
- 1 - Please note that the following is an English translation of the original Japanese version, prepared only for the convenience of shareholders residing outside Japan. In the case of any discrepancy between the translation and the Japanese original, the latter shall prevail. TAKEDA PHARMACEUTICAL COMPANY LIMITED (“TAKEDA”) HEREBY DISCLAIMS ALL REPRESENTATIONS AND WARRANTIES WITH RESPECT TO THIS TRANSLATION, WHETHER EXPRESS OR IMPLIED INCLUDING, BUT WITHOUT LIMITATION TO, ANY REPRESENTATIONS OR WARRANTIES WITH RESPECT TO ACCURACY, RELIABILITY OR COMPLETENESS OF THIS TRANSLATION. IN NO EVENT SHALL TAKEDA BE LIABLE FOR ANY DAMAGES OF ANY KIND OR NATURE INCLUDING, BUT WITHOUT LIMITATION TO, DIRECT, INDIRECT, SPECIAL, PUNITIVE, CONSEQUENTIAL OR INCIDENTAL DAMAGES ARISING FROM OR IN CONNECTION WITH THIS TRANSLATION. Items Disclosed via the Internet Concerning the Notice of Convocation of the 145th Ordinary General Meeting of Shareholders 1. Following items of the Business Report Matters Concerning the Stock Acquisition Rights of the Company Overview of the Systems that Ensure the Appropriateness of Operations of the Company and the Status of Implementation of such Systems 2.Consolidated Statement of Changes in Equity on the Consolidated Financial Statements 3.Notes to the Consolidated Financial Statements 4.Unconsolidated Statement of Changes in Net Assets on the Unconsolidated Financial Statements 5.Notes to the Unconsolidated Financial Statements Takeda Pharmaceutical Company Limited (the “Company”) The items listed above are the information which shall be deemed to have been provided to shareholders through posting on the Company’s website in the internet (https://www.takeda.com/investors/shareholders-meetings/) based on laws and regulations and Article 14 of the Company’s Articles of Incorporation.
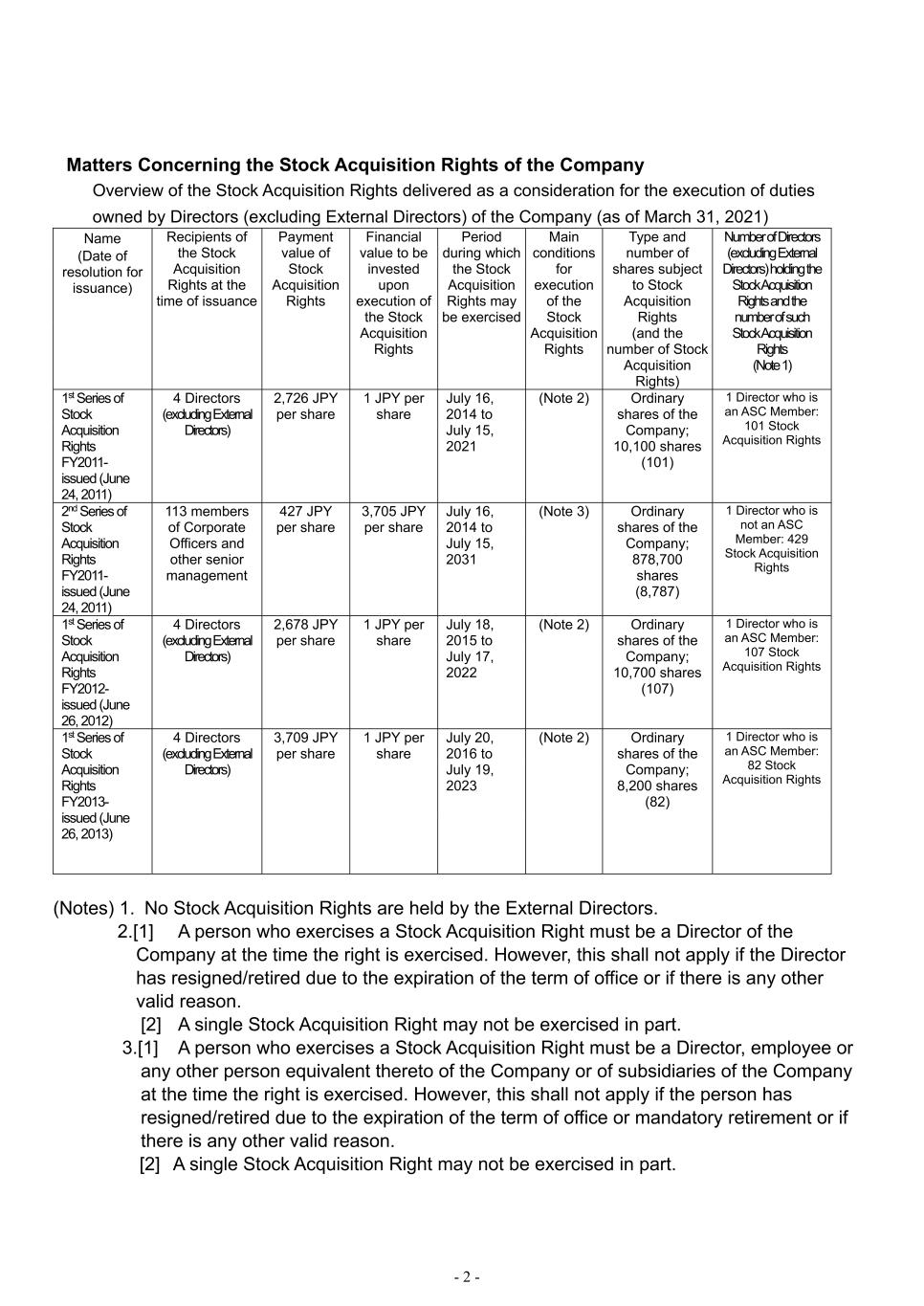
- 2 - Matters Concerning the Stock Acquisition Rights of the Company Overview of the Stock Acquisition Rights delivered as a consideration for the execution of duties owned by Directors (excluding External Directors) of the Company (as of March 31, 2021) Name (Date of resolution for issuance) Recipients of the Stock Acquisition Rights at the time of issuance Payment value of Stock Acquisition Rights Financial value to be invested upon execution of the Stock Acquisition Rights Period during which the Stock Acquisition Rights may be exercised Main conditions for execution of the Stock Acquisition Rights Type and number of shares subject to Stock Acquisition Rights (and the number of Stock Acquisition Rights) Number of Directors (excluding External Directors) holding the Stock Acquisition Rights and the number of such Stock Acquisition Rights (Note 1) 1st Series of Stock Acquisition Rights FY2011- issued (June 24, 2011) 4 Directors (excluding External Directors) 2,726 JPY per share 1 JPY per share July 16, 2014 to July 15, 2021 (Note 2) Ordinary shares of the Company; 10,100 shares (101) 1 Director who is an ASC Member: 101 Stock Acquisition Rights 2nd Series of Stock Acquisition Rights FY2011- issued (June 24, 2011) 113 members of Corporate Officers and other senior management 427 JPY per share 3,705 JPY per share July 16, 2014 to July 15, 2031 (Note 3) Ordinary shares of the Company; 878,700 shares (8,787) 1 Director who is not an ASC Member: 429 Stock Acquisition Rights 1st Series of Stock Acquisition Rights FY2012- issued (June 26, 2012) 4 Directors (excluding External Directors) 2,678 JPY per share 1 JPY per share July 18, 2015 to July 17, 2022 (Note 2) Ordinary shares of the Company; 10,700 shares (107) 1 Director who is an ASC Member: 107 Stock Acquisition Rights 1st Series of Stock Acquisition Rights FY2013- issued (June 26, 2013) 4 Directors (excluding External Directors) 3,709 JPY per share 1 JPY per share July 20, 2016 to July 19, 2023 (Note 2) Ordinary shares of the Company; 8,200 shares (82) 1 Director who is an ASC Member: 82 Stock Acquisition Rights (Notes) 1. No Stock Acquisition Rights are held by the External Directors. 2.[1] A person who exercises a Stock Acquisition Right must be a Director of the Company at the time the right is exercised. However, this shall not apply if the Director has resigned/retired due to the expiration of the term of office or if there is any other valid reason. [2] A single Stock Acquisition Right may not be exercised in part. 3.[1] A person who exercises a Stock Acquisition Right must be a Director, employee or any other person equivalent thereto of the Company or of subsidiaries of the Company at the time the right is exercised. However, this shall not apply if the person has resigned/retired due to the expiration of the term of office or mandatory retirement or if there is any other valid reason. [2] A single Stock Acquisition Right may not be exercised in part.

- 3 - Overview of the Systems to Ensure the Appropriateness of Operations of the Company and the Status of Implementation of such Systems (1) Overview of the systems to ensure the appropriateness of operations The Company refreshed its “Corporate Philosophy,” which comprises its “Purpose,” “Values: Takeda-ism,” “Vision” and “Imperatives.” The Company shares the Corporate Philosophy within the entire Takeda Group and is making an effort to further promote the creation of a corporate culture based on the “Corporate Philosophy.” Considering internal control as an important component of corporate governance that functions alongside risk management, the Company undertakes to develop its internal control system as described below. (i) Systems to ensure the appropriateness of operations in the Takeda Group As a “company with an Audit and Supervisory Committee (ASC),” the Company has developed a system that enables the ASC to effectively perform its duties relating to audit and supervision and increased the composition ratio and diversity of the External Directors in the Board of Directors. Under the appropriate audit and supervision realized through such measures, the Board of Directors makes highly transparent and objective decisions and, by its resolution, delegates authority to Directors to expedite the management of business. The objectivity and fairness of the election of Directors and the compensation paid to them are ensured by the voluntary establishment of the Nomination Committee and the Compensation Committee, as advisory bodies for the Board of Directors, in each of which an External Director serves as the chairperson and external committee members constitute a majority. By appointing one or more Directors who are ASC Members as members of such committees, the effectiveness of the ASC’s function of supervising the election, etc. of Directors who are not ASC Members and the compensation, etc. paid to them is enhanced. By resolution of the Board of Directors, the authority to decide the amount of individual remuneration of Internal Directors who are not ASC Members has been delegated to the Compensation Committee, through which the Company has realized a more transparent process in determining individual remuneration. Under the system above, the Board of Directors (i) decides on the most important matters for the business operation of the Takeda Group, including matters relating to the Corporate Philosophy and matters relating to internal control, such as compliance, and risk management, (ii) discusses business strategy, and (iii) monitors and supervises the business execution. To strengthen its global business management system, the Company has established the Takeda Executive Team (TET), which consists of the President & CEO and the members who manage and supervise each function of the Takeda Group, and also established the Business Review Committee (which is responsible for general management matters), the Portfolio Review Committee (which is responsible for R&D and product related matters), and the Risk, Ethics & Compliance Committee (which is responsible for risk management, corporate ethics and compliance matters). These committees review important matters and thereby ensure systems which enable faster and more flexible business execution and closer collaboration among the various functions. By resolution of the Board of Directors, decision-making authority on important matters of business execution is partially delegated to Directors subject to approval of decision-making bodies such as the Business Review Committee, the Portfolio Review Committee, and the Risk, Ethics & Compliance Committee, and thereby the Company conducts flexible and efficient decision-making. The Company clarifies the roles and responsibilities of each function based on the “Takeda Group’s Management Policy (T-MAP),” which summarizes the business management systems, decision-making systems, operational rules of such systems and other important management rules of the Takeda Group. The Company obliges each function to make proposals or reports to the decision-making bodies, including the Board of Directors,

- 4 - depending on the materiality of items. Concurrently, the Company delegates a certain level of decision-making authority to the President & CEO or to other TET members, and such decision-making authority is exercised under proper governance. Each TET member has developed operating procedures and rules for delegating authority and established an adequate internal control structure in the divisions which they oversee. In order to manage and supervise the entire Takeda Group in a cross-sectoral and unified manner, the Company has established the Global Policies, etc. (Global Policies mean the rules that apply to employees of three or more TET organizations) for the respective responsibilities of the specialized functions. With regard to risk management and management of a crisis that may occur in the Takeda Group, the structure of the risk management system and the system to manage the existing crisis including BCPs (Business Continuity Plans) of the Takeda Group have been laid out based on the “Global Risk Management Policy” and the “Global Crisis Management Policy.” The Global Ethics & Compliance division is working on disseminating the “Takeda Global Code of Conduct” to all group companies and developing and disseminating ethics and compliance programs for all group companies. The Global Ethics & Compliance division has developed a monitoring mechanism to ensure that the Takeda Group's business activities are in compliance with laws and regulations, internal policies and SOPs. In addition, the Global Ethics & Compliance division periodically reports to the Risk, Ethics & Compliance Committee and the ASC, and reports to the Board of Directors, as necessary, on the ethics and compliance related affairs of the Takeda Group, including issues reported through the internal reporting system for whistleblowers. The Group Internal Audit (GIA) division conducts a regular internal audit of each function of the Company and each group company based on the “Group Internal Audit Charter,” and reports the results thereof to the President & CEO, the ASC, and the Board of Directors. The head of each division and each subsidiary of the Company has developed and implements an internal control system over financial reporting based on the 2013 Internal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in order to comply with the Japanese Financial Instruments and Exchange Act and the Cabinet Office Order and the U.S. Sarbanes-Oxley Act. The Global Finance division promotes the development and implementation of the internal control system through the processes of (i) self-inspection based on questionnaires on internal control over the financial reporting completed by the head of each division and subsidiary, and (ii) implementation of the improvement plan in response to indications and/or recommendations. The GIA division assesses the effectiveness of the development and implementation of the internal control system over financial reporting. The Global Quality division, which formulated the Global Quality Policy, etc., relating to research, development, manufacturing, and post-marketing safety measures, conducts audits and monitors and supervises compliance therewith regularly or as necessary. The Corporate EHS (environment, health and safety) department in the Global Manufacturing & Supply division, which established the "Global Environment, Health and Safety Policy and Position," etc., conducts audits regularly or as necessary. Also, it provides support and advice to reduce risks regarding the environment, occupational health and safety. (ii) System for retention and management of information concerning the execution of the duties of Directors The minutes of the meetings of the Board of Directors, requests for and approvals of managerial decisions, and other information concerning the execution of the duties of Directors are appropriately retained and managed in conformity with the predetermined term, method and place of retention designated for each category of information in accordance with the “Global Records and Information Management (RIM) Policy,” in either hard copy or electronic or magnetic record, and in a manner where they are available for inspection.

