Tonix Pharmaceuticals Holding Corp. 8-K
Exhibit 99.02

© 2019 Tonix Pharmaceuticals Holding Corp. March 2019 Version P0160 3 - 5 - 19 (Doc 0438) Investor Presentation

© 2019 Tonix Pharmaceuticals Holding Corp. 2 Cautionary Note on Forward - Looking Statements Certain statements in this presentation regarding strategic plans, expectations and objectives for future operations or results are “forward - looking statements” as defined by the Private Securities Litigation Reform Act of 1995 . These statements may be identified by the use of forward - looking words such as “anticipate,” “believe,” “forecast,” “estimate” and “intend,” among others . These forward - looking statements are based on Tonix’s current expectations and actual results could differ materially . There are a number of factors that could cause actual events to differ materially from those indicated by such forward - looking statements . These factors include, but are not limited to, substantial competition ; our need for additional financing ; uncertainties of patent protection and litigation ; uncertainties of government or third party payor reimbursement ; limited research and development efforts and dependence upon third parties ; and risks related to failure to obtain U . S . Food and Drug Administration clearances or approvals and noncompliance with its regulations . As with any pharmaceutical under development, there are significant risks in the development, regulatory approval and commercialization of new products . The forward - looking statements in this presentation are made as of the date of this presentation, even if subsequently made available by Tonix on its website or otherwise . Tonix does not undertake an obligation to update or revise any forward - looking statement, except as required by law . Investors should read the risk factors set forth in the Annual Report on Form 10 - K for the year ended December 31 , 2017 , as filed with the Securities and Exchange Commission (the “SEC”) on March 9 , 2018 , and periodic reports filed with the SEC on or after the date thereof . All of Tonix's forward - looking statements are expressly qualified by all such risk factors and other cautionary statements .

© 2019 Tonix Pharmaceuticals Holding Corp. 3 Posttraumatic Stress Disorder (PTSD) – Lead program; new bedtime treatment - Tonmya ®1 • P302/RECOVERY Phase 3 clinical study with Week 4 primary endpoint to initiate in 1Q2019 • Results from 2 efficacy studies improve the new Phase 3 study design New Phase • P302/RECOVERY study design features accepted by the FDA 2 A gitation in Alzheimer’s disease (AAD) • IND 3 ready to support Phase 2 potential pivotal efficacy study Fibromyalgia Syndrome (FM) • IND 3 ready to support Phase 3 potential pivotal efficacy study TNX - 601 4 – Daytime PTSD treatment and treatment of neurocognitive dysfunction from corticosteroids • Pre - IND candidate; nonclinical development ongoing TNX - 801 5 - Smallpox - preventing vaccine candidate • Efficacy demonstrated in mouse model 1 Tonmya has been conditionally accepted by the U.S. FDA as the proposed trade name for TNX - 102 SL (cyclobenzaprine HCl sublingual tablets) for the treatment of PTSD. TNX - 102 SL is an investigational new drug and has not been approved for any indication. 2 FDA Meeting Minutes (November 26, 2018) 3 IND - Investigational New Drug Application 4 T ianeptine oxalate 5 S ynthesized live horsepox virus Tonix Development Highlights TNX - 102 SL Cyclobenzaprine Sublingual Tablets Pipeline
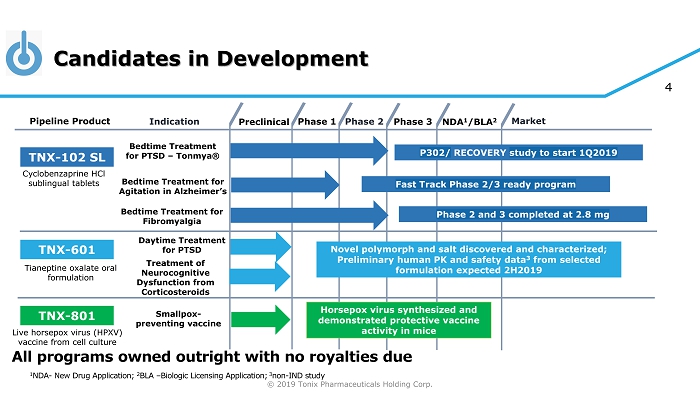
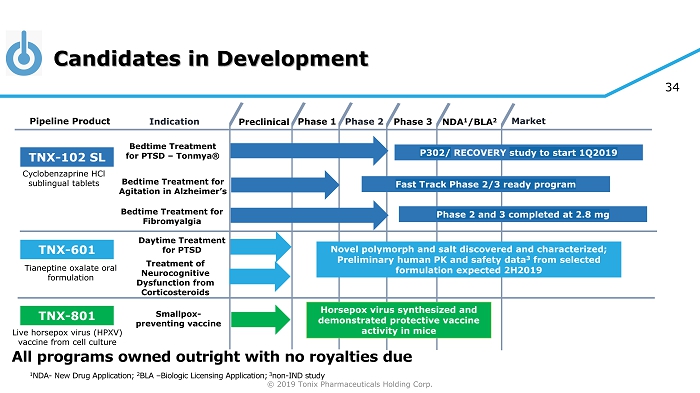
© 2019 Tonix Pharmaceuticals Holding Corp. 4 Candidates in Development Preclinical Phase 2 NDA 1 /BLA 2 Market Pipeline Product Indication Phase 3 TNX - 102 SL Bedtime Treatment for PTSD – Tonmya ® Daytime Treatment for PTSD TNX - 601 Novel polymorph and salt discovered and characterized; Preliminary human PK and safety data 3 from selected formulation expected 2H2019 TNX - 801 Horsepox virus synthesized and demonstrated protective vaccine activity in mice Smallpox - preventing vaccine Cyclobenzaprine HCl sublingual tablets Tianeptine oxalate oral formulation Live horsepox virus (HPXV) vaccine from cell culture Phase 1 1 NDA - New Drug Application; 2 BLA – Biologic Licensing Application; 3 non - IND study Bedtime Treatment for Agitation in Alzheimer’s All programs owned outright with no royalties due P302/ RECOVERY study to start 1Q2019 Bedtime Treatment for Fibromyalgia Phase 2 and 3 completed at 2.8 mg Treatment of Neurocognitive Dysfunction from Corticosteroids Fast Track Phase 2/3 ready program

© 2019 Tonix Pharmaceuticals Holding Corp. 5 Tonmya : a Potential Bedtime Treatment for PTSD First investigational new drug to show treatment effect in military - related PTSD in two potential pivotal efficacy studies • Phase 2 study (P201/ AtEase ) showed Tonmya 5.6 mg had a strong signal of treatment effect at Week 12 as measured by CAPS - 5 1 • Phase 3 study (P301/HONOR) provided evidence of effectiveness as early as 4 weeks after treatment but diminished over time due to high placebo response − Retrospective analysis showed persistent effectiveness at Week 12 in subgroup with trauma ≤9 years from screening • Both studies can be used as supportive evidence of efficacy and safety for Tonmya NDA submission • No serious or unexpected adverse events related to Tonmya were reported FDA feedback and acceptance on new Phase 3 study (P302/RECOVERY) received in November 2 Patent protection through 2034 in U.S. 3 • Composition of matter patent for transmucosal delivery of cyclobenzaprine Novel mechanism targets sleep quality • Memory processing during sleep is important to recovery from PTSD 1 CAPS - 5 = Clinician - Administered PTSD Scale for DSM - 5 2 FDA Meeting Minutes, November 26, 2018; 3 U .S. Patent No. 9,636,408 for eutectic proprietary Protectic ™ formulation

