Filed by Bristol-Myers Squibb Company
Pursuant to Rule 425 of the Securities Act of 1933
and deemed filed pursuant to Rule 14a-12
of the Securities Exchange Act of 1934
Subject Company: Celgene Corporation
Commission File No.: 001-34912
Explanatory Note: The following slides were provided to employees of Bristol-Myers Squibb Company on January 17, 2019.

Important Information for Investors and Stockholders This communication does not constitute an offer to sell or the solicitation of an offer to buy any securities or a solicitation of any vote or approval. It does not constitute a prospectus or prospectus equivalent document. No offering of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the U.S. Securities Act of 1933, as amended.In connection with the proposed transaction between Bristol-Myers Squibb Company (“Bristol-Myers Squibb”) and Celgene Corporation (“Celgene”), Bristol-Myers Squibb and Celgene will file relevant materials with the Securities and Exchange Commission (the “SEC”), including a Bristol-Myers Squibb registration statement on Form S-4 that will include a joint proxy statement of Bristol-Myers Squibb and Celgene that also constitutes a prospectus of Bristol-Myers Squibb, and a definitive joint proxy statement/prospectus will be mailed to stockholders of Bristol-Myers Squibb and Celgene. INVESTORS AND SECURITY HOLDERS OF BRISTOL-MYERS SQUIBB AND CELGENE ARE URGED TO READ THE JOINT PROXY STATEMENT/PROSPECTUS AND OTHER DOCUMENTS THAT WILL BE FILED WITH THE SEC CAREFULLY AND IN THEIR ENTIRETY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION. Investors and security holders will be able to obtain free copies of the registration statement and the joint proxy statement/prospectus (when available) and other documents filed with the SEC by Bristol-Myers Squibb or Celgene through the website maintained by the SEC at http://www.sec.gov. Copies of the documents filed with the SEC by Bristol-Myers Squibb will be available free of charge on Bristol-Myers Squibb’s internet website at https://www.bms.com/ under the tab, “Investors” and under the heading “Financial Reporting” and subheading “SEC Filings” or by contacting Bristol-Myers Squibb’s Investor Relations Department through https://www.bms.com/investors/investor-contacts.html. Copies of the documents filed with the SEC by Celgene will be available free of charge on Celgene’s internet website at https://www.celgene.com/ under the tab “Investors” and under the heading “Financial Information” and subheading “SEC Filings” or by contacting Celgene’s Investor Relations Department at ir@celgene.com.Certain Information Regarding ParticipantsBristol-Myers Squibb, Celgene, and their respective directors and executive officers may be considered participants in the solicitation of proxies in connection with the proposed transaction. Information about the directors and executive officers of Bristol-Myers Squibb is set forth in its Annual Report on Form 10-K for the year ended December 31, 2017, which was filed with the SEC on February 13, 2018, its proxy statement for its 2018 annual meeting of stockholders, which was filed with the SEC on March 22, 2018, and its Current Report on Form 8-K, which was filed with the SEC on August 28, 2018. Information about the directors and executive officers of Celgene is set forth in its Annual Report on Form 10-K for the year ended December 31, 2017, which was filed with the SEC on February 7, 2018, its proxy statement for its 2018 annual meeting of stockholders, which was filed with the SEC on April 30, 2018, and its Current Reports on Form 8-K, which were filed with the SEC on June 1, 2018, June 19, 2018 and November 2, 2018. Other information regarding the participants in the proxy solicitations and a description of their direct and indirect interests, by security holdings or otherwise, will be contained in the joint proxy statement/prospectus and other relevant materials to be filed with the SEC regarding the proposed transaction when they become available. You may obtain these documents (when they become available) free of charge through the website maintained by the SEC at http://www.sec.gov and from Investor Relations at Bristol-Myers Squibb or Celgene as described above.

