
GLPG EGM & AGM| Apr 2022 Exhibit 99.3

Disclaimer This presentation contains “forward-looking statements”. When used in this presentation, the words “anticipate,” “believe,” “could,” “expect,” “intend,” “will,” “plan,” “potential,” “should,” “estimate,” “future,” “outlook,” “guidance,” “proposal,” “intention” “expected,” “on track,” “projections,” “target,” “progress,” “promise” and similar expressions are intended to identify forward-looking statements. These statements include, but are not limited to, statements regarding: the proposed one-tier board structure, the appointment of Paul Stoffels* as combined CEO/chair, and the appointment of the two three new directors and a lead non-executive director, the rate and timing of our cash burn, the progress of our refocused R&D plan and clinical development activities, our expectations as to our novel target engine, our continued execution of our savings program, our global R&D collaboration with Gilead, our collaboration with AbbVie our R&D plans and strategy, including progress on our fibrosis portfolio, oral therapeutics and SIK platform, and potential changes in such plans and strategy, our commercialization efforts for filgotinib and any future approved products, our expectations as to commercial sales, market size, and market share for Jyseleca and commercial rollout in Europe, our expectations regarding patent exclusivity for Jyseleca, our plans to build out our commercial structure for sales of Jyseleca in Europe, guidance from management regarding our financial results (including guidance regarding the expected operational use of cash during financial year 2022), expectations regarding our ability to identify and execute on business development opportunities, statements regarding the expected timing of our ongoing and planned preclinical studies and clinical trials, statements relating to interactions with regulatory authorities, statements related to the EMA’s planned safety review of JAK inhibitors used to treat certain inflammatory disorders, including filgotinib, initiated at the request of the European Commission (EC) under Article 20 of Regulation (EC) No 726/2004, statements relating to the timing or likelihood of additional regulatory authorities’ approval of marketing authorization for filgotinib, including for additional indications, statements regarding planned changes in our leadership and governance structure and expected resulting benefits, and statements relating to the timing or likelihood of pricing and reimbursement interactions for filgotinib. Any forward-looking statements in this presentation are based on management's current expectations and beliefs, and are not guarantees of future results and performance and are subject to risks, uncertainties and other factors that could cause actual events, results, financial condition and liquidity, performance, or achievements to differ materially from any historic or future results, financial conditions and liquidity, performance or achievements and, therefore, the reader should not place undue reliance on them including. These risks, uncertainties and other factors include, without limitation: risks related to the shareholders not approving the proposed one-tier board structure or any other proposal, the risk that we may not be able to realize the expected benefits from the proposed one-tier board structure, the risk that we may not be able to realize the expected benefits of the appointment of the combined chair/CEO, the lead non-executive director, or any other director, and risks related to the ongoing COVID-19 pandemic, the risk that one or more assumptions, beliefs or expectations underlying management’s guidance regarding our 2022 revenues, operating expenses, and financial results may be incorrect (including one or more of its assumptions underlying its expense expectations), the inherent uncertainties associated with competitive developments, clinical trial and product development activities and regulatory approval requirements (including the risk that data from Galapagos’ ongoing and planned clinical research programs in rheumatoid arthritis, Crohn’s disease, ulcerative colitis, idiopathic pulmonary fibrosis, osteoarthritis, other inflammatory indications and kidney disease may not support registration or further development of its product candidates due to safety or efficacy concerns or other reasons), risks related to our reliance on collaborations with third parties (including, but not limited to, our collaboration partner Gilead), risks that our commercial build-out in Europe will be delayed or less successful than anticipated, the risk that our projections and expectations regarding the commercial potential of filgotinib and any other product candidates may be inaccurate, the risk that our planned leadership transition and change to the governance structure may be disruptive to our business operations; the risk that we will be unable to successfully achieve the anticipated benefits from our planned leadership transition and change to the governance structure, the risk that we will encounter challenges retaining or attracting talent, risks related to continued regulatory review of filgotinib following approval by relevant regulatory authorities and the EMA’s planned safety review of JAK inhibitors used to treat certain inflammatory disorders, including the risk that the EMA and/or other regulatory authorities determine that additional non-clinical or clinical studies are required with respect to filgotinib, the risk that the EMA may require that the marketing authorization for filgotinib in the EU be amended, the risk that the EMA may impose JAK class-based warnings, and the risk that the EMA’s planned safety review may negatively impact acceptance of filgotinib by patents, the medical community, and healthcare payors, and the risk that regulatory authorities may require additional post-approval trials of filgotinib or any other product candidates that are approved in the future. For a discussion of other risks and uncertainties and other important factors, any of which could cause our actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in our most recent Annual Report on Form 20-F filed with the U.S. Securities and Exchange Commission (SEC), as supplemented and/or modified by any other filings and reports that we have made or will make with the SEC in the future. All information in this presentation is as of the date of the presentation, and Galapagos undertakes no duty to update this information unless required by law or regulation. Except for filgotinib's approval for the treatment of (i) RA and UC by the European Commission and Great Britain's Medicines and Healthcare Products Regulatory Agency, and of (ii) RA by Japanese Ministry of Health, Labour and Welfare, our drug candidates are investigational; their efficacy and safety have not been fully evaluated by any regulatory authority. Under no circumstances may any copy of this presentation, if obtained, be retained, copied or transmitted. *Stoffels IMC BV, permanently represented by Mr. Paul Stoffels

