 Company Update November 2014 Exhibit 99.1 NASDAQ: |
 2 Forward Looking Statement Zogenix cautions you that statements included in this presentation that are not a description of historical facts are forward-looking statements. Words such as "believes," "anticipates," "plans," "expects," "indicates," "will," "intends," "potential," "suggests," "assuming," "designed" and similar expressions are intended to identify forward-looking statements. These forward-looking statements include: the expected timing of the FDA review and action date and potential approval of the sNDA and updated labels with abuse deterrent claims for Zohydro ER; the potential to develop an abuse deterrent formulation of Zohydro ER with Altus and the timing of related studies; the potential timing of negotiating and carrying out a co-promotion agreement for Zohydro ER; the timing of the commencement of Phase 3 clinical trials for Brabafen and a multi-dose clinical and safety study and Phase 3 clinical trials for Relday and the timing of the results; the potential timing of NDA submissions for each of the abuse deterrent formulation of Zohydro ER with Altus, Brabafen and Relday and the expected timing of the FDA review and potential approval of the NDA submissions; the size of the commercial opportunity for Zohydro ER and Relday; and the ability to secure debt financing based on the non-binding term sheet. Actual results may differ from those set forth in this release due to the risks and uncertainties associated with regulatory review and approval of the sNDA, including the risk that additional information or data requests from the FDA could significantly delay the FDA’s review period; risks and uncertainties associated with the development and regulatory approval of an abuse deterrent formulation with Altus and Zogenix's reliance on Altus and its drug delivery platform in such development efforts; unexpected adverse side effects or inadequate therapeutic efficacy of the abuse deterrent formulations that could limit approval and/or commercialization; Zogenix’s dependence on the successful commercialization of Zohydro ER; Zogenix’s ability to achieve broad market acceptance and generate revenues from sales of Zohydro ER; public concern regarding the safety of drug products such as Zohydro ER and the impact of negative publicity and political