Exhibit 99.1

CORPORATE OVERVIEW Frank Bedu-Addo Ph.D. President & CEO JUNE 2021

2 Forward-Looking Statements This presentation contains forward-looking statements about PDS Biotechnology Corporation (“PDSB”), and its businesses, business prospects, strategies and plans, including but not limited to statements regarding anticipated pre-clinical and clinical drug development activities and timelines and market opportunities. All statements other than statements of historical facts included in this presentation are forward-looking statements. The words “anticipates,” “may,” “can,” “plans,” “believes,” “estimates,” “expects,” “projects,” “intends,” “likely,” “will,” “should,” “to be,” and any similar expressions or other words of similar meaning are intended to identify those assertions as forward-looking statements. These forward-looking statements involve substantial risks and uncertainties that could cause actual results to differ materially from those anticipated.Factors that may cause actual results to differ materially from such forward-looking statements include those identified under the caption “Risk Factors” in the documents filed with the Securities and Exchange Commission (“SEC”) from time to time, including its Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this presentation. Except to the extent required by applicable law or regulation, PDSB undertakes no obligation to update the forward-looking statements included in this presentation to reflect subsequent events or circumstances.

3 PDS Biotech’s Versamune®-based immunotherapies are designed to promote a powerful in vivo tumor-specific CD8+ killer T-cell response Generate the right type and quantity of effective CD8+ killer T-cells Generate potency without systemic side effects Generate memory T-cells, to enhance durability of response A significant barrier to effective immunotherapy has been the inability to promote adequate CD8+ killer T-cell responses in vivo resulting in diminished efficacy; 70-90% of cancer patients fail check point inhibitor therapy Versamune®-based therapies also show promising potential to:

PDS Biotech is a clinical stage biotechnology company developing a pipeline of immunotherapies based on the proprietary Versamune® platform 4 NCI-initiated phase 2 combination trial of PDS0101 in advanced HPV-cancer reported strong potential clinical benefitNo new or elevated toxicities observed from the addition of PDS0101 to combination therapyPre-clinical studies demonstrated potential to work with a wide array of oncogenes and viral antigensMultiple composition and application patents valid through mid-2030s Biopharma developing novel T-cell activating cancer treatment candidates Three phase 2 oncology clinical trials in progress anticipated to release data in 2021Clinical partnerships with Merck, MD Anderson Cancer Center and National Cancer Institute~15 employees with headquarters in Florham Park, NJDebt free with approximately $29.5M in cash* Pipeline Versamune® Platform Corporate Overview *The Company received $4.5M from the sale of NJ Net Operating Loss Tax Benefits in May 2021 which has been added to the cash balance reported on our Form 10-Q as of 3/31/2021.

PDS Biotech’s robust Versamune® -based pipeline is being developed in partnership with leaders in immuno-oncology and infectious disease 5 Reference: Data on file. * *Consortium of PDS Biotech, Farmacore Biotechnology and Blanver Farmoquimica. Funding provided by The Ministry of Science, Technology and Innovation of Brazil (“MCTI”)

PDS Biotech executive team has demonstrated success in the development and commercialization of leading pharmaceutical products 6 Senior executive experience with management of strategy and execution at both large pharma and biotechsNotable drug development:Abelcet® (Liposome Company/ Elan)PEG-Intron® (Schering-Plough/ Merck) Frank Bedu-Addo, PhDChief Executive Officer Co-founder>35 years of drug development experience In-depth experience with biotech drug discovery, product development and manufacturing Gregory Conn, PhDChief Scientific Officer >30 years of translational clinical research experienceFormer Director of Clinical Research at National Cancer Institute Center for Cancer Research (Cancer Vaccine Branch) Lauren V. Wood, MDChief Medical Officer Senior executive experience with over 20 years of experience in high tech companiesIn-depth experience with M&A transactions, capital markets, business development and investor relations Seth Van Voorhees, PhDChief Financial Officer

Introduction to the Versamune® Platform

Versamune® is designed to induce a robust and targeted anti-tumor response in vivo when administered with a tumor-associated antigen 8 References: Gandhapudi SK, et al. 2019. Antigen priming with enantiospecific cationic lipid nanoparticles induces potent antitumor CTL responses through novel induction of a Type I IFN response. J Immunol. 202 (12): 3524-3536. Smalley Rumfield C et al. 2020. Immunomodulation to enhance the efficacy of an HPV therapeutic vaccine. J. for ImmunoTherapy of Cancer 8:e000612. Promotes uptake of vaccine or immunotherapy and entry into lymph nodes Promotes antigen processing and presentation to T-cells via MHC I and II pathways Activates Type I Interferon pathway, enabling a powerful anti-tumor killer CD8+ T-cell response Versamune® + Tumor-associated proteins (antigens)

Greater quantity and quality of Versamune®-induced killer T-cells may result in unique ability to eradicate HPV-positive tumors after a single dose 9 Induced a >10-fold number of highly potent T-cells and eradication of HPV-positive tumors after a single dose in preclinical studies Single treatment dose Results typical of current topclinical-stage HPV cancer vaccines Tumor rechallenge at Day 60; complete and sustained cure of cancer *Adjuvant = cytokine GM-CSFReferences: J. Immunology, 2019 (202), 1215; Studies in TC-1 tumor model with other immunotherapies reported in: Vaccine 2009, January 14, 27 (3): 431; Science Translational Medicine 2016, 13 April, Vol 8 Issue 334; Vaccine 2009, September 25, 27 (42): 5906. (PDS0101)

PDS0101 Phase 2 Clinical Development

Combinations of PDS0101 with FDA-approved standard of careFirst line treatment of recurrent/metastatic HPV-positive head and neck cancerCombination with KEYTRUDA®Treatment of locally advanced cervical cancerCombination with chemoradiotherapy 11 Novel combinations of PDS0101 with promising, investigational immunotherapeutic agentsTreatment of advanced HPV-associated cancers (anal, cervical, vaginal, head and neck etc.)Triple combination with Bintrafusp alpha (bi-functional checkpoint inhibitor - M7824) and M9241 (NHS-IL12 an antibody conjugated immuno-cytokine) Clinical strategy: Develop PDS0101 in combination with established therapies for rapid proof-of-concept and risk mitigation

