
Proteostasis Therapeutics, Inc. (PTI) October 2018 Investor Deck Exhibit 99.1

Safe Harbor and Disclaimer To the extent that statements in this presentation are not historical facts, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “aim,” “may,” “will,” “expect,” “anticipate,” “estimate,” “intend,” and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. Examples of forward-looking statements made in this presentation include, without limitation, statements regarding the expected timing of the initiation of, patient enrollment in, data from, and our completion of, our clinical studies and cohorts for PTI-428, PTI-801, PTI-808 and our combination therapy candidates as well as cash guidance. Forward-looking statements made in this presentation involve substantial risks and uncertainties that could cause actual results to differ materially from those expressed or implied by the forward-looking statements, and we therefore cannot assure you that our plans, intentions, expectations or strategies will be attained or achieved. Such risks and uncertainties include, without limitation, the possibility final or future results from our drug candidate trials (including, without limitation, longer duration studies) do not achieve positive results or are materially and negatively different from or not indicative of the preliminary results reported in this presentation (noting that these results are on a small number of patients and small data set), uncertainties inherent in the execution and completion of clinical trials (including, without limitation, the possibility FDA requires us to run cohorts sequentially or conduct additional cohorts or pre-clinical or clinical studies), in the enrollment of CF patients in our clinical trials, in the timing of availability of trial data, in the results of the clinical trials, in possible adverse events from our trials, in the actions of regulatory agencies, in endorsement, if any, by therapeutic development arms of CF patient advocacy groups, and those set forth in our Quarterly Report on Form 10-Q for the quarter ended June 30, 2018, and our other SEC filings. We assume no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. This presentation also contains estimates and other statistical data made by independent parties and by us relating to, among other items, disease incidence, market size and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. Projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of risk and uncertainty. New risks emerge from time to time, and neither we nor any other person makes any representation as to the accuracy or completeness of such data or undertakes any obligation to update such date after the date of this presentation. By attending