UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| x | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE | |
| SECURITIES EXCHANGE ACT OF 1934 | ||
| For the fiscal year ended December 31, 2008 | ||
| OR | ||
| ¨ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE | |
| SECURITIES EXCHANGE ACT OF 1934 | ||
| For the transition period from _____ to __________ |
COMMISSION FILE NO. 333-143314
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC.
(Exact name of registrant as specified in its charter)
| NEVADA | 20-8051010 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
6th Floor, Suite 608, Xue Yuan International Tower,
No. 1 Zhichun Road, Haidian District, Beijing, PRC
(Address of principal executive offices)
+86 10 8231 8626
(Issuer’s telephone number, including area code)
Securities Registered Pursuant to Section 12(b) of the Act: None
Securities Registered Pursuant to Section 12(g) of the Act: None.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes o No x
Check whether the issuer is not required to file reports pursuant to Section 13 or 15(d) of the Exchange Act.
Yes ¨ No x
Check whether the issuer (1) filed all reports required to be filed by Section 13 or 15(d) of the Exchange Act during the past 12 months (or for such shorter period that the registrant was required to file such reports); and (2) has been subject to such filing requirements for the past 90 days.
Yes x No ¨
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of “accelerated filer” and “large accelerated filer” in Rule 12b-2 of the Exchange Act.
Large Accelerated Filer ¨ Accelerated Filer ¨ Non-Accelerated Filer ¨ Smaller Reporting Company x
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes ¨ No x
The aggregate market value of the 20,000,374 voting and non-voting common equity stock held by non-affiliates of the Registrant was approximately $70,001,309 the last business day of the Registrant’s most recently completed second fiscal quarter, based on the last sale price of the registrant’s common stock on the most recent date on which a trade in such stock took place prior thereto.
There were a total of 26,760,258 shares of the registrant’s Common Stock, par value $0.001 per share, outstanding as of March 13, 2009.
DOCUMENTS INCORPORATED BY REFERENCE
None.
TABLE OF CONTENTS
| PART I | |||
| ITEM 1 | Business | 3 | |
| ITEM 1A. | Risk Factors | 19 | |
| ITEM 1B. | Unresolved Staff Comments | 30 | |
| ITEM 2 | Properties | 30 | |
| ITEM 3 | Legal Proceedings | 30 | |
| ITEM 4 | Submission Of Matters to a Vote Of Security Holders | 31 | |
| PART II | |||
| ITEM 5 | Market For Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities | 31 | |
| ITEM 6 | Selected Financial Data | 31 | |
| ITEM 7 | Management’s Discussion and Analysis of Financial Condition and Results of Operations | 32 | |
| ITEM 7A. | Quantitative and Qualitative Disclosures about Market Risk | 42 | |
| ITEM 8 | Financial Statements and Supplementary Data | 43 | |
| ITEM 9 | Changes in and Disagreements With Accountants on Accounting and Financial Disclosures | 43 | |
| ITEM 9A. | Controls and Procedures | 43 | |
| ITEM 9B. | Other Information. | 44 | |
| PART III | |||
| ITEM 10 | Directors, Executive Officers and Corporate Governance | 45 | |
| ITEM 11 | Executive Compensation | 47 | |
| ITEM 12 | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters. | 51 | |
| ITEM 13 | Certain Relationships and Related Transactions, and Director Independence | 53 | |
| ITEM 14 | Principal Accounting Fees and Services | 54 | |
| PART IV | |||
| ITEM 15 | Exhibits and Financial Statement Schedules | 54 | |
| Index to Consolidated Financial Statements | 57 | ||
| Consolidated Financial Statements |
i
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This report contains forward-looking statements and information relating to Yongye Biotechnology International, Inc., that are based on the beliefs of our management as well as assumptions made by and information currently available to us. When used in this report, the words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “plan” and similar expressions, as they relate to us or our management, are intended to identify forward-looking statements. These statements reflect our current view concerning future events and are subject to risks, uncertainties and assumptions, including among many others: a general economic downturn; a downturn in the securities markets; Securities and Exchange Commission regulations which affect trading in the securities of “penny stocks,” and other risks and uncertainties. Should any of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those described in this report as anticipated, estimated or expected. Except as required by law, we assume no obligation to update any forward-looking statements publicly, or to update the reasons actual results could differ materially from those anticipated in any forward-looking statements, even if new information becomes available in the future. Important factors that may cause actual results to differ from those projected include the risk factors specified above. Notwithstanding the above, Section 27A of the Securities Act and Section 21E of the Securities Exchange Act expressly state that the safe harbor for forward-looking statements does not apply to companies that issue penny stock. Because we may from time to time be considered as an issuer of penny stock, the safe harbor for forward-looking statements may not apply to us at certain times.
All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including statements regarding new and existing products and opportunities; statements regarding market and industry segment growth and demand and acceptance of new and existing products; any projections of sales, earnings, revenue, margins or other financial items; any statements of the plans, strategies and objectives of management for future operations; any statements regarding future economic conditions or performance; uncertainties related to conducting business in China; any statements of belief or intention; any of the factors mentioned in the “Risk Factors” section of this Form 10-K; and any statements or assumptions underlying any of the foregoing. Also, forward-looking statements represent our estimates and assumptions only as of the date of this report. You should read this report and the documents that we reference in this report, or that we filed as exhibits to this report, completely and with the understanding that our actual future results may be materially different from what we expect.
Except as required by law, we assume no obligation to update any forward-looking statements publicly, or to update the reasons actual results could differ materially from those anticipated in any forward-looking statements, even if new information becomes available in the future.
USE OF CERTAIN DEFINED TERMS
Except as otherwise indicated by the context, references in this report to:
| · | “Yongye,” “we,” “us,” “YGYB,” “the Company” or “our Company” are references to Yongye Biotechnology International, Inc.; |
| · | “Yongye Nongfeng”, “Yongye Nongfeng Biotechnology”, “CJV” or “YNFB” are reference to Yongye Nongfeng Biotechnology Co., Ltd.; |
| · | “Inner Mongolia Yongye”, “YBL”, “The Predecessor Company”, or “Yongye Biotechnology, Co.” are references to Inner Mongolia Yongye Biotechnology Co., Ltd. |
| · | “China” and “PRC” are a reference to the People’s Republic of China; |
| · | “RMB” is a reference to Renminbi, the legal currency of China; |
| · | “U.S. dollar,” “$” and “US$” are a reference to the legal currency of the United States; |
| · | “SEC” is a reference to the United States Securities and Exchange Commission; |
| · | “Securities Act” is a reference to Securities Act of 1933, as amended; and |
| · | “Exchange Act” is a reference to the Securities Exchange Act of 1934, as amended; |
PART I
| ITEM 1 | Business |
Business Overview
We are engaged in the sales of fulvic acid based liquid and powder nutrient compounds for plant and animal feed used in the agriculture industry. Fulvic acid is produced by the decomposition of plant material over a period of time and acts as a transport agent, which helps cells absorb essential minerals and elements for growth. Based on industry research and government testing, we believe our proprietary technology for extracting fulvic acid creates one of the purest and most bioactive fulvic acid and thus some of the most effective plant and animal nutrients on the market in China. Directly addressing the need for greater efficiency in the agricultural sector, Yongye’s products dramatically increase crop yields and improve the health of livestock. Our contractual relationship with our predecessor company, Inner Mongolia Yongye (defined below) throughout 2008 permitted us to purchase high quality finished goods which were produced according to stringent guidelines required by our IP and proprietary processes. This allowed us to create our own source of fulvic acid and to control our manufacturing process from procurement of raw materials to final production via outsourcing contract. Thus, we were able to provide our customers with a high quality product which was controlled end to end.
Industry and Market Overview
To understand our business, it is important to understand China’s economy. China is growing from a largely semi-subsistence economy to an urban economy almost overnight. The population shift to the first and second tier cities is already well documented and now third tier cities are developing large populations. The middle class is growing and consumers are demanding better quality agricultural products and inflation is setting in across the country. The agricultural industry in China is growing tremendously to keep up with this domestic demand, but added to this is also a global demand for China’s agriculture products. This increased demand is creating much volatility in the supply chain and farmers are at the crux of both the problem and the solution for both crop and animal production. This is where we believe our products can help to fulfill market need.
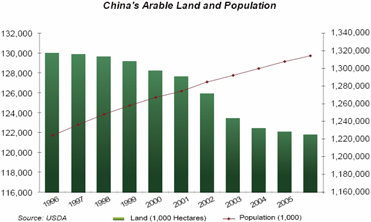
Currently, crop production in China is limited to only 1.827 billion Mu (121.8 million hectares or approximately 301 million acres) of arable farm land, which is only about 14% of all of China’s land. One principle to ensure food security is the "bottom line": 1.8 billion mu (120 million hectares) of farmland which is (0.09 hectares) per capita, about a third of the global average. In 1996, China had 1.951 billion mu (130.07 million hectares), or 1.59 mu (0.11 hectares) per person — a loss of 6.4 percent of the arable land in 11 years mainly to urbanization. China approves about 4 million mu (266,667 hectares or 658,667 acres) for construction each year which impacts about 2.82 million mu (188,000 hectares) of farmland. Currently about 70% of new construction in second and third tier cities encroaches on farmland. China's urban population was about 43.9 percent in 2006 and continues to grow with projections of 70 percent by 2050. China reported 7,438 square km of urban area in 1981 and 32,521 square km in 2005, a 340-percent increase in 25 years.
China has the world’s largest population, which it sustains on a very low amount of arable land on a per capita basis - 0.09 hectare. This is approximately 50% of that present in the United States (Source: US Census Bureau, www.census.gov). This high population density in China requires that each hectare of land feed an average of 10 people compared to the world average of 4.4 people, which means farm land is being used at close to capacity levels just for domestic production levels. Another problem is desertification. Desertification claimed 1,200 square miles of land in 2007, or 120,000 hectares. This is a major improvement from losses in the 1990’s which reached 10,000 square miles per annum. If unabated, the shortage could reach 6.67 million hectares by 2020.
3
This combination of limited arable land and a large and growing population has created a significant need to increase the output of crops per hectare in China. China’s agricultural output increased 19% from 1988 to 2004 (the total crops output was 394,080,000 tons in 1988 as compared to 469,469,000 tons in 2004). The last five years (beginning with 2004) have seen year on year increases in crop production which hasn’t been seen since economic reforms began in the late 1970s. Exports push this to maximum capacity levels, so further growth in farming capacity must come from new input technologies.
For the agricultural input industry, an increase in the use of fertilizers, approximately 8,840,000 tons in 1978 compared to approximately 46,366,000 tons used in 2004, has contributed to the growth. As a subset of the broader fertilizer market, the use of compound fertilizers in China has likewise increased, from 2,720,000 tons in 1980 to 12,040,000 in 2004. Fertilizer, however, has only a 30% impact on Chinese crops as 70% of the nutrients are lost due to poor management.
This is a common occurrence in most developing nations and has prompted the Food and Agriculture Organization of the United Nations to implement an on-going, high priority initiative to increase farmer education proper plant nutrient management. The key point is that the UN is encouraging farmers to increase nutrients to the plant without increasing the amount of fertilizer used. In case of farmer education, the FAO says that the majority of the world's farmers are females and they tend to continue cultivating in traditional ways. They need to be educated about the modern methods and the governments should take initiatives for this. Overall, this supports not only our plant nutrient approach, but also our educational approach to selling our product, which is helping farmers to increase yields via overall education and proper use of input products.
Also, with the growth of the economy has come growth in consumers’ demand for a wider choice of food options. One key area of growth is the demand for dairy products. The Chinese Government has now attached great importance to the development of this industry and it is now growing after being dormant for many years. However, average yield per cow is only about 2,000kg, indicating relatively low productivity. One major reason for this low production is mastitis, which is an inflammation of the teats which slows down milk production. This is an industry wide problem where 35-40 cows out of 100 have some form of mastitis, which is typically treated with antibiotics.
With this as a backdrop, we began selling our plant and animal nutrient products to help farmers increase their farming outputs. In crop production, our product assists farmers in generating higher yields from their crops and our first line of animal product for dairy cows assists with the reduction of mastitis to increase milk production.
Our Domestic Market
The amount of land used for agriculture in China is declining because of urban encroachment and increasing non-agricultural use of land, and a large number of farmers have moved to cities for higher paying jobs. China is going through rapid urbanization, creating pressure to use arable land for development and industrial purposes. Arable land is also being lost because of pollution (especially by heavy metals), uncontrolled erosion, overuse of chemical fertilizers and desertification. At the end of 2007, China had a total of 122 million hectares of arable farmland, which is expected to decrease to about 97 million hectares by 2015 (China National Grain and Oil Information Center).
4
As the overall economy grows, consumer demand for better quality food products is also growing. Over 60% of the nation’s population is comprised of low income, rural farmers (Asian Development Bank). The government has made raising the level of rural income, especially in Western China, a top economic and social goal. The government expects annual rural income to grow between 5% and 10% through 2010. With increased income among a large portion of the population, demand for better food products, including organic “Green Food,” is expected to grow. The need to use land efficiently has led to a genuine need to improve productivity. China’s increasingly affluent urban centers and rising concern about food quality and safety have led to greater demand for organic plant and animal nutrients.
Barely meeting domestic demand for agricultural products, food security has become a national priority in China. Agence France-Presse reported that China is self-sufficient in its ability to raise most of its staple crops, which is a part of food security, but increasingly dependent on imports of some agricultural products, such as soybeans, to meet rising domestic demand. This was after China’s agricultural output increased 19% in the period 1988 to 2004 from 394,080,000 tons to 469,469,000 tons.
The overall fertilizer market is estimated to be a $50 billion industry in China, and is estimated to grow about 30% a year from 2005 to 2009. Demand for organic plant fertilizers and nutrients is expected to grow with increasing concern over food quality and environmental issues. Overreliance on chemical fertilizers has led to soil degradation and water pollution, raising the importance of alternative means of increasing productivity. The government plans to spend approximately $169 billion, 1.6% of GDP, between 2006 and 2010 on environmental objectives. In 2007, China spent about $5.9 billion on direct subsidies for grain production and the purchase of agricultural materials, up 63% from 2006. The government is planning on additional farm subsidies, land reform initiatives, and elimination of certain agricultural taxes and is promoting the production of organically grown products by setting new standards.
Domestic competition for plant nutrients comes from companies in the traditional fertilizer industry, though the plant nutrient market does not directly compete with traditional fertilizer products. China’s fertilizer industry is highly fragmented, with over 2,800 fertilizer products registered with the government in 2007. Yongye competes against 164 other fulvic acid fertilizer products (Chinese Fertilizer Net), however, only four other similarly enhanced fulvic acid based products are truly competitors. Most of the products provided by local fertilizer companies are low quality, liquid compound fertilizers, many of which are not licensed for sale. These products do not provide plants with a full range of nutrients and international producers have higher quality offerings, but are comparatively expensive. Yongye’s animal nutrient product competes against medicines which are usually used to treat livestock after the onset of a problem. The Company’s nutrient product for dairy cows underwent testing which proved that it improved milk production and helped dairy cows avoid a number of diseases including mastitis. The use of animal nutrients promotes health and decreases the need for expensive medicines.
5
The Market for Plant Nutrients
China Market
• The overall fertilizer market is estimated to be a $50B industry in China – estimated to grow about 30% a year from 2005 to 2009.
• China has about 1.81B Mu of arable land – all cash and row crops benefit from the use of Yongye’s “Shengmingsu”
• China purchased 63% more agricultural materials in 2007 than 2006 - the market is growing rapidly
Yongye’s Market: Ten Provinces
• Yongye grew about 265% in 2008 as compared with its predecessor company in 2007 and expects to grow an additional 50% in 2009
• Our product is applied to about 2% of all arable land and we have about 37% market share
• Plant nutrient products are applied to about 6% of all arable land
Competitive Advantages
We believe that we have the following four competitive advantages:
| 1. | Unique formula for both plants and animals. Our patented plant product mixture process and patent pending animal product mixture process are for the invention of specific formulas used in these base products. We are the first company to patent such formulas in our industry and we plan to continue to improve and diversify them based on customer need. |
| 2. | Recognized and certified product offerings. We are well recognized in our markets because we work with government authorities to establish the strength of our product and company and we work with farmers in creating loyalty via our sales and support process. We also make sure our products are certified by all appropriate authorities starting with the Ministry of Agriculture which allows domestic manufacturing and sales of the product. We are also ISO 9001 certified. |
| 3. | Provide direct technical and support services to farmers who purchase products. We create strong customer loyalty by supporting farmers from product trials through initial purchase and finally into large quantity purchases. We educate farmers in yield production techniques and show them how our products are part of this process. We also show them how our product works by setting up trials in specific areas and helping them use the product throughout the season as well. |
| 4. | Cost effective extraction of fulvic acid on an industry scale. Based on internal and industry studies, our extraction process is unique in our industry in that it allows us to create a product which has been found to be more bioactive than other fulvic acid mixtures on the market. We not only create a better fulvic acid base for our products, but do it in a very cost effective manner. This allows us to create a better product at a competitive price. |
Our strategic growth plan for 2009 capitalizes on the following market conditions to build long term profitability:
| · | In October 2008 began a restructuring process to acquire the predecessor’s existing building and equipment for the 2000 tonnes per annum (TPA) facility and 120 Mu of land which will house the entire 10,000 TPA facility. This effort will not be completed until approximately September 2009. |
6
| · | Completed construction of a new 8,000 Tonnes Per Annum (TPA) processing facility in October 2008, which increased the production to 10,000 tonnes per annum. |
| · | Have employed strategic television and print advertising to support sales throughout 2008 and plan to continue this in 2009. For example, we launched an infomercial campaign on local channels to educate farmers and to help them alter usage patterns |
| · | Developing customized and enhanced plant products targeting specific crops with the intent to increase yields and market position |
| · | Increasing our line of animal product offerings to capture wide open market – revenue from these products should have the effect of reducing the seasonal sales swings in slower quarters |
| · | Building our revenue base via increased sales coverage in current provinces and increase market penetration via increased support staff coverage. |
Our Corporate History and Background
We were incorporated in the State of Nevada on December 12, 2006 under the corporate name “Golden Tan, Inc.” At that time we were engaged in the business of offering sunless tanning services and selling tanning lotions. In 2008, we began to pursue an acquisition strategy, whereby we sought to acquire an undervalued business with a history of operating revenues in markets that provide room for growth.
7
The following chart reflects our current organizational structure as of the date of this report:
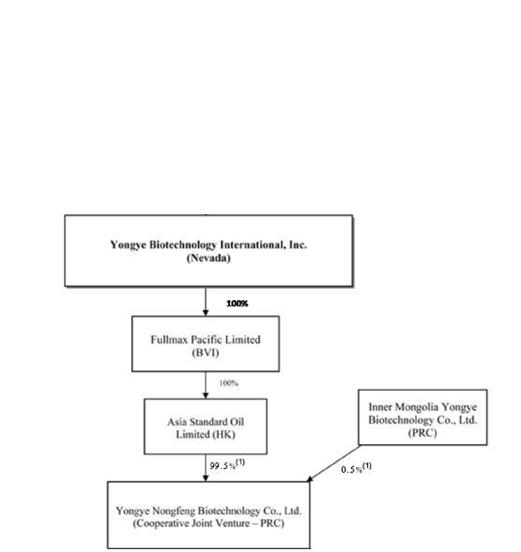
| (1) | The percentages reflected here represent the equity ownership of each entity assuming that the total amount of registered capital of the CJV, which is $21,000,000, of which $20,900,000 shall be contributed by Asia Standard Oil Limited, and $100,000 shall be contributed by Inner Mongolia Yongye. Once fully paid up, the equity agreement is such that ASO receives 99.5% ownership and Inner Mongolia Yongye receives 0.05%. |
On April 17, 2008, we entered into a Share Exchange Agreement (the “Exchange Agreement”) with Fullmax Pacific Limited, a company organized under the laws of the British Virgin Islands (“Fullmax”), the shareholders of Fullmax (the “Shareholders”), who together owned shares constituting 100% of the issued and outstanding ordinary shares of Fullmax (the “Fullmax Shares”), and our principal shareholder (the “Principal Shareholder”). Pursuant to the terms of the Exchange Agreement, the Shareholders transferred to us all of the Fullmax Shares in exchange for the issuance of 11,444,755 (the “Shares”) shares of our Common Stock (the “Share Exchange”). As a result of the Share Exchange, Fullmax became our wholly-owned subsidiary and at that time, the Shareholders acquired approximately 84.7% of our issued and outstanding Common Stock. As a result of the Share Exchange, we are now engaged in the sales of fulvic acid based plant and animal nutrients in China.
8
In November 2007, Asia Standard Oil Limited, a Hong Kong company that is the wholly owned subsidiary of Fullmax (“Asia Standard”), entered into a Sino-foreign cooperative joint venture agreement with Inner Mongolia Yongye, a PRC company that has been in the business of researching, producing and selling its own proprietary plant and animal nutrient products since 2003 (“Inner Mongolia Yongye”). Asia Standard and Inner Mongolia Yongye formed Yongye Nongfeng Biotechnology Co., Ltd., a new cooperative joint venture under PRC law (the “CJV” or “Yongye Nongfeng Biotechnology”) in January 2008. Yongye Nongfeng Biotechnology was incorporated and approved by the Inner Mongolia Department of Commerce and the Inner Mongolia Administration for Industry and Commerce on January 4, 2008.
We operate our businesses in China solely through Yongye Nongfeng Biotechnology, which is 99.5% owned by Asia Standard and 0.5% owned by Inner Mongolia Yongye. Such percentages reflect the equity ownership assuming the total amount of registered capital has been fully paid. Based upon the amount of registered capital that has been paid to date the percentages are 99.4% and 0.6%, respectively. As stipulated in the cooperative agreement between ASO and Inner Mongolia Yongye, the primary contract manufacturer of fulvic acid based products for Yongye Nongfeng Biotechnology, assigned its management team, customers, sales contracts and one of its patents to Yongye Nongfeng Biotechnology.
As part of the September Financing, we began a restructuring process which required us to purchase the land, buildings and equipment which comprised the 10,000 TPA capacity completed in October 2008. We began this process by purchasing the predecessor’s 2000TPA equipment in October 2008, but in order to complete the process we need to own the fertilizer license issued by the PRC Ministry of Agriculture. Currently, it is in the name of Inner Mongolia Yongye and we will have it issued in the name of Yongye Nongfeng Biotechnology (the “License”), and this will permit Yongye Nongfeng Biotechnology to manufacture its own fulvic acid produces. Once received, we intend to acquire the remaining land and assets related to the manufacture of fulvic acid products from Inner Mongolia Yongye (the “CJV Restructuring”).
Our Principal Products and Services
The base of our product is our own proprietary fulvic acid compound. Fulvic acid is a complex, acidic, biochemical polymer which is produced naturally by the decomposition of plant material over a long period of time. Fulvic acid binds itself to cellulose fibers and strengthens the cell walls of plants and animals and acts as a transport agent helping cells absorb the essential minerals and elements for growth. Fulvic acid usually carries 70 or more minerals and trace elements as part of its molecular complexes. These are in ideal natural form to be absorbed by plants and interact with living cells. Plants readily absorb high amounts of fulvic acid, and more readily maintain the minerals and trace elements brought in by fulvic acid. Fulvic Acid creates bioactivity in plant cells and makes them healthier.
We believe Fulvic Acid (FA) has the following key attributes when used in the agricultural industry for both plants and animals:
| · | Dissolves and then absorbs minerals into itself; |
| · | Polymer properties protect vitamins and minerals during uptake or digestion; |
| · | Contains many essential nutrients for health and growth; |
| · | Works especially well in adverse conditions; |
| · | Increases natural strength and ability of cells to fight off sickness and disease; |
| · | Scavenges free radicals and removes toxins such as heavy metals and pollutants; |
| · | Increases oxygen intake into the cells, and; |
| · | Maximizes enzyme development which results in better uptake or digestion. |
9
The principal raw material used in creating fulvic acid is Humic Acid (HA). Humic Acid is a naturally occurring humic substance. Humic acid exhibits a high cation exchange (a chemical process in which cations of like charge are exchanged equally between a solid and a solution) capacity which serves to chelate plant nutrient elements and release them as the plant requires. The chelation process holds the nutrients in the soil solution and prevents their leaching and runoff. What is more, humic acids can bind soil toxins along with plant nutrients, thereby strongly stabilizing soils. The regular use of HA organic liquid compound fertilizer enable fertilizer, insecticide, herbicide and water use to be cut by up to a half or more. This mechanism is important to environmental protection, since it prevents contamination of water sources caused by runoff.
Product Functions and Results: Plant Line
Our plant products are sold by the 100 ml bottle and in cases of 100 bottles each. The average farmer in China has a cultivated land area of 2-4 Mu and this requires about 6-12 bottles of product which is sprayed on every 15 days over a 45 day growing period. If the farmer uses our product correctly, he can decrease the use of fertilizers to normal levels and decrease overall usage of pesticides and herbicides which may reduce their overall input costs. Internal studies show that, depending upon the crop, the farmer will see increases in yields and value in the market place which should increase overall income. Each crop varies in response to the product but farmers may be expected to experience increases on par with the following results under the proper fertilizer and water conditions:
| Crop | Yield | |
| Capsicum (green pepper) | increases yield by up to 22.7%; | |
| Carrot: | increases yield by up to 26.5%; | |
| Celery: | increases yield by up to 26.3%; | |
| Cucumbers: | increases yield to 21.7%, and the leaves are greener, the plants are higher by 3.0cm, and earlier to market by 11 days; | |
| Grapes: | increases weight of individual grape 0.4g, 18.2%, increases sugar content 37.5%; | |
| Potatoes: | increases yield up to 17.3%, and the leaves are thicker and they bloom 7 to 10 days earlier; | |
| Watermelon: | increases yield by up to 16.9%, increases sugar content 0.8%-1.8%. | |
| Wheat: | increases yield up to 10.7%; |
Product Functions and Results: Animal Line
Currently, our animal product line is specifically targeted at the dairy cows, although we plan to develop products customized for other animals in the future. We believe that our animal products will help increase the capacity of the dairy supply chain by increasing the health of the dairy cows and healing their problems with mastitis. We use our base of fulvic acid and add the Chinese herbs Matrine & Oxymatrine. Matrine and Oxymatrine are non-steroidal analgesics which are anti-inflammatory in nature and are administered in treatment of mild to severe pain or treatment of inflammatory states. They also have a variety of biological activities.