© 2019 Tonix Pharmaceuticals Holding Corp. 6 No Recognized Abuse Potential in Clinical Studies Active ingredient is cyclobenzaprine, which is structurally related to tricyclic antidepressants • Cyclobenzaprine interacts with receptors that regulate sleep quality: 5 - HT 2A, a 1 - adrenergic and histamine H 1 receptors • Cyclobenzaprine does NOT interact with the same receptors as traditional hypnotic sleep drugs, benzodiazepines or non - benzodiazepines that are associated with retrograde amnesia • Cyclobenzaprine - containing product was approved 40 years ago and current labeling (May 2018) indicates no abuse or dependence concern Tonmya NDA can be filed without drug abuse and dependency assessment studies • Discussed at March 9, 2017 meeting with the FDA

© 2019 Tonix Pharmaceuticals Holding Corp. 7 TNX - 102 SL Intellectual Property – U.S. Protection until 2034 Composition of matter (eutectic) : Protection expected to 2034 • United States Patent and Trademark Office (USPTO) issued U.S. Patent No. 9,636,408 in May 2017 U.S. Patent No. 9,956,188 in May 2018 and U.S. Patent No. 10,117,936 in November 2018 • Japanese Patent Office (JPO) issued Japanese Patent No. 6310542 in March 2018 • New Zealand Intellectual Property Office (NZIPO) issued New Zealand Patent No. 631152 in May 2017 • 37 patent applications pending (2 allowed (US and South Africa)) Pharmacokinetics (PK) : Protection expected to 2033 • JPO issued Japanese Patent No. 6259452 in December 2017 • NZIPO issued New Zealand Patent No. 631144 in March 2017 • Taiwanese Intellectual Property Office issued Taiwanese Patent No. I590820 in July 2017 • 21 patent applications pending (1 allowed (Australia)) Method of use for active ingredient cyclobenzaprine : Protection expected to 2030 • USPTO issued U.S. Patent 9,918,948 in March 2018 • European Patent Office issued European Patent No. 2 501 234B1 in September 2017 (validated in 38 countries). Opposition filed in June 2018 • 2 patent applications pending

© 2019 Tonix Pharmaceuticals Holding Corp. 8 TNX - 102 SL: Sublingual Formulation is Designed for Bedtime Administration TNX - 102 SL: Proprietary sublingual formulation of cyclobenzaprine (CBP) with transmucosal absorption • Innovation by design with patent protected CBP/mannitol eutectic • Rapid systemic exposure • Increases bioavailability during sleep • Avoids first - pass metabolism • Lowers exposure to long - lived active major metabolite, norcyclobenzaprine ( norCBP ) CBP undergoes extensive first - pass hepatic metabolism when orally ingested • Active major metabolite, norCBP 1 • Long half - life (~72 hours) • Less selective for target receptors ( 5 - HT 2A, a 1 - adrenergic, histamine H 1 ) • More selective for norepinephrine transporter and muscarinic M 1 TNX - 102 SL 505(b)(2) NDA approval can rely on the safety of the reference listed drug (AMRIX ® ) 2 1 Daugherty et al., Abstract 728, Society of Biological Psychiatry 70th Annual Scientific Convention, May 14 - 16, 2015, Toronto Ont ario, Canada 2 FDA Minutes (November 26, 2018)

© 2019 Tonix Pharmaceuticals Holding Corp. 9 Tonmya: Hypothesized Novel Mechanism Targets Sleep Quality for Recovery from PTSD PTSD is a disorder of recovery • Most people exposed to extreme trauma recover over a few weeks • In PTSD, recovery process impeded due to insufficient sleep - dependent memory processing 1,2 Memory processing is essential to recovery • Vulnerability to memory intrusions and trauma triggers remains if no consolidation of new learning (extinction) Tonmya targets sleep quality 3 • The active ingredient in Tonmya, cyclobenzaprine, interacts with receptors that regulate sleep quality: strongly binds and potently blocks 5 - HT 2A , a 1 - adrenergic and histamine H 1 receptors, permissive to sleep - dependent recovery processes 1 Straus LD, Acheson DT, Risbrough VB, Drummond SPA. Sleep Deprivation Disrupts Recall of Conditioned Fear Extinction. Biol Psychiatry Cogn Neurosci Neuroimaging. 2017; 2(2):123 - 129. 2 Murkar ALA, De Koninck J. Consolidative mechanisms of emotional processing in REM sleep and PTSD. Sleep Med Rev. 2018; 41:173 - 184. 3 Daugherty et al., Abstract 728, Society of Biological Psychiatry 70th Annual Scientific Convention, May 14 - 16, 2015, Toronto Ont ario, Canada

