Exhibit 99.1

www.bioniklabs.com OTCQX:BNKL LD Micro Main Event December 3, 2015 Peter Bloch, Chief Executive Officer

Legal Disclaimer This presentation contains forward - looking statements relating to future events or the future financial performance and operations of Bionik . Forward - looking statements, which involve assumptions and describe Bionik’s intent, belief or current expectations about its business opportunities, prospects, performance and results, are generally identifiable by use of the words “may,” “could,” “should,” “will,” “would,” “expect,” “anticipate,” “plan,” “potential,” “estimate,” “believe,” “intend,” “project,” “forecast,” the negative of such words and other variations on such words or similar terminology . These forward - looking statements are not guarantees of future performance and by their nature involve known and unknown risks and uncertainties that may cause actual opportunities, prospects, performance and results to vary from those presented in this document, and those variances may be material . In evaluating such statements, prospective investors should carefully consider the various risks and uncertainties identified in Bionik’s public filings, such as market risk, liquidity risk, competitive risk, regulatory risk and other commonly recognized forms of risk relating to Bionik and its securities . In light of these risks, uncertainties and assumptions, the forward - looking events discussed in this document might not occur . Further information regarding these and other risks, as well as other information about the Company, is described from time to time in the Company’s filings with the SEC, which can be accessed at www . sec . gov . Bionik is not obligated to publicly update or revise any forward - looking statements, whether as a result of new information, future events or otherwise . 2

Bionik Overview • Pioneering medical device and robotics company with a focus in developing transformational technologies and solutions for individuals with neurological disorders • Core product ARKE™: Lower body exoskeleton initially targeting rehabilitation market • Potential Canadian and CE filings in 2H16 • Expected product launch in mid 2017 • Future expansion into broader use markets • ARKE has potential to address multi billion dollar mobility rehabilitation market • Growth strategy focused on developing earlier stage research programs and acquiring/licensing synergistic products and technologies to fuel product pipeline • Experienced Board and Management Team leading execution 3

ARKE A robotic lower body exoskeleton designed to allow paraplegics as well as other wheelchair users, the ability to rehabilitate through walking TM

ARKE: Product Details • Sensors throughout the device sense body movement and trigger motion • Constructed with carbon fiber, aluminum and steel • Fully customizable to fit patients of all heights and weights • Connects to Bionik’s cloud software where data is analyzed and displayed back to the physiotherapist in real - time • All parameters adjustable through tablet interface very easily – learning curve for therapists is very low • Addresses significant unmet need in rehabilitation and mobility as well as many secondary illness potential reduction such as bone density loss, pressure sores, and urinary tract infections 5
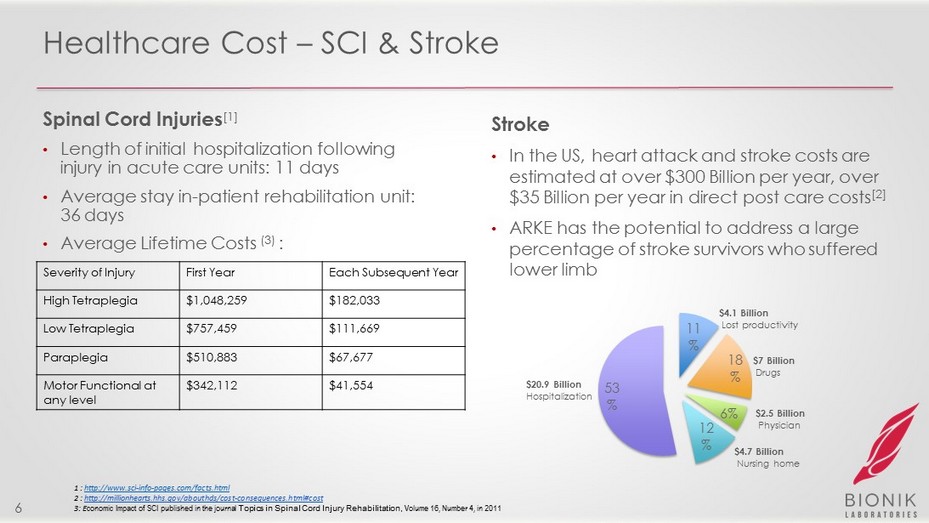
Healthcare Cost – SCI & Stroke Spinal Cord Injuries [1] • Length of initial hospitalization following injury in acute care units: 11 days • Average stay in - patient rehabilitation unit: 36 days • Average Lifetime Costs ( 3 ) : Stroke • In the US, h eart attack and stroke costs are estimated at over $300 Billion per year, over $35 Billion per year in direct post care costs [2] • ARKE has the potential to address a large percentage of stroke survivors who suffered lower limb 11 % 18 % 6% 12 % 53 % $20.9 Billion Hospitalization $4.7 Billion Nursing home $7 Billion Drugs $2.5 Billion Physician $4.1 Billion Lost productivity 1 : http://www.sci - info - pages.com/ facts.html 2 : http://millionhearts.hhs.gov/abouthds/cost - consequences.html# cost 3: E conomic Impact of SCI published in the journal Topics in Spinal Cord Injury Rehabilitation, Volume 16, Number 4, in 2011 6 Severity of Injury First Year Each Subsequent Year High Tetraplegia $1,048,259 $182,033 Low Tetraplegia $757,459 $111,669 Paraplegia $510,883 $67,677 Motor Functional at any level $342,112 $41,554

