As Filed with the Securities and Exchange Commission on January 26, 2016 |
| Registration No. |
UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
________________
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
________________
XG Sciences, INC.
(Exact name of registrant as specified in its charter)
Michigan |
| 2821 |
| 20-4998896 |
(State or other jurisdiction of |
| (Primary Standard Industrial |
| (I.R.S. Employer Identification |
3101 Grand Oak Drive
Lansing, MI 48911
(Address, including zip code, and telephone number, including area code, of registrant’s principal executive offices)
Philip L. Rose
Chief Executive Officer
XG Sciences, Inc.
3101 Grand Oak Drive
Lansing, MI 48911
Telephone: (517) 703-1110
(Name, address, including zip code, and telephone number, including area code, of agent for service)
________________
Copies to:
Clayton E. Parker, Esq.
Matthew Ogurick, Esq.
Camielle N. Green, Esq.
K&L Gates LLP
200 South Biscayne Boulevard, Suite 3900
Miami, Florida 33131-2399
Telephone: (305) 539-3306
Facsimile: (305) 358-7095
________________
Approximate date of commencement of proposed sale to the public: As soon as practicable after this Registration Statement becomes effective.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box.x
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act of 1933, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering.¨
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act of 1933, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering.¨
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act of 1933, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering.¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer¨ |
| Accelerated filer¨ |
| Non-accelerated filer¨ |
| Smaller reporting companyx |
|
|
|
| (Do not check if a smaller reporting company) | ||
CALCULATION OF REGISTRATION FEE
Title of Each Class of Securities to be Registered |
| Amount to beRegistered |
| Proposed MaximumOffering PricePer Share(1) |
| Proposed Maximum AggregateOffering Price(1) |
| Amount ofRegistration Fee | |||
Primary Offering By XG Sciences, Inc. |
|
|
|
|
|
|
|
|
|
|
|
Common stock, no par value per share |
| 2,000,000 |
| $ | 8 |
| $ | 16,000,000 |
| $ | 1,611.20 |
Secondary Offering by selling securityholders(2) |
|
|
|
|
|
|
|
|
|
|
|
Common stock, no par value per share |
| 836,544 |
| $ | 8 |
|
|
|
|
|
|
Common stock, no par value per share, underlying warrants |
| 31,625 |
|
|
|
|
|
|
|
|
|
Common stock, no par value per share, underlying shares of Series A Preferred Stock |
| 3,376,299 |
|
|
|
|
|
|
|
|
|
Common stock, no par value per share, underlying shares of Series B Preferred Stock |
| 539,974 |
|
|
|
|
|
|
|
|
|
Total for Secondary Offering: |
| 4,784,442 |
| $ | 8 |
| $ | 38,275,536 |
| $ | 3,854.35 |
Total for Primary and Secondary Offering: |
| 6,784,442 |
|
|
|
|
|
|
| $ | 5,465.55 |
____________
(1) Calculated in accordance with Rule 457(o) under the Securities Act of 1933.
(2) This Registration Statement includes and covers the sale by selling securityholders named herein of up to 4,784,442 shares of the registrant’s common stock, including: (i) up to 836,544 shares previously issued to selling securityholders in private placements, (ii) up to 31,625 shares issuable upon exercise of warrants previously issued to selling securityholders, (iii) up to 3,376,299 shares issuable upon conversion by selling securityholders of Series A Preferred Stock previously issued to selling securityholders, and (iv) up to 539,974 shares issuable upon exchange by selling securityholders of Series B Preferred Stock previously issued to selling securityholders Pursuant to Rule 416 under the Securities Act of 1933, the shares being registered hereunder include such indeterminate number of shares of common stock as may be issuable with respect to the shares being registered hereunder as a result of stock splits, stock dividends or similar transactions. The proposed offering price per share for the Selling Stockholders was estimated solely for the purpose of calculating the registration fee pursuant to Rule 457 of Regulation C. The price of $8.00 is a fixed price at which the selling securityholders may sell their shares until our common stock is quoted on the OTC Bulletin Board or OTC Markets at which time the shares may be sold at prevailing market prices or at privately negotiated prices.
The Registrant amends this registration statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which specifically states that this registration statement shall hereafter become effective in accordance with Section 8(a) of the Securities Act of 1933, or until the registration statement shall become effective on such date as the Commission, acting pursuant to Section 8(a), may determine.
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission becomes effective. This prospectus is not an offer to sell these securities and we are not soliciting offers to buy these securities in any state where the offer or sale is not permitted.
PRELIMINARY PROSPECTUS
Subject to completion, dated , 2016
6,784,442 shares of Common Stock of
XG SCIENCES, INC.
Primary Offering
This is the initial offering of common stock of XG Sciences, Inc., and no public market currently exists for the securities being offered. We are registering for sale a total of 2,000,000 shares of common stock at a fixed price of $8.00 per share to the general public in a best efforts offering. We estimate our total offering registration costs to be approximately $740,000, provided that a sales agent is engaged, as described further herein. There is no minimum number of shares that must be sold by us for the offering to proceed, and we will retain the proceeds from the sale of any of the offered shares. The offering is being conducted on a best efforts basis and the Company intends to engage the services of a sales agent to assist it with selling the shares. If the Company engages such a sales agent, we intend to pay a commission fee of up to 8%. For additional information please see the “Plan of Distribution for Primary Offering” .
The shares will be offered at a fixed price of $8.00 per share for a period of one hundred and eighty (180) days from the effective date of this prospectus. The offering shall terminate on the earlier of (i) when the offering period ends (180 days from the effective date of this prospectus), (ii) the date when the sale of all 2,000,000 shares is completed, and (iii) when our Board of Directors decides that it is in the best interest of the Company to terminate the offering prior to the completion of the sale of all 2,000,000 shares registered under the Registration Statement of which this prospectus is part.
Secondary Offering
In addition, the selling securityholders identified in this prospectus or any of their pledges, donees, transferees or other successors-in-interests may offer to sell, from time to time, in amounts at prices and on terms determined at the time of the offering, up to 4,784,442 additional shares of our common stock under this prospectus. We will not receive any proceeds from the sale of shares by the selling securityholders, but we will incur expenses in connection with the sale of those shares, including legal and accounting fees and we will receive proceeds from the cash exercise of the warrants held by selling securityholders.
Sales by the selling securityholders may occur through ordinary brokerage transactions, directly to market makers in our shares or through any other means described in the section of this prospectus entitled “Plan of Distribution for Secondary Offering”.
The selling securityholders have not engaged any underwriter in connection with the sale of their shares of common stock.
The offering price of $8.00 is a fixed price at which the selling securityholders may sell their shares until our common stock is quoted on the Over-the-Counter Bulletin Board (the “OTC Bulletin Board”) or OTC Markets at which time the common stock may be sold at prevailing market prices or at privately negotiated prices.
There has been no market for our securities and a public market may never develop, or, if any market does develop, it may not be sustained. Our common stock is not currently quoted on or traded on any exchange or on the over-the-counter market. After the effective date of this prospectus, we hope to have a market maker file an application with the Financial Industry Regulatory Authority (“FINRA”) for our common stock to be eligible for trading on the OTC Bulletin Board. To be eligible for quotation, issuers must remain current in their quarterly and annual filings with the Securities and Exchange Commission. If we are not able to pay the expenses associated with our reporting obligations we will not be able to apply for quotation on the OTC Bulletin Board. We do not yet have a market maker who has agreed to file such application. There can be no assurance that our common stock will ever be quoted on a stock exchange or a quotation service or that any market for our stock will develop. We further intend to apply for the listing of our common stock on the NASDAQ Capital Market.
We are an “emerging growth company” as that term is used in the Jumpstart Our Business Startups Act of 2012 and, as such, may elect to comply with certain reduced public company reporting requirements for future filings. Please refer to discussions under “Prospectus Summary” on page 1 and “Risk Factors” on page 8 of how and when we may lose emerging growth company status and the various exemptions that are available to us.
Investing in our securities involves a high degree of risk. See the section entitled “Risk Factors” on page 8 of this prospectus and in the documents we filed with the Securities and Exchange Commission that are incorporated in this prospectus by reference for certain risks and uncertainties you should consider.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus. Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2016.
TABLE OF CONTENTS
| 1 | |
| 5 | |
| 8 | |
| 19 | |
| 19 | |
| 20 | |
| 21 | |
| 22 | |
| 24 | |
| 25 | |
| 26 | |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND |
| 27 |
| 45 | |
| 66 | |
| 75 | |
| 78 | |
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND |
| 80 |
| 83 | |
| 89 | |
| 99 | |
| 101 | |
| 103 | |
| 103 | |
| 103 | |
| F-1 | |
| F-46 | |
| II-1 | |
| II-8 | |
| II-8 |
You should rely only on the information contained in this prospectus. We have not, and the selling securityholders have not, authorized any person to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely on it. This prospectus is not an offer to sell, nor are the selling securityholders seeking an offer to buy, securities in any state where the offer or solicitation is not permitted. The information contained in this prospectus is complete and accurate as of the date on the front cover of this prospectus, but information may have changed since that date. We are responsible for updating this prospectus to ensure that all material information is included and will update this prospectus to the extent required by law.
i
This prospectus of XG Sciences, Inc., a Michigan corporation (together with its sole subsidiary, the “Company”, “XG Sciences”, “XGS” or “we”, “us”, or “our”) is a part of a registration statement on Form S-1 that we filed with the Securities and Exchange Commission. This summary highlights selected information contained elsewhere in this prospectus. This summary does not contain all the information that you should consider before investing in our common stock. You should carefully read the entire prospectus, including “Risk Factors”, “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and the financial statements and related notes beginning on page F-1 before making an investment decision.
XG Sciences was formed in May 2006 for the purpose of commercializing certain technology to produce graphene nanoplatelets. First discovered in 2004, graphene is a single layer of carbon atoms configured in an atomic-scale honeycomb lattice. Among many noted properties, graphene is harder than diamonds, lighter than steel but significantly stronger, and conducts electricity better than copper. Graphene nanoplatelets are particles consisting of multiple layers of graphene. Graphene nanoplatelets have unique capabilities for energy storage, thermal conductivity, electrical conductivity, barrier properties, lubricity and the ability to impart strength when incorporated into plastics or other matrices.
We believe the unique properties of graphene and graphene nanoplatelets will enable numerous new product applications and the market for such products will quickly grow to be a significant market opportunity. Our business model is to design, manufacture and sell advanced materials we call xGnP® graphene nanoplatelets and value-added products based on these nanoplatelets. We currently have hundreds of customers trialing our products for numerous applications, including, but not limited to lithium ion batteries, supercapacitors, thermal shielding and heat transfer, inks and coatings, printed electronics, construction materials, composites, and military uses.
Our proprietary manufacturing processes were developed at the Composite Materials and Structures Center in the College of Engineering of Michigan State University (“MSU”) and licensed to us in 2006. We licensed four U.S. patents and patent applications from MSU. However, over time, our scientists and engineers have made many further discoveries and inventions that are embodied in the form of ten additional patent applications, and numerous trade secrets. Our general IP strategy is to keep as trade secrets those manufacturing processes that are difficult to enforce should they be disclosed and to seek patent coverage for other manufacturing processes, materials derived from those processes, unique combinations of materials and end uses of materials containing graphene nanoplatelets. We believe that the combination of our rights under the MSU license, patents and patent applications, and our trade secrets create a strong intellectual property position.
We target our xGnP® nanoplatelets for use in a range of large and growing end-use markets. Our proprietary manufacturing processes allow us to produce nanoplatelets with varying performance characteristics that can be tuned to specific end-use applications based on customer requirements. We currently offer three commercial “grades” of bulk materials, each of which is available in various particle sizes. Other grades may be made available, depending on the needs for specific applications. In addition, we sell our material in the form of pre-dispersed mixtures with water, alcohol, or other organic solvents and resins. We also formulate xGnP® nanoplatelets into value-added products and formulations that further enhance the value we deliver to our customers. We have also licensed some of our base manufacturing technology to other companies and we consider technology licensing a component of our business model.
We sell products to customers around the world and have sold materials to over 850 customers (entities that have purchased our materials) in 47 countries since 2008. Some of these customers are research organizations and some are commercial organizations. Because graphene is a new material, our customers are developing new uses for our products and purchase them in quantities consistent with development purposes. A few of our customers have indicated to us that they have introduced commercial products that use our materials, but our customers are under no obligation to report to us on the usage of our materials. Our customers have included well-known automotive and OEM suppliers around the world (Ford, Johnson Controls, Magna, Honda Engineering) world-scale lithium ion battery manufacturers in the US, South Korea and China (Samsung SDI, LG Chem, Lishen, A123) and diverse specialty material companies (3M, BASF, Henkel, Dow Chemical, Dupont) as well as many others. We also work closely with our licensees, POSCO and Cabot Corporation (“Cabot”), who further extend our technology through their customer network. Ultimately, we expect to benefit in terms of royalties on sales of xGnP® produced and sold by our licensees.
1
|
|
|
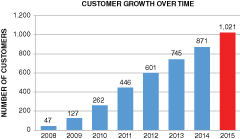
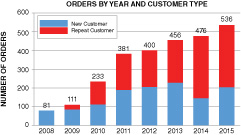
The above graphs show total orders and customers based on actual purchases of our materials and do not include free samples or materials used in joint development programs. Nevertheless, the total dollar volume of these orders is not yet sufficient to cover our fixed operating expenses. The average order size in 2014, for example, was $2,175, which indicates to us that most of these orders are for materials that are not yet incorporated into large-volume commercial products.
We also offer a sheet product, called XG Leaf®, to customers for a variety of thermal spreading and other applications. XG Leaf® is ideally suited for use in thermal management in cell phones, tablets and PC’s. As these devices continue to adopt faster electronics, higher data management capabilities, brighter displays with ever increasing definition, they generate more and more heat. Managing that heat is a key requirement for the portable electronics market and our XG Leaf® product line is well suited to address the need. In a press release dated March 3, 2015, Gartner, Inc., a leading research organization, estimated the 2014 global cell phone market at 1.88 billion units. Every cell phone has some form of thermal management system, and we believe many of the new smart phones being developed can benefit from the performance advantages we are able to achieve with XG Leaf®. In August 2015, International Data Corporation (IDC) in their Worldwide Quarterly Tablet Tracker, estimated the global shipment of tablets in 2015 at 212 million units. Thus, we believe our XG Leaf® product line is well positioned to address a very large and rapidly growing market.
According to Prismark Partners, LLC, a leading electronics industry consulting firm specializing in advanced materials, the 2014 market for finished graphitic heat spreaders as sold to the OEM and EMS companies with adhesive, PET, and/or copper backing for selected portable applications was $600 million, and is expected to reach $900 million in 2018. The market is currently in a significant expansion period driven by the demand for portable devices.
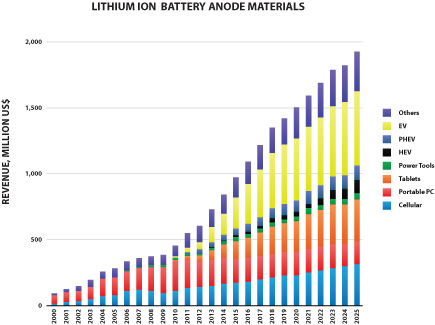
We also offer a specially formulated silicon-graphene composite material (also referred to as “SiG” in this prospectus) for use in lithium-ion battery anodes. SiG targets the never-ending need for higher battery capacity and longer life. In several customer trials, our SiG material has demonstrated the potential to increase battery energy storage capacity by 3-5x what is currently available with conventional lithium ion batteries today. The market for materials used in lithium ion battery anodes is large and growing as shown in the figure below (Avicenne Energy, “The Rechargeable Battery Market, 2014–2025”, July 2015). We believe our ability to address next generation anode materials represents a significant opportunity for us.
2
We also offer specially-formulated inks and coatings for electrical and thermal applications that are showing promise in diverse customer applications such as advanced packaging, electrostatic dissipation and thermal management.
These three product areas — custom XG Leaf® sheets, our SiG anode materials, and custom inks/coatings — comprise the set of core value-added product groups on which we are focusing our internal development resources.
We have developed and scaled-up capacity for two manufacturing processes — one based on chemical intercalation of graphite and subsequent exfoliation and classification; and the second based on a high-shear mechanical process. In March 2012, we took possession of a production facility under terms of a long-term lease and moved our headquarters to this new location. Initial production commenced in this facility in September 2012. Currently, this facility is capable of producing approximately 30 – 50 tons per year of intercalated materials (depending on product mix) if operated on a continuous basis. We also operate a separate production facility in leased manufacturing space which is used for the production of certain specialty materials. This facility is capable of producing approximately 30 – 60 tons per year of materials (depending on product mix) if operated on a continuous basis. We believe these manufacturing facilities will be sufficient to meet demands for the majority of our bulk materials for a number of years, with suitable additions of capital equipment as warranted. However, additional manufacturing capabilities for certain value-added products and certain bulk materials remain to be developed and will likely require the acquisition of additional facilities. In particular, the production processes for XG Leaf® and our silicon-graphene electrode materials and our conductive inks will require additional capital and additional facilities to meet expected future customer demand.
As of the date of this prospectus, we have not yet demonstrated sales of products at a level capable of covering our fixed expenses. Since inception, we have not demonstrated the capability to produce sufficient materials to generate the ongoing revenues necessary to sustain our operations in the long-term. Nor have we demonstrated the ability to generate sufficient sales to sustain the business. There can be no assurance that the Company will ever produce a profit.
Many of the Company’s products represent new products that have not yet been fully developed and for which manufacturing operations have not yet been fully scaled. This means that investors are subject to all the risks incident to the creation and development of multiple new products and their associated manufacturing processes, and each investor should be prepared to withstand a complete loss of their investment.
3
Because we are subject to these uncertainties, there may be risks that management has failed to anticipate and you may have a difficult time evaluating our business and your investment in our Company. Our ability to become profitable depends primarily on our ability to successfully commercialize our products in the future. Even if we successfully develop and market our products, we may not generate sufficient or sustainable revenue to achieve or sustain profitability, which could cause us to cease operations and you will lose all of your investment.
We have no sustainable base of products approved for commercial use by our customers, have never generated significant product revenues and may never achieve sufficient revenues for profitable operations, which could cause us to cease operations.
XG Sciences primarily sells bulk materials or products made with these materials to other companies for incorporation into their products. A few customers have incorporated our materials and offered them for sale in commercial markets. However, to date, there has been no incorporation of our materials or products into customer products that are released for commercial sale in sufficient volume to cover the fixed costs. Because there is limited history of commercial success for our products, it is possible that large scale adoption of our materials may never happen and that we will never achieve the level of revenues necessary to sustain our business.
Developing, manufacturing and selling nanomaterials in commercially-viable quantities requires substantial funding. As of December 31, 2014, September 30, 2015 and December 31, 2015, we had cash on hand of approximately $2.1 million, $1.6 million and $1.1 million, respectively. In addition to our cash on hand, we currently have an ongoing offering of Series B Preferred Stock, but we have no assurance that we will raise any additional funds under this offering. On December 31, 2015, we issued non-convertible promissory notes (the “December Notes”) and warrants to purchase 20,625 shares of common stock (the “December Warrants”) to several existing stockholders. The December Notes mature on June 30, 2016 and the December Warrants have a five year term and a strike price of $8.00. We cannot determine how much additional funds we might raise under the private placement. Under the terms of the private placement, the conversion price of the Series A Preferred Stock was adjusted to $6.40 per share and holders of Series B Preferred Stock received the right to exchange units each consisting of one share of Series B Preferred Stock and related warrants to purchase shares of common stock (each, a “Series B Unit”) previously purchased, for common stock at a rate of 2 shares of common stock for each Series B Unit. The Company’s financial projections show that the Company may need to raise an additional $15 million or more before it is capable of achieving sustainable cash flow from operations. There is no assurance that the Company will be able to raise these funds or that the terms and conditions of future financing will be workable or acceptable for the Company and its stockholders. In the event that the Company is not able to raise substantial additional funds in the future, the Company would be forced to cease operations and you would lose all of your investment.
Since our inception we have incurred annual losses every year and have accumulated a deficit from operations of $33,471,774 through September 30, 2015. As of September 30, 2015 our total stockholder’s deficit was $11,909,972. As of the date of this prospectus, we have cash on hand that is only sufficient to fund our operations through March 2016.
As of the date of this prospectus, we had 23 full-time employees. Employees include the following five senior managers that report to the CEO: a Vice President of Operations, a Vice President of Energy Markets, a Vice President of Research & Development, a Controller and a Senior Vice President. The Company employs a total of 6 full-time scientists and technicians in its R&D group, including the Vice President of Research & Development.
4
Common stock to be offered by the Company |
|
|
|
|
|
Common stock to be offered by the selling securityholders |
|
|
|
|
|
Secondary offering price |
| The selling securityholders may sell their shares at $8.00 per share until our shares are quoted on the OTCBB, and thereafter at prevailing market prices or privately negotiated prices. We determined this offering price arbitrarily, and the selling securityholders will be able to sell their shares once the offering is effective and would theoretically have a marketplace to sell their shares. |
|
|
|
Common stock issued and outstanding before the offerings |
|
|
|
|
|
Primary offering price |
| $8.00 per share |
|
|
|
Duration of primary offering |
| 2,000,000 shares will be offered by the Company for a period of one hundred and eighty (180) days from the effective date of this prospectus. The offering shall terminate on the earlier of (i) when the offering period ends (180 days from the effective date of this prospectus), (ii) the date when the sale of all 2,000,000 shares is completed, and (iii) when the Board of Directors decides that it is in the best interest of the Company to terminate the offering prior the completion of the sale of all 2,000,000 shares registered under the Registration Statement of which this prospectus is part. |
|
|
|
Common stock issued and outstanding after the offerings after giving effect to the sale of 2,000,000 shares by the Company and 4,784,442 shares by the selling securityholders |
|
|
|
|
|
Ticker Symbol and Market for our common stock |
|
|
5
Primary offering use of |
|
|
|
|
|
Subscriptions |
| All subscriptions, once accepted by us, are irrevocable. |
|
|
|
Risk Factors |
| The common stock offered hereby involves a high degree of risk and should not be purchased by investors who cannot afford the loss of their entire investment. See “Risk Factors” beginning on page 8. |
|
|
|
Dividend policy |
| We do not intend to pay dividends on our common stock. We plan to retain any earnings for use in the operation of our business and to fund future growth. |
There is no assurance that we will raise the full $16,000,000 anticipated from the sale by the Company of 2,000,000 shares, and there is no guarantee that we will receive any proceeds from the offering. We may sell only a small portion or none of the offered primary shares.
Unless we specifically state otherwise, the share information in this prospectus is as of December 31, 2015, and reflects or assumes:
• the exchange of each Series B Unit for 2 shares of common stock;
• the exclusion of 419,750 shares of common stock issuable upon exercise of outstanding options, at a weighted average purchase price of $12.05 per share;
• the exclusion of the additional 180,250 shares of common stock reserved for issuance pursuant to our 2007 Stock OptionPlan; and
• the exclusion of the 1,072,720 shares of common stock underlying Series A Preferred Stock underlying warrants.
Emerging Growth Company
In April 2012, the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, was enacted. Section 107 of the JOBS Act provides that an “emerging growth company,” or EGC, can take advantage of the extended transition period for complying with new or revised accounting standards. Thus, an EGC can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies.
We are in the process of evaluating the benefits of relying on other exemptions and reduced reporting requirements under the JOBS Act. Subject to certain conditions, as an EGC, we intend to rely on certain of these exemptions, including exemptions from the requirement to provide an auditor’s attestation report on our system of internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and from any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion and analysis. We will remain an EGC until the earlier of: the last day of the fiscal year in which we have total annual gross revenues of $1.0 billion or more; the last day of the fiscal year following the fifth anniversary of the date of the completion of this offering; the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
6
Corporate Information
The Company was incorporated in Michigan on May 23, 2006 and is organized as a “C” corporation under the applicable laws of the United States and State of Michigan. Our headquarters and principal executive offices are located at 3101 Grand Oak Drive, Lansing, Michigan, 48911 and our telephone number is (517) 703-1110. Our website address ishttp://www.xgsciences.com, although the information contained in, or that can be accessed through, our website is not part of this prospectus.
7
THE SECURITIES BEING OFFERED INVOLVE A HIGH DEGREE OF RISK AND, THEREFORE, SHOULD BE CONSIDERED EXTREMELY SPECULATIVE. THEY SHOULD NOT BE PURCHASED BY PERSONS WHO CANNOT AFFORD THE POSSIBILITY OF THE LOSS OF THEIR ENTIRE INVESTMENT. PROSPECTIVE INVESTORS SHOULD READ THE ENTIRE PROSPECTUS, INCLUDING ALL EXHIBITS, AND CAREFULLY CONSIDER, AMONG OTHER FACTORS, THE FOLLOWING RISK FACTORS.
Risks Relating to Our Business and Industry
We are a young company with a limited operating history, making it difficult for you to evaluate our business and your investment.
XG Sciences, Inc. was incorporated on May 23, 2006. We have not yet demonstrated sales of products at a level capable of covering our fixed expenses. Since inception, we have not demonstrated the capability to produce sufficient materials to generate the ongoing revenues necessary to sustain our operations in the long-term. Nor have we demonstrated the ability to generate sufficient sales to sustain the business. There can be no assurance that the Company will ever produce a profit.
Many of the Company’s products represent new products that have not yet been fully developed and for which manufacturing operations have not yet been fully scaled. This means that investors are subject to all the risks incident to the creation and development of multiple new products and their associated manufacturing processes, and each investor should be prepared to withstand a complete loss of their investment.
Because we are subject to these uncertainties, there may be risks that management has failed to anticipate and you may have a difficult time evaluating our business and your investment in our Company. Our ability to become profitable depends primarily on our ability to successfully commercialize our products in the future. Even if we successfully develop and market our products, we may not generate sufficient or sustainable revenue to achieve or sustain profitability, which could cause us to cease operations and you will lose all of your investment.
We have no sustainable base of products approved for commercial use by our customers, have never generated significant product revenues and may never achieve sufficient revenues for profitable operations, which could cause us to cease operations.
XG Sciences primarily sells bulk materials or products made with these materials to other companies for incorporation into their products. To date, there has been no significant incorporation of our materials or products into customer products that are released for commercial sale. Because there is no demonstrated history of commercial success for our products, it is possible that such commercial success may never happen and that we will never achieve the level of revenues necessary to sustain our business.
We will need to raise substantial additional capital in the future to fund our operations and we may be unable to raise such funds when needed and on acceptable terms, which could have a materially adverse effect on our business.
Developing, manufacturing and selling nanomaterials in commercially-viable quantities requires substantial funding. As of December 31, 2014, June 30, 2015 and December 31, 2015, we had cash on hand of approximately $2.1 million, $1.6 million and $1.1 million, respectively. In addition to our cash on hand, we currently have an ongoing offering of Series B Preferred Stock, but we have no assurance that we will raise any additional funds under this offering. The Company’s financial projections show that the Company may need to raise an additional $15 million or more before it is capable of achieving sustainable cash flow from operations. There is no assurance that the Company will be able to raise these funds or that the terms and conditions of future financing will be workable or acceptable for the Company and its stockholders. In the event that the Company is not able to raise substantial additional funds in the future, the Company would be forced to cease operations and you would lose all of your investment.
If we are unable to continue as a going concern, our securities will have little or no value.
The report of our independent registered public accounting firm that accompanies our consolidated financial statements for the year ended December 31, 2014 contains a going concern qualification in which such firm expressed substantial doubt about our ability to continue as a going concern. We currently anticipate that our cash and cash equivalents will be sufficient to fund
8
our operations through April 2016, without raising additional capital. Our continuation as a going concern is dependent upon continued financial support from our shareholders, the ability of us to obtain necessary equity and/or debt financing to continue operations, and the attainment of profitable operations. These factors raise substantial doubt regarding our ability to continue as a going concern. We cannot make any assurances that additional financings will be available to us and, if available, completed on a timely basis, on acceptable terms or at all. If we are unable to complete an equity or debt offering, or otherwise obtain sufficient financing when and if needed, it would negatively impact our business and operations, which would likely cause the price of our common stock to decline. It could also lead to the reduction or suspension of our operations and ultimately force us to cease our operations.
We have limited experience in the higher volume manufacturing that will be required to support profitable operations, and the risks associated with scaling to larger production quantities may be substantial.
We have limited experience manufacturing our products. We have established small-scale commercial or pilot-scale production facilities for our bulk powders, XG Leaf® and SiG materials, but these facilities do not have the existing production capacity to produce sufficient quantities of materials for us to reach sustainable sales levels. In order to develop the capacity to produce much higher volumes, it will be necessary to produce multiples of existing processes or engineer new production processes in some cases. There is no guarantee that we will be able to economically scale-up our production processes to the levels required. If we are unable to scale-up our production processes and facilities to support sustainable sales levels, we may be forced to cease operations and you will lose all of your investment.
Projection of fixed monthly expenses and operating losses for the near future means that investors may not earn a return on their investment or may lose all of their investment.
Because of the nature of the Company’s business, the Company projects considerable fixed expenses that lead to projected monthly deficits for the near future. Fixed manufacturing expenses to maintain production facilities, compensation expenses for scientists and other critical personnel, and ongoing rent and utilities amount to several hundred thousand dollars per month, and the Company believes that such expenses are required as a precursor to significant customer sales. However, there can be no assurance that monthly sales will ever reach a sufficient level to cover the cost of ongoing monthly expenses. If sufficient regular monthly sales are not generated to cover these fixed expenses, we will continue to experience monthly profit deficits which, if not eliminated, will require continuing new investment in the Company. If monthly deficits continue beyond levels that investors find tolerable, we may not be able to raise additional funds and may be forced to cease operations and you will lose all of your investment.
We have a long and complex sales cycle and have not demonstrated the ability to operate successfully in this environment.
It has been our experience since our inception that the average sales cycle for our products can range from one to seven years from the time a customer begins testing our products until the time that they could be successfully used in a commercial product. The product introduction timing will vary based on the target market, with automotive uses typically being toward the long end and consumer electronics toward the shorter end. We have not demonstrated a track record of success in completing customer development projects, which makes it difficult for you to evaluate the likelihood of our future success. The sales and development cycle for our products is subject to customer budgetary constraints, internal acceptance procedures, competitive product assessments, scientific and development resource allocations, and other factors beyond our control. If we are not able to successfully accommodate these factors to enable customer development success, we will be unable to achieve sufficient sales to reach profitability. In this case, the Company may not be able to raise additional funds and may be forced to cease operations and you will lose all of your investment.
We could be adversely affected by our exposure to customer concentration risk.
We are subject to customer concentration risk as a result of our reliance on a relatively small number of customers for a significant portion of our revenues. For 2014 and 2013 we had one customer whose revenue was 37% and 77%, respectively, of total revenues. In 2014 we also had another customer that represented 28% of total revenues. Due to the nature of our businessand the relatively large size of many of the applications our customers are developing, we anticipate that we will be dependent on a relatively small number of customers for the majority of our revenues for the next several years. It is possible that only one or two customers could place orders sufficient to utilize most or all of our existing manufacturing capacity. In this case, there would be a risk of significant loss of future revenues if one or more of these customers were to stop ordering our materials, which could in turn have a material adverse effect on our business and on your investment.
9
Our revenues often fluctuate significantly based on one-off orders from customers or from the recognition of grant revenues which vary from period-to-period, which may materially impact our financial results from period to period.
Because of the potential for large revenue swings from one-time large orders or grants it may be difficult to accurately forecast the needs for inventory, working capital, and other financial resources from period-to-period. Such orders would require a significant short-term increase in our production capacity and would require the financial resources to add staff and support the associated working capital. If such large one-time orders were not handled smoothly, customer confidence in us as a viable supplier could be reduced and we might not succeed in capturing the additional larger orders that may be reflected in our business plan.
We operate in an advanced technology arena where hypothesized properties and benefits of our products may not be achieved in practice, or in which technological change may alter the attractiveness of our products.
Because there is no sustained history of successful use of our products in commercial applications, there is no assurance that broad successful commercial applications may be technically feasible. Most, if not all, of the scientific and engineering data related to our products has been generated in our own laboratories or in laboratory environments at our customers or third-parties, like universities and national laboratories. It is well known that laboratory data is not always representative of commercial applications.
Likewise, we operate in a market that is subject to rapid technological change. Part of our business strategy is to monitor such change and take steps to remain technologically current, but there is no assurance that such strategy will be successful. If the Company is not able to adapt to new advances in materials sciences, or if unforeseen technologies or materials emerge that are not compatible with our products and services or that could replace our products and services, our revenues and business prospects would likely be adversely affected. Such an occurrence may have severe consequences, including the potential for our investors to lose all of their investment.
Competitors that are larger and better funded may cause the Company to be unsuccessful in selling its products.
The Company operates in a market that is expected to have significant competition in the future. Global research is being conducted by substantially larger companies who have greater financial, personnel, technical, and marketing resources. There can be no assurance that the Company’s strategy of offering better materials based on the Company’s proprietary exfoliated graphite nanoplatelets will be able to compete with other companies, many of whom will have significantly greater resources, on a continuing basis. In the event that we cannot compete successfully, the Company may be forced to cease operations and investors may lose some or all of their investment.
Because of our small size and limited operating history, we are dependent on key employees.
The Company’s operations and development are dependent upon the experience and knowledge of Philip L. Rose, our Chief Executive Officer, Michael R. Knox, our Senior Vice President, Dr. Liya Wang, Vice President of Research & Development, Robert Privette, Vice President of Energy Markets, Scott Murray, Vice President of Operations, Dr. Inhwan Do, Technical Director, and Dr. Hiroyuki Fukushima, Technical Director. If the services of any of these individuals should become unavailable, the Company’s business operations might be adversely affected. If several of these individuals became unavailable at the same time, the ability of the Company to continue normal business operations might be adversely affected to the extent that revenue or profits could be diminished and investors could lose some or all of their investment.
Our success depends in part on our ability to protect our intellectual property rights, and our inability to enforce these rights could have a material adverse effect on our competitive position.
We rely on the patent, trademark, copyright and trade-secret laws of the United States and the countries where we do business to protect our intellectual property rights. We may be unable to prevent third parties from using our intellectual property without our authorization. The unauthorized use of our intellectual property could reduce any competitive advantage we have developed, reduce our market share or otherwise harm our business. In the event of unauthorized use of our intellectual property, litigation to protect or enforce our rights could be costly, and we may not prevail.
Many of our technologies are not covered by any patent or patent application, and our issued and pending U.S. and non-U.S. patents may not provide us with any competitive advantage and could be challenged by third parties. Our inability to secure issuance of our pending patent applications may limit our ability to protect the intellectual property rights these pending patent applications were intended to cover. Our competitors may attempt to design around our patents to avoid liability for
10
infringement and, if successful, our competitors could adversely affect our market share. Furthermore, the expiration of our patents may lead to increased competition.
Our pending trademark applications may not be approved by the responsible governmental authorities and, even if these trademark applications are granted, third parties may seek to oppose or otherwise challenge these trademark applications. A failure to obtain trademark registrations in the United States and in other countries could limit our ability to protect our products and their associated trademarks and impede our marketing efforts in those jurisdictions.
In addition, effective patent, trademark, copyright and trade secret protection may be unavailable or limited in some foreign countries. In some countries, we do not apply for patent, trademark or copyright protection. We also rely on unpatented proprietary manufacturing expertise, continuing technological innovation and other trade secrets to develop and maintain our competitive position. Although we generally enter into confidentiality agreements with our employees and third parties to protect our intellectual property, these confidentiality agreements are limited in duration and could be breached, and may not provide meaningful protection of our trade secrets or proprietary manufacturing expertise. Adequate remedies may not be available if there is an unauthorized use or disclosure of our trade secrets and manufacturing expertise. In addition, others may obtain knowledge about our trade secrets through independent development or by legal means. The failure to protect our processes, apparatuses, technology, trade secrets and proprietary manufacturing expertise, methods and compounds could have a material adverse effect on our business by jeopardizing critical intellectual property.
Where a product formulation or process is kept as a trade secret, third parties may independently develop or invent and patent products or processes identical to our trade-secret products or processes. This could have an adverse impact on our ability to make and sell products or use such processes and could potentially result in costly litigation in which we might not prevail.
We could face intellectual property infringement claims that could result in significant legal costs and damages and impede our ability to produce key products, which could have a material adverse effect on our business, financial condition and results of operations.
If we fail to maintain effective internal controls over financial reporting, the price of our common stock may be adversely affected.
We are required to establish and maintain appropriate internal controls over financial reporting. Failure to establish those controls, or any failure of those controls once established, could adversely impact our public disclosures regarding our business, financial condition or results of operations. Any failure of these controls could also prevent us from maintaining accurate accounting records and discovering accounting errors and financial frauds. Rules adopted by the U.S. Securities and Exchange Commission (“SEC”) pursuant to Section 404 of the Sarbanes-Oxley Act of 2002 require annual assessment of our internal control over financial reporting, and the standards that must be met for management to assess the internal control over financial reporting as effective are new and complex, and require significant documentation, testing and possible remediation to meet the detailed standards. We may encounter problems or delays in completing activities necessary to make an assessment of our internal control over financial reporting. If we cannot assess our internal control over financial reporting as effective, investor confidence and share value may be negatively impacted.
In addition, management’s assessment of internal controls over financial reporting may identify weaknesses and conditions that need to be addressed in our internal controls over financial reporting or other matters that may raise concerns for investors. Any actual or perceived weaknesses and conditions that need to be addressed in our internal control over financial reporting, disclosure of management’s assessment of our internal controls over financial reporting, or disclosure of our independent registered public accounting firm’s report on management’s assessment of our internal controls over financial reporting may have an adverse impact on the price of our common stock.
Future adverse regulations could affect the viability of the business.
The Company’s bulk products have been approved for sale in the United States by the U.S. Environmental Protection Agency after a detailed review of our products and production processes for our H, M and C grade materials. In most cases, as far as we are aware, there are no current regulations elsewhere in the world that prevent or prohibit the sale of the Company’s products. Nevertheless, the sale of nano-materials is a subject of regulatory discussion and review in many countries around the world. In some cases, there is a discussion of potential testing requirements for toxicity or other health effects of nano-materials before they can be sold in certain jurisdictions. If such regulations are enacted in the future, the Company’s business could be adversely affected because of the requirement for expensive and time-consuming tests or other regulatory compliance. There can be no assurance that future regulations might not severely limit or even prevent the sale of the Company’s products in major markets, in which case the Company’s financial prospects might be severely limited, causing investors to lose some or all of their investment.
11
Compliance with changing regulation of corporate governance and public disclosure will result in additional expenses and will divert time and attention away from revenue generating activities.
Changing laws, regulations and standards relating to corporate governance and public disclosure, including the Sarbanes-Oxley Act of 2002 and related SEC regulations, have created uncertainty for public companies and significantly increased the costs and risks associated with accessing the public markets and public reporting. Our management team will need to invest significant management time and financial resources to comply with both existing and evolving standards for public companies, which will lead to increased general and administrative expenses and a diversion of management time and attention from revenue generating activities to compliance activities, which could have an adverse effect on our business.
Risks Relating To Our Common Stock
There is a risk of dilution of your percentage ownership of common stock in the Company.
In addition to the shares which we may sell pursuant to this offering, the Company has the right to raise additional capital or incur borrowings from third parties to finance its business. The Company may also implement public or private mergers, business combinations, business acquisitions and similar transactions pursuant to which it would issue substantial additional capital stock to outside parties, causing substantial dilution in the ownership of the Company by its existing stockholders. Our Board of Directors has the authority, without the consent of any of the stockholders, to cause the Company to issue more shares of common stock and/or preferred stock at such price and on such terms and conditions as are determined by the Board in its sole discretion.
The sale of the shares being offered by us hereunder, as well as the shares of common stock and Series A Preferred Stock issuable upon the exercise of warrants, the shares issuable upon conversion of Series A Preferred Stock, the shares issuable upon the exercise of options and warrants, shares of Series A Preferred Stock which were issued upon the conversion of secured convertible notes on December 31, 2015, shares issuable upon conversion of Series B Preferred Stock, shares issuable upon exchange of the Series B Units, and the issuance of additional shares of capital stock by the Company will dilute your ownership percentage in the Company and could impair our ability to raise capital in the future through the sale of equity securities.
Certain stockholders who are also officers and directors of the Company may have significant control over our management, which may not be in your best interests.
The directors, or the entities they represent, and executive officers of the Company owned more than 44% of the voting stock of the Company on December 31, 2015. Furthermore, these same executives and directors, or the companies they represent, converted secured convertible notes into an additional 1,456,128 shares of the Series A Preferred Stock in the Company on December 31, 2015. As of December 31, 2015, assuming the sale of no additional new equity before then, the percentage of common stock controlled by directors, or the companies they represent, and officers of the Company represented approximately 72% of total voting stock outstanding. As a result, such entities have a significant influence on the affairs and management of the Company, as well as on all matters requiring stockholder approval, including electing and removing members of the Company’s Board of Directors, causing the Company to engage in transactions with affiliated entities, causing or restricting the sale or merger of the Company, and certain other matters. Such concentration of ownership and control could have the effect of delaying, deferring or preventing a change in control of the Company even when such a change of control would be in the best interests of the Company’s stockholders.
We may, in the future, issue additional shares of common stock, which would reduce investors’ percent of ownership and may dilute our share value.
Our Articles of Incorporation, as amended, authorize the issuance of up to 25,000,000 shares of common stock and up to 8,000,000 shares of preferred stock. As of December 31, 2015, the Company had 836,544 shares of common stock, 1,800,696 shares of Series A Preferred Stock and 269,987 shares of Series B Preferred Stock issued and outstanding. We intend to convert our Series A Preferred Stock at a per share ratio of 1.875 to 1, which would result in a post-conversion common stock underlying Series A Preferred Stock of 3,376,299 shares. In addition to the 269,987 shares of Series B Preferred Stock issued, holders of Series B Preferred Stock have the right, at their discretion, to exchange each Series B Unit for two shares of common stock which result in the issuance of 539,974 shares of common stock. As of the date of this prospectus, no Series B Unit holder has exchanged their Series B Units into shares of common stock.
In addition, all outstanding secured convertible notes converted into 1,456,126 shares of Series A Preferred Stock on December 31, 2015. As of December 31, 2015, the Company had also granted options to purchase up to 419,750 shares of
12
common stock and had issued warrants to purchase up to 256,522 shares of common stock and 1,072,720 shares of Series A Preferred Stock. Each holder of a Series B Unit will exchange his, her or its Series B Unit for two shares of the Company’s common stock in accordance with the terms of the Certificate of Designations of Series B Convertible Preferred Stock (“Series B Designations”), which will result in an additional issuance of common stock of 539,974 shares. In total, therefore, we have already issued or committed to issue up to 6,276,912 shares of common stock (assuming the exchange of Series B Preferred Stock and the conversion of Series A Preferred Stock) and we are authorized to issue up to 18,723,088 additional shares of common stock. The future issuance of common stock may result in substantial dilution in the percentage of our common stock held by our then existing stockholders. We may value any common stock issued in the future on an arbitrary basis. The issuance of common stock for future services or acquisitions or other corporate actions may have the effect of diluting the value of the shares held by our investors, might have an adverse effect on any trading market for our common stock and could impair our ability to raise capital in the future through the sale of equity securities.
We have a large number of restricted shares outstanding, a portion of which may be sold under Rule 144, which may reduce the market price of our shares.
Of the 836,544 shares of common stock issued and outstanding as of December 31, 2015, the 1,800,696 outstanding shares of Series A Preferred Stock and the 269,987 shares of Series B Preferred Stock that we intend to convert into shares of common stock upon the listing of our common stock on a Qualified National Exchange (as defined in the Certificate of Designations of Series A Convertible Preferred Stock “Series A Designations”, see also “Description of Securities — Preferred Stock”), and assuming no notes, warrants or stock options are converted or exercised, 762,770 shares are held by non-affiliates and 2,144,547 are held by affiliates of the Company. The 2,144,547 shares held by affiliates are deemed “restricted securities” within the meaning of Rule 144 as promulgated under the Securities Act. Our Series A Preferred Stock will be converted at a per share ratio of 1.875 to 1, which would result in a post-conversion common stock underlying Series A Preferred Stock of 3,376,299 shares. In addition to the 269,987 shares of Series B Preferred Stock issued, holders of Series B Preferred Stock have the right, at their discretion, to exchange each Series B Unit for two shares of common stock which result in the issuance of 539,974 shares of common stock.
It is anticipated that all of the “restricted securities” will be eligible for resale under Rule 144. In general, under Rule 144, subject to the satisfaction of certain other conditions, a person, who is not an affiliate (and who has not been an affiliate for a period of at least three months immediately preceding the sale) and who has beneficially owned restricted shares of our common stock for at least six months is permitted to sell such shares without restriction, provided that there is sufficient public information about us as contemplated by Rule 144. An affiliate who has beneficially owned restricted shares of our common stock for a period of at least one year may sell a number of shares equal to one percent of our issued and outstanding common stock approximately every three months.
The possibility that substantial amounts of our common stock may be sold under Rule 144 into the public market may adversely affect prevailing market prices for the common stock and could impair our ability to raise capital in the future through the sale of equity securities.
The Company is considered a smaller reporting company and is exempt from certain disclosure requirements, which could make our stock less attractive to potential investors.
Rule 12b-2 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”) defines a “smaller reporting company” as an issuer that is not an investment company, an asset-backed issuer, or a majority-owned subsidiary of a parent that is not a smaller reporting company and that:
• Had a public float of less than $75 million as of the last business day of its most recently completed second fiscal quarter, computed by multiplying the aggregate worldwide number of shares of its voting and non-voting common equity held by non-affiliates by the price at which the common equity was last sold, or the average of the bid and asked prices of common equity, in the principal market for the common equity; or
• In the case of an initial registration statement under the Securities Act or Exchange Act for shares of its common equity, had a public float of less than $75 million as of a date within 30 days of the date of the filing of the registration statement, computed by multiplying the aggregate worldwide number of such shares held by non-affiliates before the registration plus, in the case of a Securities Act registration statement, the number of such shares included in the registration statement by the estimated public offering price of the shares; or
13
• In the case of an issuer whose public float as calculated under paragraph (1) or (2) of this definition was zero, had annual revenues of less than $50 million during the most recently completed fiscal year for which audited financial statements are available.
As a “smaller reporting company” (in addition to and without regard to our status as an “emerging growth company”) we are not required and may not include a “Compensation Discussion and Analysis” section in our proxy statements; we provide only 3 years of business development information; provide fewer years of selected financial data; and have other “scaled” disclosure requirements that are less comprehensive than issuers that are not “smaller reporting companies” which could make our stock less attractive to potential investors, which could make it more difficult for you to sell your shares.
The Company is considered an “emerging growth company” and we cannot be certain if the reduced disclosure requirements applicable to emerging growth companies will make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart our Business Startups Act of 2012, and we may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies, including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved.
We will remain an emerging growth company until the earlier of (i) the last day of the fiscal year (A) following the fifth anniversary of our first sale of common equity securities pursuant to an effective registration statement, (B) in which we have total annual gross revenue of at least $1.0 billion, or (C) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Exchange Act, which would occur if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter, and (ii) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.
We cannot predict if investors will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile when trading occurs.
We will become subject to the periodic reporting requirements of the Exchange Act, which will require us to incur audit fees and legal fees in connection with the preparation of such reports. These additional costs will negatively affect our ability to earn a profit.
Following the effective date of the registration statement in which this prospectus is included, we will be required to file periodic reports with the Securities and Exchange Commission pursuant to the Exchange Act and the rules and regulations thereunder. In order to comply with such requirements, our independent registered auditors will have to review our financial statements on a quarterly basis and audit our financial statements on an annual basis. Moreover, our legal counsel will have to review and assist in the preparation of such reports. Factors such as the number and type of transactions that we engage in and the complexity of our reports cannot accurately be determined at this time and may have a major negative effect on the cost and amount of time to be spent by our auditors and attorneys. However, the incurrence of such costs will be an expense to our operations and thus have a negative effect on our ability to meet our overhead requirements and earn a profit.
However, for as long as we remain an “emerging growth company” we intend to take advantage of certain exemptions from various reporting requirements until we are no longer an “emerging growth company.”
We also qualify as a smaller reporting company, and so long as we remain a smaller reporting company, we benefit from the same exemptions and exclusions as an emerging growth company. In the event that we cease to be an emerging growth company as a result of a lapse of the five year period, but continue to be a smaller reporting company, we would continue to be subject to the exemptions available to emerging growth companies until such time as we were no longer a smaller reporting company.
After, and if ever, we are no longer an “emerging growth company,” we expect to incur significant additional expenses and devote substantial management effort toward ensuring compliance with those requirements applicable to companies that are not “emerging growth companies,” including Section 404 of the Sarbanes-Oxley Act.
14
For so long as we are an emerging growth company, we may rely on certain exemptions provided in the JOBS Act, including reduced disclosure regarding executive compensation, not seeking an advisory vote with respect to executive compensation and not requiring our independent registered public accounting firm to attest to the effectiveness of our internal control over financial reporting, which could make our common stock less attractive to investors due to the nature of the reduced disclosure.
We are an “emerging growth company,” as defined in the JOBS Act, and may remain an emerging growth company for up to five years. For so long as we remain an emerging growth company, we are permitted and plan to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, or SOX Section 404, not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, reduced disclosure obligations regarding executive compensation and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. In this prospectus, we have provided only two years of audited financial statements and have not included all of the executive compensation related information that would be required if we were not an emerging growth company. We cannot predict whether investors will find our common stock less attractive if we rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
If securities or industry analysts do not publish research or reports or publish unfavorable research about our business, the price and trading volume of our common stock could decline.
The trading market for our common stock will depend in part on the research and reports that securities or industry analysts publish about us or our business. We do not currently have and may never obtain research coverage by securities and industry analysts. If no securities or industry analysts commence coverage of us, the trading price for our common stock and other securities would be negatively affected. In the event we obtain securities or industry analyst coverage, if one or more of the analysts who covers us downgrades our securities, the price of our securities would likely decline. If one or more of these analysts ceases to cover us or fails to publish regular reports on us, interest in the purchase of our securities could decrease, which could cause the price of our common stock and other securities and their trading volume to decline.
Because our common stock is “penny stock,” you may have greater difficulty selling your shares.
Penny stocks are generally equity securities with a price of less than $5.00, other than securities registered on certain national securities exchanges or quoted on the Nasdaq system, provided that current price and volume information with respect to transactions in such securities is provided by the exchange or quotation system. Because our securities constitute “penny stocks” within the meaning of the rules, the rules apply to us and to our securities. The rules may further affect the ability of owners of shares to sell our securities in any market that might develop for them. As long as the trading price of our common stock is less than $5.00 per share, even if our common stock is quoted on the OTCBB or the OTC Markets, the common stock will be subject to Rule 15g-9 under the Exchange Act. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock, to deliver a standardized risk disclosure document prepared by the SEC, that:
• contains a description of the nature and level of risk in the market for penny stocks in both public offerings and secondary trading;
• contains a description of the broker’s or dealer’s duties to the customer and of the rights and remedies available to the customer with respect to a violation to such duties or other requirements of securities laws;
• contains a brief, clear, narrative description of a dealer market, including bid and ask prices for penny stocks and the significance of the spread between the bid and ask price;
• contains a toll-free telephone number for inquiries on disciplinary actions;
15
• defines significant terms in the disclosure document or in the conduct of trading in penny stocks; and
• contains such other information and is in such form, including language, type, size and format, as the SEC shall require by rule or regulation.
The broker-dealer also must provide, prior to effecting any transaction in a penny stock, the customer with: (a) bid and offer quotations for the penny stock; (b) the compensation of the broker-dealer and its salesperson in the transaction; (c) the number of shares to which such bid and ask prices apply, or other comparable information relating to the depth and liquidity of the market for such stock; and (d) a monthly account statements showing the market value of each penny stock held in the customer’s account. In addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from those rules; the broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written acknowledgment of the receipt of a risk disclosure statement, a written agreement to transactions involving penny stocks, and a signed and dated copy of a written suitably statement. These disclosure requirements may have the effect of reducing the trading activity in the secondary market for our stock.
Because we do not intend to pay any cash dividends on our common stock, our stockholders will not be able to receive a return on their shares unless they sell them.
We intend to retain any future earnings to finance the development and expansion of our business. We do not anticipate paying any cash dividends on our common stock in the foreseeable future. Unless we pay dividends, our stockholders will not be able to receive a return on their shares unless they sell them. There is no assurance that stockholders will be able to sell shares when desired.
Downturns in general economic conditions could adversely affect our profitability.
Downturns in general economic conditions can cause fluctuations in demand for our products, product prices, volumes and gross margins. Future economic conditions may not be favorable to our industry. A decline in the demand for our products or a shift to lower-margin products due to deteriorating economic conditions could adversely affect sales of our products and our profitability and could also result in impairments of certain of our assets.
Furthermore, any uncertainty in economic conditions may result in a slowdown to the global economy that could affect our business by reducing the prices that our customers may be able or willing to pay for our products or by reducing the demand for our products.
An increase in the cost of raw materials or electricity might affect our profits.
Any increase in the prices of our raw materials or energy might affect the overall cost of our products. If we are not able to raise our prices to pass on increased costs to our customers, we would be unable to maintain our existing profit margins. Our major cost components include items such as graphite, sulfuric acid, and electricity, which items are normally readily available industrial commodities. During our history as a business, we have not seen any material impact (as defined by GAAP) on our cost structure from fluctuations in raw material or energy costs, but this could change in the future.
Our results of operations could deteriorate if our manufacturing operations were substantially disrupted for an extended period.
Our manufacturing operations are subject to disruption due to extreme weather conditions, floods and similar events, major industrial accidents, strikes and lockouts, adoption of new laws or regulations, changes in interpretations of existing laws or regulations or changes in governmental enforcement policies, civil disruption, riots, terrorist attacks, war, and other events. We cannot assure you that no such events will occur. If such an event occurs, it could have a material adverse affect on us.
Significant risks may be associated with future acquisitions.
Our Company may pursue the acquisition of other companies or product lines in order to add new businesses and/or product lines or simply complement our existing businesses and product lines. Along with such acquisitions come certain risks, including, but not limited to the following:
• the difficulty of identifying appropriate acquisition candidates;
• the difficulty of assimilating the operations and personnel of the acquired entities;
16
• the potential disruption of our ongoing business;
• the potential incurrence of new debt or the issuance of new equity that could increase our leverage or dilute our stockholders’ equity interests;
• our inability to capitalize on the opportunities presented by acquisitions; and
• our failure to implement and maintain uniform standards, controls, procedures and policies at any acquired businesses.
Given our limited resources, we may not effectively manage our growth.
There is no guarantee that we have the resources, financial or operational, required to manage our growth. This is particularly true as we expand facilities and manufacture our products on a greater commercial scale. Furthermore, rapid growth in our operations may place a significant strain on our management, administrative, operational and financial infrastructure. The inability to adequately manage our growth could have a material and adverse affect on our business, financial condition or results of operations, thus resulting in a lower quoted price of our common stock.
Risks Related to this Offering
Our Shareholder Agreement contains certain negative covenants and restrictions on transfer, and investors in this Offering will be required to adopt the Shareholder Agreement until such time as the Company raises $10,000,000 in a registered public offering, which could restrict such investor’s ability to sell shares of the Company’s common stock and engage in certain businesses.
On March 18, 2013, the Company and certain shareholders therein executed a Shareholders’ Agreement that contains restrictions on transfer and a number of specific provisions pertaining to the Board of Directors as well as individual Directors. Among other things, the Shareholder Agreement provides that certain of our larger shareholders have voting and nomination rights, and grants preemptive rights to shareholders. The Shareholder Agreement also gives a right of first refusal first to the Company and then to shareholders, including in the case of deceased stockholders.
Pursuant to the Shareholder Agreement, shareholders may not, while such person is a shareholder, directly or indirectly, either alone or in association with others, in any other capacity: (i) conduct, become engaged or interested in, any business that competes, directly or indirectly, with the business of the Company or any of its affiliates worldwide; (ii) solicit, divert or take away or attempt to solicit, divert or take away, directly or indirectly, any of the Company’s or its affiliates current, prior, or prospective customers: or (iii) solicit or attempt to solicit any person who is employed or engaged to perform services by the Company or its affiliates to leave his or her employment or engagement with Company or its Affiliates. After such shareholder ceases to be a shareholder, the former shareholder may not for a period of three (3) years from the date of sale, directly or indirectly, either alone or in association with others, for himself or herself or in any other capacity: (i) conduct, become engaged in, or interested in, any business that competes, directly or indirectly, with the business of the Company or any of its affiliates worldwide; (ii) solicit, divert or take away or attempt to solicit, divert or take away, directly or indirectly, any of the Company’s or its affiliates current, prior, or prospective customers; or (iii) solicit or attempt to solicit any person who is employed or engaged to perform services by the Company or its affiliates to leave his or her employment or engagement with Company or its affiliates. These restrictions on competition and solicitation will not be violated, however, by a shareholder’s passive ownership of up to 2% of a class of outstanding publicly traded shares of a corporation that is listed or quoted on a national securities exchange including any such corporation that competes with the business of the Company.
The Shareholder Agreement continues in full force and effect until the date of the closing of a public offering of our common stock pursuant to a registration statement filed with the SEC is declared effective in which the Company receives gross proceeds of at least $10,000,000, on which date it shall terminate in its entirety, unless the Shareholder Agreement is earlier terminated in accordance with its terms. Until such time as it is terminated, however, new shareholders will be required to execute an adoption agreement to the Shareholder Agreement.
Until such time as the Company raises $10,000,000 or the Shareholder Agreement is otherwise terminated, investors in the primary offering will be subject to the Shareholder Agreement and will be bound by its terms. By adopting the Shareholder Agreement, investors may not be able to sell their shares due to the restrictions on transfer, including the right of first refusal. In addition, such investors may be unable to invest freely in business ventures that may be deemed competitive with the Company’s business.
17
Because the offering price for the primary shares has been arbitrarily set by the Company, you may not realize a return on your investment upon the resale of your shares.
The offering price of the primary offering shares and other terms and conditions relative to the Company’s shares have been arbitrarily determined by us and do not bear any relationship to assets, earnings, book value or any other objective financial criteria. Additionally, as the Company was formed on May 23, 2006, and has only a limited operating history with no earnings, the price of the offered shares is not based on its past earnings, and no investment banker, appraiser, or other independent third party, has been consulted concerning the offering price for the shares or the fairness of the offering price used for the shares, as such our stockholders may not be able to receive a return on their investment when they sell their shares of common stock.
We are selling 2,000,000 shares in this offering and may engage the assistance of a sales agent; we may be unable to sell any shares.
We intend to engage the services of a sales agent to sell the shares for a commission of up to 8%. There is no guarantee that the sales agent will be able to sell all of the shares. Unless the sales agent is successful in receiving the proceeds in the amount of $16,000,000 from this offering, we may have to seek alternative financing to implement our business plan.
Our Chief Executive Officer, Mr. Philip L. Rose, has limited prior experience offering and selling securities to the public, and our offering does not require a minimum amount to be raised; as a result, we may not be able to raise sufficient funds to commence and sustain our business and investors may lose their entire investment.
If we determine not to engage a sales agent, our Chief Executive Officer, Philip Rose, will conduct the offering on our behalf. We are conducting a best efforts offering which does not require a minimum amount to be raised. Mr. Rose has limited experience conducting a public securities best efforts offering and we may not be able to successfully raise any funds. If we are not able to raise sufficient funds, we may not be able to fund our operations as planned, and our business will suffer and your investment may be materially adversely affected. Our inability to successfully conduct a best efforts offering could be the basis of your losing your entire investment in us.
Due to the lack of trading market for our securities, you may have difficulty selling any shares you purchase in this offering.
We are not registered on any market or public stock exchange. There is presently no demand for our common stock and no public market exists for the shares being offered in this prospectus. We plan to contact a market maker immediately following the completion of the offering and apply to have the shares quoted on the Over-the-Counter Bulletin Board (“OTCBB”) or the OTC Markets.
The OTCBB is a regulated quotation service that displays real-time quotes, last sale prices and volume information in over-the-counter securities. The OTCBB is not an issuer listing service, market or exchange. Although the OTCBB does not have any listing requirements, to be eligible for quotation on the OTCBB, issuers must remain current in their filings with the SEC or applicable regulatory authority. If we are not able to pay the expenses associated with our reporting obligations we will not be able to apply for quotation. Market makers are not permitted to begin quotation of a security whose issuer does not meet this filing requirement. Securities already quoted on the OTCBB that become delinquent in their required filings will be removed following a 30 to 60 day grace period if they do not make their required filing during that time. We cannot guarantee that our application will be accepted or approved and our stock listed and quoted for sale. As of the date of this filing, there have been no discussions or understandings between XG Sciences, Inc. and anyone acting on our behalf, with any market maker regarding participation in a future trading market for our securities. If no market is ever developed for our common stock, it will be difficult for you to sell any shares you purchase in this offering. In such a case, you may find that you are unable to achieve any benefit from your investment or liquidate your shares without considerable delay, if at all. In addition, if we fail to have our common stock quoted on a public trading market, your common stock will not have a quantifiable value and it may be difficult, if not impossible, to ever resell your shares, resulting in an inability to realize any value from your investment.
