Exhibit 99.1

REVISED ANNUAL INFORMATION FORM
Fiscal Year Ended February 29, 2012
September 11, 2012
TABLE OF CONTENTS
Explanatory Notes | 3 | |||
Cautionary Note Regarding Forward-Looking Statements | 3 | |||
Corporate Structure | 4 | |||
General Development of the Business | 6 | |||
Business of the Company | 8 | |||
Risk Factors | 25 | |||
Dividends | 37 | |||
Description Of The Share Capital | 37 | |||
Market for Securities | 39 | |||
Directors and Officers | 39 | |||
Cease Trade Orders, Bankruptcies, Penalties or Sanctions | 42 | |||
Legal Proceedings and Regulatory Actions | 43 | |||
Interest of Management and Others in Material Transactions | 43 | |||
Transfer Agents and Registrars | 43 | |||
Material Contracts | 44 | |||
Interest of Experts | 44 | |||
Report on Audit Committee | 44 | |||
Additional Information | 45 | |||
Schedule “A” Charter of the Audit Committee of the Board of Directors | A-1 |
EXPLANATORY NOTES
As used in this annual information form, unless the context otherwise requires, references to “Neptune”, the “Company”, “we”, “us”, “our” or similar terms refer to Neptune Technologies & Bioressources Inc. and its subsidiaries, references to “Acasti” refer to Acasti Pharma Inc. and references to “NeuroBio” refer to NeuroBioPharm Inc.
Unless otherwise noted, in this annual information form, all information is presented as of February 29, 2012. All references in this annual information form to “dollars”, “CDN$” and “$” refer to Canadian dollars, and references to “US$” refer to United States dollars, unless otherwise expressly stated.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Information Form contains certain information that may constitute forward-looking information within the meaning of Canadian securities laws and forward-looking statements within the meaning of U.S. federal securities laws, both of which we refer to as forward-looking information. Forward-looking information can be identified by the use of terms such as “may”, “will”, “should”, “expect”, “plan”, “anticipate”, “believe”, “intend”, “estimate”, “predict”, “potential”, “continue” or other similar expressions concerning matters that are not statements about the present or historical facts. Forward-looking statements in this Annual Information Form include, but are not limited to, statements about:
| • | Neptune’s ability, and the ability of its distribution partners, to continue to successfully commercialize Neptune Krill Oil (NKO®) and ECOKRILL Oil (EKO™), and the ability of Neptune’s subsidiaries, Acasti and NeuroBio, to commercialize other product candidates in the United States, Canada and internationally; |
| • | plans of Neptune’s subsidiaries, Acasti and NeuroBio, to conduct new clinical trials for product candidates, including the timing and results of these clinical trials; |
| • | the timing and cost of completion of the expansion project of Neptune’s manufacturing facility in Sherbrooke, Quebec, and the amount of increased production capacity for krill oil at the expanded facility; |
| • | Neptune’s estimates of the size of the potential markets for NKO® and EKO™ and its product candidates and the rate and degree of market acceptance of EKO™ and NKO® and its product candidates; |
| • | the benefits of NKO® and EKO™ and its product candidates as compared to others products in the nutraceutical and pharmaceutical markets; |
| • | Neptune’s ability to maintain and defend its intellectual property rights in NKO® and EKO™ and in its product candidates; and |
| • | Neptune’s expectations regarding its financial performance, including its revenues, expenses, gross margins, liquidity, capital resources and capital expenditures. |
Although the forward-looking information is based upon what we believe are reasonable assumptions, no person should place undue reliance on such information since actual results may vary materially from the forward-looking information.
In addition, the forward-looking information is subject to a number of known and unknown risks, uncertainties and other factors, including those described in this Annual Information Form under the heading “Risk Factors”, many of which are beyond our control, that could cause actual results and developments to differ materially from those that are disclosed in or implied by the forward-looking information, including, without limitation:
| • | the Company’s history of net losses and inability to achieve profitability; |
| • | the successful commercialization of NKO® and EKO™; |
3
| • | the Company’s reliance on third parties for the manufacture, supply and distribution of its products and for the supply of raw materials; |
| • | the Company’s reliance on a limited number of distributors; |
| • | the Company’s ability to manage its growth efficiently; |
| • | the Company’s ability to further penetrate core or new markets; |
| • | the Company’s dependence on a single manufacturing facility; |
| • | the Company’s ability to attract and retain skilled labor; |
| • | the Company’s ability to attract, hire and retain key management and personnel; |
| • | the success of current and future clinical trials by the Company and its subsidiaries; |
| • | the Company’s ability to achieve its publicly announced milestones on time; |
| • | product liability lawsuits brought against the Company and its subsidiaries; |
| • | intense competition from other companies in the pharmaceutical and nutraceutical industry; and |
| • | the Company’s ability to secure and defend its intellectual property rights. |
Consequently, all the forward-looking information is qualified by this cautionary statement and there can be no guarantee that the results or developments that we anticipate will be realized or, even if substantially realized, that they will have the expected consequences or effects on our business, financial condition or results of operations. Accordingly, you should not place undue reliance on the forward-looking information. Except as required by applicable law, Neptune does not undertake to update or amend any forward-looking information, whether as a result of new information, future events or otherwise. These forward-looking statements are made as of the date of this Annual Information Form.
CORPORATE STRUCTURE
Company Overview
Neptune was incorporated on October 9, 1998 pursuant to a certificate of incorporation issued under Part 1A of theCompanies Act(Québec). On February 14, 2011, theBusiness Corporations Act(Québec) came into effect and replaced theCompanies Act(Québec). Neptune is now governed by theBusiness Corporations Act(Québec). On May 30, 2000, the articles of the Company were amended in order to proceed with the restructuring of the Company’s capital stock and to convert its then issued and outstanding shares into newly-created classes of shares. The Company’s articles were also amended on May 31, 2000 to create Series A Preferred Shares. On August 29, 2000, the Company converted all its issued and outstanding Class A shares into Class B subordinate shares. On September 25, 2000, the Company further amended its share capital to eliminate its Class A shares and converted its Class B subordinate shares into common shares. On May 11, 2001, the Company amended its articles of incorporation to repeal the restrictions with respect to closed companies.
Neptune’s head office and registered office is located at 225, Promenade du Centropolis, Suite 200, Laval, Québec, Canada, H7T 0B3. The Company’s website address iswww.neptunebiotech.com. The Company is also the owner of the websiteswww.mynko.com andwww.neptunekrilloil.com.
Intercorporate Relationships
Neptune has two wholly-owned subsidiaries, Neptune Technologies & Bioressources USA Inc., or Neptune USA, and Neptune Technologies & Bioressources Hong Kong Limited, or Neptune Hong Kong, and two majority-owned subsidiaries, Acasti and NeuroBio. As of the date of this AIF, Neptune owns 57% of the voting rights attached to the securities of Acasti and 99% of the voting rights attached to the securities of NeuroBio. See “Corporate Structure — Corporate Structure Diagram”.
4
Acasti was incorporated on February 1, 2002 pursuant to a certificate of incorporation issued under Part 1A of theCompanies Act(Québec) under the name 9113-0310 Québec Inc. and, prior to its partial spin-off in 2008, was a wholly-owned subsidiary of Neptune. The common shares of Acasti are listed and posted for trading on the TSX Venture Exchange under the symbol “APO”. Acasti is a company involved in the pharmaceutical industry.
NeuroBio was incorporated on October 15, 2008 pursuant to a certificate of incorporation issued under Part 1A of theCompanies Act(Québec) under the name Neurovimer Pharma Inc. NeuroBio is also a company involved in the pharmaceutical industry.
Neptune USA was incorporated on June 1, 2006 under the laws of the State of Delaware and Neptune Hong Kong was incorporated on May 3, 2012 under the laws of Hong Kong. Neptune USA and Neptune Hong Kong do not carry on an active business at this time.
Corporate Structure Diagram
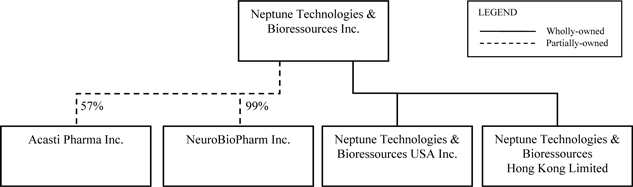
As of the date of this AIF, Neptune owns 41,381,333 Class A shares (common shares) of Acasti, representing approximately 57% of Class A shares (common shares) issued and outstanding of Acasti and 57% of the voting rights attached to the securities of Acasti. Acasti Class A shares (common shares) are voting, participating and with no par value.
As of the date of this AIF, Neptune holds 99% of the voting rights attached to the securities of NeuroBio through the holding of 8,500,990 Class A subordinate voting shares of NeuroBio, representing 99.99% of Class A subordinate voting shares issued and outstanding, 2,475,000 Class B multiple voting shares of NeuroBio, representing 99% of Class B multiple voting shares issued and outstanding, 17,325,000 Class G non-voting shares of NeuroBio, representing 99% of Class G non-voting shares issued and outstanding, and 25,740,000 Class H subordinate voting shares of NeuroBio, representing 99% of Class H subordinate voting shares issued and outstanding. As of the date of this Annual Information Form, Neptune also holds warrants of NeuroBio, namely 5,940,000 Series 2011-1 warrants, 1,885,574 Series 2011-2 warrants and 46,246 Series 2011-3 warrants to purchase 7,871,820 Class A subordinate voting shares of NeuroBio.
On September 5, 2012, a prospectus qualifying the distribution of 2,000,000 Class A subordinate voting shares and 4,000,000 Series 2011-1 warrants of NeuroBio held by Neptune by way of a dividend-in-kind was filed with Canadian securities regulatory authorities. Following the payment of the dividend, Neptune will control approximately 96% of the voting rights attached to the securities of NeuroBio in the aggregate and its holding of Class A subordinate voting shares of NeuroBio will be reduced to 6,500,990 Class A subordinate voting shares, representing approximately 76% of the Class A subordinate voting shares issued and outstanding. Neptune’s holding of Series 2011-1 warrants will also be reduced to 1,940,000 Series 2011-1 warrants following the distribution of the dividend, representing approximately 32.33% of the Series 2011-1 warrants issued and outstanding.
5
Reorganization of the Share Capital of NeuroBio
On April 12, 2011, NeuroBio proceeded with the following transactions affecting its capital structure: (i) NeuroBio consolidated all classes of its capital stock on a 2:1 basis; (ii) NeuroBio exchanged the resulting 50 Class A shares for 1,000 new Class A subordinate voting shares, 26,000,000 Class H subordinate voting shares redeemable for $0.45 per share and 6,000,000 Series 2011-1 warrants; (iii) NeuroBio exchanged the resulting 17,500,000 Class C non-voting shares, 3,500,000 Series 4 warrants and 1,500,000 Series 5 warrants for 17,500,000 Class G non-voting shares redeemable for $0.20 per share, 3,450,075 Series 2011-2 warrants and 8,050,175 Series 2011-3 warrants; and (iv) NeuroBio converted its accounts payable to Neptune in the amount of approximately $850,000 into 8,500,000 Class A subordinate voting shares.
The purpose of the transaction was to establish and freeze the estimated fair value of NeuroBio for its shareholder. Following the transaction, the valuation of the Class A subordinate voting shares was determined by the last transaction of NeuroBio; which is the conversion of its account payable to Neptune into 8,500,000 Class A subordinate voting shares, at $0.10 per share.
GENERAL DEVELOPMENT OF THE BUSINESS
Three Year History
Fiscal Year Ended February 28, 2010
During the fiscal year ended February 28, 2010, in response to increase in demand, Neptune completed an initial expansion of its Sherbrooke plant, following which the plant’s maximum yearly production of krill oil was increased from 60,000 kilograms to 100,000 kilograms. See “Business of the Company - Manufacturing and Facilities”.
During the first quarter, Neptune signed an agreement with Bayer Healthcare LLC for the commercialization of Neptune proprietary products in the United States. Neptune also entered into a distribution agreement with Inno-Vite pursuant to which Inno-Vite launched NKO® under the brand name Inno-KrillTM in health food stores across Canada. During the second quarter, Weifa launched NKO® for the first time in the Norwegian market. Neptune also presented its products at Vitafoods International 2009.
During the second quarter, Neptune received a complaint filed by Schiff Nutrition Group Inc., a former distributor of Neptune’s products alleging that Neptune failed to meet certain delivery thresholds. The parties reached a settlement and the case has been dismissed. See “Business of the Company - Economic Dependence/Litigation”.
In 2009, certain of Neptune’s competitors started marketing, advertising and selling their finished krill-based products claiming benefits based on Neptune’s research. Neptune reacted by filing suits against some of those companies in order to protect its intellectual property. On November 13, 2009 Neptune filed a patent infringement lawsuit against Aker BioMarine ASA, Jedwards International, Inc and Virgin Antartic LLC. See “Business of the Company — Economic Dependence/Litigation”.
On or around January 27, 2010, Neptune and Acasti filed a Motion for the Issuance of a Permanent Injunction relating to a patent infringement before the Quebec Superior Court against US Nutraceuticals LLC (d.b.a. Valensa). See “Business of the Company —Economic Dependence/Litigation”.
Fiscal Year Ended February 28, 2011
During the fiscal year ended February 28, 2011, Neptune managed to increase its initial plant expansion from a maximum of 100,000 kilograms of krill oil per year to a maximum of 130,000 kilograms per year. See “Business of the Company - Manufacturing and Facilities”. During the first quarter, prior to its graduation to the TSX, Neptune was named as one of the TSX Venture 50, a ranking of strong performers on the TSX Venture Exchange.
Also during the fiscal year ended February 28, 2011, Neptune launched EKO™ at Health Ingredient Europe 2010 in Madrid. See “NKO® and EKO™ — Our Lead Products”. During the second quarter, Neptune appointed two investor relations firms, The Howard Group and CEOcast, in order to increase Neptune’s visibility toward the investment community in Canada and the United States, respectively.
6
On March 9, 2010, Neptune filed an appeal with the European Patent Office’s Board of Appeal contesting a 2009 decision of the European Patent Office regarding the European composition of phospholipids and use patent #1417211. See “Business of the Company - Economic Dependence/Litigation”.
During the third quarter, Neptune completed a non-brokered private placement of $2,647,000 through the offering of common shares at a price of $1.85. Two institutional investors participated in the financing. Also during the third quarter, Health Canada approved therapeutic and risk reduction claims for NKO®, among the strongest of which was the claim that products providing 1-3g EPA and DHA per day (amounting to 3-10g of fish oil per day, or 6-20 softgels) help to reduce serum triglycerides, compared to four NKO® 500mg softgels previously approved for the same indication. Health Canada approved a claim for NKO® for cholesterol with a decrease of LDL (“bad cholesterol”) and increase of HDL (“good cholesterol”) using only two softgels per day as well as an anti-inflammatory claim using only one softgel per day and a specific claim for premenstrual syndrome.
Fiscal Year Ended February 29, 2012
During the fiscal year ended February 29, 2012, Neptune continued its investor relations efforts to increase Neptune’s visibility toward the investment community in Canada and the United States, with the objective of reaching higher trading volumes. Neptune presented at the 23rd annual Roth OC Growth Stock Conference in California. Over 400 companies selected by Roth Capital Partners were presenting at the conference and over 1,000 buy-side investors attended the conference. On the research and development front, Neptune presented at the 2011 Scientific Sessions of the American Heart Association its clinical results on the absorption of NKO® compared to competitive products. Neptune sustained its research initiatives by investing in product development, preclinical and clinical studies to validate the health benefits of its products.
On May 3, 2011, Neptune completed a non-brokered private placement of $12,438,000 through the offering of common shares at a price of $2.15 (US$2.25) plus 25% warrant coverage at $2.65 (US$2.75). In total, Neptune issued 5,787,057 common shares and 1,446,265 warrants. Following the end of the first quarter, officers and directors of Neptune exercised 550,000 options a strike price of $2.60, representing an amount of $1,430,000 in aggregate cash proceeds.
Also in May 2011, Neptune announced that it and its marine derived products successfully completed an extensive review of key environmental claims by NSF International. See “Business of the Company — Supply of Krill”.
In the second quarter, Neptune appointed Raj Nakra Associates as an agent for the Indian market. Neptune also finalized agreements with two major U.S. distributors to sell NKO® through their well-established network of U.S. national retailers and wholesalers.
In July 2011, Neptune appointed to its board of directors Dr. Anthony Holler, the former CEO of ID Biomedical, a company dedicated to the commercial development of medical products and technologies for the diagnosis, treatment and prevention of human infectious diseases. In 2011, Neptune also welcomed Mr. Michel Chartrand, a member of Neptune’s board of directors since 2005, as Chief Operating Officer.
On November 28, 2011, Neptune’s common shares started trading on the TSX following Neptune’s migration from the TSX Venture Exchange. In December 2011, Neptune announced the first phase of the currently underway expansion project of its Sherbrooke plant. See “Business of the Company — Manufacturing and Facilities”.
In September 2011, Neptune announced the conclusion of a memorandum of understanding, or MOU, with Shanghain KaiChuang Deep Sea Fisheries Co., Ltd., or SKFC, to form a 50/50 joint venture named Neptune-SKFC Biotechnology, which would manufacture and commercialize Neptune’s krill products in Asia. The initial cost and total value of the project, which includes the construction of a production facility and development of a commercial distribution network for Asia, as well as other details of this arrangement are currently being reviewed by the parties. SFKC is 43% owned by Shanghai Fisheries General Corporation, or SFGC, a large fishing conglomerate owned by the Government of China. SFGC is specializing in pelagic fishing, fishing vessels, fishing machinery, fresh grocery and storage services. It is present in more than 10 countries and employs more than 4,000 employees. SKFC also has the largest fleet of vessels of krill harvesting in the Antartic Ocean. The MOU is subject to further negociations and to approval by the boards of each party as well as by Chinese regulators.
7
On October 4, 2011, the Company filed Complaints against Aker Biomarine ASA, Aker Biomarine Antarctic USA Inc. and Schiff Nutrition International Inc. (Aker et al.) and against Enzymotec Limited, Enzymotec USA Inc., Mercola.com Health Resources, LLC and Azantis Inc. Both Complaints were for the infringement of the Company’s US patent 8,030,348 and for damages. See “Business of the Company — Economic Dependence/Litigation”.
On December 21, 2011, the Company received a motion filed by the University of Sherbrooke, asking the Court to order the transfer of certain intellectual property to Neptune. See “Business of the Company — Economic Dependence/Litigation”.
In February 2012, Neptune announced that Jamieson Laboratories was initiating commercialization of NKO® in the Canadian food, drug and mass market retail channel coast to coast. Jamieson, Canada’s largest manufacturer and distributor of dietary supplements, celebrates its 90th history in 2012 and offers more than 250 different products in over 7,000 stores in Canada.
