
Targeted Therapeutics for Inflammatory Disease January 2018 Exhibit 99.1

Forward Looking Statements/ Safe Harbor This presentation and the accompanying oral commentary contain “forward-looking” statements that involve substantial risks and uncertainties. All statements other than statements of historical facts contained in this presentation and the accompanying oral commentary, including statements regarding our future financial condition, business strategy and plans and objectives of management for future operations, are forward looking statements. In some cases, you can identify forward-looking statements by terminology such as “believe,” “will,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “might,” “approximately,” “expect,” “predict,” “could,” “potentially” or the negative of these terms or other similar expressions. Forward looking statements appear in a number of places throughout this presentation and the accompanying oral commentary and include statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things, our ongoing and planned preclinical development and clinical trials, the timing of and our ability to make regulatory filings and obtain and maintain regulatory approvals for rosiptor (AQX-1125) and our future product candidates, our intellectual property position, the degree of clinical utility of rosiptor and our future product candidates, particularly in specific patient populations, our ability to develop commercial functions, expectations regarding clinical trial data, our results of operations, cash needs, financial condition, liquidity, prospects, growth and strategies, the industry in which we operate and the trends that may affect the industry or us. Forward-looking statements involve known and unknown risks, uncertainties, assumptions and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In evaluating these statements, you should specifically consider various factors, including the risks outlined under the caption “Risk Factors” set forth in our Quarterly Report on Form 10-Q for the quarter-end ended Sept 30, 2017, which we filed with the Securities and Exchange Commission (“SEC”) on November 8, 2017 and other reports and filings we will make with the SEC from time to time. Forward-looking statements represent our management’s beliefs and assumptions only as of the date of this presentation. Except as required by law, we assume no obligation to update these forward-looking statements publicly, or to update the reasons why actual results could differ materially from those anticipated in the forward-looking statements, even if new information becomes available in the future.

Aquinox Pharmaceuticals, Inc. Founded 2006; Founder/CEO David Main NASDAQ 2014: AQXP Aquinox is discovering and developing novel drug candidates to treat inflammation, inflammatory pain, & blood cancers: Primary focus is anti-inflammatory product candidates targeting SHIP1 As of 2017: 50+ employees and growing 2 locations: Vancouver, BC and San Bruno, CA San Bruno, CA Vancouver, BC

Aquinox Summary Well capitalized into 2019 First-in-Class drug targeting novel enzyme (SHIP1) with broad anti-inflammatory potential Lead disease indication: Rosiptor (AQX-1125) for Interstitial Cystitis / Bladder Pain Syndrome (IC/BPS) Encouraging Phase 2 results in IC/BPS; published in Journal of Urology in 2016 Competitive advantage in IC/BPS; large, underserved market, suitable for independent commercialization in the US with RoW partner Actively Enrolling Patients in Phase 3 Clinical Trial; Top-Line Data Expected Q3 2018

Rosiptor Overview: A Novel, First-in-Class Anti-Inflammatory Therapy First-in-Class SHIP1 activator with broad anti-inflammatory potential Favorable ADME profile in humans Once-daily oral administration High oral bioavailability; minimal metabolism Predominantly eliminated through renal clearance as rosiptor T1/2 = 21 hours, Tmax = 1.25 hours Dose proportional PK, no food effect Well tolerated in 8 completed clinical trials More than 395 subjects dosed to date Aquinox Analysis

