
March 2017 Developing Well-Differentiated Antibiotics Exhibit 99.1

Forward Looking Statements This presentation contains forward-looking statements regarding future events. These statements are just predictions and are subject to risks and uncertainties that could cause the actual events or results to differ materially. These risks and uncertainties include, among others: our ability to address the issues identified by the FDA in the complete response letter relating to our new drug applications for solithromycin as a treatment for community acquired bacterial pneumonia; our ability to realize the cost savings of our recently initiated cost and personnel reductions; our ability to obtain FDA and foreign regulatory approval of solithromycin for community acquired bacterial pneumonia; our ability to identify and enter into strategic business transactions; our ability to meaningfully reduce our research and corporate expenses; our and our strategic commercial partners’ ability to obtain FDA and foreign regulatory approval of our other product candidates; our ability to identify and contract with regulatory-approved manufacturers for clinical and commercial supplies of our active pharmaceutical ingredients and product candidates; the impact of the recently announced reduction in force and the changes in our senior management and our ability to retain and hire necessary employees and to staff our operations appropriately; our anticipated capital expenditures and our estimates regarding our capital requirements, including the costs of addressing the complete response letter; our dependence on the success of solithromycin and fusidic acid; results of our and our strategic commercial partners' pre-clinical studies and clinical trials are not predictive of results from subsequent clinical trials for any possible therapy; risks related to the costs, sources of funding, timing, regulatory review and results of our studies, clinical trials and regulatory applications, including activities to address the complete response letter, and those of our strategic partners; our need to obtain additional funding and our ability to obtain future funding on acceptable terms; our ability to commercialize and launch whether on our own or with a strategic partner any product candidate that receives regulatory approval; the unpredictability of the size of the markets for, and market acceptance of, any of our products, including solithromycin and fusidic acid; our ability to produce and sell any approved products and the price we are able to realize for those products; the possible impairment of, or inability to obtain, intellectual property rights and the costs of obtaining such rights from third parties; our ability to compete in our industry; innovation by our competitors; and our ability to stay abreast of and comply with new or modified laws and regulations that currently apply or become applicable to our business. Please refer to the documents that we file from time to time with the Securities and Exchange Commission.

PRODUCT CANDIDATE INDICATION FORMULATION PRECLINICAL PHASE 1 PHASE 2 PHASE 3 NDA/MAA SOLITHROMYCIN Community Acquired Bacterial Pneumonia (CABP) Oral Completed IV-to-Oral Completed Pediatric: Capsule/Suspension/IV Urethritis/Gonorrhea Oral Conjunctivitis/Blepharitis/ Dry Eye Ophthalmic FUSIDIC ACID Chronic Bone and Joint Infections Oral ABSSSI Oral NON-ANTIBIOTIC MACROLIDE Diabetic Gastroparesis and GERD Portfolio Addresses Multiple Disease Areas

An ORAL antibiotic for infections, including MRSA, being developed for ABSSSI with a potential indication for CHRONIC use in bone and joint infections in the U.S. Fusidic acid for ABSSSI and BJI INDICATION FORMULATION PRECLINICAL PHASE 1 PHASE 2 PHASE 3 FUSIDIC ACID Chronic Bone and Joint Infections Oral ABSSSI Oral

