 Yuzo Toda Yuzo Toda Director, Senior Vice President Director, Senior Vice President FUJIFILM Corporation FUJIFILM Corporation March 31, 2015 March 31, 2015 Fujifilm Group Fujifilm Group Exhibit 99.1 |
 1 1. About Fujifilm |
 2 Established January 20, 1934 President & COO President & COO Shigehiro Nakajima Shigehiro Nakajima Consolidated Revenue 2,440 billions of yen FY2014/3 2,480 billions of yen FY2015/3 (rev. Plan) Operating Income FY2014/3 140.8 billions of yen FY2015/3 (rev. Plan) 170 billions of yen No. of Group Companies 275 Japan 88, Others 187 FY2014/12 No. of Employees 78,595 FY2014/3 Chairman & CEO Shigetaka Komori Shigetaka Komori |
 3 |
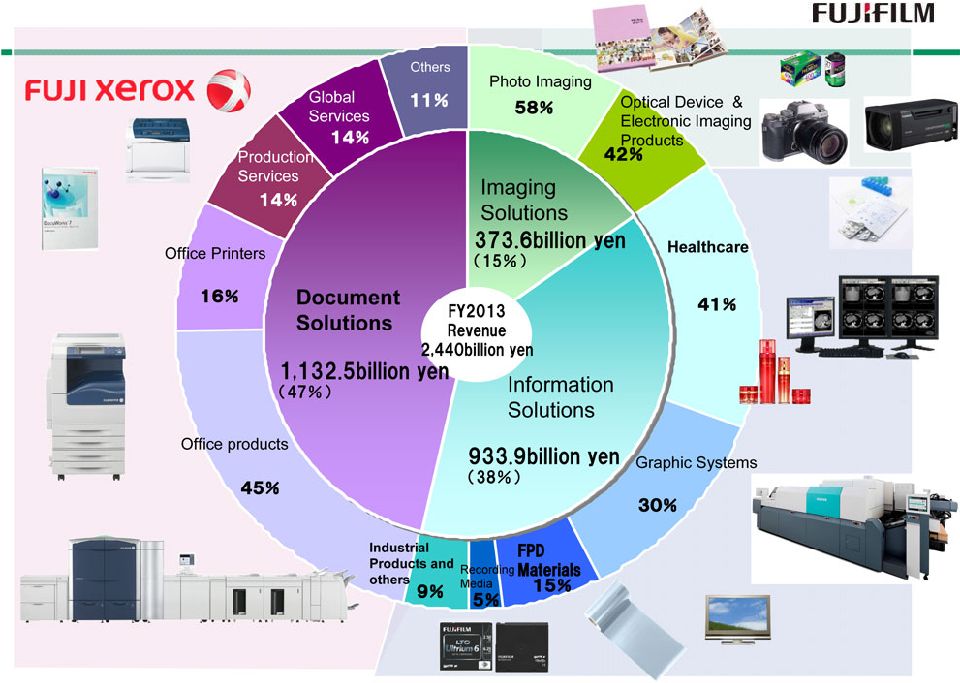
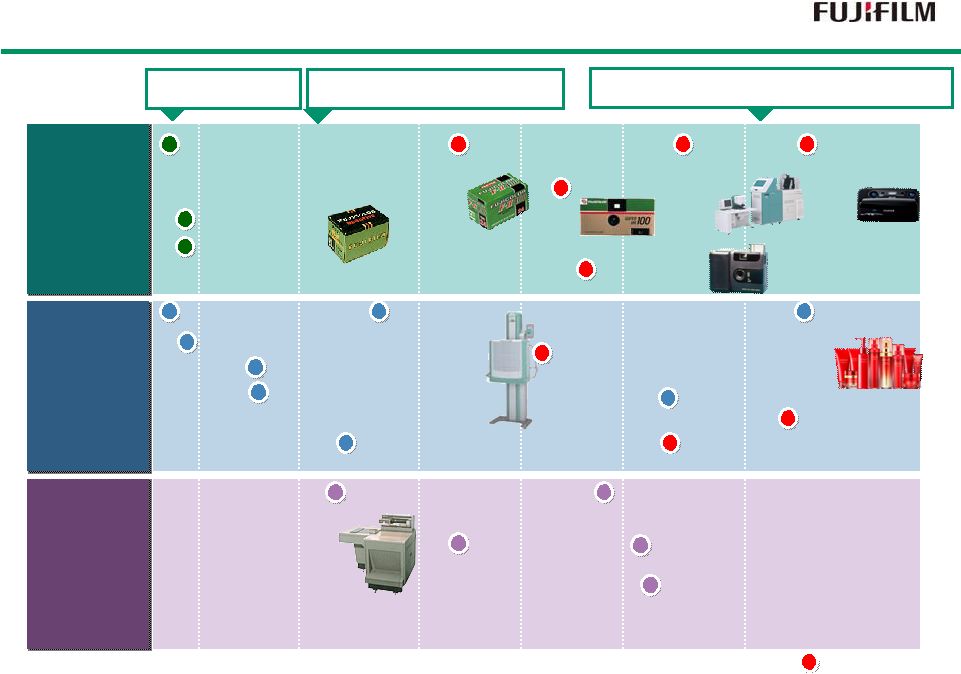 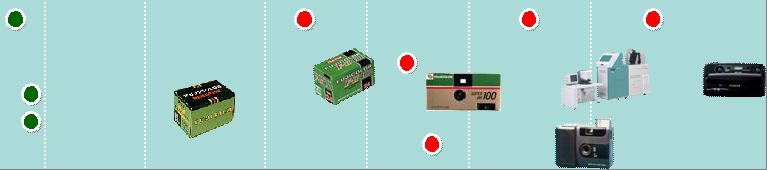  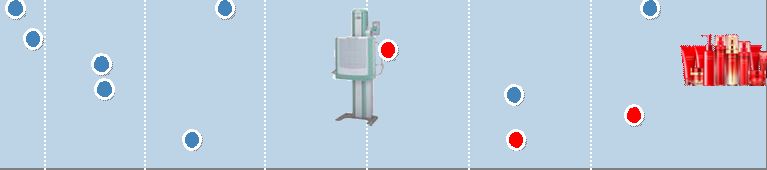    4 Imaging Solutions Imaging Solutions Information Solutions Information Solutions Document Solutions Document Solutions 1988 Digital Camera 1983 Computed Radiography FCR 1996 WV Film 1989 Digital Multi-Function Copier/Printer 1958 TAC Film 1962 Copier 1965 PS Plate 1975 Color Copier 1959 Video Tape 1936 X-Ray Film 1934 Photographic film Motion picture film 1948 Color Film 1996 Digital Mini-Lab 1992 Digital Color Copier 1965 Data Storage Media 1996 CTP Plate 1976 High-Speed Color Negative Film 1950 1960 1970 1980 1990 2000 1993 High-speed Digital Print Publishing System DocuTech 2004 Full Digital Endoscope Double balloon Endoscope World’s First 1986 Quick Snap 2009 3D- Digital Camera 2006 Skin Care Products 1948 Camera 1948 Recording Film 1934 Establishment 1962 Establishment of Fuji Xerox 2001 Fuji Xerox became a consolidated subsidiary |
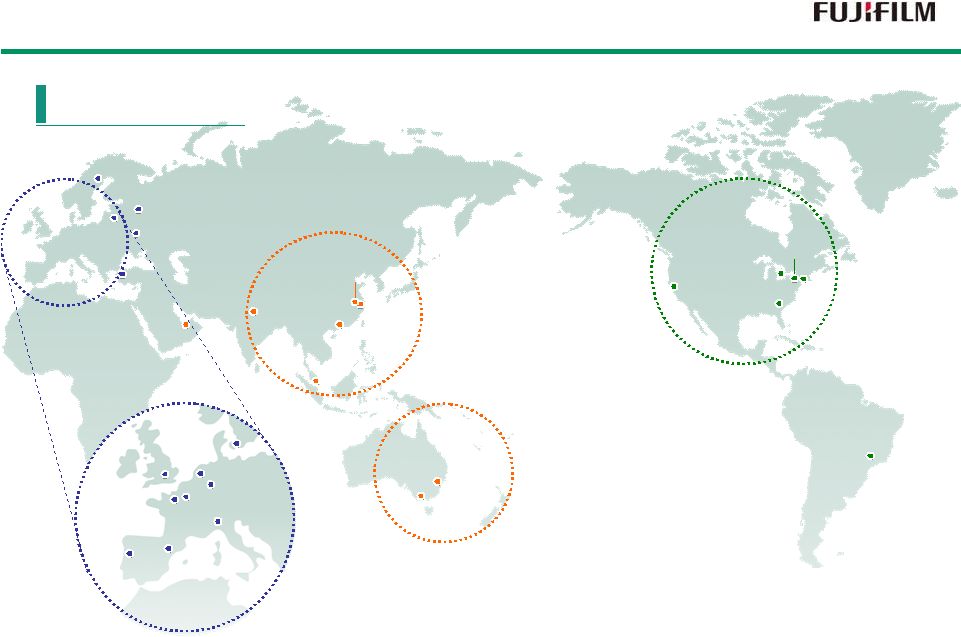 ZAO "FUJIFILM-RU" FUJIFILM Middle East FZE FUJIFILM Asia Pacific Pte. Ltd. Fuji Xerox Asia Pacific Pte. Ltd. FUJIFILM India Private Limited FUJIFILM (China) Investment Co., Ltd. FUJIFILM Medical Systems (Shanghai) Co., Ltd. Fuji Xerox (China) Limited Fuji Xerox of Shanghai Limited FUJIFILM Printing Plate (China) Co., Ltd. FUJIFILM Imaging Systems (SUZHOU) Co., Ltd. Fuji Xerox Eco-Manufacturing (Suzhou) Co., Ltd. Fuji Xerox Australia