Exhibit 4.7.10 - Appendix 5 - Bidding Document 2
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Part 1 Tender | ||
第 1 部分
Part 1
投 标 书
Tender
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
目 录
| 1. 报价书 | 2 |
| 1.1. 法定代表人资格证明书 | 2 |
| 1.2. 法人授权委托书 | 3 |
| 1.3. 授权代表人身份证复印件 | 4 |
| 1.4. 投标书 | 5 |
| 1.5. 报价一览表 | 6 |
2.分项报价清单 | 8 |
| 2.1. 压缩空气系统材料清单及报价 | 8 |
| 2.2. 氮气管道系统材料清单及报价 | 11 |
| 2.3. 氧气管道系统材料清单及报价 | 13 |
| 2.4. 二氧化碳气体管道系统材料清单及报价 | 15 |
| 2.5. 焊点费 | 17 |
| 2.6. 系统所用辅材数量清单 | 18 |
| 2.7. 验证服务报价 | 19 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
1. 报价书
1.1. 法定代表人资格证明书
法定代表人资格证明书
单位名称:上海朗脉科技有限公司
单位地址:上海市闵行区罗阳路168 号A 座1 层
经营期限:2007年7 月26 日到2017 年7 月25 日
姓名:王伟
性 别: 男
年 龄: 36
职务:法人代表
王伟先生系上海朗脉科技有限公司 的法定代表人,签署本招标项目的投标文件,进行合同谈判,签署合同和处理与之有关的一切事务。
特此证明。
投标单位(盖章): 上海朗脉科技有限公司
日期:2009-1-15
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
1.2. 法人授权委托书
法人授权委托书
沈阳三生制药有限责任公司:
本授权委托书声明:我王伟先生系上海市闵行区罗阳路168号A 座101室的上海朗脉科技有限公司的法定代表人,现代表公司授权委托下面签字的上海朗脉科技有限公司的销售经理张松为本公司的合法代理人,代表本公司参加沈阳三生制药有限责任公司组织的压缩空气及工艺气体系统项目的投标活动。代理人在本次投标中所签署的一切文件和处理与之相关的一切事务,我均予以承认。
代理人无转委权
本授权于2009 年1 月15 日签字生效,特此声明。
代理人:张松 性别:男 年龄:35
单位:上海朗脉科技有限公司 职务:销售经理
地址:上海市闵行区罗阳路168 号A 座101 室
投标人:(公章)上海朗脉科技有限公司
法定代表人:(印鉴)
2009 年1 月15 日
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
1.3. 授权代表人身份证复印件

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
1.4. 投标书
投标书
致沈阳三生制药有限责任公司:
我们收到贵方压缩空气及工艺气体系统 招标文件,经详细研究,我们决定参加该项目投标。并授权 张松销售经理 , 全权代表我投标方提交下述文件正本一份,副本叁份。并同时宣布愿意遵守下列条款:
1、承认和愿意按照招标文件中的各项规定和要求,提供压缩空气及工艺气体系统的供货、安装、调试及维保等。
2、投标总报价 1,322,028.71 元,供货周期承诺 50 日历天,安装周期承诺 60 日历天,安装质量承诺 符合GMP 要求。
3、愿意按照《合同法》和《中华人民共和国招标投标法》履行自己的责任和义务。
4、如果我们投标函被接受,我们将履行招标文件中规定的每一项要求,按期、按质、按量完成任务。
5、我们愿意提供招标方在招标文件中要求的所有资料。
6、我们理解,最低报价不是中标的唯一条件。
7、我们同意按招标文件规定,交纳投标保证金,遵守有关招标的各项规定。
8、我方的投标书在开标后60 天内有效。
9、与本投标有关的一切往来通讯请寄:
地址: 上海市闵行区罗阳路168 号A 座101 室邮编:201100
电话: 021-54306717/ 13901375676 传真: 021-54306718
投标方(公章): 上海朗脉科技有限公司
授权代表人(签字或盖章):
日期:
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 �� F: 021 54306718 | 第5页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
1.5. 报价一览表
Item 序号 | Scope of Supply 供货范围 | Price (RMB) 价格 |
| 1 | Price Breakdown for CA Distribution System 压缩空气系统材料清单及报价 | 248,861.78 |
| 2 | Price Breakdown for N2 Distribution System 氮气系统材料清单及报价 | 64,492.78 |
| 3 | Price Breakdown for O2 Distribution System 氧气系统材料清单及报价 | 54,806.58 |
| 4 | Price Breakdown for CO2 Distribution System 二氧化碳系统材料清单及报价 | 54,806.58 |
| 5 | Piping surport 辅助材料,洁净区用不锈钢支架,非洁净区用镀锌角铁 | 143,065.00 |
Subtotal 1 for Material 材料费用 | 566,032.72 | |
| 6 | Welding Point Cost 焊点费用 | 259,890.00 |
| 7 | Cost of Welding Point marking (100% of total welding point ) 20RMB /point 焊点刻码费用,100%自动焊点刻码,20 元/焊点 | 31,660.00 |
| 8 | Endoscopic Check ( 20% of total welding point ) 120 RMB /point 内窥镜检测,自动焊点的20% 取样,120 元/焊点 | 37,992.00 |
| 9 | X-ray Check ( 100% of total hand-welding point,all welded samples ) 180 RMB /point X-光检测,(100% 手工焊点,所有焊样),180 元/焊点, | 32,400.00 |
| 10 | Insurance and Transportation 保险和运输 | 10,000.00 |
Subtotal 2 for Piping fabrication 管道装配 | 371,942.00 | |
| 11 | Detail Design, Shop Drawings and As-built Drawings 详细设计,施工图纸和竣工文件 | 30,000.00 |
| 12 | Quotation For Validation Activities 验证服务报价 | 160,000.00 |
| 13 | COMPANY Management & Supervision 公司管理&监督 | 100,000.00 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
| 14 | Project Management & Supervision 项目管理&监督 | 50,000.00 |
Subtotal 3 for Project Service 项目服务 | 340,000.00 | |
Subtotal 1, 2, 3, 合计 | 1,277,974.72 | |
Tax 税金(主材部分开增值税,其他开工程发票) | 44,053.99 | |
Total Price 总价(元) | 1,322,028.71 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
2.分项报价清单
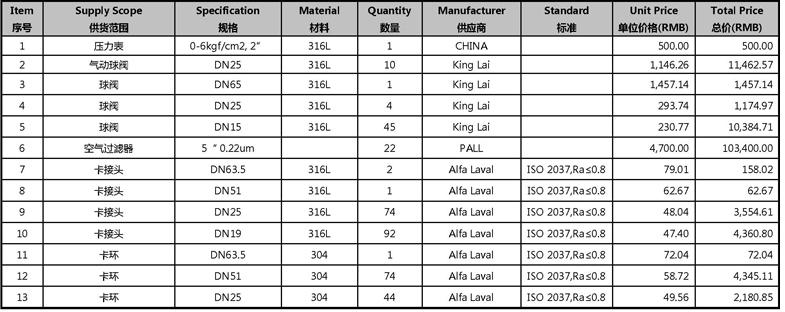
2.1. 压缩空气系统材料清单及报价
Price Breakdown for CA Distribution System
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) |
| 14 | 卡环 | DN19 | 304 | 92 | Alfa Laval | ISO 2037,Ra≤0.8 | 95.79 | 8,812.52 |
| 15 | 密封圈 | DN63.5 | PTFE | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 53.38 | 53.38 |
| 16 | 密封圈 | DN51 | PTFE | 74 | Alfa Laval | ISO 2037,Ra≤0.8 | 46.51 | 3,441.44 |
| 17 | 密封圈 | DN25 | PTFE | 44 | Alfa Laval | ISO 2037,Ra≤0.8 | 46.51 | 2,046.26 |
| 18 | 密封圈 | DN19 | PTFE | 92 | Alfa Laval | ISO 2037,Ra≤0.8 | 37.12 | 3,414.82 |
| 19 | 变径三通(WCW) | DN63.5/51 | 316L | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 264.15 | 264.15 |
| 20 | 变径三通 | DN63.5/38 | 316L | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 102.27 | 102.27 |
| 21 | 变径三通 | DN63.5/19 | 316L | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 147.81 | 147.81 |
| 22 | 变径三通 | DN51/38 | 316L | 2 | Alfa Laval | ISO 2037,Ra≤0.8 | 68.18 | 136.35 |
| 23 | 变径三通 | DN51/25 | 316L | 2 | Alfa Laval | ISO 2037,Ra≤0.8 | 129.53 | 259.06 |
| 24 | 变径三通 | DN51/19 | 316L | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 127.65 | 127.65 |
| 25 | 变径三通 | DN38/25 | 316L | 10 | Alfa Laval | ISO 2037,Ra≤0.8 | 100.27 | 1,002.71 |
| 26 | 变径三通 | DN38/19 | 316L | 4 | Alfa Laval | ISO 2037,Ra≤0.8 | 99.69 | 398.78 |
| 27 | 三通 | DN25/25 | 316L | 5 | Alfa Laval | ISO 2037,Ra≤0.8 | 65.08 | 325.41 |
| 28 | 变径三通 | DN25/19 | 316L | 30 | Alfa Laval | ISO 2037,Ra≤0.8 | 81.60 | 2,448.00 |
| 29 | 三通 | DN19 | 316L | 2 | Alfa Laval | ISO 2037,Ra≤0.8 | 262.44 | 524.87 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) |
| 30 | 大小头 | DN63.5/51 | 316L | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 163.62 | 163.62 |
| 31 | 大小头 | DN51/25 | 316L | 1 | Alfa Laval | ISO 2037,Ra≤0.8 | 136.71 | 136.71 |
| 32 | 大小头 | DN38/25 | 316L | 3 | Alfa Laval | ISO 2037,Ra≤0.8 | 77.21 | 231.64 |
| 33 | 大小头 | DN25/19 | 316L | 7 | Alfa Laval | ISO 2037,Ra≤0.8 | 83.72 | 586.02 |
| 34 | 弯头 | DN63.5 | 316L | 12 | Alfa Laval | ISO 2037,Ra≤0.8 | 56.49 | 677.93 |
| 35 | 弯头 | DN51 | 316L | 10 | Alfa Laval | ISO 2037,Ra≤0.8 | 39.61 | 396.12 |
| 36 | 弯头 | DN38 | 316L | 18 | Alfa Laval | ISO 2037,Ra≤0.8 | 34.26 | 616.66 |
| 37 | 弯头 | DN25 | 316L | 54 | Alfa Laval | ISO 2037,Ra≤0.8 | 26.95 | 1,455.46 |
| 38 | 弯头 | DN19 | 316L | 110 | Alfa Laval | ISO 2037,Ra≤0.8 | 220.33 | 24,236.24 |
| 39 | 管道 | DN63.5 | 304 | 18 | Alfa Laval | ISO 2037,Ra≤0.8 | 234.93 | 4,228.73 |
| 40 | 管道 | DN51 | 304 | 48 | Alfa Laval | ISO 2037,Ra≤0.8 | 149.74 | 7,187.58 |
| 41 | 管道 | DN38 | 304 | 84 | Alfa Laval | ISO 2037,Ra≤0.8 | 115.67 | 9,716.33 |
| 42 | 管道 | DN25 | 304 | 246 | Alfa Laval | ISO 2037,Ra≤0.8 | 85.19 | 20,956.31 |
| 43 | 管道 | DN19 | 316L | 114 | Alfa Laval | ISO 2037,Ra≤0.8 | 102.22 | 11,653.48 |
| 以上为压缩空气管道系统管材,管件配置清单及报价 | 248,861.78 | |||||||
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
2.2. 氮气管道系统材料清单及报价
Price Breakdown for N2 Distribution System
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) |
| 1 | 压力表 | 0-6kgf/cm2, 1” | 316L | 1 | CHINA | 500.00 | 500.00 | |
| 2 | 球阀 | DN25 | 316L | 1 | King Lai | 293.74 | 293.74 | |
| 3 | 球阀 | DN15 | 316L | 21 | King Lai | 230.77 | 4,846.20 | |
| 4 | 过滤器 | 5“ 0.22um | 3 | PALL | 4,700.00 | 14,100.00 | ||
| 5 | 卡接头 | DN25 | 316L | 10 | Alfa Laval | ISO 2037,Ra≤0.8 | 48.04 | 480.35 |
| 6 | 卡接头 | DN19 | 316L | 45 | Alfa Laval | ISO 2037,Ra≤0.8 | 47.40 | 2,133.00 |
| 7 | 卡环 | DN25 | 304 | 10 | Alfa Laval | ISO 2037,Ra≤0.8 | 49.56 | 495.65 |
| 8 | 卡环 | DN19 | 304 | 45 | Alfa Laval | ISO 2037,Ra≤0.8 | 95.79 | 4,310.47 |
| 9 | 密封圈 | DN25 | PTFE | 10 | Alfa Laval | ISO 2037,Ra≤0.8 | 46.51 | 465.06 |
| 10 | 密封圈 | DN19 | PTFE | 45 | Alfa Laval | ISO 2037,Ra≤0.8 | 37.12 | 1,670.29 |
| 11 | 三通 | DN25/25 | 316L | 3 | Alfa Laval | ISO 2037,Ra≤0.8 | 65.08 | 195.25 |
| 12 | 变径三通 | DN25/19 | 316L | 18 | Alfa Laval | ISO 2037,Ra≤0.8 | 81.60 | 1,468.80 |
| 13 | 大小头 | DN25/19 | 316L | 5 | Alfa Laval | ISO 2037,Ra≤0.8 | 83.72 | 418.59 |
| 14 | 弯头 | DN25 | 316L | 15 | Alfa Laval | ISO 2037,Ra≤0.8 | 26.95 | 404.29 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) |
| 15 | 弯头 | DN19 | 316L | 45 | Alfa Laval | ISO 2037,Ra≤0.8 | 220.33 | 9,914.82 |
| 16 | 管道 | DN25 | 304L | 210 | Alfa Laval | ISO 2037,Ra≤0.8 | 85.19 | 17,889.53 |
| 17 | 管道 | DN19 | 316L | 48 | Alfa Laval | ISO 2037,Ra≤0.8 | 102.22 | 4,906.73 |
| 以上为氮气管道系统管材,管件配置清单及报价 | 64,492.78 | |||||||
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
2.3. 氧气管道系统材料清单及报价
Price Breakdown for O2 Distribution System
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) |
| 1 | 压力表 | 0-6kgf/cm2, 1” | 316L | 1 | CHINA | 500.00 | 500.00 | |
| 2 | 球阀 | DN25 | 316L | 1 | King Lai | 293.74 | 293.74 | |
| 3 | 球阀 | DN15 | 316L | 20 | King Lai | 230.77 | 4,615.43 | |
| 4 | 过滤器 | 5“ 0.22um | 2 | PALL | 4,700.00 | 9,400.00 | ||
| 5 | 卡接头 | DN25 | 316L | 8 | Alfa Laval | ISO 2037,Ra≤0.8 | 48.04 | 384.28 |
| 6 | 卡接头 | DN19 | 316L | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 47.40 | 1,990.80 |
| 7 | 卡环 | DN25 | 304 | 8 | Alfa Laval | ISO 2037,Ra≤0.8 | 49.56 | 396.52 |
| 8 | 卡环 | DN19 | 304 | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 95.79 | 4,023.11 |
| 9 | 密封圈 | DN25 | PTFE | 8 | Alfa Laval | ISO 2037,Ra≤0.8 | 46.51 | 372.05 |
| 10 | 密封圈 | DN19 | PTFE | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 37.12 | 1,558.94 |
| 11 | 三通 | DN25/25 | 316L | 3 | Alfa Laval | ISO 2037,Ra≤0.8 | 65.08 | 195.25 |
| 12 | 变径三通 | DN25/19 | 316L | 17 | Alfa Laval | ISO 2037,Ra≤0.8 | 81.60 | 1,387.20 |
| 13 | 大小头 | DN25/19 | 316L | 4 | Alfa Laval | ISO 2037,Ra≤0.8 | 83.72 | 334.87 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第13页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) | |||
| 14 | 弯头 | DN25 | 316L | 12 | Alfa Laval | ISO 2037,Ra≤0.8 | 26.95 | 323.44 | |||
| 15 | 弯头 | DN19 | 316L | 45 | Alfa Laval | ISO 2037,Ra≤0.8 | 220.33 | 9,914.82 | |||
| 16 | 管道 | DN25 | 304L | 174 | Alfa Laval | ISO 2037,Ra≤0.8 | 85.19 | 14,822.75 | |||
| 17 | 管道 | DN19 | 316L | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 102.22 | 4,293.39 | |||
| 以上为氧气管道系统管材,管件配置清单及报价 | 54,806.58 | ||||||||||
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第14页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
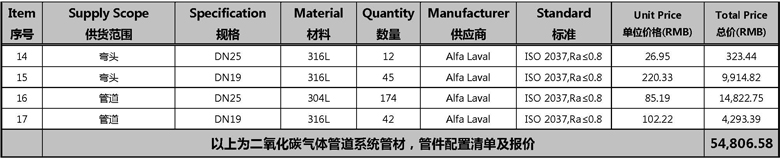
2.4. 二氧化碳气体管道系统材料清单及报价
Price Breakdown for CO2 Distribution System
Item 序号 | Supply Scope 供货范围 | Specification 规格 | Material 材料 | Quantity 数量 | Manufacturer 供应商 | Standard 标准 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) |
| 1 | 压力表 | 0-6kgf/cm2, 1” | 316L | 1 | CHINA | 500.00 | 500.00 | |
| 2 | 球阀 | DN25 | 316L | 1 | King Lai | 293.74 | 293.74 | |
| 3 | 球阀 | DN15 | 316L | 20 | King Lai | 230.77 | 4,615.43 | |
| 4 | 过滤器 | 5“ 0.22um | 2 | PALL | 4,700.00 | 9,400.00 | ||
| 5 | 卡接头 | DN25 | 316L | 8 | Alfa Laval | ISO 2037,Ra≤0.8 | 48.04 | 384.28 |
| 6 | 卡接头 | DN19 | 316L | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 47.40 | 1,990.80 |
| 7 | 卡环 | DN25 | 304 | 8 | Alfa Laval | ISO 2037,Ra≤0.8 | 49.56 | 396.52 |
| 8 | 卡环 | DN19 | 304 | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 95.79 | 4,023.11 |
| 9 | 密封圈 | DN25 | PTFE | 8 | Alfa Laval | ISO 2037,Ra≤0.8 | 46.51 | 372.05 |
| 10 | 密封圈 | DN19 | PTFE | 42 | Alfa Laval | ISO 2037,Ra≤0.8 | 37.12 | 1,558.94 |
| 11 | 变径三通 | DN25/25 | 316L | 3 | Alfa Laval | ISO 2037,Ra≤0.8 | 65.08 | 195.25 |
| 12 | 变径三通 | DN25/19 | 316L | 17 | Alfa Laval | ISO 2037,Ra≤0.8 | 81.60 | 1,387.20 |
| 13 | 大小头 | DN25/19 | 316L | 4 | Alfa Laval | ISO 2037,Ra≤0.8 | 83.72 | 334.87 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第15页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第16页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
2.5. 焊点费
Welding Point Cost
Item 序号 | Supply Scope 供货范围 | Location 位置 | Quantity 数量 | Unit Price 单位价格(RMB) | Total Price 总价(RMB) | |
| 1 | 压缩空气系统 | 自动焊点 | 835 | 150.00 | 125,250.00 | |
| 2 | 氮气系统 | 自动焊点 | 262 | 180.00 | 47,160.00 | |
| 3 | 氧气系统 | 自动焊点 | 243 | 180.00 | 43,740.00 | |
| 4 | 二氧化碳系统 | 自动焊点 | 243 | 180.00 | 43,740.00 | |
| 总价(元) | 259,890.00 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第17页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
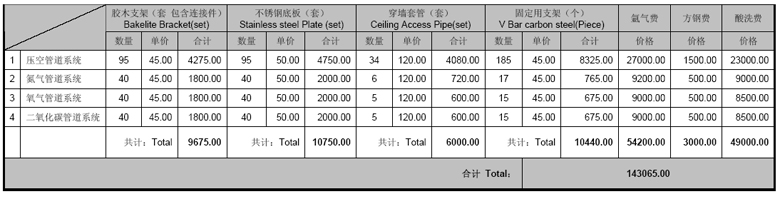
2.6. 系统所用辅材数量清单
Accessories List
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第18页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第1 部分投标书 Tender | ||
2.7. 验证服务报价
Quotation For Validation Activities
| 序号 No. | 验证项目 Scope | 报价 Quotation(RMB) | |
| 1 | 压缩空气分配系统 Compressed air distribution system | DQ,TM,RA | 10,000 |
| 2 | SAT | 5,000 | |
| 3 | IQ,OQ | 20,000 | |
| 4 | PQ 方案 PQ protocol | 5,000 | |
| 6 | 氮气分配系统 Nitrogen distribution system | DQ,TM,RA | 10,000 |
| 7 | SAT | 5,000 | |
| 8 | IQ,OQ | 20,000 | |
| 9 | PQ 方案 PQ protocol | 5,000 | |
| 11 | 氧气分配系统 Carbon dioxide distribution system | DQ,TM,RA | 10,000 |
| 12 | SAT | 5,000 | |
| 13 | IQ,OQ | 20,000 | |
| 14 | PQ 方案 PQ protocol | 5,000 | |
| 11 | 二氧化碳分配系统 Carbon dioxide distribution system | DQ,TM,RA | 10,000 |
| 12 | SAT | 5,000 | |
| 13 | IQ,OQ | 20,000 | |
| 14 | PQ 方案 PQ protocol | 5,000 | |
| 15 | 总价 | 160,000 | |
| Remarks 备注: | |||
| 1) 包含本报价服务范围内的朗脉验证人员在客户现场的差旅食宿费用。 The cost of trips and living in client site are included in above price。 | |||
| 2) 包含验证仪器费用。 The fee of validation instruments are included in above price.。 | |||
| 3) 验证不包括系统中仪表和传感器的校准工作。 The calibration work for instruments and sensors in the systems is not validation work.。 | |||
| 4) 本报价有效期:3 个月内。 | |||
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第19页/ 共19 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
第 2 部分
Part 2
公司介绍及资质文件
Company introduction and Certificate
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
目录Index
| 1. 资质证明文件Certificate | 3 |
| 1.1. 资质证书Certificate | 3 |
1.1.1. 营业执照复印件Business license | 3 |
| 1.1.2. 组织机构代码证Organization code certificate | 4 |
| 1.1.3. 税务登记证Business Registration | 5 |
| 1.1.4. 银行开户证明The opening of bank accounts permits | 6 |
1.1.5. 代理证书Agency | 7 |
1.1.6. 协会证书CAPE Certificate | 13 |
| 1.2. 部分业绩Part reference | 14 |
| 1.2.1. 部分竣工项目一览表 Part finished project List | 14 |
| 1.2.2. 部分在建同类工程一览表 Part constructing project List | 15 |
| 1.3. 2007 年财务报表Financial statement | 15 |
| 2. 公司介绍Company Introduction | 15 |
| 2.1. 公司概述Brief introduction | 15 |
2.1.1. 公司实力Strength | 15 |
| 2.1.2. 公司历史History | 15 |
| 2.1.3. 公司文化Corporate cuture | 15 |
| 2.1.4. 公司组织结构Organize chart | 15 |
| 2.2. 朗脉的服务Service | 15 |
2.2.1. 专业领域服务Professional service | 15 |
2.2.2. 工程总承包Contractor | 15 |
| 2.2.3. 植物提取工艺优化Process optimization | 15 |
| 2.2.4. 自动化控制系统业务Automation Control system | 15 |
2.2.5. 过程控制系统Process Flow | 15 |
| 2.2.6. 空调自控系统HVAC automation system | 15 |
2.2.7. 在线清洗系统Clean in place | 15 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
2.3. 洁净管道系统业务Sanitary piping system business | 15 |
2.3.1. 完善的工艺设计Perfect in process design | 15 |
2.3.2. 材料选型及采购Material Selection and Procurement | 15 |
2.3.3. 齐全的施工设备Construction facility | 15 |
2.3.4. 现场管理及现场施工Project management and construction | 15 |
2.3.5. 先进的检测设备Testing equipment | 15 |
2.3.6. 系统调试和人员培训Training and system commission | 15 |
2.3.7. 验证服务Validation service | 15 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1. 资质证明文件Certificate
1.1. 资质证书Certificate
1.1.1. 营业执照复印件Business license

