 Targeting Disease at the Nuclear Pore Corporate Presentation November 2015 NASDAQ: KPTI Exhibit 99.1 |
 2 ©2015 – Karyopharm Therapeutics, Inc. Forward-looking Statements This presentation contains forward-looking statements within the meaning of the “safe harbor” provisions of The Private Securities Litigation Reform Act of 1995. Unless otherwise noted, this presentation contains data that are interim and unaudited based on site reports. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from the company’s current expectations. For example, there can be no guarantee that Selinexor or any other drug candidate Karyopharm is developing will successfully complete necessary preclinical and clinical development phases or that development of any of Karyopharm’s drug candidates will continue. Further, there can be no guarantee that any positive developments in Karyopharm’s drug candidate portfolio will result in stock price appreciation. In addition, even if Karyopharm receives marketing approval for selinexor or another drug candidate, there can be no assurance that Karyopharm will be able to successfully commercialize that drug candidate. Management’s expectations and, therefore, any forward-looking statements in this presentation could also be affected by risks and uncertainties relating to a number of other factors, many of which are beyond Karyopharm’s control, including the following: Karyopharm’s results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; the content and timing of decisions made by the U.S. Food and Drug Administration and other regulatory authorities, investigational review boards at clinical trial sites and publication review bodies; Karyopharm’s ability to obtain and maintain requisite regulatory approvals and to enroll patients in its clinical trials; unplanned cash requirements and expenditures; development of drug candidates by Karyopharm’s competitors for diseases for which Karyopharm is currently developing its drug candidates; and Karyopharm’s ability to obtain, maintain and enforce patent and other intellectual property protection for any drug candidates it is developing. These and other risks, including those which may impact management’s expectations, are described in greater detail under the heading "Risk Factors" in Karyopharm’s Quarterly Report on Form 10-Q for the quarter ended September 30, 2015, which is on file with the Securities and Exchange Commission, and in subsequent filings filed by Karyopharm with the Securities and Exchange Commission. Any forward-looking statements contained in this presentation are for informational purposes only and speak only as of the date hereof. Other than as is required by law, Karyopharm expressly disclaims any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise. Karyopharm’s website is http://www.karyopharm.com. Karyopharm regularly uses its website to post information