Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
Related financial report
PDSB similar filings
- 9 May 24 Other Events
- 8 May 24 Other Events
- 2 May 24 PDS Biotech Appoints Stephan Toutain as Chief Operating Officer
- 27 Mar 24 PDS Biotech Announces Clinical Strategy Update and Reports Full Year 2023 Financial Results
- 13 Mar 24 Patent for composition of matter and use of Infectimune® based influenza vaccines granted in the United States
- 22 Jan 24 PDS Biotech Announces Retirement of Lauren V. Wood, M.D., and Appointment of Kirk V. Shepard, M.D., as Chief Medical Officer
- 4 Dec 23 Other Events
Filing view
External links
Exhibit 99.2

Transforming How the Immune System Targets and Fights Cancer NASDAQ: PDSB March 2024

Forward-Looking Statement This communication contains forward-looking statements (including within the meaning of Section 27E of the United States Securities Exchange Act of 7934, as amended, and Section 27A of the United States Securities Act of 7933, as amended) concerning PDS Biotechnology Corporation (the "Company") and other matters. These statements may discuss goals, intentions and expectations as to future plans, trends, events, results of operations or financial condition, or otherwise, based on current beliefs of the Company's management, as well as assumptions made by, and information currently available to, management. Forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions and include words such as "may" "will" "should" "would" "expect" "anticipate" "plan" "likely" "believe" "estimate" "project“ "intend," "forecast," "guidance", "outlook" and other similar expressions among others. Forward-looking statements are based on current beliefs and assumptions that are subject to risks and uncertainties and are not guarantees of future performance. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: the Company's ability to protect its intellectual property rights; the Company's anticipated capital requirements, including the Company's anticipated cash runway and the Company's current expectations regarding its plans for future equity financings; the Company's dependence on additional financing to fund its operations and complete the development and commercialization of its product candidates, and the risks that raising such additional capital may restrict the Company's operations or require the Company to relinquish rights to the Company's technologies or product candidates; the Company's limited operating history in the Company's current line of business, which makes it difficult to evaluate the Company's prospects, the Company's business plan or the likelihood of the Company's successful implementation of such business plan; the timing for the Company or its partners to initiate the planned clinical trials for PDS01ADC, PDS0101 and other Versamune® and lnfectimune® based product candidates; the future success of such trials; the successful implementation of the Company's research and development programs and collaborations, including any collaboration studies concerning PDS01ADC, PDS0101 and other Versamune® and lnfectimune® based product candidates and the Company's interpretation of the results and findings of such programs and collaborations and whether such results are sufficient to support the future success of the Company's product candidates; the success, timing and cost of the Company's ongoing clinical trials and anticipated clinical trials for the Company's current product candidates, including statements regarding the timing of initiation, pace of enrollment and completion of the trials (including the Company's ability to fully fund its disclosed clinical trials, which assumes no material changes to the Company's currently projected expenses), futility analyses, presentations at conferences and data reported in an abstract, and receipt of interim or preliminary results (including, without limitation, any preclinical results or data), which are not necessarily indicative of the final results of the Company's ongoing clinical trials; any Company statements about its understanding of product candidates mechanisms of action and interpretation of preclinical and early clinical results from its clinical development programs and any collaboration studies; to aid in the development of the Versamune® platform; and other factors, including legislative, regulatory, political and economic developments not within the Company's control. The foregoing review of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors included in the Company's annual, quarterly and periodic reports filed with the SEC. The forward-looking statements are made only as of the date of this press release and, except as required by applicable law, the Company undertakes no obligation to revise or update any forward-looking statement, or to make any other forward-looking statements, whether as a result of new information, future events or otherwise. Versamune® and lnfectimune® are registered trademarks of PDS Biotechnology Corporation. KEYTRUDA® is a registered trademark of Merck Sharp and Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.

