
Corporate Presentation April 2017 Exhibit 99.1

Forward-looking Statements This presentation contains forward-looking statements within the meaning of the “safe harbor” provisions of The Private Securities Litigation Reform Act of 1995. Such forward-looking statements include those regarding the therapeutic potential of and potential clinical development plans and commercialization for Karyopharm’s drug candidates, including the timing of initiation of certain trials and of the reporting of data from such trials, and assumptions of management regarding strategic and financial expectations and projections. Such statements are subject to numerous important factors, risks and uncertainties that may cause actual events or results to differ materially from the company’s current expectations. For example, there can be no guarantee that any of Karyopharm’s SINE™ compounds, including selinexor (KPT-330) and KPT-8602, Karyopharm’s second generation SINE™ compound, or KPT-9274, Karyopharm’s first-in-class oral dual inhibitor of PAK4 and NAMPT, or any other drug candidate Karyopharm is developing, will successfully complete necessary preclinical and clinical development phases, that development of any of Karyopharm’s drug candidates will continue, or that the partial clinical hold imposed by the U.S. Food and Drug Administration on selinexor clinical trials will be released in a timely manner or at all. Further, there can be no guarantee that any positive developments in Karyopharm’s drug candidate portfolio will result in stock price appreciation. In addition, even if Karyopharm receives marketing approval for selinexor or another drug candidate, there can be no assurance that Karyopharm will be able to successfully commercialize that drug candidate. Management’s expectations and, therefore, any forward-looking statements in this presentation could also be affected by risks and uncertainties relating to a number of other factors, many of which are beyond Karyopharm’s control, including the following: Karyopharm’s results of clinical trials and preclinical studies, including subsequent analysis of existing data and new data received from ongoing and future studies; the content and timing of decisions made by the FDA and other regulatory authorities, investigational review boards at clinical trial sites and publication review bodies; Karyopharm’s ability to obtain and maintain requisite regulatory approvals and to enroll patients in its clinical trials; unplanned cash requirements and expenditures; development of drug candidates by Karyopharm’s competitors for diseases for which Karyopharm is currently developing its drug candidates; and Karyopharm’s ability to obtain, maintain and enforce patent and other intellectual property protection for any drug candidates it is developing. These and other risks, including those which may impact management’s expectations, are described in greater detail under the heading "Risk Factors" in Karyopharm’s Annual Report on Form 10-K for the year ended December 31, 2016, which is on file with the Securities and Exchange Commission, and in subsequent filings filed by Karyopharm with the Securities and Exchange Commission. Any forward-looking statements contained in this presentation are for informational purposes only and speak only as of the date hereof. Other than as is required by law, Karyopharm expressly disclaims any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise. Karyopharm’s website is http://www.karyopharm.com. Karyopharm regularly uses its website to post information regarding its business, drug development programs and governance. Karyopharm encourages investors to use www.karyopharm.com, particularly the information in the section entitled “Investors,” as a source of information about Karyopharm. References to www.karyopharm.com in this presentation are not intended to, nor shall they be deemed to, incorporate information on www.karyopharm.com into this presentation by reference. Unless otherwise noted, this presentation contains data that are interim and unaudited based on site reports. ©2017 Karyopharm Therapeutics Inc.
Karyopharm Overview Innovators First to enter clinical development Oral XPO1 inhibitors for the treatment of cancer and other major diseases SINE™ Compounds Novel, unique and broadly applicable MoA Lead candidates are first-in-class, oral Selective Inhibitor of Nuclear Export (SINE™) compounds Fully owned; COM patent for selinexor with protection to at least 2032 Strong Leadership Experienced management team CEO integral in accelerated approvals of Velcade® and Kyprolis® Track record of success in oncology drug development, regulatory approval and commercialization Near Term Catalysts Several data readouts expected over the next 18 months SADAL SEAL STOMP KPT-8602 STORM BOSTON Well Capitalized Cash runway until the end of 2018 Beyond expected key data inflection points including expanded STORM data and BOSTON enrollment Selinexor Oral, take-at-home, broad single-agent activity. Combinable with favorable safety profile Potential to serve as a backbone therapy with diverse array of anti-cancer agents ©2017 Karyopharm Therapeutics Inc.
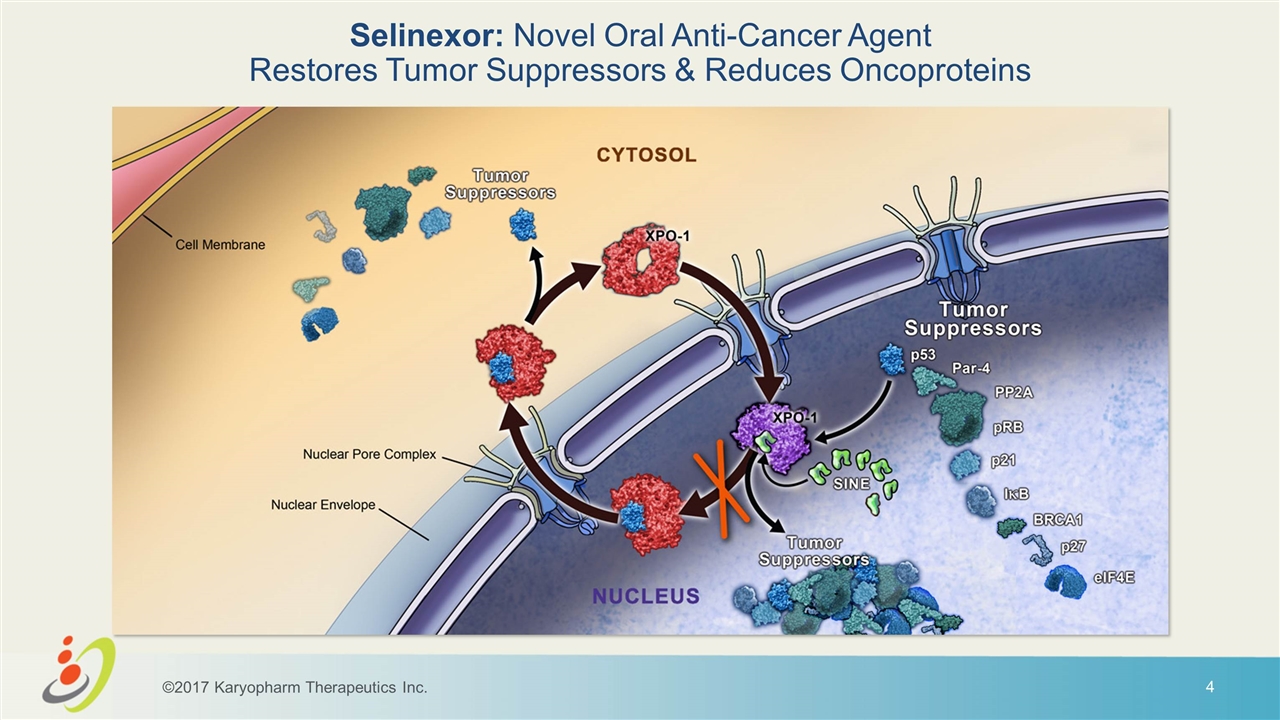
Selinexor: Novel Oral Anti-Cancer Agent Restores Tumor Suppressors & Reduces Oncoproteins ©2017 Karyopharm Therapeutics Inc.
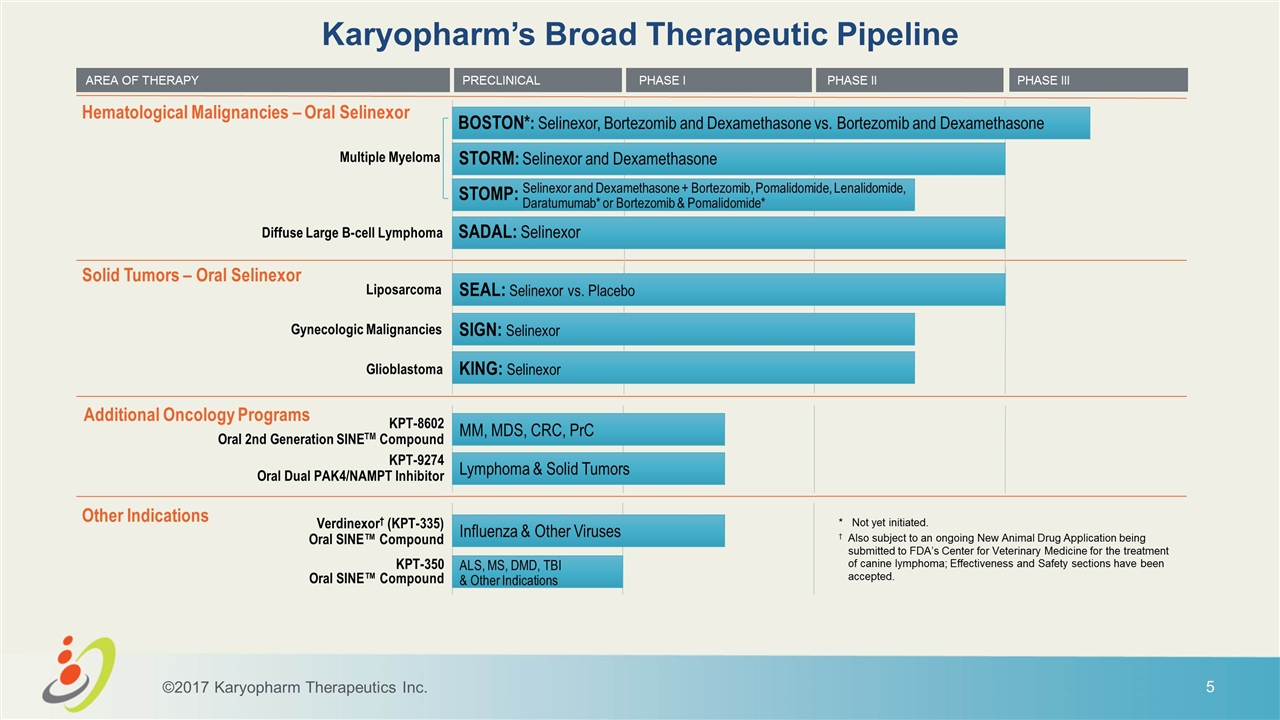
Karyopharm’s Broad Therapeutic Pipeline AREA OF THERAPY Selinexor and Dexamethasone + Bortezomib, Pomalidomide, Lenalidomide, Daratumumab* or Bortezomib & Pomalidomide* PRECLINICAL PHASE III Liposarcoma Gynecologic Malignancies Glioblastoma Diffuse Large B-cell Lymphoma Multiple Myeloma Hematological Malignancies – Oral Selinexor Solid Tumors – Oral Selinexor SEAL: Selinexor vs. Placebo SIGN: Selinexor KING: Selinexor SADAL: Selinexor Additional Oncology Programs Other Indications PHASE I PHASE II KPT-8602 Oral 2nd Generation SINETM Compound KPT-9274 Oral Dual PAK4/NAMPT Inhibitor Verdinexor† (KPT-335) Oral SINE™ Compound KPT-350 Oral SINE™ Compound MM, MDS, CRC, PrC Lymphoma & Solid Tumors Influenza & Other Viruses ALS, MS, DMD, TBI & Other Indications * Not yet initiated. †Also subject to an ongoing New Animal Drug Application being submitted to FDA’s Center for Veterinary Medicine for the treatment of canine lymphoma; Effectiveness and Safety sections have been accepted. STORM: Selinexor and Dexamethasone BOSTON*: Selinexor, Bortezomib and Dexamethasone vs. Bortezomib and Dexamethasone ©2017 Karyopharm Therapeutics Inc. STOMP:

