
Targeting cancer, differently. Susan M. Molineaux, Ph.D. | Founder, President & Chief Executive Officer Exhibit 99.2

Forward-Looking Statements This presentation and the accompanying oral commentary contain “forward‐looking” statements for purposes of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. We may, in some cases, use terms such as “believe,” “will,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “might,” “approximately,” “expect,” “predict,” “could,” “potentially” or the negative of these terms or other words that convey uncertainty of future events or outcomes to identify these forward‐looking statements. All statements other than statements of historical facts contained in this presentation and the accompanying oral commentary are forward‐looking statements, and such forward‐looking statements include statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things: plans regarding anticipated clinical trials for our product candidates, including CB-228 (sapanisertib), CB-659 (mivavotinib), CB-839 (telaglenastat), CB-280, INCB001158 and CB-708, the potential safety, efficacy and other benefits of and market opportunity of product candidates, the timing of and our ability to make regulatory filings and obtain and maintain regulatory approvals for our product candidates, statements relating to future royalties and the development, regulatory and sales milestone payments of INCB001158 and CB-708 in connection with our collaborations with Incyte and Antengene, respectively, and of CB-228 and CB-659 in connection with our asset purchase agreement with Takeda, our intellectual property position and cash needs. Forward‐looking statements involve known and unknown risks, uncertainties, assumptions and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward‐looking statements. The potential product candidates that we develop may not progress through clinical development or receive required regulatory approvals within expected timelines or at all. In addition, clinical trials may not confirm any safety, potency or other product characteristics described or assumed in this presentation and the accompanying oral commentary. Such product candidates may not be beneficial to patients or be successfully commercialized. The failure to meet expectations with respect to any of the foregoing matters may have a negative effect on our stock price. We discuss many of these risks in greater detail under the heading "Risk Factors" contained in our Quarterly Report on Form 10‐Q for the quarter ended June 30, 2021, filed with the Securities and Exchange Commission on August 5, 2021. Forward‐looking statements are not guarantees of future performance and our actual results of operations, financial condition and liquidity, and the development of the industry in which we operate may differ materially from the forward‐looking statements contained in this presentation and the accompanying oral commentary. Any forward‐looking statements that we make in this presentation and the accompanying oral commentary speak only as of the date of this presentation. We assume no obligation to update our forward‐looking statements whether as a result of new information, future events or otherwise. No Offer or Solicitation This presentation does not constitute an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction.

is a clinical-stage, precision oncology biopharmaceutical company developing targeted therapies to redefine treatment for biomarker-specific patient populations

Transformative Acquisition Sapanisertib (TAK-228; mTORC1/2 inhibitor) Calithera has acquired two Phase 2 oncology compounds from Takeda Both compounds have single agent clinical activity and the potential to be first-to-market drugs in biomarker-defined populations of cancer patients Mivavotinib (TAK-659; SYK inhibitor) Entering a Phase 2 trial for squamous NSCLC patients with NRF2 mutations Extends Calithera’s commitment to the KEAP1/NRF2 pathway Entering a Phase 2 trial for DLBCL patients with or without MyD88 or CD79 mutations TAK-228 will be referred to as CB-228 post transaction TAK-659 will be referred to as CB-659 post transaction
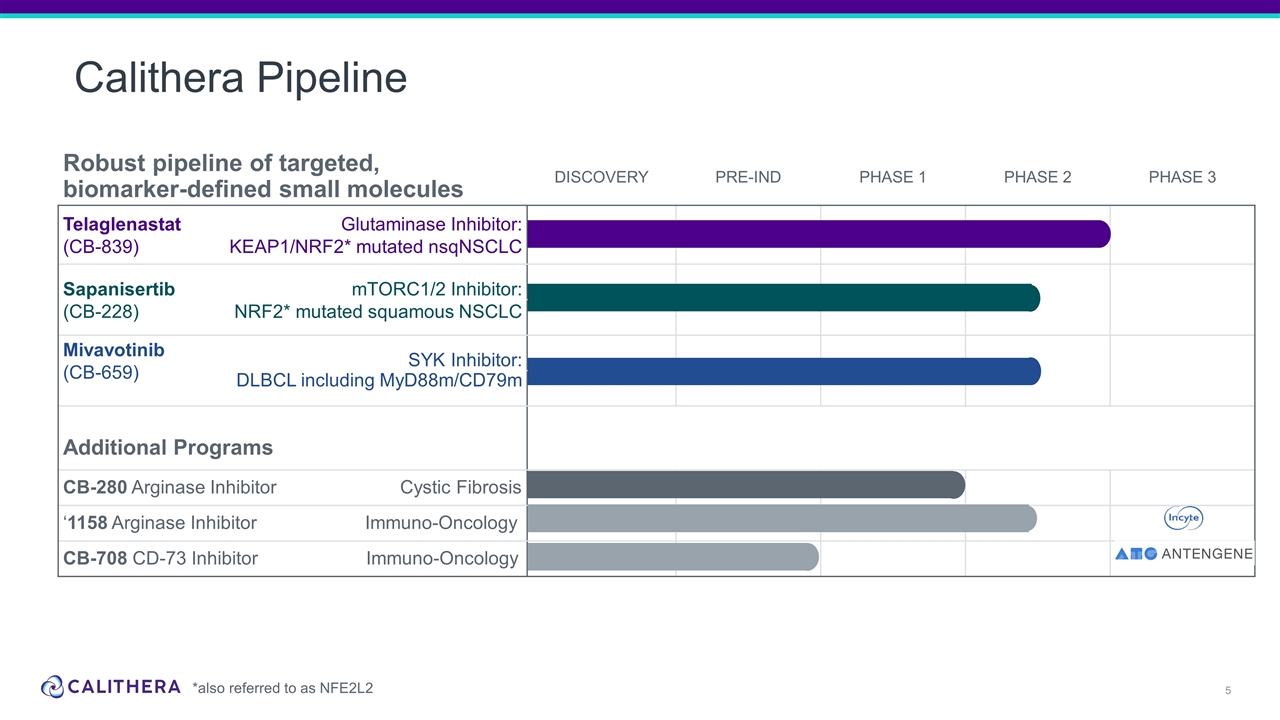
Robust pipeline of targeted, biomarker-defined small molecules DISCOVERY PRE-IND PHASE 1 PHASE 2 PHASE 3 Telaglenastat (CB-839) Glutaminase Inhibitor: KEAP1/NRF2* mutated nsqNSCLC Sapanisertib (CB-228) mTORC1/2 Inhibitor: NRF2* mutated squamous NSCLC Lung KEAP1/NRF2* mutation KEAPSAKE Mivavotinib (CB-659) SYK Inhibitor: DLBCL including MyD88m/CD79m Additional Programs CB-280 Arginase Inhibitor Cystic Fibrosis ‘1158 Arginase Inhibitor Immuno-Oncology CB-708 CD-73 Inhibitor Immuno-Oncology Calithera Pipeline *also referred to as NFE2L2

Telaglenastat and Sapanisertib KEAP1/NRF2 Programs

Telaglenastat Glutaminase Inhibitor in Development for Non-Squamous NSCLC Patients Harboring KEAP1 or NRF2 mutations
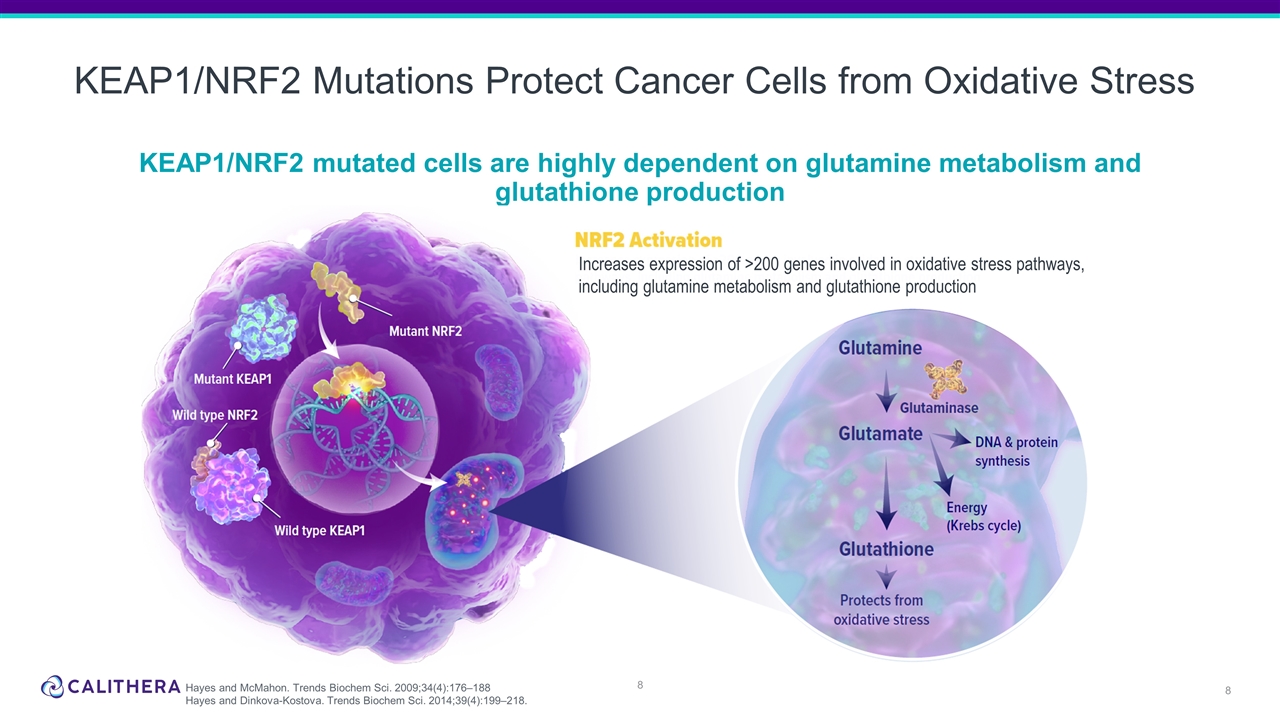
KEAP1/NRF2 Mutations Protect Cancer Cells from Oxidative Stress KEAP1/NRF2 mutated cells are highly dependent on glutamine metabolism and glutathione production Increases expression of >200 genes involved in oxidative stress pathways, including glutamine metabolism and glutathione production Hayes and McMahon. Trends Biochem Sci. 2009;34(4):176–188 Hayes and Dinkova-Kostova. Trends Biochem Sci. 2014;39(4):199–218.

