As filed with the Securities and Exchange Commission on April 8, 2014
Registration No. 333-______
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM S-1
REGISTRATION STATEMENT UNDER THE SECURITIES ACT OF 1933
STEVIA CORP.
(Exact name of registrant as specified in its charter)
| Nevada | 700 | 98-0537233 | ||
| (State or jurisdiction of | (Primary Standard Industrial | (I.R.S. Employer | ||
| incorporation or organization) | Classification Code Number) | Identification No.) |
7117 US 31S
Indianapolis, IN 46227
(888) 250-2566
(Address and telephone number of principal executive offices and principal place of business)
CSC Services of Nevada, Inc.
2215-B Renaissance Drive
Las Vegas, NV 89119
(702) 740-4244
(Name, address and telephone number of agent for service)
Copies to:
Mark C. Lee
Saxon Peters
GREENBERG TRAURIG, LLP
1201 K Street, Suite 1100
Sacramento, California 95814
Telephone: (916) 442-1111
Facsimile: (916) 448-1709
Approximate date of proposed sale to the public:
From time to time after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, check the following box.þ
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer ¨ | Accelerated filer ¨ | Non-accelerated filer ¨ | Smaller reporting company þ |
| (Do not check if a smaller reporting company) | |||
| |||
CALCULATION OF REGISTRATION FEE
| Amount of shares | Proposed maximum | Proposed maximum | Amount of | |||||||||||||
| Title of each class of | to be | offering price | aggregate | Registration | ||||||||||||
| securities to be registered | Registered | per share | offering price | Fee | ||||||||||||
| Common Stock Underlying the Principal of Convertible Notes | 10,674,182 | (1) | $ | 0.266 | (2) | $ | 2,839,332.41 | $ | 365.71 | |||||||
| Common Stock Underlying the Interest of Convertible Notes | 508,905 | (3) | $ | 0.266 | (2) | $ | 135,368.73 | $ | 17.44 | |||||||
| Common Stock Underlying the Principal of Convertible Notes | 12,809,018 | (4) | $ | 0.266 | (2) | $ | 3,407,198.79 | $ | 438.85 | |||||||
| Common Stock Underlying the Interest of Convertible Notes | 610,687 | (5) | $ | 0.266 | (2) | $ | 162,442.74 | $ | 20.92 | |||||||
| Common Stock Underlying Warrants | 20,296,139 | (6) | $ | 0.053365 | $ | 1,083,103.46 | $ | 139.50 | ||||||||
| Common Stock Underlying a Convertible Note | 3,000,000 | (7) | $ | 0.266 | (2) | $ | 798,000.00 | $ | 102.78 | |||||||
| Total | 47,898,931 | $ | 8,425,446.13 | $ | 1085.20 | |||||||||||
| (1) | Represents shares of common stock issuable by the registrant upon the conversion of the principal amount of the registrant’s Senior Convertible Note issued March 3, 2014 (the “Initial Nomis Bay Note”). |
| (2) | Estimated solely for the purpose of calculating the registration fee pursuant to Rule 457(c) under the Securities Act of 1933, as amended, based on the average high and low prices of the common stock as reported on the OTCQB on March 26, 2014. | |
| (3) | Represents shares of common stock issuable by the registrant upon the conversion of interest accrued under the Initial Nomis Bay Note. | |
| (4) | Represents shares of common stock issuable by the registrant upon the conversion of the principal amount of the registrant’s Senior Convertible Note to be issued to Nomis Bay Ltd. (the “Secondary Nomis Bay Note”) after the effectiveness of this registration statement upon the terms and conditions set forth in the Securities Purchase Agreement dated March 3, 2014. | |
| (5) | Represents shares of common stock issuable by the registrant upon the conversion of interest to be accrued under the Secondary Nomis Bay Note. |
| (6) | Represents the number of shares of common stock offered for resale following the exercise of certain warrants to purchase common stock. |
| (7) | Represents shares of common stock issuable by the registrant upon the conversion of the registrant’s promissory note issued July 10, 2013(the “JMJ Note Shares”) |
In the event of stock splits, stock dividends, or similar transactions involving the Registrant’s common stock, the number of Shares registered shall, unless otherwise expressly provided, automatically be deemed to cover the additional securities to be offered or issued pursuant to Rule 416 promulgated under the Securities Act of 1933, as amended (the “Securities Act”); provided however that any additional shares received due to subsequent equity issuances by the Registrant at a lower price per share than the current exercise price of any applicable warrants would not be covered by this registration statement and would require separate registration or an exemption prior to sale.
We hereby amend this registration statement on such date or dates as may be necessary to delay its effective date until we shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act or until the registration statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
SUBJECT TO COMPLETION, DATED APRIL 8, 2014
PROSPECTUS
47,898,931 Shares of Common Stock
STEVIA CORP.
Common Stock
This prospectus relates to the resale of 47,898,931 shares of common stock, by the selling stockholders named in this prospectus. The shares of common stock subject to this prospectus include:
| (i) | 24,602,792 shares of common stock issuable upon conversion of the principal amount of the registrant’s Senior Convertible Note issued March 3, 2014 (the “Nomis Bay Note Shares”). Based upon current market price of the Company’s common stock, the amount of Nomis Bay Note Shares being registered may be in excess of the number of shares into which the Senior Convertible Note may currently be converted, however the parties have agreed upon the aggregate number of shares to be registered to account for market fluctuations; |
| (ii) | 23,026,318 shares of common stock issuable following the exercise of certain warrants issued in accordance with a Warrant Exercise Reset Offer Letter Agreement entered into on May 3, 2013 as adjusted pursuant to its terms for certain dilutive issuances, less 5,290,665 shares of common stock previously registered pursuant to the registrant’s Registration Statement on Form S-1/A filed December 30, 2013 (the “Anson Reset Shares”); |
| (iii) | 2,560,486 shares of common stock issuable following the exercise of certain warrants issued to a selling stockholder in accordance with a Securities Purchase Agreement entered into on August 1, 2012, as adjusted pursuant to the anti-dilution provision contained therein and the Common Stock Purchase Warrant issued February 20, 2014, less 683,202 shares of common stock previously registered pursuant to the registrant’s Registration Statement on Form S-1/A filed December 30, 2013, plus an additional 683,202 shares of common stock issuable following the exercise of the warrant issued February 20, 2014 with an exercise price of $0.053365 per share (the “Cranshire Warrant Shares”); and |
| (iv) | 3,000,000 shares of common stock issuable upon the conversion of the registrant’s $400,000 Promissory Note issued July 10, 2013 (the “JMJ Note Shares”); |
We will not receive any proceeds from the resale of any of the shares offered hereby. We may receive gross proceeds of up to $1,083,103.46 if all of the warrants set forth above (the “Warrants”) are exercised for cash. The proceeds will be used for working capital or general corporate purposes. We will bear all costs associated with this registration.
Our common stock is quoted on the OTCQB under the symbol “STEV.” On March 26, 2014, the closing bid price of our common stock was $0.28 per share.
INVESTING IN OUR COMMON STOCK INVOLVES A HIGH DEGREE OF RISK. SEE “RISK FACTORS” BEGINNING ON PAGE 2 OF THIS PROSPECTUS.
NEITHER THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED OF THESE SECURITIES OR DETERMINED IF THIS PROSPECTUS IS TRUTHFUL OR COMPLETE. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENSE.
The information in this Prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the Securities and Exchange Commission becomes effective. This Prospectus is not an offer to sell these securities and we are not soliciting an offer to buy these securities in any state where the offer or sale is not permitted or would be unlawful prior to registration or qualification under the securities laws of any such state.
TABLE OF CONTENTS
| Page | |
| PART I - INFORMATION REQUIRED IN PROSPECTUS | |
| PROSPECTUS SUMMARY | 1 |
| DISCLOSURE REGARDING FORWARD-LOOKING STATEMENTS | 7 |
| RISK FACTORS | 7 |
| RISKS RELATED TO OUR BUSINESS AND INDUSTRY | 7 |
| RISKS RELATED TO DOING BUSINESS IN VIETNAM AND OTHER DEVELOPING COUNTRIES | 12 |
| RISKS RELATED TO AN INVESTMENT IN OUR SECURITIES | 14 |
| USE OF PROCEEDS | 15 |
| DETERMINATION OF OFFERING PRICE | 15 |
| SELLING SECURITY HOLDER | 15 |
| PLAN OF DISTRIBUTION | 17 |
| DESCRIPTION OF SECURITIES TO BE REGISTERED | 18 |
| INTERESTS OF NAMED EXPERTS AND COUNSEL | 20 |
| INFORMATION WITH RESPECT TO THE REGISTRANT | 20 |
| PROPERTIES | 32 |
| LEGAL PROCEEDINGS | 32 |
| MARKET FOR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS | 32 |
| MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS | 33 |
| DIRECTORS AND EXECUTIVE OFFICERS | 37 |
| EXECUTIVE COMPENSATION | 38 |
| SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT | 39 |
| CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS AND DIRECTOR INDEPENDENCE | 40 |
| DISCLOSURE OF COMMISSION POSITION OF INDEMNIFICATION FOR SECURITIES ACT LIABILITIES | 41 |
| WHERE YOU CAN FIND MORE INFORMATION | 41 |
| FINANCIAL STATEMENTS | F-1 |
| PART II - INFORMATION NOT REQUIRED IN PROSPECTUS | 42 |
| OTHER EXPENSES OF ISSUANCE AND DISTRIBUTION | 42 |
| INDEMNIFICATION OF DIRECTORS AND OFFICERS | 42 |
| RECENT SALES OF UNREGISTERED SECURITIES | 44 |
| EXHIBIT INDEX | 49 |
| UNDERTAKINGS | 50 |
| SIGNATURES | 53 |
You should rely only on the information contained in this Prospectus. We have not authorized anyone to provide you with different information. We are not making an offer of these securities in any state where the offer is not permitted.
PROSPECTUS SUMMARY
You should read the following summary together with the more detailed information and the financial statements appearing elsewhere in this Prospectus. This Prospectus contains forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those anticipated in these forward-looking statements as a result of certain factors, including those set forth under “Risk Factors” and elsewhere in this Prospectus. Unless the context indicates or suggests otherwise, references to “we,” “our,” “us,” the “Company,” “Stevia” or the “Registrant” refer to Stevia Corp., a Nevada corporation and its subsidiaries.
Overview
Stevia Corp. was incorporated on May 21, 2007 in the State of Nevada. On June 23, 2011, we closed a voluntary share exchange transaction with Stevia Ventures International Ltd., a business company incorporated in the British Virgin Islands, pursuant to which we acquired the rights to purchase certain strains of stevia leaf growing in Vietnam, including certain assignable exclusive purchase contracts and an assignable supply agreement related to the stevia leaf.
We are a farm management company primarily focused on crop agronomics from plant breeding to good agricultural practices to development of crop derived products, which can be used for human consumption as well as for aquaculture and agriculture applications.
We have established a field test research center in Vietnam on 10 Ha (25 acres) of leased land which is designed to support our commercial field trials that are on-going in Vietnam and began our first commercial trial harvests in March 2012. We confirmed elite plant varieties, developed propagation techniques, conducted field trials across several provinces, documented local operating procedures and post-harvest techniques, and began commercial harvests in February 2013.
In July 2012 we formed a joint venture with Tech-New Bio-Technology, a technology company in Hong Kong and acquired intellectual property covering several formulations utilizing stevia extracts together with probiotics and enzymes which have applications for agriculture, aquaculture and post harvest processing. We do not operate an extraction facility, but Tech-New Bio-Technology’s affiliate company in China has technologies and the facility for the extraction and refinement of high purity stevia; we entered into a multi-year supply contract in March 2012 where they are committed to purchase all of our stevia leaf production for the first two years and we also have the ability to use the resulting stevia extract to formulate our products. While we believe that our joint venture with Tech-New Bio-Technology will increase the visibility of our intended services and products, there is no guarantee that such visibility will occur.
Our formulated products consist of ecological fertilizers that address soil acidification, compaction and fertility decline caused by chemical fertilizer overuse; foliar fertilizers that help plants resist infection and disease; feed formulations for livestock, fish and shrimp that enhance digestion and help strengthen immunity; microbiological preparations that address pollution in marine environments that negatively impact aquaculture activities; and natural preparations which aid in the preservation of crops after harvest and during processing. We also provide private label pure stevia extracts which are suitable for food and beverage applications.
In August of 2012 we began to use our formulated products as feed and fertilizer inputs under our farm management model and currently service several commercial operations providing feed supplements for shrimp and fish production and fertilizer inputs for farmland as well as using it on our own trial farms.
In September 2012 we began providing samples of stevia extract to food and beverage companies and we are working closely with local parties in several South East Asian countries to provide technical information in support of recipe development. Although we believe that this product line will have growth potential, there is no guarantee that a high volume of stevia will be utilized by our customers. We expect companies will take another year to plan product launches.
In January 2014 we entered into a Farm Management and Technology Agreement with ebbu LLC to provide farm management consultancy and technical expertise related to growing the cannabis plant and extracting its cannabinoids. The cannabis plant produces many chemical compounds called cannabinoids and there are more than 85 different cannabinioids that have been identified and isolated from the cannabis plant that exhibit varied effects and many of these are being studied for their psychoactive and medical properties. Cannabis plants that have been bred to produce high levels of tetrahydrocannabinol (“THC”), a psychoactive constituent, are commonly referred to as marijuana. Current federal and most state regulations prevent us from participating directly in the marijuana industry and we cannot guarantee that our services or technology will provide value if the laws do not evolve in favor of marijuana production and the commercial sale of marijuana derived products.
In February 2014 we registered a wholly owned subsidiary, Real Hemp LLC, as part of our strategy to enter the US hemp industry to import, manufacture and license products containing hemp seeds, oil, protein, milk, fiber and cannabidiol. Hemp is the common term for cannabis plants that produce very low levels of THC and are grown for industrial purposes and foodstuff products. Cannabidiol (“CBD”) is one of the active cannabinoids in the cannabis plant and is a major constituent of hemp, accounting for up to 40% of the plants extract, and is considered to have a wider scope of medical applications than THC. Hemp products, including CBD extracts, can be legally imported and traded in the United States. We are currently exploring product opportunities and expect to launch our first products during 2014. There is no guarantee that we will launch products containing hemp or that such products will be successful.
| 1 |
All of our formulated products used for agriculture and aquaculture are approved for use in our areas of operation and the largest obstacle we will face will be farmer confidence to use new products. We believe that we can overcome this obstacle by building a successful and demonstrable track record working with the current operations of Tech-New Bio-Technology and its affiliates. All of the ingredients in the products are natural compounds and are approved by the major developed countries if we choose to expand to other markets in the future. A list of the major developed countries that have approved the use of stevia as a food additive can be found on page 29.
The stevia industry is segmented into several business processes, which can broadly be categorized as i) plant breeding and propagation, ii) farming, iii) extraction and refining, iv) product formulation, v) distribution and retail. As we achieve vertical integration along the supply chain we will continue to focus on acquisitions and intellectual property development to support further downstream integration into the agriculture and aquaculture sectors. We believe that over the long-run this will position the Company to become an industry leader, producing a number of value-added stevia-enhanced products.
Our Business
We are a farm management company primarily focused on stevia agronomics from plant breeding to good agricultural practices to development of stevia derived products which can be used for human consumption as well as for aquaculture and agriculture applications. We plan to invest in research and development and intellectual property acquisition and provide farm management services to contract growers and other industry growers integrating our stevia focused research and development and intellectual property acquisitions.
Our farm management services include training the farmers on the correct protocols and methodologies and providing ongoing technical assistance during the crop cycle as well as providing inputs such as the seedlings, fertilizers and additives they are required to use.
We employ our services under three business models which we classify as 1) contract farming model, 2) revenue share model and 3) product supply model.
Under the contract farming and revenue share models we do not charge for the services and inputs, but rather our services provide us with a competitive advantage to secure growers who are willing to dedicate their land and resources to grow crops with an expectation of high yielding, high quality crops and guaranteed purchase prices. Under these models we will generate our revenue from the crops that are grown and we only enter into production agreements with growers when there is already a committed buyer for the end crop. Under the contract farming model we will purchase the crop from the grower at a fixed price and sell to our own customer. Under the revenue share model, the grower already has their own buyer and we will share the revenue.
Under the product supply model we will market our products in combination with technical services to buyers and charge a fee. We believe that this model will contribute a small part of our overall revenue initially until we establish a proven track record and solid reputation for our services and products under the first two models. We do not expect to focus on providing strictly farm management or technical services for a fee and it is difficult to estimate what we would charge for such services.
We continue to focus on research and development to further evolve and develop new protocols, methodologies and intellectual properties and believe that this will be key to maintain our competitive advantage.
We utilize the contract farming model to produce stevia leaf for our trial harvests and use the stevia extracts to produce our proprietary formulated products, which we are applying under the revenue share model to an aquaculture operation beginning August 2012 and a chili operation beginning October 2012.
Our mission is to maximize stockholder value by consistently developing and acquiring the latest intellectual property and expanding our suite of formulated products and their applications and leveraging our farm management business model to maximize market penetration and revenue margins.
To achieve these goals we intend to develop a suite of intellectual property relating to stevia and its extracts that will enhance the value of our farm management operations. Through our relationships with Tech-New Bio-Technology, Growers Synergy and local institutes, we are exploring the market for commercial applications of stevia which will be vertically integrated into our services and production. We have engaged Growers Synergy, a regional farm management services provider, to provide farm management operations and back-office and regional logistical support for our Vietnam and Indonesia operations for a period of two years. George Blankenbaker, our president, director and stockholder is the managing director of Growers Synergy. Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and Mr. Blankenbaker controls a 49% interest in Growers Fresh.
| 2 |
Our current burn rate is approximately $150,000 per month and we currently have approximately $350,000 in cash on hand. We are dependent on additional capital to continue to operate. Failure to complete a financing will have an adverse effect on our ability to operate and execute our business plan. We believe that $3 million of funding is sufficient for us to break-even and achieve self-sufficiency on a cash flow basis. Based on the current burn rate, the Company does not currently have sufficient capital to operate and we are doing so on a very limited budget, relying primarily on our goodwill with Growers Synergy and our other vendors, and during this period we will need to raise additional capital and generate revenue. As a result, our accounts payable are expected to grow. However, there are no assurances that Growers Synergy or our other vendors will continue to extend credit to the Company, and if they cease extending credit to us, and we are unable to raise capital or generate sufficient revenue, we will have to liquidate or sell certain assets.
Our target markets are initially Vietnam, Indonesia and China where we have contracted with growers and have established our own nurseries and test fields. In China we are producing our proprietary formulated products and applying them to aquaculture projects under our revenue share model. Although our priority is Asia, our services are not limited to specific countries and we plan to pursue viable opportunities in other markets.
Our operations to-date have primarily consisted of securing purchase and supply contracts, office space and a research center, developing relationships with potential partners, and developing products derived from the stevia plant. We have earned limited revenues since inception. For the nine month period ended December 31, 2013 we incurred a net loss of $2,017,484 and for the period from inception (April 11, 2011) to March 31, 2013, we have incurred a net loss of $4,359,415. Our assets total $3,641,781 and $2,194,251 as of as of December 31, 2013 and March 31, 2013, respectively. Further, our auditors have issued a going concern opinion in their audit report dated July 15, 2013. This means that there is substantial doubt that we can continue as an on-going business for the next twelve months unless we obtain additional capital.
Recent Developments
The table below sets forth shares of our common stock that have been recently issued in exchange for certain services and rights.
| Date | Issuance of Shares for Services and/or Rights | |
| February 26, 2014 | We issued an aggregate of 28,300,000 shares of our common stock to various service providers in exchange for services rendered, including 20,000,000 shares to Blankenbaker Ventures (Asia) Pte. Ltd. on behalf of George Blankenbaker, our president, director and stockholder and 3,000,000 shares to Growers Synergy Pte Ltd., a corporation organized under the laws of Singapore (“Growers Synergy”). Thomas Ong, a director of the Company is a director of Growers Synergy and is also a 25% shareholder of Agriventure Pte Ltd., which is a 49% shareholder of Growers Synergy. Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and Mr. Blankenbaker controls a 49% interest in Growers Fresh.
| |
| February 26, 2014 | We issued 16,744,682 shares of our common stock to Blankenbaker Ventures (Asia) Pte. Ltd. on behalf of George Blankenbaker, our president, director and stockholder, in exchange for the cancellation of approximately $893,579.93 of working capital advances provided by Mr. Blankenbaker and his affiliated companies. | |
Corporate Information
Our principal executive offices are located at 7117 US 31 S., Indianapolis, IN, 46227. Our telephone number is 888-250-2566. We maintain a corporate website at http://www.steviacorp.us.
Stock Transfer Agent
Our stock transfer agent is Securities Transfer Corporation, and is located at 2591 Dallas Parkway, Suite 102, Frisco, Texas 75034. The agent’s telephone number is 469-633-0101.
| 3 |
The Offering
| Issuer | Stevia Corp. | |
| Securities Offered for Resale | 47,898,931 shares of Common Stock underling convertible notes and warrants to purchase Common Stock | |
| Common Stock Outstanding Before the Offering | 145,972,713 shares | |
| Common Stock to be Outstanding After the Offering assuming all of the Securities are Resold | 193,871,644 shares | |
| Use of Proceeds | We will not receive any proceeds from the resale of the shares of common stock underlying the Warrants. We may receive proceeds in the event the Warrants are exercised for cash. Such proceeds from the offering will be used for working capital and general corporate purposes. See “Use of Proceeds.” | |
| Trading | Our common stock is quoted on the OTCQB under the symbol “STEV.” | |
| Risk Factors | You should carefully consider the information set forth in the section entitled “Risk Factors” beginning on page 2 of this prospectus in deciding whether or not to invest in our common stock. |
Nomis Bay Note Shares
This offering, with respect to the Nomis Bay Note Shares, relates to the resale of shares of common stock of the Company underlying a Senior Convertible Note, issued March 3, 2014 (the “Nomis Bay Note”). On March 3, 2014, pursuant to a Securities Purchase Agreement (the “Purchase Agreement”) with Nomis Bay Ltd., a Bermuda company (“Nomis Bay”), we issued the Nomis Bay Note with an initial principal amount of $500,000 for a purchase price of $340,000. The Nomis Bay Note matures on December 27, 2014 (subject to extension as provided in the Nomis Bay Note) and, in addition to the 32% original issue discount, accrues interest at the rate of 8% per annum. The Purchase Agreement also provides that, upon the terms and subject to the conditions set forth therein, the Company may require Nomis Bay to purchase from the Company on or prior to the 10th trading day after the effective date of the registration statement registering the shares issuable upon conversion of the Initial Convertible Note, an additional senior convertible note with an initial principal amount of $600,000 (the “Additional Convertible Note” and together with the Nomis Bay Note, the “Convertible Notes”) for a purchase price of $600,000. If issued, the Additional Convertible Note will mature on the date that is the 10-month anniversary of the date of issuance of the Additional Convertible Note (subject to extension as provided in the Initial Convertible Note) and will accrue interest at the rate of 8% per annum. The Nomis Bay Note is convertible at any time, in whole or in part, at Nomis Bay’s option into shares of the Company’s common stock, par value $0.001 per share (the “Common Stock”), at a conversion price equal to the lesser of (i) the product of (x) the arithmetic average of the lowest three (3) volume weighted average prices of the Common Stock during the 10 consecutive trading days ending and including the trading day immediately preceding the applicable conversion date and (y) 40% (the “Variable Conversion Price”), and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). If issued, the Additional Convertible Note will be convertible at any time, in whole or in part, at Nomis Bay’s option into shares of Common Stock at a conversion price that will be equal to the lesser of (i) the Variable Conversion Price and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). At no time will Nomis Bay be entitled to convert any portion of the Convertible Notes to the extent that after such conversion, Nomis Bay (together with its affiliates) would beneficially own more than 4.99% of the outstanding shares of Common Stock as of such date. The shares of common stock underlying the Nomis Bay Note were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
Anson Reset Shares
This offering, with respect to the Anson Reset Shares, relates to the resale of shares of common stock of the Company underlying three warrants in the amounts of 1,877,333, 1,066,666 and 2,346,666, with exercise prices of $0.20, $0.25 and $0.25 per share (the “Anson Warrants”). Pursuant to the terms of a warrant exercise reset offer letter between the Company and the investor, the Anson Warrants were issued in consideration for the investor’s agreement to immediately cash exercise an existing warrant to purchase 853,333 shares of common stock of the Company at an exercise price of $0.20 per share for aggregate consideration to the Company of $170,666 originally issued pursuant to the terms of the Securities Purchase Agreement, dated August 1, 2012 (the “Purchase Agreement”). Each Anson Warrant has a five year term and was issued on May 3, 2013. Each Anson Warrant provides for adjustment of the exercise price and share amount in the event of certain dilutive issuances by the Company. On February 20, 2014, the Company issued a notice to the holder that the aggregate number of Anson Warrants had been adjusted to 23,026,318 and the exercise price of each had been adjusted to $0.053365 as a result of certain other offerings of the Company. 5,290,665 of the shares underlying the Anson Warrants were previously registered by the Company pursuant to the Registration Statement on Form S-1/A filed December 30, 2013. The warrants and the shares of common stock underlying the warrants were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
| 4 |
Cranshire Warrant Shares
This offering, with respect to the Cranshire Warrant Shares, relates to the resale of the shares of common stock underlying a warrant in the amount of 2,560,486 with an exercise price of $0.053365. The warrant has a five (5) year term and was issued pursuant to the terms of the Purchase Agreement. The warrant was originally issued in the amount of 213,334 shares at an exercise price of $0.6405. Pursuant to the anti-dilution adjustment provision contained therein, on February 20, 2014, the Company issued a notice to the holder that the total share amount had been increased to 2,560,486 and the exercise price had been reduced to $0.053365 as a result of certain other offerings of the Company. 683,202 of the shares underlying the Cranshire Warrants were previously registered by the Company pursuant to the Registration Statement on Form S-1/A filed December 30, 2013. Additionally, on February 20, 2014 in consideration for the investor’s agreement to immediately cash exercise a portion of the Cranshire Warrants for aggregate consideration to the Company of $36,459, the Company agreed to issue the investor an additional warrant to purchase 683,202 shares of common stock. The warrants and the shares of common stock underlying the warrants were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
JMJ Note Shares
This offering, with respect to the JMJ Note Shares, relates to the resale of 3,000,000 shares of common stock of the Company underlying a $400,000 Promissory Note issued July 10, 2013 (the “JMJ Note”). The JMJ Note is subject to a one-time interest charge equal to 12% of the principal sum and is due and payable July 10, 2014. The JMJ Note provides that it may be converted into common stock of the Company at any time at the election of the holder, at a conversion price equal to the lesser of $0.26 or 65% of the lowest trade price in the 25 trading days previous to the conversion. The JMJ Note and the shares of common stock underlying the JMJ Note were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
SUMMARY OF FINANCIAL INFORMATION
The following selected financial information is derived from the Company’s Financial Statements appearing elsewhere in this Prospectus and should be read in conjunction with the Company’s Financial Statements, including the notes thereto, appearing elsewhere in this Prospectus.
| 5 |
Summary of Statements of Operations
For the Three Months Ended December 31, 2013:
| Total revenue | $ | 388,746 | ||
| Net income | (831,410 | ) | ||
| Net income per common share (basic and diluted) | $ | (0.01 | ) | |
| Weighted average common shares | 79,632,959 |
For the Nine Months Ended December 31, 2013:
| Total revenue | $ | 1,893,865 | ||
| Net loss | (2,017,484 | ) | ||
| Net loss per common share (basic and diluted) | $ | (0.03 | ) | |
| Weighted average common shares | 72,842,975 |
For the Fiscal Year Ended March 31, 2013:
| Total revenue | $ | 2,168,093 | ||
| Net loss | (2,035,864 | ) | ||
| Net loss per common share (basic and diluted) | $ | (0.03 | ) | |
| Weighted average common shares | 62,092,487 |
Statement of Financial Position
| December 31, 2013 | ||||
| Cash | $ | 85,366 | ||
| Accounts receivable | $ | 164,988 | ||
| Seeds | $ | 1,807,000 | ||
| Prepayments and other current assets | $ | 74,946 | ||
| Total current assets | $ | 2,132,300 | ||
| Total assets | $ | 3,641,781 | ||
| Total current liabilities | $ | 2,591,869 | ||
| Stockholders’ equity | $ | 798,339 | ||
| Non-controlling interest | $ | (345,946 | ) | |
| Equity | $ | 452,393 | ||
| Total liabilities and equity | $ | 3,641,781 | ||
| 6 |
| March 31, 2013 | ||||
| Cash | $ | 424,475 | ||
| Accounts Receivable | 158,008 | |||
| Prepayments and other current assets | 33,096 | |||
| Total current assets | 615,579 | |||
| Total assets | $ | 2,194,251 | ||
| Total current liabilities | $ | 1,457,531 | ||
| Stockholders’ equity | $ | 464,765 | ||
| Non-controlling interest | $ | (214,158 | ) | |
| Equity | $ | 250,607 | ||
| Total liabilities and equity | $ | 2,194,251 | ||
DISCLOSURE REGARDING FORWARD-LOOKING STATEMENTS
Except for statements of historical facts, this Prospectus contains forward-looking statements involving risks and uncertainties. The words “anticipate,” “believe,” “estimate,” “expect,” “future,” “intend,” “plan” or the negative of these terms and similar expressions or variations thereof are intended to forward looking statements. Such statements reflect the current view of the Registrant with respect to future events and are subject to risks, uncertainties, assumptions and other factors (including the risks contained in the section of this registration statement on Form S-1 entitled “Risk Factors”) relating to the Registrant’s industry, the Registrant’s operations and results of operations and any businesses that may be acquired by the Registrant. Should one or more of these risks or uncertainties materialize, or should the underlying assumptions prove incorrect, actual results may differ significantly from those anticipated, believed, estimated, expected, intended or planned.
Although the Registrant believes that the expectations reflected in the forward looking statements are reasonable, the Registrant cannot guarantee future results, levels of activity, performance or achievements. Except as required by applicable law, including the securities laws of the United States, the Registrant does not intend to update any of the forward-looking statements to conform these statements to actual results. The following discussion should be read in conjunction with the Registrant’s financial statements and the related notes included in this registration statement on Form S-1.
RISK FACTORS
You should carefully consider the risks described below together with all of the other information included in our public filings before making an investment decision with regard to our securities. If any of the following events described in these risk factors actually occurs, our business, financial condition or results of operations could be harmed. In that case, the trading price of our common stock could decline, and you may lose all or part of your investment.
RISKS RELATING TO OUR BUSINESS AND INDUSTRY
We have a limited operating history on which to evaluate our business or base an investment decision.
Our business prospects are difficult to predict because of our limited operating history, early stage of development and unproven business strategy. Stevia is still a relatively new product in the sweetener marketplace and it has historically not been commercially grown in Vietnam or many of our other target locations. Both the continued growth of the stevia market in general, and our ability to introduce commercial development of stevia to new regions, face numerous risks and uncertainties. In particular, we have not proven that we can produce stevia in a manner that enables us to be profitable and meet manufacturer requirements, develop intellectual property to enhance stevia production, develop and maintain relationships with key growers and strategic partners to extract value from our intellectual property, raise sufficient capital in the public and/or private markets, or respond effectively to competitive pressures. If we are unable to accomplish these goals, our business is unlikely to succeed and you should consider our prospects in light of these risks, challenges and uncertainties.
| 7 |
We have incurred significant losses and our auditors have expressed uncertainty about our ability to continue as a going concern.
Our auditors have expressed uncertainty as to our ability to continue as a going concern as of our fiscal year ended March 31, 2013. As of December 31, 2013, we had an accumulated deficit of $6,376,899. We anticipate that our existing cash and cash equivalents will not be sufficient to fund our longer term business needs and we will need to generate additional revenue or receive additional investment in the Company to continue operations. Such financing may not be available in sufficient amounts, or on terms acceptable to us and may dilute existing stockholders.
If we fail to raise additional capital, our ability to implement our business model and strategy could be compromised.
We have limited capital resources and operations. On March 3, 2014, we raised $340,000 (subject to certain fees) through the issuance of a convertible promissory note (the “Initial Convertible Note”), pursuant to a Securities Purchase Agreement (the “Purchase Agreement”) with Nomis Bay Ltd., a Bermuda company (“Nomis Bay”). The Purchase Agreement provides that, upon the terms and subject to the conditions set forth therein, we may require Nomis Bay to purchase from the Company on or prior to the 10th trading day after the effective date of the registration statement registering the shares issuable upon conversion of the Initial Convertible Note, an additional senior convertible note with an initial principal amount of $600,000 (the “Additional Convertible Note”) for a purchase price of $600,000. There is no guarantee that we will be able to meet the conditions for the funding of the Additional Convertible Note and investors should not expect us to be able to do so.
To date, our operations have been funded entirely from the proceeds from debt and equity financings. We expect to require substantial additional capital in the near future to develop our intellectual property base and to establish the targeted levels of commercial production of stevia. We may not be able to obtain additional financing on terms acceptable to us, or at all. Even if we obtain financing for our near term operations, we expect that we will require additional capital beyond the near term. If we are unable to raise capital when needed, our business, financial condition and results of operations would be materially adversely affected, and we could be forced to reduce or discontinue our operations.
We face intense competition which could prohibit us from developing a customer base and generating revenue.
The industries within which we compete, including the sweetener industry and the fertilizer and feed industries, are highly competitive with companies that have greater capital resources, facilities and diversity of product lines. Additionally, if demand for stevia continues to grow, we expect many new competitors to enter the market as there are no significant barriers to stevia production. More established agricultural companies with much greater financial resources which do not currently compete with us may be able to easily adapt their existing operations to production of stevia. Due to this competition, there is no assurance that we will not encounter difficulties in obtaining revenues and market share or in the positioning of our services or that competition in the industry will not lead to reduced prices for the stevia leaf. Our competitors may also introduce new non-stevia based low-calorie sweeteners or be successful in developing a fermentation-derived stevia ingredient or other alternative production method which could also increase competition and decrease demand for stevia-based products.
Inability to protect our proprietary rights could damage our competitive position.
Our business will be heavily dependent upon the intellectual property we develop or acquire. Any infringement or misappropriation of our intellectual property could damage its value and limit our ability to compete. We will rely on patents, copyrights, trademarks, trade secrets, confidentiality provisions and licensing arrangements to establish and protect our intellectual property. We may have to engage in litigation to protect the rights to our intellectual property, which could result in significant litigation costs and require a significant amount of our time. In addition, our ability to enforce and protect our intellectual property rights may be limited in certain countries outside the United States, which could make it easier for competitors to capture market position in such countries by utilizing technologies that are similar to those developed or licensed by us.
Competitors may also harm our sales by designing products that mirror the capabilities of our products or technology without infringing our intellectual property rights. If we do not obtain sufficient protection for our intellectual property, or if we are unable to effectively enforce our intellectual property rights, our competitiveness could be impaired, which would limit our growth and future revenue.
A successful claim of infringement against us could result in a substantial damage award and materially harm our financial condition. Even if a claim against us is unsuccessful, we would likely have to devote significant time and resources to defending against it.
We may also find it necessary to bring infringement or other actions against third parties to seek to protect our intellectual property rights. Litigation of this nature, even if successful, is often expensive and disruptive of a company’s management’s attention, and in any event may not lead to a successful result relative to the resources dedicated to any such litigation.
We may be unable to effectively develop an intellectual property portfolio or may fail to keep pace with advances in technology.
We have a limited operating history in the agriculture industry and there is no certainty that we will be able to effectively develop a viable portfolio of intellectual property. The success of our farm management services, which are the core of our business, depends upon our ability to create such intellectual property.
| 8 |
Even if we are able to develop, manufacture and obtain any regulatory approvals and clearances necessary for our technologies and methods, the success of such services will depend upon market acceptance. Levels of market acceptance for our services could be affected by several factors, including:
| · | the availability of alternative services from our competitors; | |
| · | the price and reliability of the our services relative to that of our competitors; and | |
| · | the timing of our market entry. |
Additionally, our intellectual property must keep pace with advances by our competitors. Failure to do so could cause our position in the industry to erode rapidly.
Confidentiality agreements with employees and others may not adequately prevent disclosure of our trade secrets and other proprietary information.
