Exhibit 99.1

Developing Transformational Immunotherapies for Cancer NASDAQ: PDSB October 2023

Forward-Looking Statements This communication contains forward-looking statements (including within the meaning of Section 21E of the United States Securities Exchange Act of 1934, as amended, and Section 27A of the United States Securities Act of 1933, as amended) concerning PDS Biotechnology Corporation (the “Company”) and other matters. These statements may discuss goals, intentions and expectations as to future plans, trends, events, results of operations or financial condition, or otherwise, based on current beliefs of the Company’s management, as well as assumptions made by, and information currently available to, management. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as “may,” “will,” “should,” “would,” “expect,” “anticipate,” “plan,” “likely,” “believe,” “estimate,” “project,” “intend,” “forecast,” “guidance”, “outlook” and other similar expressions among others. Forward-looking statements are based on current beliefs and assumptions that are subject to risks and uncertainties and are not guarantees of future performance. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: the Company’s ability to protect its intellectual property rights; the Company’s anticipated capital requirements, including the Company’s anticipated cash runway and the Company’s current expectations regarding its plans for future equity financings; the Company’s dependence on additional financing to fund its operations and complete the development and commercialization of its product candidates, and the risks that raising such additional capital may restrict the Company’s operations or require the Company to relinquish rights to the Company’s technologies or product candidates; the Company’s limited operating history in the Company’s current line of business, which makes it difficult to evaluate the Company’s prospects, the Company’s business plan or the likelihood of the Company’s successful implementation of such business plan; the timing for the Company or its partners to initiate the planned clinical trials for PDS0101 and other Versamune® and Infectimune® based product candidates; the future success of such trials; the successful implementation of the Company’s research and development programs and collaborations, including any collaboration studies concerning PDS0101 and other Versamune® and Infectimune® based product candidates and the Company’s interpretation of the results and findings of such programs and collaborations and whether such results are sufficient to support the future success of the Company’s product candidates; the success, timing and cost of the Company’s ongoing clinical trials and anticipated clinical trials for the Company’s current product candidates, including statements regarding the timing of initiation, pace of enrollment and completion of the trials (including the Company’s ability to fully fund its disclosed clinical trials, which assumes no material changes to the Company’s currently projected expenses), futility analyses, presentations at conferences and data reported in an abstract, and receipt of interim or preliminary results (including, without limitation, any preclinical results or data), which are not necessarily indicative of the final results of the Company’s ongoing clinical trials; any Company statements about its understanding of product candidates mechanisms of action and interpretation of preclinical and early clinical results from its clinical development programs and any collaboration studies; to aid in the development of the Versamune® platform; and other factors, including legislative, regulatory, political and economic developments not within the Company’s control. The foregoing review of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors included in the Company’s annual, quarterly and periodic reports filed with the SEC. The forward-looking statements are made only as of the date of this press release and, except as required by applicable law, the Company undertakes no obligation to revise or update any forward-looking statement, or to make any other forward-looking statements, whether as a result of new information, future events or otherwise. Versamune® and Infectimune® are registered trademarks of PDS Biotechnology Corporation KEYTRUDA® is a registered trademark of Merck Sharp and Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA

PDS0101 to enter Phase 3 registrational trial in 2023 to treat recurrent or metastatic, HPV16-positive head and neck squamous cell cancer (HNSCC) Fast Track Designation PDS0101 addresses large and growing market with significant unmet need Transformational data generated with PDS0101 and PDS0301 in multiple Phase 2 clinical studies Financial: Cash as of June 30, 2023 - $60.6M- Adequate cash runway for the approximately the next 12 months with initiation of a registrational trial in 2023 Executive Summary: Positioned for Market Leadership Company Overview T cell activating platforms and antibody conjugated immuno-cytokine platform to develop safer, more effective and longer lasting cancer immunotherapies 1 2 3 4

Experienced Management Team Historical success in development and commercialization of leading pharmaceutical products Frank Bedu-Addo, PhD Chief Executive Officer Senior executive experience with management of strategy and execution at both large pharma and biotechs Notable drug development: Abelcet® (Liposome Company/ Elan) PEG-Intron® (Schering-Plough/ Merck) Matthew Hill Chief Financial Officer 20 years of financial and operational leadership roles for life sciences companies Former Chief Financial Officer of several publicly traded companies Lauren V. Wood, MD Chief Medical Officer 30 years of translational clinical research experience Former Vaccine Branch Clinical Director at National Cancer Institute Center for Cancer Research Gregory Conn, PhD Chief Scientific Officer Co-founder 35 years of drug development experience In-depth experience with biotech drug discovery, product development and manufacturing

PDS Biotech Versamune® Overview Designed to address limitations of current immunotherapy PLATFORM: Induces powerful, long-lasting anti-tumor response by promoting uptake of tumor-specific proteins by the immune system and activates a specific signaling pathway that promotes the production of active tumor-infiltrating multifunctional CD8 killer and CD4 helper T cells Versamune® PDS0101 PDS0102 PDS0103 PDS0104 Product Candidates