Sizing the Market • There are approximately 276,000 people living with SCI in the USA, with an estimated annual 12,500 new cases. (1) Across the G20 this translates to an estimated 4,000,000 existing SCI and annual new 180,000 cases. • Of the global, annual, 16,000,000 stroke cases, 5,000,000 people each year end up in permanent disability, (2) and 75% of stroke survivors are disabled enough to impact their employability. (3) • Mild TBI occurs in about 100 - 600 people per 100,000, (4) or about 25,000,000 per annum. Between 30 - 65% of TBI survivors will have mobility impairment (5) and require gait rehabilitation. • ARKE Generation 3 is being designed to address Stroke & TBI patient populations, therefore, expanding the addressable population by an additional 20,000,000 annual patients . 7 (1.) The National SCI Statistical Center, Birmingham, AL ( http://www.nscisc.uab.edu/ ) (2.) http :// www.strokecenter.org /patients/about - stroke/stroke - statistics/ (3.) Coffey C. Edward, Cummings Jeffrey L, Starkstein Sergio, Robinson Robert (2000). Stroke - the American Psychiatric Press Textbook of Geriatric Neuropsychiatry (Second ed.). Washington DC: American Psychiatric Press. pp. 601 – 617. (4. ) Park E, Bell JD, Baker AJ (April 2008). "Traumatic brain injury: Can the consequences be stopped?" . Canadian Medical Association Journal 178 (9): 1163 – 70. (5. ) http:// www.msktc.org / tbi /factsheets/Balance - Problems - After - Traumatic - Brain - Injury Annual USD (MM) USA Canada EU G20 SCI $100 + $10+ $160+ $1,400+ Stroke & TBI $5,000+ $500+ $8,000+ $35,000+ Estimated ARKE Rehabilitation Market Size (millions):
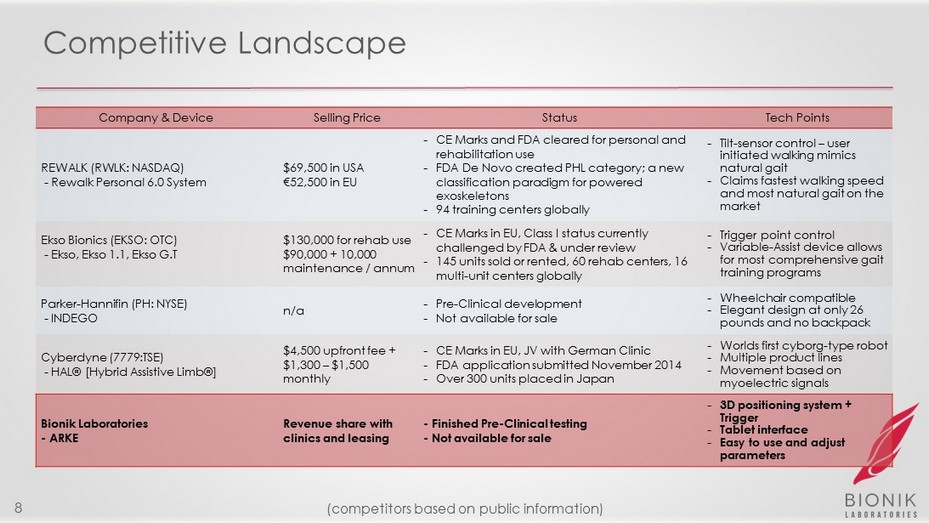
Competitive Landscape Company & Device Selling Price Status Tech Points REWALK (RWLK: NASDAQ) - Rewalk Personal 6.0 System $ 69,500 in USA € 52,500 in EU - CE Marks and FDA cleared for personal and rehabilitation use - FDA De Novo created PHL category; a new classification paradigm for powered exoskeletons - 94 training centers globally - Tilt - sensor control – user initiated walking mimics natural gait - Claims fastest walking speed and most natural gait on the market Ekso Bionics (EKSO: OTC) - Ekso , Ekso 1.1, Ekso G.T $130,000 for rehab use $90,000 + 10,000 maintenance / annum - CE Marks in EU, Class I status currently challenged by FDA & under review - 145 units sold or rented, 60 rehab centers, 16 multi - unit centers globally - Trigger point control - Variable - Assist device allows for most comprehensive gait training programs Parker - Hannifin (PH: NYSE) - INDEGO n/a - Pre - Clinical development - Not available for sale - Wheelchair compatible - Elegant design at only 26 pounds and no backpack Cyberdyne (7779:TSE) - HAL® [Hybrid Assistive Limb®] $4,500 upfront fee + $1,300 – $1,500 monthly - CE Marks in EU, JV with German Clinic - FDA application submitted November 2014 - Over 300 units placed in Japan - Worlds first cyborg - type robot - Multiple product lines - Movement based on myoelectric signals Bionik Laboratories - ARKE Revenue share with clinics and leasing - Finished Pre - Clinical testing - Not available for sale - 3D positioning system + Trigger - Tablet interface - Easy to use and adjust parameters 8 (competitors based on public information)