IC/BPS Disease State Overview

IC/BPS Takes a Physical & Mental Toll on Patients 1. Abrams P, et al. eds. Incontinence. 3rd ed. International Continence Society; 2005; 2. Berry SH, et al. J Urol 2011; 186: 540–544; 3. Suskind AM, et al. J Urol 2013; 189: 141–145; 4. Kanter G, Volpe KA, Dunivan GC, et al. Int Urogynecol J. 2016 [Epub ahead of print]. IC/BPS is a life-altering, debilitating condition - characterized by bladder pain and urinary symptoms (prevalence ~5.5M)1-3 Diagnosis of exclusion & one of the most challenging urological conditions (no new oral therapy approved in >20 years) Fear and anxiety from the unrelenting and unpredictable nature of the disease - “a life sentence…it’s always lurking”4
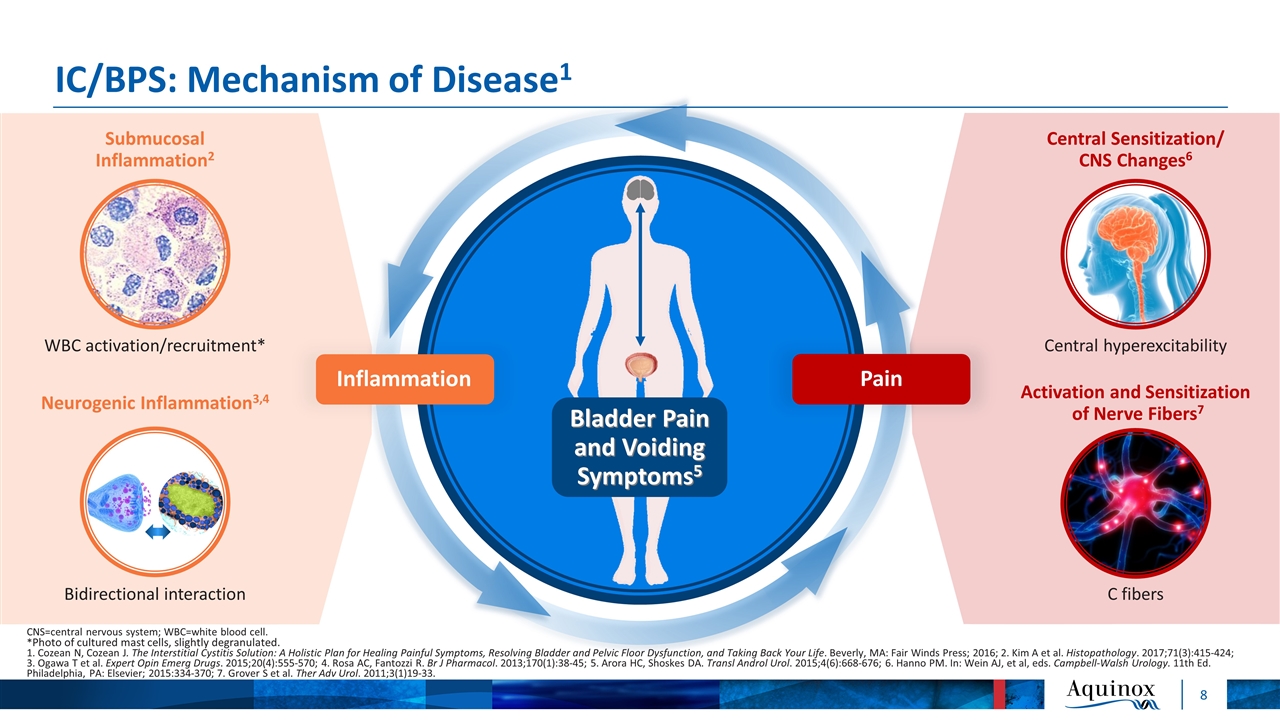
IC/BPS: Mechanism of Disease1 Pain Inflammation Bladder Pain and Voiding Symptoms5 CNS=central nervous system; WBC=white blood cell. *Photo of cultured mast cells, slightly degranulated. 1. Cozean N, Cozean J. The Interstitial Cystitis Solution: A Holistic Plan for Healing Painful Symptoms, Resolving Bladder and Pelvic Floor Dysfunction, and Taking Back Your Life. Beverly, MA: Fair Winds Press; 2016; 2. Kim A et al. Histopathology. 2017;71(3):415-424; 3. Ogawa T et al. Expert Opin Emerg Drugs. 2015;20(4):555-570; 4. Rosa AC, Fantozzi R. Br J Pharmacol. 2013;170(1):38-45; 5. Arora HC, Shoskes DA. Transl Androl Urol. 2015;4(6):668-676; 6. Hanno PM. In: Wein AJ, et al, eds. Campbell-Walsh Urology. 11th Ed. Philadelphia, PA: Elsevier; 2015:334-370; 7. Grover S et al. Ther Adv Urol. 2011;3(1)19-33. Central Sensitization/ CNS Changes6 C fibers Central hyperexcitability Activation and Sensitization of Nerve Fibers7 WBC activation/recruitment* Submucosal Inflammation2 Neurogenic Inflammation3,4 Bidirectional interaction