Our success depends upon the skills, knowledge and experience of our technical personnel, our consultants and advisors as well as our licensors and contractors. Because we operate in a highly competitive field, we will rely significantly on trade secrets to protect our proprietary technology and processes. However, trade secrets are difficult to protect. We enter into confidentiality and intellectual property assignment agreements with our corporate partners, employees, consultants, outside scientific collaborators, developers and other advisors. These agreements generally require that the receiving party keep confidential and not disclose to third parties confidential information developed by us during the course of the receiving party’s relationship with us. These agreements also generally provide that inventions conceived by the receiving party in the course of rendering services to us will be our exclusive property. However, these agreements may be breached and may not effectively assign intellectual property rights to us. Our trade secrets also could be independently discovered by competitors, in which case we would not be able to prevent use of such trade secrets by our competitors. The enforcement of a claim alleging that a party illegally obtained and was using our trade secrets could be difficult, expensive and time consuming and the outcome would be unpredictable. In addition, courts outside the United States may be less willing to protect trade secrets. The failure to obtain or maintain meaningful trade secret protection could adversely affect our competitive position.
We will produce products for consumption by consumers that may expose us to litigation based on consumer claims and product liability.
The stevia produced at our farms will be integrated into stevia-based products which will be consumed by the general public. Additionally, we may manufacture and sell private label stevia-based food products. Even though we intend to grow and sell products that are safe, we have potential product risk from the consuming public. We could be party to litigation based on consumer claims, product liability or otherwise that could result in significant liability for us and adversely affect our financial condition and operations.
If our services do not gain acceptance among stevia growers, we may not be able to recover the cost of our intellectual property development.
Our business model relies on the assumption that we will be able to develop methods and protocols, secure valuable plant strains and develop other intellectual property for stevia farming that will be attractive to both stevia growers and manufacturers. We spent $383,360 for this purpose as of March 31, 2013 and issued 3,000,000 shares to acquire intellectual property related to stevia and we estimate spending approximately fifteen percent of our operating expense budget to continue developing and improving this intellectual property portfolio. If we are unable to secure such intellectual property or if our methods and protocols do not gain acceptance among growers or manufacturers, our intellectual property will have limited value. A number of factors may affect the market acceptance of our products and services, including, among others, the perception by growers of the effectiveness of our intellectual property, the perception among manufacturers of the quality of stevia produced using our intellectual property, our ability to fund marketing efforts, and the effectiveness of such marketing efforts. If such products and services do not gain acceptance by growers and/or manufacturers, we may not be able to fund future operations, including the expansion of our own farming projects and development and/or acquisition of additional intellectual property, which inability would have a material adverse effect on our business, financial condition and operating results.
Any failure to adequately establish a network of growers and manufacturers will impede our growth.
We expect to be substantially dependent on manufacturers to purchase the stevia produced both at our own farms and at those of our customers. We have entered into a supply agreement with a manufacturer and two purchase agreements with growers and are in the process of establishing a network of growers to produce stevia using the methods and protocols we are developing. The relationship with this manufacturer and its perception of the stevia produced using our farm management services will determine its willingness to enter into purchase contracts with us and our customers on attractive terms. Our ability to secure such contracts will influence our attractiveness to growers who are potentially interested in partnering with us. Achieving significant growth in revenue will depend, in large part, on our success in establishing this production network. If we are unable to develop an efficient production network, it will make our growth more difficult and our business could suffer.
| 9 |
If we are unable to deliver a consistent, high quality stevia leaf at sufficient volumes, our relationship with our manufacturers may suffer and our operating results will be adversely affected.
Manufacturers will expect us to be able to consistently deliver stevia at sufficient volumes, while meeting their established quality standards. If we are unable to consistently deliver such volumes either from our own farms, or those of our grower partners, our relationship with these manufacturers could be adversely affected which could have a negative impact on our operating results.
Laws and regulations affecting the cannabis and marijuana industries are constantly changing, which could detrimentally affect our contemplated business, and we cannot predict the impact that future regulations may have on us.
Local, state and federal cannabis and marijuana laws and regulations are constantly changing and they are subject to evolving interpretations, which could require us to incur substantial costs associated with compliance or to alter one or more of our contemplated service offerings. In addition, violations of these laws, or allegations of such violations, could disrupt our contemplated business and result in a material adverse effect on our revenues, profitability, and financial condition. We cannot predict the nature of any future laws, regulations, interpretations or applications, nor can we determine what effect additional governmental regulations or administrative policies and procedures, when and if promulgated, could have on our contemplated business. Any change in law or interpretation could have a material adverse effect on our contemplated business, financial condition, and results of operations.
Marijuana remains illegal under federal law.
Marijuana remains illegal under federal law. It is a schedule-I controlled substance. Even in those jurisdictions in which the use of medical marijuana has been legalized at the state level, its prescription is a violation of federal law. Federal law criminalizing the use of marijuana trumps state laws that legalize its use for medicinal purposes, although the current President’s administration has expressed a reluctance to enforce federal law in this regard in jurisdictions where it conflicts with state law. However, a change in the federal attitude towards enforcement could occur at any time and could cripple the industry.
It is possible that our contemplated activities could be deemed to be facilitating the selling or distribution of marijuana in violation of the federal Controlled Substances Act, or to constitute aiding or abetting, or being an accessory to, a violation of that Act. Federal authorities have not focused their resources on such tangential or secondary violations of the Act, nor have they threatened to do so. However, if the federal government were to change its practices, or were to expend its resources attacking providers of services or equipment that could be usable by participants in the marijuana industry, such action could have a materially adverse effect on our contemplated business, financial condition, and results of operations.
Hemp remains illegal to grow under federal law.
Hemp remains illegal to grow in the United States under federal law due to its relation to marijuana. However, it may be legally imported and sold in the United States. In certain states, the cultivation of hemp is legal, however federal law criminalizing such cultivation trumps state laws in this regard. The current President’s administration has expressed a reluctance to enforce federal law in this regard in jurisdictions where it conflicts with state law. However, a change in the federal attitude towards enforcement could occur at any time and could cripple the industry.
It is possible that our contemplated activities could be deemed to be facilitating hemp cultivation in violation of the federal Controlled Substances Act, or to constitute aiding or abetting, or being an accessory to, a violation of that Act. Federal authorities have not focused their resources on such tangential or secondary violations of the Act, nor have they threatened to do so. However, if the federal government were to change its practices, or were to expend its resources attacking providers of services or equipment that could be usable by participants in the hemp cultivation industry, such action could have a materially adverse effect on our contemplated business, financial condition, and results of operations.
Changes in consumer preferences or negative publicity or rumors may reduce demand for our products.
Recent data suggests consumers are adopting stevia as a sweetener in many products. However, stevia is a relatively new ingredient in consumer products and many consumers are not familiar with it. Therefore, any negative reports or rumors regarding either the taste or perceived health effects of stevia, whether true or not, could have a severe impact on the demand for stevia-based products. Manufacturers may decide to rely on alternative sweeteners which have a more established history with consumers. Primarily operating at the grower level, we will have little opportunity to influence these perceptions and there can be no assurance that the increased adoption of stevia in consumer food and beverage products will continue. Additionally, new sweeteners with similar characteristics to stevia may emerge which could be cheaper to produce or be perceived to have other qualities superior to stevia. Any of these factors could adversely affect our ability to produce revenues and our business, financial condition and results of operations would suffer.
| 10 |
Failure to effectively manage growth of internal operations and business may strain our financial resources.
We intend to significantly expand the scope of our farming operations and our research and development activities in the near term. Our growth rate may place a significant strain on our financial resources for a number of reasons, including, but not limited to, the following:
| · | The need for continued development of our financial and information management systems; | |
| · | The need to manage strategic relationships and agreements with manufacturers, growers and partners; and | |
| · | Difficulties in hiring and retaining skilled management, technical and other personnel necessary to support and manage our business. |
Additionally, our strategy envisions a period of rapid growth that may impose a significant burden on our administrative and operational resources. Our ability to effectively manage growth will require us to substantially expand the capabilities of our administrative and operational resources and to attract, train, manage and retain qualified management and other personnel. Our failure to successfully manage growth could result in our sales not increasing commensurately with capital investments. Our inability to successfully manage growth could materially adversely affect our business.
Adverse weather conditions, natural disasters, crop disease, pests and other natural conditions can impose significant costs and losses on our business.
Weather-related events could significantly affect our results of operations. We do not currently maintain insurance to cover weather-related losses and if we do obtain such insurance it likely will not cover all weather-related events and, even when an event is covered, our retention or deductible may be significant. Cooler temperatures in the regions where we operate could negatively affect us, while not affecting our competitors in other regions.
Our crops, and those of our grower partners, could also be affected by drought, temperature extremes, hurricanes, windstorms and floods. In addition, such crops could be vulnerable to crop disease and to pests, which may vary in severity and effect, depending on the stage of agricultural production at the time of infection or infestation, the type of treatment applied and climatic conditions. Unfavorable growing conditions caused by these factors can reduce both crop size and crop quality. In extreme cases, entire harvests may be lost. These factors may result in lower production and, in the case of farms we own or manage, increased costs due to expenditures for additional agricultural techniques or agrichemicals, the repair of infrastructure, and the replanting of damaged or destroyed crops. We may also experience shipping interruptions, port damage and changes in shipping routes as a result of weather-related disruptions.
Competitors and industry participants may be affected differently by weather-related events based on the location of their production and supply. If adverse conditions are widespread in the industry, it may restrict supplies and lead to an increase in prices for stevia leaf, but our typical fixed-price supply contracts may prevent us from recovering these higher costs.
Our operations and products are regulated in the areas of food safety and protection of human health and the environment.
Our operations and products are subject to inspections by environmental, food safety, health and customs authorities and to numerous governmental regulations, including those relating to the use and disposal of agrichemicals, the documentation of food shipments, the traceability of food products, and labeling of our products for consumers, all of which involve compliance costs. Changes in regulations or laws may require, operational modifications or capital improvements at various locations. If violations occur, regulators can impose fines, penalties and other sanctions. The costs of these modifications and improvements and of any fines or penalties could be substantial. We can be adversely affected by actions of regulators or if consumers lose confidence in the safety and quality of stevia, even if our products are not implicated.
If we are unable to continually innovate and increase efficiencies, our ability to attract new customers may be adversely affected.
In the area of innovation, we must be able to develop new processes, plant strains, and other technologies that appeal to stevia growers. This depends, in part, on the technological and creative skills of our personnel and on our ability to protect our intellectual property rights. We may not be successful in the development, introduction, marketing and sourcing of new technologies or innovations, that satisfy customer needs, achieve market acceptance or generate satisfactory financial returns.
Global economic conditions may adversely affect our industry, business and result of operations.
Disruptions in the global credit and financial market could result in diminished liquidity and credit availability, a decline in consumer confidence, a decline in economic growth, an increased unemployment rate, and uncertainty about economic stability. These economic uncertainties can affect businesses such as ours in a number of ways, making it difficult to accurately forecast and plan our future business activities. Such conditions can lead consumers to postpone spending, which can cause manufacturers to cancel, decrease or delay orders with us. We are unable to predict the likelihood of the occurrence, duration or severity of such disruptions in the credit and financial markets and adverse global economic conditions and such economic conditions could materially and adversely affect our business and results of operations.
Our business depends substantially on the continuing efforts of our executive officers and our business may be severely disrupted if we lose their services.
Our future success depends substantially on the continued services of our executive officers, especially our President and director, Mr. George Blankenbaker. We do not maintain key man life insurance on any of our executive officers and directors. If one or more of our executive officers are unable or unwilling to continue in their present positions, we may not be able to replace them readily, if at all. Therefore, our business may be severely disrupted, and we may incur additional expenses to recruit and retain new officers. In addition, if any of our executives joins a competitor or forms a competing company, we may lose some of our customers.
| 11 |
Our engagement of Growers Synergy Pte Ltd. may represent a potential conflict of interest.
We have engaged Growers Synergy Pte Ltd, a regional farm management services provider, to provide farm management operations and back-office and regional logistical support for our Vietnam and Indonesia operations for a period of two years. During the fiscal year ended March 31, 2012, Growers Synergy received $180,000 for consulting services rendered to the Company and during the fiscal year ended March 31, 2013, Growers Synergy received $240,000 for consulting services rendered to the Company. George Blankenbaker, our president, director and stockholder is the managing director of Growers Synergy. Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and Mr. Blankenbaker controls a 49% interest in Growers Fresh. As a result, there is a potential conflict of interest on Mr. Blankenbaker’s role in the Company and Growers Synergy and such potential conflict could materially affect the terms of any engagement entered into by the Company and Growers Synergy. Such terms, if not negotiated at arms length may not be in the best interest of the Company and our stockholders.
Litigation may adversely affect our business, financial condition and results of operations.
From time to time in the normal course of our business operations, we may become subject to litigation that may result in liability material to our financial statements as a whole or may negatively affect our operating results if changes to our business operation are required. The cost to defend such litigation may be significant and may require a diversion of our resources. There also may be adverse publicity associated with litigation that could negatively affect customer perception of our business, regardless of whether the allegations are valid or whether we are ultimately found liable. As a result, litigation may adversely affect our business, financial condition and results of operations.
We may be required to incur significant costs and require significant management resources to evaluate our internal control over financial reporting as required under Section 404 of the Sarbanes-Oxley Act, and any failure to comply or any adverse result from such evaluation may have an adverse effect on our stock price.
As a smaller reporting company as defined in Rule 12b-2 under the Securities Exchange Act of 1934, as amended, we are required to evaluate our internal control over financial reporting under Section 404 of the Sarbanes-Oxley Act of 2002 (“Section 404”). Section 404 requires us to include an internal control report with our Annual Report on Form 10-K. This report must include management’s assessment of the effectiveness of our internal control over financial reporting as of the end of the fiscal year. This report must also include disclosure of any material weaknesses in internal control over financial reporting that we have identified. Failure to comply, or any adverse results from such evaluation could result in a loss of investor confidence in our financial reports and have an adverse effect on the trading price of our equity securities. As of December 31, 2013, the management of the Company assessed the effectiveness of the Company’s internal control over financial reporting based on the criteria for effective internal control over financial reporting established inInternal Control - Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”) and SEC guidance on conducting such assessments. Management concluded, as of the quarter ended December 31, 2013, that its internal controls and procedures were not effective to detect the inappropriate application of U.S. GAAP rules. Management realized there were deficiencies in the design or operation of our internal control that adversely affected our internal controls which management considers to be material weaknesses including those described below:
| · | We have not achieved the optimal level of segregation of duties relative to key financial reporting functions. | |
| · | We do not have an audit committee or an independent audit committee financial expert. While not being legally obligated to have an audit committee or independent audit committee financial expert, it is the management’s view that to have an audit committee, comprised of independent board members, and an independent audit committee financial expert is an important entity-level control over our financial statements. |
Achieving continued compliance with Section 404 may require us to incur significant costs and expend significant time and management resources. No assurance can be given that we will be able to fully comply with Section 404 or that we and our independent registered public accounting firm would be able to conclude that our internal control over financial reporting is effective at fiscal year end. As a result, investors could lose confidence in our reported financial information, which could have an adverse effect on the trading price of our securities, as well as subject us to civil or criminal investigations and penalties. In addition, our independent registered public accounting firm may not agree with our management’s assessment or conclude that our internal control over financial reporting is operating effectively.
RISKS RELATED TO DOING BUSINESS IN VIETNAM AND OTHER DEVELOPING COUNTRIES
Our international operations will be subject to the laws of the jurisdictions in which we operate.
A significant portion of our initial business operations will occur in Vietnam. We will be generally subject to laws and regulations applicable to foreign investment in Vietnam. The Vietnamese legal system is based, at least in part, on written statutes. However, since these laws and regulations are relatively new and the Vietnamese legal system continues to rapidly evolve, the interpretations of many laws, regulations and rules are not always uniform and enforcement of these laws, regulations and rules involves uncertainties.
| 12 |
In April 2012, we announced plans to begin field tests in Indonesia. Similar to Vietnam, the modern Indonesia legal system was formed relatively recently and is continuing to evolve. As we continue our expansion into Indonesia and other developing countries, we will face similar risks and uncertainties regarding the legal system as we currently face in Vietnam.
We cannot predict the effect of future developments in the legal systems of developing countries, including the promulgation of new laws, changes to existing laws or the interpretation or enforcement thereof, the preemption of local regulations by national laws, or the overturn of local government’s decisions by the superior government. These uncertainties may limit legal protections available to us.
Our international operations involve the use of foreign currencies, which subjects us to exchange rate fluctuations and other currency risks.
The revenues and expenses of our international operations are generally denominated in local currencies, which subjects us to exchange rate fluctuations between such local currencies and the U.S. dollar. These exchange rate fluctuations will subject us to currency translation risk with respect to the reported results of our international operations, as well as to other risks sometimes associated with international operations. In the future, we could experience fluctuations in financial results from our operations outside of the United States, and there can be no assurance we will be able, contractually or otherwise, to reduce the currency risks associated with our international operations.
We may be adversely affected by economic and political conditions in the countries where we operate.
We operate in Vietnam and other countries throughout the world. Economic and political changes in these countries, such as inflation rates, recession, foreign ownership restrictions, restrictions on transfer of funds into or out of a country and similar factors may adversely affect results of operations.
While it is our understanding that the economy in Vietnam has grown significantly in the past 20 years, the growth has been uneven, both geographically and among various economic sectors. The government of Vietnam has implemented various measures to encourage or control economic growth and guide the allocation of resources. Some of these measures benefit the overall Vietnamese economy, but may also have a negative effect on us. For example, our financial condition and results of operations may be adversely affected by government control over capital investments or changes in tax regulations that are applicable to us.
The Vietnamese economy has been transitioning from a planned economy to a more market-oriented economy. Although in recent years the Vietnamese government has implemented measures emphasizing the utilization of market forces for economic reform, the reduction of state ownership of productive assets and the establishment of sound corporate governance in business enterprises, a substantial portion of the productive assets in Vietnam are still owned by the Vietnamese government. The continued control of these assets and other aspects of the national economy by Vietnam government could materially and adversely affect our business. The Vietnamese government also exercises significant control over Vietnamese economic growth through the allocation of resources, controlling payment of foreign currency-denominated obligations, setting monetary policy and providing preferential treatment to particular industries or companies. Efforts by the Vietnamese government to slow the pace of growth of the Vietnamese economy could negatively affect our business.
Our insurance coverage may be inadequate to cover all significant risk exposures.
We will be exposed to liabilities that are unique to the products we provide. While we intend to maintain insurance for certain risks, the amount of our insurance coverage may not be adequate to cover all claims or liabilities, and we may be forced to bear substantial costs resulting from risks and uncertainties of our business. It is also not possible to obtain insurance to protect against all operational risks and liabilities. The failure to obtain adequate insurance coverage on terms favorable to us, or at all, could have a material adverse effect on our business, financial condition and results of operations. In addition, because the insurance industry in Vietnam and other developing countries are still in their early stages of development, business interruption insurance available in such countries relating to our intended services and products offers limited coverage compared to that offered in many other developed countries. We do not have any business interruption insurance. Any business disruption or natural disaster could result in substantial costs and diversion of resources.
It will be extremely difficult to acquire jurisdiction and enforce liabilities against our officers, directors and assets outside the United States.
Substantially all of our assets are currently located outside of the United States and a significant number of our officers and directors may reside outside of the United States as well. As a result, it may not be possible for United States investors to enforce their legal rights, to effect service of process upon our directors or officers or to enforce judgments of United States courts predicated upon civil liabilities and criminal penalties of our directors and officers under Federal securities laws. Moreover, we have been advised that Vietnam in particular does not have treaties providing for the reciprocal recognition and enforcement of judgments of courts with the United States. Further, it is unclear if extradition treaties now in effect between the United States and Vietnam would permit effective enforcement of criminal penalties of the Federal securities laws.
| 13 |
RISKS RELATED TO AN INVESTMENT IN OUR SECURITIES
The relative lack of public company experience of our management team may put us at a competitive disadvantage.
Our management team lacks public company experience and is generally unfamiliar with the requirements of the United States securities laws and U.S. Generally Accepted Accounting Principles, which could impair our ability to comply with legal and regulatory requirements such as those imposed by Sarbanes-Oxley Act of 2002. The individuals who now constitute our senior management team have never had responsibility for managing a publicly traded company. Such responsibilities include complying with federal securities laws and making required disclosures on a timely basis. Our senior management may not be able to implement programs and policies in an effective and timely manner that adequately responds to such increased legal, regulatory compliance and reporting requirements. Our failure to comply with all applicable requirements could lead to the imposition of fines and penalties and distract our management from attending to the growth of our business.
Our stock is categorized as a penny stock. Trading of our stock may be restricted by the SEC’s penny stock regulations which may limit a stockholder’s ability to buy and sell our stock.
Our stock is categorized as a “penny stock”. The SEC has adopted Rule 15g-9 which generally defines “penny stock” to be any equity security that has a market price (as defined) less than $4.00 per share or an exercise price of less than $5.00 per share, subject to certain exceptions. Our securities are covered by the penny stock rules, which impose additional sales practice requirements on broker-dealers who sell to persons other than established customers and accredited investors. The penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document in a form prepared by the SEC which provides information about penny stocks and the nature and level of risks in the penny stock market. The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction and monthly account statements showing the market value of each penny stock held in the customer’s account. The bid and offer quotations, and the broker-dealer and salesperson compensation information, must be given to the customer orally or in writing prior to effecting the transaction and must be given to the customer in writing before or with the customer’s confirmation. In addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from these rules, the broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written agreement to the transaction. These disclosure requirements may have the effect of reducing the level of trading activity in the secondary market for the stock that is subject to these penny stock rules. Consequently, these penny stock rules may affect the ability of broker-dealers to trade our securities. We believe that the penny stock rules discourage investor interest in and limit the marketability of our common stock.
FINRA sales practice requirements may also limit a stockholder’s ability to buy and sell our stock.
In addition to the “penny stock” rules described above, the Financial Industry Regulatory Authority (“FINRA”) has adopted rules that require that in recommending an investment to a customer, a broker-dealer must have reasonable grounds for believing that the investment is suitable for that customer. Prior to recommending speculative low priced securities to their non-institutional customers, broker-dealers must make reasonable efforts to obtain information about the customer’s financial status, tax status, investment objectives and other information. Under interpretations of these rules, FINRA believes that there is a high probability that speculative low priced securities will not be suitable for at least some customers. The FINRA requirements make it more difficult for broker-dealers to recommend that their customers buy our common stock, which may limit your ability to buy and sell our stock and have an adverse effect on the market for our shares.
We expect to experience volatility in our stock price, which could negatively affect stockholders’ investments.
Although our common stock is quoted on the OTCQB under the symbol “STEV”, there is a limited public market for our common stock. No assurance can be given that an active market will develop or that a stockholder will ever be able to liquidate its shares of common stock without considerable delay, if at all. Many brokerage firms may not be willing to effect transactions in the securities. Even if a purchaser finds a broker willing to effect a transaction in these securities, the combination of brokerage commissions, state transfer taxes, if any, and any other selling costs may exceed the selling price. Furthermore, our stock price may be impacted by factors that are unrelated or disproportionate to our operating performance. These market fluctuations, as well as general economic, political and market conditions, such as recessions, interest rates or international currency fluctuations may adversely affect the market price and liquidity of our common stock.
In the past, securities class action litigation has often been brought against a company following periods of volatility in the market price of its securities. Due to the volatility of our common stock price, we may be the target of securities litigation in the future. Securities litigation could result in substantial costs and divert management’s attention and resources.
Stockholders should also be aware that, according to SEC Release No. 34-29093, the market for “penny stock”, such as our common stock, has suffered in recent years from patterns of fraud and abuse. Such patterns include (1) control of the market for the security by one or a few broker-dealers that are often related to the promoter or issuer; (2) manipulation of prices through prearranged matching of purchases and sales and false and misleading press releases; (3) boiler room practices involving high-pressure sales tactics and unrealistic price projections by inexperienced sales persons; (4) excessive and undisclosed bid-ask differential and markups by selling broker-dealers; and (5) the wholesale dumping of the same securities by promoters and broker-dealers after prices have been manipulated to a desired level, along with the resulting inevitable collapse of those prices and with consequent investor losses. Our management is aware of the abuses that have occurred historically in the penny stock market. Although we do not expect to be in a position to dictate the behavior of the market or of broker-dealers who participate in the market, management will strive within the confines of practical limitations to prevent the described patterns from being established with respect to our securities. The occurrence of these patterns or practices could increase the future volatility of our share price.
| 14 |
To date, we have not paid any cash dividends and no cash dividends will be paid in the foreseeable future.
We do not anticipate paying cash dividends on our common stock in the foreseeable future and we may not have sufficient funds legally available to pay dividends. Even if the funds are legally available for distribution, we may nevertheless decide not to pay any dividends. We presently intend to retain all earnings for our operations.to pay dividends. Even if the funds are legally available for distribution, we may nevertheless decide not to pay any dividends. We presently intend to retain all earnings for our operations.
The elimination of monetary liability against our directors, officers and employees under Nevada law and the existence of indemnification rights to our directors, officers and employees may result in substantial expenditures by our company and may discourage lawsuits against our directors, officers and employees.
Our Articles of Incorporation contain a provision permitting us to eliminate the personal liability of our directors to our company and stockholders for damages for breach of fiduciary duty as a director or officer to the extent provided by Nevada law. We may also have contractual indemnification obligations under our employment agreements with our officers. The foregoing indemnification obligations could result in the Company incurring substantial expenditures to cover the cost of settlement or damage awards against directors and officers, which we may be unable to recoup. These provisions and resultant costs may also discourage our company from bringing a lawsuit against directors and officers for breaches of their fiduciary duties, and may similarly discourage the filing of derivative litigation by our stockholders against our directors and officers even though such actions, if successful, might otherwise benefit our company and stockholders.
If the Company’s outstanding warrants and convertible notes are exercised and/or converted it will result in the dilution of our existing stockholders.
The outstanding warrants and convertible notes being registered hereunder are exercisable for an aggregate of 47,898,931 shares of our common stock. Although the Company has a call right or repayment right with respect to certain of the notes and warrants, the Company may not be financially capable of exercising such call right or may otherwise choose not to do so, and therefore the Company may not control if and when the notes and warrants are exercised and/or converted. The exercise of the notes and warrants would result in dilution to our existing stockholders and could contribute to a reduction in the market price of the outstanding shares of our common stock.
USE OF PROCEEDS
The “Selling Security Holders” set forth in “The Selling Security Holders Table” below may sell all of the common stock underlying the Warrants offered by this Prospectus from time-to-time. We will not receive any proceeds from the sale of those shares of common stock. We may, however, receive gross proceeds of up to $1,083,103.46 upon the cash exercise of the Warrants. Any such proceeds we receive will be used for working capital and general corporate matters.
DETERMINATION OF OFFERING PRICE
There currently is a limited public market for our common stock. Selling Security Holders will determine at what price they may sell the offered shares, and such sales may be made at prevailing market prices or at privately negotiated prices. See “Plan of Distribution” below for more information.
SELLING SECURITY HOLDERS
Nomis Bay Note Shares
This offering, with respect to the Nomis Bay Note Shares, relates to the resale of shares of common stock of the Company underlying a Senior Convertible Note, issued March 3, 2014 (the “Nomis Bay Note”). On March 3, 2014, pursuant to a Securities Purchase Agreement (the “Purchase Agreement”) with Nomis Bay Ltd., a Bermuda company (“Nomis Bay”), we issued the Nomis Bay Note with an initial principal amount of $500,000 for a purchase price of $340,000. The Nomis Bay Note matures on December 27, 2014 (subject to extension as provided in the Nomis Bay Note) and, in addition to the 32% original issue discount, accrues interest at the rate of 8% per annum. The Purchase Agreement also provides that, upon the terms and subject to the conditions set forth therein, the Company may require Nomis Bay to purchase from the Company on or prior to the 10th trading day after the effective date of the registration statement registering the shares issuable upon conversion of the Initial Convertible Note, an additional senior convertible note with an initial principal amount of $600,000 (the “Additional Convertible Note” and together with the Nomis Bay Note, the “Convertible Notes”) for a purchase price of $600,000. If issued, the Additional Convertible Note will mature on the date that is the 10-month anniversary of the date of issuance of the Additional Convertible Note (subject to extension as provided in the Initial Convertible Note) and will accrue interest at the rate of 8% per annum. The Nomis Bay Note is convertible at any time, in whole or in part, at Nomis Bay’s option into shares of the Company’s common stock, par value $0.001 per share (the “Common Stock”), at a conversion price equal to the lesser of (i) the product of (x) the arithmetic average of the lowest three (3) volume weighted average prices of the Common Stock during the 10 consecutive trading days ending and including the trading day immediately preceding the applicable conversion date and (y) 40% (the “Variable Conversion Price”), and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). If issued, the Additional Convertible Note will be convertible at any time, in whole or in part, at Nomis Bay’s option into shares of Common Stock at a conversion price that will be equal to the lesser of (i) the Variable Conversion Price and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). At no time will Nomis Bay be entitled to convert any portion of the Convertible Notes to the extent that after such conversion, Nomis Bay (together with its affiliates) would beneficially own more than 4.99% of the outstanding shares of Common Stock as of such date. The shares of common stock underlying the Nomis Bay Note were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
| 15 |
Anson Reset Shares
This offering, with respect to the Anson Reset Shares, relates to the resale of shares of common stock of the Company underlying three warrants in the amounts of 1,877,333, 1,066,666 and 2,346,666, with exercise prices of $0.20, $0.25 and $0.25 per share (the “Anson Warrants”). Pursuant to the terms of a warrant exercise reset offer letter between the Company and the investor, the Anson Warrants were issued in consideration for the investor’s agreement to immediately cash exercise an existing warrant to purchase 853,333 shares of common stock of the Company at an exercise price of $0.20 per share for aggregate consideration to the Company of $170,666 originally issued pursuant to the terms of the Securities Purchase Agreement, dated August 1, 2012 (the “Purchase Agreement”). Each Anson Warrant has a five year term and was issued on May 3, 2013. Each Anson Warrant provides for adjustment of the exercise price and share amount in the event of certain dilutive issuances by the Company. On February 20, 2014, the Company issued a notice to the holder that the aggregate number of Anson Warrants had been adjusted to 23,026,318 and the exercise price of each had been adjusted to $0.053365 as a result of certain other offerings of the Company. 5,290,665 of the shares underlying the Anson Warrants were previously registered by the Company pursuant to the Registration Statement on Form S-1/A filed December 30, 2013. The warrants and the shares of common stock underlying the warrants were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
Cranshire Warrant Shares
This offering, with respect to the Cranshire Warrant Shares, relates to the resale of the shares of common stock underlying a warrant in the amount of 2,560,486 with an exercise price of $0.053365. The warrant has a five (5) year term and was issued pursuant to the terms of the Purchase Agreement. The warrant was originally issued in the amount of 213,334 shares at an exercise price of $0.6405. Pursuant to the anti-dilution adjustment provision contained therein, on February 20, 2014, the Company issued a notice to the holder that the total share amount had been increased to 2,560,486 and the exercise price had been reduced to $0.053365 as a result of certain other offerings of the Company. 683,202 of the shares underlying the Cranshire Warrants were previously registered by the Company pursuant to the Registration Statement on Form S-1/A filed December 30, 2013. Additionally, on February 20, 2014 in consideration for the investor’s agreement to immediately cash exercise a portion of the Cranshire Warrants for aggregate consideration to the Company of $36,459, the Company agreed to issue the investor an additional warrant to purchase 683,202 shares of common stock. The warrants and the shares of common stock underlying the warrants were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
JMJ Note Shares
This offering, with respect to the JMJ Note Shares, relates to the resale of 3,000,000 shares of common stock of the Company underlying a $400,000 Promissory Note issued July 10, 2013. The JMJ Note is subject to a one-time interest charge equal to 12% of the principal sum and is due and payable July 10, 2014. The JMJ Note provides that it may be converted into common stock of the Company at any time at the election of the holder, at a conversion price equal to the lesser of $0.26 or 65% of the lowest trade price in the 25 trading days previous to the conversion. The JMJ Note and the shares of common stock underlying the JMJ Note were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
The Selling Security Holders Table
The following table sets forth the names of the Selling Security Holders, the number of shares of common stock beneficially owned by each Selling Security Holder as of the date hereof and the number of shares of common stock being offered by each Selling Security Holder. The shares being offered hereby are being registered to permit public secondary trading, and the Selling Security Holders may offer all or part of the shares for resale from time to time. However, the Selling Security Holders are under no obligation to sell all or any portion of such shares nor are the Selling Security Holders obligated to sell any shares immediately upon effectiveness of this prospectus. All information with respect to share ownership has been furnished by the Selling Security Holders. The “Amount Beneficially Owned After Offering” column assumes the sale of all shares offered.
| Shares | Amount | Percent | ||||||||||||||
| Beneficially | Beneficially | Beneficially | ||||||||||||||
| Owned Prior To | Shares to | Owned After | Owned | |||||||||||||
| Name | Offering(1) | be Offered | Offering(1)(5) | After Offering(6) | ||||||||||||
| Nomis Bay Ltd.(2) | 6,361,323 | 24,602,792 | 0 | 0 | % | |||||||||||
| Anson Investments Master Fund LP(3) | 17,735,653 | 17,735,653 | 0 | 0 | % | |||||||||||
| Cranshire Capital Master Fund, Ltd.(4) | 2,560,486 | 2,560,486 | 0 | 0 | % | |||||||||||
| JMJ Financial | 0 | 3,000,000 | 0 | 0 | % | |||||||||||
| 16 |
| (1) | Beneficial ownership has been determined in accordance with Rule 13d-3 under the Exchange Act. Pursuant to the rules of the SEC, shares of common stock which an individual or group has a right to acquire within 60 days pursuant to the exercise of options or warrants are deemed to be outstanding for the purpose of computing the percentage ownership of such individual or group, but are not deemed to be beneficially owned and outstanding for the purpose of computing the percentage ownership of any other person shown in the table. |
| (2) | Includes 6,361,323 shares of common stock issuable to Nomis Bay Ltd. upon the conversion of the principal amount of the registrant’s Senior Convertible Note issued March 3, 2014, based upon a current conversion price of $0.0786. EOM Management Ltd. (“EOM”) is the investment manager of Nomis Bay Ltd. and has voting control and investment discretion over securities held by Nomis Bay Ltd. Chaja Carlebach (“Ms. Carlebach”) owns all of the outstanding equity of EOM and has voting control over EOM. As a result, each of Ms. Carlebach and EOM may be deemed to have beneficial ownership (as determined under Section 13(d) of the Securities Exchange Act of 1934, as amended) of the securities held by Nomis Bay Ltd. We have been advised that neither EOM nor Nomis Bay Ltd. is a member of the Financial Industry Regulatory Authority (“FINRA”) or an independent broker-dealer, and that neither EOM, Nomis Bay Ltd. or any of their respective affiliates is an affiliate or an associated person of any FINRA member or independent broker-dealer. |
| (3) | Moez Kassam has voting and dispositive control over the shares beneficially owned by Anson Investments Master Fund LP. |
| (4) | Cranshire Capital Advisors, LLC (“CCA”) is the investment manager of Cranshire Capital Master Fund, Ltd. (“Cranshire Master Fund”) and has voting control and investment discretion over securities held by Cranshire Master Fund. Mitchell P. Kopin (“Mr. Kopin”), the president, the sole member and the sole member of the Board of Managers of CCA, has voting control over CCA. As a result, each of Mr. Kopin and CCA may be deemed to have beneficial ownership (as determined under Section 13(d) of the Securities Exchange Act of 1934, as amended) of the securities held by Cranshire Master Fund. |
| (5) | Includes the shares of common stock issuable upon exercise of the Warrants. |
| (6) | Applicable percentage ownership is based on 145,972,713 shares of our common stock outstanding as of March 26, 2014. |
All expenses incurred with respect to the registration of the common stock will be borne by us, but we will not be obligated to pay any underwriting fees, discounts, commission or other expenses incurred by the Selling Security Holders in connection with the sale of the Shares.
Neither the Selling Security Holders nor any of their associates or affiliates has held any position, office, or other material relationship with us in the past three years.
PLAN OF DISTRIBUTION
This prospectus relates to the resale of 47,898,931 shares of common stock, by the Selling Security Holders listed under “Selling Security Holders” on page 15, issuable upon the exercise of the outstanding convertible notes and warrants.