Phase 2 NCI-led clinical trial evaluating the triple combination of PDS0101, Bintrafusp alfa and M9241 in advanced HPV-associated cancer 12 Indication Patients with advanced HPV-associated cancer who have failed prior treatment Clinical Agents Bintrafusp alfa: Bifunctional “trap” fusion proteinM9241: Antibody-conjugated immuno-cytokinePDS0101: Versamune®-based immunotherapy generating HPV-specific CD8+ T-cells Study goals Group 1: Objective response rate (ORR) in checkpoint inhibitor (CPI) naïve patientsGroup 2: ORR in patients who have failed checkpoint inhibitor therapy (CPI refractory) Timing Full enrollment of 56 patientsComplete enrollment expected by Q4 2021/Q1 2022 Trial Sponsor The objective of this trial is to confirm that PDS0101 enhances the therapeutic benefit of Bintrafusp alfa and M9241 and may lead to expanded evaluation in several pipeline products
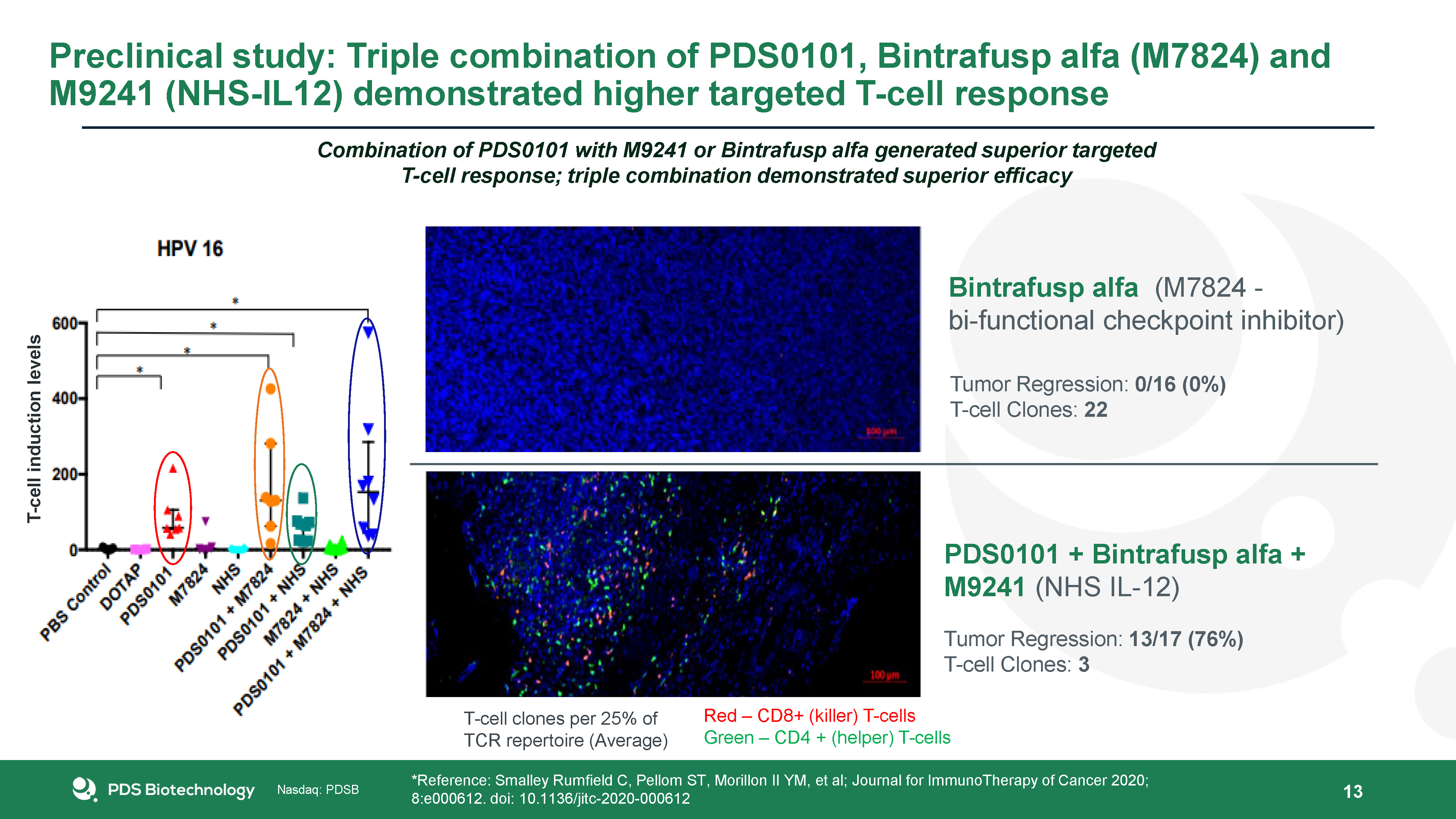
13 Bintrafusp alfa (M7824 - bi-functional checkpoint inhibitor) Tumor Regression: 0/16 (0%)T-cell Clones: 22 PDS0101 + Bintrafusp alfa + M9241 (NHS IL-12) Tumor Regression: 13/17 (76%)T-cell Clones: 3 *Reference: Smalley Rumfield C, Pellom ST, Morillon II YM, et al; Journal for ImmunoTherapy of Cancer 2020; 8:e000612. doi: 10.1136/jitc-2020-000612 Red – CD8+ (killer) T-cellsGreen – CD4 + (helper) T-cells T-cell clones per 25% of TCR repertoire (Average) Combination of PDS0101 with M9241 or Bintrafusp alfa generated superior targeted T-cell response; triple combination demonstrated superior efficacy T-cell induction levels Preclinical study: Triple combination of PDS0101, Bintrafusp alfa (M7824) and M9241 (NHS-IL12) demonstrated higher targeted T-cell response

