Exhibit 99.1
IMMUNOME’S BUSINESS
Overview
We are a biopharmaceutical company focused on the development of targeted oncology therapies. We believe that the pursuit of novel or underexplored targets will be central to the next generation of transformative therapies. For that reason, we pursue therapeutics that we believe have best-in-class or first-in-class potential. Our goal is to establish a broad pipeline of preclinical and clinical assets which we can efficiently develop to value inflection points. To support that goal, we pair business development activity with significant investment in our internal discovery programs.
Our pipeline is centered on three preclinical assets: IM-1021, a ROR1 ADC; IM-4320, an anti-IL-38 immunotherapy candidate; and a currently undisclosed candidate that is a FAP radioligand therapy, or RLT, candidate. We anticipate submitting investigational new drug applications, or INDs, for each of these programs in the first quarter of 2025. We are not aware of any active development programs targeting IL-38 and believe that IM-4320, if successfully developed and approved, would be a first-in-class immunotherapy. We believe that each of these drugs has the potential to improve outcomes for patients across multiple indications.
On February 5, 2024, we signed a definitive asset purchase agreement to acquire AL102, an investigational gamma secretase inhibitor, or GSI, currently under evaluation in a Phase 3 trial for the treatment of desmoid tumors. We expect the purchase of AL102 (which also includes AL101, a related asset) to close in late Q1 or early Q2 2024. Following the closing of that deal, we will become a clinical stage company. Based on our evaluation of Phase 2 data, we believe that AL102 has the potential, if approved, to establish a new standard of care for patients with desmoid tumors.
Immunome’s business model is built upon our expertise in discovering and developing targeted therapies as well as its ability to evaluate and acquire high-potential assets. We believe that the successful track record of the leadership team will make the Company more attractive to companies selling assets, especially early-stage biotechnology companies that lack resources to efficiently develop their assets.
Our perspective is that the most important considerations when acquiring an asset are the quality of its preclinical or clinical data and the economic terms it can be acquired on. Accordingly, we are willing to consider assets across multiple modalities, including antibody drug conjugates, or ADCs, RLTs, naked antibodies, small molecules and more. We believe that effectively pursuing a novel target requires selecting a modality that is appropriate to the target biology.
At present, our internal discovery efforts are centered on ADCs and RLTs. We believe that a broad toolbox of linkers and payloads is necessary to design and develop a broad pipeline of ADCs, as different targets may require different payloads to achieve optimal efficacy and therapeutic index. The novel linker-effector unit we exclusively licensed from Zentalis is an important component of this toolbox, and we have efforts underway to develop additional linkers and payloads. We also believe that the incorporation of albumin binders into radioligand therapies provides a differentiated approach that can increase the dose of radiation absorbed by patient tumors.
Immunome’s Discovery Platform utilizes proprietary hybridoma technology to immortalize memory B cells isolated from oncology patient samples. This enables the production of sufficient quantities of antibodies to perform high-throughput functional screening, allowing for the recognition of antibodies and targets whose role in cancer was not previously appreciated. In January 2023, we announced an agreement with AbbVie under which AbbVie paid $30 million upfront for access to up to 10 targets identified by the Discovery Platform.
Immunome is led by Clay Siegall, PhD, President and Chief Executive Officer. Dr. Siegall previously served as CEO of Seagen, which he co-founded in 1997 and led for nearly 25 years. During his tenure, Seagen earned FDA approvals for four cancer therapies. Pfizer purchased Seagen in December 2023. Dr. Siegall joined Immunome in connection with Immunome’s acquisition of Morphimmune, a preclinical biotechnology company led by Dr. Siegall, in October 2023.
In addition to Dr. Siegall, three members of our current management team joined Immunome from Morphimmune in October 2023. Jack Higgins, PhD, Immunome’s Chief Scientific Officer, held the same role at Morphimmune, He was previously the Chief Development Sciences Officer at Molecular Templates, where he led discovery and development efforts for multiple clinical candidates and co-invented the company’s Engineered Toxin Body platform. Bruce Turner, MD, PhD, Immunome’s Chief Strategy Officer, held the same role at Morphimmune and previously founded several biotechnology companies including Xanadu Bio and Gennao Bio. Max Rosett, Executive Vice President, Operations, and Interim Chief Financial Officer at Immunome, served as Acting Chief Operating Officer at Morphimmune. Mr. Rosett previously served as Principal at Research Bridge Partners, where he led Research Bridge Partners’ investment in Morphimmune’s Series A financing.
Bob Lechleider, MD, serves as the Chief Medical Officer of Immunome. Dr. Lechleider was most recently the Chief Medical Officer of OncoResponse and previously worked with Dr. Siegall at Seagen, where he was responsible for directing the development of early and late-stage portfolios. Phil Roberts, serves as the Chief Technical Officer of Immunome. Dr. Roberts previously served as SVP, Technical Operations at Mirati Therapeutics, where he led the CMC development of Krazati, Mirati’s first approved product. Sandra Stoneman, JD, serves as Chief Legal Officer of Immunome. She joined Immunome from Duane Morris LLP, where she was an equity partner. Kinney Horn serves as our Chief Business Officer. He previously served in the same role at Olema Oncology and spent more than 15 years at Genentech.
Immunome’s Pipeline

ROR1 ADC (IM-1021)
On January 8, 2024, we announced that we had entered into an exclusive, worldwide license agreement under which we licensed from Zentalis ZPC-21 (now IM-1021), a preclinical-stage ADC targeting the receptor tyrosine kinase like orphan receptor 1, or ROR1. ROR1 has an oncofetal expression pattern, with little or no expression in healthy tissue, and is expressed on solid and liquid tumors. We believe ROR1 has been clinically validated as an ADC target through clinical trials of a competitor ADC in multiple B-cell malignancies.
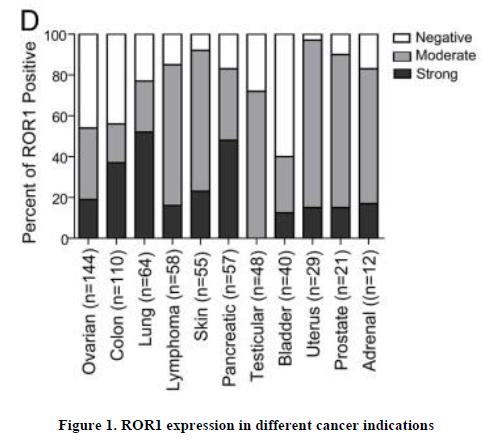
The expression pattern of ROR1 suggests that it may have clinical utility as a therapeutic target in multiple solid and liquid tumor indications, including diseases with large patient populations and high unmet need (Figure 1). However, the moderate-to-low expression and slow internalization of ROR1 present challenges to developing a successful ADC for the treatment of ROR1-positive solid tumors. Our approach to overcoming these challenges is focused on pursuing development of IM-1021, which incorporates an ROR1 antibody that is designed to promote internalization; uses a linker-payload combination that we believe provides a potentially improved therapeutic index and may allow for higher clinical dosing; and contains a payload that is designed to maximize the potential bystander effect and supports a drug-antibody ratio, or DAR, of 8.
IM-1021 incorporates a cleavable, undisclosed linker that is used to conjugate a camptothecin derivative (a topoisomerase I inhibitor) to the ROR1 antibody via cysteine conjugation, and provides a DAR of 8. In preclinical studies, IM-1021 showed sustained tumor regression in a mouse model triple-negative breast cancer, or TNBC. In this model, IM-1021 dosed weekly for three weeks at 2.5 mg/kg or 5.0 mg/kg demonstrated superior reductions in tumor volume compared with the same respective dose of a competitor, vedotin payload ROR1 ADC, with no meaningful weight loss observed (Figure 2).

Figure 2. IM-1021 showed favorable activity and safety findings in a TNBC mouse model
We expect to submit an IND for the IM-1021 program to the FDA in the first quarter of 2025. Subject to obtaining an IND, our IM-1201 clinical strategy is designed to efficiently evaluate dose escalation in patients with solid tumors or lymphoma, followed by potential expansion of the solid tumor clinical program into targeted indications, potentially including non-small cell lung cancer, breast prostate, pancreatic, and gastric cancer, and expansion of the lymphoma program into diffuse large B-cell lymphoma and mantle cell lymphoma. Concurrent with the dose escalation and expansion studies, we plan to conduct non-clinical studies evaluating IM-1021 in combination with other therapies, particularly in B-cell malignancies, and to evaluate and develop potential companion diagnostics that could help identify patients most likely to respond to IM-1021. Our strategy is to pursue pivotal clinical studies in indications that have shown compelling clinical outcomes in earlier-stage trials, present significant commercial opportunities, have the potential for enhanced outcomes using a companion diagnostic, and offer potential for accelerated approval.
177Lu-FAP Radioligand Therapy
Through our merger with Morphimmune, we acquired a FAP-targeted Lu-177 radiotherapy development candidate for the treatment of solid tumors. FAP, or fibroblast activation protein, serves as a tumor-specific marker due to its broad expression on cancer associated fibroblasts. Cancer-associated fibroblasts are the most common tumor stromal cell, with expression in 75% of solid tumors. Our FAP-Lu RLT candidate is designed to deliver radioactive 177Lu directly to FAP-expressing cells, where the “bystander” effect of the radiation may target nearby tumor cells. We believe this RLT approach could overcome the limitations, such as poor internalization and low expression on tumor cells, that make FAP an unsuitable target for ADCs.
Our FAP-targeted radiotherapy candidate has four functional domains:
| • | A small molecule FAP-specific ligand |
| • | A linker tuned to drive tumor-specific uptake |
| • | An albumin-binding domain to improve tumor retention |
| • | A chelator to deliver the radionuclide |

We have conducted preclinical studies demonstrating that incorporating albumin binders into RLTs improved biodistribution and in vivo pharmacokinetic profiles. Strong albumin binding resulted in higher total absorbed doses in tumor compared with liver and kidney (Figure 4) and also led to increased and prolonged FAP-RLT levels in serum when administered intravenously (Figure 5).

We are evaluating a series of potential drug candidates that explore options for each of the four domains in order to select the combination that we believe may be most likely to deliver therapeutic benefits in cancer patients. Several candidates have shown substantial tumor regression in an animal model of glioblastoma (Figure 6A), with no weight loss observed (Figure 6B).
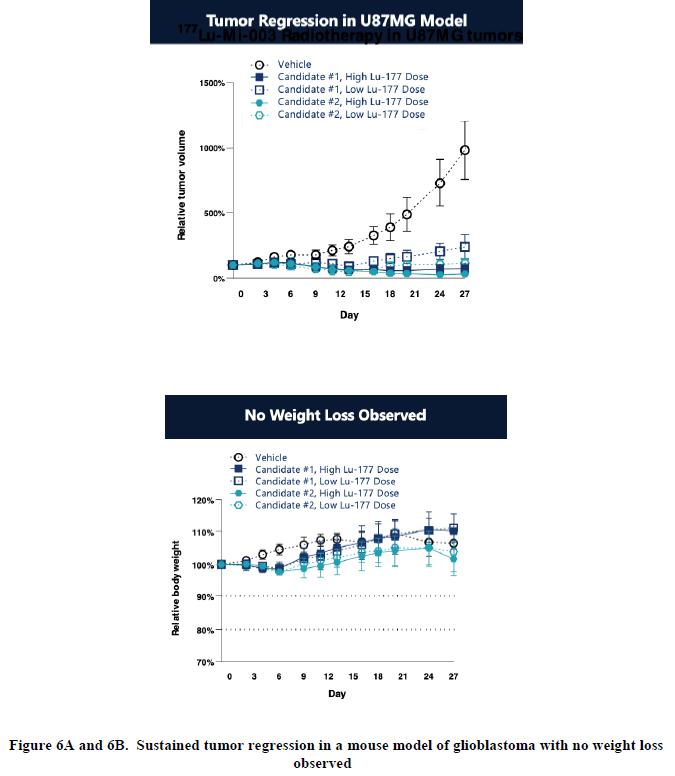
We believe our growing body of preclinical data for these candidates demonstrates desirable characteristics that support the further development of a FAP RLT. These preclinical characteristics include sub-nanomolar affinity, high specificity, radiostability, superior dose retention and tumor absorbed dose, and preclinical activity and tolerability.
We expect to submit an IND for this program to the FDA in Q1 2025.
Anti-IL-38 Immunotherapy (IM-4320)
Our lead oncology program targets IL-38, which we believe is a novel, negative regulator of inflammation capable of promoting tumor evasion of the immune system. IL-38 was identified as the target of an antibody isolated from a hybridoma library generated from the memory B cells of a patient with squamous head and neck cancer.
