Exhibit (a)(5)(Q)

Acquisition of OSI Pharmaceuticals, Inc.
Becoming a Global Category Leader in Oncology-May 17, 2010

Table of Contents
Transaction Summary
Strategic Rationale
Overview of OSI Pharmaceuticals
Financial Impact
2 |
|

I. Transaction Summary
3 |
|

Transaction Summary
Purchase price: $57.50 per share in cash (55% premium to the closing price on February 26, 2010, the last trading day before the announcement of tender offer)
Acquisition amount: Approximately $4.0 Billion (Fully diluted basis)
Tender offer period: Expires no later than 10 business days after the amendment to the Schedule TO is filed (which is expected to be filed on or before May 21st), unless extended
Financing: Fully financed with cash and cash equivalents on Astellas’ balance sheet
The acquisition is unanimously approved by the Board of Directors of both Astellas and OSI
4 |
|

II. Strategic Rationale
5 |
|

Business Model of Astellas
Global Category Leader (GCL)
A “GCL”, by providing value-added products “globally”, takes over the position of “leader” in a “category” where high unmet medical needs exist and a high degree of expertise is required.
6 |
|
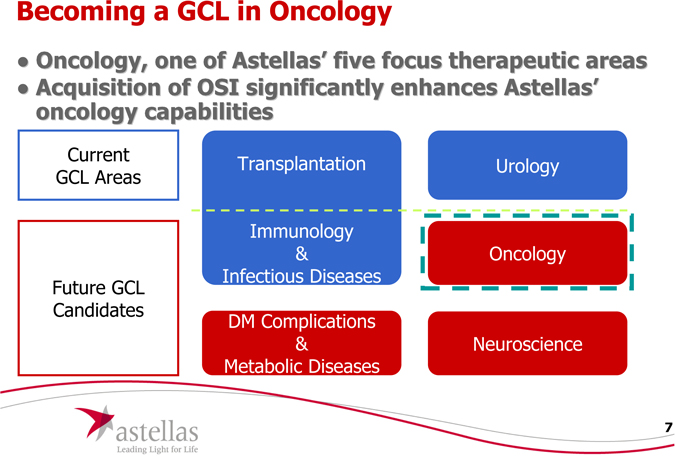
Becoming a GCL in Oncology
Oncology, one of Astellas’ five focus therapeutic areas
Acquisition of OSI significantly enhances Astellas’ oncology capabilities
Current GCL Areas
Future GCL Candidates
Transplantation
Immunology
& Infectious Diseases
Urology
Oncology
Neuroscience
7 |
|

