Exhibit 99.3
|
Fujifilm’s Initiatives in
Regenerative Medicine
Shigetaka Komori Chairman and CEO
FUJIFILM Holdings Corporation
March 30, 2015
|
Priority Business: Healthcare
Expand business with focus on six core business areas
[Graphic Appears Here]
1
|
Comprehensive Healthcare Company
—\–h Diagnostics Treatment
Functional
X-Ray Imaging (FCR/DR/Film) Low Molecular Drugs
Cosmetics Business Business
Biopharmaceuticals
Perseus Proteomics Supplements Fujifilm Diosynth
Expansion Expansion Biotechnologies
Kalon
Radio Diagnostic
Endoscopes
Medicines
Regenerative Medicine
HairCare
Influenza Ultrasound Detection
Autologous Autologous
cultured cultured
epidermis cartilage
2
|
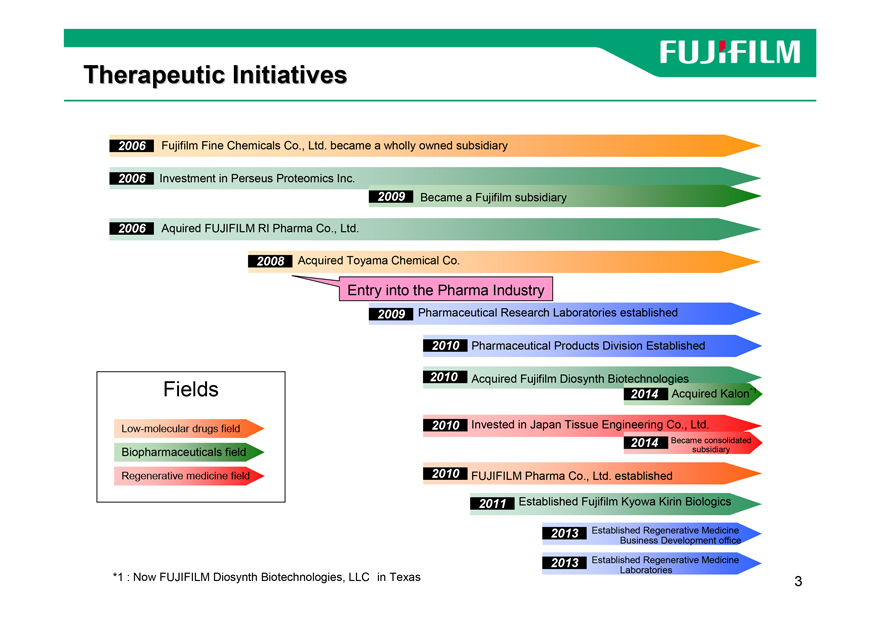
Therapeutic Initiatives
2006 Fujifilm Fine Chemicals Co., Ltd. became a wholly owned subsidiary
2006 Investment in Perseus Proteomics Inc.
2009 Became a Fujifilm subsidiary
2006 Aquired FUJIFILM RI Pharma Co., Ltd.
2008 Acquired Toyama Chemical Co.
Entry into the Pharma Industry
2009 Pharmaceutical Research Laboratories established
2010 Pharmaceutical Products Division Established
2010 Acquired Fujifilm Diosynth Biotechnologies
Fields 2014 Acquired Kalon*1
2010 Invested in Japan Tissue Engineering Co., Ltd.
Low-molecular drugs field
2014 Became consolidated
Biopharmaceuticals field subsidiary Regenerative medicine field 2010 FUJIFILM Pharma Co., Ltd. established 2011 Established Fujifilm Kyowa Kirin Biologics
2013 Established Regenerative Medicine Business Development office
2013 Established Regenerative Medicine Laboratories
*1 : Now FUJIFILM Diosynth Biotechnologies, LLC in Texas 3
|
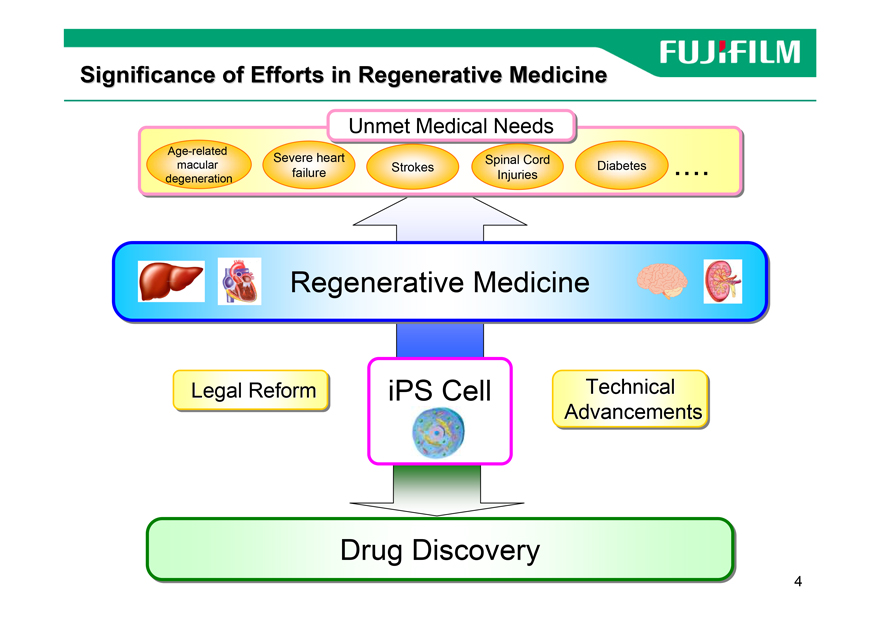