716 adult patients, randomized 1:1 (double blind) to oral fusidic acid or linezolid (Zyvox) Fusidic Acid: 1500 mg loading dose x2 + 600mg every 12 hrs for 10 days total Linezolid: 600 mg every 12 hrs for 10 days total 62 sites in the United States Patients stratified by type of infection (cellulitis, wound infection, major cutaneous abscess), by age and by prior use of an antibiotic within 36 hours prior to randomization. Primary objective: Show non-inferiority vs Zyvox for early clinical response (ECR) 67.5% of study subjects had an infection associated with IV drug abuse Most common pathogens: Staphylococcus aureus, Streptococcus anginosus group species, Streptococcus pyogenes and Clostridium species Outstanding Data from Phase 3 Study of Fusidic Acid in ABSSSI
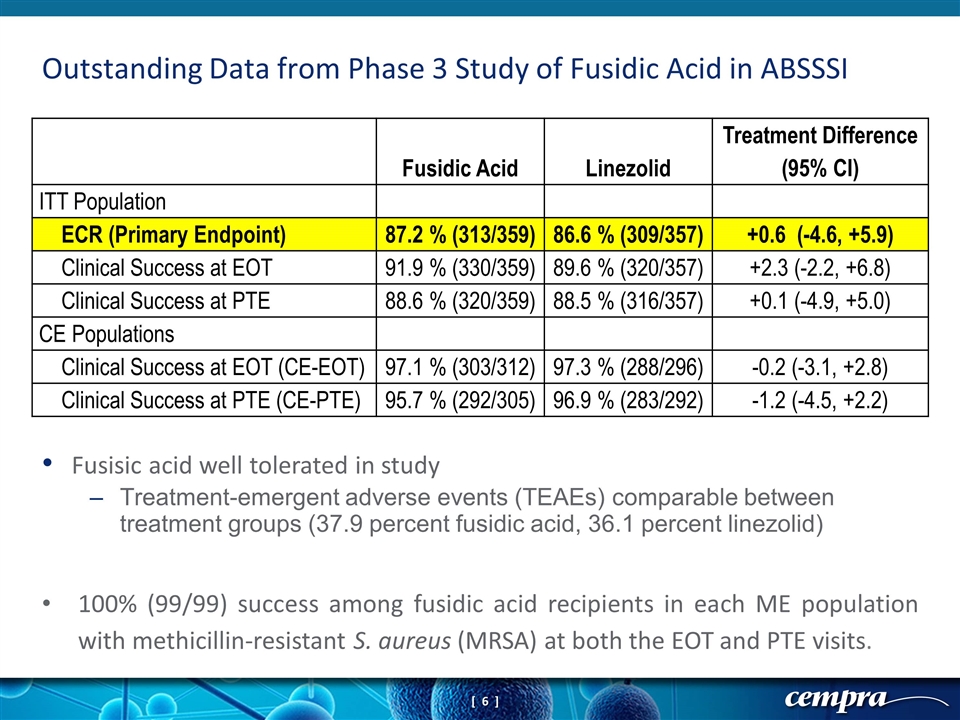
Outstanding Data from Phase 3 Study of Fusidic Acid in ABSSSI Fusidic Acid Linezolid Treatment Difference (95% CI) ITT Population ECR (Primary Endpoint) 87.2 % (313/359) 86.6 % (309/357) +0.6 (-4.6, +5.9) Clinical Success at EOT 91.9 % (330/359) 89.6 % (320/357) +2.3 (-2.2, +6.8) Clinical Success at PTE 88.6 % (320/359) 88.5 % (316/357) +0.1 (-4.9, +5.0) CE Populations Clinical Success at EOT (CE-EOT) 97.1 % (303/312) 97.3 % (288/296) -0.2 (-3.1, +2.8) Clinical Success at PTE (CE-PTE) 95.7 % (292/305) 96.9 % (283/292) -1.2 (-4.5, +2.2) Fusisic acid well tolerated in study Treatment-emergent adverse events (TEAEs) comparable between treatment groups (37.9 percent fusidic acid, 36.1 percent linezolid) 100% (99/99) success among fusidic acid recipients in each ME population with methicillin-resistant S. aureus (MRSA) at both the EOT and PTE visits.

QIDP granted for ABSSSI – exclusivity and priority review Data from refractory Phase 2/3 BJI study anticipated by year-end 2017 Primary objective: clinical success at 6 months in intent-to-treat (ITT) population Current enrollment: 27 (target=30) Next Step: Meet with FDA to discuss path forward Expectation: 2nd Phase 3 study required for potential approval in ABSSSI LPAD may offer opportunities to accelerate potential approval Progressing Fusidic Acid to Potential Approval