The OTC Market is a privately owned, interdealer quotation service that permits market participants to quote over-the-counter equity securities. The OTC Market is a significantly more limited market than a national securities exchange such as the New York Stock Exchange or NASDAQ, with generally lower trading volumes and higher price volatility. Broker-dealers often decline to trade in OTC Market stocks given that the market for such securities is often limited, the stocks are more volatile, and the risk to investors is greater than with stocks listed on other national securities exchanges. The OTC Markets group consists of three tiers, two of which require that issuers are current in their reporting obligations. We have not applied for quotation on the OTC Markets. An over-the-counter equity security can be quoted simultaneously on both OTCBB and OTC Markets. If our common stock were quoted on the OTC Market, the liquidity and price of our shares could be limited and that could adversely impact our ability to raise capital in the future. Accordingly, investors must be able to bear the financial risks associated with losing their entire investment in our common stock.
18
This prospectus contains forward-looking statements which relate to future events or our future financial performance. In some cases, you can identify forward-looking statements by terminology such as “may,” “should,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “predicts,” “potential” or “continue” or the negative of these terms or other comparable terminology. These statements are only predictions and involve known and unknown risks, uncertainties and other factors, including the risks in the section entitled “Risk Factors” that may cause our or our industry’s actual results, levels of activity, performance or achievements to be materially different from any future results, levels of activity, performance or achievements expressed or implied by these forward-looking statements. In addition, you are directed to factors discussed in the Business section and in the Management’s Discussion of Financial Condition and Results of Operations section and those discussed elsewhere in this prospectus.
Although these forward-looking statements, and any assumptions upon which they are based, are made in good faith and reflect our current judgment regarding the direction of our business, actual results will almost always vary, sometimes materially, from any estimates, predictions, projections, assumptions or other future performance suggested herein. Except as required by applicable law, including by the securities laws of the United States, we do not intend to update any of the forward-looking statements to conform these statements to actual results.
MARKET AND INDUSTRY DATA AND FORECASTS
This prospectus includes estimates of market share and industry data and forecasts that we obtained from industry publications and surveys and/or internal company sources. Industry publications and surveys and forecasts generally state that the information contained therein has been obtained from sources believed to be reliable, but there can be no assurance as to the accuracy or completeness of included information. We have not independently verified any of the data from third-party sources, nor have we ascertained the underlying economic assumptions relied upon therein. Similarly, while we believe our internal estimates with respect to our industry are reliable, our estimates have not been verified by any independent sources. Although we are not aware of any misstatements regarding any industry data presented in this prospectus, our estimates, in particular as they relate to market share and our general expectations, involve risks and uncertainties and are subject to change based on various factors, including those discussed under the section entitled “Risk Factors” beginning on page 8. Unless otherwise noted, all market share data is based on net sales in the applicable market.
19
Our primary offering is being made on a best efforts basis: no minimum number of shares must be sold in order for the offering to proceed. The offering price per share is $8.00. The following table sets forth the uses of proceeds assuming the sale of 10%, 25%, 50%, 75% and 100%, respectively, of the securities offered for sale by the Company. There is no assurance that we will raise the full $16,000,000 as anticipated and there is no guarantee that we will receive any proceeds from the offering. We intend to use the estimated net proceeds from the sale of the shares of common stock being offered by the Company hereunder as follows:
Description |
| If 10% of Primary Shares are Sold |
| If 25% of Primary Shares are Sold |
| If 50% of Primary Shares are Sold |
| If 75% of Primary Shares are Sold |
| If 100% of Primary Shares are Sold | |||||
GROSS PROCEEDS |
| $ | 1,600,000 |
| $ | 4,000,000 |
| $ | 8,000,000 |
| $ | 12,000,000 |
| $ | 16,000,000 |
Offering Expenses |
| $ | 100,000 |
| $ | 100,000 |
| $ | 100,000 |
| $ | 100,000 |
| $ | 100,000 |
Offering Expenses Attributable to Sales Agent* |
| $ | 64,000 |
| $ | 160,000 |
| $ | 320,000 |
| $ | 480,000 |
| $ | 640,000 |
NET PROCEEDS |
| $ | 1,436,000 |
| $ | 3,740,000 |
| $ | 7,580,000 |
| $ | 11,420,000 |
| $ | 15,260,000 |
Capital Expenditures |
|
| — |
| $ | 500,000 |
| $ | 2,000,000 |
| $ | 4,000,000 |
| $ | 7,000,000 |
Working Capital |
|
| — |
| $ | 500,000 |
| $ | 1,000,000 |
| $ | 2,000,000 |
| $ | 3,000,000 |
General Corporate Expenses |
| $ | 1,436,000 |
| $ | 2,740,000 |
| $ | 4,580,000 |
| $ | 5,420,000 |
| $ | 5,260,000 |
____________
* The above estimates assume that we engage the services of a sales agent at a commission of 8%.
The above estimates assume that, if the primary offering is completely successful and we raise gross proceeds of $16 million, we will fund general corporate expenses of approximately $5.2 million over the next two years, by which time the Company might begin generating positive cash flow from operations, we will fund working capital increases of $3 million, and we will spend the remaining funds to expand capacity during the two year period following the close of the offering. On the other hand, if we sell only 10% of the primary shares, we will use all net proceeds from the offering to fund general corporate expenses. In between these two scenarios, we would plan to primarily fund general corporate expenses but would begin to invest in working capital and capacity expansion with the remaining funds. Pending such use, we reserve the right to temporarily invest the proceeds.
We will not receive any proceeds from the sale of the shares of common stock by the selling securityholders. The proceeds from the cash exercise of warrants by selling securityholders, if any, will be used by us for working capital and other general corporate purposes.
20
The following table sets forth our cash and cash equivalents and our capitalization as of September 30, 2015:
Cash and cash equivalents |
| $ | 1,585,105 |
|
|
|
|
|
|
Convertible notes payable, net: |
| $ | 11,803,934 |
|
Derivative liablity – warrants |
| $ | 5,812,856 |
|
Series A convertible preferred stock, 3,000,000 shares authorized, 339,430 shares issued and outstanding, liquidation value of $4,020,600 |
| $ | 3,732,701 |
|
Series B convertible preferred stock, 3,000,000 shares authorized, 269,987 shares issued and outstanding, liquidation value of $4319,792 |
| $ | 3,651,533 |
|
Common stock, no par value, 25,000,000 shares authorized and 836,544 shares outstanding |
| $ | 8,565,225 |
|
Additional paid-in-capital |
| $ | 5,612,343 |
|
Accumulated deficit |
| $ | (33,471,774 | ) |
Total stockholders’ deficit |
| $ | (11,909,972 | ) |
Total capitalization |
| $ | 5,706,818 |
|
The table above assumes the following:
• the exchange of each Series B Unit for two shares of common stock;
• the exclusion of 419,750 shares of common stock issuable upon exercise of outstanding options, at a weighted average purchase price of $12.05 per share;
• the exclusion of the additional 180,250 shares of common stock reserved for issuance pursuant to our 2007 Stock Option Plan; and
• the exclusion of the 1,072,720 shares of common stock underlying Series A Preferred Stock underlying warrants.
21
MARKET FOR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS
Market for Common Equity
This is our initial public offering and no public market currently exists for our shares and a public market may never develop, or, if any market does develop, it may not be sustained. Our common stock is not traded on any exchange or on the over-the-counter market. After the effective date of the registration statement relating to this prospectus, we hope to have a market maker file an application with FINRA for our common stock to be eligible for trading on the OTC Bulletin Board. To be eligible for quotation, issuers must remain current in their quarterly and annual filings with the SEC. If we are not able to pay the expenses associated with our reporting obligations we will not be able to apply for quotation on the OTC Bulletin Board. We do not yet have a market maker who has agreed to file such application. There can be no assurance that our common stock will ever be quoted on a stock exchange or a quotation service or that any market for our stock will develop. We further intend to apply for the listing of our common stock on the NASDAQ Capital Market.
Holders of Equity
As of December 31, 2015, we had 29 record holders of our common stock, and a total of 836,544 shares of common stock issued and outstanding. We had 13 record holders of Series A Preferred Stock and a total of 1,800,696 shares of Series A Preferred Stock issued and outstanding. We also had 65 record holders of Series B Preferred Stock and a total of 269,987 shares of Series B Preferred Stock issued and outstanding. In addition to the 269,987 shares of Series B Preferred Stock issued, holders of Series B Preferred Stock have the right to exchange each Series B Unit for two shares of common stock. As of the date of this prospectus, no Series B Unit holder has exchanged their Series B Units into shares of common stock. Under the terms of the Series A Designations and the Series B Designations, all outstanding shares of Series A Preferred Stock and all of the outstanding shares of Series B Preferred Stock automatically convert into shares of common stock at a ratio of 1.875 to 1 upon the listing of the common stock on a Qualified National Exchange, which is defined in the Certificate of Designations of the Series B Preferred Stock as a securities exchange registered with the U.S. Securities and Exchange Commission under Section 6(a) of the Exchange Act as well as the over the counter markets administered by the OTCQX® and the OTCQB®, and their successor-entities (see also “Description of Securities — Preferred Stock”). Notwithstanding the foregoing, the Series B Preferred Stock shall not be subject to such mandatory conversion until all outstanding Convertible Securities (any evidences of indebtedness, shares or other securities convertible into or exchangeable for common stock or preferred stock) are also converted into Common Stock. The OTCBB is not considered a Qualified National Exchange and therefore listing on the OTCBB is not sufficient to trigger the Company’s ability to convert the Series B Preferred Stock.
Further, each share of Series A Preferred Stock shall be convertible, at the option of the holder thereof, at any time after the date of issuance of such share and prior to the listing of the common stock on a Qualified National Exchange, into that number of fully paid, non-assessable shares of common stock determined by dividing the Series A Original Issue Price by the Conversion Price, as such terms are defined in the Series A Designations. The conversion price of the Series A Preferred Stock is subject to adjustments pursuant to the occurrence of certain specified events.
In addition, the Company issued secured convertible notes that converted into 1,456,126 shares of Series A Preferred Stock on December 31, 2015. Upon listing of our stock on a Qualified National Exchange (as defined in the Series A Designations, see also “Description of Securities — Preferred Stock”) all of these shares will convert into shares of common stock. As of December 31, 2015, the Company had also granted options to purchase up to 419,750 shares of common stock and had issued warrants to purchase up to 256,522 shares of common stock (comprised of warrants to purchase 11,000 shares that were issued in 2009 and 2012 to Michael Knox in connection with loans to the Company, and warrants to purchase 224,897 shares that were issued in April and June of 2015 and 20,625 December Warrants issued in December 2015), and 1,800,696 shares of Series A Preferred Stock (which will be converted into 3,376,299 shares of common stock upon the listing of our stock on a Qualified National Exchange). Therefore, we have already issued or committed to issue up to 6,276,912 shares of common stock (assuming conversion of the Series A Preferred Stock and the exchange of the Series B Preferred Stock) and we are authorized to issue up to 18,723,088 additional shares of common stock. The future issuance of common stock may result in substantial dilution in the percentage of our common stock held by our then existing stockholders.
We are registering 6,784,442 shares of common stock hereunder, of which up to 2,000,000 shares of common stock are being sold by us, and we have agreed to register 4,784,442 shares of common stock currently held and issuable upon conversion of Series A Preferred Stock or exchange of Series B Units, as applicable, by our selling securityholders, of which (i) up to 836,544 shares were previously issued and sold to the selling securityholders in private placements during the calendar years 2007 through 2014, (ii) up to 31,625 shares issuable upon exercise of outstanding warrants to purchase shares of common stock that were issued to selling securityholders, (iii) up to 3,376,299 shares issuable upon the conversion of shares of our Series A Preferred Stock that were previously issued to selling securityholders, and (iv) up to 539,974 shares issuable upon the exchange of shares of our Series B Units that were previously issued to selling securityholders. See also “Selling Securityholders”.
22
Securities Authorized for Issuance under Equity Compensation Plans
As of December 31, 2014 and December 31, 2015, the Company had granted options to purchase up to 464,750 and 419,750 shares of common stock, respectively.
In 2007, the Company implemented a Stock Option Plan and initially reserved 75,000 shares of common stock to cover stock options that might be issued under the Stock Option Plan. As of December 31, 2014 and November 23, 2015, the number of shares reserved for issuance under the plan was 600,000 and 600,000 shares, respectively.
As of December 31, 2014, stock options to purchase a total of 464,750 shares at prices ranging from $8.00 to $13.20 per share had been granted to Company employees and Directors, with expiration dates ranging from April 2015 to November 2022, with a weighted average purchase price of $12.04 per share.
As of December 31, 2015, stock options to purchase a total of 419,750 shares at prices ranging from $8.00 to $13.20 per share had been granted to Company employees and Directors, with expiration dates ranging from December 2017 to March 2022, with a weighted average purchase price of $12.05 per share.
Equity Compensation Plans as of December 31, 2014
Equity Compensation Plan Information | |||||||
Plan category |
| Number of securities to be issued upon exercise of outstanding options, warrants and rights |
| Weighted-average exercise price of outstanding options, warrants and rights |
| Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) | |
Equity compensation plans approved by security holders(1) |
| 464,750 |
| $ | 12.04 |
| 135,250 |
Equity compensation plans not approved by security holders |
| — |
|
| — |
| — |
| 464,750 |
| $ | 12.04 |
| 135,250 | |
____________
(1) Reflects our 2007 Stock Option Plan for the benefit of our directors, officers, employees and consultants. As of December 31, 2014, we had reserved 600,000 shares of common stock for such persons pursuant to that plan.
Securities that May be Sold under Rule 144
Of the 836,544 shares of common stock issued and outstanding as of December 31, 2015, and the 1,800,696 outstanding shares of Series A Preferred Stock and the 269,987 shares of Series B Preferred Stock that may be exchanged for shares of common stock, and assuming no notes, warrants or stock options are converted or exercised, 762,770 shares are held by non-affiliates and 2,144,547 are held by affiliates of the Company. This means that 2,144,547 securities are deemed “restricted securities” within the meaning of Rule 144 as promulgated under the Securities Act.
It is anticipated that all of the “restricted securities” will be eligible for resale under Rule 144. In general, under Rule 144, subject to the satisfaction of certain other conditions, a person, who is not an affiliate (and who has not been an affiliate for a period of at least three months immediately preceding the sale) and who has beneficially owned restricted shares of our common stock for at least six months is permitted to sell such shares without restriction, provided that there is sufficient public information about us as contemplated by Rule 144. An affiliate who has beneficially owned restricted shares of our common stock for a period of at least one year may sell a number of shares equal to one percent of our issued and outstanding common stock approximately every three months. No underwriters were utilized and no commissions or fees were paid with respect to any of the above transactions. These persons were the only offerees in connection with these transactions. We relied on Regulation S, Section 4(2), 4(a)/2 and/or Rule 506 of Regulation D of the Securities Act for the transactions set forth above since none of the transactions involved any public offering.
23
Since inception, we have not paid any dividends on our common stock. We currently do not anticipate paying any cash dividends in the foreseeable future on our common stock, because we intend to retain our earnings, if any, to finance the growth of our business. Our Board of Directors will have the discretion to declare and pay dividends in the future. Payment of dividends in the future will depend upon our earnings, capital requirements, and other factors, which our Board of Directors may deem relevant.
24
DETERMINATION OF OFFERING PRICE
The offering price of the primary offering shares and other terms and conditions relative to the Company’s shares have been arbitrarily determined by us and do not bear any relationship to assets, earnings, book value or any other objective financial criteria. In determining the number of shares to be offered and the offering price, we took into consideration our cash on hand and the amount of money we would need to implement our business plan. Accordingly, the offering price should not be considered an indication of the actual value of the securities. The offering price of the secondary offering shares is a fixed price of $8.00 until our common stock is quoted on the OTCBB at which time the common stock may be sold at prevailing market prices or at privately negotiated prices.
25
If you invest in our shares of common stock, you will incur immediate, substantial dilution based on the difference between the public offering price per share you will pay in the primary offering and the net tangible book value per share of common stock immediately after the primary offering.
Our net tangible book value as of September 30, 2015 was approximately $(12,275,664), or $(14.67) per share, based on 836,544 shares of common stock outstanding on September 30, 2015. Based on the per share offering price of $8.00, investors will incur further dilution from the sale by us of shares of our common stock offered in the primary offering, and after deducting the estimated offering expenses of $740,000, our as adjusted net tangible book value as of September 30, 2015 would have been $2,984,336, or $1.05 per share, based on shares outstanding after the primary offering. This represents an immediate increase in net tangible book value of $15.73 per share to our existing stockholders and an immediate dilution of $6.95 per share to the new investors purchasing shares of our common stock in the primary offering.
The following table illustrates this per share dilution:
Assumed public offering price per share |
| $ | 8.00 |
|
Net tangible book value per share as of September 30, 2015 |
|
| (14.67 | ) |
Increase per share attributable to new public investors |
|
| 15.95 |
|
Net tangible book value per share after this offering |
|
| 1.05 |
|
Dilution per share to new public investors |
| $ | 6.95 |
|
26
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION
AND RESULTS OF OPERATIONS
This section of the prospectus includes a number of forward-looking statements that reflect our current views with respect to future events and financial performance. Forward-looking statements are often identified by words like “believe”, “expect”, “estimate”, “anticipate”, “intend”, “project” and similar expressions, or words which, by their nature, refer to future events. You should not place undue certainty on these forward-looking statements. These forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from our predictions.
Overview of our Business
We manufacture and sell advanced materials we call xGnP® graphene nanoplatelets and value-added products based on these materials. We have also licensed some of our base manufacturing technology to other companies and we consider technology licensing a component of our business model.
We sell our products to customers around the world and have sold materials to over 850 customers in 47 countries since 2008. We offer four main types of products, each of which may be customized for a particular customer:
1. Bulk materials, which we sell under the trademarked brand name of xGnP® graphene nanoplatelets. These materials are produced in various grades, which are analogous to average particle thickness, average particle diameters and with varying levels of oxygen-containing surface functionality. Overall, there are three standard grades, each of which is available in three different standard particle sizes and multiple customer sizes. Other grades may be available, depending upon customer performance requirements. These bulk materials are especially applicable for use as additives in polymeric or metallic composites, or in coatings or other formulations where particular electrical, thermal or barrier applications are desired by our customers.
2. Energy storage materials. These consist of specialty advanced materials that have been formulated for specific applications in the energy storage segment. Chief among these is our silicon-graphene composite material (SiG) that we offer to manufacturers of li-ion batteries. Additionally, we offer various bulk materials for use as conductive additives for cathodes and anodes in li-ion batteries, as an additive to anode slurries for lead-carbon batteries and are investigating the use of our materials as part of other battery components.
3. XG Leaf®. XG Leaf® is a family of sheet products that we manufacture for industrial, electronics, and automotive markets. These sheets are made using special formulations of xGnP® graphene nanoplatelets as precursors, along with other materials for specific applications. There are several different types of XG Leaf® available in various thicknesses, depending on the end-use requirements for thermal conductivity, electrical conductivity, or resistive heating.
4. Inks and Coatings. These consist of specially-formulated dispersions of xGnP® together with solvents, binders, and other additives to make electrically or thermally conductive products designed for printing or coating and which are suitable for a variety of industrial applications.
The process of “designing-in” new materials is a relatively complex process that involves the use of relatively small amounts of the new material in laboratory and engineering development for an extended period of time. Following successful development, we expect customers that incorporate our materials into their products will then order much larger quantities of material to support commercial production. Thus, while many of our customers are currently purchasing our materials in kilogram (one or two pounds) quantities, we expect many of our commercial customers will require tons or even hundreds of tons of material. The majority of our customers are still in the development stage and our product sales thus consist of relatively many orders for relatively small quantities of materials being used in a variety of research or development activities and early phase adoption in limited commercial applications.
Operating Segment
We have one reportable operating segment that manufactures xGnP® graphene nanoplatelets and products produced therefrom, that conducts research on graphene nanoplatelets and related products and licenses our technology as appropriate. As of September 30, 2015 we shipped products on a worldwide basis, but all of our assets were located within the United States.
27
Our Critical Accounting Policies
The preparation of financial statements in conformity with United States generally accepted accounting principles requires our management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates. Our management routinely makes judgments and estimates about the effects of matters that are inherently uncertain. For a complete description of our significant accounting policies, see Note 2 to our Consolidated Financial Statements included in this prospectus.
Our critical accounting policies are those where we have made difficult, subjective or complex judgments in making estimates, and/or where these estimates can significantly impact our financial results under different assumptions and conditions. Our critical accounting policies are:
• Revenue Recognition
• Stock Based Compensation
• Fair Value Measurements
• Beneficial Conversion Features of Convertible Notes
• Going Concern
Revenue Recognition
We recognize revenues when (a) the price is fixed or determinable, (b) persuasive evidence of a sales arrangement exists, (c) the service is performed or delivery has occurred and (d) collectability of the resulting receivable is reasonably assured.
We recognize product revenues when products are shipped to customers. At that time, product ownership and risk have transferred to the customer and we have no further obligations. We record product sales at net selling prices that are reflective of discounts and allowances. Shipping and handling costs are recorded as a component of direct costs, as are shipping and handling costs billed to customers.
Revenue related to licensing agreements is recorded upon substantial performance of the terms of the licensing contract. In the case of licensing arrangements that involve up-front payments, revenue is recorded when management determines that the appropriate terms of the contract have been fulfilled. For example, this may occur when technology has been transferred via written documents or, if training is involved, whenever all contracted training has occurred. In the case of licenses where product delivery is also embedded in the deliverable, a portion of revenue would be recognized when products are delivered.
In the case of licensing arrangements that involve ongoing royalties based on sales of products produced with our technology, royalty income is recorded when received or, in the case of minimum royalties due, in the period when due.
Grant contract revenue is recognized over the life of the contracts as the services are performed.
Amounts received in excess of revenues earned are recorded as deferred revenue.
Stock-Based Compensation
We recognize compensation expense in our statement of operations for all share-based option and stock awards, based on estimated grant-date fair values.
We estimate the grant-date fair value of stock-based compensation awards using the Black-Scholes option valuation model. This model is affected by the estimated value of our common stock on the date of the grant as well as assumptions regarding a number of highly complex and subjective variables. These variables include the expected term of the option, the exercise price, expected risk-free rates of return, the expected volatility of our common stock, and expected dividend yield, each of which is more fully described below. The assumptions for the estimated value of our common stock, expected term and expected volatility are the assumptions that most significantly affect the grant date fair value.
28
Estimated Value of our Common Stock: The estimated value of our common stock was determined on the basis of recent third-party investment transactions that established a price of $12.00 per share as the value at which outsiders were willing to invest in our stock, as of the January 2014 grant date.
Expected Term: Because we have limited experience related to the exercise of employee stock options, we use the simplified method permitted by SEC Staff Accounting Bulletin Topic 14 to estimate the expected term of the stock options. The expected term of an option is estimated to be equal to the mid-point between the vesting and expiration dates of the option.
Risk-free Interest Rate: We base the risk-free interest rate used on the implied yield at the grant date of U.S. Treasury zero-coupon issues with a term approximately equal to the expected term of the stock-based award being valued.
Expected Stock Price Volatility: Because we are a private company with very limited stock sales history. we use a blended average weekly volatility of certain publicly traded peer companies. We believe that the use of this blended average peer volatility is reflective of market conditions and a reasonable indicator of our expected future volatility.
Dividend Yield: Because we have never paid a dividend and do not expect to begin doing so in the foreseeable future, we have assumed a 0% dividend yield in valuing our stock-based awards.
The grant-date fair value of the award is recognized as expense over the requisite service period using the straight-line method.
Fair Value Measurements
FASB ASC 820: “Fair Value Measurements and Disclosures” defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. ASC 820 also establishes a fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure fair value:
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Inputs other than quoted prices included within Level 1 which are either directly or indirectly observable.
Level 3: Inputs to the valuation methodology are unobservable and significant to the fair value measurement.
A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement. Our derivative liabilities are classified as Level 3 within the fair value hierarchy because they were valued using other unobservable inputs. The valuation technique used to measure fair value of the derivative liabilities is based on a lattice model with significant assumptions and inputs determined by the Company. A lattice model was used to estimate the fair value of the derivative liabilities because management believes it reflects all the assumptions that market participants would likely consider including early exercise of the warrants. The fair value of the derivative liabilities will be significantly influenced by the fair value of our common stock, stock price volatility and the risk free interest components of the lattice technique.
Beneficial Conversion Feature of Convertible Notes
If the embedded conversion option in a convertible note is not required to be separated and accounted for as a derivative instrument liability, then ASC 470-20: “Debt with Conversion and Other Options” requires separate recognition of any beneficial conversion feature (BCF) related to the convertible instrument. Accordingly, we recognized a BCF related to the Notes that we issued in 2013. The BCF for the convertible instruments is recognized by allocating to additional paid-in capital a portion of the proceeds received equal to the intrinsic value of the conversion feature. Generally, the intrinsic value is calculated at the commitment date as the difference between the effective conversion price and the fair value of the common stock or other securities into which the security is convertible, multiplied by the number of shares into which the security is convertible. When certain other securities, such as warrants, are issued in conjunction with the convertible security, the proceeds are allocated among the different components. The portion of the proceeds allocated to the convertible security is divided by the contractual number of the conversion shares to determine the effective conversion price which is then used to measure the BCF. The value of the BCF recognized is limited to the amount of proceeds received that is initially allocated to the convertible security.
29
Going Concern
We have historically incurred recurring losses from operations and we may continue to generate negative cash flows as we implement our business plan. Our unaudited condensed consolidated financial statements are prepared using US GAAP as applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business.
We currently do not have sufficient cash or commitments for financing to sustain our operations for the next twelve months. We currently anticipate that our cash and cash equivalents will be sufficient to fund our operations through April 2016, without raising additional capital. Our plan is to develop customer relationships and increase our revenues derived from our products and IP licensing. Although we have historically incurred operating losses, we have been able to fund such losses primarily by selling common and preferred stock and convertible notes. We expect that our cash on hand and proceeds from anticipated financings (private placements of debt and equity securities) will sustain our operations for the next twelve months. However, there can be no assurance of our continued success in raising capital to fund our business plan.
In the event we are unable to fund our operations from existing cash on hand, operating cash flows, additional borrowings or raising equity capital, we may be forced to reduce our expenses, slow down our growth rate, or discontinue operations. Our consolidated financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary should we be unable to continue as a going concern.
On April 18, 2015 we commenced an investment offering of up to $18,000,000 in Series B Units. The offering price is $16 per unit.
The Company is authorized to issue up to 1,500,000 shares of Series B Preferred Stock. Each share of the Series B Preferred Stock, which has a liquidation preference of $16 per share, is convertible at any time, at the option of the holder, into one share of common stock. The Series B Preferred Stock may, at the option of the holder, be exchanged for any new securities issued by the Company until the earlier of such time as the Company completes the sale of at least $18 million in new financing or December 31, 2017. The Series B Preferred Stock also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. The Series B is subject to mandatory conversion into Common Stock upon the listing of the Company’s common stock on a Qualified National Exchange, provided that all outstanding Convertible Securities have also converted into common stock.
In conjunction with this investment offering, all holders of the outstanding secured convertible notes (see Note 6 of September 30, 2015 condensed consolidated financial statements) agreed to modify the original terms of the Notes such that the Notes will automatically convert into shares of Series A Preferred Stock upon the earlier to occur of 1) the Company raising not less than $12,000,000 or 2) December 31, 2015. The Notes were converted on December 31, 2015 at a conversion price of $12.00.
As of September 30, 2015 we had accepted subscriptions for 269,987 Series B Units valued at $4,319,792 and had issued 269,987 shares of Series B Preferred Stock and Warrants to purchase 224,897 shares of common stock, which includes 3,100 shares of Series B Preferred Stock and 2,635 warrants issued pursuant to the preemptive rights described in Note 6 to the September 30, 2015 condensed consolidated financial statements. The Warrants were offered with either 85%, 50% or 15% coverage, depending on the time the Company received the subscription. With the exception of 2 investors, all Warrants were subscribed for at 85% coverage. Accordingly, the number of shares of common stock underlying the Warrants included as part of the Series B Units differ from the number of units subscribed for.
On December 31, 2015, we issued the December Notes and December Warrants to purchase 20,625 shares of common stock to several existing stockholders. The December Notes mature on June 30, 2016 and the December Warrants have a five year term and a strike price of $8.00. We cannot determine how much additional funds we might raise under this private placement. Under the terms of this private placement, the conversion price of the Series A Preferred Stock was adjusted to $6.40 per share and holders of Series B Preferred Stock received the right to exchange their Series B Units for common stock at a rate of 2 shares of common stock for each Series B Unit.
If we raise gross proceeds of $16 million in the primary offering, we will fund general corporate expenses of approximately $5.2 million over the next two years, by which time the Company might begin generating positive cash flow from operations.
Recent Accounting Pronouncements
In May 2014, the Financial Accounting Standards Board, or FASB, issued Accounting Standards Update No. 2014-09,Revenue From Contracts With Customers, or ASU 2014-09. Pursuant to this update, an entity should recognize revenue to depict the transfer of promised goods or services to customers in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. The amendments in this update are currently effective for annual reporting periods beginning after December 15, 2016, including interim periods within that reporting period, and are to be applied
30
retrospectively, or on a modified retrospective basis. Early application is not permitted. In July 2015, the FASB approved a one year deferral of the effective date for annual reporting periods beginning after December 15, 2017 with early adoption permitted for annual periods beginning after December 15, 2016. We are currently evaluating the impact of adopting ASU 2014-09 on our financial statements.
In June 2014, the FASB issued ASU No. 2014-10,Development Stage Entities (Topic 915): Elimination of Certain Financial Reporting Requirements, Including an Amendment to Variable Interest Entities Guidance in Topic 810, Consolidation, or Topic 915. The guidance set forth in Topic 915 is intended to reduce the overall cost and complexity associated with financial reporting for development stage entities without reducing the availability of relevant information. The FASB also believes the changes will simplify the consolidation accounting guidance by removing the differential accounting requirements for development stage entities. As a result of these changes, there no longer will be any accounting or reporting differences in generally accepted accounting principles, or GAAP, between development stage entities and other operating entities. For organizations defined as public business entities, the presentation and disclosure requirements in Topic 915 will no longer be required starting with the first annual period beginning after December 15, 2014, including interim periods therein. Early application is permitted for any annual reporting period or interim period for which the entity’s financial statements have not yet been issued (public business entities) or made available for issuance (other entities). This standard had no impact on our financial position, results of operations or cash flows.
In August 2014, FASB issued ASU 2014-15,Disclosure of Uncertainties about an Entity’s Ability to Continue as a Going Concern, or ASU 2014-15. ASU 2014-15 explicitly requires a company’s management to assess an entity’s ability to continue as a going concern, and to provide related footnote disclosures in certain circumstances. The new standard will be effective in the first annual period ending after December 15, 2016, although early application is permitted. We are currently evaluating the potential impact of the adoption of this standard, but believe its adoption will have no impact on our financial position, results of operations or cash flows.
In November 2014, the FASB issued ASU No. 2014-16,Determining Whether the Host Contract in a Hybrid Financial Instrument Issued in the Form of a Share is more akin to Debt or to Equity, or ASU 2014-16. ASU 2014-16 clarifies how current GAAP should be interpreted in evaluating the economic characteristics and risks of a host contract in a hybrid financial instrument that is issued in the form of a share. Specifically, ASU 2014-16 provides that an entity should consider all relevant terms and features, including the embedded derivative feature being evaluated for bifurcation, in evaluating the nature of the host contract. ASU 2014-16 is effective for public companies for fiscal years and interim periods within those fiscal years beginning after December 15, 2015 with early adoption permitted. We adopted this guidance for the year ended December 31, 2014 and have properly applied it to hybrid financial instruments.
In April 2015, the FASB issued ASU No. 2015-03,Simplifying the Presentation of Debt Issuance Costs, or ASU 2015-03. ASU 2015-03 requires debt issuance costs to be presented in the balance sheet as a direct deduction from the carrying value of the associated debt liability, consistent with the presentation of a debt discount. The standard also aligns the GAAP presentation with International Financial Reporting Standards and will remedy the long-standing conflict with the guidance in FASB Concepts Statement No. 6, Elements of Financial Statements, which indicates that debt issuance costs do not meet the definition of an asset, because they provide no future economic benefit. ASU No. 2015-03 is effective for financial statements issued for fiscal years beginning after December 15, 2015, and interim periods within those fiscal years. Early adoption is permitted for financial statements that have not been previously issued. The new guidance will be applied on a retrospective basis. The adoption of this guidance during the nine months ended September 30, 2015 did not have a material impact on our balance sheets.
JOBS Act
In April 2012, the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, was enacted. Section 107 of the JOBS Act provides that an “emerging growth company,” or EGC, can take advantage of the extended transition period for complying with new or revised accounting standards. Thus, an EGC can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies.
We are in the process of evaluating the benefits of relying on other exemptions and reduced reporting requirements under the JOBS Act. Subject to certain conditions, as an EGC, we intend to rely on certain of these exemptions, including exemptions from the requirement to provide an auditor’s attestation report on our system of internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and from any requirement that may be adopted by the Public Company
31
Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion and analysis. We will remain an EGC until the earlier of: the last day of the fiscal year in which we have total annual gross revenues of $1.0 billion or more; the last day of the fiscal year following the fifth anniversary of the date of the completion of this offering; the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
Internal Control Over Financial Reporting
Assessing our staffing and training procedures to improve our internal control over financial reporting is an ongoing process. We are not currently required to comply with Section 404 of the Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act, and are therefore not required to make an assessment of the effectiveness of our internal control over financial reporting. As a result, our management did not perform an evaluation of our internal control over financial reporting as of December 31, 2014. Further, our independent registered public accounting firm has not been engaged to express, nor have they expressed, an opinion on the effectiveness of our internal control over financial reporting.
Results of Operations for the Year Ended December 31, 2014 Compared with the Year Ended December 31, 2013
The following table summarizes the results of our operations for the years ended December 31, 2014 and 2013.
Summary Income Statement |
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||||
Total Revenues |
| $ | 1,635,002 |
|
| $ | 1,814,302 |
|
| (179,300 | ) |
| (9.9 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of Goods Sold |
|
| 2,749,331 |
|
|
| 2,255,155 |
|
| 494,176 |
|
| 21.9 |
|
Gross Profit |
|
| (1,114,329 | ) |
|
| (440,853 | ) |
| (673,476 | ) |
| 152.8 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research & Development expense |
|
| 1,449,478 |
|
|
| 1,269,456 |
|
| 180,022 |
|
| 14.2 |
|
Sales, General & Administrative expense |
|
| 4,405,266 |
|
|
| 2,739,956 |
|
| 1,665,310 |
|
| 60.8 |
|
Total operating expense |
|
| 5,854,744 |
|
|
| 4,009,412 |
|
| 1,845,332 |
|
| 46.0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating Loss |
|
| (6,969,073 | ) |
|
| (4,450,265 | ) |
| (2,518,808 | ) |
| 56.6 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other Income (expense) |
|
| (4,454,985 | ) |
|
| (362,191 | ) |
| (4,092,794 | ) |
| 1,130.0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Loss |
| $ | (11,424,058 | ) |
| $ | (4,812,456 | ) |
| (6,611,602 | ) |
| 137.4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deemed dividend on Series A convertible preferred stock |
|
| (316,366 | ) |
|
| — |
|
| (316,366 | ) |
| n/a |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | (11,740,424 | ) |
| $ | (4,812,456 | ) |
| (6,927,968 | ) |
| 144.0 |
|
Revenues
We categorize revenues into three broad categories: product sales, licensing revenue, and research contracts. Revenues for the past two years, by category, are shown below.
Revenue Summary
|
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||
Product Sales |
| $ | 874,469 |
| $ | 239,767 |
| 634,702 |
|
| 264.7 |
|
Grants |
|
| 660,533 |
|
| 174,535 |
| 485,998 |
|
| 278.5 |
|
Licensing Revenues |
|
| 100,000 |
|
| 1,400,000 |
| (1,300,000 | ) |
| (92.9 | ) |
Total |
| $ | 1,635,002 |
| $ | 1,814,302 |
| (179,300 | ) |
| (9.9 | ) |
32
Product sales consist primarily of material sold to customers for research or development purposes, although a few small commercial applications are beginning to result in larger sales. Product sales increased by $634,702, or about 265% from 2013 to 2014 primarily because of one large order for $600,000 from a development customer. We do not consider this to be a significant indicator of future sales or overall company performance, because the size and number of “research” orders is not representative of the commercial potential represented by specific customers and applications.
We also classify product sales according to the type of product, and we divide our products into four main classes of materials. The table below shows a breakdown of product sales into these classes.
Product Sales Summary
|
| Years ended December 31 |
| Change 2013 – 2014 | |||||||||
|
| 2014 |
| 2013 |
| $ |
| % | |||||
xGnP Bulk Material |
| $ | 809,609 |
| $ | 192,894 |
|
| 616,715 |
|
| 319.7 |
|
Battery Anode Materials (SiG) |
|
| 37,388 |
|
| 23,350 |
|
| 14,038 |
|
| 60.1 |
|
XG Leaf |
|
| 20,504 |
|
| 12,686 |
|
| 7,818 |
|
| 61.6 |
|
Inks and Coatings |
|
| 6,968 |
|
| 10,837 |
|
| (3,869 | ) |
| (35.7 | ) |
Total |
| $ | 874,469 |
| $ | 239,767 |
| $ | 634,699 |
|
| 264.7 |
|
The table below shows a comparison of orders received and the average order size, both in dollars and in physical size. These numbers indicate that our customer base remains active with development or research projects that use our materials, and the total number of customer orders increased 4% from 2013 to 2014. However, the small average order size indicates that almost all orders are for quantities that are far smaller than anticipated orders once the Company’s products are commercialized in an industrial or consumer product.
Order Summary
|
| Years ended December 31 |
| Change 2013 – 2014 | |||||||
|
| 2014 |
| 2013 |
| Amt |
| % | |||
Number of orders – total |
|
| 476 |
|
| 456 |
|
| 20 |
| 4.4 |
Average order size – |
| $ | $2,175 |
| $ | 768 |
| $ | 1,407 |
| 183.2 |
Average order size – kg. |
|
| 8.3 |
|
| 5.0 |
|
| 3.3 |
| 66.0 |
Grant revenues of $660,533 in 2014 were up by 278% from the previous year, primarily because of a Phase II Small Business Innovation Research (“SBIR”) grant from the U.S. Department of Energy (“DOE”) as well as a one-time award from the Michigan Economic Development Corporation (“MEDC”). The following table shows the individual grants that made up grant revenues during 2014 and 2013.
Grant Revenues
|
| Years ended December 31 |
| Change 2013 – 2014 | ||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||
US DOE |
| $ | 463,093 |
| $ | 147,785 |
| 315,308 |
| 213.4 |
MEDC Advanced Manufacturing |
|
| 42,440 |
|
| 6,750 |
| 35,690 |
| 528.7 |
MEDC Advance Materials |
|
| 30,000 |
|
| 20,000 |
| 10,000 |
| 50.0 |
MEDC Emerging Technologies |
|
| 125,000 |
|
| — |
| 125,000 |
| na |
Total |
| $ | 660,533 |
| $ | 174,535 |
| 485,998 |
| 278.5 |
The US DOE grant revenues recognized during 2014 consisted of the first year of a two-year grant received in December 2013, which represented a continuation of a previously awarded Phase I grant entitled “Low-cost, High-Energy Si/Graphene Anodes for Li-Ion Batteries.” This award was for a total of $1,147,684 to fund a research project over a planned two-year period commencing in January 2014. The first-year milestone review has been completed for this project and award funding for the second half of the program, to be accomplished during 2015, has been approved.
33
The individual MEDC awards were awards granted as a result of competitive award processes for various research or development projects intended to advance the state of manufacturing or materials development in Michigan. All of the work under each of the MEDC awards has been completed and we do not expect further revenue from any of these grants.
Licensing revenues declined significantly during 2014, as compared to 2013, because of the completion of required work under a license agreement executed in late 2011. Under the terms of the license agreement, the licensee agreed to pay us an up-front payment of $2.5 million. In addition, the licensee also committed to pay $750,000 per year for each of two years for either: (1) access to XGS facilities for purposes of product development; or (2) purchase of XGS products. Near the end of the two-year period, the licensee and XGS completed this agreement with a series of product orders, shipped during the first half of 2014. Thus, the final accounting for the license agreement amortized the net revenues over the two-year period of 2012 and 2013 as follows: $2.5 million in up-front payments, amortized equally over 2012 and 2013 at $1.25 million per year; $750,000 in plant access and product purchase charges paid and fulfilled in 2012 was recorded as 2012 licensing revenue; $750,000 in plant access and product purchase charges paid in 2013 was recorded as $150,000 in 2013 licensing revenue and $600,000 in deferred revenues and these revenues recognized as product revenues in 2014. The $100,000 in licensing revenues booked during 2014 is the minimum payment required under a different license agreement with a different licensee.
Cost of Goods Sold
We use a standard cost system to estimate the direct costs of products sold. Direct costs include estimates of raw material costs, packaging, freight charges net of those billed to customers, and an allocation for direct labor and manufacturing overhead. Because of the nature of our production processes, there is a substantial fixed manufacturing expense requirement that represents the ongoing cost of maintaining production facilities that are not directly related to products sold, so we use a “full capacity” allocation of overhead based on an estimate of what product costs would be if the manufacturing facilities were operating on a full-time basis and producing products at the designed capacity. This estimate involves estimating both the level of expenses as well as production amounts as if the manufacturing facility were operating on a continuous, three-shift, production basis.
The following table shows the relationship of direct costs and unallocated manufacturing expenses to product sales for the two most recent calendar years:
Gross Profit Summary
|
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||||
Product Sales |
| $ | 874,469 |
|
| $ | 239,767 |
|
| 634,702 |
|
| 264.7 |
|
Direct Costs |
|
| 226,678 |
|
|
| 129,243 |
|
| 97,435 |
|
| 75.4 |
|
Direct Cost Margin |
| $ | 647,791 |
|
| $ | 110,524 |
|
| 537,267 |
|
| 486.1 |
|
% of sales |
|
| 74.1 | % |
|
| 46.1 | % |
|
|
|
|
|
|
Unallocated Manufacturing Expense |
|
| 2,522,653 |
|
|
| 2,125,912 |
|
| 396,741 |
|
| 18.7 |
|
Gross profit (Loss) on Product Sales |
| $ | (1,874,862 | ) |
| $ | (2,015,388 | ) |
| 140,526 |
|
| (7.0 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Grants and Licensing Revenue |
|
| 760,533 |
|
|
| 1,574,535 |
|
| (814,002 | ) |
| (51.7 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross Profit (Loss) |
| $ | (1,114,329 | ) |
| $ | (440,853 | ) |
| (673,476 | ) |
| 152.8 |
|
We believe that the fluctuations in direct cost from period to period are not material. Direct costs vary depending on the size of an order, the specific products being ordered, and other factors like shipping destination. We normally expect direct cost margins to fluctuate with a range of plus or minus ten percentage points of sales, depending on product mix. Because of one large order during the first half of 2014, the Direct Cost Margin, as a percentage of sales, during 2014 was higher than in 2013.
Costs associated with grant revenues tend to be a mixture of facilities use, management time, labor from scientists, technicians and manufacturing personnel, and some supplies. Because of the difficulty of developing and maintaining an administrative system to gather direct costs for grants, together with the relatively small size of grant revenues, we do not track direct costs for
34
grant revenues as a separate cost category. Therefore, we do not calculate direct cost margins associated with grant revenues but, rather, we view these revenues as being supported by indirect corporate expenses.
Costs associated with licensing revenue tend to be a mixture of IP costs as well as management and administrative expenses that are indirect in nature. As such, we do not assign direct costs to licensing revenues. Where revenues from a license agreement can be assigned to specific product revenues, we classify these revenues as product sales and, using our standard cost system, assign direct costs to those sales.
The remaining “non-direct” costs of operating our manufacturing facilities are recorded as unallocated manufacturing expenses. This expense includes personnel costs, rent, utilities, indirect supplies, depreciation, and related indirect expenses. Unallocated manufacturing expenses are expensed as incurred.
Total unallocated manufacturing expenses increased by $396,741, or 19%, from 2013 to 2014. The primary cause of this increase, accounting for about half of the overall dollar increase, was increased payroll, tax and benefit costs caused by additions of 10 new personnel associated with new production processes being implemented. In particular, an XG Leaf® pilot production line was initiated during the late third quarter of the 2013 and incurred expenses throughout 2014. This line incurred expenses for installing and testing equipment as well as an increase in headcount of 8 people. Other expense increases were recorded materials used, supplies, utilities and depreciation. These expense increases reflect the increase in production over the previous year as well as the continued addition of new equipment to the manufacturing facilities.
The following table shows the major expense categories that make up unallocated manufacturing expense as well as the changes from 2013 to 2014:
Unallocated Manufacturing Expense
|
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||||
Payroll, tax & benefits |
| $ | 881,948 |
|
| $ | 659,496 |
|
| 222,452 |
|
| 33.7 |
|
Materials used |
|
| 189,899 |
|
|
| 93,779 |
|
| 96,120 |
|
| 102.5 |
|
Supplies |
|
| 218,724 |
|
|
| 136,217 |
|
| 82,507 |
|
| 60.6 |
|
Equipment repairs & maintenance |
|
| 188,223 |
|
|
| 205,697 |
|
| (17,474 | ) |
| (8.5 | ) |
Building lease |
|
| 192,223 |
|
|
| 201,345 |
|
| (9,122 | ) |
| (4.5 | ) |
Utilities |
|
| 191,231 |
|
|
| 114,988 |
|
| 76,243 |
|
| 66.3 |
|
Depreciation |
|
| 681,983 |
|
|
| 544,999 |
|
| 136,984 |
|
| 25.1 |
|
Tools & equipment |
|
| 121,675 |
|
|
| 187,607 |
|
| (65,932 | ) |
| (35.1 | ) |
All other |
|
| 16,416 |
|
|
| 34,666 |
|
| (18,250 | ) |
| (52.6 | ) |
Allocated to direct costs |
|
| (159,669 | ) |
|
| (52,882 | ) |
| (106,787 | ) |
| 201.9 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | 2,522,653 |
|
| $ | 2,125,912 |
|
| 396,741 |
|
| 18.7 |
|
Research and Development Expenses
Research and development costs increased from about 14% from 2013 to 2014 primarily as a result of one additional staff member and increased contracted services to support our increased research under grant awards. The following table shows the main individual components of research and development expenses.
Increased depreciation, which was up by 20% over the previous year, resulted from the addition of equipment, primarily instruments and test apparatuses, to our research facilities.
35
Research and Development Expenses
|
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||
Payroll, tax & benefits |
| $ | 876,183 |
| $ | 807,068 |
| 69,115 |
|
| 8.6 |
|
Building lease |
|
| 108,080 |
|
| 105,768 |
| 2,312 |
|
| 2.2 |
|
Depreciation |
|
| 142,785 |
|
| 119,429 |
| 23,356 |
|
| 19.6 |
|
Contracted services |
|
| 203,271 |
|
| 114,947 |
| 88,324 |
|
| 76.8 |
|
All other |
|
| 119,159 |
|
| 122,244 |
| (3,085 | ) |
| (2.5 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | 1,449,478 |
| $ | 1,269,456 |
| 180,022 |
|
| 14.2 |
|
Sales, General and Administrative Expenses
Overall expenses in this category increased by 61% from 2013 to 2014, primarily as a result of increases in personnel, stock option compensation and professional fees. The following table shows the major expense categories that make up sales, general and administrative expenses as well as the changes from 2013 to 2014:
Sales, General and Administrative Expenses
|
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||
Payroll, tax & benefits |
| $ | 2,343,456 |
| $ | 1,423,930 |
| 919,526 |
|
| 64.6 |
|
Non-cash stock option compensation |
|
| 644,787 |
|
| 233,002 |
| 411,785 |
|
| 176.7 |
|
Building lease |
|
| 51,104 |
|
| 41,346 |
| 9,758 |
|
| 23.6 |
|
Utilities |
|
| 32,400 |
|
| 84,660 |
| (52,260 | ) |
| (61.7 | ) |
Professional fees |
|
| 856,385 |
|
| 606,663 |
| 249,722 |
|
| 41.2 |
|
Travel & entertainment |
|
| 121,885 |
|
| 77,989 |
| 43,896 |
|
| 56.3 |
|
Depreciation & amortization |
|
| 65,790 |
|
| 66,786 |
| (996 | ) |
| (1.5 | ) |
Contract engineering |
|
| 101,059 |
|
| 25,010 |
| 76,049 |
|
| 304.1 |
|
Insurance |
|
| 46,209 |
|
| 14,345 |
| 31,864 |
|
| 222.1 |
|
All other |
|
| 142,191 |
|
| 166,225 |
| (24,034 | ) |
| (14.5 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | 4,405,266 |
| $ | 2,739,956 |
| 1,665,310 |
|
| 60.8 |
|
On January 16, 2014 our current CEO, Dr. Philip Rose was hired and our former CEO, Mr. Michael Knox became a Senior Vice President. As part of his compensation package, Dr. Rose was awarded 220,000 stock options that vest over time and under different circumstances. This option grant accounts for the majority of the non-cash stock option compensation recorded during 2014. Other than the addition of the new CEO, there were no other net headcount additions in this expense category during the year. The majority of the year-over-year increase from 2013 to 2014 was due to the fact that 6 individuals were hired in this category during 2013 but only part of their compensation was recorded during 2013.
Professional fees increased by about $250,000 during 2014, primarily because of expenses associated with legal, audit, professional search fees, and investment banking expenses associated with fundraising. The professional search fees related to the search for a CEO, which was closed in January 2014 as well as a search for a Chief Financial Officer. The increased audit fees related to a change in our auditors made for the 2013 audit and beyond because of our desire to work with a PCAOB registered firm. Many of the legal fees, as well as investment banking fees, related to the $9.6 million in new investment raised during the year as well as additional fundraising efforts underway at the end of the year. Travel expenses increased by $43,896 during the year because of additional travel, by a broader group of managers, to Southeast Asia. This reflects our recent investment from, and joint development work with, Samsung as well as the fact that most of our customers are in Southeast Asia. Finally, contract engineering expense increased by $76,049 during the year primarily because of engineering projects associated with scale-up of manufacturing processes and development of new manufacturing processes.
36
Other Income (Expense)
The following table shows a comparison of other income and expense by major expense component for the years ended December 31, 2013 and 2014:
Other Income (Expense) |
| Years ended December 31 |
| Change 2013 – 2014 | |||||||||
|
| 2014 |
| 2013 |
| $ |
| % | |||||
Incentive refund & interest income |
| $ | 117,940 |
|
| $ | 72,467 |
|
| 45,473 |
|
| 62.7 |
Interest expense |
|
| (1,507,374 | ) |
|
| (471,102 | ) |
| (1,036,272 | ) |
| 220.0 |
Gain on fair value – derivative liability – warrants |
|
| 503,186 |
|
|
| 81,339 |
|
| 421,847 |
|
| 518.6 |
Extinguishment loss on convertible notes |
|
| (3,513,770 | ) |
|
| — |
|
| (3,513,770 | ) |
| n/a |
Loss on disposal of equipment |
|
| (54,967 | ) |
|
| (44,895 | ) |
| (10,072 | ) |
| 22.4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | (4,454,985 | ) |
| $ | (362,191 | ) |
| (4,092,794 | ) |
| 1,130.0 |
Our 2013 and 2014 financing agreements primarily involved the sale of secured convertible notes. These notes carry an annual interest rate of 12%, which accrues and is compounded monthly. The large increase of $1 million in interest expense from 2013 to 2014 reflects the issuance of these notes beginning in January 2013 and continuing through June of 2014. At December 31, 2013 and 2014 there were $3.4 million and $10.4 million in convertible notes outstanding, respectively.
Stock warrants were issued throughout 2013 and in 2014 in association with the sale of secured convertible notes and preferred stock. These warrants are accounted for as derivative liabilities and are revalued to fair market value on a quarterly basis. Changes in value are recorded as non-cash income or expense.
The incentive refund and interest income category primarily reflects ongoing receipts from the State of Michigan as part of a jobs incentive package awarded to us in 2011. This is a five-year program under which the State of Michigan partially funds new jobs created for eligible companies.
Liquidity and Capital Resources
The following table shows our cash flow statement for the past two years with sub-totals for these major categories:
Summary Cash Flow |
| Years ended December 31 |
| Change 2013 – 2014 | ||||||||||
|
| 2014 |
| 2013 |
| $ |
| % | ||||||
Cash, beginning of period |
| $ | 95,342 |
|
| $ | 79,470 |
|
| 15,872 |
|
| 20.0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating activities |
|
| (6,490,914 | ) |
|
| (3,490,294 | ) |
| (3,000,620 | ) |
| 86.0 |
|
Investing activities (purchase of assets) |
|
| (1,083,196 | ) |
|
| (1,087,575 | ) |
| 4,379 |
|
| (0.4 | ) |
Financing activities |
|
| 9,567,634 |
|
|
| 4,593,741 |
|
| 4,973,893 |
|
| 108.3 |
|
Net increase in cash |
|
| 1,993,524 |
|
|
| 15,872 |
|
| 1,977,652 |
|
| 12,460.0 |
|
Cash, end of period |
| $ | 2,088,866 |
|
| $ | 95,342 |
|
| 1,993,524 |
|
| 2,090.9 |
|
Our cash position increased by about $2 million from 2013 to 2014 primarily as a result of financing activities that raised about $9 million in new funds during 2014. The remaining portion of the funds raised during the year were used to fund increases in working capital ($1.3 million), purchase of additional capital assets ($1.1 million), and to fund our net losses caused by continuing business, manufacturing and technology development expenses that exceeded our revenues.
Offsetting some of these items, a large amount of non-cash expense items on our income statement are associated with the securities and related warrants we issued over the past two years. Accrued interest expense of $1.5 million was a non-cash expense during 2014. In January of 2014, in association with a debt offering to Samsung Ventures, we consolidated previously-issued debt and issued additional warrants to our previous debt-holders. These actions resulted in a large, one-time, non-cash charge of $3.5 million recorded as an extinguishment loss on convertible notes.
During 2014, we invested $924,947 in purchases of property and equipment, down somewhat from the corresponding amount of $1,051,920 in 2013. We also invested $158,249 in purchases of intangible assets during 2014, up from $35,655 in 2013. These expenses are primarily legal fees related to the ongoing management of our patent portfolio as well as new patent filings.
37
Results of Operations for the Nine Months Ended September 30, 2015 Compared with the Nine Months Ended September 30, 2014
The following table summarizes the results of our operations for the nine month periods ended September 30, 2015 and 2014.
|
| Nine Months ended September 30 |
| Change 2014 – 2015 | ||||||||||
Summary Income Statement |
| 2015 |
| 2014 |
| $ |
| % | ||||||
Total Revenues |
| $ | 531,487 |
|
| $ | 1,425,508 |
|
| (894,021 | ) |
| (62.7 | ) |
Cost of Goods Sold |
|
| 1,341,948 |
|
|
| 2,045,036 |
|
| (703,088 | ) |
| (34.4 | ) |
Gross Profit |
|
| (810,461 | ) |
|
| (619,528 | ) |
| (190,933 | ) |
| (30.8 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Research & Development Expense |
|
| 1,135,326 |
|
|
| 988,149 |
|
| 147,177 |
|
| 14.9 |
|
Sales, General & Administrative Expense |
|
| 3,133,375 |
|
|
| 3,420,125 |
|
| (286,750 | ) |
| (8.4 | ) |
Loss on disposal of equipment and intangible |
|
| — |
|
|
| 38,177 |
|
| (38,177 | ) |
| (100.0 | ) |
Total Operating Expense |
|
| 4,268,701 |
|
|
| 4,446,451 |
|
| (177,750 | ) |
| (4.0 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating Loss |
|
| (5,079,162 | ) |
|
| (5,065,979 | ) |
| (13,183 | ) |
| (0.3 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other Income (Expense) |
|
| (1,703,889 | ) |
|
| (4,061,282 | ) |
| 2,357,393 |
|
| 58.0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net Loss |
| $ | (6,783,051 | ) |
| $ | (9,127,261 | ) |
| 2,344,210 |
|
| 25.7 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deemed dividend on Series A Convertible preferred stock |
|
| — |
|
|
| (316,366 | ) |
| 316,366 |
|
| 100.0 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss attributable to common stockholders |
| $ | (6,783,051 | ) |
| $ | (9,443,627 | ) |
| 2,660,576 |
|
| 28.2 |
|
Revenues
Revenues for the nine month periods ended September 30, 2015 and 2014, by category, are shown below.
Revenue Summary
|
| Nine Months ended |
| Change 2014 – 2015 | ||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||
Product Sales |
| $ | 156,608 |
| $ | 798,881 |
| (642,273 | ) |
| (80.4 | ) |
Grants |
|
| 299,879 |
|
| 551,627 |
| (251,748 | ) |
| (45.6 | ) |
Licensing Revenues |
|
| 75,000 |
|
| 75,000 |
| — |
|
| 0.0 |
|
Total |
| $ | 531,487 |
| $ | 1,425,508 |
| (894,021 | ) |
| (62.7 | ) |
Product sales consist of two broad categories: 1) material sold to customers for research or development purposes; and 2) production orders for customers. Typically, the order sizes for the first category are relatively small, however we expect orders in the second category to be much larger. In 2015, product sales were down $642,273 or 80%. The main reason for the decreases in the current year was that in the prior year there was one large customer order for approximately $600,000 in the first half of the year for a custom version of our graphene nanoplatelets. The table below shows a comparison of product sales by product line.
38
Product Sales Summary
|
| Nine Months ended |
| Change 2014 – 2015 | ||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||
Product |
|
|
|
|
|
|
|
|
|
|
|
|
xGnP Bulk Material |
| $ | 112,594 |
| $ | 766,486 |
| (653,892 | ) |
| (85.3 | ) |
Battery Anode Materials (SiG) |
|
| 21,965 |
|
| 14,023 |
| 7,942 |
|
| 56.6 |
|
XG Leaf |
|
| 12,134 |
|
| 13,873 |
| (1,739 | ) |
| (12.5 | ) |
Inks and Coatings |
|
| 9,915 |
|
| 4,499 |
| 5,416 |
|
| 120.4 |
|
TOTAL |
| $ | 156,608 |
| $ | 798,881 |
| (642,273 | ) |
| (80.4 | ) |
We ship our products from our Lansing manufacturing facility to customers around the world. During the first nine months of 2015 we shipped materials to customers in 21 different countries, versus 24 countries during the corresponding period in 2014. Shipments to only one country other than the United States accounted for more than 10% of product revenues during the nine months ended September 30, 2015 and 2014. That country (South Korea) accounted for approximately 36% and 9% of total product revenues during the respective periods.
The table below shows a comparison of orders received, both domestic and international, and the average order size in dollars. These numbers indicate that our customer base remains active with development or research projects that use our materials and indicate the breadth of our geographic coverage. However, the small average order size indicates that almost all orders are for quantities that are far smaller than anticipated orders once our products are commercialized in an industrial or consumer product. Year to date orders have increased nearly 45% as compared to previous year periods. Note that the average order size decreased compared to previous year periods due to the previously mentioned custom order placed in 2014. We do not consider these fluctuations to be especially significant, given the relatively small order size in comparison to our expectations for future commercial orders.
Order Summary
|
| Nine Months Ended |
| Change 2014 – 2015 | |||||||||
|
| 2015 |
| 2014 |
| $ |
| % | |||||
Number of orders – domestic |
|
| 259 |
|
| 179 |
|
| 80 |
|
| 44.7 |
|
Number of orders – international |
|
| 158 |
|
| 155 |
|
| 3 |
|
| 1.9 |
|
Number of orders – total |
|
| 417 |
|
| 334 |
|
| 83 |
|
| 24.9 |
|
Average order size – $ |
| $ | 1,771 |
| $ | 2,798 |
| $ | (1,027 | ) |
| (36.7 | ) |
Grant revenues of $299,879 in 2015 consisted entirely of revenue from the Phase II SBIR grant from the US Department of Energy. This Phase II SBIR was a grant awarded from the DOE as a follow-on for continued development of the materials investigated under the Phase I program entitled “Low-cost, High-Energy Si/Graphene Anodes for Li-Ion Batteries.” This award was for a total of $999,899 to fund a research project over a planned two-year period commencing in January 2014 and expiring December 2015. A total of $762,972 has been billed against the grant, $463,093 in 2014, and $299,879 as of September 30, 2015. All 2014 and 2015 grant revenues are recorded as time and expenses are incurred according to the grant contracts. The table below shows the components of grant revenue.
Components of Grant Revenue |
| Nine Months Ended September 30 | ||||
|
| 2015 |
| 2014 | ||
US Department of Energy grant |
| $ | 299,879 |
| $ | 354,187 |
Navitas Systems sub-contract award |
|
| — |
|
| 30,000 |
Michigan Economic Development Corporation Emerging Technologies award |
|
| — |
|
| 128,000 |
Pure Michigan Connect Technology award |
|
| — |
|
| 39,440 |
|
| $ | 299,879 |
| $ | 551,627 |
39
Licensing revenues during the nine months ended September 30, 2015 and 2014 were accrued based on minimum royalty payments from a production license granted in 2011, which stipulates a minimum of $100,000 in royalties are due to XGS starting in 2014 and annually thereafter. These revenues are being accrued at the rate of $25,000 per quarter. However a dispute has arisen regarding interpretation of the licensing agreement, so we reserved half of these revenues as a potential bad debt. Please see the discussion of bad debt expense.