Significance of Efforts in Regenerative Medicine
Unmett Medicali l Needs
Age-related
Severe heart Spinal Cord
macular Strokes Diabetes
degeneration failure Injuries EEEE
Regenerative Medicine
Legal Reform iPS Cell Technical
Advancements
Drug Discovery
4
|
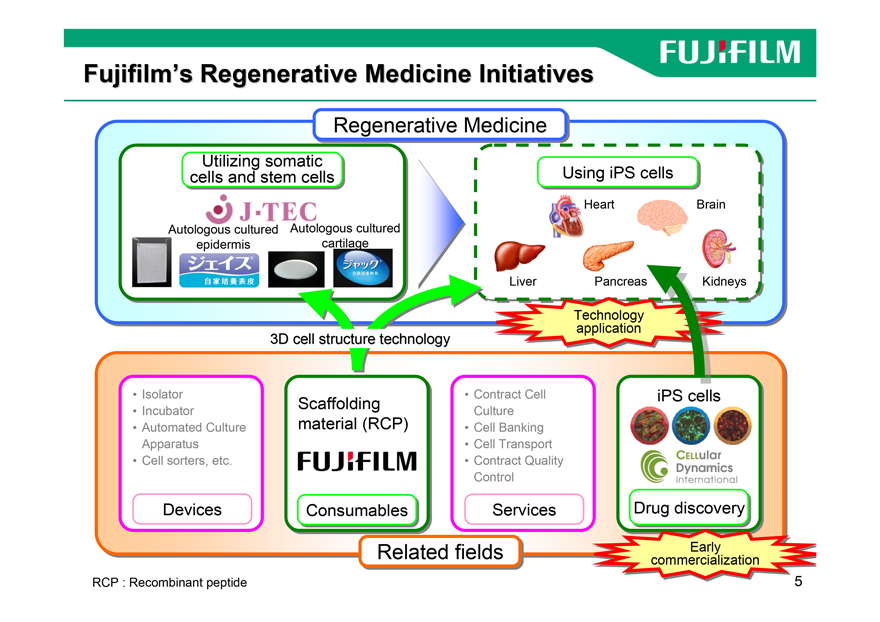
Fujifilm’s Regenerative Medicine Initiatives
Regenerativer ti Medicinei i
Utilizingtilizing somaticso atic
cellscells andand stemste cellscells iPS×–E,ðŽg,¤ Usingsing iPSi cellscells
Heart Brain
Autologous cultured Autologous cultured
epidermis cartilage
Liver Pancreas Kidneys
TechnologyTechnology
applicationapplication
3D cell structure technology
• Isolator Scaffolding • Contract Cell iPSi cellscells
Incubator Culture
• Automated Culture material (RCP) • Cell Banking
Apparatus • Cell Transport
• Cell sorters, etc. • Contract Quality
Control
Devices Consumablesonsu ables Services Drugrug discoverydiscovery
Relatedl t fieldsfi l commercializationcommercialization EarlyEarly
RCP : Recombinant peptide 5
|
Acquisition of U.S.-based Cellular Dynamics International, Inc., a leading developer and manufacturer of iPS cells
Towards a new stage in the regenerative medicine business
|
Acquisition of Cellular Dynamics International, Inc. (USA), a leading developer and manufacturer of iPS cells
Yuzo Toda
Director, Senior Vice President
FUJIFILM Corporation
March 30, 2015
|
Cellular Dynamics International, Inc. Summary
Name: Cellular Dynamics International, Inc. (“CDI”)
Founded: 2004
Founded by world-renowned researcher, Dr. James Thomson of
University of Wisconsin-Madison, the first in the world to generate ES cells.