Selinexor in Myeloma: First Oral Agent with Novel Mechanism and Single Agent Activity Since the IMiDs

Selinexor Advantages in Multiple Myeloma Novel agent with anti-MM activity, including in penta-refractory disease Oral, taken at home Combinable with observed additive or synergistic activity and potential to resensitize to prior therapies PIs: Velcade and Kyprolis IMiDs: Revlimid and Pomalyst Combination regimens appear to be well tolerated Side effects reported in the SVd arm of STOMP are similar to, or reduced, compared with Sd Phase 3 SVd regimen uses 40% less Velcade and 25% less Dex than standard Vd dosing Planned additional combination regimens (plus Darzalex, plus Ninlaro) Worldwide MM market estimated ~$11 billion in 2015, increasing to > $22 billion by 2023 Individual MM drugs may generate peak sales in excess of $1-2B Selinexor could be the next anti-MM agent with potential as a “backbone” therapy ©2017 Karyopharm Therapeutics Inc.
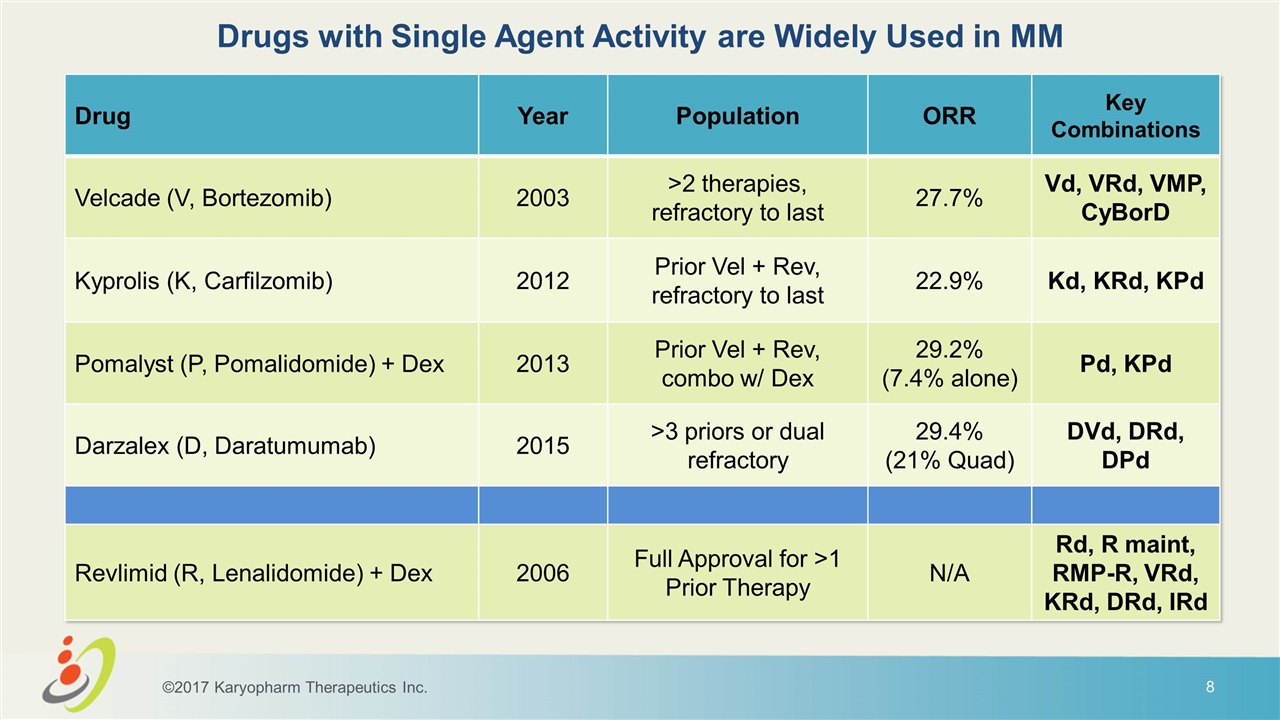
Drugs with Single Agent Activity are Widely Used in MM Drug Year Population ORR Key Combinations Velcade (V, Bortezomib) 2003 >2 therapies, refractory to last 27.7% Vd, VRd, VMP, CyBorD Kyprolis (K, Carfilzomib) 2012 Prior Vel + Rev, refractory to last 22.9% Kd, KRd, KPd Pomalyst (P, Pomalidomide) + Dex 2013 Prior Vel + Rev, combo w/ Dex 29.2% (7.4% alone) Pd, KPd Darzalex (D, Daratumumab) 2015 >3 priors or dual refractory 29.4% (21% Quad) DVd, DRd, DPd Revlimid (R, Lenalidomide) + Dex 2006 Full Approval for >1 Prior Therapy N/A Rd, R maint, RMP-R, VRd, KRd, DRd, IRd ©2017 Karyopharm Therapeutics Inc.

