| Building Bridges in Medical Dermatology Investor Presentation June 2022 NYSE American: TMBR |
| NYSE American: TMBR Safe Harbor Statement 2 Certain statements contained in this PowerPoint presentation describing Timber’s technology and development program, including, without limitation, statements containing the words “expects,” “anticipates,” “believes,” and words of similar import, constitute “forward - looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 .. These forward - looking statements are subject to various risks and uncertainties that could cause actual future results and events to differ materially from those currently anticipated, including risks discussed in Timber’s Form 10 - K and any subsequent filings with the Securities and Exchange Commission .. Potential investors are cautioned not to place undue reliance on these forward - looking statements .. |
| NYSE American: TMBR 3 3 NYSE American: TMBR Timber Pharmaceuticals is a specialty pharmaceutical company devoted to transforming the lives of patients with rare, high impact dermatologic diseases by utilizing advanced translational science. |
| NYSE American: TMBR Timber Business Model 4 Focused on Orphan Drug Indications and Leveraging the 505(b)(2) pathway • Timber can be first to market by pursuing conditions for which there are no current FDA approved treatments • Requires potentially smaller and less costly drug trials with greater flexibility from the FDA • Orphan Drug Market Exclusivity limits additional market entrants and conveys pricing power • 7 years in U.S., 10 years in E.U. and Japan • Bypasses Phase 1 trials by utilizing proven drugs and mechanisms of action • Safety and efficacy already established in the referenced drug • Can immediately begin Phase 2 studies • Potentially mitigates risk from a CMC, Safety and clinical development standpoint • Greater flexibility from the FDA Why Orphan Drugs? Why Leverage the 505(b)(2) Pathway? |
| NYSE American: TMBR Investment Highlights 5 • Positioning to become a leading rare disease dermatology company with a focus on rare and orphan dermatologic diseases, led by a management team with a proven track record in development and commercialization • Lead product candidate TMB - 001 achieved multiple FDA Milestones • Breakthrough therapy status received in May 2022; Fast Track designation awarded in April 2022 • Phase 3 ASCEND study to be initiated by end of June 2022 • Phase 2b CONTROL study completed, and positive top - line data has been reported • Data demonstrated reduction in targeted and overall severity of CI in patients treated with topical IPEG TM TMB - 001 • Large market opportunity for TMB - 001 • U.S. market estimated at $250 million annually at peak* • Recently granted patents provide strong IP protection; additional patents pending • Additional market expansion potential in rare and/or broader dermatology indications • High potential multi - product candidate pipeline • Proven clinical Proof of Concept (POC) • All strategic programs have Orphan designations and no currently approved treatments • Lean operating model and cost structure • $13.9 million cash position as of March 31, 2022 * Based on Timber estimates |