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.1.2. 组织机构代码证Organization code certificate

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.1.3. 税务登记证Business Registration

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第5页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.1.4. 银行开户证明The opening of bank accounts permits

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.1.5. 代理证书Agency

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文�� Part 2 Company intruduction and Certificate | ||
1.1.6. 协会证书CAPE Certificate

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第13页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.2. 部分业绩Part reference
1.2.1. 部分竣工项目一览表 Part finished project List
| 序号 | 客户名称 | 地点 | 项目概况 | 认证情况 |
| 1 | 北京凯因生物技术有限公司 | 北京 | 纯水/注射用水/纯蒸气分配系统/干扰素发酵物料管道转运系统和空 调自控(工艺管道) | |
| 2 | 北京万泰制药有限公司 | 北京 | 纯水/注射用水/纯蒸汽/物料分配系统 | |
| 3 | 北京三元基因工程有限公司 | 北京 | 纯水/注射用水/纯蒸汽/物料分配系统 | |
| 4 | 常州四药有限公司 | 常州 | 注射用水/纯化水管道分配系统 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 5 | 河北恒利药业有限公司 | 河北 | CIP 系统 | |
| 6 | 成都普仕医塑公司 | 成都 | 纯水/注射用水/纯蒸汽及物料管道分配系统 | |
| 7 | 扬子江药业集团广州海瑞药业有限公司 | 广州 | 纯蒸汽/纯水/注射用水/物料分配管道系统 | |
| 8 | 齐鲁海南药业有限公司 | 海南 | 纯蒸汽/注射用水/纯水以及工艺管道系统 | |
| 9 | 杭州天元生物制药有限公司 | 杭州 | 注射用水/纯蒸汽/纯化水/洁净压缩空气/氮气等工艺管道系统 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 10 | 杭州九源基因工程有限公司 | 杭州 | 纯蒸汽/注射用水/纯水以及工艺管道系统和空调自控 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第14页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
| 序号 | 客户名称 | 地点 | 项目概况 | 认证情况 |
| 11 | 上海博莱科信谊制药有限公司 | 上海 | (蒸馏水生产装置MS304S,纯蒸汽生产装置PSG500DTS,纯水& 蒸馏水&纯蒸汽分配系统) | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 12 | 齐鲁安替比奥制药有限公司 | 济南 | 纯水& 注射用水分配系统/ 发酵物料转运管道系统(二期) | 无菌原料药已通过欧盟认证 |
| 13 | 齐鲁制药厂 | 济南 | 纯水/注射用水/纯蒸汽/物料分配系统 | |
| 14 | 齐鲁安替比奥制药有限公司 | 济南 | 注射用水/纯化水/纯蒸汽管道分配系统 | |
| 15 | 齐鲁天和惠世制药有限公司 | 济南 | 注射用水/纯化水/纯蒸汽管道分配系统 | |
| 16 | 山东鲁抗辰欣药业有限公司 | 济宁 | 注射用水/纯化水/纯蒸汽管道分配系统和空调自控 | |
| 17 | 山东鲁抗立科制药有限公司 | 济宁 | 注射用水/纯化水/纯蒸汽管道分配系统和空调自控 | |
| 18 | 浙江耐斯康制药有限公司 | 金华 | 注射用水/纯蒸汽/纯化水分配系统改造项目 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 19 | 扬子江药业集团南京海陵药业有限公司 | 南京 | 纯蒸汽,注射用水分配系统工艺管道工程 | |
| 20 | 宝利化(南京)制药有限公司 | 南京 | 注射用水/纯化水管道分配系统 | 无菌注射剂已通过TGA 认证 |
| 21 | 青岛生化制药有限公司 | 青岛 | 纯蒸汽/纯水/注射用水/压缩空气分配系统 | |
| 22 | GE Healthcare | 上海 | 纯蒸汽/注射用水工艺管道工程 | 无菌注射剂已通过FDA 认证 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第15页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
| 序号 | 客户名称 | 地点 | 项目概况 | 认证情况 |
| 23 | 石药集团中润制药有限公司 | 石家庄 | 注射用水/纯化水/纯蒸汽及物料分配系统 | |
| 24 | 上海天士力药业有限公司 | 上海 | 纯蒸汽/注射用水分配系统工艺管道工程 | |
| 25 | 上海百特医疗用品有限公司 | 上海 | 纯水/注射用水/配液分配系统 | |
| 26 | 上海惠生生物工程有限公司 | 上海 | 纯水/注射用水/纯蒸汽管道分配系统 | |
| 27 | 上海迪赛诺制药有限公司 | 上海 | 注射用水/纯化水管道分配系统 | 符合WHO 规范 |
| 28 | 上海惠生生物工程有限公司 | 上海 | 抗体实验室纯化水分配系统(二期) | |
| 29 | 上海莱士血液制品股份有限公司 | 上海 | 注射用水分配系统过热水灭菌系统改造项目 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 30 | 深圳海王英特龙生物科技有限责任公司 | 深圳 | 纯蒸汽/注射用水/纯水系统工艺管道系统 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 31 | 华北制药奥奇德有限公司 | 石家庄 | 纯水/注射用水/纯蒸气分配系统/干扰素发酵物料管道转运系统 | |
| 32 | 华北制药华胜有限公司 | 石家庄 | 纯蒸汽/注射用水/纯水系统及物料工艺管道分配系统 | |
| 33 | 华北制药集团北元有限公司 | 石家庄 | 注射用水,纯化水,纯蒸汽分配系统改造项目 | 依照FDA cGMP 规范, |
| 34 | 石家庄欧意制药有限公司 | 石家庄 | 纯化水分配系统改造项目 | 依照FDA cGMP 规范, |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第16页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
| 序号 | 客户名称 | 地点 | 项目概况 | 认证情况 |
| 35 | 无锡台裕制药有限公司 | 无锡 | 注射用水/纯化水/纯蒸汽系统及物料分配管道系统 | |
| 36 | 厦门北大之路药业有限公司 | 厦门 | 注射用水/纯化水纯蒸汽系统及物料分配管道系统 | |
| 37 | 苏州东瑞药业有限公司 | 苏州 | 纯蒸汽/注射用水/纯水系统及物料工艺管道分配系统 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 38 | 贝朗医疗(苏州)有限公司 | 苏州 | 纯水/注射用水/配液分配系统 | |
| 39 | 苏州东瑞制药有限公司 | 苏州 | 104 车间物料系统改造 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 40 | 苏州东瑞制药有限公司 | 苏州 | 原料车间物料系统改造 | 依照FDA cGMP 规范,提供IQ/OQ 文件 |
| 41 | 贝朗医疗(苏州)有限公司 | 苏州 | 注射用水系统改造项目 | |
| 42 | 扬子江药业集团有限公司 | 泰州 | 纯蒸汽,注射用水分配系统工艺管道工程 | |
| 43 | 天津天士力药业有限公司 | 天津 | 纯蒸汽/纯水/蒸馏水/压缩空气分配系统 | |
| 44 | 天津百特医疗用品有限公司 | 天津 | 纯蒸汽/注射用水分配系统工艺管道工程 | |
| 45 | 天津天士力药业有限公司 | 天津 | 实验室纯水管道系统 | |
| 46 | 通化东宝药业 | 通化 | (蒸馏水生产装置MS3005S, 纯蒸汽生产装置PSG500DTS, 纯水& 蒸馏水&纯蒸汽分配系统, 大输液配制系统) |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第17页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
| 序号 | 客户名称 | 地点 | 项目概况 | 认证情况 |
| 47 | 香港美达制药有限公司 | 香港 | 纯水分配系统 | |
| 48 | 厦门科兴生物制品有限公司 | 厦门 | (预处理,二级反渗透,蒸馏水生产装置MS504S, 纯蒸汽生产装置PSG500DTS ,纯水&蒸馏水&纯蒸汽分配系统) | |
| 49 | 劲牌有限公司 | 湖北 | 保健酒仪表、计量控制系统 | |
| 50 | 贝朗医疗(苏州)有限公司 | 苏州 | 改造公用洁净设施工程 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第18页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.2.2. 部分在建同类工程一览表 Part constructing project List
| 序号 | 客户名称 | 地点 | 项目概况 | 认证情况 |
| 1 | 成都生物制品所 | 成都 | 洁净室配套公用工程系统 | 符合WHO 标准 |
| 2 | 株洲千金药业有限公司 | 湖南 | 煮提、浓缩自控总包项目 | |
| 3 | 浙江普洛康裕药业有限公司 | 浙江 | 冻干粉针洁净管道和配料系统工程合同 | |
| 4 | 赣州菊隆高科技食品有限公司 | 江西 | 萃取、提纯车间自控系统工程项目合同 | |
| 5 | 山东先声麦得津生物制药有限公司 | 山东 | 工艺管道安装工程项目 | |
| 6 | 成都生物制品所 | 成都 | 乙脑疫苗车间改造 | 符合WHO 标准 |
| 7 | 成都生物制品所 | 成都 | 病毒疫苗车间PW/WFI/PS 系统 | |
| 8 | 杭州中美华东制药有限公司 | 杭州 | 冻干粉针201 车间洁净工艺管道系统工程 | |
| 9 | 苏州东瑞制药有限公司 | 苏州 | 103 车间洁净管道系统改造 | |
| 10 | 江苏正大丰海制药有限公司 | 江苏 | 输液车间注射水及物料洁净管道系统 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第19页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
1.3. 2007 年财务报表Financial statement
针对本招标文件,公司提供2007 年度财务审计报告复印件作为参考。
We provided the copy of 2007 financial statement as reference for the project bidding requirement.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第20页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
2. 公司介绍Company Introduction
2.1. 公司概述Brief introduction
2.1.1. 公司实力Strength
公司上海朗脉科技有限公司由上海双赢洁净科技有限公司和上海朗和系统集成有限公司整合重组而成,公司注册资金一千万,现有员工一百多人。我们致力于成为中国最好的制药工程专业提供商。
Shanghai Macroprocess Co., Ltd. was founded by integrating Shanghai WinWin Process System Co., Ltd. and Shanghai Longhorn System Integration Co., Ltd. We have over one hundred employee with RMB 10 million pay up capital. We are committed to become a leading supplier for the total solution in pharmaceutical industry.
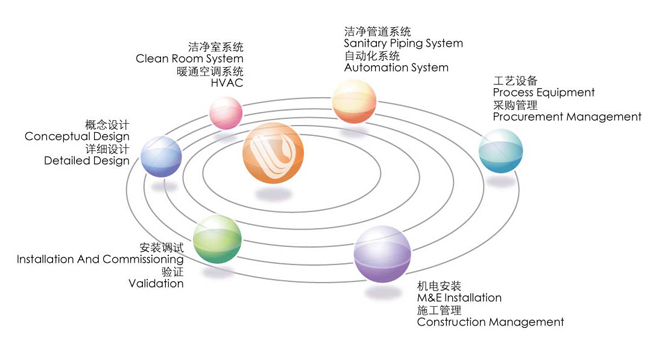
“朗脉”本着“以人为本、技术为核心”的原则,聘请多位国内外技术专家,经过长期发展,打造了一支经验丰富的设计、安装、调试及验证的专业队伍。我们为您的药厂建设提供专业的概念设计 、详细设计、工艺设备、洁净管道、暖通空调系统、洁净室系统、自动化系统、采购管理、施工管理、安装调试、验证等一体化服务。

Following the principle “ putting employee first, setting technology as core”, Macroprocess has accomplished an experienced professional team made up of experts on design , installation and validation by recuiting many technical experts both from home and abroad after a long-time development. We can provide your
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第21页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
pharmaceutical factory contruction with integrated services, including professional conceptual design, detailed design , process equipments , sanitary pipelines , HVAC system, cleanroom system, automation system , procurement management, construction management, installation with commissioning , validation and etc.
“朗脉”的客户涵盖了许多国内外知名的制药企业,其中已有多家客户通过FDA 、EME A、TGA 等国外认证,并建立了良好的长期合作关系。
Macroprocess has served many famous pharmaceutical enterprises both from home and abroad, of which many enterprises have passed foreign validation ( USA FDA, EU EMEA, Australian TGA and etc.) We are maintaining excellent cooperative relationship with our clients.
目前我们用户中已经通过国外验证的有some of our clients have gained International certification:
u | GE Healthcare 通用医疗(上海)药业有限公司是美国GE 通用的独资企业,是目前为止国内唯一一家无菌小容量注射剂通过美国FDA 认证的制药企业;GE Healthcare Located in Shanghai City , Sterility injection was certified by USA FDA and also this is the only one which approvaled in China. |
u | 山东齐鲁安替比奥制药有限公司是意大利安替比奥公司与齐鲁制药有限公司的合资企业,是国内第一家无菌原料药(相当于注射剂)通过欧盟EMEA 认证的制药企业;Qilu Antibiotics Pharmaceutical Co., Ltd. is the joint venture company of Italy Antibiotics and Qilu |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第22页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
pharmceutical company, it is the first one of China which Sterile API was certified by EU EMEA.
u | Polifarma 宝利化(南京)制药有限公司是意大利Polifarma 公司控股的企业,无菌制剂以及口服固体制剂于2007 年通过了澳大利亚TGA 认证。Polifarma (Nanjing) Co. Ltd. is a holding company of Italy Polifarma, Sterile injection was certified by TGA in 2007. |
我公司在生物制药领域的部分客户有as the following there are some reference in bio pharmaceutical domain:
u | 北京凯因生物技术有限公司作为病毒生物技术国家工程研究中心,是由中国疾病预防控制中心病毒所和大连国际合作(集团)股份有限公司共同投资建立,我公司多次承接其洁净管道系统安装工程;Kawin Technology's core R&D organization – National Engineering Research Center for Viral Bio-Technology (NERC) was established with the approval & support of National Development and Reform Commission. Beijing Kawin Technology Share-Holding Co., Ltd. (Kawin Technology) was invested by Beijing Kawin Bio-Tech Co., Ltd. We cooperation in piping system many times. |
u | 成都生物制品研究所作为我国西南六省、市、自治区计划免疫协作中心,是中国最大的生物制品研究、生产基地之一,我公司先后承接乙脑疫苗车间水系统、病毒性疫苗项目水系统、洁净室配套公用工程系统等多个符合GMP 标准的工程; The Chengdu Institute of Biological Products (CDIBP), part of the China National Biotec Corporation, |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第23页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
is one of the largest research and manufacturing bases for biological products in China.And we are participated in some sanitary piping system with GMP standard.
u | 浙江天元生物药业股份有限公司是中国国内规模最大的流感疫苗生产企业之一。所有的生产车间都通过国家药监局的GMP 认证。我公司先后承接其疫苗车间的洁净管道施工安装项目,按照WHO 的标准实施项目。Zhejiang Tianyuan Bio-Pharmaceutical Co., Ltd. is one of China's largest private manufacturers of influenza vaccine. All manufacturing lines of Tianyuan have been granted with cGMP certificates from the State Food and Drug Administration (SFDA) of China. Our company carried on some sanitary piping system project in bacterin factory with WHO standard. |

"朗脉" 以 "专业成就未来" 为使命,打造中国真正意义上的制药领域的专业工程公司,不断增强公司的核心竞争力,推动更多的本土药厂走向世界。
With the mission that "expertise for future", Macroprocess is committed to become a Chinese professional engineering company that can bring more and more local pharmaceutical companies to the world by continuously improving core competiton capability in pharmaceutical industry.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第24页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
2.1.2. 公司历史History
u | 上海双赢洁净科技有限公司成立。Shanghai WinWin Process System Co., Ltd was founded |
u | 上海双赢洁净科技有限公司与意大利STILMAS 建立合作关系。Shanghai WinWin Process System Co., Ltd commenced business relationships with Italian STILMAS. |
u | 上海朗和系统集成有限公司成立,进入到中药现代化领域。Shanghai Longhorn System Integration Co., Ltd was founded with the entry into TCM modernization field. |
u | 上海双赢洁净科技有限公司将国际化的验证概念融入项目实施。WinWin brought the international validation concept into project execution. |
u | 聘请国外资深工艺及验证专家。International experienced process and validation experts were recruited. |
u | "朗脉" 的多名技术成员成为国际制药工程协会(ISPE )会员。Several Macroprocess engineers are members of ISPE |
u | 融合工艺、管道、自控、HVAC 及洁净室系统、验证等各专业,成立技术中心。A technical center was operated with integration of process, piping, automation, HVAC and validation sections. |
u | 双赢与朗和整合,成立上海朗脉科技有限公司。WINWIN and LONGHORN were intergrated into MACROPROCESS. |
u | "朗脉" 与国外公司合作开发了连续式真空带式干燥机。Macroprocess invented Vacuum belt dryer with cooperation with foreign enterprises. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第25页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

2.1.3. 公司文化Corporate cuture
 | 朗脉的愿景The Vision of MACROPROCESS: |
我们致力于成为中国最好的制药工程专业提供商。
We are committed to become a leading supplier for the total solution in pharmaceutical industry.
 | 朗脉的使命Mission of Macroprocess: |
| 专业成就未来!Experitise for future! |
 | 朗脉的核心价值观Core values of Macroprocess: |
合作、创新、负责、分享 Collaboration, Innovation, Responsibility and Sharing. |
 | 朗脉的核心竞争力Core Competitiveness of Macroprocess: |
| ♦ | 朗脉的专业能力将永远是我们引以为荣的核心竞争力Expertise of Macroprocess is always our core competitiveness; |
| ♦ | 因为我们专业,完成了一个又一个的高标准项目Owing to our expertise, we have successfully completed a large |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第26页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
number of high-standard projects;
♦ | 因为我们专业,建立了与客户的长久合作关系Owing to our expertise, we have built long-term cooperation relationship with our clients; |
♦ | 专业,成就朗脉的未来,也将成就朗脉伙伴的未来Our expertise is creating the future for both MACROPROCESS and our clients! |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第27页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
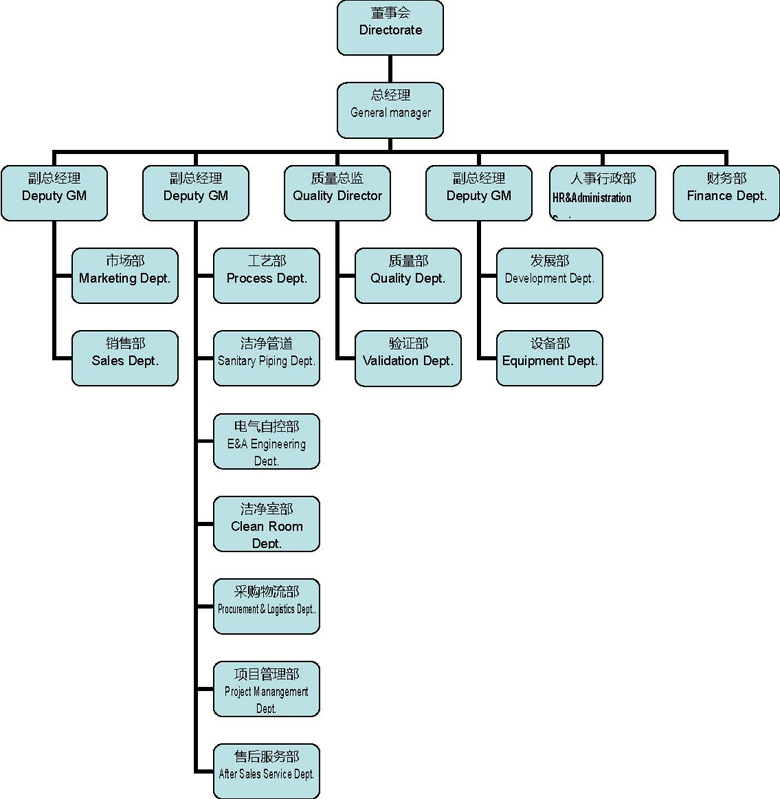
2.1.4. 公司组织结构Organize chart
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第28页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
2.2. 朗脉的服务Service
2.2.1. 专业领域服务Professional service
“朗脉”在引进吸收国际先进项目管理理念的基础上,经过多年积累,建立了完善的项目管理体系,对工程整个周期进行全方位的管理,最大限度提高项目的成功率,降低过程控制的成本,规避项目风险。
With the introduction of advanced international project management ideology and many years’ experiences, Macroprocess has established a perfect project management system to conduct all-round management to the entire cycle of project. This system can facilitate project success , cost reduction and risk avoidance.
“朗脉”真诚的为您提供以下各专业服务Macroprocess can offer the following professional service:
 | 洁净管道系统Sanitary piping system |
♦ | 配料系统Formulation system: |
● | 小容量注射剂配料及输送系统SVP formulation and distribution system |
● | 大输液配料及输送系统LVP formulation and distribution system |
● | 冻干制剂配料及输送系统Lyophilized product formulation and distribution system |
● | 生物工程、血液制品等配料及输送系统preparation and distribution system for bioengineering and blood products |
● | 无菌原料药生产工艺系统等Sterile raw material production |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第29页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
process system etc.
♦ | 工艺用水、汽系统Process used water and steam system: |
| ● | 纯化水系统、注射用水系统、纯蒸汽系统等Purified water system, water for injection system and pure steam system etc. |
♦ | 洁净气体(空气、氮气等)输送系统Clean gas( air/nitrogen etc.) delivering system |
♦ | CIP 在线清洗工作站及系统CIP(Clean In Place)work stations and systems |
♦ | SIP 在线消毒工作站及系统SIP(Sterilization-in-Place)work stations and systems |
 | 自动化系统Automation control system |
♦ | 工艺过程自动化控制系统Automation control system of process procedure |
♦ | 洁净室空调自动化控制系统HVAC automation system |
 | 洁净厂房系统Clean room system |
♦ | 洁净厂房系统的设计、安装、调试及验证(中国GMP、欧盟EMEA 及美国FDA 等) 。Professional design , installation, commissioning and validation (Chinese GMP, EMEA, FDA , etc.) of clean room system. |
 | 验证服务Validation services |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第30页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
“朗脉”专门的验证部门为您研究全球的认证规范,涉及FDA 、EMEA 、TGA、WHO 等。我们熟悉验证的重要性和复杂性,为顾客提供切实有效的验证服务,确保项目符合国内外GMP 法规要求。我们提供以下专业验证服务 Macroprocess Validation department can provide world class validation service, such as cGMP of SFDA, FDA, EMEA, TGA, WHO and etc. With great familiarity and complexity of validtion, we can provide pragmatic and effective validation services to ensure the uniformity of your project with GMP standards. We can provide the following professional validation services:
♦ | 编写验证主计划Writing of VMP (Validation Master Plan) |
♦ | 设计cGMP 审核Design cGMP Review |
♦ | 编写URS Writing of URS |
♦ | HVAC 系统和洁净室的验证Validation of HVAC system and Cleanroom |
| ♦ | 水系统、纯蒸汽和压缩空气系统的验证Validation of Water system, Pure steam and compressed air system |
♦ | 灭菌设备的性能确认PQ of Sterilization equipments |
♦ | 关键工艺设备的验证指导Guiding of Critical Process Equipments Validation |
♦ | 清洗验证的指导Guiding of Cleaning Validation |
♦ | 工艺验证的指导Guiding of Process Validation |
♦ | 计算机系统的验证Computer System Validation |
♦ | 验证培训Validation Training |
2.2.2. 工程总承包Contractor
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第31页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
对于任何一个项目,工程设计、安装调试、投资控制、工期控制和质量控制以及环保健康和安全管理等对于投资商的利益和发展都是至关重要。“朗脉”提供的专业化工程服务最大限度地避免了工程建设的风险,给投资方带来最大的经济效益。
For any project, the key to the benefit and development of investors are engineering design, installation and commissioning, investment control, project period control, quality control, environment protection, safety and security managemnt. Besides, GMP inspection consultation and validation services also play a special role in pharmaceutical factory project. Professional engineering services that offered by Macroprocess maximize the project risk avoidance and the benefit.
“朗脉”通过采用国际通用的工程总承包(EPC )模式,将“朗脉”自身的各子专业服务整合及与国际知名的工程公司合作,为您的药厂建设提供符合国际cGMP 规范的概念设计 、详细设计、工艺设备、洁净管道、暖通空调系统、洁净室系统、自动化系统、采购管理、施工管理、安装调试、验证等一体化整体解决方案。
Through adoption of EPC formula, integration of different specialties and cooperation with international famous engineering companies, Macroprocess can provide integrated total solution ,including conceptual design, detailed design, process equipments, sanitary piping , HVAC, cleanroom system, automation system , procurement management, construction management, installation, commissioning and validation etc. which are in conformity with international cGMP for pharmaceutical factory construction .
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第32页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
| 2.2.3. | 植物提取工艺优化Process optimization “朗脉”致力于植物提取工艺的优化及中药现代化。 |
we are committed to process optimization of plant extraction and TCM modernization
 | 植物提取工艺优化Optimization of Plant extraction |
我们提供植物提取生产线的工艺设计优化、工艺成套设备、管道安装及自动化集成等专业服务。
We can provide process optimization for plant extraction production line, process equipment, piping installation and integration of automation control and etc.
 | VBD 系列全自动连续式真空带式干燥机VBD series --fully automatic continuous vaccum band-type dryer |
“朗脉”在引进消化国外先进技术基础上,研制开发的一种全新概念的高效节能、环保型干燥设备。彻底解决了其它干燥设备难以解决的高粘度、高脂、高糖、高热敏性等物料的干燥。
Based on the introduction of foreign advanced technology ,
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第33页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
Macroprocess has developed this kind of new drying machine of high efficiency and ecological characteristics which can thoroughly solve the problem of drying substances of high vicosity, high fat, high sugar content and heat sensitivity etc. that other drying machines cannot overcome.