influences relating to the regulation of the pain management market in general and opioids and Zohydro ER in particular; competition from other pharmaceutical or biotechnology companies; risks associated with the acquisition of Brabant and integration of Brabant's operations into Zogenix's business, including an increase in near and long-term expenditures, exposure to unknown liabilities and diversion of Zogenix's management's time and attention; Zogenix’s dependence on its contract manufacturers and its ability to ensure an adequate and continued supply of Zohydro ER to meet market demand; Zogenix’s dependence on third parties to develop an abuse deterrent formulation of Zohydro ER, Relday and Brabafen; the potential that earlier clinical trials may not be predictive of future results; Zogenix’s ability to successfully enforce its marketing exclusivities and intellectual property rights, and to defend the patents covering Zohydro ER, including the potential for Paragraph IV litigation relating to the product; the potential product liability exposure associated with pharmaceutical products such as Zohydro ER and other products Zogenix may in-license or acquire; difficulties in identifying, negotiating and carrying out a co-promotions agreement for Zohydro ER; Zogenix’s ability to fully comply with numerous federal, state and local laws and regulatory requirements that apply to its commercial activities; the risk that Zogenix will be unable to negotiate and enter into definitive agreements for a potential debt financing, on acceptable terms, or at all; Zogenix's ability to raise additional funding that it may need to continue to pursue its commercial and business development plans; and other risks detailed under “Risk Factors”and elsewhere in Zogenix’s periodic reports and other filings made with the Securities and Exchange Commission from time to time. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, and Zogenix undertakes no obligation to revise or update this presentation to reflect events or circumstances after the date hereof, except as required by law. All forward-looking statements are qualified in their entirety by this cautionary statement. This caution is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. |