The Need for Solithromycin: Spectrum of in-vitro Activity Addresses CABP Pathogens GRAM ORGANISMS SOLITHROMYCIN AZITHROMYCIN CEFTRIAXONE LEVOFLOXACIN or MOXIFLOXACIN Positive Streptococcus pneumoniae P O P P Negative Haemophilus influenzae P P P P Positive Staphylococcus aureus P O P P Atypical Legionella pneumophila P P O P Atypical Mycoplasma pneumoniae P P/ O O P Atypical Chlamydophila pneumoniae P P O P Overcomes macrolide resistance that limits existing macrolides Azithromycin Monotherapy not used to Treat Moderate to Severe Pneumonia – Potency, Spectrum and Resistance Allow Use Only in Simpler Infections or Add-On To Ceftriaxone Data on file. Cempra Pharmaceuticals, Inc.
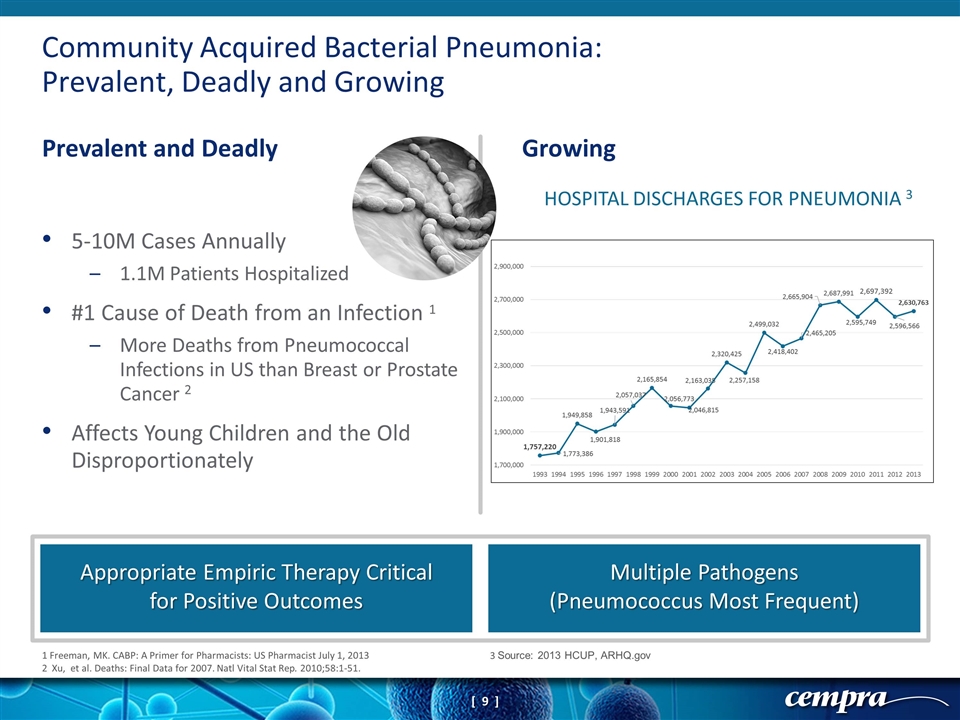
Community Acquired Bacterial Pneumonia: Prevalent, Deadly and Growing Prevalent and Deadly Growing Appropriate Empiric Therapy Critical for Positive Outcomes Multiple Pathogens (Pneumococcus Most Frequent) 1 Freeman, MK. CABP: A Primer for Pharmacists: US Pharmacist July 1, 2013 2 Xu, et al. Deaths: Final Data for 2007. Natl Vital Stat Rep. 2010;58:1-51. 