or receiving this presentation you acknowledge you are solely responsible for your own assessment of the market and our market position and that you will conduct your own analysis and are solely responsible for forming your own view of the potential future performance of our business. The trademarks included in this presentation are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of the Company or its securities.

Introduction to Proteostasis Therapeutics (PTI) Clinical stage biopharmaceutical company developing small molecule CFTR modulators for Cystic Fibrosis (CF) PTI-428: Amplifier (increases unfolded CFTR protein) with POC as ORKAMBI® add on PTI-801: Corrector (increases folded CFTR protein) with POC as ORKAMBI® add on PTI-808: Potentiator (improves ion transport across the CFTR protein) Drugs can be combined as either add-ons or proprietary combinations to offer potential therapeutic benefit PTI-428 and PTI-801 as add-on therapies to current and future standard of care CFTR modulators; SYMDEKO® add on studies underway PTI-801 and PTI-808 as a stand-alone doublet PTI-428, PTI-801, and PTI-808 as a stand-alone triplet As of June 30, 2018, PTI has $60.8 million in cash, cash equivalents and short-term investments Cash position sufficient to fund operations into early 2019 ORKAMBI® and SYMDEKO® are registered trademarks of Vertex Pharmaceuticals Inc.

CORRECTOR PTI-801 PTI CF Portfolio Address 3 Distinct Regions of CFTR Protein Processing PTI-808 increases the opening time of the CFTR channel, which results in higher ion flow; novel and KALYDECO®-like POTENTIATOR PTI-808 AMPLIFIER PTI-428 PTI-808 Potentiator PTI-801 facilitates the processing of mutated CFTR protein, which leads to improved delivery of the channel to the cell membrane; additive to first and second generation correctors PTI-801 Corrector PTI-428 Amplifier PTI-428 selectively increases the amount of immature CFTR protein in the cell, which provides additional substrate for correctors and potentiators to act upon KALYDECO® is registered trademark of Vertex Pharmaceuticals Inc.
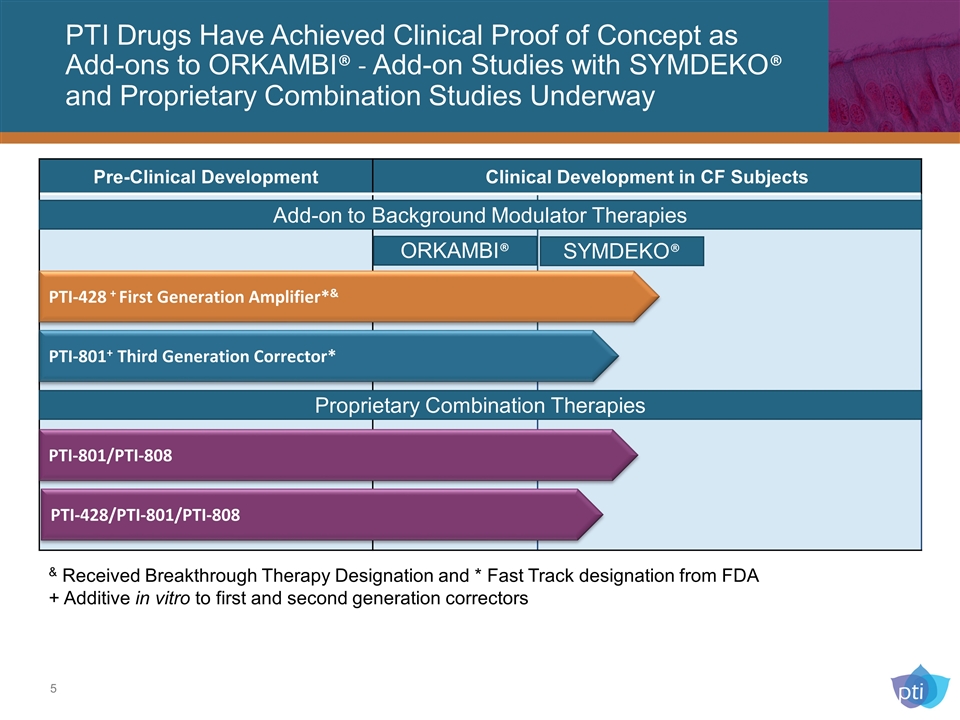
PTI Drugs Have Achieved Clinical Proof of Concept as Add-ons to ORKAMBI® - Add-on Studies with SYMDEKO® and Proprietary Combination Studies Underway Pre-Clinical Development Clinical Development in CF Subjects PTI-428 + First Generation Amplifier*& PTI-801+ Third Generation Corrector* & Received Breakthrough Therapy Designation and * Fast Track designation from FDA + Additive in vitro to first and second generation correctors Proprietary Combination Therapies PTI-428/PTI-801/PTI-808 PTI-801/PTI-808 Add-on to Background Modulator Therapies ORKAMBI® SYMDEKO®
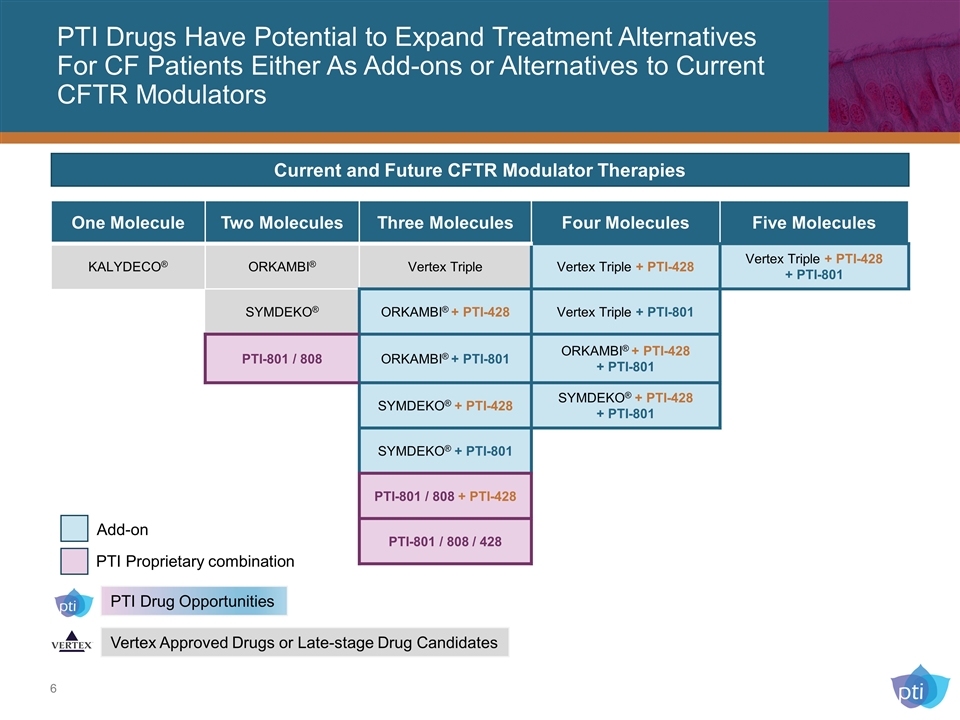