 3 CHRONIC PAIN SCHIZOPHRENIA $16B Market Launch March 2014 Co-Promote Opportunity ADF potential approvals 2015 and 2016 Orphan Designation US and EU Phase 3 2Q2015 $29B+ Market Worldwide Market Rights Phase 1b Jan 2015 Co-Development/ Commercialization Opportunities DRAVET SYNDROME (low-dose fenfluramine) Developing and Commercializing High Value CNS Products Dravet syndrome Product Candidate With Long-Term Data Subcutaneous Once-Monthly Antipsychotic Oral, 12-Hour Extended Release Hydrocodone without Acetaminophen CHRONIC PAIN * *12 months ended March 2014 Retail/Institutional Dollars Symphony Health Systems (SHS) Source ® April 2013 – March 2014. Pain - Retail TRx Dollars. Antipsychotic - Retail and Institution Dollars |
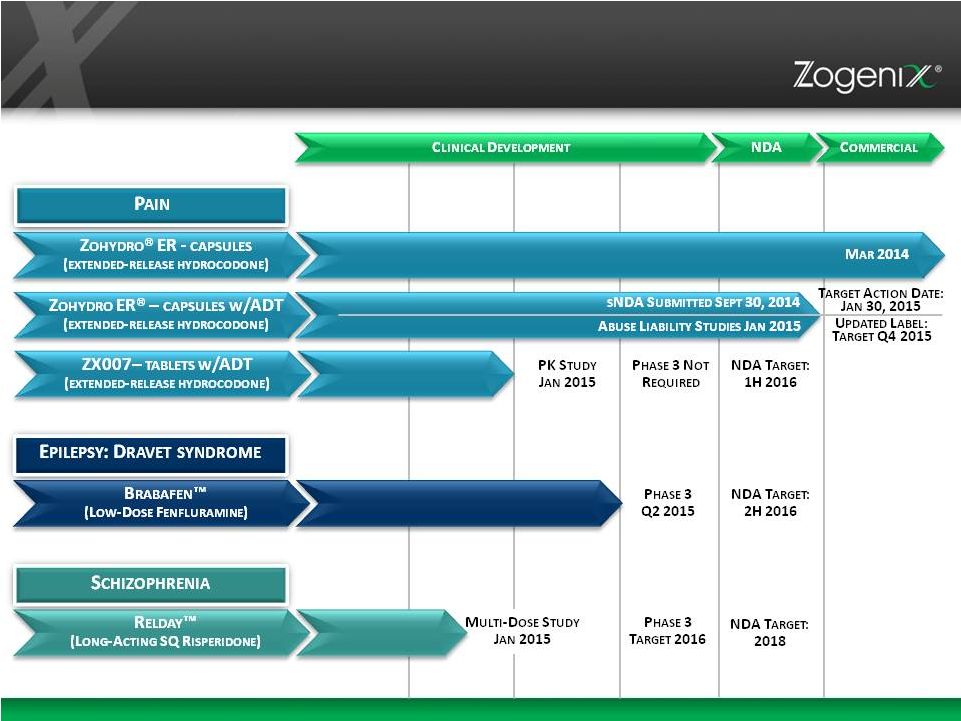 CNS Pipeline – Innovative Development and Regulatory Timelines 4 |
 5 Oral, Extended Release Hydrocodone Acetaminophen -Free Hydrocodone Product FDA Approved October 2013 Launched March 2014 |
 6 Zohydro ER Commercial Update • 15,134 Prescriptions in 3Q 2014, up 47% over 2Q 2014 • 31,000 Prescriptions and 4000 Prescribers from Launch Through October 24, 2014 • 8 out of 10 Prescriptions Approved by Payers • Average NSP $240 • >7000 Pharmacies Stocking Zohydro ER METRICS • sNDA for modified formulation with abuse deterrent properties filed September 30, 2014 – target action date January 30, 2015 • Hydrocodone Combination Products Rescheduled from Schedule III to Schedule II (October 2014) • Dr. Janet Woodcock, FDA, BioCentury TV, November 9, 2014: ADT is in “infancy” stage and rather than forcing conventional opioids off the market, FDA is focused on getting more abuse- deterrent products approved MARKET DYNAMICS Symphony Health Solutions, Source® PHAST Prescription Monthly, July – September 2014 Symphony Health Solutions, Source® PHAST Prescription Weekly, February 2014 - October 24, 2014 |
 7 Zohydro ER Fills an Important Medical Need • About 5% of patients on hydrocodone combination products are using it to manage chronic pain – Unintentional acetaminophen overdose* is the leading cause of acute liver failure – No extended-release version of hydrocodone had been available • Zohydro ER is a more appropriate treatment than IR-HC/APAP for severe chronic pain patients requiring around-the-clock opioid therapy – No acetaminophen – Extended-release allows more consistent pain relief – 12-hour dosing reduces total pills in 30-day prescription – Simple transition from IR-HC to ER-HC vs. switching to a different type of extended-release molecule such morphine or oxycodone *Blieden M, et.al. Expert Rev Clin Pharmacol. 2014 |
