
Proteostasis Therapeutics, Inc. (PTI) March 25, 2019 Exhibit 99.2

Safe Harbor and Disclaimer To the extent that statements in this presentation are not historical facts, they are forward-looking statements reflecting the current beliefs and expectations of management made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “aim,” “may,” “will,” “expect,” “anticipate,” “estimate,” “intend,” and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) are intended to identify forward-looking statements. Examples of forward-looking statements made in this presentation include, without limitation, statements regarding the potential of our proprietary combination therapies for the treatment of CF, the potential benefit to patients of our proprietary combination therapies, the expected timing of the initiation of, patient enrollment in, data from, and our completion of, our clinical studies and cohorts for PTI-428, PTI-801, PTI-808 and our combination therapy candidates, as well as cash guidance, and realization of the potential of the HIT-CF initiative. Forward-looking statements made in this presentation involve substantial risks and uncertainties that could cause actual results to differ materially from those expressed or implied by the forward-looking statements, and we therefore cannot assure you that our plans, intentions, expectations or strategies will be attained or achieved. Such risks and uncertainties include, without limitation, the possibility that final or future results from our drug candidate trials (including, without limitation, longer duration studies) do not achieve positive results or are materially and negatively different from or not indicative of the preliminary results reported in this presentation (noting that these results are on a small number of patients and small data set), uncertainties inherent in the execution and completion of clinical trials (including, without limitation, the possibility FDA requires us to run cohorts sequentially or conduct additional cohorts or pre-clinical or clinical studies), in the enrollment of CF patients in our clinical trials, in the timing of availability of trial data, in the results of the clinical trials, in possible adverse events from our trials, in the actions of regulatory agencies, in endorsement, if any, by CF patient advocacy groups, and those set forth in our Annual Report on Form 10-K for the year ended December 31, 2018, and our other SEC filings. We assume no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. This