PTI Drugs Have Potential to Expand Treatment Alternatives For CF Patients Either As Add-ons or Alternatives to Current CFTR Modulators One Molecule Two Molecules Three Molecules Four Molecules Five Molecules KALYDECO® ORKAMBI® Vertex Triple Vertex Triple + PTI-428 Vertex Triple + PTI-428 + PTI-801 SYMDEKO® ORKAMBI® + PTI-428 Vertex Triple + PTI-801 PTI-801 / 808 ORKAMBI® + PTI-801 ORKAMBI® + PTI-428 + PTI-801 SYMDEKO® + PTI-428 SYMDEKO® + PTI-428 + PTI-801 SYMDEKO® + PTI-801 PTI-801 / 808 + PTI-428 PTI-801 / 808 / 428 PTI Drug Opportunities Vertex Approved Drugs or Late-stage Drug Candidates Current and Future CFTR Modulator Therapies Add-on PTI Proprietary combination
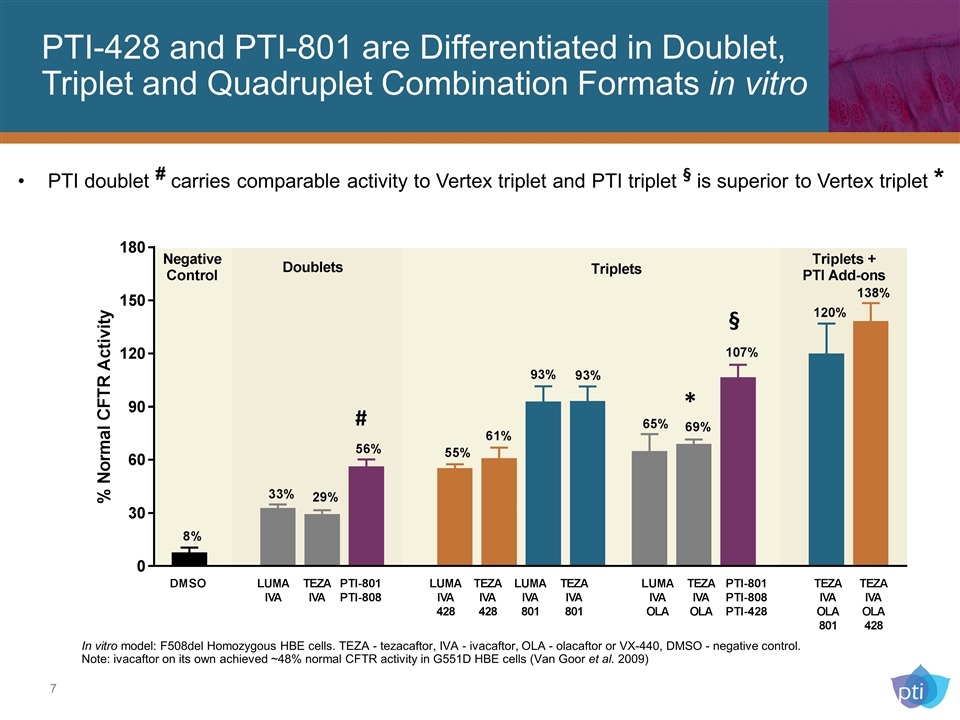
PTI-428 and PTI-801 are Differentiated in Doublet, Triplet and Quadruplet Combination Formats in vitro PTI doublet # carries comparable activity to Vertex triplet and PTI triplet § is superior to Vertex triplet * In vitro model: F508del Homozygous HBE cells. TEZA - tezacaftor, IVA - ivacaftor, OLA - olacaftor or VX-440, DMSO - negative control. Note: ivacaftor on its own achieved ~48% normal CFTR activity in G551D HBE cells (Van Goor et al. 2009) § * #
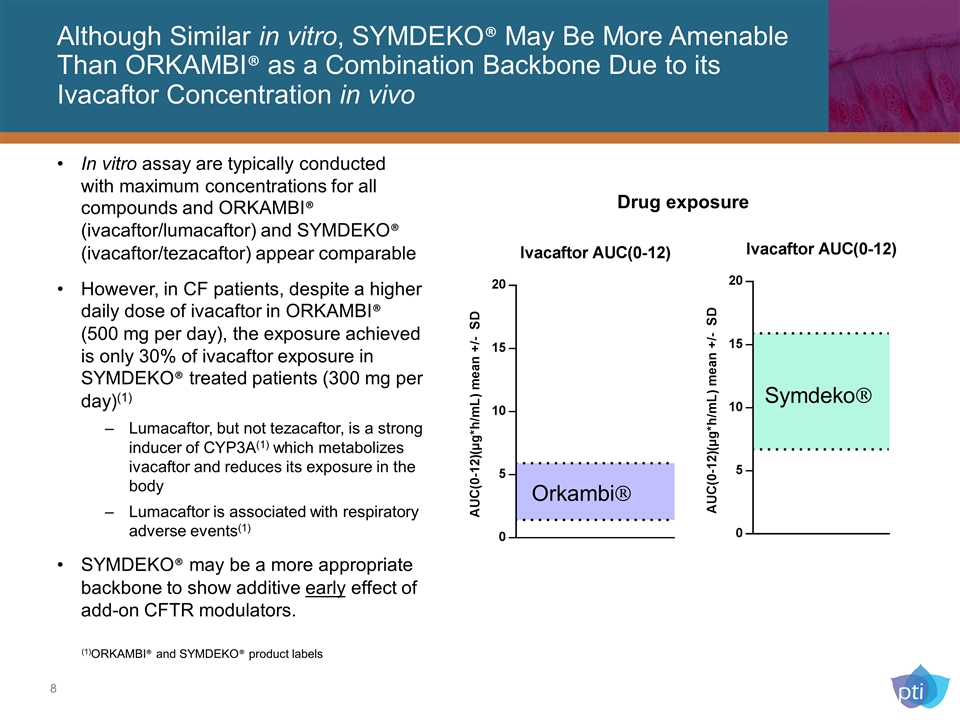
Although Similar in vitro, SYMDEKO® May Be More Amenable Than ORKAMBI® as a Combination Backbone Due to its Ivacaftor Concentration in vivo In vitro assay are typically conducted with maximum concentrations for all compounds and ORKAMBI® (ivacaftor/lumacaftor) and SYMDEKO® (ivacaftor/tezacaftor) appear comparable However, in CF patients, despite a higher daily dose of ivacaftor in ORKAMBI® (500 mg per day), the exposure achieved is only 30% of ivacaftor exposure in SYMDEKO® treated patients (300 mg per day)(1) Lumacaftor, but not tezacaftor, is a strong inducer of CYP3A(1) which metabolizes ivacaftor and reduces its exposure in the body Lumacaftor is associated with respiratory adverse events(1) SYMDEKO® may be a more appropriate backbone to show additive early effect of add-on CFTR modulators. (1)ORKAMBI® and SYMDEKO® product labels Drug exposure

CF Study Updates

PTI-801 Unaffiliated Orkambi Patients Recruited to PTI Clinical Studies Have Experienced a ppFEV1 Decline Before Study Start Comparable to PROGRESS Outcomes(1) PTI has recruited ~100 Subjects on ORKAMBI® for add on clinical studies with PTI-801 and PTI-428 PTI-428 + ORKAMBI®, PTI’s first study in subjects on background ORKAMBI® therapy, recruited subjects in the US that have been on ORKAMBI® for an average of 1.8 years PTI-801 + ORKAMBI®, PTI’s second study in subjects on background ORKAMBI® therapy, recruited subjects in the US that have been on ORKAMBI® for an average of 2.3 years As a group, screened subjects experienced a lung function decline of approximately 3 ppFEV1 