About AIPAC
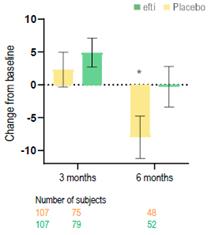
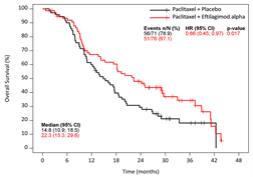
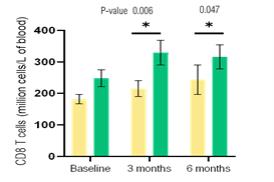
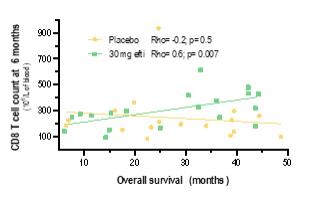
Active Immunotherapy Paclitaxel (AIPAC) is a multicentre, placebo-controlled, double-blind, 1:1 randomised Phase IIb clinical trial in HER2-negative/HR positive metastatic breast cancer.
The study is evaluating the combination of efti with paclitaxel chemotherapy. 227 HER2-negative/HR positive metastatic breast cancer patients are randomised 1:1 to a chemo-immunotherapy arm (efti plus paclitaxel) or to a comparator arm (placebo plus paclitaxel). Patients receive weekly paclitaxel at days 1, 8 and 15, with either efti or placebo injected subcutaneously on days 2 and 16 of each 4-week cycle, repeated for 6 cycles. Thereafter, patients pass over to the maintenance phase with efti alone.
For more information regarding the AIPAC trial, visit clinicaltrials.gov (identifier: NCT02614833) and https://www.ncbi.nlm.nih.gov/pubmed/30977393.
About TACTI-002
TACTI-002 (Two ACTive Immunotherapies) is being conducted in collaboration with Merck & Co., Inc., Kenilworth, NJ, USA (known as “MSD” outside the United States and Canada). The study is evaluating the combination of efti with MSD’s KEYTRUDA® (pembrolizumab) in up to 183 patients with second line head and neck squamous cell carcinoma or non-small cell lung cancer in first and second line.
The trial is a Phase II, Simon’s two-stage, non-comparative, open-label, single-arm, multicentre clinical study that is taking place in study centres across Australia, Europe, the UK and US.
Patients participate in one of the following:
| | • | | Part A - first line non-small cell lung cancer (NSCLC), PD-X naive |
| | • | | Part B - second line NSCLC, PD-X refractory |
| | • | | Part C - second line head and neck squamous cell carcinoma (HNSCC), PD-X naive |
TACTI-002 is an all-comer study in terms of PD-L1 status, a well-known predictive marker for response to pembrolizumab monotherapy especially in NSCLC and HNSCC. PD-L1 expression is typically reported in three groups for NSCLC: < 1%, 1-49% and ³ 50% (Tumour Proportion Score or TPS) and in HNSCC: < 1, 1-19 and ³ 20 (Combined Positive Score or CPS). Patients with a high PD-L1 status are typically more responsive to anti-PD-1 therapy such as pembrolizumab, whereas those with low PD-L1 status are overall less responsive.
More information about the trial can be found on Immutep’s website or on ClinicalTrials.gov (Identifier: NCT03625323).
KEYTRUDA® is a registered trademark of Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA.
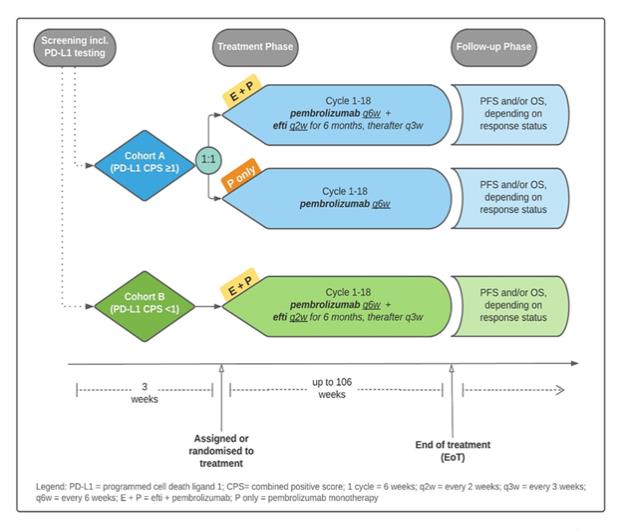
About TACTI-003
TACTI-003 is a Phase IIb clinical trial in first line head and neck squamous cell carcinoma (HNSCC) in collaboration with Merck & Co., Inc., Kenilworth, NJ, USA, known as “MSD” outside the United States and Canada. It will evaluate efti in combination with MSD’s KEYTRUDA® (pembrolizumab) as a first line therapy in unresectable recurrent or metastatic HNSCC patients with PD-L1 negative and PD-L1 positive (CPS ³ 1) tumours. It will be a randomised, controlled clinical study in approximately 154 first line HNSCC patients and will take place across Australia, Europe and the US in up to 35 clinical sites.
7