STORM: Selinexor in Refractory Myeloma: Quad and Penta Populations
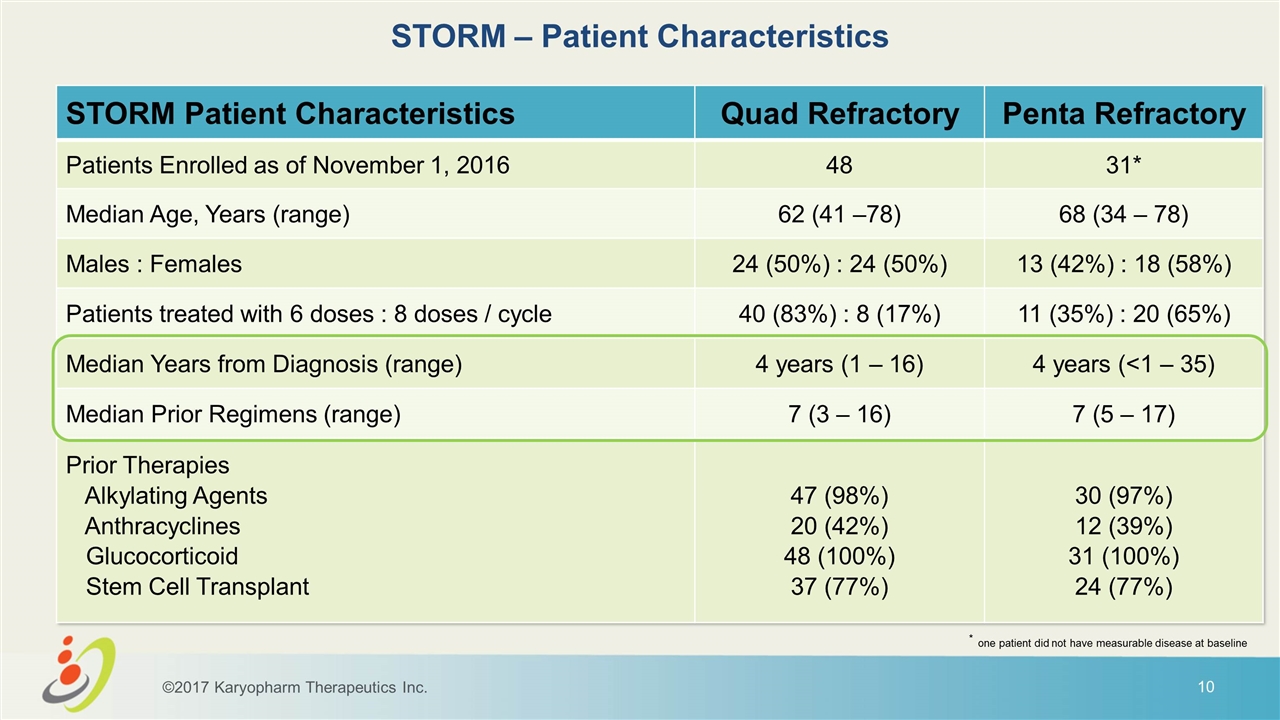
STORM – Patient Characteristics STORM Patient Characteristics Quad Refractory Penta Refractory Patients Enrolled as of November 1, 2016 48 31* Median Age, Years (range) 62 (41 –78) 68 (34 – 78) Males : Females 24 (50%) : 24 (50%) 13 (42%) : 18 (58%) Patients treated with 6 doses : 8 doses / cycle 40 (83%) : 8 (17%) 11 (35%) : 20 (65%) Median Years from Diagnosis (range) 4 years (1 – 16) 4 years (<1 – 35) Median Prior Regimens (range) 7 (3 – 16) 7 (5 – 17) Prior Therapies Alkylating Agents Anthracyclines Glucocorticoid Stem Cell Transplant 47 (98%) 20 (42%) 48 (100%) 37 (77%) 30 (97%) 12 (39%) 31 (100%) 24 (77%) ©2017 Karyopharm Therapeutics Inc. * one patient did not have measurable disease at baseline
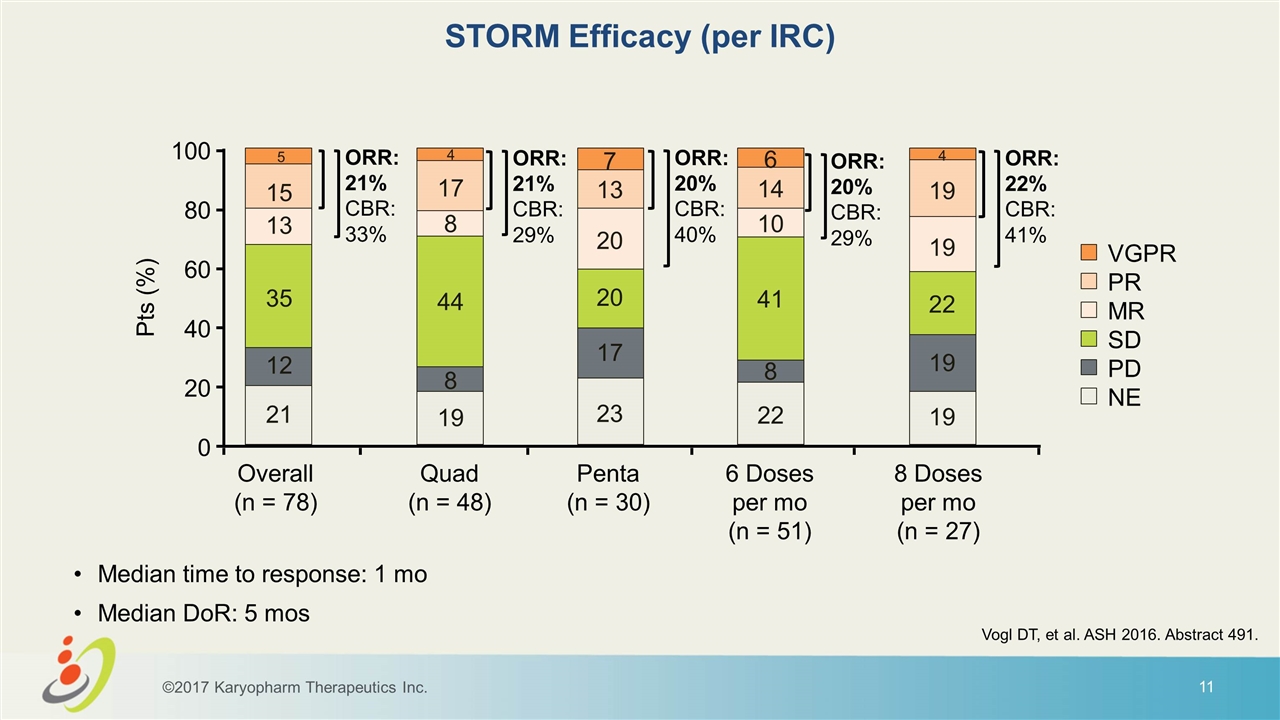
STORM Efficacy (per IRC) ©2017 Karyopharm Therapeutics Inc. ORR: 21% CBR: 33% 5 15 13 35 12 21 4 17 8 44 8 19 7 13 20 20 17 23 6 14 10 41 8 22 4 19 19 22 19 19 Median time to response: 1 mo Median DoR: 5 mos Vogl DT, et al. ASH 2016. Abstract 491. 100 80 60 40 20 0 Overall (n = 78) Quad (n = 48) Penta (n = 30) 6 Doses per mo (n = 51) 8 Doses per mo (n = 27) VGPR PR MR SD PD NE Pts (%) ORR: 21% CBR: 29% ORR: 20% CBR: 40% ORR: 20% CBR: 29% ORR: 22% CBR: 41%
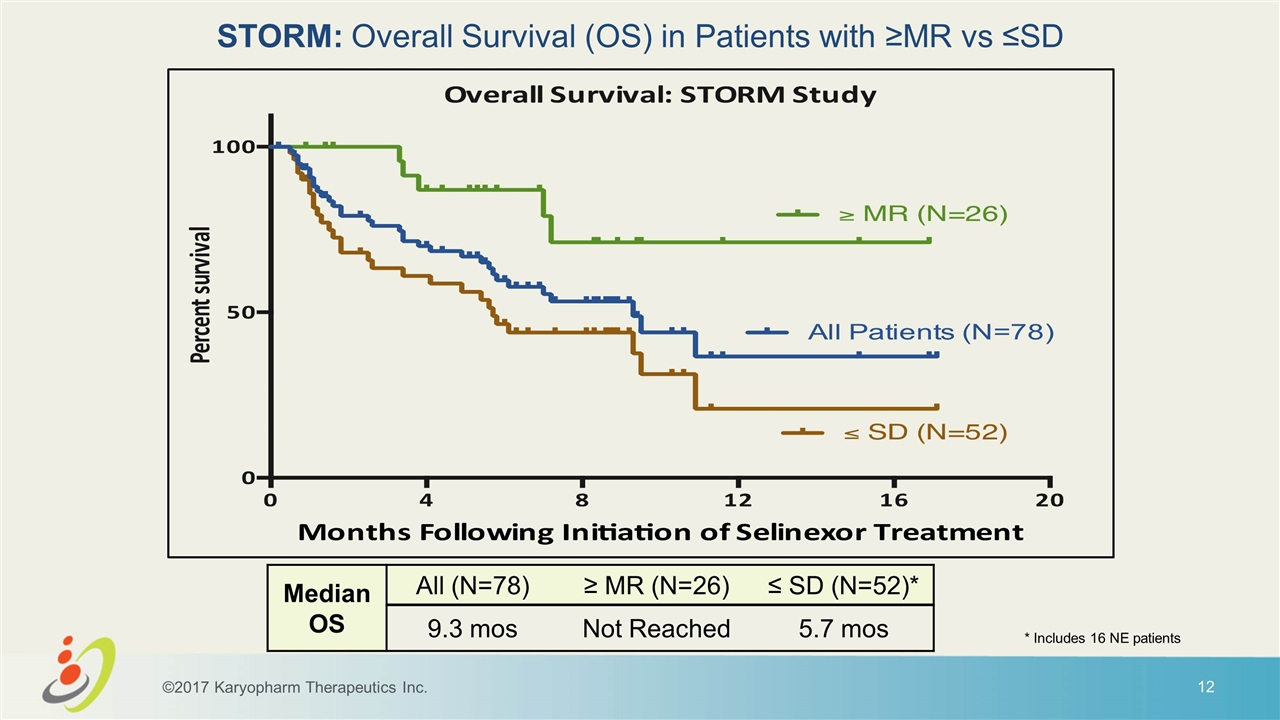
STORM: Overall Survival (OS) in Patients with ≥MR vs ≤SD Median OS All (N=78) ≥ MR (N=26) ≤ SD (N=52)* 9.3 mos Not Reached 5.7 mos ©2017 Karyopharm Therapeutics Inc. * Includes 16 NE patients
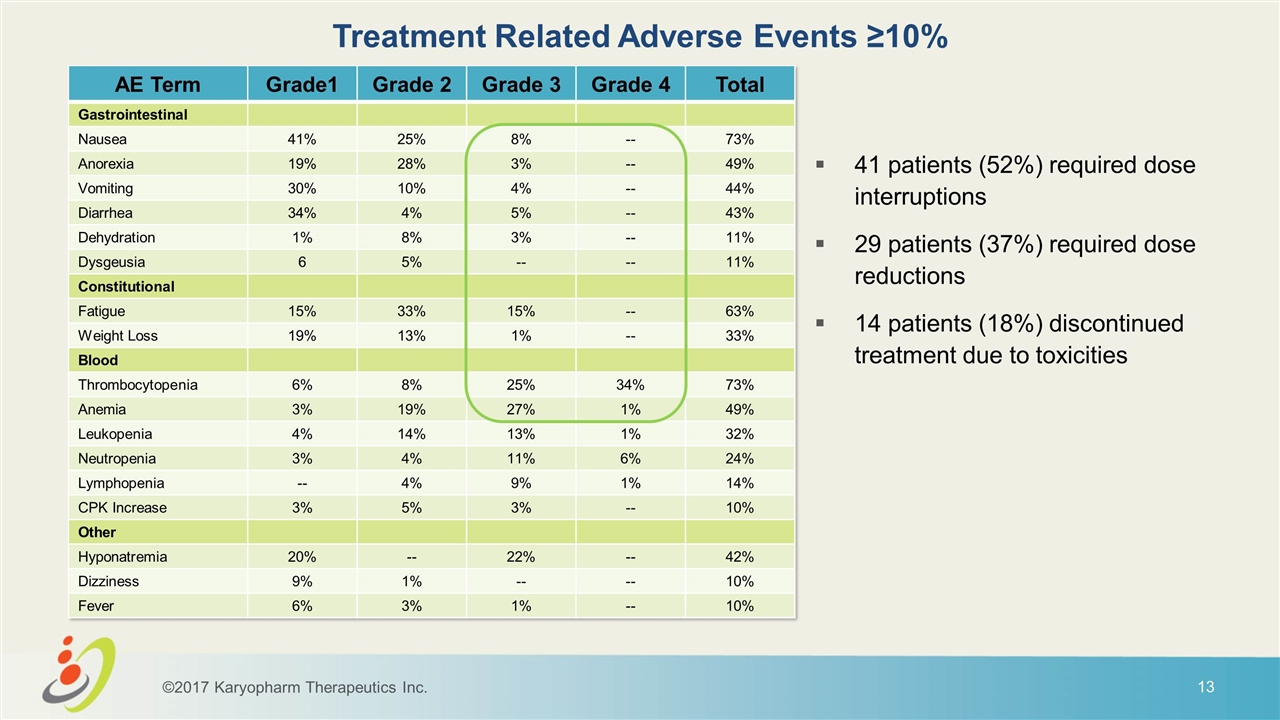
Treatment Related Adverse Events ≥10% 41 patients (52%) required dose interruptions 29 patients (37%) required dose reductions 14 patients (18%) discontinued treatment due to toxicities AE Term Grade1 Grade 2 Grade 3 Grade 4 Total Gastrointestinal Nausea 41% 25% 8% -- 73% Anorexia 19% 28% 3% -- 49% Vomiting 30% 10% 4% -- 44% Diarrhea 34% 4% 5% -- 43% Dehydration 1% 8% 3% -- 11% Dysgeusia 6 5% -- -- 11% Constitutional Fatigue 15% 33% 15% -- 63% Weight Loss 19% 13% 1% -- 33% Blood Thrombocytopenia 6% 8% 25% 34% 73% Anemia 3% 19% 27% 1% 49% Leukopenia 4% 14% 13% 1% 32% Neutropenia 3% 4% 11% 6% 24% Lymphopenia -- 4% 9% 1% 14% CPK Increase 3% 5% 3% -- 10% Other Hyponatremia 20% -- 22% -- 42% Dizziness 9% 1% -- -- 10% Fever 6% 3% 1% -- 10% ©2017 Karyopharm Therapeutics Inc.

