OMB APPROVAL | |
| OMB Number: | 3235-0258 |
| Expires: | December 31, 2017 |
| Estimated average burden hours per response: | 1802.12 |
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM F-1
ABATTIS BIOCEUTICALS CORP.
(Exact name of registrant as specified in its charter)
| British Columbia | 2833 | |||
| (State or other jurisdiction of incorporation or organization) | (Primary Standard Industrial Classification Code Number) | (I.R.S. Employer Identification Number) |
1040 - 885 West Georgia Street, Vancouver, British Columbia, V6C 3H1
604-336-0881
(Address, including zip code, and telephone number,
including area code, of Registrant's principal executive office)
William Fleming
1040 - 885 West Georgia Street, Vancouver, British Columbia, V6C 3H1
604-336-0881
(Name, address, including zip code, and telephone number,
including area code, of agent for service)
Approximate date of commencement of proposed sale to the public: As soon as practical after the effective date of this registration statement.
If any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check the following box: o
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. o
1
Calculation of Registration Fee
Title of Each Class of Securities to be Registered | Amount to be Registered(1) | Proposed Maximum Offering Price Per Common Share(1) | Proposed Maximum Aggregate Offering Price(2) | Amount of Registration Fee(3) | |
| Common Shares | 180,000,000 Common Shares | $0.1388 | $25,000,000 | $3,001.18 | |
Notes:
| (1) | We are registering 180,000,000 shares of our common stock issued to Dutchess Opportunity Fund, II, LP ("Dutchess" or the "Selling Security Holder") pursuant to an Investment Agreement dated January 28, 2015 (the "Investment Agreement"). | |
| (2) | In the event of stock splits, stock dividends, or similar transactions involving the registrant's common stock, the number of shares of common stock registered shall, unless otherwise expressly provided, automatically be deemed to cover the additional securities to be offered or issued pursuant to Rule 416 promulgated under the Securities Act of 1933, as amended (the "Securities Act"). In the event that adjustment provisions of the Investment Agreement require the registrant to issue more shares than are being registered in this registration statement, for reasons other than those stated in Rule 416 of the Securities Act, the registrant will file a new registration statement to register those additional shares | |
| (3) | The registration fee is calculated in accordance with Rule 457(i) of the Securities Act, based upon the conversion price set forth in the Investment Agreement when using the highest trading price of the Company's common stock in the last thirty (30) calendar days. |
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with section 8(a) of the Securities Act of 1933 or until this registration statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said section 8(a), may determine.
2
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the U.S. Securities and Exchange Commission is effective. This prospectus is not an offer to sell these securities and is not soliciting an offer to buy these securities in any state where the offer or sale is not permitted.
PRELIMINARY PROSPECTUS
SUBJECT TO COMPLETION, DATED April 27, 2015
ABATTIS BIOCEUTICALS CORP.
Distribution of
180,000,000 Shares of Common Stock
The selling stockholders identified in this prospectus may offer and sell up to 180,000,000 shares of our common stock (the "Shares"), which will consist of 180,000,000 shares of common stock issued to Dutchess Opportunity Fund, II, LP ("Dutchess" or the "Selling Security Holder") pursuant to an Investment Agreement dated January 28, 2015 (the "Investment Agreement").
The selling stockholders may sell all or a portion of the shares being offered pursuant to this prospectus at fixed prices, at prevailing market prices at the time of sale, at varying prices or at negotiated prices.
The Investment Agreement with Dutchess provides that, for a period of thirty-six (36) months commencing on the effective date of the registration statement, Dutchess is committed to purchase up to $25,000,000 of our common stock. We may draw on the facility from time to time, as and when we determine appropriate in accordance with the terms and conditions of the Investment Agreement. The 180,000,000 Shares included in this prospectus represent the Shares issuable to the Selling Security Holder under the Investment Agreement.
Dutchess is an "underwriter" within the meaning of the Securities Act of 1933 (the "Securities Act") in connection with the resale of our common stock under the Investment Agreement. No other underwriter or person has been engaged to facilitate the sale of shares of our common stock in this offering. Dutchess will pay us ninety-five percent (95%) of the volume weighted average price of the Company's common stock for the five (5) consecutive trading days after the Company delivers to Dutchess an advance notice in writing requiring Dhess to advance funds (an "Advance") to the Company, subject to the terms of the Investment Agreement.
We will not receive any proceeds from the sale of these Shares offered by the Selling Security Holder. However, we will receive proceeds from the sale of our Shares under the Investment Agreement. The proceeds will be used for working capital or general corporate purposes. We will bear all costs associated with this registration.
We are currently listed on the OTCQX. Our common stock is quoted on the OTCQX under the symbol "ATTBF". The Shares registered hereunder are being offered for sale by the Selling Security Holder at prices established on the OTCQX during the term of this offering. On April 24, 2015, the closing price as reported on the OTCQX was $0.101 per share. This price will fluctuate based on the demand for our common stock.
We are also listed on the Canadian Securities Exchange (the "CSE") under the trading symbol "ATT". On April 24, 2015, the closing price as reported on the CSE was CDN $0.12 per share. This price will fluctuate based on the demand for our common stock.
3
The Company qualifies as an "emerging growth company" as defined in the Jumpstart Our Business Startups Act, which became law in April 2012 and will be subject to reduced public company reporting requirements. See "The Company: Jumpstart Our Business Startups Act" contained herein.
The information in this prospectus is not complete and may be changed. We may not sell these securities until the registration statement filed with the U.S. Securities and Exchange Commission (the "SEC") is effective. This prospectus is not an offer to sell these securities and it is not soliciting an offer to buy these securities in any state where the offer or sale is not permitted.
We are an "emerging growth company" as defined under the federal securities laws and, as such, may elect to comply with certain reduced public company reporting requirements for future filings.
This investment involves a high degree of risk. You should purchase shares only if you can afford a complete loss. See "Risk Factors" beginning on page 13.
Neither the U.S. Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is April 27, 2015.
4
TABLE OF CONTENTS
| 7 | |
| 7 | |
| 8 | |
| 13 | |
| 25 | |
| 26 | |
| 26 | |
| 27 | |
| 28 | |
| 28 | |
| 29 | |
| 30 | |
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATION | 32 |
| 43 | |
| 63 | |
| 63 | |
| 67 | |
| 75 | |
| 77 | |
| 79 | |
| 80 | |
| 81 | |
| 84 | |
| 88 | |
| 89 | |
| 89 | |
| 89 | |
| 98 | |
| 98 | |
DISCLOSURE OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES | 99 |
| 99 | |
| 99 |
5
| 100 | |
| 211 | |
| 211 | |
| 211 | |
| 213 | |
| 219 | |
| 220 | |
| 222 |
6
DEALER AND PROSPECTUS DELIVERY OBLIGATIONS
Until 90 days after the effective date of this prospectus, all dealers that effect transactions in these securities, whether participating in this offering, may be required to deliver a prospectus. This is in addition to the dealers' obligation to deliver a prospectus when acting as underwriters with respect to their unsold allotments or subscriptions.
All dollar amounts in this prospectus are expressed in Canadian dollars unless otherwise indicated. The Company's accounts are maintained in Canadian dollars and the Company's financial statements are prepared in accordance with International Financial Reporting Standards as issued by the International Accounting Standards Board. All reference to "U.S. dollars", "USD", or to "US$" are to United States dollars.
The following table sets forth the rate of exchange for the Canadian dollar, expressed in United States dollars in effect at the end of the periods indicated, the average of exchange rates in effect during such periods, and the high and low exchange rates during such periods based on the noon rate of exchange as reported by the Bank of Canada for conversion of Canadian dollars into United States dollars.
| Years Ended September 30, | |||||
| Canadian Dollars to U.S. Dollars | 2014 | 2013 | 2012 | 2011 | 2010 |
| Rate at end of period | High - 0.8973 Low - 0.8922 | High - 0.9732 Low - 0.9702 | High - 1.0200 Low - 1.0150 | High - 0.9641 Low - 0.9540 | High - 0.9775 Low - 0.9684 |
| Average rate for period | 0.9233 | 1.0299 | 0.9996 | 0.9891 | 1.0407 |
| High for period | 0.9724 | 1.0272 | 1.0371 | 1.0630 | 1.0845 |
| Low for period | 0.8888 | 0.9426 | 0.9383 | 0.9540 | 0.9961 |
FORWARD-LOOKING STATEMENTS
This prospectus contains forward-looking statements that involve risks and uncertainties. We use words such as "project", "believe", "anticipate", "plan", "expect", "estimate", "intend", "should", "would", "could", or "may", or other such words, verbs in the future tense and words and phrases that convey similar meaning and uncertainty of future events or outcomes to identify these forward-looking statements. There are a number of important factors beyond our control that could cause actual results to differ materially from the results anticipated by these forward-looking statements. While we make these forward-looking statements based on various factors and using numerous assumptions, you have no assurance the factors and assumptions will prove to be materially accurate when the events they anticipate actually occur in the future.
The forward-looking statements are based upon our beliefs and assumptions using information available at the time we make these statements. We caution you not to place undue reliance on our forward-looking statements as (i) these statements are neither predictions nor guaranties of future events or circumstances, and (ii) the assumptions, beliefs, expectations, forecasts and projections about future events may differ materially from actual results. We undertake no obligation to publicly update any forward-looking statement to reflect developments occurring after the date of this prospectus.
7
This summary highlights selected information contained elsewhere in this prospectus. This summary does not contain all the information that you should consider before investing in the common stock of the Company. You should carefully read the entire prospectus, including "Risk Factors", "Management's Discussion and Analysis of Financial Condition and Results of Operations" and the Financial Statements, before making an investment decision. In this prospectus, the terms "Company," "we," "us" and "our" refer to Abattis Bioceuticals Corp.
Corporate Overview
Abattis Bioceuticals Corp. ("Abattis" or the "Company"), was incorporated in British Columbia on June 30, 1997 under the name "Sinocan Capital Group Inc." The Company changed its name to Sican Ventures Inc. on September 29, 1997, to Abattis Biologix Corporation on September 14, 2009 and to Abattis Bioceuticals Corp. on September 5, 2012. As of April 27, 2015, there were 69,667,951 shares of common stock outstanding.
Our principal executive offices are located at 1040 - 885 West Georgia Street, Vancouver, British Columbia, V6C 3H1. Our telephone number is (604) 336-0881. Our internet address is www.abattis.com. Information on our website does not constitute part of this prospectus.
We are a reporting issuer in the Canadian provinces of British Columbia, Alberta, and Ontario. Our common shares are listed on the Canadian Securities Exchange under the symbol "ATT" and are quoted under the OTCQX under the symbol "ATTBF".
Our Current Business
Overview
Abattis is a specialty biotechnology company who intends with capabilities through its wholly owned subsidiaries of cultivating, licensing, and marketing proprietary ingredients, bio-similar compounds, patented equipment and consulting services to medicinal markets in North America. Management believes the Company is positioned to capitalize on the fast growing trend toward marijuana legalization in the United States and for medicinal use in Canada and international jurisdictions by supplying and collaborating with companies to employ its vertical cultivation systems, extraction equipment/technology, and strategic marketing support to licensed growers. The Company also has an extensive pipeline of high-quality products and intellectual property for the rapidly expanding botanical drug market. We follow strict Standard Operating Protocols and adhere to the laws of Canada and Foreign Jurisdictions. As of the date of this prospectus, we have not commenced the actual production and sale of medical marijuana but are seeking to lay the foundation to commence this business.
Canadian Regulatory Changes Regarding Medical Marijuana
Until recently, the medical use of marijuana in Canada was governed by the Marihuana Medical Access Regulations (or MMAR), a regulation to the federal Controlled Drugs and Substances Act (or CDSA), which allowed qualified patients to access medical marijuana by growing their own marijuana, designating a third party to grow marijuana for them, or purchasing marijuana directly from Health Canada, the department of the federal Canadian government which oversees public health matters. Although the MMAR was repealed effective March 31, 2014, preliminary injunction orders rendered by the Federal Court of Canada on March 21, 2014 and May 7, 2014 have exempted certain MMAR patients from the repeal, pending a trial of the issues raised in those proceedings.
8
Since April 1, 2014, access to medical marijuana in Canada has been exclusively pursuant to the Marihuana for Medical Purposes Regulations (or MMPR), the bulk of which came into force in 2013. The MMPR, which is also a regulation to the CDSA creates a licensing scheme for the commercial production and distribution of dried marijuana for medical purposes. Patients (other than those subject to the preliminary injunction orders previously mentioned) are now prohibited from growing their own medical marijuana. Rather, the production and distribution of medical marijuana is restricted to licensed producers pursuant to the MMPR. Licensed producers are permitted to grow strains of their choosing based on market demand and to freely set pricing for their various marijuana strains.
Our Products and Their Markets
Through our Northern Vine Canada Inc. ("N. Vine") subsidiary, we have applied to obtain a license through Health Canada for handling, extraction and analytics of marijuana. N. Vine, under Health Canada's new regulations, intends to begin operations in a commercial property in Langley, British Columbia. N. Vine will produce medical marijuana, conduct research and development, and coordinate its distribution efforts at the Langley facility. We also plan to retrofit and secure a second location in Squamish, British Columbia.
The Company has a four-pronged approach to furthering its products offered and services provided: research and development, horticultural supremacy, distribution and market penetration, and asset security. These four principles will be the focus of our growth and potential partnerships in the future.
Upon initial entry into the market, we expect to offer a number of products based on different strains of marijuana and undergo regular review based on customer feedback, sales data and customer data to ensure the optimal suite of products are available that not only maximizes sales but also meets customers' evolving needs.
For a more detailed description of the Company, please refer to the section "DESCRIPTION OF BUSINESS".
Selling Security Holder
Dutchess Opportunity Fund, II, LP
Dutchess is the manager for several global investment funds that provide capital to start-up, pre-IPO and publicly-traded companies worldwide. Since 1996, Dutchess has made over 400 investments globally, with a total transaction value exceeding $2 billion.
Dutchess provides plain vanilla, as well as structured equity and debt financings, for companies located throughout North America, UK and Continental Europe, Australia, Asia and Latin America.
For more information, visit www.dutchesscapital.com.
Dutchess may sell all or a portion of the shares being offered pursuant to this prospectus at fixed prices, at prevailing market prices at the time of sale, at varying prices or at negotiated prices.
Dutchess Investment Agreement
This prospectus relates to the resale of up to 180,000,000 Shares by Dutchess of which up to 180,000,000 Shares are to be issued to Dutchess pursuant to a Put Notice(s) under the Investment Agreement.
9
Dutchess will obtain our common stock pursuant to the Investment Agreement entered into by Dutchess and us, dated January 28, 2015.
Although we are not mandated to sell shares under the Investment Agreement, the Investment Agreement gives us the option to sell to Dutchess, up to $25,000,000 worth of Shares, without par value per share over a thirty-six (36) month period following effectiveness of the registration of which this prospectus forms a part. We may draw on the facility from time to time, as and when we determine appropriate in accordance with the terms and conditions of the Investment Agreement. The purchase price of the common stock shall be set at ninety-five percent (95%) of the volume weighted average price ("VWAP") of the common stock during the pricing period. The pricing period shall be the five (5) consecutive trading days immediately after the Put Notice Date (the "Pricing Period"). The Put Amount shall be equal to up to either (1) two-hundred-and-fifty-thousand ($250,000); (2) two hundred percent (200%) of the average daily volume (U.S. market only) of the Common Stock for the three (3) Trading Days prior to the applicable Put Notice Date. The obligation of Dutchess to purchase our shares is contingent upon our filing of a registration statement registering the shares of our Common Stock with the SEC and such registration statement being declared effective, as well as our Common Stock continuing to be listed for trading, among other things. There are no assurances that our registration statement will be deemed effective, or that we will elect to take advantage of this opportunity.
On each Put Notice submitted to Dutchess by us, we have the option to specify a suspension price ("Suspension Price") for that Put. In the event the Common Stock price falls below the Suspension Price, the Put shall be temporarily suspended. The Put shall resume at such time as the Common Stock is above the Suspension Price, provided the dates for the Pricing Period for that particular Put are still valid. In the event the Pricing Period has been completed, any shares above the Suspension Price due to Dutchess shall be sold by us at the Suspension Price under the terms of this Agreement. The Suspension Price for a Put may not be changed by us once submitted to Dutchess.
In addition, there is an ownership limit for Dutchess of 4.99%.
On any Closing Date, we shall deliver to Dutchess the number of shares of the Common Stock registered in the name of Dutchess as specified in the Put Notice. In addition, we must deliver the other required documents, instruments, and writings. Dutchess is not required to purchase the shares unless:
Our Registration Statement with respect to the resale of the shares of Common Stock delivered in connection with the applicable Put shall have been declared effective. |
We shall have obtained all material permits and qualifications required by any applicable state for the offer and sale of the Registerable Securities. |
| We shall have filed with the SEC in a timely manner all reports, notices and other documents required. |
Dutchess will not engage in any "short-sale" (as defined in Rule 200 of Regulation SHO) of our common stock at any time during this Agreement. On January 28, 2015 and as amended on April 1, 2015, we entered into a Registration Rights Agreement with Dutchess requiring, among other things that we prepare and file with the SEC a Registration Statement on Form F-1 covering the resale of the shares issuable to Dutchess under the Investment Agreement. As per the Investment Agreement, Dutchess' obligations are not assignable.
10
Offering
The following is a brief summary of this offering:
| Securities being offered | The selling stockholders identified in this prospectus may offer and sell up to 180,000,000 shares of our common stock pursuant to the Investment Agreement.
|
| Offering price per share | The selling stockholders may sell all or a portion of the shares being offered pursuant to this prospectus at fixed prices, at prevailing market prices at the time of sale, or at varying prices or at negotiated prices.
|
| Offering period | The period during which the Company may make a "Put Notice" as defined in the Investment Agreement, is thirty-six (36) months from the date the SEC declares the Registration Statement is effective.
|
| Number of shares of the Company before the offering | 69,667,951 Common Shares are currently issued and outstanding.
|
Terms of the offering | The actual price of the stock will be determined by using the prevailing market prices at the time of sale as adjusted in accordance with the Investment Agreement (which is 95% of the lowest reported trade of the Company's common stock during the "Put Period" as defined in the Investment Agreement) and the Selling Security Holder will sell the common stock offered in this prospectus. The Company will receive no proceeds from any future re-sale of any of the registered shares by Dutchess.
|
Trading market | As of the day before the date of this prospectus, the closing price of the common stock on the CSE was CDN $0.12.
As of the day before the date of this prospectus, the closing price of the common stock on the OTCQX was $0.101.
|
Number of shares of the Company after the offering | 249,667,951 Common Shares.
|
11
Summary Financial Data
The summary financial information set forth below has been derived from the financial statements of Abattis for periods presented and should be read in conjunction with the financial statements and the notes thereto included elsewhere in this prospectus and in the information set forth in the section titled "Management's Discussion and Analysis of Financial Condition and Results of Operations."
| Years Ended September 30, | ||||
| 2014 | 2013 | |||
| Revenues | $ | 7,720 | $ | 17,448 |
| Operating Expenses | $ | 7,378,559 | $ | 1,049,151 |
| Other Income (Expenses) | $ | (280,562) | $ | (58,982) |
| Net Income (Loss) | $ | 7,611,540 | $ | (1,102,491) |
| Total Assets | $ | 5,903,803 | $ | 1,629,346 |
| Total Liabilities | $ | 1,260,767 | $ | 1,071,329 |
| Shareholders' Equity (Deficit) | $ | 4,043,225 | $ | 558,017 |
| Total Liabilities and Shareholders' Equity | $ | 5,903,803 | $ | 1,629,346 |
12
This section of the prospectus discloses all material risks known to us. We do not make, nor have we authorized any other person to make, any representation about the future market value of our common stock. In addition to the other information contained in this prospectus, the following factors should be considered carefully in evaluating an investment in our securities. If any of the risks discussed below materialize, our common stock could decline in value or become worthless.
Risk Factors Related to the Offering
Existing stockholders may experience significant dilution from the sale of our common stock pursuant to the Investment Agreement.
The sale of our common stock to Dutchess in accordance with the Investment Agreement may have a dilutive impact on our shareholders. As a result, our net income per share could decrease in future periods and the market price of our common stock could decline. In addition, the lower our stock price is at the time we exercise our put options, the more shares of our common stock we will have to issue to Dutchess in order to exercise a put under the Investment Agreement. If our stock price decreases, then our existing shareholders would experience greater dilution for any given dollar amount raised through the offering.
The perceived risk of dilution may cause our stockholders to sell their shares, which may cause a decline in the price of our common stock. Moreover, the perceived risk of dilution and the resulting downward pressure on our stock price could encourage investors to engage in short sales of our common stock. By increasing the number of shares offered for sale, material amounts of short selling could further contribute to progressive price declines in our common stock.
The issuance of shares pursuant to the Investment Agreement may have a significant dilutive effect.
Depending on the number of shares we issue pursuant to the Investment Agreement, it could have a significant dilutive effect upon our existing shareholders. Although the number of shares that we may issue pursuant to the Investment Agreement will vary based on our stock price (the higher our stock price, the less shares we have to issue) the information set out below indicates the potential dilutive effect to our shareholders, based on different potential future stock prices, if the full amount of the Investment Agreement is realized.
Dilution based upon common stock put to Dutchess and the stock price discounted to Dutchess' purchase price of 95% of the Market Price of the VWAP during the pricing period. The example below illustrates dilution based upon a $0.20 market price/$0.19 purchase price and other increased/decreased prices (without regard to Dutchess' 4.99% ownership limit):
13
$25,000,000 Put
| Stock Price (Dutchess Purchase Price) | Shares Issued | Percentage of Outstanding Shares(1) | |||
| $1.00 x 95% = $0.95 | 26,315,789 | 37.77% | |||
| $0.80 x 95% = $0.76 | 32,894,737 | 47.22% | |||
| $0.60 x 95% = $0.57 | 43,859,649 | 62.96% | |||
| $0.45 x 95% = $0.43 | 58,139,535 | 83.45% | |||
| $0.30 x 95% = $0.29 | 86,206,896 | 123.74% | |||
| $0.20 x 95% = $0.19 | 131,578,947 | 188.87% | |||
| $0.12 x 95% = $0.11 | 227,272,727 | 326.22% |
Note:
| (1) | Based on 69,667,951 shares outstanding as of April 27, 2015. |
Dutchess will pay less than the then-prevailing market price of our common stock, which could cause the price of our common stock to decline.
Our common stock to be issued under the Investment Agreement will be purchased at a discount of 95% of the Market Price (subject to a minimum price of $0.05) of the VWAP during the five trading days immediately following our notice to Dutchess of our election to exercise our "put" right.
Dutchess has a financial incentive to sell our shares immediately upon receiving the shares to realize the profit between the discounted price and the market price. If Dutchess sells our shares, the price of our common stock may decrease. If our stock price decreases, Dutchess may have a further incentive to sell such shares. Accordingly, the discounted sales price in the Investment Agreements may cause the price of our common stock to decline.
Dutchess has entered into similar agreements with other public companies and may not have sufficient capital to meet our put notices.
Dutchess has entered into similar Investment Agreements with other public companies, and some of those companies have filed registration statements with the intent of registering shares to be sold to Dutchess pursuant to the Investment Agreement. We do not know if management at any of the companies who have or will have effective registration statements intend to raise funds now or in the future, what the size or frequency of each put request would be, if floors will be used to restrict the amount of shares sold, or if the Investment Agreement will ultimately be cancelled or expire before the entire amount of shares are put to Dutchess. Since we do not have any control over the requests of these other companies, if Dutchess receives significant requests, it may not have the financial ability to meet our requests. If so, the amount of available funds may be significantly less than we anticipate.
14
We are registering an aggregate of 180,000,000 shares of common stock to be issued under the Investment Agreement. The sale of such shares could depress the market price of our common stock.
We are registering an aggregate of 180,000,000 shares of common stock under the registration statement of which this prospectus forms a part for issuance pursuant to the Investment Agreement. The sale of these shares into the public market by Dutchess could depress the market price of our common stock.
Risks Related to Our Business and Industry
We are reliant on our senior management team. The loss of any of these key personnel may hamper our ability to manage a publicly traded company while developing our products, which may harm our business.
The Company strongly depends on the business and technical expertise of its management and consultants, and it is unlikely that this dependence will decrease in the near term. Loss of the Company's key personnel could slow the Company's ability to innovate, although the effect on ongoing operations would be manageable as experienced key operations personnel could be put in place. As the Company's operations expand, additional general management resources will be required.
If the Company expands its operations, the ability of the Company to recruit, train, integrate and manage a large number of new employees is uncertain and failure to do so would have a negative impact on the Company's business plans.
Given our lack of revenue and cash flow, we will need to raise additional capital, which may be unavailable to us or, even if consummated, may cause dilution or place significant restrictions on our ability to operate.
According to our management's estimates, based on our current cash on hand and further based on our budget, we believe that we will have sufficient resources to continue our activities only into June, 2015.
Since we are likely to be unable to generate sufficient, if any, revenue or cash flow to fund our operations for the foreseeable future, we will need to seek additional equity or debt financing to provide the capital required to establish or expand our operations. We may also need additional funding for developing products and services, increasing our sales and marketing capabilities, promoting brand identity, and acquiring complementary companies, technologies and assets, as well as for working capital requirements and other operating and general corporate purposes.
Other than our agreement with Dutchess, we do not currently have any arrangements or credit facilities in place as a source of funds, and there can be no assurance that we will be able to raise sufficient additional capital on acceptable terms, or at all. If such financing is not available on satisfactory terms, or is not available at all, we may be required to delay, scale back or eliminate the development of business opportunities, our operations and financial condition may be materially adversely affected and our business might fail.
If we raise additional capital by issuing equity securities, the percentage ownership of our existing stockholders may be reduced, and accordingly these stockholders may experience substantial dilution. We may also issue equity securities that provide for rights, preferences and privileges senior to those of our common stock. Given our need for cash and that equity raising is the most common type of fundraising for companies like ours, the risk of dilution is particularly significant for stockholders of our company.
Debt financing, if obtained, may involve agreements that include liens on our assets, covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, could increase our
15
expenses and require that our assets be provided as a security for such debt. Debt financing would also be required to be repaid regardless of our operating results.
If we raise additional funds through collaborations and licensing arrangements, we may be required to relinquish some rights to our technologies or candidate products, or to grant licenses on terms that are not favorable to us.
We are subject to significant regulation by the Canadian Federal Government. There is no assurance that we or our affiliates will be granted licensed producer status by Health Canada. Any failure or delay in obtaining such status would materially and adversely affect our operations.
The Company depends heavily on the success of acquiring a Production License from Health Canada to be able to grow, store and distribute medical marijuana in Canada. There is no assurance that we or our affiliates will be approved by Health Canada or will be granted licensed producer status. Should we or our affiliates be unable to obtain all required licenses, or if the regulations in Canada continue to change, our business would not be able to operate or there could be significant cost to change our operations to remain compliant with the laws and regulations.
Once a Production License is obtained, any failure to comply with the terms of the Production License, or any failure to renew the Production License after its expiry date, would have a material adverse impact on the financial condition and operations of our business.
The Company's operations are subject to regulations promulgated by government regulatory agencies from time to time. The cost of compliance with changes in governmental regulations has the potential to reduce the profitability of operations. However, there can be no guarantee that the Company will be able to obtain and maintain, at all times, all necessary licenses and permits required to carry out its business.
There are many regulations and governmental agencies that regulate the medical marijuana industry, and there will likely be increased and/or changing regulations as the industry becomes more mainstream with more participants.
The Company's operations are subject to a variety laws, regulations and guidelines relating to the manufacture, management, transportation, storage and disposal of medical marijuana but also including laws and regulations relating to health and safety, the conduct of operations and the protection of the environment. While to the knowledge of management, the Company is currently in compliance with all such laws, changes to such laws, regulations and guidelines due to matters beyond the control of the Company may cause adverse effects to its operations.
The MMPR is a new regime established in June 2013. As such, revisions to the regime could be implemented which could have an impact on the Company's operations. There is also some uncertainty regarding the likely interpretation of certain regulatory provisions by the regulator. Changes in legislation or regulator interpretation could negatively impact the operations of the Company. Similarly, a change in government could result in meaningful changes to the regulatory regime under which the Company operates, which could negatively impact its operations.
On March 21, 2014, the Federal Court of Canada issued an order affecting the repeal of the MMAR and the application of certain portions of the MMPR which are inconsistent with the MMAR in response to a motion brought by four individuals. As of the date of this prospectus, the Government of Canada has indicated its intention to appeal the order but it is unclear whether this will be successful or how the Federal Court of Canada might ultimately decide the case to which the order relates. The risks to the business of the Company represented by this or similar actions are that they might lead to court rulings or legislative changes that allow those with existing licenses to possess and/or grow medical marijuana and perhaps others to opt out of the regulated supply system implemented through the MMPR, in which the Company is a Licensed Producer. This could significantly reduce the addressable market for the
16
Company's products and could materially and adversely affect the business, financial condition and results of operations of the Company.
While the impact of such changes are uncertain and are highly dependent on which specific laws, regulations or guidelines are changed and on the outcome of any such court actions, it is not expected that any such changes would have an effect on the Company's operations that is materially different than the effect on similar-sized companies in the same business as the Company.
There are sales risks associated with the cannabis and medical marijuana industry because cannabis is a controlled substance.
As cannabis is a controlled substance in Canada, direct consumer marketing is not allowed. All products can only be prescribed by a physician and, to be successful, companies will have to develop programs targeted to this group. Traditionally in this sector, growers have targeted users as opposed to the doctors. The new regulations will change this traditional approach and will require growers to target doctors. If we are unable to properly conduct sales in a regulated environment or target the appropriate audiences for our medical marijuana products, our results of operations and business prospects could be substantially impaired.
Cannabis is not legal to grow in the U.S. under federal law, although it may be imported and sold in the U.S. Importation is subject to a "zero tolerance" policy as a controlled substance under the U.S. Controlled Substances Act.
In certain states, the growth and cultivation of cannabis is legal (California, Colorado, Hawaii, Maine, Maryland, Michigan, Montana, New Mexico, Oregon, Rhode Island, Vermont and Washington), although states are resistant to allow the cultivation of cannabis due to resistance from the U.S. Department of Drug Enforcement Agency and prohibitions of federal law. Upon the production and sale of marijuana-based products to consumers, our products will be required to comply with various regulations at the province and Canadian levels. At this time, our product will only be sold in Canada under the MMPR. We will be unable for the foreseeable future to sell our products in the United States, which would impair our prospects for revenue and profit.
There are safety operational risks related to the cultivation and storage of our products at the facilities.
Given the nature of the product and its lack of legal availability outside of the therapeutic channels, as well as the concentration of abundant stock within one facility, despite meeting or exceeding Health Canada's security requirements, there remains a risk of shrinkage as well as theft. Also, as an agricultural product, despite meeting or exceeding Health Canada's requirements regarding good production practices, there remains a risk of diseases and pests impacting not only yield and revenue but overall product quality to consumers.
We may not be able to use the facilities as planned and will therefore not be able to commence operations on the timetable or the scale that we have planned.
To date, the Company's activities and resources have been primarily focused on its facility near Vancouver, BC and the Company will continue to be focused on this facility for the foreseeable future. Adverse changes or developments affecting the facility, including but not limited to a breach of security, could have a material and adverse effect on the Company's business, financial condition and prospects. Any breach of the security measures and other facility requirements, including any failure to comply with recommendations or requirements arising from inspections by Health Canada, could also have an impact on the Company's ability to continue operating under the License.
17
We may not acquire market share or achieve profits due to competition in the medical marihuana industry.
There is potential that the Company will face intense competition from other companies, some of which can be expected to have longer operating histories and more financial resources and manufacturing and marketing experience than the Company. Increased competition by larger and better financed competitors could materially and adversely affect the business, financial condition and results of operations of the Company.
Because of the early stage of the industry in which the Company operates, the Company expects to face additional competition from new entrants. If the number of users of medical marijuana in Canada increases, the demand for products will increase and the Company expects that competition will become more intense, as current and future competitors begin to offer an increasing number of diversified products. To remain competitive, the Company will require a continued high level of investment in research and development, marketing, sales and client support. The Company may not have sufficient resources to maintain research and development, marketing, sales and client support efforts on a competitive basis which could materially and adversely affect the business, financial condition and results of operations of the Company.
We are subject to risks inherent in an agricultural business.
The Company's business involves the growing of medical marijuana, an agricultural product. As such, the business is subject to the risks inherent in the agricultural business, such as insects, plant diseases and similar agricultural risks. Although the Company intends to grow its products indoors under climate controlled conditions and carefully monitor the growing conditions with trained personnel, there can be no assurance that natural elements will not have a material adverse effect on the production of its products.
We are subject to risks related to the availability of seed supply.
Our ability to commence and continue operations will be dependent on our ability to acquire starting materials. There are four legal sources of starting materials under the MMPR: Health Canada; Personal-Use Production License holders under the MMAR regime; Designated-Person Production License holders under the MMAR regime; and importation. There is no guarantee that we will be able to acquire seeds from such sources and a failure to do so would limit our ability to produce the predicted amounts of product. This would have an adverse effect on our operations and financial results.
We are subject to risks related to rising energy costs.
Medical marijuana growing operations consume considerable energy, making the Company vulnerable to rising energy costs. Rising or volatile energy costs may adversely impact the business of the Company and its ability to operate profitably.
We are subject to risks related to transportation disruptions.
Due to the perishable and premium nature of the Company's products, the Company will depend on fast and efficient courier services to distribute its product. Any prolonged disruption of this courier service could have an adverse effect on the financial condition and results of operations of the Company. Rising costs associated with the courier services used by the Company to ship its products may also adversely impact the business of the Company and its ability to operate profitably.
No commercial products have been developed.
We have not completed the development of any commercial products, and accordingly we have not begun to market or generate revenues from sales of the products we are developing.
18
There can be no assurance that any of our product candidates will meet applicable health regulatory standards, obtain required regulatory approvals, be capable of being produced in commercial quantities at reasonable costs, be successfully marketed or that the investment made in such product candidates will be recouped through sales or related royalties. There can be no assurance that we will ever achieve profitability. As a result, an investment in our common shares involves a high degree of risk and should be considered only by those persons who can afford a total loss of their investment.
Our reputation in the industry will be very important as we grow the business, and any negative impact on our reputation could be damaging to our business.