Versamune® Induces the Right Type, Potency and Quantity of Multifunctional Killer and Helper T Cells Comprised of positively charged lipid (R-DOTAP) co-administered with proprietary tumor-specific proteins, delivered via subcutaneous injection Presents antigen to CD4 and CD8 T cells. Activates the Type I Interferon pathway, leading to potent, multifunctional, antigen specific T cell responses Human clinical trials confirm induction and accumulation of multifunctional T cells in the tumor, which correlated with clinical response and elimination of circulating tumor DNA and clinical response (SITC 2022) Recruits T cells to lymph nodes Trains T cells to target tumors Arms T cells to kill tumor cells 1 2 3 Injection Site Lymph Node Tumor Site References: Gandhapudi SK, et al. 2019. Antigen priming with enantiospecific cationic lipid nanoparticles induces potent antitumor CTL responses through novel induction of a Type I IFN response. J Immunol. 202 (12): 3524-3536. Smalley Rumfield C et al. 2020. Immunomodulation to enhance the efficacy of an HPV therapeutic vaccine. J. for ImmunoTherapy of Cancer 8:e000612.
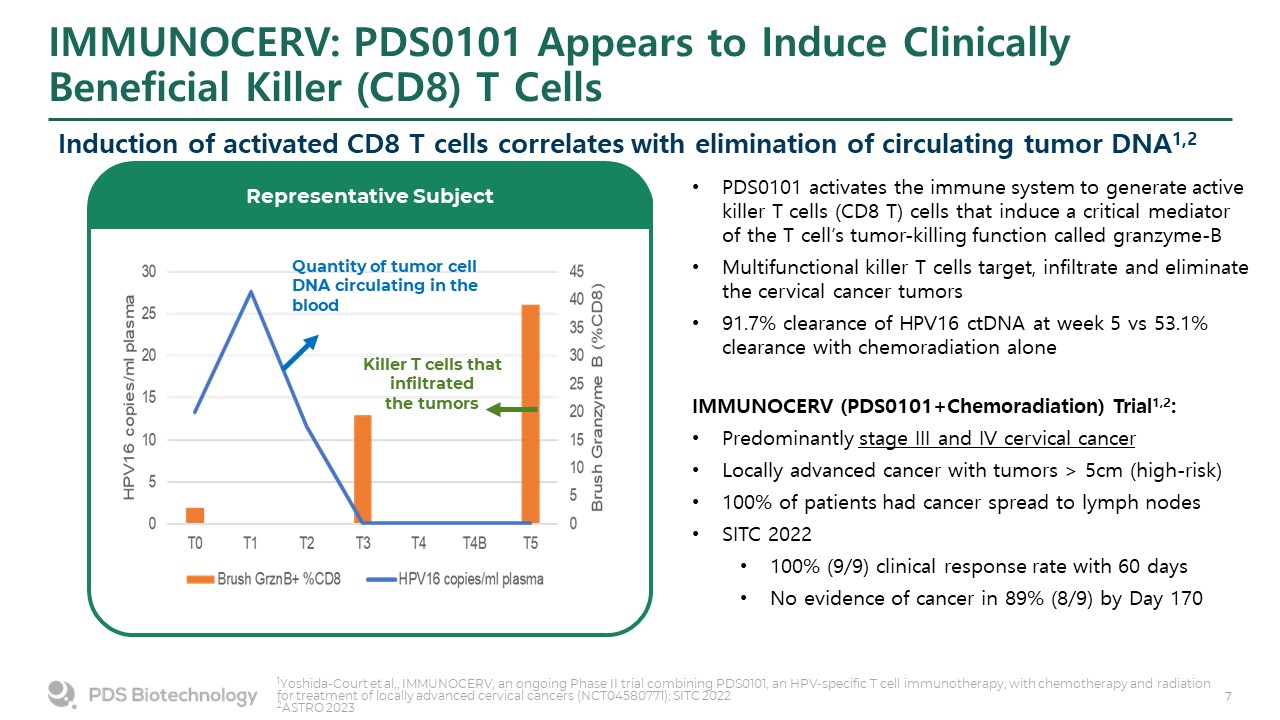
IMMUNOCERV: PDS0101 Appears to Induce Clinically Beneficial Killer (CD8) T Cells PDS0101 activates the immune system to generate active killer T cells (CD8 T) cells that induce a critical mediator of the T cell’s tumor-killing function called granzyme-B Multifunctional killer T cells target, infiltrate and eliminate the cervical cancer tumors 91.7% clearance of HPV16 ctDNA at week 5 vs 53.1% clearance with chemoradiation alone Quantity of tumor cell DNA circulating in the blood Killer T cells that infiltrated the tumors Representative Subject IMMUNOCERV (PDS0101+Chemoradiation) Trial1,2: Predominantly stage III and IV cervical cancer Locally advanced cancer with tumors > 5cm (high-risk) 100% of patients had cancer spread to lymph nodes SITC 2022 100% (9/9) clinical response rate with 60 days No evidence of cancer in 89% (8/9) by Day 170 Induction of activated CD8 T cells correlates with elimination of circulating tumor DNA1,2 1Yoshida-Court et al,, IMMUNOCERV, an ongoing Phase II trial combining PDS0101, an HPV-specific T cell immunotherapy, with chemotherapy and radiation for treatment of locally advanced cervical cancers (NCT04580771); SITC 2022 2ASTRO 2023