- 5 - (iii) Rules and other systems for managing the risk of loss Based on the “Global Risk Management Policy,” Enterprise Risk Management (ERM) is conducted through a five-step approach, which is the identification, assessment, mitigation, reporting, and monitoring of the risk. The Company has developed a system where major potential risks and their mitigation plans are reported to the Risk, Ethics & Compliance Committee and the Board of Directors on an annual basis. Specifically, with respect to all risk factors, the heads of functions or the risk owner(s) of the major potential risks control and manage such risk factors in each area under their charge, and take the necessary measures to mitigate such risk factors, depending on the degree and content of the risk. In addition, where deemed necessary, Business Continuity Plans have been developed for key risks concerning, for example, manufacturing sites and IT cybersecurity. For crisis management in emergency situations, the Company has developed a crisis management system structured around the Crisis Management Committee in accordance with the “Global Crisis Management Policy.” (iv) System to ensure that the duties of Directors are executed efficiently A system under which the duties of Directors are executed appropriately and efficiently is ensured by the “Bylaws of Board of Directors” and other internal company regulations relating to authorities and rules for decision-making. (v) Systems to ensure that Directors and employees comply with laws and regulations and the Company’s Articles of Incorporation in executing their duties The Company has established the Chief Ethics & Compliance Officer and the Global Ethics & Compliance division to support each division. The Company also implements ethics and compliance programs across the organization. The Company has established procedures for the receipt, retention, investigation and handling of reports by whistleblowers related to any violations of laws and regulations, Takeda’s Global Code of Conduct, Global Policy or internal policies or SOPs, including those related to the Company’s accounting, internal accounting controls, or accounting audits. The Company has also established procedures for confidential and anonymous whistleblowing by Takeda employees through the Takeda Ethics Line. (vi) System to ensure that the audits by the Audit and Supervisory Committee are conducted effectively In accordance with the “Rules of Audit and Supervisory Committee’s Audit, etc.”: A full-time ASC Member is appointed, and the ASC Office, which is a clerical section dedicated to the ASC, is established to assist operations of the ASC and serve as its secretariat. Appointment and the personnel change of the members of the ASC Office require consent of the ASC in order to secure the independence of the ASC Office from persons executing the business and the effectiveness of instructions from the ASC. Directors inform the ASC of matters concerning the Company’s basic management policy and plans, and material matters including those involving subsidiaries and affiliates in advance (provided, however, that this does not apply if the ASC Members attend the meeting of the Board of Directors or any other meeting at which such matter is deliberated or reported). If a Director becomes aware of any fact that might cause material damage to the Takeda Group, such Director immediately reports such fact to the ASC. The ASC has appointed the “Appointed ASC Members” who have the authority to request Directors and employees to report on matters relating to the performance of their duties, investigate the status of the operations and properties of the Company and perform part of the other duties of the ASC. Based on the status of development and implementation of the internal control system and other relevant circumstances, the ASC closely communicates with the internal audit division, the internal control promotion division and the Accounting Auditor, to which the ASC is

- 6 - authorized to give instructions, and enhances the effectiveness and efficiency of the audit by conducting a systematic audit utilizing the information received from them. The ASC Members request the Company to reimburse their costs for performing their duties, and submit budget thereof to the Company every year. The ASC makes proposals or states its opinions to the Board of Directors, as necessary, with respect to systems to ensure that any person who makes a report to the ASC and the internal audit division, etc., including a report made through the internal reporting system for whistleblowers, would not be subject to any unfavorable treatment on account of such reporting. (2) Overview of the status of the implementation of systems to ensure the appropriateness of operations During this fiscal year, the Company made efforts to appropriately implement the systems described in (1) above. The major efforts made by the Company in this fiscal year that are considered important for internal control include the following: [Dissemination of the Corporate Philosophy] The Company refreshed its “Corporate Philosophy” consisting of the “Purpose,” “Values: Takeda-ism,” “Vision” and “Imperatives.” TET members, including the President & CEO, have been making efforts to disseminate the Corporate Philosophy across the Takeda Group and to employees by sending messages internally, holding town hall meetings and other means. [Strengthening of the Corporate Governance Structure] Along with the Company’s conversion into a “company with an Audit and Supervisory Committee” in 2016, the Company increased the composition ratio and diversity of its External Directors so that the Board of Directors and the ASC can conduct their respective responsibilities more appropriately. Of the 16 members of the Board of Directors (including one female director) as of the end of this fiscal year, 11 are External Directors, and eight Directors are Japanese and eight are foreign nationals. Furthermore, four Directors make up the ASC, and three of them are External Directors. [Status of the Board of Directors] The Board of Directors held eight meetings during this fiscal year. At the Board of Directors meetings, the Chairman of the Board, who is an Independent and External Director, acted as a chairperson, and diverse Directors, including the External Directors who are highly independent from the Company, stated opinions as appropriate from their own perspectives. As mentioned above, by delegating to Directors the authority to decide on important matters on business execution, the Board of Directors allocates more time to deliberate issues that can have a significant impact on the Takeda Group and its management strategies and to oversee Directors' performance on business execution. Before every Board of Directors meeting, External Directors are given an explanation of the agenda items of the meeting by Directors who are not External Directors. In addition, when the External Directors are newly appointed, the Company makes sure they thoroughly understand their legal obligations and also provides them with information relating to the business environment, strategy, etc., of the Company. They are also provided with opportunities to further deepen their understanding thereof. At the Board of Directors meetings, each External Director expressed their opinions as appropriate during the deliberations on the agenda items of the Board of Directors meetings based on (i) their advanced insight derived from experience in corporate management, or (ii) their high level of knowledge in highly specialized areas such as accounting and law. In addition, a meeting of only the External Directors was held to allow them to share their understanding and exchange views and opinions on the management of the Board of Directors and how it should be involved in the business operation. In this fiscal year, the Company conducted an evaluation of the performance and effectiveness of the Board of Directors. A third party organization was appointed for this purpose, and the evaluation was conducted in a manner to facilitate obtaining of opinions from each person, such as having all Directors individually complete a questionnaire and be individually interviewed. Based on the results of this evaluation, the Board of Directors of the Company is considered to be effective, and (i) improvements were confirmed with regard to

- 7 - matters that were pointed out in evaluations in the past, and (ii) there was no important matter which was newly pointed out. After incorporating the analysis and recommendations made by the third party organization, the results of this evaluation were discussed by all members of the Board of Directors. The Company considers this process to be an opportunity to gain a deeper understanding of the strengths of the Board of Directors and to further enhance its functions. [Efforts to develop the internal control system in the Takeda Group] With regard to matters other than those that need to be resolved by decision-making bodies, including the Board of Directors, the Business Review Committee, the Portfolio Review Committee, and the Risk, Ethics & Compliance Committee, the decision-making authority is delegated to the members of the TET which consists of the President & CEO and the representatives of each function. The delegation of authority from TET members to their subordinates is conducted based on the “Global Policy - Delegation of Authorities.” The GIA division conducted an internal audit of each function of the Company and each company under the Takeda Group, as well as an evaluation of the status of development and implementation of the internal control system to secure the reliability of financial reporting. With regard to the status of internal control on financial reporting, the Global Finance division confirmed the effectiveness of the internal control of each division and key subsidiaries of the Company based on the answers to self-inspection through questionnaires received from the head of each division and key subsidiaries. Further, the Global Finance division reported the results of Management’s internal control testing to the ASC. The Global Quality division clarified the Company's commitment to, and vision for, quality, and conducts global quality assurance for the Takeda Group based on the “Global Quality Policy.” The Corporate EHS department clarified the roles and responsibilities in order to promote activities for the environment, occupational health and safety management of the Company, and conducted internal audits within the Takeda Group from the perspective of management of the environment, occupational health and safety, and compliance by setting specific targets based on the "Global Environment, Health and Safety Policy and Position," etc. [Efforts to promote compliance] The monitoring of fields with potentially high compliance-related risks was conducted at each division, and continuous improvements are being made. Takeda Group’s compliance-related issues are regularly reported to the Risk, Ethics & Compliance Committee and the ASC, and to the Board of Directors and the TET in a timely manner. [Efforts relating to risk management] This fiscal year, important risks for each region and division were discussed and validated at the Risk, Ethics & Compliance Committee. Thereafter, such risks were registered as corporate risks and a risk map was developed. The risk map was reported to the Board of Directors. Also, a risk mitigation plan for important risks was developed and the effectiveness thereof was monitored based on evaluation indicators. Other concrete efforts relating to risk management for this fiscal year are as follows: Through the risk coordinator community within the Takeda Group, the Company promotes upskilling in risk management practices and knowledge sharing. In addition, the Company undertook education and drills for its employees, the purpose of which was to enhance consciousness on responding to crises, including earthquakes, pandemics, and product recalls. The Company conducted the following actions for cybersecurity: The Information Security & Governance Board continued to meet monthly and on an ad hoc basis. This body consists of representatives from all Takeda business units/functions and discussed relevant information risk topics and reviewed the status of actions taken to mitigate such risks. Enhanced training modules were provided to all the employees in order to strengthen cybersecurity awareness and address emerging threats.