presentation also contains estimates and other statistical data made by independent parties and by us relating to, among other items, disease incidence, market size and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. Projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of risk and uncertainty. New risks emerge from time to time, and neither we nor any other person makes any representation as to the accuracy or completeness of such data or undertakes any obligation to update such date after the date of this presentation. By attending or receiving this presentation you acknowledge you are solely responsible for your own assessment of the market and our market position and that you will conduct your own analysis and are solely responsible for forming your own view of the potential future performance of our business. The trademarks included in this presentation are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of the Company or its securities.

Key Highlights as of March 25, 2019 High dose triplet shows mean absolute improvement in ppFEV1 of 8 percentage points at 14 days compared to placebo (p<0.05) in subjects with two F508del mutations (F/F) not predisposed to rapid pulmonary decline No plateau in ppFEV1 response observed at the end of dosing at day 14 with high dose triplet All combinations studied were well-tolerated, including in difficult to treat patients predisposed to rapid pulmonary decline Doublet and triplet combinations progressing to 28 day studies Phase 3 initiation remains on track for mid-2020

Potential Best-in-class doublet offers differentiated backbone to patients who cannot tolerate or do not have access to current options Global clinical development footprint established 500 total subjects dosed, 10 clinical studies, 7 countries, with 3 novel CFTR modulators since 2016 240 CF patients already dosed in studies with individual or proprietary combinations Completed 3 proof of concept studies with lumacaftor/ivacaftor and tezacaftor/ivacaftor as of Q1’19 Generated proof of concept data for proprietary doublet and triplet All combinations studied well-tolerated, including in difficult to treat patients ineligible for other CFTR modulator combinations studies Optimizing the path to success in pivotal studies targeted to begin in mid-2020 PTI Potential to Become a Second Entrant in CFTR Modulator Combination Market
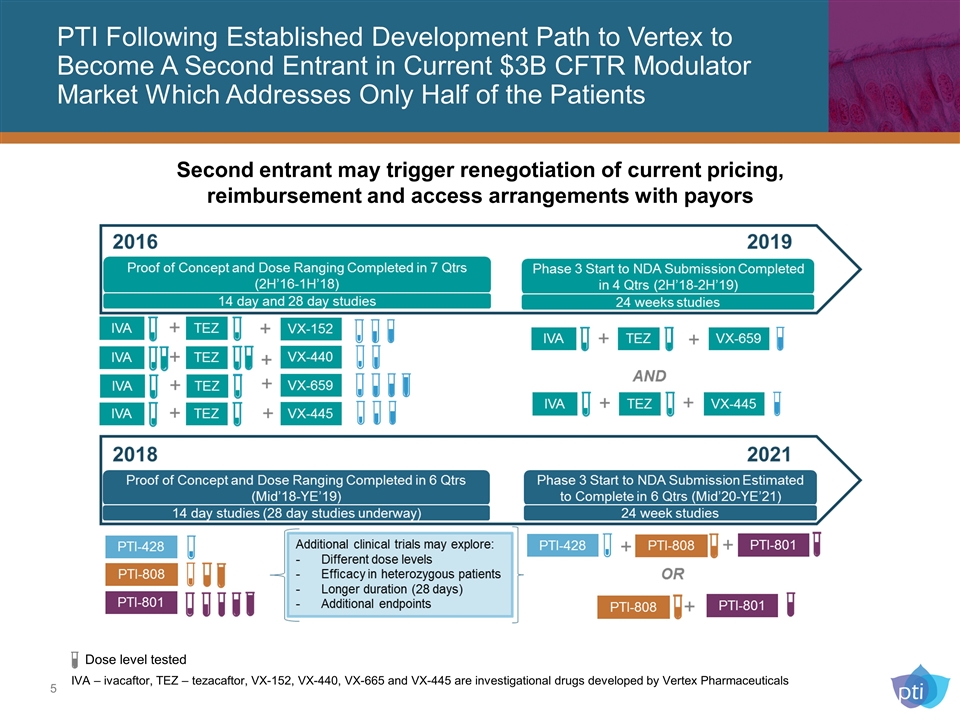