Telaglenastat In KEAP1/NRF2*- Mutated nonsquamous NSCLC SOC=pembrolizumab, carboplatin, pemetrexed (PCP) *also referred to as NFE2L2 https://www.cbioportal.org/ Gadjeel et al. LB-397, AACR 2020 Skoulidis et al. J Clin Oncol 38: 2020 (suppl; abstr TPS9627) ASCO 2020 https://clinicaltrials.gov/ct2/show/NCT04698681?cond=keapsake&draw=2&rank=1. KEAPSAKE is a randomized Phase 2 trial of telaglenastat with SOC in 1L adeno NSCLC patients with KEAP1/NRF2 mutations (20% of adeno NSCLC) 1 Strong biologic rationale KEAP1/NRF2 mutations protect cells from oxidative stress; these mutations result in high dependence on glutamine metabolism for glutathione production Targeted biomarker-driven approach KEAP1m/NRF2m detected through NGS / Liquid biopsy 4 Interim data expected 4Q 2021 Potential first-in-class molecule Telaglenastat is the first glutaminase inhibitor to enter clinical trials High unmet need Activating KEAP1/NRF2 mutations occur early in tumor development and drive aggressive growth Patients with KEAP1m/NRF2m NSCLC have poor outcomes with SOC therapy 2 Evidence of clinical benefit in KEAP1m NSCLC patients from prior telaglenastat trial 3 KEAP1m tumors are sensitive to telaglenastat in preclinical models
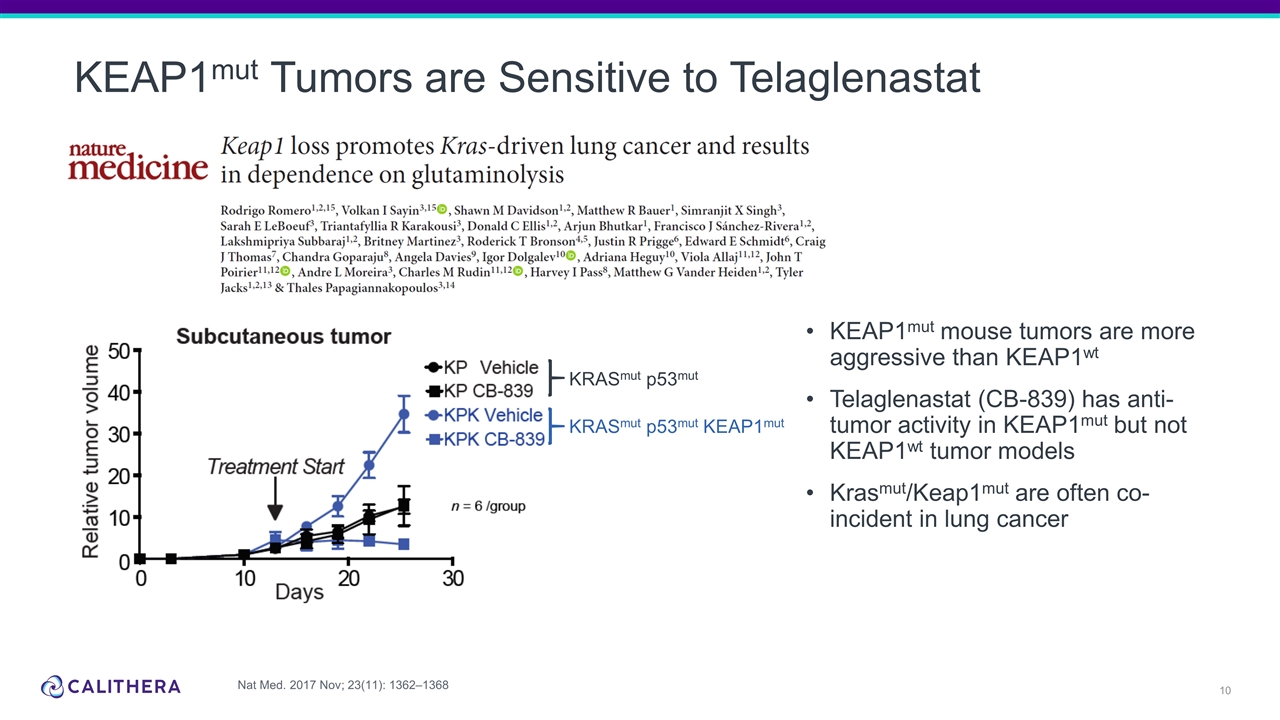
KEAP1mut Tumors are Sensitive to Telaglenastat KRASmut p53mut KRASmut p53mut KEAP1mut KEAP1mut mouse tumors are more aggressive than KEAP1wt Telaglenastat (CB-839) has anti-tumor activity in KEAP1mut but not KEAP1wt tumor models Krasmut/Keap1mut are often co-incident in lung cancer Nat Med. 2017 Nov; 23(11): 1362–1368

KEAPSAKE: Telaglenastat and Standard of Care in 1L Patients With KEAP1/NRF2-mutated Non-squamous NSCLC Ongoing Randomized Ph2 Trial Placebo Pembro + Chemo 1L nsqNSCLC Stage IV N = 120 KEAP1/NRF2-mutation detected by NGS/liquid biopsy Double-blinded and placebo controlled Telaglenastat Pembro + Chemo Primary Endpoint: PFS Interim Data expected 4Q21 STK11/LKB status Metastasis stage (M1a/b vs M1c) https://clinicaltrials.gov/ct2/show/NCT04265534 Pembro=pembrolizumab Chemo= carboplatin + pemetrexed Stratified by: Potential to be the first-in-class glutaminase inhibitor and the first drug approved for KEAP1/NRF2-mutated non-squamous NSCLC
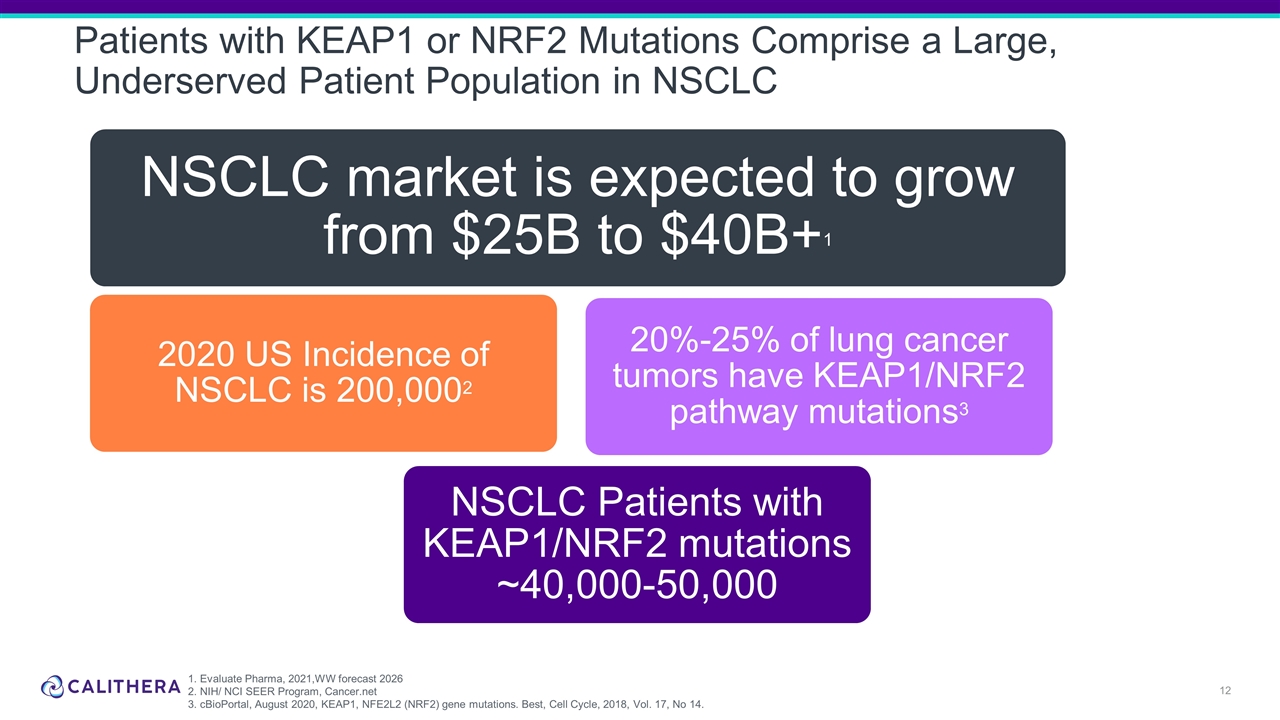
Patients with KEAP1 or NRF2 Mutations Comprise a Large, Underserved Patient Population in NSCLC 1. Evaluate Pharma, 2021,WW forecast 2026 2. NIH/ NCI SEER Program, Cancer.net 3. cBioPortal, August 2020, KEAP1, NFE2L2 (NRF2) gene mutations. Best, Cell Cycle, 2018, Vol. 17, No 14. NSCLC market is expected to grow from $25B to $ 40B+ 1 NSCLC Patients with KEAP1/NRF2 mutations ~40,000-50,000 20%-25% of lung cancer tumors have KEAP1/NRF2 pathway mutations 3 2020 US Incidence of NSCLC is 200,000 2