IPO: July 30, 2013, listed on NASDAQ
Location: Madison, Wisconsin, USA
Chairman and CEO: Robert J. Palay
Core product: Human iPSC derived cells from a single genetic background, e.g.
cardiomyocytes, neurons, hepatocytes, etc.(iCell® products),
Human iPSC’s and iPSC derived cells from custom genetic backgrounds
(MyCell® products)
Sales: $ U.S.16.7 million (FY 2014/12)
Number of employees: 155 (as of Dec. 31, 2014)
8
|
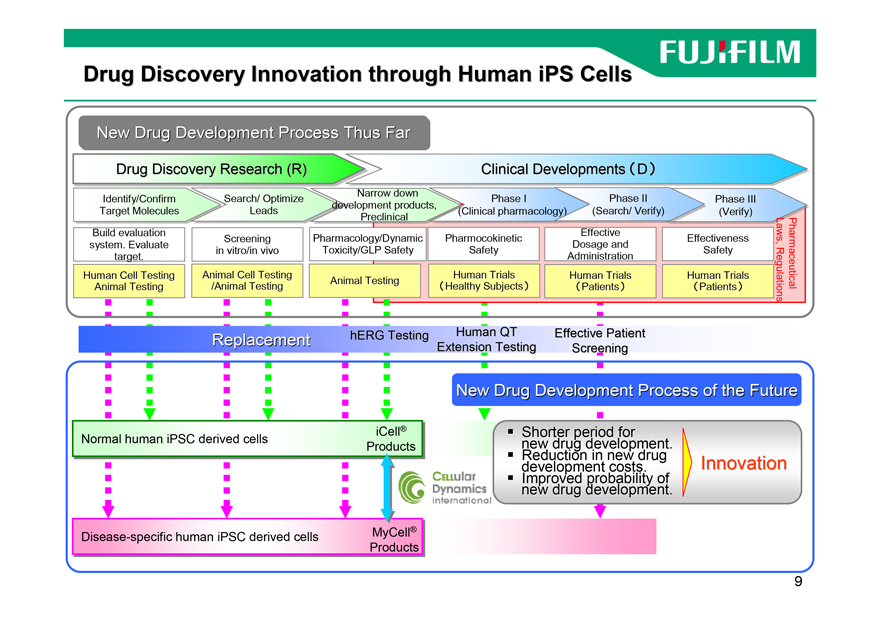
Drug Discovery Innovation through Human iPS Cells
New Drugr Developmentl t Processr Thus Farr
Drug Discovery Research (R) Clinical Developments (D)
Narrow down
Identify/Confirm Search/ Optimize Phase I Phase II Phase III
development products,
Target Molecules Leads Clinical pharmacolo(Search/ Verify)(Verify)
Preclinical L
Build evaluation Effective
Screening Pharmacology/Dynamic Pharmocokinetic Effectiveness aws,
system. Evaluate Dosage and
in vitro/in vivo Toxicity/GLP Safety Safety Safety
target. Administration
Human Cell Testing Animal Cell Testing Human Trials Human Trials Human Trials
Animal Testing
Animal Testing /Animal Testing (Healthy Subjects) (Patients) (Patients) Regulations
Replacement hERG Testing Human QT Effective Patient
Extension Testing Screening
New Drug Development Process of the Future
iCell® Normal human iPSC derived cells Products
? Shorter period for new drug development.
