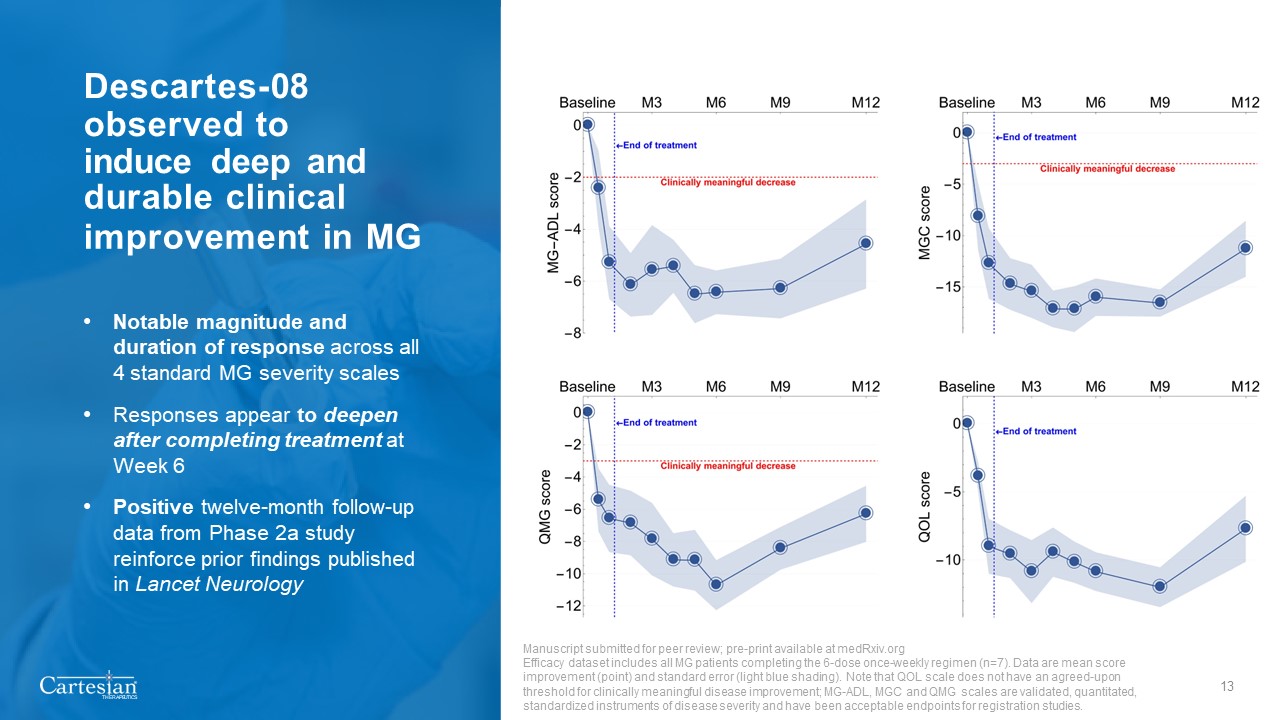
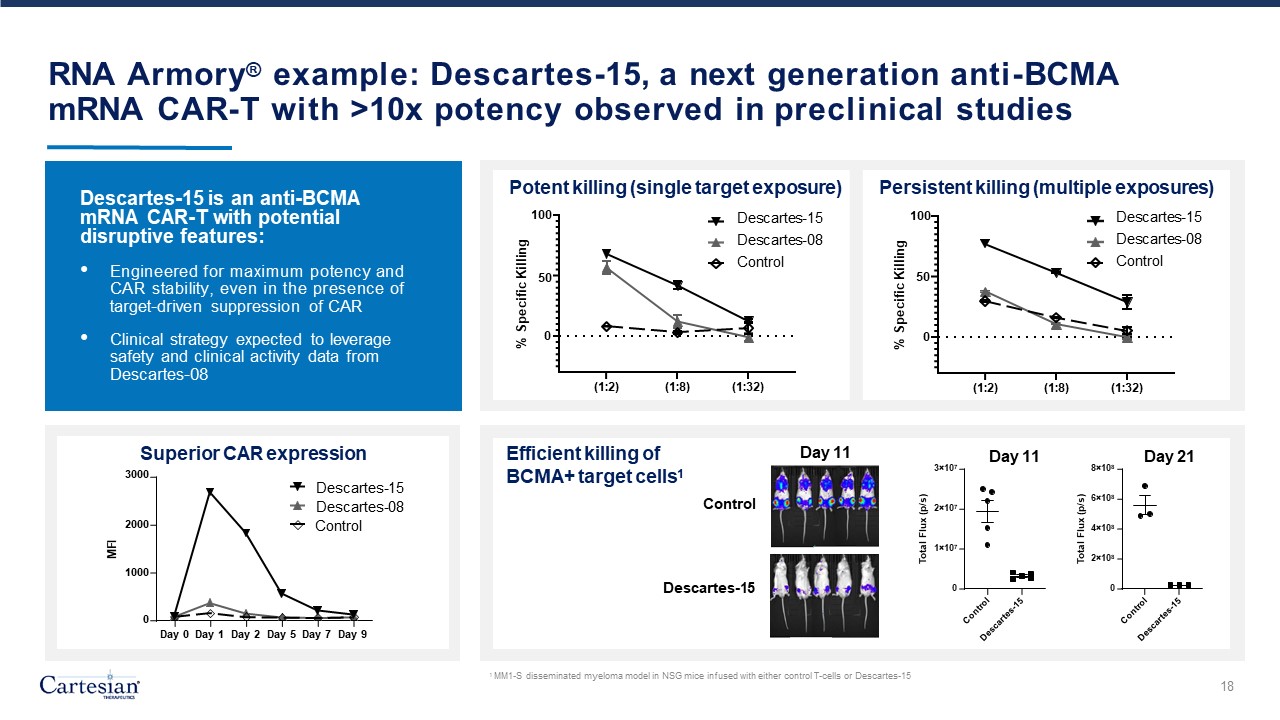
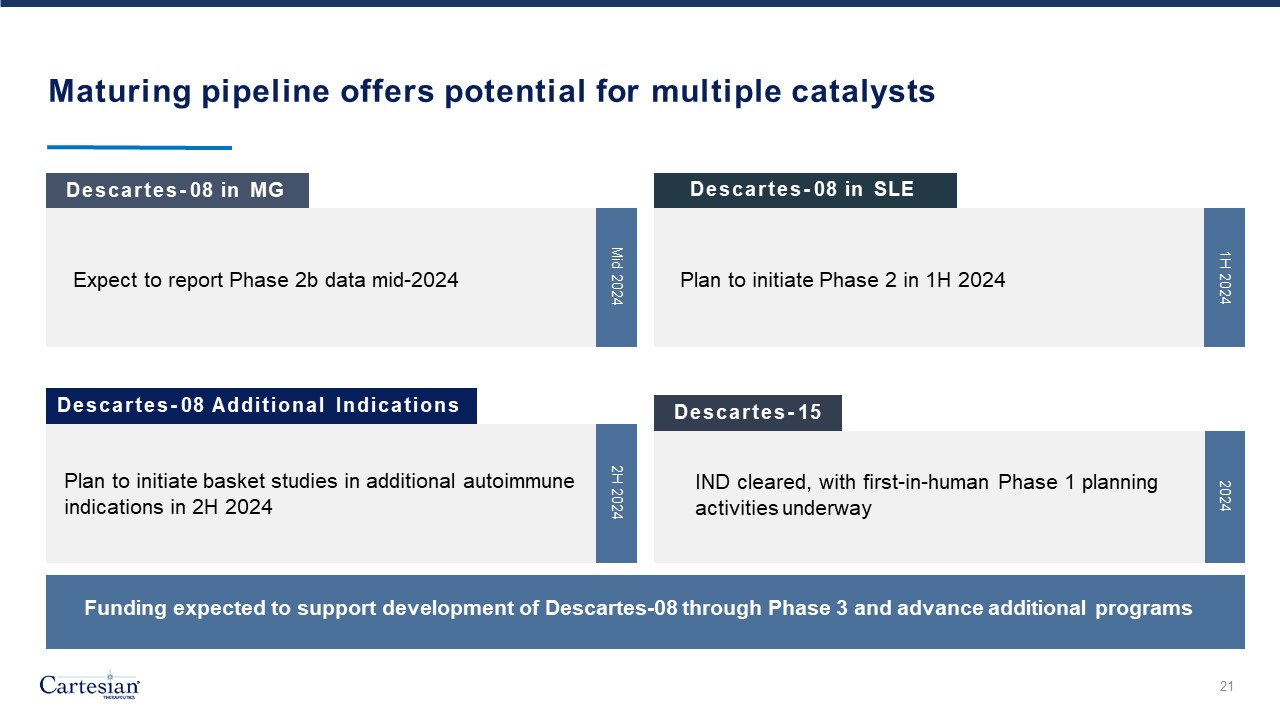
Forward-Looking Statements Disclosures For the purposes of this notice, the “presentation” that follows shall mean and include the slides that follow, the oral presentation of the slides by members of management of Cartesian Therapeutics, Inc. (the “Company”) or any person on their behalf, any question-and-answer session that follows such oral presentation, hard copies of this document and any materials distributed at, or in connection with, such oral presentation. Forward-looking Statements Any statements in this presentation about the future expectations, plans and prospects of the Company, including without limitation, statements regarding the Company’s expected cash resources and cash runway, the Company’s estimated cash on hand, the expected receipt of proceeds from the Company’s November 2023 private placement, conversion of the Company’s Series A Non-Voting Convertible Preferred Stock, the potential of RNA Armory® to enable precision control and optimization of engineered cells for diverse cell therapies leveraging multiple modalities, the potential of Descartes-08, Descartes-15, Descartes-33 and the Company’s other product candidates to treat myasthenia gravis, systemic lupus erythematosus, or any other disease, the anticipated timing or the outcome of ongoing and planned clinical trials, studies and data readouts, the anticipated timing or the