© 2019 Tonix Pharmaceuticals Holding Corp. 10 PTSD: Not Well - Served by Approved Treatments FDA - approved SSRIs, paroxetine and sertraline, are indicated as a treatment for PTSD • Neither drug has shown efficacy in military - related PTSD • Majority of male PTSD patients unresponsive or intolerant to current treatments • Side effects relating to sexual dysfunction, sleep disturbance and weight gain are commonly reported Characteristics of an ideal drug therapy that would be compatible and complementary with behavioral therapy • Lack of retrograde amnesia (e.g., unlike off - label use of benzodiazepines and non - benzodiazepines) • Lack of interference on sleep (e.g., unlike approved SSRIs) Tonmya is being investigated in both military and civilian PTSD and is expected to be indicated as a “treatment for PTSD”
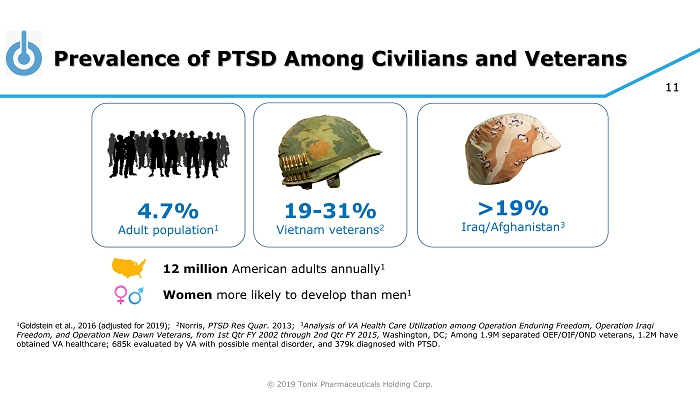
© 2019 Tonix Pharmaceuticals Holding Corp. 11 Prevalence of PTSD Among Civilians and Veterans 1 Goldstein et al., 2016 (adjusted for 2019) ; 2 Norris, PTSD Res Quar . 2013; 3 Analysis of VA Health Care Utilization among Operation Enduring Freedom, Operation Iraqi Freedom, and Operation New Dawn Veterans, from 1st Qtr FY 2002 through 2nd Qtr FY 2015, Washington, DC ; Among 1.9M separated OEF/OIF/OND veterans, 1.2M have obtained VA healthcare; 685k evaluated by VA with possible mental disorder, and 379k diagnosed with PTSD. >19% Iraq/Afghanistan 3 4.7% Adult population 1 19 - 31% Vietnam veterans 2 12 million American adults annually 1 Women more likely to develop than men 1
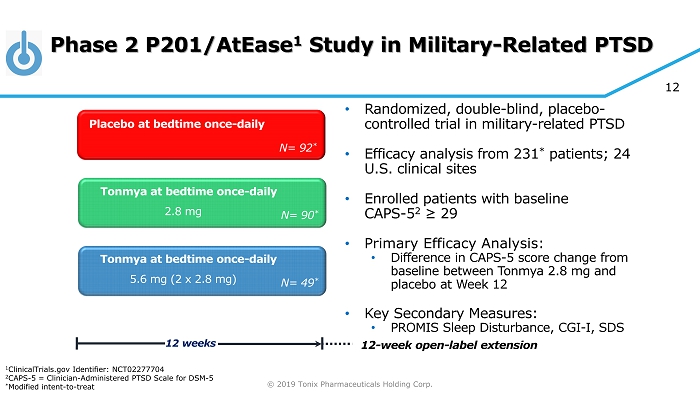
© 2019 Tonix Pharmaceuticals Holding Corp. 12 Phase 2 P201/AtEase 1 Study in Military - Related PTSD • Randomized, double - blind, placebo - controlled trial in military - related PTSD • Efficacy analysis from 231 * patients; 24 U.S. clinical sites • Enrolled patients with baseline CAPS - 5 2 ≥ 29 • Primary Efficacy Analysis: • Difference in CAPS - 5 score change from baseline between Tonmya 2.8 mg and placebo at Week 12 • Key Secondary Measures: • PROMIS Sleep Disturbance, CGI - I, SDS Tonmya at bedtime once - daily Placebo at bedtime once - daily 12 weeks N= 90 * Tonmya at bedtime once - daily N= 92 * N= 49 * 2.8 mg 5.6 mg (2 x 2.8 mg) 12 - week open - label extension 1 ClinicalTrials.gov Identifier: NCT02277704 2 CAPS - 5 = Clinician - Administered PTSD Scale for DSM - 5 * Modified intent - to - treat