Cost of Goods Sold
We use a standard cost system to estimate the direct costs of products sold. Direct costs include estimates of raw material costs, packaging, freight charges net of those billed to customers, and an allocation for direct labor and manufacturing overhead. Because of the nature of our production processes, there is a substantial fixed manufacturing expense requirement that represents the ongoing cost of maintaining production facilities that are not directly related to products sold, so we use a “full capacity” allocation of overhead based on an estimate of what product costs would be if the manufacturing facilities were operating on a full-time basis and producing products at the designed capacity. This estimate involves estimating both the level of expenses as well as production amounts as if the manufacturing facility were operating on a continuous, three-shift, production basis.
The following table shows the relationship of direct costs to product sales for the nine month periods ended September 30, 2015 and 2014:
Gross Profit Summary
|
| Nine Months Ended September 30 |
| Change 2014 – 2015 | ||||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||||
Product Sales |
| $ | 156,608 |
|
| $ | 798,881 |
|
| (642,273 | ) |
| (80.4 | ) |
Direct Costs |
|
| 77,516 |
|
|
| 209,858 |
|
| (132,342 | ) |
| (63.1 | ) |
Direct Cost Margin |
| $ | 79,092 |
|
| $ | 589,023 |
|
| (509,931 | ) |
| (86.6 | ) |
% of Sales |
|
| 50.5 | % |
|
| 73.7 | % |
|
|
|
|
|
|
Unallocated Manufacturing Expense |
|
| 1,264,432 |
|
|
| 1,835,178 |
|
| (570,746 | ) |
| (31.1 | ) |
Gross Profit (Loss) on Product Sales |
| $ | (1,185,340 | ) |
| $ | (1,246,155 | ) |
| 60,815 |
|
| 4.9 |
|
We believe that the fluctuations in direct cost from period to period are not indicative of overall performance or of future margins because of the relatively small size of our sales in comparison to our future expectations. Direct costs vary depending on the size of an order, the specific products being ordered, and other factors like shipping destination. Furthermore, the sales for the first half of 2014 were influenced by one large order for custom materials which was priced with higher than average prices as compared with our standard materials.
Costs associated with grant revenues tend to be a mixture of facilities use, management time, labor from scientists, technicians and manufacturing personnel, and some supplies. Because of the difficulty of developing and maintaining an administrative system to gather direct costs for grants, together with the relatively small size of grant revenues, we do not track direct costs for grant revenues as a separate cost category. Therefore, we do not calculate direct cost margins associated with grant revenues but, rather, we view these revenues as being supported by indirect corporate expenses.
Costs associated with licensing revenue tend to be a mixture of IP costs as well as management and administrative expenses that are indirect in nature. As such, we do not assign direct costs to licensing revenues. Where revenues from a license agreement can be assigned to specific product revenues, we classify these revenues as product sales and, using our standard cost system, assign direct costs to those sales.
The remaining “non-direct” costs of operating our manufacturing facilities are recorded as unallocated manufacturing expenses. This expense includes personnel costs, rent, utilities, indirect supplies, depreciation, and related indirect expenses. Unallocated manufacturing expenses are expensed as incurred.
Overall product margins are greatly influenced by unallocated manufacturing expenses, which are those overhead costs that represent the fixed costs of our factory, production personnel, utilities, and so forth that are not yet being covered by our sales level. We allocate these costs as direct product costs on the basis of the proportion of these expenses that would be representative product costs if we were operating our factory at full capacity.
40
For the nine months ended September 30, 2015, unallocated manufacturing expenses were down by $570,746 or 31% from the comparable period in 2014. The largest part of this decrease was decreased payroll, tax and benefit costs caused by a reduction in work force. Total manufacturing personnel fluctuated from period to period. On September 30, 2015 there were 15 manufacturing personnel as compared to 27 on September 30, 2014. Overall decreases in other unallocated expense areas were due to cost containment measures.
The following table shows a comparison of the major expense categories that make up unallocated manufacturing expense for the periods ended September 30, 2015 and 2014:
Manufacturing Expense |
| Nine Months ended |
| Change 2014 – 2015 | ||||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||||
Payroll, Tax & Benefits |
| $ | 316,757 |
|
| $ | 661,725 |
|
| (344,968 | ) |
| (52.1 | ) |
Materials Used |
|
| 70,909 |
|
|
| 114,849 |
|
| (43,940 | ) |
| (38.3 | ) |
Supplies |
|
| 41,944 |
|
|
| 68,159 |
|
| (26,215 | ) |
| (38.5 | ) |
Equipment Repairs & Maintenance |
|
| 97,506 |
|
|
| 153,630 |
|
| (56,124 | ) |
| (36.5 | ) |
Building Lease |
|
| 165,205 |
|
|
| 143,012 |
|
| 22,193 |
|
| 15.5 |
|
Utilities |
|
| 132,231 |
|
|
| 144,524 |
|
| (12,293 | ) |
| (8.5 | ) |
Depreciation |
|
| 504,909 |
|
|
| 509,770 |
|
| (4,861 | ) |
| (1.0 | ) |
Tools & Equipment |
|
| 15,943 |
|
|
| 44,314 |
|
| (28,371 | ) |
| (64.0 | ) |
All Other |
|
| 42,615 |
|
|
| 177,952 |
|
| (135,337 | ) |
| (76.1 | ) |
Allocated to Direct Costs |
|
| (123,587 | ) |
|
| (182,757 | ) |
| 59,170 |
|
| 32.4 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | 1,264,432 |
|
| $ | 1,835,178 |
|
| (570,746 | ) |
| (31.1 | ) |
Research and Development Expenses
The following table shows a comparison of the major expense categories that make up research and development expenses for the periods ended September 30, 2015 and 2014:
Research & Development Expenses |
| Nine Months ended September 30 |
| Change 2014 – 2015 | ||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||
Payroll, Tax & Benefits |
| $ | 658,127 |
| $ | 637,539 |
| 20,588 |
|
| 3.2 |
|
Lab Supplies |
|
| 77,089 |
|
| 31,951 |
| 45,138 |
|
| 141.3 |
|
Equipment Repairs & Maintenance |
|
| 15,606 |
|
| 1,891 |
| 13,715 |
|
| 725.3 |
|
Building Lease |
|
| 84,859 |
|
| 81,026 |
| 3,833 |
|
| 4.7 |
|
Utilities |
|
| 24,043 |
|
| 16,700 |
| 7,343 |
|
| 44.0 |
|
Depreciation |
|
| 110,700 |
|
| 105,324 |
| 5,376 |
|
| 5.1 |
|
Tools & Equipment |
|
| 38,867 |
|
| 11,905 |
| 26,962 |
|
| 226.5 |
|
Contracted Services |
|
| 94,051 |
|
| 100,532 |
| (6,481 | ) |
| (6.4 | ) |
All Other |
|
| 31,984 |
|
| 1,281 |
| 30,703 |
|
| 2396.8 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | 1,135,326 |
| $ | 988,149 |
| 147,177 |
|
| 14.9 |
|
There were sizable increases in the current year for purchases of lab supplies, tools, equipment and maintenance as well as contracted services. Most of the current year increases are attributable to increased activity on the US Department of Energy (DOE) grant previously mentioned. Payroll related expenses in the current year are higher than prior year because 2015 includes nine months of a senior research scientist who was only here one month in the prior year.
41
Sales, General and Administrative Expenses
The following table shows a comparison of the major expense categories that make up sales, general and administrative expenses for the periods ended September 30, 2015 and 2014:
Sales, General & Administrative Expenses |
| Nine Months ended |
| Change 2014 – 2015 | ||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||
Payroll, Tax & Benefits |
| $ | 1,818,317 |
| $ | 1,870,601 |
| (52,284 | ) |
| (2.8 | ) |
Non-Cash Stock Option Compensation |
|
| 387,998 |
|
| 395,403 |
| (7,405 | ) |
| (1.9 | ) |
Building Lease |
|
| 43,874 |
|
| 37,950 |
| 5,924 |
|
| 15.6 |
|
Utilities |
|
| 23,218 |
|
| 22,439 |
| 779 |
|
| 3.5 |
|
Professional Fees |
|
| 348,690 |
|
| 556,114 |
| (207,424 | ) |
| (37.3 | ) |
General Business Insurance |
|
| 53,914 |
|
| 27,976 |
| 25,938 |
|
| 92.7 |
|
Capital Funding Expenses |
|
| — |
|
| 56,287 |
| (56,287 | ) |
| (100.0 | ) |
Travel & Entertainment |
|
| 57,398 |
|
| 94,701 |
| (37,303 | ) |
| (39.4 | ) |
Directors Fees & Expense |
|
| 54,550 |
|
| 58,875 |
| (4,325 | ) |
| (7.3 | ) |
Samples |
|
| 73,498 |
|
| 7,286 |
| 66,212 |
|
| 908.8 |
|
Bad Debt Expense |
|
| 94,027 |
|
| — |
| 94,027 |
|
| 100.0 |
|
Office Expense and Supplies |
|
| 41,337 |
|
| 70,046 |
| (28,709 | ) |
| (41.0 | ) |
All Other |
|
| 136,554 |
|
| 222,447 |
| (85,893 | ) |
| (38.6 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Total |
| $ | 3,133,375 |
| $ | 3,420,125 |
| (286,750 | ) |
| (8.4 | ) |
During 2015, the overall expenses in this category decreased 8%, from the corresponding period in 2014, primarily because of sizable decreases in payroll, professional fees, capital funding expenses, travel and entertainment, and office expenses and all other general costs.
The most notable increases can be seen in our sample costs and our bad debt expense. The current year includes $37,000 of sample cost for one large customer. The current year also includes a charge for $75,000 to bad debt expense which represents an allowance for the potential that a portion of the $150,000 recorded in Other Current Assets at September 30, 2015 may not be collectible from the licensing agreement with POSCO (mentioned previously). The terms of the agreement are being disputed by POSCO and may have to be settled by arbitration.
Other Income (Expense)
The following table shows a comparison of other income and expense by major expense component for nine month periods ended September 30, 2015 and 2014:
Other Income (Expense) |
| Nine Months ended September 30 |
| Change 2014 – 2015 | ||||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||||
Incentive Refund & Interest Income |
| $ | 80,624 |
|
| $ | 54,584 |
|
| 26,040 |
|
| 47.7 |
|
Interest Expense |
|
| (1,640,668 | ) |
|
| (1,064,760 | ) |
| (575,908 | ) |
| 54.1 |
|
Gain (loss) from change in fair value of derivative liability – warrants |
|
| (143,845 | ) |
|
| 462,664 |
|
| (606,509 | ) |
| (131.1 | ) |
Extinguishment Loss on Covertible Notes |
|
| — |
|
|
| (3,513,770 | ) |
| 3,513,770 |
|
| (100.0 | ) |
Total |
| $ | (1,703,889 | ) |
| $ | (4,061,282 | ) |
| 2,357,393 |
|
| (58.4 | ) |
Interest expense for the nine months ended September 30, 2015 is $575,908 higher as compared to the same period in the prior year because the current year reflects interest on $13.7 million of convertible notes and in prior year the convertible note balances fluctuated between $4.5 million and $13.7 million as proceeds from the note sales occurred throughout the year. The loss on extinguishment of convertible notes in 2014 was a one-time entry to reflect the note modifications executed on January 15, 2014.
42
Cash Flow Summary
Ongoing cash usage for operations has decreased in the first nine months of 2015 as compared to the same period in 2014 because of reduced operating expenses, as discussed above. The following simplified cash flow statement compares cash usage for the first 9 months of the past two years.
Summary Cash Flow |
| Nine Months ended September 30 |
| Change 2014 – 2015 | ||||||||||
|
| 2015 |
| 2014 |
| $ |
| % | ||||||
Cash, beginning of period |
| $ | 2,088,866 |
|
| $ | 95,342 |
|
| 1,993,524 |
|
| 2090.9 |
|
Net Cash provided (used) by: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (4,591,143 | ) |
|
| (5,315,873 | ) |
| 724,730 |
|
| (13.6 | ) | |
|
| (217,646 | ) |
|
| (820,749 | ) |
| 603,103 |
|
| (73.5 | ) | |
|
| 4,305,028 |
|
|
| 9,582,433 |
|
| (5,277,405 | ) |
| (55.1 | ) | |
Net increase (decrease) in cash |
|
| (503,761 | ) |
|
| 3,445,811 |
|
| (3,949,572 | ) |
| (114.6 | ) |
Cash, end of period |
| $ | 1,585,105 |
|
| $ | 3,541,153 |
|
| (1,956,048 | ) |
| (55.2 | ) |
Accounts receivable balances of $67,258 and $96,457 at September 30, 2015 and 2014 respectively are included in the current assets line item in the table above (net of a $10,000 allowance for uncollectible accounts in each year.
Inventory balances of $211,052 and $140,836 at September 30, 2015 and 2014 respectively are included in the current assets line item in the table above. The increase of $70,216 represents increased finished product on hand in the current year as the Company ramps up for anticipated sales volume in the future.
Current assets at September 30, 2015 are $1,872,171 lower than the previous year. The decrease is primarily caused by the decrease in available cash due to the reduction of new financing from 2014 to 2015 discussed further below
A decrease in accounts payable of $179,472 for 2015 and $118,065 for 2014 reflects the pay down of high accounts payable balances at both fiscal year-ends. The payables balances were high at year-end because of cash flow management related to the timing of new financings.
During 2014, we had significant financing activity and raised a total of $9.7 million in new financing, primarily from the sale of convertible notes, giving us a cash balance on September 30, 2014 of $3.5 million. In the first half of 2015 we raised an additional $4.3 million in new financing through the sale of Series B Units at a price of $16 per unit.
On December 31, 2015, we issued the December Notes and December Warrants to purchase 20,625 shares of common stock to several existing stockholders. The December Notes mature on June 30, 2016 and the December Warrants have a five year term and a strike price of $8.00. As of the date of this prospectus, we have raised $550,000 under this December 2015 capital raise. We cannot determine how much additional funds we might raise under this private placement.
Net capital expenditures for the purchase of property and equipment were $157,983 during 2015, down from $682,845 during the corresponding period in 2014. These levels of capital expenditures are relatively lower than expected in the future as we begin to ramp up our production capacity to meet customer orders. Therefore, these expenditures should not be interpreted as indicative of future expenditures in this area. Overall investment activities used a total of $217,646 during 2015, much lower than the $820,749 spent during 2014.
Liquidity and Capital Expenditures
Ongoing cash flow from operations has been negative throughout our history. In addition, we have invested significant amounts in research and manufacturing capabilities. We anticipate that we will need to invest further to support ongoing operations for a minimum of another one to two years. We believe that cash on hand is sufficient to finance our business through at least April 2016 and we are, therefore, contemplating raising additional funds from private sources that may be in addition to any funds resulting from the sale of shares of common stock in the primary offering hereunder.
We also currently forecast a need for additional capital expenditures to execute on our business plan. The amount and timing of such expenditures will be dependent on the timing and magnitude of sales orders received from customers, but we currently anticipate a need to invest approximately $1 million to $6 million per year during the upcoming three-year period. We plan to fund these investments through a mixture of capital lease financing, debt financing, and equity financing. If we are unable to obtain such financing, we may be forced to curtail our investment activities and this may limit our ability to grow our revenues as fast as we would like.
43
Additionally, we anticipate that as our sales increase we will need to invest funds in working capital to support additional inventory and accounts receivable. We anticipate that, for every $1 million increase in annual sales, we may need to invest as much as $250,000 in working capital.
Taken together, these multiple cash requirements will likely exceed our cash flow from operations for at least two to three years, so we foresee a need to continue investing in our business. Although there can be no guarantee of successful funding in the future, we intend to pursue the sale of additional equity, as well as additional debt or lease financing in the future, until such time as ongoing revenues begin to generate a positive monthly cash flow from the business sufficient to cover our cash requirements.
44
Overview
XG Sciences, Inc. was formed in May 2006 for the purpose of commercializing certain technology to produce graphene that had been developed at MSU. First discovered in 2004, graphene is a single layer of carbon atoms configured in an atomic-scale honeycomb lattice, and is noted for its strength, density, impermeability, light weight, and both electrical and thermal conductivity. Among many noted properties, graphene is harder than diamonds, lighter than steel but 200–300 times stronger, and conducts electricity better than copper. Graphene nanoplatelets are particles consisting of multiple layers of graphene. The technology licensed from MSU consists of several U.S. patents, patent applications in process and MSU Invention Disclosures. In addition, our in-house personnel have made many further discoveries and inventions that are embodied in the form of issued patents, additional patent applications, and trade secrets and other proprietary information. (See also subsection entitled “Description of Our Business — Intellectual Property” below).
We manufacture and sell a material we call xGnP® brand graphene nanoplatelets. This material consists of ultrathin particles of graphite that are short stacks of graphene. We currently offer this material in different thicknesses, designated by the “grade” of material, as well as different particle sizes within each grade. There are three commercially available grades, each of which is currently offered in three standard particle sizes. We have other developmental grades that may be made available on a commercial basis if their need in the market warrants commercialization. We believe graphene nanoplatelets are a game changer for numerous industries and we estimate the market for graphene will quickly grow to be a billion dollar opportunity with XGS as a first mover. Graphene is a new material and is just beginning to find commercial applications. However, not all graphene is the same. The primary manufacturing processes in use today are plotted below as a function of their ability to tailor both thickness (correlates with surface area) and particle size.
These attributes, as summarized below, will impact their fit for various applications. XG Sciences uses two primary, proprietary processes to create our xGnP® nanoplatelets. We believe we are well positioned from both a cost and performance perspective to be able to address a wide range of end-use applications. We have chosen to not pursue a graphene oxide process as it is inherently more costly, produces nanoplatelets with more surface functional groups and higher levels of dislocation (e.g., holes) in the basel planes, both of which tend to degrade electrical and thermal performance. Gas phase processes produce very small particle sizes which make them slightly better for applications where optical purity is a requirement. However, gas phase processes are both costly and difficult to scale. Expanded graphite is created by swelling natural graphite in a solvent like NMP and then sonicating (the act of applying sound energy to agitate particles) to separate the layers. The process tends to make rather thick material which limits the amount of platelet overlap that can be achieved and therefore degrades properties such as electrical and thermal conductivity. In addition, many applications will not tolerate residual solvent that is difficult and costly to remove.
45
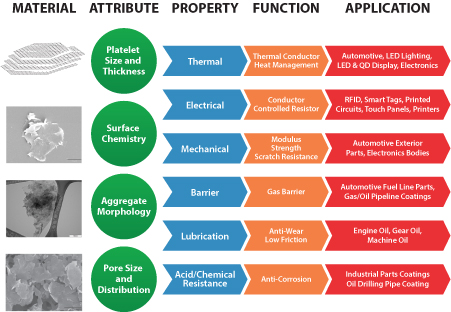
Graphene has been hailed as a “miracle material” because of its many potential uses in products ranging from hand-held electronics to automobiles to spacecraft. Its material properties include very high strength, very high thermal conductivity, good electrical conductivity, strong barrier properties, and very high stiffness. These properties make graphene potentially useful in thousands of specific products.
The term “graphene” is used in the popular press to cover a variety of specific forms of the material, but we generally think about two broad classes of graphene materials:
1. One-atom thick films of carbon, manufactured from gases by assembling molecules to form relatively large, transparent sheets of material. We do not manufacture these films and do not participate in the markets for these films.
2. Ultra-thin particles of carbon that usually consist of a few layers of graphene sheets. Because they are thin and can be manufactured in a range of diameters, these graphene particles are useful for a wide variety of applications. Our typical particles are about 5 nanometers thick — or about 50,000 times thinner than a human hair and range in diameter from less than 1 micron to 100 microns. The manufacture of these graphene particles is our main area of expertise, and their use in practical applications is the focus of our marketing and development efforts.
XG Sciences offers an advanced material platform in that we can produce varying grades of graphene nanoplatelets, each of which can be customized in many ways. Our manufacturing processes control the attributes of the graphene nanoparticles as summarized below. These attributes contribute to a range of properties that can be mapped to various functions and applications. Because we have multiple production processes and because we have invested in application know-how and development of value-added product formulations, we are able to address a range of market needs in multiple market segments, providing breadth to our base capabilities and product portfolio.
Graphene market projections are difficult to accurately predict. This is primarily related to the fact that it is a relatively new material for which no historical use data exists on a commercial scale. However, because graphene is a new form of carbon and with unique performance attributes, one may consider other carbon forms such as carbon nanotubes or carbon fibers or activated carbon as a basis for estimating the potential market size for graphene where graphene’s enhanced performance may
46
take market share from one or more of the more established forms of carbon. We also estimate target market sizes by using a bottom-up approach where we look at existing applications for graphene and estimate sales by year as a function of graphene’s unique performance attributes and value proposition for specific customers. In some instances, we are able to get fairly granular data on market estimates. In other areas, we must infer a potential market size from related materials.
The figure given below provides estimates for the 2018 total available market in a range of target applications. Since our advanced materials are sometimes used as a replacement material to supplant the current materials used in these applications and are sometimes used as an additive material to augment the incumbent materials, it is difficult to determine exact market potential by segment. This information does provide an indication the relevant markets and their respective sizes. Naturally, we do not expect to capture all of these markets, but we do see opportunities to capture significant market share (1% to 20%) in these targeted markets. However, there can be no guarantees. The following discussion applies to the individual segments of the chart:
• “High strength composites” is the size of the carbon fiber market used in advanced composites for automobiles and industrial applications. xGnP® offers improvements in physical properties in a range of matrices and is a candidate to augment and/or substitute carbon fibers in high strength composites.
• “Lithium Ion Battery Anode Active Materials” is a forecast for total carbon materials sold into the application, for which our SiG materials are a potential direct replacement with significant performance advantages.
• “Thermal Paper” is the forecast annual sales of “carbon-based” sheet products used to control thermal spreading or provide heat management in portable electronic devices.
• “Automotive” includes miscellaneous automotive applications such as electronic shielding of engine or control components, circuits printed with conductive inks, and thermal shielding.
• “Sensors” includes carbon-based sheets or printed substrates into which our materials may be incorporated to improve results.
• “Packaging” includes conductive inks or coatings printed on packages as part of circuits or sensors.
• “RFID” means Radio Frequency Identification Devices that are printed or installed on packaged items to facilitate tracking. Our materials can be used in conductive inks and components used to print circuits, batteries, or antennas.
• “Supercapacitors” means high-capacity capacitors that are typically fabricated with electrodes made from high-surface area carbon materials.
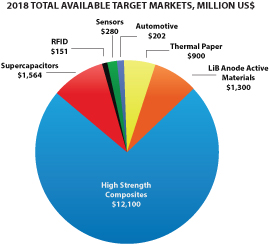
47
Note: Market sizes estimated by XG Sciences, based on customer input as well as reference market sizes for equivalent or identical materials from outside market research sources including:
(1) ID Tech “Graphene Markets, Technologies and Opportunities 2013 – 2018.”
(2) ID Tech “Conductive Ink Markets 2014 – 2024, Silver & Copper Inks & Pastes — and Beyond.”
(3) Avicenne Energy “The Rechargeable Battery Market, 2014 – 2025” July 2015.
(4) Prismark Partners “Thin Carbon-Based Heat Spreaders” August 2014.
ID Tech, in a recently published study (Graphene Markets, Technologies and Opportunities 2013–2018), estimates 2018 global graphene sales slightly in excess of $100 million and in a range of applications. Their projections are based largely on replacement of existing carbon-based materials and with very minimal market penetration. They do not consider such products as SiG, and XG Leaf® that are new products incorporating our ability to tailor the form and function of xGnP® into value-added product platforms. We expect to generate sales from the design and manufacture of both our xGnP® nanoplatelets and products derived therefrom. As such, we are not able to rely on market projections such as those provided by ID Tech and use a combination of total available market data, customer input and internal estimates. We have sold materials to more than 850 customers, many of whom are household names, evaluating graphene for numerous different applications, including, but not limited to lithium ion batteries, supercapacitors, thermal shielding and heat transfer, inks and coatings, printed electronics, construction materials, composites, and military uses.
In addition to our bulk material, which normally ships in the form of a dry powder, we also offer our materials in the form of dispersions of nanoplatelets in liquids such as water, alcohol, or organic solvents, or mixed into resins or polymers such as epoxies or urethanes. Additionally, we offer a sheet product, called XG Leaf®, to potential customers for a variety of thermal spreading and other applications. XG Leaf® is ideally suited for use in thermal management in cell phones, tablets and PC’s. As these devices continue to adopt faster electronics, higher data management capabilities, brighter displays with ever increasing definition, they generate more and more heat. Managing that heat is a key requirement for the portable electronics market and our XG Leaf® product line is well suited to address the need. In a press release dated, March 3, 2015, Gartner, Inc., a leading research organization, estimated the 2014 global cell phone market at 1.88 billion units. Every cell phone has some form of thermal management system, and we believe many of the new smart phones being developed can benefit from the performance advantages we are able to achieve with XG Leaf®. In August 2015, IDC in their Worldwide Quarterly Tablet Tracker estimated the global shipment of tablets 2015 at 212 million units. Thus, we believe our XG Leaf® product line is well positioned to address a very large and rapidly growing market.
We also offer a specially formulated silicon-graphene composite material (also referred to as “SiG” in this prospectus) for use in lithium-ion battery anodes. SiG targets the never-ending need for higher battery capacity and longer life. In several customer trials, our SiG material has demonstrated the potential to increase battery energy storage capacity by 3-5x what is currently available with conventional lithium ion batteries today. The market for materials used in lithium ion battery anodes is large and growing as shown in the figure below (Avicenne Energy, “The Rechargeable Battery Market, 2014-2025”, July 2015). We believe our ability to address next generation anode materials represents a significant opportunity for us.
48
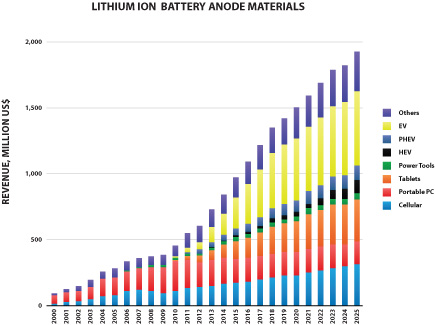
We also offer specially-formulated inks for electrical and thermal applications that are showing promise in diverse customer applications such as advanced packaging, electrostatic dissipation and thermal management.
These three product areas — custom XG Leaf® sheets, our XG SiG™ anode materials, and custom inks/coatings — comprise the set of core value-added product groups on which we are focusing our internal development resources.
Significant Accomplishments
Since our founding in May 2006, we have been one of the global pioneers in graphene nanoplatelet markets. As a prospective manufacturer of a newly discovered advanced material, our development efforts required us to invent appropriate manufacturing processes and methods to scale these processes at economic costs, to hire world-class employees, to introduce our products to customers around the world, and to demonstrate the scientific merits of our materials. Specific accomplishments related to these tasks include, but are not limited to, the following:
• Developed and launched three grades (Grades C, H & M) of xGnP® graphene nanoplatelets bulk powders, each of which is offered in three standard particle sizes, for a standard bulk product portfolio of nine different materials.
• Developed and launched three higher margin, value-added product families:
- XG Leaf® sheet products for a variety of electronics and industrial applications.
- Silicon-graphene composite anode materials for lithium-ion batteries.
- Custom formulated inks and coatings designed to take advantage of the special properties of our graphene nanoplatelets in targeted customer applications.
• Established sales distributors in Japan, South Korea, Taiwan, Italy, Germany and China.
49
• Completed a research contract from the US Air Force for high energy-density ultra-capacitors.
• Won a competition that resulted in a research contract from the U.S. Department of Energy (DOE) for ongoing development of high-capacity anode materials for electric vehicles based on the Company’s SiG material.
• Secured an additional $1.0 million grant from the DOE for further research into silicon graphene.
• Collaborated with Oak Ridge National Laboratory to explore metal-matrix materials using xGnP graphene nanoplatelets to modify the properties of metals like titanium.
• Was recognized as a world leader in the graphene market space by Lux Research and named one of the “Top Ten Innovative Companies” profiled by Lux.
• In March 2015 was named a “Michigan 50 Companies to Watch” by Michigan Celebrates Small Businesses.
• Hired Dr. Philip L. Rose as CEO. Prior to joining XGS, Dr. Rose was President of SAFC Hitech, a $100 million division of Sigma-Aldrich Corporation that makes precursors and performance materials for the LED, energy and display, and semiconductor markets.
• Hired Dr. Liya Wang, who led the A123 Systems next generation cathode development efforts, as Director of Emerging Technologies.
• Raised $31.2 million of equity and equity-linked capital from inception to August 31, 2015.
In addition, set forth below are tables which evidence our customer growth over time and orders by year from 2008 through 2014:
|
|
|
The above graphs show total orders and customers based on actual purchases of our materials and do not include free samples or materials used in joint development programs. Nevertheless, the total dollar volume of these orders is not yet sufficient to cover our fixed operating expenses. The average order size in 2014, for example, was $2,175, which indicates to us that most of these orders are for materials that are not yet incorporated into large-volume commercial products.
Products
We manufacture and sell a material we call xGnP® brand graphene nanoplatelets. This material consists of ultrathin particles of graphite that are short stacks of graphene. We currently offer this bulk material in different thicknesses, designated by the “grade” of material, as well as different particle sizes within each grade. There are three commercial grades (Grades C, H & M), each of which is offered in three standard particle sizes. We use two different commercial processes to produce these bulk materials:
50
• Grade H/M materials are produced through chemical intercalation of natural graphite followed by thermal exfoliation. The “grade” designates the thickness of the material, and each grade is available in various average particle diameters. Surface area, calculated by the Brunauer, Emmet, and Teller (BET) Method, is used as a convenient proxy for thickness, so each grade in this area is designated by its average surface area, which ranges from 50 to 150 m2/g of material.
• Grade C materials and some related composite materials are produced through a high-shear mechanical exfoliation using a process and equipment that we invented, designed, and constructed. The Grade C materials are smaller particles than the other grades, and Grade C materials are designated by their BET surface area, which ranges from 300 to 800 m2/g.
In addition to our bulk material, which normally ships in the form of a dry powder, we also offer our material in the form of dispersions of nanoplatelets in liquids such as water, alcohol, or organic solvents, or mixed into resins or polymers such as epoxies or urethanes. We recently introduced a new Grade of xGnP® powders, R-Grade, with improved electrical conductivity targeting use in LiB as a cathode conductive additive and for use in various conductive ink applications.
Additionally, we offer a value-added product line we call XG Leaf®, which consists of thin sheets formulated with the Company’s materials to meet a variety of thermal management and heat shielding applications.
We also offer a mixture of materials that we call “silicon-graphene composite material” (“SiG”, or “XG SiGTM”) that is intended for use in formulating anodes of lithium-ion batteries. This new material recently demonstrated 3-5 times the energy capacity of traditional carbon graphite anodes used in lithium ion batteries today, which the Company believes may be a revolutionary breakthrough for the battery market.
We also offer a set of standardized ink formulations suitable for printing. These inks offer the capability to print electrical circuits or antennas, or might be suitable for other electrical or thermal applications. All of these formulations can be customized for specific customer requirements.
Thus, we manage our business with four major product lines at this time:
• Bulk materials, which we sell under the trademarked brand name of xGnP® graphene nanoplatelets. These materials are produced in various grades, which are analogous to average particle thickness, and average particle diameters. Overall, there are three different standard commercial grades, each of which is available in three different standard particle sizes and multiple customer sizes. These bulk materials are especially applicable for use as additives in polymeric or metallic composites, or in coatings or other formulations where particular electrical, thermal or barrier applications are desired by our customers.
• Energy storage materials. These consist of specialty advanced materials that have been formulated for specific applications in the energy storage segment. Chief among these is our silicon-graphene composite material that we offer to manufacturers of li-ion batteries. Additionally, we offer various bulk materials for use as conductive additives for cathodes and anodes in li-ion batteries, as an additive to anode slurries for lead-carbon batteries and are investigating the use of our materials as part of other battery components.
• XG Leaf®. XG Leaf® is a family of sheet products that we manufacture for industrial, electronics, and automotive markets. These sheets are made using special formulations of xGnP® graphene nanoplatelets as precursors, along with other materials for specific applications. There are several different types of XG Leaf®, varying based on their composition and available in various thicknesses, depending on the end-use requirements for thermal conductivity, electrical conductivity, or resistive heating.
• Inks and Coatings. These consist of specially-formulated dispersions of xGnP® together with solvents, binders, and other additives to make electrically or thermally conductive products designed for printing or coating and which are suitable for a variety of industrial applications.
Our general business plan is to use our scientific and manufacturing expertise to develop specific products based on our xGnP® graphene nanoplatelets for end-use applications in industrial, electronics, aerospace, automotive, military, and similar OEM markets. Our initial focus is on developing high-value products that use our bulk materials as a significant component. We work closely with customers to design our products to meet their needs. Because these needs may be unique to a given customer and/or a given application, we may formulate and ultimately commercialize customer-specific formulations. These
51
formulations, to the extent possible, will be made available to other customers. It is more likely that a new customer will require a combination of our technologies to derive a customer-specific performance outcome. Successful execution requires a close development relationship with customers and an ability to formulate products for use in multiple markets and for multiple customers.
Within the various markets we serve, we prioritize our efforts in specific areas and with specific customers that we believe represent opportunities for either relatively near-term revenue or especially large and attractive markets. At this time, we focus our highest priority in two areas:
• Silicone-graphene composite materials for lithium-ion battery anodes. This is a material that has shown superior early results in laboratory and early prototype battery testing. Although there is still scientific development, testing and prototyping that remain to be done, we perceive a significant market opportunity in this area and we devote almost half of our development efforts to this product.
• XG Leaf® materials for thermal applications in electronics. Early testing with customers has produced promising results in applications requiring high thermal conductivity in thin (20–40 microns) and thick (70–120 microns) sheets, depending upon the end-use application. Typical applications include the use of these materials in smart phones, tablets, and portable computers. While there are many other applications for XG Leaf® that include electrostatic dissipation (“ESD”), electromagnetic interference (“EMI”) shielding and resistive heating, our initial focus is on thermal management.
The following diagram shows some of the specific application areas within our major target markets, and gives a general indication of the time horizons we assign to these application areas at this time.
GRAPHENE NANOPLATELETS, VALUE-ADDED PRODUCTS/APPLICATIONS AND RELATED 2018 TOTAL AVAILABLE MARKETS.
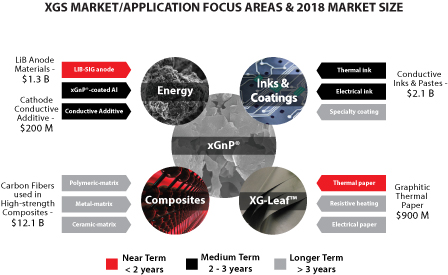
* Near Term means less than 2 years; Medium Term means 2-3 years; and Longer Term means longer than 3 years.
52
Markets
Energy Storage Markets
One of the more exciting near term market opportunities is the market for rechargeable batteries. In general, lithium-ion battery (“LIB”) technology is the dominant technology for rechargeable batteries throughout the world. The global LIB market is large and growing rapidly. The figure below is a representation of the LIB value chain and the associated 2014 revenue, according to Avicenne Energy (The Rechargable Battery Market 2014-2015, July 2015). The 2014 LIB market was $13.6 billion at the cell level and $19.6billion at the pack level (multiple cells sold at the aggregate level). The primary materials used in the production of LIB’s represent $6.0 billion in 2014 revenue. We target our products and technology toward both the cathode and anode material segments. The 2014 market for cathode materials was $2.5 billion with a 2004 to 2014 compound annual growth rate of 16%. The 2014 market for anode materials was $0.8 billion with a 2004 to 2014 compound annual growth rate of 14%. The second figure, 2014 LIB Anode Materials, provides further segmentation of the materials comprising LIB anodes and their relative volume contributing to the $0.8 billion in 2014 revenue.
Our SiG materials target replacement of the carbon and graphite(s) used in current generation LIBs. Because our materials have a much higher energy density than these traditional materials, these market sizes do not translate exactly into the potential for our SiG materials, but we estimate that we are addressing a segment that represents in excess of $500 million sales opportunity by 2016. Although the anode segment that we address with our SIG materials is relatively large, it comprises only about 6% of the overall value of the finished battery. Thus, we believe there is room for premium products in this segment in exchange for higher overall battery performance. XGS is actively engaged with LIB manufacturers to design our SiG material for use as a component of anodes in next generation LIBs. We believe that our SiG material, once successfully developed, qualified and used commercially in next-generation LIBs, will allow for improved storage capacity and battery life — key performance features of interest within the LIB industry.
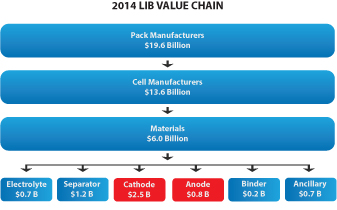
53
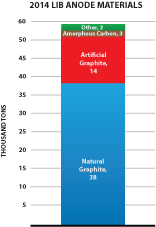
We further target use of our xGnP® materials as conductive additives in LIB cathodes. The figure below provides details of the 2014 market volume for active materials used in LIB cathodes including: nickel cobalt aluminum, lithium iron phosphate (“LFP”), spinel-based lithium-ion (“LMO”), nickel cobalt magnesium and cobalt-based lithium ion (Avicenne Energy, The Rechargeable Battery Market 2014-2025, July 2015). Active cathode materials will vary by end-use performance requirements. Another material used in the cathode is a conductive additive, whose incorporation improves the electron transport characteristics of the cathode — a key performance feature of interest within the LIB industry. LFP- and LMO-based cathodes have relatively low inherent conductivity and can most benefit from use of a conductive additive. Conductive additives are typically used in 1–3% of the weight of the active cathode materials. We are engaged with LIB manufacturers to design our xGnP® materials for use as a cathode conductive additive in next generation LIBs.
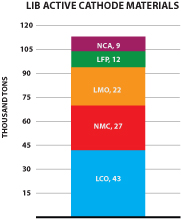
The LIB market is often divided into two major segments, each of which has different technical, cost, and efficiency requirements:
- The industrial market consists of electric and hybrid vehicles, grid storage, and similar heavy-duty applications. These applications require long life, extreme safety, high-power capacity, durability, and the ability to operate in extreme ranges of environmental conditions.
54
- The market for consumer electronics uses LIB technology to power the devices we carry in our pockets, purses and briefcases. These devices need small batteries, with high power density, that charge rapidly and do not cost much. This market is dynamic and has an insatiable appetite for smaller, cheaper, faster-charging, higher capacity batteries.
These market segments have different development timelines and the marketing strategies and the route-to-market that we pursue differs markedly in these two segments.
For industrial applications, we work closely with the DOE, which is the primary driving force in the US, as well as large US battery manufacturers. The DOE operates a number of national laboratories like Argonne, Oak Ridge, Lawrence Berkeley, and Los Alamos national labs, where much of the advanced battery research is performed. The DOE has laid out a “road map” for battery development in the US, with guidelines and targets for performance and costs for the next decade. The DOE spurs development of new technology by sponsoring research through grants and challenge programs. Therefore, we work to maintain close ties with the national laboratories as well as vehicle program managers in Washington D.C. and to participate in DOE grant programs, conferences, and joint development programs. Although we hope to receive grants to offset some of the research costs, the main advantages from this approach are that we stay abreast of technology developments and we get our technology dispersed through the battery research community in a highly credible manner. The development cycle for industrial batteries is a multi-year effort that is spearheaded by billions of dollars of government spending.
For consumer applications, we focus on the large consumer battery manufacturers, which are almost all in Asia. These companies perform their own research, development, and battery engineering. Names like Samsung, Panasonic, and LG Chem are familiar to most Western consumers, but other companies like ATL, Envision, Wanxiang, BYD, Lishen, BAK, and many other Chinese companies are rapidly growing and represent potentially large sales opportunities for XGS. Our general strategy in the Asian markets is to engage customers on sampling and joint development to design our technology into our customer’s products. We have a network of distributors, who have close connections to the customers in their countries and also provide assistance in customer collaboration.
Our main internal development focus has been on the use of our proprietary manufacturing processes and proprietary materials to produce a composite material composed primarily of silicon and graphene. We also develop xGnP® materials for use as conductive aids in both the cathode and anode formulations. We believe that our manufacturing processes and ability to control the structure of the SiG composite material at the nano-level offer the opportunity for us to develop new battery anode materials. During the past year, we made significant progress with our development efforts and have demonstrated in our labs a silicon-graphene nanocomposite material that delivers 3–5 times the energy storage capacity of traditional carbon graphite anodes. This material is produced using our proprietary process using readily available, low-cost precursors.
We believe this is a significant breakthrough for lithium-ion battery development for two reasons. First, this material has the highest capacity of any electrode that we are aware of that is currently available on the commercial market. Second, we believe we can offer this material at prices that are acceptable to most commercial battery manufacturers and at the needed scale. We believe that the combination of commercially viable pricing, demonstrated manufacturing scale and high performance for this new anode material will give us access to many of the more significant battery makers in the world today.
In January 2014, we announced an investment by Samsung Ventures in the Company’s convertible secured notes. As part of the investment, we agreed with Samsung Ventures that the two companies would engage in a joint development program to develop anode materials for lithium-ion batteries for use in certain Samsung applications. One Samsung subsidiary, Samsung SDI, is one of the world’s largest manufacturers of lithium-ion batteries. XGS and Samsung SDI entered into an agreement on April 8, 2014 to jointly develop and optimize our SiG for use in SDI’s lithium ion batteries.
XG Leaf® Markets
XG Leaf® is a unique family of thin sheets based on our graphene nanoplatelets. These sheets can be used for a variety of applications such as heat dissipation in electronics, ESD, EMI shielding, and resistive heating. Among these, thermal management applications in portable electronics have the biggest short-term market potential. There is an increased need for advanced thermal management in electronics of all sorts, but especially consumer electronics. As these products become smaller, thinner and more powerful, thermal management challenges are a major engineering barrier that needs to be overcome if the industry is to provide continued improvements in device and system performance. We developed XG Leaf® to meet such a market need.
55
The key requirements for these products are thermal conductivity, thickness, mechanical strength, and cost. Currently, there are high-end products in the market such as highly ordered pyrolytic graphite (HOPG) papers which can be very thin and conductive, but are expensive due to the high-temperature, crystal-growth process involved in their manufacture. On the contrary, XG Leaf® is manufactured with a low-temperature process more akin to that of paper manufacture that offers acceptable thermal conductivity, thickness, and mechanical strength but with a lower cost. In 2012, the XG Leaf® team focused on product design and verification, process design and verification, and product/process improvement and validation. The product achieved its performance goals and samples were produced for testing by several potential customers. In late 2013, we built a pilot production line for XG Leaf® and we expanded our production capacity for XG Leaf® during the latter part of 2014 and into early 2015. As with many of our other advanced products, we are developing new production processes to produce materials for new applications and cannot be sure exactly when we will move into commercial production.
The total available market for carbon-based heat spreaders as sold to the OEM or EMS companies with adhesive, PET and/or copper backing, and for selected portable applications, is estimated by Prismark Partners as $600 million in 2014 and growing to $900 million in 2018. The market can be further segmented by both the desired thickness and thermal conductivity of the heat spreaders as shown in the table below (Source: Prismark Partners LLC, “Thin Carbon-Based Heat Spreaders”, August 2014). XG Leaf is currently ideally suited to meet the needs for >50% of the total available market and can be made in thicknesses ranging from approximately 20 microns to greater than 200 microns and with in-plane thermal conductivity up to 600 W/m.K. We also target improved performance allowing us to meet the remaining market needs.
|
| Thermal Conductivity |
| Area (Thousand m2) |
|
| ||
Thickness (µm) |
| (W/mK, in Plane) |
| 2014 |
| 2018 |
| CAAGR |
<25 |
| 1,600 – 1,900 |
| 200 |
| 500 |
| 26% |
25 – 40 |
| 1,300 – 1,600 |
| 800 |
| 1,500 |
| 17% |
25 – 40 |
| 900 – 1,200 |
| 400 |
| 800 |
| 19% |
25 – 40 |
| 400 – 800 |
| 1,300 |
| 3,000 |
| 23% |
50 – 70 |
| 700 – 1,300 |
| 300 |
| 500 |
| 14% |
50 – 70 |
| 400 – 600 |
| 400 |
| 700 |
| 15% |
100 – 150 |
| 600 – 800 |
| 50 |
| 60 |
| 5% |
100 – 150 |
| 300 – 500 |
| 150 |
| 200 |
| 7% |
Total |
|
|
| 3,600 |
| 7,260 |
| 19% |
Markets for Ink and Coating Products
Our xGnP® graphene nanoplatelets can be made into coatings or inks for thermal, electrical, anti-corrosion, or lubricant applications. Various types of processing and application methods can be used, depending on customer systems and requirements. We have developed several products and also work with customers to provide formulations of inks or coatings to achieve targeted electrical and thermal properties.
xGnP® is potentially suited for formulating into ink-based products for deposition by flexo, gravure and screen printing processes. These inks show intermediate performance when compared to silver-based inks, the standard product used today for electrically-conductive ink requirements. Silver-based inks have high conductivity but are relatively expensive due to their use of silver. Carbon-based inks are lower cost, but have poor conductive performance and are well suited for few applications. xGnP® inks, although still a few orders of magnitude lower in conductivity than silver-based inks, are potentially less expensive to produce and have cost/performance benefits allowing them to be used in many applications. ID Tech, in a recently published report (Conductive Ink Markets 2014-2024), estimated the 2014 conductive ink market at just over $1.5 billion. In the short term, our xGnP®-based inks are designed for applications where silver-based inks exceed the cost and performance targets required for specific applications.
Other Markets
We may also choose to participate in a number of other broad market segments which we believe are large and growing and where the performance of our products may find a good fit with existing and emerging market needs. These market segments are variously referred to as “advanced materials” or “composites” or “nanotechnology” markets. Because we may compete with and replace existing materials or may create new opportunities, it is difficult to accurately quantify specific market segments. However, we believe that the broad range of existing materials and existing applications might account for hundreds of millions of dollars in annual sales. We may also participate in an emerging market segment called “nano-composites” that consists of
56
new materials specifically designed to incorporate “nano” sized additives. Other product applications that have been identified for graphene and that we may pursue include, but are not limited to:
• The production of electrically-conductive composites for use in applications where electro-static dissipation (ESD), radio-frequency interference (RFI), and electro-magnetic interference (EMI) are important.
• The production of thermally-conductive composites for use in applications where heat transference is important or for applications where it is important that thermal expansion and contraction be limited.
• The production of composites that feature improved barrier performance (impermeability) to liquids or gases like engine fuels.
• The production of elastomeric-based (rubber) materials that are longer wearing and have a higher surface toughness than some alternative materials currently used.
• The production of materials designed to improve the performance of electrodes in EDLC capacitors (supercapacitors).
Multiple Applications and Products within Markets
It is also worth noting that many of our markets contain multiple applications for our products. For example, automotive markets may offer potential applications for multiple uses of our coatings technologies within one automobile, along with potential applications for our bulk material, battery anode materials, and composites made with our products. As another example, the following diagram shows possible uses for our materials within an electronic tablet.

Distribution Channels
Our business model includes the production of bulk materials and value-added products based on our proprietary production processes and the sale of these materials directly to customers around the world. We generate sales through our own sales and scientific personnel and also maintain a network of independent representatives who may assist in sales to smaller customers throughout North America. Additionally, we maintain a network of distributors and sales agents for handling customers in overseas locations. Currently, we have signed Sales Agency Agreements with companies in South Korea, Japan, Taiwan, China, and Italy and will add other distributors on a worldwide basis as needed.
We pursue a national and international customer base. We plan to develop a broader sales and marketing organization and also plan to extend production of our products to overseas locations as appropriate in the next several years. We plan to recruit and train a sales organization located at headquarters as well as locations around the world. We also plan to partner with other companies in the United States and other countries in order to further leverage our time-to-market advantage.
57
Manufacturing Capacity and Concentration of Business
Our current manufacturing capacity depends on the specific grade and type of product that we are manufacturing but, given an average mix of orders, would generally be about 100 tons of annual production on a three-shift basis. We have plans to increase this capacity as needed, by adding additional equipment, but our existing facilities have capacity limits simply because of available space. If we are successful at building our revenues beyond our existing facilities’ capacity, we will consider adding additional capacity in the U.S. or overseas by adding new facilities as necessary.
Because of the nature of the applications that our customers are developing, we anticipate that we may see a few customers that account for the majority of our revenues. For example, in 2014 and 2013 we had one customer whose revenue was 37% and 77%, respectively, of total revenues. In 2014 we also had another customer that represented 28% of total revenues. It is very conceivable that, as we go forward, we will continue to have only a few customers that order enough materials to effectively utilize all of our manufacturing capacity. This would, of course, raise certain risks if those customers were to leave us in the future. Likewise, it is possible that we may find opportunities to serve these large customers with dedicated cooperative manufacturing facilities located near end-use points, or otherwise find ways to accommodate concentration risks.
Sources and Availability of Raw Materials
The raw materials we use for our products consist primarily of graphite and standard industrial chemicals that are readily available. Although we purchase raw materials used in the manufacturing of our products from a small number of sources in order to maintain the stability of the quality of the raw materials, we have the ability to purchase from numerous sources. We believe that all necessary raw materials for our products are readily available and will continue to be so in the foreseeable future. We have never had, nor do we anticipate experiencing, any shortages of such materials.
Strategic Investors
In addition to our sales of products, we also emphasize the importance of strategic investors, for several reasons. We see an opportunity to partner with larger companies as a way to accelerate our customer reach — particularly in parts of the world where we have not yet established a physical presence. In addition, many of our prospective customers are global companies with multiple manufacturing facilities in various parts of the world. These companies often seek local suppliers. In addition, these companies often seek multiple suppliers to avoid the risk of single-sourced supply. Thus, an important part of our business strategy has been to seek to develop relationships with other companies that we see as complementary partners that can help us develop our business for mutual benefit. These strategic partners typically have much greater resources than we do and represent manufacturing or technology partners or both.
During the past several years, XGS has formed relationships with a number of larger companies that it considers to be “strategic partners” where both companies can mutually benefit from combining their resources in specific ways. We foresee a likelihood of continuing partnerships with large companies as we focus on local production in non-U.S locations. The following is a short list of transactions with strategic partners or licensees:
• Hanwha Chemical. In December 2010, we sold 150,000 shares of Common Stock at $20.00 per share to Hanwha Chemical Company (“Hanwha Chemical”), a global company with headquarters in South Korea. At the same time, we signed a Mutual Collaboration Agreement that gave Hanwha Chemical a non-exclusive right to distribute XGS products in East and Southeast Asia.
• POSCO. In June 2011, the Company sold 200,000 shares of Common Stock at $20.00 per share to POSCO, a global corporation with headquarters in Pohang, South Korea. At the same time, the Company granted to POSCO a non-exclusive license to certain XGS production technology for use in Southeast Asia region defined therein, as well as a non-exclusive license to sell XGS products on a worldwide basis. POSCO currently maintains a seat on the XGS Board as a result of its investment in the Company. In March, 2014, POSCO purchased an additional 100,000 shares of Series A convertible preferred stock at $12 per share as part of a preemptive rights share offering. The license to POSCO terminates on December 31, 2024 unless terminated earlier by mutual consent.
• Cabot Corporation. In November 2011 (as amended in January 2014), the Company granted a non-exclusive, royalty-bearing, worldwide license to certain of its production technology to Cabot Corporation (“Cabot”), a global corporation with headquarters in Boston, Massachusetts. Cabot is a customer of XGS and is also a development partner and licensee. We are not currently receiving royalty payments from Cabot.
58
• Samsung. On January 15, 2014 the Company sold $3,000,000 of notes to Samsung Ventures. In conjunction with the sale of these notes, the Company also entered into a Joint Development Agreement with Samsung SDI on April 8, 2014, under which we will jointly develop anode materials for future versions of lithium-ion batteries to be manufactured by Samsung SDI. Samsung Ventures currently maintains the right to appoint a seat on the XGS Board. The Joint Development Agreement continued through June 2015. It is no longer active.
Samsung Ventures purchased secured convertible notes on terms identical to Aspen Advanced Opportunity Fund, LP’s (“AAOF”) terms with XGS. Such notes are convertible into Series A convertible preferred stock which, in turn, is convertible into Common Stock at the lower of: (a) $12.00 per share, or (b) 80% of the price at which XGS sells any equity or equity-linked securities in the future. Samsung Ventures also received 40% warrant coverage on their investment with such warrants vesting according to the amount of cash payments made to XGS in connection with the joint development agreement and other commercial arrangements; the warrants have a four year term. In contrast, AAOF received 100% warrant coverage on its investment with up front vesting on any amounts funded, and the warrants have a ten year term.
Intellectual Property
We believe that our intellectual property (IP) is an important asset. Our strategy is to keep our production processes as legally defined and protected trade secrets rather than to patent them. We believe that by patenting our processes, we would simply be teaching others how to produce materials like ours. In addition, it would be difficult or impossible to detect infringement. Therefore, our basic intercalation and exfoliation processes are not patented. We have, however, patented some of the equipment used in these processes.
Our intellectual property consists of four main types of assets:
1. Employee knowledge and skills
2. Corporate “trade-secrets” and processing know-how
3. Exclusive license from Michigan State University on certain inventions
4. Patents and trademarks owned by the Company
In January 2014 in connection with the Samsung Ventures investment, we transferred all of our intellectual property into a newly created, wholly owned subsidiary of the Company called XG Sciences IP, LLC, a Michigan limited liability company.
Employee Knowledge and Skills
Our founding stockholders include three research scientists and one experienced entrepreneur. Our Chief Scientist, Dr. Lawrence Drzal, is a University Distinguished Professor at Michigan State University and has a worldwide reputation in the field of materials science. Dr. Drzal has been engaged in research in our product areas for over eleven years. Dr. Hiroyuki Fukushima, another founding stockholder, has been engaged in research in this area for over ten years and is a co-inventor of one of our proprietary manufacturing processes. Dr. Inhwan Do, also a founding stockholder, has been engaged in research in this area for over ten years and is a co-inventor of one of our proprietary production processes as well as a number of application inventions. Mr. Michael Knox, the Company’s Senior Vice President, is a successful entrepreneur with over forty years of experience in global corporate management as well as business development. He previously founded or co-founded five other successful businesses, four of which were sold in private transactions or went public.
Beginning in mid-2011, we started adding research scientists to our staff. As these scientists gather experience working with our materials, the cumulative research experience becomes increasingly valuable. We believe the cumulative knowledge of our employees is an important aspect of our intellectual property. In mid-2012, we hired Dr. Liya Wang as Vice President of Research & Development. Having served in a number of senior scientific positions for global battery makers, Dr. Wang significantly strengthened our experience base in the energy storage sector.
In January 2014, we hired Dr. Philip L. Rose as CEO. Prior to joining XGS, Dr. Rose was President of SAFC Hitech, a $100 million division of Sigma-Aldrich Corporation that makes precursors and performance materials for the LED, energy and
59
display, and semiconductor markets. Before moving to Sigma Aldrich, Dr. Rose spent almost 20 years with Rohm and Haas in various leadership positions in the semiconductor and flat panel display industries.
Corporate Trade-Secrets and Processing Know-How
The production of xGnP® brand graphene nanoplatelets involves significant processing know-how that we maintain as secret internal knowledge, commonly referred to as “trade-secrets”. These trade secrets have been internally documented and safeguarded through the use of limited documentation, stringent employee confidentiality agreements, and strict disclosure policies. We have chosen not to pursue patent protection for some of our processing know-how because of the public disclosure involved. Nevertheless, we believe that this processing know-how has a significant value and we believe that a prospective competitor would be forced to spend millions of dollars to duplicate the processing know-how necessary to produce products of a similar quality to our products. In part, this reasoning has been validated by two large global companies: POSCO and Cabot. Each of these companies independently researched worldwide technologies for production of graphene and each of them chose to license XG Sciences’ production processes. These two companies have paid millions of dollars for access to our trade secrets and the experience and knowledge of our personnel. In addition, we signed in April, 2014 an agreement with Samsung SDI, the world’s largest producer of lithium ion batteries for handheld devices, outlining a joint development program aimed at engineering our Silicon Graphene material into a next generation lithium ion battery platform.
Michigan State University License
We have acquired an exclusive license for a number of inventions from Michigan State University. These rights are embodied in a Technology Licensing Agreement between XGS and Michigan State University dated July 27, 2007 and amended on May 24, 2010 and May 27, 2011, which owns the rights to all intellectual property resulting from research performed at the University. This Agreement gives us the exclusive worldwide rights to the subject intellectual property in its field. In exchange for the rights to the technology covered by this Agreement, we paid MSU an initial fee and agreed to pay ongoing royalties on the basis of material sold by us in future years. Under a previous version of the Licensing Agreement, we also awarded a small amount of common stock to MSU.
The intellectual property covered by the Agreement is in various stages of development and legal protection. Because there is an active research program underway in this area, we also anticipate that future inventions will be made that will also fall under the terms of our license agreement with MSU. In some limited cases, depending on the source of research funds, other parties may have limited rights to use the inventions covered by these agreements. This is typical in the case of research funded by U.S. government agencies and is sometimes the case when research has been funded or partially funded by private corporations.
As of November 1, 2015 the following technologies were included in the MSU License Agreements with XGS and were being actively prosecuted at the U.S. Patent Office:
• U.S. Patent Application No. 12/587,645. “Electrically Conductive, Optically Transparent Films of Exfoliated Graphite NanoParticles & Methods of Making the Same”
• U.S. Patent Application No. 13/200,764. “Method of Preparing Metal Nanoparticles”
In addition, one licensed MSU invention has recently been granted a US Patent:
• U.S. Patent No. 8,834,959. “Method for the Preparation of Doped Single Graphene Sheets.”
In general, the current license arrangement with MSU provides us with an exclusive option to relevant MSU inventions, as documented by MSU invention disclosures. We, in turn, have an obligation to patent these disclosures in a timely manner. Disclosures that we do not pursue or that are not allowed as patentable by the USPTO are eventually considered as abandoned and drop out of the licensed technology package. It is contemplated that periodic amendments to the MSU — XGS License Agreement will update the list of existing technologies covered by the license.
In general, the MSU licensed technology relates to end-use applications for graphene nanoplatelets. Of particular interest are the discoveries and claims related to the use of graphene nanoplatelets in electrodes for lithium-ion batteries and supercapacitors.
60
Our Patent Filings
In addition to the patent applications related to MSU technology discussed above, we also file for patents on some of our own inventions. As of November 1, 2015 the following patent applications were being managed by us and our patent attorneys:
• U.S. Patent Application No. 13/435,260. “Mechanical Exfoliation Apparatus”, allowed but not yet granted
• U.S. Patent Application No. 13/474,860. “Process of Dry Milling Particulate Materials”
• U.S. Patent Application No. 13/686,961. “Single Mode Microwave Device for Producing Exfoliated Graphite”
• U.S. Patent Application No. 61/786,735. “Graphene Carbon Compositions”
• U.S. Patent Application No. 61/786,745. “Electrodes for Capacitors from Mixed Carbon Compositions”
• U.S. Patent Application No. 13/858,149. “xGnP as conductive filler for resistor materials
• U.S. Patent Application No. 61/879,225. “Flexible Resin-Free Composites Containing Graphite & Fillers
• U.S. Patent Application No. 14/079,057. “Silicon-Graphene Nanocomposites for Electrochemical Applications”
• U.S. Patent Application No. 62/060,319. “LiF-Embedded SiG Powder for Lithium-Ion Battery.”
• U.S. Patent Application No. 62/482,797. “Thermal Interface Materials using Graphene Coated Fillers”
• U. S. Patent Application No. 62/244,927. “Heat Exchanger Elements and Devices”
In addition, some of our early patent filings have been granted:
• US Patent 8,715,720: “A Cloud Mixer and Method of Minimizing Agglomeration of Particles.”
• US Patent 9,061,259 B2 “Cloud Mixer and Method of Minimizing Agglomeration of Particles.”
Global Patent Filings
For each patent application filed in the US, we make a determination on the nature and value of the patent. For many of the applications filed in the US, additional filings are made in other countries such as the European Union, Japan, South Korea, China, Taiwan or other applicable countries. These filings and analyses are made on a case-by-case basis. Typically, patents that are defensive in nature are not filed abroad, while those that are protective of active XGS products or application areas are filed in relevant countries abroad.
Trademarks
We have obtained a registered trademark on the following:
• “xGnP®,” which is the brand name by which we designate our graphene nanoplatelets.
• The corporate logo “XG Sciences” (design logo plus words)
• The tag line “The Material Difference” which is used in conjunction with the corporate logo
• The product designation “XG Leaf®”, which is the brand name for our family of sheet products.
• The product designation “XG SiGTM”, which is the brand name for our battery anode materials.
We also recently applied for protection for the product designation “XG TIM™”, which is the brand name for our thermal interface materials and which application is pending.
61
Protection of IP
We protect our IP through various agreements. Employee agreements, non-disclosure agreements, and confidentiality agreements all include provisions to prevent our confidential information from disclosure outside very tightly controlled circumstances. A sign-in sheet non-disclosure agreement is maintained at the entrance to each facility. Access to the lab and production facility is controlled on a need to know basis for non-XGS personnel.
Research and Development
During 2014 and 2013 we spent $1,449,478 and $1,269,456 on research and development activities, respectively. Of these amounts, $660,533 and $174,535 were funded by government agencies in the form of grants, recorded as revenues during 2014 and 2013, respectively. During the nine months ended September 30, 2015 and September 30, 2014 we spent $1,135,326 and $988,149 on research and development activities, respectively. Of these amounts, $299,879 and $551,627 were funded by government agencies in the form of grants during 2014 and 2013, respectively. With the exception of these government grants, none of our research and development activities are funded directly by customers.
