Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
NUWE similar filings
- 5 Jun 23 Nuwellis Announces the Appointment of Mike McCormick to its Board of Directors
- 22 May 23 Submission of Matters to a Vote of Security Holders
- 9 May 23 Nuwellis, Inc. Announces First Quarter 2023 Financial Results
- 14 Mar 23 March 2023 Investor Presentation ©2023 Nuwellis, Inc
- 28 Feb 23 Nuwellis, Inc. Announces Fourth Quarter and Full Year 2022 Financial Results
- 18 Jan 23 Departure of Directors or Certain Officers
- 9 Jan 23 Nuwellis Reports Preliminary Fourth Quarter and Full Year 2022 Revenue Results
Filing view
External links
Exhibit 99.1

March 2023 Investor Presentation ©2023 Nuwellis, Inc

Safe Harbor Statement Forward Looking Statement This presentation contains forward‐looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended and Section 21E of the Securities and Exchange Act, as amended regarding our plans, expectations, beliefs, estimates, goals and outlook for the future that are intended to be covered by the Private Securities Litigation Reform Act of 1995. Except for statements of historical fact, all forward‐looking statements are management’s present expectations and are not guarantees of future events and are subject to a number of known and unknown risks and uncertainties and other factors that may cause actual results to differ materially from those expressed in, or implied by, such forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “could,” “would,” “should,” “plan,” “predict,” “potential,” “project,” “promising,” “expect,” “estimate,” “anticipate,” “intend,” “goal,” “strategy,” “milestone,” and similar expressions and variations thereof. Various factors could cause actual results to differ materially from these statements including our ability to execute on our commercial strategy and to grow our Aquadex® business, the possibility that we may be unable to raise sufficient funds necessary for our anticipated operations, our post-market clinical data collection activities, benefits of our products to patients, our expectations with respect to product development and commercialization efforts, our ability to increase market and physician acceptance of our products, potentially competitive product offerings, intellectual property protection, our expectations regarding anticipated synergies with and benefits of the Aquadex business, our business strategy, market size, potential growth opportunities and the other risks set forth under the caption “Risk Factors” and elsewhere in our periodic and other reports filed with the U.S. Securities and Exchange Commission (“SEC”), including our Annual Report on Form 10‐K for the fiscal year ended December 31, 2022 and subsequent reports. We are providing this information as of the date of this presentation, and we undertake no obligation to update any forward-looking statements contained in this presentation as a result of new information, future events or otherwise. Although the Company believes that the forward‐looking statements are reasonable and based on information currently available, it can give no assurances that the Company’s expectations are correct. All forward-looking statements are expressly qualified in their entirety by this cautionary statement. Financial and Statistical Data This presentation also contains estimates and other statistical data made by independent parties and by us relating to market shares and other data about our industry. These data involve a number of assumptions and limitations and have not been reviewed or audited by our independent registered accounting firm. You are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. Neither we nor our advisors or representatives makes any representations as to the accuracy or completeness of that data or undertake to update such data after the date of this presentation. Trademarks The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products. Additional Information You should read the documents that we have filed with the SEC for more complete information about us. We encourage you to read such documents in full for more detailed information, statistics, reports and clinical trials referenced in this presentation. You may access these documents for free by visiting EDGAR on the SEC website at http://www.sec.gov. Aquadex FlexFlow® and Aquadex SmartFlow® are registered trademarks of Nuwellis, Inc. Aquadex ® is a trademark of Nuwellis, Inc. ©2023 Nuwellis, Inc

is dedicated to transforming the lives of patients suffering from fluid overload through science, collaboration, and innovation. 3 Our Mission ©2023 Nuwellis, Inc

$2B+ and growing addressable market Outpatient market opportunity adds $0.5B+ to addressable market (heart failure and advanced liver disease) Positive ROI and attractive clinical + economic benefits to hospitals and healthcare system Robust body of clinical evidence Scalable consumables driven growth Commercial infrastructure leverage Novel product pipeline Experienced leadership FOR INVESTOR PURPOSES ONLY: NOT FOR PRODUCT PROMOTION ©2022 Nuwellis, Inc. Investment Highlights ©2023 Nuwellis, Inc

Over 200 years’ collective experience in clinical practice and the medical device industry, working with companies including Medtronic, Boston Scientific and Abbott/St. Jude Medical Management team with proven success commercializing many therapies John Jefferies, M.D., joined as Chief Medical Officer Nestor Jaramillo, Jr. President & Chief Executive Officer Lynn Blake Chief Financial Officer Neil P. Ayotte General Counsel, SVP & Chief Compliance Officer Sandra Eayrs Chief Human Resources Officer John Kowalczyk Senior Vice President of Sales & Marketing Laurent Duhoux Vice President of International Business Development Vitaliy Epshteyn Senior Vice President of Operations & Engineering Al Saalabi Vice President of Quality and Regulatory Megan Cease Vice President of Clinical Research and Reimbursement Executive Leadership Team John Jefferies, M.D. Chief Medical Officer ©2023 Nuwellis, Inc

