Exhibit 99.2

Bristol Myers Squibb to Acquire MyoKardia October 5, 2020

Additional Information and Where to Find It The tender offer described in this report has not yet commenced, and this communication is neither an offer to purchase nor a solicitation of an offer to sell securities. At the time the tender offer is commenced, Bristol Myers Squibb will cause Merger Sub to file with the U.S. Securities and Exchange Commission (“SEC”) a tender offer statement on Schedule TO. Investors and MyoKardia stockholders are strongly advised to read the tender offer statement (including an offer to purchase, letter of transmittal and related tender offer documents) and the related solicitation/recommendation statement on Schedule 14D-9 that will be filed by MyoKardia with the SEC, because they will contain important information. These documents will be available at no charge on the SEC’s website at www.sec.gov. In addition, a copy of the offer to purchase, letter of transmittal and certain other related tender offer documents (once they become available) may be obtained free of charge at www.bms.com or by directing a request to Bristol Myers Squibb, Office of the Corporate Secretary, 430 East 29th Street, 14th Floor, New York, New York 10154-0037. A copy of the tender offer statement and the solicitation/recommendation statement will be made available to all stockholders of MyoKardia free of charge at www.myokardia.com or by contacting MyoKardia at ir@myokardia.com, telephone number 650-351-4690. In addition to the offer to purchase, the related letter of transmittal and certain other offer documents, as well as the solicitation/recommendation statement, Bristol Myers Squibb and MyoKardia file annual, quarterly and special reports, proxy statements and other information with the SEC. You may read and copy any reports, statements or other information filed by Bristol Myers Squibb or MyoKardia at the SEC public reference room at 100 F Street, N.E., Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information on the public reference room. Bristol Myers Squibb’s and MyoKardia’s filings with the SEC are also available to the public from commercial document-retrieval services and at the website maintained by the SEC at www.sec.gov. Forward Looking Statements This report contains “forward-looking statements” relating to the acquisition of MyoKardia by Bristol Myers Squibb and the development and commercialization of certain biological compounds. Such forward-looking statements are based on current expectations and involve inherent risks and uncertainties, including factors that could delay, divert or change any of them, and could cause actual outcomes and results to differ materially from current expectations. No forward-looking statement can be guaranteed. Among other risks, there can be no guarantee that the acquisition will be completed, or if it is completed, that it will close within the anticipated time period or that the expected benefits of the acquisition will be realized. The actual financial impact of this transaction may differ from the expected financial impact described in this report. In addition, the compounds described in this report are subject to all the risks inherent in the drug development process, and there can be no assurance that the development of these compounds will be commercially successful. Forward-looking statements in this report should be evaluated together with the many uncertainties that affect Bristol Myers Squibb’s business, particularly those identified in the cautionary factors discussion in Bristol Myers Squibb’s Annual Report on Form 10-K for the year ended December 31, 2019, and its subsequent Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. Bristol Myers Squibb undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events, or otherwise. Use of Non-GAAP Financial Information and Financial GuidanceThis earnings release contains non-GAAP financial guidance, which is adjusted to exclude certain costs, expenses, gains and losses and other specified items that are evaluated on an individual basis. These non-GAAP items are adjusted after considering their quantitative and qualitative aspects and typically have one or more of the following characteristics, such as being highly variable, difficult to project, unusual in nature, significant to the results of a particular period or not indicative of future operating results. Similar charges or gains were recognized in prior periods and will likely reoccur in future periods. Non-GAAP information is intended to portray the results of the company’s baseline performance, supplement or enhance management, analysts and investors overall understanding of the company’s underlying financial performance and facilitate comparisons among current, past and future periods. For example, non-GAAP earnings and EPS information are indications of the company’s baseline performance before items that are considered by us to not be reflective of the company’s ongoing results. In addition, this information is among the primary indicators that we use as a basis for evaluating performance, allocating resources, setting incentive compensation targets and planning and forecasting for future periods. This information is not intended to be considered in isolation or as a substitute for net earnings or diluted EPS prepared in accordance with GAAP and may not be the same as or comparable to similarly titled measures presented by other companies due to possible differences in method and in the items being adjusted. We encourage investors to review our financial statements and publicly-filed reports in their entirety and not to rely on any single financial measure. There is no reliable or reasonably estimable comparable GAAP measure for this non-GAAP financial guidance because we are not able to reliably predict the impact of specified items beyond 2020. As a result, reconciliation of this non-GAAP measure to the most directly comparable GAAP measure is not available without unreasonable effort. In addition, the company believes such a reconciliation would imply a degree of precision and certainty that could be confusing to investors. The variability of the specified items may have a significant and unpredictable impact on our future GAAP results. In addition, the non-GAAP financial guidance in this release excludes the impact of any potential additional future strategic acquisitions and divestitures and any specified items that have not yet been identified and quantified. The guidance also excludes macro-economic effects due to the COVID-19 pandemic that are not yet quantifiable. The financial guidance is subject to risks and uncertainties applicable to all forward-looking statements as described elsewhere in this press release. 2