Sapanisertib mTORC1/2 Inhibitor in Development for Treatment of Squamous NSCLC patients whose tumors harbor NRF2 mutations
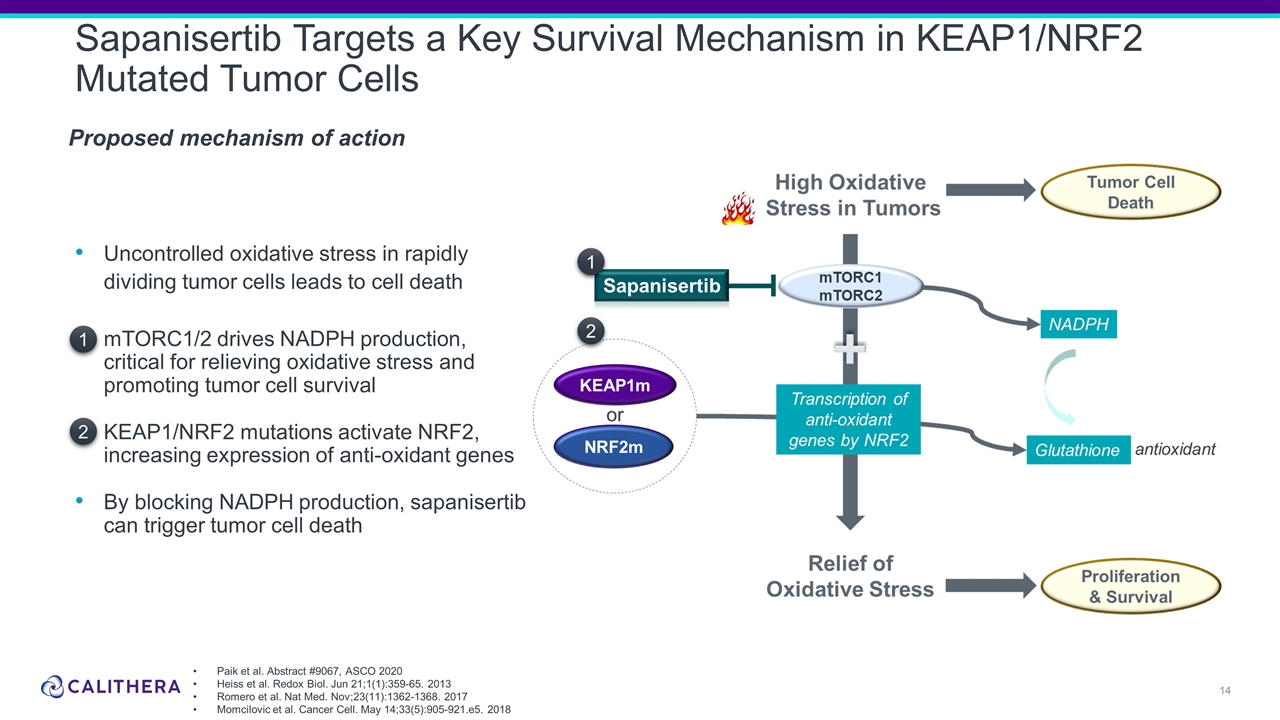
Uncontrolled oxidative stress in rapidly dividing tumor cells leads to cell death mTORC1/2 drives NADPH production, critical for relieving oxidative stress and promoting tumor cell survival KEAP1/NRF2 mutations activate NRF2, increasing expression of anti-oxidant genes By blocking NADPH production, sapanisertib can trigger tumor cell death Sapanisertib Targets a Key Survival Mechanism in KEAP1/NRF2 Mutated Tumor Cells Proposed mechanism of action Paik et al. Abstract #9067, ASCO 2020 Heiss et al. Redox Biol. Jun 21;1(1):359-65. 2013 Romero et al. Nat Med. Nov;23(11):1362-1368. 2017 Momcilovic et al. Cancer Cell. May 14;33(5):905-921.e5. 2018 1 2 1 2
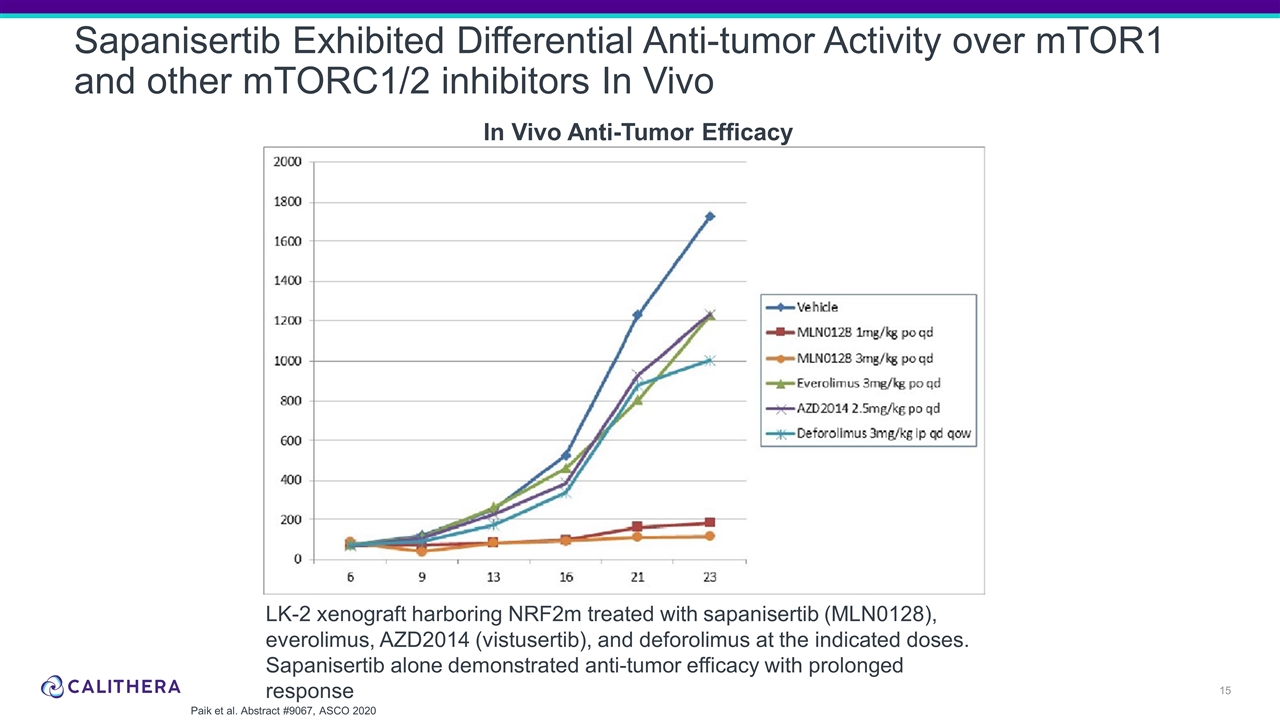
Sapanisertib Exhibited Differential Anti-tumor Activity over mTOR1 and other mTORC1/2 inhibitors In Vivo LK-2 xenograft harboring NRF2m treated with sapanisertib (MLN0128), everolimus, AZD2014 (vistusertib), and deforolimus at the indicated doses. Sapanisertib alone demonstrated anti-tumor efficacy with prolonged response Paik et al. Abstract #9067, ASCO 2020 In Vivo Anti-Tumor Efficacy

