Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
AQST similar filings
- 7 Aug 23 Results of Operations and Financial Condition
- 2 Aug 23 Entry into a Material Definitive Agreement
- 27 Jul 23 Aquestive Therapeutics Announces Positive Topline Pharmacokinetic Data for Anaphylm™ (epinephrine) Sublingual Film
- 29 Jun 23 Advancing medicines. Solving problems. Improving lives. Corporate Presentation June 2023 1
- 23 Jun 23 Submission of Matters to a Vote of Security Holders
- 31 May 23 Regulation FD Disclosure
- 2 May 23 Aquestive Therapeutics Reports First Quarter 2023 Financial Results and Provides Business Update
Filing view
External links
Exhibit 99.1

Advancing medicines. Solving problems. Improving lives. Corporate Presentation June 2023 1

Disclaimer This presentation and the accompanying oral commentary has been prepared by Aquestive Therapeutics, Inc. (the “Company”, “our” or “us”) and contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believe,” “anticipate,” “plan,” “expect,” “estimate,” “intend,” “may,” “will,” or the negative of those terms, and similar expressions, are intended to identify forward-looking statements. These forward-looking statements include, but are not limited to, statements regarding the advancement of Libervant™, Anaphylm™ (“AQST-109”), and other product candidates through the regulatory and development pipeline and ability to bring those products to market and achieve market acceptance of those products; the potential benefits our products could bring to patients; the outlicensing of our products in the U.S. and abroad; our growth and future financial and operating results and financial position, including regarding the profitability of the Company's manufacturing operations and the current and future financial outlook of the Company; ability to refinance the Company’s current debt; and business strategies, market opportunities (including total addressable market size), and other statements that are not historical facts. These forward-looking statements are subject to the uncertain impact of global business or macroeconomic conditions, including as a result of inflation, rising interest rates, instability in the global banking system, and geopolitical conflicts, including the war in Ukraine, our business including with respect to our clinical trials including site initiation, patient enrollment and timing and adequacy of clinical trials; on regulatory submissions and regulatory reviews and approvals of our product candidates; pharmaceutical ingredient and other raw materials supply chain, manufacture, and distribution; sale of and demand for our products; our liquidity and availability of capital resources; customer demand for our products and services; customers’ ability to pay for goods and services; and ongoing availability of an appropriate labor force and skilled professionals. Given these uncertainties, the Company is unable to provide assurance that operations can be maintained as planned prior to the global business or macroeconomic conditions discussed above. These forward-looking statements are based on our current expectations and beliefs and are subject to a number of risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Such risks and uncertainties include, but are not limited to, risks associated with the Company's development work, including any delays or changes to the timing, cost and success of our product development activities and clinical trials and plans for Anaphylm and our other drug candidates; risk of failure to address the concerns identified in the FDA End-of-Phase 2 meeting for Anaphylm; risk of delays in FDA approval of Anaphylm and our other drug candidates or failure to receive approval at all; risk of the failure to receive FDA approval for U.S. market access for Libervant, including by establishing a major contribution to patient care within the meaning of FDA regulations, as well as risks related to other potential pathways or positions which are or may in the future be advanced to the FDA, to overcome the seven year orphan drug market exclusivity granted by the FDA for a nasal spray product of another company, and there can be no assurance that the Company will be successful in such endeavors; risk of our ability to out-license our proprietary products in the U.S. or abroad and risks that such product candidates will receive regulatory approval in those licensed territories and risk of the rate and degree of market acceptance of our product and product candidates in those territories; risk to growing our manufacturing revenues and generate cash and capabilities to support demand for current and future licensed products; risk of eroding market share for Suboxone® and risk of a sunsetting product, which accounts for the substantial part of our current operating revenue; risk inherent in commercializing a new product (including technology risks, financial risks, market risks and implementation risks and regulatory limitations); the success of any competing products, including generics; risks of compliance with all FDA and other governmental and customer requirements for our manufacturing facilities; risks associated with intellectual property rights and infringement claims relating to the Company's products; risk of unexpected patent developments; the impact of existing and future legislation and regulatory provisions on product exclusivity; legislation or regulatory actions affecting pharmaceutical product pricing, reimbursement or access; claims and risks that may arise regarding the safety or efficacy of the Company's products and product candidates; risk of loss of significant customers; risks related to legal proceedings, including patent infringement, investigative and antitrust litigation matters and associated costs; changes in government laws and regulations; risk of product recalls and withdrawals; risk regarding the Company’s future financial and operating results and financial position; risk of insufficient capital and cash resources, including insufficient access to available debt and equity financing and revenues from operations, to satisfy all of the Company’s short-term and longer term liquidity and cash requirements and other cash needs, at the times and in the amounts needed; risk of failure to satisfy all financial and other debt covenants and of any default under debt financings; uncertainties related to general economic, political, business, industry, regulatory, financial and market conditions and other unusual items; uncertainties related to general economic, financial, political, business, industry, regulatory and market conditions and other unusual items; and other uncertainties affecting the Company described in the “Risk Factors” section and in other sections included in our Annual Report on Form 10-K, in our Quarterly Reports on Form 10-Q, and in our Current Reports on Form 8-K filed with the Securities Exchange Commission (“SEC”). Given those uncertainties, you should not place undue reliance on these forward-looking statements, which speak only as of the date made. All subsequent forward-looking statements attributable to us or any person acting on our behalf are expressly qualified in their entirety by this cautionary statement. The Company assumes no obligation to update forward-looking statements or outlook or guidance after the date of this press release whether as a result of new information, future events or otherwise, except as may be required by applicable law. This presentation also contains estimates, projections and other information concerning the Company’s business and the markets for the Company’s products and product candidates, including data regarding the estimated size of those markets, and the incidence and prevalence of certain medical conditions. Information that is based on estimates, forecasts, projections, market research, or similar methodologies is inherently subject to uncertainties and actual events, or circumstances may differ materially from events and circumstances reflected in this information. Unless otherwise expressly stated, the Company obtained this industry, business, market and other data from reports, research surveys, clinical trials studies and similar data prepared by market research firms and other third parties, from industry, medical and general publications, from other publicly available information, and from government data and similar sources. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of these securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction. PharmFilm®, Sympazan® and the Aquestive logo are registered trademarks of Aquestive Therapeutics, Inc. The trade name for AQST-109 “Anaphylm” has been conditionally approved by the FDA. Final approval of the Anaphylm™ proprietary name is conditioned on FDA approval of the product candidate, AQST-109. All other registered trademarks referenced herein are the property of their respective owners. © 2023 Property of Aquestive Therapeutics, Inc. 2

Corporate Overview 3

Our Quest Advancing medicines, solving therapeutic problems, and improving lives Our pipeline of product candidates aims to overcome barriers that patients face with existing treatment options and provide new paradigms for treating critical and complex conditions 4
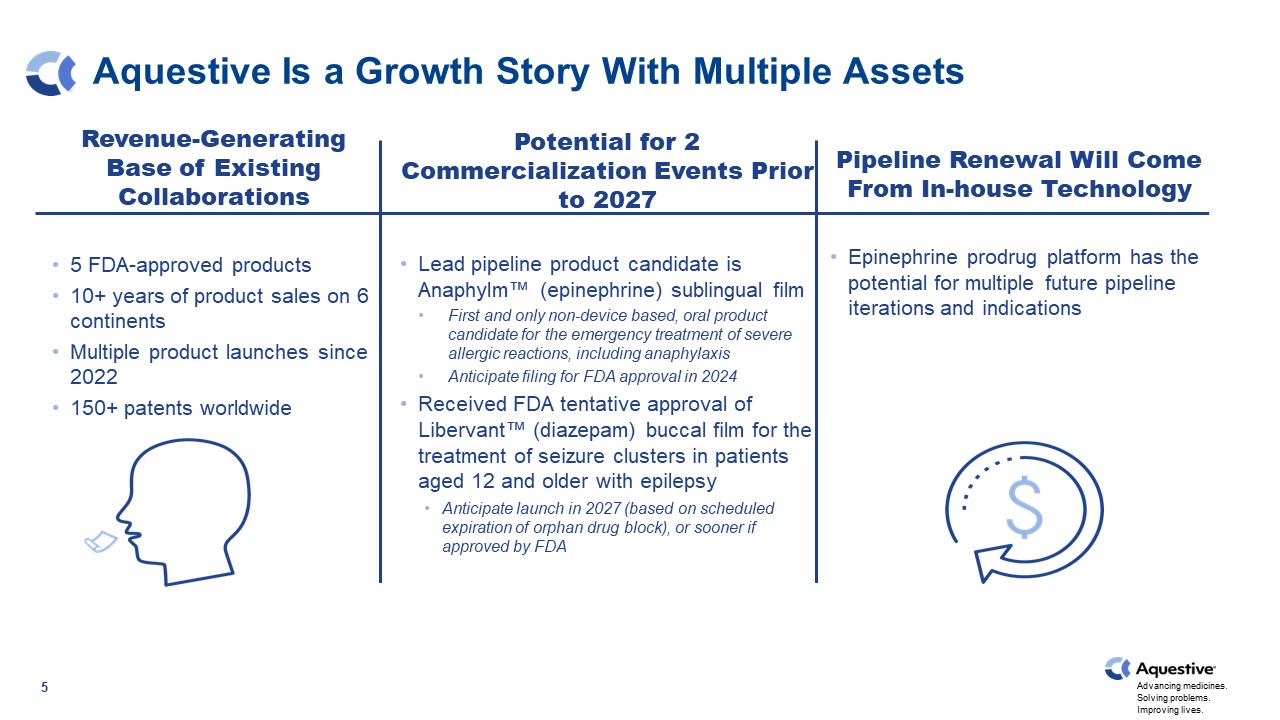
Aquestive Is a Growth Story With Multiple Assets 5 Revenue-Generating Base of Existing Collaborations 5 FDA-approved products 10+ years of product sales on 6 continents Multiple product launches since 2022 150+ patents worldwide Potential for 2 Commercialization Events Prior to 2027 Lead pipeline product candidate is Anaphylm™ (epinephrine) sublingual film First and only non-device based, oral product candidate for the emergency treatment of severe allergic reactions, including anaphylaxis Anticipate filing for FDA approval in 2024 Received FDA tentative approval of Libervant™ (diazepam) buccal film for the treatment of seizure clusters in patients aged 12 and older with epilepsy Anticipate launch in 2027 (based on scheduled expiration of orphan drug block), or sooner if approved by FDA Pipeline Renewal Will Come From In-house Technology Epinephrine prodrug platform has the potential for multiple future pipeline iterations and indications

We Have a Strong Vision for Building the Company In the next five years, we aim to: Grow the existing and ex-U.S. collaboration revenue Secure FDA approval for Anaphylm in the U.S. Launch Libervant in the U.S. in 2027 Utilize our epinephrine prodrug platform for future product launches after Anaphylm and Libervant, if approved by the FDA 6 1. Estimate is based on an orphan drug market exclusivity block until January of 2027 by a competing nasal spray product.