In general, the financial impact for farmers from using our product has been an annual net profit increase per cow just due to a normal increase in production, and if used for treatment of mastitis, a decrease in the costs associated with the purchase of antibiotics, which also increases annual net profit. We sell our product in 300g bags which contain ten (10) 30g packets in each bag. A typical regime of use would be one cow taking 1.5g daily over a 100 day period of time.
New Products for 2009
In 2008, we did not roll out any customized products within our two product lines because the market demand for our universal product for plants and for animals was sufficient to gain market share and drive revenue. However, for 2009, we will continue to look at opportunities to develop market driven additions to our product lines as demand exists.
10
Plant Products
Currently, we use a universal product which can be applied to all types of crops, but will increase our product offerings to the following:
Corn, La Jiao Pepper, Wheat, Rice, Cucumber, Tomato, Cotton, Potato, Sunflower, Grapes, Tropical Fruits and Flowers.
When we introduce these products into the market place, we plan to charge a small percentage more than we do for the universal product, which should increase our revenue by a small amount. This will not replace our universally applied product as we will leave it in the market as well. We also expect to increase the price as demanded by the market.
Animal Product
After successful sales in our test market, we will continue to offer our Dairy Cow products, but will increase sales of the product in a targeted fashion in selected provinces. We are also working on the introduction of products for pigs, chickens and sheep.
Our Contract Manufacturing Outsourced Process
Our competitive advantage begins with our core intellectual property (“IP”) and cost effective production capability, which is attributed to our contractual relationship with Inner Mongolia Yongye. Our chief scientist has been working specifically on Inner Mongolia Yongye products for the last five years and has over 40 years of experience in the industry. This has led to two invention patents pending held by Inner Mongolia Yongye (the “Predecessor Company”) which are used in the manufacturing process. These invention patents cover the formulation and stabilization of our unique plant and animal nutrient products. Our products are approved and certified by the PRC Ministry of Agriculture.
Inner Mongolia Yongye’s production procedure is scientifically designed to ensure that our back-end product takes advantage of our front-end Intellectual Property and our vertical integration of our main raw materials provider to ensure constantly high quality product. Inner Mongolia Yongye is ISO 9001:2000 (quality control certified (July 2007), a Hohhot Industry and Commerce Bureau AAA trusted company (awarded July 2007) and a Greenfood certified (August 2008) production facility. To control the quality and the intellectual property of our products, Yongye Nongfeng has built into its joint venture contract a process to mitigate the potential loss of IP during the outsourced manufacturing process. We also inspect all raw materials shipments to Inner Mongolia Yongye and manage the arrangement of mixing parcels which are then mixed together by Inner Mongolia Yongye’s manufacturing department based on a schedule given them each day. The production facility is housed in a 2,000 sq. meter building which is adjacent to a 4,000 sq meter building used for heating and water filtration. The actual production process for Fulvic Acid is the key intellectual property component. This process, generally described, is as follows:
| · | Humic Acid is mixed with water and sodium hydroxide to form a solution. |
| · | The Humic Acid is precipitated as a solid while maintaining the solubilized Fulvic Acid in solution. |
| · | The solid Humic Acid and the solubilized Fulvic acid are separated. |
| · | The Fulvic Acid Compound is then mixed with special nutrients for its plant and animal product lines. |
| · | The animal product is turned into a powder. |
11
| · | Other customization is completed as required by customers |
Our products are packaged in bottles, bags and boxes. Each type of packaging material, along with packaging labels, is purchased from three to four manufacturers. These materials are readily available in the market. The products are then assembled and packaged in Inner Mongolia and shipped to distributors and retailers.
Manufacturing Outsourcing Contract (10,000 Tonnes Per Annum Capacity)
Currently, Yongye Nongfeng Biotechnology has an outsourcing contract with Inner Mongolia Yongye for the production of our finished nutrient product. From January until September, the manufacturer was running at 2,000TPA capacity, but after constructing a new 8,000TPA facility, capacity was increased to 10,000TPA. All employees have transferred to Yongye Nongfeng except those in the manufacturing and Research and Development departments and they will be transferred at a later date as part of the restructuring process. Because we are a small to medium enterprise and are located in an economic development zone, we have favorable tax treatment. The contract between the two companies has the following stipulations:
| · | Yongye Nongfeng Biotechnology has negotiated a flat fee arrangement with Inner Mongolia Yongye of RMB 350 per unit for our plant product and RMB 120 per unit for our animal product. The term for this agreement is five (5) years with quarterly options to renew based on general prevailing conditions at the time. |
| · | Yongye Nongfeng Biotechnology will work towards purchasing the existing site and/or expanding to new production lines in the future. We will also work towards building other equipment manufacturer (“OEM”) relationships with other manufacturers in a way which will give us avenues for additional capacity while also protecting our IP. |
| · | Yongye Nongfeng Biotechnology will have the option to purchase all the equipment, facilities and land use right of Inner Mongolia Yongye during the first two (2) years of the agreement, at the minimum purchase price permitted by the Chinese government or a book price. |
| · | The amount of rent to be paid during the term of the agreement depends on the amount of space used by Yongye Nongfeng Biotechnology, with the fee equaling RMB 2 per square meter per day. |
| · | We have been granted favorable tax benefits by the local tax authority because we are designated a high technology company and are located in a economic development zone in Hohhot city. This applies to the tax rate we incur for revenue, profit and VAT and will remain in place until 2010. |
Our Marketing and Sales Support
In conjunction with our distributors, we have established verbal agreements with 1,125 independently owned agricultural stores to operate in our branded store network. By June 30, 2008, we had 775 of these stores in our network and by December 31, 2008, had added an additional 350 of these independent stores to our network on a trial basis. These stores are dedicated to selling our product in their market location. Our sales staff is trained to work with our branded stores, and distributor network to ensure that our customers receive the right product and after-sales support. We overlay this sales and support network on top of our store and distributor network in a way that our sales and support staff are project managers who oversee three to ten stores each depending upon their region and capability level. They in turn hire contractors to assist them in various areas to ensure enough coverage exists in each area.
Our staff shares its knowledge base by walking through farming communities, organizing training courses, inviting local agricultural experts and university professors to speak on proper agricultural techniques as well as the use of our product. The Predecessor Company ended 2007 with marketing and sales staff of 91, which included temporary staff, and at December 31, 2008 had 65 full time staff. We expect that we will grow in 2009 to meet demand and support the sales of the product to our distributors. Our management in Beijing works with this staff to coordinate all marketing and sales activities.
12
In the past we have grown via market trials and word of mouth, but in 2008 we introduced many larger market media programs. We work with our independent distributors to coordinate television advertisements on local channels and arrange other mass media events. We will continue to use conferences and seminars, newspaper ads and pamphlets to get customer recognition and product branding. Our staff emphasizes the technological components of our products to help end users understand the differences in products available and how to use them. Word-of-mouth advertising and sample trials of new products in new areas are essential.
One new strategy will include an infomercial campaign to promote and educate farmers on benefits of Yongye’s nutrient products and provide in-store training for farmers on the use of the products. In this way, we hope to increase the predictability of operational and sales performance for both the franchisee and the farmer.
Our Distribution & Sales Network
The marketing and distribution of the product is a key element of our growth strategy. Our goals are to control our distribution channels, penetrate our target markets and retain our customers. Currently we are implementing a three pronged approach to accomplish this: Corporate Direct Sales, Community-Direct sales and Distributor Network sales. We have observed successful distribution models in our industry and other industries such as the Pharmaceutical and IT industries and we have take elements of them all to create a very unique approach among agriculture companies. These are described below for both our plant and animal product lines.
Corporate Direct Sales
Though a much smaller part of our business, Corporate Direct Sales allows us to sell product directly to large farms at a fixed price. In our plant line, this occurs most frequently in Xinjiang Province where most of our customers are larger farm owners. In our animal line, this includes large dairies primarily in Inner Mongolia and Xinjiang province.
Community Direct Sales
This is done through our “community-direct” footprint model which creates a “branded” feel in an independently owned agricultural store where our products are on center stage and prominently displayed in each store. This creates a local feel for a national product in the minds of the store owners and the end users also. We expect to grow the stores at a faster rate than we could implement corporate stores in an organic fashion. The owners will in turn receive a proven system of resources, tools, training and local promotion in getting their products into the market. Here is a list of stores by province for the last two years.
| Provinces | Year End 2008 | Year End 2007 | ||||||
| Hebei Subtotals | 452 | 36 | ||||||
| Hubei Subtotals | 235 | 23 | ||||||
| Xingjiang Subtotals | 145 | 100 | ||||||
| Inner Mongolia Subtotals | 51 | 25 | ||||||
| Shandong Subtotals | 133 | 16 | ||||||
| Other Subtotals | 109 | 0 | ||||||
| Grand Total | 1125 | 200 | ||||||
13
Distributor Network
Our Distributor Network channel is comprised of agents who take on our product and sell it through a chain of agents whose terminal sales point is a branded store. Our top 5 distributors accounted for approximately $44,109,813 (92%) of our revenue and each province more than one active distributor. When we take on distributors, we set up mutually agreed upon sales target agreements which enable us to become contract manufacturers for them once they order product. In these agreements, we do not allow distributors to return product once shipped. The targeted sales price per unit of plant product at each level of distribution is as follows:
| Province | $ | 107 | ||
| City | $ | 112 | ||
| County | $ | 126 | ||
| Administrative District | $ | 156 | ||
| Village | $ | 174 |
Plant Products
For the year 2008, our 3 largest sales areas are Hebei at $18,986,572 (42%), Xinjiang at $13,177,694 (29%) and Gansu at $5,663,011 (13%).These three provinces represented 84% of all plant product sales.
Animal Products
For the year 2008, our largest three sales areas are Hebei at $1,554,695 (48%), Inner Mongolia at $1,261,462 (39%) and Shandong at $430,895 (13%). This represented 100% of all of our animal product sales. We anticipate the sale of our animal product to grow in revenue, but when compared to our overall growth in plant products, it will continue to be a small percentage of revenue .
Raw Materials and Our Principal Suppliers
The humic acid our contract manufacturer, Inner Mongolia Yongye, uses comes from lignite coal which is mined in Inner Mongolia and it can be purchased for approximately USD $273 per metric ton. Humic Acid is mined from lignite or Leonardite coal. Leonardite is defined as highly oxidized low grade lignite that contains a relatively high concentration of the smaller molecular units (fulvic acids (FA)). China has approximately 12% of the world’s lignite reserves according to the survey of energy resources published by the World Energy Council.
Inner Mongolia Yongye is able to produce its high quality fulvic acid base product by controlling the input of humic acid from its direct, contracted suppliers. Currently, they have four principal suppliers which are all in Hohhot City: Heng Ya Trading Company, Bo Yi Ze Trading Company, Feng Li Trading Company and Sinochem. Their main supplier has dedicated one production line to us and has based their production design on our specific technical requirements. This line produces much of the humic acid we need, but only constitutes about 40% of their capacity. The other suppliers take up slack in supply when needed.
In addition to humic acid, we also utilize up to 18 different components in our production process, all of which can be readily obtained from numerous sources in local markets and require no special purchase requirements.
Competition
The Chinese fertilizer industry is highly fragmented. By 2007, there were over 2,000 fertilizer products in the government’s registry. We compete more specifically with producers of fulvic acid products and there are 164 of these in the registry (Source: Chinese Fertilizer Net). Of these 164, only four other products are similar to ours in the type of raw materials added. The top three producers of these products based on revenue generated in 2007 were:
14
| 1. | Dry Dragon – USD $16M. Based in Xinjiang and in business for 15 years. |
| 2. | Penshibao – USD $13.5M. Based in Guangxi. Founded in 1985. |
| 3. | Inner Mongolia Yongye – USD $13.1M. |
Competitive Advantages
We believe that we have the following four competitive advantages:
| 1. | Unique formula for both plants and animals. Our pending patents, as listed below, are for the invention of the specific formulas for our base plant and animal products. We will continue to improve and diversify them based on customer need. |
| 2. | Recognized and certified product offerings. We are well recognized in our markets because we work with government authorities to establish the strength of our product and company and we work with farmers in creating loyalty via our sales and support process. This leads to high recognition. We also make sure our products are certified in all the appropriate ways. |
| 3. | Provide direct technical and support services to farmers who purchase products. We create strong customer loyalty by supporting farmers from product trials through initial purchase and finally into large quantity purchases. We will educate farmers in yield production techniques and how our products are part of this process. We will then show them how our product works and even set up trials in specific areas. We will then help them use the product throughout the season as well. |
| 4. | Cost effective extraction of fulvic acid on an industry scale. Based on internal and industry studies, our extraction process is unique in our industry and we not only create a better fulvic acid base for our products, but do it in a very cost effective manner. This allows us to create a better product at a competitive price. |
Employees
The past few years have seen tremendous growth in the company and our employee base has also scaled with the business model. In focusing on our distribution base rather than on direct sales to farmers, we have decreased the number of temporary employees which are reflected in the 2007 numbers, and have hired full time sales professionals who work directly with distributors and branded stores. Here are our 2007 and 2008 numbers broken out between Yongye Nongfeng Biotechnology Co., Ltd. (YNFB), and its contracted manufacturing company Inner Mongolia Yongye (YBL) :
| Category | 2007 | 2008 | ||||||
| YBL | YNFB | |||||||
| Admin | 31 | 88 | ||||||
| Manufacturing | 60 | 0 | ||||||
| Research & Development | 15 | 0 | ||||||
| Sales & Support | 91 | 65 | ||||||
| Total | 197 | 153 | ||||||
As of December 31, 2008, all employees, excluding manufacturing staff and R&D staff, have signed contracts with Yongye Nongfeng Biotechnology Company, Ltd. and work exclusively for us. The manufacturing and R&D staff will be transferred over in 2009 as part of the restructuring process. None of our employees are under collective bargaining agreements. We believe that we maintain a satisfactory working relationship with our employees and we have not experienced any significant labor disputes or any difficulty in recruiting staff for our operations.
15
Research and Development
The product development life cycle is an important part of the way we do business. We bring new products to market in the following way: market research, funding approval, R&D on product, trials, approvals, model for marketing and market entry. The typical process may take up to three years depending upon the governmental approval process. Currently, the R&D function is still under transition from the predecessor company to Yongye Nongfeng, but should be fully transferred in 2009.
Intellectual Property
Inner Mongolia Yongye has carried on independent research for many years in the area of biochemistry including humic acid and fulvic acid research, development and industrialization. This research has produced the intellectual property we currently use. Inner Mongolia Yongye filed two invention patent applications with the State of Intellectual Property Office of the PRC with the application numbers 200610131953.7 and 200510118240.2. Currently, one of the two patent applications has been granted (plant patent) and is in the name of the CJV while the other one is still are pending (animal patent). When issued, it will be issued in the name of Yongye Nongfeng Biotechnology Co., Ltd. We also filed two trademark registration applications with the Trademark Bureau of the State Administration of Industry and Commerce of the PRC.
Our invention patents cover the mixture of both the base formulas for the plant and animal nutrient products and we will work with our exclusive contract manufacturers to ensure that this mixture process is consistently carried out while also protecting our Intellectual Property. The Inner Mongolia Science & Technology Department has tested and compared our fulvic acid product with other fulvic acid products and found that it has a lighter weight and higher bio-activity than the other products it tested. Our extraction process for fulvic acid remains a trade secret and is protected by a non-compete contract with Professor Gao Jing.
In addition to trademark and patent protection law in China, we also rely on contractual confidentiality and non-compete provisions to protect our intellectual property rights and brand. We also take the further steps of limiting the number of people involved in the production process and, when taking in raw materials and preparing them for mixture, we refer to each ingredient by a number rather than its name.
Governmental Regulation
Our products and services are subject to regulation by central and provincial governmental agencies in the PRC. Business and company registrations, along with the products, are certified on a regular basis and must be in compliance with the laws and regulations of the PRC and provincial and local governments and industry agencies, which are controlled and monitored through the issuance of licenses. Our licenses include:
Operating license
Our operating license enables us to undertake research and development, sales and services of humic acid liquid fertilizer, sales of pesticides, and export and import of products, technology and equipment. The registration No. is 150000400000679, and it is valid between January 4, 2008 and May 7, 2009. Once the term has expired, the license is renewable.
Green Food Certified
All of our fertilizer products are certified by the PRC government as green products for growing Grade AA “Green” foods which means they contain little or no chemical materials and can be used to grow organic foods. This is given by the China Green Food Research Center which has been researching organic food issues since 1992 and is part of the PRC Ministry of Agriculture. Our certificate is valid from August 2007 to August 2010 and requires an annual inspection which we passed in 2008.
16
Fertilizer Registration
Fertilizer registration is required for the production of liquid fertilizer and issued by the Ministry of Agriculture of the PRC. Our registration number is Agriculture Fertilizer No. 2630 (2008).
Financing Transactions During 2008
During the fiscal year we consummated two financing transactions in which we issued 12,568,625 shares of our Common Stock and warrants to purchase up to 4,399,019 at an exercise price of $1.848 for aggregate gross proceeds of approximately $20 million. All of the shares of Common Stock and underlying the warrants were registered for resale on a Registration Statement on Form S-1, which was declared effective on September 11, 2008.
April Private Placement
Concurrent with the Share Exchange, in April 2008 we completed a private placement for aggregate gross proceeds equal to $10,000,655, in which we sold 6,495,619 shares of our Common Stock and issued warrants to purchase up to 2,273,467 shares of Common Stock at an exercise price of $1.848 per share (the “April Offering”).
April “Make Good” Escrow Agreement
In connection with the April Offering, we entered into an escrow agreement with ROTH Capital Partners, LLC, (the “Placement Agent”), a representative of the Investors, Tri-State Title & Escrow LLC, as escrow agent, and Full Alliance International Limited, one of the Shareholders, pursuant to which 2,000,000 of the Shares issued to Full Alliance in the Share Exchange were delivered into escrow to be held as security for the achievement of $10,263,919 in net income for the year ended December 31, 2008 (the “2008 Net Income Threshold”). If we achieve the 2008 Net Income Threshold, the Escrow Shares will be released back to Full Alliance. If the 2008 Net Income Threshold is not achieved, the Escrow Shares will be distributed pro-rata to the investors in the April Offering.
April Lock-Up Agreement
In connection with April Offering, Full Alliance became a party to a Lock-Up Agreement with Roth Capital Partners, LLC (the “Placement Agent”) dated on April 17, 2008, under which the Director of Full Alliance agreed with the Placement Agent, without the prior written consent of the Placement Agent, not to sell any shares of our common stock or any securities convertible into or exercisable for our common stock, or enter into any swap or other agreement that transfers any of ownership of our common stock, during the period commencing on the closing date of the April Offering and expiring on the first anniversary of the date that is the effective date of the registration statement covering the resale of all the securities in connection with the April Offering (the “Lock-up Period”).
September Private Placement
In September 2008, we completed a private placement for aggregate gross proceeds equal to $9,350,000 with Qualified Institutional Buyers, in which we sold 6,073,006 shares of our Common Stock and issued warrant to purchase up to 2,125,552 shares of Common Stock at an exercise price of $1.848 per share (the “September Warrants”).
17
September “Make Good” Escrow Agreement
In connection with the September Offering, we entered into a second escrow agreement with Roth, Tri-State Title & Escrow LLC and Full Alliance, pursuant to which 4,000,000 of the Shares issued to Full Alliance in the Share Exchange (the “September Escrow Shares”) were delivered into escrow. Of the September Escrow Shares, 2,000,000 shares (the “Make Good Shares”) are being held as security for the achievement of (i) the 2008 Net Income Threshold, and (ii) fully diluted earnings per share reported in the 2008 Annual Report on Form 10-K filed with the SEC (the “2008 Annual Report”), of no less than $0.42 (the “2008 Guaranteed EPS”). If we achieve the 2008 Net Income Threshold and the 2008 Guaranteed EPS, the Make Good Shares will remain in escrow as security for the achievement of certain net income and fully diluted earnings per share targets for the year ending December 31, 2009. If the 2008 Annual Report is not filed timely with the SEC and remains unfiled for a period in excess of 45 days after the date due (taking into account the relief permitted under Rule 12(b)-25 of the Securities Exchange Act of 1934, as amended), then the 2008 Net Income Threshold will be deemed not to have been met and all of the Make Good Shares shall be distributed on a pro rata basis to the investors in the September Offering.
If prior to the second anniversary of the filing of the 2008 Annual Report, we report or recognize, or our auditors’ report or recognize that the financial statements contained in the 2008 Annual Report require an amendment or a restatement such that the Company would recognize or report adjusted 2008 After Tax Net Income (“ATNI”) of less than the 2008 Net Income Threshold or adjusted earnings per share less than the 2008 Guaranteed EPS, then notwithstanding the retention of the Make Good Shares in the escrow, or any prior return of Make Good Shares to Full Alliance, Full Alliance will, within 10 business days following the earlier of the filing of such amendment or restatement or recognition, deliver the Make Good Shares to the investors in the September Offering. “2008 After Tax Net Income” means our operating income after taxes for the fiscal year ended December 31, 2008, determined in accordance with GAAP as reported in the 2008 Annual Report.
If the Make Good Shares are retained in the escrow after we achieve the 2008 Net Income Threshold and 2008 Guaranteed EPS, we are then subject to an additional escrow for one year with respect to our 2009 operations. . In the event that (i) the 2009 After Tax Net Income equals or exceeds $12,649,248 and is less than $15,811,560, or (ii) the fully diluted earnings per share reported in the 2009 Annual Report on Form 10-K filed with the SEC (the “2009 Annual Report”), equals or exceeds $0.42 and is less than $0.53, then Make Good Shares equal to the product of (i)(A) $15,811,560 minus the 2009 After Tax Net Income, divided by (B) $15,811,560, and (ii) the Make Good Shares, shall be distributed on a pro-rata basis to the investors in the September Offering and the remaining Make Good Shares shall be returned to Full Alliance. In the event that (i) the 2009 After Tax Net Income is less than $12,649,248 or (ii) the fully diluted earnings per share reported in the 2009 Annual Report is less than $0.42, then all of the Make Good Shares shall be distributed on a pro-rata basis to the investors in the September Offering. In the event that the (i) 2009 After Tax Net Income is equal to or greater than $15,811,560 and (ii) the fully diluted earnings per share reported in the 2009 Annual Report is equal to or greater than $0.53, then all of the Make Good Shares shall be returned to Full Alliance. If the 2009 Annual Report is not filed timely with the SEC and remains unfiled for a period in excess of days after the date due (taking into account the relief permitted under Rule 12(b)-25 of the Securities Exchange Act of 1934, as amended), then the 2009 After Tax Net Income shall be deemed to be less $12,649,248, and all of the Make Good Shares shall be distributed on a pro-rata basis to the investors in the September Offering. If prior to the second anniversary of the filing of the 2009 Annual Report, we report or recognize, or our auditors’ report or recognize that the financial statements contained in the 2009 Annual Report require an amendment or restatement such that the Company would recognize or report adjusted 2009 After Tax Net Income of less than $12,649,248 or adjusted earnings per share less than $0.42, then notwithstanding any prior return of the Make Good Shares, or any portion thereof, to Full Alliance, Full Alliance will, within 10 business days following the earlier of the filing of such amendment or restatement or recognition, deliver the relevant Make Good Shares to the investors in the September Offering; provided, however, that if any portion of the Make Good Shares have been previously distributed, Full Alliance shall only be responsible for transferring such number of Make Good Shares up to the number of Make Good Shares that were previously returned to Full Alliance. In no event shall the Full Alliance be responsible for transferring any number of Make Good Shares in excess of what has been previously returned to Full Alliance. “2009 After Tax Net Income” shall mean the Company’s operating income after taxes for the fiscal year ending December 31, 2009, determined in accordance with GAAP as reported in the 2009 Annual Report.
18
Yongye Nongfeng Biotechnology Restructuring Escrow Agreement
2,000,000 shares of the September Escrow Shares (the “Restructuring Make Good Shares”) are being held as security for the timely issuance by the relevant governmental authority of a fertilizer license to the CJV and completion of the restructuring of Yongye Nongfeng Biotechnology (the “CJV Restructuring”). In the event that (1) the license has not been issued to Yongye Nongfeng Biotechnology by June 30, 2009, or such later date as agreed to by us and the investors in the September Offering holding a majority of the shares issued in the September Offering at such time (the “License Grant Date”), or (2) the license has been issued by the License Grant Date (the “Restructuring Completion Date”), but the CJV Restructuring is not completed by the date that is 132 calendar days after the License Grant Date, the Restructuring Make Good Shares shall be distributed on a pro-rata basis to the investors in the September Offering. If the license is issued by the License Grant Date and the CJV Restructuring is completed by the Restructuring Completion Date, the Restructuring Make Good Shares shall be returned to Full Alliance.
