 January 5, 2015 Solitaire-Oral Phase 3 Trial Design and Topline Data Developing Well-Differentiated Antibiotics Exhibit 99.2 |
 [ 2 ] [ 2 ] Forward Looking Statement This presentation contains forward-looking statements regarding future events. These statements are just predictions and are subject to risks and uncertainties that could cause the actual events or results to differ materially. These risks and uncertainties include, among others: risks related to the costs, sources of funding, timing, regulatory review and results of our studies and clinical trial and those of our strategic partners; our need to obtain additional funding and our ability to obtain future funding on acceptable terms; our anticipated capital expenditures and our estimates regarding our capital requirements; our and our strategic partners’ ability to obtain FDA and foreign regulatory approval of our product candidates; our dependence on the success of solithromycin and TAKSTA; the possible impairment of, or inability to obtain, intellectual property rights and the costs of obtaining such rights from third parties; the unpredictability of the size of the markets for, and market acceptance of, any of our products, including solithromycin and TAKSTA; our ability to produce and sell any approved products and the price we are able to realize for those products; our ability to retain and hire necessary employees and to staff our operations appropriately; our ability to compete in our industry; innovation by our competitors; and our ability to stay abreast of and comply with new or modified laws and regulations that currently apply or become applicable to our business. Please refer to the documents that we file from time to time with the Securities and Exchange Commission. |
 [ 3 ] [ 3 ] Two Differentiated Antibiotics in Late Stage Clinical Development LEAD INDICATION FORMULATIONS GEOGRAPHY Global Oral and IV Oral Suspension for Pediatrics Community-Acquired Bacterial Pneumonia (CABP) Refractory Bone and Joint Infections Oral US SOLITHROMYCIN Potent 4 th Generation Macrolide; 1 st Fluoroketolide TAKSTA TM Long History of Use in EU for Bone and Joint Infections and Skin Infections |