Agreement toacquire MyoKardiafor:$13.1B$225/share in cash Bristol Myers Squibbto gain access to:MavacamtenFirst-in-class specialty CV medicinewith significant commercial potential CV pipeline and discovery capabilities Significantmedium- and long-term growth opportunity; accretive to revenue and Non-GAAP EPS starting in 2023 All-cash transactionvia tender offer expected to closein Q4 2020 subject to customary reviews Transaction overview 3 Significant opportunity for value creation for BMS

We are well positioned to advance our strategy Pipeline New product approvals: Reblozyl, Zeposia, Onureg (CC-486)Multiple BLAs/NDAs in progress: liso-cel, ide-cel Two 1L lung approvals: Checkmate 227 and Checkmate 9LADelivered positive results on key clinical trials: Zeposia in UC, Checkmate 9ER, Checkmate 743, Checkmate 649, Checkmate 577, Checkmate 274 CommercialStrong commercial execution, delivering continued topline growth IntegrationActivities proceeding well, synergies on track FinancialFinancial strength and P&L discipline 4

Consistent approach to sustaining innovation and renewing our portfolio 5 Research & DevelopmentWorld class talent & approachProprietary datasets & platformsRobust pipeline Business DevelopmentA top priority for capital allocationConsistent evaluation criteriaEnabled by financial strength & flexibility 17 marketed medicines, many have opportunities for additional indications 7 assets in registrational development>50 assets in Ph I or Ph II trials

MyoKardia: Innovative biotech focused on specialty CV 6 mavacamten: A first-in-class myosin inhibitor De-risked lead-indicationPositive Ph3 (EXPLORER) trial for obstructive Hypertrophic Cardiomyopathy (HCM) with strong efficacy and good tolerability profileReceived Breakthrough Therapy Designation inJuly 2020; on track to be filed with FDA in Q1 2021Additional indications e.g. non-obstructive HCM ~300 employees with significant expertise in cardiovascular Located inSan Francisco Bay Area Complementary patient-centric cultures Obstructive HCM (completed) P1 P3 Non-obstructive HCM Precision diastolic disease (mavacamten HFpEF) HCM Systolic dysfunction and atrial fibrillation Genetic dilated cardiomyopathy (DCM) P2 LUS-1 ACT-1 Precision diastolic diseases (preclinical) Precision systolic diseases (preclinical) MYK-224 danicamtiv mavacamten PIPELINE

FXIa inhibitor Transaction accelerates expansion of leading CV franchise 7 *Assuming District Court ruling upheld upon appeal, no other generics overturn current patents; VTEp: Venous Thromboembolism prevention; TKR: Total Knee Replacement; SSP: Secondary Stroke Prevention Best-in-class medicine with strengthened IP positionPotential additional exclusivity post 2026 Fully-owned growth driver with potential 2021 launchBMS commercial capabilities are well positioned to realize the full value of the opportunity Potential next-generation antithromboticPh2 data in VTEp TKR study and SSP study expected next year and 2022 Eliquis mavacamten