Sapanisertib Overview Sapanisertib is an mTORC1/2 inhibitor with single-agent clinical activity in NRF2*m sqNSCLC1 NRF2 mutations occur in ~15% of sqNSCLC patients2 Sapanisertib exhibits differential anti-tumor activity compared to rapalog inhibitors of TORC1 in NRF2-mutant squamous NSCLC in vivo models1 Potential for single agent activity in other tumor types with KEAP1 and NRF2 mutations Sapanisertib has a well-characterized clinical safety profile1,3 IP COM protection through 2036 US/2034 EU assuming full 5-year patent term extension4 *NRF2 is also known as NFE2L2 WT=wild type COM=composition of matter Paik et al.. Abstract #9607, ASCO 2020 Data from TCGA BioPortal https://www.cbioportal.org/ Voss et al. British Journal of Cancer (2020) 123:1590–1598; US 8,476,282 B2 and WO 2010/051043 Fast-to-market strategy in relapsed/refractory squamous NSCLC with potential to be the first targeted treatment for NRF2-mutated cancer

Squamous Non-Small Cell Lung Cancer Treatment Overview >235,000 diagnosed with NSCLC in the US each year, ~25-30% of those are squamous NSCLC1-2 Actionable mutations are only found in 1-5% of squamous NSCLC patients, leaving patients few options after PD1 inhibitors and chemo3,4 The standard-of-care options in R/R sqNSCLC have a mPFS of 3-4.5 mos5,6 NRF2 mutations occur in ~15% of sqNSCLC patients3 NRF2/KEAP1 mutations confer a poorer prognosis than WT for NSCLC patients7 https://seer.cancer.gov/statfacts/html/lungb.html https://www.cancer.gov/types/lung/hp/non-small-cell-lung-treatment-pdq#_359 Data from TCGA BioPortal https://www.cbioportal.org/ NCCN NSCLC guidelines June 2021 Garon et al. Lancet. Aug 23;384(9944):665-73. 2014 Leong et al. J Thorac Oncol. Mar;2(3):230-6. 2007 Rizvi et al. Oral Abstract OA04.07: World Conference on Lung Cancer, 2019 Sapanisertib has the potential to address a substantial underserved patient population
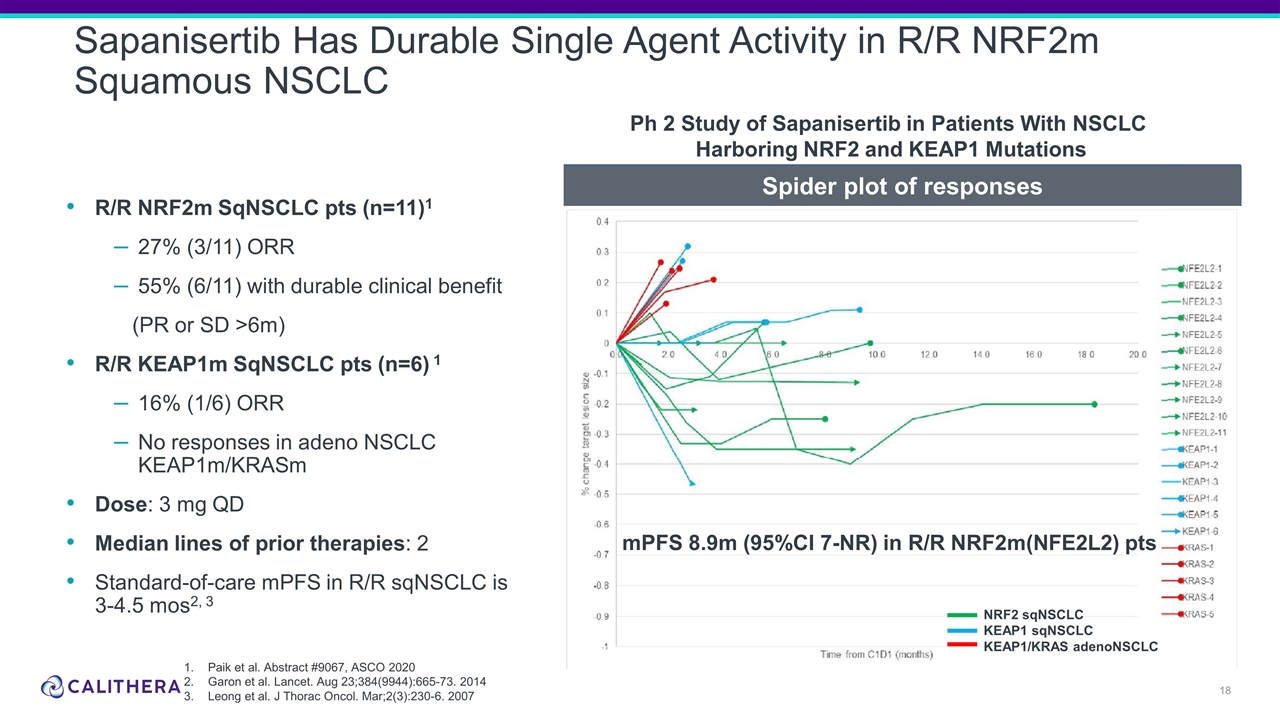