| NYSE American: TMBR 6 Strategic Pipeline in Rare Dermatologic Disease TMB - 001 Received FDA Breakthrough Therapy Designation in May 2022 |
| NYSE American: TMBR 7 7 NYSE American: TMBR TMB - 001 – Topical Isotretinoin Moderate to Severe Congenital Ichthyosis (CI) |
| NYSE American: TMBR 8 TMB - 001 has Received Multiple FDA Milestones May 2022 – FDA Breakthrough therapy status granted • A process designed to expedite the development and review of drugs that are intended to treat serious or life - threatening conditions April 2022 – FDA Fast Track designation received • Facilitates the development and expedites the review of drugs that treat serious conditions and fill an unmet medical need September 2018 - Received $1.5 million Grant from the FDA’s Orphan Products Clinical Trials Grants Program towards the costs of the Phase 1b/2a and the Phase 2b CONTROL studies April 2014 – designated as an Orphan drug by the FDA |
| NYSE American: TMBR Unmet Need: An efficacious and safe treatment that can be used long term to cover a large body surface area .. 9 Congenital Ichthyosis – Quick facts A rare disorder of keratinization affecting ~ 80,000 people in the U.S. and >1.5 million globally Prevalence • X - Linked Ichthyosis has a prevalence of 1:2,000 - 1:6,000 males • Lamellar Ichthyosis has a prevalence of 1:100,000 - 200,000 Quality of life is significantly affected • Skin inflammation and fragility, pruritus, fissuring and cracking of thickened skin, ectropion, anhidrosis, and in some severe cases, an increased susceptibility to infection • In Lamellar Ichthyosis, almost the whole body can be covered and be severe • Increased rates of depression, anxiety and decreased quality of life on multiple validated clinical measures No FDA Approved treatments 2017 Yale Copyright 2017 Yale Copyright 2017 Yale Copyright |
| NYSE American: TMBR 10 TMB - 001 - Topical Isotretinoin in CI Patented topical formulation that minimizes systemic absorption, thereby allowing for chronic treatment Isotretinoin is well known as the most effective therapy for several skin conditions, including ichthyosis, but it is only available orally, where high dose, chronic oral therapy cannot be tolerated due to systemic toxicity TMB - 001 utilizes patented IPEG ™ delivery system to target isotretinoin delivery to the epidermis and dermis which minimizes systemic absorption and removes irritating excipients We believe TMB - 001 has strong market protection • Orphan exclusivity - 7 years in U.S. and 10 years in E.U. and Japan • Patents awarded for formulation, additional patents pending Market Opportunity • $250 million annual estimated U.S. market size at peak* Development Stage • Phase 1b/2a POC study in CI completed in 2018 • Positive data on clinically meaningful safety and efficacy endpoints and no significant systemic absorption • Phase 2b CONTROL study completed in Q4 2021 • Demonstrated clinically meaningful efficacy with a favorable safety profile • Successful end - of - Phase 2 meeting with FDA competed in Jan 2022 • Phase 3 ASCEND study being initiated by June 30, 2022 2018 FDA Orphan Products Grant Recipient • Awarded $1.5 million from the FDA to run the Phase 1b/2a and Phase 2b Clinical Trials * Based on Timber estimates |