Our query of public and proprietary databases of cancer gene expression revealed over-expression of IL-38 in multiple solid tumors. Further, a correlation with low levels of tumor-infiltrating immune effector cells, a hallmark of immune suppression in some of these patients’ tumors, and high IL-38 expression was also observed, suggesting a role for IL-38 as an immune modulator.
Data obtained from preclinical testing showed that blocking IL-38 function using inhibitory antibodies increased the immune response to the tumor and resulted in anti-tumor activity in select animal models, suggesting that anti-IL-38 antibodies could have therapeutic utility as single agents or in combination with other therapeutic modalities. Our recent analysis further showed IL-38 expression was frequently elevated in samples of select patient tumor subtypes, in cancers such as head and neck, lung and gastroesophageal.

IM-4320 is our lead anti-IL-38 antibody. Our data indicates that IM-4320 bound to IL-38, inhibiting its binding ILRAPL1 and IL-36R. Furthermore, it inhibited tumor growth in an immune cold melanoma model. We believe IM-4320 has shown preclinical activity consistent with an active immunotherapy agent.
We expect to submit an IND for this program to the FDA in Q1 2025.
AL102
On February 5, 2024, we signed an Asset Purchase Agreement with Ayala Pharmaceuticals, Inc. or Ayala, for AL102, a gamma secretase inhibitor, or GSI, currently under evaluation in a Phase 3 trial for the treatment of desmoid tumors. We expect the purchase of AL102 (which also include AL101, a related asset) to close in late Q1 or early Q2 2024.
Desmoid tumors are painful, aggressive soft-tissue tumors that can lead to significant disability if left untreated. They are most commonly diagnosed in young adults, and they are more common in women than in men. An estimated 1,000-1,650 patients are diagnosed with desmoid tumors each year in the US, and an estimated 5,500 to 7,000 patients have desmoid tumors that are actively managed. In November 2023, OSGIVEO (nirogacestat) became the first FDA-approved systemic treatment for desmoid tumors. Like AL102, nirogacestat is a gamma secretase inhibitor.
Our interest in AL102 was largely a response to Phase 2 data, as shared by Ayala at ESMO in October 2023. Ayala’s data showed an objective response rate of 64% in the intent-to-treat population and 75% among evaluable patients. Other measures of response including tumor volume, as measured by MRI and cellularity as estimated via T2 imaging, also showed deep responses.
We believe the data published at that time suggests that AL102’s safety results were similar to nirogacestat.

ADC Strategy
We are pursuing a target-driven development strategy intended to establish a broad pipeline of next-generation ADCs focused on oncology indications with high unmet need. We are working to identify novel or under-explored targets that we believe will enable the development of first-in-class ADCs. We also are working to identify clinically validated ADC targets for which competitor programs have shown suboptimal efficacy and/or safety, with the goal of advancing best-in-class ADCs against these targets that overcome these limitations. Our ability to achieve these goals is predicated on our deep understanding of ADC target biology and our ability to deploy a broad toolbox of antibodies, linkers, and payloads in combinations that best match this biology.
Our development process is intended to efficiently advance ADC pipeline candidates through clinical proof-of-concept. We believe that key steps in this process include:
| • | Optimize the antibody portion of the ADC for binding and internalization |
| • | Incorporate proven or novel linkers |
| • | Select payloads that provide consistent cytotoxic effects |
| • | Optimize ADC pharmacology for clinical activity |
| • | Enable early go/no-go decisions via well-designed clinical trials |
We believe that identifying appropriate targets is a key challenge of ADC development. One piece of evidence for this is the concentration of current ADC development activity, with 54% of active ADC clinical programs focused on the same ten targets (Figure 7).

We believe there are several downsides to pursuing these targets, including:
| • | Potential difficulty in overcoming limitations of existing ADCs against these targets due to heterogeneity of target expression on tumor cells and/or the likelihood that changes in payload and/or linker technology will yield only incremental gains in efficacy. |
| • | Challenging development and commercialization pathways |
| • | Lower unmet patient need |
Given these downsides, we are systematically evaluating novel targets that we believe will have first-in-class potential using multiple target and antibody sources. Our Immunome Discovery Platform (described above) has already identified more than 30 novel targets and, subject to satisfaction of the closing terms of the definitive asset purchase agreement with Atreca, Inc. announced December 26, 2023 (discussed below) we will have access to more than 25 antibodies that may support the development of ADCs against novel or underexplored targets. We also screen public and proprietary expression databases to identify other potential targets for ADC development. Potential targets identified through this systematic approach are evaluated for differential expression on tumor cells compared with normal cells and additional factors.
We believe that our proprietary camptothecin derivative/topoisomerase I inhibitor payload provides a significant opportunity to develop ADCs. This payload, which is in the same class as another camptothecin derivative (deruxtecan) used in an FDA-approved ADC targeting HER2, was developed by Zentalis and is exclusively licensed by us. This proprietary payload is designed to have enhanced ADME properties, including the potential for greater in vivo potency, increased permeability that may lead to superior bystander effects, and faster clearance that may improve tolerability after cleavage. We have conducted studies in the JIMT-1 breast cancer model demonstrating that a HER2 ADC constructed with our proprietary payload provided improved activity compared with a deruxtecan-containing HER2 ADC when dosed intravenously weekly for three weeks.

We believe that camptothecin derivatives are well-suited for ADCs targeting solid tumor because they achieve a higher DAR and higher clinical doses. Additionally, a third-party comparison of two FDA-approved HER2 ADCs that contain the same antibody (trastuzumab) but different payloads showed that percentage of patients with progression-free survival was significantly higher for the ADC containing the camptothecin derivative.
In addition to our portfolio of targets, antibodies, and payloads, we also have access to novel linker technologies under our exclusive worldwide license agreement with Zentalis (discussed below).
Immunome Discovery Platform
Immunome’s Discovery Platform provides a proprietary approach to identifying cancer-associated targets. Although many of these targets are known in scientific literature, many of them were not previously known to be associated with cancer.
The workflow for our platform is as follows:
Patient Sampling: Our discovery process begins with obtaining a patient’s lymph node, tumor or blood sample and then purifying and expanding the memory B cell population. In oncology, patients sampled include those who are treatment naïve, treated with standard regimens, or have been treated with immunity enhancing therapies.
Patient Response: We fuse and immortalize thousands of these patient-derived memory B cells using proprietary methods, capturing them as hybridomas, each of which typically express an individual antibody in quantities sufficient for extensive functional screening.
Antibody Screening: For oncology, we screen individual antibodies by assessing their binding to intact cancer cells or normal cells, or by assessing their binding to a large number of different extracts of authentic tumor samples and cancer cell lines. Using our proprietary approach, we can screen up to 18,400 antibodies on a single array. Hybridomas producing antibodies that show both high-affinity binding, by typically binding at single digit nanomolar concentrations, and specific binding, by showing much higher binding to a subset of tumor cells compared to normal cells, are designated as screening “hits.” Hybridomas producing those hits can be sequenced, their immunoglobulin genes can be cloned into expression vectors, and the individual antibodies can then be produced recombinantly.
Antibody Validation: The next step in our process is to identify the specific antigen to which the antibody appears to bind with high affinity and specificity. We use one of two complementary approaches for this activity: the first method involves an assessment of antibody binding to known human proteins spotted on a protein microarray with high selectivity. If the target is not represented on the array or no specific binding is seen, we attempt to use the antibody to “pull out” the antigen from its source using immunoprecipitation, and then identify the antigen sequence using mass spectrometry. Using these two approaches we are largely successful in identifying the antigen to which newly identified antibodies are binding. We then conduct experiments to assess whether the binding of the antibody to the specific antigen can produce a change in the biology of a cancer cell expressing the target, which we refer to as target validation. Additional tests, such as measurements of changes in cell growth, cell survival, cell migration, or internalization of the antigen after it has been bound by the antibody, are used to further assess the potential that the antibody could be of therapeutic interest.
Strategic Transactions
Transactions Subject to Completion
Acquisition of Assets from Ayala Pharmaceuticals, Inc.
On February 5, 2024, the Company and Ayala Pharmaceuticals, Inc., or Ayala, entered into an Asset Purchase Agreement, or the Ayala Purchase Agreement, pursuant to which the Company will acquire Ayala’s AL101 and AL102 programs and assume certain of Ayala’s liabilities associated with the acquired assets, or the Ayala Asset Purchase. Pursuant to the Ayala Purchase Agreement, at the closing of the Ayala Asset Purchase (the Closing), the Company will (i) pay Ayala $20,000,000, subject to certain adjustments, (ii) issue Ayala 2,175,489 shares of Company common stock, or the Ayala Shares, and (iii) assume specified liabilities. The Company is obligated to pay Ayala up to $37,500,000 in development and commercial milestones.
Each party’s obligation to consummate the Ayala Asset Purchase is subject to customary closing conditions, including the accuracy of the other party’s representations and warranties as of the Closing, subject, in certain instances, to certain materiality and other thresholds, the performance by the other party of its obligations and covenants under the Ayala Purchase Agreement in all material respects, the Company’s receipt of the Stockholder Consents (defined below) from the Ayala stockholders with the requisite vote to approve a sale of substantially all of the assets of Ayala and the lapse of at least twenty (20) calendar days from the date Ayala mails a definitive information statement to its stockholders in accordance with Rule 14c-2 promulgated under the Exchange Act of 1934, as amended, receipt of certain third party consents, the delivery of certain related ancillary documents by the other party, and the absence of any injunction or other legal prohibitions preventing consummation of the Ayala Asset Purchase.
The Ayala Purchase Agreement also provides that until the six-month anniversary of the Closing, Ayala will hold and not sell 50% of the Ayala Shares, subject to certain exceptions. Further, Ayala has agreed, subject to certain exceptions, that until the one-year anniversary of the Closing, any transfer of the Ayala Shares by Ayala that exceed 15% of the average daily trading volume of the Company’s stock over the five-trading day period ending on the trading day immediately prior to such trading date shall be made pursuant to a block trade or other disposition through a market participant designated by the Company.
The Company has agreed to use its commercially reasonable efforts to (x) file a resale registration statement with the SEC registering the Ayala Shares for resale on or before the date seven days following the earlier of (i) April 1, 2024 and (ii) the date the Company files its annual report on Form 10-K for the year ended December 31, 2023 and (y) cause such resale registration statement to be declared effective as soon as practicable after the filing thereof but no later than 90 calendar days after the filing thereof or by five trading days from when the Company is notified that the SEC will not review the resale registration statement or that it will not be subject to further review.
Acquisition of Assets from Atreca, Inc.
On December 22, 2023, the Company entered into an asset purchase agreement with Atreca, Inc, or Atreca, pursuant to which the Company will acquire certain antibody-related assets and materials for an upfront payment of $5.5 million and up to $7.0 million in clinical development milestones. The closing of the transaction is subject to customary conditions, including the approval of Atreca’s stockholders.
Completed Transactions
Merger with Morphimmune
On October 2, 2023, the Company completed its merger with Morphimmune Inc., or Morphimmune. Under the terms of the Agreement and Plan of Merger and Reorganization dated as of June 28, 2023, or the Merger Agreement, among the Company, Morphimmune and Ibiza Merger Sub, Inc., a wholly owned subsidiary of the Company, or Merger Sub, Morphimmune merged with and into Merger Sub, with Morphimmune surviving as a wholly-owned subsidiary of Immunome, or the Merger. In connection with the Merger, on October 2, 2023, the Company issued and sold 21,690,871 shares of its common stock pursuant to the subscription agreements in a Private Investment in Public Equity, or PIPE, transaction which provided the Company with gross proceeds of $125.0 million.
Strategic Collaborations, License Agreements and Other Material Agreements
We believe that our technology has broad utility and could enable us to organically expand our pipeline by internal discovery. We are also dedicated to expanding our pipeline through disciplined mergers and acquisitions. We believe our approach provides us the flexibility needed to maintain a pipeline of potential product candidates and to maximize their value through internal development and strategic collaborations. The material asset collaborations, licensing and other related agreements entered into by us to date are described in greater detail below.
License Agreement with Zentalis Pharmaceuticals, Inc.
In January 2024, the Company entered into a license agreement, or the Zentalis License Agreement, with Zentalis Pharmaceuticals, Inc., or Zentalis, pursuant to which the Company received an exclusive, worldwide, royalty-bearing, sublicensable license under certain intellectual property relating to Zentalis’ proprietary ADC platform technology, ROR1 antibodies and ADCs targeting ROR1 to exploit products covered by or incorporating the licensed intellectual property rights. Under the Zentalis License Agreement, the Company is required to use commercially reasonable efforts to develop an ADC targeting ROR1, two additional ADCs, and commercialize any product that has received regulatory approval.