Strategic Rationale for OSI Acquisition
Accelerates development of Astellas’ oncology franchise
Acquire fully integrated oncology capabilities in the U.S. including discovery, development and commercialization
Expand clinical stage oncology pipeline
Access to small molecule discovery research platform in oncology
Existing revenue stream and improved profitability
Marketed blockbuster product with significant growth and late stage pipeline
Outstanding partnership with Roche/Genentech Growing DPP-IV royalty income
8 |
|
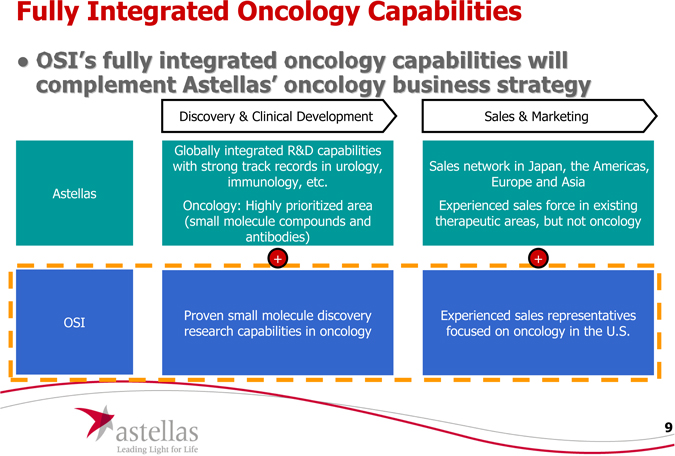
Fully Integrated Oncology Capabilities
OSI’s fully integrated oncology capabilities will complement Astellas’ oncology business strategy
Discovery & Clinical Development
Sales & Marketing
Astellas
Globally integrated R&D capabilities
with strong track records in urology,
immunology, etc.
Oncology: Highly prioritized area
(small molecule compounds and
antibodies)
Sales network in Japan, the Americas,
Europe and Asia
Experienced sales force in existing therapeutic areas, but not oncology
OSI
P
roven small molecule discovery research capabilities in oncology
Experienced sales representatives focused on oncology in the U.S.
9
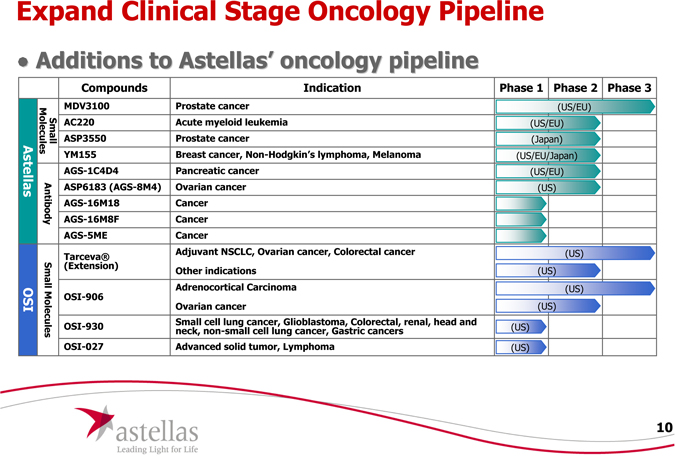
Expand Clinical Stage Oncology Pipeline
Additions to Astellas’ oncology pipeline
Abtibody
Compounds Indication Phase 1 Phase 2 Phase 3
MDV3100 Prostate cancer(US/EU)
AC220 Acute myeloid leukemia(US/EU)
ASP3550 Prostate cancer(Japan)
YM155 Breast cancer, Non-Hodgkin’s lymphoma, Melanoma(US/EU/Japan)
AGS-1C4D4 Pancreatic cancer(US/EU)
ASP6183 (AGS-8M4) Ovarian cancer(US)
AGS-16M18 Cancer
AGS-16M8F Cancer
AGS-5ME Cancer
Tarceva® Adjuvant NSCLC, Ovarian cancer, Colorectal cancer(us)
(Extension) Other indications(US)
OSI-906 Adrenocortical Carcinoma(US)
OSI-906 Ovarian cancer(US)
OSI-930 Small cell lung cancer, Glioblastoma, Colorectal, renal, head and neck, non-small cell lung cancer, Gastric cancers
OSI-027 Advanced solid tumor, Lymphoma
10
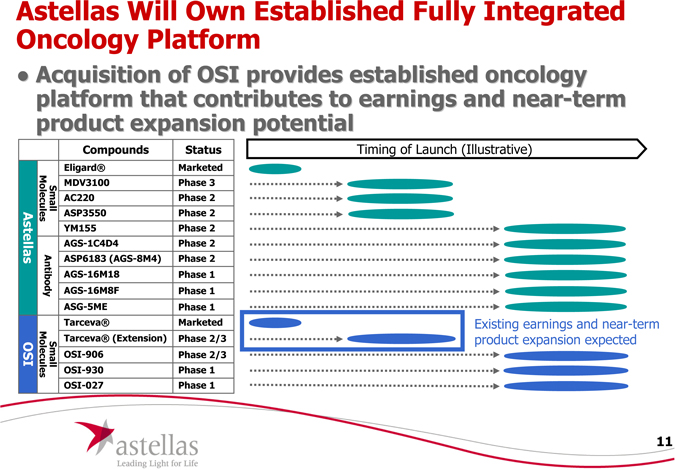
Astellas Will Own Established Fully Integrated Oncology Platform
Acquisition of OSI provides established oncology platform that contributes to earnings and near-term product expansion potential
Compounds Status
Eligard® Marketed
Sma Mole MDV3100 Phase 3
AC220 Phase 2
Small Molecules ASP3550 Phase 2
YM155 Phase 2
AGS-1C4D4 Phase 2
ASP6183 (AGS-8M4) Phase 2
AGS-16M18 Phase 1
AGS-16M8F Phase 1
ASG-5ME Phase 1
Tarceva® Marketed
Tarceva® (Extension) Phase 2/3
ma ecu OSI-906 Phase 2/3
OSI-930 Phase 1
OSI-027 Phase 1
Timing of Launch (Illustrative)
Existing earnings and near-term product expansion expected
11