? Reduction in new drug Innovation development costs.
Improved probability of new drug development.
Disease-specific human iPSC derived cells MyCell®
Products
9
|
Strengths and Features of CDI
Merging University of Wisconsin-Madison’s iPS cell technology with CDI’s unique iPS cell culturing technology
Patent portfolio includes over 800 entities (includes licences and pending patents)
Awarded US patent covering automated production of iPSC’c
Rich product line from iPS cell-derived differentiated cells
Ready made iCell? : 12 types (incl. prototypes) such as cardiomyocytes, neurons, endothelial cells, dopaminergic neurons, hepatocytes
Custom made MyCell? : reprogramming, genetic engineering, and cell differentiation
Pioneer in iPS cell banking
Currently contracted to establish and bank iPS cells from over 3000 people via CIRM (USA)*1
2 cGMP HLA superdonor iPSC lines established
Applying iPS cells to Cell Therapy
12 types of iPS cell-derived differentiated cells can be used to treat over 50 diseases
Providing US NEI*2 with retinal pigmented cells for preclinical studies in the treatment of age-related macular degeneration
*1 : California Institute for Regenerative Medicine *2 : National Eye Institute 10
|
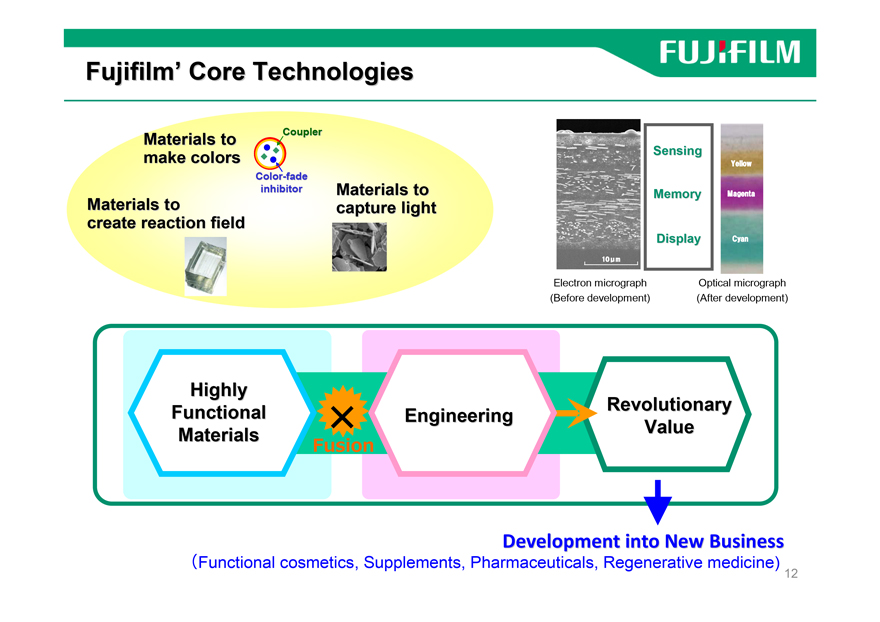
Fujifilm’ Core Technologies
Coupler
Materials to
make colors Sensing
Yellow
Color-fade
inhibitor Materials to Memory Magenta
Materials to capture light
create reaction field
Display Cyan
10ìm
Electron micrograph Optical micrograph
(Before development)(After development)
Highly
Functional Engineering Revolutionary