R&D Novel target engine & differentiated pipeline Commercial EU roll-out Jyseleca in RA & UC BD Bring in opportunities Financial guidance 2022 Jyseleca* €65-75M Cash burn €450-490M Foundations for future growth Dr. Paul Stoffels** starting as new CEO effective April 1, 2022 *Guidance on European net sales; **Stoffels IMC BV, permanently represented by Mr. Paul Stoffels

Governance changes & 2022 EGM AGM proposals

Key points in upcoming AGM/EGM Combined chair/CEO to fully leverage Paul’s leadership capabilities, incl BD Lead non-executive director Checks and balances & defined responsibilities New board appointments Paul Stoffels Jérôme Contamine Dan Baker One-tier board Strengthen decision-making process & organizational agility Amended remuneration policy Following introduction of one-tier structure

Reasons for a one-tier board A unitary board would allow Dr. Paul Stoffels to be part of the (new) board strengthen decision-making process & organizational agility allow increased information flow, greater understanding of business & strategy provide a fast approval stream to execute on strategy to drive long-term shareholder value State of business has changed materially over past 1.5 years CRL* by the FDA for our key product filgotinib, late-stage pipeline failures, management changes (CEO, CSO) *CRL: Complete Response Letter

Rationale for a combined chair/CEO role If unitary board is approved, intention to nominate Paul Stoffels as chair leverage Paul’s leadership capabilities, efficiently set & implement company direction & strategy incl BD Fully supported by current supervisory board

Dr. Paul Stoffels appointed as CEO Leadership Network icon Entrepreneurial GLPG connection Qualifications: Inspirational industry leader, strong scientific roots Track record of accelerated product development 25 innovative drugs to market Start effective April 1, 2022

Why a lead non-executive director? A strong counter-balance & support to the chair Note: For a full list of responsibilities will become available in our company CG Charter (subject to approval) and is described in letter to AGM/EGM Responsibilities: principal liaison between non-executive members & chair/CEO ensure independence of BoD from chair/CEO, executive management ensure continuity within board Authority to call meetings of non-executive directors at any time Vice-chair of BoD

Appointment of new board members *if and as long as the CEO serves as chair Paul Stoffels, MD as director, and proposal to nominate as chair fully leverage leadership capabilities efficiently set & implement company direction & strategy, incl BD Jérôme Contamine as independent director Dan Baker, MD as independent director Intention to appoint lead non-executive director as counter-balance to CEO*

Executive Remuneration Under our remuneration policy, the CEO’s cash bonus can be maximum 75% of base salary. The aggregate cash bonuses of the other members of the management board can be maximum 50% of the aggregate base salaries. An equivalent number of RSUs will be granted to the CEO and the other members of the management board under the RSU Annual Long-Term Incentive Plan Amended policy reflects shift to one-tier structure appointment of new chair/CEO Possibility for sign-on remuneration, removal of references to old grants, and plans and agreements that are no longer relevant

Transparency on pay for performance Corporate objectives published in annual report, evaluated by supervisory board and aligned with long-term interest of GLPG and SH Continuously monitor possible concerns raised by SH and amendments to our policy are made when required Company is at turning point with strategic changes expected in coming year(s) New (unitary) board to periodically review remuneration policy