Recent Developments
To date during the 2013 fiscal year, which ends on February 28, 2013, Neptune has continued the first phase of the expansion project of its Sherbrooke plant, which is anticipated to be completed by the end of our current fiscal year. See “Business of the Company — Manufacturing and Facilities”. On September 7, 2012, Neptune announced that pursuant to a final prospectus dated September 5, 2012, 2,000,000 Class A subordinate voting shares and 4,000,000 Series 2011-1 warrants of NeuroBio held by Neptune will be distributed on October 31, 2012 to holders of record of Neptune’s shares at the close of business on October 15, 2012 by way of a dividend-in-kind. See “Corporate Structure — Corporate Structure Diagram”.
BUSINESS OF THE COMPANY
Overview
Neptune is a biotechnology company engaged primarily in the development, manufacture and commercialization of marine-derived omega-3 polyunsaturated fatty acids, or PUFAs. Neptune produces omega-3 PUFAs through its patented process of extracting oils from Antarctic krill, which omega-3 PUFAs are then principally sold as bulk oil to Neptune’s distributors who commercialize them under their private label primarily in the U.S., European and Australian nutraceutical markets. Neptune’s lead products, Neptune Krill Oil (NKO®) and ECOKRILL Oil (EKO™), generally come in capsule form and serve as a dietary supplement to consumers.
Having commenced commercial krill oil production in 2002, Neptune pioneered the commercialization of omega-3 PUFAs extracted from krill for human health maintenance and it now continues to further progress its product development based on its proprietary technology. We believe that our ability to provide a safe and effective product is a key factor in building and sustaining our credibility with our distribution partners. In fiscal year 2012, we produced 130,000 kilograms of krill oil, which at the time was our maximum capacity of production at our manufacturing facility. We are in the process of completing an expansion of our facility that, when completed, is expected to enable us to produce approximately 300,000 kilograms of krill oil annually. We believe this increase in production capacity will help position us to meet growing market demand for Neptune’s krill oil products. See “Business of the Company — Manufacturing and Facilities” and “Risk Factors — Risks Related to the Company’s Business — The Company is dependent on a single manufacturing facility.”
Through Neptune’s subsidiaries, Acasti and NeuroBio, in which Neptune respectively holds 57% and 99% of the voting rights, Neptune is also pursuing opportunities in the pharmaceutical market, namely in the over-the-counter, or OTC, medical food and prescription drug market. Neptune has granted licensing rights to both Acasti and NeuroBio which allow them to leverage the intellectual property, clinical data and know-how developed by Neptune to focus on, respectively, the research and development of safe and therapeutically effective compounds for highly prevalent atherosclerotic conditions, such as cardiometabolic disorders and cardiovascular diseases, and for neurodegenerative and inflammation related conditions.
8
The Krill Industry
Krill, which resembles shrimp, is a generic term designating approximately 85 species of deep and cold water pelagic marine planktonic animals (zooplankton) constituting of the global marine biomass. According to the Australian government’s Department of Sustainability, Environment, Water, Population and Communities (Australian Antarctic Division), krill is the most abundant animal biomass on the planet and is found in schools that can sometimes cover several square kilometres of ocean.
Because krill feeds on phytoplankton, namely diatoms and dinoflagellates, its lipid content is a major source of PUFAs, mainly docosahexaenoic acid, or DHA, and eicosapentaenoic acid, or EPA, two types of marine omega-3 fatty acids beneficial for health maintenance. Krill contains proteins offering a range of amino acids and effective digestive enzymes. In addition, it contains powerful antioxidants, including astaxanthin. Krill also contains phospholipids, amino acids and minerals providing clinically proven benefits in the absorption and digestion of nutrients for humans and animals.
Neptune’s patented krill oil extraction process produces a compound substance that contains enhanced levels of EPA and DHA, phospholipids and antioxidants, making it highly bioavailable (capable of absorption) and resistant to oxidation. Based on our internal research, we believe Neptune’s krill oil has a lower level of oxidation than fish oil due to its high natural content of antioxidants, which also results in a longer shelf life of our commercialized products.
Despite the higher price per kilogram of krill oil compared to fish oil, the krill oil market had global revenues of US$51.1 million in 2011, and is projected to grow at a compound annual growth rate, or CAGR, of 16.4% between 2011 and 2016, according to a Frost & Sullivan industry report entitled the2012 Global Overview of the EPA and DHA Omega 3 Ingredients Markets, or the Frost & Sullivan July 2012 Report.
NKO® and EKO™ – Our Lead Products
Neptune Krill Oil (NKO®) and ECOKRILL Oil (EKO™)
NKO®, which was first commercialized in 2003, is a marine oil extracted from Antarctic krill (Euphasia superba) that contains the two essential omega-3 PUFAs, EPA and DHA, and provides a blend of nutritional elements. Its elevated content in phospholipids rich in omega-3 and omega-9 fatty acids and antioxidants such as astaxanthin and vitamin A and vitamin E offer a safe and effective product free of preservatives with clinically proven health benefits.
NKO® has a biomolecular profile of phospholipids, omega-3 fatty acids and important antioxidants that surpasses the usual profile of fish oils. The combination of phospholipids and omega-3 fatty acids highly facilitates the passage of fatty acids molecules through the body’s intestinal wall, increasing the bioavailability of omega-3 fatty acids. Independent research has shown that astaxanthin has a stronger antioxidant activity than vitamin A and vitamin E and other antioxidants such as lycopene and lutein. Neptune believes that NKO® contains higher amounts of astaxanthin compared to all other krill oil products on the market.
EKO™, which was commercialized in 2010, is similar to NKO® in that it undergoes the same krill oil extraction process except it has slightly lower specifications of PUFAs, phopholipids and antioxidants and, as a result, EKO™ has a lower price point than NKO®. For the 2012 fiscal year, sales of NKO® and EKO™ together accounted for nearly all of Neptune’s consolidated revenues.
Neptune believes that NKO® is the first and only krill oil product with clinically proven human health benefits in cardiovascular, joint, cognitive and women’s health. In 2004, the Alternative Medicine Review published the results of a 12-week, double-blind, randomized trial which demonstrated that daily doses of 1-3g NKO® are significantly more effective than 3g EPA/DHA fish oil in the management of cholesterol levels (hyperlipidemia). Daily doses of 1-3g NKO® have been proven effective in that trial to decrease low density lipoprotein (“LDL” – “bad cholesterol”) by 33.9%, triglycerides by 11.5% and increase high density lipoprotein (“HDL” – “good cholesterol”) by 43.3%.
9
The results of a double blind clinical study performed in May 2003 by Fotini Sampalis M.D., Ph.D., et. al., which were published in the Alternative Medicine Review, support the proposition that NKO® can reduce certain physical and emotional symptoms of premenstrual syndrome, such as stress, irritability and abdominal pain, and that NKO® is more effective than omega-3 fish oils for the management of such premenstrual symptoms.
An analysis of the Framingham Risk Score (which is used to estimate the 10-year cardiovascular risk of an individual and based on data obtained from the Framingham Heart Study, a long-term, ongoing cardiovascular study on residents of the town of Framingham, Massachusetts) data completed by Dr. Roxandra Bunea in 2003 suggests that individuals taking NKO® can significantly reduce cardiovascular disease risk, with an attributable-risk-reduction between 52% and 53%.
A double-blind clinical study performed in 2007 found that NKO® at a daily dose of 300 mg may within a short time to reaction (7-14 days) significantly inhibit inflammation by reducing C-reactive protein as well as significantly alleviate symptoms caused by osteoarthritis and rheumatoid arthritis.
A double-blind clinical trial undertaken by BioTeSys GmbH in February 2009 supports the benefits of NKO® versus a range of other omega-3 products for improving the EPA to arachidonic acid ratio and the omega-3 index. The main objective of the trial was to show the bioavailability of a physiological dosage of omega-3 fatty acids. Within the clinical trial, different sources of EPA and DHA, including different chemical bounds of EPA and DHA, were compared to each other. The obtained data reflects that uptake of EPA and DHA out of NKO® was most prominent and showed significant higher bioavailability in comparison to fish oil and a blend of lecithin, astaxanthin and fish oil. The study stated that, overall, the NKO® product showed clear superiority followed by ethyl esters, fish oil and the blend of lecithin, astaxanthin and fish oil.
Other Nutraceutical Products
Neptune Krill Aquatein™( NKA™)
Neptune Krill Aquatein (krill protein concentrate), or NKA™, is a product that features a range of marine amino acids, including the eight essential amino acids. NKA™ contains pre-digested proteins that are an important source of short-chain amino acids in the form of peptides that facilitate digestion by more effective assimilation.
More complete analyses of the composition of NKA™ were performed and different methods for improving quality and efficiency of production have been investigated. NKA™ is being positioned to be sold for both human and animal nutrition. For the fiscal year ended 2012, NKA™ did not account for any revenues and Neptune believes NKA™ will not generate meaningful revenues during the current fiscal year.
Pharmaceutical Products and Product Candidates - Acasti
Our majority owned subsidiary, Acasti, focuses on the research and development of safe and therapeutically effective compounds for highly prevalent atherosclerotic conditions, such as cardiometabolic disorders and cardiovascular diseases.
ONEMIA™
In 2011, ONEMIA™ became Acasti’s first product to be commercialized. ONEMIA™, marketed as a “medical food”, is only administered under the supervision of a physician and is intended for the dietary management of illnesses associated with omega-3 phospholipid deficiency related to cardiometabolic disorders. The term medical food is defined in the United States Orphan Drug Act as a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.
ONEMIA™ consists of concentrated omega-3 phospholipids and antioxidants, derived from Neptune krill oil. Studies have shown ONEMIA™ to be safe and effective for the dietary management of omega-3 phospholipid deficiency and the consequent abnormal lipid profiles. Omega-3 phospholipid deficiency can lead to a number of conditions, including hyperlipidemia (which generally manifests as high LDL (“bad cholesterol”) and high triglycerides), atherosclerosis (the buildup of plaque on the inside of blood vessels), diabetes and metabolic syndrome.
10
ONEMIA™ is now in the early stages of commercialization and is being distributed in the United States by Acasti to physicians (who then can either provide it to their patients directly or via a website by using a dedicated medical food code). Acasti also makes ONEMIA™ available via distributors and behind-the-counter in pharmacies. Acasti also intends to secure distribution partners to commercialize ONEMIA™ outside of the U.S. See “Risk Factors — Risks Related to the Company’s Business — The Company may not be able to further penetrate core or new markets.”
CaPre®
Acasti’s lead prescription drug candidate is CaPre®, which is a purified high omega-3 phospolipid concentrate derived from Neptune krill oil. CaPre® is being developed to address the prevention and treatment of cardiometabolic disorders, including hypertriglyceridemia, which is characterized by abnormally high levels of triglycerides.
CaPre® is designed to be used as a medical treatment in conjunction with positive lifestyle changes and administered either alone or in conjunction with other treatments such as statins (a class of drug used to reduce cholesterol levels) and, potentially, for use by statin-intolerant patients. In addition to targeting the reduction of moderate and very high triglycerides, preclinical research has indicated that CaPre® may also normalize blood lipids overall by also reducing LDL (“bad cholesterol”) and increasing HDL (“good cholesterol”). See “Business of the Company — Studies & Trials for Pharmaceutical Product Candidates — Acasti’s Product Candidate: CaPre®”. Clinical research is required in order to confirm an analogous efficacy in humans.
CaPre® is currently being evaluated in two Phase II clinical trials: (i) a prospective randomized double blind placebo control clinical study designed to evaluate the safety and efficacy of CaPre® for the management of moderate to high hypertriglyceridemia, and (ii) a prospective randomized open-label clinical trial designed to assess the safety, efficacy and dose response of CaPre® for patients with moderate to high hypertriglycerimedia. Over 600 patients will be enrolled for these trials, some of which were enrolled at the end of 2011. Following the completion of the trials, Acasti intends to prepare an investigational new drug, or IND, submission for a Phase III clinical trial for CaPre® in the U.S. CaPre® is currently undergoing the regulatory approval process in Canada and the United States, which requires, among other things, the safety of the product and its effectiveness in sufficiently reducing triglycerides. See “Business of the Company — Regulatory Environment”.
Based on preclinical evaluations and subject to validation through ongoing clinical trials, we believe that CaPre® could be used to treat high levels of triglycerides (hypertriglyceridemia) and LDL and low HDL. We also believe that the competitive advantages of CaPre® may include a range of clinical benefits at lower dosage levels than other products currently in the market. Generally, lower dosage of medications tends to reduce the risks of certain side effects in patients, including gastrointestinal disorders.
Pharmaceutical Products and Product Candidates - NeuroBio
Our subsidiary, NeuroBio, is in the early stages of developing omega-3 phospholipids medical foods, over-the-counter products and prescription drugs. NeuroBio is dedicated to the research, development and commercialization of active pharmaceutical ingredients for the management of neurodevelopmental, memory, concentration, learning and neurological disorders, from prevention to treatment. NeuroBio addresses mental and neurological conditions, specifically mood disorders such as depression, attention-deficit hyperactivity disorder, or ADHD, and cognitive decline associated with aging.
MPL VI, MPL VII and MPL VIII – Medical Food / OTC
MPL VI is customized for the dietary management of cognitive decline associated with neurodegenerative conditions.
MPL VII is well-positioned to exhibit an intrinsic biological activity, because of its distinctive DHA-bounded phosphatidylcholine content, in memory, concentration and learning disorders, allowing a variety of applications. For this specific product, NeuroBio has an innovative clinical approach to quantify cognitive improvement and reach rapidly the market with conclusive results.
11
MPL VIII was designed and intended to supplement nutrition intake by children and adults suffering from ADHD for which phospholipid deficiency may represent a key risk factor. MPL VIII is an original and a proprietary formulation that contains a specific API having a high concentration in selected phospholipids and with a specific omega-3 profile.
MPL IX – Prescription Drug
MPL IX is under preclinical evaluation for neurological disorders and will be tested in several preclinical models, such as dogs (2 sub-species) and rats (2 sub-species). Various daily doses and durations of treatment will be administered orally to assess the safety and efficacy of given compositions and to determine the pharmacokinetic profile.
Data is expected to demonstrate that MPL IX will dose-dependently and significantly reduce important neurological disorders and improve cognitive functions in these animal models. Most importantly, these effects will need to be achieved without the common side-effect of other traditional treatments.
NeuroBio’s product candidates are at different development and/or validation stages and are expected to require the approval of the U.S. Food and Drugs Administration, or FDA, and/or Health Canada before clinical studies are conducted. Approvals from similar regulatory organizations are also expected to be required before sales are authorized. See “Business of the Company — Studies & Trials for Pharmaceutical Product Candidates — NeuroBio’s Product Candidates” and “Business of the Company — Regulatory Environment”.
Our Market
Neptune’s Market: The Nutraceutical Market
The nutraceutical market encompasses functional foods and dietary supplements, which include a wide range of nutrients such as vitamins, minerals, fatty acids, amino acids and herbal supplements. Neptune focuses on dietary supplements. According to Agriculture and Agri-Food Canada, a government organization that provides statistics on the nutraceutical market, the nutraceutical market is growing rapidly, in part driven by the health demands of an aging population. According to a report published by RNCOS Industry Research Solutions in May 2012 entitledUS Nutraceuticals Market Analysis, the nutraceutical market has become one of the fastest growing industries in the United States. In 2008, the U.S. Census Bureau, using data from the 2000 U.S. Census, projected that by 2030, the number of Americans 65 years old and older will increase from 40.3 million to just over 72.0 million, then representing over 19% of the population in the United States.
The Company believes that health issues such as high (and in some cases low) cholesterol, heart disorders, cognitive function and brain performance disorders and joint issues (including inflammation) are driving the nutraceutical market expansion. We believe the following factors, among others, favor the growth of the nutraceutical market:
| • | improved understanding and scientific knowledge of the contribution of diet in health maintenance and disease prevention; |
| • | increased consumer demand for dietary supplements that help to maintain vitality and promote health; and |
| • | increased health care costs and the trend towards self-treatment with a focus on natural products. |
Omega-3 PUFAs extracted during Neptune’s krill oil extraction process are sold primarily into the nutraceutical market. The most predominant omega-3 fatty acids are DHA and EPA derived from plant and marine sources.
The omega-3 fatty acids contained in Neptune’s products are sourced from krill, a zooplankton, with the advantage that omega-3 fatty acids from krill are carried by phospholipids and not triglycerides such as in fish oil. Phospholipids, a major component of biological membranes, are more easily absorbed by the body than triglycerides, resulting in a higher bioavailability of omega-3 fatty acids contained in krill oil.
12
The FDA announced in 2004 the availability of a qualified health claim for reduced risk of coronary heart disease for conventional foods that contain EPA and DHA omega-3 fatty acids. In 2000, the FDA announced a similar qualified health claim for dietary supplements containing EPA and DHA omega-3 fatty acids and the reduced risk of coronary heart disease.
In addition, extensive research, including Neptune’s clinical trial work, has further demonstrated certain clinical benefits of omega-3. Omega-3 fatty acids reduce inflammation and prevent risk factors associated with chronic diseases, such as heart disease and arthritis, and appear to be particularly important for cognitive (memory and concentration) and behavioural functions. Many forms of arthritis, such as osteoarthritis and rheumatoid arthritis, are inflammatory disorders and lead to pain, stiffness, swelling and functional impairment. Osteoarthritis is the most common form of arthritis and affects approximately 27 million people in the United States, according to a January 2008 publication of the medical journal called Arthritis Rheum. It is caused by the breakdown and eventual loss of the cartilage between the bones of the joints. Non-surgical treatment options for osteoarthritis include analgesic and anti-inflammatory pain medications, nutritional supplementation, physical therapy, exercise and weight loss.
The PUFAs ingredient market and, more specifically, sales of omega-3 ingredients, are experiencing sustained growth, driven by the world retail market for dietary supplements and functional food. Based on the trends reported in the Frost & Sullivan July 2012 Report, the worldwide omega-3 market is expected to exceed US$3.1 billion in annual ingredient sales by 2016 and general market data indicates that sales of higher quality and higher performance omega-3’s are generating increasing revenues.
According to the Frost & Sullivan July 2012 Report, the global market revenue for marine and algae EPA/DHA omega-3 ingredients was US$1.8 billion in 2011, and projected to grow at a combined annual growth rate, or CAGR, of 11.8% from 2012 to 2016. Global consumption was measured at 103,284 metric tons in 2011, and projected to grow at a CAGR of 9.4% from 2012 to 2016.
The world retail market for dietary supplements is highly fragmented, and is comprised of a large number of products and many small manufacturers. According to the Frost & Sullivan July 2012 Report, dietary supplements continued to be the largest consumer of marine omega-3 oils in the global market in 2011 with a 46.2% share, with a total of US$834.6 million in revenue. The Frost & Sullivan July 2012 Report also estimates that pharmaceuticals, infant formulas and foods and beverages were the next largest consumers of marine oil omega-3, with 19.8%, 14.3% and 13.4% shares, respectively, in 2011.