© 2019 Tonix Pharmaceuticals Holding Corp. 13 P201/AtEase Study P201 was a large adequate well - controlled Phase 2 study in military - related PTSD • Primary endpoint (Week 12 CAPS - 5) did not separate from placebo for TNX - 102 SL 2.8 mg • No safety or tolerability issue discovered • Retrospective analyses showed TNX - 102 SL 5.6 mg had a strong signal of treatment effect at Week 12 CAPS - 5 (P=0.053) and CGI - I (P=0.041) scores • Retrospective analyses suggested CAPS - 5 ≥ 33 enrollment criteria for Phase 3
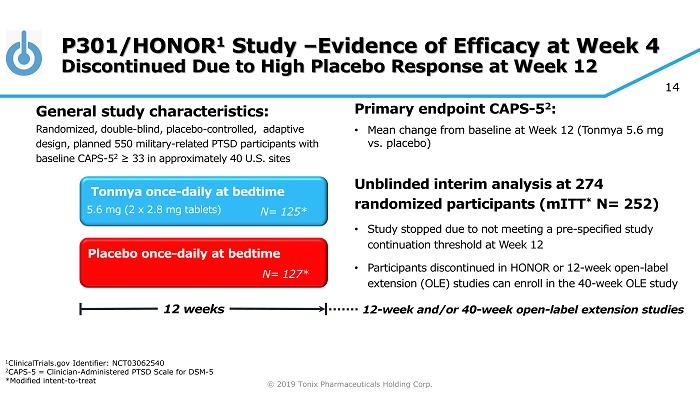
© 2019 Tonix Pharmaceuticals Holding Corp. 14 P301/HONOR 1 Study – Evidence of Efficacy at Week 4 Discontinued Due to High Placebo Response at Week 12 Primary e ndpoint CAPS - 5 2 : • Mean change from baseline at W eek 12 (Tonmya 5.6 mg vs. placebo ) Unblinded interim analysis at 274 randomized participants ( mITT * N= 252) • Study stopped due to not meeting a pre - specified study continuation threshold at Week 12 • Participants discontinued in HONOR or 12 - week open - label extension (OLE) studies can enroll in the 40 - week OLE study Placebo once - daily at bedtime 12 weeks Tonmya once - daily at bedtime N= 127* N= 125* 5.6 mg (2 x 2.8 mg tablets) General s tudy c haracteristics: Randomized, double - blind, placebo - controlled , adaptive design, planned 550 military - related PTSD participants with baseline CAPS - 5 2 ≥ 33 in approximately 40 U.S. sites 12 - week and/or 40 - week open - label extension studies 1 ClinicalTrials.gov Identifier: NCT03062540 2 CAPS - 5 = Clinician - Administered PTSD Scale for DSM - 5 *Modified intent - to - treat
© 2019 Tonix Pharmaceuticals Holding Corp. 15 P301/HONOR Study Stopped After Interim Analysis (July 2018) P301 was a large adequate well - controlled Phase 3 study in military - related PTSD • Separation on primary endpoint at Week 12 did not cross pre - specified study continuation threshold at Week 12 (p=0.602) • No safety or tolerability issue discovered • Retrospective analyses showed Week 4 CAPS - 5 (P=0.019) and CGI - I (P=0.015) scores in Tonmya group had a strong signal of treatment effect P301 dataset is complex and rich • Retrospective analyses presented at Military Health System Research Symposium (MHSRS) in Kissimmee, FL on August 22, 2018 • Results discussed with the FDA 1 and helped to design the new Phase 3 P302/RECOVERY study with high probability of success 1 FDA Meeting Minutes (November 26, 2018)
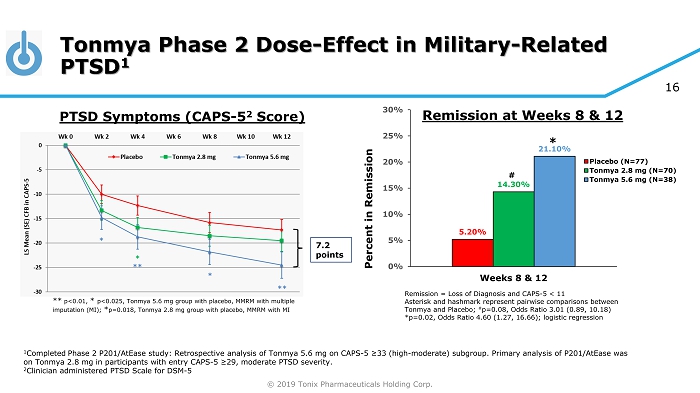
© 2019 Tonix Pharmaceuticals Holding Corp. 16 Tonmya Phase 2 Dose - Effect in Military - Related PTSD 1 1 Completed Phase 2 P201/AtEase study: Retrospective analysis of Tonmya 5.6 mg on CAPS - 5 ≥33 (high - moderate) subgroup. Primary analysis of P 201/AtEase was on Tonmya 2.8 mg in participants with entry CAPS - 5 ≥29 , moderate PTSD severity. 2 Clinician administered PTSD Scale for DSM - 5 7.2 points ** p<0.01, * p<0.025, Tonmya 5.6 mg group with placebo, MMRM with multiple imputation (MI); * p=0.018, Tonmya 2.8 mg group with placebo, MMRM with MI PTSD Symptoms (CAPS - 5 2 Score) Remission = Loss of Diagnosis and CAPS - 5 < 11 Asterisk and hashmark represent pairwise comparisons between Tonmya and Placebo; # p=0.08, Odds Ratio 3.01 (0.89, 10.18) *p=0.02, Odds Ratio 4.60 (1.27, 16.66); logistic regression 5.20% 14.30% 21.10% 0% 5% 10% 15% 20% 25% 30% Weeks 8 & 12 Percent in Remission Placebo (N=77) Tonmya 2.8 mg (N=70) Tonmya 5.6 mg (N=38) # * Remission at Weeks 8 & 12
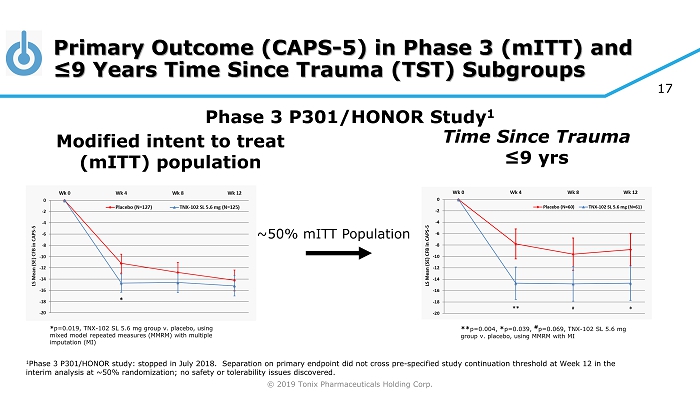
© 2019 Tonix Pharmaceuticals Holding Corp. Primary Outcome (CAPS - 5) in Phase 3 ( mITT ) and ≤9 Years Time Since Trauma (TST) Subgroups Modified intent to treat ( mITT ) population Phase 3 P301/HONOR Study 1 ** p=0.004, * p=0.039, # p=0.069, TNX - 102 SL 5.6 mg group v. placebo, using MMRM with MI ~50% mITT Population 17 * p=0.019, TNX - 102 SL 5.6 mg group v. placebo, using mixed model repeated measures (MMRM) with multiple imputation (MI) Time Since Trauma ≤9 yrs 1 Phase 3 P301/HONOR study: stopped in July 2018. Separation on primary endpoint did not cross pre - specified study continuation t hreshold at Week 12 in the interim analysis at ~50% randomization; no safety or tolerability issues discovered.
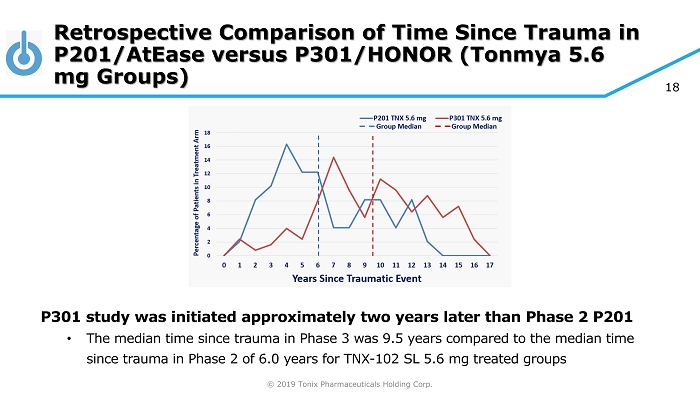
© 2019 Tonix Pharmaceuticals Holding Corp. 18 Retrospective Comparison of Time Since Trauma in P201/AtEase versus P301/HONOR (Tonmya 5.6 mg Groups) P301 study was initiated approximately two years later than Phase 2 P201 • The median time since trauma in Phase 3 was 9.5 years compared to the median time since trauma in Phase 2 of 6.0 years for TNX - 102 SL 5.6 mg treated groups
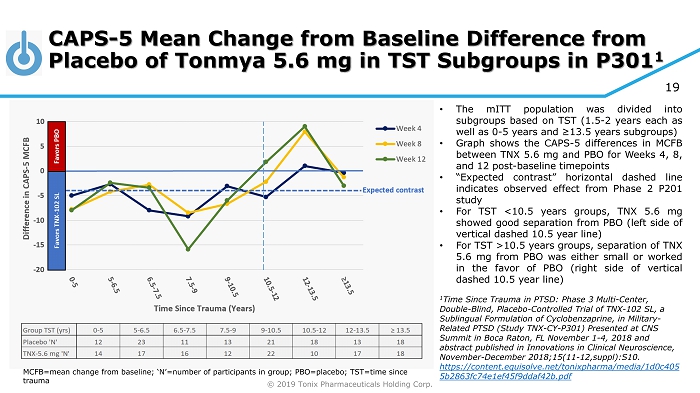
© 2019 Tonix Pharmaceuticals Holding Corp. 19 CAPS - 5 Mean Change from Baseline Difference from Placebo of Tonmya 5.6 mg in TST Subgroups in P301 1 MCFB=mean change from baseline; ‘N’=number of participants in group; PBO=placebo; TST=time since trauma • The mITT population was divided into subgroups based on TST ( 1 . 5 - 2 years each as well as 0 - 5 years and ≥ 13 . 5 years subgroups) • Graph shows the CAPS - 5 differences in MCFB between TNX 5 . 6 mg and PBO for Weeks 4 , 8 , and 12 post - baseline timepoints • “Expected contrast” horizontal dashed line indicates observed effect from Phase 2 P 201 study • For TST < 10 . 5 years groups, TNX 5 . 6 mg showed good separation from PBO (left side of vertical dashed 10 . 5 year line) • For TST > 10 . 5 years groups, separation of TNX 5 . 6 mg from PBO was either small or worked in the favor of PBO (right side of vertical dashed 10 . 5 year line) 1 Time Since Trauma in PTSD: Phase 3 Multi - Center, Double - Blind, Placebo - Controlled Trial of TNX - 102 SL, a Sublingual Formulation of Cyclobenzaprine, in Military - Related PTSD (Study TNX - CY - P301) Presented at CNS Summit in Boca Raton, FL November 1 - 4, 2018 and abstract published in Innovations in Clinical Neuroscience, November - December 2018;15(11 - 12,suppl):S10. https://content.equisolve.net/tonixpharma/media/1d0c405 5b2863fc74e1ef45f9ddaf42b.pdf