2.2.4. 自动化控制系统业务Automation Control system
制药及相近行业自动化系统的设计、安装、调试及验证(中国GMP、欧盟EMEA 及美国FDA 的cGMP)的工程是上海朗脉科技有限公司主营业务之一。
Professional design, installation and validation (Chinese GMP,EU EMEA and USA FDA ) of automation systems for the pharmaceutical and relevant industry fields is one of Macroprocess Technology’s core business.
“朗脉科技”本着“以技术为核心”的原则,聘用多位国内外技术专家,经过长期发展,造就了一批经验丰富的设计、安装、调试及验证的专业团队。
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第34页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
In line with the principle “take the technology as the core”, we recruits lots of domestic and foreign technical experts. After the extensive experience we have cultivated and accomplished a group of specialized experts in design, installation and validation for domestic and foreign projects.
2.2.5. 过程控制系统Process Flow
在工业生产中,各单元操作集合而成的生产流程,例如:提取、发酵、合成、萃取、过滤、配料、浓缩、精馏、沉淀、结晶、干燥等,实现生产过程的自动化,并且可以大大提高生产效率,减少人为差错,保证产品质量,降低生产成本,提高生产质量管理水品,为企业现代化和信息化管理搭建数据平台。
In pharmaceutical factory,the process flow integrated by each unit operation, i.e. extraction, fermentation, chemosynthesis, filtration, preparation, concentration, distillation, deposition, crystallization, drying and so on, and the process flow with automation could maximize production efficiency, reduce human error, ensure product quality, reduce production cost, advance the quality management level, set up the data platform for company modernization and informationization.

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第35页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

2.2.6. 空调自控系统HVAC automation system
上海朗脉科技有限公司拥有自己的洁净室项目实施管理团队,对制药厂房实施项目实施。
Macroprocess has own team to manage clean room project, and to proceed the project for whole pharmaceutical facotory.

朗脉公司采用国际先进的板材保证洁净厂房的质量,采用先进的HVAC 系统,尤其对HVAC 系统进行自动化控制和门禁一卡通系统,实现EMS 管理和空气缓冲的互锁,符合FDA 的要求;对于空气质量要求高的厂房引入文丘里风阀系统保证压差。
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第36页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
朗脉公司认为一个好的洁净室项目的实施,离不开好的集成化的设计,公司各个富有施工经验的专业人员利用三维设计对项目进行预演。
We believe that to proceed a HVAC Automation system project without a integrated design, and we will preview the system via 3D software.
空调运行需要用大量的能源,系统在满足生产要求的前提下,实现能耗最小化、运行效率最高化,可以提高设备运行安全可靠性和使用寿命。
HVAC’s operation needs lot of energy. BA system could minimize consumption, maximize operation efficiency, enhancing the reliability of operation and working life and achieving the process requirements.
系统具有数据记录、查询、报警、打印及趋势曲线图功能,利于管理。

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第37页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

BA System has the functions of data logging, search, alarm, printout and trend graph for easy operation.
2.2.7. 在线清洗系统Clean in place
全自动CIP 清洗是指设备(罐体、管道、泵等)及整个生产线在封闭的系 统中,无需人工拆卸便能进行有效的清洗和消毒。系统由酸、碱、热水、纯化水、注射用水、酸碱配置罐和换热器组成,根据工艺特点设定酸碱浓度、清洗流程、清洗液温度、清洗时间、保证产品质量,便于QA 工作的自动化和清洗工作的规范化。
CIP means that the equipment and the whole production line could be efficiently cleaned and sanitized without disassembly in close system. A CIP system is composed by detergents, hot water, purified water, WFI, preparation tank and heat exchanger. Concentration of detergents, clean flow, water temperature and cleaning time can be set
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第38页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
according to process specification, which could ensure the product quality, automation of QA affairs and standardization of cleaning.
| ● | 可分区同时清洗一个或者两个以上区域,大大缩短CIP 清洗时间,使生产 |
| ● | 减少工艺用水量、节省清洗剂、减低生产成本CIP system conduces to reduce process water consumption, saving detergent and reducing production cost |
| ● | 对清洗过程进行自动检测、显示和控制CIP system conduces to automatic measurement, display and control in cleaning process. |
| ● | 形成电子记录,符合GAMP4 和FDA 21 CFR PART 11 的要求CIP system generates electronic record compliance with the requirement of GAMP4 and 21 CFR PART 11 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第39页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
2.3. 洁净管道系统业务Sanitary piping system business
2.3.1. 完善的工艺设计Perfect in process design
对于我们承接的每一个项目,我们的设计团队在公司具有丰富管理施工经验的项目经理的领导下,并充分结合客户的URS 进行详细的优化设计,为客户做出具精确的P&ID 图、平面布局图、乃至施工图。这种优化设计是重要的,也是必须的,精确了材料数量,节省了施工时间,是为客户争取最大利益的保证。
For each project in which we are participated, our project manager in construction; Our design team can provide fully integrated and optimized detailed design according to the URS of our clients, the precise P&ID chart, layout plans, as well as construction plans. This kind of optimized design is a must. The precise amount of material, the organization of time, guarantees to act in the best interests of customers.
对于复杂的工艺管道系统,根据客户的需要和现场实际情况,我们的设计团队可以制作模拟现场的3D 效果图,从而保证了整个系统的精确定位和合理布置。3D 效果图可以衍生出相应的平面图,单管图及材料清单便于控制投资成本及施工周期,大大提高了施工效率和质量,是为提供给客户良好工程的重要保证。
Regarding complex piping systems, the needs of the customers and
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第40页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
the actual conditions, our design team can produce 3D simulation of the complete site, thus ensuring the accurate positioning and rational layout of the whole system. The corresponding plan can be developed from the 3D effect chart; singular pipeline plans can facilitate inventory control and materials costs and also the investment in the construction cycle, greatly increasing the efficiency and quality of construction.
2.3.2. 材料选型及采购Material Selection and Procurement
“质量和信誉是企业生存之本”,优质的材料供应商是高质量工程的基石。
Quality materials suppliers -- the cornerstone of high-quality works.
采购部门,根据技术部门的提供的参数和要求,为客户选择可靠的供应商及高性价比的产品。通过对供应商的评估、采购数据、所购产品的认证并结合客户的URS 为我们良好的工程提供重要的保证。Procurement departments, according to provided technical parameters and requirements, purchase products from reliable suppliers with high performance. Through vendor evaluation, procurement data, certificate of products and integration with URS provide an important guarantee for our work.
目前“朗脉科技”已经成为国内外许多知名公司的长期合作伙伴,如:After years of development, "Win Win" becomes the long-term partner of many well-known domestic and foreign enterprises:
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第41页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
瑞典:Alfa laval
德国:Gemu、Burker t、E+H、+GF+、Siemens
瑞士:Mettler-Toledo
丹麦:Grundfos
台湾:Kinglai
优质的材料供应商,加上“朗脉科技”先进的优化设计理念、科学的工程管理、丰富的国内外验证经验,以及科学严谨地规范每一个工作流程;确保您的整个自控系统工程准时、高质量地完成。
Good cooperative relations with suppliers and ensuring their quality plans to ensure that the project completed on time and according to the specified quality of the Client.

2.3.3. 齐全的施工设备Construction facility
我们齐全的施工设备能够承担任何作业。Our organization is fully equipped with hardware to undertake any task.
洁净管道专业切割设备Import cutting machinery specially for pipelines:
| ● | 切割端面平整Flat cutting surface |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第42页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
| ● | 切割端面无氧化Non-oxidized cutting surface |
| ● | 切割端面无变形Non-deformed cutting surface 平口机完成专用切割机无法完成的其他任务,如特殊位置用的管件需要修整或对管道的端面进行平整等等。Tube square machine that can complete the mission that special cutting machine can not complete, e.g. special positions and surface in the pipeline which need trimming. |
| ● | 全自动轨道焊接设备World-class automatic orbital welding machinery |
| ● | 焊缝内外充氩气保护,无氧化Welding is argon-protected without being oxidized |
| ● | 操作简单、方便Simple and convenient handling |
| ● | 避免人为的焊接缺陷Manual welding defect can be avoided |
所有的焊接点都是经过专业处理,设备与材料具有可追溯型,手动焊接由受过培训的具有职业资格证书的人员操作。All welding joints are professionally treated; all materials and equipments are certified and traceable; Manual welding is accomplished by trained personnel with appropriate qualifications and certification.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第43页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

2.3.4. 现场管理及现场施工Project management and construction
项目经理根据公司制定的良好工程规范组建项目组来进行质量管理、设计 变更控制、文件和数据的控制、采购控制、过程控制、检查和测试控制、检测 设备的控制、不合格品的控制、纠正和预防措施、安全防护制、包装、运输、储存控制、培训以及验证等。
The project manager sets up the project team according to Clients Company for complete project quality management. Control in change of design, document and data control, procurement control, process control, inspection and control testing, testing of equipment control, control of defective products, corrective and preventive measures, security control, packaging, transport, storage control, training, testing and validation is also strictly monitored
严谨规范的安装队伍是良好工程的保证。Installation team with stringent standards is our Guarantee for good work.
公司富有多年施工经验的施工人员将会按照公司制定的操作规范和客户要求进行施工操作。Our experienced construction staff are rich in experience and they will strictly follow the rules and regulations of operation, and customer requirements.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第44页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||

2.3.5. 先进的检测设备Testing equipment
先进的检测设备确保高质量的完成工程安装,公司拥有完善的焊接质量检测系统。Advanced testing equipment ensure the quality of the completed work.and Welding quality examination system as the follow aim:
| ● | 焊缝质量检测Welded joint quality test |
| ● | 焊缝质量评定Welded joint quality assessment |
| ● | 提供焊接质量检测报告表Provide welding quality inspection reports |
为了确保每一个焊缝的焊接质量。公司结合科学的检测手段配备了先进的检测仪表,如X 光烫伤机、内窥镜、钝化检测仪等。To ensure the quality of welding, the company utilises the scientific detection means equipped with advanced instrumentation, such as the X-ray crack detection machine、Slope meter 、Passivation test and endoscope.
2.3.6. 系统调试和人员培训Training and system commission
根据包含的系统及其范围,我们将编制详细的调试计划。这个计划将包括范围内的每一个系统和设备的描述,包括自控系统;确定个系统进行调试顺利,
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第45页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
及相关前后工序;与项目施工进度结合在一起。
According to the scope and contents of the system, we will establish the detailed commissioning plan. This plan will include descriptions of the scope for each system and equipment, including automatic control system and ensure all systems are commissioned in the correct order, and in their correlation to working procedures.

我们确保所有人员的培训是有文件证明的。直到接受培训的人员能胜任他们各自的任务。
We ensure that all staff training is well documented with certification. Until those trained personnel are capable of performing their respective tasks.
所有人员的培训过程是必须可以被记录的。对于所有人员来说,都需要资格、经验和培训的记录。所有室内和室外的记录都将被保存。
All the training process must be documented for easy reference. For all the staffs they are required to acquire qualifications, experience and training records. All training records will be preserved.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第46页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第2 部分公司介绍及资质文件 Part 2 Company intruduction and Certificate | ||
2.3.7. 验证服务Validation service
作为药品生产过程中的关键系统。洁净管道在设计施工中面对的是严格的规范要求和质量标准。朗脉聘用的是曾经在跨国制药及工程公司服务多年的技术专家,按照国际GMP 规范进行洁净管道系统的验证。
As one of the critical systems in pharmaceutical production facility, clean piping faces more and more stringent requirement on quality assurance and regulatory compliance. Macroprocess employs experts who have served at multinational pharmaceutical and engineering companies and provide qualification/validation services in accordance with international GMP guidelines.
我们在洁净管道系统方面的验证范围包括The scope of our qualification service include:
| ● | 系统的风险分析 System Risk Analysis |
| ● | 确认文件的准备 Preparation of IQ/OQ/PQ protocol |
| ● | 确认执行 Execution of IQ/OQ/PQ |
| ● | 验证报告的编制 Final Qualification Report |
| ● | 现有系统的差距分析及整改建议 Gap analysis on existing system and recommendations on retrofits |
| ● | 回顾性验证 Retrospective Validation |

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第47页/ 共47 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
第 3 部分
Part 3
技术及控制方案
Technical and control system proposal
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
目 录
| 1. 技术方案Technical | 2 |
| 1.1. 项目背景Project Background | 2 |
| 1.2. 管道系统设计标准和规范Design Standards | 4 |
| 1.3. 施工规范和法规Construction Regulations and Guidance .. | 8 |
| 1.3.1. GMP 法规 GMP regulations | 8 |
| 1.3.2. ISPE 指南 | 9 |
| 1.3.3. 工艺管道标准 Process pipe system standards | 10 |
| 1.4. 生产工艺描述Process Description | 10 |
| 1.4.1. 概要General | 10 |
1.4.2. 洁净压缩空气系统Compressed Air (CDA) Distribution .... | 11 |
| 1.4.3. 氮气系统Nitrogen Distribution | 11 |
| 1.4.4. 氧气系统Oxygen Distribution | 12 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
1. 技术方案 Technical
1.1. 项目背景Project Background
沈阳三生制药有限责任公司是集生物药品的研发、生产和销售一体化的高新技术企业。公司的生产基地位于沈阳经济技术开发区,生产车间面积3000 平方米,通过了国家GMP 认证。公司计划建设新的厂房,提高生产能力,来满足日益增长的市场需求。
Shenyang Sunshine Pharmaceutical Company Limited (SUNSHINE) is an integrated biopharmaceutical company engaged in research/development, manufacture and marketing/sales of biopharmaceutical products in China. Its clinical and commercial manufacturing operations are based in Shenyang, China, where Chinese current Good Manufacturing Practice, or cGMP, certified, 3,000 square meters, and state-of-the-art facility resides. SUNSHINE plan to expand the plant in Shenyang to increase the manufacturing capacity and take further advantage of his favorable cost structure.