Neptune has conducted clinical trials for functional food applications of NKO® with the multinational corporations Nestlé and Yoplait. However, the parties have decided not to pursue the development of these functional food applications. Neptune is instead currently focusing on the dietary supplement market, particularly in light of growing sales of its NKO® and EKO™ products, Neptune’s determination that supplement sales provide it with higher margins than potential margins in the functional food market and the limits on Neptune’s current maximum production capacity.
In 2008, Neptune received a first payment of €500,000 out of several payments scheduled under the terms of a partnership agreement in connection with its functional food trials. An amount of up to 62.5% of such initial payment may be reimbursable by Neptune in certain circumstances where the clinical trials are not proceeding further.
Acasti’s and NeuroBio’s Market: The Pharmaceutical Market
Cardiometabolic Disorder Treatments - Acasti
Cardiometabolic disorders are considered among the leading health problems worldwide arising from the combined impact of obesity and cardiovascular disease. According to the American Heart Association’s Heart Disease and Stroke Statistics — 2012 Update, an estimated 82.6 million American adults (more than one in three) have one or more types of cardiovascular disease, 41.8 million have low HDL (“good cholesterol”) and 149 million are overweight or obese. According to the American Heart
13
Association, these cardiometabolic risks will lead to an estimated 758,000 Americans having a coronary attack in 2012. The American Heart Association also estimates that direct and indirect costs of cardiovascular disease and stroke in the United States totalled US$297.7 billion in 2008, of which US$32.9 billion was spent on prescribed medications, and these direct and indirect costs are projected to triple before 2030.
Cardiovascular diseases include a wide range of conditions and treatment is focused on reducing cardiovascular risk factors to prevent an acute cardiovascular event and on preventing or delaying the onset of chronic cardiovascular disease. Important risk factors for cardiovascular disease are abnormal levels of lipids and/or lipoproteins such as triglycerides and cholesterol. Increased serum levels of LDL (“bad cholesterol”) and low levels of HDL (“good cholesterol”), the former being recognized as the most important risk factor for the development of cardiovascular disease, are known as dyslipidemia.
Dyslipidemia promotes plaque formation on the interior walls of the arteries thereby impeding the passage of blood. This leads to myocardial infarction (heart attack), coronary artery disease, stroke and peripheral vascular and neurodegenerative disease. According to the Centers for Disease Control and Prevention, coronary heart disease mortality in the United States in 2008 was over 400,000. The Centers for Disease Control and Prevention estimated that in 2011, 71 million American adults had total blood cholesterol values considered borderline-high (200 to 240 mg/dL) or high (above 240 mg/dL) making them potentially eligible for a cholesterol lowering agent.
Neurodegenerative and inflammation related conditions - NeuroBio
NeuroBio focuses on mental and neurological conditions, specifically mood disorders such as depression, ADHD and cognitive decline associated with aging. The prevalence of these disorders in North America is summarized in the following table:
Disorder | Market | Prevalence | Source | |||
Memory, learning, and concentration and neurological disorders | OTC / Medical Food | Affecting throughout a lifespan the majority of people during the educational and professional stage and later 19% of adults aged >65 years | Alzheimer’s Association, 2010 Alzheimer’s Disease Facts and Figures,Alzheimer’s & Dementia,Volume 6 | |||
ADHD (attention- deficit hyperactivity disorder) | OTC / Medical food | 9.0% of children 13-18 yrs (lifetime prevalence) | Merikangas KR, He J, Burstein M, Swanson SA, Avenevoli S, Cui L, Benjet C, Georgiades K, Swendsen J.; Lifetime prevalence of mental disorders in U.S. adolescents: Results from the National Comorbidity Study-Adolescent Supplement (NCS-A).J Am Acad Child Adolesc Psychiatry.2010 Oct;49(10):980-989. | |||
Studies & Trials for Pharmaceutical Product Candidates
General
Neptune is continuously investing in medical research and development aimed at demonstrating the benefits of its products on human health. From time to time, Neptune enters into clinical research programs with strategic partners for the completion of clinical trial.
Acasti’s Product Candidate: CaPre®
Initial nonclinical research designed to evaluate the safety and efficacy of CaPre® was completed in 2011. The efficacy of CaPre® on dyslipidemia was evaluated on Zucker Diabetic Fatty, or ZDF, rats, a commonly used diseased rat phenotype,
14
characterized by established type 2 diabetes, glucose intolerance and severely elevated triglycerides and cholesterol. After 4, 8 and 12 weeks of chronic daily treatment with human equivalent daily dosing of 500mg and 2,500mg, CaPre® was shown to significantly increase HDL cholesterol (“good cholesterol”), by 40% at the lower dose and by up to 61% at higher dose after 3 months treatment in ZDF. These results indicate that CaPre® could potentially be effectively used in patients with metabolic syndrome and/or lipid disorders.
In conjunction with initial nonclinical research, preclinical research was completed by Acasti in late 2011 to further evaluate the potentially broader spectrum of therapeutic efficacy of CaPre®. CaPre® was administered for 3 months at a daily human equivalent dose of 500mg and 2,500mg in both ZDF diabetic and normal healthy rats. Both rat phenotypes were subjected to oral glucose tolerance tests, or OGTT. In medical practice, the OGTT is commonly used to test for diabetes and insulin resistance. It involves the oral administration of high amounts of glucose in order determine how quickly it is cleared from the blood. The test may be performed as part of a test panel, such as the comprehensive metabolic test panel. Treatment of ZDF rats with CaPre® was shown to significantly reduce impaired glucose intolerance within 1 month of treatment, with the higher dose being only slightly more effective than the lower dose. After 3 months, the ZDF rats had established a normal tolerance to glucose analogous to the tolerance of healthy rats. Also, the healthy rats continued to tolerate glucose normally, indicating another safety parameter for CaPre®.
Acasti has also worked with a team dedicated to functional testing and development of therapeutic candidates for arresting and reversing atherosclerosis through modulation of HDL, Reverse Cholesterol Transport and Immune Mediators. The first series of experiments, which was conducted in three mouse models reflecting healthy state and moderate to severe dyslipidemia, took place in 2010 to evaluate the active pharmaceutical ingredients, or APIs, of CaPre®. After six weeks of treatment at very low doses ranging from 500mg and 2,500mg of CaPre®, a statistically significant increase of HDL and reduction of LDL was observed, as well as a reduction of up to 60% of triglycerides.
CaPre® is currently being evaluated in two Phase II clinical trials. See “Business of the Company — Pharmaceutical Products and Product Candidates — Acasti — CaPre®”.
NeuroBio’s Product Candidates
Certain preclinical results have indicated the safety and efficacy of NeuroBio’s APIs portfolio in either nutritional intervention or therapeutic management of memory, concentration and learning disorders, ADHD and cognitive decline associated with aging.
The NeuroBio product portfolio includes highly concentrated phospholipids extracted and purified from different marine species, including krill, which functionalize EPA and DHA most often stabilized by potent antioxidant esters. The NeuroBio product portfolio benefits from efforts over the last twelve years at Neptune and the last three (3) years at NeuroBio. NeuroBio’s product portfolio consists of MPL VI, MPL VII, MPL VIII and MPL VIX, each being at different development and/or validation stage before commercialization as indicated in the table below.
Product | Channel | Indication | Stage of | |||
| MPL VI | Medical Food / OTC | Prevention of cognitive decline | Preclinical/clinical | |||
| MPL VII | Medical Food / OTC | Memory, concentration and learning disorders | Preclinical/clinical | |||
| MPL VIII | Medical Food / OTC | ADHD | Preclinical/clinical | |||
| MPL IX | Prescription Drug | Neurological disorders | Preclinical | |||
NeuroBio still requires approvals from Health Canada and/or the FDA before clinical studies are conducted. Regulatory approval specific to each pathway (medical food, OTC and prescription drug) will also be required before sales are authorized. See “Business of the Company — Regulatory Environment” and “Risk Factors — Risks Related to the Company’s Industry — The Company is subject to significant government regulations.”
15
Supply of Krill
Neptune sources the krill used in the manufacturing of its products generally from three suppliers. Neptune considers its relationship with its suppliers to be good and believes it is not dependent upon any of these suppliers since alternative sources of krill supply are readily available.
There are two primary ocean regions where krill is harvested: the Southern Ocean (Antarctic krill) and the North Pacific Ocean (Pacific krill, mainly off the coasts of Japan and Canada). The total quantity of the krill species in these two oceans is conservatively estimated to be at least 500,000,000 metric tonnes. The World Health Organization estimates that approximately 271,000 metric tonnes of both krill species are harvested annually from these two oceans. From 2002 to 2011, between 105,000 to 212,000 metric tonnes originated from the Southern Ocean (Antarctic krillEuphausia superba) and, on average, 60,000 metric tonnes originated from the Northern Pacific Ocean (Pacific krillEuphausia pacifica) each year. The annual Antarctic krill catches represent an estimated 0.05% of the existing resource. Neptune uses Antarctic krill.
According to the Commission for the Conservation of Antarctic Marine Living Resources, or CCAMLR, from 2008 to 2011, annual quotas for Antarctic krill have increased by 33%. Annual allowable quotas of 6.555 million tonnes for 2009 and 2010 were increased to 8.695 million tons for 2010/11. As a result, the Company believes that krill is an abundant and accessible resource with potential for long-term sustainable exploitation with adequate traceability measures. The average market price for whole frozen krill is around US$900 per metric tonne.
Neptune harvests less than 0.0006% of the total estimated krill biomass and less than 0.03% of the precautionary catch limit. Neptune commits 100% of its krill capture for human health benefits. Worldwide, approximately 88% of total catches are used by fisheries for low valued products, such as fishing baits (45%) and krill meal for aquaculture (43%). Approximately 12% of the total krill catch is used for direct human consumption as food (whole or processed).
In May 2011, NSF International, an independent, not-for-profit organization that provides standards development, product certification, auditing, education and risk management for public health and the environment, completed a review of key environmental claims for Neptune and its marine derived products. The audit performed by NSF International was conducted to ensure clarity and conformance with the strict criteria of the International Organization for Standardization (ISO) 14021: Environmental labels and declaration, as well as Federal Trade Commission (16 CFR PART 260): Guides for the Use of Environmental Marketing Claims. Based on the results of the audit, Neptune was approved by NSF International to make the following five claims: (i) Neptune only uses krill captured by fisheries that follow the Antarctic Treaty (1961) rules and respects the annual capture quota of the CCAMLR, (ii) Neptune obtains krill from fisheries that use only mid-water trawl, which reduces the impact on other species as by-catch, (iii) Neptune krill oils are alternative sources of marine omega-3 which reduce the pressure on fish populations, (iv) Neptune’s OceanExtract™ patented process recycles 99% of the extraction solvent used during the manufacture of Neptune Krill oils, and (v) Neptune only uses krill that is 100% traceable to the source of capture.
Manufacturing and Facilities
Neptune produces all of its products at its plant located on Pepin Street in Sherbrooke, Quebec, Canada.
Since 2010, the production capacity of the Sherbrooke plant has steadily increased. During the 2010 fiscal year, in response to increase in demand from its distributors, Neptune completed an initial expansion of the production capacity of the Sherbrooke plant increasing production capacity from 60,000 kilograms to 100,000 kilograms. In the 2011 fiscal year, Neptune increased its production capacity from 100,000 kilograms per year to 130,000 kilograms per year. An additional $21.0 million expansion of the Sherbrooke plant is currently ongoing, which Neptune expects will increase its annual krill oil production capacity to 300,000 kilograms. Neptune will be required to obtain a permit from the Minister of Environment Quebec that will allow it to bring its krill oil capacity under its current permit from 100,000 kilograms per year to 300,000 kilograms per year. See “Risk Factors — Risks Related to the Company’s Business — The Company may be adversely affected by environmental and safety regulations or concerns.” The costs of the expansion project are expected to be funded primarily by a Canadian Federal government grant and interest-free loan, certain investment tax credits, a secured credit facility and a portion of Neptune’s working capital. The expansion is anticipated to be completed by the end of our current fiscal year. Following the completion of its ongoing expansion project and before the end of 2014, Neptune intends to further expand its Sherbrooke plant to increase its annual production capacity to 500,000 kilograms of krill oil.
16
The new two-level facility currently being constructed is adjacent to Neptune’s initial production plant and will have a gross area of approximately 40,000 square feet. The facility will almost entirely be dedicated to Neptune’s production process. Neptune will continue to operate its initial facilities, which have a gross area of approximately 12,000 square feet and accommodate Neptune’s laboratories, administrative offices and initial production plant. The structure is designed to allow greater flexibility for Neptune’s production lines and is expected to improve Neptune’s efficiency and productivity.
Neptune adheres to Good Manufacturing Practices, or GMP, mandated by the Natural Health Products Directorate of Health Canada, or NHPD, and successfully passed the audit performed by the NHPD in May 2011.
Neptune also leases office space in facilities located at 225, Promenade du Centropolis, in Laval, Quebec, Canada, but anticipates a move to its new headquarters at 545, Promenade du Centropolis, in Laval, Quebec on October 1, 2012.
Sales/Distribution
Neptune sells NKO® and EKO™ in bulk oil or in capsules to multiple distributors, who commercialize these products under their private label in different market segments, including health food stores, mass (food and drug), direct sales (multi-level marketing, internet, catalogue, radio) and via healthcare professional recommendation. The encapsulation process is subcontracted to third parties in Canada, United States, Asia and Europe. While the company may have purchase orders in place with approximately 40 to 50 different distributors at any one time, the majority of the Company’s sales are concentrated with a relatively small group of distributors. As at February 29, 2012, five customers represented 73% of total trade accounts receivable of the Company. Agreements with these distribution partners may be terminated or altered by them unilaterally in certain circumstances. See “Risk Factors — Risks Related to the Company’s Business —The Company has a significant concentration of its accounts receivable and its revenue from a limited number of distributors.” In addition, the agreements between Neptune and its distributors contain certain customary indemnification provisions with respect to liability incurred from claims resulting from items that are the responsibility of the distributor, such as encapsulation or packaging.
ONEMIA™ is now in the early stages of commercialization and is being distributed in the United States by Acasti to physicians (who then can either provide it to their patients directly or via a website by using a dedicated ONEMIA™ medical food code website). Acasti also makes ONEMIA™ available via distributors and behind-the-counter in pharmacies. Acasti also intends to secure distribution partners to commercialize ONEMIA™ outside of the United States. See “Risk Factors — Risks Related to the Company’s Business — The Company may not be able to further penetrate core or new markets.”
During the 2012 fiscal year, approximately 41% of Neptune’s sales were made to customers in the United States, 23% to customers in Europe, 23% to customers in Australia and 12% to customers in Canada. Sales of Neptune products for the fiscal year ended February 29, 2012 amounted to $19,124,000, from $16,685,000 for the fiscal year ended February 28, 2011. Neptune’s sales are not cyclical or seasonal.
Intellectual Property
It is an important part of our business to obtain intellectual property protection for our technology, products, applications and processes and/or to maintain trade secrets. Our success depends, in part, on our ability to obtain, license and enforce patents, protect our proprietary information and maintain trade secret protection without infringing the proprietary rights of third parties. Our strategic approach is to file and/or license patent applications whenever possible to obtain patent protection. We also rely on trade secrets, proprietary unpatented information and trademarks to protect our technology and enhance our competitive position. We have confidence in our patents and will continue to take all appropriate actions needed to protect our intellectual property rights in the United States and elsewhere in the world as required.
The Company has a firm policy to protect its intellectual property rights, including its patents, trademarks and trade secrets, through legal action.Certain of Neptune’s competitors have been marketing, advertising and selling finished krill-based products
17
which we believe infringe on patents owned by Neptune or for which Neptune has exclusive rights. Neptune is taking legal actions against those companies in order to protect its intellectual property and its business. See “Risk Factors — Risks Related to the Company’s Intellectual Property —A failure by the Company to protect its intellectual property may have a material adverse effect on its ability to develop and commercialize its products.”
Brand Names and Trademarks
Neptune has filed and registered the trademarks OPA 3™ and NKO® in over thirty countries and has filed numerous trademark applications in various jurisdictions. Neptune OceanExtract™ and NKA™ are other trademarks of Neptune.
NKO® distributors use private labels with the NKO® logo displayed on them and with names and trademarks pre-approved by Neptune.
Acasti has applied for worldwide trademark protection of CaPre®, and has filed for U.S. trademark protection of ONEMIA™. Acasti also is the owner of the trademark BREAKING DOWN THE WALLS OF CHOLESTEROL™ in Canada and the United States. The trademark CaPre® is now registered in Canada, the United States, the European Union, Australia and China.
Patents
Neptune has the following patent portfolio:
Category | Description | Countries | ||||||||
| Issued | Pending | |||||||||
Novel Phospholipid / Flavonoid | Composition of Matter | 26 | 5 | |||||||
Cardiovascular Neurological health | Method of Use | 22 | 11 | |||||||
Extraction Process | Process | 32 | 1 | |||||||
In Canada, the United States and Europe, a patent is generally valid for 20 years from the date of first filing. Patent terms can vary slightly for other jurisdictions, with 20 years from filing being the norm. In certain jurisdictions exclusivity can be formally extended beyond the normal patent term to compensate for regulatory delays during the pre-market approval process.
Licensing Arrangements
Even though the Company uses, for its production, its own process technology, the Company also strategically exploits, within its intellectual property portfolio, an exclusive, irrevocable worldwide license on a patent related to an extraction process belonging to the University of Sherbrooke, province of Quebec (the “University”). The license agreement applies to the process of oil extracted from krill and from other marine and/or aquatic biomasses.
The license agreement clearly stipulates that the Company shall remain the sole owner of any improvement and/or modification and/or enhancement of the extraction process done and/or paid by the Company. This clause is significantly important. The license agreement also stipulates that the University shall remain the sole owner of any improvement and/or modification and/or enhancement of the extraction process done and/or paid by the University. Thus, the Company, for a period of 24 months following any such improvement and/or modification and/or enhancement by the University, has the right to enter into an exclusive license agreement with the University with respect to any such improvement and/or modification and/or enhancement. No such improvement and/or modification and/or enhancement have been, to this date, reported to the Company by the University.
The license agreement may be terminated (i) by way of agreement between the University and the Company; (ii) in the event of a default by the Company or the University; (iii) in the event of the insolvency or bankruptcy of the Company; or (iv) if the Company ceases to carry on its activities in the normal course of business.
18
The Company also benefits from a right of first refusal with respect to any research project for the development of a process to extract and purify oils originating from marine and freshwater biomasses like krill among others and from an option to purchase the intellectual property rights with respect to the results of the research, as it relates to krill, or other crustaceans, conducted by the University. The exercise price for this purchase option has been set at $275,000 by mutual agreement between the University and the Company, this price was contested by the researcher but has remained the same based on the decision of the Quebec Court of appeal rendered in January 2010. See “Business of the Company — Economic Dependence/Litigation”.
The Company has undertaken to pay an annual commission to a Company controlled by Mr. Henri Harland for services rendered as well as for the transfer in February 2001 to the Company of the license rights with the University, including the right of first refusal and of the option to purchase the intellectual property rights. This royalty of 1% on any sales and on other income of the Company is for an indeterminate period of time and it shall be paid semi-annually and disbursement of such royalty payment per year will not be superior to the Company’s net earnings before interest, taxes, depreciation and amortization (EBITDA).