regarding its business, drug development programs and governance. Karyopharm encourages investors to use www.karyopharm.com, particularly the information in the section entitled “Investors,” as a source of information about Karyopharm. References to www.karyopharm.com in this presentation are not intended to, nor shall they be deemed to, incorporate information on www.karyopharm.com into this presentation by reference. Such forward-looking statements include those regarding the therapeutic potential of and potential clinical development plans and commercialization for Karyopharm’s lead drug candidate, selinexor (KPT-330), including the timing of initiation of certain trials and of the reporting of data from such trials, other drug product candidate development plans, and assumptions of management regarding strategic and financial expectations and projections. |
 3 Novel mechanism First in class Oral, small molecule Fully Owned Topline data in lead indications expected Q4 ‘16 Tumor Suppressor Protein (TSP) activation and oncoprotein reduction Potentially relevant to any type of cancer 4 ongoing later stage Heme trials 3 ongoing Ph 2 Solid Tumor trials >40 combination studies ongoing or planned Prepare to commercialize selinexor in NA and Western EU Seek collaborators for other geographies Selinexor New Drug Class in Oncology Commercial Strategy Clinical Development Strategy Initial focus on the regulatory approval and commercialization of oral selinexor as a single-agent, while developing SINE™ compounds as novel approaches to treat a variety of diseases in areas of unmet medical need Karyopharm’s wholly owned, lead SINE™ compound selinexor offers an entirely new approach to cancer therapy that restores the ability of a cell to detect cancerous changes and commit suicide ©2015 – Karyopharm Therapeutics, Inc. Karyopharm: At the Nucleus of Cancer Care |
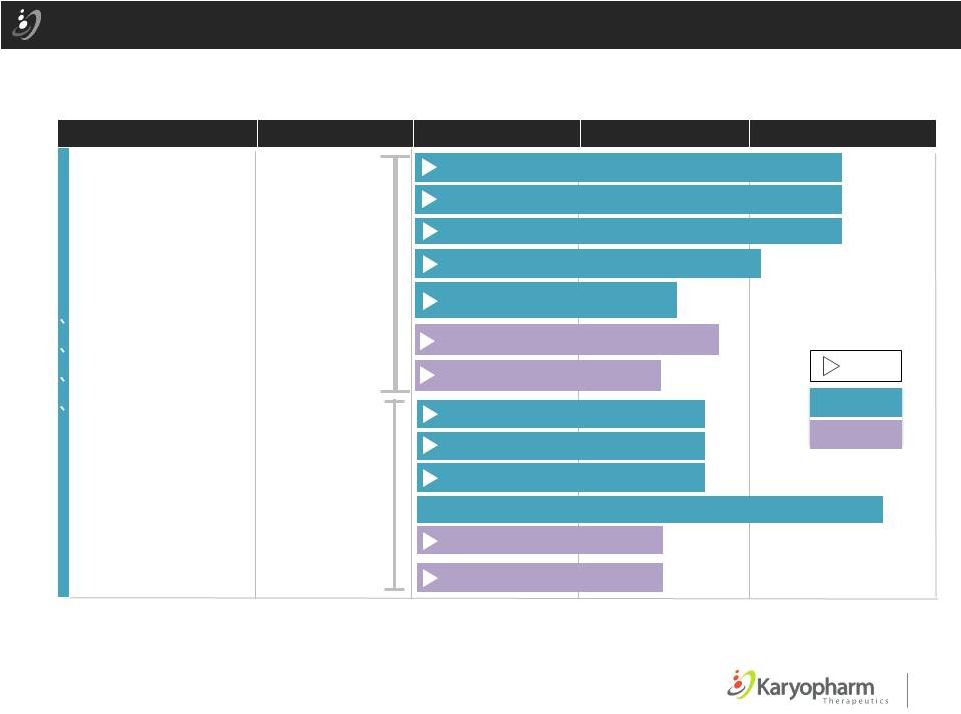 4 Selinexor: SINE™ Compound with Broad Clinical Development Strategy Oral Selinexor (KPT-330) Hematological Malignancies AML (SOPRA) DLBCL (SADAL) Richter’s (SIRRT) Multiple Myeloma (STOMP) Sel+Pom+Dex; Sel+Bort+Dex; Sel+Len+Dex Gynecologic Malignancies (SIGN) Prostate (SHIP) Glioblastoma (KING) PRECLINICAL PHASE 1 & PHASE 2 PHASE 2 or 3 AREA OF THERAPY DRUG CANDIDATE ` ` ` ` Solid Tumors = Initiated Multiple Myeloma (STORM) Liposarcoma Ovarian or Endometrial Selinexor + Paclitaxel + Carboplatin Company Sponsored Investigator Sponsored Various Solid Tumors Standard Chemotherapy Regimens Various Hematological Malignancies Combination Regimens Multiple Myeloma Sel+Carfil+Dex |
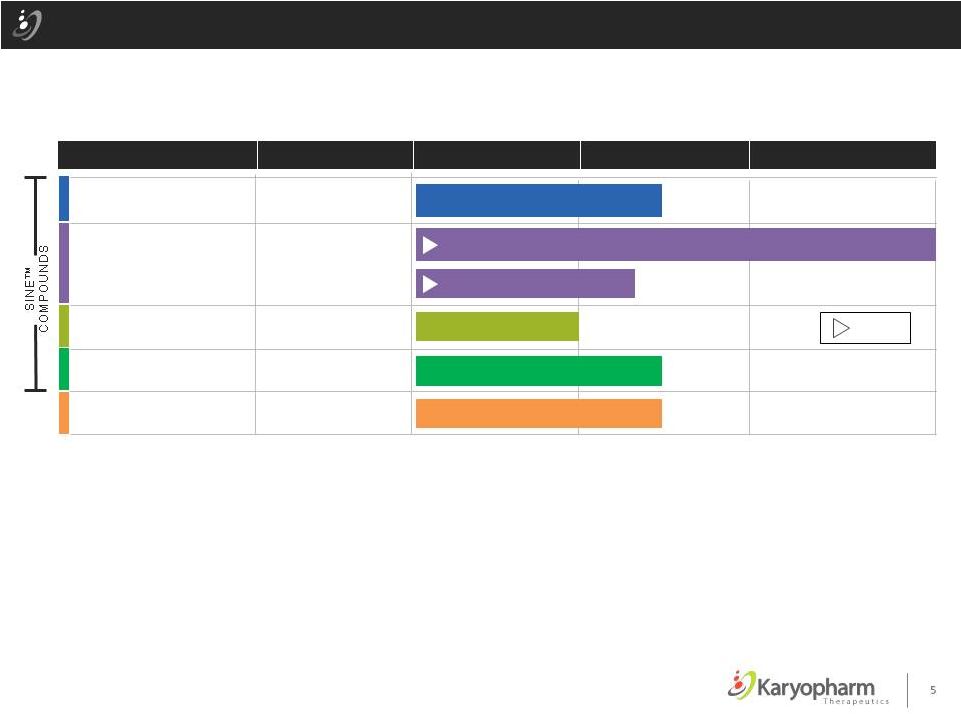 Karyopharm’s Broad Therapeutic Pipeline Topical Selinexor (KPT-330) Oral Verdinexor (KPT-335) Oral KPT-8602 PRECLINICAL PHASE 1 & PHASE 2 REGISTRATION AREA OF THERAPY DRUG CANDIDATES Wound Healing Diabetic Foot Ulcers Influenza Antiviral Oral KPT-350 Inflammation & Autoimmune ALS, TBI, MS, SLE Oncology Multiple Myeloma = Initiated Hematological Malignancies Canine Lymphoma – NADA Submission in Progress (MUMS designation received) PAK4 Inhibitors Oncology Lymphoma & Solid Tumors 5 |