The Selling Security Holders and any of their respective pledges, donees, assignees and other successors-in-interest may, from time to time, sell any or all of their shares of our common stock on any stock exchange, market or trading facility on which the shares are traded or in private transactions. The Selling Security Holders may use any one or more of the following methods when selling shares:
| · | ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| · | block trades in which the broker-dealer will attempt to sell the shares as agent, but may position and resell a portion of the block as principal to facilitate the transaction; |
| · | purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
| · | an exchange distribution in accordance with the rules of the applicable exchange; |
| · | privately negotiated transactions; |
| · | broker-dealers may agree with a Selling Security Holder to sell a specified number of such shares at a stipulated price per share; |
| · | through the writing of options on the shares; |
| · | a combination of any such methods of sale; and |
any other method permitted pursuant to applicable law.
| 17 |
The Selling Security Holders or their respective pledgees, donees, transferees or other successors in interest, may also sell the shares directly to market makers acting as principals and/or broker-dealers acting as agents for themselves or their customers. Such broker-dealers may receive compensation in the form of discounts, concessions or commissions from the Selling Security Holders and/or the purchasers of shares for whom such broker-dealers may act as agents or to whom they sell as principal or both, which compensation as to a particular broker-dealer might be in excess of customary commissions. Market makers and block purchasers purchasing the shares will do so for their own account and at their own risk. It is possible that the Selling Security Holders will attempt to sell shares of common stock in block transactions to market makers or other purchasers at a price per share which may be below the then market price. The Selling Security Holders cannot assure that all or any of the shares offered in this prospectus will be issued to, or sold by, the Selling Security Holders. In addition, the Selling Security Holders and any brokers, dealers or agents, upon effecting the sale of any of the shares offered in this prospectus may be deemed “underwriters” as that term is defined under the Securities Act or the Exchange Act, or the rules and regulations under such acts. In such event, any commissions received by such broker-dealers or agents and any profit on the resale of the shares purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act.
Discounts, concessions, commissions and similar selling expenses, if any, attributable to the sale of shares will be borne by a Selling Security Holder. The Selling Security Holders may agree to indemnify any agent, dealer or broker-dealer that participates in transactions involving sales of the shares if liabilities are imposed on that person under the Securities Act.
The Selling Security Holders may from time to time pledge or grant a security interest in some or all of the shares of common stock owned by them, and, if they default in the performance of their secured obligations, the pledgees or secured parties may offer and sell the shares of common stock from time to time under this prospectus after we have filed an amendment to this prospectus under Rule 424(b)(3) or any other applicable provision of the Securities Act amending the list of Selling Security Holders to include the pledgee, transferee or other successors in interest as Selling Security Holder under this prospectus.
The Selling Security Holders also may transfer the shares of common stock in other circumstances, in which case the transferees, pledgees or other successors in interest will be the selling beneficial owners for purposes of this prospectus and may sell the shares of common stock from time to time under this prospectus after we have filed an amendment to this prospectus under Rule 424(b)(3) or other applicable provision of the Securities Act amending the list of Selling Security Holders to include the pledgee, transferee or other successors in interest as a Selling Security Holder under this prospectus.
The Selling Security Holders acquired or will acquire the securities offered hereby in the ordinary course of business and have advised us that they have not entered into any agreements, understandings or arrangements with any underwriters or broker-dealers regarding the sale of their shares of common stock, nor is there an underwriter or coordinating broker acting in connection with a proposed sale of shares of common stock by any Selling Security Holder. We will file a supplement to this prospectus if a Selling Security Holder enters into a material arrangement with a broker-dealer for sale of common stock being registered. If the Selling Security Holders use this prospectus for any sale of the shares of common stock, it will be subject to the prospectus delivery requirements of the Securities Act.
Pursuant to a requirement by the Financial Industry Regulatory Authority, or FINRA, the maximum commission or discount to be received by any FINRA member or independent broker/dealer may not be greater than eight percent (8%) of the gross proceeds received by us for the sale of any securities being registered pursuant to SEC Rule 415 under the Securities Act.
The anti-manipulation rules of Regulation M under the Exchange Act, may apply to sales of our common stock and activities of the Selling Security Holders. The Selling Security Holders will act independently of us in making decisions with respect to the timing, manner and size of each sale.
We will pay all expenses incident to the registration, offering and sale of the shares of our common stock to the public hereunder other than commissions, fees and discounts of underwriters, brokers, dealers and agents. We estimate that the expenses of the offering to be borne by us will be approximately $16,145.53. We will not receive any proceeds from the resale of any of the shares of our common stock in this Offering, other than $1,083,103.46 of potential proceeds upon the exercise of the Warrants for cash.
DESCRIPTION OF SECURITIES TO BE REGISTERED
General
The following summary includes a description of material provisions of our capital stock.
Authorized and Outstanding Securities
The Company is authorized to issue 250,000,000 shares of common stock, par value $0.001 per share. As of March 26, 2014, there were issued and outstanding 145,972,713 shares of our common stock.
Common Stock
The holders of our common stock are entitled to one vote for each share on all matters to be voted on by the stockholders. Holders of common stock do not have cumulative voting rights. Holders of common stock are entitled to share ratably in dividends, if any, as may be declared from time to time by the board of directors in its discretion from funds legally available therefore. In the event of a liquidation, dissolution or winding up of the Company, the holders of common stock are entitled to share pro rata all assets remaining after payment in full of all liabilities. Holders of common stock have no preemptive rights to purchase the Company’s common stock. There are no conversion or redemption rights or sinking fund provisions with respect to the common stock.
| 18 |
Dividends
Dividends, if any, will be contingent upon our revenues and earnings, if any, capital requirements and financial conditions. The payment of dividends, if any, will be within the discretion of our board of directors. We intend to retain earnings, if any, for use in its business operations and accordingly, the board of directors does not anticipate declaring any dividends in the foreseeable future.
Convertible Notes and Warrants
Nomis Bay Note Shares
This offering, with respect to the Nomis Bay Note Shares, relates to the resale of shares of common stock of the Company underlying a Senior Convertible Note, issued March 3, 2014 (the “Nomis Bay Note”). On March 3, 2014, pursuant to a Securities Purchase Agreement (the “Purchase Agreement”) with Nomis Bay Ltd., a Bermuda company (“Nomis Bay”), we issued the Nomis Bay Note with an initial principal amount of $500,000 for a purchase price of $340,000. The Nomis Bay Note matures on December 27, 2014 (subject to extension as provided in the Nomis Bay Note) and, in addition to the 32% original issue discount, accrues interest at the rate of 8% per annum. The Purchase Agreement also provides that, upon the terms and subject to the conditions set forth therein, the Company may require Nomis Bay to purchase from the Company on or prior to the 10th trading day after the effective date of the registration statement registering the shares issuable upon conversion of the Initial Convertible Note, an additional senior convertible note with an initial principal amount of $600,000 (the “Additional Convertible Note” and together with the Nomis Bay Note, the “Convertible Notes”) for a purchase price of $600,000. If issued, the Additional Convertible Note will mature on the date that is the 10-month anniversary of the date of issuance of the Additional Convertible Note (subject to extension as provided in the Initial Convertible Note) and will accrue interest at the rate of 8% per annum. The Nomis Bay Note is convertible at any time, in whole or in part, at Nomis Bay’s option into shares of the Company’s common stock, par value $0.001 per share (the “Common Stock”), at a conversion price equal to the lesser of (i) the product of (x) the arithmetic average of the lowest three (3) volume weighted average prices of the Common Stock during the 10 consecutive trading days ending and including the trading day immediately preceding the applicable conversion date and (y) 40% (the “Variable Conversion Price”), and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). If issued, the Additional Convertible Note will be convertible at any time, in whole or in part, at Nomis Bay’s option into shares of Common Stock at a conversion price that will be equal to the lesser of (i) the Variable Conversion Price and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). At no time will Nomis Bay be entitled to convert any portion of the Convertible Notes to the extent that after such conversion, Nomis Bay (together with its affiliates) would beneficially own more than 4.99% of the outstanding shares of Common Stock as of such date. The shares of common stock underlying the Nomis Bay Note were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
Anson Reset Shares
This offering, with respect to the Anson Reset Shares, relates to the resale of shares of common stock of the Company underlying three warrants in the amounts of 1,877,333, 1,066,666 and 2,346,666, with exercise prices of $0.20, $0.25 and $0.25 per share (the “Anson Warrants”). Pursuant to the terms of a warrant exercise reset offer letter between the Company and the investor, the Anson Warrants were issued in consideration for the investor’s agreement to immediately cash exercise an existing warrant to purchase 853,333 shares of common stock of the Company at an exercise price of $0.20 per share for aggregate consideration to the Company of $170,666 originally issued pursuant to the terms of the Securities Purchase Agreement, dated August 1, 2012 (the “Purchase Agreement”). Each Anson Warrant has a five year term and was issued on May 3, 2013. Each Anson Warrant provides for adjustment of the exercise price and share amount in the event of certain dilutive issuances by the Company. On February 20, 2014, the Company issued a notice to the holder that the aggregate number of Anson Warrants had been adjusted to 23,026,318 and the exercise price of each had been adjusted to $0.053365 as a result of certain other offerings of the Company. 5,290,665 of the shares underlying the Anson Warrants were previously registered by the Company pursuant to the Registration Statement on Form S-1/A filed December 30, 2013. The warrants and the shares of common stock underlying the warrants were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
Cranshire Warrant Shares
This offering, with respect to the Cranshire Warrant Shares, relates to the resale of the shares of common stock underlying a warrant in the amount of 2,560,486 with an exercise price of $0.053365. The warrant has a five (5) year term and was issued pursuant to the terms of the Purchase Agreement. The warrant was originally issued in the amount of 213,334 shares at an exercise price of $0.6405. Pursuant to the anti-dilution adjustment provision contained therein, on February 20, 2014, the Company issued a notice to the holder that the total share amount had been increased to 2,560,486 and the exercise price had been reduced to $0.053365 as a result of certain other offerings of the Company. 683,202 of the shares underlying the Cranshire Warrants were previously registered by the Company pursuant to the Registration Statement on Form S-1/A filed December 30, 2013. Additionally, on February 20, 2014 in consideration for the investor’s agreement to immediately cash exercise a portion of the Cranshire Warrants for aggregate consideration to the Company of $36,459, the Company agreed to issue the investor an additional warrant to purchase 683,202 shares of common stock. The warrants and the shares of common stock underlying the warrants were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
| 19 |
JMJ Note Shares
This offering, with respect to the JMJ Note Shares, relates to the resale of 3,000,000 shares of common stock of the Company underlying a $400,000 Promissory Note issued July 10, 2013. The JMJ Note is subject to a one-time interest charge equal to 12% of the principal sum and is due and payable July 10, 2014. The JMJ Note provides that it may be converted into common stock of the Company at any time at the election of the holder, at a conversion price equal to the lesser of $0.26 or 65% of the lowest trade price in the 25 trading days previous to the conversion. The JMJ Note and the shares of common stock underlying the JMJ Note were issued in reliance upon an exemption from the registration requirements of the Securities Act and/or Rule 506 of Regulation D promulgated thereunder.
Registration Rights
Nomis Bay Registration Rights Agreement
In connection with the execution of the Purchase Agreement, the Company and Nomis Bay also entered into a registration rights agreement (the “Registration Rights Agreement”) dated March 3, 2014 (the “Closing Date”). Pursuant to the Registration Rights Agreement, the Company has agreed to file this registration statement (“Registration Statement”) with the SEC to register the resale of 24,602,792 shares of Common Stock into which the Convertible Notes may be converted, on or prior to the 45th calendar day after the Closing Date and have it declared effective at the earlier of (i) the 120th calendar day after the Closing Date and (ii) the fifth business day after the date the Company is notified by the SEC that such Registration Statement will not be reviewed or will not be subject to further review.
If at any time all of the shares of Common Stock underlying the Convertible Notes are not covered by the initial Registration Statement, the Company has agreed to file with the SEC one or more additional Registration Statements so as to cover all of the shares of Common Stock underlying the Convertible Notes not covered by such initial Registration Statement, in each case, as soon as practicable, but in no event later than the applicable filing deadline for such additional Registration Statements as provided in the Registration Rights Agreement.
Piggyback Registration Rights
The JMJ Note contains a provision whereby the Company agreed to include on its next registration statement all shares issuable upon conversion of such debentures and notes. Such conversion shares are included on this registration statement. All fees and expenses incident to the performance of or compliance with such registration rights shall be borne by the Company.
INTERESTS OF NAMED EXPERTS AND COUNSEL
No expert or counsel named in this prospectus as having prepared or certified any part of this prospectus or having given an opinion upon the validity of the securities being registered or upon other legal matters in connection with the registration or offering of the common stock was employed on a contingency basis, or had, or is to receive, in connection with the offering, a substantial interest, direct or indirect, in the registrant or any of its parents or subsidiaries. Nor was any such person connected with the registrant or any of its parents or subsidiaries as a promoter, managing or principal underwriter, voting trustee, director, officer, or employee.
The consolidated financial statements included in this prospectus and in the registration statement have been audited by Li and Company, PC, and are included in reliance upon such report given upon the authority of said firm as experts in auditing and accounting.
The validity of the issuance of the common stock hereby will be passed upon for us by Greenberg Traurig, LLP.
INFORMATION WITH RESPECT TO THE REGISTRANT
Background
We are a farm management company primarily focused on agronomics from plant breeding to good agricultural practices and the development of stevia derived products which can be used for human consumption as well as for aquaculture and agriculture applications.
We were incorporated on May 21, 2007 in the State of Nevada under the name Interpro Management Corp. On March 4, 2011, we changed our name to Stevia Corp. and effectuated a 35 for 1 forward stock split of all of our issued and outstanding shares of common stock. Effective November 15, 2013,we filed a Certificate of Amendment to the Company’s Articles of Incorporation to increase the total number of authorized shares of Common Stock from one hundred million (100,000,000) shares of Common Stock to two hundred fifty million (250,000,000) shares of Common Stock, each with a par value of $0.001.
On June 23, 2011, we closed a voluntary share exchange transaction (the “Share Exchange Transaction”) with Stevia Ventures International Ltd., a business company incorporated in the British Virgin Islands, pursuant to which we acquired certain rights relating to stevia production, including certain exclusive purchase contracts and a supply agreement related to stevia. In connection with the Share Exchange Transaction, on June 23, 2011, Mohanad Shurrab, a stockholder of the Company, surrendered 33,000,000 shares of the Company’s common stock to the Company for cancellation.
| 20 |
On March 19, 2012, we formed a wholly-owned subsidiary, Stevia Asia Limited, a company incorporated under the companies ordinance of Hong Kong (“Stevia Asia”) that will allow the Company to expand its China operations. Hero Tact Limited, a wholly-owned subsidiary of Stevia Asia, was incorporated under the companies ordinance of Hong Kong and renamed Stevia Technew Limited on April 28, 2012.
On July 5, 2012, Stevia Asia entered into a Cooperative Agreement with Technew Technology Limited (“Technew Technology”), a company incorporated under the companies ordinance of Hong Kong, and Zhang Ji, a Chinese citizen (together with Technew Technology, the “Partners”) pursuant to which Stevia Asia and Partners have agreed to engage in a joint venture to be owned 70% by Stevia Asia and 30% by Technew Technology, through the entity Stevia Technew Limited (the “Joint Venture”). The Partners will be responsible for managing the Joint Venture and Stevia Asia has agreed to contribute $200,000 per month, up to a total of $2,000,000 in financing to be applied on a project by project basis and subject to those projects remaining on target to generate positive EBITDA (earnings before interest, tax, depreciation and amortization) of at least 1.5 times the investment in any particular project and subject to Stevia Asia’s financial capabilities in terms of completing a financing or series of financings that provides the Company with the ability to contribute at least $200,000 in any given month. Completion of a financing or series of financings depends on the size of any private placements with investors that the Company may complete. Although Stevia Asia or Technew Technology may believe that a project is on target to generate positive EBITDA of at least 1.5 times the investment, there is no guarantee that any particular project will generate revenue. Stevia Asia contributed $200,000 to the Joint Venture in August 2012 which was applied to a specific aquaculture project that is ongoing but has not and will not contribute additional funds until it completes a financing or series of financings that provides the Company the ability to contribute at least $200,000 in any given month. The aquaculture project is focused on producing prawns and fish using the Company’s formulated products. The Joint Venture will participate in the revenue of specific ponds based on the pro-rata capital contribution allowing the flexibility to expand its participation as and when it has the ability to contribute additional funds. The Joint Venture also participated in an agriculture project in Vietnam where 102.5 acres of chili were cultivated using the Company’s formulated products. Part of the harvest occurred during the last quarter of the 2013 fiscal year generating revenue of $2,167,812.54 for the Company. The delay of additional capital is permissible pursuant to the joint venture agreement and will only impact the number and size of specific projects and the companies continue to explore potential stevia commercial applications but failure to complete a financing or series of financings sufficient to make additional contributions will have an adverse effect on our ability to execute our business plan. The Cooperative Agreement shall automatically terminate upon either Stevia Asia or Technew ceasing to be a stockholder in the Joint Venture, or may be terminated by either Stevia Asia or Technew upon a material breach by the other party which is not cured within 30 days of notice of such breach.
On October 1, 2013, we formed SC Brands Pte. Ltd., a Singapore corporation and a subsidiary in which we own a 70% equity interest. SC Brands will allow us to develop consumer brand products.
On February 24, 2014, we formed Real Hemp LLC, a wholly owned Indiana limited liability company that will focus on application of our proprietary processes to the commercial farming of the cannabis plant.
On February 26, 2014, we entered into a farm management and technology agreement with ebbu LLC to advise on scaling commercial extraction of identified cannabinoids from the cannabis plant.
The following diagram illustrates our corporate structure:
| 21 |
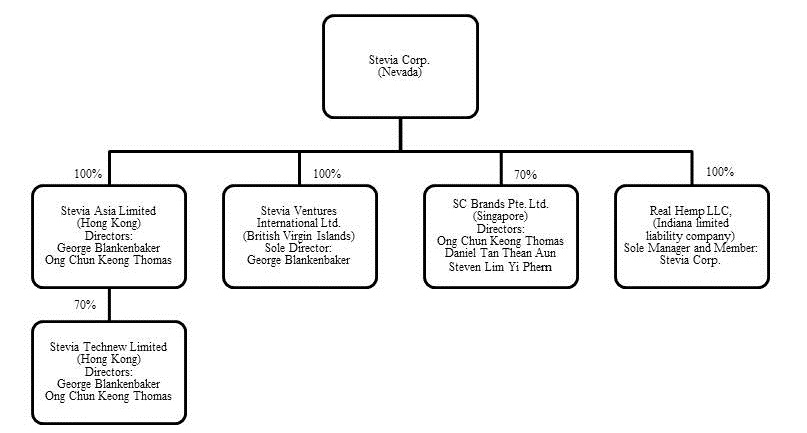
Overview
Our focus is on implementing quality agribusiness solutions to our partners, contract growers and customers to maximize the production of agri-products and the economic development of stevia and its extracts.
Our mission is to maximize shareholder value by consistently developing and acquiring the latest intellectual property and expanding our suite of formulated products and their applications and leveraging our farm management business model to maximize market penetration and revenue margins.
To achieve these goals we intend to develop a suite of intellectual property relating to stevia and its extracts that will enhance the value of our farm management operations. Through our relationships with Tech-New Bio-Technology, Growers Synergy and local institutes, we are exploring the market for commercial applications of stevia which will be vertically integrated into our services and production.
Our target markets are initially Vietnam, Indonesia and China. In Vietnam and Indonesia we have contracted with growers and have established our own nurseries and test fields. In China we are producing our proprietary formulated products and applying them to aquaculture projects under our revenue share model. Although our priority is Asia, our services are not limited to specific countries and we plan to pursue viable opportunities in other markets.
The Industry and Our Opportunity
Stevia as a Food Additive
We believe that health issues created by the modern diet are causing consumers to look for more natural products and simpler ingredient lines on the foods and beverages they purchase and causing governments to put pressure on the food industry to offer products with reduced calories.
In evaluating potential sweetener alternatives, manufacturers focus on taste, pricing, and a sustainable and scalable supply. We believe stevia fulfills these four criteria and has the added advantage of contributing no calories to food and beverage with a near zero glycemic index, making it safe for diabetics.
Originating from Paraguay, stevia leaf has been valued for centuries because of its sweetening and herbal properties and has been used as an approved sweetener in Japan and Korea for decades. Extracts from stevia contain a mixture of different molecules that vary depending upon climate and growing conditions and it was historically impossible to come up with clear and consistent specifications of the product needed to make it a reliable ingredient as well as conduct clinical trials required by the FDA for the approval process. This issue was only overcome in recent years by identifying the steviol glycoside molecules with the best taste profiles and by developing innovative and unique process technologies to separate and purify stevia extract to pharmaceutical levels of purity on a reliable and consistent basis: and, importantly, to do so in commercially viable volumes.
| 22 |
In 2008, Rebaudioside A, a steviol glycoside, was granted GRAS (Generally Recognized as Safe) status by the U.S. Food and Drug Administration following applications by Cargill and Merisant. Since then, approval by legislators across the world has opened the door to new formulations and reformulations of foods and beverages with zero or reduced calorie content. In 2009, stevia was incorporated into leading soft drinks brands manufactured by Coca-Cola and PepsiCo and has since been incorporated into many categories of food and beverages.
The stevia industry is segmented into several business processes, which can broadly be categorized as i) plant breeding and propagation, ii) farming, iii) extraction and refining, iv) product formulation, v) distribution and retail.
A significant portion of the cost of Rebaudioside A is a result of the leaf cost and we believe there remains considerable opportunity to build value in the supply chain by focusing on stevia agronomics. The stevia genus includes more than 100 species and each species contains unique sweet compounds. However, only two of these species contain steviol glycosides and of these two the variety with the sweetest compounds is stevia rebaudiana bertoni. There is relatively little technical knowledge of this species and almost all commercial growing of stevia has occurred in China because of the traditional Japanese and Korean markets. Now with the global market demand for high TSG (total steviol glycoside) and high Reb-A (Rebaudioside A) producing plants, there is an increased demand for agronomic and farm management expertise to establish new plantations and rapidly scale leaf production.
The primary competitors within this market segment include: PureCircle, which has extensive operations in China as well as subsidiaries in South America (Paraguay) and Africa (Kenya); Stevia One, an independent grower established in Peru; S&W Seed Company, who signed a supply agreement with PureCircle in July of 2010 to grow stevia in North America under its subsidiary, Stevia California; and GLG Life Tech Corporation, a China-centric company which has chosen to continue to focus on building and expanding its supply chain within China.
Stevia as a Commercial Product for Agriculture Use
Stevia is classified as a medicinal herb in China where more than 80% of the world’s supply of stevia is grown and stevia has been used as a medicinal herb as well as a sweetener for centuries in its native country of Paraguay. Japan is the largest consumer of stevia extract and stevia has accounted for more than 40% of Japan’s entire sweetener market consumption since 1992. Research articles studying the efficacy of stevia as a feed supplement and fertilizer have been published by several universities in Japan, China and South Korea for more than ten years. There are also several small local companies in Japan, South Korea and China that produce feed and fertilizer products that are formulated using stevia extracts and they have been supplying these products to their local markets for several years. We believe that the feed and fertilizer markets provide additional growth opportunities for stevia.
In July 2012, we obtained the rights to product formulations that add stevia extracts to an existing probiotic and enzyme product line produced by our technology partner, Tech-New Bio-Technology. We then obtained government approval in Vietnam to use the stevia product formulations for agricultural use such as fertilizer and animal feed supplement.
The first commercial application was started in August of 2012 for the production of approximately 2.5 acres of shrimp in China under the revenue share model with the intention to expand as we confirm available funding. In October 2012, a commercial application was started for the production of 102.5 acres of chili in Vietnam which was harvested during the first and second quarter of 2013. We are also using the formulations for the commercial stevia trial fields in Vietnam.
Our product line includes aquaculture feed for shrimp and fish, feed for livestock, granular fertilizers and foliar spray, each of which, we believe, holds the potential to open new revenue opportunities to us.
By vertically integrating down the supply chain, we believe we significantly enhance our revenue potential. An average hectare of stevia will produce approximately 6 tons of dry leaf per year. We have entered into a five year supply contract with an option to renew for an additional four years with a leaf buyer. Under the agreement, we will set a fixed price for the leaf each year based on the yearly average market prices for the quality of leaf provided. We are growing elite strains and we believe prices for high quality leaf will continue to be more stable than lower quality leaf, and as such, we believe our leaf prices will be more stable and predictable. We expect to generate approximately $2,000 for each ton of dry leaf, so each hectare will potentially produce $12,000 of dry stevia leaf. On average, dry stevia leaf produces approximately 10% of net usable extract by weight and the average price for the extract is approximately $100,000 per ton, so each hectare can potentially produce $60,000 of extract (6 tons x 10% x $100,000) which is five times the value of the dry stevia leaf and when we use the extracts to create our proprietary formulations, we can increase our revenue potential further.
Alternative Applications of Our Technology – Commercial Farming of Cannabis
We believe our expertise growing stevia to produce specific types of extracts and our technologies for post-harvest processing, extraction and product formulation may be applicable to the cannabis industry. In February 2014 we entered into a farm management and technology agreement with ebbu LLC to provide farm management consultancy and technical expertise related to growing the cannabis plant and extracting its cannabinoids. The cannabis plant produces many chemical compounds called cannabinoids and there are more than 85 different cannabinioids that have been identified and isolated from the cannabis plant that exhibit varied effects and many of these are being studied for their psychoactive and medical properties. Cannabis plants that have been bred to produce high levels of tetrahydrocannabinol (“THC”), a psychoactive constituent, are commonly referred to as marijuana. In 2004 the United Nations estimated that global consumption of marijuana indicated that approximately 4% of the adult world population (162 million people) used marijuana annually, and that approximately 0.6% (22.5 million) people used marijuana daily. Current federal and most state regulations prevent us from participating directly in the marijuana industry and we cannot guarantee that our services or technology will provide value if the laws do not evolve in favor of marijuana production and the commercial sale of marijuana derived products, but it is our goal to position the Company to take advantage of the shifting regulatory landscape where possible.
| 23 |
Cannabis plants that produce very low levels of THC and are grown for industrial purposes and foodstuff products are commonly referred to as hemp. Hemp products such as seeds, oil, protein, milk, fiber and cannabidiol (“CBD”) can be legally imported and traded in the United States, but it is not legal for a U.S. company to grow hemp because of its relationship to marijuana. Seventy percent of the world’s hemp production is currently produced in China. In February 2014 we registered a wholly owned subsidiary, Real Hemp LLC, to import, manufacture, license and sell hemp products in the U.S. The 2012 retail value of North American hemp food, vitamin and body care products was estimated to be in the range of $156 to $171 million by the Hemp Industries Association (HIA). When clothing, auto parts, building materials and other non-food or body care products are included, the HIA estimates that the total retail value of U.S. hemp products is about $500 million. Food and fiber uses for industrial hemp are growing rapidly and have increased over 300 percent, to an estimated 25,000 products, in the past few years. Much of that growth is coming from the increased sales of hemp food products. CBD is one of the active cannabinoids in the cannabis plant and is a major constituent of hemp, accounting for up to 40% of the plants extract, and is considered to have a wider scope of medical applications than THC. Similar to our goals in the marijuana industry, we intend to position the Company to take advantage of regulatory changes in the hemp industry.
Products and Services
Our farm management services include training the farmers on the correct protocols and methodologies and providing ongoing technical assistance during the crop cycle as well as providing inputs such as the seedlings, fertilizers and additives they are required to use. We apply our services under three business models which we classify as 1) contract farming model, 2) revenue share model and 3) product supply model.
Under the contract farming and revenue share models we do not charge for the services and inputs, but rather our services provide us with a competitive advantage to secure growers who are willing to dedicate their land and resources to grow crops with an expectation of high yielding, high quality crops and guaranteed purchase prices. Under these models we will generate our revenue from the crops that are grown and we only enter into production agreements with growers when there is already a committed buyer for the end crop. Under the contract farming model we will purchase the crop from the grower at a fixed price and sell to our own customer. Under the revenue share model, the grower already has their own buyer and we will share the revenue.
Under the product supply model we will market our products in combination with technical services to buyers and charge a fee. We believe that this model will contribute a small part of our overall revenue initially until we establish a proven track record and solid reputation for our services and products under the first two models. We do not expect to focus on providing strictly farm management or technical services for a fee and it is difficult to estimate what we would charge for such services.
To support our farm management services we established a research center in Vietnam on 25 acres of leased land. We confirmed elite plant varieties, developed propagation techniques, conducted field trials across several provinces, documented local operating procedures and post-harvest techniques, and began trial harvests in March 2012.
We continue to focus on research and development to further evolve and develop new protocols, methodologies and intellectual properties and believe that this will be key to maintain our competitive advantage.
We utilize the contract farming model to produce stevia leaf for our trial harvests and use the stevia extracts to produce our proprietary formulated products, which we are applying under the revenue share model to an aquaculture operation beginning August 2012 and a chili operation beginning October 2012.
In September 2012 we began providing samples of stevia extract to food and beverage companies and we are working closely with local parties in several South East Asian countries to provide technical information in support of recipe development. Although we believe that this product line will have growth potential, there is no guarantee that a high volume of stevia will be utilized by our customers. We expect companies will take another year to plan product launches.
Growth Cycle - The stevia plant is a perennial but the growing cycle varies greatly depending on the particular strain and location. Stevia is sensitive to frost and in China where most stevia is grown today, it is common to only have one or two harvests. Closer to the equator it is possible to harvest year round with some dormancy during the winter months. It is also possible to manipulate the harvest cycle and in developing countries where manual labor is the preferred method, a short cycle of as little as 45 to 60 days between harvests is preferred. However, in more developed countries where mechanization is the focus, a longer growing cycle is preferred and cycles of more than 120 days have been achieved.
Yield - Expected annual dry leaf yields of plant varieties commonly sourced from China is three to six tons per hectare (“Ha”). Field trial data indicates that six tons or more per Ha can be achieved working with elite strains. By continuing to build our inventory of elite strains and refine our farm management practices and technologies, we plan to improve yield and plant performance and exploit the economic value of our intellectual property.
| 24 |
Harvest - Stevia is a very labor intensive plant and traditionally has been harvested by hand. As larger commercial operations have begun to focus on stevia, a considerable amount of research is being put into the mechanization of planting, harvesting and leaf removal. While we will need to maximize mechanization in the United States to be economical, in many Asian locations there is both an abundance of low cost labor and an expectation that stevia will provide an economic stimulus and employ many of the farmers in poor rural areas. So the adoption of mechanization will need to consider both economic and social factors.
Location - Currently over 80% of stevia is grown in China and almost all of the high Reb-A variety stevia leaf is being produced in China. China is the center of commercial stevia growing for historical reasons due to its proximity to Japan and Korea, which have historically been the major markets for stevia. Due to its climate, we believe China is likely not the most geographically optimal location to grow stevia, as stevia is sensitive to frost and China typically produces only one or two crops per year, requiring leaf processors to purchase and store sufficient leaf for an entire year of production.
We believe that diversifying the supply chain of stevia leaf would provide several advantages:
| · | Incorporating Southern Hemisphere production provides two major growing seasons; | |
| · | Incorporation Equatorial production provides for year round production; | |
| · | Enables better control of leaf quality where major propagation of stevia varieties is controlled; | |
| · | Provides protection against country-specific political, regulatory, disease, and natural disaster risk; and | |
| · | Provides operations closer to end markets. |
We believe infrastructure is a major criteria for field site selection and can be especially challenging in developing countries. In addition, we believe a viable site must have the proper weather and soil that is suitable for plant growth as well as being in a location that satisfies logistical business considerations, such as being easily accessible and in close proximity to a capable labor pool. It is our belief that access to water can often be a challenge and greatly limits the areas where an irrigation model can be applied. We believe Vietnam has excellent road infrastructure and our fields are easily accessible by passenger car or lorry and most potential growing areas are located within hours of a major port city. Indonesia has an abundance of low cost labor and land available for acquisition that is suitable for new varieties of stevia that we are breeding and/or acquiring to grow in the equatorial zone.
Land Use and Capital Requirements - As we expand our operations, there are two primary business models available to manage farm operations. The plantation model will involve us controlling the land and assets through lease or purchase arrangements and hiring the necessary workers which will require higher upfront capital cost but enable rigorous control over operations with potentially higher revenue per acre. The contract farm model involves entering into agreements with existing farmers to utilize our agriculture inputs and protocols in order to produce specified crops under contract at negotiated prices. The contract farm model requires lower upfront capital and enables us to more quickly scale over larger areas in those instances where we are able to efficiently manage operations and implement supervisory control. If successfully implemented, we believe the contract farming model provides the fastest ramp to positive cash flow while also conserving capital.
We are managing our trial harvest stevia farms under the contract farming model and plan to continue using this model.
Under the revenue share model the grower owns, leases or contracts the land and we provide our farm management services and products as part of the agriculture inputs and then we share the revenue. This does not require us to have any obligations or liability for land and enables us to expand rapidly and maximize revenue by leveraging existing operations with minimal capital commitment.
We intend to scale the use of our formulated products using both the contract farm model and revenue share model which we started implementing in August of 2012. We will initially work on projects with our joint venture partner.
Labor and Research and Development - Our initial research and development funding was used to establish our research center and engage specialists who have secured elite plant varieties, culled the original planted varieties, developed propagation techniques, conducted field trials, documented local operating procedures and developed post-harvest techniques. We target spending approximately fifteen percent of our operating expense budget towards research and development to continue improving and develop new intellectual properties.
Financial - The value of the stevia leaf fluctuates based on supply and demand and the quality of the leaf. Wide seasonal variances on the open market are common and can make long-term planning difficult. Because we have entered into a long-term supply contract with a leaf buyer and we are growing elite strains, we believe our prices will be more stable and predictable and we will be able to plan our growth and commit to large contract growers. In addition, buyers of leaf pay a substantial premium for high quality leaf. This places strong economic value on our intellectual property, including our elite stevia strains, and our farm management solutions.
Current contracted selling price for leaf that meets the minimum standards is set at a fixed price. Leaf exceeding the minimum standards will receive a premium for which the benchmarks and price tiers will be reviewed each year based on comparative market leaf quality and supply and demand.
| 25 |
Historically, leaf that produced 13% TSG and 70% Reb-A was purchased at a premium. Elite strains can potentially deliver TSG well above 12% and Reb-A above 80% providing significant economic advantage. Minimum standards require a TSG of 12% or more, Reb-A to be at least 60% of TSG, maximum of 5% impurities and a maximum moisture content of 10%. During the refining process, the net yields of usable extract will be slightly lower.
Our Key Contracts and Relationships
Growers Synergy
Effective November 1, 2011, we engaged Growers Synergy Pte Ltd, a regional farm management services provider (“Growers Synergy”), to provide farm management operations and back-office and regional logistical support for our Vietnam and Indonesia operations for a period of two years at a cost of $20,000 per month and the agreement was renewed on November 1, 2013 for an additional year. In addition, Growers Synergy will enter into an agreement to purchase from us all the non-stevia crops produced at the farms for which they are providing management services.
We believe that the relationship with Growers Synergy will provide us with a strategic advantage and potential synergistic partnership by providing us with guaranteed off-take agreements for agriculture crops other than stevia, which will be produced as part of inter-cropping practices to maintain optimal soil conditions for stevia farming. Growers Synergy will work with us and our technology partner, Tech-New Bio-Technology, to combine the agronomy protocol with the farming models. Models and their related protocols have been commercially field tested during the first two years working with the provincial and national programs and establishing 100 Ha of field trials.
A local farm management service, such as Growers Synergy, is critical to assist us in training local teams with the documented protocol sufficient to scale to 1,000 Ha to create a turnkey project. Our goal is to be vested with fully documented protocols, local teams of trained staff capable of supporting the scale up to 1,000 Ha and farmer communities that are capable of growing stevia and other crops. To help us achieve this Growers Synergy will provide the necessary resources and assign staff to fill certain managerial and support staff positions.
Tech-New Bio-Technology
In March 2012, we entered into both a Supply Agreement and Cooperative Agreement with Guangzhou Health China Technology Development Company Limited, operating under the trade name Tech-New Bio-Technology (“TechNew”). TechNew is a developer and manufacturer of hi-tech biotechnology products which offers a series of specialized ecological fertilizers, microbiological preparations and management systems for the agriculture and aquaculture industry as well as technologies for the extraction and refinement of high purity stevia. Under the terms of the Supply Agreement, we are able to sell dry stevia plant product exclusively to TechNew including all leaf and stem for a term of five years with an option to renew for a further four years with the price to be negotiated by the parties on a yearly basis to reflect changes in the specifications and market price. During the first two years TechNew is obligated to purchase all of our production with quantity to be negotiated from the third year onwards. Under the terms of the Cooperative Agreement, we agreed to explore potential technology partnerships with TechNew, with the intent to formalize a joint venture to pursue promising technologies and businesses. These include the inclusion of stevia extracts in its current product formulations for use in agriculture and aquaculture applications including fertilizers and feed.