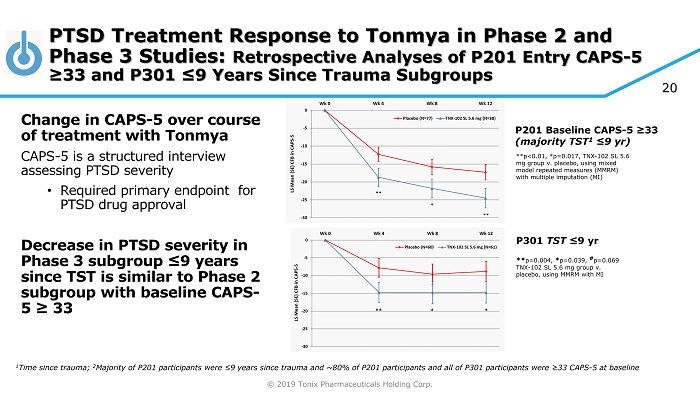
© 2019 Tonix Pharmaceuticals Holding Corp. P201 Baseline CAPS - 5 ≥33 (majority TST 1 ≤9 yr ) **p<0.01, *p=0.017, TNX - 102 SL 5.6 mg group v. placebo, using mixed model repeated measures (MMRM) with multiple imputation (MI) P301 TST ≤9 yr ** p=0.004, * p=0.039, # p=0.069 TNX - 102 SL 5.6 mg group v. placebo, using MMRM with MI PTSD Treatment Response to Tonmya in Phase 2 and Phase 3 Studies: Retrospective Analyses of P201 Entry CAPS - 5 ≥33 and P301 ≤9 Years Since Trauma Subgroups Change in CAPS - 5 over course of treatment with Tonmya CAPS - 5 is a structured interview assessing PTSD severity • Required primary endpoint for PTSD drug approval Decrease in PTSD severity in Phase 3 subgroup ≤9 years since TST is similar to Phase 2 subgroup with baseline CAPS - 5 ≥ 33 1 Time since trauma; 2 Majority of P201 participants were ≤ 9 years since trauma and ~80% of P201 participants and all of P301 participants were ≥33 CAPS - 5 at baseline 20
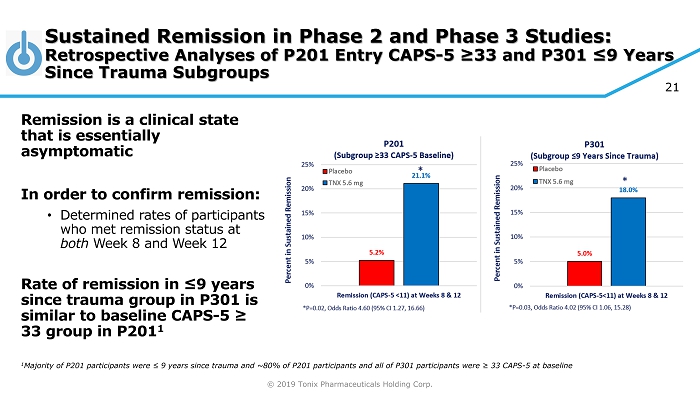
© 2019 Tonix Pharmaceuticals Holding Corp. 21 Sustained Remission in Phase 2 and Phase 3 Studies: Retrospective Analyses of P201 Entry CAPS - 5 ≥33 and P301 ≤9 Years Since Trauma Subgroups Remission is a clinical state that is essentially asymptomatic In order to confirm remission: • Determined rates of participants who met remission status at both Week 8 and Week 12 Rate of remission in ≤9 years since trauma group in P301 is similar to baseline CAPS - 5 ≥ 33 group in P201 1 1 Majority of P201 participants were ≤ 9 years since trauma and ~80% of P201 participants and all of P301 participants were ≥ 33 CAPS - 5 at baseline
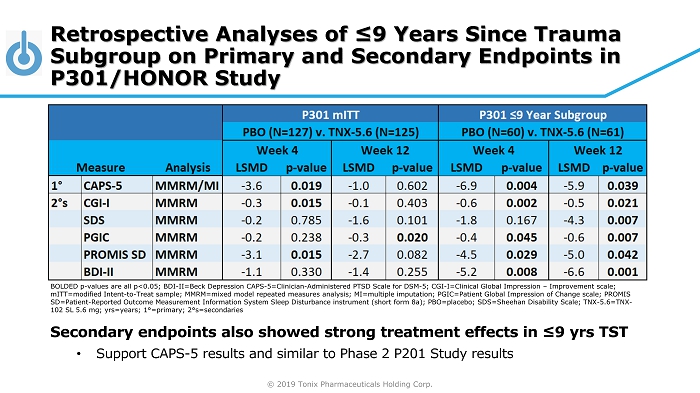
© 2019 Tonix Pharmaceuticals Holding Corp. BOLDED p - values are all p<0.05; BDI - II=Beck Depression CAPS - 5=Clinician - Administered PTSD Scale for DSM - 5; CGI - I=Clinical Global Impression – Improvement scale; mITT =modified Intent - to - Treat sample; MMRM=mixed model repeated measures analysis; MI=multiple imputation; PGIC=Patient Global Impre ssion of Change scale; PROMIS SD=Patient - Reported Outcome Measurement Information System Sleep Disturbance instrument (short form 8a); PBO=placebo; SDS=Sheeha n Disability Scale; TNX - 5.6=TNX - 102 SL 5.6 mg; yrs =years; 1 ° =primary; 2 ° s=secondaries Retrospective Analyses of ≤9 Years Since Trauma Subgroup on Primary and Secondary Endpoints in P301/HONOR Study Secondary endpoints also showed strong treatment effects in ≤9 yrs TST • Support CAPS - 5 results and similar to Phase 2 P201 Study results
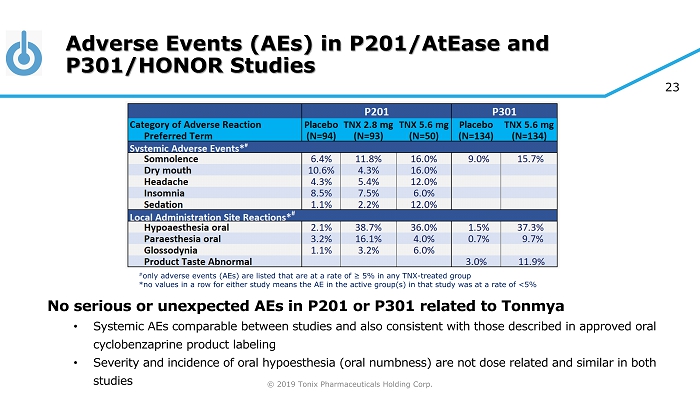
© 2019 Tonix Pharmaceuticals Holding Corp. 23 Adverse Events (AEs) in P201/AtEase and P301/HONOR Studies No serious or unexpected AEs in P201 or P301 related to Tonmya • Systemic AEs comparable between studies and also consistent with those described in approved oral cyclobenzaprine product labeling • Severity and incidence of oral hypoesthesia (oral numbness) are not dose related and similar in both studies # only adverse events (AEs) are listed that are at a rate of ≥ 5% in any TNX - treated group *no values in a row for either study means the AE in the active group(s) in that study was at a rate of <5%
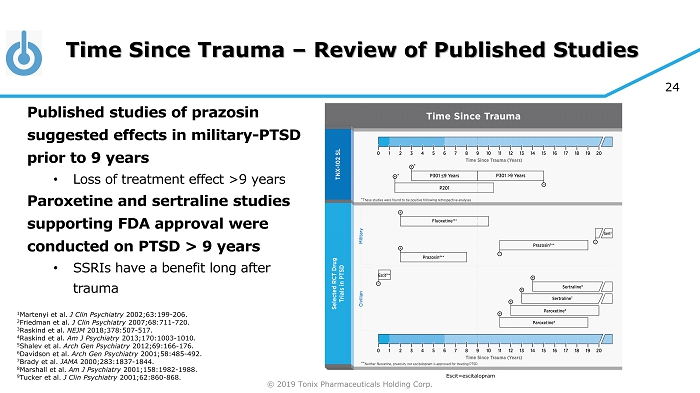
© 2019 Tonix Pharmaceuticals Holding Corp. 24 Time Since Trauma – Review of Published Studies Published studies of prazosin suggested effects in military - PTSD prior to 9 years • Loss of treatment effect >9 years Paroxetine and sertraline studies supporting FDA approval were conducted on PTSD > 9 years • SSRIs have a benefit long after trauma 1 Martenyi et al. J Clin Psychiatry 2002;63:199 - 206. 2 Friedman et al. J Clin Psychiatry 2007;68:711 - 720. 3 Raskind et al. NEJM 2018;378:507 - 517. 4 Raskind et al. Am J Psychiatry 2013;170:1003 - 1010. 5 Shalev et al. Arch Gen Psychiatry 2012;69:166 - 176. 6 Davidson et al. Arch Gen Psychiatry 2001;58:485 - 492. 7 Brady et al. JAMA 2000;283:1837 - 1844. 8 Marshall et al. Am J Psychiatry 2001;158:1982 - 1988. 9 Tucker et al. J Clin Psychiatry 2001;62:860 - 868. Escit =escitalopram
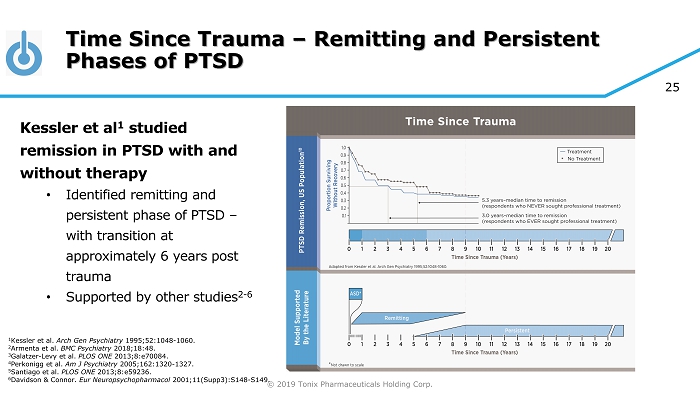
© 2019 Tonix Pharmaceuticals Holding Corp. 25 Time Since Trauma – Remitting and Persistent Phases of PTSD Kessler et al 1 studied remission in PTSD with and without therapy • Identified remitting and persistent phase of PTSD – with transition at approximately 6 years post trauma • Supported by other studies 2 - 6 1 Kessler et al. Arch Gen Psychiatry 1995;52:1048 - 1060. 2 Armenta et al. BMC Psychiatry 2018;18:48. 3 Galatzer - Levy et al. PLOS ONE 2013;8:e70084. 4 Perkonigg et al. Am J Psychiatry 2005;162:1320 - 1327. 5 Santiago et al. PLOS ONE 2013;8:e59236. 6 Davidson & Connor. Eur Neuropsychopharmacol 2001;11(Supp3):S148 - S149.
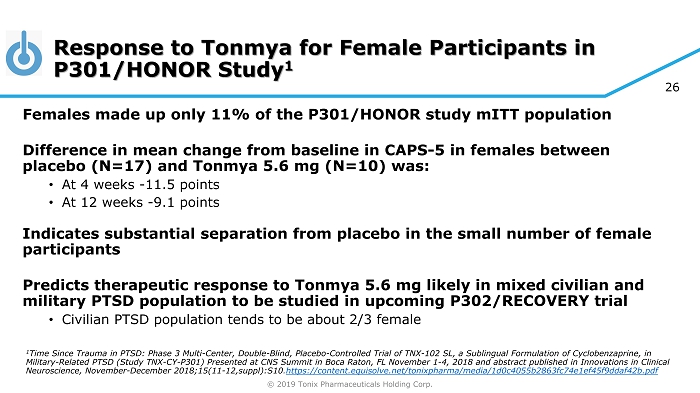
© 2019 Tonix Pharmaceuticals Holding Corp. 26 Response to Tonmya for Female Participants in P301/HONOR Study 1 Females made up only 11% of the P301/HONOR study mITT population Difference in mean change from baseline in CAPS - 5 in females between placebo (N=17) and Tonmya 5.6 mg (N=10) was: • At 4 weeks - 11.5 points • At 12 weeks - 9.1 points Indicates substantial separation from placebo in the small number of female participants Predicts therapeutic response to Tonmya 5.6 mg likely in mixed civilian and military PTSD population to be studied in upcoming P302/RECOVERY trial • Civilian PTSD population tends to be about 2/3 female 1 Time Since Trauma in PTSD: Phase 3 Multi - Center, Double - Blind, Placebo - Controlled Trial of TNX - 102 SL, a Sublingual Formulation of Cyclobenzaprine, in Military - Related PTSD (Study TNX - CY - P301) Presented at CNS Summit in Boca Raton, FL November 1 - 4, 2018 and abstract published in Innovations in Clinical Neuroscience, November - December 2018;15(11 - 12,suppl):S10 . https://content.equisolve.net/tonixpharma/media/1d0c4055b2863fc74e1ef45f9ddaf42b.pdf