Where You Need It, When You Need It™ 7 Our Core Technology is Branded as PharmFilm®
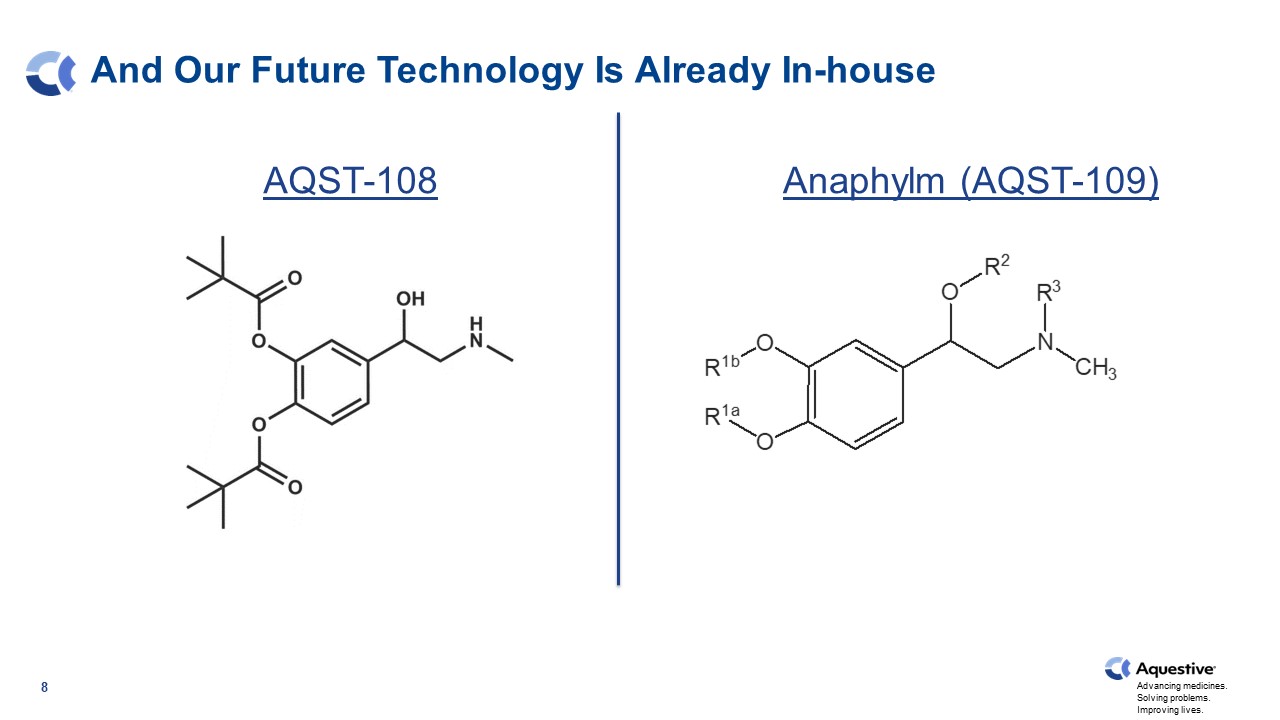
8 And Our Future Technology Is Already In-house AQST-108 Anaphylm (AQST-109)
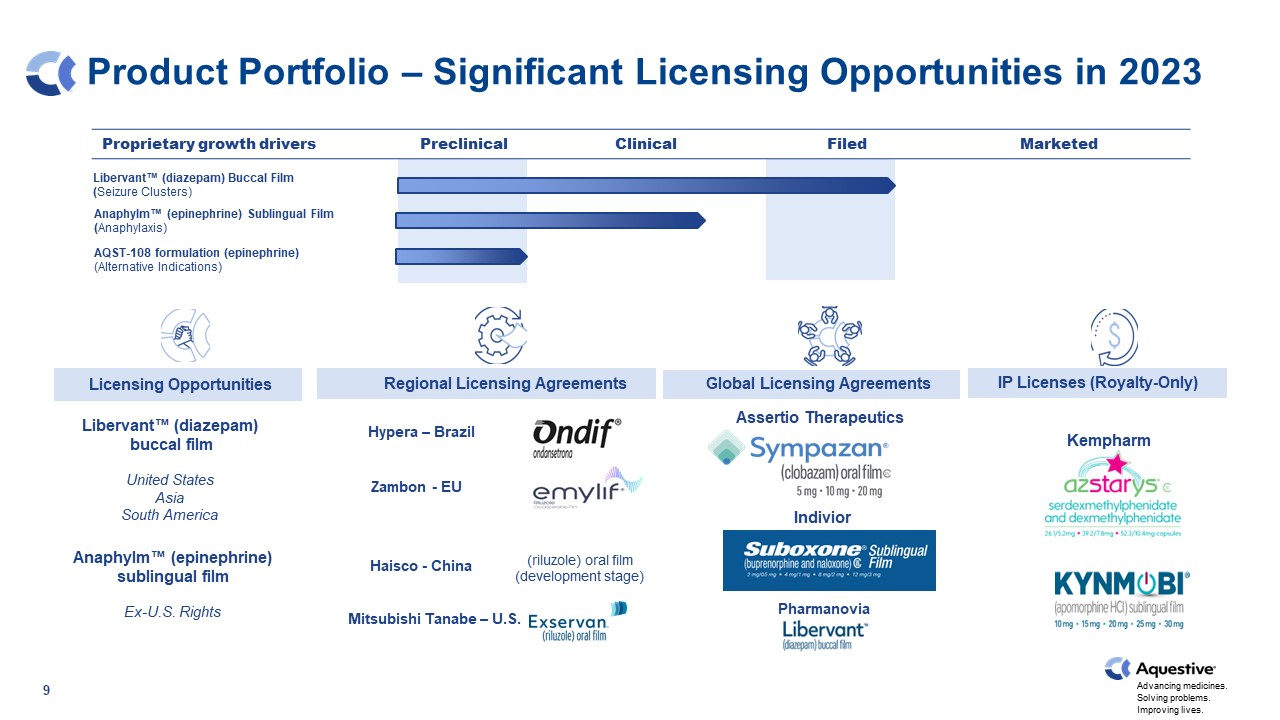
Product Portfolio – Significant Licensing Opportunities in 2023 AQST-108 formulation (epinephrine) (Alternative Indications) Indivior license Zambon EU license / Mitsubishi Tanabe US license Sunovion license Hypera Brazil license Proprietary growth drivers Preclinical Clinical Filed Marketed 9 Hypera – Brazil Haisco - China Libervant™ (diazepam) Buccal Film (Seizure Clusters) Libervant™ (diazepam) buccal film United States Asia South America Anaphylm™ (epinephrine) sublingual film Ex-U.S. Rights Assertio Therapeutics Indivior Kempharm Kempharm Mitsubishi Tanabe – U.S. Hypera – Brazil Zambon - EU Haisco - China Pharmanovia Licensing Opportunities Regional Licensing Agreements IP Licenses (Royalty-Only) Global Licensing Agreements (riluzole) oral film (development stage) Anaphylm™ (epinephrine) Sublingual Film (Anaphylaxis)

Potential for Two Transformative Launches 10 Anaphylm™ (epinephrine) Sublingual Film Potential indication of treatment of severe allergic reactions, including anaphylaxis Anticipate submitting New Drug Application (NDA) by the end of 2024 Estimated Total Addressable Market of ~ $1B 1. Estimated total addressable market is an Aquestive Therapeutic’s calculations based on (i) WAC Price for generic EipPen as of March 2020 and (ii) epinephrine market TRx volume as of December 2022. 2. Estimate is based on an orphan drug market exclusivity block until January of 2027 by a competing nasal spray product. Libervant™ (diazepam) Buccal Film Indicated for the treatment of seizure clusters in patients aged 12 and older with epilepsy Tentatively approved by FDA Expected Launch 2027 1 2

Strong Leadership Team Daniel Barber President, CEO and Director Lori J. Braender SVP, General Counsel Ken Marshall Chief Commercial Officer Mark Schobel Chief Innovation & Technology Officer Ernie Toth Chief Financial Officer 11 Cassie Jung SVP, Operations Steve Wargacki SVP, R&D Gary Slatko Chief Medical Officer Strong Operations & Partnering Team Experienced Science/IP/Development Team Peter Boyd SVP, IT, HR, & Communications

Executed on Key Deliverables in the Last 12 Months Since management change in May 2022, the team has: Raised $47M in non-dilutive financing Signed 3 new licensing agreements on 3 continents Supported two new product launches of licensees Received FDA tentative approval for Libervant Successfully closed 4 litigation cases Continued to advance Anaphylm towards an NDA submission Reduced existing debt by 17.6% 12 1 1. As of December 31, 2022, the outstanding debt was $51,500,000. As of March 31, 2023, the outstanding debt was $42,414,025, resulting in a difference of $9,085,975, or 17.6%.

We Are Now Focused on the Next Chapter Over the next 90 days, the Company aims to: Continue to strengthen the balance sheet Refinance the existing debt (anticipate standard 5 yr, 3 yr i/o deal) Out-license Libervant in China Submit pivotal protocol for Anaphylm to the FDA for review 13

14 Potential Near-term Milestones Targeted Complete debt refinancing Start Anaphylm pivotal study License Libervant in China H1 2024 Receive Anaphylm topline pivotal data AQST-108 first-in-human data License ex-U.S. rights to Anaphylm H2 2023 Anaphylm pre-NDA meeting Anaphylm NDA submission Ex-U.S. Libervant launches begin Epinephrine prodrug pipeline additions H2 2024

Anaphylm™ (epinephrine) Sublingual Film 15

Anaphylaxis Market Overview 16

Anaphylaxis: A Serious Systemic Hypersensitivity Reaction That is Usually Rapid in Onset And May Be Fatal 17 1. Turner PJ, et al. World Allergy Org J. 2019;12100066. 2. FARE, 2022; https://www.foodallergy.org/resources/facts-and-statistics. 3. Fromer L. The American Journal of Medicine (2016);129, 1244-1250. As many as 32 million people in the United States are at chronic risk for acute anaphylactic episodes 52% of patients in a nationwide patient survey who had previously experienced anaphylaxis had never received an epinephrine auto-injector prescription Direct costs of anaphylaxis have been estimated at $1.2 billion per year 60% of respondents in same patient survey did not have an epinephrine auto-injector currently available 1 2 3 3 3

Advancing medicines. Solving problems. Improving lives. 18 Treatment of Anaphylaxis – Epinephrine 0.10 mg (for children 16.5 to 33 pounds) — AUVI-Q® brand only 0.15 mg (for children under 66 pounds) 0.3 mg (for children and adults over 66 pounds) 1 Epinephrine is the first line of treatment for anaphylaxis Epinephrine is the only medication proven to stop a life-threatening allergic reaction 1. Epinephrine in the Management of Anaphylaxis. Brown JC, Simons E, Rudders SA. J Allergy Clin Immunol Pract. 2020 Apr;8(4):1186-1195. doi: 10.1016/j.jaip.2019.12.015 PMID: 32276687. 2. EpiPen® Package Insert. 2 Epinephrine dosage (current medication delivery systems): 0.3-0.5 mg intramuscularly (IM) or subcutaneously Children’s dosage is weight based: 2 A second dose of epinephrine can be given as needed

Epinephrine Market 4M TRx in 2022 1. Symphony Health Data April 2023. The 2022 Epinephrine market surpassed 4 million TRx and has rebounded to historical highs following a downturn due to generics and the Covid-19 pandemic. TRx counts have exceeded prior year for 9 consecutive months. 1

Generic Market With High Levels of Dissatisfaction and Unmet Need 1. KOL feedback; Aquestive Market Research. 2. Fromer L. The American Journal of Medicine (2016);129, 1244-1250. 3. Warren et al. Ann Allergy Asthma Immunol (2018). 4. Brooks et al. Ann Allergy Asthma Immunol (2017). 5. Asthma and Allergy Foundation of America Patient Survey Report (2019). 6. El Turki et al. EmergMed J (2017). Current Standard of Care = Large, Needle Based Injectors Numerous Studies and Patient Surveys Articulate Significant Dissatisfaction with Current Offerings Right place, right time <50% of patients carry their EpiPen® – often due to hassle factor Refusal of treatment 25-50% of patients refuse treatment with EpiPen® – often due to needle reluctance Time to treat post exposure 60% of patients/caregivers delay treatment – often due to needle reluctance Failed administration in the field 23-35% of patients and caregivers fail to dose correctly Oversized devices Hard to carry Medical guidelines recommend always having 2 doses on hand Needle based High prevalence of needle phobia (especially in children) Not always intuitive to use Even trained health care providers have been shown to incorrectly inject 3,4,5 1 1 6 2
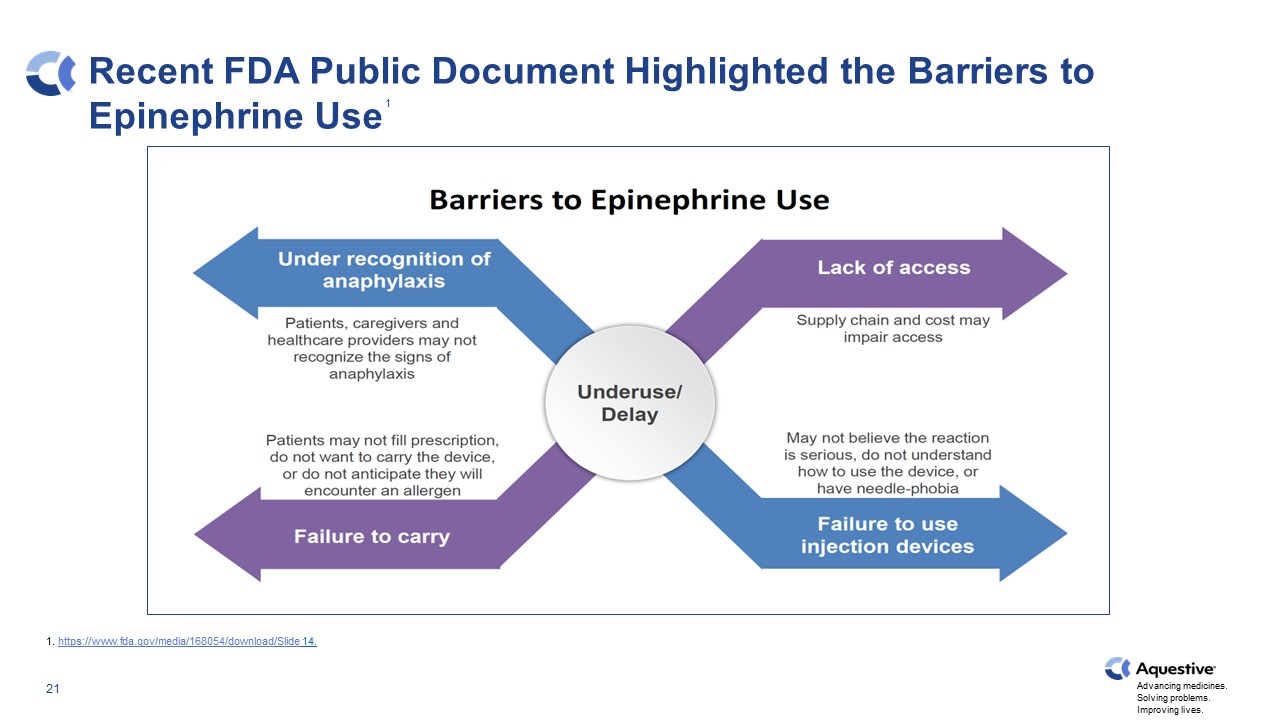
Recent FDA Public Document Highlighted the Barriers to Epinephrine Use 1 1. https://www.fda.gov/media/168054/download/Slide 14.