Under the Zentalis License Agreement, the Company paid to Zentalis upfront consideration totaling $35 million in cash and shares of Company common stock. The Company is obligated to pay Zentalis up to $150 million in development and regulatory milestones for the first product containing an ADC targeting ROR1 (a ROR1 ADC Product) to achieve such milestones and commercial milestones on ROR1 ADC Products. The Company is also obligated to pay to Zentalis mid-to-high single digit royalties on ROR1 ADC Products. In addition, the Company is obligated to pay Zentalis $25 million in development and regulatory milestones for the first product from each of the first five additional development programs using the licensed platform technology to generate products, and mid-single digit royalties on products from each such program. The Company’s royalty payment obligation will commence, on a product-by-product and country-by-country basis, on the first commercial sale of such product in such country and will expire on the latest of (a) the ten (10)-year anniversary of such first commercial sale for such product in such country, (b) the expiration of regulatory exclusivity for such product in such country, and (c) the expiration of the last-to-expire valid claim of a licensed patent covering such product in such country.
The Zentalis License Agreement will continue until the expiration of all royalty payment obligations. The Zentalis License Agreement may be terminated early by (a) either party in its entirety upon (i) the other party’s uncured material breach, subject to a notice and cure period, (ii) any insolvency event of the other party or (iii) prolonged force majeure, (b) the Company, either in its entirety or in part, for convenience upon a specified period prior written notice, or (c) Zentalis (i) in its entirety if the Company challenges one of the licensed patents or (ii) fails to meet certain development activity benchmarks within specified time periods.
Collaboration with AbbVie
On January 4, 2023, the Company entered into a collaboration and option agreement, or the Collaboration Agreement, with AbbVie Global Enterprises Ltd., or AbbVie, pursuant to which the Company will use its proprietary discovery engine to discover and validate targets derived from patients with three specified tumor types, and antibodies that bind to such targets, which may be the subject of further development and commercialization by AbbVie. The research term is at least 66 months, subject to extension in certain circumstances by specified extension periods. Pursuant to the terms of the Collaboration Agreement, with respect to each novel target-antibody pair that the Company generates that meets certain mutually agreed criteria (each, a Validated Target Pair or VTP), the Company granted to AbbVie an exclusive option (up to a maximum of 10 in total) to purchase all rights in and to such Validated Target Pair, for all human and non-human diagnostic, prophylactic and therapeutic uses throughout the world, including without limitation the development and commercialization of certain products derived from the assigned Validated Target Pair and directed to the target comprising such VTP (Products). No rights are granted by the Company to AbbVie under any of Company’s platform technology covering the Company’s discovery engine. Until the expiration of the research term, the Company is not permitted to conduct any activities in connection with targets or antibodies derived from patients with the specified tumor types, whether independently or with other third parties, except in limited circumstances with respect to certain target-antibody pairs that are no longer subject to the collaboration with AbbVie. In addition, during the term of the Collaboration Agreement, the Company is not permitted to develop products directed to targets that are included in VTPs purchased by AbbVie, or to which AbbVie still has rights under the Collaboration Agreement, whether independently or with other third parties.
Under the Collaboration Agreement, AbbVie will pay the Company an upfront payment of $30.0 million, plus certain additional platform access payments in the aggregate amount of up to $70.0 million based on the Company’s use of our discovery engine in connection with activities under each stage of the research plan, and delivery of VTPs to AbbVie. AbbVie will also pay an option exercise fee in the low single digit millions for each of the up to 10 VTPs for which it exercises an option. If AbbVie progresses development and commercialization of a Product, AbbVie will pay the Company development and first commercial sale milestones of up to $120.0 million per target, and sales milestones based on achievement of specified levels of net sales of Products of up to $150.0 million in the aggregate per target, in each case, subject to specified deductions in certain circumstances. On a Product-by-Product basis, AbbVie will pay the Company tiered royalties on net sales of Products at a percentage in the low single digits, subject to specified reductions and offsets in certain circumstances. AbbVie’s royalty payment obligation will commence, on a Product-by-Product and country-by-country basis, on the first commercial sale of such Product in such country and will expire on the earlier of (a) (i) the ten (10)-year anniversary of such first commercial sale for such Product in such country, or (ii) solely with respect to a Product that incorporates an antibody comprising a VTP (or certain other antibodies derived from such delivered antibody), the expiration of all valid claims of patent rights covering the composition of matter of any such antibody (whichever out of (i) or (ii) is later), and (b) the expiration of regulatory exclusivity for such Product in such country. The Company is potentially eligible to receive up to $2.8 billion from AbbVie under the Collaboration Agreement from the sources described above.
The Collaboration Agreement will expire upon the expiration of the last to expire royalty payment obligation with respect to all Products in all countries, subject to earlier expiration if all option exercise periods for all Validated Target Pairs expire without AbbVie exercising any option. In addition, the research term will terminate if AbbVie does not elect to make certain platform access payments at specified points during the research term, in order for the Company to continue the target discovery activities under the collaboration. The Collaboration Agreement may be terminated by (a) either party upon the other party’s uncured material breach, or upon any insolvency event of the other party, (b) AbbVie for convenience upon a specified period prior written notice, or (c) AbbVie for the Company’s breach of representations and warranties with respect to debarment or compliance with anti-bribery and anti-corruption laws. If AbbVie has the right to terminate the Collaboration Agreement for the Company’s uncured material breach or a breach of representations and warranties with respect to debarment or compliance with anti-bribery and anti-corruption laws, AbbVie may elect to continue the Collaboration Agreement, subject to certain specified reductions applicable to certain of AbbVie’s payment obligations (with a specified floor on such reductions).
Whitehead Patent License Agreement
In June 2009, we entered into an exclusive patent license agreement, or the Whitehead Agreement, with the Whitehead Institute for Biomedical Research, or Whitehead, and the Massachusetts Institute of Technology, or MIT, as licensing agent for Whitehead, pursuant to which we obtained from MIT and Whitehead a royalty-bearing exclusive license under certain patent rights of Whitehead and a royalty-bearing non-exclusive license under certain biological and chemical material of Whitehead that relate to our antibody screening platform, in each case to develop, manufacture, use, and commercialize licensed products and to develop and perform licensed processes and perform licensed services for all purposes in the United States. The foregoing license grant included the right to grant sublicenses with certain restrictions. Pursuant to the Whitehead Agreement, we are obligated to pay Whitehead up to $725,000 in the aggregate for certain development, regulatory and commercial milestones and up to $275,000 for each product or derivative that we discover using the licensed product or processes or Discovered Products. We are also obligated to pay Whitehead a low single digit royalty on net sales of licensed products and licensed processes when sold as a therapeutic or diagnostic product, a mid-single digit royalty on net sales of such licensed products or processes when sold as a research reagent, and a less than one percent royalty on net sales of Discovered Products when sold as a therapeutic or diagnostic product. Our obligation to pay royalties on net sales of Discovered Products is limited to a period of seven years from the first commercial sale of each Discovered Product. We are obligated to pay Whitehead a high single digit royalty on service income received in connection with the provision of licensed services the provision of which, absent the license granted under the Whitehead Agreement, would infringe a claim of a licensed patent. We are obligated to pay Whitehead a high first decile percentage of certain payments received from sublicensees, subject to certain reductions to single-digit percentages, and we are obligated to pay Whitehead a mid-teen percentage royalty on certain payments received from non-sublicensee corporate partners.
On November 17, 2022, the Company entered into a Letter Agreement, or Letter Agreement, with Whitehead, which became effective on January 4, 2023, upon the satisfaction of the conditions described therein. The Letter Agreement supplements the Whitehead Agreement. Pursuant to the Letter Agreement, Whitehead and the Company agreed that certain payments received by the Company from the Collaborator (as defined in the Letter Agreement) (i.e., a corporate partner, as defined in the License Agreement) would be excluded from the Company’s payment obligations to Whitehead. The Company and Whitehead further agreed, among other things, that the Company will make certain payments to Whitehead (i) as Net Sales (as defined in the License Agreement) as long as the Company receives those payments from the Collaborator on a specified number of products purchased by the Collaborator and (ii) upon the achievement of certain milestones whether by the Company or the Collaborator.
We have the right to terminate the Whitehead Agreement upon specified prior written notice to Whitehead. Whitehead may terminate the Whitehead Agreement in the event of our uncured material breach or insolvency. Additionally, Whitehead may terminate the Whitehead Agreement if we or any of our affiliates or sublicensees challenges the validity, patentability, enforceability or non-infringement of the licensed patents.
License Agreement with Purdue Research Foundation
In January 2022, Morphimmune entered into a Master License Agreement (Purdue License Agreement) with Purdue Research Foundation, or PRF. Under the Purdue License Agreement, PRF granted Morphimmune a royalty-bearing, transferable, worldwide, exclusive license, sublicensable through multiple tiers, under certain patents and technology owned by PRF relating to, among other subject matter, drugs to target Fibroblast Activation Protein (FAP), to research, develop, manufacture, and commercialize products covered by the licensed patents in all fields of use with limited exceptions. The license is subject to certain rights of the U.S. government and rights retained by PRF (i) to practice and to license any government agencies, universities or other educational institutions to practice, make, and use the intellectual property licensed to Morphimmune on a royalty-free basis for non-commercial uses, (ii) to conduct activities required under sponsored research agreements with Morphimmune, and (iii) to disseminate and publish materials and scientific findings from PRF’s research related to the intellectual property licensed to Morphimmune. Morphimmune is obligated to use commercially reasonable efforts to develop and commercialize the licensed products in accordance with a development and commercialization plan and to achieve agreed development milestones according to a specified timeline. PRF is obligated to prosecute and maintain the licensed patents at Morphimmune’s cost and expense.
Under the Purdue License Agreement, Morphimmune paid PRF a one-time upfront payment of $200,000 upon execution and $100,000 on each of the first and second anniversary of the effective date of the Purdue License Agreement. During the period commencing on the date of first commercial sale of a licensed product and ending upon the date of expiration of the last valid claim of the licensed patents covering such licensed product in a country, referred to as the royalty term, Morphimmune will pay PRF an earned unit royalty of a low single-digit percentage on gross receipts from sale of the licensed product, and beginning with the first sale of a licensed product, a tiered minimum annual royalty from the low to mid six-digit figure range less the unit royalties due for the annual period. Upon the achievement of specified development and commercialization milestones, Morphimmune will pay PRF the milestone payments as specified in the Purdue License Agreement, which may be up to $3.75 million in the aggregate. Morphimmune is also required to pay PRF an annual maintenance fee ranging from a low five-digit figure to a low six-digit figure prior to first sale of a licensed product and a low double-digit percentage of sublicense income received for sublicenses of licensed intellectual property, with such percentage depending upon the timing of execution of the sublicense.
The Purdue License Agreement expires on a licensed product-by-licensed product and country-by-country basis, upon expiration of the royalty term for such licensed product for the applicable country. Morphimmune may terminate the Purdue License Agreement upon at least one month’s prior written notice to PRF. PRF may terminate the Purdue License Agreement and the licenses granted thereunder if Morphimmune fails to cure a payment default or other material breach of the Purdue License Agreement after written notice from PRF, or if Morphimmune becomes insolvent.
Manufacturing
We produce our lead antibodies at the laboratory scale necessary for early research and development activities and some preclinical assessments. For later stage preclinical assessment, such as IND-enabling studies and safety assessment and early-stage clinical assessment, we use third-party manufacturers to produce our antibodies, antibody drug conjugates and drug substance for our FAP program, and any other necessary intermediates or reagents. We do not have, and we do not currently plan to acquire or develop the infrastructure, facilities or capabilities to conduct these manufacturing activities ourselves. We intend to continue to utilize third-party manufacturers to produce, package, label, test and release product for clinical and non-clinical testing and for future commercial use, as needed. We expect to continue to rely on such third parties to manufacture our products for the foreseeable future. Our expected future contractual manufacturing organizations will each have successful track records of producing products for other companies under applicable compliance regulations, such as cGMP compliance in case of the FDA.
Competition
The development and commercialization of new product candidates is highly competitive. We compete in the segments of the pharmaceutical, biotechnology and other related markets that develop therapies for the treatment of cancer, which is highly competitive with rapidly changing standards of care. As such, our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer or less severe side effects, are more convenient, or are less expensive than any products that we may develop or that would render any products that we may develop obsolete or non-competitive. Our competitors also may obtain marketing approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market.
