
1 NASDAQ: OPHT January 2018 36th Annual J.P. Morgan Healthcare Conference Glenn Sblendorio, Chief Executive Officer and President

2 Forward‐looking statements Any statements in this presentation about Ophthotech’s future expectations, plans and prospects constitute forward‐looking statements for purposes of the safe harbor provisions under the Private Securities Litigation Reform Act of 1995. Forward‐looking statements include any statements about Ophthotech’s strategy, future operations and future expectations and plans and prospects for Ophthotech, and any other statements containing the words “anticipate,” “believe,” “estimate,” “expect,” “intend”, “goal,” “may”, “might,” “plan,” “predict,” “project,” “target,” “potential,” “will,” “would,” “could,” “should,” “continue,” and similar expressions. In this presentation, Ophthotech’s forward looking statements include statements about the implementation of its strategic plan, Ophthotech's projected use of cash and cash balances, the timing, progress and results of clinical trials and other development activities, and the potential for its business development strategy, including any potential in‐license or acquisition opportunities. Such forward‐looking statements involve substantial risks and uncertainties that could cause Ophthotech’s clinical development programs, future results, performance or achievements to differ significantly from those expressed or implied by the forward‐looking statements. Such risks and uncertainties include, among others, those related to the initiation and conduct of clinical trials, availability of data from clinical trials, expectations for regulatory matters and negotiation and consummation of in‐license and/or acquisition transactions, need for additional financing and other factors discussed in the “Risk Factors” section contained in the quarterly and annual reports that Ophthotech files with the Securities and Exchange Commission. Any forward‐looking statements represent Ophthotech’s views only as of the date of this presentation. Ophthotech anticipates that subsequent events and developments will cause its views to change. While Ophthotech may elect to update these forward‐looking statements at some point in the future, Ophthotech specifically disclaims any obligation to do so except as required by law.