| NYSE American: TMBR 11 TMB - 001 Phase 2b Study Design Title A Vehicle Controlled Study to Evaluate Safety and Efficacy of Topical TMB - 001 for Treatment of Congenital Ichthyosis Primary Objective Proportion of subjects with Visual Index of Ichthyosis Severity (VIIS) treatment success, defined as 50% or greater decrease in VIIS scaling score Secondary Objectives • IGA decrease in disease severity by at least 2 grades • Patient Reported Outcomes (Itch, Dermatology Life Quality Index) Study Design • Randomized, parallel, double - blind, vehicle - controlled study • Evaluating the safety and efficacy of two concentrations of topical TMB - 001 • Multi - center global trial (U.S. and Australia) Study Population Moderate to Severe X - linked and Lamellar Ichthyosis 9 years and older Patient Number 34 patients Therapy Twice daily application Time Schedule 12 weeks |
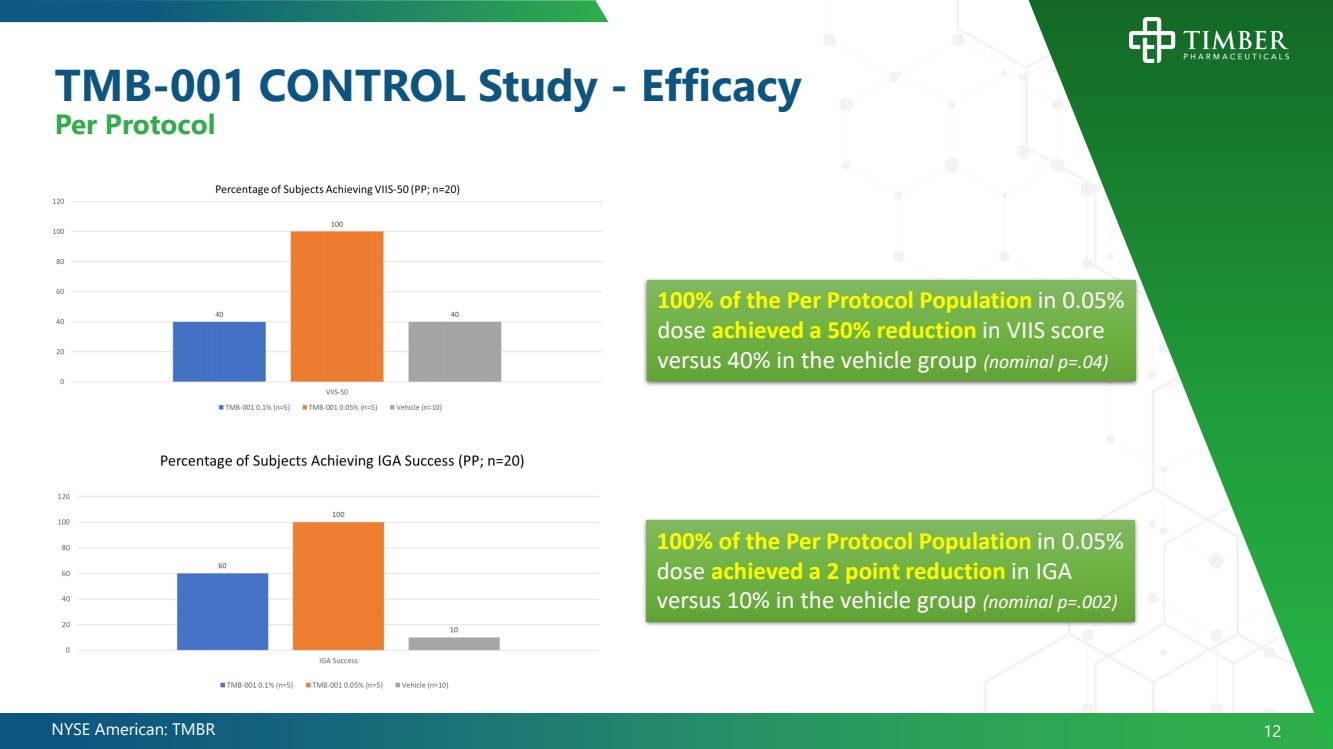
| NYSE American: TMBR 12 TMB - 001 CONTROL Study - Efficacy Per Protocol 100% of the Per Protocol Population in 0.05% dose achieved a 50% reduction in VIIS score versus 40% in the vehicle group (nominal p=.04) 100% of the Per Protocol Population in 0.05% dose achieved a 2 point reduction in IGA versus 10% in the vehicle group (nominal p=.002) |
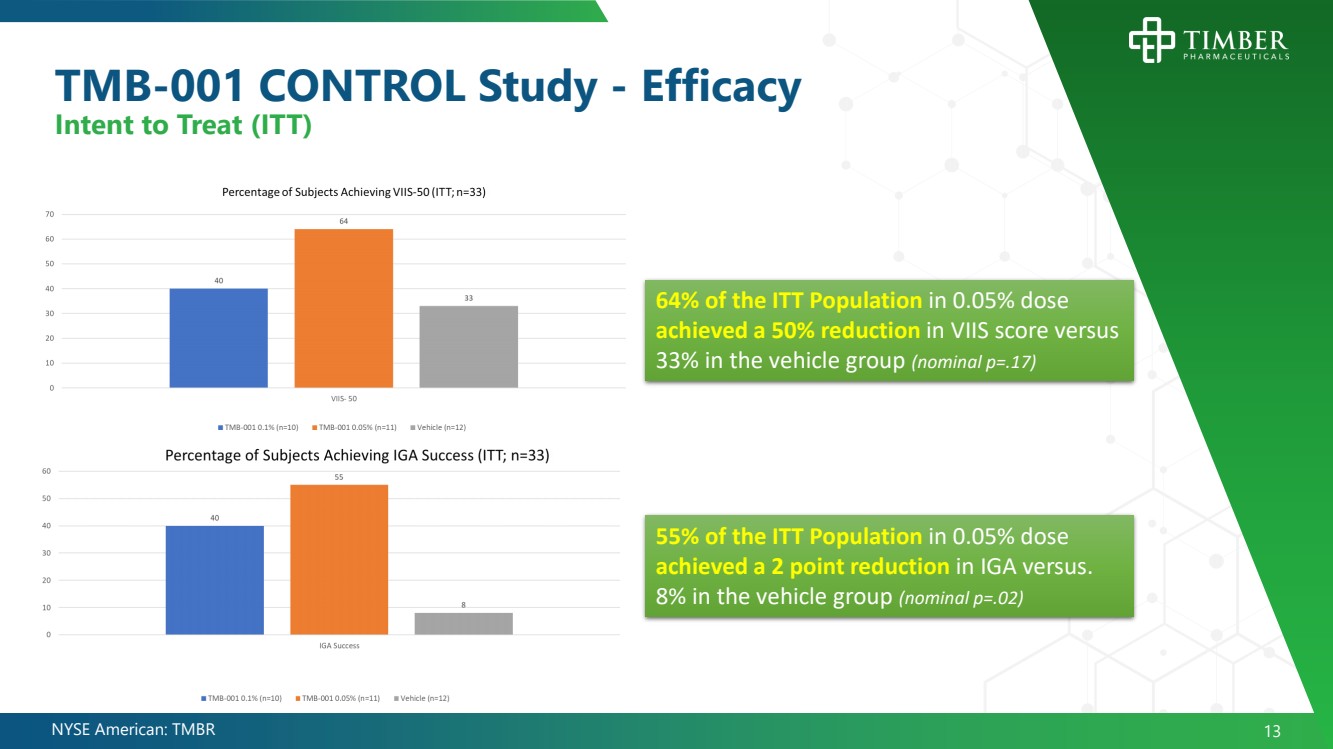
| NYSE American: TMBR 13 TMB - 001 CONTROL Study - Efficacy Intent to Treat (ITT) 64% of the ITT Population in 0.05% dose achieved a 50% reduction in VIIS score versus 33% in the vehicle group (nominal p=.17) 55% of the ITT Population in 0.05% dose achieved a 2 point reduction in IGA versus. 8% in the vehicle group (nominal p=.02) |
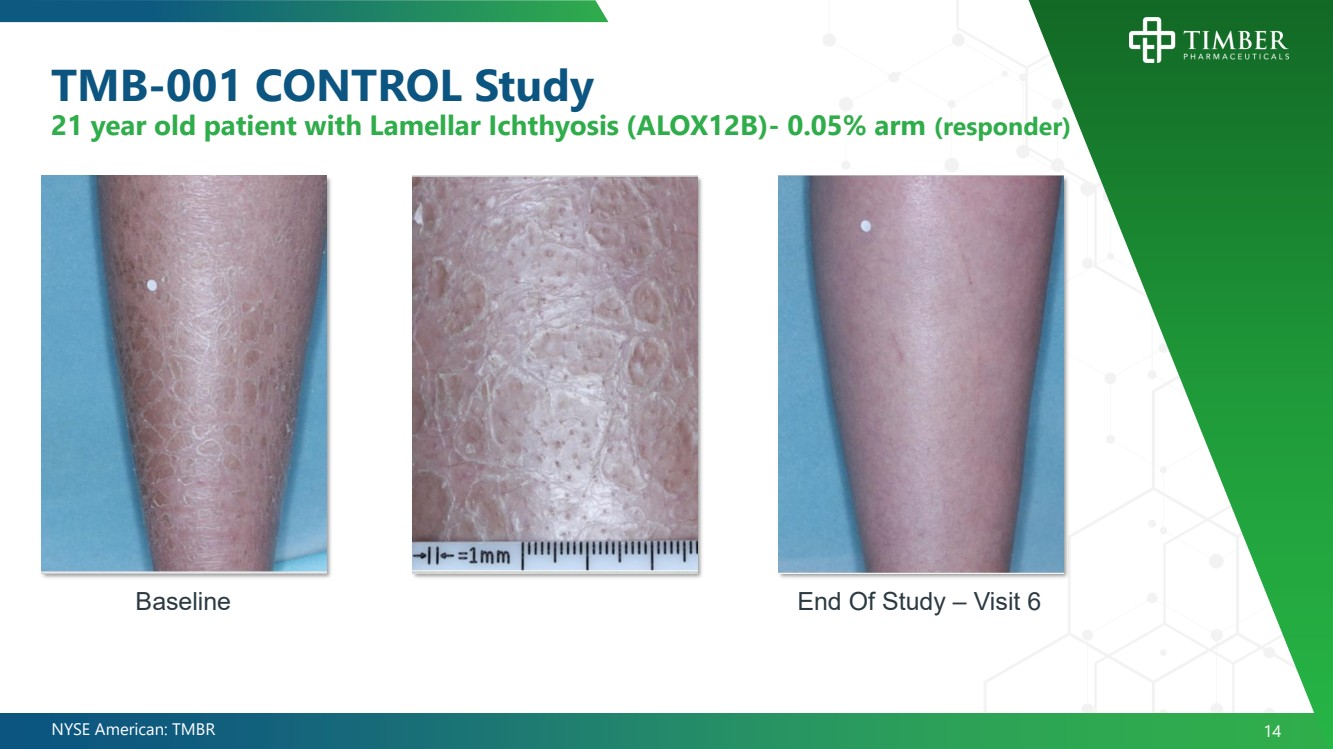
| NYSE American: TMBR 14 TMB - 001 CONTROL Study 21 year old patient with Lamellar Ichthyosis (ALOX12B) - 0.05% arm (responder) End Of Study – Visit 6 Baseline |
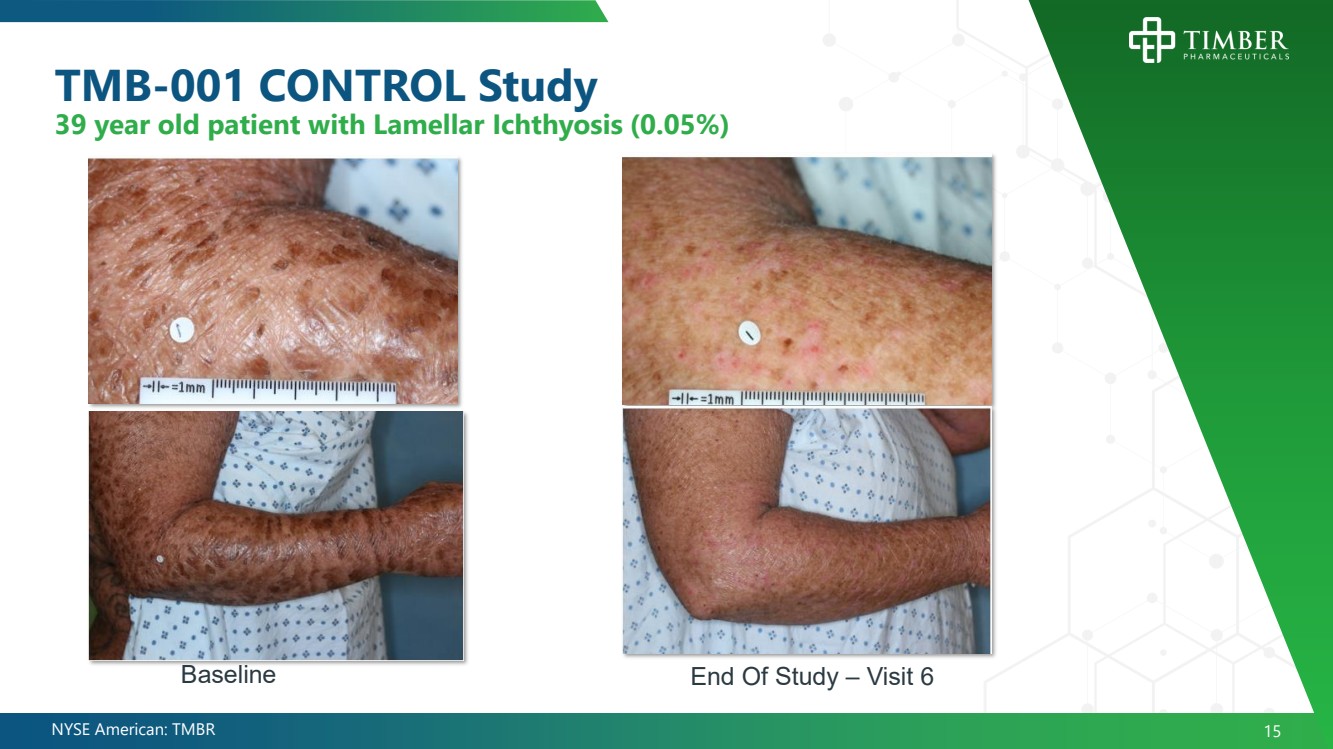
| NYSE American: TMBR 15 TMB - 001 CONTROL Study 39 year old patient with Lamellar Ichthyosis (0.05%) End Of Study – Visit 6 Baseline |
| NYSE American: TMBR 16 TMB - 001 Phase 3 Study Design Title The ASCEND Study: A Phase 3, Multicenter, Double Blind Vehicle Controlled Study of TMB - 001 with a parallel Optional Maximal Use Arm - in the treatment of RXLI or ARCI Ichthyosis in Subjects Aged > 6 Years Primary Objectives • To ascertain the efficacy of TMB - 001 0.05% topical ointment as a treatment for CI compared with Vehicle during a 12 - week treatment using IGA (>2 point reduction in IGA from Baseline to Week 12) followed by a 12 - week randomized maintenance study • To determine systemic exposure of isotretinoin and metabolites after TMB - 001 0.05% applications Secondary Objectives • VIIS decrease in disease severity by at least 50% from Baseline • Patient Reported Outcomes (Itch, Quality of Life) Study Design • Randomized, parallel, double - blind, vehicle - controlled study • Evaluating the safety and efficacy topical TMB - 001 0.05% • Multi - center global trial (U.S., Canada and E.U.) Study Population Moderate to Severe X - linked and ARCI Ichthyosis; 6 years and older Patient Number 110 patients 32 patients Maximal Use Arm Therapy Induction Period (3 weeks) of once daily application, followed by twice daily application Time Schedule 12 weeks therapy + 12 weeks maintenance |