- 8 - Investments continued to be made to strengthen security in the process and technical aspects of Takeda’s data and IT infrastructure. Insurance is held to cover certain costs related to significant cybersecurity events that Takeda may face in the future. The Global Crisis Management Committee on COVID-19 was established, which issued timely guidance such as travel restrictions and recommendations for working from home to encourage employees to act appropriately in an effort to prevent the spread of the disease. [Efforts by the Audit and Supervisory Committee] The ASC is managed based on the “Rules of Audit and Supervisory Committee’s Audit, etc.” The ASC meetings, at which an ASC Member who is an External Director serves as the chairperson, were held ten times during this fiscal year, and the members exchanged information and opinions relating to matters such as the agenda at the Board of Directors meetings, status of the execution of the business by Directors and the internal control system. All ASC Members shared information obtained by a full-time ASC Member based on activities such as attendance at important meetings, collection of information on a regular basis and periodical hearing of reports relating to the business performance of the division in charge of executing the business operation, and through cooperation or collaboration with the internal audit division or the internal control promotion division. The audit opinions were formed in the ASC through the activities mentioned above. The ASC reported on the result of the activities of the previous fiscal year and its action policy and activity plan for this fiscal year, and exchanged opinions at the Board of Directors meeting. Also, as necessary, the ASC gave its opinion on the execution of the business by Directors. The ASC had meetings to exchange opinions with the GIA division regularly or as necessary and conducted a systematic audit by providing instructions or making requests, in addition to receiving a report relating to the plan and result of the internal audit. The Appointed ASC Members attended the Nomination Committee and the Compensation Committee as members of those committees, and stated their opinions relating to the election, etc. of Directors who are not ASC Members and their compensation, etc. Also, information obtained from those committees was shared at the ASC, and through this and other relevant processes, the ASC formulated its opinion, and performed its duties of supervision.
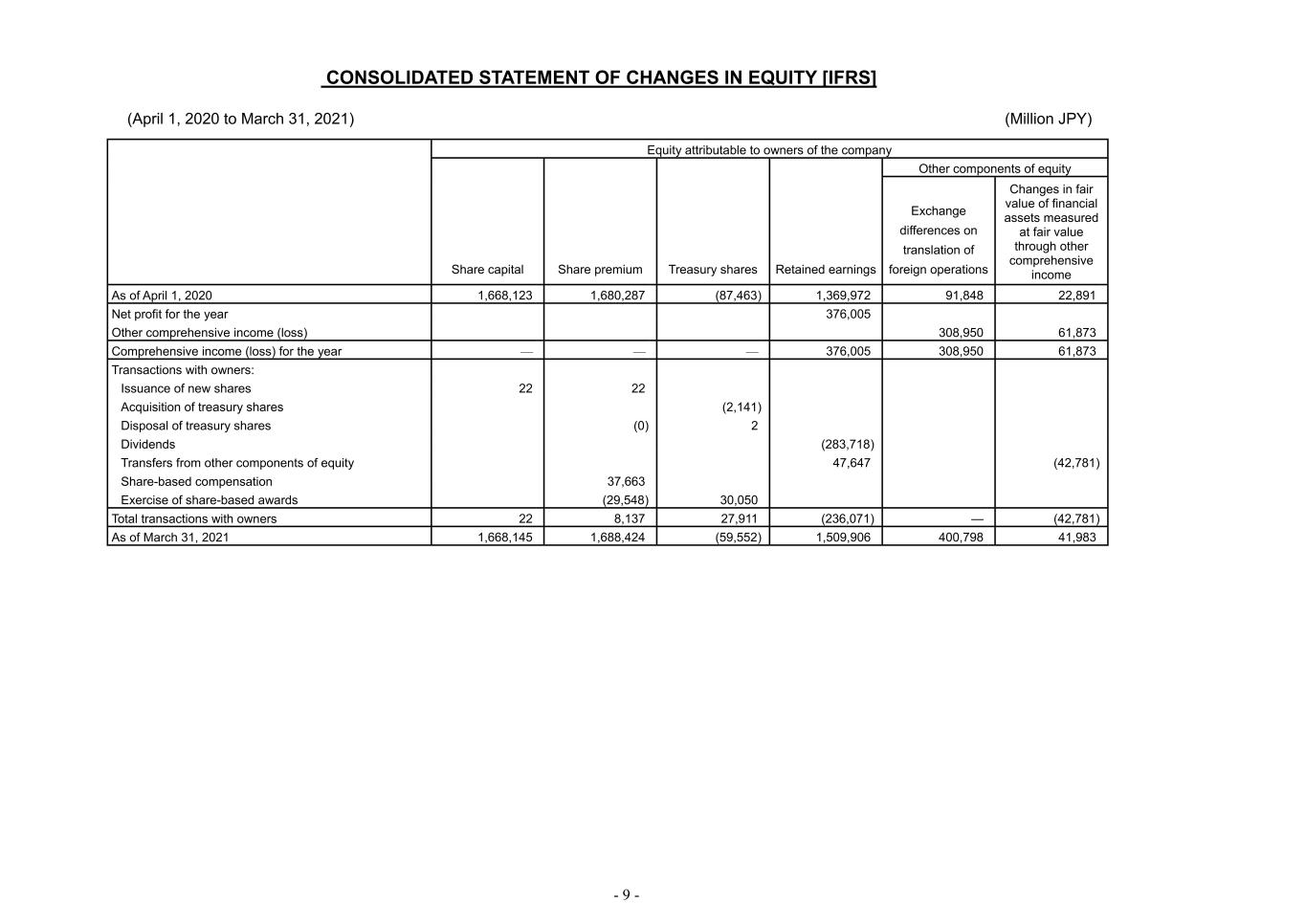
- 9 - CONSOLIDATED STATEMENT OF CHANGES IN EQUITY [IFRS] (April 1, 2020 to March 31, 2021) (Million JPY) Equity attributable to owners of the company Share capital Share premium Treasury shares Retained earnings Other components of equity Exchange differences on translation of foreign operations Changes in fair value of financial assets measured at fair value through other comprehensive income As of April 1, 2020 1,668,123 1,680,287 (87,463) 1,369,972 91,848 22,891 Net profit for the year 376,005 Other comprehensive income (loss) 308,950 61,873 Comprehensive income (loss) for the year — — — 376,005 308,950 61,873 Transactions with owners: Issuance of new shares 22 22 Acquisition of treasury shares (2,141) Disposal of treasury shares (0) 2 Dividends (283,718) Transfers from other components of equity 47,647 (42,781) Share-based compensation 37,663 Exercise of share-based awards (29,548) 30,050 Total transactions with owners 22 8,137 27,911 (236,071) — (42,781) As of March 31, 2021 1,668,145 1,688,424 (59,552) 1,509,906 400,798 41,983
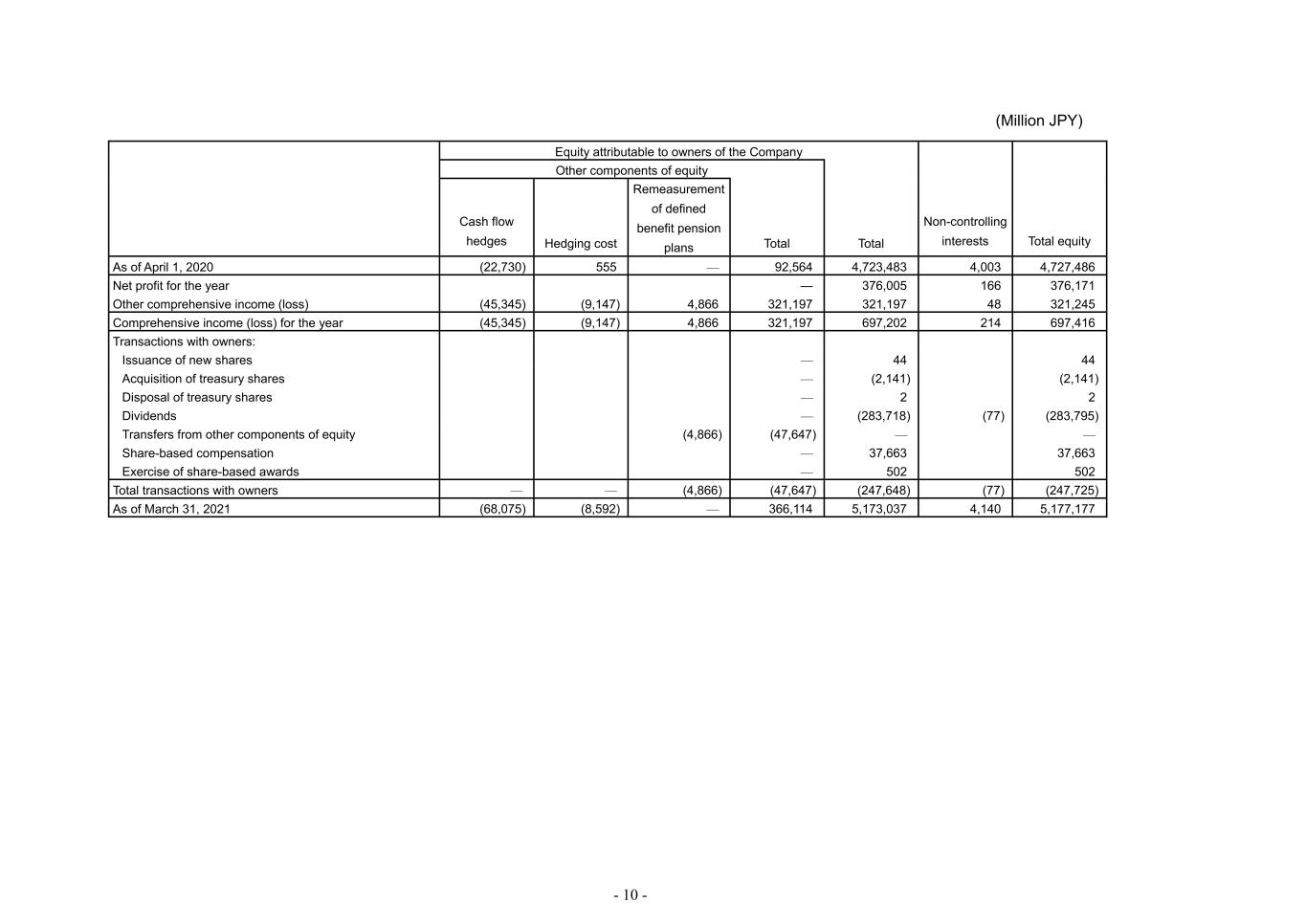
- 10 - (Million JPY) Equity attributable to owners of the Company Non-controlling interests Total equity Other components of equity Total Cash flow hedges Hedging cost Remeasurement of defined benefit pension plans Total As of April 1, 2020 (22,730) 555 — 92,564 4,723,483 4,003 4,727,486 Net profit for the year — 376,005 166 376,171 Other comprehensive income (loss) (45,345) (9,147) 4,866 321,197 321,197 48 321,245 Comprehensive income (loss) for the year (45,345) (9,147) 4,866 321,197 697,202 214 697,416 Transactions with owners: Issuance of new shares — 44 44 Acquisition of treasury shares — (2,141) (2,141) Disposal of treasury shares — 2 2 Dividends — (283,718) (77) (283,795) Transfers from other components of equity (4,866) (47,647) — — Share-based compensation — 37,663 37,663 Exercise of share-based awards — 502 502 Total transactions with owners — — (4,866) (47,647) (247,648) (77) (247,725) As of March 31, 2021 (68,075) (8,592) — 366,114 5,173,037 4,140 5,177,177

- 11 - Notes to the Consolidated Financial Statements [Notes for Items that Form the Basis of Preparing Consolidated Financial Statements] 1. Accounting Standards of Consolidated Financial Statements The consolidated financial statements are prepared in accordance with International Financial Reporting Standards ("IFRS"), in compliance with Article 120, Paragraph 1 of the Regulation on Corporate Accounting. In compliance with the second sentence of the same paragraph, certain disclosures required under IFRS are omitted. 2. Scope of Consolidation (1) Number of consolidated subsidiaries: 239 Names of major consolidated subsidiaries: (Domestic) Nihon Pharmaceutical Co., Ltd. (Overseas) Takeda Pharmaceuticals U.S.A., Inc., Millennium Pharmaceuticals, Inc., Dyax Corp., Baxalta US Inc., Takeda Pharmaceuticals International AG, Takeda GmbH, Shire Pharmaceuticals International Unlimited Company, Shire Pharmaceuticals Ireland Limited, Shire Ireland Finance Trading Limited, Baxalta GmbH (2) Increase and decrease of consolidated subsidiaries: Increase : 5 (due to establishment) Decrease : 94 (due to merger, liquidation and divestiture) 3. Application of the Equity Method (1) Number of associates accounted for using the equity method: 21 Names of major associates accounted for using the equity method: Teva Takeda Pharma Ltd., Amato Pharmaceutical Products, Ltd. (2) Increase and decrease of associates accounted for using the equity method: Increase : - Decrease : 1 (due to divestiture) 4. Significant Accounting Policies (1) Valuation Standards and Methods for Major Assets (excluding Financial Instruments) 1) Property, Plant and Equipment Property, plant and equipment are measured using the cost model and is stated at cost less accumulated depreciation and accumulated impairment loss. Acquisition cost includes mainly the costs directly attributable to the acquisition and the initial estimated dismantlement, removal, and restoration costs associated with the asset. 2) Goodwill Goodwill arising from business combinations is stated at its cost less accumulated impairment losses. Goodwill is not amortized. Goodwill is allocated to cash-generating units or groups of cash- generating units based on expected synergies and tested for impairment annually or whenever there is any indication of impairment. Impairment losses on goodwill are recognized in the consolidated statements of profit or loss and no subsequent reversal will be made.