3 Science Driven and Retina Focused • Deep Expertise in Ophthalmic Drug Development - Multiple retina specialists - Strong global KOL network to facilitate clinical execution - Highly experienced clinical development team • Current Clinical Programs - Age‐related Clinical trials in wet and dry AMD currently ongoing Multi‐billion dollar market opportunities - Orphan Significant unmet medical need Multiple programs ongoing or planned, led by a program in autosomal recessive Stargardt disease • Business Development Strategy - Orphan ophthalmic and retinal diseases with therapeutic and gene therapy solutions • Strong Cash Position – ~$167 million in cash and cash equivalents as of 12/31/171 1 Unaudited estimate Ophthalmology: Age‐related and Orphan Indications
4 Value Creation: Multiple Track Strategy Age‐related Diseases Multiple opportunities in large markets where medical need remains for patients Orphan Diseases Focus on underserved patients with the potential for an accelerated path to market Business Development Disciplined approach to evaluation of therapeutic and gene therapy solutions to ophthalmic diseases
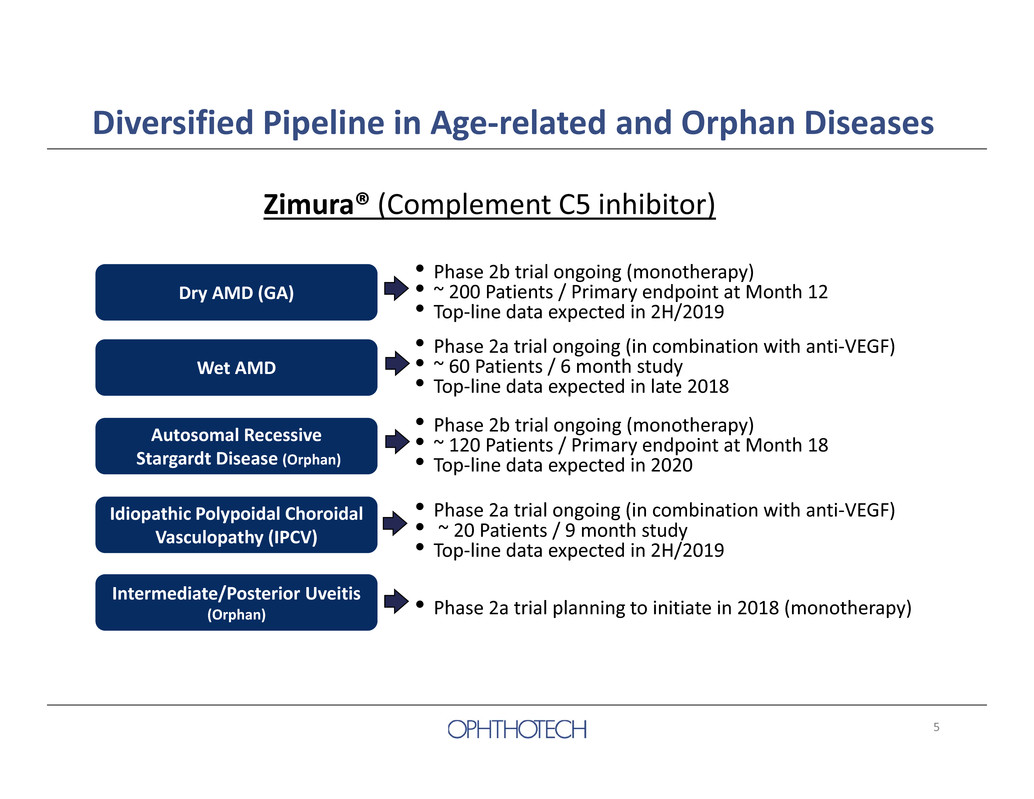
5 Diversified Pipeline in Age‐related and Orphan Diseases Zimura® (Complement C5 inhibitor) Autosomal Recessive Stargardt Disease (Orphan) • Phase 2b trial ongoing (monotherapy) • ~ 120 Patients / Primary endpoint at Month 18 • Top‐line data expected in 2020 Wet AMD • Phase 2a trial ongoing (in combination with anti‐VEGF) • ~ 60 Patients / 6 month study • Top‐line data expected in late 2018 Idiopathic Polypoidal Choroidal Vasculopathy (IPCV) • Phase 2a trial ongoing (in combination with anti‐VEGF) • ~ 20 Patients / 9 month study • Top‐line data expected in 2H/2019 Intermediate/Posterior Uveitis (Orphan) • Phase 2a trial planning to initiate in 2018 (monotherapy) Dry AMD (GA) • Phase 2b trial ongoing (monotherapy) • ~ 200 Patients / Primary endpoint at Month 12 • Top‐line data expected in 2H/2019
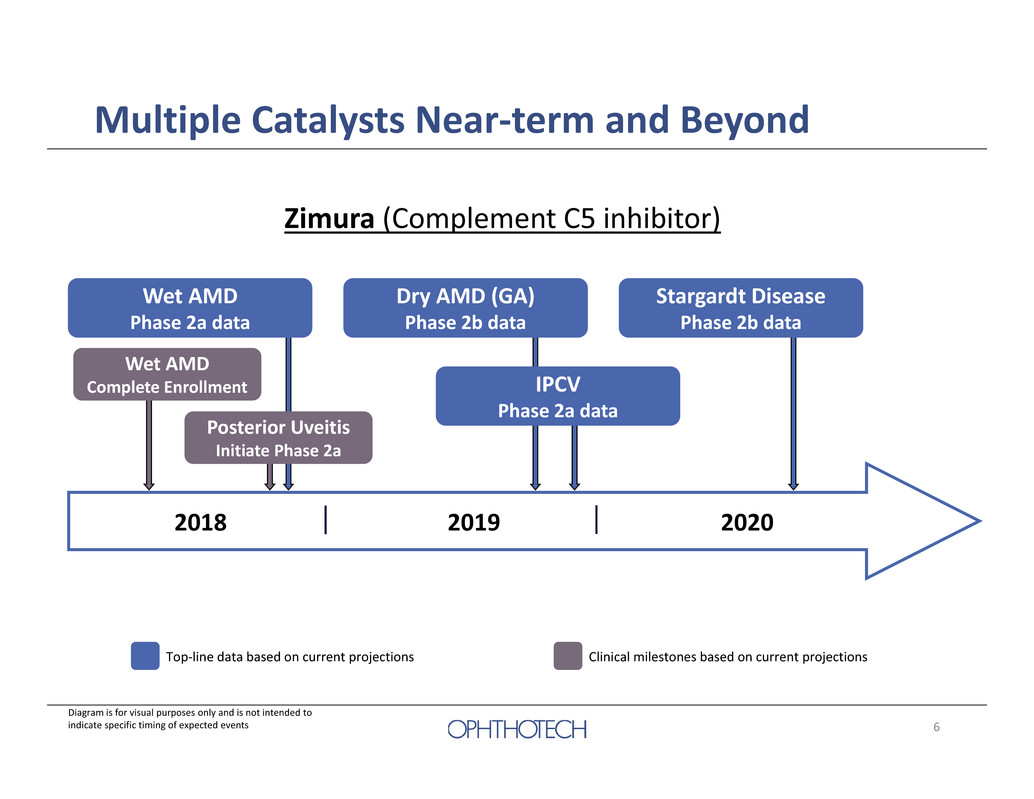
6 Multiple Catalysts Near‐term and Beyond Zimura (Complement C5 inhibitor) Wet AMD Complete Enrollment 2018 2019 2020 Wet AMD Phase 2a data Dry AMD (GA) Phase 2b data Stargardt Disease Phase 2b data Top‐line data based on current projections Posterior Uveitis Initiate Phase 2a IPCV Phase 2a data Diagram is for visual purposes only and is not intended to indicate specific timing of expected events Clinical milestones based on current projections