Competition
We manufacture xGnP® graphene nanoplatelets and various value-added products made therefrom. In one sense, this is a new type of material that requires application development before it can be incorporated into customer products. In another sense, our material is similar to other carbon products in its chemistry and molecular composition. Therefore, there are many instances when our materials might be used interchangeably or in combination with other types of carbon materials like carbon nanotubes, carbon fibers, or carbon black. Because these other forms of carbon materials are much older, with markets that are significantly more developed, there are many companies that are much larger and more well-established than we are and that might compete in the same markets we address.
In addition, there are many different materials that may sometimes be used as full or partial substitutes for our materials. A large number of companies offer a wide variety of materials that may compete with us. Almost all of these competitors that manufacture alternative materials are significantly larger and better financed than we are. In addition to alternative materials, there are a number of emerging companies in the United States that have announced plans to offer graphene platelet materials purported to be similar to ours. In addition, several new companies in Europe and Asia have announced the availability of materials purported to be similar to ours. Many of these companies currently offer materials only in limited quantities, but we anticipate that it will face competition from a variety of companies in most of the markets in which we compete.
Graphene has excited great interest in the scientific community and there are hundreds of university groups and private corporations that are interested in this material. In addition to us, many other small companies are beginning to appear that have announced products based on graphene or graphene nanoplatelets in some form, and we expect this trend to continue. We believe that we are a first mover in this market and believe that our production processes are the lowest cost, but we expect competition from various sources in the future. Given the huge projected market sizes, we expect that several companies will share these markets, perhaps focused on specific niches.
Our strategy is to compete with other start-ups on the basis of our business model, our manufacturing techniques, our management team, our scientific credibility, and our strategic partners. Recognizing that the worldwide market for products and materials similar to ours is developing very rapidly and will be quite large, we intend to compete vigorously on a worldwide basis by:
• Implementing strategic partnerships with customers and other partners in many different geographic localities as well as specialized markets.
• Focusing our resources on remaining a low-cost supplier of high-quality materials with sufficient production capacity to meet the needs of large, global customers.
• Developing “value-added” products like XG Leaf®, XG SiG® battery anode materials, inks and coatings, and similar specialized products for specific markets.
• Investing in intellectual property, especially in areas covering novel formulations of our products and use of our products in end-use applications.
62
Our Competitive Strengths
We believe that we are a world leader in the emerging global market for graphene nanoplatelets. The following competitive strengths distinguish us in our industry:
The strength of our intellectual property. Because of our focus on manufacturing process development, we believe we have one of the world’s strongest internal knowledge bases in graphene nanoplatelet manufacturing, with most of our proprietary knowledge maintained as trade secrets to avoid the disclosures required by patenting. The 14 patents/applications we are currently managing add value by protecting specific equipment, or high-value end-user product applications. The fact that two global companies have evaluated and licensed our production technology provides independent evidence of our technology’s effectiveness.
The breadth of our product offering. As far as we are aware, we have the broadest product offering in our industry. In addition to offering three standard particle sizes in three different grades of bulk materials, we offer four different grades of XG Leaf® in multiple thicknesses, two different grades of silicon-graphene composite materials, three standard ink formulations, and optional custom dispersions and formulations of our bulk materials.
The low-cost nature of our manufacturing processes. As far as we are aware, our manufacturing processes have the potential to be the lowest-cost approaches to the manufacture of graphene nanoplatelets (subject to economies of scale) based on our internal modelling of competitive processes as well as our analysis of alternative technologies.
Our corporate and strategic partners. Three global corporations have made the following investments in XGS, giving us a significant global reach as well as the ability to leverage the assets of our partners:
• Samsung Ventures — $3.0 Million (January 2014)
• POSCO — $5.2 Million (June 2011 and March 2014)
• Hanwha Chemical — $3 Million (December 2010)
Our licensees will accelerate our entry into large markets. Cabot Corporation, the largest U.S.-based manufacturer of carbon particles, and POSCO, one of the world’s largest steel producers, have licensed parts of our production technology. We believe these licensees will help us distribute our products and value-added products made with our xGnP® nanoplatelets more rapidly than we could do on our own.
The number of development partners that are working with our materials. As of November 23, 2015, we had supplied materials to 202 universities or government laboratories in 40 different countries around the world. A recent search of the U.S. patent database revealed 327 citations of XG Sciences in patents filed by other organizations. These other organizations include Goodrich Corporation, PPG Industries, Exxonmobil Research & Engineering, Toray, Solvay, Honda, Eastman Kodak, Baker Hughes, GM, Rohm and Haas and Sekisui Chemical.
The number of customers purchasing and working with our materials. As of August 31, 2014, we have supplied materials to more than 600 commercial companies around the world (in addition to universities and research laboratories) who are assessing their performance and potentially designing them into products. We have more than 20 active development relationships where we are working with end-use customers to design products for commercial use. We expect that these relationships will continue to expand.
Our know-how and ability to tailor our products for use in multiple applications. Many of our products and product-development activities target use of our xGnP® graphene nanoplatelets in various matrices to form composite products that are then used by our customers. We have extensive knowledge of how to tailor our products to deliver performance as composite products in various applications and we also have knowledge of how to tailor other components of a composite to adjust the performance of the composite for use in various applications.
We believe that the combination of these factors provides objective evidence that XGS is a world leader in the emerging global graphene particle industry. Other independent observers have agreed with this assessment. For example, Lux Research, in a July 2015 release listed XGS as a leading worldwide player in its industry. Further, Lux analysts wrote: “XG’s march of strategic relationship announcements — Hanwha Chemical in December 2010, POSCO in June 2011, and Cabot in November 2011
63
— arguably give it the strongest partnership portfolio in the space, and its recent expansion (see the May 7, 2012 LRMJ) makes it one of the low cost and capacity leaders.”
Legal Proceedings
To our knowledge, neither we nor any of our officers or directors is a party to any material legal proceeding or litigation and such persons know of no material legal proceeding or contemplated or threatened litigation. There are no judgments against us or our officers or directors. None of our officers or directors has been convicted of a felony or misdemeanor relating to securities or performance in corporate office.
Governmental Regulation and Environmental Compliance
We believe we are in material compliance with all applicable governmental regulations, and that the cost and effect of compliance with environmental laws is not material. Most new chemical substances or materials sold in the U.S or in many other countries require regulation by government authorities. In the U.S., we have specifically reviewed our grades H, M and C materials and the associated production processes with the U.S. Environmental Protection Agency and have received written confirmation from that agency as to the appropriate classification of our materials and of our ability to offer these materials on a commercial basis within the U.S. In most other countries, there are no specific regulations that require additional regulation but some countries do have registration requirements with which we comply to the best our ability.
Our Production Facilities and Employees
In March of 2012, we took possession of a larger production facility at 3101 Grand Oak Drive, Lansing, Michigan, 48911 under terms of a long-term lease and moved our headquarters to this new location. We and Dart Container of Michigan LLC, a Michigan limited liability company (“Dart”) entered into a lease pursuant to which Dart, as landlord, agreed to lease such property for ten years at an annual base rent for each of the ten years in twelve equal monthly instalments each year. Such annual base rent for years one through ten were $189,996, $193,800, $197,676, $201,636, $205,668, $209,772, $213,972, $218,256, $222,612, and $227,064, respectively. In addition to base rent, we agreed to pay additional rent consisting of certain taxes, insurance costs, assessments and common area maintenance charges. Furthermore, we agreed to provide Dart with a letter of credit in the amount of $189,996, the terms of which are more specifically detailed in the lease. Additionally, under the terms of the lease, the parties agreed that Dart would pay costs up to $640,000 in connection with specified improvements (described in an exhibit to the lease) to the leased premises and that any costs over such amount would be borne by us.
Effective as of August 15, 2011, we and Dart amended certain provisions whereby the parties agreed that taxes payable shall be paid directly to the “appropriate taxing authority” instead of to Dart; that we shall provide copies of the tax or assessment being paid along with a copy of the applicable check; and that we shall pay our respective share of Taxes (as such term is defined in the lease) either for the entire year or its pro rata share of any partial year in which we occupied the leased premises. Furthermore, the parties amended the obligation of the landlord to deliver estimates of certain costs payable by us. The revision removed the requirement imposed on Dart to provide a Tax Estimate (as such term is defined in the lease) to us. The lease was also amended to provide for a default in the event the “appropriate taxing authority fails to receive any payment of Taxes within ten days after written notice from either landlord or the taxing authority that such payment is past due.”
The lease was further amended by us and Dart effective as of November 16, 2012 to include additional space as part of the leased premises from January 1, 2013 through December 31, 2017, and as such, we agreed to an increase to base rent by $6,667, $6,871, $7,074, $7,288 and $7,503 per month for the periods ranging from January 1, 2013 through December 31, 2013, January 1, 2014 through December 31, 2014, January 1, 2015 through December 31, 2015, January 1, 2016 through December 31, 2016, and January 1, 2017 through December 31, 2017, respectively. The parties further agreed that common cost estimates for such additional space was $2,147 per month for the calendar year 2013, said cost to be adjusted for each subsequent year. Additionally, we now have the ability to terminate the lease of the above-mentioned additional space by providing six months’ notice. A second amendment addresses utility payments in connection with additional rental space.
We also operate a separate production facility in leased manufacturing space, which is used for the production of certain specialty materials. On January 3, 2008, we entered into a lease agreement with Quality Dairy Company, as landlord, pursuant to which the landlord agreed to lease to us such 6,600 square feet of a building commonly referred to as “Building C” and an area approximately 16’ X 80’ of the parking lot adjacent to the aforementioned area located at 2100 S. Washington Ave., Lansing, MI 48910. We were also granted the non-exclusive right to use common facilities. The term of the lease was for six months, commencing on January 15, 2008 and ending on July 31, 2008. We agreed to pay base rent in monthly instalments
64
in the amount of $12,798 for the term of the lease, and $1,969 per month thereafter. In addition to base rent, we agreed to pay certain utilities, janitorial services, and personal property taxes as they become due. Subsequently, we have extended this lease and now maintain our presence in this facility on a month-to-month basis.
We believe our manufacturing facilities will be sufficient to meet demands for the majority of our bulk materials for a number of years, with suitable additions of capital equipment as warranted. However, additional manufacturing capabilities for certain value-added products and certain bulk materials remain to be developed and may require the acquisition of additional facilities. In particular, the production processes for XG Leaf® and our silicon-graphene electrode materials have not yet been scaled and will require additional capital to meet expected customer demand.
Our Employees
As of November 16, 2015, we had 23 full-time employees. Employees include the following five senior managers that report to the CEO: a Vice President of Operations, a Vice President of Energy Markets, a Vice President of Research & Development, a Controller and a Senior Vice President. The Company employs a total of 6 full-time scientists and technicians in its R&D group, including the Vice President of Research & Development.
Corporate Information
XG Sciences, Inc. was incorporated on May 23, 2006 in the State of Michigan and is organized as a “C” corporation under the applicable laws of the United States and State of Michigan. We do not currently have any affiliated companies or joint venture partners, and we have one wholly-owned subsidiary called XG Sciences IP, LLC. This subsidiary was created in 2014 for the purpose of holding our intellectual property. Our headquarters and principal executive offices are located at 3101 Grand Oak Drive, Lansing, Michigan, 48911 and our telephone number is (517) 703-1110.
Our website address ishttp://www.xgsciences.com, although the information contained in, or that can be accessed through, our website is not part of this prospectus. You may also contact Dr. Philip L. Rose, our Chief Executive Officer via email atp.rose@xgsciences.com.
65
The following table sets forth the name and age of the Company’s executive officers, Directors and significant employees as of the date of this prospectus. There are no family relationships among any of our executive officers or directors.
Name |
| Age |
| Position(s) |
Philip L. Rose |
| 53 |
| Chief Executive Officer, President, Treasurer & Director |
Michael R. Knox |
| 67 |
| Senior Vice President, Secretary & Director |
Arnold A. Allemang |
| 71 |
| Director, Chairman of the Executive Committee, and member of the Audit and Compensation Committees |
Ravi Shanker |
| 49 |
| Director and member of the Executive and Compensation Committees |
Steven C. Jones |
| 51 |
| Director, AAOF representative to the Board and member of the Audit, Executive and Compensation Committees |
Wonil Kim |
| 47 |
| Director, POSCO representative to the Board |
Michael Pachos |
| 37 |
| Director, Samsung representative on the Board and Audit Committee member |
Scott Murray |
| 60 |
| Vice President, Operations |
Robert Privette |
| 53 |
| Vice President of Energy Markets |
Liya Wang |
| 56 |
| Vice President of Research & Development |
In addition to the persons listed above, Mr. David Pendell, a principal of AAOF, has observer rights to all meetings of the Board of Directors and is an advisor to the Company.
Biographies of Officers and Directors
The following short biographies of our officers and Directors as of the date of this prospectus:
Philip L. Rose, Ph.D.
Dr. Rose joined the Company in January 2014 and currently serves as our Chief Executive Officer, President, Treasurer and is a Director of the Company. Dr. Rose has extensive international business management experience in the electronic and specialty materials markets. Prior to joining XG Sciences, he spent 4 years as President of SAFC Hitech, a $100 million division of Sigma-Aldrich that makes precursors and performance materials for the LED, energy and display, and semiconductor markets. He also served concurrently for a period of time as CEO of Soulbrain-Sigma Aldrich based in South Korea and as an independent Director for Pixtronix, a company based in the Boston area. Before joining Sigma Aldrich, Dr. Rose spent almost 20 years with Rohm and Haas in various leadership positions in the semiconductor and flat panel display industries that include general management, mergers and acquisitions, business development and marketing. Dr. Rose has 7 years of experience living in Japan and South Korea and has travelled extensively in Asia over the past 15 years. Dr. Rose earned his Ph.D. in Physical Chemistry from Duke University and holds a B.S. in Chemistry from the University of Southern California and a certificate in Business Management from The Wharton School at the University of Pennsylvania. In light of the aforementioned experience qualifications, attributes and skills, we believe Dr. Rose is qualified to serve as a director.
Michael R. Knox
Mr. Knox is a Company co-founder and currently serves as the Senior Vice President and Secretary, and is a Director of the Company. He served as Chief Executive Officer, President, Treasurer and Secretary of XGS from May 2006 to January 2014. Prior to founding XG Sciences, Mr. Knox was involved in a number of different businesses as an owner or an officer. From 1998 to 2003, he was the President and CEO of Polyphasic, Inc., a company that he founded to develop software and technology related to the World Wide Web. In 2003, Polyphasic merged into TravWell, Inc., another company that Mr. Knox founded, and of which he served as CEO. TravWell was sold to a consortium of private investors in 2005 and he left the company shortly thereafter. From 1989 to 1995, Mr. Knox served as Chief Executive Officer and Chief Financial Officer of Harmony Brook, Inc., a business that he co-founded in 1989 to develop and manufacture water purification products. While in that capacity, he directed all aspects of business development during a period in which the Company grew from one employee to almost 100, expanded operations into forty US states and Mexico, and achieved annualized operating revenues of over $7 million. The Culligan Corporation purchased Harmony Brook in 1997. From 1985 to 1989, Mr. Knox was president of Knox & Associates, Inc., a management-consulting firm. From 1976 to 1985, Mr. Knox worked in various general management and financial positions for Donaldson Company, Inc., a worldwide leader in the air and liquid filtration industry. Mr. Knox has a BA in
66
Economics and an MBA in finance from the University of Minnesota. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Knox is qualified to serve as a director.
Arnold A. Allemang
Mr. Allemang has served as a Director of XG Sciences since March 2010. Mr. Allemang is also a member of the Board of Directors of The Dow Chemical Company, headquartered in Midland, Michigan. Mr. Allemang joined Dow in 1965 in Freeport, Texas. After five years in the Solvents Development Lab, he transferred to Stade, Germany. In 1972, he returned to the United States for various technical and managerial assignments. Mr. Allemang was named unit manager for Freeport’s Chlorinated Ethanes in 1981, and two years later assumed the same role for Light Hydrocarbons and Acetylene. He transferred to Terneuzen, The Netherlands, in 1984 to become production manager for Light Hydrocarbons I, and in 1986 became responsible for process control and engineering functions. In 1988, he returned to Freeport to manage the site’s hydrocarbons production, and moved to Midland in 1989 as Director of Technology Centers. Mr. Allemang was named Manufacturing general manager for Dow Benelux in 1992, and in early 1993 was named regional vice president, Manufacturing and Administration, Dow Benelux. He was named vice president, Manufacturing Operations Dow Europe in late 1993. In August 1995, Mr. Allemang was named vice president, Operations, which included global manufacturing and engineering activities. He then became executive vice president of The Dow Chemical Company in 2000, and was named senior advisor in 2004. Mr. Allemang was elected to the Dow Board of Directors in July 1996. In March 2008, he retired as a Dow employee and remains on the Board as a non-employee Director. Mr. Allemang received a bachelor’s degree in chemistry from Sam Houston State University in Huntsville, Texas. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Allemang is qualified to serve as a director.
Ravi Shanker, Ph.D.
Dr. Shanker has served as a Director of XG Sciences since March 2014. Since 1991, Dr. Shanker has been employed by the Dow Chemical Company (“Dow”) in various capacities. Dr. Shanker is currently the General Manager of the Lightweighting Platform, a position he has held since March 2015. From March 2013 until March 2015, Dr. Shanker was the Global Business Advisor at Dow. In this role, Dr. Shanker provides strategic insight into the portfolio of businesses that include Dow Epoxy Intermediates, Polymers and Systems. Previous to this role, Ravi served as the president and CEO of Dow Kokam. He led the team that created the Dow Kokam joint venture, and has many years of experience building, growing and running businesses with the Dow Chemical Company. Dr. Shanker first joined Dow in 1991 and has grown Dow’s business via new product and new business development. Starting in the research department, he focused on developing new manufacturing processes. Dr. Shanker also supported the office of the CEO in strategy development for near- and long-term business opportunities in emerging geographies. He was then appointed Global Business Director of Specialty Plastics in the Performance Products Portfolio. Before assuming his role with Dow Kokam, Dr. Shanker served as Corporate Director of Ventures and Business Development, where he incubated a portfolio of innovative business opportunities for Dow, driving many to commercialization. Ravi holds a Ph.D. in Mechanical Engineering from the University of Delaware and B. Tech in Mechanical Engineering from B.I.T. Sindri, in India. He also has an MBA from the University of Houston. In light of the aforementioned experience qualifications, attributes and skills, we believe Dr. Shanker is qualified to serve as a director.
Steven C. Jones
Mr. Jones has served as a Director of XG Sciences since March 2014. Mr. Jones has been the Executive Vice President of Finance and Chief Compliance Officer of NeoGenomics, Inc. (NASDAQ: NEO) (“NEO”) since November 30, 2009 and February 7, 2013, respectively, and serves as a Director. He previously served as the Chief Financial Officer for NEO from October 2003 until November 30, 2009. In December 2014, he was appointed to serve as a Director by the Aspen Advanced Opportunity Fund, LP in connection with their investment in XGS. Mr. Jones is the Chairman of Aspen Capital Group, LLC, a private equity firm headquartered in Naples, FL, which manages four private equity funds. Mr. Jones also serves as the President of Aspen Capital Advisors, LLC, which is Aspen Capital Group’s investment management subsidiary. Aspen believes its highest value is to be a “hands on” partner with the management teams of portfolio investments to help them achieve their growth objectives by bringing capital, strategic partners, customers, additional management and other business advice. Mr. Jones has substantial expertise in developing and financing emerging growth companies. In addition to his involvement with Aspen Capital Group, Mr. Jones serves on the Boards of T3 Communications, Inc., a business communications company, and SpinaDyne, Inc., a medical device company. Prior to his career in structured private equity, among other positions, Mr. Jones was a Vice President in the Telecommunications, Media and Technology Investment Banking Group of Merrill Lynch & Co in New York and was the chief executive officer or chief financial officer of various public and private companies. Mr. Jones has a BS degree in Computer
67
Engineering from the University of Michigan and an MBA from the Wharton School of the University of Pennsylvania. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Jones is qualified to serve as a director.
Wonil Kim
Mr. Wonil Kim has been the Director of the Advanced Materials Investment & Technology Group within South Korea’s POSCO, one of the largest multinational steel companies operating worldwide, since 2014. He has served the company for over 25 years and currently applies his diverse experiences into supervising and developing new businesses and technologies, expanding and broadening POSCO’s existing portfolio. Since 2010, Mr. Wonil Kim, primarily as a Director in Technology Strategy, has been involved in various industries within POSCO These include projects concerning carbon materials, anode/cathode technologies for rechargeable batteries, lithium and nickel extraction, magnesium, and other advanced materials. Prior to this role, from 2004 to 2009, he spent five years in Europe serving as a Senior Manager in Technology Scouting and Project Management and was also responsible for cultivating and producing partnerships across the European steel and automotive industry, academia, and consulting firms. From 2002 to 2004, Mr. Wonil Kim directed and streamlined processes concentrated on POSCO’s cold rolled steel, the business segment he had previously managed from 1990 to 2002. Mr. Wonil Kim has a Master of Science in the field of Mechanical Engineering from Lehigh University.
Michael Pachos
Michael Pachos is a Principal at Samsung Ventures. He joined Samsung in 2008, first working out of the Seoul headquarters, and then transferring to their Silicon Valley offices in 2012, and has been a director at Samsung since early 2015. He focuses on Consumer, Display and Energy related investments. Prior to joining Samsung Ventures, Michael worked in the Samsung Group’s Global Strategy Group (“GSG”). GSG was established under the Chairman’s Office to address strategic and operational issues at the CEO level of Group companies. Michael worked on projects involving M&A, convergence and digital media, and channel strategy. Before joining Samsung, Michael was a CPU architect at DEC, Compaq/HQ and Intel. Michael is also a co-founder of Global Velocity; an enterprise security related Hardware Company. Michael earned his MBA at the Wharton School and completed law related coursework. Michael holds his B.S. degree in Computer Engineering from Washington University in St. Louis.
Scott Murray
Scott Murray has extensive experience as a senior operations executive and is skilled in business management, product and process development, and strategic leadership of growth of businesses. These experiences ranged from small, privately owned companies to Fortune 500 Companies. Mr. Murray has been the Vice President of Operations of the Company since October 2007. Prior to his tenure with the Company, Mr. Murray was with Motor Wheel Corporation for 17 years having held positions in engineering, marketing, and plant management. He served in the positions of Chief Engineer and Director of Manufacturing prior to the leaving to form a new Company. Mr. Murray founded and was CEO of Uretech International Inc. from 1995 through 2004. He directed all process, product, marketing, and commercial activities for the Company. As a manufacturer of specialty chemicals and urethane products, Uretech International supplied products to many markets including the automotive, medical, and office furniture industries as well as a variety of industrial applications. Most recently, as Director of Development for McKechnie Automotive, Mr. Murray was responsible for the technical and market introduction of a new product line and the startup of a manufacturing operation for that product line in Kentucky. Mr. Murray is a graduate of Michigan Tech University, where he earned a degree in Metallurgical Engineering. As a member of the Society of Automotive Engineers and the American Iron and Steel Institute served on numerous task forces and technical committees for professional organizations in the automotive industry. He is a past Committee Chairman for the American Society of Non-Destructive Testing. Mr. Murray is a registered Professional Engineer and holds a Six Sigma Black Belt certification.
Robert Privette
Robert Privette is a technical business executive with more than 25 years of hands-on experience in technology development and commercialization related to fuel cells, batteries and other energy related devices. He has experience in managing large, complex programs with the US Department of Energy, DARPA, the US Army and the US Navy focusing on technology development, product design, new manufacturing process, and market-based product qualification testing with multiple corporate, university and laboratory partners. Mr. Privette was hired as the Vice President, Energy Markets for the Company in May 2011. In his current role as Vice President, Energy Markets, he is responsible for developing product markets and sales of graphene nanoplatelets and associated value-added XGS products for energy storage and conversion applications. Prior to joining XGS he was Director for Product Development at Energy Conversion Device (ECD) - Ovonic Fuel Cell Company. In this role he
68
led the development and execution of the company business plan and technology roadmap for a 5 kW fuel cell product for stationary and portable power applications. Before joining ECD in 2006, he worked for Degussa, a large German materials and specialty chemicals supplier where he led customer product qualification programs developed for North American customers focused on accelerating market introduction and commercialization of state-of-the-art fuel cell membrane electrode assemblies and fuel processing catalysts for automotive and stationary fuel cell power applications. During 1989 through 2000 he worked for Babcock & Wilcox where he led a US Department of Energy development program for a 50 kW on-board gasoline fuel processor prototype for fuel cell vehicles. He received his MS degree in Mechanical Engineering from Purdue University in 1986 and has completed MBA coursework.
Liya Wang, Ph.D.
Dr. Liya Wang leads the research and development activities of the Company for a variety of applications. Previously, Dr. Wang was Principal Scientific Director at CIC Energigune in Spain from 2010 to 2012. There he helped build a world-class new energy research center and led the development of advanced batteries and capacitors. Prior to arriving at CIC Energigune, Dr. Wang was Director of Emerging Technologies from 2006 to 2010 at A123 Systems, a global Li-ion battery manufacturer based in US. He led the development of new generations of Li-ion battery cathodes and the transition of technologies into production. From 2003 to 2006, Dr. Wang worked as R&D Director at Pacific Industrial Development Corp and coordinated the development of nano materials for catalysis and luminescence applications. From 1999 to 2003, he was Manager of Materials Development and Vice President at T/J technologies where he built and led a multi-million dollar battery research program. From 1994 to 1999, he worked at IMRA America as a Researcher on electrochemical capacitors and high power lithium ion batteries. Dr. Wang received a Bachelor’s and a Master’s degree in Metallurgy from Beijing University of Science and Technology in China, and a Master’s and a PhD degree in Materials Science from University of Michigan in US. He is a Guest Professor at University of Electronic Science and Technology in China and an Adjunct Associate Professor at University of Michigan in USA.
Shareholder Agreement
In conjunction with a financing with AAOF, we and our stockholders listed therein entered into a Shareholder Agreement on March 18, 2013 that contains a number of specific provisions pertaining to the Board of Directors as well as individual Directors. Among other things, the Shareholder Agreement provides for certain voting and nomination rights to be calculated on the basis of “Full Conversion” stock ownership (under which calculation, all convertible notes, preferred shares, or other convertible equity securities are deemed converted into common stock) as follows:
• So long as AAOF or its affiliates own 10% of more of the aggregate outstanding Shareholder Stock (as defined in the Shareholder Agreement):
– the size of the Board of Directors shall be set at seven individuals.
– one person nominated by AAOF shall be elected to the Board of Directors.
– two members of the Board of Directors, other than those nominated by AAOF, POSCO or Hanwha Chemical, shall qualify as independent Directors.
• So long as POSCO owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by POSCO shall be elected to the Board of Directors.
• So long as Hanwha Chemical owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by Hanwha Chemical shall be elected to the Board of Directors.
As of December 31, 2015, the ownership percentage of AAOF and POSCO, as calculated for purposes of Director voting, required the stockholders to vote for a Director nominated by AAOF and for a director nominated by POSCO. Mr. Jones is the AAOF representative to the Board and Mr. Oh is the POSCO representative to the Board pursuant to the terms of the Shareholder Agreement.
On January 15, 2014, AAOF, POSCO, SVIC, XGS II, Mike Knox and the Company entered into a letter agreement pursuant to which AAOF agreed to consent to, approve or otherwise sanction certain actions of the Company specified in the Shareholder Agreement so long as investors owning at least 66% of the voting securities on a fully diluted basis consent to the proposed Company action. These actions include if the Company (i) creates, obligates itself to create, authorizes or issues (by reclassification or otherwise) any new class or classes of securities which has a preference over or being on a parity with the Series A Preferred Stock in any respect; (ii) sells any material portion of the tangible or intangible assets of the Company that secures the indebtedness to AAOF; or (iii) incurs additional secured indebtedness.
69
The Shareholder Agreement grants preemptive rights to shareholders and holders of convertible notes who are parties to the Shareholder Agreement. Pursuant to the terms therein, such shareholders and noteholders have the right to purchase their pro rata share of all shareholder stock that the Company may, from time to time, propose to sell, issue, or exchange after the date of the Shareholder Agreement, other than certain excluded stock which includes stock granted to employees or as merger consideration. Each shareholder’s pro rata shares shall be equal to the ratio of (i) the aggregate number of shares of the Company’s common stock on a fully diluted basis, owned by the such shareholder at the time of the delivery of a preemptive rights notice to (ii) the aggregate number of shares of Company’s common stock on a fully diluted basis owned by all of the Company’s shareholders at the time of the delivery of a preemptive rights notice.
The Shareholder Agreement continues in full force and effect until the date of the closing of a public offering of our common stock pursuant to a registration statement filed with the SEC is declared effective in which the Company receives gross proceeds of at least $10,000,000, on which date it shall terminate in its entirety, unless the Shareholder Agreement is earlier terminated in accordance with its terms. Until such time as it is terminated, however, new shareholders will be required to execute an adoption agreement to the Shareholder Agreement. By adopting the Shareholder Agreement, such new shareholders will be bound by its terms, including the transfer restrictions provided therein.
Voting Agreement
In conjunction with a financing from Samsung Ventures on January 15, 2014, we and our stockholders entered into a voting agreement with Samsung Ventures whereby for so long as Samsung Ventures owns 10% or more of the aggregate outstanding common stock of XGS (assuming Full Conversion, but excluding any shares of common stock issuable upon the exercise of any warrants held by Samsung Ventures), each stockholder made a party thereto shall vote all of his, her or its voting securities from time to time in whatever manner is necessary to ensure that at each annual or special meeting of the stockholders at which an election of Directors is held or pursuant to any written consent of the stockholders, one person nominated by Samsung Ventures is elected to our Board as a Director.
The rights granted to Samsung Ventures under a voting agreement are effective until the first to occur of (1) the date on which the Minimum Ownership Requirement is no longer satisfied, (2) the date on which the Shareholder Agreement (which is described above) is terminated for any reason and (3) the date that Samsung Ventures agrees in writing to terminate the Agreement.
Code of Ethics
We have a Code of Ethics applicable to our principal executive, financial and accounting officers.
Board Committees
Audit Committee
Our Board has established an Audit Committee, which is composed of Steven C. Jones and Arnold A. Allemang and Michael Pachos. Pursuant to our Audit Committee charter, the Audit Committee was established for the primary purpose of assisting the Board in its oversight of the Company’s tax, legal regulatory and ethical compliance. The Audit Committee assists the Board in certain areas, including, but not limited to:
• Oversight and monitoring of the Company’s financial statements, accounting and financial reporting processes, financial statement audits, and other financial information provided by the Company to its shareholders and others;
• Overseeing the Company’s compliance with legal, regulatory, and public disclosure requirements;
• Oversight of the Company’s registered public accounting firm’s (“independent auditor”) qualifications and independence;
• Overseeing the performance of the Company’s independent auditor and the internal audit function;
• Overseeing the Company’s systems of disclosure controls and procedures, internal controls over financial reporting, and compliance with ethical standards adopted by the Company;
• Oversight of treasury and finance matters;
• Oversight and monitoring of enterprise risk management, privacy, and data security;
• Oversight of the auditing, accounting, and financial reporting process generally;
70
• Preparation of a report of the Committee to be included in the Company’s annual proxy statement in accordance with any applicable rules of the SEC; and
• Review and approval of related-party transactions (as defined by any applicable rules of the SEC and any applicable listing standards of the NASDAQ).
The members of the Committee are appointed by the Board at its annual meeting from among the Company’s directors. Members are appointed to serve until their successors are duly elected and qualified by the Board, or until their resignation or removal. The Board determines the number of members on the Committee from time to time, but in any event the Committee is to be composed of at least three Board members or any greater minimum number as required by applicable law, the Company’s Bylaws, or the Company’s contractual obligations. The Board may appoint a chairperson and secretary for the Committee. If the Board does not appoint a chairperson or a secretary, the members of the Committee may elect a chairperson or secretary, respectively, by majority vote.
Each member of the Committee is and must be “independent” in accordance with the Company’s contractual obligations and any applicable SEC and NASDAQ rules. The Board determines the standards that are currently applicable to determining whether a member is “independent” and whether each member or nominee member of the Committee satisfies those standards.
The members of the Committee must also satisfy other applicable qualification rules of NASDAQ and the SEC. Generally, each member of the Committee must have a strong level of accounting or financial acumen and must be able to read and understand fundamental financial statements. A member of the Committee may not have participated in the preparation of financial statements of the Company or any current subsidiary of it at any time during the past three years. To the extent required by applicable rules of the SEC, at least one member of the Committee must be a “financial expert” as defined by the applicable rules of the SEC. In general, to be considered a “financial expert,” an audit committee member must have the following attributes:
• An understanding of generally accepted accounting principles and financial statements.
• The ability to assess the general application of generally accepted accounting principles in connection with the accounting for estimates, accruals, and reserves.
• Experience preparing, auditing, analyzing, or evaluating financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of issues that can reasonably be expected to be raised by the Company’s financial statements, or experience actively supervising one or more persons engaged in such activities.
• An understanding of internal controls and procedures for financial reporting.
• An understanding of audit committee functions.
Steven C. Jones qualifies as an “audit committee financial expert” as the term is defined under the SEC rules, as does Michael Pachos.
Nominating and Corporate Governance Committee
Our Board has established and adopted a charter for a Nominating and Corporate Governance Committee. The Committee is composed of Steven C. Jones, Ravi Shanker and Wonil Kim. As provided in its charter, the Nomination and Corporate Governance Committee was established for the primary purposes of considering and reporting to the Board on matters relating to the identification, selection, and qualification of Board members and candidates nominated to the Board, and assisting the Board with respect to corporate governance matters. The Committee is responsible for providing support to the Board in certain areas, including:
• Assisting the Board by identifying individuals qualified to become Board members.
• Recommending to the Board the director nominees for the next annual meeting of shareholders.
• Leading the Board in an annual review of the Board’s performance.
• Recommending to the Board director nominees for each committee.
• Developing, maintaining, and overseeing the Company’s corporate governance guidelines.
• Making recommendations to the Board with respect to corporate governance matters.
71
The members of the Committee are appointed by the Board at its annual meeting from among the Company directors. The members of the Committee are appointed to serve until their successors are duly elected and qualified by the Board, or until their resignation or removal. The Board determines the number of members on the Committee from time to time, but in any event the Committee must be composed of at least three Board members or any greater minimum number as required by applicable law, the Company’s Bylaws, or the Company’s contractual obligations. The Board may appoint a chairperson and secretary for the Committee. If the Board does not appoint a chairperson or a secretary, the members of the Committee may elect a chairperson or secretary, respectively, by majority vote.
Each member of the Committee is and must be “independent” in accordance with the Company’s contractual obligations and any applicable SEC and NASDAQ rules. The Board determines the standards that are currently applicable to determining whether a member is “independent” and whether each member or nominee member of the Committee satisfies those standards.
Compensation Committee
Our Board has established a Compensation Committee, which is composed of Arnold A. Allemang, Ravi Shanker and Steven C. Jones. The Committee was established for the primary purpose of assisting the Board with the review and determination of executive compensation and the oversight, review, and approval of significant employee benefits programs, policies, and plans. The Board has adopted a Compensation Committee charter.
The members of the Committee are appointed by the Board at its annual meeting from among the Company’s directors. The members of the Committee are appointed to serve until their successors are duly elected and qualified by the Board, or until their resignation or removal. The Board will determine the number of members on the Committee from time to time, but in any event the Committee must be composed of at least three Board members or any greater minimum number as required by applicable law, the Company’s Bylaws, or the Company’s contractual obligations. The Board may appoint a chairperson and secretary for the Committee. If the Board does not appoint a chairperson or a secretary, the members of the Committee may elect a chairperson or secretary, respectively, by majority vote.
Each member of the Committee must be “independent” in accordance with the Company’s contractual obligations and any applicable SEC and NASDAQ rules. Each member of the Committee must also qualify as an “outside director” for purposes of 162(m) of the Internal Revenue Code of 1986, as amended. The Board shall determine the standards that are currently applicable to determining whether a member is “independent” and whether each member or nominee member of the Committee satisfies those standards.
Executive Committee
Our Board has established an Executive Committee, which is composed of Arnold A. Allemang, Steven C. Jones and Dr. Ravi Shanker. Pursuant to our Executive Committee charter, the Executive Committee was established for the primary purpose of exercising the powers and duties of the Board between Board Meetings and while the Board is not in session and to implement policy decisions of the Board.
During the intervals between meetings of the Board, the Committee may exercise all of the powers and authority of the Board of Directors for the purpose of acting upon matters that should not be postponed until the next scheduled meeting of the Board. The members of the Executive Committee are to exercise their business judgment to act in what they reasonably believe to be in the best interests of the Corporation and its shareholders. The Committee will have all powers and authority of the Board enumerated in the Bylaws of the Corporation, except to:
1. Take action specifically reserved for another committee of the Board;
2. Amend the Corporation’s Articles of Incorporation or Bylaws;
3. Adopt an agreement of merger, conversion, or share exchange;
4. Recommend to shareholders the sale, lease, or exchange of all or substantially all of the Corporation’s property and assets;
5. Recommend to shareholders a dissolution of the Corporation or a revocation of a dissolution;
6. Fill vacancies in the Board;
72
7. Declare a distribution or dividend or to authorize the issuance of shares; or
8. Take action with respect to any other matter that the Board may not delegate to the Committee under the Michigan Business Corporation Act, the Corporation’s Articles of Incorporation, the Corporation’s Bylaws, or the Corporation’s contractual commitments.
Notwithstanding the foregoing, the Board may, by resolution or an amendment to this Charter, restrict the powers and authority of the Committee, in its sole discretion. In addition, the Committee shall comply with all directions of the Board and shall discharge all duties and responsibilities expressly delegated by the Board to the Committee.
The Committee may delegate any of its responsibilities, along with the authority to take action in relation to such responsibilities, to one or more subcommittees as the Committee may determine to be appropriate.
Conflicts of Interest
Certain potential conflicts of interest are inherent in the relationships between our officers and directors and us.
From time to time, one or more of our affiliates may form or hold an ownership interest in and/or manage other businesses both related and unrelated to the type of business that we own and operate. These persons expect to continue to form, hold an ownership interest in and/or manage additional other businesses which may compete with our business with respect to operations, including financing and marketing, management time and services and potential customers. These activities may give rise to conflicts between or among the interests of us and other businesses with which our affiliates are associated. Our affiliates are in no way prohibited from undertaking such activities, and neither we nor our stockholders will have any right to require participation in such other activities.
Further, because we have transacted business and intend to continue to do so with some of our officers, directors and affiliates, as well as with firms in which some of our officers, directors or affiliates have a material interest, including, without limitation, Samsung Ventures, XGS II and AAOF, potential conflicts may arise between the respective interests of us and these related persons or entities. We believe that such transactions will be effected on terms at least as favorable to us as those available from unrelated third-parties.
With respect to transactions involving real or apparent conflicts of interest, we have adopted policies and procedures which require that: (i) the fact of the relationship or interest giving rise to the potential conflict be disclosed or known to the directors who authorize or approve the transaction prior to such authorization or approval; and (ii) the transaction be fair and reasonable to us at the time it is authorized or approved by our directors.
Risk Oversight
The Board of Directors is actively involved in the oversight of risks, including strategic, operational and other risks, which could affect our business. The Board of Directors does not have a standing risk management committee, but administers this oversight function directly through the Board of Directors as a whole, which oversee risks relevant to their respective functions. The Board of Directors considers strategic risks and opportunities and administers its respective risk oversight function by evaluating management’s monitoring, assessment and management of risks, including steps taken to limit our exposure to known risks, through regular interaction with our senior management and in board and committee deliberations that are closed to members of management. The interaction with management occurs not only at formal board and committee meetings but also through periodic and other written and oral communications.
Meetings of the Board and Committees
During 2014, there were seven meetings of the Board of Directors. During the first half of 2015, there were three meetings of the Board of Directors. The Board of Directors also acted at times by unanimous written consent, as authorized by our Bylaws and the Michigan Business Corporation Act.
Director Independence
We have five independent directors on our Board of Directors: Arnold A. Allemang, Ravi Shanker, Steven C. Jones, Michael Pachos and Wonil Kim. Philip L. Rose and Michael R. Knox are not considered independent. Because our Common Stock is not currently listed on a national securities exchange, we have used the definition of “independence” of The NASDAQ Stock Market to make this determination.
73
NASDAQ Listing Rule 5605(a)(2) provides that an “independent director” is a person other than an officer or employee of the Company or any other individual having a relationship that, in the opinion of the Company’s Board, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. The NASDAQ listing rules provide that a director cannot be considered independent if:
• the director is, or at any time during the past three years was, an employee of the Company;
• the director or a family member of the director accepted any compensation from the Company in excess of $120,000 during any period of 12 consecutive months within the three years preceding the independence determination (subject to certain exclusions, including, among other things, compensation for board or board committee service);
• a family member of the director is, or at any time during the past three years was, an executive officer of the Company;
• the director or a family member of the director is a partner in, controlling stockholder of, or an executive officer of an entity to which the Company made, or from which the Company received, payments in the current or any of the past three fiscal years that exceed 5% of the recipient’s consolidated gross revenue for that year or $200,000, whichever is greater (subject to certain exclusions);
• the director or a family member of the director is employed as an executive officer of an entity where, at any time during the past three years, any of the executive officers of the Company served on the compensation committee of such other entity; or
• the director or a family member of the director is a current partner of the Company’s outside auditor, or at any time during the past three years was a partner or employee of the Company’s outside auditor, and who worked on the Company’s audit.
The following is a summary of certain of the experience, qualifications, attributes and skills that led the Company’s Board of Directors to conclude that such person should serve as a director or officer This information supplements the biographical information provided above.
Philip L. Rose, Ph.D., Chief Executive Officer, President, Treasurer & Director. Dr. Rose has extensive international business management experience, having previously served as the chief executive officer, president and director of several companies based both in the United States and abroad, and significant experience in the electronic and specialty materials markets. Based on his practical leadership and industry experience, Dr. Rose provides valuable experience and knowledge.
Michael R. Knox, Senior Vice President, Secretary & Director. As a founder of the Company, Mr. Knox’s knowledge of the Company’s history and operations provides a crucial link between management and the Board, enabling the Board to provide its oversight function.
Arnold A. Allemang, Director. Mr. Allemang’s previous board service for several corporations and substantial managerial and operational history will prove valuable to the Board as it seeks to implement and maintain growth strategies that will enable the Company to succeed.
Ravi Shanker, Director. Mr. Shanker’s experience supporting and advising companies with respect to their growth and business development will enable him to assist the Board and management as they endeavor to expand the Company.
Steven C. Jones, Director. Mr. Jones’ background in investment banking and in investing, as well as his prior experience serving as a member of several boards, enables him to provide the Board with valuable insight and expertise.
Wonil Kim, Director. Mr. Kim’s extensive business development and operational expertise will enable him to effectively serve the Board.
Michael Pachos, Director. Mr. Pachos has extensive experience in the investment and securities industries, as well as in implementing strategy, all of which experience will prove useful to the Board.
74
Summary Compensation
The following table sets forth all compensation earned and accrued, in all capacities, during the fiscal years ended December 31, 2015 and 2014 by our named executive officers:
Name and Position |
| Year |
| Salary |
| Bonus |
| Option Expense(1) |
| Other |
| Total | |||||
Philip L. Rose,Chief Executive |
| 2015 |
| $ | 268,654 |
| $ | 18,000 |
| — |
|
| — |
|
| $ | 286,673 |
Officer, Treasurer |
| 2014 |
| $ | 259,135 |
|
| — |
| — |
|
| — |
|
|
| — |
Michael R. Knox,Former Chief |
| 2015 |
| $ | 160,000 |
|
| — |
| — |
| $ | 254,400 | (2) |
| $ | 417,000 |
Executive Officer, Current Chief Financial Officer, Senior |
| 2014 |
| $ | 161,154 |
| $ | 13,500 |
| — |
| $ | 120,200 | (2) |
| $ | 294,854 |
Vice President and Secretary |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Scott Murray,Vice President of |
| 2015 |
| $ | 140,000 |
| $ | 6,725 |
| — |
|
| — |
|
| $ | 148,824 |
Operations |
| 2014 |
| $ | 140,000 |
| $ | 3,000 |
| — |
| $ | 712 | (2) |
| $ | 143,712 |
Robert Privette,Vice President of |
| 2015 |
| $ | 190,000 |
| $ | 10,938 |
| — |
|
| — |
|
| $ | 200,957 |
Energy Markets |
| 2014 |
| $ | 190,000 |
| $ | 9,000 |
| — |
|
| — |
|
| $ | 199,000 |
Liya Wang,Vice President of |
| 2015 |
| $ | 200,000 |
| $ | 14,800 |
| — |
|
| — |
|
| $ | 214,806 |
Research & Development |
| 2014 |
| $ | 200,000 |
| $ | 10,000 |
| — |
|
| — |
|
| $ | 210,000 |
____________
(1) Option expense is calculated using the fair value of options that vested during the period. See Note 12 to our December 31, 2014 financial statements included in this prospectus.
(2) Mr. Knox’s and Mr. Murray’s other compensation included payment of compensation previously deferred under the Company’s Phantom Stock Plan.
Outstanding Equity Awards
The Board of Directors of the Company occasionally awards stock options under the Company’s 2007 Stock Option Plan. Additionally, stock warrants have been issued to certain officers of the Company in conjunction with financing agreements. The following table sets forth information regarding outstanding stock option and stock warrant awards as of December 31, 2015:
Name and Position(s) |
| Number of Securities Underlying Unexercised Options and Warrants that are currently Exercisable |
| Number of Securities Underlying Unexercised Options and Warrants that are currently Unexercisable |
| Exercise Price |
| Expiration Date | ||
Philip L. Rose,Chief Executive Officer, Treasurer |
| 56,665 |
| 103,335 |
|
| $ | 12.00 |
| 1/6/2022 |
Michael R. Knox,Former Chief Executive Officer, |
| 6,000 |
| 15,000 | (1) |
| $ | 8.00 |
| 7/1/2019 |
Current Senior Vice President and |
| 5,000 |
|
|
|
| $ | 12.00 |
| 10/8/2027 |
Secretary |
| 35,000 |
|
|
|
|
| 13.20 |
| 6/1/2021 |
Scott Murray,Vice President of Operations |
| 10,000 |
| 3,333 | (2) |
| $ | 8.00 |
| 12/1/2017 |
|
| 6,667 |
|
|
|
| $ | 12.00 |
| 6/1/2021 |
Robert Privette,Vice President of Energy Markets |
| 13,333 |
| 6,667 | (3) |
| $ | 12.00 |
| 6/1/2021 |
Liya Wang,Vice President of Research & Development |
| 20,000 |
| 10,000 | (4) |
| $ | 12.00 |
| 6/1/2021 |
• Mr. Knox’s remaining unvested options vest at the rate of 15,000 per year on June 1, 2016 in proportion to the amount of time Knox has worked as an active XGS employee during the previous year compared with 1,500 hours per year.
• Mr. Murray’s remaining unvested options vest on June 1, 2016 providing he remains an employee.
75
• Mr. Privette’s unvested options vest on June 1, 2016 providing he remains an employee.
• Mr. Wang’s unvested options vest on June 1, 2016 providing he remains an employee.
Director Compensation
Each of our non-employee Directors who was not appointed as a representative of a corporate investor in XGS is entitled to receive compensation in accordance with Director Compensation plans as amended by the full Board of Directors from time to time. The following table sets forth information concerning the compensation of eligible Directors for the year ended December 31, 2015:
Name |
| Period |
| Cash Compensation |
| Stock Option Expense(1) |
| Total Compensation | |||
Arnold Allemang |
| 2015 |
| $ | 7,500 |
| $ | 9,330 |
| $ | 16,830 |
Steven Jones |
| 2015 |
| $ | 7,500 |
| $ | 3,584 |
| $ | 11,084 |
Ravi Shanker |
| 2015 |
| $ | 7,500 |
| $ | 9,330 |
| $ | 16,830 |
____________
(1) Option expense is calculated using the fair value of options that vested during the period. See Note 12 to the December 31, 2013 financial statements included in this prospectus.
2007 Stock Option Plan
We have established an incentive stock option plan under which we may grant to key employees and Directors options to purchase our common stock at not less than fair market value as of the grant date. Options for up to 600,000 shares may be awarded under the plan. Each option is exercisable into one share of our common stock. The plan expires December 2017. The fair value of the options granted was estimated on the date of grant using the Black Scholes option-pricing model. As of December 31, 2014 and 2013, respectively, 464,750 and 152,500 option shares have been granted. As of September 30, 2015 the number of options granted and outstanding was 389,750. Vesting of the option shares with the employees range from immediately to 20% per year. Rights to exercise the options vest immediately upon a change in control of XGS or termination of the employee’s continuous service due to death or disability. The options expire at various dates through January 2022.
Phantom Stock Plan
On January 1, 2015, the former CEO, Michael R. Knox, was paid $254,400 in deferred compensation under the terms of the phantom stock plan. At the same time, he reinvested the entire payout in a Secured Promissory Note having a maturity on July 1, 2015. This Note was subsequently repaid by the Company in May 2015.
Employment Agreements and Potential Payments Upon Termination
The Company is party to two employment contracts. The following descriptions summarize the commitments in these agreements.
On December 16, 2013 the Company entered into an Employment Agreement with Philip L. Rose to serve as the Company’s Chief Executive Officer commencing on January 6, 2014 and continuing indefinitely, subject to termination by the Company for cause or without cause, or resignation by Dr. Rose with or without cause. If the Company terminates Dr. Rose without cause, or if Dr. Rose resigns with cause, the Company has agreed to pay Dr. Rose’s base salary for a period of six months, any permitted COBRA health insurance premiums for Dr. Rose and his family, and any pro-rata bonus amounts that are deemed to have been earned during Dr. Rose’s employment period prior to termination. In the event that the Company terminates Dr. Rose with cause, or if Dr. Rose resigns without cause, the Company has no further obligations beyond the severance date. The Agreement provides that Dr. Rose will be paid an initial base salary of $275,000 annually, with an annual Target Bonus opportunity of 30% of base salary, which may be earned up to a level of 150% of the Target Bonus under certain conditions. Additionally, Dr. Rose was awarded an option to purchase a total of 220,000 shares of common stock at $12.00 per share and with a life of eight years from the award date. These stock options vest in-part based on based on time and in-part based on the achievement of certain milestones. In addition, Dr. Rose was granted a temporary commuting allowance of up to $50,000 for the calendar years 2014 and 2015 and through August 31, 2016, and a one-time relocation allowance of up to $25,000 for a permanent relocation prior to August 31, 2016.
76
On January 13, 2014 the Company entered into an Employment Agreement with Michael R. Knox, the Company’s former CEO and co-founder. Under the terms of the Agreement, Mr. Knox accepted employment as Senior Vice President for a period of three years unless terminated, with the understanding that Mr. Knox would work approximately 1,500 hours per year on a schedule to be mutually-agreed with the CEO. The Agreement provides Mr. Knox with a base salary of $160,000 annually and includes a continuation of regular employee benefits except paid time off. The Agreement is subject to termination by the Company at any time upon a 30-day notice. If such termination is without cause, the Company has agreed to continue to pay 50% of Mr. Knox’s base salary for the remaining term of the Agreement. This Agreement also references and incorporates a stock option award that vests over the term of the Agreement.
77
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
Related Party Transactions
On March 18, 2013, the Company entered into financing arrangements with two private funds (AAOF and XGS II) that were formed for the sole purpose of investing in the Company by two investors affiliated with ASC-XGS, LLC, a stockholder of the Company. During 2013, these two funds purchased $4,503,847 of secured convertible notes and also provided $635,770 in equipment lease financing. In 2014, these two funds purchased $5,500,000 of secured convertible notes and also provided $390,321 in equipment lease financing. No payments have been made on the secured convertible note and the balance due, including interest, is approximately $15,604,429.
On January 15, 2014, we sold $3 million of secured convertible Notes to Samsung Ventures, a new lender and stockholder of the Company. As part of this investment, Samsung SDI, the largest maker of lithium ion batteries in the world, committed to entering into a joint development agreement with XGS to jointly development a new lithium ion battery anode material based on the Company’s proprietary silicon graphene material.
On March 31, 2014, we sold 100,000 shares of Series A convertible preferred stock to POSCO for $1.2 million as part of a preemptive rights offering triggered by the AAOF and Samsung investments, bringing the total POSCO investment in XGS to $5.2 million.
During 2014, we issued 29,169 shares of Series A convertible preferred stock to AAOF as payment for lease financing obligations under the terms of a Master Leasing Agreement.
During the nine months ended September 30, 2015, Michael R. Knox was paid $254,000 under the terms of a phantom stock agreement.
During the first nine months of 2015, we issued 19,040 shares of Series A convertible preferred stock to AAOF as payment for lease financing obligations under the terms of a Master Leasing Agreement.
In April 2015, we commenced a private placement offering of up to $18,000,000 in units consisting of up to 1,125,000 shares of Series B Convertible Preferred Stock and Warrants to purchase Common Stock at an offering price of $16.00 per Unit. Each of Steven Jones and Arnold Allemang invested $250,000 in the offering of Series B Units.
On December 31, 2015, we issued December Notes and December Warrants to existing stockholders. The December Notes mature on June 30, 2016 and the December Warrants have a five year term and a strike price of $8.00. Each of Steven Jones, Arnold Allemang, and Dave Pendell purchased December Notes for $250,000, $250,000 and $50,000 respectively, and purchased December Warrants for 9,375, 9375, and 1,875 shares of common stock respectively.
Shareholder Agreement
In conjunction with a financing with AAOF, the Company and the stockholders of the Company listed therein entered into a Shareholder Agreement on March 18, 2013 that contains a number of specific provisions pertaining to the Board of Directors as well as individual Directors. Among other things, the Shareholder Agreement provides for certain voting and nomination rights to be calculated on the basis of “Full Conversion” stock ownership (under which calculation, all convertible notes, preferred shares, or other convertible equity securities are deemed converted into common stock) as follows:
So long as AAOF or its affiliates own 10% of more of the aggregate outstanding Shareholder Stock:
• the size of the Board of Directors shall be set at seven individuals.
• one person nominated by AAOF shall be elected to the Board of Directors.
• two members of the Board of Directors, other than those nominated by AAOF, POSCO or Hanwha Chemical, shall qualify as independent Directors.
78
• So long as POSCO owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by POSCO shall be elected to the Board of Directors.
• So long as Hanwha Chemical owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by Hanwha Chemical shall be elected to the board of directors.
As of December 31, 2015, the ownership percentage of AAOF and POSCO, as calculated for purposes of Director voting, required the stockholders to vote for a Director nominated by AAOF and for a director nominated by POSCO. Mr. Jones is the AAOF representative to the Board and Mr. Oh is the POSCO representative to the Board pursuant to the terms of the Shareholder Agreement.
On January 15, 2014, AAOF, POSCO, SVIC, XGS II, Mike Knox and the Company entered into a letter agreement pursuant to which AAOF agreed to consent to, approve or otherwise sanction certain actions of the Company specified in the Shareholder Agreement so long as investors owning at least 66% of the voting securities on a fully diluted basis consent to the proposed Company action. These actions include if the Company (i) creates, obligates itself to create, authorizes or issues (by reclassification or otherwise) any new class or classes of securities which has a preference over or being on a parity with the Series A Preferred Stock in any respect; (ii) sells any material portion of the tangible or intangible assets of the Company that secures the indebtedness to AAOF; or (iii) incurs additional secured indebtedness.
The Shareholder Agreement grants preemptive rights to shareholders and holders of convertible notes who are parties to the Shareholder Agreement. Pursuant to the terms therein, such shareholders and noteholders have the right to purchase their pro rata share of all shareholder stock that the Company may, from time to time, propose to sell, issue, or exchange after the date of the Shareholder Agreement, other than certain excluded stock which includes stock granted to employees or as merger consideration. Each shareholder’s pro rata shares shall be equal to the ratio of (i) the aggregate number of shares of the Company’s common stock on a fully diluted basis, owned by the such shareholder at the time of the delivery of a preemptive rights notice to (ii) the aggregate number of shares of Company’s common stock on a fully diluted basis owned by all of the Company’s shareholders at the time of the delivery of a preemptive rights notice.
The Shareholder Agreement shall continue in full force and effect until the date of the closing of a public offering of our common stock pursuant to a registration statement filed with the SEC is declared effective in which the Company receives gross proceeds of at least $10,000,000, on which date it shall terminate in its entirety, unless the Shareholder Agreement is earlier terminated in accordance with its terms. Until such time as it is terminated, however, new shareholders will be required to execute an adoption agreement to the Shareholder Agreement. By adopting the Shareholder Agreement, such new shareholders will be bound by its terms, including the transfer restrictions provided therein.
Voting Agreement
In conjunction with a financing from Samsung Ventures on January 15, 2014, the Company and certain of its stockholders entered into a voting agreement with Samsung Ventures whereby for so long as Samsung Ventures owns 10% or more of the aggregate outstanding common stock of the Company (assuming Full Conversion, but excluding any shares of common stock issuable upon the exercise of any warrants held by Samsung Ventures), each stockholder made a party thereto shall vote all of his, her or its voting securities from time to time in whatever manner is necessary to ensure that at each annual or special meeting of the stockholders at which an election of Directors is held or pursuant to any written consent of the stockholders, one person nominated by Samsung Ventures is elected to the Company’s Board as a Director.
The rights granted to Samsung Ventures under a voting agreement are effective until the first to occur of (1) the date on which the Minimum Ownership Requirement is no longer satisfied, (2) the date on which the Shareholder Agreement (which is described above) is terminated for any reason and (3) the date that Samsung Ventures agrees in writing to terminate the Agreement.
79
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND
RELATED STOCKHOLDER MATTERS
As of December 31, 2015, the following table sets forth certain information with respect to the beneficial ownership of our common stock, Series A Preferred Stock and Series B Preferred Stock by (i) by each of our directors and executive officers, (ii) by all of our director and executive officers as a group, and (iii) by each person or entity known by us to beneficially own more than 5% of any class of our outstanding shares. We have determined the number and percentage of shares beneficially owned by such person in accordance with Rule 13d-3 under the Exchange Act.
Name and Address of Beneficial Owner(1)(2) |
| Shares of Common Stock Beneficially Owned(3) |
| Percentage of Shares of Common Stock Beneficially Owned(4) |
| Shares of Series A Preferred Stock Beneficially Owned(5) |
| Percentage of Shares of Series A Preferred Stock Beneficially Owned(6) |
| Shares of Series B Preferred Stock Beneficially Owned(7) |
| Percentage of Shares of Series B Preferred Stock Beneficially Owned(8) |
| Total Percentage of Outstanding Shares of Voting Stock Beneficially Owned(9) | ||||
Directors, Executive Officers, and Significant Employees | ||||||||||||||||||
Phillip L. Rose(10) |
| 59,790 |
| 6.67 | % |
| — |
| — |
|
| 3,125 |
| 1.14 | % |
| 2.02 | % |
Michael R. Knox(11) |
| 210,074 |
| 20.07 | % |
| 72,226 |
| 3.86 | % |
| — |
| — |
|
| 6.61 | % |
Arnold A. Allemang(12) |
| 21,125 |
| 2.46 | % |
| — |
| — |
|
| 15,625 |
| 5.47 | % |
|
| * |
Steven C. Jones(13) |
| 33,156 |
| 3.94 | % |
| — |
| — |
|
| 15,625 |
| 5.47 | % |
| 1.11 | % |
Wonil Kim(14) |
| — |
| — |
|
| — |
| — |
|
| — |
| — |
|
| — |
|
Ravi Shanker(15) |
| 4,000 |
|
| * |
| — |
| — |
|
| 1,000 |
|
| * |
|
| * |
Michael Pachos(16) |
| — |
| — |
|
| — |
| — |
|
| — |
| — |
|
| — |
|
Scott Murray(17) |
| 13,334 |
| 1.57 | % |
| — |
| — |
|
| — |
| — |
|
|
| * |
Robert Privette(18) |
| 13,333 |
| 1.57 | % |
| — |
| — |
|
| — |
| — |
|
|
| * |
Liya Wang(19) |
| 20,000 |
| 2.33 | % |
| — |
| — |
|
| — |
| — |
|
|
| * |
Directors & Executive Officers as a Group (10 persons) |
| 374,812 |
| 38.60 | % |
| 72,227 |
| 3.86 | % |
| 35,375 |
| 11.58 | % |
| 11.20 | % |
Certain Other Beneficial Owners – Over 5% Ownership | ||||||||||||||||||
Aspen Advanced Opportunity Fund, LP(20) |
| 1,851,081 |
| 68.87 | % |
| 1,851,081 |
| 50.69 | % |
| — |
| — |
|
| 38.90 | % |
POSCO(21) |
| 350,000 |
| 29.50 | % |
| 150,000 |
| 7.69 | % |
| — |
| — |
|
| 10.75 | % |
ASC-XGS, LLC(22) |
| 166,023 |
| 16.56 | % |
| 166,023 |
| 8.44 | % |
| — |
| — |
|
| 5.40 | % |
Hanwha Chemical(23) |
| 150,000 |
| 17.93 | % |
| — |
| — |
|
| — |
| — |
|
| 4.91 | % |
XGS II, LLC(24) |
| 192,505 |
| 18.71 | % |
| 109,172 |
| 5.72 | % |
| — |
| — |
|
| 6.21 | % |
SVIC No. 15 New Technology Business Investment LLP(25) |
| 314,710 |
| 27.34 | % |
| 314,710 |
| 14.88 | % |
| — |
| — |
|
| 9.25 | % |
Inhwan Do(26) |
| 50,500 |
| 5.69 | % |
| 500 |
|
| * |
| — |
| — |
|
| 1.71 | % |
Hiroyuki Fukushima(27) |
| 50,500 |
| 5.69 | % |
| 500 |
|
| * |
| — |
| — |
|
| 1.71 | % |
Lawrence T. Drzal(28) |
| 51,150 |
| 5.76 | % |
| — |
| — |
|
| 600 |
|
| * |
| 1.73 | % |
____________
* Less than 1%
(1) Beneficial ownership is determined in accordance with the rules of the Securities and Exchange Commission and generally includes voting or investment power with respect to securities. In accordance with SEC rules, shares of stock issuable upon the exercise of options, warrants and other convertible securities which are currently exercisable and convertible or which become exercisable or convertible within sixty (60) days following the date of the information in this table are deemed to be beneficially owned by, and outstanding with respect to, the holder of such option, warrant or other convertible security. Subject to community property laws where applicable, to our knowledge, each person listed is believed to have sole voting and investment power with respect to all shares owned by such person.