| NYSE American: TMBR 17 17 NYSE American: TMBR TMB - 002 – Topical Rapamycin Facial Angiofibromas (FAs) in Tuberous Sclerosis Complex (TSC) TMB - 003 - Topical Sitaxsentan Sclerotic Skin Diseases |
| NYSE American: TMBR 18 TMB - 002 Topical Rapamycin (Pascomer®) Pascomer® is being developed for territories outside of the United States where Facial Angiofibromas remain a high unmet need. Proprietary topical formulation of rapamycin (1.0%, 0.5%, and 0.1%) • Additional 5% strength assessed in preclinical toxicity studies Utilizes trade secret lipid crystalline vehicle system designed to improve stability and delivery of unstable APIs • Compounded formulations that are sometimes currently used lack consistent quality, potency, and homogeneity • Other topical rapamycin formulations have short shelf lives and require refrigeration • Pascomer has demonstrated room temperature stability for 0.5%, 1.0%, and 5.0% formulations Development Stage: • Efficacy of topical rapamycin demonstrated in multiple well controlled studies and dozens of published case reports • Recruitment on Phase 2b study (one of two pivotal studies required for registration) in FAs in TSC has been completed; Data expected in Q3 2022 • Timber will evaluate all potential strategic opportunities for the asset in markets outside of the U.S. and/or other indications |
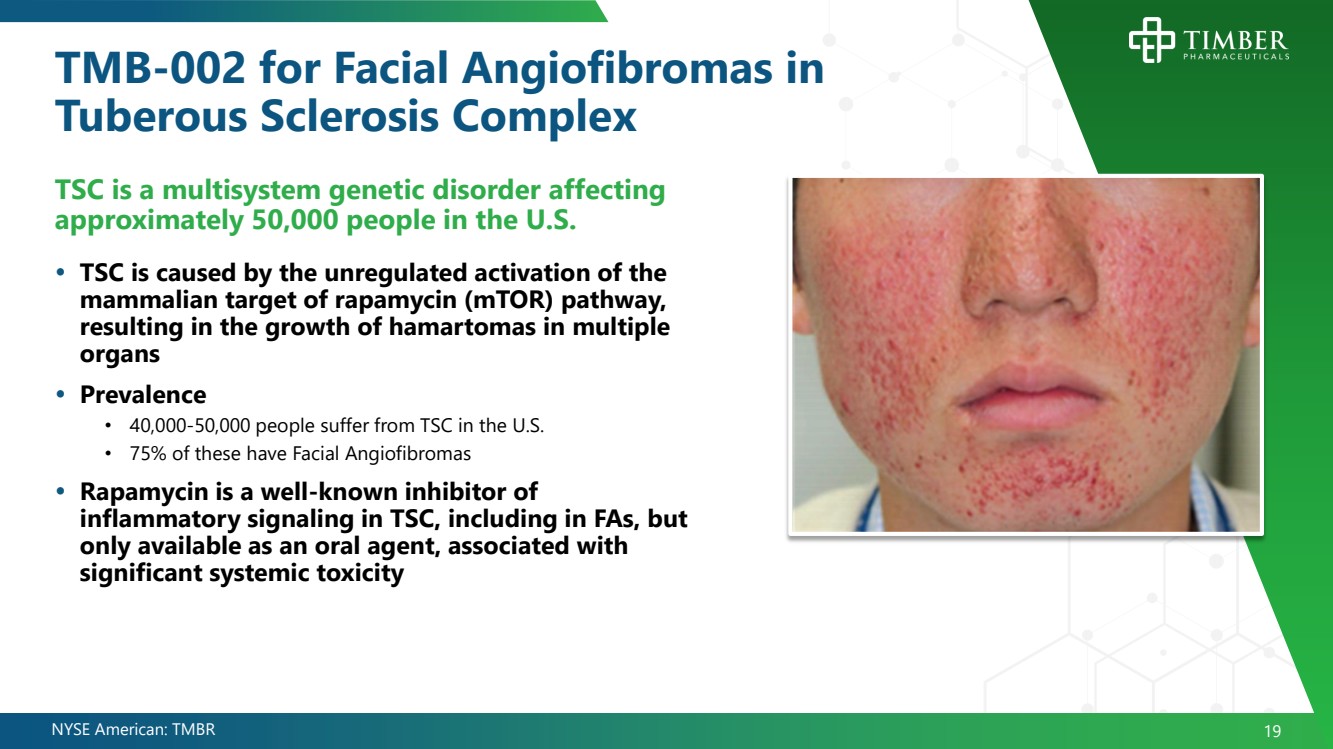
| NYSE American: TMBR 19 TMB - 002 for Facial Angiofibromas in Tuberous Sclerosis Complex TSC is a multisystem genetic disorder affecting approximately 50,000 people in the U.S. TSC is caused by the unregulated activation of the mammalian target of rapamycin (mTOR) pathway, resulting in the growth of hamartomas in multiple organs Prevalence • 40,000 - 50,000 people suffer from TSC in the U.S. • 75% of these have Facial Angiofibromas Rapamycin is a well - known inhibitor of inflammatory signaling in TSC, including in FAs, but only available as an oral agent, associated with significant systemic toxicity |
| NYSE American: TMBR 20 TMB - 002 Phase 2b Study Design Title Dose - Ranging Efficacy and Safety Study of Topical Rapamycin Cream for Facial Angiofibromas Associated With Tuberous Sclerosis Complex Primary Objective • Proportion of subjects with IGA treatment success defined as clear or almost clear with an improvement of at least two grades from baseline Secondary Objectives • Time from the first dose to IGA success • Change in IGA from baseline • Change in the Facial Angiofibroma Severity Index (FASI) from baseline • Percentage improvement in FA, as assessed by the participant or parent/caregiver • Percentage improvement in FA, as assessed by the clinician • Change in FA on a 5 - point scale, as assessed by the participant or parent/caregiver Study Design • Phase 2/3, multi - center, double - blind, placebo - controlled, randomized, parallel - group, dose - response comparison • Efficacy and safety of two concentrations of TMB - 002 • Multi - center global trial (U.S., Europe, Australia, NZ) Study Population Patients aged 6 – 65 with FA severity score of 2 or 3 on the IGA Patient Number 108 patients randomized Therapy Once daily application Time Schedule 26 weeks |