7 Zimura, C5 Complement Inhibitor Geographic Atrophy Secondary to Dry AMD

8 • Major unmet medical need – Large market with no approved treatment options available • Role of complement in dry AMD1 – Complement deposition increases with aging – Complement activation leads to the formation and accumulation of inflammasomes and Membrane Attack Complex (MAC) – Inflammasomes and MAC lead to retinal pigment epithelial (RPE) cell death – RPE degeneration leads to photoreceptor cell death and loss of vision 1 The Journal of Biological Chemistry Vol. 290, NO. 52, pp. 31189–31198, December 25, 2015. Invest Ophthalmol Vis Sci. 2013;54:110–120. J Immunol. 2015; 195:3382‐3389. Med Sci Monit, 2010; 16(1): BR17‐23. Am J Ophthalmol 2002;134:411–431. Proc Natl Acad Sci USA. 2005, 102(20), 7053‐7054. Development of Zimura for Geographic Atrophy Secondary to Dry AMD
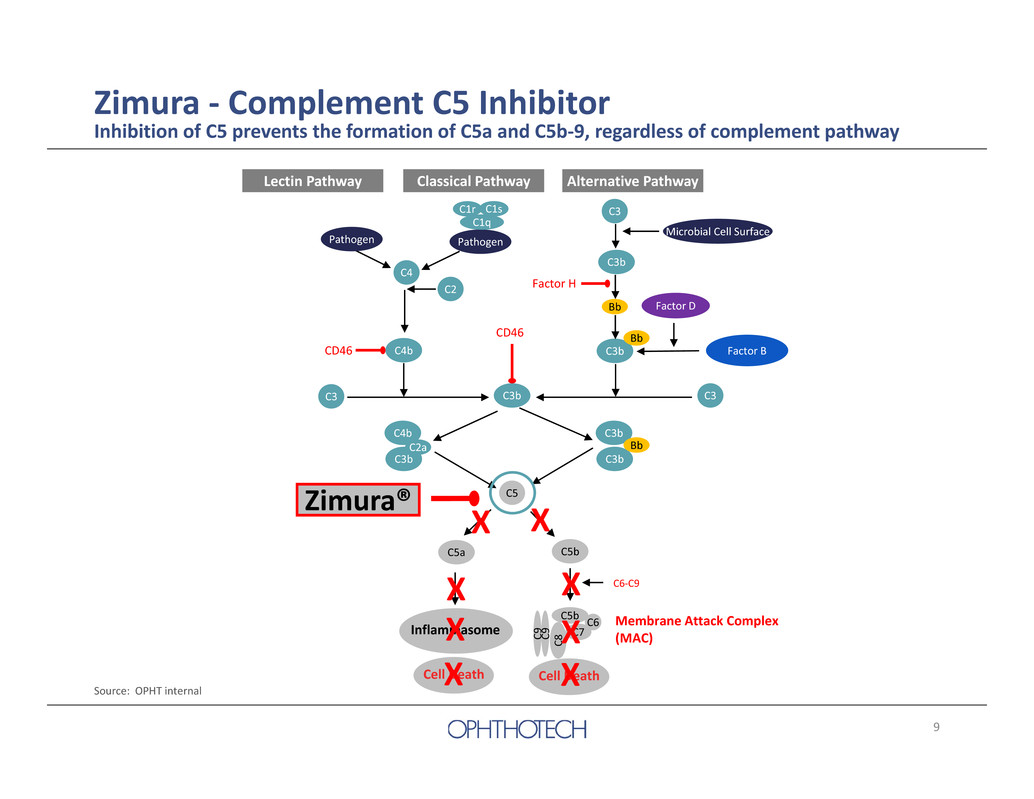
9 C3 C3b C5 C4 C2 C3 C3 C3b C3b C4b C4b C3b C5b C5b C 8 C 9 C 9 C6 C7 Pathogen C3bC3b C2a C6‐C9 Factor H C1q C1r C1s CD46 CD46 Microbial Cell Surface Alternative PathwayLectin Pathway Classical Pathway Pathogen Zimura ‐ Complement C5 Inhibitor Inhibition of C5 prevents the formation of C5a and C5b‐9, regardless of complement pathway Source: OPHT internal Cell Death Membrane Attack Complex (MAC) C5a Cell Death Inflammasome Bb Bb Bb Factor B Factor D Zimura® X X X XX X X X