Terms of the License Granted to Acasti
In 2008, Neptune and Acasti entered into a license agreement that provides Acasti with the right to use certain intellectual property rights of Neptune in order to develop novel APIs into commercial products for specific pharmaceutical markets, namely the OTC, the medical food and the prescription drug market. Effective August 7, 2011 and in accordance with the license agreement, Acasti abandoned its rights to develop products for the OTC.
Pursuant to the license agreement, Acasti has been granted a license to use Neptune’s intellectual property rights solely for the development, distribution and sale of products for use in the human cardiovascular field. Acasti is responsible for carrying out the research and development of the APIs, as well as required regulatory submissions and approvals and intellectual property filings. The license agreement provides that the products developed by Acasti must have a specified concentration of phospholipids.
Acasti is obligated under the license to pay Neptune until the expiration of Neptune’s patents on licensed intellectual property a royalty equal to the sum of (a) in relation to sales of products in the licensed field, if any, the greater of: (i) 7.5% of net sales, and (ii) 15% of Acasti’s gross margin; and (b) 20% of revenues from sub-licenses granted by Acasti to third parties, if any. The license will expire on the date of expiration of the last-to-expire of the licensed patent claims and/or continuation in part and/or divisional of the licensed patent claims. After the expiration of Neptune’s patents on licensed intellectual property, which is currently expected to occur in 2022, the license agreement will automatically renew for an additional period of 15 years, during which period royalties will equal half of those calculated according to the above formula. In addition, the license provides for minimum royalty payments notwithstanding the above of: year 1 - nil; year 2 - $50,000; year 3 - $200,000; year 4 - $225,000 (initially $300,000, but reduced to $225,000 following Acasti’s abandonment of its rights to develop products for the OTC market pursuant to the license agreement); year 5 - $700,000; and year 6 and thereafter - $750,000. Minimum royalties are based on contract years based on the effective date of the license, August 7, 2008.
Acasti has the option to pay future royalties in advance, in cash or through the issuance of shares, in whole or in part, based on an established economic model contained in the license agreement. Acasti can also abandon its rights under all or part of the license agreement and consequently remove itself from the obligation to pay all or part of the minimum royalties by paying a penalty equal to half of the next year’s minimum royalties. In addition, Acasti is committed to have its products, if any, manufactured by Neptune at prices determined according to different cost-plus rates for each of the product categories under the license.
Under the license, Acasti must buy its raw materials from Neptune at prices determined according to different cost-plus rates for each of the product categories under the license agreement. A copy of the Acasti license agreement is available on SEDAR atwww.sedar.com.
Terms of the License Granted to NeuroBio
In 2008, Neptune also entered into a license agreement that provides NeuroBio the same rights and obligations as provided to Acasti. See “Business of the Company — Intellectual Property — Licensing Arrangements — Terms of the License Granted to Acasti”. Pursuant to the license agreement, NeuroBio is permitted to use the licensed intellectual property rights solely for the development, distribution and sale of products for use in the human neurological field (all conditions, abnormalities and/or diseases related to cognitive function and/or affective and/or neurological systems).
19
The Neptune patents subject to the license with NeuroBio are the following:
Patent description | Patent # | Exclusivity | ||
Composition of Matter | WO 2003/011873 | 2024 | ||
Method of Use | WO 2002/102394 | 2024 | ||
Composition and Use | USSN 61/256,106 | 2030 |
NeuroBio is obligated under the license to pay Neptune until the expiration of Neptune’s patents on licensed intellectual property a royalty equal to the sum of (a) in relation to sales of products in the licensed field, if any, the greater of: (i) 7.5% of net sales, and (ii) 15% of NeuroBio’s gross margin; and (b) 20% of revenues from sub-licenses granted by NeuroBio to third parties, if any. The license will expire on the date of expiration of the last-to-expire of the licensed patent claims and/or continuation in part and/or divisional of the licensed patent claims. After the expiration of Neptune’s patents on licensed intellectual property, which is currently expected to occur in 2022, the license agreement will automatically renew for an additional period of 15 years, during which period royalties will equal half of those calculated according to the above formula. In addition, the license provides for minimum royalty payments notwithstanding the above of: years 1 and 2 - nil; year 3 - $50,000; year 4 - $200,000; year 5 - $300,000; year 6 - $900,000 and year 7 and thereafter - $1,000,000. Minimum royalties are based on contract years based on the effective date of the license, October 15, 2008.
NeuroBio has the option to pay future royalties in advance, in cash or through the issuance of shares, in whole or in part, based on an established economic model contained in the license. NeuroBio can also abandon its rights under all or part of the license agreement and consequently remove itself from the obligation to pay all or part of the minimum royalties by paying a penalty equal to half of the next year’s minimum royalties. In addition, NeuroBio is committed to have its products, if any, manufactured by Neptune at prices determined according to different cost-plus rates for each of the product categories under the license.
Under the license, NeuroBio must buy its raw materials from Neptune at prices determined according to different cost-plus rates for each of the product categories under the license agreement. A copy of the NeuroBio license agreement is available on SEDAR atwww.sedar.com.
Regulatory Environment
Commercial products developed or under development by Neptune, directly or through its subsidiaries, can be categorized as ingredients to be used in foods, dietary supplements or medical foods, or as APIs to be used in drug products.
Those ingredients may qualify as “novel foods” or “new dietary ingredients”, depending on final applications and countries where they are or will be marketed. Generally speaking, novel foods are defined as food substances that do not have a prior history of safe use or result from a process previously not used for foods. Similarly, a new dietary ingredient refers to a substance not previously used as a dietary supplement in humans. In the United States, the FDA (Centre for Food Safety and Nutrition) regulates matters associated with the safety of ingredients for use in food and dietary supplements. Any substance intentionally added to food is a food additive, thus requiring approval by the FDA, unless the substance is “Generally Recognized As Safe”, or GRAS, under the conditions of its intended use, or unless the use of the substance is otherwise excluded from the definition of a food additive. GRAS status may be achieved through a voluntary notification procedure. A mandatory notification process for a new dietary ingredient is also in place according to the Federal Food, Drug, and Cosmetic Act which requires that manufacturers and distributors who wish to market dietary supplements that contain new dietary ingredients notify the FDA.
In Canada, novel foods are regulated by the Novel Foods Regulation (under theFood and Drugs Act) which requires that a notification be made to the Health Products and Food Branch prior to the marketing or advertising of a novel food in the Canadian marketplace. Natural health products (equivalent to dietary or food supplements) sold in Canada are subject to theNatural Health
20
Products Regulations, which came into force on January 1, 2004. All natural health products must have a product license before they can be sold in Canada, which requires applicants to gather and provide detailed information about the quality, safety and efficacy of ingredients to be used for assessment and pre-market approval.
In Europe, the legislation governing nutritional supplements is enacted and enforced by each individual country’s governmental authorities. In an effort to harmonize the often differing regulations of its member states, the European Union adopted in 2002 the Food Supplements Directive. This directive seeks to harmonize the rules governing the composition, labelling and marketing of nutritional supplements throughout the European Union. The Food Supplements Directive outlines a specific process and timetable for the member states to bring their domestic legislation in line with the directive’s provisions. The directive, upon recommendation by the European Food Safety Authority, or EFSA, specifies what nutrients and nutrient sources may be used, identifies the levels at which these nutrients may be found in a supplement and the labelling and other information which must be provided on packaging.
APIs developed or under development by Acasti and NeuroBio are regulated through different procedures and requirements. In Canada, biopharmaceutical product candidates are regulated by theFood and Drugs Act and the rules and regulations promulgated thereunder, which are enforced by the Therapeutic Products Directorate. In the United States, drugs and biological product candidates are subject to regulation by the FDA (Center for Drug Evaluation and Research).
In Europe, the European Medicines Agency, or EMA, is the regulatory agency which controls all aspects of the development, manufacture and commercialization of drug products for the countries of the European Union. Each country of the European Union also has its own national regulatory agency which works under the umbrella of the EMA. These laws and regulations require the licensing of manufacturing and contract research facilities, carefully controlled research and testing of product candidates and governmental review and approval of results prior to marketing therapeutic product candidates. Additionally, they require adherence to good laboratory practices for pre-clinical safety testing in animals, good clinical practices during clinical testing and good manufacturing practices during production. The systems of new drug approvals in Canada, the United States and the European Union are generally considered to be among the most rigorous in the world.
In general, the steps required for approval of a new drug in Canada, the United States and Europe are:
1.Research
Prior to preclinical studies, a research phase takes place which involves characterization of the physical chemical properties and biological activity of the product. This is often followed by evaluation of efficacy in animal models.
2.Preclinical Studies
Preclinical studies involve evaluations of animal pharmacology and toxicity, pharmacokinetics and metabolism of a drug in animals to provide evidence of the safety, bioavailability and activity of the drug in animals. The results of these studies as well as the comprehensive descriptions of proposed human clinical studies are then submitted as part of the IND application to the FDA, its Canadian equivalent, a Clinical Trial Application, to the Biologics and Genetic Therapeutics Directorate, or BGTD, or its European equivalent, an Investigational Medicinal Product Dossier.
3.Clinical Trials
Phase I Clinical Trials: Phase I clinical trials are usually first-in-man trials and take from a few months to two years to complete. They are generally conducted on a small number of healthy human subjects to evaluate the drug’s safety, schedule and dose, pharmacokinetics and pharmacodynamics.
Phase II Clinical Trials: Phase II clinical trials take approximately one to three years to complete and are carried out on a relatively small to moderate number of patients (compared to Phase III) suffering from the targeted condition or disease to determine the drug’s efficacy, optimal doses, treatment regimens, pharmacokinetics, pharmacodynamics and dose response relationships. This phase also provides additional safety data and serves to identify possible common short-term side effects and risks in a larger group of patients. Phase II clinical trials often include randomization of patients as well as a placebo arm.
21
Phase III Clinical Trials: Phase III clinical trials take approximately two to five years to complete and involve tests on a much larger population of patients (several hundred to several thousand patients) suffering from the targeted condition or disease. These studies usually include randomization of patients, a placebo arm and blinding of both patients and investigators at geographically dispersed test sites (multi-centre trials) to establish clinical safety effectiveness.
New Drug Application: Upon completion of the Phase III clinical studies, the company sponsoring the new drug then assembles all the pre-clinical, clinical and manufacturing data and submits it to the FDA, the BGTD or the EMA as part of a New Drug Application in the United States, a New Drug Submission in Canada or a Market Authorization Application in Europe, respectively. The submission or application is then reviewed by the regulatory body for approval to market the product candidate. This process usually takes six months to two years to complete.
Obtained regulatory approvals, permits and authorizations:
Neptune has obtained the following regulatory approvals, permits and authorizations:
| • | European Food Safety Authority (EFSA) has approved NKO® as PARNUTS (food for particular nutritional use) for commercialization in the European Union. |
| • | European Food Safety Authority (EFSA) has approved NKO® as a Novel Food for commercialization in the European Union. |
| • | NKO® has received FDA GRAS (Generally Recognized as Safe) notification as a food ingredient in the United States. |
| • | NKO® has obtained approval as a Complementary Medicine from the Therapeutic Good Administration (TGA) in Australia. |
| • | NKO® has a natural product number (NPN) issued by Health Canada. |
| • | Health claims in Canada — Multiple claims for health benefits of NKO® approved by NHPD (7 claims). |
| • | Neptune’s production plant in Sherbrooke has been accredited as a nutraceutical GMP audit performed by NHPD. |
Competition
General
The nutraceutical and pharmaceutical industries are highly competitive. There are many pharmaceutical companies, biotechnology companies, public and private universities and research organizations actively engaged in the research and development of products that may be similar to our products. It is probable that the number of companies seeking to develop products and therapies similar to our products will increase. Many of these and other existing or potential competitors have substantially greater financial, technical and human resources than we do and may be better equipped to develop, manufacture and market products.
For instance, Aker BioMarine ASA, a Norway-based corporation that is in the business of harvesting and commercializing marine ingredients, launched a krill oil product under the brand name SuperbaTM in 2009. Enzymotec Ltd., an Israel-based biotechnology corporation, is also a krill oil supplier. These companies and others may develop and introduce products and processes competitive with ours.
Acasti’s potential competitors in the United States, Europe and Asia include large, well-established pharmaceutical companies as well as specialty pharmaceutical sales and marketing companies and specialized cardiovascular biopharmaceutical companies. These companies include GlaxoSmithKline plc, which currently markets Lovaza®, a prescription only omega-3 fatty acid indicated for patients with very high triglycerides, and Abbott Laboratories, which currently markets Tricor and Trilipix for the
22
treatment of very high triglycerides and mixed dyslipidemia. In addition, in July 2012, the FDA approved Vascepa™, a drug developed by Amarin Corporation plc, as an adjunct to diet to reduce triglyceride levels in adult patients with severe (triglycerides greater than or equal to 500mg/dL) hypertriglyceridemia (very high triglycerides).
Also, we are aware of other pharmaceutical companies that are developing products that, if approved, would compete with CaPre®. These include a free fatty acid form of omega-3 which is being developed by Omthera Pharmaceuticals and an omega-3 based drug candidate for hypertriglyceridemia being developed by Trygg Pharma. We also believe that certain other pharmaceutical companies are developing potential treatments for inflammatory and metabolic diseases based on omega-3 fatty acids. See “Risk Factors — Risks Related to the Company’s Business — The Company’s industry is subject to rapid technological change and competition.”
Employees
As at the date of this Annual Information Form, Neptune, along with Acasti and NeuroBio, has approximately 100 full-time employees working at its business offices in Laval and the Sherbrooke plant. We believe that Neptune employees possess specialized skills and knowledge in the following fields, which are valuable assets of the Company: (i) intellectual property protection, (ii) legal matters, (iii) marine biomasses, (iv) scientific issues, (v) marine oil extraction processes, (vi) quality assurance/quality control, (vii) clinical validation of biological therapeutic properties, (viii) regulatory compliance related to the Company’s operations, and (ix) commercialization and business development. Neptune is not a party to any collective bargaining agreement. Neptune considers its relations with its employees to be good and its operations have never been interrupted as the result of a labor dispute.
Economic Dependence/Litigation
Economic Dependence
Neptune sources the krill used in the manufacturing of its products generally from three suppliers. Neptune considers its relationship with its suppliers to be good and believes it is not dependent upon any of these suppliers since alternative sources of krill supply are readily available. See “Business of the Company — Supply of Krill”.
Litigation
University of Sherbrooke
In early 2001, Neptune acquired the rights to an exclusive and irrevocable worldwide license agreement with the University of Sherbrooke, province of Quebec (the “University”) on a patent related to an extraction process belonging to the University. The license applies to the process of oil extracted from krill and from other marine and/or aquatic biomasses. On August 18, 2004, the Company notified the University of its intention to exercise its $275,000 purchase option relating to the intellectual property. On August 23, 2004, University researchers filed an injunction against Neptune and the University demanding cancellation of the purchase option granted to Neptune by the University. In December 2008, a ruling was rendered against the Company. The judge determined that the Company had not exercised its option to purchase the intellectual property in August 2004, as claimed by the Company, and it had to pay additional royalties in the amount of $1,031,134 in addition to $145,000 in fees. The judge furthermore set at $1,776,000 the purchase price for the intellectual property.
Following the December 2008 judgement, Neptune appealed the ruling and requested an immediate stay of its execution. In January 2010, the Quebec Court of Appeal ruled in favor of the Company confirming in its ruling the Neptune’s right to exercise its purchase option relating to the intellectual property at a purchase price of $275,000 plus interests of $36,000, for a total of $311,000. The court also confirmed that the Company had exercised its option in August 18, 2004 and rejected all royalty claims to the exception of $36,000 plus interests of $11,000, for a total of $47,000 (for a grand total of $358,000 including the purchase price).
On December 21, 2011, the Company received a motion filed by the University, the worldwide registered owner of patents relating to the extraction process licensed to the Company, asking the Court to order the transfer of the intellectual property to Neptune for the purchase price of $358,000 set by the 2010 Quebec Court of Appeal ruling, as well as to order Neptune to pay the University $206,100 for the maintenance fees of the intellectual property. The case is currently pending and no trial dates have been set.
23
Schiff Nutrition Group Inc.
In August 2009, the Company received a complaint filed by Schiff Nutrition Group Inc., or Schiff, a former distributor of the Company’s products, in the US District Court for the District of Utah, alleging inter alia that the Company had failed to meet a certain delivery threshold. The Company filed a response denying all the allegations and a counterclaim wherein it alleged that Schiff had failed to pay the Company for shipments and products and that Schiff had committed trademark infringement, and claimed damages. The parties reached a settlement in December 2011 and the case was dismissed by the US District Court for the District of Utah in January 2012.
US Neutraceuticals LLC
On or around January 27, 2010, the Company and Acasti Pharma Inc. filed a Motion for the Issuance of a Permanent Injunction before the Quebec Superior Court against US Nutraceuticals LLC (d.b.a. Valensa), a US based Company. Neptune and Acasti are seeking inter alia an injunction ordering Valensa to amend some patent applications filed by Valensa to add Neptune as co-owner, or in the alternative to have Valensa assign these patent applications to Neptune, as well as punitive damages, loss of profit and loss of business opportunity for an amount currently established at $3,000,000. On September 28, 2011, Valensa filed its Defence wherein it denied Neptune/Acasti’s allegations and requested a dismissal of the Motion. Valensa also filed a Cross-Demand but only against Neptune, wherein it alleged breach of contract and damages in the amount of $2,300,000. The Company denies all material allegations made by Valensa. The case is currently pending and no trial dates have been set.
Aker Biomarine ASA and others
On November 13, 2009, Neptune filed a patent infringement lawsuit against Aker BioMarine ASA, Jedwards International, Inc and Virgin Antartic LLC, asserting its U.S. patent relating to a method of extraction of total lipids fractions from Krill. Neptune alleges that the Defendants have used solvents for the extraction of their krill oil, which are covered by the patent (US6,800,299) licensed to Neptune. As of the date of this Annual Information Form, the case is still pending before the federal district court in Massachusetts.
On October 4, 2011, the Company filed a Complaint in the US District Court for the District of Delaware against Aker Biomarine ASA, Aker Biomarine Antarctic USA Inc. and Schiff Nutrition International Inc. (Aker et al.) for the infringement of the Company’s US patent 8,030,348 and for damages. On December 19, 2011, Aker et al. filed Counterclaims denying any infringement, seeking the invalidity of the Company’s patent, and seeking an award for costs and damages. The proceedings have been stayed due to the reexamination of the patent.