| NYSE American: TMBR 21 TMB - 003 Topical Sitaxsentan for Sclerotic Skin Diseases Highly Selective ET - A inhibitor Sitaxsentan is a highly selective (6,500:1) ET - A receptor antagonist. • It was previously on the market in the E.U., Canada and Australia as an oral treatment for PAH but was voluntarily withdrawn from global market in 2010 due to risks of liver toxicity • Demonstrated benefit in cutaneous symptoms associated with connective tissue disease (CTD) in multiple case reports TMB - 003 • Topical Sitaxsentan in development • Systemic safety concerns may be addressed via local administration • We believe TMB - 003 has strong market protection based on: • Method of Use patents pending for conditions of cutaneous fibrosis and hyperpigmentation • Orphan Drug Designation (ODD) was granted in Systemic Sclerosis, plan to pursue additional designations in other indications • New Chemical Entity (NCE) exclusivity Role of ET - A Inhibition Endothelin - 1 is a potent and long - lasting vasoconstrictor and profibrotic known to play a role in skin fibrosis • Endothelin - 1 acts through two receptors, ET - A and ET - B, known to have broadly opposing functions • Hypothesized that selectively antagonizing ET - A may lead to more effective reductions in collagen production Endothelin Receptor Antagonists are a class of drugs currently on the market (in oral form) for the treatment of Pulmonary Arterial Hypertension (PAH) • A review of the literature showed no difference in endothelin selectivity in PAH, however in the subset of PAH patients with connective tissue disease, selective ET - A inhibition was beneficial in improving 6 - minute walk measurements |
| NYSE American: TMBR 22 Lichen Sclerosis Rare chronic disease of vulvae and perianal areas which affects approximately 160,000 women in the U.S.* Lichen Sclerosis affects both sexes, but is predominant in women • Prevalence 0.1 - 1.7% of women; diagnosed more by gynecologists than dermatologists • Bimodal presentation: pre - pubertal girls, post - menopausal women • Associated with other autoimmune diseases in 28% of women (e.g., morphea, autoimmune thyroiditis, alopecia areata, vitiligo, pernicious anemia) Quality of life can be significantly affected • Disfiguring and extremely pruritic • May be precursor to squamous cell carcinoma of the vulva • Extremely painful No FDA Approved treatments for any cutaneous symptoms in lichen sclerosis • Ultrapotent corticosteroid ointments (e.g., clobetasol) are primary therapy • 96% improvement in symptoms but only 23% of patients return to normal skin, 68% partial return to normal skin * Based on Timber estimates |