(2) Unless otherwise specified, the address of the beneficial owner shall be the business address of the Company, c/o XG Sciences, Inc., 3101 Grand Oak Drive, Lansing, MI 48911.
(3) Number of shares of common stock includes, as of December 31, 2015: (i) shares of common stock registered in the name of the respective stockholder; (ii) shares of Series A Preferred Stock registered in the name of the respective stockholder which are immediately convertible into shares of common stock; (iii) shares of Series B Preferred Stock registered in the name of the respective stockholder which are immediately convertible into shares of common stock; (iv) shares of common stock underlying options and/or warrants of the respective stockholder and which are exercisable within sixty (60) days of
80
December 31, 2015; (v) shares of Series A Preferred Stock which are immediately convertible into shares of common stock underlying warrants and/or convertible notes of the respective stockholder which are exercisable or convertible into shares of Series A Preferred Stock within sixty (60) days of December 31, 2015; and (vi) shares of Series B Preferred Stock which are immediately convertible into shares of common stock underlying warrants and/or convertible notes of the respective stockholder which are exercisable or convertible into shares of Series B Preferred Stock within sixty (60) days of days of December 31, 2015. The shares of common stock herein does not include the common stock issuable upon conversion of the Series A Preferred Stock at a ration of 1.875 to 1 and does not include the common stock issuable upon exchange of the Series B Units for shares of common stock.
(4) Applicable percentage of ownership is based on 836,544 shares of common stock issued and outstanding as of December 31,2015, together with: (i) shares of common stock registered in the name of the respective stockholder; (ii) shares of Series A Preferred Stock registered in the name of the respective stockholder which are immediately convertible into shares of common stock; (iii) shares of Series B Preferred Stock registered in the name of the respective stockholder which we will convert into shares of common stock; (iv) shares of common stock underlying options and/or warrants of the respective stockholder and which are exercisable within sixty (60) days of December 31, 2015.
(5) Number of shares of Series A Preferred Stock includes, as of December 31, 2015: (i) shares of Series A Preferred Stock registered in the name of the respective stockholder; and (ii) shares of Series A Preferred Stock underlying warrants and/or secured notes of the respective stockholder which are exercisable or convertible within sixty (60) days of December 31, 2015. The number of shares of Series A Preferred Stock does not assume the conversion of the Series A Preferred Stock on a 1.875 to 1 ratio.
(6) Applicable percentage of ownership is based on 1,800,696 shares of Series A Preferred Stock issued and outstanding as of December 31, 2015, together with shares of Series A Preferred Stock underlying warrants and/or secured notes of the respective stockholder which are exercisable or convertible within sixty (60) days of December 31, 2015.
(7) Number of shares of Series B Preferred Stock includes, as of December 31, 2015, (i) shares of Series B Preferred Stock registered in the name of the respective stockholder; and (ii) shares of Series B Preferred Stock underlying warrants and/or secured notes of the respective stockholder which are exercisable or convertible within sixty (60) days of December 31, 2015. The number of Series B Preferred Stock used in the table does not assume the exchange of the Series B Unit for common stock.
(8) Applicable percentage of ownership is based on 269,987 shares of Series B Preferred Stock issued and outstanding as of December 31, 2015 together with shares of Series B Preferred Stock underlying warrants and/or secured notes of the respective stockholder which are exercisable or convertible within sixty (60) days of December 31, 2015.
(9) Applicable percentage of ownership is based on 836,544 shares of common stock, 1,800,696 shares of Series A Preferred Stock, and 269,987 shares of Series B Preferred Stock issued and outstanding as of December 31, 2015, together with shares of common stock underlying options and/or warrants of the respective stockholder which are exercisable within sixty (60) days of December 31, 2015 and shares of Series A Preferred Stock and Series B Preferred Stock underlying warrants and/or convertible notes of the respective stockholder which are convertible into shares of Series A Preferred Stock within sixty (60) days of December 31, 2015.
(10) Includes 56,665 shares of common stock underlying currently exercisable options and 3,125 shares of common stock underlying immediately convertible shares of Series B Preferred Stock.
(11) Includes 127,047 shares of common stock, 35,000 shares of common stock underlying currently exercisable options, 11,000 shares underlying vested warrants to purchase common stock, and 72,226 shares of common stock underlying shares of immediately convertible Series A Preferred Stock which are currently issuable upon the conversion of outstanding convertible notes. Mr. Knox’s interest also includes 5,380 shares of stock issued in the name of his wife, Linnea Van Dyne.
(12) Includes 5,500 shares of common stock underlying currently exercisable options and 15,625 shares of common stock underlying immediately convertible shares of Series B Preferred Stock. The shares of Series B Preferred Stock are held in the name of the Arnold Avery Allemang Revocable Trust.
(13) Includes (i) 4,250 shares of common stock underlying currently exercisable options held by Steven Jones; (ii) 9,375 shares of common stock underlying Series B Preferred Stock and 7,969 shares of common stock underlying warrants, held in thename Jones Network, LP, of which Mr. Jones, a Director, is a partner; (ii) 3,125 shares of common stock underlying Series B Preferred Stock and 2,656 shares of common stock underlying warrants, held in the name Jones Extended Family Trust, of which Mr. Jones is trustee; and (iii) 3,125 shares of common stock underlying Series B Preferred Stock and 2,656 shares of common stock underlying warrants, held in the name Steven & Carisa Jones 401K Plan and Trust, of which Mr. Jones is trustee. Mr. Jones is an affiliate of Aspen Advanced Opportunity Fund, LP (“AAOF”), which owns 48,209 shares of Series A Preferred Stock which are issuable upon the conversion of immediately convertible notes. Mr. Jones disclaims beneficial ownership of the shares owned by AAOF.
81
(14) Mr. Kim is a director of POSCO and disclaims beneficial ownership of any shares owned by POSCO.
(15) Includes 3,000 shares of common stock underlying currently exercisable options, and 1,000 shares of common stock underlying immediately convertible shares of Series B Preferred Stock.
(16) Mr. Pachos was appointed to the Board by Samsung and disclaims beneficial ownership of any shares owned by Samsung.
(17) Includes 13,334 shares of common stock underlying currently exercisable options.
(18) Includes 13,333 shares of common stock underlying currently exercisable options.
(19) Includes 20,000 shares of common stock underlying currently exercisable options.
(20) Includes 1,017,748 shares of common stock underlying shares of Series A Preferred Stock which were issued upon the conversion of convertible notes, and 833,333 shares of Series A Preferred Stock issuable upon the exercise of warrants.
(21) Includes 200,000 shares of common stock, 100,000 shares of common stock underlying immediately convertible shares of Series A Preferred Stock and 50,000 shares of Series A Preferred Stock issuable upon the exercise of warrants.
(22) Includes 166,023 shares of common stock underlying immediately convertible shares of Series A Preferred Stock.
(23) Includes 150,000 shares of common stock.
(24) Includes 83,333 shares of Series A Preferred Stock issuable upon the exercise of warrants and 102,845 shares of common stock underlying Series A Preferred Stock which are issuable upon the conversion of immediately convertible notes.
(25) Includes 296,471 shares of common stock underlying Series A Preferred Stock which are issuable upon the conversion of immediately convertible notes.
(26) Includes 50,000 shares of common stock and 500 shares of common stock underlying immediately convertible shares of Series A Preferred Stock.
(27) Includes 50,000 shares of common stock and 500 shares of common stock underlying immediately convertible shares of Series A Preferred Stock.
(28) Includes 50,550 shares of common stock and 600 shares of common stock underlying immediately convertible shares of Series B Preferred Stock. The 50,550 shares of common stock are held in the name of the Drzal Revocable Trust, for which Mr. Drzal is the Trustee.
Change in Control Arrangements
We are not aware of any arrangements that could result in a change of control.
82
The 4,784,442 shares of common stock being offered for resale by the selling securityholders consist of (i) up to 836,544 shares were previously issued to the selling securityholders in private placements during the calendar years 2007 through 2014, (ii) up to 31,625 shares issuable upon exercise of outstanding warrants to purchase shares of common stock that were issued to selling securityholders, (iii) up to 3,376,299 shares issuable upon the conversion of shares of our Series A Preferred Stock that were previously issued to selling securityholders, and (iv) up to 539,974 shares issuable upon the exchange of shares of our Series B Preferred Stock that were previously issued to selling securityholders, (including 269,987 shares issuable upon an exchange for units).
The following table sets forth the names of the selling securityholders, the number of shares of common stock beneficially owned by each of the selling securityholders as of December 31, 2015 and the number of shares of common stock being offered by the selling securityholders. The shares being offered hereby are being registered to permit public secondary trading, and the selling securityholders may offer all or part of the shares for resale from time to time. However, the selling securityholders are under no obligation to sell all or any portion of such shares nor are the selling securityholders obligated to sell any shares immediately upon effectiveness of this prospectus.
To our knowledge, except as set forth in the footnotes in the table below, none of the selling securityholders listed in this prospectus has had a material relationship with us other than as a securityholder at any time since inception of the Company and none of the selling securityholders are broker-dealers or affiliated with any broker-dealers.
We do not have any predecessors and are not affiliated with any other entities with the exception of XG Sciences IP, LLC, our wholly owned subsidiary.
All information with respect to share ownership has been furnished to us by the selling securityholders.
Name of Selling Securityholder |
| Shares BeneficiallyOwnedPrior ToOffering(1) |
| Shares to be Offered(2) |
| Amount BeneficiallyOwnedAfterOffering |
| Percent BeneficiallyOwned afterOffering(3) | |
Michael R. Knox(4) |
| 210,274 |
| 210,274 |
| 35,000 |
|
| * |
Aspen Advanced Opportunity Fund(5) |
| 1,851,081 |
| 1,017,748 |
| 833,333 |
| 13 | % |
POSCO(6) |
| 350,000 |
| 300,000 |
| 50,000 |
|
| * |
ASC-XGS, LLC(7) |
| 166,023 |
| 166,023 |
| — |
| 0 |
|
XGS II, LLC(8) |
| 192,505 |
| 109,172 |
| 83,333 |
|
| * |
SVIC No. 15(9) |
| 314,710 |
| 314,710 |
| — |
| 0 |
|
Hanwha Chemical(10) |
| 150,000 |
| 150,000 |
| — |
| 0 |
|
Hiroyuki Fukushima(11) |
| 50,500 |
| 50,500 |
| — |
| 0 |
|
Inhwan Do(12) |
| 50,500 |
| 50,500 |
| — |
| 0 |
|
Lawrence T Drzal, Trustee of the Drzal Revocable Trust(13) |
| 51,150 |
| 51,150 |
| — |
| 0 |
|
Colin D. Cronin(14) |
| 14,495 |
| 12,728 |
| 1,767 |
|
| * |
John W. Dourjalian(15) |
| 5,813 |
| 4,772 |
| 1,041 |
|
| * |
Paul Nordstrom(16) |
| 12,121 |
| 10,000 |
| 2,121 |
|
| * |
Peter S. Bosanic & Lisa Kendzioski Bosanic (JTWROS)(17) |
| 4,425 |
| 3,800 |
| 625 |
|
| * |
Steven & Carisa Jones 401K Plan & Trust(18) |
| 33,156 |
| 33,156 |
| — |
| 0 |
|
Robert Skandalaris |
| 34,977 |
| 34,977 |
| — |
| 0 |
|
Mark S. Kerrins & Rosemary Kerrins (JTWROS) |
| 32,500 |
| 32,500 |
| — |
| 0 |
|
Ann Arbor Spark |
| 36,257 |
| 36,257 |
| — |
| 0 |
|
David J. Donovan |
| 16,257 |
| 16,257 |
| — |
| 0 |
|
Edward J. Demmer |
| 30,000 |
| 30,000 |
| — |
| 0 |
|
Gary Griffin |
| 1,500 |
| 1,500 |
| — |
| 0 |
|
Iris K. Linder |
| 1,667 |
| 1,667 |
| — |
| 0 |
|
83
Name of Selling Securityholder |
| Shares BeneficiallyOwnedPrior ToOffering(1) |
| Shares to be Offered(2) |
| Amount BeneficiallyOwnedAfterOffering |
| Percent BeneficiallyOwned afterOffering(3) |
Jeffery A. Wesley |
| 9,005 |
| 9,005 |
| — |
| 0 |
Kevin B. Miller |
| 2,400 |
| 2,400 |
| — |
| 0 |
Marjorie E. Frisch |
| 2,087 |
| 2,087 |
| — |
| 0 |
Mark E. Hooper Revocable Living Trust(21) |
| 3,625 |
| 3,625 |
| — |
| 0 |
Mathew H. Frisch |
| 2,087 |
| 2,087 |
| — |
| 0 |
MSU Foundation |
| 9,543 |
| 9,543 |
| — |
| 0 |
William D. Hamilton and Susan M. Heathfield |
| 7,251 |
| 7,251 |
| — |
| 0 |
Stephen J. Linder Trust No. 1(22) |
| 5,251 |
| 5,251 |
| — |
| 0 |
William G. Lutz |
| 3,053 |
| 3,053 |
| — |
| 0 |
Reed Shick |
| 880 |
| 880 |
| — |
| 0 |
Robert L. McKellar |
| 592 |
| 592 |
| — |
| 0 |
Dave and Vicky Pendell |
| 3,157 |
| 3,157 |
| — |
| 0 |
George Cardoza(19) |
| 2,500 |
| 2,500 |
| — |
| 0 |
William Robison(19) |
| 57,813 |
| 57,813 |
| — |
| 0 |
Steven Purcell(19) |
| 11,563 |
| 11,563 |
| — |
| 0 |
James D. Chambers(19) |
| 11,100 |
| 11,100 |
| — |
| 0 |
Golden Hills Investments, LLLP(19)(23) |
| 28,906 |
| 28,906 |
| — |
| 0 |
Muriel F. Licht Trust(19)(24) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Hewit Hawn(19) |
| 579 |
| 579 |
| — |
| 0 |
First Trust Company of Onaga FBO Kenneth RJ Rettig(19)(25) |
| 2,081 |
| 2,081 |
| — |
| 0 |
David G. Pendell Revocable Trust(19)(26) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Edelgard Traut(19) |
| 1,850 |
| 1,850 |
| — |
| 0 |
First Trust Company of Onaga FBO George A. Shaw(19)(27) |
| 5,781 |
| 5,781 |
| — |
| 0 |
John M. O’Neill(19) |
| 1,850 |
| 1,850 |
| — |
| 0 |
L.D. Sipes, Jr.(19) |
| 18,500 |
| 18,500 |
| — |
| 0 |
Lexy and Steven Shroyer Family Trust 2004(19)(28) |
| 17,344 |
| 17,344 |
| — |
| 0 |
Robert F. Moyer Revocable Trust dated |
| 8,673 |
| 8,673 |
| — |
| 0 |
Samuel H. Jones Jr.(19) |
| 11,563 |
| 11,563 |
| — |
| 0 |
Elaine D. Cavanna(19) |
| 3,006 |
| 3,006 |
| — |
| 0 |
Shannondoah, LLC(19)(30) |
| 1,500.0 |
| 1,500.0 |
| — |
| 0 |
Traut Family Trust UTD 7/3/13(19)(31) |
| 1,850 |
| 1,850 |
| — |
| 0 |
Ravi Shanker(19) |
| 1,850 |
| 1,850 |
| — |
| 0 |
David Fuhrhop(19) |
| 1,077 |
| 1,077 |
| — |
| 0 |
Thomas Bombeck IV(19) |
| 5,550 |
| 5,550 |
| — |
| 0 |
Arnold Avery Allemang Revocable Trust(19)(32) |
| 28,906 |
| 28,906 |
| — |
| 0 |
George J. and/or Beverly A. Biltz(19) |
| 6,938 |
| 6,938 |
| — |
| 0 |
Philip Rose(19)(20) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Laurie L. Bock Administrative Trust(19)(33) |
| 1,735 |
| 1,735 |
| — |
| 0 |
Kay B. Coin(19) |
| 2,892 |
| 2,892 |
| — |
| 0 |
LMRAY, LLC(19)(34) |
| 868 |
| 868 |
| — |
| 0 |
Spinnaker Brown, LLC(19)(35) |
| 3,123 |
| 3,123 |
| — |
| 0 |
Who Dat Nation Trust(19)(36) |
| 3,256 |
| 3,256 |
| — |
| 0 |
Arthur Baumann(19) |
| 1,504 |
| 1,504 |
| — |
| 0 |
N. Rich Holdings, LLC(19)(37) |
| 3,700 |
| 3,700 |
| — |
| 0 |
84
Name of Selling Securityholder |
| SharesBeneficiallyOwnedPrior ToOffering(1) |
| Shares to be Offered(2) |
| AmountBeneficiallyOwnedAfterOffering |
| PercentBeneficiallyOwned afterOffering(3) |
M. Rich Holdings, LLC(19)(38) |
| 11,100 |
| 11,100 |
| — |
| 0 |
David P. Stern Trust UA 12-19-200(19)(39) |
| 925 |
| 925 |
| — |
| 0 |
Arnold S. and Bette G. Hoffman(19) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Tomas R. Manson Family Trust(19)(40) |
| 2,775 |
| 2,775 |
| — |
| 0 |
Thomas and Carmelita B. Karacic(19) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Erik J. Falconer(19) |
| 5,550 |
| 5,550 |
| — |
| 0 |
Brian G. Martindale Trust(19)(41) |
| 5,920 |
| 5,920 |
| — |
| 0 |
OK Enterprises(19)(42) |
| 1,735 |
| 1,735 |
| — |
| 0 |
Mary V. Murfey Family Trust #2(19)(43) |
| 7,400 |
| 7,400 |
| — |
| 0 |
Galt Asset Management(19)(44) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Paul Anthony Radomski(19) |
| 18,500 |
| 18,500 |
| — |
| 0 |
Stevens Financial Group, LLC(19)(45) |
| 57,813 |
| 57,813 |
| — |
| 0 |
First Trust Company of Onaga FBO James M Rajewski IRA#TW000918(19)(46) |
| 4,338 |
| 4,338 |
| — |
| 0 |
David Ari Johnson and Gabrielle Johnson Revocable Trust 1-13-14(19)(47) |
| 2,892 |
| 2,892 |
| — |
| 0 |
Timothy Long Living Trust(19)(48) |
| 1,156 |
| 1,156 |
| — |
| 0 |
Thomas W. Rouse(19) |
| 2,255 |
| 2,255 |
| — |
| 0 |
Morgan Duke(19) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Howard Moskow(19) |
| 2,892 |
| 2,892 |
| — |
| 0 |
Hoffman Investment Company(19)(49) |
| 5,781 |
| 5,781 |
| — |
| 0 |
Russell S. Deane Trust 1/4/2008(19)(50) |
| 2,313 |
| 2,313 |
| — |
| 0 |
Scott Lizenby(19) |
| 740 |
| 740 |
| — |
| 0 |
Michael D. Bierlein Intervivos Trust(19)(51) |
| 3,282.0 |
| 3,282.0 |
| — |
| 0 |
Lillian Elizabeth Deane Trust dated July 2, 1993(19)(52) |
| 625.0 |
| 625.0 |
| — |
| 0 |
Jennifer Dana Dean(19) |
| 1,850 |
| 1,850 |
| — |
| 0 |
First Trust Company of Onaga FBO Robert L Hartsock(19)(53) |
| 6,072 |
| 6,072 |
| — |
| 0 |
Robert and Carlita Gasparini(19) |
| 2,892 |
| 2,892 |
| — |
| 0 |
Rodney Boulanger(19) |
| 5,781 |
| 5,781 |
| — |
| 0 |
James R. DeVore and Joan Mobley-DeVore(19) |
| 15,000 |
| 15,000 |
| — |
| 0 |
Lawrence T Drzal |
| 600 |
| 600 |
| — |
| 0 |
____________
* Represents less than 1%.
(1) The number of shares beneficially owned before the offering includes shares of common stock and shares of common stock issuable upon conversion of Series A Preferred Stock, common stock issuable upon conversion of Series B Preferred Stock, common stock issuable upon exercise of warrants and common stock underlying shares of Series A Preferred Stock resulting from the conversion of convertible notes.
(2) Represents shares of common stock and shares of common stock issuable upon conversion of the convertible notes, common stock issuable upon conversion of the Series A Preferred Stock, common stock issuable upon conversion of the Series B Preferred Stock and common stock issuable upon exercise of the warrants.
(3) Assumes that (i) all of the shares of common stock to be registered on the registration statement, including all shares of common stock underlying Series A Preferred Stock, Series B Preferred Stock, warrants and convertible notes held by the selling securityholders, are sold in the offering and (ii) that no other shares of common stock are acquired or sold by the selling securityholder prior to the completion of the offering.
(4) Includes 121,667 shares of common stock, including 5,380 shares held by Mr. Knox’s wife, Linnea Van Dyne, 11,000 shares of common stock issuable upon the conversion of warrants, and 72,227 shares of common stock underlying Series A Preferred Stock issuable upon the conversion of notes held by Michael R Knox and Linnea Van Dyne JTWROS, of which Mr. Knox is a trustee. Mr. Knox is a co-founder of the Company and serves as the Company’s Senior Vice President, Secretary, and as a Director.
85
(5) Includes 57,729 shares of common stock underlying Series A Preferred Stock and 960,019 shares of common stock underlying Series A Preferred Stock issued upon the conversion of notes, and 833,333 shares of common stock underlying warrants which are not being registered in this prospectus. Mr. Steven Jones is the Manager of Aspen Advanced Opportunity Fund and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Advanced Opportunity Fund. Mr. Jones has sole voting and disposition power over such shares being offered under this prospectus.
(6) Includes 200,000 shares of common stock and 100,000 shares of common stock underlying Series A Preferred Stock, and 50,000 shares of common stock underlying warrants which are not being registered in this prospectus. Wonil Kim is the authorized representative of POSCO and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by POSCO. Wonil Kim has shared voting and disposition power over such shares being offered under this prospectus.
(7) Includes 166,023 shares of common stock underlying Series A Preferred Stock. David G. Pendell is the Manager of ASC-XGS and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by ASC-XGS. David G. Pendell has shared voting and disposition power over such shares being offered under this prospectus.
(8) Includes 109,172 shares of common stock underlying Series A Preferred Stock issued upon the conversion of notes, and 83,333 shares of common stock underlying warrants which are not being registered in this prospectus. David G. Pendell is the Manager of XGS II and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by XGS II. David G. Pendell has shared voting and disposition power over such shares being offered under this prospectus.
(9) Includes 314,710 shares of common stock underlying Series A Preferred Stock issued upon the conversion of notes. Excludes 100,000 shares of common stock underlying warrants which have not yet vested. Michael Pachos is the authorized representative of SVIC No. 15 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by SVIC No. 15. Michael Pachos has shared voting and disposition power over such shares being offered under this prospectus.
(10) Includes 150,000 shares of common stock. Hong Man Park is the authorized representative of Hanwha and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Hanwha. Hong Man Park has sharedvoting and disposition power over such shares being offered under this prospectus.
(11) Includes 50,000 shares of common stock and 500 shares of common stock underlying Series A Preferred Stock. Hiroyuki Fukushima is a founder of the Company and is employed by the Company as a Senior Scientist.
(12) Includes 50,000 shares of common stock and 500 shares of common stock underlying Series A Preferred Stock. Inhwan Do is a founder of the Company and, until recently, was employed by the Company as a Senior Scientist.
(13) Includes 50,500 shares of common stock and 600 shares of common stock underlying Series B Preferred Stock. Lawrence T. Drzal is the trustee of the Drzal Revocable Trust and as such may be deemed to be the beneficial owner of all of the shares of common stock owned by Drzal Revocable Trust. Mr. Drzal has sole voting and disposition power over such shares being offered under this prospectus. Mr. Drzal is a founder of the Company and is its Chief Scientist. Mr. Drzal served as a director from 2006 to 2013.
(14) Includes 2,803 shares of common stock, 5,300 shares of common stock underlying Series A Preferred Stock, 2,500 shares of common stock underlying Series B Preferred Stock, and 3,892 shares of common stock underlying warrants, 1,767 of which are not being registered hereunder.
(15) Includes 1,649 shares of common stock, 3,123 shares of common stock underlying Series A Preferred Stock and 1,041 shares of common stock underlying warrants, which shares underlying warrants are not being registered hereunder.
(16) Includes 3,637 shares of common stock, 6,363 shares of common stock underlying Series A Preferred Stock and 2,121 shares of common stock underlying warrants, which shares underlying warrants are not being registered hereunder.
(17) Includes 1,925 shares of common stock, 1,875 shares of common stock underlying Series A Preferred Stock and 625 shares of common stock underlying warrants, which shares underlying warrants are not being registered hereunder.
(18) Includes (i) 9,375 shares of common stock underlying Series B Preferred Stock and 7,969 shares of common stock underlying warrants, held in the name Jones Network, LP, of which Steven Jones, a Director, is a partner; (ii) 3,125 shares of common stock underlying Series B Preferred Stock and 2,656 shares of common stock underlying warrants, held in the name Jones Extended Family Trust, of which Mr. Jones is trustee; and (iii) 3,125 shares of common stock underlying Series B Preferred Stock and 2,656 shares of common stock underlying warrants, held in the name Steven & Carisa Jones 401K Plan and Trust, of which Mr. Jones is trustee.
(19) Includes shares of common stock underlying Series B Preferred Stock and shares of common stock underlying warrants with 85% warrant coverage.
(20) Includes shares of common stock underlying Series B Preferred Stock and shares of common stock underlying warrants with 85% warrant coverage. Mr. Rose is the Company’s Chief Executive Officer, President, Treasurer and a Director.
86
(21) Mark E. Hooper is the trustee of the Mark E. Hooper Revocable Living Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Mark E. Hooper has sole voting and disposition power over such shares being offered under this prospectus.
(22) Stephen J. Linder is the trustee of the Stephen J. Linder Trust No. 1 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust Stephen J. Linder has sole voting and disposition power over such shares being offered under this prospectus.
(23) John Sudduth is the General Partner of Golden Hill Investments, LLP and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Golden Hill. John Sudduth has shared voting and disposition power over such shares being offered under this prospectus.
(24) Benjamin P. Licht is the trustee of the Muriel F. Licht Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Benjamin P. Licht has sole voting and disposition power over such shares being offered under this prospectus.
(25) Stacy Schwartz is the trustee of the First Trust Company of Onaga FBO Kenneth RJ Rettig and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Stacy Schwartz has shared voting and disposition power over such shares being offered under this prospectus.
(26) David G. Pendell is the trustee of the David G. Pendell Revocable Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. David G. Pendell has sole voting and disposition power over such shares being offered under this prospectus.
(27) Joyce Matson is the trustee of the First Trust Company of Onaga FBO George A. Shaw and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Joyce Matson has sole voting and disposition power over such shares being offered under this prospectus.
(28) Steven Shroyer is the trustee of the Lexy and Steven Shroyer Family Trust 2004 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Steven Shroyer has sharedvoting and disposition power over such shares being offered under this prospectus.
(29) Robert F. Moyer is the trustee of theRobert F. Moyer Revocable Trust dated 9/20/2011 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Robert F. Moyer has sole voting and disposition power over such shares being offered under this prospectus.
(30) Bryan A Weber is the manager of Shannondoah, LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Shannondoah, LLC. Bryan Weber has voting and disposition power over such shares being offered under this prospectus.
(31) John M O’Neill is the trustee of the Traut Family Trust UTD 7/3/13 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. John M O’Neill has sole voting and disposition power over such shares being offered under this prospectus.
(32) Arnold A Allemang is the trustee of the Arnold Avery Allemang Revocable Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Arnold A Allemang has sole voting and disposition power over such shares being offered under this prospectus.
(33) Mitchell H Bock is the trustee of the Laurie L. Bock Administrative Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Mitchell H Bock has sole voting and disposition power over such shares being offered under this prospectus.
(34) Michael L. Ray is the manager of LMRAY, LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by LMRAY, LLC. Michael L. Ray has shared voting and disposition power over such shares being offered under this prospectus.
(35) Charles W. Brown is the manager of Spinnaker Brown, LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Spinnaker Brown, LLC. Charles W. Brown has shared voting and disposition power over such shares being offered under this prospectus.
(36) Paul Ng is the trustee of the Who Dat Nation Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Paul Ng has sole voting and disposition power over such shares being offered under this prospectus.
(37) Steven Spector is the manager of N. Rich Holdings, LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by N. Rich Holdings, LLC. Steven Spector has shared voting and disposition power over such shares being offered under this prospectus.
(38) Steven Spector is the manager of M. Rich Holdings, LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by M. Rich Holdings, LLC. Steven Spector has shared voting and disposition power over such shares being offered under this prospectus.
87
(39) David P. Stern is the trustee of the David P. Stern Trust UA 12-19-200 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. David P. Stern has sole voting and disposition power over such shares being offered under this prospectus.
(40) Tomas R. Manson is the trustee of the Tomas R. Manson Family Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Tomas R. Manson has sole voting and disposition power over such shares being offered under this prospectus.
(41) Brian G. Martindale is the trustee of the Brian G. Martindale Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Brian G. Martindale has sole voting and disposition power over such shares being offered under this prospectus.
(42) William B. Larson is the manager of OK Enterprises LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by OK Enterprises LLC. William B. Larson has sharedvoting and disposition power over such shares being offered under this prospectus.
(43) Mary V. Murfey is the trustee of the Mary V. Murfey Family Trust #2 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Mary V. Murfey has sole voting and disposition power over such shares being offered under this prospectus.
(44) Brian P. Vitale is the manager of Galt Asset Management and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Galt Asset Management. Brian P. Vitale has sharedvoting and disposition power over such shares being offered under this prospectus.
(45) Michael R. Stevens is the manager of Stevens Financial Group, LLC and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Stevens Financial Group, LLC. Michael R. Stevens has shared voting and disposition power over such shares being offered under this prospectus.
(46) Joyce Matson is the trustee of the First Trust Company of Onaga FBO James M Rajewski IRA#TW000918 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Joyce Matson has sole voting and disposition power over such shares being offered under this prospectus.
(47) David A. Johnson is the trustee of the David Ari Johnson and Gabrielle Johnson Revocable Trust 1-13-14 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. David A. Johnson has sharedvoting and disposition power over such shares being offered under this prospectus.
(48) Timothy Long is the trustee of the Timothy Long Living Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Timothy Long has sole voting and disposition power over such shares being offered under this prospectus.
(49) Andrew Hoffman is the manager of Hoffman Investment Company and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by Hoffman Investment Company. Andrew Hoffman has sharedvoting and disposition power over such shares being offered under this prospectus.
(50) Russell S. Deane is the trustee of the Russell S. Deane Trust 1/4/2008 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Russell S. Deane has sole voting and disposition power over such shares being offered under this prospectus
(51) Michael D. Bierlein is the trustee of the Michael D. Bierlein Intervivos Trust and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Michael D. Bierlein has sole voting and disposition power over such shares being offered under this prospectus.
(52) Lillian Elizabeth Deane is the trustee of the Lillian Elizabeth Deane Trust dated July 2, 1993 and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Lillian Elizabeth Deane has sole voting and disposition power over such shares being offered under this prospectus.
(53) Joyce Matson is the trustee of the First Trust Company of Onaga FBO Robert L Hartsock and as such, may be deemed to be the beneficial owner of all of the shares of common stock owned by the trust. Joyce Matson has shared voting and disposition power over such shares being offered under this prospectus.
88
In the discussion that follows, we have summarized selected provisions of our Articles of Incorporation, as amended relating to our capital stock. This summary is not complete. This discussion is subject to the relevant provisions of the Michigan Business Corporation Act and is qualified in its entirety by reference to our Articles of Incorporation, as amended and our Bylaws. You should read the provisions of our Articles of Incorporation, as amended and our Bylaws as currently in effect for provisions that may be important to you.
Common Stock
As of December 31, 2015, we had 25,000,000 shares of common stock, no stated par value per share, authorized for issuance and 836,544 shares of common stock issued and outstanding.
In accordance with the terms of our Bylaws, each outstanding share of capital stock is entitled to one vote on each matter submitted to a vote, unless otherwise provided in our Articles of Incorporation, as amended. A vote may be cast either orally or in writing. When an action, other than the election of Directors, is to be taken by vote of the stockholders, it shall be authorized by a majority of the votes cast by the holders of shares of capital stock entitled to vote thereon, unless a plurality is required by the Articles of Incorporation, as amended or by the laws of the State of Michigan. Except as otherwise provided by our Articles of Incorporation, as amended, the Directors shall be elected by a plurality of the votes cast at an election of Directors. Holders of common stock do not have cumulative voting rights in the election of Director.
According to our Articles of Incorporation, as amended, and Bylaws, any action required or permitted to be taken at an annual or special meeting of stockholders may be taken without a meeting, without prior notice, and without a vote, if consents in writing, setting forth the action so taken, are signed by the holders of outstanding shares having not less than the minimum number of votes that would be necessary to authorize or take the action at a meeting at which all shares entitled to vote on the action were present and voted.
Each stockholder is entitled to receive dividends, as the Board of Directors may, from time to time, declare in a manner and upon the terms and conditions provided by the laws of the State of Michigan and our Articles of Incorporation, as amended.
Holders of the common stock have no preemptive or other preferential rights to purchase additional shares of any class of the Company’s capital stock in subsequent stock offerings.
Preferred Stock
As of December 31, 2015, the Company has 8,000,000 shares of preferred stock authorized for issuance, no stated par value per share. As of December 31, 2015, 3,000,000 preferred shares have been designated as Series A Preferred Stock, of which 1,800,696 shares of Series A Preferred Stock are issued and are outstanding, 1,500,000 shares have been designated as Series B Preferred Stock, of which 269,987 are issued and outstanding, and the remaining 3,500,000 authorized shares are shares of blank check preferred stock which may be issued in one or more series, each of such series to have such designations, powers, preferences, and relative participating, optional, or other rights, and such qualifications, limitations, or restrictions, as may be stated in a resolution or resolutions providing for the issue of such series adopted by the Board of Directors.
Series A Convertible Preferred Stock
Pursuant to the Series A Designations, the Company’s Series A convertible preferred stock have the following powers, preferences, rights, qualifications, limitations and restrictions:
Upon any liquidation, dissolution or winding up of the Company, either voluntary or involuntary, the holders of the Series A convertible preferred stock shall be entitled to receive, prior and in preference to any distribution of any of the assets of the Company to the holders of Common Stock by reason of their ownership of such stock, an amount per share for each share of Series A convertible preferred stock held by them equal to (i) the Liquidation Preference, as such term is defined in the Series A Designations, specified for such share of Series A convertible preferred stock less (ii) all dividends (if any) paid on such share of Series A convertible preferred stock.
With respect to any matter presented to the stockholders of the Company for their action or consideration at any meeting of stockholders of the Company, or by written consent of stockholders in lieu of a meeting, each holder of Series A convertible preferred stock shall be entitled to cast the number of votes equal to the number of whole shares of Common Stock into which
89
the shares of Series A convertible preferred stock held by such holder are convertible as of the record date for determining stockholders entitled to vote on such matter.
Each share of Series A convertible preferred stock shall be convertible, at the option of the holder thereof, at any time after the date of issuance of such share, into that number of fully paid, non-assessable shares of Common Stock determined by dividing the Series A Original Issue Price (which is twelve dollars ($12.00) per share, subject to certain adjustments from time to time for recapitalizations), by the Conversion Price (which is twelve dollars ($12.00), subject to adjustment from time to time for recapitalizations or as otherwise set forth in the Series A Designations). The conversion price of the Series A convertible preferred stock is subject to adjustments pursuant to the occurrence of certain specified events. The number of shares of Common Stock into which each share of Series A Preferred Stock may be converted is the “Conversion Rate”.
Furthermore, the Company has the right to effect a mandatory conversion of each share of Series A convertible preferred stock outstanding into fully paid, non-assessable shares of Common Stock at the then effective Conversion Rate, for such share upon the listing by the Company of its Common Stock on a Qualified National Exchange, defined in the Series A Designations as a securities exchange registered with the SEC under Section 6(a) of the Exchange Act, as well as the over the counter markets administered by the OTCQX and the OTCQB and their successor entities. The OTCBB is not considered a qualified national exchange and therefore listing on the OTCBB is not sufficient to trigger the Company’s ability to convert the Series A convertible preferred stock.
Upon any liquidation, dissolution or winding up of the Company, either voluntary or involuntary, the holders of the Series A Preferred Stock shall be entitled to receive, prior and in preference to any distribution of any of the assets of the Company to the holders of the common stock by reason of their ownership of such stock, an amount per share for each share of Series A Preferred Stock held by them equal to (i) the Liquidation Preference, as such term is defined in the Series A Designations, specified for such share of Series A Preferred Stock less (ii) all dividends (if any) paid on such share of Series A Preferred Stock.
With respect to any matter presented to the stockholders of the Company for their action or consideration at any meeting of stockholders of the Company, or by written consent of stockholders in lieu of a meeting, each holder of Series A Preferred Stock shall be entitled to cast the number of votes equal to the number of whole shares of common stock into which the shares of Series A Preferred Stock held by such holder are convertible as of the record date for determining stockholders entitled to vote on such matter.
Series B Convertible Preferred Stock
Pursuant to the Series B Designations, the Company’s Series B convertible preferred stock has the following powers, preferences, rights, qualifications, limitations and restrictions:
Series B Preferred Stock shall rank (a) senior to all other equity or equity equivalent securities of the Company other than those securities which are explicitly senior orpari passu in rights and liquidation preference to the Series B Preferred Stock (“Junior Securities”) and (b)pari passu with the Company’s Series A convertible preferred stock and (c) junior to all other series of the Company’s preferred stock that are explicitly senior in rights and liquidation preference which, as of the date hereof, is none.
In the event of any liquidation, dissolution or winding up of the Company, either voluntary or involuntary, holders of the Series B Preferred Stock shall be entitled to receive, prior and in preference to any distribution of any of the assets of the Company to the holders of Junior Securities, an amount per share for each share of Series B Preferred held by them equal to $16.
With respect to any matter presented to the stockholders of the Company for their action or consideration at any meeting of stockholders of the Company, or by written consent of stockholders in lieu of a meeting, each holder of Series B convertible preferred stock shall be entitled to cast the number of votes equal to the number of whole shares of Common Stock into which the shares of Series B convertible preferred stock held by such holder are convertible as of the record date for determining stockholders entitled to vote on such matter.
Each share of Series B convertible preferred stock shall be convertible, at the option of the holder thereof, at any time after the date of issuance of such share, into that number of fully paid, non-assessable shares of Common Stock determined by dividing the Series B Original Issue Price (which is currently $16, subject to adjustments), by the Conversion Price (which is currently $16, subject to adjustments), as such terms are further defined in the Series B Designations. The conversion price of the Series B convertible preferred stock is subject to adjustments pursuant to the occurrence of certain specified events.
90
Furthermore, the Company shall have the right to effect the conversion of each share then currently outstanding into shares of common stock upon the listing by the Company of its common stock on a Qualified National Exchange (a securities exchange registered with the SEC under Section 6(a) of the Exchange Act, as well as the OTCQX® and the OTCQB®markets, and their successor-entities maintained by the OTC Markets Group, Inc.). Notwithstanding the foregoing, the Series B Preferred Stock shall not be subject to such mandatory conversion until all outstanding Convertible Securities (any evidences of indebtedness, shares or other securities convertible into or exchangeable for common stock or preferred stock) are also converted into common stock. The OTCBB is not considered a qualified national exchange and therefore listing on the OTCBB is not sufficient to trigger the Company’s ability to convert the Series B convertible preferred stock.
Warrants
In conjunction with previous loans to the Company in 2009 and 2012 by Michael Knox, warrants to purchase 5,000 and 6,000 shares of common stock, respectively, were issued to Mr. Knox. In conjunction with the sale of Series B Preferred Stock in April and June of 2015, an additional 224,897 warrants to purchase shares of common stock were issued. In connection with the December Notes and December Warrants issuance, warrants to purchase 20,625 shares of common stock were issued. In 2014, warrants to purchase 1,072,720 shares of Series A Convertible Preferred Stock were issued in conjunction with the sale of Series A Convertible Preferred stock. All of these warrants are vested. These warrants have exercise prices ranging $8.00 to $16.00 per share, with a weighted average purchase price of $12.67 per share. These warrants expire on various dates ranging from July 2019 to October 2027.
As of December 31, 2015, outstanding warrants were as follows:
Holder |
| Issue Date |
| Warrant Shares |
| Expiration Date |
| Exercise Price |
| Total Vested on 6/30/2015 |
| Total Not Vested on 6/30/2015 | |
Warrants to Purchase Common Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
Michael R. Knox |
| 10/8/2012 |
| 5,000 |
| 10/8/2017 |
| $ | 12.00 |
| 5,000 |
| — |
Michael R. Knox |
| 7/1/2009 |
| 6,000 |
| 7/1/2019 |
| $ | 8.00 |
| 6,000 |
| — |
Jones Network, LP |
| 4/21/2015 |
| 7,969 |
| 4/21/2022 |
| $ | 16.00 |
| 7,969 |
| — |
George Cardoza |
| 4/22/2015 |
| 2,125 |
| 4/22/2022 |
| $ | 16.00 |
| 2,125 |
| — |
William Robison |
| 4/22/2015 |
| 26,563 |
| 4/22/2022 |
| $ | 16.00 |
| 26,563 |
| — |
Steven Purcell |
| 4/23/2015 |
| 5,313 |
| 4/23/2022 |
| $ | 16.00 |
| 5,313 |
| — |
James D. Chambers |
| 4/24/2015 |
| 5,100 |
| 4/24/2022 |
| $ | 16.00 |
| 5,100 |
| — |
Golden Hills Investments, LLLP |
| 4/24/2015 |
| 13,281 |
| 4/24/2022 |
| $ | 16.00 |
| 13,281 |
| — |
Muriel F. Licht Trust |
| 4/24/2015 |
| 2,656 |
| 4/24/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Hewit Hawn |
| 4/24/2015 |
| 266 |
| 4/24/2022 |
| $ | 16.00 |
| 266 |
| — |
First Trust Company of Onaga FBO Kenneth RJ Rettig |
| 4/27/2015 |
| 956 |
| 4/27/2022 |
| $ | 16.00 |
| 956 |
| — |
Jones Extended Family Trust |
| 4/27/2015 |
| 2,656 |
| 4/27/2022 |
| $ | 16.00 |
| 2,656 |
| — |
David G. Pendell Revocable |
| 4/28/2015 |
| 2,656 |
| 4/28/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Edelgard Traut |
| 4/28/2015 |
| 850 |
| 4/28/2022 |
| $ | 16.00 |
| 850 |
| — |
First Trust Company of Onaga FBO George A. Shaw |
| 4/28/2015 |
| 2,656 |
| 4/28/2022 |
| $ | 16.00 |
| 2,656 |
| — |
John M. O’Neill |
| 4/28/2015 |
| 850 |
| 4/28/2022 |
| $ | 16.00 |
| 850 |
| — |
L.D. Sipes, Jr. |
| 4/28/2015 |
| 8,500 |
| 4/28/2022 |
| $ | 16.00 |
| 8,500 |
| — |
Lexy and Steven Shroyer Family Trust 2004 |
| 4/28/2015 |
| 7,969 |
| 4/28/2022 |
| $ | 16.00 |
| 7,969 |
| — |
Robert Moyer |
| 4/28/2015 |
| 3,985 |
| 4/28/2022 |
| $ | 16.00 |
| 3,985 |
| — |
Samuel H. Jones Jr. |
| 4/28/2015 |
| 5,313 |
| 4/28/2022 |
| $ | 16.00 |
| 5,313 |
| — |
91
Holder |
| Issue Date |
| Warrant Shares |
| Expiration Date |
| Exercise Price |
| Total Vested on 6/30/2015 |
| Total Not Vested on 6/30/2015 | |
John and Elaine D. Cavanna JTWROS |
| 4/29/2015 |
| 1,381 |
| 4/29/2022 |
| $ | 16.00 |
| 1,381 |
| — |
Shannondoah, LLC |
| 4/29/2015 |
| 1,275 |
| 4/29/2022 |
| $ | 16.00 |
| 1,275 |
| — |
Traut Family Trust UTD 7/3/13 |
| 4/29/2015 |
| 850 |
| 4/29/2022 |
| $ | 16.00 |
| 850 |
| — |
Ravi Shanker |
| 4/29/2015 |
| 850 |
| 4/29/2022 |
| $ | 16.00 |
| 850 |
| — |
David Fuhrhop and Marcia L. Furhop |
| 4/29/2015 |
| 495 |
| 4/29/2022 |
| $ | 16.00 |
| 495 |
| — |
Thomas Bombeck IV |
| 4/29/2015 |
| 2,550 |
| 4/29/2022 |
| $ | 16.00 |
| 2,550 |
| — |
Arnold Avery Allemang |
| 4/29/2015 |
| 13,281 |
| 4/29/2022 |
| $ | 16.00 |
| 13,281 |
| — |
Steven & Carisa Jones 401K Plan & Trust |
| 4/29/2015 |
| 2,656 |
| 4/29/2022 |
| $ | 16.00 |
| 2,656 |
| — |
George J. and/or Beverly A. Biltz |
| 4/29/2015 |
| 3,188 |
| 4/29/2022 |
| $ | 16.00 |
| 3,188 |
| — |
Philip Rose |
| 4/29/2015 |
| 2,656 |
| 4/29/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Laurie L. Bock Administrative Trust |
| 4/29/2015 |
| 797 |
| 4/29/2022 |
| $ | 16.00 |
| 797 |
| — |
Kay B. Coin |
| 4/29/2015 |
| 1,329 |
| 4/29/2022 |
| $ | 16.00 |
| 1,329 |
| — |
LMRAY, LLC |
| 4/29/2015 |
| 399 |
| 4/29/2022 |
| $ | 16.00 |
| 399 |
| — |
Spinnaker Brown, LLC |
| 4/29/2015 |
| 1,435 |
| 4/29/2022 |
| $ | 16.00 |
| 1,435 |
| — |
Who Dat Nation Trust |
| 4/29/2015 |
| 1,496 |
| 4/29/2022 |
| $ | 16.00 |
| 1,496 |
| — |
Arthur Baumann |
| 4/29/2015 |
| 691 |
| 4/29/2022 |
| $ | 16.00 |
| 691 |
| — |
N. Rich Holdings, LLC |
| 4/29/2015 |
| 1,700 |
| 4/29/2022 |
| $ | 16.00 |
| 1,700 |
| — |
M. Rich Holdings, LLC |
| 4/29/2015 |
| 5,100 |
| 4/29/2022 |
| $ | 16.00 |
| 5,100 |
| — |
David P. Stern Trust UA 12-19-2001 |
| 4/29/2015 |
| 425 |
| 4/29/2022 |
| $ | 16.00 |
| 425 |
| — |
Arnold S. and Bette G. Hoffman |
| 4/29/2015 |
| 2,656 |
| 4/29/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Tomas R. Manson Family Trust |
| 4/29/2015 |
| 1,275 |
| 4/29/2022 |
| $ | 16.00 |
| 1,275 |
| — |
Thomas and Carmelita B. Karacic |
| 4/29/2015 |
| 2,656 |
| 4/29/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Erik J. Falconer |
| 4/29/2015 |
| 2,550 |
| 4/29/2022 |
| $ | 16.00 |
| 2,550 |
| — |
Brian G. Martindale Trust |
| 4/29/2015 |
| 2,720 |
| 4/29/2022 |
| $ | 16.00 |
| 2,720 |
| — |
OK Enterprises |
| 4/30/2015 |
| 797 |
| 4/30/2022 |
| $ | 16.00 |
| 797 |
| — |
Mary V. Murfey Family Trust #2 |
| 4/30/2015 |
| 3,400 |
| 4/30/2022 |
| $ | 16.00 |
| 3,400 |
| — |
Galt Asset Management |
| 4/30/2015 |
| 2,656 |
| 4/30/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Paul Anthony Radomski |
| 4/30/2015 |
| 8,500 |
| 4/30/2022 |
| $ | 16.00 |
| 8,500 |
| — |
Stevens Financial Group, LLC |
| 4/30/2015 |
| 26,563 |
| 4/30/2022 |
| $ | 16.00 |
| 26,563 |
| — |
First Trust Company of Onaga FBO James M Rajewski IRA#TW000918 |
| 4/30/2015 |
| 1,993 |
| 4/30/2022 |
| $ | 16.00 |
| 1,993 |
| — |
David Ari Johnson and Gabrielle Johnson Revocable Trust 1-13-14 |
| 4/30/2015 |
| 1,329 |
| 4/30/2022 |
| $ | 16.00 |
| 1,329 |
| — |
Timothy Long Living Trust (no paperwork) |
| 4/30/2015 |
| 531 |
| 4/30/2022 |
| $ | 16.00 |
| 531 |
| — |
Thomas W. Rouse |
| 4/30/2015 |
| 1,036 |
| 4/30/2022 |
| $ | 16.00 |
| 1,036 |
| — |
Morgan Duke |
| 4/30/2015 |
| 2,656 |
| 4/30/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Howard Moskow |
| 4/30/2015 |
| 1,329 |
| 4/30/2022 |
| $ | 16.00 |
| 1,329 |
| — |
Hoffman Investment Company |
| 4/30/2015 |
| 2,656 |
| 4/30/2022 |
| $ | 16.00 |
| 2,656 |
| — |
Russell S. Deane Trust 1/4/2008 |
| 4/30/2015 |
| 1,063 |
| 4/30/2022 |
| $ | 16.00 |
| 1,063 |
| — |
Scott Lizenby (assumed internal 53 transfer – no paperwork) |
| 4/30/2015 |
| 340 |
| 4/30/2022 |
| $ | 16.00 |
| 340 |
| — |
92
Holder |
| Issue Date |
| Warrant Shares |
| Expiration Date |
| Exercise Price |
| Total Vested on 6/30/2015 |
| Total Not Vested on 6/30/2015 | |
Michael D. Bierlein Intervivos Trust |
| 4/30/2015 |
| 2,790 |
| 4/30/2022 |
| $ | 16.00 |
| 2,790 |
| — |
Lillian Elizabeth Deane Trust dated July 2, 1993 |
| 4/30/2015 |
| 531 |
| 4/30/2022 |
| $ | 16.00 |
| 531 |
| — |
Jennifer Dana Dean |
| 4/30/2015 |
| 850 |
| 4/30/2022 |
| $ | 16.00 |
| 850 |
| — |
First Trust Company of Onaga FBO Robert L Hartsock |
| 4/30/2015 |
| 1,275 |
| 4/30/2022 |
| $ | 16.00 |
| 1,275 |
| — |
Robert and Carlita Gasparini |
| 4/30/2015 |
| 1,329 |
| 4/30/2022 |
| $ | 16.00 |
| 1,329 |
| — |
Colin Cronin |
| 5/26/2015 |
| 2,125 |
| 5/26/2022 |
| $ | 16.00 |
| 2,125 |
| — |
Lawrence T. Drzal |
| 5/26/2015 |
| 510 |
| 5/26/2022 |
| $ | 16.00 |
| 510 |
| — |
Rodney Boulanger |
| 6/26/2015 |
| 1,563 |
| 6/26/2022 |
| $ | 16.00 |
| 1,563 |
| — |
James R. DeVore and Joan Mobley-DeVore |
| 6/30/2015 |
| 5,000 |
| 6/30/2022 |
| $ | 16.00 |
| 5,000 |
| — |
Steven Jones |
| 12/31/2015 |
| 9,375 |
| 12/31/2020 |
| $ | 8.00 |
| 9,375 |
|
|
Arnold Allemang |
| 12/31/2015 |
| 9,375 |
| 12/31/2020 |
| $ | 8.00 |
| 9,375 |
|
|
David Pendell |
| 12/31/2015 |
| 1,875 |
| 12/31/2020 |
| $ | 8.00 |
| 1,875 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 256,522 |
|
|
|
|
|
| 256,522 |
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants to Purchase Series A Preferred Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
Hiroyuki Fukushima |
| 3/31/2014 |
| 250 |
| 1/15/2024 |
| $ | 12.00 |
| 250 |
| — |
Inwhan Do |
| 3/31/2014 |
| 250 |
| 1/15/2024 |
| $ | 12.00 |
| 250 |
| — |
Peter S. Bosanic & Lisa Kendzioski Bosanic |
| 1/15/2014 |
| 625 |
| 1/15/2024 |
| $ | 12.00 |
| 625 |
| — |
John W. Dourjalian |
| 1/15/2014 |
| 1,041 |
| 1/15/2024 |
| $ | 12.00 |
| 1,041 |
| — |
Colin D. Cronin |
| 1/15/2014 |
| 1,767 |
| 1/15/2024 |
| $ | 12.00 |
| 1,767 |
| — |
Paul Nordstrom |
| 1/15/2014 |
| 2,121 |
| 1/15/2024 |
| $ | 12.00 |
| 2,121 |
| — |
POSCO |
| 3/31/2014 |
| 50,000 |
| 1/15/2024 |
| $ | 12.00 |
| 50,000 |
| — |
XGS II, LLC |
| 1/15/2014 |
| 83,333 |
| 1/15/2024 |
| $ | 12.00 |
| 83,333 |
| — |
SVIC No. 15 (Samsung) |
| 1/15/2014 |
| 100,000 |
| 1/15/2024 |
| $ | 12.00 |
| — |
| 100,000 |
Aspen Advanced Opportunity Fund, LP |
| 1/15/2014 |
| 833,333 |
| 1/15/2024 |
| $ | 12.00 |
| 833,333 |
| — |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Series A Preferred Warrants |
|
|
| 1,072,720 |
|
|
|
|
|
| 972,720 |
| 100,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Grand Total Warrants |
|
|
| 256,522 |
|
|
|
|
|
| 1,208,617 |
| 100,000 |
Stock Options
As of December 31, 2015, the Company had granted options to purchase up to 419,750 shares of common stock.
In 2007, the Company implemented a Stock Option Plan and initially reserved 75,000 shares of common stock to cover stock options that might be issued under the Stock Option Plan. As of March 31, 2014 the number of shares reserved for issuance under the plan was 600,000 shares.
As of December 31, 2013, stock options to purchase a total of 152,500 shares at prices ranging from $8.00 to $13.20 per share had been granted to Company employees and Directors, with expiration dates ranging from March 2014 to June 2021, with a weighted average purchase price of $12.13 per share.
During 2014, additional stock options to purchase 295,000 shares were granted to employees, bringing the total stock options outstanding under the Stock Option Plan to 464,750 at the end of the year. During 2015, certain options expired and other options were granted, bringing the total stock options outstanding as of December 31, 2015 to 419,750.
93
The following table shows the number of stock options issued and outstanding as of December 29, 2015:
Holder |
| Issue Date |
| Total Options |
| Exp. Date |
| Exercise Price |
| Amount Vested 12/30/2015 |
| Amount Non - Exercisable 12/30/2015 | |
Philip Rose |
| 1/6/2014 |
| 160,000 |
| 1/6/2022 |
| $ | 12.00 |
| 63,331 |
| 96,669 |
Philip Rose |
| 1/6/2014 |
| 40,000 |
| 1/6/2022 |
| $ | 12.00 |
| — |
| 40,000 |
Philip Rose |
| 1/6/2014 |
| 20,000 |
| 1/6/2022 |
| $ | 12.00 |
| — |
| 20,000 |
Michael R. Knox |
| 6/1/2013 |
| 50,000 |
| 6/1/2021 |
| $ | 13.20 |
| 35,000 |
| 15,000 |
Liya Wang |
| 6/1/2013 |
| 30,000 |
| 6/1/2021 |
| $ | 12.00 |
| 20,000 |
| 10,000 |
Robert Privette |
| 6/1/2013 |
| 20,000 |
| 6/1/2021 |
| $ | 12.00 |
| 13,333 |
| 6,667 |
Scott Murray |
| 12/1/2007 |
| 10,000 |
| 12/1/2017 |
| $ | 8.00 |
| 10,000 |
| — |
Scott Murray |
| 6/1/2013 |
| 10,000 |
| 6/1/2021 |
| $ | 12.00 |
| 6,667 |
| 3,333 |
Corinne Lyon |
| 6/1/2013 |
| 10,000 |
| 6/1/2021 |
| $ | 12.00 |
| 6,667 |
| 3,333 |
Arnold Allemang |
| 6/1/2013 |
| 3,750 |
| 6/1/2021 |
| $ | 12.00 |
| 3,750 |
| — |
Steven Jones |
| 6/1/2013 |
| 2,500 |
| 6/1/2021 |
| $ | 12.00 |
| 2,500 |
| — |
Arnold Allemang |
| 4/1/2014 |
| 1,750 |
| 3/31/2022 |
| $ | 12.00 |
| 1,750 |
| — |
Arnold Allemang |
| 4/1/2014 |
| 9,000 |
| 3/31/2022 |
| $ | 12.00 |
| 438 |
| 8,562 |
Steven Jones |
| 4/1/2014 |
| 1,750 |
| 3/31/2022 |
| $ | 12.00 |
| 1,750 |
| — |
Steven Jones |
| 4/1/2014 |
| 9,000 |
| 3/31/2022 |
| $ | 12.00 |
| 438 |
| 8,562 |
Ravi Shanker |
| 4/1/2014 |
| 12,000 |
| 3/31/2022 |
| $ | 12.00 |
| 3,750 |
| 8,250 |
Albert Kim |
| 10/19/2015 |
| 20,000 |
| 10/19/2023 |
| $ | 12.00 |
| — |
| 20,000 |
Percy Chinoy |
| 10/19/2015 |
| 10,000 |
| 10/19/2023 |
| $ | 12.00 |
| — |
| 10,000 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total Options |
|
|
| 419,750 |
|
|
|
|
|
| 169,374 |
| 250,376 |
The Company intends to continue to use stock options as one method of attracting and retaining key employees and expects that additional shares will be reserved to cover stock options that might be issued under the plan.
Secured Convertible Notes
On March 18, 2013, the Company entered into a series of agreements with Knox and AAOF. Under these agreements, Knox and AAOF agreed to provide financing to the Company in the form of secured convertible notes (the “Notes”) as well as optional lease financing. The agreements provided for Knox to lend the Company a sum of $700,000, to be funded by converting advances he had made under a revolving line of credit, and for AAOF to lend the Company a minimum of $2.5 million and, at the option of AAOF, a maximum of $5 million, with a provision that up to 50% of the AAOF funding could be funded under lease financing arrangements.
On July 12, 2013, the agreements described above were amended and restated to provide for expanded financing commitments from AAOF as well as a related entity called XGS II, LLC (“XGS II”). Under the terms of these amended and restated agreements, the financing commitments from AAOF were increased to a minimum of $6 million and, at the option of AAOF, a maximum of $10 million, of which a maximum of $3 million could be provided in the form of lease financing. Also under these amended agreements, XGS II agreed to provide a minimum of $250,000 and, at the option of XGS II, a maximum of $750,000. All of the funding with the exception of the Knox financing, was scheduled according to minimum financing commitments on calendar dates ranging from the time the agreements were executed to a period of eighteen months into the future.
With the exception of any lease financing, the financing provided by Knox, AAOF, and XGS II takes the form of purchases of Notes with the following identical key terms and provisions:
• Each Note bears interest at a rate of 12% per annum and has a maturity date of March 18, 2018;
• The Notes have provisions providing an option to the Company to accrue the interest payments on the Notes through December 31, 2014 and add the accrued interest to the principal of the Notes;
• After December 31, 2014 the holder, at their option, can choose to receive ongoing interest payments: (a) in the form of cash; (b) receive all or a portion of currently due interest in the form of shares of Series A Preferred Stock at a price
94
per share equal to the then effective Series A Original Issue Price as defined in the Series A Designations; (c) receive all or a portion of currently due interest payments in the form of any other series of preferred stock outstanding at the time interest is due; or (d) elect to accrue such interest and add it to the balance of the Note;
• The Notes are secured by all of the assets, both tangible and intangible, of the Company;
• The Notes have provisions allowing conversion into shares of Series A Preferred Stock (or such other form of preferred stock as may be outstanding at that time) at the option of the holder, in the event that the Company chooses to repay the Notes prior to maturity; and
• The Notes contain various affirmative and negative covenants that provide for various reporting, information, notice, maintenance of business, insurance, audits and reappraisal requirements. These covenants also prohibit the Company from issuing new secured debt that would have security or liquidation provisions senior to or equivalent to the Notes, and these covenants also prohibit the issuance of dividends or stock redemptions, creation of subsidiaries, certain transactions with subsidiaries, or transfer of assets without the permission of the holder.