Materials × V,µ,¢‰¿’l Value
Fusion
Development into New Business
(Functional cosmetics, Supplements, Pharmaceuticals, Regenerative medicine)
12
|
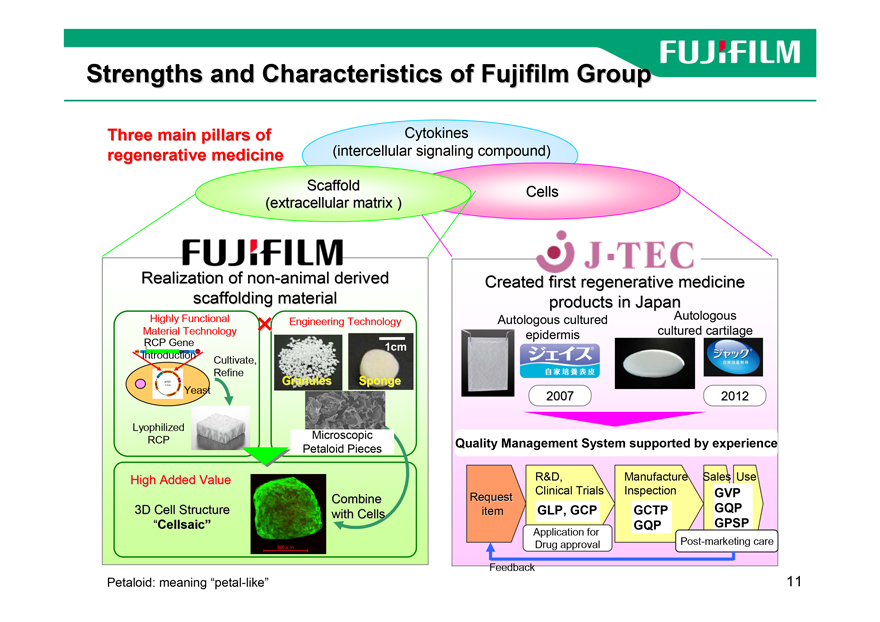
Strengths and Characteristics of Fujifilm Group
Three main pillars of Cytokines
regenerative medicine(intercellular signaling compound)
Scaffold
(extracellular matrix )
Realization of non-animal derived
scaffolding material
Highly Functional
Material Technology × Engineering Technology
RCP Gene 1cm
Introduction Cultivate,
Refine
Granules Sponge
Yeast
Lyophilized
RCP Microscopic
Petaloid Pieces
High Added Value
Combine
3D Cell Structure with Cells
“Cellsaic”
Petaloid: meaning “petal-like”
Cells
Created first regenerative medicine
products in Japan
Autologous cultured Autologous
epidermis cultured cartilage
2007 2012
Quality Management System supported by experience
R&D, Manufacture Sales Use
Request Clinical Trials Inspection GVP
item GLP, GCP GCTP GQP
GQP GPSP
Application for
Drug approval Post-marketing care
Feedback
11
|
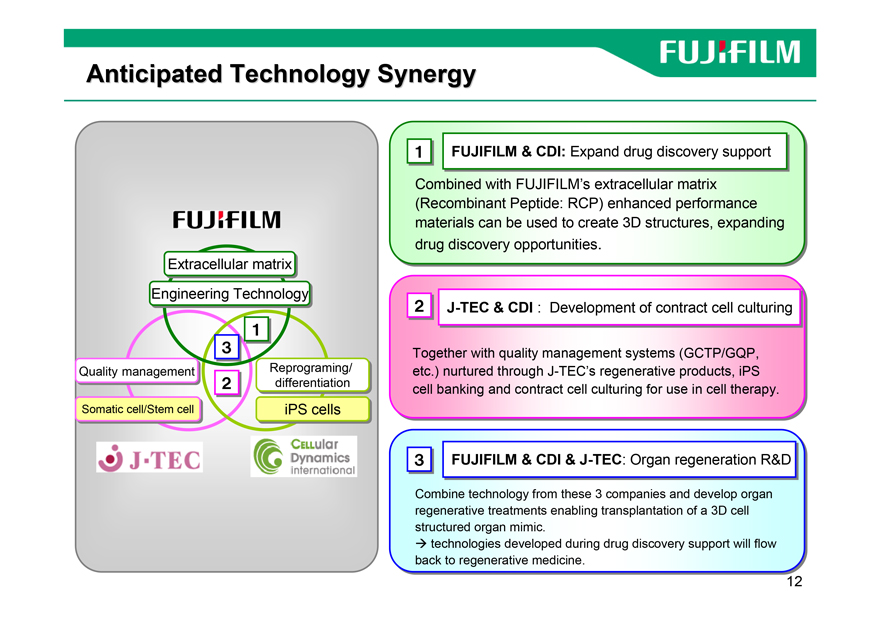
Anticipated Technology Synergy
ExtracellularExtracellular matrixmatrix
EngineeringEngineering TechnologyTechnology
1
2
QualityQuality managementmanagement Reprograming/Reprograming/
3 differentiationdifferentiation
SomaticSomatic cell/Stemcell/Stem cell cell iPSiPS cellscells
1 FUJIFILM & CDI: Expand drug discovery support
Combined with FUJIFILM’s extracellular matrix
(Recombinant Peptide: RCP) enhanced performance materials can be used to create 3D structures, expanding drug discovery opportunities.