本次项目为沈阳三生制药有限责任公司的新建车间压缩空气和工艺气体系统的设计、材料、安装、检查和测试、调试、文件和交付。
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
This project is invited tender for design, supply & install of distribution loops for new workshop of SUNSHINE Company. And it is Compressed Air and Process Gas System . this is a turnkey project.
上海朗脉科技有限公司被邀请参加本次洁净管路项目的招标,我们的系统方案以及设计完全按照业主2009 年1 月发布的招标要求以及系统设计要求。
Macroprocess was invited to provide a quotation for the clean piping/utilities systems of this facility. Our proposal is based on SUNSHINE’s tendering specification distributed in January 2009.
本次方案我们提��以下各系统的施工图设计,交货,安装,测试,调试和确认:
Our Scope of Work consists of final detail design, delivery, installation, testing, commissioning and qualification of following packages:
| · | Compressed Air and Process Gas System 压缩空气和工艺气体系统 |
| · | Related control system 相关的自控系统 |
| · | Other related Accessories 其他相关辅助材料系统 |
各循环系统根据客户的要求进行详细设计,具体详见相关技术描述和PID 图。我们按照GMP 的要求对贵公司的系统进行验证,此部分验证内容
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
和报价可以参见报价清单说明。
All loop is designed to meet owner’s requirement and for details see technical description and PID.and we also provide the quotation on validtion to meet GMP requirement. And this part of validation scope and pricelist please refer to the related section.
公司聘认了国外专家专门从事FD A、WHO、欧盟、TGA 等法规的验证工作。“朗脉”的客户涵盖了许多国内外知名的制药企业,其中已有多家客户通过FDA 、EMEA 、TGA 等国外认证,并建立了良好的长期合作关系。
Macroprocess has accomplished an experienced professional team made up of experts on FDA ,WHO,EMEA,TGA validation by recuiting many technical experts both from home and abroad . Macroprocess has served many famous pharmaceutical enterprises both from home and abroad, of which many enterprises have passed foreign validation ( USA FDA, EU EMEA, Australian TGA and etc.) We are also maintaining excellent cooperative relationship with our clients.
1.2. 管道系统设计标准和规范Design Standards
· | According to related national and local government pipeline construction standards and stipulations 根据国家及当地政府管道施工的相关标准及规定 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
| · | WHO about Guidelines for Drinking-water Quality WHO 关于饮用水水质的指南 |
| · | WHO about water treatment guide & aqueous system Examination guide WHO 关于水处理指南和水系统检测指南 |
| · | Drugs production quality control in 1998 amended 药品生产质量管理规范1998 年修订 |
| · | ASE BPE 2002 Edition ASE BPE 2002 年版 |
| · | ICH Q7A GMP of Bulk Pharmaceutical Chemicals, November 2000 ICH Q7A 原料药的GMP, 2000 年11 月 |
| · | USP-29 2006 Edition USP-29 2006 年版 |
| · | FDA federation laws & regulations related stipulation FDA 联邦法规有关规定 |
| · | European pharmacopoeia 欧洲药典 |
| · | According to JBJ23- 96 edition (Code for Construction & acceptance of machinery and equipment installation) 根据JBJ23- 96 版《机械设备安装工程施工及验收规范》 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第5页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
| · | According to HGJ203-83 edition (Code for Construction & acceptance of chemical machinery installation) 根据HGJ203-83 版 《化工机器安装工程施工及验收规范》 |
| · | According to GB50236-98 Edition (Code for Construction & acceptance of field equipment installation & industry pipeline welding) 根据GB50236-98 版《现场设备、工业管道焊接工程施工及验收规范》 |
| · | According to DL5017-93 Edition (Code for Construction, Manufacturing & acceptance of Pressure Piping) 根据DL5017-93 版 《压力管道制造安装及验收规范》 |
| · | According to (Pharmaceutical chemicals overall examination Guide) 根据《医药化学品总体检验指南》 |
| · | According to (Code for Drugs production & management) 根据《药品生产管理规范》 |
| · | According to (Code for Clean room construction and Acceptance) 根据《洁净室施工及验收规范》 |
| · | According to Planning & Design Ordinance of Macroprocess Technology 根据朗脉科技设计条例 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
| · | According to PW piping system design & standards of Macroprocess Technology 根据朗脉科技高纯度管道系统的设计标准 |
| · | According to Construction requirements for Clean Room & High purified water pipeline system of Macroprocess Technology 根据朗脉科技高纯度管道工程洁净厂房施工守则 |
| · | According to guide to Construction safety of Macroprocess Technology 根据朗脉科技安全施工注意事项 |
| · | According to Construction procedure for High purified water piping system of Macroprocess Technology 根据朗脉科技高纯度管道系统的施工程序 |
| · | According to operational procedure for testing of Macroprocess Technology 根据朗脉科技高纯度管道系统的测试作业程序 |
| · | Code & requirements of China, the United States and the European in production of vaccine for Human 用于生产人用疫苗的中国、欧洲和美国的规范和要求 |
| · | Laws and regulations for Electric appliance & installation 电器设备和安装的法规 |
| · | Laws & regulations for safety standards |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
安全标准、法典和规范
| · | According to drawings and relevant requirements of products provided by owner. 根据业主提供的图纸及产品的相关要求 |
1.3. 施工规范和法规Construction Regulations and Guidance
本洁净管道系统项目,将按照以下法规和指南进行设计、施工、调试和验证:This project for sanitary piping system will be assured that its design, construction, commissioning and qualification will be in accordance with the following regulations and guidance:
1.3.1. GMP 法规 GMP regulations
| a) | (SFDA) 药品生产质量管理规范 1998 年修订版 |
Good Manufacturing Practice, revised in 1998
| b) | (SFDA) 药品生产质量管理规范附录 1998 年 |
Annex of GMP, in 1998
| c) | (FDA) 联邦法规第21 篇第210-211 部分,现行的成品药生产质量管理规范 21 CFR parts 210 and 211, Current Good Manufacturing Practice (cGMP) for finished pharmaceuticals. |
| d) | (FDA) 联邦法规第21 篇第11 部分,电子记录和电子签名 |
21 CFR part 11, Electronic Records; Electronic Signatures
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control | ||
| e) | (FDA) 无菌工艺生产的无菌产品的GMP 的行业指南, 2004 年9 月 Guidance for Industry, Sterile Drug Products Produced by Aseptic Processing- Current Good Manufacturing Practice, September 2004 |
| f) | (FDA) 高纯水系统的检查指南,1993年7 月 Guide to Inspections of High Purity Water Systems, July 1993 |
| g) | (Europe) 欧盟最新的GMP European Current GMP |
1.3.2. ISPE 指南
| a) | ISPE 工程指南卷3:无菌生产厂房设施,2000年6 月 ISPE Baseline Guide Volume 3: Sterile Manufacturing Facilities, June 2000 |
| b) | ISPE 工程指南卷4:水和蒸汽系统,2000年12 月 ISPE Baseline Guide Volume 4: Water and Steam System, December 2000 |
| c) | ISPE 工程指南卷5:调试和确认,2000年6 月 ISPE Baseline Guide Volume 5: Commissioning and Qualification, June 2000 |
| d) | ISPE 工程指南卷6:生物制药,2003年1 月 ISPE Baseline Guide Volume 6: Biopharmaceuticals, January 2003 |
| e) | GAMP 5 : 制药生产中的自动化系统验证的良好规范 Good Automated Manufacturing Practice for Validation of Automated Systems in Pharmaceutical Manufacture |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
1.3.3. 工艺管道标准 Process pipe system standards
| a) | GB50235-97 工业金属管道工程施工及验收规范 Code for construction and acceptance of industrial metallic piping. |
| b) | ASME BPE-2005 生物工艺设备 Bioprocess equipment |
1.4. 生产工艺描述Process Description
1.4.1. 概要General
以下技术描述基于UR S(用户需求规格)。我们提供各系统的详细设计包括设备,控制系统,以及各类仪表,并按照标准提供整个系统的设计、安装、交货、调试、验证、培训方案。我们的设计图纸(包括管路平面走向示意图、系统流程图和P&ID’s 等)将作为主要设计依据。另外,我们提供必要的条款,确保整个设计系统符合生产要求。
The following description is based on the URS. Upon award of the bid, we shall carry out detail design of the entire system including equipment, controls, and instrumentation, and use the approved design as guideline for the procurement, delivery, installation, commissioning and qualification of the system. Our design drawings, including Piping and Instrument Diagrams (P&ID's), piping layout drawing, piping isometric drawing and so on, will become the master documents to which and for which the Chengdu Institute’s documents will be modified. We shall provide necessary items to ensure that the overall system performs to meet the process requirements.
整个系统的符合中国、WHO、EU和FDA 的标准。
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
The sanitary aspects of the system will meet GMP requirements for China, WHO, EU and FDA.
1.4.2. 洁净压缩空气系统Compressed Air (CDA) Distribution
通过洁净压缩空气管路,压缩空气分配给各个使用点。使用点采用球阀,管子及阀门全部为316L 不锈钢材质,为了防止污染的压缩空气回流,管路中应设置止回阀。
Piping will connect CDA to all use points from air compressor. Interface point with the general CDA pipe will be ball valves. Pipe material for general CDA system is 316L Stainless Steel. To prevent back-flow contamination to general CDA system, check valves shall be installed after the connected valves
1.4.3. 氮气系统Nitrogen Distribution
通过氮气管路,氮气分配给各个使用点。使用点采用球阀,管子及阀门全部为304 不锈钢材质,为了防止污染的氮气回流,管路中应设置止回阀。
Piping will connect nitrogen to all use points from nitrogen generation plant. Interface point with the general nitrogen pipe will be ball valves. Pipe material for general nitrogen system is 304 Stainless Steel. To prevent back-flow contamination to general nitrogen system, check valves shall be installed after the connected valves.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第3 部分技术及控制方案 Technical and control system | ||
1.4.4. 氧气系统Oxygen Distribution
通过氧气管路,氧气分配给各个使用点。使用点采用球阀,管子及阀门全部为304 不锈钢材质,为了防止污染的氧气回流,管路中应设置止回阀。
Piping will connect Oxygen to all use points from Oxygen generation plant. Interface point with the general Oxygen pipe will be ball valves. Pipe material for general Oxygen system is 304 Stainless Steel. To prevent back-flow contamination to general Oxygen system, check valves shall be installed after the connected valves.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共12 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
第 4 部分
Part 4
验证方案
Validation
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
目 录
Index
| 1. 介绍Introduction | 3 |
| 2. 验证范围Work scope of validation | 3 |
| 3. 验证方法Validation approach | 5 |
| 3.1. 总要求General Requirement | 5 |
| 3.2. 风险评估Risk Assessment | 6 |
| 3.3. 追溯矩阵表Traceability Matrix | 8 |
| 3.4. 设计确认Design Qualification | 9 |
| 3.5. 工厂验收测试 Factory Acceptance Test | 12 |
| 3.6. 现场验收测试 Site Acceptance Test | 14 |
| 3.7. 校准Calibration | 14 |
| 3.8. 安装确认Installation Qualification | 17 |
| 3.9. 运行确认Operation Qualification | 19 |
| 3.10. 控制系统的验证 Validation of Automatic Control System | 21 |
| 3.11. 性能确认Performance Qualification | 21 |
| 4. 质量项目计划Quality and Project Plan | 22 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
验证服务范围
Work Scope of Validation Service
压缩空气、氮气、氧气和二氧化碳分配系统
Distribution System of Compressed air,
Nitrogen, Oxygen and Carbon Dioxide
提供方:上海朗脉科技有限公司
Provided by: Shanghai Macroprocess Co., Ltd.
January 2009
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
1. 介绍Introduction
沈阳三生制药有限责任公司(“沈阳三生”) 将新建一个生物制药车间,现对压缩空气、氮气、氧气和二氧化碳分配系统的详细设计、施工、调试和确认(验证)的工程服务项目进行招标。
Shenyang Sunshine Pharmaceutical Co., Ltd. (3SBIO) will build one new biopharmaceutical plant, and now invite the bidding for the project of detailed design, construction, commissioning and qualification (validation) of distribution system of compressed air, Nitrogen, Oxygen and Carbon dioxide.
上海朗脉受邀参与投标,本文件将为本次的项目中验证范围进行做出说明,并给出最优的报价。
Shanghai Macroprocess is invited to bid for this project. This document will define the work scope of validation activities and give a best quotation.
2. 验证范围Work scope of validation
我们提供的验证服务范围,见以下验证矩阵表。
We provide the validation services shown in Validation Matrix.
表Table 1-验证矩阵表Validation Matrix
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
| 系统System | RA | TM | DQ | FAT | SAT | IQ | OQ | PQ* |
压缩空气分配系统 compressed air distribution system | √ | √ | √ | - | √ | √ | √ | √ |
氮气分配系统 Nitrogen distribution system | √ | √ | √ | - | √ | √ | √ | √ |
氧气分配系统 Oxygen distribution system | √ | √ | √ | - | √ | √ | √ | √ |
二氧化碳分配系统 Carbon dioxide distribution system | √ | √ | √ | - | √ | √ | √ | √ |
备注Remark:
| 1) | RA- 风险评估Risk Assessment ; TM- 追溯矩阵表Traceability Matrix; DQ- 设计确认Design Qualification; FAT -工厂验收测试Factory Acceptance Test; SAT- 现场验收测试Site Acceptance Test; IQ-安装确认Installation Qualification; OQ- 运行确认Operational Qualification; PQ- 性能确认 Performance Qualification |
| 2) | PQ 方案由朗脉提供,实施由客户负责。 PQ protocols are prepared by Macroprocess, but the execution will be done by client. |
详细的验证要求描述见后面章节。
Detailed validation requirement description will be found in the following section.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
3. 验证方法Validation approach
3.1. 总要求General Requirement
我们将按照以下V 模型对关键系统开展验证工作。
We will conduct the validation/qualification activities according to the following V-Model.
Figure 1- V-model
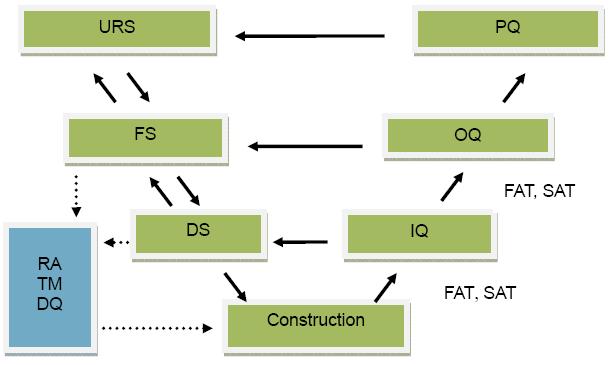
在设计开始之时,验证工作也将同时开展。在详细设计完成的后期,进行设计确认,并对系统进行风险评估,编写追溯矩阵表。在采购和施工过程中,任何的关键性变更(影响产品质量或安全的变更) , 需要提交给验证人员进行审核。
Validation activities should begin with design. In the end of detailed design stage, Design Qualification and Risk assessment will be
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第5页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
conducted, and also Traceability matrix will be prepared. During purchase and construction, any significant change(impact product quality or safety) should be submitted to validation person for review. 系统施工结束后,进行调试,在合适的时候组织客户相关人员,进行FAT 或SA T。然后,进行安装确认和运行确认。性能确认,由客户自己负责实施。 When the finish of construction and commissioning, we will invite related persons from client to conduct FAT or SAT. Then Installation and Operational qualification will be done by us with witness by client. Performance qualification will be done by client own.
3.2. 风险评估 Risk Assessment
风险评估将决定哪些系统/部件需在GEP 基础上还须进行确认,哪些仅仅需要按照GEP 规范进行安装和调试。 Risk assessment is the process of determining which systems/components should be subject to qualification practice in addition to Good Engineering Practices (GEP) and which systems/components should be commissioned in accordance with GEP.
评估通过衡量某一系统对产品质量的影响进行。在系统完成评估之后,系统的部件也将被评估,来确定它们是否对保证和维持产品质量有关键影响。 有直接影响的系统和部件将进行确认,而有间接影响的仅按照GEP 进行设计,
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
安装和调试。
The assessment is made by evaluating the impact that a system has on the quality of the product. After the system assessment, the components of the system are evaluated to determine if they are critical to establish or maintain the quality of the product. The appropriate qualification practices are then applied to the ‘direct impact’ systems and components, while the ‘indirect impact’ systems and their components shall be designed, installed and commissioned according to GEP.
根据这种方式,工作的重点集中在对产品质量有关的系统和部件上。评估的完成途径多种多样,由每个公司最终确定。
Following this approach, the appropriate effort and focus can be maintained on the quality impacting systems and components. The assessment process can be done in a variety of ways, which will be ultimately defined by each individual operation company.
风险评估的基本步骤Basic Steps of Risk Assessment:
| - | 了解系统或设备,以及工艺流程 Identification of System/Equipment, Process |
| - | 识别可能出现风险、产生的原因、带来的影响 Identification of the risk event, cause and effects |
| - | 评估该风险出现的可能性 Evaluation of the probability that the event occurs |
| - | 评估该风险带来的影响的严重程度 Evaluation of the Impact of the Risk |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
| - | 评估该风险被发现的可能性 Evaluation of the probability of detection of the event |
| - | 建议采取的措施 |
Suggested actions
报告Reporting:
风险评估的结果将报告下列内容:
The results of the risk assessment will be reported with the following typical contents:
| - | 目的 |
Objective
| - | 风险评估的参与者 |
Participants in the risk analysis
| - | 工艺参数、功能、关键组件 Process parameters, Function and Components |
| - | 潜在的风险 |
Potential risk
| - | GMP 关键性的判断 |
Identification if it is GMP critical
| - | 风险的原因和后果 |
Cause and consequences of risk
| - | 风险评估(出现的可能性、影响的程度、被发现的可能性) Risk Assessment (probability of occur, Impact, probability of detection) |
| - | 处理的措施(如测试要求、特别措施等) Measures(e.g. Test requirement, specific measures etc.) |
3.3. 追溯矩阵表Traceability Matrix
追溯矩阵表对验证项目的质量关键及基本要求自用户需求标准到功能标准直至测试方案进行追溯。
Traceability Matrix traces the quality critical and essential
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
requirements from the User Requirements Specification (URS) to the functional specifications and onto the test protocols for this validation project.
3.4. 设计确认 Design Qualification
DQ 是对系统的设计进行审核,以确认它们符合预定功能及运行要求,并符合GMP 要求。
DQ is the review for the design of system which ensures that these are according to advanced function and operation requirement as well as GMP requirement.
DQ是贯穿于项目设计阶段的一个持续的过程,它保证与特定设备和系统相关的所有的设计文件符合现行GMP的要求。我们将积极采取行动,保证项目的设计符合国际GMP 及客户的内控标准。
DQ is a continuous progress throughout project design stage. DQ ensure the design documents related to specific equipment and system according to current GMP requirement. We will act for it and ensure the project design meet the international GMP and internal standard of Client.
DQ 的关键工作是对系统的设计进行GMP 审查。该审查由朗脉验证专家主持,双方人员均参加。此审查将提供一个正式文件,证明设计按照GMP 各
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
方面要求。会议的记录,完整的文件检查清单及变更清单将是DQ 的质量记录,被妥善保存。
The critical work of DQ is GMP review to system design. This review is conducted by specialist from Macroprocess, both sides would join it. This review will provide an official document to prove the design meet the GMP requirement of each aspect. Meeting memo, complete document check list and change list will be quality record of DQ and will be preserved appropriately.
设计确认(DQ )并非法规的恶强制要求。然而,从正式性和加强的设计审核角度,我们推荐执行DQ。
Design Qualification (DQ) is not a mandatory requirement of the regulations. However, in this formal format or through enhanced design reviews, it is recommended that it be carried out.
DQ 通过对系统进行有组织的设计审查来进行。这种审查将在严格要求下进行,因为它在IQ、OQ之前能够更好地发现设计问题或不符合要求的方面,否则将对项目的工期和费用造成巨大影响。
The DQ will be executed by applying a structured review of the design of the systems. The review should be rigorous as it is better to discover any design problems or non-conformance issues before reaching IQ or OQ, where remedial work may have a significant
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
impact on the project schedule and cost.
方案将用采用书面格式。详细的检查清单将确保设计和文件的所有方面均被涉及。方案被各方正式签署后执行。
A protocol will be prepared with a written description of the format and approach for the reviews. Detailed checklists will be prepared to enable all aspects of the design and documentation to be interrogated. The requirements of the risk assessment and cGMP reviews will also be reviewed to ensure that their requirements are incorporated. The protocol will be formally signed off as approved prior to execution and post execution by all parties.
DQ 文件的通常标准包括: Typical criteria, which would be used to generate the DQ protocol are:
| - | 设计确保满足GMP 的要求 Design satisfies GMPs and regulatory requirements |
| - | 设计符合性能标准 Design meets performance criteria |
| - | 材料材质 Materials of construction |
| - | 清洗要求 Cleaning requirements |
| - | 操作的可靠性和有效性 Reliability and Efficiency of operation |
| - | 设备安装 Equipment Installation |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
| - | 系统调试 System commissioning |
| - | 关键仪表的参数,量程和精度是否满足工艺要求和控制要求 parameters of critical instruments, verify if the measuring range and accuracy satisfy the process and control requirement |
| - | 关键组件的结构 Structure of critical components |
| - | 文件要求 Documentation requirements |
3.5. 工厂验收测试 Factory Acceptance Test
工厂验收测试应该关注设备关键的方面,包括进行测试活动。这些测试将在设备起运之前进行,以保证该设备已按照设计标准进行制造,并提供所需要的文件。因此,FAT 可帮助避免可能影响现场测试的问题的产生。
Factory acceptance test could be applied to any critical aspect and includes test activity, that will be carried out before the shipment, to get assurance that a new critical equipment/ package has been built following the design specifications and it is provided with the required documentation; moreover the FAT will help to avoid problems that could affect the on site activities progress.
FAT 可适用于所有关键的设备或设备包。
FAT could be applied to any critical equipment or components.
FAT 将包括IQ OQ 所包含的一些测试内容。任何不受运输或安装所影响的测试内容,如果其得到合适的执行、复核和记录,在以后的确认中,可以不需重复进行。下面是一些被认为可以作为确认目的的测试内容,但不仅限于
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空��及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
此:
FAT will already include some tests foreseen by the IQ and OQ; any test, which reliability will not be affected by the shipment and the installation could not be repeated for qualification purpose, if properly executed, witnessed and documented. Test activity that typically could be considered validated for qualification purpose, but not limited, are the following:
| - | 文件确认 Documents verification |
| - | 图纸的确认(P&ID、布置图、电子和气路图) Drawings verification (P&ID, Lay Out, electrical and pneumatic scheme) |
| - | 材料和表面抛光的检查 Materials and surfaces finishing verification |
| - | 控制系统图形界面的检查 Control system graphical pages check |
| - | 报警和联锁 Alarms and interlocks |
FAT 将要求进行记录;相关的测试报告或方案,将被提交给客户审核和批准。
FAT 将在客户批准后进行。
FAT will be documented; the relevant test report or protocol, will be submitted to client for review, comments and approval; FAT execution will begin after client approval.
本项目不需要进行FAT。
It doesn’t need to conduct FAT for this project.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第13页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
3.6. 现场验收测试 Site Acceptance Test
此工作适用于关键的系统或设备。 SAT 包含动态的和静态的测试活动,测试活动在现场由供应商在移交系统给客户之前进行。每一项SAT 工作将被文件记录下来。我们将提交测试文件(定义为SAT 报告或方案)给客户,以审核和批准。
Activity Applicable to critical system or equipment; SAT consists of Dynamic and static testing activities performed on site by the vendor before hand over the system to the client. Each SAT activity will be documented. Macroprocess will submit the test document (defined as SAT report or protocol) to Client (for comments and approval).
那些不会因为系统/设备的改变而影响可靠性的测试,如果已得到正确的执行、证明和文件记录,那么在验证过程中不必重复进行。
All tests which reliability will not be affected by the system/equipment modification could not be repeated for qualification purpose, if properly executed (in conformity with Qualification Protocols), witnessed and documented.
3.7. 校准 Calibration
因为是新的安装, 所有的仪器仪表和变送器应被校准并产生校准证书。
Since this is a new installation all transmitter instruments will be calibrated, and calibration certificate will be produced.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第14页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
所有不受安装影响的仪器仪表将经过校准后送到现场,并提供校准证书(溯源为NIST 或中国国家计量基准)。其它的仪器仪表将在现场使用计量标准进行校准。在任何情况下,应提供参考仪器的校准证书和校准程序。
All instruments not affected by the installation will be bench calibrated and then, supplied to the field, provided with NIST or Chinese National measurement standards traceable calibration certificate; the others will be calibrated in field using traceable standards references. In a case copy of the reference instrument calibration certificate and approved procedures will be provided.
下列是对不同等级仪器仪表的处理方法。
On the following different dedicated approach for class of instruments.
不受安装影响的仪表
Instruments not affected by the installation:
温度和压力变送器,需要校准证书(溯源为NIST 或中国国家计量基准)。确认工作,仅为确认安装情况(是否有合适的配置、范围、计量单位、信号)和带有相关显示器或相关监督和控制站的信号定位。
Temperature and pressure transmitters; for which will be required (to
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第15页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
the vendor) a bench calibration certificate (NIST or Chinese National measurement standards traceable) and during the qualification will only be verified the set up (if smart the relevant configuration, range, engineering unit, signals) and the signal alignment with the relevant indicator or the eventual supervision and control station
会受安装影响的仪表
Instruments affected by the installation:
流量和液位变送器,不需要实验室校准。校准将在现场使用可溯源为NIST 或中国国家计量标准的仪器进行校准。
Flow rate and level transmitters; for which will not be required a bench calibration. The calibration will be performed after the installation in field using NIST or Chinese National measurement standards traceable reference instruments.
电导仪和PH 计将在现场通过使用相关的指示器与标准溶液进行确认/校准。conductivity and pH probe will be verified/calibrated in field reading the measurement using the relevant indicator in comparison with a reference standard solution.
仪表的校准工作不属于验证工作。本项目的仪表校准工作由哪方负责,将由合同进行约定。
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第16页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
Calibration is not included into Validation work. The responsible party for calibration work will be according to the contract.
3.8. 安装确认Installation Qualification
安装确认将检查每一个设备/系统的安装是否符合设计和相应的GMP 规范。IQ 将确认所有的需要用于设备/系统的管理和操作的文件的可用性。
The Installation Qualification will determine that each equipment/system has been installed in accordance with the design (if new or renovated) and the applicable current good manufacturing practice (either if new or existing). IQ will confirm the availability of all the required documentation useful to manage and operate with the equipment/system itself.
在执行IQ 之前,系统的机械安装工作已完成。
Prior to the IQ, systems will have to be mechanically completed.
IQ 方案编写 IQ Protocols Preparation:
在所有对产品质量有影响的系统上都要进行IQ。每个IQ 方案都是针对特定系统的,但是所有方案都基本包含以下内容:
For all of the system that affect product quality must undergo IQ. Each IQ protocol is targeted at specific system, but all protocols usually contain the following elements:
| - | 方案批准页Protocol Review and Approval Sheet |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第17页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
| - | 目的 Purpose |
| - | 验证范围 Scope of Validation |
| - | 职责 Responsibility |
| - | 参考文件Reference documents |
| - | 系统描述 System description |
| - | 文件检查 Document verification |
| - | 系统图和布局图检查 P&ID and Layout verification |
| - | 设备确认 Equipment verification |
| - | 仪表确认 Instrument verification |
| - | 材质和表面抛光的检查 Materials and Surface finishes verification |
| - | 组件结构的检查 Components construction verification |
| - | 死角的确认 Deadleg verification |
| - | 安装情况的检查 Installation verification |
| - | 公用设施的检查 Utilitites verification |
| - | 人员的确认 Personnel identification |
| - | 偏差报告 Deivation report |
| - | 实施结果批准 Execution review and approval |
IQ 接受标准 IQ Acceptance Criteria:
| - | 所有的参与IQ 执行的人员在实施前须经相关培训。 All personnel participation in IQ execution must pass through the correlation pre-job training. |
| - | 所有文件(如操作手册,维修手册,焊点图,焊接记录,焊接检查,坡度确认,压力测试文件,内窥镜检查测试记录等)将被检查,文档位置将被记录。 All documents (such as operating manuals, maintenance manuals, solder joint plans, records of welding, welding inspection, slope confirmation, pressure test document, endoscopies test records, etc.) will be examined, file location will be recorded. |
| - | 所有的图纸(如管道和仪表流程图,管道轴测图,电气图,气动图等)和材质证明等将被检查,文档位置将被记录。 All drawings (For example pipeline and measuringappliance flow chart, three dimensional pipelineschematic drawing, electrical chart, pneumatic operated |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第18页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
chart and so on) and with the material quality proved and inspection file location will be recorded.
| - | 特定系统的备件清单须完备 The detailed list of spare parts for specific system must be completed |
| - | 所有的设备,仪表和配套将被检查并须符合预设标准 All equipment, instrument and accessory will be inspected and they are required to meet the pre-set standards |
| - | 仪表的校正已经成功完成 Instrument calibration has been successfully completed |
3.9. 运行确认Operation Qualification
运行确认是按照预设的操作范围和机械性能进行,通过记录的文件,证明系统和设备能正确运行。
OQ could be executed and operated according to the pre-set operation arrange and mechanism performance through the recorded documentations prove that the system and equipment have been proper operated.
OQ 方案编写 OQ Protocols Preparation:
在所有对产品质量有影响的系统上都要进行OQ。我们会为每一个系统写一份OQ 方案。每个OQ 方案都是针对特定系统的,但是所有方案都包含以下内容:
All systems which influence the product quality are needed to execute OQ. We will write a OQ protocol for each system. Each OQ protocol aims at specific system separately, but all protocols are including:
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第19页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
| - | 方案批准页 Protocol Review and Approval Sheet |
| - | 目的 Purpose |
| - | 验证范围 Scope of Validation |
| - | 职责 Responsibility |
| - | 参考文件 Reference documents |
| - | 系统描述 System description |
| - | 验证仪器的检查 Validation Instruments verification |
| - | 功能检查 Function Verification |
| - | 报警和联锁的检查 Alarms and interlocks verification |
| - | 人员的确认 Personnel identification |
| - | 偏差报告 Deivation report |
| - | 实施结果批准 Execution review and approval |
OQ 接受标准 OQ Acceptance Criteria:
| - | 所有的参与IQ 执行的人员在实施前须经相关培训。 All personnel participation in IQ execution must pass through the correlation pre-job training. |
| - | 所有的OQ 工作将按照已批准的方案执行 All OQ activities should be conducted according to approved protocols. |
| - | OQ 测试仪器应被校准过的, 相关的证书应可溯源NIST 或中国国家计量基准, 并附在相应的方案中 OQ Test instruments should be calibrated; the relevant certificate should be NIST or Chinese National measurement standards traceable to the National Institute of Standard Technology and attached to the relevant protocol. |
| - | 系统报警和联锁的情况应符合设计标准 The system’s alarms and interlocks should operate in accordance with the design specification. |
| - | 系统功能运行应符合设计标准 The system’s function should operate in accordance with the design specification. |
| - | 系统所用的公用设施应当在规定范围以内( 温度、压力、流量) Utilities, used by the system, should be available at the battery limit within the specified range (temperature, pressure, flow-rate). |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第20页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
3.10. 控制系统的验证 Validation of Automatic Control System
所有的跟产品生产、质量控制和分销有关的自控系统及其他数据采集,存储和处理系统都属于验证的范围。自控/计算机系统的验证将依据美国FDA (包括21CFR 第11 部分有关电子记录和电子签名的规定)、欧盟的GMP 指南和良好自控生产规范(GAMP)进行。
All automatic control system and other data collection, storage and treatment system related to production, quality control and distribution are involved in the scope of validation. The validation of automatic system would be held according to the US FDA (21 CFR Part 11), GMP guideline of EU and GAMP.
3.11. 性能确认 Performance Qualification
OQ 的成功完成之后,将进行PQ 工作。PQ 将整合考虑规程、人员、物料、设备和工艺。
Upon successful completion of the OQ, Performance Qualification will be carried out. The PQ will integrate procedures, personnel, material, equipment, and process.
测试、规程和接受标准将在系统的挑战性实验之前确立,并在执行完PQ 之后进行审核。
Tests, procedures and acceptance criteria will be defined before challenging the systems and review after PQ execution.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第21页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第4 部分验证方案 Validation Service | ||
PQ 将证明设备、工艺步骤和参数、公用设施、原料、中间体和标准操作程序可以生产出预先所建立的生产和质量要求。
PQ will demonstrate that equipment, process phases and parameters, utilities, raw materials, intermediates and standard operating procedures allow to reach the pre-established manufacturing and quality requirements.
测试可能包括化学的、物理的、微生物的分析。
Testing may include analysis for chemical, physical, microbiological constituents.
任何显著变化的情况将被要求进行审核和调查,以最终决定适当的纠偏措施。
Any outstanding condition will be identified for review and investigated to eventually determine the appropriate corrective action.
4. 质量项目计划Quality and Project Plan
参见附件See attachment.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第22页/ 共22 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
第 5 部分
Part 5
施工组织
Construction Scheme
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
目 录
Index
| 1.工程施工人员组织设计Staff organization | 2 |
| 1.1. 人员组成结构图Organization Chart | 2 |
| 1.2. 项目组成人员角色及职责team and responsibility | 2 |
| 1.3. 验证经理资质Validation manager qualification | 5 |
| 1.4. 施工人员组织Construction organize | 10 |
| 1.5. 项目经理负责人资质project manager qualification | 10 |
| 1.6. 质量控制组织图Quality control | 15 |
| 2.施工设备材料投入计划Tools list | 16 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
1.工程施工人员组织设计Staff organization
1.1. 人员组成结构图 Organization Chart
目前拟定结构如下:At present,we study out the staff organization chart as the following:

1.2. 项目组成人员角色及职责team and responsibility
 项目经理 Project manager
项目经理 Project manager项目负责人;协调和负责整个项目执行,确保项目执行达到各节点要求;安全第一负责人;是业主用户和上海朗脉科技有限公司的联系人;确认来往资料及传真,负责所有项目资料的归档。
Take charge of the whole project , ensure the project proceed to the right requirement and conduct the construction staff. As the window between the owner and Macroprocess; clarify the letter
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
and fax and all document of the project.
 项目组 project team
项目组 project team负责监督完成系统的工艺设计、自动化控制系统设计、CAD 图纸制作等的设计,出图、资料准备、组态及实施监控,完成整个项目设计工作。
Work together to take charge of all design 、drawings of technical and control system. Also prepare the document 、configration and inspection to achieve the design task.
 施工经理 construction manager
施工经理 construction manager负责项目系统的现场调度、实施、安装、检测和系统正常运行等现场技术支持服务,同时负责协调工程培训事宜,包括培训日程、地点、人员安排等。该职位作为项目实施过程中朗脉公司人员直接与业主沟通。
Take charge of technical support for implement 、installation 、test and operation of whole project, and also response to training including the p lace、schedul e。This position is used to communicate with owner directly.
 品质管理和质量 quality manager
品质管理和质量 quality manager负责协调和监督工程实施过程中项目质量符合系统设计要求,满足客户需求,同时监控项目实施中的安全操作,确保人力物力的安全。
Ensure the quality meet the owner’s requirement and comply with sepecification, and also control the operation in the construction work, ensure the safty of human resource and material resources.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
 采购经理 purchase manager
采购经理 purchase manager具体负责系统设备的预算、采购、物流等管理和支持工作,是项目正常实施的物流枢纽。
Take charge of project’s budget 、purchase 、logistic and other support. As a hinge of all project.
 验证经理 Validation manager
验证经理 Validation manager评估项目质量要求和提供项目实施验证方案确保符合顾客要求,验证与行业规范相符合。
Evaluate the whole project quality requirement and provide the validation proposal to meet the client regulation requirement.
 用户项目经理 owner project manager
用户项目经理 owner project manager为了使双方的共同渠道畅通,顾客需任命一名项目经理,该项目经理要保证用户的招标承诺实现。并且确认上海朗脉科技有限公司的项目经理定期与用户沟通。用户项目经理是用户与上海朗脉科技有限公司的交流窗口。
In order to push the project smothly,client need to appoint one project manager to ensure promises of whole work. And also need to communicate with Macroprocess stablely.this postion is also a window between owner and client.
 用户工程组 user team
用户工程组 user team用户工程组为上海朗脉科技有限公司提供实施项目质量保证的支持工作。作为设计、质量、验证、工程、安全卫生环保的沟通机构。
This team shall provide support to Macroprocess to ensure
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
quality.
 用户工程师代表 Engineer representation
用户工程师代表 Engineer representation工程的成功需要用户工程组的努力及配合,参与设计方案的准备有助于他们理解系统,而系统最终要由他们来维护。参与设计及以后的培训使他们能够更加彻底的了解系统。
Engineer representation of client need join with Macroprocess to understand the whole design and the proposal, and they will maintain the system by themselves. That to take part in the design is good for them to familiar with whole project.
1.3. 验证经理资质Validation manager qualification
验证是项目不可或缺的组成部分,验证的过程将直接确保项目的实施是否符合国际法规的要求,确认是否满足验证指定要求。上海朗脉科技有限公司的验证部门派出质量总监全权与其验证团队监督项目的执行。
Validation is one important part of the whole project, Macroprocess will delegate quality director with his team to monitor the project. And his qualification as the following:
他的履历情况如下:
验证经理 Validtion manager 陈国笋 Chen Guosun
 学历教育 Education and Qualification:
学历教育 Education and Qualification:浙江大学药学院 药学专业本科 执业药师
Bachelor in Pharmacy,
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第5页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
Pharmacy College of Zhejiang University
Licensed Pharmacist
 专业技能Professional Competence:
专业技能Professional Competence:有8 年多的制药行业经验,尤其在验证和质量管理方面。Has over 8 years of professional experience in Pharmaceutical Industry, especially in Validation and Quality Management.
| ü | 熟悉中国、美国和欧盟GMP 法规和指南。Be familiar with Chinese, USA and EU GMP Regulations and Guidance. |
| ü | 有GMP 培训和验证的经验,如HVAC 系统的确认、水系统的确认、设备确认、清洗验证和工艺验证等。Experience in GMP training and validation (i.e. HVAC system qualification, water system qualification, equipment qualification, cleaning validation, process validation etc.). |
| ü | 有在制药公司进行生产和质量管理的经验。Experience in Production and Quality Management in Pharmaceutical Company. |
 主要项目Key Projects:
主要项目Key Projects: 石家庄欧意药业 口服固体制剂 Shijiazhuang Ouyi Pharma. Oral Solid Dosage Forms
石家庄欧意药业 口服固体制剂 Shijiazhuang Ouyi Pharma. Oral Solid Dosage Forms验证经理。负责按照中国GMP 和FDA cGMP 的要求领导验证工作。对HVAC 系统和纯化水系统进行设计审核。编写HVAC 系统、洁净室和纯化水系统的IQOQ 方案,并实施。
Validation Manager
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, FDA cGMP.
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, FDA cGMP.
Design review of HVAC system and purified water system.
IQOQ Protocols writing and execution of HVAC system, Clean room and purified water system. IQOQ protocols
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
writing and execution of HVAC computerized system.
 杭州九源基因 原料药 Hangzhou Jiuyuan Bio-Gene. API
杭州九源基因 原料药 Hangzhou Jiuyuan Bio-Gene. API验证经理。负责按照中国GMP 和FDA cGMP 的要求领导验证工作。
纯化水和注射用水系统的设计审核。纯化水和注射用水系统的IQOQ 方案的审核。
Validation Manager
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, FDA cGMP.
Design review of Purified water, WFI systems. Review of IQOQ protocols of purified water, WFI systems.
 齐鲁安替制药 无菌原料药 Qilu Antibiotics Pharma Sterile API
齐鲁安替制药 无菌原料药 Qilu Antibiotics Pharma Sterile API验证经理。负责按照中国GMP 和FDA cGMP 的要求领导验证工作。
纯化水、注射用水和纯蒸汽系统的设计审核。
Validation Manager
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, FDA cGMP.
Design review of Purified water, WFI system and pure steam system.
 成都生物研究所 疫苗
成都生物研究所 疫苗Chengdu Institute of biological product Vccine
验证经理。负责按照中国GMP 和WHO GMP 的要求领导验证工作。
纯化水、注射用水、纯蒸汽和压缩空气的设计确认。
Validation Manager
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, WHO GMP.
DQ of Purified water, WFI, Pure steam, compressed air.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
 浙江华海药业 口服固体制剂
浙江华海药业 口服固体制剂Zhejiang Huahai Pharma. Oral Solid Dosage Forms
GMP 咨询师。按照中国GMP、FDA cGMP 和欧盟GMP 对概念设计进行GMP 审核。(已于2007 年通过FDA GMP 检查)
GMP Consultant
GMP review of conceptual design in accordance with the requirement of Chinese GMP, FDA cGMP and EU GMP. (Already passed FDA GMP Inspection in 2007)
 大连美罗制药厂 滴眼剂
大连美罗制药厂 滴眼剂 Dalian Merro Pharma. Eye Drops
验证经理。负责按照中国GMP 和欧盟GMP 的要求领导验证工作。
负责验证主计划的编写。编写HVAC 系统、洁净室、纯化水、注射用水系统的IQOQ 方案,并实施。
Validation manager
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, EU GMP. Responsible for VMP writing.
IQOQ protocols writing and execution of HVAC system, Clean room, PW/WFI system.
 北京四环生物 疫苗
北京四环生物 疫苗Beijing Four ring Bio-pharma Vaccine
验证经理。负责按照中国GMP 和欧盟GMP 的要求领导验证工作。
编写HVAC 系统、洁净室的IQOQ 方案,并实施。
Validation manager
Responsible for Design GMP review and VMP writing in accordance with the requirement of Chinese GMP, EU GMP.
IQOQ protocols writing and execution of HVAC system
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
and Clean room.
 石家庄恩必普药业 软胶囊
石家庄恩必普药业 软胶囊Shijiazhuang NBP Pharma. Soft capsule
验证经理。负责按照中国GMP 和FDA cGMP 的要求领导验证工作。
负责验证主计划的编写。编写HVAC 系统、洁净室、纯化水系统的IQOQ 方案,并实施。
工艺验证、清洗验证和分析方法验证的培训。
工艺设备的计算机系统验证。
Validation manager
Responsible for Lead Validation work in accordance with the requirement of Chinese GMP, FDA GMP. Responsible for VMP writing.
IQOQ protocols writing and execution of HVAC system, Clean room, PW system.
Training of process validation, cleaning validation and analytical method validation.
Computer validation of process equipments.
 北京费森尤斯卡比医药 片剂
北京费森尤斯卡比医药 片剂Beijing Fresenius Kabi Pharma. Tablet
验证工程师。HVAC 系统和洁净室的DQ/IQ/OQ 方案编写和实施。
(已于2005 年通过EMEA 的GMP 检查)
Validation engineer
DQ/IQ/OQ protocols writing and execution of HVAC system and Clean room.
(Already passed EMEA GMP Inspection in 2005)
 上海莱士 血制品
上海莱士 血制品Shanghai RAAS Blood Products
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
验证工程师。HVAC 系统、纯化水和注射用水系统的DQ/IQ/OQ 方案编写和实施。
Validation engineer
DQ/IQ/OQ protocols writing and execution of HVAC system, Purified water and WFI systems.
1.4. 施工人员组织 Construction organize
为保质,按时完成贵公司的管道系统的施工,特组织人员如下:In order to guarantee the quality, & to complete the distribution loop on time, we especially organize our personnel as follows:
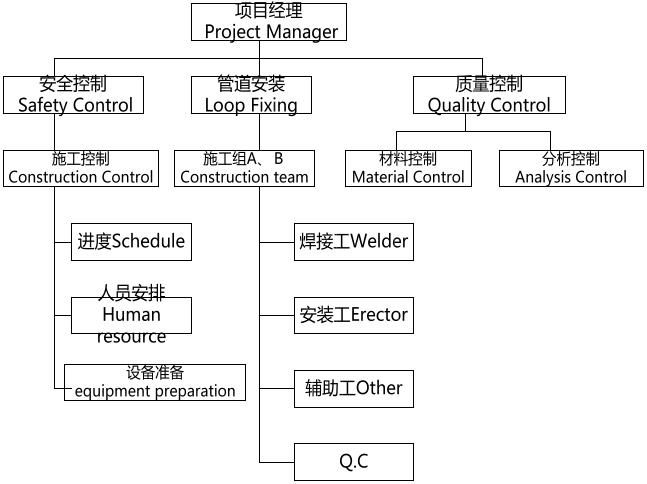
1.5. 项目经理人负责人资质 project manager qualification
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
针对本次项目的情况,目前暂时确定选择经验丰富的程千振经理作为项目经理人负责整个项目的实施。他的履历情况如下:
Evaluating this project,we choose professional manager named Chen Qianzhen as project manager to take charge the technical of whole peoject. And the folloing is the qualification:
Cheng Qianzhen
程千振
Education and Qualifications 学历教育
| • | Bachelor of Science,Mechanic Design (1992-1996) 理学学士,机械设计(1992-1996) Shandong University 山东大学 |
Professional Experience 专业经验
Mr. Cheng has over 10 years of professional experience in project co-ordination, procurement and planning, facility design, generation of facility design drawings, commissioning in pharmaceutical engineering field.
程千振先生在制药工程领域内已有10 年以上专业的项目统筹经验,其中包括采购计划,设施设计,生产设备的设计图,设备调试等。
Key Projects 重点工程
MACROPROCESS TECHNOLOGY CO., LTD.
上海朗脉科技有限公司
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
Feb 2005 – 2005 年2 月至今 | CHENGDU INSTITUTE OF BIOLOGICAL PRODUCTS 成都生物制品研究所 Project Manager, responsible for engineering design, construction, commissioning and validation execution for clean utilities system which complies to WHO GMP 项目经理,负责疫苗工厂按照WHO 的GMP 进行水系统设计,安装和验证执行。 NCPC NORTHBEST 华北制药集团北元有限公司 Project Manager, responsible for complete a retrofitted Clean Piping system and validation execution which complies to FDA. Water for Injection distribution system, Purified Water distribution system, Pure Steam piping system and Clean Compressed air piping system are included. 项目经理,制药厂按照美国FDA 的GMP 标准进行水系统的改造和验证执行。水系统包括注射用水分配系统,纯化水分配系统,纯蒸汽管道系统,洁净压缩空气管道系统等。 B.BRAUN MEDICAL (SUZHOU) CO., LTD. 贝朗医疗(苏州)有限公司 Project Manager, responsible for complete a retrofitted material preparation system and validation execution which complies to EMEA. 项目经理,制药厂按照欧盟的GMP 标准进行对大输液配料系统进行改造和验证执行。 SHANGHAI WISON BIOENGINEERING CO., LTD. 上海惠生生物工程有限公司 Project Manager, responsible for complete a new clean piping system for a new vaccine plant and an antibody laboratory. Including WFI and PW distribution system. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
项目经理,负责完成一个新的疫苗工厂和抗体实验室的洁净管道系统。包括注射用水系统和纯化水分配系统。
HANGZHOU TIANYUAN BIOPHARMA
杭州天元生物药业
Project Manager, responsible for engineering design, construction, commissioning and validation execution for clean utilities system which complies to WHO GMP
项目经理,负责疫苗工厂按照WHO 的GMP 进行水系统设计,安装和验证执行。
POLIFARMA (NANJING) CO., LTD.
南京宝利化制药有限公司
Project Manager, responsible for complete a retrofitted Clean Piping system and validation execution which complies to TGA. Water for Injection distribution system, Purified Water distribution system are included.
项目经理,制药厂按照澳大利亚TGA 的GMP 标准进行水系统的改造和验证执行。水系统包括注射用水分配系统,纯化水分配系统等。
GE HEALTHCARE (SHANGHAI) CO., LTD.
通用电气(上海)药业有限公司
Project Manager, responsible for complete a retrofitted WFI piping and PS piping system which complies to FDA. This company has been certificated by FDA
项目经理,制药厂按照美国FDA 的标准进行注射用水系统和纯蒸汽系统的改造。该厂的无菌大容量注射剂已通过美国FDA 的认证。
Project Manager, responsible for complete the clean piping system for their extension project which complies to FDA. WFI distribution system, PW distribution system, PS system and CA system.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第13页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
项目经理,扩建项目按照美国FDA 的标准进行洁净管道系统的建设。包括注射用水分配系统,纯化水分配系统,纯蒸汽系统以及压缩空气系统。
SUZHOU DOWNRAYS
苏州东瑞制药有限公司
Project Manager, responsible for process design, construction and commissioning for a clean piping for sterile API production.
项目经理, 负责无菌原料药生产,洁净管道的工艺设计,安装和调试。
BAXTER HEALTHCARE (SHANGHAI) CO., LTD.
百特医疗用品(上海)有限公司
Project Manager, responsible for complete a retrofitted material preparation system and validation execution which complies to EMEA.
项目经理,制药厂按照欧盟的GMP 标准进行对大输液配料系统进行改造和验证执行。
QILU ANTIBIOTICOS PHARMACEUTICAL CO., LTD.
齐鲁安替比奥制药有限公司
1996 – 2005
Project Engineer. Mr. Cheng, who has gotten rich engineering experience in this ten years, through organize and participate all the pharmaceutical project constructed by QILU Group. Such as sterile API project, sterile small volume injection dosage, lyo-product project and so on.
And the sterile API project of Qilu Antibioticos has passed the qualification of EU GMP. Mr. Cheng organized and participate the whole duration.
项目工程师,十年内,通过组织并参与了所有齐鲁集团建设的制药项目,有无菌原料药,无菌小容量液体制剂,冻干产品等
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第14页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
一系列项目,他获得了丰富的工程经验。其中齐鲁安替比奥制药有限公司的无菌原料药通过了欧盟GMP 的检查。他参与了全过程。
1.6. 质量控制组织图 Quality control
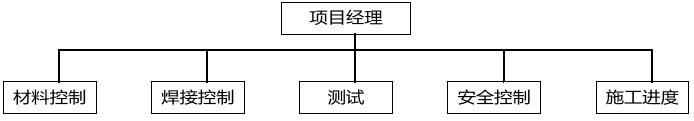
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第15页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||
2. 施工设备材料投入计划Tools list
齐全的施工设备准备是一个项目能够顺利执行的前提条件,朗脉公司已经准备了设备随时按需入场,按照项目施工要求,公司拟定安排以下主要施工机械设备投入计划,清单参照如下(最终设备材料计划以项目经理实际安排为最终确认实施清单):
Our construction equipment is ready to undertake any task. And acoording to the requirement,we draw a draft plan to launch into project. The list as the following: (the finally plan is refer to project manager’s arrangement)

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第16页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第17页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第5 部分施工组织 Construction Scheme | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第18页/ 共18 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
第 6 部分
Part 6
施工工艺
Construction Technics
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
目 录Index
| 1 主要施工工艺Construction Technics | 2 |
| 1.1. 施工准备Construction preparations | 2 |
| 1.2. 施工步骤Construction Flow | 4 |
| 1.3. 配管质量检查流程 Pipline Quality Testing flow chart | 6 |
| 1.4. 安全施工管理规范Safety and Good Engineering Plan | 6 |
| 1.4.1. 安全目标safety target | 6 |
| 1.4.2. 安全管理机构management organization chart | 6 |
| 1.4.3. 安全管理规范health and safety guidance | 7 |
| 1.4.4. 安全管理措施safety management measures | 11 |
| 1.5. 施工质量管理Construction quality management | 15 |
| 1.5.1. 质量保证体系quality assurance system | 15 |
| 1.5.2. 质量保证措施quality assurance measures | 17 |
| 1.6. 施工进度管理schedule manage | 26 |
| 1.6.1. 进度管理及程序progress management and procedure | 26 |
| 1.6.2. 全面资源保障Strategy for Deploying All Resources | 26 |
| 1.6.3. 应急预案emergency preparation | 27 |
| 1.6.4. 排除施工干扰之政策Strategy to Expel Interference | 29 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
1 主要施工工艺Construction Technics
1.1. 施工准备Construction preparations
施工准备按以下几个过程:Construction preparations refer to the following procedure:
 | 直观检查Visual inspection |
 | 抽样检查,确认无损伤并符合要求,检查是否有塑料帽密封10% random checks, confirmed no damage and meet requirements, check whether there are any sealed plastic caps 10% |
 | 检查型号,数量及证书Check model No., quantities & Certificates |
 | 核对定货单Verification with materials order form |
 | 加工现场的准备Preparations for field fabrication |
 | 验收及接收由负责人完成确认Acceptance and receipt of completion to be recognize by the person in charge |
 | 材料储存在指定区域内并根据型号,尺寸,洁净级别分类保存Materials stored in the designated areas under the model, size and sanitary class for preservation & identification |
 | 材料运输不可弯曲,撞击,挤压等When transport materials do not bending, impact and have other extrusion. |
 | 材料取用需有详细记录Material shall be released from storage with detailed records |
 | 现场测量,相关设备的检查Field survey and inspection of Related Equipment |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
 | 样本制作Samples production |
 | 材料预制:管道切割,面处理,弯曲及预制焊接。Prefabricated materials: Pipeline Cutting, surface treatment. Bending and prefabricated welding |

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
Field welding include 现场焊接包括:
 | 管道定位,现场焊接,质量检查。为保证每一焊缝内表面无氧化变色。必须使用高纯度保护气体,保证冲气时间不小于30 分钟。Pipeline positioning; field welding; quality inspection. To ensure that no surface oxidation color on inner weld surfaces; protection must use high-purity gases, air time is guaranteed – not less than 30 minutes. |
 | 非焊接连接:配件、设备连接Non-Welding connection: accessories, equipment connection |
 | 施工后处理:管道检查。气密性测试。Procedure after Construction: pipeline inspection, gas tightness test. |
 | 文件移交:样本记录,施工记录及测试报告Documents submission: samples records, test reports and construction records |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第5页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
1.3. 配管质量检查流程 Pipline Quality Testing flow chart