Executive Office
Our principal executive offices are located on the 6th Floor, Suite 608, at Xue Yuan International Tower, No. 1 Zhichun Road, Haidian District, Beijing, PRC. Our telephone number at that address is +82-10-8231-8626. Our corporate website is www.yongyebiotech.com. Information contained on or accessed through our website is not intended to constitute and shall not be deemed to constitute part of this Form 10-K.
| ITEM 1A. | Risk Factors |
An investment in our Common Stock is speculative and involves a high degree of risk and uncertainty. You should carefully consider the risks described below, together with the other information contained in this prospectus, including the consolidated financial statements and notes thereto, before deciding to invest in our Common Stock. The risks described below are not the only ones facing our Company. Additional risks not presently known to us or that we presently consider immaterial may also adversely affect our Company. If any of the following risks occur, our business, financial condition and results of operations and the value of our Common Stock could be materially and adversely affected.
Risks Related to Our Business
The CJV is still in the process of transitioning its business operations from our predecessor company.
We established the Cooperative Joint Venture, Yongye Nongfeng Biotechnology, on January 4th, 2008, with the intention and ultimate goal of carrying out the business of marketing and distributing our fulvic acid plant and animal nutrient products. We have transitioned all personnel, services, and control issues for the distribution and sales operations from our predecessor company Inner Mongolia Yongye (which has a 0.5/% ownership interest in the Cooperative Joint Venture and is under the control of Mr. Zishen Wu), to the Cooperative Joint Venture (Mr. Wu is also the CEO of the Cooperative Joint Venture). All personnel, services, and control issues relating to the manufacturing operations - from procurement of raw materials to final production, still reside with Inner Mongolia Yongye, our primary contract manufacturing company for acquiring finished goods. However, under the current restructuring plan, we will begin to transition the manufacturing entity to the CJV over a period of approximately 14 months which begins upon the date that the fertilizer license issued by the Ministry of Agriculture is transferred into the name of the CJV.
We have complied with all of the stipulations in the supplemental agreement related to the transition of the business. The IP related transfer for the plant product has been officially completed with the patent being granted to the predecessor company and then transferred into the name of the CJV. However, the patent for the animal product has not been granted though the transfer agent has approved the transfer of the patent into the name of the CJV once it has been granted.
To the extent that the current corporate structure is ineffective in facilitating our business operations as contemplated, we may decide to unwind or modify the current Cooperative Joint Venture in favor of a more efficient corporate structure, which may include formation of a wholly foreign owned entity. This may be accomplished without seeking approval from investors in the financing.
19
Currently, all of the distributorship agreements are in the name of Inner Mongolia Yongye, while these agreements have been transferred to Yongye Nongfeng, there are no formal agreements between Yongye Nongfeng and the branded stores. The limited operating history and the early stage of development of the Cooperative Joint Venture make it difficult to evaluate its business and future prospects. Although the “Predecessor” company, Inner Mongolia Yongye’s revenues have risen quickly and has transferred the same agreements to the CJV, we cannot assure you that the Cooperative Joint Venture will continue to maintain such profitability or that it will not incur net losses in the future. We expect that our operating expenses will increase as we expand. Any significant failure to realize anticipated revenue growth could result in operating losses.
Our reliance upon our contract manufacturer for finished goods may hinder our ability to be profitable.
We are dependent upon our relationship with our contract manufacturer, Inner Mongolia Yongye, which provides us with 100% of their production of goods. We have the opportunity to purchase from other suppliers, but Inner Mongolia Yongye are required to sell us 100% of their production and they have supplied us with approximately 100% of our finished goods in 2008. Should they be unable to procure sufficient amounts of their raw materials, they may be unable to meet all of our demand. Or, if they have production restrictions and cannot perform their obligations as agreed, we may be unable to specifically enforce our agreements and will need to find other suppliers. If they are unable to obtain adequate quantities of humid acid at economically viable prices, our business could be unprofitable and investors may lose their entire investment in us.
Inner Mongolia Yongye’s reliance upon third party suppliers for raw materials may hinder our ability to be profitable.
Inner Mongolia Yongye is dependent upon its relationships with third parties for its supply of humic acid. Inner Mongolia Yongye has three major suppliers of humic acid, which provided approximately 100% of its feedstock in 2007, and 100% in 2008. Should any of these suppliers terminate their supply relationships, Inner Mongolia Yongye may be unable to procure sufficient amounts of humic acid to meet its obligations under its manufacturing contract with us and our profitability may be limited. In addition, these suppliers may not perform their obligations as agreed, and it may not be possible to specifically enforce the related agreements. If Inner Mongolia Yongye is unable to obtain adequate quantities of humid acid at economically viable prices, our business could become unprofitable and investors may lose their entire investment in us.
Adverse weather conditions could reduce demand for fertilizer products.
The demand for our nutrient products fluctuates significantly with weather conditions, which may delay the application of the fertilizer or render it unnecessary at all. If any natural disasters, such as flood, drought, hail, tornado or earthquake, occur, demands for our products will be reduced.
Our business will suffer if Inner Mongolia Yongye loses its land use rights.
There is no private ownership of land in China and all land ownership is held by the government of the PRC, its agencies and collectives. Land use rights can be obtained from the government for a period up to 70 years, and are typically renewable. Land use rights can be transferred upon approval by the land administrative authorities of the PRC (State Land Administration Bureau) upon payment of the required land transfer fee. Inner Mongolia Yongye has received the necessary land use right certificate for its primary operating facilities, but we can give no assurance that these land use rights will be renewed on favorable terms or renewed at all. If Inner Mongolia Yongye loses its land right certificates we may lose access to production facilities that may be difficult or impossible to replace. Should we have to relocate, our workforce may be unable or unwilling to work in the new location and our operations will be disrupted during the relocation. The relocation or loss of facilities could cause us to lose sales and/or increase our costs of production, which will negatively impact our financial results.
Our business will be harmed if our major distributors reduce their orders or discontinue doing business with us.
For the year ended December 31, 2008, we sold our products primarily through 5 major distributors in our top 4 provinces. These five major customers accounted for 92% (and one major customer accounted for 43%) of our net revenue for the year ended December 31, 2008. These five major customers accounted for 82% (and the same one major customer accounted for 29%) of the predecessor’s net revenue for the year ended December 31, 2007 Although we believe that our relationship with these distributors is good, we have no long term supply agreements with them and any or all of them could termination their relationship with us in favor of competitors with increased productions capabilities or offering lower prices or other favorable terms. If some or all of these distributors reduce their orders or discontinue doing business with us, we could have difficulties finding new distributors to distribute our products and our revenues and net income could in turn decline considerably. Our reliance on these major distributors could also affect our bargaining power in getting favorable prices for our products. In addition, untimely payment and/or failure to pay by these major distributors would negatively affect our cash flow.
20
If we cannot renew our fertilizer registration certificate, which expires in one year, we will be unable to sell some of our products which will cause our sales revenues to significantly decrease.
All fertilizers produced in China must be registered with the PRC Ministry of Agriculture. No fertilizer can be manufactured without such registration. We have obtained a Fertilizer Registration Certificate from the PRC Ministry of Agriculture. Such certificate was issued in February 2008 and it will be reissued in March 2009. We are in the process of renewing it as required by the PRC Ministry of Agriculture and it should be granted in March 2009. Upon issuance, we will transfer the license into the name of the CJV.
Our belief is that the PRC Ministry of Agriculture generally will grant an application for renewal in the absence of illegal activity by the applicant. However, there is no guarantee that the PRC Ministry of Agriculture will grant renewal of our Fertilizer Registration Certificate. If we cannot obtain the necessary renewal, we will not be able to manufacture and sell our fertilizer products in China which will cause the termination of our commercial operations.
Key employees are essential to growing our business.
Mr. Zishen Wu and Professor Gao Jing are essential to our ability to continue to grow our business. Mr. Zishen Wu has established relationships within the industries in which we operate. Professor Gao has performed all of the independent research and knows our main suppliers and has other important industry relationships. If they or other members of our senior management were to leave us, our growth strategy might be hindered, which could limit our ability to increase revenue. In addition, we face competition for attracting skilled personnel. If we fail to attract and retain qualified personnel to meet current and future needs, this could slow our ability to grow our business, which could result in a decrease in market share.
We will continue to encounter risks and difficulties in implementing our business model, including potential failure to:
| · | increase awareness of our products, protect our reputation and develop customer loyalty; |
| · | manage our expanding operations and service offerings; |
| · | maintain adequate control of our expenses; and |
| · | anticipate and adapt to changing conditions in the markets in which we operate as well as the impact of any changes in government regulation, mergers and acquisitions involving our competitors, technological developments and other significant competitive and market dynamics. |
If we are not successful in addressing any or all of these risks, our business may be materially and adversely affected.
The markets in which we operate are highly competitive and fragmented and we may not be able to maintain market share.
We operate in highly competitive markets and expect competition to persist and intensify in the future. Our competitors are mainly domestic leaders in the fertilizer markets in China. We face the risk that new competitors with greater resources than us will enter our markets.
21
If we need additional financing, we may not be available to find such financing on satisfactory terms or at all.
Our capital requirements may be accelerated as a result of many factors, including timing of development activities, underestimates of budget items, unanticipated expenses or capital expenditures, future product opportunities with collaborators, future licensing opportunities and future business combinations. Consequently, we may need to seek additional debt or equity financing, which may not be available on favorable terms, if at all, and which may be dilutive to our stockholders.
We may seek to raise additional capital through public or private equity offerings, debt financings or additional corporate collaboration and licensing arrangements. To the extent we raise additional capital by issuing equity securities, our stockholders may experience dilution. To the extent that we raise additional capital by issuing debt securities, we may incur substantial interest obligations, may be required to pledge assets as security for the debt and may be constrained by restrictive financial and/or operational covenants. Debt financing would also be superior to our stockholders’ interest in bankruptcy or liquidation. To the extent we raise additional funds through collaboration and licensing arrangements, it may be necessary to relinquish some rights to our technologies or product candidates, or grant licenses on unfavorable terms.
Financial damages may be imposed on us if we are unable to retain certain “financial professionals” as required by the Purchase Agreement.
The Purchase Agreement obligates us to hire a chief financial officer who has experience as a senior financial officer of a United States public reporting company and who is (i) fluent in English, (ii) residing or will reside, upon employment by us, in Asia, and (iii) familiar with (x) GAAP and (y) auditing procedures and compliance for the United States public companies and to enter into an employment agreement with such professional for a term of no less than two years. If we fail to comply timely with the above obligation regarding such financial professional, we may incur financial damages in the amount of 1% of the proceeds of the financing, monthly, up to an aggregate amount of 6% of the amount of the financing.
The imposition of such financial damages would require the use of capital that we had planned to use, and may require, in connection with its business.
If we fail to adequately protect or enforce our intellectual property rights, or to secure rights to patents and trademarks of others, the value of our intellectual property rights could diminish.
Our success, competitive position and future revenues will depend in part on our ability to obtain and maintain patent protection for our products, methods, processes and other technologies, to preserve our trade secrets, to prevent third parties from infringing on our proprietary rights and to operate without infringing the proprietary rights of third parties.
To date, we have filed two patent applications to the State Intellectual Property Office of the PRC and one trademark registration application to the Trademark Bureau of State Administration of Industry and Commerce of the PRC. However, we cannot predict the degree and range of protection patents and trademarks will afford us against competitors. Third parties may find ways to invalidate or otherwise circumvent our proprietary technology and trademark. Third parties may attempt to obtain patents claiming aspects similar to our patent and trademark applications. If we need to initiate litigation or administrative proceedings, such actions may be costly whether we win or lose.
Our success also depends on the skills, knowledge and experience of our scientific and technical personnel, consultants, advisors, licensors and contractors. To help protect our proprietary know-how and inventions for which patents may be unobtainable or difficult to obtain, we rely on trade secret protection and confidentiality agreements. If any of our intellectual property is disclosed, our value would be significantly impaired, and our business and competitive position would suffer.
22
If we infringe the rights of third parties, we could be prevented from selling products, forced to pay damages and compelled to defend against litigation.
If our products, formula, methods, processes and other technologies infringe proprietary rights of other parties, we could incur substantial costs, and may have to obtain licenses (which may not be available on commercially reasonable terms, if at all), redesign our products or processes, stop using the subject matter claimed in the asserted patents, pay damages, or defend litigation or administrative proceedings, which may be costly whether it wins or loses. All of the above could result in a substantial diversion of valuable management resources.
We believe we have taken reasonable steps, including comprehensive internal and external prior patent searches, to ensure we have freedom to operate and that our development and commercialization efforts can be carried out as planned without infringing others’ proprietary rights. However, we cannot guarantee that no third party patent has been filed or will be filed that may contain subject matter of relevance to our development, causing a third party patent holder to claim infringement. Resolving such issues has traditionally resulted, and could in our case result, in lengthy and costly legal proceedings, the outcome of which cannot be predicted accurately.
We have never paid cash dividends and are not likely to do so in the foreseeable future.
We have never declared or paid any cash dividends on our Common Stock. We currently intend to retain any future earnings for use in the operation and expansion of our business. We do not expect to pay any cash dividends in the foreseeable future but will review this policy as circumstances dictate.
We do not have a majority of independent directors serving on our board of directors, which could present the potential for conflicts of interest.
We do not have a majority of independent directors serving on our board of directors. In the absence of a majority of independent directors, our executive officers could establish policies and enter into transactions without independent review and approval thereof. This could present the potential for a conflict of interest between us and our stockholders, generally, and the controlling officers, stockholders or directors.
If we are unable to establish appropriate internal financial reporting controls and procedures, it could cause us to fail to meet our reporting obligations, result in the restatement of our financial statements, harm our operating results, subject us to regulatory scrutiny and sanction, cause investors to lose confidence in our reported financial information and have a negative effect on the market price for shares of our Common Stock.
Effective internal controls are necessary for us to provide reliable financial reports and effectively prevent fraud. We maintain a system of internal control over financial reporting, which is defined as a process designed by, or under the supervision of, our principal executive officer and principal financial officer, or persons performing similar functions, and effected by our board of directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles.
As a public company, we will have significant additional requirements for enhanced financial reporting and internal controls. We are required to document and test our internal control procedures in order to satisfy the requirements of Section 404 of the Sarbanes-Oxley Act of 2002, which requires annual management assessments of the effectiveness of our internal controls over financial reporting and, beginning with our next annual report, a report by our independent registered public accounting firm addressing these assessments. The process of designing and implementing effective internal controls is a continuous effort that requires us to anticipate and react to changes in our business and the economic and regulatory environments and to expend significant resources to maintain a system of internal controls that is adequate to satisfy our reporting obligations as a public company. We cannot assure you that we will not, in the future, identify areas requiring improvement in our internal control over financial reporting. We cannot assure you that the measures we will take to remediate any areas in need of improvement will be successful or that we will implement and maintain adequate controls over our financial processes and reporting in the future as we continue our growth. If we are unable to establish appropriate internal financial reporting controls and procedures, it could cause us to fail to meet our reporting obligations, result in the restatement of our financial statements, harm our operating results, subject us to regulatory scrutiny and sanction, cause investors to lose confidence in our reported financial information and have a negative effect on the market price for shares of our Common Stock.
23
Lack of experience as officers of publicly-traded companies of our management team may hinder our ability to comply with Sarbanes-Oxley Act.
It may be time consuming, difficult and costly for us to develop and implement the internal controls and reporting procedures required by the Sarbanes-Oxley Act. of 2002. We may need to hire additional financial reporting, internal controls and other finance staff in order to develop and implement appropriate internal controls and reporting procedures. If we are unable to comply with the Sarbanes-Oxley Act’s internal controls requirements, we may not be able to obtain the independent auditor certifications that Sarbanes-Oxley Act requires publicly-traded companies to obtain.
We have incurred increased costs as a result of being a public company.
As a public company, we have incurred significant legal, accounting and other expenses that we did not incur as a private company. In addition, the Sarbanes-Oxley Act of 2002, as well as new rules subsequently implemented by the SEC, has required changes in corporate governance practices of public companies. We expect these new rules and regulations to increase our legal, accounting and financial compliance costs and to make certain corporate activities more time-consuming and costly. In addition, we will incur additional costs associated with our public company reporting requirements. We are currently evaluating and monitoring developments with respect to these new rules, and we cannot predict or estimate the amount of additional costs we may incur or the timing of such costs.
Risks Associated With Doing Business In China
There are substantial risks associated with doing business in China, as set forth in the following risk factors.
Our operations and assets in China are subject to significant political and economic uncertainties.
Changes in PRC laws and regulations, or their interpretation, or the imposition of confiscatory taxation, restrictions on currency conversion, imports and sources of supply, devaluations of currency or the nationalization or other expropriation of private enterprises could have a material adverse effect on our business, results of operations and financial condition. Under our current leadership, the Chinese government has been pursuing economic reform policies that encourage private economic activity and greater economic decentralization. There is no assurance, however, that the Chinese government will continue to pursue these policies, or that it will not significantly alter these policies from time to time without notice.
We derive a substantial portion of ours sales from China.
Substantially all of our sales are generated from China. We anticipate that sales of our products in China will continue to represent a substantial proportion of our total sales in the near future. Any significant decline in the condition of the PRC economy could adversely affect consumer demand of our products, among other things, which in turn would have a material adverse effect on our business and financial condition.
Currency fluctuations and restrictions on currency exchange may adversely affect our business, including limiting our ability to convert Chinese renminbi into foreign currencies and, if Chinese renminbi were to decline in value, reducing our revenue in U.S. dollar terms.
24
Our reporting currency is the U.S. dollar and our operations in China use their local currency as their functional currencies. Substantially all of our revenue and expenses are in Chinese renminbi. We are subject to the effects of exchange rate fluctuations with respect to any of these currencies. For example, the value of the renminbi depends to a large extent on Chinese government policies and China’s domestic and international economic and political developments, as well as supply and demand in the local market. Since 1994, the official exchange rate for the conversion of renminbi to the U.S. dollar had generally been stable and the renminbi had appreciated slightly against the U.S. dollar. However, on July 21, 2005, the Chinese government changed its policy of pegging the value of Chinese renminbi to the U.S. dollar. Under the new policy, Chinese renminbi may fluctuate within a narrow and managed band against a basket of certain foreign currencies. This change in policy has resulted in an approximately 21.3% appreciation of the Renminbi against the U.S. dollar between July 21, 2005 and December 31, 2008.
It is possible that the Chinese government could adopt a more flexible currency policy, which could result in more significant fluctuation of Chinese renminbi against the U.S. dollar. We can offer no assurance that Chinese renminbi will be stable against the U.S. dollar or any other foreign currency.
The income statements appearing elsewhere herein are translated into U.S. dollars at the average exchange rates in each applicable period. To the extent the U.S. dollar strengthens against foreign currencies, the translation of these foreign currencies denominated transactions will result in reduced revenue, operating expenses and net income for our international operations. Similarly, to the extent the U.S. dollar weakens against foreign currencies, the translation of these foreign currency denominated transactions will result in increased revenue, operating expenses and net income for our international operations. We are also exposed to foreign exchange rate fluctuations as we convert the financial statements of Fullmax Ltd and Asia Standard Oil, Ltd into U.S. dollars in consolidation. If there is a change in foreign currency exchange rates, the conversion of the foreign subsidiaries’ financial statements into U.S. dollars will lead to a translation gain or loss which is recorded as a component of other comprehensive income. In addition, we may have certain assets and liabilities that are denominated in currencies other than the relevant entity’s functional currency. Changes in the functional currency value of these assets and liabilities create fluctuations that will lead to a transaction gain or loss. We have not entered into agreements or purchased instruments to hedge our exchange rate risks, although we may do so in the future. The availability and effectiveness of any hedging transaction may be limited and we may not be able to successfully hedge our exchange rate risks.
Although Chinese governmental policies were introduced in 1996 to allow the convertibility of Chinese renminbi into foreign currency for current account items, conversion of Chinese renminbi into foreign exchange for capital items, such as foreign direct investment, loans or securities, requires the approval of the State Administration of Foreign Exchange, or SAFE, which is under the authority of the People’s Bank of China. These approvals, however, do not guarantee the availability of foreign currency conversion. We cannot be sure that we will be able to obtain all required conversion approvals for our operations or that Chinese regulatory authorities will not impose greater restrictions on the convertibility of Chinese renminbi in the future. Because a significant amount of our future revenue may be in the form of Chinese renminbi, our inability to obtain the requisite approvals or any future restrictions on currency exchanges could limit our ability to utilize revenue generated in Chinese renminbi to fund our business activities outside of China, or to repay foreign currency obligations, including our debt obligations, which would have a material adverse effect on our financial condition and results of operations.
25
We may have limited legal recourse under PRC law if disputes arise under our contracts with third parties.
The Chinese government has enacted some laws and regulations dealing with matters such as corporate organization and governance, foreign investment, commerce, taxation and trade. However, their experience in implementing, interpreting and enforcing these laws and regulations is limited, and our ability to enforce commercial claims or to resolve commercial disputes is unpredictable. If our new business ventures are unsuccessful, or other adverse circumstances arise from these transactions, we face the risk that the parties to these ventures may seek ways to terminate the transactions, or, may hinder or prevent us from accessing important information regarding the financial and business operations of these acquired companies. The resolution of these matters may be subject to the exercise of considerable discretion by agencies of the Chinese government, and forces unrelated to the legal merits of a particular matter or dispute may influence their determination. Any rights we may have to specific performance, or to seek an injunction under PRC law, in either of these cases, are severely limited, and without a means of recourse by virtue of the Chinese legal system, we may be unable to prevent these situations from occurring. The occurrence of any such events could have a material adverse effect on our business, financial condition and results of operations.
We must comply with the Foreign Corrupt Practices Act.
We are required to comply with the United States Foreign Corrupt Practices Act, which prohibits U.S. companies from engaging in bribery or other prohibited payments to foreign officials for the purpose of obtaining or retaining business. Foreign companies, including some of our competitors, are not subject to these prohibitions. Corruption, extortion, bribery, pay-offs, theft and other fraudulent practices occur from time-to-time in mainland China. If our competitors engage in these practices, they may receive preferential treatment from personnel of some companies, giving our competitors an advantage in securing business or from government officials who might give them priority in obtaining new licenses, which would put us at a disadvantage. Although we inform our personnel that such practices are illegal, we cannot assure you that our employees or other agents will not engage in such conduct for which we might be held responsible. If our employees or other agents are found to have engaged in such practices, we could suffer severe penalties.
We may not be guaranteed of a continuance to receive the preferential tax treatment we currently enjoy under PRC law, and dividends paid to us from our operations in China may become subject to income tax under PRC law.
The rate of income tax on companies in China may vary depending on the availability of preferential tax treatment or subsidies based on their industry or location. The PRC government promulgated on March 16, 2007 the new Enterprise Income Tax Law that will be effective as of January 1, 2008. Pursuant to the new law, the enterprise income tax of 25% shall be applied to any enterprise. While the Company was approved by the local tax authority in April 2008 to pay revenue tax at a rate of 1.25% of gross revenue rather than on 25% of net income as defined by PRC accounting principles, and to receive a tax rebate of 31.2% on all income taxes paid prior to this approval, and continue to receive the rebate after the approval date, we do not know how long this will continue or if such new law may change the preferential treatment will be granted to us. Any loss or substantial reduction of the tax benefits enjoyed by us would reduce our net profit.
Changes in foreign exchange regulations in the PRC may affect our ability to pay dividends in foreign currency or conduct other foreign exchange business.
The Renminbi is not currently a freely convertible currency, and the restrictions on currency exchanges may limit our ability to use revenues generated in RMB to fund our business activities outside the PRC or to make dividends or other payments in United States dollars. The PRC government strictly regulates conversion of RMB into foreign currencies. Over the years, foreign exchange regulations in the PRC have significantly reduced the government’s control over routine foreign exchange transactions under current accounts. In the PRC, the State Administration for Foreign Exchange, or the SAFE, regulates the conversion of the RMB into foreign currencies. Pursuant to applicable PRC laws and regulations, foreign invested enterprises incorporated in the PRC are required to apply for “Foreign Exchange Registration Certificates.” Currently, conversion within the scope of the “current account” (e.g. remittance of foreign currencies for payment of dividends, etc.) can be effected without requiring the approval of SAFE. However, conversion of currency in the “capital account” (e.g. for capital items such as direct investments, loans, securities, etc.) still requires the approval of SAFE. In addition, on October 21, 2005, SAFE issued the Notice on Issues Relating to the Administration of Foreign Exchange in Fundraising and Reverse Investment Activities of Domestic Residents Conducted via Offshore Special Purpose Companies (“Notice 75”), which became effective as of November 1, 2005. Notice 75 replaced the two rules issued by SAFE in January and April 2005. According to Notice 75:
26
| · | prior to establishing or assuming control of an offshore company for the purpose of obtaining overseas equity financing with assets or equity interests in an onshore enterprise in the PRC, each PRC resident, whether a natural or legal person, must complete the overseas investment foreign exchange registration procedures with the relevant local SAFE branch; |
| · | an amendment to the registration with the local SAFE branch is required to be filed by any PRC resident that directly or indirectly holds interests in that offshore company upon either (1) the injection of equity interests or assets of an onshore enterprise to the offshore company, or (2) the completion of any overseas fund raising by such offshore company; and |
| · | an amendment to the registration with the local SAFE branch is also required to be filed by such PRC resident when there is any material change in the capital of the offshore company that does not involve any return investment, such as (1) an increase or decrease in its capital, (2) a transfer or swap of shares, (3) a merger or division, (4) a long term equity or debt investment, or (5) the creation of any security interests. |
Moreover, Notice 75 applies retroactively. As a result, PRC residents who have established or acquired control of offshore companies that have made onshore investments in the PRC in the past are required to complete the relevant overseas investment foreign exchange registration procedures by March 31, 2006. Under the relevant rules, failure to comply with the registration procedures set forth in Notice 75 may result in restrictions being imposed on the foreign exchange activities of the relevant onshore company, including the payment of dividends and other distributions to its offshore parent or affiliate and the capital inflow from the offshore entity, and may also subject relevant PRC residents to penalties under PRC foreign exchange administration regulations.