LEADERSHIP 201: Phase 2 Trial with Rosiptor in IC/BPS

Phase 2 LEADERSHIP Trial: A Comprehensive IC/BPS Trial Population and entry demographics: 69 female patients across US and Canadian sites with moderate to severe IC/BPS symptoms: Mean bladder pain > 5/10 Mean BPIC-SS and O’Leary Sant ICSI/PI symptom scores ≥ 19 and ≥ 8 respectively On background medication (excluding opiates) Primary endpoint: Reduction of average daily bladder pain at 6 weeks with once daily AQX-1125 vs. placebo Pre-specified secondary endpoints: Maximum daily bladder pain based on an 11-point NRS recorded by eDiary Average and maximum daily bladder pain score measured by 11-point NRS recorded at clinic Multiple urological and QoL symptom assessments (O’Leary-Sant ICSI/PI, BPIC-SS) Voiding frequency over a 24-hour period Safety, pharmacokinetics Nickel JC, et al. J Urol. 2016;196(3):747-754
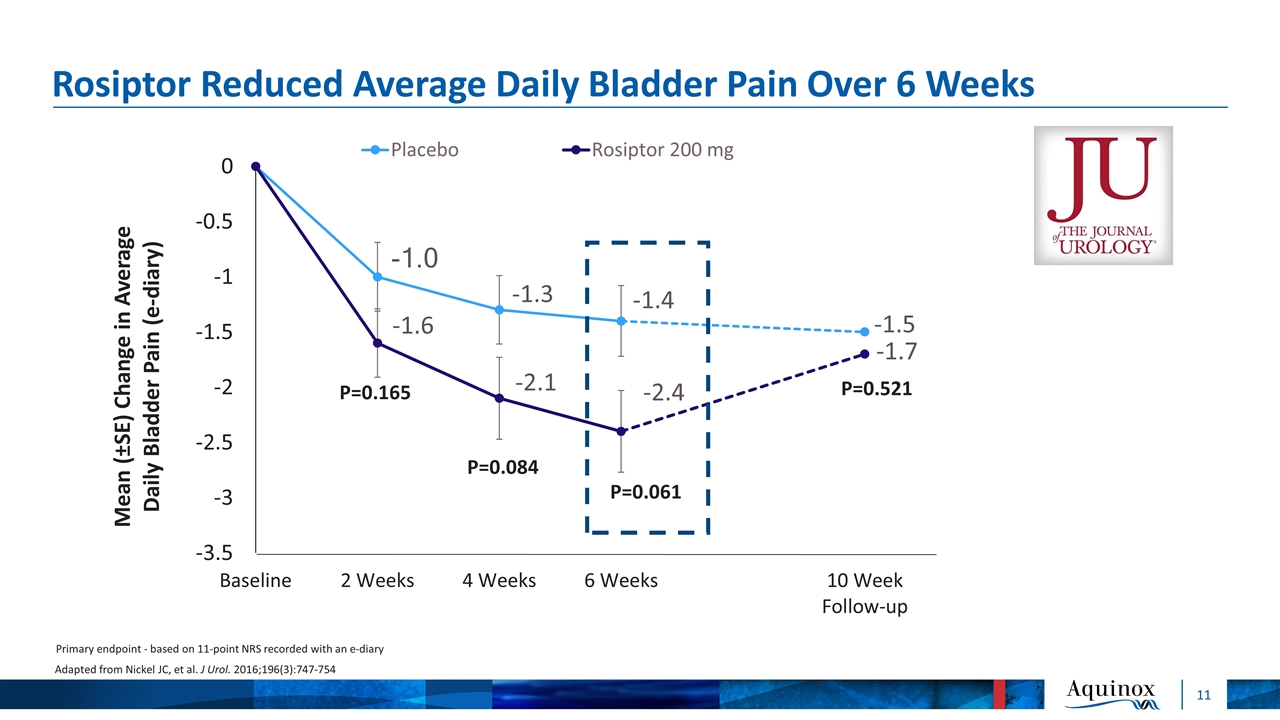
Rosiptor Reduced Average Daily Bladder Pain Over 6 Weeks P=0.165 P=0.084 P=0.061 Primary endpoint - based on 11-point NRS recorded with an e-diary Adapted from Nickel JC, et al. J Urol. 2016;196(3):747-754
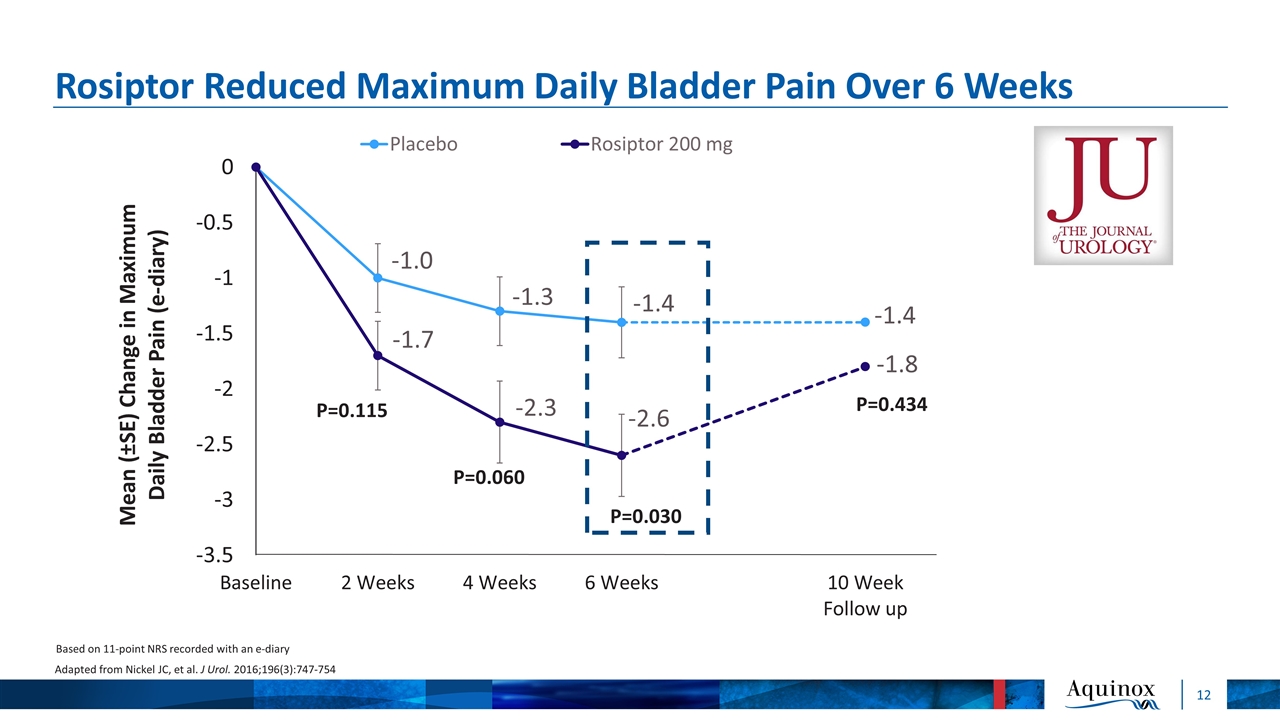
Rosiptor Reduced Maximum Daily Bladder Pain Over 6 Weeks Based on 11-point NRS recorded with an e-diary Adapted from Nickel JC, et al. J Urol. 2016;196(3):747-754 P=0.115 P=0.060 P=0.030