In oncology, we expect to compete with companies advancing antibodies, antibody drug conjugates, or ADCs, small molecules, targeted radiotherapies, and other therapeutic modalities. We are aware of competitors who are pursuing antibody-based discovery approaches, including, but not limited to, AbCellera Biologics, Inc.; Adaptive Biotechnologies Corporation, or Adaptive; AIMM Therapeutics B.V.; IGM Biosciences, Inc.; OncoReponse, Inc. We also expect to compete with companies pursuing targeted radiotherapies, including, but not limited to, RayzeBio, Fusion Pharmaceuticals, POINT Biopharma, Aktis Oncology, Actinium Pharmaceuticals, and Yantai LNC Biotechnology. In addition, we expect to compete with large, multinational pharmaceutical companies that discover, develop and commercialize antibodies, ADCs, small molecules, targeted radiotherapies, and other therapeutics for use in treating cancer such as Immunogen (acquired by AbbVie Inc.), AstraZeneca; Amgen; Bayer AG, Bristol-Myers Squibb Company; Eli Lilly and Company; Genentech, Inc. (a member of Roche group); Merck & Co. Inc.; Novartis; Seagen (acquired by Pfizer) and Johnson & Johnson. If any future product candidates identified through our current lead programs are eventually approved for sale, they will likely compete with a range of treatments that are either in development or currently marketed for use in those same disease indications.
Subject to the closing of the Ayala Transaction and our acquisition of AL102, we expect to compete with companies advancing treatment of desmoid tumors, including but not limited to, SpringWorks Therapeutics, Inc. In November 2023, Springworks received FDA approval for its oral gamma secretase inhibitor, OGSIVEOTM (nirogacestat), for the treatment of adult patients with progressing tumors who require systemic treatment. Desmoid tumors treatments also include surgery, hormonal therapy, targeted therapy and chemotherapy.
There are several other companies developing FAP-targeted radioligand therapies which may represent the most direct competition to our 177Lu-FAP program. Novartis is advancing a FAP-targeted radioligand therapy (177Lu-FAP-2286) that was acquired from Clovis Oncology and is currently in Phase 1/2. Clovis previously presented Phase 1 data for FAP-2286 (June 2022.) In December 2023, Eli Lilly and Company acquired POINT Biopharma, which is developing a FAP-targeted radioligand therapy (PNT2004) that is currently in Phase 1. POINT presented a trial-in-progress poster discussing trial design (June 2023) and expects to release data from that trial in the first half of 2024. In addition, POINT has disclosed two preclinical radioligand programs targeting FAP. Yantai LNC Biotechnology has also initiated a Phase 1 trial for another FAP-targeted radioligand therapy (LNC1004.) Additionally, our 177Lu-FAP program faces competition from competitors who may have superior access to a consistent supply of radioactive isotopes.
In January 2023, we exclusively licensed a preclinical ROR1 ADC program from Zentalis with the potential to address hematologic and solid tumor indications. There are several other companies developing antibodies, antibody-drug conjugates, and CAR-T therapies targeting ROR1, and they may represent the most direct competition to our ROR1 ADC program. Merck has an ADC program (Zilovertamab vedotin) in a Phase 2/3 clinical trial for B-cell lymphoma. Boehinger Ingelheim has an ADC program in a Phase 1/2 clinical trial (NBE-002) in solid tumors, and Legochem Biosciences has an ADC program (LCB71) in a Phase 1 trial in solid tumors. Companies advancing clinical ROR1-CAR T therapy programs include Octernal Therapeutics (ONCT-808) in a Phase 1/2 in B-cell malignancies, and Lyell Immunopharma (LYL797) in a Phase 1 trial.
Many of our competitors have significantly greater financial resources and expertise in research and development, manufacturing, preclinical studies, conducting clinical studies, obtaining regulatory approvals and marketing approved products than we have. These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical study sites and patient registration for clinical studies, as well as in acquiring technologies complementary to, or necessary for, our programs. In addition, these larger companies may be able to use their greater market power to obtain more favorable supply, manufacturing, distribution and sales-related agreements with third parties, which could give them a competitive advantage over us.
Further, as more product candidates within a particular class of drugs proceed through clinical development to regulatory review and approval, the amount and type of clinical data that may be required by regulatory authorities may increase or change. Consequently, the results of our clinical trials for product candidates in that class will likely need to show a risk benefit profile that is competitive with or more favorable than those products and product candidates in order to obtain marketing approval or, if approved, a product label that is favorable for commercialization. If the risk benefit profile is not competitive with those products or product candidates, or if the approval of other agents for an indication or patient population significantly alters the standard of care with which we tested our product candidates, we may have developed a product that is not commercially viable, that we are not able to sell profitably or that is unable to achieve favorable pricing or reimbursement. In such circumstances, our future product revenue and financial condition would be materially and adversely affected.
Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These third parties compete with us in recruiting and retaining qualified scientific and management personnel, establishing clinical study sites and subject enrollment for clinical studies, as well as in acquiring technologies complementary to, or necessary for, our current or future products or programs.
Intellectual Property
Intellectual property is of vital importance in our field and in biotechnology generally. We seek to protect and enhance proprietary technology, inventions, and improvements that are commercially important to the development of our business by seeking, maintaining, and defending patent rights, whether developed internally, acquired or licensed from third parties. We will also seek to rely on regulatory protection afforded through orphan drug designations, inclusion in expedited development and review, data exclusivity, market exclusivity and patent term extensions where available.
We utilize various types of intellectual property assets to provide multiple layers of protection. For example, we seek a variety of patents to protect our inventions including, for example, compositions of matter and uses in treatment and diagnostic and methods for novel antibodies, including methods of treatment for diseases expressing novel targets. We believe our current layered patent estate, together with our efforts to develop and patent next generation technologies, provides us with substantial intellectual property protection.
As of February 8, 2024, we own or exclusively in-license 1 issued U.S. patent, 6 pending U.S. non-provisional patent applications, 1 pending U.S. provisional patent application, and 53 pending patent applications in Australia (3), Brazil (3), Canada (4), China (4), Europe (5), Hong Kong (1), Israel (3), India (3), Japan (4), Korea (3), Mexico (3), New Zealand (3), Russia (3), Singapore (3), Taiwan (3), South Africa (3), and the UAE (2) , in total 6 patent families, covering our IM-4320 (IL-38), IM-1021 (ROR1), and IM-3050 (177Lu-FAP) products. Our portfolio includes issued and/or pending claims directed to the composition of matter and methods of use for IM-4320, IM-1021 and IM-3050. Patent applications covering IM-4320, if issued, are expected to expire between 2040 and 2042, absent any patent term extensions or adjustments and without accounting for terminal disclaimers. Patent applications covering IM-1021, if issued, are expected to expire in 2042, absent any patent term extensions or adjustments and without accounting for terminal disclaimers. Patent applications covering IM-3050, if issued, are expected to expire in 2045, absent any patent term extensions or adjustments and without accounting for terminal disclaimers. However, we recognize that the area of patent and other intellectual property rights in biotechnology is an evolving one with many risks and uncertainties, which may affect those rights.
Our commercial success will depend in significant part upon obtaining and maintaining patent protection and trade secret protection for our targeted effector-based therapeutics and the methods used to develop and manufacture them, as well as successfully defending these patents against third-party challenges and operating without infringing on the proprietary rights of others. Our ability to stop third parties from making, using, selling, offering to sell or importing our products depends on the extent to which we have rights under valid and enforceable patents or trade secrets that cover these activities. We cannot be sure that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents, or any patents granted to us in the future will be commercially useful in protecting our targeted effector-based therapeutics, current programs and processes. For this and more comprehensive risks related to our intellectual property, please see the section titled “Risk Factors — Risks Related to Our Intellectual Property.”
The term of individual patents depends upon the legal term of the patents in the countries in which they are obtained. In most countries in which we file, including the United States, the patent term is 20 years from the earliest date of filing a non-provisional patent application. In the United States, a patent’s term may potentially be lengthened by patent term adjustment, or PTA, which compensates a patentee for administrative delays by the USPTO in examining and granting a patent. In the United States, the patent term of a patent that covers an FDA-approved drug may also be eligible for patent term extension, or PTE, which permits patent term restoration as compensation for the patent term lost during the FDA regulatory review process. The Hatch-Waxman Act permits PTE of up to five years beyond the expiration of the patent. The length of the PTE accorded a patent is related to the length of time the drug is under regulatory review by the FDA. PTE cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval. Further, only one patent applicable to an approved drug may be extended, and only those claims covering the approved drug, a method for using it, or a method for manufacturing it may be extended. Similar provisions for extending the term of a patent that covers an approved drug are available in multiple European countries and other foreign jurisdictions. In the future, if and when our products receive FDA approval, we expect to apply for patent term extensions on patents covering those products. We expect to seek patent term extensions to all of our issued patents in any jurisdiction where these are available; however, there is no guarantee that the applicable authorities, including the FDA in the United States, will agree with our assessment of whether such extensions should be granted, and if granted, the length of such extensions. Patent term in the U.S. may be shortened if a patent is terminally disclaimed over an earlier-filed patent.
In some instances, we file provisional patent applications directly in the USPTO. Provisional patent applications are designed to provide a lower-cost first patent filing in the United States. Corresponding non-provisional patent applications must be filed not later than 12 months after the provisional application filing date. The corresponding non-provisional application benefits in that the priority date(s) of the non-provisional patent application is/are the earlier provisional application filing date(s), and the patent term of the finally issued patent is calculated from the earliest non-provisional application filing date. This system allows us to obtain an early priority date, obtain a later start to the patent term and to delay prosecution costs, which may be useful in the event that we decide not to pursue examination in a subsequent non-provisional application. While we intend, as appropriate, to timely file non-provisional patent applications relating to our provisional patent applications, we cannot predict whether any such non-provisional patent applications will result in the issuance of patents that provide us with any competitive advantage.
We intend to file U.S. non-provisional applications and/or international Patent Cooperation Treaty, or PCT, applications that claim the benefit of the priority date of earlier filed provisional or non-provisional applications, when applicable. The PCT system allows for a single PCT application to be filed within 12 months of the priority filing date of a corresponding priority patent application, such as a U.S. provisional or non-provisional application, and to designate all of the 157 PCT contracting states in which national phase patent applications can later be pursued based on the PCT application. The PCT International Searching Authority performs a patentability search and issues a non-binding patentability opinion which can be used to evaluate the chances of success for the national applications in foreign countries prior to having to incur the filing fees. Although a PCT application does not issue as a patent, it allows the applicant to establish a patent application filing date in any of the member states and then seek patents through later-filed national-phase applications. No later than either 30 or 31 months from the earliest priority date of the PCT application, separate national phase patent applications can be pursued in any of the PCT member states, depending on the deadline set by individual contracting states. National phase entry can generally be accomplished through direct national filing or, in some cases, through a regional patent organization, such as the European Patent Organization. The PCT system delays application filing expenses, allows a limited evaluation of the chances of success for national/regional patent applications and allows for substantial savings in comparison to having filed individual countries rather than a PCT application in the event that no national phase applications are filed.
For all patent applications, we determine claiming strategy on a case-by-case basis. Advice of counsel and our business model and needs are always considered. We file patent applications containing claims for protection of all commercially-relevant uses of our proprietary technologies and any products, as well as all new applications and/or uses we discover for existing technologies and products, assuming these are strategically valuable. We may periodically reassess the number and type of patent applications, as well as the pending and issued patent claims to ensure that coverage and value are obtained for our processes, and compositions, given existing patent law and court decisions. Further, claims may be modified during patent prosecution to meet our intellectual property and business needs.
We recognize that the ability to obtain patent protection and the degree of such protection depends on a number of factors, including the extent of the prior art, the novelty and non-obviousness of the invention, and the ability to satisfy subject matter, written description, and enablement requirements of the various patent jurisdictions. In addition, the coverage claimed in a patent application can be significantly reduced before the patent is issued, and its scope can be reinterpreted or further altered even after patent issuance. Consequently, we may not obtain or maintain adequate patent protection for any of our targeted effector-based therapeutics. We cannot predict whether the patent applications we are currently pursuing will issue as patents in any particular jurisdiction or whether the claims of any issued patents will provide sufficient proprietary protection from competitors. Any patents that we hold may be challenged, circumvented or invalidated by third parties.
In addition to patent protection, we also rely on trademark registration, trade secrets, know how, other proprietary information and/or continuing technological innovation to develop and maintain our competitive position. We seek to protect and maintain the confidentiality of proprietary information to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection. Although we take steps to protect our proprietary information and trade secrets, including through contractual means with our employees and consultants, third parties may independently develop substantially equivalent proprietary information and techniques or otherwise gain access to our trade secrets or disclose our technology. Thus, we may not be able to meaningfully protect our trade secrets. It is our policy to require our employees, consultants, outside scientific collaborators, sponsored researchers and other advisors to execute confidentiality agreements upon the commencement of employment or consulting relationships with us. These agreements provide that all confidential information concerning our business or financial affairs developed or made known to the individual during the course of the individual’s relationship with us is to be kept confidential and not disclosed to third parties except in specific circumstances. Our agreements with employees also provide that all inventions conceived by the employee in the course of employment with us or from the employee’s use of our confidential information are our exclusive property. However, such confidentiality agreements and invention assignment agreements can be breached, and we may not have adequate remedies for any such breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our consultants, contractors or collaborators use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting trade secrets, know-how and inventions.