© 2019 Tonix Pharmaceuticals Holding Corp. 27 Non - combat traumas studied are similar to traumas experienced in civilian populations with PTSD To determine the therapeutic effects of Tonmya 5.6 mg in a mixed civilian and military population, difference in MCFB in CAPS - 5 was assessed in non - combat traumas in ≤9 years TST subgroup (placebo N=14, Tonmya 5.6 mg N=10): • At 4 weeks - 4.8 points • At 12 weeks - 4.4 points Non - combat traumas treated with Tonmya 5.6 mg showed clinically meaningful separation from placebo at Weeks 4 and 12, suggesting a mixed civilian and military sample within 9 years of index trauma may show a therapeutic response to Tonmya 1 Time Since Trauma in PTSD: Phase 3 Multi - Center, Double - Blind, Placebo - Controlled Trial of TNX - 102 SL, a Sublingual Formulation of Cyclobenzaprine, in Military - Related PTSD (Study TNX - CY - P301) Presented at CNS Summit in Boca Raton, FL November, 2018; and abst ract published in Innovations in Clinical Neuroscience, November - December 2018;15(11 - 12,suppl):S10. https://content.equisolve.net/ tonixpharma /media/1d0c4055b2863fc74e1ef45f9ddaf42b.pdf Response to Tonmya for Non - Combat Traumas in P301/HONOR Study in ≤9 Years Time Since Trauma Subgroup 1 CAPS - 5=Clinician - Administered PTSD Scale for DSM - 5; MCFB=mean change from baseline; mITT =modified Intent - to - Treat sample; TST=time since trauma