Survival (days) 90-day mortality by intravenous fluid median 1.000 0.975 0.950 0.925 0.900 0 10 20 30 40 50 60 70 80 90 Survival rates with No Excess Fluid Survival rates with Excess Fluid IVF > Median IVF ≤ Median Fluid overload is the leading cause of death for critically ill patients in the ICU within 90 days1 Excess fluid following cardiac surgery leads to three-fold increase in mortality at 90 days2 90% of heart failure hospitalizations are due to signs and symptoms of fluid overload3 Vaara ST et al. Crit Care.2012; 16: 1-11. Pradeep, A. et al. HSR Proc IC and Car An. 2010 Mar; 2(4): 287-296. Costanzo MR, et al. J Am Coll Cardiol. 2017 May 16;69(19):2428-2445. Fluid Overload is Associated with Greater Mortality ©2023 Nuwellis, Inc

Diuretics: Significant Limitations as Current Standard of Care High risk of rehospitalization1 Long-term use of diuretics is associated with kidney damage1-4 Diuretics provide insufficient symptom relief and are associated with worsening heart failure and increased mortality after discharge1 >40% of heart failure patients have poor diuretic response5 1. Costanzo MR, et al. JACC. 2017;69(19)2428-2445. 2.Felker MG & Mentz RJ. JACC. 2012;59(24):2145-53. 3. Al‐Naher et al. Br J Clin Pharmacol. 2018 Jan; 84(1): 5–17. 4. Butler J et al. Am Heart J. 2004 Feb;147(2):331-8. 5. Testani JM, et al. Circ Heart Fail. 2016;9(1):e002370. ©2023 Nuwellis, Inc

SIMPLE FLEXIBLE SMART Reduces hospitalization by 81%1 compared to diuretics Rehospitalizations with Aquadex were 48% lower than the national average at 30 days1 Reduces length of hospital stay when initiated early, resulting in average savings of $3,975 (14%)6-7 No significant changes to kidney function1 Stabilizes or improves cardiac hemodynamics2-5 Predictably removes excess fluid Safe, easy to use and flexible in application Clinically Superior Solution for Fluid Overload 1. Watson R et al. J Cardiac Fail. 2020; 26(10): s56. 2. Kiziltepe, U, et al. Ann Thorac Surg. 2001;71(2): 684-93. 3. Sahoo, TK, et al. Indian J Thorac Cardiovas Surg. 2007;23(2): 116-24. 4. Boga et al. Perfusion. 2000;15:143-50. 5. Onoe et al. Perfusion. 2001;16:37-42.65. 6. Costanzo MR et al. JACC. 2005; 46(11); 2457-51. 7. Costanzo, et. al., ISPOR 23rd Annual Int’l Mtg., May 19-23, 2018, Baltimore, MD, USA. The only device of its kind in the market: Saving lives, time & money ©2023 Nuwellis, Inc

$1B Market1 ~30% of sales Heart Failure $130M Market1 ~30% of sales PEDIATRIC $900M Market1 ~40% of sales CRITICAL CARE TREATING THE MOST VULNERABLE From children2 to the elderly, our therapy is critical to improving care and outcomes $2B+ TAM 1. See Appendix. 2. Approved for use in pediatric patients weighing 20 kg or more. Multiple Growth Opportunities Moving Aquadex to standard of care in fluid overload ©2023 Nuwellis, Inc