PDS0101 Phase 1 clinical trial: Powerful CD8+ T-cell response resulted in regression of CIN cervical lesions & supported continued clinical studies 14 * When treated with selected human clinical trial dosage (1mg and 3mg Versamune®)References: L. Wood et al. A Novel Enantio-Specific Cationic Lipid R-DOTAP + HPV16 E6 & E7 Antigens Induces Potent Antigen-Specific CD8+ T Cell Responses In-Vivo in Subjects with CIN and High-Risk Human Papillomavirus Infection. Nov 8, 2019. SITC. Presentation O17. Most patients infected with multiple strains of HPV CIN lesion regression as early as 1-3 months 60% 20% 20% Phase 1 trial results showed no serious or dose-limiting toxicities of PDS0101 monotherapy Monotherapy distinguished from key limitation of immuno-oncology: > 20-fold increase in circulating dual INF-γ & Granzyme-b inducing killer T-cells vs. pre-treatment at day 14 led to rapid clearance of lesions*
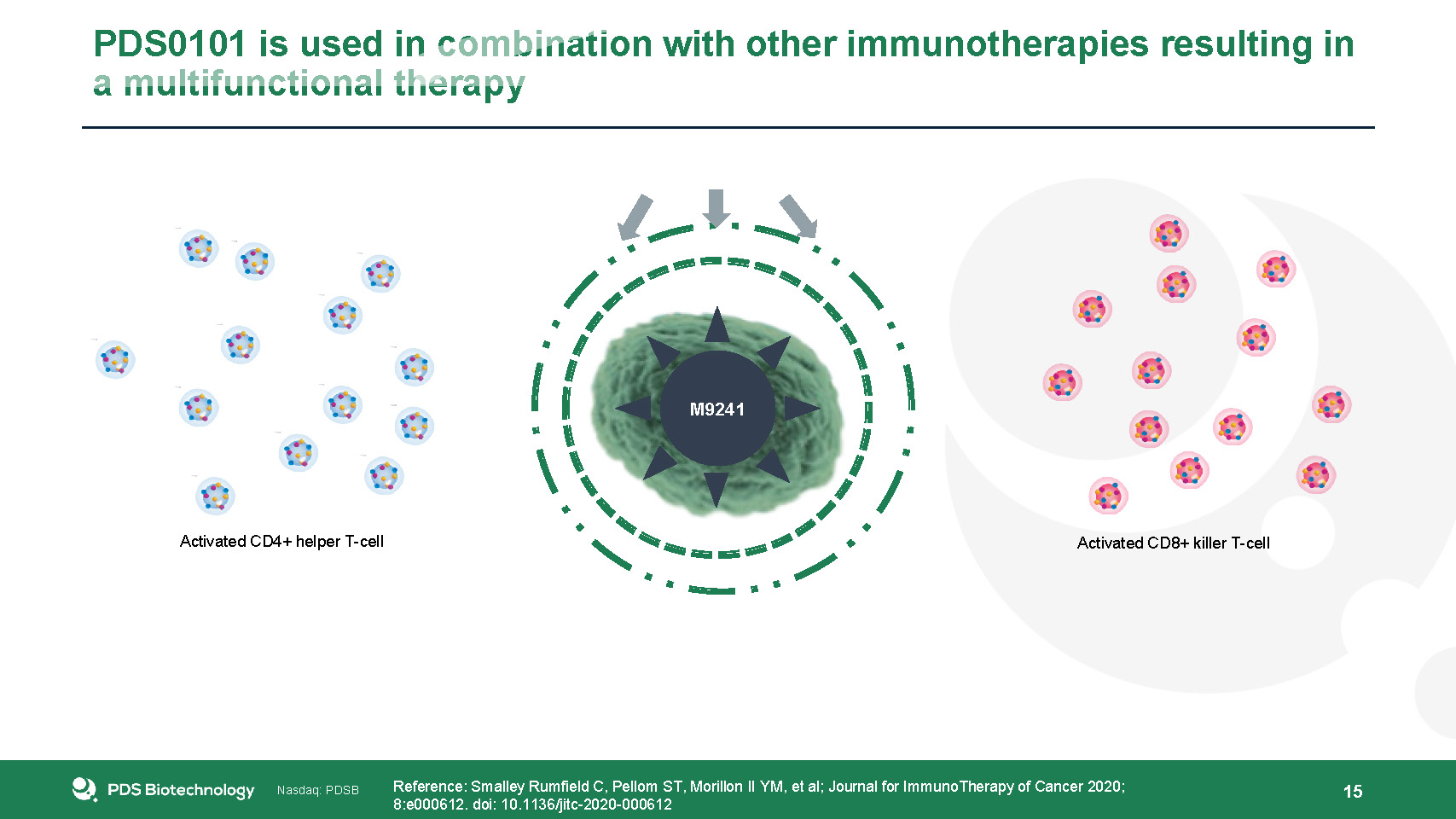
PDS0101 is used in combination with other immunotherapies resulting in a multifunctional therapy 15 Bintrafusp alfa exposes the tumor to attack and M9241 issues a signal calling T-cells to the tumor Reference: Smalley Rumfield C, Pellom ST, Morillon II YM, et al; Journal for ImmunoTherapy of Cancer 2020; 8:e000612. doi: 10.1136/jitc-2020-000612 Bintrafusp ALFA M9241 PDS0101 induces a powerful, HPV16-targeted CD8+ and CD4+ T-cell response PDS0101 Activated CD8+ killer T-cell Activated CD4+ helper T-cell The triple combination works in synergy to prompt targeted T-cells to infiltrate and destroy the tumor TUMOR DESTROYED