 [ 4 ] [ 4 ] Cephalosporin (e.g. Ceftriaxone) Macrolide (e.g. Azithromycin) Fluoroquinolone (e.g., Levofloxacin, Moxifloxacin) 2 1 Current CABP Therapies Have Use-Limiting Formulations and Safety Issues IDSA / ATS Recommends Broad Spectrum, Empiric Coverage 1 Freeman, MK. US Pharmacist. July 1, 2013 2 Magill, SS. And CDC and Emory Authors. NEJM 2014. 1198-1208, 2014 • Requires IV Ceftriaxone AND Hospitalization • No Oral Switch Therapy Replacement • Treatment Failures from Resistant Strain Selection • Kill Bowel Flora – Increased frequency of C. difficile Colitis • Adverse Tendonitis, Achilles Tendon Rupture, Hepatotoxicity and Peripheral Neuritis, Retinal Detachment • Not Approved for Use in Pediatrics • No Longer Used in Several Countries NO ORAL OPTION NO ORAL OPTION LACK OF SAFETY LACK OF SAFETY Issues Hospitalization Issues • 23% Hospitalized CABP Mortality Rate 1 • Hospital-Acquired Infections Costs and Hazards 2 2 Primary Options |
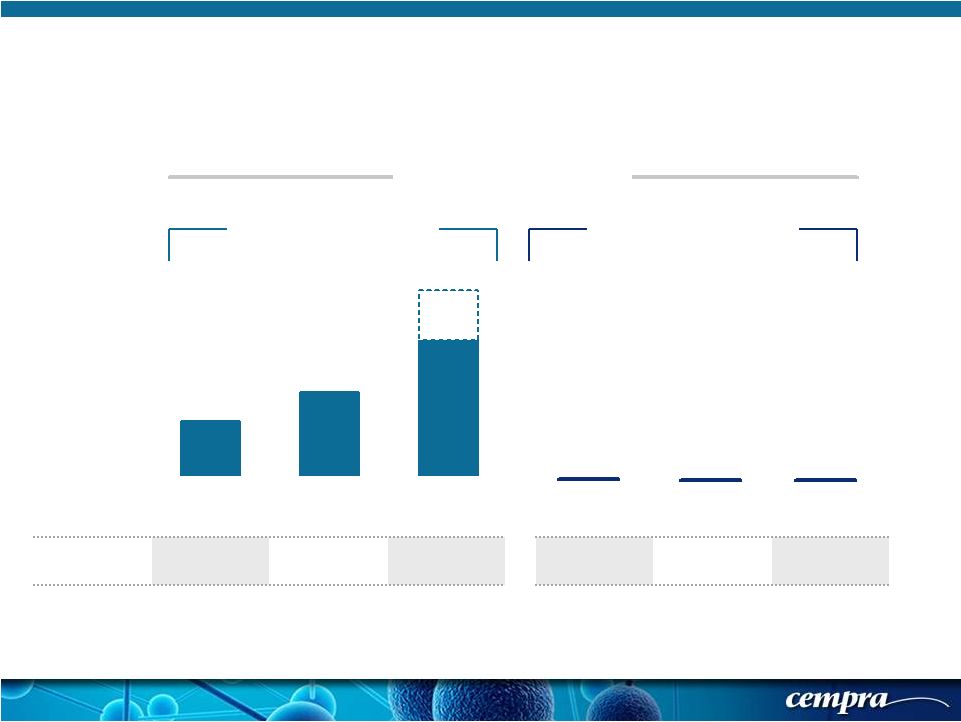 [ 5 ] [ 5 ] 28.0%* 44.0%*** 70.5%* Europe North America Asia 0% 0% 0% Europe North America Asia Resistance Drives Need for New Macrolide * Morrissey, I. ECCMID 2014. Abstr. P1584 ***Jones, RN.DMID 2013; 75:107-109. ** Kim,SH , AAC, 2012 , 56: 1418-1426 % Resistance AZITHROMYCIN SOLITHROMYCIN * MIC 90% (µg/mL) >1 >1 >1 0.06 0.25 0.5 China** 96.4% |
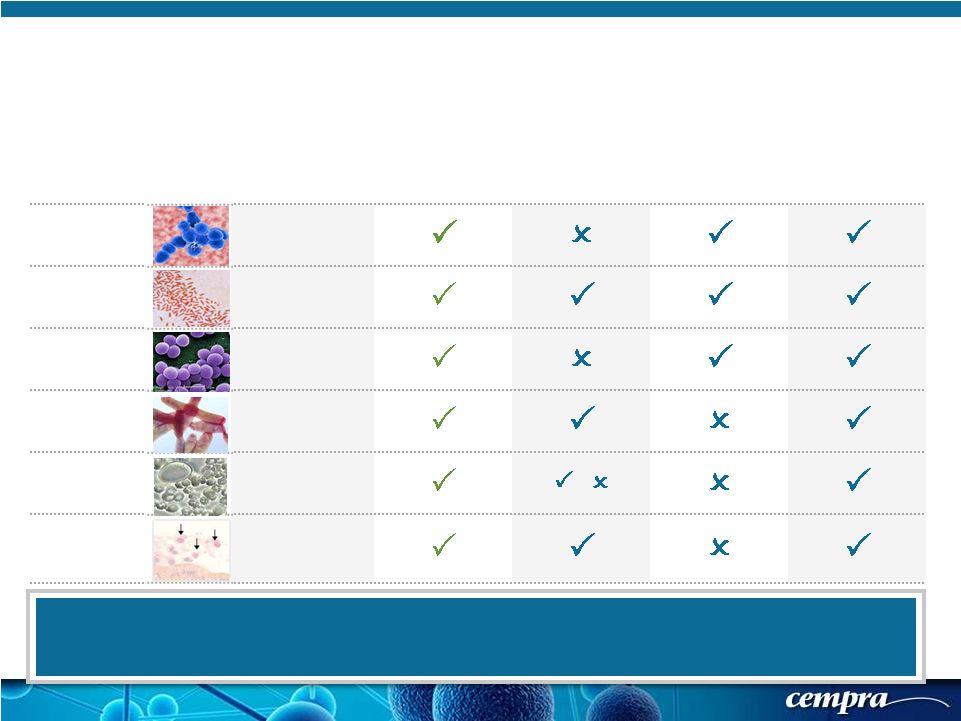 [ 6 ] [ 6 ] Solithromycin – Spectrum of Activity That Addresses CABP Pathogens GRAM ORGANISMS SOLITHROMYCIN AZITHROMYCIN CEFTRIAXONE LEVOFLOXACIN or MOXIFLOXACIN Positive Streptococcus pneumoniae Negative Haemophilus influenzae Positive Staphylococcus aureus Atypical Legionella pneumophila Atypical Mycoplasma pneumoniae / Atypical Chlamydophila pneumoniae Solithromycin Has Class-Leading Potency and Spectrum In Vitro Against CABP Pathogens Azithromycin is Not Used in Monotherapy to Treat Moderate to Severe Pneumonia – Potency, Spectrum and Resistance Allow Use Only in Simpler Infections or Add-On To Ceftriaxone |