Enzymotec Limited and others
On October 4, 2011, the Company filed a Complaint in the US District Court for the District of Delaware against Enzymotec Limited, Enzymotec USA Inc., Mercola.com Health Resources, LLC and Azantis Inc. for the infringement of the Company’s US patent 8,030,348 and for damages. On December 30, 2011, Enzymotec USA Inc. filed a Counterclaim denying any infringement, seeking the invalidity of the Company’s patent, and seeking an award for costs and damages. On December 30, 2011, Mercola.com Health Resources, LLC filed a Counterclaim denying any infringement, seeking the invalidity of the Company’s patent, and seeking an award for costs and damages. On December 30, 2011, Enzymotec Limited and Azantis Inc. filed a motion to dismiss for alleged lack of personal jurisdiction. The proceedings have been stayed for the reasons mentioned above.
Patent #1417211
On March 9, 2010, Neptune filed an appeal with the European Patent Office’s Board of Appeal contesting a 2009 decision of the European Patent Office regarding the European composition of phospholipids and use patent #1417211. As of the date of this Annual Information Form, the appeal process is currently pending.
24
RISK FACTORS
Investing in securities of the Company involves a high degree of risk. Prospective investors should carefully consider the following risks, as well as the other information contained in this Annual Information Form and the other information in our publicly filed documents before investing in securities of the Company. If any of the following risks actually occurs, the Company’s business, financial condition, liquidity, results of operation and prospects could be materially harmed. Additional risks and uncertainties, including those of which the Company is currently unaware or that it deems immaterial, may also adversely affect the Company’s business, financial condition, liquidity, results of operation and prospects.
Risks Related to the Company’s Business
The Company has a history of net losses and the Company may never achieve high profitability.
The Company has been reporting losses since the Company’s inception and, as at May 31, 2012, the Company has an accumulated deficit of $32,956,652. It is expected that the Company will continue to generate losses until income from product sales generate sufficient revenues to fund Neptune’s and its subsidiaries’ continuing operations, including research and product development, which the Company cannot assure you will occur in the near term or at all.
The Company’s near term success depends largely on the continued commercialization of NKO® and EKO™.
The Company’s ability to generate revenues in the foreseeable future is primarily based on the commercialization success of NKO® and EKO™. For the fiscal year 2012, revenues generated from the sale of NKO® and EKO™ to our distribution partners accounted for nearly all of the Company’s total consolidated revenues. Although the Company is developing other products that contain krill, all of them are at earlier stages of development and none of them may reach the clinical trial phase, obtain regulatory approval or, even if approved, be successfully commercialized.
The overall commercialization success of NKO® and EKO™ depends on several factors, including:
| • | continued market acceptance of NKO® and EKO™ by the nutraceutical market and medical community; |
| • | the amount of resources devoted by the Company’s distribution partners to continue the commercialization efforts of NKO® and EKO™ in our core geographic markets; |
| • | maintaining supply agreements to ensure the availability of krill in order to produce sufficient krill oil to meet the order demands of the Company’s distribution partners for NKO® and EKO™; |
| • | receipt of regulatory approvals for NKO® and EKO™ from regulatory agencies in certain territories in which the Company wishes to expand its commercialization efforts; |
| • | the number of competitors in the Company’s market; and |
| • | protecting and enforcing the Company’s intellectual property and avoiding patent infringement claims. |
The Company relies on third parties for the supply of raw materials and the distribution and commercialization of its products and such reliance may adversely affect the Company if the third parties are unable or unwilling to fulfill their obligations.
Part of the Company’s strategy is to enter into and maintain arrangements with third parties related to the development, clinical testing, marketing, distribution and commercialization of its products. The Company’s revenues are dependent on the successful efforts of these third parties, including the efforts of the Company’s distribution partners. Entering into strategic relationships can be a complex process and the interests of the Company’s distribution partners may not be or remain aligned with the Company’s interests. Some of the Company’s current and future distribution partners may decide to compete with the Company, refuse or be unable to fulfill or honour their contractual obligations to the Company, or change their plans to reduce their commitment to, or even abandon,
25
their relationships with the Company. There can be no assurance that our distribution partners will market the Company’s products successfully or that any such third-party collaboration will be on favourable terms. The Company may not be able to control the amount and timing of resources the Company’s distribution partners devote to the Company’s products. In addition, the Company may incur liabilities relating to the distribution and commercialization by its distributors of its krill oil products. While the agreements with such distributors generally include customary indemnification provisions indemnifying the Company for liabilities relating to the encapsulation or packaging of its krill oil products, there can be no assurance that these indemnification rights will be sufficient in amount, scope or duration to fully offset the potential liabilities associated with the Company’s distributors handling and use of our products. Any such liabilities, individually or in the aggregate, could have a material adverse effect on our business, financial condition or results of operations.
The Company has a significant concentration of its accounts receivable and its revenue from a limited number of distributors.
As at February 29, 2012, five distributors represented 73% of total trade accounts receivable of the Company. During the year ended February 29, 2012, the Company realized sales from the nutraceutical segment equalling $6,414,659 from two distributors. Sales to these distributors represented 20.8% and 12.8% of the Company’s consolidated sales. Agreements with these or other significant distribution partners may be terminated or altered by them unilaterally in certain circumstances. Any adverse change in the relationship with the Company’s principal distributors could have a material adverse effect on the Company’s business, consolidated results of operations, financial condition and cash flows.
The Company may be unable to manage its growth efficiently.
The Company’s future financial performance and its ability to commercialize its products and to compete effectively will depend, in part, on its ability to manage any future growth effectively. To that end, the Company must be able to increase its production capabilities, hire, train and integrate additional management, and potentially administer internal sales and marketing personnel on an effective and efficient basis. The Company is currently undergoing an expansion project of its manufacturing facility with the expectation that the new facility, when completed, will double production capacity. Even after completion of the expansion, there can be no guarantee that the Company will be able to meet the product order demands of its distributors.
The Company may not be able to accomplish any of the above actions, and its failure to do so could prevent it from successfully growing. Any increase in resources devoted to manufacturing, research, product development and sales, marketing and distribution efforts without a corresponding increase in the Company’s operational, financial and management information systems could have a material adverse effect on the Company’s business, financial condition and results of operations.
The Company may not be able to further penetrate core or new markets.
If the Company fails to further penetrate its core markets and existing geographic markets or successfully expand its business into new markets, the growth in sales of the Company’s products, along with the Company’s operating results, could be negatively impacted. The Company’s ability to further penetrate its core markets and existing geographic markets or to successfully expand its business into additional countries in Europe, Asia or elsewhere, to the extent the Company believes that it has identified attractive geographic expansion opportunities in the future, is subject to numerous factors, many of which are beyond the Company’s control. The Company cannot assure that its efforts to increase market penetration in its core markets and existing geographic markets will be successful. The Company’s failure to do so could have a material adverse effect on the Company’s operating results.
The Company is dependent on a single manufacturing facility.
The Company owns, manages and operates a manufacturing, processing and packaging facility in Sherbrooke, Québec that handles the production of all of the Company’s krill oil. Accordingly, it is highly dependent on the uninterrupted and efficient operation of its manufacturing facility. Currently, the manufacturing plant is undergoing a significant expansion project in an effort to increase its krill oil production capabilities. We cannot assure you that the expansion project will be implemented in a timely and cost efficient manner, and that our current production of krill oil will not be adversely affected by the operational challenges of implementing the expansion project. If operations at the Company’s manufacturing plant were to be disrupted as a result of equipment
26
sfailures, natural disasters, fires, accidents, work stoppages, power outages or other reasons, the Company’s business, financial condition and/or results of operations could be materially adversely affected. Lost sales or increased costs that the Company may experience during the disruption of operations may not be recoverable under the Company’s insurance policies, and longer-term business disruptions could result in a loss of customers. If this were to occur, the Company’s business, financial condition and operations could be negatively impacted.
The Company must attract and retain skilled labor in order to maintain and increase its business.
The Company’s ability to sustain and expand its operations depends in part on its ability to attract and retain skilled manufacturing workers, equipment operators, engineers and other technical personnel. Demand for these workers is currently high and the supply is limited, particularly in the case of skilled and experienced machinists and engineers. The Company will be required to retain additional skilled workers upon the completion of its manufacturing facility. Further, the Company may be faced with increased training costs and reduced productivity as it trains new employees hired to meet the Company’s increasing krill oil production needs. Additionally, a significant increase in the wages paid by competing employers could result in a reduction in the Company’s skilled labor force, increases in the rates of wages it must pay or both. If the Company’s compensation costs increase or it cannot attract and retain skilled labor, including engineers and machinists, the Company’s earnings could be reduced, and production capacity and growth potential could be impaired.
The Company may not be able to attract, hire and retain key management and personnel could negatively affect its business.
We depend substantially on our ability to hire, train, motivate and retain high quality personnel, especially our scientists and management team. Particularly, in light of the limited number of employees that cover our numerous programs and key functions, if we are unable to retain existing personnel or identify or hire additional personnel, we may not be able to research, develop, commercialize or market our products and product candidates as expected or on a timely basis and we may not be able to adequately support current and future alliances with strategic partners.
Furthermore, if we were to lose key management personnel, such as Henri Harland, our President and Chief Executive Officer, we would lose a portion of our institutional knowledge and technical know-how, potentially causing a substantial delay in one or more of our development programs until adequate replacement personnel could be hired and trained. Mr. Harland has been President and Chief Executive Officer of the Company since its incorporation on October 9, 1998. He is the founder of the Company and has been involved in our krill research project since 1991. Other than our stock option plan, we have not adopted any policies or entered into any agreements specifically designed to motivate officers or other employees to remain with us. We do not have key man life insurance policies on the lives of most of our key personnel, including Henri Harland.
The Company’s current and future clinical trials may prove unsuccessful or be delayed by certain factors.
The Company is not able to predict the results of pre-clinical and clinical testing of its product candidates. It is not possible to predict, based on studies or testing in laboratory conditions or in animals, whether a product candidate will prove to be safe or effective in humans. Further, preclinical data may not be sufficient for regulators to accept positive clinical data for approval to commercialize a product. Pre-clinical data must have been conducted to high regulatory standards and may be found, on review by health regulatory authorities, to be of insufficient quality to support an application for commercialization of a product. In addition, success in one stage of testing is not necessarily an indication that the particular product will succeed in later stages of testing and development. Further, clinical trials require the enrollment of patients and the Company may experience difficulties identifying and enrolling suitable human subjects for ongoing and future trials of its products. This could be as a result of a number of factors including, but not limited to, design protocol, the size of the available patient population, the eligibility criteria for participation in the clinical trials, and the availability of clinical trial sites.
For example, Acasti is developing CaPre®, a prescription drug candidate being developed to address the treatment of hypertriglyceridemia. CaPre® is currently being evaluated in two Phase II clinical trials. The Company’s ability to commercialize any of its products, including CaPre®, is dependent upon the success of product development efforts and the success of clinical studies. If these clinical trials and product development efforts fail to produce satisfactory results, or if the Company is unable to maintain the financial and operational capability to complete these development efforts, it may be unable to generate revenues for this and other product candidates.
27
A number of companies in the life sciences industry have suffered significant setbacks in advanced clinical trials, even after positive results in earlier trials. Share prices of biotechnology companies have declined significantly in certain instances where clinical results were not favourable, were perceived negatively or otherwise did not meet expectations. Unfavourable results or negative perceptions regarding the results of clinical trials for any of the Company’s product candidates currently under development could cause the Company’s share price to decline significantly.
The Company may not achieve its publicly announced milestones on time.
From time to time, the Company publicly announces the timing of certain events it expects to occur. These statements are forward-looking and are based on the best estimate of management at the time relating to the occurrence of such events. However, the actual timing of such events may differ from what has been publicly disclosed. Events such as completion of a clinical program, discovery of a new product candidate, filing of an application to obtain regulatory approval, beginning of commercialization of certain products or product candidates, or announcement of additional clinical programs for a product candidate may vary from what is publicly disclosed. CaPre®, Acasti’s leading drug candidate, is currently being evaluated in two Phase II clinical trials. The Company cannot assure you that the clinical trials for CaPre® or any other of the Company’s or its subsidiaries’ product candidates will be completed, that it will make regulatory submissions or receive regulatory approvals as planned, or that it will be able to adhere to its current schedule for the scale-up of manufacturing and launch of any of its products. These variations in timing may occur as a result of a series of events, including the nature of the results obtained during a clinical trial or during a research phase, problems with a supplier or a distribution partner or any other event having the effect of delaying the publicly announced timeline. The Company undertakes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, after the distribution of this Annual Information Form, except as otherwise required by law. Any variation in the timing of certain events having the effect of postponing such events could have a material adverse effect on the Company’s business plan, financial condition or operating results.
The Company may require additional funding and may not be able to raise the capital necessary to fund all or part of its capital requirements.
The Company may require substantial additional funds to increase production capacity and/or for further research and development, scheduled clinical testing, regulatory approvals and the commercialization of its products. The Company may seek additional funding for these purposes through public or private equity or debt financing, joint venture arrangements, and collaborative arrangements with other pharmaceutical companies, and/or from other sources. There can be no assurance that additional funding will be available on acceptable terms or at all to enable us to continue and complete the research and development of our product candidates and their successful commercialization. Should the Company fail to obtain the necessary capital, it may be required to delay, reduce or eliminate one or more of its various research and development programs or seek financial support from one of its strategic partners or from third-parties who may require that the Company waive significant rights regarding protection of its proprietary technologies or offer it financial support on less favourable terms than those normally acceptable to the Company.
If product liability lawsuits are brought against the Company, they could result in costly and time-consuming litigation and significant liabilities.
The development of human therapeutic products involves an inherent risk of product liability claims and associated adverse publicity. The Company’s products may be found to be, or to contain substances that are, harmful to the health of its consumers. This sort of finding may expose the Company to substantial risk of litigation and liability and/or forced to discontinue production of certain products.
The Company has secured a product liability insurance policy, renewable on an annual basis, to cover civil liability claims relating to its products in an amount equal to $5,000,000 per year for all such claims. The Company also maintains a quality-assurance process that is QMP (Quality Management Program) certified by the Canadian Food Inspection Agency. However, this coverage may not insure against all claims made.
28
Product liability insurance is costly, often limited in scope, and could be unavailable or only available on terms unfavourable to the Company. There can be no assurance that the Company will be able to obtain or maintain insurance on reasonable terms or to otherwise protect itself against potential product liability claims that could impede or prevent commercialization of the Company’s future products and product candidates. Furthermore, a product liability claim could tarnish the Company’s reputation, whether or not such claims are covered by insurance or are with or without merit. A product liability claim against the Company or the withdrawal of a product from the market could have a materially adverse effect on the Company’s business or its financial condition.
The Company may be adversely affected by environmental and safety regulations or concerns.
The Company’s krill oil extraction process involves the use of certain hazardous materials, including acetone. The Company is subject to federal, provincial and municipal laws and regulations governing the use, manufacture, storage, handling and disposal of such materials and certain waste products. In the event of an accident that involves hazardous materials, the Company could be held liable for damages, which could exceed the resources of the Company. There can be no assurance that the Company will not be required to incur significant costs to comply with regulatory requirements in the future, or that the operations, business or assets of the Company will not be materially adversely affected by current or future legislative or regulatory requirements. The Company currently has no immediate plans for major capital expenditures in respect of environmental protection installations.
The Company will be required to obtain a permit from the Minister of Environment Quebec that will allow it to produce in excess of the 100,000 kilograms currently permitted. We may not be successful in obtaining such permit on favourable terms or at all, or in a timely manner. Any of the foregoing would have a material adverse effect on our business, operations and financial condition.
The Company is heavily dependent on third parties to obtain certain raw materials necessary to develop and produce its products.
The Company depends on third parties to obtain certain raw materials necessary to develop and produce its products. If the Company is no longer able to obtain raw materials, including krill, from one or more of its suppliers on terms reasonable to the Company or at all, the Company’s revenues could suffer. This could also have a significant impact on the Company’s capacity to complete certain of its current research and development projects and, accordingly, would negatively affect its projected commercial and financial growth. In addition, a significant increase in the price of raw materials that cannot be passed on to the Company’s distributors could have a material adverse effect on the Company’s results of operations and financial condition. While other potential alternative suppliers of raw materials may be identified, they must first pass intensive validation tests to ensure their compliance with product specifications. No assurance can be given regarding the successful outcomes of such tests or the Company’s ability to secure alternate sources of supply at competitive pricing and upon fair and reasonable contractual terms and conditions.
The Company’s industry is subject to rapid technological change and competition.
The Company operates in a sector that is subject to rapid and substantial change. There can be no assurance that products developed by others will not render the Company’s products, product candidates or technologies non-competitive or that the Company will be able to keep pace with technological developments. Competitors may have developed or may be in the process of developing technologies that could be the basis for competitive products. Some of these products may prove more effective and less costly than products developed by the Company or its product candidates. Scientific and technological developments and regulatory requirements may, within a relatively short timeframe, render the products and processes developed or planned by the Company obsolete.
Competition in the health and nutrition industry and in the pharmaceutical sector is extremely intense. Many companies, as well as research organizations, currently engage in, or have in the past engaged in, efforts related to the development of products similar to the Company’s products and product candidates. The Company competes with companies that produce similar or identical products or that proposes different approaches to the separation or purification of components of krill.
For instance, Aker BioMarine ASA, a Norway-based corporation that is in the business of harvesting and commercializing marine ingredients, launched a krill oil product under the brand name SuperbaTM in 2009. Enzymotec Ltd., an Israel-based
29
biotechnology corporation, is also a krill oil supplier. These companies and others may develop and introduce products and processes competitive with ours. Acasti’s potential competitors both in the United States, Europe and Asia include large, well-established pharmaceutical companies as well as specialty pharmaceutical sales and marketing companies and specialized cardiovascular biopharmaceutical companies. These companies include GlaxoSmithKline plc, which currently markets Lovaza®, a prescription only omega-3 fatty acid indicated for patients with very high triglycerides, and Abbott Laboratories, which currently markets Tricor and Trilipix for the treatment of very high triglycerides and mixed dyslipidemia. In addition, in July 2012, the FDA approved Vascepa™, a drug developed by Amarin Corporation plc, as an adjunct to diet to reduce triglyceride levels in adult patients with severe (triglycerides greater than or equal to 500mg/dL) hypertriglyceridemia (very high triglycerides). Also, we are aware of other pharmaceutical companies that are developing products that, if approved, would compete with CaPre®. These include a free fatty acid form of omega-3 which is being developed by Omthera Pharmaceuticals and an omega-3 based drug candidate for hypertriglyceridemia being developed by Trygg Pharma. We also believe that certain other pharmaceutical companies are developing potential treatments for inflammatory and metabolic diseases based on omega-3 fatty acids.
These and other competitors may have greater resources than the Company. Accordingly, no assurance can be given that products developed by these other companies or their technology will not affect the Company’s ability to compete in the nutraceutical market. There is a risk that one or more of the Company’s competitors may develop more effective or more affordable products than the Company, or may achieve earlier patent protection or product commercialization that the Company, or that such competitors will commercialize products that will render the Company’s product candidates obsolete, possibly before the Company is able to commercialize them.
The Company is subject to foreign currency fluctuations.