Business Model 9 In - Patient Rehab • Lease/Rental model • Monthly fee to the hospital/clinic for use of the product Out - Patient Rehab • Revenue share or Lease/Rental model • Percentage of the per session therapy to Bionik Home Use • Rental or direct product sale • ARKE revenue can be generated by a product sale, product rental/lease, and revenue share with partner clinics for treatments • Rental /lease is optimal for highest - use / early recovery areas, such as specialized SCI in - patient facilities • Revenue - share is best suited for out - patient rehabilitation programs, once a financing plan and treatment program has been indicated for the patient • Product sale presents the most challenge, as high upfront cost must overcome lengthy capital expenditure cycles

Reimbursement • No universal coverage; however successful reimbursement on a case - by - case has been achieved • Etiology of injury impacts financing plan – e.g. vehicle accidents (automobile liability and Personal Injury Protection); workplace (workman’s compensation); fall at home (home insurance); etc. • Case Managers & Social Workers work with patients to prepare a comprehensive financing plan from multiple sources • Industry - wide efforts, all competitors, academics, etc., drive the case for universal reimbursement (indicated by granting of HCPCS Code), whenever they demonstrate beneficial outcomes in published studies. • Successful cases include: broad coverage at Bronx - VA, top 3 private insurers in Germany, & German Statutory Accident Insurance (DGUV) • E.g. DGUV offers reimbursement of EUR 500 / session for up to 61 sessions (1) • Applicable Center for Medicare & Medicaid Services HCPCS codes currently include: Therapeutic Exercises, Neuromuscular re - education, Therapeutic activities, Gait training, Physical performance tests, assistive technology assessments, orthotic management/training and prosthetic management training. Average Reimbursement of $34.03 / 15 minute interval (2) 10 (1.) http:// www.cyberdyne.jp / english /services/ HALTherapy.html (2.) Centers for Medicare and Medicaid Services
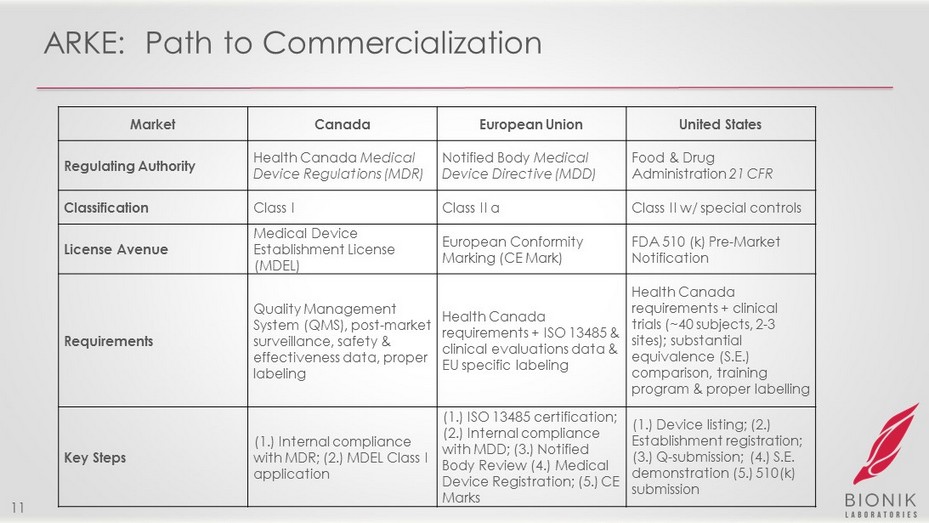
ARKE: Path to Commercialization 11 Market Canada European Union United States Regulating Authority Health Canada Medical Device Regulations (MDR) Notified Body Medical Device Directive (MDD) Food & Drug Administration 21 CFR Classification Class I Class II a Class II w/ special controls License Avenue Medical Device Establishment License (MDEL) European Conformity Marking (CE Mark) FDA 510 (k) Pre - Market Notification Requirements Quality Management System (QMS), post - market surveillance, safety & effectiveness data, proper labeling Health Canada requirements + ISO 13485 & clinical evaluations data & EU specific labeling Health Canada requirements + clinical trials (~40 subjects, 2 - 3 sites); substantial equivalence (S.E.) comparison, training program & proper labelling Key Steps (1.) Internal compliance with MDR; (2.) MDEL Class I application (1.) ISO 13485 certification; (2.) Internal compliance with MDD; (3.) Notified Body Review (4.) Medical Device Registration; (5.) CE Marks (1.) Device listing; (2.) Establishment registration; (3.) Q - submission; (4.) S.E. demonstration (5.) 510(k) submission