 [ 7 ] [ 7 ] • Blinded, Randomized 1:1, Global • Soli = 5 days; Avelox = 7 days TRIAL DESIGN • 2x Sites + More Countries than Oral • 7 Day IV: MD Determines Oral Switch Avelox (moxifloxacin) COMPARATOR Avelox (moxifloxacin) 860 PATIENTS (n) 800 Early Response ITT (Non-Inferiority) PRIMARY ENDPOINT Early Response ITT (Non-Inferiority) Microbial ITT; Safety; SFU SECONDARY ENDPOINT Microbial ITT; Safety; SFU Completed Enrollment Q3 2014 Topline Data Announced on Jan 4, 2015 STATUS Update on Enrollment in April 2015 Solithromycin - Solitaire Phase 3 CABP Trials SOLITAIRE FORMULATION PRECLINICAL PHASE 1 PHASE 2 PHASE 3 Oral IV-to-Oral SOLITAIRE IV-to-ORAL 1 1 Combined Data For NDA 2 2 SOLITAIRE-ORAL |
 [ 8 ] [ 8 ] Randomized 860 Patients Received Either Solithromycin or Moxifloxacin Solitaire-Oral Phase 3 CABP Study Outline Randomization stratified by geographic region, history of asthma and/or COPD, and by pneumonia severity index (PORT) risk class PORT II severity pneumonia capped at 50% Enrollment of patients <65 years of age limited to 80% Enrollment of patients outside North America limited to 75% Treatment and Follow-up The duration of study drug administration - 5 days for solithromycin and 7 days for moxifloxacin Assessment of early clinical response (ECR) - 72 hours (-12/+36 hours) after the first dose of study drug Clinical response evaluated at the End of Treatment (EOT) Visit (last dose of study drug) and the SFU Visit (5 to 10 days after the EOT) - Short Term Follow up (SFU) is EMA’s primary end point Long Term Follow Up (LFU) visit for all-cause mortality at 28-35 days after the first dose of study drug |
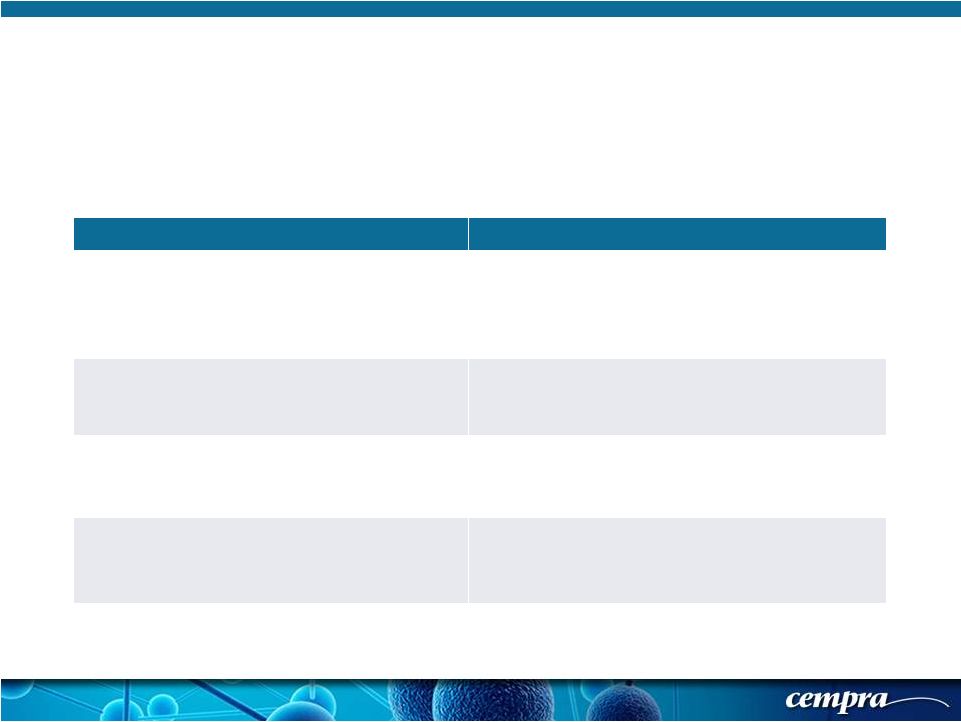 [ 9 ] [ 9 ] Randomized (1:1) 860 Patients Received Either Solithromycin or Moxifloxacin: Solitaire-Oral Phase 3 CABP: Inclusion/Exclusion Criteria Key Inclusion Criteria Key Exclusion Criteria Clinical - At least 3 of: cough, dyspnea, purulent sputum, and chest pain AND At least 1 of: fever, hypothermia, rales/ evidence of consolidation Hospital- or ventilator-acquired pneumonia Known viral, fungal pneumonia, CF, TB, Pneumocystis