Through our cooperative agreement with TechNew, we will also explore a potential relationship to integrate extraction and refining technology to produce high purity Reb-A and other steviol glycosides for the consumer market. We believe that vertically integrating our technologies for both commercial and consumer products may provide advantages of a diversified market, but we do not intend to enter the consumer market with a finished stevia product. It is our goal to develop core strengths in farm management and developing technologies for production and post harvest processes, and we believe that the consumer market for stevia is extremely competitive.
We supplied leaf to TechNew from our trial harvests and all of the leaf we have supplied has been used to produce products formulated with stevia extract. It is our intention to apply as much leaf as possible towards producing the higher value added products rather than sell the leaf as a commodity under the supply contract.
TechNew Technology Limited
On July 5, 2012, our wholly-owned subsidiary, Stevia Asia entered into a Cooperative Agreement with Technew Technology Limited (“Technew Technology”), a company incorporated under the companies ordinance of Hong Kong, and Zhang Ji, a Chinese citizen (together with Technew Technology, the “Partners”) pursuant to which Stevia Asia and Partners have agreed to engage in a joint venture to be owned 70% by Stevia Asia and 30% by Technew Technology (the “Joint Venture”), through the entity Stevia Technew Limited. The Joint Venture will allow us to further explore potential stevia commercial applications, which we would integrate into our farm management services and our own stevia production.
Independent Grower Relationships
We plan to develop a network of partner growers who we can market our production methods and technologies to and who will also help supply us with the stevia product necessary to fulfill our supply obligations. To date we have entered into initial purchase agreements for stevia under the contract farming model where we provide the seedlings, fertilizer additives, protocols and technical supervision with an obligation to purchase the stevia leaf at a fixed price per ton and the grower is responsible for the land, labor and all other inputs. The agreements are reviewed annually to negotiate price and quantity for the subsequent renewal year to reflect changes in specifications, market prices and demand. We have also entered into revenue share agreements with growers where we provide our proprietary feed or fertilizer additives and farm management services in return for a share of the revenue. These agreements are reviewed each growing/harvest cycle with renewal terms to be negotiated and confirmed for each subsequent cycle.
| 26 |
Our Farm Management Services and Intellectual Property
Our objective is to provide a full spectrum of farm management services to manage our contract farms, service industry growers and provide for optimal production. To achieve this objective, our focus is on intellectual property development and continued development and improvement of cultivar varieties for intended growing sites, propagation protocol, cultivation technology including an intercropping system and regional adaptability test, and post-harvest and refinery processes.
We are also continuing to develop and improve local SOP (standard operating procedures) manuals specific to each growing location and plant variety, which document the proper use of all inputs including a proprietary crop production system that we believe is more efficient and cost effective than traditional methods. We believe these customized operating manuals will result in advanced propagation and growing techniques that can improve the quality and efficiency of a variety of crops.
We are also developing a wide portfolio of highly efficient and environmentally friendly crop nutrition products. These products are performance minerals, plant phyto-chemicals, functional nutrients and microbial formulations. All products are derived from natural sources and can be used as sustainable agriculture solutions and/or for organic farming. While it is our intent to develop the foregoing highly efficient and environmentally friendly crop nutrition products, there is no guarantee we will be successful in developing such a portfolio of products.
We are still developing protocols regarding stevia production and we plan to provide a wide spectrum of agricultural consulting and solutions for stevia growers, including:
TechNew Suite of Products - through our technology partner, TechNew, we are able to contract manufacture the extraction and refinement of high purity stevia and we acquired their formulas for using stevia extract in feed and fertilizer applications. We have also entered into a joint venture with Technew Technology to further explore potential stevia commercial applications, which we would integrate into our farm management services and our own stevia production.
Elite Germplasm - high performance mother stock suitable for varied regions and environment.
Advanced Propagation Techniques- methods that are efficient, more cost effective, and produce a higher quality plant.
To date we have not filed patents or registered trademarks and we do not license any of our technologies. We previously had a license arrangement with Agro-Genesis, however, such license was cancelled when we partnered with TechNew.
Our Competitive Advantage
We believe our intellectual property suite that we are developing and our ability to serve across a wide spectrum of agricultural consulting and solutions will provide us with a competitive advantage against our competitors.
We also believe our intellectual property, particularly our fertilizers and feed additives and other input products used in our protocols, have the potential to create a dedicated customer base because the protocols once implemented on a farm call for continual use of our fertilizers and feed additives and other products as a mandatory production input. We believe this long-term customer relationship can enable us to create a substantial barrier to entry to potential new competitors, while at the same time providing networking benefits that could further propagate our business.
Our ability to fully develop our suite of products and apply them to a customer base is dependent on our ability to raise sufficient capital to fund our business operations.
Market Trends
The original products launched that used stevia were zero calorie beverages. Subsequent product launches included a blend of sugar and stevia that advertised reduced calories. Stevia is now used across 38 categories of food and beverages with most of the applications involving a blend of sugar and stevia for a reduced calorie product using all natural sweeteners.
Our Properties
Our primary focus is on providing farm management services to our contract growers. We have acquired two grower supply contracts and three nursery fields in Vietnam. More than twenty fields have been established in five provinces in the northern half of Vietnam with total propagation exceeding 100 Ha (250 acres).
The provincial locations include Vinh Phuc, Tuyen Quang, Thanh Hoa, Ha Tinh and Lai Chau.
| 27 |
On December 14, 2011 we entered into a land lease agreement with Stevia Ventures Corporation, one of our Suppliers, and Vinh Phuc Province People’s Committee Tam Dao Agriculture & Industry Co., Ltd (“Vinh Phuc”) whereby Stevia Ventures Corporation leased 10 Ha (25 acres) of land over 5 years and we developed a research facility that will also serve as a propagation center for farms located in the surrounding provinces and particularly those serving the provincial and national sponsored projects.
To better service multiple farms located across the many provinces stretching from north central Vietnam to the Chinese border, we will utilize the greenhouse facilities of our local grower partners in a decentralized model that more efficiently addresses the logistical challenges presented by the contract farming model. It is assumed that the commercial fields will be scaled by stem cutting and we will provide the seedlings to the growers as one of the inputs.
In addition to our Vietnam operations, in April 2012, we established a 1 Ha (2.5 acres) initial field trial in Indonesia which utilizes our intercropping model.
We lease office space with Leverage Investments, LLC, an entity owned and controlled by our President, for $500 per month on a month-to-month basis since July 1, 2011.
Regulation
Stevia extracts may be used in a wide variety of consumer products including soft drinks, vegetable products, tabletop sweeteners, confectioneries, fruit products and processed seafood products, in a wide range of countries, including almost all major markets, and as a dietary supplement in others. Clinical studies have supported the safety and stability of stevia’s various high purity compounds used in food and beverages. There is no documented health threat.
Cargill and Merisant each submitted applications to the United Stated Food and Drug Administration (FDA) in 2008 for GRAS approval. On December 17, 2008 the stevia extract, Rebaudioside A (Reb-A), received GRAS approval.
In December 2008, Australia and New Zealand approved highly purified forms of stevia extracts as safe for use in food and beverages. Previously, such extracts had only been permitted for use as a dietary supplement in these countries.
Stevia extracts have been sanctioned by the Ministry of Health of China to be used as a food additive, and are listed in the Sanitation Standard of Food Additives.
In July 2010 the FDA issued GRAS clearance for PureCircle’s high purity SG95 stevia product which opened up opportunities for many more applications as well as more cost effective solutions.
In November 2011, the European Union cleared stevia for use as a food additive in its twenty seven member states.
Further regulatory clearances were secured for Reb-A in nearly all of the developed countries and South East Asian countries confirming the growing regulatory support for high purity stevia.
Our proprietary fertilizer and feed additive products are approved for use in China and South East Asia and we have started using them in commercial operations. All of the ingredients in the products are natural compounds and are approved by the major developed countries, but registration of the products will be required in each country before importation is allowed.
Foreign Currency Exchange Rate
The Company expects that international revenues will account for a majority of our total revenues. Our international operations expose the Company to foreign currency fluctuations. Revenues and related expenses generated from our international subsidiaries will generally be denominated in the functional currencies of the local countries. For example, revenues derived from the People’s Republic of China (“PRC”) will be denominated in Renminbi, or RMB.
Our statements of income of our international operations are translated into United States dollars at the average exchange rates in each applicable period. To the extent the United States dollar strengthens against foreign currencies, the translation of foreign currency denominated transactions will result in reduced revenues, operating expenses and net income for our business. Similarly, our revenues, operating expenses and net income will increase if the United States dollar weakens against foreign currencies.
We are also exposed to foreign exchange rate fluctuations as we convert the financial statements of our foreign subsidiaries and our investments in equity interests into United States dollars in consolidation. If there is a change in foreign currency exchange rates, the conversion of the foreign subsidiaries’ financial statements into United States dollars will lead to a translation gain or loss which is recorded as a component of accumulated other comprehensive income which is part of stockholders’ equity. In addition, we may have certain assets and liabilities that are denominated in currencies other than the relevant entity’s functional currency. Changes in the functional currency value of these assets and liabilities create fluctuations that will lead to a transaction gain or loss.
| 28 |
China – The Company expects to derive revenue from China. Pursuant to the Foreign Currency Administration Rules promulgated in 1996 and amended in 2008 and various regulations issued by the State Administration of Foreign Exchange (“SAFE”), and other relevant PRC government authorities, RMB is freely convertible only to the extent of current account items, such as trade-related receipts and payments, interest and dividends. Capital account items, such as direct equity investments, loans and repatriation of investments, require the prior approval from the SAFE or its local counterpart for conversion of RMB into a foreign currency, such as U.S. dollars, and remittance of the foreign currency outside the PRC.
Payments for transactions that take place within the PRC must be made in RMB. Unless otherwise approved, PRC companies must repatriate foreign currency payments received from abroad. Foreign-invested enterprises may retain foreign exchange in accounts with designated foreign exchange banks subject to a cap set by the SAFE or its local counterpart. Unless otherwise approved, domestic enterprises must convert all of their foreign currency receipts into RMB. The value of the RMB against the U.S. dollar and other currencies is affected by, among other things, changes in China’s political and economic conditions. Since July 2005, the RMB has no longer been pegged to the U.S. dollar. The RMB may appreciate or depreciate significantly in value against the U.S. dollar in the medium to long term. Moreover, it is possible that in the future, PRC authorities may lift restrictions on fluctuations in the RMB exchange rate and lessen intervention in the foreign exchange market.
Because some of our revenue is expected to come from China, appreciation or depreciation in the value of the RMB relative to the U.S. dollar would affect our financial results reported in U.S. dollar terms without giving effect to any underlying change in our business or results of operations. As a result, we face exposure to adverse movements in currency exchange rates as the financial results of our Chinese derived revenue are translated from local currency into U.S. dollar upon consolidation. Our operations are subject to risks typical of international business, including, but not limited to, differing economic conditions, changes in political climate, differing tax structures, other regulations and restrictions, and foreign exchange rate volatility.
International Laws
A significant portion of our initial business operations will occur in Vietnam. We will be generally subject to laws and regulations applicable to foreign investment in Vietnam. Similarly, as we expand into Indonesia and other markets, we will be subject to the laws and regulations of such jurisdictions. The Vietnam legal system is based, at least in part, on written statutes. However, since these laws and regulations are relatively new and the Vietnamese legal system continues to rapidly evolve, the interpretations of many laws, regulations and rules are not always uniform and enforcement of these laws, regulations and rules involves uncertainties. Similar to Vietnam, the modern Indonesia legal system was formed relatively recently and is continuing to evolve.
| Country | Type of Approval | |
| North America | ||
| USA | Food additive | |
| Canada | Food additive | |
| Mexico | Food additive | |
| Latin America | ||
| Argentina | Food additive | |
| Brazil | Food additive | |
| Chile | Food additive | |
| Colombia | Food additive | |
| Ecuador | Food additive | |
| Paraguay | Food additive | |
| Peru | Food additive | |
| Uruguay | Food additive | |
| Venezuela | Food additive | |
Asia Pacific | ||
| Australia | Food additive | |
| Brunei | Food additive | |
| China | Food additive | |
| Hong Kong | Food additive | |
| Indonesia | Food additive | |
| Japan | Food additive | |
| Malaysia | Food additive | |
| New Zealand | Food additive | |
| Singapore | Food additive | |
| South Korea | Food additive | |
| Taiwan | Food additive | |
| Thailand | Food additive | |
| Vietnam | Food additive |
| 29 |
| Europe | ||
| Austria | Food additive | |
| Belgium | Food additive | |
| Bulgaria | Food additive | |
| Cyprus | Food additive | |
| Czech Republic | Food additive | |
| Denmark | Food additive | |
| Estonia | Food additive | |
| Finland | Food additive | |
| France | Food additive | |
| Germany | Food additive | |
| Hungary | Food additive | |
| Ireland | Food additive | |
| Italy | Food additive | |
| Latvia | Food additive | |
| Lithuania | Food additive | |
| Luxembourg | Food additive | |
| Malta | Food additive | |
| The Netherlands | Food additive | |
| Poland | Food additive | |
| Portugal | Food additive | |
| Romania | Food additive | |
| Slovakia | Food additive | |
| Slovenia | Food additive | |
| Spain | Food additive | |
| Sweden | Food additive | |
| Switzerland | Food additive | |
| Russia | Food additive | |
| United Kingdom | Food additive |
We cannot predict the effect of future developments in the legal systems of developing countries, including the promulgation of new laws, changes to existing laws or the interpretation or enforcement thereof, the preemption of local regulations by national laws, or the overturn of local government’s decisions by the superior government. These uncertainties may limit legal protections available to us.
Marketing
We believe it is important to educate the local governments and farmer communities on the merits of stevia becoming a new commercial crop and its potential as a new economic stimulus for rural farmers. Our President, Mr. George Blankenbaker, and our local partner have been conducting talks and training sessions for more than three years in Vietnam and have fostered local support at many levels. To support the farmer’s transition to stevia farming and provide an opportunity to showcase the stevia opportunity to farmers’ communities, the Vietnam government provided financial support at both the provincial and national level to plant 20 Ha (50 acres) and 50 Ha (125 acres) respectively, both of which were completed in 2012. The fields were small plots located in several villages and served as demonstration fields and stepping stones to gain wide support from growers in several villages.
We have entered into formal cooperative agreements with several local institutes, including the National Institute of Medicinal Materials in Hanoi and the Agricultural Science Institute of Northern Central Vietnam. The terms of these agreements generally provide that we will provide stevia seedlings and other products and services, at prices and in quantities as will be mutually agreed by the parties, at the clients’ nurseries and provide the clients with off-take agreements for crops produced using our systems. As part of our services, we provide technical assistance to assure the clients adhere to our established growing protocols. We also agree to work cooperatively with the clients on research projects relating to stevia development, the cost of such projects to be shared between the parties as may be mutually agreed. These agreements provide local technical assistance for our grower partners and also provide additional credibility when our grower partners present the stevia opportunity to the local farmers’ communities.
We are also in contact with non-governmental organizations (NGO) that are seeking programs to bring to the communities that they serve which are generally located in poor rural areas in need of economically sound projects. If the stevia model proves to be viable for these locations, the NGOs have indicated that they will be interested in introducing and funding stevia farming programs. However, many of these poor rural areas are located in areas of poor soil quality, that lack adequate access to water or that suffer from other environmental constraints which limit the opportunities for this approach.
We also hope to generate many local testimonials from our field trials and the farmers in Vietnam are very fluid and willing to adopt new crops if the new crops are proven to be more economically viable than their current crops.
In connection with commercial opportunities for stevia derived products, we intend to develop a mark that can be applied to a buyer’s brand which would signify premium quality stevia-derived products.
| 30 |
Currently our marketing efforts are focused on educating our growers on our new proprietary formulations. These efforts are more administrative in nature and we do not currently anticipate a need for a large marketing budget to support current operations.
Product Alternatives
As a full service stevia farm management service provider we will face competition from both non-stevia sweetener products and from other service providers within the stevia industry.
Food Additive Product Alternatives - We believe stevia is the leader among natural zero calorie sweeteners at this time and it takes years to develop and bring to market new sweeteners of which few end up possessing all the qualities needed to be adopted mainstream. At this time we are not aware of any proven and viable alternative which possesses all of the positive qualities of stevia. As discussed above, the other sweeteners currently on the market lack many of the qualities that make stevia attractive to consumers and manufacturers, including the zero calorie/near zero glycemic index combination.
Therefore, we believe that the most likely threat to stevia growers will come from alternative “natural” methods to produce stevia extracts that obviate the need to farm stevia, such as fermentation-derived stevia.
A fermentation-derived stevia ingredient can be produced in a lab where low cost plant materials are converted into sweet steviol glycosides through controlled fermentation methods that duplicate the natural biochemical pathways that are involved in the natural production of the sweet components of the stevia leaf and would still meet the requirements to be classified as a “natural” ingredient and when done at volume could potentially be produced more economically than the farming method and without impurities.
Major known companies that are progressing down this track include Evolva Holding SA of Switzerland who has acquired San Francisco based Abunda Nutrition, Inc., Blue California of Rancho Santa Margarita, California and Stevia First Corporation of Yuba City, California.
There are four areas on which we will focus to reduce the risk and/or impact of alternative methods of stevia ingredient production.
| 1. | Increase farming efficiencies . The more efficient and scaled farming becomes, the higher the economic hurdle will be for other methods of production. We believe that our intellectual property and continued research and development activities will allow our farms and those of our customers to increase efficiencies, decrease cost of production and produce better quality leaf. |
| 2. | Intellectual Property Protections. We have a strong focus on developing protectable intellectual property which we believe should create barriers to entry and protect our methodologies. Additionally, where applicable we will continue to consider the acquisition of potentially synergistic intellectual property. |
| 3. | Crop Diversification. Our farm management infrastructure and the majority of our intellectual property is applicable to most crops providing us with the flexibility to diversify our crops and the customer base for our farm management solutions. |
| 4. | Product Diversification . We will explore additional markets and uses for stevia and seek to acquire technology to diversify its applications. |
Commercial Product Alternatives
Small regional companies in Japan, China, and South Korea have been producing commercial stevia products for several years, focusing on their local markets. We believe with the awareness of stevia on a global scale, this will provide an opportunity to develop a large commercial market. Once the market reaches critical mass, large companies will likely enter the market.
We intend to protect our market by positioning ourselves as both the primary provider of raw extract to companies as well as establishing our own vertical markets utilizing our farm management core competency to contract farm using our commercial stevia products.
Employees
George Blankenbaker, our President and a director, is our sole employee.
Our relationship with our farm management partner, Growers Synergy, currently provides the staffing necessary to operate our farms and our technology partner, TechNew, provides the staffing for our technical operations.
We chose to outsource the operations management during our development phase to minimize expenses and provide a team of qualified experienced staff to lead us through the development phase until we are ready to commercialize. As we begin commercialization and revenue generation, we intend to begin to hire full time staff.
| 31 |
Summary Plan of Operation
The following table provides our current and intended plan of operation, including our goals, estimated costs and timelines in connection with our aim of expanding our operations to provide farm management services and products in China and South East Asia. There can be no assurance that our plan of operation and goals will be accomplished on the timelines set forth below, at the costs noted, or at all.
| Goals | Status | Requirements | Timeline | Estimated Budget | ||||||
| Build elite strains of Stevia | Completed (Continue Improving) | - | Ongoing | * | ||||||
| Develop Intellectual Property | Completed (Continue Improving) | - | Ongoing | * | ||||||
| Develop Farming Protocols | Completed (Continue Improving) | - | Ongoing | * | ||||||
| Develop Operating Manuals | Completed (Continue Improving) | - | Ongoing | * | ||||||
| Conduct Product Testing | Completed (Continue Improving) | Continue R&D | Ongoing | $ | 10,000/month | |||||
| Trial harvests (multiple countries) | Completed (Continue Improving) | Continue R&D | Ongoing | $ | 100,000/year | |||||
| Feed Product approvals | Approved in South East Asia & China | Register in other countries | by 2015 | negligible | ||||||
| Fertilizer Product approvals | Approved in South East Asia & China | Register in other countries | by 2015 | negligible | ||||||
| Hire Additional Full Time Staff | Staff are Identified | Adequate working capital | 2nd Quarter 2014 (est) | $ | 50,000/month | |||||
| Increase revenue by expanding farm management operations under joint venture with Technew | Begun in August 2012 | Adequate working capital | 1st Quarter 2015 (est) | $ | 3,000,000 | ** | ||||
* These goals have initially been completed but improvements are ongoing and the costs will depend on opportunities to make such improvements as they arise.
** This is what the Company is targeting through a combination of revenue generation and external financing.
PROPERTIES
Our international corporate office is located at 14 Chin Bee Road, Singapore 619824. We also maintain an office in Vietnam at No. 602, CC2A, Thanh Ha‘s building, Bac Linh Dam, Hoang Mai district, Hanoi, Vietnam and in Hong Kong, at 19/F Kam Chung Comm Bldg 19-21, Hennessy Rd, Hong Kong and in the United States, at 7117 US 31 South, Indianapolis, IN 46227.
We have also developed a research facility on 10 Ha (25 Acres) of land leased by Stevia Ventures Corporation and have prepaid the first year lease payment of $30,000 and the six month lease payment of $15,000 as security deposit.
LEGAL PROCEEDINGS
None.
MARKET FOR COMMON EQUITY AND RELATED SHAREHOLDER MATTERS
Market Information
Our common stock is quoted on the OTCQB under the symbol STEV. The closing bid price for our stock as of March 26, 2014, was $0.28.
The following is the range of high and low bid prices for our common stock for the periods indicated. The quotations reflect inter-dealer prices, without retail mark-up, mark-down or commissions and may not represent actual transactions.
| Interim Period From April 1, 2013 to March 26, 2014 | High | Low | ||||||
| First Quarter (April 1, 2013 to June 30, 2013) | $ | .349 | $ | .20 | ||||
| Second Quarter (July 1, 2013 to September 30, 2013) | $ | .2595 | $ | .1234 | ||||
| Third Quarter (October 1, 2013 to December 31, 2013) | $ | .164 | $ | .097 | ||||
| Fourth Quarter (January 1, 2014 to March 26, 2014) | $ | .28 | $ | .083 | ||||
| Fiscal Year Ended March 31, 2013 | High | Low | ||||||
| First Quarter (June 30, 2012) | $ | 1.69 | $ | .75 | ||||
| Second Quarter (September 30, 2012) | $ | .83 | $ | .26 | ||||
| Third Quarter (December 31, 2012) | $ | .341 | $ | .101 | ||||
| Fourth Quarter (March 31, 2013) | $ | .41 | $ | .146 | ||||
| Fiscal Year Ended March 31, 2012 | High | Low | ||||||
| First Quarter (June 30, 2011) | $ | 1.60 | $ | .25 | ||||
| Second Quarter (September 30, 2011) | $ | 1.00 | $ | .85 | ||||
| Third Quarter (December 31, 2011) | $ | 1.05 | $ | .56 | ||||
| Fourth Quarter (March 31, 2012) | $ | 2.75 | $ | .667 | ||||
| 32 |
Stockholders
As of March 26, 2014, there were 145,972,713 shares of common stock issued and outstanding held by 19 stockholders of record (including street name holders).
Dividends
We have not paid dividends to date and do not anticipate paying any dividends in the foreseeable future. Our Board of Directors intends to follow a policy of retaining earnings, if any, to finance our growth. The declaration and payment of dividends in the future will be determined by our Board of Directors in light of conditions then existing, including our earnings, financial condition, capital requirements and other factors.
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis of the results of operations and financial condition for the fiscal years ended March 31, 2012 and 2013, and the three and nine months ended December 31, 2013, should be read in conjunction with the financial statements and related notes and the other financial information that are included elsewhere in this Prospectus. This discussion includes forward-looking statements based upon current expectations that involve risks and uncertainties, such as our plans, objectives, expectations and intentions. Actual results and the timing of events could differ materially from those anticipated in these forward-looking statements as a result of a number of factors, including those set forth under the Risk Factors, Cautionary Notice Regarding Forward-Looking Statements and Business sections in this registration statement on Form S-1. We use words such as “anticipate,” “estimate,” “plan,” “project,” “continuing,” “ongoing,” “expect,” “believe,” “intend,” “may,” “will,” “should,” “could,” and similar expressions to identify forward-looking statements.
The following discussion should be read in conjunction with our consolidated financial statements and notes thereto included elsewhere in this Registration Statement on Form S-1. Forward looking statements are statements not based on historical information and which relate to future operations, strategies, financial results or other developments. Forward-looking statements are based upon estimates, forecasts, and assumptions that are inherently subject to significant business, economic and competitive uncertainties and contingencies, many of which are beyond our control and many of which, with respect to future business decisions, are subject to change. These uncertainties and contingencies can affect actual results and could cause actual results to differ materially from those expressed in any forward-looking statements made by us, or on our behalf. We disclaim any obligation to update forward-looking statements.
Overview
We were incorporated on May 21, 2007 in the State of Nevada under the name Interpro Management Corp. On March 4, 2011, we changed our name to Stevia Corp. and effectuated a 35 for 1 forward stock split of all of our issued and outstanding shares of common stock. Effective November 15, 2013,we filed a Certificate of Amendment to the Company’s Articles of Incorporation to increase the total number of authorized shares of Common Stock from one hundred million (100,000,000) shares of Common Stock to two hundred fifty million (250,000,000) shares of Common Stock, each with a par value of $0.001.
We generated revenues during the 2013 fiscal year. We expect our primary sources of revenue will be (i) providing farm management services, which will provide protocols and other services to agriculture, aquaculture, and livestock operators, (ii) the sale of inputs such as fertilizer and feed additives to agriculture, aquaculture and livestock operators, (iii) the sale of crops and seafood produced under contract farming, (iv) the sale of products derived from the stevia plant and other agriculture crops, (v) providing extraction and refining technology services related to stevia and other medicinal herbs and (v) the sale of branded consumer products made from natural ingredients.
During 2012, we completed our first commercial trials of stevia production in Vietnam. In connection with such production we have entered into supply agreements for the off-take of the stevia we produce and entered into an agreement with Growers Synergy Pte Ltd to assist in the management of our Asia day-to-day operations. We have also developed commercial applications of stevia derived products and have developed and acquired certain proprietary technology relating to stevia development which we can integrate into our own stevia production and our farm management services. In connection with our intellectual property development efforts we have engaged TechNew Technology Limited (“TechNew), as our technology partner in Vietnam and on July 5, 2012 we entered into a Cooperative Agreement (the “Cooperative Agreement”) through our subsidiary Stevia Asia Limited (“Stevia Asia”), with Technew and Zhang Ji, a Chinese citizen (together with Technew, the “Partners”) pursuant to which Stevia Asia and Partners have agreed to engage in a joint venture to develop certain intellectual property related to stevia development, such joint venture to be owned 70% by Stevia Asia and 30% by Technew (the “Joint Venture”). Pursuant to the Cooperative Agreement Stevia Asia agreed to contribute $200,000 per month, up to a total of $2,000,000 in financing, subject to the performance of the Joint Venture and Stevia Asia’s financial capabilities.
We have also continued to establish research and production relationships with local institutions and companies in Vietnam. In April, 2012 we announced plans to begin field trials in Indonesia.
On March 19, 2012, we formed a wholly-owned subsidiary, Stevia Asia Limited, a company incorporated under the companies ordinance of Hong Kong (“Stevia Asia”) that will allow the Company to expand its China operations. Hero Tact Limited, a wholly-owned subsidiary of Stevia Asia, was incorporated under the companies ordinance of Hong Kong and renamed Stevia Technew Limited on April 28, 2012.
On October 1, 2013, we formed SC Brands Pte. Ltd., a Singapore corporation and a subsidiary in which we own a 70% equity interest. SC Brands will allow us to develop branded consumer products.
On February 24, 2014, we formed Real Hemp LLC, a wholly owned Indiana limited liability company that will focus on developing hemp products to be sold in the US.
| 33 |
Results of Operations
The following discussion of the financial condition, results of operations, cash flows, and changes in our financial position should be read in conjunction with our audited consolidated financial statements and notes included in our Annual Report on Form 10-K for the fiscal year ended March 31, 2013, filed July 15, 2013. Such financial statements have been prepared in conformity with U.S. GAAP and are stated in United States dollars.
Comparison of Three Month Periods Ended December 31, 2013 and December 31, 2012
For the three month period ended December 31, 2013 we incurred a net loss of $831,410, compared to a net loss of $364,601 for the three month period ended December 31, 2012. The increase was mainly attributed to a $561,077 loss attributed to debt settlement during the three month period ended December 31, 2013.
General and administration expenses and professional fees for the three month period ended December 31, 2013 amounted to $133,641 and $178,135 respectively, compared to $69,302 and $123,356 during the three month period ended December 31, 2012. Research and development fees for the three month period ended December 31, 2013 were $71,930 compared to $0 during the three month period ended December 31, 2012. Directors fees, officer salary and compensation and other salary and compensation were $7,813, $0 and $378 respectively, compared to $93,750, $0 and $59,105 during the three month period ended December 31, 2012.
Comparison of Nine Month Periods Ended December 31, 2013 and December 31, 2012
For the nine month period ended December 31, 2013 we incurred a net loss of $2,017,484, compared to a net loss of $1,342,473 for the nine month period ended December 31, 2012. The increase was mainly attributed to an increase in officer salary and compensation expense from $0 to $600,000, an increase in other income/expenses from a gain of $247,341 to a loss of $786,508, and offset by a an increase in revenues from $0 to $758,809.
General and administration expenses and professional fees for the nine month period ended December 31, 2013 amounted to $387,040 and $487,867 respectively, compared to $204,616 and $281,250 during the nine month period ended December 31, 2012. Research and development fees for the nine month period ended December 31, 2013 were $262,810 compared to $118,669 during the nine month period ended December 31, 2012. Directors fees, officer salary and compensation and other salary and compensation were $195,313, $600,000 and $66,556 respectively, compared to $281,250, $0 and $110,982 during the nine month period ended December 31, 2012.
Results of Operations for the Fiscal Years Ended March 31, 2012 and 2013
For the fiscal year ended March 31, 2013, we incurred a net loss of $2,035,864 and for the fiscal year ended March 31, 2012, we incurred a net loss of $2,323,551.
Revenues
Our revenues during the fiscal year ended March 31, 2013 totaled $2,168,093, compared to $1,300 in the fiscal year ended March 31, 2012. This increase in revenue was the result of the Company successfully completing its first commercial crop harvest in Vietnam.
Cost of Revenues
Cost of revenues during the fiscal year ended March 31, 2013 totaled $2,617,381, compared to $711,246 during the fiscal year ended March 31, 2012. The largest components of our cost of revenues are farm produce, which was $1,789,034 and farm management services – related parties, which was $712,550.
Gross Margin
Gross margin for the fiscal year ended March 31, 2013 was negative $449,288, compared to a negative $709,946 for the fiscal year ended March 31, 2012. The decrease was attributable to the significant increase in revenues during the fiscal year ended March 31, 2013.
General and Administrative Expenses, Salary and Compensation and Directors’ and Professional Fees
General and administration expenses for the fiscal year ended March 31, 2013, amounted to $412,409 compared to $113,742 in the fiscal year ended March 31, 2012. Research and development fees for the fiscal year ended March 31, 2013 were $177,169 compared to $206,191 for the fiscal year ended March 31, 2012. Salary and compensation expenses amounted to $190,549, directors’ fees amounted to $375,000 and professional fees amounted to $454,958 in the fiscal year ended March 31, 2013 compared to $0, $187,500 and $255,959 for the fiscal year ended March 31, 2012.
| 34 |
Liquidity and Capital Resources
As at December 31, 2013 we have $2,132,300 in current assets, and $2,591,869 in current liabilities. As at December 31, 2013 we have $85,366 in cash. As at December 31, 2013, our total assets were $3,641,781 and our total liabilities were $3,189,388. Our net working capital deficit as at December 31, 2013 was $459,569.
During the nine month period ended December 31, 2013, we used cash of $895,651 in operating activities and used cash of $16,475 in investing activities, respectively. During the nine month period ended December 31, 2013, we funded our operations from revenue from operations and the proceeds of private sales of convertible notes and the exercise proceeds of warrants. During the nine month period ended December 31, 2013, we raised $431,500 through the issuance of convertible notes and $152,012 through the proceeds of warrant exercises.
As of December 31, 2013, convertible promissory notes, net of discount, in the aggregate amount of $1,071,569 remained outstanding.
In July, 2012 outstanding convertible promissory notes in the principal amount of $500,000 were converted into an aggregate of 634,193 shares of our common stock.
On August 1, 2012, we entered into a Securities Purchase Agreement with certain accredited investors (the “Financing Stockholders”) to raise $500,000 in a private placement financing (the “Offering”). On August 6, 2012, after the satisfaction of certain closing conditions, the Offering closed and the Company issued to the Financing Stockholders: (i) an aggregate of 1,066,667 shares of the Company's common stock at a price per share of $0.46875 and (ii) warrants to purchase an equal number of shares of the Company's common stock at an exercise price of $0.6405 with a term of five (5) years, for gross proceeds of $500,000. Garden State Securities, Inc. (“GSS”) served as the placement agent for such equity financing. Per the engagement agreement signed between GSS and the Company on June 18, 2012, in consideration for services rendered as the placement agent, the Company agreed to: (i) pay GSS cash commissions equal to $40,000, or 8.0% of the gross proceeds received in the equity financing, and (ii) issue to GSS or its designee, a warrant to purchase up to 85,333 shares of the Company's common stock representing 8% of the Shares sold in the Offering) with an exercise price of $0.6405 per share and a term of five (5) years. Pursuant to the anti-dilution adjustment provision included in the Offering, the total share amount under the Cranshire Warrant has been increased to 2,036,381 and the exercise price has been reduced to $0.0671 as a result of certain other offerings of the Company. We may receive gross proceeds of up to $136,640.40 upon the cash exercise of the Cranshire Warrants. Any such proceeds we receive will be used for working capital and general corporate matters.
On May 3, 2013, in consideration for the immediate cash exercise of outstanding warrants to purchase 853,333 shares of common stock of the Company at a price per share of $0.20, the Company issued the Anson Warrants which are included in this registration statement. The warrant to purchase 1,877,333 shares of common stock is subject to a right of repurchase by the Company upon the satisfaction of certain conditions, at a price of $0.001 per warrant share. The warrant to purchase 2,346,666 shares is only exercisable upon the investor’s exercise in full of the warrant to purchase 1,877,333 shares. We will not receive any proceeds from the sale of those shares of common stock. We may, however, receive gross proceeds of up to $1,228,799.60 upon the cash exercise of the Anson Warrants. Any such proceeds we receive will be used for working capital and general corporate matters.
On July 16, 2013, the Company entered into a $400,000 Promissory Note (the “June 2013 Note”) with an accredited investor (the “Investor”) whereby the Investor agreed to loan to the Company up to $400,000 pursuant to the terms of the June 2013 Note. The June 2013 Note provides for the first $100,000 to be advanced upon closing and additional amounts will be advanced at the Investor’s sole discretion. Each advance is subject to a 10% original issue discount, such that the total amount which may actually be received by the Company pursuant to the June 2013 Note is only $360,000. The maturity date for each advance made under the June 2013 Note is one year from the date of such advance. If the Company repays the June 2013 Note on or before 90 days from the effective date, the interest rate shall be 0%, otherwise a one-time interest charge of l2% shall be applied to the principal sum. The June 2013 Notes are convertible into common stock of the Company on a cashless basis at any time, at a conversion price equal to the lesser of $0.26 or 65% of the lowest trade price in the 25 trading days prior to the conversion. If the conversion shares are not deliverable by DWAC an additional 10% discount will apply, and if the shares are ineligible for deposit into the DTC system and only eligible for Xclearing deposit an additional 5% discount will apply. So long as the June 2013 Note is outstanding, upon any issuance by the Company or any of its subsidiaries of any security with any term more favorable to the holder of such security or with a term in favor of the holder of such security that was not similarly provided to the Investor in the June 2013 Note, then the Company shall notify the Investor of such additional or more favorable term and such term, at the Investor's option, shall become a part of the transaction documents with the Company.
On August 22, 2013, we issued a convertible promissory note to Asher Enterprises, Inc. in the principal amount of $153,500 (the “Asher Note”), pursuant to the terms of a Securities Purchase Agreement. The Note matures on May 26, 2014, incurs interest at the rate of 8% per annum, and is convertible into shares of our common stock at a 35% discount to the average of the lowest three trading prices for our common stock during the 30 day trading period prior to the conversion date.
On September 26, 2013, we issued a convertible note in the principal amount of $27,778 with a 10% original issuance discount and a one-time interest charge of 12%. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date.
| 35 |
On October 15, 2013, we issued a Convertible Debenture in the principal amount of $58,000 (the “Debenture”), to Black Mountain Equities, Inc. (“Black Mountain”). On March 31, 2014, the Debenture was converted in full into 1,119,299 shares of common stock. The Debenture provides that on the next registration statement the Company files, the Company will include the shares issuable upon conversion of the Debenture. Black Mountain also received a warrant to purchase 1,000,000 shares of our common stock with an exercise price of $0.25 per share, subject to adjustment, and a term of five years.