| NYSE American: TMBR 23 Scleroderma Rare connective tissue disorder characterized by abnormal thickening of the skin Two potential markets: • Moderate to Severe Localized Scleroderma which generally only affects the skin • More common in children, with a prevalence of approximately 90,000 people in the U.S. • Systemic sclerosis where fibrotic changes may affect the connective tissue in the skin plus other organs • More common in adults, with a prevalence of approximately 100,000 people in the U.S. Quality of life can be significantly affected • Disfiguring • Can cover joints and cause pain, affect movement / mobility, etc. • Extremely painful No FDA Approved treatments for any cutaneous symptoms in scleroderma • Off - label therapies have significant side effects and rarely lead to benefit • Bosentan ( Tracleer ®), a non - selective endothelin receptor antagonist approved in the E.U. for reducing the number of new digital ulcers in patients with systemic sclerosis and ongoing digital ulcer disease Ref: Progressive CutaneoU.S .. Scleroderma | Consultant360 |
| NYSE American: TMBR Multiple Barriers to Entry 24 Orphan exclusivity granted for all three strategic assets • 7 years in U.S., 10 years in E.U. and Japan Patents granted and pending globally Trade secrets in formulation/manufacturing • Cytotoxic, requires containment, photolabile, prone to oxidation, temperature sensitivities, stability challenges, etc. TMB - 001 TMB - 002 TMB - 003 • WO 2017/074982 A1 – Isotretinoin Formulations and Uses and Methods Thereof • Patent granted in the U.S. (U.S. Patent No. 10,933,018) • Decision of Grant in Japan and Australia • Pending applications in Canada, China, EPO, Mexico, South Korea and U.S. • Protection through at least 2035 • Freedom to Operate (FTO) complete • Further expansion planned around dosing, PK, and other methods of use • AUS 2020277132 – formulation application licensed from AFT, expansion is planned around other methods of use • Will be converted to PCT and pursued in key countries • Priority date of November 2020 • FTO complete • WO 2019/173215 A1 – Compositions and methods for treating cutaneous fibrosis • Pending in Australia, Brazil, Canada, China, EPO, Israel, Japan, Korea, and the U.S. • Priority date of March 2018 • WO 2019/173219 – Compositions and methods for treating pigmentation disorders • Pending in the U.S. • Priority date of March 2018 • Landscape search complete • Expansion planned around dosing, PK, and other methods of use |
| NYSE American: TMBR World Class Clinical Advisors and Partners 25 • Amy S. Paller , MD Chair and Professor of Dermatology and Pediatrics, Northwestern University • Keith Choate, MD, PhD Professor of Dermatology, Pathology, and Genetics, Yale University • Joyce Teng, MD Professor and Director of Pediatric Dermatology, Stanford University • Christopher Bunick , MD, PhD Associate Professor of Dermatology, Yale School of Medicine Development Partner on TMB - 002 (Pascomer®) program Patient Advocacy Groups related to TMB - 001 and TMB - 002 Clinical Advisors Partners |
| NYSE American: TMBR John Koconis, MBA, Chairman and CEO • Seasoned Pharmaceutical executive with over 25 years experience leading successful teams in global markets and having launche d multiple market leading products in Dermatology • President & CEO of LEO Pharma (U.S.), established a market leading dermatology company with focus on Psoriasis and AK • Global Lead for Dermatology and Respiratory Disease at Sanofi Genzyme, led the Global launch of Dupilumab in AD • EVP & CCO at Castle Creek Pharmaceuticals, built a rare/orphan