The patent positions of biotechnology companies like ours are generally uncertain and involve complex legal, scientific and factual questions. Our commercial success will also depend in part on not infringing upon the proprietary rights of third parties. It is uncertain whether the issuance of any third-party patent would require us to alter our development or commercial strategies, or our products or processes, obtain licenses or cease certain activities. Our breach of any license agreements or our failure to obtain a license to proprietary rights required to develop or commercialize our future products may have a material adverse impact on us. If third parties prepare and file patent applications in the United States that also claim technology to which we have rights, we may have to participate in interference or derivation proceedings in the USPTO to determine priority of invention.
When available to expand market exclusivity, our strategy is to obtain, or license additional intellectual property related to current or contemplated development platforms, core elements of technology and/or programs and targeted effector-based therapeutics.
For more information regarding the risks related to our intellectual property, see the section titled “Risk Factors — Risks Related to Our Intellectual Property.”
Government Regulation
The FDA and other regulatory authorities at federal, state, and local levels, as well as in foreign countries, extensively regulate, among other things, the research, development, testing, manufacture, quality control, import, export, safety, effectiveness, labeling, packaging, storage, distribution, record keeping, approval, advertising, promotion, marketing, post-approval monitoring, and post-approval reporting of drugs and biologics. We, along with our third-party contractors, will be required to navigate the various preclinical, clinical and commercial approval requirements of the governing regulatory agencies of the countries in which we wish to conduct studies or seek approval or licensure of our programs and development candidates.
U.S. Government Regulation of Drugs and Biologics
In the United States, the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act, or FDCA, and its implementing regulations and biologics under the FDCA, the Public Health Service Act, or PHSA, and their implementing regulations. Both drugs and biologics also are subject to other federal, state and local statutes and regulations. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state and local statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements may subject an applicant to administrative or judicial sanctions, such as FDA refusal to approve pending new drug applications, or NDAs, or biologics license applications, or BLAs, or the agency's issuance of warning letters, or the imposition of fines, civil penalties, product recalls, product seizures, total or partial suspension of production or distribution, injunctions and/or criminal prosecution brought by the FDA and the U.S. Department of Justice or other governmental entities.
Nonclinical and Clinical Development
Nonclinical studies include laboratory evaluation of product chemistry and formulation and may involve in vitro testing or in vivo animal studies to assess the potential for toxicity, adverse events, and other safety characteristics of the program or development candidate, and in some cases to establish a rationale for therapeutic use. The conduct of nonclinical studies is subject to federal regulations and requirements, including good laboratory practice regulations for safety/toxicology studies.
The sponsor must submit the results of the preclinical studies, together with manufacturing information, analytical data, any available clinical data or literature and a proposed clinical protocol, as well as other information, to the FDA as part of the Investigational New Drug application, or IND. Some long-term nonclinical testing as well as manufacturing process development and product quality evaluation, continues after the IND is submitted.
Human clinical trials in support of an NDA or BLA
Prior to beginning the first clinical trial with a program or development candidate, the sponsor must submit an IND to the FDA. The central focus of an IND submission is on the general investigational plan and the protocol(s) for clinical trials. An IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, raises concerns or questions related to the proposed clinical trial and places the IND on a clinical hold. In such a case, the IND sponsor must resolve all outstanding concerns or questions posed by the FDA before the clinical trial can begin. Submission of an IND therefore may not result in FDA authorization to begin a clinical trial.
Clinical trials involve the administration of the investigational product to human subjects under the supervision of qualified investigators in accordance with Good Clinical Practices, or GCPs, which include the requirement that all research subjects provide their informed consent for their participation in any clinical study. Clinical trials are conducted under protocols detailing, among other things, the objectives of the study, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. A separate submission to the existing IND must be made for each successive clinical trial conducted during product development and for any subsequent protocol amendments. Furthermore, an independent institutional review board, or IRB, for each site proposing to conduct the clinical trial must review and approve the plan for any clinical trial and its informed consent form before the clinical trial begins at that site and must monitor the study until completed. Regulatory authorities, the IRB or the sponsor may suspend a clinical trial at any time on various grounds, including a finding that the subjects are being exposed to an unacceptable health risk or that the trial is unlikely to meet its stated objectives. Some studies also include oversight by an independent group of qualified experts organized by the clinical trial sponsor, known as a data safety monitoring board, or DSMB, which provides authorization for whether a study may move forward at designated check points based on review of certain data from the study, to which only the DSMB has access, and may recommend halting the clinical trial if it determines that there is an unacceptable safety risk for subjects or other grounds, such as no demonstration of efficacy. Progress reports detailing the results of the clinical trials, among other information, must be submitted at least annually to the FDA, and written IND safety reports must be submitted to the FDA and the investigators for serious and unexpected suspected adverse reactions, findings from other studies suggesting a significant risk to humans exposed to the investigational product, findings from animal or in vitro testing that suggest a significant risk for human subjects, and any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator brochure.
Sponsors of clinical trials of certain FDA-regulated products must register and disclose certain clinical trial information to a public registry maintained by the National Institutes of Health, or NIH. In particular, information related to the investigational product, patient population, phase of investigation, trial sites and investigators and other aspects of the clinical trial is made public as part of the registration of the clinical trial. Although sponsors are also obligated to disclose the results of their clinical trials after completion, disclosure of the results can be delayed in some cases for up to two years after the date of completion of the trial. Failure to timely register a covered clinical study or to submit study results as provided for in the law can give rise to civil monetary penalties and also prevent the non-compliant party from receiving future grant funds from the federal government.
For purposes of NDA or BLA approval, human clinical trials are typically conducted in three sequential phases that may overlap.
| Phase 1 — The investigational product is initially introduced into healthy human subjects or directly into patients with the target disease or condition for certain therapies targeting severe or life-threatening diseases where the investigational product may be too inherently toxic to administer ethically to healthy volunteers. In either case, these studies are designed to test safety, dosage tolerance, absorption, metabolism, distribution and excretion of the investigational product in humans, the side effects associated with increasing doses, and, if possible, to gain early evidence on effectiveness. |
| Phase 2 — The investigational product is administered to a limited patient population with a specified disease or condition to evaluate the preliminary efficacy, optimal dosages and dosing schedule and to assess adverse events and potential side effects. Multiple Phase 2 clinical trials may be conducted to obtain information prior to beginning larger and more expensive Phase 3 clinical trials. |
Phase 3 — The investigational product is administered to an expanded patient population to further evaluate dosage, to provide statistically significant evidence of clinical efficacy and to further test for safety, generally at multiple geographically dispersed clinical trial sites. These clinical trials are intended to establish the overall risk/benefit ratio of the investigational product and, if appropriate, to provide an adequate basis for product approval. These trials may include comparisons with placebo and/or other comparator treatments. The duration of treatment is often extended to mimic the actual use of a product during marketing. |
In some cases, the FDA may require, or companies may voluntarily pursue, additional clinical trials after a product is approved to gain more information about the product. These so-called Phase 4 studies may be made a condition to approval of the NDA or BLA; failure to exhibit due diligence with regard to conducting these Phase 4 clinical trials could result in withdrawal of approval for products. Concurrent with clinical trials, companies may complete additional nonclinical studies and develop additional information about the characteristics of the investigational product and must finalize a process for manufacturing the product in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the candidate and, among other things, must develop methods for testing the identity, strength, quality and purity of the final product, or for biologics, the safety, purity and potency. Additionally, appropriate packaging must be selected and tested, and stability studies must be conducted to demonstrate that the candidate does not undergo unacceptable deterioration over its shelf life.
BLA and NDA Submission and Review
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, the results of product development, nonclinical studies and clinical trials, along with information relating to the product’s chemistry, manufacturing, and controls and proposed labeling, are submitted to the FDA as part of an NDA or BLA requesting approval to market the product for one or more indications. An NDA or BLA must contain sufficient evidence of the candidate’s safety, purity, potency and efficacy for its proposed indication or indications. Data may come from company-sponsored clinical trials or from a number of alternative sources, including studies initiated by investigators. To support marketing approval, the data submitted must be sufficient in quality and quantity to establish the safety and efficacy of the investigational product to the satisfaction of the FDA. The testing and approval processes require substantial time and effort and there can be no assurance that the FDA will accept the NDA or BLA for filing and, even if filed, that any approval will be granted on a timely basis, if at all.
Under the Prescription Drug User Fee Act, as amended, or PDUFA, each NDA or BLA must be accompanied by a significant user fee, and the sponsor of an approved application is also subject to an annual program fee. The FDA adjusts the PDUFA user fees on an annual basis. Fee waivers or reductions are available in certain circumstances, including a waiver of the application fee for the first application filed by a small business. Additionally, no user fees are assessed for products designated as orphan drugs, unless the product also includes a non-orphan indication.
Within 60 days following submission of the application, the FDA reviews it to determine if it is substantially complete before the agency accepts it for filing. The FDA may refuse to file any application that it deems incomplete or not properly reviewable at the time of submission and may request additional information. In this event, the application must be resubmitted with the additional information. The resubmitted application also is subject to review before the FDA accepts it for filing.
Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews BLA to determine, among other things, whether the proposed product is safe, pure, and potent for its intended use, and whether the facility (or facilities) in which it is manufactured, processed, packed, or held meets standards designed to assure the product’s continued safety, purity and potency. The FDA reviews an NDA to determine, among other things, whether a product is safe and effective for its intended use and whether its manufacturing is current good manufacturing practice, or cGMP-compliant to assure and preserve the product's identity, strength, quality and purity. Most such applications are meant to be reviewed within ten months from the date they are accepted for filing, and most applications for “priority review” products are meant to be reviewed within six months from the date the application is accepted for filing. The review process may be extended by the FDA for three additional months to consider new information or in the case of a clarification provided by the applicant deemed a major amendment to the application. The FDA likely will re-analyze the clinical trial data, which could result in extensive discussions between the FDA and the applicant during the review process.
The FDA may refer applications for novel products or products that present difficult questions of safety or efficacy to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making final decisions on approval.
Before approving an NDA or BLA, the FDA will typically inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities comply with cGMP requirements and are adequate to assure consistent production of the product within required specifications. Additionally, the FDA will typically inspect one or more clinical trial sites to assure that the clinical trials were conducted in compliance with good clinical practices, or GCP. To assure cGMP and GCP compliance, an applicant must incur significant expenditure of time, money and effort in the areas of training, record keeping, production, and quality control.
After the FDA evaluates an NDA or BLA and conducts inspections of manufacturing facilities where the investigational product and/or its drug substance will be produced, the FDA may issue an approval letter or a Complete Response Letter, or CRL. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. A CRL indicates that the review cycle of the application is complete, and the application will not be approved in its present form. A CRL generally outlines the deficiencies that the FDA identified in the application, except that where the FDA determines that the data supporting the application are inadequate to support approval, the FDA may issue the CRL without first conducting required inspections, testing submitted product lots, and/or reviewing proposed labeling. In issuing the CRL, the FDA may recommend actions that the applicant might take to place the application in condition for approval, including requests for additional clinical or other data, additional clinical trial(s) and/or other significant and time-consuming requirements related to clinical trials, preclinical studies or manufacturing. If a CRL is issued, the applicant may choose to either resubmit the NDA or BLA addressing all of the deficiencies identified in the letter or withdraw the application. If and when those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA or BLA, the FDA will issue an approval letter. The FDA has committed to reviewing such resubmissions in response to an issued CRL in either two or six months depending on the type of information included. Even with the submission of this additional information, however, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
If the FDA grants regulatory approval of a product, such approval is limited to the conditions of use (e.g., patient population, indication) described in the application and may entail limitations on the indicated uses for which such product may be marketed. For example, the FDA may approve the product with a risk evaluation and mitigation strategy, or REMS, to ensure the benefits of the product outweigh its risks and to assure the safe use of the drug or biological product. A REMS is a safety strategy to manage a known or potential serious risk associated with a product and to enable patients to have continued access to such medicines by managing their safe use, and could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. The FDA determines the requirement for a REMS, as well as the specific REMS provisions, on a case-by-case basis. If the FDA concludes a REMS is needed, the sponsor of the NDA or BLA must submit a proposed REMS. The FDA will not approve the application without a REMS, if required. The FDA also may condition approval on, among other things, changes to proposed labeling (e.g., the addition of specific contraindications, warnings or precautions) or the development of adequate controls and specifications. Once approved, the FDA may withdraw the product approval if compliance with pre- and post-marketing requirements is not maintained or if problems occur after the product reaches the marketplace. The FDA may require one or more Phase 4 post-market trials and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization and may limit further marketing of the product based on the results of these post-marketing studies. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are subject to further testing requirements and FDA review and approval.