On November 21, 2013, we issued a convertible note in the principal amount of $53,000, convertible at 65% of the three lowest bids for 30 trading days before the conversion date with interest at 8% per annum, due on August 25, 2014.
On December 9, 2013, we issued a convertible note in the principal amount of $55,556 with a 10% original issuance discount and 12% one time interest. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date.
Subsequent to the three months ended December 31, 2013, on February 7, 2014, we issued a Convertible Debenture to an investor in the principal amount of $80,000. The Convertible Debenture matures on February 6, 2015, incurs interest at the rate of 8% per annum, and is convertible into shares of our common stock at a conversion price of $0.10 per share.
Subsequent to the three months ended December 31, 2013, on February 13, 2014, an investor exercised warrants to purchase 1,877,333 shares of the Company’s common stock with an exercise price of $0.0585 per share, yielding aggregate gross proceeds to the Company of $109,824 in cash.
Subsequent to the three months ended December 31, 2013, on February 20, 2014, an investor exercised warrants to purchase 683,202 shares of the Company’s common stock with an exercise price of $0.053365 per share, yielding aggregate gross proceeds to the Company of $36,459 in cash.
Subsequent to the three months ended December 31, 2013, on February 24, 2014, an investor exercised warrants to purchase 1,413,326 shares of the Company’s common stock with an exercise price of $0.053365 per share, yielding aggregate gross proceeds to the Company of $75,422 in cash.
Subsequent to the three months ended December 31, 2013, on February 26, 2014, the Company converted $893,579 of working capital advances, provided to the Company by George Blankenbaker and his affiliated companies, into an aggregate of 16,744,682 shares of common stock.
Subsequent to the three months ended December 31, 2013, on February 28, 2014, an investor exercised warrants to purchase 250,000 shares of the Company’s common stock with an exercise price of $0.053365 per share, yielding aggregate gross proceeds to the Company of $13,341 in cash.
Subsequent to the three months ended December 31, 2013, on March 3, 2014, an investor exercised warrants to purchase 1,750,006 shares of the Company’s common stock with an exercise price of $0.053365 per share, yielding aggregate gross proceeds to the Company of $93,389 in cash.
Subsequent to the three months ended December 31, 2013, on March 3, 2014, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with Nomis Bay Ltd., a Bermuda company (“Nomis Bay”). The Purchase Agreement provides that, upon the terms and subject to the conditions set forth therein, (i) Nomis Bay shall purchase from the Company on the Closing Date a senior convertible note with an initial principal amount of $500,000 (the “Initial Convertible Note”) for a purchase price of $340,000 (a 32% original issue discount) and (ii) the Company shall have the right to require Nomis Bay to purchase from the Company on or prior to the 10th trading day after the effective date of this registration statement an additional senior convertible note with an initial principal amount of $600,000 (the “Additional Convertible Note” and, together with the Initial Convertible Note, the “Convertible Notes”) for a purchase price of $600,000. The Initial Convertible Note matures on December 27, 2014 (subject to extension as provided in the Initial Convertible Note) and, in addition to the 32% original issue discount, accrues interest at the rate of 8% per annum. If issued, the Additional Convertible Note will mature on the date that is the 10-month anniversary of the date of issuance of the Additional Convertible Note (subject to extension as provided in the Initial Convertible Note) and will accrue interest at the rate of 8% per annum. The Initial Convertible Note is convertible at any time, in whole or in part, at Nomis Bay’s option into shares of the Company’s common stock, par value $0.001 per share (the “Common Stock”), at a conversion price equal to the lesser of (i) the product of (x) the arithmetic average of the lowest three (3) volume weighted average prices of the Common Stock during the 10 consecutive trading days ending and including the trading day immediately preceding the applicable conversion date and (y) 40% (the “Variable Conversion Price”), and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). If issued, the Additional Convertible Note will be convertible at any time, in whole or in part, at Nomis Bay’s option into shares of Common Stock at a conversion price that will be equal to the lesser of (i) the Variable Conversion Price and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). At no time will Nomis Bay be entitled to convert any portion of the Convertible Notes to the extent that after such conversion, Nomis Bay (together with its affiliates) would beneficially own more than 4.99% of the outstanding shares of Common Stock as of such date. The Company has the right at any time to redeem all, but not less than all, of the total outstanding amount then remaining under the Initial Convertible Note and/or the Additional Convertible Note in cash at a price equal to 140% of the total amount of such Convertible Note then outstanding.
We do not expect that our revenues from operations will be wholly sufficient to fund our operating plan, so we are currently seeking further financing and we believe that, along with our revenues, will provide sufficient working capital to fund our operations for at least the next six months. Changes in our operating plans, increased expenses, acquisitions, or other events, may cause us to seek additional equity or debt financing in the future.
| 36 |
Our current cash requirements are significant due to the planned development and expansion of our business. The successful implementation of our business plan is dependent upon our ability to develop valuable intellectual property relating to stevia through our research programs, as well as our ability to develop and manage our own crop and aquaculture production operations. These planned research and agricultural development activities require significant cash expenditures. We do not expect to generate the necessary cash from our operations during the next 6 to 12 months to expand our business as desired. As such, in order to fund our operations during the next 6 to 12 months, we anticipate that we will have to raise additional capital through debt and/or equity financings, which may result in substantial dilution to our existing stockholders. There are no assurances that we will be able to raise the required working capital on terms favorable, or that such working capital will be available on any terms when needed. In addition, the terms of the Securities Purchase Agreement contain certain restrictions on our ability to engage in financing transactions. Specifically, for a period of two years after the effective date of the Securities Purchase Agreement, the Securities Purchase Agreement contains restrictions on certain types of financing transactions. The Securities Purchase Agreement contains carveouts to such financing restrictions for certain exempted transactions including (i) issuances pursuant to a stock option plan, (ii) securities issued upon the conversion of outstanding securities, (iii) securities issued pursuant to acquisitions or other strategic transactions, (iv) up to $500,000 in stock and warrants on the same terms as set forth in the Securities Purchase Agreement, and (v) up to $3,000,000 of the Company’s securities.
Contractual Obligations and Off-Balance Sheet Arrangements
As of March 31, 2013, the end of our latest fiscal year, we did not have any long-term debt or purchase obligations.
We have not entered into any other financial guarantees or other commitments to guarantee the payment obligations of any third parties. We have not entered into any derivative contracts that are indexed to our shares and classified as stockholder’s equity or that are not reflected in our consolidated financial statements. Furthermore, we do not have any retained or contingent interest in assets transferred to an unconsolidated entity that serves as credit, liquidity or market risk support to such entity. We do not have any variable interest in any unconsolidated entity that provides financing, liquidity, market risk or credit support to us or engages in leasing, hedging or research and development services with us.
Critical Accounting Estimates
The preparation of financial statements in conformity with generally accepted accounting principles of the United States (GAAP) requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the year. The more significant areas requiring the use of estimates include asset impairment, stock-based compensation, and future income tax amounts. Management bases its estimates on historical experience and on other assumptions considered to be reasonable under the circumstances. However, actual results may differ from the estimates.
The discussion and analysis of our financial condition and results of operations are based upon our financial statements, which have been prepared in accordance with GAAP. We believe certain critical accounting policies affect our more significant judgments and estimates used in the preparation of the financial statements. A description of our critical accounting policies is set forth in our Annual Report on Form 10-K for the fiscal year ended March 31, 2013, filed on July 16, 2013. As of, and for the three months ended December 31, 2013, there have been no material changes or updates to our critical accounting policies.
DIRECTORS AND EXECUTIVE OFFICERS
The following table sets forth the names and ages of our current directors and executive officers, the principal offices and positions held by each person:
| Person | Age | Position | ||
| George Blankenbaker | 48 | Director, President, Secretary and Treasurer | ||
| Dr. Pablo Erat | 42 | Director | ||
| Thomas Ong | 42 | Director |
The information below with respect to our directors includes such director’s experience, qualifications, attributes, and skills that led us to the conclusion that they should serve as a director.
George Blankenbaker - President, Secretary, Treasurer and Director
Mr. Blankenbaker became our President, Secretary, Treasurer and Director in June, 2011. Since November 2008, Mr. Blankenbaker has been leading the development of high Reb-A stevia farming in Vietnam. Mr. Blankenbaker was raised on a farm and became involved in large scale commercial agriculture in 2002 when he began working with the Agri-Food Veterinary Authority of Singapore (AVA) to provide strategically important food supplies to Singapore and has extensive experience managing agriculture projects in South East Asia. Mr. Blankenbaker received a Bachelors of Science in Business Finance from Indiana University in 1988, where he also studied Asian Political Science. Mr. Blankenbaker’s recent activities and experience in Vietnam have laid the groundwork for the Company’s current business strategy, and his in-depth knowledge of such matters are invaluable to our Board of Directors.
| 37 |
Dr. Pablo Erat - Director
Dr. Erat was elected to our board of directors on October 4, 2011. Since January 2009, Dr. Erat has served as CEO of Pal & Partners AG, a Swiss-based group domiciled in Zug with offices in Zurich and Mumbai and with a focus on the Indian agriculture industry. Prior to joining Pal & Partners AG, in 2008 Dr. Erat served as a consultant to corporations and start-up companies in various industries to assist in the development and implementation of innovative strategies. In April 2001, he co-founded Executive Insight, a strategy consulting firm and in January 2003, he co-founded DocsLogic, a company specialized on the development of knowledge applications, where he remained through 2007. Dr. Erat is also Assistant Professor at the ETH Zurich and regularly delivers speeches and workshops on strategic management principles for educational and business communities. Dr. Erat received a Doctorate from the University of St. Gallen in Switzerland in June 2003. Dr. Erat’s extensive knowledge and experience working for and advising early stage companies as well as his experience in the agriculture industry will be extremely relevant to the Board of Directors.
Thomas Ong - Director
Since November 1, 2011, Mr. Ong has served as our Director of Operations, Asia. Since November 6, 2009, Mr. Ong also serves as a Director of the Singapore registered farm management firm Growers Synergy Pte Ltd, an agriculture consultancy and farm management company producing and trading crops for the domestic and export markets. He is a member of the SPRING Start-up Enterprise Development Scheme (SPRING SEEDS) Investment Panel, a wholly owned subsidiary of SPRING Singapore, a statutory board under the Singapore Ministry of Trade and Industry, that provides equity-based co-financing options for Singapore-based early-stage companies. Prior to focusing on the food supply sector, Mr. Ong was a director of A.D. Venture Limited, a Singapore-registered fund investment and management company with operating arms in Hong Kong and the People's Republic of China (PRC). Previously, Mr. Ong served 5 years with the Ministry of the Environment and subsequently joined the National Environment Agency (NEA) and worked with the Economic Development Board (EDB), International Enterprise Singapore (IE Singapore), Workforce Development Agency (WDA) and related industry groups to promote high value environmental services to the domestic and international markets. Mr. Ong received his Bachelor of Business Administration from the National University of Singapore in 1995 and his Master of Science in Information Studies from Nanyang Technological University in 2000. Mr. Ong's familiarity with our operations specifically and Asian farm management generally will be of great value to our Board of Directors.
Involvement In Certain Legal Proceedings
No director, executive officer, significant employee or control person of the Company has been involved in any legal proceeding listed in Item 401(f) of Regulation S-K in the past 10 years.
Term of Office
Our directors are appointed for a one-year term to hold office until the next annual general meeting of our stockholders or until removed from office in accordance with our bylaws. Our officers are appointed by our Board of Directors and hold office until removed by the Board, absent an employment agreement.
EXECUTIVE COMPENSATION
Executive Compensation
The summary compensation table below shows certain compensation information for services rendered in all capacities to us by our principal executive officer and principal financial officer and by each other executive officer whose total annual salary and bonus exceeded $100,000 during the fiscal periods ended March 31, 2012 and March 31, 2013. Other than as set forth below, no executive officer’s total annual compensation exceeded $100,000 during our last fiscal period.
Summary Compensation Table
| Name and Principal Position (a) | Year (b) | Salary ($) (c) | Bonus ($) (d) | Stock Awards ($) (e) | Option Awards ($) (f) | Non Equity Incentive Plan Compensation ($) (g) | Non-qualified Deferred Compensation Earnings ($) (h) | All Other Compensation ($) (i) | Total ($) (j) | |||||||||||||||||||||||||||
| George Blankenbaker | 2013 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | |||||||||||||||||||
| President, Secretary, Treasurer, Director (Principal Executive Officer and Principal Financial Officer) | 2012 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 0 | $ | 750,000 | $ | 750,000 | |||||||||||||||||||
| 38 |
On June 23, 2011, as a result of the Share Exchange Agreement, the sole stockholder of Stevia Ventures International Ltd. (“Stevia Ventures”) received 12,000,000 shares of our common stock in exchange for 100% of the issued and outstanding common stock of Stevia Ventures. Mr. Blankenbaker, our President and director, was the sole stockholder and officer of Stevia Ventures. Accordingly, he was a recipient of 12,000,000 shares of our common stock issued in connection with the Share Exchange Transaction, 6,000,000 of which were to be held in escrow pending the achievement by the Company of certain business milestones (the “Escrow Shares”). On December 23, 2011, 3,000,000 of the 6,000,000 Escrow Shares were earned and released to Mr. Blankenbaker upon achievement of certain business objectives by the Company. Those shares were valued at $0.25 per share or $750,000 on the date of release and recorded as compensation. The remaining 3,000,000 Escrow Shares were earned and released from escrow on July 12, 2013 upon achievement of certain business objectives by the Company. Those shares were valued at $0.20 per share or $600,000 on the date of release and recorded as compensation.
Other than as set forth above, none of our executive officers received, nor do we have any arrangements to pay out, any bonus, stock awards, option awards, non-equity incentive plan compensation, or non-qualified deferred compensation.
Director Compensation
On October 14, 2011 we issued 1,500,000 shares to each of Rodney L. Cook and Pablo Erat, as newly appointed members of our Board of Directors, as compensation for future services. These shares shall vest with respect to 750,000 shares of restricted stock for each director on each of the first two anniversaries of the date of grant, subject to the director’s continuous service to the Company. These shares were valued at $0.25 per share, or an aggregate of $750,000, on the date of grant and are being amortized over the vesting period of two (2) years or $93,750 per quarter.
We recorded $187,500 in directors’ fees for the period from April 11, 2011 (inception) through March 31, 2012 and $375,000 in directors’ fees for the fiscal year ended March 31, 2013.
We have no standard arrangement to compensate directors for their services in their capacity as directors. Except as set forth above, directors are not paid for meetings attended. All travel and lodging expenses associated with corporate matters are reimbursed by us, if and when incurred.
Employment Agreements
None of our executive officers currently have employment agreements with us and the manner and amount of compensation for the above-referenced new officer and director has not yet been determined.
Potential Payments Upon Termination or Change-in-Control
We currently have no employment agreements with any of our executive officers, nor any compensatory plans or arrangements resulting from the resignation, retirement or any other termination of any of our executive officers, from a change-in-control, or from a change in any executive officer’s responsibilities following a change-in-control. As a result, we have omitted this table.
Compensation Committee Interlocks and Insider Participation
No interlocking relationship exists between our Board of Directors and the Board of Directors or compensation committee of any other company, nor has any interlocking relationship existed in the past.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT
The following table sets forth certain information as of March 26, 2014 with respect to the beneficial ownership of our common stock for (i) each director and officer, (ii) all of our directors and officers as a group, and (iii) each person known to us to own beneficially 5% or more of the outstanding shares of our common stock. To our knowledge, except as indicated in any footnotes to this table or pursuant to applicable community property laws, the persons named in the table have sole voting and investment power with respect to the shares of common stock indicated.
| 39 |
| Name and Address of Beneficial Owner (1) | Amount and Nature of Beneficial Ownership | Percentage of Class (2) | ||||||
| George Blankenbaker(3) | 52,244,682 | 35.79 | % | |||||
| President, Secretary, Treasurer, | ||||||||
| and Director | ||||||||
| 6451 Buck Creek Pkwy | ||||||||
| Indianapolis, IN 46227 | ||||||||
| Thomas Ong(4) | 5,000,000 | 3.42 | % | |||||
| Director | ||||||||
| 7117 US 31S | ||||||||
| Indianapolis, IN 46227 | ||||||||
| Pablo Erat | 1,500,000 | 1.03 | % | |||||
| Director | ||||||||
| Ludretikonerstrasse 53 | ||||||||
| 880 Thalwil | ||||||||
| Switzerland | ||||||||
| All Officers and Directors as a Group | 55,244,982 | 40.24 | % | |||||
(1) Beneficial ownership has been determined in accordance with Rule 13d-3 under the Exchange Act. Pursuant to the rules of the SEC, shares of common stock which an individual or group has a right to acquire within 60 days pursuant to the exercise of options or warrants are deemed to be outstanding for the purpose of computing the percentage ownership of such individual or group, but are not deemed to be beneficially owned and outstanding for the purpose of computing the percentage ownership of any other person shown in the table.
(2) Based on 145,972,713 shares of our common stock outstanding as of March 26, 2014.
(3) Mr. Blankenbaker is the beneficial owner of 52,244,982 shares of common stock. Mr. Blankenbaker owns 12,000,000 shares of common stock directly. 3,500,000 shares of common stock are owned by Growers Synergy Pte Ltd. (“Growers Synergy”). Mr. Blankenbaker is the managing director of Growers Synergy. Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and the Reporting Person controls a 49% interest in Growers Fresh. Mr. Blankenbaker may be deemed to be the indirect beneficial owner of the shares held by Growers Synergy under Rule 13d-3(a) promulgated under the Securities Exchange Act of 1934 (the “Exchange Act”). However, pursuant to Rule 13d-4 promulgated under the Exchange Act, Mr. Blankenbaker disclaims that he is a beneficial owner of such shares, except to the extent of his pecuniary interest herein. 36,744,682 shares of common stock are owned by Blankenbaker Ventures (Asia) Pte. Ltd. (“BV Asia”). Mr. Blankenbaker owns a 65% controlling interest in BV Asia.
(4) Mr. Ong is the beneficial owner of 5,000,000 shares of common stock. Mr. Ong owns 1,500,000 shares of common stock directly and 3,500,000 shares of common stock are owned by Growers Synergy. Mr. Ong, a director of the Company, is a director of Growers Synergy and is also a 25% shareholder of Agriventure Pte Ltd., which is a 49% shareholder of Growers Synergy. Mr. Ong may be deemed to be the indirect beneficial owner of the shares held by Growers Synergy under Rule 13d-3(a) promulgated under the Securities Exchange Act of 1934 (the “Exchange Act”). However, pursuant to Rule 13d-4 promulgated under the Exchange Act, Mr. Ong disclaims that he is a beneficial owner of such shares, except to the extent of his pecuniary interest herein.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
AND DIRECTOR INDEPENDENCE
Certain Relationships and Related Party Transactions
On June 23, 2011, as a result of the Share Exchange Agreement, the sole stockholder of Stevia Ventures International Ltd. (“Stevia Ventures”) received 12,000,000 shares of our common stock in exchange for 100% of the issued and outstanding common stock of Stevia Ventures. Mr. Blankenbaker, our President and director, was the sole stockholder and officer of Stevia Ventures. Accordingly, he was a recipient of 12,000,000 shares of our common stock issued in connection with the Share Exchange Transaction, 6,000,000 of which were to be held in escrow pending the achievement by the Company of certain business milestones (the “Escrow Shares”). On December 23, 2011, 3,000,000 of the 6,000,000 Escrow Shares were earned and released to Mr. Blankenbaker upon achievement of certain business objectives by the Company. Those shares were valued at $0.25 per share or $750,000 on the date of release and recorded as compensation. The remaining 3,000,000 Escrow Shares were earned and released from escrow on July 12, 2013 upon achievement of certain business objectives by the Company. Those shares were valued at $0.20 per share or $600,000 on the date of release and recorded as compensation.
On November 1, 2011, the Company entered into a Management and Off-Take Agreement (the "Management Agreement") with Growers Synergy Pte Ltd. ("Growers Synergy"), a Singapore corporation. Mr. Ong, a director of the Company, is a director of Growers Synergy and is also a 25% shareholder of Agriventure Pte Ltd., which is a 49% shareholder of Growers Synergy. Mr. Blankenbaker is the managing director of Growers Synergy. Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and Mr. Blankenbaker controls a 49% interest in Growers Fresh. Under the terms of the Management Agreement, the Company engaged Growers Synergy to supervise the Company's farm management operations, recommend quality farm management programs for stevia cultivation, assist in the hiring of employees and provide training to help the Company meet its commercialization targets, develop successful models to propagate future agribusiness services, and provide back-office and regional logistical support for the development of proprietary stevia farm systems in Vietnam, Indonesia and potentially other countries. Growers Synergy will provide services for a term of two (2) years from the date of signing, at $20,000 per month. The Management Agreement may be terminated by the Company upon 30 day notice. In connection with the Management Agreement, the parties agreed to enter into an off-take agreement whereby Growers Synergy agreed to purchase all of the non-stevia crops produced at the Company's Growers Synergy supervised farms. On July 5, 2012, the Company issued 500,000 shares of its common stock to Growers Synergy as consideration for services rendered by Growers Synergy to the Company. On February 26, 2014, the Company issued 3,000,000 shares of its common stock to Growers Synergy as consideration for services rendered by Growers Synergy to the Company.
| 40 |
On February 26, 2014, we issued 20,000,000 shares to Blankenbaker Ventures (Asia) Pte. Ltd., on behalf of George Blankenbaker, our president, director and stockholder in exchange for services to be rendered by Mr. Blankenbaker. 4,000,000 of the shares were fully vested at the time of grant and the remainder vest in four equal installments on each anniversary of February 26, 2014. Mr. Blankenbaker owns a 65% controlling interest in BV Asia.
On February 26, 2014, we issued 16,744,682 shares of our common stock to Blankenbaker Ventures (Asia) Pte. Ltd., on behalf of George Blankenbaker, our president, director and stockholder, in exchange for the cancellation of approximately $893,579.93 of working capital advances received from Mr. Blankenbaker and his affiliated companies.
Review, Approval or Ratification of Transactions with Related Persons
Although we have adopted a Code of Ethics, we still rely on our Board to review related party transactions on an ongoing basis to prevent conflicts of interest. Our Board reviews a transaction in light of the affiliations of the director, officer or employee and the affiliation’s of such person’s immediate family. Transactions are presented to our Board for approval before they are entered into or, if this is not possible, for ratification after the transaction has occurred. If our Board finds that a conflict of interest exists, then it will determine the appropriate remedial action, if any. Our Board approves or ratifies a transaction if it determines that the transaction is consistent with the best interests of the Company.
Director Independence
During the year ended March 31, 2013, we had two independent directors on our Board, Dr. Erat and Mr. Cook. Mr. Blankenbaker is not independent. We evaluate independence by the standards for director independence established by applicable laws, rules, and listing standards including, without limitation, the standards for independent directors established by The New York Stock Exchange, Inc., the NASDAQ National Market, and the SEC.
Subject to some exceptions, these standards generally provide that a director will not be independent if (a) the director is, or in the past three years has been, an employee of ours; (b) a member of the director’s immediate family is, or in the past three years has been, an executive officer of ours; (c) the director or a member of the director’s immediate family has received more than $120,000 per year in direct compensation from us other than for service as a director (or for a family member, as a non-executive employee); (d) the director or a member of the director’s immediate family is, or in the past three years has been, employed in a professional capacity by our independent public accountants, or has worked for such firm in any capacity on our audit; (e) the director or a member of the director’s immediate family is, or in the past three years has been, employed as an executive officer of a company where one of our executive officers serves on the compensation committee; or (f) the director or a member of the director’s immediate family is an executive officer of a company that makes payments to, or receives payments from, us in an amount which, in any twelve-month period during the past three years, exceeds the greater of $1,000,000 or 2% of that other company’s consolidated gross revenues.
DISCLOSURE OF COMMISSION POSITION OF
INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Sections 78.7502 and 78.751 of the Nevada Revised Statutes authorizes a court to award, or a corporation’s board of directors to grant indemnity to directors and officers in terms sufficiently broad to permit indemnification, including reimbursement of expenses incurred, under certain circumstances for liabilities arising under the Securities Act of 1933, as amended. In addition, the registrant’s Bylaws provide that the registrant has the authority to indemnify the registrant’s directors and officers and may indemnify the registrant’s employees and agents (other than officers and directors) against liabilities to the fullest extent permitted by Nevada law. The registrant is also empowered under the registrant’s Bylaws to purchase insurance on behalf of any person whom the registrant is required or permitted to indemnify.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling us pursuant to the foregoing provisions, or otherwise, we have been advised that in the opinion of the SEC, such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
WHERE YOU CAN FIND MORE INFORMATION
We have filed a registration statement on Form S-1, together with all amendments and exhibits, with the SEC. This Prospectus, which forms a part of that registration statement, does not contain all information included in the registration statement. Certain information is omitted and you should refer to the registration statement and its exhibits. With respect to references made in this Prospectus to any of our contracts or other documents, the references are not necessarily complete and you should refer to the exhibits attached to the registration statement for copies of the actual contracts or documents. You may read and copy any document that we file at the Commission’s Public Reference Room at 100 F Street, N.E., Washington, D.C. 20549. Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference rooms. Our filings and the registration statement can also be reviewed by accessing the SEC’s website at http://www.sec.gov. We maintain a website at http://www.steviacorp.us
| 41 |
FINANCIAL STATEMENTS
Our interim unaudited consolidated financial statements as of and for the three and nine months ended December 31, 2013 and 2012, and audited consolidated financial statements as of and for the fiscal year ended March 31, 2013 and for the period from April 11, 2011 (inception) to March 31, 2012 are included herewith.
| Contents | Page(s) |
| Report of Independent Registered Public Accounting Firm | F-2 |
| Consolidated Balance Sheets at March 31, 2013 and 2012 | F-3 |
| Consolidated Statements of Operations for the Fiscal Year Ended March 31, 2013 and for the Period from April 11, 2011 (Inception) through March 31, 2012 | F-4 |
| Consolidated Statement of Equity (Deficit) for the Fiscal Year Ended March 31, 2013 and for the Period from April 11, 2011 (Inception) through March 31, 2012 | F-5 |
| Consolidated Statements of Cash Flows for the Fiscal Year Ended March 31, 2013 and for the Period from April 11, 2011 (Inception) through March 31, 2012 | F-6 |
| Notes to the Consolidated Financial Statements | F-7 |
| March 31, 2013 | March 31, 2012 | |||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash | $ | 424,475 | $ | 15,698 | ||||
| Accounts receivable | 158,008 | - | ||||||
| Prepayments and other current assets | 33,096 | 168,874 | ||||||
Total current assets | 615,579 | 184,572 | ||||||
| Non-current assets: | ||||||||
| Property and equipment | 7,925 | 3,036 | ||||||
| Accumulated depreciation | (1,234 | ) | - | |||||
| Property and equipment, net | 6,691 | 3,036 | ||||||
| Acquired technology | 1,635,300 | - | ||||||
| Accumulatd amortization | (81,765 | ) | - | |||||
| Acquired technology, net | 1,553,535 | - | ||||||
| Website development costs | 5,315 | 5,315 | ||||||
| Accumulated amortization | (1,869 | ) | (801 | ) | ||||
| Website development costs, net | 3,446 | 4,514 | ||||||
| Security deposit | 15,000 | 15,000 | ||||||
| Total assets | $ | 2,194,251 | $ | 207,122 | ||||
| Liabilities and equity (deficit) | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 948,073 | $ | 237,288 | ||||
| Accounts payable - president and CEO | 89,193 | 20,220 | ||||||
| Accrued expenses | 19,700 | 5,400 | ||||||
| Accrued interest | 21,627 | 15,521 | ||||||
| Advances from president and significant stockholder | 21,238 | 19,138 | ||||||
| Convertible notes payable - net of discount | 357,700 | 700,000 | ||||||
| Current portion of derivative liability | - | - | ||||||
Total current liabilities | 1,457,531 | 997,567 | ||||||
| Non-Current liabilities: | ||||||||
| Derivative liability | 486,113 | - | ||||||
Total non-current liabilities | 486,113 | - | ||||||
| Total liabilities | 1,943,644 | 997,567 | ||||||
| Equity (Deficit) | ||||||||
| Stevia Corp stockholders' equity (deficit): | ||||||||
| Preferred stock at $0.001 par value: 1,000,000 shares authorized; | ||||||||
| none issued or outstanding | - | - | ||||||
| Common stock at $0.001 par value: 100,000,000 shares authorized, | ||||||||
| 63,555,635 and 58,354,775 shares issued and outstanding, respectively | 63,556 | 58,355 | ||||||
| Additional paid-in capital | 4,760,624 | 1,474,751 | ||||||
| Accumulated deficit | (4,359,415 | ) | (2,323,551 | ) | ||||
Total Stevia Corp stockholders' equity (deficit) | 464,765 | (790,445 | ) | |||||
| Non-controlling interest in subsidiary | (214,158 | ) | - | |||||
| Total Equity (Deficit) | 250,607 | (790,445 | ) | |||||
| Total Liabilities and Equity (Deficit) | $ | 2,194,251 | $ | 207,122 | ||||
| For the Period from | |||||||||
| For the Fiscal Year | April 11, 2011 | ||||||||
| Ended | (inception) through | ||||||||
| March 31, 2013 | March 31, 2012 | ||||||||
| Revenues | $ | 2,168,093 | $ | 1,300 | |||||
| Cost of revenues | |||||||||
| Farm produce | 1,789,034 | - | |||||||
| Farm expenses | 94,547 | 523,746 | |||||||
| Farm field lease | 21,250 | 7,500 | |||||||
| Farm management services - related parties | 712,550 | 180,000 | |||||||
| Total cost of revenues | 2,617,381 | 711,246 | |||||||
| Gross margin | (449,288) | (709,946) | |||||||
| Operating expenses: | |||||||||
| Directors' fees | 375,000 | 187,500 | |||||||
| Professional fees | 454,958 | 255,959 | |||||||
| Research and development | 177,169 | 206,191 | |||||||
| Salary and compensation - officer | - | 750,000 | |||||||
| Salary and compensation - others | 190,549 | - | |||||||
| General and administrative expenses | 412,409 | 113,742 | |||||||
| Total operating expenses | 1,610,085 | 1,513,392 | |||||||
| Loss from operations | (2,059,373) | (2,223,338) | |||||||
| Other (income) expense: | |||||||||
| Change in fair value of derivative liability | 74,308 | - | |||||||
| Financing cost | 28,625 | 70,500 | |||||||
| Foreign currency transaction gain (loss) | 1,316 | - | |||||||
| Interest expense | 86,400 | 29,757 | |||||||
| Interest income | - | (44) | |||||||
| Total other (income) expense | 190,649 | 100,213 | |||||||
| Loss before income tax provision and non-controlling interest | (2,250,022) | (2,323,551) | |||||||
| Income tax provision | - | - | |||||||
| Net loss before non-controlling interest | (2,250,022) | (2,323,551) | |||||||
| Net loss attributable to the non-controlling interest | (214,158) | - | |||||||
| Net loss attributable to Stevia Corp. | $ | (2,035,864) | $ | (2,323,551) | |||||
| Net loss per common share | |||||||||
| - Basic and diluted: | $ | (0.03) | $ | (0.05) | |||||
| Weighted average common shares outstanding | |||||||||
| - basic and diluted | 62,092,487 | 45,093,271 | |||||||
| Total STEV | ||||||||||||||||||||||||||||
| Common Stock, $0.001 Par Value | Additional | Accumulated | Stockholders' | Non-controlling | Total | |||||||||||||||||||||||
| Number of Shares | Amount | paid-in Capital | Deficit | Equity (Deficit) | Interest | Equity (Deficit) | ||||||||||||||||||||||
| Balance, April 11, 2011 (inception) | 6,000,000 | $ | 6,000 | $ | (5,900 | ) | $ | - | $ | 100 | $ | - | $ | 100 | ||||||||||||||
| Common shares deemed issued in reverse acquisition | 79,800,000 | 79,800 | (198,088 | ) | (118,288 | ) | (118,288 | ) | ||||||||||||||||||||
| Common shares cancelled in reverse acquisition | (33,000,000 | ) | (33,000 | ) | 33,000 | - | - | |||||||||||||||||||||
| Common shares issued for cash at $0.25 per share | ||||||||||||||||||||||||||||
| on October 4, 2011 | 400,000 | 400 | 99,600 | 100,000 | 100,000 | |||||||||||||||||||||||
| Common shares issued for notes conversion | ||||||||||||||||||||||||||||
| at $0.25 per share on October 4, 2011 | 1,400,000 | 1,400 | 348,600 | 350,000 | 350,000 | |||||||||||||||||||||||
| Common shares issued for conversion of accrued interest | ||||||||||||||||||||||||||||
| at $0.25 per share on October 4, 2011 | 74,850 | 75 | 18,638 | 18,713 | 18,713 | |||||||||||||||||||||||
| Common shares cancelled by significant stockholder | ||||||||||||||||||||||||||||
| on October 4, 2011 | (3,000,000 | ) | (3,000 | ) | 3,000 | - | - | |||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||
| services on October 4, 2011 | 3,000,000 | 3,000 | 747,000 | 750,000 | 750,000 | |||||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||
| services on October 4, 2011 | (750,000 | ) | (750,000 | ) | (750,000 | ) | ||||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||
| services on October 4, 2011 | ||||||||||||||||||||||||||||
| earned during the period | 187,500 | 187,500 | 187,500 | |||||||||||||||||||||||||
| Make good shares released to officer for achieving | ||||||||||||||||||||||||||||
| the first milestone on December 23, 2011 | 3,000,000 | 3,000 | 747,000 | 750,000 | 750,000 | |||||||||||||||||||||||
| Common shares issued for notes conversion | ||||||||||||||||||||||||||||
| at $0.25 per share on January 18, 2012 | 600,000 | 600 | 149,400 | 150,000 | 150,000 | |||||||||||||||||||||||
| Common shares issued for conversion of accrued interest | ||||||||||||||||||||||||||||
| at $0.25 per share on January 18, 2012 | 17,425 | 17 | 4,339 | 4,356 | 4,356 | |||||||||||||||||||||||
| Common shares issued for financing services upon agreement | ||||||||||||||||||||||||||||
| at $1.50 per share on January 26, 2012 | 35,000 | 35 | 52,465 | 52,500 | 52,500 | |||||||||||||||||||||||
| Common shares issued for consulting services | ||||||||||||||||||||||||||||
| at $1.39 per share on March 31, 2012 | 27,500 | 28 | 38,197 | 38,225 | 38,225 | |||||||||||||||||||||||
| Net loss | (2,323,551 | ) | (2,323,551 | ) | (2,323,551 | ) | ||||||||||||||||||||||
| Balance, March 31, 2012 | 58,354,775 | 58,355 | 1,474,751 | (2,323,551 | ) | (790,445 | ) | - | (790,445 | ) | ||||||||||||||||||
| Restricted common shares issued for farm management services to a relatd party | ||||||||||||||||||||||||||||
valued at $0.79 per share discounted at 69% on July 5, 2012 | 500,000 | 500 | 272,050 | 272,550 | 272,550 | |||||||||||||||||||||||
| Restricted common shares issued for technology rights | ||||||||||||||||||||||||||||
valued at $0.79 per share discounted at 69% on July 5, 2012 | 3,000,000 | 3,000 | 1,632,300 | 1,635,300 | 1,635,300 | |||||||||||||||||||||||
| Common shares issued for notes conversion | ||||||||||||||||||||||||||||
| at $0.832143 per share on July 6, 2012 | 600,858 | 601 | 499,399 | 500,000 | 500,000 | |||||||||||||||||||||||
| Common shares issued for conversion of accrued interest | ||||||||||||||||||||||||||||
| at $0.832143 per share on July 6, 2012 | 33,335 | 33 | 27,707 | 27,740 | 27,740 | |||||||||||||||||||||||
| Common shares and warrants issued to two investors for cash | ||||||||||||||||||||||||||||
| at $0.46875 per unit on August 6, 2012 | 1,066,667 | 1,067 | 498,933 | 500,000 | 500,000 | |||||||||||||||||||||||
| Warrants issued to investors in connection with the sale of | ||||||||||||||||||||||||||||
| equity units on August 6, 2012 classified as derivative liability | (381,300 | ) | (381,300 | ) | (381,300 | ) | ||||||||||||||||||||||
| Commissions and legal fees paid in connection with the sale of | ||||||||||||||||||||||||||||
| equity units on August 6, 2012 | (52,500 | ) | (52,500 | ) | (52,500 | ) | ||||||||||||||||||||||
| Warrants issued to placement agent in connection with the sale of | ||||||||||||||||||||||||||||
| equity units on August 6, 2012 classified as derivative liability | (30,504 | ) | (30,504 | ) | (30,504 | ) | ||||||||||||||||||||||
| Issuance of warrants in connection with | ||||||||||||||||||||||||||||
| convertible note payable issued in February and March 2013 | 220,438 | 220,438 | 220,438 | |||||||||||||||||||||||||
| Beneficial conversion feature in connection with | ||||||||||||||||||||||||||||
| convertible note payable issued in February and March 2013 | 224,350 | 224,350 | 224,350 | |||||||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||
services on October 4, 2011 earned during the period | 375,000 | 375,000 | 375,000 | |||||||||||||||||||||||||
| Net loss | (2,035,864 | ) | (2,035,864 | ) | (214,158 | ) | (2,250,022 | ) | ||||||||||||||||||||
| Balance, March 31, 2013 | 63,555,635 | $ | 63,556 | $ | 4,760,624 | $ | (4,359,415 | ) | $ | 464,765 | $ | (214,158 | ) | $ | 250,607 | |||||||||||||
| For the Period from | ||||||||
| For the Fiscal Year | April 11, 2011 | |||||||
| Ended | (inception) through | |||||||
| March 31, 2013 | March 31, 2012 | |||||||
| Cash flows from operating activities: | ||||||||
| Adjustments to reconcile net loss to net cash used in operating activities | ||||||||
| Depreciation expense | 1,234 | - | ||||||
| Amortization expense - acquired technology | 81,765 | - | ||||||
| Amortization expense - website development costs | 1,068 | 801 | ||||||
| Discount of convertible notes payable | (412,738 | ) | - | |||||
| Change in fair value of derivative liability | 74,308 | - | ||||||
| Common shares issued for compensation | - | 750,000 | ||||||
| Common shares issued for director services earned during the period | 375,000 | 187,500 | ||||||
| Common shares issued for services-related party | 272,550 | - | ||||||
| Common shares issued for outside services | - | 90,725 | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | (158,008 | ) | - | |||||
| Prepaid expenses | 135,778 | (168,874 | ) | |||||
| Accounts payable | 710,785 | 141,530 | ||||||
| Accounts payable - President and CEO | 68,973 | 20,220 | ||||||
| Accrued expenses | 14,300 | (1,290 | ) | |||||
| Accrued interest | 54,284 | 38,589 | ||||||
| Net cash used in operating activities | (1,030,723 | ) | (1,279,350 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Purchases of property, plant and equipment | (4,889 | ) | (3,036 | ) | ||||
| Website development costs | - | (5,315 | ) | |||||
| Cash received from reverse acquisition | - | 3,199 | ||||||
| Net cash used in investing activities | (4,889 | ) | (5,152 | ) | ||||
| Cash flows from financing activities: | ||||||||
| Advances from president and stockholder | 2,100 | 200 | ||||||
| Proceeds from issuance of convertible notes | 550,000 | 1,200,000 | ||||||
| Proceeds from sale of common stock, net of costs | 892,289 | 100,000 | ||||||
| Net cash provided by financing activities | 1,444,389 | 1,300,200 | ||||||
| Net change in cash | 408,777 | 15,698 | ||||||
| Cash at beginning of the fiscal year | 15,698 | - | ||||||
| Cash at end of the fiscal year | $ | 424,475 | $ | 15,698 | ||||
| Supplemental disclosure of cash flows information: | ||||||||
| Interest paid | $ | - | $ | - | ||||
| Income tax paid | $ | - | $ | - | ||||
| Non-cash investing and financing activities: | ||||||||
| Issuance of common stock for conversion of convertible notes | $ | 500,000 | $ | 500,000 | ||||
| Converstion of accrued interest to convertible notes | $ | 20,438 | $ | - | ||||
| Issuance of common stock for conversion of accrued interest | $ | 27,740 | $ | 23,068 | ||||
Stevia Corp.