The Company believes the medical marijuana industry is highly dependent upon consumer perception regarding the safety, efficacy and quality of the medical marijuana produced. Consumer perception of the Company's products can be significantly influenced by scientific research or findings, regulatory investigations, litigation, media attention and other publicity regarding the consumption of medical marijuana products. There can be no assurance that future scientific research, findings, regulatory proceedings, litigation, media attention or other research findings or publicity will be favourable to the medical marijuana market or any particular product, or consistent with earlier publicity. Future research reports, findings, regulatory proceedings, litigation, media attention or other publicity that are perceived as less favourable than, or that question, earlier research reports, findings or publicity could have a material adverse effect on the demand for the Company's products and the business, results of operations, financial condition and cash flows of the Company.
There are risks related to the quality and quality control of our products.
As a manufacturer and distributor of products designed to be ingested by humans, the Company faces an inherent risk of exposure to product liability claims, regulatory action and litigation if its products are alleged to have caused significant loss or injury. In addition, the manufacture and sale of the Company's products involve the risk of injury to consumers due to tampering by unauthorized third parties or product contamination. Previously unknown adverse reactions resulting from human consumption of the Company's products alone or in combination with other medications or substances could occur. The Company may be subject to various product liability claims, including, among others, that the Company's products caused injury or illness, include inadequate instructions for use or include inadequate warnings concerning possible side effects or interactions with other substances. A product liability claim or regulatory action against the Company could result in increased costs, could adversely affect the Company's reputation with its clients and consumers generally, and could have a material adverse effect on our results of operations and financial condition of the Company.
There can be no assurances that the Company will be able to obtain or maintain product liability insurance on acceptable terms or with adequate coverage against potential liabilities. Such insurance is expensive and may not be available in the future on acceptable terms, or at all. The inability to obtain sufficient insurance coverage on reasonable terms or to otherwise protect against potential product liability claims could prevent or inhibit the commercialization of the Company's potential products.
There are risks related to potential product recalls.
Manufacturers and distributors of products are sometimes subject to the recall or return of their products for a variety of reasons, including product defects, such as contamination, unintended harmful side effects or interactions with other substances, packaging safety and inadequate or inaccurate labeling disclosure. If any of the Company's products are recalled due to an alleged product defect or for any other reason, the Company could be required to incur the unexpected expense of the recall and any legal proceedings that might arise in connection with the recall. The Company may lose a significant amount of sales and may not be able to replace those sales at an acceptable margin or at all. In addition, a product recall may require significant management attention. Although the Company has detailed procedures in place for
19
testing finished products, there can be no assurance that any quality, potency or contamination problems will be detected in time to avoid unforeseen product recalls, regulatory action or lawsuits. Additionally, if one of the Company's significant brands were subject to recall, the image of that brand and the Company could be harmed. A recall for any of the foregoing reasons could lead to decreased demand for the Company's products and could have a material adverse effect on the results of operations and financial condition of the Company. Additionally, product recalls may lead to increased scrutiny of the Company's operations by Health Canada or other regulatory agencies, requiring further management attention and potential legal fees and other expenses.
There are risks related to product liability claims.
Our product candidates subject us to the risk of product liability claims for which we may not be able to maintain or obtain adequate insurance coverage. Inherent in the use of our product candidates in clinical trials, as well as in the manufacturing and distribution in the future of any approved products, is the risk of financial exposure to product liability claims and adverse publicity in the event that the use of such products results in personal injury or death. There can be no assurance that we will not experience losses due to product liability claims in the future.
There are risks related to potential delays or impairment of future sales.
Even if any of our product candidates receives regulatory approval, we and our collaborators may still face development and regulatory difficulties that may delay or impair future sales. If we or our collaborators obtain regulatory approval for any of our product candidates, we and our collaborators will continue to be subject to extensive regulation by Health Canada, the FDA, other federal authorities, certain state agencies and regulatory authorities elsewhere. These regulations will impact many aspects of our operations and the drug manufacturer's operations including manufacture, record keeping, quality control, adverse event reporting, storage, labelling, advertising, promotion, sale and distribution, export and personnel. The FDA and state agencies may conduct periodic inspections to assess compliance with these requirements. We, together with our collaborators, will be required to conduct post-marketing surveillance of the product. We also may be required to conduct post-marketing studies. Our or our collaborators' failure to comply with applicable FDA and other regulatory requirements, or the later discovery of previously unknown problems, may result in restrictions including:
| delays in commercialization; |
| refusal by Health Canada, the FDA or other similar regulatory agencies to review pending applications or supplements to approved applications; |
| product recalls or seizures; |
| warning letters; |
| suspension of manufacturing; |
| withdrawals of previously approved marketing applications; |
| fines or other civil penalties; |
| injunctions, suspensions or revocations of marketing licenses; |
| refusals to permit products to be imported to or exported from the United States; and |
| criminal prosecutions. |
There are risks related to intellectual property.
Our success depends on our ability to protect our proprietary rights and operate without infringing the proprietary rights of others; we may incur significant expenses or be prevented from developing and/or commercializing products as a result of an intellectual property infringement claim.
Our success will depend in part on our ability and that of our corporate collaborators to obtain and enforce patents and maintain trade secrets, in Canada, the United States and in other countries.
20
Patent law relating to the scope and enforceability of claims in the fields in which we operate is still evolving. The patent positions of biotechnology and biopharmaceutical companies, including us, is highly uncertain and involves complex legal and technical questions for which legal principles are not firmly established. The degree of future protection for our proprietary rights, therefore, is highly uncertain. In this regard there can be no assurance that patents will issue from any of the pending patent applications. In addition, there may be issued patents and pending applications owned by others directed to technologies relevant to our or our corporate collaborators' research, development and commercialization efforts. There can be no assurance that our or our corporate collaborators' technology can be developed and commercialized without a license to such patents or that such patent applications will not be granted priority over patent applications filed by us or one of our corporate collaborators.
Our commercial success depends significantly on our ability to operate without infringing the patents and proprietary rights of third parties, and there can be no assurance that our and our corporate collaborators' technologies and products do not or will not infringe the patents or proprietary rights of others.
There can be no assurance that third parties will not independently develop similar or alternative technologies to ours, duplicate any of our technologies or the technologies of our corporate collaborators or our licensors, or design around the patented technologies developed by us, our corporate collaborators or our licensors. The occurrence of any of these events would have a material adverse effect on our business, financial condition and results of operations.
Litigation may also be necessary to enforce patents issued or licensed to us or our corporate collaborators or to determine the scope and validity of a third party's proprietary rights. We could incur substantial costs if litigation is required to defend ourselves in patent suits brought by third parties, if we participate in patent suits brought against or initiated by our corporate collaborators or if we initiate such suits, and there can be no assurance that funds or resources would be available in the event of any such litigation. An adverse outcome in litigation or an interference to determine priority or other proceeding in a court or patent office could subject us to significant liabilities, require disputed rights to be licensed from other parties or require us or our corporate collaborators to cease using certain technology or products, any of which may have a material adverse effect on our business, financial condition and results of operations.
The Company will have to expand its patent protection to other countries. There can be no assurances that the Company will be able to do so successfully. The Company may not have the financial resources to enforce its patents should another company compete with a similar or identical product that infringes on the Company's patents.
There is no assurance that we will be able to obtain required supply of materials and skilled labor.
The ability of the Company to compete and grow will be dependent on it having access, at a reasonable cost and in a timely manner, to skilled labour, equipment, parts and components. No assurances can be given that the Company will be successful in maintaining its required supply of skilled labour, equipment, parts and components. It is also possible that the final costs of the major equipment contemplated by the Company's capital expenditure program may be significantly greater than anticipated by the Company's management, and may be greater than funds available to the Company, in which circumstance the Company may curtail, or extend the timeframes for completing, its capital expenditure plans. This could have an adverse effect on the financial results of the Company.
The Company's business is dependent on a number of key inputs and their related costs including raw materials and supplies related to its growing operations, as well as electricity, water and other local utilities. Any significant interruption or negative change in the availability or economics of the supply chain for key inputs could materially impact the business, financial condition and operating results of the Company. Any inability to secure required supplies and services or to do so on appropriate terms could
21
have a materially adverse impact on the business, financial condition and operating results of the Company.
Our forecasts are highly speculative in nature and we cannot predict results in a development stage company with a high degree of accuracy.
The Company must rely largely on its own market research to forecast sales as detailed forecasts are not generally obtainable from other sources at this early stage of the medical marijuana industry in North America. A failure in the demand for its products to materialize as a result of competition, technological change or other factors could have a material adverse effect on the business, results of operations and financial condition of the Company.
If we incur substantial liability from litigation, complaints, or enforcement actions our financial condition could suffer.
The Company may become party to litigation from time to time in the ordinary course of business which could adversely affect its business. Should any litigation in which the Company becomes involved be determined against the Company such a decision could adversely affect the Company's ability to continue operating and the market price for the Company's Common Shares and could use significant resources. Even if the Company is involved in litigation and wins, litigation can redirect significant Company resources.
Risks Related to the Ownership of Our Common Stock
Our common shares are thinly traded and you may be unable to sell at or near asking prices, or at all.
We do not have a liquid market for our common stock, and we cannot predict the extent to which an active public market for trading our common stock will be achieved or sustained.
This situation is attributable to a number of factors, including the fact that we are a small company that is relatively unknown to stock analysts, stockbrokers, institutional investors and others in the investment community who generate or influence sales volume. Even if we came to the attention of such persons, those persons tend to be risk-averse and may be reluctant to follow, purchase, or recommend the purchase of shares of an unproven company such as ours until such time as we become more seasoned and viable. As a consequence, there may be periods of several days or more when trading activity in our shares is minimal or non-existent, as compared to a seasoned issuer which has a large and steady volume of trading activity that will generally support continuous sales without an adverse effect on share price. We cannot give you any assurance that a broader or more active public trading market for our common stock will develop or be sustained, or that current trading levels will be sustained.
The market price for our common stock is particularly volatile given our status as a relatively small company, which could lead to wide fluctuations in our share price. You may be unable to sell your common stock at or above your purchase price if at all, which may result in substantial losses to you.
Shareholders should be aware that, according to SEC Release No. 34-29093, the market for penny stocks has suffered in recent years from patterns of fraud and abuse. Such patterns include (1) control of the market for the security by one or a few broker-dealers that are often related to the promoter or issuer; (2) manipulation of prices through prearranged matching of purchases and sales and false and misleading press releases; (3) boiler room practices involving high-pressure sales tactics and unrealistic price projections by inexperienced sales persons; (4) excessive and undisclosed bid-ask differential and markups by selling broker-dealers; and (5) the wholesale dumping of the same securities by promoters and broker-dealers after prices have been manipulated to a desired level, along with the resulting inevitable collapse of those prices and with consequent investor losses. Our management is aware of the abuses that have occurred historically in the penny stock market. Although we do not expect to be in a
22
position to dictate the behavior of the market or of broker-dealers who participate in the market, management will strive within the confines of practical limitations to prevent the described patterns from being established with respect to our securities. The occurrence of these patterns or practices could increase the volatility of our share price.
Our stock price may be volatile, which may result in losses to our shareholders.
The market price for the Common Shares may be volatile and subject to wide fluctuations in response to numerous factors, many of which are beyond the Company's control, including the following:
| actual or anticipated fluctuations in the Company's quarterly results of operations; |
| recommendations by securities research analysts; |
| changes in the economic performance or market valuations of companies in the industry in which the Company operates; |
| addition or departure of the Company's executive officers and other key personnel; |
| release or expiration of transfer restrictions on outstanding Common Shares; |
| sales or perceived sales of additional Common Shares; |
| operating and financial performance that vary from the expectations of management, securities analysts and investors; |
| regulatory changes affecting the Company's industry generally and its business and operations; |
| announcements of developments and other material events by the Company or its competitors; |
| changes in global financial markets and global economies and general market conditions, such as interest rates and pharmaceutical product price volatility; |
| significant acquisitions or business combinations, strategic partnerships, joint ventures or capital commitments by or involving the Company or its competitors; |
| operating and share price performance of other companies that investors deem comparable to the Company or from a lack of market comparable companies; and |
| news reports relating to trends, concerns, technological or competitive developments, regulatory changes and other related issues in the Company's industry or target markets. |
Financial markets have recently experienced significant price and volume fluctuations that have particularly affected the market prices of equity securities of companies and that have often been unrelated to the operating performance, underlying asset values or prospects of such companies. Accordingly, the market price of the Common Shares may decline even if the Company's operating results, underlying asset values or prospects have not changed. Additionally, these factors, as well as other related factors, may cause decreases in asset values that are deemed to be other than temporary, which may result in impairment losses. There can be no assurance that continuing fluctuations in price and volume will not occur. If such increased levels of volatility and market turmoil continue, the Company's operations could be adversely impacted and the trading price of the Common Shares may be materially adversely affected.
We do not expect to declare any dividends in the foreseeable future.
We do not anticipate declaring any cash dividends to holders of our common stock in the foreseeable future. Consequently, investors may need to rely on sales of their common stock after price appreciation, which may never occur, as the only way to realize any future gains on their investment. Investors seeking cash dividends should not purchase our common stock.
Because the SEC imposes additional sales practice requirements on brokers who deal in shares of penny stocks, some brokers may be unwilling to trade our securities. This means that you may have difficulty reselling your shares, which may cause the value of your investment to decline.
23
Our shares are classified as penny stocks. The SEC has adopted regulations which generally define a "penny stock" to be any equity security that has a price of less than $5.00 per share or an exercise price of less than $5.00 per share, subject to certain exceptions (including the issuer of the securities having net tangible assets (i.e., total assets less intangible assets and liabilities) in excess of $2,000,000 or average revenue of at least $6,000,000 for the last three years). As a result, our common stock could be subject to these rules that impose additional sales practice requirements on broker-dealers who sell our securities to persons other than established customers and accredited investors (generally persons with a net worth in excess of $1,000,000 or annual income exceeding $200,000, or $300,000 together with their spouse). For transactions covered by these rules, the broker-dealer must make a special suitability determination for the purchase of such securities and have received the purchaser's written consent to the transaction prior to the purchase. Additionally, for any transaction involving a "penny stock," unless exempt, the rules require the delivery, prior to the transaction, of a risk disclosure document mandated by the SEC relating to the "penny stock" market. The broker-dealer must also disclose the commissions payable to both the broker-dealer and the registered representative, current quotations for the securities and, if the broker-dealer is the sole market maker, the broker-dealer must disclose this fact and the broker-dealer's presumed control over the market. Finally, monthly statements must be sent disclosing recent price information for the "penny stock" held in the account and information on the limited market in "penny stocks." Consequently, although the "penny stock" rules do not currently apply to our securities, if these rules do become applicable in the future, this may restrict the ability of broker-dealers to sell our securities.
Securities analysts may not cover our common stock and this may have a negative impact on our common stock's market price.
The trading market for our common stock may depend on the research and reports that securities analysts publish about us or our business. We do not have any control over these analysts. There is no guarantee that securities analysts will cover our common stock. If securities analysts do not cover our common stock, the lack of research coverage may adversely affect our common stock's market price, if any. If we are covered by securities analysts, and our stock is downgraded, our stock price would likely decline. If one or more of these analysts ceases to cover us or fails to publish regularly reports on us, we could lose visibility in the financial markets, which could cause our stock price or trading volume to decline.
Investment Industry Regulatory Organization of Canada (IIROC) and Financial Industry Regulatory Authority ("FINRA") sales practice requirements may limit your ability to buy and sell our common stock, which could depress the price of our shares.
IIROC and FINRA rules require broker-dealers to have reasonable grounds for believing that an investment is suitable for a customer before recommending that investment to the customer. Prior to recommending speculative low-priced securities to their non-institutional customers, broker-dealers must make reasonable efforts to obtain information about the customer's financial status, tax status and investment objectives, among other things. Under interpretations of these rules, IIROC and FINRA believes that there is a high probability such speculative low-priced securities will not be suitable for at least some customers. Thus, IIROC and FINRA requirements make it more difficult for broker-dealers to recommend that their customers buy our common stock, which may limit your ability to buy and sell our shares, have an adverse effect on the market for our shares, and thereby depress our share price.
Volatility in our common share price may subject us to securities litigation.
The market for our common stock is characterized by significant price volatility as compared to seasoned issuers, and we expect that our share price will continue to be more volatile than a seasoned issuer for the indefinite future. In the past, plaintiffs have often initiated securities class action litigation against a company following periods of volatility in the market price of its securities. Although we are not currently the subject of any pending litigation, we may, in the future, be the target of such litigation.
24
Securities litigation could result in substantial costs and liabilities and could divert management's attention and resources.
Dilution of common stock
To conduct its business, the Company may from time to time require additional funds. The Company may have to issue additional securities including, but not limited to, common shares or some form of convertible security, the effect of which will result in a dilution of the equity interests of any existing shareholders.
CSE
The principal market on which the common shares of the Company are traded is the Canadian Securities Exchange ("CSE") under the symbol "ATT".
Trading the Company's common stock on the CSE began on December 23, 2010. Shown below are the range of high and low sales prices of common stock for the two most recent fiscal years as reported on www.quotemedia.com.
| Quarter Ended | High | Low |
| March 31, 2015 | $0.215 | $0.09 |
| December 31, 2014 | $0.36 | $0.15 |
| September 30, 2014 | $0.70 | $0.24 |
| June 30, 2014 | $1.98 | $0.40 |
| March 31, 2014 | $2.46 | $0.02 |
| December 31, 2013 | $0.09 | $0.02 |
| September 30, 2013 | $0.15 | $0.05 |
| June 30, 2013 | $0.19 | $0.05 |
| March 31, 2013 | $0.28 | $0.14 |
As of the day before the date of this prospectus, the closing price of the common stock on the CSE was CDN $0.12.
OTCQX
In the United States, the common shares of the Company are quoted on the OTCQX under the symbol "ATTBF". Trading of the Company's common stock on the OTCQX began on August 5, 2014. Prior to that date the Company's stock was quoted on the OTC Pinks under the same symbol: "ATTBF".
Shown below are the high and low sales prices of the common stock as reported by the OTCQX for the two most recent fiscal years as reported on by the OTC Markets Group Inc.
| Quarter Ended | High | Low |
| March 31, 2015 | $0.168 | $0.072 |
| December 31, 2014 | $0.3280 | $0.1309 |
| September 30, 2014 | $0.4900 | $0.2250 |
| June 30, 2014 | $0.7150 | $0.3716 |
| March 31, 2014 | $2.2100 | $0.0161 |
| December 31, 2013 | $0.1187 | $0.0250 |
| September 30, 2013 | $0.1520 | $0.0463 |
| June 30, 2013 | $0.1792 | $0.1000 |
| March 31, 2013 | $0.2870 | $0.1560 |
25
As of the day before the date of this prospectus, the closing price of the common stock on the OTCQX was U.S. $0.101.
The above quotations reflect inter-dealer prices, without retail mark-up, mark-down, or commission and may not necessarily represent actual transactions.
Holders
As of the date of this prospectus, there are 69,667,951 issued and outstanding shares of common stock and 193 shareholders of record.
We will not receive any proceeds from the sale of the shares of our common stock by the selling stockholders. All proceeds from the sale of such shares will be for the account of the selling stockholders. We will pay for expenses of this offering, except that the selling stockholders will pay any broker discounts or commissions or equivalent expenses applicable to the sale of their shares.
We agreed to register for resale shares of common stock of the selling security holder, Dutchess. Dutchess may from time to time offer and sell any or all of their shares that are registered under this prospectus. Dutchess and any participating broker-dealers are "underwriters" within the meaning of Securities Act, as amended. All expenses incurred with respect to the registration of the common stock will be borne by us, but we will not be obligated to pay any underwriting fees, discounts, commissions or other expenses incurred by Dutchess in connection with the sale of such shares.
The following table provides information regarding the beneficial ownership of our common stock held by Dutchess as of the date of this prospectus, including:
| the number of shares owned by Dutchess prior to this offering; |
| the total number of shares that are to be offered by Dutchess; |
| the total number of shares that will be owned by Dutchess upon completion of the offering; |
| the percentage owned by Dutchess upon completion of the offering; and |
the identity of the beneficial holder of any entity that owns the shares. |
The named party beneficially owns and has sole voting and investment power over all shares or rights to the shares, unless otherwise shown in the table. The numbers in this table assumes that Dutchess sells shares of common stock being offered in this prospectus or purchases additional shares of common stock,
26
and assumes that all shares offered are sold. The percentages are based on 69,667,951 shares of common stock outstanding on April 27, 2015.
Name of Selling Shareholder | Shares Owned Prior to this Offering | Total Number of Shares to be Offered for | Total Shares to be Owned Upon Completion of this Offering | Percent Owned Upon Completion of this Offering(2) |
Dutchess Opportunity Fund, II, LP(3) 50 Commonwealth Avenue, Suite 2, Boston, MA 02116 | 0 | 180,000,000 | 0 | 0%(4) |
Notes:
| (1) | The actual number of shares of common stock offered in this prospectus, and included in the registration statement of which this prospectus is a part, includes such additional number of shares of common stock as may be issued or issuable upon drawn down under the Agreement with Dutchess. |
| (2) | Based on the assumption 249,667,951 shares of common stock of the Company will be outstanding at the close of this offering. |
| (3) | Douglas H. Leighton is the Managing Director of Dutchess and, in that capacity, has the authority to direct voting and investment decisions regarding the securities. |
| (4) | Dutchess has not: (i) has had a material relationship with us other than as a shareholder at any time within the past three years or (ii) has ever been one of our officers or directors. |
Investment Agreement / Registration Rights Agreement
On January 28, 2015, we entered into the Investment Agreement and a Registration Rights Agreement with Dutchess in order to establish a possible source of funding for us.
Under the Investment Agreement, Dutchess has agreed to provide us with up to $25,000,000 of funding over a thirty-six (36) month period from the effective date of this prospectus. 180,000,000 Shares are being registered pursuant to this prospectus; of which up to 180,000,000 shares of common stock are to be issued to Dutchess pursuant to a Put Notice(s) under the Investment Agreement. During this period, we can deliver a put under the Investment Agreement by selling shares of our common stock to Dutchess and Dutchess will be obligated to purchase the shares. A put transaction must close before we can deliver another put notice to Dutchess.
We may request a put by sending a put notice to Dutchess, stating the amount of the put of either $250,000 or 200% of the average daily dollar volume multiplied by the average of the three daily closing prices immediately preceding the Put Date. During the five trading days following a notice, we will calculate the amount of shares we will sell to Dutchess and the purchase price per share. The number of shares of Common Stock that Dutchess shall purchase pursuant to each put notice shall be determined by dividing the amount of the put by the purchase price.
27
The purchase price per share of common stock will be set at a discount of 95% if the Market Price is up to $0.50 (subject to a minimum price of $0.05) of the VWAP during the five trading days immediately following our notice to Dutchess of our election to exercise our "put" right. On each Put Notice submitted to Dutchess by us, we have the option to specify a Suspension Price for that Put. In the event the Common Stock price falls below the Suspension Price, the Put shall be temporarily suspended. The Put shall resume at such time as the Common Stock is above the Suspension Price, provided the dates for the Pricing Period for that particular Put are still valid. In the event the Pricing Period has been completed, any shares above the Suspension Price due to Dutchess shall be sold Dutchess by us at the Suspension Price under the terms of this Agreement. The Suspension Price for a Put may not be changed by us once submitted to Dutchess.
There is no minimum amount we can put to Dutchess at any one time. Upon effectiveness of the Registration Statement, the Company shall deliver instructions to its transfer agent to issue shares of common stock to Dutchess free of restrictive legends on or before each closing date.
Pursuant to the Investment Agreement, Dutchess and its affiliates shall not be issued shares of our common stock that would result in its beneficial ownership equaling more than 4.99% of our outstanding common stock.
Dutchess will not engage in any "short-sale" (as defined in Rule 200 of Regulation SHO) of our common stock at any time during this Agreement. On January 28, 2015 and as amended on April 1, 2015, we entered into a Registration Rights Agreement with Dutchess requiring, among other things, which we prepare and file with the SEC a Registration Statement on Form F-1 covering the shares issuable to Dutchess under the Investment Agreement.
As per the Investment Agreement, none of Dutchess' obligations thereunder are transferrable and may not be assigned to a third party.
DETERMINATION OF OFFERING PRICE
Dutchess may sell its shares in the over-the-counter market or otherwise, at market prices prevailing at the time of sale, at prices related to the prevailing market price, or at negotiated prices. We will not receive any proceeds from the sale of shares by Dutchess.
We have arbitrarily established the offering price of the common stock for the purpose of this prospectus and it should not be considered to bear any relationship to our assets, book value or net worth and should not be considered to be an indication of our value. No valuation or appraisal has been prepared for our business. Among the factors considered by our management were:
| the market price for our common stock on the OTCQX and CSE; |
| the potential of our properties; |
| our capital structure; |
| the background of our management; |
| the proceeds to be raised by the offering; and |
| our cash requirements relative to our business operations. |
"Dilution" represents the difference between the Offering price of the shares of common stock and the net tangible book value per share of common stock immediately after completion of the Offering. "Net
28
Tangible Book Value" is the amount that results from subtracting total liabilities from total tangible assets. Please refer to the following table presenting the number of shares issued and the corresponding price per share paid before this Offering for more information. Following is a table detailing dilution as of April 27, 2015, to investors if 100%, 75%, 50%, or 10% of the Offering is sold.
| Percent of Offering sold* | 100% | 75% | 50% | 25% | ||||||||||||
| Net Tangible Book Value Per Share Prior to Stock Sale | $ | 0.0287 | $ | 0.0287 | $ | 0.0287 | $ | 0.0287 | ||||||||
| Pro Forma Net Tangible Book Value Per Share After Stock Sale | 0.1030 | 0.0967 | 0.0868 | 0.0692 | ||||||||||||
| Increase in Net Book Value Per Share Due to Stock Sale | 0.0743 | 0.0680 | 0.0581 | 0.0405 | ||||||||||||
| Net Dilution (Purchase Price of $0.1388 less Pro Forma Net Tangible Book Value Per Share) | $ | 0.0358 | $ | 0.0421 | $ | 0.0520 | $ | 0.0696 |
* Table assumes 180,000,000 shares in Offering are sold at $0.1388 per share and Company receives 95% of that Offering amount or $0.1318 per share. Based on 69,667,951 Common Shares of the Company issued and outstanding immediately prior to stock sale and net tangible book value per share as set out in September 2014 audited financial statements.
Trading in our securities is subject to penny stock considerations. Broker-dealer practices in connection with transactions in "penny stocks" are regulated by certain penny stock rules adopted by the SEC.
Penny stocks generally are equity securities with a price of less than $5.00 (other than securities registered on certain national securities exchanges or quoted on the NASDAQ system). Penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from the rules, to deliver a standardized risk disclosure document that provides information about penny stocks and the risks in the penny stock market. The broker-dealer also must provide the customer with current bid and offer quotations for the penny stock, the compensation of the broker-dealer and its salesperson in the transaction, and monthly account statements showing the market value of each penny stock held in the customer's account. The broker-dealer must also make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser's written agreement to the transaction. These requirements may have the effect of reducing the level of trading activity, if any, in the secondary market for a security that becomes subject to the penny stock rules. The additional burdens imposed upon broker-dealers by such requirements may discourage broker-dealers from effecting transactions in our securities, which could severely limit their market price and liquidity of our securities. These requirements may restrict the ability of broker-dealers to sell our common stock and may affect your ability to resell our common stock.
State Securities - Blue Sky Laws
There is a limited public market for our common stock, and there can be no assurance that any market will develop in the foreseeable future. Transfer of our common stock may also be restricted under the securities regulations or laws promulgated by various states and foreign jurisdictions, commonly referred to as "Blue Sky" laws. Absent compliance with such individual state laws, our common stock may not be traded in such jurisdictions. Because the securities registered hereunder have not been registered for resale under the Blue Sky laws of any state, the holders of such shares and persons who desire to purchase them in any trading market that might develop in the future, should be aware that there may be significant
29
state Blue Sky law restrictions upon the ability of investors to sell the securities and of purchasers to purchase the securities. Accordingly, investors may not be able to liquidate their investments and should be prepared to hold the shares of our common stock for an indefinite period of time.
Selling security holders may contact us directly to ascertain procedures necessary for compliance with Blue Sky Laws in the applicable states relating to sellers and/or purchasers of our shares of common stock.
We intend to apply for a listing in Mergent, Inc. Securities Manual which, once published, will provide us with "manual" exemptions (as described below) in approximately 33 states as indicated in CCH Blue Sky Law Desk Reference at Section 6301 entitled "Standard Manuals Exemptions."
Thirty-three states have what is commonly referred to as a "manual exemption" for secondary trading of securities such as those to be resold by selling security holders under this prospectus. In these states, so long as we obtain and maintain a listing in Mergent, Inc. or Standard and Poor's Corporate Manual, secondary trading of our common stock can occur without any filing, review or approval by state regulatory authorities in these states. These states are Alaska, Arizona, Arkansas, Colorado, Connecticut, District of Columbia, Florida, Hawaii, Idaho, Indiana, Iowa, Kansas, Maine, Maryland, Massachusetts, Michigan, Mississippi, Missouri, Nebraska, New Jersey, New Mexico, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Rhode Island, South Carolina, Texas, Utah, Washington, West Virginia and Wyoming. We cannot secure this listing, and thus this qualification, until after the Registration Statement, of which this prospectus forms a part, is declared effective. Once we secure this listing, secondary trading can occur in these states without further action.
We currently do not intend to and may not be able to qualify our common stock for resale in other states which require shares to be qualified before they can be resold by our security holders.
Limitations Imposed by Regulation M
Under applicable rules and regulations under the Securities Exchange Act of 1934, as amended, (the "Exchange Act") any person engaged in the distribution of the shares may not simultaneously engage in market making activities with respect to our common stock for a period of two business days prior to the commencement of such distribution. In addition and without limiting the foregoing, each selling security holder will be subject to applicable provisions of the Exchange Act and the associated rules and regulations thereunder, including, without limitation, Regulation M, which may limit the timing of purchases and sales of shares of our common stock by the selling security holders. We will make copies of this prospectus available to the selling security holders and will inform them of the need for delivery of copies of this prospectus to purchasers at or prior to the time of any sale of the shares offered hereby. We assume no obligation to so deliver copies of this prospectus or any related prospectus supplement.
SELECTED ANNUAL FINANCIAL INFORMATION
The selected consolidated financial data set forth below as of and for the years ended September 30, 2014, September 30, 2013 and September 30, 2012 is derived from our audited consolidated financial statements, which have been prepared in accordance with International Financial Reporting Standards ("IFRS") as issued by the International Accounting Standards Board ("IASB") and are presented elsewhere in this prospectus.
You should read the following selected consolidated financial data in conjunction with, and it is qualified in its entirety by reference to our consolidated financial statements and the related notes appearing
30
elsewhere in this prospectus and other information provided in this prospectus, including "Management's Discussion and Analysis of Financial Condition and Results of Operations." The historical results set forth below are not necessarily indicative of the results to be expected in future periods.
| �� | For the years ended | ||||||
| September 30, 2014 | September 30, 2013 | September 30, 2012 | |||||
| Statements of Net Loss and Comprehensive Loss: | |||||||
| Total revenue | $ | 7,720 | $ | 17,448 | $ | - | |
| Research expenditure | 129,057 | 123,559 | 124,308 | ||||
| Total operating expenses | 7,378,559 | 1,060,957 | 1,016,571 | ||||
| Other expenses (income) | 280,562 | 58,982 | (24,182) | ||||
| Net loss and comprehensive loss | 7,611,540 | 1,102,491 | 992,389 | ||||
| Basic and diluted loss per common share | $ | 0.16 | $ | 0.04 | $ | 0.09 | |
(Expressed in Canadian Dollars)
| For the years ended | |||||||
September 30, 2014 | September 30, 2013 | September 30, 2012 | |||||
| Statements of Financial Position: | |||||||
| Cash | $ | 135,171 | $ | 5,327 | $ | 5,499 | |
| Intangible assets | 2,641,083 | 1,074,161 | 752,964 | ||||
| Total assets | 5,903,803 | 1,629,346 | 1,325,025 | ||||
| Total liabilities | 1,260,767 | 1,071,329 | 701,558 | ||||
| Shareholders' equity | 4,043,225 | 558,017 | 623,467 | ||||
(Expressed in Canadian Dollars)
31
MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATION
The following discussion and analysis should be read in conjunction with "Selected Consolidated Financial Data" and our consolidated financial statements and the related notes included elsewhere in this prospectus. This discussion contains forward-looking statements that are based on our management's current expectations, estimates and projections for our business, which are subject to a number of risks and uncertainties. Our actual results may differ materially from those anticipated in these forward-looking statements as a result of many factors, including those set forth under "Special Note Regarding Forward-Looking Statements" and "Risk Factors."
Overview
Abattis is a specialty biotechnology company who intends with capabilities, through its wholly owned subsidiaries, of producing, licensing and marketing proprietary ingredients and formulas for use in the Medical Marijuana, BioPharma, Neutraceutical, Cosmetic and Animal Nutrition markets. The Company's strategic plan entails a fully integrated bioceutical company that cultivates, extracts, develops and markets proprietary, natural health and wellness products derived from Cannabis that address unmet health and wellness needs and chronic illnesses through targeted nutritional and medicinal support. It has a deep pipeline of proprietary products ready for sale in high growth areas of the Functional Foods and Supplements business. The Company's wholly owned subsidiaries have the capacity to refine and bring to market a portfolio of proprietary neutraceutical formulations of botanical agents with a demonstrated ability to inhibit the growth of, and reduce the severity and duration of, various strains of human influenza and other viral diseases. Continuing research is being conducted by the Company. The Company has identified targeted channels, to market and license proprietary patent pending antiviral products, throughout the world.
We are considered a development-stage company and have only a limited operating history. Since our inception in 2009, we have devoted substantially all of our resources to identify, acquire and develop our nutraceutical and bioceutical product candidates and providing general and administrative support for these operations. We have funded our operations to date primarily from the issuance and sale of common stock.
We generated $7,720 in revenue for the year ended September 30, 2014 ($17,448 - September 30, 2013) and have incurred net losses since inception. Our net losses were $7,611,540 and $1,102,491 for the year ended September 30, 2014 and September 30, 2013, respectively. As of September 30, 2014, we had an accumulated deficit of $9,108,966. Substantially all of our net losses have resulted from costs incurred in connection with our share based payments, research and development programs and from general and administrative expenses associated with our operations. Our cash and short-term available-for-sale investment balances at September 30, 2014 totaled $1,084,863.