per year compared to the first 6 months of ORKAMBI® treatment, suggesting a higher than previously described annual lung function decline despite CFTR modulator therapy PTI-801 CFF TDN PTI-801 ECFS CTN PTI-428 CFF TDN Overlapping CFF TDN Sites PTI-428 Unaffiliated Overlapping Unaffiliated Sites PTI-428 ECFS CTN Overlapping ECFS CTN Sites (1) MW Konstan et al. The Lancet Respiratory Medicine, 2017, Volume 5 , Issue 2 , 107 – 118
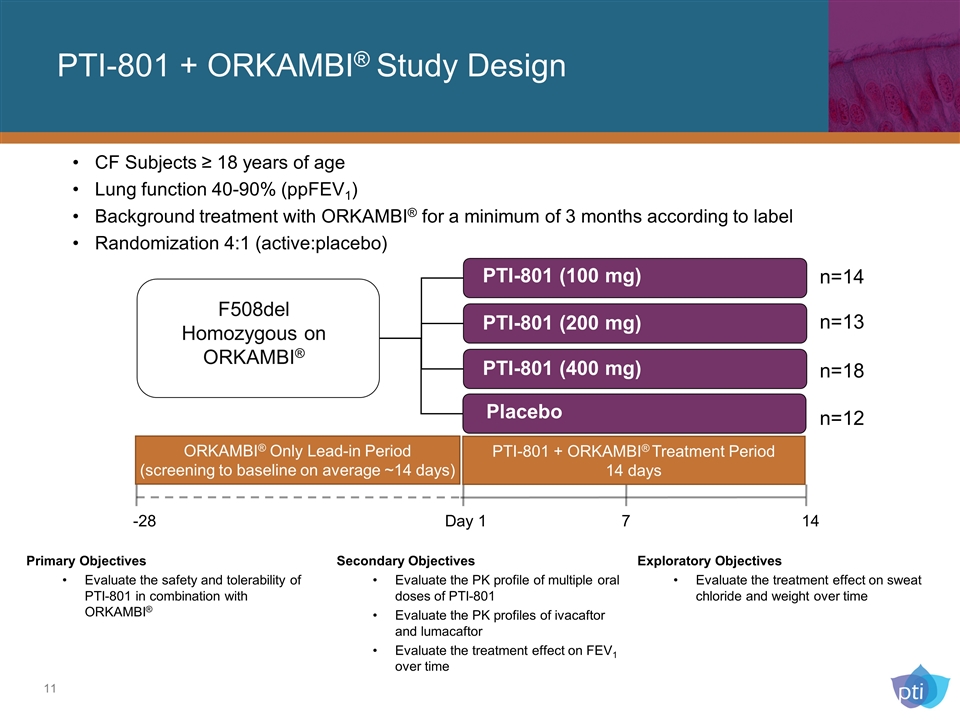
CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Background treatment with ORKAMBI® for a minimum of 3 months according to label Randomization 4:1 (active:placebo) Placebo F508del Homozygous on ORKAMBI® -28 Day 1 7 14 PTI-801 (400 mg) PTI-801 (200 mg) PTI-801 (100 mg) n=14 n=13 n=18 n=12 Primary Objectives Evaluate the safety and tolerability of PTI-801 in combination with ORKAMBI® Exploratory Objectives Evaluate the treatment effect on sweat chloride and weight over time Secondary Objectives Evaluate the PK profile of multiple oral doses of PTI-801 Evaluate the PK profiles of ivacaftor and lumacaftor Evaluate the treatment effect on FEV1 over time PTI-801 + ORKAMBI® Study Design ORKAMBI® Only Lead-in Period (screening to baseline on average ~14 days) PTI-801 + ORKAMBI® Treatment Period 14 days
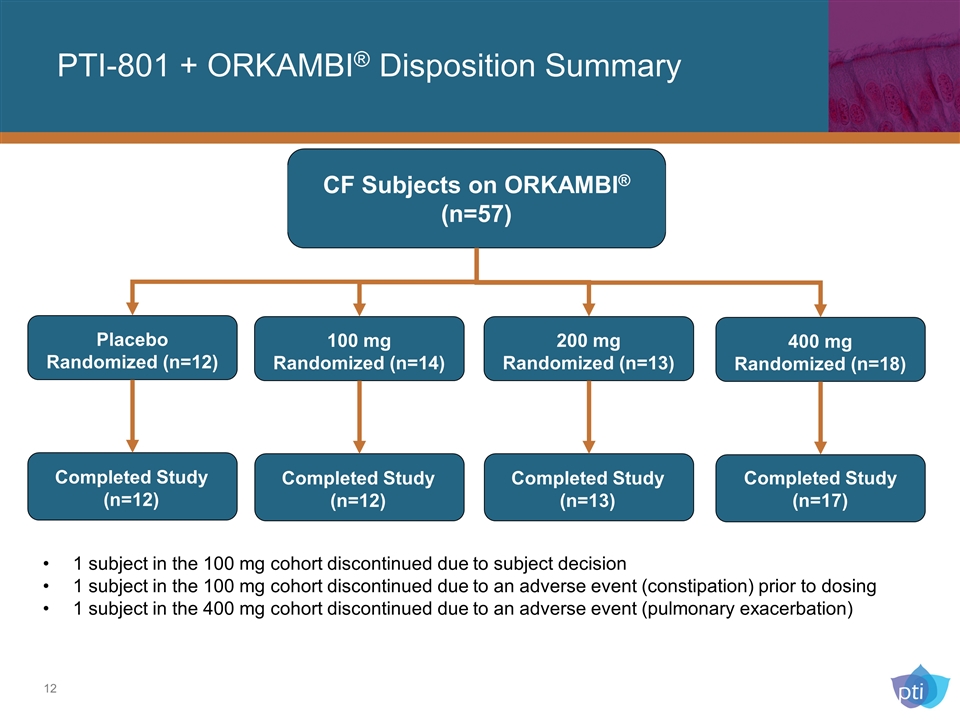
PTI-801 + ORKAMBI® Disposition Summary Placebo Randomized (n=12) Completed Study (n=12) CF Subjects on ORKAMBI® (n=57) 100 mg Randomized (n=14) Completed Study (n=12) 200 mg Randomized (n=13) Completed Study (n=13) 400 mg Randomized (n=18) Completed Study (n=17) 1 subject in the 100 mg cohort discontinued due to subject decision 1 subject in the 100 mg cohort discontinued due to an adverse event (constipation) prior to dosing 1 subject in the 400 mg cohort discontinued due to an adverse event (pulmonary exacerbation)
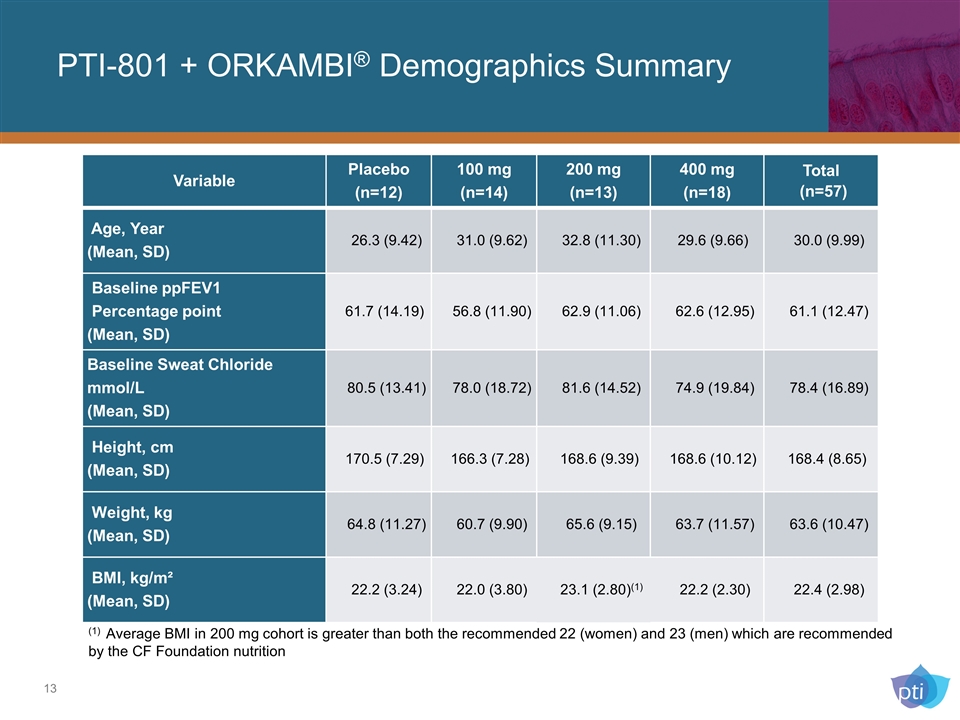
PTI-801 + ORKAMBI® Demographics Summary Variable Placebo (n=12) 100 mg (n=14) 200 mg (n=13) 400 mg (n=18) Total (n=57) Age, Year (Mean, SD) 26.3 (9.42) 31.0 (9.62) 32.8 (11.30) 29.6 (9.66) 30.0 (9.99) Baseline ppFEV1 Percentage point (Mean, SD) 61.7 (14.19) 56.8 (11.90) 62.9 (11.06) 62.6 (12.95) 61.1 (12.47) Baseline Sweat Chloride mmol/L (Mean, SD) 80.5 (13.41) 78.0 (18.72) 81.6 (14.52) 74.9 (19.84) 78.4 (16.89) Height, cm (Mean, SD) 170.5 (7.29) 166.3 (7.28) 168.6 (9.39) 168.6 (10.12) 168.4 (8.65) Weight, kg (Mean, SD) 64.8 (11.27) 60.7 (9.90) 65.6 (9.15) 63.7 (11.57) 63.6 (10.47) BMI, kg/m² (Mean, SD) 22.2 (3.24) 22.0 (3.80) 23.1 (2.80)(1) 22.2 (2.30) 22.4 (2.98) (1) Average BMI in 200 mg cohort is greater than both the recommended 22 (women) and 23 (men) which are recommended by the CF Foundation nutrition
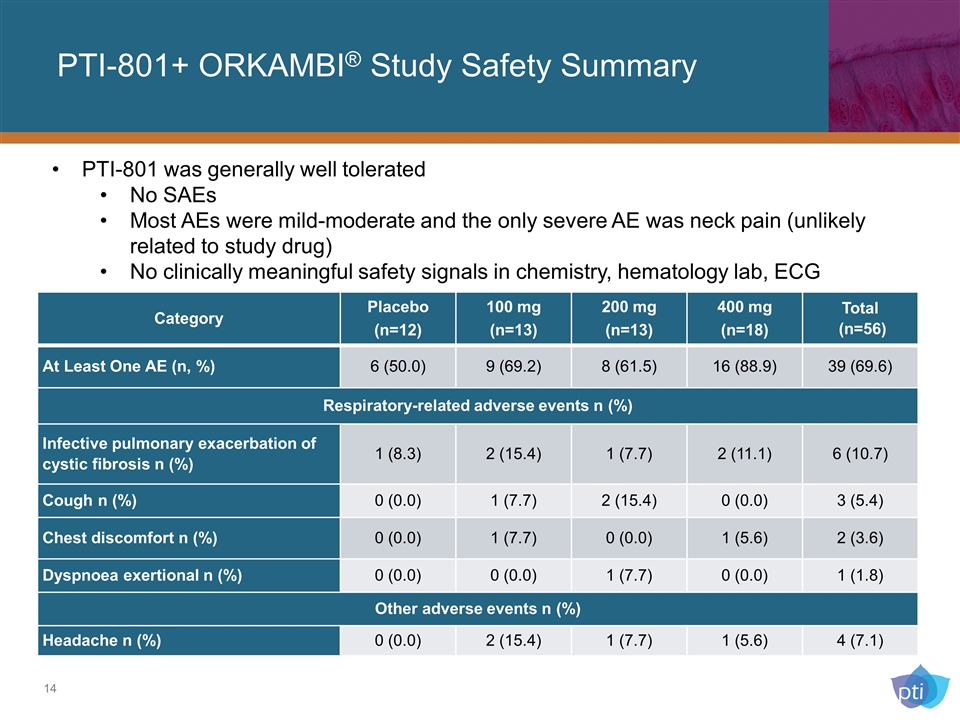