Late-Stage Head and Neck Cancer Program as Value Catalyst Pivotal trial planned for PDS01ADC + Versamune® HPV (PDS0101) + KEYTRUDA® in head and neck cancer High-Value Lead Program PDS01ADC + Versamune® disrupts tumor’s inside defenses, and generates potent, targeted killer T-cell attack from outside Durable Phase 2 survival and ORR data Novel Investigational “Inside-Outside” MOA PDS01ADC safety demonstrated in >300 patients Versamune® HPV administered to >100 HNSCC patients Robust Phase 2 Data in 400+ Patients Cash runway into Q4 2025 (without pivotal trial)1 Financials 1. Company’s 10-K for year ended 12/31/2023 includes going concern opinion. Cash runway estimate based on currently available cash resources and cash flow projections and assumes no initiation of pivotal trial and Company debt not being called by lenders.

Strategy Addresses Why Immunotherapies Fail in Solid Tumors References: Darvin et al. Immune Checkpoint Inhibitors, Recent Progress and Potential Biomarkers; Experimental & Molecular Medicine (2018) 50:165. Chen, D. S. & Mellman, I. Elements of cancer immunity and the cancer–immune set point. Nature 541, 321 (2017). Two Critical Limitations Remain Inability to generate the right type and quantity of effective tumor-infiltrating and tumor-killing T cells Inadequate T Cell Response Immune-Desert Tumors: Lack T cells because T cells don’t get activated or recognize the cancer Immune-Excluded Tumors: Contain immune suppressive cytokines and inhibitory factors that prevent T cell infiltration TME Prevents Immunogenicity TME = Tumor Microenvironment

Infiltrates TME to Suppress the Tumor’s Defenses & Promotes T Cell Infiltration/Immunogenicity Combination Platform Enables Inside-Outside Attack to Address Limits Immuno-cytokine IL-12 Fused Antibody Drug Conjugate PDS01ADC First immuno-cytokine antibody drug conjugate NHS76 (Tumor Necrosis Targeting Ab – Binds to exposed DNA) De-immunized Junction IL-12 (p40 clipping-resistant) IL-12 (p40 clipping-resistant) IL-12 fused antibody drug conjugate TME = Tumor Microenvironment Induces Right Type & Quantity of Powerful Tumor-Targeting Killer T Cells Versamune® Water-insoluble fatty acids/ hydrocarbon chains Water-soluble, positively charged head-group coats particle surface Immunologically active R-enantiomer of 1,2-dioleoyl-trimethyl-ammonium (R-DOTAP) Inside Outside
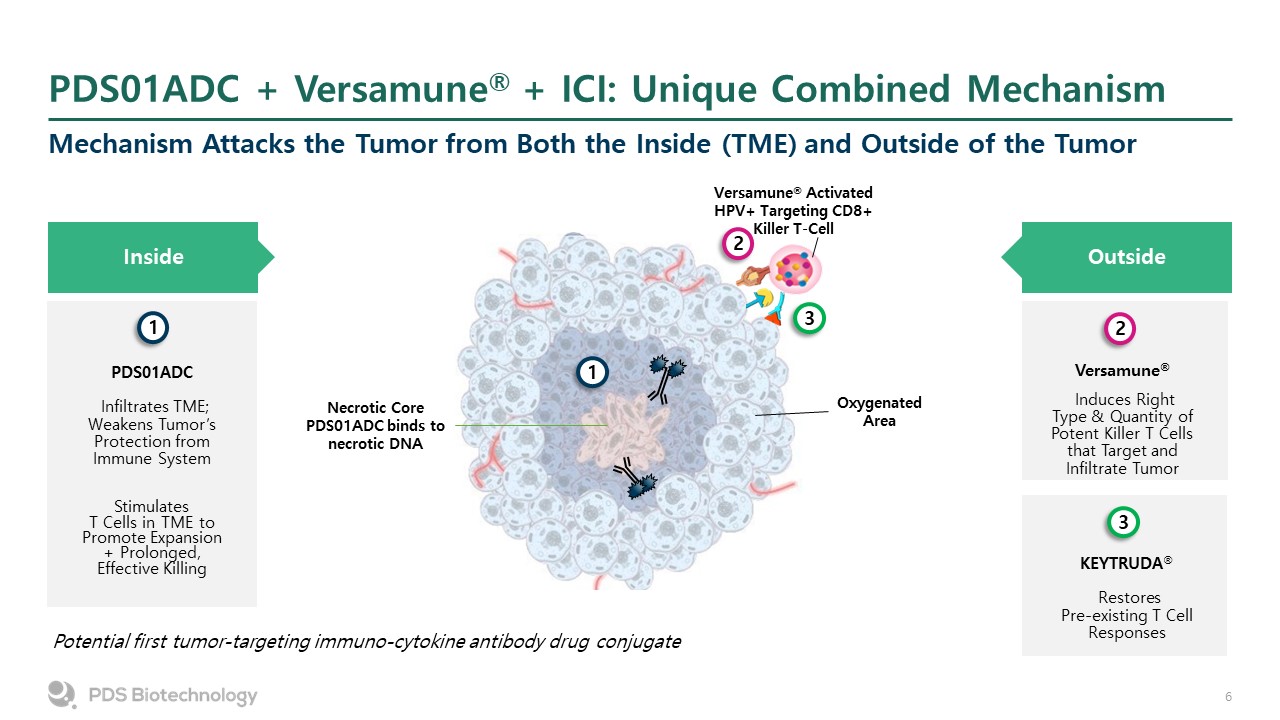
PDS01ADC + Versamune® + ICI: Unique Combined Mechanism Mechanism Attacks the Tumor from Both the Inside (TME) and Outside of the Tumor Inside 1 PDS01ADC Infiltrates TME; Weakens Tumor’s Protection from Immune System StimulatesT Cells in TME to Promote Expansion + Prolonged, Effective Killing Versamune® Induces Right Type & Quantity of Potent Killer T Cells that Target and Infiltrate Tumor Outside 2 KEYTRUDA® Restores Pre-existing T Cell Responses 3 Potential first tumor-targeting immuno-cytokine antibody drug conjugate 1 2 3 Necrotic Core PDS01ADC binds to necrotic DNA Oxygenated Area Versamune® Activated HPV+ Targeting CD8+ Killer T-Cell