On December 31, 2013, a number of agreements related to financing transactions had been signed, with signature pages escrowed, pending final approval by legal representatives of Samsung Ventures. On January 15, 2014 these agreements were completed. Under these agreements:
• The Company sold $3 million of Notes to Samsung Ventures. The Notes had terms and conditions that were substantially similar to those previously issued to Knox, AAOF and XGS II. In connection with the sale of the Notes, the Company issued to Samsung Ventures a total of 100,000 warrants with a term of 4 years that are exercisable into shares of Series A Preferred Stock at a price of $12.00 per share. These warrants vest in four installments on the first, second, third and fourth anniversary of the issuance of the warrants according to the following formula: On each anniversary, the warrant vests according to the ratio of (a) total Samsung Ventures payments to the Company during the previous twelve-month period divided by (b) 250. Thus, the warrants shall vest in four annual increments, proportionately in relation to the first $25 million, or a fraction thereof, that Samsung Ventures pays, in cash, to the Company over the four years commencing on January 15, 2014. Samsung Ventures payments are defined as future total cash payments, including licensing, royalties and product purchases but not joint development or the purchase of Notes.
• We consolidated the Notes previously issued to AAOF and XGS II, together with accrued interest, into a single new Note for each lender, with terms and conditions identical to the Notes issued to Samsung Ventures and exchanged existing warrants to purchase 447,916 shares of Series A Preferred Stock for new warrants to purchase 916,666 shares of Series A Preferred Stock. Because the cash flows, including fees and other instruments issued, associated with the new instruments exceeded the cash flows of the old instruments by more than 10%, in accordance with ASC 450, the modification of the Notes has been accounted for as a debt extinguishment. Accordingly, the difference of $3,513,770 between the carrying amounts of the original Notes and the fair value of the newly issued Notes plus the fair value of the additional warrants was recorded as a loss on extinguishment.
• We transferred all of our intellectual property into a newly created, wholly owned subsidiary of the Company called XG Sciences IP, LLC, a Michigan limited liability company.
• We revised the terms and amounts related to the timing of future investments from AAOF and XGS II by increasing the XGS II optional funding level to $1 million and extending the optional financing periods for both AAOF and XGS II through the end of 2014.
• A new provision was added granting us (as the borrower) the right to force conversion of all of the Notes into Series A Preferred Stock at any time after we raise an aggregate of $15 million in new equity.
• We revised certain provisions in our Bylaws to allow for special stockholder meetings to be called by holders of ten percent (10%) or more of the Company’s stock and to clarify certain voting provisions.
• We further amended and restated the Series A Designations to clarify the conditions relating to mandatory conversions into common stock upon a listing of the Company’s common stock on a Qualified National Exchange (as defined in the Series A Designations) and to add a provision that would exempt holders of Series A Preferred Stock from future mandatory financing participation.
95
Additional funding from AAOF and XGS II occurred during 2014 and, as of December 31, 2014 a total of $14,090,678.90 in principle had been funded that, together with accrued interest, brought the total balance due on the convertible notes to $15,604,429.36.
On April 10, 2015 all of the secured noteholders entered into a Consent and Waiver Agreement which modified the original terms of the Convertible Secured Notes such that all previously issued Notes, together with accrued interest, shall automatically convert into shares of Series A Preferred Stock upon the earlier to occur of 1) the Company raising not less than $12,000,000 (as opposed to $15,000,000) in new funding, and 2) December 31, 2015. All Notes, together with accrued interest, converted to Series A Preferred Stock on December 31, 2015 at a rate of $12 per share.
The following table shows the total balances on the Notes that were converted on December 31, 2015 as well as the resulting number of Series A Preferred Shares issued at that time.
|
| Face Value of Notes |
| Total Interest & Principle as of 12/30/2015 |
| No. of shares converted @ $12/share | ||
AAOF |
| $ | 9,268,906 |
| $ | 11,520,232 |
| 960,019 |
Knox |
|
| 772,998 |
|
| 866,722 |
| 72,227 |
XGS II LLC |
|
| 1,048,775 |
|
| 1,310,068 |
| 109,172 |
Samsung Ventures |
|
| 3,000,000 |
|
| 3,776,518 |
| 314,710 |
Grand Total Notes |
| $ | 14,090,679 |
| $ | 17,473,540 |
| 1,456,128 |
Non-convertible Promissory Notes
On December 31, 2015, the Company issued non-secured, non-convertible promissory notes in an aggregate amount of $550,000 to existing stockholders of the Company who are also accredited investors, along with warrants to purchase 20,625 shares of common stock. The December Notes mature on June 30, 2016 and bear interest at 8% per annum.
Preemptive Rights
On July 5, 2006, the Company entered into a stock redemption agreement with Michael R. Knox, Lawrence T. Drzal, Hiroyuki Fukushima and Inhwan Do whereby, among other things, each holder of common stock of the Company was granted the first right to purchase shares of common stock that may thereafter from time to time be issued in the ratio that the number of shares of common stock they hold at the time of the issue bears to the total number of shares of common stock outstanding. The right shall be deemed waived by any holder of common stock who does not exercise the right and pay for the stock preemptive within thirty days of receipt of a notice in writing from the Company inviting them to exercise the right.
On December 1, 2010, the Company entered into a stock redemption agreement with Hanwha Chemical whereby, among other things, Hanwha Chemical was granted the first right, on equal terms with each of the other stockholders of the Company who had been granted preemptive rights pursuant to a stock redemption agreement with the Company to purchase stock of the Company as of June 8, 2011, to purchase shares of common stock that may thereafter from time to time be issued in the ratio that the number of shares of common stock they hold at the time of the issue bears to the total number of shares of common stock outstanding. The right shall be deemed waived by any holder of common stock who does not exercise the right and pay for the stock preemptive within thirty days of receipt of a notice in writing from the Company inviting them to exercise the right.
On June 8, 2011, the Company entered into a stock redemption agreement with POSCO whereby, among other things, POSCO was granted the first right, on equal terms with each of the other stockholders of the Company who had been granted preemptive rights pursuant to a stock redemption agreement with the Company to purchase stock of the Company as of June 8, 2011, to purchase shares of common stock that may thereafter from time to time be issued in the ratio that the number of shares of common stock they hold at the time of the issue bears to the total number of shares of common stock outstanding. The right shall be deemed waived by any holder of common stock who does not exercise the right and pay for the stock preemptive within thirty days of receipt of a notice in writing from the Company inviting them to exercise the right.
As part of our overall financing agreements with AAOF, we agreed to allow conversion of previously held shares of common stock into Series A Preferred Stock for AAOF affiliates or other stockholders who exercised preemptive rights to purchase Series A Preferred Stock on or before October 31, 2013. The preemptive rights allowed the stockholders to purchase one share of Series A Preferred Stock at a price of $12.00 per share for every two shares of Series A Preferred Stock or common stock
96
owned by the stockholder. Stockholders exercised these preemptive rights to purchase 11,107 shares of Series A Preferred Stock for $133,284. In addition, a total of 174,734 shares of common stock were cancelled on conversion into a like number of shares of Series A Preferred Stock.
On January 15, 2014, as part of our financing agreements with Samsung Ventures, AAOF and XGS II, we agreed to allow all stockholders to purchase one share of Series A Preferred Stock at a price of $12.00 per share for every two shares of Series A Preferred Stock or Common Stock owned by the stockholder. In addition, for every two preemptive shares purchased, the Company issued the stockholder a warrant to purchase one additional share of Series A Preferred Stock with the same terms as the warrants issued to AAOF and XGS II. The Company also agreed to issue warrants with the same terms to those stockholders who exercised preemptive rights in October 2013. These preemptive rights expired on March 31, 2014.
During the quarter ended March 31, 2014, 101,000 shares of Series A Preferred Stock were sold to existing stockholders at a price of $12.00 per share under the January 15, 2014 preemptive rights offering. In addition, Stock Warrants indexed to 56,054 shares of Series A Preferred Stock were issued in conjunction with these stock purchases, including 5,554 warrants related to the preemptive rights exercised in October 2013.
During the six months ended June 30, 2015, 3,100 shares of Series B Preferred Stock were sold to existing stockholders at a price of $16 per share under a preemptive rights offering associated with the April 18, 2015 offering of Series B Preferred Stock Units.
Our Shareholder Agreement grants preemptive rights to shareholders and holders of convertible notes who are parties to the Shareholder Agreement. Pursuant to the terms therein, such shareholders and note holders have the right to purchase their pro rata share of all shareholder stock that the Company may, from time to time, propose to sell, issue, or exchange after the date of the Shareholder Agreement, other than certain excluded stock which includes stock granted to employees or as merger consideration. Each shareholder’s pro rata shares shall be equal to the ratio of (i) the aggregate number of shares of the Company’s common stock on a fully diluted basis, owned by the such shareholder at the time of the delivery of a preemptive rights notice to (ii) the aggregate number of shares of Company’s common stock on a fully diluted basis owned by all of the Company’s shareholders at the time of the delivery of a preemptive rights notice. The Shareholder Agreement’s preemptive rights provision supersedes the preemptive rights granted by the stock redemption agreement.
Michigan Anti-Takeover Law and Charter Provisions
Provisions of our Articles of Incorporation, as amended may delay or discourage transactions involving an actual or potential change in our control or change in our management. Therefore, these provisions could adversely affect the price of our common stock. Our Articles of Incorporation, as amended, permit our Board of Directors to issue up to 5,000,000 shares of blank check preferred stock in one or more series, each of such series to have such designations, powers, preferences, and relative participating, optional, or other rights, and such qualifications, limitations, or restrictions, as may be stated in a resolution or resolutions providing for the issue of such series adopted by the Board of Directors.
Registration Rights
On December 31, 2015, the Company issued promissory notes and warrants to several existing investors in a private placement. Pursuant to the terms of the subscription agreement therein, the Company agreed to use its best efforts to register its shares of common stock on Form S-1.
Pursuant to the terms of the Series B Unit offering, investors therein were granted registration rights with respect to the common stock underlying the shares of Series B Preferred Stock. The Company agreed to use its best efforts to file a registration statement on Form S-1 with the SEC to register shares of common stock within six (6) months of the last closing of the offering of Series B Units, subject to certain extensions of the registration timeframe.
On December 20, 2013, the Company, AAOF, XGS II, LLC and Samsung Ventures executed the Second Amended and Restated Registration Rights Agreement whereby AAOF, XGS II, LLC and Samsung Ventures were granted demand and piggy-back registration rights with respect to any shares of common stock held by such investors or issuable upon conversion, exercise or exchange of any securities owned by the investors.
97
On May 24, 2010, the Company entered into an amended and restated license agreement with MSU whereby the Company, in further consideration of the license, issued 9,543 shares of common stock to MSU and granted to MSU tag-along and piggyback registration rights. The Company agreed to pay all expenses of such registrations.
Transfer Agent and Registrar
The Company does not currently have an independent stock transfer agent. The Company intends to engage an ndependent stock transfer agent prior to the commencement of the primary offering.
98
PLAN OF DISTRIBUTION FOR PRIMARY OFFERING
We intend to engage the services of a sales agent to assist us in selling the shares. If we do engage a sales agent, the sales agent will not purchase or sell any securities offered by this prospectus, nor will it be required to arrange the purchase or sale of any specific number or dollar amount of securities, but must agree to use its best efforts to arrange for the sale of all of the securities offered hereby.
The sales agent may retain other brokers or dealers to act as a sub-agents or selected-dealers on its behalf in connection with the offering. Therefore, we will enter into a securities purchase agreement directly with investors in connection with this offering and we may not sell the entire amount of securities offered pursuant to this prospectus. We intend to pay the sales agent a cash sale agent’s fee equal to up to eight percent (8%) of the gross proceeds of this primary offering.
The following table shows the per share sales agent’s fees that we intend to pay to the sales agent in connection with the sale of the shares offered pursuant to this primary offering, assuming the purchase of all of the shares offered hereby:
|
| Maximum Selling Commissions as a % of Proceeds from Sale of Common Stock |
Shares of Common Stock |
| 8% |
Our obligations to issue and sell securities to the purchasers is subject to the conditions set forth in the subscription agreement, which may be waived by us at our discretion. A purchaser’s obligation to purchase securities is subject to the conditions set forth in the securities purchase agreement as well, which may also be waived. We estimate that the total expenses of the offering, including registration, filing and listing fees, printing fees and legal and accounting expenses, including the sales agent fees, will be approximately $740,000, all of which are payable by us.
The foregoing does not purport to be a complete statement of the terms and conditions of the subscription agreement. If sales of shares offered under this prospectus are made to broker-dealers as principals, we would be required to file a post-effective amendment to the registration statement of which this prospectus is a part. In the post-effective amendment, we would be required to disclose the names of any participating broker-dealers and the compensation arrangements relating to such sales. Once we have engaged the services of a sales agent, we will file a post-effective amendment to this registration statement to identify such sales agent and disclose in greater detail the terms of any compensation arrangement.
All expenses of the registration statement including, but not limited to, legal, accounting, printing and mailing fees are and will be borne by us.
Penny Stock Regulations
You should note that our stock is a penny stock. The SEC has adopted Rule 15g-9 which generally defines “penny stock” to be any equity security that has a market price (as defined) less than $5.00 per share or an exercise price of less than $5.00 per share, subject to certain exceptions. Our securities are covered by the penny stock rules, which impose additional sales practice requirements on broker-dealers who sell to persons other than established customers and “accredited investors”. The term “accredited investor” refers generally to institutions with assets in excess of $5,000,000 or individuals with a net worth in excess of $1,000,000 or annual income exceeding $200,000 or $300,000 jointly with their spouse. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document in a form prepared by the SEC which provides information about penny stocks and the nature and level of risks in the penny stock market. The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction and monthly account statements showing the market value of each penny stock held in the customer’s account. The bid and offer quotations, and the broker-dealer and salesperson compensation information, must be given to the customer orally or in writing prior to effecting the transaction and must be given to the customer in writing before or with the customer’s confirmation. In addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from these rules, the broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written agreement to the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for the stock that is subject to these penny stock rules. Consequently, these penny stock rules may affect the ability of broker-dealers to trade our securities. We believe that the penny stock rules discourage investor interest in and limit the marketability of our common stock.
99
Procedures for Subscribing
If you decide to subscribe for any shares in this offering, you must (1) execute and deliver a subscription agreement; and (2) deliver a check or certified funds to us for acceptance or rejection.
All checks for subscriptions must be made payable to “XG Sciences, Inc.” The Company will deliver stock certificates attributable to shares of common stock purchased directly to the purchasers.
Right to Reject Subscriptions
We have the right to accept or reject subscriptions in whole or in part, for any reason or for no reason. All monies from rejected subscriptions will be returned immediately by us to the subscriber, without interest or deductions. Subscriptions for securities will be accepted or rejected with letter by mail within 48 hours after we receive them.
100
PLAN OF DISTRIBUTION FOR SECONDARY OFFERING
The selling securityholders may sell the securities described in this prospectus on a continuous or delayed basis directly to purchasers, through underwriters, broker-dealers or agents that may receive compensation in the form of discounts, concessions or commissions from us or the purchasers of the securities, in “at the market offerings” within the meaning of Rule 415(a)(4) of the Securities Act, to or through a market maker or into an existing trading market, on an exchange, or otherwise or through a combination of any such methods of sale. Discounts, concessions or commissions as to any particular underwriter, broker-dealer or agent may be in excess of those customary in the types of transactions involved. The securities may be sold from time to time in one or more transactions at fixed prices, which may be changed from time to time, at prevailing market prices at the time of sale, at varying prices determined at the time of sale or at negotiated prices. These sales may be effected in transactions, which may involve crosses or block transactions:
• on any national securities exchange or quotation service on which the securities may be listed or quoted at the time of sale, including, as of the date of this prospectus, the NASDAQ Capital Market in the case of our common stock;
• in the over-the-counter market;
• in transactions otherwise than on these exchanges or services or in the over-the-counter market; or
• through the writing of options, whether the options are listed on an options exchange or otherwise.
Each time that the selling securityholders use this prospectus to sell shares of common stock the selling securityholders shall also provide a prospectus supplement. For each series of securities, the applicable prospectus supplement will set forth the terms of the offering including:
• the public offering price;
• the name or names of any underwriters, dealers or agents;
• the purchase price of the securities;
• the proceeds from the sale of the securities to us;
• any underwriting discounts, agency fees, or other compensation payable to underwriters or agents;
• any discounts or concessions allowed or reallowed or repaid to dealers; and
• the securities exchanges on which the securities will be listed, if any.
The offering price of $8.00 is a fixed price at which the selling securityholders may sell their shares until our common stock is quoted on the OTCBB or OTC Markets at which time the common stock may be sold at prevailing market prices or at privately negotiated prices. There can be no assurance that a market maker will agree to file the necessary documents with the Financial Industry Regulatory Authority (FINRA), which operates the OTC Electronic Bulletin Board, nor can there be any assurance that such an application for quotation will be approved.
If the selling securityholder uses underwriters in the sale of securities, the securities will be acquired by the underwriters for their own account. The underwriters may then resell the securities in one or more transactions at a fixed public offering price or at varying prices determined at the time of sale or thereafter. The securities may be either offered to the public through underwriting syndicates represented by managing underwriters, or directly by underwriters. The obligations of the underwriters to purchase the securities will be subject to certain conditions. The underwriters will be obligated to purchase all the securities offered if they purchase any securities. The public offering price and any discounts or concessions allowed or re-allowed or paid to dealers may be changed from time to time.
If the selling securityholders use dealers in the sale of securities, the selling securityholders will sell securities to such dealers as principals. The dealers may then resell the securities to the public at varying prices to be determined by such dealers at the time of resale. The selling securityholders may solicit offers to purchase the securities directly, and the selling securityholders may sell the securities directly to institutional or other investors, who may be deemed underwriters within the meaning of the Securities Act with respect to any resales of those securities. The terms of these sales will be described in the applicable
101
prospectus supplement. If the selling securityholders use agents in the sale of securities, unless otherwise indicated in the prospectus supplement, they will use their reasonable best efforts to solicit purchases for the period of their appointment. Unless otherwise indicated in a prospectus supplement, if we sell directly, no underwriters, dealers or agents would be involved. We will not make an offer of securities in any jurisdiction that does not permit such an offer.
The selling securityholders may grant underwriters who participate in the distribution of securities an option to purchase additional securities to cover over-allotments, if any, in connection with the distribution. Any underwriter may engage in over-allotment, stabilizing transactions, short covering transactions and penalty bids in accordance with SEC orders, rules and regulations and applicable law. To the extent permitted by applicable law and SEC orders, rules and regulations, an over-allotment involves sales in excess of the offering size, which create a short position. Stabilizing transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed a specified maximum. To the extent permitted by applicable law and SEC orders, rules and regulations, short covering transactions involve purchases of the common stock in the open market after the distribution is completed to cover short positions. Penalty bids permit the underwriters to reclaim a selling concession from a dealer when the common stock originally sold by the dealer is purchased in a covering transaction to cover short positions. Those activities may cause the price of the common stock to be higher than it would otherwise be. If commenced, the underwriters may discontinue any of the activities at any time.
Any underwriters who are qualified market makers on the NASDAQ Stock Market may engage in passive market making transactions in the common stock on the NASDAQ Stock Market in accordance with Rule 103 of Regulation M, during the business day prior to the pricing of the offering, before the commencement of offers or sales of the common stock. Passive market makers must comply with applicable volume and price limitations and must be identified as passive market makers. In general a passive market maker must display its bid at a price not in excess of the highest independent bid for such security; if all independent bids are lowered below the passive market maker’s bid, however, the passive market maker’s bid must then be lowered when certain purchase limits are exceeded.
We have advised the selling securityholders that the anti-manipulation rules of Regulation M may apply to sales of shares in the market and to the activities of the selling securityholders’ affiliates. This regulation may limit the timing of purchases and sales of any of the shares of common stock offered in this prospectus by the selling securityholders. The anti-manipulation rules under the Exchange Act may apply to sales of shares in the market and to the activities of the selling securityholdes and their affiliates. Furthermore, Regulation M may restrict the ability of any person engaged in the distribution of the shares to engage in market-making activities for the particular securities being distributed for a period of up to five business days before the distribution. The restrictions may affect the marketability of the shares and the ability of any person or entity to engage in market-making activities for the shares.
Underwriters, dealers and agents that participate in any distribution of securities may be deemed to be underwriters as defined in the Securities Act. Any discounts, commissions or profit they receive when they resell the securities may be treated as underwriting discounts and commissions under the Securities Act. Only underwriters named in the prospectus supplement are underwriters of the securities offered in the prospectus supplement. The selling securityholders may have agreements with underwriters, dealers and agents to indemnify them against certain civil liabilities, including certain liabilities under the Securities Act, or to contribute with respect to payments that they may be required to make.
The selling securityholders may authorize underwriters, dealers or agents to solicit offers from certain institutions whereby the institution contractually agrees to purchase the securities from the selling securityholders on a future date at a specific price. This type of contract may be made only with institutions that the selling securityholders specifically approve. Such institutions could include banks, insurance companies, pension funds, investment companies and educational and charitable institutions. The underwriters, dealers or agents will not be responsible for the validity or performance of these contracts.
Each series of securities will be a new issue of securities. It has not presently been established whether the underwriters, if any, of the securities will make a market in the securities. If the underwriters make a market in the securities, such market making may be discontinued at any time without notice.
Agents, dealers and underwriters may be entitled to indemnification by us against certain civil liabilities, including liabilities under the Securities Act, or to contribution with respect to payments which the agents, dealers or underwriters may be required to make in respect thereof. Agents, dealers or underwriters may be customers of, engage in transactions with, or perform services for us and our subsidiaries in the ordinary course of business.
102
The validity of the securities being offered by this prospectus has been passed upon for us by Foster Swift Collins & Smith PC, with an address at 313 South Washington Square, Lansing, MI 48933-2114.
The 2013 financial statements included in this prospectus have been so included in reliance on the report of Kingery & Crouse P.A., an independent registered public accounting firm, given on the authority of said firm as experts in auditing and accounting. The 2014 and subsequent financial statements included in this prospectus have been so included in reliance on the report of Frazier & Deeter, LLC, an independent registered public accounting firm, given on the authority of said firm as experts in auditing and accounting.
No expert or counsel named in this prospectus as having prepared or certified any part of this prospectus or having given an opinion upon the validity of the securities being registered or upon other legal matters in connection with the registration or offering of the common stock was employed on a contingency basis, or had, or is to receive, in connection with the offering, a substantial interest, direct or indirect, in the registrant or any of its parents or subsidiaries. Nor was any such person connected with the registrant or any of its parents or subsidiaries as a promoter, managing or principal underwriter, voting trustee, director, officer, or employee.
WHERE YOU CAN FIND ADDITIONAL INFORMATION
We filed with the Securities and Exchange Commission a registration statement under the Securities Act for the shares of common stock in this offering. This prospectus does not contain all of the information in the registration statement and the exhibits that were filed with the registration statement. For further information with respect to us and our common stock, we refer you to the registration statement and the exhibits that were filed with the registration statement. Statements contained in this prospectus about the contents of any contract or any other document that is filed as an exhibit to the registration statement are not necessarily complete but do contain the material provisions for each such contract and other document, and we refer you to the full text of the contract or other document filed as an exhibit to the registration statement. A copy of the registration statement and the exhibits that were filed with the registration statement may be inspected without charge at the Public Reference Room maintained by the Securities and Exchange Commission at 100 F Street, N.E. Washington, DC 20549, and copies of all or any part of the registration statement may be obtained from the Securities and Exchange Commission upon payment of the prescribed fee. Information regarding the operation of the Public Reference Room may be obtained by calling the Securities and Exchange Commission at 1-800-SEC-0330. The Securities and Exchange Commission maintains a website that contains reports, proxy and information statements, and other information regarding registrants that file electronically with the SEC. The address of the website iswww.sec.gov.
We will become obligated to file periodic reports upon the effectiveness of the registration statement of which this prospectus is made a part and we will file periodic reports under the Exchange Act, including annual, quarterly and special reports, and other information with the SEC. These periodic reports and other information will be available for inspection and copying at the regional offices, public reference facilities and website of the Securities and Exchange Commission referred to above.
We will also make available free of charge on or through our internet website our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Exchange Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the Securities and Exchange Commission.
103
DISCLOSURE OF COMMISSION POSITION ON INDEMNIFICATIONFOR SECURITIES ACT LIABILITY
Our Articles of Incorporation, as amended, state that a Director of the Company shall not be personally liable to the Company or its stockholders for money damages for any action taken or any failure to take any action as a Director, except liability for any of the following:
(a) The amount of a financial benefit received by a Director to which the Director is not entitled;
(b) The intentional infliction of harm on the Company or its stockholders;
(c) A violation of Section 551 of the Michigan Business Corporation Act, as amended (the “Act”); or
(d) An intentional criminal act.
In the event the Act is amended, after approval by the stockholders of the appurtenant article in the Company’s Articles of Incorporation, as amended to authorize corporate action further eliminating or limiting the personal liability of Directors, then the liability of a Director of the Company shall be eliminated or limited to the fullest extent permitted by the Act, as so amended. Any repeal, modification or adoption of any provision in our Articles of Incorporation, as amended inconsistent with this Article in shall not adversely affect any right or protection of a Director of the Company existing at the time of such repeal, modification or adoption.
Our Bylaws states that each person who is or was or had agreed to become a Director or officer of the Company, or each such person who is or was serving or who had agreed to serve at the request of the Board of Directors as an employee or agent of the Company, or as a Director, officer, employee or agent of another corporation (whether for profit or not), partnership, joint venture, trust or other enterprise (including the heirs, executors, administrators or estate of such person), shall be indemnified by the Company to the full extent permitted by the Act or any other applicable laws as presently or hereafter in effect.
The right to indemnification conferred in the Bylaws shall not be exclusive of any right any person may have or acquire under any statute, provision of the Articles of Incorporation, as amended, Bylaws, agreement, vote of stockholders or disinterested Directors, or otherwise.
The Company shall have the power to purchase and maintain insurance on behalf of any person who is or was a Director, officer, employee or agent of the Company or is or was serving at the request of the Company as a Director, officer, partner, trustee, employees or agent of another foreign or domestic corporation, partnership, joint venture, trust or other enterprise, whether for profit or not, against any liability asserted against him or her and incurred by him or her in any such capacity or arising out of his or her status as such, whether or not the Company would have the power to indemnify him or her against such liability under the appurtenant provision of the Company’s Bylaws.
Under Michigan law, however, such provisions do not eliminate the personal liability of a director unless the director (i) acted in good faith; (ii) acted in a manner he or she reasonably believed to be in or not opposed to the best interests of the corporation or its shareholders; and (iii) with respect to a criminal action or proceeding, if the director had no reasonable cause to believe his or her conduct was unlawful. Furthermore, under Section 551of the Act, directors who vote for, or concur in, any of the following corporate actions are jointly and severally liable to the corporation for the benefit of its creditors or shareholders, to the extent any legally recoverable injury suffered by its creditors or shareholders as a result of the action but not to exceed the difference between the amount paid or distributed and the amount that lawfully could have been paid or distributed: (i) a declaration of a share dividend or distribution to shareholders contrary to the Act or contrary to any restriction in the articles of incorporation; (ii) distribution to shareholders during or after dissolution of the corporation without paying or providing for debts obligations and liabilities of the corporation as required by Section 855a of the Act; and (iii) making a loan to a director, officer, or employee of the corporation or of a subsidiary of the corporation contrary to the Act.
We have been advised that in the opinion of the Securities and Exchange Commission indemnification for liabilities arising under the Securities Act is against public policy as expressed in the Securities Act, and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities is asserted by one of our directors, officers, or controlling persons in connection with the securities being registered, we will, unless in the opinion of our legal counsel the matter has been settled by controlling precedent, submit the question of whether such indemnification is against public policy to a court of appropriate jurisdiction. We will then be governed by the court’s decision.
104
XG SCIENCES, INC.
CONSOLIDATED FINANCIAL STATEMENTS
INDEX TO FINANCIAL STATEMENTS
TABLE OF CONTENTS
|
| Page |
Consolidated Financial Statements as of and for the years ended December 31, 2014 and 2013 |
|
|
|
|
|
| F-2 | |
|
|
|
| F-4 | |
|
|
|
| F-5 | |
|
|
|
Consolidated Statement of Changes in Stockholders’ Equity (Deficit) |
| F-6 |
|
|
|
| F-7 | |
|
|
|
| F-8 – F-28 | |
|
|
|
Unaudited Condensed Consolidated Financial Statements as of and for the Nine Months Ended September 30, 2015 and 2014 |
|
|
|
|
|
| F-29 | |
|
|
|
| F-30 | |
|
|
|
Condensed Consolidated Statement of Changes in Stockholders’ Equity (Deficit) |
| F-31 |
|
|
|
| F-32 | |
|
|
|
| F-33 – F-45 |
F-1

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors
XG Sciences, Inc.
We have audited the accompanying consolidated balance sheet of (the “Company”) as of December 31, 2014, and the related consolidated statements of operations, changes in stockholders’ equity (deficit), and cash flows for the year then ended. These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2014, and the results of its operations and its cash flows for the year then ended in conformity with accounting principles generally accepted in the United States of America.
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to such financial statements, the Company has incurred recurring losses from operations, has recurring negative cash flows from operations, has shareholder deficits and currently has ongoing requirements for additional capital investment. These factors raise doubt about the Company’s ability to continue as a going concern. Management’s plans in regard to these matters are described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
/s/ Frazier & Deeter, LLC
Frazier & Deeter, LLC
Tampa, Florida
February 26, 2015
F-2

REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors
XG Sciences, Inc.
We have audited the accompanying consolidated balance sheet of (the “Company”) as of December 31, 2013, and the related statements of operations, changes in stockholders’ equity (deficit), and cash flows for the year then ended. These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these financial statements based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements, assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December 31, 2013 and the results of its operations and its cash flows for the year then ended in conformity with accounting principles generally accepted in the United States of America.
/s/ Kingery & Crouse, PA
Kingery & Crouse, PA
Tampa, Florida
March 24, 2014
F-3
XG SCIENCES, INC.
CONSOLIDATED BALANCE SHEETS AS OF DECEMBER 31, 2014 AND 2013
|
| 2014 |
| 2013 | ||||
ASSETS |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT ASSETS |
|
|
|
|
|
|
|
|
Cash |
| $ | 2,088,866 |
|
| $ | 95,342 |
|
Accounts receivable, less allowance for doubtful accounts of $10,000 in 2014 and 2013, respectively |
|
| 124,983 |
|
|
| 201,258 |
|
Inventories |
|
| 132,459 |
|
|
| 67,160 |
|
Incentive refunds receivable |
|
| 178,430 |
|
|
| 133,268 |
|
Other current assets |
|
| 18,117 |
|
|
| 12,362 |
|
|
| 2,542,855 |
|
|
| 509,390 |
| |
|
|
|
|
|
|
|
|
|
PROPERTY, PLANT AND EQUIPMENT, NET |
|
| 4,652,473 |
|
|
| 4,237,284 |
|
|
|
|
|
|
|
|
|
|
RESTRICTED CASH FOR LETTER OF CREDIT |
|
| 190,915 |
|
|
| 190,526 |
|
|
|
|
|
|
|
|
|
|
INTANGIBLE ASSETS, NET |
|
| 327,134 |
|
|
| 229,745 |
|
|
|
|
|
|
|
|
|
|
TOTAL ASSETS |
| $ | 7,713,377 |
|
| $ | 5,166,945 |
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ (DEFICIT) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES |
|
|
|
|
|
|
|
|
Accounts payable and other current liabilities |
| $ | 1,073,938 |
|
| $ | 1,501,142 |
|
Deferred revenue |
|
| — |
|
|
| 600,000 |
|
Line of credit |
|
| — |
|
|
| 103,447 |
|
Current portion of capital lease obligations |
|
| 152,593 |
|
|
| 128,563 |
|
|
| 1,226,531 |
|
|
| 2,333,152 |
| |
|
|
|
|
|
|
|
|
|
LONG-TERM LIABILITIES |
|
|
|
|
|
|
|
|
Deferred compensation |
|
| — |
|
|
| 254,400 |
|
Long-term portion of capital lease obligations |
|
| 532,970 |
|
|
| 455,063 |
|
Convertible notes payable, net |
|
| 10,422,641 |
|
|
| 3,410,440 |
|
Derivative liability – warrants |
|
| 5,000,752 |
|
|
| 1,190,261 |
|
|
| 15,956,363 |
|
|
| 5,310,164 |
| |
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES |
|
| 17,182,894 |
|
|
| 7,643,316 |
|
|
|
|
|
|
|
|
|
|
STOCKHOLDERS’ (DEFICIT) |
|
|
|
|
|
|
|
|
Series A convertible preferred stock, 3,000,000 shares authorized, 316,010 and |
|
| 3,475,686 |
|
|
| 2,230,092 |
|
Common stock, no par value, 25,000,000 shares authorized, 835,144 and 833,477 shares issued and outstanding at December 31, 2014 and 2013, respectively |
|
| 8,551,225 |
|
|
| 8,531,221 |
|
Additional paid-in capital |
|
| 5,192,295 |
|
|
| 2,026,981 |
|
Accumulated (deficit) |
|
| (26,688,723 | ) |
|
| (15,264,665 | ) |
|
| (9,469,517 | ) |
|
| (2,476,371 | ) | |
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES AND STOCKHOLDERS’ (DEFICIT) |
| $ | 7,713,377 |
|
| $ | 5,166,945 |
|
See notes to consolidated financial statements
F-4
XG SCIENCES, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS FOR THE YEARS ENDED DECEMBER 31, 2014 AND 2013
|
| 2014 |
| 2013 | ||||
REVENUES |
|
|
|
|
|
|
|
|
Product sales |
| $ | 874,469 |
|
| $ | 239,767 |
|
Grants |
|
| 660,533 |
|
|
| 174,535 |
|
Licensing revenue |
|
| 100,000 |
|
|
| 1,400,000 |
|
Total revenues |
|
| 1,635,002 |
|
|
| 1,814,302 |
|
|
|
|
|
|
|
|
|
|
COST OF GOODS SOLD |
|
|
|
|
|
|
|
|
Direct costs |
|
| 226,678 |
|
|
| 129,243 |
|
Unallocated manufacturing expenses |
|
| 2,522,653 |
|
|
| 2,125,912 |
|
Total cost of goods sold |
|
| 2,749,331 |
|
|
| 2,255,155 |
|
|
|
|
|
|
|
|
|
|
GROSS LOSS |
|
| (1,114,329 | ) |
|
| (440,853 | ) |
|
|
|
|
|
|
|
|
|
OPERATING EXPENSES |
|
|
|
|
|
|
|
|
Research and development |
|
| 1,449,478 |
|
|
| 1,269,456 |
|
Sales, general and administrative |
|
| 4,405,266 |
|
|
| 2,739,956 |
|
Total operating expenses |
|
| 5,854,744 |
|
|
| 4,009,412 |
|
|
|
|
|
|
|
|
|
|
OPERATING LOSS |
|
| (6,969,073 | ) |
|
| (4,450,265 | ) |
|
|
|
|
|
|
|
|
|
OTHER INCOME (EXPENSE) |
|
|
|
|
|
|
|
|
Incentive refund and interest income |
|
| 117,940 |
|
|
| 72,467 |
|
Interest expense |
|
| (1,507,374 | ) |
|
| (471,102 | ) |
Gain from change in fair value of derivative liability – warrants |
|
| 503,186 |
|
|
| 81,339 |
|
Extinguishment loss on convertible notes payable |
|
| (3,513,770 | ) |
|
| — |
|
Loss on disposal of equipment and intangible assets |
|
| (54,967 | ) |
|
| (44,895 | ) |
|
|
|
|
|
|
|
|
|
Total other (expense) |
|
| (4,454,985 | ) |
|
| (362,191 | ) |
|
|
|
|
|
|
|
|
|
NET LOSS |
|
| (11,424,058 | ) |
|
| (4,812,456 | ) |
|
|
|
|
|
|
|
|
|
Deemed dividend on Series A convertible preferred stock |
|
| (316,366 | ) |
|
| — |
|
|
|
|
|
|
|
|
|
|
NET LOSS ATTRIBUTABLE TO COMMON STOCKHOLDERS |
| $ | (11,740,424 | ) |
| $ | (4,812,456 | ) |
|
|
|
|
|
|
|
|
|
WEIGHTED AVERAGE NUMBER OF SHARES OUTSTANDING – Basic and diluted |
|
| 834,400 |
|
|
| 993,371 |
|
NET LOSS PER SHARE– Basic and diluted |
| $ | (14.07 | ) |
| $ | (4.84 | ) |
See notes to consolidated financial statements
F-5
XG SCIENCES, INC.
CONSOLIDATED STATEMENT OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT)
FOR THE YEARS ENDED DECEMBER 31, 2014 and 2013
|
| Preferred stock |
| Common stock |
| Additional |
| Accumulated |
|
| |||||||||||||||
|
| Shares |
|
| Amount |
|
| Shares |
|
|
| Amount |
|
|
| paid-in capital |
|
|
| Deficit |
|
|
| Total |
|
Balances, December 31, 2012 |
| — |
| $ | — |
|
| 1,008,211 |
|
| $ | 10,628,029 |
|
| $ | 660,850 |
|
| $ | (10,452,209 | ) |
| $ | 836,670 |
|
Preferred stock issued for cash |
| 11,107 |
|
| 133,284 |
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| 133,284 |
|
Common stock converted to preferred stock |
| 174,734 |
|
| 2,096,808 |
|
| (174,734 | ) |
|
| (2,096,808 | ) |
|
| — |
|
|
| — |
|
|
| — |
|
Preferred stock issued – beneficial conversion feature |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| 1,098,184 |
|
|
| — |
|
|
| 1,098,184 |
|
Common stock warrants reclassified from derivative liabilities |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| 34,945 |
|
|
| — |
|
|
| 34,945 |
|
Employee stock option expense |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| 233,002 |
|
|
| — |
|
|
| 233,002 |
|
Net loss |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| — |
|
|
| (4,812,456 | ) |
|
| (4,812,456 | ) |
Balances, December 31, 2013 |
| 185,841 |
|
| 2,230,092 |
|
| 833,477 |
|
|
| 8,531,221 |
|
|
| 2,026,981 |
|
|
| (15,264,665 | ) |
|
| (2,476,371 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock issued for cash |
| 101,000 |
|
| 895,634 |
|
| 1,667 |
|
|
| 20,004 |
|
|
| — |
|
|
| — |
|
|
| 915,638 |
|
Preferred stock issued – beneficial conversion feature |
| — |
|
| (316,366 | ) |
| — |
|
|
| — |
|
|
| 316,366 |
|
|
| — |
|
|
| — |
|
Preferred stock issued – deemed dividend from beneficial conversion feature |
| — |
|
| 316,366 |
|
| — |
|
|
| — |
|
|
| (316,366 | ) |
|
| — |
|
|
| — |
|
Preferred stock issued to pay capital lease obligations |
| 29,169 |
|
| 349,960 |
|
| — |
|
|
| — |
|
|
| — |
|
|
| — |
|
|
| 349,960 |
|
Convertible notes issued – beneficial conversion feature |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| 2,520,526 |
|
|
| — |
|
|
| 2,520,526 |
|
Employee stock option expense |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| 644,788 |
|
|
| — |
|
|
| 644,788 |
|
Net loss |
| — |
|
| — |
|
| — |
|
|
| — |
|
|
| — |
|
|
| (11,424,058 | ) |
|
| (11,424,058 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balances, December 31, 2014 |
| 316,010 |
| $ | 3,475,686 |
|
| 835,144 |
|
| $ | 8,551,225 |
|
| $ | 5,192,295 |
|
| $ | (26,688,723 | ) |
| $ | (9,469,517 | ) |
See notes to consolidated financial statements
F-6
XG SCIENCES, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
DECEMBER 31, 2014 AND 2013
|
| 2014 |
| 2013 | ||||
CASH FLOWS FROM OPERATING ACTIVITIES |
|
|
|
|
|
|
|
|
Net loss |
| $ | (11,424,058 | ) |
| $ | (4,812,456 | ) |
Adjustments to reconcile net loss to net cash used in operating activities |
|
|
|
|
|
|
|
|
Depreciation and amortization |
|
| 866,889 |
|
|
| 717,408 |
|
Amortization of intangible assets |
|
| 25,096 |
|
|
| 18,946 |
|
Loss on disposal of equipment and intangible assets |
|
| 54,967 |
|
|
| 44,895 |
|
Provision for (recovery of) bad debts |
|
| 3,038 |
|
|
| (2,691 | ) |
Stock-based compensation expense |
|
| 644,788 |
|
|
| 233,002 |
|
Non-cash interest expense |
|
| 1,501,138 |
|
|
| 437,938 |
|
Deferred compensation – phantom stock |
|
| (120,000 | ) |
|
| (249,590 | ) |
Gain from change in fair value of derivative liability – warrants |
|
| (503,186 | ) |
|
| (81,339 | ) |
Extinguishment loss on convertible notes payable |
|
| 3,513,770 |
|
|
| — |
|
(Increase) Decrease in: |
|
|
|
|
|
|
|
|
Accounts receivable |
|
| 173,238 |
|
|
| 33,711 |
|
Inventories |
|
| (65,299 | ) |
|
| (26,965 | ) |
Incentive refund receivable |
|
| (45,162 | ) |
|
| (72,000 | ) |
Other current assets |
|
| (106,537 | ) |
|
| 23,556 |
|
Other assets |
|
| (389 | ) |
|
| (162 | ) |
Increase (Decrease) in: |
|
|
|
|
|
|
|
|
Note payable for compensation |
|
| — |
|
|
| (91,375 | ) |
Accounts payable and other liabilities |
|
| (468,173 | ) |
|
| 1,006,392 |
|
Accrued compensation |
|
| 58,967 |
|
|
| (19,564 | ) |
Deferred revenue |
|
| (600,000 | ) |
|
| (650,000 | ) |
NET CASH USED IN OPERATING ACTIVITIES |
|
| (6,490,914 | ) |
|
| (3,490,294 | ) |
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM INVESTING ACTIVITIES |
|
|
|
|
|
|
|
|
Purchases of property and equipment |
|
| (924,947 | ) |
|
| (1,051,920 | ) |
Purchases of intangible assets |
|
| (158,249 | ) |
|
| (35,655 | ) |
NET CASH USED IN INVESTING ACTIVITIES |
|
| (1,083,196 | ) |
|
| (1,087,575 | ) |
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM FINANCING ACTIVITIES |
|
|
|
|
|
|
|
|
Advances (repayments) on line of credit, net |
|
| (103,447 | ) |
|
| (18,401 | ) |
Repayments of capital lease obligations |
|
| (60,923 | ) |
|
| (24,989 | ) |
Proceeds of convertible notes and warrants |
|
| 8,500,000 |
|
|
| 4,503,847 |
|
Proceeds from issuance of preferred stock |
|
| 1,212,000 |
|
|
| 133,284 |
|
Proceeds from issuance of common stock |
|
| 20,004 |
|
|
| — |
|
NET CASH PROVIDED BY FINANCING ACTIVITIES |
|
| 9,567,634 |
|
|
| 4,593,741 |
|
|
|
|
|
|
|
|
|
|
NET INCREASE IN CASH |
|
| 1,993,524 |
|
|
| 15,872 |
|
CASH AT BEGINNING OF PERIOD |
|
| 95,342 |
|
|
| 79,470 |
|
|
|
|
|
|
|
|
|
|
CASH AT END OF PERIOD |
| $ | 2,088,866 |
|
| $ | 95,342 |
|
|
|
|
|
|
|
|
|
|
SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: |
|
|
|
|
|
|
|
|
Cash paid for interest |
| $ | 6,236 |
|
| $ | 29,037 |
|
SUPPLEMENTAL DISCLOSURE OF NON-CASH INVESTING AND FINANCING: |
|
|
|
|
|
|
|
|
Conversion of common stock to preferred stock |
| $ | — |
|
| $ | 2,096,808 |
|
Value of preferred stock issued for AAOF capital lease obligations |
| $ | 349,960 |
|
| $ | — |
|
Property and equipment additions under capital leases |
| $ | 390,321 |
|
| $ | 756,110 |
|
Conversion of line of credit to secured convertible notes |
| $ | — |
|
| $ | 700,000 |
|
Reclassification of derivative warrant liability to additional paid-in capital |
| $ | — |
|
| $ | 34,945 |
|
See notes to consolidated financial statements
F-7
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 1 — NATURE OF BUSINESS AND BASIS OF PRESENTATION
XG Sciences, Inc., a Michigan company located in Lansing, Michigan and its subsidiary XGS IP, LLC (“we”, “us”, “our”, or the “Company”) manufactures graphene nanoplatelets made from graphite, using a proprietary manufacturing process to split natural flakes of crystalline graphite into very small and thin particles. These nanoplatelets are then used in products like battery electrodes, thin sheets, films, inks and coatings that we sell to other companies. We also sell our xGnP® graphene nanoplatelets in the form of bulk powders or dispersions to other companies for use as additives to make composite and other materials with specially engineered characteristics. Additionally, we license our technology to other companies in exchange for royalties and other fees.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“US GAAP”). All normal adjustments considered necessary for a fair presentation have been included.
The consolidated financial statements include the accounts of XG Sciences, Inc. and our wholly-owned subsidiary, XGS IP, LLC. We operate as one reportable segment. All intercompany accounts, transactions and profits have been eliminated in consolidation.
Going Concern
We have historically incurred recurring losses from operations and we may continue to generate negative cash flows as we implement our business plan. Our consolidated financial statements are prepared using US GAAP as applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business.
We currently do not have sufficient cash or commitments for financing to sustain our operations for the next twelve months. Our plan is to develop customer relationships and increase our revenues derived from our products and IP licensing. Although we have historically incurred operating losses, we have been able to fund such losses primarily by selling common and preferred stock and convertible notes. We expect that our cash on hand at December 31, 2014 of $2,088,866 and proceeds from anticipated financings (private placements of equity securities) will sustain our operations for the next twelve months. However, there can be no assurance of our continued success in raising capital to fund our business plan.
In the event we are unable to fund our operations from existing cash on hand, operating cash flows, additional borrowings or raising equity capital, we may be forced to reduce our expenses, slow down our growth rate, or discontinue operations. Our consolidated financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary should we be unable to continue as a going concern.
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Use of Estimates
US GAAP requires management to make estimates, judgments and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses, together with amounts disclosed in the related notes to the consolidated financial statements. Actual results and outcomes may differ from management’s estimates, judgments and assumptions. Significant estimates, judgments and assumptions used in these consolidated financial statements include, but are not limited to, those related to revenues, accounts receivable and related allowances, contingencies, useful lives and recovery of long-term assets, income taxes, and the fair values of stock-based compensation and derivative financial instrument liabilities. These estimates, judgments, and assumptions are reviewed periodically and the effects of material revisions in estimates are reflected in the financial statements prospectively from the date of the change in estimate.
F-8
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(cont.)
Revenue Recognition
We recognize revenues when (a) the price is fixed or determinable, (b) persuasive evidence of a sales arrangement exists, (c) the service is performed or delivery has occurred and (d) collectability of the resulting receivable is reasonably assured.
We recognize product revenues when products are shipped to customers. At that time, product ownership and risk have transferred to the customer and we have no further obligations. We record product sales at net selling prices that are reflective of discounts and allowances. Shipping and handling costs are recorded as a component of Direct Costs, as are shipping and handling costs billed to customers.
Revenue related to licensing agreements is recorded upon substantial performance of the terms of the contract. In the case of licensing arrangements that involve ongoing royalties based on sales of products produced with our technology, royalty income is recorded when received or, in the case of minimum royalties due, in the period when due.
Grant contract revenue is recognized over the life of the contracts as the services are provided.
Amounts received in excess of revenues earned are recorded as deferred revenue.
Cost of Products Sold
We use a standard cost system to estimate the direct costs of products sold. Direct costs include estimates of raw material costs, packaging, freight charges net of those billed to customers, and an allocation for direct labor and manufacturing overhead. Because of the nature of our production processes, there is a substantial fixed manufacturing expense requirement that represents the ongoing cost of maintaining production facilities that are not directly related to products sold, so we use a “full capacity” allocation of overhead based on an estimate of what product costs would be if the manufacturing facilities were operating on a full-time basis and producing products at the designed capacity. This estimate is updated annually and involves estimating both the level of expenses as well as production amounts as if the manufacturing facility were operating on a continuous, three-shift, production basis.
The remaining costs of operating the Company’s manufacturing facilities are recorded as Unallocated Manufacturing Expenses. Manufacturing expense includes the costs of operating our manufacturing facilities including personnel costs, rent, utilities, indirect supplies, depreciation, and related indirect expenses. Manufacturing expenses are expensed as incurred.
Research and Development
Research and development expenses include the compensation costs of research personnel, laboratory rent and utilities, depreciation of laboratory equipment, travel and laboratory supplies and are expensed as incurred.
General and Administrative Expense
General and Administrative expenses include the compensation costs of personnel, rent, utilities, supplies, travel, depreciation of office equipment, and related expenses not included in other expense categories. General and Administrative Expense also includes non-cash compensation expenses related to the Company’s deferred compensation, management incentive bonus, and employee stock option programs.
Sales and Marketing Expense
Sales and marketing costs include compensation, travel, and business development expenses including free samples provided to customers. These costs are expensed as incurred. Product marketing allowances are recorded at the estimated out of pocket cost necessary to produce the product in the period the allowance is granted. Advertising costs are expensed at the time they are incurred and were not material for the years ended December 31, 2014 and 2013.
F-9
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(cont.)
Income Taxes
It is our policy to provide for uncertain tax positions and the related interest and penalties based upon management’s assessment of whether a tax benefit is more likely than not to be sustained upon examination by tax authorities. To the extent that the probable tax outcome of these uncertain tax position changes, such changes in estimate will impact the income tax provision in the period in which such determination is made. At December 31, 2014, we believe we have appropriately accounted for any unrecognized tax benefits. We are not aware of any uncertain tax positions. To the extent we prevail in matters for which a liability for an unrecognized tax benefit is established or we are required to pay amounts in excess of the liability, our effective tax rate in a given financial statement period may be affected.
We account for income taxes using an asset and liability approach. The difference between the financial statement and tax basis of assets and liabilities is determined annually. Deferred income tax assets and liabilities are computed for those differences that have future tax consequences using the currently enacted tax laws and rates that apply to the periods in which they are expected to affect taxable income. Income tax expense is the current tax payable or refundable for the period plus or minus the net change in the deferred tax assets and liabilities. The deferred tax effects of state and local income taxes are considered immaterial and have not been recorded. Valuation allowances are established, if necessary, to reduce deferred tax assets to the amount that is estimated to be realized. Because of the uncertainty related to future realization of deferred tax assets (see Note 15), we have established a valuation allowance equal to one hundred percent of the deferred tax assets.
Net Income (Loss) Per Common Share
We compute net income or (loss) per share in accordance with Financial Accounting Standards Board (“FASB”) Accountings Standards Codification (“ASC��) Topic 260: Earnings Per Share. Under the provisions of ASC 260, basic net income (loss) per share is computed by dividing the net income (loss) available to common stockholders by the weighted average number of common shares outstanding during the period. Diluted net loss per share is computed using the weighted average number of shares outstanding during the applicable period, plus the effect of potentially dilutive securities. Potentially dilutive securities consist of shares potentially issuable pursuant to stock options and warrants as well as shares that would result from full conversion of all outstanding convertible securities. During periods of net loss per share, these potentially dilutive securities are excluded from diluted net loss per share calculations because they are anti-dilutive. As a result, for the years ended December 31, 2014 and 2013, basic and diluted net loss per share were the same.
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable are stated at the amount management expects to collect from outstanding balances. Management provides for probable uncollectible amounts through a provision for bad debt expense and an adjustment to a valuation allowance based on their assessment of the current status of individual accounts. Balances that are still outstanding after management has used reasonable collection efforts are written off through a charge to the allowance account and a credit to accounts receivable. Changes in the valuation allowance are disclosed on the balance sheet.
Statements of Cash Flows
For the purposes of the statements of cash flows, we consider all highly liquid investments purchased with an original maturity of three months or less to be cash equivalents.
Fair Value of Financial Instruments and Concentrations of Credit Risk
Financial instruments, which potentially subject the Company to concentrations of credit risk, consist principally of cash and accounts receivable. The Company places its cash with FDIC insured financial institutions. Although such balances may exceed the federally insured limits at certain times, in the opinion of management they are subject to minimal risk.
F-10
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(cont.)
The Company has established policies for extending credit to customers based upon factors including the customers’ credit worthiness, historical trends and other information. Nonetheless the collectability of accounts receivable is affected by regional economic conditions and other factors. During 2013, the Company earned $1,400,000 of licensing revenue from one entity and also recorded $600,000 in deferred revenues from the same entity as part of a negotiated contract. At December 31, 2013, $187,500 of accounts receivable was outstanding from this entity. At December 31, 2014, there was no outstanding balance due from this entity and the deferred revenue of $600,000 is reflected in revenues.
Inventory
Inventory consists of raw materials, work-in-process and finished goods, all of which are valued at standard cost, which approximates average cost.
Property and Equipment
Property and equipment are recorded at cost, net of accumulated depreciation and amortization. Property and equipment generally includes purchases of items with a cost greater than $3,000 and a useful life greater than one year. Depreciation and amortization are computed on the straight line basis over the estimated useful lives of the assets. Leasehold improvements are amortized over the shorter of the related lease terms or their estimated useful lives.
We periodically review the estimated useful lives of property and equipment. Changes to the estimated useful lives are recorded prospectively from the date of the change. Upon retirement or sale, the cost of the assets disposed of and the related accumulated depreciation are removed from the accounts and any resulting gain or loss is included in the statement of operations. Repairs and maintenance costs are expensed as incurred.
Recoverability and Impairment of Long-Lived Assets
We review our long-lived assets for recoverability if events or changes in circumstances indicate the assets may be impaired. This circumstance exists when the carrying amount of the asset exceeds the sum of the undiscounted cash flows expected to result from its use and eventual disposition. At December 31, 2014, we believe the carrying value of our long-lived assets is recoverable.
Intangible Assets
We have entered into a License Agreement with Michigan State University under which we have licensed certain intellectual property in the form of patents and patent applications and invention disclosures. We are responsible for managing the patent process and ongoing filings for this licensed intellectual property and for bearing the cost thereof. We capitalize all costs related to the acquisition and ongoing administration of this License Agreement and we amortize these costs over 15 years or the remaining life of the License Agreement, whichever is shorter.
In addition to the costs of managing in-licensed intellectual property, we also file for patent protection for inventions and other intellectual property generated by our employees. All patents are evaluated for filing in international markets on a case-by-case basis, and are filed in the United States and in selected international markets as considered appropriate. All external legal and filing costs related to patent applications, patent filings, ongoing registrations, overseas filings, and legal opinions related thereto are capitalized as intangible assets at cost and amortized over a period of 15 years from the date incurred, or the remaining useful life of the associated patent, whichever is shorter.
The cost of royalties or minimum payments specified under the license agreement for in-licensed technology is expensed as incurred.
F-11
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(cont.)
We have also out-licensed certain of our intellectual property to licensees under terms and conditions of license agreements that specify the intellectual property licensed, the territory, and the type of license. In exchange for these licenses, we have recorded revenues associated with the initial granting of the license and expect to receive royalties based on sales of products produced under these licenses. License revenues are recorded to reflect our performance of requirements under these license agreements. In addition, we record royalty revenues from licensees at the time they are earned.
Our intangible assets are assessed for impairment on an annual basis. As part of this process, we take into account the cash flows which we have received from licensing certain of our intellectual property as well as our plans to continue licensing our intellectual property as part of our ongoing business model.
Incentive Refund
As of December 31, 2014 and 2013, we had $178,430 and $133,268 of incentive refunds due from the Michigan Economic Growth Authority under a five-year agreement signed on January 19, 2011. These incentive payments are awarded annually based on a pre-determined formula relating to the number of jobs created, average compensation, and total payroll and benefits for jobs created by XG Sciences in Michigan. These refunds are paid in cash during the year following the refund period. There is no requirement to have taxable income to receive these incentive payments.
Stock-Based Compensation
We recognize compensation expense in our statement of operations for all share-based option and stock awards, based on estimated grant-date fair values.
We estimate the grant-date fair value of stock-based compensation awards using the Black-Scholes option valuation model. This model is affected by the estimated value of our common stock on the date of the grant as well as assumptions regarding a number of highly complex and subjective variables. These variables include the expected term of the option, the exercise price, expected risk-free rates of return, the expected volatility of our common stock, and expected dividend yield, each of which is more fully described below. The assumptions for the estimated value of our common stock, expected term and expected volatility are the assumptions that most significantly affect the grant date fair value.
Estimated Value of our Common Stock: The estimated value of our common stock was determined on the basis of recent third-party investment transactions that established a price of $12 per share as the value at which outsiders were willing to invest in our stock.
Expected Term: Because we have limited experience related to the exercise of employee stock options, we use the simplified method permitted by SEC Staff Accounting Bulletin Topic 14 to estimate the expected term of the options. The expected term of an option is estimated to be equal to the mid-point between the vesting and expiration dates of the option.
Risk-free Interest Rate: We base the risk-free interest rate used on the implied yield at the grant date of U.S. Treasury zero-coupon issues with a term approximately equal to the expected term of the stock-based award being valued.
Expected Stock Price Volatility: Because we are a private company with very limited stock sales history, we use a blended average weekly volatility of certain publically-traded peer companies. We believe that the use of this blended average peer volatility is reflective of market conditions and a reasonable indicator of our expected future volatility.
Dividend Yield: Because we have never paid a dividend and do not expect to begin doing so in the foreseeable future, we have assumed a 0% dividend yield in valuing our stock-based awards.
The grant-date fair value of the award is recognized as expense over the requisite service period using the straight-line method.
F-12
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(cont.)
Fair Value Measurements
FASB ASC 820: “Fair Value Measurements and Disclosures” defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. ASC 820 also establishes a fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure fair value:
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Inputs other than quoted prices included within Level 1 which are either directly or indirectly observable.
Level 3: Inputs to the valuation methodology are unobservable and significant to the fair value measurement.
A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement. Our derivative liabilities are classified as Level 3 within the fair value hierarchy because they were valued using other unobservable inputs. The valuation technique used to measure fair value of the derivative liabilities is based on a lattice model with significant assumptions and inputs determined by the Company. A lattice model was used to estimate the fair value of the derivative liabilities because management believes it reflects all the assumptions that market participants would likely consider including early exercise of the warrants. The fair value of the derivative liabilities will be significantly influenced by the fair value of our common stock, stock price volatility and the risk free interest components of the lattice technique.
The following is a reconciliation of the beginning and ending balances for liabilities measured at fair value on a recurring basis using significant unobservable inputs (Level 3) during the years ended December 31, 2014 and 2013:
|
| 2014 |
| 2013 | ||||
Balance at January 1 |
| $ | 1,190,261 |
|
| $ | 60,865 |
|
Warrants issued with additional financing |
|
| 2,676,570 |
|
|
| 1,245,680 |
|
Warrants issued with preferred stock sold under preemptive rights |
|
| 316,366 |
|
|
| — |
|
Warrants issued for debt modification |
|
| 1,320,741 |
|
|
| — |
|
Gain recognized in earnings |
|
| (503,186 | ) |
|
| (81,339 | ) |
Warrants reclassified to equity |
|
| — |
|
|
| (34,945 | ) |
Balance at December 31 |
| $ | 5,000,752 |
|
| $ | 1,190,261 |
|
Derivative Financial Instruments
We do not use derivative instruments to hedge exposures to cash flow, market or foreign currency risk. The terms of convertible preferred stock and convertible notes that we issue are reviewed to determine whether or not they contain embedded derivative instruments that are required by ASC 815: “Derivatives and Hedging” to be accounted for separately from the host contract, and recorded at fair value. In addition, freestanding warrants are also reviewed to determine if they achieve equity classification. Certain stock warrants that we have issued did not meet the conditions for equity classification and are classified as derivative instrument liabilities measured at fair value. The fair values of these derivative liabilities are revalued at each reporting date, with the change in fair value recognized in earnings. See Note 10 for additional information.
Beneficial Conversion Feature of Convertible Notes
If the embedded conversion option in a convertible note is not required to be separated and accounted for as a derivative instrument liability, then ASC 470: “Debt with Conversion and Other Options” requires separate recognition of any beneficial conversion feature (“BCF”) related to the convertible instrument. Accordingly, we recognized a BCF related to the Secured Convertible Notes that we issued in 2013 and 2014. The BCF for the convertible instruments is recognized by allocating to additional paid-in capital a portion of the proceeds received equal to the intrinsic value of the conversion feature. Generally, the intrinsic value is calculated at the commitment
F-13
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(cont.)
date as the difference between the effective conversion price and the fair value of the common stock or other securities into which the security is convertible, multiplied by the number of shares into which the security is convertible. When certain other securities, such as warrants, are issued in conjunction with the convertible security, the proceeds are allocated among the different components. The portion of the proceeds allocated to the convertible security is divided by the contractual number of the conversion shares to determine the effective conversion price which is then used to measure the BCF. The value of the BCF recognized is limited to the amount of proceeds received that is initially allocated to the convertible security. See Note 10 for additional information related to the BCF resulting from our Secured Convertible Notes.
Beneficial Conversion Feature of Series A Preferred Stock
Equity instruments that contain a BCF are recorded as a deemed dividend to the holders of the convertible equity instruments. The deemed dividend amount is recorded as an increase to additional paid-in capital. For the year ended December 31, 2014, we recorded a deemed dividend of $316,366 related to the BCF resulting from the issuance of Series A Preferred Stock preemptive shares. See Note 10 for additional information related to the BCF that resulted from the issuance of Series A Preferred Stock upon exercise of the preemptive rights.
Segment Information
We have one reportable operating segment that manufactures xGnP® graphene nanoplatelets and products produced therefrom, that conducts research on graphene nanoplatelets and related products and licenses our technology as appropriate. As of December 31, 2014, we shipped products on a worldwide basis, but all of our assets were located within the United States.