R/R NRF2m SqNSCLC pts (n=11)1 27% (3/11) ORR 55% (6/11) with durable clinical benefit (PR or SD >6m) R/R KEAP1m SqNSCLC pts (n=6) 1 16% (1/6) ORR No responses in adeno NSCLC KEAP1m/KRASm Dose: 3 mg QD Median lines of prior therapies: 2 Standard-of-care mPFS in R/R sqNSCLC is 3-4.5 mos2, 3 Sapanisertib Has Durable Single Agent Activity in R/R NRF2m Squamous NSCLC Paik et al. Abstract #9067, ASCO 2020 Garon et al. Lancet. Aug 23;384(9944):665-73. 2014 Leong et al. J Thorac Oncol. Mar;2(3):230-6. 2007 mPFS 8.9m (95%CI 7-NR) in R/R NRF2m(NFE2L2) pts NRF2 sqNSCLC KEAP1 sqNSCLC KEAP1/KRAS adenoNSCLC Ph 2 Study of Sapanisertib in Patients With NSCLC Harboring NRF2 and KEAP1 Mutations Spider plot of responses
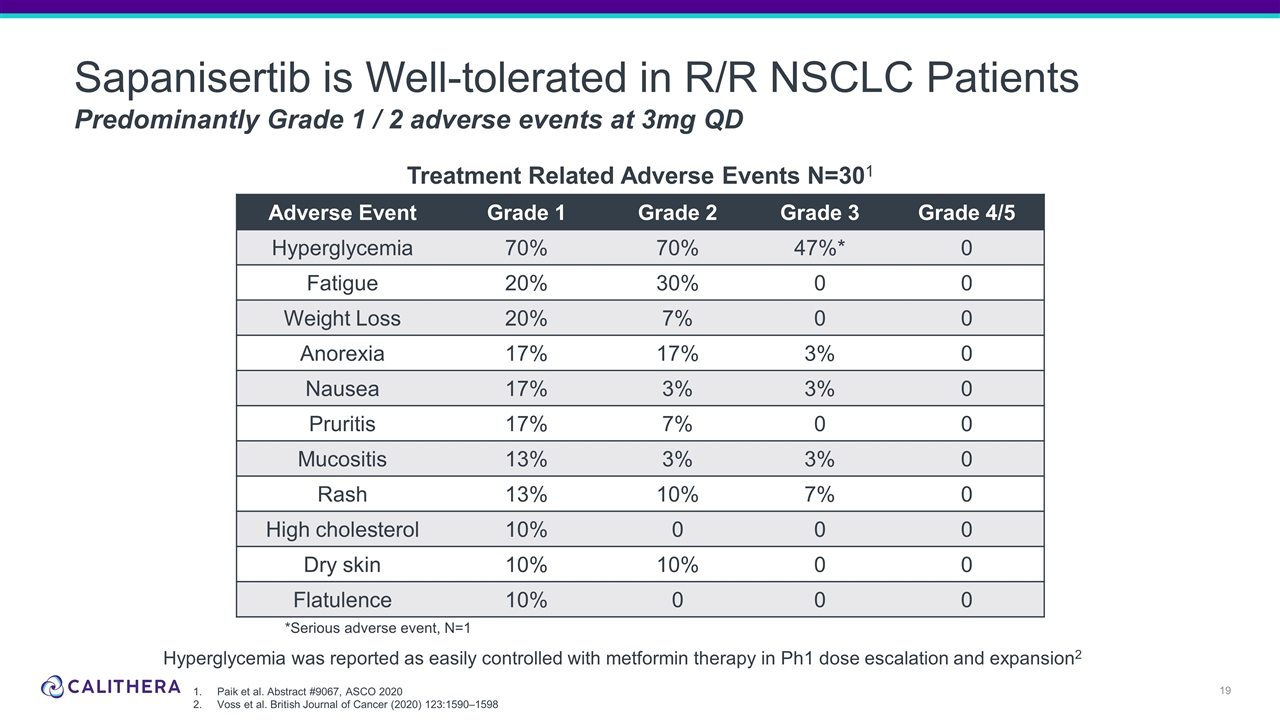
Sapanisertib is Well-tolerated in R/R NSCLC Patients Predominantly Grade 1 / 2 adverse events at 3mg QD Adverse Event Grade 1 Grade 2 Grade 3 Grade 4/5 Hyperglycemia 70% 70% 47%* 0 Fatigue 20% 30% 0 0 Weight Loss 20% 7% 0 0 Anorexia 17% 17% 3% 0 Nausea 17% 3% 3% 0 Pruritis 17% 7% 0 0 Mucositis 13% 3% 3% 0 Rash 13% 10% 7% 0 High cholesterol 10% 0 0 0 Dry skin 10% 10% 0 0 Flatulence 10% 0 0 0 Treatment Related Adverse Events N=301 *Serious adverse event, N=1 Paik et al. Abstract #9067, ASCO 2020 Voss et al. British Journal of Cancer (2020) 123:1590–1598 Hyperglycemia was reported as easily controlled with metformin therapy in Ph1 dose escalation and expansion2
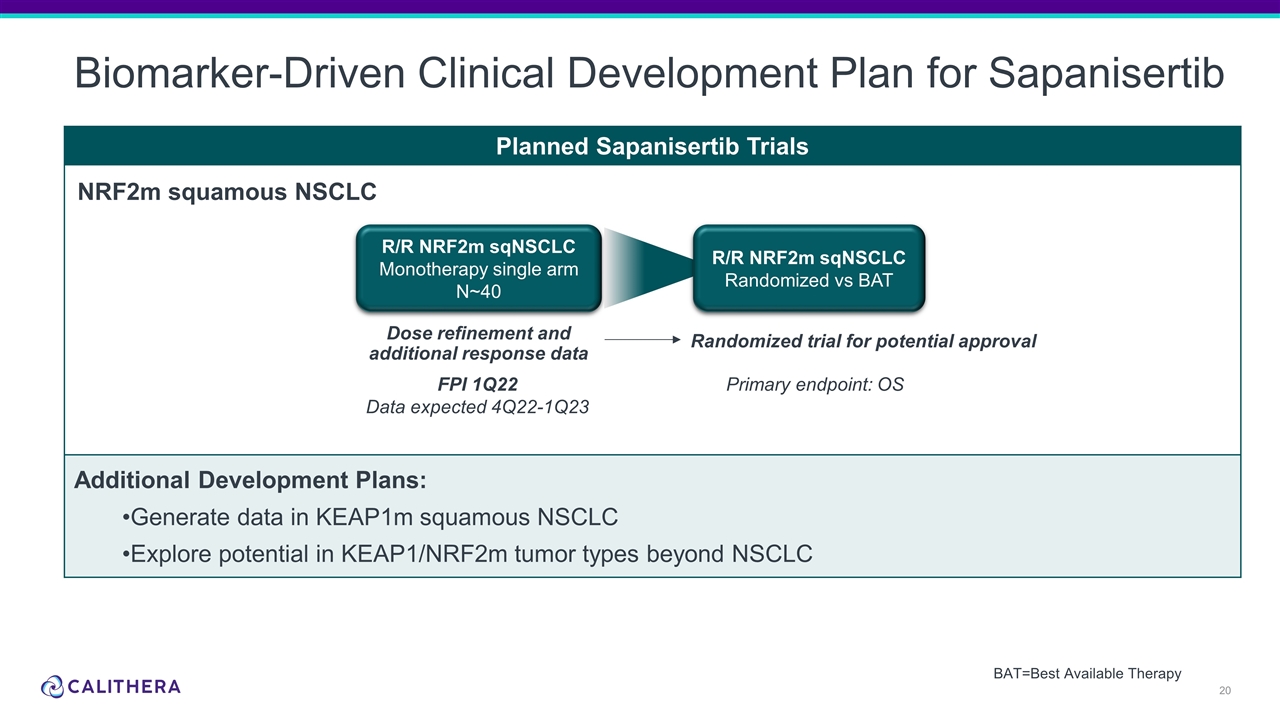
Planned Sapanisertib Trials Additional Development Plans: Generate data in KEAP1m squamous NSCLC Explore potential in KEAP1/NRF2m tumor types beyond NSCLC Biomarker-Driven Clinical Development Plan for Sapanisertib NRF2m squamous NSCLC Primary endpoint: OS R/R NRF2m sqNSCLC Monotherapy single arm N~40 BAT=Best Available Therapy Dose refinement and additional response data Randomized trial for potential approval R/R NRF2m sqNSCLC Randomized vs BAT FPI 1Q22 Data expected 4Q22-1Q23
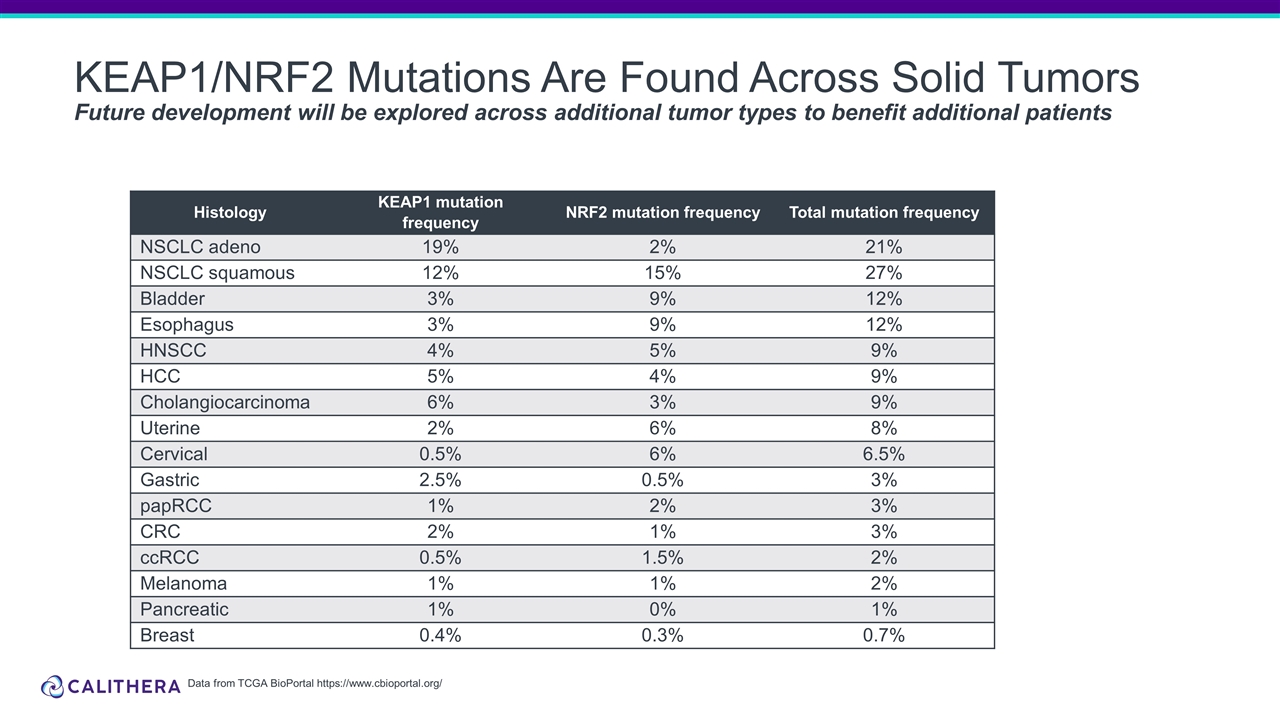
KEAP1/NRF2 Mutations Are Found Across Solid Tumors Future development will be explored across additional tumor types to benefit additional patients Histology KEAP1 mutation frequency NRF2 mutation frequency Total mutation frequency NSCLC adeno 19% 2% 21% NSCLC squamous 12% 15% 27% Bladder 3% 9% 12% Esophagus 3% 9% 12% HNSCC 4% 5% 9% HCC 5% 4% 9% Cholangiocarcinoma 6% 3% 9% Uterine 2% 6% 8% Cervical 0.5% 6% 6.5% Gastric 2.5% 0.5% 3% papRCC 1% 2% 3% CRC 2% 1% 3% ccRCC 0.5% 1.5% 2% Melanoma 1% 1% 2% Pancreatic 1% 0% 1% Breast 0.4% 0.3% 0.7% Data from TCGA BioPortal https://www.cbioportal.org/