HPV16-Positive Head and Neck Squamous Cell Cancer (HNSCC) Disease Overview and Market Size

Est. HPV16Locally Advanced,Unresectable andMetastatic HPV16-positive HNSCC Presents a Significant Market Opportunity Largely Attributed to the High Rate of Oral HPV Infections in Men Data sources: 1 PD-L1 negative and PD-L1 positive populations (> 9000 incidence PD-L1 Positive) https://seer.cancer.gov/statfacts/html/oralcav.html; https://www.cdc.gov/cancer/hpv/basic_info/hpv_oropharyngeal.htm; https://virologyj.biomedcentral.com/articles/10.1186/s12985-021-01688-9; https://seer.cancer.gov/statistics-network/explorer/application.html?site=3&data_type=1&graph_type=4&compareBy=sex&chk_sex_1=1&race=1&age_range=1&advopt_precision=1&hdn_view=0; https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5002133/ ~18,300 ~34,000 ~38,100 ~54,400 Est. U.S. HPV Positive Oral and Pharyngeal Cancer Est. U.S. Oral and Pharyngeal Cancer Est. HPV16 Genotype $2-3B Market Opportunity in US1 HPV cancer incidences continue to increase despite preventive HPV vaccine HPV vaccination is not expected to impact the rate of HPV-positive cancer incidence for the near-term Existing immunotherapies cost $150,000+ annually per patient1 No available HPV specific therapy Initial commercial opportunity for PDS0101

Targeted treatment option to address the growing population of HPV16-positive HNSCC and improve outcomes Novel MOA that is clinically effective in a broader patient population and provides more durable responses. Safer and more effective treatments that may be used with or in place of current standard of care Better tolerability and less toxic alternatives to chemotherapy Despite the Availability of Treatments, Significant Unmet Needs Remain in Recurrent or Metastatic HNSCC KEYTRUDA® KEYTRUDA® Plus Chemo Chemotherapy + EGFR Inhibitor Objective Response Rate (ORR) 19% 36% 35% Progression Free Survival (PFS) 3.2 mos 5.0 mos 5.0 mos 12-Month Survival Rate 50% 55% 44% Median Overall Survival (OS) 12.3 mos 13.6 mos 10.3 mos Key Toxicities Anemia Fatigue Weight loss Hypokalemia Additional to KEYTRUDA®: Neutropenia Mucosal inflammation Thrombocytopenia Stomatitis Neutropenia ��Anemia Thrombocytopenia Nausea/vomiting Hypokalemia Rash Fatigue Mucosal inflammation Treatment Related Grade 3+ Toxicities 17% 72% 69% Oncologist2 – Stated Unmet Medical Needs in HNSCC Standard of Care for Recurrent or Metastatic HNSCC – Published Results*1 1KEYNOTE-048 Study Burtness B et al, Lancet 2019 2Primary Market Research 2022 * No control or comparative studies have been conducted between immune checkpoint inhibitors and PDS0101

PDS0101 for HPV16-Positive HNSCC

VERSATILE-002 Phase 2 Clinical Trial – Results Support Initiation of Phase 3 Clinical Trial in ICI Naïve R/M HNSCC Objective: To assess the combination of PDS0101 and KEYTRUDA® in ICI naïve subjects with recurrent or metastatic HPV-positive HNSCC KEYTRUDA® (pembrolizumab) FDA Approved Standard of Care Partner Study Design Open-label, non-randomized, adaptive design study N=54 Enrollment complete Key Entry Criteria for ICI Naïve Subjects Recurrent or metastatic HNSCC ≥18 years of age HPV16-Positive tumor Combined positive score (CPS) ≥1 Pembrolizumab 200mg IV Q3W up to 35 Cycles (2 years) PDS0101 1 mL subcutaneous injection at Cycles 1, 2, 3, 4 and 12 Study Treatment Primary: Best overall response (BOR) of confirmed complete response (CR) or confirmed partial response (PR) per RECIST 1.1 Key Secondary: Progression Free Survival (PFS) per RECIST 1.1 Overall Survival (OS) Safety and tolerability Achievement of Statistical Threshold for Efficacy Endpoints Fast TrackDesignation

Disease Stabilization or Tumor Reduction in 81% of Patients Tumor Shrinkage in 60% (31/52) with Confirmed Objective Response in 27%1 (14/52) to Date Assessments based on Investigator assessment per RECIST 1.1 Data on File. 08/02/23 Data Cut. Burtness B et al., Lancet. 2019; 394:1915-1928. Best Percentage Change from Baseline in Target Lesions (mITT population) VERSATILE-002 Months (95% CI) PDS0101+KEYTRUDA® 8.1 KEYNOTE-048 (CPS≥1) Months (95% CI) KEYTRUDA® Monotherapy 3.2 KEYTRUDA® + Chemo 5.0 EXTREME Chemo 5.0 Progression Free Survival 1Achievement of Statistical Threshold for Efficacy

