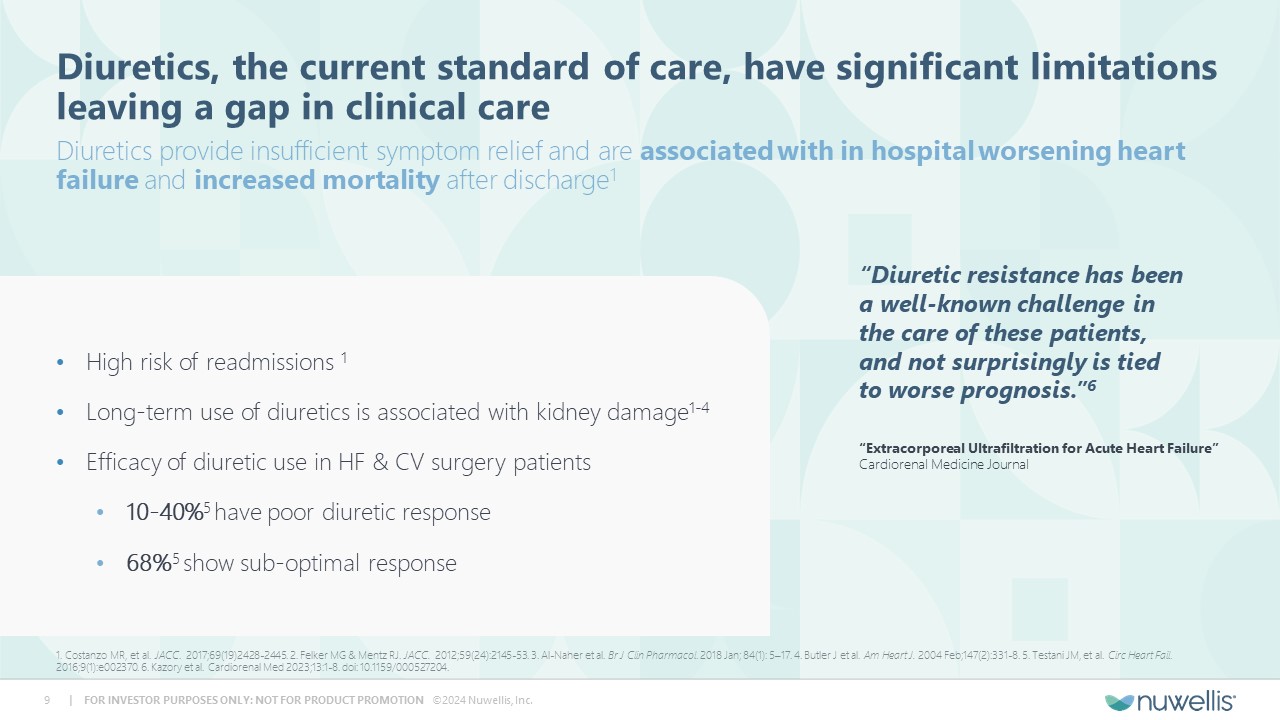
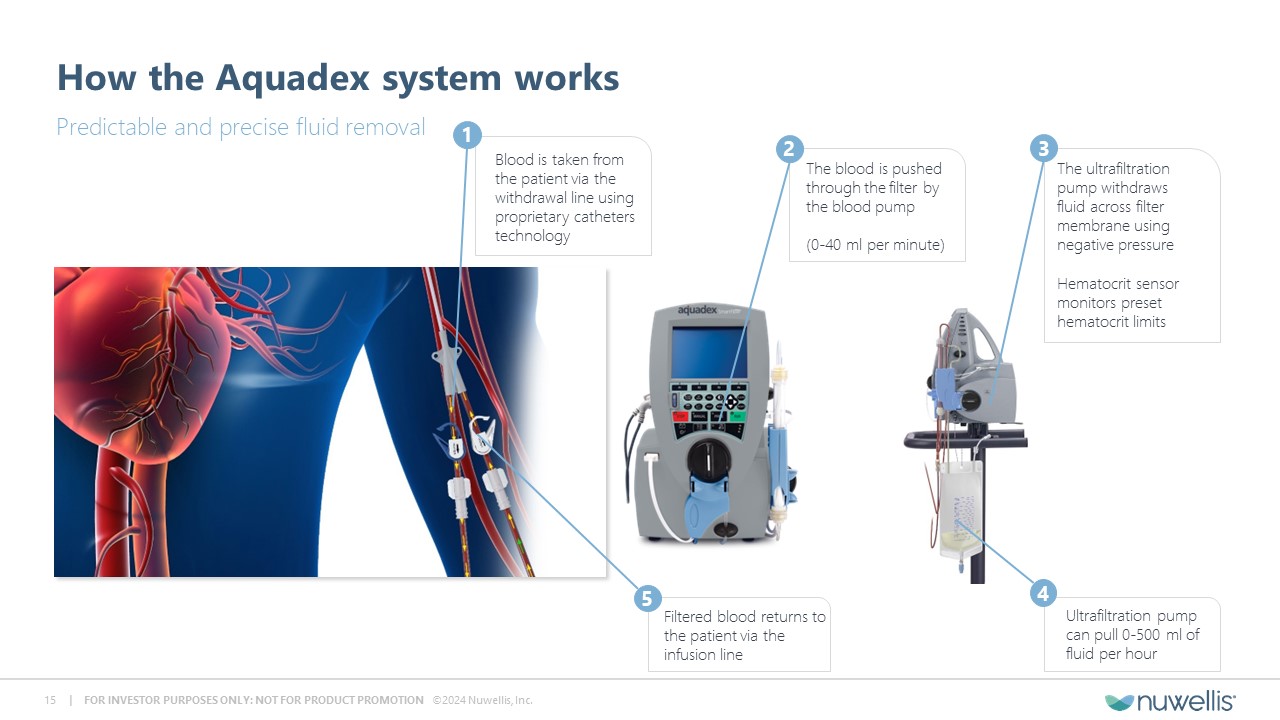
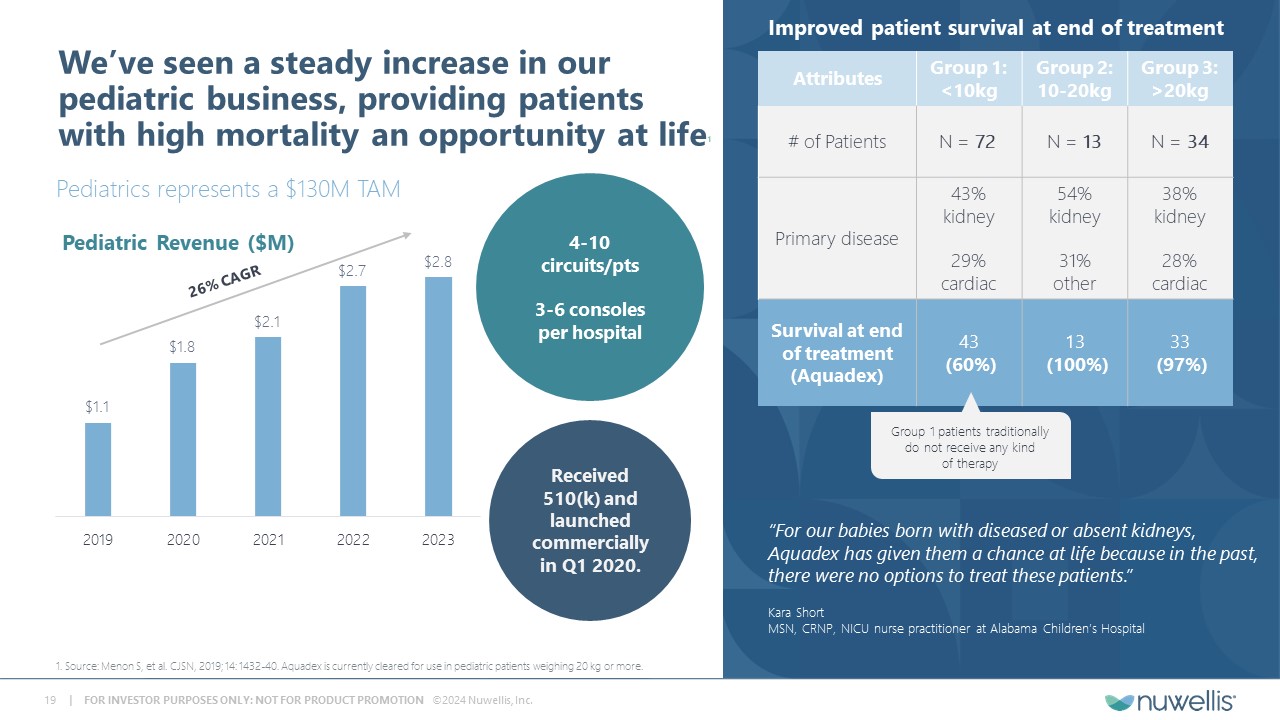
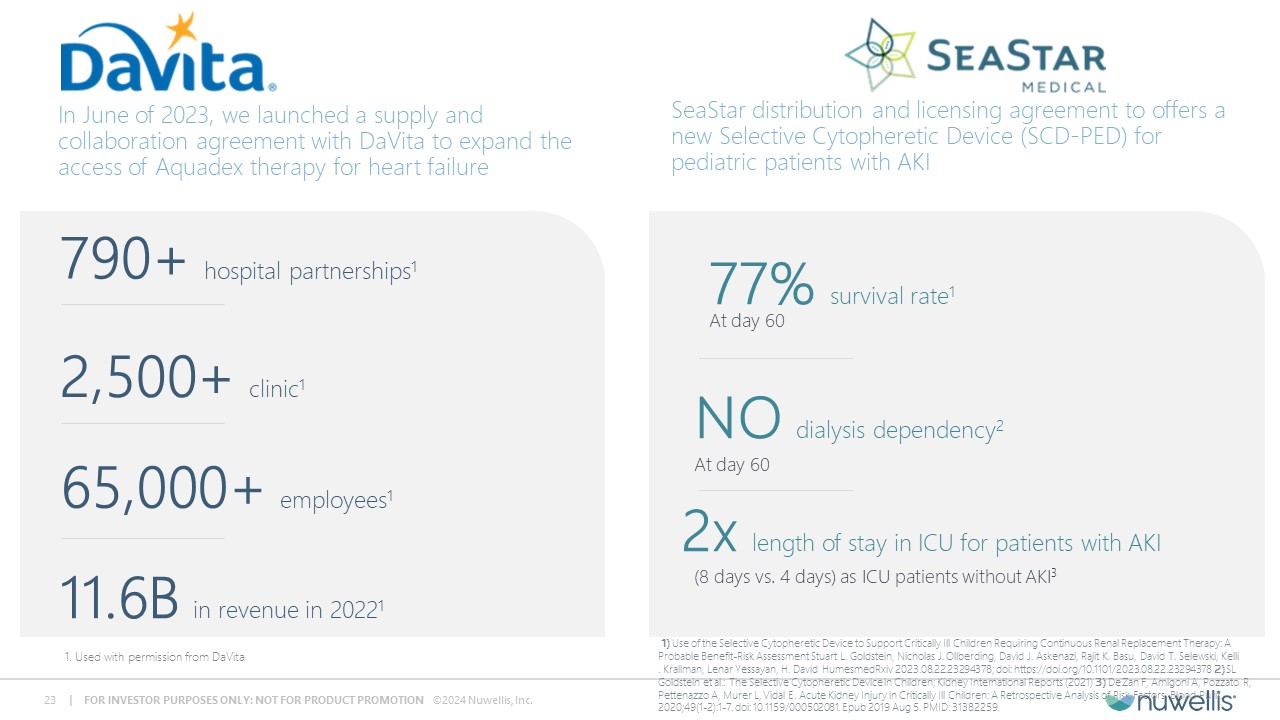
Aquadex FlexFlow® and Aquadex SmartFlow® are registered trademarks of Nuwellis, Inc. Aquadex ® is a trademark of Nuwellis, Inc. Safe Harbor Statement Financial and Statistical DataThis presentation also contains estimates and other statistical data made by independent parties and by us relating to market shares and other data about our industry. These data involve a number of assumptions and limitations and have not been reviewed or audited by our independent registered accounting firm. You are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. Neither we nor our advisors or representatives makes any representations as to the accuracy or completeness of that data or undertake to update such data after the date of this presentation. TrademarksThe trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of such products. Additional InformationYou should read the documents that we have filed with the SEC for more complete information about us. We encourage you to read such documents in full for more detailed information, statistics, reports and clinical trials referenced in this presentation. You may access these documents for