Pty. Ltd. FUJIFILM Holdings Australasia Pty Ltd. Upstream Print Solutions Pty Ltd FUJIFILM Danmark A/S FUJIFILM Manufacturing Europe B.V. FUJIFILM Europe GmbH FUJIFILM Italia S.P.A. FUJIFILM UK Ltd. FUJIFILM Holdings France SAS FUJIFILM Dimatix, Inc. FUJIFILM Manufacturing U.S.A., Inc. FUJIFILM Canada Inc. FUJIFILM Holdings America Corporation FUJIFILM North America Corporation FUJIFILM Medical Systems U.S.A., Inc. Fuji Xerox of Shenzhen Ltd. FUJIFILM do Brasil Ltda. Global Operation FUJIFILM Europe GmbH, Spanish branch FUJIFILM Europe GmbH, Portuguese branch FUJIFILM Belgium NV FUJIFILM Nordic AB FUJIFILM dis Ticaret A.S. FUJIFILM Ukraine FUJIFILM Diosynth Biotechnologies UK Limited FUJIFILM Europe GmbH, Polish branch 5 |
 6 2. Fujifilm’s Second Foundation 2. Fujifilm’s Second Foundation |
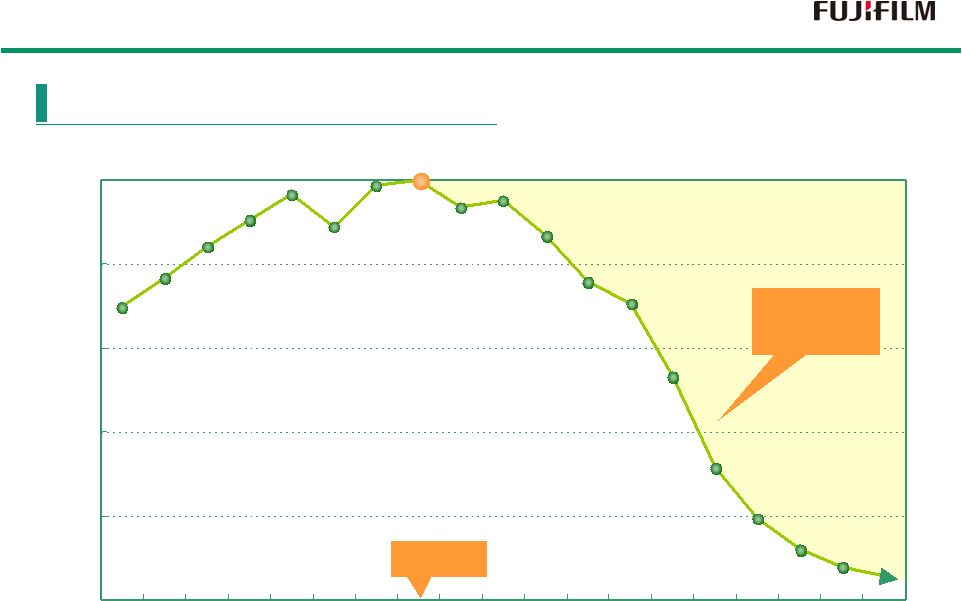 7 0 20 40 60 80 100 93 94 95 96 97 98 99 0 1 2 3 4 5 6 7 8 9 10 11 93 94 95 96 97 98 99 00 01 02 03 04 05 06 07 08 09 Peak Rapid Decline 10 (Index) Index based on an aggregate demand of 100 in the year 2000 (FY) Global Demand Trend of Color Film |
 8 VISION75 Medium-term Management Plan VISION75 (FY2003) |
 9 Structural Reform of Imaging Solutions Segment In order to continue to nurture the culture of photography… In order to continue to nurture the culture of photography… Rebuild the imaging business to ensure stable earnings Cumulative structural reform expenses: approx. 200 billion yen Restructuring and consolidation of factory, photo processing labs, R&D and sales function Structural Reform |
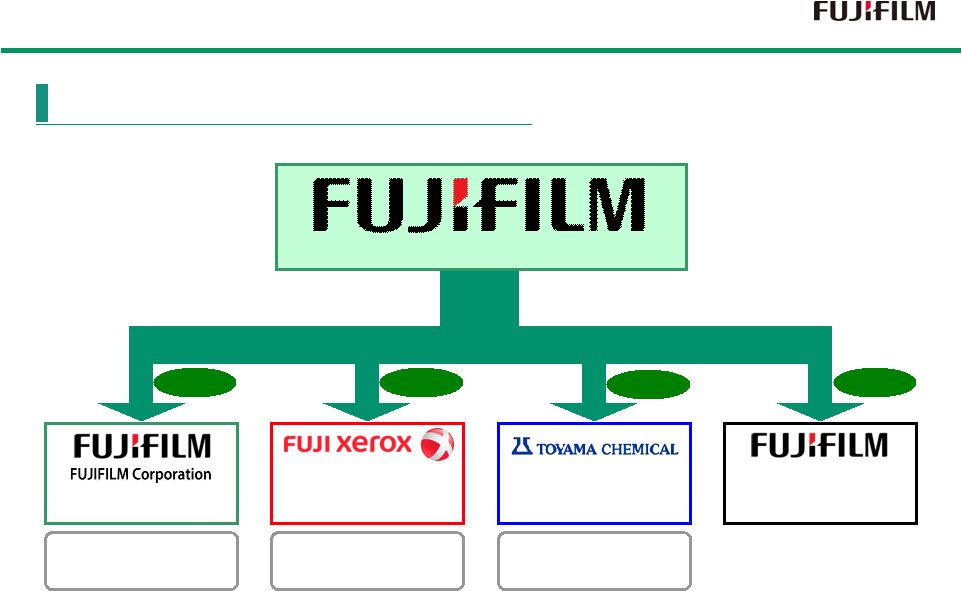 10 (Shared services company) TOYAMA CHEMICAL CO., LTD 100% 75% 66% 100% FUJIFILM Holdings Corporation Operating Company Operating Company Operating Company FUJIFILM Business Expert Shift to Holding Company Structure October, 2006 |
 11 Fujifilm’s Technologies in Four-quadrants Can we stay competitive in this area? Do we have the technology to be applied in this area? Is it a growing area? Keys to Determine Priority Businesses Existing Business New Business New Growth Strategies |