Objective response rate is measured by RECIST 1.1 and represents at least a 30% reduction in tumor sizeAdvanced HPV-related cancer that is checkpoint inhibitor naïve:Patients who fail chemotherapy and/or radiation progress to checkpoint inhibitor therapy12-24% ORR with standard of care checkpoint inhibitors30% ORR reported with experimental monotherapy Bintrafusp alfa is the highest reported to dateAdvanced HPV-related cancer that is checkpoint inhibitor refractory:Few treatment options exist for these patients5-12% ORR reported with checkpoint inhibitors PDS0101 phase 2 triple combination trial: Evaluated potential for superior preclinical tumor regression in advanced HPV-related cancer 16 A critical limitation of immunotherapy is the inability to induce large numbers of powerful tumor-attacking CD8+ (killer) T-cells within the body, that can result in tumor reduction or elimination in a significant number of advanced cancer patients Reference: Strauss J, et al. J Immunother Cancer. 2020 Dec;8(2):e001395
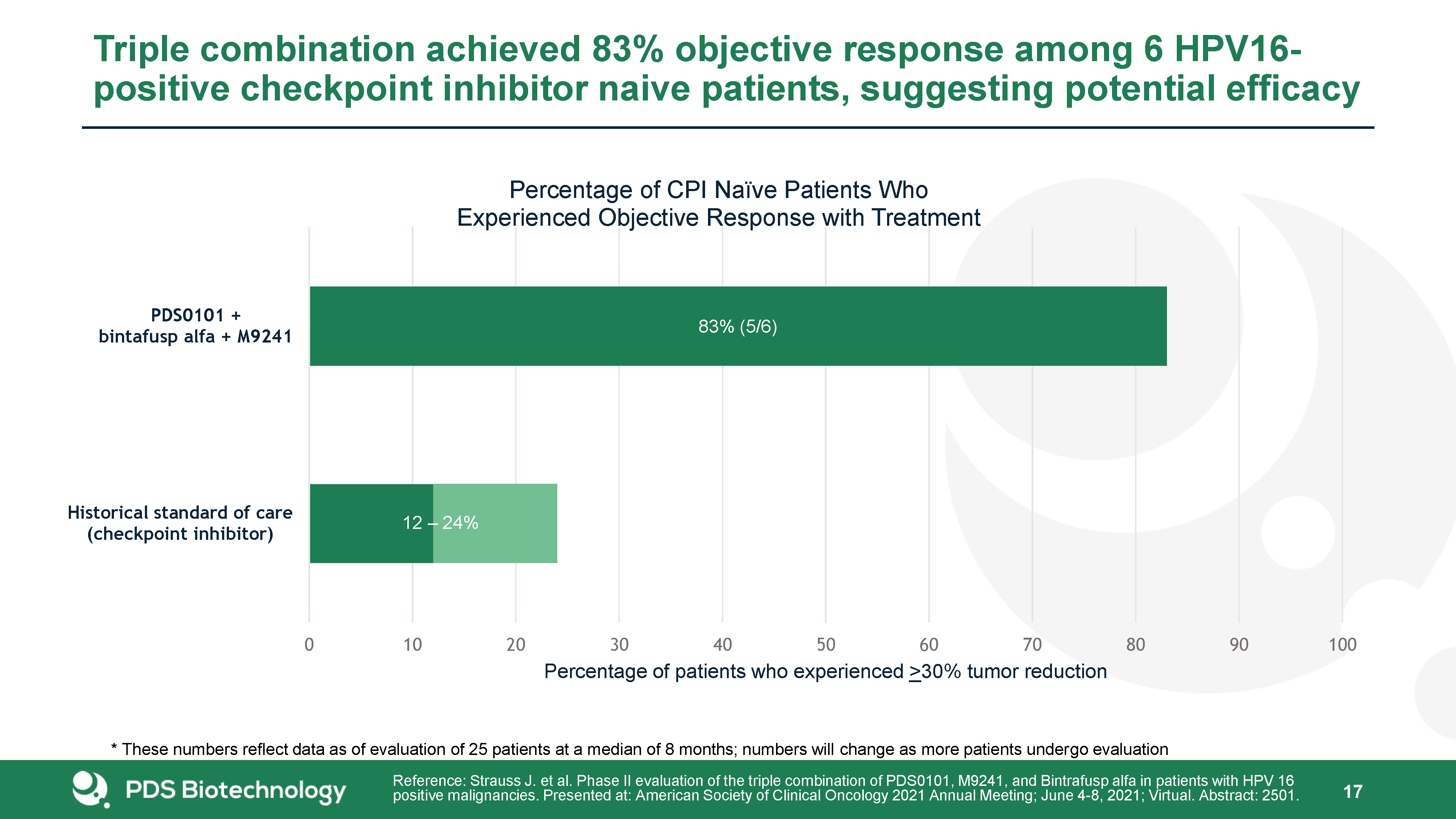
Triple combination achieved 83% objective response among 6 HPV16-positive checkpoint inhibitor naive patients, suggesting potential efficacy 17 Reference: Strauss J. et al. Phase II evaluation of the triple combination of PDS0101, M9241, and Bintrafusp alfa in patients with HPV 16 positive malignancies. Presented at: American Society of Clinical Oncology 2021 Annual Meeting; June 4-8, 2021; Virtual. Abstract: 2501. * These numbers reflect data as of evaluation of 25 patients at a median of 8 months; numbers will change as more patients undergo evaluation
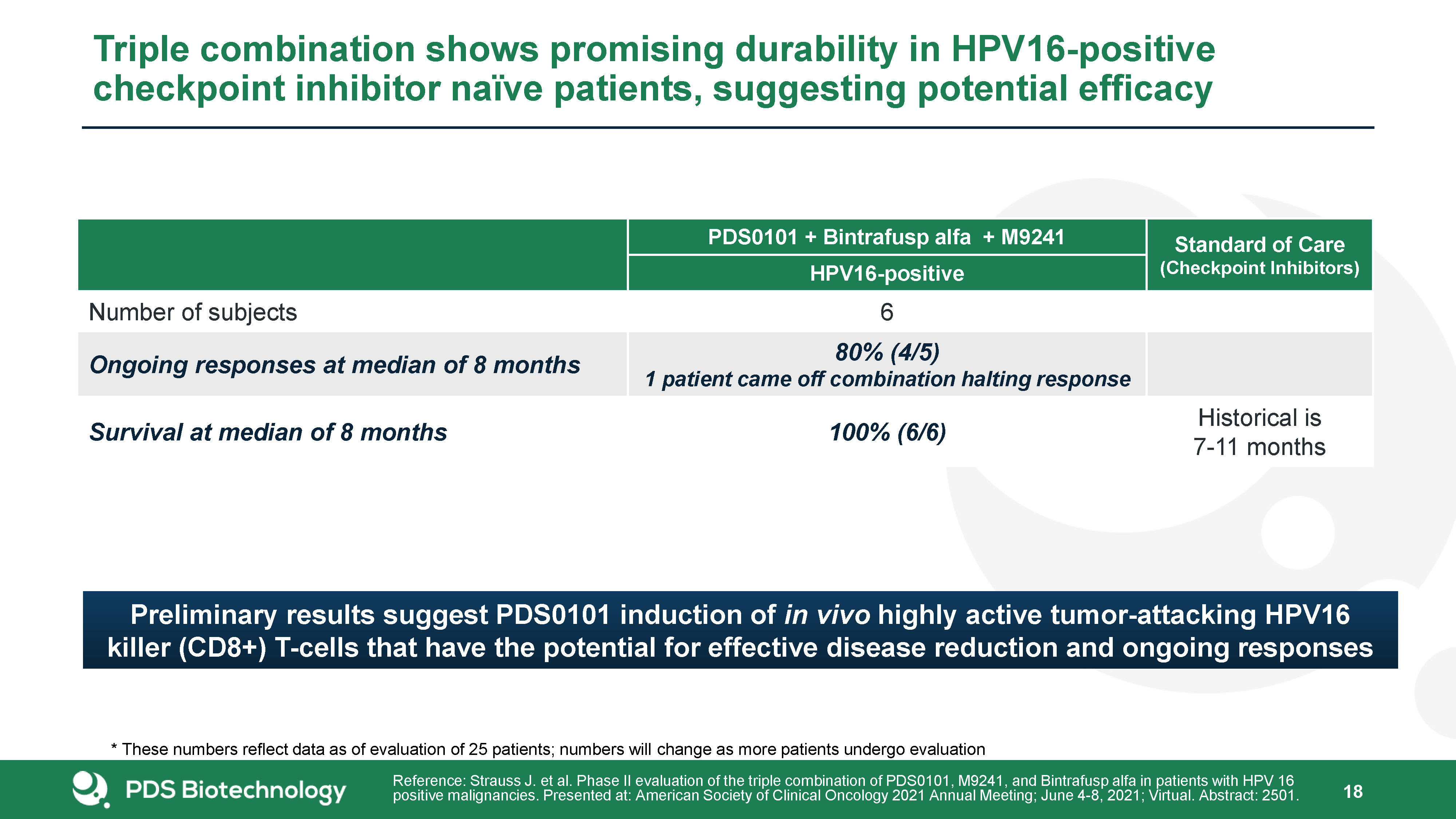