outcome of the FDA’s review of the Company’s regulatory filings, the Company’s ability to conduct its clinical trials and preclinical studies, the timing or making of any regulatory filings, the anticipated timing or outcome of selection of developmental product candidates, the ability of the Company to consummate any expected agreements and licenses and to realize the anticipated benefits thereof, the novelty of treatment paradigms that the Company is able to develop, the potential of any therapies developed by the Company to fulfill unmet medical needs, the Company’s ability to enter into and maintain its strategic partnerships, and enrollment in the Company’s clinical trials and other statements containing the words “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “hypothesize,” “intend,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “target,” “would,” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including, but not limited to, the following: the uncertainties inherent in the initiation, completion and cost of clinical trials including proof of concept trials, including uncertain outcomes, the availability and timing of data from ongoing and future clinical trials and the results of such trials, whether preliminary results from a particular clinical trial will be predictive of the final results of that trial and whether results of early clinical trials will be indicative of the results of later clinical trials, the ability to predict results of studies performed on human beings based on results of studies performed on non-human subjects, the unproven approach of the Company’s RNA Armory® technology, potential delays in enrollment of patients, undesirable side effects of the Company’s product candidates, its reliance on third parties to conduct its clinical trials, the Company’s inability to maintain its existing or future collaborations, licenses or contractual relationships, its inability to protect its proprietary technology and intellectual property, potential delays in regulatory approvals, the availability of funding sufficient for its foreseeable and unforeseeable operating expenses and capital expenditure requirements, the Company’s recurring losses from operations and negative cash flows, substantial fluctuation in the price of the Company’s common stock, risks related to geopolitical conflicts and pandemics and other important factors discussed in the “Risk Factors” section of the Company’s most recent Annual Report on Form 10-K and Quarterly Reports on Form 10-Q, and in other filings that the Company makes with the Securities and Exchange Commission. In addition, any forward-looking statements included in this presentation represent the Company’s views only as of the date of its publication and should not be relied upon as representing its views as of any subsequent date. The Company specifically disclaims any intention to update any forward-looking statements included in this presentation, except as required by law. 2