JOBS Act
In April 2012, the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, was enacted. Section 107 of the JOBS Act provides that an “emerging growth company,” or EGC, can take advantage of the extended transition period for complying with new or revised accounting standards. Thus, an EGC can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies.
We are in the process of evaluating the benefits of relying on other exemptions and reduced reporting requirements under the JOBS Act. Subject to certain conditions, as an EGC, we intend to rely on certain of these exemptions, including exemptions from the requirement to provide an auditor’s attestation report on our system of internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and from any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion and analysis. We will remain an EGC until the earlier of: the last day of the fiscal year in which we have total annual gross revenues of $1.0 billion or more; the last day of the fiscal year following the fifth anniversary of the date of the completion of this offering; the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
NOTE 3 — INVENTORY
As of December 31, the following amounts were included in inventory:
|
| 2014 |
| 2013 | ||
Raw materials |
| $ | 69,600 |
| $ | 34,390 |
Finished goods |
|
| 62,859 |
|
| 32,770 |
Total |
| $ | 132,459 |
| $ | 67,160 |
F-14
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 4 — PROPERTY, PLANT AND EQUIPMENT
Property, plant and equipment as of December 31 consist of the following:
|
| Depreciable |
| 2014 |
| 2013 | ||||
Plant and equipment not yet placed in service |
|
|
| $ | 820,861 |
|
| $ | 184,903 |
|
Leasehold improvements |
| 5–10 |
|
| 382,230 |
|
|
| 324,302 |
|
Lab equipment |
| 3–7 |
|
| 893,068 |
|
|
| 717,978 |
|
Production and other equipment |
| 3–7 |
|
| 5,070,344 |
|
|
| 4,711,891 |
|
Software |
| 3 |
|
| 39,144 |
|
|
| 39,144 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| 7,205,647 |
|
|
| 5,978,218 |
|
Less accumulated depreciation and amortization |
|
|
|
| (2,553,174 | ) |
|
| (1,740,934 | ) |
|
|
|
|
|
|
|
|
|
|
|
Net property, plant and equipment |
|
|
| $ | 4,652,473 |
|
| $ | 4,237,284 |
|
Depreciation and amortization expense on property and equipment, including leased assets, for the years ended December 31, 2014 and 2013, was $866,889 and $717,408 respectively. These amounts are included as part of our Statement of Operations in Cost of Goods Sold, Research and Development, and General and Administrative Expenses - $681,983, $142,785, $42,121 and $545,000, $119,428, $52,980 respectively in 2014 and 2013.
Property and equipment under capital leases included above consists of the following at December 31:
|
| 2014 |
| 2013 | ||||
Lab equipment |
| $ | 160,202 |
|
| $ | 160,202 |
|
Production and other equipment |
|
| 986,230 |
|
|
| 595,908 |
|
|
|
|
|
|
|
|
|
|
|
|
| 1,146,432 |
|
|
| 756,110 |
|
Less accumulated depreciation |
|
| (128,220 | ) |
|
| (14,864 | ) |
|
|
|
|
|
|
|
|
|
Net property, plant and equipment under capital leases |
| $ | 1,018,212 |
|
| $ | 741,246 |
|
NOTE 5 — INTANGIBLE ASSETS
Intangible assets and related accumulated amortization as of December 31, 2014 and 2013 are as follows:
|
| Carrying Amount |
| Less Accumulated Amortization |
| Net Carrying Amount | ||||
Licenses |
| $ | 137,308 |
| $ | (55,908 | ) |
| $ | 81,400 |
Patents |
|
| 268,805 |
|
| (27,264 | ) |
|
| 241,541 |
Trademarks, other intangibles |
|
| 5,698 |
|
| (1,505 | ) |
|
| 4,193 |
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2014 |
| $ | 411,811 |
| $ | (84,677 | ) |
| $ | 327,134 |
|
|
|
|
|
|
|
|
|
|
|
Licenses |
| $ | 134,698 |
| $ | (46,394 | ) |
| $ | 88,304 |
Patents |
|
| 169,771 |
|
| (31,292 | ) |
|
| 138,479 |
|
|
|
|
|
|
|
|
|
|
|
Trademarks, other intangibles |
|
| 4,098 |
|
| (1,136 | ) |
|
| 2,962 |
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2013 |
| $ | 308,567 |
| $ | (78,822 | ) |
| $ | 229,745 |
Amortization expense of $25,095 and $18,946 was recorded for the years ended December 31, 2014 and 2013, respectively. Amortization expense for the next five years is estimated to be approximately $30,000 annually.
F-15
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 6 — LINE OF CREDIT
We previously had a $1,000,000 revolving line of credit which bore interest at 8%, and which was collateralized by substantially all assets of the Company. On March 18, 2013, the line of credit was paid down through the issuance of $700,000 of Secured Convertible Notes, bearing interest at 12% per annum and having a due date of March 18, 2018. See Note 10 for additional information. The remaining balance outstanding under the line of credit of $103,447 as of December 31, 2013, was repaid in January 2014 and the line of credit is no longer available to us.
As consideration for providing the line of credit, we issued warrants on October 8, 2012 to purchase 5,000 shares of common stock. The warrants were exercisable at a price of $20 per share, or if lower, the price per share at which we priced our next offering of common stock. The warrants expire on October 8, 2027. Derivative liability classification was required at inception because the variable exercise price is not consistent with the instruments being indexed only to the Company’s own stock. The fair value of the warrants was estimated to be $63,060 at inception using a lattice model with the following inputs: estimated value of underlying stock: $20.00; exercise price: $20.00; equivalent risk-free interest rate: 1.05%; expected term: 15 years; equivalent stock price volatility: 43.48%; expected dividend yield: none. Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions.
On March 18, 2013, we entered into our next offering of common stock for $12.00 per share, at which time, based on the terms of the warrants, the exercise price of the warrants was reduced from $20.00 to $12.00. The resulting increase in fair value of $6,915 was recorded in the Statements of Operations as a component of the Change in fair value of derivative liability — warrants. Once the exercise price of the Stock Warrants was fixed at $12.00, the Stock Warrants met the definition of being indexed only to the Company’s own stock and achieved equity classification. Accordingly, on March 18, 2013, the fair value of the Stock Warrants, estimated at that time to be $34,945, was reclassified from a derivative liability to equity.
NOTE 7 — ACCOUNTS PAYABLE AND OTHER CURRENT LIABILITIES
As of December 31, 2014 and 2013, our accounts payable and other current liabilities consisted of the following:
|
| 2014 |
| 2013 | ||
Accounts payable |
| $324,096 |
| $1,177,317 | ||
Product marketing allowance |
| 35,045 |
| 36,993 | ||
Accrued compensation |
| 210,800 |
| 151,832 | ||
Accrued expenses |
| 195,390 |
| 15,000 | ||
401(k) employer contribution expense |
| 54,207 |
| — | ||
Current portion of deferred compensation (see Note 9) |
| 254,400 |
| 120,000 | ||
|
|
|
|
|
|
|
Accounts payable and other liabilities |
| $ | 1,073,938 |
| $ | 1,501,142 |
NOTE 8 — DEFERRED REVENUE
During 2013, we invoiced $600,000, of which $412,500 was received and $187,500 of which was included as accounts receivable as of December 31, 2013, to a single large customer in exchange for a commitment to ship a variety of our products during the period from January 1, 2014 to June 30, 2014. The invoiced amounts were recorded as Deferred Revenue. The related services were performed, and the revenue recognized, during 2014.
NOTE 9 — DEFERRED COMPENSATION — PHANTOM STOCK
Certain key employees have deferred compensation through a phantom stock plan (the “plan”) established by the Company in 2009. These key employees can elect to defer up to 100% of their compensation for a specific period. Amounts deferred are allocated quarterly into phantom units of our stock and are 100% vested. The cost of each phantom unit is established annually by the plan administrator prior to the beginning of each plan year. The cost established by the plan administrator for the plan years December 31, 2014 and 2013 was $12 and $20 per unit, respectively. Amounts accumulated under the plan are paid out as elected by the employee and accepted by the plan administrator. The value of accumulated phantom stock units at payout for
F-16
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 9 — DEFERRED COMPENSATION — PHANTOM STOCK(cont.)
events other than a change in control of the Company is based on the average per share value of common stock sales for the previous twelve months or based on an independent appraisal if no such stock sales have occurred within a twelve month period. As of December 31, 2014 and 2013, the fair value of the phantom stock units was deemed to be $12 per unit and the liability for outstanding units was adjusted based on that value. In the event of a change of control of the Company, accumulated phantom stock units are to be paid to the employee based on the per share value of common stock at that time. Provisions for payout of the value of the units also exist in the event of death, disability or separation from service as summarized in the plan.
Phantom stock unit activity for the years ended December 31, 2014 and 2013 was as follows:
|
| 2014 |
| 2013 | ||||
Balance, beginning of year |
| $ | 374,400 |
|
| $ | 623,990 |
|
Phantom stock paid out during the year |
|
| (120,000 | ) |
|
| — |
|
Change in value of phantom units outstanding |
|
| — |
|
|
| (249,590 | ) |
|
|
|
|
|
|
|
|
|
Balance, end of year |
|
| 254,400 |
|
|
| 374,400 |
|
Less current portion |
|
| (254,400 | ) |
|
| (120,000 | ) |
|
|
|
|
|
|
|
|
|
Non-current portion |
| $ | — |
|
| $ | 254,400 |
|
|
|
|
|
|
|
|
|
|
Phantom units outstanding |
|
| 21,200 |
|
|
| 31,200 |
|
The amount outstanding at December 31, 2014 is to be paid out in early 2015. The 31,200 units outstanding at December 31, 2013 were valued at an estimated market price of $12 per share as of December 31, 2013, down from $20 per share on December 31, 2012. This reduction in the estimated payout value of these shares resulted in a decrease to compensation expense of $249,590 during 2013.
NOTE 10 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS
On March 18, 2013 we entered into a series of agreements with our former Chief Executive Officer, Michael R. Knox and Aspen Advance Opportunity Fund, LP (“AAOF”) to fund continuing operations of the business. Under these agreements, Mr. Knox and AAOF agreed to provide financing to the Company in the form of Secured Convertible Notes as well as optional lease financing. The agreements provided for Mr. Knox to lend the Company $700,000, to be funded by converting advances he had made under a revolving line of credit, and for AAOF to lend the Company a minimum of $2.5 million and, at the option of AAOF, a maximum of $5 million, with a provision that up to 50% of the AAOF funding could be funded under lease financing arrangements.
Subsequently, on July 12, 2013, these agreements were amended and restated to provide for expanded financing commitments from AAOF as well as a related entity, XGS II, LLC (“XGS II”). Under the terms of these amended agreements, the financing commitments from AAOF were increased to a minimum of $6 million and, at the option of AAOF, a maximum of $10 million, of which a maximum of $3 million could be provided in the form of lease financing. Also under these amended agreements, XGS II agreed to provide a minimum of $250,000 and, at the option of XGS II, a maximum of $750,000. All of the funding with the exception of the Knox financing, was scheduled according to minimum financing commitments on calendar dates ranging from the time the agreements were executed to a period of eighteen months into the future. With the exception of any lease financing, the financing provided by Knox, AAOF, and XGS II takes the form of Secured Convertible Notes with identical terms and provisions.
On January 15, 2014, we amended our agreements with AAOF and XGS II to revise the terms and amounts related to the timing of future investments from AAOF and XGS II by increasing the XGS II optional funding level to $1 million and extending the optional financing periods for both AAOF and XGS II through the end of 2014. In connection with the amendments, we also i) transferred all of our intellectual property into a newly-created, wholly-owned subsidiary called XGS IP, LLC, ii) revised certain terms of the Company’s By-Laws to allow for special stockholder meetings to be called by holders of ten percent or
F-17
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 10 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(cont.)
more of the Company’s stock and to clarify certain voting provisions, and iii) amended the previously issued Certificate of Designation of Series A Convertible Preferred Stock to clarify the conditions related to Mandatory Conversion into Common Stock upon listing of the Company’s stock on a Qualified National Exchange and to add a provision that would exempt holders of Series A from future mandatory financing participation.
The Secured Convertible Notes issued under these agreements bear interest at 12% per annum and have a due date of March 18, 2018. These notes have provisions providing an option to the Company, at its choice, to accrue the interest payments on the notes through December 31, 2014 and add the accrued interest to the principal of the notes. After December 31, 2014, the note holder, at its option, can choose to receive ongoing interest payments: 1) in the form of cash; 2) receive all or a portion of currently due interest in the form of Series A Convertible Preferred Stock at a price per share (currently $12) equal to the then effective Series A Original Issue Price as defined in the Company’s Certificate of Designation of Series A Convertible Preferred Stock; 3) receive all or a portion of currently due interest payments in the form of any other series of Preferred Stock outstanding at the time interest is due; or 4) elect to accrue such interest and add it to the balance of the notes. These notes are secured by all of the assets, both tangible and intangible, of the Company. Further, these notes have provisions allowing the conversion of these notes into Series A Convertible Preferred Stock (or such other form of preferred stock as may be outstanding at that time), at the option of the holder at a conversion price of $12.00 per share. In addition, these notes contain various affirmative and negative covenants that provide for various reporting, information, notice, maintenance of business, insurance, audits and reappraisal requirements. These covenants also prohibit the Company from issuing new secured debt that would have security or liquidation provisions senior to, or equivalent to, the Secured Convertible Notes, and these covenants also prohibit the issuance of dividends or stock redemptions, creation of subsidiaries, certain transactions with subsidiaries, or transfer of assets without the permission of the holders.
The embedded conversion feature in the Secured Convertible Notes was analyzed under ASC 815 to determine if it achieved equity classification or required bifurcation as a derivative instrument. In order to be a derivative, one of the criteria is that the embedded component must be net settleable. The Company’s common stock is not publicly traded, there is no mechanism outside the notes that would permit the holder to achieve net settlement and the underlying shares are not readily convertible to cash. Accordingly, the embedded conversion feature does not meet the definition of a derivative, and therefore, does not require bifurcation from the host instrument. Certain default put provisions were not considered to be clearly and closely related to the host instrument but management concluded that the value of these default put provisions wasde minimus at December 31, 2014. Management will reconsider the value of the default put provisions each reporting period to determine if the value becomes material to the consolidated financial statements.
In conjunction with the sale of the Secured Convertible Notes to AAOF and XGS II during 2013, Stock Warrants to purchase an aggregate of 447,916 shares of Series A Convertible Preferred Stock were issued that vest as the financing occurs at a rate of one Stock Warrant for every $24.00 funded. The Stock Warrants have an exercise price of $12.00 per share and are indexed to Series A Convertible Preferred Stock, which contains a feature that requires adjustment of the exercise price of the Preferred Stock if the Company issues any Common Stock, or common stock equivalents, for a price less than the exercise price in effect immediately before the issuance. The Stock Warrants expire on the earlier of March 18, 2023 or the acquisition of the Company by another entity.
Although the Stock Warrants did not fall within the scope of ASC 480, they required derivative liability accounting because the conversion price reset protection terms in the underlying Series A Convertible Preferred Stock was not consistent with the definition for financial instruments indexed only to a company’s own stock. As such, they are required to be marked to fair value each reporting period.
On January 15, 2014, we consolidated the Secured Convertible Notes issued in 2013 to AAOF and XGS II, together with accrued interest, into a single note for each lender with terms and conditions similar to those previously issued to AAOF and XGS II. We also exchanged existing warrants to purchase 447,916 shares of Series A Preferred Stock for new warrants to purchase 916,666 shares of Series A Preferred Stock. As of January 15, 2014, 434,314 Stock Warrants were vested and their fair value was estimated to be $2,525,822 using a lattice model with the following inputs: estimated value of underlying
F-18
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 10 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(cont.)
stock: $12.00; equivalent risk-free interest rate: 1.40%; expected term: 10 years; equivalent stock price volatility: 37.16%; expected dividend yield: none. Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. The fair value of the Stock Warrants immediately prior to the exchange was $1,205,081, resulting in an increase in fair value of $1,320,741.
The cash flows of the newly consolidated Secured Convertible Notes plus the fair value of the additional Stock Warrants issued exceeded the cash flows of the old Secured Convertible Notes by more than 10%. Therefore, in accordance with ASC 470 “Debt”, the modification of the notes was accounted for as a debt extinguishment. Because the conversion price of the Secured Convertible Notes is the same as the estimated value of the underlying stock on the date of the modification, no beneficial conversion feature was recognized. The difference of $2,193,029 between the carrying amounts of the original Secured Convertible Notes and the fair value of the newly consolidated Secured Convertible Notes plus the increase in fair value resulting from the issuance of additional Stock Warrants of $1,320,741 was recorded as a loss on extinguishment of $3,513,770.
During the year ended December 31, 2014, we issued an additional $5,500,000 in Secured Convertible Notes to AAOF and XGS II with terms similar to those issued previously. Stock Warrants indexed to 454,166 shares of Series A Preferred Stock vested as result of the 2014 financings. In accordance with the terms of the January 15, 2014 amendment, the previously-issued Stock Warrants associated with these financings vest as the financings occur at a rate of one Stock Warrant for every $12.00 funded. As of December 31, 2014, all 833,332 Stock Warrants issued in conjunction with the Secured Convertible Notes were vested. The initial fair values of the Stock Warrants that vested during the year ended December 31, 2014 were estimated using a binomial lattice model with the following assumptions:
Fair value of underlying stock |
| $12.00 |
Equivalent risk free interest rate |
| 1.23% – 1.41% |
Expected term (in years) |
| 9.05 – 9.99 |
Equivalent stock price volatility |
| 33.79% – 37.16% |
Expected dividend yield |
| — |
Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. Because the Company is not publicly traded, there is currently no active market for the Company’s stock. Therefore, the expected volatility of the Company’s stock was developed using historical volatility of a peer group for a period equal to the expected term of the Stock Warrants. The estimated value of our common stock was determined on the basis of recent third-party investment transactions that established a price of $12 per share as the value at which outsiders were willing to invest in our stock.
After recording the Stock Warrants at fair value, a determination was made as to whether, in accordance with ASC 470, there was a BCF associated with the Secured Convertible Notes. A BCF is a non-detachable conversion feature that is in-the-money at the commitment date. An option is in-the-money if its effective conversion price is less than the current fair value of the share. For purposes of measuring a BCF, the effective conversion price is based on the proceeds allocated to the convertible debt instrument after considering allocations to detachable warrants.
The cash proceeds from the AAOF and XGS II financings during the year ended December 31, 2014 were allocated first to the derivative liabilities resulting from the Stock Warrants, at their fair values, then to the beneficial conversion feature, with the residual allocated to the host debt contracts, as follows:
|
| Allocation | |
Derivative liabilities – warrants |
| $ | 2,520,527 |
Beneficial Conversion Feature |
|
| 2,520,527 |
Secured Convertible Notes – initial carrying value |
|
| 458,946 |
Total allocated proceeds |
| $ | 5,500,000 |
F-19
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 10 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(cont.)
The amount allocated to the derivative liabilities and the BCF was recorded as a discount on the Secured Convertible Notes and is being amortized to interest expense over the remaining term of the notes using the effective interest method. The effective interest rates on the Secured Convertible Notes are approximately 28% – 85%. Total interest expense of $1,360,308 and $402,962 was recognized for the year ended December 31, 2014 and 2013, respectively.
At December 31, 2014, the total funding from the AAOF and XGS II agreements was $10,026,091 and $1,000,000, respectively. Because the lease commitments of $1,026,091 exceed the $1,000,000 initial commitment by $26,091, AAOF increased the maximum potential funding from $10,000,000 to $10,026,091.
Samsung Note
On January 15, 2014, we sold $3,000,000 of Secured Convertible Notes to SVIC No. 15 New Technology Business Investment L.L.P, a subsidiary of the Samsung Group (“Samsung”). These notes had terms and conditions that were substantially similar to those previously issued to Knox, AAOF, and XGS II. In connection with the sale of the notes, the Company issued to Samsung a total of 100,000 Stock Warrants with a term of 4 years that are exercisable into shares of Series A Convertible Preferred Stock at a price of $12.00. These Stock Warrants vest in four installments on the first, second, third and fourth anniversary of the issuance of the Stock Warrants according to the following formula: On each anniversary, the warrant vests according to the ratio of total Samsung payments to us during the previous twelve-month period divided by 250. Thus, the warrants will vest proportionately in relation to the first $25 million, or fraction thereof, that Samsung pays, in cash, to us over the four years beginning January 15, 2014. Samsung payments are defined as future total cash payments, including licensing, royalties and product purchases but not joint development or purchase of Secured Convertible Notes. As of December 31, 2014, and through January 15, 2015, and through the date our consolidated financial statements were issued, no payments had been made by Samsung and none of the Stock Warrants associated with the Samsung note had vested. If and when Samsung makes payments to the Company in the future, the Stock Warrants will be measured at fair value and expensed at that time.
The following table summarizes the Secured Convertible Notes outstanding and the number of shares of Series A Convertible Preferred Stock into which the Notes may be converted at December 31, 2014:
|
| AAOF |
| XGS II |
| KNOX |
| SAMSUNG |
| Total | ||||||||
Original face value of notes – issuance |
| $ | 9,268,906 |
|
| $ | 1,048,775 |
|
| $ | 772,998 |
| $ | 3,000,000 |
| $ | 14,090,679 |
|
Accrued interest added to face value |
|
| 954,715 |
|
|
| 113,844 |
|
|
| 93,723 |
|
| 351,468 |
|
| 1,513,750 |
|
Amount due – December 31, 2014 |
|
| 10,223,621 |
|
|
| 1,162,619 |
|
|
| 866,721 |
|
| 3,351,468 |
|
| 15,604,429 |
|
Unamortized (discount) |
|
| (4,890,785 | ) |
|
| (291,003 | ) |
|
| — |
|
| — |
|
| (5,181,788 | ) |
Carrying value |
| $ | 5,332,836 |
|
| $ | 871,616 |
|
| $ | 866,721 |
| $ | 3,351,468 |
| $ | 10,422,641 |
|
Series A Shares |
|
| 851,968 |
|
|
| 96,885 |
|
|
| 72,227 |
|
| 279,289 |
|
| 1,300,369 |
|
Preemptive Rights
On January 15, 2014, as part of our financing agreements with Samsung, AAOF and XGS II, we agreed to allow all stockholders to purchase one share of Series A Convertible Preferred Stock at a price of $12.00 per share for every two shares of Series A Convertible Preferred Stock or Common Stock owned by the stockholder. In addition, for every two preemptive shares purchased, the Company issued the stockholder a warrant to purchase one additional share of Series A Convertible Preferred Stock with the same terms as the warrants issued to AAOF and XGS II. The Company also agreed to issue warrants with the same terms to those stockholders who exercised preemptive rights in October 2013. These preemptive rights expired on March 31, 2014.
During the quarter ended March 31, 2014, 101,000 shares of Series A Convertible Preferred Stock were sold to existing stockholders at a price of $12.00 per share under the January 15, 2014 preemptive rights offering. In addition, Stock Warrants indexed to 56,054 shares of Series A Convertible Preferred Stock were issued in conjunction with these stock purchases, including 5,554 warrants related to the preemptive rights exercised in October 2013.
F-20
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 10 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(cont.)
The proceeds from the exercise of the preemptive rights were allocated first to the derivative liabilities resulting from the Stock Warrants, at their fair values, then to the beneficial conversion feature, with the residual allocated to the Series A Preferred Stock, as follows:
Derivative liabilities – warrants |
| $ | 316,366 |
Beneficial Conversion Feature |
|
| 316,366 |
Series A Preferred Stock |
|
| 712,552 |
Total allocated proceeds |
| $ | 1,345,284 |
ASC 470 provides for a BCF to be accreted over the longer of the term to maturity or the period to when the holder can first convert the instrument. Because the Series A Convertible Preferred Stock has no stated maturity and is convertible on the issuance date, the BCF is accreted immediately by crediting the Series A Convertible Preferred Stock, with an offsetting charge to retained earnings or, in the absence of retained earnings, to paid-in capital.
Warrants
As discussed above, 833,332 vested Stock Warrants were issued in conjunction with our Secured Convertible Notes. In addition, 83,334 vested Stock Warrants were issued with our lease financings. Also, certain stockholders exercised their preemptive rights to purchase Series A Convertible Preferred Stock which resulted in the issuance of 56,054 Stock Warrants.
The Stock Warrants require liability classification because the conversion price reset protection terms in the underlying Series A Convertible Preferred Stock is not consistent with the definition for financial instruments indexed only to a company’s own stock.
The Stock Warrants issued as consideration for the equipment financing leases are recorded as derivative liabilities at fair value. The initial value of these warrants was recorded as a reduction of the capital lease obligation and is being amortized as part of the effective interest cost on the capital lease obligation (see Note 14).
As discussed in Note 6 above, in conjunction with a line of credit, the Company also issued Stock Warrants which were recorded as derivative liabilities as of December 31, 2012 but which were reclassified to equity on March 18, 2013.
The Stock Warrants issued in conjunction with the Secured Convertible Notes, the equipment financing leases and the preemptive rights are derivative liabilities which require re-measurement at fair value each reporting period. The Company estimated the fair value of these derivative liabilities as of December 31, 2014 and 2013, using a lattice model and the following assumptions:
|
| 2014 |
| 2013 | |
Fair value of underlying stock |
| $12.00 |
|
| $12.00 |
Equivalent risk-free interest rate |
| 1.25 | % |
| 1.32% – 1.37% |
Expected term (in years) |
| 9.05 |
|
| 9.22 – 9.53 |
Equivalent stock price volatility |
| 34.27 | % |
| 37.02% – 37.10% |
Expected dividend yield |
| — |
|
| — |
Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. Because the Company is not publicly traded, there is no active market for the Company’s stock. Therefore, the expected volatility of the Company’s stock was developed using historical volatility for a peer group for a period equal to the expected term of the Stock Warrants. The estimated value of our common stock was determined on the basis of recent third-party investment transactions that established a price of $12 per share as the value at which outsiders were willing to invest in our stock.
F-21
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 10 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(cont.)
The following table summarizes the fair value of the derivative liabilities as of December 31, 2014 and 2013:
|
| 2014 |
| 2013 | ||
Warrants issued with Secured Convertible Notes |
| $ | 4,284,161 |
| $ | 1,043,347 |
Warrants issued with equipment financing leases |
|
| 428,417 |
|
| 146,914 |
Warrants issued with preemptive rights |
|
| 288,174 |
|
| — |
|
|
|
|
|
|
|
Total derivative liabilities |
| $ | 5,000,752 |
| $ | 1,190,261 |
|
|
|
|
|
|
|
Shares indexed to derivative liabilities |
|
| 972,720 |
|
| 214,149 |
Changes in the fair value of Derivative Liabilities, carried at fair value, are reported as “Change in fair value of derivative liability - warrants” in the Statement of Operations, and were as follows:
|
| Year ended December 31, | ||||||
|
| 2014 |
| 2013 | ||||
Warrants issued with line of credit |
| $ | — |
|
| $ | (25,920 | ) |
Warrants issued with Secured Convertible Notes |
|
| (439,579 | ) |
|
| (54,837 | ) |
Warrants issued with equipment financing leases |
|
| (35,417 | ) |
|
| (582 | ) |
Warrants issued with preemptive rights |
|
| (28,190 | ) |
|
| — |
|
|
|
|
|
|
|
|
|
|
Total Derivative (Gain) |
| $ | (503,186 | ) |
| $ | (81,339 | ) |
NOTE 11 — SERIES A CONVERTIBLE PREFERRED STOCK
The Company is authorized to issue up to 3,000,000 shares of Series A Convertible Preferred Stock (“Series A”). Each share of the Series A, which has a liquidation preference of $12.00 per share, is convertible at any time, at the option of the holder, into one share of Common Stock at the lower of: $12.00 per share, or 80% of the price at which the Company sells any equity or equity-linked securities in the future. The Series A also participates on a pro-rata basis with Common stock on residual liquidation rights after the Series A liquidation preference has been satisfied. The Series A also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. The Series A is subject to mandatory conversion into Common Stock upon the listing of the Company’s Common Stock on a Qualified National Exchange.
The Company concluded that the Series A is more similar to an equity-type instrument than a debt-type instrument. Accordingly, the embedded conversion option is clearly and closely related to an equity-type host and the conversion option does not require classification and measurement as a derivative financial instrument.
NOTE 12 — INCENTIVE STOCK OPTION PLAN
We have established an incentive stock option plan (the “Plan”) under which the Company may grant key employees and directors options to purchase common stock of the Company at not less than fair market value as of the grant date. Options for up to 600,000 shares may be awarded under the Plan. Each option is exercisable into one share of common stock of the Company. The Plan expires in December 2017. The fair value of the options granted was estimated on the dates of grant using the Black Scholes option-pricing model. As of December 31, 2014 and 2013, respectively, 464,750 and 152,500 option shares have been granted. Vesting of the options ranges from immediately to 20% per year, with most options vesting on a straight-line basis over a three or four year period from the date issued. (See discussion of CEO options below.) Rights to exercise the options vest immediately upon a change in control of the Company or termination of the employee’s continuous service due to death or disability. The options expire at various dates through June 2021.
F-22
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 12 — INCENTIVE STOCK OPTION PLAN(cont.)
As of December 31, 2014, we had outstanding options to purchase 464,750 shares of Common Stock. As of December 31, 2013, we had 152,500 outstanding options. The following table shows the stock option activity during the years ended December 31, 2014 and 2013:
|
| 2014 |
| 2013 | ||||||||
|
| Number |
| Weighted |
| Number |
| Weighted | ||||
|
|
|
|
|
|
|
|
|
|
|
|
|
Options outstanding at beginning of year |
| 152,500 |
|
| $ | 12.13 |
| 50,200 |
|
| $ | 15.92 |
Changes during the year: |
|
|
|
|
|
|
|
|
|
|
|
|
Granted – above market price |
| — |
|
|
| — |
| 50,000 |
|
|
| 13.20 |
Granted – at market price |
| 328,500 |
|
|
| 12.00 |
| 85,000 |
|
|
| 12.47 |
Exercised |
| (1,667 | ) |
|
| 12.00 |
| — |
|
|
| — |
Expired |
| (14,583 | ) |
|
| 12.00 |
| — |
|
|
| — |
Cancelled |
| — |
|
|
| — |
| (32,700 | ) |
|
| 20.47 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Options outstanding at end of year |
| 464,750 |
|
|
| 12.04 |
| 152,500 |
|
|
| 12.13 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Options exercisable at end of year |
| 60,333 |
|
|
| 11.73 |
| 30,000 |
|
|
| 10.87 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average fair value of options granted during the year |
|
|
|
| $ | 12.00 |
|
|
|
| $ | 12.74 |
Costs incurred in respect of stock based compensation for employees and directors, for the years ended were $644,788 and $233,002, respectively. Unrecognized compensation cost as of December 31, 2014 was $1,589,463, which amount is expected to be recognized over approximately 47 months.
As of December 31, 2014, the aggregate intrinsic value of currently exercisable stock options was approximately $40,000. The intrinsic value of each option share is the difference between the fair market value of our common stock and the exercise price of such option share to the extent it is “in-the-money”. Aggregate intrinsic value represents the value that would have been received by the holders of in-the-money options had they exercised their options on the last trading day of the year and sold the underlying shares at the closing stock price on such day. The intrinsic value calculation is based on the assumed market value of our common stock on December 31, 2014 of $12 per share. The total number of in-the-money options outstanding and exercisable as of December 31, 2014 was 10,000. The exercise prices of the remaining stock options outstanding and expected to vest were all either equal to or greater than the fair value of our common stock. Therefore, these options currently have no intrinsic value.
The following table presents changes in the number of non-exercisable options during 2014:
Non-exercisable options |
| Number |
| Average Exercise Price | ||
| 122,500 |
|
| $ | 12.44 | |
| 328,500 |
|
| $ | 12.00 | |
| (14,583 | ) |
| $ | 12.00 | |
| (32,000 | ) |
| $ | 11.73 | |
|
|
|
|
|
|
|
| 404,417 |
|
|
|
| |
F-23
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 12 — INCENTIVE STOCK OPTION PLAN(cont.)
The Company’s new Chief Executive Officer commenced his employment on January 6, 2014. As part of his compensation, on January 6, 2014, he was awarded 220,000 stock options, with an exercise price of $12 per share. Of the total option award, 160,000 options vest over the four year period ending January 6, 2018. The grant date fair value of these options was $987,648, which is expected to be recognized over the four year vesting period. The remaining 60,000 options are subject to certain performance targets being met. The probability of those targets being met is periodically re-assessed and compensation cost begins to be recognized when the targets are considered probable of achievement. As of December 31, 2014, $17,070 of expense had been recognized when achievement of certain targets was deemed probable.
The total fair value of options granted during the years ended December 31, 2014 and 2013 was $1,662,047 and $473,426, respectively.
The fair value of the options granted in 2014 and 2013 was estimated on the grant date using the Black Scholes option-pricing model with the following assumptions:
|
| 2014 |
| 2013 |
Fair value of underlying stock |
| $12.00 |
| $12.00 |
Expected option life |
| 4.29 years – 6.00 years |
| 4.33 – 4.77 years |
Expected stock price volatility |
| 26.03% – 52.89% |
| 30.90% – 48.00% |
Risk free interest rate |
| 1.63% – 2.04% |
| 1.05% |
Expected dividend yield |
| 0.00% |
| 0.00% |
The following table presents summary information concerning the options outstanding as of December 31, 2014:
Exercise prices |
| Number |
| Number |
| Weighted |
$8.00 |
| 10,000 |
| 10,000 |
| 2.92 |
$12.00 |
| 404,750 |
| 30,333 |
| 6.91 |
$13.20 |
| 50,000 |
| 20,000 |
| 6.42 |
|
| 464,750 |
| 60,333 |
| 6.65 |
NOTE 13 — STOCK WARRANTS
As described in Note 10, we issued a total of 916,666 Stock Warrants to AAOF and XGS II in connection with our Secured Convertible Notes and equipment lease financing, 100,000 Stock Warrants to Samsung in connection with Secured Convertible Notes issued to them and 56,054 Stock Warrants to holders of Series A Preferred Stock in connection with their exercise of preemptive rights. As of December 31, 2014, all 972,720 Stock Warrants issued to AAOF, XGS II and holders of Series A Preferred Stock had vested. These warrants have an exercise price of $12.00 per share and expire on January 15, 2024. The 100,000 warrants issued to Samsung will vest only if, and when, payments are made by Samsung as described in Note 10. As described in Note 10, all of these warrants are accounted for as derivative liabilities at fair value.
In addition, we have 42,694 Common Stock Warrants outstanding at December 31, 2014 that were issued in 2012 and prior years and which are accounted for as equity instruments. In July 2014, 22,694 of these warrants expired unexercised. The remaining 20,000 warrants have exercise prices ranging from $8.00 to $20.00 and expire at various dates through 2027.
F-24
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 13 — STOCK WARRANTS(cont.)
The following table summarizes the warrants outstanding at December 31, 2014, all of which are exercisable:
Date Issued |
| Expiration Date |
| Exercise Price |
| Number of Warrants |
7/1/2009 |
| 7/1/2019 |
| $8.00 |
| 6,000 |
4/1/2010 |
| 4/1/2015 |
| $10.00 |
| 5,000 |
7/12/2011 |
| 7/12//2015 |
| $20.00 |
| 4,000 |
10/8/2012 |
| 10/8/2027 |
| $12.00 |
| 5,000 |
|
|
|
|
|
| 20,000 |
NOTE 14 — CAPITAL LEASES
As of December 31, 2014, we have capital lease obligations to AAOF (see Note 10) and other lessors as follows:
|
| 2014 |
| 2013 | ||||
Capital lease obligations |
| $ | 935,174 |
|
| $ | 731,122 |
|
Unamortized warrant discount |
|
| (249,611 | ) |
|
| (147,496 | ) |
Net obligations |
|
| 685,563 |
|
|
| 583,626 |
|
Short-term portion of obligations |
|
| (152,593 | ) |
|
| (128,563 | ) |
|
|
|
|
|
|
|
|
|
Long-term portion of obligations |
| $ | 532,970 |
|
| $ | 455,063 |
|
In connection with the lease agreements with AAOF, we issued to them preferred stock warrants with an initial fair value of $156,043 and $147,496 in 2014 and 2013, respectively (see Note 10). The initial fair value has been accounted for as a discount on the capital lease obligation and will be amortized as part of the interest expense on the leases. As discussed in Note 10, the warrants are accounted for as derivative instrument liabilities at fair value.
Our AAOF capital lease obligations are four year leases starting on January 1, 2014 and January 1, 2015. The effective interest rates on the leases are 50% and 32% for the leases executed in 2014 and 2013, respectively. The present value of the lease payments are more than 90% of the fair value of the equipment and therefore the leases were capitalized.
Our other capital leases expire at various dates through 2017, have average effective interest rates of 8% and contain bargain purchase options that allow us to purchase the leased property for a minimal amount upon the expiration of the lease term.
Future minimum lease payments under capital lease obligations are as follows:
For the year ending December 31: |
|
|
|
|
2015 |
| $ | 373,754 |
|
2016 |
|
| 347,033 |
|
2017 |
|
| 342,686 |
|
2018 |
|
| 130,356 |
|
Total future minimum lease payments |
|
| 1,193,829 |
|
Less amount representing interest |
|
| (508,266 | ) |
Present value of future minimum lease payments |
|
| 685,563 |
|
Less current maturities |
|
| (152,593 | ) |
Obligations under capital leases – long term |
| $ | 532,970 |
|
Property and equipment acquired under capital lease agreements is pledged as collateral to secure the performance of the future minimum lease payments above.
F-25
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 15 — INCOME TAXES
Deferred tax assets (liabilities) consist of the following at December 31:
|
| 2014 |
| 2013 | ||||
Current: |
|
|
|
|
|
|
|
|
| $ | — |
|
| $ | 180,000 |
| |
|
| 15,000 |
|
|
| 35,000 |
| |
Non-current: |
|
|
|
|
|
|
|
|
|
| 6,085,000 |
|
|
| 4,075,000 |
| |
|
| 75,000 |
|
|
| 75,000 |
| |
|
| (210,000 | ) |
|
| (145,000 | ) | |
|
| 410,000 |
|
|
|
|
| |
|
| 425,000 |
|
|
| 325,000 |
| |
|
|
|
|
|
|
|
|
|
| $ | 6,800,000 |
|
| $ | 4,545,000 |
| |
|
|
|
|
|
|
|
|
|
Deferred tax valuation allowance |
| $ | (6,800,000 | ) |
| $ | (4,545,000 | ) |
|
|
|
|
|
|
|
|
|
Net deferred tax asset |
| $ | — |
|
| $ | — |
|
Net operating loss carry forwards of $20,290,000 and $13,590,000 exist at December 31, 2014 and 2013, respectively. The primary difference between the net operating loss carry forwards and the accumulated deficit arises from certain stock option, warrants and other debt and equity transactions that are considered permanent differences These losses were incurred in the years 2006 through 2014 and will expire between 2026 and 2034 and their utilization may be limited if we experience significant ownership changes. The research and development credits will expire between 2028 and 2034. A rate of 30% has been used to calculate the deferred tax assets and liabilities based on the expected effective rate, net of applicable credits, upon reversal of the differences above. A valuation allowance has been established against the entire deferred tax asset at December 31, 2014 and 2013. The valuation allowance is considered a significant estimate subject to material change in the near term.
Below is a reconciliation of the statutory federal income tax rate to our effective tax rate for the fiscal years ended December 31, 2014 and 2013:
|
| 2014 |
| 2013 | ||
Federal tax provision |
| 34.0 | % |
| 34.0 | % |
State tax provision |
| 6.0 | % |
| 6.0 | % |
Valuation allowance |
| (40.0 | )% |
| (40.0 | )% |
|
| 0.0 | % |
| 0.0 | % |
We file income tax returns in the U.S. federal jurisdiction and in Michigan. Tax regulations within each jurisdiction are subject to the interpretation of the related tax laws and regulations and require significant judgment to apply. For federal and state purposes, we have open tax years for all years in which we have filed tax returns. We are not currently subject to any ongoing income tax examinations.
NOTE 16 — Customer, Supplier, country and Product Concentrations
Product Concentration
For 2014, we had concentrations of revenue from two products that were each greater than 10% of total revenues. Revenue from a grant was 28% of total revenues and revenue from one of the Company’s graphene nanoplatelets materials was 26%. For 2013, only one concentration was greater than 10% and this was licensing fees from one customer which represented 77% of total revenues. No other products in either year represented 10% or more of our revenue in 2014 or 2013. We attempt to minimize the risk associated with product concentrations by continuing to develop new products to add to its portfolio.
F-26
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 16 — Customer, Supplier, country and Product Concentrations(cont.)
Customer Concentration
Because of the nature of the applications that our customers are developing, we anticipate that we may see a few customers that account for the majority of our revenues. For 2014 and 2013 we had one customer whose revenue was 37% and 77%, respectively, of total revenues. In 2014 we also had another customer that represented 28% of total revenues. There were no other customers in either year that represented more than 10% of our revenues. The corresponding accounts receivable balances of customers from which revenues were in excess of 10% of total revenue were $3,389 and $187,500 at December 31, 2014 and 2013, respectively. At December 31, 2014 there were a total of five customers who had accounts receivable balances greater than 10% of our outstanding receivable balance, at December 31, 2013 there was one customer.
Country Concentration
We sell our products on a worldwide basis. International revenues in 2014 and 2013 were 17% and 8% of total revenues, respectively. All of these sales are denominated in U.S. dollars.
Shipments to only one country other than the United States accounted for more than 10% of revenue in the year ended December 31, 2014, that country (South Korea) represented approximately 12% of total revenues. There were no concentrations greater than 10% of revenue outside the United States in 2013.
Suppliers
We buy raw materials used in manufacturing from several sources. These materials are available from a large number of sources. A change in suppliers has no material effect on the Company’s operations. We did not have any purchases to one supplier that was more than 10% of total purchases in either 2014 or 2013.
NOTE 17 — RELATED PARTY TRANSACTIONS AND COMMITMENTS
We have a licensing agreement for exclusive use of patents and pending patents with Michigan State University (MSU), a stockholder via the MSU Foundation. During 2014 and 2013, we incurred $25,000 and $20,000, respectively, for royalties for these licenses. We have also entered into product licensing agreements with certain other stockholders. No royalty expenses have been recognized related to these agreements during 2014 and 2013.
The line of credit disclosed in Note 6 was from a shareholder and officer of the Company. As partial consideration for the line of credit, warrants for 5,000 shares of common stock with a fair value of $63,060 were issued in 2012. The subsequent conversion of this line of credit in 2013 resulted in the issuance of $700,000 of Secured Convertible Notes to the same shareholder and officer of the Company. The outstanding balance of this line of credit, $103,447 as of December 31, 2013, was repaid in January 2014. During 2013, a total of $32,269 in interest expense was paid to this shareholder and officer. An additional $69,152 was accrued as interest on the Secured Convertible Notes.
The financing arrangements disclosed in Note 10 were provided by two private funds that were formed for the sole purpose of investing in the Company by two investors affiliated with ASC-XGS, LLC, a shareholder of the Company. Under a Voting Agreement signed by our shareholders, a principal of the private funds serves as a director of the Company. During 2013, these two funds purchased $4,503,847 of Secured Convertible Notes and also provided $635,770 in equipment lease financing. In 2014, these two funds purchased $5,500,000 and also provided $390,321 in equipment lease financing.
We entered into an employment agreement with our CEO during December 2013. The agreement calls for an annual salary of $275,000. In addition, this officer will receive an aggregate of 220,000 options to purchase common stock at $12 per share. The options have a life of 8 years provided the employee remains employed by the Company. The options vest as follows: 160,000 over a 48 month period with 40,000 options vesting on the first anniversary of the agreement and the balance over the remaining 36 months; 40,000 vesting over a provisional time based schedule and 20,000 vest based on certain performance
F-27
XG SCIENCES, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
DECEMBER 31, 2014 AND 2013
NOTE 17 — RELATED PARTY TRANSACTIONS AND COMMITMENTS(cont.)
requirements. The agreement has no termination date but may be terminated by either party at any time. If the Company terminates the agreement without cause it will be liable for 6 months of severance pay.
We entered into an employment agreement with our senior vice president during January 2014. The agreement is for a period of three years and has a base salary of $160,000 per annum. The Company may terminate the agreement at any time, however if it terminates the agreement without cause it will be liable for severance pay at 50% of the employees monthly salary for the remainder of the agreement.
NOTE 18 — OPERATING LEASES
We lease our primary manufacturing facility, laboratory and administrative office under two separate operating leases expiring in December 2017 and March 2022. A second manufacturing facility is leased on a month to month basis. Total rent expense, including common area maintenance costs, was $351,406 and $348,459 during 2014 and 2013, respectively. Operating lease commitments for the next 5 years are as follows:
For the year ending December 31: |
|
|
|
2015 |
| $ | 324,798 |
2016 |
| $ | 331,380 |
2017 |
| $ | 338,046 |
2018 |
| $ | 226,422 |
2019 |
| $ | 230,685 |
NOTE 19 — RETIREMENT PLAN
We maintain a defined-contribution 401(k) retirement plan covering substantially all employees (as defined by our Plan Document). Employees may make voluntary contributions to the plan, subject to limitations based on IRS regulations and compensation. The plan allows for an employer match contribution. There were no employer match contributions for 2014 or 2013. The plan was deemed to be top-heavy for 2014. A plan is top heavy when more than 60% of plan assets are in the accounts of key employees. Key employees in 401(k) plans are defined by the IRS. For years when a plan is top heavy, the plan must make a “top heavy minimum contribution” to non-key employees. An amount of $54,207 is accrued at December 31, 2014 for this required contribution. This represents 3% of eligible compensation for non-key employees who were employed at December 31, 2014. The contribution has a 6 year vesting schedule and forfeitures reduce future lan expenses. The top heavy contribution is due by the close of 2015.
NOTE 20 — LETTER OF CREDIT
We are required by one of our lease agreements to maintain a letter of credit of approximately $190,000 through February 2022. To support this letter of credit, we are required to maintain an equivalent cash deposit. As of December 31, 2014, there were no amounts outstanding on the letter of credit. The cash deposit is restricted and classified as a non-current asset. As of December 31, 2014 and 2013, the cash deposit for the letter of credit was $190,915 and $190,526, respectively.
NOTE 21 — SUBSEQUENT EVENTS
On January 1, 2015, the former CEO, Michael R. Knox, was paid $254,400 in deferred compensation under the terms of the phantom stock plan. At the same time, he reinvested the entire payout in a Secured Promissory Note having a maturity on July 1, 2015. This Note bears interest at a rate of 12% per annum and has security provisions substantially identical to the other Secured Notes previously issued to Knox, AAOF, XGSII, and Samsung.
Subsequent events have been evaluated through the date these financial statements were submitted within the Registration Statement on Form S-1 to the Securities and Exchange Commission.
F-28
XG SCIENCES, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
|
| September30, 2015 |
| December 31, 2014 | ||||
|
| (unaudited) |
|
| ||||
ASSETS |
|
|
|
|
|
|
|
|
|
|
|
|
| ||||
CURRENT ASSETS |
|
|
|
|
|
|
|
|
| $ | 1,585,105 |
|
| $ | 2,088,866 |
| |
|
| 67,258 |
|
|
| 24,983 |
| |
|
| 211,052 |
|
|
| 132,459 |
| |
|
| 264,630 |
|
|
| 296,547 |
| |
|
|
|
|
| ||||
|
| 2,128,045 |
|
|
| 2,542,855 |
| |
|
|
|
|
|
|
|
|
|
PROPERTY, PLANT AND EQUIPMENT, NET |
|
| 4,169,153 |
|
|
| 4,652,473 |
|
|
|
|
|
|
|
|
|
|
RESTRICTED CASH FOR LETTER OF CREDIT |
|
| 195,132 |
|
|
| 190,915 |
|
|
|
|
|
|
|
|
|
|
INTANGIBLE ASSETS, NET |
|
| 365,692 |
|
|
| 327,134 |
|
|
|
|
|
|
|
|
|
|
TOTAL ASSETS |
| $ | 6,858,022 |
|
| $ | 7,713,377 |
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ DEFICIT |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CURRENT LIABILITIES |
|
|
|
|
|
|
|
|
| $ | 582,098 |
|
| $ | 1,073,938 |
| |
|
| 165,103 |
|
|
| 152,593 |
| |
|
| 747,201 |
|
|
| 1,226,531 |
| |
|
|
|
|
|
|
|
|
|
LONG TERM LIABILITIES |
|
|
|
|
|
|
|
|
|
| 404,003 |
|
|
| 532,970 |
| |
|
| 11,803,934 |
|
|
| 10,422,641 |
| |
|
| 5,812,856 |
|
|
| 5,000,752 |
| |
|
| 18,020,793 |
|
|
| 15,956,363 |
| |
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES |
|
| 18,767,994 |
|
|
| 17,182,894 |
|
|
|
|
|
|
|
|
|
|
STOCKHOLDERS’ DEFICIT |
|
|
|
|
|
|
|
|
Series A convertible preferred stock, 3,000,000 shares authorized, 337,430 and 316,010 shares issued and outstanding, liquidation value of $4,049,160 and $3,792,120 respectively |
|
| 3,732,701 |
|
|
| 3,475,686 |
|
Series B Preferred Stock, 1,500,000 shares authorized, 269,987 and 0 shares issued and outstanding, liquidation value of $4,319,792 |
|
| 3,651,533 |
|
|
| — |
|
Common stock, no par value, 25,000,000 shares authorized, 836,544 and 835,144 shares issued and outstanding, respectively |
|
| 8,565,225 |
|
|
| 8,551,225 |
|
Additional paid in capital |
|
| 5,612,343 |
|
|
| 5,192,295 |
|
Accumulated deficit |
|
| (33,471,774 | ) |
|
| (26,688,723 | ) |
|
| (11,909,972 | ) |
|
| (9,469,517 | ) | |
|
|
|
|
|
|
|
|
|
TOTAL LIABILITIES AND STOCKHOLDERS’ DEFICIT |
| $ | 6,858,022 |
|
| $ | 7,713,377 |
|
See notes to unaudited condensed consolidated financial statements.
F-29
XG SCIENCES, INC.
CONDENSED CONSOLIDATED STATEMENT OF OPERATIONS (Unaudited)
|
| For the Nine Months ended September 30, | ||||||
|
| 2015 |
| 2014 | ||||
REVENUES |
|
|
|
|
|
|
|
|
| $ | 156,608 |
|
| $ | 798,881 |
| |
|
| 299,879 |
|
|
| 551,627 |
| |
|
| 75,000 |
|
|
| 75,000 |
| |
|
| 531,487 |
|
|
| 1,425,508 |
| |
|
|
|
|
| ||||
COST OF GOODS SOLD |
|
|
|
|
|
|
|
|
|
| 77,516 |
|
|
| 209,858 |
| |
|
| 1,264,432 |
|
|
| 1,835,178 |
| |
|
| 1,341,948 |
|
|
| 2,045,036 |
| |
GROSS PROFIT (LOSS) |
|
| (810,461 | ) |
|
| (619,528 | ) |
|
|
|
|
|
|
|
|
|
OPERATING EXPENSES |
|
|
|
|
|
|
|
|
|
| 1,135,326 |
|
|
| 988,149 |
| |
|
| 3,133,375 |
|
|
| 3,420,125 |
| |
|
| — |
|
|
| 38,177 |
| |
|
| 4,268,701 |
|
|
| 4,446,451 |
| |
OPERATING LOSS |
|
| (5,079,162 | ) |
|
| (5,065,979 | ) |
|
|
|
|
|
|
|
|
|
OTHER INCOME (EXPENSE) |
|
|
|
|
|
|
|
|
|
| 80,624 |
|
|
| 54,584 |
| |
|
| (1,640,668 | ) |
|
| (1,064,760 | ) | |
|
| (143,845 | ) |
|
| 462,664 |
| |
|
| — |
|
|
| (3,513,770 | ) | |
|
| (1,703,889 | ) |
|
| (4,061,282 | ) | |
|
|
|
|
|
|
|
|
|
NET LOSS |
|
| (6,783,051 | ) |
|
| (9,127,261) |
|
|
|
|
|
|
|
|
|
|
|
| — |
|
|
| (316,366 | ) | |
|
|
|
|
|
|
|
|
|
NET LOSS ATTRIBUTABLE TO COMMON SHAREHOLDERS |
| $ | (6,783,051 | ) |
| $ | (9,443,626 | ) |
|
|
|
|
|
|
|
|
|
WEIGHTED AVERAGE NUMBER OF SHARES OUTSTANDING |
|
| 836,544 |
|
|
| 835,144 |
|
NET LOSS PER SHARE –Basic and diluted |
| $ | (8.11 | ) |
| $ | (11.31 | ) |
See notes to unaudited condensed consolidated financial statements.
F-30
XG SCIENCES, INC.
CONDENSED CONSOLIDATED STATEMENT OF STOCKHOLDERS’ (DEFICIT) (unaudited)
|
| Preferred stock (A) |
| Preferred stock (B) |
| Common stock |
| Additional paid-in |
| Accumulated |
|
| |||||||||||||||
|
| Shares |
| Amount |
| Shares |
| Amount |
| Shares |
| Amount |
| capital |
| deficit |
| Total | |||||||||
Balances, December 31, 2014 |
| 316,010 |
| $ | 3,475,686 |
| — |
| $ | — |
|
| 835,144 |
| $ | 8,551,225 |
| $ | 5,192,295 |
| $ | (26,688,723 | ) |
| $ | (9,469,517 | ) |
Stock issued for cash |
| — |
|
| — |
| 269,987 |
|
| 4,319,792 |
|
| 1,400 |
|
| 14,000 |
|
| — |
|
| — |
|
|
| 4,333,792 |
|
Proceeds allocated to warrants |
| — |
|
| — |
|
|
|
| (668,259 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
| (668,259 | ) |
Preferred stock issued to pay capital lease obligations |
| 21,420 |
|
| 257,015 |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
|
| 257,015 |
|
Employee stock option expense |
| — |
|
| — |
| — |
|
| — |
|
| — |
|
| — |
|
| 420,048 |
|
| — |
|
|
| 420,048 |
|
Net loss |
| — |
|
| — |
| — |
|
| — |
|
| — |
|
| — |
|
| — |
|
| (6,783,051 | ) |
|
| (6,783,051 | ) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |||||||||
Balances, September 30, 2015 |
| 337,430 |
| $ | 3,732,701 |
| 269,987 |
| $ | 3,651,533 |
|
| 836,544 |
| $ | 8,565,225 |
| $ | 5,612,343 |
| $ | (33,471,774 | ) |
| $ | (11,909,972 | ) |
See notes to unaudited condensed consolidated financial statements.
F-31
XG SCIENCES, INC.
CONDENSED CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE NINE MONTHS ENDED SEPTEMBER 30, 2015 AND 2014 (unaudited)
|
| 2015 |
| 2014 | ||||
CASH FLOWS FROM OPERATING ACTIVITIES |
|
|
|
| ||||
| $ | (6,783,051 | ) |
| $ | (9,127,261 | ) | |
|
|
|
|
|
|
|
| |
|
| 641,304 |
|
|
| 646,686 |
| |
|
| 21,105 |
|
|
| 18,523 |
| |
|
| — |
|
|
| 38,177 |
| |
|
| 420,048 |
|
|
| 436,277 |
| |
|
| 1,550,616 |
|
|
| 1,059,372 |
| |
|
| — |
|
|
| (120,000 | ) | |
|
| 143,845 |
|
|
| (462,664 | ) | |
|
| — |
|
|
| 3,513,770 |
| |
|
|
|
|
|
|
|
| |
|
| (42,275 | ) |
|
| 104,801 |
| |
|
| (78,593 | ) |
|
| (73,676 | ) | |
|
| 27,700 |
|
|
| (76,458) |
| |
|
| (491,842 | ) |
|
| (682,206 | ) | |
|
| — |
|
|
| (591,214 | ) | |
NET CASH USED IN OPERATING ACTIVITIES |
|
| (4,591,143 | ) |
|
| (5,315,873 | ) |
|
|
|
|
|
|
|
|
|
CASH FLOWS FROM INVESTING ACTIVITIES |
|
|
|
|
|
|
|
|
|
| (157,983 | ) |
|
| (682,845 | ) | |
|
| (59,663 | ) |
|
| (137,904 | ) | |
NET CASH USED IN INVESTING ACTIVITIES |
|
| (217,646 | ) |
|
| (820,749 | ) |
|
|
|
|
| ||||
CASH FLOWS FROM FINANCING ACTIVITIES |
|
|
|
|
|
|
|
|
|
| — |
|
|
| (103,447 | ) | |
|
| (28,764 | ) |
|
| (46,124 | ) | |
|
| — |
|
|
| 8,500,000 |
| |
|
| 4,319,792 |
|
|
| 1,212,000 |
| |
|
| 14,000 |
|
|
| 20,004 |
| |
NET CASH PROVIDED BY FINANCING ACTIVITIES |
|
| 4,305,028 |
|
|
| 9,582,433 |
|
|
|
|
|
|
|
|
|
|
NET INCREASE (DECREASE) IN CASH |
|
| (503,761 | ) |
|
| 3,445,811 |
|
CASH AT BEGINNING OF PERIOD |
|
| 2,088,866 |
|
|
| 95,342 |
|
|
|
|
|
|
|
|
|
|
CASH AT END OF PERIOD |
| $ | 1,585,105 |
|
| $ | 3,541,153 |
|
|
|
|
|
|
|
|
|
|
SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: |
|
|
|
|
|
|
|
|
| $ | 90,052 |
|
| $ | 5,388 |
| |
|
|
|
|
|
|
|
|
|
SUPPLEMENTAL DISCLOSURE OF NON-CASH INVESTING AND FINANCING: |
|
|
|
|
|
|
|
|
Value of preferred stock issued for AAOF capital lease obligations |
| $ | 257,015 |
|
| $ | 241,112 |
|
See notes to unaudited condensed consolidated financial statements.
F-32
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 1 — NATURE OF BUSINESS AND BASIS OF PRESENTATION
XG Sciences, Inc., a Michigan company located in Lansing, Michigan and its subsidiary, XGS IP, LLC (collectively referred to as “we”, “us”, “our”, or the “Company”) manufactures graphene nanoplatelets made from graphite, using a proprietary manufacturing process to split natural flakes of crystalline graphite into very small and thin particles, which we sell as xGnP® graphene nanoplatelets. These particles are then used in products like battery electrodes, thin sheets, films, inks and coatings that we sell to other companies. We also sell our nanoparticles in the form of bulk powders or dispersions to other companies for use as additives to make composite and other materials with specially engineered characteristics. Additionally, we license our technology to other companies in exchange for royalties and other fees.
Basis of Presentation
The accompanying interim condensed consolidated financial statements are unaudited and have been prepared in accordance with accounting principles generally accepted in the United States of America (“GAAP”) for interim financial information. All intercompany transactions have been eliminated in consolidation.
Certain information and footnote disclosures normally included in the Company’s annual audited consolidated financial statements and accompanying notes have been condensed or omitted in these interim condensed consolidated financial statements. Accordingly, the unaudited condensed consolidated financial statements included herein should be read in conjunction with the audited financial statements for the year ended December 31, 2014.
The results of operations presented in this quarterly report are not necessarily indicative of the results of operations that may be expected for any future periods. In the opinion of management, these unaudited condensed consolidated financial statements include all adjustments and accruals, consisting only of normal recurring adjustments that are necessary for a fair statement of the results of all interim periods reported herein.
NOTE 2—SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Going Concern
We have historically incurred recurring losses from operations and we may continue to generate negative cash flows as we implement our business plan. Our unaudited condensed consolidated financial statements are prepared using US GAAP as applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business.
We currently do not have sufficient cash or commitments for financing to sustain our operations for the next twelve months. We currently anticipate that our cash and cash equivalents will be sufficient to fund our operations through April 2016, without raising additional capital. Our plan is to develop customer relationships and increase our revenues derived from our products and IP licensing. Although we have historically incurred operating losses, we have been able to fund such losses primarily by selling common and preferred stock and convertible notes. We expect that our cash on hand September 30 and proceeds from anticipated financings (private placements of debt and equity securities) will sustain our operations for the next seven months. However, there can be no assurance of our continued success in raising capital to fund our business plan.
In the event we are unable to fund our operations from existing cash on hand, operating cash flows, additional borrowings or raising equity capital, we may be forced to reduce our expenses, slow down our growth rate, or discontinue operations. Our consolidated financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary should we be unable to continue as a going concern.
On April 18, 2015 we commenced an investment offering of up to $18,000,000 in units consisting of shares of Series B Convertible Preferred Stock and Warrants to purchase common stock. The offering price is $16 per unit.
On December 31, 2015 we issued promissory notes and warrants to purchase common stock of the Company to existing stockholders. The notes are non-convertible and mature on June 30, 2016. The warrants have a five year maturity date with a strike price of $8 per share.
F-33
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(Cont.)
The Company is authorized to issue up to 1,500,000 shares of Series B Convertible Preferred Stock. Each share of the Series B, which has a liquidation preference of $16 per share, is convertible at any time, at the option of the holder, into one share of Common Stock. The Series B may, at the option of the holder, be exchanged for any new securities issued by the Corporation until the earlier of such time as the Corporation completes the sale of at least $18 million in new financing or December 31, 2017. The Series B also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. The Series B is subject to mandatory conversion into Common Stock upon the listing of the Company’s Common Stock on a Qualified National Exchange, provided that all outstanding Convertible Securities have also converted into Common Stock.