The Company's head office is located at Suite 1040 - 885 West Georgia Street, Vancouver, British Columbia, V6C 3H1, and the Company's Canadian operating facility is located at 104 - 9295 198th Street, Langley, British Columbia.
Highlights and Performance Summary
On October 31, 2013, the Company submitted its application to Health Canada to become a licensed producer under the new MMPR. In order to be granted a producer license from Health Canada, an applicant must have a facility that conforms to government regulations concerning security, cleanliness, traceability, as well as a workforce that has passed an enhanced security screening. In addition to its existing proposed facilities, the Company continues to pursue additional financially viable locations and partnerships that offer synergies and opportunities for growth. |
32
| On November 5, 2013, the Company presented its Vertical Growing Technology to Arcview Group in Seattle, Washington, which was well received by some of the top executives in the American medical marijuana business. |
| On November 13, 2013, the Company filed an international PCT patent application on its novel supplement technology that increases nitric oxide levels of tissue in mammalian patients. |
On November 19, 2013, the Company announced the appointment of Mr. Rene David as its Chief Financial Officer ("CFO") and Chief Operating Officer ("COO"). Mr. Rene David has been valuable in helping the Company recent milestones and his vast business experience will be invaluable as the Company moves into the approval stages of the Health Canada licenses. |
| On December 6, 2013, the Company issued 3,500,000 common shares with a fair value of $87,500 to settle a trade payable of $105,000 with the CEO of the Company. |
| On December 11, 2013, the Company extended the expiry date of 2,100,000 share purchase warrants previously issued on December 12, 2012 from December 12, 2013 to December 12, 2014. |
On December 18, 2013, the Company's wholly owned subsidiary, Animo Wellness Corporation doing business as Medical Marijuana Labs ("MMLC"), signed a five-year lease with PurGenesis Technologies, Inc. for the lease of approximately 5,000 square feet of lab and production space at a cost of $120,000 in annual gross rent. Amino Wellness Corporation changed its name to iJuana Cannabis Inc. on January 28, 2014. |
On December 23, 2013, Northern Vine Canada Inc. submitted its application to Health Canada to obtain a Controlled Substance License for handling, analytics and extraction of Marihuana. On February 20, 2014, the application was reviewed and assessed by the Office of Controlled Substances of Health Canada and there were only three deficiencies in the application that needed to be addressed. |
On January 16, 2014, the Company was added to The Marijuana IndexTM. The index's website, mmj-index.com, states: "The Marijuana IndexTM (AKA 'The Medical Marijuana IndexTM, or 'The Cannabis Index') is the first and only registered equity tracking index which monitors the performance, news and general pulse of qualified marijuana stocks or cannabis stocks." |
| On January 28, 2014, the Company issued 625,000 common shares with a fair value of $31,250 to settle a trade payable of $31,250 with the CFO of the Company. |
| On January 28, 2014, the Company issued 1,500,000 common shares with a fair value of $210,000 to settle a trade payable of $75,000 with the CEO of the Company. |
| On February 5, 2014, the Company was notified by the U.S. Patent and Trademark Office that certain claims of its patent pending method for treating and reducing the risk of avian influenza in poultry were allowable. The resulting patent is expected to be issued in the 4th quarter of 2014. |
On February 11, 2014, the Company completed a non-brokered private placement of 12,000,000 units at a price of $0.05 per unit for gross proceeds of $600,000. Each unit consists of one common share and one-half share purchase warrant of the Company. Each share purchase warrant entitles the holder to purchase one additional common share at a price of $0.10 per share for a period of one year. |
On February 21, 2014, the Company, through its wholly owned subsidiary, Biocube Green Grow Systems Corp., which was incorporated on February 12, 2014, acquired five-year Exclusive World Wide Distribution Rights to Jiangsu Jiahui New Material Co Ltd.'s innovative MgO ("Magnesium Oxide") products for use in any building or facility designed to cultivate botanicals. The Company paid 100,000 shares as consideration and 10,000 common shares as a finder's fee. Total fair value of these 110,000 common shares was $18,700. |
33
On March 7, 2014, the Company acquired a 51% membership interest in Phytalytics LLC in exchange for a cash payment of US$20,000 ($22,196) to Phytalytics LLC. In addition, the Company issued 827,657 common shares to consultants of Phytalytics LLC that had a fair value of $579,360. This fair value of the shares was included in the determination of the purchase of the 51% membership interest. On May 13, 2014, Phytalytics LLC opened an analytical laboratory in Kirkland, WA. |
On March 12, 2014, the Company through its wholly owned subsidiary, Biocube Green Grow Systems Corp., acquired all of the organic and hydroponic fertilizer and nutritional formulas owned by Green-Gro Garden Products Ltd. for consideration of 300,000 common shares and 15,000 common shares as finder fees. Total fair value of the 315,000 common shares was $252,000. |
| On March 12, 2014, the Company sent its newly designed Biocube drawings to China to have its first order manufactured. |
On March 17, 2014, the Company completed a non-brokered private placement of 5,333,331 units at a price of $0.45 per unit for gross proceeds of $2,399,999. Each unit consists of one common share and one share purchase warrant of the Company to purchase one additional common share at a price of $0.50 per share for a period of 18 months, subject to acceleration terms. |
| On March 31, 2014, the Company incorporated a wholly owned subsidiary Abattis Bioceuticals International Inc. in the United States. |
| On April 1, 2014, the Company issued 43,440 common shares to a law firm, of which a director is a one-third partner, to settle a trade payable of US $62,554. |
| On April 7, 2014, the Company retained TDM Financial as its full service investor relations firm. Pursuant to the agreement, the Company will pay US $25,000 consulting fee in common shares to TDM Financial. |
| On April 8, 2014, the Company incorporated another subsidiary National Access Pharmacy Corp. in Canada. |
On April 10, 2014, the Company, through its wholly owned subsidiary, Northern Vine Canada Inc. ("Northern Vine"), entered into a share exchange agreement with Experion Biotechnologies Inc. ("Experion"), whereby Experion and Northern Vine exchanged 25% of each parties' issued and outstanding common shares. Abattis maintains a 75% ownership in Northern Vine. Experion is located in Greater Vancouver and is nearing the completion of its Marijuana for Medical Purposes Regulations "MMPR" licence application with Health Canada. |
| On April 28, 2014, 40,000 warrants were exercised for $10,000 in cash. |
| On May 1, 2014, the Company issued 33,669 common share at deemed price of $0.99 per share as consideration for services provided from two consulting agreements with arm's length parties TDM Financial and ThinkSharp Inc. |
On May 2, 2014, the Company, through its wholly owned subsidiary, Abattis Bioceuticals International Inc., acquired 34% interest in Instant Payment Systems LLC ("IPS"), a U.S. entity based in Washington State, in consideration for 200,000 common share at a deemed price of $1.15 per share and $100,000 cash. Under the terms of the agreement, Abattis will receive a 60% economic interest with the responsibility of being the marketing and sales arm of the organization. |
On May 16, 2014, the Company changed auditors to MNP LLP, Chartered Accountants. |
34
On May 26, 2014, the Company leased a 16,200 square foot facility zoned for medical marijuana near Vancouver, BC, with the potential to expand up to 254,000 square feet. The facility was sub-leased from Crimson Opportunities Ltd., which is owned by the Company's CFO. Until it secures a license, the Company will use the facility to manufacture and warehouse its proprietary Biocube systems. |
| On June 2, 2014, the Company issued 35,665 common shares to TDM Financial. Total fair value of the 35,665 common shares was US$24,609 ($26,392). |
| On June 17, 2014, the Company issued 50,000 common shares to William Fleming and 50,000 common shares to Manny Montenegrino. Total fair value of the 100,000 common shares was US$43,000 ($38,000). |
| On June 19, 2014, the shareholders re-elected the previous Board of Directors, and added two new directors: Mr. William Fleming and Mr. Emanuel Montenegrino. |
| On June 23, 2014, the Company announced that its International Patent Application for refining the optimum natural sources of nitric oxide in the human body had been published and was publicly available for inspection. |
| On July 2, 2014, the company announced that Brazos Minshew was appointed the President of BioCell Labs, a wholly-owned subsidiary of Abattis Bioceuticals Corp. |
| During the year ended September 30, 2014, 9,217,200 incentive stock options with an estimated fair value of $4,701,541 were granted by the Company to certain directors, officers and consultants of the Company to purchase up to an aggregate of 9,217,200 common shares of the Company. |
On July 31, 2014, the Company has entered into a sub-licensing agreement with its subsidiary, Biocube Green Grow Systems Corp. ("Biocube"), for the non-exclusive use of Vertical Design Ltd.'s ("VDL") vertical growing systems for which Abattis owns certain exclusive worldwide rights. Under the terms of the agreement, Biocube has agreed to pay Abattis certain licensing fees and ongoing royalties for products grown and sold within the pharmaceutical, nutraceutical, cosmetic, and wellness markets. |
On August 5, 2014, the Company entered into a US$5 million standby equity financing agreement with Kodiak Capital Group, LLC ("Kodiak"), a Newport Beach-based institutional investor. The Company has agreed to file a registration statement with the SEC covering the Company's shares that may be issued to Kodiak under this financing. After the SEC has declared the registration statement related to the transaction effective, the Company has the right at its sole discretion over a period of one year to sell up to US$5 million of common shares to Kodiak. The Company has agreed to pay Kodiak a 5% commitment fee in shares. Of these shares, 277,373 shares shall be held in escrow in accordance with an escrow agreement to be released if and when the Company accessed the facility. On August 7, 2014, 243,460 shares were issued to Kodiak with a deemed value of US$102,253 ($111,991). The Company terminated this agreement in writing on January 26, 2015. |
On August 6, 2014, Biocube, its wholly owned subsidiary, entered into a worldwide non-exclusive licensing agreement with TerraSphere Systems LLC ("TerraSphere") for the company's proprietary and patented vertical farming technology. The TerraSphere license complements Biocube's previously announced sublicensing agreement for VDL's vertical farming technology. |
| On September 8, 2014, Phytalytics LLC ("Phytalab.com"), a 51% controlled US subsidiary of ABI, has received provisional certification from the Washington State Liquor Control Board. This milestone enables the lab to legally offer service to the new legal Cannabis industry. |
35
| On September 16, 2014, the Company announced that Guy Dancosse joined the Board effective as an independent Director. |
| On November 17, 2014, Jaouad Fichtali joined the Company as the Chief Technology Officer. |
| On January 15, 2015, the Company announced a manufacturing partnership with Empirical Labs., of Ft. Collins, Colorado. |
On January 27, 2015, Mike Withrow resigned as the President, CEO and Director of the Company to assume the role of International Business Advisor for Abattis, effective February 1, 2015. William (Bill) Fleming was appointed the CEO of the Company, effective February 1, 2015. Jaouad Fichtali resigned as the Chief Technology Officer to assume the role of Extraction Advisor of Abattis, effective as of January 27, 2015. |
On January 28, 2015, the Company entered into a US$25 million Standby equity financing agreement with Dutchess Opportunity Fund, II, LP ("Dutchess"), a Boston, MA-based institutional investor. The Company has agreed to file a registration statement with the U.S. Securities and Exchange Commission ("SEC") covering the Abattis shares that may be issued to Dutchess under this financing. After the SEC has declared the registration statement related to the transaction effective, the Company has the right at its sole discretion over a period of 36 months to sell up to US$25 million of common shares to Dutchess. The Company plans to access and use these funds in the event of an acquisition opportunity and for expansion in the US. |
On February 4, 2015, Michael Bessler was appointed as corporate communications consultant for the Company. |
On February 17, 2015, the Company, through its wholly-owned subsidiary BioCell, launched 8 phytocannabinoid formulas at CannaCon Seattle 2015. One formula consists of a beverage known as ECS Supreme, which contains phytocannabinoids and Abattis' patented nitric oxide blend. Other condition-specific cannabinoid formulas address nausea, anxiety and traumatic brain injury. |
| On February 18, 2014, Robert Hedley resigned as a director of the Company. |
On March 19, 2015, the Company completed a non-brokered private placement of 2,365,072 units at a price of $0.13 per unit for gross proceeds of $307,459. Each unit consists of one common share and one share purchase warrant of the Company to purchase one additional common share at a price of $0.18 per share for a period of 18 months, subject to acceleration terms. |
Results of Operation
Year Ended September 30, 2014 Compared to Year Ended September 30, 2013
The Company incurred a net loss and comprehensive loss of $7,611,540 during the year ended September 30, 2014, an increase of $6,509,049 when compared with the loss of $1,102,491 for year ended September 30, 2013.
Revenue
The Company generated $7,720 of revenue for the year ended September 30, 2014, as compared to $17,448 for the year ended September 30, 2013. We do not expect to receive any revenues from any product candidates that we develop until we obtain regulatory approval and commercialize our products or enter into collaborative agreements with third parties.
Research and Development Expenses
Research and development expenses were $129,057 for the year ended September 30, 2014 compared to $123,559 for the year ended September 30, 2013, an increase of 4%.
36
Management and Consulting Fees
Management and consulting fees, were $804,755 for the year ended September 30, 2014, compared to $313,433 for the year ended September 30, 2013, an increase of 157%. This increase is attributable to an increase in fees paid in respect of general business consulting services.
General and Administrative Expenses
General and administrative expenses were $369,627 for the year ended September 30, 2014, compared to $83,906 for the year ended September 30, 2013, an increase of 340%. The increase in expenses is primarily attributable to increased professional services and office expenses.
Legal Fees
Legal fees were $722,707 for the year ended September 30, 2014, compared to $167,663 for the year ended September 30, 2013, an increase of 331%. The increase in legal fees in 2014 is attributable to increased professional services.
Accounting and Audit Fees
Accounting and audit were $116,813 for the year ended September 30, 2014, compared to $64,543 for the year ended September 30, 2013. Accounting and audit fees were higher due to implementation of new accounting software, additional work performed for the acquisitions, and the employment of additional accounting staff.
Liquidity and Capital Resources
Since inception, we have funded our operations primarily through the sale of equity securities and convertible notes to investors in private placements.
As at September 30, 2014, the Company had a cash balance of $135,171 (September 30, 2013 - $5,327). During year ended September 30, 2014, cash used for operating activities was $2,108,551 (September 30, 2013 - $230,151); $4,276,505 was received from financing activities (September 30, 2013 - $231,979), and cash used for investing activities was $2,038,110 (September 30, 2013 - $2,000).
The Company continues to utilize its cash resources to fund its administrative requirements and product development. As the Company does not currently generate revenue, cash balances, unless replenished by capital fundraising, will continue to decline as funds are utilized to conduct its operations.
In order to fund the Company's ongoing operational needs, the Company will need funding through equity or debt financing, joint venture arrangements or a combination thereof. The Company's operations to date have been financed by the issuance of its common shares, debt instruments and government assistance. The Company continues to seek capital through various means including the issuance of equity and debt. While the Company has been successful in raising funds in the past, there is no assurance that it will continue to do so in the future or that it will be available on a timely basis or on terms acceptable to the Company.
The financial statements have been prepared on a going concern basis which assumes that the Company will be able to realize its assets and discharge its liabilities in the normal course of business. The continuing operations of the Company are dependent upon its ability to continue to raise adequate financing and to commence profitable operations in the future. If the Company is unable to obtain sufficient funding, the ability of the Company to meet its obligations as they come due and, accordingly,
37
the appropriateness of the use of accounting principles applicable to a going concern will be in significant doubt. The Company has incurred $9,108,966 in losses from inception including a net loss of $7,611,540 for the year ended September 30, 2014 (September 30, 2013 - $1,102,491), and has a working capital of $695,889 as at September 30, 2014 (September 30, 2013 - working capital deficiency of $999,440).
Cash Flows
Net Cash Used in Operating Activities
Net cash used in operating activities was $2,108,551 and $230,151 in each of the years ended September 30, 2014 and 2013, respectively. Our net losses in each of the years were offset primarily by non-cash expenses and by net changes in our working capital.
Net Cash Used in Investing Activities
Net cash used in investing activities was $2,038,110 and $2,000 in each of the years ended September 30, 2014 and 2013, respectively. Investing activities in these years consisted of purchases of term deposits, marketable securities, property and equipment.
Net Cash Provided by (Used in) Financing Activities
Our financing activities have consisted of the issuance of common shares and the sale of common shares and units. Net cash provided by financing activities was $4,276,505 and $231,979 for the years ended September 30, 2014 and 2013, respectively. Financing activities in these years consisted of common shares for cash, modest funding from the Agricultural Foundation and a short term loan from a related party.
Income Tax
At September 30, 2014, the Company has accumulated non-capital losses for income tax purposes of approximately $5,711,413 (September 30, 2013 - $2,974,000). If unused, the non-capital losses will expire as follows:
| Year of expiration | Non-capital loss | |
| 2028 | $ | 65,764 |
| 2029 | 64,815 | |
| 2030 | 609,226 | |
| 2031 | 682,635 | |
| 2032 | 834,223 | |
| 2033 | 904,700 | |
| 2034 | 2,550,050 | |
| Total | $ | 5,711,413 |
The Company has net operating loss carryforwards of approximately $108,204 which may be carried forward to apply against future year income tax for US tax purposes.
| 2034 | $ | 108,204 |
| Total | $ | 108,204 |
38
Critical Accounting Policies
Our consolidated financial statements are prepared in accordance with IFRS as issued by the IASB. The preparation of our financial statements requires us to make estimates, judgments and assumptions that can affect the reported amounts of assets and liabilities, disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. We base our estimates, judgments and assumptions on historical experience and other factors that we believe to be reasonable under the circumstances. Materially different results can occur as circumstances change and additional information becomes known. Besides the estimates identified above that are considered critical, we make many other accounting estimates in preparing our financial statements and related disclosures. See Note 2 to our audited consolidated financial statements presented elsewhere in this prospectus for a description of the significant accounting policies that we used to prepare our consolidated financial statements. The critical accounting policies that were impacted by the estimates, judgments and assumptions used in the preparation of our consolidated financial statements are discussed below.
Share-Based Compensation
Option Valuations
The fair value of employee options is measured at the option's grant date, and the fair value of non-employee options is measured at the date when goods or services are received. The fair value of each tranche of options granted which do not vest immediately on grant, is recognized over the period during which each tranche of options vest. The fair value of the options granted is measured using the Black-Scholes option pricing model taking into account the terms and conditions upon which the options were granted. At each financial position reporting date, the amount recognized as an expense is adjusted to reflect the actual number of share options that are expected to vest.
Share-based compensation expense is credited to the equity settled share-based payment reserve. If the options are later exercised, their fair value is transferred from the reserve to share capital.
The changes in options during the year ended September 30, 2014 are as follows:
| Number Outstanding | Weighted Average Exercise Price | ||||
| Balance, beginning of year | 2,575,000 | $ | 0.17 | ||
| Granted | 9,217,200 | $ | 0.87 | ||
| Exercised | (3,131,000) | $ | 0.17 | ||
| Expired | (605,000) | $ | 0.13 | ||
| Cancelled | (2,613,100) | $ | 1.99 | ||
| Balance, end of year | 5,443,100 | $ | 0.49 |
39
Quantitative and Qualitative Disclosure about Market Risk
Foreign Currency Risk
Our results of operations and cash flows are affected by fluctuations due to changes in foreign currency exchange rates. Approximately half of our expenses were denominated in U.S. dollars in 2013, with the remainder of our expenses being denominated in Canadian dollars. Accordingly, changes in the value of the Canadian dollar relative to the U.S. dollar in 2014 has affected the amounts recorded on our consolidated statements of operations in 2014. We expect that the denominations of our revenue and expenses in 2014 will be consistent with what we experienced in 2014.
The following table presents information about the changes in the exchange rates at noon of the Canadian dollar against the U.S. dollar in 2014 as provided by the Bank of Canada:
| Years Ended September 30, | |||||
| 2014 | 2013 | 2012 | 2011 | 2010 | |
| Average exchange rate CDN$ per US$1.00 | 1.0831 | 1.0155 | 1.0074 | 0.9866 | 1.0407 |
| Average exchange rate US$ per CDN$1.00 | 0.9233 | .9847 | 0.9927 | 1.0136 | 0.9609 |
The high and low exchange rates between the Canadian dollar and the U.S. dollar for each of the six months ended March 31, 2015 are as follows:
| Exchange rate CDN$ per US$1.00 | |||||||
| Month | Low | High | |||||
| March 2015 | 1.2440 | 1.2803 | |||||
| February 2015 | 1.2403 | 1.2635 | |||||
| January 2015 | 1.1728 | 1.2717 | |||||
| December 2014 | 1.1344 | 1.1643 | |||||
| November 2014 | 1.1236 | 1.1426 | |||||
| October 2014 | 1.1136 | 1.1289 | |||||
A 10% increase or decrease in the value of the Canadian dollar against the U.S. dollar would have decreased or increased our net loss for the year by approximately $761,154 in 2014.
We have not entered into any hedging arrangements with financial institutions and do not see a need to do so in the upcoming fiscal year.
Other Market Risks
We do not believe that we have material exposure to interest rate risk due to the fact that we have no long-term borrowings.
We do not believe that we have any material exposure to inflationary risks.
New and Revised Financial Accounting Standards
The Jumpstart our Business Startups Act ("JOBS Act") permits emerging growth companies such as us to delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
40
Recently Issued and Adopted Accounting Pronouncements
There are no recently adopted accounting pronouncements that have a material effect on our company.
Off-balance sheet arrangements
There are no off-balance sheet arrangements that have or are reasonably likely to have a current or future effect on our financial condition, changes in financial condition, revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that is material investors.
Subsequent events
We have evaluated subsequent events from our September 30, 2014 audited financial statements through to April 27, 2015, and report the following events:
| On November 3, 2014, 33,334 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On November 4, 2014, Stone Throw exercised 500,000 warrants at $0.10 per share for gross proceeds of $50,000. |
On November 14, 2014, New Horizons Inc. exercised 150,000 warrants at $0.10 per share for gross proceeds of $15,000. | ||
| On November 18, 2014, Michael Sweeney exercised 25,000 warrants at $0.10 per share for gross proceeds of $2,500. |
| On November 20, 2014, Michael Sweeney exercised 75,000 warrants at $0.10 per share for gross proceeds of $7,500. |
| On November 27, 2014, New Horizons Inc. exercised 42,000 warrants at $0.10 per share for gross proceeds of $4,200. |
| On December 1, 2014, 14,535 common shares were issued to Jaouad Fichatli for consulting services to the Company. |
| On December 1, 2014, 40,000 common shares were issued to ThinkSharp Inc., a consulting firm. |
| On December 1, 2014, 33,334 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On December 3, 2014, New Horizons Inc. exercised 40,000 warrants at $0.10 per share for gross proceeds of $4,000. |
On December 29, 2014, the Company obtained a obtained a preliminary injunction from the Washington state court in King County against Herbal Analytics, LLC, James Baxter, Kaleb Lund, Lauren Hilty, and Erin Leary, Affinor Growers, LLC, and Nicholas Brusatore. Affinor Growers, LLC is a wholly owned subsidiary of defendant Affinor Growers, Inc. (collectively the "Defendants"). According to the conclusions of law, the Defendants shall cease and desist from any and all use of PhytaLabs trade secrets and confidential information and documents; (b) are restrained from copying, transferring, using or disclosing to any other person or entity any documentation taken from PhytaLab; (c) shall retain and preserve all existing documents and files that mention, refer to, or are derived from PhytaLab, PhytaLab and Abattis' customers, or PhytaLab and Abattis' prospective customers; and (d) shall keep a detailed, complete, and accurate accounting of its business operations. |
| On January 2, 2015, 283,334 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On January 2, 2015, New Horizons Inc. exercised 68,000 warrants at $0.10 per share for gross proceeds of $6,800. |
41
| On January 2, 2015, 18,382 common shares were issued to Jaouad Fichatli for consulting services to the Company. |
On January 15, 2015, the Company entered into a manufacturing partnership with Empirical Labs, a nutraceutical manufacturing facility. This relationship with Empirical Labs will allow the Company the exclusive license rights for cannabidiol rich hemp oil products delivered directly to licensed physicians and dispensaries. |
| On January 27, 2015, the Company granted 175,000 stock options to certain of its directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. |
On January 28, 2015, the Company entered into a US$25 million Standby equity financing agreement with Dutchess Opportunity Fund, II, LP ("Dutchess"), a Boston, MA-based institutional investor. The Company has agreed to file a registration statement with the U.S. Securities and Exchange Commission ("SEC") covering the Abattis shares that may be issued to Dutchess under this financing. After the SEC has declared the registration statement related to the transaction effective, the Company has the right at its sole discretion over a period of 36 months to sell up to US$25 million of common shares to Dutchess. The Company plans to access and use these funds in the event of an acquisition opportunity and for expansion in the US. |
VDL sent a letter advising they were terminating the license agreement by citing that the Company failed to comply with certain terms and conditions included in the license agreement. The Company believes that the terms in the license agreement have been followed; as a result, the license agreement should be valid. The Company intends to continue to honor the agreement and make any payments or provide any information required under the license. The Company provides for costs related to contingencies when a loss is probable and the amount is reasonably determinable. In the opinion of management, no grounds exist that justify the termination of the license agreement. It is the opinion of management, based in part on advice of legal counsel, that the ultimate resolution of the termination of the license agreement is undeterminable; therefore there has been no provision made with respect to the license in the consolidated financial statements for the year ended September 30, 2014. |
| The Company was granted a patent which was originally acquired on April 16, 2009 from PRB and Pacific Bio. |
One of the directors of the Company forgave $235,418 in amounts owing to him for consulting services in exchange for cash settlement of $32,000. | ||
On February 3, 2015, 55,926 common shares were issued to certain consultants of the Company. | ||
On February 11, 2015, 50,000 warrants were exercised at a price of $0.10 per share for gross proceeds of $5,000. | ||
On February 11, 2015, 250,000 warrants were exercised at a price of $0.10 per share for gross proceeds of $25,000. | ||
On March 4, 2015, 539,498 common shares were issued to certain directors, officers and consultants of the Company. | ||
On March 6, 2015, the Company granted 125,000 stock options to certain of its directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. | ||
On March 19, 2015, the Company completed a non-brokered private placement of 2,365,072 units at a price of $0.13 per unit for gross proceeds of $307,459. Each unit consists of one common share and one share purchase warrant of the Company to purchase one additional common share at a price of $0.18 per share for a period of 18 months, subject to acceleration terms. |
42
On April 10, 2015, The Company amicably resolved and dismissed the Washington litigation against Affinor Growers, LLC and Nick Brusatore. The Company will continue to pursue its claims against Herbal Analytics, LLC, James Baxter, Kaleb Lund, Lauren Hilty and Erin Leary. | ||
On April 14, 2015, 86,436 common shares were issued to certain consultants of the Company. | ||
On April 14, 2015, 72,414 common shares were issued to Allan Echino to settle a debt. |
Outstanding share data
At April 27, 2015, we had 69,667,951 issued and outstanding Common Shares, 3,123,100 stock options and 8,948,903 share purchase warrants outstanding. On a fully diluted basis, 81,739,954 common shares were outstanding.
Corporate History
The Company was incorporated as Sinocan Capital Group Inc. under the Company Act (British Columbia) on June 30, 1997 for the purpose of listing on the predecessor to the TSX Venture Exchange as a Capital Pool Corporation as defined in the TSX Venture Exchange Policy 2.4. The Company was unsuccessful in finding a suitable target and was delisted from the TSX Venture Exchange.
On September 29, 1997, the Company changed its name to Sican Ventures Inc.
Beginning in April 2009, we began our entry into the bio-pharmaceutical and nutraceutical industry.
On April 16, 2009, the Company entered into an agreement with PRB Pharmaceuticals, Inc. ("PRB") and Pacific Bio-Pharmaceuticals, Inc. ("Pacific Bio") for the purchase of their interest in patents and intellectual property related to antiviral products designed to prevent avian influenza in humans and poultry. The Company issued 5,000,000 (pre-consolidation 25,000,000) common shares and assigned a value of $500,000 for this acquisition. The shares issued by the Company have been distributed by PRB and Pacific Bio to the shareholders of those companies.
The Antiviral formulations were developed and designed to inhibit growth over multiple segments of the influenza virus life cycle - viral binding, encoding, replication, and release. To date, the Company's research has shown the formulations to be highly effective in reducing the severity and duration of influenza illness symptoms in humans. The Company had previously planned to build its business by marketing the Antiviral formulations throughout the world.
On September 14, 2009, the Company amended its articles of incorporation to change its name to Abattis Biologix Corporation to reflect its new business and changed the authorized share capital to set no maximum on the number of common shares without par value.
The Company was listed and began trading on the Canadian Securities Exchange (formerly the Canadian National Stock Exchange) ("CSE") on December 23, 2010. On September 5, 2012, the Company changed its name to Abattis Bioceuticals Corp. The Company is a reporting issuer in British Columbia, Alberta and Ontario.
In March 2013, we began our entry into the medicinal marijuana business, which is now the focus of our business plan.
43
The Company's head office is located at 1040 - 885 West Georgia Street, Vancouver, British Columbia, V6C 3H1, and the Company's Canadian operating facility is located at 104 - 9295 198th Street, Langley, British Columbia.
Overview
Abattis is a specialty biotechnology company who intends with capabilities through its wholly owned subsidiaries of cultivating, licensing, and marketing proprietary ingredients, bio-similar compounds, patented equipment and consulting services to medicinal markets in North America. Management believes the Company is positioned to capitalize on the fast growing trend toward marijuana legalization in the United States and for medicinal use in Canada and international jurisdictions by supplying and collaborating with companies to employ its vertical cultivation systems, extraction equipment/technology, and strategic marketing support to licensed growers. The Company also has an extensive pipeline of high-quality products and intellectual property for the rapidly expanding botanical drug market. We follow strict Standard Operating Protocols and adhere to the laws of Canada and Foreign Jurisdictions.
Abattis aims to become one of the leaders in the growing, testing, and distribution of medical and adult-use cannabis in North America. Through its network of subsidiaries and partners, the Company has developed an innovative GDERS (grow, dry, extract, refine, sell) strategy spanning the entire industry supply chain from seed to sale. These subsidiaries and partners specialize in cultivating, licensing and marketing proprietary ingredients, bio-similar compounds, patented equipment, and consulting services across the continent. The Company follows strict standard operating protocols and adheres to both U.S. and Canadian laws pertaining to the cultivation, testing, and sale of cannabis. As of the date of this prospectus, we have not commenced the actual the production and sale of medical marijuana but are seeking to lay the foundation to commence this business.
We currently have seven subsidiaries through which we plan to operate our business.
Recent Developments
In the first quarter of 2011, the Company acquired Biocell Algae (Immune System Support), a complimentary Natural Health Product to the Anti Viral Flu Intellectual Property and hired a new President and CEO who brought in a new board of directors and that team acquired three new formulations. The formulae are comprised of all-natural ingredients and target and address 1) Flu like symptoms and other viral conditions, 2) migraine headaches (Cognitive) and 3) blood flow to muscles (Cardiovascular and Erectile Dysfunction).
On June 17, 2011, the Company was listed on the OTC Markets Pink Sheets to enable easier access to American Investors. During the 2011 calendar, year management worked to structure the company and identify assets that would be useful in nutraceutical and bioceutical production. In December 2011 the Company entered into an agreement to acquire Northern Vine Canada Inc. (a cGMP nutraceutical production facility in Langley, BC) and the assets with the Sci-Naturals brand and closed the acquisitions in August 2012.
In March 2012, the Company acquired Animo Wellness Corporation, which owns 77 Natural Health Product Licenses issued by Health Canada. These range from (A) Aloe Vera to (Z) Zinc. Amino Wellness Corporation changed its name to iJuana Cannabis Inc. on January 28, 2014.
On July 23 2012, the Company held its AGM and shareholders approved a 5:1 reverse split, and the acquisition of proprietary Flash Freeze Extraction Equipment and a large portfolio of natural health product internet domain names. The Company changed its name to more accurately reflect the nature of
44
its business of bioceuticals and botanical drugs from Abattis Biologix Corporation to Abattis Bioceuticals Corp.
In March 2013, the Company's management decided to focus its efforts on the legal medical cannabis industry following Health Canada's introduction of its Licensed Producer Program on December 15, 2012.
In November 2013, the Company, through its subsidiary, filed its initial MMPR Licensed Producer application with Health Canada and received a list of deficiencies in the application in February 2014. Since then, the Company has been actively working to expand its cannabis-related products and services portfolio, while moving towards securing an MMPR Licensed Producer status.
On September 8, 2014, Phytalytics LLC, a 51% controlled U.S. subsidiary of the Company, received provisional certification by the Washington State Liquor Control Board to legally offer cannabis analysis laboratory services in Washington State. Phytalytics LLC intends to provide quality control testing services for I-502 Market Marijuana Producers and Processors in Washington.
On October 8, 2014, the Company through its wholly owned subsidiary, Northern Vine Canada Inc., Experion Biotechnologies Inc. has received a written update from the Fraser Valley Regional District on the amended zoning regulations for its property which has now been approved for "the use of cultivation, growth, storage, or distribution, testing, or research, of marijuana for medical purposes as lawfully permitted and authorized under the applicable federal or provincial law."
On December 29, 2014, the Company obtained a obtained a preliminary injunction from the Washington state court in King County against Herbal Analytics, LLC, James Baxter, Kaleb Lund and Lauren Hilty (former employees of PhytaLab), and Erin Leary, Affinor Growers, LLC, and Nicholas Brusatore. Affinor Growers, LLC is a wholly owned subsidiary of defendant Affinor Growers, Inc. (collectively the "Defendants"). According to the conclusions of law, the Defendants shall cease and desist from any and all use of PhytaLabs trade secrets and confidential information and documents; (b) are restrained from copying, transferring, using or disclosing to any other person or entity any documentation taken from PhytaLab; (c) shall retain and preserve all existing documents and files that mention, refer to, or are derived from PhytaLab, PhytaLab and Abattis' customers, or PhytaLab and Abattis' prospective customers; and (d) shall keep a detailed, complete, and accurate accounting of its business operations.
On January 15, 2015, the Company announced a manufacturing partnership with Empirical Labs., of Ft. Collins, Colorado.
On February 17, 2015, the Company, through its wholly-owned subsidiary BioCell, launched eight phytocannabinoid formulas at CannaCon Seattle 2015, the nation's largest cannabis expo. One formula consists of a beverage known as ECS Supreme, which contains phytocannabinoids and Abattis' patented nitric oxide blend. Other condition-specific cannabinoid formulas address nausea, anxiety and traumatic brain injury.