dermatology company with a focus on EBS Alan Mendelsohn, MD, CMO • Board certified in Pediatrics and Pediatric Cardiology with strong management skills and over 20 years of drug development a nd medical affairs experience in major pharmaceutical companies • Associate Vice President of Dermatology Medical Affairs at Sun Pharma, focused on the post - clinical development of Tildrakizumab (ILUMYA), an IL - 23p19 inhibitor for patients with moderate to severe plaque psoriasis • Senior Director, Rheumatology Team Leader, U.S. Medical Affairs .at Pfizer leading the U.S. organization in supporting the pre - launch and launch medical activities for Xeljanz (tofacitinib) • Senior Director of Immunology R&D at Johnson & Johnson/Janssen) in Phase II/III clinical trial development for multiple biolo gic agents targeting rheumatoid arthritis and psoriatic arthritis • Authored or co - authored over 200 peer reviewed abstracts and manuscripts in the areas of dermatology, rheumatology and cardiolog y Leadership Team 26 Strong Balance between Entrepreneurial Acumen and Proven Operational Leadership |
| NYSE American: TMBR Joseph Lucchese, CFO • Chief Financial Officer of Timber Pharmaceuticals • Founding member and Managing Director of Oncology Partners; Managing Director at BlueStone Capital Partners, headed the Private Placement desk • Portfolio Manager and Chief Financial Officer of the MicroCap Fund, a NASDAQ - listed private equity fund that invested in early - stage companies in the healthcare and information technology sectors • Began career as a member of Chase Manhattan Bank’s Merchant Banking Group, part of the team that financed highly leveraged tr ans actions and managed the bank’s private equity portfolio Jessica Bruno - Raiz , MPH, VP Global Program Management and Clinical Operations • Over 20 years experience working in R&D with a proven track record of influencing global strategy for optimal clinical develo pme nt. • Has held roles with increasing responsibilities within Clinical Development/ Clinical Operations where she spearheaded design ing , developing and implementing innovative processes, best practice methodologies and strategic initiatives with the aim of identifying effi cie ncies and optimizing project outcomes • While at Novartis and GSK, led cross functional teams focused on Phase II/III global clinical studies in Oncology and Rx to O TC switch. Also has held positions in Learning and Development, allowing her to develop a curriculum to educate peers within R&D as well as a Pr ocess Improvement Manager with training in Lean Six Sigma Leadership Team 27 Strong Balance between EntreprenE.U.rial Acumen and Proven Operational Leadership |
| NYSE American: TMBR Independent Board of Directors 28 Lead Independent Board Member Respected expert in SEC reporting and internal controls with extensive financial leadership experience as CFO at several public life sciences companies. Independent Board Member Professor of Dermatology at New York University School of Medicine, where he also serves as Chief of Allergy and Contact Dermatitis, Vice Chairman of Clinical Affairs, and Director of Occupational and Environmental Dermatology. Independent