Cardiac Surgery Liver Transplants Sepsis Advanced Liver Disease 550,000 patients/year1 12,000 patients/year2 VAD 6,000 patients/year3 1.8M patients/year4 700,000 patients/year5 Adult ECMO 15,000 patients/year6 NEAR-TERM OPPORTUNITIES (In the U.S.) MID to LONG-TERM OPPORTUNITIES (In the U.S.) Expanding Use of Ultrafiltration Across Hospital Specialty UnitsImproves outcomes for fluid overloaded patients 1. Derived from: https://www.grandviewresearch.com/industry-analysis/coronary-artery-bypass-graft-cabg-market and growth rate from: https://www.hcup-us.ahrq.gov/reports/statbriefs/sb171-Operating-Room-Procedure-Trends.pdf. 2. Derived from: https://www.healthline.com/health/liver-transplant-survival and this for growth rate: https://www.marketwatch.com/press-release/organ-transplantation-market-size-to-grow-at-934-cagr-during-the-forecast-period-of-2022-2027-100-report-pages-2022-09-23. 3. Derived from: https://www.grandviewresearch.com/industry-analysis/ventricular-assist-devices-market ($600m estimated market in 2018 / Avg cost per procedure of $200k = 3k procedures) and growth rate from same source. 4. Derived from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6557150/. 5. Derived from: https://www.ncbi.nlm.nih.gov/pubmed/25291348. 6. Derived from: https://www.uclahealth.org/medical-services/heart/ecmo/research/statistics and growth rate from same source. ©2023 Nuwellis, Inc
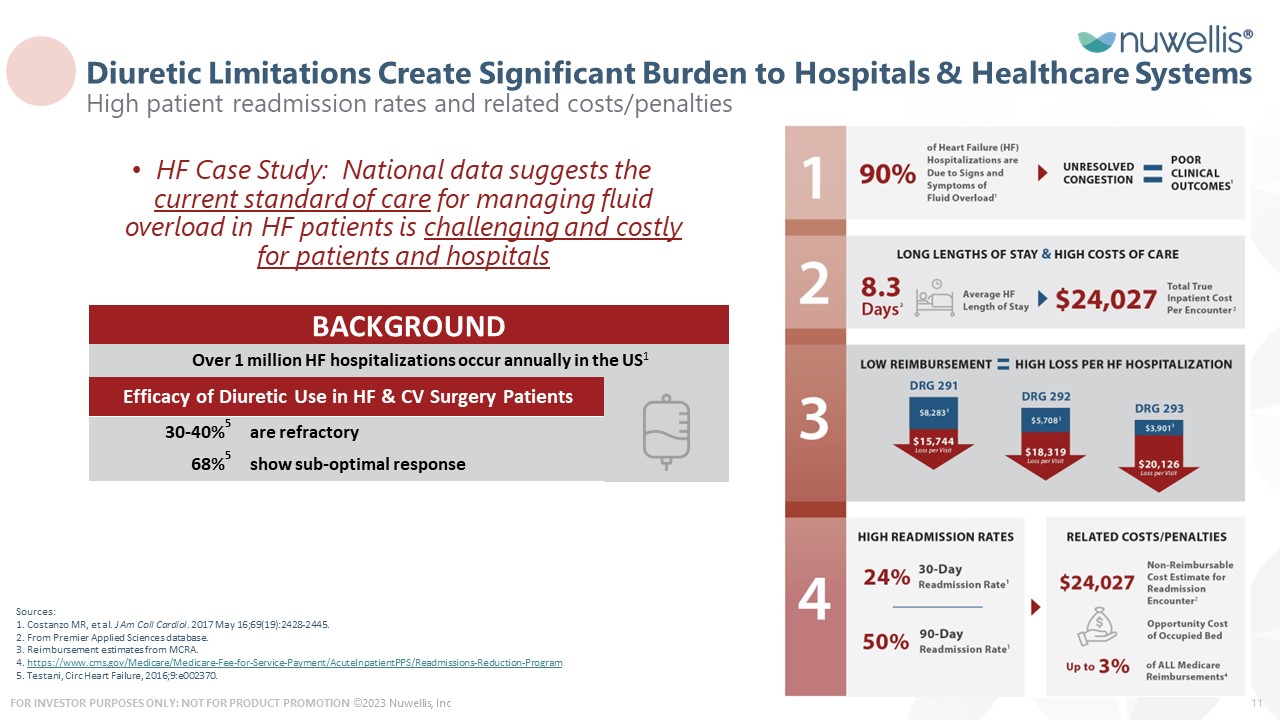
HF Case Study: National data suggests the current standard of care for managing fluid overload in HF patients is challenging and costly for patients and hospitals Sources: 1. Costanzo MR, et al. J Am Coll Cardiol. 2017 May 16;69(19):2428-2445. 2. From Premier Applied Sciences database. 3. Reimbursement estimates from MCRA. 4. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/Readmissions-Reduction-Program 5. Testani, Circ Heart Failure, 2016;9:e002370. 30-40%5 68%5 Efficacy of Diuretic Use in HF & CV Surgery Patients are refractory show sub-optimal response BACKGROUND Over 1 million HF hospitalizations occur annually in the US1 Diuretic Limitations Create Significant Burden to Hospitals & Healthcare Systems High patient readmission rates and related costs/penalties ©2023 Nuwellis, Inc

Aquapheresis Versus Intravenous Diuretics and Hospitalizations for Heart Failure (AVOID-HF) Trial Finkelstein-Schoenfeld Method of Win Ratios Analysis Provides New Insights Into Clinical Data Favors Adjustable Ultrafiltration (AUF) over Adjustable Loop Diuretics (ALD) in Treating Fluid Overloaded Heart Failure Patients *Sean Pinney1, Maria DeVita2, Maria Rosa Costanzo3 1. University of Chicago Medicine, Division of Cardiology, Chicago, IL (through February 2023). Mount Sinai Morningside, Division of Cardiology, New York, NY (March 2023 to present); 2. Lenox Hill Hospital, Division of Nephrology, New York, NY. 3. Midwest Cardiovascular Institute, Division of Cardiology, Naperville, IL. Aquadex Ultrafiltration: A Safe and More Effective Therapy Than Diuretics ©2023 Nuwellis, Inc Adjustable Ultrafiltration “won” more times than Adjustable IV Loop Diuretics, resulting in Win Ratios that all favored Ultrafiltration over diuretics in reducing CV mortality and HF events in congested HF patients

HF HOSPITALIZATIONS 10-Year, Real-World Experience with Ultrafiltration1 ABINGTON HOSPITAL JEFFERSON HEALTH Retrospective, single center analysis 334 consecutive acutely decompensated heart failure patients Cohort of patients in study were sicker than those in other clinical trials Treated with adjustable-rate UF using Aquadex Weight loss due to fluid removal Unchanged kidney function Average 2.14 hospitalizations Year before Aquadex Ultrafiltration 1 Year After Aquadex ultrafiltration Average 0.4 hospitalizations 12.4% at 30 days 14.9% at 90 days 27.3% at 1 year NATIONAL AVERAGE 24% at 30 days2 50% at 6 months HOSPITAL READMISSIONS Watson R et al. J Cardiac Fail. 2020; 26(10): s56. 2. Costanzo MR, et al. JACC. 2017 May 16;69(19):2428-2445. Significant quality of life improvement for the patients and savings to the healthcare system and to the individual hospitals Ultrafiltration: Positive ROI, Clinical and Economic Benefits 81% reduction in heart failure hospitalizations per year Newly published ©2023 Nuwellis, Inc