18 Triple combination shows promising durability in HPV16-positive checkpoint inhibitor naïve patients, suggesting potential efficacy PDS0101 + Bintrafusp alfa + M9241 Standard of Care(Checkpoint Inhibitors) HPV16-positive Number of subjects 6 Ongoing responses at median of 8 months 80% (4/5)1 patient came off combination halting response Survival at median of 8 months 100% (6/6) Historical is 7-11 months Reference: Strauss J. et al. Phase II evaluation of the triple combination of PDS0101, M9241, and Bintrafusp alfa in patients with HPV 16 positive malignancies. Presented at: American Society of Clinical Oncology 2021 Annual Meeting; June 4-8, 2021; Virtual. Abstract: 2501. Preliminary results suggest PDS0101 induction of in vivo highly active tumor-attacking HPV16 killer (CD8+) T-cells that have the potential for effective disease reduction and ongoing responses * These numbers reflect data as of evaluation of 25 patients; numbers will change as more patients undergo evaluation
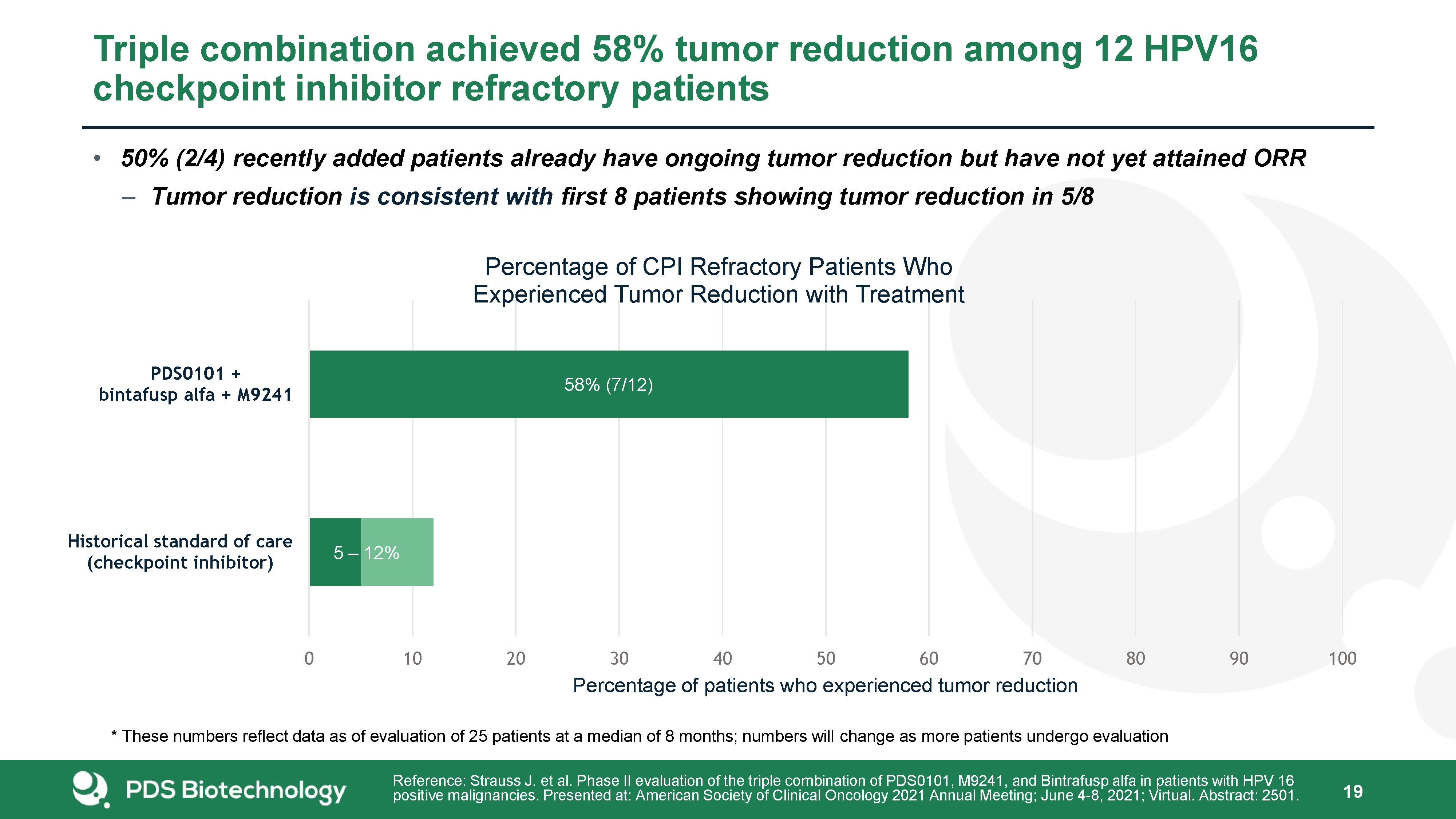
Triple combination achieved 58% tumor reduction among 12 HPV16 checkpoint inhibitor refractory patients 19 Reference: Strauss J. et al. Phase II evaluation of the triple combination of PDS0101, M9241, and Bintrafusp alfa in patients with HPV 16 positive malignancies. Presented at: American Society of Clinical Oncology 2021 Annual Meeting; June 4-8, 2021; Virtual. Abstract: 2501. * These numbers reflect data as of evaluation of 25 patients at a median of 8 months; numbers will change as more patients undergo evaluation 50% (2/4) recently added patients already have ongoing tumor reduction but have not yet attained ORRTumor reduction is consistent with first 8 patients showing tumor reduction in 5/8
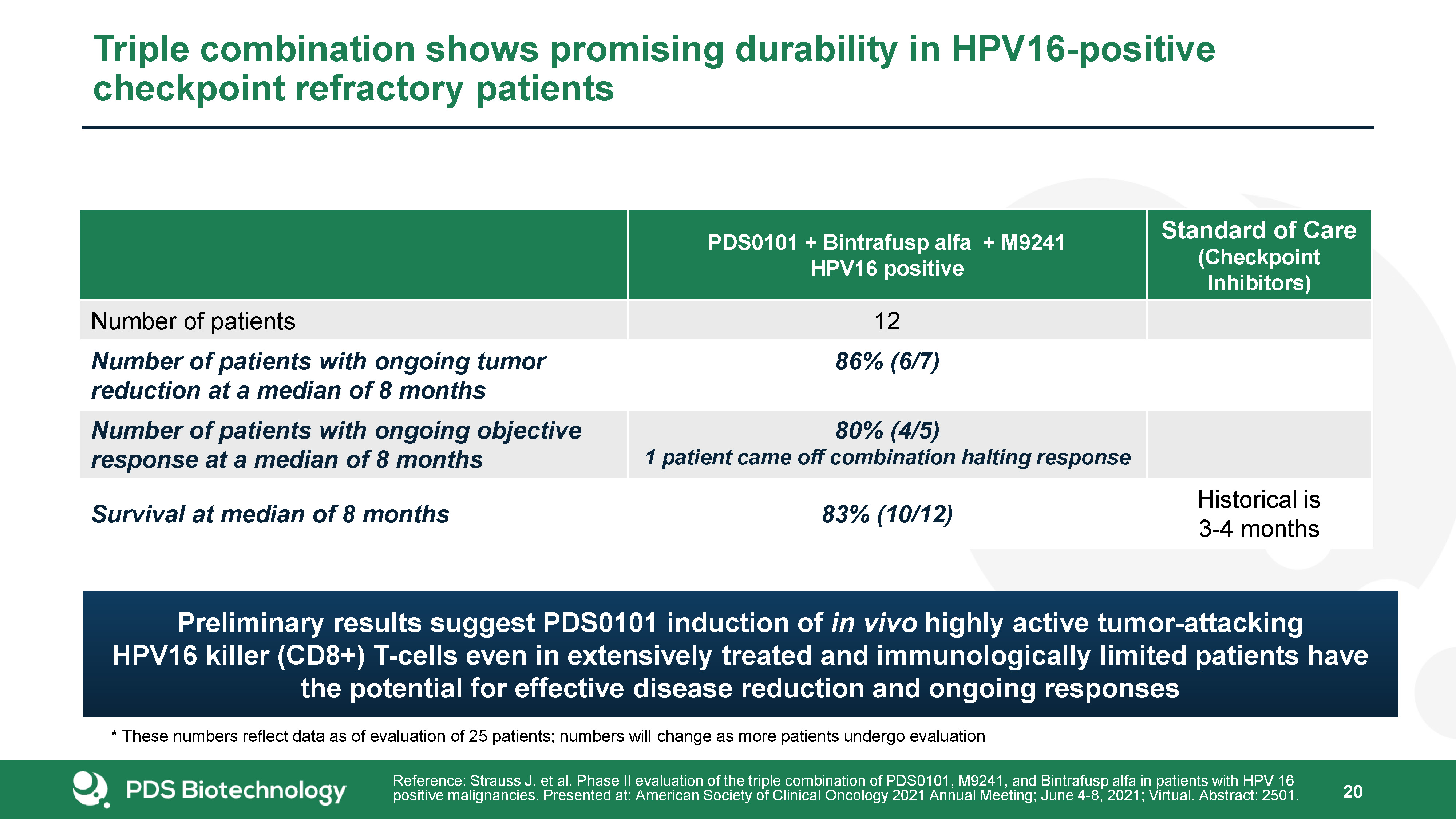