On April 10, 2015, The Company amicably resolved and dismissed the Washington litigation against Affinor Growers, LLC and Nick Brusatore. The Company will continue to pursue its claims against Herbal Analytics, LLC, James Baxter, Kaleb Lund, Lauren Hilty and Erin Leary.
45
Emerging Growth Company Status
We qualify as an "emerging growth company" as defined in Section 101 of the JOBS Act as we do not have more than $1,000,000,000 in annual gross revenue and did not have such amount as of September 30, 2014, being the last day of our last fiscal year.
We may lose our status as an emerging growth company on the last day of our fiscal year during which (i) our annual gross revenue exceeds $1,000,000,000 or (ii) we issue more than $1,000,000,000 in non-convertible debt in a three-year period. We will lose our status as an emerging growth company if at any time we are deemed to be a large accelerated filer. We will lose our status as an emerging growth company on the last day of our fiscal year following the fifth anniversary of the date of the first sale of common equity securities pursuant to an effective registration statement.
As an emerging growth company under the JOBS Act, we have elected to opt out of the extended transition period for complying with new or revised standards pursuant to Section 107(b) of the Securities Act. This election is irrevocable.
As an emerging growth company, we are exempt from Section 404(b) of the Sarbanes-Oxley Act of 2002 and Section 14A(a) and (b) of the Exchange Act. Such sections are provided below:
Section 404(b) of the Sarbanes-Oxley Act of 2002 requires a public company's auditor to attest to, and report on, management's assessment of its internal controls.
Sections 14A(a) and (b) of the Exchange Act, implemented by Section 951 of the Dodd-Frank Act, require companies to hold shareholder advisory votes on executive compensation and golden parachute compensation.
As long as we qualify as an emerging growth company, we will not be required to comply with the requirements of Section 404(b) of the Sarbanes-Oxley Act of 2002 and Section 14A(a) and (b) of the Exchange Act.
Geographic and Segment Information
We have one reportable segment, consisting of the research, cultivation, licensing, testing and marketing of medicinal chemicals & botanical products which activities are principally conducted in British Columbia. We reported no material revenues during 2014, 2013 and 2012.
Intercorporate Relationships
We currently have seven subsidiaries through which we plan to operate our business. At April 27, 2015, the Company's subsidiaries are as follows:
Name | Jurisdiction of Incorporation | Principal Activity | % Owned Company Interest |
| Abattis Bioceuticals International Inc. ("ABI") | Washington State | Biotechnology | 100% |
| BioCell Labs Inc. ("BLI") | Canada | Biotechnology | 100% |
| Biocube Green Grow Systems Corp. ("Biocube") | British Columbia | Biotechnology | 100% |
| iJuana Cannabis Inc. ("iJuana") | British Columbia | Holds certain licenses | 100% |
| True Plant Technologies Inc. ("TPL") | British Columbia | Biotechnology | 100% |
| North American BioExtracts Inc. ("NAB") | British Columbia | Biotechnology | 100% |
| Northern Vine Canada Inc. ("N. Vine") | British Columbia | Biotechnology | 75% |
46
On April 10, 2014, the Company through its wholly owned subsidiary, Northern Vine Canada Inc., ("N. Vine") entered into a share exchange agreement with Experion Biotechnologies Inc. ("Experion"). Pursuant to the terms of the agreement, Experion and N. Vine have exchanged 25% of each parties' issued and outstanding common shares. Abattis maintains a 75% ownership in Northern Vine.
On April 7, 2014, the Company through its wholly owned subsidiary Abattis Bioceuticals International Inc. ("ABI") entered into an agreement with Phytalytics LLC ("Phytalytics") to obtain a 51% membership interest in Phytalytics. On April 30, 2014, ABI entered into an agreement with Instant Payment Systems LLC ("IPS"), a Washington State online payment company, to obtain a 34% equity interest in IPS.
The following corporate chart sets forth all of our material subsidiaries:
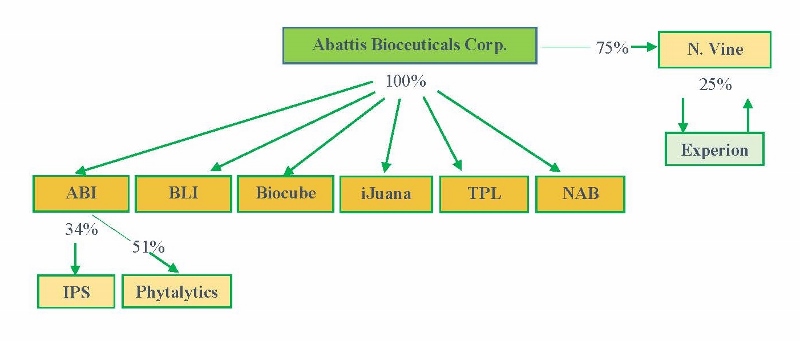
Abattis Bioceuticals International Inc.
ABI was formed to hold and advance the Company's interests in the U.S. It currently holds the Company's interest in IPS and Phytalytics.
BioCell Labs Inc.
BLI is focused on supplying standardized pharmaceutical grade products. BLI has made application to Health Canada to become a licensed producer under Canada's MMPR program and is in the review stage of the process. The company has a term sheet agreement to co-own a $13 million, 17,000 square foot botanical drug facility that it will use to perform quality testing and refining using proprietary water extraction technologies. Currently, the company has 13 proprietary formulations for out-licensing to cosmetic, nutraceutical and pharmaceutical companies.
Biocube Green Grow Systems Corp.
Biocube has exclusive worldwide rights to a building product derived from magnesium oxide known as MgO board, which has applications in all areas of the building and construction industry as a fireproof, antimicrobial, antibacterial, waterproof, and recyclable material.
47
Experion Biotechnologies Inc.
Experion operates a ten-acre property in Mission, British Columbia. Experion has developed strains of cannabis for use across many medical conditions. Experion, in conjunction with their partners, is developing proprietary techniques to derive pharmaceutical grade materials from various sources of biomass. To complement their research, the Company has applied to become a licensed producer under Canada's MMPR and Experion's application has passed the review stage and Experion is awaiting a "Ready-to-Build" letter from Health Canada.
iJuana Cannabis Inc.
iJuana has leased a 27,000 sq. ft. facility in a pre-approved zoned area to become a Licensed Producer under Canada's MMPR. With operations in Squamish, British Columbia, the company aims to become the licensed cannabis growing arm and consumer-facing entity for the Company.
True Plant Technologies Inc.
TPL was incorporated as "National Access Pharmacy Corp." on April 8, 2014 under the Business Corporations Act (British Columbia). On September 23, 2014, National Access Pharmacy Corp. changed its name to "True Plant Technologies Inc." TPL has no operations at this date.
North American BioExtracts Inc.
NAB will focus on cannabis, cannabis derivatives, and ancillary products like vaporizers where legally permitted. NAB aims to use its proprietary extraction technologies to develop customized THC extract oils and CBD extract oils, as well as customized vaporizer flavors to enhance the end user's experience. NAB has no operations as of this date.
Northern Vine Canada Inc.
N. Vine has applied for a Controlled Substance License with Health Canada, with an existing cannabis processing, testing, and packaging facility located in Langley, British Columbia. In addition to these services, the Company will support its operations by providing analytical services for Licensed Producers and charging a fee to produce a Certificate of Analysis. N. Vine will also formulate compositions for sprays, creams, and tinctures that it plans to patent. N. Vine has limited operations as of this date.
Phytalytics LLC
Phytalytics operating in Kirkland, Washington, performs testing services under Washington State's Initiative 502, with a fully functional testing laboratory for nutraceuticals, biopharmaceuticals, and plant-based medicines.
Instant Payment Systems LLC
IPS is a payment systems company. It enables patients to make payments through either a mobile app or loyalty card to purchase medical marijuana, which avoids the many problems associated with using cash to complete transactions. Dispensaries and other medical marijuana providers in the U.S. and Canada can also use the technology to retain purchase information to meet auditing and regulatory compliance requirements. The Company will act as the marketing and sales arm of IPS, signing up new clients in the botanical drug space. IPS has limited operations as of this date.
48
Overall strategy
Development of our Medical Marihuana Business
In March 2013, management of the Company began identifying and evaluating opportunities for entry into the medical marihuana industry in Canada.
Medical marijuana laws have changed in the US and laws are being confirmed in Canada. New markets are continually opening up, and there is a massive need to develop, as plant-based phyto compounds appear to be in huge demand globally. Pharma companies have patents expiring and the Abattis/Vertical capabilities intends to create solutions to these market needs. 2014 and 2013 were milestone years for Abattis in its pursuit of becoming a fully integrated bioceutical company and it brings the Company another step closer to implementing the Company's Grow, Dry, Extract, Refine and Sell (GDERS) model.
Moreover, the Company is conducting continuing research and development while it is preparing and filing MMPR license applications. The Company recognizes the newly emerging opportunity in connection with medical marijuana and intends to further integrate it into the Company's overall botanical strategy. As the regulatory environment in Canada and in the US on Cannabis changed during this reporting period, management believes the Company, with its research team and growing equipment and proprietary extraction equipment, has the ability to become a leader in the medical marijuana industry worldwide. The Company's recent acquisition of exclusive worldwide rights to patent pending proprietary technologies will enable it to license, produce and deliver highly effective phyto-compounds for use in nutraceuticals, topical cremes, medical foods, botanical drugs and pharmaceuticals. Management's expertise we believe will enable the Company to produce bio-pharma proteins and compounds to meet all federal regulations and laws.
Abattis plans to control its own proprietary and pending patented products from seed to producing finished product at facilities owned by its wholly-owned subsidiary, N. Vine. The Company believes this will give it a competitive edge in the markets that the Company has identified. The Company believes its plans will ensure control in the supply chain and traceability at a Drug and Pharma level for its ingredients and finished products. The Company plans to target expansion of turn-key operations throughout Canada and the USA. N. Vine has limited operations as of this date.
License Application
In November 2013, the Company filed its initial MMPR Licensed Producer application with Health Canada (the "Application"). In general terms, following a submission of application form, the process to become a licensed producer under the MMPR is broken down by Health Canada into the following seven steps: (1) preliminary screening; (2) enhanced screening; (3) security clearance; (4) review; (5) ready to build letter, if required by applicant; (6) pre-licensing inspection; and (7) licensing. The Company, through its two subsidiaries, received a list of deficiencies in both applications. Since then, the Company has re-submitted the Application addressing deficiencies.
MMPR requires that the Minister of Health, after examining the application and any supplementary information requested, issue a renewed licence unless:
| (a) | the applicant is not an adult who ordinarily resides in Canada or a corporation that has its head office in Canada or operates a branch office in Canada and whose officers and directors are all adults; |
49
| (b) | the requirements regarding notification of local authorities in sections 38 or 39 of the MMPR have not been met (such notifications would only be required with a renewal if there are changes to the information since the original application); |
| (c) | an inspector, who has requested an inspection, has not been given the opportunity by the applicant to inspect; |
| (d) | the Minister has reasonable grounds to believe that false or misleading information or false or falsified documents were submitted in or with the application; |
| (e) | information received from a peace officer, a competent authority or the United Nations raises reasonable grounds to believe that the applicant has been involved in the diversion of a controlled substance or precursor to an illicit market or use; |
| (f) | the applicant does not have in place the security measures set out in the Security Directive and Division 3 regarding an activity for which the license is sought; |
| (g) | the applicant contravenes or has contravened in the past 10 years |
| (i) | a provision of the CDSA or its regulations or the Food and Drugs Act, or |
| (ii) | a term or condition of another license or a permit issued to it under any of those regulations; |
| (h) | the renewal of the license would likely create a risk to public health, safety or security, including the risk of cannabis being diverted to an illicit market or use; |
| (i) | any of the following persons does not hold a security clearance: |
| (i) | the senior person in charge, |
| (ii) | the responsible person in charge, |
| (iii) | if applicable, the alternate responsible person in charge, |
| (iv) | if the applicant is an individual, that individual, and |
| (v) | if the applicant is a corporation, any of its officers or directors; |
| (j) | the proposed method of record keeping does not meet the requirements of the MMPR; or |
| (k) | if applicable, any supplemental information requested has not been provided or is insufficient to process the application. |
Once Health Canada approves the Application, the Company expects to receive a full license (the "License") permitting the Company to possess, produce, sell, transport, deliver and destroy marijuana. The License, if granted, will allow for the production of up to 15,000 kilograms of dried marijuana annually within the 5,000 square feet licensed area in a facility in British Columbia.
The Company sub-leased a 16,200 square foot facility from Crimson Opportunities Ltd., which is owned by Rene David. The facility is near Vancouver, British Columbia and is zoned for medical marijuana production. Once the License has been granted, the facility will grow medical marijuana, as well as being converted to house up to 28 Biocube systems, an in-house laboratory, and a vault area for the secure storage of cannabis. Two additional buildings on the 6.5 acre property could also be used to expand cultivation to 254,000 square feet.
Narcotic Control Regulation's Licensing
Our subsidiary, N. Vine has applied for a Controlled Drugs and Substances Dealer's License, and is considered eligible for this license under the Narcotic Control Regulations of Canada as a corporation that has its head office in Canada or operates a branch office in Canada. To apply for a dealer's licence, a person shall submit an application to the Minister containing:
| The corporation's name and any other name registered with a province, under which it intends to carry out the activities specified in its dealer's licence or intends to identify itself; |
50
| The address, telephone number and, if applicable, the facsimile number and e-mail address for the premises to which the dealer's licence would apply and, if different, the mailing address for the premises; |
| The name, date of birth and gender of the individual in charge of the premises; |
| With respect to the proposed qualified person in charge and, if applicable, the proposed alternate qualified person in charge: |
| Their name, date of birth and gender, |
| Their academic qualifications, training and work experience relevant to their duties, |
| Their hours of work at the premises, |
| Their title at the premises, |
| The name and title of their immediate supervisor at the premises, |
| The name and gender of the individuals authorized to place an order for a narcotic on behalf of the applicant; |
| The activities for which the licence is sought that would be carried out at the premises to which the dealer's licence would apply; |
| If the licence is sought to produce a narcotic other than a product or compound that contains a narcotic; |
| A detailed description of the security measures at the premises, determined in accordance with the Security Directive; |
| A detailed description of the method that the applicant proposes to use for recording their narcotic transactions. |
An application for a dealer's licence must:
| Be signed by the individual in charge of the premises to which the licence would apply; and |
| Be accompanied by a statement signed by the individual in charge indicating that |
| All information and documents submitted in support of the application are correct and complete to the best of their knowledge, and |
| The individual has the authority to bind the applicant. |
An application for a dealer's licence must be accompanied by:
| Declarations signed by the individual in charge of the premises to which the application applies, the proposed qualified person in charge and, if applicable, the proposed alternate qualified person in charge, stating that they have not been convicted, as an adult, during the preceding 10 years, of: |
| a designated drug offence, |
| a designated criminal offence, or |
| an offence committed outside Canada that, if committed in Canada, would have constituted an offence referred to above; |
| A document issued by a Canadian police force with respect to each of the persons referred to here, stating whether the person has or has not been convicted, as an adult, during the previous 10 years, of a designated drug offence or a designated criminal offence; |
If any of the persons referred to here has ordinarily resided in a country other than Canada during the preceding 10 years, a document issued by a police force of that country stating whether the person has or has not been convicted in that country, as an adult, during the preceding 10 years, of an offence that would have constituted a designated drug offence or a designated criminal offence if committed in Canada; |
51
| A statement, signed and dated by the individual in charge of the premises to which the application applies, stating that the proposed qualified person in charge and, if applicable, the proposed alternate qualified person in charge have the knowledge and experience required; |
If the proposed qualified person in charge or, if applicable, the proposed alternate qualified person in charge is not a pharmacist or a practitioner of medicine, dentistry or veterinary medicine registered with a provincial professional licensing authority, a copy of the person's degree and a copy of the course transcript for that degree; |
| If the applicant is a corporation, a copy of: |
| the certificate of incorporation or other constituting instrument, and |
| any document filed with the province in which the premises to which the licence would apply are located that states its corporate name or any other name registered with the province, under which the applicant intends to carry out the activities specified in its dealer's licence or intends to identify itself. |
The method proposed by the applicant for recording their narcotic transactions must:
| Allow for the recording of narcotic transactions in accordance with section 15 of the Narcotic Control Regulations; and |
| Permit the Minister to audit the activities of the licensed dealer with respect to narcotics. |
The Minister may, on receiving an application made under these Regulations, require the submission of any additional information that pertains to the information contained in the application and that is necessary for the Minister to process the application.
Subject to section 9.4 of the Narcotic Control Regulations, the Minister shall, after examining the information and documents required as stated above, issue a dealer's licence that contains:
| The licence number; |
| The name of the licensed dealer or the title of the position they hold, or, if the licensed dealer is a corporation, its corporate name; |
| A list of the activities that are permitted; |
| The address of the premises at which the licensed dealer may carry on the permitted activities; |
| The name of the narcotic for which the activities are permitted; |
| The security level at the premises, determined in accordance with the Security Directive; |
| The effective date of the licence; |
| The expiry date of the licence, which may not be later than three years after its effective date; |
| Any conditions to be met by the licensed dealer to: |
| ensure that an international obligation is respected, |
| provide the security level referred to above, or |
| reduce the potential security, public health or safety hazard, including the risk of the narcotic being diverted to an illicit market or use; |
In the case of a producer of a narcotic, the quantity of the narcotic that may be produced under the licence and the period during which that quantity may be produced. |
The Minister shall refuse to issue, renew or amend a dealer's licence if:
| The applicant is not eligible under section 8.2 of the Narcotic Control Regulations; |
| An inspector who has requested an inspection has not been given the opportunity by the applicant to conduct an inspection under section 16 of the Narcotic Control Regulations; |
52
| False or misleading information or false or falsified documents were submitted in or with the application; |
| An activity for which the licence is requested would not be in compliance with an international obligation; |
| Information received from a competent authority or the United Nations raises a reasonable belief that the applicant has been involved in the diversion of a narcotic to an illicit market or use or has been involved in an activity that was not in compliance with an international obligation; |
| The applicant does not have in place the security measures set out in the Security Directive in respect of an activity for which the licence is requested; |
| The applicant is in contravention of or has contravened during the preceding 10 years: |
| a provision of the Act or the regulations made or continued under it, or |
| a term or condition of another dealer's licence or of an import or export permit issued to the applicant under any regulations made or continued under the Act; |
The issuance, amendment or renewal of the licence would likely create a risk to public health, safety or security, including the risk of a narcotic being diverted to an illicit market or use; |
| The individual in charge of the premises, the proposed qualified person in charge or, if applicable, the proposed alternate qualified person in charge has been convicted, as an adult, within the preceding 10 years, of: |
| a designated drug offence, |
| a designated criminal offence, or |
| an offence committed outside Canada that, if committed in Canada, would have constituted an offence referred to above; |
| The proposed method referred to above is not capable of recording narcotic transactions as required under section 15 of the Narcotic Control Regulations or of permitting the Minister to audit the applicant's activities with respect to narcotics in a timely manner; or |
| The additional information required under section 9.1 of the Narcotic Control Regulations has not been provided or is insufficient to process the application. |
Unless it is necessary to do so to protect public health, safety or security, including preventing a narcotic from being diverted to an illicit market or use, the Minister shall not refuse to issue, renew or amend a licence under paragraph (1)(c) or (g) of the Narcotic Control Regulations if the applicant:
| Does not have a history of non-compliance with the Act or any regulation made or continued under it; and |
| Has carried out, or signed an undertaking to carry out, the necessary corrective measures to ensure compliance with the Act, these Regulations and the Marihuana for Medical Purposes Regulations. |
Operations
The Company operates in the newly created regulated medical marijuana sector in Canada. This sector is defined by specific legislation that regulates the production, storage and sale of narcotics. This new regime borrowed from existing pharmaceutical regulations that mandated storage, production and security requirements which could be easily enforced. Part of the new legislation was a requirement to create the facility and be inspected prior to licensing.
To date the Company's focus has been on setting-up its various subsidiaries, entering into strategic relationships and establishing facilities that meet the requirements to obtain a MMPR Licensed Producer designation from Health Canada.
53
The primary specialized skill unique to the medical marijuana industry is with respect to the growing of product. While a background in the growing of marijuana specifically may be helpful, the nature of growing marijuana does not differ substantially from the nature of growing any other greenhouse product. These skills are generally available. The Company will also require client care staff when its business becomes operational.
Customer care staff is a skill set that is also generally available in the market.
Differentiation in the strains of medical marijuana is primarily achieved through the procurement of seeds.
Obtaining seeds for growing medical marijuana must be done in accordance with the MMPR. Seeds must be obtained from a legal source which includes seeds acquired from Health Canada and seeds imported from a jurisdiction allowed to export seeds. An authorization from Health Canada is required to conduct such a transaction.
Equipment used is specialized, but is readily available and not specific to the cultivation of medical marijuana.
Subject to available funding, the Company does not anticipate any difficulty in obtaining equipment as needed.
All aspects of Company's business are governed by standard operating procedures, from security procedures (rounds/incidents) to cleaning protocols. The majority of regulations have little to do with the actual growing of marijuana but are security and quality related.
Principal Products
Through our subsidiary BLI, we have 13 proprietary formulations for out-licensing to cosmetic, nutraceutical and pharmaceutical companies. BLI has identified several other botanicals as candidates to produce. On February 17, 2015, BLI launched eight phytocannabinoid formulas at CannaCon Seattle 2015. One formula consists of a beverage known as ECS Supreme, which contains phytocannabinoids and Abattis' patented nitric oxide blend. Other condition-specific cannabinoid formulas address nausea, anxiety and traumatic brain injury.
Through our subsidiary iJuana, we have 77 Natural Health Product Licenses issued by Health Canada. These range from (A) Aloe Vera to (Z) Zinc.
Through our subsidiary Biocube, we have the worldwide rights to a building product derived from magnesium oxide known as MgO board, which has applications in all areas of the building and construction industry as a fireproof, antimicrobial, antibacterial, waterproof, and recyclable material
Through our investment in Experion, we have developed strains of cannabis for use across many medical conditions when our MMPR application is approved.
Through our affiliate IPS, we own a secure banking software which provides a pre-paid cash component for financial transactions. We would use a mobile app and a loyalty card which allows patients to easily purchase medical marijuana.
Through our subsidiaries iJuana Cannabis Inc. and N. Vine; and investment in Experion, the Company intends to grow, package and sell medical marijuana in Canada. These subsidiaries have no operations in this business area as of this date.
54
Through our subsidiary NAB and N. Vine, the Company intends to develop customized THC extract oils and CBD extract oils, customized vaporizer flavors, sprays, creams and tinctures all related to the medical marijuana industry. These subsidiaries have no operations in this business area as of this date.
Through our affiliate Phytalytics and subsidiary N. Vine, the Company offers analytical and laboratory services for nutraceuticals, biopharmaceuticals and plant based medicines including medical marijuana.
To date, we have not generated any significant revenues and we remain in the development stage. Our ability to pursue our business plan and generate revenues is subject to our ability to obtain additional financing, and we cannot give any assurance that we will be able to do so.
Market Plans and Strategies
Medical Marijuana
Health Canada in 2014 suggested there are over 41,000 patients registered, and by 2024, that number is projected to rise to over 450,000 patients.
Direct Sales to Consumers via Online or Telephone Channels
Upon registration, consumers in Canada will be able to directly and securely purchase or re-order the Company's products via the company's website which will be further enhanced through search engine optimization ("SEO") strategies. According to the regulations of Health Canada, only patients within Canada will be able to purchase these products through these channels.
In order for customers to register with the Company and order its products they must provide the following documents:
the Company will ensure the highest level of security of its systems to ensure the confidentiality and security of customer and transactional information; system will be integrated with, a provider of technology solutions and support for commercial cannabis growing operations, for patient profiles, document storage and e-commerce functionalities. |
| the Company's website will provide the relevant and necessary information in order to educate and advise customers to make informed purchasing decisions. |
| Online channels can further be expanded into mobile channels to provide greater ease of access by customers to the Company's products. |
| Shipping will be completed by Canada Post Secured Shipping (the only approved carrier for Controlled Substances) requiring customer signature upon receipt of the product. |
| Per MMPR Section 5 "Packaging, Labelling and Shipping", shipments will include the following features: |
| Product will only be sent to the customer's home, shelter or the health care practitioner who provided the customer's medical document. |
| Container for shipping will have no smell or visible indicators for the product, tamper evident and child resistant. |
| Shipment will contain prescribed information about the product and client specific label. |
| Maximum package size will be 30 day supply or a maximum of 150g max per shipment. |
| Package will be clearly labelled "Keep Out of Reach of Children" as well as list the expiration date based on stability testing prior to shipping. |
The Company anticipates it will have additional revenue opportunities from merchandise sales, consulting, brand licensing, and proprietary technology and software licensing.
55
Sales via Intermediaries (Medical Professionals)
The Company also intends to market and promote its medicinal marijuana product to health care professionals. The Company intends to work closely with medical professionals to provide educational and awareness training to ensure they understand the benefits of its products and incentivize professionals to recommend the Company's products to patients that may benefit from its use, the Company will also verify the physician is able to practice and is not prohibited from prescribing controlled substances and/or narcotics. Further verification will be made with the physician's office to ensure that the medical documentation and quantity is complete and correct. Medical professionals will be able to scan bar codes to place direct orders online or via the phone through a dedicated line.
Once the Company has been able to establish a stronger brand and gain recognition of its product quality, medical professionals can be strategically approached to expand this channel. However, it should be noted that intermediary channels are strongly dependent on brand and quality development and thus must be developed concurrently to optimize our strategy.
Competition
Marijuana Industry
The Company has applied for Licensed Producer Status under the MMPR in Canada. As of April 27, 2015, there are 24 Licensed Producers in Canada each intending to produce medical marijuana.
| Name(1) | Location | Private/Public | Trading Symbol |
| ABcann Medicinals Inc.(2) | Ontario | Private | n/a |
| Agripharm Corp. (2) | Ontario | Public(3) | TSX-V: MT |
| Aphria | Ontario | Public | TSXV: APH |
| Aurora Cannabis Enterprises Inc. (2) | Alberta | Public(4) | CSE: ACB |
Bedrocan Canada Inc. NKA: Bedrocan Cannabis Corp. | Ontario | Private | TSXV: BED |
| Broken Coast Cannabis Ltd. | British Columbia | Private | n/a |
| Canna Farms Ltd. | British Columbia | Private | n/a |
| CanniMed Ltd. | Saskatchewan | Private | n/a |
| CannTrust Inc.(2) | Ontario | Private | n/a |
| Delta-9-Bio Tech LP | Manitoba | Private | n/a |
| Hydropothecary(2) | Quebec | Private(5) | n/a |
| In the Zone Produce Inc. | British Columbia | Private | n/a |
| MariCann Inc. | Ontario | Private | n/a |
| MedReleaf Corp. | Ontario | Private | n/a |
| Mettrum Ltd. | Ontario | Public(6) | TSX-V: MT |
| OrganiGram Inc. | New Brunswick | Public | TSX-V: OGI OTC PK: OGRMF |
| Prairie Plant Systems Inc. (2) | Saskatchewan | Private | n/a |
| RedeCan Pharm(2) | Ontario | Private | n/a |
| The Peace Naturals Project Inc. | Ontario | Private | n/a |
| Thunderbird Biomedical Inc.(2) | British Columbia | Public(7) | TSX-V: TPI |
| Tilray | British Columbia | Private | n/a |
| Tweed Farms Inc. (2) | Ontario | Public(8) | TSXV: TWD |
| Tweed Inc. | Ontario | Public(9) | TSXV: TWD |
| Whistler Medical Marijuana Corp. | British Columbia | Private | n/a |
56
Notes:
| (1) | The list of current Licensed Producers is available on the Health Canada website at: http://www.hc-sc.gc.ca/dhp-mps/marihuana/info/list-eng.php |
| (2) | Licensed producers who are authorized to produce marijuana but are not authorized to sell to the public as other licensed producers on this list. |
| (3) | Agripharm Corp. is a wholly owned subsidiary of Mettrum Ltd., a TSX-V listed company. This is Mettrum's second license under the MMPR. |
| (4) | Aurora Cannabis Enterprises Inc. is a wholly owned subsidiary of Aurora Cannabis Inc., a CSE company. |
| (5) | Hydropothecary has a reverse takeover pending with BFK Capital Corp. TSX-V: BFK. Company will be listed on TSX-V on close of the transaction. |
| (6) | Mettrum Ltd. is a wholly owned subsidiary of Mettrum Health Corp., a TSX-V company. |
| (7) | Thunderbird Biomedical Inc. is a wholly owned subsidiary of T-Bird Pharma Inc., a TSX-V company. |
| (8) | Tweed Farms Inc. is a wholly owned subsidiary of Tweed Marijuana Inc., a TSX-V company. |
| (9) | Tweed Inc. is a wholly owned subsidiary of Tweed Marijuana Inc., a TSX-V company. |
A number of other entities have applications pending, or will seek to obtain Licensed Producer Status under the MMPR.
The differentiators between competitors are expected to be price, quality (smell/taste/appearance), organic purity (zero additive, pesticide, mold treatment or anti-biological), and production process. The cost of growing an inexpensive strain (i.e. mass market) is identical to growing premium strains and the crop risks are identical (disease, pests and infrastructure failure). The majority of firms with listed product often overlap in strains and strengths (THC/CBD).
Bioceutical Industry
Notwithstanding the foregoing, the bioceutical and nutraceutical industries are highly competitive and subject to significant and rapid technological change. Developments by our competitors may render our products obsolete or noncompetitive. Numerous companies compete in our market, many of which have greater size and financial, personnel, distribution and other resources greater than ours. Our principal competition in the distribution channels where we are marketing our current products and where we intend to market other products comes from a limited number of large nationally known manufacturers and many smaller manufacturers of nutraceutical supplements. In addition, large pharmaceutical companies compete with us on a limited basis in the nutraceutical supplement market. Increased competition from such companies could have a material adverse effect on us because such companies have greater financial and other resources available to them and possess distribution and marketing capabilities far greater than ours. We also face competition in mass market distribution channels from private label nutraceutical supplements offered by health and natural food store chains and drugstore chains. We cannot assure that we will be able to compete.
Intellectual Property
Patents
The proprietary nature of, and protection for, our product candidates, processes, and know-how are important to our business. Our success depends in part on our ability to protect the proprietary nature of our product candidates, technology, and know-how, to operate without infringing on the proprietary rights
57
of others, and to prevent others from infringing our proprietary rights. We seek patent protection in Canada and internationally for our product candidates and other technology. Our policy is to patent or in-license the technology, inventions and improvements that we consider important to the development of our business. In addition to patent protection, we intend to use other means to protect our proprietary rights, including pursuing marketing or data exclusivity periods, orphan drug status, and similar rights that are available under regulatory provisions in certain countries, including the United States, Europe, Japan and China.
We also rely on trade secrets, know-how, and continuing innovation to develop and maintain our competitive position. We cannot be certain that patents will be granted with respect to any of our pending patent applications or with respect to any patent applications filed by us in the future, nor can we be sure that any of our existing patents or any patents granted to us in the future will be commercially useful in protecting our technology.
Despite these measures, any of our intellectual property and proprietary rights could be challenged, invalidated, circumvented, infringed or misappropriated, or such intellectual property and proprietary rights may not be sufficient to permit us to take advantage of current market trends or otherwise to provide competitive advantages.
Subsequent to the year ended September 30, 2014, the Company was granted a patent, which was originally acquired on April 16, 2009 from PRB and Pacific Bio.
The following table lists the intangible assets managed by the Company and the full value of the asset. The shares and cash paid by the Company for its part or full ownership of the asset are also listed:
| Consideration | |||||
| Acquisition Date | Items | Vendor Name | Fair Value | Shares issued | Cash paid |
| April 16, 2009 | Patents | PRB and Pacific Bio | $ 500,000 | $ 5,000,000 | $ - |
| May 17, 2011 | Formulae | Dr. Samuel Brant LLC | 125,000 | 200,000 | - |
| May 17, 2011 | Formulae | Biocell Labs | 125,000 | 200,000 | - |
| February 29, 2012 | Licenses (77 NPNs) | Animo Wellness Corporation | 50,000 | 100,000 | 25,000 |
| July 10, 2012 | Licenses (1 NPN) | - | 3,188 | - | 3,188 |
August 15, 2012 | Licenses (16 NPNs) | Sci-Natural Wellness Corporation | 56,000 | - | 56,000 |
| August 15, 2012 | Licenses (11 NPNs) | Northern Vine Canada Inc. | 7,143 | - | - |
| December 27, 2012 | Licenses to the Bio-Pharma patent license | Vertical Designs Ltd. | 500,000 | 6,000,000 | - |
| March 28, 2013 | Formulae | Dr. Paula Brown | 78,000 | 400,000 | - |
| September 30, 2013 | Patents - abandoned | PRB and Pacific Bio | (250,000) | - | - |
| February 24, 2014 | Licenses - innovative MgO products | Jiangsu Jiahui New Material Co. Ltd. | 21,120 | 110,000 | 2,420 |
| March 12, 2014 | Formulae | Green-Gro Garden Products Ltd. | 257,840 | 315,000 | 5,840 |
| April 7, 2014 | Licenses | Phytalytics | 1,245,812 | 827,657 | 88,782 |
| August 6, 2014 | Licenses | Terrasphere | 109,500 | - | 109,500 |
| October 1, 2014 | Trademarks | Oliver Hunt | 309 | - | 309 |
| October 29, 2014 | Formulae | Empirical Labs, Inc | 2,227 | - | 2,227 |
| November 28, 2014 | Trademarks | Oliver Hunt | 215 | - | 215 |
| November 30, 2014 | Trademarks | Oliver Hunt | 280 | - | 280 |
| Total | $ 2,831,634 | $ 13,152,657 | $ 293,761 | ||
58
With respect to any patents that may issue in the United States and Europe, we may also be entitled to obtain a patent term extension to extend the patent expiration date.
Trademarks
As of the date of this prospectus, we have three trademarks.
Other
We rely upon unpatented trade secrets, know-how, and continuing technological innovation to develop and maintain our competitive position. We seek to protect our ownership of know-how and trade secrets through an active program of legal mechanism including assignments, confidentiality agreements, material transfer agreements, research collaborations, and licenses.