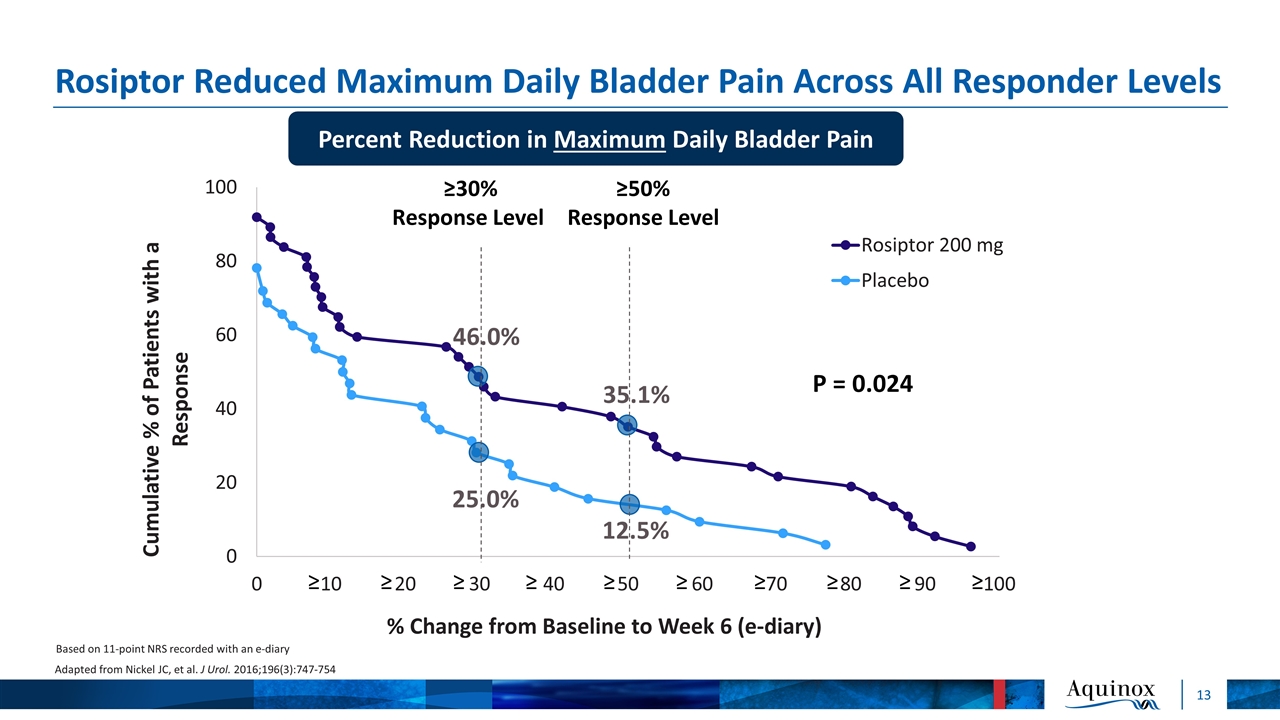
Rosiptor Reduced Maximum Daily Bladder Pain Across All Responder Levels P = 0.024 46.0% 25.0% 35.1% 12.5% ≥30% Response Level ≥50% Response Level Based on 11-point NRS recorded with an e-diary Adapted from Nickel JC, et al. J Urol. 2016;196(3):747-754 ≥ ≥ ≥ ≥ ≥ ≥ ≥ ≥ ≥ ≥ Percent Reduction in Maximum Daily Bladder Pain
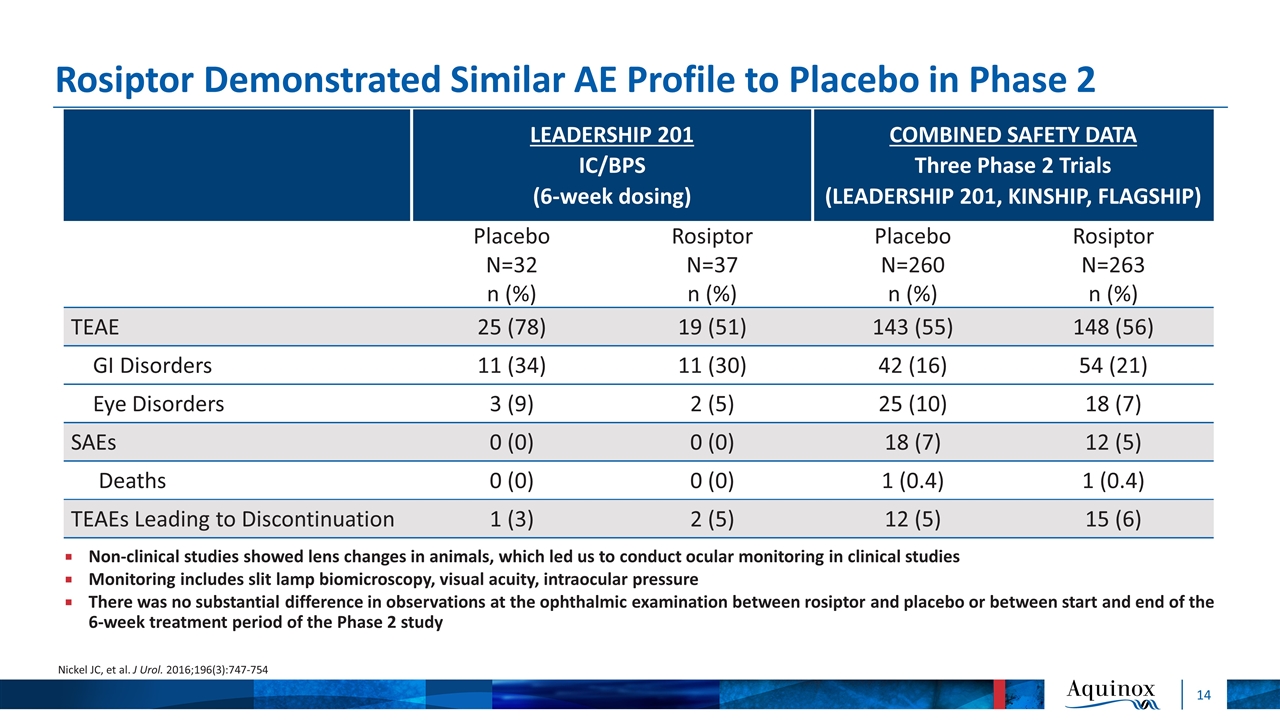
Rosiptor Demonstrated Similar AE Profile to Placebo in Phase 2 LEADERSHIP 201 IC/BPS (6-week dosing) COMBINED SAFETY DATA Three Phase 2 Trials (LEADERSHIP 201, KINSHIP, FLAGSHIP) Placebo N=32 n (%) Rosiptor N=37 n (%) Placebo N=260 n (%) Rosiptor N=263 n (%) TEAE 25 (78) 19 (51) 143 (55) 148 (56) GI Disorders 11 (34) 11 (30) 42 (16) 54 (21) Eye Disorders 3 (9) 2 (5) 25 (10) 18 (7) SAEs 0 (0) 0 (0) 18 (7) 12 (5) Deaths 0 (0) 0 (0) 1 (0.4) 1 (0.4) TEAEs Leading to Discontinuation 1 (3) 2 (5) 12 (5) 15 (6) Nickel JC, et al. J Urol. 2016;196(3):747-754 Non-clinical studies showed lens changes in animals, which led us to conduct ocular monitoring in clinical studies Monitoring includes slit lamp biomicroscopy, visual acuity, intraocular pressure There was no substantial difference in observations at the ophthalmic examination between rosiptor and placebo or between start and end of the 6-week treatment period of the Phase 2 study