20 Triple combination shows promising durability in HPV16-positive checkpoint refractory patients PDS0101 + Bintrafusp alfa + M9241HPV16 positive Standard of Care(Checkpoint Inhibitors) Number of patients 12 Number of patients with ongoing tumor reduction at a median of 8 months 86% (6/7) Number of patients with ongoing objective response at a median of 8 months 80% (4/5)1 patient came off combination halting response Survival at median of 8 months 83% (10/12) Historical is 3-4 months Reference: Strauss J. et al. Phase II evaluation of the triple combination of PDS0101, M9241, and Bintrafusp alfa in patients with HPV 16 positive malignancies. Presented at: American Society of Clinical Oncology 2021 Annual Meeting; June 4-8, 2021; Virtual. Abstract: 2501. * These numbers reflect data as of evaluation of 25 patients; numbers will change as more patients undergo evaluation Preliminary results suggest PDS0101 induction of in vivo highly active tumor-attacking HPV16 killer (CD8+) T-cells even in extensively treated and immunologically limited patients have the potential for effective disease reduction and ongoing responses
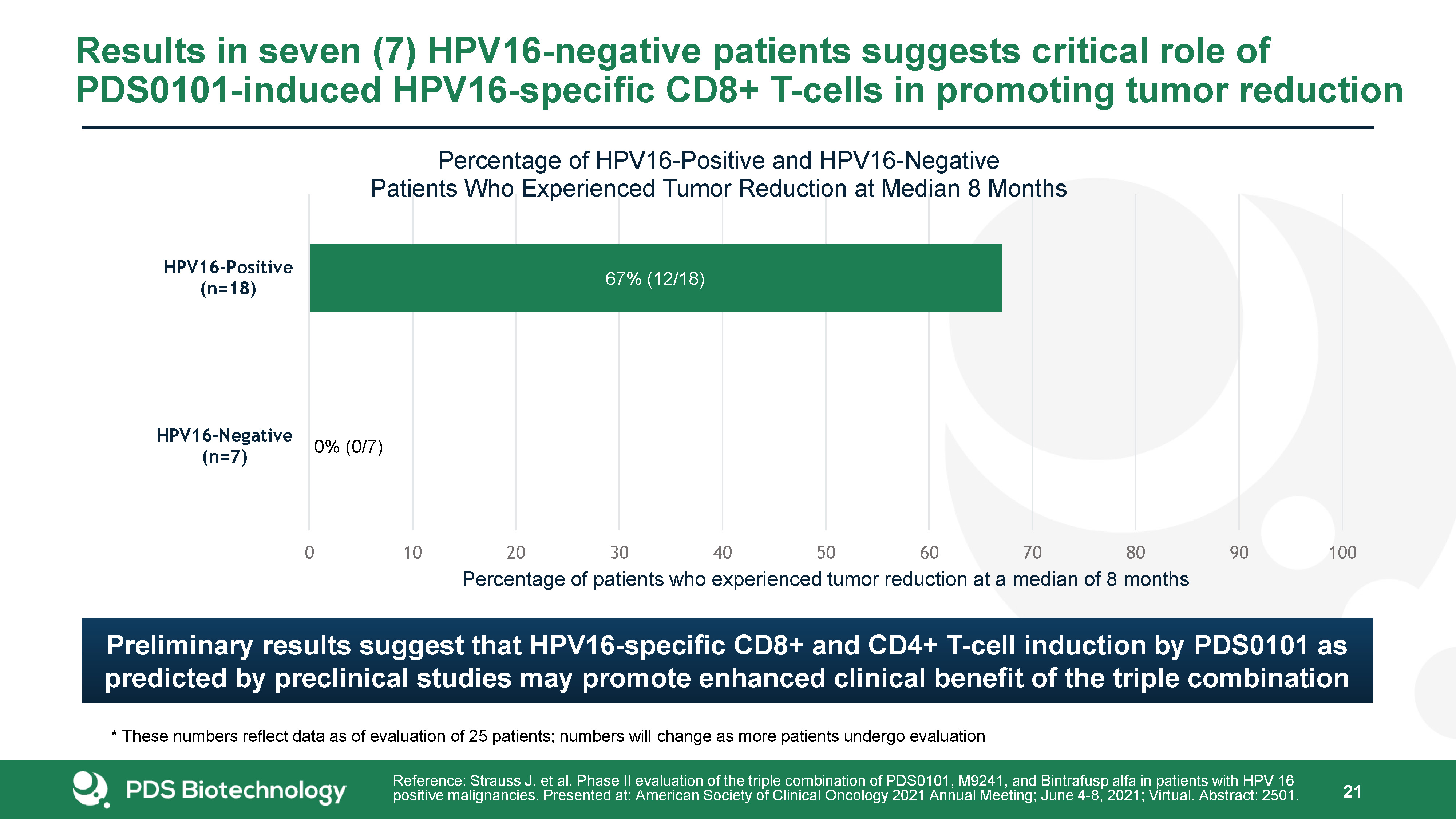
Results in seven (7) HPV16-negative patients suggests critical role of PDS0101-induced HPV16-specific CD8+ T-cells in promoting tumor reduction 21 Reference: Strauss J. et al. Phase II evaluation of the triple combination of PDS0101, M9241, and Bintrafusp alfa in patients with HPV 16 positive malignancies. Presented at: American Society of Clinical Oncology 2021 Annual Meeting; June 4-8, 2021; Virtual. Abstract: 2501. * These numbers reflect data as of evaluation of 25 patients; numbers will change as more patients undergo evaluation Preliminary results suggest that HPV16-specific CD8+ and CD4+ T-cell induction by PDS0101 as predicted by preclinical studies may promote enhanced clinical benefit of the triple combination