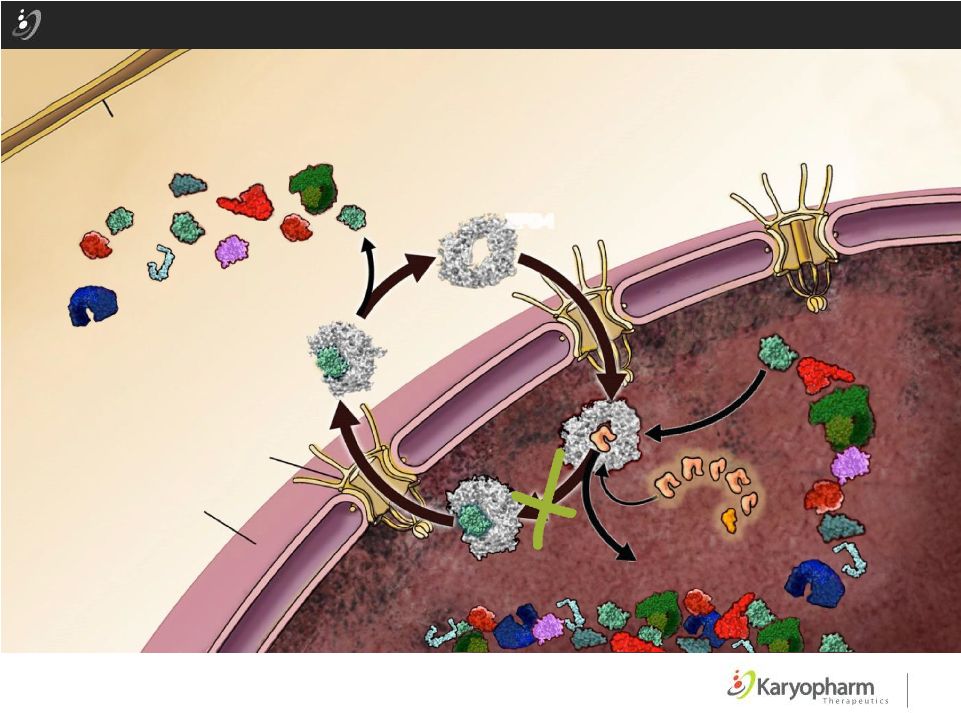 Selinexor: Novel Anti-Cancer Agent: Restores Tumor Suppressors & Reduces Oncoproteins Tumor Suppressors Cell Membrane Nuclear Envelope Nuclear Pore Complex XPO-1 CYTOPLASM ©2015 – Karyopharm Therapeutics, Inc. 6 SINE p53 elF4E (myc,bcl2 mRNA) pRB PP2A Par-4 p21 IkB BRCA1 p27 Suppressors Tumor Tumor Suppressors |
 Initial Focus for Single-Agent Approval of Selinexor in: • DLBCL • Acute Myeloid Leukemia • Multiple Myeloma • Richter’s Transformation ©2015 – Karyopharm Therapeutics Inc. 7 |
 Selinexor’s Broad and Durable Oral Single-Agent Anti-Tumor Activity Patients treated to date with single agent or in combination 1,250+ Selinexor Broad and durable single-agent anti-tumor activity in a pill Broad and durable single-agent anti-tumor activity in a pill Disease control across hematologic and solid tumor malignancies Durability – some patients on >1 year (longest 2+ years) Recommended Phase 2 dose for most indications is 60mg twice weekly – generally well-tolerated up to 50% >6 month Dosing ©2015 – Karyopharm Therapeutics, Inc. 8 |
 9 Aggressive form of Non-Hodgkin’s Lymphoma (NHL) Incidence: 25,000 new cases annually in the US • ~40% of incident patients will succumb to their disease • ~10,000 deaths per year in USA (~22,500 worldwide) Greatest unmet need currently in GCB and Double-Hit Subtypes of DLBCL Selinexor has shown durable single-agent activity across all forms of DLBCL Diffuse Large B-Cell Lymphoma: The Opportunity ©2015 – Karyopharm Therapeutics, Inc. Citations: ACS and Leukemia and Lymphoma Society, Pasqualucci et al, 2013 |
 10 ©2015 – Karyopharm Therapeutics, Inc. Best Responses in DLBCL Patients • 31% ORR and 51% DCR for all evaluable DLBCL patients • 43% ORR and 71% DCR for evaluable DLBCL patients on study > 1 month • ORR and DCR are comparable across DLBCL origin or subtype • Duration of response was >9 months • Responses were also observed in “double-hit” DLBCL Category Total Evaluable ORR CR PR SD PD DCR All Patients 39* 31% 4 (10%) 8 (21%) 8 (21%) 19 (49%) 51% Patients on study > 1 Month 28 43% 4 (14%) 8 (29%) 8 (29%) 8 (29%) 71% Origin De novo 28 25% 3 (11%) 4 (14%) 6 (21%) 15 (54%) 46% Transformed 11 45% 1 (9%) 4 (36%) 2 (18%) 4 (36%) 64% Subtype GCB 14 43% 3 (21%) 3 (21%) 5 (36%) 3 (21%) 79% non-GCB 4 25% 1 (25%) -- 3 (75%) -- 100% *Three patients were non-evaluable for response due to consent withdrawal with lack of disease assessment prior to one cycle on study. Responses (as of 1-June-2015) were adjudicated according to the International Working Group Response Criteria for Non-Hodgkin’s Lymphoma (NHL) 2007 based on interim unaudited data. ORR=Objective Response Rate (CR+PR), CR=Complete Response, PR=Partial Response, SD=Stable Disease, PD=Progressive Disease, DCR=Disease Control Rate (CR+PR+SD) GCB=Germinal Center B Cell. GCB/non-GCB subtypes were not defined for all patients. All patients |