Anaphylm™ (epinephrine) Sublingual Film First and only non-device based, orally delivered epinephrine product candidate Fast absorption into the bloodstream Non-device administration Portability + +
PharmFilm® Platform Projecting Robust Stability Chemical Stability > 2 years room temperature > 6 months accelerated conditions Successful Scale Up Executed Environmental Stability Light resistant Water resistant Withstands extreme cold conditions High temperature excursions while maintaining shelf-life

Patents/Patent Applications Extending into 2042 Title Patent Status ENHANCED DELIVERY EPINEPHRINE COMPOSITIONS Granted U.S. Patent 11,191,737 (5/4/2037) 8 Foreign applications Priority date: May 5, 2016 Possible patent term to 2037 ENHANCED DELIVERY EPINEPHRINE AND PRODRUG COMPOSITIONS 2 U.S. applications 8 Foreign applications Priority date: May 4, 2017 Possible patent term to 2037 PRODRUG COMPOSITIONS AND METHODS OF TREATMENT 2 U.S. applications 1 Foreign application Priority date: late 2019 Possible patent term to 2041 PHARMACEUTICAL COMPOSITIONS WITH ENHANCED STABILITY PROFILES 1 U.S. application Priority date: October 2021 Possible patent term to 2042

Competitive Product Summary ORAL AUTO INJECTOR INTRA NASAL Company Brand Anaphylm EpiPen/Generic Adrenaclick® Auvi-Q® Symjepi® neffy® Utuly™ N/A Administration Sublingual Auto-Injector Auto-Injector Auto-Injector Syringe Device Nasal Spray Nasal Spray Nasal Spray Dosing (Adult/Jr) TBD 0.3 / 0.15 mg 0.3 / 0.15 mg 0.3 / 0.15 / 0.10 mg 0.3 / 0.15 mg 2 mg 6.6 mg Not Reported Market Position 1st & Only Oral 90%+ Share Negligible <10% Negligible 1 Dose per Device 2 Doses per Device Potentially 3rd Nasal to Market Regulatory Status (FDA) Expected NDA Filing 2024 Approved/Marketed Filed Fall ‘22 Expected Filing 1H ‘23 Expected NDA Filing 2023 1 2 3 2 2, 4 2 1. The data presented on this slide are based on cross-study comparisons and are not based on any heat-to-head trials as a result, comparability may be limited/inaccurate. Cross-study comparisons are inherently limited and may suggest misleading similarities or difference. 2. Pending FDA Review. 3. VIATRIS: Formerly Mylan. 4. US WorldMeds markets for Adamis.

Anaphylm:Product Development 26

Scientific Advisory Board David Bernstein, MD University of Cincinnati Carlos Camargo, MD Harvard Medical School David M. Fleischer, MD Children’s Hospital Colorado David Golden, MD Sinai Hospital, Baltimore Matthew Greenhawt, MD Children’s Hospital Colorado Ruchi Gupta, MD, MPH Northwestern Jay Lieberman, MD University of Tennessee John Oppenheimer, MD University of Medicine and Dentistry of NJ - Rutgers

Anaphylm Clinical Trials to Date Study Description Study Status N 210010 First-in-Human (FIH), Single Ascending Dose (SAD) study to evaluate safety and tolerability, as well as pharmacokinetic (PK) performance and pharmacodynamic (PD) effect, of DESF (Anaphylm) Complete 44 EPIPHAST Part 1 Evaluate multiple formulations and strengths of DESF (Anaphylm) Benchmark against epinephrine 0.5mg manual intramuscular (IM) injection Complete 35 Part 2 Confirm benchmarking vs. epinephrine 0.3mg manual IM injection Evaluate intrasubject variability and adequacy of washout period Complete 24 Part 3 Characterize conditions of use and effect of use errors (different saliva hold times and directly swallowing film) Film performance after ingestion of sticky substance (peanut butter) Complete 24 EPIPHAST II Characterize: repeat dose performance of DESF (Anaphylm) performance against EpiPen Complete 24 AQ109102 Evaluate: differences in PK and PD results based on changes to administration instructions additional repeat dose data on DESF (Anaphylm) performance of various approved auto-injectors Complete 30 AQ109106 Evaluate differences in PK and PD results based on changes to administration instructions Complete 35 AQ109103 Further characterization of PK performance and PD effect of DESF (Anaphylm) to inform pivotal study design Ongoing 24