PDS0101 and KEYTRUDA® Combination in ICI Naïve HNSCC Demonstrates Promising Patient Survival to Date Median OS Not Yet Estimable Data on File. 08/02/23 Data Cut. Kaplan-Meier Estimates of Overall Survival (OS) (Intent-to-Treat Population) No. of Subjects at Risk (Events) 55 (0) 53 (2) 50 (3) 42 (5) 41 (6) 36 (7) 32 (8) 27 (8) 24 (8) 21 (8) 19 (9) 17 (9) 17 (9) 17 (9) 15 (9) 14 (9) 11 (10) 11 (10) 11 (10) 9 (10) 8 (10) 7 (10) 6 (10) 4 (10) 2 (10) 0 (10) 12 Month OS Rate – 80% 24 Month OS Rate – 74% 12-month OS Rates CPS 1-19 = 75% CPS ≥ 20 = 85% 24-month OS Rates CPS 1-19 = Not yet estimable CPS ≥ 20 = 85%

PDS0101 and KEYTRUDA® Combination in ICI NaïveHNSCC Demonstrates Promising Patient Survival to Date Overall Survival is Primary Endpoint in Planned Phase 3 Study VERSATILE-003 * No controlled or comparative studies have been conducted between checkpoint inhibitors and PDS0101Data on File. 08/02/23 Data CutBurtness B et al., Lancet. 2019; 394:1915-1928 OS Rate (%) VERSATILE-002 VERSATILE-002 ® ® ® KEYNOTE-048(CPS≥1) KEYNOTE-048(CPS≥1) 12-month OS Rate 24-month OS Rate
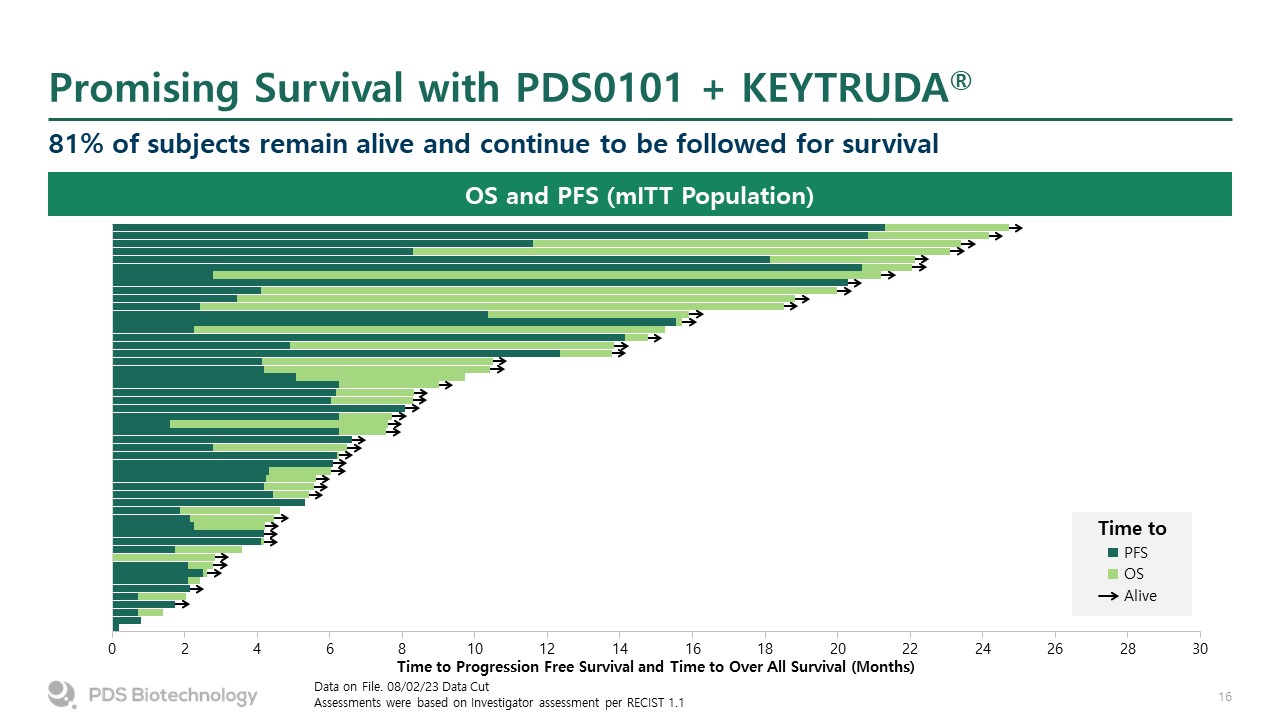
Promising Survival with PDS0101 + KEYTRUDA® 81% of subjects remain alive and continue to be followed for survival Data on File. 08/02/23 Data Cut Assessments were based on Investigator assessment per RECIST 1.1 OS and PFS (mITT Population) Time to PFS OS Alive