In addition, SAFE issued updated internal implementing rules (“Implementing Rules”) in relation to Notice 75. The Implementing Rules were promulgated and became effective on May 29, 2007. Such Implementing Rules provide more detailed provisions and requirements regarding the overseas investment foreign exchange registration procedures. However, even after the promulgation of Implementing Rules there still exist uncertainties regarding the SAFE registration for PRC residents’ interests in overseas companies. It remains uncertain whether PRC residents shall go through the overseas investment foreign exchange registration procedures under Notice 75 or Implementing Rules, who may indirectly hold our shares through the participation and exercise of incentive stock options granted to our members of our management by Full Alliance.
As a result, we cannot predict how they will affect our business operations following a business combination. For example, our ability to conduct foreign exchange activities following a business combination, such as remittance of dividends and foreign-currency-denominated borrowings, may be subject to compliance with the SAFE registration requirements by such PRC residents, over whom we have no control. In addition, we cannot assure you that such PRC residents will be able to complete the necessary approval and registration procedures required by the SAFE regulations. We will require all our shareholders, following a business combination, who are PRC residents to comply with any SAFE registration requirements, if required by Notice 75, Implementing Rules or other applicable PRC laws and regulations, although we have no control over either our shareholders or the outcome of such registration procedures. Such uncertainties may restrict our ability to implement our business combination strategy and adversely affect our business and prospects following a business combination.
Recent PRC regulations relating to mergers and acquisitions of domestic enterprises by foreign investors may increase the administrative burden we face and create regulatory uncertainties.
On September 8, 2006, six PRC regulatory agencies, namely, the PRC Ministry of Commerce, or MOFCOM, the State Assets Supervision and Administration Commission, or SASAC, the State Administration for Taxation, the State Administration for Industry and Commerce, the China Securities Regulatory Commission, or CSRC, and the State Administration of Foreign Exchange, or SAFE, jointly adopted the Regulations on Mergers and Acquisitions of Domestic Enterprises by Foreign Investors, or New M&A Rule, which became effective on September 8, 2006. The New M&A Rule purports, among other things, to require offshore special purpose vehicles, or SPVs, formed for overseas listing purposes through acquisitions of PRC domestic companies and controlled by PRC companies or individuals, to obtain the approval of the CSRC prior to publicly listing their securities on an overseas stock exchange.
27
On September 21, 2006, pursuant to the New M&A Rule and other PRC laws and regulations, the CSRC, in its official website, promulgated relevant guidance with respect to the issues of listing and trading of domestic enterprises’ securities on overseas stock exchanges (the “Administrative Permits”), including a list of application materials with respect to the listing on overseas stock exchanges by SPVs.
Based on our understanding of current PRC Laws, we are not sure whether the New M&A Rule would require us or our entities in China to obtain the CSRC approval in connection with the transaction contemplated by the Exchange Agreement in connection with the share exchange.
Further, if the PRC government finds that we or our management members did not obtain the CSRC approval, which CSRC may think we should have obtained before our executing the Exchange Agreement, we could be subject to severe penalties. The New M&A Rule does not stipulate the specific penalty terms, so we are not able to predict what penalties we may face, and how such penalties will affect our business operations or future strategy.
The Chinese government exerts substantial influence over the manner in which we must conduct our business activities.
China only recently has permitted provincial and local economic autonomy and private economic activities, and, as a result, we are dependent on our relationship with the local government in the province in which we operate our business. Chinese government has exercised and continues to exercise substantial control over virtually every sector of the Chinese economy through regulation and state ownership. Our ability to operate in China may be harmed by changes in its laws and regulations, including those relating to taxation, environmental regulations, land use rights, property and other matters. We believe that our operations in China are in material compliance with all applicable legal and regulatory requirements. However, the central or local governments of these jurisdictions may impose new, stricter regulations or interpretations of existing regulations that would require additional expenditures and efforts on our part to ensure our compliance with such regulations or interpretations. Accordingly, government actions in the future, including any decision not to continue to support recent economic reforms and to return to a more centrally planned economy or regional or local variations in the implementation of economic policies, could have a significant effect on economic conditions in China or particular regions thereof, and could require us to divest ourselves of any interest we then hold in Chinese properties.
Future inflation in China may inhibit our activity to conduct business in China.
In recent years, the Chinese economy has experienced periods of rapid expansion and high rates of inflation. During the past ten years, the rate of inflation in China has been as high as 20.7% and as low as (2.2)%. These factors have led to the adoption by Chinese government, from time to time, of various corrective measures designed to restrict the availability of credit or regulate growth and contain inflation. While inflation has been more moderate since 1995, high inflation may in the future cause Chinese government to impose controls on credit and/or prices, or to take other action, which could inhibit economic activity in China, and thereby harm the market for our products.
Government regulations on environmental matters in China may adversely impact on our business.
Our manufacturing operations are subject to numerous laws, regulations, rules and specifications relating to human health and safety and the environment. These laws and regulations address and regulate, among other matters, wastewater discharge, air quality and the generation, handling, storage, treatment, disposal and transportation of solid and hazardous wastes and releases of hazardous substances into the environment. In addition, third parties and governmental agencies in some cases have the power under such laws and regulations to require remediation of environmental conditions and, in the case of governmental agencies, to impose fines and penalties. We make capital expenditures from time to time to stay in compliance with applicable laws and regulations.
28
We have obtained all permits and approvals and filed all registrations required for the conduct of its business, except where the failure to obtain any permit or approval or file any registration would not have a material adverse effect on our business, financial condition and results of operations. We are in compliance in all material respects with the numerous laws, regulations, rules, specifications and permits, approvals and registrations relating to human health and safety and the environment except where noncompliance would not have a material adverse effect on our business, financial condition and results of operations.
The PRC governmental authorities have not revealed any material environmental liability that would have a material adverse effect on us. We have not been notified by any governmental authority of any continuing noncompliance, liability or other claim in connection with any of our properties or business operations, nor are we aware of any other material environmental condition with respect to any of our properties or arising out of our business operations at any other location. However, in connection with the ownership and operation of its properties (including locations to which we may have sent waste in the past) and the conduct of its business, we potentially may be liable for damages or cleanup, investigation or remediation costs.
No assurance can be given that all potential environmental liabilities have been identified or properly quantified or that any prior owner, operator, or tenant has not created an environmental condition unknown to us. Moreover, no assurance can be given that (i) future laws, ordinances or regulations will not impose any material environmental liability or (ii) the current environmental condition of the properties will not be affected by the condition of land or operations in the vicinity of the properties (such as the presence of underground storage tanks), or by third parties unrelated to us. State and local environmental regulatory requirements change often.
It is possible that compliance with a new regulatory requirement could impose significant compliance costs on us. Such costs could have a material adverse effect on our business, financial condition and results of operations.
We may have difficulty establishing adequate management, legal and financial controls in the PRC.
The PRC historically has been deficient in Western style management and financial reporting concepts and practices, as well as in modern banking, computer and other control systems. We may have difficulty in hiring and retaining a sufficient number of qualified employees to work in the PRC. As a result of these factors, we may experience difficulty in establishing management, legal and financial controls, collecting financial data and preparing financial statements, books of account and corporate records and instituting business practices that meet Western standards. We may have difficulty establishing adequate management, legal and financial controls in the PRC.
We face risks related to health epidemics and other outbreaks.
Our business could be adversely affected by the effects of avian flu, severe acute respiratory syndrome, or SARS, or another epidemic or outbreak. From 2005 to 2007, there have been reports on the occurrences of avian flu in various parts of China and elsewhere in Asia, including a few confirmed human cases and deaths. Any prolonged recurrence of avian flu, SARS or other adverse public health developments in China may have a material adverse effect on our business operations. Our operations may be impacted by a number of health-related factors, including, among other things, quarantines or closures of our factories and the facilities of our supplier and customers which could severely disrupt our operations, and a general slowdown in the Chinese economy. Any of the foregoing events or other unforeseen consequences of public health problems could adversely affect our business and results of operations. We have not adopted any written preventive measures or contingency plans to combat any future outbreak of avian flu, SARS or any other epidemic.
29
Risks Related to the Common Stock
There may not be sufficient liquidity in the market for our securities in order for investors to sell their securities.
There is currently only a limited public market for our common stock, which is listed on the OTC Bulletin Board. The share volume of our common stock during the month of February 2009 was 3,651 shares. As of March 13, 2009, the closing price of our common stock was $1.50.
The market price of our Common Stock may be volatile.
The market price of our Common Stock has been and will likely continue to be highly volatile, as is the stock market in general, and the market for OTC quoted stocks in particular. Some of the factors that may materially affect the market price of our Common Stock are beyond our control, such as changes in financial estimates by industry and securities analysts, conditions or trends in the industry in which we operate or sales of our Common Stock. These factors may materially adversely affect the market price of our Common Stock, regardless of our performance. In addition, the public stock markets have experienced extreme price and trading volume volatility. This volatility has significantly affected the market prices of securities of many companies for reasons frequently unrelated to the operating performance of the specific companies. These broad market fluctuations may adversely affect the market price of our Common Stock.
Our Common Stock is considered “penny stock.”
The SEC has adopted regulations which generally define “penny stock” to be an equity security that has a market price of less than $5.00 per share, subject to specific exemptions. The market price of our Common Stock is less than $5.00 per share and therefore is a “penny stock.” Brokers and dealers effecting transactions in “penny stock” must disclose certain information concerning the transaction, obtain a written agreement from the purchaser and determine that the purchaser is reasonably suitable to purchase the securities. These rules may restrict the ability of brokers or dealers to sell our Common Stock and may affect your ability to sell shares.
| ITEM 1B. | Unresolved Staff Comments |
None.
| ITEM 2 | Properties |
Our principal executive offices are located at 6th floor, Xue Yuan International Tower, No. 1 Zhichun Road, Haidian District, Beijing PRC and the telephone number is +86-10-8231-8626. The office space is approximately 1,000 square meters in area. Inner Mongolia Yongye’s main production facility is in the High Tech Economic Development Zone in Hohhot City in Inner Mongolia.
There is no private ownership of land in China. All land ownership is held by the government of the PRC, its agencies and collectives. Land use rights can be transferred upon approval by the land administrative authorities of the PRC (State Land Administration Bureau) upon payment of the required land transfer fee. Inner Mongolia Yongye owns the land use rights for the land on which its manufacturing facility is situated, which have a term of 50 years from 2003.
| ITEM 3 | Legal Proceedings |
We are not a party to any material legal proceedings nor are we aware of any circumstance that may reasonably lead a third party to initiate legal proceedings against us.
30
| ITEM 4 | Submission Of Matters to a Vote Of Security Holders |
No matter was submitted to a vote of security holders during the fourth quarter of the fiscal year ended December 31, 2008.
PART II
| ITEM 5 | Market For Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
Market Information
Our Common Stock is quoted on the OTC Bulletin Board (“OTCBB”) under the trading symbol “YGYB”. Until April 29, 2008, our common stock was traded under the symbol “GDTN.OB”. The last reported price for our Common Stock on the OTCBB on March 13, 2009 was $1.50 per share..
The following table shows by each fiscal quarter and partial period, where applicable the range of high and low bid quotations reported by the OTCBB in each fiscal quarter from January 1, 2008 to December 31, 2008 and the quarter from January 1, 2009 to March 13, 2009. There were no reported bids for our common stock during 2007, 2006 and the first quarter of 2008. The OTCBB quotations reflect inter-dealer prices, without retail mark-up, mark-down or commission and may not represent actual transactions.
Year | Period | High | Low | |||||||
| 2008 | First Quarter | |||||||||
| Second Quarter | $ | 3.75 | $ | 1.75 | ||||||
| Third Quarter | 4.65 | 2.00 | ||||||||
| Fourth Quarter | 2.00 | 1.25 | ||||||||
| 2009 | First Quarter (January 1 – March 13) | $ | 1.60 | $ | 0.65 | |||||
Holders
As of December 31, 2008, there were approximately 48 active record holders of our common stock.
Dividends
We have not paid any cash dividends on shares of our common stock and do not plan to do so in the near future. We currently plan to retain future earnings to fund the development and growth of our business. Any future determination related to our dividend policy will be made at the discretion of our Board of Directors.
Securities Authorized for Issuance Under Equity Compensation Plans
As of December 31, 2008, we did not have any equity compensation plans; however we may wish to implement such plans in the future.
Equity Repurchases
No repurchases of our common stock were made during the fourth quarter of our fiscal year ended December 31, 2008.
| ITEM 6 | Selected Financial Data |
As a smaller reporting company, we are not required to provide this information.
31
| ITEM 7 | Management’s Discussion and Analysis of Financial Condition and Results of Operations |
Except for the historical information contained herein, the matters discussed in this “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” and elsewhere in this report are forward-looking statements that involve risks and uncertainties. The factors listed in the section captioned “Risk Factors,” as well as any cautionary language in this report, provided examples of risks, uncertainties and events that may cause our actual results to differ materially from those projected. Except as may be required by law, we undertake no obligation to update any forward-looking statement to reflect events after the date of this prospectus.
Overview
We are engaged in the sales of fulvic acid based liquid and powder nutrient compounds. Based on industry research and government information, we believe our proprietary technology for fulvic acid extraction creates some of the purest and most bioactive fulvic acid in China and thus some of the most effective plant and animal nutrients on the market. Our contractual relationship with our predecessor company, Inner Mongolia Yongye, is to create our own fulvic acid and to control our manufacturing process from procurement of raw materials to final production, we believe that we can provide a high quality product to our customers with the expectation of more reliable results season to season.
We are headquartered in Beijing, China and Inner Mongolia Yongye’s manufacturing plant is located in the Inner Mongolia province of China. Currently, we sell two lines of product based on our fulvic acid base: plant nutrition liquid compound and animal nutrition food additive. Our products start with our fulvic Acid base then, in addition, we add other natural substances to customize the base for use in our plant or animal lines of products. Our plant products add naturally occurring macro and micro nutrients such as nitrogen, phosphorus, potassium, boron and zinc. Our animal products add natural herbs which provide antibiotic type properties.
Our predecessor company, Inner Mongolia Yongye, began recording sales of its first plant product in 2005 and by 2007 marketed and sold both plant product in 10 provinces and animal product in 2 provinces. In 2008 we sold approximately 5,100 tons of plant product (427,200 units) which represented 93% of revenue at USD $44.8M and sold it to approximately 150,000 farmers. We also sold approximately 6 tons of our animal product (approximately 98,000 units) to farmers to treat over 100,000 animals. This represented 7% of revenue at USD $3.25M. In its highly concentrated form, our plant product was sprayed on approximately 2% of all available arable land in our 10 provinces and in our largest province, Xinjiang, it was applied to 5% of the land available for cultivation. Yongye’s top 3 provinces by revenue for 2008 represented 82% of sales and were Hebei at $20,541,267 (43%), Xinjiang at 13,177,694 (27%) and Gansu at $ 5,663,011 (12%).
Recent Development
The financial statements we are reporting for 2007 are for our predecessor company, Inner Mongolia Yongye, which has now transferred all 2008 and 2009 sales contracts and income, intellectual property and patents, and personnel, exclusive of manufacturing and R&D personnel, into the name of Yongye Nongfeng Biotechnology, the new Cooperative Joint Venture. After this transfer, Inner Mongolia Yongye became the primary contract manufacturing company for the new Cooperative Joint Venture and provided product at a cost plus price for the entire year of 2008. Inner Mongolia Yongye also kept the existing assets and long-term liabilities on its balance sheet.
32
Factors affecting our operating results
Demand for our products
One major tenet of the PRC government’s 11th Five-Year National Economic and Social Plan (the “NESDP”) (2006-2010) is the focus towards developing China’s western region. This is one of the top-five economic priorities of the nation. The goal is to increase rural income growth which will in turn increase demand for more food and agriculture products. Currently, a large majority of our products are sold in this western region and this government focus will increase our opportunity to sell more plant and animal nutrients to farmers who have to keep up with the demand for higher quantity and higher quality of products.
According to the Asian Development Bank statistics, well over 60% of the nation’s 1.3 billion total population is comprised of low-income, rural farmers. According to the 11th Five-Year NESDP (2006-2010), raising the level of rural income is a top economic and social goal for the country. Many government initiatives, including removal of certain agricultural and local product taxes, have been implemented to spur rural income development. The government expects annual rural income to grow between 5% and 10% through 2010. Additionally, according to the National Population and Family Planning Commission, China's population will reach 1.5 billion by 2030. Therefore, the country has the challenge of producing approximately 100 million more tons of crops needed to feed the additional 200 million people. This put pressure on the agricultural system to increase production capacity.
Supply of Finished Goods
Currently, we purchase our finished goods from our main supplier, Inner Mongolia Yongye and then sell it through our distribution system. In order to generate greater profit margins, we set out to control our cost of goods sold and have put into place a fixed rate contract with our main supplier and this will extend over the next five years. Each quarter we will go through a review process with our supplier to adjust the fixed rate for the next quarter. We have not received any rate increases in 2008.
Earthquake in Sichuan
The earthquake in Sichuan was a devastating event in the recent history of China. While the impact was felt all the way to Beijing, the disruption of business and the ensuing relief efforts were largely contained to the province itself and mainly to the areas nearest the epicenter. Because of this, the impact to our business was minimal. China’s Agriculture Minister Sun Zhengcai said in an interview with Xinhua that, “The earthquake will not change the nation-wide situation of agricultural production this year since local output of the affected area is quite small compared to that of the whole country,” Sun acknowledged that, “The damage was mainly to planted crops and livestock,” he said, adding an urgent harvesting and planting effort has helped minimize the impact and which had no national implications.” Furthermore, he said that “food security remains guaranteed.”
Seasonality
We typically face the seasonal demand patterns similar to other companies in our sector. In general, the first and fourth quarters are typically our slowest quarters and in 2008 we brought in approximately 20% and 6% of sales in these quarters. The second and third quarters drive the bulk of our overall sales with 36% and 38% respectively of the year’s net sales. Our Shengmingsu plant line faces the most seasonality of our two product lines with our Shengmingsu for animals experiencing less fluctuation during the year due to seasonal buying patterns. This year, while we did experience fluctuations by quarter of our animal line, this was due more in part to fluctuations in our sales and marketing efforts and not due to seasonal buying patterns. We will not typically face this type of revenue fluctuations for this product which doesn’t experience the same seasonal tapering which our plant product faces.
33
Drought
In the last half year of 2008, there have been many news reports of the drought in certain areas of China and the obvious impact it would have on agriculture and thus on our company. Through our contact with government agencies, sales and support staff, distributors, and branded store network, we have a very good connection with the farming community in the markets we serve and thus have good information about the real impacts of the drought on the Chinese farmer. Thus far the impact has not been felt on our revenue for the year end 2008 and we do not believe the impact will curtail our sales activities in the near term for several reasons.
Since the news hit about the drought in late 2008, there have been several events which have occurred to mitigate the water shortages faced by farmers in drought conditions such as additional governmental spending on increased efforts to irrigate land from other water sources and additional rainfall on arable land has come in northern China. The State Flood Control and Drought Relief Headquarters recently said that there has been a reduction of farmland affected. The government has also begun their stimulus injections into the agriculture community to help ward off the affects of any drought induced financial hardships.
Several market conditions also bode well for us in the sales of our plant product during this time. Overall, the drought has impacted northern China and primarily large field crop growers. We are currently focused mostly on economic crops except for the Xinjiang province. Additionally, we emphasize that the drought resistance nature of our plant product will actually assist farmers because the water that is used will be held more effectively. Thus, while the drought will impact China, we feel there are many factors working for the company to help us ward off financial impact due to the drought.
FINANCIAL HIGHLIGHTS
Yongye Biotechnology International, Inc. and Subsidiaries
CONSOLIDATED STATEMENTS OF INCOME AND COMPREHENSIVE INCOME
| Yongye Biotechnology | ||||||||
| International, Inc. | The Predecessor | |||||||
| and Subsidiaries | Inner Mongolia Yongye | |||||||
| FOR YEAR ENDED | FOR YEAR ENDED | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| SALES | $ | 48,092,271 | $ | 13,137,406 | ||||
| COST | 23,165,684 | 7,274,710 | ||||||
| GROSS PROFIT | 24,926,587 | 5,862,696 | ||||||
| SELLING EXPENSES | 8,665,755 | 449,168 | ||||||
| GENERAL AND ADMINISTRATIVE EXPENSES | 2,573,017 | 476,828 | ||||||
| INCOME FROM OPERATIONS | 13,687,815 | 4,936,700 | ||||||
| OTHER EXPENSES | ||||||||
| Interest expenses | (3,135 | ) | (212,239 | ) | ||||
| Other expenses | (526,039 | ) | (365,907 | ) | ||||
| TOTAL OTHER INCOME (EXPENSES) | (529,174 | ) | (578,146 | ) | ||||
| INCOME BEFORE PROVISION FOR INCOME TAXES AND MINORITY INTEREST | 13,158,641 | 4,358,554 | ||||||
| PROVISION FOR INCOME TAXES | 864,292 | - | ||||||
| NET INCOME BEFORE MINORITY INTEREST | 12,294,349 | 4,358,554 | ||||||
| PROVISION FOR MINORITY INTEREST | 1,102,388 | - | ||||||
| NET INCOME | 11,191,961 | 4,358,554 | ||||||
34
RESULTS OF OPERATIONS
Fiscal year ended December 31, 2008 compared with Fiscal year ended December 31, 2007
Our business for the year ended December 31, 2008 exceeded previous estimates and grew at a rate of 266% in net sales or a $34.9M increase over the same period in 2007. This demonstrated our ability to scale our distribution and sales mechanisms and drive robust revenue based on our new business model throughout the full year of 2008. This was primarily driven by higher volumes shipped through the first three quarters with the fourth quarter being the slowest due to seasonality. Numerous factors contributed to our strong growth: strong market demand, continued cost containment and steady pricing model, government policy support, industry growth, and sales and marketing leadership in our markets.
The following table shows, for the periods indicated, information derived from our consolidated statements of income.
| Predecessor | ||||||||||||
| Yongye Biotechnology | Inner Mongolia | |||||||||||
| International, Inc. | Yongye | Increase/ (decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Net Sales | $ | 48,092,271 | $ | 13,137,406 | 266 | % | ||||||
| Gross Profit | $ | 24,926,587 | $ | 5,862,696 | 325 | % | ||||||
| Operating Income | $ | 13,687,815 | $ | 4,936,700 | 177 | % | ||||||
| Net Income | $ | 11,191,961 | $ | 4,358,554 | 157 | % | ||||||
| Gross Margins | 52 | % | 45 | % | 7 | % | ||||||
| Net Margins | 23 | % | 33 | % | (10 | )% | ||||||
| EPS- Basic | $ | 0.66 | $ | 0.38 | 74 | % | ||||||
| EPS- Diluted | $ | 0.64 | $ | 0.38 | 67 | % | ||||||
Our financial position at December 31, 2008 and December 31, 2007:
| Predecessor | ||||||||||||
| Yongye Biotechnology | Inner Mongolia | |||||||||||
| International Inc. | Yongye | Increase /(decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Cash | $ | 4,477,477 | $ | 376,002 | 1091 | % | ||||||
| Accounts Receivable, net | $ | 2,748,042 | $ | 1,630,609 | 69 | % | ||||||
| PP&E, net | $ | 5,368,074 | $ | 2,486,487 | 116 | % | ||||||
| Total assets | $ | 34,504,261 | $ | 23,131,656 | 49 | % | ||||||
| Short term liability | $ | 3,493,911 | $ | 10,208,031 | (66 | )% | ||||||
| Long term debt | $ | 397,773 | $ | 12,153 | 3,173 | % | ||||||
| Total stockholders’ equity | $ | 29,410,189 | $ | 12,911,472 | 128 | % | ||||||
35
We increased our cash position to $4,477,477 at December 31, 2008 from $376,002 at December 31, 2007, which is an overall increase of $4,101,475, or 1091%. This was primarily due to increased collections of accounts receivable and the April and September financings. Accounts receivable increased by 69% to $2,748,042 as of December 31, 2008 from $1,630,609 as of December 31, 2007 due to increased growth in sales for the year. Property, plant and equipment increased to $5,368,074 at December 31, 2008 from $2,486,487 at December 31, 2007, which was a 116% increase and was largely due to the addition of the 8,000TPA facility in the third quarter. Additionally, shareholders’ equity increased by $16,498,717 to $29,410,189 as of December 31, 2008, which is an overall 128% increase compared to $12,911,472 on December 31, 2007, and was due primarily to the influx of our two financing transactions on April 17, 2008 and September 5, 2008 and net income for the year.
Net Sales
| Yongye Biotechnology | Predecessor | |||||||||||
| International Inc. | Inner Mongolia Yongye | Increase/(decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Sales | $ | 48,092,271 | $ | 13,137,406 | 266 | % | ||||||
| Gross Profit | $ | 24,926,587 | $ | 5,862,696 | 325 | % | ||||||
| Gross Margin | 52 | % | 45 | % | 7 | % | ||||||
Sales revenue for the year ended December 31, 2008 was $48,092,271, an increase of $34,954,865 or 266%, compared with the corresponding period in 2007. This increase was the result of an increase in sales volume due to rapid expansion of our sales network and an increased footprint in each community via our branded stores.