III. Overview of OSI Pharmaceuticals
12

Overview of OSI Pharmaceuticals
Established in 1983 in the U.S., listed on NASDAQ
Based in Melville, NY with facilities in NY, CO, NJ in the U.S. and UK (U.S. facilities consolidating in Ardsley, NY by the end of 2010)
Company with unique, profitable, and fully integrated operations in discovery, development and commercialization of innovative molecular targeted therapies for the treatment of oncology, diabetes and obesity
Fully integrated oncology platform
Marketed lead product: Tarceva® (er/otinib, HER1/EGFR inhibitor)
Partnership with Roche/Genentech
Promising clinical pipeline in oncology and diabetes/obesity Robust financial performance with strong growth
Number of full-time employees: 512 (As of December 31, 2009)
13
Established Oncology Business Platform in the U.S.
A profitable and growing oncology business anchored around a blockbuster product with considerable remaining patent life (Tarceva®)
An experienced and capable commercial organization
A proven track record of success based on an established high quality drug discovery infrastructure and know-how
Discovery & Clinical Development
Sales & Marketing
14

Tarceva®—A Blockbuster Oncology Drug
One of the most successful oncology products in terms of patients treated in U.S. history
Achieved ~$1.2billion of worldwide sales in 2009
Approved for sale in 109 countries for treatment of advanced
non-small cell lung cancer (NSCLC) Strategically valuable long-term cash flows throughout
exclusivity period
Life cycle plan
Life cycle plan expected to yield multiple data and label
expansion opportunities EGFR-mutation and ovarian cancer milestones expected over
the next 15 months Additional opportunities in liver cancer and multiple molecular
targeted therapy combination trials
15
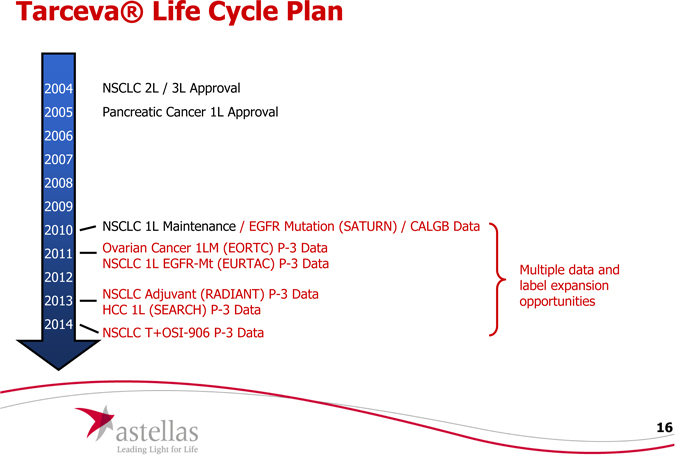
Tarcevafft Life Cvrle Plan
2004 2005 2006 2007 2008 2009 2010
NSCLC 2L / 3L Approval Pancreatic Cancer 1L Approval
NSCLC 1L Maintenance / EGFR Mutation (SATURN) / CALGB Data
Ovarian Cancer 1LM (EORTC) P-3 Data
NSCLC 1L EGFR-Mt (EURTAC) P-3 Data Multiple data and
NSCLC Adjuvant (RADIANT) P-3 Data
HCC 1L (SEARCH) P-3 Data opportunities
NSCLC T+OSI-906 P-3 Data
16