Mivavotinib SYK inhibitor for hematologic malignancies

Mivavotinib Overview Mivavotinib is a SYK inhibitor with single-agent clinical activity and best-in-class potential in NHL Recent preclinical data identify NHL with MyD88 or CD79 mutations as particularly vulnerable to SYK inhibition2 Preliminary mivavotinib data suggests early signal in NHL patients with CD79 mutations3 Potential fast-to-market development strategy in DLBCL patients with or without MyD88 and/or CD79 mutations Deep and durable single agent activity in unselected 3L+ DLBCL and other NHLs in completed Phase 1/2 clinical trials1 IP COM protection through 2036 US/2035 EU assuming full 5-year patent term extension4 m=mutation r=rearrangement COM=composition of matter Gordon at al. Clin Cancer Res. Jul 15;26(14):3546-3556. AACR 2020. Munshi et al. Blood Cancer J. Jan 31;10(1):12. 2020 Takeda data on file US 8,440,689 B2 and WO 2011/079051 Potential First-to-Market approach for DLBCL patients with MyD88/CD79 mutations
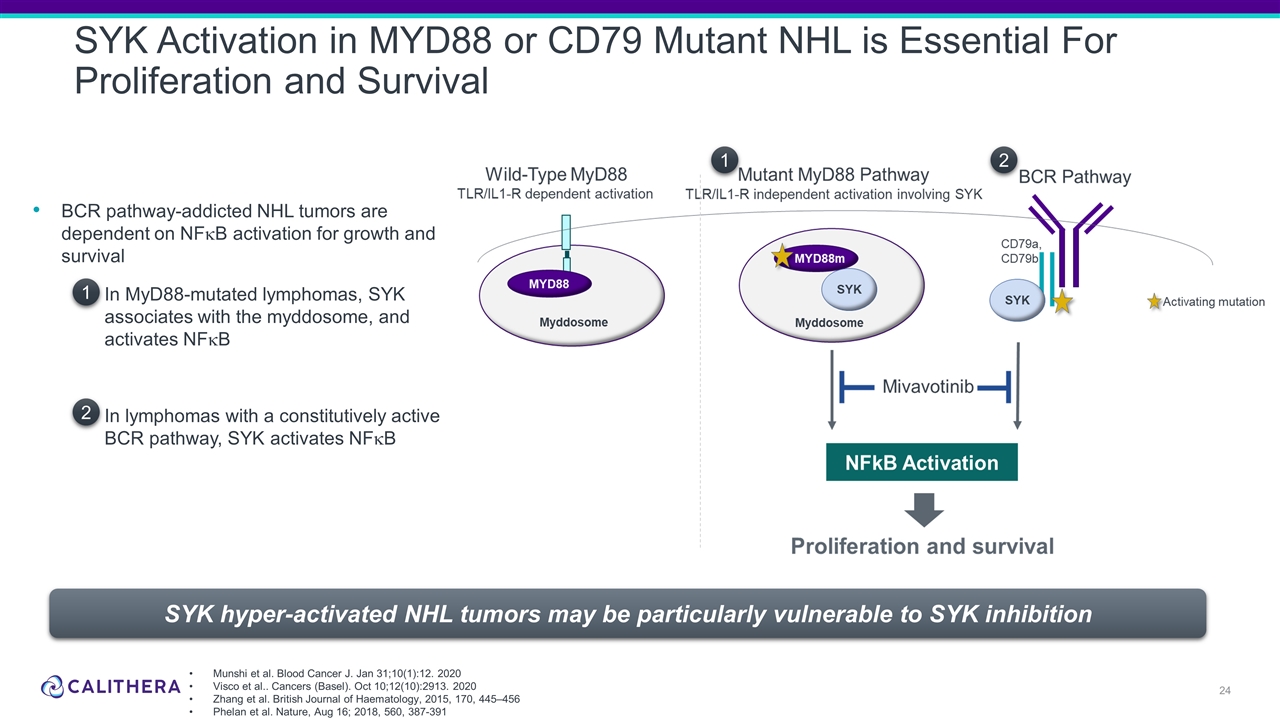
SYK Activation in MYD88 or CD79 Mutant NHL is Essential For Proliferation and Survival BCR pathway-addicted NHL tumors are dependent on NFkB activation for growth and survival In MyD88-mutated lymphomas, SYK associates with the myddosome, and activates NFkB In lymphomas with a constitutively active BCR pathway, SYK activates NFkB 1 2 1 2 SYK hyper-activated NHL tumors may be particularly vulnerable to SYK inhibition Munshi et al. Blood Cancer J. Jan 31;10(1):12. 2020 Visco et al.. Cancers (Basel). Oct 10;12(10):2913. 2020 Zhang et al. British Journal of Haematology, 2015, 170, 445–456 Phelan et al. Nature, Aug 16; 2018, 560, 387-391

