Exhibit 99.01

1 Investor Presentation January 2015 OTC QB : GNSZ Craig A. Dionne, PhD President & CEO

2 Safe Harbor Statement Safe Harbor Statement Under the Private Securities Litigation Reform Act of 1995: Any statements that are not historical facts are forward - looking statements that involve risks and uncertainties that could cause actual results to differ materially from those in the forward - looking statements, which may include, but are not limited to, factors related to GenSpera's anticipated growth strategies, the outcome of its clinical trials, future business development, ability to develop new products, expand to other related industries or markets in other geographical locations, and other information detailed from time to time in the Company's filings and future filings made with the United States Securities and Exchange Commission. Readers are advised that this information is intended for the use of investment professionals. Anyone interested in obtaining information on GenSpera should contact GenSpera directly. This presentation was developed by GenSpera and is intended solely for informational purposes and is not to be construed as an offer to sell or the solicitation of an offer to buy the Company's stock. This presentation is based upon information available to the public, as well as other information from sources which management believes to be reliable, but is not guaranteed by the Company as being accurate nor does it purport to be complete. Opinions expressed herein are those of management as of the date of the presentation and are subject to change without notice.

3 • An innovation driven biotech company that unlocks conventional thinking • Deep experience in cancer drug discovery and development • Cost - efficient business model • Robust global IP position • World class strategic partners • 2015 is tipping point for value creation GenSpera Profile

4 GenSpera Clinical Activity • GenSpera is a leader in developing prodrug therapeutics for the treatment of cancer • Unique, naturally derived, cancer killing drug has advantages in potency and significantly reduced side - effects • Mipsagargin, GenSpera’s lead candidate, is showing excellent clinical results • Highly differentiated from other anti - cancer approaches • Outstanding clinical safety profile • Data strongly suggestive of clinical activity in HCC • Positive proof of concept in patients at level of tumor physiology • Will be evaluated in 4 different cancer indications by Q2 2015 • Multiple development opportunities based on our prodrug platform • Four drug candidates in the pipeline

5 • World Class pedigree - technology developed at John Hopkins University and the University of Copenhagen • Our patents provide REAL value • Future company acquisition/partnerships • Expansive by design • Licensing • Current portfolio • Patent coverage in U.S. to 2023 • Orphan Drug Designation and patent restoration add significant coverage • Data exclusivity outside of U.S. - up to 10 years after drug approval in EU • Portfolio expansion strategy • Composition of matter PCT filed August 2013 • Worldwide patent exclusivity expected through 2033 Intellectual Property