2 J-TEC & CDI : Development of contract cell culturing
Together with quality management systems (GCTP/GQP, etc.) nurtured through J-TEC’s regenerative products, iPS cell banking and contract cell culturing for use in cell therapy.
3 FUJIFILM & CDI & J-TEC: Organ regeneration R&D
Combine technology from these 3 companies and develop organ regenerative treatments enabling transplantation of a 3D cell structured organ mimic.
_technologies developed during drug discovery support will flow back to regenerative medicine.
12
|
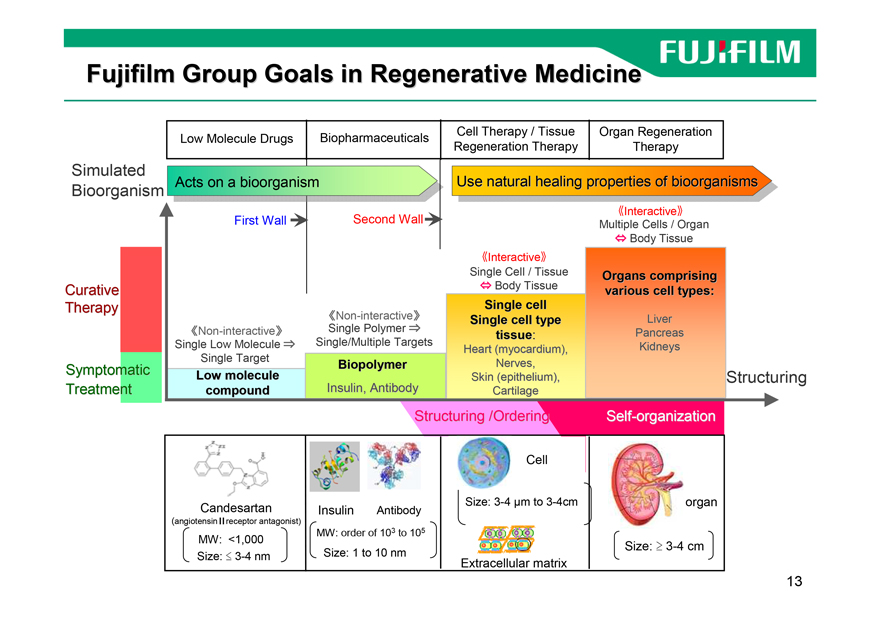
Fujifilm Group Goals in Regenerative Medicine
Low Molecule Drugs Biopharmaceuticals Cell Therapy / Tissue Organ Regeneration
Regeneration Therapy Therapy
Simulated
Acts on a bioorganism Use natural healing properties of bioorganisms
Bioorganism
sInteractivet
First Wall Second Wall Multiple Cells / Organ
? Body Tissue
sInteractivet
Single Cell / Tissue Organs comprising
Curative ? Body Tissue various cell types:
Therapy Single cell
sNon-interactivet Single cell type Liver
sNon-interactivet Single Polymer ? tissue: Pancreas
Single Low Molecule ? Single/Multiple Targets Heart (myocardium), Kidneys
Single Target
Symptomatic Biopolymer Nerves,
Low molecule Skin (epithelium), Structuring
Treatment compound Insulin, Antibody Cartilage
Structuring /Ordering Self-organization
Cell
Candesartan Insulin Antibody Size: 3-4 µm to 3-4cm organ
(angiotensin?receptor antagonist)
MW: order of 103 to 105
MW: <1,000
Size: ? 3-4 cm
Size: ? 3-4 nm Size: 1 to 10 nm
Extracellular matrix
13
|
Overview of the Acquisition
Method: Tender offer for common stock of CDI by a special purpose
company formed as a wholly-owned subsidiary of Fujifilm’s
U.S. holding company
(If necessary, following the tender offer, exercise of top-up
option to acquire new shares), followed by a short-form
merger
Price: USD 16.5 per share
Represents a 108% premium over the USD 7.94 closing
price on March 27.