PDS01ADC and Versamune® Have Broad Therapeutic Potential Synergistic Effect With SoC Modalities Across a Spectrum of Tumors PDS01ADC Versamune® Conjugate limits systemic toxicity of IL-12 Activates/expands T cells in tumor & limits T cell exhaustion Changes tumor to become more permissive to T cell attack Designed to deliver and sustain IL-12 in tumor Activates the right type of multifunctional CD8 killer T cells Promotes the right quantity and potency of T cells Promotes a long-lasting memory T cell response Designed to train T cells to recognize the cancer

Patients Had Sustained Tumor Shrinkage, Objective Response Rate ICI Resistant Cancer 1. Burtness B et al., Lancet. 2019; 394:1915-1928. 2. Triple combination Phase 2 trial (NCT04287868, Investigator assessment (11/2023) 1. Strauss J et al, Journal for ImmunoTherapy of Cancer 2020;8:e001395. 2. INTERLINK-1 (NCT04590963) 3. Triple combination Phase 2 trial (NCT04287868), Investigator assessment (11/2023). ICI Naïve Cancer Optimal Dose PDS01ADC % of Patients with Sustained Tumor Shrinkage of ≥30% No controlled or comparative studies have been conducted between PDS0101 + PDS01ADC and checkpoint inhibitors or Chemotherapy. * *38% Complete Response (CR)