Clinical Pipeline
Compounds Characteristics Indication Status
Oncology
Tarceva® (erlotinib) (Extension) HERl/EGFR tyrosine kinase inhibitor Adjuvant NSCLC, Ovarian cancer, Colorectal cancer Other indications Phase 3 Phase 2
OSI-906 IGF-1R/IR tyrosine kinase inhibitor Adrenocortical Carcinoma Ovarian cancer Phase 3 Phase 2
OSI-930
(Out-licensed to Simcere Pharma in China) c-kit/VEGFR-2 tyrosine kinase inhibitor Small cell lung cancer, glioblastoma, Colorectal, renal, head and neck, non-small celt lung cancer, Gastric cancers Phase 1
OSI-027 mTOR kinase inhibitor Advanced solid tumor, lymphoma Phase 1
Diabetes / Obesity
PSN821 GPR119 agonist Type 2 diabetes / obesity Phase 2
PSN010
(Out-licensed to Eli Lilly) Glucokinase activator Type 2 diabetes Phase 2
17

Discovery Capabilities
Focus on novel targets with biological validation
Has multiple small molecule based drug discovery programs in
both oncology and diabetes / obesity that employ both
traditional and novel assays Drug targets are predominantly kinase pathways that are
either clinically validated or validated through extensive
elucidation of the biology and role in disease Competitive edge is targeting multiple pathways and in
selectivity of drug candidates / Novel cell based assay (EMT—Epithelial-Mesenchymal
Transition) is unique in the industry
Oncology research in the U.S., diabetes / obesity research through a subsidiary in the UK
18
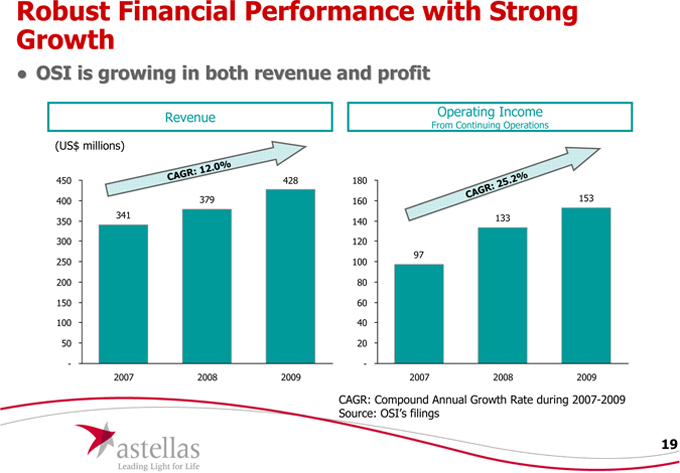
Robust Financial Performance with Strong Growth
• OSI is growing in both revenue and profit
Revenue
Operating Income
From Continuing Operations
(US$ millions)
450 -
400 -
350 -
300 -
250 -
200 -
150 -
100 -
50 -
341
CAGR: 12.0%
379
428
180
160
140
120
100
80
60
40
20
97
133
153
2007
2008
2009
2007
2008
2009
CAGR: Compound Annual Growth Rate during 2007-2009 Source: OSI’s filings
19

IV. Financial Impact
20
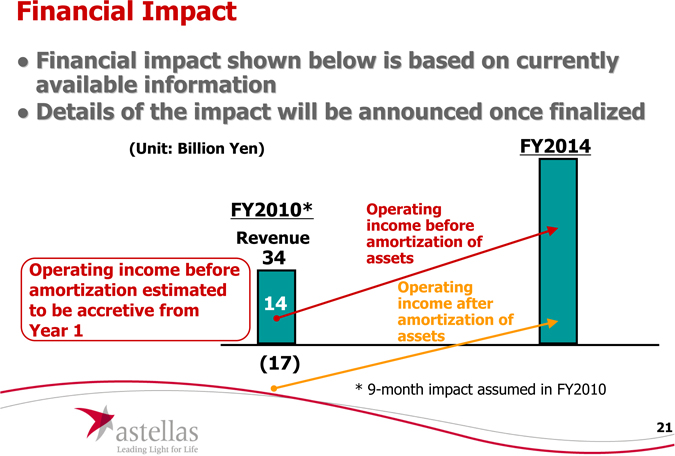
Financial Impact
Financial impact shown below is based on currently available information
Details of the impact will be announced once finalized
(Unit: Billion Yen)
FY2014
FY2010* Revenue
34
Operating income before amortization estimated to be accretive from
year 1
Operating income before amortization of assets
Operating income after amortization of assets
(17) |
|
* |
| 9-month impact assumed in FY2010 |
21