Agency Interactions on Anaphylm Program to Date Interaction Key Takeaways Pre-IND Meeting (December 1, 2021) 505(b)(2) NDA regulatory approval pathway acceptable (no efficacy trials required) Bracket PK to 0.3mg IM and safety to 0.5mg IM Evaluate potential for extrinsic factors to impact DESF (ANAPHYLM) absorption Stability Excursion Protocol Review (July 29, 2022) Design and planned analysis of the proposed excursions are reasonable and can be expected to provide data to support product and patient labeling End Of Phase 2 (EOP2) Meeting-CMC Meeting Feedback (October 4, 2022) Proposed Chemistry Manufacturing and Controls (CMC) package for both active pharmaceutical ingredients (API) and DESF (Anaphylm) considered sufficient and reasonable for future NDA filing Nonclinical Study Plans (October 11, 2022) Aligned with FDA on NDA, enabling nonclinical toxicology package EOP2 Meeting (November 15, 2022) Reaffirmed 505(B)(2) regulatory approval pathway acceptable (no efficacy trials required) Modified bracketing strategy to compare PK performance to IM and autoinjectors Use during conditions of anaphylaxis to be considered in overall risk/benefit profile FDA Response to General Correspondence (March 1, 2023) FDA agreed to review pivotal protocol FDA agreed to separate meeting to align on risk/benefit characterization after pivotal study alignment
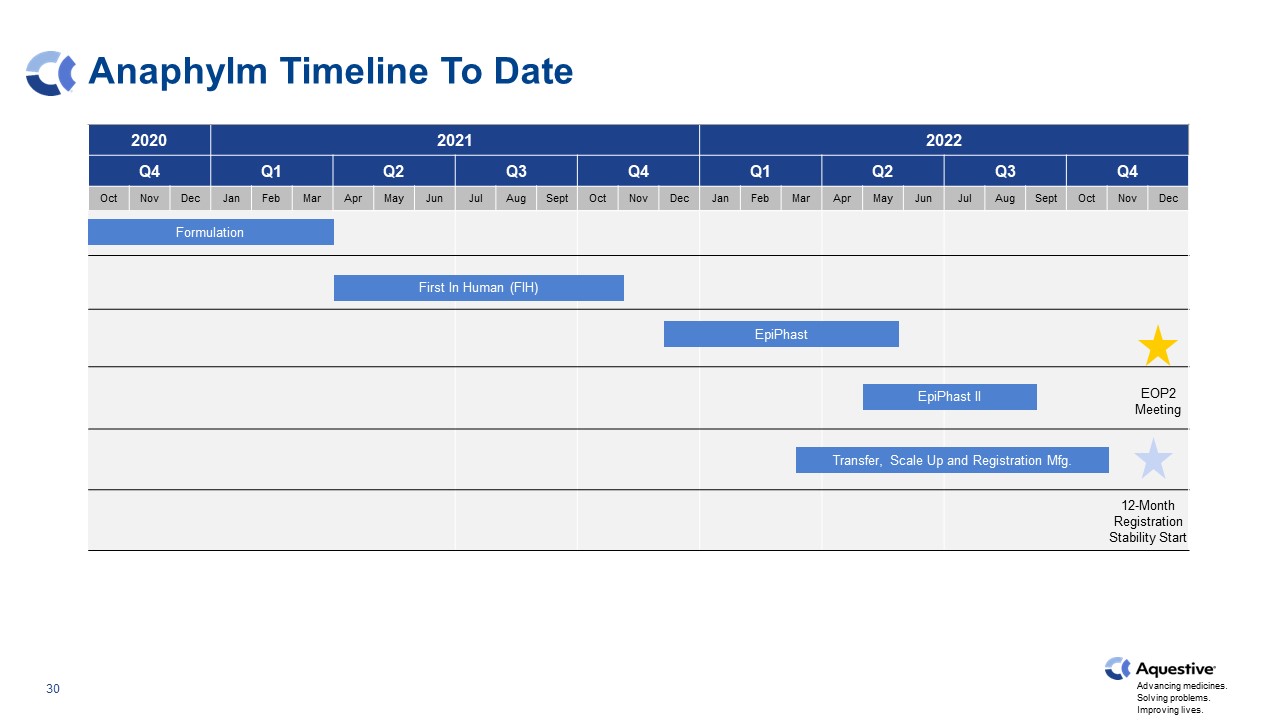
Anaphylm Timeline To Date 2020 2020 2021 2023 2022 Q4 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Oct Nov Dec Jan Feb Mar Apr May Jun Jul Aug Sept Oct Nov Dec Jan Feb Mar Apr May Jun Jul Aug Sept Oct Nov Dec EpiPhast II First In Human (FIH) Transfer, Scale Up and Registration Mfg. Formulation EpiPhast EOP2 Meeting 12-Month Registration Stability Start
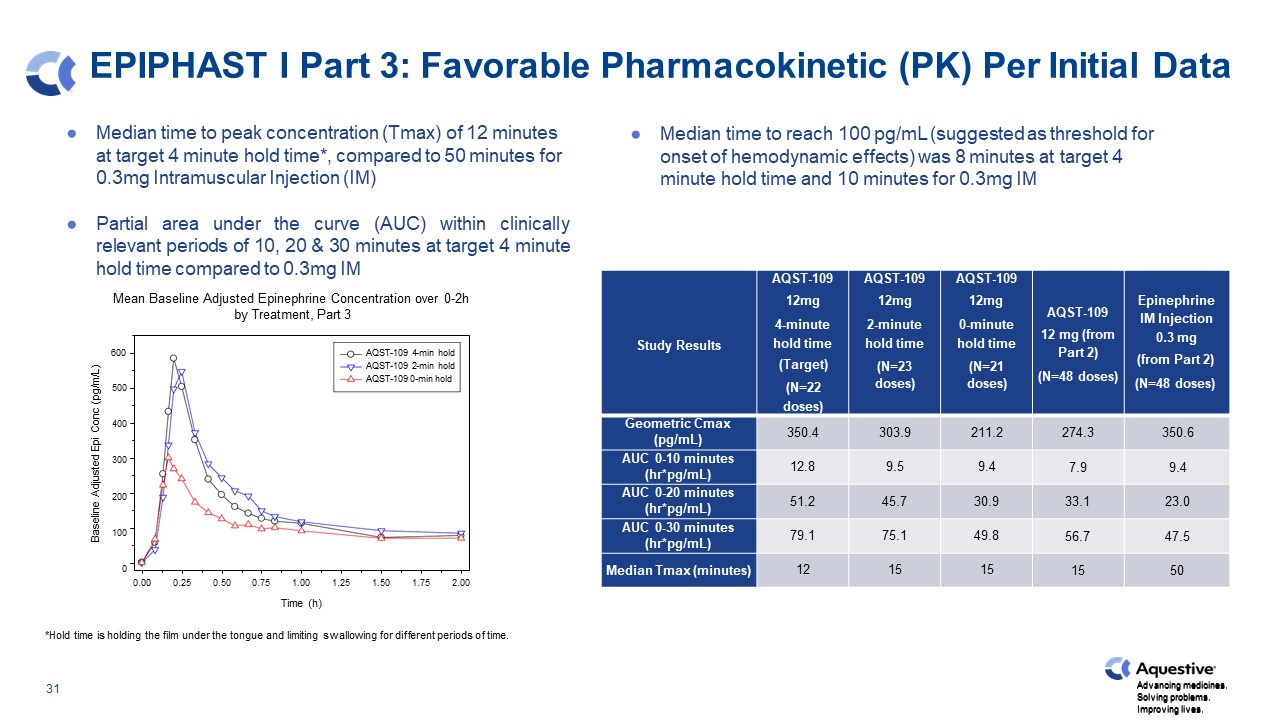
EPIPHAST I Part 3: Favorable Pharmacokinetic (PK) Per Initial Data Median time to peak concentration (Tmax) of 12 minutes at target 4 minute hold time*, compared to 50 minutes for 0.3mg Intramuscular Injection (IM) Partial area under the curve (AUC) within clinically relevant periods of 10, 20 & 30 minutes at target 4 minute hold time compared to 0.3mg IM Mean Baseline Adjusted Epinephrine Concentration over 0-2h by Treatment, Part 3 Median time to reach 100 pg/mL (suggested as threshold for onset of hemodynamic effects) was 8 minutes at target 4 minute hold time and 10 minutes for 0.3mg IM *Hold time is holding the film under the tongue and limiting swallowing for different periods of time. Study Results AQST-109 12mg 4-minute hold time (Target) (N=22 doses) AQST-109 12mg 2-minute hold time (N=23 doses) AQST-109 12mg 0-minute hold time (N=21 doses) AQST-109 12 mg (from Part 2) (N=48 doses) Epinephrine IM Injection 0.3 mg (from Part 2) (N=48 doses) Geometric Cmax (pg/mL) 350.4 303.9 211.2 274.3 350.6 AUC 0-10 minutes (hr*pg/mL) 12.8 9.5 9.4 7.9 9.4 AUC 0-20 minutes (hr*pg/mL) 51.2 45.7 30.9 33.1 23.0 AUC 0-30 minutes (hr*pg/mL) 79.1 75.1 49.8 56.7 47.5 Median Tmax (minutes) 12 15 15 15 50 0.75 1.00 1.25 Time (h) 0.00 0.25 0.50 1.50 1.75 2.00 Baseline Adjusted Epi Conc (pg/m/L) Advancing medicines. Solving problems. Improving lives. 500 400 300 200 100 0 600 AQST-109 4-min hold AQST-109 2-min hold AQST-109 0-min hold
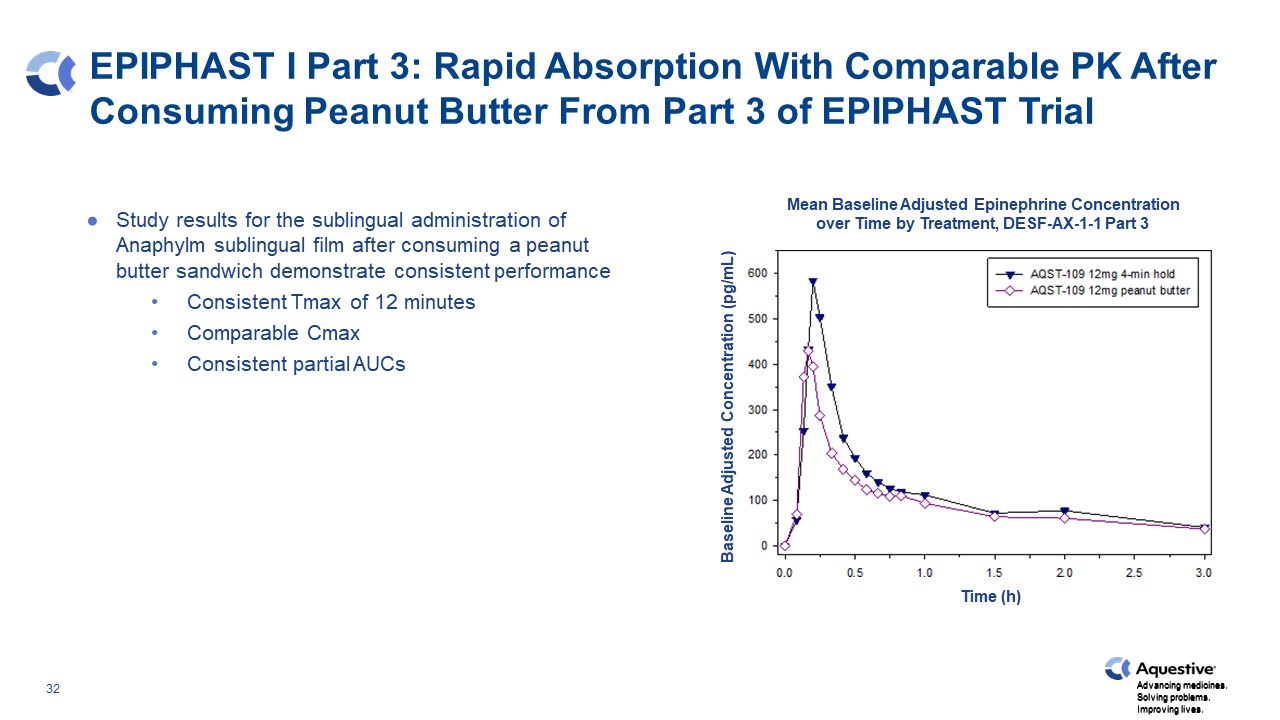
EPIPHAST I Part 3: Rapid Absorption With Comparable PK After Consuming Peanut Butter From Part 3 of EPIPHAST Trial Study results for the sublingual administration of Anaphylm sublingual film after consuming a peanut butter sandwich demonstrate consistent performance Consistent Tmax of 12 minutes Comparable Cmax Consistent partial AUCs Mean Baseline Adjusted Epinephrine Concentration over Time by Treatment, DESF-AX-1-1 Part 3 Baseline Adjusted Concentration (pg/mL) Time (h) Advancing medicines. Solving problems. Improving lives.
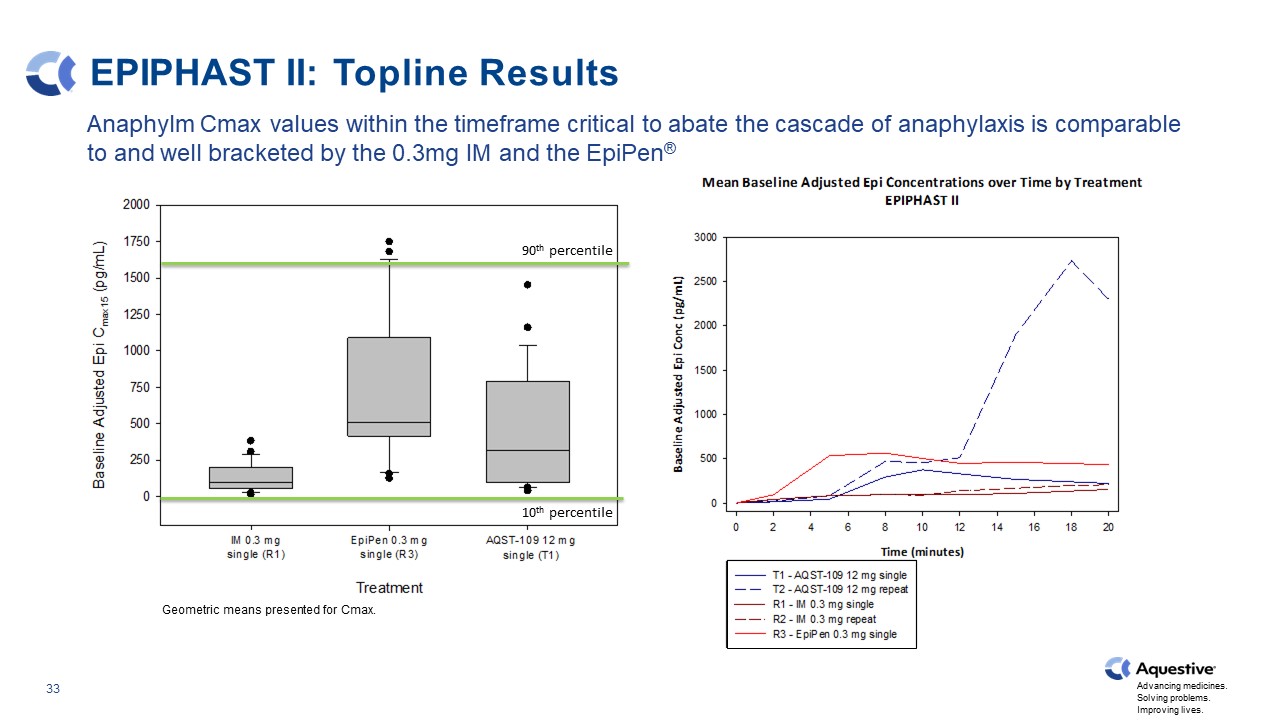
EPIPHAST II: Topline Results Geometric means presented for Cmax. Anaphylm Cmax values within the timeframe critical to abate the cascade of anaphylaxis is comparable to and well bracketed by the 0.3mg IM and the EpiPen® 90th percentile 10th percentile
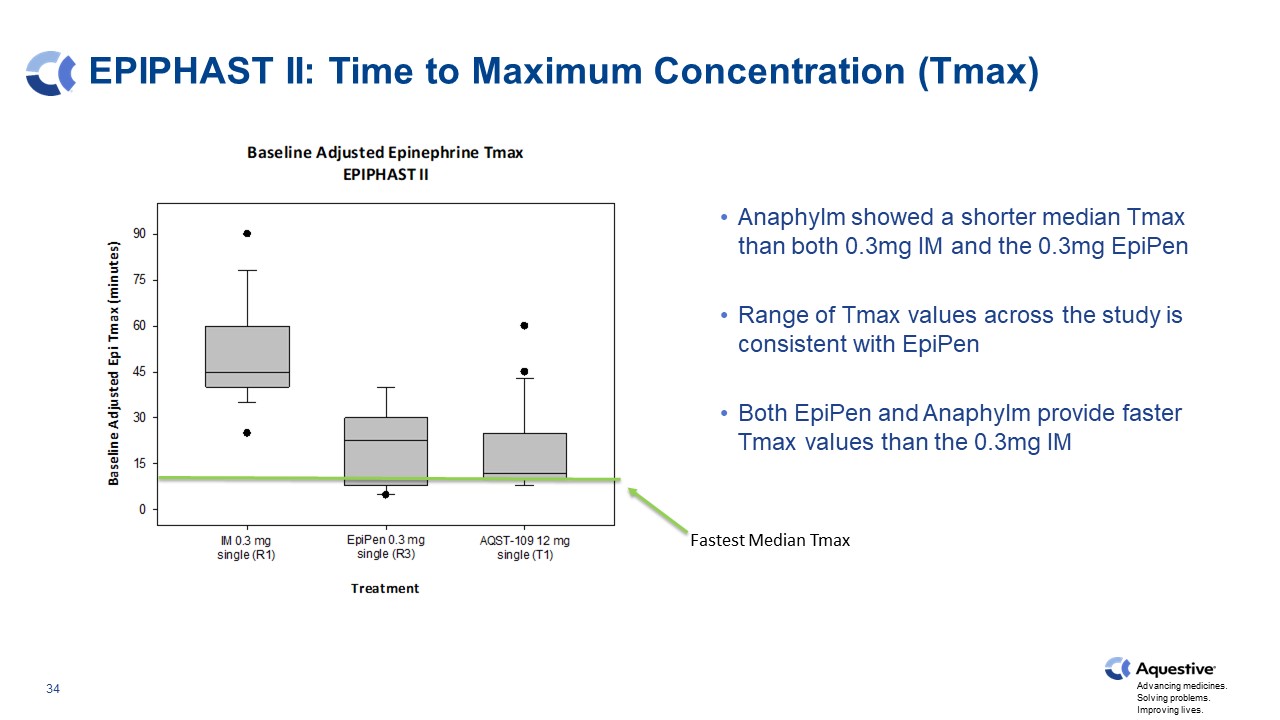
EPIPHAST II: Time to Maximum Concentration (Tmax) Anaphylm showed a shorter median Tmax than both 0.3mg IM and the 0.3mg EpiPen Range of Tmax values across the study is consistent with EpiPen Both EpiPen and Anaphylm provide faster Tmax values than the 0.3mg IM Fastest Median Tmax
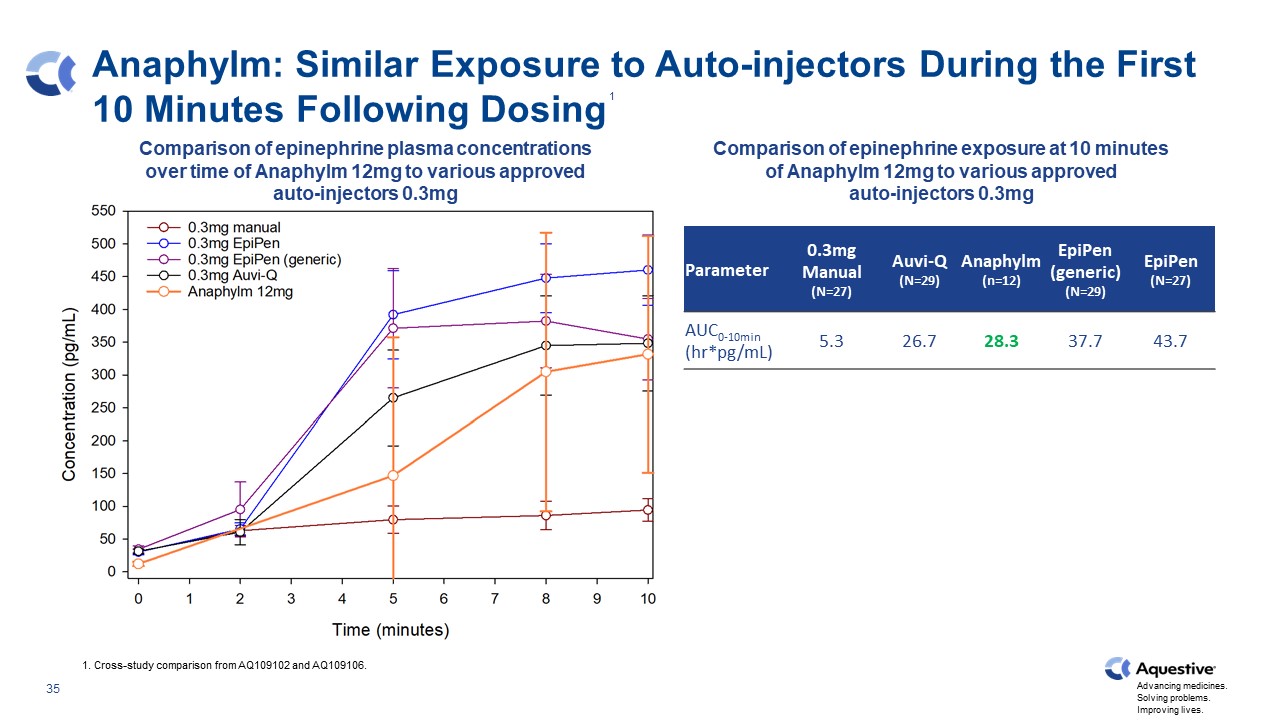
Anaphylm: Similar Exposure to Auto-injectors During the First 10 Minutes Following Dosing Parameter 0.3mg Manual (N=27) Auvi-Q (N=29) Anaphylm (n=12) EpiPen (generic) (N=29) EpiPen (N=27) AUC0-10min (hr*pg/mL) 5.3 26.7 28.3 37.7 43.7 1. Cross-study comparison from AQ109102 and AQ109106. Comparison of epinephrine plasma concentrations over time of Anaphylm 12mg to various approved auto-injectors 0.3mg Comparison of epinephrine exposure at 10 minutes of Anaphylm 12mg to various approved auto-injectors 0.3mg 1