10 Zimura Phase 1/2a Dry AMD (GA) – Completed* Study Design Intravitreal Zimura was administered for a maximum of 5 injections at one of two dose levels (0.3 mg/eye or 1mg/eye) 47 Patients Enrolled 0.3 mg dose group (n=24) 1 mg dose group (n=23) 0 4 8 12 16 20 24 28 32 36 40 44 48Week Zimura Dosed *Uncontrolled safety trial; small sample size
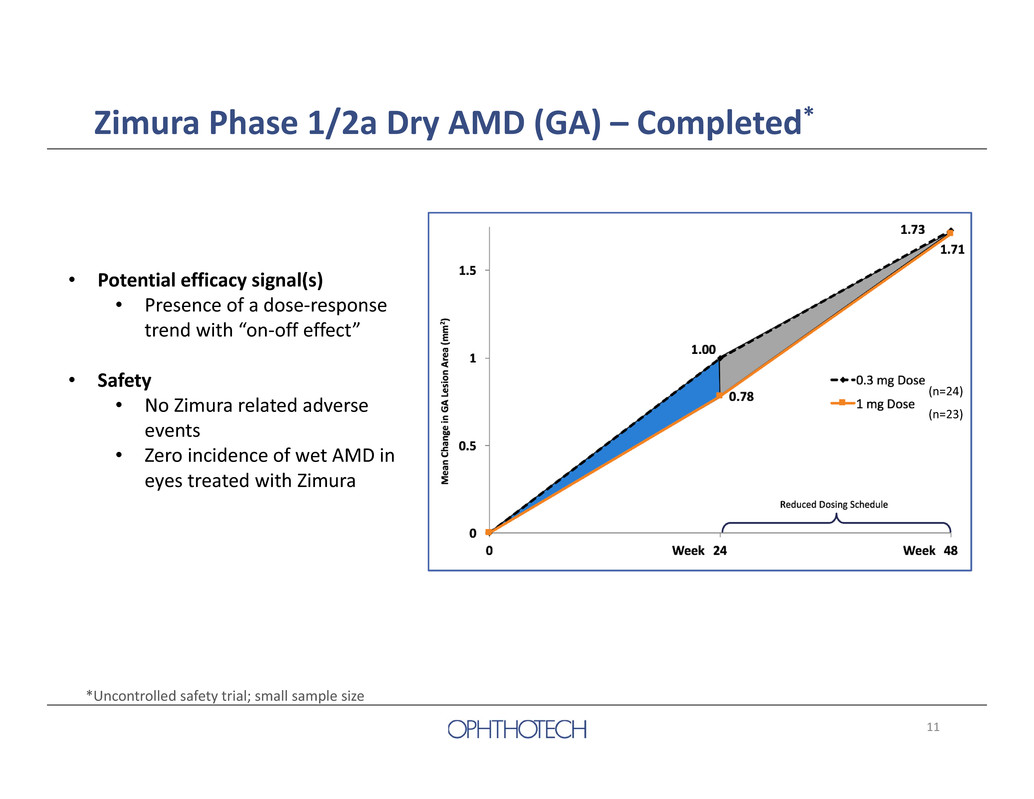
11 Zimura Phase 1/2a Dry AMD (GA) – Completed* • Potential efficacy signal(s) • Presence of a dose‐response trend with “on‐off effect” • Safety • No Zimura related adverse events • Zero incidence of wet AMD in eyes treated with Zimura *Uncontrolled safety trial; small sample size (n=24) (n=23)

12 Zimura Phase 2b Dry AMD (GA) Clinical Trial – Ongoing • Phase 2b, randomized, double masked, sham controlled clinical trial • Study recently amended to accelerate anticipated timeline to obtain data • ~ 200 subjects will be treated with monthly study treatment (Zimura or Sham) for 18 months • Primary Efficacy Endpoint – Mean rate of change in GA over 12 months measured by fundus autofluorescence (FAF) at three time points Top‐line data expected in 2H 2019

13 Zimura, C5 Complement Inhibitor Wet Age‐Related Macular Degeneration

14 “The processes responsible for the decrease in vision in CATT and other studies are multiple, but seem to be related to an increase in the proportion patients with an abnormally thin retina (< 120 µm), an increase in prevalence of geographic atrophy, . . . " “These data highlight the need for agents that can prevent or minimize geographic atrophy . . . ” Unmet Need Current Standard of Care – Anti‐VEGF Monotherapy Source: Ophthalmology 2016;123:1751‐1761