3 Source: 2013 HCUP, ARHQ.gov 5-10M Cases Annually 1.1M Patients Hospitalized #1 Cause of Death from an Infection 1 More Deaths from Pneumococcal Infections in US than Breast or Prostate Cancer 2 Affects Young Children and the Old Disproportionately HOSPITAL DISCHARGES FOR PNEUMONIA 3
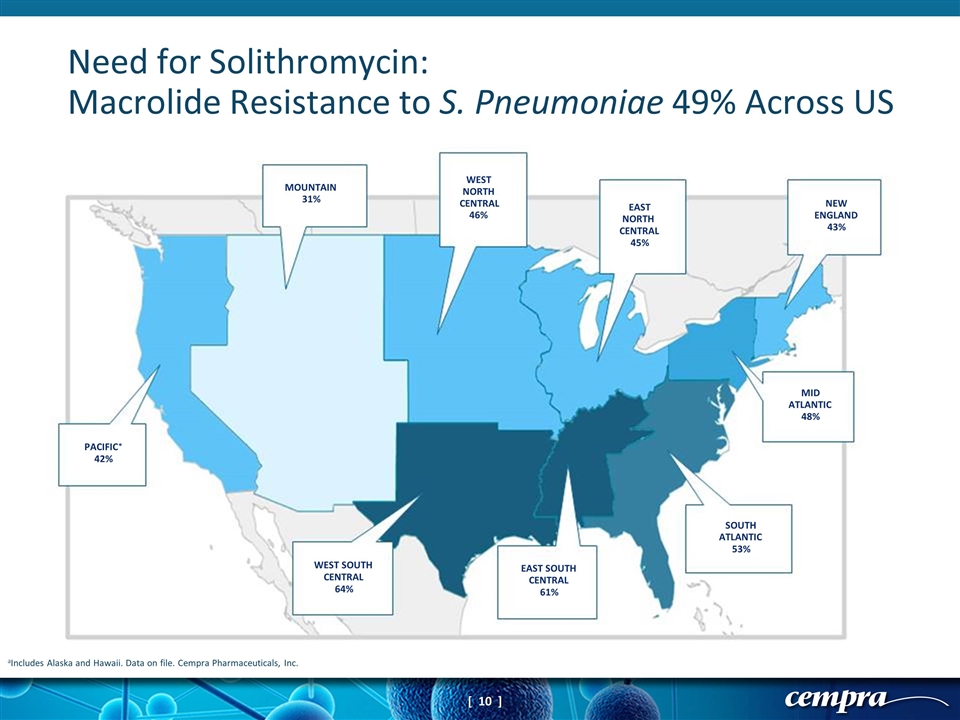
aIncludes Alaska and Hawaii. Data on file. Cempra Pharmaceuticals, Inc. Need for Solithromycin: Macrolide Resistance to S. Pneumoniae 49% Across US NEW ENGLAND 43% SOUTH ATLANTIC 53% MID ATLANTIC 48% EAST SOUTH 61% PACIFIC 42% WEST SOUTH CENTRAL 64% CENTRAL MOUNTAIN 31% WEST NORTH CENTRAL EAST NORTH CENTRAL 45% 46% *
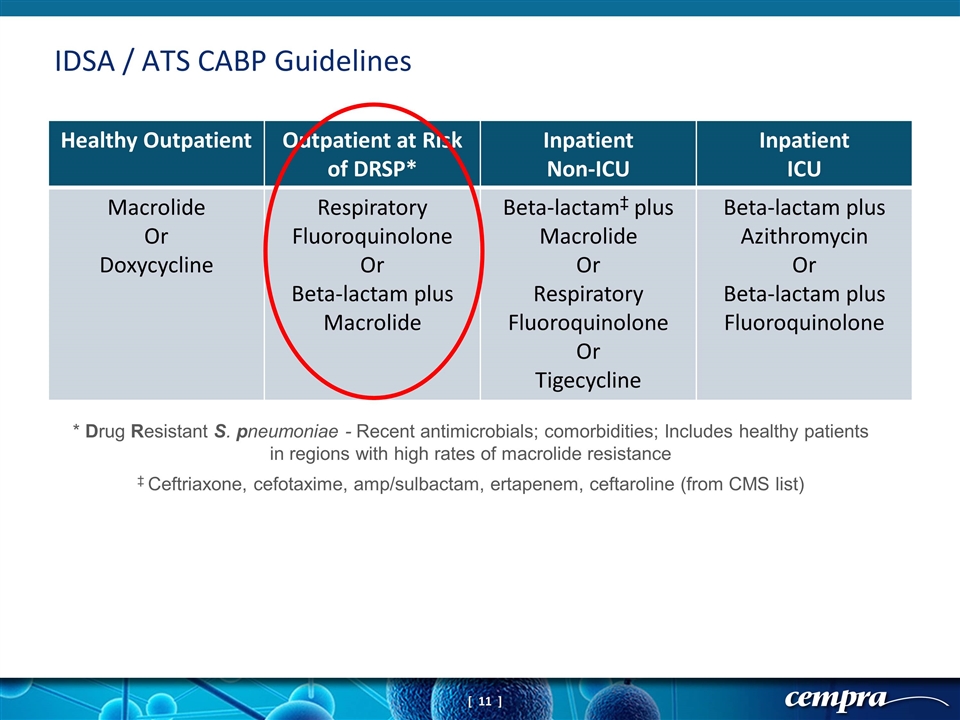
IDSA / ATS CABP Guidelines Healthy Outpatient Outpatient at Risk of DRSP* Inpatient Non-ICU Inpatient ICU Macrolide Or Doxycycline Respiratory Fluoroquinolone Or Beta-lactam plus Macrolide Beta-lactam‡ plus Macrolide Or Respiratory Fluoroquinolone Or Tigecycline Beta-lactam plus Azithromycin Or Beta-lactam plus Fluoroquinolone * Drug Resistant S. pneumoniae - Recent antimicrobials; comorbidities; Includes healthy patients in regions with high rates of macrolide resistance ‡ Ceftriaxone, cefotaxime, amp/sulbactam, ertapenem, ceftaroline (from CMS list)