Triple Combination Has Shown Compelling Survival Data Proof-of-Concept in ICI-resistant Cancer Validates MOA and Preclinical Results One-Year Survival Median Overall Survival 1. Strauss J et al, Journal for ImmunoTherapy of Cancer 2020;8:e001395. 2. Pestana, et al. Oral Oncology. 2022:104523. 3. Triple combination Phase 2 trial (NCT04287868), Investigator assessment (11/2023). Bila M, et al. Frontiers in Oncology. Jan 2022;12:761428. Pestana, et al. Oral Oncology. 2022:104523. Triple combination Phase 2 trial (NCT04287868), Investigator assessment (11/2023). No controlled or comparative studies have been conducted between PDS0101 and checkpoint inhibitors or EGFR Inhibitor or Chemotherapy or PDS01ADC.
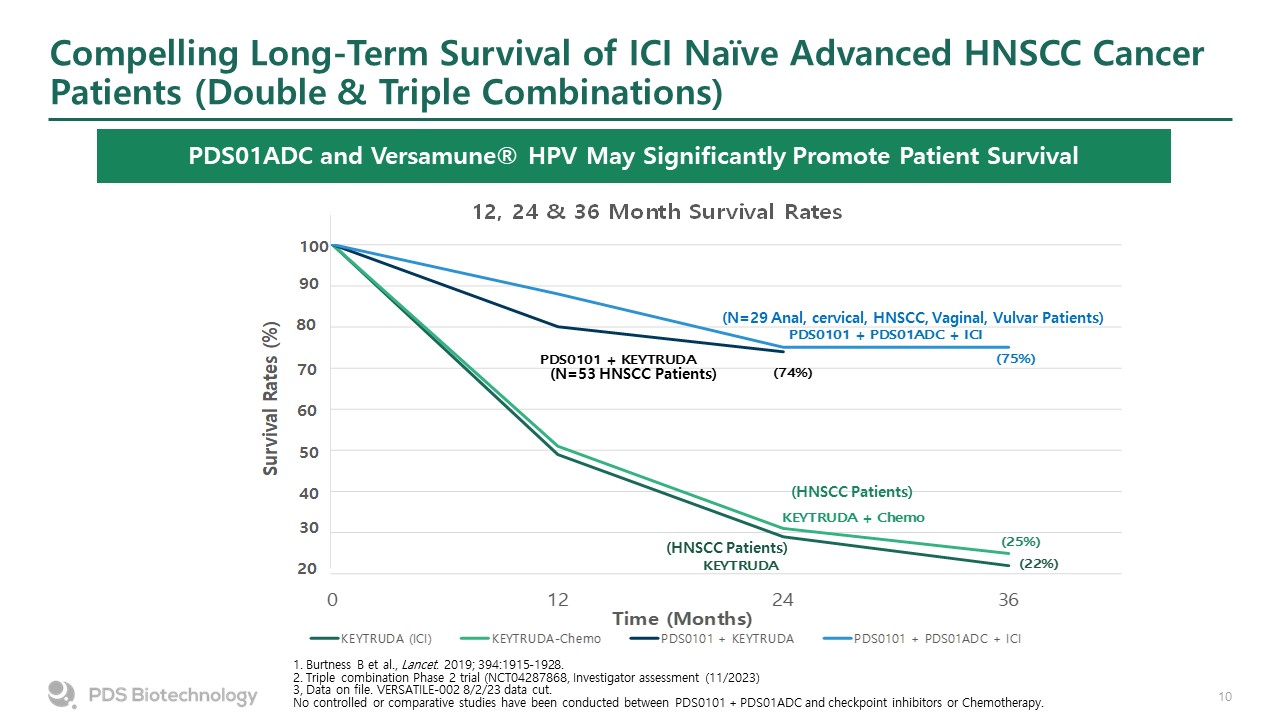
Compelling Long-Term Survival of ICI Naïve Advanced HNSCC Cancer Patients (Double & Triple Combinations) PDS01ADC and Versamune® HPV May Significantly Promote Patient Survival (N=53 HNSCC Patients) (HNSCC Patients) (HNSCC Patients) (N=29 Anal, cervical, HNSCC, Vaginal, Vulvar Patients) 1. Burtness B et al., Lancet. 2019; 394:1915-1928. 2. Triple combination Phase 2 trial (NCT04287868, Investigator assessment (11/2023) 3, Data on file. VERSATILE-002 8/2/23 data cut. No controlled or comparative studies have been conducted between PDS0101 + PDS01ADC and checkpoint inhibitors or Chemotherapy.