 8 SaferLock ™ Locking Bottle for safe storage Zogenix Commitment to Responsible Commercialization • Broad, Integrated Educational Resources for Patients, Physicians and Pharmacists EDUCATION Commercial Activity Focused On Select Physicians – High Decile ER/LA Opioid Prescribers (~60% of ER/LA opioid market) MARKETING • Certification of Zogenix Territory Representatives • Incentive Compensation Based On Achieving Safe Use Goals SALES • Surveillance and Monitoring to Detect Potential Misuse, Abuse and Diversion MONITORING • Distribution of Safe Storage Mechanisms STORAGE • Abuse Deterrent Formulations ADF |
 9 • Addition of Inert Beads That Form a Viscous Gel When Crushed and Dissolved in Aqueous Solvents – Designed to be injection and insufflation resistant • Supplemental Application to Approved NDA – Formulation submission September 2014 – Further submission with Abuse Liability Studies (additional AD label claims) H1 2015 • Hardened Tablet to Avoid Crushing and Chewing • Maintains Zohydro ER’s Unique Release Profile • Extraction Resistant • Insufflation and Injection Resistant • Separate NDA - Referencing Current Approved NDA for Safety and Efficacy – No Phase 3 Studies Required – Submission H2 2016 • Extended IP Runway (>2030) Abuse Deterrent (AD) Formulations In Development TWO Simultaneous APPROACHES Modification of Approved Capsule Formulation Development of Novel Tablet Formulation with Altus |
 10 Low-dose fenfluramine for the treatment of Dravet syndrome, a rare and catastrophic form of intractable epilepsy that begins in infancy Phase 3 Candidate Orphan drug designation in Europe and U.S. BRABAFEN ™ |
 11 Brabant Pharma Acquisition • Acquired October 27, 2014 for $20 million in cash and $15 million in stock plus potential future milestone and royalty payments • Worldwide development and commercialization rights to Brabafen TM , low- dose fenfluramine, for the treatment of Dravet syndrome, a rare and catastrophic form of intractable epilepsy that begins in infancy – Orphan drug designation in Europe and the U.S. – Phase 3 clinical studies of Brabafen planned for second quarter of 2015 – Longest duration data (26 years) in treatment of Dravet syndrome • Signed a non-binding term sheet for a $20 million term loan plus an additional $4 million in a revolving line of credit – Potential financing expected to help fund R&D expenses for Brabafen |