Hypertrophic cardiomyopathy (HCM) disease profile 8 Normal Heart Hypertrophic Heart Decreased left ventricular volume Thickened heart muscle and septum LVOT1 obstruction Diagnosed by echo-cardiogram Subset of patients have severe symptoms Typical age of diagnosis in the ~40s-50s HCM Pathophysiology Enhanced cardiac actin-myosin interactionsHypercontractility and thickening of the heart muscle HCM Prevalence ~1 in 500 people (total population)Most common genetic heart disease Classification Obstructive HCM: mechanical obstruction of blood flow out of left ventricle (~2/3 of patients)Non-obstructive HCM: ~1/3 patients ~25% of obstructiveHCM and ~10% of non-obstructive HCM patients are symptomatic and diagnosed 1. LVOT = Left ventricular outflow tractSource: Olivotto. Lancet. 2020; Maron. NEJM. 2018; Marian. Circ Res. 2017; Maron. J Am Coll Cardiol. 2016; Veselka. Lancet. 2016; Maron. J Am Coll Cardiol. 2015; Ahmad. Annu Rev Genomic Hum Genet. 2005; Maron. JAMA. 1999; Maron. Circulation. 1995.

Significant unmet need for patients with symptomatic obstructive HCM 9 Symptoms have a meaningful negative impact on quality of lifeComplications can be severe, including:heart failuremitral valve prolapse and regurgitationatrial fibrillationsudden cardiac death Source: Maron. NEJM. 2018; Geske. JJAC Heart Fail. 2018; Marian. Circ Res. 2017; Maron. Am J Cardiol. 2016; Veselka. Lancet. 2016. Maron. Circulation. 1995. Patients with Symptomatic Obstructive HCM 80–100K U.S. 80–100KEU Currently no approved medicines that address underlying disease Current management is limited to symptomatic relief, compensation via lifestyle changes, or invasive proceduresMedical therapy: non-specific and only offers symptomatic improvementSurgical therapy: typically for patients with severe obstructive HCM and include septal or apical myectomy, septal ablation

Mavacamten: a potential first-in-class medicine that could treat underlying disease 10 Source: Olivotto. Lancet. 2020. Normal Sarcomere HCMSarcomere HCM Sarcomere with mavacamten Actin thin filament Actin-myosin cross-bridge Myosin thick filament First-in-class Myosin ATPase inhibitor Mavacamten’s mechanism of action is specific to cardiac muscleInhibition of myosin cross-bridge formation has a direct impact on underlying disease Reduction of cross-bridges inhibits excessive contractility and hypertrophic stimulus mavacamten

Mavacamten: EXPLORER-HCM Study Design 11 1Ho CY et al. Circ Heart Fail. 2020; 13(6):e006853 Pivotal Phase 3, Multicenter, Randomized, Double-blind, Placebo-controlled Trial in Patients With Obstructive HCM1 NYHA Class II/IIIPeak LVOT gradient ≥ 50mmHgLVEF ≥ 55%Background medications allowed Primary endpoint: functional composite of either (1) ≥ 1.5-mL/kg/min improvement in peak VO2 plus improvement of > 1 NYHA functional class, or (2) ≥ 3.0-mL/kg/min improvement of peak VO2 with no worsening in NYHA functional class at week 30Secondary endpoints: post-exercise LVOT gradient, peak VO2, NYHA class, and quality of life and shortness of breath scores Inclusion criteria N=251 Screening Randomization Washout mavacamten placebo 5mg QD 10mg QD5mg QD2.5mg QD 15mg QD10mg QD5mg QD2.5mg QD -W4 D1 W1 W2 W6 W12 W13 W14 W28 W30 W38 W7 W8 DOSE ADJUSTMENT DOSE ASSESSMENT