pneumonia, aspiration pneumonia No prior antibiotic treatment (past 7 days) (1 dose of short acting allowed in 25% of the population) HIV, current treatment for HCV, myasthenia gravis, chemotherapy PORT Class II-IV (score of 51 to 105) QTcF > 450 msec CrCl < 30 mL/min; AST/ALT/T bili > 2xULN ANC < 500; Platelets < 50,000 Chest X-Ray: Lobar/multilobar infiltrates, or patchy parenchymal QTc greater than 450 msec or use of class Ia or III anti- arrhythmics |
 [ 10 ] [ 10 ] PORT* Score Determination – Measure of Pneumonia Severity PORT Score Determination Patient Characteristic Point Assignment Age One point for each year of age Female? -10 if yes Neoplastic disease history? +30, if yes Liver disease? +20, if yes Congestive heart disease? +10, if yes Cerebrovascular disease? +10, if yes Renal disease? +10, if yes Altered mental status? +20, if yes Respiratory rate 30/minute? +20, if yes Systolic blood pressure <90 mm Hg? +20, if yes Temperature <35 º C (95ºF) or 40ºC (104ºF) +15, if yes Pulse 125 +10, if yes pH <7.35 (from ABG 2 )? +30, if yes (+0 if ABG not obtained) Blood urea nitrogen >30 mg/dL (Urea >11 mmol/L)? +20, if yes Sodium <130 mmol/L? +20, if yes Glucose 250 (14 mmol/L)? +10, if yes Hematocrit <30%? +10, if yes Partial pressure of arterial oxygen <60 mmHg (from ABG if medically indicated) or oxygen saturation <90% (by pulse oximetry)? +10, if yes Pleural effusion on radiograph? +10, if yes PORT Score Sum of Applicable Numbers Above PORT Risk Class Determination PORT Risk Class PORT Score I (Ineligible for Study) 0-50 II 51-70 III 71-90 IVa 91-105 IVb - V (Ineligible for Study) >105 to 131 *PORT : Patient Outcome Research Team |
 [ 11 ] [ 11 ] Solitaire-Oral Phase 3 Trial: Analysis Populations Intent-to-treat (ITT) population All patients who were randomized Safety population All patients who received at least one capsule of study drug Microbial-ITT population (mITT) All randomized patients who have a baseline bacterial pathogen known to cause CABP Clinically evaluable (CE) or per-protocol populations Patients who meet the definition for the ITT population and who follow key protocol procedures Microbiologically evaluable populations (ME) Micro-ITT population who follow protocol (intersection of CE and mITT) |