PTI-801+ ORKAMBI® Study Safety Summary PTI-801 was generally well tolerated No SAEs Most AEs were mild-moderate and the only severe AE was neck pain (unlikely related to study drug) No clinically meaningful safety signals in chemistry, hematology lab, ECG Category Placebo (n=12) 100 mg (n=13) 200 mg (n=13) 400 mg (n=18) Total (n=56) At Least One AE (n, %) 6 (50.0) 9 (69.2) 8 (61.5) 16 (88.9) 39 (69.6) Respiratory-related adverse events n (%) Infective pulmonary exacerbation of cystic fibrosis n (%) 1 (8.3) 2 (15.4) 1 (7.7) 2 (11.1) 6 (10.7) Cough n (%) 0 (0.0) 1 (7.7) 2 (15.4) 0 (0.0) 3 (5.4) Chest discomfort n (%) 0 (0.0) 1 (7.7) 0 (0.0) 1 (5.6) 2 (3.6) Dyspnoea exertional n (%) 0 (0.0) 0 (0.0) 1 (7.7) 0 (0.0) 1 (1.8) Other adverse events n (%) Headache n (%) 0 (0.0) 2 (15.4) 1 (7.7) 1 (5.6) 4 (7.1)
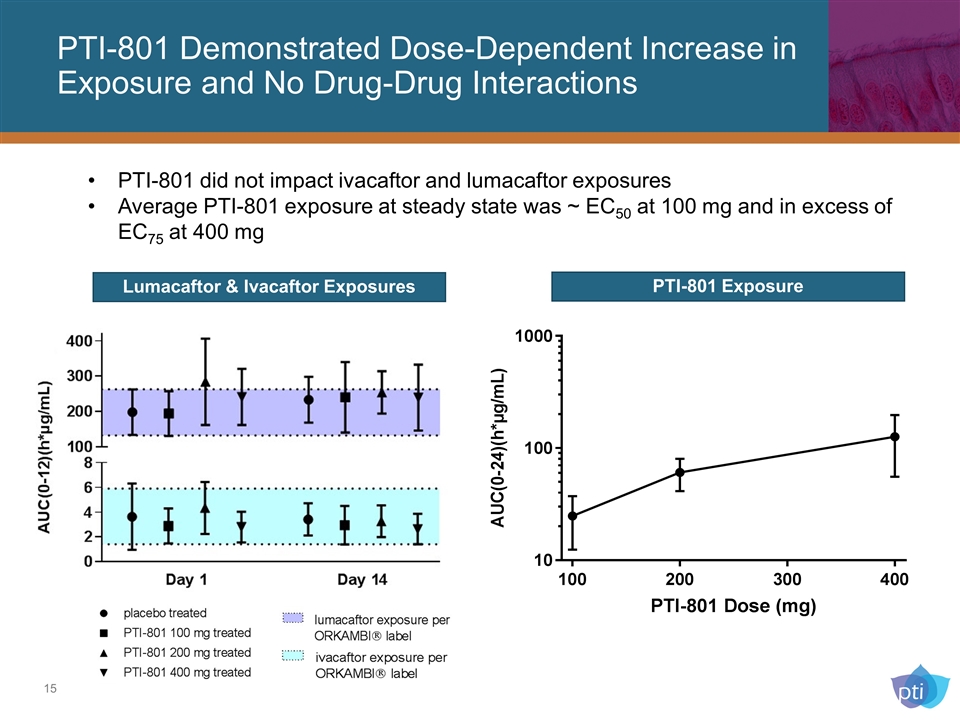
PTI-801 did not impact ivacaftor and lumacaftor exposures Average PTI-801 exposure at steady state was ~ EC50 at 100 mg and in excess of EC75 at 400 mg PTI-801 Demonstrated Dose-Dependent Increase in Exposure and No Drug-Drug Interactions Lumacaftor & Ivacaftor Exposures PTI-801 Exposure
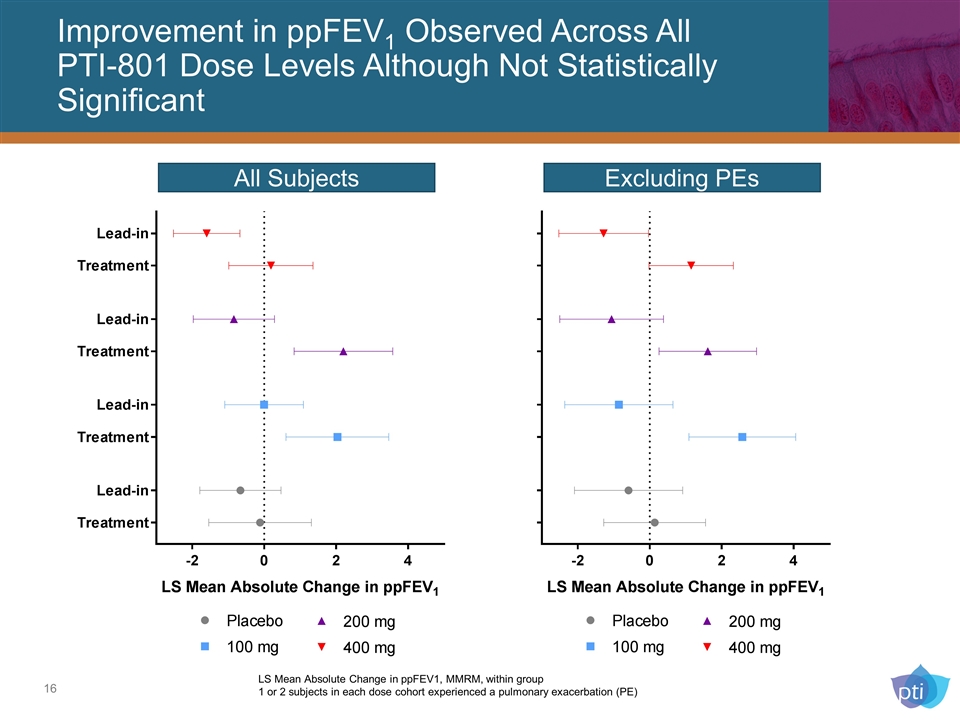
Improvement in ppFEV1 Observed Across All PTI-801 Dose Levels Although Not Statistically Significant All Subjects Excluding PEs LS Mean Absolute Change in ppFEV1, MMRM, within group 1 or 2 subjects in each dose cohort experienced a pulmonary exacerbation (PE)
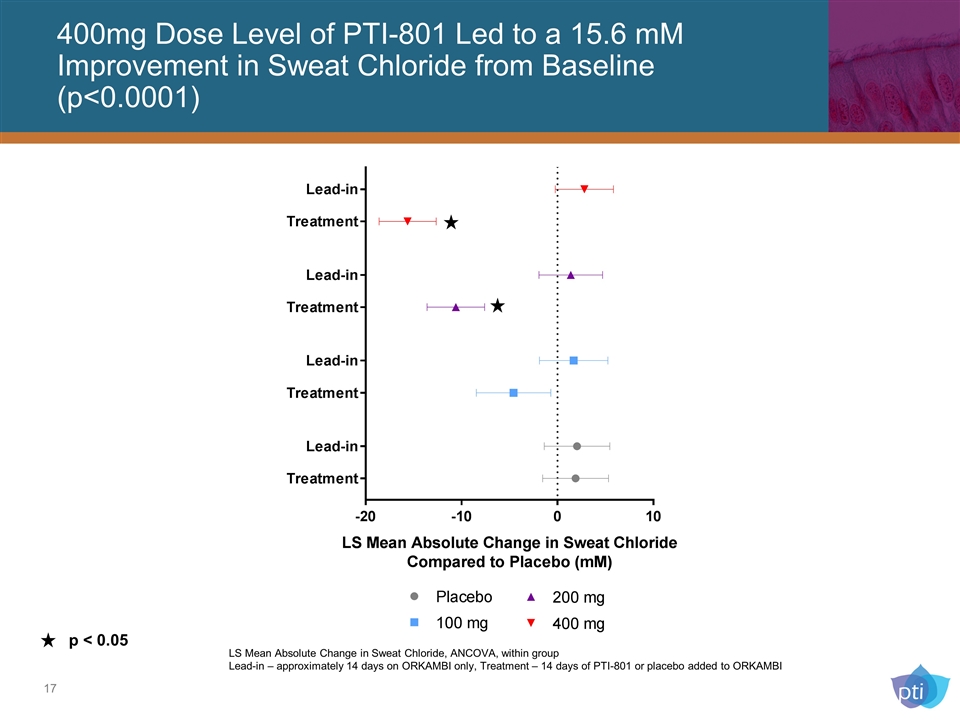
p < 0.05 LS Mean Absolute Change in Sweat Chloride, ANCOVA, within group Lead-in – approximately 14 days on ORKAMBI only, Treatment – 14 days of PTI-801 or placebo added to ORKAMBI 400mg Dose Level of PTI-801 Led to a 15.6 mM Improvement in Sweat Chloride from Baseline (p<0.0001)
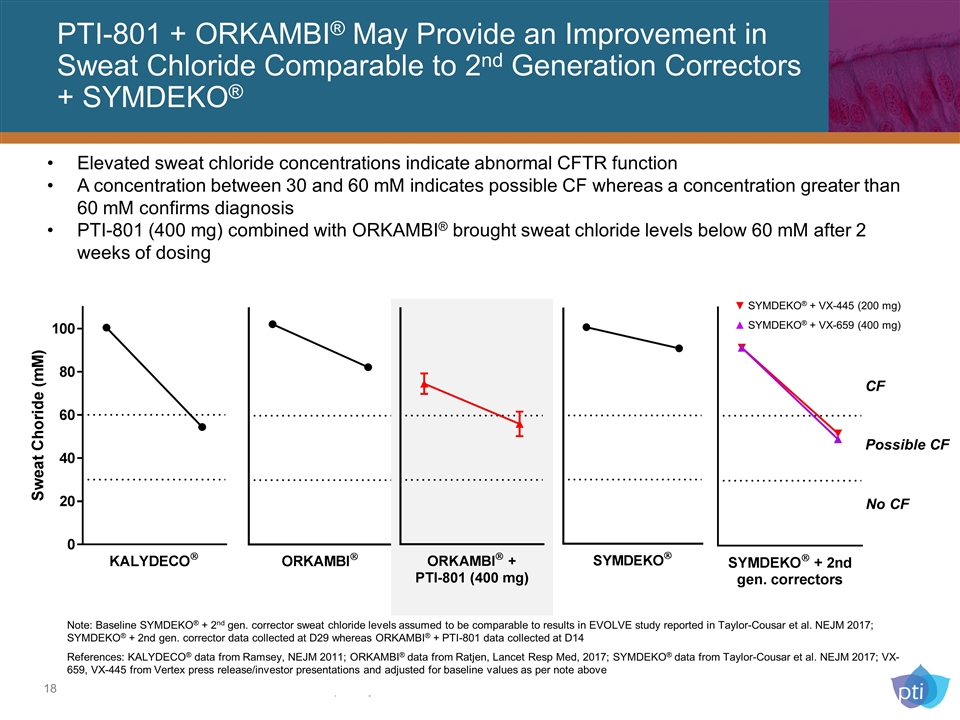
PTI-801 + ORKAMBI® May Provide an Improvement in Sweat Chloride Comparable to 2nd Generation Correctors + SYMDEKO® Elevated sweat chloride concentrations indicate abnormal CFTR function A concentration between 30 and 60 mM indicates possible CF whereas a concentration greater than 60 mM confirms diagnosis PTI-801 (400 mg) combined with ORKAMBI® brought sweat chloride levels below 60 mM after 2 weeks of dosing CF Possible CF No CF Note: Baseline SYMDEKO® + 2nd gen. corrector sweat chloride levels assumed to be comparable to results in EVOLVE study reported in Taylor-Cousar et al. NEJM 2017; SYMDEKO® + 2nd gen. corrector data collected at D29 whereas ORKAMBI® + PTI-801 data collected at D14 References: KALYDECO® data from Ramsey, NEJM 2011; ORKAMBI® data from Ratjen, Lancet Resp Med, 2017; SYMDEKO® data from Taylor-Cousar et al. NEJM 2017; VX-659, VX-445 from Vertex press release/investor presentations and adjusted for baseline values as per note above ▼ SYMDEKO® + VX-445 (200 mg) ▲ SYMDEKO® + VX-659 (400 mg)