Multiple Myeloma Accelerated Approval Strategy STORM Expansion Underway (N=122 additional patients) Patients with penta-refractory disease are considered an unmet medical need Additional sites being added in the US and EU to support expansion As of March 30 2017, partial clinical hold lifted by U.S. FDA Division of Hematology Products Target topline data from the expanded cohort in early 2018 Potential NDA Submission in Mid-2018 Engage regulatory agencies on potential expedited review opportunities Assuming positive data from STORM expansion, plan to seek accelerated approval (FDA) and conditional approval (EMA) ©2017 Karyopharm Therapeutics Inc.

STOMP: Selinexor and Backbone Treatments for Relapsed/Refractory Multiple Myeloma
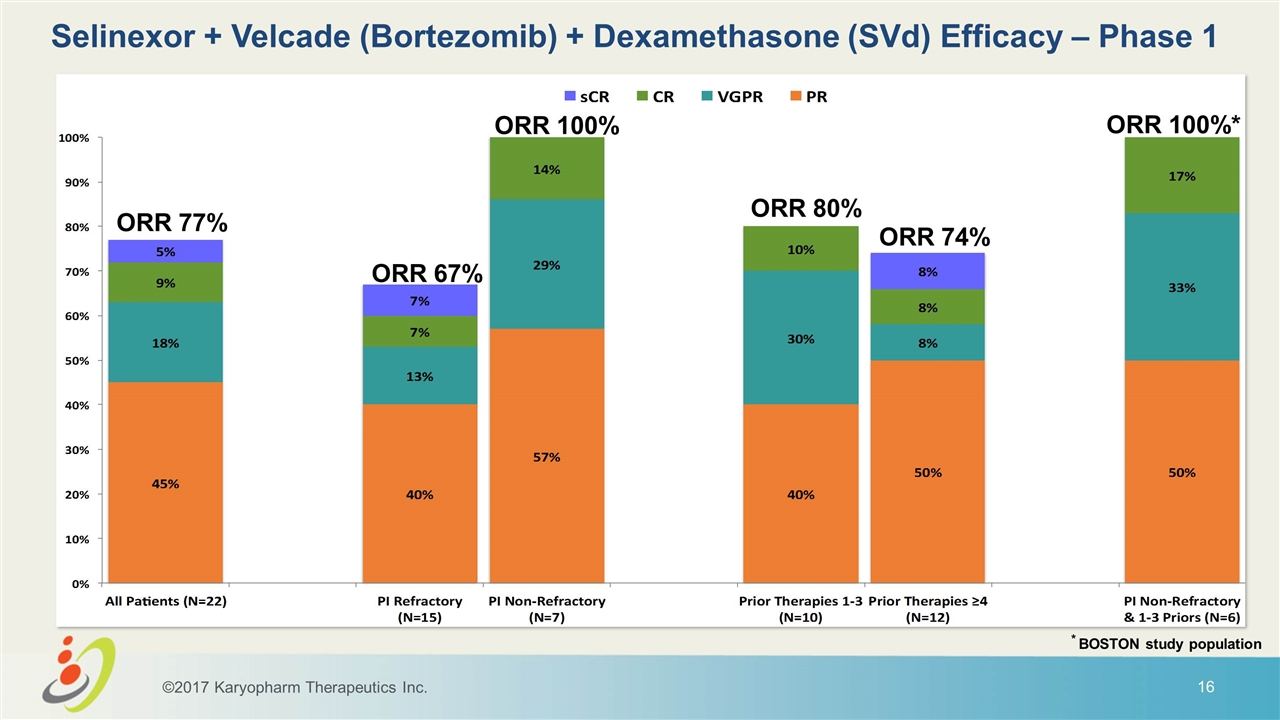
Selinexor + Velcade (Bortezomib) + Dexamethasone (SVd) Efficacy – Phase 1 ORR 67% ORR 77% ORR 100% ORR 100%* ORR 80% ORR 74% ©2017 Karyopharm Therapeutics Inc. * BOSTON study population
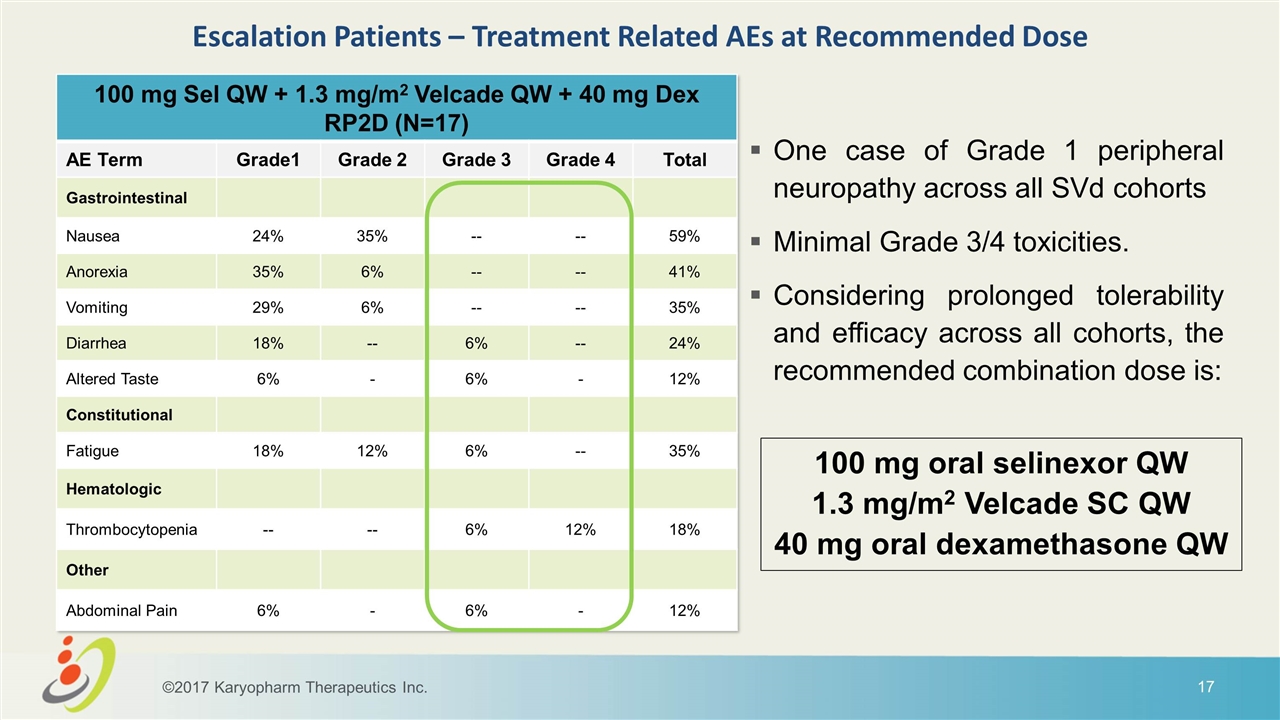
100 mg Sel QW + 1.3 mg/m2 Velcade QW + 40 mg Dex RP2D (N=17) AE Term Grade1 Grade 2 Grade 3 Grade 4 Total Gastrointestinal Nausea 24% 35% -- -- 59% Anorexia 35% 6% -- -- 41% Vomiting 29% 6% -- -- 35% Diarrhea 18% -- 6% -- 24% Altered Taste 6% - 6% - 12% Constitutional Fatigue 18% 12% 6% -- 35% Hematologic Thrombocytopenia -- -- 6% 12% 18% Other Abdominal Pain 6% - 6% - 12% Escalation Patients – Treatment Related AEs at Recommended Dose One case of Grade 1 peripheral neuropathy across all SVd cohorts Minimal Grade 3/4 toxicities. Considering prolonged tolerability and efficacy across all cohorts, the recommended combination dose is: 100 mg oral selinexor QW 1.3 mg/m2 Velcade SC QW 40 mg oral dexamethasone QW ©2017 Karyopharm Therapeutics Inc.
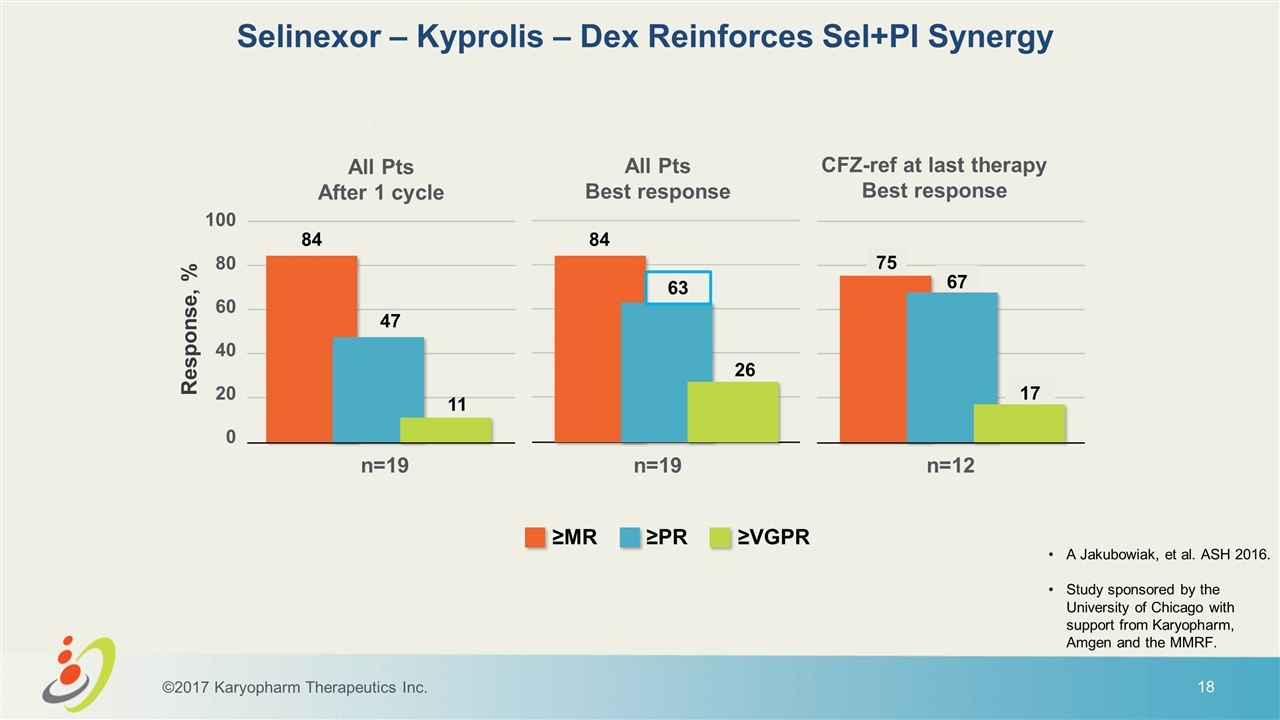
Selinexor – Kyprolis – Dex Reinforces Sel+PI Synergy A Jakubowiak, et al. ASH 2016. Study sponsored by the University of Chicago with support from Karyopharm, Amgen and the MMRF. ©2017 Karyopharm Therapeutics Inc. * 84 47 11 84 63 26 75 67 17 All Pts After 1 cycle All Pts Best response CFZ-ref at last therapy Best response ≥MR ≥PR ≥VGPR n=19 n=19 n=12 100 80 60 40 20 0 Response, %

†Responses were adjudicated according to the International Myeloma Working Group criteria, ‡ included one unconfirmed PR. ORR=Overall Response Rate (VGPR+PR), VGPR=Very Good Partial Response, PR=Partial Response, MR=Minor Response, SD=Stable Disease, PD=Progressive Disease, CBR=Clinical Benefit Rate (VGPR+PR+MR). Responses as of November 28, 2016 based on interim unaudited site data. Selinexor + Pomalyst (Pomalidomide) + Dexamethasone (SPd) Efficacy – Phase 1 Best Responses† in Evaluable SPd Patients as of November 28, 2016 Category N ORR(%) CBR(%) VGPR(%) PR‡ (%) MR(%) SD(%) PD(%) All Patients 15 9 (60%) 11 (73%) 3 (20%) 6 (40%) 2 (13%) 3 (20%) 1 (7%) ©2017 Karyopharm Therapeutics Inc.