Anaphylm Data Brackets Existing Products to 45 Minutes FDA recommended bracketing between the exposures produced by auto-injectors and manual injection across a range of relevant time points characterized as pAUC. Manual Injection Level and Below Auto-injector Levels Anaphylm Critical early phase similar to auto-injectors Not statistically different from manual injection and supported by PD data Partial AUC (%CV) Manual Injection Anaphylm AUC0-45min 94.4 (75.6%) 117.0 (93.6%) AUC0-50min 117.2 (72.3%) 123.3 (92.4%) AUC0-60min 160.4 (66.2%) 135.1 (90.1%) 1 1. Bracketing end points subject to alignment with FDA. Cross-study comparison from AQ109102 and AQ109106.

Key PK Parameters Compare Favorably to Existing Treatments Bars above show highest and lowest 75% quartile ranges of approved products. 1. Cross-study comparison of AQ109102 and AQ109106. Anaphylm 12mg provides a consistently fast Tmax with median and mean Cmax levels bracketed by the current FDA approved products. Median and Mean Cmax Median Tmax Tmax (minutes) Cmax (pg/mL) 1 1

Clinically Favorable PD from Anaphylm Anaphylm demonstrates a rapid increase in systolic blood pressure (SBP), pulse and diastolic blood pressure (DBP) within 2 minutes. Injected epinephrine produces moderate increases in SBP and pulse with no measurable effect on DBP. Mean Baseline Adjusted Changes in Systolic Blood Pressure Following Administration Mean Baseline Adjusted Changes in Pulse Following Administration 1. Cross-study comparison of AQ109102 and AQ109106. 1
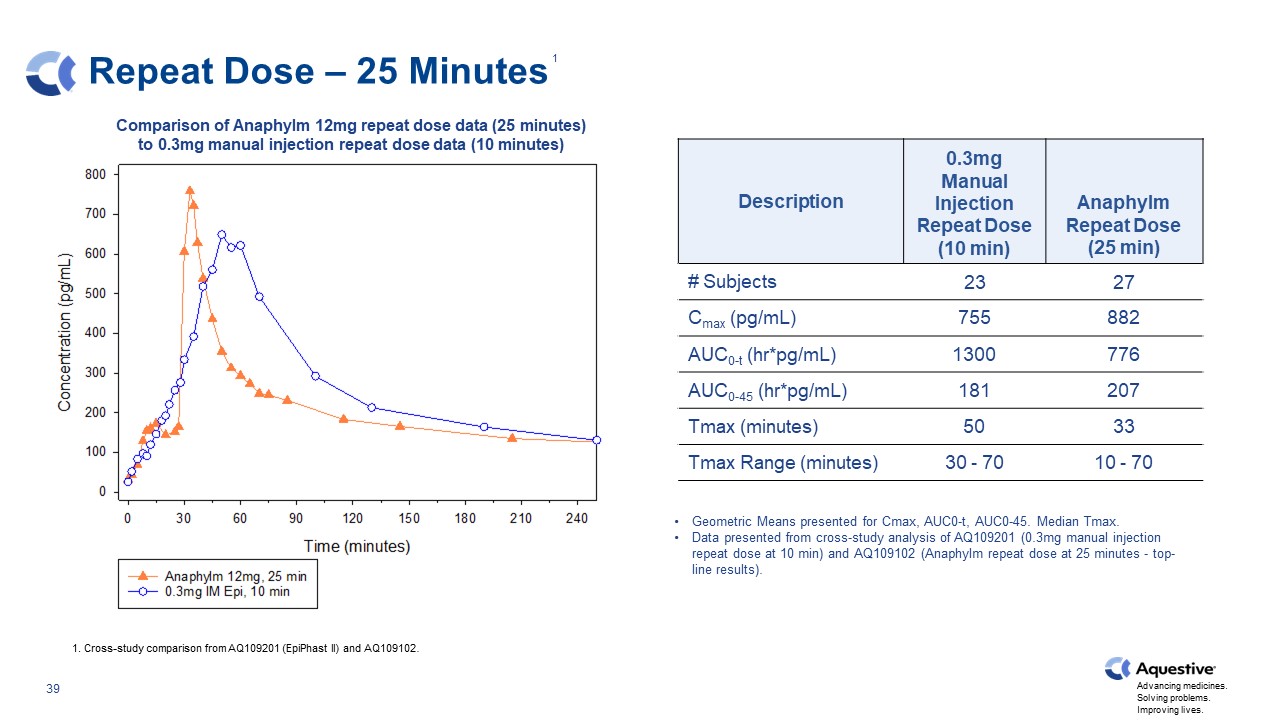
Repeat Dose – 25 Minutes Description 0.3mg Manual Injection Repeat Dose (10 min) Anaphylm Repeat Dose (25 min) # Subjects 23 27 Cmax (pg/mL) 755 882 AUC0-t (hr*pg/mL) 1300 776 AUC0-45 (hr*pg/mL) 181 207 Tmax (minutes) 50 33 Tmax Range (minutes) 30 - 70 10 - 70 Geometric Means presented for Cmax, AUC0-t, AUC0-45. Median Tmax. Data presented from cross-study analysis of AQ109201 (0.3mg manual injection repeat dose at 10 min) and AQ109102 (Anaphylm repeat dose at 25 minutes - top-line results). 1. Cross-study comparison from AQ109201 (EpiPhast II) and AQ109102. Comparison of Anaphylm 12mg repeat dose data (25 minutes) to 0.3mg manual injection repeat dose data (10 minutes) 1

Summary and Next Steps AQ109102 compared Anaphylm to multiple epinephrine auto-injectors Confirmation of target range between existing reference listed drug (RLD) epinephrine injections AQ109106 focused on administration instructions Confirmation of Anaphylm Cmax comparability Confirmation that Anaphylm early pAUC parameters are bracketed by other RLDs Next Steps Refine administration instructions in ongoing pilot study (AQ109103) Finalize pivotal study protocol – expect to submit for FDA review/alignment in Q3 2023 Expect to begin execution of pivotal study in Q4 2023

Regulatory Path Potentially De-risked by Recent Epinephrine Nasal Spray FDA Advisory Committee Meeting VOTING RESULTS 16:6 VOTING RESULTS 17:5 VOTE: Do the PK/PD results support a favorable benefit-risk assessment for ARS-1 in adults for the emergency treatment of allergic reactions (Type 1) and anaphylaxis? a. If not, what additional data are needed? VOTE: Do the PK/PD results support a favorable benefit-risk assessment for ARS-1 in children (<18 years of age) > 30 kg for the emergency treatment of allergic reactions (Type 1) and anaphylaxis? a. If not, what additional data are needed? VOTING QUESTION 1 1. https://www.fda.gov/media/168054/download.
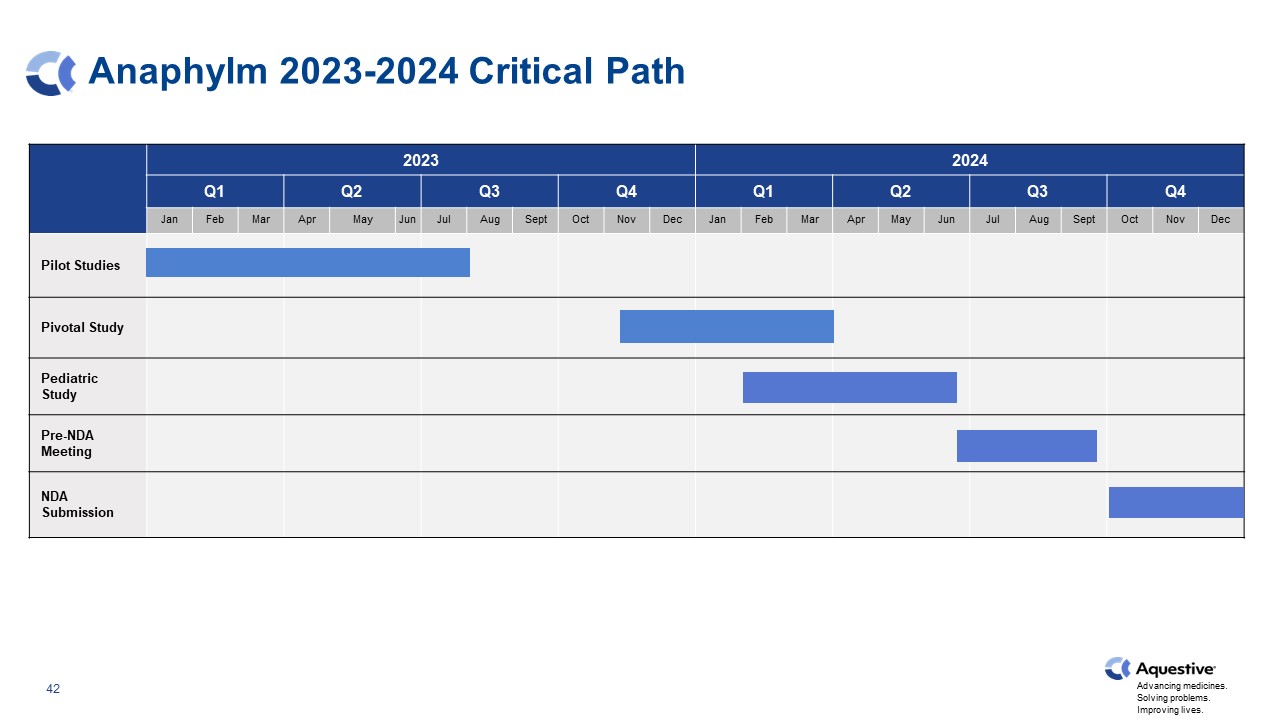
Anaphylm 2023-2024 Critical Path 2023 2023 2024 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Jan Feb Mar Apr May Jun Jul Aug Sept Oct Nov Dec Jan Feb Mar Apr May Jun Jul Aug Sept Oct Nov Dec Pilot Studies Pivotal Study Pediatric Study Pre-NDA Meeting NDA Submission