Mavacamten: Compelling Ph3 efficacy profile in symptomatic obstructive HCM 12 [Key Data] [Key Data] Mavacamten group (n=123) Placebo group (n=128) Difference* (95% CI),p value Primary endpoint† Either ≥1.5 mL/kg per min increase in pVO2 with ≥1 NYHA class improvement or ≥3.0 mL/kg per min increase in pVO2 with no worsening of NYHA class 45 (37%) 22 (17%) 19.4 (8.7 to 30.1; p=0.0005) Both ≥3.0 mL/kg per min increase in pVO2 and ≥1 NYHA class improvement 25 (20%) 10 (8%) 12.5 (4.0 to 21.0) Secondary endpoints‡ Post-exercise LVOT gradient change from baseline to week 30, mm Hg –47 (40), n=117 –10 (30), n=122 –35.6 (–43.2 to –28.1; p<0.0001) pVO2 change from baseline to week 30, mL/kg per min 1.4 (3.1), n=120 –0.1 (3.0), n=125 1.4 (0.6 to 2.1; p=0.0006) ≥1 NYHA class improvement from baseline to week 30§ 80 (65%) 40 (31%) 34% (22 to 45; p<0.0001) Change from baseline to week 30 in KCCQ-CSS§ (QoL) 13.6 (14.4), n=92 4.2 (13.7), n=88 9.1 (5.5 to 12.7; p<0.0001) Change from baseline to week 30 in HCMSQ-SoB§ (QoL) -2.8 (2.7), n=85 –0.9 (2.4), n=86 –1.8 (–2.4 to –1.2; p<0.0001) Exploratory endpoint Complete response, n/N (%) Defined as NYHA Class I and all LVOT gradients <30 mm Hg 32/117 (27%) 1/126 (1%) 26.6% (18.3% to 34.8%) Data are n (%) or mean (SD). HCMSQ-SoB = Hypertrophic Cardiomyopathy Symptom Questionnaire Shortness-of-Breath subscore. KCCQ-CSS = Kansas City Cardiomyopathy Questionnaire-Clinical Symptom Score. LVOT = Left ventricular outflow tract. pVO2 = Peak oxygen consumption. NYHA=New York Heart Association. *Model estimated least-square mean differences were reported for continuous variables. †Patients with a non-evaluable primary endpoint and NYHA secondary endpoint were considered as non-responders. The response rates were calculated with the N value as the denominator. ‡N was the number analysable for secondary endpoints based on availability of both baseline and week 30 values. §Due to the smaller numbers evaluable for patient-reported outcome endpoints, additional post-hoc analyses compared the reasons for missing data. Adapted from Olivotto. Lancet. 2020.
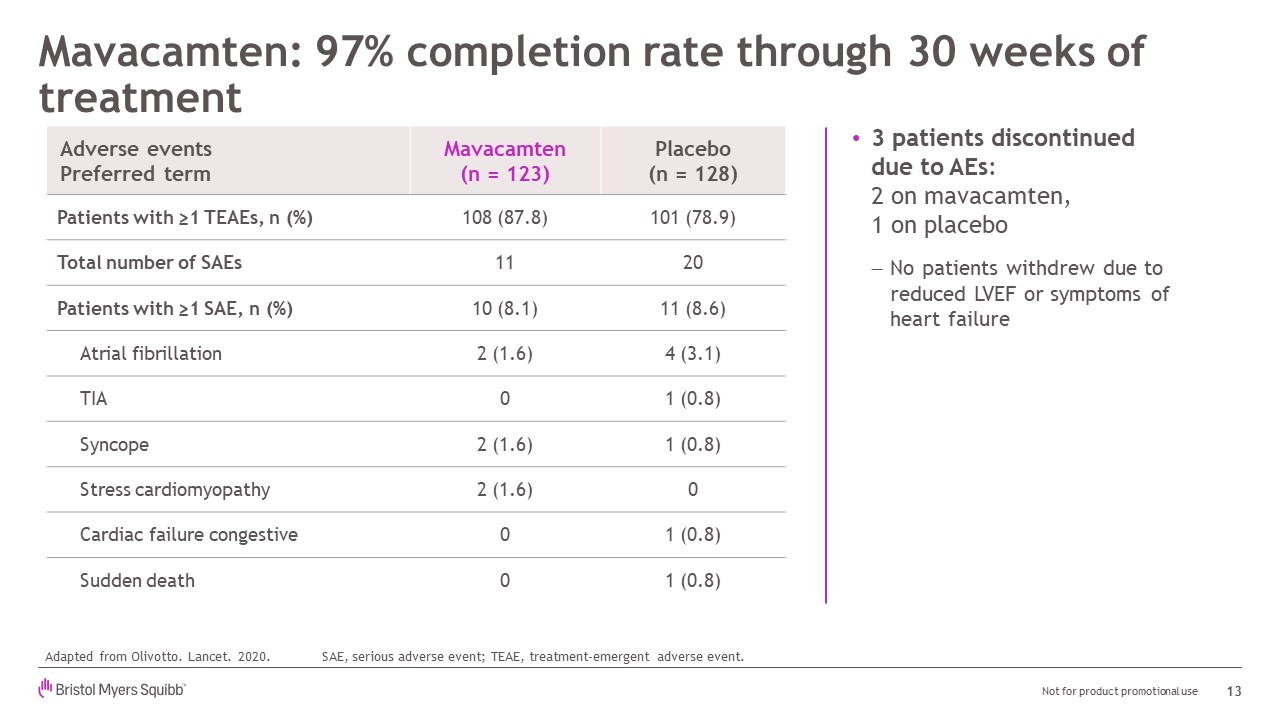
Mavacamten: 97% completion rate through 30 weeks of treatment Adverse eventsPreferred term Mavacamten(n = 123) Placebo(n = 128) Patients with ≥1 TEAEs, n (%) 108 (87.8) 101 (78.9) Total number of SAEs 11 20 Patients with ≥1 SAE, n (%) 10 (8.1) 11 (8.6) Atrial fibrillation 2 (1.6) 4 (3.1) TIA 0 1 (0.8) Syncope 2 (1.6) 1 (0.8) Stress cardiomyopathy 2 (1.6) 0 Cardiac failure congestive 0 1 (0.8) Sudden death 0 1 (0.8) 13 3 patients discontinueddue to AEs:2 on mavacamten,1 on placeboNo patients withdrew due to reduced LVEF or symptoms of heart failure SAE, serious adverse event; TEAE, treatment-emergent adverse event. Adapted from Olivotto. Lancet. 2020.