Government Regulation
Medical Marihuana Production in Canada
On July 30, 2001, the Government of Canada implemented the Marihuana Medical Access Regulations (MMAR) under subsection 55(1) of the Controlled Drugs and Substances Act, which defines the circumstances and the manner in which marijuana can be used in Canada for medical purposes. The MMAR and regulations granted access to marijuana for Canadians suffering from symptoms (pain, muscle spasms, nausea, and weight loss) related to multiple sclerosis, cancer, HIV, spinal cord injury, epilepsy, arthritis or other debilitating symptoms as determined by a medical doctor.
59
On June 7, 2013 the Canadian regulations concerning the production and sale of medical marijuana were amended by the Marihuana for Medical Purposes Regulations (MMPR) which permit licensing commercial growers beginning April 1, 2014, while eliminating provisions for its production on a personal-use basis. Applications for personal-use production ceased to be processed as of October 1, 2013 and, individuals authorized to possess medical marihuana under the MMPR must transition to the new licensed producer regime.
The revised regulations create conditions for a commercial industry responsible for medical marijuana production and distribution, by eliminating small-scale, personal-use production. Commercial growers can now submit applications to Health Canada for the production of medical marijuana and, if licensed, supply patients who qualify for the product at a price established by market forces and at the discretion of producers.
The MMPR only permits the sale of dried marihuana; the production of concentrated or edible forms (oils, resins, teas or infusions) is not permitted. On March 21, 2014, the Court of Appeal of the Province of British Columbia ruled with R v. Owen Edward Smith that the MMPR`s restriction on the production of edible marihuana products for medicinal purposes is unconstitutional. It is expected that the court`s decision will be appealed to the Supreme Court of Canada where, if it is affirmed, would compel the Government of Canada to regulate access to edible medical marihuana products.
Other relevant requirements for applicants and licensed producers under the MMPR include:
| production facilities may only be located indoors(greenhouses are also acceptable); |
| production facilities must meet specified advanced security requirements to prevent and detect unauthorized access; |
| producers may not operate storefronts; |
| producers may not wholesale products except to other licensed producers; they must sell directly to authorized consumers or, if requested, to their physicians; |
producers must notify their local government, local police force and local fire officials of their intention to apply to Health Canada, so local authorities know of their proposed location and activities. Producers are also required to communicate with local authorities whenever there is a change in the status of their license; |
| producers must comply with all federal, provincial/territorial and municipal laws and by-laws, including municipal zoning by-laws; |
| there are no applicable federal fees payable regarding the application or maintenance of the license to produce marihuana under the MMPR; |
producer must have an employee designated as a quality assurance person responsible for assuring the quality of the dried marihuana, before it is provided for sale. This employee must have the training, experience and technical knowledge related to the proposed licensed activities and the MMPR; and |
| applicants must submit a detailed description of their proposed record keeping methods. This must describe the process used for recording transactions relating to licensed activities, including maintaining records of transactions and dealings with both suppliers and clients. |
Other aspects of the MMPR relevant to our business include:
| The MMPR contain no limitations on the conditions for which a health care practitioner can support the use of marijuana for medical purposes; |
| The MMPR imposes no limit on the number of production licenses; |
60
| There are no restrictions under the new MMPR on the daily marihuana that may be prescribed, there is an individual possession cap of the lesser of 150 grams or 30 times the daily amount. If an individual has a daily amount of 2 grams per day, their possession cap would be 60 grams. |
To date, Health Canada has not published or established any timelines or detailed procedures for the processing, evaluation or vetting of applications to obtain a producer license under the MMPR. Based on third party public information, we estimate that applications will be processed within 6 to 18 months from submission. Following acknowledgement of a complete application, applicant producers are subject to interviews, site visits and other due diligence procedures at the discretion of Health Canada.
Licensed Products under the MMPR are required to report of the following additional information to the Office of Controlled Substances of Health Canada on a monthly basis, unless otherwise stated:
| 1. | the total amount of dried marihuana (in kilograms) produced in the reporting period; |
| 2. | the total amount of dried marihuana (in kilograms) sold to the following during the reporting period: |
| (a) | Registered clients; |
| (b) | Other licensed producers; |
| (c) | Licensed dealers; and |
| (d) | Other clients; |
| 3. | the total number of persons that were registered clients of the Company at the end of the reporting period, including only those persons whose registrations were valid on the last day of the reporting period, and the total number of persons that were registered as new clients of the Company during the reporting period; |
| 4. | the number of registered clients who tried to register with the Company, but could not be registered, regardless of the reason and the number of clients who placed orders or tried to place orders that could not be filled, regardless of the reason; |
| 5. | the total amount of dried marihuana (in kilograms) as of the final day of the reporting period; |
| 6. | the total amount of dried marihuana (in kilograms) that the Company imported and exported during the reporting period; |
| 7. | the total amount of dried marihuana (in grams) lost and/or stolen and destroyed during the reporting period; |
| 8. | the total number of shipments sent to the following during the reporting period: |
| (a) | Registered clients; |
| (b) | Other licensed producers; |
| (c) | Licensed dealers; and |
| (d) | Other clients; |
| 9. | the total number of shipments sent to the following in each province and territory. |
| (a) | Registered clients; |
| (b) | Other licensed producers; |
| (c) | Licensed dealers; and |
| (d) | Other clients; |
| 10. | the average and median daily amount of dried marihuana (in grams) supported by Health Care Professionals to be used by the registered clients of the Company; |
| 11. | the average and median shipment size (in grams) sent to registered clients during the reporting period; |
| 12. | the ten highest and ten lowest amounts of dried marihuana shipped to registered clients in the reporting period (The name or other information of the registered client must not be identified.); |
61
| 13. | the total number of shipments of dried marihuana to registered clients in various defined ranges (in grams); and |
| 14. | a list of all physicians and all nurse practitioners who provided a medical document for a registered client in the reporting period, and include the following information for each: |
| (a) | the location; and | |
| (b) | the number of medical documents the physician or nurse practitioner signed during the reporting period. |
Marihuana Production in the United States
Our Company is focused on the Medical Marihuana Industry in Canada supported by the Canadian Federal Government and administered by Health Canada under the MMPR. Our Company is following the strict guidelines outlined regarding security, quality control and safety of the product under the current federal MMPR program.
In the United States it is still illegal under federal law to grow, cultivate and sell medical or adult use marijuana. However, 23 states have approved medical marihuana for use and two states have approved adult use regulations. The United States Federal government justice department has released memo's that will respect the individual states where strict guidelines are followed and enforced so the health, safety and security are protected by state authorities. If the individual state framework fails to protect the public the Federal government will act in enforcing the Controlled Substances Act of 1970 and the DEA will enforce the federal law.
As at the date of this registration statement, our Company has not entered into any prospective or definitive arrangements to produce or distribute marihuana products in the United States. However, our Company continually reviews opportunities in the medical marihuana sector in both Canada and the United States.
Other Regulations
We are also subject to numerous federal, provincial, state and local laws relating to such matters as safe working conditions, manufacturing practices, environmental protection, fire hazard control, and disposal of hazardous or potentially hazardous substances. We may incur significant costs to comply with such laws and regulations now or in the future.
Research and Development
Our subsidiary BLI specializes in research and development of botanicals for out-licensing our proprietary formulas to third parties.
We anticipate our subsidiaries NAB and N. Vine; and our investment in Experion, will conduct further research and development to increase production efficiencies, advance new medicinal marijuana strains and formulate compositions for sprays, creams and tinctures for sale.
As of September 30, 2014, the Company has spent $129,057 on research and development (September 30, 2013 - $123,559). The research and development expenses incurred in 2013 and 2012 are directly related to the activities of BLI.
Employees
As of April 27, 2015, our staff included one person, comprised of one full time employee. We primarily use the services of sub-contractors and consultants for our business operations.
62
Our employee is not part of a labor union or covered by collective bargaining agreement. We consider our relationship with our employee to be good. Our company uses a cost-effective business model, only retaining top management as company employees or dedicated consultants, and drawing upon other consultants to advance our development programs. As of April 27, 2015, our management consisted of our chief executive officer, chief financial officer, chief operating officer, tech advisor and global product development advisor.
Environmental Laws
As of the date of this prospectus, the Company is in compliant with local environmental laws. The Company has no reason to believe it is in violation of any environmental laws both provincially and federally.
The Company leases a 3742 square foot office in Langley, British Columbia, from Toro Holdings Ltd. The lease is for a 3-year term ending October 31, 2015 unless otherwise renewed. In 2015, the Company will pay $36,291 under the lease agreement.
The Company sub-leases a 16,200 square foot facility from Crimson Opportunities Ltd., which is owned by Rene David. The facility is located in Squamish, British Columbia and is zoned for medical marijuana production. Until the Company secures a license, the facility will be used to manufacture and warehouse its proprietary Biocube systems. Once a license has been granted, the facility will be used to grow medical marijuana, as well as being converted to house up to 28 Biocube systems, an in-house laboratory, and a vault area for the secure storage of cannabis. Two additional buildings on the 6.5 acre property could also be used to expand cultivation to 254,000 square feet. The lease is for a 5-year term ending May 31, 2019 unless otherwise renewed. The Company will pay approximately $437,400 annually under the lease.
The Company's wholly owned subsidiary, Animo Wellness Corporation, which conducts business through Medical Marijuana Labs, entered into a 5-year lease with PurGenesis Technologies, Inc. for the lease of approximately 5,000 square feet of lab and production space at a cost of $120,000 in annual gross rent. The facility is located in the Province of Quebec. The Company intends to produce medical marijuana in this facility. Amino Wellness Corporation changed its name to iJuana Cannabis Inc. on January 28, 2014.
The current directors and officers of the Company are listed below. Directors are generally elected at an annual general meeting of
63
shareholders and hold office until the next annual general meeting of shareholders, or until their successors are elected and qualified executive officers are elected by directors and serve at the Board's discretion.
| Name | Age | Position | Date Appointed | Number of Common Shares |
William Fleming(1) Nova Scotia, Canada | 48 | President, CEO and Director | June 18, 2014 | 350,000 |
Mike Withrow(2)(3) British Columbia, Canada | 52 | International Business Advisor | April 30, 2011 | 3,423,300(4) |
Tim Fealey(2) California, United States | 70 | Director and Global Product Development Advisor | April 30, 2011 | 69,000(5) |
Douglas Sorocco(2) Oklahoma, United States | 44 | Director | April 30, 2011 | 118,400(6) |
Emanuel Montenegrino Ontario, Canada | 59 | Director | June 18, 2014 | 90,000 |
Rene David British Columbia, Canada | 45 | CFO and COO | November 19, 2013 | 853,488 |
| Guy Dancosse Quebec, Canada | 71 | Director | September 16, 2014 | 50,000(7) |
Notes:
| (1) | Mr. Fleming was appointed CEO of the Company effective February 1, 2015. | |
| (2) | Member of Audit Committee. Mr. Withrow resigned as an officer of the Company effective February 1, 2015. He will be replaced on the Audit Committee in the upcoming quarter. | |
| (3) | Mr. Withrow resigned from his position as CEO, President and Director effective February 1, 2015. | |
| (4) | Does not include options to purchase 300,000 Common Shares and warrants to purchase 555,556 Common Shares. | |
| (5) | Does not include options to purchase 275,000 Common Shares and warrants to purchase 600,000 Common Shares. . | |
| (6) | Does not include options to purchase 375,000 Common Shares and warrants to purchase 40,000 Common Shares. | |
| (7) | Does not include options to purchase 100,000 Common Shares. |
William "Bill" Fleming
Mr. William "Bill" James Fleming has an extensive business background with national and international companies. Mr. Fleming has been VP E-Commerce for Bermuda based GlobeNet Communications, presently a Co-Founder of Goose Bay Capital Corporation and is the CFO and Director of NWest Energy Corporation. Mr. Fleming is the Founder of Mernova Medicinal Inc., an Aboriginal owned company focused on becoming a Canadian producer of legal marijuana. Mr. Fleming also has served with senior executive teams dealing with equity and debt financings. Mr. Fleming is a graduate of St. Francis Xavier University and a recipient of the ROTP Military Scholarship.
Mike Withrow
Mr. Mike Withrow is founder of North American BioExtracts Inc., an extraction technology equipment company and Biocell Labs Inc. and has served as VP of Business Development in two nutracuetical companies: Canadian Pacific Algae Inc. and Canadian Pacific Vital Plankton. He brings an in-depth
64
knowledge of supplement innovation, ingredients, is well connected in the ingredients and natural health product industry. Mike is a valued member of any team and is capable of managing intellectual property, ingredient research, extraction technologies, and formulation. He also has a keen understanding of animal and human testing, grant funding, and product and market development of a company's nutraceutical ingredient and finished product strategies. Mike is a board member of a publicly traded mining and exploration company and is a board Member of the Saskatoon Berry Council of Canada. During the past five years, Mr. Withrow was also a director of Newton Gold Corp., a TSXV junior resource company.
Mr. Withrow continues to consult to nutracuetical companies and has been instrumental in helping companies in very different industries receive over $3,800,000 in grant money. He was a founding partner of BFuel Canada Ltd., a planned bio-diesel facility in Lethbridge AB. Prior to BFuel Mike co-founded CBN Systems Inc., a company that unified and patented all known color spaces via the "CBN Code" and linked spectrophotometry to user friendly designer software and a colour formulation system. Early in his career Mike founded PowerTrader Inc., which was the first company to offer real-time financial data via a stand-alone MS Windows platform and on the Internet. Prior to that Mike was a floor trader on the Vancouver Stock Exchange.
Terrence (Tim) Fealey
Dr. Tim Fealey is a partner at OTC Nutrition LLC and formerly held the position of Senior VP Global Food and Beverage Strategic Planning for The Proctor and Gamble Company. Additionally, he has had senior R&D and Strategic leadership responsibilities with The Coca-Cola Company and recently served as Senior VP Chief Innovation Officer of Martek Bioscience Corporation, a leading nutrition biotechnology company based in Maryland, USA. Dr. Fealey has over 30 years of combined R&D and general management experience in all aspects of technology management and global strategic planning. He brings extensive business experience, knowledge, and a broad network, both private companies and public institutions, of personal contacts in all regions of the world. Dr. Fealey was a key contributor to the highly successful global expansion of the Pringles brand as well as other well-known consumer product brands. He has extensive experience in the business of foods and beverages including suppliers and external capabilities and to the global academic and regulatory communities. Dr. Fealey holds a PhD in Physical Chemistry from Georgetown University and MBA from University of Chicago.
Douglas Sorocco
Mr. Douglas Sorocco practices law in the areas of intellectual property, technology, licensing, life sciences and patents and is involved in counseling and transactional work involving all aspects of intellectual property. He is ranked among Oklahoma's top intellectual property practitioners by the highly regarded Chambers USA: America's Leading Lawyers for Business. Mr. Sorocco was recently selected by attorney peers for inclusion in Oklahoma Super Lawyers--Rising Stars Edition (2010).
Mr. Sorocco's scientific background has focused on all areas of biotechnology and life sciences (including molecular biology, cell biology, glycobiology, biochemistry, developmental biology, immunology, microbiology, virology, and genetics; pharmaceutical compositions; molecular diagnostics and techniques; medical devices and equipment) as well as chemistry and chemical engineering.
Mr. Sorocco has significant experience in providing strategic and tactical intellectual property counsel to individual clients, universities, large pharmaceutical and manufacturing companies, and start-up biotechnology companies. He is an adjunct faculty member at the Oklahoma City University School of Law and the Physiology Department at Oklahoma University Health Sciences Center.
65
Emanuel "Manny" Montenegrino
Mr. Emanuel (Manny) Montenegrino is the President and CEO of Think Sharp Inc., Think Sharp Inc. is an invaluable service to corporate clients who seek expeditious, creative solutions for their business objectives while facing government barriers. Mr. Montenegrino was previously Chair of the Government Relations Group at Lang Michener LLP, now McMillan LLP and has 32 years' experience as a business lawyer.
Rene David
Mr. Rene David has over 20 years of experience in real estate development, finance, and business strategy. He started his career at VanCity, Canada's largest Credit Union as a real estate developer and then went into investment banking in Western Canada. He later expanded his interests to include the resource, food and water industries, and assisting corporations who are dealing with the globalization of local sources. In his normal course of duties, Mr. David's responsibilities include internal financial controls and analysis, procurement and contracts management. His continuous review of operating and capital plans is geared towards improving the internal controls and financial best practices in order to deliver highest value product and strong company values.
Guy Dancosse
Mr. Guy Dancosse, Q.C. has extensive experience in arbitration, negotiation and mediation, nationally and internationally, in many areas of business, including the public sector. He has appeared before Courts of all jurisdictions in Canada. He acted as counsel in commercial arbitration, both nationally and internationally (International Chamber of Commerce). He also served as Arbitrator in commercial cases in Canada. Finally, he acted as counsel, and as party appointed Arbitrator in Labour and Employment matters across Canada, and in the United States.
He was part of Canadian Task Forces and Inquiry Commissions (Pilotage in the St. Lawrence River and Native Land Claims), and also headed World Bank and African Development Bank missions with mandates to review and recommend reforms to the legal and judicial systems of countries in Africa (Tanzania - Chad).
He served in the Canadian Foreign Service as a foreign service officer posted in Washington, D.C. and worked at General Dynamics' legal department (Canadair and Flextrack Nodwell) on their international aircraft sales programs.
He has undergone post-graduate training in mediation and arbitration at Harvard University, Boston, and is a member of the Advisory Board for Groupe Ocean Inc. and the Board of Directors for the Royal Canadian Mint. He is also a member of the Canadian Arbitration Committee of the ICC International Court of Arbitration (Paris). He has completed the Directors Education Program at Rotman School of Management in Toronto, jointly developed by the Institute of Corporate Directors (ICD) and the Rotman School of Management to help board directors clarify their mission and fully exercise their leadership potential as a Board member.
Family Relationships
There are no family relationships between any of the executive officers and directors. Each director is elected at our annual meeting of shareholders, if management deems it advisable to hold an annual
66
meeting of shareholders, and holds office for a term of one year, or until his successor is elected and qualified.
Cease Trade Orders or Bankruptcies
To the knowledge of the Company, no director of the Company is or has been, within the past 10 years, a director, chief executive officer or chief financial officer of any Company that:
| (a) | was subject to an order that was issued while the proposed director was acting in the capacity as director, chief executive officer or chief financial officer; or |
| (b) | was subject to an order that was issued after the proposed director ceased to be a director, chief executive officer or chief financial officer and which resulted from an event that occurred while that person was acting in the capacity as director, chief executive officer or chief financial officer. |
To the knowledge of the Company, no proposed director of the Company is or has been, within the past 10 years, a director or executive officer of any Company that, while that person was acting in that capacity or within a year of that person ceasing to act in that capacity, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency, or was subject to or instituted any proceedings, arrangement or compromise with creditors, or had a receiver, receiver manager or trustee appointed to hold its assets.
Individual Bankruptcies
To the knowledge of the Company, no proposed director of the Company has, within the past 10 years, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency, or become subject to or instituted any proceedings, arrangement or compromise with creditors, or had a receiver, receiver manager or trustee appointed to hold the assets of the proposed director.
Penalties or Sanctions
To the knowledge of the Company, no proposed director has been subject to any penalties or sanctions imposed by a court relating to securities legislation or by a securities regulatory authority or has entered into a settlement agreement with a securities regulatory authority, or has been subject to any other penalties or sanctions imposed by a court or regulatory body that would likely be considered important to a reasonable securityholder in deciding whether to vote for a proposed director.
The following disclosure of all direct and indirect compensation provided to certain executive officers and directors for, or in connection with, services they have provided to the Company or a subsidiary of the Company.
Summary Compensation Table for Named Executive Officers
The following summary compensation tables set forth information concerning the annual and long-term compensation for services in all capacities to our Company for the year ended September 30, 2014 of those persons who were, at September 30, 2014: (i) the principal executive officer (Mike Withrow); (ii) the principal financial officer (Rene David) and (iii) the two other most highly compensated executive
67
officers of the Company, whose annual base salary and bonus compensation was in excess of US $100,000 (none):
| Name and Position | Year | Salary ($) | Stock-based Awards ($) | Option-based Awards ($)(1) | Non-equity Incentive Plan Compensation | Pension Value ($) | All Other Compensation ($) | Total ($) |
Mike Withrow Former President, CEO and acting CFO(2) | 2014 | 172,134 | Nil | 2,226,591 | Nil | Nil | Nil | 2,398,725 |
| 2013 | 142,500 | Nil | 14,549 | Nil | Nil | Nil | 157,049 | |
| 2012 | 120,000 | Nil | Nil | Nil | Nil | Nil | 120,000 | |
Rene David CFO and COO(3) | 2014 | 127,425 | Nil | 983,105 | Nil | Nil | Nil | 1,110,530 |
| 2013 | - | - | - | - | - | - | - | |
| 2012 | - | - | - | - | - | - | - |
Notes:
| (1) | The Black Scholes valuation methodology was used to determine value on the date of grant. |
| (2) | Mr. Withrow resigned as President, CEO and Director of the Company effective February 1, 2015. Mr. Withrow was the acting CFO of the Company from September 11, 2011 until November 18, 2013. |
| (3) | Mr. Rene David was appointed the CFO and COO of the Company on November 19, 2013. |
The compensation amounts reported as option-based awards in the above table represent the estimated grant date fair value of the stock options granted during the year. All of these stock options were granted with an exercise price equal to the market price of the Common Shares on the date of grant. The amounts reported do not represent the net cash proceeds received by the individuals from the exercise of stock options.
General Compensation Strategy
The Company has, as of yet, no significant revenues from operations and often operates with limited financial resources to ensure that funds are available to complete strategic acquisitions and to develop its products. As a result, the directors of the Company have to consider not only the financial situation of the Company at the time of the determination of executive compensation, but also the estimated financial situation of the Company in the mid and long term. An important element of executive compensation is that of stock options and incentive and performance share bonuses, which do not require cash disbursements by the Company.
Remuneration plays an important role in attracting, motivating, rewarding and retaining knowledgeable and skilled individuals to the Company's management team. The main objectives the Company hopes to achieve through its compensation arrangements are:
| to attract and retain executives critical to the Company's success, who will be key in helping the Company achieve its corporate objectives and increase shareholder value; | ||
| to motivate the Company's management team to meet or exceed targets; | ||
| to recognize the contribution of the Company's executive officers to the overall success and strategic growth of the Company; and |
68
| to align the interests of management and the Company's shareholders by providing performance-based compensation in addition to consulting fees or salary. |
The key elements of executive compensation awarded by the Company are: (i) base consulting fees/salary; (ii) potential annual and other incentive awards; (iii) potential performance based incentive awards; and (iv) incentive stock options. The goal of the Company's executive compensation system is to attract, motivate and retain the best talent possible while at the same time being fair to its shareholders. As the Company continues to establish itself as a viable business, the Board has created and expects to continue to create a comprehensive set of business performance metrics or industry standard benchmarks to determine executive compensation. Variables that factor into establishing its executive compensation system include, but are not limited to, the following, which are set out in no particular order:
| available working capital; | ||
| pre-determined annual and monthly budgets; | ||
| comparable base salary for positions in similar size Companies; | ||
| weighted criteria relative to the Company's development and growth stage; | ||
| education and experience relative to the industry and position; | ||
| balance between short-term and long-term goals and be relative to achievements within those time periods; and | ||
| paying performance based incentive bonuses for achieving or exceeding pre-determined short and long term goals (i.e., such as: acquisitions/grants of MMPR licenses or controlled substance licenses; financing targets; revenue growth; profit margins; etc.), which may include any or a combination of the following: |
| stock options | ||
| cash bonus | ||
| stock bonus | ||
| benefits | ||
| retirement package | ||
| other allowances |
Executive Compensation Agreements
Consulting Agreement - Michael Withrow (former President, CEO & Director)
Mr. Withrow was appointed as the President and CEO of the Company on February 1, 2011. On January 10, 2011, Mr. Withrow entered into a consulting agreement with the Company pursuant to which he was engaged as an advisor to the Company. This agreement was subsequently terminated and superseded by an executive consulting agreement dated effective May 1, 2011, as amended January 1, 2014 between the Company and Chiron Capital Inc. (the "Withrow Agreement") with respect to his executive positions held with the Company since February 1, 2011. Mr. Withrow resigned as President and CEO of the Company effective February 1, 2015. He remains a director and a consultant to the Company.
Pursuant to the terms of the Withrow Agreement, Mr. Withrow initially received $120,000 per annum plus applicable HST/GST, which has subsequently been increased to $175,000 per annum plus applicable GST. In addition, from and after January 1, 2014, Mr. Withrow is entitled to receive an incentive bonus of 500,000 Common Shares upon each MMPR license and/or a controlled substance license being granted by Health Canada to the Company or a subsidiary of the Company, or upon the Company or a subsidiary otherwise acquiring an MMPR license and/or controlled substance license, which number of shares may be pro rated in certain circumstances where the Company or a subsidiary does not own a 100% interest in
69
the entity holding such license. Mr. Withrow is also entitled to receive additional variable performance bonuses, incentive shares and/or stock options, at the sole discretion of the Board. In the event the Company provides a benefits package to its consultants and employees, then Mr. Withrow is also entitled to receive benefits thereunder. All reasonable documented expenses incurred by Mr. Withrow in connection with his duties are also reimbursed by the Company.
Mr. Withrow may terminate the Withrow Agreement at any time by providing the Company with 60 days' written notice. The Company may, at any time, terminate the Withrow Agreement for cause, without notice and without liability for any claim, action or demand. Notwithstanding the aforementioned, the Company may terminate the Agreement by paying to Mr. Withrow a lump sum amount equal to three months' salary plus one additional month's salary for each full year of service provided by Mr. Withrow (up to a maximum of 12 months' salary), plus any other amounts owed by the Company to Mr. Withrow under the Withrow Agreement at the time of termination.
In the event that Withrow Agreement is terminated within 12 months of a change in control of the Company, then Mr. Withrow will be entitled to a lump sum payment equal to nine months' salary. In addition, the Company is required to pay all accrued but unpaid consulting fees and bonuses owing to Mr. Withrow, and any previously issued but unvested options granted to Mr. Withrow vest immediately on termination and remain exercisable for the period of time as is permitted under the Option Plan.
Consulting Agreement - Crimson Opportunities Ltd. (Rene David - CFO & COO)
On November 19, 2013, Rene David was appointed as the CFO and Chief Operations Officer ("COO") of the Company. Pursuant to an executive consulting agreement with Crimson Opportunities Ltd. ("Crimson") dated October 1, 2013, as amended January 1, 2014 (the "Crimson Agreement"), Crimson was engaged to provide management consulting services, including the services of Rene David to act as the CFO and COO of the Company.
Pursuant to the terms of the Crimson Agreement, Crimson receives $125,000 per annum plus applicable GST, Crimson is entitled to receive an incentive bonus of 400,000 Common Shares upon each MMPR license and/or a controlled substance license being granted by Health Canada to the Company or a subsidiary of the Company, or upon the Company or a subsidiary otherwise acquiring an MMPR license and/or controlled substance license, which number of shares may be pro rated in certain circumstances where the Company or a subsidiary does not own a 100% interest in the entity holding such license. Pursuant to the Crimson Agreement, Crimson is also entitled to receive 1,000,000 incentive stock options, as follows: (i) 250,000 options upon execution of the Crimson Agreement (which options have been granted); (ii) 250,000 options upon the completion of a $400,000 financing (which options have been granted); (iii) 250,000 options upon the completion of a $1,000,000 financing (which options have been granted); and (iv) 250,000 options upon the announcement by Health Canada that the Company or one of its subsidiaries is a licensed producer under MMPR. Crimson is also entitled to additional variable performance bonuses, incentive shares and/or stock options, at the sole discretion of the Board. All reasonable documented expenses incurred by Crimson in connection with providing services under the Crimson Agreement are also reimbursed by the Company.
Crimson may terminate the Crimson Agreement at any time by providing the Company with three months' written notice until September 30, 2014 and thereafter by providing six months' notice. The Company may, at any time, terminate the Crimson Agreement for cause or upon the death or disability of Rene David, without notice and without liability for any claim, action or demand.
70
Incentive Plan Awards for Executive Officers of the Company
Outstanding Option-Based Awards
The following table (and notes thereto) states the names of each person who is a Named Executive Officer, and the value of all compensation provided for the most recently completed financial year of the Company ended September 30, 2014.
| Name | Option-based Awards | Share-based Awards | |||||
| Number of securities underlying unexercised options | Option exercise price ($) | Option expiration date | Value of unexercised in-the-money options(1) ($) | Number of shares that have not vested | Market or payout value of share-based awards that have not vested | ||
| Mike Withrow(2) | 300,000 | 0.35 | May 12, 2016 | $7,500 | N/A | N/A | |
| Rene David(3) | 0 | N/A | N/A | N/A | N/A | N/A | |
Notes:
| (1) | The value of "in-the-money-options" is calculated based on the difference between the market value of the Company's Common Shares underlying the options at the end of the most recently completed financial year and the exercise price of the options. The last trading price of the Company's Common Shares on the CSE as of September 30, 2014 was $0.375 per share. |
| (2) | During the year ended September 30, 2014, 3,230,000 stock options, with an estimated fair value of $2,226,591 were granted by the Company to Mr. Withrow and Chiron Capital Inc., a company controlled by Mr. Withrow. |
| (3) | During the year ended September 30, 2014, 2,315,000 stock options, with an estimated fair value of $983,105 were granted by the Company to Crimson Opportunities Ltd., a company controlled by Mr. David. |
During the year ended September 30, 2014, 890,000 options were exercised by Mike Withrow directly or through a company controlled by Mr. Withrow.
During the year ended September 30, 2014, 550,000 options were exercised by Rene David directly or through a company controlled by Rene David.
The Company did not have a long-term incentive plan pursuant to which cash or non-cash compensation intended to serve as an incentive for performance (whereby performance is measured by reference to financial performance or the price of the Company's securities) was paid or distributed to the Named Executive Officers during the most recently completed financial year.
The significant terms of the Option Plan, pursuant to which all current option-based awards have been granted to executive officers of the Company are set out below under the heading "Equity Compensation Plan" below.
There was no re-pricing of stock options under the Option Plan or otherwise during the Company's most recently completed financial year ended September 30, 2014.
Pension Plan Benefits for Executives of Company
As of April 27, 2015 and as at the year ended September 30, 2014, the Company did not maintain any defined benefit plans, defined contribution plans or deferred compensation plans.
71
Termination and Change of Control Benefits and Employment Contracts
As at April 27, 2015, the Company had the following plans or arrangements whereby NEOs could be compensated in the event of such NEO's resignation, retirement or other termination of employment, or in the event of a change of control of the Company:
| (a) | Pursuant to the terms of the Withrow Agreement, which includes amendments made subsequent to the fiscal year ended September 30, 2014, in the event the Company terminates the Withrow Agreement for other than cause, subject to the change of control provisions contained in the agreement, the Company is required to pay Mr. Withrow a lump sum amount equal to three months' salary plus one additional month's salary for each full year of service provided by Mr. Withrow (up to a maximum of 12 months' salary), plus any other amounts owed by the Company to Mr. Withrow under the Withrow Agreement at the time of termination. In the event that the Withrow Agreement is terminated within 12 months of a change in control of the Company, then Mr. Withrow will be entitled to a lump sum payment equal to nine months' salary. In addition, the Company is required to pay all accrued but unpaid consulting fees and bonuses owing to Mr. Withrow, and any previously issued but unvested options granted to Mr. Withrow vest immediately on termination and remain exercisable for the period of time as is permitted under the Option Plan. | |
| (b) | Pursuant to the Option Plan: |
| (i) | in the event that an optionee is dismissed from employment or services for cause, all options granted to the optionee terminate without right to exercise same immediately upon such dismissal; |
| (ii) | in the event that an optionee ceases to be employed/engaged by the Company other than by reason of death or dismissal for cause, the expiry date of all options granted to such optionee will be 30 days from the date the optionee ceases to be employed with/provide services to the Company and the expiry date otherwise applicable to such options, but only to the extent that such options are vested at the date the optionee ceases to be so employed/provide services to the Company; and |
| (iii) | in the case of the death of an optionee, any vested options held by him or her at the date of death will become exercisable by the optionee's lawful personal representatives, heirs or executors until the earlier of one year after the date of death of such optionee and the date of expiration of the term otherwise applicable to such option. |
Director Compensation
The Company has five directors as at April 27, 2015.
The Company has no arrangements, standard or otherwise, pursuant to which directors are compensated by the Company for their services in their capacity as directors, or for committee participation or involvement in special assignments during the most recently completed financial year or subsequently, up to and including the date of this registration statement.
The Company grants stock options to directors pursuant to the terms of the Option Plan (see "Equity Compensation Plan" below for details). The purpose of granting such stock options is to assist the Company in compensating, attracting, retaining, and motivating the directors of the Company and to align the personal interests of such persons to that of Shareholders.
72
Director Compensation Table
The following table (and notes thereto) states the names of each director, who is not also a Named Executive Officer, and the value of all compensation provided for the most recently completed financial year of the Company ended September 30, 2014.
| Name(1) | Fees earned ($) | Share-based awards ($) | Option-based awards ($)(2) | Non-equity incentive plan compensation ($) | Pension value ($) | All Other Compensation ($) | Total ($) |
| Tim Fealey | Nil | Nil | Nil | Nil | Nil | 104,182 | 104,182 |
| Douglas Sorocco | Nil | Nil | 357,306 | Nil | Nil | Nil | 357,306 |
| Robert Hedley(3) | Nil | Nil | 89,326 | Nil | Nil | Nil | 89,326 |
| Guy Dancosse | 15,000 | Nil | 20,422 | Nil | Nil | Nil | 35,422 |
| Emanuel Montenegrino | 19,000 | Nil | Nil | Nil | Nil | 110,860 | 129,860 |
| William Fleming | 19,000 | Nil | Nil | Nil | Nil | Nil | 19,000 |
Notes:
| (1) | Please see "Statement of Executive Compensation - Summary Compensation Table for Named Executive Officers" above for details of compensation paid by the Company to those directors who are also NEOs. |
| (2) | The Black Scholes valuation methodology was used to determine the value on the date of grant. |
| (3) | Mr. Robert Hedley resigned as a Director of the Company effective February 16, 2015. |
During the fiscal year ended September 30, 2014, $53,000 in director's fees were paid to directors of the Company for services in their capacity as directors of the Company and $215,042 was paid to directors of the Company for consultant services in their capacity as directors of the Company.
During the fiscal year ended September 30, 2014, the Company paid Mr. Tim Fealey $8,000/month for providing senior industry advisory services to the Company pursuant to a consulting agreement dated March 1, 2012 (the "Fealey Agreement"). The term of the Fealey Agreement is for three years and may be extended upon written notice at least 5 days prior to the expiry date. The Fealey Agreement will not expire unless terminated by Mr. Fealey.