PTI Following Established Development Path to Vertex to Become A Second Entrant in Current $3B CFTR Modulator Market Which Addresses Only Half of the Patients IVA – ivacaftor, TEZ – tezacaftor, VX-152, VX-440, VX-665 and VX-445 are investigational drugs developed by Vertex Pharmaceuticals Dose level tested Second entrant may trigger renegotiation of current pricing, reimbursement and access arrangements with payors
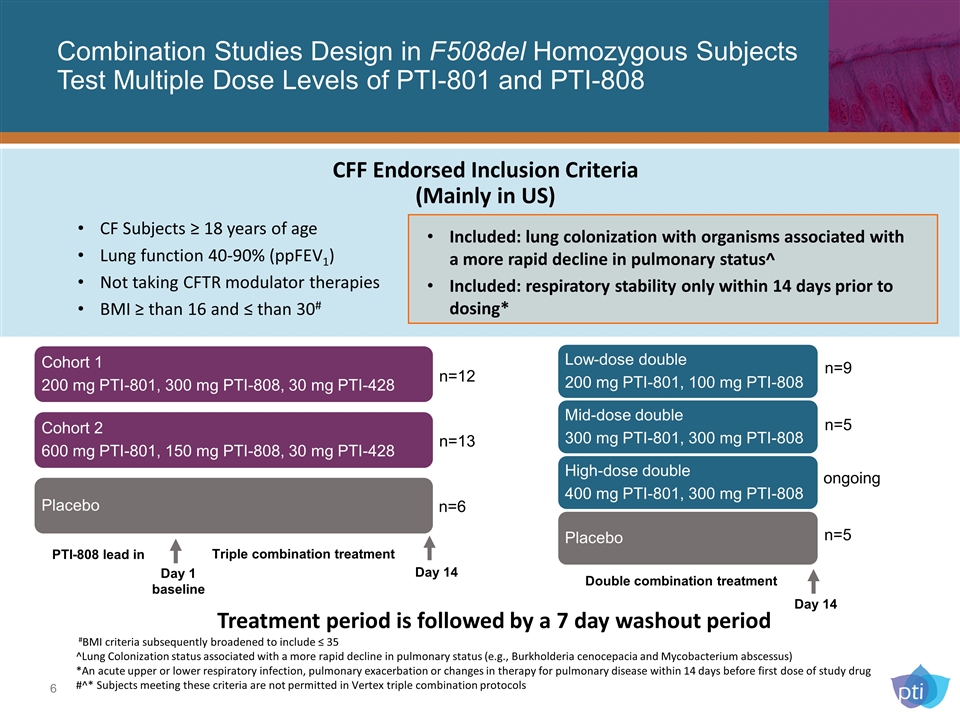
Combination Studies Design in F508del Homozygous Subjects Test Multiple Dose Levels of PTI-801 and PTI-808 n=9 n=5 ongoing n=5 n=12 n=13 n=6 #BMI criteria subsequently broadened to include ≤ 35 ^Lung Colonization status associated with a more rapid decline in pulmonary status (e.g., Burkholderia cenocepacia and Mycobacterium abscessus) *An acute upper or lower respiratory infection, pulmonary exacerbation or changes in therapy for pulmonary disease within 14 days before first dose of study drug #^* Subjects meeting these criteria are not permitted in Vertex triple combination protocols Day 1 baseline Day 14 Day 14 PTI-808 lead in Triple combination treatment Double combination treatment Treatment period is followed by a 7 day washout period CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Not taking CFTR modulator therapies BMI ≥ than 16 and ≤ than 30# Included: lung colonization with organisms associated with a more rapid decline in pulmonary status^ Included: respiratory stability only within 14 days prior to dosing* CFF Endorsed Inclusion Criteria (Mainly in US) Low-dose double 200 mg PTI-801, 100 mg PTI-808 Mid-dose double 300 mg PTI-801, 300 mg PTI-808 High-dose double 400 mg PTI-801, 300 mg PTI-808 Placebo Cohort 1 200 mg PTI-801, 300 mg PTI-808, 30 mg PTI-428 Cohort 2 600 mg PTI-801, 150 mg PTI-808, 30 mg PTI-428 Placebo
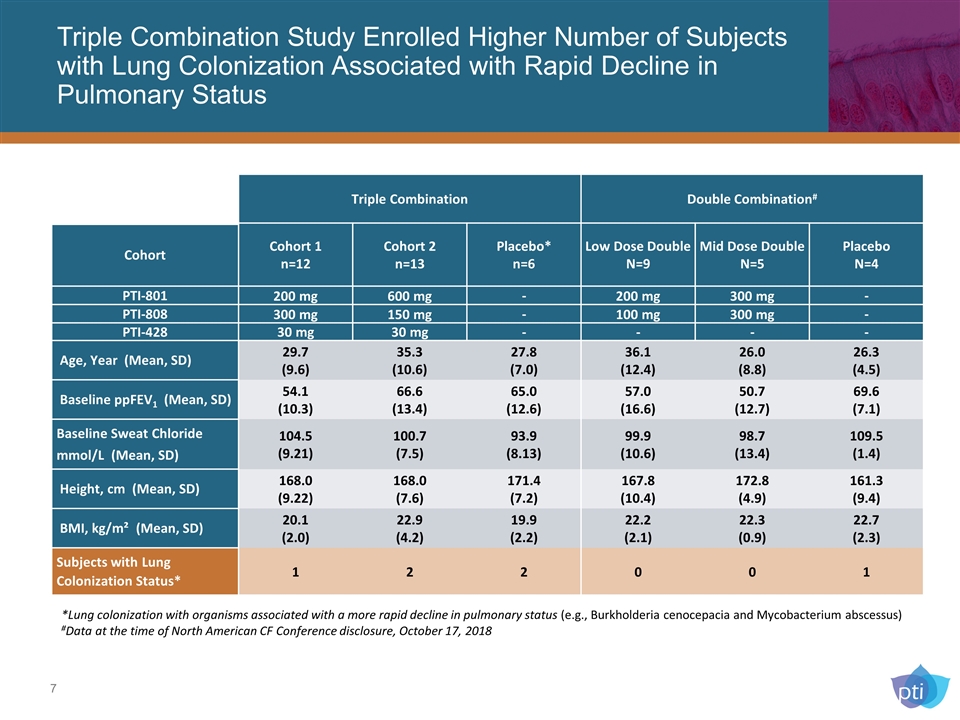
Triple Combination Study Enrolled Higher Number of Subjects with Lung Colonization Associated with Rapid Decline in Pulmonary Status Triple Combination Double Combination# Cohort Cohort 1 n=12 Cohort 2 n=13 Placebo* n=6 Low Dose Double N=9 Mid Dose Double N=5 Placebo N=4 PTI-801 200 mg 600 mg - 200 mg 300 mg - PTI-808 300 mg 150 mg - 100 mg 300 mg - PTI-428 30 mg 30 mg - - - - Age, Year (Mean, SD) 29.7 (9.6) 35.3 (10.6) 27.8 (7.0) 36.1 (12.4) 26.0 (8.8) 26.3 (4.5) Baseline ppFEV1 (Mean, SD) 54.1 (10.3) 66.6 (13.4) 65.0 (12.6) 57.0 (16.6) 50.7 (12.7) 69.6 (7.1) Baseline Sweat Chloride mmol/L (Mean, SD) 104.5 (9.21) 100.7 (7.5) 93.9 (8.13) 99.9 (10.6) 98.7 (13.4) 109.5 (1.4) Height, cm (Mean, SD) 168.0 (9.22) 168.0 (7.6) 171.4 (7.2) 167.8 (10.4) 172.8 (4.9) 161.3 (9.4) BMI, kg/m² (Mean, SD) 20.1 (2.0) 22.9 (4.2) 19.9 (2.2) 22.2 (2.1) 22.3 (0.9) 22.7 (2.3) Subjects with Lung Colonization Status* 1 2 2 0 0 1 *Lung colonization with organisms associated with a more rapid decline in pulmonary status (e.g., Burkholderia cenocepacia and Mycobacterium abscessus) #Data at the time of North American CF Conference disclosure, October 17, 2018