Libervant™ (diazepam) Buccal Film 43

Laxer, Ketal, The consequences of Refractory Epilepsy and its treatment; Epilepsy & Behavior, Vol 37, Aug 2014, Pgs 59 –70; https://doi.org/10.1016/j.yebeh.2014.05.031, 2. Triangle Insights Group (2017). Synthesis of Epilepsy (ARS) Primary Research. Internal Aquestive report: unpublished, 3. Epilepsy Data and Statistics | CDC - 1.2% of the US population had active epilepsy (95% CI* = 1.1-1.4). This is about 3.4 million people with epilepsy nationwide: 3 million adults and 470,000 children. 4. Breakthrough Seizures: Causes, Treatment, and Prevention (healthline.com) - About 1 in 3 people with epilepsy experience breakthrough seizures. 5. 2022 Symphony Data shows 420,000 labeled rescue rxs, if a patient fills 2.5 times a year that’s 168,000 patients. 6. Seizure visits to ED: ht t p s:/ /www.ncbi.nlm.nih.gov/pmc/articles/PMC2657249/. The Unmet Need in Refractory Seizures… ~1M Suffer from uncontrolled, refractory seizures Epilepsy patients1 1M Seizures Account for EMERGENCY DEPARTMENT visits annually6 of patients with refractory seizures will not interact with the historically available Treatment2,3,4,5 ~85%
Current Treatments are Either Rectal or Intra Nasal Options

Seizure Rescue Market The seizure rescue market continues to grow with new products being promoted. Based upon publicly available data, Valtoco® has flat to growing market share in all age groups in which it competes. 1. Symphony Health Data April 2023. 1

Seizure Rescue Market by Age 1. Symphony Health Data April 2023. 1
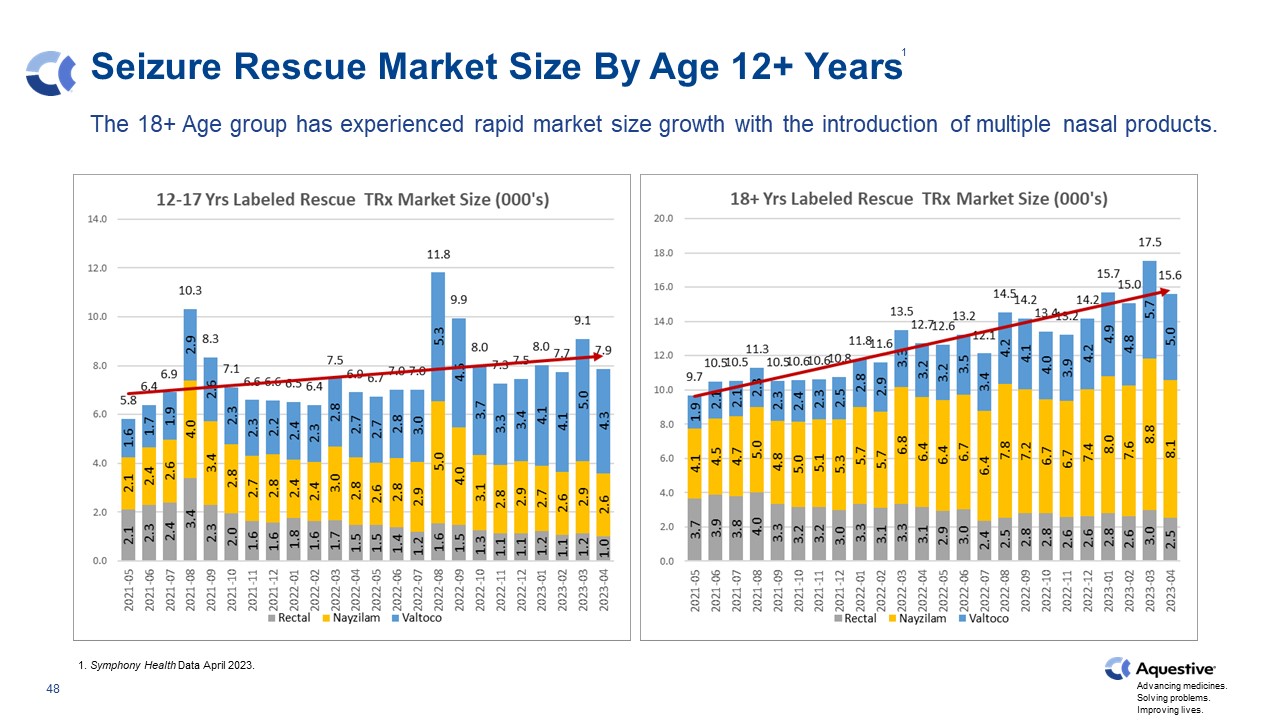
Seizure Rescue Market Size By Age 12+ Years The 18+ Age group has experienced rapid market size growth with the introduction of multiple nasal products. 1. Symphony Health Data April 2023. 1
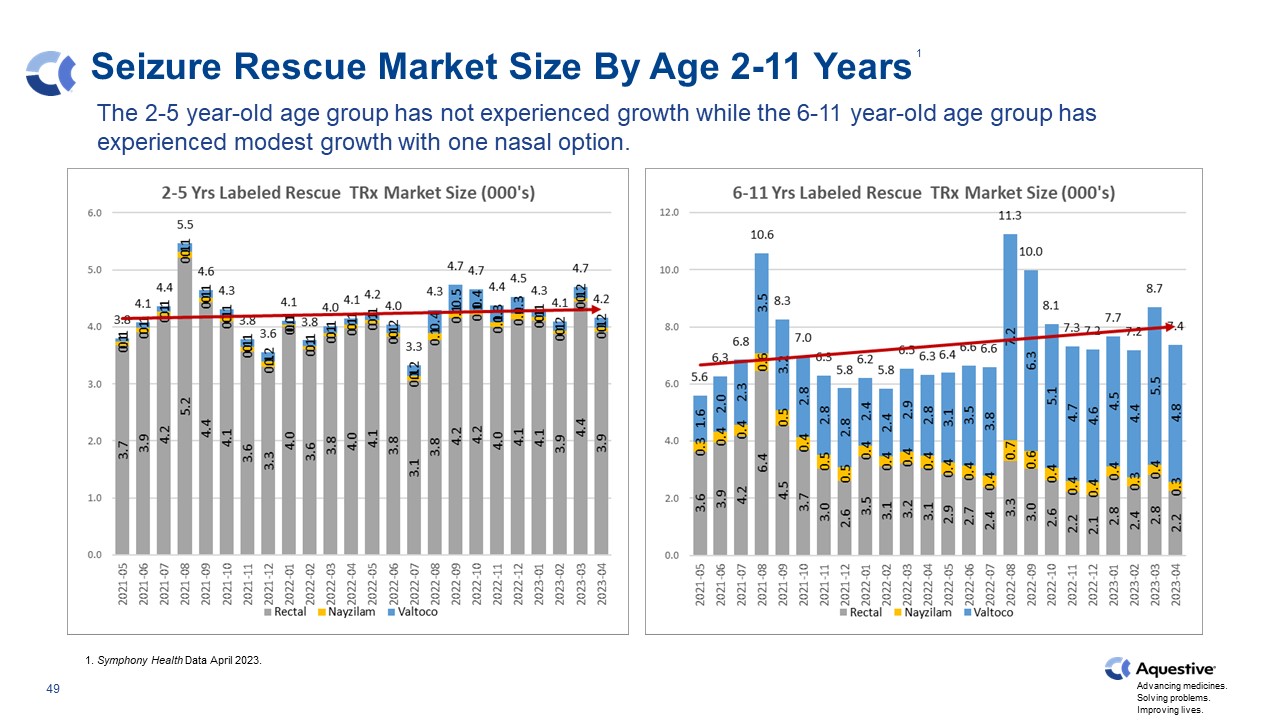
Seizure Rescue Market Size By Age 2-11 Years The 2-5 year-old age group has not experienced growth while the 6-11 year-old age group has experienced modest growth with one nasal option. 1. Symphony Health Data April 2023. 1
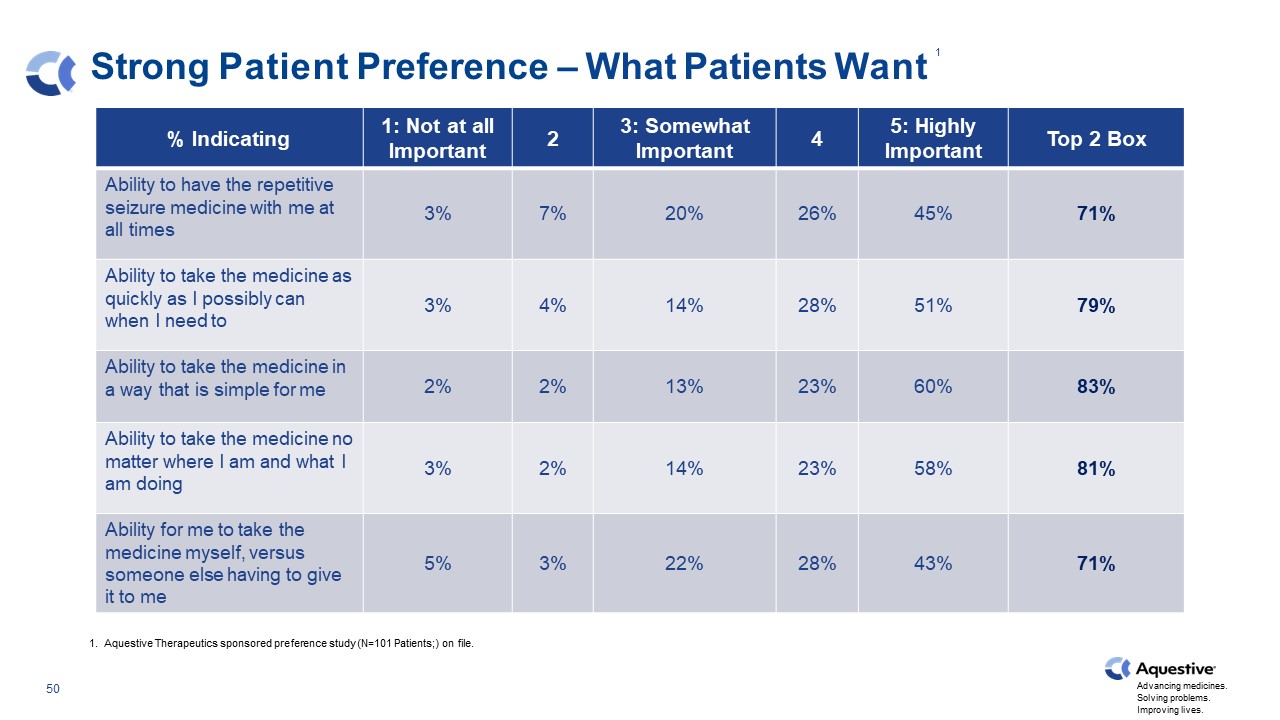
Strong Patient Preference – What Patients Want % Indicating 1: Not at all Important 2 3: Somewhat Important 4 5: Highly Important Top 2 Box Ability to have the repetitive seizure medicine with me at all times 3% 7% 20% 26% 45% 71% Ability to take the medicine as quickly as I possibly can when I need to 3% 4% 14% 28% 51% 79% Ability to take the medicine in a way that is simple for me 2% 2% 13% 23% 60% 83% Ability to take the medicine no matter where I am and what I am doing 3% 2% 14% 23% 58% 81% Ability for me to take the medicine myself, versus someone else having to give it to me 5% 3% 22% 28% 43% 71% 1. Aquestive Therapeutics sponsored preference study (N=101 Patients;) on file. 1
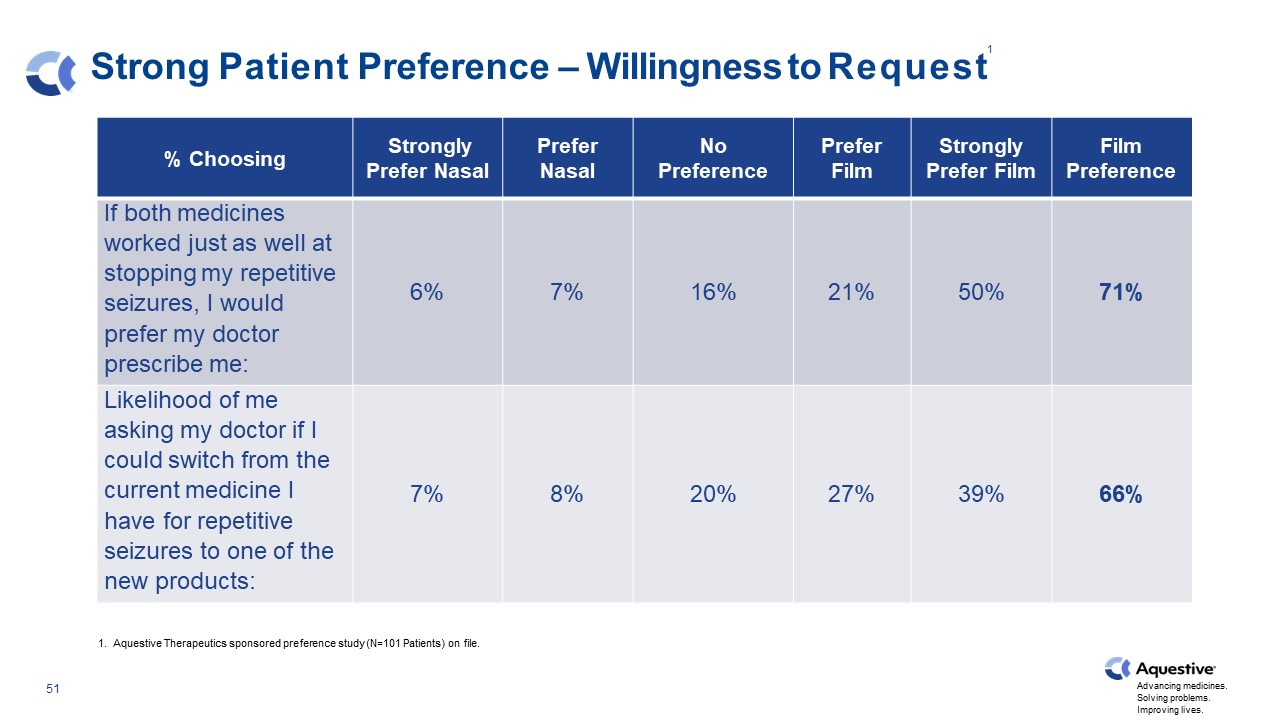
Strong Patient Preference – Willingness to Request % Choosing Strongly Prefer Nasal Prefer Nasal No Preference Prefer Film Strongly Prefer Film Film Preference If both medicines worked just as well at stopping my repetitive seizures, I would prefer my doctor prescribe me: 6% 7% 16% 21% 50% 71% Likelihood of me asking my doctor if I could switch from the current medicine I have for repetitive seizures to one of the new products: 7% 8% 20% 27% 39% 66% 1. Aquestive Therapeutics sponsored preference study (N=101 Patients) on file. 1
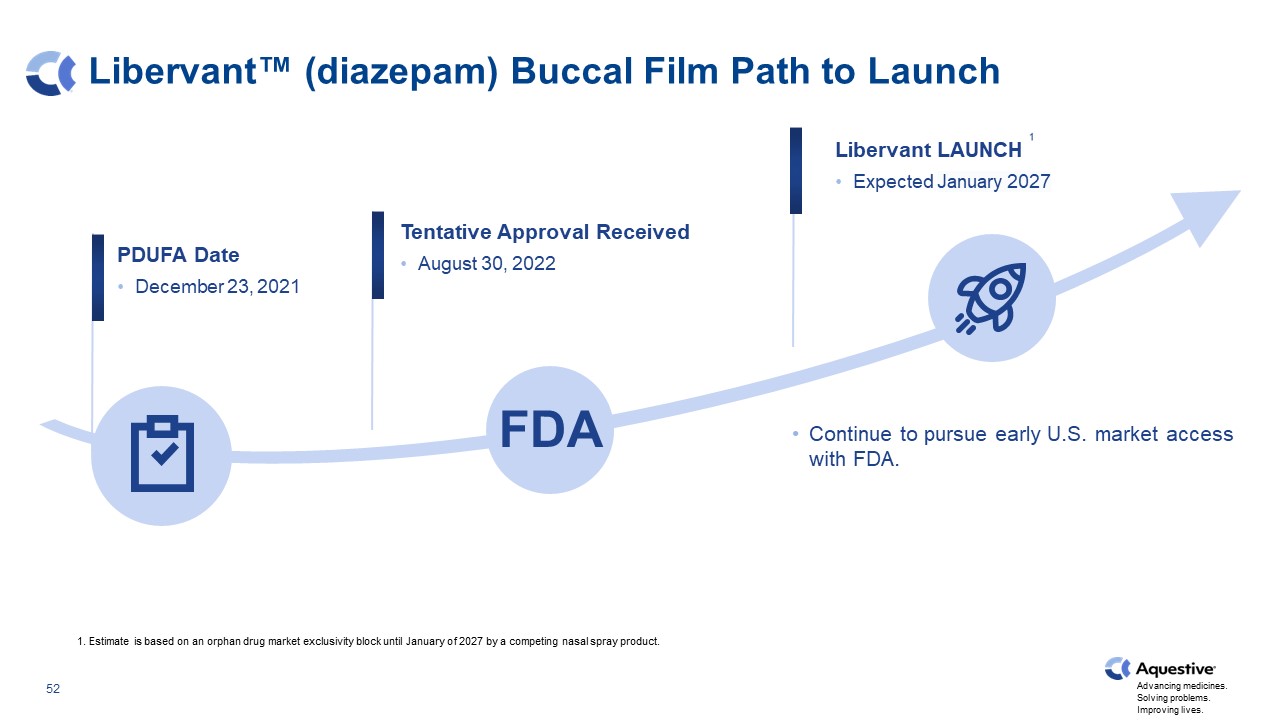
Libervant™ (diazepam) Buccal Film Path to Launch Libervant LAUNCH Expected January 2027 PDUFA Date December 23, 2021 Tentative Approval Received August 30, 2022 Continue to pursue early U.S. market access with FDA. 1. Estimate is based on an orphan drug market exclusivity block until January of 2027 by a competing nasal spray product. FDA