A private company, owned by Mr. Hedley, Hedley Enterprises Ltd. ("HEL"), a Canadian distributer of natural health products, entered into a distribution agreement with the Company on April 20, 2012, whereby HEL was granted the exclusive right to purchase, resell and distribute various health products, including certain nitric oxide natural products that may be manufactured by the Company during the term of the agreement. The term of the agreement is for five years, renewable at the sole discretion of the Company. No consideration was paid to Mr. Hedley pursuant to this agreement during the financial year ended September 30, 2014.
A law firm of which Mr. Sorocco is a 1/3 partner provides legal services to the Company from time to time.
During the year ended September 30, 2014, the Company paid this law firm $75,618 (fiscal year ended September 30, 3013: $33,150). Directors are entitled to be reimbursed for reasonable expenditures incurred in performing their duties as directors.
73
Directors are also entitled to participate in the Option Plan (see "Equity Compensation Plans" below for details), which is designed to give each option holder an interest in preserving and maximizing shareholder value in the longer term. Individual grants are determined by an assessment of each individual director's current and expected future performance, level of responsibilities and the importance of his position and contribution to the Company.
Incentive Plan Awards for Non-Executive Directors
Outstanding Option-Based Awards - Non-Executive Directors
The following table provides the option-based awards outstanding to the non-executive directors of the Company as of September 30, 2014.
| Name | Option-based Awards | Share-based Awards | ||||
| Number of securities underlying unexercised options | Option exercise price ($) | Option expiration date | Value of unexercised in-the-money options(1) ($) | Number of shares that have not vested | Market or payout value of share-based awards that have not vested | |
| Tim Fealey | 200,000 | 0.35 | May 12, 2016 | $5,000 | N/A | N/A |
| 75,000 | 0.10 | Dec. 24, 2017 | $20,625 | N/A | N/A | |
| Douglas Sorocco | 100,000 | 0.35 | May 12, 2016 | $2,500 | N/A | N/A |
| 75,000 | 0.10 | Dec. 24, 2017 | $20,625 | N/A | N/A | |
| 200,000 | 0.64 | July 22, 2019 | $53,000 | N/A | N/A | |
| Robert Hedley(2) | 50,000 | 0.64 | July 22, 2019 | $13,250 | N/A | N/A |
| Guy Dancosse | 100,000 | 0.33 | Sept. 16, 2019 | $4,500 | N/A | N/A |
| Emanuel Montenegrino | 0 | N/A | N/A | $0 | N/A | N/A |
| William Fleming | 0 | N/A | N/A | $0 | N/A | N/A |
Notes:
| (1) | The value of "in-the-money-options" is calculated based on the difference between the market value of the Company's Common Shares underlying the options at the end of the most recently completed financial year and the exercise price of the options. The last trading price of the Company's Common Shares on the CSE as of September 30, 2014 was $0.375 per share. |
| (2) | Mr. Robert Hedley resigned as a Director of the Company effective February 16, 2015. |
74
Option-Based Awards - Directors
The following table summarizes the value of incentive plan awards vested or earned by non-NEO directors as of September 30, 2014.
| Name | Option-based awards - Value vested during the year(1) ($) | Share-based awards - Value vested during the year ($) | Non-equity incentive plan compensation - Value earned during the year ($) |
| Tim Fealey | $25,625 | N/A | N/A |
| Douglas Sorocco | $76,125 | N/A | N/A |
| Robert Hedley(2) | $13,250 | N/A | N/A |
| Guy Dancosse | $4,500 | N/A | N/A |
| Emanuel Montenegrino | N/A | N/A | N/A |
| William Fleming | N/A | N/A | N/A |
Notes:
| (1) | Value vested or earned during the year means the aggregate dollar value that would have been realized if the options under the option-based award had been exercised on the vesting date. This amount is calculated by determining the difference between the market price of the underlying securities at exercise and the exercise or base price of the options under the option-based award on the vesting date. |
| (2) | Mr. Robert Hedley resigned as a Director of the Company effective February 16, 2015. |
Pension Plan Benefits - Directors
The Company does not have a pension plan that provides for payments to the directors at, following or in connection with retirement.
Equity Compensation Plan Information
We have no long-term incentive plans other than the stock option plan described below:
The following table sets out securities authorized for issuance under equity compensation plans as of September 30, 2014.
| Plan Category | Number of Securities to be issued upon exercise of outstanding options, warrants and rights | Weighted-average exercise price of outstanding options, warrants and rights | Number of securities remaining available for future issuance under equity compensation plans |
| Equity compensation plans approved by Shareholders (Option Plan)(1) | 2,575,000 | $0.17 | 283,408 |
| Equity compensation plans not approved by Shareholder | N/A | N/A | N/A |
75
Note:
| (1) | The aggregate number of Common Shares issuable upon the exercise of all options granted under the Option Plan shall not exceed 10% of the issued and outstanding Common Shares from time to time. |
2012 Incentive Stock Option Plan
The Company maintains a stock option plan dated for reference June 18, 2012 (the "Option Plan") for its directors, officers, employees and consultants. The Option Plan was approved by the shareholders of the Company at its annual general meeting held on July 23, 2012. As of the date of April 27, 2015, the Company has 3,123,100 stock options outstanding (4.48% of the issued capital as at such date) and an additional 3,843,695 stock options were available for grant (5.52% of the issued capital as at such date).
The purpose of the Option Plan is to advance the interests of the Company by encouraging the directors, officers, employees and consultants of the Company to acquire Common Shares thereby increasing their proprietary interest in the Company, encouraging them to remain associated with the Company and furnishing them with additional incentive in their efforts on behalf of the Company in the conduct of its affairs.
Under the Option Plan, the aggregate number of optioned shares that may be issued may not exceed 10% of the number of issued and outstanding Common Shares of the Company at the time of granting of options.
The Board has the discretion to grant options pursuant to the terms of the Option Plan. Options may be granted to eligible persons, being: directors, executive officers, employees or consultants. Limitations on issue include:
| (a) | no more than 5% of the issued Common Shares of the Company, calculated at the date of the grant of options, may be granted to any one optionee in any 12 month period (unless disinterested shareholder approval is obtained where permitted by applicable regulators); | |
| (b) | no more than an aggregate of 1% of the issued Common Shares of the Company, calculated at the date of the grant of options, may be granted to all employees conducting investor relations activities within any 12 month period (which percentage interest may be increased from an aggregate of 1% to an aggregate of 2% if permitted by applicable regulators); and | |
| (c) | if required by applicable regulators, no more than 2% of the issued Common Shares of the Company, calculated at the date of the grant of options, may be granted to any one consultant in any 12 month period. |
Pursuant to the Option Plan, the exercise price of options is set by the Board and cannot be less than the closing market price of the Common Shares on the trading day immediately prior to the date of grant less any allowable discounts if permitted under applicable exchange policies. Options may be granted for a maximum of 10 years from the date of grant.
All options granted under the Option Plan are non-transferable and non-assignable.
Where permitted by applicable regulators, vesting provisions are at the sole discretion of the Board except that options granted to consultants conducting investor relations activities will vest, at a minimum, over a period of not less than 12 months as to 25% on the date that is three months from the date of grant and a further 25% on each successive date that is three months from the date of the previous vesting.
76
Any reduction in exercise price of an option previously granted to an insider requires disinterested shareholder approval. Any other amendments to the Option Plan or to any options granted under the Option Plan will be required to comply with applicable Exchange policies.
Options will expire immediately upon the optionee leaving his or her employment/office except that:
| (a) | in the case of death of an optionee, any vested options held by the deceased at the date of death will become exercisable by the optionee's estate until the earlier of one year after the date of death and the date of expiration of the term otherwise applicable to such option; | |
| (b) | options granted to an optionee may be exercised in whole or in part by the optionee for a period of 30 days after the optionee ceases to be employed/provide services but only to the extent that such optionee was vested in the option at the date the optionee ceased to be employed/provide services; and | |
| (c) | in the case of an optionee dismissed from employment/service for cause, such options, whether vested or not, will immediately terminate without right to exercise same. |
Corporate governance relates to activities of the Board, the members of which are elected by and are accountable to the shareholders, and takes into account the role of the individual members of management who are appointed by the Board and who are charged with the day-to-day management of the Company. The Board is committed to sound corporate governance practices, which are both in the interest of its shareholders and contribute to effective and efficient decision making. In Canada, National Instrument 58-101 - Disclosure of Corporate Governance Practices ("NI 58-101") requires that each reporting company in Canada disclose its corporate governance practices on an annual basis. The Company's general approach to corporate governance is summarized below.
Board of Directors
Independence
The Company's Board is comprised of five (5) directors: Tim Fealey, Douglas Sorocco, Bill Fleming, Manny Montenegrino and Guy Dancosse.
Section 1.4 of National Instrument 52-110 - Audit Committee ("NI 52-110") sets out the standard for director independence. Under NI 52-110, a director is independent if he has no direct or indirect material relationship with the Company in Canada. A material relationship is a relationship which could, in the view of the Board, be reasonably expected to interfere with the exercise of a director's independent judgment. NI 52-110 also sets out certain situations where a director will automatically be considered to have a material relationship to the Company.
Applying the definition set out in section 1.4 of NI 52-110, the only members of the Board who are independent are Manny Montenegrino and Guy Dancosse. Bill Fleming is not independent by virtue of the fact that he is an executive officer of the Company. Mike Withrow is not independent since he was an executive officer of the Company. Mr. Fealey is not independent as he is a paid consultant of the Company. Mr. Hedley is not independent as a private company of which he is a shareholder and director has an exclusive distribution agreement with the Company. Mr. Sorocco is not independent as a law firm, of which he is a 1/3 partner, provides legal services to the Company from time to time.
The directors are responsible for managing and supervising the management of the business and affairs of the Company. Each year, the Board must review the relationship that each director has with the Company in order to satisfy themselves that the relevant independence criteria have been met.
77
Nomination Committee
The Board does not have a nominations committee or a formal procedure with respect to the nomination of directors. Nominees have historically been recruited by the efforts of existing Board members, and the recruitment process has involved both formal and informal discussions among Board members. New nominees must have at track record in general business management, special expertise in an area of strategic interest to the Company, the ability to devote the required time, show support for the Company's mission and strategic objectives and have a willingness to serve.
The Board monitors, but does not formally assess, the performance of individual Board members and their contributions. The Board does not, at present, have a formal process in place for assessing the effectiveness of the Board as a whole, its committees or individual directors, but will consider implementing one in the future should circumstances warrant. Based on the Company's size, its stage of development and the limited number of individuals on the Board, the Board considers a formal assessment process to be inappropriate at this time.
Compensation Committee
The Board does not currently have a compensation committee or a formal procedure with respect to determining compensation for the directors and the CEO. All employment, consulting or other compensation arrangements between the Company, or its subsidiary, and the directors or executive officers are considered and approved by disinterested members of the Board. The Board annually reviews and approves corporate goals and objectives relevant to the compensation of the CEO, evaluates the CEO's performance in light of those goals and objectives and sets the CEO's compensation level based on this evaluation. The Board meets without the presence of other executive officers when approving the CEO's compensation but may invite the CEO to be present during approval of other executive officer compensation.
Audit Committee
The Company has an Audit Committee whose primary function is to assist the Board in fulfilling its financial oversight responsibilities by reviewing the financial reports and other financial information provided by the Company to regulatory authorities and Shareholders, the Company's systems of internal controls regarding finance and accounting, and the Company's auditing, accounting and financial reporting processes.
Audit Committee Charter
The Audit Committee operates under a written charter that sets out its responsibilities and composition requirements.
Composition of the Audit Committee
As at the year ended September 30, 2014, the Company's Audit Committee is comprised of members consisting of Mike Withrow, Tim Fealey and Douglas Sorocco. The following table sets out the
78
names of the members of the Audit Committee and whether they are 'independent' and 'financially literate' for the purposes of NI 52-110.
| Name of Member | Independent(1) | Financially Literate(2) |
| Mike Withrow(3) | No | Yes |
| Tim Fealey | No | Yes |
| Douglas Sorocco | No | Yes |
Notes:
| (1) | To be independent, a member of the Audit Committee must not have any direct or indirect 'material relationship' with the Company. A material relationship is a relationship which could, in the view of the Board, reasonably interfere with the exercise of a member's independent judgment. Accordingly, an executive officer of the Company is not independent, nor is a director that is paid consulting fees for non-director services provided to the Company. |
| (2) | To be considered financially literate, a member of the Audit Committee must have the ability to read and understand a set of financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of the issues that can reasonably be expected to be raised by the Company's financial statements. |
| (3) | Mr. Withrow resigned as an officer and director of the Company effective February 1, 2015. He will be replaced on the Audit Committee in the upcoming quarter. |
The following table shows the major shareholders that are beneficial owners of 5% or more of our common stock as of the date of this prospectus. Unless otherwise indicated, each owner has sole voting and investment powers over his shares of common stock. Unless otherwise indicated, beneficial ownership is determined in accordance with the Rule 13d-3 promulgated under the Securities and Exchange Act of 1934, as amended, and includes voting or investment power with respect to shares beneficially owned.
All of our shareholders, including the shareholder(s) listed below, have the same voting rights attached to their common shares. See "Description of Securities - Common Stock." Unless otherwise noted below, each shareholder's address is Suite 1040 - 885 West Georgia Street, Vancouver, British Columbia, V6C 3H1.
A description of any material relationship that our principal shareholders have had with us or any of our predecessors or affiliates within the past three years is included under "Certain Relationships and Related Party Transactions."
| Name of Shareholder | Number of Common Shares | Percentage of Class(1) |
Nicholas Brusatore(2) | 3,908,500 | 5.61% |
Notes:
| (1) | Based on 69,667,951 issued and outstanding common shares of the Company. |
| (2) | Nicholas Brusatore is a former director of the Company who resigned on November 29, 2013. |
79
Common Stock
The Company is authorized to issue an unlimited number of shares of common stock without par value. As of the date of this prospectus, the Company has 69,667,951 shares of common stock outstanding.
Holders of common stock are entitled to one vote for each share held of record on all matters presented to shareholders.
Holders of common stock are entitled to receive such dividends as may be declared by the Board out of funds available and, in the event of liquidation, to share pro rata in any distribution of the Company's assets after paying off liabilities. It is not anticipated that dividends will be paid in the foreseeable future.
Holders of common stock do not have pre-emptive rights to subscribe to any additional shares which may be issued in the future. There are no conversions, redemptions, sinking funds or similar provisions regarding the common stock. All outstanding shares of common stock are fully paid and non-assessable.
Stock Options
The Company has a share purchase option plan (dated June 18, 2012) which specifies that a maximum of 10% of the issued and outstanding common shares of the Company may be reserved for issuance pursuant to the exercise of share options. The term of the share options granted are fixed by the board of directors and are not to exceed ten years. The exercise prices of the share options shall not be less than the closing price of the Company's common shares on the day preceding the day on which the directors grant the share purchase options, less any discount permitted by the CSE. Vesting of options will be at the discretion of the Board.
As of the date of this prospectus, the Company has 3,123,100 outstanding stock options to purchase common shares of the Company.
Warrants
As of the date of this prospectus, the Company has 8,948,903 outstanding share purchase warrants to purchase common shares of the Company.
Loans and Convertible Notes
On March 14, 2014, the Company entered into a one-year loan agreement with Phytalytics with principal amount of $66,318 (US$60,000). The loan to Phytalytics matures on March 14, 2015 and bears interest at 10% annually.
On September 3, 2014, the Company entered into a one-year loan agreement with Phytalytics with principal amount of US$19,690. The loan matures on September 3, 2015 and bears interest at 5% annually on any balance unrepaid after one year.
On September 29, 2014, the Company entered into a one-year loan agreement with Phytalytics with principal amount of US$26,000. The loan matures on September 29, 2015 and bears interest at 10% annually on any balance unrepaid after one year.
During the year ended September 30, 2014, the Company provided a short-term loan of $24,543 to IPS (September 30, 2013 - $nil). This amount remains outstanding as at September 30, 2014 (September 30, 2013 - $nil).
80
On December 18, 2014, the Company provided a short-term loan to Terracity Lawrence LLC ("Terracity") for an amount of $116,290 (USD 100,000). This loan matures on February 18, 2015. Under the loan agreement, the Company has an option to convert outstanding principal and interest amount to Terracity's common shares at a conversion price of $0.05 upon Terracity successful closing of its public transaction on the TSX Venture Exchange.
Dividends
We have not paid a cash dividend on our common stock and do not expect to pay a cash dividend in the foreseeable future. Our board of directors has the sole authority to declare dividends. Our payment of dividends in the future will depend on our earnings, capital requirements, expansion plans, financial condition and other relevant factors.
Transfer Agent and Registrar
The Company's transfer agent and registrar is Computershare Investor Services Inc. in Canada in Vancouver and Toronto. Transfers may be effected at, and registration facilities are maintained in British Columbia, Canada at 3rd Floor, 510 Burrard Street, Vancouver, British Columbia, V6C 3B9.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
The following includes a summary of transactions since October 1, 2013 to which we have been a party in which the amount involved exceeded or will exceed $120,000, and in which any of our board members, executive officers or, to our knowledge, beneficial owners of more than 5% of our capital shares or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described under "Management Compensation." We also describe below certain other transactions with our board members, executive officers and shareholders.
Management transactions
Management transactions with related parties for the year ended September 30, 2014 is as follows:
| Name | Position | Management and consulting fees | Director's fees | Share-based compensation | Total |
| Mike Withrow(1) | CEO | $ 172,134 | $ - | $ 2,226,591 | $ 2,398,725 |
| Rene David(2) | CFO | 127,425 | - | 983,105 | 1,110,530 |
| Terence Fealey(3) | Director | 104,182 | - | - | 104,182 |
| Brazos Minshew(4) | Consultant | - | 30,333 | - | 30,333 |
| Guy Dancosse(5) | Director | - | 15,000 | 20,422 | 35,422 |
| Douglas Sorocco(6) | Director | - | - | 357,306 | 357,306 |
| Robert Hedley(7) | Director | - | - | 89,326 | 89,326 |
| Dunlap Codding, P.C.(8) | Consultant | 75,618 | - | 325,683 | 401,301 |
| Emanuel "Manny" Montenegrino(9) | Director | 110,860 | 19,000 | - | 129,800 |
| William Fleming(10) | Director | - | 19,000 | - | 19,000 |
| $ 590,219 | $ 83,333 | $ 4,002,433 | $ 4,675,985 | ||
81
Notes:
| (1) | Mr. Withrow resigned as President, CEO and Director of the Company effective February 1, 2015. The Company paid management fees of $172,134 to Chiron Capital Inc., a company controlled by Mr. Withrow during the year ended September 30, 2014 (September 30, 2013 - $142,500). |
During the year ended September 30, 2014, 3,230,000 stock options (September 30, 2013 - 390,000), with an estimated fair value of $2,226,591 (September 30, 2013 - $14,549) were granted by the Company to Mr. Withrow and Chiron Capital Inc., a company controlled by Mr. Withrow.
During the year ended September 30, 2014, 890,000 options were exercised for cash proceeds of $124,000 (September 30, 2013 - nil) and 1,150,000 options were cancelled (September 30, 2013 - nil).
During the year ended September 30, 2014, 1,360,000 warrants were exercised for cash proceeds of $220,000 (September 30, 2013 - nil).
During the year ended September 30, 2014, 5,000,000 common shares (September 30, 2013 - 747,500), with an estimated fair value of $297,500 (September 30, 2013 - $119,475), were issued by the Company to Mr. Withrow to settle a trade payable of $180,000 (September 30, 2013 - $68,988).
At September 30, 2014, $18,270 due to Mr. Withrow was included in trade and other payables (September 30, 2013 - $142,344). In addition, as at September 30, 2013, operating cash of $260,159 provided by Mr. Withrow and $30,000 in common shares due from the Company were included in short-term loans and accrued interest of $14,468 was included in interest payable. This amount was fully paid during the year ended September 30, 2014.
| (2) | The Company paid management fees of $127,425 to Crimson Opportunities Ltd., a company controlled by Mr. David during the year ended September 30, 2014 (September 30, 2013 - $nil). |
During the year ended September 30, 2014, 2,315,000 stock options (September 30, 2013 - nil), with an estimated fair value of $983,105 (September 30, 2013 - $nil) were granted by the Company to Crimson Opportunities Ltd., a company controlled by Mr. David.
During the year ended September 30, 2014, 550,000 options were exercised for cash proceeds of $76,000 (September 30, 2013 - nil) and 580,000 options were cancelled (September 30, 2013 - nil).
During the year ended September 30, 2014, 625,000 common shares (September 30, 2013 - nil), with an estimated fair value of $31,250 (September 30, 2013 - $nil) were issued by the Company to Crimson Opportunities Ltd. to settle a trade payable of $31,250 (September 30, 2013 - $nil).
82
During the year ended September 30, 2014, the Company sub-leased a facility from Crimson Opportunities Ltd. Until the Company secures a license, the Company will use the facility to manufacture and warehouse its proprietary Biocube systems.
Minimum lease payments are as follows:
| Year | |||
| 2015 | 36,450 | ||
| 2016 | 36,450 | ||
| 2017 | 36,450 | ||
| 2018 | 36,450 | ||
| 2019 | 24,300 | ||
| $ | 170,100 | ||
During the year ended September 30, 2014, the Company recognized $11,340 in related lease expense (September 30, 2013 - $nil). The amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014.
At September 30, 2014, $10,809 due to Mr. David was included in trade and other payables (September 30, 2013 - $nil).
| (3) | The Company paid consulting fees of $104,182 to Mr. Fealey during the year ended September 30, 2014 (September 30, 2013 - $97,816). |
During the year ended September 30, 2014, nil stock options (September 30, 2013 - 75,000), with an estimated fair value of $nil (September 30, 2013 - $2,798) were granted by the Company to Mr. Fealey.
At September 30, 2014, $235,418 due to Mr. Fealey was included in trade and other payables (September 30, 2013 - $118,302).
| (4) | During the year ended September 30, 2014, the Company issued 66,666 common shares to Mr. Minshew for the directors' fees of $30,333 (September 30, 2013 - $nil). |
During the year ended September 30, 2014, 244,500 warrants were exercised for cash proceeds of $61,125 (September 30, 2013 - nil).
| (5) | During the year ended September 30, 2014, the Company issued 50,000 common shares to Mr. Dancosse for the directors' fees of $15,000 (September 30, 2013 - $nil). |
During the year ended September 30, 2014, 100,000 stock options (September 30, 2013 - nil), with an estimated fair value of $20,422 (September 30, 2013 - $nil) were granted by the Company to Mr. Dancosse.
| (6) | During the year ended September 30, 2014, 400,000 stock options (September 30, 2013 - 75,000), with an estimated fair value of $357,306 (September 30, 2013 - $2,798) were granted by the Company to Mr. Sorocco. |
During the year ended September 30, 2014, 200,000 options were cancelled (September 30, 2013 - nil).
| (7) | Mr. Hedley resigned as a director of the Company effective February 16, 2015. During the year ended September 30, 2014, 100,000 stock options (September 30, 2013 - 75,000), with an estimated fair value of $89,326 (September 30, 2013 - $2,798) were granted by the Company to Mr. Hedley. |
During the year ended September 30, 2014, 75,000 options were exercised for cash proceeds of $7,500 (September 30, 2013 - nil) and 50,000 options were cancelled (September 30, 2013 - nil).
| (8) | The Company paid legal fees of $75,618 to Dunlap Codding, P.C., of which of Mr. Sorocco is a one-third partner, during the year ended September 30, 2014 (September 30, 2013 - $33,150). |
83
During the year ended September 30, 2014, 200,000 stock options (September 30, 2013 - nil), with an estimated fair value of $325,683 (September 30, 2013 - $nil) were granted by the Company to Dunlap Codding, P.C.
During the year ended September 30, 2014, 200,000 options were cancelled (September 30, 2013 - nil).
During the year ended September 30, 2014, 43,400 common shares (September 30, 2013 - nil), with a deemed value of $78,626 (September 30, 2013 - $nil) were issued by the Company to Dunlap Codding, P.C. to settle a trade payable of $68,591 (September 30, 2013 - $nil).
At September 30, 2013, $31,040 (September 30, 2014 - $45,717) due to Dunlap Codding, P.C. was included in trade and other payables.
| (9) | The Company paid consulting fees of $110,860 and directors' fees of $19,000 to Think Sharp, a company controlled by Emanuel Montenegrino, the director of the Company, during the year ended September 30, 2014 (September 30, 2013 - $nil). During the year ended September 30, 2014, the Company issued 80,000 common shares with a fair value of $36,880 in lieu of cash payments (September 30, 2013 - nil). |
At September 30, 2013, $14,187 (September 30, 2013 - $nil) due to Think Sharp was included in trade and other payables.
| (10) | Mr. Fleming was appointed CEO of the Company effective February 1, 2015. During the year ended September 30, 2014, the Company issued 50,000 common shares to Mr. Fleming for the directors' fees of $19,000 (September 30, 2013 - $nil). |
Subsequent to September 30, 2014, 442,919 common shares with a fair value of US$74,320 issued to the Company's directors and officers for the services provided to the Company.
Subsequent to September 30, 2014, one of the directors of the Company forgave $235,418 in amounts owing to him for consulting services in exchange for cash settlement of $32,000.
Transactions with related parties have been in the normal course of operations and, in management's opinion, undertaken on the same terms and conditions as transactions with unrelated parties. These costs are measured at exchange amounts agreed upon by the parties.
We have not entered into any material contract other than in the ordinary course of business and other than those described below or in this prospectus.
On March 1, 2012, the Company entered into a 3-year consulting agreement with one of the directors of the Company. Under the agreement, the Company will pay US $8,000 per month to this director for consulting and research and development services. The contract expires on March 1, 2015 and if the contract is terminated at the Company's discretion, the director is entitled to receive three months' fees over and above the thirty-day notice period.
On April 20, 2012, the Company entered into an exclusive distribution agreement with Hedley Enterprises Ltd. ("Hedley") to purchase, resell and distribute Abattis' line of natural products in Canada. Under the terms of the Agreement Hedley has acquired the exclusive right to sell and distribute Abattis' products to all retail distribution channels, which include health food stores, grocery stores, fitness facilities, and similar retail establishments.
84
On May 21, 2012, the Company entered into a two-year finder's fee agreement with VentureCorp 2 Capital Inc. Under the agreement, the Company will pay to the finder a cash fee equal to 10% of the gross proceeds raised by the Company through the sale of shares or units to investors introduced by the finder. The Company will also issue to the finder non-transferable compensation warrants in an amount equal to 5% of the aggregate number of shares or units subscribed for by the investors introduced by the finder. Each finder's warrant will entitle the finder to acquire one common share of the Company, exercisable for a period of 12 months from closing date at an exercise price equal to the price per share or unit.
On November 1, 2012, the Company renewed a three-year office lease with Toro Holdings Ltd. The Company's minimum annual lease commitments over the next two years are as follows:
| Year | |||
| 2015 | $ | 36,291 | |
| 2016 | 3,063 | ||
| $ | 39,354 |
On December 27, 2012, the Company entered into a license agreement with Vertical Designs Ltd. ("Vertical Designs"). Under the agreement, the Company has been granted the exclusive, worldwide rights to a patent license, with the right to grant sublicenses, to use the Bio Pharma technology for growing products at licensed facilities, which products may only be used as ingredients in the pharmaceutical, nutraceutical, cosmetic and wellness markets. The royalty provisions of the license agreement reflect that: (i) the royalty payable on net sales of all products sold by Abattis was 4%; (ii) in consideration for the grant of the Company's right to grant sublicenses, the Company will pay to Vertical Designs Ltd. a sublicense royalty of 15% of any monies or other consideration that the Company receives from any sublicense; and (iii) after two years, the Company will be required to pay to Vertical Designs Ltd. a minimum royalty payment of $25,000 per year and if the combined royalty payments paid from (i) and (ii) above do not equal $25,000 in any given year then the Company will be permitted to top up such amount with a cash payment. Under the terms of the agreement, the patent license will revert to Vertical Designs Ltd. in certain circumstances, including: (i) if the Company terminates the agreement; (ii) if the Company materially breaches or defaults in the performance of the agreement and has not cured such default within 60 days, or in the case of failure to pay any amounts due, then within 30 days, after receiving written notice from Vertical Designs Ltd. specifying the breach; (iii) if the Company discontinues its business of producing ingredients for pharmaceutical, nutraceutical, cosmetic or wellness markets; (iv) if the Company fails to pay the annual $25,000 minimum royalty payment for any year ending after the second anniversary of the agreement; or (v) if the Company becomes insolvent, makes an assignment for the benefit of creditors or has a petition of bankruptcy filed by or against it, which petition is not vacated or otherwise removed within 90 days after the filing thereof. The Company also agreed to pay Vertical Designs $250,000 for the purchase and sale of six complete Vertical Designs operational units. The purchase price will be paid in instalments, dates and amounts are to be determined between the parties, with the first payment due on or before the earlier of five business days following the Company completing an equity and/or debt financing of any amount or the first business day in the seventh month following the date of the Bill of Sale.
On January 6, 2013, the Company entered into a two-year consulting agreement with Georges Laraque Management Inc. Under the agreement, the Company will pay consulting fees of $5,000 per month.
On October 1, 2013, the Company entered into a consulting agreement with Crimson Opportunities Ltd., a company controlled by the CFO of the Company for his services as CFO and COO. Under the agreement, the Company will pay annual consulting fees of $125,000 (excluding GST).
85
In December 2013, the Company's wholly owned subsidiary, Animo Wellness Corporation doing business as Medical Marijuana Labs ("MMLC"), signed a five-year lease with PurGenesis Technologies, Inc. for the lease of approximately 5,000 square feet of lab and production space at a cost of $120,000 in annual gross rent. Amino Wellness Corporation changed its name to iJuana Cannabis Inc. January 28, 2014.
On January 1, 2014, the Company entered into a consulting agreement with the Chiron Capital Corp., a company controlled by the CEO of the Company for his services as CEO. Under the agreement, the Company will pay annual consulting fees of $175,000 (excluding GST).
On January 1, 2014, the Company entered into a consulting agreement with Growing Strategies Inc. Under the agreement, the Company will pay monthly consulting fees of $4,000 (excluding GST).
On January 1, 2014, the Company entered into a consulting agreement with Voelpel Gold Medal Investment Ltd. Under the agreement, the Company will pay monthly consulting fees of $3,000 (excluding GST).
On February 1, 2014, the Company entered into a consulting agreement with Golden Straw Consulting Group Inc. Under the agreement, the Company will pay monthly consulting fees of $5,000 (excluding GST).
On March 16, 2014, the Company entered into a one-year consulting agreement with Think Sharp Inc. Under the agreement, the Company will pay monthly consulting fees of $10,000 and monthly administration fees of $100 (excluding GST) in cash and 6,000 common shares per month.
On April 1, 2014, the Company entered into a consulting agreement with TDM Financial as its full service investor relations firm to provide online branding, marketing, outreach, and investor relations management services. Pursuant to the agreement, the Company will pay US $25,000 consulting fee in common share to TDM Financial.
On April 10, 2014, the Company through its wholly owned subsidiary, Northern Vine Canada Inc. ("Northern Vine"), entered into a share exchange agreement with Experion Biotechnologies Inc. ("Experion"). Pursuant to the terms of the agreement, Experion and Northern Vine have exchanged 25% of each parties' issued and outstanding common shares. Abattis maintains a 75% ownership in Northern Vine. Experion is located in Greater Vancouver and has passed the review stage by Health Canada and is now awaiting a ready-to-build letter from Health Canada in order to proceed.
On April 30, 2014, the Company through its wholly owned subsidiary, Abattis Bioceuticals International Inc., acquired 34% interest in Instant Payment Systems LLC ("IPS"), a US entity based in Washington State, in consideration for $100,000 cash payments and 200,000 common shares of the Company with a fair value of $180,000. IPS was incorporated under the laws of Washington, US.
On June 25, 2014, the Company entered into an 18 month consulting agreement with Brazos Minshew for his services as the President of one of the Company's subsidiaries. Pursuant to the agreement, the Company will pay, for consulting services, an aggregate of 200,000 shares of the Company payable in monthly instalments for the period of July 1, 2014 to December 31, 2014. Following this period, the Company will pay the consultant $5,000 per month.
On July 31, 2014, the Company entered into a sub-licensing agreement with its subsidiary, Biocube Green Grow Systems Corp. ("Biocube"), for the non-exclusive use of Vertical Design's vertical growing systems for which Abattis owns certain exclusive worldwide rights.
86
On August 5, 2014, the Company entered into a US$5 million Standby equity financing agreement with Kodiak, a Newport Beach, CA-based institutional investor. Under the terms of the agreement between the parties, the Company was to file a registration statement with the SEC covering the Abattis shares that may be issued to Kodiak under this financing. After the SEC has declared the registration statement related to the transaction effective, the Company would have the right at its sole discretion over a period of 24 months to sell up to US$5 million of common shares to Kodiak. The Company agreed to pay Kodiak a 5% commitment fee in shares. Of these shares, 277,373 shares were held in escrow in accordance with an escrow agreement to be released if and when the Company accessed the facility. On August 7, 2014, 243,460 shares were issued to Kodiak with a deemed value of US$102,253 ($111,991). The Company terminated this agreement in writing on January 26, 2015.
On August 6, 2014, the Company through its wholly owned subsidiary, Biocube, entered into a worldwide non-exclusive licensing agreement with TerraSphere Systems LLC ("TerraSphere") for the company's proprietary and patented vertical farming technology. The TerraSphere license complements Biocube's previously announced sublicensing agreement for Vertical Design's vertical farming technology.
During the year ended September 30, 2014, the Company entered into 34 month office lease. The Company's minimum annual lease payments are as follows:
| Year | |||
| 2015 | $ | 68,860 | |
| 2016 | 71,727 | ||
| 2017 | 73,962 | ||
| $ | 214,549 |
During the year ended September 30, 2014, the Company paid $5,591 lease payments (September 30, 2013 - $nil). These amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014.
During the year ended September 30, 2014, the Company entered into 13 month facility lease. The Company's minimum annual lease payments are as follows:
| Year | |||
| 2015 | $ | 15,618 |
During the year ended September 30, 2014, the Company paid $10,879 lease payments (September 30, 2013 - $nil). These amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014.