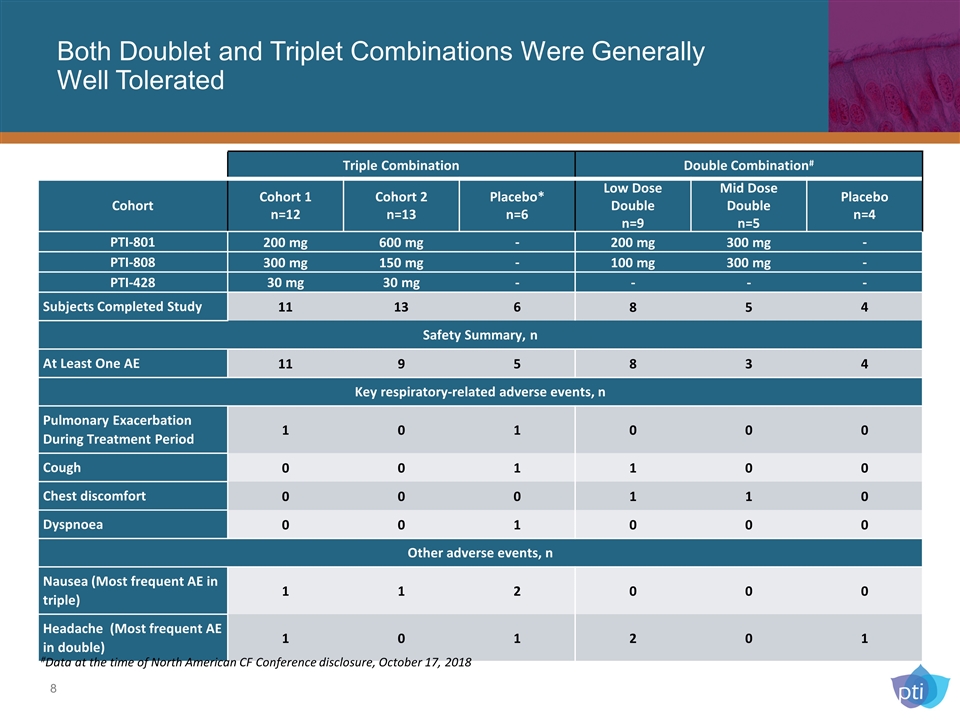
Both Doublet and Triplet Combinations Were Generally Well Tolerated Triple Combination Double Combination# Cohort Cohort 1 n=12 Cohort 2 n=13 Placebo* n=6 Low Dose Double n=9 Mid Dose Double n=5 Placebo n=4 PTI-801 200 mg 600 mg - 200 mg 300 mg - PTI-808 300 mg 150 mg - 100 mg 300 mg - PTI-428 30 mg 30 mg - - - - Subjects Completed Study 11 13 6 8 5 4 Safety Summary, n At Least One AE 11 9 5 8 3 4 Key respiratory-related adverse events, n Pulmonary Exacerbation During Treatment Period 1 0 1 0 0 0 Cough 0 0 1 1 0 0 Chest discomfort 0 0 0 1 1 0 Dyspnoea 0 0 1 0 0 0 Other adverse events, n Nausea (Most frequent AE in triple) 1 1 2 0 0 0 Headache (Most frequent AE in double) 1 0 1 2 0 1 #Data at the time of North American CF Conference disclosure, October 17, 2018
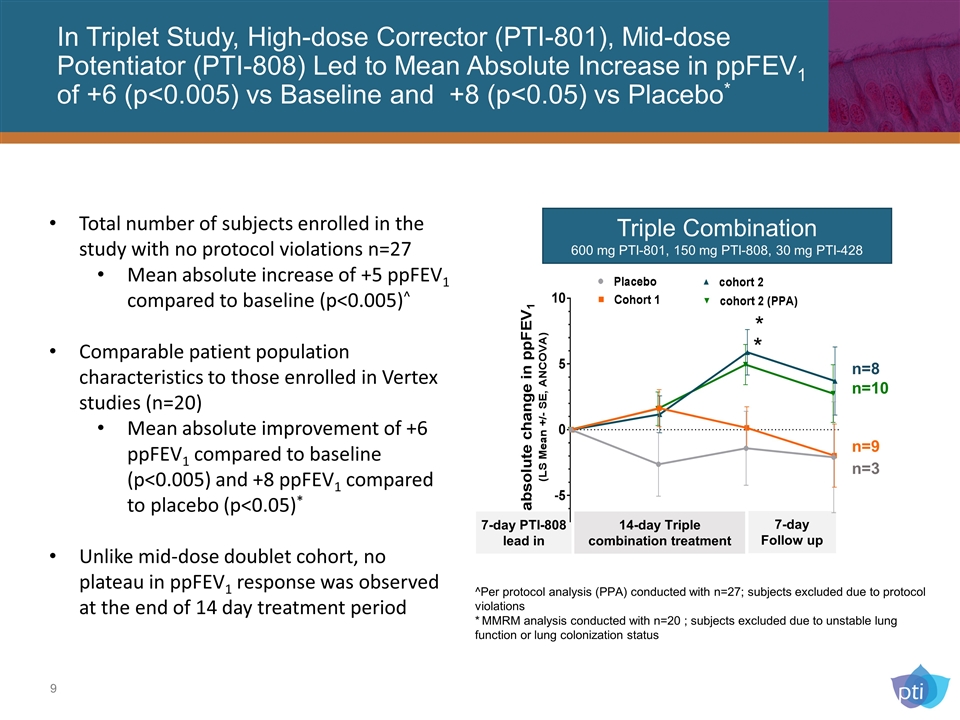
In Triplet Study, High-dose Corrector (PTI-801), Mid-dose Potentiator (PTI-808) Led to Mean Absolute Increase in ppFEV1 of +6 (p<0.005) vs Baseline and +8 (p<0.05) vs Placebo* Triple Combination 600 mg PTI-801, 150 mg PTI-808, 30 mg PTI-428 14-day Triple combination treatment 7-day Follow up n=10 n=3 7-day PTI-808 lead in n=9 n=8 * * Total number of subjects enrolled in the study with no protocol violations n=27 Mean absolute increase of +5 ppFEV1 compared to baseline (p<0.005)^ Comparable patient population characteristics to those enrolled in Vertex studies (n=20) Mean absolute improvement of +6 ppFEV1 compared to baseline (p<0.005) and +8 ppFEV1 compared to placebo (p<0.05)* Unlike mid-dose doublet cohort, no plateau in ppFEV1 response was observed at the end of 14 day treatment period ^Per protocol analysis (PPA) conducted with n=27; subjects excluded due to protocol violations * MMRM analysis conducted with n=20 ; subjects excluded due to unstable lung function or lung colonization status
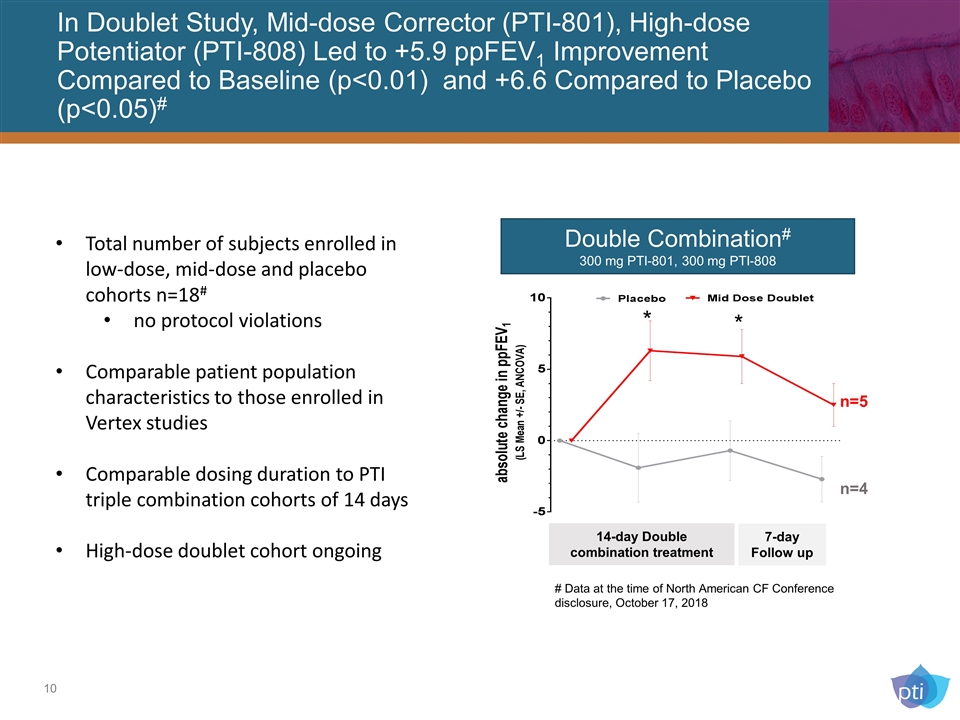