 Selinexor in DLBCL: Promising Responses in Single Hit and Double Hit Patients (ICML) ©2015 – Karyopharm Therapeutics, Inc. 11 |
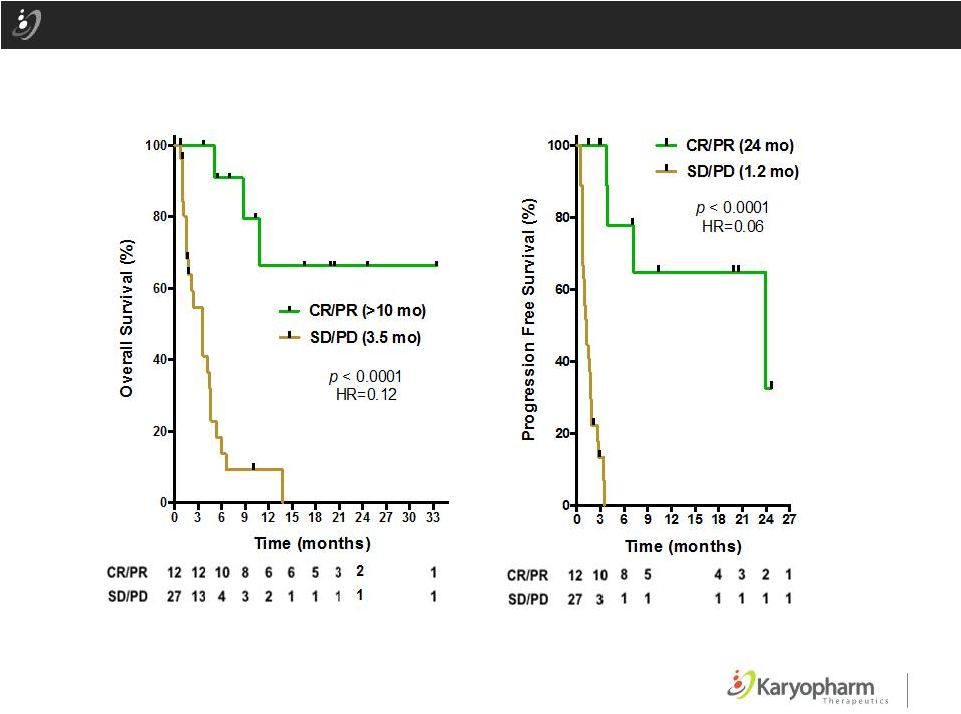 Phase 1 DLBCL Data: Response to Selinexor Correlates with Overall Survival (EHA 2015) Data as of May 16, 2015 ©2015 – Karyopharm Therapeutics, Inc. Presented at the 20 Congress of the European Hematology Association, Vienna, Austria 2015 12 th OS (CR/PR vs SD/PD) PFS (CR/PR vs SD/PD) Patients at risk |
 SADAL: Selinexor Against Diffuse Aggressive Lymphoma Ongoing Randomized Trial for Accelerated Approval • Relapsed / Refractory >3rd line • Twice-weekly randomized single-agent selinexor 1:1: selinexor 60 mg vs. selinexor 100 mg • >50% of patients with GCB-DLBCL • Targeting ~200 patients • Primary Endpoint: Overall Response Rate • Data read out anticipated in Q4 2016 SADAL: Randomized Phase 2 trial in DLBCL ©2015 – Karyopharm Therapeutics, Inc. 13 |
 Potential Selinexor NHL Development 2014 H1 H2 2015 H1 H2 2016 H1 H2 2017 H1 H2 Slide Key * Investigator Sponsored CHOP-R Cyclophosphamide, Doxorubicin Hydrochloride (Hydroxydaunomycin), Vincristine Sulfate (Oncovin), Predisone, Rituxan Dex dexamethasone Ibrut IMBRUVICA® (ibrutinib) Ritux Rituxan®(rituximab) R-ICE Rituxan, fosfamide, carboplatin, etoposide Ph 1 Sel + Ritux in NHL Ph 2 Selinexor in PTCL and CTCL SADAL: Ph 2 Selinexor in DLBCL (high vs. low Sel dose) Ph 2 Selinexor in Richter’s transformation (single arm) Ph 1/2 (2 /3 Line) Sel + Ritux + X in NHL Ph 1/2 Sel + Ibrut in NHL + CLL Ph 1/2 (2 Line) Sel + R-ICE in DLBCL Ph 1/2 (1 /2 Line) Sel + CHOP-R in NHL Phase 3 Studies: Randomized Studies with Selinexor- Rituximab in Combination * * * ©2015 – Karyopharm Therapeutics, Inc. nd st rd nd 14 nd |
 Multiple Myeloma represents a significant patient population for selinexor Second most commonly diagnosed blood cancer, after NHL • 114,000 new cases worldwide each year • In the US, a prevalence of approximately 83,000, with 24,050 new cases and 11,090 deaths in 2014 • Initial focus on patients with recurrent disease following multiple lines of therapy Unmet need for patients with recurrent or refractory MM for patients after proteasome inhibitors and immunomodulatory drugs (IMIDs) Multiple Myeloma: The Opportunity Citations: ACS Facts and Figures 2014; GLOBOCAN 2012, IARC, http:///seer.cancer.gov/ ©2015 – Karyopharm Therapeutics, Inc. 15 |