The Company is exposed to the financial risk related to the fluctuation of foreign exchange rates and the degrees of volatility of those rates. Foreign currency risk is limited to the portion of the Company’s business transactions denominated in currencies other than the Canadian dollar. During the 2012 fiscal year, approximately 72% of the Company’s revenues were in United States dollars and 24% were in Euros. If the values of foreign currencies including the United States dollar and Euro fluctuate significantly more than expected in the foreign exchange markets, the Company’s operating results and financial condition may be adversely affected.
The Company enters into currency forwards to purchase or sell amounts of foreign currency in the future at predetermined exchange rates. The purpose of these currency forwards is to fix the risk of fluctuations in future exchange rates. Significant fluctuations in the rate of exchange could adversely affect the Company’s financial performance. There is a risk of loss arising from an eventual weakening of the United States dollar or Canadian dollar.
The Company may be negatively impacted by the value of its intangible assets.
The Company is required to review the carrying value of its intangible assets for impairment annually or when events change. Intangible assets include net book value of product rights, trademarks and process know-how covered by certain patented and non-patented information. Management reviews the carrying value based on projected future results. If events such as generic competition or inability to manufacture or obtain supply of product occur that may cause sales of the related products to decline, the Company adjusts the projected results accordingly. Any impairment in the carrying value results in a write-down of the intangible asset that is charged to income during the period in which the impairment is determined. Any write-down of intangible assets may have a material adverse effect on the Company’s results of operations in the period in which the write-down occurs.
Risks Related to the Company’s Intellectual Property
The Company’s commercial success depends, in part, on its intellectual property rights.
The Company’s success depends in part on its ability to develop products, obtain patents, protect its trade secrets and operate without infringing third-party exclusive rights or without others infringing the Company’s exclusive rights or those granted to it under license. The Company has filed and is actively pursuing patent applications in Canada, the United States, Europe and elsewhere in the world. The patent position of pharmaceutical firms is generally uncertain and involves complex legal, factual and scientific issues, several of which remain unresolved. The Company does not know whether all of its pending patent applications will be granted and
30
whether the Company will be able to develop other patentable proprietary technology and/or products. Furthermore, the Company cannot be completely certain that its existing or future patents provide a definitive and competitive advantage or afford protection against competitors with similar technology. Furthermore, the Company cannot give any assurance that such patents will not be challenged or circumvented by others using alternative technology or whether existing third-party patents will prevent the Company from marketing its products. In addition, competitors or potential competitors may independently develop, or have independently developed products as effective as those of the Company or invent or have invented other products based on the Company’s patented products.
If third-party licenses are required, the Company may not be able to obtain them, or if obtainable, that they will be available on reasonable terms. Furthermore, the Company could develop or obtain alternative technologies related to third-party patents that may inadvertently cover its products. Inability to obtain such licenses or alternative technologies could delay the market launch of certain Neptune products, or even prevent the Company from developing, manufacturing or selling certain products. In addition, the Company could incur significant costs in defending itself in patent infringement proceedings initiated against it or in bringing infringement proceedings against others.
In some cases, the Company cannot determine with any certainty whether it has priority of invention in relation to any new product or new process covered by a patent application or if it was the first to file a patent application for any such new invention. Furthermore, in the event of patent litigation there can be no assurance that the Company’s patents would be held valid or enforceable by a court of competent jurisdiction or that a court would rule that the competitor’s products or technologies constitute patent infringement.
Moreover, a significant part of the Company’s technological know-how constitutes trade secrets. The Company requires that its employees, consultants, advisers and collaborators sign confidentiality agreements. However, these agreements may not provide adequate protection in the event of unauthorized use or disclosure of the Company’s trade secrets, know-how or other proprietary information.
Claims that the Company’s technology or products infringe on intellectual property rights of others could be costly to defend or settle, could cause reputational injury and would divert the attention of management and key personnel, which in turn could have a material adverse effect on the Company’s business, results of operations, financial condition and cash flows.
A failure by the Company to protect its intellectual property may have a material adverse effect on its ability to develop and commercialize its products.
The Company will be able to protect its intellectual property rights from unauthorized use by third parties only to the extent that its intellectual property rights are covered and protected by valid and enforceable patents or are effectively maintained as trade secrets. The Company tries to protect its intellectual property position by, among other things, filing patent applications related to its proprietary technologies, inventions and improvements that are important to the development of its business.
The Company is a plaintiff in multiple ongoing patent infringement cases with several parties. For instance, on October 4, 2011, the Company filed a complaint in the US District Court for the District of Delaware against Aker Biomarine ASA, Aker Biomarine Antarctic USA Inc. and Schiff Nutrition International Inc. for the infringement of the Company’s US patent 8,030,348 and for damages. On December 19, 2011, the parties filed counterclaims denying any infringement, seeking the invalidity of the Company’s patent, and seeking an award for costs and damages. The proceedings have been stayed due to the reexamination of the patent.
Because the patent position of pharmaceutical companies involves complex legal and factual questions, the issuance, scope, validity, and enforceability of patents cannot be predicted with certainty. Patents, if issued, may be challenged, invalidated or circumvented. If the Company’s patents are invalidated or found to be unenforceable, it would lose the ability to exclude others from making, using or selling the inventions claimed. Moreover, an issued patent does not guarantee the Company the right to use the patented technology or commercialize a product using that technology. Third parties may have blocking patents that could be used to prevent the Company from developing its product candidates, selling its products or commercializing its patented technology. Thus, patents that the Company owns may not allow it to exploit the rights conferred by its intellectual property protection.
31
The Company also relies on trade secrets, know-how and technology, which are not protected by patents, to maintain its competitive position. The Company tries to protect this information by entering into confidentiality agreements with parties who have access to such confidential information, such as its current and prospective suppliers, distributors, manufacturers, commercial partners, employees and consultants. Any of these parties may breach the agreements and disclose confidential information to the Company’s competitors. It is possible that a competitor will make use of such information, and that the Company’s competitive position could be disadvantaged.
Enforcing a claim that a third party infringes on, has illegally obtained or is using an intellectual property right, including a trade secret or know-how, is expensive and time-consuming and the outcome is unpredictable. In addition, enforcing such a claim could divert management’s attention from the Company’s business. If any intellectual property right were to be infringed, disclosed to or independently developed by a competitor, the Company’s competitive position could be harmed. Any adverse outcome of such litigation or settlement of such a dispute could subject the Company to significant liabilities, could put one or more of its patents at risk of being invalidated or interpreted narrowly, could put one or more of its pending patent applications at risk of not issuing, or could facilitate the entry of generic products. Any such litigation could also divert the Company’s research, technical and management personnel from their normal responsibilities.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of the Company’s confidential information could be compromised by disclosure during this type of litigation. For example, confidential information may be disclosed, inadvertently or as ordered by the court, in the form of documents or testimony in connection with discovery requests, depositions or trial testimony. This disclosure would provide the Company’s competitors with access to its proprietary information and may harm its competitive position.
Risks Related to the Company’s Industry
The Company is subject to significant government regulations.
The research, development, production and commercialization of the Company’s products is generally subject to comprehensive regulations under Health Canada’s Therapeutic Products Program and other regulatory bodies in Canada and various regional, national and local regulatory bodies, including the FDA in the United States. See “Business of the Company - Regulatory Environment”. These regulations may require the (i) approval of manufacturing facilities, including adhering to GMPs during the production, storage, controlled research and quality testing of products, (ii) review and approval of applications to establish the safety and efficacy of the product for each marketing claim sought, and (iii) the control of marketing activities. The process of obtaining required approvals (such as from the FDA and Health Canada) can be costly, time consuming and without guaranteed certainty of approval. Regulatory authorities may change processes, laws, regulations and policies related to product development or commercialization and business operations and require the Company to make changes to the product, its claims or its operations. The Company could encounter difficulties or incur excessive costs in obtaining the necessary approvals or permits, which could delay or prevent the commercialization and production of its new products.
In December 2006, Congress passed legislation requiring companies that manufacture or distribute OTC products or dietary supplements to report serious adverse events allegedly associated with their products to the FDA and institute recordkeeping procedures for all alleged adverse events (serious and non-serious). The legislation requires manufacturers and distributors of OTC products or dietary supplements to report to the FDA any serious adverse event reports received, even if the party making the report provides no medical or other information to the manufacturer or distributor. There is a risk that consumers, the press or government regulators could misinterpret adverse event reports as evidence of causation by the ingredient or product complained of, which could lead to consumer confusion, damage to our reputation, banned or recalled ingredients or products, increased insurance costs, class action litigation and a potential increase in product liability litigation, among other things. Distribution of the Company’s products outside Canada and the United States is also subject to comprehensive government regulation. Regulations, specifically requirements in respect of product releases on the market and the time involved in respect of regulatory assessment and the sanctions imposed in the event of infringement vary from country to country. No assurance can be given that the Company will obtain the requisite approvals in the relevant countries or that it will not incur significant expense in obtaining regulatory approvals or maintaining them in effect.
32
Failure to obtain the necessary regulatory approvals, the suspension or revocation of current approvals or any failure to comply with regulatory requirements may have a material adverse effect on the Company’s operations, its financial situation and its operating results.
Neptune’s majority owned subsidiaries, Acasti and NeuroBio, are developing products and product candidates for the pharmaceutical market. Products intended for therapeutic use for humans are governed by a wide array of regulatory agencies. For most of these products, applicable regulations require testing and government review and approval prior to marketing the product. See “Business of the Company - Regulatory Environment”. This procedure can take a number of years and involves the expenditure of substantial resources. Any failure or delay by the Company to obtain regulatory approvals or clearances could adversely affect the marketing of any products it developed and its ability to generate product revenue. There can be no assurance that any of the Company’s pharmaceutical product candidates will be approved by any regulatory agency on a timely basis, or at all. Regulatory approval in Canada, Europe and the United States does not assure approval by other national regulatory agencies, although often test results from one country may be used in applications for regulatory approval in another country.
In the event that a regulatory authority revokes any clearances or approvals granted in respect of the Company’s pharmaceutical products, the Company’s business and financial condition could be adversely affected. Numerous statutes and regulations govern the manufacture and sale of pharmaceutical products in Canada, the United States and other countries where the Company markets or intends to market its products. Such laws and regulations govern, among other things, the approval of manufacturing facilities, testing procedures and controlled research, non-clinical and clinical data required prior to and after marketing approval, compliance with GMP affecting production and storage, the advertising and labelling of products and the reporting of adverse events. Failure to comply with statutes and regulations could result in warning letters, fines and other civil penalties, unanticipated expenditures, withdrawal of regulatory approval, delays in approving or refusing to approve a product, product recall or seizure, interruption of production, operating restrictions, injunctions or criminal sanctions. The Company and its manufacturers and suppliers are also subject to numerous federal, state, provincial and local laws relating to such matters as safe working conditions, manufacturing practices, environmental protection, fire hazard control and disposal of hazardous or potentially hazardous substances.
The global regulatory environment continues to evolve with changes to regulations, rules, standards and guidelines and the establishment of new health authorities and/or mergers of divisions within them. The Company’s existing or future regulatory clearances or approvals may be negatively affected as a result of such changes or reorganization.
The market for the Company’s products has not been fully defined.
The Company believes that products based on its core technology will have numerous applications and that there is a growing market for the products that it has developed. However, there can be no assurance that these assumptions will prove justified, particularly considering competition from existing or new products and considering the uncertain commercial viability of the Company’s products. Therefore, there can be no assurance that any of the Company’s products in development or products recently launched will achieve market acceptance.
The degree of market acceptance for the Company’s products and those of its customers will depend upon a number of factors, including competitive pricing, the extent to which the products fulfill customer expectations and demands, the receipt of regulatory approvals, the establishment and demonstration in the medical community of the clinical efficacy and safety of the products, the establishment and demonstration of the potential advantages over competing products and, in the case of pharmaceuticals, the establishment and demonstration of the potential advantages over existing and new treatment methods and the reimbursement policies of government and third-party payers, and in the case of the Company’s nutraceuticals, the acceptance of the listing of the product and appropriate distribution with large retailers. There can be no assurance that consumers, physicians, patients, payers, the medical community in general, distributors or retailers will accept and utilize any existing or new products that may be developed by the Company.
33
Legislative or regulatory reform of the health care system may adversely affect the Company’s business and financial condition.
The Company’s revenues from sales of pharmaceutical products will depend in part on reimbursement policies and regulations of government health administration authorities, private health insurers and other organizations. The business and financial condition of pharmaceutical companies will continue to be affected by the efforts of governments and third-party payers to contain or reduce the costs of health care through various means. For example, in certain markets, including Canada, pricing or profitability of prescription pharmaceuticals is subject to government control. In the United States there have been, and the Company expects that there will continue to be, a number of federal and state proposals to implement similar government controls. In addition, an increasing emphasis on managed health care in the United States has increased and will continue to increase the pressure on pharmaceutical pricing. In Canada, the United States and elsewhere, sales of prescription pharmaceutical products are dependent, in part, on the availability of reimbursement to the consumer from third-party payers, such as government and private insurance plans. Third-party payers are increasingly challenging the prices charged for medical products and services. To the extent the Company succeeds in bringing new products to market, there can be no assurance that these products will be considered cost-effective and reimbursement to consumers will be available or will be sufficient to allow the sale of these products on a competitive basis. The Company may not be able to obtain prices for its products under development that will make them commercially viable.
Risks Related to the Company’s Securities
The following risk factors apply with respect to the securities of the Company.
The price of the Company’s shares may fluctuate.
Market prices for securities in general, and that of pharmaceutical and nutraceutical companies in particular, tend to fluctuate. Factors such as the announcement to the public or in various scientific or industry forums of technological innovations, new commercial products, patents, exclusive rights obtained by the Company or others, results of pre-clinical and clinical studies by the Company or others, a change of regulations, publications, financial results, public concerns over the risks of pharmaceutical products and dietary supplements, future sales of securities by the Company or its shareholders and many other factors could have considerable effects on the price of the Company’s securities.
The market price of the Company’s shares could decline as a result of future issuances or actual or potential sales.
The market price of the common shares could decline as a result of future issuances by the Company or sales by its existing holders of common shares, or the perception that these sales could occur. Sales by shareholders might also make it more difficult for Neptune to sell equity securities at a time and price that Neptune deems appropriate, which could reduce its ability to raise capital and have an adverse effect on its business.
The market price of the Company’s shares could decline as a result of operating results falling below the expectations of investors or fluctuations in operating results each quarter.
The Company’s revenues and expenses may fluctuate significantly and any failure to meet financial expectations may disappoint securities analysts or investors and result in a decline in the price of the Company’s common shares. The Company’s revenues and expenses have fluctuated in the past and are likely to do so in the future. These fluctuations could cause the Company’s share price to decline. Some of the factors that could cause revenues and expenses to fluctuate include the following:
| • | the inability to complete product development in a timely manner that results in a failure or delay in receiving the required regulatory approvals or allowances to commercialize product candidates; |
| • | the timing of regulatory submissions and approvals; |
| • | the timing and willingness of any current or future collaborators to invest the resources necessary to commercialize the Company’s products; |
34
| • | the outcome of any litigation; |
| • | changes in foreign currency fluctuations; |
| • | the timing of achievement and the receipt of milestone payments from current or future third parties; |
| • | failure to enter into new or the expiration or termination of current agreements with third parties; and |
| • | failure to introduce the Company’s products to the market in a manner that generates anticipated revenues. |
If the Company’s quarterly operating results fall below the expectations of investors or securities analysts, the price of the Company’s common shares could decline substantially. Furthermore, any quarterly fluctuations in the Company’s operating results may, in turn, cause the price of its stock to fluctuate substantially.
The Company does not currently intend to pay any cash dividends on its common shares in the foreseeable future.
The Company has never paid any cash dividends on its common shares. The Company does not anticipate paying any cash dividends on its common shares in the foreseeable future because, among other reasons, the Company currently intends to retain any future earnings to finance its business. The future payment of cash dividends will be dependent on factors such as cash on hand and achieving profitability, the financial requirements to fund growth, the Company’s general financial condition and other factors the board of directors of the Company may consider appropriate in the circumstances. Until the Company pays cash dividends, which it may never do, its shareholders will not be able to receive a return on their common shares unless they sell them.
There can be no assurance that an active market for the Company’s securities will be sustained.
There can be no assurance that an active market for Neptune’s securities will be sustained. Holders of securities of the Company may be unable to sell their investments on satisfactory terms. As a result of any risk factor discussed herein, the market price of the securities of the Company at any given point in time may not accurately reflect the long-term value of the Company. Furthermore, responding to these risk factors could result in substantial costs and divert management’s attention and resources. Substantial and potentially permanent declines in the value of the securities may result.
Other factors unrelated to the performance of the Company that may have an effect on the price and liquidity of its securities include: extent of analytical coverage; lessening in trading volume and general market interest in the securities; the size of the Company’s public float; and any event resulting in a delisting of securities.
The Company’s shareholder rights plan and certain Canadian laws could delay or deter a change of control.
The Company’s shareholder rights plan entitles a rights holder, other than a person or group holding 20% or more of our common shares, to subscribe for our common shares at a discount of 50% to the market price at that time, subject to certain exceptions. See “Description Of The Share Capital — Shareholder Rights Plan”.
TheInvestment Canada Act(Canada) subjects an acquisition of control of a company by a non-Canadian to government review if the value of the assets as calculated pursuant to the legislation exceeds a threshold amount. A reviewable acquisition may not proceed unless the relevant minister is satisfied that the investment is likely to be a net benefit to Canada.
Any of the foregoing could prevent or delay a change of control and may deprive or limit strategic opportunities for our shareholders to sell their shares.
The Company may pursue opportunities or transactions that may adversely affect its business and financial condition.
Management of Neptune, in the ordinary course of Neptune’s business, regularly explores potential strategic opportunities and transactions. These opportunities and transactions may include strategic joint venture relationships, significant debt or equity investments in Neptune by third parties, the acquisition or disposition of material assets, the licensing, acquisition or disposition of material intellectual property, the development of new product lines or new applications for its existing products, significant
35
distribution arrangements, the sale of all of the shares of Neptune and other similar opportunities and transactions. The public announcement of any of these or similar strategic opportunities or transactions might have a significant effect on the price of the securities of the Company. Neptune’s policy is to not publicly disclose the pursuit of a potential strategic opportunity or transaction unless it is required to do so by applicable law, including applicable securities laws relating to continuous disclosure obligations. There can be no assurance that investors who buy or sell securities of Neptune are doing so at a time when Neptune is not pursuing a particular strategic opportunity or transaction that, when announced, would have a significant effect on the price of the securities of the Company.
In addition, any such future corporate development may be accompanied by certain risks, including exposure to unknown liabilities of the strategic opportunities and transactions, higher than anticipated transaction costs and expenses, the difficulty and expense of integrating operations and personnel of any acquired companies, disruption of the Company’s ongoing business, diversion of management’s time and attention, and possible dilution to shareholders. The Company may not be able to successfully overcome these risks and other problems associated with any future acquisitions and this may adversely affect the Company’s business and financial condition.