The Addition of PDS0101 to KEYTRUDA® Does not Appear to Compound Toxicity * No controlled or comparative studies have been conducted between checkpoint inhibitors and PDS0101Data on File. 08/02/23 Data CutBurtness B et al., Lancet. 2019; 394:1915-1928 Ferris RL, et al. NEJM. 2016;375:1856-67. Only 8 subjects (13%) had Grade 3 PDS0101-KEYTRUDA®: Fatigue (2), Rash, Alanine aminotransferase increased, Blood alkaline phosphatase increased, Lymphocyte count decreased, Autoimmune colitis, Colitis, Headache, Acute kidney injury, Hyponatraemia, Hyperglycaemia Published results 13-17% grade 3-5 TRAE with approved ICI immunotherapy No subjects had Grade 4 or 5 TRAEs PDS0101+KEYTRUDA® Treatment Related Adverse Events (TRAE) ≥5% (Safety Population, N=62) Safety data in over 130 patients to date across multiple Phase 1 and 2 Studies Safety data, anti-tumor responses and patient survival suggest that the Versamune® based therapies such as PDS0101 could be ideal candidates for combination oncology treatments Preferred Term n (%) Any Combination-TRAE 49 (79.0) Grade 1 23 (37.1) Grade 2 18 (29.0) Grade 3 8 (12.9) Grade 4 0 Grade 5 0

PDS0101 with KEYTRUDA® Well Tolerated in VERSATILE-002 to Date with Few Serious Toxicities Serious (Grade 3–5) Treatment Related Adverse Events *No controlled or comparative studies have been conducted between checkpoint inhibitors and PDS0101 Data on File. 08/02/23 Data Cut Burtness B et al. Lancet. 2019;394:1915-1928 ® ® ® VERSATILE-002 No Grade 4 or 5 TRAE Subjects (%) KEYNOTE-048(CPS≥1)

Data Continues to Hold Up as More Patients are Enrolled and Follow up Time is Extended Data Summary Table Shows Consistency in Results PDS0101 KEYNOTE-048 Comments ASCO 2023 (06/05/23) KOL event (09/27/23) Cut-off Date January 13, 2023 August 02, 2023 Patients with CPS 1-19 50% 60% 48% Aug 02 cut off has a higher percentage of patients with CPS 1-19 (lower response to KEYTRUDA®). KEYNOTE rates among subjects with CPS ≥1. Patients with CPS ≥20 50% 40% 52% Disease Control Rate (SD+PR+CR) 70.6% 80.8% Patients with tumor reduction 67.6% 59.6% Confirmed ORR on cut-off date 26.5% 26.9% 19% ORR steady as more patients enroll Median OS (CPS ≥1) Not yet estimable Not yet estimable 12.3 months KEYTRUDA® exposes the cancer to the immune system (improves with higher CPS score). Injection of PDS0101 activates T cells to infiltrate, weaken (disable) and, or kill the exposed cancer cells, thus providing the potential to promote prolonged survival. 12-month OS rate (CPS ≥1) 87% 80% 50% KEYTRUDA® exposes the cancer to the immune system (improves with higher CPS score). Injection of PDS0101 activates T cells to infiltrate, weaken (disable) and, or kill the exposed cancer cells, thus providing the potential to promote prolonged survival. 12-month OS rate (CPS 1-19) N/A 75% 44% 12-month OS rate (CPS ≥20) N/A 85% 56% 24-month OS rate (CPS ≥1) N/A 74% 29% PFS (CPS ≥1) 10.4 months 8.1 months 3.2 months PDS0101 promotes prolonged PFS even in patients with lower CPS score. PFS for CPS ≥1 remains extended despite increased percentage of patients in lower CPS score group. PFS (CPS 1-19) 5.1 months 2.2 months PFS (CPS ≥20) 11.6 months 3.4 months * No control or comparative studies have been conducted between immune checkpoint inhibitors and PDS0101 Burtness B et al. Lancet. 2019;394:1915-1928 Burtness, B. et al. J Clin Oncol. 2022;40:2321-2332.

Combination of PDS0101 & KEYTRUDA® Continues to Show Promising Safety and Survival Outcomes VERSATILE-002 Study Shows Potential Of PDS0101 to Safely Modify the Tumor Microenvironment to Promote Patient Survival The 24-month OS rate in the ICI naïve cohort is 74%; Published results of 29% in KEYNOTE-048 The 12-month OS rate in the ICI naïve cohort is 80%; Published results of 50% in KEYNOTE-048 The addition of PDS0101 to KEYTRUDA® does not appear to compound toxicity in ICI naïve patients 13% (8/62) Grade 3 and 0% Grade 4 & 5 Treatment Related Adverse Events * No controlled or comparative studies have been conducted between checkpoint inhibitors and PDS0101 Data on File. 08/02/23 Data Cut. Burtness B, et al. Lancet. 2019;394:1915-28.