LEADERSHIP 301: Phase 3 Trial with Rosiptor in IC/BPS

LEADERSHIP 301: Assessing Bladder Pain and Urinary Symptoms 12-week treatment phase followed by 52-week extension phase Primary endpoint: Change from baseline to Week 12 in the maximum daily bladder pain score based on an 11-point NRS recorded by e-diary Key secondary endpoints: Change from baseline to Week 12 for the following: Voiding frequency over a 24-hour period Bladder Pain/Interstitial Cystitis Symptom Score (BPIC-SS) Subject’s Global Response Assessment (GRA) at Week 12 AEs over 12-week treatment phase followed by 52-week extension phase Top-Line Data Expected Q3 2018
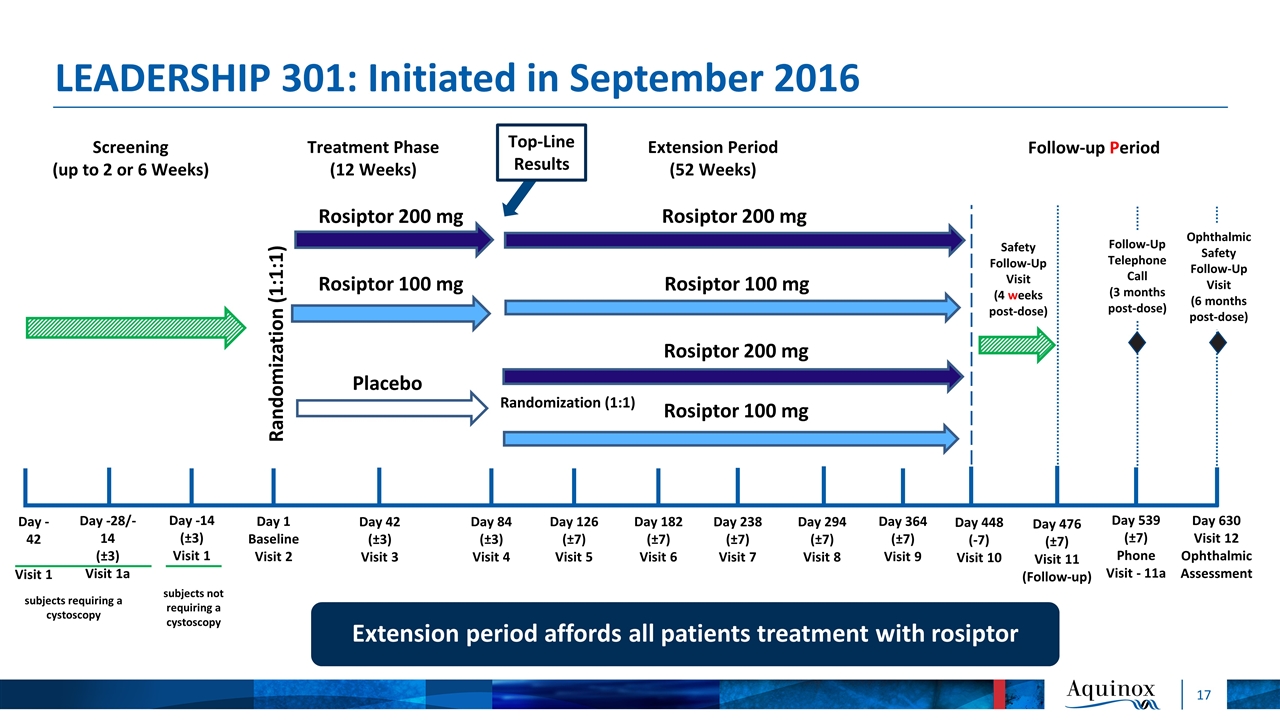
LEADERSHIP 301: Initiated in September 2016 Day -42 Visit 1 Day -28/-14 (±3) Visit 1a Day -14 (±3) Visit 1 Day 1 Baseline Visit 2 Day 42 (±3) Visit 3 Day 84 (±3) Visit 4 Day 126 (±7) Visit 5 Day 182 (±7) Visit 6 Day 238 (±7) Visit 7 Day 294 (±7) Visit 8 Day 364 (±7) Visit 9 Day 476 (±7) Visit 11 (Follow-up) subjects not requiring a cystoscopy Randomization (1:1:1) Randomization (1:1) Rosiptor 200 mg Rosiptor 100 mg Placebo Rosiptor 200 mg Rosiptor 100 mg Screening (up to 2 or 6 Weeks) Treatment Phase (12 Weeks) Extension Period (52 Weeks) Safety Follow-Up Visit (4 weeks post-dose) Rosiptor 200 mg Rosiptor 100 mg Ophthalmic Safety Follow-Up Visit (6 months post-dose) Day 448 (-7) Visit 10 Day 539 (±7) Phone Visit - 11a Day 630 Visit 12 Ophthalmic Assessment Follow-up Period subjects requiring a cystoscopy Follow-Up Telephone Call (3 months post-dose) Extension period affords all patients treatment with rosiptor Top-Line Results