15 • Unmet medical need remains– major market opportunity • Anti‐VEGF monotherapy: – Shown to reach a ceiling effect – Majority of patients do not reach a visual acuity of 20/40 or better – In the real world most patients lose vision over time • Role of Complement in Wet AMD1 – VEGF Increases Complement Factor H (CFH) (regulator of complement activation) – CFH decreases complement activation – Anti‐VEGF Increases complement activation – Patients receiving anti‐VEGF monotherapy may develop geographic atrophy2 • Adding Zimura to anti‐VEGF therapy may improve the efficacy and safety of anti‐VEGF 1J Clin Invest. 2017;127(1):199‐214 2 Ophthalmology 2014; 121:150‐161. Development of Zimura for Wet AMD
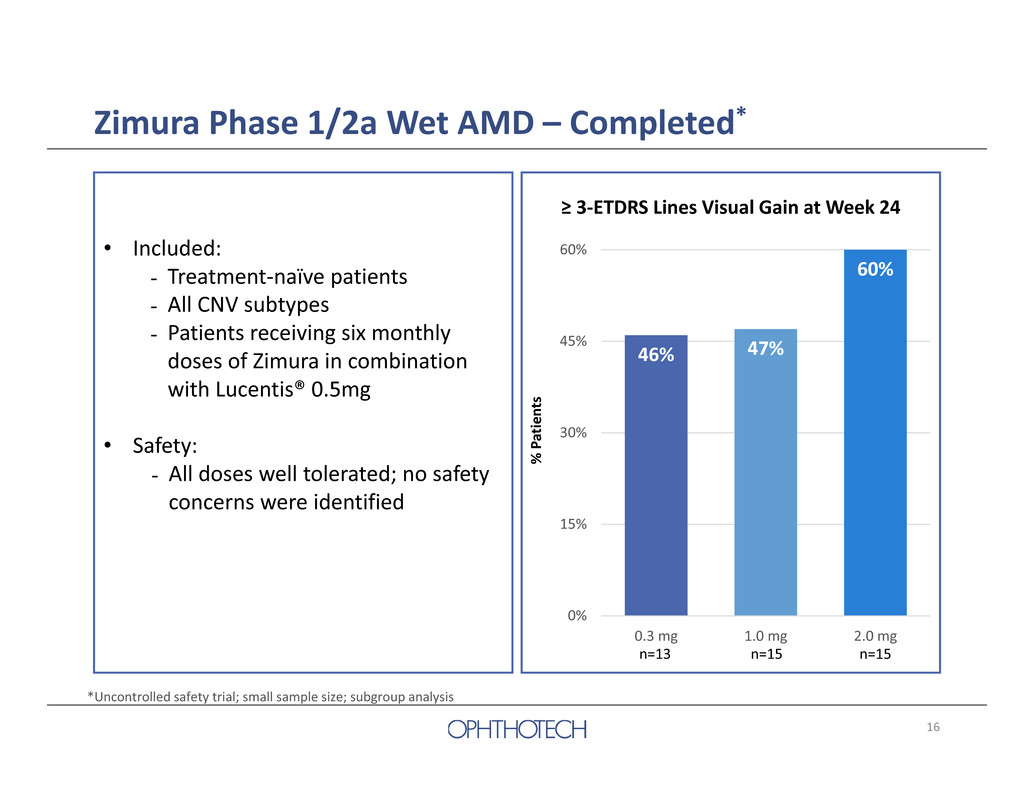
16 Zimura Phase 1/2a Wet AMD – Completed* 46% 47% 60% 0% 15% 30% 45% 60% 0.3 mg 1.0 mg 2.0 mg n=13 n=15 n=15 ≥ 3‐ETDRS Lines Visual Gain at Week 24 % P a t i e n t s • Included: - Treatment‐naïve patients - All CNV subtypes - Patients receiving six monthly doses of Zimura in combination with Lucentis® 0.5mg • Safety: - All doses well tolerated; no safety concerns were identified *Uncontrolled safety trial; small sample size; subgroup analysis

17 • Phase 2a open label clinical trial • N = ~ 60 subjects • Objectives: – To assess the safety of intravitreal Zimura administered in combination with Lucentis® 0.5 mg in treatment naïve subjects with wet AMD – Dose ranging – Validate results from previously completed Phase 1/2a • Duration: 6 months Top‐line data expected in late 2018 Zimura Wet AMD Clinical Trial – Ongoing