1.4. 安全施工管理规范Safety and Good Engineering Plan
1.4.1. 安全目标safety target 本项目的安全目标是“零事故”。 The safety target for this project is Zero Accident.
1.4.2. 安全管理机构management organization chart
 项目经理Project Manager:
项目经理Project Manager:上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
项目经理是健康安全环保工作的第一责任人,对健康安全环保工作负全面的责任。 Project manager is the first responsible person for safety works. He is fully responsible for the safety work. |
 | 安全员Safety Officer: 协助项目经理管理和执行健康安全环保计划,为健康安全环保工作的具体负责人。Assist the project manager to manage and execute safety plan. He is the specific person in charge for the safety work. |
1.4.3. 安全管理规范health and safety guidance
 | 施工现场应有专职安全员,每天应定期、不定期巡查,发现安全隐患可责令相关人员停工整顿。In the constructing site, there should have full time safety inspectors who go on a tour of inspection periodically or aperiodically. If any hidden problem is found, order relative personnel shutdown and make amendment. |
 | 进入施工现场应戴好安全帽,系紧帽扣,穿好工作服,工作鞋,并佩带工作牌。When entering the constructing site, wear safety helmet, tie the helmet buckle tightly, wear working clothes and shoes and carry working tablet. |
 | 严禁穿拖鞋、高跟鞋、泡沫底鞋在工地行走、作业;特别是高空作业(高空作业是指操作点高度在离地面或楼面 1.5m 以上的施工作业)。Strictly forbidden walking and working on the constructing site with slipper and high-heel shoe on, especially high altitude working (high altitude working indicates working at a position more than 1.5mhigh from the ground or floor). |
 | 在现场作业人员严禁与其它施工队发生纠打等现象。Forbidden in site working personnel fighting with other construction team. |
 | 施工现场人员在工作期间严禁赌博、喝酒。No gambling and drinking is allowed during working time for the in site construction |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
personnel.
 | 施工机械(设备)进场前,应检查设备的完好状态;重要的工具在每次使用前,应检查其是否完好。Check the construction mechanism before entering the construction place. Important tools should be checked each time before usage. |
 | 施工现场应规范用电,严禁私拉乱接,所有接线应由电工进行;电箱内应有漏电保护开关,保险丝。每一个用电回路均应由单一的断路器(或刀闸) 控制。Regulate electricity usage in the construction site. No private electricity connection is allowed. All the wire connection should be carried out by electrician. The electricity box should be equipped with leakage protection switch and fuse. Each electric loop should be controlled by signal switch (or brake). |
 | 所有电气工具操作人员应穿绝缘鞋或带绝缘手套。All electric facility operators should wear insulating gloves or shoes. |
 | 电焊机、手工氩弧焊机、自动轨迹焊机均应有可靠接地,接线端子有防护罩,在雷雨天气应暂停焊接作业。 Electric weld machine, manual argon arc weld machine and automatic orbit weld machine should all reliably connected to the earth. The connection should have protective cover. Stop welding in thunder storm situation. |
 | 高速旋转的电动工具(如型材切割机、砂轮机、台钻)在操作时严禁带纱质手套。工件的夹持应牢固,不得松动,防止随转动轴旋转或飞出。Wearing gauze gloves when operating the electric equipments with high revolving (such as cutting machine, grinding wheel machine) is forbidden. Hold the tools tightly to prevent their revolving or flying out along the revolving of axis. |
 | 各种气品在存放使用时,要距离明火 10 米以上,并且避免在阳光下爆晒,钢瓶在搬动过程中应轻拿轻放,严禁在地面上滚动或拖拉,严禁抛掷。All kinds of gases shall be over 10 meters away from open flame and avoid being insolated in the sun when they are stored or used. Slightly lift and put sown the steel tank. No rolling, |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
dragging and throwing is allowed.
 | 登高作业时(高度超过 1. 5 米),应有专人配合,配合人员负责工具,材料的接送,并负责看护;作业人员应佩带安全带,安全带的搭扣应系在操作点的上方的牢固构件上。 Climbing work should be assisted by special person who is in charge of delivering materials and tools and watching. Working person should wear safety belt with its agraffe tied to substantial component on top of the operating point. |
 | 撑梯的张角应合适(一般为 60-70°为宜),梯子在摆放前应先清理地面,梯子严禁接长使用。Folding ladder should have a suitable open angle (60-70°preferred). Make up the ground before placing the ladder. Prolonging the ladder for usage is forbidden. |
 | 严禁病时酒后作业,严禁有恐高症、高血压等疾病的人员进行高空作业。Do not work when in sickness or after drinking alcohol. High altitude working is forbidden if operated by people with acrophobia, high blood pressure, etc. |
 | 在施工现场动火,应得到业主、工地安全员的书面批准,并在火源处摆放手提式灭火器。Making fire in the construction site should be approved by written permission given by the owner or safety personnel in construction place and a portable fire extinguisher should be put on the fire place. |
 | 动火前应先清理操作点附近的易燃易爆品,并由专业人员进行。Remove the flammable and explosive material around the fire place. This should be operated by professionals. |
 | 所有操作人员应按规定在进入施工现场施工时,签定安全施工责任书,实行安全奖惩制度。 All the operation personnel should sign safe construction duty sheet before entering construction site. Put the safety rewards and punishments regulation into use. |
 | 在彩钢板上施工时。应先检查彩钢板是否已安装完成,(特别是彩钢板加强吊筋是否已安装。人员在行走时应尽量在隔墙、隔断、吊筋处或附近, |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
重物应摆放在隔墙、隔断处。Before constructing on the colored steel sheet, first inspect if it has been completely installed (especially whether the reinforced hanger has been installed). People should walk on or near the partition, obstruct and hanger. Heavy things should be put at the place of partition and obstruct.
 | 严禁在彩钢板开口处(如风口、灯孔等)行走、作业。 Walking and working at the colored steel sheet opening (e.g. air pot, lamp hole) are forbidden. |
 | 在容器和狭小管道内焊接时,应加强通风,严格执行双人监护制,监护人必须坚守岗位,发现问题要及时采取措施。When welding in containers or small-spaced pipes, ventilation shall be reinforced. Strictly execute the double-guardian system. The guardians shall stick to their post and take necessary measures once finding problems. |
 | 受压容器、密封容器、各种油桶、管道沾有可燃气体和溶液的工作进行时,必须先进行检查,并经过冲洗除掉有害、易燃、易爆物质并解除容器管道压力,清除容器 (包括密封空心、工件、管子 )的密封状态 (敞开口旋开盖)再进行工作,禁止在已做油漆或喷漆塑料的容器内焊接。When performing operations involving combustible gas and solution on the pressurized tanks, sealed tanks, various oil tanks and pipes, you should check them first, remove the harmful, combustive, and explosive material by flushing them, release the pressure of tanks and pipes, and remove the seals of the tanks (including the sealed cavity, work pieces, and pipes) before any work is done. To weld in the painted tanks is forbidden. |
 | 在做酸洗时,先试压,保压后用碱液试纸擦抹每一个焊缝,观察有无泄漏,严格执行双人坚督制、签字确认制,应穿带好防酸手套及防毒面具防酸胶鞋等防护用品。有关硝酸、盐酸、硫酸的化学用品使用中应注意:配比稀释时应逐渐地缓慢地小剂量的混合,切忌大剂量强制溶合,防止爆溅伤人。When performing pickling, have a pressure test first. Then |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
keep the pressure and erase each weld seam with alkali test paper to check if there is any leakage. Strictly execute the double-guardian system and signature-conforming system. Protective articles should be worn, such as acidproof gloves, gas mask, and acidproof rubber shoes. When using chemicals such as nitric acid, hydrochloric acid, and sulfuric acid, take care: When carrying out dilution practices, gradually and slowly mix with small volume. Do not mix with large volume, or it will cause explosion, spattering and damage.
 | 废料、废水应妥善处理、不应污染工作环境Waste material and waste water should be treated properly without polluting the working environment. |
 | 进入甲方施工现场,应遵守甲方的一切安全规章制度。Obey party A‘s all safety regulations while entering party A’s construction site. |
 | 未尽事宜,应按国家有关规定的标准执行。Unmentioned matters are carried out in accordance with relative national criteria. |