 12 Dravet Syndrome – Rare And Severe Condition • Orphan disease with incidence between 1:20,000 and 1:40,000 live births • Genetic condition associated with mutations of the SCN1A gene – Severe, frequent seizures; drug-refractory – Cognitive decline; increasing motor problems – Children do not outgrow condition, require constant care and supervision • Significant mortality (approx. 15 - 20%), more frequent during childhood – Patients often suffer from Status Epilepticus (severe, continuous seizures) and are at increased risk of Sudden Unexpected Death in Epilepsy (SUDEP) • Current treatments are limited and inadequate – No US product FDA-approved for Dravet syndrome – Stiripentol is the only drug specifically approved in EU for Dravet syndrome and has limited and restricted use |
 13 Long Term Results of Brabafen In Dravet Syndrome Showed Significant Reduction in Seizures (n=15) Brabafen Responder Analysis 100% 80% 60% 40% 20% 0% >50% >70% >90% Seizure Free FDA Assessment of Primary Efficacy In Dravet syndrome 93% N=14 93% N=14 87% N=12 67% N=10 • Average length of treatment >12 years; longest duration = 26 years – Mean age at last evaluation = 19 yr (3-35 yr) • Fenfluramine (5-10 mg BID) added on to existing antiepileptic drug therapy • Seizure diary follow-up twice a year; clinical & echocardiogram every year • Efficacy: – Evidence that seizures in some patients ceased within days of starting treatment – On average patients were seizure free for approximately 6 years (1-19 years) • Safety – Mild/transient side effects – loss of appetite, fatigue, somnolence – Evidence of clinically insignificant heart valve thickening in 3/15 patients (resolved in 1 patient) • Not progressive – all patients remain in study – No pulmonary hypertension |
 14 Remaining Development Plan for Brabafen • Agreed upon pre-clinical and clinical requirements from FDA and EMA • Phase 3 pivotal clinical study protocol – Two placebo controlled studies (United States and Europe) – 40-60 patients each – Add-on therapy – 12-week evaluation period – Evaluating number of seizures compared to baseline • Phase 3 initiation planned for second quarter 2015 – Phase 3 data in early 2016 – U.S. potential approval in late 2016 – European potential approval in 2017 |
