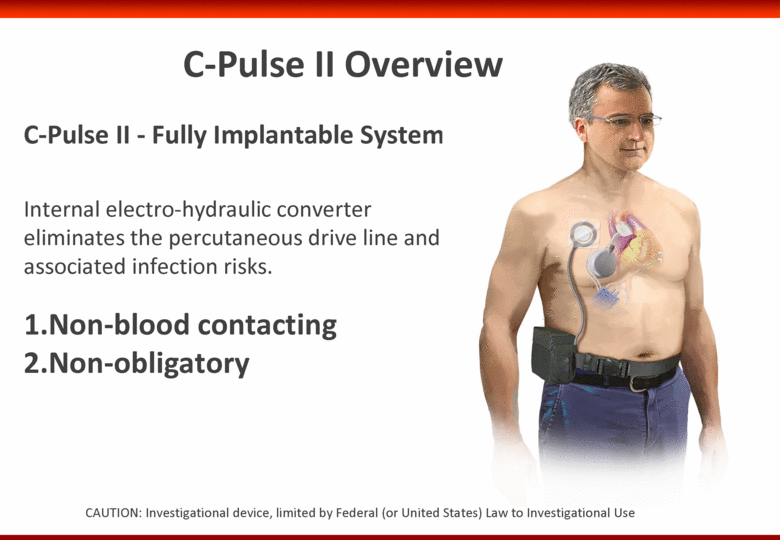
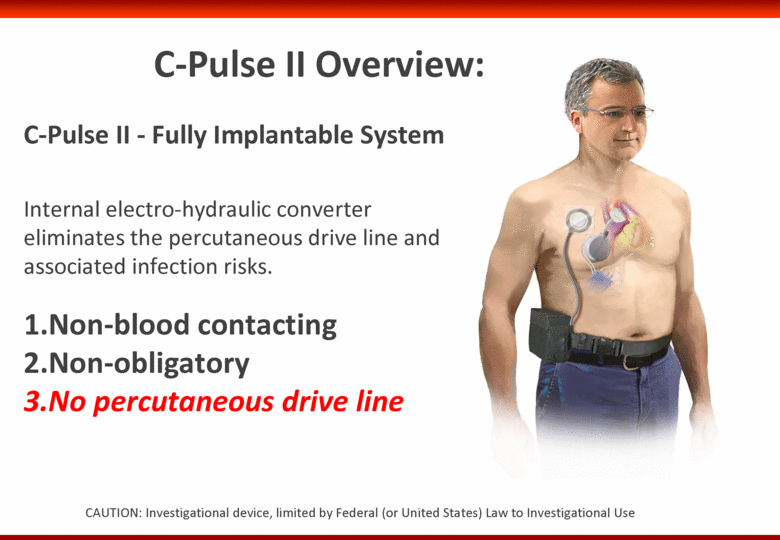
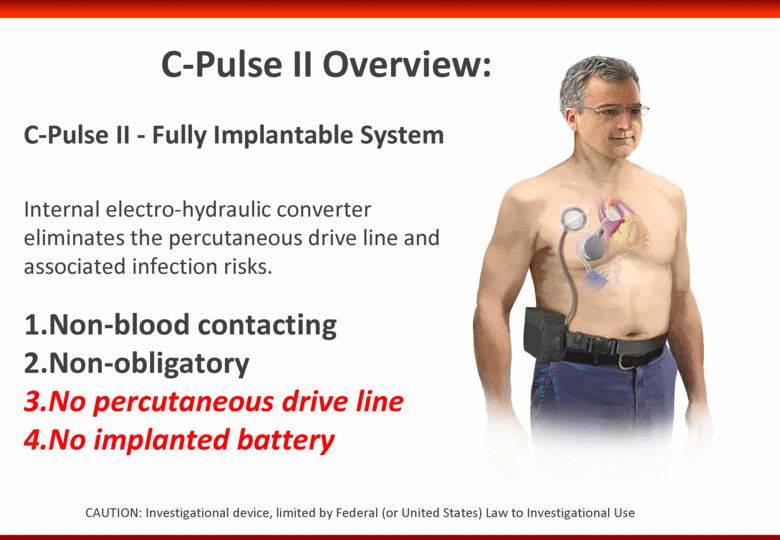
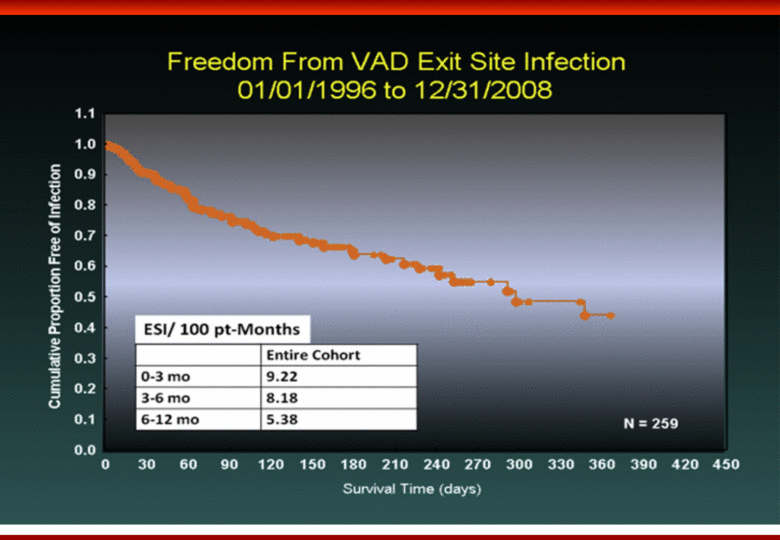
Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
NUWE similar filings
- 21 Mar 14 Entry into a Material Definitive Agreement
- 5 Mar 14 Regulation FD Disclosure
- 13 Jan 14 Sunshine Heart Provides Corporate Update
- 29 Oct 13 Regulation FD Disclosure
- 19 Sep 13 Sunshine Heart, Inc. Prices $40.0 Million Public Offering of Common Shares
- 18 Sep 13 Sunshine Heart, Inc. Announces Proposed Public Offering of Common Shares
- 10 Sep 13 Departure of Directors or Certain Officers
Filing view
External links