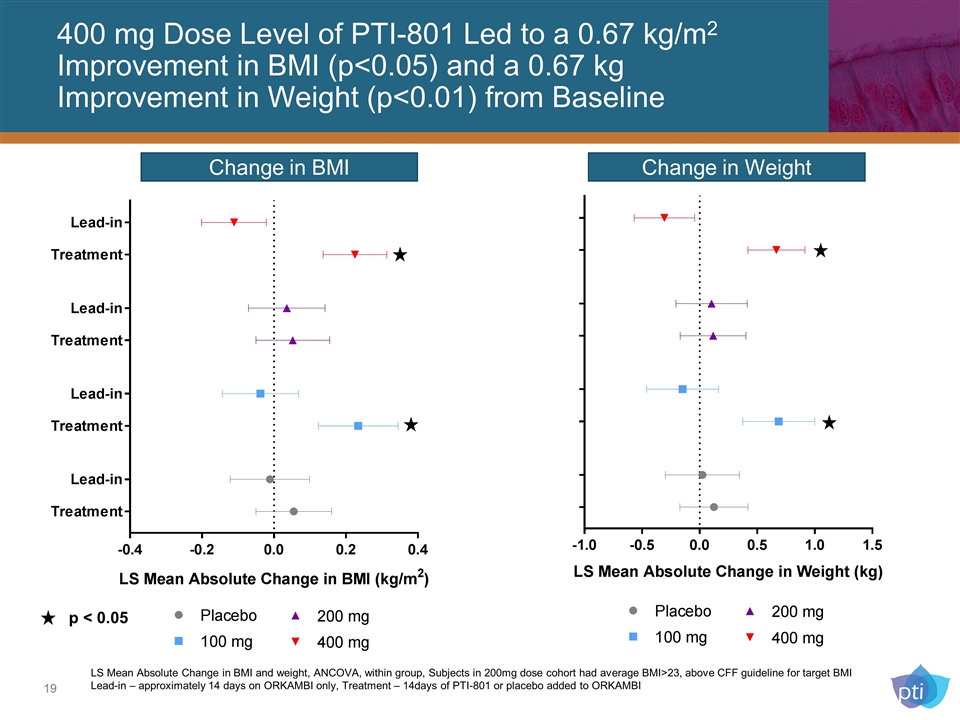
400 mg Dose Level of PTI-801 Led to a 0.67 kg/m2 Improvement in BMI (p<0.05) and a 0.67 kg Improvement in Weight (p<0.01) from Baseline Change in BMI Change in Weight p < 0.05 LS Mean Absolute Change in BMI and weight, ANCOVA, within group, Subjects in 200mg dose cohort had average BMI>23, above CFF guideline for target BMI Lead-in – approximately 14 days on ORKAMBI only, Treatment – 14days of PTI-801 or placebo added to ORKAMBI
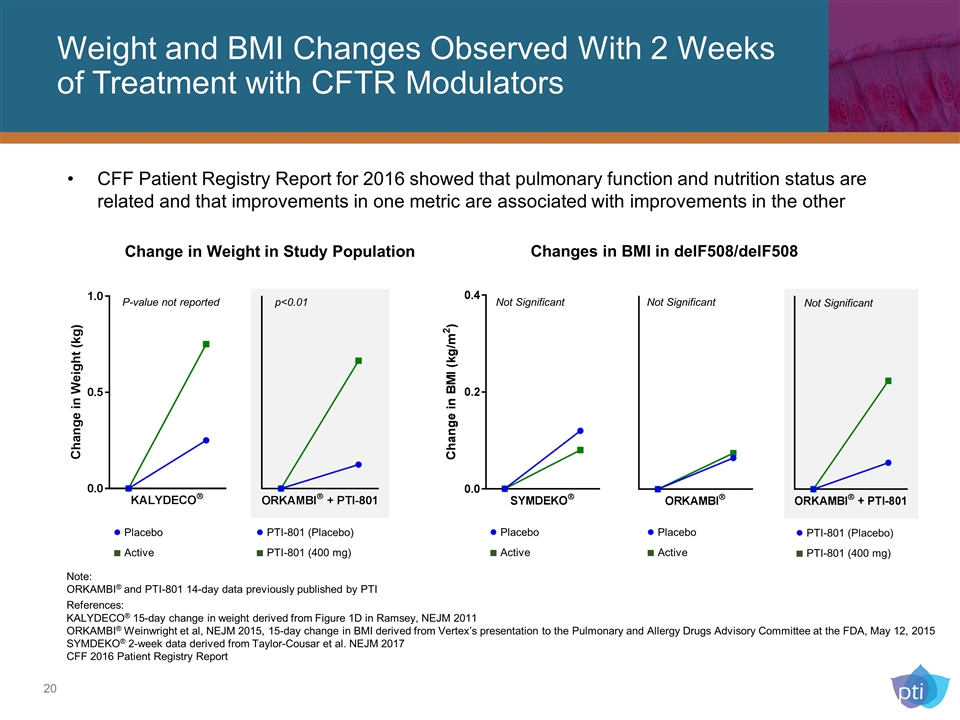
Weight and BMI Changes Observed With 2 Weeks of Treatment with CFTR Modulators Change in Weight in Study Population Changes in BMI in delF508/delF508 Note: ORKAMBI® and PTI-801 14-day data previously published by PTI References: KALYDECO® 15-day change in weight derived from Figure 1D in Ramsey, NEJM 2011 ORKAMBI® Weinwright et al, NEJM 2015, 15-day change in BMI derived from Vertex’s presentation to the Pulmonary and Allergy Drugs Advisory Committee at the FDA, May 12, 2015 SYMDEKO® 2-week data derived from Taylor-Cousar et al. NEJM 2017 CFF 2016 Patient Registry Report Not Significant Not Significant ● PTI-801 (Placebo) ■ PTI-801 (400 mg) p<0.01 P-value not reported ● Placebo ■ Active ● Placebo ■ Active ● Placebo ■ Active ● PTI-801 (Placebo) ■ PTI-801 (400 mg) Not Significant CFF Patient Registry Report for 2016 showed that pulmonary function and nutrition status are related and that improvements in one metric are associated with improvements in the other
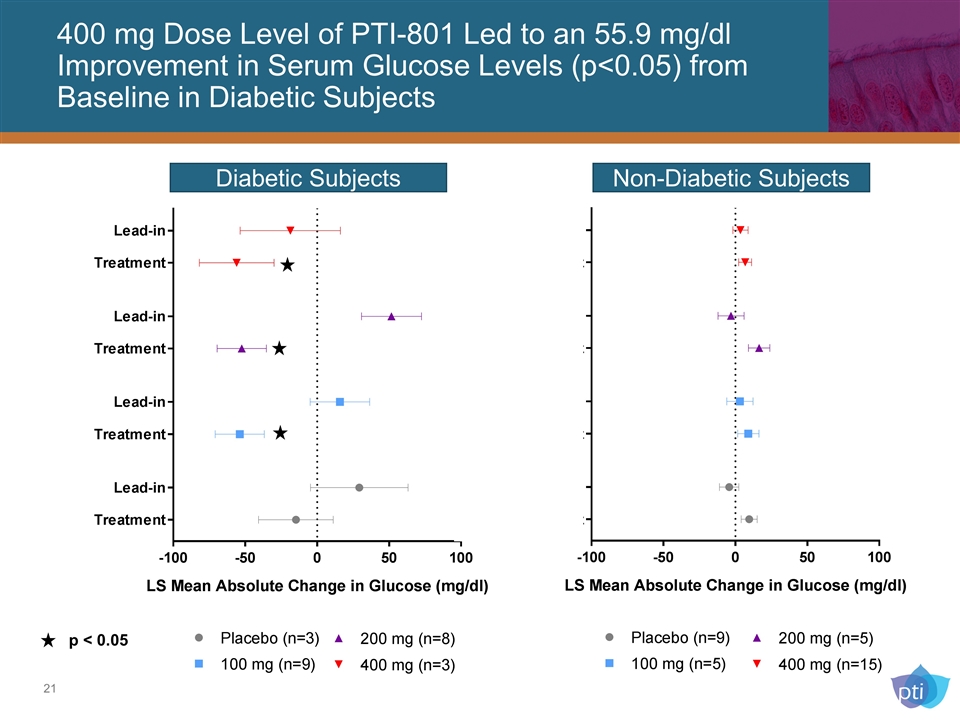
Diabetic Subjects Non-Diabetic Subjects p < 0.05 400 mg Dose Level of PTI-801 Led to an 55.9 mg/dl Improvement in Serum Glucose Levels (p<0.05) from Baseline in Diabetic Subjects

Life expectancy for CF has increased drastically over the past 2 decades The rise in life expectancy has led to an increase in comorbidities with CFRD being the most common one 12,000 CF patients have been diagnosed with CFRD in the US and Europe CFRD profoundly affects nutritional status and weight, resulting in higher morbidity than in the general CF population Preclinical studies suggest that CFTR may play a role in dysfunctional insulin-secreting β-cells In small studies, ivacaftor has been shown to improve diabetes related outcomes in CF patients. No conclusive evidence is available that ivacaftor/lumacaftor can lead to improved glucose control in CFRD patients Cystic Fibrosis-Related Diabetes (CFRD) Cystic Fibrosis Foundation (CFF) Report 2016., European Cystic Fibrosis Society (ECFS) Report 2016. M.S. Stalvey, et al. Cystic fibrosis transmembrane conductance regulator deficiency exacerbates islet cell dysfunction after beta-cell injury, Diabetes, 