Cautionary Statement Regarding Forward Looking Statements This communication contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. You can generally identify forward-looking statements by the use of forward-looking terminology such as “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “explore,” “evaluate,” “intend,” “may,” “might,” “plan,” “potential,” “predict,” “project,” “seek,” “should,” or “will,” or the negative thereof or other variations thereon or comparable terminology. These forward-looking statements are only predictions and involve known and unknown risks and uncertainties, many of which are beyond Bristol-Myers Squibb’s and Celgene’s control. Statements in this communication regarding Bristol-Myers Squibb, Celgene and the combined company that are forward-looking, including projections as to the anticipated benefits of the proposed transaction, the impact of the proposed transaction on Bristol-Myers Squibb’s and Celgene’s business and future financial and operating results, the amount and timing of synergies from the proposed transaction, the terms and scope of the expected financing for the proposed transaction, the aggregate amount of indebtedness of the combined company following the closing of the proposed transaction, expectations regarding cash flow generation, accretion to non-GAAP earnings per share, capital structure, debt repayment, adjusted leverage ratio and credit ratings following the closing of the proposed transaction, Bristol-Myers Squibb’s ability and intent to conduct a share repurchase program and declare future dividend payments, the combined company’s pipeline, intellectual property protection and R&D spend, the timing and probability of a payment pursuant to the contingent value right consideration, and the closing date for the proposed transaction, are based on management’s estimates, assumptions and projections, and are subject to significant uncertainties and other factors, many of which are beyond Bristol-Myers Squibb’s and Celgene’s control. These factors include, among other things, effects of the continuing implementation of governmental laws and regulations related to Medicare, Medicaid, Medicaid managed care organizations and entities under the Public Health Service 340B program, pharmaceutical rebates and reimbursement, market factors, competitive product development and approvals, pricing controls and pressures (including changes in rules and practices of managed care groups and institutional and governmental purchasers), economic conditions such as interest rate and currency exchange rate fluctuations, judicial decisions, claims and concerns that may arise regarding the safety and efficacy of in-line products and product candidates, changes to wholesaler inventory levels, variability in data provided by third parties, changes in, and interpretation of, governmental regulations and legislation affecting domestic or foreign operations, including tax obligations, changes to business or tax planning strategies, difficulties and delays in product development, manufacturing or sales including any potential future recalls, patent positions and the ultimate outcome of any litigation matter. These factors also include the combined company’s ability to execute successfully its strategic plans, including its business development strategy, the expiration of patents or data protection on certain products, including assumptions about the combined company’s ability to retain patent exclusivity of certain products, the impact and result of governmental investigations, the combined company’s ability to obtain necessary regulatory approvals or obtaining these without delay, the risk that the combined company’s products prove to be commercially successful or that contractual milestones will be achieved. Similarly, there are uncertainties relating to a number of other important factors, including: results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; the content and timing of decisions made by the U.S. FDA and other regulatory authorities, investigational review boards at clinical trial sites and publication review bodies; the ability to enroll patients in planned clinical trials; unplanned cash requirements and expenditures; competitive factors; the ability to obtain, maintain and enforce patent and other intellectual property protection for any product candidates; the ability to maintain key collaborations; and general economic and market conditions. Additional information concerning these risks, uncertainties and assumptions can be found in Bristol-Myers Squibb’s and Celgene’s respective filings with the SEC, including the risk factors discussed in Bristol-Myers Squibb’s and Celgene’s most recent Annual Reports on Form 10-K, as updated by their Quarterly Reports on Form 10-Q and future filings with the SEC. It should also be noted that projected financial information for the combined businesses of Bristol-Myers Squibb and Celgene is based on management’s estimates, assumptions and projections and has not been prepared in conformance with the applicable accounting requirements of Regulation S-X relating to pro forma financial information, and the required pro forma adjustments have not been applied and are not reflected therein. None of this information should be considered in isolation from, or as a substitute for, the historical financial statements of Bristol-Myers Squibb or Celgene. Important risk factors could cause actual future results and other future events to differ materially from those currently estimated by management, including, but not limited to, the risks that: a condition to the closing of the proposed acquisition may not be satisfied; a regulatory approval that may be required for the proposed acquisition is delayed, is not obtained or is obtained subject to conditions that are not anticipated; Bristol-Myers Squibb is unable to achieve the synergies and value creation contemplated by the proposed acquisition; Bristol-Myers Squibb is unable to promptly and effectively integrate Celgene’s businesses; management’s time and attention is diverted on transaction-related issues; disruption from the transaction makes it more difficult to maintain business, contractual and operational relationships; the credit ratings of the combined company declines following the proposed acquisition; legal proceedings are instituted against Bristol-Myers Squibb, Celgene or the combined company; Bristol-Myers Squibb, Celgene or the combined company is unable to retain key personnel; and the announcement or the consummation of the proposed acquisition has a negative effect on the market price of the capital stock of Bristol-Myers Squibb and Celgene or on Bristol-Myers Squibb’s and Celgene’s operating results. No assurances can be given that any of the events anticipated by the forward-looking statements will transpire or occur, or if any of them do occur, what impact they will have on the results of operations, financial condition or cash flows of Bristol-Myers Squibb or Celgene. Should any risks and uncertainties develop into actual events, these developments could have a material adverse effect on the proposed transaction and/or Bristol-Myers Squibb or Celgene, Bristol-Myers Squibb’s ability to successfully complete the proposed transaction and/or realize the expected benefits from the proposed transaction. You are cautioned not to rely on Bristol-Myers Squibb’s and Celgene’s forward-looking statements. These forward-looking statements are and will be based upon management’s then-current views and assumptions regarding future events and operating performance, and are applicable only as of the dates of such statements. Neither Bristol-Myers Squibb nor Celgene assumes any duty to update or revise forward-looking statements, whether as a result of new information, future events or otherwise, as of any future date.