Outpatient Aquadex – Potential Savings for a Hospital Account With inpatient therapy only: 23% HF patient readmission rate $41.7M non reimbursable loss With outpatient Aquadex: 12% HF patient readmission rate 35% cost savings over inpatient therapy only HOSPITAL NAME Actual Hospital Account INPATIENT TREATMENT WITH DIURETICS ONLY ESTIMATED HEART FAILURE (HF) 30-DAY READMISSION COSTS Est Annual HF Admissions1 7,537 30-Day Readmission Rate1 23% Est Annual HF Readmissions 1,734 Est Cost Per HF Patient Readmission (Inpatient)2 $24,027 Est Reimbursement Per HF Readmission (Inpatient) $0 Est Loss Per HF Patient (Inpatient) $24,027 EST LOSS FOR ALL 30-DAY READMISSIONS ($41,662,818) ESTIMATED PER PATIENT REIMBURSEMENT FOR OUTPATIENT AQUADEX Outpatient Facility Payment $1,216 Venous Access Payment $1,436 Physician Payment $366 TOTAL ESTIMATED REIMBURSEMENT (PER PATIENT) $3,018 NET OUPATIENT AQUADEX COSTS (PER PATIENT) ($3,490) ESTIMATED COSTS FOR OUTPATIENT AQUADEX Total Costs (All Patients) ($11,284,433) Total Reimbursement (All Patients) $5,233,472 NET OUPATIENT AQUADEX COSTS (ALL PATIENTS) ($6,050,961) 30-DAY READMISSIONS (AQUADEX TREATED PATIENTS) % Readmission Reduction 50% Readmission Reduction (Number of Patients) 867 READMISSION REDUCTION COST ($20,831,409) 30-DAY READMISSIONS (NON-AQUADEX TREATED PATIENTS) % Readmission Reduction 0% Readmission Reduction (Number of Patients) 0 READMISSION REDUCTION SAVINGS $0 ADJUSTED REAMISSIONS AND COSTS ADJUSTED READMISSIONS (NUMBER OF PATIENTS) 867 ADJUSTED COSTS FOR READMISSIONS ($20,831,409) NET COSTS FOR ADMINISTRATION OF OUTPATIENT AQUADEX ($6,050,961) TOTAL COSTS FOR OUTPATIENT AQUADEX ($26,882,370) NET SAVINGS OVER INPATIENT THERAPY ONLY $14,780,448 % COST SAVINGS 35% ADJUSTED READMISSION RATE 12% 1. Definitive Health Database for year 2019. 2. Premier Applied Sciences database. ©2023 Nuwellis, Inc *Figures above are not guaranteed, are used for illustrative purposes only and actual results can and will vary by location
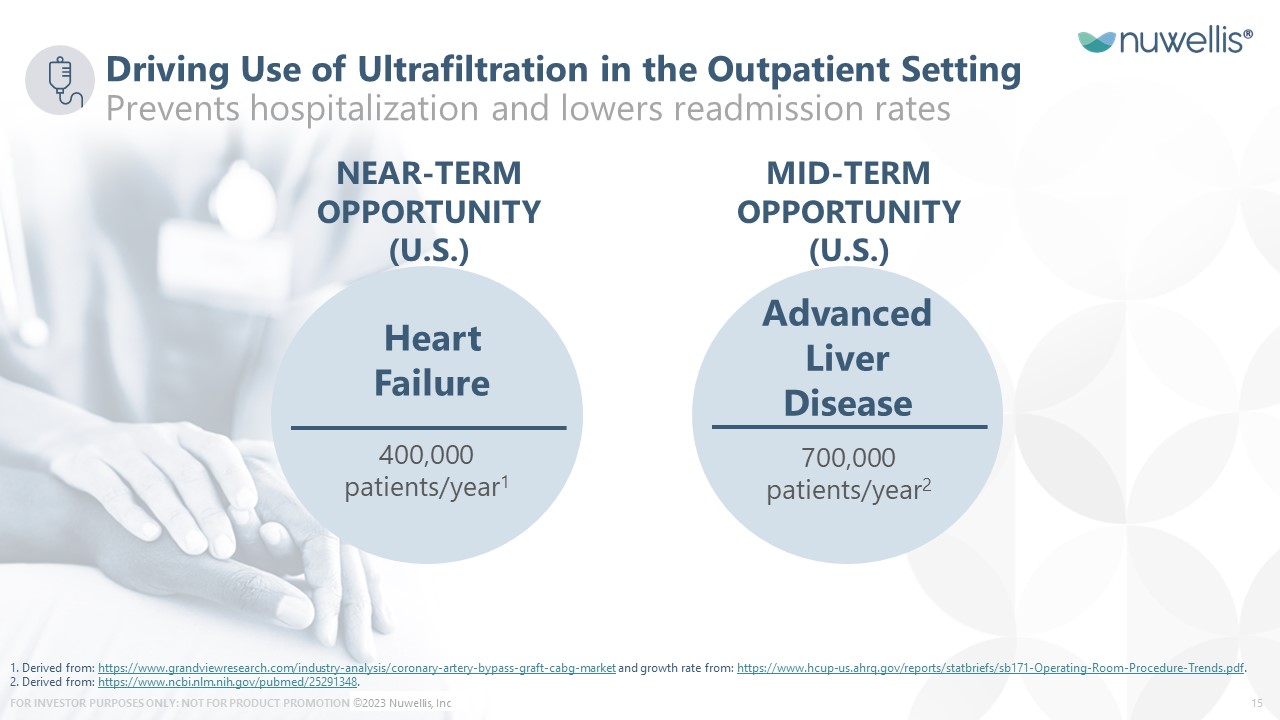
Heart Failure Advanced Liver Disease 400,000 patients/year1 700,000 patients/year2 NEAR-TERM OPPORTUNITY (U.S.) Driving Use of Ultrafiltration in the Outpatient SettingPrevents hospitalization and lowers readmission rates MID-TERM OPPORTUNITY (U.S.) 1. Derived from: https://www.grandviewresearch.com/industry-analysis/coronary-artery-bypass-graft-cabg-market and growth rate from: https://www.hcup-us.ahrq.gov/reports/statbriefs/sb171-Operating-Room-Procedure-Trends.pdf. 2. Derived from: https://www.ncbi.nlm.nih.gov/pubmed/25291348. ©2023 Nuwellis, Inc