- 12 - 3) Intangible Assets Intangible assets are measured by using the cost model and are stated at cost less accumulated amortization and accumulated impairment losses. Takeda regularly enters into collaboration and in-license agreements with third parties for products and compounds for research and development projects. Payments for collaboration agreements generally take the form of subsequent development milestone payments. Payments for in-license agreements generally take the form of up-front payments and subsequent development milestone payments. Up-front payments for in-license agreements are capitalized upon commencement of the in-license agreements, and development milestone payments are capitalized when the milestone is triggered. If and when Takeda obtains approval for the commercial application of a product in development, the related in-process research and development assets will be reclassified to intangible assets associated with marketed products and amortized over its estimated useful life from marketing approval. 4) Impairment of Non-financial Assets Takeda assesses whether there is any indication of impairment for non-financial assets at the end of each reporting period, excluding inventories, deferred tax assets, assets held for sale, and assets arising from employee benefits. If any such indication exists, or in cases in which an impairment test is required to be performed each year, the recoverable amount of the asset is estimated. In cases in which the recoverable amount cannot be estimated for each asset, they are estimated at the cash-generating unit level. The recoverable amount of an asset or a cash-generating unit is determined at the higher of its fair value less cost of disposal, or its value in use. In determining the value in use, the estimated future cash flows are discounted to their present value using a discount rate that reflects the time value of money and the risks specific to the asset. If the carrying amount of the asset or cash-generating unit exceeds the recoverable amount, impairment loss is recognized in profit or loss and the carrying amount is reduced to the recoverable amount. An asset or a cash-generating unit other than goodwill, for which impairment losses were recognized in prior years, is assessed at the end of the reporting period to determine whether there is any indication that the impairment loss recognized in prior periods may no longer exist or may have decreased. If any such indication exists, the recoverable amount of the asset or cash-generating unit is estimated. In cases in which the recoverable amount exceeds the carrying amount of the asset or cash-generating unit, the impairment loss is reversed up to the lower of the estimated recoverable amount or the carrying amount that would have been determined if no impairment loss had been recognized in prior years. The reversal of impairment loss is immediately recognized in profit or loss. 5) Inventories Inventories are measured at the lower of cost and net realizable value. The cost of inventories is determined mainly using the weighted-average cost formula. The cost of inventories includes purchase costs, costs of conversion, and other costs incurred in bringing the inventories to the present location and condition. Net realizable value is the estimated selling price in the ordinary course of business less the estimated costs of completion and the estimated costs necessary to make the sale. (2) Depreciation and Amortization Methods of Assets 1) Property, Plant and Equipment Except for assets that are not subject to depreciation, such as land and construction in progress, assets are depreciated mainly using the straight-line method over the estimated useful life of the asset. Right-of-use assets are depreciated using the straight-line method over the shorter of the

- 13 - lease term or the estimated useful life unless it is reasonably certain that Takeda will obtain ownership by the end of the lease term. The depreciation of these assets begins when they are available for use. The estimated useful life of major asset items is as follows: Buildings and structures 3 to 50 years Machinery and vehicles 2 to 20 years Tools, furniture and fixtures 2 to 20 years 2) Intangible Assets An intangible asset associated with a product (an intangible asset associated with a marketed product) is amortized on a straight-line basis over the estimated useful life, which is based on expected exclusivity period, ranging from 3 to 20 years. Software is amortized on a straight-line basis over the expected useful life. The useful life used for this purpose is 3 to 10 years. (3) Valuation Standards and Methods for Financial Instruments 1) Financial Assets (i) Initial Recognition and Measurement • Investments in debt instruments measured at amortized cost: Assets such as trade and other receivables that are held within a business model whose objective is to hold financial assets in order to collect contractual cash flows and whose contractual terms give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding are measured at amortized cost. Trade receivables are initially recognized at their invoiced amounts, including any related sales taxes less adjustments for estimated deductions such as impairment loss allowance and cash discounts. • Investments in debt instruments measured at fair value through other comprehensive income ("FVTOCI"): Assets that are held within a business model objective whose objective is achieved by both collecting contractual cash flows and selling financial assets whose contractual terms give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding are measured at FVTOCI. • Investments in debt Instruments measured at fair value through net profit or loss ("FVTPL"): Assets that do not meet the criteria for amortized cost or FVTOCI are measured at FVTPL. • Equity instruments measured at FVTOCI: On initial recognition, Takeda makes an irrevocable FVTOCI election (on an instrument-by-instrument basis) to present the subsequent changes in the fair value of equity instruments in other comprehensive income for certain equity instruments held for the long term for strategic purposes. At the reporting date, Takeda designates all of its equity instruments as financial assets measured at FVTOCI. (ii) Subsequent Measurement and Derecognition • Investments in debt Instruments measured at amortized cost: These assets are subsequently measured at amortized cost using the effective interest method. The amortized cost is reduced by impairment losses. Interest income, foreign exchange gains and losses and impairment are recognized in profit or loss. Any gain or loss on derecognition is recognized in profit or loss. • Investments in debt instruments measured at FVTOCI: These assets are subsequently measured at fair value. Interest income calculated using the effective interest method, foreign exchange gains and losses and impairment are recognized in profit or loss. Other net gains and losses arising from changes in fair value are recognized in other comprehensive income. Upon derecognition of the investments, the gains and losses accumulated in other comprehensive income related to the investments are reclassified to profit or loss. • Investments in debt instruments measured at FVTPL: These assets are subsequently measured at fair value, and a gain or loss on debt instruments that is subsequently measured at FVTPL is recognized in net profit or loss.

- 14 - • Equity Instruments measured at FVTOCI: These assets are subsequently measured at fair value. Dividends are recognized as income in profit or loss unless the dividend clearly represents a recovery of part of the cost of the investment. Other net gains and losses are recognized in other comprehensive income and are never reclassified to profit or loss. Upon derecognition of the investments, the amounts in other comprehensive income related to the investments are reclassified within equity to retained earnings. (iii) Impairment Loss allowances for trade receivables are established using an Expected Credit Loss ("ECL") model. The provisions are based on a forward-looking ECL, which includes possible default events on the trade receivables over the entire holding period of the trade receivables. Takeda has elected to measure provisions for trade receivables, contract assets, and lease receivables at an amount equal to lifetime ECL. Takeda uses a provisions matrix based on historical loss rates adjusted for forward looking information to calculate ECL. These provisions represent the difference between the contractual amount of the trade receivables, contract assets, and the lease receivables in the consolidated statements of financial position and the estimated collectible net amount. 2) Financial Liabilities (i) Initial Recognition and Measurement Financial liabilities are recognized in the consolidated statements of financial position when Takeda becomes a party to the contract of financial instruments. Financial liabilities are classified, at initial recognition, as financial liabilities measured at FVTPL, bonds and loans, or payables. Financial liabilities, except for those measured at FVTPL, are initially measured at fair value less transaction costs that are directly attributable to the issuance. (ii) Subsequent Measurement • Financial liabilities measured at FVTPL: Financial liabilities measured at FVTPL are measured at fair value, and any gains or losses arising on re-measurement are recognized in profit or loss. Financial liabilities measured at FVTPL include derivatives and the financial liabilities associated with contingent consideration arrangements . • Other financial liabilities, including bonds and loans: Other financial liabilities are measured at amortized cost mainly using the effective interest method. (iii) Derecognition Takeda derecognizes a financial liability only when the obligation specified in the contract is discharged, canceled, or expires. On derecognition of a financial liability, the difference between the carrying amount and the consideration paid or payable is recognized in profit or loss. 3) Derivatives Takeda hedges the risks arising mainly from its exposure to fluctuations in foreign currency exchange rates and interest rates using derivatives such as foreign exchange forward contracts, currency options, interest rate swaps, cross currency interest rate swaps and interest rate future. Takeda does not enter into derivative transactions for trading or speculative purposes. Derivatives are measured at FVTPL unless the derivative contracts are designated as hedging instruments. The gains and losses on derivatives that are not designed as hedging instruments are recognized in profit or loss. 4) Hedge Accounting For foreign currency exposure as a result of translation risk, Takeda designates certain non- derivatives, such as foreign currency denominated debt and certain derivatives such as foreign currency forwards, as net investment hedges of foreign operations. For foreign currency exposure due to foreign currency denominated transactions, Takeda designates certain derivatives, such as foreign currency forwards, currency options and cross currency interest rate swaps, as cash flow hedges of forecasted transactions. For interest risk exposure, Takeda designates derivatives such

- 15 - as interest and cross currency interest rate swaps and forward rate agreements, as cash flow hedges of forecasted transactions. Within the designation documentation at inception, Takeda documents the risk management objective, nature of the risk being hedged, and relationship between hedging instruments and hedged risk based on the strategy for undertaking the hedging relationships. At inception and on a quarterly basis, Takeda also assesses whether the hedging instruments are highly effective in offsetting changes in the hedged transactions or net investment. • Cash flow hedges: the effective portion of changes in the fair value of derivatives designated and qualifying as cash flow hedges is recognized in other comprehensive income. The gain or loss relating to the ineffective portion is recognized immediately in profit or loss. The cumulative gain or loss that was previously recognized in other comprehensive income is reclassified to profit or loss in the same period when the cash flows of the hedged items are recognized in profit or loss and in the same line item in the consolidated statements of profit or loss. The currency basis spread and the time value of the foreign currency options are accounted for and presented as hedging cost under other components of equity separately from cash flow hedges. • Net investment hedges in foreign operations: the gain or loss on hedging instruments in foreign operations is recognized in other comprehensive income. At the time of disposal of the foreign operations, the cumulative gain or loss recognized in other comprehensive income is reclassified to profit or loss. Hedge accounting is discontinued when the hedging instrument expires or is sold, terminated or exercised, or when the hedge no longer qualifies for hedge accounting. (4) Provisions Takeda recognizes rebates and return reserves if Takeda receives consideration from a customer and expects to refund some or all of that consideration to the customer. In addition, Takeda recognizes provisions when Takeda has present legal or constructive obligations as a result of past events, it is probable that outflows of resources embodying economic benefits will be required to settle the obligations and reliable estimates can be made of the amount of the obligations. Takeda’s provisions consist primarily of rebates and return reserves, as well as provisions for litigation and restructuring. 1) Rebates and Return Reserves Takeda has recognized a provision related mainly to sales rebates and returns for products and merchandises, including for U.S. government health programs such as the U.S. Medicaid Drug Rebate Program, the U.S. Medicare Part-D Rebate Program and the U.S. Commercial Managed Care Program. 2) Provisions for Litigation Provisions for litigation are recorded, after taking appropriate legal and other specialist advice, where an outflow of resources is considered probable and a reliable estimate can be made of the likely outcome of the dispute. For certain product liability claims, Takeda will record a provision where there is sufficient history of claims made and settlements to make a reliable estimate of the provision required to cover unasserted claims. 3) Provisions for Restructuring A restructuring provision is recorded when Takeda has a detailed formal plan for the restructuring. Takeda records the provision and associated expenses based on estimated costs associated with the plan.

- 16 - (5) Post-Employment Benefit Takeda sponsors lump-sum payments on retirement, pensions and other plans such as post- retirement medical care as post- employment benefit plans. They are classified into defined benefit plans and defined contribution plans, depending on the characteristics of the plans. 1) Defined Benefit Plans Takeda uses the projected unit credit method to determine the present value, the related current service cost, and the past service cost by each defined benefit obligation. The discount rate is determined by reference to market yields on high quality corporate bonds at the end of the reporting period. The net defined benefit liabilities (assets) in the consolidated statements of financial position are calculated by deducting the fair value of the plan assets from the present value of the defined benefit obligations. If the defined benefit plan has a surplus, the net defined benefit asset is limited to the present value of any future economic benefits available in the form of refunds from the plan or reductions in future contributions to the plan. Past service cost defined as the change in the present value of the defined benefit obligation resulting from a plan amendment or curtailment is recognized in profit or loss upon occurrence of the plan amendment or curtailment. Re-measurement of net defined benefit plans is recognized in full as other comprehensive income and transferred to retained earnings in the period in which they are recognized. 2) Defined contribution plans The costs for defined contribution plans are recognized as expenses when the employees render the related service. (6) Other Significant Accounting Policies for the Consolidated Financial Statements 1) Stated Amount All amounts shown are rounded to the nearest million JPY. 2) Consumption taxes Consumption taxes are excluded from the items in the consolidated statement of profit and loss. [Notes for Accounting Estimates and Assumptions] The items which were recorded on the consolidated financial statements as of March 31, 2021 using accounting estimates or assumptions and could have a material impact on the consolidated financial statements as of March 31, 2022 are described below. Provisions for Contractual and Statutory Rebates Payable under Commercial Healthcare Provider Contracts and U.S. State and Federal Government Health Programs 267,254 million JPY In order to estimate provisions for contractual and statutory rebates payable under Commercial healthcare provider contracts and U.S. state and Federal government health programs such as U.S. Medicaid and U.S. Medicare as well as U.S. commercial managed care programs, expected product specific assumptions are used in estimating which of Takeda’s revenue transactions will ultimately be subject to the respective programs. Therefore, the change in the product specific assumptions could have a material impact on the amount of provisions to be recorded on the consolidated financial statements as of March 31, 2022. Intangible Assets 3,909,106 million JPY Intangible assets are generally considered impaired when their balance sheet carrying amount exceeds their estimated recoverable amount. The recoverable amount is estimated for each individual asset or at the larger cash generating unit level when cash is generated in combination with other assets. The estimation on recoverable amount of intangible assets requires us to make a number of assumptions including amount and timing of projected future cash flows, behavior of competitors (launch of competing products, marketing initiatives, etc.), probability of obtaining regulatory approvals, future tax rates, terminal growth rate, and