Name of consolidated subsidiary or entity | State or other jurisdiction of incorporation or organization | Date of incorporation or formation (date of acquisition, if applicable) | Attributable interest | |||
| Stevia Ventures International Ltd. | The Territory of the British Virgin Islands | April 11, 2011 | 100% | |||
| Stevia Asia Limited | Hong Kong SAR | March 19, 2012 | 100% | |||
| Stevia Technew Limited | Hong Kong SAR | April 28, 2012 | 70% |
| Level 1 | Quoted market prices available in active markets for identical assets or liabilities as of the reporting date. | |
| Level 2 | Pricing inputs other than quoted prices in active markets included in Level 1, which are either directly or indirectly observable as of the reporting date. | |
| Level 3 | Pricing inputs that are generally observable inputs and not corroborated by market data. |
| · | Expected term of share options and similar instruments: The expected life of options and similar instruments represents the period of time the option and/or warrant are expected to be outstanding. Pursuant to Paragraph 718-10-50-2(f)(2)(i) of the FASB Accounting Standards Codification the expected term of share options and similar instruments represents the period of time the options and similar instruments are expected to be outstanding taking into consideration of the contractual term of the instruments and employees’ expected exercise and post-vesting employment termination behavior into the fair value (or calculated value) of the instruments. Pursuant to paragraph 718-10-S99-1, it may be appropriate to use thesimplified method,i.e.,expected term = ((vesting term + original contractual term) / 2), if (i) A company does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term due to the limited period of time its equity shares have been publicly traded; (ii) A company significantly changes the terms of its share option grants or the types of employees that receive share option grants such that its historical exercise data may no longer provide a reasonable basis upon which to estimate expected term; or (iii) A company has or expects to have significant structural changes in its business such that its historical exercise data may no longer provide a reasonable basis upon which to estimate expected term. The Company uses the simplified method to calculate expected term of share options and similar instruments as the company does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term. |
| · | Expected volatility of the entity’s shares and the method used to estimate it. Pursuant to ASC Paragraph 718-10-50-2(f)(2)(ii) a thinly-traded or nonpublic entity that uses the calculated value method shall disclose the reasons why it is not practicable for the Company to estimate the expected volatility of its share price, the appropriate industry sector index that it has selected, the reasons for selecting that particular index, and how it has calculated historical volatility using that index. The Company uses the average historical volatility of the comparable companies over the expected contractual life of the share options or similar instruments as its expected volatility. If shares of a company are thinly traded the use of weekly or monthly price observations would generally be more appropriate than the use of daily price observations as the volatility calculation using daily observations for such shares could be artificially inflated due to a larger spread between the bid and asked quotes and lack of consistent trading in the market. |
| · | Expected annual rate of quarterly dividends. An entity that uses a method that employs different dividend rates during the contractual term shall disclose the range of expected dividends used and the weighted-average expected dividends. The expected dividend yield is based on the Company’s current dividend yield as the best estimate of projected dividend yield for periods within the expected term of the share options and similar instruments. |
| · | Risk-free rate(s). An entity that uses a method that employs different risk-free rates shall disclose the range of risk-free rates used. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for periods within the expected term of the share options and similar instruments. |
| · | Expected term of share options and similar instruments: Pursuant to Paragraph 718-10-50-2 of the FASB Accounting Standards Codification the expected term of share options and similar instruments represents the period of time the options and similar instruments are expected to be outstanding taking into consideration of the contractual term of the instruments and holder’s expected exercise behavior into the fair value (or calculated value) of the instruments. The Company uses historical data to estimate holder’s expected exercise behavior. If the Company is a newly formed corporation or shares of the Company are thinly traded the contractual term of the share options and similar instruments is used as the expected term of share options and similar instruments as the Company does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term. |
| · | Expected volatility of the entity’s shares and the method used to estimate it. An entity that uses a method that employs different volatilities during the contractual term shall disclose the range of expected volatilities used and the weighted-average expected volatility. A thinly-traded or nonpublic entity that uses the calculated value method shall disclose the reasons why it is not practicable for the Company to estimate the expected volatility of its share price, the appropriate industry sector index that it has selected, the reasons for selecting that particular index, and how it has calculated historical volatility using that index. The Company uses the average historical volatility of the comparable companies over the expected contractual life of the share options or similar instruments as its expected volatility. If shares of a company are thinly traded the use of weekly or monthly price observations would generally be more appropriate than the use of daily price observations as the volatility calculation using daily observations for such shares could be artificially inflated due to a larger spread between the bid and asked quotes and lack of consistent trading in the market. |
| · | Expected annual rate of quarterly dividends. An entity that uses a method that employs different dividend rates during the contractual term shall disclose the range of expected dividends used and the weighted-average expected dividends. The expected dividend yield is based on the Company’s current dividend yield as the best estimate of projected dividend yield for periods within the expected contractual life of the option and similar instruments. |
| · | Risk-free rate(s). An entity that uses a method that employs different risk-free rates shall disclose the range of risk-free rates used. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for periods within the contractual life of the option and similar instruments. |
| Potentially Outstanding DilutiveCommon Shares | ||||||||
For Fiscal Year Ended March 31, 2013 | For the Period from April 11, 2011 (inception) through March 31, 2012 | |||||||
| Make Good Escrow Shares | ||||||||
| Make Good Escrow Agreement shares issued and held with the escrow agent in connection with the Share Exchange Agreement consummated on June 23, 2011 pending the achievement by the Company of certain post-Closing business milestones (the “Milestones”). | 3,000,000 | 6,000,000 | ||||||
| Sub-total Make Good Escrow Shares | 3,000,000 | 6,000,000 | ||||||
| Convertible Note Shares | ||||||||
| On March 7, 2012, the Company issued a convertible note in the amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the next private placement price on a per share basis, provided that the Company completes a private placement with gross proceeds of at least $100,000. On August 6, 2012, the Company completed the very next private placement at $0.46875 per share with gross proceeds of at least $100,000. On March 15, 2013, the above note was cancelled and reissued with a new convertible note consisting of the prior principal amount and all the accrued unpaid interest for a total amount of $220,438 with interest at 12% per annum due on September 30, 2013 with the conversion price is $0.25 per share. | 881,752 | - | ||||||
| On May 30, 2012, the Company issued a convertible note in the amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the next private placement price on a per share basis, provided that the Company completes a private placement with gross proceeds of at least $100,000. On August 6, 2012, the Company completed the very next private placement at $0.46875 per share with gross proceeds of at least $100,000. | 426,667 | - | ||||||
| On February 26, 2013 , the Company issued two (2) convertible notes in the amount of $250,000 and $100,000, respectively, with interest at 12% per annum due on September 30, 2013 with the conversion price at $0.25 per share | 1,400,000 | - | ||||||
| Sub-total Convertible Note Shares | 2,708,419 | - | ||||||
| Warrant Shares | ||||||||
On August 6, 2012, the Company issued (i) warrants to purchase 1,066,667 shares, in the aggregate, of the Company’s common stock to the investors (the “investors warrants”) and (ii) warrants to purchase 85,333 shares of the Company's common stock to the placement agent (the "agent warrants") with an exercise price of $0.6405 per share subject to certain adjustments pursuant to Section 3(b) Subsequent Equity Sales of the SPA expiring five (5) years from the date of issuance. On February 26, 2013, thenew warrants issuedtriggered a reset of the above warrants exercise price to $0.25 per share and the shares to be issued under the warrants were adjusted accordingly. | 2,951,424 | - | ||||||
| On February 26, 2013, the Company issued warrants to purchase 1,000,000 and 400,000 shares respectively, 1,400,000 shares in the aggregate, of the Company’s common stock to two notes holders in connection with the issuance of convertible notes. | 1,400,000 | - | ||||||
| On March 15, 2013, the Company issued a warrant to purchase 881,753 shares of the Company’s common stock to the note holder in connection with the issuance of the convertible note. | 881,753 | |||||||
| Sub-total Warrant Shares | 5,233,177 | - | ||||||
| Total potentially outstanding dilutive common shares | 10,941,596 | 6,000,000 |
| March 31, 2013 | March 31, 2012 | |||||||
| Prepaid research and development | $ | 25,546 | $ | 128,445 | ||||
| Prepaid rent | 6,667 | 21,250 | ||||||
| Retainer | 451 | 15,000 | ||||||
| Other | 432 | 4,179 | ||||||
| $ | 33,096 | $ | 168,874 | |||||
| Estimated Useful Life (Years) | March 31, 2013 | March 31, 2012 | |||||||
| Property and equipment | 5 | $ | 7,925 | $ | 3,036 | ||||
| Less accumulated depreciation | (1,234 | ) | (- | ) | |||||
| $ | 6,691 | $ | 3,036 | ||||||
| Estimated Useful Life (Years) | March 31, 2013 | March 31, 2012 | |||||||
| Technology right | 15 | $ | 1,635,300 | $ | - | ||||
| Less accumulated amortization | (81,765 | ) | (- | ) | |||||
| $ | 1,553,535 | $ | - | ||||||
| Estimated Useful Life (Years) | March 31, 2013 | March 31, 2012 | |||||||
| Website development costs | 5 | $ | 5,315 | $ | 5,315 | ||||
| Accumulated amortization | (1,869 | ) | (801 | ) | |||||
| $ | 3,446 | $ | 4,514 | ||||||
| Related Parties | Relationship | |
| George Blankenbaker | President and significant stockholder of the Company | |
| Leverage Investments LLC | An entity owned and controlled by the president and significant stockholder of the Company | |
| Technew Technology Limited | Non-controlling interest holder | |
| Growers Synergy Pte Ltd. | An entity owned and controlled by the president and significant stockholder of the Company | |
| Guangzhou Health Technology Development Company Limited | An entity owned and controlled by Non-controlling interest holder |
For the Fiscal Year Ended March 31, 2013 | For the Period from April 11, 2011 (inception) through March 31, 2012 | |||||||
| Farm management services – related parties | $ | 240,000 | $ | 180,000 | ||||
| $ | 240,000 | $ | 180,000 | |||||
| Fiscal Year Ending March 31: | ||||
| 2014 | $ | 140,000 | ||
| $ | 140,000 | |||
| Expected option life (year) | 3.00 | |||
| Expected volatility | 74.53 | % | ||
| Risk-free interest rate | 0.37 | % | ||
| Dividend yield | 0.00 | % | ||
| Expected option life (year) | 3.00 | |||
| Expected volatility | 75.11 | % | ||
| Risk-free interest rate | 0.40 | % | ||
| Dividend yield | 0.00 | % | ||
| March 31, 2013 | March 31, 2012 | |||||||||
| On November 16, 2011, the Company issued a convertible note in the amount of $250,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the private placement price on a per share basis provided the Company completes a private placement with gross proceeds of at least $100,000. On July 6, 2012, the note holder converted the entire principal of $250,000 and accrued interest through the date of conversion of $15,959 to 319,607 shares of the Company’s common stock at $0.83 per share. | $ | - | $ | 250,000 | ||||||
| On January 16, 2012, the Company issued a convertible note in the amount of $250,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the private placement price on a per share basis provided the Company completes a private placement with gross proceeds of at least $100,000. On July 6, 2012, the note holder converted the entire principal of $250,000 and accrued interest through the date of conversion of $11,781 to 314,586 shares of the Company’s common stock at $0.83 per share. | - | 250,000 | ||||||||
| On March 7, 2012, the Company issued a convertible note in the amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the next private placement price on a per share basis provided that the Company completes a private placement with gross proceeds of at least $100,000. On August 6, 2012, the Company completed the very next private placement at $0.46875 per share with gross proceeds of at least $100,000. On March 15, 2013, the above note was cancelled and reissued with a new convertible note consisting of the prior principal amount and all the accrued unpaid interest for a total amount of $220,438 with interest at 12% per annum due on September 30, 2013 with the conversion price $0.25 per share | 220,438 | 200,000 | ||||||||
| On May 30, 2012, the Company issued a convertible note in the amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the next private placement price on a per share basis provided that the Company completes a private placement with gross proceeds of at least $100,000. On August 6, 2012, the Company completed the very next private placement at $0.46875 per share with gross proceeds of at least $100,000. | 200,000 | - | ||||||||
| On February 26, 2013, the Company issued two (2) convertible notes in the amount of $250,000 and $100,000, respectively, for an aggregate of $350,000 with interest at 12% per annum, due on September 30, 2013, with the conversion price at $0.25 per share. In connection with the issuance of the convertible notes, the Company issued to both notes holders a warrant to purchase 1,000,000 shares and 400,000 shares, respectively, in the aggregate of 1,400,000 shares of the Company’s common stock. | 350,000 | - | ||||||||
| Sub-total: convertible notes payable | 770,438 | 700,000 | ||||||||
| Discount representing (i) the relative fair value of the warrants issued and (ii) the beneficial conversion features | (444,788 | ) | - | |||||||
| Accumulated amortization of discount on convertible notes payable | 32,050 | - | ||||||||
| Remaining discount | (412,738 | ) | - | |||||||
| $ | 357,770 | $ | 700,000 | |||||||
| · | The stock price would fluctuate with the Company projected volatility. |
| · | The stock price would fluctuate with an annual volatility. The projected volatility curve was based on historical volatilities of the Company for the valuation periods. |
| · | The Holder would exercise the warrant as they become exercisable (effective registration is projected 4 months from issuance and the earliest exercise is projected 180 days from issuance) at target prices of 2 times the higher of the projected reset price or stock price. |
| · | The Holder would exercise the warrant at maturity if the stock price was above the project reset prices. |
| · | A 100% probability of a reset event and a projected financing each quarter for 3 years at prices approximating 93% of market |
| · | The 2,732,801 Investor Warrants with an exercise price of $0.25 per share (1,066,667 Investor Warrants with an exercise price of $0.6405 per share prior to the occurrence of the February 26, 2013 reset event) is projected to reset to $0.095 at maturity; and the 218,623 Placement Agent Warrants with an exercise price of $0.25 per share (85,333 Placement Agent Warrants with an exercise price of $0.6405 per share prior to the occurrence of the February 26, 2013 reset event) is projected to reset to $0.095 at maturity. |
| · | No warrants have been exercised or expired. |
| · | The projected volatility curve for the valuation dates was: |
| 1 Year | 2 Tear | 3 Year | 4 Year | 5 Year | |||||||||||
| August 6, 2012 | 129% | 178% | 218% | 252% | 281% | ||||||||||
| September 30, 2012 | 127% | 173% | 211% | 244% | 272% | ||||||||||
| March 31, 2013 | 122% | 167% | 205% | 236% | 264% |
| Fair Value Measurement Using Level 3 Inputs | ||||||||
| Derivative warrants | ||||||||
| Assets (Liability) | Total | |||||||
| Balance, August 6, 2012 | $ | (411,805 | ) | $ | (411,805 | ) | ||
| Total gains or losses (realized/unrealized) included in: | ||||||||
| Net income (loss) | 231,521 | 231,521 | ||||||
| Other comprehensive income (loss) | - | - | ||||||
| Purchases, issuances and settlements | - | - | ||||||
| Transfers in and/or out of Level 3 | - | - | ||||||
| Balance, August 6, 2012 | (180,284 | ) | (180,284 | ) | ||||
| Total gains or losses (realized/unrealized) included in: | ||||||||
| Net income (loss) | (305,829 | ) | (305,829 | ) | ||||
| Other comprehensive income (loss) | - | - | ||||||
| Purchases, issuances and settlements | - | - | ||||||
| Transfers in and/or out of Level 3 | - | - | ||||||
| Balance, March 31, 2013 | $ | (486,113 | ) | $ | (486,113 | ) | ||
| 2012 Warrant Activities | APIC | (Gain) Loss | |||||||||||||||||
Derivative Shares | Non-derivative Shares | Total Warrant Shares | Fair Value of Derivative Warrants | Reclassification of Derivative Liability | Change in Fair Value of Derivative Liability | ||||||||||||||
| Derivative warrant at August 6, 2012 | 1,152,000 | - | 1,152,000 | (411,805 | ) | - | - | ||||||||||||
| Mark to market | 231,521 | (231,521 | ) | ||||||||||||||||
| Derivative warrant at September 30, 2012 | 1,152,000 | - | 1,152,000 | (180,284 | ) | (231,521 | ) | ||||||||||||
| Mark to market | (73,723 | ) | (73,723 | ) | |||||||||||||||
| Derivative warrant at December 31, 2012 | 1,152,000 | - | 1,152,000 | (106,561 | ) | (305,244 | ) | ||||||||||||
| Reset of warrant shares | 1,799,424 | ||||||||||||||||||
| Mark to market | (379,552 | ) | 379,552 | ||||||||||||||||
| Derivative warrant at March 31, 2013 | 2,951,424 | - | 2,951,424 | (486,113 | ) | 74,308 | |||||||||||||
| Number of | Exercise Price | Weighted Average | Fair Value at Date | Aggregate | ||||||||||||||||
| Warrant Shares | RangePer Share | Exercise Price | of Issuance | IntrinsicValue | ||||||||||||||||
| Balance, March 31, 2012 | - | $ | - | $ | - | $ | - | $ | - | |||||||||||
| Granted | 5,233,177 | 0.25 | 0.25 | 620,350 | - | |||||||||||||||
| Canceled | - | - | - | - | ||||||||||||||||
| Exercised | - | - | - | - | ||||||||||||||||
| Expired | - | - | - | - | ||||||||||||||||
| Balance, March 31, 2013 | 5,233,177 | $ | 0.25 | $ | 0.25 | $ | 620,350 | $ | - | |||||||||||
| Earned and exercisable, March 31, 2013 | 5,233,177 | $ | 0.25 | $ | 0.25 | $ | 620,350 | $ | - | |||||||||||
| Unvested, March 31, 2013 | - | $ | - | $ | - | $ | - | $ | - | |||||||||||
| Warrants Outstanding | Warrants Exercisable | ||||||||||||||||||
| Range of Exercise Prices | Number Outstanding | Average Remaining Contractual Life (in years) | Weighted Average Exercise Price | Number Exercisable | Average Remaining Contractual Life (in years) | Weighted Average Exercise Price | |||||||||||||
| $0.25 | 5,233,177 | 3.73 | $ | 0.25 | 5,233,177 | 3.73 | $ | 0.25 | |||||||||||
| $0.25 | 5,233,177 | 3.73 | $ | 0.25 | 5,233,177 | 3.73 | $ | 0.25 | |||||||||||
| Milestones | Completion Date | Number of EscrowShares | |||||||||
I. (1)Enter into exclusive international license agreement for all Agro Genesis intellectual property and products as it applies to stevia (2)Enter into cooperative agreements to work with Vietnam Institutes (a) Medical Plant Institute in Hanoi; (b) Agricultural Science Institute of Northern Central Vietnam (3)Enter into farm management agreements with local growers including the Provincial and National projects; (4)Take over management of three existing nurseries | Within 180 days of the Closing Date | 3,000,000 shares only if and when ALL four (4) milestones reached (*) | |||||||||
| II. Achieve 100 Ha field trials and first test shipment of dry leaf | Within two (2) years of the Closing Date | 1,500,000 shares (**) | |||||||||
| III. Test shipment of dry leaf to achieve minimum specs for contracted base price (currently $2.00 per kilogram) | Within two (2) years of the Closing Date | 1,500,000 shares (**) | |||||||||
| * | On December 23, 2011, 3,000,000 out of the 6,000,000 Escrow Shares have been earned and released to Ventures stockholder upon achievement of the First Milestone within 180 days of June 23, 2011, the Closing Date associated with the First Milestone. These shares were valued at $0.25 per share or $750,000 on the date of release and recorded as compensation. |
| ** | On June 23, 2013, the remaining 3,000,000 Escrow Shares have been earned to Ventures stockholder upon achievement of the Second and the Third Milestones within two (2) years of June 23, 2011, the Closing Date associated with the Milestones. These shares were valued at $0.20 per share or $600,000 on June 23, 2013 and recorded as compensation. |
Contributed andadditional paid-in capital | Earnings and losses | Other Comprehensive Income | Total non-controlling interest | |||||||||||||
| Balance at March 31, 2012 | $ | - | $ | - | $ | - | $ | - | ||||||||
| Current period earnings and losses | - | (214,082 | ) | - | (214,082 | ) | ||||||||||
| Balance at March 31, 2013 | $ | - | $ | (214,082 | ) | $ | - | $ | (214,082 | ) | ||||||
| Fiscal Year Ending March 31: | ||||
| 2014 | $ | 30,000 | ||
| 2015 | 30,000 | |||
| 2016 | 30,000 | |||
| $ | 90,000 | |||
| March 31, 2013 | March 31, 2012 | |||||||
| Net deferred tax assets – Non-current: | ||||||||
| Expected income tax benefit from NOL carry-forwards | 1,311,552 | 790,007 | ||||||
| Less valuation allowance | (1,311,552 | ) | (790,007 | ) | ||||
| Deferred tax assets, net of valuation allowance | $ | - | $ | - | ||||
For the Fiscal Year Ended March 31, 2013 | For the Period from April 11, 2011 (inception) through March 31, 2012 | |||||||
| Federal statutory income tax rate | 34.0 | % | 34.0 | % | ||||
| Change in valuation allowance on net operating loss carry-forwards | (34.0 | ) | (34.0 | ) | ||||
| Effective income tax rate | 0.0 | % | 0.0 | % | ||||
| Accounts Payable at | Net Purchases | |||||||||||||||
| March 31, 2013 | March 31, 2012 | March 31, 2013 | March 31, 2012 | |||||||||||||
| Growers Synergy Pte. Ltd. – related party | 50.1 | % | 16.4 | % | 26.4 | % | 13.5 | % | ||||||||
| Stevia Ventures Corporation | 16. 9 | % | 54.1 | % | 55.7 | % | 14.4 | % | ||||||||
| 67.0 | % | 70.5 | % | 82.1 | % | 27.9 | % | |||||||||
| Contents | Page(s) | |
| Consolidated Balance Sheets at December 31, 2013 (Unaudited) and March 31, 2013 | 5 | |
| Consolidated Statements of Operations for the Nine Months and Three Months Ended December 31, 2013 and 2012 (Unaudited) | 6 | |
| Consolidated Statement of Equity (Deficit) for the Interim Period Ended December 31, 2013 (Unaudited) and for the Fiscal Year Ended March 31, 2013 | 7 | |
| Consolidated Statements of Cash Flows for the Nine Months Ended December 31, 2013 and 2012 (Unaudited) | 8 | |
| Notes to the Consolidated Financial Statements (Unaudited) | 9 |
| December 31, 2013 | March 31, 2013 | ||||||||
| (Unaudited) | |||||||||
| Assets | |||||||||
| Current assets: | |||||||||
| Cash | $ | 85,366 | $ | 424,475 | |||||
| Accounts receivable | 164,988 | 158,008 | |||||||
| Seeds | 1,807,000 | - | |||||||
| Prepayments and other current assets | 74,946 | 33,096 | |||||||
| Total current assets | 2,132,300 | 615,579 | |||||||
| Non-current assets: | |||||||||
| Property and equipment | 24,400 | 7,925 | |||||||
| Accumulated depreciation | (4,334) | (1,234) | |||||||
| Property and equipment, net | 20,066 | 6,691 | |||||||
| Acquired technology | 1,635,300 | 1,635,300 | |||||||
| Accumulatd amortization | (163,530) | (81,765) | |||||||
| Acquired technology, net | 1,471,770 | 1,553,535 | |||||||
| Website development costs | 5,315 | 5,315 | |||||||
| Accumulated amortization | (2,670) | (1,869) | |||||||
| Website development costs, net | 2,645 | 3,446 | |||||||
| Security deposit | 15,000 | 15,000 | |||||||
| Total assets | $ | 3,641,781 | $ | 2,194,251 | |||||
| Liabilities and equity | |||||||||
| Current liabilities: | |||||||||
| Accounts payable | $ | 1,092,237 | $ | 948,073 | |||||
| Accounts payable - president and CEO | 262,576 | 89,193 | |||||||
| Accrued expenses | 9,600 | 19,700 | |||||||
| Accrued interest | 145,144 | 21,627 | |||||||
| Advances from president and significant stockholder | 10,743 | 21,238 | |||||||
| Convertible notes payable - net of discount | 1,071,569 | 357,700 | |||||||
| Current portion of derivative liability | - | - | |||||||
| Total current liabilities | 2,591,869 | 1,457,531 | |||||||
| Non-Current liabilities: | |||||||||
| Derivative note liabilities | 227,772 | - | |||||||
| Derivative warrant liabilities | 369,747 | 486,113 | |||||||
| Total non-current liabilities | 597,519 | 486,113 | |||||||
| Total liabilities | 3,189,388 | 1,943,644 | |||||||
| Equity | |||||||||
| Stevia Corp stockholders' equity: | |||||||||
| Preferred stock par value $0.001: 1,000,000 shares authorized; | |||||||||
| none issued or outstanding | - | - | |||||||
| Common stock par value $0.001: 250,000,000 shares authorized, | |||||||||
| 82,695,634 and 63,555,635 shares issued and outstanding, respectively | 82,695 | 63,556 | |||||||
| Additional paid-in capital | 6,813,321 | 4,760,624 | |||||||
| Common stock to be issued | 279,222 | - | |||||||
| Accumulated deficit | (6,376,899) | (4,359,415) | |||||||
| Total Stevia Corp stockholders' equity | 798,339 | 464,765 | |||||||
| Non-controlling interest in subsidiary | |||||||||
| Noncontrolling interest - retained earnings in consolidated subsidiaries | (345,946) | (214,158) | |||||||
| Non-controlling interest in subsidiary | (345,946) | (214,158) | |||||||
| Total Equity | 452,393 | 250,607 | |||||||
| Total Liabilities and Equity | $ | 3,641,781 | $ | 2,194,251 | |||||
| For the Nine Months | For the Three Months | For the Nine Months | For the Three Months | |||||||||||||
| Ended | Ended | Ended | Ended | |||||||||||||
| December 31, 2013 | December 31, 2013 | December 31, 2012 | December 31, 2012 | |||||||||||||
| (Unaudited) | (Unaudited) | (Unaudited) | (Unaudited) | |||||||||||||
| Revenues | $ | 1,893,865 | $ | 388,746 | $ | 120,939 | $ | 8,142 | ||||||||
| Cost of revenues | ||||||||||||||||
| Farm produce | 758,809 | - | - | - | ||||||||||||
| Farm expenses | 318,234 | 4,925 | 66,743 | 66,743 | ||||||||||||
| Farm field lease | - | - | 21,250 | 6,250 | ||||||||||||
| Farm management services - related parties | 180,000 | 60,000 | 652,550 | 60,000 | ||||||||||||
| Total cost of revenues | 1,257,043 | 64,925 | 740,543 | 132,993 | ||||||||||||
| Gross margin | 636,822 | 323,821 | (619,604 | ) | (124,851 | ) | ||||||||||
| Operating expenses: | ||||||||||||||||
| Directors' fees | 195,313 | 7,813 | 281,250 | 93,750 | ||||||||||||
| Professional fees | 487,867 | 178,135 | 352,031 | 123,356 | ||||||||||||
| Research and development | 262,810 | 71,930 | 118,669 | - | ||||||||||||
| Salary and compensation - officer | 600,000 | - | - | - | ||||||||||||
| Salary and compensation - others | 66,556 | 378 | 110,982 | 59,105 | ||||||||||||
| General and administrative expenses | 387,040 | 133,641 | 204,616 | 69,302 | ||||||||||||
| Total operating expenses | 1,999,586 | 391,897 | 1,067,548 | 345,513 | ||||||||||||
| Loss from operations | (1,362,764 | ) | (68,076 | ) | (1,687,152 | ) | (470,364 | ) | ||||||||
| Other (income) expense: | ||||||||||||||||
| Change in fair value of derivative liability | (675,949 | ) | (40,105 | ) | (305,244 | ) | (73,723 | ) | ||||||||
| Debt discount | 626,446 | 63,263 | - | - | ||||||||||||
| Debt settlement loss | 561,077 | 561,077 | - | - | ||||||||||||
| Excess of fair value of warrants over notes, net of OID | 38,075 | 38,075 | - | - | ||||||||||||
| Financing cost | 30,000 | 8,000 | 16,125 | 2,807 | ||||||||||||
| Foreign currency transaction gain (loss) | - | - | 1,316 | 1 | ||||||||||||
| Interest expense | 123,516 | 75,613 | 40,462 | 10,130 | ||||||||||||
| Other (income) expense | 83,343 | 83,343 | - | - | ||||||||||||
| Total other (income) expense | 786,508 | 789,266 | (247,341 | ) | (60,785 | ) | ||||||||||
| Loss before income tax provision and non-controlling interest | (2,149,272 | ) | (857,342 | ) | (1,439,811 | ) | (409,579 | ) | ||||||||
| Income tax provision | - | - | - | - | ||||||||||||
| Net loss | ||||||||||||||||
| Net loss before non-controlling interest | (2,149,272 | ) | (857,342 | ) | (1,439,811 | ) | (409,579 | ) | ||||||||
| Net loss attributable to the non-controlling interest | (131,788 | ) | (25,932 | ) | (97,338 | ) | (44,978 | ) | ||||||||
| Net income (loss) attributable to Stevia Corp. | $ | (2,017,484 | ) | $ | (831,410 | ) | $ | (1,342,473 | ) | $ | (364,601 | ) | ||||
| Net income (loss) per common share | ||||||||||||||||
| - Basic and diluted: | $ | (0.03 | ) | $ | (0.01 | ) | $ | (0.02 | ) | $ | (0.01 | ) | ||||
| Weighted average common shares outstanding | ||||||||||||||||
| - basic and diluted | 72,842,975 | 79,632,959 | 61,613,572 | 63,225,253 | ||||||||||||
| Common Stock Par Value $0.001 | ||||||||||||||||||||||||||||||||
| Number of | Additional | Common stock | Accumulated | Stockholders' | Non-controlling | Total | ||||||||||||||||||||||||||
| Shares | Amount | paid-in Capital | to be Issued | Deficit | Equity (Deficit) | Interest | Equity (Deficit) | |||||||||||||||||||||||||
| Balance, March 31, 2012 | 58,354,775 | $ | 58,355 | $ | 1,474,751 | $ | - | $ | (2,323,551 | ) | $ | (790,445 | ) | $ | - | $ | (790,445 | ) | ||||||||||||||
| Restricted common shares issued for farm management services to | ||||||||||||||||||||||||||||||||
| a related party valued at $0.79 per share discounted at 69% | ||||||||||||||||||||||||||||||||
| on July 5, 2012 | 500,000 | 500 | 272,050 | 272,550 | 272,550 | |||||||||||||||||||||||||||
| Restricted common shares issued for technology rights | ||||||||||||||||||||||||||||||||
| valued at $0.79 per share discounted at 69% | ||||||||||||||||||||||||||||||||
| on July 5, 2012 | 3,000,000 | 3,000 | 1,632,300 | 1,635,300 | 1,635,300 | |||||||||||||||||||||||||||
| Common shares issued for notes conversion | ||||||||||||||||||||||||||||||||
| at $0.832143 per share on July 6, 2012 | 600,858 | 601 | 499,399 | 500,000 | 500,000 | |||||||||||||||||||||||||||
| Common shares issued for conversion of accrued interest | ||||||||||||||||||||||||||||||||
| at $0.832143 per share on July 6, 2012 | 33,335 | 33 | 27,707 | 27,740 | 27,740 | |||||||||||||||||||||||||||
| Common shares and warrants issued to two investors for cash | ||||||||||||||||||||||||||||||||
| at $0.46875 per unit on August 6, 2012 | 1,066,667 | 1,067 | 498,933 | 500,000 | 500,000 | |||||||||||||||||||||||||||
| Warrants issued to investors in connection with the sale of | ||||||||||||||||||||||||||||||||
| equity units on August 6, 2012 classified as derivative liability | (381,300 | ) | (381,300 | ) | (381,300 | ) | ||||||||||||||||||||||||||
| Commissions and legal fees paid in connection with the sale of | ||||||||||||||||||||||||||||||||
| equity units on August 6, 2012 | (52,500 | ) | (52,500 | ) | (52,500 | ) | ||||||||||||||||||||||||||
| Warrants issued to placement agent in connection with the sale of | ||||||||||||||||||||||||||||||||
| equity units on August 6, 2012 classified as derivative liability | (30,504 | ) | (30,504 | ) | (30,504 | ) | ||||||||||||||||||||||||||
| Issuance of warrants in connection with | ||||||||||||||||||||||||||||||||
| convertible note payable issued in February and March 2013 | 220,438 | 220,438 | 220,438 | |||||||||||||||||||||||||||||
| Beneficial conversion feature in connection with | ||||||||||||||||||||||||||||||||
| convertible note payable issued in February and March 2013 | 224,350 | 224,350 | 224,350 | |||||||||||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||||||
services on October 4, 2011 earned during the period | 375,000 | 375,000 | 375,000 | |||||||||||||||||||||||||||||
| Net loss | (2,035,864 | ) | (2,035,864 | ) | (214,158 | ) | (2,250,022 | ) | ||||||||||||||||||||||||
| Balance, March 31, 2013 | 63,555,635 | 63,556 | 4,760,624 | - | (4,359,415 | ) | 464,765 | (214,158 | ) | 250,607 | ||||||||||||||||||||||
| Common shares issued for consulting services | ||||||||||||||||||||||||||||||||
| valued at $0.20 per share on April 30, 2013 | 500,000 | 500 | 99,500 | 100,000 | 100,000 | |||||||||||||||||||||||||||
| Exercise of warrant with exercise price adjusted | ||||||||||||||||||||||||||||||||
| to $0.20 per share on May 6, 2013 | 853,333 | 853 | 169,813 | 170,666 | 170,666 | |||||||||||||||||||||||||||
| Commissions and legal fees paid in connection with the exercise of | ||||||||||||||||||||||||||||||||
| warrants on May 6, 2013 | (18,653 | ) | (18,653 | ) | (18,653 | ) | ||||||||||||||||||||||||||
| Reclassification of derivative liability to additional paid-in capital | ||||||||||||||||||||||||||||||||
| associated with the exercise of warrants | 595,852 | 595,852 | 595,852 | |||||||||||||||||||||||||||||
| Warrants issued to investors in connection with warrants | ||||||||||||||||||||||||||||||||
| exercised on May 6, 2013 classified as derivative liability | (833,106 | ) | (833,106 | ) | (833,106 | ) | ||||||||||||||||||||||||||
| Make good shares released to officer for achieving | ||||||||||||||||||||||||||||||||
| the second and third milestones on June 21, 2013 | 3,000,000 | 3,000 | 597,000 | 600,000 | 600,000 | |||||||||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||||||
services on October 4, 2011 earned during the period endng June 30,2013 | 93,750 | 93,750 | 93,750 | |||||||||||||||||||||||||||||
| Reclassification to derivative liability for warrants | ||||||||||||||||||||||||||||||||
| that became derivatives | (167,949 | ) | (167,949 | ) | (167,949 | ) | ||||||||||||||||||||||||||
| Common shares issued for future director | ||||||||||||||||||||||||||||||||
services on October 4, 2011 earned during the period endng September 30, 2013 | 93,750 | 93,750 | 93,750 | |||||||||||||||||||||||||||||
| Anti-dilution shares issued in accordance with the Security Purchase | ||||||||||||||||||||||||||||||||
| Agreement dated August 1, 2012 on October 1, 2013 | 286,666 | 286 | (286 | ) | - | - | ||||||||||||||||||||||||||
| Common shares issued for future director service | ||||||||||||||||||||||||||||||||
| on December 4, 2013 | 1,500,000 | 1,500 | 186,000 | 187,500 | 187,500 | |||||||||||||||||||||||||||
| Common shares issued for future director service | ||||||||||||||||||||||||||||||||
| on December 4, 2013 | (187,500 | ) | (187,500 | ) | (187,500 | ) | ||||||||||||||||||||||||||
| Common shares issued per debt settlement agreement | ||||||||||||||||||||||||||||||||
| for past due accounts payable and related settlement costs | 13,000,000 | 13,000 | 1,416,715 | 279,222 | 1,708,937 | 1,708,937 | ||||||||||||||||||||||||||
| Common shares issued for future director service on December 4, 2013 | ||||||||||||||||||||||||||||||||
| earned during the period endng December 31, 2013 | 7,811 | 7,811 | 7,811 | |||||||||||||||||||||||||||||
| Net loss | (2,017,484 | ) | (2,017,484 | ) | (131,788 | ) | (2,149,272 | ) | ||||||||||||||||||||||||
| Balance, December 31, 2013 | 82,695,634 | $ | 82,695 | $ | 6,813,321 | $ | 279,222 | $ | (6,376,899 | ) | $ | 798,339 | $ | (345,946 | ) | $ | 452,393 | |||||||||||||||
| For the Nine months | For the Nine Months | |||||||
| Ended | Ended | |||||||
| December 31, 2013 | December 31, 2012 | |||||||
| (Unaudited) | (Unaudited) | |||||||
| Cash flows from operating activities: | ||||||||
| Net loss before non-controlling interest | $ | (2,149,272 | ) | $ | (1,439,811 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities | ||||||||
| Depreciation expense | 3,100 | 762 | ||||||
| Amortization expense - acquired technology | 81,765 | 54,510 | ||||||
| Amortization expense - website development costs | 801 | 801 | ||||||
| Amortization of discount of convertible notes payable | 626,446 | - | ||||||
| Debt settlement loss | 561,077 | - | ||||||
| Excess of fair value of warrants over notes, net of OID | 38,075 | - | ||||||
| Change in fair value of derivative liability | (675,949 | ) | (305,244 | ) | ||||
| Common shares issued for director services earned during the period | 195,312 | 281,250 | ||||||
| Common shares issued for services-related party | 600,000 | 272,550 | ||||||
| Common shares issued for outside services | 205,860 | - | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Accounts receivable | (6,980 | ) | (120,659 | ) | ||||
| Seeds | (765,000 | ) | - | |||||
| Prepayments and other current assets | (41,850 | ) | 142,858 | |||||
| Accounts payable | 144,164 | 412,110 | ||||||
| Accounts payable - president and CEO | 173,383 | - | ||||||
| Accrued expenses | (10,100 | ) | 6,045 | |||||
| Accrued interest | 123,517 | 40,397 | ||||||
| Net cash used in operating activities | (895,651 | ) | (654,431 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Purchases of property, plant and equipment | (16,475 | ) | (4,889 | ) | ||||
| Net cash used in investing activities | (16,475 | ) | (4,889 | ) | ||||
| Cash flows from financing activities: | ||||||||
| Advances from (repayments to) president and significant stockholder | (10,495 | ) | 2,100 | |||||
| Proceeds from issuance of convertible notes, net of costs | 431,500 | 200,000 | ||||||
| Proceeds from sale of common stock, net of costs | - | 447,500 | ||||||
| Proceeds from exercise of warrants, net of costs | 152,012 | - | ||||||
| Net cash provided by financing activities | 573,017 | 649,600 | ||||||
| Net change in cash | (339,109 | ) | (9,720 | ) | ||||
| Cash at beginning of the period | 424,475 | 15,698 | ||||||
| Cash at end of the period | $ | 85,366 | $ | 5,978 | ||||
| Supplemental disclosure of cash flows information: | ||||||||
| Interest paid | $ | - | $ | - | ||||
| Income tax paid | $ | - | $ | - | ||||
| Non-cash investing and financing activities: | ||||||||
| Issuance of common stock for past due payables | $ | 1,042,000 | $ | - | ||||
| Issuance of common stock for conversion of convertible notes | $ | - | $ | 500,000 | ||||
| Issuance of common stock for conversion of accrued interest | $ | - | $ | 27,739 | ||||
| (i) | Assumption as a going concern:Management assumes that the Company will continue as a going concern, which contemplates continuity of operations, realization of assets, and liquidation of liabilities in the normal course of business. |
| (ii) | Allowance for doubtful accounts:Management’sestimate of the allowance for doubtful accounts is based on historical sales, historical loss levels, and an analysis of the collectability of individual accounts;and general economic conditions that may affect a client’s ability to pay. The Company evaluated the key factors and assumptions used to develop the allowance in determining that it is reasonable in relation to the financial statements taken as a whole. |
| (iii) | Inventory Obsolescence and Markdowns:The Company’s estimate of potentially excess and slow-moving inventories is based on evaluation of inventory levels and aging, review of inventory turns and historical sales experiences. The Company’s estimate of reserve for inventory shrinkage is based on the historical results of physical inventory cycle counts. |
| (iv) | Fair value of long-lived assets:Fair value is generally determined using the asset’s expected future discounted cash flows or market value, if readily determinable. If long-lived assets are determined to be recoverable, but the newly determined remaining estimated useful lives are shorter than originally estimated, the net book values of the long-lived assets are depreciated over the newly determined remaining estimated useful lives. The Company considers the following to be some examples of important indicators that may trigger an impairment review: (i) significant under-performance or losses of assets relative to expected historical or projected future operating results; (ii) significant changes in the manner or use of assets or in the Company’s overall strategy with respect to the manner or use of the acquired assets or changes in the Company’s overall business strategy; (iii) significant negative industry or economic trends; (iv) increased competitive pressures; (v) a significant decline in the Company’s stock price for a sustained period of time; and (vi) regulatory changes. The Company evaluates acquired assets for potential impairment indicators at least annually and more frequently upon the occurrence of such events. |
| (v) | Valuation allowance for deferred tax assets:Management assumes that the realization of the Company’s net deferred tax assets resulting from its net operating loss (“NOL”) carry–forwards for Federal income tax purposes that may be offset against future taxable income was not considered more likely than not and accordingly, the potential tax benefits of the net loss carry-forwards are offset by a full valuation allowance. Management made this assumption based on (a) the Company has incurred recurring losses, (b) general economic conditions, and (c) its ability to raise additional funds to support its daily operations by way of a public or private offering, among other factors. |
| (vi) | Estimates and assumptions used in valuation of equity instruments: Management estimatesexpected term of share options and similar instruments, expected volatility of the Company’s common shares and the method used to estimate it, expected annual rate of quarterly dividends, and risk free rate(s) to value share options and similar instruments. |
These significant accounting estimates or assumptions bear the risk of change due to the fact that there are uncertainties attached to these estimates or assumptions, and certain estimates or assumptions are difficult to measure or value.