22 PDS Biotech-sponsored phase 2 trial evaluating the combination of PDS0101 and KEYTRUDA for first-line treatment of HPV-associated metastatic/recurrent head and neck cancer (VERSATILE-002) Indication First line treatment of patients with HPV-associated head and neck cancer whose cancer has spread or returned Clinical Agents KEYTRUDA (Standard of Care): Anti-PD1 checkpoint inhibitor (ORR ~20%)PDS0101: Versamune®-based immunotherapy generating HPV-specific CD8+ and CD4+ T-cells Study goals Group 1: Objective response rate (ORR) in checkpoint inhibitor (CPI) naïve patientsGroup 2: ORR in patients who have failed checkpoint inhibitor therapy (CPI refractory) Timing Preliminary data anticipated Q4 2021/Q1 2022: ORR minimum of 4 of 17 in CPI naïve and 2 of 21 in CPI refractory required for subsequent stage 2 enrollment (n=95 patients) Trial Partner If achieved, confirmation that PDS0101 enhances the therapeutic benefit of checkpoint inhibitors could expand evaluation of Versamune®-based therapies in multiple cancer indications

23 A Phase 2, investigator-initiated clinical trial evaluating PDS0101 in combination with chemoradiation therapy in patients with locally advanced cervical cancer (IMMUNOCERV) Indication Treatment of patients with locally advanced cervical cancer – Stages IB3-IVA Clinical Agents Chemoradiotherapy (CRT – Standard of Care): Cisplatin & radiation therapyPDS0101: Versamune®-based immunotherapy generating HPV-specific CD8+ and CD4+ T-cells Study goals Safety, rate of regression and local control in patients with primary tumor ≥5cm (n=35 patients) Timing Preliminary data anticipated Q4 2021/1H 2022 – Rate of complete response by PET-CT at 6 months and rate of tumor volume reduction by MRI at 30-40 days from start of treatment Trial Sponsor If successful, this study could support further investigation of Versamune®-based immunotherapies in combination with chemotherapy or CRT to treat multiple cancers