2006 Hayes D Jr, et al. Resolution of cystic fibrosis-related diabetes with ivacaftor therapy. American Journal of Respiratory and Critical Care Medicine, 2014 Bellin MD, et al. Insulin secretion improves in cystic fibrosis following ivacaftor correction of CFTR: a small pilot study. Pediatr Diabetes, 2013 JC Thomassen et al, Effect of Lumacaftor/Ivacaftor on glucose metabolism and insulin secretion in Phe508del homozygous cystic fibrosis patients, Journal of CF 2018
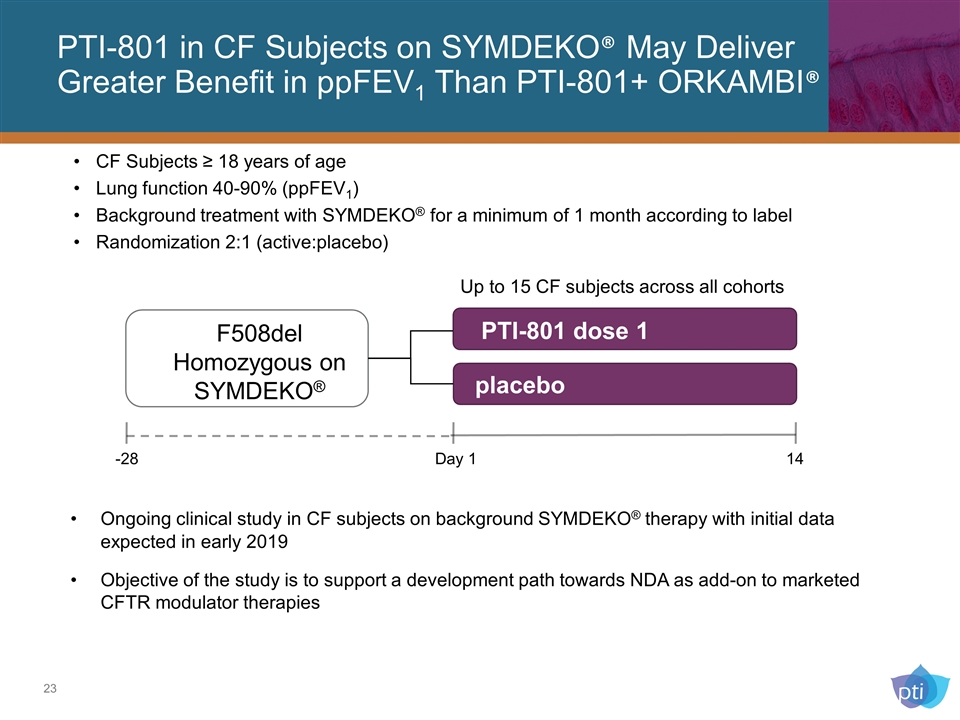
PTI-801 in CF Subjects on SYMDEKO® May Deliver Greater Benefit in ppFEV1 Than PTI-801+ ORKAMBI® PTI-801 dose 1 F508del Homozygous on SYMDEKO® -28 Day 1 14 placebo Ongoing clinical study in CF subjects on background SYMDEKO® therapy with initial data expected in early 2019 Objective of the study is to support a development path towards NDA as add-on to marketed CFTR modulator therapies Up to 15 CF subjects across all cohorts CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Background treatment with SYMDEKO® for a minimum of 1 month according to label Randomization 2:1 (active:placebo)
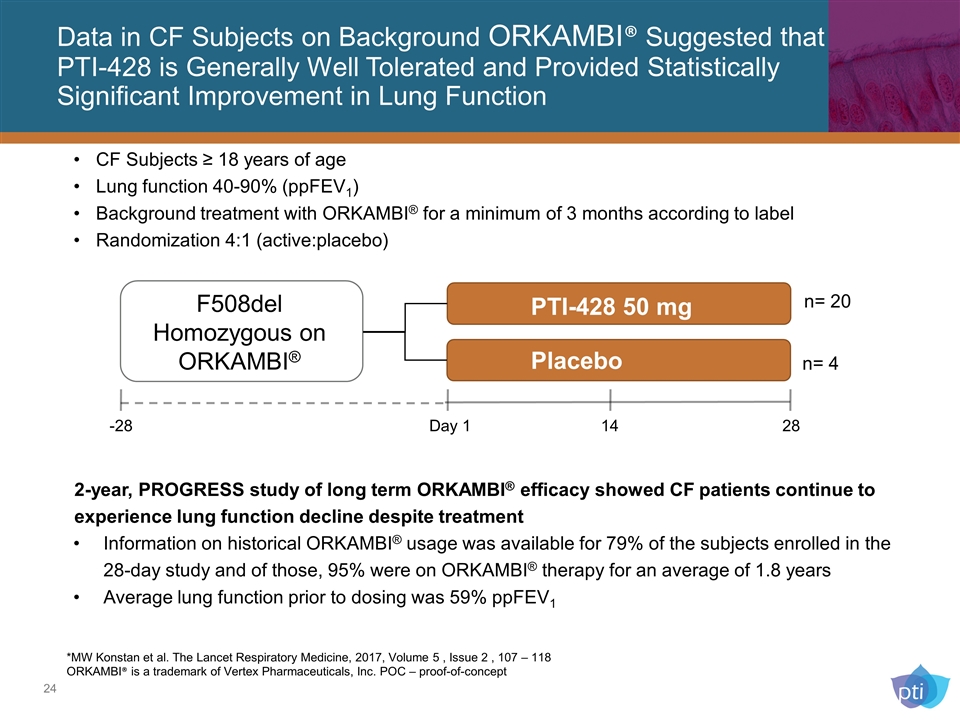
Data in CF Subjects on Background ORKAMBI® Suggested that PTI-428 is Generally Well Tolerated and Provided Statistically Significant Improvement in Lung Function *MW Konstan et al. The Lancet Respiratory Medicine, 2017, Volume 5 , Issue 2 , 107 – 118 ORKAMBI® is a trademark of Vertex Pharmaceuticals, Inc. POC – proof-of-concept 2-year, PROGRESS study of long term ORKAMBI® efficacy showed CF patients continue to experience lung function decline despite treatment Information on historical ORKAMBI® usage was available for 79% of the subjects enrolled in the 28-day study and of those, 95% were on ORKAMBI® therapy for an average of 1.8 years Average lung function prior to dosing was 59% ppFEV1 PTI-428 50 mg Placebo F508del Homozygous on ORKAMBI® -28 Day 1 14 28 n= 4 n= 20 CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Background treatment with ORKAMBI® for a minimum of 3 months according to label Randomization 4:1 (active:placebo)
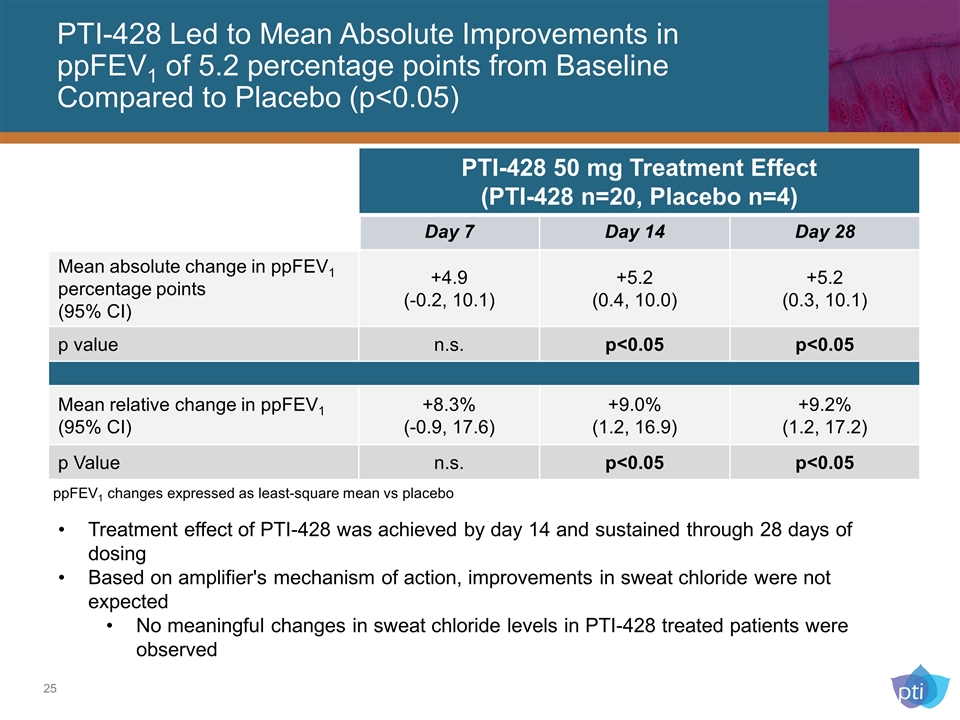
PTI-428 Led to Mean Absolute Improvements in ppFEV1 of 5.2 percentage points from Baseline Compared to Placebo (p<0.05) Treatment effect of PTI-428 was achieved by day 14 and sustained through 28 days of dosing Based on amplifier's mechanism of action, improvements in sweat chloride were not expected No meaningful changes in sweat chloride levels in PTI-428 treated patients were observed PTI-428 50 mg Treatment Effect (PTI-428 n=20, Placebo n=4) Day 7 Day 14 Day 28 Mean absolute change in ppFEV1 percentage points (95% CI) +4.9 (-0.2, 10.1) +5.2 (0.4, 10.0) +5.2 (0.3, 10.1) p value n.s. p<0.05 p<0.05 Mean relative change in ppFEV1 (95% CI) +8.3% (-0.9, 17.6) +9.0% (1.2, 16.9) +9.2% (1.2, 17.2) p Value n.s. p<0.05 p<0.05 ppFEV1 changes expressed as least-square mean vs placebo