 12 Healthcare Healthcare Optical Devices Optical Devices Highly Functional Materials Highly Functional Materials Document Solutions Document Solutions Graphic Arts Systems Graphic Arts Systems Digital Imaging Digital Imaging R&D Investment New Growth Strategies Capital Investment M&A Selectively Concentrating Management Resources to Growing Business Fields |
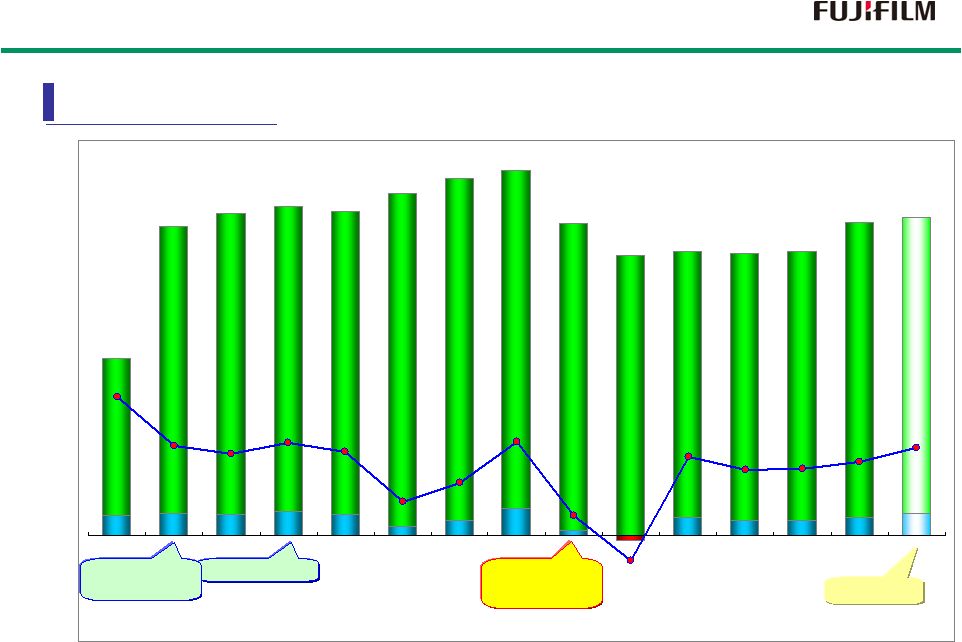 13 150 169 160 185 164 70 113 207 37 136 113 114 141 170 -42 1,383 2,408 2,512 2,567 2,527 2,667 2,783 2,847 2,434 2,182 2,217 2,195 2,215 2,440 2,480 11% 7% 6% 7% 7% 3% 4% 7% 2% 6% 5% 5% 6% 7% -2% 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 Unit : Billion Yen Total Total sales sales Operating Operating profit profit Operating Operating profit % profit % Forecast FY2000 : April 1st, 2000 to March 31st, 2001 Fiscal Fiscal year year Lehman Lehman Brothers Brothers Bankruptcy Bankruptcy VISION75 VISION75 FUJI Xerox FUJI Xerox Consolidated Consolidated Sales and Profit |
 14 3. Fujifilm’s Healthcare Business |
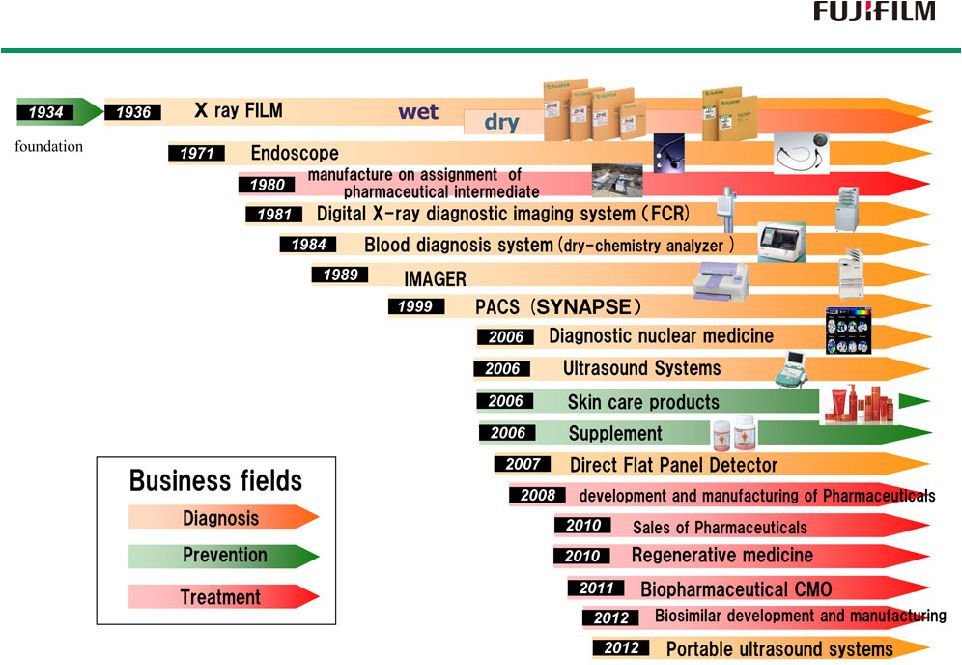 History of Healthcare Products & Services History of Healthcare Products & Services 15 |
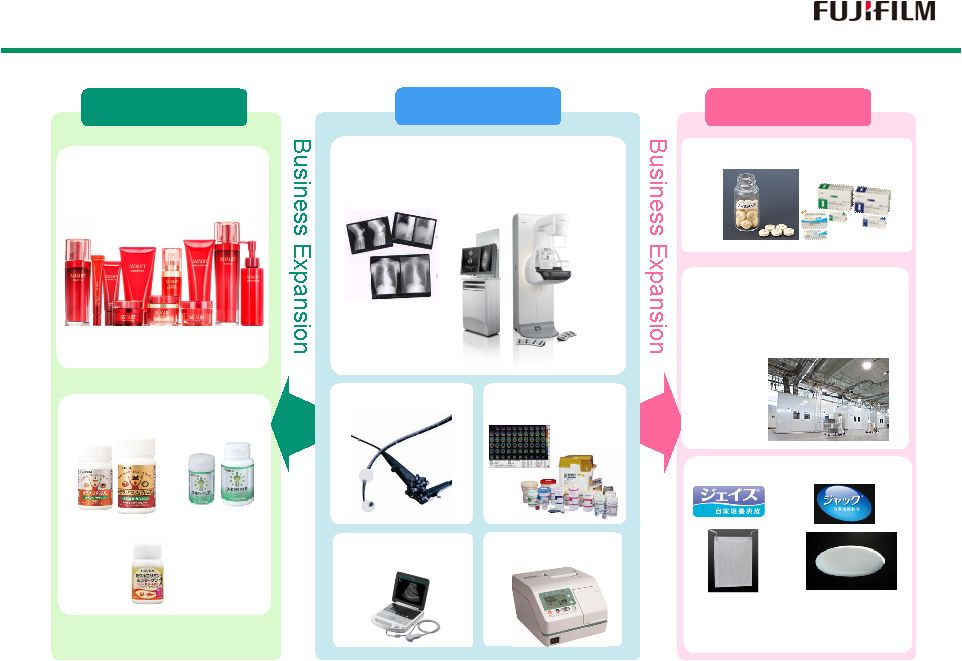 16 Diagnostics Prevention Treatment Endoscopes Low Molecular Drugs Biopharmaceuticals Comprehensive Healthcare Company Comprehensive Healthcare Company Functional Cosmetics Supplements Fujifilm Diosynth Biotechnologies Kalon Regenerative Medicine Autologous cultured epidermis Autologous cultured cartilage Perseus Proteomics X-Ray Imaging (FCR/DR/Film) Radio Diagnostic Medicines Influenza Detection Ultrasound |
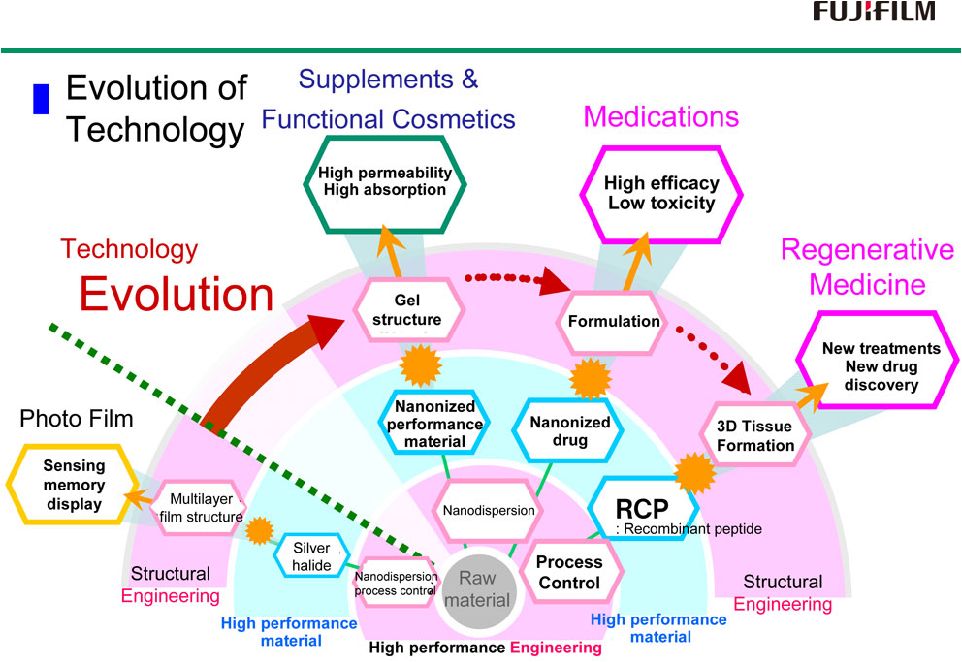 17 Core technology applied to Treatment Core technology applied to Treatment |
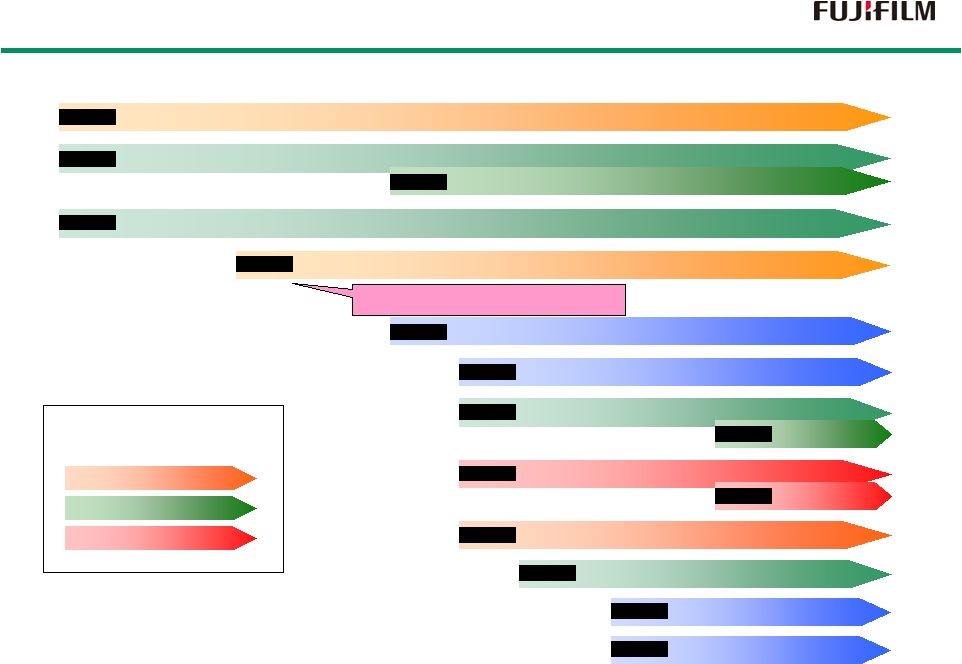 18 Regenerative medicine field Biopharmaceuticals field Low-molecular drugs field Fields 2006 2006 2006 2008 2009 2009 2010 2010 2010 2014 2014 2011 2010 2013 2013 Fujifilm Fine Chemicals Co., Ltd. became a wholly owned subsidiary Investment in Perseus Proteomics Inc. Became a Fujifilm subsidiary Aquired FUJIFILM RI Pharma Co., Ltd. Acquired Toyama Chemical Co. Entry into the Pharma Industry Pharmaceutical Research Laboratories established Pharmaceutical