Appendix

Our vision & mission Discover & develop innovative medicines that address high unmet medical needs and to improve the quality of life of patients Fully integrated, independent European biopharma Target discovery platform & pipeline Growing Jyseleca® franchise in EU Long-term Gilead collaboration €4.7B* cash & cash equivalents *At 31 Dec 2021
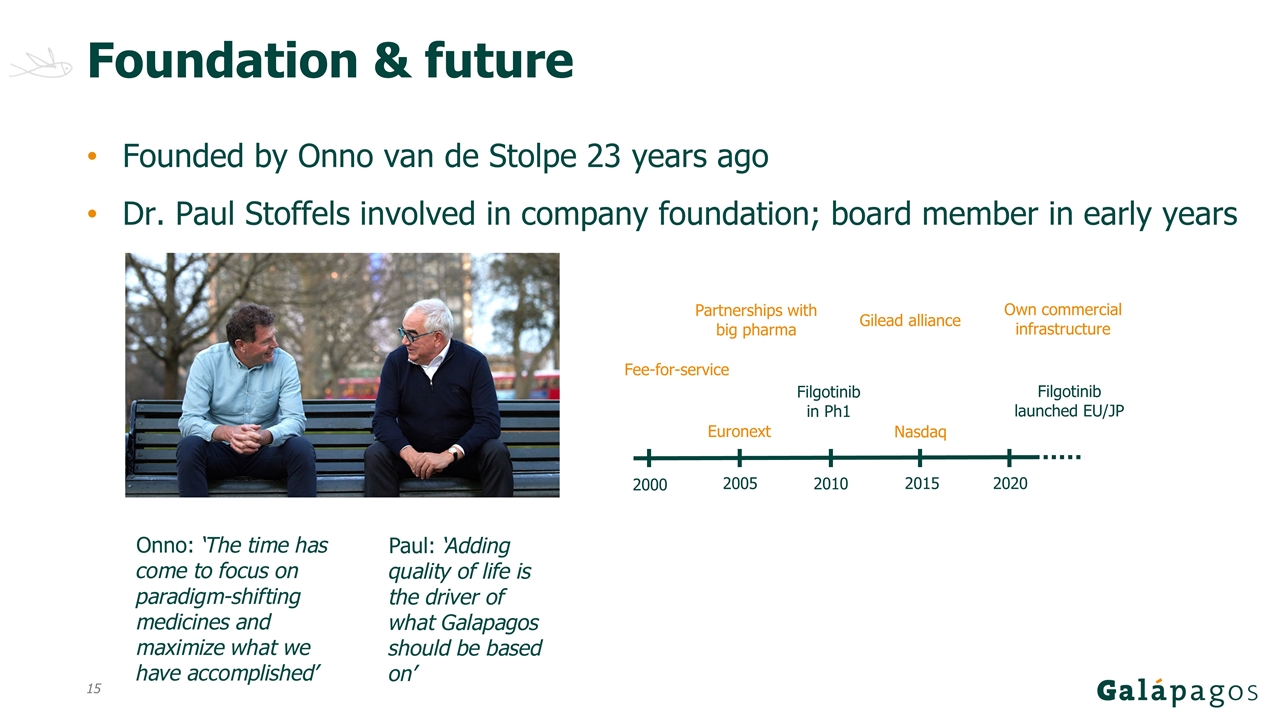
Foundation & future Founded by Onno van de Stolpe 23 years ago Dr. Paul Stoffels involved in company foundation; board member in early years Onno: ‘The time has come to focus on paradigm-shifting medicines and maximize what we have accomplished’ Paul: ‘Adding quality of life is the driver of what Galapagos should be based on’ Euronext Nasdaq Fee-for-service Partnerships with big pharma Filgotinib in Ph1 Gilead alliance Filgotinib launched EU/JP Own commercial infrastructure 2010 2005 2015 2020 2000