- 17 - discount rate. Events that may result in the change in amount and timing of cash flows include IPR&D projects which are not successfully developed, fail during development, are abandoned or subject to significant delay or do not receive the relevant regulatory approvals and/or commercially marketed products whose value becomes impaired. If these events were to occur, Takeda may not realize the future cash flows that Takeda has estimated nor recover the value of the initial or subsequent R&D investments made subsequent to acquisition of the asset project. Therefore, these events could have a material impact on the amount of the intangible assets to be recorded on the consolidated financial statements as of March 31, 2022. The balance of goodwill as of March 31, 2021 was 4,033,917 million JPY. Recoverable amount of goodwill was determined on a fair value less costs to dispose basis using the market value of Takeda’s outstanding shares. Takeda's market capitalization was compared to the book value of the company’s net assets and this indicated a significant surplus as of March 31, 2021. Provision for Litigation 73,395 million JPY The factors Takeda considers in developing the provision for litigation and other contingent liability amounts include the merits and jurisdiction of the litigation, the nature and the number of other similar current and past litigation cases, the nature of the product and the current assessment of the science subject to the litigation, and the likelihood of settlement and current state of settlement discussions, if any. In addition, Takeda records a provision for product liability claims incurred, but not filed, to the extent Takeda can formulate a reasonable estimate of their costs based primarily on historical claims experience and data regarding product usage. In cases Takeda may become involved in significant legal proceedings for which it is not possible to make a reliable estimate of the expected financial effect, if any, which may result from ultimate resolution of the proceedings, no provision is recognized for such cases. The estimates of these provisions and contingent liabilities are dependent upon the outcome of litigation proceedings, investigations and possible settlement negotiations, and therefore could have a material impact on the amount of provisions to be recorded on the consolidated financial statements as of March 31, 2022. Income Taxes Payable 145,203 million JPY, Deferred Tax Assets 353,769 million JPY Takeda prepares and files the tax returns based on an interpretation of tax laws and regulations, and records estimates based on these judgments and interpretations. In the normal course of business, Takeda’s tax returns are subject to examination by various tax authorities, which may result in additional tax, interest or penalty assessment by these authorities. Inherent uncertainties exist in estimates of many uncertain tax positions due to changes in tax law resulting from legislation, regulation, and/or as concluded through the various jurisdictions’ tax court systems. When Takeda concludes that it is not probable that a tax authority will accept an uncertain tax position, Takeda recognizes the best estimate of the expenditure required to settle a tax uncertainty. The amount of unrecognized tax benefits is adjusted for changes in facts and circumstances. For example, adjustments could result from significant amendments to existing tax law, the issuance of regulations or interpretations by the tax authorities, new information obtained during a tax examination, or resolution of a tax examination. Therefore, these adjustment could have a material impact on the amount of income tax payable to be recorded on the consolidated financial statements as of March 31, 2022. Takeda also assesses deferred tax assets to determine the realizable amount at the end of each period. In assessing the recoverability of deferred tax assets, Takeda considers the scheduled reversal of taxable temporary differences, projected future taxable profits, and tax planning strategies. Future taxable profits according to profitability is estimated based on Takeda’s business plan. Therefore, the change in judgment upon determining the revenue forecast used for Takeda’s business plan could have a material impact on the amount of the deferred tax assets to be recorded on the consolidated financial statements as of March 31, 2022. Provisions for Restructuring 32,297 million JPY Takeda incurs restructuring costs associated with planned initiatives to reduce the costs and in connection with the integration of the acquisitions. Takeda’s most significant restructuring costs are severance payments and lease termination costs. Takeda establishes a provision for restructuring costs when Takeda has developed a detailed formal plan for the restructuring. The recognition of restructuring provision requires estimates including timing of

- 18 - payments and the number of individuals impacted by the restructuring. The actual restructuring costs may differ from the estimates, and therefore the difference, if any, could have a material impact on the amount of the provisions to be recorded on the consolidated financial statements as of March 31, 2022. [Notes on Consolidated Statement of Profit or Loss] 1. Other operating income Other operating income included 228,923 million JPY gain from divestitures including 139,470 million JPY gain on sale of shares and relevant assets of Takeda Consumer Healthcare Company Ltd. and other non-core assets amounting to 89,453 million JPY and 60,179 million JPY revaluation gain triggered by an update to previously recognized liabilities for pipeline compound SHP647 and certain associated rights ("SHP647") to reflect a change in expected future costs, such as program termination costs. This change was a result of the European Commission's decision in May 2020 to release Takeda's obligation to divest SHP647. 2. Other operating expenses Other operating expenses included 115,875 million JPY restructuring expenses from reductions in the workforce and consolidation of sites. The major factors of restructuring expenses were related to the Shire integration costs after the Shire Acquisition and expenses related to the business transformation in Japan. In addition, other operating expenses also included 72,940 million JPY loss from changes in the fair value of financial assets associated with contingent consideration arrangements driven by changes in assumptions related to the future sales of XIIDRA, including the impact from Novartis' withdrawal of the Marketing Authorisation Application in Europe. 3. Income tax benefit Takeda recorded an income tax benefit of 9,936 million JPY. This was mainly due to tax benefits from legal entity reorganizations, from previously unrecognized deferred tax assets and tax credits offsetting tax provision on pretax earnings and tax impacts of divestitures. [Notes on Consolidated Statement of Financial Position] 1. Accumulated depreciation on assets (including accumulated impairment losses) Property, plant and equipment 737,817 million JPY Investment property 5,608 million JPY 2. Impairment loss allowance directly deducted from trade and other receivables Trade and other receivables 8,637 million JPY 3. Contingent liabilities (1) Irish Revenue Authority assessment Shire received a tax assessment from the Irish Revenue Authority on November 28, 2018 for 398 million EUR. This assessment relates to a potential taxable gain from a 1,635 million USD break fee Shire received from AbbVie, Inc. ("AbbVie") in connection with the terminated offer to acquire Shire made by AbbVie in 2014. Takeda is currently in the appeal process with regards to this assessment as it does not believe a tax liability should arise from the break fee. In late-November / early-December 2020, a hearing took place before the Irish Tax Appeals Commission and Takeda anticipates receiving a ruling within twelve months after the hearing. (2) Litigation Takeda is involved in various legal and administrative proceedings. The most significant matters are described below.

- 19 - Takeda may become involved in significant legal proceedings for which it is not possible to make a reliable estimate of the expected financial effect, if any, which may result from ultimate resolution of the proceedings. In these cases, appropriate disclosures about such cases would be included in this note, but no provision would be made for the cases. With respect to each of the legal proceedings described below, other than those for which a provision has been made, Takeda is unable to make a reliable estimate of the expected financial effect at this stage. This is due to a number of factors, including, but not limited to, the stage of proceedings, the entitlement of parties to appeal a decision, if any, and lack of clarity as to the merits of theories of liability, the merits of Takeda’s defenses, the amount and recoverability of damages and/or governing law. The Company does not believe that information about the amount sought by the plaintiffs, if that is known, is, by itself, meaningful in every instance with respect to the outcome of those legal proceedings. Legal expenses incurred and charges related to legal claims are recorded in selling, general and administrative expenses. Provisions are recorded, after taking appropriate legal and other specialist advice, where an outflow of resources is considered probable and a reliable estimate can be made of the likely outcome of the dispute. For certain product liability claims, Takeda will record a provision where there is sufficient history of claims made and settlements to enable management to make a reliable estimate of the provision required to cover unasserted claims. The factors Takeda considers in developing a provision include the merits and jurisdiction of the litigation, the nature and the number of other similar current and past litigation, the nature of the product and the current assessment of the science subject to the litigation, and the likelihood of settlement and current state of settlement discussions, if any. As of March 31, 2021, Takeda’s aggregate provisions for legal and other disputes were 73,395 million JPY. The ultimate liability for legal claims may vary from the amounts provided and is dependent upon the outcome of litigation proceedings, investigations and possible settlement negotiations. Unless otherwise stated below, Takeda is unable to predict the outcome or duration of these matters at this time. Takeda’s position could change over time, and, therefore, there can be no assurance that any losses that result from the outcome of any legal proceedings will not exceed, by a material amount, the amount of the provisions reported in these consolidated financial statements. Matters that were previously disclosed may no longer be reported because, as a result of rulings in the case, settlements, changes in our business or other developments, in our judgment, they are no longer material to our financial condition or operating results. Product Liability and Related Claims Pre-clinical and clinical trials are conducted during the development of potential products to determine the safety and efficacy of products for use by humans following approval by regulatory bodies. Notwithstanding these efforts, when drugs and vaccines are introduced into the marketplace, unanticipated safety issues may become, or be claimed by some to be, evident. Takeda is currently a defendant in a number of product liability lawsuits related to its products. For the product liability lawsuits and related claims, other than those for which a provision has been made, Takeda is unable to make a reliable estimate of the expected financial effect at this stage. The Company’s principal pending legal and other proceedings are disclosed below. The outcomes of these proceedings are not always predictable and can be affected by various factors. For those legal and other proceedings for which it is considered at least reasonably possible that a loss has been incurred, the Company discloses the possible loss or range of possible loss in excess of the recorded loss contingency provision, if any, where such excess is both material and estimable. ACTOS Product Liability Claims First initiated in 2011, Takeda has been named as a defendant in lawsuits in U.S. federal and state courts in which plaintiffs allege to have developed bladder cancer or other injuries as a result of taking products containing type 2 diabetes treatment pioglitazone (U.S. brand name ACTOS). Eli Lilly and Company (“Lilly”), which co-promoted ACTOS in the U.S. for a period of time, also is named as a

- 20 - defendant in many of these lawsuits. Under the parties’ co- promotion agreement, Takeda agreed to defend and indemnify Lilly in the U.S. matters. Outside the U.S., lawsuits and claims have also been brought by persons claiming similar injuries. In April 2015, Takeda entered into a settlement agreement with the lead plaintiffs’ lawyers that resolved more than 95% of ACTOS product liability lawsuits pending against Takeda and Lilly in the U.S. The settlement covered all bladder cancer claims pending in any U.S. court as of the date of settlement. Claimants with unfiled claims in the U.S. represented by counsel as of the date of settlement and within three days thereafter were also eligible to participate. As part of the settlement, Takeda paid 2.4 billion USD (approximately 288 billion JPY) into a qualified settlement fund. Takeda received insurance proceeds totaling approximately 58 billion JPY under various policies covering product liability claims against Takeda. Takeda also established provisions for the remaining ACTOS claims and lawsuits. Although Takeda subsequently received additional claims from plaintiffs not resolved by the settlement, nearly all of those claims have now been resolved. These lawsuits are no longer material to Takeda. Economic Loss Cases Takeda has been named in several ACTOS-related lawsuits brought by plaintiffs that do not assert any claims for personal injuries. Instead plaintiffs claim they suffered an economic loss by paying for ACTOS prescriptions that allegedly would not have been written had Takeda provided additional information about the alleged risks of bladder cancer associated with ACTOS. A putative class of third party payors and consumers brought suit against Takeda in the U.S. District Court for the Central District of California. Discovery is ongoing in that case. A case brought by a separate group of third party payors asserting similar claims was filed in the U.S. District Court for the Southern District of New York in June 2019. Takeda’s motion to dismiss is pending in that case. The States of Mississippi and Louisiana also filed lawsuits against Takeda and Lilly alleging that defendants did not warn about bladder cancer and other risks of ACTOS. The lawsuits sought reimbursement of the cost of ACTOS, paid by the states on behalf of patients through programs such as Medicaid, and for medical treatment of patients allegedly injured by ACTOS, attorneys’ fees and expenses, and punitive damages. Takeda and Lilly entered into settlement agreements that resolved the lawsuits brought by the State of Mississippi and the State of Louisiana, in November 2018 and September 2019, respectively. Proton Pump Inhibitor (“PPI”) Product Liability Claims As of March 31, 2021, more than 6,600 product liability lawsuits related to the use of PREVACID and DEXILANT have been filed against Takeda in U.S. federal and state courts. Most of these cases are pending in U.S. federal court and are consolidated for pre-trial proceedings in a multi- district litigation in federal court in New Jersey. The plaintiffs in these cases allege they developed kidney injuries or, in some cases, gastric cancer as a result of taking PREVACID and/or DEXILANT, and that Takeda failed to adequately warn them of these potential risks. Similar cases are pending against other manufacturers of drugs in the same PPI class as Takeda’s products, including AstraZeneca plc (“AstraZeneca”), Procter & Gamble Company (“Procter & Gamble”) and Pfizer Inc. (“Pfizer”). Outside the U.S., three proposed class actions have been filed in three provinces in Canada (Quebec, Ontario, and Saskatchewan). The defendants in these actions include Takeda, AstraZeneca, Janssen Pharmaceutical Companies (“Janssen”) and several generic manufacturers. Intellectual property Intellectual property claims include challenges to the validity and enforceability of Takeda’s patents on various products or processes as well as assertions of non-infringement of those patents. A loss in any of these cases could result in loss of patent protection for the product at issue. The consequences of any such loss could be a significant decrease in sales of that product and could materially affect future results of operations for Takeda. TRINTELLIX Takeda has received notices from sixteen generic pharmaceutical companies that they have submitted ANDAs with paragraph IV certifications seeking to sell generic versions of TRINTELLIX. Takeda filed