Who Is Celgene? Celgene Mission and Vision

Celgene is a Global Biotech Based in Summit, NJ > 8,500 Employees* ~70 CountriesWhere Products are Sold �� Total U.S. locations: 20 Total Ex-U.S. locations: 43 EU: 30APAC: 10Latin America: 2Canada: 1 Function: Manufacturing R&D Commercial / enabling ~2,200R&D ~3,000Commercial ~2,100Manufacturing ~1,200Enabling Functions

Celgene’s History Is Marked By Bold & Courageous Choices 1960 2000 2010 1990 1961: Thalidomide tragedy 1986: Celgene spin off from 1987: IPO ~1990-95: Academic discovery of new therapeutic uses for thalidomide 1999: Thalidomide active in myeloma (NEJM) 1998: approval 2005: approval 2011: Cereblon ID’d target of IMiDs 2012-13: Key approvals 2014: approval Scientific milestones Corporate milestones 2002: Publication of more potent derivatives of thalidomide
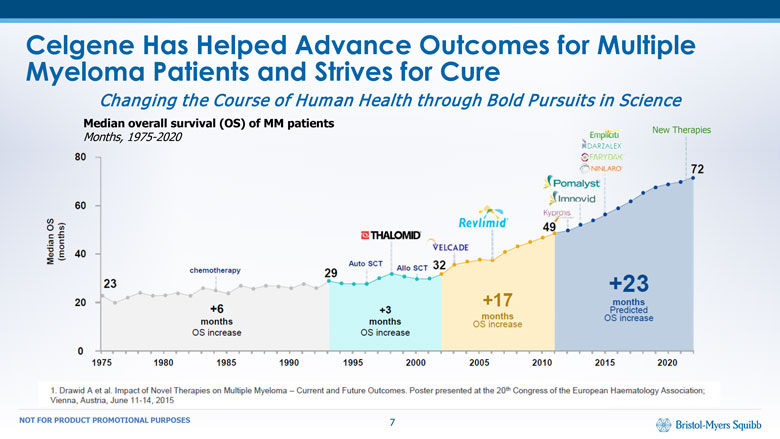
Celgene Has Helped Advance Outcomes for Multiple Myeloma Patients and Strives for Cure Median overall survival (OS) of MM patientsMonths, 1975-2020 New Therapies Changing the Course of Human Health through Bold Pursuits in Science
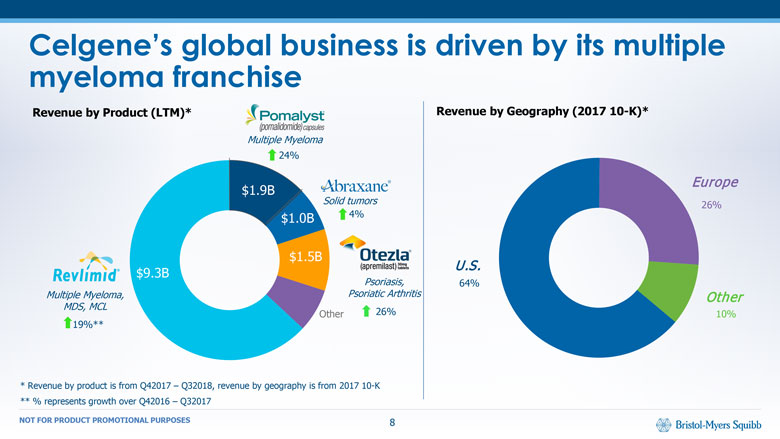
Psoriasis,Psoriatic Arthritis Celgene’s global business is driven by its multiple myeloma franchise Other 19%** 24% Multiple Myeloma Solid tumors 4% 26% Revenue by Product (LTM)* Revenue by Geography (2017 10-K)* U.S. 64% Europe 26% Other 10% * Revenue by product is from Q42017 – Q32018, revenue by geography is from 2017 10-K ** % represents growth over Q42016 – Q32017 $9.3B $1.0B $1.9B $1.5B Multiple Myeloma,MDS, MCL