In Doublet Study, Mid-dose Corrector (PTI-801), High-dose Potentiator (PTI-808) Led to +5.9 ppFEV1 Improvement Compared to Baseline (p<0.01) and +6.6 Compared to Placebo (p<0.05)# Double Combination# 300 mg PTI-801, 300 mg PTI-808 14-day Double combination treatment 7-day Follow up n=5 n=4 * * Total number of subjects enrolled in low-dose, mid-dose and placebo cohorts n=18# no protocol violations Comparable patient population characteristics to those enrolled in Vertex studies Comparable dosing duration to PTI triple combination cohorts of 14 days High-dose doublet cohort ongoing # Data at the time of North American CF Conference disclosure, October 17, 2018
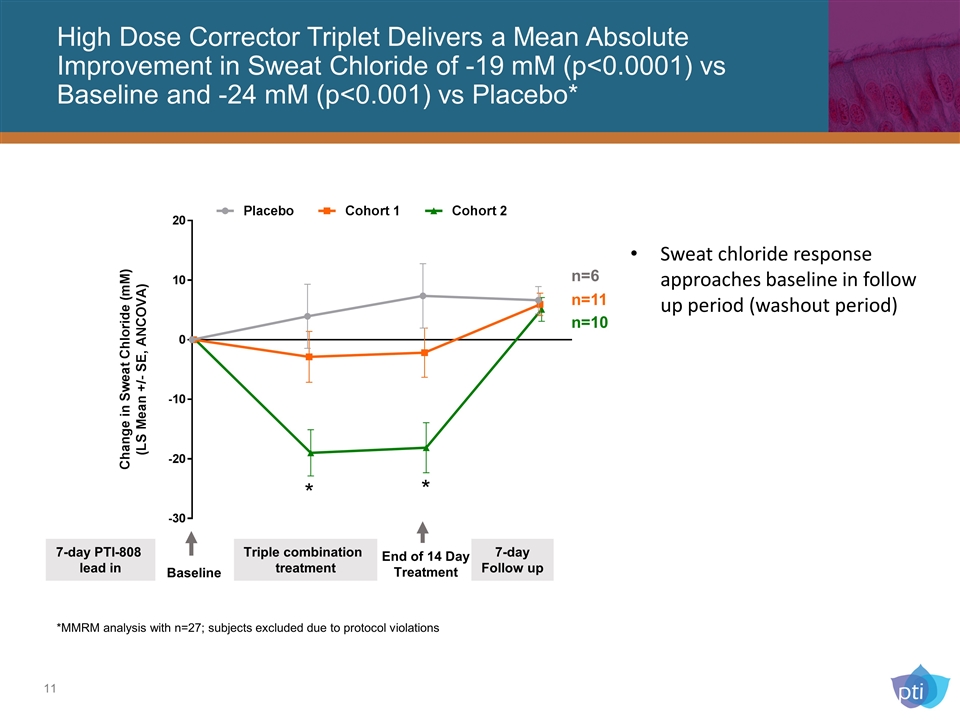
High Dose Corrector Triplet Delivers a Mean Absolute Improvement in Sweat Chloride of -19 mM (p<0.0001) vs Baseline and -24 mM (p<0.001) vs Placebo* Sweat chloride response approaches baseline in follow up period (washout period) *MMRM analysis with n=27; subjects excluded due to protocol violations End of 14 Day Treatment Triple combination treatment 7-day Follow up Baseline 7-day PTI-808 lead in * * n=10 n=11 n=6
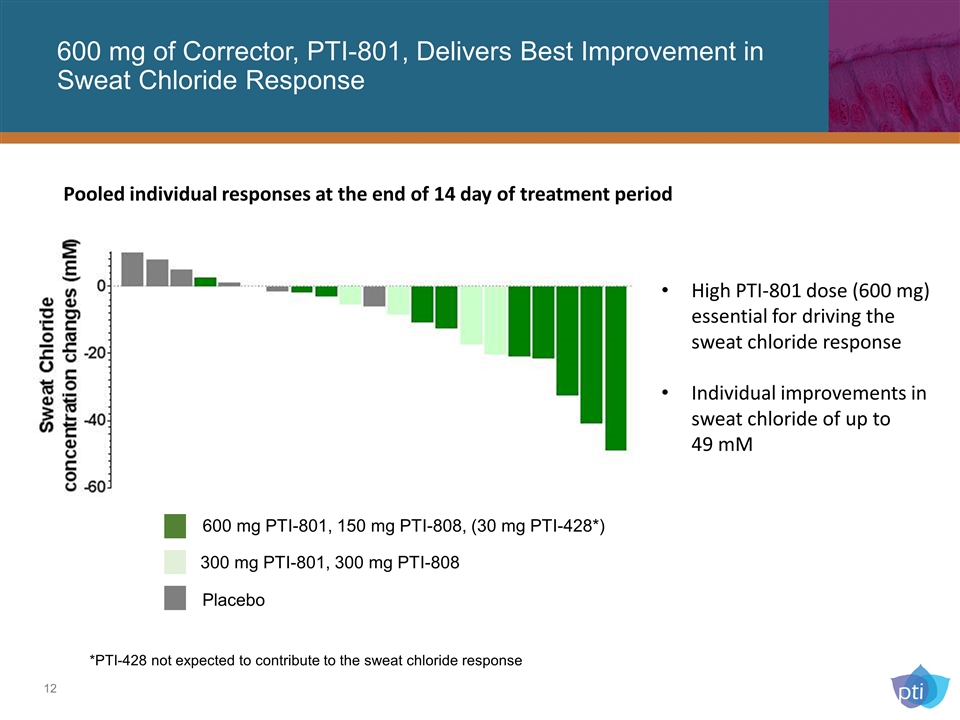
600 mg of Corrector, PTI-801, Delivers Best Improvement in Sweat Chloride Response 600 mg PTI-801, 150 mg PTI-808, (30 mg PTI-428*) 300 mg PTI-801, 300 mg PTI-808 *PTI-428 not expected to contribute to the sweat chloride response High PTI-801 dose (600 mg) essential for driving the sweat chloride response Individual improvements in sweat chloride of up to 49 mM Pooled individual responses at the end of 14 day of treatment period Placebo
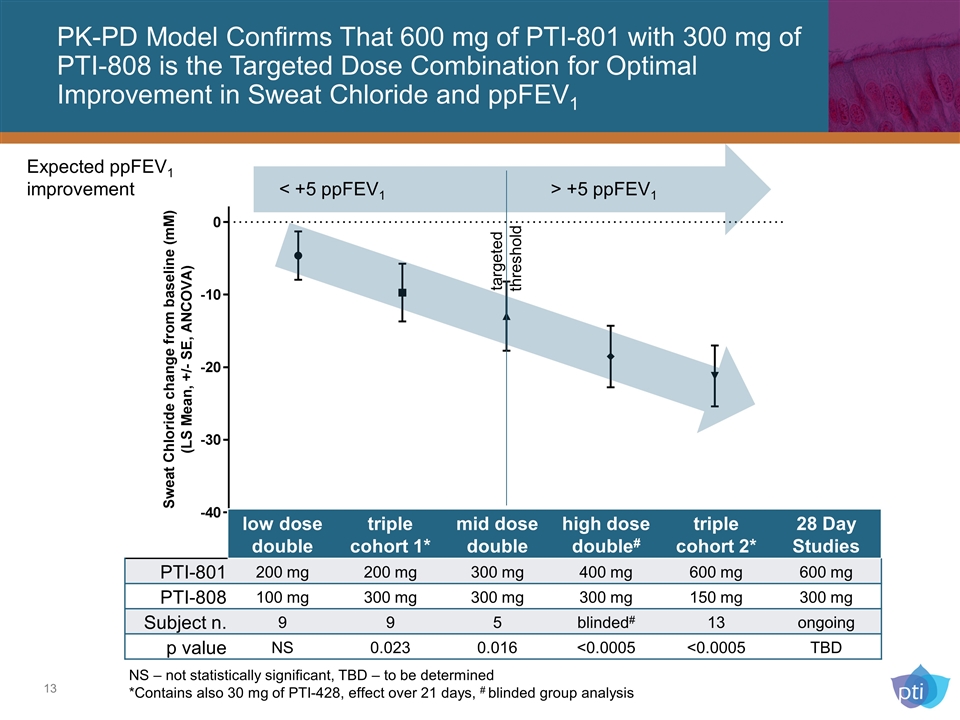
PK-PD Model Confirms That 600 mg of PTI-801 with 300 mg of PTI-808 is the Targeted Dose Combination for Optimal Improvement in Sweat Chloride and ppFEV1 NS – not statistically significant, TBD – to be determined *Contains also 30 mg of PTI-428, effect over 21 days, # blinded group analysis Expected ppFEV1 improvement < +5 ppFEV1 > +5 ppFEV1 low dose double triple cohort 1* mid dose double high dose double# triple cohort 2* 28 Day Studies PTI-801 200 mg 200 mg 300 mg 400 mg 600 mg 600 mg PTI-808 100 mg 300 mg 300 mg 300 mg 150 mg 300 mg Subject n. 9 9 5 blinded# 13 ongoing p value NS 0.023 0.016 <0.0005 <0.0005 TBD targeted threshold
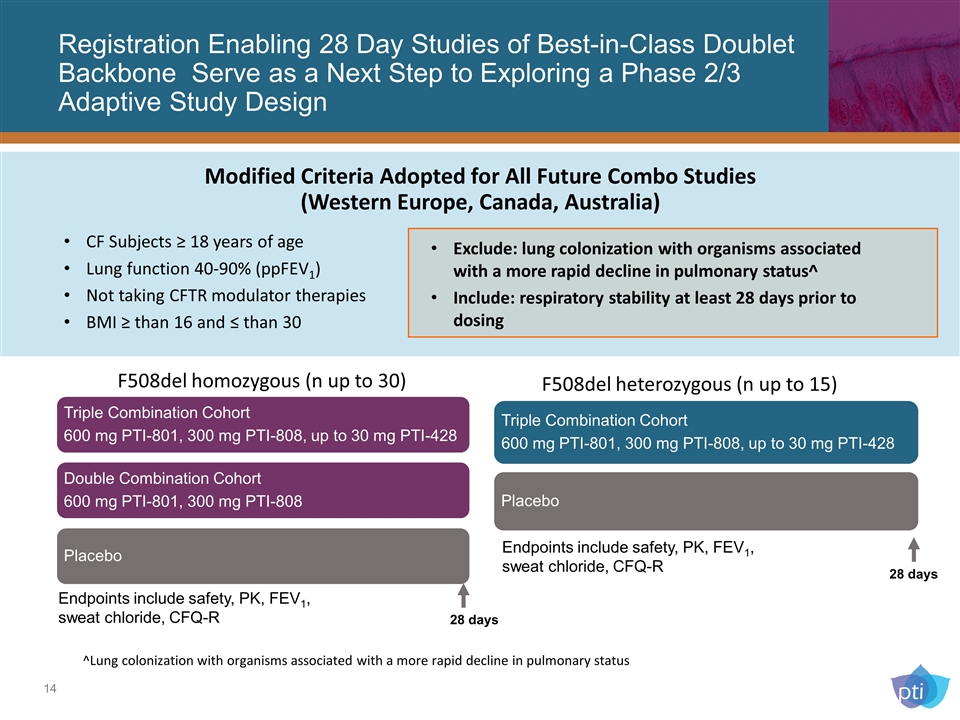
Registration Enabling 28 Day Studies of Best-in-Class Doublet Backbone Serve as a Next Step to Exploring a Phase 2/3 Adaptive Study Design ^Lung colonization with organisms associated with a more rapid decline in pulmonary status 28 days 28 days F508del homozygous (n up to 30) F508del heterozygous (n up to 15) Endpoints include safety, PK, FEV1, sweat chloride, CFQ-R Endpoints include safety, PK, FEV1, sweat chloride, CFQ-R CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Not