Significant Number of DLBCL and Waldenstrom’s Patients Have MyD88/CD79 Mutations Vulnerable to SYK Inhibition Preclinical validation MyD88/CD79m DLBCL cell lines have high levels of activated SYK and are sensitive to inhibition by SYK inhibitors DLBCL represents the most common subtype of NHL ~30% of all NHL cases1 ~30,000 people diagnosed in the US each year with ~60% 5 year survival1,2 MyD88 mutations occur in ~30% DLBCL3-6 CD79 mutations occur in 10-15% of DLBCL3-6 Waldenstrom macroglobulinemia (WM) is a rare type of lymphoma with 1,000-1,500 people diagnosed each year in the US7 36% 5 year survival for patients with high risk7 90% of patients have mutations in MyD883,7 https://seer.cancer.gov/statfacts/html/dlbcl.html Decision Resources Group, epidemiology, DLBCL incidence 2021 de Groen et al. Haematologica. Dec;104(12):2337-2348. 2019 Visco et al.. Cancers (Basel). Oct 10;12(10):2913. 2020 Munshi et al. Blood Cancer J. Jan 31;10(1):12. 2020 Vermaat et al. Haematologica 2020; 105(2):424-434 https://www.cancer.org/cancer/waldenstrom-macroglobulinemia/about/key-statistics.html Varettoni et al. Haematologica 2017 Volume 102(12):2077-2085
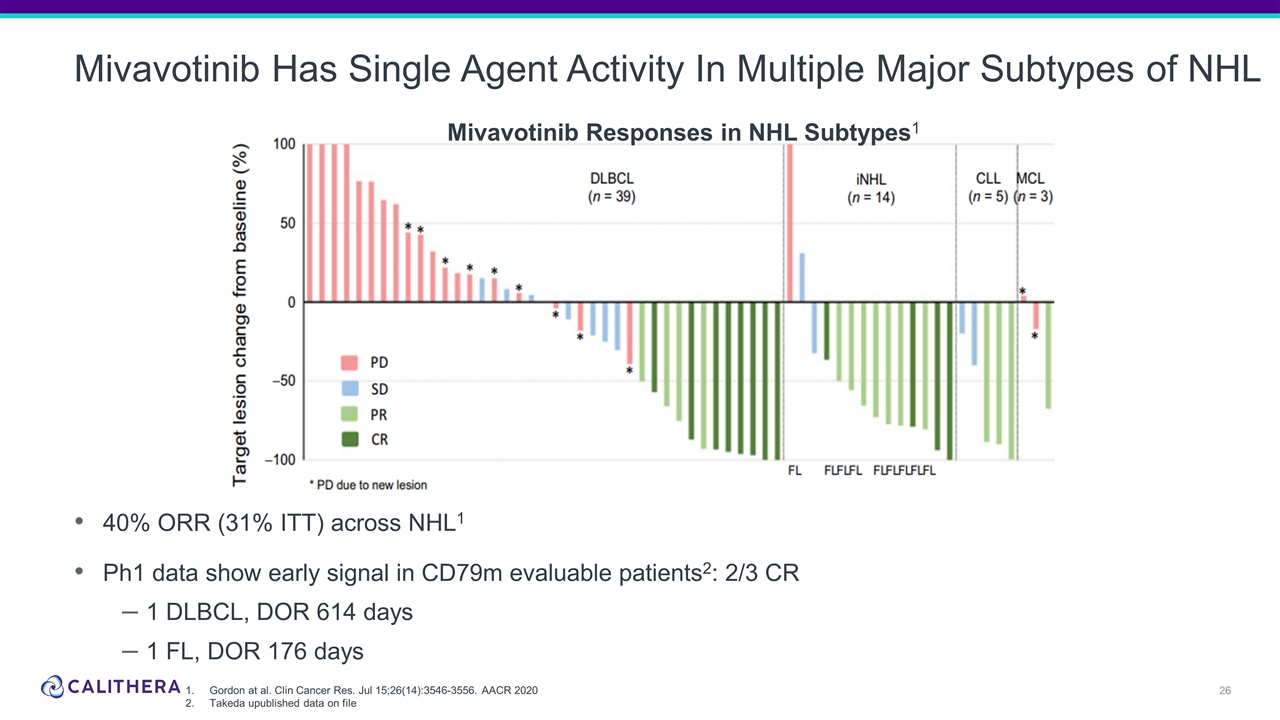
Mivavotinib Has Single Agent Activity In Multiple Major Subtypes of NHL 40% ORR (31% ITT) across NHL1 Ph1 data show early signal in CD79m evaluable patients2: 2/3 CR 1 DLBCL, DOR 614 days 1 FL, DOR 176 days Mivavotinib Responses in NHL Subtypes1 Gordon at al. Clin Cancer Res. Jul 15;26(14):3546-3556. AACR 2020 Takeda upublished data on file
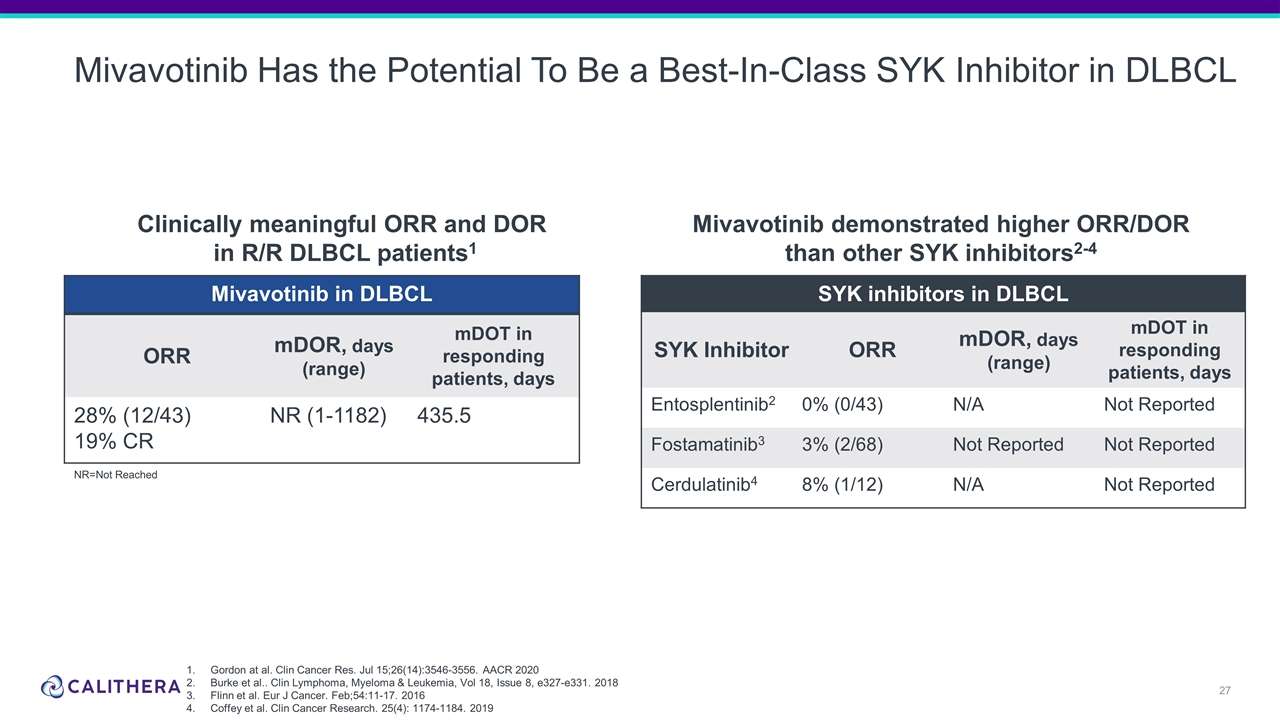
Mivavotinib Has the Potential To Be a Best-In-Class SYK Inhibitor in DLBCL Clinically meaningful ORR and DOR in R/R DLBCL patients1 Mivavotinib in DLBCL ORR mDOR, days (range) mDOT in responding patients, days 28% (12/43) 19% CR NR (1-1182) 435.5 Gordon at al. Clin Cancer Res. Jul 15;26(14):3546-3556. AACR 2020 Burke et al.. Clin Lymphoma, Myeloma & Leukemia, Vol 18, Issue 8, e327-e331. 2018 Flinn et al. Eur J Cancer. Feb;54:11-17. 2016 Coffey et al. Clin Cancer Research. 25(4): 1174-1184. 2019 Mivavotinib demonstrated higher ORR/DOR than other SYK inhibitors2-4 NR=Not Reached SYK inhibitors in DLBCL SYK Inhibitor ORR mDOR, days (range) mDOT in responding patients, days Entosplentinib2 0% (0/43) N/A Not Reported Fostamatinib3 3% (2/68) Not Reported Not Reported Cerdulatinib4 8% (1/12) N/A Not Reported
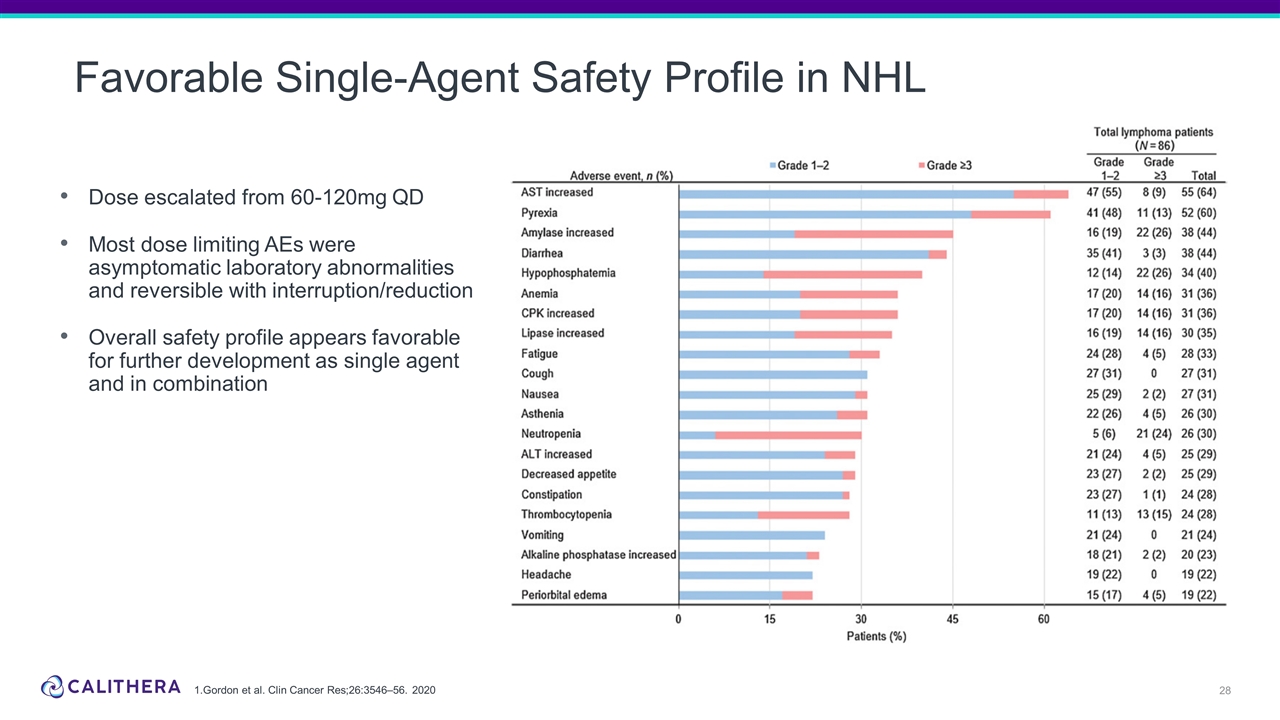
Favorable Single-Agent Safety Profile in NHL Dose escalated from 60-120mg QD Most dose limiting AEs were asymptomatic laboratory abnormalities and reversible with interruption/reduction Overall safety profile appears favorable for further development as single agent and in combination 1.Gordon et al. Clin Cancer Res;26:3546–56. 2020
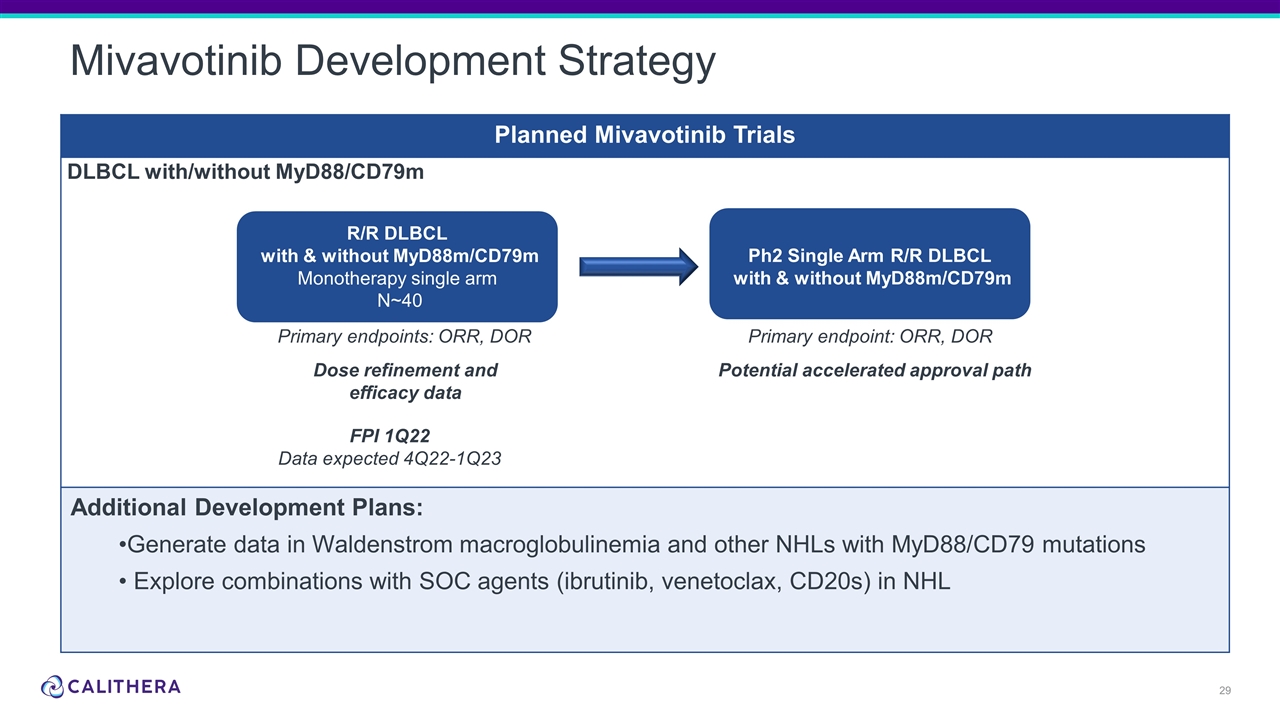
Mivavotinib Development Strategy Planned Mivavotinib Trials Additional Development Plans: Generate data in Waldenstrom macroglobulinemia and other NHLs with MyD88/CD79 mutations Explore combinations with SOC agents (ibrutinib, venetoclax, CD20s) in NHL DLBCL with/without MyD88/CD79m Primary endpoints: ORR, DOR R/R DLBCL with & without MyD88m/CD79m Monotherapy single arm N~40 Potential accelerated approval path FPI 1Q22 Data expected 4Q22-1Q23 Ph2 Single Arm R/R DLBCL with & without MyD88m/CD79m Primary endpoint: ORR, DOR Dose refinement and efficacy data