Timeline to Registrational Trial Initiation Worldwide Randomized, Controlled Clinical Study to Be Initiated Q4 2023 PDS0101 + KEYTRUDA® in Recurrent or Metastatic HPV16-Positive HNSCC 2Q 2022 3Q 2022 1Q 2023 2Q 2023 3Q 2023 4Q 2023 FDA Fast Track designation for PDS0101 + KEYTRUDA® Successful EOP2 meeting with FDA Initiated PDS0101 tech-transfer, scale up at selected Phase 3 clinical/commercial manufacture Completed Phase 3 clinical manufacturing of PDS0101 Obtained visibility to potential OS and PFS information for VERSATILE-002 trial needed to finalize VERSATILE-003 trial design Completed CMC-related activities for PDS0101 Obtained feedback from EU regulatory agencies on protocol Received feedback from FDA allowing for initiation of VERSATILE-003 Initiate site activation and related clinical, operational activities (4- to 6-month process) InitiateVERSATILE-003 Phase 3 Trial

Assessing the Role of PDS0101 in Extending Survival in the Absence of a VERSATILE-002 KEYTRUDA® Control Arm Strauss J, et al. Journal for ImmunoTherapy of Cancer. 2020;8:e001395. Evaluation of the combination of PDS0101 and KEYTRUDA in patients who have failed KEYTRUDA therapy provides an “internal control” Important Consideration: ICI refractory patients have more advanced disease than ICI naïve patients and are much more difficult to treat with immunotherapy Presents a higher treatment bar than ICI naïve patients On alternative ICI therapy, historical overall survival rates in HPV-positive ICI refractory cancer is reported to be approximately only 3-4 months Results provide useful information regarding Role of PDS0101 targeted immunotherapy in promising VERSATILE-002 survival rates OS endpoint in potential triple combination study with ICI, PDS0101 & PDS0301(NHS-IL12) Evaluation of PDS0101 + KEYTRUDA® in HPV16-positive head and neck patients who have failed/progressed on KEYTRUDA® therapy (ICI Refractory)

Versamune® Based Oncology Pipeline Partnerships with World Class Institutions in Immuno-Oncology Reference: Data on file. PDS Biotech Funded Candidate/ Study Indication Combination PC P1 P2 P3 R Partner(s) Clinical (Lead) PDS0101 (HPV)/ VERSATILE-002 Recurrent or metastatic HPV16-positive head and neck cancer Arm 1: ICI naïve Arm 2: ICI refractory KEYTRUDA® (standard of care) IIT Studies PDS0101 (HPV)/ IMMUNOCERV 1st-line treatment of locally advanced (IB3-IVA) cervical cancer Chemo-radiation (standard of care) PDS0101 (HPV)/ Mayo Clinic Pre-metastatic HPV-positive oropharyngeal cancer (OPSCC) Arm 1: PDS0101 monotherapy Arm 2: PDS0101 + KEYTRUDA® KEYTRUDA® (standard of care) Preclinical Candidates PDS0102(TARP) TARP-positive AML, prostate and breast cancers TBD PDS0103 (MUC1) MUC1-positive breast, colon, lung, ovarian and other cancers TBD PDS0104(TRP2) Melanoma TBD Partner Co-Funded Fast Track Designation

VERSATILE-003 Phase 3 Study Design Global Randomized, Controlled Clinical Study with Estimated 90–100 Sites Overall survival (OS) Primary Endpoint For the treatment of recurrent or metastatic HPV16-positive HNSCC Targeted Indication Randomization 2:1 PDS0101 +KEYTRUDA® PDS0101 + KEYTRUDA® KEYTRUDA® KEYTRUDA® Planned Interim Analysis: OS Final Analysis:OS Enrollment Follow-up

Survival Rates Demonstrate Potential Contribution of PDS0101 to Survival in Advanced Head and Neck Cancer PDS0101 + KEYTRUDA Shows Promising Survival Benefit even in ICI Refractory Patients * No controlled or comparative studies have been conducted between checkpoint inhibitors and PDS0101 Data on File. 08/02/23 Data Cut Burtness B et al., Lancet. 2019;394:1915-1928 Ferris RL, et al. NEJM. 2016;375:1856-67. Bila M, et al. Frontiers in Oncology. Jan 2022;12:761428. 12-month Overall Survival Rate ® First-line R/M HNSCC ICI Refractory HNSCC 12-month Overall Survival Rate Progression ®

Antibody-Drug Conjugate (ADC) IL-12 (PDS0301)

PDS Biotech Antibody-Drug Conjugate IL-12 (PDS0301) Overview Antibody-Conjugated IL-12 Opportunities PDS0301 is a novel investigational tumor-targeting IL-12 that enhances the proliferation, potency and longevity of T cells in the tumor microenvironment Proprietary combination of PDS0301 with Versamune® shows reduction of myeloid derived suppressor cells (MDSC) in tumors, a mechanism by which tumors suppress attack by T cells Monotherapy Combinations: Versamune® Based Immunotherapies Chemotherapy Radiation HDAC Inhibitors* *Histone Deacetylase Inhibitor