 15 Once-Monthly Risperidone Phase 1b – Jan 2015 Proof of Concept Established Schizophrenia With A Unique PK Profile, Available as a Subcutaneous Injection for Treatment of |
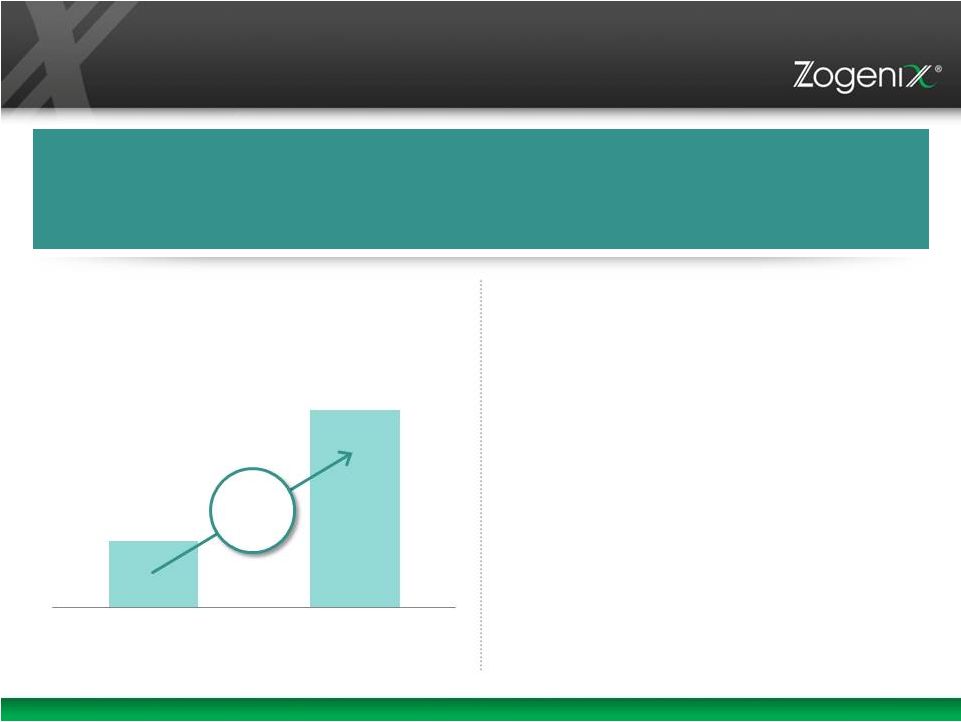 16 • Highly Concentrated U.S. Market with 11,000 Psychiatrists Prescribing Long- Acting Injectable Antipsychotics • Growing U.S. and EU Market for LAI Products • Risperdal Consta, 100mg/Month >$1,200 in U.S. $2.8 Billion Market in U.S.+ Europe in 2013 Long Acting Injections (LAIs): A Highly Attractive Market Source: Credit Suisse, May 2014 Note: Includes 2013 WAC $ from SHS JNJ reported ex-U.S. sales. U.S. LAI Sales (Schizophrenia) $ Millions $400M $1.2B 2007 2013 20% CAGR Further Clinical Evidence of Earlier LAI Usage Will Likely Drive Additional Upside |
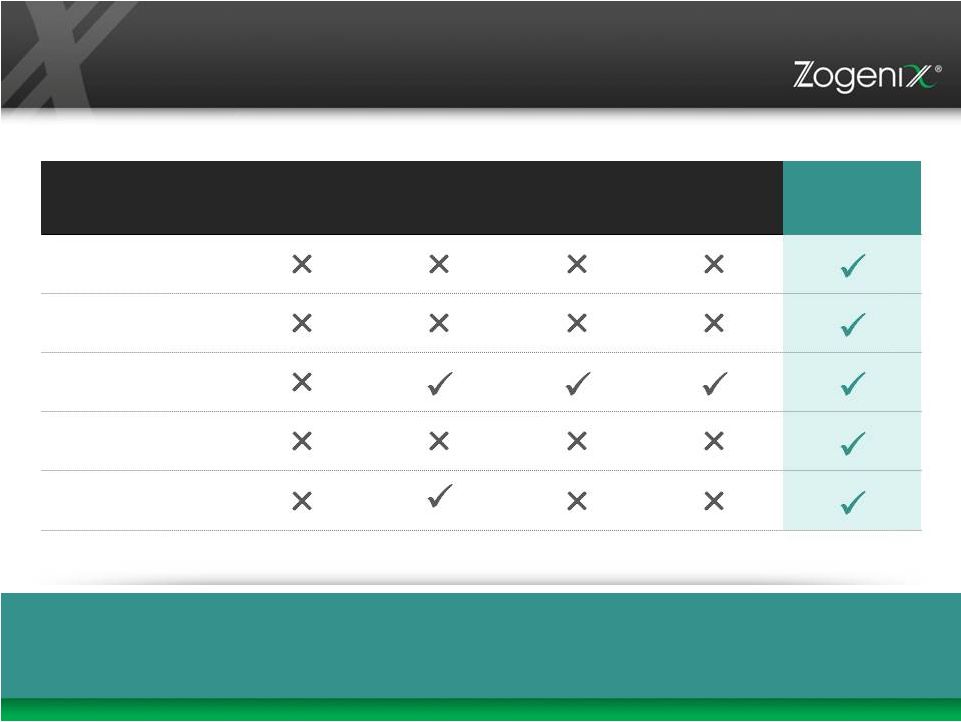 17 RELDAY Product Profile vs. Current LAI Atypical Antipsychotics (AA) KEY ATTRIBUTES FROM PHYSICIAN RESEARCH RISPERDAL CONSTA (RISPERIDONE) INVEGA SUSTENNA (PALIPERIDONE) ZYPREXA RELPREVV (OLANZAPINE) ABILIFY MAINTENA (ARIPIPRAZOLE) RELDAY (RISPERIDONE) Therapeutic Levels on Day 1 No Loading Dose/ Oral Supplementation 1X Monthly Subcutaneous No Reconstitution Substantial Market Research Supports Key Features Physicians Want 17 |
 18 RELDAY Clinical Program Status • Established in the First IND Study • Subjects with Schizophrenia (n=40) • Pharmacokinetics (PK) – Therapeutic Levels within 8 hours of Dosing – First Day Levels Consistent with Comparable Oral Dose – Duration Consistent with Once-monthly Dosing – Dose-Proportionality Across the Entire Therapeutic Range • Safety /Tolerability – No Unique Safety or Tolerability Issues with SC Dosing – Injection Site Reactions Were Mild and Spontaneously Resolved REGULATORY STRATEGY PROOF OF CONCEPT • Confirmed 505(b)(2) NDA in US • Abridged Hybrid MAA likely in EU |
 19 RELDAY Clinical Study Design • Starts in January 2015, Top-Line Results Q3 2015 • ROW Partnering With Data (H2 2015) • End of Phase II Meeting (Q3/Q4 2015) • Begin Phase III Trial (H1 2016) OVERVIEW INJECTIONS TIMING NEXT STEPS (Target Dates) • Relday Patients Will Receive 4 Injections Over 16 Weeks • Consta Patients Will Receive 5 Injections (Required to Reach