Name of consolidated subsidiary or entity | State or other jurisdiction of incorporation or organization | Date of incorporation or formation (date of acquisition, if applicable) | Attributable interest | |||||
| Stevia Ventures International Ltd. | The Territory of the British Virgin Islands | April 11, 2011 | 100 | % | ||||
| Stevia Asia Limited | Hong Kong SAR | March 19, 2012 | 100 | % | ||||
| Stevia Technew Limited | Hong Kong SAR | April 28, 2012 | 70 | % | ||||
| SC Brands Pte Ltd | Singapore | October 1, 2013 | 70 | % | ||||
| Level 1 | Quoted market prices available in active markets for identical assets or liabilities as of the reporting date. | |
| Level 2 | Pricing inputs other than quoted prices in active markets included in Level 1, which are either directly or indirectly observable as of the reporting date. | |
| Level 3 | Pricing inputs that are generally observable inputs and not corroborated by market data. |
| Estimated Useful Life (Years) | ||||
| Acquired technology | 15 | |||
| Website development costs | 5 | |||
| · | Expected term of share options and similar instruments: The expected life of options and similar instruments represents the period of time the option and/or warrant are expected to be outstanding. Pursuant to Paragraph 718-10-50-2(f)(2)(i) of the FASB Accounting Standards Codification the expected term of share options and similar instruments represents the period of time the options and similar instruments are expected to be outstanding taking into consideration of the contractual term of the instruments and employees’ expected exercise and post-vesting employment termination behavior into the fair value (or calculated value) of the instruments. Pursuant to paragraph 718-10-S99-1, it may be appropriate to use thesimplified method,i.e.,expected term = ((vesting term + original contractual term) / 2), if (i) A company does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term due to the limited period of time its equity shares have been publicly traded; (ii) A company significantly changes the terms of its share option grants or the types of employees that receive share option grants such that its historical exercise data may no longer provide a reasonable basis upon which to estimate expected term; or (iii) A company has or expects to have significant structural changes in its business such that its historical exercise data may no longer provide a reasonable basis upon which to estimate expected term. The Company uses the simplified method to calculate expected term of share options and similar instruments as the company does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term. |
| · | Expected volatility of the entity’s shares and the method used to estimate it. Pursuant to ASC Paragraph 718-10-50-2(f)(2)(ii) a thinly-traded or nonpublic entity that uses the calculated value method shall disclose the reasons why it is not practicable for the Company to estimate the expected volatility of its share price, the appropriate industry sector index that it has selected, the reasons for selecting that particular index, and how it has calculated historical volatility using that index. The Company uses the average historical volatility of the comparable companies over the expected contractual life of the share options or similar instruments as its expected volatility. If shares of a company are thinly traded the use of weekly or monthly price observations would generally be more appropriate than the use of daily price observations as the volatility calculation using daily observations for such shares could be artificially inflated due to a larger spread between the bid and asked quotes and lack of consistent trading in the market. |
| · | Expected annual rate of quarterly dividends. An entity that uses a method that employs different dividend rates during the contractual term shall disclose the range of expected dividends used and the weighted-average expected dividends. The expected dividend yield is based on the Company’s current dividend yield as the best estimate of projected dividend yield for periods within the expected term of the share options and similar instruments. |
| · | Risk-free rate(s). An entity that uses a method that employs different risk-free rates shall disclose the range of risk-free rates used. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for periods within the expected term of the share options and similar instruments. |
| · | Expected term of share options and similar instruments: Pursuant to Paragraph 718-10-50-2 of the FASB Accounting Standards Codification the expected term of share options and similar instruments represents the period of time the options and similar instruments are expected to be outstanding taking into consideration of the contractual term of the instruments and holder’s expected exercise behavior into the fair value (or calculated value) of the instruments. The Company uses historical data to estimate holder’s expected exercise behavior. If the Company is a newly formed corporation or shares of the Company are thinly traded the contractual term of the share options and similar instruments is used as the expected term of share options and similar instruments as the Company does not have sufficient historical exercise data to provide a reasonable basis upon which to estimate expected term. |
| · | Expected volatility of the entity’s shares and the method used to estimate it. An entity that uses a method that employs different volatilities during the contractual term shall disclose the range of expected volatilities used and the weighted-average expected volatility. A thinly-traded or nonpublic entity that uses the calculated value method shall disclose the reasons why it is not practicable for the Company to estimate the expected volatility of its share price, the appropriate industry sector index that it has selected, the reasons for selecting that particular index, and how it has calculated historical volatility using that index.The Company uses the average historical volatility of the comparable companies over the expected contractual life of the share options or similar instruments as its expected volatility. If shares of a company are thinly traded the use of weekly or monthly price observations would generally be more appropriate than the use of daily price observations as the volatility calculation using daily observations for such shares could be artificially inflated due to a larger spread between the bid and asked quotes and lack of consistent trading in the market. |
| · | Expected annual rate of quarterly dividends. An entity that uses a method that employs different dividend rates during the contractual term shall disclose the range of expected dividends used and the weighted-average expected dividends. The expected dividend yield is based on the Company’s current dividend yield as the best estimate of projected dividend yield for periods within the expected contractual life of the option and similar instruments. |
| · | Risk-free rate(s). An entity that uses a method that employs different risk-free rates shall disclose the range of risk-free rates used. The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the time of grant for periods within the contractual life of the option and similar instruments. |
| Potentially Outstanding Dilutive Common Shares | |||||
For Interim PeriodEnded December 31, 2013 | For Interim Period Ended December 31, 2012 | ||||
| Make Good Escrow Shares | |||||
| Make Good Escrow Agreement shares issued and held with the escrow agent in connection with the Share Exchange Agreement consummated on June 23, 2011 pending the achievement by the Company of certain post-Closing business milestones (the “Milestones”). | - | 3,000,000 | |||
| Sub-total Make Good Escrow Shares | - | 3,000,000 | |||
| Convertible Note Shares | |||||
| On March 7, 2012, the Company issued a convertible note in the principal amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the conversion price to be the same as the next private placement price on a per share basis, provided that the Company completes a private placement with gross proceeds of at least $100,000. On August 6, 2012, the Company completed the very next private placement at $0.46875 per share with gross proceeds of at least $100,000. On March 15, 2013, the above note was cancelled and reissued with a new convertible note consisting of the prior principal amount and the entire accrued unpaid interest for the total amount of $220,438 with interest at 12% per annum convertible at $0.25 per share due on September 30, 2013. The note is currently past due with no penalty and the Company continues to accrue the interest at 10% per annum. | 881,752 | 426,667 | |||
| On May 30, 2012, the Company issued a convertible note in the principal amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the unpaid principal and any accrued and unpaid interest thereon convertible, as of the Conversion Date, at the lower of (a) the price per share at which shares of capital stock issued in the Financing are sold in the Financing, or (b) the closing price of the Company's securities if traded on a securities exchange, or if actively traded over-the-counter, the average closing bid price for the securi1ies, in each case over the thirty (30) day period prior to the Conversion Date; provided however, that if no active trading market for the securities exists at the time of the conversion, such amount shall be the fair market value of a share of the Company's common stock as determined in good faith by Company's Board of Directors. A "Financing" means the closing of the sale of shares of capital stock of the Company in the first equity financing transaction after the date first set forth above, in which the Company receives gross proceeds of at least $100,000, excluding conversion of this Note. The note is currently past due with no penalty and the Company continues to accrue the interest at 10% per annum. | 1,739,130 | 426,667 | |||
| On February 26, 2013, the Company issued two (2) convertible notes in the principal amount of $250,000 and $100,000, respectively, convertible at $0.25 per share, with interest at 12% per annum due on September 30, 2013. The note is currently past due with no penalty and the Company continues to accrue the interest at 12% per annum. | 1,400,000 | - | |||
| On July 16, 2013, the Company issued a convertible note in the principal amount of $111,111 with a 10% Original Issuance Discount ("OID") and a one-time interest charge of 12% after 90 days. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date. | 1,762,228 | - | |||
| On August 27, 2013, the Company issued a convertible note in the principal amount of $153,500, convertible at 65% of the three lowest bids for 30 trading days before the conversion date with interest at 8% per annum, due on May 26, 2014. | 2,353,573 | - | |||
| On September 26, 2013, the Company issued a convertible note in the principal amount of $27,778 with a 10% Original Issuance Discount ("OID") and a one-time interest charge of 12%. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date. | 440,571 | - | |||
| On October 15, 2013, the Company issued a convertible note in the principal amount of $58,000 with an $8,000 Original Issuance Discount ("OID") and with interest at 10% per annum, convertible at $0.20 per share, due on May 1, 2014. | 290,000 | - | |||
| On November 21, 2013, the Company issued a convertible note in the principal amount of $53,000, convertible at 65% of the three lowest bids for 30 trading days before the conversion date with interest at 8% per annum, due on August 25, 2014. | 812,634 | - | |||
| On December 9, 2013, the Company issued a convertible note in the principal amount of $55,556 with a 10% Original Issuance Discount ("OID") and 12% one time interest. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date . | 881,142 | - | |||
| Sub-total Convertible Note Shares | 10,561,070 | 853,334 |
| Warrant Shares | |||||
| On August 6, 2012, the Company issued (i) warrants to purchase 1,066,667 shares, in the aggregate, of the Company’s common stock to investors (the “investor warrants”) and (ii) warrants to purchase 85,333 shares of the Company's common stock to the placement agent (the "agent warrants") with an exercise price of $0.6405 per share, subject to certain adjustments pursuant to Section 3(b) Subsequent Equity Sales of the SPA, expiring five (5) years from the date of issuance. On February 26, 2013, warrantsissued subsequent to these warrantstriggered a reset of these warrants exercise price to $0.25 per share and the shares to be issued under the warrants were adjusted to 2,951,424 shares accordingly. On May 8, 2013, the Company completed a private placement at $0.20 per share with gross proceeds more than $100,000; this event triggered the reset of the conversion price of the convertible note to $0.20 per share and the shares to be issued under the warrants were adjusted to 3,689,280 shares accordingly. On May 8, 2013, investors exercised the warrants to purchase 2,732,799 shares (853,333 original shares) at $0.20 per share. | 956,481 | 1,152,000 | |||
| On February 26, 2013, the Company issued warrants to purchase 1,000,000 and 400,000 shares respectively, or 1,400,000 shares in the aggregate, of the Company’s common stock to two (2) note holders in connection with the issuance of convertible notes. | 1,400,000 | - | |||
| On March 15, 2013, the Company issued a warrant to purchase 881,753 shares of the Company’s common stock to the note holder in connection with the issuance of the convertible note. | 881,753 | - | |||
On May 6, 2013, the Company issued three (3) series of warrants: Series A warrants include (i) warrants to purchase 1,877,333 shares of the Company’s common stock to the investor and (ii) warrants to purchase 150,187 shares of the Company's common stock to the placement agent (the "agent warrants") with an exercise price of $0.20 per share expiring five (5) years from the date of issuance. | 2,027,520 | - | |||
Series B warrants include (i) warrants to purchase 1,066,666 shares of the Company’s common stock to the investor and (ii) warrants to purchase 85,333 shares of the Company's common stock to the placement agent (the "agent warrants") with an exercise price of $0.25 per share expiring five (5) years from the date of issuance. | 1,151,999 | - | |||
Series C warrants include (i) warrants to purchase 2,346,666 shares of the Company’s common stock to the investor and (ii) warrants to purchase 187,733 shares of the Company's common stock to the placement agent (the "agent warrants") with an exercise price of $0.25 per share expiring five (5) years from the date of issuance. The warrants are exercisable under the condition of Series A warrants are exercised. | 2,534,399 | - | |||
| On October 15, 2013, the Company issued warrants to purchase 1,000,000 shares of the Company’s common stock to a note holder with an exercise price of $0.25 per share in connection with the issuance of convertible note. | 1,000,000 | - | |||
| Sub-total Warrant Shares | 9,952,152 | 1,152,000 | |||
| Total potentially outstanding dilutive common shares | 20,513,222 | 5,005,334 |
Subsequent Events
| December 31, 2013 | March 31, 2013 | |||||||
| Seeds (*) | $ | 1,807,000 | $ | - | ||||
| $ | 1,807,000 | $ | - | |||||
| Related Parties | Relationship | |
| George Blankenbaker | President and significant stockholder of the Company | |
| Leverage Investments LLC | An entity owned and controlled by the president and significant stockholder of the Company | |
| Technew Technology Limited | Non-controlling interest holder |
| Growers Synergy Pte Ltd. | An entity owned and controlled by the president and significant stockholder of the Company | |
| Guangzhou Health Technology Development Company Limited | An entity owned and controlled by Non-controlling interest holder |
For the interim period ended December 31, 2013 | For the interim period ended December 31, 2012 | |||||||
| Farm management services – related parties | $ | 180,000 | $ | 180,000 | ||||
| $ | 180,000 | $ | 180,000 | |||||
| Fiscal Year Ending March 31: | ||||
| 2014 (remainder of the fiscal year) | $ | 240,000 | ||
| 2015 | 240,000 | |||
| 2016 | 240,000 | |||
| 2017 | 140,000 | |||
| $ | 860,000 | |||
| Expected option life (year) | 3.00 | |||
| Expected volatility | 74.53 | % | ||
| Risk-free interest rate | 0.37 | % | ||
| Dividend yield | 0.00 | % |
| Expected option life (year) | 3.00 | |||
| Expected volatility | 75.11 | % | ||
| Risk-free interest rate | 0.40 | % | ||
| Dividend yield | 0.00 | % |
| December 31, 2013 | March 31, 2013 | |||||||
| On May 30, 2012, the Company issued a convertible note in the principal amount of $200,000 with interest at 10% per annum due one (1) year from the date of issuance with the unpaid principal of this note and any accrued and unpaid interest thereon, as of the Conversion Date, at the lower of (a) the price per share at which shares of capital stock issued in the Financing are sold in the Financing, or (b) the closing price of the Company's securities if traded on a securities exchange, or if actively traded over-the-counter, the average closing bid price for the securi1ies, in each case over the thirty (30) day period prior to the Conversion Date; provided however, that if no active trading market for the securities exists at the time of the conversion, such amount shall be the fair market value of a share of the Company's common stock as determined in good faith by Company's Board of Directors. A "Financing" means the closing of the sale of shares of capital stock of the Company in the first equity financing transaction after the date first set forth above, in which the Company receives gross proceeds of at least $100,000, excluding conversion of this Note. The note is currently past due with no penalty and the Company continues to accrue the interest at 10% per annum. | 200,000 | 200,000 | ||||||
| February 26, 2013 convertible notes | 350,000 | 350,000 | ||||||
| March 15, 2013 convertible note | 220,438 | 220,438 | ||||||
| On July 16, 2013, the Company issued a convertible note in the principal amount of $111,111 with a 10% Original Issuance Discount ("OID") and a one-time interest charge of 12% after 90 days. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date . | 111,111 | - | ||||||
| On August 27, 2013, the Company issued a convertible notes in the principal amount of $153,500 convertible at 65% of the three lowest bids for 30 trading days before the conversion date with interest at 8% per annum due on May 26, 2014. | 153,500 | - | ||||||
| On September 26, 2013, the Company issued a convertible note in the principal amount of $27,778 with a 10% Original Issuance Discount ("OID") and a one-time interest charge of 12%. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date . | 27,778 | - | ||||||
| On October 15, 2013, the Company issued a convertible note in the principal amount of $58,000 convertible at $0.20 per share, with an $8,000 Original Issue Discount ("OID") and interest at 10% per annum maturing on May 1, 2014. The Debenture is secured by 1,250,000 restricted common shares of the Company. In connection with the issuance of the convertible note, the Company granted the note holder a warrant to purchase 1,000,000 common shares with an exercise price of $0.25 per share, subject to certain adjustments pursuant to Section 3(b) Subsequent Equity Sales and Section 3(c) Subsequent Rights Offerings of the warrant ("full price and share reset provisions") expiring five (5) years from the date of issuance. | 58,000 | - | ||||||
| On November 21, 2013, the Company issued a convertible note in the principal amount of $53,000, convertible at 65% of the three lowest bids for 30 trading days before the conversion date, with interest at 8% per annum, due on August 25, 2014. | 53,000 | - | ||||||
| On December 9, 2013, the Company issued a convertible note in the principal amount of $55,556 with a 10% Original Issuance Discount ("OID") and a one-time interest charge of 12%. The note is due one (1) year from the date of issuance with the conversion price at 65% of the lowest trade price for the 25 trade day period before the conversion date . | 55,556 | - | ||||||
| Sub-total: convertible notes payable | 1,229,383 | 770,438 | ||||||
| Discount representing (i) the relative fair value of the warrants issued, (ii) the beneficial conversion features and (iii) the derivative liability on conversion features | (816,310 | ) | (444,788 | ) | ||||
| Accumulated amortization of discount on convertible notes payable | 658,496 | 32,050 | ||||||
| Remaining discount | (157,814 | ) | (412,738 | ) | ||||
| $ | 1,071,569 | $ | 357,770 |
| · | The stock price would fluctuate with the Company projected volatility. |
| · | The stock price would fluctuate with an annual volatility. The projected volatility curve was based on historical volatilities of the Company for the valuation periods. |
| · | The Holder would exercise the warrant as they become exercisable (effective registration is projected 4 months from issuance and the earliest exercise is projected 180 days from issuance) at target prices of 2 times the higher of the projected reset price or stock price. |
| · | The Holder would exercise the warrant at maturity if the stock price was above the project reset prices. |
| · | A 100% probability of a reset event and a projected financing each quarter for 3 years at prices approximating 93% of market |
| · | The Warrants with an exercise price of $0.25 exercise price is projected to reset to $0.047 at maturity; the Warrants with an exercise price of $0.20 per share is projected to reset to $0.043at maturity |
| · | The Company had no reset event during this quarter period ending 12/31/2013. Prior reset events occurred on 2/26/2013 to $0.25 and 5/6/2013 to $0.20. |
| · | No warrants have expired. Warrants with full reset feature issued during this quarter period ending 12/31/2013 |
| · | The projected volatility curve for the valuation dates was: |
| 1 Year | 2 Year | 3 Year | 4 Year | 5 Year | ||||||||||||
| August 6, 2012 | 129% | 178% | 218% | 252% | 281% | |||||||||||
| September 30, 2012 | 127% | 173% | 211% | 244% | 272% | |||||||||||
| March 31, 2013 | 122% | 167% | 205% | 236% | 264% | |||||||||||
| December 31, 2013 | 111% | 168% | 202% | 233% | 261% |
Derivative warrants Assets (Liability) | Total | ||||||||
| Balance, September 30, 2012 | $ | (180,284 | ) | $ | (180,284 | ) | |||
| Total gains or losses (realized/unrealized) included in: | |||||||||
| Net income (loss) | (305,829 | ) | (305,829 | ) | |||||
| Other comprehensive income (loss) | - | - | |||||||
| Purchases, issuances and settlements | - | - | |||||||
| Transfers in and/or out of Level 3 | - | - | |||||||
| Balance, March 31, 2013 | (486,113 | ) | (486,113 | ) | |||||
| Total gains or losses (realized/unrealized) included in: | |||||||||
| Net income (loss) | 675,949 | 675,949 | |||||||
| Other comprehensive income (loss) | - | - | |||||||
| Purchases, issuances and settlements | (787,355 | ) | (787,355 | ) | |||||
| Transfers in and/or out of Level 3 | - | - | |||||||
| Balance, December 31, 2013 | $ | (597,519 | ) | $ | (597,519 | ) | |||
| Warrant Activities | APIC | (Gain) Loss | |||||||||||||||||
Derivative Shares | Non-derivative Shares | Total Warrant Shares | Fair Value of Derivative Warrants | Reclassification of Derivative Liability | Change in Fair Value of Derivative Liability | ||||||||||||||
| Derivative warrant at August 6, 2012 | 1,152,000 | - | 1,152,000 | (411,805 | ) | - | - | ||||||||||||
| Mark to market | 231,521 | (231,521 | ) | ||||||||||||||||
| Derivative warrant at September 30, 2012 | 1,152,000 | - | 1,152,000 | (180,284 | ) | ||||||||||||||
| Mark to market | (73,723 | ) | (73,723 | ) | |||||||||||||||
| Derivative warrant at December 31, 2012 | 1,152,000 | - | 1,152,000 | (106,561 | ) | ||||||||||||||
| Reset of warrant shares | 1,799,424 | ||||||||||||||||||
| Mark to market | (379,552 | ) | 379,552 | ||||||||||||||||
| Derivative warrant at March 31, 2013 | 2,951,424 | - | 2,951,424 | (486,113 | ) | ||||||||||||||
| Exercise of warrants on May 6, 2013 | (2,732,799 | ) | - | (2,732,799 | ) | - | - | - | |||||||||||
| Issuance of warrants on May 6, 2013 | 5,713,918 | - | 5,713,918 | (106,360 | ) | - | - | ||||||||||||
| Reset of warrant shares | 737,856 | 737,856 | |||||||||||||||||
| Issuance of warrants on Oct 15, 2013 | 1,000,000 | 1,000,000 | (76,647 | ) | |||||||||||||||
| Mark to market | 299,373 | (299,373) | |||||||||||||||||
| Derivative warrant at December 31, 2013 | 7,670,399 | - | 7,670,399 | (369,747 | ) |
Number of Warrant Shares | Exercise Price Range Per Share | Weighted Average Exercise Price | Fair Value at Date of Issuance | Aggregate Intrinsic Value | ||||||||||||||||
| Balance, March 31, 2013 | 5,233,177 | $ | 0.20 | $ | 0.20 | $ | 620,325 | $ | - | |||||||||||
| Issuance of warrant shares Pursuant to Section 3(b) Subsequent Equity Sales | 737,856 | 0.20 | 0.20 | - | ||||||||||||||||
| Granted | 6,713,918 | 0.20 - 0.25 | 0.23 | 183,007 | - | |||||||||||||||
| Canceled | - | - | - | - | ||||||||||||||||
| Exercised | (2,732,799 | ) | 0.20 | - | - | |||||||||||||||
| Expired | - | - | - | - | ||||||||||||||||
| Balance, December 31, 2013 | 9,952,152 | $ | 0.20 - 0.25 | $ | 0.23 | $ | 803,332 | $ | - | |||||||||||
| Earned and exercisable, December 31, 2013 | 9,952,152 | $ | 0.20 - 0.25 | $ | 0.23 | $ | 803,332 | $ | - | |||||||||||
| Unvested, December 31, 2013 | - | $ | - | $ | - | $ | - | $ | - | |||||||||||
| Warrants Outstanding | Warrants Exercisable | ||||||||||||||
| Range of Exercise Prices | Number Outstanding | Average Remaining Contractual Life (in years) | Weighted Average Exercise Price | Number Exercisable | Average Remaining Contractual Life (in years) | Weighted Average Exercise Price | |||||||||
| $0.20 - 0.25 | 9,952,152 | 4.36 | $ | 0.23 | 9,952,152 | 4.36 | $ | 0.23 | |||||||
| $0.20 - 0.25 | 9,952,152 | 4.36 | $ | 0.23 | 9,952,152 | 4.36 | $ | 0.23 | |||||||
| * | On December 23, 2011, 3,000,000 out of the 6,000,000 Escrow Shares have been earned by and released to Ventures stockholder upon achievement of the First Milestone within 180 days of June 23, 2011, the Closing Date associated with the First Milestone. These shares were valued at $0.25 per share or $750,000 on the date of release and recorded as salary and compensation - officer. |
| ** | On June 23, 2013, the remaining 3,000,000 Escrow Shares have been earned by and released to Ventures stockholder upon achievement of the Second and the Third Milestones within two (2) years of June 23, 2011, the Closing Date associated with the Milestones. These shares were valued at $0.20 per share or $600,000 on June 23, 2013 and recorded as salary and compensation - officer. |
| Fiscal Year Ending March 31: | ||||
| 2014 (remainder of the year) | $ | 7,500 | ||
| 2015 | 30,000 | |||
| 2016 | 30,000 | |||
| $ | 67,500 | |||
| Accounts Payable at | Net Purchases | ||||||||||||||
| December 31, 2013 | March 31, 2013 | For the Reporting Period Ended December 31, 2013 | For the Reporting Period Ended December 31, 2012 | ||||||||||||
| Growers Synergy Pte. Ltd. – related party | 45.0 | % | 50.1 | % | 6.0 | % | 49.8 | % | |||||||
| Stevia Ventures Corporation | 19.3 | % | 16. 9 | % | 26.6 | % | 10.3 | % | |||||||
| SG Agro Tech Pte Ltd | - | % | - | % | 52.2 | % | - | % | |||||||
| 64.3 | % | 67.0 | % | 84.8 | % | 60.1 | % | ||||||||
PART II - INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution
The following table sets forth the costs and expenses payable by us in connection with the issuance and distribution of the securities being registered hereunder. No expenses will be borne by the Selling Security Holders. All of the amounts shown are estimates, except for the SEC registration fee.
| SEC registration fee | $ | 1,145.53 | ||
| Accounting fees and expenses | $ | 5,000.00 | ||
| Legal fees and expenses | $ | 10,000.000 | ||
| Total | $ | 16,145.53 |
Item 14. Indemnification of Directors and Officers
Nevada Law
Section 78.7502 of the Nevada Revised Statutes permits a corporation to indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, except an action by or in the right of the corporation, by reason of the fact that he is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise, against expenses, including attorneys’ fees, judgments, fines and amounts paid in settlement actually and reasonably incurred by him in connection with the action, suit or proceeding if he:
| (a) | is not liable pursuant to Nevada Revised Statute 78.138, or |
| (b) | acted in good faith and in a manner which he reasonably believed to be in or not opposed to the best interests of the corporation, and, with respect to any criminal action or proceeding, had no reasonable cause to believe his conduct was unlawful. |
In addition, Section 78.7502 permits a corporation to indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of the corporation to procure a judgment in its favor by reason of the fact that he is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against expenses, including amounts paid in settlement and attorneys’ fees actually and reasonably incurred by him in connection with the defense or settlement of the action or suit if he:
| (a) | is not liable pursuant to Nevada Revised Statute 78.138; or |
| (b) | acted in good faith and in a manner which he reasonably believed to be in or not opposed to the best interests of the corporation. |
To the extent that a director, officer, employee or agent of a corporation has been successful on the merits or otherwise in defense of any action, suit or proceeding referred to above, or in defense of any claim, issue or matter, the corporation is required to indemnify him against expenses, including attorneys’ fees, actually and reasonably incurred by him in connection with the defense.
Section 78.751 of the Nevada Revised Statutes provides that such indemnification may also include payment by the Company of expenses incurred in defending a civil or criminal action or proceeding in advance of the final disposition of such action or proceeding upon receipt of an undertaking by the person indemnified to repay such payment if he shall be ultimately found not to be entitled to indemnification under Section 78.751. Indemnification may be provided even though the person to be indemnified is no longer a director, officer, employee or agent of the Company or such other entities.
| 42 |
Section 78.752 of the Nevada Revised Statutes allows a corporation to purchase and maintain insurance or make other financial arrangements on behalf of any person who is or was a director, officer, employee or agent of the corporation or is or was serving at the request of the corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise for any liability asserted against him and liability and expenses incurred by him in his capacity as a director, officer, employee or agent, or arising out of his status as such, whether or not the corporation has the authority to indemnify him against such liability and expenses.