Randomized Phase 3 Global “BOSTON” Trial A Phase 3 Randomized, Open-Label Multicenter Study of Selinexor, Velcade (bortezomib) and Dexamethasone (SVd) vs. Velcade (bortezomib) and Dexamethasone (Vd) in Patients with Relapsed/Refractory Myeloma Key Aspects of Study Design 1-3 prior line of therapy, naïve to proteasome inhibitors (PIs), or if patient had prior PI, then must have had ≥PR and at least 6 months since last PI SVd Arm: Weekly Velcade sc dosing 4 / 5 weeks with weekly selinexor and dexamethasone Vd Arm: Standard Velcade sc BIW 2/3 weeks x 8 cycles then QW 4 / 5 weeks for cycles 9+ 1:1 randomization Interim PFS analysis after 75% of the PFS events for potential sample size adjustment For Vd arm, cross-over to SVd at objective progression permitted Planned in US, Canada, Europe, and Asia Primary Endpoint (EP) PFS Key Secondary EPs ORR, DOR, PFS 1 + PFS 2, OS, QOL Statistical Assumptions Vd (9.4 mo., based on ENDEAVOR), 65% ORR SVd (13.5 mo.; HR ~ 0.67), 85% ORR ~360 pts (180/arm) ©2017 Karyopharm Therapeutics Inc.
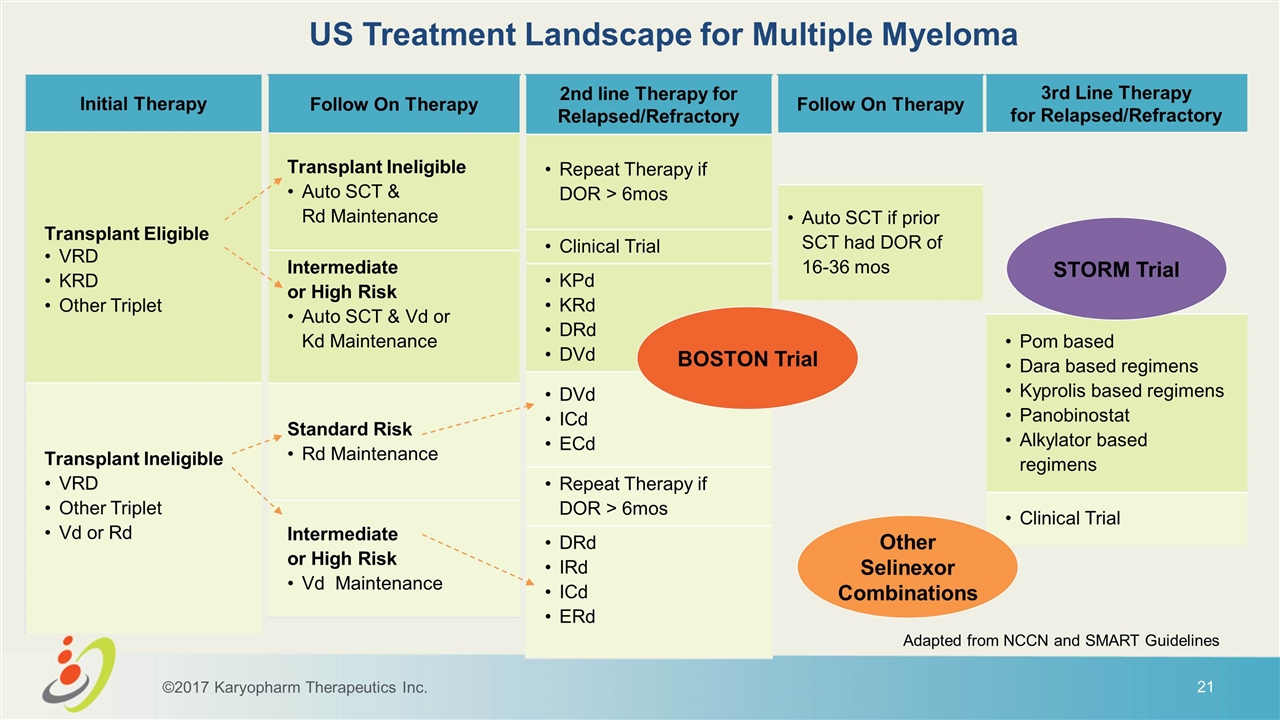
Follow On Therapy Auto SCT if prior SCT had DOR of 16-36 mos Follow On Therapy Transplant Ineligible Auto SCT & Rd Maintenance Intermediate or High Risk Auto SCT & Vd or Kd Maintenance Standard Risk Rd Maintenance Intermediate or High Risk Vd Maintenance 2nd line Therapy for Relapsed/Refractory Repeat Therapy if DOR > 6mos Clinical Trial KPd KRd DRd DVd DVd ICd ECd Repeat Therapy if DOR > 6mos DRd IRd ICd ERd 3rd Line Therapy for Relapsed/Refractory Pom based Dara based regimens Kyprolis based regimens Panobinostat Alkylator based regimens Clinical Trial Initial Therapy Transplant Eligible VRD KRD Other Triplet Transplant Ineligible VRD Other Triplet Vd or Rd US Treatment Landscape for Multiple Myeloma ©2017 Karyopharm Therapeutics Inc. Adapted from NCCN and SMART Guidelines Other Selinexor Combinations STORM Trial BOSTON Trial