 STORM: Selinexor Treatment of Refractory Myeloma • Planned Single Arm Trial for Relapsed/Refractory after > 4 approved agents • Quadruple drug exposed, dual-class refractory: REVLIMID®, POMALYST®, KYPROLIS® and VELCADE® • Initiated May 2015, ~80 patients • Primary Endpoint: Overall Response Rate • Interim data anticipated mid-2016; Study may be expanded to >200 patients for potential accelerated approval STOMP: Selinexor in Combination w Backbone Treatments for Relapsed/Refractory Multiple Myeloma • Initiated in October 2016 • Sel+Pom+Dex & Sel+Bortez+Dex & Sel+Len+Dex • Striking preclinical synergy in animal models • Dose escalation study, targeting up to 220 patients • “Pick the winner” in advance of phase 3 studies • Data anticipated in 2017 STORM & STOMP ©2015 – Karyopharm Therapeutics, Inc. 16 |
 17 Selinexor + Dexamethasone • Phase 1/2 combination study with ten evaluable patients treated with selinexor (45 mg/m ) + low dose dexamethasone (20 mg) • Heavily pretreated with a median of 7 prior therapies • In ten patients: 6 responses (5 PR, 1 CR), 2 MR, 1 PD, 1 not evaluable, Median DOR ~ 7 months Multiple Myeloma: Phase 1 Data 76 IgG- –71% PR 7 Dox-Vinc-Dex, Thal-Dex, Carfil-Dex, VRD, Cyclo-Pred-BCNU, Sel, Doxil-Carfil-Dex 391 77 FLC- -- NE 8 Len-Dex, Cyclo-Etop-Cis-Mel-Dex-ASCT, VRD, Carfil-Cyclo-Dex, Carfil-Cyclo-Dex-Len, Carm-Thal-Cis-Etop-Cyta-Vel-Mel, Cyclo-Carfil-Pom-Dex, Vor-Len-Dex 15 79 FLC- –53% PR 3 Thal-Pred-Dex-ASCT, Cyclo-Vel-Dex, Len-Dex 52 81 FLC- –99% sCR 6 VAD-ASCT, Cyclo-Pred-ASCT, Rev-Dex, Cyclo-BorD (x2), Pom-Carfil-Dex 280 84 IgG- –84% PR 9 Vel-Dex, ASCT, Len-Dex, Vel-Dex, Vel, Carfil, Pom-Dex, Carfil, DT-PACE-Thal 170 90 IgG- 41% PD 5 Cyclo-Vel-Len-Dex (x2), Carfil-Mel-ASCT, Cyclo-Vel-Dex, Pom-Carfil-Dex 31 92 IgA- –55% PR 9 Vel-Dex, VRD, ASCT, Len-Dex, Reolysin, TG02, Carfil-Dex, Carfil-Cyclo-Dex, Carfil-Pom-Dex 121 93 IgG- –41% MR 9 VAD, VTD+ASCT, Vel-Len-Dex, Experim, Carfil-Panob, Len-Elotu-Dex, Experim, Pom-Dex, Benda-Pom-Dex 114 98 IgG- –48% MR 16 Len-Dex, ASCT (x2), Vel-Len-Dex, Vid-Len, Benda-Vel-Dex, VAD, Ritux, Vel-Thal, Carfil-Dex, Carfil-Dex-Cis- Adria, Len-Ritux-Inter, Carfil-Pom, Vel-Thal-Dex-Adria-Cis-ATRA-Arsenic Trioxide, Len-Dex, TG02-Carfil 79 99 IgA- –82% PR 6 Sal, Thal-Dex, Len-Dex, Vel-Dex, ASCT, Ibrut-Dex 285 ©2015 – Karyopharm Therapeutics, Inc. Prior Therapies (As of 10-May-2015) Group A Patients with Rel/Ref MM Treated with Twice Weekly Oral Combination – Selinexor 45 mg/m + Dexamethasone 20 mg Study Days Patient ID MM Type Maximal Response # Prior Tx 2 2 |
 18 Additional patient responses to be presented at ASH 2015 Promising Potential in Patients with Carfilzomib-Refractory MM SELINEXOR + CARFILZOMIB IN PATIENTS WITH CARFILZOMIB-REFRACTORY MM (Selinexor 30 mg/m 2 + Carfilzomib (20/27 mg/m 2 ) Patient Age Days on Tx Best Response # Prior Tx 59 113+ VGPR 4 73 85+ PR 2 64 71+ PR 5 Data as of November 1, 2014 Presented at ASH 2014 ©2015 – Karyopharm Therapeutics, Inc. |