Other financial arrangements made by the corporation pursuant to Section 78.752 may include the following:
| (a) | the creation of a trust fund; |
| (b) | the establishment of a program of self-insurance; |
| (c) | the securing of its obligation of indemnification by granting a security interest or other lien on any assets of the corporation; and |
| (d) | the establishment of a letter of credit, guaranty or surety |
No financial arrangement made pursuant to Section 78.752 may provide protection for a person adjudged by a court of competent jurisdiction, after exhaustion of all appeals, to be liable for intentional misconduct, fraud or a knowing violation of law, except with respect to the advancement of expenses or indemnification ordered by a court.
Any discretionary indemnification pursuant to NRS 78.7502, unless ordered by a court or advanced pursuant to an undertaking to repay the amount if it is determined by a court that the indemnified party is not entitled to be indemnified by the corporation, may be made by the corporation only as authorized in the specific case upon a determination that indemnification of the director, officer, employee or agent is proper in the circumstances. The determination must be made:
| (a) | by the stockholders; |
| (b) | by the board of directors by majority vote of a quorum consisting of directors who were not parties to the action, suit or proceeding; |
| (c) | if a majority vote of a quorum consisting of directors who were not parties to the action, suit or proceeding so orders, by independent legal counsel in a written opinion, or |
| (d) | if a quorum consisting of directors who were not parties to the action, suit or proceeding cannot be obtained, by independent legal counsel in a written opinion. |
Charter Provisions and Other Arrangements of the Registrant
Pursuant to the provisions of Nevada Revised Statutes, the Registrant has adopted the following indemnification provisions in its Bylaws for its directors and officers:
The Company shall indemnify, to the maximum extent permitted by the law, any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, except an action by or in the right of the Company, by reason of the fact that such person is or was a director, officer, employee, or agent of the Company, or is or was serving at the request of the Company as a director, officer, employee, or agent of another company, partnership, joint venture, trust, or other enterprise, against expenses, including attorneys’ fees, judgments, fines, and amounts paid in settlement actually and reasonably incurred by such person in connection with such action, suit, or proceeding if such person acted in good faith and in a manner which such person reasonably believed to be in or not opposed to the best interests of the Company, and, with respect to any criminal action or proceeding, had no reasonable cause to believe such person’s conduct was unlawful. The termination of any action, suit or proceeding by judgment, order, settlement, conviction, or upon a plea ofno lo contendere or its equivalent, shall not, of itself, create a presumption that the person did not act in good faith and in a manner which such person reasonably believed to be in or not opposed to the best interests of the Company, and that, with respect to any criminal action or proceeding, such person had reasonable cause to believe that his conduct was unlawful.
The Company shall indemnify, to the maximum extent permitted by the law, any person who was or is a party or is threatened to be made a party to any threatened, pending or completed action or suit by or in the right of the Company to procure a judgment in its favor by reason of the fact that such person is or was a director, officer, employee, or agent of the Company, or is or was serving at the request of the Company as a director, officer, employee or agent of another company, partnership, joint venture, trust, or other enterprise against expenses, including attorneys’ fees, actually and reasonably incurred by such person in connection with the defense or settlement of such action or suit if such person acted in good faith and in a manner which such person reasonably believed to be in or not opposed to the best interests of the Company, but no indemnification shall be made in respect of any claim, issue or matter as to which such person has been adjudged to be liable for negligence or misconduct in the performance of such person’s duty to the Company unless and only to the extent that the court in which such action or suit was brought determines upon application that, despite the adjudication of liability but in view of all the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses as the court deems proper.
| 43 |
To the extent that a director, officer, employee or agent of the Company has been successful on the merits or otherwise in defense of any action, suit or proceeding referred to in the prior two paragraphs, or in defense of any claim, issue or matter therein, such person shall be indemnified by the Company against expenses, including attorneys’ fees, actually and reasonably incurred by such person in connection with such defense. Any indemnification under the prior two paragraphs, unless ordered by a court, shall be made by the Company only as authorized in the specific case upon a determination that indemnification of the director, officer, employee or agent is proper in the circumstances because such person has met the applicable standard of conduct set forth in the prior two paragraphs. Such determination shall be made:
| (i) | by the stockholders; |
| (ii) | by the board of directors by majority vote of a quorum consisting of directors who were not parties to such act, suit or proceeding; |
| (iii) | if such a quorum of disinterested directors so orders, by independent legal counsel in a written opinion; or |
| (iv) | if such a quorum of disinterested directors cannot be obtained, by independent legal counsel in a written opinion. |
Expenses incurred in defending a civil or criminal action, suit or proceeding may be paid by the Company in advance of the final disposition of such action, suit or proceeding as authorized by the board of directors unless it is ultimately determined that such director, officer, employee or agent is not entitled to be indemnified by the Company as authorized in the Bylaws or as provided by law.
The indemnification provided by the Bylaws:
| (i) | does not exclude any other rights to which a person seeking indemnification may be entitled under any bylaw, agreement, vote of stockholders, or disinterested directors or otherwise, both as to action in such person’s official capacity and as to action in another capacity while holding such office; and |
| (ii) | shall continue as to a person who has ceased to be a director, officer, employee or agent and shall inure to the benefit of the heirs, executors, and administrators of such a person. |
The Company may purchase and maintain insurance on behalf of any person who is or was a director, officer, employee or agent of the Company, or is or as serving at the request of the Company as a director, officer, employee, or agent of another company, partnership, joint venture, trust, or other enterprise against any liability asserted against such person and incurred by such person in any such capacity, or arising out of such person’s status as such, whether or not the Company would have the power to indemnify such person against such liability under the provisions of the Bylaws.
Item 15. Recent Sales of Unregistered Securities
Promissory Notes
On February 14, 2011 we issued a convertible promissory note in the principal amount of $250,000 to Vantage Associates SA (“Vantage”) and on June 23, 2011, we issued an additional convertible promissory note to Vantage in the principal amount of $100,000 (the “Original Notes”). The Original Notes were convertible into shares of the Company’s common stock upon the closing by the Company of an equity financing yielding aggregate gross proceeds of at least $100,000. The Original Notes convert at the price per share of the securities issued in such financing. The Original Notes were issued in reliance upon exemption from registration under the Securities Act pursuant to Regulation S thereof. The Original Notes were converted into common stock on October 4, 2011 and are no longer outstanding.
Share Exchange Transaction
In connection with the Share Exchange Transaction, on June 23, 2011 we issued a total of 12,000,000 shares of our common stock in exchange for 100% of the issued and outstanding common stock of BVI. The common stock was issued in reliance upon exemption from registration under the Securities Act pursuant to Rule 506 of Regulation D thereof, and comparable exemptions under state securities laws. The common stock was issued to “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act, based upon representations made by such investor.
Sale of Common Stock and Additional Promissory Notes
On October 6, 2011, we raised $100,000 through the sale of 400,000 shares of our common stock at a price of $0.25 per share (the “October Shares”).
On October 6, 2011, we raised $150,000 from the proceeds of a convertible note (the “October Note”). The October Note was based upon the Company’s standard form of promissory note, accrues interest at the rate of ten percent per annum, simple interest and the principal balance of the October Note and any accrued interest thereon is convertible into our common stock at a $0.25 per share conversion price. The October Note was converted into common stock on January 18, 2012 and is no longer outstanding.
| 44 |
On November 16, 2011, we raised $250,000 from the proceeds of a convertible note (the “November Note”). The November Note was based upon the Company’s standard form of promissory note, accrues interest at the rate of ten percent per annum, simple interest and the principal balance of the November Note and any accrued interest thereon is convertible into our common stock at the lower of (a) the price per share at which shares of capital stock are sold in our next equity financing, or (b) the closing price of our securities if traded on a securities exchange, or if actively traded over-the-counter, the average closing bid price for the securities, in each case over the thirty (30) day period prior to the date of conversion; provided however, that if no active trading market for the securities exists at the time of the conversion, such conversion price shall be the fair market value of a share of our common stock as determined in good faith by our board of directors. The November Note was converted into common stock on July 6, 2012 and is no longer outstanding.
On January 16, 2012, March 7, 2012 and May 30, 2012, we raised $250,000, $200,000 and $200,000 respectively from the proceeds of convertible notes (the “Subsequent Notes”). The Subsequent Notes were based upon the Company’s standard form of promissory note, accrue interest at the rate of ten percent per annum, simple interest and the principal balance of the Subsequent Notes and any accrued interest thereon is convertible into our common stock at the lower of (a) the price per share at which shares of capital stock are sold in our next equity financing, or (b) the closing price of our securities if traded on a securities exchange, or if actively traded over-the-counter, the average closing bid price for the securities, in each case over the thirty (30) day period prior to the date of conversion; provided however, that if no active trading market for the securities exists at the time of the conversion, such conversion price shall be the fair market value of a share of our common stock as determined in good faith by our board of directors. The Note issued on January 16, 2012 was converted into common stock on July 16, 2012 and is no longer outstanding. The other Subsequent Notes remain outstanding.
On January 26, 2012, we entered into an Equity Purchase Agreement (the “Southridge Agreement”) with Southridge Partners II, LP, a Delaware limited partnership (“Southridge”). Upon execution of the Southridge Agreement, we issued 35,000 shares of our common stock to Southridge as a commitment fee (the “Southridge Shares”).
On March 19, 2012, we issued 27,500 shares of our common stock to Empire Relations Group (“Empire”) as consideration for consulting services rendered by Empire to the Company (the “Empire Shares”).
On July 5, 2012, we entered into a Technology Acquisition Agreement (the “Technology Agreement”) with Technew, pursuant to which we acquired the rights to certain technology from Technew in exchange for 3,000,000 shares of our common stock (the “Technew Shares”).
On July 5, 2012, we issued 500,000 shares of our common stock (the “Growers Synergy Shares”) to Growers Synergy Pte Ltd., a corporation organized under the laws of Singapore (“Growers Synergy”), as consideration for services rendered by Growers Synergy to the Company. George Blankenbaker, our president, director and stockholder is the managing director of Growers Synergy. Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and Mr. Blankenbaker controls a 49% interest in Growers Fresh. owned and controlled by the president and major stockholder of the Company.
On February 26, 2013 we raised an aggregate of $350,000, from the proceeds of convertible notes. Additionally, on March 15, 2013, we issued a convertible promissory note in the aggregate principal amount of $220,438.36 in exchange for the cancellation of an outstanding promissory note in the principal amount of $200,000 issued March 7, 2012 (collectively, the "2013 Notes").
The 2013 Notes were each based upon our standard form of promissory note, accrue interest at the rate of twelve percent per annum simple interest, and the principal balance of each 2013 Note and any accrued interest thereon is convertible into our common stock at a price per share of $0.25.
In connection with the issuance of the Notes, we issued to each investor a warrant to purchase such number of shares of common stock as is equal to four times the principal amount of their 2013 Note (the “2013 Warrants”). The 2013 Warrants have a three year term and an exercise price of $0.25 per share.
The issuance of the October Shares, the Notes, the Technew Shares and the Growers Synergy Shares were conducted in reliance upon Regulation S of the Securities Act of 1933, as amended and the rules and regulations promulgated thereunder (the “Securities Act”), to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act, in offshore transactions (as defined in Rule 902 under Regulation S of the Securities Act), based upon representations made by such investors.
The issuance of the Southridge Shares and the Empire Shares were conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act, based upon representations made by such investors.
The issuances of the 2013 Notes and the 2013 Warrants were conducted in reliance upon Regulation D and Regulation S of the Securities Act of 1933, as amended and the rules and regulations promulgated thereunder (the "Securities Act"), to investors who are "accredited investors," as such term is defined in Rule 501(a) under the Securities Act or in offshore transactions (as defined in Rule 902 under Regulation S of the Securities Act), based upon representations made by such investors.
| 45 |
2012 Financing
On August 6, 2012, we raised $500,000 in a private placement financing (the “Offering”) through the sale of (i) an aggregate of 1,066,667 shares of common stock at a price per share of $0.46875 and (ii) warrants to purchase an equal number of shares of the Company’s common stock at an exercise price of $0.6405 with a term of 5 years (the “Financing Securities”). The Company intends to use the net proceeds from this offering to advance the Company’s ability to execute its growth strategy and to aid in the commercial development of the recently announced launch of the Company’s majority-owned subsidiary, Stevia Technew Limited.
Garden State Securities, Inc. (the “Placement Agent”) served as the placement agent of the Company for the Offering. In consideration for services rendered as the Placement Agent, the Company agreed to: (i) pay to the Placement Agent cash commissions equal to $40,000, or 8.0% of the gross proceeds received in the Offering, and (ii) issue to the Placement Agent, or its designee, a Warrant to purchase up to 85,333 shares of the Company’s common stock (representing 8% of the Shares sold in the Offering) with an exercise price of$0.6405 per share and a term of 5 years (the “GSS Securities”).
The issuance of the Financing Securities and the GSS Securities were conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act, based upon representations made by such investors.
Reset Warrants
On May 3, 2013, the Company entered into a Warrant Exercise Reset Offer Letter Agreement (the “Reset Letter”) with an accredited investor (the “Investor”) whereby the Company and the Investor agreed that the Investor would immediately cash exercise its warrant to purchase 853,333 shares of common stock of the Company at an exercise price of $0.20 per share. In consideration for the Investor’s immediate exercise, the Company agreed to issue to the Investor three new warrants in the amounts of 1,877,333, 1,066,666 and 2,346,666, with exercise prices of $0.20, $0.25 and $0.25 per share, respectively (the “Series A Warrants”, “Series B Warrants” and “Series C Warrants”, respectively, and collectively the “New Warrants”). With the exception of the different exercise prices, the Warrants all contain the same terms, except that the Series A Warrants are subject to the Company’s call right, and the Series C Warrants are only exercisable upon the Investor’s exercise in full of the Series A Warrants, pursuant to the terms of a Warrant Exercise Reset Offer Letter Agreement.
In connection with the Reset Letter, the Company agreed to use its best efforts to file a registration statement (the “Registration Statement”) with the Securities and Exchange Commission (the “SEC”) within ten (10) business days. The Company will use its best efforts to have the Registration Statement declared effective by the SEC within thirty (30) days.
Garden State Securities, Inc. (the “Placement Agent”) served as the placement agent of the Company for the Offering. In consideration for services rendered as the Placement Agent, the Company agreed to: (i) pay to the Placement Agent cash commissions equal to $13,600, (ii) warrants equal to eight percent (8%) of the aggregate number of shares exercised by the Investor, and (iii) upon exercise of the New Warrants by the Company, the Placement Agent will receive additional warrants equal to eight percent (8%) of the number of shares issued upon exercise of the New Warrants (collectively, the “Agent Warrants”).
The New Warrants and the Agent Warrants (including the shares of the Company’s common stock underlying the New Warrants and the Agent Warrants) were offered and sold in reliance on the exemption from registration afforded by Section 4(2) and Regulation D (Rule 506) under the Securities Act and corresponding provisions of state securities laws.
Stock Issuance for Services
On April 30, 2013, the Company issued 1,000,000 shares of our common stock (the “Mountain Sky Shares”) to Mountain Sky International Limited, a Hong Kong corporation (“Mountain Sky”), in partial consideration for consulting services rendered by Mountain Sky. 500,000 of the shares vest at the time of grant, and 500,000 vest on the one year anniversary of the date of grant.
June 2013 Note
On July 16, 2013, the Company entered into a $400,000 Promissory Note (the “June 2013 Note”) with an accredited investor (the “Investor”) whereby the Investor agreed to loan to the Company up to $400,000 pursuant to the terms of the June 2013 Note. The June 2013 Note provides for the first $100,000 to be advanced upon closing and additional amounts will be advanced at the Investor’s sole discretion. Each advance is subject to a 10% original issue discount, such that the total amount which may actually be received by the Company pursuant to the June 2013 Note is only $360,000. The maturity date for each advance made under the June 2013 Note is one year from the date of such advance. If the Company repays the June 2013 Note on or before 90 days from the effective date, the interest rate shall be 0%, otherwise a one-time interest charge of l2% shall be applied to the principal sum.
The June 2013 Notes are convertible into common stock of the Company on a cashless basis at any time, at a conversion price equal to the lesser of $0.26 or 65% of the lowest trade price in the 25 trading days prior to the conversion. If the conversion shares are not deliverable by DWAC an additional 10% discount will apply, and if the shares are ineligible for deposit into the DTC system and only eligible for Xclearing deposit an additional 5% discount will apply. Unless otherwise agreed in writing by both parties, at no time will the Investor convert any amount of the June 2013 Note into common stock that would result in the Investor owning more than 4.99% of the common stock outstanding. The Company will reserve at least 3,000,000 shares of Common Stock for issuance upon conversion of the Preferred Stock.
| 46 |
The Company agreed to include all shares issuable upon conversion of the June 2013 Note in its next registration statement filed with the Securities and Exchange Commission.
So long as the June 2013 Note is outstanding, upon any issuance by the Company or any of its subsidiaries of any security with any term more favorable to the holder of such security or with a term in favor of the holder of such security that was not similarly provided to the Investor in the June 2013 Note, then the Company shall notify the Investor of such additional or more favorable term and such term, at the Investor's option, shall become a part of the transaction documents with the Company.
Garden State Securities, Inc. (the “Placement Agent”) served as the placement agent of the Company for the June 2013 Note. In consideration for services rendered as the Placement Agent, the Company and Investor agreed that eight percent (8%) of any amounts advanced under the June 2013 Note would be wired directly to the Placement Agent by the Investor as payment of the Placement Agent’s fee.
Settlement Shares
On July 25, 2013, we issued 7,500,000 shares of our common stock to Hanover Holdings I, LLC, a New York limited liability company (“Hanover”), in full and final settlement, subject to adjustment, of past-due accounts payable of the Company, which Hanover had purchased from a farming supplies vendor of the Company, plus fees and costs (the “Hanover Settlement”).
On September 30, 2013, pursuant to the adjustment provision included in the Hanover Settlement, an additional 2,000,000 shares of our common stock were issued to Hanover based upon the volume weighted average price of our common stock.
The issuance of shares of our common stock to Hanover was exempt from the registration requirements of the Securities Act pursuant to Section 3(a)(10) thereof, as an issuance of securities in exchange for bona fide outstanding claims, where the terms and conditions of such issuance are approved by a court after a hearing upon the fairness of such terms and conditions at which all persons to whom it is proposed to issue securities in such exchange shall have the right to appear.
Asher Note
On August 22, 2013, we issued the Asher Note, pursuant to the terms of a Securities Purchase Agreement. The Note matures on May 26, 2014, incurs interest at the rate of 8% per annum, and is convertible into shares of our common stock at a 35% discount to the average of the lowest three trading prices for our common stock during the 30 day trading period prior to the conversion date. The Asher Note was offered and sold in reliance on the exemption from registration afforded by Section 4(2) and Regulation D (Rule 506) under the Securities Act and corresponding provisions of state securities laws.
Black Mountain Shares
On October 15, 2013, we issued a Convertible Debenture in the principal amount of $58,000 (the “Debenture”), to Black Mountain Equities, Inc. (“Black Mountain”). The Debenture matures on May 1, 2014, incurs a one-time interest charge of 10%, and is convertible into shares of our common stock at a conversion price of $0.20 per share. The Debenture is secured by 1,250,000 shares of our common stock. The Debenture provides that on the next registration statement the Company files, the Company will include the shares issuable upon conversion of the Debenture. Black Mountain also received a warrant to purchase 1,000,000 shares of our common stock, with an exercise price of $0.25 per share and a term of five years. The Debenture was offered and sold in reliance on the exemption from registration afforded by Section 4(2) and Regulation D (Rule 506) under the Securities Act and corresponding provisions of state securities laws.
Note Issuances
On November 21, 2013, we issued a convertible note in the principal amount of $53,000, convertible at 65% of the three lowest bids for 30 trading days before the conversion date with interest at 8% per annum, due on August 25, 2014 and on December 9, 2013, we issued a convertible note in the principal amount of $55,556 with a 10% original issuance discount and 12% one time interest. The convertible notes were offered and sold in reliance on the exemption from registration afforded by Section 4(2) and Regulation D (Rule 506) under the Securities Act and corresponding provisions of state securities laws.
Foster Debenture and Warrants
Effective February 7, 2014, we issued a Convertible Debenture to an investor in the principal amount of $80,000. The Convertible Debenture matures on February 6, 2015, incurs interest at the rate of 8% per annum, and is convertible into shares of our common stock at a conversion price of $0.10 per share. The Convertible Debenture requires that we reserve at least 2,000,000 shares for issuance upon its conversion and provides the holder with participation rights in future financings and piggyback registration rights on the next registration statement the Company files. In connection with the issuance of the Convertible Debenture, the investor also received a Common Stock Purchase Warrant to purchase 1,000,000 shares of our common stock, with an exercise price of $0.10 per share, subject to adjustment, and a term of five years. The Convertible Debenture and Common Stock Purchase Warrant were offered and sold in reliance on the exemption from registration afforded by Section 4(2) and Regulation D (Rule 506) under the Securities Act and corresponding provisions of state securities laws.
| 47 |
Supplemental Warrant
On February 20, 2014, in consideration for Cranshire Capital Master Fund, Ltd.’s (“Cranshire”) immediate cash exercise of an outstanding warrant to purchase common stock of the Company, we agreed to issue Cranshire an additional warrant to purchase 683,202 shares of common stock (the “Supplemental Warrant”). The issuance of the Supplemental Warrant was conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act.
Restricted Stock Awards
On February 26, 2014, we issued an aggregate of 28,300,000 shares of common stock pursuant to restricted stock award agreements to employees and consultants of the Company for services rendered (the “Restricted Shares”). 20,000,000 of the Restricted Shares were issued to Blankenbaker Ventures (Asia) Pte. Ltd. on behalf of George Blankenbaker, the Company’s President and director; 4,000,000 of such shares vest at the time of issuance and the remainder vest over the following four years in equal annual installments. 3,000,000 of the shares were issued to Growers Synergy Pte Ltd., a corporation organized under the laws of Singapore (“Growers Synergy”), all of which were fully vested at the time of issuance. Mr. Blankenbaker is the managing director of Growers Synergy and Growers Fresh Pte Ltd (“Growers Fresh) owns a 51% interest in Growers Synergy and Mr. Blankenbaker controls a 49% interest in Growers Fresh. Thomas Ong, a director of the Company is a director of Growers Synergy andis also a 25% shareholder of Agriventure Pte Ltd., which is a 49% shareholder of Growers Synergy. The issuance of the Restricted Stock was conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act and Regulation S of the Securities Act, in offshore transactions (as defined in Rule 902 under Regulation S of the Securities Act).
Accounts Payable Conversion
On February 26, 2014, the Company agreed to convert an aggregate of approximately $893,579.93 of advances for working capital received from George Blankenbaker, the Company’s President and director, and entities affiliated with Mr. Blankenbaker, into an aggregate of 16,744,682 shares of common stock at a deemed fair market value of $0.053365 per share. The issuance was conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act.
Nomis Bay Transaction
On March 3, 2014, pursuant to a Securities Purchase Agreement (the “Purchase Agreement”) with Nomis Bay Ltd., a Bermuda company (“Nomis Bay”), we issued a senior convertible note with an initial principal amount of $500,000 (the “Initial Convertible Note”) for a purchase price of $340,000. The Purchase Agreement provides that, upon the terms and subject to the conditions set forth therein, we shall have the right to require Nomis Bay to purchase from the Company on or prior to the 10th trading day after the effective date of a registration statement registering the shares issuable upon conversion of the Initial Convertible Note, an additional senior convertible note with an initial principal amount of $600,000 (the “Additional Convertible Note” and, together with the Initial Convertible Note, the “Convertible Notes”) for a purchase price of $600,000.
The Initial Convertible Note matures on December 27, 2014 (subject to extension as provided in the Initial Convertible Note) and, in addition to the 32% original issue discount, accrues interest at the rate of 8% per annum. If issued, the Additional Convertible Note will mature on the date that is the 10-month anniversary of the date of issuance of the Additional Convertible Note (subject to extension as provided in the Initial Convertible Note) and will accrue interest at the rate of 8% per annum. The Initial Convertible Note is convertible at any time, in whole or in part, at Nomis Bay’s option into shares of the Company’s common stock, par value $0.001 per share (the “Common Stock”), at a conversion price equal to the lesser of (i) the product of (x) the arithmetic average of the lowest three (3) volume weighted average prices of the Common Stock during the 10 consecutive trading days ending and including the trading day immediately preceding the applicable conversion date and (y) 40% (the “Variable Conversion Price”), and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). If issued, the Additional Convertible Note will be convertible at any time, in whole or in part, at Nomis Bay’s option into shares of Common Stock at a conversion price that will be equal to the lesser of (i) the Variable Conversion Price and (ii) $0.30 (as adjusted for stock splits, stock dividends, stock combinations or other similar transactions). At no time will Nomis Bay be entitled to convert any portion of the Convertible Notes to the extent that after such conversion, Nomis Bay (together with its affiliates) would beneficially own more than 4.99% of the outstanding shares of Common Stock as of such date.
The issuances were conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act.
Warrant Exercises
On August 1, 2012, the Company issued to Cranshire a warrant to purchase an aggregate of 213,334 shares of the Company’s common stock at an exercise price of $0.6405 with a term of 5 years (the “Cranshire Warrant”). On May 1, 2013, the Company issued to Anson Investments Master Fund LP (“Anson”), three warrants to purchase 1,877,333, 1,066,666 and 2,346,666 shares respectively, at exercise prices of $0.20, $0.25 and $0.25 respectively (the “Anson Warrants” and together with the Cranshire Warrants, the “Investor Warrants”). The Investor Warrants each contained certain adjustment provisions in the event the Company undertook subsequent stock issuances at a price per share less than the exercise price of the Investor Warrants. As of February 20, 2014, as a result of dilutive issuances, the Investor Warrants were each adjusted to an exercise price of $0.053365. As of February 20, 2014, the Cranshire Warrant is exercisable for an aggregate of 2,560,486 shares and the Anson Warrants are exercisable for an aggregate of 7,035,820, 4,997,029 and 10,993,469 shares respectively. As of February 28, 2014 Cranshire had exercised an aggregate of 683,202 shares pursuant to the Cranshire Warrant and Anson had exercised an aggregate of 3,540,659 shares pursuant to the Anson Warrants. The issuances were conducted in reliance upon Regulation D of the Securities Act to investors who are “accredited investors,” as such term is defined in Rule 501(a) under the Securities Act.
| 48 |
Item 16. Exhibit Index
The following exhibits are included as part of this registration statement by reference:
| Number | Description | |
| 2.1 | Share Exchange Agreement, dated June 23, 2011 (incorporated by reference to Exhibit 2.1 of the Registrant’s Current Report on Form 8-K filed on June 29, 2011) | |
| 3.1 | Articles of Incorporation of the Registrant, dated May 18, 2007, including all amendments to date (Incorporated by reference to the Quarterly Report on Form 10-Q filed on November 20, 2013) | |
| 3.2 | Amended and Restated Bylaws of the Registrant, as amended, dated March 18, 2011 (incorporated by reference to Exhibit 3.2 of the Registrant’s Current Report on Form 8-K filed on March 22, 2011) | |
| 4.1 | Specimen Common Stock Certificate (incorporated by reference to Exhibit 4.1 of the Registrant’s Registration Statement on Form S-1 filed on July 16, 2008) | |
| 5.1 | Opinion of Greenberg Traurig, LLP* | |
| 10.1 | Supply Agreement with Asia Stevia Investment Development Company Ltd, dated April 12, 2011 (incorporated by reference to the registrant’s Form 8-K filed on June 29, 2011) | |
| 10.2 | Supply Agreement with Stevia Ventures Corporation, dated April 12, 2011 (incorporated by reference to the registrant’s Form 8-K filed on June 29, 2011) | |
| 10.3 | Convertible Promissory Note, with Vantage Associates SA, dated February 14, 2011 (incorporated by reference to the registrant’s Form 8-K filed on June 29, 2011) | |
| 10.4 | Convertible Promissory Note, with Vantage Associates SA, dated June 23, 2011 (incorporated by reference to the registrant’s Form 8-K filed on June 29, 2011) | |
| 10.5 | Form of Convertible Promissory Note (incorporated by reference to the registrant’s Form 10-Q filed on November 21, 2011) | |
| 10.6 | Stock Purchase Agreement (incorporated by reference to the registrant’s Form 10-Q filed on November 21, 2011) | |
| 10.7 | Management and Off-Take Agreement with Growers Synergy Pte Ltd., effective November 1, 2011 (incorporated by reference to the registrant’s Form 8-K filed on October 31, 2011) |
| 10.8 | The Minutes for Land Transferring Agreement for New Crop Plants Variety, dated December 14, 2011 (incorporated by reference to the registrant’s Form 10-Q filed on February 17, 2012) | |
| 10.9 | Supply Agreement with Guangzhou Health China Technology Development Company Limited, dated February 21, 2012 (incorporated by reference to the registrant’s Form 8-K filed on February 27, 2012) | |
| 10.10 | Cooperative Agreement (incorporated by reference to the registrant’s Current Report on Form 8-K filed on July 11, 2012) | |
| 10.11 | Technology Acquisition Agreement (incorporated by reference to the registrant’s Current Report on Form 8-K filed on July 11, 2012) | |
| 10.12 | Securities Purchase Agreement (incorporated by reference to the Current Report on Form 8-K filed on August 7, 2012) | |
| 49 |
| 10.13 | Registration Rights Agreement (incorporated by reference to the Current Report on Form 8-K filed on August 7, 2012) | |
| 10.14 | Form of Warrant (incorporated by reference to the Current Report on Form 8-K filed on August 7, 2012) | |
| 10.15 | Reset Letter with Anson Investments Master Fund LP, dated May 1, 2013 (incorporated by reference to the Current Report on Form 8-K filed on May 6, 2013) | |
| 10.16 | Form of Warrant (incorporated by reference to the Current Report on Form 8-K filed on May 6, 2013) | |
| 10.17 | Stipulation of Settlement with Hanover Holdings I, LLC, dated July 16, 2013 (incorporated by reference to the Current Report on Form 8-K filed on July 29, 2013) | |
| 10.18 | $400,000 Promissory Note, dated July 16, 2013(incorporated by reference to the Quarterly Report on Form 10-Q filed August 19, 2013) | |
| 10.19 | Form of Senior Convertible Note (incorporated by reference to the Current Report on Form 8-K filed March 4, 2014) | |
| 10.20 | Securities Purchase Agreement, dated as of March 3, 2014, by and between Nomis Bay Ltd. and Stevia Corp. (incorporated by reference to the Current Report on Form 8-K filed March 4, 2014) | |
| 10.21 | Registration Rights Agreement, dated as of March 3, 2014, by and between Nomis Bay Ltd. and Stevia Corp. (incorporated by reference to the Current Report on Form 8-K filed March 4, 2014) | |
| 10.22 | Note Purchase Agreement, dated as of April 2, 2014, by and between Stevia Corp. and YOPCP, LLC (incorporated by reference to the Current Report on Form 8-K filed on April 3, 2014) | |
| 10.23 | Form of Senior Secured Convertible Promissory Note (incorporated by reference to the Current Report on Form 8-K filed on April 3, 2014) | |
| 10.24 | Security Agreement, dated as of April 2, 2014, by and between Stevia Corp. and YOPCP, LLC (incorporated by reference to the Current Report on Form 8-K filed on April 3, 2014) | |
| 21 | List of Subsidiaries * | |
| 23.1 | Consent of Li and Company, PC* | |
| 23.2 | Consent of Greenberg Traurig, LLP (filed as part of Exhibit 5.1)* | |
| 24 | Power of Attorney (included on the signature page of this Registration Statement)* | |
| 101 | Interactive Data File* |
*Filed Herewith
Item 17. Undertakings
The undersigned registrant hereby undertakes to:
(a) Rule 415 Offering :
1. To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
i. To include any prospectus required by section 10(a)(3) of the Securities Act of 1933;
ii. To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) (§ 230.424 of this chapter) if, in the aggregate, the changes in volume and price represent no more than 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement.
iii. To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement;
Provided, however, that:
(A) Paragraphs (a)(1)(i) and (a)(1)(ii) of this section do not apply if the registration statement is on Form S-8 (§ 239.16b of this chapter), and the information required to be included in a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant to section 13 or section 15(d) of the Securities Exchange Act of 1934 (15 U.S.C. 78m or 78o(d)) that are incorporated by reference in the registration statement; and
| 50 |
(B) Paragraphs (a)(1)(i), (a)(1)(ii) and (a)(1)(iii) of this section do not apply if the registration statement is on Form S-3 (§ 239.13 of this chapter) or Form F-3 (§ 239.33 of this chapter) and the information required to be included in a post-effective amendment by those paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant to section 13 or section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference in the registration statement, or is contained in a form of prospectus filed pursuant to Rule 424(b) (§ 230.424(b) of this chapter) that is part of the registration statement.
(C) Provided further, however, that paragraphs (a)(1)(i) and (a)(1)(ii) do not apply if the registration statement is for an offering of asset-backed securities on Form S-1 (§ 239.11 of this chapter) or Form S-3 (§ 239.13 of this chapter), and the information required to be included in a post-effective amendment is provided pursuant to Item 1100(c) of Regulation AB (§ 229.1100(c)).
2. That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
3. To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
4. If the registrant is a foreign private issuer, to file a post-effective amendment to the registration statement to include any financial statements required by “Item 8.A. of Form 20-F (17 CFR 249.220f)” at the start of any delayed offering or throughout a continuous offering. Financial statements and information otherwise required by Section 10(a)(3) of the Act need not be furnished, provided that the registrant includes in the prospectus, by means of a post-effective amendment, financial statements required pursuant to this paragraph (a)(4) and other information necessary to ensure that all other information in the prospectus is at least as current as the date of those financial statements. Notwithstanding the foregoing, with respect to registration statements on Form F-3 (§239.33 of this chapter), a post-effective amendment need not be filed to include financial statements and information required by Section 10(a)(3) of the Act or §210.3-19 of this chapter if such financial statements and information are contained in periodic reports filed with or furnished to the Commission by the registrant pursuant to section 13 or section 15(d) of the Securities Exchange Act of 1934 that are incorporated by reference in the Form F-3.
5. That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser:
i. If the registrant is relying on Rule 430B (§230.430B of this chapter):
(A) Each prospectus filed by the registrant pursuant to Rule 424(b)(3) (§230.424(b)(3) of this chapter) shall be deemed to be part of the registration statement as of the date the filed prospectus was deemed part of and included in the registration statement; and
(B) Each prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) (§230.424(b)(2), (b)(5), or (b)(7) of this chapter) as part of a registration statement in reliance on Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) (§230.415(a)(1)(i), (vii), or (x) of this chapter) for the purpose of providing the information required by section 10(a) of the Securities Act of 1933 shall be deemed to be part of and included in the registration statement as of the earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such effective date, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such effective date; or
ii. If the registrant is subject to Rule 430C (§230.430C of this chapter), each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A (§230.430A of this chapter), shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
6. That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities:
The undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser:
| 51 |
i. Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424 (§230.424 of this chapter);
ii. Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
iii. The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and
iv. Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
Insofar as indemnification for liabilities arising under the Securities Act of 1933 may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Act and will be governed by the final adjudication of such issue.
| 52 |
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, the registrant has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized in the City of Indianapolis, State of Indiana, on April 8, 2014.
| STEVIA CORP. | |
| a Nevada corporation | |
| Dated: April 8, 2014 | /s/ George Blankenbaker |
| By: George Blankenbaker | |
| Its: President, Secretary, Treasurer and Director | |
| (Principal Executive Officer, Principal Financial Officer and Principal Accounting Officer) |
SIGNATURES AND POWER OF ATTORNEY
Known All Persons By These Present, that each person whose signature appears below appoints Mr. George Blankenbaker as his true and lawful attorney-in-fact and agent, with full power of substitution, for him and in his name, place and stead, to sign any amendment (including post-effective amendments) to this registration statement, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact and agent, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he may do in person, hereby ratifying and confirming all that said attorney-in-fact and agent or any of them, or of his/her substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated:
| Dated: April 8, 2014 | /s/ George Blankenbaker |
| George Blankenbaker | |
| President, Secretary, Treasurer and Director (Principal Executive Officer, Principal Financial Officer and Principal Accounting Officer) | |
| Dated: April 8, 2014 | /s/ Pablo Erat |
| Pablo Erat | |
| Director | |
| Dated: April 8, 2014 | /s/ Thomas Ong |
| Thomas Ong | |
| Director |
| 53 |