Arginase Inhibitor CB-280 Cystic Fibrosis, regardless of CFTR genotype

Rationale for Arginase Inhibitors in Cystic Fibrosis Despite recent advances with CFTR modulators, many patients still have impaired lung function Arginase plays a critical role in CF airway disease1, 2, 3 Decreases nitric oxide (NO) production4, increases production of polyamines and proline Inhibition of arginase should increase NO, increasing anti-microbial activity and improving airway function5 Potential for additional benefit when combined with standard of care therapies6 Potential benefit in all CF patients, regardless of CFTR genotype CB-280 is an investigational first-in-class orally-dosed arginase inhibitor in Phase 1b trials supported by a grant from the Cystic Fibrosis Foundation Grasemann et al., Am J Respir Crit Care Med 172: 1523-1528, 2005 Grasemann et al., Respiratory Research 7: 87, 2006 Jaecklin et al., J Appl Physiol 117: 284-288, 2014 Grasemann et al., Eur Respir J 25: 62-68, 2005 Mermis et al, NACF-2020-CX-280-202-TIP-POSTER-08Sep2020 Wu, Mol Pharmacol 96:515-525, 2019

CB-280 Randomized Ph1b Trial In Cystic Fibrosis Ongoing Randomized Double Blind Ph1b Trial in CF Placebo Cystic Fibrosis N = approximately 32 ≥ 18 years old FEV1 40-90% predicted On stable regimen of CF therapies Chronic microbial infection Trial initiated July 2020 CB-280 Dose Escalation Cohorts Primary Endpoint: Safety Secondary Endpoints: PK/PD: arginase inhibition and arginine increase assessed in plasma and sputum Exploratory Endpoints: FENO, sweat chloride, sputum colonization Interim Analysis will be presented at NACFC in November 2021 Randomized 3:1 Potential to benefit all CF patients, regardless of CFTR genotype, as the first-in-class orally dosed arginase inhibitor
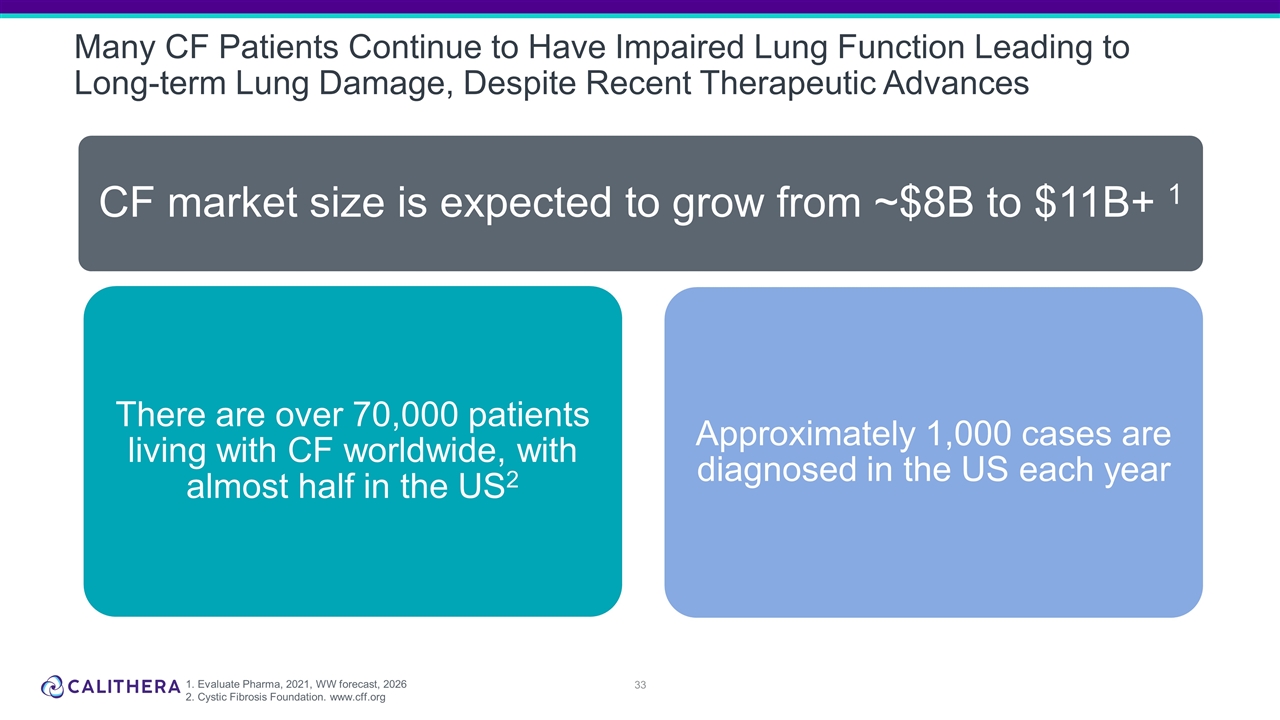
Many CF Patients Continue to Have Impaired Lung Function Leading to Long-term Lung Damage, Despite Recent Therapeutic Advances 1. Evaluate Pharma, 2021, WW forecast, 2026 2. Cystic Fibrosis Foundation. www.cff.org CF market size is expected to grow from ~$8B to $11B+ 1 Approximately 1,000 cases are diagnosed in the US each year There are over 70,000 patients living with CF worldwide, with almost half in the US 2

Well Positioned for Success

Interim data read outs in Q4 Data expected 4Q22-1Q23 Calithera Milestones Legacy Clinical Programs New Clinical Programs Robust Discovery Engine Telaglenastat (CB-839): KEAPSAKE interim data expected 4Q 2021 CB-280: Phase 1b interim data at North American Cystic Fibrosis Conference November 2021 Sapanisertib trial in NRF2m squamous R/R NSCLC Mivavotinib trial in R/R DLBCL with and without MyD88m/CD79m Preclinical pipeline of undisclosed synthetic lethality targets with a focus on paralog genes to be announced as lead molecules are identified and IND timelines are in focus