 [ 12 ] [ 12 ] Solitaire-Oral Phase 3 Trial: Primary Objectives and Endpoints Primary objective and endpoint (for EMA) Primary objective and endpoint (for FDA) NI in SFU success rate in the ITT-SFU and CE-SFU populations. Clinical Success at the SFU visit (Day 5-7 after EOT visit) in the ITT and CE-SFU populations. Clinical success was as defined by the investigator. Noninferiority (NI) in Early Clinical Response (ECR) rate in the ITT population. Improvement at day 3 to day 5 in at least two of the following symptoms: chest pain, cough, difficulty with sputum production, and dyspnea, without worsening in any symptom. Symptoms evaluated on a four-point scale—absent, mild, moderate, severe as experienced by the patient— from baseline to day 3 to day 5 (e.g., from severe to moderate, from moderate to mild, or from mild to absent). |
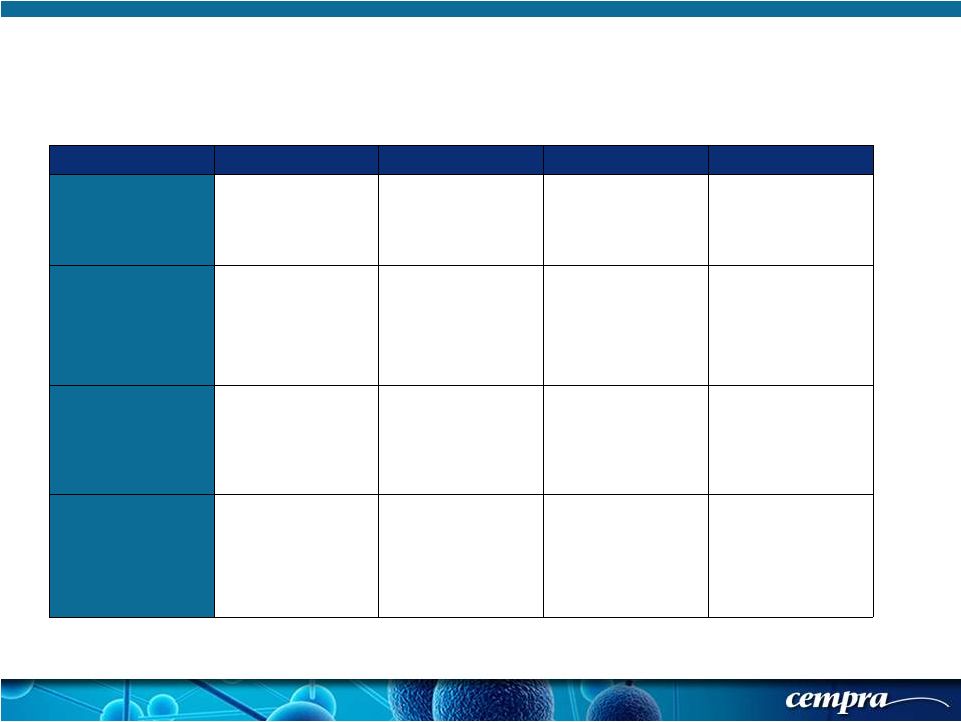 [ 13 ] [ 13 ] Solitaire-Oral Phase 3: Symptom Scores for ECR Assessment Symptom Absent (0) Mild (1) Moderate (2) Severe (3) Cough Resolution (to pre- CABP baseline) or absence of cough. Frequent, interferes with normal activity or sleep Constant, interferes with most or all activities or sleep Dyspnea/Shortness of Breath Resolution (to pre- CABP baseline) or absence of dyspnea Dyspnea on exertion (e.g. climbing stairs) Dyspnea with normal/routine activities (e.g. walking) Dyspnea at rest or requiring oxygen therapy Chest Pain due to Pneumonia Resolution or absence of chest pain related to CABP Transient, does not interfere with normal activity Frequent, interferes with normal activity or sleep Constant, interferes with most or all activities or sleep Difficulty with Sputum Production Resolution (to pre- CABP baseline) or absence of sputum production Sputum production rarely causes difficulty or distress Sputum production often causes difficulty or distress Constant difficulty with sputum production Transient, does not interfere with normal activity |