On January 15, 2015, the Company entered into a manufacturing partnership with Empirical Labs, a nutraceutical manufacturing facility. This relationship with Empirical Labs will allow the Company the exclusive license rights for cannabidiol rich hemp oil products delivered directly to licensed physicians and dispensaries.
On January 28, 2015, the Company entered into an Investment Agreement with Dutchess, pursuant to which Dutchess will purchase up to $25,000,000 of our common stock for a period of thirty-six (36) months commencing on the effective date of the Form F-1 Registration Statement. On January 28, 2015 and as amended April 1, 2015, the Company also entered into a Registration Rights Agreement with Dutchess, pursuant to which the Company has agreed to issue and sell to Dutchess up to 180,000,000
87
shares of the Company's common stock, with no par value per share, to be purchased pursuant to the terms and subject to the conditions set forth in the Investment Agreement.
As of April 27, 2015, four claims were pending against the Company. The plaintiffs are claiming an aggregate of approximately $474,000. The outcomes of these claims are not determinable and therefore no amounts have been recorded for any potential payments, which may have to be made. Details about each of these claims follows.
On September 20, 2012, Sasko Despotovski filed a statement of claim in the Provincial Court of British Columbia against the Company for CDN$ 37,356 plus interest. Mr. Despotovski alleges the Company breached a settlement agreement relating to his employment with the Company. The Company believes it has complied with the settlement agreement and no amount is owing. The Court has decided in Despotovski's favour, however, the amount has yet to be determined.
On February 24, 2012, 0740567 B.C. Ltd. (Ted Nitta) filed a statement of claim in the Supreme Court of British Columbia against the Company for $145,000 plus costs, interest, and punitive damages. Mr. Nitta appears to be alleging breach of contract in the statement of claim. The Company denies the claim in its entirety.
On February 18, 2014, Linda Byer doing business as Byer Advertising & Public Relations filed a statement of claim against the Company in the Supreme Court of British Columbia for U.S. $23,000 plus interest and costs. Ms. Byer alleges breach of contract for marketing services. The Company has filed counter-claim for U.S. $29,000, return of shares plus interest and costs. The Company alleges Ms. Byer failed to provide any services or alternatively provided sub-standard services.
On November 11, 2014, the Company obtained an Emergency Temporary Restraining Order against Herbal Analytics, LLC; James Baxter; Kaleb Lund; Lauren Hilty; Erin Leary; Affinor Growers, LLC and Nicholas Brusatore (the "Defendants") regarding the alleged infringement of intellectual property owned by the Company. On December 25, 2014, the Company obtained a preliminary injunction from the Washington state court in King County against the Defendants, ordering that they:
| (a) | cease and desist from any and all use of PhytaLabs trade secrets and confidential information and documents; |
| (b) | are restrained from copying, transferring, using or disclosing to any other person or entity any documentation taken from PhytaLab; |
| (c) | retain and preserve all existing documents and files that mention, refer to, or are derived from PhytaLab, PhytaLab and the Company's customers, or PhytaLab and the Company's prospective customers; and |
| (d) | keep a detailed, complete, and accurate accounting of its business operations. |
On April 10, 2015, The Company amicably resolved and dismissed the Washington litigation against Affinor Growers, LLC and Nick Brusatore. The Company will continue to pursue its claims against Herbal Analytics, LLC, James Baxter, Kaleb Lund, Lauren Hilty and Erin Leary.
On January 8, 2015, John Gregory Dennison filed a Notice of Civil Claim in the Supreme Court of British Columbia against the Company for breaching the consulting contract which the plaintiff should entitle for 75,000 options of the Company. Legal advice received supports the Company's belief that the claim is
88
without merit. The outcome of this claim are not determinable and therefore no amounts have been recorded for any potential payments which may have to be made.
On January 15, 2015, White Rock Holdings Inc. filed a Notice of Civil Claim in the Supreme Court of British Columbia against the Company or amounts payable to him which he claims were to be settled in common shares. The plaintiff has claimed damages of approximately $300,000. Abattis has offered to settle with White Rock Holdings Inc.; however, no settlement between the parties has been reached.
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS
On May 16, 2014, the Company changed its auditors from Hay & Watson, Chartered Accountants (the "Former Auditors") to MNP LLP, Chartered Accountants (the "Successor Auditors"). The Former Auditors resigned as the Company's auditors effective May 16, 2014 at the request of the Company. The resignation of the Former Auditors and the appointment of the Successor Auditors was considered and approved by the Company's audit committee of the Company's board of directors. There were no reservations contained in the Former Auditors' report on any of the Company's financial statements for the period commencing at the beginning of the Company's most recently completed financial year and ending on the date of resignation of the Former Auditors. Furthermore, there have been no reportable events (including disagreements, unresolved issues, and consultations) in connection with the audits of the two most recently completed financial years of the Company and with respect to any subsequent period to date.
There are currently no laws, decrees, regulations or other legislation in Canada that restricts the export or import of capital or that affects the remittance of dividends, interest or other payments to non-resident holders of our securities other than withholding tax requirements. There is no limitation imposed by Canadian law or by our Articles of Incorporation or our other organizational documents on the right of a non-resident of Canada to hold or vote our common shares, other than as provided in the North American Free Trade Agreement Implementation Act (Canada) and in the Investment Canada Act, as amended by the World Trade Organization Agreement Implementation Act.
The Investment Canada Act requires notification and, in certain cases, advance review and approval by the Government of Canada of the acquisition by a "non-Canadian" of "control of a Canadian business", all as defined in the Investment Canada Act. Generally, the threshold for review will be higher in monetary terms, and in certain cases an exemption will apply, for an investor ultimately controlled by persons who are nationals of a WTO Member or have the right of permanent residence in relation thereto.
Canadian Income Tax Consequences
We consider that the following summary fairly describes the principal Canadian federal income tax consequences applicable to a holder of our common shares who at all material times deals at arm's length with our company, who holds all common shares as capital property, who is resident in the United States, who is not a resident of Canada and who does not use or hold, and is not deemed to use or hold, his common shares of our company in connection with carrying on a business in Canada (a "non-resident
89
holder"). It is assumed that the common shares will at all material times be listed on a stock exchange that is prescribed for purposes of the Income Tax Act (Canada) (the "ITA") and regulations thereunder. Investors should be aware that the Canadian federal income tax consequences applicable to holders of our common shares will change if, for any reason, we cease to be listed on a prescribed stock exchange. Accordingly, holders and prospective holders of our common shares should consult with their own tax advisors with respect to the income tax consequences of them purchasing, owing and disposing of our common shares should we cease to be listed on a prescribed stock exchange.
This summary is based upon the current provisions of the ITA, the regulations there under, the Canada-United States Tax Convention as amended by the Protocols thereto (the "Treaty") as at the date of the registration statement and the currently publicly announced administrative and assessing policies of the Canada Revenue Agency (the "CRA"). This summary does not take into account Canadian provincial income tax consequences. This description is not exhaustive of all possible Canadian federal income tax consequences and does not take into account or anticipate any changes in law, whether by legislative, governmental or judicial action. This summary does, however, take into account all specific proposals to amend the ITA and regulations there under, publicly announced by the Government of Canada to the date hereof.
This summary does not address potential tax effects relevant to our company or those tax considerations that depend upon circumstances specific to each investor. Accordingly, holders and prospective holders of our common shares should consult with their own tax advisors with respect to the income tax consequences to them of purchasing, owning and disposing of common shares in our company.
Dividends
The ITA provides that dividends and other distributions deemed to be dividends paid or deemed to be paid by a Canadian resident corporation (such as our company) to a non-resident of Canada shall be subject to a non-resident withholding tax equal to 25% of the gross amount of the dividend of deemed dividend. Provisions in the ITA relating to dividend and deemed dividend payments to and gains realized by non-residents of Canada, who are residents of the United States, are subject to the Treaty. The Treaty may reduce the withholding tax rate on dividends as discussed below.
Article X of the Treaty as amended by the US-Canada Protocol ratified on November 9, 1995 provides a 5% withholding tax on gross dividends or deemed dividends paid to a United States corporation which beneficially owns at least 10% of the voting stock of the company paying the dividend. In cases where dividends or deemed dividends are paid to a United States resident (other than a corporation) or a United States corporation which beneficially owns less than 10% of the voting stock of a company, a withholding tax of 15% is imposed on the gross amount of the dividend or deemed dividend paid. We would be required to withhold any such tax from the dividend and remit the tax directly to CRA for the account of the investor.
The reduction in withholding tax from 25%, pursuant to the Treaty, will not be available:
| (a) | if the shares in respect of which the dividends are paid formed part of the business property or were otherwise effectively connected with a permanent establishment or fixed base that the holder has or had in Canada within the 12 months preceding the disposition, or |
| (b) | the holder is a U.S. LLC which is not subject to tax in the U.S. |
90
The Treaty generally exempts from Canadian income tax dividends paid to a religious, scientific, literary, educational, or charitable organization or to an organization exclusively administering a pension, retirement or employee benefit fund or plan, if the organization is resident in the U.S. and is exempt from income tax under the laws of the U.S.
Capital Gains
A non-resident holder is not subject to tax under the ITA in respect of a capital gain realized upon the disposition of one of our shares unless the share represents "taxable Canadian property" to the holder thereof. Our common shares will be considered taxable Canadian property to a non-resident holder only if:
| (a) | the non-resident holder; |
| (b) | persons with whom the non-resident holder did not deal at arm's length - or |
| (c) | the non-resident holder and persons with whom he did not deal at arm's length, |
owned not less than 25% of the issued shares of any class or series of our company at any time during the five year period preceding the disposition. In the case of a non-resident holder to whom shares of our company represent taxable Canadian property and who is resident in the United States, no Canadian taxes will generally be payable on a capital gain realized on such shares by reason of the Treaty unless:
| (a) | the value of such shares is derived principally from real property (including resource property) situated in Canada, |
| (b) | the holder was resident in Canada for 120 months during any period of 20 consecutive years preceding, and at any time during the 10 years immediately preceding, the disposition and the shares were owned by him when he ceased to be a resident of Canada, |
| (c) | they formed part of the business property or were otherwise effectively connected with a permanent establishment or fixed base that the holder has or had in Canada within the 12 months preceding the disposition, or |
| (d) | the holder is a U.S. LLC which is not subject to tax in the U.S. |
If subject to Canadian tax on such a disposition, the taxpayer's capital gain (or capital loss) from a disposition is the amount by which the taxpayer's proceeds of disposition exceed (or are exceeded by) the aggregate of the taxpayer's adjusted cost base of the shares and reasonable expenses of disposition. For Canadian income tax purposes, the "taxable capital gain" is equal to one-half of the capital gain.
U.S. Federal Income Tax Consequences
The following is a discussion of the material United States Federal income tax consequences, under current law, applicable to a U.S. Holder (as defined below) of our common shares who holds such shares as capital assets. This discussion does not address all potentially relevant Federal income tax matters and it does not address consequences peculiar to persons subject to special provisions of Federal income tax law, such as those described below as excluded from the definition of a U.S. Holder. In addition, this discussion does not cover any state, local, or foreign tax consequences. (See "Canadian Federal Income Tax Consequences" above.)
The following discussion is based on the Internal Revenue Code of 1986, as amended (the "Code"), Treasury Regulations, published Internal Revenue Service ("IRS") rulings, published administrative positions of the IRS and court decisions that are currently applicable, any or all of which could be materially and adversely changed, possibly on a retroactive basis, at any time. In addition, this discussion
91
does not consider the potential effects, both adverse and beneficial, of any recently proposed legislation, which, if enacted, could be applied, possibly on a retroactive basis, at any time.
The discussion below does not address potential tax effects relevant to our company or those tax considerations that depend upon circumstances specific to each investor. In addition, this discussion does not address the tax consequences that may be relevant to particular investors subject to special treatment under certain U.S. Federal income tax laws, such as, dealers in securities, tax-exempt entities, banks, insurance companies, and non-U.S. Holders. Purchasers of the common stock should therefore satisfy themselves as to the overall tax consequences of their ownership of the common stock, including the State, local and foreign tax consequences thereof (which are not reviewed herein), and should consult their own tax advisors with respect to their particular circumstances.
U.S. Holders
As used herein, a "U.S. Holder" includes a beneficial holder of common shares of our company who is a citizen or resident of the United States, a corporation or partnership created or organized in or under the laws of the United States or of any political subdivision thereof, any trust if a US court is able to exercise primary supervision over the administration of the trust and one or more US persons have the authority to control all substantial decisions of the trust, any entity created or organized in the United States which is taxable as a corporation for U.S. tax purposes and any other person or entity whose ownership of common shares of our company is effectively connected with the conduct of a trade or business in the United States. A U.S. Holder does not include persons subject to special provisions of Federal income tax law, such as tax-exempt organizations, qualified retirement plans, financial institutions, insurance companies, real estate investment trusts, regulated investment companies, broker-dealers, non-resident alien individuals or foreign corporations whose ownership of our common shares is not effectively connected with the conduct of a trade or business in the United States and shareholders who acquired their shares through the exercise of employee stock options or otherwise as compensation.
Dividend Distribution on Shares of our Company
U.S. Holders receiving dividend distributions (including constructive dividends) with respect to the common shares of our company are required to include in gross income for United States Federal income tax purposes the gross amount of such distributions to the extent that we have current or accumulated earnings and profits, without reduction for any Canadian income tax withheld from such distributions. Such Canadian tax withheld may be deducted or may be credited against actual tax payable, subject to certain limitations and other complex rules, against the U.S. Holder's United States Federal taxable income. See "Foreign Tax Credit" below. To the extent that distributions exceed our current or accumulated earnings and profits, they will be treated first as a return of capital to the extent of the shareholder's basis in the common shares of our company and thereafter as gain from the sale or exchange of the common shares of our company. Preferential tax rates for net long term capital gains may be applicable to a U.S. Holder which is an individual, estate or trust.
In general, dividends paid on our common shares will not be eligible for the dividends received deduction provided to corporations receiving dividends from certain United States corporations.
Foreign Tax Credit
A U.S. Holder who pays (or who has had withheld from distributions) Canadian income tax with respect to the ownership of our common shares may be entitled, at the election of the U.S. Holder, to either a deduction or a tax credit for such foreign tax paid or withheld. This election is made on a year-by-year
92
basis and generally applies to all foreign income taxes paid by (or withheld from) the U.S. Holder during that year. There are significant and complex limitations which apply to the credit, among which is the general limitation that the credit cannot exceed the proportionate share of the U.S. Holder's United States income tax liability that the U.S. Holder's foreign source income bears to his or its world-wide taxable income. In determining the application of this limitation, the various items of income and deduction must be classified into foreign and domestic sources. Complex rules govern income such as "passive income", "high withholding tax interest", "financial services income", "shipping income" and certain other classifications of income. A U.S. Holder who is treated as a domestic U.S. corporation owning 10% or more of our voting stock is also entitled to a deemed paid foreign tax credit in certain circumstances for the underlying foreign tax of our company related to dividends received or Subpart F income received from us. (See the discussion below of Controlled Foreign Corporations). The availability of the foreign tax credit and the application of the limitations on the foreign tax credit are fact specific and holders and prospective holders of our common shares should consult their own tax advisors regarding their individual circumstances.
Disposition of Common Shares
If a "U.S. Holder" is holding shares as a capital asset, a gain or loss realized on a sale of our common shares will generally be a capital gain or loss, and will be long-term if the shareholder has a holding period of more than one year. However, gains realized upon sale of our common shares may, under certain circumstances, be treated as ordinary income, if we were determined to be a "collapsible corporation" within the meaning of Code Section 341 based on the facts in existence on the date of the sale (See below for definition of "collapsible corporation"). The amount of gain or loss recognized by a selling U.S. Holder will be measured by the difference between (i) the amount realized on the sale and (ii) his tax basis in our common shares. Capital losses are deductible only to the extent of capital gains. However, in the case of taxpayers other than corporations (U.S.) $3,000 ($1,500 for married individuals filing separately) of capital losses are deductible against ordinary income annually. In the case of individuals and other non-corporate taxpayers, capital losses that are not currently deductible may be carried forward to other years. In the case of corporations, capital losses that are not currently deductible are carried back to each of the three years preceding the loss year and forward to each of the five years succeeding the loss year.
A "collapsible corporation" is a corporation that is formed or availed principally to manufacture, construct, produce, or purchase prescribed types or property that the corporation holds for less than three years and that generally would produce ordinary income on its disposition, with a view to the stockholders selling or exchanging their stock and thus realizing gain before the corporation realizes two thirds of the taxable income to be derived from prescribed property. Prescribed property includes: stock in trade and inventory; property held primarily for sale to customers in the ordinary course of business; unrealized receivables or fees, consisting of rights to payment for noncapital assets delivered or to be delivered, or services rendered or to be rendered to the extent not previously included in income, but excluding receivables from selling property that is not prescribed; and property gain on the sale of which is subject to the capital gain/ordinary loss rule. Generally, a shareholder who owns directly or indirectly 5 percent or less of the outstanding stock of the corporation may treat gain on the sale of his shares as capital gain.
Other Considerations for U.S. Holders
In the following circumstances, the above sections of this discussion may not describe the United States Federal income tax consequences resulting from the holding and disposition of common shares of the
93
Registrant. Our management is of the opinion that there is little, if not, any likelihood that we will be deemed a "Foreign Personal Holding Company", a "Foreign Investment Company" or a "Controlled Foreign Corporation" (each as defined below) under current and anticipated conditions.
Foreign Personal Holding Company
If at any time during a taxable year more than 50% of the total combined voting power or the total value of our outstanding shares is owned, actually or constructively, by five or fewer individuals who are citizens or residents of the United States and 60% or more of our gross income for such year was derived from certain passive sources (e.g., from dividends received from its subsidiaries), we would be treated as a "foreign personal holding company." In that event, U.S. Holders that hold common shares in our capital would be required to include in income for such year their allocable portion of our passive income which would have been treated as a dividend had that passive income actually been distributed.
Foreign Investment Company
If 50% or more of the combined voting power or total value of our outstanding shares are held, actually or constructively, by citizens or residents of the United States, United States domestic partnerships or corporations, or estates or trusts other than foreign estates or trusts (as defined by the Code Section 7701(a)(31)), and we are found to be engaged primarily in the business of investing, reinvesting, or trading in securities, commodities, or any interest therein, it is possible that we might be treated as a "foreign investment company" as defined in Section 1246 of the Code, causing all or part of any gain realized by a U.S. Holder selling or exchanging our common shares to be treated as ordinary income rather than capital gains.
Passive Foreign Investment Company
A U.S. Holder who holds stock in a foreign corporation during any year in which such corporation qualifies as a passive foreign investment company ("PFIC") is subject to U.S. federal income taxation of that foreign corporation under one of two alternative tax methods at the election of each such U.S. Holder.
Section 1297 of the Code defines a PFIC as a corporation that is not formed in the United States and, for any taxable year, either (i) 75% or more of its gross income is "passive income," which includes interest, dividends and certain rents and royalties or (ii) the average percentage, by value (or, if the company is a controlled foreign corporation or makes an election, adjusted tax basis), of its assets that produce or are held for the production of "passive income" is 50% or more. For taxable years of U.S. persons beginning after December 31, 1997, and for tax years of foreign corporations ending with or within such tax years, the Taxpayer Relief Act of 1997 provides that publicly traded corporations must apply this test on a fair market value basis only. We believe that we currently do not qualify as a PFIC because our passive income producing assets are less than 50% of our total assets.
As a PFIC, each U.S. Holder must determine under which of the alternative tax methods it wishes to be taxed. Under one method, a U.S. Holder who elects in a timely manner to treat the Registrant as a Qualified Electing Fund ("QEF"), as defined in the Code, (an "Electing U.S. Holder") will be subject, under Section 1293 of the Code, to current federal income tax for any taxable year in which we qualify as a PFIC on his pro-rata share of our (i) "net capital gain" (the excess of net long-term capital gain over net short-term capital loss), which will be taxed as long-term capital gain to the Electing U.S. Holder and (ii) "ordinary earnings" (the excess of earnings and profits over net capital gain), which will be taxed as ordinary income to the Electing U.S. Holder, in each case, for the U.S. Holder's taxable year in which (or
94
with which) our taxable year ends, regardless of whether such amounts are actually distributed. Such an election, once made shall apply to all subsequent years unless revoked with the consent of the IRS.
A QEF election also allows the Electing U.S. Holder to (i) generally treat any gain realized on the disposition of his common shares (or deemed to be realized on the pledge of his common shares) as capital gain; (ii) treat his share of our net capital gain, if any, as long-term capital gain instead of ordinary income, and (iii) either avoid interest charges resulting from PFIC status altogether (see discussion of interest charge below), or make an annual election, subject to certain limitations, to defer payment of current taxes on his share of our annual realized net capital gain and ordinary earnings subject, however, to an interest charge. If the Electing U.S. Holder is an individual, such an interest charge would be not deductible.
The procedure a U.S. Holder must comply with in making a timely QEF election will depend on whether the year of the election is the first year in the U.S. Holder's holding period in which we are a PFIC. If the U.S. Holder makes a QEF election in such first year, (sometimes referred to as a "Pedigreed QEF Election"), then the U.S. Holder may make the QEF election by simply filing the appropriate documents at the time the U.S. Holder files its tax return for such first year. If, however, we qualified as a PFIC in a prior year, then the U.S. Holder may make an "Unpedigreed QEF Election" by recognizing as an "excess distribution" (i) under the rules of Section 1291 (discussed below), any gain that he would otherwise recognize if the U.S. Holder sold his stock on the qualification date (Deemed Sale Election) or (ii) if we are a controlled foreign corporation ("CFC"), the Holder's pro rata share of the corporation's earnings and profits (Deemed Dividend Election) (But see "Elimination of Overlap Between Subpart F Rules and PFIC Provisions"). The effect of either the deemed sale election or the deemed dividend election is to pay all prior deferred tax, to pay interest on the tax deferral and to be treated thereafter as a Pedigreed QEF as discussed in the prior paragraph. With respect to a situation in which a Pedigreed QEF election is made, if we no longer qualify as a PFIC in a subsequent year, normal Code rules and not the PFIC rules will apply.
If a U.S. Holder has not made a QEF Election at any time (a "Non-electing U.S. Holder"), then special taxation rules under Section 1291 of the Code will apply to (i) gains realized on the disposition (or deemed to be realized by reason of a pledge) of his common shares and (ii) certain "excess distributions", as specially defined, by our company. An "excess distribution" is any current-year distribution in respect of PFIC stock that represents a ratable portion of the total distributions in respect of the stock during the year that exceed 125 percent of the average amount of distributions in respect of the stock during the three preceding years.
A Non-electing U.S. Holder generally would be required to pro-rate all gains realized on the disposition of his common shares and all excess distributions over the entire holding period for the common shares. All gains or excess distributions allocated to prior years of the U.S. Holder (other than years prior to our first taxable year during such U.S. Holder's holding period and beginning after January, 1987 for which it was a PFIC) would be taxed at the highest tax rate for each such prior year applicable to ordinary income. The Non-electing U.S. Holder also would be liable for interest on the deferred tax liability for each such prior year calculated as if such liability had been due with respect to each such prior year. A Non-electing U.S. Holder that is an individual is not allowed a deduction for interest on the deferred tax liability. The portions of gains and distributions that are not characterized as "excess distributions" are subject to tax in the current year under the normal tax rules of the Internal Revenue Code.
If we are a PFIC for any taxable year during which a Non-electing U.S. Holder holds common shares, then we will continue to be treated as a PFIC with respect to such common Shares, even if it is no longer
95
by definition a PFIC. A Non-electing U.S. Holder may terminate this deemed PFIC status by electing to recognize gain (which will be taxed under the rules discussed above for Non-Electing U.S. Holders) as if such common shares had been sold on the last day of the last taxable year for which it was a PFIC.
Under Section 1291(f) of the Code, the Department of the Treasury has issued proposed regulations that would treat as taxable certain transfers of PFIC stock by Non-electing U.S. Holders that are generally not otherwise taxed, such as gifts, exchanges pursuant to corporate reorganizations, and transfers at death. If a U.S. Holder makes a QEF Election that is not a Pedigreed Election (i.e., it is made after the first year during which we are a PFIC and the U.S. Holder holds our shares) (a "Unpedigreed Election"), the QEF rules apply prospectively but do not apply to years prior to the year in which the QEF first becomes effective. U.S. Holders should consult their tax advisors regarding the specific consequences of making a Non-Pedigreed QEF Election.
Certain special, generally adverse, rules will apply with respect to the common shares while we are a PFIC whether or not it is treated as a QEF. For example under Section 1297(b)(6) of the Code (as in effect prior to the Taxpayer Relief Act of 1997), a U.S. Holder who uses PFIC stock as security for a loan (including a margin loan) will, except as may be provided in regulations, be treated as having made a taxable disposition of such stock.
The foregoing discussion is based on currently effective provisions of the Code, existing and proposed regulations thereunder, and current administrative rulings and court decisions, all of which are subject to change. Any such change could affect the validity of this discussion. In addition, the implementation of certain aspects of the PFIC rules requires the issuance of regulations which in many instances have not been promulgated and which may have retroactive effect. There can be no assurance that any of these proposals will be enacted or promulgated, and if so, the form they will take or the effect that they may have on this discussion. Accordingly, and due to the complexity of the PFIC rules, U.S. Holders of the Registrant are strongly urged to consult their own tax advisors concerning the impact of these rules on their investment in our company. For a discussion of the impact of the Taxpayer Relief Act of 1997 on a U.S. Holder of a PFIC, see "Mark-to-Market Election For PFIC Stock Under the Taxpayer Relief Act of 1997" and "Elimination of Overlap Between Subpart F Rules and PFIC Provisions" below.
Mark-to-Market Election for PFIC Stock under the Taxpayer Relief Act of 1997
The Taxpayer Relief Act of 1997 provides that a U.S. Holder of a PFIC may make a mark-to-market election with respect to the stock of the PFIC if such stock is marketable as defined below. This provision is designed to provide a current inclusion provision for persons that are Non-Electing Holders. Under the election, any excess of the fair market value of the PFIC stock at the close of the tax year over the Holder's adjusted basis in the stock is included in the Holder's income. The Holder may deduct any excess of the adjusted basis of the PFIC stock over its fair market value at the close of the tax year. However, deductions are limited to the net mark-to-market gains on the stock that the Holder included in income in prior tax years, or so called "unreversed inclusions." For purposes of the election, PFIC stock is marketable if it is regularly traded on (1) a national securities exchange that is registered with the SEC, (2) the national market system established under Section II A of the Exchange Act, or (3) an exchange or market that the IRS determines has rules sufficient to ensure that the market price represents legitimate and sound fair market value.
A Holder's adjusted basis of PFIC stock is increased by the income recognized under the mark-to-market election and decreased by the deductions allowed under the election. If a U.S. Holder owns PFIC stock indirectly through a foreign entity, the basis adjustments apply to the basis of the PFIC stock in the hands of the foreign entity for the purpose of applying the PFIC rules to the tax treatment of the U.S. owner.
96
Similar basis adjustments are made to the basis of the property through which the U.S. persons hold the PFIC stock.
Income recognized under the mark-to-market election and gain on the sale of PFIC stock with respect to which an election is made is treated as ordinary income. Deductions allowed under the election and loss on the sale of PFIC with respect to which an election is made, to the extent that the amount of loss does not exceed the net mark-to-market gains previously included, are treated as ordinary losses. The U.S. or foreign source of any income or losses is determined as if the amount were a gain or loss from the sale of stock in the PFIC.
If PFIC stock is owned by a CFC (discussed below), the CFC is treated as a U.S. person that may make the mark-to-market election. Amounts includible in the CFC's income under the election are treated as foreign personal holding company income, and deductions are allocable to foreign personal holding company income.
The above provisions apply to tax years of U.S. persons beginning after December 31, 1997, and to tax years of foreign corporations ending with or within such tax years of U.S. persons.
The rules of Code Section 1291 applicable to nonqualified funds as discussed above generally do not apply to a U.S. Holder for tax years for which a mark-to-market election is in effect. If Code Section 1291 is applied and a mark-to-market election was in effect for any prior tax year, the U.S. Holder's holding period for the PFIC stock is treated as beginning immediately after the last tax year of the election. However, if a taxpayer makes a mark-to-market election for PFIC stock that is a nonqualified fund after the beginning of a taxpayer's holding period for such stock, a co-ordination rule applies to ensure that the taxpayer does not avoid the interest charge with respect to amounts attributable to periods before the election.
Controlled Foreign Corporation Status
If more than 50% of the voting power of all classes of stock or the total value of the stock of our company is owned, directly or indirectly, by U.S. Holders, each of whom own after applying rules of attribution 10% or more of the total combined voting power of all classes of stock of our company, we would be treated as a "controlled foreign corporation" or "CFC" under Subpart F of the Code. This classification would bring into effect many complex results including the required inclusion by such 10% U.S. Holders in income of their pro rata shares of "Subpart F income" (as defined by the Code) of our company and our earnings invested in "U.S. property" (as defined by Section 956 of the Code). In addition, under Section 1248 of the Code if we are considered a CFC at any time during the five year period ending with the sale or exchange of its stock, gain from the sale or exchange of common shares of our company by such a 10% U.S. Holder of our common stock at any time during the five year period ending with the sale or exchange is treated as ordinary dividend income to the extent of our earnings and profits attributable to the stock sold or exchanged. Because of the complexity of Subpart F, and because we may never be a CFC, a more detailed review of these rules is beyond the scope of this discussion.
Elimination of Overlap between Subpart F Rules and PFIC Provisions
Under the Taxpayer Relief Act of 1997, a PFIC that is also a CFC will not be treated as a PFIC with respect to certain 10% U.S. Holders. For the exception to apply, (i) the corporation must be a CFC within the meaning of section 957(a) of the Code and (ii) the U.S. Holder must be subject to the current inclusion rules of Subpart F with respect to such corporation (i.e., the U.S. Holder is a "United States Shareholder," see "Controlled Foreign Corporation," above). The exception only applies to that
97
portion of a U.S. Holder's holding period beginning after December 31, 1997. For that portion of a United States Holder before January 1, 1998, the ordinary PFIC and QEF rules continue to apply.
As a result of this new provision, if we were ever to become a CFC, U.S. Holders who are currently taxed on their pro rata shares of Subpart F income of a PFIC which is also a CFC will not be subject to the PFIC provisions with respect to the same stock if they have previously made a Pedigreed QEF Election. The PFIC provisions will however continue to apply to U.S Holders for any periods in which Subpart F does not apply (for example he is no longer a 10% Holder or we are no longer a CFC) and to U.S. Holders that did not make a Pedigreed QEF Election unless the U.S. Holder elects to recognize gain on the PFIC shares held in our company as if those shares had been sold.
ALL PROSPECTIVE INVESTORS ARE ADVISED TO CONSULT THEIR OWN TAX ADVISORS WITH RESPECT TO THE SPECIFIC TAX CONSEQUENCES OF PURCHASING THE COMMON SHARES OF OUR COMPANY.
We have filed with the SEC a registration statement on Form F-1 to register the securities offered in this prospectus. This prospectus, which forms a part of the registration statement, does not contain all of the information included in the registration statement and its exhibits and schedules. References in this prospectus to any contract or other document are not necessarily complete and, if we filed the contract or document as an exhibit to the registration statement, you should refer to the exhibit for more information.
We will be subject to periodic reporting and other informational requirements of the Exchange Act as applicable to foreign private issuers. Accordingly, we will be required to file reports, including annual reports on Form 20-F, and other information with the SEC. As a foreign private issuer, we are exempt from the rules under the Exchange Act that prescribe the furnishing and content of proxy statements, and our officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. We are not currently required under the Exchange Act to publish financial statements as frequently or as promptly as are United States companies subject to the Exchange Act. We will, however, continue to furnish our shareholders with annual reports containing audited financial statements and will issue semi-annual press releases on Form 6-K containing unaudited results of operations as well as such other reports as may from time to time be authorized by our board of directors or as may be otherwise required.
All information filed with the SEC can be inspected and copied at the public reference facilities maintained by the SEC at 100 F Street, N.E., Washington, D.C. 20549. You can request copies of these documents upon payment of a duplicating fee, by writing to the SEC. Please call the SEC at 1-800-SEC-0330 for further information on the operation of the public reference rooms. In addition, the SEC maintains a web site at http://www.sec.gov which contains the Form F-1 and other reports, proxy and information statements, and information regarding issuers that file electronically with the SEC.
ENFORCEABILITY OF CIVIL LIABILITIES
We are organized under the laws of the province of British Columbia, Canada and our executive offices are located outside of the United States in Vancouver, British Columbia. A majority of our directors and officers, as well as the expert named in this prospectus, reside outside the United States. In addition, a
98
substantial portion of their assets and currently all of our assets are located outside of the United States. As a result, you may have difficulty serving legal process within the United States upon us or any of these persons. You may also have difficulty enforcing, both in and outside of the United States, judgments you may obtain in U.S. courts against us or these persons in any action, including actions based upon the civil liability provisions of U.S. Federal or state securities laws. Furthermore, there is substantial doubt as to the enforceability in Canada against us or against any of our directors, officers and the expert named in this prospectus who are not residents of the United States, in original actions or in actions for enforcement of judgments of U.S. courts, of liabilities based solely upon the civil liability provisions of the U.S. federal securities laws.
DISCLOSURE OF COMMISSION POSITION ON INDEMNIFICATION FOR SECURITIES ACT LIABILITIES
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers and controlling persons pursuant to the charter provision, by-law, contract, arrangements, statute or otherwise, we acknowledge that in the opinion of the Securities and Exchange Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable.
Venture Law Corporation is passing upon the validity of the voting common shares being offered hereby for us.
Venture Law Corporation does not have and is not to receive in connection with the offering, a substantial interest, direct or indirect, in the Company or any of its subsidiaries nor was it connected with the Company or any of its subsidiaries as a promoter, managing or principal underwriter, voting trustee, director, officer, or employee.
Our consolidated financial statements for the years ended September 30, 2014 and September 30, 2013 have been included herein in reliance upon the report of MNP LLP, independent registered public accounting firm, appearing elsewhere herein, and upon the authority of such firm as experts in accounting and auditing.
MNP LLP does not have and is not to receive in connection with the offering, a substantial interest, direct or indirect, in the Company or any of its subsidiaries nor was it connected with the Company or any of its subsidiaries as a promoter, managing or principal underwriter, voting trustee, director, officer, or employee.