Products Division Established Acquired Fujifilm Diosynth Biotechnologies Invested in Japan Tissue Engineering Co., Ltd. Became consolidated subsidiary FUJIFILM Pharma Co., Ltd. established Established Fujifilm Kyowa Kirin Biologics Established Regenerative Medicine Business Development office Established Regenerative Medicine Laboratories Acquired Kalon *1 *1 : Now FUJIFILM Diosynth Biotechnologies, LLC in Texas History of Treatment Business History of Treatment Business |
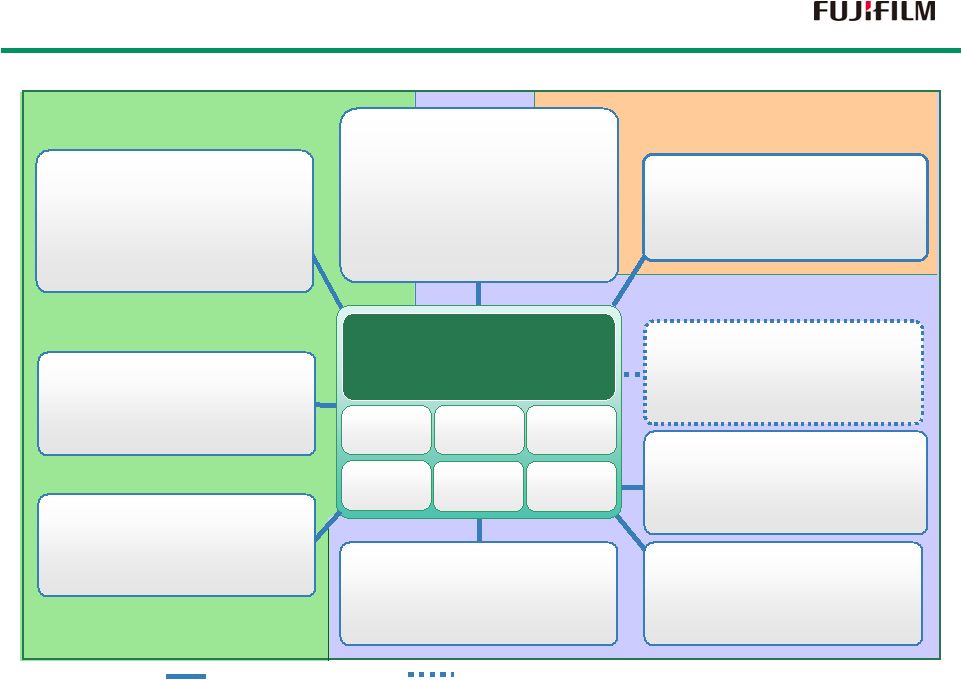 19 Consolidated Subsidiary Unconsolidated Affiliates (Equity-method affiliated companies) Japan Tissue Japan Tissue Engineering Engineering Perseus Proteomics Perseus Proteomics FUJIFILM RI Pharma FUJIFILM RI Pharma FUJIFILM Diosynth FUJIFILM Diosynth Biotechnologies Biotechnologies Low-molecular drugs Regenerative medicines Biopharmaceuticals FUJIFILM FUJIFILM Finechemicals Finechemicals Toyama Chemical Toyama Chemical FUJIFILM KYOWA FUJIFILM KYOWA KIRIN Biologics KIRIN Biologics FUJIFILM FUJIFILM Drug Discovery Drug Discovery Research Laboratory Research Laboratory FUJIFILM Pharma FUJIFILM Pharma FUJIFILM Pharma Manufacturing of raw materials Development/Manufacture of low-molecular drugs Development/Sales of pharmaceuticals Technological Resources Technological Technological Resources Resources Development/Manufacture/Sales of regenerative medical products Discovery of antibody medicines Manufacture of biopharmaceuticals Development/Sales of radiopharmaceuticals Development/Manufacture of biosimilars Discovery of low-molecular/DDS drugs Development of regenerative medical materials Development of FTD technologies Manufacturing technology Precision material designing technology Collagen technology Analysis/ assessment imaging technology Quality control Original nano- technology Summary of Treatment Business Summary of Treatment Business |
 20 4. FUJIFILM’s Assets for Regenerative Medicine 4. FUJIFILM’s Assets for Regenerative Medicine |
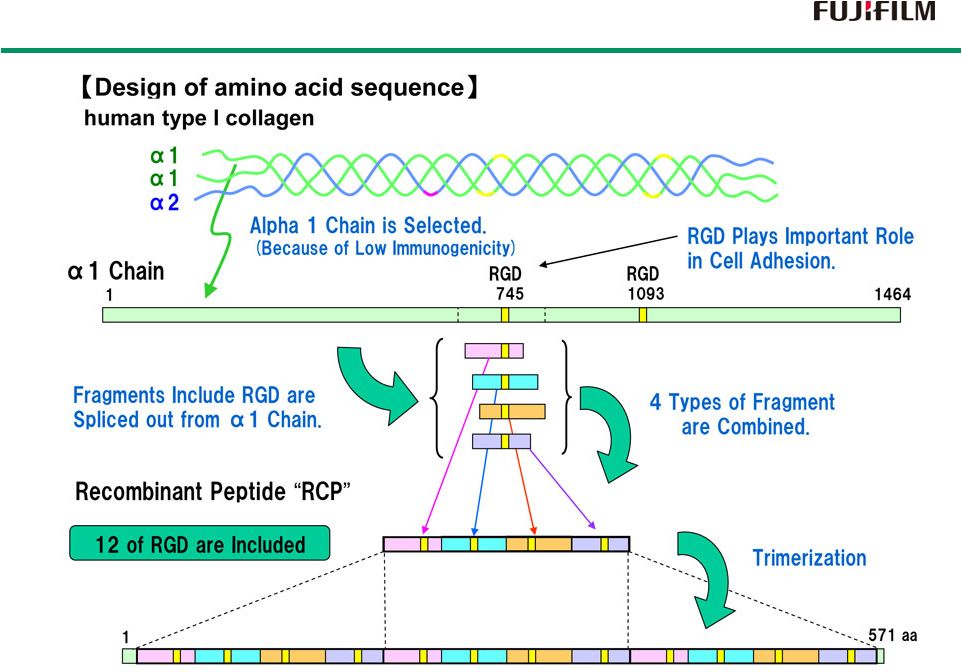 21 Our Technology, Recombinant Peptide (RCP) Our Technology, Recombinant Peptide (RCP) |