NCI-led Triple Combination: PDS0301 + PDS0101 + ICI Advanced HPV16-Positive Anal, Cervical, Head and Neck, Penile, Vaginal, Vulvar Cancer Patients Who Are ICI Refractory 172% grade 3 and higher adverse events reported in KEYNOTE-048 Burtness 2019 https://doi.org/10.1016/S0140-6736(19)32591-7 Goswami 2022 http://dx.doi.org/10.1136/jitc-2022-SITC2022.0695 Partner FDA Approved Standard of Care None Immunology/Immune Correlates SITC, November 2022: Greater than two-fold increase in HPV16-specific T cells in the blood of 11/14 (79% ) of the evaluated patients Induction of multifunctional killer (CD8) T cells Increases in granzyme B (associated with active killer T cells), IFN-γ, TNF-α, etc., signal a pro-inflammatory response and role in overcoming tumor immune suppression Safety Safety results (Arms 1 & 2)1 24/50 (48%) of patients experienced grade 3 and higher adverse events 2/50 (4%) experienced grade 4 adverse events

Phase 2 Results in Recurrent Metastatic ICI Refractory HPV-Positive Cancer (CPS>0; PD-L1 agnostic) Plot Includes Published Data in HSNCC NCI-led Triple Combination: PDS0301 + PDS0101 + ICI Advanced HPV16-Positive ICI Refractory Cancer Patients 1Published results for ICI monotherapy <10%, Strauss J, et al. J Immunother Cancer 2020;8:e001395. doi:10.1136/jitc-2020-001395 2Pestana RC et al. Oral Oncology 2020;101:104523. https://doi.org/10.1016/j.oraloncology.2019.104523 N=29 Published median OS data in ICI refractory head and neck cancer population 8.2 months2 Objective Response (ORR) in High Dose PDS0301 Group = 63% (5/8)* 1 No controlled or comparative studies have been conducted between immune checkpoint inhibitors and PDS0101 2

Antibody-Drug Conjugate (PDS0301) Pipeline Reference: Data on file. Candidate/ Study Indication Combination PC P1 P2 P3 R Partner(s) IIT Studies PDS0301/ NCI-led Triple Combination HPV-positive anal, cervical, head and neck, penile, vaginal, vulvar cancers Arm 1: ICI naive Arm 2: ICI refractory PDS0101 & ICI IIT Studies PDS0301 Advanced Kaposi Sarcoma Monotherapy PDS0301 Metastatic Castration sensitive and Castration Resistant Prostate Cancer Docetaxel PDS0301 Localized High and Intermediate Risk Prostate Cancer Radiation Therapy PDS0301 ICI Refractory HPV-related, colon and small-bowel cancer HDAC Inhibitor Partner Co-Funded

3Q23 4Q23 1Q24 2Q24 3Q24 PDS0101 Submit IND with FDA for registrational trial (VERSATILE-003) Anticipate ICI naïve/refractory data – KOL Event (VERSATILE-002) Updated OS data from PDS0101-PDS0301 based triple combination Immune response data (VERSATILE-002) (ESMO 2023) Initiate registrational trial (VERSATILE-003) Anticipate updated data (IMMUNOCERV) (ASTRO 2023) Anticipate preliminary efficacy data (Mayo Clinic) Final data VERSATILE-002 PDS0301 Interim safety and immune data (PDS0301 + docetaxel) (Cytokines 2023) PDS0103 Estimated IND filing in MUC1-related cancers PDS0202 Universal flu preclinical ferret data (ESWI 2023) Projected Milestones Through 3Q24 Investigator-initiated trials and data read-outs are outside the control of PDS Biotech.
Infectimune® Infectious Disease Platform

License agreement with University of Georgia for proprietary influenza antigens Top-line preclinical data announced; effective delivery of flu proteins activate the critical immune signals necessary to generate neutralizing antibody responses to all flu strains tested in animals Preclinical data presented at the Ninth ESWI Influenza Conference and 41st Annual meeting of the American Society Virology Meeting Universal Flu Market Opportunity in 2021 PDS0202: Universal Influenza Vaccine with Potential to Revolutionize Protection Against Influenza Reference: Ross T. and Woodward J. et al. evaluation of the PDS0202 (Infectimune® + COBRA) Universal flu formulation. Universal Influenza Vaccines $7 Billion

PDS0202 Promotes Induction of Antibodies that Neutralize a Broad Range of Influenza Viral Strains in Preclinical Studies Proprietary Computationally Designed Influenza Protein Studies in Ferrets – Model Viewed as Gold Standard in Influenza Research

PDS0202 Promotes Induction of Antibodies and T Cells that Prevent Viral Replication in Lungs in Preclinical Studies Proprietary Computationally Designed Influenza Protein Studies in Ferrets – Model Viewed as Gold Standard in Influenza Research

PDS0101 to enter Phase 3 registrational trial in 2023 to treat recurrent or metastatic, HPV16-positive head and neck squamous cell cancer (HNSCC) Fast Track Designation PDS0101 addresses large and growing market with significant unmet need Transformational data generated with PDS0101 and PDS0301 in multiple Phase 2 clinical studies Financial: Cash as of June 30, 2023 - $60.6M cash runway for the approximately next 12 months with initiation of a registrational trial in 2023 Executive Summary: Positioned for Market Leadership Company Overview T cell activating platforms and antibody conjugated immuno-cytokine platform to develop safer, more effective and longer lasting cancer immunotherapies 1 2 3 4