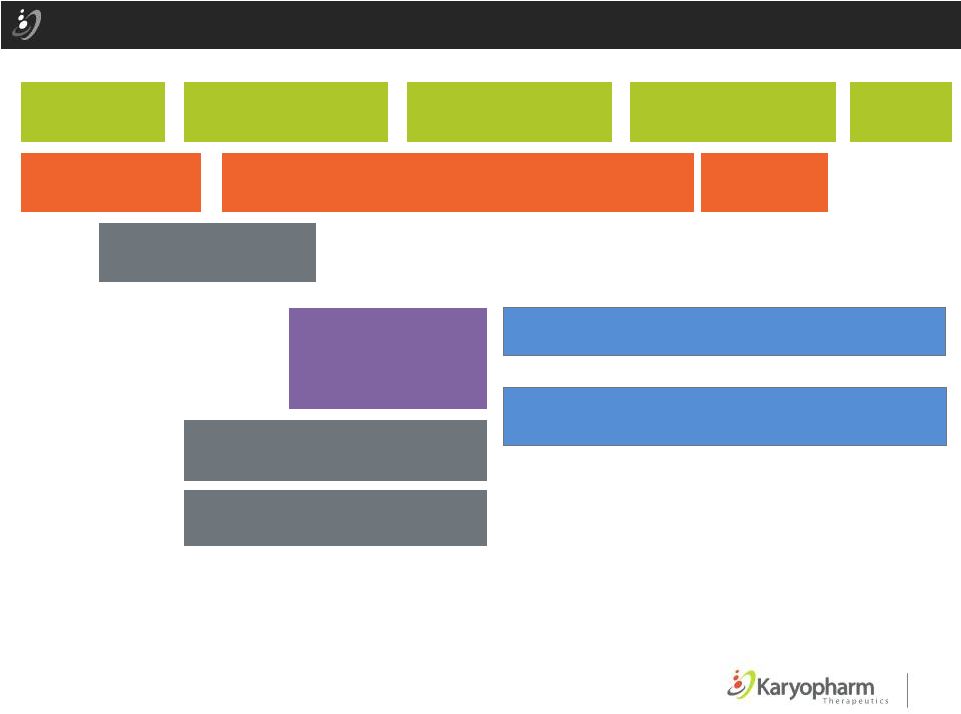 19 Potential Selinexor MM Development 2014 H2 2015 H1 H2 2016 H1 H2 2017 H1 H2 Ph 1 Selinexor + Dex STORM: Ph 2 Selinexor + Low Dose Dex (80 patients initially; potential upsize) Ph 1/2 Selinexor + Carfil + Dex STOMP: Ph 1/2 Selinexor + Pom + Dex Selinexor + Bortez + Dex Selinexor + Len + Dex Ph 1/2: Pre-Transplant Selinexor + Bortez + Dex Ph 3: Selinexor + Pom + Dex vs. Pom + Dex Prep/Submit NDA/EMA Ph 3: Selinexor + PI (Carfil / Bortez / Len) + Dex vs. PI + Dex AND/OR Ph 1/2 Selinexor + PLD * * * 2018 H1 LAUNCH ©2015 – Karyopharm Therapeutics, Inc. Slide Key * Investigator Sponsored Bortez bortezomib (Velcade®) Carfil carfilzomib (Kyprolis®) Dex dexamethasone PI proteasome inhibitor PLD pegylated liposomal doxorubicin Pom pomalidomide Rev Revlimid®(lenalidomide) |
 20 Significant opportunity exists for selinexor in AML as few treatment options exist for patients • Approximately 18,860 new diagnoses of AML and approximately 7,330 deaths per year in the US • The average age of a patient with AML is 66 Older patients with relapsed AML have limited treatment options with poorer outcomes than younger patients due to comorbidities and increased resistance to chemotherapy • Median Survival of older patients unfit for chemotherapy is ~9 months Approval Plans • SOPRA: Older patients with AML after at least 1 line of therapy; topline survival data end of 2016 • Combination studies ongoing to inform use in first line therapy for older patients Additional opportunities for younger patients with “high risk” AML in front line • About 20% of patients at first diagnosis • All patients with relapse after initial chemotherapy • Pediatric patients with relapsed disease (>50% of pediatric AML) Acute Myeloid Leukemia: The Opportunity Citations: ACS Facts and Figures 2014 ©2015 – Karyopharm Therapeutics, Inc. |
 SOPRA: Selinexor in Older Patients with Relapsed/Refractory AML Ongoing Randomized Study in AML in pts > 60 years old – after at least one therapy • Primary Endpoint: Overall Survival • Randomized 2:1 – single-agent selinexor (60mg fixed dose) vs. “Physician’s Choice” (hypomethylating agents or LDAC or Supportive Care only) • Dose adjusted in July 2015; ~170 patients • A trend towards an increase in the frequency of sepsis was identified in elderly patients with relapsed or refractory AML receiving very high doses of selinexor (typically ~100 mg). There was no apparent increase in the risk of neutropenia or febrile neutropenia in these patients. • Interim analysis in mid-2016; Full top-line data read out anticipated Q4 2016 SOPRA: Randomized Phase 2 Trial in 2 Line AML ©2015 – Karyopharm Therapeutics, Inc. 21 nd |
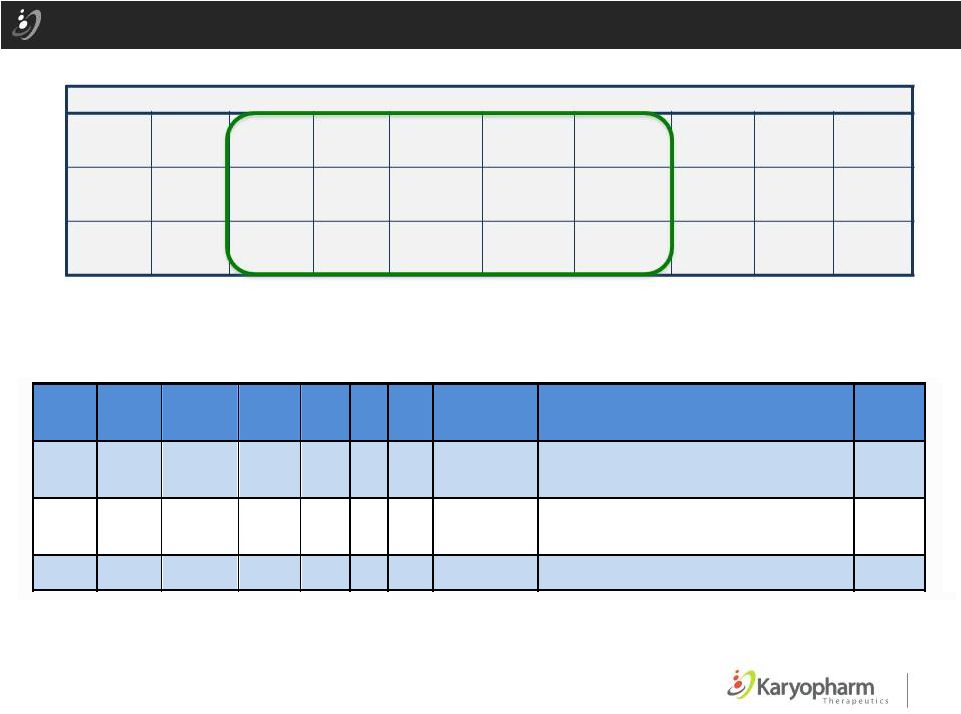 Selinexor AML Phase 1 Study: Activity in Relapsed/Refractory AML N DCR ORR CR CR(i) PR MLFS SD PD NE 65 32 10 5 2 1 2 22 16 17 % 49% 15% 8% 3% 2% 3% 34% 25% 26% Selinexor Phase 1 AML Patients: Prolonged SD ©2015 – Karyopharm Therapeutics, Inc. Pat ient ID Age 110 73 147 79 149 63 mg/m 23 40 70 ose ( ) Doses/ ycle MDS Flt3 NPM Cy ogenic k D 2 C rior t Ris Pr ior Ther pies Da s on St udy 10 N N N Favo abl T, FL A, FIL, VID CY U, ID , DA , MIT 312 8 Y N N Inter me di ate 132+ 8 N N N Inter me di te DAU , CY T, EP O, M IT, RO S 120+ P a y r e U No Pr or i s a 22 Best Responses in Patients with AML as 10-June-2014 |