SADAL Study in DLBCL

SADAL: Randomized Phase 2b Trial in DLBCL SADAL: Selinexor Against Diffuse Aggressive Lymphoma Ongoing Randomized Trial for Accelerated Approval Relapsed / refractory ≥3rd line; At least 50% of patients with GCB-DLBCL 28.6% ORR in the first 63 patients in Phase 2b DLBCL study with median DOR >7 months Planned protocol amendment following consultation with FDA Discontinue 100mg arm Targeting an additional 90 patients at 60mg in single arm Primary endpoint: overall response rate Recent FDA correspondence confirms potential for accelerated approval pending data and treatment landscape Top-line expanded 60mg data readout anticipated in mid-2018 ©2017 Karyopharm Therapeutics Inc.
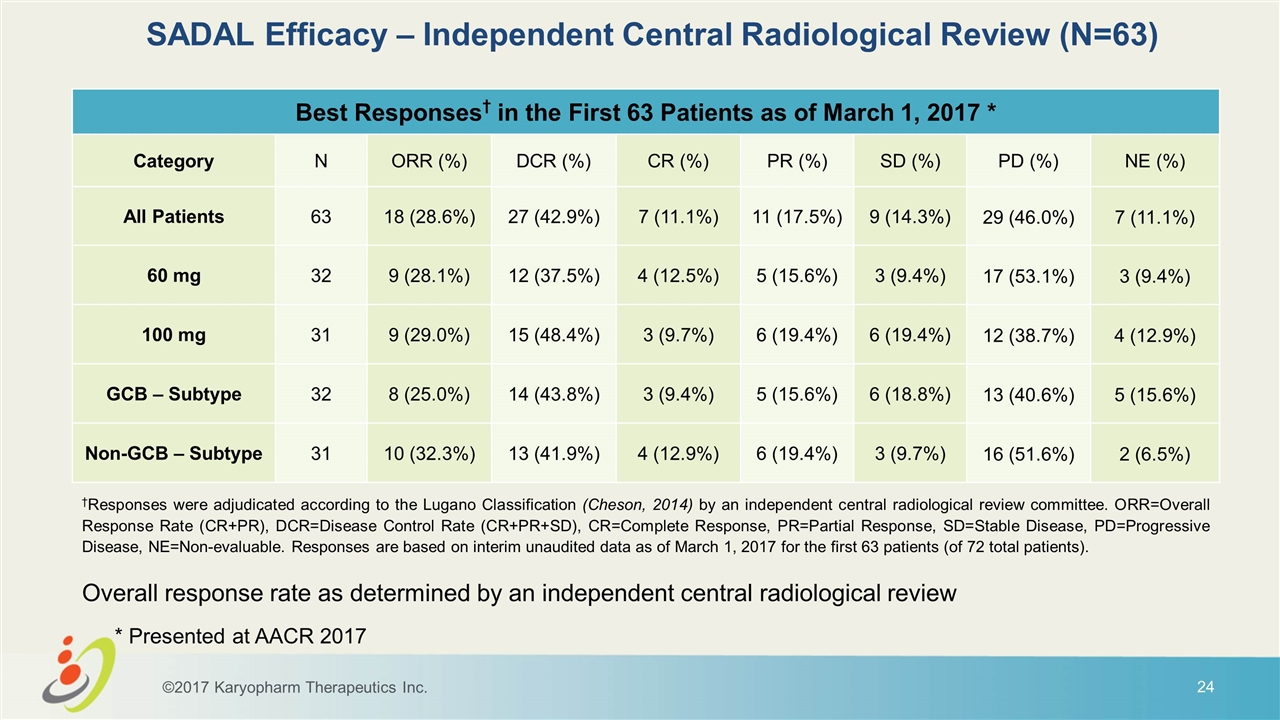
SADAL Efficacy – Independent Central Radiological Review (N=63) Best Responses† in the First 63 Patients as of March 1, 2017 * Category N ORR (%) DCR (%) CR (%) PR (%) SD (%) PD (%) NE (%) All Patients 63 18 (28.6%) 27 (42.9%) 7 (11.1%) 11 (17.5%) 9 (14.3%) 29 (46.0%) 7 (11.1%) 60 mg 32 9 (28.1%) 12 (37.5%) 4 (12.5%) 5 (15.6%) 3 (9.4%) 17 (53.1%) 3 (9.4%) 100 mg 31 9 (29.0%) 15 (48.4%) 3 (9.7%) 6 (19.4%) 6 (19.4%) 12 (38.7%) 4 (12.9%) GCB – Subtype 32 8 (25.0%) 14 (43.8%) 3 (9.4%) 5 (15.6%) 6 (18.8%) 13 (40.6%) 5 (15.6%) Non-GCB – Subtype 31 10 (32.3%) 13 (41.9%) 4 (12.9%) 6 (19.4%) 3 (9.7%) 16 (51.6%) 2 (6.5%) Overall response rate as determined by an independent central radiological review †Responses were adjudicated according to the Lugano Classification (Cheson, 2014) by an independent central radiological review committee. ORR=Overall Response Rate (CR+PR), DCR=Disease Control Rate (CR+PR+SD), CR=Complete Response, PR=Partial Response, SD=Stable Disease, PD=Progressive Disease, NE=Non-evaluable. Responses are based on interim unaudited data as of March 1, 2017 for the first 63 patients (of 72 total patients). ©2017 Karyopharm Therapeutics Inc. * Presented at AACR 2017