 [ 14 ] [ 14 ] Solitaire-Oral Phase 3 Trial: Secondary Endpoints Secondary Endpoints: NI in early clinical response rate at 72 (-12/+36) hours in the pooled mITT population from the two Phase 3 trials. NI in early clinical response rate at 72 (-12/+36) hours in the individual study mITT population. Safety and tolerability of oral solithromycin in comparison with oral moxifloxacin. |
 [ 15 ] [ 15 ] Solitaire-Oral Phase 3 Trial: Statistical Considerations Sample size based on response rate in Phase 2 trial in the ITT analysis population – 90% power NI margin of 10.0 % at the ECR endpoint at day 3 to day 5 for the ITT population NI margin for mITT analyses is 12.5% in the pooled population of the two Phase 3 trials – However, we have analyzed mITT in the single study as a guide The NI hypothesis test is a 1-sided hypothesis test performed at the 2.5% level of significance, based on the lower limit of the 2-sided 95% confidence interval (CI) for the observed difference in the early clinical response rate (solithromycin group minus the moxifloxacin group). The CI is calculated using an unadjusted continuity corrected Z-test - If the lower limit of the 95% CI for the difference in response rates in the ITT population is greater than -10%, then solithromycin is NI to moxifloxacin |
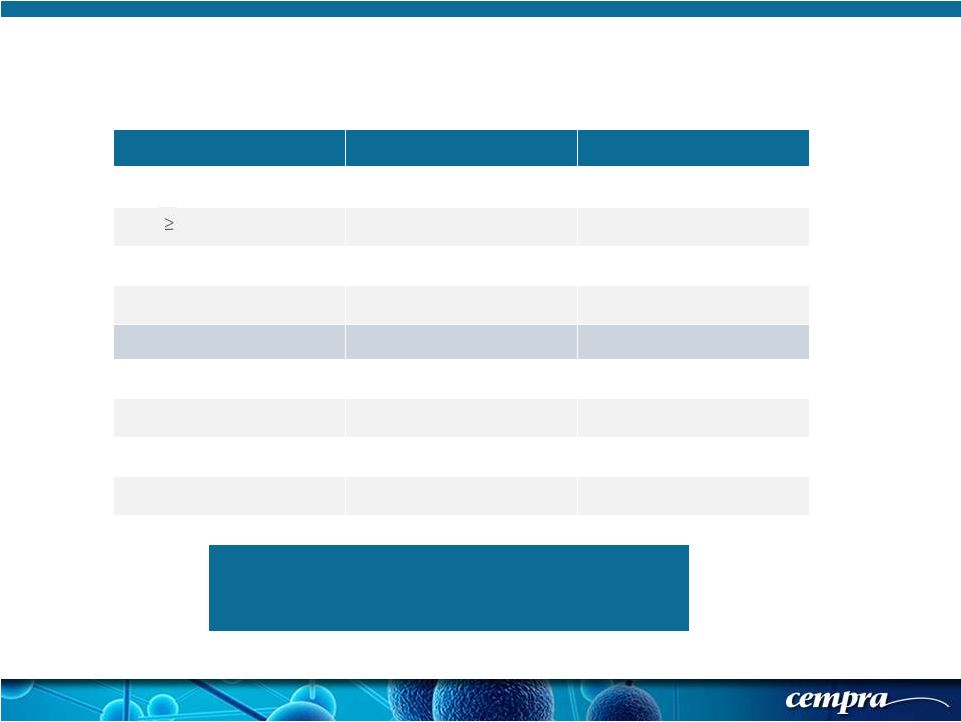 [ 16 ] [ 16 ] Solithromycin Moxifloxacin 58.5 56.7 36.4% 31.6% 53.3% 52.8% 14.6% 14.7% PORT I* 1* (0.2%) 0 PORT II 209 (49.1%) 223 (51.4%) PORT III 168 (39.4%) 173 (39.9%) PORT IV 48 (11.3%) 38 (8.8%) Solitaire-Oral Phase 3 Trial: Baseline Characteristics and Enrolling Geographical Regions/Countries Enrollment by Regions/ Countries North America 23.7%, Europe 52.1%, Latin America 12.3%, South Africa 11.9% Male gender (%) Mean Age (years) * PORT score incorrectly calculated by investigator * PORT score incorrectly calculated by investigator h/o asthma or COPD % 65 years of age |