- 21 - patent infringement lawsuits against the ANDA filers in federal court in Delaware. A first Markman hearing took place on May 29, 2019 and a claim construction ruling was issued on July 16, 2019. A second Markman hearing took place on December 18, 2019. A trial took place from January 15 to January 28, 2021. ADYNOVATE On December 5, 2016, Bayer Healthcare LLC (“Bayer”) filed a lawsuit in the U.S. District Court for the District of Delaware against Baxalta Incorporated and Baxalta US Inc. (collectively “Baxalta”), which are now subsidiaries of Takeda, and Nektar Therapeutics (“Nektar”) filed alleging infringement of U.S. Patent No. 9,364,520 in connection with the sales of ADYNOVATE [antihemophilic factor (recombinant), PEGylated]. The case was tried before a jury beginning on January 28, 2019. The jury found in favor of Bayer determining that the patent is infringed. The jury further awarded damages in the amount of 155.2 million USD. Takeda has filed an appeal with the Court of Appeals of the Federal Circuit (CAFC) in September 2019. The CAFC upheld the District Court’s decision on March 1, 2021. The Appeal Mandate was issued on April 7, 2021. Takeda established a provision against this case in purchase accounting. NINLARO Takeda received a paragraph IV notice letter from Sun Pharmaceutical Industries Limited (“Sun”) on January 17, 2020. Sun alleged that U.S. Patent numbers 7,442,830, 8,859,504, and 9,175,017 are invalid, unenforceable, and/or will not be infringed. Takeda filed a complaint against Sun in the U.S. District Court for the District of Delaware on February 27, 2020. Other In addition to the individual patent litigation cases described above, Takeda is party to a number of cases where Takeda has received notices that companies have submitted ANDAs with paragraph IV certifications to sell generic versions of other Takeda products. These include other Takeda products including Alogliptin. Takeda has filed patent infringement lawsuits against parties involved in these situations. Sales, Marketing, and Regulation Takeda has other litigations related to its products and its activities, the most significant of which are describe below. ACTOS Antitrust Case In December 2013, the first of two antitrust class action lawsuits was filed against Takeda in the U.S. District Court for the Southern District of New York by a putative class of patients who were prescribed ACTOS. The second class action was filed against Takeda in the same court in April 2015 by a putative class of wholesalers that purchased ACTOS from Takeda. In both actions, plaintiffs allege, inter alia, that Takeda improperly characterized certain patents for ACTOS in the FDA Orange Book, which they claim imposed requirements on generic companies that filed Abbreviated New Drug Applications and, in turn, resulted in delayed market entry for generic forms of ACTOS. In October 2019, the District Court denied Takeda’s motion to dismiss. Takeda subsequently sought an interlocutory appeal of the District Court’s decision which is still pending. Investigation of Patient Assistance Programs In November 2016, the U.S. Department of Justice (through the U.S. Attorneys’ Office in Boston) issued a subpoena to ARIAD, which was acquired by Takeda during the year ended March 31, 2017, seeking information from January 2010 to the present relating to ARIAD’s donations to 501(c) (3) co-payment foundations, financial assistance programs, and free drug programs available to Medicare beneficiaries and the relationship between these co-payment foundations and specialty pharmacies, hubs or case management programs. Takeda is cooperating in the investigation.

- 22 - In June 2019, the U.S. Department of Justice (through the U.S. Attorney’s Office in Boston) issued a subpoena to Shire Pharmaceuticals LLC, which was acquired by Takeda during the year ended March 31, 2019 (through Takeda’s acquisition of Shire plc). The subpoena generally seeks information about Shire’s interactions with 501(c)(3) organizations that provide financial assistance to Medicare patients taking Shire drugs, including the hereditary angioedema medications Firazyr and Cinryze. Takeda is cooperating with the investigation. Department of Justice Civil Investigative Demands On February 19, 2020, Takeda received a Civil Investigative Demand (“CID”) from the U.S. Department of Justice (through its office in Washington, DC). The CID seeks information as part of an investigation of possible off-label promotion and violations of the Anti-kickback Statute in connection with the promotion and sale of TRINTELLIX. Takeda is cooperating with the DOJ’s investigation. On February 28, 2020, Takeda received a CID from the U.S. Department of Justice (through its office in Washington, DC). The CID seeks information as part of an investigation of possible kickbacks to a Florida allergy center in connection with the promotion and sale of Takeda’s subcutaneous IG products, CUVITRU, HYQVIA and GAMMAGARD. Takeda is cooperating with the DOJ’s investigation.

- 23 - [Notes on Consolidated Statement of Changes in Equity] 1. Class and total number of shares issued as of March 31, 2021 Common Stock 1,576,388 thousand shares 2. Dividends (1) Amount of dividends paid Resolution Class of Shares Total dividends Dividends per share Record date Effective date Ordinary General Meeting of Shareholders Common Stock 141,858 million JPY 90.00 JPY March 31, 2020 June 25, 2020 (June 24, 2020) Meeting of Board of Directors Common Stock 141,860 million JPY 90.00 JPY September 30,2020 December 1, 2020 (October 29, 2020) Total 283,718 million JPY (2) Dividends declared whose record date falls in the fiscal year ended March 31, 2021 and the effective date falls in the following fiscal year Matters with respect to dividends on shares of common stock will be proposed at the Ordinary General Meeting of Shareholders to be held on June 29, 2021 as follows: (i) Total dividends 141,859 million JPY (ii) Dividends per share 90.00 JPY (iii) Record date March 31, 2021 (iv) Effective date June 30, 2021 Dividends will be paid from retained earnings. 3. Class and number of shares underlying stock acquisition rights as of March 31, 2021 (excluding rights whose exercise period has yet to begin) Common stock 2,672,000 shares [Per Share Information] 1. Equity attributable to owners of the Company per share 3,308.93 JPY 2. Basic earnings per share 240.72 JPY

- 24 - [Notes on Financial Instruments] 1. Overview of Financial Instruments Takeda promotes risk management to reduce the financial risks arising from business operations. The principal risks to which Takeda is exposed include market risk, counterparty credit risk, and liquidity risk caused by changes in the market environment such as fluctuations in foreign exchange rates, interest rates and market prices of commodities and other financial holdings. Each of these risks is managed in accordance with Takeda’s policies. (1) Market Risk Major market risks to which Takeda is exposed are 1) foreign currency risk, 2) interest rate risk and 3) price fluctuation risk. Financial instruments affected by market risk include loans and borrowings, deposits, equity investments and derivative financial instruments. 1) Foreign Currency Risk Takeda’s exposure to the risk of changes in foreign exchange rates primarily relates to its operations (when revenue or expense is denominated in a foreign currency) and Takeda’s net investments in foreign subsidiaries. Takeda manages foreign currency risks in a centralized manner using derivative financial instruments. Takeda's policy does not permit the use of speculative foreign currency financial instruments or derivatives. Takeda uses forward exchange contracts, currency swaps, and currency options to hedge individually significant foreign currency transactions. Takeda has also designated loans and bonds denominated in the US dollar and Euro and forward exchange contracts as hedging instruments of net investments in foreign operations. 2) Interest Rate Risk Takeda’s exposure to the risk of changes in benchmark interest rates and foreign exchange rate relates primarily to the outstanding debts with floating interest rates. Takeda may use interest and currency swaps that fix the amount of future payments to manage interest and foreign exchange rate risks through cash flow hedge strategies. 3) Price Fluctuation Risk Management Commodity Price Risk For its business operations, Takeda is exposed to risks from commodity price fluctuations. Takeda manages this risk primarily by utilizing fixed price contracts, but may also use financial instruments to lock in a fixed price. Market Price Risk Market pricing and valuations of Takeda’s fixed-income financial assets and liabilities are impacted by changes in currency rates, interest rates and credit spreads, which are managed as described in this Notes. For equity instruments, Takeda manages the risk of price fluctuations in the instruments by regularly reviewing share prices and financial positions of the issuers. (2) Credit Risk Takeda is exposed to credit risk from its operating activities (primarily trade receivables) and from its financing activities, including deposits with banks and financial institutions, foreign exchange transactions, and other financial instruments. The maximum exposure to credit risk, without taking into account any collateral held at the end of the reporting period, is represented by the carrying amount of the financial instruments which is exposed to credit risk on the consolidated statement of financial position. 1) Customer Credit Risk Trade and other receivables are exposed to customer credit risk. Takeda monitors the status of overdue balances, reviews outstanding balances for each customer and regularly examines the credibility of major customers in accordance with Takeda’s policies for credit management to facilitate the early evaluation and