Both studies achieved their primary efficacy endpoint: Non-inferior to moxifloxacin in CABP (5 to 7 day course of therapy) Observed ALT/AST elevations, consistent with other approved macrolides SOLITAIRE-ORAL 860 adult CABP patients randomized (1:1) to receive oral solithromycin or moxifloxacin SOLITAIRE IV 863 adult CABP patients randomized (1:1) to receive IV solithromycin or moxifloxacin with option for transition to oral Studies designed in accordance w/FDA Guidance for Industry for CABP trials Study design and scope discussed with FDA at end of Phase 2 meeting Strong Phase 3 Clinical Program

NDAs (Oral and IV) submitted:April 27 (oral) & 28 (IV), 2016 Proposed indication: 5-7 day one-time course of therapy for adults with CABP Advisory committee meeting: November 4, 2016 Is there substantial evidence of the efficacy of solithromycin for CABP? YES (13-0) Has the risk of hepatotoxicity been adequately characterized? NO (12-1) Does the efficacy of solithromycin for the treatment of CABP outweigh the risks, including hepatotoxicity? YES (7-6) PDUFA dates:December 27 (oral) & 28 (IV), 2016 Complete Response Letter received Solithromycin FDA Regulatory Path

No further information on solithromycin efficacy for CABP requested FDA determined risk of hepatotoxicity is not adequately characterized The FDA noted size of the safety database is limited to 920 patients FDA recommending a comparative study of approximately 9,000 patients to evaluate the safety of solithromycin in patients with CABP CRL states even in the absence of a case of Hy’s Law or another form of serious DILI in future studies, labeling will need to include adequate information about potential for hepatotoxicity, limiting use to patients who have limited treatment options and limitations regarding duration of therapy Satisfactory resolution of CMC deficiencies at Wockhardt and Hospira required prior to approval CRL Requests Additional Clinical Safety and CMC Information

February 2017 meeting with FDA to discuss CRL items and paths to resolution Details on required clinical safety study Specific deficiencies at Wockhardt and Hospira Updated FDA on CEMP progress with Uquifa FDA encouraged CEMPRA to submit protocol for discussion Cempra developing protocol which will propose: <9,000 patients at time of CRL response Delivering data in cohorts as study progresses Data in CRL response could support approval in limited group of pts w/unmet need Company would continue to accumulate safety data post-approval to support potential label expansion to broader CABP population If company and FDA agree on protocol, Cempra would seek non-dilutive financing Next Steps with FDA