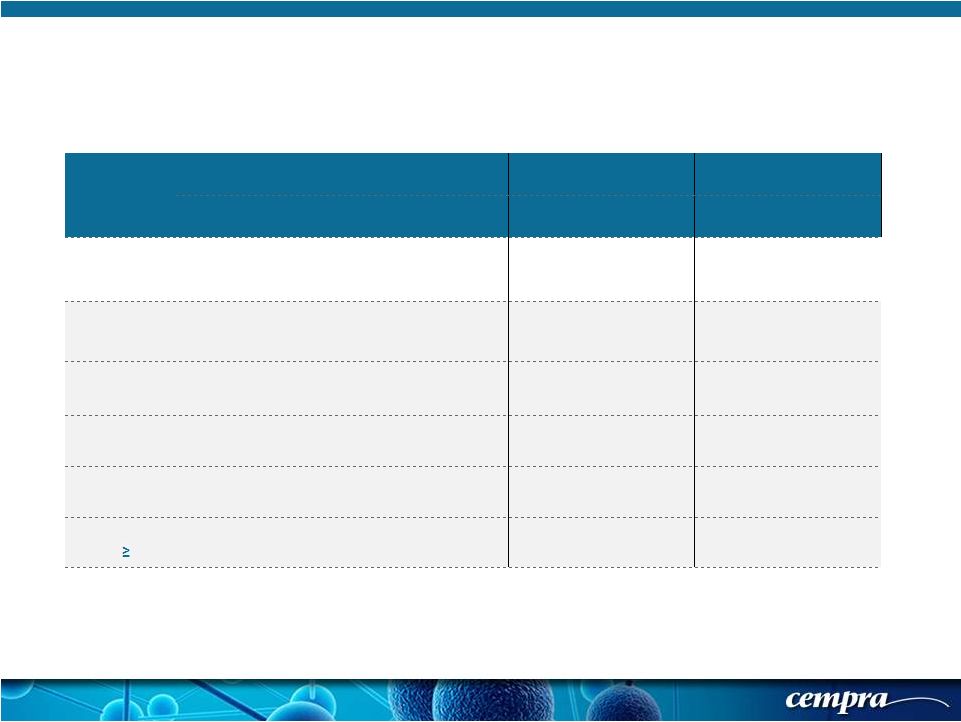 [ 17 ] [ 17 ] Population SOLITHROMYCIN MOXIFLOXACIN Success Rate % Success Rate % Difference 95% CI ECR-IT T 78.2 (333/426) 77.9 (338/434) 0.29 % (-5.5, 6.1) ECR-PORT I/II 80.5 (169/210) 80.7 (180/223) -0.24 % (-8.2, 7.7) ECR-PORT III/IV 75.9 (164/216) 74.9 (158/211) 1.04 % (-7.6, 9.7) ECR- Age < 65 77.9 (211/271) 80.8 (240/297) -2.95 % (-10.0, 4.1) ECR- Age 65-74 75.3 (70/93) 73.0 (54/74) 2.30 % (-12.3, 16.9) ECR- Age 75 83.9 (52/62) 69.8 (44/63) 14.03 % (-2.1, 30.2) Solitaire-Oral Phase 3 Trial: Primary Objective (FDA) - Early Clinical Response (ECR) in ITT Populations (& Subgroups) |
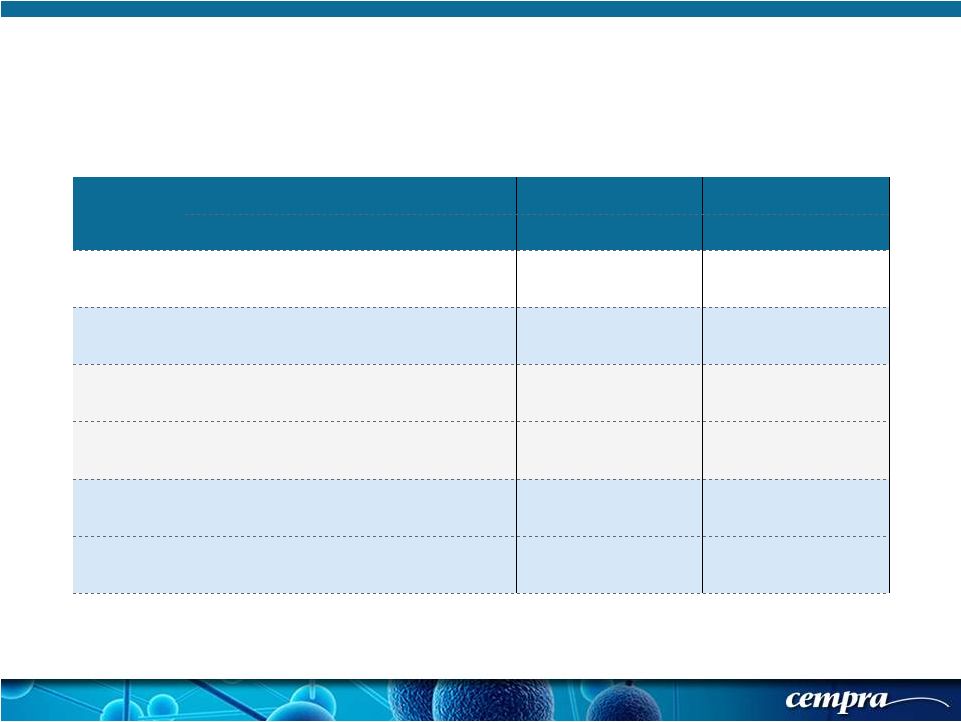 [ 18 ] [ 18 ] Population SOLITHROMYCIN MOXIFLOXACIN Success Rate % Success Rate % Difference 95% CI SFU-ITT 84.5 (360/426) 86.6 (376/434) -2.13% (-7.1, 2.8) SFU-CE 88.1 (342/388) 91.3 356/390 -3.14% (-7.7, 1.4) SFU-ITT PORT II 86.2 (181/210) 89.2 (199/223) -3.05% (-9.7, 3.6) SFU-ITT PORT III/IV 82.9 (179/216) 83.9 (177/211) -1.02% (-8.5, 6.5) SFU-CE PORT II 89.3 (175/196) 92.1 (187/203) -2.83% (-9.0, 3.4) SFU-CE PORT III/IV 87.0 (167/192) 90.4 (169/187) -3.40% (-10.3, 3.5) Solitaire-Oral Phase 3 Trial: Secondary Objective (FDA, Primary for EMA) - Short Term Follow Up (SFU) in ITT Populations (& Subgroups) |
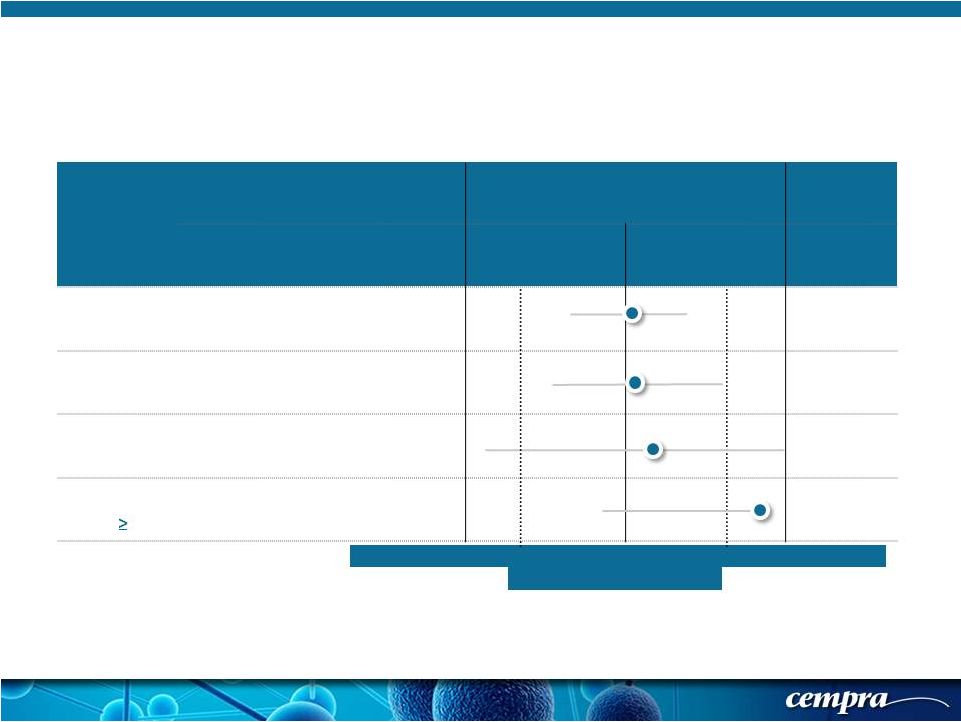 [ 19 ] [ 19 ] Population SOLITHROMYCIN MOXIFLOXACIN Favors Moxifloxacin Favors Solithromycin Success Rate % Success Rate % Delta (CI) ECR-IT T 78.2 (333/426) 77.9 (338/434) +0.24 (-5.5, 6.1) ECR-PORT III/IV 75.9 (176/235) 74.98 (178/226) +1.04 (-7.6, 9.7) ECR- Age 65-74 75.3 (70/93) 73.0 (54/74) +2.30 (-12.3, 16.9) ECR- Age 75 83.9 (52/62) 69.8 (44/63) +14.03 (-2.1, 30.2) Solitaire-Oral Phase 3 Trial: ECR-ITT Results Sub-Grouped To Demonstrate Efficacy in Older Age Groups -12 -10 -8 -6 -4 -2 0 2 4 6 8 10 12 Treatment difference |
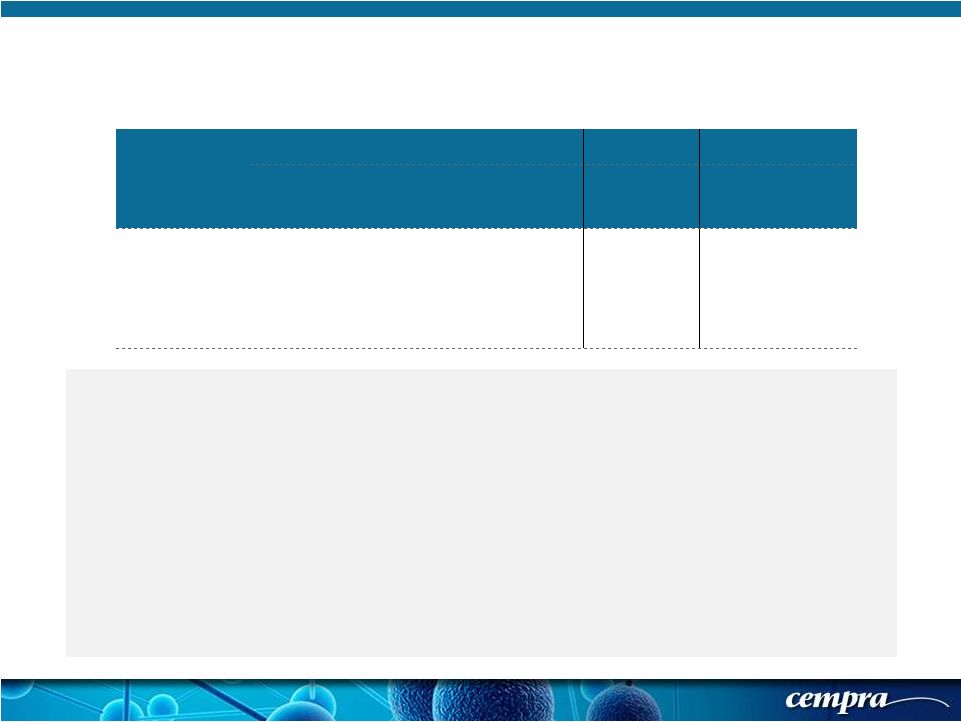 [ 20 ] [ 20 ] Population SOLITHROMYCIN MOXIFLOXACIN Success Rate % Success Rate % Delta CI ECR-mITT 74.9 (176/235) 78.8 (178/226) -3.87 (-12.0, 4.3) Solitaire-Oral Phase 3 Trial: Secondary Objective for FDA - Demonstration of NI for mITT mITT analysis will be finalized after the second study to generate the pooled weighted mITT First data set where we have met the