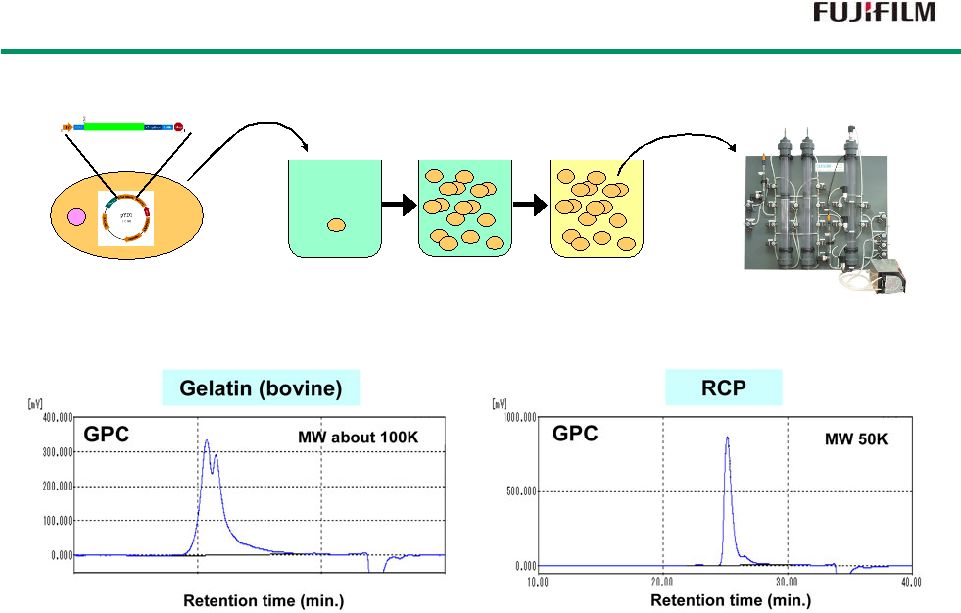 22 RCP Manufacturing RCP Manufacturing Fermentation process Purification process Purification Production of RCP Cultivation RCP gene Yeast Molecular weight distribution Batches of RCP produced in GMP production facility of FF’s subsidiary. |
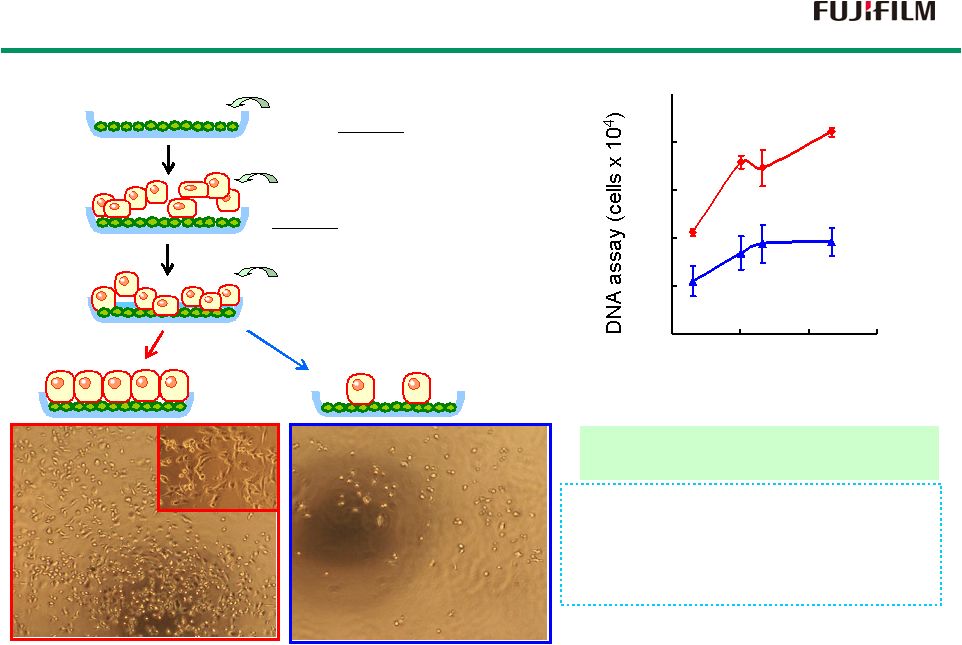 23 RCP Cell Adhesion Capability RCP Cell Adhesion Capability RCP Bovine gelatin 1.0 2.0 0.01 0.1 1 10 Amount of coating (µ 0 Inoculation and cultivation of HUVEC. (Human Umbilical Vein Endothelial Cell) Wash out with new medium. Method Extent of cell adhesion effects on; 1) Cell viability 2) Cell growth 3) Cell differentiation RCP is more effective than bovine gelatin for cell adhesion. Results Coat the material on culture dishes. (RCP or bovine gelatin) RCP RCP Bovine Gelatin Bovine Gelatin g/well) |
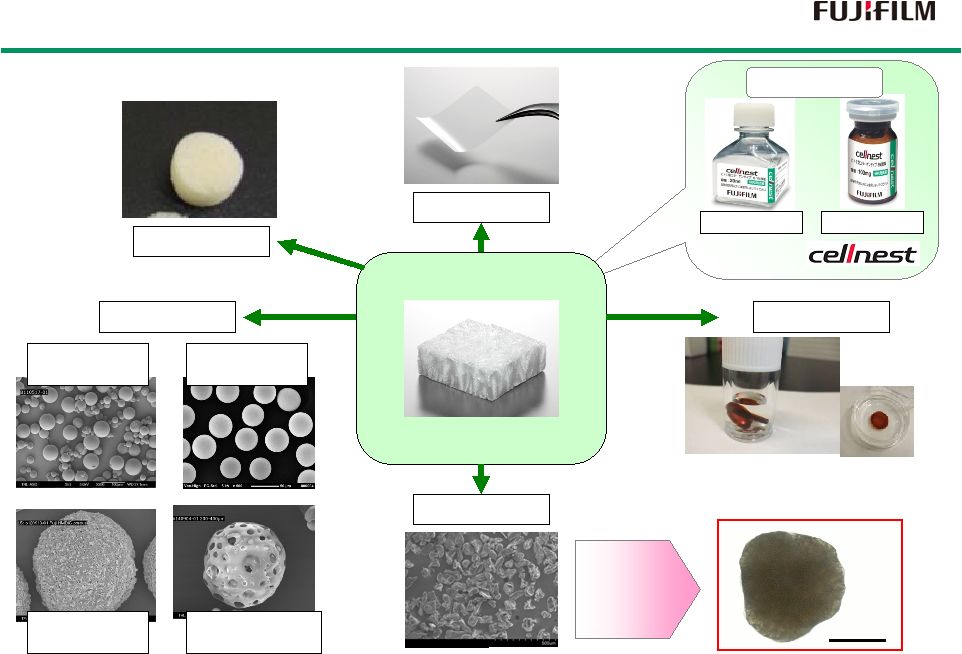 24 Various Formulations (Engineering Aptitude) Various Formulations (Engineering Aptitude) Film RCP Lyophilized 1mm Lyophilized Solution Hydro-gel Sponge Reagent Micro-Piece Micro-Sphere polydisperse smooth surface monodisperse rough surface monodisperse smooth surface Macro porous Launched as Cell culture With Micro-Piece 50-100 µ _ |
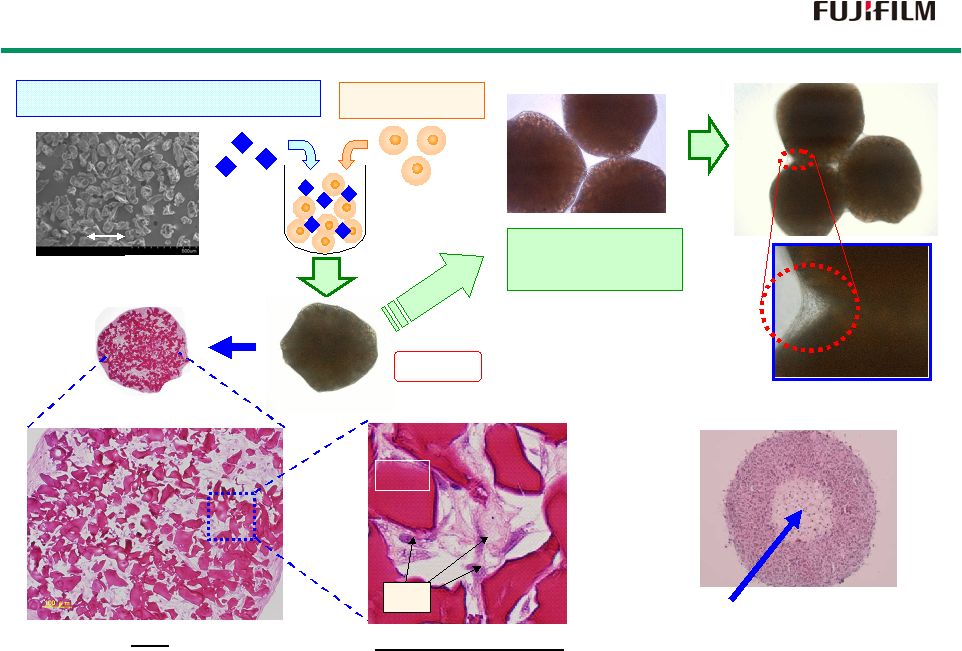 25 RCP Mosaic Cell Cluster (CellSaic) RCP Mosaic Cell Cluster (CellSaic) 50um Scaffold: RCP micro-block Cell:hMSC 100 µ m Mixed culture 100 µ m HE staining RCP mosaic cell cluster RCP RCP Cell Conventional spheroid Cell death (central necrosis) Mixed culture of cell clusters CellSaic CellSaic |
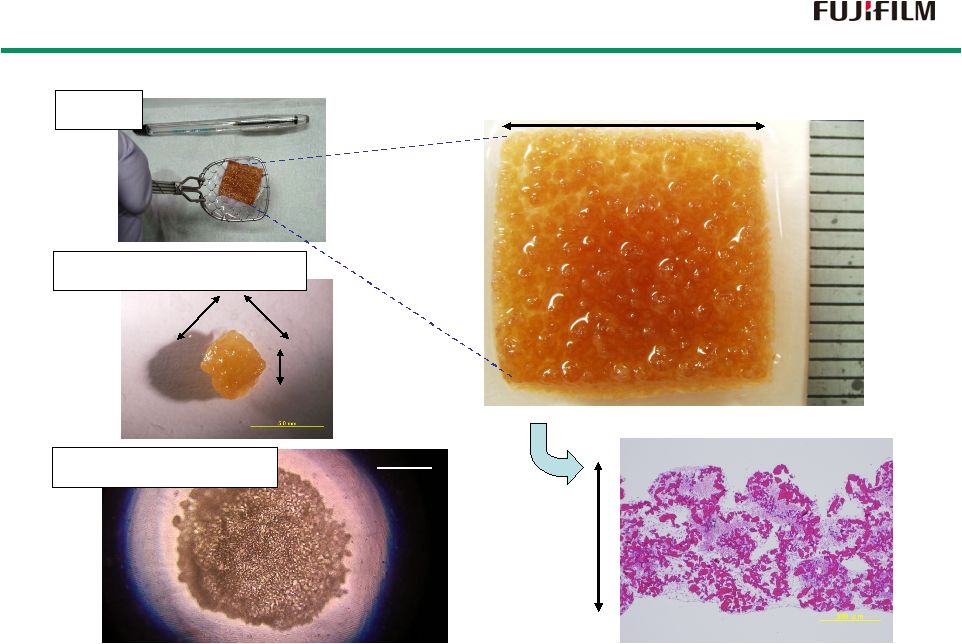 26 1.5 cm 1 mm hMSC Thickness 1.2 mm HE staining Rat Skeletal Myoblast 4mm 4mm 3mm 1 mm Rat Cardiomyocyte Regenerating Larger Tissue by CellSaic Regenerating Larger Tissue by CellSaic |
 27 High Safety High Safety 100% free from animal derived material Non toxic sequence of human type I collagen High Efficacy High Efficacy High cell adhesiveness: enriched RGD sequence Bioabsorbable and biodegradable Fujifilm developed highly safe and effective new material for regenerative medicine. High Flexibility High Flexibility Flexible to formulate into various forms such as sponges, granules, film and microsphere High Quality High Quality Pharmaceutical grade quality produced in a GMP-compliant biopharmaceutical facility Cross-sectional image of the rat’s calvarial bone a month later after transplant (H&E stain). Implanted RCP as a scaffold, regenerated bone in several weeks. Summary of RCP Summary of RCP |