Proof-of-Concept Results Indicate Favorable Tolerability 48% Had Grade 3 TRAEs, 4% Grade 4 Safety Population: All enrolled subjects who received at least 1 dose of any drug National Cancer Institute. (2023). Combination Immunotherapy in Subjects With Advanced HPV Associated Malignancies. [Data set] Preferred Term n (%) Myocarditis 1 (2) Anemia 15 (30) HLH* 1 (2) Flu-like Symptoms 1 (2) Lymphopenia 3 (6) CPK Elevation 1 (2) Preferred Term n (%) Leukopenia 1 (2) Neutropenia 1 (2)** Hematuria 5 (10) GI Bleeding 2 (4) AST/ALT Elevation 4 (8)*** Mucositis 1 (2) Grade 3/4 Adverse Events (AE) Grade 3/4 Adverse Events (cont.) *HLH, hemophagocytic lymphohistiocytosis **Grade 4 TRAE ***1 patient had Grade 4 TRAE

Program Is Supported and Validated by Robust Clinical Data Experience in More Than 430 Patients PDS01ADC + Versamune® HPV + Bintrafusp alfa (triple) and Versamune® HPV + KEYTRUDA® (double) administered to 110+ head & neck cancer patients to date HNSCC Patient Exposure Across Product Portfolio Well-tolerated to date at 3.0 mg per dose every 3 weeks 170+ Patients Treated to Date Versamune® HPV Acceptable tolerability and safety profile to date at 8.0, 12.0 and 16.8 ug/kg every 2 or 4 weeks 300+ Patients Treated to Date PDS01ADC

HNSCC: Devastating Cancers with High Prevalence and Mortality References: Johnson, D.E., Burtness, B., Leemans, C.R.et al.Head and neck squamous cell carcinoma.Nat Rev Dis Primers6, 92 (2020) Noseyaba et al. 2018. Cancer. Suicide Risk Among Cancer Survivors: Head and Neck Versus Other Cancers https://virologyj.biomedcentral.com/articles/10.1186/s12985-021-01688-9 https://www.cdc.gov/cancer/hpv/basic_info/hpv_oropharyngeal.html *Human Papillomavirus Oral and PharyngealCancers (~40% of HNSCC) Genotype of *HPV-Positive Oral and Pharyngeal Cancer Paranasalsinuses Nasopharynx Oropharynx Hypopharynx Larynx Pharynx Tongue Salivaryglands OralCavity NasalCavity Increasing Incidence Driven Largely by HPV16+
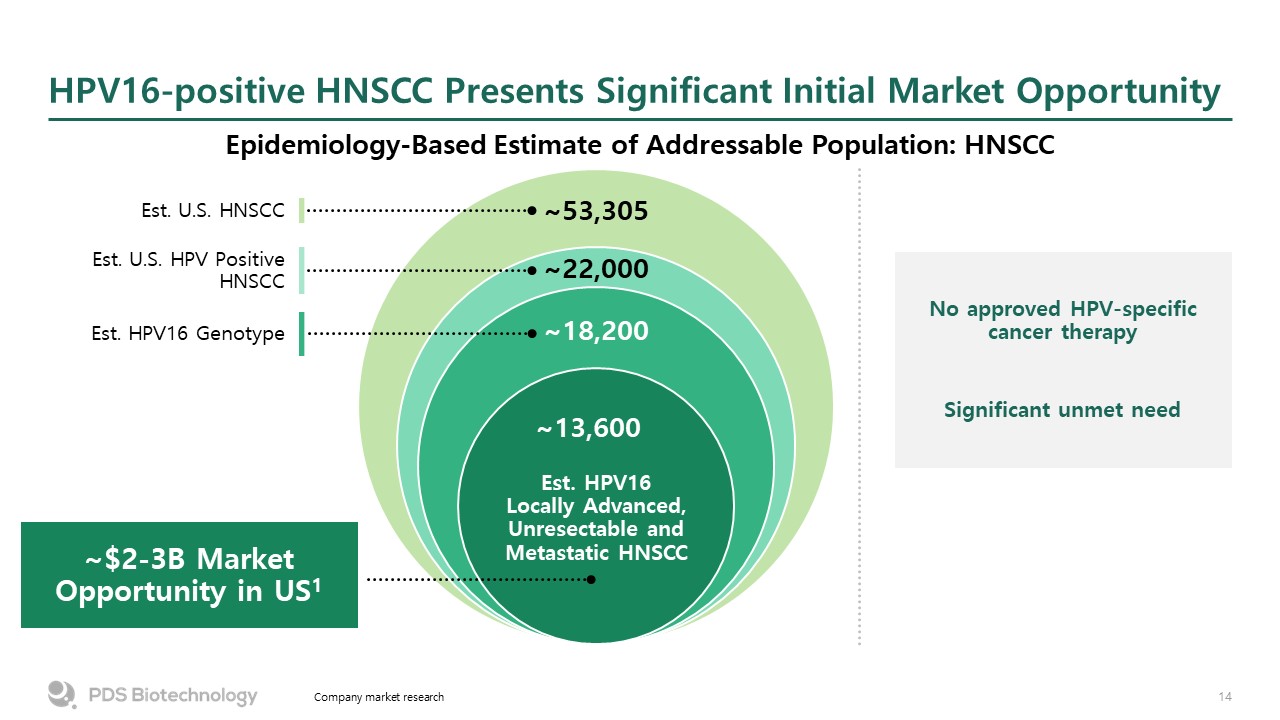
Est. HPV16Locally Advanced,Unresectable andMetastatic HNSCC HPV16-positive HNSCC Presents Significant Initial Market Opportunity Epidemiology-Based Estimate of Addressable Population: HNSCC Company market research ~13,600 ~18,200 ~22,000 ~53,305 Est. U.S. HPV Positive HNSCC Est. U.S. HNSCC Est. HPV16 Genotype ~$2-3B Market Opportunity in US1 No approved HPV-specific cancer therapy Significant unmet need