- 25 - the reduction of potential credit risk. If necessary, Takeda obtains rights to collateral or guarantees on the receivables. 2) Other Counterparty Credit Risk Cash reserves of Takeda are concentrated mostly with Takeda and entities acting as the cash pool leader in the United States and Europe. These cash reserves are managed exclusively by investments in highly rated short-term bank deposits and bonds of highly rated issuers within the investment limits determined by reviewing the investment ratings and terms under Takeda’s policies for fund management, resulting in limited credit risk. Cash reserves, other than those subject to the group cash pooling system, are managed by each consolidated subsidiary in accordance with Takeda’s fund management policies. For derivatives, Takeda enters into contracts only with financial counterparties rated investment grade or higher in order to minimize counterparty risk. (3) Liquidity Risk The Company manages liquidity risk and establishes an adequate management framework for liquidity risk to secure stable short-, mid-, and long-term funds and sufficient liquidity for operations. Takeda manages liquidity risk by monitoring forecasted cash flows and actual cash flows. In addition, Takeda has commitment lines with some counterparty financial institutions to manage liquidity risk. Takeda strives to maximize the available liquidity with a combination of liquid short-term investments and committed credit lines with strong rated counterparties. The objective is to maintain levels in excess of project cash needs to mitigate the risk of contingencies. 2. Fair value of financial instruments The carrying amount and fair value of financial instruments at the reporting date are set forth in the table below. Financial instruments measured at fair value whose fair value is the same as their carrying amount, short- term financial assets and liabilities whose fair value is approximately the same as their carrying amount, and lease liabilities whose fair value is not required to be disclosed are not included in the table below. (Million JPY) Carrying amount Fair value Bonds 3,532,202 3,762,266 Long-term loans 1,103,100 1,098,526 Long-term debt is recognized at its carrying amount. The fair value of bonds is measured at quotes whose significant inputs to the valuation model used are based on observable market data. The fair value of loans is measured at the present value of future cash flows discounted using the applicable market rate on the loans in consideration of the credit risk by each group classified in a specified period. [Significant Subsequent Events] On April 1, 2021, Takeda provided a notice of prepayment to the lenders of the JBIC Loan in respect of 2,000 million USD of the outstanding loan amount of 3,700 million USD that has an original maturity date of December 11, 2025. The prepayment of the JBIC Loan will be made on June 11, 2021. On April 16, 2021, Takeda provided a notice of redemption to the holders of the remaining 200 million USD of unsecured U.S. dollar-denominated senior notes issued in July 2017 in advance of their original maturity date of January 18, 2022. The redemption date of the unsecured U.S. dollar-denominated senior notes will be May 17, 2021. The impact from the accelerated debt prepayments on the consolidated statements of profit or loss is not expected to be material.
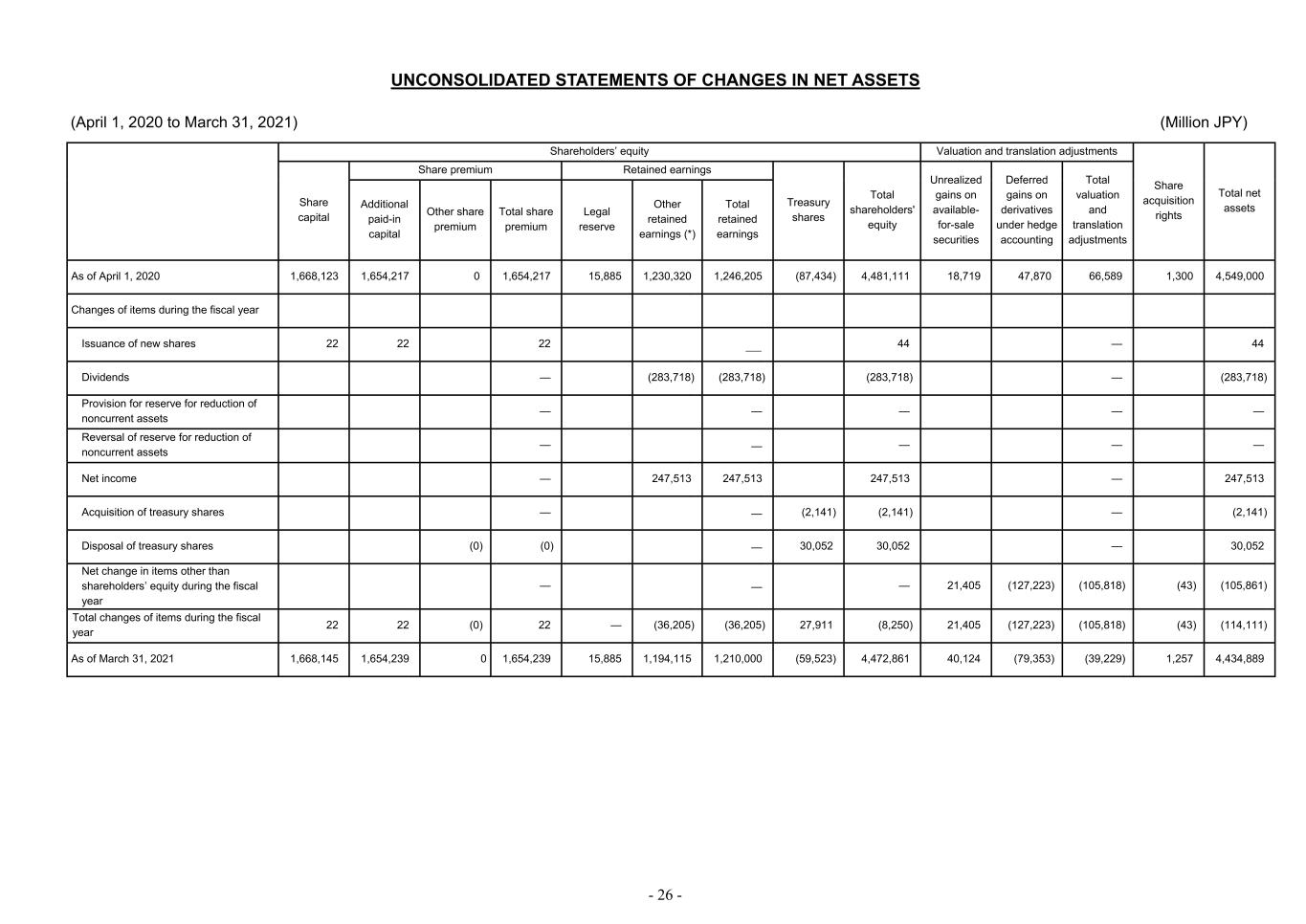
- 26 - UNCONSOLIDATED STATEMENTS OF CHANGES IN NET ASSETS (April 1, 2020 to March 31, 2021) (Million JPY) Shareholders’ equity Valuation and translation adjustments Share acquisition rights Total net assets Share capital Share premium Retained earnings Treasury shares Total shareholders' equity Unrealized gains on available- for-sale securities Deferred gains on derivatives under hedge accounting Total valuation and translation adjustments Additional paid-in capital Other share premium Total share premium Legal reserve Other retained earnings (*) Total retained earnings As of April 1, 2020 1,668,123 1,654,217 0 1,654,217 15,885 1,230,320 1,246,205 (87,434) 4,481,111 18,719 47,870 66,589 1,300 4,549,000 Changes of items during the fiscal year Issuance of new shares 22 22 22 — 44 — 44 Dividends — (283,718) (283,718) (283,718) — (283,718) Provision for reserve for reduction of noncurrent assets — — — — — Reversal of reserve for reduction of noncurrent assets — — — — — Net income — 247,513 247,513 247,513 — 247,513 Acquisition of treasury shares — — (2,141) (2,141) — (2,141) Disposal of treasury shares (0) (0) — 30,052 30,052 — 30,052 Net change in items other than shareholders’ equity during the fiscal year — — — 21,405 (127,223) (105,818) (43) (105,861) Total changes of items during the fiscal year 22 22 (0) 22 — (36,205) (36,205) 27,911 (8,250) 21,405 (127,223) (105,818) (43) (114,111) As of March 31, 2021 1,668,145 1,654,239 0 1,654,239 15,885 1,194,115 1,210,000 (59,523) 4,472,861 40,124 (79,353) (39,229) 1,257 4,434,889

- 27 - *Breakdown of other retained earnings (Million JPY) Reserve for retirement benefits Reserve for dividends Reserve for research and development Reserve for capital improvements Reserve for promotion of exports Reserve for reduction of noncurrent assets General reserve Unappropriated retained earnings Total As of April 1, 2020 5,000 11,000 2,400 1,054 434 26,659 814,500 369,273 1,230,320 Changes of items during the fiscal year Issuance of new shares — Dividends (283,718) (283,718) Provision for reserve for reduction of noncurrent assets 14,356 (14,356) — Reversal of reserve for reduction of noncurrent assets (5,942) 5,942 — Net income 247,513 247,513 Acquisition of treasury shares — Disposal of treasury shares — Net change in items other than shareholders’ equity during the fiscal year — Total changes of items during the fiscal year — — — — — 8,414 — (44,619) (36,205) As of March 31, 2021 5,000 11,000 2,400 1,054 434 35,073 814,500 324,654 1,194,115

- 28 - Notes to the Unconsolidated Financial Statements [Notes for Significant Accounting Policies] 1. Valuation of Significant Assets (1) Valuation of Securities Shares of subsidiaries and affiliates: Valued at cost using the moving-average method Available-for-sale securities With market values: Valued at market prices on the balance sheet date (Unrealized gains and losses are included in net assets, and cost of securities sold is calculated using the moving-average method.) Without market values: Valued at cost using the moving-average method (2) Valuation of Derivatives: Valued at market value (3) Valuation of Inventories Merchandise and products: Cost determined by gross average method (Balance sheet values are calculated by write-down of the book value based on decreases in profitability) Work in process: Cost determined by gross average method (Balance sheet values are calculated by write-down of the book value based on decreases in profitability) Raw materials and Supplies: Cost determined by gross average method (Balance sheet values are calculated by write-down of the book value based on decreases in profitability) 2. Depreciation Methods for Significant Noncurrent Assets (1) Tangible noncurrent assets (excluding lease assets) The Company uses the declining-balance method. However, for buildings (excluding building improvements) acquired on or after April 1, 1998, the straight-line method is applied. Estimated useful lives are mainly as follows: Buildings and structures: 15-50 years Machinery and equipment: 4-15 years (2) Intangible noncurrent assets (excluding lease assets) The Company uses the straight line depreciation method for intangible noncurrent assets. The depreciation period is based on the period of availability. (3) Lease assets The Company depreciates lease assets related to finance leases with no transfer of ownership rights over the lease term, with a nil residual value. 3. Significant Reserves (1) With respect to allowance for doubtful receivables, in order to account for potential losses from uncollectible notes and accounts receivable, the Company recognizes reserve for uncollectible receivables based on historical loss ratios. Specific claims, including doubtful claims, are individually evaluated in light of their recoverability, and the allowance for doubtful receivables is recognized at the amount deemed unrecoverable. (2) Reserve for employees’ bonuses is stated at the estimated amount of bonuses required to be paid to eligible employees at the balance sheet date based on the applicable payments period in order to cover payment of bonuses to employees.

- 29 - (3) Reserve for bonuses for directors and corporate auditors is stated as the estimated amount to be paid in order to cover payments of bonuses to directors and corporate auditors. (4) Reserve for retirement benefits is based on the present value of the projected retirement benefit obligation as of the balance sheet date estimated at the beginning of each fiscal year, less pension assets under the corporate pension plans measured at fair value in order to cover payments of retirement benefits to employees. In calculating retirement benefit obligations, the benefit formula basis is used as the method of attributing expected benefit to periods up to this fiscal year end. Prior service cost is amortized using the straight-line method over a fixed number of years (five years) within the average remaining years of service when obligations arise. Unrecognized net actuarial gains and losses are expensed from the period of occurrence in proportional amounts, on a straight-line basis over the fixed number of years (five years) within the average remaining years of service in each period when obligations arise. (5) Reserve for litigation is recorded, after taking appropriate legal and other specialist advice, where an outflow of resources is considered probable and a reliable estimate can be made for the likely outcome of the dispute. (6) Reserve for share-based payments is stated at the estimated amount of share-based obligations as of the balance sheet date mainly in order to grant the Company's share to directors and employees in accordance with the share-based payment rules. (7) Reserve for restructuring costs is reasonably estimated based on costs expected to arise from the R&D transformation and the integration with Shire. 4. Other Significant Accounting Policies for the Unconsolidated Financial Statements (1) Hedge Accounting a. Methods of hedge accounting The Company uses deferred hedging. The allocation treatment is adopted for forward exchange transactions that meet the requirements for that method and special treatment is adopted for interest rate swaps that meet the requirements for special treatment. b. Hedging instruments, hedged items and hedging policies The Company uses interest rate swaps to hedge a portion of future cash flow related to financial income or expense that is linked to short-term variable interest rates. In addition, the Company uses forward foreign exchange transactions, etc. to hedge a portion of risk of changes in future cash flow arising from changes in foreign exchanges. Foreign currency risk of the investments in foreign operations is managed through the use of foreign-currency-denominated bonds and borrowings. These hedge transactions are conducted in accordance with established policies regarding the scope of usage and standards for selection of financial institutions. c. Method of assessing effectiveness of hedges Preliminary testing is conducted using statistical methods such as regression analysis, and post-transaction testing is conducted using ratio analysis. The Company omits the assessment if material terms of the transaction are the same and also the hedging effect is extremely high. (2) Stated Amount All amounts shown are rounded to the nearest million JPY (i.e., a half of a million or more is rounded up to a full one million and less than a half of a million is disregarded). (3) Consumption taxes Consumption taxes are excluded from the items in the statement of operations. (4) Consolidated taxation system The Company has adopted the consolidated taxation system. (5) Application of Tax Effect Accounting for the Transition from the Consolidated Taxation System to the Group Tax Sharing System Regarding the transition to the Group Tax Sharing System established by "Act for Partial Revisions of the Income Tax Act, etc." (Act No.8 of 2020), the Company did not apply paragraph 44 of "Implementation Guidance on Tax Effect Accounting" (ABSJ Guidance No.28, February 16, 2018) to the items under the Standalone Tax System whose treatment was revised in line with the transition to the Group Tax Sharing System, and calculated deferred

- 30 - tax assets and deferred tax liabilities based on the tax law before the revision according to paragraph 3 of "Practical Solution on the Treatment of Tax Effect Accounting for the Transition from the Consolidated Taxation System to the Group Tax Sharing System" (Practical Issues Task Force No.39, March 31, 2020). [Notes for Changes in Presentation] (Applying the “Accounting Standard for Disclosure of Accounting Estimates”) The Company applied the “Accounting Standard for Disclosure of Accounting Estimates” (ASBJ Statement No.31, March 31, 2020) from this fiscal year and disclosed Notes for Accounting Estimated and Assumption. (Changes in Unconsolidated Balance Sheet) "Reserve for SMON compensation" (989 million JPY as of March 31, 2020), which was presented separately in the previous year, is included in "Reserve for litigation" for the fiscal year ended March 31, 2021 since the amounts became immaterial. [Notes for Accounting Estimates and Assumptions] The items which were recorded on the financial statements as of March 31, 2021 using accounting estimates or assumptions and could have a material impact on the financial statements as of March 31, 2022 are described below. Deferred Tax Assets 179,650 million JPY The Company recognized deferred tax assets of 179,650 million JPY on the balance sheet as of March 31, 2021. As discussed in the note (Accounting for Deferred Income Taxes), the amount of deferred tax assets before offsetting with the deferred tax liabilities is 229,727 million JPY, which is a net of gross deferred tax assets for deductible temporary differences and net operating loss carryforward of 1,193,704 million JPY with valuation allowances of 963,977 million JPY. These deferred tax assets are recorded to the extent that it is probable that future taxable income will be available against which the reversal of deductible temporary differences or utilization of the net operating losses carryforward will generate a tax benefit for the Company. The Company also assesses deferred tax assets to determine the realizable amount at the end of each period. In assessing the recoverability of deferred tax assets, The Company considers the scheduled reversal of taxable temporary differences, projected future taxable profits, and tax planning strategies. Future taxable profits according to profitability is estimated based on the Company's business plan. Therefore, the change in judgment upon determining the revenue forecast used for the Company’s business plan could have a material impact on the amount of the deferred tax assets to be recorded on the financial statements as of March 31, 2022.