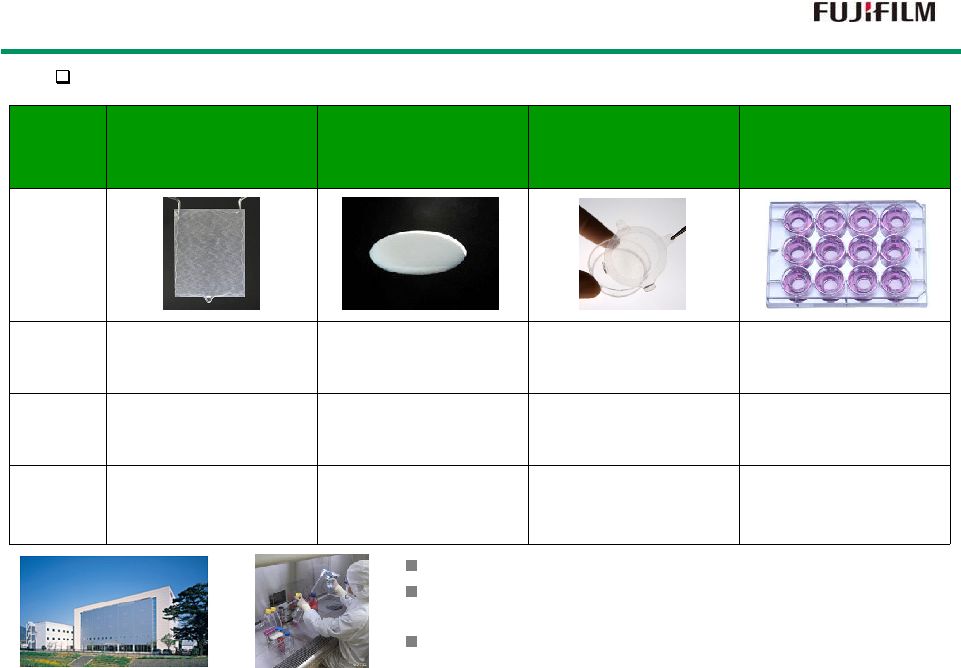 28 J-TEC carries regenerative medicinal products which are authorized in Japan. Autologous Cultured Epidermis (JACE) Autologous Cultured Cartilage (JACC) Cultured Corneal Epithelium LabCyte Products Revenue FY 2013 JPY 801million JPY 52million JPY 73million JPY 80million Indication Burn Cartilage defect Limbal stem cell deficiency Cultured human tissue for R&D use Status Authorized : 2007 Oct Insurance : 2009 Jan Authorized : 2012 Jul Insurance : 2013 Apr In discussion with PMDA (Outsourced by NIDEK) Non regulated products J-TEC has extensive capability in cell culturing. Fujifilm runs several investigational study of regenerative medicine projects with J-TEC. (critical limb ischemia etc.) J-TEC became a subsidiary of FUJIFILM at the end of 2014. Our Subsidiary, Japan Tissue Engineering Our Subsidiary, Japan Tissue Engineering |
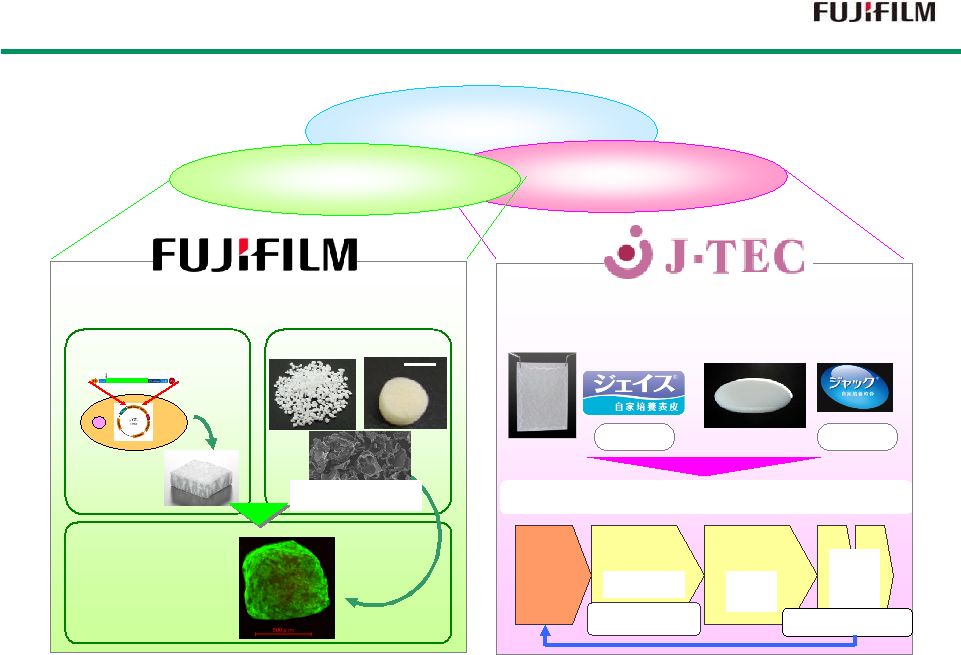 29 Strengths and Characteristics of Fujifilm Group Strengths and Characteristics of Fujifilm Group Cells Three main pillars of Three main pillars of regenerative medicine regenerative medicine Created first regenerative medicine products in Japan Autologous cultured epidermis Autologous cultured cartilage Scaffold Scaffold (extracellular matrix ) 2007 2007 2012 2012 (intercellular signaling compound) Cytokines Petaloid: meaning “petal-like” Request Request item item R&D, R&D, Clinical Trials Clinical Trials Manufacture Manufacture Inspection Inspection Sales Sales Use Use GVP GQP GPSP GCTP GQP GLP, GCP Application for Drug approval Post-marketing care Feedback Quality Management System supported by experience Realization of non-animal derived scaffolding material 1cm 1cm Granules Granules Sponge Sponge Engineering Technology Engineering Technology 3D Cell Structure “Cellsaic” Highly Functional Highly Functional Material Technology Material Technology Yeast RCP Gene Introduction Lyophilized RCP × × High Added Value High Added Value Cultivate, Refine Microscopic Petaloid Pieces Combine Combine with Cells with Cells |