Candidate/ Study Indication PC P1 P2 P3 Partner PDS01ADC + Versamune® HPV + ICI Recurrent or metastatic HPV16-positive HNSCC PDS01ADC + Versamune® MUC1 + ICI (Phase 1/2 anticipated 2024) Recurrent or metastatic colorectal cancer Versamune® HPV + ICI (KEYTRUDA) Recurrent or metastatic HPV16-positive HNSCC PDS01ADC + Versamune® Agent + SoC* Immune Deficiency and Infectious Diseases* Pipeline Continues to Validate Platforms, Drive Future Opportunities Versamune® Initiate pivotal study in HNSCC: PDS01ADC+Versamune® HPV+ KEYTRUDA® Triple Combination - 2024 Update on regulatory confirmation of potentially registrational study - Q2/Q3-2024 Readout of VERSATILE-002 Phase 2 in HNSCC (Versamune® HPV + KEYTRUDA®) – April 2024 Confirm path to triple combination Phase 1/2 study in r/m colorectal cancer to be performed under funded collaboration with NCI - Q4-2024 Upcoming Milestones

Veteran New Leadership to Execute Strategy Record of Execution in Development, Commercialization of Leading Pharmaceutical Products Frank Bedu-Addo, PhD Chief Executive Officer Senior executive experience with management of strategy and execution at both large pharma and biotechs Notable drug development: Abelcet® (Liposome Company/ Elan) PEG-Intron® (Schering-Plough/ Merck) Lars Boesgaard Chief Financial Officer 20 years of financial leadership roles in healthcare Former Chief Financial Officer of publicly traded healthcare and biotech companies Kirk Shepard, M.D. Chief Medical Officer US board-certified medical oncologist and hematologist 30+ years of experience in the pharmaceutical industry Gregory Conn, PhD Chief Scientific Officer Co-founder 35 years of drug development experience In-depth experience with biotech drug discovery, product development and manufacturing

Thank You NASDAQ: PDSB