Gross profit for the year ended December 31, 2008 was $24,926,587, and represented 52% of sales. This was an increase of $19,063,891, or 325%, when compared with $5,862,696 in the corresponding period in 2007. However, when compared as a percentage of revenues, Gross Profit Margin increased 7% from 45% to 52% from 2007 to 2008. The overall increase in margin was largely due to our fixed rate contract for finished goods as opposed to the “Predecessor” company’s model of buying raw materials and producing goods which put them at the risk of fluctuation of raw materials prices.
Additionally, Yongye Nongfeng did not pay VAT for the plant products sold, which increased its gross sales in 2008, whereas Inner Mongolia Yongye paid VAT for all products sold in 2007, which led to an approximate 4% decrease in gross sales.
| Predecessor | ||||||||||||
| Yongye Biotechnology | Inner Mongolia | Increase / | ||||||||||
| International Inc. | Yongye | (decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Branded Stores | 775 | 200 | 288 | % | ||||||||
| Branded Stores in Trials | 350 | 0 | N/A | |||||||||
| Totals | 1,125 | 200 | 463 | % | ||||||||
Our number of branded stores grew to 1,125 in the period ended December 31, 2008 from 200 in the same period ended December 31, 2007 which was an increase of 463%.
36
Sales by Product Line
| Yongye Biotechnology | Predecessor | |||||||||||||||||||||||
| International Inc. | Inner Mongolia Yongye | |||||||||||||||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||||||||||||||
Units Shipped | Total sales | % of Total Sales | Units Shipped | Total sales | % of Total Sales | |||||||||||||||||||
| Plant | 427,196 | 44,842,062 | 93 | % | 107,269 | 11,147,125 | 85 | % | ||||||||||||||||
| Animals | 98,058 | 3,250,209 | 7 | % | 55,305 | 1,990,282 | 15 | % | ||||||||||||||||
| Total | 525,254 | 48,092,271 | 100 | % | 162,574 | 13,137,407 | 100 | % | ||||||||||||||||
Sales of plant product increased 298% to 427,196 units for the year ended December 31, 2008 from 107,269 units in the same period ended December 31, 2007. Sales of animal product increased 77% to 98,058 units for the year ended December 31, 2008 from 55,305 units in the same period ended December 31, 2007.
Customers
| Yongye Biotechnology | Predecessor | |||||||||||||||
| International Inc. | Inner Mongolia Yongye | |||||||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||||||
| % of Sales | Sales | % of Sales | Sales | |||||||||||||
| 5 Major Customers | 92 | % | $ | 44,109,813 | 83 | % | $ | 10,924,986 | ||||||||
| 3 Major Customers | 70 | % | $ | 33,718,961 | 67 | % | $ | 8,810,806 | ||||||||
| 1 Major Customer | 43 | % | $ | 20,541,267 | 29 | % | $ | 3,853,891 | ||||||||
Five major customers accounted for 92% and one major customer accounted for 43% of the Company’s net revenue for the twelve months period ended December 31, 2008. Five major customers accounted for 83% and one major customer accounted for 29% of the Predecessor’s net revenue for the twelve months period ended December 31, 2007. The Company and the Predecessor’s total sales to five major customers were $44,109,813 and $10,924,986, for the twelve months period ended December 31, 2008 and 2007, respectively.
Cost of Goods Sold
| Predecessor | ||||||||||||
| Yongye Biotechnology | Inner Mongolia | Increase | ||||||||||
| International Inc. | Yongye | /(decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Cost of Sales | $ | 23,165,684 | $ | 7,274,710 | 218 | % | ||||||
| Percentage of Sales | 48 | % | 55 | % | (7 | )% | ||||||
Cost of goods sold for the year ended December 31, 2008 was $23,165,684 which is 48% of revenues. This is an increase of $15,890,974 over the previous period which represents a 218% increase overall. However, as a percent of revenue, this represented an overall decrease of 7% when compared with the corresponding period in 2007 which was 55%. The overall decrease in cost of goods sold as a percentage was primarily due to the significant increase of sales and the fixed costs allocated to each unit of product throughout the twelve months ended December 31, 2008.
The major reason for the drop in cost by percentage is the change in business models between the two periods of 2008 and 2007. In year 2007, Inner Mongolia Yongye simply bought raw materials and produced them into finished goods at relatively small levels of production. This led to lower margins due to lower economies of scale and lower volume discounts on raw materials.
37
However, in 2008, Inner Mongolia Yongye became a contract manufacturer to Yongye Nongfeng which purchased 100% of the finished goods produced and did so at approximately 223% increase over 2007 with 525,254 units being sold in 2008 as opposed to 162,574 units in 2007. Based on this projected high volume of purchases, Inner Mongolia Yongye offered Yongye Nongfeng a fixed rate contract for finished goods for 2008. This helped Inner Mongolia Yongye reduce its fixed costs allocated to each unit of product which increased its overall margins and this was passed on to Yongye Nongfeng via the fixed rate contract. This helped increase the margin to 52%.
Selling, General & Administrative Expenses
| Yongye | Predecessor | |||||||||||
| Biotechnology | Inner Mongolia | Increase / | ||||||||||
| International Inc. | Yongye | (decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Selling, General and Administrative | $ | 11,238,772 | $ | 925,996 | 1114 | % | ||||||
| Percentage of Sales | 23 | % | 7 | % | 16 | % | ||||||
Selling, general and administrative (“SG&A”) expenses for the year ended December 31, 2008 was $11,238,772, an overall increase of $10,312,776 or 1114% when compared with the corresponding period in 2007. The increase in selling, general and administrative expenses was primarily due to increased sales activities in a larger number of provinces, increased staffing at the administrative level, executive salaries, advertising activities and an increase in allowance for doubtful accounts. Much of this is due to the cost of being a public company. However, while we increased revenues by 266%, we only increased spending on SG&A by 16% as a percentage of revenue.
Enterprise Income Taxes
The Company did not carry on any business and did not maintain any branch office in the United States during the years ended December 31, 2008 and 2007 and does not intend to repatriate any earnings from the Chinese operations. Therefore, no provision for withholding or U.S. federal income taxes or tax benefits on the undistributed earnings and/or losses of the Company has been made.
Yongye Nongfeng is subject to PRC Enterprise Income Tax at a rate of 25% of net income from its foundation on January 4, 2008 to March 31, 2008, and 1.25% of gross revenue since April 1, 2008. Since the predecessor is located in the economic development area in Inner Mongolia Autonomous Region, the predecessor is exempt from income tax according to the tax law in China.
For the year ended December 31, 2008, the Company’s income tax expense was $864,292 and income taxes payable was $219,366 as of December 31, 2008 as compared to $0 and $0 for the same period in 2007.
Net Income
| Predecessor | ||||||||||||
| Yongye Biotechnology | Inner Mongolia | Increase / | ||||||||||
| International Inc. | Yongye | (decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Net Income | $ | 11,191,961 | $ | 4,358,554 | 157 | % | ||||||
| Net Margins | 23 | % | 33 | % | (10 | )% | ||||||
| EPS- Basic | $ | 0.66 | $ | 0.38 | 74 | % | ||||||
| EPS- Diluted | $ | 0.64 | $ | 0.38 | 67 | % | ||||||
38
Net income for the period ended December 31, 2008 increased by $6,833,407 to $11,191,961 from $4,358,554 in the same period ended December 31, 2007, which is a 157% increase. However, this also represented a decrease in the overall percentage of net income to sales by 10% going from 33% in the period ended December 31, 2007 to 23% in December 31, 2008. This was primarily due to increased SG&A over the period as described above.
Basic and diluted earnings per share (EPS) for the year ended December 31, 2008, were $0.66 and $0.64, respectively, compared to $0.38 and $0.38, in the same period last year. The weighted average shares outstanding used to calculate basic and diluted EPS for the comparative periods were 16.9 million and 17.5 million for 2008 and 11.4 million and 11.4 million for year 2007, respectively.
Foreign Currency Translation Gains
The reporting currency of the Company is the US dollar. We use our local currency, Renminbi (RMB), as our functional currency. Results of operations and cash flows are translated at average exchange rates during the period, and assets and liabilities are translated at the unified exchange rate at the end of the period. Translation adjustments resulting from this process are included in accumulated other comprehensive income in the statement of shareholders’ equity. Transaction gains and losses that arise from exchange rate fluctuations on transactions denominated in a currency other than the functional currency are included in the results of operations as incurred.
The value of the USD versus the RMB continued to decline during the fourth quarter of 2008. As a result of the appreciation of the RMB, we recognized a foreign currency translation gain of $331,100 for the year ended December 31, 2008 compared to a gain of $723,298 for the same period in 2007. Given the uncertainty of exchange rate fluctuations, we cannot estimate the effect of these fluctuations on our future business, product pricing, and results of operations or financial condition. All of our revenues and expenses were denominated in RMB Yuan. The income statement accounts were translated at 1 RMB Yuan to 0.1459 USD and balance sheet amounts were translated at 1 RMB Yuan to 0.1436 USD.
Liquidity and Capital Resources
Inner Mongolia Yongye, the predecessor company, has historically financed its operations and capital expenditures principally through shareholder loans, bank loans, and cash provided by operations. As a newly formed Cooperative Joint Venture, Yongye Nongfeng, has used the net proceeds of both the April and September Offerings of approximately $20 million to finance the purchase of raw materials and finished inventory from Inner Mongolia Yongye, capital equipment and an expansion of our facilities and production, build out of our distribution network and increasing the number of our branded stores.
As is customary in the industry, we provide payment terms to most of our distributors which typically exceed the terms that we ourselves receive from our finished goods suppliers. Therefore, the Company’s liquidity needs have generally consisted of working capital necessary to finance receivables and raw material inventory. We believe that over the next 12 months our existing cash, cash equivalents and cash flows from operations will be sufficient to meet our anticipated future cash needs. We may, however, require additional cash resources due to changing business conditions or other future developments, including any investments or acquisitions we may decide to pursue. We will determine how to meet these specific cash flow needs as they arise. Therefore, there can be no assurance that such additional investment will be available to us, or if available, that it will be available on terms acceptable to us. Cash and Cash Equivalents balance amounted to $4,477,477 and $376,002 as of December 31, 2008 and December 31, 2007, respectively.
39
Financial Cash Flow Highlights for Fiscal Years Ended
Yongye Biotechnology | Predecessor | |||||||||||
International Inc. | Inner Mongolia Yongye | Increase / (decrease) | ||||||||||
| YEAR 2008 | YEAR 2007 | |||||||||||
| Net cash used in operating activities | $ | (8,666,893 | ) | $ | (4,618,338 | ) | 73 | % | ||||
| Net cash used in investing activities | $ | (5,475,572 | ) | $ | (309,221 | ) | 1705 | % | ||||
| Net cash provided by financing activities | $ | 18,286,765 | $ | 5,197,237 | 240 | % | ||||||
| Effect of exchange rate change on cash and cash equivalents | $ | 325,041 | $ | 17,301 | 1814 | % | ||||||
| Net increase in cash and cash equivalents | $ | 4,469,341 | $ | 286,979 | 1457 | % | ||||||
| Cash and cash equivalents at beginning of period | $ | 8,136 | $ | 89,023 | (91 | )% | ||||||
| Cash and cash equivalents at end of period | $ | 4,477,477 | $ | 376,002 | 1091 | % | ||||||
The Company’s working capital at December 31, 2008, increased $22,891,033 to $25,546,823 from $2,655,790 on December 31, 2007. The increase in working capital by 862% over the same period in 2007 resulted primarily from the April and September financings, increased receipts of accounts receivables during the fourth quarter, increased build up of inventory in preparation for shipments beginning in Q1 of 2009, and the substantially different business model of the CJV which carries very little current liabilities.
Accounts receivable Days Sales Outstanding for the fiscal year ended December 31, 2008 decreased 12 days to 18 days at December 31, 2008 from 30 days at December 31, 2007 and Days Sales in Inventory decreased by 55 days to 242 days at December 31, 2008 from 297 days at December 31, 2007. As is customary in China’s agriculture industry, we give credit terms which allow our distributors to pay over a longer period of time than is traditionally done in other industries.
For the fiscal year ended December 31, 2008, net cash used in operating activities increased $4,048,555 to $8,666,893 from $4,618,338 for the year ended December 31, 2007. Reductions in cash were primarily attributable to growth in the business which required larger finished goods purchases and resulted in a large inventory balance of $20,708,193 at the end of 2008 which is needed for peak season sales in 2009. Also, while we significantly reduced our accounts receivable balance from Q3 to Q4, this was still $3,053,380 by year end December 31, 2008.
For the fiscal year ended December 31, 2008, net cash used in investing activities increased $5,166,351 to $5,475,572 from $309,221 in the same period in 2007 and was primarily attributable to acquisitions of plant, property and equipment of $5,475,572.
For the fiscal year ended December 31, 2008, gross cash from financing activities increased $14,639,056 to $19,848,424 from $5,209,368 year ended 2007. This resulted from our offerings received on April 17, 2008 and September 8, 2008 and bank loans for property and equipment. This was offset by expenses incurred in issuance of stock to shareholders of $1,461,659 resulting in net cash of $18,286,765. Net cash from financing activities increased $ 13,089,528 to $ 18,286,765 year ended 2008 from $ 5,197,237 year ended 2007.
Impact of inflation
We are subject to commodity price risks arising from price fluctuations in the market prices of the raw materials. We have generally been able to pass on cost increases through price adjustments. However, the ability to pass on these increases depends on market conditions influenced by the overall economic conditions in China. We manage our price risks through productivity improvements and cost-containment measures. We do not believe that inflation risk is material to our business or our financial position, results of operations or cash flows.
40
Off Balance Sheet Arrangements
We do not have any off-balance sheet arrangements as defined by standards issued by the Financial Accounting Standards Board, and accordingly, no such arrangements are likely to have a current or future effect on our financial position, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources.
Critical Accounting Policies
The preparation of financial statements and related disclosures in conformity with accounting principles generally accepted in the United States requires management to make judgments, assumptions, and estimates that affect the amounts reported in the consolidated financial statements and accompanying notes. The second footnote to the Company’s financial statements (Summary of Significant Accounting Policies) describes the major accounting policies and methods used in the preparation of the financial statements.
The following are considered to be the Company’s crucial accounting policies
Revenue recognition
The Company’s revenue recognition policies are in compliance with Staff Accounting Bulletin (“SAB”) 104. Sales revenue is recognized at the date of shipment to customers when a formal arrangement exists, the price is fixed or determinable, the delivery is completed, no other significant obligations of the Company exist and collectability is reasonably assured. Payments received before all of the relevant criteria for revenue recognition are recorded as customer deposits. Sales revenue represents the invoiced value of goods, net of value-added tax (VAT). The Company’s animal products sold in PRC are subject to a Chinese value-added tax at a rate of 4% of the gross sales price in 2008 because Yongye Nongfeng is a small-scale tax payer in PRC. This VAT cannot be offset by VAT paid by the Company on raw materials and other materials included in the cost of the finished product. The Company’s plant products sold in PRC are exempted from any VAT.
Use of estimates
The preparation of financial statements in conformity with generally accepted accounting principles of the United States of America requires management to make estimates and assumptions that affect the amounts reported in the financial statements and accompanying notes. Significant accounting estimates reflected in the Company’s financial statements include the useful lives of and impairment for property, plant and equipment, and potential losses on uncollectible receivables. Actual results could differ from these estimates.
Inventory costing
Inventory is stated at the lower of cost, which takes into account historical prices on a continuing basis, or market. Cost is determined by the weighted average method. Provision for diminution in value on inventories is made using specific identification method.
41
| ITEM 7A. | Quantitative and Qualitative Disclosures about Market Risk |
Interest Rate Risk
We are exposed to interest rate risk due primarily to our short-term bank loans for the acquisition of automobiles. Although the interest rates are fixed for the terms of the loans, the terms are typically twelve months and interest rates are subject to change upon renewal. Since July 20, 2007, the People’s Bank of China has increased the interest rate of Renminbi bank loans with a term of six months or less by 0.2% and loans with a term of six to 12 months by 0.3%. The new interest rates are approximately 6.0% and 6.8% for Renminbi bank loans with a term six months or less and loans with a term of six to 12 months, respectively. The change in interest rates has no impact on our bank loans secured before July 28, 2007. We monitor interest rates in conjunction with our cash requirements to determine the appropriate level of debt balances relative to other sources of funds. We have not entered into any hedging transactions in an effort to reduce our exposure to interest rate risk.
Credit Risk
The Company is exposed to credit risk from its cash in bank and fixed deposits and bills and accounts receivable. The credit risk on cash in bank and fixed deposits is limited because the counterparties are recognized financial institutions. Accounts receivable are subjected to credit evaluations. An allowance has been made for estimated irrecoverable amounts which have been determined by reference to past default experience and the current economic environment.
Foreign Exchange Risk
The value of the Renminbi against the U.S. dollar and other currencies is affected by, among other things, changes in China’s political and economic conditions. Since July 2005, the Renminbi has no longer been pegged to the U.S. Dollar at a constant exchange rate. Although the People’s Bank of China regularly intervenes in the foreign exchange market to prevent significant short-term fluctuations in the exchange rate, the Renminbi may appreciate or depreciate within a flexible peg range against the U.S. dollar in the medium to long term. Moreover, it is possible that in the future, PRC authorities may lift restrictions on fluctuations in the Renminbi exchange rate and lessen intervention in the foreign exchange market.
Because substantially all of our earnings and cash assets are denominated in Renminbi, but our reporting currency is the U.S. dollar, fluctuations in the exchange rate between the U.S. dollar and the Renminbi will affect our balance sheet, income statement and our earnings per share in U.S. dollars. In addition, appreciation or depreciation in the value of the Renminbi relative to the U.S. dollar would affect our financial results reported in U.S. dollar terms without giving effect to any underlying change in our business or results of operations. Fluctuations in the exchange rate will also affect the relative value of any dividend we issue in the future that will be exchanged into U.S. dollars and earnings from, and the value of, any U.S. dollar-denominated investments we make in the future.
Very limited hedging transactions are available in China to reduce our exposure to exchange rate fluctuations. To date, we have not entered into any hedging transactions in an effort to reduce our exposure to foreign currency exchange risk. While we may enter into hedging transactions in the future, the availability and effectiveness of these transactions may be limited, and we may not be able to successfully hedge our exposure at all. In addition, our foreign currency exchange losses may be magnified by PRC exchange control regulations that restrict our ability to convert Renminbi into foreign currencies.
Most of the transactions of the Company are settled in Renminbi and U.S. dollars. In the opinion of the directors, the Company is not exposed to significant foreign currency risk.
Inflation
Inflationary factors, such as increases in the cost of the finished goods we buy could impair our operating results. Although we do not believe that inflation has had a material impact on our financial position or results of operations to date, a high rate of inflation in the future may have an adverse effect on our ability to maintain current levels of gross margin and selling, general and administrative expenses as a percentage of sales revenue if the selling prices of our products do not increase with these increased costs.
42
Company’s Operations are Substantially in Foreign Countries
Substantially all of our operations are conducted in China and are subject to various political, economic, and other risks and uncertainties inherent in conducting business in China. Among other risks, the Company and its subsidiaries’ operations are subject to the risks of restrictions on transfer of funds; export duties, quotas, and embargoes; domestic and international customs and tariffs; changing taxation policies; foreign exchange restrictions; and political conditions and governmental regulations. Additional information regarding such risks can be found under the heading “Risk Factors” in this Form 10-K.
| ITEM 8 | Financial Statements and Supplementary Data |
Consolidated Financial Statements
The information required by Item 8 appears after the signature page to this report.
| ITEM 9 | Changes in and Disagreements With Accountants on Accounting and Financial Disclosures |
None.
ITEM 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Disclosure Controls and Procedures
The Company maintains disclosure controls and procedures that are designed to ensure that information required to be disclosed in reports filed by the Company under the Securities Exchange Act of 1934, as amended, is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and regulations and that such information is accumulated and communicated to our management, including its Chief Executive Officer and Interim Principal Financial Officer, as appropriate, to allow for timely decisions regarding required disclosure. Our Chief Executive Officer and Interim Principal Financial Officer evaluated, with the participation of other members of management, the effectiveness of our disclosure controls and procedures (as defined in Exchange Act Rule 15d-15(e)), as of the end of the period covered by this Annual Report on Form 10-K. Based on this evaluation, our Chief Executive Officer and Interim Chief Financial Officer concluded that the Company’s disclosure controls and procedures were effective.
Although the management of our Company, including the Chief Executive Officer and the Interim Principal Financial Officer, believes that our disclosure controls and internal controls currently provide reasonable assurance that our desired control objectives have been met, management does not expect that our disclosure controls or internal controls will prevent all error and all fraud. A control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, within our Company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the controls. The design of any system of controls is also based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions.
43
Management’s Report on Internal Control over Financial Reporting
The management of the Company is responsible for establishing and maintaining adequate internal control over financial reporting as such term is defined in Exchange Act Rules 13a-15(f) and 15d-15(f). Our internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States of America.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Under the supervision and with the participation of the Chief Executive Officer and Interim Principal Financial Officer, management conducted an evaluation of the effectiveness of its internal control over financial reporting as of December 31, 2008. The framework on which such evaluation was based is contained in the report entitled “Internal Control — Integrated Framework” issued by the Committee of Sponsoring Organizations of the Treadway Commission (the “COSO Report”). Based on that evaluation and the criteria set forth in the COSO Report, management concluded that its internal control over financial reporting was effective as of December 31, 2008.
This annual report on Form 10-K does not include an attestation report of our registered independent public accounting firm regarding management’s assessment of the Company’s internal control over financial reporting. Management’s report was not subject to audit by our registered independent public accounting firm pursuant to rules of the SEC that permit us to provide only management’s report in this annual report.
Changes in Internal Controls over Financial Reporting
There were no significant changes in our internal controls over financial reporting identified in connection with this evaluation that occurred during our last fiscal quarter that have materially affected, or are reasonably likely to materially affect, the Company’s internal controls over financial reporting.
ITEM 9B. Other Information.
There is no information required to be disclosed in a report on Form 8-K during the fourth quarter of the year covered by this Form 10-K but not reported.
44
PART III
| ITEM 10 | Directors, Executive Officers and Corporate Governance |
Directors and Executive Officers
Set forth below is information regarding our current directors and executive officers as of the date of this report:
Name | Age | Position | ||
| Zishen Wu | 40 | Chief Executive Officer, President and Chairman | ||
| Sun Taoran | 39 | Vice Chairman | ||
| Zhao Qiang | 43 | VP Sales and Marketing and Director | ||
| Larry Gilmore | 46 | Interim Principal Financial Officer and VP Corporate Strategy | ||
| Zhang Haiming | 48 | Independent Director | ||
| Guo Xiaochuan | 43 | Independent Director |
Zishen Wu, Chief Executive Officer, President and Chairman
Mr. Wu is CEO and Chairman of the Board of Directors of Yongye Nongfeng and Yongye Biotechnology International, Inc. Mr. Wu began his career as official at the State Planning Department in Inner Mongolia from 1984 to 1988. From 1989 to 2000, Mr. Wu had been appointed in various managerial positions from General Manager to Chairman of several State Owned Conglomerates in textile, diary and agriculture industries. In 2001, Mr. Wu founded Yongye Technology Company in Inner Mongolia to distribute consumer electronics with annual sales of 80 million RMB. In 2003 Mr. Wu founded Yongye Organism Technology Company to product plant and animal nutrients. Mr. Wu currently is the deputy director for the Inner Mongolia Charmer of Commerce and a member of executive committee for industry and commerce association in Inner Mongolia.
Sun Taoran, Vice Chairman
Mr. Sun joined Yongye Biotechnology International as the Vice Chairman in 2008. Mr. Sun has over 16 years experience with leading media and marketing companies in China. In 1991, Mr. Sun co-founded a media company with several well known publications. He was managing one of the key managers and share holders in the media company until 2001. In 1998, Mr. Sun co-founded High Tech Wealth (HTW), a Chinese well known branded Personal Digital Assistant (PDA). Mr. Sun was the General Manager of HTW until 2002. In 2003, Mr. Sun also founded and served as chairman of an electronic payment system company. Mr. Sun received his B.A. in Economic from Beijing University in 1991.
Mr. Zhao Qiang, VP Sales and Marketing and Director
Mr. Zhao is a well known marketing professional and recently published his first book entitled, “Learn Marketing from Chairman Mao.” Prior to joining the Company as the Chief Marketing Officer and director in April 2008, Mr. Zhao had over 16 years of marketing experiences where he worked for famous consumer product companies as Head of Marketing. Mr. Zhao was Senior Vice President of Marketing for Guangdong Galanz Group, a famous household appliance company. Prior to Galanz Group Mr. Zhao was President of Gracewell, a well known undergarment brand in China, and Senior Vice President of Marketing at Meijin, a famous PDA manufacture. Mr. Zhao received his B.A. in Journalism from China Media University, and his MBA from Cheung Kong Graduate School of Business.
45
Larry Gilmore, Interim Principal Financial Officer and VP Corporate Strategy
Prior to joining the Company, Mr. Gilmore was SVP of operations for Asia Standard Energy from 2005 to 2007 and had the responsibility for raising private equity for PRC small to medium enterprises and renewable energy projects and corporate oversight on finance and accounting. Mr. Gilmore served as Managing Director of GC Global from 2001 to 2004 and assisted large organizations in major scale change initiatives. Prior to this he was the Manager of Human Resources at Alcatel and Senior Consultant at Deloitte and Touche. As a result of the resignation of Ms. Vini Dharmawan on July 23, 2008, Mr. Gilmore was appointed by the Board of Directors to serve as Interim Principal Financial Officer until a permanent replacement for this position has been identified.