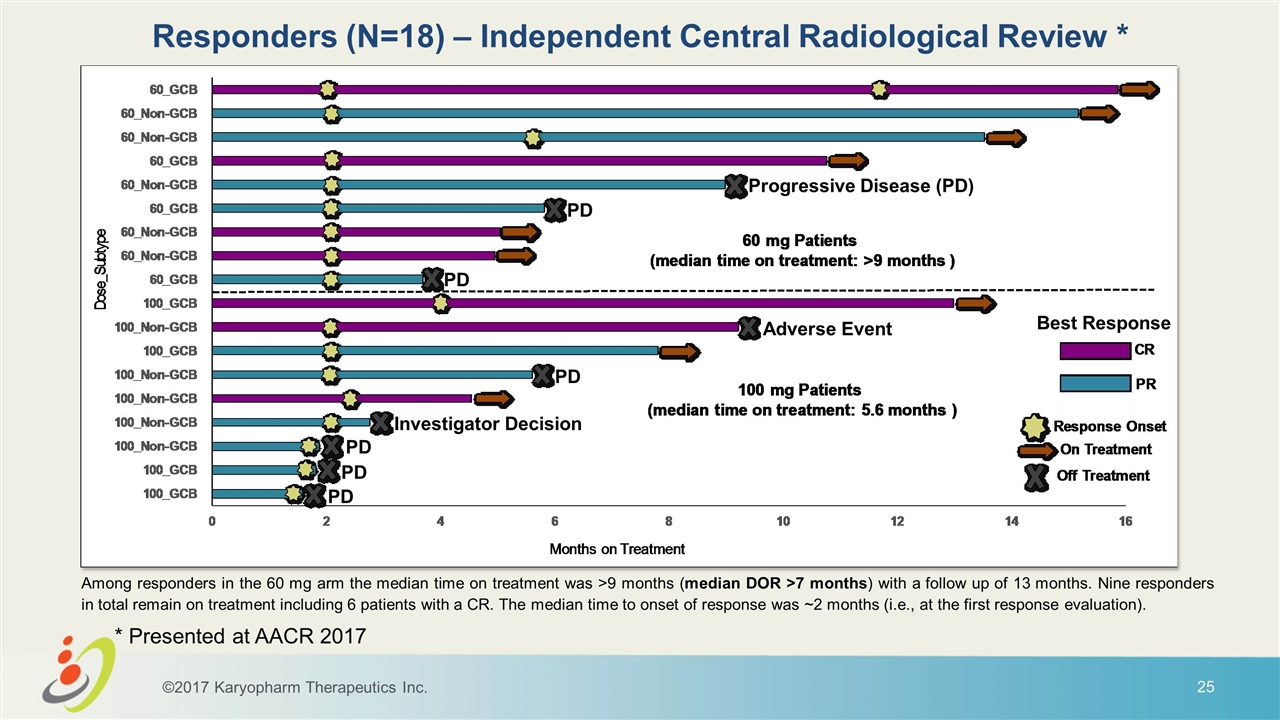
Among responders in the 60 mg arm the median time on treatment was >9 months (median DOR >7 months) with a follow up of 13 months. Nine responders in total remain on treatment including 6 patients with a CR. The median time to onset of response was ~2 months (i.e., at the first response evaluation). Best Response Progressive Disease (PD) PD PD Adverse Event PD Investigator Decision PD PD PD Responders (N=18) – Independent Central Radiological Review * ©2017 Karyopharm Therapeutics Inc. * Presented at AACR 2017
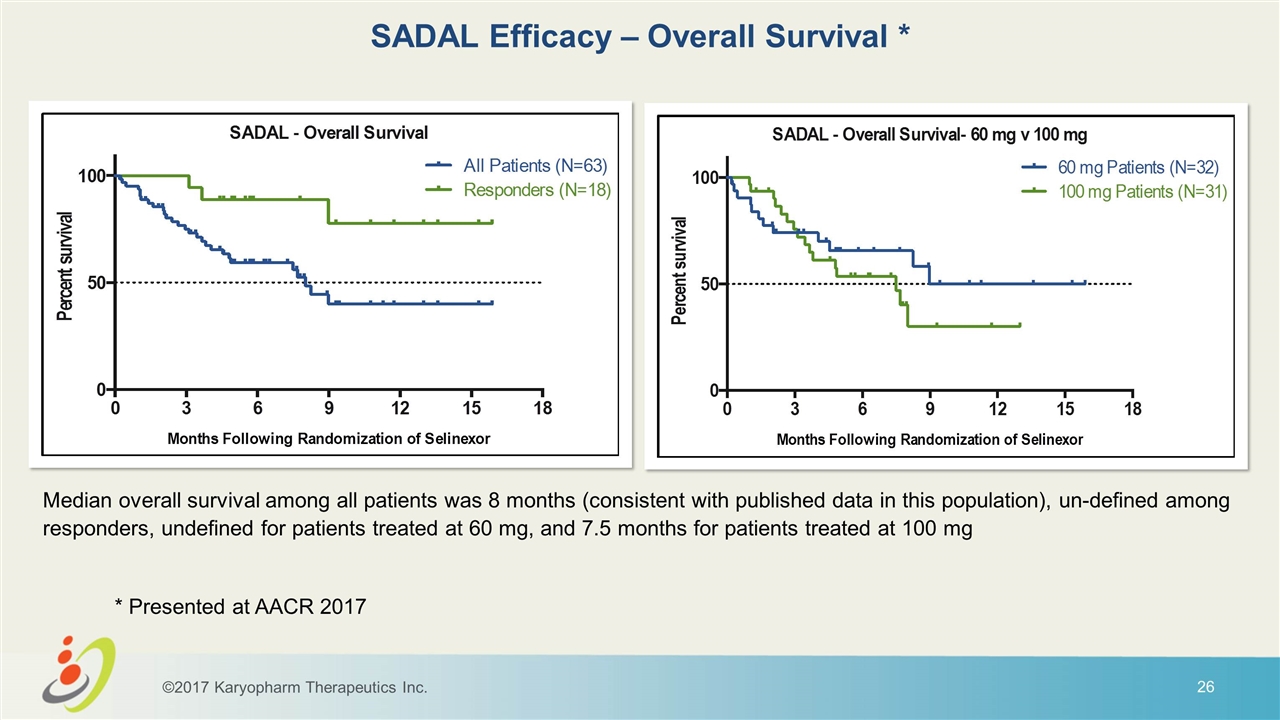
Median overall survival among all patients was 8 months (consistent with published data in this population), un-defined among responders, undefined for patients treated at 60 mg, and 7.5 months for patients treated at 100 mg SADAL Efficacy – Overall Survival * ©2017 Karyopharm Therapeutics Inc. * Presented at AACR 2017
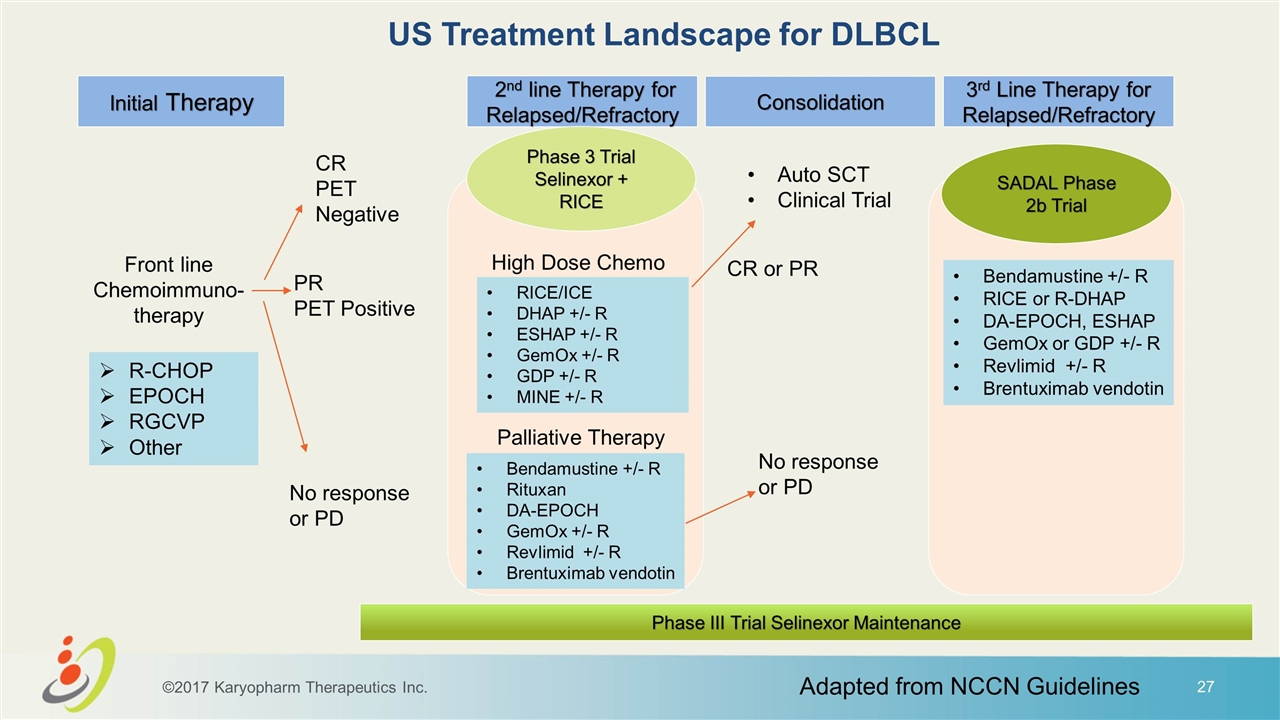
US Treatment Landscape for DLBCL Front line Chemoimmuno- therapy CR PET Negative PR PET Positive No response or PD Initial Therapy 2nd line Therapy for Relapsed/Refractory Consolidation 3rd Line Therapy for Relapsed/Refractory Auto SCT Clinical Trial R-CHOP EPOCH RGCVP Other RICE/ICE DHAP +/- R ESHAP +/- R GemOx +/- R GDP +/- R MINE +/- R Bendamustine +/- R Rituxan DA-EPOCH GemOx +/- R Revlimid +/- R Brentuximab vendotin CR or PR No response or PD Bendamustine +/- R RICE or R-DHAP DA-EPOCH, ESHAP GemOx or GDP +/- R Revlimid +/- R Brentuximab vendotin SADAL Phase 2b Trial Phase 3 Trial Selinexor + RICE Phase III Trial Selinexor Maintenance Adapted from NCCN Guidelines ©2017 Karyopharm Therapeutics Inc. High Dose Chemo Palliative Therapy

