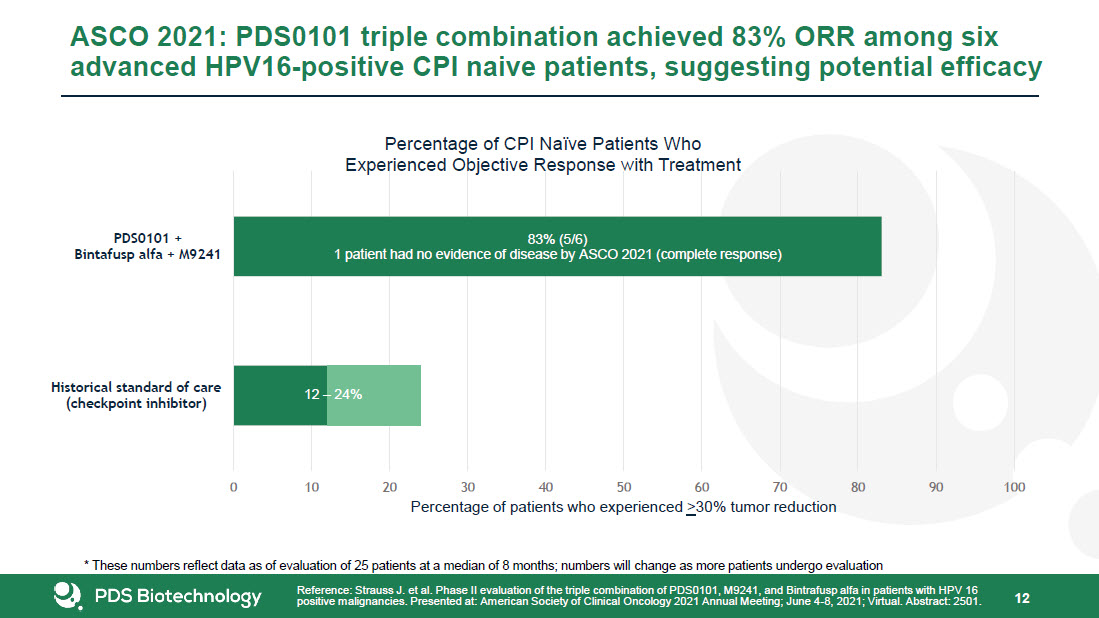
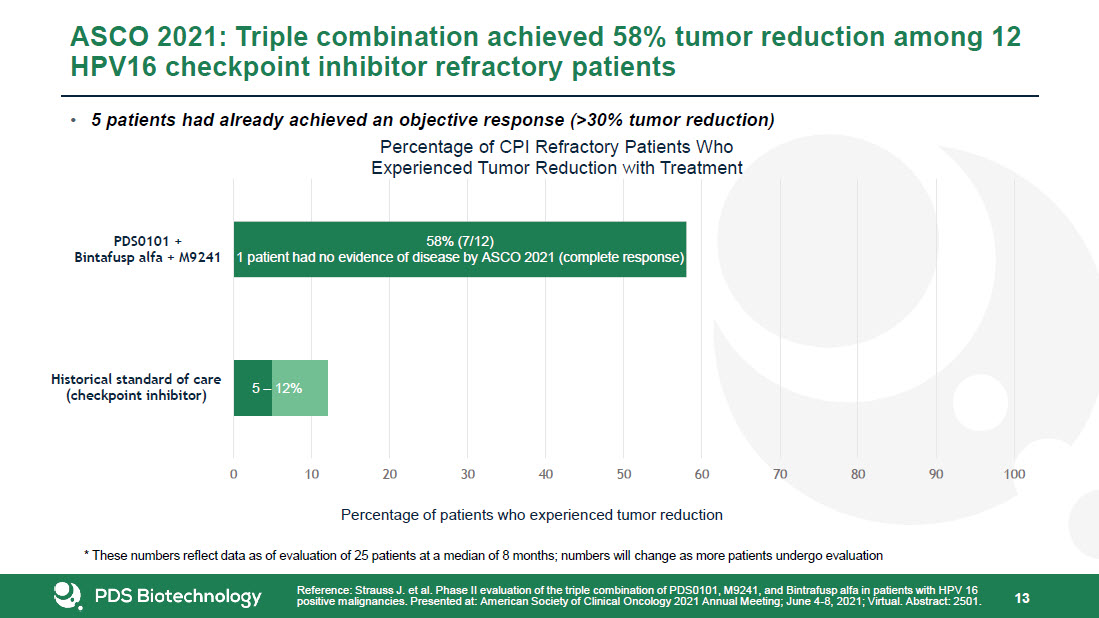
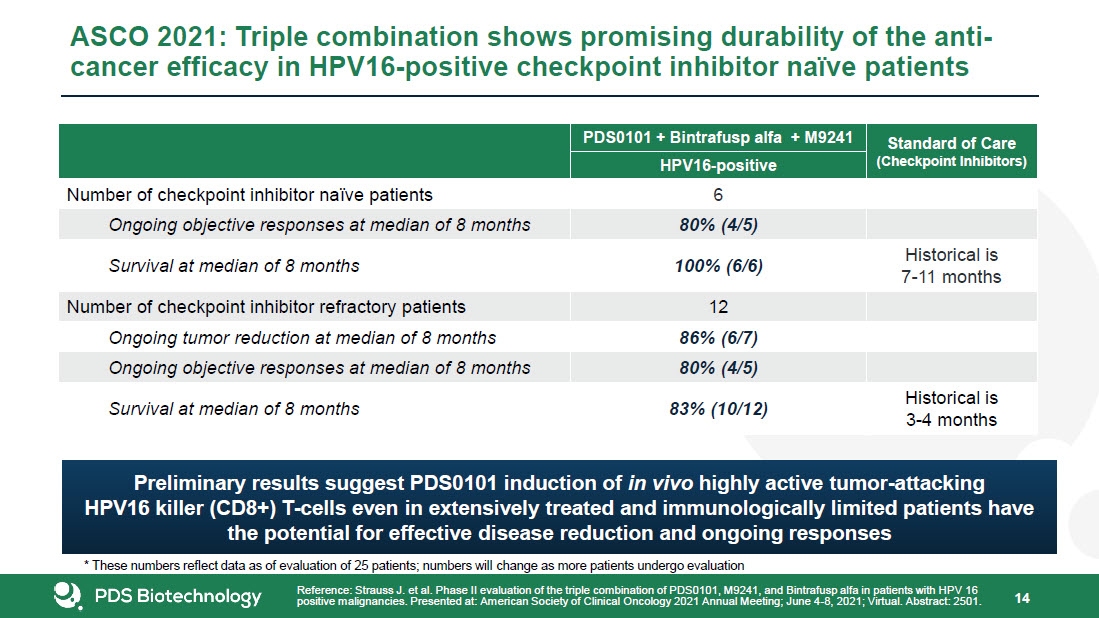
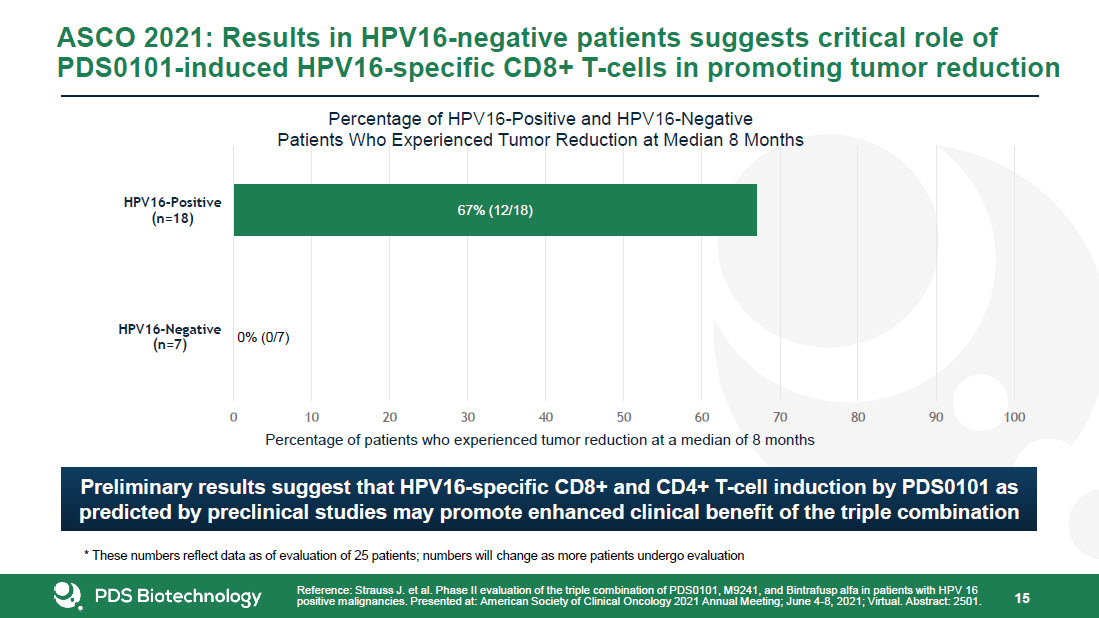
* Forward-Looking Statements This presentation contains forward-looking statements about PDS Biotechnology Corporation (“PDSB”), and its businesses, business prospects, strategies and plans, including but not limited to statements regarding anticipated pre-clinical and clinical drug development activities and timelines and market opportunities. All statements other than statements of historical facts included in this presentation are forward-looking statements. The words “anticipates,” “may,” “can,” “plans,” “believes,” “estimates,” “expects,” “projects,” “intends,” “likely,” “will,” “should,” “to be,” and any similar expressions or other words of similar meaning are intended to identify those assertions as forward-looking statements. These forward-looking statements involve substantial risks and uncertainties that could cause actual results to differ materially from those anticipated.Factors that may cause actual results to differ materially from such forward-looking statements include those identified under the caption “Risk Factors” in the documents filed with the Securities and Exchange Commission (“SEC”) from time to time, including its Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this presentation. Except to the extent required by applicable law or regulation, PDSB undertakes no obligation to update the forward-looking statements included in this presentation to reflect subsequent events or circumstances.