1.4.4. 安全管理措施safety management measures
 | 健康安全管理措施health and safety management measures 总的原则是执行业主的健康安全计划,执行我公司安全管理的制度及政府的有关法规。The general principle is to execute the owner’s safety plan, our company’s safety management system and relative official regulations. |
 | 安全教育safety education |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
安全教育的目的是提高职工的安全意识和自我保护的能力,避免各类事故的发生。 The safety education aims to improve employee’s safety awareness and self-protection ability and avoid accidents.
 | 安全教育safety education |
 | 进场安全教育*(*表示教育后需进行考试) Safety education on field entrance * (*indicates tests will be held after education) |  |
 | 方案技术交底教育 Technical notification on design proposal |
 | 星期一安全例会的教育 Regular safety meeting on Mondays |
 | 班前安全教育 Safety education before work |
 | 每一次教育都必须记录 ,记录教育的时间、地点、参加人员名单、教育的内容,进行考试的还应保存好考试成绩及试卷。 Each time the education should have a record to note the education time, location, person list and contents. Keep the test scores and test papers if test is taken. |
 | 安全作业单元与安全许可证safety operation unit and safety license |
 | 安全作业单 ,施工任务的布置通过安全作业单的方式下达 , 以保证安全工作与施工过程 ”五同时 ”(即计划、布置、检查、总结、评比五同时)Safety operation sheet. The construction tasks are assigned by safety operation sheets, which ensure the “five simultaneous” (simultaneous plan, assign, inspect, summarize, compare) of safety work and construction procedures |
 | 安全许可证,以下特殊作业必须有安全许可证:Safety license. The following special operations require safety licenses: |
■ | 进入设备作业许可证Equipment entry operation license |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
 | ■ | 电工作业许可证 Electrician operation license |
| ■ | 有毒有害作业许可证 Poisonous and deleterious operation license |
 | 安全检查safety inspection |
 | 安全检查是做好健康安装工作的重要手段之一,能及时发现事故隐患,及时制止“三违”(违章作业、违章指挥、违反劳动纪律)的行为发生,保证各项安全管理规章制度落到实处,从而杜绝事故的发生。Safety inspection is one of the key measures in safe installation. It can timely find the potential accident, prevent the “three breaches” activities (regulation-breaching operation, regulation-breaching direction, breach labor laws), and ensure all the safety regulations can be executed, so as to prevent the possibility of accidents. |
 | 安全检查的内容是:查领导、查制度、查机械设备、查安全设施、查教育培训、查操作行为、查劳保用品使用、查事故处理等。Contents of safety inspection: inspect the leaders, inspect the system, inspect the equipment, inspect the safety devices, inspect the education and training, inspect the operation, inspection the labor protection appliance, inspect the accident reasons, etc. |
 | 季节性安全检查。冬季应以防雪、解冻、跑漏、防临时建筑物倒塌为重点,秋季以防火防爆为重点。Seasonal safety inspection. In winters focus on snowproof, defreezing, and preventing the building collapse. In autumns focus on fireprotection and blameproof. |
 | 所有检查均必须按安全检查表进行,并做好记录。 All the inspections should be taken as per safety inspection sheet with relative records done. |
 | 基层检查均必须按安全检查表进行,并做好记录。Grass roots inspection should accord with safety inspection sheet with |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第13页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
relative records done. |
 | 代表性的安全检查种类 Typical Safety Inspection Category |
 | 领导检查中发现的隐患应下达书面的《事故隐患整改通知单》限期整改。If any hidden trouble is found during leader inspection, send out a Modification Notification on Hidden Trouble and ask them to modify it within a certain time. |
 | 隐患整改hidden trouble modification |
 | 接《事故隐患整改通知单》后,责任人应立即组织整改,做到“三定”(定具体整改人员、定整改措施、定整改完成时间); After receiving the Modification Notification on Hidden Trouble, the person in charge should arrange modification immediately, accomplishing “three establishes” (establish the persons involved in modification, establish the modification measures, and establish the finish time) |
 | 隐患整改完毕及时将整改结果书面反馈给通知单发出人。After finishing modifying the hidden troubles, timely feedback the modification results in paper to the person who sends out the modification notification. |
 | 通知发出人收到反馈情况后,要复验整改结果。The notification sender should re-inspect the modification results after having received the feedback. |
 | 应急计划 emergency |
 | 目的正确、及时处理意外情况,防止事故扩大,将事故损失控制在最小范围内。 The plan aims to correctly and timely treat the accidental conditions, prevent the accident from extending, and control the damages to a smallest limit. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第14页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
1.5. 施工质量管理Construction quality management
1.5.1. 质量保证体系quality assurance system
1.5.1.1. 公司质量方针guidance on quality
质量是永恒的追求,提供满足业主要求的工程和服务。
Quality is the goals to meet the requirement of owner on project and service.
1.5.1.2. 本项目质量目标target quality for this project
目标:保证我公司所提供的工程和服务,一次性通过相关认证。
Target: Ensure our engineering and services can pass relative certification only one time
1.5.1.3. 基本质量要求basic quality requirements
 | 恰当的控制各项工程步骤;连续的保持各项质量的有效性。Control all the construction procedures correctly. Maintain all the quality indexes effective continuously. |
 | 要具有及时预防和及时纠偏的能力。Have the ability to timely prevent and correct the errors. |
 | 具有可追溯性,能正确方便的追溯到各项质量、生产状况、检验情况,能追溯到事和人,便于分析改进不断完善体系和提高工程质量。Traceability. We can trace back to quality, manufacturing, and checking status as well as matters and persons for the convenience of analyse and improving system and project quality. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第15页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
 | 确保整个工程能顺利通过国家 GMP 验证,并达到美国 FDA 中要求的相应水平。 Ensure the whole project can pass the national GMP validation and FDA standards. |
1.5.1.4. 现场质量保证体系construction field quality assurance system
 | 项目质量实行公司经理负责制,对现场项目质量负有全面监督和保证职责。The project quality is ensured by manager responsibility system, which will monitor the on-site project quality. |
 | 现场项目经理对现场质量负有组织、全面管理的职责。 The field project manager is responsible to organize and manage the field construction quality. |
 | 建立以自检、互检和专职检查相结合的检验体系,把检验工作贯穿于预制、安装的全过程。对各个质量控制点,一定要按有关规范要求进行严格的检查、验收,验收不合格的工序坚决不允许转入下道工序。Establish a checking system combining self-check, cross check, and special check. The checking operation should be implemented through the prefabrication and installation. The quality control points should be checked and accepted by all relative regulations. Do not turn to the next procedure of the process if the previous procedure is unqualified. |
 | 质量员负责现场项目施工中的质量的检查监督、检验和试验、质量记录和不合格品的控制,以及推动、监督现场质量保证体系的正常运行。Quality officer is in charge of the quality supervision, inspection, experiment, quality record, and unqualified product control during field project construction, so as to promote and monitor the normal operation of on-site quality assurance system. |
 | 材料员负责自行采购和业主提供设备、材料的质量控制。The material officer is in charge of on-site purchasing and the |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第16页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
quality control of the equipment and materials provided by the owner |
 | 现场作业层的各专业、施工单位,统一在现场质量保证机构领导和控制下进行质量活动。All the construction companies will carry out activities involving quality under the united supervision and control of on-site quality assurance organization. |
 | 恰当的控制各项工程步骤;连续的保持各项质量的有效性。Correctly control all the engineering steps. Maintain all the quality index continuously effective. |
 | 具有及时预防和及时纠偏的能力。This system has the capacity to timely prevent and correct errors. |
 | 具有可追溯性,能正确方便的追溯到各项质量、生产状况、检验情况,能追溯到事和人,便于分析改进不断完善体系和提高工程质量。This system is traceable. We can trace back to quality, manufacturing, and checking status as well as matters and persons for the convenience of analyse and improving system and project quality. |
 | 确保整个工程能顺利通过国家 GMP 验证,并达到美国 FDA 中要求的相应水平。Ensure the whole project can pass the national GMP validation and FDA standards |
1.5.2. 质量保证措施quality assurance measures
严格按公司质量体系要求进行,特别注意人员培训,资质审核,材料检验,焊接工艺评定,工机具、量器具检验,各质量控制点等方面严格把关。
Strictly operate according to the quality system. Pay special attentions to personnel training, qualification verification, material checking, welding verification, tools and instruments checking. Check all the quality control points.
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第17页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
1.5.2.1. 图纸资料管理drawings management
对下发到各部门的图纸、方案、技术规范联络笺、材料代用单等必须指定专人统一管理,并登记收发,各技术员应做好施工原始资料,交工资料的整理工作,并协助施工班组填写各种施工记录表、卡,施工技术员每天必须记录施工日记。
All the drawings, proposals, technical standards, contact list, material sheet should be unitedly managed by a special person and have a registration record. The technicians should compile and pack all the original construction data and project handing over data, and assist the construction group to fill in all kinds of construction records and cards. The technicians should write the construction log daily.
1.5.2.2. 材料管理material management
 | 材料方面的资料由材料员管理,如产品合格证、材质说明书、收发料单等。根据专业技术负责人审核过单线图,材料表和预算并填写限额领料单到仓��领料时,对材质、规格、数量进行核实和验收。并且有合格证件(或复印件)确认无误,并在计算机上进行消料处理。收货人签字。领回后入库时应规格、型号、材质、分类挂牌存放并计入帐卡,按上述程序材料员向班组发料,每天将领、发料单装订成册备查,领回后不入库直接运到现场的材料,也应计入帐卡,交接清楚,按指定地方堆放整齐。The data concerning material, such as product certificates, material specification, material accepting and delivery sheet, are administered by material officer. Check the material, specification, and quality when receive material in the warehouse after fill in a material receiving sheet according to the drawings, material list, and budget. Check whether the certificates (or their copies) are valid and input it into the computer. The person who receives |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第18页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
the material should sign on the receiving sheet. After accepting the material, put it into the inventory. Separate and store them according to the specifications, models, materials and category. Distribute the material to the construction teams according to the above procedures. Compile a material receiving and distribution booklet each day. If a certain material is moved to the construction field instead of being put into the warehouse after being received, it should also be registered with clear notes to identify its location. |
 | 对所有材料特别是不锈钢,在切割后必须进行材质标识移置。The material marks should be moved after welding for all the materials, especially stainless steel. |
1.5.2.3. 焊接管理welding management
 | 项目部设置焊接专业工程师、焊接组长及系统验证专职人员,具体负责焊接工作,包括在单线图上标注焊口号、焊工代号、无损检验部位,填写无损检验委托单,联系与配合试验室无损检验。焊缝预热和热处理记录,透视报告的保管,焊接及热处理施工记录,管理焊条的烘烤、存放和保管记录等验证文件都由专职人员进行处理。Project department possesses the welding technician, welding team leader, and system validation personnel. They are in charge of welding, including note welding port number, welder code, non-destruction inspection parts on the single line diagram, fill in the non-destruction inspection sheet, contact and coordination with the lab non-destruction inspection, welding seam preheating and heat treatment record, perspective report maintenance, welding and heat treatment record, welding rot roasting, and record storage and maintenance. These works will be done by special personnel. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第19页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
 | 每个焊工均有一个固定的代号。公司焊接试验室应把经考试合格的焊工姓名、代号及合格项目表及时发给焊工所在的施工队及检验部门备查。焊后在其所焊的焊缝旁用记号笔写上代号,并要标注在单线图上。 Each welder has his own stable code. Our company will hand over the welder’s name, code, and qualified project sheet to the construction team and inspection department for reference in time. The welder will mark his code close to the weld seam and note on the single line diagram after welding. |
 | 焊好的管线经自检、互检合格后按规定的 X 射线透视比例由施工队委托试验进行透视,X光底片以区域为单位,以管线号为依据,建立流水号。同一焊口在透视委托单、底片、透视记录、检验报告以及在单线图上的标注必须一致,使抽查任何一张底片就可知道焊接位置。透视一定要及时,未经透视或抽透不足及需要重新返修的管线一般不要去安装,以减少高空作业,对透视不合格的焊口应立即在管线上作出明显标志,并及时组织返修,直至合格为止。返修次数不锈钢管不超过两次,并作好返修记录。 Self-check and cross check the pipelines after welding. If it is qualified, the construction team shall carry out perspective operation according to the specified X-ray perspective ratio. The X-ray negative film is divided by units of area. Set up a numerical order according to the pipeline number. The marks should be unified for the same weld port on perspective sheet, negative file, perspective record, inspection record, and single line diagram for the convenience of knowing the welding position just by checking only one negative film. The perspective should be taken in time. The pipeline without perspective or with less insufficient perspective or repair needed should not be installed so as to reduce aloft work. Make obvious marks on the pipelines that have welding ports with unqualified perspective. Arrange repairs timely, until it is qualified. The |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第20页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
repair times should not be more than twice for stainless steel pipes. Do not forget to make repair records. |
 | 对所有预制完并经检验合格的管段,暂不安装的,管口应予以封闭。To all the pipes that have being prefabricated and qualified, but will not be installed immediately, seal the pipe ends. |
 | 对不锈钢管道,在预制和安装时不得与其它管道接触。 To the stainless steel pipes, do not contact them with other pipes when under prefabracation and installation. |
1.5.2.4. 检验管理checking management
 | 建立以自检、互检和专职检查相结合的检验体系,把检验工作贯穿于预制、安装的全过程。对各个质量控制点,一定要按有关规范要求进行严格的检查、验收,验收不合格的工序坚决不允许转入下道工序。Establish a checking system combining self-check, cross check, and special check. The checking operation should be implemented through the prefabrication and installation. The quality control points should be checked and accepted by all relative regulations. Do not turn to the next procedure of the process if the previous procedure is unqualified. |
1.5.2.5. 质量控制
 | 项目质量控制重点参照业主工艺的要求,结合本公司在类似工程的 施工经历,初步明确下列质量控制重点: according to the owner’s requirements and our similar construction experiences in the past, we’ve established the following key quality control points: |
 | 设备、材料的选择与采购The selection and purchasing of equipment and material |
 | 焊接工程 Welding |
 | 电气、仪表 Electricity and instruments |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第21页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
 | 清洁清扫Cleaning |
 | 资料管理 Documentation management |
 | 质量控制的原则the principles for quality control |
 | 预防为主、检验把关; Focus on prevention and inspection |
 | 以 FD A、SFDA 的标准作为项目施工和验收的依据;Set FDA and SFDA criteria as the standards for project construction and acceptance |
 | 设备、材料的选择和采购必须经过技术部检验合格后方可使用;The equipment and material can be used only after being verified by the technique department |
 | 工序质量否决制:即上一道工序不合格,坚决不允许进入下一道工序;Process quality veto system: Namely no entrance to the next procedure if the previous one is unqualified. |
 | 施工记录、质量记录与施工同步,确保施工过程得到有效控制;Construction record and quality record should be simultaneous with the construction, ensuing the construction procedure can be effectively controlled. |
 | 所有施工机具、检验、检测、计量设备符合项目施工技术、施工质量要求;All the construction devices and checking, measuring, and accounting instruments accord with the technical and quality requirements |
 | 参加和参与施工的人员必须符合施工技术的要求,主要工种须持证上岗,所有人员须经技术、安全培训合格。People who are involved in the construction should meet the technical requirements for construction. Person at important job position shall have certificate. All the staff must pass technical and safety training. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第22页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
1.5.2.6. 针对性质量保证措施Pertinent quality assurance measures
 | 焊接质量保证措施Welding quality assurance measures 焊接质量是施工质量中关键的组成部分。本工程为制药项目,有洁净度要求,为保证管道焊接质量,我们采取以下措施:Welding quality is the key in construction quality. This is a pharmaceutical project, which have high requirement on sanitation. To ensure the pipe welding quality, we will take the following measures: |
 | 不锈钢管道采用全自动氩气保护焊接;The stainless steel pipes are auto welded with argon gas protection |
 | 设置专门的不锈钢材料堆放场所;Set up special stainless steel storage place |
 | 成立专门的不锈钢管道施工队;Establish a special stainless steel pipe engineering team |
 | 每个焊工均有一个固定的代号。公司焊接试验室应把经考试合格的焊工姓名、代号及合格项目表及时发给焊工所在的施工队及检验部门备查。焊后在其所焊的焊缝旁用记号笔写上代号,并要标注在单线图上;Each welder has his own code. Our company will hand over the welder’s name, code, and qualified project sheet to the construction team and inspection department for reference in time. The welder will mark his code close to the weld seam and note on the single line diagram after welding. |
 | 对所有预制完并经检验合格的管段,暂不安装的,管口应予以封闭。To all the pipes that have being prefabricated and qualified, but will not be installed immediately, seal the pipe ends. |
 | 对不锈钢管道,在预制和安装时不得与其它管道接触。To the stainless steel pipes, do not contact them with other pipes when under prefabracation and |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第23页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
installation. |
 | 试压时使用的水源必须为纯化水,必须有效控制游离氯和氯离子的含量. The source water for pressure test should be purified water, which can effectively control the free chlorine and chlorine ion. |
 | 保温工程质量保证措施 Quality assurance measures for heat preservation |
 | 管道保温须进行错缝连接;Breaking joints for pipe heat preservation |
 | 保温层的包扎应严密不漏,符合规范的要求。The bandage for heat insulating layer should be tight and accords with the specification 。 |
 | 清洁文明保证措施Assurance measures for cleaning |
 | 施工现场保持清洁文明,特别是有洁净要求的厂房,每个工种完成工作后,清理干净工作区域;Keep the construction field clean. Especially in the plant building that have special requirements on sanitation, clean the working area each time after finish operation. |
 | 进入洁净厂房施工前,工具、设备、材料、防护用品都必须进行清洁,经检查后方可进入洁净厂房施工;Before entering the sanitary plant for construction, the tools, equipment, materials, and protective articles should be cleaned and inspected. |
 | 洁净厂房管道施工内外壁清理干净,焊口内外清除干净,并及时封闭,室内所有缝隙、电仪管口、接线盒必须清扫干净。Clean the inner and outer pipe walls. Clean the weld ports and timely seal them. Clean all the seams, electric instrument pipe ports, and connection box |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第24页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
1.5.2.7. 资料管理
 | 对下发到各部门的图纸、方案、技术规范联络笺、材料代用单等必须指定专人统一管理,并登记收发,各技术员应做好施工原始资料,交工资料的整理工作,并协助施工班组填写各种施工记录表、卡,施工技术员每天必须记录施工日记。All the drawings, proposals, technical standards, contact list, material sheet should be unitedly managed by a special person and have a registration record. The technicians should compile and pack all the original construction data and project handing over data, and assist the construction group to fill in all kinds of construction records and cards. The technicians should write the construction log daily. |
 | 项目部设立专职验证员和资料员,专门负责相关文件的填写、交接、整理、归档等管理工作,使在施工过程中的每一道工序、每一个变更、每一次变更等都有相应的记录,具有可追溯性。施工过程中材料的质量证明文件,材料的自查、委托检查记录;施工方案的确定;施工人员的安排、施工机械的调配以及所有施工资源的分配全部有案可查。施工过程中与建设单位,其他施工单位的协调也相应作详尽的记录。最终目标使工程顺利通过建设单位、国家职能部门等检查,验证。The project department has verification officer and data officer to be specially in charge of document filling handing over, management and filing. Each procedure or each change should have relative record and is traceable.We have the history documents for material quality certificates, material self-check and cross check record, construction proposal determination, construction personnel arrangement, construction mechanics arrangement, and construction resources deployment. The detailed record of coordination with the constructed company and other construction |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第25页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
companies should also be available. |
 |  |
安装现场临时库房--分类存放
 |  |
1.6. 施工进度管理schedule manage
1.6.1. 进度管理及程序progress management and procedure
 | 向业主及工程总包递交的进度文件清单:The Progress list supplied to the owner: |
 | 进度周报、进度月报、进度计划(总体计划、季度计划、月度计划)、纠偏计划 Progress Weekly, Progress Monthly, Progress Plan (whole plan, quarterly plan, monthly plan), Correction Plan |
 | 进度管理职责表:Progress Management Responsibility Sheet |
 | 进度管理程序图Progress Management Process Chart |
1.6.2. 全面资源保障Strategy for Deploying All Resources
 | 充分调动公司内部的资源Sufficiently Deploy Resources in Our |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第26页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
Company |
 | 我们充分认识到此项目的重要性,公司总部将从各个方面对此项目进行支持政策的倾斜 We fully understand the importance of this project. Our headquarter will offer privileged support on this project |
 | 项目经理长驻现场。Project manager will stay at the construction field for a long time. |
 | 向此项目选择经验的项目经理,优秀的工程师,有丰富经验的工人和完好的施工设备。We will select experienced project manager, excellent engineers, skillful workers, and whole package of engineering equipment |
 | 我们已做好劳动力的充分保障。We have sufficient labor insurance |
 | 充分调动当地资源 Sufficiently Deploy Local Resources |
 | 充分与兄弟施工单位加强联系,利用当地在水、电、气、材料供应,人力资源,加工能力,运输能力等优势。Sufficiently contact with other construction companies and make advantages of local water, electricity, gas, material, labor, and machining ability |
1.6.3. 应急预案emergency preparation
 | 冬季施工工期保证措施Assuring Measures for Construction in Winter |
 | 因工程主要施工时间在主体厂房施工完成之后,且装修、装饰基本施工完成之后,所以在冬季施工的寒冷气候对工程施工的影响并不是特别大。Our construction will start after the main part of the plant building is finished and its decoration is done, therefore the cold weather won’t have big influences to our construction |
 | 尽量避免室外施工,应全面检查防冻保暖工作,充分做好防寒、防冻、防滑、防火和防止一氧化碳中毒的措施。Try to |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第27页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
avoid outdoor construction. Fully check the antifreeze and heat protection measures. Take measures for anti-cold, anti-freeze, skid proof, fire proof, and carbon monoxide poisoning proof. |
 | 尽量避免冬季登高作业。Try to avoid ascending work |
 | 遇暴风雪应停止室外工作,严禁在冰冻的湿地上施工。Stop working at snowstorm. Do not work on cold and moist field. |
 | 夏季施工工期保证措施Assuring Measures in Summer |
 | 首先调整作息时间,使施工的主要时间集中在早晚比较凉爽的时间进行。 Adjust the work and rest time at first to focus the engineering time on morning and evening, when it is cold. |
 | 要配备必要的防暑降温设施、器材、药品和茶水饮料。Prepare necessary heatstroke preventative devices, equipment, drugs, and waters. |
 | 高温作业场所,应加强通风和降温措施。Reinforce ventilation and cooling measures for |
 | 通风机和排风扇等设备应由防护网罩。The ventilator and exhaust fan should have protection shield |
 | 要加强食堂的清洁卫生工作,严格执行检查消毒制度,防止职工食物中毒。Strengthen the sanitary conditions in dining room. Strictly execute the disinfection system. Avoid food poisoning. |
 | 雨季施工工期保证措施Assuring Measures in Rain Season |
 | 暴雨前后,要检查工地临时设施,脚手架、机电设备、临时用电线路等,发现倾斜、变形、下沉、漏雨、漏电等现象应及时修理加固,有严重危险的应立即排除;After rainstorm, check the temporary equipment, scaffold, electric devices, temporary electric loop, etc. Repair it if any |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第28页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
incline, deformation, sinking, rain leakage, and electricity leakage is found. Remove it if it is severely dangerous. |
 | 雷雨时,严禁管道焊接工作。Pipe welding is strictly forbidden at thunder storm. |
1.6.4. 排除施工干扰之政策Strategy to Expel Interference
 | 当设备材料、设计图纸因特殊原因不能正点到位时,立即调整施工计划,制定补救工期的对策。一旦到位,立即施工,将失去的工期补回来。If the equipment, material, or design drawings can not arrive in place in time, adjust the construction plan immediately and set up countermeasure. Once they arrive, carry out construction immediately to offset the time limit. |
 | 供的材料合理安排采购计划,确保不影响施工进度计划,对于因施工计划改变可能需紧急采购的材料,应在施工前做工供应商的评估。 For the materials that are supposed to be purchased by ourselves, reasonably arrange the purchasing plan. Verify the supplier before construction if urgent purchasing is necessary because of construction plan changes. |
 | 与生产运行有交叉的,在施工开始前做好防护隔离措施,保证施工与生产的安全顺利进行。 Take protective measures before construction if production operation is involved, so as to ensure the successful operation of construction and manufacturing. |
 | 坚持逐月检查,警惕和预防传染病的袭击,发现病员,立即隔离。Carry out checking month by month. Look out and guard against the attack of infection. If any patients are found, separate them from others. |
 | 管线及其它设施的加固措施 Solidifying Measures for Pipes and Other Devices |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第29页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第6 部分施工工艺 Construction Technics | ||
 | 管道在安装过程中,严格执行工艺作业规程,一般现做支架固定,并手工点焊固定,并在短期内施焊完成。During installation procedures, strictly implement the process working specifications. Normally use a bracket for the sake of solidifying and weld it in short time. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第30页/ 共30 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation for Piping System | ||
第 7 部分
Part 7
管道系统施工规范
Construction Regulation for
Piping System
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
目 录
| 1. 管道系统施工规范Construction Regulation | 3 |
| 1.1. 焊接总则General | 3 |
| 1.2. 焊接规范Welding regulation | 4 |
| 1.2.1. 总体要求General requirements | 4 |
| 1.2.2. 材料Material | 5 |
| 1.2.3. 焊接准备Weld joint preparation and fit-up | 6 |
| 1.2.4. 预热温度Preheating temperature | 8 |
| 1.2.5. 供电Electrical characteristics | 8 |
| 1.2.6. 焊接程序Welding procedure | 8 |
| 1.2.7. 焊接修补Weld repair | 8 |
| 1.2.8. 自动焊接程序Automatic welding program | 9 |
| 1.2.9. 文档Documentation | 10 |
| 1.2.10. 非破坏性检查Non destructive examination | 11 |
| 1.2.11. 每日焊接样品Weld daily samples | 11 |
| 1.3. 装配和安装Fabrication and erection | 12 |
| 1.3.1. 范围Scope | 12 |
| 1.3.2. 管件材料接收储存Tubing materials receipt storage | 12 |
| 1.3.3. 分类Identification | 13 |
| 1.3.4. 装配Fabrication | 13 |
| 1.3.5. 布管,切割和装备Lay-out, cutting and fitting up | 13 |
| 1.3.6. 焊接程序和资格Welding procedure and qualifications | 14 |
| 1.3.7. 安装Erection | 15 |
| 1.3.8. 焊接接口Welded joints | 15 |
| 1.3.9. 法兰接口Flanged joints | 15 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
| 1.3.10. 快装接口Clamped joints | 16 |
| 1.3.11. 支撑和穿管Support of tubing and penetrations | 16 |
| 1.4. 检查Inspection | 17 |
| 1.4.1. 范围Scope | 17 |
| 1.4.2. 配件和焊接编号Component & weld identification | 17 |
| 1.4.3. 焊接准备Weld preparation | 18 |
| 1.4.4. 焊接检验Weld inspection | 20 |
| 1.4.5. 预装配件的保护Protection of prefabricated spools | 22 |
| 1.4.6. 验收Acceptance | 22 |
| 1.4.7. 其它要求Other requirements | 22 |
| 1.5. 水压测试与清洁water pressure test and cleaning | 23 |
| 1.5.1. 压力测试Pressure testing | 23 |
| 1.5.2. 测试报告Test reports | 25 |
| 1.5.3. 水压压力测试Hydrostatic test pressure | 25 |
| 1.5.4. 系统清洁和清洗System cleaning and flushing | 26 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
1. 管道系统施工规范Construction Regulation
1.1. 焊接总则General
 | 本规范适用于卫生管道的焊接、装配和安装。The specification applies for welding, fabrication and erection of sanitary piping. |
 | 本规范特别用于不锈钢316L 管件间之圆周对接焊头之连接In particular such specification is to be used for circumferential butt joints between sanitary piping components made of stainless steel type AISI 316 L. 。 |
 | 在项目中与本规范有冲突的其它规格必须与朗脉公司讨论,以便澄清和定义。Existing discrepancies between this specification and others applicable for the project, must be discussed with MACROPROCESS for clarification and definition |
 | 本规范中的焊接连接将需要符合FDA 的验收标准和3A 标准。Welded joints in the scope of this specification shall require the acceptance by FDA and shall comply with Sanitary Standard 3A. |
 | 本规范中术语装配者是指朗脉公司,他负责管道的装配和安装。In the scope of this specification the term FABRICATOR is referred to MACROPROCESS, who is encharged for fabrication and erection of piping. |
 | “业主”是指用户或其代表。the term OWNER is referred to CUSTOMER or his representative. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 焊接检查员”是指业主或用户代表,他将负责检查管道承建商的焊接操作。the term WELDING INSPECTOR is referred to the Owner or Customer representative who is en charged to inspect the welding operation and examination performed by piping construction Contractor. |
1.2. 焊接规范Welding regulation
本规范覆盖用自动轨迹焊接机或手动TIG 设备的气体钨电弧不锈钢焊接设备。
This specification covers the gas tungsten arc welding of austenitic stainless steel using automatic orbital tube welding machines or manual TIG equipments.
“管道”应包括所有需要焊接元件。
The term TUBING shall include all components of the welding.
1.2.1. 总体要求General requirements
 | 采用这些程序的手动TIG 焊接工,将用实际工作材料作焊接试样测 试以确认合格。这些焊接试样和自动焊接试样进行同样的独立检验。Manual Tungsten Inert Gas (TIG) welders using these procedure shall be qualified by making coupon test welds using actual job material. These coupons shall be subject to the same Independent examination as the automatic welds. |
 | 自动焊接机的操作人员将用样品材料做焊接测试以确认其资格。焊 接测试应该选用项目常用的管材尺寸材料。Automatic welding |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
machine operators shall be qualified by making acceptable test welds on sample materials. The welds shall be made on the prevalent job size tube material. |
 | 组件的定位点焊必须避免对完全焊接的不良影响。Tack welding of the components must avoid deleterious effect on the complete weld. |
 | 定位点焊应该尽可能的轻,以降低过热而导致材料的内部结构变化。Tack should be as light as possible to reduce over heat that might cause structural changes to the granular composition of the material. |
 | 定位点焊不应焊透内表面。Tacks should not penetrate the inner surfaces. |
 | 定位点焊完成后,需要检查线定位焊接的对准性和缝隙。After the tack welding completion it’s necessary to check the alignment and gap |
1.2.2. 材料Material
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第5页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 用于焊接的材料须符合甲方的技术规格。The material to be welded shall comply with the owner specification, . |
 | 所有管道和配件须完全焊透。All tubing and fittings shall have complete weld penetration. |
 | 采用的焊接电极为2%涂钍平头钨焊电极。Welding electrode to be used shall be a pointed 2% thoriated tungsten electrode with a flat tip. |
 | 焊接电极如有污染须更换。The electrode shall be replaced if contaminated.。 |
 | 焊接管道内、外部保护氩气的最低纯度为99.99%。Welding grade argon gas shall be use inside the pipe for back-up purge and shall be 99.99%minimum purity. |
 | 焊接过程中必须要通入氩气进行保护,以防止焊接的内表面被氧化。在开始焊接前,吹走焊接区域的所有氧气。Argon gas back-up has to be provided to prevent oxidative attack of the inner weld surface.Before starting the welding all oxygen is purged from the welding area. |
1.2.3. 焊接准备Weld joint preparation and fit-up
 | 当焊接表面潮湿时,不得进行任何焊接。除非有适当的保护措施,在严酷气候或风速高或不良大气条件下不得进行室外焊接。No weld is carried out when surfaces to be welded are wet. No outside welding is carried cut during inclement weather or during periods of high wind or negative atmospheric agent unless the work is properly protected. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第6页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 焊接接头两端面的清洁,圆整和平直是重要的。It is essential that the two surfaces of the joint are clean, circular and squared. |
 | 金属加工方法应确保焊接端表面光滑,无划痕或其它有害的不规则形状。The machining method of base metal preparation shall leave the weld end with smooth surfaces, free from notches or other harmful irregularities. |
 | 建议采用G+F 或同类设备来处理切割和最终处理。It is recommended to carry out the cut-off and final preparation using a G+F equipment or equivalent. |
 | 切割必须平直和不会造成管件变形或损坏。砂轮切割的管件端面必须清洁以除去磨屑。Cut-off must be squared and not cause any distortion or damage to the tubing. The pipe cut-off by an abrasive wheel must be cleaned to remove the abrasive dust. |
 | 碳钢切割工具在任何情况下都不得使用。Carbon steel cutting tools are not admitted in any circumstance, |
 | 用于焊接端面的处理、装配和安装工具,在使用前须检查是否是没有生锈的认可合金钢,清洁无油脂,和无其它污染残留物。Tools used for weld joint and preparation, fabrication and installation, shall be stainless of approved alloy steel, cleaned free of oil, grease and other contamination residuals before they are used. |
 | 不得使用硫基切割油,除非在焊接前管件的内外表面都被彻底清除所有残留油渍。Non sulphur based cutting oil may be used, provided all traces of such oil are completely removed from both the interior and exterior surfaces on the tubing prior to |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第7页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
welding. |
 | 需与焊接检查员一起设定焊接设备。The welding equipment shall be set up together with WELDING INSPECTOR. |
 | 此检查员将检查所有设置是否符合主程序。他将签署焊接日志和注明日期。The inspector will check that all settings comply with the master program. He shall sign and date the welding log to that effect. |
 | 每天的开始时,每个操作人员要作焊接测试。这些焊样将编号,并呈交给焊接检查员。每台自动焊接机都需做测试。At the start of each day, each operator shall make acceptable test welds on plain tubing, These pieces shall be identified and made available to the welding inspector. Test shall be made for each automatic welding machine. |
1.2.4. 预热温度Preheating temperature
 | 在焊接材料的温度不得低于10 ºC。一般来说,不需要预热。The minimum base material temperature shall not be less than 10 0 C prior to welding. In general, no preheating shall be required. |
1.2.5. 供电Electrical characteristics
 | 电流应与注明在相应的焊接表相符。Current shall be as specified on the applicable welding schedule. |
1.2.6. 焊接程序Welding procedure
 | 焊接工艺,如焊接次序,焊接程序设置,焊极位置等,都将与相应的焊接表相符。The welding technique, such as weld sequence, welding program settings, electrode positioning etc., shall be as shown in the applicab le welding schedule。 |
1.2.7. 焊接修补Weld repair
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第8页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 经检查,如发现不符合标准的缺陷并且此缺陷是可修复的,有必要进行修复焊接。最可能的修复是在已有的焊接上进行再焊接;A weld repair is required when, through inspection, it is determined that an unacceptable defect exists and that the defect is correctable . The first repair may be a re-weld over one existing. |
 | 可做修补的缺陷包括不佳的准备,不良线性,过量或不适当的焊接穿透,多孔性和沙眼;The allowable defects which may be repaired include poor fit up, misalignment, excessive or inadequate weld penetration, porosity and pin holes. |
 | 所有有裂纹的焊接将被视为不合格,并须与受热区一起去除。All welds with cracks shall be rejected and removed together with the heat affected zone. |
1.2.8. 自动焊接程序Automatic welding program
 | 所有主程序都规定了不同尺寸管件焊接的一组参数;Calibration of the automated tube welding system shall be in accordance with the procedures to be defined by the welding operator together with the inspector. |
 | 自动管道焊接系统的焊接程序都须符合焊接操作员与检查员一起制定的程序; Any master program defined a set of parameters for different size of tubing to be welded. |
 | 正式和测试焊接需采用已认可的焊接程序;Production and test welds shall be made only using approved welding programs. |
 | 焊接样品必须按顺序编号(如T1,T2 等)。焊接样品,具备完全的可追朔性,将备独立机构检验;Welding samples must be |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第9页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
numbered in sequence ( ex.: T1, T2, etc.). Welding samples, with complete traceability, are subject to independent laboratory examination. |
 | 在整个施工阶段,所有每天的现场样品将留予业主;All daily samples will be available in site to the owner, during all the construction period. |
 | 自动管道焊接系统的校正都须符合焊接操作员与检查员一起制定的程序。Welding programs of the automated tube welding system shall be in accordance with the procedures to be defined by the welding operator together with the inspector. |
1.2.9. 文档Documentation
 | 朗脉将提供此系统所有焊接记录报告。此焊接记录报告表格,必须完全填写,最少包含:MACROPROCESS will provide a written log on all welds carried out on the system. The weld log forms, that must be fully filled, will minimally contain : |
 | 系统名称system name |
 | 焊接号weld number |
 | 焊接工姓名welder name |
 | 设备编号machine identification |
 | 焊接日期welding date |
 | 焊接程序编号welding program number |
 | 焊接操作工(如有,还加上检查员)签名welding operator ( and in addition inspector, if any ) signature |
 | 设备将打印相应自动焊接的报告。在此焊接报告中要有焊接编号。In addition the machine will print out an automatic log that will refer to the weld carried. This weld number shall be then |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第10页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
identified on the welding log. |
 | 透视简图 (标示相应的坡度,尺寸和长度,所有焊点编号)将在整个施工期间使用并且更新,在工程完成生成竣工图。Each weld shall be numbered in progressive way. All welds will be shown on isometric sketches and on the related Welding Report ( FORMAT 5, already mentioned ). |
 | 每个焊点以累进方式编号。所有焊点标示在透视简图和焊接报告中。Isometrics sketches (showing installed elevations for proper sloping, dimensions and lengths, all weld numbers) will be available during site activity and will be updated ( “ as-built” issue ) at the end of job. |
1.2.10. 非破坏性检查Non destructive examination
 | 当相应的条例或合同规格要求时,非破坏性检查将在符合条例或合同规格下进行。须考虑到因此而造成的时间延误。Non destructive examination, when required by the applicable by the applicable code or contract specification, shall be performed in accordance with the code rules of contract specification. The time delay due to these controls must be considered. |
1.2.11. 每日焊接样品Weld daily samples
 | 每日焊接样品应采用设备焊接程序。这些样品必须按顺序编号(如P1,P2 等)。所有这些焊接样品将在整个施工期间提供给业主;Weld daily samples shall be made using the machine welding programs. These samples must be numbered in sequence ( ex.: P1, P2, etc.). All of those will be available to the owner in site during all the construction period. |
 | 每日样品结果必须收集在完全填写的自动焊接设备测试报告中。The daily samples results must be collected in the Automatic |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第11页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | Welding Machine Test Report that must be fully f illed。 |
1.3. 装配和安装Fabrication and erection
1.3.1. 范围Scope
 | 本节和引用的图纸标准以及条例将适合所有管道现场装配和安装。This section and drawings standard and codes referenced shall apply to the field fabrication and erection of all tubing. |
 | 本规格中的内容不得用于解释使供应商从完全符合引用条例的责任中解脱。Nothing in this specification shall be interpreted as in any way relieving SUPPLIER from the responsibility for complete compliance with the referenced codes wherever they apply. |
1.3.2. 管件材料接收储存Tubing materials receipt storage
管件材料接收储存和现场处理如下:Tubing materials receipt storage and handling at site:
 | 管件材料需由其他人确认和检验;Tubing materials shall be identified and inspected by others. |
 | 管材和预装配件的存放应注意不会被油脂,潮湿或其它物质污染;管材和预装配件应处理得当,以避免变形,刮伤或任何有形损伤;It shall be taken care to keep all tubing materials and prefabricated assemblies free from contamination by grease, moisture or other matter does not occur, Tubing materials and prefabricated assemblies must be handed in order to avoid any denting, scraping or any other physical damage. |
 | 所有不锈钢材料应与其它施工材料分开检查和储存。储存区必须在 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第12页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 建筑物内,并且有防护任何腐蚀性环境和温度变化的措施;All stainless steel materials should be checked and stocked separately from other construction materials. The storage area must be located inside a building and protected from any corrosive atmosphere and temperature changes. |
 | 通道应严格限制以确保材料不会受损伤和丢失。只有在施工需要时才能从储存区提取材料。Access shall be strictly limited in order to guarantee a protection against physical damage and losses. Material shall be released from storage only when needed for construction purposes. |
1.3.3. 分类Identification
 | 所有材料将按规格进行分类,并且适当储存和处理以便于辨认。All materials will be identified in accordance with the applicable specification and shall be stored and handled in order to allow their proper identification. |
1.3.4. 装配Fabrication
 | 装配应按照此规格的要求和如有的详细安装要求(图纸或其它)进行。Fabrication shall be in accordance with the requirements of this specification and with installation specific details ( drawings or others ), if any. |
1.3.5. 布管,切割和装备Lay-out, cutting and fitting up
 | 管件应按图纸上的尺寸精确切割以适应现场情况。Tubing shall be cut accurately to measurements shown on drawing to suit field conditions. |
 | 所有待焊接的管件端面需清理毛刺,并且切割面应平直,边角无切 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第13页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
割园弧。管件边缘的端面处理和清理毛刺应使用终端处理工具,并且其公差与配件一致以确保适当的焊接线性。All tube ends to be welded shall be deburred and have square cut edges, free of shear radii at the corner. Tube edge facing and deburring shall be done with an end prep tool and be of equal tolerance as the fittings to assure proper alignment for welding. |
 | 检查切割管件终端,以确保其平直。The end of the cutted pipe shall be checked to insure its squareness. |
 | 清除毛刺应避免破坏内外径,或损伤管件的内抛光。Burs shall be removed in such a manner as not to level I.D. or O.D. or damage polished interior of the tubing. |
 | 在管件端口积聚的磨屑必须擦拭干净。Abrasive dust accumulated near the end of the tube must be wiped clean. |
 | 所有安装在管线上的管件不应受过分弹力和受力下装配。All tubing shall be worked in place without excessive springing or forcing. |
 | 不允许通过管件加热和弯曲以达到线性整齐。Heating and bending tubing to achieve alignment is not allowed. |
1.3.6. 焊接程序和资格Welding procedure and qualifications
 | 所有焊工应具备6 个月现场工作经验经过工厂培训All welders shall be qualified either by 6 months field experience or by factory training.; |
 | 在开始焊接前,焊接程序应提交给焊接检查员核准Welding |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第14页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
procedures shall be submitted to welding inspector for approval before starting the welding activities. |
1.3.7. 安装Erection
 | 所有组装管件应在不受过分弹力和受力下装配。不允许有切割或弱化的支撑物作管道安装;All assembled tubing shall be installed without excessive springing of forcing . Cutting or other weakening of structural members to facilitate tubing installation will be not permitted. |
 | 所有管件安装应可以自由膨胀和收缩,而不至于损坏连接和支撑;All tubing shall be installed to permit free expansion and contraction without damage to joints or supports. |
 | 所有管道必须有适当的坡度(0.3% 最大0.5% )以确保完全排水。All lines must guarantee the complete drainage with proper slope (0.3% max. 0.5%). |
1.3.8. 焊接接口Welded joints
 | 所有焊接应按照本规格所述的要求进行准备和装配。All welded joints shall be prepared and made in accordance with described of this specification. |
1.3.9. 法兰接口Flanged joints
 | 所有法兰接口,应用全新的密封圈和螺栓。在拧紧螺栓前应确保所有密封圈定位准确。应注意防止将螺栓拧的过紧。所有被拧得过紧的螺栓螺帽都应更换;All flanged joints, if any, shall be made with gaskets and bolting materials brand new. It shall be taken care to assure that all gaskets are properly positioned prior to bolts tightening. It shall be taken care to avoid |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第15页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
overtorquing any bolts. All bolts and nuts which are overtorqued shall be replaced. |
 | 法兰螺栓孔应跨骑于管道中心线,除非图纸上另有说明。 Flange bolt holes shall straddle pipe center lines except when noted otherwise on drawings. |
1.3.10. 快装接口Clamped joints
 | 不得使用螺纹接口。Threated joints shall not be used. |
1.3.11. 支撑和穿管Support of tubing and penetrations
 | 管件的支撑应按管件支撑图纸上的参数和细节设计、装配和安装。Support to tubing shall be designed, fabricated and installed in accordance with the details and parameters of the pipe support drawings. |
 | 不锈钢管件的安装按制造商的建议进行。Stainless steel tubing is to be installed in accordance with manufacturers recommendations. |
 | 吊架和支撑的安装应尽量的经济,并符合支撑图纸或规格上的描述。Hangers and/or support should be installed as economically possible and be in accordance with the charts and notes on the pipe support drawings or specifications. |
 | 管道的支撑应符合管道图纸上要求的坡度。Tubing shall be supported in such a way to comply with the slope required on piping drawings. |
 | 所有装配的管道应最少符合检查,检验和测试要求规格。All |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第16页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
fabricated tubing shall as a minimum meet the examination, inspection and testing requirements specifications. |
1.4. 检查Inspection
1.4.1. 范围Scope
 | 本技术规格适用于不锈钢管件自动焊接检查,该焊接检查按照技术规格中的焊接要求;This specification covers the inspection of automatic welding stainless steel tube and fittings, in accordance with the welding specification. |
 | 焊接日志检查清单将用于查核主要信息,准备和质量要求;The weld log inspection check list shall be used to verify the principal information, preparation and quality requirements. |
 | 焊接检查员按照焊接要求,检视分包商的焊接记录、检查和设置、非破坏性测试,以及目检。在双方同意并理解相应的时间延误下,此工作须在任何时间进行;The welding inspector shall witness the required subcontractor records, checks and setups, non-destructive tests, and visually inspect the welds, as specified herein and in accordance with the welding specification. This activity must be carried out at any time with the agreement of both parties involved and knowing the related time delay. |
 | 焊接检查员或其指定代表应目检所有焊接。The welding inspector or his appointed representative shall visually examine all welds . |
1.4.2. 配件和焊接编号Component & weld identification
 | 每个焊接应在其立体图中编号;Each weld shall be numbered on |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第17页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
his isometrics. |
 | 检查员应保留焊接检查清单,而且实际的焊点以此编号辨别;The inspector shall maintain a weld inspection check list and the actual weld shall be identified primarily by this number. |
 | 每个焊点的记录应包括所有相关的条目。The record for each weld which shall include all items listed |