Steady-State) • Open-Label, PK and Safety, in Patients with Chronic, Stable Schizophrenia or Schizoaffective Disorder (n=40) • Patients Randomized into One of Four Cohorts: – Cohort 1: Relday, 60mg Once-Monthly SQ Depot Injection – Cohort 2: Relday, 90mg Once-Monthly SQ Depot Injection – Cohort 3: Relday, 120mg Once-Monthly SQ Depot Injection – Cohort 4: Risperdal ® Consta ® - 25 mg IM Bi-Weekly Depot Injection |
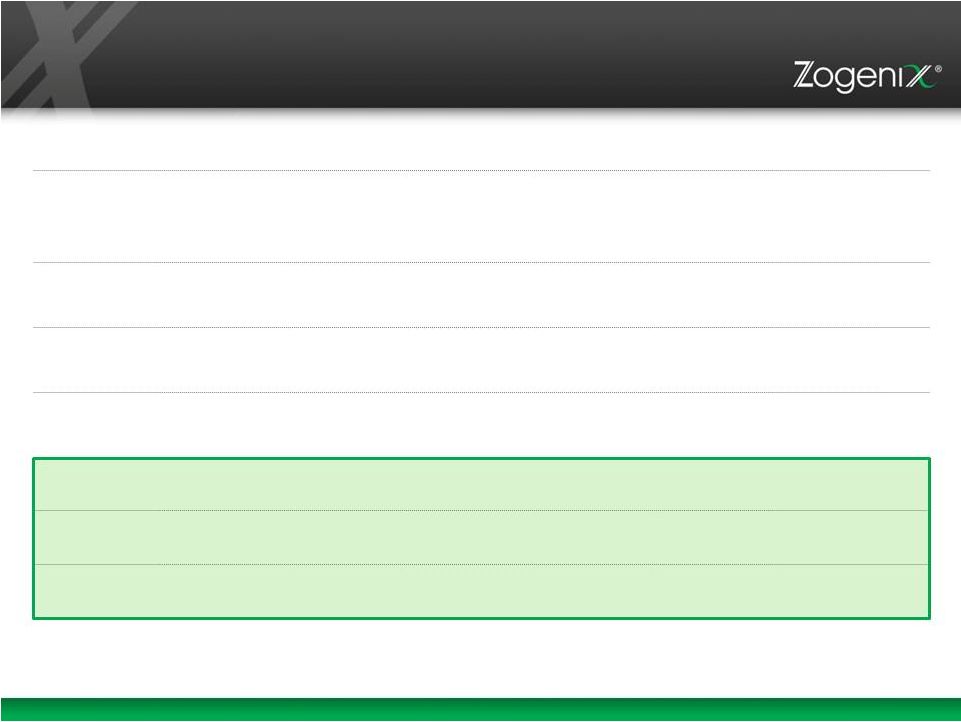 20 Financial Highlights Q3 2014 Total Revenue (Zohydro ER revenue of $4.1M) $8.8M Market Capitalization (8-6-14 ~139,539,151 Shares) $190M Q3 2014 Cash Balance (1) $50.5M and $8.5M (escrow) $0M Common Stock Outstanding 153,040,066 Fully Diluted Shares (3) 185,657,387 Employees 220 (1) Excludes $24m in debt facility secured with Brabant acquisition and $13.5m related to the Purdue waiver exchange and Teva right of reference agreement (2) Excludes $7m working capital advance from Endo International and pending $24m in debt facility (3) Fully diluted shares as of 10/17/14 include options to purchase 16,893,600 shares and warrants to purchase 15,723,721 shares with weighted average exercise prices of $2.67 and $2.73 respectively Debt (2) |
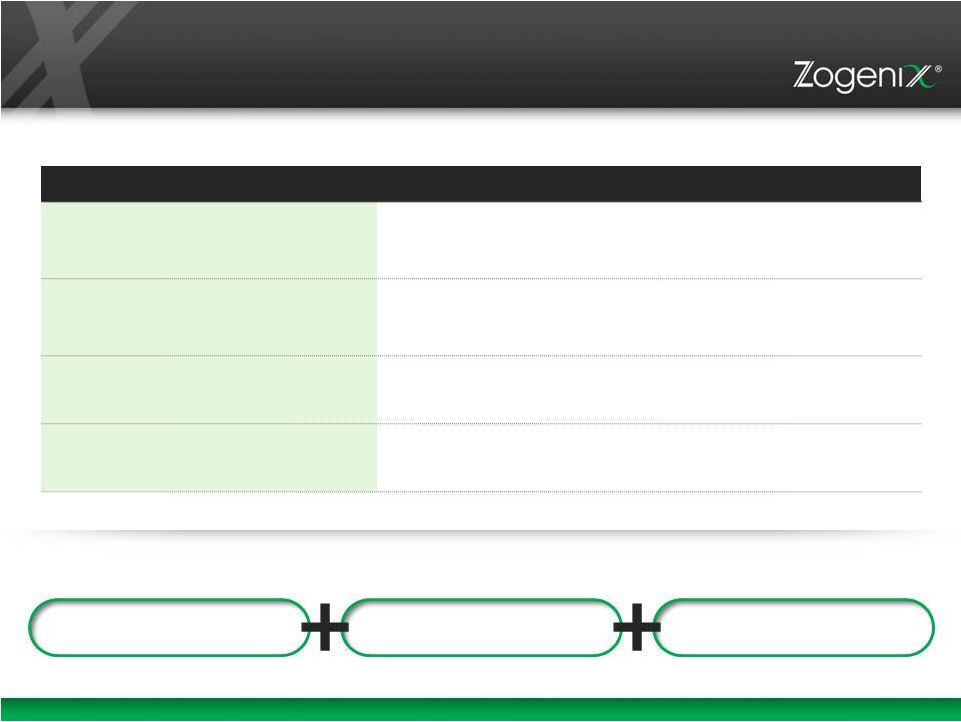 21 Experienced Management Team INDUSTRY EXPERIENCE Roger Hawley Chief Executive Officer Co-Founder 27 Glaxo, GSK, Elan, InterMune Stephen Farr, Ph.D. President Co-Founder 21 Aradigm, Cardiff University Ann Rhoads Chief Financial Officer 20 Premier, Sprout, DLJ, Bain & Co, Merrill Lynch Bradley Galer, M.D. Chief Medical Officer 19 Nuvo Research, Endo MANUFACTURING DEVELOPMENT COMMERCIALIZATION History of Successful Execution |
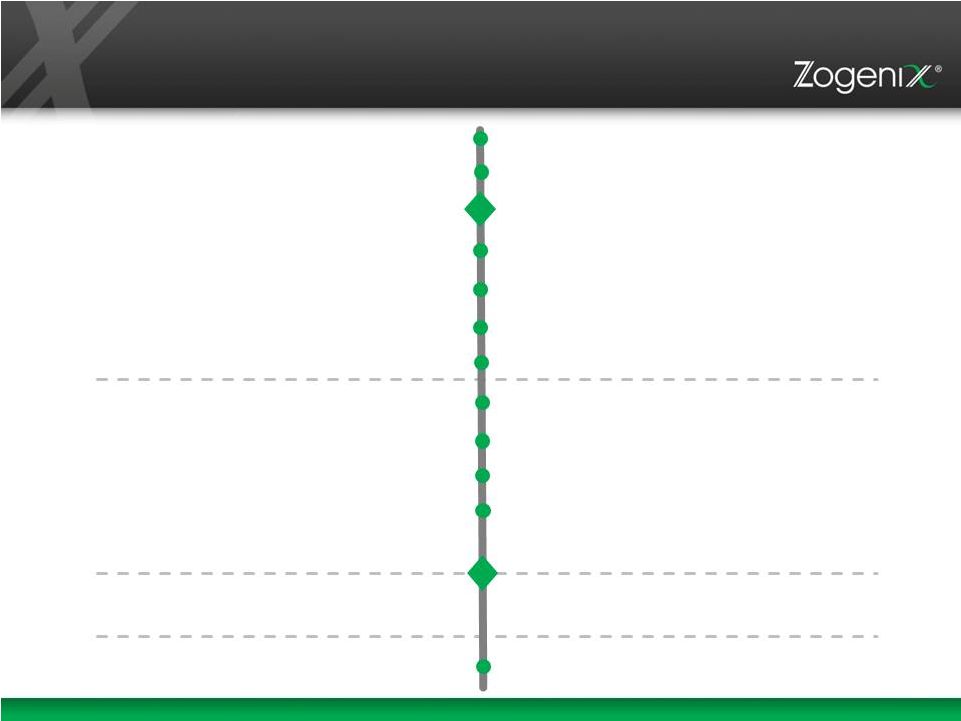 22 Significant Cadence of Potential Value Creating Milestones Through 2018 2015 Relday Multi-Dose Clinical Trial Begins Abuse Liability Studies Zohydro ER AD capsules Begins Potential Zohydro ER Co-Promotion Brabafen Phase 3 Clinical Trial Begins Relday Multi-Dose Trial Data Results NDA for Altus ADF Zohydro ER Brabafen Phase 3 Data Relday Phase 3 Clinical Trial Brabafen EU Approval & Altus ADF Zohydro ER 2016 2017 2018 sNDA Potential Approval Zohydro ER (AD capsules) Updated Zohydro ER label with AD claims NDA Potential Approval Zohydro ER (AD tablets) NDA Potential Approval Brabafen Relday Target NDA Brabafen Target NDA |
 |