Making Aquadex the Standard of Care in Fluid Overload Growing body of clinical evidence; Advocating for medical society guidelines and improved provider reimbursement 1. Pinney S et al Poster presented HFSA Annual Meeting 2022. 2. Haas D et al. Ten year real world experience with ultrafiltration for the management of acute decompensated heart failure. American Heart Journal, 2022. 3. Beckles DL et al. The Use of Simple Ultrafiltration Technology as a Fluid Management Strategy for High‐Risk Coronary Artery Bypass Grafting Surgery. J Cardiac Surg, 2022. DOI: 10.1111/jocs.16867 4. Sutherland SM, Davis AS, Powell D, Tanaka J, Woo M, Josephs S, Wong CJ. Kidney Replacement Therapy in Low Birth Weight Preterm Newborns. Pediatrics. 2022 Sep 1;150(3):e2022056570. doi: 10.1542/peds.2022-056570. PMID: 35945293. 5. Engelman DT, Shaw AD. A Turnkey Order Set for Prevention of Cardiac Surgery-Associated Acute Kidney Injury. 2023 Jan 1. The Annals of Thoracic Surgery. doi.org/10.1016/j.athoracsur.2022.10.022. Change practice guidelines to Ultrafiltration Reimbursement expansion into outpatient setting New clinical evidence in Heart Failure: Finkelstein-Schoenfeld Win Ratio (WR) analysis favored ultrafiltration in reducing cardiovascular mortality and heart failure rehospitalization as compared to intravenous diuretics at 30 days and 90 days1 New peer-reviewed publication: Data from ten-year, real-world experience demonstrated 81% reduction in heart failure hospitalizations per year and 48% decrease in 30-day hospital readmission rates, as well as improvements in renal function response, with ultrafiltration2 New clinical evidence in Critical Care: 100% survival at 30 days following use of ultrafiltration in high-risk postoperative coronary artery bypass grafting (CABG) patients3 Recent peer-reviewed publication of promising clinical data demonstrating 71% survival with kidney replacement therapy with ultrafiltration to treat, low weight preterm neonates with end-stage kidney disease4 2023 peer-reviewed publication of a turnkey order set for cardiac-surgery associated acute kidney injury, viewed as a template to guide clinicians in creating institution-specific, evidence-based protocols for patient care, that provides a recommendation to consider ultrafiltration if unresponsive to diuretics5 Ongoing REVERSE-HF randomized controlled trial to support driving ultrafiltration to standard of care ©2023 Nuwellis, Inc

Target Outpatient Accounts Driving Penetration and Increased Utilization Across Territories Scaling up field sales organization, education programs and social platforms Aquadex in Hospital Specialty Units ©2023 Nuwellis, Inc