© 2019 Tonix Pharmaceuticals Holding Corp. 28 Summary of Clinical Experience with Tonmya/ TNX - 102 SL in PTSD Median time since trauma (TST) in TNX - 102 SL 5.6 mg group in the P301/HONOR study (9.5 years) was longer than P201/AtEase study (6 years) • Both studied military - related PTSD • Time has passed since the surge in Iraq In retrospective analysis, the ≤ 9 year subgroup of P301 study had similar results as the P201 study (primary and secondary) • TST is important in placebo - controlled clinical study • Potential enrichment in ≤ 9 years TST subgroup for treatment responders The ≤ 9 year subgroup of P301 may be enriched for “Remitting Phase” of PTSD 1 - 4 • Expect remitting phase of PTSD is more amenable to drug studies Results from retrospective analyses lead to improved Phase 3 study design 1 Kessler et al. Arch Gen Psychiatry 1995;52:1048 - 1060. 2 Armenta et al. BMC Psychiatry 2018;18:48. 3 Galatzer - Levy et al. PLOS ONE 2013;8:e70084. 4 Perkonigg et al. Am J Psychiatry 2005;162:1320 - 1327.

© 2019 Tonix Pharmaceuticals Holding Corp. 29 New Phase 3 P302/RECOVERY Study – Expected to Start 1Q 2019 Primary e ndpoint: • CAPS - 5 1 mean change from baseline at Week 4 ( Tonmya 5.6 mg vs. placebo) Key Secondary e ndpoint s include: • CAPS - 5 m ean change from baseline at W eek 12 (Tonmya 5.6 mg vs. placebo ) • Change from baseline Clinical Global Impression – Severity scale • Change from baseline Sheehan Disability Scale total score Potential pivotal efficacy study to support NDA approval Placebo once - daily at bedtime 12 weeks Tonmya once - daily at bedtime 5.6 mg (2 x 2.8 mg tablets) General s tudy c haracteristics: • Randomized, double - blind, placebo - controlle d study with baseline CAPS - 5 1 ≥ 33 in approximately 30 U.S. sites • Enrollment restricted to study participants with PTSD who experienced an index trauma ≤ 9 years from the date of screening • Both civilian and military - related PTSD to be included 1 CAPS - 5 = Clinician - Administered PTSD Scale for DSM - 5 Primary e ndpoint CAPS - 5 1 at Week 4 N= 125 N= 125

© 2019 Tonix Pharmaceuticals Holding Corp. 30 Late - Stage PTSD Drug Candidates Tonmya • Phase 3 development focused on military - related and civilian PTSD; showed activity in treatment of military - related PTSD in large multi - center trials MDMA - assisted psychotherapy • Breakthrough therapy that is Phase 3 - ready; showed activity in a Phase 2 study of PTSD; enrolling in Phase 3 Other drugs currently (or recently) in Phase 2 development • Rexulti ® ( brexpiprazole ) - Otsuka/Lundbeck; atypical antipsychotic; positive clinical results from Phase 2 study reported in November 2018 for brexpiprazole , when used in combination with an approved PTSD medication, sertraline, but not as monotherapy • NYX - 783 - Aptinyx ; NMDA receptor modulator (enrolling for 8 - week Phase 2 study of 144 patients using 50 mg either once daily or once weekly) • BNC - 201 – Bionomics; nicotinic receptor modulator (program planned to resume after reformulation)

© 2019 Tonix Pharmaceuticals Holding Corp. 31 TNX - 102 SL – Bedtime Treatment for Multiple Potential Indications Ma nagement of Fibromyalgia (FM) – chronic pain condition • TNX - 102 SL studied at low dose (2.8 mg) – half the dose being developed for PTSD – did not separate from placebo on primary endpoint, average pain improvement (responder analysis) • Retrospective analysis showed average pain improvement (secondary endpoint) after 12 weeks of treatment showed statistical significance (P< 0.05, MMRM) • Low dose TNX - 102 SL (2.8 mg) showed an improvement in sleep quality in Phase 2 and Pha se 3 FM trials • Efficacy of TNX - 102 SL 5.6 mg in FM can be studied in a potential pivotal study to support product registration Agitation in Alzheimer’s Disease • Fast Track designation granted July 2018 • Phase 2/ potential pivotal efficacy study protocol received FDA comments in October 2018

© 2019 Tonix Pharmaceuticals Holding Corp. 32 Agitation in Alzheimer’s Disease – Additional Indication Being Developed for TNX - 102 SL FDA designated Fast Track development program Significant unmet need • No FDA approved drugs for the treatment of agitation in Alzheimer’s Mechanism of improving sleep quality • Sleep disturbance is a significant and common symptom in Alzheimer’s Pharmacological advantages outweigh potential concerns of using TNX - 102 SL in treating agitation in Alzheimer’s disease • Blocks 3 receptors, not just one (e.g., 5 - HT 2A )

© 2019 Tonix Pharmaceuticals Holding Corp. 33 TNX - 102 SL for Agitation in Alzheimer’s – Regulatory Status and Registration Strategy FDA confirmed no additional study was needed prior to IND submission • Pre - IND meeting established open dialogue with the FDA on pivotal clinical study design and efficacy endpoints to support product registration Proposed Phase 2 IND study can potentially serve as a pivotal efficacy study to support NDA approval • FDA comments on final protocol received October 2018 Registration Strategy of TNX - 102 SL for agitation in Alzheimer’s disease • Efficacy Supplement (sNDA 1 ) may be leveraged from the PTSD/FM development program and supported by Initial NDA approval for PTSD/FM 1 Supplemental New Drug Application
© 2019 Tonix Pharmaceuticals Holding Corp. 34 Candidates in Development Preclinical Phase 2 NDA 1 /BLA 2 Market Pipeline Product Indication Phase 3 TNX - 102 SL Bedtime Treatment for PTSD – Tonmya ® Daytime Treatment for PTSD TNX - 601 Novel polymorph and salt discovered and characterized; Preliminary human PK and safety data 3 from selected formulation expected 2H2019 TNX - 801 Horsepox virus synthesized and demonstrated protective vaccine activity in mice Smallpox - preventing vaccine Cyclobenzaprine HCl sublingual tablets Tianeptine oxalate oral formulation Live horsepox virus (HPXV) vaccine from cell culture Phase 1 1 NDA - New Drug Application; 2 BLA – Biologic Licensing Application; 3 non - IND study Bedtime Treatment for Agitation in Alzheimer’s All programs owned outright with no royalties due P302/ RECOVERY study to start 1Q2019 Bedtime Treatment for Fibromyalgia Phase 2 and 3 completed at 2.8 mg Treatment of Neurocognitive Dysfunction from Corticosteroids Fast Track Phase 2/3 ready program