6 Initial Market Opportunities Current drugs either are not very effective and/or have significant side effects Tremendous unmet needs in numerous types of cancer Mipsagargin attacks a universal attribute of all solid tumors Liver Cancer • 3rd largest cancer - killer worldwide • $10 billion by 2020 Brain Cancer • 13,000 deaths annually in the U.S. • Avastin expected to generate $460 million in revenue by 2017 Prostate Cancer • 240,000 new cases diagnosed annually in the U.S. • $6.7 billion by 2020 Kidney Cancer • 64,000 new cases diagnosed annually in the U.S. • 14,000 deaths annually in the U.S .
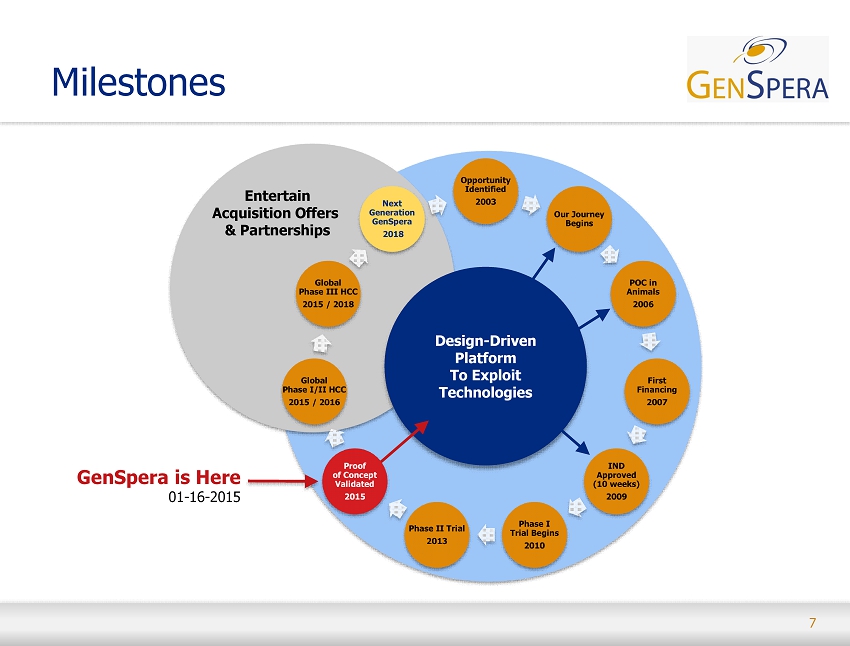
7 Milestones Opportunity Identified 2003 Our Journey Begins POC in Animals 2006 First Financing 2007 IND Approved (10 weeks) 2009 Phase I Trial Begins 2010 Phase II Trial 2013 Global Phase I/II HCC 2015 / 2016 Global Phase III HCC 2015 / 2018 Next Generation GenSpera 2018 GenSpera is Here 01 - 16 - 2015 Entertain Acquisition Offers & Partnerships Design - Driven Platform To Exploit Technologies Proof of Concept Validated 2015
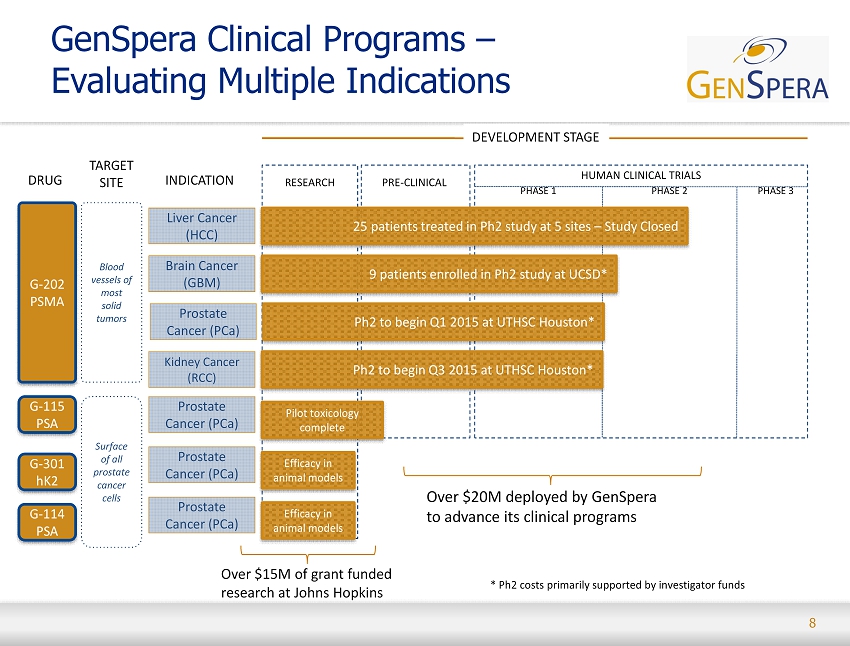
8 GenSpera Clinical Programs – Evaluating Multiple Indications RESEARCH PRE - CLINICAL PHASE 1 PHASE 2 Liver Cancer (HCC) TARGET SITE DRUG INDICATION Brain Cancer (GBM) Prostate Cancer (PCa) Prostate Cancer (PCa) Prostate Cancer ( PCa ) Prostate Cancer (PCa) G - 202 PSMA Blood vessels of most solid tumors Surface of all prostate cancer cells G - 114 PSA G - 301 hK2 G - 115 PSA DEVELOPMENT STAGE HUMAN CLINICAL TRIALS Over $15M of grant funded research at Johns Hopkins PHASE 3 25 patients treated in Ph2 study at 5 sites – Study Closed 9 patients enrolled in Ph2 study at UCSD* Ph2 to begin Q1 2015 at UTHSC Houston* Efficacy in animal models Efficacy in animal models Pilot toxicology complete * Ph2 costs primarily supported by investigator funds Kidney Cancer (RCC) Ph2 to begin Q3 2015 at UTHSC Houston* Over $20M deployed by GenSpera to advance its clinical programs

9 Mechanism of Action 1. Blood vessels feed living tumor 2. Drug circulates in bloodstream in benign fashion 3. Enzyme within tumor blood vessels activates drug 4. Activated drug kills blood vessels and tumor cells 5. Death of t umor As a result: • Fewer Side Effects • Potential for Complete Tumor Kill Better by Design