18 Zimura, C5 Complement Inhibitor Autosomal Recessive Stargardt Disease (STGD1) (Orphan Indication)

19 • High unmet medical need – Orphan disease – No FDA or EMA approved treatment available • Role of Complement in Stargardt Disease1 – Bisretinoids (visual cycle waste) activate complement – Complement inhibition rescues photoreceptor cells in a Stargardt animal model – Anti‐C5 improved RPE cell viability in bisretinoid/complement cell culture model 1 The Journal of Biological Chemistry. 2011; 286(21): 18593–18601. Proc Natl Acad Sci U S A. 2017; 114(15):3987‐3992. Invest Ophthalmol Vis Sci. 2013;54:2669‐2677 Development of Zimura in Autosomal Recessive Stargardt Disease
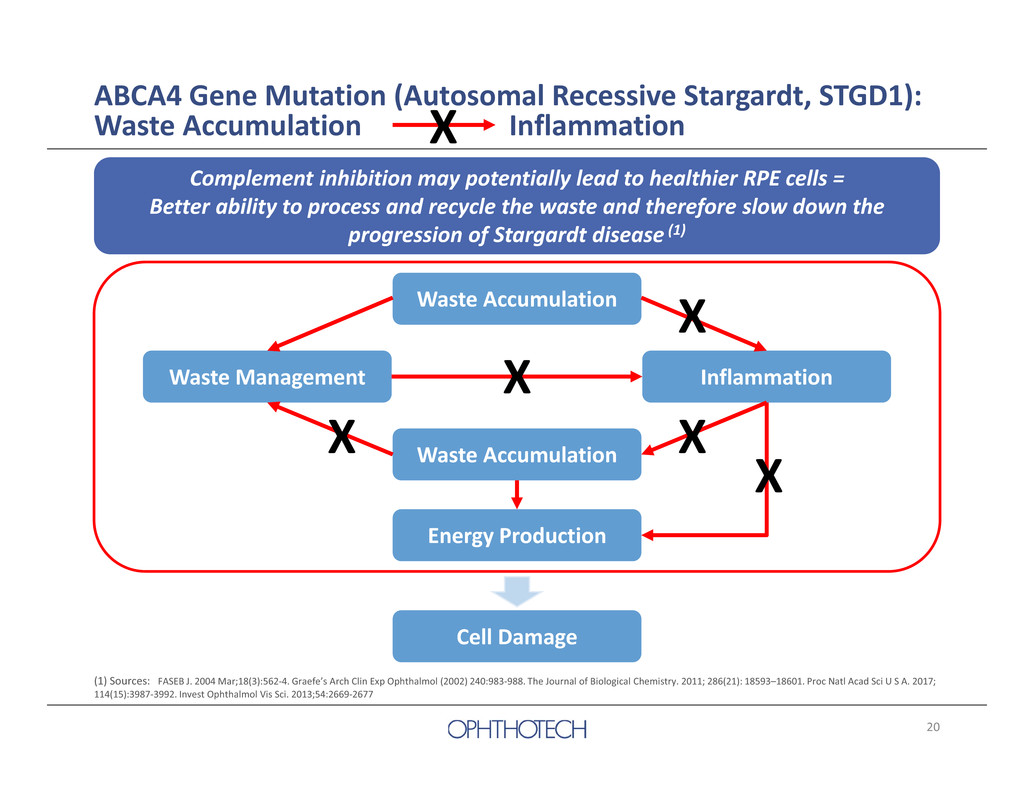
20 ABCA4 Gene Mutation (Autosomal Recessive Stargardt, STGD1): Waste Accumulation InflammationX Complement inhibition may potentially lead to healthier RPE cells = Better ability to process and recycle the waste and therefore slow down the progression of Stargardt disease (1) Waste Accumulation Waste Accumulation Energy Production Waste Management Inflammation Cell Damage X XX X X (1) Sources: FASEB J. 2004 Mar;18(3):562‐4. Graefe’s Arch Clin Exp Ophthalmol (2002) 240:983‐988. The Journal of Biological Chemistry. 2011; 286(21): 18593–18601. Proc Natl Acad Sci U S A. 2017; 114(15):3987‐3992. Invest Ophthalmol Vis Sci. 2013;54:2669‐2677

21 Stargardt Albino Abca4‐/‐Mice: Complement Inhibition Rescues Photoreceptors Expression of Complement Inhibitory Protein (CRRY) ~2 fold decrease in bisretinoid accumulation ~30% increase in the number of photoreceptor nuclei Normalized Complement Activity Source: Proc Natl Acad Sci U S A. 2017; 114(15):3987‐3992.

22 • Phase 2b, randomized, double masked, sham controlled clinical trial • N = ~ 120 subjects • Duration of treatment: 18 months • Primary Endpoint: Mean rate of change in the area of ellipsoid zone defect measured by en face SD‐OCT Top‐line data expected in 2020 Zimura Stargardt Disease (STGD1) Clinical Trial ‐ Initiated