Dr. Zhang Haiming, Independent Director
Dr. Zhang received his B.S. and M.S. in Genetic Breeding from Inner Mongolia Agricultural University, and his PhD at China Agricultural University. Dr. Zhang went to the University of Tokyo and Tokyo University of Agriculture and Technology as Senior Visiting Scholar in 1995 and 2002. He has over 10 years of management experience in agricultural and animal husbandry industry where he worked for several academic colleges and departments at Beijing University of Agriculture and Inner Mongolia Agricultural University. He joined some international projects as Chief Chinese Scientist supported by Asian Development Bank and UNIDO. Dr. Zhang has published over 40 papers, been responsible for 6 projects with funding over RMB6M.
Guo Xiaochuan, Independent Director
Professor Guo Xiaochuan joined the Company as an Independent Director. Professor Guo received his B.S., M.S. and PhD in management science in Fudan University. He is currently the Dean of College of Economic & Management and Director of MBA Center of Inner Mongolia University. Professor Guo worked as lecturer for Inner Mongolia University from 1988 to 1992. Professor Guo was the founder of the MBA program at Inner Mongolia University. Professor Guo serves as an Independent Director of Inner Mongolia PingZhuang Energy Resource Co., Ltd. and an Independent Director of Inner Mongolia Yili Industrial Group Co., Ltd. He has served as director and Independent Directors in several enterprises, such as Inner Mongolia Shunxin Ningcheng Laojiao Co., Ltd., Inner Mongolia Rixin Group, Rising Securities and Baotou Aluminum (Group) Co., Ltd., etc.
Family Relationships
There are no family relationships among our directors or officers.
Section 16(A) Beneficial Ownership Reporting Compliance
Under U.S. securities laws, directors, certain executive officers and persons holding more than 10% of our common stock must report their initial ownership of the common stock, and any changes in that ownership, to the SEC. The SEC has designated specific due dates for these reports. Based solely on our review of copies of such reports filed with the SEC and written representations of our directors and executive offers, we believe that all persons subject to reporting filed the required reports on time in 2008.
Code of Ethics
We have not adopted a code of business conduct and ethics relating to the conduct of our business by our employees, officers and directors.
Board Composition and Meetings of the Board of Directors
The Board of Directors is currently composed of five members. All actions of the Board of Directors require the approval of a majority of the directors in attendance at a meeting at which a quorum is present. Through December 31, 2008, our Board of Directors met in person 0 times and acted by written consent 1 time.
46
Committees
We do not have a standing nominating or compensation committee or any committee performing a similar function, although we may form such committees in the near future.
Audit Committee and Audit Committee Financial Expert
Our Audit Committee is currently composed of Messrs. Zhang Haiming and Guo Xiaochuan, each of which is an “independent director” as defined by Rule 4200(a)(15) of the Marketplace Rules of The Nasdaq Stock Market, Inc. Mr. Guo Xiaochuan acts as chairman of the Audit Committee. We do not currently have a member of the Audit Committee who qualifies as an “audit committee financial expert” as defined in Item 401(h) of Regulation S-K. Our Board of Directors is in the process of searching for a suitable candidate for this position.
Independent Directors
Our Board of Directors is currently composed of five members. Messrs. Zhang Haiming and Guo Xiaochuan serve on our board of directors as an “independent director” as noted above.
Policy Regarding Board Attendance.
Our directors are expected to attend Board meetings as frequently as necessary to properly discharge their responsibilities and to spend the time needed to prepare for each such meeting. Our directors are expected to attend annual meetings of stockholders, but we do not have a formal policy requiring them to do so.
Shareholder Communications
The Company has a process for shareholders who wish to communicate with the Board of Directors. Shareholders who wish to communicate with the Board may write to it at the Company’s address given above. These communications will be reviewed by one or more employees of the Company designated by the Board, who will determine whether they should be presented to the Board. The purpose of this screening is to allow the Board to avoid having to consider irrelevant or inappropriate communications.
| ITEM 11 | Executive Compensation |
Compensation of Officers
The Company’s executive compensation program for the named executive officers (NEOs) is administered by the Board of Directors.
Background and Compensation Philosophy
Our Board of Directors has not adopted or established a formal policy or procedure for determining the amount of compensation paid to our executive officers. No pre-established, objective performance goals or metrics have been used by the Board of Directors in determining the compensation of our executive officers.
As our executive leadership and Board of Directors grows, our Board of Directors may decide to form a compensation committee charged with the oversight of executive compensation plans, policies and programs, but we have no current plans to establish a compensation committee.
47
Elements of Compensation
Some of our executive officers receive a base salary to compensate them for services rendered during the year. Our policy of compensating our certain executives with a cash salary has served the Company well. Because of our history of attracting and retaining executive talent, we do not believe it is necessary at this time to provide our executives discretionary bonuses, equity incentives, or other benefits for the Company to continue to be successful.
Base Salary and Bonus. The value of base salary and bonus for each our executive reflects his skill set and the market value of that skill set in the sole discretion of the Board of Directors.
Equity Incentives. The Company and its subsidiaries have not established an equity based incentive program and have not granted stock based awards as a component of compensation. In the future, we may adopt and establish an equity incentive plan pursuant to which awards may be granted if our Board of Directors determines that it is in the best interest of the Company and its stockholders to do so.
Retirement Benefits. Our executive officers are not presently entitled to company-sponsored retirement benefits.
Perquisites. We have not provided our executive officers with any material perquisites and other personal benefits and, therefore, we do not view perquisites as a significant or necessary element of our executive’s compensation.
Deferred Compensation. We do not provide our executives the opportunity to defer receipt of annual compensation.
Summary Compensation Table
The following table sets forth all cash compensation paid by the Company, as well as certain other compensation paid or accrued, for each of the last two fiscal years of our company to each named executive officers.
Summary Compensation of Named Executive Officers
Name and Principal Position | Fiscal Year | Salary ($) | Bonus ($) | Stock Awards ($) | Nonequity Incentive Plan Compensation ($) | Nonqualified Deferred Compensation Earnings ($) | All Other Compensation ($) | Total ($) | ||||||||||||||||||||||
| Zishen Wu- Chief Executive Officer, President and Chairman | 2008 | 171,233 | 17,123 | – | – | – | – | 188,356 | ||||||||||||||||||||||
| 2007 | 14,623 | – | – | – | – | – | 14,623 | |||||||||||||||||||||||
| Larry Gilmore, Principal Financial Officer & VP Corporate Strategy | 2008 | 62,500 | 4,566 | 67,066 | ||||||||||||||||||||||||||
| Zhao Qiang, VP Sales & Marketing | 2008 | 114,155 | 11,416 | – | – | – | – | 125,571 | ||||||||||||||||||||||
| 2007 | – | – | – | – | – | – | – | |||||||||||||||||||||||
48
During each of the last two fiscal years, none of our other officers had salary and bonus greater than $100,000. In addition, our executive officers and/or their respective affiliates will be reimbursed by us for any out-of-pocket expenses incurred in connection with activities conducted on our behalf. There is no limit on the amount of these out-of-pocket expenses and there will be no review of the reasonableness of such expenses by anyone other than our Board of Directors, which includes persons who may seek reimbursement, or a court of competent jurisdiction if such reimbursement is challenged.
Director Compensation
Our non-independent directors are paid a stipend of RMB 25,000 per month or RMB 300,000 annually and also reimbursed for expenses incurred by them in connection with attending Board of Directors’ meetings, and may also participate in the Company’s incentive compensation program. Our Independent Directors receive an annual stipend of $13,700 per year plus reimbursement of travel related fees.
Bonuses and Deferred Compensation
We do not have any bonus, deferred compensation or retirement plan. We do not have a compensation committee. All decisions regarding compensation are determined by our full Board of Directors.
Options and Stock Appreciation Rights
We do not currently have a stock option or other equity incentive plan. We may adopt one or more such programs in the future.
Employment Contracts
We have entered into an employment agreement with each of Wu Zishen, Zhao Qiang and Larry Gilmore. The CJV entered into an employment contract on April 17, 2008 with Mr. Wu Zishen to employ him as its Chairman and CEO. Mr. Wu is entitled to an annual gross salary in an amount of $205,479 including all allowances, social insurance and housing fund. This also includes a stipend of RMB 25,000 per month for board activities. He may also pay premiums for social security insurance schemes such as pension, unemployment, medical insurance and other social insurance coverage in accordance with relevant PRC laws and regulations. The CJV has a right to adjust the salary and welfare benefits of Mr. Wu appropriately based on his capability, experience, attitude, performance, achievement, working-age and position as well as its salary and position adjustment policies and business conditions experienced. Either party to the agreement has a right to terminate the agreement, subject to the terms and conditions therein.
The CJV entered into an employment contract on April 17, 2008 with Mr. Zhao Qiang to employ him as its VP Sales & Marketing. Mr. Zhao is entitled to an annual gross salary in an amount of $136,986 including all allowances, social insurance and housing fund. This also includes a stipend of RMB 25,000 per month for board activities. He also pays premiums for social security insurance schemes such as pension, unemployment, medical insurance and other social insurance coverage in accordance with relevant PRC laws and regulations. The CJV has a right to adjust the salary and welfare benefits of Mr. Zhao appropriately based on his capability, experience, attitude, performance, achievement, working-age and position as well as its salary and position adjustment policies and business conditions experienced. Either party to the agreement has a right to terminate the agreement, subject to the terms and conditions therein.
49
The CJV entered into an employment contract on April 17, 2008 with Mr. Larry Gilmore to employ him as its VP Corporate Strategy and was later amended to include the role of interim Principal Financial Officer. Mr. Gilmore’ annual gross salary for 2008 amounted to $62,500 including all allowances in accordance with relevant PRC laws and regulations. The CJV has a right to adjust the salary and welfare benefits of Mr. Gilmore appropriately based on his capability, experience, attitude, performance, achievement, working-age and position as well as its salary and position adjustment policies and business conditions experienced. Either party to the agreement has a right to terminate the agreement, subject to the terms and conditions therein.
Payment of Post-Termination Compensation
The Company does not have change-in-control agreements with any of its executive officers, and the Company is not obligated to pay severance or other enhanced benefits to executive officers upon termination of their employment.
Indemnification of Directors and Officers
Our officers and directors are indemnified as provided by the Nevada Revised Statutes and our bylaws.
Under the Nevada Revised Statutes, director immunity from liability to a company or its shareholders for monetary liabilities applies automatically unless it is specifically limited by a company’s articles of incorporation that is not the case with our articles of incorporation. Excepted from that immunity are:
(1) a willful failure to deal fairly with the company or its shareholders in connection with a matter in which the director has a material conflict of interest;
(2) a violation of criminal law (unless the director had reasonable cause to believe that his or her conduct was lawful or no reasonable cause to believe that his or her conduct was unlawful);
(3) a transaction from which the director derived an improper personal profit; and
(4) willful misconduct.
Our bylaws provide that we will indemnify our directors and officers to the fullest extent not prohibited by Nevada law; provided, however, that we may modify the extent of such indemnification by individual contracts with our directors and officers; and, provided, further, that we shall not be required to indemnify any director or officer in connection with any proceeding (or part thereof) initiated by such person unless:
(1) such indemnification is expressly required to be made by law;
(2) the proceeding was authorized by our Board of Directors;
(3) such indemnification is provided by us, in our sole discretion, pursuant to the powers vested us under Nevada law; or
(4) such indemnification is required to be made pursuant to the bylaws.
Our bylaws provide that we will advance all expenses incurred to any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, by reason of the fact that he is or was our director or officer, or is or was serving at our request as a director or executive officer of another company, partnership, joint venture, trust or other enterprise, prior to the final disposition of the proceeding, promptly following request. This advanced of expenses is to be made upon receipt of an undertaking by or on behalf of such person to repay said amounts should it be ultimately determined that the person was not entitled to be indemnified under our bylaws or otherwise.
50
Our bylaws also provide that no advance shall be made by us to any officer in any action, suit or proceeding, whether civil, criminal, administrative or investigative, if a determination is reasonably and promptly made: (a) by the board of directors by a majority vote of a quorum consisting of directors who were not parties to the proceeding; or (b) if such quorum is not obtainable, or, even if obtainable, a quorum of disinterested directors so directs, by independent legal counsel in a written opinion, that the facts known to the decision- making party at the time such determination is made demonstrate clearly and convincingly that such person acted in bad faith or in a manner that such person did not believe to be in or not opposed to our best interests.
| ITEM 12 | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters. |
The following table sets forth certain information regarding beneficial ownership of our Common Stock as of December 31, 2008 by (i) each person (or group of affiliated persons) who is known by us to own more than five percent (5%) of the outstanding shares of our Common Stock, (ii) each director and named executive officer, and (iii) all of our directors and executive officers as a group. As of December 31, 2008, we had 26,760,258 shares of Common Stock outstanding.
Beneficial ownership is determined in accordance with SEC rules and generally includes voting or investment power with respect to securities. Unless otherwise noted, the principal address of each of the stockholders, directors and officers listed below is 6th Floor Xue Yuan International Tower, No. 1 Zhichun Road, Haidian District, Beijing, PRC.
Unless otherwise indicated, all share ownership figures include shares of our Common Stock issuable upon securities convertible or exchangeable into shares of our Common Stock within sixty (60) days of March 13, 2009, which are deemed outstanding and beneficially owned by such person for purposes of computing his or her percentage ownership, but not for purposes of computing the percentage ownership of any other person.
Name and Address of Beneficial Owner | Amount and Nature of Beneficial Ownership | Percentage of Outstanding Shares of Common Stock | ||||||
| Full Alliance International Limited (1) | 6,748,107 | 25.2 | % | |||||
| Knight Bridge Group Limited (2) | 2,861,189 | 10.7 | % | |||||
| Ardsley Partners Fund II, LP (3) | 2,313,683 | 8.5 | % | |||||
| Ardsley Offshore Fund, Ltd. (3) | 900,000 | 3.34 | % | |||||
| Ardsley Partners Institutional Fund, LP (3) | 1,517,500 | 5.61 | % | |||||
| Philip J. Hempleman (3) | 5,196,683 | 18.69 | % | |||||
| Ardsley Partners I (3) | 3,831,183 | 13.78 | % | |||||
| Ardsely Advisory Partners (3) | 4,790,433 | 17.23 | % | |||||
| Black River Small Capitalization Fund Ltd. (5) | 2,441,495 | 9.0 | % | |||||
| Black River Asset Management LLC | 2,684,665 | 9.9 | % | |||||
| Black River Commodity Select Fund Ltd. (5) | 771,304 | 2.9 | % | |||||
| Special Situations Private Equity Fund, L.P. (4) | 909,328 | 3.4 | % | |||||
| Special Situations Cayman Fund, L.P. (4) | 1,688,750 | 6.3 | % | |||||
| Zishen Wu | — | * | ||||||
| Sun Taoran | — | * | ||||||
| Zhao Qiang | — | * | ||||||
| Larry Gilmore | — | * | ||||||
| Zhang Haiming | — | * | ||||||
| Guo Xiaochuan | * | |||||||
| All Directors and Executive Officers, as a group | — | — | ||||||
51
* Less than one percent
(1) The business address of Full Alliance International Limited is OMC Chambers, P.O. Box 3152, Road Town, Tortola, British Virgin Islands. Full Alliance is wholly owned by Ms. Xingmei Zhang and such shares may be deemed to be beneficially owned by Ms. Zhang.
(2) The business address of Knight Bridge Group Limited is 20/F, Sunning Plaza, 10 Hysan Avenue, Causeway Bay, Hong Kong. Knight Bridge Group Limited is controlled by Mr. Dickson Kwok, who may be deemed to beneficially own such shares.
(3) These are all affiliated entities with a business address c/o Ardsley Partners, 262 Harbor Drive, 4th Floor, Stamford, CT 06902, except in the case of Ardsley Offshore Fund, Ltd., which has a business address of Romasco Place, Wickham’s Cay 1, Roadtown, Tortola, British Virgin Islands. Philip J. Hempleman has voting and investment control over securities held by Ardsley Partners Fund II, LP, Ardsley Partners Institutional Fund, LP, Marion Lynton, Ardsley Offshore Fund, Ltd., and the 1998 Hempleton Family Trust may be deemed to beneficially own such shares. This information has been derived from a Schedule 13G filed on February 13, 2009.
(4) These are affiliated entities with a business address c/o Special Situations Private Equity Fund, 527 Madison Avenue, Suite 2600, New York, NY 10022. Austin Marxe and David Greenhouse have voting and investment control over securities held by Special Situations Private Equity Fund, L.P. and Special Situations Cayman Fund, L.P. and may be deemed to beneficially own such shares. The amount of beneficial ownership set forth in the table above does not include a total of 649,520 April Warrants or the September Warrants. Based upon the terms of such Warrants the holders may not exercise such Warrants if after giving effect to the issuance of shares of Common Stock issuable upon exercise of any portion of such Warrants, on any such date the holders, together with their affiliates, would be deemed the beneficial owner of more than 4.99% of the then outstanding shares of our Common Stock; however, any such holder can elect to increase or decrease the cap upon 61 days’ prior notice to us, provided that the cap in no event exceeds 9.99%. This information has been derived from a Schedule 13G filed on October 10, 2008.
(5) The business address is c/o Black River Asset Management LLC, 12700 Whitewater Drive, Minnetonka, MN 55343-943 8. Black River Asset Management LLC has voting and investment control over securities held by Black River Small Capitalization Fund Ltd. and Black River Commodity Select Fund Ltd. and may be deemed to beneficially own such shares. The calculation of the amount of beneficial ownership as set forth in the table above includes the September Warrants. This information has been derived from a Schedule 13G filed on February 18, 2009.
Changes in Control
There are no arrangements known to us, including any pledge by any person of our securities, the operation of which may at a subsequent date result in a change in control of the Company.
52
| ITEM 13 | Certain Relationships and Related Transactions, and Director Independence |
Transactions with Related Persons
Based on CJV contractual terms, Inner Mongolia Yongye is a 0.5% owner of Yongye Nongfeng and Asia Standard Oil, Ltd. is a 99.5% owner in the CJV. However, based upon actual capital injection into the CJV, Inner Mongolia Yongye is a 0.6% owner of Yongye Nongfeng Biotechnology and Asia Standard Oil, Ltd. is a 99.4% owner. Mr. Zishen Wu controls Inner Mongolia Yongye and is the CEO of Yongye Nongfeng Biotechnology. He also sits on the boards of both companies as Chairman. During the years ended December 31, 2008 and 2007, Inner Mongolia Yongye entered into several intercompany loan transactions with affiliated entities, none of which have been assumed by Yongye Nongfeng Biotechnology. In addition, in January 2008, Yongye Nongfeng Biotechnology entered into a Cooperation Agreement with Inner Mongolia Yongye providing for the terms of contract manufacturing of nutrient product on terms disclosed elsewhere herein. In January 2008 Inner Mongolia Yongye and Yongye Nongfeng Biotechnology also entered into a Sales Agreement providing for the sale of existing nutrient inventory on terms disclosed elsewhere herein.
As of December 31, 2008, the predecessor is Yongye Nongfeng’s only vendor that provided $43,509,906 (100%) of the Company’s purchased finished goods for the year ended December 31, 2008. As of December 31, 2008, due from related party is $192,741 and represents the payment the Company made for the predecessor for its professional fees and research & development fee. According to the contract, the predecessor sells to Yongye Nongfeng at fixed prices of RMB 350 per case for plant products and RMB 120 per case for animal products.
During the year ended December 31, 2008, the Company borrowed $1,617,293 from Ms. Yin’s (Mr. Wu’s wife) company Inner Mongolia Chilechuan Culture Development Co., Ltd. The amounts are unsecured and non-interest bearing, and has been repaid before December 31, 2008.
Yongye Nongfeng and the predecessor entered lease-exchange arrangements to lease land, buildings and equipments to each other. On June 1, 2008, a land lease agreement was entered into in which Yongye Nongfeng would lease a land of 74,153 square meters from the predecessor from June 1, 2008 to May 31, 2009. On September 28, 2008, a building lease agreement and an equipment lease agreement were entered into in which the predecessor would lease a building of 3,967 square meters and a set of equipments from Yongye Nongfeng from September 28, 2008 to September 27, 2009. The estimated value of rentals of land lease and the combination of building and equipment lease are not materially different. Therefore, pursuant to the agreements, both Yongye Nongfeng and the predecessor would not charge any rental to each other for the lease.
As of December 31, 2007, the predecessor has borrowed $2,507,371 from stockholders. These loans are short term in nature, unsecured and non-interest bearing. Also, at December 31, 2007 the predecessor has $12,153 of long-term, unsecured and non-interest bearing loans from shareholders.
Other than the foregoing, there have been no transactions since January 1, 2007 or any currently proposed transaction, or series of transactions, to which the Company was or is to be a party in which the amount involved exceeds $120,000 and in which any current or former director or officer of the Company, any 5% or greater shareholder of the Company or any member of the immediate family of any such persons had or will have a direct or indirect material interest other than as disclosed below.
Policies and Procedures for Review, Approval or Ratification of Transactions with Related Persons
It is the Company’s policy that the Company will not enter into transactions required to be disclosed under item 404 of the SEC’s Regulation S-K unless the committee or another independent body of the board first reviews and approves the transactions.
Promoters and Certain Control Persons
We did not have any promoters at any time during the past five fiscal years. Except as set forth in our discussion above, none of our directors, director nominees or executive officers has been involved in any transactions with us or any of our directors, executive officers, affiliates or associates which are required to be disclosed pursuant to the rules and regulations of the SEC.
53
| ITEM 14 | Principal Accounting Fees and Services |
Fees Paid to Independent Public Accountants
The following table presents fees for professional audit services rendered by Bernstein & Pinchuk LLP for the audit of the Company’s annual financial statements for the year ended December 31, 2007 and fees billed for other services rendered by them during this period, and fees for professional audit services billed and expected to be billed by MSPC for the audit of the Company’s annual financial statements for the year ended December 31, 2008 and fees billed for other services rendered by them during this year.
| Fiscal 2008 | Fiscal 2007 | |||||||
| Audit fees (1) | $ | 140,000 | $ | 11,000 | ||||
| Audit Related Fees | — | — | ||||||
| Tax Fees | — | — | ||||||
| All Other Fees | — | — | ||||||
| TOTAL | $ | 140,000 | $ | 11,000 | ||||
(1) Audit Fees consist of fees billed for professional services rendered for the audit of the Company’s consolidated annual financial statements and review of the interim consolidated financial statements included in quarterly reports and services that are normally provided by our auditors in connection with statutory and regulatory filings or engagements.
Policy on Pre-Approval of Audit and Non-Audit Services of Independent Auditor
PART IV
| ITEM 15 | Exhibits and Financial Statement Schedules |
Exhibit No. | Description | |
| 2.1 | Share Exchange Agreement, dated as of April 17, 2008.(1) | |
| 3.1 | Amended Articles of Incorporation.(1) | |
| 4.1 | Form of Investor Warrant (i).(1) | |
| 4.2 | Form of Investor Warrant (ii). (1) | |
| 4.3 | Form of Placement Agent Warrant.(1) | |
| 4.4 | Registration Rights Agreement, dated as of April 17, 2008.(1) | |
| 4.5 | Registration Rights Agreement, dated as of September 5, 2008. (2) | |
| 4.7 | Form of Investor Warrant. (2) | |
| 4.8 | Form of Placement Agent Warrant (2) | |
| 10.1 | Securities Purchase Agreement, dated as of April 17, 2008.(1) | |
| 10.2 | Lockup Agreement, dated as of April 17, 2008.(1) | |
| 10.3 | Make Good Escrow Agreement, dated as of April 17, 2008.(1) | |
| 10.4 | Closing Escrow Agreement, dated as of April 17, 2008.(1) | |
| 10.5 | Sales Agreement, dated April 1, 2008 by and between Inner Mongolia Yongye Biotechnology Co., Ltd. and Yongye Nongfeng Biotechnology Co., Ltd .(1) | |
| 10.6 | Cooperation Agreement dated January 15, 2008 by and between Inner Mongolia Yongye Biotechnology Co., Ltd. and Yongye Nongfeng Biotechnology Co., Ltd.(1) | |
| 10.7 | Sino-foreign Cooperative Joint Venture Contract, dated November 16, 2007 by and between Inner Mongolia Yongye Biotechnology Co., Ltd. and Asia Standard Oil Limited.(1) |
54
| 10.8 | Supplemental Agreement to the Sino-foreign Cooperative Joint Venture Contract by and between Inner Mongolia Yongye Biotechnology Co., Ltd. and Asia Standard Oil Limited.(1) | |
| 10.9 | Securities Purchase Agreement, dated as of September 5, 2008. (2) | |
| 10.10 | Make Good Escrow Agreement, dated as of September 5, 2008.(2) | |
| 23.1 | Consent | |
| 23.2 | Consent . | |
| 31.1 | Certification of Chief Executive Officer pursuant to Rule 13a-14(a)/15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002, filed herewith.* | |
| 31.2 | Certification of Interim Principal Financial Officer pursuant to Rule 13a-14(a)/15d-14(a) of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley Act of 2002, filed herewith.* | |
| 32 | Certification of Chief Executive Officer and Interim Principal Financial Officer Pursuant to 18 U.S.C. 1350 (Section 906 of the Sarbanes-Oxley Act of 2002), filed herewith.* |
(1) Incorporated by reference herein to the Report on Form 8-K filed with the Securities and Exchange Commission on April 22, 2008.
(2) Incorporate by reference herein to the Registration Statement on Form S-1/A (Reg. No. 333-150949) filed with the Securities and Exchange Commission on September 9, 2008.
55
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities and Exchange Act, the Registrant has duly caused this Report to be signed on its behalf by the undersigned thereunto duly authorized.