Fast Track, Breakthrough Therapy and Priority Review Designations
The FDA is authorized to designate certain products for expedited development or review if they are intended to address an unmet medical need in the treatment of a serious or life-threatening disease or condition. These programs include fast track designation, breakthrough therapy designation and priority review designation.
To be eligible for a fast track designation, the FDA must determine, based on the request of a sponsor, that a product is intended to treat a serious or life-threatening disease or condition and demonstrates the potential to address an unmet medical need for such diseases or condition. Fast track designation provides opportunities for more frequent interactions with the FDA review team to expedite development and review of the product. The FDA may also review sections of the NDA or BLA for a fast track product on a rolling basis before the complete application is submitted, if the sponsor and the FDA agree on a schedule for the submission of the application sections and the sponsor pays any required user fees upon submission of the first section of the application. In addition, fast track designation may be withdrawn by the sponsor or rescinded by the FDA if the designation is no longer supported by data emerging from the clinical trial process.
In addition, the FDA may designate a drug or biologic as a “breakthrough therapy” upon a request made by the IND sponsor. A breakthrough therapy is a drug or biologic that is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug or biologic may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The FDA must take certain actions with respect to breakthrough therapies, such as holding timely meetings with and providing advice to the product sponsor, which are intended to expedite the development and review of an application for approval of a breakthrough therapy.
Finally, the FDA may designate an application for priority review if it is for a drug or biologic that treats a serious condition and, if approved, would provide a significant improvement in safety or effectiveness over existing therapy. The FDA determines at the time that the marketing application is submitted, on a case- by-case basis, whether the proposed drug represents a significant improvement in treatment, prevention or diagnosis of disease when compared with other available therapies. Significant improvement may be illustrated by evidence of increased effectiveness in the treatment of a condition, elimination or substantial reduction of a treatment-limiting drug reaction, documented enhancement of patient compliance that may lead to improvement in serious outcomes, or evidence of safety and effectiveness in a new subpopulation. A priority review designation is intended to direct overall attention and resources to the evaluation of such applications, and to shorten the FDA’s goal for taking action on an original marketing application from ten months to six months from the date of filing.
Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened. Furthermore, fast track designation, breakthrough therapy designation and priority review do not change the standards for approval and may not ultimately expedite the development or approval process.
Accelerated Approval Pathway
In addition, products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval from the FDA and may be approved on the basis of adequate and well-controlled clinical trials establishing that the drug product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit. The FDA may also grant accelerated approval for such a drug or biologic when the product has an effect on an intermediate clinical endpoint that can be measured earlier than an effect on irreversible morbidity or mortality, or IMM, and that is reasonably likely to predict an effect on IMM or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA will require that a sponsor of a drug receiving accelerated approval perform post-marketing clinical trials to verify and describe the predicted effect on IMM or other clinical endpoint, and the product may be subject to expedited withdrawal procedures. Drugs and biologics granted accelerated approval must meet the same statutory standards for safety and effectiveness as those granted traditional approval.
For the purposes of accelerated approval, a surrogate endpoint is a marker, such as a laboratory measurement, radiographic image, physical sign, or other measure that is thought to predict clinical benefit but is not itself a measure of clinical benefit. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. An intermediate clinical endpoint is a measurement of a therapeutic effect that is considered reasonably likely to predict the clinical benefit of a drug or biologic, such as an effect on IMM.
The accelerated approval pathway is usually contingent on a sponsor’s agreement to conduct, in a diligent manner, additional post-approval confirmatory studies to verify and describe the drug’s clinical benefit. As a result, a program or development candidate approved on this basis is typically subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post-approval clinical trials to establish the effect on the clinical endpoint. Failure to conduct required post-approval studies, or to confirm the predicted clinical benefit of the product during post-marketing studies, may allow the FDA to withdraw approval of the drug. The FDA may require the sponsor of a product granted accelerated approval to have a confirmatory trial underway prior to approval. The sponsor must also submit progress reports on a confirmatory trial every six months until the trial is complete, and such reports are published on FDA’s website.
All promotional materials for products approved under the accelerated approval program are subject to prior review by the FDA.
Pediatric Trials
Under the Pediatric Research Equity Act, or PREA, an NDA or BLA or supplement thereto must contain data to assess the safety and efficacy of the product for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may grant deferrals for submission of such data or full or partial waivers. The FDCA requires that a sponsor who is planning to submit a marketing application for a drug or biologic product that includes a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration submit an initial Pediatric Study Plan, or PSP, within sixty days of an end-of-Phase 2 meeting or as may be agreed between the sponsor and FDA, if there is no such meeting, as early as practicable before the initiation of Phase 3 or Phase 2/3 clinical trials. The initial PSP must include an outline of the pediatric study or studies that the sponsor plans to conduct, including study objectives and design, age groups, relevant endpoints and statistical approach, or a justification for not including such detailed information, and any request for a deferral of pediatric assessments or a full or partial waiver of the requirement to provide data from pediatric studies along with supporting information. The FDA and the sponsor must reach agreement on the PSP. A sponsor can submit amendments to an agreed-upon initial PSP at any time if changes to the pediatric plan need to be considered based on data collected from nonclinical studies, early phase clinical trials, and/or other clinical development programs. Unless otherwise required by regulation, the PREA does not apply to any product for an indication for which orphan designation has been granted. However, if only one indication for a product has orphan designation, a pediatric assessment may still be required for any applications to market that same product for the non-orphan indication(s).
Orphan Drug Designation and Exclusivity
Under the Orphan Drug Act, the FDA may grant orphan drug designation to a drug or biologic product intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the United States, or more than 200,000 individuals in the United States and for which there is no reasonable expectation that the cost of developing and making available in the United States a drug or biologic for this type of disease or condition will be recovered from sales in the United States for that drug or biologic. Orphan drug designation must be requested before submitting an NDA or BLA. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use will be disclosed publicly by the FDA; the posting will also indicate whether the drug or biologic is no longer designated as an orphan drug. More than one program or development candidate may receive an orphan drug designation for the same indication. Orphan drug designation does not convey any advantage in or shorten the duration of the regulatory review and approval process.
If a product that has orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to seven years of orphan product exclusivity. During the seven-year exclusivity period, the FDA may not approve any other applications to market a product containing the same active moiety for the same disease, except in very limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity. A product is clinically superior if it is safer, more effective or makes a major contribution to patient care. Thus, orphan drug exclusivity could block the approval of one of our potential products for seven years if a competitor obtains approval of the same product as defined by the FDA and we are not able to show the clinical superiority of our program or development candidate or if our program or development candidate’s indication is determined to be contained within the competitor’s product orphan indication. In addition, the FDA will not recognize orphan drug exclusivity if a sponsor fails to demonstrate upon approval that the product is clinically superior to a previously approved product containing the same active moiety for the same orphan condition, regardless of whether or not the previously approved product was designated an orphan drug or had orphan drug exclusivity. A product that has received orphan drug designation may not receive orphan exclusivity if it is approved for a use that is broader than the indication for which it received the designation. Orphan exclusivity does not prevent the FDA from approving a different drug or biological product for the same disease or condition, or the same product for a different disease or condition.
Post-Approval Requirements
Any products that we may manufacture or distribute pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, monitoring and record-keeping requirements, reporting of adverse experiences with the product, periodic reporting requirements, providing the FDA with updated safety and efficacy information, product sampling and distribution requirements, as well as advertising and promotion requirements, which include, among others, standards for direct-to-consumer advertising, restrictions on promoting products for uses or in patient populations that are not described in the product’s approved uses (known as off-label use), limitations on industry-sponsored scientific and educational activities, and requirements for promotional activities involving the Internet. After approval, most changes to the approved product, such as adding new indications or other labeling claims, are subject to prior FDA review and approval of a new application or supplement, which may require the applicant to develop additional data or conduct additional pre-clinical studies and clinical trials. The FDA may also place other conditions on approvals, including the requirement for a REMS, to assure the safe use of the product. A REMS could include medication guides, physician communication plans or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution, prescription or dispensing of products.
In addition, quality control and manufacturing procedures must continue to conform to applicable manufacturing requirements after approval to ensure the quality and long-term stability of the product. The cGMP regulations include requirements relating to organization of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, packaging and labeling controls, holding and distribution, laboratory controls, records and reports and returned or salvaged products. The manufacturing facilities for our programs and development candidates must meet cGMP requirements and satisfy the FDA or comparable foreign regulatory authorities before any product is approved and our commercial products can be manufactured. Third-party manufacturers must comply with cGMP regulations that require, among other things, quality control and quality assurance, the maintenance of records and documentation and the obligation to investigate and correct any deviations from cGMP. Manufacturers, including third-party manufacturers, and other entities involved in the manufacture and distribution of approved biologics are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP and other laws. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance. Future inspections by the FDA and other regulatory agencies may identify compliance issues at the facilities of our contract manufacturing organizations, or CMOs, that may disrupt production or distribution or require substantial resources to correct. In addition, the discovery of conditions that violate these rules, including failure to conform to cGMP regulations, could result in enforcement actions, and the discovery of problems with a product after approval may result in restrictions on a product, manufacturer, or sponsor of an approved NDA or BLA, including, among other things, voluntary recall and regulatory sanctions as described below.
The FDA may withdraw approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, could result in adverse consequences to the Company. Examples of these consequences include, without limitation, the following: may result in revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical studies to assess new safety risks; or imposition of distribution restrictions or other restrictions under a REMS program; complete withdrawal of the product from the market or other limits on marketing or manufacture of the product; imposition of civil or criminal penalties.
The FDA closely regulates the marketing, labeling, advertising and promotion of biopharmaceutical products. A company can make only those claims relating to safety and efficacy, purity and potency that are approved by the FDA and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off label uses. Failure to comply with these requirements can result in, among other things, adverse publicity, warning letters, corrective advertising and potential civil and criminal penalties. Physicians may prescribe legally available products for uses that are not described in the product’s labeling and that differ from those tested by us and approved by the FDA. The FDA does not regulate the behavior of physicians in their choice of treatments. The FDA does, however, restrict manufacturer’s communications on the subject of off-label use of their products.
Pediatric exclusivity
Pediatric exclusivity is a type of non-patent marketing exclusivity available in the United States and, if granted, it provides for the attachment of an additional six months of marketing protection to the term of any existing regulatory exclusivity or listed patents. This six-month exclusivity may be granted if a sponsor submits pediatric data that fairly responds to a Written Request from the FDA for such data. The data do not need to show the product to be effective in the pediatric population studied; rather, if the clinical trial is deemed to fairly respond to the FDA’s request, the additional protection is granted. If reports of requested pediatric studies are submitted to and accepted by the FDA within the statutory time limits, whatever statutory or regulatory periods of exclusivity or patent protection cover the product are extended by six months. The issuance of a Written Request does not require the sponsor to undertake the described studies.
Biosimilars and Reference Product Exclusivity
The Biologics Price Competition and Innovation Act of 2009, or BPCIA, created an abbreviated approval pathway for biological products that are biosimilar to or interchangeable with an FDA-licensed reference biological product. Biosimilarity, which requires that there be no clinically meaningful differences between the biological product and the reference product in terms of safety, purity, and potency, can be shown through analytical studies, animal studies, and a clinical study or studies. Interchangeability requires that a product is biosimilar to the reference product and the product must demonstrate that it can be expected to produce the same clinical results as the reference product in any given patient and, for products that are administered multiple times to an individual, the biologic and the reference biologic may be alternated or switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biologic without such alteration or switch. Upon licensure by the FDA, an interchangeable biosimilar may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product.
The biosimilar applicant must demonstrate that the product is biosimilar based on data from (1) analytical studies showing that the biosimilar product is highly similar to the reference product; (2) animal studies (including toxicity); and (3) one or more clinical studies to demonstrate safety, purity and potency in one or more appropriate conditions of use for which the reference product is approved. In addition, the applicant must show that the biosimilar and reference products have the same mechanism of action for the conditions of use on the label, route of administration, dosage and strength, and the production facility must meet standards designed to assure product safety, purity and potency.
A reference biological product is granted 12 years of data exclusivity from the time of first licensure of the product, and the first approved interchangeable biologic product will be granted an exclusivity period of up to one year after it is first commercially marketed. In addition, the FDA will not accept an application for a biosimilar or interchangeable product based on the reference biological product until four years after the date of first licensure of the reference product.