IV. Existing Collaborations 53

Services and Capabilities Formulation Development Systematic approach applied to address permeation barriers Robust formulation design capabilities utilize quality- by-design principles to control risk and optimize performance Analytical Systematic approach utilized to characterize complex formulations and evaluate critical quality attributes Specialized techniques employed to adapt to specialized dosage forms Constant focus on maintaining highly efficient and discriminating methodologies Tech Transfer Multiple scales of analogous equipment Broad experience in multiple thin-film manufacturing techniques Process analytical technology (PAT) to continually drive innovation Regulatory Experienced with the health authorities' approval process Leadership provided during engagements with health authorities throughout the development and approval process 54

Product Licenses Across the Globe Indivior Assertio Hypera Haisco Pharmanovia Zambon Mitsubishi Tanabe Pharma America We currently have eight active worldwide licensing and manufacturing contracts; five more than just two years ago.

Existing Product Portfolio Has Generated Over $500M In Revenue Exservan (2021 Launch) Emylif (2023 Launch) Pharmanovia (ex-US, China) TBD(US) Expected 2024 Launch Expected 2027 Launch1 Riluzole FDA Approval 2019 Diazepam FDA Approval 2022 Haisco (China) Zambon (EU) TBD (China) Clobazam FDA Approval 2018 Ondansetron FDA Approval 2010 Buprenorphine/Naloxone FDA Approval 2010 Mitsubishi Tanabe (US) Expected 2025 Launch Indivior Suboxone (2010 Launch) Hypera (SAM) TBD (US) Ondif (2022 Launch) Assertio (Global) Sympazan (2018 Launch) Product Development Collaborations Commercial Manufacturing 1. Estimate is based on an orphan drug market exclusivity block until January of 2027 by a competing nasal spray product.

Global Diversification of Suboxone 1. Aquestive Therapeutics data on file. 2. Data from Indivior Jeffries Healthcare Conference Presentation June 7, 2023. Suboxone ROW Market2 N. America Canada Europe & Middle East EU France Italy Germany Denmark, Norway Sweden Finland Switzerland UK Israel Australasia Australia New Zealand Available Approved Indivior has presence in 39 countries Suboxone Film is approved in 36 countries Suboxone ROW business is expected to grow to 47% of the Suboxone Revenue by 20291 reducing the reliance on the Suboxone US market. Suboxone Film is currently distributed in Denmark, Finland, Germany, Italy, Norway, U.K., Sweden, Australia, Canada, Israel, and Malaysia. SUBOXONE Film – Approved in 36 countries ex-U.S. Filings under review in Kuwait, Kingdom of Saudi Arabia and Colombia.
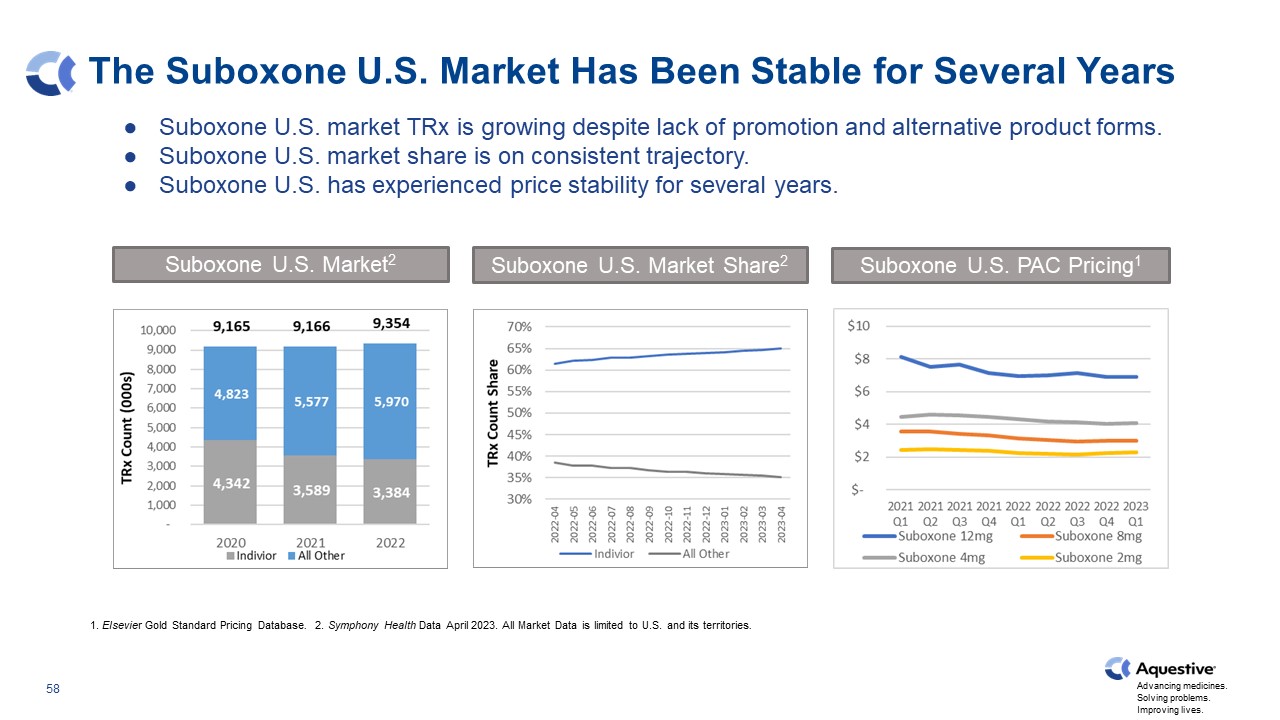
The Suboxone U.S. Market Has Been Stable for Several Years Suboxone U.S. market TRx is growing despite lack of promotion and alternative product forms. Suboxone U.S. market share is on consistent trajectory. Suboxone U.S. has experienced price stability for several years. 1. Elsevier Gold Standard Pricing Database. 2. Symphony Health Data April 2023. All Market Data is limited to U.S. and its territories. Suboxone U.S. Market2 Suboxone U.S. Market Share2 Suboxone U.S. PAC Pricing1