taking CFTR modulator therapies BMI ≥ than 16 and ≤ than 30 Modified Criteria Adopted for All Future Combo Studies (Western Europe, Canada, Australia) Exclude: lung colonization with organisms associated with a more rapid decline in pulmonary status^ Include: respiratory stability at least 28 days prior to dosing Triple Combination Cohort 600 mg PTI-801, 300 mg PTI-808, up to 30 mg PTI-428 Placebo Triple Combination Cohort 600 mg PTI-801, 300 mg PTI-808, up to 30 mg PTI-428 Double Combination Cohort 600 mg PTI-801, 300 mg PTI-808 Placebo

Add-On Studies Results
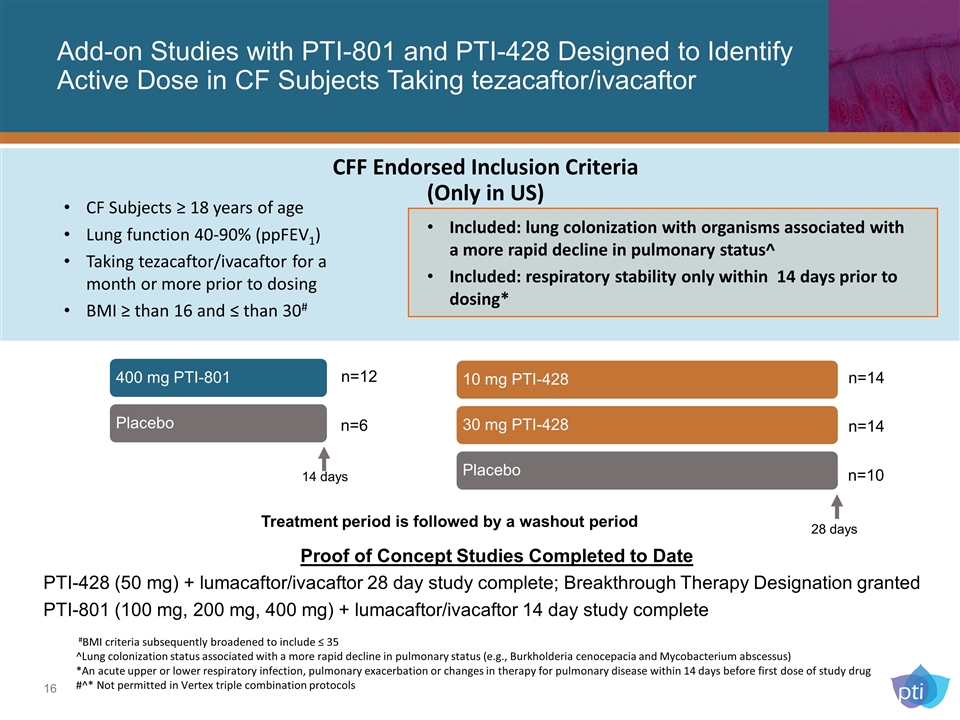
CFF Endorsed Inclusion Criteria (Only in US) Add-on Studies with PTI-801 and PTI-428 Designed to Identify Active Dose in CF Subjects Taking tezacaftor/ivacaftor n=14 n=14 n=10 n=12 n=6 Proof of Concept Studies Completed to Date PTI-428 (50 mg) + lumacaftor/ivacaftor 28 day study complete; Breakthrough Therapy Designation granted PTI-801 (100 mg, 200 mg, 400 mg) + lumacaftor/ivacaftor 14 day study complete 14 days 28 days CF Subjects ≥ 18 years of age Lung function 40-90% (ppFEV1) Taking tezacaftor/ivacaftor for a month or more prior to dosing BMI ≥ than 16 and ≤ than 30# #BMI criteria subsequently broadened to include ≤ 35 ^Lung colonization status associated with a more rapid decline in pulmonary status (e.g., Burkholderia cenocepacia and Mycobacterium abscessus) *An acute upper or lower respiratory infection, pulmonary exacerbation or changes in therapy for pulmonary disease within 14 days before first dose of study drug #^* Not permitted in Vertex triple combination protocols Treatment period is followed by a washout period Included: lung colonization with organisms associated with a more rapid decline in pulmonary status^ Included: respiratory stability only within 14 days prior to dosing* 400 mg PTI-801 Placebo 10 mg PTI-428 30 mg PTI-428 Placebo
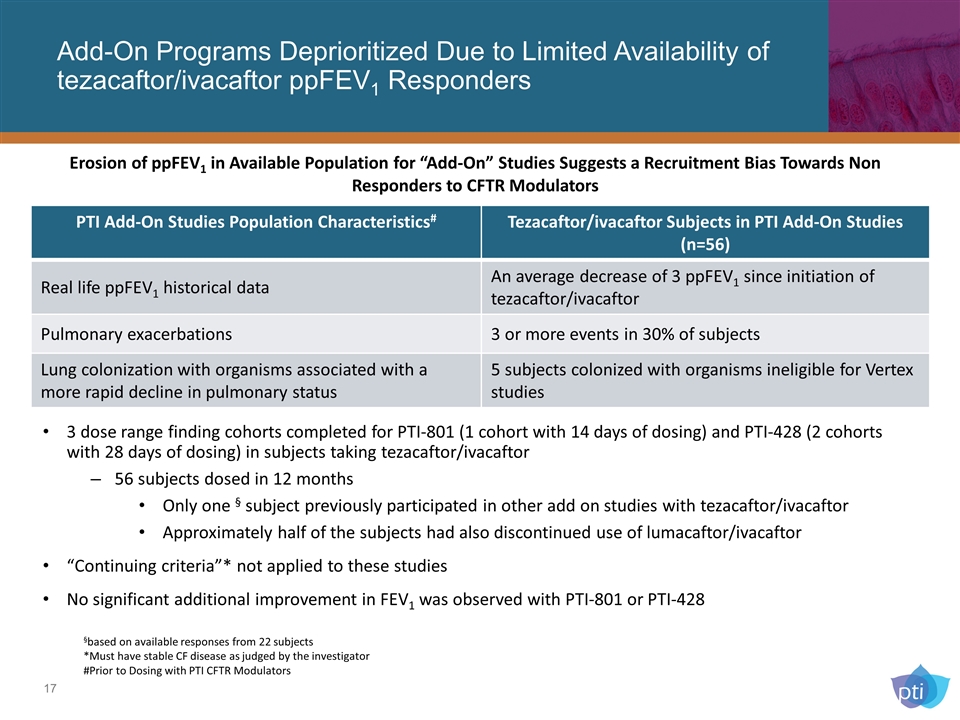
Add-On Programs Deprioritized Due to Limited Availability of tezacaftor/ivacaftor ppFEV1 Responders PTI Add-On Studies Population Characteristics# Tezacaftor/ivacaftor Subjects in PTI Add-On Studies (n=56) Real life ppFEV1 historical data An average decrease of 3 ppFEV1 since initiation of tezacaftor/ivacaftor Pulmonary exacerbations 3 or more events in 30% of subjects Lung colonization with organisms associated with a more rapid decline in pulmonary status 5 subjects colonized with organisms ineligible for Vertex studies §based on available responses from 22 subjects *Must have stable CF disease as judged by