Celgene’s Near-term Pipeline Includes Five Highly Promising Assets luspatercept liso-cel (JCAR017) bb2121 fedratinib ozanimod Biologic Myelodysplastic syndrome, Beta-thalassemia Cell therapyDiffuse large B-cell lymphoma, Chronic lymphocytic leukemia Cell therapy Multiple myeloma Small moleculeMyelofibrosis Small moleculeRelapsing-remitting multiple sclerosis, Ulcerative colitis, Crohn’s disease Oncology: Hematology I&I Est. 2019 launch Est. 2020 launch Est. 2020 launch Est. 2019 launch Est. 2020 launch
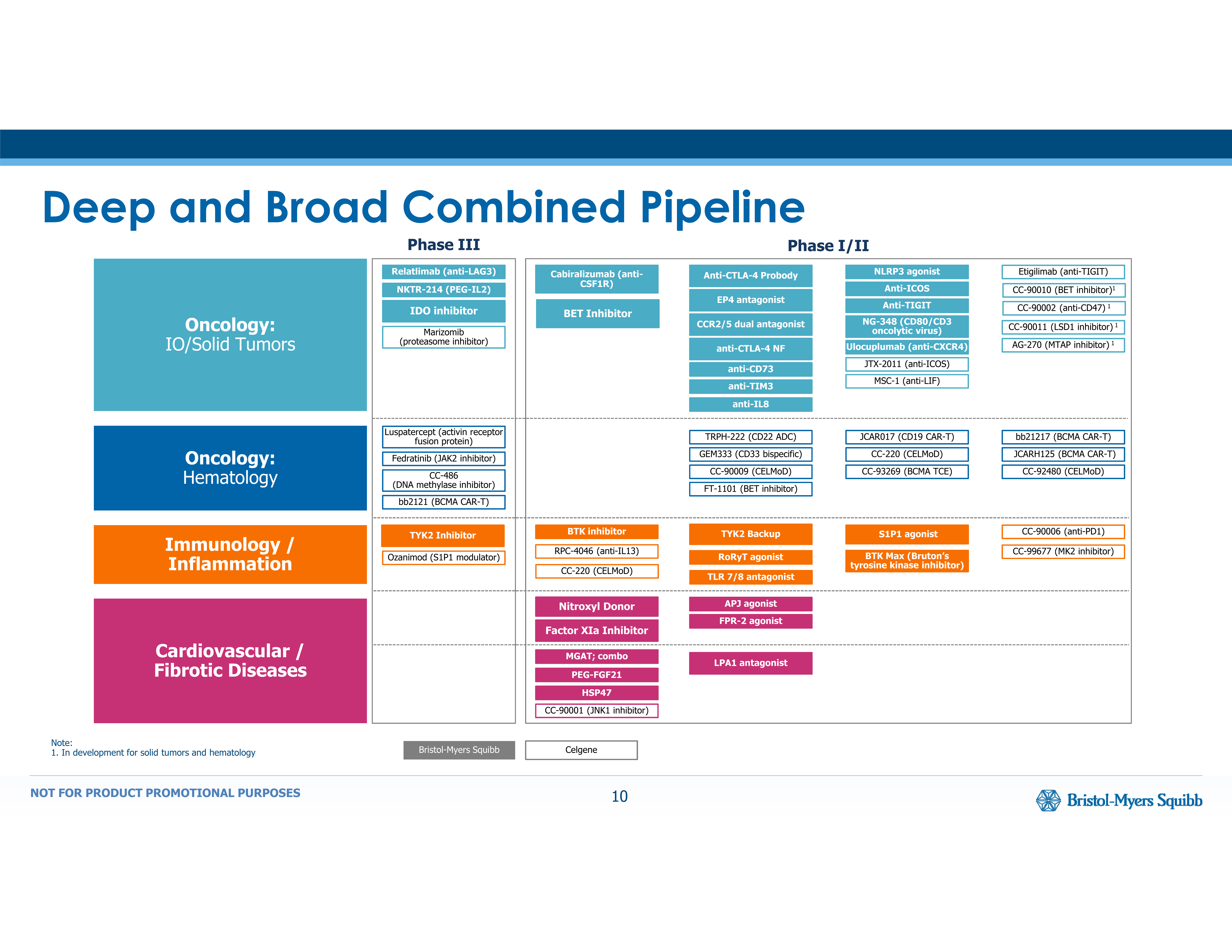
Deep and Broad Combined Pipeline Oncology: IO/Solid Tumors Relatlimab (anti-LAG3) Luspatercept (activin receptor fusion protein) Ozanimod (S1P1 modulator) CC-486 (DNA methylase inhibitor) Fedratinib (JAK2 inhibitor) Cabiralizumab (anti-CSF1R) Marizomib (proteasome inhibitor) TYK2 Inhibitor Nitroxyl Donor NKTR-214 (PEG-IL2) bb2121 (BCMA CAR-T) RPC-4046 (anti-IL13) Factor XIa Inhibitor HSP47 CC-90001 (JNK1 inhibitor) PEG-FGF21 CC-220 (CELMoD) EP4 antagonist Anti-CTLA-4 Probody S1P1 agonist APJ agonist CCR2/5 dual antagonist anti-CTLA-4 NF CC-90011 (LSD1 inhibitor) 1 TRPH-222 (CD22 ADC) BTK Max (Bruton’s tyrosine kinase inhibitor) anti-CD73 anti-TIM3 FPR-2 agonist LPA1 antagonist CC-90009 (CELMoD) GEM333 (CD33 bispecific) NLRP3 agonist bb21217 (BCMA CAR-T) CC-90010 (BET inhibitor)1 CC-93269 (BCMA TCE) JCARH125 (BCMA CAR-T) CC-92480 (CELMoD) CC-90002 (anti-CD47) 1 JCAR017 (CD19 CAR-T) anti-IL8 NG-348 (CD80/CD3 oncolytic virus) CC-220 (CELMoD) Phase III Phase I/II FT-1101 (BET inhibitor) CC-99677 (MK2 inhibitor) CC-90006 (anti-PD1) AG-270 (MTAP inhibitor) 1 MSC-1 (anti-LIF) Etigilimab (anti-TIGIT) JTX-2011 (anti-ICOS) Oncology: Hematology Immunology / Inflammation Cardiovascular / Fibrotic Diseases MGAT; combo Anti-ICOS Anti-TIGIT Ulocuplumab (anti-CXCR4) BTK inhibitor RoRyT agonist TLR 7/8 antagonist TYK2 Backup IDO inhibitor BET Inhibitor Bristol-Myers Squibb Celgene Note:1. In development for solid tumors and hematology

Celgene R&D Strengths Are Amplified Through Active External Partnerships Immuno-Oncology/Cell Therapy Protein Homeostasis Epigenetics Inflammation & Immunology Neuroscience & Imaging Platform Technologies Academics & IKU 50+ programs21 unique compounds in clinical development20+ IND Files
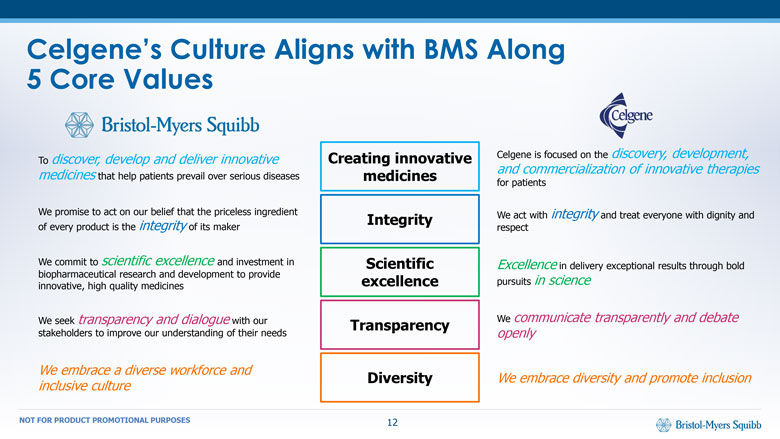
Celgene’s Culture Aligns with BMS Along5 Core Values Creating innovative medicines To discover, develop and deliver innovative medicines that help patients prevail over serious diseases Celgene is focused on the discovery, development, and commercialization of innovative therapies for patients Integrity We promise to act on our belief that the priceless ingredient of every product is the integrity of its maker We act with integrity and treat everyone with dignity and respect Scientific excellence We commit to scientific excellence and investment in biopharmaceutical research and development to provide innovative, high quality medicines Excellence in delivery exceptional results through bold pursuits in science Transparency We seek transparency and dialogue with our stakeholders to improve our understanding of their needs We communicate transparently and debate openly Diversity We embrace a diverse workforce and inclusive culture We embrace diversity and promote inclusion