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| Date: March 24, 2009 | By: | /s/ Zishen Wu | |
| Name: | Zishen Wu | ||
| Title: | Chief Executive Officer |
Pursuant to the requirements of the Securities and Exchange Act of 1934, this Report has been signed below by the following person on behalf of the Registrant and in the capacities and on the dates indicated.
| Dated: March 24, 2009 | By: | /s/ Zishen Wu | |
| Name: | Zishen Wu | ||
| Title: | Chief Executive Officer and Chairman | ||
| Date: March 24, 2009 | By: | /s/ Larry Gilmore | |
| Name: | Larry Gilmore | ||
| Title: | Interim Principal Financial Officer | ||
| Dated: March 24, 2009 | By: | /s/ Taoran Sun | |
| Name: | Taoran Sun | ||
| Title: | Vice Chairman | ||
| Dated: March 24, 2009 | By: | /s/ Zhao Qiang | |
| Name: | Zhao Qiang | ||
| Title: | Director | ||
| Dated: March 24, 2009 | By: | /s/ Guo Xiaochuan | |
| Name: | Guo Xiaochuan | ||
| Title: | Director | ||
| Dated March 24, 2009 | By: | /s/ Zhang Haiming | |
| Name: | Zhang Haiming | ||
| Title: | Director | ||
56
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC.
AND SUBSIDIARIES
CONTENTS
| PAGE | F-1 | REPORTS OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRMS |
| PAGE | F-3 | CONSOLIDATED BALANCE SHEETS. |
| PAGE | F-4 | CONSOLIDATED STATEMENTS OF INCOME AND COMPREHENSIVE INCOME. |
| PAGE | F-5 | CONSOLIDATED STATEMENT OF CHANGES IN STOCKHOLDERS’ EQUITY. |
| PAGE | F-7 | CONSOLIDATED STATEMENTS OF CASH FLOWS. |
| PAGES | F-8 - F-19 | NOTES TO CONSOLIDATED FINANCIAL STATEMENTS. |
57
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Yongye Biotechnology International, Inc.
We have audited the accompanying consolidated balance sheet of Yongye Biotechnology International, Inc. and Subsidiaries as of December 31, 2008 and the related consolidated statements of income and comprehensive income, changes in stockholders' equity, and cash flows for the year then ended. These consolidated financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these consolidated financial statements based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Yongye Biotechnology International, Inc. and Subsidiaries as of December 31, 2008 and the results of their operations and their cash flows for the year then ended in conformity with United States generally accepted accounting principles.
MSPC
Certified Public Accountants and Advisors, P.C.
New York, New York
March 19, 2009
F-1
Report of Independent Registered Public Accounting Firm
To the Board of Directors
Yongye Biotechnology Co.
We have audited the accompanying balance sheet of Yongye Biotechnology Co. as of December 31, 2007, and the related statements of operations and comprehensive income, stockholders’ equity, and cash flows for the year then ended. These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on these financial statements based on our audits.
We conducted our audit in accordance with auditing standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a reasonable basis for our opinion.
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of Yongye Biotechnology Co. as of December 31, 2007, and the results of their operations and cash flows for the year then ended in conformity with accounting principles generally accepted in the United States of America.
PATRIZIO & ZHAO, LLC
Parsippany, New Jersey
January 25, 2008
F-2
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
| Yongye Biotechnology | The Predecessor | |||||||
| International, Inc. | Inner Mongolia | |||||||
| and Subsidiaries | Yongye | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| CURRENT ASSETS | ||||||||
| Cash and cash equivalents | $ | 4,477,477 | $ | 376,002 | ||||
| Accounts receivable, net | 2,748,042 | 1,630,609 | ||||||
| Inventories | 20,708,193 | 9,851,788 | ||||||
| Advance payments | 44,051 | - | ||||||
| Due from related party | 192,741 | - | ||||||
| Due from affiliates | - | 978,384 | ||||||
| Prepaid expenses | 189,478 | - | ||||||
| Other receivables | 680,752 | 27,038 | ||||||
| Total Current Assets | 29,040,734 | 12,863,821 | ||||||
| PROPERTY AND EQUIPMENT, NET | 5,368,074 | 2,486,487 | ||||||
| INTANGIBLE ASSETS, NET | 95,453 | 3,665,584 | ||||||
| LONG-TERM INVESTMENTS | - | 4,115,764 | ||||||
| TOTAL ASSETS | $ | 34,504,261 | $ | 23,131,656 | ||||
| CURRENT LIABILITIES | ||||||||
| Short-term bank loans | - | 5,484,000 | ||||||
| Accounts payable and accrued expenses | $ | 630,619 | 1,271,852 | |||||
| Due to shareholders | - | 2,507,371 | ||||||
| Taxes payable | 366,981 | 893,892 | ||||||
| Advance from customers | 1,869,400 | - | ||||||
| Other payables | 626,911 | 50,916 | ||||||
| Total Current Liabilities | 3,493,911 | 10,208,031 | ||||||
| LONG-TERM LOANS | 397,773 | 12,153 | ||||||
| Minority interest | 1,202,388 | - | ||||||
| STOCKHOLDERS' EQUITY | ||||||||
| Capital stock: par value $.001; 26,760,258 shares authorized and issued at December 31, 2008 | 26,760 | - | ||||||
| Capital contribution | - | 7,260,000 | ||||||
| Additional paid-in capital- Common stock | 13,976,900 | - | ||||||
| Additional paid-in capital- Warrants | 3,883,432 | - | ||||||
| Retained earnings | 9,984,085 | 4,024,111 | ||||||
| Statutory reserve | 1,207,912 | 480,629 | ||||||
| Accumulated other comprehensive income | 331,100 | 1,146,732 | ||||||
| Total Stockholders' Equity | 29,410,189 | 12,911,472 | ||||||
| TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY | $ | 34,504,261 | $ | 23,131,656 | ||||
The accompanying notes are an integral part of these consolidated financial statements.
F-3
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF INCOME AND COMPREHENSIVE INCOME
| Yongye Biotechnology | ||||||||
| International, Inc. | The Predecessor | |||||||
| and Subsidiaries | Inner Mongolia Yongye | |||||||
| FOR YEAR ENDED | FOR YEAR ENDED | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| SALES | $ | 48,092,271 | $ | 13,137,406 | ||||
| COST OF SALES | 23,165,684 | 7,274,710 | ||||||
| GROSS PROFIT | 24,926,587 | 5,862,696 | ||||||
| SELLING EXPENSES | 8,665,755 | 449,168 | ||||||
| GENERAL AND ADMINISTRATIVE EXPENSES | 2,573,017 | 476,828 | ||||||
| INCOME FROM OPERATIONS | 13,687,815 | 4,936,700 | ||||||
| OTHER EXPENSES | ||||||||
| Interest expenses | (3,135 | ) | (212,239 | ) | ||||
| Other expenses | (526,039 | ) | (365,907 | ) | ||||
| TOTAL OTHER EXPENSES | (529,174 | ) | (578,146 | ) | ||||
| INCOME BEFORE PROVISION FOR INCOME TAXES AND MINORITY INTEREST | 13,158,641 | 4,358,554 | ||||||
| PROVISION FOR INCOME TAXES | 864,292 | - | ||||||
| NET INCOME BEFORE MINORITY INTEREST | 12,294,349 | 4,358,554 | ||||||
| PROVISION FOR MINORITY INTEREST | 1,102,388 | - | ||||||
| NET INCOME | 11,191,961 | 4,358,554 | ||||||
| Foreign Currency Translation Adjustment | 331,100 | 723,298 | ||||||
| COMPREHENSIVE INCOME | $ | 11,523,061 | $ | 5,081,952 | ||||
| Net income per share: | ||||||||
| Basic | $ | 0.66 | $ | 0.38 | ||||
| Diluted | $ | 0.64 | $ | 0.38 | ||||
| Weighted average shares used in computation: | ||||||||
| Basic | 16,937,852 | 11,444,775 | ||||||
| Diluted | 17,546,796 | 11,444,775 | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC. AND SUBSIDIARIES
STATEMENTS OF CHANGES IN STOCKHOLDERS' EQUITY
FOR THE YEAR ENDED DECEMBER 31, 2008
| Shares of | Common | Additional | Additional | |||||||||||||
| Common | Stock | Paid-in Capital | Paid-in Capital | |||||||||||||
| Stock | Amount | -Common Stock | -Warrants | |||||||||||||
| Balance at January 1, 2008 | 4,960,000 | 4,960 | (6,860 | ) | - | |||||||||||
| Stock issued for Fullmax merger | 11,444,755 | 11,445 | (11,445 | ) | - | |||||||||||
| McElroy shares cancelled | (2,900,000 | ) | (2,900 | ) | 2,900 | - | ||||||||||
| Stock issued for cash April 17, 2008 | 6,495,619 | 6,495 | 7,234,148 | 1,953,849 | ||||||||||||
| Stock issued for cash September 8, 2008 | 6,073,006 | 6,073 | 6,758,844 | 1,929,583 | ||||||||||||
| Warrants exercised | 686,878 | 687 | (687 | ) | - | |||||||||||
| Foreign currency translation adjustment | - | - | - | - | ||||||||||||
| Transfer to statutory reserve | - | - | - | |||||||||||||
| Net income for year ended December 31, 2008 | - | - | - | - | ||||||||||||
| Balance at December 31, 2008 | 26,760,258 | $ | 26,760 | $ | 13,976,900 | $ | 3,883,432 | |||||||||
F-5
| Accumulated other | Statutory | Retained | ||||||||||||||
| Comprehensive | Reserve | Earnings | Total | |||||||||||||
| Income | (Deficits) | Equity | ||||||||||||||
| Balance at January 1, 2008 | - | - | - | (1,900 | ) | |||||||||||
| Stock issued for Fullmax merger | - | - | 36 | 36 | ||||||||||||
| McElroy shares cancelled | - | - | - | - | ||||||||||||
| Stock issued for cash April 17, 2008 | - | - | - | 9,194,492 | ||||||||||||
| Stock issued for cash September 8, 2008 | - | - | - | 8,694,500 | ||||||||||||
| Warrants exercised | - | - | - | - | ||||||||||||
| Foreign currency translation adjustment | 331,100 | - | - | 331,100 | ||||||||||||
| Transfer to statutory reserve | - | 1,207,912 | (1,207,912 | ) | - | |||||||||||
| Net income for year ended December 31, 2008 | - | - | 11,191,961 | 11,191,961 | ||||||||||||
| Balance at December 31, 2008 | $ | 331,100 | $ | 1,207,912 | $ | 9,984,085 | $ | 29,410,189 | ||||||||
THE PREDECESSOR
INNER MONGOLIA YONGYE, CO.
STATEMENT OF CHANGES IN STOCKHOLDERS' EQUITY
FOR THE YEAR ENDED DECEMBER 31, 2007
Capital Contribution | Retained Earnings (Deficits) | Statutory Reserve | Accumulated other Comprehensive Income | Total Equity | ||||||||||||||||
| Balance at December 31, 2006 | $ | 7,260,000 | $ | 101,412 | $ | 44,774 | $ | 423,434 | $ | 7,829,620 | ||||||||||
| Net income | - | 4,358,554 | - | - | 4,358,554 | |||||||||||||||
| Statutory reserve | - | (435,855 | ) | 435,855 | - | - | ||||||||||||||
| Other comprehensive income | - | - | - | 723,298 | 723,298 | |||||||||||||||
| Balance at December 31, 2007 | $ | 7,260,000 | $ | 4,024,111 | $ | 480,629 | $ | 1,146,732 | $ | 12,911,472 | ||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-6
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
| Yongye Biotechnology | ||||||||
| International, Inc. | The Predecessor | |||||||
| And Subsidiaries | Inner Mongolia Yongye | |||||||
| FOR YEAR ENDED | FOR YEAR ENDED | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES | ||||||||
| Net income | $ | 11,191,961 | $ | 4,358,554 | ||||
| Adjustments to reconcile net income to net cash used in operating activities | ||||||||
| Depreciation and amortization | 118,104 | 212,423 | ||||||
| Provision for bad debts | 305,338 | 31,907 | ||||||
| Provision for minority interest | 1,102,388 | - | ||||||
| Loss on disposal of fixed assets | - | 149,853 | ||||||
| Changes in assets and liabilities: | ||||||||
| Accounts receivable | (3,053,380 | ) | (1,124,042 | ) | ||||
| Inventories | (20,708,193 | ) | (7,814,789 | ) | ||||
| Advanced payments | (44,051 | ) | 93,091 | |||||
| Due from related party | (192,741 | ) | - | |||||
| Due from related affiliates | - | (267,345 | ) | |||||
| Prepaid expense | (189,478 | ) | 5,741 | |||||
| Other receivables, net | (680,752 | ) | 66,926 | |||||
| Accounts payable and accrued expenses | 630,619 | 1,068,613 | ||||||
| Taxes payable | 366,981 | 835,137 | ||||||
| Advance from customers | 1,869,400 | - | ||||||
| Other payables | 616,911 | (2,234,407 | ) | |||||
| Total Adjustments | (19,858,854 | ) | (8,976,892 | ) | ||||
| Net Cash Used in Operating Activities | (8,666,893 | ) | (4,618,338 | ) | ||||
| CASH FLOWS FROM INVESTING ACTIVITIES | ||||||||
| Acquisition of property and equipment | (5,475,572 | ) | (308,312 | ) | ||||
| Additions to intangible assets | - | (909 | ) | |||||
| Net Cash Used in Investing Activities | (5,475,572 | ) | (309,221 | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES | ||||||||
| Proceeds from bank loans | 397,773 | 4,345,110 | ||||||
| Proceeds from shares issued | 19,350,651 | - | ||||||
| Proceeds from shareholder loans | - | 864,258 | ||||||
| Repayment of long-term loans | - | (12,131 | ) | |||||
| Payment for stock issuance costs | (1,461,659 | ) | - | |||||
| Net Cash Provided by Financing Activities | 18,286,765 | 5,197,237 | ||||||
| EFFECT OF FOREIGN CURRENCY TRANSLATION ON CASH | 325,041 | 17,301 | ||||||
| NET INCREASE IN CASH AND CASH EQUIVALENTS | 4,469,341 | 286,979 | ||||||
| CASH AND CASH EQUIVALENTS BEGINNING | 8,136 | 89,023 | ||||||
| CASH AND CASH EQUIVALENTS - ENDING | $ | 4,477,477 | $ | 376,002 | ||||
| Supplemental cash flow information: | ||||||||
| Cash paid for income taxes | 648,331 | - | ||||||
| Cash paid for interest expense payment | 11,301 | 212,239 | ||||||
| Noncash investing and financing activities: | ||||||||
| The minority shareholder of one of our subsidiaries contributed a patent valued at $100,000 to the subsidiary. | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-7
YONGYE BIOTECHNOLOGY INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2008
NOTE 1 - -ORGANIZATION AND DESCRIPTION OF BUSINESS
Organization
Yongye Biotechnology International Inc. (the “Company”, formerly known as “Golden Tan, Inc.”) was incorporated in the State of Nevada on December 12, 2006. On April 17, 2008, Yongye Biotechnology International Inc. entered into a Share Exchange Agreement (the “Exchange Agreement”) with Fullmax Pacific Limited, a company organized on May 23, 2007 under the laws of the British Virgin Islands (“Fullmax”), the shareholders of Fullmax (the “Shareholders”), who together own shares constituting 100% of the issued and outstanding ordinary shares of Fullmax (the “Fullmax Shares”), and the Company’s then principal shareholder . Pursuant to the terms of the Exchange Agreement, the Shareholders transferred to the Company all of the Fullmax Shares in exchange for the issuance of 11,444,755 (the “Shares”) shares of our Common Stock (the “Share Exchange”). As a result of the Share Exchange, Fullmax became our wholly-owned subsidiary and the Shareholders acquired approximately 84.7% of our issued and outstanding Common Stock. The transaction is accounted for as a reverse acquisition with a public shell.
The audited consolidated balance sheet as of December 31, 2008 includes Yongye Biotechnology International Inc., Fullmax, Asia Standard Oil Limited (“ASO”, subsidiary of Fullmax) and Yongye Nongfeng Biotechnology (“Yongye Nongfeng”, subsidiary of ASO) and the audited consolidated statements of operations and cash flows for the year ended December 31, 2008 includes Yongye Nongfeng for the full period and Fullmax, ASO and Yongye Biotechnology International Inc. from April 17, 2008. All the historical financial statements prior to January 1, 2008 are solely those of Inner Mongolia Yongye Biotechnology Co., Ltd., (formerly Yongye Biotechnology, Co.), (the “Predecessor”).
As part of the above financing described below, the Company agreed to begin a restructuring process whereby our Cooperative Joint Venture ('CJV") subsidiary, Yongye Nongfeng Biotechnology Co. ("Yongye Nongfeng"), will acquire all of the land rights, buildings, equipment and permits that currently belong to our Predecessor and outsourced manufacturing partner Inner Mongolia Yongye. This will enable Yongye Nongfeng to centralize and manage the Company’s product research and development, manufacturing and distribution and result in a more tightly integrated business model with greater control over our product quality and intellectual property. The full restructuring process should be completed by approximately September 2009. We will begin by purchasing the production equipment used in the existing 2,000 Tonnes Per Annum (TPA) production facility, which was purchased from the Inner Mongolia Yongye pursuant to an Asset Transfer Agreement on October 31, 2008.
In order to support our future growth, we have completed the construction of a new 8,000 Tonnes per Annum (“TPA”) production facility. This new facility is located at the same site as Inner Mongolia Yongye’s 2,000TPA production facility and has already been put into testing and should be into full production within the first quarter of 2009. The combined 10,000 TPA capacity of both facilities will allow the Company to produce its own finished goods in the quantities needed to fulfill its current and future sales goals. After the completion of the equipment purchase and the integration of the equipment with the Company’s new 8,000 TPA facility, the Company will discontinue its current exclusive contract manufacturing agreement with Inner Mongolia Yongye.
F-8
The Company, through its subsidiary Yongye Nongfeng, is engaged in the sales of fulvic acid based liquid and powder nutrient compounds used in the agriculture industry. Based on industry research and government testing, the Company’s proprietary technology for fulvic acid extraction creates fulvic acid and this is the base material for the plant and animal nutrients on the market in China. Yongye Nongfeng’s contractual relationship with the predecessor company Inner Mongolia Yongye is set up so that the predecessor conducts the manufacturing process and provides Yongye Nongfeng fulvic acid final product, which will allow Yongye Nongfeng to provide product to the customers.
Shares Issued and Registration Matters
Concurrent with the Share Exchange, we entered into a securities purchase agreement (the “April Purchase Agreement”) with certain investors (the “April Investors”) for the sale in a private placement of an aggregate of 6,495,619 shares of our Common Stock (the “April Investor Shares”), and 1,623,905 warrants to purchase 1,623,905 shares of Common Stock (the “April Warrants”) for aggregate gross proceeds equal to $10,000,651 (the “April Offering”). The warrants issued have a 5 years exercise period with an exercise price of $1.848. There were another 649,562 warrants issued to Roth Capital as the “Placement Agent” and these were issued with the same exercise price and term as the April Warrants. Expenses of the April Offering were $1,162,022 allocated as follows: fees for the issuance of stock of $806,159 as a reduction of Additional Paid In Capital on the December 31, 2008 Consolidated Balance Sheet and $355,863 as an expense on the Consolidated Statement of Income and Comprehensive Income for the year ended December 31, 2008.
In connection with the April Offering, we also entered into a registration rights agreement (the “April Registration Rights Agreement”) with the April Investors, in which we agreed to file a registration statement (the “April Registration Statement”) with the Securities and Exchange Commission (the “SEC”) to register for resale the April Investor Shares and the shares underlying the April Warrants, within 45 calendar days of the closing date of the April Offering, and use our best efforts to have the registration statement declared effective within 150 calendar days of the closing date of the April Offering. We filed the April Registration Statement on Form S-1 on May 15, 2008.
In connection with the April Offering, we also entered into an escrow agreement with ROTH Capital Partners, LLC, a representative of the Investors (“Roth”), Tri-State Title & Escrow LLC (the “Escrow Agent”) and Full Alliance International Limited (The “Full Alliance”), one of the Shareholders (the “April Escrow Agreement”), pursuant to which 2,000,000 of the Shares (the “April Escrow Shares”) were delivered to the Escrow Agent. The Escrow Shares are being held as security for the achievement of $10,263,919 after tax net income (“ATNI”) for the year ending December 31, 2008 (the 2008 “Net Income Threshold”). If we achieve the Net Income Threshold, the Escrow Shares will be released back to Full Alliance. If the Net Income Threshold is not achieved, the Escrow Shares will be distributed pro-rata to the April Investors. As of the filing of this Form 10-K, the ATNI threshold has been achieved.
On September 5, 2008, we entered into a securities purchase agreement (the “September Purchase Agreement”), with certain Qualified Institutional Buyers (the “September Investors”), for the sale in a private placement of an aggregate of 6,073,006 shares of our Common Stock (the “September Investor Shares”), and the issuance of 1,518,253 warrants to purchase 1,518,253 shares of Common Stock (the “September Warrants”), for aggregate gross proceeds equal to approximately $9,350,000 (the “September Offering”). The warrants issued have a 5 years exercise period with an exercise price of $1.848. There were another 607,301 warrants issued to Roth Capital as the “Placement Agent” and these were issued with the same exercise price and term as the September Warrants. Expenses of the September Offering were $995,500 allocated as follows: fees for the issuance of stock at $655,500 as a reduction of Additional Paid In Capital on the December 31, 2008 Consolidated Balance Sheet and $340,000 as an expense on the Consolidated Statement of Income and Comprehensive Income for the year ended December 31, 2008.
On September 12, 2008 Roth Capital executed an irrevocable cashless exercise of its warrants and purchased 686,878 shares in the aggregate of Common Stock of the Company pursuant to the April 17, 2008 and September 5, 2008 Placement Agent Warrants. In exchange for the issuance of 354,987 shares, Roth surrendered 649,562 warrants received in the April Offering; and in exchange for 331,891 shares, Roth surrendered 607,301 warrants received in the September Offering. At December 31, 2008 there are 3,142,158 warrants outstanding with a weighted average exercise price of $1.848. Of this total, 1,623,905 expire in April 2013 and 1,518,235 expire in September 2013.
F-9
In connection with the September Offering, we also entered into a registration rights agreement (the “September Registration Rights Agreement”) with the September Investors, in which we agreed to file a registration statement (the “September Registration Statement”) with the SEC to register for resale the September Investor Shares and the shares underlying the September Warrants, on or prior to 45 calendar days after the closing date of the September Offering, and use our best efforts to have the September Registration Statement declared effective within 150 calendar days of the closing date of the September Offering. We registered the September Investor Shares and the shares underlying the September Warrants in the April Registration Statement by using a “piggy back” registration process. We requested that the SEC accelerate the effectiveness of our S-1 Registration Statement on September 8, 2008 which was declared effective on September 11, 2008
In connection with the September Offering, we entered into an escrow agreement with Roth, the Escrow Agent and Full Alliance (the “September Escrow Agreement”), pursuant to which 4,000,000 of the Shares issued to Full Alliance in the Share Exchange (the “September Escrow Shares”) were delivered to the Escrow Agent. Of the September Escrow Shares, 2,000,000 shares (the “Make Good Escrow Shares”) are being held as security for the achievement of 2008 and 2009 Make Good ATNI in the following manner. If the Company achieves (i) the 2008 Net Income Threshold, and (ii) fully diluted earnings per share reported in the 2008 Annual Report on Form 10-K filed with the SEC (the “2008 Annual Report”), of no less than $0.42 (the “2008 Guaranteed EPS”), then the provisions described in the following paragraph apply with respect to the achievement of 2009 net income and fully diluted earnings per share targets and the Make Good Escrow Shares will be retained in escrow for the achievement of certain net income and fully diluted earnings per share targets for the year ending December 31, 2009. If the Company does not achieve the Make Good ATNI, the Make Good Shares will be released pro-rata to the September Offering investors. As of the filing of this Form 10-K, the ATNI threshold has been achieved.
In the event that (i) the 2009 After Tax Net Income equals or exceeds $12,649,248 and is less than $15,811,560, or (ii) the fully diluted earnings per share reported in the 2009 Annual Report on Form 10-K filed with the SEC (the “2009 Annual Report”), equals or exceeds $0.42 and is less than $0.53, then Make Good Shares equal to the product of (i)(A) $15,811,560 minus the 2009 After Tax Net Income, divided by (B) $15,811,560, and (ii) the Make Good Escrow Shares, shall be transferred to the September Investors on a pro-rata basis, and the remaining Make Good Shares shall be returned to Full Alliance. If the 2009 ATNI exceeds $15,811,560, the 2,000,000 Make Good Escrow Shares will be released back to Full Alliance.
The remaining 2,000,000 escrow shares are being held as security for the timely issuance of Inner Mongolia Yongye ’s fertilizer License into the name of Yongye Nongfeng Biotechnology and completion of the CJV Restructuring as defined below (the “Restructuring Make Good Shares”). This license is issued by the Ministry of Agriculture and gives the owner the right to manufacturer and sell fertilizer products domestically. In the event that (1) the License has not been issued to Yongye Nongfeng Biotechnology by June 30, 2009, or such later date as agreed to by us and the September Investors holding a majority of the September Investor Shares at such time (the “License Grant Date”), or (2) the License has been issued by the License Grant Date, but the CJV Restructuring is not completed by the Restructuring Completion Date, the Restructuring Make Good Shares shall be transferred in accordance with the September Escrow Agreement to the September Investors on a pro-rata basis for no consideration other than their respective investment amounts paid to us at the closing of September Offering. The “Restructuring Completion Date” shall be the date that is 132 calendar days after the License Grant Date. If the License is issued by the License Grant Date and the CJV Restructuring is completed by the Restructuring Completion Date, the Restructuring Make Good Shares shall be returned to Full Alliance.