Pipeline Expansion Opportunities Pipeline Products Discovery Prototyping Optimization Pre - clinical Clinical Approval ARKE TM PARAPLEGIC REHABILITATION Pipeline Expansion Opportunities Research Products Discovery Prototyping Optimization Pre - clinical Clinical Approval BNKL - 002 ARKE TM STROKE BNKL - 003 ARKE TM SUPPORT APOLLO TM CHRONOS TM 12 An exoskeleton technology specific for stroke rehabilitation Potential future application specific for elderly support Intelligent above knee prosthetic with ultimate natural fluidity and walking gait Cloud based intelligent patient queuing system with predictive technology
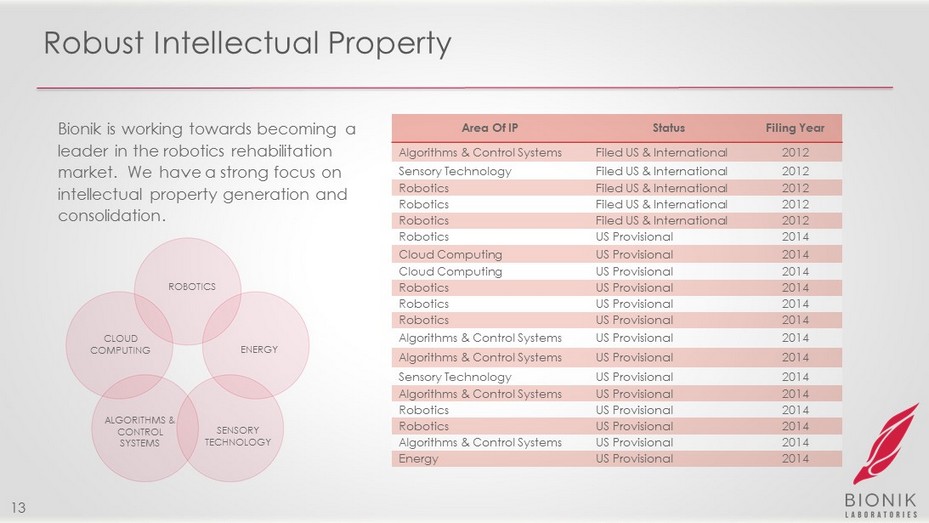
Robust Intellectual Property Area Of IP Status Filing Year Algorithms & Control Systems Filed US & International 2012 Sensory Technology Filed US & International 2012 Robotics Filed US & International 2012 Robotics Filed US & International 2012 Robotics Filed US & International 2012 Robotics US Provisional 2014 Cloud Computing US Provisional 2014 Cloud Computing US Provisional 2014 Robotics US Provisional 2014 Robotics US Provisional 2014 Robotics US Provisional 2014 Algorithms & Control Systems US Provisional 2014 Algorithms & Control Systems US Provisional 2014 Sensory Technology US Provisional 2014 Algorithms & Control Systems US Provisional 2014 Robotics US Provisional 2014 Robotics US Provisional 2014 Algorithms & Control Systems US Provisional 2014 Energy US Provisional 2014 ROBOTICS SENSORY TECHNOLOGY ALGORITHMS & CONTROL SYSTEMS ENERGY CLOUD COMPUTING 13 Bionik is working towards becoming a leader in the robotics rehabilitation market. We have a strong focus on intellectual property generation and consolidation.
Growth Strategy Supports Further Pipeline Expansion • Leveraging internal expertise and broad network to develop new synergistic technologies • Team dedicated to expanding product pipeline through strategic partnering and acquisitions with products and enabling software 14 Expanded and Focused Development Programs In - house R&D Licensing / Acquisitions Industry Partners University Networks