Hatch-Waxman exclusivity
Market exclusivity provisions under the FDCA can delay the submission or the approval of certain marketing applications. The FDCA provides a five-year period of non-patent data exclusivity within the United States to the first applicant to obtain approval of an NDA for a new chemical entity. A drug is a new chemical entity if the FDA has not previously approved any other new drug containing the same active moiety, which is the molecule or ion responsible for the action of the drug substance. During the exclusivity period, the FDA may not accept for review an abbreviated new drug application, or ANDA, or an NDA submitted under Section 505(b)(2) (505(b)(2) NDA) submitted by another company for another drug based on the same active moiety, regardless of whether the drug is intended for the same indication as the original innovative drug or for another indication, where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement to one of the patents listed with the FDA by the innovator NDA holder.
The FDCA alternatively provides three years of non-patent exclusivity for an NDA, or supplement to an existing NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example new indications, dosages or strengths of an existing drug. This three-year exclusivity covers only the modification for which the drug received approval on the basis of the new clinical investigations and does not prohibit the FDA from approving ANDAs or 505(b)(2) NDAs for drugs containing the active agent for the original indication or condition of use. Five-year and three-year exclusivity will not delay the submission or approval of a full NDA. However, an applicant submitting a full NDA would be required to conduct, or obtain a right of reference to, all of the preclinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
FDA Regulation of Companion Diagnostics
We believe that certain of our product candidates may require an in vitro diagnostic to identify appropriate patient populations for investigation and/or use of our product candidates. These diagnostics, often referred to as companion diagnostics, are regulated as medical devices. In the United States, the FDCA and its implementing regulations, and other federal and state statutes and regulations govern, among other things, medical device design and development, preclinical and clinical testing, premarket clearance or approval, registration and listing, manufacturing, labeling, storage, advertising and promotion, sales and distribution, export and import, and post-market surveillance. Unless an exemption applies, diagnostic tests require marketing clearance or approval from the FDA prior to commercial distribution. The two primary types of FDA marketing authorization applicable to a medical device are premarket notification, also called 510(k) clearance, and premarket approval, or PMA. Most companion diagnostics for oncology product candidates utilize the PMA pathway.
If use of companion diagnostic is deemed essential to the safe and effective use of a drug product, then the FDA generally will require approval or clearance of the diagnostic contemporaneously with the approval of the therapeutic product. On August 6, 2014, the FDA issued a final guidance document addressing the development and approval process for “In Vitro Companion Diagnostic Devices.” According to the guidance, for novel product candidates, a companion diagnostic device and its corresponding drug candidate should be approved or cleared contemporaneously by FDA for the use indicated in the therapeutic product labeling. The guidance also explains that a companion diagnostic device used to make treatment decisions in clinical trials of a drug generally will be considered an investigational device, unless it is employed for an intended use for which the device is already approved or cleared. If used to make critical treatment decisions, such as patient selection, the diagnostic device may be considered a significant risk device under the FDA's Investigational Device Exemption, or IDE, regulations. In which case, the sponsor of the diagnostic device will be required to submit and obtain approval of an IDE application, and subsequently comply with the IDE regulations. However, according to the guidance, if a diagnostic device and a drug are to be studied together to support their respective approvals, both products can be studied in the same investigational study, if the study meets both the requirements of applicable IDE regulations and the IND regulations. The guidance provides that, depending on the details of the study plan and degree of risk posed to subjects, a sponsor may seek to submit an IND alone, or both an IND and an IDE.
The FDA has generally required companion diagnostics intended to select the patients who will respond to cancer treatment to obtain approval of a PMA for that diagnostic simultaneously with approval of the therapeutic. The PMA process, including the gathering of clinical and preclinical data and the submission to and review by the FDA, can take several years or longer. It involves a rigorous premarket review during which the applicant must prepare and provide the FDA with reasonable assurance of the device's safety and effectiveness and information about the device and its components regarding, among other things, device design, manufacturing and labeling. In addition, PMAs for certain devices must generally include the results from extensive preclinical and adequate and well-controlled clinical trials to establish the safety and effectiveness of the device for each indication for which FDA approval is sought. In particular, for a diagnostic, the applicant must demonstrate that the diagnostic produces reproducible results when the same sample is tested multiple times by multiple users at multiple laboratories. As part of the PMA review, the FDA will typically inspect the manufacturer's facilities for compliance with the Quality System Regulation, or QSR, which imposes elaborate testing, control, documentation and other quality assurance requirements.
If the FDA's evaluation of the PMA application is favorable, the FDA may issue an approvable letter requiring the applicant's agreement to specific conditions, such as changes in labeling, or specific additional information, such as submission of final labeling, in order to secure final approval of the PMA. If the FDA's evaluation of the PMA or manufacturing facilities is not favorable, the FDA will deny approval of the PMA or issue a not approvable letter. A not approvable letter will outline the deficiencies in the application and, where practical, will identify what is necessary to make the PMA approvable. The FDA may also determine that additional clinical trials are necessary, in which case the PMA approval may be delayed for several months or years while the trials are conducted and then the data submitted in an amendment to the PMA. If and when the FDA concludes that the applicable criteria have been met, the FDA will issue a PMA for the approved indications, which can be more limited than those originally sought by the applicant. The PMA can include post-approval conditions that the FDA believes necessary to ensure the safety and effectiveness of the device, including, among other things, restrictions on labeling, promotion, sale and distribution. Once granted, PMA approval may be withdrawn by the FDA if compliance with post approval requirements, conditions of approval or other regulatory standards are not maintained or problems are identified following initial marketing.
After a device is commercialized, it remains subject to significant regulatory requirements. Medical devices may be marketed only for the uses and indications for which they are cleared or approved. Device manufacturers must also establish registration and device listings with the FDA. A medical device manufacturer's manufacturing processes and those of its suppliers are required to comply with the applicable portions of the QSR, which cover the methods and documentation of the design, testing, production, processes, controls, quality assurance, labeling, packaging and shipping of medical devices. Domestic facility records and manufacturing processes are subject to periodic unscheduled inspections by the FDA. The FDA also may inspect foreign facilities that export products to the United States.
Other U.S. Health Care Laws and Compliance Requirements
Although we currently do not have any products on the market, our business operations and current and future arrangements with investigators, health care professionals, consultants, third-party payors and customers may be subject to regulation and enforcement by various federal, state and local authorities in addition to the FDA, including but not limited to, the Centers for Medicare and Medicaid Services, or CMS, other divisions of the U.S. Department of Health and Human Services, or HHS, (such as the Office of Inspector General and the Health Resources and Service Administration), the Department of Justice, or the DOJ, and individual U.S. Attorney offices within the DOJ, and state and local governments. For example, sales, marketing and scientific/educational grant programs may have to comply with the anti-fraud and abuse provisions of the Social Security Act, the false claims laws, the privacy and security provisions of the Health Insurance Portability and Accountability Act, or HIPAA, and similar state laws, each as amended, as applicable.
The federal Anti-Kickback Statute prohibits, among other things, any person or entity, from knowingly and willfully offering, paying, soliciting or receiving any remuneration, directly or indirectly, overtly or covertly, in cash or in kind, to induce or in return for purchasing, leasing, ordering or arranging for the purchase, lease or order of any item or service reimbursable, in whole or in part, under Medicare, Medicaid or other federal health care programs. The term remuneration has been interpreted broadly to include anything of value. Further, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. The federal Anti-Kickback Statute has been interpreted to apply to arrangements between therapeutic product manufacturers on one hand and prescribers and purchasers on the other. There are a number of statutory exceptions and regulatory safe harbors protecting some common activities from prosecution. The exceptions and safe harbors are drawn narrowly and practices that involve remuneration that may be alleged to be intended to induce prescribing, purchasing or recommending may be subject to scrutiny if they do not qualify for an exception or safe harbor. Further, courts have found that if “one purpose” of remuneration is to induce referrals, the federal Anti-Kickback statute is violated. Failure to meet all of the requirements of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the federal Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review of all of its facts and circumstances. Our practices, including our arrangements with physicians, may not in all cases meet all of the criteria for protection under a statutory exception or regulatory safe harbor.
The federal false claims and civil monetary penalty laws, including the False Claims Act, or FCA, which can be enforced by private citizens through civil qui tam actions, prohibit any person or entity from, among other things, knowingly presenting, or causing to be presented, a false or fraudulent claim for payment to, or approval by, the federal health care programs, including Medicare and Medicaid, or knowingly making, using, or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government. A claim includes “any request or demand” for money or property presented to the U.S. government. For instance, historically, pharmaceutical and other health care companies have been, and continue to be, prosecuted under these laws for allegedly providing free product to customers with the expectation that the customers would bill federal programs for the product. A violation of the Anti-Kickback Statute makes any claim submitted as a result of the violation of the Anti-Kickback Statute a false claim under the FCA. Other companies have been prosecuted for causing false claims to be submitted because of the companies’ marketing of the product for unapproved, off-label, and thus generally non-reimbursable, uses.
HIPAA created additional federal criminal statutes that prohibit, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud or to obtain, by means of false or fraudulent pretenses, representations or promises, any money or property owned by, or under the control or custody of, any health care benefit program, including private third-party payors, willfully obstructing a criminal investigation of a health care offense, and knowingly and willfully falsifying, concealing or covering up by trick, scheme or device, a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services. Like the federal Anti-Kickback Statute, under HIPAA, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
Also, many states have similar, and typically more prohibitive, fraud and abuse statutes or regulations that apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Additionally, to the extent that our products are approved by and sold in a foreign country, we may be subject to similar foreign laws.
We may be subject to data privacy and security regulations by both the federal government and the states in which we conduct our business. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and their implementing regulations, impose requirements relating to the privacy, security and transmission of individually identifiable health information on certain health care providers, health care clearinghouses, and health plans, known as covered entities, as well as independent contractors, or agents of covered entities that create, receive or obtain individually identifiable health information in connection with providing a service on behalf of a covered entity, known as a business associates. Among other things, the passage of HITECH made HIPAA’s privacy and security standards directly applicable to business associates and their covered subcontractors. HITECH also created four new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce HIPAA and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, many state laws govern the privacy and security of health information in specified circumstances, many of which differ from each other in significant ways, are often not pre-empted by HIPAA, and may have a more prohibitive effect than HIPAA, thus complicating compliance efforts.
Additionally, the federal Physician Payments Sunshine Act, or the Sunshine Act, within the Patient Protection and Affordable Care Act, or ACA, and its implementing regulations, require that certain manufacturers of drugs, devices, biological and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program (with certain exceptions) report annually to CMS information related to certain payments or other transfers of value made or distributed to physicians, as broadly defined by such law, certain advanced non-physician health care practitioners, and teaching hospitals, or to entities or individuals at the request of, or designated on behalf of, such individuals or entities, and to report annually certain ownership and investment interests held by physicians and their immediate family members. In addition, many states also govern the reporting of payments or other transfers of value, many of which differ from each other in significant ways, are often not pre-empted, and may have a more prohibitive effect than the Sunshine Act, thus further complicating compliance efforts.
In order to distribute products commercially, we must comply with state laws that require the registration of manufacturers and wholesale distributors of drug and biological products in a state, including, in certain states, manufacturers and distributors who ship products into the state even if such manufacturers or distributors have no place of business within the state. Some states also impose requirements on manufacturers and distributors to establish the pedigree of product in the chain of distribution, including some states that require manufacturers and others to adopt new technology capable of tracking and tracing product as it moves through the distribution chain. Several states have enacted legislation requiring pharmaceutical and biotechnology companies to establish marketing compliance programs, file periodic reports with the state, make periodic public disclosures on sales, marketing, pricing, clinical trials and other activities, and/or register their sales representatives, as well as to prohibit pharmacies and other health care entities from providing certain physician prescribing data to pharmaceutical and biotechnology companies for use in sales and marketing, and to prohibit certain other sales and marketing practices. All of our activities are potentially subject to federal and state consumer protection and unfair competition laws.
Ensuring business arrangements with third parties comply with applicable health care laws and regulations is a costly endeavor. If our operations are found to be in violation of any of the federal and state health care laws described above or any other current or future governmental regulations that apply to us, we may be subject to penalties, including without limitation, significant civil, criminal and/or administrative penalties, damages, fines, disgorgement, individual imprisonment, exclusion from participation in government programs, such as Medicare and Medicaid, injunctions, private “qui tam” actions brought by individual whistleblowers in the name of the government, or refusal to allow us to enter into government contracts, contractual damages, reputational harm, administrative burdens, diminished profits and future earnings, additional reporting obligations and integrity oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws, and the curtailment or restructuring of our operations, any of which could adversely affect our ability to operate our business and our results of operations. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. The complex compliance environment and the need to build and maintain robust and expandable systems to comply with multiple jurisdictions with different compliance or reporting requirements increases the possibility that a health care company may run afoul of one or more of the requirements.