Board Member Edward Sitar, CPA David Cohen, MD Lubor Gaal , PhD Gianluca Pirozzi , MD, PhD Independent Board Member 20 years’ experience in drug development, currently Senior Vice President, Head of Clinical Development and Translational Sciences at Alexion Pharmaceuticals. CFO at Targovax ASA, previously Managing Director at Locust Walk and prior Head of Business Development at Almirall , a global leader in Dermatology. Dr. Gaal brings extensive experience in pharmaceutical corporate and business development transactions across various therapeutic areas and geographies |
| NYSE American: TMBR Key Takeaways 29 Rare, High Need Medical Dermatology Large Market Potential Multiple Shots on Goal De - Risked Portfolio Management Expertise • High unmet needs with large market potential • All strategic programs have been granted Orphan Drug status • No approved treatments for indications • Portfolio of orphan indications • Market expansion potential in broader dermatology indications for all programs • Breakthrough therapy status for Topical Isotretinoin for Congenital Ichthyosis • Topical Rapamycin for Facial Angiofibromas in TSC outside of North America, Australia, NZ and SE Asia • Topical sitaxsentan for sclerotic skin diseases • Clinical proof of concept established in two assets • Proven mechanisms of action with decades of clinical experience • Well - known CMC and safety profiles • Deep experience in dermatology and rare disease • Independent Board of Directors with life sciences expertise in drug development, dermatology, and finance |
| www.timberpharma.com For additional Investor Information please contact: John Koconis Stephanie Prince Chairman and CEO PCG Advisory jkoconis@timberpharma.com sprince@pcgadvisory.com (646) 863 - 6341 NYSE American: TMBR |
| www.timberpharma.com APPENDIX/Additional Slides NYSE American: TMBR |
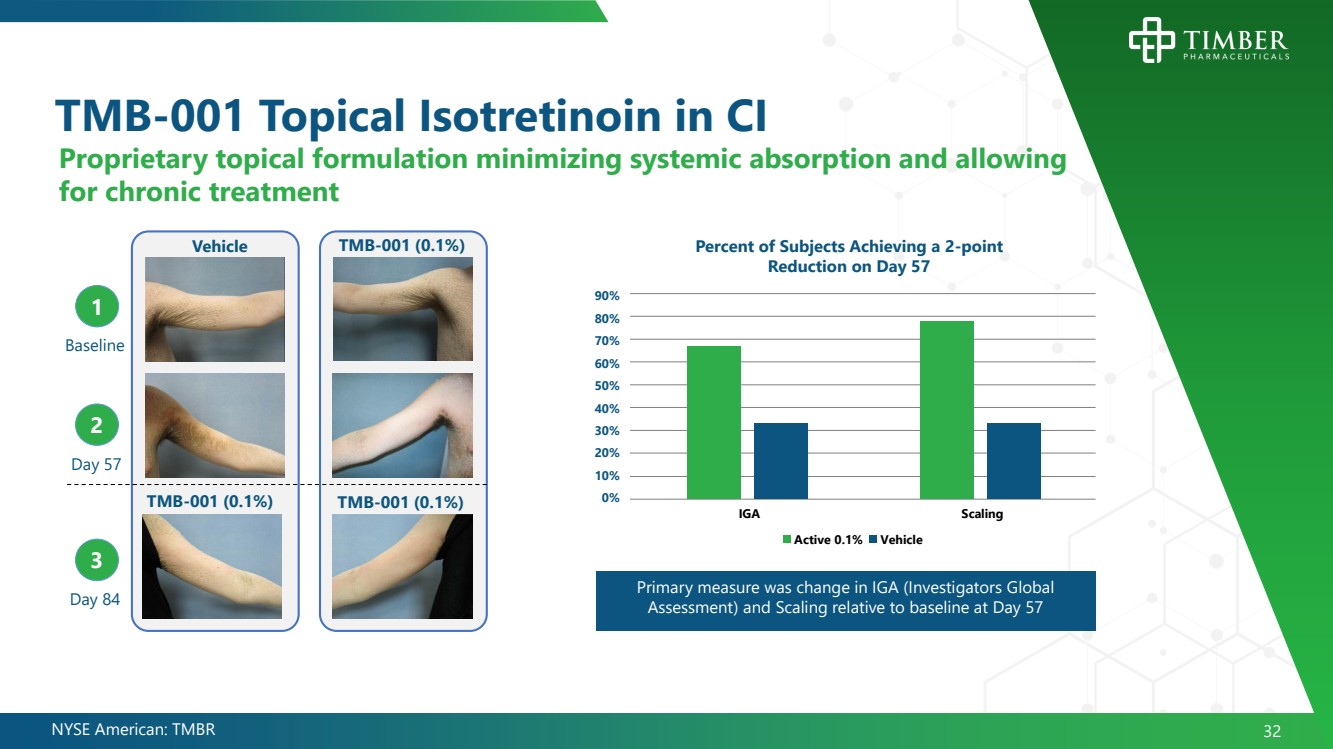
| NYSE American: TMBR 32 TMB - 001 Topical Isotretinoin in CI Baseline Day 57 Day 84 2 3 1 Vehicle TMB - 001 (0.1%) TMB - 001 (0.1%) TMB - 001 (0.1%) Percent of Subjects Achieving a 2 - point Reduction on Day 57 90% 80% 70% 60% 50% 40% 30% 20% 10% 0% IGA Scaling Vehicle Active 0.1% Primary measure was change in IGA (Investigators Global Assessment) and Scaling relative to baseline at Day 57 Proprietary topical formulation minimizing systemic absorption and allowing for chronic treatment |
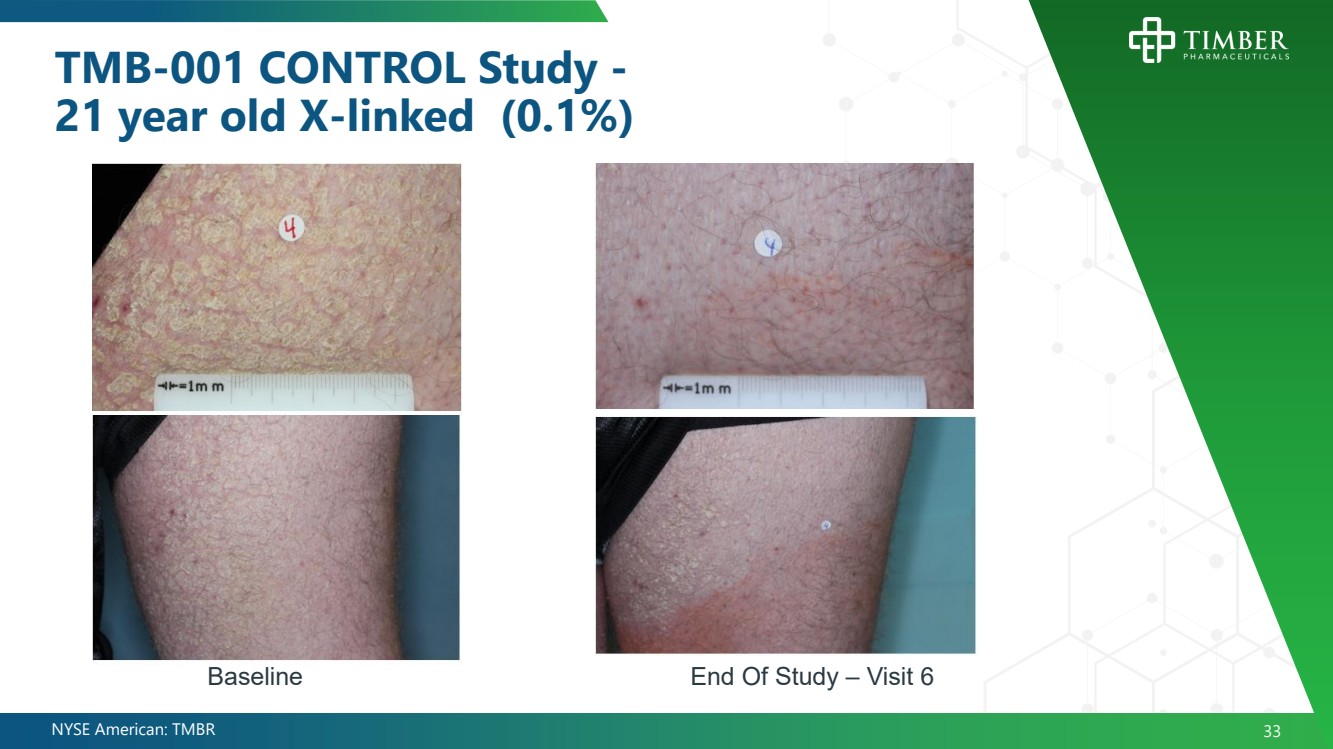
| NYSE American: TMBR 33 TMB - 001 CONTROL Study - 21 year old X - linked (0.1%) End Of Study – Visit 6 Baseline |
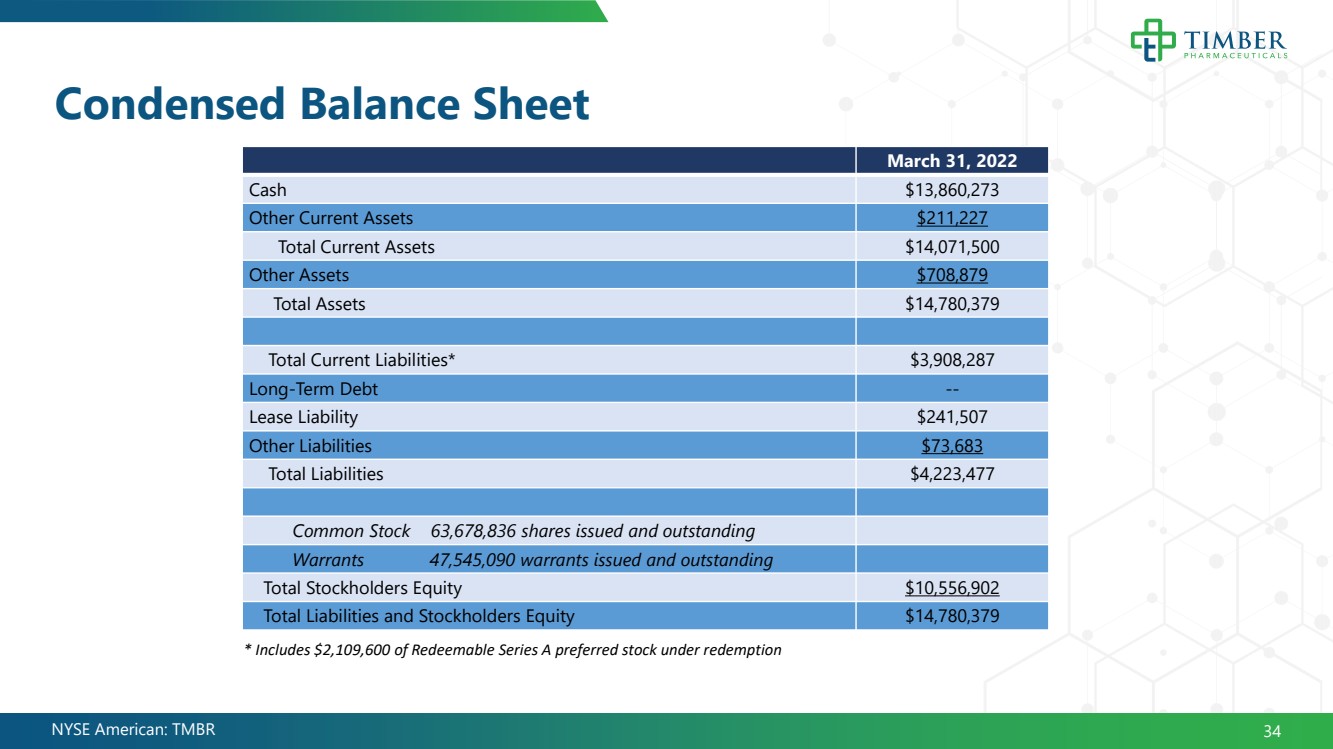
| NYSE American: TMBR Condensed Balance Sheet 34 March 31, 2022 Cash $13,860,273 Other Current Assets $211,227 Total Current Assets $14,071,500 Other Assets $708,879 Total Assets $14,780,379 Total Current Liabilities* $3,908,287 Long - Term Debt -- Lease Liability $241,507 Other Liabilities $73,683 Total Liabilities $4,223,477 Common Stock 63,678,836 shares issued and outstanding Warrants 47,545,090 warrants issued and outstanding Total Stockholders Equity $10,556,902 Total Liabilities and Stockholders Equity $14,780,379 * Includes $2,109,600 of Redeemable Series A preferred stock under redemption |