10 Impressive clinical activity • Highly differentiated from other anti - cancer approaches • Outstanding clinical safety profile • Data strongly suggestive of clinical activity in HCC • Will be evaluated in 4 different cancer indications by Q2 2015 • Unique molecular mechanism of action • Prolonged disease stabilization in a significant percentage of patients • 35% with stable disease > 7 months • Minimal and manageable side effects • Fatigue, nausea, rash, reversible creatinine increase • No apparent effect on bone marrow Mipsagargin Phase II Results
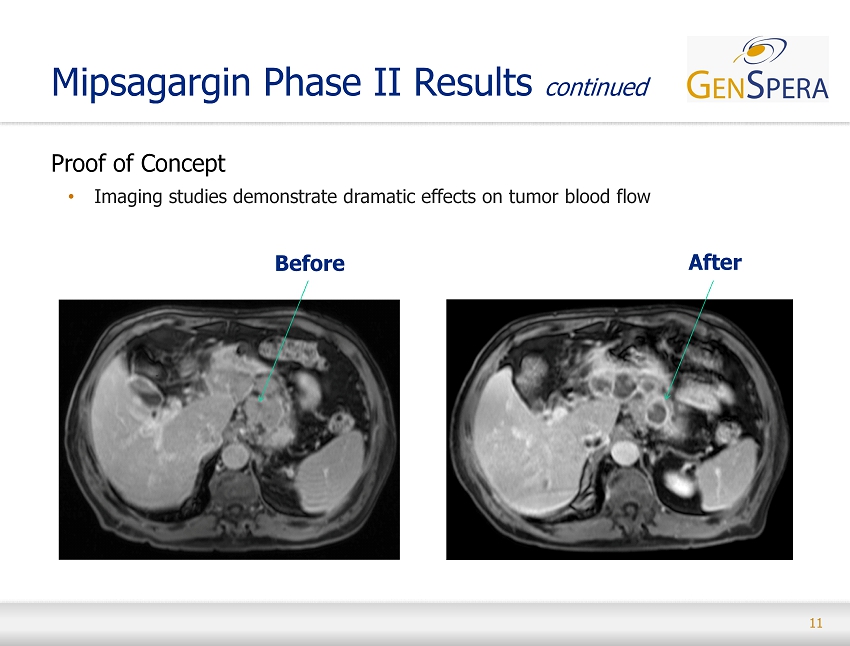
11 Proof of Concept • Imaging studies demonstrate dramatic effects on tumor blood flow Mipsagargin Phase II Results continued Before After

12 Utilize world class strategic partnerships as avenues to global commercialization • Expanded global clinical trials – heavy focus Asia/China • Manufacture at commercial scale • International development guided by focus on long - term value creation and sustainability • Collaboration with, and adoption by, leading physicians, scientists and institutions Partner Strategy
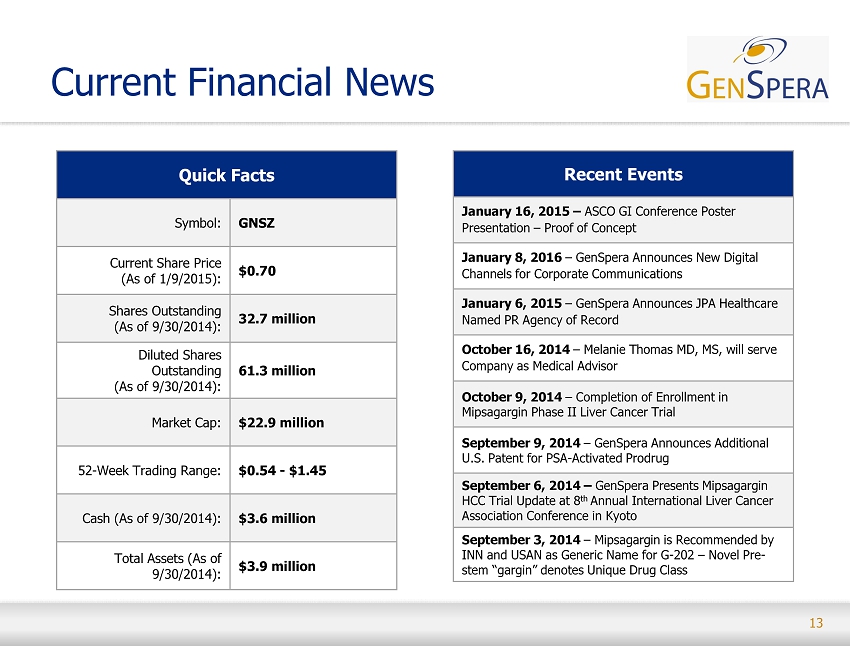
13 Current Financial News Quick Facts Symbol: GNSZ Current Share Price (As of 1/9/2015): $0.70 Shares Outstanding (As of 9/30/2014): 32.7 million Diluted Shares Outstanding (As of 9/30/2014): 61.3 million Market Cap: $22.9 million 52 - Week Trading Range : $0.54 - $1.45 Cash (As of 9/30/2014 ) : $3.6 million Total Assets (As of 9/30/2014): $3.9 million Recent Events January 16, 2015 – ASCO GI Conference Poster Presentation – Proof of Concept January 8, 2016 – GenSpera Announces New Digital Channels for Corporate Communications January 6, 2015 – GenSpera Announces JPA Healthcare Named PR Agency of Record October 16, 2014 – Melanie Thomas MD, MS, will serve Company as Medical Advisor October 9, 2014 – Completion of Enrollment in Mipsagargin Phase II Liver Cancer Trial September 9, 2014 – GenSpera Announces Additional U.S. Patent for PSA - Activated Prodrug September 6, 2014 – GenSpera Presents Mipsagargin HCC Trial Update at 8 th Annual International Liver Cancer Association Conference in Kyoto September 3, 2014 – Mipsagargin is Recommended by INN and USAN as Generic Name for G - 202 – Novel Pre - stem “ gargin ” denotes Unique Drug Class