Risks Related to the Company’s Status as a Foreign Private Issuer
As a foreign private issuer, the Company is subject to different U.S. Securities laws and regulations than a domestic U.S. issuer, which may limit the information publicly available to the Company’s U.S. shareholders.
The Company is a foreign private issuer under applicable U.S. federal securities laws, and therefore, it is not required to comply with all the periodic disclosure and current reporting requirements of the U.S. Exchange Act. As a result, the Company does not file the same reports that a U.S. domestic issuer would file with the SEC, although the Company is required to file with or furnish to the SEC the continuous disclosure documents that it is required to file in Canada under Canadian securities laws. In addition, the Company’s officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions of Section 16 of the U.S. Exchange Act. Therefore, the Company’s shareholders may not know on as timely a basis when the Company’s officers, directors and principal shareholders purchase or sell common shares as the reporting periods under the corresponding Canadian insider reporting requirements are longer. In addition, as a foreign private issuer, the Company is exempt from the proxy rules under the U.S. Exchange Act.
The Company may lose its foreign private issuer status in the future, which could result in significant additional costs and expenses to the Company.
In order to maintain its current status as a foreign private issuer, a majority of the Company’s common shares must be either directly or indirectly owned by non-residents of the United States unless the Company also satisfies one of the additional requirements necessary to preserve this status. The Company may in the future lose its foreign private issuer status if a majority of the Company’s common shares are held in the United States and it fails to meet the additional requirements necessary to avoid loss of foreign private issuer status. The regulatory and compliance costs to the Company under U.S. federal securities laws as a U.S. domestic issuer may be significantly more than the costs it incurs as a Canadian foreign private issuer eligible to use MJDS. If the Company is not a foreign private issuer, it would not be eligible to use the MJDS or other foreign issuer forms and would be required to file periodic and current reports and registration statements on U.S. domestic issuer forms with the SEC, which are more detailed and extensive than the forms available to a foreign private issuer. In addition, the Company may lose the ability to rely upon exemptions from NASDAQ corporate governance requirements that are available to foreign private issuers.
U.S. investors may be unable to enforce certain judgments.
Neptune is a company existing under theBusiness Corporations Act (Québec). A number of the Company’s directors and officers are residents of Canada or other jurisdictions outside of the United States, and a significant portion of the Company’s assets are located outside the United States. As a result, it may be difficult to effect service within the United States upon the Company or upon its directors and officers. Execution by United States courts of any judgment obtained against the Company or any of the Company’s directors or officers in United States courts may be limited to the assets of such companies or such persons, as the case may be, located in the United States. It may also be difficult for holders of securities who reside in the United States to realize in the United States upon judgments of courts of the United States predicated upon civil liability and the civil liability of the Company’s
36
directors and executive officers under the United States federal securities laws. The Company has been advised that a judgment of a U.S. court predicated solely upon civil liability under U.S. federal securities laws or the securities or “blue sky” laws of any state within the United States, would likely be enforceable in Canada if the United States court in which the judgment was obtained has a basis for jurisdiction in the matter that would be recognized by a Canadian court for the same purposes. However, there may be doubt as to the enforceability in Canada against these non-U.S. entities or their controlling persons, directors and officers who are not residents of the United States, in original actions or in actions for enforcement of judgments of courts of the United States, of liabilities predicated solely upon U.S. federal or state securities laws.
DIVIDENDS
The Company does not anticipate paying any cash dividend on its common shares in the foreseeable future. We presently intend to retain future earnings to finance the expansion and growth of our business. Any future determination to pay dividends will be at the discretion of our Board of Directors and will depend on our financial condition, results of operations, capital requirements and other factors the Board of Directors deems relevant. In addition, the terms of any future debt or credit facility may preclude the Company from paying dividends.
On September 5, 2012, a prospectus qualifying the distribution of 2,000,000 Class A subordinate voting shares and 4,000,000 Series 2011-1 warrants of NeuroBio held by Neptune by way of a dividend-in-kind was filed with Canadian securities regulatory authorities. Following the payment of the dividend, Neptune will control approximately 96% of the voting rights attached to the securities of NeuroBio in the aggregate and its holding of Class A subordinate voting shares of NeuroBio will be reduced to 6,500,990 Class A subordinate voting shares, representing approximately 76% of the Class A subordinate voting shares issued and outstanding. Neptune’s holding of Series 2011-1 warrants will also be reduced to 1,940,000 Series 2011-1 warrants following the distribution of the dividend, representing approximately 32.33% of the Series 2011-1 warrants issued and outstanding. See “General Development of the Business — Recent Developments”.
Besides the dividend declaration mentioned in the preceding paragraphs, the Company does not anticipate paying any dividend on its common shares in the foreseeable future. We presently intend to retain future earnings to finance the expansion and growth of our business. Any future determination to pay dividends will be at the discretion of our Board of Directors and will depend on our financial condition, results of operations, capital requirements and other factors the Board of Directors deems relevant. In addition, the terms of any future debt or credit facility may preclude the Company from paying dividends.
DESCRIPTION OF THE SHARE CAPITAL
The authorized share capital of the Company is comprised of an unlimited number of common shares, or Common Shares, and an unlimited number of preferred shares, or Preferred Shares, issuable in one or more series. By way of by-law, in accordance with its articles of incorporation, the Company created the “Series A Preferred Shares”, which are non-voting shares.
As at September 10, 2012, there were a total of (i) 50,161,061 Common Shares and no Preferred Shares issued and outstanding, (ii) 1,278,125 warrants to purchase Common Shares issued and outstanding, and (iii) 6,492,500 options to purchase Common Shares issued and outstanding.
Common Shares
Voting Rights
Each Common Share entitles its holder to receive notice of, and to attend and vote at, all annual or special meetings of the shareholders of the Company. Each Common Share entitles its holder to one vote at any meeting of the shareholders, other than meetings at which only the holders of a particular class or series of shares are entitled to vote due to statutory provisions or the specific attributes of this class or series.
37
Dividends
Subject to the prior rights of the holders of Preferred Shares ranking before the Common Shares as to dividends, the holders of Common Shares are entitled to receive dividends as declared by the board of directors of the Company from the Company’s funds that are duly available for the payment of dividends.
Winding-up and Dissolution
In the event of the Company’s voluntary or involuntary winding-up or dissolution, or any other distribution of the Company’s assets among its shareholders for the purposes of winding up its affairs, the holders of Common Shares shall be entitled to receive, after payment by the Company to the holders of Preferred Shares ranking prior to Common Shares regarding the distribution of the Company’s assets in the case of winding-up or dissolution, share for share, the remainder of the property of the Company, with neither preference nor distinction.
Preferred Shares
The Preferred Shares carry no voting rights. Preferred Shares may be issued at any time, in one or more series. The Company’s board of directors has the power to set the number of Preferred Shares and the consideration per share, as well as to determine the provisions attaching to each series of Preferred Shares (including dividends, redemption rights and conversion rights, where applicable). The shares in each series of Preferred Shares rank prior to the Common Shares of the Company with regard to payment of dividends, reimbursement of capital and division of assets in the event of the Company’s winding-up or dissolution. The holders of Preferred Shares shall not be entitled to receive notice of, or to attend or vote at the meetings of the shareholders, except: (i) in the event of a separate meeting or vote by class or by series as specified by law, (ii) where entitled to vote by class or series on amendments to the attributes attaching to the class or series, or (iii) where applicable, in the event of the Company’s omission to pay the number of periodical dividends, whether consecutive or not, as applicable to any series.
The board of directors of the Company has passed a by-law creating the Series A Preferred Shares. Series A Preferred Shares may be issued only as part of an acquisition by the Company of other companies or material assets. Series A Preferred Shares are non-voting, and entitle holders thereof to a fixed, preferential and non-cumulative annual dividend of 5% of the amount paid for the said shares.
Shareholder Rights Plan
On May 26, 2010, we entered into a shareholder rights plan agreement, or “Rights Plan”. The Rights Plan entitles a holder of rights (other than the Acquiring Person, as defined below, or any affiliate or associate of an Acquiring Person or any person acting jointly or in concert with an Acquiring Person or any affiliate or associate of an Acquiring Person) to purchase our Common Shares at a discount of 50% to the market price upon a person becoming an “Acquiring Person”, subject to certain exceptions and the terms and conditions set out in the Rights Plan. An “Acquiring Person” is defined in the Rights Plan as a beneficial owner of 20% or more of our Common Shares. The Rights Plan will expire at the close of our annual meeting of shareholders in 2013.
In order to implement the Rights Plan, we issued one right in respect of each Common Share outstanding as of 5:01 p.m. (Montreal time) on May 26, 2010, the “Effective Date”. One right will also be issued and attached to each subsequently issued Common Share. The rights will separate and trade separately from the Common Shares to which they are attached and will become exercisable after the “Separation Time”. The “Separation Time” is the close of business on the tenth business day following the earliest of:
| (a) | the date of the first public announcement or disclosure made by us or an Acquiring Person that a person has become an Acquiring Person; |
| (b) | the date of the commencement of, or first public announcement of the intent of any person to commence, a take-over bid (other than a Permitted Bid (as defined in the Rights Plan) or a Competing Permitted Bid (as defined in the Rights Plan) by any person for our Common Shares; |
| (c) | the date upon which a Permitted Bid or Competing Permitted Bid ceases to be such; or |
| (d) | such later date as may be determined by the board of directors. |
38
After the time at which a person becomes an Acquiring Person, and subject to the terms and conditions set out in the Rights Plan, each right would, upon exercise, entitle a rights holder, other than the Acquiring Person and related persons, to purchase Common Shares at a 50% discount to the market price at the time.
Under the Rights Plan, a “Permitted Bid” is a bid made to all holders of the Common Shares and which is open for acceptance for not less than 60 days. If at the end of 60 days at least 50% of the outstanding Common Shares, other than those owned by the offeror and certain related parties, have been tendered, the offeror may take up and pay for the Common Shares but must extend the bid for a further 10 days to allow other shareholders to tender.
A copy of the Rights Plan is available on SEDAR atwww.sedar.com.
MARKET FOR SECURITIES
On March 31, 2011, Acasti completed its listing application on the TSX-Venture Exchange. As a result Acasti had its shares listed on the TSX-Venture Exchange on March 31, 2011 under the symbol APO. The authorized share capital of Acasti is composed of an unlimited number of Class “A”, “B”, “C”, “D” and “E” shares. Each holder of Class “A” and Class “B” shares of Acasti has the right to vote at any meeting of the shareholders of Acasti.
As at May 15, 2012, there were 72,680,638 issued and outstanding Class A shares of Acasti, each share entitling its holder to one (1) vote per Class “A” share.
On November 28, 2011, the Company migrated from the TSX-Venture Exchange to the Toronto Stock Exchange, or TSX. The Company’s Common Shares are listed and posted for trading on (i) the Toronto Stock Exchange, or TSX, under the symbol “NTB”, and (ii) the Nasdaq Stock Market, or NASDAQ, under the symbol “NEPT”.
Trading Prices and Volumes for Neptune
The price ranges and trading volume of Company’s Common Shares for the most recently completed financial year on the TSX and the NASDAQ was as follows:
Period | TSX (CDN$) | NASDAQ (US$) | ||||||||||||||||||||||
| High | Low | Volume (daily average) | High | Low | Volume (daily average) | |||||||||||||||||||
February 2012 | 3.25 | 2.44 | 52,924 | 3.29 | 2.46 | 84,480 | ||||||||||||||||||
January 2012 | 2.89 | 2.29 | 32,937 | 2.86 | 2.25 | 54,102 | ||||||||||||||||||
December 2011 | 3.10 | 2.25 | 22,345 | 3.05 | 2.18 | 44,001 | ||||||||||||||||||
November 2011 | 3.17 | 2.55 | 27,824 | 3.10 | 2.51 | 47,242 | ||||||||||||||||||
October 2011 | 2.84 | 2.10 | 50,361 | 2.86 | 2.02 | 115,447 | ||||||||||||||||||
September 2011 | 3.74 | 2.60 | 71,901 | 3.78 | 2.46 | 102,372 | ||||||||||||||||||
August 2011 | 4.46 | 3.00 | 143,670 | 4.66 | 3.06 | 135,202 | ||||||||||||||||||
July 2011 | 4.25 | 3.55 | 91,078 | 4.62 | 3.65 | 137,578 | ||||||||||||||||||
June 2011 | 3.85 | 2.95 | 82,590 | 3.99 | 3.02 | 129,895 | ||||||||||||||||||
May 2011 | 4.13 | 2.70 | 110,970 | 4.05 | 2.61 | 101,250 | ||||||||||||||||||
April 2011 | 3.77 | 2.05 | 178,004 | 3.95 | 2.11 | 166,363 | ||||||||||||||||||
March 2011 | 2.46 | 2.07 | 36,534 | 2.55 | 2.08 | 27,216 | ||||||||||||||||||
DIRECTORS AND OFFICERS
Name, Occupation and Security Holding of Directors
The following table sets forth each director and executive officer’s name, province and country of residence, his/her principal occupation, including the committees of the Board, the year in which he or she first became a director, as at the date of this annual information form. All members of the Board of Directors herein below will hold their positions until the next annual meeting of shareholders of the Company.
39
Name and Province and Country of | Principal Occupation | Position Within the Company | Year of Nomination as a Director of the Company | |||||
Henri Harland(3,4,5) Québec, Canada | President and Chief Executive Officer of the Company | Director, President and Chief Executive Officer of the Company | 1998 | |||||
Ronald Denis(1,2,3,4) Québec, Canada | Chief of Surgery at Hôpital du Sacré-Coeur, Montréal | Director and Chairman of the Board of the Company | 2000 | |||||
Daniel Perry(1,2,3,9) France | President/Manager Société ADG 7 Tours | Director of the Company | 2000 | |||||
Michel Chartrand(3,4) Québec, Canada | Chief Operating Officer of the Company | Director of the Company | 2006 | |||||
Jean-Claude Debard(1,2,3,9) France | President of M Motors Automobiles France | Director of the Company | 2009 | |||||
Martin Godbout(8) Québec, Canada | President, Hodran Consultants | Director of Acasti | 2011 | |||||
Marc Lebel(8) Québec, Canada | Interim CEO and Director of Warnex Inc. | Director of Acasti | 2011 | |||||
Harlan W. Waksal(3) New York, United States | Vice-President, Business and Scientific Affairs at Acasti | Director | 2012 | |||||
Tina Sampalis, M.D., Ph.D.(5) Québec, Canada | Chief Global Strategy Officer of the Company | Chief Global Strategy Officer of the Company | — | |||||
André Godin(5,6) Québec, Canada | Vice-President, Administration and Finance of the Company | Vice-President, Administration and Finance of the Company | — | |||||
Pierre Lemieux(5,7) Québec, Canada | Chief Operating Officer of Acasti | Chief Operating Officer of Acasti | — | |||||
Xavier Harland(5,7) Québec, Canada | Chief Financial Officer of Acasti | Chief Financial Officer of Acasti | — | |||||
| (1) | Member of the Audit Committee of the Company |
| (2) | Member of the Compensation Committee of the Company |
| (3) | Member of the Corporate Governance Committee of the Company |
| (4) | Director of Acasti and NeuroBio |
| (5) | Officer of Acasti and NeuroBio |
| (6) | Chief Financial Officer of Acasti until March 20, 2011 |
| (7) | Chief Financial Officer of Acasti since March 21, 2011 |
| (8) | Director of Acasti only |
| (9) | Director of NeuroBio |
As of February 29, 2012, the directors and executive officers of the Company, as a group, beneficially owned or exercised control or direction over approximately 4,306,500 (8.6%) of the outstanding Common Shares of Neptune.
In Neptune’s and Acasti’s Management Proxy Circular dated respectively May 29 and May 15, 2012, all of the above listed directors were nominated by management for election and/or re-election.
Following are brief biographies of Neptune’s directors and executive officers:
Mr. Henri Harland – Director, Chief Executive Officer and President
Mr. Henri Harland is an Actuary and holds a MBA (Finance) from Laval University. Mr. Henri Harland has been a director and the President and Chief Executive Officer of the Company since its incorporation on October 9, 1998. He is the Founder of the Company and has been involved in the krill research project since 1991. For more than ten years he has also held the position of President and Chief Executive Officer of Groupe Conseil Harland Inc., a financial engineering group. Previously, he acted has an independent financial consultant guiding companies from different industrial sectors in both North America and Europe in their capital restructure, financing and business development.
40
Dr. Ronald Denis – Chairman of the Board and Director
Dr. Ronald Denis is currently Chief of Surgery and Co-Director of the Trauma Program at Hôpital du Sacré-Coeur in Montréal. Also, since 1987, Dr. Denis has been medical co-director of the Canadian Formula 1 Grand Prix. Dr. Denis sits on several scientific boards and management committees.
Mr. Michel Chartrand – Chief Operating Officer and Director
On September 12, 2011, Mr. Michel Chartrand joined the Company has its Chief Operating Officer. Before joining the Company, he was the Vice-President of Retail Partner Solutions at McKesson Canada between 2009 and 2011. From 2004 to 2009 Mr. Chartrand was the President and Chief Executive Officer of Groupe PharmEssor inc. which included, due to a merger, Gestion Santé Services Obonsoins Inc. and Groupe Essaim Inc., two important Quebec pharmacy franchisors in Quebec. From 1998 to 2004, Mr. Chartrand was the Executive Vice President of Gestion Santé Services Obonsoins Inc.
Mr. Jean-Claude Debard – Director
Mr. Jean-Claude Debard has been President of M Motors Automobiles France, Subaru France, Daihatsu France, SsangYong France since 2012 and Executive President of Group Emil Frey France since 2008.
Mr. Daniel Perry – Director
Since March 1993, Mr. Daniel Perry has been General Manager of companies operating recreational/tourism complexes in France. Mr. Perry is also a specialist and consultant for different corporations involved in the marketing of new products in Europe.
Dr. Harlan W. Waksal – Director
Dr. Harlan W. Waksal is a retired physician. Dr. Waksal is the Vice-President, Business and Scientific Affairs at Acasti Pharma Inc. He received his B.A. from Oberlin College and M.D. from Tufts University School of Medicine, and his post graduate training in Internal Medicine and in Pathology. In addition, he did research in immunology at the Weizmann Institute of Science. Dr. Waksal was a founder of Imclone Systems Incorporated; a New York based pharmaceutical Company specializing in developing new treatment for various forms of cancer. He served as the Chief Operating Officer and member of the Board of Directors from 1986 until 2001 and as President/CEO from 2001 until 2002. During his tenure, he was responsible for building the scientific and operation infrastructure of the Company. Dr. Waksal is the author of over 50 scientific publications and has been the author of multiple patents and patent applications. His current activities are focused on managing various real estate developments and serving on select Board of Directors. Dr. Waksal currently serves on the Boards of the Oberlin College, Senesco Technologies, Inc. He also serves on the Advisory Board of Northern Rivers Funds.