Current state of business
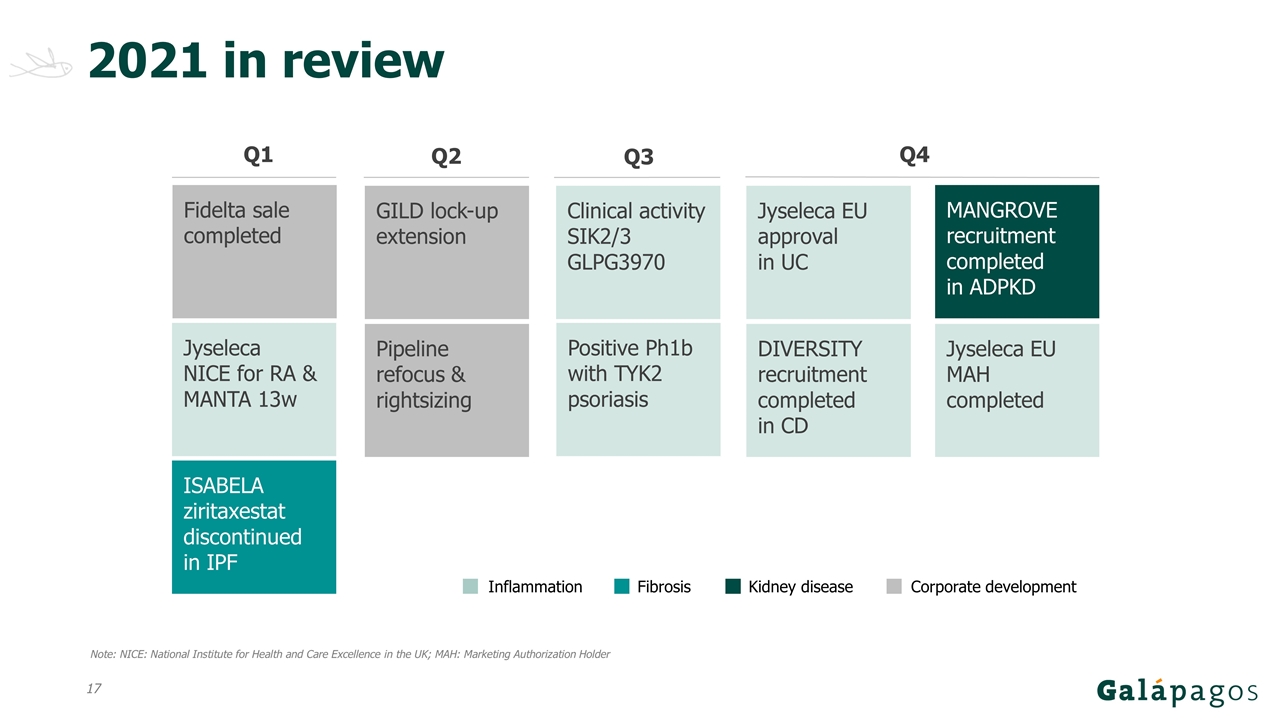
2021 in review Inflammation Fibrosis Q2 Corporate development Q3 Clinical activity SIK2/3 GLPG3970 Positive Ph1b with TYK2 psoriasis Q4 GILD lock-up extension Jyseleca EU approval in UC ISABELA ziritaxestat discontinued in IPF Q1 Fidelta sale completed Jyseleca NICE for RA & MANTA 13w Pipeline refocus & rightsizing DIVERSITY recruitment completed in CD MANGROVE recruitment completed in ADPKD Kidney disease Jyseleca EU MAH completed Note: NICE: National Institute for Health and Care Excellence in the UK; MAH: Marketing Authorization Holder