- 31 - [Notes on Unconsolidated Balance Sheet] 1. Accumulated depreciation on assets: Tangible noncurrent assets 325,312 million JPY 2. Contingent liabilities (Guarantees) The Company has provided guarantees to the following persons/subsidiaries mainly for obligations to cover the repayment of bonds, rental fees based on the real-estate lease contracts and foreign exchange derivatives. Employees of Takeda Pharmaceutical Company Limited 23 million JPY Shire Acquisitions Investments Ireland Designated Activity Company 608,355 million JPY (USD) 5,504 million Baxalta Incorporated 170,033 million JPY (USD) 1,539 million Pharma International Insurance Designated Activity Company 50,942 million JPY (USD) 461 million Millennium Pharmaceuticals, Inc 28,036 million JPY (USD) 254 million Shire Ireland Finance Trading 12,103 million JPY (USD) 110 million Takeda UK Limited 104 million JPY (GBP) 1 million Takeda Pharma, S.A.(Argentine) 43 million JPY (ARS) 35 million Baxalta Columbia S.A.S. 56 million JPY (USD) 500 thousand (Litigation) For details of major litigation matters, please refer to [Notes on Consolidated Statement of Financial Position] 3. Contingent liabilities, (2) Litigation Product Liability and Related Claims ACTOS Proton Pump Inhibitor (“PPI”) Product Liability Claims 3. Receivables from and payables to subsidiaries and affiliates (including those separately presented) Short-term receivables: 82,341 million JPY Long-term receivables: 2,150 million JPY Short-term payables: 1,393,027 million JPY Long-term payables: 634,824 million JPY

- 32 - [Notes on Unconsolidated Statement of Operations] 1. Transactions with subsidiaries and affiliates Operating transactions: Sales 104,943 million JPY Purchases 66,906 million JPY Other 36,904 million JPY Non-operating transactions: Non-operating income 33,998 million JPY Non-operating expenses 11,855 million JPY Extraordinary income 6,779 million JPY Extraordinary loss 18,075 million JPY Sales of assets 1,651,907 million JPY Purchases of assets 1,804,901 million JPY Acquisition amount of shares in subsidiaries as a result of in- kind dividends and share exchange 4,849,028 million JPY Please refer to [Transaction with Related Parties] for the major transactions included above. 2. Research and development costs: 122,631 million JPY 3. Extraordinary income (Gain on divestment of business) The gain was from sale of shares and related assets of Takeda Consumer Healthcare Company Ltd. (Gain on sales of noncurrent assets) The gain was recognized primarily from the sale of patent rights of select over-the-counter and prescription pharmaceutical products. 4. Extraordinary loss (Restructuring costs) The loss was from restructuring expenses to build an efficient operating model, including reductions in the workforce and consolidation of sites. (Loss on restructuring of subsidiaries and affiliates) The loss was recognized primarily from restructuring of subsidiaries in relation to our group restructuring. [Notes on Unconsolidated Statement of Changes in Net Assets] 1. Class and total number of shares of treasury shares as of March 31, 2021 Common Stock 12,945 thousand shares

- 33 - [Transaction with Related Parties] Association Company Name Ownership of Voting Rights Relationship with Related Party Nature of the Transaction Transaction Amount Account Item Balance at the end of year million JPY million JPY Consolidated subsidiary Shire Acquisitions Investments Ireland Designated Activity Company Direct 100.0% Debt guarantee Debt guarantee (Note) 1 608,355 - — Receipt of in-kind dividends (Note) 3 201,673 - — Exchange loss associated with in- kind dividends (Note) 3 18,075 - — Consolidated subsidiary Baxalta Incorporated Indirect 100.0% Debt guarantee Debt guarantee (Note) 1 170,033 - — Consolidated subsidiary Shire Holdings Luxembourg S.à r.l. Direct 100.0% (Note) 4 Investment Receipt of in-kind dividends (Note) 2,4 2,715,045 - — Consolidated subsidiary Shire Holdings Ireland No.2 Limited Indirect 100.0% (Note) 5 Funding Acquisition of share of subsidiary (Note) 2,5 1,666,852 - — Consolidated subsidiary Shire Ireland Finance Trading Limited Direct 100.0% Funding Transfer of share of subsidiary (Note) 2,5 1,644,854 - — Inter-company borrowings (Note) 6 498,969 Short-term loans 496,811 Interest expenses (Note) 6 6,305 Accrued interest expenses 1,014 Consolidated subsidiary Shire Ireland Investment Limited Direct 100.0% Investment Exchange of share of subsidiary (Note) 2,7 226,554 - — Receipt of in-kind dividends (Note) 2,8 910,474 - — Consolidated subsidiary Baxalta Holding B.V. Direct 100.0% (Note) 9 Investment Receipt of in-kind dividends (Note) 2,9 795,282 - — Consolidated subsidiary Baxalta GmbH Indirect 100.0% Funding Acquisition of share of subsidiary (Note) 2,10 126,075 Short-term loans (Note) 10 129,783 Interest expenses (Note) 6 286 Accrued interest expenses 24 Consolidated subsidiary Takeda Financing GK Indirect 100.0% Funding Interest expenses (Note) 6 1,389 Long-term loans 634,824 Consolidated subsidiary Takeda Pharmaceuticals International AG Direct 100.0% Funding Inter-company borrowings (Note) 6 703,392 Short-term loans 624,917 Interest expenses (Note) 6 822 Accrued interest expenses 99

- 34 - Terms and conditions of the transactions and the policy for determining the terms and conditions (Notes) 1. Debt guarantee is guarantee for redemption of bonds. Guarantee fees are reasonably determined based on market rates. 2. The exchange values of shares of subsidiary are determined in consideration of the carrying amount of the shares of subsidiary delivered before the transaction. The acquisition values of shares of subsidiary are determined in consideration of the carrying amount of the investments received before the transaction. The acquisition values of shares of subsidiary received through a capital reduction or in-kind dividends are determined based on the amount of shareholder's equity of the company subject to reorganization. 3. This was recognized in associated with the in-kind dividends of the loan to Shire Ireland Finance Trading Limited which had been owned by Shire Acquisitions Investments Ireland Designated Activity Company as a result of the capital reduction of Shire Acquisitions Investments Ireland Designated Activity Company. The difference between the carrying amount of the shares of the subsidiary held by the Company and the loans received was recognized as an exchange loss associated with in-kind dividends. 4. This was the receipt of in-kind dividend of share of Takeda Pharmaceuticals USA Inc. which had been owned by Shire Holdings Luxembourg S.à r.l as a result of the liquidation of Shire Holdings Luxembourg S.à r.l. Since the liquidation of Shire Holdings Luxembourg S.à r.l was completed during the current fiscal year, the above transaction amount is the transaction amount for the period when the company was a related party. In addition, the ownership of voting rights and other information for the company is at the time when the company was removed from a related party. 5. This was the acquisition of 100% share of Shire Ireland Finance Trading Limited which had been wholly owned by Shire Holdings Ireland No.2 Limited and the transfer of 100% shares of Shire Holdings Ireland No.2 Limited to Shire Ireland Finance Trading Limited as a result of the merger of Shire Holdings Ireland No.2 Limited by Shire Ireland Finance Trading Limited.Since the merger of Shire Holdings Ireland No.2 Limited was completed during the current fiscal year, the above transaction amount is the transaction amount for the period when the company was a related party. In addition, the ownership of voting rights and other information for the company is at the time when the company was removed from a related party. 6. Interest rates of inter-company loans are reasonably determined in consideration of market rates and in accordance with the agreement through mutual consultation. 7. This was the acquisition of shares of Takeda Pharmaceuticals USA Inc. which had been owned by Shire Ireland Investment Limited and the transfer of share of Shire Ireland Finance Trading Limited. 8. This was the receipt of in-kind dividends of shares of Takeda Pharmaceuticals USA Inc. which had been owned by Shire Ireland Investment Limited in preparation of the liquidation of Shire Ireland Investment Limited. 9. This was the receipt of in-kind dividend of share of Takeda Pharmaceuticals International AG which had been owned by Baxalta Holding B.V. as a result of its liquidation..Since the liquidation of Baxalta Holding B.V. was completed during the current fiscal year, the above transaction amount is the transaction amount for the period when the company was a related party. In addition, the ownership of voting rights and other information for the company is at the time when the company was removed from a related party. 10. This was the acquisition of 100% share of Baxalta Recombinant S.à r.l. which had been wholly owned by Baxalta GmbH and recognition of the intercompany loan as a consideration. Since Baxalta Recombinant S.à r.l. was merged into Baxalta Manufacturing S.à r.l. in September 2020, the company hold the same amount of share of Baxalta Manufacturing S.à r.l. as of the end of this fiscal year. [Per Share Information] 1. Net assets per share 2,835.81 JPY 2. Net income per share 158.45 JPY

- 35 - [Tax Effect Accounting] 1. Major components of deferred tax assets and deferred tax liabilities: (Million JPY) (Deferred tax assets) Reserve for employees’ bonuses 5,346 Research and development costs 13,675 Inventories 9,228 Deferred hedge gains or losses on derivatives under hedge accounting 17,778 Accrued expenses 13,718 Deferred income 2,224 Reserve for retirement benefits 4,242 Reserve for restructuring costs 2,993 Excess depreciation of tangible noncurrent assets 5,148 Patent rights 14,489 Sales rights 12,724 Investment in subsidiaries and affiliates (Note)2 671,894 Securities 3,515 Net operating loss carryforward (Note)1 392,506 Other 24,224 Deferred tax assets - subtotal 1,193,704 Valuation allowance for net operating loss carryforward (Note)1 (282,940) Valuation allowance for deductible temporary difference (Note)2 (681,037) Total valuation allowance (963,977) Total deferred tax assets 229,727 (Deferred tax liabilities) Prepaid pension costs (15,805) Unrealized gain on available-for-sale securities (17,607) Reserve for reduction of noncurrent assets (15,450) Other (1,215) Total deferred tax liabilities (50,077) Net deferred tax assets 179,650 (Note) 1. As part of integration with the Shire, the subsidiaries were liquidated in order to reorganize capital in subsidiaries. As a result of this liquidation, losses from liquidation of subsidiaries were treated as a tax deductible expense, which resulted in a substantial amount of Net operating loss. Of 392,506 million JPY of Net operating loss carryforwards 109,566 million JPY was considered as recoverable based on the estimation of future taxable profit income. 2. The valuation allowance mainly related to the deductible temporary difference arose from the recognition of the stock of sub-subsidiaries as a dividend in kind at fair value for tax purposes in association with liquidation of subsidiaries in the previous fiscal year. 2. The effective income tax rate of the Company after applying of deferred tax accounting differs from the statutory tax rate for the following reasons: (%) Statutory tax rate 30.6 (Adjustments) Non-deductible tax expenses 1.6 Dividend income and other nontaxable income (84.9) Changes in valuation allowance (21.9) Unitary tax on overseas subsidiaries 68.6 Increase in unrecognized deferred tax liabilities (6.3) Japanese earnings stripping rules 6.7 Other 0.5 Effective tax rate after application of deferred tax accounting (5.1)

- 36 - [Significant Subsequent Events] On April 1, 2021, the Company provided a notice of prepayment to the lenders of the JBIC Loan in respect of 2,000 million USD of the outstanding loan amount of 3,700 million USD that has an original maturity date of December 11, 2025. The prepayment of the JBIC Loan will be made on June 11, 2021. On April 16, 2021, the Company provided a notice of redemption to the holders of the remaining 200 million USD of unsecured U.S. dollar-denominated senior notes issued in July 2017 in advance of their original maturity date of January 18, 2022. The redemption date of the unsecured U.S. dollar-denominated senior notes will be May 17, 2021. The impact from the debt prepayments on the unconsolidated statements of operations is not expected to be material.