PTI-428 Granted Breakthrough Designation from the FDA PTI-428 received Breakthrough Therapy Designation by FDA in March 2018 for treatment of CF patients homozygous for F508del on background ORKAMBI® therapy Grant was given based on clinical evidence of increased lung function during the 28-day, once-a-day treatment with PTI-428 in CF subjects on ORKAMBI® background The study provides first evidence that genotype agnostic CFTR amplifiers have the potential to provide additional benefit to CF patients on existing CFTR modulator therapy Since the inception of the program, less than half of the drugs submitted for Breakthrough Therapy under the FDA Safety and Innovation Act have been granted the designation. In the CFTR modulator category, BTD has been granted to KALYDECO®, ORKAMBI® and SYMDEKO®

Ongoing Study PTI-428 in CF Subjects on SYMDEKO® May Deliver Greater Benefit in FEV1 Than PTI-428+ ORKAMBI® PTI-428 dose 1 F508del Homozygous on SYMDEKO® -28 Day 1 14 28 PTI-428 dose 2 placebo Ongoing clinical study in CF subjects on background SYMDEKO® therapy with initial data expected in early 2019 Objective of the study is to support a development path towards NDA as add-on to marketed CFTR modulator therapies CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Background treatment with SYMDEKO® for a minimum of 1 months according to label Randomization 4:1 (active:placebo) Up to 40 CF subjects across all cohorts
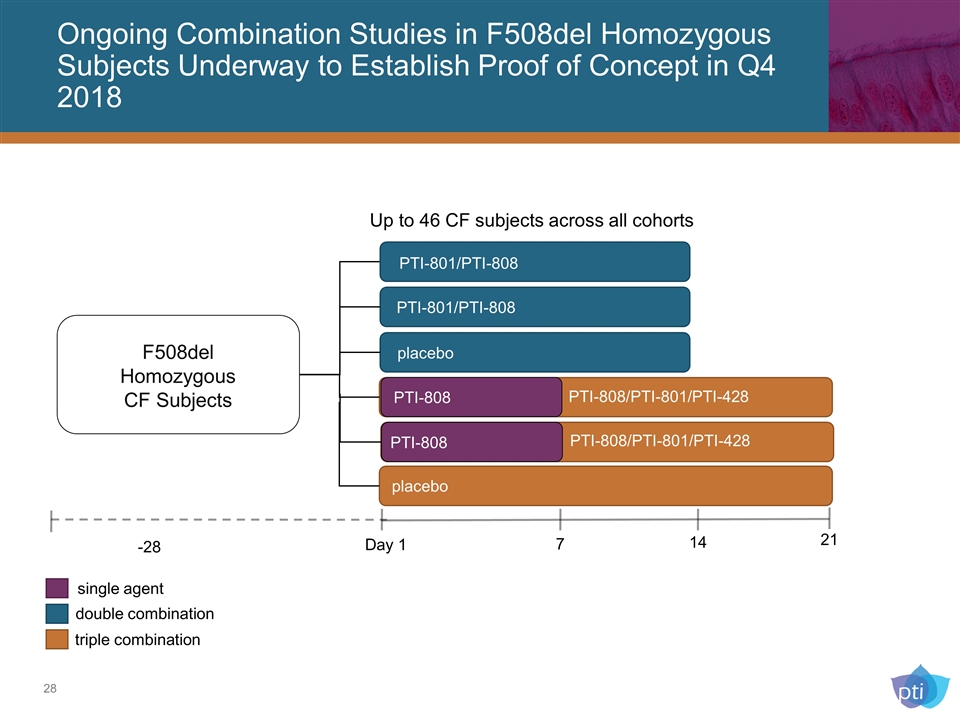
Ongoing Combination Studies in F508del Homozygous Subjects Underway to Establish Proof of Concept in Q4 2018 Placebo F508del Homozygous CF Subjects -28 Day 1 7 14 placebo PTI-801/PTI-808 PTI-801/PTI-808 PTI-808 PTI-808 placebo PTI-808/PTI-801/PTI-428 PTI-808/PTI-801/PTI-428 21 single agent double combination triple combination Up to 46 CF subjects across all cohorts

PTI-801/808/428 combination study protocol endorsed by patient advocacy groups in the US and EU PTI-801/808 combination study initiated in CF patients PTI-428 granted Breakthrough Therapy Designation by the FDA PTI-801 dosing in CF subjects on ORKAMBI® completed PTI-801/808/428 combination study initiated in CF subjects PTI-428 dosing in CF Subjects on SYMDEKO® initiated Year to Date and Near-term Clinical Milestones Year to Date Near-term Clinical Milestones PTI-801/808 preliminary data from combination study in CF subjects PTI-801/808/428 preliminary data from combination study in CF subjects PTI-428 preliminary data from study in CF Subjects on SYMDEKO® PTI-801 preliminary data from study in CF Subjects on SYMDEKO®

Thank You