Future mavacamten indications and pipeline opportunities 14 Obstructive HCM (completed) P1 P3 Non-obstructive HCM Precision diastolic disease (mavacamten HFpEF) HCM Systolic dysfunction and atrial fibrillation Genetic dilated cardiomyopathy (DCM) P2 LUS-1 ACT-1 Precision diastolic diseases (preclinical) Precision systolic diseases (preclinical) MYK-224 danicamtiv mavacamten PIPELINE

Mavacamten: significant commercial opportunity in symptomatic obstructive HCM 15 Robust Clinical Data First-to-market Strong SpecialtyValue Source: Maron. NEJM. 2018; Marian. Circ Res. 2017; Maron. Am J Cardiol. 2016; Maron. Circulation. 1995. Addresses underlying disease and improves quality of lifeFirst-to-marketOpportunity to achieve value consistent with chronic specialty CV products 160–200K symptomatic patients across the U.S. and EU are in immediate need of treatment mavacamten
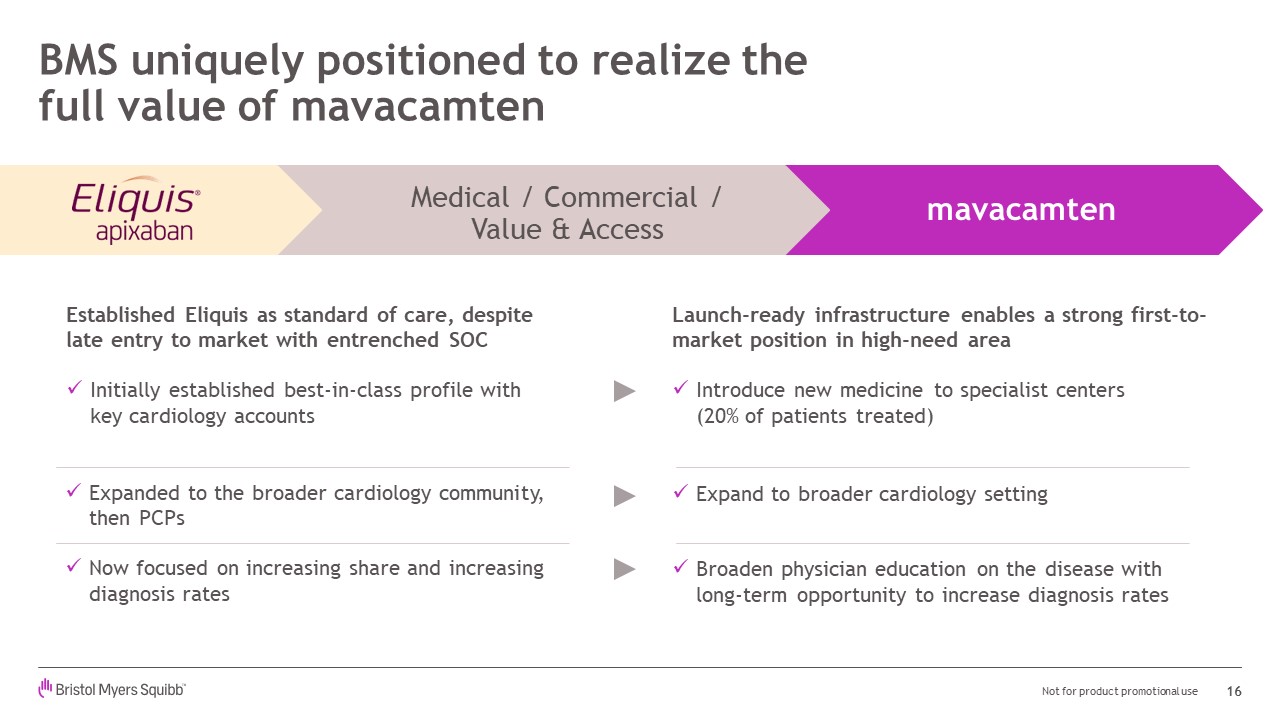
Expanded to the broader cardiology community, then PCPsNow focused on increasing share and increasing diagnosis rates BMS uniquely positioned to realize the full value of mavacamten 16 Established Eliquis as standard of care, despite late entry to market with entrenched SOCInitially established best-in-class profile with key cardiology accounts mavacamten Medical / Commercial /Value & Access Launch-ready infrastructure enables a strong first-to-market position in high-need areaIntroduce new medicine to specialist centers(20% of patients treated)Expand to broader cardiology settingBroaden physician education on the disease withlong-term opportunity to increase diagnosis rates