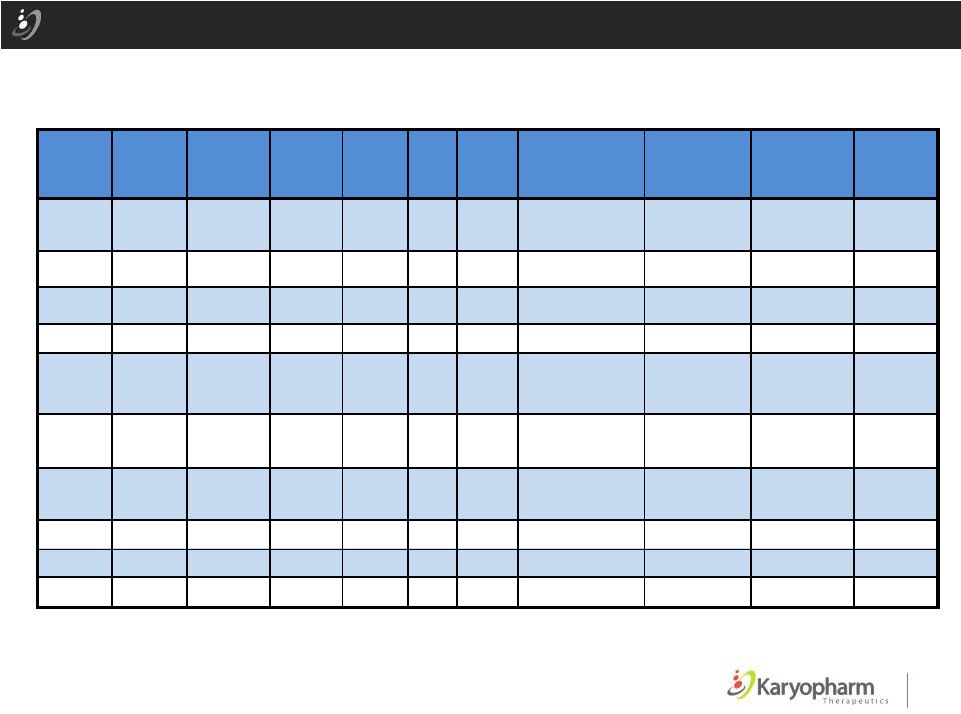 Selinexor Phase 1 AML Patients with Responses ©2015 – Karyopharm Therapeutics, Inc. 23 Pa tie nt Age 138 39 40 8 N N N 111 70 23 10 Y N N 505 82 40 8 N N N 501 71 16 .8 10 N Y Y 133 81 40 8 N N N 2 ID Dose mg/m Dose s/ Cyc le Adverse me rab 102 70 6.8 10 N N N In medi ( ) MDS Flt3 NPM Cy ogenic Risk ior Ther pies sponse Da ys on udy CR(i) 81 150 61 40 8 N Unk Unk CR( i) 119+ 121 77 30 8 N N N MLFS 170 155 83 40 8 Y N N ter MLFS 92+ 114 70 30 8 N N N PR 87 In me ate AU, C YT Adverse VID, CYT In medi ID In rm edi ate CYT, IDA te V , DEC te C ter di D a Pr ior t r a e t P R S 178 157 142 113 78 Favor le AU, C YT MITO, CR EC YT CR CYT CR AU, C YT CR DEC CR ab D , In med ate D , C ter di te D Favo le ter i In a E 1 ter ate VID , D |
 Slide Key * Investigator Sponsored Ida idarubicin LDAC Low Dose AraC AraC Arabinoside Cytosine Dauno Daunorubicin MEC Mitoxantrone + Etoposide + AraC CLAG Clofarabine + AraC + G-CSF Potential Selinexor AML Development 2014 H1 H2 2015 H1 H2 2016 H1 H2 2017 H1 H2 Launch Ph 1 Sel in R/R AML SOPRA: Ph 2 Selinexor vs. Physician’s Choice (2:1) 170 pts; OS Endpoint Prep/Submit NDA/EMA Front Line (Older): Selinexor + LDAC Pick the Winner (L1 Study); Interim 2015-6 Data Selinexor + Decitabine (OSU) Induction: Dauno/AraC + Sel Induction: Ida/AraC + Sel Relapse: MEC + Sel Relapse: CLAG + Sel Pediatric Acute Leukemias Front Line Induction High Risk AML: Dauno/AraC + Sel * * * * * * * Phase 3 : Randomized Studies with Selinexor in Combination * ©2015 – Karyopharm Therapeutics, Inc. 24 |
 ©2015 – Karyopharm Therapeutics Inc. 25 Selinexor in Solid Tumors & Combination Studies |
 26 ASCO Summary & Conclusions – KING, SIGN, & Sarcoma Selinexor shows anti-tumor activity with 13% ORR and 38% DCR in patients with GBM that progressed after temozolomide and radiation Selinexor levels in brain tumor tissue 2 hours after dosing were at or higher than those with known anti-cancer activity Selinexor showed broad anti-tumor activity across all three heavily pretreated gynecological cancer populations: Selinexor induced meaningful single-agent anti-cancer activity in patients with ovarian and endometrial cancers with disease control rates (PR+ 12 week SD) of 55% and 62% respectively and several patients remaining on study for 6 to >11 months. Single agent oral selinexor demonstrated durable stable disease in liposarcoma, leiomyosarcoma and other sarcomas In patients with previously treated liposarcoma, PFS on selinexor was longer than the patient’s most recent anti-cancer regimen ©2015 – Karyopharm Therapeutics, Inc. |