Commercial Opportunity January 2018
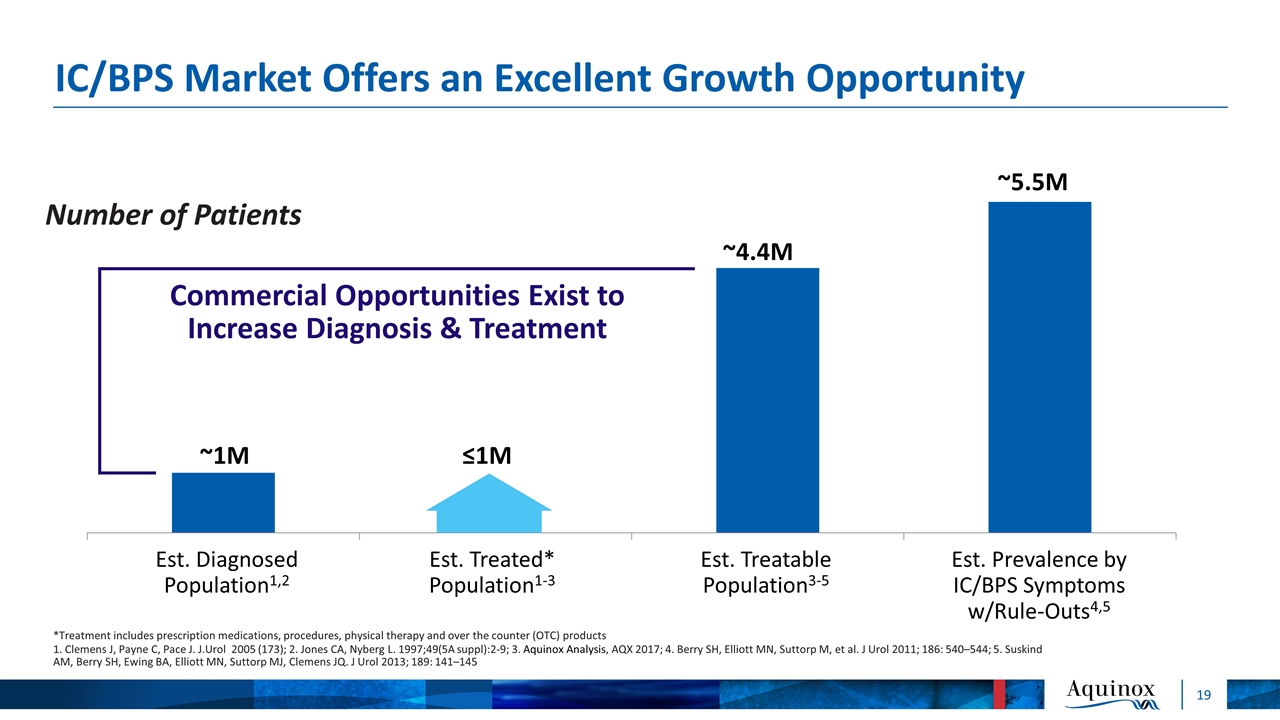
IC/BPS Market Offers an Excellent Growth Opportunity Number of Patients *Treatment includes prescription medications, procedures, physical therapy and over the counter (OTC) products 1. Clemens J, Payne C, Pace J. J.Urol 2005 (173); 2. Jones CA, Nyberg L. 1997;49(5A suppl):2-9; 3. Aquinox Analysis, AQX 2017; 4. Berry SH, Elliott MN, Suttorp M, et al. J Urol 2011; 186: 540–544; 5. Suskind AM, Berry SH, Ewing BA, Elliott MN, Suttorp MJ, Clemens JQ. J Urol 2013; 189: 141–145 Est. Treated* Population1-3 Est. Diagnosed Population1,2 Est. Treatable Population3-5 Est. Prevalence by IC/BPS Symptoms w/Rule-Outs4,5 ≤1M ~1M ~4.4M ~5.5M Commercial Opportunities Exist to Increase Diagnosis & Treatment

Second-Line Treatments Appropriate manual physical therapy techniques Oral: amitriptyline, cimetidine, hydroxyzine, or Elmiron® Intravesical: DMSO, heparin, or lidocaine Pain management Few Clinically Effective and/or FDA Approved Treatment Options AUA Guidelines Adapted from Hanno PM, Burks DA, Clemens JQ et al. American Urological Association (AUA) Guideline. Approved September 2014. First-Line Treatments General relaxation/stress management Pain management Patient education Self-care/behavioral modification Third, Fourth, Fifth, & Sixth-Line Treatments Additional treatment options to consider depending on symptom severity, clinician judgement, and patient preferences Only 4 Oral Options Recommended