free by visiting EDGAR on the SEC website at http://www.sec.gov. Forward Looking StatementThis presentation contains forward‐looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended and Section 21E of the Securities and Exchange Act, as amended regarding our plans, expectations, beliefs, estimates, goals and outlook for the future that are intended to be covered by the Private Securities Litigation Reform Act of 1995. Except for statements of historical fact, all forward‐looking statements are management’s present expectations and are not guarantees of future events and are subject to a number of known and unknown risks and uncertainties and other factors that may cause actual results to differ materially from those expressed in, or implied by, such forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “could,” “would,” “should,” “plan,” “predict,” “potential,” “project,” “promising,” “expect,” “estimate,” “anticipate,” “intend,” “goal,” “strategy,” “milestone,” and similar expressions and variations thereof. Various factors could cause actual results to differ materially from these statements including our ability to execute on our commercial strategy and to grow our Aquadex® business, the possibility that we may be unable to raise sufficient funds necessary for our anticipated operations, our clinical data collection activities, benefits of our products to patients, our expectations with respect to product development and commercialization efforts, our ability to increase market and physician acceptance of our products, potentially competitive product offerings, intellectual property protection, our expectations regarding anticipated synergies with and benefits of the Aquadex business, our business strategy, market size, potential growth opportunities and the other risks set forth under the caption “Risk Factors” and elsewhere in our periodic and other reports filed with the U.S. Securities and Exchange Commission (“SEC”), including our Annual Report on Form 10‐K for the fiscal year ended December 31, 2023 and subsequent reports. We are providing this information as of the date of this presentation, and we undertake no obligation to update any forward-looking statements contained in this presentation as a result of new information, future events or otherwise. Although the Company believes that the forward‐looking statements are reasonable and based on information currently available, it can give no assurances that the Company’s expectations are correct. All forward-looking statements are expressly qualified in their entirety by this cautionary statement.