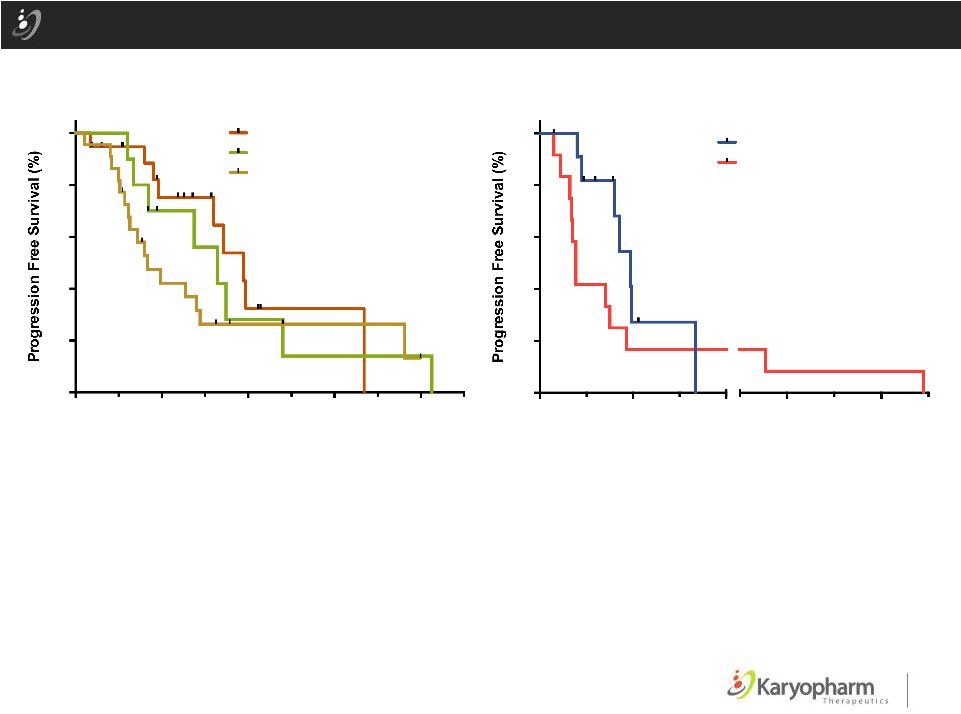 27 ©2015 – Karyopharm Therapeutics, Inc. Progression Free Survival (PFS) by Sarcoma Type (A) Progression Free Survival (PFS) by sarcoma type and (B) Liposarcoma PFS for selinexor vs last prior regimen. Comparison of PFS vs last prior regimen is limited to liposarcoma patients with known time to progression for last prior regimen. Median PFS and N are listed in the legends in parentheses. P values and hazard ratios (by Gehan-Breslow- Wilcoxon and logrank respectively) are listed for statistically significant differences. A B Time (weeks) Time (weeks) 0 10 20 30 40 0 20 40 60 80 100 All Sarcomas Other sarcoma (8.3 wks, N=22) Leiomyosarcoma (16.4 wks, N=10) Liposarcoma (19.4 wks, N=19) p<0.03, HR = 0.55 for liposarcoma vs other 0 20 40 100 120 0 20 40 60 80 100 Liposarcoma Selinexor (19.4 wks) Last prior regimen (7.7 wks) N=11 p<0.02, HR = 0.53 |
 28 Hematological Malignancies • Selinexor + Carfilzomib + Dexamethasone in patients with R/R MM • Selinexor + Fludarabine + Cytarabine in pediatric patients with relapsed or refractory leukemia or myelodysplastic syndrome • Selinexor + Bortezomib + Dexamethasone in patients with progressive of refractory MM • Selinexor + Ibrutinib in patients with R/R CLL and NHL • Selinexor + Decitabine in patients with AML Solid Tumors • Selinexor + Paclitaxel + Carboplatin in patients with advanced ovarian or endometrial malignancies • Selinexor + Gemcitabine + Abraxane in patients with advanced pancreatic cancer • Selinexor + FOLFOX in patients with metastatic colorectal cancer • Selinexor + Irinotecan in patients with adenocarcinoma of the stomach and distal esophagus • Selinexor + Docetaxel in patients with relapsed squamous cell lung cancer ©2015 – Karyopharm Therapeutics, Inc. ISTs Using Selinexor: Planned or Ongoing in Combination With Other Therapies |
 Financial and Commercial Overview ©2015 – Karyopharm Therapeutics Inc. 29 |
 Cash, Cash Equivalents & Investments: • ~$230.2 MM at September 30, 2015 • Expected to fund the Company into 2018 Shares Outstanding at September 30, 2015 • Basic: ~35.7 MM as of September 30, 2015 • Fully diluted: ~40.2 MM as of September 30, 2015 Financial Overview ©2015 – Karyopharm Therapeutics, Inc. 30 |
 Karyopharm: At the Nucleus of Cancer Care ©2015 – Karyopharm Therapeutics, Inc. Selinexor is a novel, oral selective inhibitor of XPO1-mediated nuclear export with broad single-agent anti-cancer activity Karyopharm wholly owns the worldwide rights to selinexor with patent protection through at least 2032 Three ongoing later phase studies (SOPRA, SADAL and SIRRT) are expected to have topline data by the end of 2016 Two ongoing later phase studies (SOPRA and STORM) are expected to have interim analyses in mid-2016 Multiple combination studies are ongoing or planned to incorporate selinexor into treatment regimens across many cancer types 31 |
 NASDAQ: KPTI Karyopharm Therapeutics Inc. |