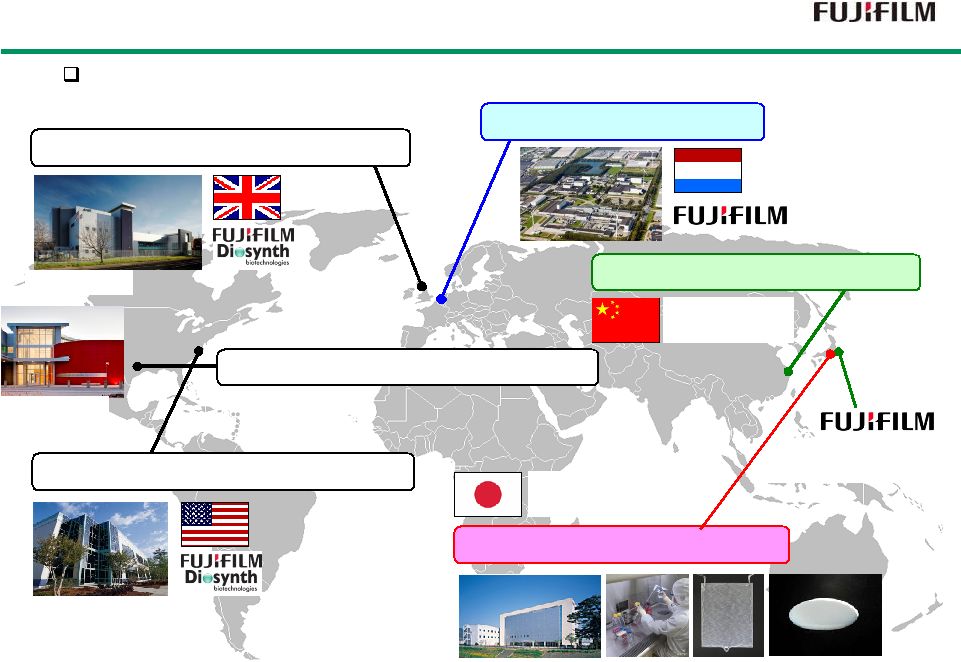 30 Fujifilm’s Group Companies Fujifilm’s Group Companies FUJIFILM Diosynth Biotechnologies UK FUJIFILM Diosynth Biotechnologies US Fujifilm is expanding regenerative medicine business platform worldwide. J-TEC (Japan Tissue Engineering) FUJIFILM Tissue Engineering Co. Biologics CMO Biologics CMO To develop cultured epidermis Developing RCP Developing RM products Tilburg Research Laboratory Research Laboratory Business Development Office FUJIFILM Diosynth Biotechnologies TX Biologics CMO |
 31 4. Fujifilm Policy for Regenerative Medicine 4. Fujifilm Policy for Regenerative Medicine |
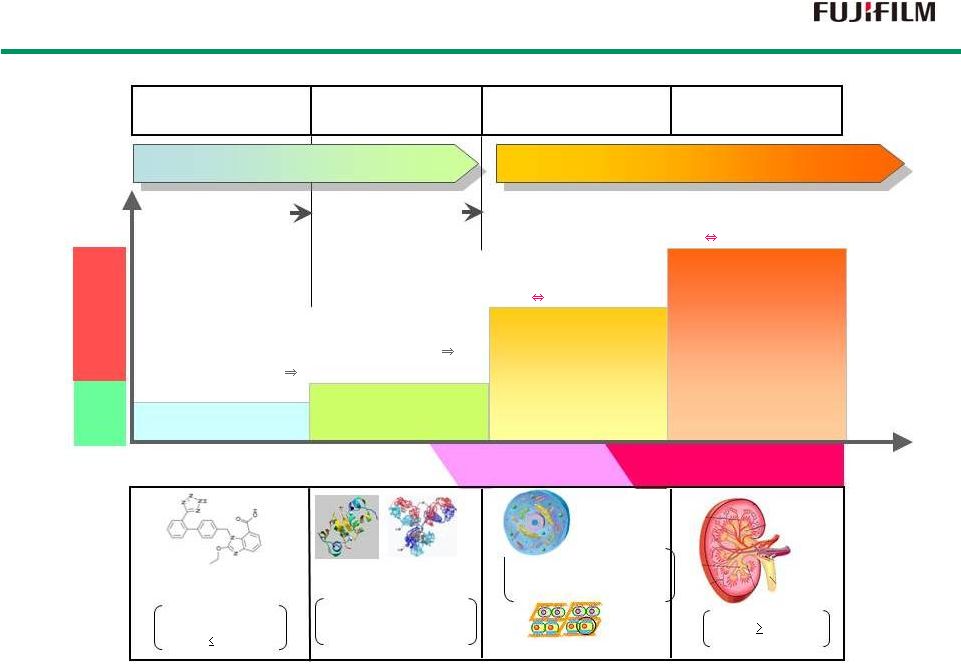 32 Self-organization Structuring /Ordering Simulated Bioorganism Structuring Low molecule compound Single cell Single cell type tissue: Heart (myocardium), Nerves, Skin (epithelium), Cartilage Organs comprising various cell types: Liver Pancreas Kidneys Low Molecule Drugs Organ Regeneration Therapy Biopharmaceuticals Cell Therapy / Tissue Regeneration Therapy Biopolymer Insulin, Antibody Use natural healing properties of bioorganisms Acts on a bioorganism Candesartan (angiotensin II receptor antagonist) MW: <1,000 Size: 3-4 nm Size: 3-4 µm to 3-4cm Cell Size: 3-4 cm organ Insulin Antibody MW: order of 10 3 to 10 5 Size: 1 to 10 nm Extracellular matrix Symptomatic Treatment Curative Therapy Fujifilm Group Goals in Regenerative Medicine Fujifilm Group Goals in Regenerative Medicine Single Low Molecule Single Target «Non-interactive» Single Polymer Single/Multiple Targets «Non-interactive» Single Cell / Tissue Body Tissue Multiple Cells / Organ Body Tissue «Interactive» «Interactive» First Wall Second Wall |
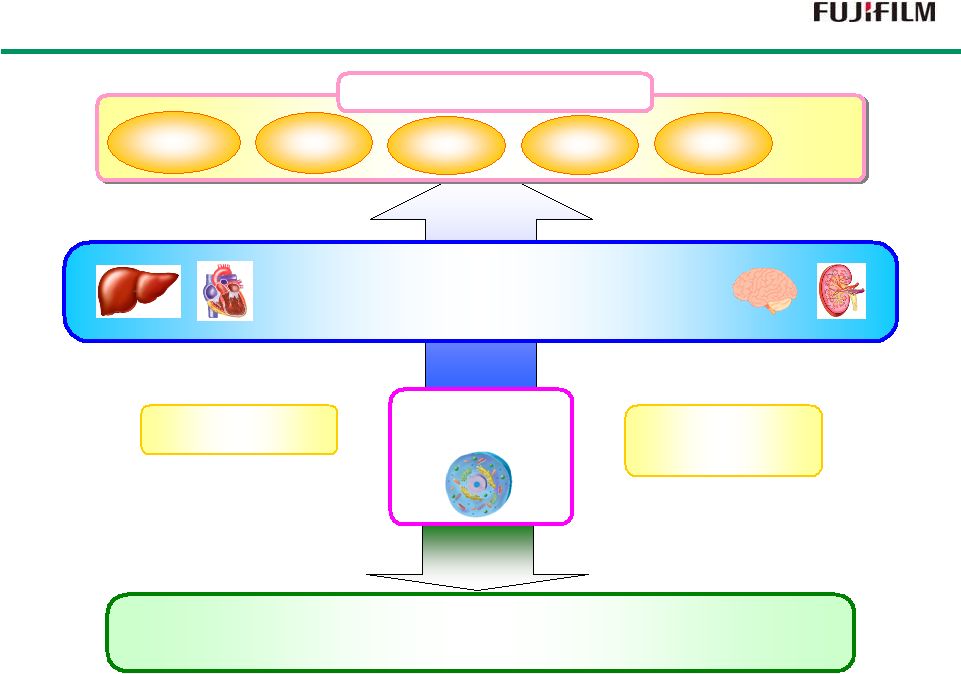 33 Significance of Efforts in Regenerative Medicine Significance of Efforts in Regenerative Medicine Drug Discovery Legal Reform Legal Reform Technical Technical Advancements Advancements Regenerative Medicine Unmet Medical Needs Age-related macular degeneration Severe heart failure Strokes iPS Cell Spinal Cord Injuries Diabetes · · · · |