Symptomatic obstructive HCM opportunity High unmet needMavacamten: first medicine with potential to treat underlying diseaseSignificant commercial opportunityBMS uniquely positioned to commercialize 17

Significant financial benefits 18 Clear opportunity for value creation and P&L growthTransaction IRR in excess of MyoKardia’s WACCMavacamten launch indication is significant and supports the transaction Additional indications and pipeline provide incremental valueSignificant medium- and long-term growth opportunity Accretive to revenue and Non-GAAP EPS starting in 2023 Transaction details$13.1B total considerationAll-cash deal via tender offer, no financing contingencyExpect to use cash and debt while retaining strong investment grade credit ratings

Consistent approach to capital allocation 19 Continued commitment to the dividend* Committed to reducing debt:<1.5x Debt / EBITDA by end of 2024 Future innovation through business development *Subject to board approval

MyoKardia: Strong strategic fit and financially attractive 20 Compelling Opportunity First-in-class, specialty CV medicine addressing high unmet need, with significant commercial potential in the lead indication and upside potential from additional indications and pipeline; Obstructive HCM to be filed in Q1 2021 Strengthens CV franchise Broadens and accelerates expansion of CV portfolio & pipelineFull ownership Strong Value Rationale Generates IRR in excess of MyoKardia’s WACCMavacamten in obstructive HCM (launch indication) is a significant opportunitythat supports the transaction, with further opportunities from additional indications and pipeline Supports P&L Growth Significant medium- and long-term growth opportunity; Accretive to revenue and Non-GAAP EPS starting in 2023 & substantial impact in the second half of the decadeMeaningful growth driver in the medium- and long-term including into the2H of the decade Consistent Capital Allocation Remains focused on disciplined BD, improving leverage and reaching 1.5x Debt/EBITDA in 2024 while committed to the dividend

Q&A Giovanni Caforio, M.D.Board Chair,Chief Executive OfficerDavid ElkinsExecutive VP,Chief Financial Officer 21 Chris Boerner, Ph.D.Executive VP,Chief Commercialization OfficerSamit Hirawat, M.D.Executive VP,Chief Medical Officer,Global Drug Development