Appendix 59
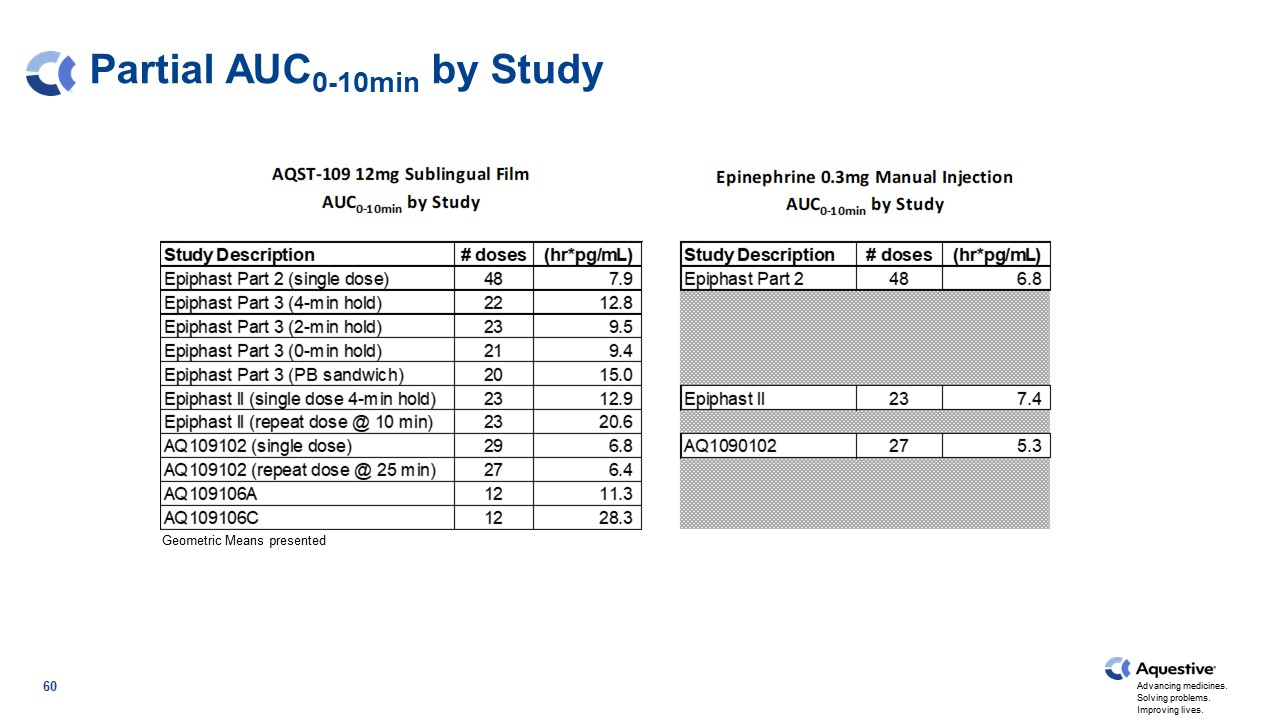
Partial AUC0-10min by Study Geometric Means presented 60
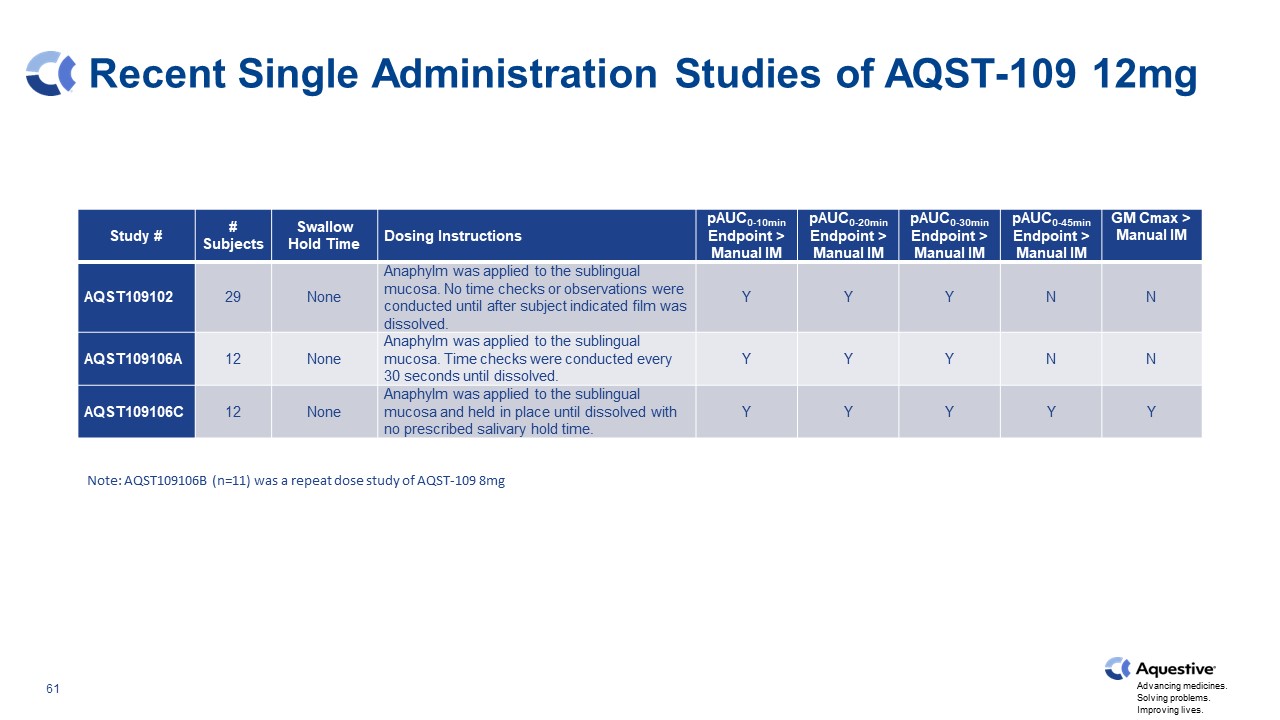
Recent Single Administration Studies of AQST-109 12mg Study # # Subjects Swallow Hold Time Dosing Instructions pAUC0-10min Endpoint > Manual IM pAUC0-20min Endpoint > Manual IM pAUC0-30min Endpoint > Manual IM pAUC0-45min Endpoint > Manual IM GM Cmax > Manual IM AQST109102 29 None Anaphylm was applied to the sublingual mucosa. No time checks or observations were conducted until after subject indicated film was dissolved. Y Y Y N N AQST109106A 12 None Anaphylm was applied to the sublingual mucosa. Time checks were conducted every 30 seconds until dissolved. Y Y Y N N AQST109106C 12 None Anaphylm was applied to the sublingual mucosa and held in place until dissolved with no prescribed salivary hold time. Y Y Y Y Y Note: AQST109106B (n=11) was a repeat dose study of AQST-109 8mg
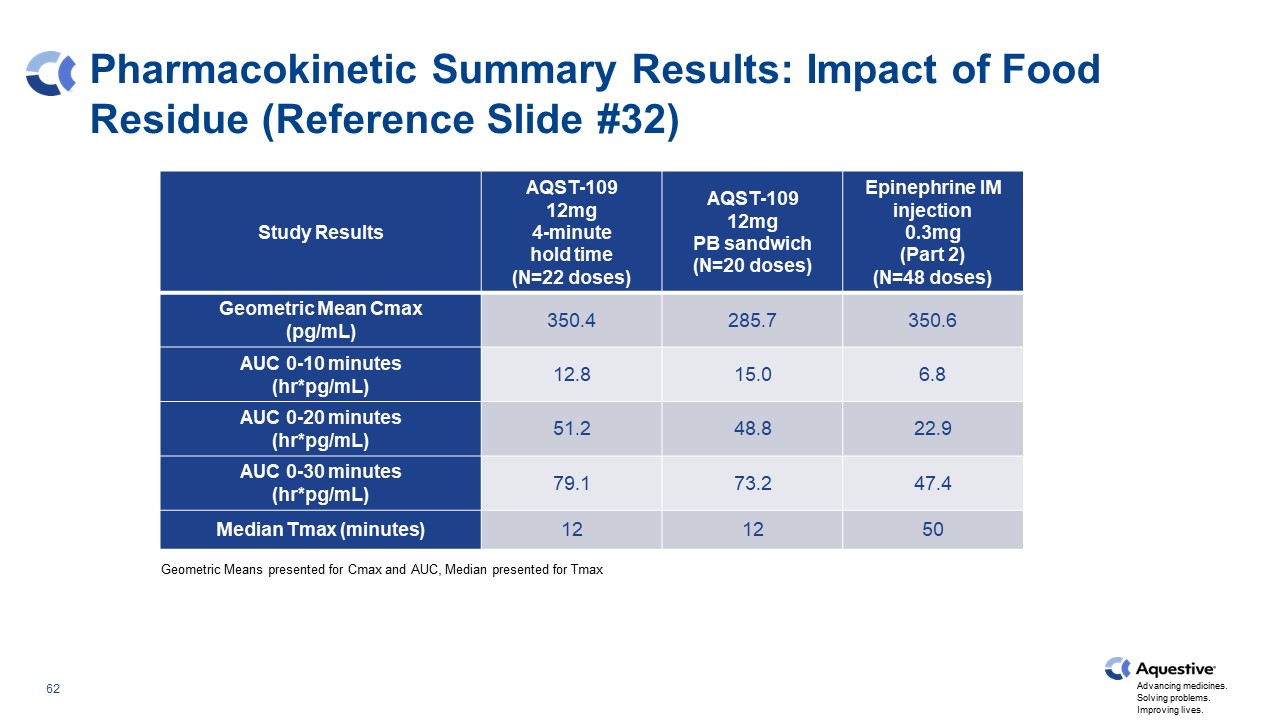
Pharmacokinetic Summary Results: Impact of Food Residue (Reference Slide #32) Geometric Means presented for Cmax and AUC, Median presented for Tmax Study Results AQST-109 12mg 4-minute hold time (N=22 doses) AQST-109 12mg PB sandwich (N=20 doses) Epinephrine IM injection 0.3mg (Part 2) (N=48 doses) Geometric Mean Cmax (pg/mL) 350.4 285.7 350.6 AUC 0-10 minutes (hr*pg/mL) 12.8 15.0 6.8 AUC 0-20 minutes (hr*pg/mL) 51.2 48.8 22.9 AUC 0-30 minutes (hr*pg/mL) 79.1 73.2 47.4 Median Tmax (minutes) 12 12 50
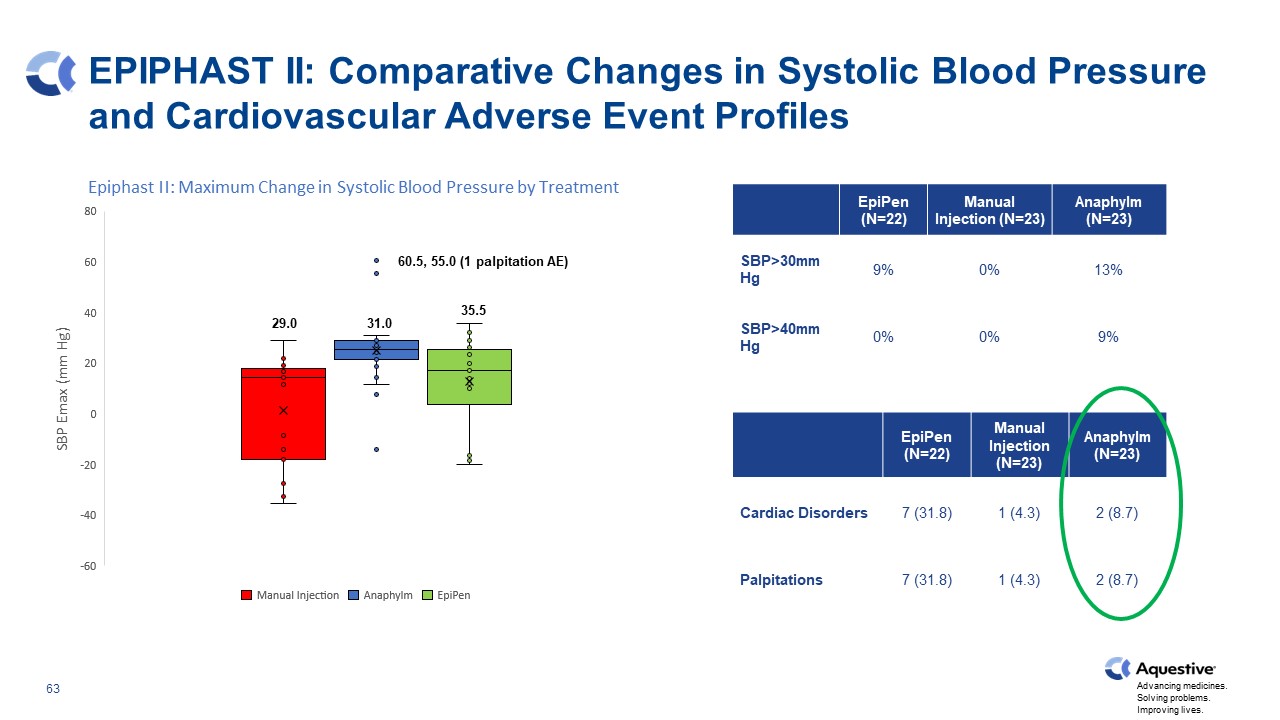
EPIPHAST II: Comparative Changes in Systolic Blood Pressure and Cardiovascular Adverse Event Profiles EpiPen (N=22) Manual Injection (N=23) Anaphylm (N=23) SBP>30mm Hg 9% 0% 13% SBP>40mm Hg 0% 0% 9% EpiPen (N=22) Manual Injection (N=23) Anaphylm (N=23) Cardiac Disorders 7 (31.8) 1 (4.3) 2 (8.7) Palpitations 7 (31.8) 1 (4.3) 2 (8.7) 29.0 31.0 35.5 60.5, 55.0 (1 palpitation AE)

Study 102 & 106: Comparative Changes in Systolic Blood Pressure and Cardiovascular Adverse Event Profiles EpiPen (102, N=27) EpiPen Generic (102, N=29) Auvi-Q (102, N=29) Manual Injection (102, N=27) Anaphylm (106, N=12) SBP>30mm Hg 7% 7% 7% 7% 42% SBP>40mm Hg 4% 3% 0% 4% 8% EpiPen (102, N=27) EpiPen Generic (102, N=29) Auvi-Q (102, N=29) Manual Injection (102, N=27) Anaphylm (106, N=12) Cardiac Disorders 2 (7.4) 2 (6.9) 0 1(3.7) 0 Palpitations 2 (7.4) 2 (6.9) 0 0 0 Atrial Tachycardia 0 0 0 1 (3.7) 0 45.5 45.0 43.5 40.5 38.5

Thank You 71