Solid Tumors

SEAL: Phase 2/3 Trial in Liposarcoma SEAL: Selinexor in Advanced Liposarcoma Phase 2 portion fully enrolled = 51 patients Evaluating patients with advanced unresectable dedifferentiated liposarcoma Randomized 1:1, double blind, single-agent selinexor (60mg fixed dose, twice weekly) vs. placebo Primary endpoint: progression-free survival Trial design and endpoints agreed with FDA & EMA Partial clinical hold lifted by U.S. FDA Division of Oncology Products 2 as of April 4, 2017 Top-line data readout from Phase 2 portion anticipated in mid-2017 Following interim analysis, may initiate Phase 3 expansion in late 2017 ©2017 Karyopharm Therapeutics Inc.
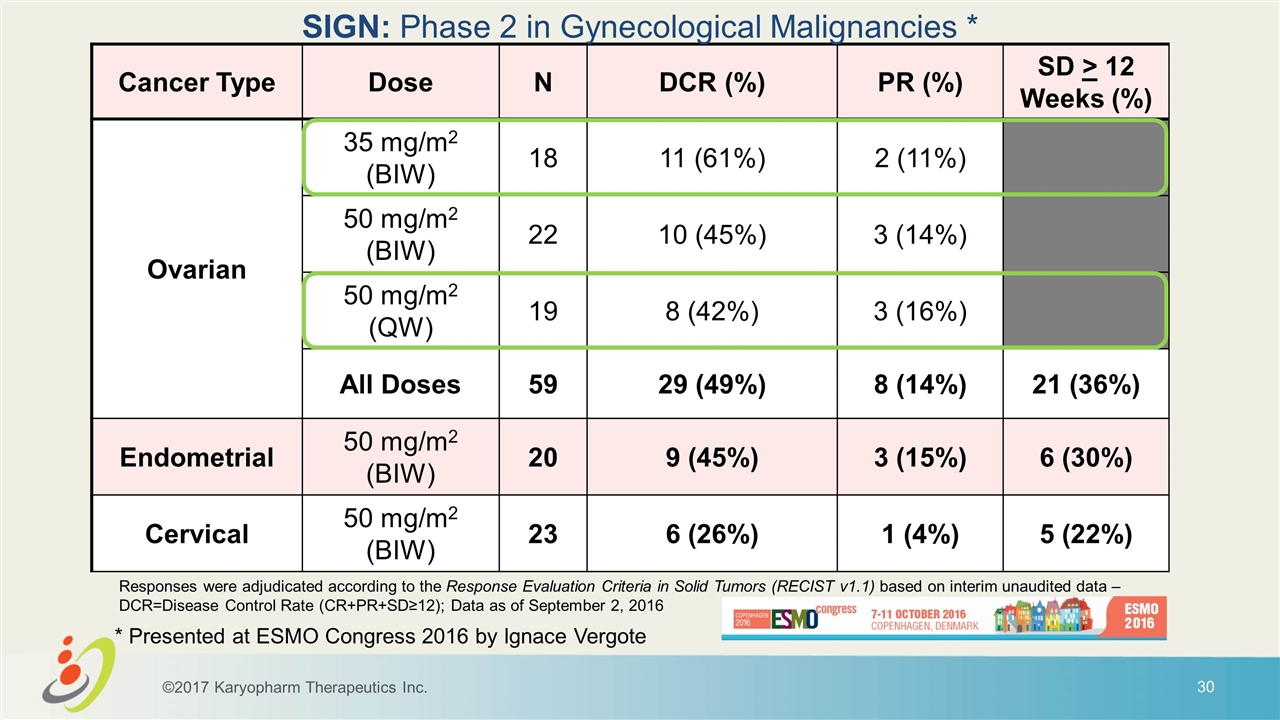
Cancer Type Dose N DCR (%) PR (%) SD > 12 Weeks (%) Ovarian 35 mg/m2 (BIW) 18 11 (61%) 2 (11%) 50 mg/m2 (BIW) 22 10 (45%) 3 (14%) 50 mg/m2 (QW) 19 8 (42%) 3 (16%) All Doses 59 29 (49%) 8 (14%) 21 (36%) Endometrial 50 mg/m2 (BIW) 20 9 (45%) 3 (15%) 6 (30%) Cervical 50 mg/m2 (BIW) 23 6 (26%) 1 (4%) 5 (22%) SIGN: Phase 2 in Gynecological Malignancies * * Presented at ESMO Congress 2016 by Ignace Vergote Responses were adjudicated according to the Response Evaluation Criteria in Solid Tumors (RECIST v1.1) based on interim unaudited data – DCR=Disease Control Rate (CR+PR+SD≥12); Data as of September 2, 2016 ©2017 Karyopharm Therapeutics Inc.

KPT-8602: Second Generation SINE™ Compound Karyopharm is committed to maintaining its leadership position in the discovery and development of SINETM compounds KPT-8602 belongs to a novel chemical series with pharmacological properties distinct from selinexor More reversible binding to XPO1 at Cys528 versus selinexor Similar potency in cell-based assays (IC50 ~20nM) Substantially reduced brain penetration KPT-8602 can be dosed daily over five days per week in animals with tolerability at least equivalent to twice weekly dosing with selinexor Apparent reduction in fatigue in animals More frequent dosing with continued XPO1 inhibition has potential to overcome selinexor resistance Phase 1/2 open label, dose escalation trial is still ongoing with good tolerability Current protocol being amended to include patients with MDS, colorectal & castrate resistant prostate cancers ©2017 Karyopharm Therapeutics Inc.

Financial Overview – Unaudited Projections Cash, Cash Equivalents & Investments ~$175 MM at December 31, 2016 Expected to fund the Company until the end of 2018 Shares Outstanding as of December 31, 2016 Basic: ~41.9 MM common shares Fully diluted: ~47.7 MM common shares ©2017 Karyopharm Therapeutics Inc.

KPT-8602: Phase 1/2 top-line data Late 2017 KPT-9274: Phase 1/2 top-line data (safety and tolerability) Mid 2017 Anticipated Corporate Milestones STORM: Phase 2b expansion top-line dataEarly 2018 BOSTON: Phase 3 top-line data2019 Multiple Myeloma SEAL: Phase 2 top-line data (PFS)Mid 2017 Solid Tumors Pipeline Expansion ©2017 Karyopharm Therapeutics Inc. SADAL: DLBCL Expanded Phase 2b top-line data (ORR) Mid 2018 Report Phase 2b top-line data (ORR) AACR 2017
Karyopharm Overview Innovators First to enter clinical development Oral XPO1 inhibitors for the treatment of cancer and other major diseases SINE™ Compounds Novel, unique and broadly applicable MoA Lead candidates are first-in-class, oral Selective Inhibitor of Nuclear Export (SINE™) compounds Fully owned; COM patent for selinexor with protection to at least 2032 Strong Leadership Experienced management team CEO integral in accelerated approvals of Velcade® and Kyprolis® Track record of success in oncology drug development, regulatory approval and commercialization Near Term Catalysts Several data readouts expected over the next 18 months SADAL SEAL STOMP KPT-8602 STORM BOSTON Well Capitalized Cash runway until the end of 2018 Beyond expected key data inflection points including expanded STORM data and BOSTON enrollment Selinexor Oral, take-at-home, broad single-agent activity. Combinable with favorable safety profile Potential to serve as a backbone therapy with diverse array of anti-cancer agents ©2017 Karyopharm Therapeutics Inc.

Thank You