23 • OPHT agreement with Foundation Fighting Blindness (FFB) – Highly‐distinguished organization recognized for its scientific commitment to orphan inherited retinal diseases – Established network of scientists and a robust patient registry • Access to FFB’s publicly available ProgStar study – Largest Natural History Study of Stargardt Disease – Data leveraged for Zimura Stargardt study design OPHT / Foundation Fighting Blindness
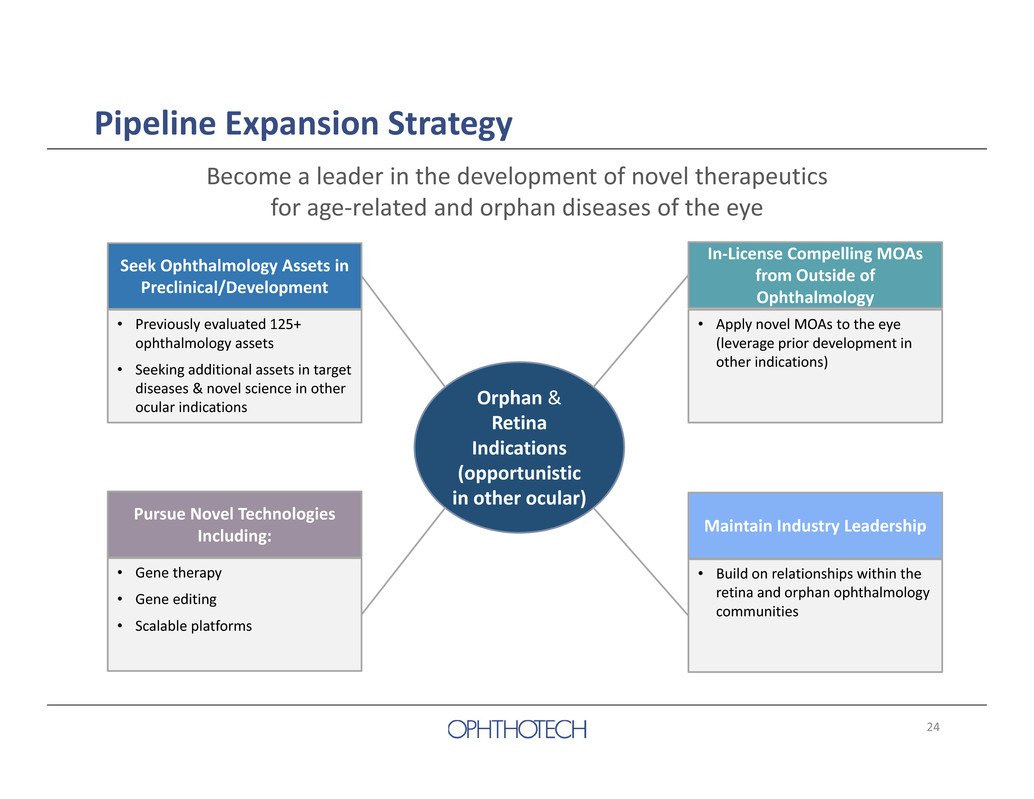
24 Become a leader in the development of novel therapeutics for age‐related and orphan diseases of the eye Orphan & Retina Indications (opportunistic in other ocular) Seek Ophthalmology Assets in Preclinical/Development • Previously evaluated 125+ ophthalmology assets • Seeking additional assets in target diseases & novel science in other ocular indications Pursue Novel Technologies Including: • Gene therapy • Gene editing • Scalable platforms Maintain Industry Leadership • Build on relationships within the retina and orphan ophthalmology communities In‐License Compelling MOAs from Outside of Ophthalmology • Apply novel MOAs to the eye (leverage prior development in other indications) Pipeline Expansion Strategy