Significant Opportunity to Improve Treatment Paradigm 1. Hanno PM. Urology. 1997;49(suppl 5A):93-99. * IQVIA (formerly IMS) data, FY 2016; IC/BPS only not other uses Elmiron® (PPS), the only oral product indicated for IC/BPS, has many limiting attributes: Limited efficacy in clinical trials and practice TID dosing Unpleasant side effect profile Delayed symptom improvement if patient responds (~3 months) Low adherence/persistence rates1 Elmiron is the most frequently prescribed IC/BPS treatment with ~340K total prescriptions in the U.S. & ~$300M in U.S. revenues*
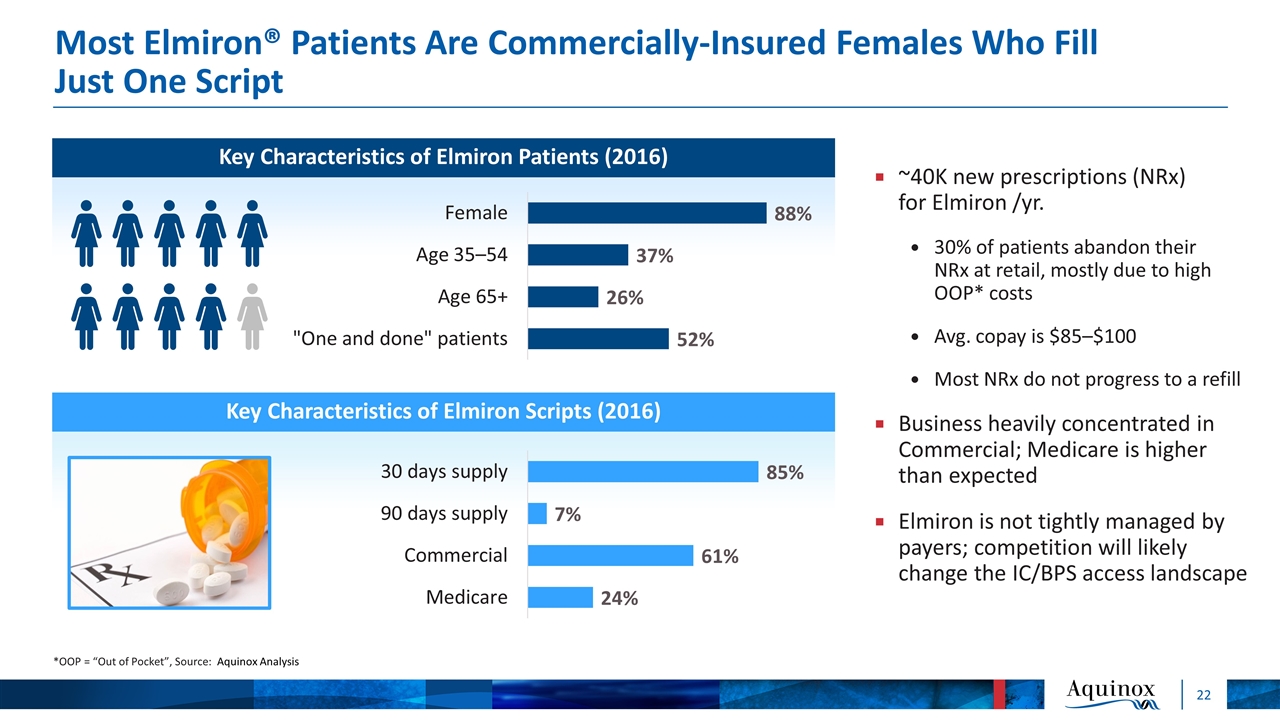
Most Elmiron® Patients Are Commercially-Insured Females Who Fill Just One Script *OOP = “Out of Pocket”, Source: Aquinox Analysis Key Characteristics of Elmiron Patients (2016) ~40K new prescriptions (NRx) for Elmiron /yr. 30% of patients abandon their NRx at retail, mostly due to high OOP* costs Avg. copay is $85–$100 Most NRx do not progress to a refill Business heavily concentrated in Commercial; Medicare is higher than expected Elmiron is not tightly managed by payers; competition will likely change the IC/BPS access landscape Key Characteristics of Elmiron Scripts (2016)