BARDA funding recently initiated Phase 2/3 pediatric trial Current treatment options for children even more limited Enrolling 400 children age two months to 17 years Patients receive IV, oral suspension or oral capsule formulations of solithromycin Active comparator: standard of care therapy 3:1 randomization (~300 patients to receive solithromycin) Significant Pediatric CABP Opportunity for Solithromycin
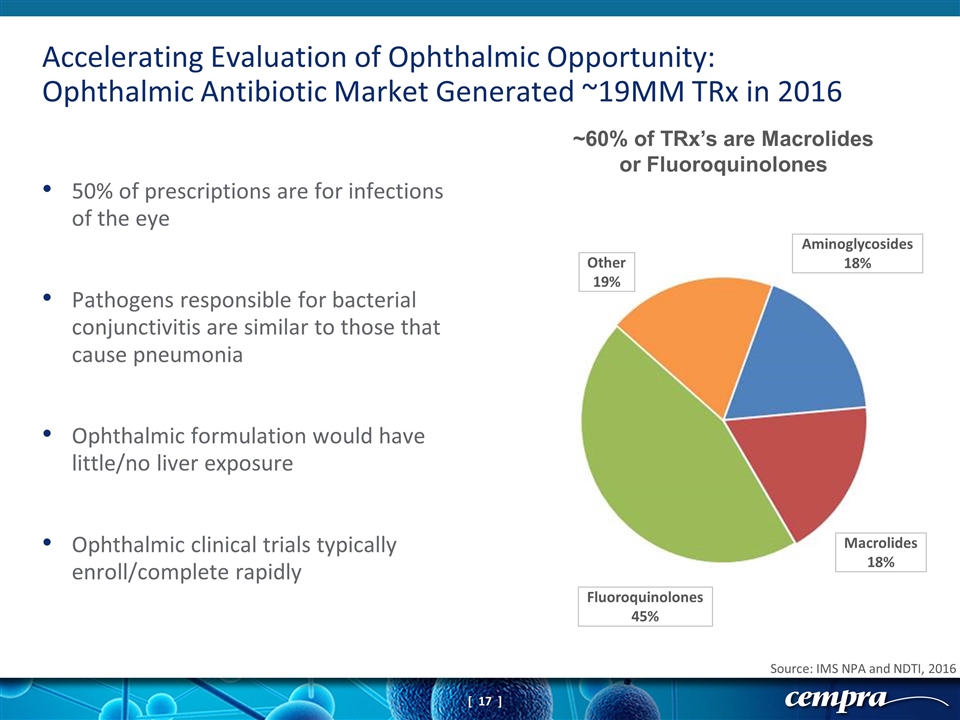
50% of prescriptions are for infections of the eye Pathogens responsible for bacterial conjunctivitis are similar to those that cause pneumonia Ophthalmic formulation would have little/no liver exposure Ophthalmic clinical trials typically enroll/complete rapidly Accelerating Evaluation of Ophthalmic Opportunity: Ophthalmic Antibiotic Market Generated ~19MM TRx in 2016 Source: IMS NPA and NDTI, 2016 ~60% of TRx’s are Macrolides or Fluoroquinolones Aminoglycosides 18% Macrolides 18% Fluoroquinolones 45% Other 19%

Workforce reduced approximately 67 percent from 136 to 45 Significant reductions in commercial & non-essential external spending Actions to reduce 2H 2017 expenses by >70% compared to 2H 2016 expenses Does not assume solithromycin launch expenses or any additional clinical trials with any of our product candidates Cash and equivalents at 12/31/16: $231.6 million Goal: Conserve financial resources as we evaluate best path forward with existing pipeline and potential business development opportunities Solithromycin patent life in U.S extends to 2032 Accelerating ophthalmic program + ongoing studies in GC, NASH, pediatrics Companywide Cost & Personnel Reductions Extend Cash Runway

Process underway to assess external late-stage assets and other potential strategic business opportunities Wide-ranging review of business opportunities Not limited to anti-infective space Open to variety of options Goal: Fully-informed assessment of external and internal opportunities to determine best use of significant cash resources and clinical programs to deliver value to patients and shareholders Evaluating External Opportunities to Drive Shareholder Value

Preserve cash Identify external business opportunities to consider Identify best path forward with fusidic acid BJI data by YE 2017 Progress manufacturing work necessary for CRL response Develop/agree to protocol to enable data for CRL response and potential approval using non-dilutive funding to support IND for ophthalmic solithromycin Cempra: 2017 Areas of Focus

Developing Well-Differentiated Antibiotics