14 Experienced Scientific and Development Team Management, Directors, Advisors Craig A. Dionne, PhD Chairman & CEO Over 25 years of experience in pharmaceutical industry, SVP Drug Discovery at Cephalon , Inc. (acquired by Teva Pharmaceuticals), EVP Prostate Cancer Foundation Russell Richerson , PhD COO Over 25 years of experience in biotechnology industry, Previously COO Molecular Profiling Institute, VP Diagnostic R&D Prometheus, Abbott Labs Peter E. Grebow , PhD Director Over 37 years of experience in pharmaceutical industry, EVP Drug Development Cephalon , Inc., Board member Optimer Pharmaceuticals, Q Holdings, Complexa . Bo Jesper Hansen, MD, PhD Director Executive Chairman of Swedish Orphan Biovitrum AB, Board member CMC AB, Orphazyme ApS , Newron , Onxeo SAS, Hyperion Therapeutics and Ablynx NV Scott Ogilvie Director President and CEO AFIN International, Inc., Board member Research Solutions, Inc., and Neuralstem , Inc. Søren Brøgger Christensen, PhD Scientific Advisor Professor at University of Copenhagen, Expert in chemistry of thapsigargin, Exploring further derivatives and manufacturing efficiencies for thapsigargin Samuel R. Denmeade , MD Chief Clinical Advisor Professor at Johns Hopkins and Board Certified Medical Oncologist, Co - Inventor of GenSpera’s technologies John T. Isaacs, PhD Chief Scientific Advisor Professor at Johns Hopkins School of Medicine, Co - Inventor of GenSpera’s technologies Melanie Thomas, MD, MS Medical Advisor Medical Director of Clinical Research, Gibbs Cancer Center and Research Institute, Principal Investigator on 29 separate clinical trials, primarily in gastrointestinal cancers.

15 Proof of Concept creates future value • Shareholders • Company options 2015 Tipping Point for growth • Additional Phase II studies • Design Phase III Liver study • World Class Partnerships GenSpera in the News • Public Relations • Social Media Channels • 3rd Party Endorsements Undervalued • $ 25mm Market Cap, comparable between $200mm and $600mm • Share price at $ 0.77 vs $3.00 or higher Summary
16 GenSpera , Inc. (OTC QB: GNSZ) 2511 N Loop 1604 W, Suite 204 San Antonio, TX 78258 Craig Dionne, PhD President, CEO and Chairman (210) 479 - 8112 info@genspera.com Investor Relations Alan Sheinwald Capital Markets Group, LLC (914) 669 - 0222 Alan@capmarketsgroup.com Connect with GenSpera https://www.facebook.com/GenSpera https://twitter.com/GenSperaNews https://www.linkedin.com/company/genspera - inc - https://plus.google.com/u/0/b/111021404725787491742/11 1021404725787491742/posts https://www.youtube.com/channel/UCdvATHFsc6ZLVgVMTJtPq3A http://www.thechairmansblog.com/genspera