Pediatric revenue has outpaced total growth over past two years 18 FOR INVESTOR PURPOSES ONLY: NOT FOR PRODUCT PROMOTION ©2021 Nuwellis, Inc. Received 510(k) and launched commerciallyin Q1 2020. Growing Pediatric Business Development of novel pediatric pipeline product on track. Growing patent portfolio supporting pediatric products. “For our babies born with diseased or absent kidneys, Aquadex has given them a chance at life because in the past, there were no options to treat these patients.” Kara Short, MSN, CRNP, NICU nurse practitioner at Alabama Children’s Hospital 3
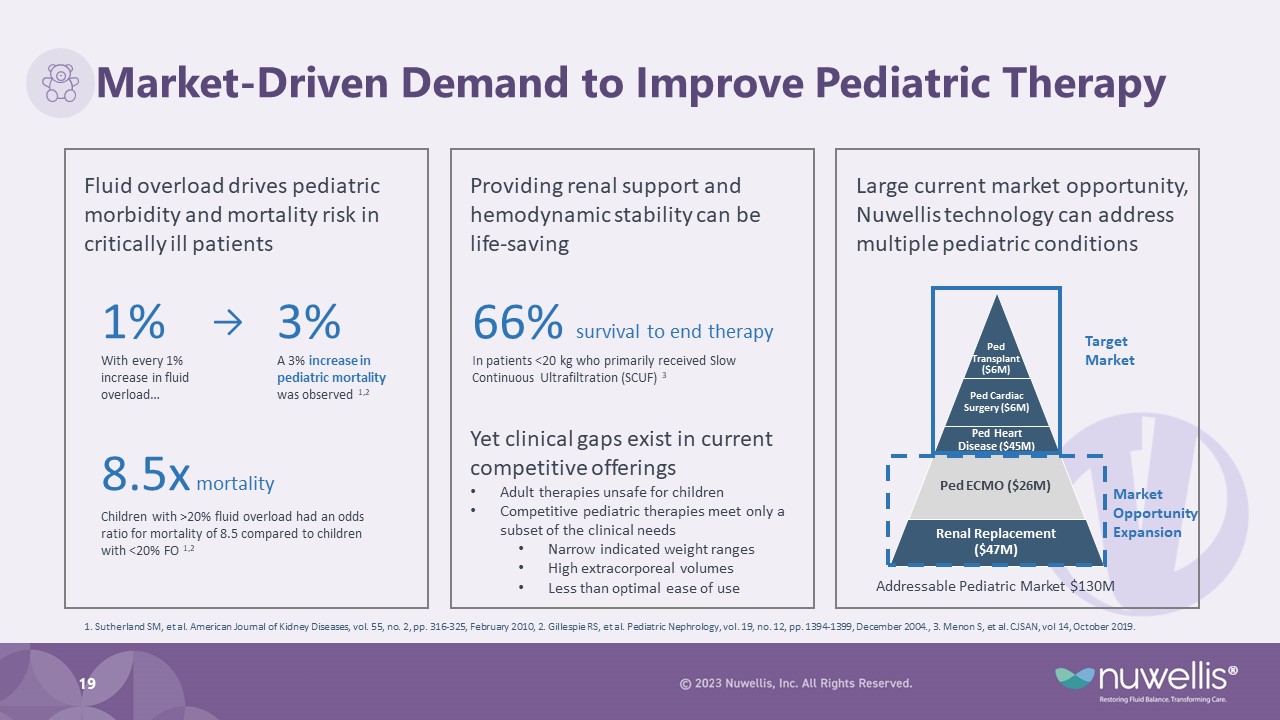
Market-Driven Demand to Improve Pediatric Therapy 19 1% With every 1% increase in fluid overload… 3% A 3% increase in pediatric mortality was observed 1,2 → Fluid overload drives pediatric morbidity and mortality risk in critically ill patients 8.5x mortality Children with >20% fluid overload had an odds ratio for mortality of 8.5 compared to children with <20% FO 1,2 Providing renal support and hemodynamic stability can be life-saving Large current market opportunity, Nuwellis technology can address multiple pediatric conditions Addressable Pediatric Market $130M 66% survival to end therapy In patients <20 kg who primarily received Slow Continuous Ultrafiltration (SCUF) 3 1. Sutherland SM, et al. American Journal of Kidney Diseases, vol. 55, no. 2, pp. 316-325, February 2010, 2. Gillespie RS, et al. Pediatric Nephrology, vol. 19, no. 12, pp. 1394-1399, December 2004., 3. Menon S, et al. CJSAN, vol 14, October 2019. Yet clinical gaps exist in current competitive offerings Adult therapies unsafe for children Competitive pediatric therapies meet only a subset of the clinical needs Narrow indicated weight ranges High extracorporeal volumes Less than optimal ease of use Target Market Market Opportunity Expansion