Thank you

Appendix

FDA Requires OS as Primary Endpoint PFS and ORR will not support a BLA approval From FDA’s Clinical Trial Endpoints for the Approval of Cancer Drugs and Biologics White Paper1: “In the 1970’s, the FDA usually approved cancer drugs based on objective response rate (ORR), determined by tumor assessments from radiological tests or physical examinations. In the early 1980s, after discussion with the Oncologic Drugs Advisory Committee (ODAC), the FDA determined that cancer drug approval should be based on more direct evidence of clinical benefit, such as improvement in survival, improvement in a patient’s quality of life, improved physical functioning, or improved tumor-related symptoms. These benefits may not always be predicted by, or correlate with, ORR.” FDA Quote from Minutes of PDS Biotech End of Phase 2 Meeting with the FDA: “If Study PDS0101-HNC-301 is to be conducted with registrational intent, overall survival (OS) should be the primary endpoint, particularly given that recent recently approved immunotherapies for HNSCC have demonstrated in median OS but not always in PFS.” 1https://www.fda.gov/media/71195/download

VERSATILE-002 ICI Naïve Key Demographics and Treatment Exposure Majority of Patients Are CPS 1-19 Demographic ITT Population (N=55) mITT Population (N=52) Age, Median (Min, Max) 64.0 (46, 83) 64.0 (46, 83) Sex, n (%) Male Female 51 (92.7) 4 (7.3) 48 (92.3) 4 (7.7) Race, n (%) American Indian or Alaska Native Asian Black or African American Pacific Islander White Other 0 1 (1.8) 1 (1.8) 0 52 (94.5) 1 (1.8) 0 1 (1.9) 1 (1.9) 0 49 (94.2) 1 (1.9) ECOG, n (%) 0 1 32 (58.2) 23 (41.8) 29 (55.8) 23 (44.2) CPS, n (%)* <1 1–19 ≥20 0 33 (60.0) 22 (40.0) 0 31 (59.6) 21 (40.4) Data on File. 08/02/23 Data Cut. Treatment Exposure(ITT Population) Median number of PDS0101 doses: 4 (range 1–5) 72.7% received ≥4 doses25.5% received 5 doses (5th dose is 6 months after dose 4) Median number of KEYTRUDA® doses: 7 (range 1–33) 32.7% received ≥10 doses

Expanding Evidence of Consistent and Durable PDS0101 Clinical Results Across Multiple Phase 2 Trial Indications PDS0101 is an HPV16-targeted immunotherapy PDS0101 is providing strong proof of concept data for the Versamune® technology platform Efficacy data in >90 patients to date Strong agreement between preclinical and clinical results Versamune® mechanism of action shows clear translation between preclinical and human results Anti-tumor responses and biomarker data show strong correlation across all types of HPV-positive cancer and at all stages of the disease Safety data in approximately 130 patients to date Safety data, anti-tumor responses and patient survival suggest that the Versamune® based therapies such as PDS0101 could be ideal candidates for combination oncology treatments

HNSCC is a Devastating Group of Cancers Reference: Noseyaba et al. 2018. Cancer. Suicide Risk Among Cancer Survivors: Head and Neck Versus Other Cancers https://virologyj.biomedcentral.com/articles/10.1186/s12985-021-01688-9 https://www.cdc.gov/cancer/hpv/basic_info/hpv_oropharyngeal.html *Human Papillomavirus Oral and PharyngealCancers Genotype of *HPV-Positive Oral and Pharyngeal Cancer Paranasalsinuses Nasopharynx Oropharynx Hypopharynx Larynx Pharynx Tongue Salivaryglands OralCavity NasalCavity

Antibody Drug Conjugate IL-12: PDS0301 Targets Tumors and Enhances T Cell Infiltration and Proliferation in the Tumor Tumor Site Injection Site Non-Immunogenic (Cold Tumor) Immunogenic (Hot Tumor) Tumor Site PDS0301 travels direct to tumor Increased tumor inflammation from PDS0301 PDS0301 increases T cell infiltration and expansion in tumor CD8 T cells kill tumor cells 1 2 3 4

PDS0101: A Novel Investigational HPV-Targeted Immunotherapy PDS0101 is given by subcutaneous injection and stimulates a potent targeted T cell attack against HPV-positive cancers Interim VERSATILE-002 data suggests PDS0101 generates clinically effective immune responses The combination of PDS0101 with KEYTRUDA® has demonstrated a favorable safety profile to date PDS0101 with KEYTRUDA® demonstrates significant disease control by shrinking tumors, delaying disease progression and prolonging survival

Antibody Drug Conjugate IL-12 (PDS0301) A novel investigational tumor-targeting Interleukin 12 (IL-12) that enhances the proliferation, potency and longevity of T cells in the tumor microenvironment Together with Versamune® based immunotherapies PDS0301 works synergistically to promote a targeted T cell attack against cancers PDS0301 is given by a simple subcutaneous injection Clinical data suggest the addition of PDS0301 to Versamune® based immunotherapies demonstrate disease control by shrinking tumors and prolonging survival in recurrent or metastatic cancers with poor survival prognosis