1.4.3. 焊接准备Weld preparation
焊接检查员决定是否进行下列检查/确认。 The following inspection/verification could be made at the welding inspector option.
 | 管道截面确认Tube section verification |
 | 管道的长度按要求在管道图纸中标示;The length of tube is as required to give dimensions as shown on the piping drawings. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第18页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 要去掉管道端口切割产生的任何可能之污物;Tube ends are removed any possible contamination from the cut off. |
 | 工具不得被碳钢所污染;No tool has been contaminated by contact with carbon steel. |
 | 确认管道圆度公差。Verify tube roundness to tolerance. |
 | 清洁确认Cleaning verification |
 | 用认可的溶剂清洁配件。Components are cleaned with approved solvent. |
 | 在焊接前,已清洁和准备好的端口不得接触皮肤。The cleaned and prepared ends should not come into contact with the skin prior to welding. |
 | 在部件的端口不得有可目见的异物,此包括清洁布的纤维。No visible foreign material is acceptable at the weld ends of the components. This includes lint from cleaning cloths. |
 | 钨焊极必须清洁和无污染。Tungsten electrode must be clean and uncontaminated. |
 | 接口准备Joint fit-up |
 | 焊接组件端口间隙用厚度塞尺测量,不得超过0.25 mm。The gap between ends of welding components shall not exceed 0.25 mm as checked by thickness gauge. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第19页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 钨焊极应置于焊点中央。The tungsten electrode shall be centered on the joint。 |
 | 焊机设置Weld machine settings |
 | 参数的设置应按焊接程序中相应的焊接表进行。Parameters shall be set in accordance with appropriate welding schedule as shown in the welding procedure. |
 | 确认气源就位。Verify that purge bladder is in place. |
1.4.4. 焊接检验Weld inspection
每个焊接样品和所有装配焊都要目检管道内侧。如焊接有可修复的缺陷,在焊接部件拆离焊机前进行第二次焊接以修复缺陷。如果重新焊接未修复缺陷,则此焊接视为不合格并不得装配。
Every sample weld and all production welds shall be visually inspected on the interior, If the weld has correctable defects, a second weld pass may be made to try to correct them before the spool is removed from the welding machine . If the rewelding does not remove the defect, the weld shall be rejected and not mounted.
所有自动焊接按如下控制:
All automatic welds will be controlled as follows:
 | 对每日样品,用选定的焊接程序完成一个焊接,进行X 射线检验For each daily sample a weld carried out with the same welding program will be chosen. The check will be done with Rx will be carried out. |
 | 对于所有完成的自动焊接,含有焊接参数的焊机打印报告For all executed automatic welds, a welding machine print out with |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第20页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
welding parameters will be annexed. |
 | 10%的自动焊接,进行X 射线检验10% of executed automatic welds, Rx will be carried out. |
所有手工焊接要进行X 射线检验。All the manual weldings will be Rx inspected.
如有表面缺陷,则本规格条目适用。For surface defects, the headlines of this specification are valid.
表1:Table 1
| A | B | ||
| 须进行重新焊接的缺陷 | 不合格的缺陷:须与受热区(棕色)一起切除: | ||
| Defects for which rewelding may be | Defects to be rejected : Cut out | ||
| attempted | required together with its heat affected | ||
| zone ( brown color ): | |||
1. 未焊透incomplete penetration 2. 多孔porosity 3. 沙眼pin hole 4. 冷隔cold lap 5. 过度穿透( <0.4 mm )excessive penetration 6. 未对准( <0.55 mm)misalignment | 1. 裂纹cracks 2. 过烧burn out 3. 解块sugaring 4. 冷隔cold lap 5. 过度穿透(>0.4 mm ) excessive penetration 6. 未对准(> 0.55 mm)misalignment |
点焊如有破裂、完全焊透、过烧、组件未对准,则视为不合格。Tack welds shall be rejected if : cracked, full penetrated, burned, components misalignment.
焊件移离焊机后,对焊接外观进行目检。The outside of the weld is visually inspected after the spool is removed from the welding machine.

上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第21页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
1.4.5. 预装配件的保护Protection of prefabricated spools
 | 所有开口应有适当的保护,保持密封;All openings shall be kept sealed at all times with proper protection. |
 | 快装接口和法兰端面应有保护措施以防止损坏;Clamp ends and flange face shall be protected from damage in handling by approved protective devices. |
 | 除了在工作台上,在处理和存放期间,预装配件应置于保护区内的木支架上。 During handling and storage other than on work tables, spools shall be on wooden skids of racks in a protected area. |
1.4.6. 验收Acceptance
 | 焊接检查员应完成焊接检查清单并须在完成的清单上共同签署和注明日期。如在任何地方有不合格焊接,则不合格原因应被记录。焊接检查员应在焊接完成时签署清单。The welding inspector shall complete the weld inspection check-list. When it has been completed. It shall be jointly signed and dated. If at any place the weld is rejected, the reason for rejection shall be recorded. The welding supervisor shall sign each list at the time the weld is made. |
1.4.7. 其它要求Other requirements
 | 检查员应确认吹刷气体和后备气体流量计处于准确工作状态。The inspector shall insure that the flow gauges on the purge gas and back up gas be in accurate working condition, where applicable. |
 | 如任何工具或组件被怀疑有污染,应按ASTM 标准程序进行清洁和 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第22页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
测试。If any tool or component is suspected of having contamination, it shall be cleaned and tested in accordance with ASTM standard procedures. |
1.5. 水压测试与清洁water pressure test and cleaning
本规程包含在管道系统完成后,为测试系统性能和特性而进行的所有工作。This specification covers the activities to be carried out after the system pipe construction, to test and improve his capability to perform the required characteristics.
1.5.1. 压力测试Pressure testing
 | 所有压力测试应当同检查员一起进行;All pressure tests shall be complete system tests carried out together with the inspector. |
 | 所有测试进行时应对操作人员的安全作好预防,要进行水压测试的系统应该带有合适的表和压力安全泻载装置;Every test shall be carried out with all the precaution needed to insure the safety of the operator, Systems to be hydraulically tested will be provided with appropriate gauges and pressure relieving devices. |
 | 所有连接点,包含焊缝应该去除绝热保温以便在测试过程中观察;All joints, including welds, are to be left un-insulated and exposed for examination during testing. |
 | 不需要进行压力测试的设备,在测试过程中应该与系统断开或使用其他合适的方式与测试系统隔离开;Equipment which is not to be |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第23页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
subjected to the pressure test shall be either disconnected from the tubing or isolated by proper means during the test. |
 | 可以安装测试过程需要的阀门;Valves may be used provided that they are suitable for the proposed test procedure. |
 | 在管线上的压力计不应在超过量程的压力下测试。没有表明测试压力的部件和设备应排除在测试内容之外;In line pressure gauges shall not be subjected to pressure in excess of their scale range. All pieces of equipment which do not have their test pressures indicated or declared or not proper shall be excluded from these tests. |
 | 在进行所有测试之前,需目测管线系统以保障没有可见的缺陷且所有连接紧密;Before every test the tubing systems shall have visually inspected to assure that there are no visual defects and that all connections are tightened. |
 | 控制阀门只要不是正在被测试,应该打在全开的位置;Control valves, unless being tested, shall be set and maintained in the wide open position. |
 | 只要有可能应将所有在管线上的设备,表玻璃,流量计,漂浮液位计和所有其他受压部分包含在测试之内;All in-line instruments, gauge glasses, flow meters, liquid level float gauges and all other pressure parts of instruments shall be included in these test, where feasible. |
 | 水压测试的压力应由测试压力在其量程一半处的计量表测量;Hydrostatic test pressure must be measured by gauges having the test pressure value approximately in its mid-range. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第24页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
 | 有缺陷的连接点应该被移除并重新测试;Joints found to be defective shall be removed and retested. |
 | 在修复后重新进行测试后测试压力需和原来指定测试压力相同。Retesting of lines after repair shall be done at pressures originally specified for the test |
1.5.2. 测试报告Test reports
承包商应为每个测试做相应记录,记录包含‘水压测试证书’和测试编号。The subcontractor shall make a record of the test as per specimen included “Hydraulic Test Certificate “, Certification Code
“ CER-003 ”
 | 测试编号test number; |
 | 测试日期date of test |
 | 系统名称system name; |
 | 测试媒介,压力和相应压力下维持的时间test media, pressure applied and length of time at test pressure 。 |
1.5.3. 水压压力测试Hydrostatic test pressure
 | 水压测试压力需根据项目技术要求进行,不需进行压力测试的容器和组件应被隔离。The hydrostatic test pressure shall be calculated in accordance with the project specifications: vessels or components which are not designed for the testing pressure shall be isolated. |
 | 所有水压测试系统应在设计压力的1.5 倍和至少3.5 巴的压力下进行测试。在进行连接点目测前测试压力需维持至少15 分钟。All |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第25页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第7 部分管道系统施工规范 Construction Regulation | ||
hydrostatically tested systems shall be tested to a one-half ( 1 1/2 ) times the design pressure or to a minimum pressure for 3.5 bar whichever is greater. All test pressures shall be maintained a minimum of 15 minutes before visual examination of joints beings. |
 | 在待测试的管线系统和测试的媒介达到热平衡之后再进行水压测试。Hydrostatic test pressure shall not be applies until the tubing system and the testing medium reached thermal equilibrium. |
 | 在测试过程中需对所有连接点进行目测。All joints shall be visually examinated for leakage during the test. |
 | 水压测试压力至少需维持两个小时。Hydrostatic test pressure will be maintained for at least two hours. |
 | 在含有水的管线部分不能做任何修复焊接。No repairs welding shall be done on any section of tubing that contains water. |
 | 所有不锈钢管线在水压测试结束后需立即排空。All stainless steel lines shall be emptied immediately after hydrostatic tests completion. |
1.5.4. 系统清洁和清洗System cleaning and flushing
 | 清洗和清洁全部安装好的管线,应按照供应商要求:不锈钢清洗,酸洗和钝化规程执行。Washing and cleaning of the whole piping installed will be carried out according to Supplier document requirement : STAINLEES STEEL WASHING, PICKLING AND PASSIVATION”. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第26页/ 共26 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第8 部分售后服务和技术承诺 Technical Promise and after-sale Service | ||
第 8 部分
Part 8
售后服务和技术承诺
Technical Promise and
After-sale Service
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第8 部分售后服务和技术承诺 Technical Promise and after-sale Service | ||
目 录
1.售后服务和承诺Technical Promise and after-sale Service | 2 |
1.1. 技术承诺Technical Promise | 2 |
1.2. 售后服务After-sale Service | 2 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共3 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第8 部分售后服务和技术承诺 Technical Promise and after-sale Service | ||
1.售后服务和承诺Technical Promise and after-sale Service
1.1. 技术承诺Technical Promise
上海朗脉科技有限公司,拥有先进的焊接设备和质量检测设备,以及完全符合中国GMP 标准,FDA 认证以及其他国家标准的文件系统。工程中将使用验证过的全自动轨迹焊机进行焊接。所有的焊工都具有相关的上岗资格,至少要有2 年的实际工作经验或者经过工厂培训。上海朗脉科技有限公司承诺,我们承接的工程将完全按照GMP 标准进行施工。
工程质量保证期为1 年。
Macroprocess Technologies Co., Ltd. has advanced welding machine, quality-checking equipment and integrated files system according with Chinese GMP standard, FDA GMP standard and other country’s standards. Qualified automatic orbital welding machines will be used in our projects. All welders are qualified either by 2 year field experience or by factory training. The seller promises that the project we will do is completely according to GMP.
The Quality Guarantee Period of project will be 1 year.
1.2. 售后服务After-sale Service
 | 售后服务联络:买方可直接与卖方联系,并将派自己的维修人员到买方工厂进行售后服务。After-sale service contact: The Buyer can contact the Seller directly. The seller will dispatch its’ own technicians to provide after-sale service on the Buyer’s site. |
 | 售后服务的响应时间:接到买方报修传真后2 天内 (公共假日除外)。Responding time: within two day from the receipt of written report from the Buyer (public holidays are excluded). |
 | 售后服务的质量要求:卖方应派遣有实际工作经验的工程技术人员来买方工厂进行维修。卖方技术人员需在接到通知后48 小时内到达 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共3 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第8 部分售后服务和技术承诺 Technical Promise and after-sale Service | ||
买方现场 (公共假日除外)。Requirement of service quality: The seller shall dispatch engineers with good experience to the Buyer’s premise. The seller should arrive the buyer’s site within 48 hours after receipt of notice (public holidays are excluded). |
 | 售后服务的费用:质保期内免费维修,维修人员的差旅费、人工费、食宿费等由卖方承担 (因买方操作不当、未采用合同文件中规定的主材和操作环境条件、合同供货范围以外的工作除外)。质保期外的维修费由买方承担,收费标准由双方友好协商。Expense for after-sale service: The seller is responsible to provide free technical service within the Quality Guarantee Period. All the travel and accommodation expenses as well as the labor costs of the service engineer are to be born by the seller (exclusive of situations that problems are brought by incorrect operation and works beyond the scope of supply). The Buyer should be responsible for the service expenses after the guarantee period and the price is determined through friendly discussion. |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共3 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第9 部分偏离和图纸 Deviation and Drawings | ||
第 9 部分
Part 9
偏离及图纸
Deviation and Drawings
上海朗脉科技有限公司
ShangHai MacroProcess Technology Co.,Ltd
海朗脉科技有限公司 上海市闵行区罗阳路168 号A 座101 室 201100 T:021 54306717 F: 021 54306718 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第9 部分偏离和图纸 Deviation and Drawings | ||
目 录
9. 偏离及图纸Deviation and Drawings | 2 |
9.1. 商务偏离表Deviation Form for Commercial Terms | 2 |
9.2. 技术偏离表Deviation Form for Technical Specifications | 3 |
9.3. 图纸Drawings | 4 |
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第1页/ 共4 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第9 部分偏离和图纸 Deviation and Drawings | ||
9. 偏离及图纸Deviation and Drawings
9.1. 商务偏离表Deviation Form for Commercial Terms
商务偏离表
Deviation Form for Commercial Terms
| 项目名称 | 压缩空气及工艺气体系统 Compressed Air and Process Gas System | ||
| 数 量 | 1 套 | 规格型号 | |
| 标 书要求 | 投标参数及其它 | ||
付款方式: 合同签订后10 日内,甲方支付合同总额10%给乙方,同时乙方向甲方提供相同金额的发票; 乙方根据甲方提供的设计资料完成工程的二次设计,并经甲方书面确认后10 日内,甲方支付合同总额20%给乙方,同时乙方向甲方提供相同金额的发票;全部设备和材料运输到施工现场指定位置,甲乙双方开箱验收完毕,10 日内甲方支付合同总额30%给乙方, 同时乙方向甲方提供相同金额的发票。安装、调试、验收、验证合格并交付甲方使用后,10日内甲方再支付合同总价30%,同时乙方向甲方提供相同金额的发票;余合同总价10%的作为质量保证金,质量保证期1 年, 质保期满,无质量问题,10 日内甲方付清全部余款,同时乙方向甲方提供相同金额的发票。 | 付款方式: 合同签订后,10日内甲方支付合同总额30%预付款;全部设备和材料运输到施工现场指定位置前,甲方支付合同总额40%; 安装、调试、验收合格���交付甲方使用后,10 日内甲方再支付合同总额25%, 余合同总价5%作为质量保证金,质量保证期1 年, 质保期满,无质量问题,10日内甲方付清全部余款。 | ||
投标人(公章): 上海朗脉科技有限公司
授权代表人(签字或盖章):
日期:
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第2页/ 共4 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第9 部分偏离和图纸 Deviation and Drawings | ||
9.2. 技术偏离表Deviation Form for Technical Specifications
技术偏离表
Deviation Form for Technical Specifications
| 项目名称 | 压缩空气及工艺气体系统 Compressed Air and Process Gas System | ||||
| 数 量 | 1 套 | 规格型号 | |||
| 标 书 要 求 | 投标参数及其它 | ||||
无偏离 | |||||
投标人(公章): 上海朗脉科技有限公司
授权代表人(签字或盖章):
日期:
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第3页/ 共4 页 |
 | 沈阳三生制药有限责任公司 Shenyang Sunshine Pharmaceutical Company Limited |  | |
压缩空气及工艺气体系统 Compressed Air and Process Gas System | |||
投标文件 Bidding Documents | 第9 部分偏离和图纸 Deviation and Drawings | ||
9.3. 图纸Drawings
●PID 流程图 PID
●项目进度表 Schedule
上海市闵行区罗阳路168 号A 座101 室 201100 1F, building A, No.168 LuoYang Rd, Shanghai, China 201100 T:021 54306717 F: 021 54306718 | 第4页/ 共4 页 |