The Nuwellis Solution – Introducing Vivian™ 20 Business Objectives Launch best-in-class pediatric Continuous Renal Replacement Therapy (CRRT) system in 1H 2025 IDE approval anticipated in 1Q 2024 Continuously expand pediatric therapy value proposition with planned roadmap of product enhancements Product Strategy & Differentiation Deliver a solution that provides the broadest pediatric indication on the market Drive adoption and penetration with user experience (UX) design principles Uncompromising focus on patient safety Vivian System Details Integrates Ultrafiltration with Hemofiltration and Hemodialysis capabilities Broadest weight indication: 2.5 kg + Safety features: lowest extra-corporeal blood volume; built-in hematocrit sensor Clinician-driven UX design Clinical & Regulatory Plan 15 patient IDE to support 510(k) submission To be confirmed with FDA via pre-submission in May 2023 IDE submission anticipated in late 2023 Commercialization Plan Distribute with established Nuwellisfield force Grow from current pediatric customer base Offer competitive pricing PEDIATRIC
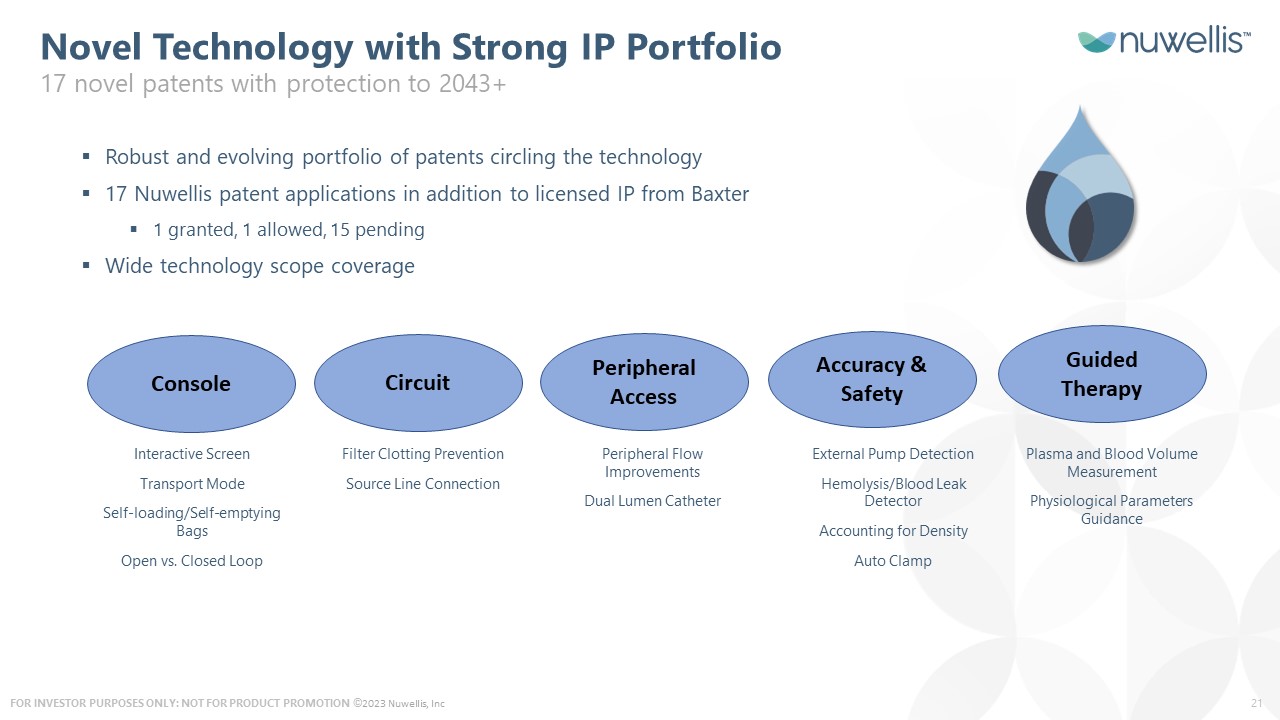
©2023 Nuwellis, Inc Novel Technology with Strong IP Portfolio 17 novel patents with protection to 2043+ Console Circuit Peripheral Access Accuracy & Safety Guided Therapy Interactive Screen Transport Mode Self-loading/Self-emptying Bags Open vs. Closed Loop Filter Clotting Prevention Source Line Connection Peripheral Flow Improvements Dual Lumen Catheter External Pump Detection Hemolysis/Blood Leak Detector Accounting for Density Auto Clamp Plasma and Blood Volume Measurement Physiological Parameters Guidance Robust and evolving portfolio of patents circling the technology 17 Nuwellis patent applications in addition to licensed IP from Baxter 1 granted, 1 allowed, 15 pending Wide technology scope coverage