© 2019 Tonix Pharmaceuticals Holding Corp. 35 TNX - 601 ( Tianeptine Oxalate): A Potential Clinical Candidate for PTSD Pre - IND Candidate Targeted as a 1 st line monotherapy for PTSD: oral formulation for daytime dosing x Leverages expertise in PTSD (clinical and regulatory experience, market analysis, etc.) x Mechanism of Action (MOA) is different from TNX - 102 SL • Tianeptine sodium (amorphous) has been approved in EU, Russia, Asia and Latin America for depression since 1987 with established post - marketing experience • Identified new oxalate salt polymorph with improved pharmaceutical properties ideal for reformulation • Preliminary human pharmacokinetic and safety data (non - IND study) from selected formulation expected in second half 2019 Filed patent application on novel salt polymorph • Issued patent on steroid - induced cognitive impairment and memory loss issues Targeting a Condition with Significant Unmet Need Clinical evidence for PTSD • Several studies have shown tianeptine to be active in the treatment of PTSD 1 - 4 1 Frančišković T, et al. Psychiatr Danub . 2011 Sep;23(3):257 - 63. PMID: 21963693 2 Rumyantseva GM and, Stepanov AL. Neurosci Behav Physiol. 2008 Jan;38(1):55 - 61. PMID: 18097761 3 Aleksandrovskiĭ IA, et al. Zh Nevrol Psikhiatr Im S S Korsakova . 2005;105(11):24 - 9. PMID: 16329631 [Russian] 4 Onder E, et al. Eur Psychiatry. 2006 (3):174 - 9. PMID: 15964747

© 2019 Tonix Pharmaceuticals Holding Corp. 36 TNX - 801 (Synthesized Live Horsepox Virus): A Smallpox - Preventing Vaccine Candidate Pre - IND Stage Potential improvement over current biodefense tools against smallpox ✓ Leverages Tonix’s government affairs effort ✓ Collaboration with Professor David Evans and Dr. Ryan Noyce at University of Alberta ✓ Demonstrated protective vaccine activity in mice ✓ Patent application on novel vaccine submitted Regulatory strategy • We intend to meet with FDA to discuss the most efficient and appropriate investigational plan to support the licensure, either: ✓ Application of the “Animal Rule”, or ✓ Conducting an active comparator study using ACAM2000 • Good Manufacturing Practice (GMP) viral production process in development Targeting a Potential Public Health Issue Material threat medical countermeasure under 21 st Century Cures Act • Qualifies for Priority Review Voucher (PRV) upon licensure * ✓ PRVs have no expiration date, are transferrable and have sold for ~$125 M *BLA/NDA priority 6 - month review is expected.
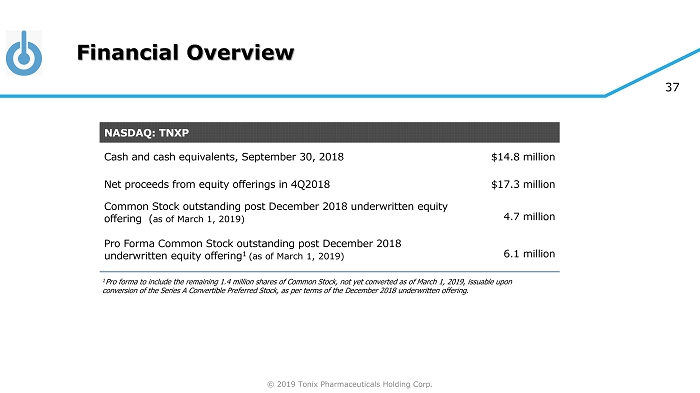
© 2019 Tonix Pharmaceuticals Holding Corp. 37 Financial Overview NASDAQ: TNXP Cash and cash equivalents, September 30, 2018 $14.8 million Net proceeds from equity offerings in 4Q2018 $17.3 million Common Stock outstanding post December 2018 underwritten equity offering ( a s of March 1, 2019) 4.7 million Pro Forma Common Stock outstanding post December 2018 underwritten equity offering 1 ( a s of March 1, 2019) 6.1 million 1 Pro forma to include the remaining 1.4 million shares of Common Stock, not yet converted as of March 1, 2019, issuable upon conversion of the Series A Convertible Preferred Stock, as per terms of the December 2018 underwritten offering.

© 2019 Tonix Pharmaceuticals Holding Corp. 38 Management Team Seth Lederman, MD President & CEO Jessica Morris Chief Operating Officer Gregory Sullivan, MD Chief Medical Officer Bradley Saenger, CPA Chief Financial Officer
© 2019 Tonix Pharmaceuticals Holding Corp. 39 Board of Directors Seth Lederman, MD Chairman Adeoye “Oye” Olukotun, MD Squibb, BMS, Mallinckrodt, Esperion John Rhodes Chair, NYS Public Service Commission, CEO, NYS Dept. of Public Service, Booz Allen James Treco First Chicago, Salomon Brothers/Citigroup Gen. David Grange (US Army, ret.) Pharm - Olam, PPD, McCormick Foundation Patrick Grace ( qp ) global family offices, Grace Institute Foundation, WR Grace, Chemed Donald Landry, MD, PhD Chair of Medicine, Columbia University Margaret Smith Bell Standard Life Investments, Putnam Investments, State Street Research

© 2019 Tonix Pharmaceuticals Holding Corp. 40 Milestones – Recently Completed and Upcoming □ July 201 8 Completed P301/HONOR study interim analysis - result did not support study continuation but strengthened new Phase 3 study □ August 201 8 Presentation of P301/HONOR study results at Military Health System Scientific Symposium □ October 201 8 Met with FDA and received preliminary agreement on the design of new Phase 3 study of Tonmya for PTSD (P302/RECOVERY study) □ November 201 8 Received FDA minutes confirming agreement on the design of P302/RECOVERY study □ First Quarter 2019 P302/RECOVERY study to be initiated □ First Quarter 2019 FM FDA Clinical Guidance meeting □ Second Half 2019 P reliminary human pharmacokinetic and safety data (non - IND study) from selected TNX - 601 (tianeptine oxalate) formulation expected □ First Half 2020 Topline data from P302/RECOVERY study expected x x x x

© 2019 Tonix Pharmaceuticals Holding Corp. 41 Summary Phase 3 development of new bedtime treatment for PTSD, including military - related PTSD • Major unmet need; ~12 million Americans annually • Benefited from FDA 505(b)(2) NDA approval requirement Complimentary day - time PTSD treatment in development • Leverages development expertise in PTSD, i.e., regulatory, trial recruitment and execution New indication in development for agitation in Alzheimer’s Disease • Unmet medical need, no approved drug available • Fast Track Phase 2/3 ready program Fibromyalgia bedtime treatment in development • IND ready to support Phase 3 potential pivotal efficacy study Innovative vaccine in development to prevent Smallpox • Opportunity to supply stockpiling requirement; short development path • Studies in mice suggest improved safety profile

© 2019 Tonix Pharmaceuticals Holding Corp. Thank you ! NASDAQ: TNXP