Total value Approximately 307 million USD (fully-diluted basis)
Offer period: Commences within 5 business days after March 30, 2015
Ends 20 business days from commencement
(subject to extension)
14
|
CAUTIONARY STATEMENT ABOUT FORWARD-LOOKING STATEMENTS
This presentation contains certain statements which constitute “forward-looking statements”. These forward-looking statements may be identified by words such as ‘believes’, ‘expects’, ‘anticipates’, ‘projects’, ‘intends’, ‘should’, ‘seeks’, ‘estimates’, ‘future’ or similar expressions or by discussion of, among other things, strategy, goals, plans or intentions. The forward-looking statements involve risks and uncertainties that could cause actual results to differ materially from those expressed in the forward-looking statements. Many of these risks and uncertainties relate to factors that are beyond Fujifilm’s and CDI’s abilities to control or estimate precisely, such as future market conditions, the behaviors of other market participants, the effects of the transaction making it more difficult to maintain existing relationships with employees, customers or business partners, and other business effects, including the effects of industry, economic or political conditions, and therefore undue reliance should not be placed on such statements. Examples of forward-looking statements in this press release include, but are not limited to, statements regarding the proposed acquisition of CDI by Fujifilm, such as: the timing of the tender offer and the merger; results of the review of the transaction by regulatory agencies, and any conditions imposed in connection with consummation of the transaction; and satisfaction of various other conditions to the closing of the transaction. Actual results may differ materially from those in the forward-looking statements. For information regarding other related risks, please see the “Risk Factors” section of CDI’s filings with the Securities and Exchange Commission (the “SEC”), including its most recent filings on Form 10-K and Form 10-Q. CDI and Fujifilm assume no obligation to update these forward-looking statements, except as required pursuant to applicable law.
NOTE TO INVESTORS
The tender offer to purchase shares of CDI common stock referenced in this press release has not yet commenced, and this press release is neither an offer to purchase, nor a solicitation of an offer to sell, any securities. The tender offer to purchase shares of CDI common stock will be made only pursuant to a Tender Offer Statement on Schedule TO containing an offer to purchase, forms of letters of transmittal and other documents relating to the tender offer (the “Tender Offer Statement”), which Fujifilm will file with the SEC and mail to CDI stockholders. At the time the tender offer is commenced, CDI will file a Solicitation / Recommendation Statement on Schedule 14D-9 with respect to the tender offer (the “Recommendation Statement”). Investors and security holders of CDI are advised to read the Tender Offer Statement and Recommendation Statement carefully when they become available, before making any investment decision with respect to the tender offer because they will contain important information about the tender offer. Investors and security holders of CDI also are advised that they may obtain free copies of the Tender Offer Statement and other documents filed by Fujifilm with the SEC (when these documents become available) and the Recommendation Statement and other documents filed by CDI (when these documents become available) on the SEC’s website at http://www.sec.gov. In addition, free copies of the Tender Offer Statement and related materials may be obtained (when these documents become available) from Fujifilm’s website at http://www.fujifilmholdings.com/en/investors/index.html; and free copies of the Recommendation Statement and related materials may be obtained (when these documents become available) from CDI’s website at www.cellulardynamics.com.
15