Coverage, Pricing and Reimbursement
Significant uncertainty exists as to the coverage and reimbursement status of any programs or development candidates for which we may obtain regulatory approval. In the United States and in foreign markets, sales of any products for which we receive regulatory approval for commercial sale will depend, in part, on the extent to which third-party payors provide coverage and establish adequate reimbursement levels for such products. In the United States, third-party payors include federal and state health care programs, private managed care providers, health insurers and other organizations. Coverage and adequate reimbursement from governmental health care programs, such as Medicare and Medicaid in the United States, and commercial payors are critical to new product acceptance.
Third-party payors decide which therapeutics they will pay for and establish reimbursement levels. In the United States, the principal decisions about reimbursement for new medicines are typically made by CMS. CMS decides whether and to what extent our products will be covered and reimbursed under Medicare and private payors tend to follow CMS to a substantial degree. Coverage and reimbursement by a third-party payor may depend upon a number of factors, including the third-party payor’s determination that use of a therapeutic is a covered benefit under its health plan, safe, effective and medically necessary, appropriate for the specific patient, cost-effective and neither experimental nor investigational.
We cannot be sure that reimbursement will be available for any product that we commercialize and, if coverage and reimbursement are available, we cannot be sure that the level of reimbursement will be adequate. Coverage may also be more limited than the purposes for which the product is approved by the FDA or comparable foreign regulatory authorities. Limited coverage and less than adequate reimbursement may reduce the demand for, or the price of, any product for which we obtain regulatory approval.
Third-party payors are increasingly challenging the price, examining the medical necessity, and reviewing the cost-effectiveness of medical products, therapies, and services, in addition to questioning their safety and efficacy. Obtaining reimbursement for our products may be particularly difficult because of the higher prices often associated with branded drugs and biologics, as well as drugs and biologics administered under the supervision of a physician. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of our products, in addition to the costs required to obtain FDA approvals. Our programs and development candidates may not be considered medically necessary or cost-effective. Obtaining coverage and reimbursement approval of a product from a third-party payor is a time-consuming and costly process that could require us to provide to each payor supporting scientific, clinical and cost-effectiveness data for the use of our product on a payor-by-payor basis, with no assurance that coverage and adequate reimbursement will be obtained. A third-party payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved. Additionally, in the United States there is no uniform policy among third-party payors for coverage or reimbursement. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting their own coverage and reimbursement policies, but also have their own methods and approval processes. Therefore, one third-party payor’s determination to provide coverage for a product does not ensure that other payors will also provide coverage for the product. Adequate third-party payor reimbursement may not be available to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development. If reimbursement is not available or is available only at limited levels, we may not be able to successfully commercialize any program or development candidate that we successfully develop.
Many pharmaceutical manufacturers must calculate and report certain price reporting metrics, such as average sales price and best price, to the government, such as average sales price and best price. These prices for drugs or biologics may be reduced by mandatory discounts or rebates required by government health care programs or private payors and by any future relaxation of laws that presently restrict imports of drugs or biologics from countries where drugs may be sold at lower prices than in the United States. Further, certain of our products, if approved, may be administered by a physician. Under currently applicable U.S. law, certain products that are not self-administered by the patient (including injectable drugs) may be eligible for coverage under Medicare through Medicare Part B. Medicare Part B is part of original Medicare, the federal health care program that provides health care benefits to the aged and disabled, and covers outpatient services and supplies, including certain drug and biological products, that are medically necessary to treat a beneficiary’s health condition. As a condition of receiving Medicare Part B reimbursement for a manufacturer’s eligible drugs or biologicals, the manufacturer is required to participate in other government health care programs, including the Medicaid Drug Rebate Program and the 340B Drug Pricing Program. The Medicaid Drug Rebate Program requires biopharmaceutical manufacturers to enter into and have in effect a national rebate agreement with the Secretary of HHS as a condition for states to receive federal matching funds for the manufacturer’s outpatient therapeutic products furnished to Medicaid patients. Under the 340B Drug Pricing Program, the manufacturer must extend discounts to entities that participate in the program.
Different pricing and reimbursement schemes exist in other countries. In the European Union, governments influence the price of pharmaceutical products through their pricing and reimbursement rules and control of national health care systems that fund a large part of the cost of those products to consumers. Some jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost effectiveness of a particular program or development candidate to currently available therapies. Other member states allow companies to fix their own prices for medicines but monitor and control company profits. The downward pressure on health care costs has become intense. As a result, increasingly high barriers are being erected to the entry of new products. In addition, in some countries, cross-border imports from low-priced markets exert a commercial pressure on pricing within a country.
The marketability of any programs or development candidates for which we receive regulatory approval for commercial sale may suffer if the government and third-party payors fail to provide coverage and adequate reimbursement. In addition, emphasis on managed care, the increasing influence of health maintenance organizations, and additional legislative changes in the United States has increased, and we expect will continue to increase, the pressure on health care pricing. The downward pressure on the rise in health care costs has become very intense. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
Health Care Reform
In the United States and some jurisdictions outside the United States, there have been, and continue to be, proposed legislative and regulatory changes to the current health care systems that could prevent or delay marketing approval of programs and development candidates, restrict or regulate post-approval activities, and affect the ability to profitably sell programs and development candidates for which marketing approval is obtained. The FDA’s and other regulatory authorities’ policies may change, and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product and therapeutic candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may lose any marketing approval that we otherwise may have obtained and we may not achieve or sustain profitability, which would adversely affect our business, prospects, financial condition and results of operations. Moreover, among policy makers and payors in the United States and elsewhere, there is significant interest in promoting changes in health care systems with the stated goals of containing health care costs, improving quality and/or expanding access.
For example, the ACA was enacted in March 2010 and has had a significant impact on the health care industry in the United States. The ACA expanded coverage for the uninsured while at the same time containing overall health care costs. With regard to biopharmaceutical products, the ACA, among other things, addressed a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted or injected, increased the minimum Medicaid rebates owed by manufacturers under the Medicaid Drug Rebate Program and extended the rebate program to individuals enrolled in Medicaid managed care organizations, established annual fees on manufacturers of certain branded prescription drugs, and created a new Medicare Part D coverage gap discount program. Additionally, the Creating and Restoring Equal Access to Equivalent Samples Act, or CREATES Act, was enacted on December 20, 2019 to address the concern articulated by both the FDA and others in the industry that some brand manufacturers have improperly restricted the distribution of their products, including by invoking the existence of a REMS for certain products, to deny generic product developers access to samples of brand products. Because generic product developers need samples to conduct certain comparative testing required by the FDA, some have attributed the inability to timely obtain samples as a cause of delay in the entry of generic products. To remedy this concern, the CREATES Act establishes a private cause of action that permits a generic product developer to sue the brand manufacturer to compel it to furnish the necessary samples on “commercially reasonable, market-based terms.” Whether and how generic product developments will use this new pathway, as well as the likely outcome of any legal challenges to provisions of the CREATES Act, remain highly uncertain and its potential effects on any of our future commercial products are unknown.
Following several years of litigation in the federal courts, in June 2021, the U.S. Supreme Court dismissed a challenge on procedural groups that argued the ACA is unconstitutional in its entirety because the individual mandate was repealed by Congress. In addition, on August 16, 2022, President Biden signed the Inflation Reduction Act of 2022, or the IRA, into law, which among other things, extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. The IRA also eliminates the “donut hole” under the Medicare Part D program beginning in 2025 by significantly lowering the beneficiary maximum out-of-pocket cost and creating a new manufacturer discount program. Further legislative and regulatory changes under the ACA remain possible, but it is unknown what form any such changes or any law would take, and how or whether it may affect the biopharmaceutical industry as a whole or our business in the future. We expect that changes or additions to the ACA, the Medicare and Medicaid programs, and changes stemming from other health care reform measures, especially with regard to health care access, financing or other legislation in individual states, could have a material adverse effect on the health care industry in the United States.
In addition, other legislative changes have been proposed and adopted in the United States since the ACA that affect health care expenditures. These changes include aggregate reductions to Medicare payments to providers pursuant to the Budget Control Act of 2011, which began in 2013 and, due to subsequent legislative amendments, will remain in effect through 2032, unless additional Congressional action is taken.
Moreover, there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several Congressional inquiries and proposed and enacted federal and state legislation designed to, among other things, bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs, and reform government program reimbursement methodologies for drug products.
In addition, the IRA has multiple provisions that may impact the prices of drug products that are both sold into the Medicare program and throughout the United States. Starting in 2023, a manufacturer of a drug or biological product covered by Medicare Parts B or D must pay a rebate to the federal government if the drug product’s price increases faster than the rate of inflation. This calculation is made on a drug product by drug product basis and the amount of the rebate owed to the federal government is directly dependent on the volume of a drug product that is paid for by Medicare Parts B or D. Additionally, starting in payment year 2026, CMS will negotiate drug prices annually for a select number of single source Part D drugs without generic or biosimilar competition. On August 29, 2023, the list of the first ten drugs that will be subject to price negotiations was published, although the Medicare drug price negotiation program is currently subject to legal challenges. CMS will also negotiate drug prices for a select number of Part B drugs starting for payment year 2028. If a drug product is selected by CMS for negotiation, it is expected that the revenue generated from such drug will decrease. The IRA permits HHS to implement many of these provisions through guidance, as opposed to regulation, for the initial years. HHS has and will continue to issue and update guidance as these programs as implemented. These provisions take effect progressively starting in fiscal year 2023. It is unclear how the IRA will be implemented but is likely to have a significant impact on the pharmaceutical industry. Further, on February 14, 2023, HHS released a report outlining three new models for testing by the Centers for Medicare & Medicaid Services Innovation Center which will be evaluated on their ability to lower the cost of drugs, promote accessibility, and improve quality of care. It is unclear whether the models will be utilized in any health reform measures in the future. Additionally, on December 7, 2023, the Biden administration announced an initiative to control the price of prescription drugs through the use of march-in rights under the Bayh-Dole Act. On December 8, 2023, the National Institute of Standards and Technology published for comment a Draft Interagency Guidance Framework for Considering the Exercise of March-In Rights which for the first time includes the price of a product as one factor an agency can use when deciding to exercise march-in rights. While march-in rights have not previously been exercised, it is uncertain if that will continue under the new framework.
Individual states in the United States have also increasingly passed legislation and implemented regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. For example, on January 5, 2024, the FDA approved Florida’s Section 804 Importation Program, or SIP, proposal to import certain drugs from Canada for specific state healthcare programs. It is unclear how this program will be implemented, including which drugs will be chosen, and whether it will be subject to legal challenges in the United States or Canada. Other states have also submitted SIP proposals that are pending review by the FDA. Any such approved importation plans, when implemented, may result in lower drug prices for products covered by those programs.
We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative or executive action, either in the United States or abroad. We expect that additional state and federal health care reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for health care products and services, including any future drug products for which we secure marketing approval.
Additional Regulation
In addition to the foregoing, state and federal laws regarding environmental protection and hazardous substances, including the Occupational Safety and Health Act, the Resource Conservancy and Recovery Act and the Toxic Substances Control Act, affect our business. These and other laws govern our use, handling and disposal of various biological, chemical and radioactive substances used in, and wastes generated by, our operations. If our operations result in contamination of the environment or expose individuals to hazardous substances, we could be liable for damages and governmental fines. We believe that we are in material compliance with applicable environmental laws and that continued compliance therewith will not have a material adverse effect on our business. We cannot predict, however, how changes in these laws may affect our future operations.
We are also subject to numerous federal, state and local laws relating to such matters as safe working conditions, manufacturing practices, environmental protection, fire hazard control, and disposal of hazardous or potentially hazardous substances. We may incur significant costs to comply with such laws and regulations now or in the future.
Employees and Human Capital Resources
As of December 31, 2023, we had 55 full-time employees, including 38 who hold advanced degrees. Of these 55 employees, 26 were engaged in research and development activities and 29 were engaged in general and administrative activities. None of our employees are represented by labor unions or covered by collective bargaining agreements. We consider our relationship with our employees to be good.
Our human capital resources objectives include, as applicable, identifying, recruiting, retaining a diverse pool of qualified talent, training, incentivizing, and integrating our existing new employees, advisors, and consultants. We offer a competitive total rewards package, updated in 2024 based on market research. We incentivize high performers through an annual bonus program based on our company and individual performance for which all employees are eligible. We also offer equity incentive plans, the purpose of which are to attract, retain and reward employees through the granting of share-based compensation awards in order to increase stockholder value and the success of our company by motivating team members to perform to the best of their abilities and achieve our objectives.
Facilities
We currently lease 11,113 square feet of office space and laboratory space in Exton, Pennsylvania and under a lease that expires on March 31, 2025 and 14,348 square feet of office and laboratory space in Bothell, Washington under a lease that expires on October 31, 2028, or the Bothell Lease. We believe the leased spaces are sufficient to meet our immediate facility needs, and that any additional space we may require will be available on commercially reasonable terms.