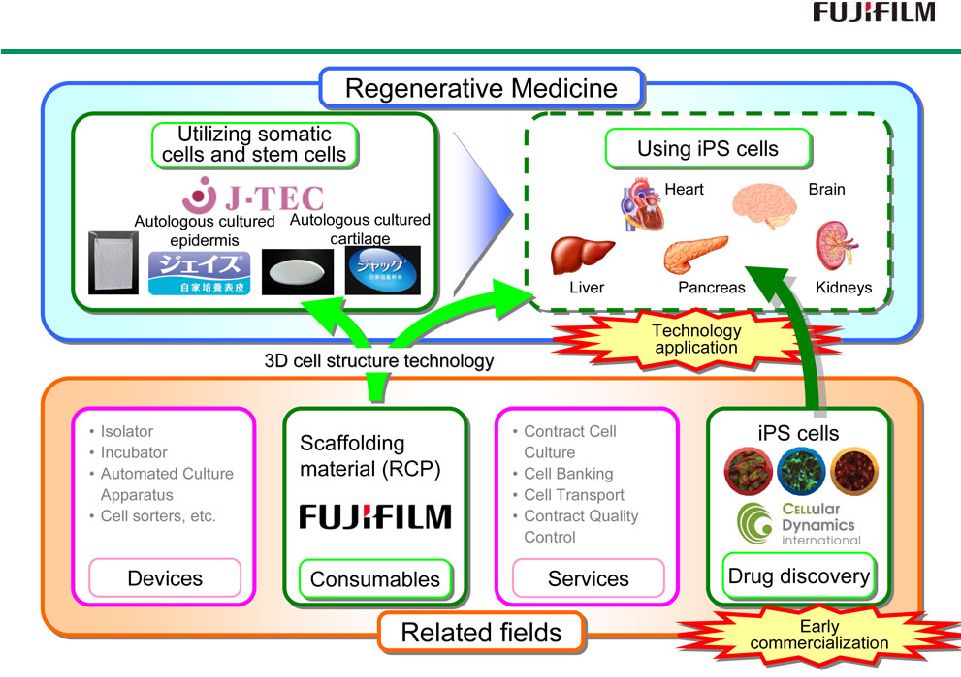 Fujifilm’s Regenerative Medicine Fujifilm’s Regenerative Medicine Initiatives Initiatives RCP : Recombinant peptide 34 |
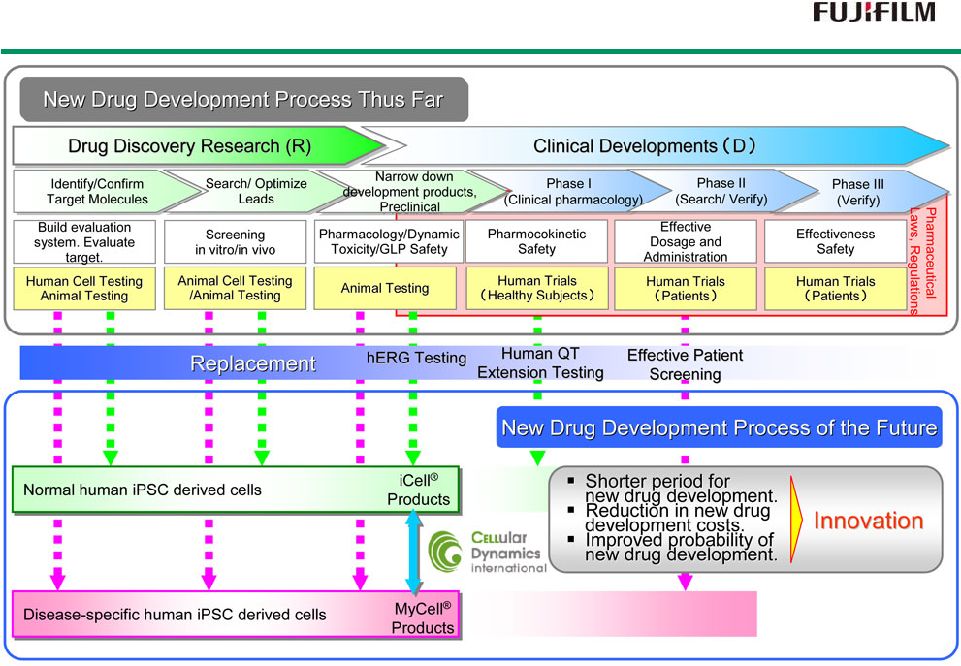 35 Drug Discovery Innovation through Human iPS Cells Drug Discovery Innovation through Human iPS Cells |
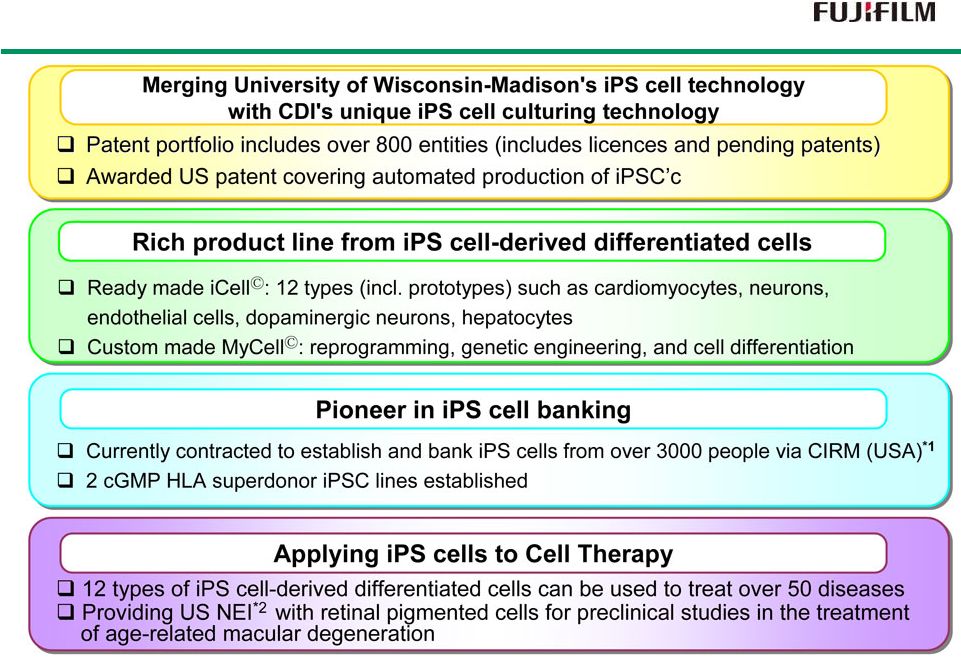 36 Strengths and Features of CDI *1 : California Institute for Regenerative Medicine *2 : National Eye Institute |
 37 Anticipated Technology Synergy Anticipated Technology Synergy 1 2 3 1 3 2 Extracellular matrix Engineering Technology Reprograming/ differentiation iPS cells Quality management Somatic cell/Stem cell FUJIFILM & CDI: Expand drug discovery support J-TEC & CDI : Development of contract cell culturing FUJIFILM & CDI & J-TEC: Organ regeneration R&D Combined with FUJIFILM’s extracellular matrix (Recombinant Peptide: RCP) enhanced performance materials can be used to create 3D structures, expanding drug discovery opportunities. Together with quality management systems (GCTP/GQP, etc.) nurtured through J-TEC’s regenerative products, iPS cell banking and contract cell culturing for use in cell therapy. Combine technology from these 3 companies and develop organ regenerative treatments enabling transplantation of a 3D cell structured organ mimic. technologies developed during drug discovery support will flow back to regenerative medicine. |
 38 Finally Finally |
 39 We will use leading-edge, proprietary technologies to provide top-quality products and services that contribute to the advancement of culture, science, technology and industry, as well as improved health and environmental protection in society. Our overarching aim is to help enhance the quality of life of people worldwide. Fujifilm Group Corporate Philosophy |
 40 Thank you for your attention. Thank you for your attention. We are proud of your excellent people We are proud of your excellent people and cutting-edge technologies. and cutting-edge technologies. |
 41 |