25 Financial Highlights • ~$167 million in cash and cash equivalents as of 12/31/171 • External costs to bring Zimura programs to next phase of development expected to range between $25 million and $35 million2 • Cash corporate overhead expenses expected to average less than $2 million per month and continues to decline2, 3 1Unaudited estimate 2Guidance as of 11/8/17 and excludes any potential business development activities or any other changes to the Company’s current clinical development programs 3Cash corporate overhead expenses consist of cash expenditures for employees and external G&A expenses Strong Cash Position
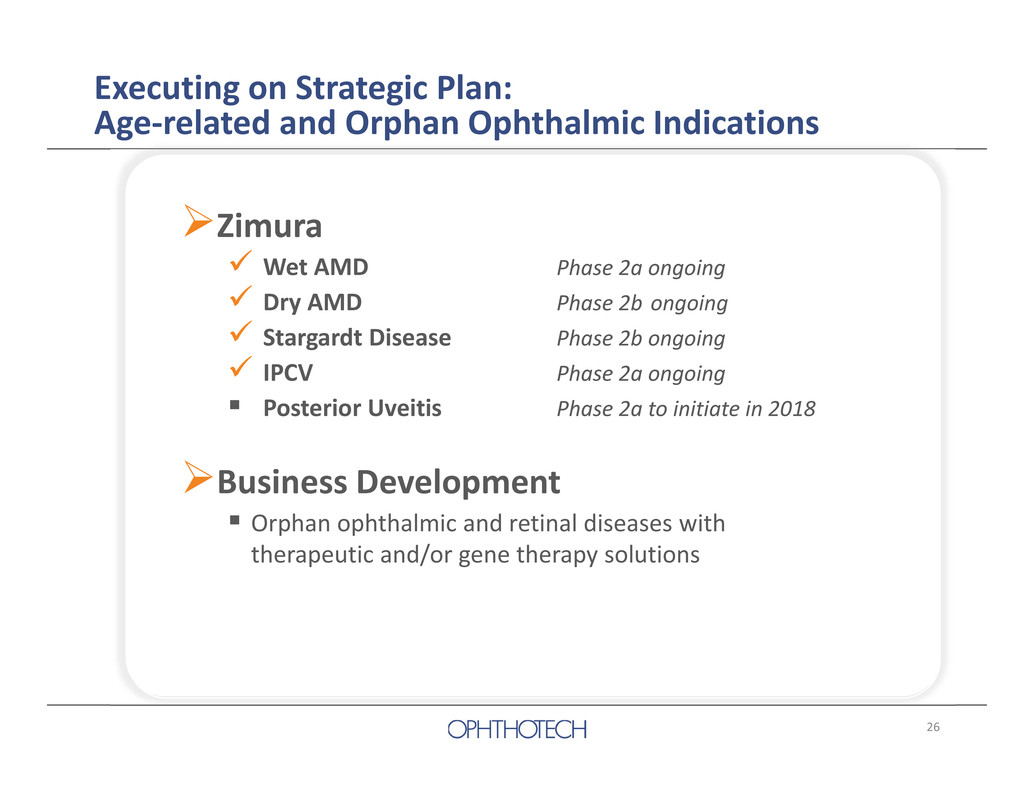
26 Zimura Wet AMD Phase 2a ongoing Dry AMD Phase 2b ongoing Stargardt Disease Phase 2b ongoing IPCV Phase 2a ongoing Posterior Uveitis Phase 2a to initiate in 2018 Business Development Orphan ophthalmic and retinal diseases with therapeutic and/or gene therapy solutions Executing on Strategic Plan: Age‐related and Orphan Ophthalmic Indications
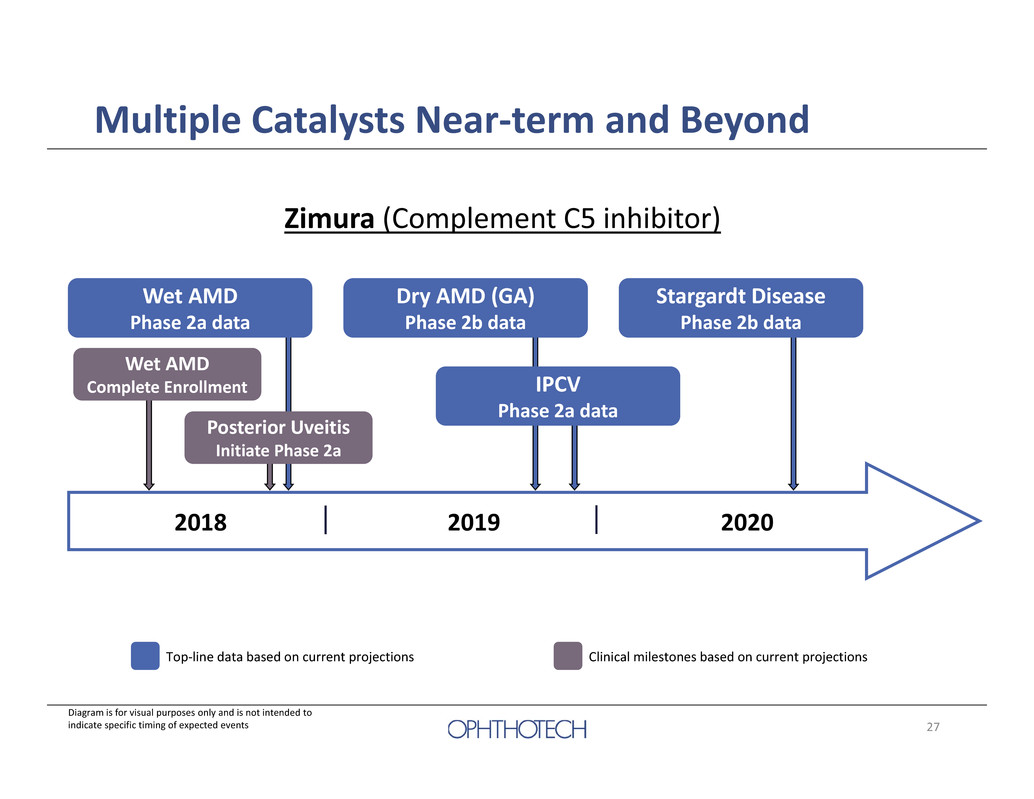
27 Multiple Catalysts Near‐term and Beyond Zimura (Complement C5 inhibitor) Wet AMD Complete Enrollment 2018 2019 2020 Wet AMD Phase 2a data Dry AMD (GA) Phase 2b data Stargardt Disease Phase 2b data Top‐line data based on current projections Posterior Uveitis Initiate Phase 2a IPCV Phase 2a data Diagram is for visual purposes only and is not intended to indicate specific timing of expected events Clinical milestones based on current projections

28 NASDAQ: OPHT January 2018 36th Annual J.P. Morgan Healthcare Conference Glenn Sblendorio, Chief Executive Officer and President