Dr. Tina Sampalis M.D., Ph.D. – Chief Scientific Officer
Dr. Tina Sampalis is an Oncology Surgeon, trained in Physiology at McGill University, Medicine at the University of Patras (Greece), Dermatology at Göttingen University (Germany) and Marselisborg University (Denmark), Pediatric, General and Oncology Surgery at the University of Athens (Greece), graduate training (PhD) in Surgical Research at the University of Athens and a second PhD in Epidemiology and Experimental Surgery at McGill University. She has received several international scholarships and awards for her work on the clinical implementation of retinols skin and breast cancer and for her work on scintimammography. U.S. and Canadian patent applications have been filed for the development and implementation of innovative micro-invasive and stereotactic robotic surgical techniques for breast cancer. Between May 2000 and June 2007, she has held the position of Vice-President of Research and Business Development and since June 2007 the position of Chief Scientific Officer of the Company. She has ceased to occupy this position following her nomination as Chief Global Strategy Officer of the Company, which was announced on May 25, 2012.
41
Mr. André Godin – Chief Financial Officer
Mr. André Godin, C.A., has a Bachelor in Administration and has been a Member of the Canadian Institute of Chartered Accountants since 1988. He has more than 10 years experience in the Biotech/Pharma industry as former President of a Dietary Supplement Company and as a Corporate Controller for a pharmaceutical Company in OTC products. Mr. Godin has been Vice-President, Administration and Finance for Neptune since 2003.
Dr. Pierre Lemieux Ph.D. – Chief Operating Officer (Acasti)
Mr. Pierre Lemieux holds a post-doctoral degree in Oncology from the Health Science Center, University of Texas, USA, and a PhD in biochemistry from Laval University, Canada, jointly with University of Nottingham, England. Mr. Lemieux joined Neptune’s subsidiary Acasti as the Chief Operating Officer on April 12, 2010. Prior to joining Acasti, Mr. Lemieux was the President, CEO and the Chairman of the Board as well as being the founder of Technologie Biolactis Inc., a late-stage biotechnology Company specialized in the valorization of proteins to better serve the nutraceutical, the cosmetical and pharmaceutical industries.
Mr. Xavier Harland – Chief Financial Officer (Acasti)
Mr. Xavier Harland recently joined Acasti as Chief Financial Officer. He graduated from Laval University in Actuarial Science in 2003. He is also a CFA charter holder since 2007 and FRM holder since 2006. Xavier Harland has been working as Director of Finance for Neptune since 2004. Mr. Harland works full time for the Neptune group, which includes Acasti and NeuroBio.
Mr. Marc Lebel – Director (Acasti)
Mr. Marc LeBel is the co-founder of Anapharm, a Phase I contract research organization, that reached 1,200 employees. Mr. LeBel was Executive Vice-President of Pharmanet, from 2005 to 2007, following its acquisition of Anapharm. Mr. LeBel is currently Interim CEO and Director of Warnex Inc., Director of Acasti Pharma Inc. and Nuchem Inc. His recent venture in the film industry made him Executive Producer “Ruby McCollum”, and Associate Producer of the 3D animation movie “Sarila”. He received the following honors: Excelsia 2006 BioQuébec, Grand Diplômé Université Laval, and Leadership Prize, Canadian Society Pharmaceutical Sciences.
Dr. Martin Godbout – Director (Acasti)
Mr. Martin Godbout holds a B.Sc. in Biochemistry (1979) and a doctorate in physiology and molecular endocrinology from Laval University. From 1985 to 1990, he received a postdoctoral fellowship from the Medical Research Council of Canada (MRC) and went to San Diego, California, where he continued research work in molecular neurobiology at the Scripps Research Institute. From May 1994 to May 1997, he was President and CEO of Innovatech Quebec, a technology investment fund of 60 million dollars. In May 1997, he became Vice-President of BioCapital, a Canadian venture fund specialized in private financing of start-up companies demonstrating strong potential in the areas of health and biotechnology. From 200 to 2009 he was President of Genome Canada. Mr. Godbout is an Officer of the Order of Canada (2005). Since 2004, Mr. Godbout is a director of MethylGene, a public Company listed on the Toronto Stock Exchange. Mr. Godbout is currently a director on several boards of high technology companies, foundations and scientific organizations such as AmorChem, AngioChem, Asmacure, BioContact Québec, Génome Québec, BioQuébec, Montréal In Vivo, Fonds de Recherche Québec-Santé and the Ataxia Charlevoix Foundation.
CEASE TRADE ORDERS, BANKRUPTCIES, PENALTIES OR SANCTIONS
To the knowledge of Neptune, none of the directors or executive officers of the Company:
| (a) | is, or has been, within the last ten years, a director, chief executive officer or chief financial officer of any Company that: |
42
| (i) | was subject to a cease trade order, an order similar to a cease trade order, or an order that denied the relevant Company access to any exemption under applicable securities legislation, that was in effect for a period of more than 30 consecutive days (an “Order”), which Order was issued while the director or executive officer was acting in the capacity as director, chief executive officer or chief financial officer; or |
| (ii) | was subject to an Order that was issued after the director or executive officer ceased to be a director, chief executive officer or chief financial officer and which resulted from an event that occurred while that person was acting in the capacity as director, chief executive officer or chief financial officer; |
To the knowledge of Neptune, no director or executive officer of the Company, or shareholder holding a sufficient number of securities of the Company to affect materially the control of the Company:
| (a) | is, or has been, within the last ten years, a director or executive officer of any Company that, while that person was acting in that capacity, or within a year of that person ceasing to act in that capacity, became bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency or was subject to or instituted any proceedings, arrangement or compromise with creditors or had a receiver manager or trustee appointed to hold its assets; or |
| (b) | has, within the last ten years, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency, or become subject to or instituted any proceedings, arrangement or compromise with creditors, or had a receiver, receiver manager or trustee appointed to hold his or its assets of the proposed director. |
To the knowledge of Neptune, no director, executive officer or shareholder holding a sufficient number of securities of the Company to affect materially the control of the Company has been subject to:
| (a) | any penalties or sanctions imposed by a court relating to securities legislation or by a securities regulatory authority or has entered into a settlement agreement with a securities regulatory authority; or |
| (b) | any other penalties or sanctions imposed by a court or regulatory body that would likely be considered important to a reasonable security holder in deciding whether to vote for a proposed director. |
LEGAL PROCEEDINGS AND REGULATORY ACTIONS
The Company is not aware of any legal proceedings or regulatory actions in which it is involved and no such proceedings or regulatory actions are known by the Company to be contemplated, except in regards of what is mentioned in the section “Business of the Company — Economic Dependence/Litigation”.
INTEREST OF MANAGEMENT AND OTHERS IN MATERIAL TRANSACTIONS
Except for what is stated below, none of the insiders of the Company, the Directors, or any of their respective associates or affiliates, has or has had any material interest, direct or indirect, in any material transaction whether proposed or concluded, since the beginning of the Company’s most recently completed financial year and for the three (3) last completed financial years.
The Company entered into an agreement with a Company controlled by Mr. Henri Harland, as of February 23, 2001, calling for royalties to be paid in semi-annual instalments equal to 1% of the Company’s annual revenues, for an unlimited period. Each year disbursement of royalties cannot exceed net annual earnings before interest, taxes and amortization. See “Business of the Company —Intellectual Property — Licensing Arrangements”.
TRANSFER AGENTS AND REGISTRARS
Computershare Trust Company of Canada, at its offices in Montreal, is the transfer agent and registrar for our Common Shares.
43
MATERIAL CONTRACTS
The Company has not entered into any material contract, other than those entered into in the normal course of business, within the most recently completed financial year, or before the most recently completed financial year, which is still in effect except for Technology License Agreement with Acasti Pharma Inc. on August 7, 2008 and Technology License Agreement with NeuroBioPharm Inc. on October 15, 2008. See “Business of the Company — Intellectual Property”.
INTEREST OF EXPERTS
KPMG LLP, Chartered Professional Accountants (“KPMG”), has audited our consolidated financial statements for the years ended as at February 29, 2012 and February 28, 2011. KPMG are independent with respect to Neptune Technologies & Bioressources Inc. and Acasti Pharma Inc. within the meaning of the Rules of Professional Conduct/Code of Ethics of the Quebec Order of Chartered Professional Accountants.
REPORT ON AUDIT COMMITTEE
Audit Committee’s Charter
The Charter of the Audit Committee is annexed to this circular as Schedule A. The Charter was adopted by the Board of Directors on June 6, 2007.
Composition of the Audit Committee
The Audit Committee is composed of 3 members of the Board of Directors. Dr. Ronald Denis, Mr. Daniel Perry, and Mr. Jean-Claude Debard are the directors that seat on the Audit Committee. From the experience set forth below, the Company believes that these persons have sufficient knowledge and background to actively participate on the Audit Committee. Mr. Michel Chartrand ceased to be a member of the Audit Committee on September 12, 2011, following his nomination as the Company’s Chief Operating Officer.
Under Multilateral Instrument 52-110Audit Committees (“MI 52-110”), a director of an Audit Committee is “independent” if he or she has no direct or indirect material relationship with the issuer, that is, a relationship which could, in the view of the Board of Directors, reasonably interfere with the exercise of the member’s independent judgment.
The following describes the relevant education and experience of each member of the Audit Committee of the Company that provides him or her with (a) an understanding of the accounting principles used by the Company to prepare its financial statements, (b) the ability to assess the general application of such accounting principles, (c) experience preparing, auditing, analyzing or evaluating financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to those that can reasonably be expected to be raised by the Company’s financial statements or experience actively supervising one or more persons engaged in such activities and (d) an understanding of internal controls and procedures for financial reporting.
Dr. Ronald Denis– Dr. Denis has been Chief of Surgery and Director of the Trauma Program at Hôpital Sacré-Coeur since 1997. In his duties, Mr. Denis has to manage Sacré-Coeur Hospital Trauma Program budget and staff, also he has had to regularly review and analyze financial statements. Dr. Denis’ experience required and contributed to the development of his ability to analyze financial statements and understand IFRS.
Jean-Claude Debard – Mr. Debard has been President of Hyundai Automobile France and FEA Services as well as an officer of Frey Accessories and Parts since 1999 and most recently Executive President of Group Emil Frey France since 2008. Since 1999, Mr. Debard has served on the Oversight Committee of Holding (SERGESA), SsangYong France and Hyundai Finances. Mr. Debard also has a graduate degree in Management and Strategic Management. Mr. Debard’s experience required and contributed to the development of his ability to analyze financial statements and understand IFRS.
Daniel Perry– Since March 1993, Mr. Perry is General Manager of a corporation operating a recreational/tourism complex in France. Also, Mr. Perry is a specialist and consultant in the marketing of new products on the European continent. Mr. Perry’s experience required and contributed to the development of his ability to analyze financial statements and understand IFRS.
44
External Auditor Fees
Audit Fees
“Audit fees” consist of fees for professional services for the audit of the Company’s annual financial statements. For the fiscal year ended February 29, 2012, KPMG LLP, the Company’s external auditors, billed $267,000 to the Company, respectively $195,000 for Neptune, $40,000 for Acasti and $32,000 for NeuroBio, for audit fees. For the fiscal year ended February 28, 2011, these fees were $290,225 to the Company, respectively $188,225 for Neptune and $102,000 for Acasti.
Audit-Related Fees
“Audit-related fees” consist of fees for professional services that are reasonably related to the performance of the audit or review of the Company’s financial statements and which are not reported under “Audit Fees” above. For the fiscal year ended February 29, 2012, KPMG LLP, the Company’s external auditors, billed $170,750 to the Company, respectively $82,500 for Neptune (US registration statement, IFRS consultations and translation of documents), $30,750 for Acasti (IFRS consultations, translation) and $57,500 for NeuroBio (preliminary prospectus and interim reviews). The Company’s auditors billed no fees as to this matter for fiscal year ended February 28, 2011.
Tax Fees
“Tax fees” consist of fees for professional services for tax compliance, tax advice and tax planning. KPMG LLP, the Company’s external auditors, billed a total of $56,000 to the Company, respectively $47,500 for Neptune, $7,000 for Acasti and 1,500 for NeuroBio, for tax fees for fiscal year ended February 29, 2012 and a total of $63,814 to the Company, respectively $48,151 for Neptune and $15,663 for Acasti the fiscal period ended February 28, 2011. Tax fees include, but are not limited to, preparation of tax returns.
All Other Fees
The “other fees” include all other fees billed for professional services other than those mentioned hereinabove. KPMG LLP, chartered accountants, the Company’s external auditors, billed no fees as to this matter the fiscal years ended February 29, 2012 and February 28, 2011.
ADDITIONAL INFORMATION
Additional information relating to the Company may also be found on the SEDAR website at www.sedar.com, and on EDGAR atwww.sec.gov.
Additional information, including directors’ and officers’ remuneration and indebtedness, principal holders of our securities, options to purchase securities and interests of informed persons in material transactions, if applicable, is contained in Neptune’s Management Proxy Circular dated May 28, 2012 and available on SEDAR. Additional financial information is also provided in our financial statements and MD&A for the most recently completed financial year.
45
SCHEDULE “A”
CHARTER OF THE AUDIT COMMITTEE OF THE BOARD OF DIRECTORS
The Audit Committee of the Board of Directors assists the Board in fulfilling its oversight responsibilities relating to the quality and integrity of the accounting, auditing and reporting practices of the Company and such other duties as directed by the Board of Directors or imposed by legislative authorities or stock exchanges.
Structure and Organization
| 1. | The membership of the Committee will consist of at least three independent members of the Board of Directors, the majority of whom will not be employees, controlling shareholders or executives of the Company or of any associates or affiliates of the Company. Committee members and the Committee Chairman shall be designated by and serve at the pleasure of the Board of Directors. All members must be financially literate and at least one member must have accounting or related financial management expertise, in each case in the judgment of the Board of Directors. |
| 2. | The Committee shall meet at least four times per year or more frequently as circumstances require. The Committee may ask members of management or others to attend meetings and provide pertinent information as necessary. The required quorum for the Committee will be the majority of the members forming the Committee. |
| 3. | The Committee is expected to maintain free and open communication with management and the external auditors. |
| 4. | The Committee has the authority to investigate any matter brought to its attention and to retain outside counsel for this purpose if, in its judgment, that is appropriate. |
General Responsibilities
The Committee shall:
| 1. | Meet periodically with representatives of the external auditors, the internal audit manager (if any) and management in separate sessions to discuss any matters that the Committee or these groups believe should be discussed privately with the Committee. Provide sufficient opportunity for the external auditors to meet with the internal auditors as appropriate without members of management being present. |
| 2. | Prepare the minutes of all Committee meetings and report of such meetings to the Board of Directors. |
| 3. | Review and reassess the adequacy of this Charter annually. |
Responsibilities for Engaging External Auditors
The Committee shall:
| 1. | Recommend for approval by the Board of Directors and ratification by the shareholders the selection and retention of an independent firm of chartered accountants as external auditors, approve compensation of the external auditors, and review and approve in advance the discharge of the external auditors. |
| 2. | Review the independence of the external auditors. In considering the independence of the external auditors, the Committee will review the nature of the services provided by the external auditors and the fees charged, and such other matters as the Committee deems appropriate. |
| 3. | Ensure that the external auditors are in good standing with the Canadian Public Accountability Board (CPAB) and that the CPAB has not imposed any sanction on them. The Audit Committee is also responsible for ensuring that the external auditors comply with the rotation requirements with respect to partners involved in the audit of the Company. |
| 4. | Arrange for the external auditors to be available to the Board of Directors at least annually to help provide a basis for the Board’s approval of the external auditors’ appointment. |
A-1
| 5. | Approve all allowable non-audit related services to be provided to the Company or one of its subsidiaries by the Company’s external auditors if applicable. |
| 6. | Non-audit services of minimal amount satisfy the pre-approval requirements on the following conditions: |
| (a) | that the aggregate amount of all non-audit services that were not pre-approved is reasonably expected to constitute no more than five per cent of the total amount of fees paid by the Company and its subsidiaries to the Company’s external auditors during the fiscal year in which the services are provided; |
| (b) | that the Company or its subsidiaries, as the case may be, did not recognize the services as non-audit services at the time of the engagement; and |
| (c) | that the services are promptly brought to the attention of the Audit Committee and approved, prior to the completion of the audit, by the Audit Committee or by one or more of its members to whom authority to grant such approvals had been delegated by the Audit Committee. |
Responsibilities for Oversight of the Quality and Integrity of Accounting, Auditing and Reporting Practices of the Company
The Committee shall:
| 1. | Directly review the work of the external auditors engaged for the purpose of preparing or issuing an auditor’s report or performing other audit, review or attestation services for the Company. The Committee shall be directly responsible of the resolution of disagreements between management and the external auditors regarding financial reporting. |
| 2. | Review the Company’s financial statements, management’s discussion and analysis (MD&A) and annual and interim earnings press releases together with management and the external auditors, if applicable, before the Company publicly discloses this information. This review should cover the quality of the financial reporting and such other matters as the Committee deems appropriate. |
| 3. | Review with the external auditors and management the audit plan of the external auditors for the current year and the following year. |
| 4. | Review with financial and accounting personnel, the adequacy and effectiveness of the accounting, financial, and computerized information systems controls of the Company, and the results of any external audit procedures, if applicable. |
| 5. | Establish procedures for the receipt, retention and treatment of complaints received regarding accounting, internal accounting controls or auditing matters. Such complaints are to be treated confidentially and anonymously. |
| 6. | Review and approve all related party transactions undertaken by the Company. |
Periodic Responsibilities
The Committee shall:
| 1. | Review periodically with management any legal and regulatory matters that may have a material impact on the Company’s financial statements, compliance policies and compliance programs. |
| 2. | Review with management and approve transactions involving management and/or members of the Board of Directors, which would require disclosure under TSX Venture Exchange rules. |
| 3. | Supervise the corporate compliance program and periodically review whether any improvements should be made thereto and make appropriate recommendations to management. |
| 4. | Perform such other functions assigned by law, the Company’s Articles or bylaws, or by the Board of Directors. |
A-2
| 5. | Review services and related fees for work done by the external auditors as well as an updated projection of the total costs for the fiscal year. |
| 6. | Review and approve the engagement policy of the Company with respect to partners, employees, former partners and employees of the current and previous external auditors of the Company. |
| 7. | Implement a process for the identification of the principal business risks and monitor the implementation of appropriate methods of risk management. This process will require consultation with management in order to determine how risks are handled and to solicit the opinion of the internal audit department with respect to the effectiveness of the risk limitation strategies. |
Authority of the Audit Committee
The Committee shall have the authority to:
| 1. | Engage independent counsel and other advisors as it determines necessary to carry out its duties. |
| 2. | Pay the compensation for any advisors employed by the Committee. The Committee shall notify the Board of Directors on the extent of the financing required to pay for the compensation of the independent expert advisors retained to advise the Committee. |
| 3. | Communicate directly with the internal and external auditors. |
A-3