the investigator #Prior to Dosing with PTI CFTR Modulators 3 dose range finding cohorts completed for PTI-801 (1 cohort with 14 days of dosing) and PTI-428 (2 cohorts with 28 days of dosing) in subjects taking tezacaftor/ivacaftor 56 subjects dosed in 12 months Only one § subject previously participated in other add on studies with tezacaftor/ivacaftor Approximately half of the subjects had also discontinued use of lumacaftor/ivacaftor “Continuing criteria”* not applied to these studies No significant additional improvement in FEV1 was observed with PTI-801 or PTI-428 Erosion of ppFEV1 in Available Population for “Add-On” Studies Suggests a Recruitment Bias Towards Non Responders to CFTR Modulators
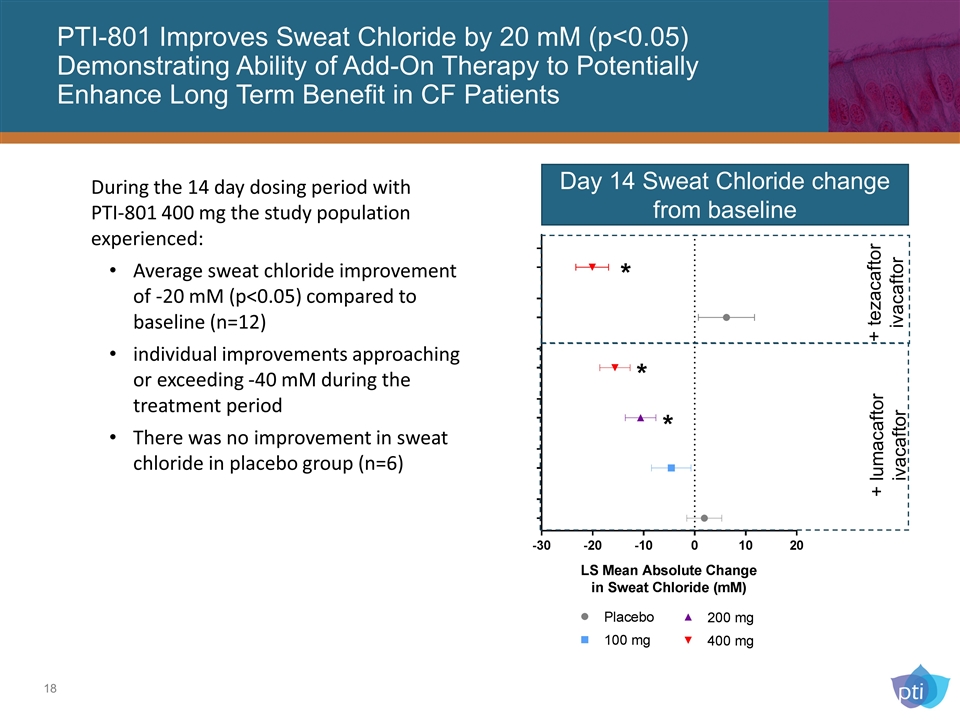
PTI-801 Improves Sweat Chloride by 20 mM (p<0.05) Demonstrating Ability of Add-On Therapy to Potentially Enhance Long Term Benefit in CF Patients Day 14 Sweat Chloride change from baseline * * * + lumacaftor ivacaftor + tezacaftor ivacaftor During the 14 day dosing period with PTI-801 400 mg the study population experienced: Average sweat chloride improvement of -20 mM (p<0.05) compared to baseline (n=12) individual improvements approaching or exceeding -40 mM during the treatment period There was no improvement in sweat chloride in placebo group (n=6)
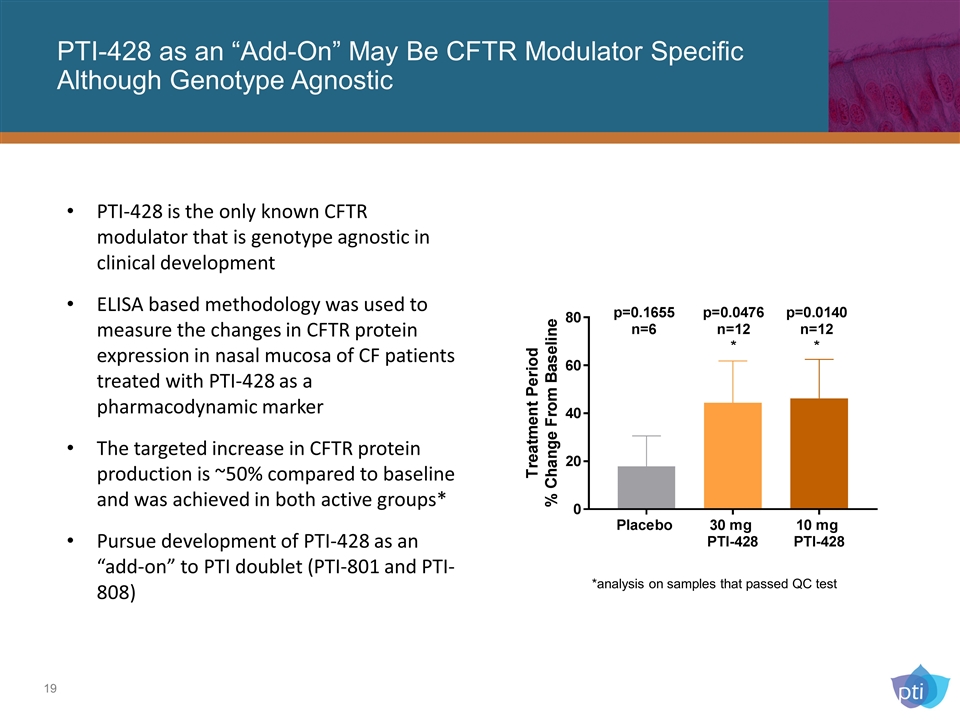
PTI-428 as an “Add-On” May Be CFTR Modulator Specific Although Genotype Agnostic PTI-428 is the only known CFTR modulator that is genotype agnostic in clinical development ELISA based methodology was used to measure the changes in CFTR protein expression in nasal mucosa of CF patients treated with PTI-428 as a pharmacodynamic marker The targeted increase in CFTR protein production is ~50% compared to baseline and was achieved in both active groups* Pursue development of PTI-428 as an “add-on” to PTI doublet (PTI-801 and PTI-808) *analysis on samples that passed QC test

Go Forward Strategy Driven by Proprietary Combination Study Results Built a robust PK-PD model to address combination rule consistent with established regulatory guidance in order to justify dose selection for subsequent studies including Phase 3 Established a dose response with PTI-801 and PTI-808 as seen by a sweat chloride and ppFEV1 improvement Establish an efficacy plateau currently not achieved by end of dosing at day 14 for the triple combination Proceed to 28 day studies to confirm or further strengthen efficacy signal In a competitive patient enrollment landscape, prioritize geographies with