Key financials FY’21 Revenues & other income: €539 M Operating costs:- €703 M Net loss:- €103 M €236M revenue recognition for filgotinib development €43M net other financial income, gain on disposal of Fidelta €22M Flat versus FY’2020 €231M revenue recognition for the platform €15M Jyseleca sales (out of €26M total EU27 Jyseleca sales), €4M royalties
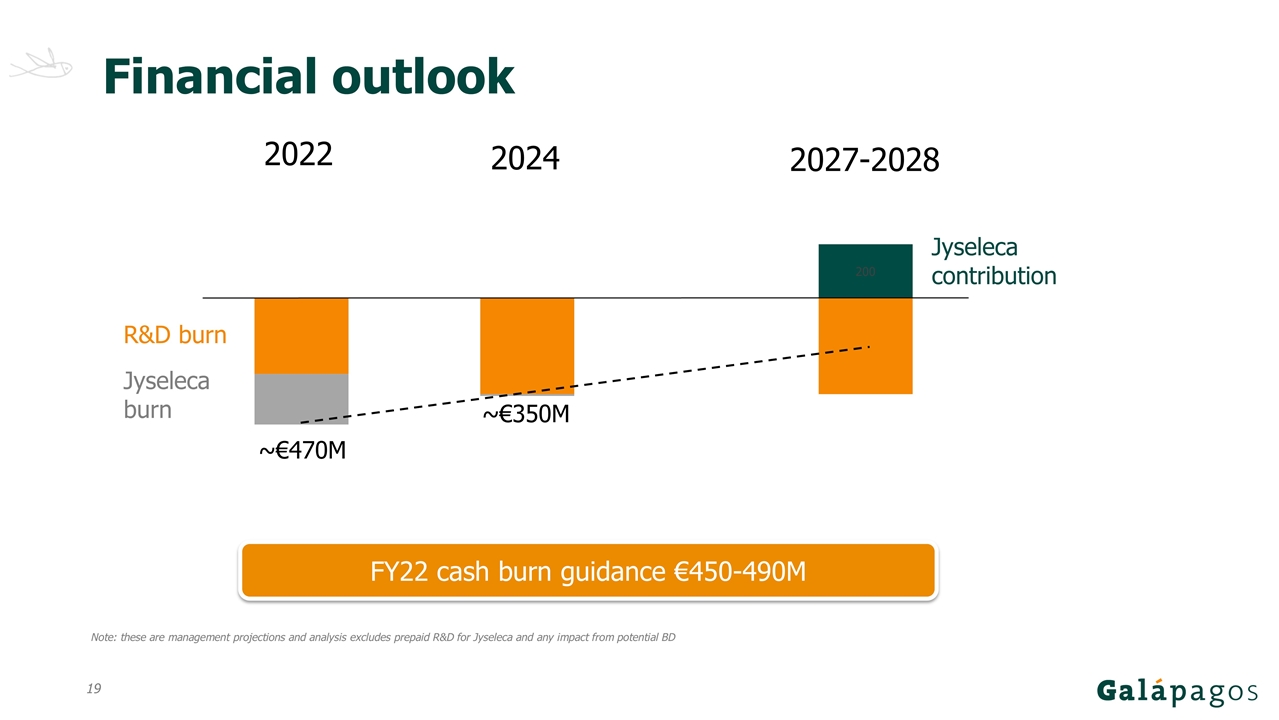
Note: these are management projections and analysis excludes prepaid R&D for Jyseleca and any impact from potential BD Jyseleca burn R&D burn Jyseleca contribution ~€470M ~€350M 2022 2024 2027-2028 Financial outlook FY22 cash burn guidance €450-490M

Legal structure & board
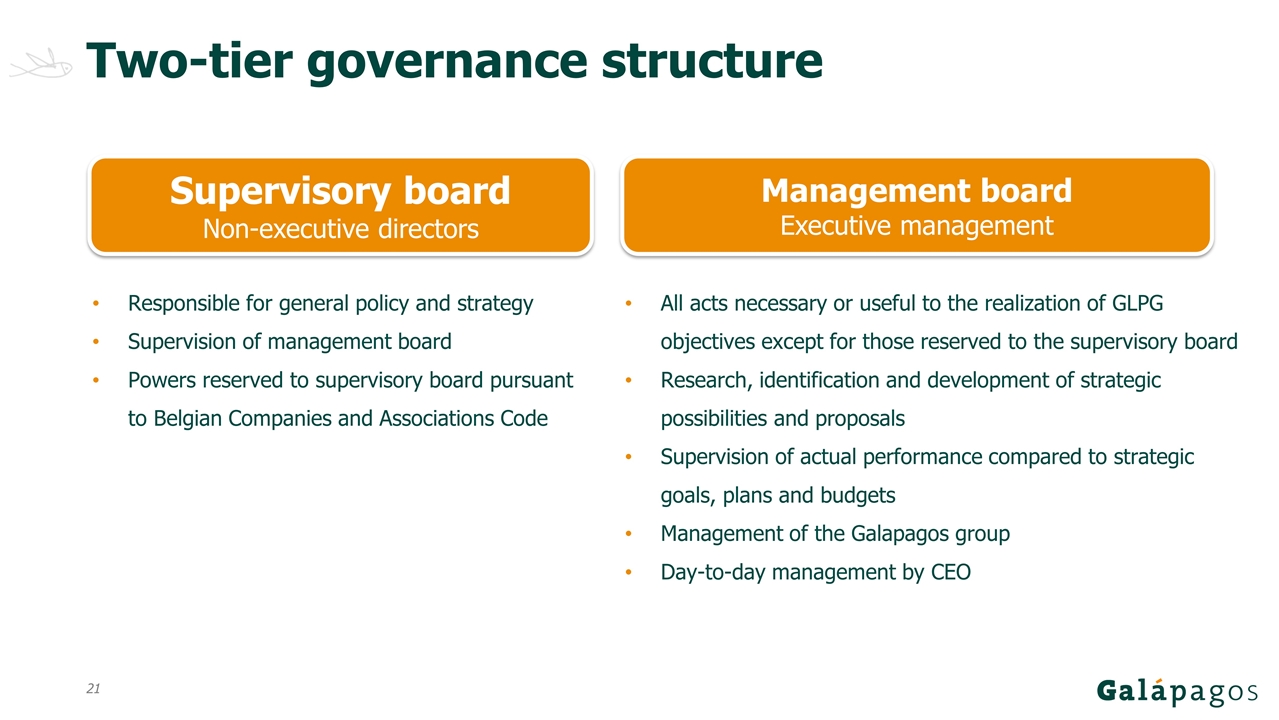
Two-tier governance structure Supervisory board Non-executive directors Management board Executive management Responsible for general policy and strategy Supervision of management board Powers reserved to supervisory board pursuant to Belgian Companies and Associations Code All acts necessary or useful to the realization of GLPG objectives except for those reserved to the supervisory board Research, identification and development of strategic possibilities and proposals Supervision of actual performance compared to strategic goals, plans and budgets Management of the Galapagos group Day-to-day management by CEO