In conjunction with this investment offering, all holders of the outstanding Secured Convertible Notes (see Note 6) agreed to modify the original terms of the Notes such that the Notes will automatically convert into shares of Series A Convertible Preferred Stock upon the earlier to occur of 1) the Company raising not less than $12,000,000 or 2) December 31, 2015.
As of September 30, 2015 we had accepted subscriptions for 269,987 units valued at $4,319,792 and had issued 269,987 shares of Series B convertible preferred stock and Warrants to purchase 224,897 shares of common stock, which includes 3,100 shares of Series B convertible preferred stock and 2,635 warrants issued pursuant to the preemptive rights described in Note 6.
Use of Estimates
We prepare our condensed consolidated financial statements in conformity with accounting principles generally accepted in the United States of America. These principles require management to make estimates, judgments and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses, together with amounts disclosed in the related notes to the financial statements. Actual results and outcomes may differ from management’s estimates, judgments and assumptions. Significant estimates, judgments and assumptions used in these financial statements include, but are not limited to, those related to revenues, accounts receivable and related allowances, contingencies, useful lives and recovery of long-term assets, income taxes, the fair value of stock-based compensation and derivative financial instrument liabilities. These estimates, judgments, and assumptions are reviewed periodically and the effects of material revisions in estimates are reflected in the financial statements prospectively from the date of the change in estimate.
Inventory
Inventory consists of raw materials, work-in-process and finished goods, all of which are valued at standard cost, which approximates average cost.
Recoverability and Impairment of Long-Lived Assets
We review our long-lived assets for recoverability if events or changes in circumstances indicate the assets may be impaired. This circumstance exists when the carrying amount of the asset exceeds the sum of the undiscounted cash flows expected to result from its use and eventual disposition. At September 30, 2015, we believe the carrying value of our long-lived assets is not impaired.
Common and Preferred Stock Valuation Methodologies
The valuation of our Common and Preferred stock was prepared in accordance with the guidelines in the AICPA Practice Guide, which prescribes several valuation approaches for determining the value of an enterprise, such as the cost, market and income approaches, and various methodologies for allocating the value of an enterprise to its capital structure.
We considered valuations implied by arm’s length transactions involving the sale of our securities to independent investors, taking into consideration the various rights and preferences of the equity securities transacted. We concluded that the terms of our Series B Convertible Preferred Stock sales were representative of fair value. Using the Series B Preferred Stock pricing, we utilized the back-solve method (a form of the market approach defined in the AICPA Practice Guide) to estimate the implied
F-34
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(Cont.)
enterprise value and the resulting common and preferred stock fair values at certain dates. As of September 30, 2015, the fair value of our Common Stock, Series A Preferred and Series B Preferred was $8.77, $12.64 and $15.42, respectively.
Stock-Based Compensation
We recognize compensation expense in our consolidated statements of operations for all share-based option and stock awards, based on estimated grant-date fair values.
We estimate the grant-date fair value of stock-based compensation awards using the Black-Scholes option valuation model. This model is affected by the estimated value of our common stock on the date of the grant as well as assumptions regarding a number of highly complex and subjective variables. These variables include the expected term of the option, the exercise price, expected risk-free rates of return, the expected volatility of our common stock, expected forfeiture rates and expected dividend yield. The assumptions for the estimated value of our common stock, expected term and expected volatility are the assumptions that most significantly affect the grant date fair value.
The grant-date fair value of the award is recognized as expense over the requisite service period using the straight-line method.
Derivative Financial Instruments
We do not use derivative instruments to hedge exposures to cash flow, market or foreign currency risk. The terms of convertible preferred stock and convertible notes that we issue are reviewed to determine whether or not they contain embedded derivative instruments that are required by ASC 815: “Derivatives and Hedging” to be accounted for separately from the host contract, and recorded at fair value. In addition, freestanding warrants are also reviewed to determine if they achieve equity classification. Certain stock warrants that we have issued did not meet the conditions for equity classification and are classified as derivative instrument liabilities measured at fair value. The fair values of these derivative liabilities are revalued at each reporting date, with the change in fair value recognized in earnings.
Fair Value Measurements
Financial Accounting Standards Board Accounting Standards Codification (“ASC”) 820: “Fair Value Measurements and Disclosures” defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. ASC 820 also establishes a fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure fair value:
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Inputs other than quoted prices included within Level 1 which are either directly or indirectly observable.
Level 3: Inputs to the valuation methodology are unobservable and significant to the fair value measurement.
A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement. Our derivative liabilities are classified as Level 3 within the fair value hierarchy because they were valued using unobservable inputs that were significant to the valuation. The valuation technique used to measure fair value of the derivative liabilities is based on a lattice model with significant assumptions and inputs determined by the Company. A lattice model was used to estimate the fair value of the derivative liabilities because management believes it reflects all the assumptions that market participants would likely consider including early exercise of the warrants. The fair value of the derivative liabilities will be significantly influenced by the fair value of our common stock, stock price volatility and the risk free interest components of the lattice technique.
F-35
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES(Cont.)
The following is a reconciliation of the beginning and ending balances for liabilities measured at fair value on a recurring basis using significant unobservable inputs (Level 3) during the nine months ended September 30, 2015 and 2014:
|
| 2015 |
| 2014 | |||
Balance at January 1 |
| $ | 5,000,752 |
| $ | 1,190,261 |
|
Warrants issued with additional financing |
|
| — |
|
| 2,520,527 |
|
Warrants issued with private placement of Series B Preferred Stock |
|
| 660,378 |
|
|
|
|
Warrants issued with preferred stock sold under preemptive rights |
|
| 7,881 |
|
| 316,366 |
|
Warrants issued for debt modification |
|
| — |
|
| 1,320,740 |
|
Gain (Loss) recognized in earnings |
|
| 143,845 |
|
| (462,664 | ) |
Balance at September 30 |
| $ | 5,812,856 |
| $ | 4,885,230 |
|
Beneficial Conversion Feature of Convertible Notes and Convertible Preferred Stock
If the embedded conversion option in a convertible note or convertible preferred stock is not required to be separated and accounted for as a derivative instrument liability, then ASC 470-20: “Debt with Conversion and Other Options” requires separate recognition of any beneficial conversion feature (“BCF”) related to the convertible instrument. Accordingly, we recognized a BCF related to the Secured Convertible Notes that we issued in 2014 and the Convertible Preferred Stock sold under preemptive rights during 2014. The BCF for the convertible instruments is recognized by allocating to additional paid-in capital a portion of the proceeds received equal to the intrinsic value of the conversion feature. Generally, the intrinsic value is calculated at the commitment date as the difference between the effective conversion price and the fair value of the common stock or other securities into which the security is convertible, multiplied by the number of shares into which the security is convertible. When certain other securities, such as warrants, are issued in conjunction with the convertible security, the proceeds are allocated among the different components. The portion of the proceeds allocated to the convertible security is divided by the contractual number of the conversion shares to determine the effective conversion price which is then used to measure the BCF. The value of the BCF recognized is limited to the amount of proceeds received that is initially allocated to the convertible security. See Note 6 for additional information related to the BCF resulting from our Secured Convertible Notes.
NOTE 3 — PROPERTY, PLANT AND EQUIPMENT
Property, plant and equipment as of September 30, 2015 and December 31, 2014 consist of the following:
|
| Depreciable life |
| September 30, 2015 |
| December 31, 2014 | ||||
Production and equipment not yet placed in service |
|
|
| $ | — |
|
| $ | 820,861 |
|
Leasehold improvements |
| 5 – 10 |
|
| 399,060 |
|
|
| 382,230 |
|
Lab equipment |
| 3 – 7 |
|
| 899,746 |
|
|
| 893,068 |
|
Production and other equipment |
| 3 – 7 |
|
| 6,025,681 |
|
|
| 5,070,344 |
|
Software |
| 3 |
|
| 39,144 |
|
|
| 39,144 |
|
|
|
|
|
|
|
| ||||
|
|
|
|
| 7,363,631 |
|
|
| 7,205,647 |
|
Less accumulated depreciation and amortization |
|
|
|
| (3,194,478 | ) |
|
| (2,553,174 | ) |
|
|
|
|
|
|
|
|
|
|
|
Net property, plant and equipment |
|
|
| $ | 4,169,153 |
|
| $ | 4,652,473 |
|
Depreciation and amortization expense on property and equipment, including leased assets, for the nine months ended September 30, 2015 and 2014 was $641,304 and $646,686, respectively.
F-36
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 4 — INTANGIBLE ASSETS
Intangible assets and related accumulated amortization at September 30, 2015 and December 31, 2014 was as follows:
|
| Carrying Amount |
| Less Accumulated Amortization |
| Net Carrying Amount | ||||
Licenses |
| $ | 137,533 |
| $ | (62,979 | ) |
| $ | 74,554 |
Patents |
|
| 328,244 |
|
| (41,011 | ) |
|
| 287,233 |
Trademarks, other tangibles |
|
| 5,698 |
|
| (1,793 | ) |
|
| 3,905 |
Balance, September 30, 2015 |
| $ | 471,475 |
| $ | (105,783 | ) |
| $ | 365,692 |
|
|
|
|
|
|
|
|
|
|
|
Licenses |
| $ | 137,308 |
| $ | (55,908 | ) |
| $ | 81,400 |
Patents |
|
| 268,805 |
|
| (27,264 | ) |
|
| 241,541 |
Trademarks, other intangibles |
|
| 5,698 |
|
| (1,505 | ) |
|
| 4,193 |
Balance, December 31, 2014 |
| $ | 411,811 |
| $ | (84,677 | ) |
| $ | 327,134 |
Amortization expense for the nine months ended September 30, 2015 and 2014 was $21,105 and $18,523, respectively.
NOTE 5 — ACCOUNTS PAYABLE AND OTHER CURRENT LIABILITIES
Accounts payable and other current liabilities at September 30, 2015 and December 31, 2014 are as follows:
|
| September 30, 2015 |
| December 31, 2014 | ||
Accounts payable |
| $ | 200,013 |
| $ | 324,096 |
Product marketing allowance |
|
| 18,390 |
|
| 35,045 |
Accrued compensation |
|
| 246,440 |
|
| 210,800 |
Accrued expenses |
|
| 40,315 |
|
| 195,390 |
401(k) employer contribution expense |
|
| 76,940 |
|
| 54,207 |
Current portion of deferred compensation |
|
| — |
|
| 254,400 |
|
|
|
|
| ||
Accounts payable and other liabilities |
| $ | 582,098 |
| $ | 1,073,938 |
A promissory note for $254,400 was executed on January 1, 2015 to satisfy the deferred compensation with a shareholder and officer of the Company. The note was due and payable on July 1, 2015 with accrued interest at a rate of 12%. On May 28, 2015 the note balance of $254,400 and accrued interest in the amount of $12,800 was paid in full.
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS
On March 18, 2013 we entered into a series of agreements with our former Chief Executive Officer, Michael R. Knox and Aspen Advance Opportunity Fund, LP (“AAOF”). Under these agreements, Mr. Knox and AAOF agreed to provide financing to the Company in the form of Secured Convertible Notes as well as optional lease financing. The agreements provided for Mr. Knox to lend the Company $700,000, to be funded by converting advances he had made under a revolving line of credit, and for AAOF to lend the Company a minimum of $2.5 million and, at the option of AAOF, a maximum of $5 million, with a provision that up to 50% of the AAOF funding could be funded under lease financing arrangements.
Subsequently, on July 12, 2013, these agreements were amended and restated to provide for expanded financing commitments from AAOF as well as a related entity, XGS II, LLC (“XGS II”). Under the terms of these amended agreements, the financing commitments from AAOF were increased to a minimum of $6 million and, at the option of AAOF, a maximum of $10 million, of which a maximum of $3 million could be provided in the form of lease financing. Also under these amended agreements, XGS II agreed to provide a minimum of $250,000 and, at the option of XGS II, a maximum of $750,000. All of the funding with the exception of the Knox financing, was scheduled according to minimum financing commitments on calendar dates ranging from the time the agreements were executed to a period of eighteen months into the future. With the exception of any
F-37
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(Cont.)
lease financing, the financing provided by Knox, AAOF, and XGS II takes the form of Secured Convertible Notes with identical terms and provisions.
On January 15, 2014, we amended our agreements with AAOF and XGS II to revise the terms and amounts related to the timing of future investments from AAOF and XGS II by increasing the XGS II optional funding level to $1 million and extending the optional financing periods for both AAOF and XGS II through the end of 2014. In connection with the amendments, we also i) transferred all of our intellectual property into a newly-created, wholly-owned subsidiary called XGS IP, LLC, ii) revised certain terms of the Company’s By-Laws to allow for special shareholder meetings to be called by holders of ten percent or more of the Company’s stock and to clarify certain voting provisions, and iii) amended the previously issued Certificate of Designation of Series A Convertible Preferred Stock to clarify the conditions related to Mandatory Conversion into Common Stock upon listing of the Company’s stock on a Qualified National Exchange and to add a provision that would exempt holders of Series A from future mandatory financing participation.
The Secured Convertible Notes issued under these agreements bear interest at 12% per annum and have a due date of March 18, 2018. These notes have provisions providing an option to the Company, at its choice, to accrue the interest payments on the notes through December 31, 2014 and add the accrued interest to the principal of the notes. After December 31, 2014, the note holder, at its option, can choose to receive ongoing interest payments: 1) in the form of cash; 2) receive all or a portion of currently due interest in the form of Series A Convertible Preferred Stock at a price per share (currently $12) equal to the then effective Series A Original Issue Price as defined in the Company’s Certificate of Designation of Series A Convertible Preferred Stock; 3) receive all or a portion of currently due interest payments in the form of any other series of Preferred Stock outstanding at the time interest is due; or 4) elect to accrue such interest and add it to the balance of the Note. Beginning January 1, 2015, Mr. Knox chose to receive ongoing interest payments in cash, while all other note holders continue to have interest accrued and added to the balance of their Notes. These Notes are secured by all of the assets, both tangible and intangible, of the Company. Further, these notes have provisions allowing the conversion of these notes into Series A Convertible Preferred Stock (or such other form of Preferred Stock as may be outstanding at that time), at the option of the holder at a conversion price of $12 per share. In addition, these Notes contain various affirmative and negative covenants that provide for various reporting, information, notice, maintenance of business, insurance, audits and reappraisal requirements. These covenants also prohibit the Company from issuing new secured debt that would have security or liquidation provisions senior to or equivalent to the Secured Convertible Notes, and these covenants also prohibit the issuance of dividends or stock redemptions, creation of subsidiaries, certain transactions with subsidiaries, or transfer of assets without the permission of the holders.
The embedded conversion feature in the Secured Convertible Notes was analyzed under ASC 815 to determine if it achieved equity classification or required bifurcation as a derivative instrument. In order to be a derivative, one of the criteria is that the embedded component must be net settleable. The Company’s common stock is not publicly traded, there is no mechanism outside the notes that would permit the holder to achieve net settlement and the underlying shares are not readily convertible to cash. Accordingly, the embedded conversion feature does not meet the definition of a derivative, and therefore, does not require bifurcation from the host instrument. Certain default put provisions were not considered to be clearly and closely related to the host instrument but management concluded that the value of these default put provisions wasde minimus at September 30, 2015. Management will reconsider the value of the default put provisions each reporting period to determine if the value becomes material to the financial statements.
In conjunction with the sale of the Secured Convertible Notes to AAOF and XGS II during 2013, Stock Warrants to purchase an aggregate of 447,916 shares of Series A Convertible Preferred Stock were issued that vest as the financing occurs at a rate of one Stock Warrant for every $24.00 funded. The Stock Warrants have an exercise price of $12.00 per share and are indexed to Series A Convertible Preferred Stock which contains a feature that requires adjustment of the exercise price of the Preferred Stock if the Company issues any Common Stock or common stock equivalents for a price less than the exercise price in effect immediately before the issuance. The Stock Warrants expire on the earlier of i) March 18, 2023 or ii) the acquisition of the Company by another entity.
F-38
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(Cont.)
Although the Stock Warrants did not fall within the scope of ASC 480, they required derivative liability accounting because the conversion price reset protection terms in the underlying Series A Convertible Preferred Stock was not consistent with the definition for financial instruments indexed only to a company’s own stock. As such, they are required to be marked to fair value each reporting period.
On January 15, 2014, we consolidated the Secured Convertible Notes issued in 2013 to AAOF and XGS II, together with accrued interest, into a single note for each lender with terms and conditions similar to those previously issued to AAOF and XGS II. We also exchanged existing warrants to purchase 447,916 shares of Series A Preferred Stock for new warrants to purchase 916,666 shares of Series A Preferred Stock. As of January 15, 2014, 434,314 Stock Warrants were vested and their fair value was estimated to be $2,525,822 using a lattice model with the following inputs: estimated value of underlying stock: $12.00; equivalent risk-free interest rate: 1.40%; expected term: 10 years; equivalent stock price volatility: 37.16%; expected dividend yield: none. Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. The fair value of the Stock Warrants immediately prior to the exchange was $1,205,081, resulting in an increase in fair value of $1,320,741.
The cash flows of the newly consolidated Secured Convertible Notes plus the fair value of the additional Stock Warrants issued exceeded the cash flows of the old Secured Convertible Notes by more than 10%. Therefore, in accordance with ASC 470 “Debt”, the modification of the notes was accounted for as a debt extinguishment. Because the conversion price of the Secured Convertible Notes is the same as the estimated value of the underlying stock on the date of the modification, no beneficial conversion feature was recognized. The difference of $2,193,029 between the carrying amounts of the original Secured Convertible Notes and the fair value of the newly consolidated Secured Convertible Notes plus the increase in fair value resulting from the issuance of additional Stock Warrants of $1,320,741 was recorded as a loss on extinguishment of $3,513,770.
During the year ended December 31, 2014, we issued an additional $5,500,000 in Secured Convertible Notes to AAOF and XGS II with terms similar to those issued previously. Stock Warrants indexed to 454,166 shares of Series A Preferred Stock vested as result of the 2014 financings. In accordance with the terms of the January 15, 2014 amendment, the previously-issued Stock Warrants associated with these financings vest as the financings occur at a rate of one Stock Warrant for every $12.00 funded. As of September 30, 2015, all 833,332 Stock Warrants issued in conjunction with the Secured Convertible Notes were vested. The initial fair value of the Stock Warrants that vested during the year ended December 31, 2014 were estimated using a binomial lattice model with the following assumptions:
Fair value of underlying stock |
| $12.00 |
|
Equivalent risk free interest rate |
| 1.23% – 1.41 | % |
Expected term (in years) |
| 9.64 – 9.99 |
|
Equivalent stock price volatility |
| 34.94% – 37.16 | % |
Expected dividend yield |
| — |
|
Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. Because the Company is not publically traded, there is currently no active market for the Company’s stock. Therefore, the expected volatility of the Company’s stock was developed using historical volatility of a peer group for a period equal to the expected term of the Stock Warrants.
After recording the Stock Warrants at fair value, a determination was made as to whether, in accordance with ASC 470, there was a BCF associated with the Secured Convertible Notes. A BCF is a non-detachable conversion feature that is in-the-money at the commitment date. An option is in-the-money if its effective conversion price is less than the current fair value of the share. For purposes of measuring a BCF, the effective conversion price is based on the proceeds allocated to the convertible debt instrument after considering allocations to detachable warrants.
F-39
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(Cont.)
The cash proceeds from the AAOF and XGS II financings during the year ended December 31, 2014 were allocated first to the derivative liabilities resulting from the Stock Warrants, at their fair values, then to the beneficial conversion feature, with the residual allocated to the host debt contracts, as follows:
|
| Allocation | |
|
|
| |
Derivative liabilities – warrants |
| $ | 2,520,527 |
Beneficial Conversion Feature |
|
| 2,520,527 |
Secured Convertible Notes – initial carrying value |
|
| 458,946 |
Total allocated proceeds |
| $ | 5,500,000 |
The amount allocated to the derivative liabilities and the BCF was recorded as a discount on the Secured Convertible Notes and is being amortized to interest expense over the remaining term of the notes using the effective interest method. The effective interest rates on the Secured Convertible Notes are approximately 28% - 85%. Total interest expense of $1,459,297 and $951,601 was recognized for the nine months ended September 30, 2015 and 2014, respectively.
At September 30, 2015, the total funding from the AAOF and XGS II agreements was $11,026,091. Because the lease commitments of $1,026,091 exceed the $1,000,000 initial commitment by $26,091, AAOF increased the maximum potential funding from $11,000,000 to $11,026,091 which resulted in the issuance of an additional 2,174 Stock Warrants to AAOF.
Samsung Note
On January 15, 2014, we sold $3,000,000 of Secured Convertible Notes to SVIC No. 15 New Technology Business Investment L.L.P, a subsidiary of the Samsung Group (“Samsung”). These notes had terms and conditions that were substantially similar to those previously issued to Knox, AAOF, and XGS II. In connection with the sale of the notes, the Company issued to Samsung a total of 100,000 Stock Warrants with a term of 4 years that are exercisable into shares of Series A Convertible Preferred Stock at a price of $12.00. These Stock Warrants vest in four installments on the first, second, third and fourth anniversary of the issuance of the Stock Warrants according to the following formula: On each anniversary, the warrant vests according to the ratio of total Samsung payments to us during the previous twelve-month period divided by 250. Thus, the warrants will vest proportionately in relation to the first $25 million, or fraction thereof, that Samsung pays, in cash, to us over the four years beginning January 15, 2014. Samsung payments are defined as future total cash payments, including licensing, royalties and product purchases but not joint development or purchase of Secured Convertible Notes. As of September 30, 2015, no payments had been made by Samsung and none of the Stock Warrants associated with the Samsung note had vested. If and when Samsung provides funding to the Company in the future, the Stock Warrants will be measured at fair value and expensed at that time.
Summary
The following table summarizes the Secured Convertible Notes outstanding and the number of shares of Series A Convertible Preferred Stock in which the Notes may be converted at September 30, 2015:
|
| AAOF |
| XGS II |
| KNOX |
| SAMSUNG |
| Total | ||||||||
Face value of notes at issuance |
| $ | 9,000,000 |
|
| $ | 1,000,000 |
|
| $ | 700,000 |
| $ | 3,000,000 |
| $ | 13,700,000 |
|
Accrued interest on face value |
|
| 2,181,423 |
|
|
| 271,539 |
|
|
| 166,721 |
|
| 665,451 |
|
| 3,285,134 |
|
Amount due – September 30, 2015 |
|
| 11,181,423 |
|
|
| 1,271,539 |
|
|
| 866,721 |
|
| 3,665,451 |
|
| 16,985,134 |
|
Unamortized (discount) |
|
| (4,891,261 | ) |
|
| (289,940 | ) |
|
| — |
|
| — |
|
| (5,181,201 | ) |
Carrying value |
| $ | 6,290,162 |
|
| $ | 981,599 |
|
| $ | 866,721 |
| $ | 3,665,451 |
| $ | 11,803,934 |
|
Indexed shares |
|
| 931,785 |
|
|
| 105,962 |
|
|
| 72,227 |
|
| 305,454 |
|
| 1,415,428 |
|
Private Placement
In April 2015, we commenced a private placement offering of up to $18,000,000 in units consisting of up to 1,125,000 shares of Series B Convertible Preferred Stock and Warrants to purchase Common Stock at an offering price of $16.00 per Unit. As of September 30, 2015, we had sold 266,887 shares of Series B Convertible Preferred Stock and Warrants to purchase
F-40
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(Cont.)
222,262 shares of Common Stock, for aggregate gross proceeds of $4,270,192. In addition, existing shareholders and holders of convertible notes exercised their preemptive rights and purchased 3,100 shares of Series B Convertible Preferred Stock and Warrants to purchase 2,635 shares of Common Stock, for which we received aggregate gross proceeds of $49,600 (see below). The Series B Preferred Stock has a stated value of $16.00 per share and is convertible, at the option of the holder, at a conversion price of $16.00 per share, subject to adjustments for stock dividends, splits, combinations and similar events. The Warrants have an exercise price of $16.00 per share and expire in 7 years. During the period from closing of the offering and ending on the earlier of i) December 31, 2017 and ii) the date the Company consummates the sale of new securities resulting in gross proceeds of at least $18 million, the holder has the right to exchange their Series B Preferred Stock and Warrants on a price per share basis into the new security on the relative price per share terms as the new securities were sold to the third party.
The cash proceeds from the private placement were allocated first to the derivative liabilities resulting from the Stock Warrants, at their fair values. After allocating a portion of the proceeds to the warrants it was determined there was no BCF present because the effective conversion price of the Series B Preferred Stock was greater than the current fair value of the shares. Therefore, the residual was allocated to the Series B Preferred Stock, as follows:
|
| Allocation | |
Derivative liabilities – warrants |
| $ | 660,378 |
Series B Preferred Stock |
|
| 3,609,814 |
Total allocated proceeds |
| $ | 4,270,192 |
As mentioned in further detail below, the private placement triggered a preemptive rights offering which resulted in additional proceeds of $49,600. Proceeds of $7,881 were allocated to derivative liabilities-warrants with the remaining $41,719 being allocated to the Series B Preferred Stock.
The Stock Warrants issued in the private placement required derivative liability accounting and are required to be marked to fair value each reporting period. The initial fair value of the Stock Warrants was estimated using a binomial lattice model with the following assumptions:
Fair value of underlying stock |
| $12.00 |
|
Equivalent risk free interest rate |
| 0.91% – 1.03% | |
Expected term (in years) |
| 7.00 |
|
Equivalent stock price volatility |
| 30.47% – 31.22% | |
Expected dividend yield |
| — |
|
Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. The expected volatility of the Company’s stock was developed using historical volatility of a peer group for a period equal to the expected term of the Stock Warrants.
Preemptive Rights
On January 15, 2014, as part of our financing agreements with Samsung, AAOF and XGS II, we agreed to allow all shareholders to purchase one share of Series A Convertible Preferred Stock at a price of $12 per share for every two shares of Series A Convertible Preferred Stock or Common Stock owned by the shareholder. In addition, for every two preemptive shares purchased, the Company issued the shareholder a warrant to purchase one additional share of Series A Convertible Preferred Stock with the same terms as the warrants issued to AAOF and XGS II. The Company also agreed to issue warrants with the same terms to those shareholders who exercised preemptive rights in October 2013. These preemptive rights expired on March 31, 2014.
During the quarter ended March 31, 2014, 101,000 shares of Series A Convertible Preferred Stock were sold to existing stockholders at a price of $12.00 per share under the January 15, 2014 preemptive rights offering. In addition, Stock Warrants indexed to 56,054 shares of Series A Convertible Preferred Stock were issued in conjunction with these stock purchases, including 5,554 warrants related to the preemptive rights exercised in October 2013.
F-41
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(Cont.)
As part of our private placement in April 2015, shareholders and holders of convertible notes were provided the right to purchase their pro rata share of any class of stock that the Company sells or issues. The sale of Series B Preferred Stock in the April 2015 offering triggered the preemptive rights. As of September 30, 2015, 3,100 shares of Series B Convertible Preferred Stock have been sold to existing stockholders at a price of $16.00 per share. In addition, Stock Warrants indexed to 2,635 shares of Series B convertible preferred stock were issued in conjunction with these stock purchases.
The proceeds from the exercise of the preemptive rights were allocated first to the derivative liabilities resulting from the Stock Warrants, at their fair values, then to the beneficial conversion feature (if applicable), with the residual allocated to the Preferred Stock, as follows:
|
| January 2014 Preemptive Rights |
| April 2015 Preemptive Rights | ||
Derivative liabilities – warrants |
| $ | 316,366 |
| $ | 7,881 |
Beneficial Conversion Feature |
|
| 316,366 |
|
| — |
Series A Preferred Stock |
|
| 712,552 |
|
| — |
Series B Preferred Stock |
|
| — |
|
| 41,719 |
Total allocated proceeds |
| $ | 1,345,284 |
| $ | 49,600 |
ASC 470 provides for a BCF to be accreted over the longer of the term to maturity or the period to when the holder can first convert the instrument. Because the Series A Convertible Preferred Stock had no stated maturity and are convertible on the issuance date, the BCF is accreted immediately by crediting the Series A Convertible Preferred Stock, with an offsetting charge to retained earnings or, in the absence of retained earnings, to paid-in capital.
Warrants
As of September 30, 2015, all 1,197,617 Stock Warrants issued to AAOF, XGS II and holders of Series A and Series B Preferred Stock have vested.
The Stock Warrants issued in conjunction with the convertible notes, the equipment financing leases, the 2015 private placement transaction and the preemptive rights are derivative liabilities which require re-measurement at fair value each reporting period. Liability classification is required because the conversion price reset protection terms in the underlying Series A Convertible Preferred Stock and the Series B Designations is not consistent with the definition for financial instruments indexed only to a company’s own stock.
The initial value of the Stock Warrants issued as consideration for the equipment financing leases was recorded as a reduction of the capital lease obligation and is being amortized as part of the effective interest cost on the capital lease obligation (see Note 10).The Company estimated the fair value of these derivative liabilities as of September 30, 2015 and December 31, 2014, using a lattice model and the following assumptions:
|
| September 30, |
| December 31, | ||
Fair value of underlying stock |
| $8.77 – $12.64 |
|
| $12.00 |
|
Equivalent risk free interest rate |
| 0.86% – 1.04% |
| 1.25% | ||
Expected term (in years) |
| 6.59 – 8.30 |
|
| 9.05 |
|
Equivalent stock price volatility |
| 35.00% – 35.61% |
| 34.27% | ||
Expected dividend yield |
| — |
|
| — |
|
Equivalent amounts reflect the net results of multiple modeling simulations that the lattice model applies to underlying assumptions. Because the Company is not publically traded, there is no active market for the Company’s stock. Therefore, the
F-42
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 6 — SECURED CONVERTIBLE NOTES, WARRANTS AND FINANCING AGREEMENTS(Cont.)
expected volatility of the Company’s stock was developed using historical volatility for a comparable company peer group for a period equal to the expected term of the Stock Warrants.
The following table summarizes the fair value of the derivative liabilities as of September 30, 2015 and December 31, 2014:
|
| 2015 |
| 2014 | ||
Warrants issued with Secured Convertible Notes |
| $ | 4,664,160 |
| $ | 4,284,161 |
Warrants issued with equipment financing leases |
|
| 466,418 |
|
| 428,417 |
Warrants issued with preemptive rights |
|
| 318,048 |
|
| 288,174 |
Warrants issued with 2015 private placement |
|
| 364,230 |
|
| — |
Total derivative liabilities |
| $ | 5,812,856 |
| $ | 5,000,752 |
Shares indexed to derivative liabilities as of September 30, 2015 and December 31, 2014 were as follows:
|
| 2015 |
| 2014 |
Warrants issued with Secured Convertible Notes |
| 833,332 |
| 833,332 |
Warrants issued with equipment financing leases |
| 83,334 |
| 83,334 |
Warrants issued with preemptive rights |
| 58,689 |
| 56,054 |
Warrants issued with 2015 private placement |
| 222,262 |
| — |
Total shares indexed to derivative liabilities |
| 1,197,617 |
| 972,720 |
All derivative Stock Warrants have an exercise price of $12.00, except for the Stock Warrants issued in conjunction with the 2015 private placement and related preemptive rights which have an exercise price of $16.00.
Changes in the fair value of Derivative Liabilities, carried at fair value, are reported as “Change in fair value of derivative liability — warrants” in the Statement of Operations, as follows:
|
| Nine months ended | |||||
|
| 2015 |
| 2014 | |||
Warrants issued with Secured Convertible Notes |
| $ | (379,999 | ) |
| $ | 403,746 |
Warrants issued with equipment financing leases |
|
| (38,001 | ) |
|
| 33,137 |
Warrants issued with preemptive rights |
|
| (21,993 | ) |
|
| 25,781 |
Warrants issued with 2015 private placement |
|
| 296,148 |
|
|
| — |
Total Derivative Gain (Loss) |
| $ | (143,845 | ) |
| $ | 462,664 |
NOTE 7 — CONVERTIBLE PREFERRED STOCK
Series A Convertible Preferred Stock
The Company is authorized to issue up to 3,000,000 shares of Series A Convertible Preferred Stock. Each share of the Series A, which has a liquidation preference of $12 per share, is convertible at any time, at the option of the holder, into one share of Common Stock at the lower of: (a) $12.00 per share, or (b) 80% of the price at which the Company sells any equity or equity-linked securities in the future. The Series A also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. The Series A is subject to mandatory conversion into Common Stock upon the listing of the Company’s Common Stock on a Qualified National Exchange.
Series B Convertible Preferred Stock
The Company is authorized to issue up to 1,500,000 shares of Series B Convertible Preferred Stock. Each share of the Series B, which has a liquidation preference of $16 per share, is convertible at any time, at the option of the holder, into one share of
F-43
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 7 — CONVERTIBLE PREFERRED STOCK(Cont.)
Common Stock at $16 per share. The Series B also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. The Series B is subject to mandatory conversion into Common Stock upon the listing of the Company’s Common Stock on a Qualified National Exchange and after all outstanding Convertible Securities are also converted into Common Stock. As mentioned in Note 6, the Series B provides investors with the right to exchange their Series B and Warrants on a price per share basis into new securities on the relative price per share terms as new securities sold to a third party on the earlier of i) December 31, 2017 and ii) the date the Company consummates the sale of new securities resulting in gross proceeds of at least $18 million.
The Series A and B are not redeemable for cash and the Company concluded that they are more akin to equity-type instruments than debt-type instruments. Accordingly, the embedded conversion option in each agreement is clearly and closely related to an equity-type host and the conversion option does not require classification and measurement as a derivative financial instrument. Therefore, the securities are classified in stockholders’ equity.
NOTE 8 — INCENTIVE STOCK OPTION PLAN
We have established an incentive stock option plan (the “Plan”) under which the Company may grant key employees and directors options to purchase common stock of the Company at not less than fair market value as of the grant date. Options for up to 600,000 shares may be awarded under the Plan. Each option is exercisable into one share of common stock of the Company. The Plan expires in December 2017. The fair value of the options granted was estimated on the dates of grant using the Black Scholes option-pricing model. As of 464,750 option shares have been granted. Vesting of the options range from immediately to 20% per year. Rights to exercise the options vest immediately upon a change in control of the Company or termination of the employee’s continuous service due to death or disability. The options expire at various dates through January 2022.
The following table shows the stock option activity during the nine months ended September 30, 2015.
|
| Number of options |
| Weighted average exercise price | |
Options outstanding – beginning of period |
| 464,750 |
| $ | 12.04 |
Granted at market price |
| — |
|
|
|
Exercised |
| — |
|
|
|
Expired |
| — |
|
|
|
Options outstanding – end of period |
| 464,750 |
| $ | 12.04 |
|
|
|
|
| |
Options exercisable – end of period |
| 161,082 |
| $ | 12.04 |
NOTE 9 — STOCK WARRANTS
In addition to the warrants described in Note 6, which are accounted for as derivative liabilities, we had 42,694 Common Stock Warrants that were issued in 2012 and prior years and which are accounted for as equity instruments. In July 2014 and April 2015, 22,694 and 3,600 of these warrants, respectively, expired unexercised and 1,400 of the warrants were exercised in May 2015. The remaining 11,000 warrants have exercise prices ranging from $8.00 to $12.00 and expire at various dates through 2027.
The following table summarizes the warrants outstanding at September 30, 2015, all of which are exercisable:
Date Issued |
| Expiration Date |
| Exercise Price |
| Number of Warrants |
7/1/2009 |
| 7/1/2019 |
| $8.00 |
| 6,000 |
10/8/2012 |
| 10/8/2027 |
| $12.00 |
| 5,000 |
|
|
|
|
|
| 11,000 |
F-44
XG SCIENCES, INC.
NOTES TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
NOTE 10 — CAPITAL LEASES
As of September 30, 2015 and December 31, 2014, we have capital lease obligations as follows:
|
| September 30, 2015 |
| December 31, 2014 | ||||
Capital lease obligations |
| $ | 742,716 |
|
| $ | 935,174 |
|
Unamortized warrant discount |
|
| (173,610 | ) |
|
| (249,611 | ) |
Net obligations |
|
| 569,106 |
|
|
| 685,563 |
|
Short-term portion of obligations |
|
| (165,103 | ) |
|
| (152,593 | ) |
|
|
|
|
| ||||
Long-term portion of obligations |
| $ | 404,003 |
|
| $ | 532,970 |
|
The 83,334 Stock Warrants issued as consideration for the equipment financing leases are recorded as derivative liabilities at fair value. The initial value of these warrants was recorded as a reduction of the capital lease obligation and is being amortized as part of the effective interest cost on the capital lease obligations.
NOTE 11 — OTHER RELATED PARTY TRANSACTIONS
We have a licensing agreement for exclusive use of patents and pending patents with Michigan State University (MSU), a stockholder via the MSU Foundation. During nine months ended September 30, 2015 and 2014, we incurred expenses of $37,500 and $18,500, respectively. We have also entered into product licensing agreements with certain other stockholders. No royalty expenses have been recognized related to these agreements during the nine months ended September 30, 2015 and 2014.
In 2015 and 2014, POSCO, one of our shareholders, has a contractual obligation to pay us a minimum of $100,000 per year to license certain technologies we license from MSU. This obligation is due annually on February 28 of the following year. We recorded this license revenue at a rate of $25,000 per quarter for the nine months ended September 30, for 2015 and 2014, respectively. POSCO is disputing that they are obligated to pay the royalties. This case is being arbitrated. An allowance in the amount of $87,500 has been recorded at September 30, 2015 to reflect an estimate of what portion of the 2014 and 2015 royalties may not be collectible. The accrued royalty and allowance are netted together and reflected in other current assets on the balance sheet.
The financing arrangements disclosed in Note 6 were provided by AAOF and XGS II, two private funds that were formed for the sole purpose of investing in the Company by two investors affiliated with ASC-XGS, LLC, a shareholder of the Company. Under a Voting Agreement signed by our shareholders, a principal of the private funds serves as a director of the Company.
NOTE 12 — RETIREMENT PLAN
We maintain a defined-contribution 401(k) retirement plan covering substantially all employees (as defined). Employees may make voluntary contributions to the plan, subject to limitations based on IRS regulations and compensation. The plan allows for an employer match and/or profit sharing contribution. For the nine months ended September 30, 2015 and 2014 the 401(k) employer expense was $70,056 and $61,074, respectively.
NOTE 13 — LETTER OF CREDIT
We are required by one of our lease agreements to maintain a letter of credit of approximately $190,000 through February 2022. To support this letter of credit, we are required to maintain an equivalent cash deposit. As of September 30, 2015 and December 31, 2014, there were no amounts outstanding on the letter of credit. The cash deposit is restricted and classified as a non-current asset.
NOTE 14 — SUBSEQUENT EVENTS
Subsequent events have been evaluated through the date these financial statements were submitted within the Registration Statement on Form S-1 to the Securities and Exchange Commission.
F-45
YOU SHOULD RELY ONLY ON THE INFORMATION CONTAINED IN THIS DOCUMENT OR THAT WE HAVE REFERRED YOU TO. WE HAVE NOT AUTHORIZED ANYONE TO PROVIDE YOU WITH INFORMATION THAT IS DIFFERENT. THIS PROSPECTUS IS NOT AN OFFER TO SELL COMMON STOCK AND IS NOT SOLICITING AN OFFER TO BUY COMMON STOCK IN ANY STATE WHERE THE OFFER OR SALE IS NOT PERMITTED.
6,784,442 SHARES OF COMMON STOCK
_______________
PROSPECTUS
_______________
Until _____________, all dealers that effect transactions in these securities, whether or not participating in this offering, may be required to deliver a prospectus. This is in addition to a dealer’s obligation to deliver a prospectus when acting as underwriter and with respect to their unsold allotments or subscriptions.
The Date of This Prospectus is , 2016
PART II: INFORMATION NOT REQUIRED IN THE PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
|
| Amount to be Paid | |
Registration fee |
|
| 5,465.55 |
Printing and engraving expenses |
|
| — |
Legal fees and expenses |
|
| 140,000 |
Accounting fees and expenses |
|
| 20,000 |
|
|
|
|
Total |
| $ | 165,465.55 |
Item 14. Indemnification of Directors and Officers.
Our Articles of Incorporation, as amended states that a Director of the Company shall not be personally liable to the Company or its stockholders for money damages for any action taken or any failure to take any action as a Director, except liability for any of the following:
(a) The amount of a financial benefit received by a Director to which the Director is not entitled;
(b) The intentional infliction of harm on the Company or its stockholders;
(c) A violation of Section 551 of the Michigan Business Corporation Act, as amended (the “Act”); or
(d) An intentional criminal act.
In the event the Act is amended, after approval by the stockholders of the appurtenant article in the Company’s Articles of Incorporation, as amended to authorize corporate action further eliminating or limiting the personal liability of Directors, then the liability of a Director of the Company shall be eliminated or limited to the fullest extent permitted by the Act, as so amended. Any repeal, modification or adoption of any provision in our Articles of Incorporation, as amended inconsistent with this Article shall not adversely affect any right or protection of a Director of the Company existing at the time of such repeal, modification or adoption.
Our Bylaws state that each person who is or was or had agreed to become a Director or officer of the Company, or each such person who is or was serving or who had agreed to serve at the request of the Board of Directors as an employee or agent of the Company, or as a Director, officer, employee or agent of another corporation (whether for profit or not), partnership, joint venture, trust or other enterprise (including the heirs, executors, administrators or estate of such person), shall be indemnified by the Company to the full extent permitted by the Act or any other applicable laws as presently or hereafter in effect.
The right to indemnification conferred in the Bylaws shall not be exclusive of any right any person may have or acquire under any statute, provision of our Articles of Incorporation, as amended, Bylaws, agreement, vote of stockholders or disinterested Directors, or otherwise.
The Company shall have the power to purchase and maintain insurance on behalf of any person who is or was a Director, officer, employee or agent of the Company or is or was serving at the request of the Company as a Director, officer, partner, trustee, employees or agent of another foreign or domestic corporation, partnership, joint venture, trust or other enterprise, whether for profit or not, against any liability asserted against him or her and incurred by him or her in any such capacity or arising out of his or her status as such, whether or not the Company would have the power to indemnify him or her against such liability under the appurtenant provision of the Company’s Bylaws.
II-1
Under Michigan law, however, such provisions do not eliminate the personal liability of a director unless the director (i) acted in good faith; (ii) acted in a manner he or she reasonably believed to be in or not opposed to the best interests of the corporation or its shareholders; and (iii) with respect to a criminal action or proceeding, if the director had no reasonable cause to believe his or her conduct was unlawful. Furthermore, under Section 551of the Act, directors who vote for, or concur in, any of the following corporate actions are jointly and severally liable to the corporation for the benefit of its creditors or shareholders, to the extent any legally recoverable injury suffered by its creditors or shareholders as a result of the action but not to exceed the difference between the amount paid or distributed and the amount that lawfully could have been paid or distributed: (i) a declaration of a share dividend or distribution to shareholders contrary to the Act or contrary to any restriction in the articles of incorporation; (ii) distribution to shareholders during or after dissolution of the corporation without paying or providing for debts obligations and liabilities of the corporation as required by Section 855a of the Act; and (iii) making a loan to a director, officer, or employee of the corporation or of a subsidiary of the corporation contrary to the Act.
We have been advised that in the opinion of the Securities and Exchange Commission indemnification for liabilities arising under the Securities Act is against public policy as expressed in the Securities Act, and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities is asserted by one of our directors, officers, or controlling persons in connection with the securities being registered, we will, unless in the opinion of our legal counsel the matter has been settled by controlling precedent, submit the question of whether such indemnification is against public policy to a court of appropriate jurisdiction. We will then be governed by the court’s decision.
Item 15. Recent Sales of Unregistered Securities.
During the three years preceding the filing of this registration statement, we issued and sold the following securities that were not registered under the Securities Act:
On October 8, 2012, we issued warrants to Michael R. Knox to purchase 5,000 shares of common stock at $20.00 per share, which was subsequently adjusted in August 2013 to $12.00 per share.
In March 2013, we issued a secured convertible Note to AAOF in the principal amount of $603,846.58.
On April 3, 2013, we issued a secured convertible Note to AAOF in the principal amount of $250,000.
On April 12, 2013, we issued a secured convertible Note to AAOF in the principal amount of $250,000.
On April 26, 2013, we issued a secured convertible Note to AAOF in the principal amount of $1,400,000.
In July 2013, we issued a secured convertible Note to AAOF in the principal amount of $800,000 (as adjusted to $309,425 pursuant to the July 12, 2013 amended and restated purchase agreement with AAOF).
In September 2013, we issued a secured convertible Note to AAOF in the principal amount of $200,000.
In October 2013, various individual stockholders exercised preemptive rights to purchase 11,107 shares of Series A Preferred Stock at a price of $12.00 per share. These purchases, along with an investment in convertible secured debt, enabled the conversion of 172,824 previously issued shares of common stock into 172,824 shares of Series A Preferred Stock.
In October 2013, we issued a secured convertible Note to AAOF in the principal amount of $300,000.
In October 2013, we issued a secured convertible Note to XGS II, LLC in the principal amount of $200,000.
In December 2013, we issued a secured convertible Note to AAOF in the principal amount of $500,000.
On January 7, 2014, we issued a secured convertible Note to AAOF in the principal amount of $100,000.
On January 15, 2014, we issued a secured convertible Note to AAOF in the principal amount of $4,178,330.92 which restated and consolidated all previously issued Notes to AAOF.
On January 15, 2014, we issued a secured convertible Note to XGS II, LLC in the principal amount of $739,349.83 which restated the $200,000 secured convertible Note previously issued in October 2013, and adjusted to reflect an increase in the principal pursuant to a reallocation of principal and accrued interest.
On January 15, 2014, we issued a secured convertible Note to Samsung Ventures in the principal amount of $3,000,000.
II-2
On January 15, 2014, we issued warrants to AAOF, XGS II, LLC and Samsung Ventures to purchase 833,333, 83,333 and 100,000 shares of Series A Preferred Stock, respectively at an exercise price of $12.00 per share.
In February 2014, we issued a secured convertible Note to AAOF in the principal amount of $1,000,000.
In March 2014, various individual shareholders exercised preemptive rights to purchase 101,000 shares of Series A Preferred Stock at a price of $12.00 per share.
On March 18, 2014, we issued a secured convertible Note to AAOF in the principal amount of $1,300,000.
On March 31, 2014, we issued a secured convertible Note to AAOF in the principal amount of $900,000.
In early 2014, we issued 12,722 shares of Series A Preferred Stock to AAOF as payment for lease financing obligations under the terms of a Master Leasing Agreement.
In June 2014 an individual stockholder exercised a stock option to purchase 1,667 shares of Common Stock at $12 per share.
In March 2015 an individual stockholder exercised a warrant to purchase 1,400 shares of Common Stock at a price of $10 per share.
On April 18, 2015 we commenced an offering consisting of units of Series B Preferred Stock and Warrants priced at $16 per unit. During the period April 18, 2015 through June 30, 2015 we sold 269,987 shares of Series B Preferred Stock and issued warrants to purchase an additional 224,897 shares of common stock for a total of $4,319,792.
On December 31, 2015 we issued promissory notes in an aggregate amount of $550,000 and warrants to purchase 20,625 shares of common stock of the Company to existing stockholders. The notes are non-convertible and mature on June 30, 2016. The warrants have a five year maturity date with a strike price of $8 per share.
No underwriters were utilized and no commissions or fees were paid with respect to any of the above transactions. These persons were the only offerees in connection with these transactions. We relied on Regulation S, Section 4(a)(2) and/or Rule 506 of Regulation D of the Securities Act for the transactions set forth above since none of the transactions involved any public offering.
Item 16. Exhibits and Financial Statement Schedules
EXHIBIT NUMBER |
| DESCRIPTION |
| LOCATION |
3.1 |
| Articles of Incorporation of XG Sciences, Inc. (formerly known as XG Nano, Inc.) dated May 23, 2006 |
| Filed herewith |
|
|
|
|
|
3.2 |
| Certificate of Amendment to Articles of Incorporation of XG Sciences, Inc. dated August 31, 2006 (name change) |
| Filed herewith |
|
|
|
|
|
3.3 |
| Certificate of Amendment to Articles of Incorporation of XG Sciences, Inc. dated March 23, 2009 (increase of authorized common stock) |
| Filed herewith |
|
|
|
|
|
3.4 |
| Certificate of Designations of Series A Convertible Preferred Stock, dated March 18, 2013 |
| Filed herewith |
|
|
|
|
|
3.5 |
| Certificate of Amendment to Articles of Incorporation of XG Sciences, Inc. dated June 26, 2013 (increase of authorized common stock and authorization of preferred stock) |
| Filed herewith |
|
|
|
|
|
3.6 |
| Amended and Restated Certificate of Designations of Series A Convertible Preferred Stock, dated June 26, 2013 |
| Filed herewith |
|
|
|
|
|
3.7 |
| First Amendment to the Amended and Restated Certificate of Designations of Series A Convertible Preferred Stock, dated November 20, 2013 |
| Filed herewith |
|
|
|
|
|
3.8 |
| Restated Bylaws dated January 15, 2014 |
| Filed herewith |
|
|
|
|
|
3.9 |
| Certificate of Designations of Series B Convertible Preferred Stock, effective September 9, 2015 |
| Filed herewith |
|
|
|
|
|
II-3
EXHIBIT NUMBER |
| DESCRIPTION |
| LOCATION |
4.1 |
| Warrant to Purchase 5,000 Shares of Common Stock, dated October 8, 2012, issued by XG Sciences, Inc. to Michael R. Knox, together with Notice and Certificate of Adjustment to Warrant, dated August 21, 2013 |
| Filed herewith |
|
|
|
|
|
4.2 |
| Warrant to Purchase 833,333 Shares of Series A Convertible Preferred Stock, dated January 15, 2014, issued by XG Sciences, Inc. to Aspen Advanced Opportunity Fund, LP |
| Filed herewith |
|
|
|
|
|
4.3 |
| Warrant to Purchase 83,333 Shares of Series A Convertible Preferred Stock, dated January 15, 2014, issued by XG Sciences, Inc. to XGS II, LLC |
| Filed herewith |
|
|
|
|
|
4.4 |
| Warrant to Purchase 100,000 Shares of Series A Convertible Preferred Stock, dated January 15, 2014, issued by XG Sciences, Inc. to SVIC No. 15 New Technology Business Investment L.L.P. |
| Filed herewith |
|
|
|
|
|
5.1 |
| Opinion of Foster Swift Collins & Smith PC |
| Filed herewith |
|
|
|
|
|
10.1 |
| Technology Licensing Agreement between XGS and Michigan State University dated July 27, 2007 and amended on May 24, 2010 and May 27, 2011 |
| Filed herewith |
|
|
|
|
|
10.2 |
| Lease between XG Sciences, Inc. and Quality Dairy Company, dated January 3, 2008 |
| Filed herewith |
|
|
|
|
|
10.3 |
| Amendment to Technology Licensing Agreement between XGS and Michigan State University dated May 24, 2010 |
| Filed herewith |
|
|
|
|
|
10.4 |
| Amendment to Technology Licensing Agreement between XGS and Michigan State University dated May 27, 2011 |
| Filed herewith |
|
|
|
|
|
10.5 |
| Lease between XG Sciences, Inc. and Dart Container of Michigan LLC, dated September 1, 2011 |
| Filed herewith |
|
|
|
|
|
10.6 |
| Amendment to Lease between XG Sciences, Inc. and Dart Container of Michigan LLC, dated August 15, 2011 |
| Filed herewith |
|
|
|
|
|
10.7 |
| Amendment to Lease between XG Sciences, Inc. and Dart Container of Michigan LLC, dated November 16, 2012 |
| Filed herewith |
|
|
|
|
|
10.8 |
| $300,000 Line of Credit Note, dated March 18, 2013, issued by |
| Filed herewith |
|
|
|
|
|
10.9 |
| Letter Agreement, dated March 18, 2013, by and between XG Sciences, Inc. and Michael R. Knox |
| Filed herewith |
|
|
|
|
|
10.10 |
| Shareholder Agreement, dated March 18, 2013, by and among XG Sciences, Inc. and the stockholder signatories thereto. |
| Filed herewith |
|
|
|
|
|
10.11 |
| Master Lease Agreement, dated March 18, 2013, by and between XG Sciences, Inc. and Aspen Advanced Opportunity Fund, LP |
| Filed herewith |
|
|
|
|
|
10.12 |
| Employment Agreement dated December 16, 2013, by and between Philip L. Rose and XG Sciences |
| Filed herewith |
|
|
|
|
|
10.13 |
| Employment Agreement dated January 13, 2014 by and between Michael R. Knox and XG Sciences |
| Filed herewith |
|
|
|
|
|
10.14 |
| $772,998.15 Secured Convertible Promissory Note, dated January 15, 2014, issued by XG Sciences, Inc. to Michael R. Knox |
| Filed herewith |
|
|
|
|
|
II-4
EXHIBIT NUMBER |
| DESCRIPTION |
| LOCATION |
10.15 |
| Second Amended & Restated Purchase Agreement, dated January 15, 2014, by and between XG Sciences, Inc. and Aspen Advanced Opportunity Fund, LP |
| Filed herewith |
|
|
|
|
|
10.16 |
| $4,178,330.92 Secured Convertible Promissory Note, dated January 15, 2014, issued by XG Sciences, Inc. and XG Sciences IP, LLC to Aspen Advanced Opportunity Fund, LP |
| Filed herewith |
|
|
|
|
|
10.17 |
| $1,300,000 Secured Convertible Promissory Note, dated March 18, 2014, issued by XG Sciences, Inc. and XG Sciences IP, LLC to Aspen Advanced Opportunity Fund, LP |
| Filed herewith |
|
|
|
|
|
10.18 |
| $900,000 Secured Convertible Promissory Note, dated March 31, 2014, issued byXG Sciences, Inc. and XG Sciences IP, LLC to Aspen Advanced Opportunity Fund, LP |
| Filed herewith |
|
|
|
|
|
10.19 |
| Amended & Restated Purchase Agreement, dated January 15, 2014, by and between XG Sciences, Inc. and XGS II, LLC |
| Filed herewith |
|
|
|
|
|
10.20 |
| $739,349.83 Secured Convertible Promissory Note, dated January 15, 2014, issued by XG Sciences, Inc. and XG Sciences IP, LLC to XGS II, LLC |
| Filed herewith |
|
|
|
|
|
10.21 |
| Purchase Agreement, dated January 15, 2014, by and between XG Sciences, Inc. and SVIC No. 15 New Technology Business Investment L.L.P. |
| Filed herewith |
|
|
|
|
|
10.22 |
| $3,000,000 Secured Convertible Promissory Note, dated January 15, 2014, issued by XG Sciences, Inc. and XG Sciences IP, LLC to SVIC No. 15 New Technology Business Investment L.L.P. |
| Filed herewith |
|
|
|
|
|
10.23 |
| License Agreement, dated January 15, 2014, by and between XG Sciences, Inc. and XG Sciences IP, LLC |
| Filed herewith |
|
|
|
|
|
10.24 |
| Second Amended and Restated Security Agreement, dated January 15, 2014, by and among XG Sciences, Inc., Aspen Advanced Opportunity Fund, LP, XGS II, LLC, SVIC No. 15 New Technology Business Investment L.L.P. |
| Filed herewith |
|
|
|
|
|
10.25 |
| Second Amended and Restated Intellectual Property Security Agreement, dated January 15, 2014, by and among XG Sciences, Inc., Aspen Capital Advisors LLC, Aspen Advanced Opportunity Fund, LP, XGS II, LLC, SVIC No. 15 New Technology Business Investment L.L.P. and Michael R. Knox |
| Filed herewith |
|
|
|
|
|
10.26 |
| Second Amended and Restated Intercreditor Agreement, dated January 15, 2014 by and among XG Sciences, Inc., Aspen Advanced Opportunity Fund, LP, XGS II, LLC, SVIC No. 15 New Technology Business Investment L.L.P. and Michael R. Knox |
| Filed herewith |
|
|
|
|
|
10.27 |
| Second Amended and Restated Registration Rights Agreement, dated January 15, 2014, by and among XG Sciences, Inc., Aspen Advanced Opportunity Fund, LP, XGS II, LLC and SVIC No. 15 New Technology Business Investment L.L.P. |
| Filed herewith |
|
|
|
|
|
10.28 |
| Voting Agreement, dated January 15, 2014, by and among XG Sciences, Inc., SVIC No. 15 New Technology Business Investment L.L.P and the stockholder signatories thereto. |
| Filed herewith |
|
|
|
|
|
10.29 |
| Assignment of Intellectual Property, dated January 15, 2014, by and between XG Sciences, Inc. and XG Sciences IP, LLC |
| Filed herewith |
|
|
|
|
|
II-5
EXHIBIT NUMBER |
| DESCRIPTION |
| LOCATION |
10.30 |
| Letter Agreement dated January 15, 2014, by and among XG Sciences, Inc., Aspen Advanced Opportunity Fund, LP, XGS II, LLC and SVIC No. 15 New Technology Business Investment L.L.P. and Michael Knox |
| Filed herewith |
|
|
|
|
|
10.31 |
| Form of Sales Agency Agreement |
| Filed herewith |
|
|
|
|
|
10.32 |
| Form of Subscription Agreement for Series B Unit Offering |
| Filed herewith |
|
|
|
|
|
10.33 |
| Form of Warrant for Series B Unit Offering |
| Filed herewith |
|
|
|
|
|
10.34 |
| Form of Note for December 2015 Private Placement |
| Filed herewith |
|
|
|
|
|
10.35 |
| Form of Warrant for December 2015 Private Placement |
| Filed herewith |
|
|
|
|
|
10.36 |
| Form of Subscription Agreement for December 2015 Private Placement |
| Filed herewith |
|
|
|
|
|
10.37 |
| Form of Subscription Agreement for Primary Offering |
| Filed herewith |
|
|
|
|
|
14 |
| Code of Ethics |
| Filed herewith |
|
|
|
|
|
21 |
| Subsidiary |
| Filed herewith |
|
|
|
|
|
23.1 |
| Consent of Kingery & Crouse P.A. |
| Filed herewith |
|
|
|
|
|
23.2 |
| Consent of Frazier & Deeter, LLC |
| Filed herewith |
|
|
|
|
|
23.3 |
| Consent of Foster Swift Collins & Smith PC |
| Incorporated by reference to Exhibit 5.1 herein |
|
|
|
|
|
23.4 |
| Consent of Prismark Partners, LLC |
| Filed herewith |
|
|
|
|
|
24.1 |
| Power of Attorney (included on signature page) |
| Filed herewith |
Item 17. Undertakings
The undersigned registrant hereby undertakes:
(1) To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) To include any prospectus required by Section 10(a)(3) of the Securities Act;
(ii) To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of a prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20 percent change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
(iii) To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement;
(2) That, for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
(3) To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
II-6
(4) That, for the purpose of determining liability of the registrant under the Securities Act to any purchaser:
If the registrant is subject to Rule 430C, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
(5) That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities:
The undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser:
(i) Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424 (§ 230.424 of this chapter);
(ii) Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
(iii) The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and
(iv) Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
(6) Include the following in a registration statement permitted by Rule 430A under the Securities Act of 1933 (§ 230.430A of this chapter):
The undersigned registrant hereby undertakes that:
(1) For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in a form of prospectus filed by the registrant pursuant to Rule 424(b) (1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective.
(2) For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the Registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
II-7
Pursuant to the requirements of the Securities Act, the registrant has duly caused this amendment to the Registration Statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Lansing, State of Michigan, on the 26 day of January, 2016.
|
|
|
| XG SCIENCES, INC. |
|
|
|
|
|
|
| By: |
| /s/ Philip L. Rose |
|
| Name: |
| Philip L. Rose |
|
| Title: |
| Chief Executive Officer, President, |
We, the undersigned officers and directors of XG Sciences, Inc., hereby severally constitute and appoint Philip L. Rose and Michael R. Knox and each of them singly (with full power to each of them to act alone), our true and lawful attorneys-in-fact and agents, with full power of substitution and resubstitution in each of them for him and in his name, place and stead, and in any and all capacities, to sign any and all amendments (including post-effective amendments) to this Registration Statement on Form S-1 (or any other Registration Statement for the same offering that is to be effective upon filing pursuant to Rule 462(b) under the Securities Act), and to file the same, with all exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite or necessary to be done in and about the premises, as full to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them or their or his substitute or substitutes may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act, this Registration Statement has been signed by the following persons in the capacities and on the dates indicated.
Name |
| Title(s) |
| Date |
/s/ Philip L. Rose |
|
|
|
|
Philip L. Rose |
| Chief Executive Officer, President, Treasurer and Director, Principal Executive Officer |
| January 26, 2016 |
|
|
|
|
|
/s/ Michael R. Knox |
|
|
|
|
Michael R. Knox |
| Senior Vice President, Secretary and Director, Principal Financial Officer |
| January 26, 2016 |
|
|
|
|
|
/s/ Arnold A. Allemang |
|
|
|
|
Arnold A. Allemang |
| Director |
| January 26, 2016 |
|
|
|
|
|
/s/ Wonil Kim |
|
|
|
|
Wonil Kim |
| Director |
| January 26, 2016 |
|
|
|
|
|
/s/ Steven C. Jones |
|
|
|
|
Steven C. Jones |
| Director |
| January 26, 2016 |
|
|
|
|
|
/s/ Michael D. Pachos |
|
|
|
|
Michael D. Pachos |
| Director |
| January 26, 2016 |
|
|
|
|
|
/s/ Ravi Shanker |
|
|
|
|
Ravi Shanker |
| Director |
| January 26, 2016 |
II-8