of New Patients Drop-Off When Time 2nd Fill 52% 180 Day Refill Rates for Elmiron New-to-Brand Patients First Fill Elmiron® Persistence is Low; Nearly 70% of Elmiron Patients Have Discontinued Treatment by 90 Days Half of all Elmiron patients fill within 15 days suggesting patients are relatively adherent. However, among new patients, Elmiron refill rates drop off with half of all new users not filling beyond the 1st Rx indicating persistence challenges Time between determined as end date - beginning date - days of supply divided by number of Rxs Source: Aquinox Analysis Gap Between Fills % of Elmiron Patients 0 ~25% 15 Days (Median) ~25% >15 Days ~50% 50% Average Rx/Patient/Year: 4 New Patient (44% of Patients): 2 Long-standing (56% of Patients): 5 Days Post First Fill
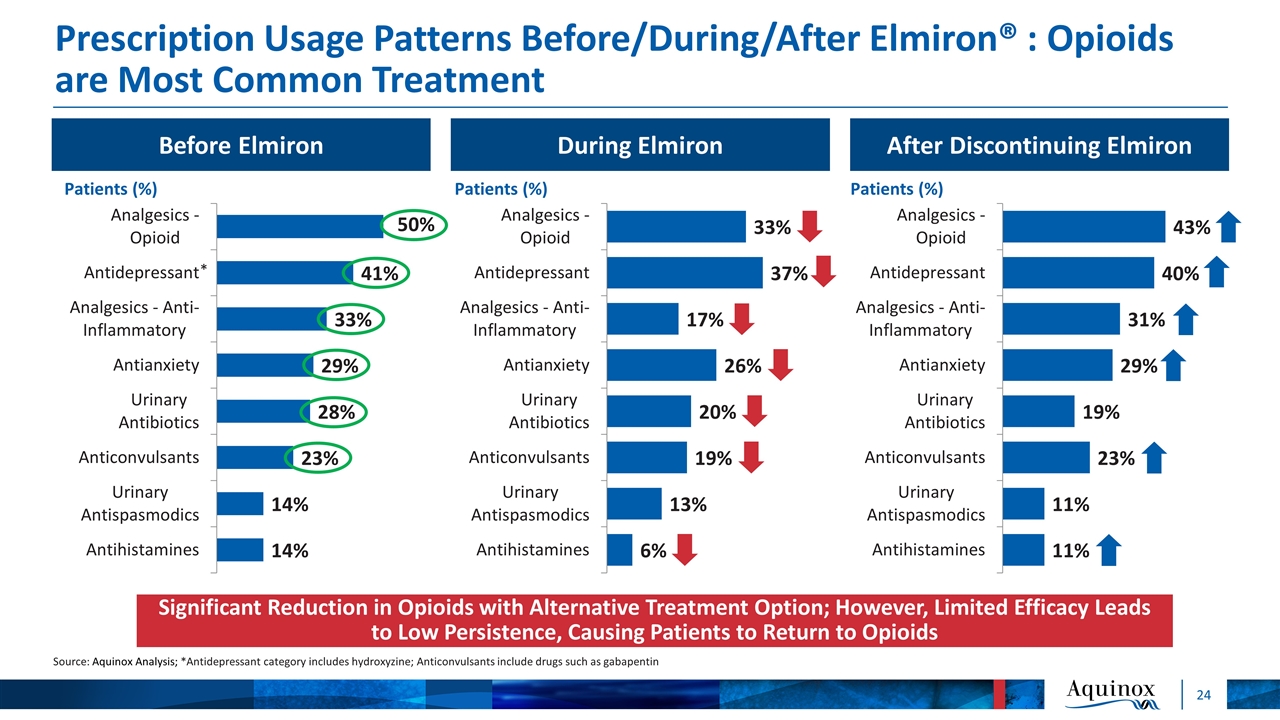
Before Elmiron Prescription Usage Patterns Before/During/After Elmiron® : Opioids are Most Common Treatment Source: Aquinox Analysis; *Antidepressant category includes hydroxyzine; Anticonvulsants include drugs such as gabapentin After Discontinuing Elmiron During Elmiron Significant Reduction in Opioids with Alternative Treatment Option; However, Limited Efficacy Leads to Low Persistence, Causing Patients to Return to Opioids * 50%

Urologists Will Be Key in Driving Diagnosis & Treatment Decisions 1. IMS data 2016; 2. Aquinox Analysis Most commonly (60%) visited by patients seeking medical care for IC/BPS1 Most frequently diagnose IC/BPS (patient reported)2 Write ~60% of all prescriptions for IC/BPS1 Generate the highest volume of Elmiron® prescriptions Patients visiting a Urologist for IC/BPS leave with at least one prescription Aquinox is building relationships within the Urology community
Aquinox is Committed to Raising Awareness for IC/BPS Building Relationships with Key Advocacy Groups Patient Healthcare Provider American Urogynecologic Society American Urology Association (AUA) Urology Care Foundation (UCF)
Exploring Additional Opportunities with Rosiptor Potential Development with Rosiptor Expanding Urology Gastrointestinal

Chronic Prostatitis/Chronic Pelvic Pain Syndrome (CP/CPPS)

Chronic Prostatitis/Chronic Pelvic Pain Syndrome (CP/CPPS) Offers Aquinox a Synergistic Commercial Opportunity in Urology Characterized by pelvic pain (unrelated to urinary bladder filling or emptying), lower urinary tract symptoms without evidence of urinary tract infection1-3 Unmet medical need No FDA-approved products & few in development Antibiotics used regardless of (-) culture findings, followed by alpha-blockers1 No single agent has proven efficacious to date across a broad population Inflammation is believed to be a critical driver in IC/BPS and CP/CPPS4,5 CP/CPPS has a lower prevalence rate than IC/BPS, but similar number of patients diagnosed and treated (~1M)6-9 1. Nickel J, et al. CUAJ. 2011;5(5): 306-315; 2. Clemens JQ, et al. Urology. 2009;74(5):983-987; 3. Hanno PM, et al. J Urol. 1999;161:553-557; 4. Nickel JC, et al. J Urol 2017;198:1-7; 5. Grover S, et al. Ther Adv Urol. 2011;3(1):19-33; 6. Suskind AM, et al. J Urol 2013;189(1):141-145; 7. Clemens J, Payne C, Pace J. J.Urol 2005 (173); 8. Jones CA, Nyberg L. 1997;49(5A suppl):2-9; 9. Data on File, AQX 2017. #1 Area of Unmet Need: Effective Therapy to Improve Symptomatology

Prospective Near-Term Milestones Publication of LEADERSHIP 201 Data ü Initiation of IC/BPS – LEADERSHIP 301 ü Complete ADME Trial ü Initiate Carcinogenicity Studies ü FDA Advisory Committee Meeting on IC/BPS ü Initiation of Ethnobridging Trial (Q1 2018) Initiation of Drug/Drug Interaction Trial (Q1 2018) LEADERSHIP 301 Last Patient Randomized (Q1 2018) CP/CPPS Phase 2 Trial 1st Patient Randomized (Early 2018) LEADERSHIP 301 Top-Line Data (Q3 2018) Near-term Data, Expanded Market Opportunities and Pipeline Advancement

Financial & Stock Information ~ $119.7M cash, cash equivalents and short-term and long-term investments as of Sept 30, 2017 ~$37.6M operating expenses in 2016 Existing cash to fund first LEADERSHIP 301 trial and supporting activities 23.5M shares outstanding (~25.5M fully diluted) NASDAQ : AQXP