Current composition of supervisory board 75% independence, 4 different nationalities, 50% female, median tenure of 3.5 years Supervisory board member Position Nationality Year of initial appointment Rajesh Parekh* Chairman, Independent director British 2004 Howard Rowe** Independent director British, US 2010 Katrine Bosley** Independent director US 2013 Mary Kerr Independent director British 2016 Peter Guenter Independent director Belgian 2019 Elisabeth Svanberg Independent director Swedish 2020 Daniel O’Day*** Director US 2019 Linda Higgins*** Director US 2019 *Under Nasdaq listing rules we have 2 dependent directors and 6 independent directors **Mandate expires 2022 ***In September 2021 Galapagos entered into a 10-year collaboration with Gilead and two Gilead representatives became board members based on majority shareholdership

Current composition of nomination/remuneration committee 100% independence, 3 different nationalities, 66.7% female, median tenure of 8 years Nomination/ remuneration committee member Position Nationality Year of initial appointment Rajesh Parekh* Chairman; Independent director British 2004 Katrine Bosley** Independent director US 2013 Elisabeth Svanberg Independent director Swedish 2020 *Under Nasdaq listing rules **Mandate expires 2022

Current composition of audit committee 100% independence, 3 different nationalities, 33.3% female, median tenure of 5 years Audit committee member Position Nationality Year of initial appointment Howard Rowe* Independent director British, US 2010 Mary Kerr Independent director British 2016 Peter Guenter Independent director Belgian 2019 *Mandate expires 2022

Paul Stoffels, MD Former VC of the Ex Com and CSO of J&J Set wide innovation agenda, including single-shot COVID-19 vaccine within a record time frame Prior WW Chairman Pharmaceuticals of J&J, rejuvenated its product pipeline and adopted a transformational R&D operating model, resulting in the launch of 25 innovative medicines Joined J&J in 2002, with the acquisition of Virco and Tibotec, where he was CEO and Chairman respectively, leading development of treatments of HIV Member of Galapagos board of directors from its foundation (1999) until 2002 Paul studied Medicine and Infectious Diseases & Tropical Medicine at the Institute of Tropical Medicine in Antwerp, Belgium

Jérôme Contamine CFO of Sanofi from 2009 until 2018 CFO of Veolia from 2000 to 2009 Graduate of France’s École polytechnique, ENSAE (École Nationale de la Statistique et de l’Administration Économique) and École nationale d’administration Non-executive director at Valeo from 2006 to 2017 Non-executive director on the boards of Société Générale and Total Energies

Dan Baker, MD Joined Janssen/Centocor in 2000 – 2019; as VP Immunology R&D developed Remicade, Simponi, Stelara, and other rheumatology and dermatology programs Supervised trials in multiple disease areas, and oversaw >15 regulatory approvals in the US, Europe and Japan Responsible for evaluating BD opportunities in immunology Recently raised capital to fund and start an immunology company, KiRA Biotech, where he now acts as CEO and executive director B.A. in Biology from Gettysburg College and medical degree from the University of Pennsylvania