Additional Information
Further details related to this announcement can be found on www.oncologyleader.com
Media Contacts
Brunswick New York +1 212 333 3810 Stan Neve Sarah Lubman
Brunswick Hong Kong +852 9850 5033 Joseph Lo
Information Agent Georgeson
+ 1 212 440 9872 Thomas Gardiner
About Astellas
Astellas Pharma Inc., located in Tokyo, Japan, is a pharmaceutical company dedicated to improving the health of people around the world through the provision of innovative and reliable Pharmaceuticals. Astellas has approximately 15,000 employees worldwide. The organization is committed to becoming a global category leader in urology, immunology & infectious diseases, neuroscience, DM complications & metabolic diseases and oncology. For more information on Astellas Pharma Inc., please visit our website at http://astellas.com/en.
About OSI Pharmaceuticals
OSI Pharmaceuticals, Inc. is committed to “shaping medicine and changing lives” by discovering, developing and commercializing high-quality, novel and differentiated targeted medicines designed to extend life and improve the quality of life for patients with cancer and diabetes/obesity. For additional information about OSI, please visit http://www.osip.com.
Important additional information
This press release is for informational purposes only and does not constitute an offer to purchase or a solicitation of an offer to sell OSI’s common stock. The tender offer (“Tender Offer”) is being made pursuant to a tender offer statement on Schedule TO (including the offer to purchase, letter of transmittal and other related tender offer materials) initially filed by Astellas with the Securities and Exchange Commission (the “SEC”) on March 2, 2010. These materials, as they may be amended from time to time, contain important information, including the terms and conditions of the offer, that should be read carefully before any decision is made with respect to the Tender Offer. Investors and shareholders can obtain a free copy of these materials and other documents filed by Astellas with the SEC at the website maintained by the SEC at www.sec.gov. The Tender Offer materials may also be obtained for free by contacting the information agent for the tender offer, Georgeson Inc. at (212) 440-9800 for banks and brokers and at (800) 213-0473 for persons other than banks and brokers.
OSI’s stockholders should read the company’s solicitation/recommendation statement on schedule 14D-9, which was initially filed with the SEC on March 15, 2010, and any amendments or supplements thereto. The company’s solicitation/recommendation statement will set forth the reasons for the recommendation of the OSI’s board and related information. The solicitation/recommendation statement and other public filings made from time to time by OSI with the SEC are available without charge from the SEC’s website at www.sec.gov, at OSI’s website at www.osip.com or from OSI’s information agent, MacKenzie Partners, Inc. by calling 800-322-2885 (toll free) or 212-929-5500 or by emailing osipharma@mackenziepartners.com.
Statement of Cautionary Factors
This document contains certain forward-looking statements. These forward-looking statements may be identified by words such as ‘believes’, ‘expects’, ‘anticipates’, ‘projects’, ‘intends’, ‘should’, ‘seeks’, ‘estimates’, ‘future’ or similar expressions or by discussion of, among other things, strategy, goals, plans or intentions. Various factors may cause actual results to differ materially in the future from those reflected in forward-looking statements contained in this document, among others: (1) pricing and product initiatives of competitors; (2) legislative and regulatory developments and economic conditions; (3) delay or inability in obtaining regulatory approvals or bringing products to market; (4) fluctuations in currency exchange rates and general financial market conditions; (5) uncertainties in the discovery, development or marketing of new products or new uses of existing products, including without limitation negative results of clinical trials or research projects, unexpected side-effects of pipeline or marketed products; (6) increased government pricing pressures; (7) interruptions in production; (8) loss of or inability to obtain adequate protection for intellectual property rights: (9) litigation; (10) loss of key executives or other employees; and (11) adverse publicity and news coverage. The statement regarding earnings growth is not a profit forecast and should not be interpreted to mean that Astellas’ earnings or earnings per snare for any current or future period will necessarily match or exceed the historical published earnings or earnings per share of Astellas.