Studies are designed to demonstrate efficacy and broad applicability of PDS0101 and the Versamune® T-cell activating platform 24 Potential to enhance anti-cancer efficacy of various cancer treatments: Combinations with checkpoint inhibitors, chemoradiotherapy and novel therapies may further demonstrate Versamune®’s versatility. Broad potential for additional partnerships: Successful phase 2 studies with PDS0101 could enable a broad pipeline of Versamune®-based oncology products containing various antigens. Potential to treat all types of HPV-cancer: PDS0101 Phase 2 clinical studies address multiple types of HPV-associated cancers. Potential applications beyond oncology: PDS0203 COVID-19 phase 1/2 trials may demonstrate protection and may induce durable T-cell responses against conserved regions of mutating viruses.

PDS0101 Near-Term Milestones and Market Opportunities

Projected PDS0101 milestones through 2022* *Based on current enrollment and forecast modeling as of June 2021. Subject to change. 4Q22 3Q22 2Q22 1Q22 4Q21 3Q21 2Q21 1Q21 Preliminary efficacy Data from advanced HPV-associated cancer trial (NCI) Interim data from HPV-associated Cancer trial (NCI) expected Potential preliminary data from ImmunoCerv combo trial (MD Anderson) expected Potential preliminary data from VERSATILE-002 (KEYTRUDA® combo) Expected completion of accrual HPV-associated cancer trial (NCI) PDS0101 26 PDS Biotech Funded Clinical Trials Partner Co-Funded Clinical Trials
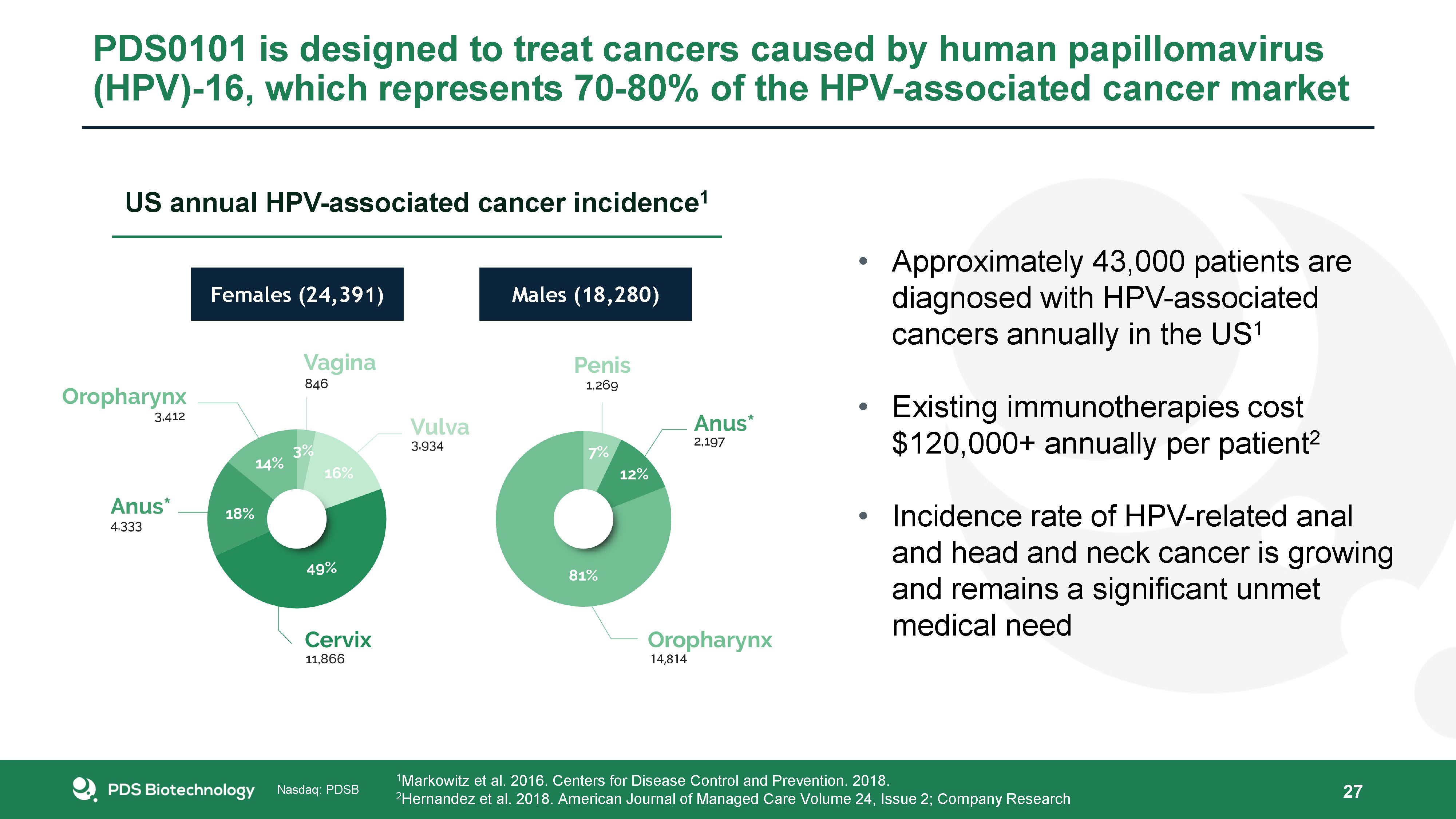
27 Approximately 43,000 patients are diagnosed with HPV-associated cancers annually in the US1Existing immunotherapies cost $120,000+ annually per patient2Incidence rate of HPV-related anal and head and neck cancer is growing and remains a significant unmet medical need 1Markowitz et al. 2016. Centers for Disease Control and Prevention. 2018.2Hernandez et al. 2018. American Journal of Managed Care Volume 24, Issue 2; Company Research Females (24,391) Males (18,280) US annual HPV-associated cancer incidence1 PDS0101 is designed to treat cancers caused by human papillomavirus (HPV)-16, which represents 70-80% of the HPV-associated cancer market

Versamune® has demonstrated the potential for immunological compatibility with a wide array of tumor and pathogenic antigens Reference: Clin Cancer Res. 2009 Sep 1;15(17):5323-37. doi: 10.1158/1078-0432.CCR-09-0737 Versamune®’s unique flexibility means it may work well with a wide range of identified tumor and infectious disease antigens3 tumor antigens are currently being utilized with the Versamune® platform beyond HPVPDS0102 (TARP) for the treatment of AML, prostate and breast cancerPDS0103 (MUC1) for the treatment of breast, colorectal, ovarian and NSCLCPDS0104 (TRP2) for the treatment of melanomaMore than 70 tumor antigens have been identified to dateProof-of-concept data from ongoing clinical trials could trigger development activities for Versamune®-based products through partnerships and licensing 28

PDS Biotech’s robust Versamune® -based oncology pipeline is being developed in partnership with the leaders in immuno-oncology 29 Reference: Data on file.

Farmacore License Agreement for Versamune® -Based COVID Vaccine

Development of PDS0203, if successful, may offer potential advantages as a second generation COVID-19 vaccine May be effective against multiple COVID-19 variantsDemonstrated induction of killer CD8+ and helper CD4+ T cells that can target less variable regions of the SARS-CoV-19 virus and may be effective against currently circulating variants.May induce long-lasting immunityDemonstrated induction of long-lasting, virus-specific memory T-cells necessary for longer term protection.High potential for safetyPDS0203 is a subunit vaccine, and does not require the use of attenuated viruses, traditional adjuvants, DNA or RNA. Versamune®-based vaccines have shown no serious or dose limiting reactions. 31

Consortium has received a commitment of up to ~US$60 million from MCTI, Brazil to support phase 1-3 clinical development and manufacturing scale-upPDS Biotech has licensed Versamune® technology to Farmacore to develop a COVID-19 vaccine candidate Farmacore is responsible for antigen manufacturing and clinical developmentPhase 1/2 study anticipated to start following Farmacore’s submission of a full data package to and subsequent approval from Anvisa (Brazilian regulatory agency)PDS Biotech is closely monitoring the evolving political situation in Brazil as which may result in potential challenges to developing a Versamune®-based COVID-19 vaccineIntellectual property protections for a Versamune-based COVID-19 vaccine potentially at riskBrazil’s senate has voted for compulsory licensing for COVID-19 vaccine technology Legislative process is ongoingFunding release is subject to negotiations among Farmacore, MCTI, and Brazilian authorities PDS0203, if development is successful, could offer another option to address the COVID-19 global health crisis 32

Final Comments

Investment Highlights 34 Promising early data in both oncology and infectious disease: Early clinical data and preclinical data suggest potential efficacy, safety and versatility of the Versamune® platformNear-term milestones: Preliminary data from MD Anderson-led study and PDS Biotech-led VERSATILE-002 as well as additional data from NCI-led Phase 2 anticipated Q4 2021 - Q2 2022Validation of approach: All three on-going phase 2 clinical trials supported and partnered with leading and top-tier institutions in the field of cancer and immuno-oncologyCommercialization path: Clinical studies demonstrating enhancement of FDA-approved anti-cancer therapies may offer potential for expedited programsRapid adoption strategy: Evaluation of PDS0101 in combination with standard of care in multiple HPV-associated cancers Potential Advantages and Differentiators *Based on current enrollment and forecast modeling as of May 2021. Subject to change.