Management Team • Peter Bloch, CPA, CA – Chief Executive Officer • Over 25 years of executive management experience with proven track record of building public and private technology companies. Former CFO and joint Interim CEO of Sanofi Canada, CFO of Intellivax , Gennum TSX:GNM (n/k/a Semtech NASDAQ:SMTC), Just Energy NYSE/TSX:JE and Founder of Tribute Pharmaceuticals • Michal Prywata – Co - Founder and Chief Operating Officer • Biomedical engineering experience with a track record of winning technology showcases and developing technologies that address significant and untapped markets • Thiago Caires – Co - Founder and Chief Technical Officer • Biomedical engineering experience with a track record of winning technology showcases and developing technologies that address significant and untapped markets • Leslie N. Markow , CPA, CA, CPA(Illinois) C.Dir – Chief Financial Officer • Over 25 years of finance/accounting leadership experience. Former CFO of SunOpta (NASDAQ: STKL) 15

Board and Advisors Strengthens Capabilities Advisors • Gary Henley • Significant medtech experience • Former CEO of United Orthopedic Group, Wright Medical Technology, CeCorp and Electronic Systems • Board Director of numerous companies • Dr. Isador Lieberman, MD MBA FRCSC • Medical Director, Clinical Advisor; Texas Back Institute Orthopedic and Spinal Surgeon • Dr. Edward Lemaire • Clinical Trial Advisor & Investigator; Clinical Investigator, Centre for Rehabilitation Research, Clinical Epidemiology, Ottawa Hospital Research Institute • Dr. Wendy Cukier • Grant Advisor; Vice - President of Research and Innovation, Ryerson University • Dr. Kaamran Raahemifar • Control Systems & Electronics Advisor; PhD in Biomedical Engineering; Electrical & Computer Eng. professor at Ryerson University • Dr. Dany Gagnon • Clinical Trial Advisor & Investigator; Clinical Investigator : Centre de Recherche Interdisciplinaire en readaptation du Grand Montreal Board of Directors • Dr. Robert Hariri • Independent Director • Chairman, Founder, Chief Scientific Officer, and former Chief Executive Officer of Celgene Cellular Therapeutics; Board member of Myos Corporation and Provista Diagnostics • Marc Mathieu • Independent Director • Chief Marketing Officer of Samsung North America; Former Senior Vice President of Global Marketing at Unilever; Chairman and Co - founder of We&Co • Peter Bloch, CPA, CA • Chairman and Chief Executive Officer • Michal Prywata • Co - Founder and Chief Operating Officer • Thiago Caires • Co - Founder and Chief Technical Officer 16

Financial Snapshot - OTCQX: BNKL Stock Ticker Symbol OTCQX: BNKL Market Capitalization as of November 30 , 2015 $87.6 M Shares Outstanding 72.4 M** Warrants at exercise price of $1.40 16.4 M Warrants at exercise price of $0.80 1.64 M R ecent financings: February - July , 2015 $13.1 M Cash as of September 30 , 2015 $7.8 M Amount received in grants to date $2.5 M Based on management’s current projections, cash position is sufficient to fund operations through initial commercialization of ARKE* *Additional funding would be required to execute growth and expansion strategy ** Includes 50 million Exchangeable Shares which are exchangeable into common stock on a 1 for 1 basis 17

Near - term Milestones 201 5 H2 201 6 H1 201 6 H2 201 7 H1 Pre - clinical v erification testing gen 2 - ARKE Clinical evaluations - ARKE First clinical data readout - ARKE Targeted Health Canada MDEL & CE Mark Filings - ARKE Expected Health Canada license & CE Marks Registration - ARKE Formal Clinical Trials for FDA Substantial Equivalence Expansion of Board Seeking up - list to a national exchange Expansion of intellectual property Expansion of product portfolio through acquisitions/licensing 18

www.bioniklabs.com OTCQX:BNKL LD Micro Main Event December 3, 2015 Peter Bloch, Chief Executive Officer