The Predecessor Company
The Company’s Predecessor, Inner Mongolia Yongye Biotechnology Co., Ltd. (the “Predecessor” or “Inner Mongolia Yongye”) was formed on September 16, 2003, under PRC law. Its primary business is to research, manufacture, and sell biological products for use in plants and animal growth. The Company is located in the City of Hohhot, Inner Mongolia Autonomous Region., and People’s Republic of China (“PRC”). Yongye Nongfeng Biotechnology Co., Ltd. was incorporated on January 4, 2008, under the corporate laws of the People’s Republic of China (“PRC”) by Inner Mongolia Yongye and ASO, a Hong Kong based company. Inner Mongolia Yongye was a 10% owner of Yongye Nongfeng and ASO 90% pursuant to a Sino-Foreign Cooperative Joint Venture Contract (the “Contract”). In May 2008, the Contract was revised that Inner Mongolia Yongye is a 0.5% owner of Yongye Nongfeng and ASO 99.5%. The terms of the Contract provide that the purpose of the Company is the promotion and sales of Inner Mongolia Yongye’s products. All of the Yongye Nongfeng’s products will be purchased from Inner Mongolia Yongye. The term of the contract is for ten years and may be extended by written application submitted to the Approval Authority for approval no less than six (6) months prior to the expiration of the term. However, Asia Standard Oil, Ltd didn’t fully pay the contractual registered capital into Yongye Nongfeng. Based upon actual capital injection into the CJV, Inner Mongolia Yongye is a 0.6% owner of Yongye Nongfeng Biotechnology and Asia Standard Oil, Ltd. is a 99.4% owner.
F-10
NOTE 2 - -SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
PRINCIPLES OF CONSOLIDATION AND BASIS OF PRESENTATION
The accompanying consolidated financial statements of the Company have been prepared in accordance with generally accepted accounting principles in the United States of America (“GAAP”) and include the financial statements of the Company and its majority-owned subsidiaries. All significant intercompany transactions and balances are eliminated on consolidation.
USE OF ESTIMATES
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenues, expenses, and related disclosures at the date of the financial statements and during the reporting period. Actual results could differ from those estimates.
CASH AND CASH EQUIVALENTS
In accordance with Statement of Financial Accounting Standards ("SFAS") No. 95, “Statement of Cash Flows,” the Company and the Predecessor considers all highly liquid instruments with original maturities of three months or less to be cash and cash equivalents.
ACCOUNTS RECEIVABLE AND BAD DEBT RESERVE
The Company performs certain credit evaluation procedures and does not require collateral for financial instruments subject to credit risk. The Company believes that credit risk is limited because the Company routinely assesses the financial strength of its customers and, based upon factors surrounding the credit risk of its customers, establishes an allowance for uncollectible accounts receivable. As a consequence, the Company believes that its accounts receivable credit risk exposure beyond such allowances is limited. The Company recognizes an allowance for doubtful accounts to ensure accounts receivable are not overstated due to uncollectability and are maintained for all customers based on a variety of factors, including the length of time the receivables are past due, significant one-time events and historical experience. Based on the age of the receivables, the Company reserves 10% of accounts receivable balances that have been outstanding for more than 6 months but less than one year, 20% of accounts receivable balances that have been outstanding between one year and two years, 50% of receivable balances that have been outstanding between two year and three years, and 100% of receivable balances that have been outstanding for more than three years. An additional reserve for individual accounts is recorded when the Company becomes aware of a customer's inability to meet its financial obligation, such as in the case of bankruptcy filings or deterioration in the customer's operating results or financial position. If circumstances related to customers change, estimates of the recoverability of receivables would be further adjusted .An allowance for doubtful accounts of $305,338 was provided as of December 31, 2008.
INVENTORY
Inventory is stated at the lower of weighted average cost, which takes into account historical prices on a continuing basis, or market. Cost is determined by the weighted average method. Provision for diminution in value on inventories is made using specific identification method.
F-11
PROPERTY AND EQUIPMENT
Property and equipment other than leasehold improvements are stated at cost and depreciated using the straight-line method over the estimated useful lives of the assets. Leasehold improvements are stated at cost and depreciated using the straight-line method over the estimated useful life or lease period, whichever is shorter. Estimated useful lives are as follows:
| Estimated Useful Life | |
| Yongye Biotechnology International, Inc. | |
| Buildings and structures | 30 years |
| Office equipment and furniture | 5 years |
| Machinery and equipment | 10 years |
| Vehicles | 10 years |
| Software | 10 years |
| Leasehold improvements | 3 years |
| The Predecessor- Inner Mongolia Yongye | |
| Buildings | 50 years |
| Machinery and equipment | 10-20 years |
| Transportation equipment | 10 years |
REVENUE RECOGNITION
Revenue from product sales is recognized when title has been transferred, which is generally at the time of customer’s receipt of product, the risks and rewards of ownership have been transferred to the customer, the fee is fixed and determinable, and the collection of the related receivable is probable. The Company reports revenue net of value added taxes if applicable.
ADVERTISING COSTS
Advertising costs are expensed as incurred. Advertising costs for the years ended December 31, 2008 and 2007 were $5,109,502 and $15,800, respectively.
IMPAIRMENT OF LONG-LIVED ASSETS
The Company follows SFAS No. 144, “Accounting for the Impairment or Disposal of Long-Lived Assets” (“SFAS 144”), which addresses financial accounting and reporting for the impairment or disposal of long-lived assets. Per SFAS 144, the Company is required to periodically evaluate the carrying value of long-lived assets and to record an impairment loss when indicators of impairment are present and the undiscounted cash flows estimated to be generated by those assets are less than the asset’s carrying amounts.
In that event, a loss is recognized based on the amount by which the carrying amount exceeds the fair market value of the long-lived assets. Loss on long-lived assets to be disposed of is determined in a similar manner, except that fair market values are reduced for the cost of disposal. Based on its review, the Company and the Predecessor concluded that as of December 31, 2008 and 2007 there were no significant impairments of their long-lived assets.
INCOME TAXES
Deferred income taxes are computed using the asset and liability method, such that deferred tax assets and liabilities are recognized for the expected future tax consequences of temporary differences between financial reporting amounts and the tax basis of existing assets and liabilities based on currently enacted tax laws and tax rates in effect in the People’s Republic of China for the periods in which the differences are expected to reverse. Income tax expense is the tax payable for the period plus the change during the period in deferred income taxes.
A valuation allowance is provided when it is more likely than not that some portion or all of the deferred tax assets will not be realized. No material differences were noted between the book and tax bases of the Company and the Predecessor’s assets and liabilities, respectively, therefore, there are no deferred tax assets or liabilities as of December 31, 2008 and 2007. Yongye Nongfeng is subject to PRC Enterprise Income Tax at a rate of 25% of net income from its foundation on January 4, 2008 to March 31, 2008, and 1.25% of gross revenue since April 1, 2008. Since the Predecessor is located in the economic development area in Inner Mongolia Autonomous Region, the Predecessor is exempt from income tax according to the tax law in China. .
F-12
FOREIGN CURRENCY TRANSLATION AND TRANSACTIONS
The financial position and results of operations of the Company’s Chinese subsidiaries are determined using the local currency (Chinese Yuan) as the functional currency, while the reporting currency is the US dollar. Assets and liabilities of the subsidiaries are translated at the prevailing exchange rate in effect at each period end. Contributed capital accounts are translated using the historical rate of exchange when capital is injected. Income statement accounts are translated at the average rate of exchange during the period. Translation adjustments arising from the use of different exchange rates from period to period are included in the cumulative translation adjustment account in shareholders’ equity. Gains and losses resulting from foreign currency transactions denominated in other than the functional currency are included in operations as incurred. Such gains and losses were immaterial for the years ended December 31, 2008 and 2007.
The PRC government imposes significant exchange restrictions on fund transfers out of the PRC that are not related to business operations. These restrictions have not had a material impact on the Company because it has not engaged in any significant transactions that are subject to the restrictions.
FAIR VALUE OF FINANCIAL INSTRUMENTS
The carrying amounts of cash and cash equivalents, trade, related party and other receivables, accounts, related party and other payables approximate their fair value due to the short-term nature of these instruments.
NET INCOME PER SHARE
Basic net income per share is computed by dividing net income attributable to common shareholders by the weighted average number of common shares outstanding during the period, but excluding the 2,000,000 “Make Good Shares” according to the “Make Good Escrow Agreement” in April 2008, and 4,000,000 “September Escrow Shares” according to the “September Escrow Agreement” (see Note 1). Diluted net income per share reflects the potential dilution that would occur upon the exercise of outstanding warrants. Common share equivalents are excluded from the computation of the diluted net income per share in periods when their effect would be anti-dilutive.
RECENTLY ISSUED ACCOUNTING PRONOUNCEMENTS
In September 2006, the Financial Accounting Standards Board (“FASB”) issued Statement No. 157, “Fair Value Measurement” (FAS 157). While this statement does not require new fair value measurements, it provides guidance on applying fair value and expands required disclosures. FAS 157 is effective for the Company beginning in the first quarter of fiscal 2009. This pronouncement should not have a material impact on the Company’s financial statements.
In February 2008, the FASB issued “Effective Date of FASB Statement No.157” FASB Staff Position (FSP) No. 157-2 (FSP No. 157-2). FSP No.157-2 delays the effective date of SFAS No. 157 until fiscal years beginning after November 15, 2008, for fair value measurements of non-financial assets and non-financial liabilities, except for items that are recognized or disclosed at fair value in an entity’s financial statements on a recurring basis (at least annually).
In February 2007, the FASB issued Statement No. 159, “The Fair Value Option for Financial Assets and Financial Liabilities” (FAS 159). The statement, which is expected to expand fair value measurement, permits entities to choose to measure many financial instruments and certain others items at fair value. FAS 159 is effective for the Company beginning in the first quarter of 2009. This pronouncement should not have a material impact on the Company’s financial statements.
F-13
In March 2008, the FASB issued SFAS No. 161, “Disclosures about Derivative Instruments and Hedging Activities” (FAS 161). The new standard is intended to improve financial reporting about derivative instruments and hedging activities by requiring enhanced disclosures to enable investors to better understand their effects on an entity’s financial position, financial performance, and cash flows. It is effective for financial statements issued for fiscal years and interim periods beginning after November 15, 2008, with early application encouraged. The Company does not expect the adoption of FAS 161 to have a material impact on its financial statements.
NOTE 3-INVENTORIES
Inventories at December 31, 2008 and 2007 consisted of the following:
| Yongye Biotechnology | The Predecessor | |||||||
| International, Inc. and Subsidiaries | Inner Mongolia Yongye | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| Raw materials | $ | - | $ | 384,361 | ||||
| Packing supplies | - | 195,127 | ||||||
| Work-in process | - | 4,969,350 | ||||||
| Finished goods | 20,664,930 | 4,302,950 | ||||||
| Consumables | 43,263 | - | ||||||
| Total | $ | 20,708,193 | $ | 9,851,788 | ||||
NOTE 4-DUE FROM AFFILIATES
The balance due from the Predecessor’s affiliated entity, Huimin Biotechnology Co., Ltd., at December 31, 2007 was $978,384. The balance had no stated terms for repayment and was not interest-bearing.
NOTE 5-PROPERTY AND EQUIPMENT
Property and equipment at December 31, 2008 and 2007 consisted of the following:
| Yongye Biotechnology | ||||||||
International, Inc. and Subsidiaries | The Predecessor Inner Mongolia Yongye | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| Buildings and structures | $ | 3,656,992 | $ | 1,560,251 | ||||
| Manufacturing equipment | 673,480 | 788,641 | ||||||
| Office equipment and furniture | 85,087 | 33,724 | ||||||
| Vehicles | 824,013 | 419,529 | ||||||
| Software | 17,156 | - | ||||||
| Leasehold improvement | 218,844 | - | ||||||
| Construction-in-process | - | 1,797 | ||||||
| 5,475,572 | 2,803,942 | |||||||
| Less: Accumulated depreciation | 107,498 | 317,455 | ||||||
| Total | $ | 5,368,074 | $ | 2,486,487 | ||||
Depreciation expense for the years ended December 31, 2008 and 2007 was $107,498 and $125,931, respectively.
Among the vehicles, 11 cars in the amount of $692,982 were pledged for the long-term banks loans of $438,563 which were received for purchasing those cars (see Note 10).
F-14
NOTE 6- INTANGIBLE ASSETS
Net intangible assets at December 31, 2008 and 2007 were as follows:
| Yongye Biotechnology | ||||||||
International, Inc. and Subsidiaries | The Predecessor Inner Mongolia Yongye | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| Rights to use land | $ | - | $ | 4,028,099 | ||||
| Patent | 106,059 | - | ||||||
| 106,059 | 4,028,099 | |||||||
| Less: accumulated amortization | 10,606 | 362,515 | ||||||
| Total | $ | 95,453 | $ | 3,665,584 | ||||
Product patent was acquired by the Company in March 2008 with an estimated useful life of 10 years. It is amortized using the straight-line method over its useful life commencing on April 1, 2008. The predecessor’s rights to use land are also amortized using the straight-line method over its useful life of 50 years. Amortization expense for the years ended December 31, 2008 and 2007 amounted to $10,606 and $86,492, respectively. The estimated aggregate amortization expense for each of the five succeeding fiscal years is $10,606.
NOTE 7 - LONG-TERM INVESTMENTS
Long-term investments of the Predecessor as of December 31, 2007 consist of medicinal plants and trees which the Predecessor purchased in conjunction with the right to use land. These medicinal plants and trees are to be used for human medical treatments and the Predecessor intends to sell them in future years as they mature.
NOTE 8 – SHORT-TERM BANK LOANS
On March 27, 2007, the Predecessor obtained a loan in the amount of $5,484,000 from Inner Mongolia Agriculture Development Bank, of which the principal is to be paid in full by March 26, 2008. The interest is to be calculated using an annual fixed interest rate of 6.39% and paid monthly. The loan is secured by the Predecessor’s property and equipment.
NOTE9 – DUE TO SHAREHOLDERS
As of December 31, 2007, the Predecessor has $2,507,371 in loans from shareholders. These loans are short-term in nature, unsecured and non-interest bearing.
NOTE 10 – LONG-TERM LOANS
From August to December 2008, the Company financed the purchase of 11 cars by with bank loans of $438,563. The Company pledged those 11 cars with an initial value of $692,982 to the loans. The loans all have 3-year terms and are paid in monthly installments. Interest on the loans is range from 5.45% to 14.54% annually. These loans were obtained by individuals who are employees of the Company on behalf of the Company. The Company and the individuals entered into agreements of trust whereby the Company is entitled to the cars and is responsible for payments on the loans. Under the loan agreements, the Company must make specified payments monthly. The aggregate amount of such required payments at December 31, 2008 is as follows:
F-15
| 2009 | $ | 167,652 | ||
| 2010 | 167,652 | |||
| 2011 | 119,758 | |||
| Total | 455,062 | |||
| Less: Amount representing interest | (57,289 | ) | ||
| Total at present value | $ | 397,773 |
The Company's total payments under the agreement were $47,151 which included interest expenses $11,301 during the year ended December 31, 2008.
As of December 31, 2007 the Predecessor has $12,153 of long-term, unsecured and non-interest bearing loans from shareholders.
NOTE 11 MINORITY INTEREST
The Company’s main operating subsidiary, Yongye Nongfeng, is a Cooperative Joint Venture by ASO and the Predecessor, Inner Mongolia Yongye. During the year ended December 31, 2008, the Predecessor invested $100,000 in Yongye Nongfeng by contributing a patent and ASO made 4 cash investments totaling $ 16,778,771.
NOTE 12 STATUTORY COMMON WELFARE FUND
As stipulated by the PRC, net income after taxation can only be distributed as dividends after appropriation has been made for the following:
| (i) | Making up cumulative prior years’ losses, if any; |
| (ii) | Allocations to the “Statutory surplus reserve” of at least 10% of income after tax, as determined under PRC accounting rules and regulations, until the fund amounts to 50% of the company’s registered capital; |
| (iii) | Allocation of 5-10% of income after tax, as determined under PRC accounting rules and regulations, to the company’s “Statutory common welfare fund”, which is established for the purpose of providing employee facilities and other collective benefits to the company’s employees. Chinese companies invested by companies registered outside mainland China, including joint ventures, are exempted from contributing to this fund; |
| (iv) | Allocations to the discretionary surplus reserve, if approved in the shareholders’ annual general meeting. |
The predecessor did not provide a reserve for the welfare fund for the year ended December 31, 2007. The Company provided $1,207,912 to the statutory surplus reserve for the year ended December 31, 2008.
NOTE 13 EMPLOYEE BENEFIT PLANS
The employees of the Company who are domiciled in the PRC receive coverage under a comprehensive benefit plan as required by the local social security governing bureau. The calculation for contribution by eligible employees is based on 20% of the base salary. The contribution paid by the Company on behalf of their employees for this defined benefit plan was $31,053 for the year ended December 31, 2008. The Predecessor was not obliged to pay any contribution for the year ended December 31, 2007.
In addition, the Company is required to contribute a portion of the employees base salary for those employees domiciled in Beijing in the following manner- approximately 10% for medical benefits, 1.5% for unemployment benefits and 1.3% for workers compensation. Contributions for the employees located in Inner Mongolia for these benefits is not required for the year ended December 31, 2008. The PRC government is directly responsible for the payments of the benefits to these employees. The amounts contributed by the Company were $19,651 for the year ended December 31, 2008. The Predecessor was not obliged to pay any contribution for the year ended December 31, 2007.
F-16
NOTE 14 – INCOME TAXES
A reconciliation between taxes computed at the United States statutory rate of 34% and the Company’s and the Predecessor’s effective tax rate is as follows:
| Yongye Biotechnology | ||||||||
International, Inc. and Subsidiaries | The Predecessor Inner Mongolia Yongye | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| Income before income taxes | $ | 13,158,641 | $ | 4,358,554 | ||||
| Income tax on pretax income at statutory rate | 4,473,938 | 1,481,908 | ||||||
| Effect of different tax rates of subsidiary operating in other jurisdictions | (3,609,646 | ) | - | |||||
| Tax exemption | - | (1,481,908 | ) | |||||
| Income tax at effective rate | $ | 864,292 | $ | - | ||||
NOTE 15 – LEASE COMMITMENTS
The Company has entered into a building lease for our Beijing office. The lease for Beijing office is from January 1, 2008 to December 31, 2010. The lease expense for Beijing office was $227,606 for the year ended December 31, 2008. Future minimum lease payments under non-cancellable operating lease agreements at December 31, 2008 were as follows:
| December 31, 2009 | $ | 222,969 | ||
| December 31, 2010 | 231,194 | |||
| Total | $ | 454,163 |
NOTE 16 – RELATED PARTY TRANSACTIONS AND BALANCES
As of December 31, 2008, the Predecessor is a 0.6% shareholder of the Company’s main operating subsidiary, Yongye Nongfeng, and is Yongye Nongfeng’s only vendor, providing $43,509,906 (100%) of the Company’s purchased finished goods for the year ended December 31, 2008. According to the contract, the Predecessor sells to Yongye Nongfeng at fixed prices of RMB 350 per case for plant products and RMB 120 per case for animal products.
As of December 31, 2008, due to the Predecessor is $46,739 and represents the payable generated in purchasing inventory from the Predecessor; due from related party is $192,741 and represents the payment the Company made for the Predecessor for its professional fees and research & development fee. The amounts are unsecured and non-interest bearing, and has no defined payment terms.
During the year ended December 31, 2008, the Company borrowed $1,617,293 from Ms. Yin’s (Mr. Wu’s wife) company, Inner Mongolia Chilechuan Culture Development Co., Ltd. The amounts were unsecured and non-interest bearing, and were repaid before December 31, 2008.
Yongye Nongfeng and Inner Mongolia Yongye entered into two lease-exchange arrangements related to the land, buildings and equipment of the 2,000TPA and 8,000TPA facilities. On June 1, 2008, Yongye Nongfeng entered into a land lease agreement to lease 74,153 square meters of land from Inner Mongolia Yongye for a term beginning June 1, 2008 and ending May 31, 2009. On September 28, 2008, Inner Mongolia Yongye entered into a building lease agreement with Yongye Nongfeng to lease a building of 3,967 square meters and the 8,000 TPA production equipment for a term beginning September 28, 2008 and ending September 27, 2009. The estimated value of rentals of land lease and the combination of buildings and equipment were not materially different. Therefore, pursuant to the agreements, both Yongye Nongfeng and the Inner Mongolia Yongye did not charge lease fees to each other. Additionally, the rental income to be received by the Company and the rental expense to be paid are not material to the Company’s 2008 results of operations and therefore have not been included.
F-17
As of December 31, 2007, the Predecessor has borrowed $2,507,371 from stockholders. These loans are short term in nature, unsecured and non-interest bearing. Also, at December 31, 2007 the Predecessor has $12,153 of long-term, unsecured and non-interest bearing loans from shareholders.
NOTE 17-NET INCOME PER SHARE
The following table sets forth the computation of basic and diluted income per share for the periods indicated:
| Yongye Biotechnology | ||||||||
| International, Inc. | The Predecessor | |||||||
| and Subsidiaries | Inner Mongolia Yongye | |||||||
| FOR YEAR ENDED | FOR YEAR ENDED | |||||||
| DECEMBER 31, 2008 | DECEMBER 31, 2007 | |||||||
| Numerator used in basic net income per share: | ||||||||
| Net income | $ | 11,191,961 | $ | 4,358,554 | ||||
| Shares (denominator): | ||||||||
| Weighted average ordinary shares outstanding | 16,937,852 | 11,444,775 | ||||||
| Plus: weighted average incremental shares from assumed exercise of warrants | 608,944 | - | ||||||
| Weighted average ordinary shares outstanding used in computing diluted net income per ordinary share | 17,546,796 | 11,444,775 | ||||||
| Net income per ordinary share-basic | $ | 0.66 | $ | 0.38 | ||||
| Net income per ordinary share-diluted | $ | 0.64 | $ | 0.38 | ||||
NOTE 18 - -CONCENTRATIONS AND CREDIT RISKS
At December 31, 2008 and 2007, the Company and the Predecessor have a credit risk exposure of uninsured cash in banks of approximately $4,477,500 and $376,000, respectively. Neither the Company nor the Predecessor requires collateral or other securities to support financial instruments that are subject to credit risk.
Five major customers accounted for 92% and one major customer accounted for 43% of the Company’s net revenue for the year ended December 31, 2008. Five major customers accounted for 82% and one major customer accounted for 29% of the Predecessor’s net revenue for the year ended December 31, 2007. The Company and the Predecessor’s total sales to five major customers were $44,109,813 and $10,767,153 for the years ended December 31, 2008 and 2007, respectively.
Yongye Biotechnology International, Inc. and Subsidiaries | The Predecessor Inner Mongolia Yongye | |||||||||||||||||
| YEAR ENDED DECEMBER 31, 2008 | YEAR ENDED DECEMBER 31, 2007 | |||||||||||||||||
| Largest Customers | Amount of Sales | % Total Sales | Largest Customers | Amount of Sales | % Total Sales | |||||||||||||
| Hebei | $ | 20,541,267 | 43 | % | Xinjiang | $ | 3,853,891 | 29 | % | |||||||||
| Xinjiang | $ | 6,886,624 | �� | 14 | % | Beijing | $ | 2,980,234 | 23 | % | ||||||||
| Gansu | $ | 6,291,070 | 13 | % | Hebei | $ | 1,976,680 | 15 | % | |||||||||
| Inner Mongolia | $ | 5,663,011 | 12 | % | Dalian | $ | 1,216,097 | 9 | % | |||||||||
| Shandong | $ | 4,727,842 | 10 | % | Jiangsu | $ | 740,251 | 6 | % | |||||||||
| Total | $ | 44,109,813 | 92 | % | Total | $ | 10,767,153 | 82 | % | |||||||||
F-18
The Predecessor is the Company’s only vender who provided 100% of the Company purchased finished goods for the year ended December 31, 2008 in the amount of $43,509,906. The Predecessor had four major vendors who provided 73% of its raw materials for the year ended December 31, 2007. Total purchases from these vendors were $11,088,687 for the year ended December 31, 2007.
The Company and the Predecessor’s operations are carried out in the PRC. Accordingly, the Company and the Predecessor’s business, financial condition and results of operations may be influenced by the political, economic and legal environments in the PRC as well as by the general state of the PRC’s economy. The business may be influenced by changes in governmental policies with respect to laws and regulations, anti-inflationary measures, currency conversion and remittance abroad, and rates and methods of taxation, among other things.
NOTE 19 – SUBSEQUENT EVENTS
On October 29, 2008 on behalf of Yongye Nongfeng, Jones Lange LaSalle, an internationally recognized valuation company, completed the valuation of the 2,000TPA equipment owned by the predecessor company, Inner Mongolia Yongye Biotechnology in order to acquire it as required by the September financing. The purchase contract between the two companies allowed for payment 10 days subsequent to the “Completion Date” and the “Completion Date” was specified as two (2) months after the effective date of this Agreement or based upon normal performance of such conditions; the last date for satisfaction of all such conditions shall be the Completion Date. The satisfaction date for this was March 12, 2009 and the payment for the assets was made on that date. As such, on March 12, 2009 RMB 6,439,000 (USD $939,849), which fairly represented the market value of the machinery and equipment, was paid to Inner Mongolia Yongye Biotechnology by Yongye Nongfeng.
F-19