2024 KEY MILESTONES 2023 2022 Q4 2022 Completed system integration and test for pediatric CRRT device Q2 2023 Enroll 50th patient in REVERSE-HF clinical trial Q4 2023 Treat 50th heart failure patient in outpatient setting Q4 2022 Acceptance by peer-reviewed journal: Ten-Year Real-World Experience with UF for the Management of ADHF Q2 2023 Penetrate 15th Premier GPO account Q3 2023 Expand IDN-based penetration strategy with 3 health systems Q3 2023 Submit claims data to support higher APC reimbursement Q2 / Q3 2023 FDA 510(k) clearance of next-generation dual-lumen extended-length catheter (dELC) Key Near-Term Milestones Legend Clinical Milestone Commercial Milestone Product Milestone Partnership/Acquisition Q1 2024 Anticipated IDE approval for Vivian™ Q2 / Q3 2023 Commercial introduction of SCD for pediatric AKI following HDE approval ©2023 Nuwellis, Inc
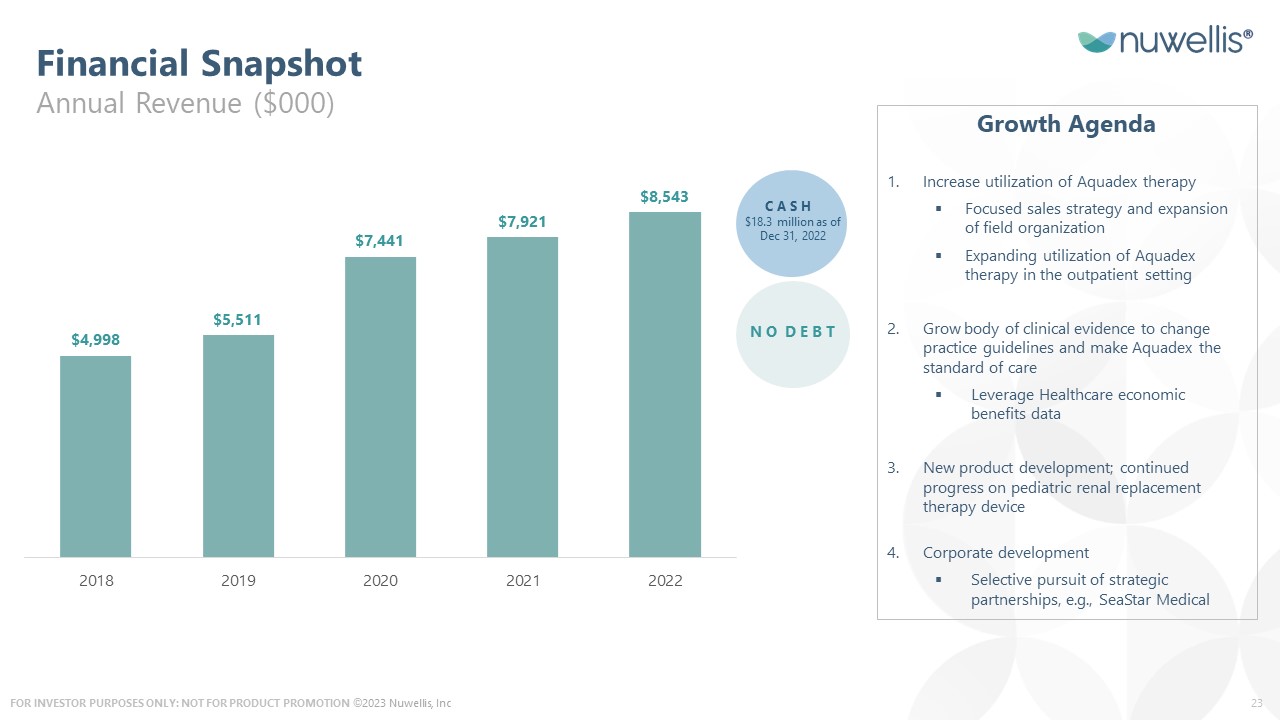
Financial Snapshot Annual Revenue ($000) Growth Agenda Increase utilization of Aquadex therapy Focused sales strategy and expansion of field organization Expanding utilization of Aquadex therapy in the outpatient setting Grow body of clinical evidence to change practice guidelines and make Aquadex the standard of care Leverage Healthcare economic benefits data New product development; continued progress on pediatric renal replacement therapy device Corporate development Selective pursuit of strategic partnerships, e.g., SeaStar Medical Cash $18.3 million as of Dec 31, 2022 NO Debt ©2023 Nuwellis, Inc

$2B+ and growing addressable market Outpatient market opportunity adds $0.5B+ to addressable market (heart failure and advanced liver disease) Positive ROI and attractive clinical + economic benefits to hospitals and healthcare system Robust body of clinical evidence Scalable consumables driven growth Commercial infrastructure leverage Novel product pipeline Experienced leadership FOR INVESTOR PURPOSES ONLY: NOT FOR PRODUCT PROMOTION ©2022 Nuwellis, Inc. Investment Highlights ©2023 Nuwellis, Inc

25 dedicated to transforming the lives of patients suffering from fluid overload through science, collaboration, and innovation ©2023 Nuwellis, Inc

MARKET SIZE SOURCES Heart Failure – Inpatient ($1B+) Incidence of HF: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5494150/ Annual HF Hospitalizations: Costanzo MR, et al. J Am Coll Cardiol. 2017 May 16;69(19):2428-2445 Insufficient diuretic response: https://www.ahajournals.org/doi/10.1161/CIRCHEARTFAILURE.115.002370?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed Heart Failure – Outpatient ($0.5B+) Incidence of HF: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5494150/ Annual HF Hospitalizations: Costanzo MR, et al. J Am Coll Cardiol. 2017 May 16;69(19):2428-2445 Diuretic resistance rate: https://www.ahajournals.org/doi/10.1161/CIRCHEARTFAILURE.115.002370?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed Critical Care ($900m) VADs: https://www.grandviewresearch.com/industry-analysis/ventricular-assist-devices-market CABG: https://www.grandviewresearch.com/industry-analysis/coronary-artery-bypass-graft-cabg-market Valves: https://idataresearch.com/over-182000-heart-valve-replacements-per-year-in-the-united-states/ Liver Transplants: https://www.healthline.com/health/liver-transplant-survival Liver Disease: https://www.ncbi.nlm.nih.gov/pubmed/25291348 Kidney Disease: https://www.kidney.org/news/newsroom/factsheets/KidneyDiseaseBasics Sepsis: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6557150/ ECMO: https://www.uclahealth.org/medical-services/heart/ecmo/research/statistics Pediatrics ($130m) Renal Replacement/AKI: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3789331/#:~:text=The%20hospitalized%20population%20at%20risk,are%20shown%20in%20Table%201 Heart Disease: https://www.cdc.gov/ncbddd/heartdefects/data.html#:~:text=Congenital%20heart%20defects%20are%20conditions,the%20United%20States%20each%20year Pediatric Transplantations: https://www.organdonor.gov/about/donors/child-infant.html Pediatric ECMO: https://www.ncbi.nlm.nih.gov/pubmed/23246046 ©2023 Nuwellis, Inc