sufficient subjects that are comparable to those enrolled in recent phase 3 combination studies Examined overlap in subjects enrolled in PTI studies and non-PTI triple combination trials in order to better understand subject availability in US, western Europe, Canada and Australia Examined differences in inclusion and exclusion criteria across PTI and non-PTI protocols that could impact quality and pace of recruitment Predisposition to rapid decline in lung function based on bacterial colonization status and rate of pulmonary exacerbations, treatment compliance, broadening of inclusion/exclusion criteria

“Better than what we actually have” “Competitive” with the “best therapy profile” “Better than Symdeko in pulmonary function and CFTR activity” “Impressive FEV1” “Beneficial to have more than one option” and a “good alternative worth trying” “Easy formulation and dosing” “Good new option for CF patients” and an “option for non-responders” Physicians Remain Steadfast to Switch Between CFTR Combinations Based on 6 Month Data Not Short Term ppFEV1 Outcomes PTI conducted a blinded and independent primary market research survey of 82 pulmonologists in the US and EU to learn how physicians assess, rank, and prescribe CFTR modulators Physicians assess the impact of a modulator based on 6 month efficacy data in product label as well as the safety profile Prescribing decisions are not driven by 14 or 28 day ppFEV1 data Short term ppFEV1 data viewed merely as a “check and balance” to proceed to longer studies; greater priority assigned to sweat chloride Additional factors that influence prescribing behavior are clinical experience, patient preference, drug access, and extra-pulmonary considerations Physicians Support the Profile of a “Doublet-Like” Combination Product CF doctors

Summary and 2019 Milestones PK PD model enables effective dose selection PTI-801 and PTI-808 efficacious dose threshold established PTI-428 is a highly potent molecule, benefit to be determined +/- doublet No ppFEV1 reached for triplet, 28-day studies underway New data provides leverage for future studies Selection criteria consistent with current SOC Pulmonary stability central to interpreting results and capturing benefit Build Foundation for Planned Initiation of Phase 3 Studies in Mid-2020 YE 2019 PTI-801/808/428 28-day study in CF subjects (F508del homozygotes) PTI-801/808 28-day study in CF subjects (F508del homozygotes) PTI-801/808/428 28-day study in CF subjects (F508del heterozygotes) Cash runway into Q2 2021

Thank You