non-inferiority endpoint In the second Phase 3 trial the drug is administered intravenously for 7 days of treatment, not 5 days as in the oral study Solithromycin 400 mg dose is 63% orally bioavailable (better than 38% for azithromycin) but is less than the 86% for moxifloxacin. In the Solitaire-IV trial, both drugs will be administered intravenously at the same dose for the same duration |
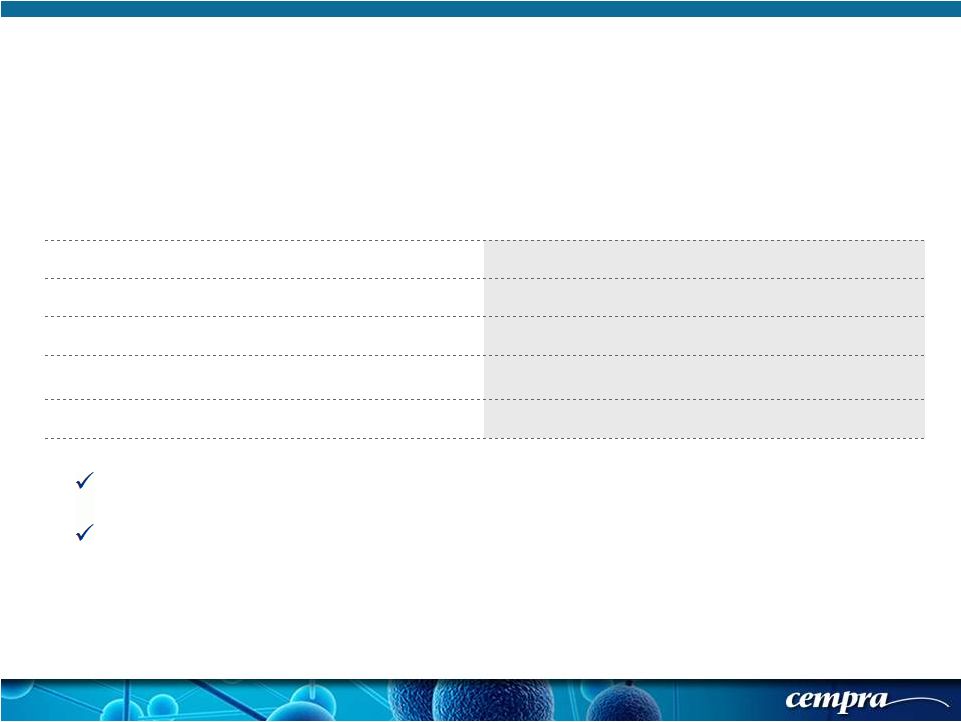 [ 21 ] [ 21 ] Solithromycin 800/400 mg QD (N=424) Moxifloxacin 400 mg QD (N=432) Any Treatment-Emergent Adverse Event (TEAE) 155 (36.6%) 154 (35.6%) Any Study Drug Related TEAE 43 (10.1%) 54 (12.5%) Any Serious Adverse Event (SAE) 28 (6.6%) 27 (6.3%) Premature Discontinuation of Study Drug from Adverse Events 16 (3.8%) 13 (3.0%) Deaths 6 (1.4%) 6 (1.4%) Solitaire-Oral Phase 3 Trial: Safety Outcomes Solithromycin Demonstrated an Acceptable Safety and Tolerability Profile, Comparable to Moxifloxacin No SAEs attributed to Solithromycin |
 Solithromycin 800/400 mg QD (N=424) Moxifloxacin 400 mg QD (N=432) Headache 4.5% 2.5% Diarrhea 4.2% 6.5%* Nausea 3.5% 3.9% Emesis 2.4% 2.3% Dizziness 2.1% 1.6% ALTs** - Grade 3 4.6% 2.1% Grade 4 0.5% 1.2% Solitaire-Oral Phase 3 Trial: Treatment Emergent Adverse Events * Not included in the diarrhea definition are 2 patients with C. difficile associated diarrhea, both of whom received moxifloxacin ** No patient in either arm of the study developed treatment emergent elevation of both ALT and bilirubin as defined by Hy’s Law criteria. Observed ALT elevations reversible and asymptomatic [ 22 ] [ 22 ] |
 [ 23 ] [ 23 ] Primary and Secondary Objectives and Endpoints Were Met for the FDA and the EMA Solitaire-Oral Phase 3 Trial: Conclusion These data will be combined with the data from the Solitaire IV Phase 3 study when that study is completed to submit the NDAs to the FDA and the EMA CABP definition and objectives and endpoints are similar between the two studies Solitaire-IV is enrolling more patients with PORTIII/IV pneumonia (75%) than in the Solitaire-Oral study (50%) and treatment is for 7 days for both drugs |