99
Unaudited Consolidated Financial Statements for the three months ended December 31, 2014
Notice of No Auditor Review of Interim Financial Statements
Interim Consolidated Statements of Financial Position
Interim Consolidated Statements of Loss and Comprehensive Loss
Interim Consolidated Statements of Changes in Shareholders' Equity
Interim Consolidated Statements of Cash Flows
Notes to the Interim Consolidated Financial Statements
Consolidated Financial Statements for the years ended September 30, 2014 and 2013
Independent Auditor's Report of MNP LLP for year ended September 30, 2014
Consolidated Statements of Financial Position
Consolidated Statements of Loss and Comprehensive Loss
Consolidated Statements of Changes in Shareholders' Equity
Consolidated Statements of Cash Flows
Notes to the Consolidated Financial Statements
Consolidated Financial Statements for the years ended September 30, 2013 and 2012
Independent Auditor's Report of Hays & Watson LLP for years ended September 30, 2013 and September 30, 2012
Consolidated Statements of Financial Position
Consolidated Statements of Loss and Comprehensive Loss
Consolidated Statements of Changes in Shareholders' Equity
Consolidated Statements of Cash Flows
Notes to the Consolidated Financial Statements
100

Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
101
NOTICE OF NO AUDITOR REVIEW OF INTERIM FINANCIAL STATEMENTS
Under National Instrument 51 102, Part 4, subsection 4.3(3) (a), if an auditor has not performed a review of the interim financial statements, they must be accompanied by a notice indicating that the financial statements have not been reviewed by an auditor. The Company's independent auditor has not performed a review of these financial statements in accordance with standards established by the Canadian Institute of Chartered Accountants for a review of interim financial statements by an entity's auditor.
102
Abattis Bioceuticals Corp.
Condensed Interim Consolidated Statements of Financial Position
(Expressed in Canadian Dollars)
As at | December 31, 2014 | September 30, 2014 | ||
$ | $ | |||
| ASSETS | ||||
| CURRENT ASSETS | ||||
Cash | 139,032 | 135,171 | ||
Cash held in trust (note 3) | 248,990 | 248,990 | ||
Term deposits (note 4) | 164,692 | 949,692 | ||
Marketable securities (note 5) | 160,518 | 25,475 | ||
Trade and other receivables | 174,823 | 152,693 | ||
Loan receivable (note 7) | 116,290 | - | ||
Prepaid expenses and other deposits | 83,520 | 60,920 | ||
1,087,865 | 1,572,941 | |||
| NON CURRENT ASSETS | ||||
Property and equipment (note 8) | 893,485 | 793,024 | ||
Intangible assets (note 9) | 2,624,314 | 2,641,083 | ||
Other assets | 2,000 | 2,000 | ||
Investments (note 10) | 200,000 | 200,000 | ||
Investment in associates (note 11) | 279,045 | 271,179 | ||
Goodwill | 423,576 | 423,576 | ||
4,422,420 | 4,330,862 | |||
| TOTAL ASSETS | 5,510,285 | 5,903,803 | ||
| LIABILITIES | ||||
| CURRENT LIABILITIES | ||||
Trade and other payables (note 12) | 1,143,885 | 858,181 | ||
Advances payable (note 13) | 18,871 | 18,871 | ||
1,162,756 | 877,052 | |||
| LONG TERM LIABILITIES | ||||
Deferred tax liability | 383,715 | 383,715 | ||
| TOTAL LIABILITIES | 1,546,471 | 1,260,767 | ||
| SHAREHOLDERS' EQUITY | ||||
Share capital (note 14) | 10,239,488 | 10,073,190 | ||
Equity settled share-based payments (note 14) | 1,637,196 | 1,637,196 | ||
Warrants (note 14) | 1,381,736 | 1,441,805 | ||
Accumulated deficit | (9,850,901) | (9,108,966) | ||
| TOTAL SHAREHOLDERS' EQUITY | 3,407,519 | 4,043,225 | ||
| NON-CONTROLLING INTEREST (note 15) | 556,295 | 599,811 | ||
| TOTAL LIABILITIES AND SHAREHOLDERS' EQUITY | 5,510,285 | 5,903,803 |
Nature of operations and going concern (note 1)
The accompanying notes are an integral part of these consolidated financial statements.
These condensed interim consolidated financial statements were authorized for issue by the Board of Directors on March 2, 2015. They are signed on behalf of the Board of Directors by:
| "Douglas Sorocco" | "Bill Fleming" | |
| Director | Director | |
103
Abattis Bioceuticals Corp.
Condensed Interim Consolidated Statements of Loss and Comprehensive Loss
(Expressed in Canadian Dollars)
| For the three months ended | ||||
December 31, 2014 | December 31, 2013 | |||
$ | $ | |||
| REVENUE | ||||
Sales | 11,331 | - | ||
| EXPENSES | ||||
Accounting and audit fees | 16,213 | 18,658 | ||
Advertising | 29,359 | 18,266 | ||
Amortization | 19,801 | 15,621 | ||
Bank service charge | 1,707 | 536 | ||
Depreciation | 21,066 | 3,944 | ||
Domain names | - | 1,000 | ||
Interest | 96 | 3,205 | ||
Legal fees | 103,229 | 52,137 | ||
Management and consulting fees | 237,426 | 98,620 | ||
Office and general administration | 407,905 | 12,873 | ||
Regulatory and transfer agents fees | 12,894 | 2,737 | ||
Research | 29,741 | 26,180 | ||
| (879,437) | (253,777) | |||
| OTHER INCOME (EXPENSES) | ||||
Foreign exchange loss | (93) | (6,804) | ||
Gain (loss) on cancellation and settlement of trade payables | - | 17,500 | ||
Investment income | 80,562 | - | ||
Gain from investment in associates (note 11) | 7,866 | - | ||
Other income | 12,052 | - | ||
Financing costs | (17,732) | - | ||
| 82,655 | 10,696 | |||
| TOTAL COMPREHENSIVE LOSS FOR THE PERIOD | (785,451) | (243,081) | ||
| Comprehensive loss for the period attributable to: | ||||
Common shareholders | (741,935) | (243,081) | ||
Non-controlling interest | (43,516) | - | ||
| (785,451) | (243,081) | |||
Basic and diluted loss per share basic, for loss for the period attributable to common shareholders (options and warrants not included as the impact would be anti-dilutive) | (0.01) | (0.01) | ||
| Weighted average number of common shares outstanding | 65,439,280 | 29,535,181 | ||
The accompanying notes are an integral part of these consolidated financial statements.
104
Abattis Bioceuticals Corp.
Condensed Interim Consolidated Statements of Changes in Shareholders' Equity
(Expressed in Canadian Dollars)
| Share Capital | Reserves | |||||||||||||||
Number of shares
| Amount $ | Equity settled share-based payments $ | Warrants reserve $ | Total $ | Deficit $ | Non-controlling interests $ | Total $ | |||||||||
| Balance at September 30, 2013 | 28,584,094 | 4,291,204 | 316,900 | 606,459 | 923,359 | (4,656,546) | - | 558,017 | ||||||||
| Shares issued as settlement of trade payables | 3,500,000 | 87,500 | - | - | - | - | - | 87,500 | ||||||||
| Net loss for the period | - | - | - | - | - | (243,081) | - | (243,081) | ||||||||
| Balance at December 31, 2013 | 32,084,094 | 4,378,704 | 316,900 | 606,459 | 923,359 | (4,899,627) | - | 402,436 | ||||||||
| Balance at September 30, 2014 | 64,925,686 | 10,073,190 | 1,637,196 | 1,441,805 | 3,079,001 | (9,108,966) | 599,811 | 4,643,036 | ||||||||
| Shares issued for consulting fees | 54,535 | 10,362 | - | - | - | - | - | 10,362 | ||||||||
| Shares issued for directors' fees | 66,668 | 12,667 | - | - | - | - | - | 12,667 | ||||||||
| Shares issued for cash - warrant exercise | 832,000 | 83,200 | - | - | - | - | - | 83,200 | ||||||||
| Reclassification of grant-date fair value on exercise of warrants | - | 60,069 | - | (60,069) | (60,069) | - | - | - | ||||||||
| Net loss for the period | - | - | - | - | - | (741,935) | (43,516) | (785,451) | ||||||||
| Balance at December 31, 2014 | 65,878,889 | 10,239,488 | 1,637,196 | 1,381,736 | 3,018,932 | (9,850,901) | 556,295 | 3,963,814 | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
105
Abattis Bioceuticals Corp.
Condensed Interim Consolidated Statements of Cash Flows
(Unaudited, Expressed in Canadian Dollars)
| For the three months ended | ||||
December 31, 2014 | December 31, 2013 | |||
$ | $ | |||
| Cash flows provided from (used by): | ||||
| OPERATING ACTIVITIES | ||||
| Net loss for the period | (785,451) | (243,081) | ||
Adjustments for items not affecting cash: | ||||
Amortization | 19,801 | 15,621 | ||
Depreciation | 21,066 | 3,944 | ||
Investment loss (gain) | (86,043) | - | ||
Loss on settlement on trade payables | - | (17,500) | ||
Gain from investment in associates | (7,866) | - | ||
Shares issued for consulting fees | 10,362 | - | ||
Shares issued for directors' fees | 12,667 | - | ||
| (815,464) | (241,016) | |||
| Net changes in non-cash working capital items: | ||||
Trade and other receivables | (22,130) | (8,842) | ||
Loan receivable | (116,290) | - | ||
Prepaid expenses and other deposits | (22,600) | 18,044 | ||
Trade and other payables | 285,704 | 225,692 | ||
Interest payable | - | 3,205 | ||
| Net cash flows used in operating activities | (690,780) | (2,917) | ||
| FINANCING ACTIVITIES | ||||
Common shares issued for cash, net of share issue costs | 83,200 | - | ||
Short-term loan received from related party | - | 1,450 | ||
| Net cash flows from financing activities | 83,200 | 1,450 | ||
| INVESTING ACTIVITIES | ||||
Proceeds on redemption of term deposits | 785,000 | - | ||
Purchase of intangible assets | (3,032) | - | ||
Purchase of equipment | (121,527) | - | ||
| Net cash flows from investing activities | 611,441 | - | ||
| Net increase (decrease) in cash | 3,861 | (1,467) | ||
| Cash, beginning of period | 135,171 | 5,327 | ||
| Cash, end of period | 139,032 | 3,860 | ||
| Cash paid during the period for interest | - | - | ||
| Cash paid during the period for income taxes | - | - | ||
Supplementary cash flow information (Note 16)
The accompanying notes are an integral part of these consolidated financial statements.
106
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 1. | NATURE OF OPERATIONS AND GOING CONCERN |
Abattis Bioceuticals Corp. (the "Company" or "Abattis") was incorporated as Sinocan Capital Group Inc. under the Company Act (Canada British Columbia) on June 30, 1997 and listed and began trading on the Canadian National Stock Exchange ("the Exchange") under the symbol "FLU" on December 23, 2010. From February 21, 2014, the Company commenced trading under the new symbol "ATT". The Company's head office is located at Suite 1000 - 355 Burrard Street, Vancouver, British Columbia, V6C 2G8, Canada.
Abattis is a biotechnology company with capabilities, through its wholly owned subsidiaries, of producing, licensing and marketing proprietary ingredients and formulas for use in the BioPharma, Nutraceutical, Cosmetic and Animal Nutrition markets.
These condensed interim consolidated financial statements have been prepared on a going concern basis, which assumes the realization of assets and liquidation of liabilities in the normal course of business in the foreseeable future. These consolidated financial statements do not include any adjustments to the carrying value and classification of recorded asset amounts and classification of liabilities that might be necessary should the Company be unable to continue as a going concern.
The Company's operations to date have been financed by issuing common shares, debt instruments and government assistance. The Company's ability to continue as a going concern is dependent upon profitable commercialization of its technologies and the continuing ability to obtain debt or equity financing to fund ongoing operations and research and development activities. The current cash position on hand and expected cash flows for the next 12 months are not sufficient to fund the Company's ongoing operational needs. Therefore, the Company will need funding through equity or debt financing, joint venture arrangements or a combination thereof. There is no assurance that additional funding or suitable joint venture arrangements will be available on a timely basis or on terms acceptable to the Company. If the Company is unable to obtain sufficient funding in this fashion, the ability of the Company to meet its obligations as they come due and, accordingly, the appropriateness of the use of the going concern assumption will be in significant doubt.
During the three months ended December 31, 2014, the Company has incurred a net loss of $785,451. As at December 31, 2014, the Company had a working capital deficiency of $74,891 (September 30, 2014 - working capital of $695,889) and an accumulated deficit of $9,850,901 (September 30, 2014 - $9,108,966). These factors indicate the presence of a material uncertainty that may cast significant doubt on the ability of the Company to continue as a going concern.
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION |
Statement of compliance to International Financial Reporting Standards
These unaudited condensed consolidated interim financial statements have been prepared in accordance with International Accounting Standard ("IAS") 34, Interim Financial Reporting. Accordingly, they do not include all of the information and disclosures required by International Financial Reporting Standards ("IFRS") for annual consolidated statements.
These unaudited condensed consolidated interim financial statements have been prepared using the same accounting policies and methods of application as the Company's most recent annual audited consolidated financial statements for the year ended September 30, 2014.
Critical accounting estimates
Critical accounting estimates are estimates and assumptions made by Management that may result in a material adjustment to the carrying amounts of assets and/or liabilities within the next financial year and are disclosed in Note 2 of the Company's annual audited consolidated financial statements for the year ended September 30, 2014. There have been no changes to the Company's critical accounting estimates and judgments during the three months ended December 31, 2014.
107
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (continued) |
Presentation and functional currency
The presentation and functional currency of the Company and its subsidiaries is the Canadian dollar. All amounts in these consolidated financial statements are expressed in Canadian dollars, unless otherwise indicated.
New accounting standards
The following standards are not effective until fiscal years beginning on or after January 1, 2015, and, unless otherwise indicated, have no effect on the Company's financial performance:
IFRS 9, Financial Instruments ("IFRS 9") IFRS 9 replaces the guidance in IAS 39 Financial Instruments: Recognition and Measurement, on the classification and measurement of financial assets. The Standard eliminates the existing IAS 39 categories of held to maturity, available-for-sale and loans and receivable. Financial assets will be classified into one of two categories on initial recognition, financial assets measured at amortized cost or financial assets measured at fair value. Gains and losses on re-measurement of financial assets measured at fair value will be recognized in profit or loss, except that for an investment in an equity instrument which is not held-for-trading, IFRS 9 provides, on initial recognition, an irrevocable election to present all fair value changes from the investment in other comprehensive income (OCI). |
IFRS 15, Revenue from Contracts with Customers ("IFRS 15") In May 2014, the IASB issued IFRS 15, Revenue from Contracts with Customers. IFRS 15 is effective for periods beginning on or after January 1, 2017 and is to be applied retrospectively. IFRS 15 clarifies the principles for recognizing revenue from contracts with customers. IFRS 15 will also result in enhanced disclosures about revenue, provide guidance for transactions that were not previously addressed comprehensively (i.e. service revenue and contract modifications) and improve guidance for multiple-element arrangements. The Company intends to adopt IFRS 15 in its financial statements for the annual period beginning October 1, 2018, and may consider earlier adoption. The extent of the impact of adoption of IFRS 15 has not yet been determined. |
IAS 32, Financial Instruments: Presentation IAS 32 is effective for annual periods beginning on or after January 1, 2014, is amended to provide guidance on the offsetting of financial assets and financial liabilities. |
108
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 3. | CASH HELD IN TRUST |
As at December 31, 2014, the Company has $248,990 held in trust (September 30, 2014 - $248,990). This amount was held in a lawyer's trust account at year end. Subsequent to three months ended December 31,2014, this cash has been returned less $78,895 in legal fees.
| 4. | TERM DEPOSITS |
The Company's term deposit matures on March 17, 2015, and bears interest at an annual rate of prime - 1.95%.
| 5. | MARKETABLE SECURITIES |
December 31, 2014 | September 30, 2014 | ||||||
| Quantity | Carrying Value | Quantity | Carrying Value | ||||
| BI Optic Ventures Inc. | 1,234,750 | $ | 160,518 | $ | 254,750 | $ | 25,475 |
| 1,234,750 | $ | 160,518 | $ | 254,750 | $ | 25,475 | |
| 6. | ACCOUNTS RECEIVABLE |
December 31, 2014 | September 30, 2014 | |||
| GST receivable | $ | 129,631 | $ | 26,763 |
| Due from an associate | 24,543 | - | ||
| $ | 174,823 | $ | 26,763 |
| 7. | LOAN RECEIVABLE |
On December 18, 2014, the Company's provided a short-term loan to Terracity Lawrence LLC ("Terracity") for an amount of $116,290 (USD 100,000). This loan matures on February 18, 2015. Under the loan agreement, the Company has an option to convert outstanding principal and interest amount to Terracity's common shares at a conversion price of $0.05 upon Terracity successful closing of its public transaction on the TSX Venture Exchange.
109
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 8. | PROPERTY AND EQUIPMENT |
| Computer equipment | Office equipment | Plant equipment | Leasehold Improvement | Total | ||||||
| Cost | ||||||||||
As at September 30, 2014 | $ | 30,671 | $ | 110,233 | $ | 655,604 | $ | 56,882 | $ | 853,390 |
Additions | 1,641 | 4,959 | 114,926 | - | 121,527 | |||||
| Balance as at December 31, 2014 | $ | 32,312 | $ | 115,192 | $ | 770,530 | $ | 56,882 | $ | 974,917 |
| Depreciation | ||||||||||
As at September 30, 2014 | $ | (2,260) | $ | (10,157) | $ | (42,668) | $ | (5,281) | $ | (60,366) |
Charged for the period | (2,213) | (6,659) | (11,600) | (593) | (21,066) | |||||
| Balance as at December 31, 2014 | $ | (4,473) | $ | (16,816) | $ | (54,268) | $ | (5,874) | $ | (81,432) |
| Net book value | ||||||||||
| As at September 30, 2014 | $ | 28,411 | $ | 100,076 | $ | 612,936 | $ | 51,601 | $ | 793,024 |
| As at December 31, 2014 | $ | 27,839 | $ | 98,376 | $ | 716,262 | $ | 51,008 | $ | 893,485 |
Equipment which is not ready for use as at December 31, 2014 with a cost of $375,000 (September 30, 2014 - $375,000) is not being depreciated.
| 9. | INTANGIBLE ASSETS |
| Patents | Formulae | Licenses | Trademark | Proprietary processes | Total | |||||||
| Cost | ||||||||||||
As at September 30, 2014 | $ | 250,000 | $ | 585,840 | $ | 746,951 | $ | - | $ | 1,245,812 | $ | 2,828,603 |
Additions | - | 2,227 | - | 805 | - | 3,032 | ||||||
| Balance as at December 31, 2014 | $ | 250,000 | $ | 588,067 | $ | 746,951 | $ | 805 | $ | 1,245,812 | $ | 2,831,635 |
| Amortization | ||||||||||||
As at September 30, 2014 | $ | (73,012) | $ | (54,798) | $ | (59,710) | $ | - | $ | - | $ | (187,520) |
Charged for the period | (3,125) | (7,342) | (9,328) | (6) | - | (19,801) | ||||||
| Balance as at December 31, 2014 | $ | (76,137) | $ | (62,140) | $ | (69,038) | $ | (6) | $ | - | $ | (207,321) |
| Net book value | ||||||||||||
| As at September 30, 2014 | $ | 176,988 | $ | 531,042 | $ | 687,241 | $ | - | $ | 1,245,812 | $ | 2,641,083 |
| As at December 31, 2014 | $ | 173,863 | $ | 525,928 | $ | 677,913 | $ | 799 | $ | 1,245,812 | $ | 2,624,314 |
Amortization of intangible assets is included in 'Amortization' on the statement of comprehensive loss.
The Company's intangible assets consist of assets for both finite and indefinite life. The Company amortized the intangible assets based on their expected useful life.
110
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 9. | INTANGIBLE ASSETS (CONTINUED) |
The intangible assets include the following key agreements:
On April 16, 2009 the Company entered into an agreement with PRB and Pacific Bio for the purchase of their interest in patents and intellectual property related to anti-viral products designed to prevent avian influenza in humans and poultry. Accordingly, Pacific Bio relinquished its license for the use of the patents to PRB in return for 4,800,000 common shares in the Company, assigned a value of $480,000 and PRB sold its interest in the patents to the Company for 200,000 common shares at an assigned value of $20,000. During the year ended September 30, 2013, the Company abandoned the patent related to humans and wrote off the amortized value of $189,489. The Company retained the patent related to animals. (Note 25) | ||
On December 27, 2012 the Company entered into a worldwide exclusive agreement with Vertical Designs Ltd. ("VDL") and acquired the license to apparatus engineered using vertical farming technology. The license allows the Company to grow plants using the technology for use as ingredients in pharmaceuticals, nutraceutical, wellness and cosmetics, among other uses. Total consideration for the intangible asset was $500,000 paid by way of issuance of 6,000,000 common shares of the Company. (Note 23) | ||
On August 6, 2014 the Company entered into an agreement with TerraSphere Systems LLC which granted the Company non-exclusive rights to proprietary and patented vertical farming technology for cash consideration of $109,500. The patent related to the technology has a remaining life of 15 years. | ||
On February 27, 2014 the Company purchased organic and hydroponic fertilizer and nutritional proprietary formulas from Green-Grow Garden Products Ltd. in consideration for 300,000 common shares of the Company. | ||
On April 7, 2014, the Company acquired a 51% membership interest in Phytalytics LLC. by making a cash payment of US$20,000 ($22,196), issuing 827,657 common shares with a fair value of $579,360 to the members of Phytalytics LLC and advancing a loan of US$60,000 ($66,588). At the date of acquisition, the Company determined the fair value of the net identified assets of Phytalytics and recognized an intangible asset of $1,245,813 which related to the accumulated research, trade secrets and established standard operating procedures for cannabis analysis laboratory services. Key management personnel remain shareholders in Phytalytics and have active management contracts with the Company. |
111
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 10. | INVESTMENTS |
During the year ended September 30, 2014, the Company purchased 800,000 shares of True Leaf Medicine Corp. at a cost of $200,000. These securities are classified as available for sale. The shares of True Leaf Medicine Corp. do not have a quoted price in an active market and their fair value cannot be reliably measured; accordingly, these shares are measured at their cost in the consolidated financial statements.
| 11. | INVESTMENT IN ASSOCIATES |
The following is a summary of the investment in associates for the three months ended December 31, 2014:
Experion Biotechnologies Inc. | Instant Payment Systems LLC | |||
Carrying value as at September 30, 2014 | $ | - | $ | 271,179 |
Share of profit | - | 7,866 | ||
Carrying value as at December 31, 2014 | $ | - | $ | 279,045 |
On April 10, 2014, the Company through its wholly owned subsidiary, Northern Vine, entered into a share exchange agreement with Experion Biotechnologies Inc. ("Experion"). Experion is incorporated under the laws of British Columbia, Canada and is located in Vancouver, BC. Pursuant to the terms of the agreement, Experion and Northern Vine have exchanged 25% of each parties' issued and outstanding common shares. The Company maintains a 75% ownership in Northern Vine.
April 30, 2014, the Company through its wholly owned subsidiary, Abattis Bioceuticals International Inc., is acquired 34% interest in Instant Payment Systems LLC (IPS), a US entity based in Washington State, in consideration for $100,000 cash payments and 200,000 common shares of the Company with a fair value of $180,000. IPS was incorporated under the laws of Washington, US.
The following table summarizes the financial information of Experion and IPS for the three months ended December 31, 2014:
December 31, 2014 | ||||
Experion Biotechnologies Inc. | Instant Payment Systems LLC | |||
| Current assets | $ | - | $ | 2,135 |
| Current liabilities | (14,298) | (2,000) | ||
| Revenue | - | - | ||
| Net income | - | 23,134 | ||
112
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 12. | TRADE AND OTHER PAYABLES |
December 31, 2014 | September 30, 2014 | |||
| Trade payables | $ | 590,631 | $ | 317,475 |
| Accrued liabilities | 203,330 | 225,675 | ||
| Due to related parties | 343,010 | 309,725 | ||
| Payroll liabilities | 6,914 | 5,306 | ||
| $ | 1,143,885 | $ | 858,181 |
| 13. | ADVANCES PAYABLE |
On January 30, 2013, the Investment Agriculture Foundation provided $18,871 to a subsidiary acquired by the Company on March 1, 2013 to develop high value, high quality fractionation processes for surplus berries. Focus has moved away from this project during the year ended September 30, 2013 and therefore funds advanced by the Investment Agriculture Foundation will be repaid. During the three months ended December 31, 2014, no funds were repaid by the Company (December 31, 2013 - $nil).
| 14. | SHARE CAPITAL |
Authorized share capital
Unlimited number of common shares without par value.
Issued share capital
At December 31, 2014, there were 65,878,889 issued and fully paid common shares (September 30, 2014 - 64,925,686).
During the three months ended December 31, 2014:
The Company issued 66,668 common shares for the directors' fees of $12,667. |
The Company issued 54,535 common shares for the consulting fees of $10,362. |
832,000 warrants were exercised for proceeds of $83,200. A fair value of $60,069 was transferred to share capital from reserves in connection with these exercises. |
During the three months ended December 31, 2014:
The Company issued 3,500,000 common shares with a fair value of $87,500 to settle a trade payable of $105,000. |
113
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 14. | SHARE CAPITAL (CONTINUED) |
Share purchase warrants
The changes in warrants during the three months ended December 31, 2014 are as follows:
| Number outstanding | Weighted average exercise price | ||
| Balance, September 30, 2014 | 7,853,831 | $ | 0.40 |
Exercised | (832,000) | 0.10 | |
| Balance, December 31, 2014 | 7,021,831 | $ | 0.44 |
The following summarizes information about stock options outstanding and exercisable at December 31, 2014:
| Expiry date | Warrants outstanding | Exercise price | Estimated grant date fair value | Weighted average remaining contractual life (in years) | ||
February 14, 2015 | 468,000 | $ | 0.100 | $ | 35,837 | 0.12 |
September 18, 2015 | 5,308,331 | 0.500 | 999,705 | 0.72 | ||
October 7, 2015 | 1,245,500 | 0.250 | 251,681 | 0.77 | ||
| 7,021,831 | $ | $ | 1,287,223 | 0.68 |
114
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 14. | SHARE CAPITAL (CONTINUED) |
Stock options
The Company has a share purchase option plan (dated June 18, 2012) which specifies that a maximum of 10% of the issued and outstanding common shares of the Company may be reserved for issuance pursuant to the exercise of share options. The term of the share options granted are fixed by the board of directors and are not to exceed ten years. The exercise prices of the share options shall not be less than the closing price of the Company's common shares on the day before the day on which the directors grant the share purchase options, less any discount permitted by the Exchange. Vesting of options will be at the discretion of the Board.
No options were granted, exercised, expired or cancelled during the three months ended December 31, 2014 and 2013.
The following summarizes information about stock options outstanding and exercisable at December 31, 2014:
| Expiry date | Options outstanding | Options exercisable | Exercise price | Estimated grant date fair value | Weighted average remaining contractual life (in years) | ||
May 12, 2016 | 660,000 | 660,000 | $ | 0.350 | $ | 189,050 | 1.36 |
December 24, 2017 | 350,000 | 350,000 | 0.100 | 13,057 | 2.98 | ||
December 28, 2017 | 20,000 | 20,000 | 0.100 | 1,635 | 2.99 | ||
January 29, 2019 | 200,000 | 200,000 | 0.115 | 9,051 | 4.08 | ||
February 18, 2019 | 175,000 | 175,000 | 0.170 | 13,151 | 4.14 | ||
March 4, 2019 | 440,000 | 440,000 | 0.450 | 91,782 | 4.18 | ||
July 22, 2019 | 2,438,100 | 2,438,100 | 0.640 | 978,427 | 4.56 | ||
August 8, 2019 | 605,000 | 605,000 | 0.480 | 182,984 | 4.61 | ||
August 13, 2019 | 455,000 | 455,000 | 0.430 | 127,227 | 4.62 | ||
September 16, 2019 | 100,000 | 100,000 | 0.330 | 20,422 | 4.71 | ||
| 5,443,100 | 5,443,100 | $ | $ | 1,626,786 | 4.02 |
115
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 15. | NON-CONTROLLING INTEREST |
The following table summarizes the information relating to the non-controlling interest ("NCI") of the Company's subsidiaries before any inter-company eliminations:
Phytalytics LLC |
| Northern Vine | ||
NCI percentage |
| 49% |
| 25% |
As at December 31, 2014 | ||||
Current assets | $ | 2,162 | $ | 15,564 |
Property and equipment | 83,634 | 94,224 | ||
Intangible assets | 1,245,813 | |||
Goodwill | 423,576 | |||
Current liabilities | (26,279) | (10,737) | ||
Deferred income tax liability | (383,715) | |||
Other non-current liabilities | (185,313) | (213,692) | ||
Net assets | 1,159,878 | (114,641) | ||
Carrying amount for NCI | $ | 568,340 | $ | (28,660) |
For the three months ended December 31, 2014 | ||||
| Revenue | $ | 11,331 | $ | - |
| Loss | (77,673) | (21,822) | ||
| Loss allocated to NCI | $ | (38,060) | $ | (5,456) |
As the December 31, 2014, the carrying value of NCI was $556,295 (September 30, 2014 - $599,811) which was determined as follows:
Phytalytics LLC |
| Northern Vine | ||
| As at September 30, 2014 | $ | 606,400 | $ | (6,589) |
| During the three months ended December 31, 2014 | ||||
Loss allocated to NCI | (38,060) | (5,456) | ||
| As at December 31, 2014 | $ | 568,340 | $ | (12,045) |
| 16. | SUPPLEMENTAL CASH FLOW INFORMATION |
For the three months ended | ||||
| December 31, 2014 | December 31, 2013 | ||
Shares issued for settlement of trade payable | $ | - | $ | 87,500 |
Shares issued for consulting fees |
| 10,363 | - | |
Shares issued for directors' fees |
| 12,666 | - | |
116
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 17. | RELATED PARTY TRANSACTIONS |
Transactions with associates
During the year ended September 30, 2014, the Company provided a short-term loan of $24,543 to IPS. This amount remains outstanding as at December 31, 2014 (September 30, 2014 - $24,543).
Key management personnel compensation
| For the three months ended December 31, 2014 | |||||||
| Name | Position | Management and consulting fees | Director's fees | Total | |||
| Mike Withrow (i) | CEO | $ | 43,749 | $ | - | $ | 43,749 |
| Rene David (ii) | CFO | 31,250 | - | 31,250 | |||
| Terence Fealey (iii) | Director | 27,683 | - | 27,683 | |||
| Brazos Minshew (iv) | Director | - | 26,599 | 26,599 | |||
| Dunlap Codding, P.C. (v) | Director | 66,528 | - | 66,528 | |||
| Emanuel "Manny" Montenegrino (vi) | Director | 32,320 | - | 32,320 | |||
| $ | 201,530 | $ | 26,599 | $ | 228,129 | ||
| For the three months ended December 31, 2013 | |||||||
| Name | Position | Management and consulting fees | Director's fees | Total | |||
| Mike Withrow (i) | CEO | $ | 37,500 | $ | - | $ | 37,500 |
| Rene David (ii) | CFO | 31,250 | 31,250 | ||||
| Terence Fealey (iii) | Director | 25,200 | - | 25,200 | |||
| $ | 93,950 | $ | - | $ | 93,950 | ||
Key management personnel compensation (continued)
i)
| The Company paid management fees of $43,749 to Chiron Capital Inc., a company controlled by Mr. Withrow during the three months ended December 31, 2014 (December 31, 2013 - $37,500). At December 31, 2014, $9,099 due to Mr. Withrow was included in trade and other payables (September 30, 2014 - $18,270). | |
ii)
| The Company paid management fees of $31,250 to Crimson Opportunities Ltd. , a company controlled by Mr. David during the three months ended December 31, 2014 (December 31, 2013 - $31,250). During the year ended September 30, 2014, the Company leased a facility from Crimson Opportunities Ltd. Until the Company secures a license, the Company will use the facility to manufacture and warehouse its proprietary Biocube systems. |
117
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 17. | RELATED PARTY TRANSACTIONS (CONTINUED) |
Key management personnel compensation (continued)
|
| As at December 31, 2014, the minimum lease payments are as follows: |
| Year | ||
| 2015 | 27,338 | |
| 2016 | 36,450 | |
| 2017 | 36,450 | |
| 2018 | 36,450 | |
| 2019 | 24,300 | |
| $ | 160,988 |
|
| At December 31, 2014, $530 due to Mr. David was included in trade and other payables (September 30, 2014 - $10,809). |
| iii) | The Company paid consulting fees of $27,683 to Mr. Fealey the three months ended December 31, 2014 (December 31, 2013 - $25,200). At December 31, 2014, $273,561 due to Mr. Fealey was included in trade and other payables (September 30, 2014 - $235,418). |
| iv) | During the three months ended December 31, 2014, the Company issued 66,668 common shares to Mr. Minshew for the directors' fees of $12,667 (December 31, 2014 - $nil). At December 31, 2014, $6,333 due to Mr. Minshew was included in trade and other payables (September 30, 2014 - $nil). |
| v) | The Company paid legal fees of $66,528 to Dunlap Codding, P.C., of which of Mr. Sorocco is a one-third partner, during the three months ended December 31, 2014 (December 31, 2014 - $nil). At December 31, 2014, $99,245 (September 30, 2014 - $31,040) due to Dunlap Codding, P.C. was included in trade and other payables. |
| vi) | The Company paid consulting fees of $32,320 to Think Sharp, a company controlled by Emanual Montenegrino, the director of the Company, during the three months ended December 31, 2014 (December 31, 2013 - $nil). During the three months ended December 31, 2014, the Company issued 40,000 common shares with a fair value of $7,600 in lieu of cash payments (December 31, 2013 - nil). At December 31, 2014 and September 30, 2014, $nil and $14,187 due to Think Sharp was included in trade and other payables, respectively. |
Transactions with related parties are measured at the exchange amount of consideration established and agreed to by the related parties.
118
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 18. | COMMITMENTS |
i) | On March 1, 2012, the Company entered into a three year consulting agreement with one of the directors of the Company. Under the agreement, the Company will pay US $8,000 per month to this director for consulting and research and development services. The contract expires on March 1, 2015 and if the contract is terminated at the Company's discretion, the director is entitled to receive three months' fees over and above the thirty day notice period. | |
ii) | On April 20, 2012, the Company entered into a five year exclusive distribution agreement with Hedley Enterprises Ltd. ("Hedley") to purchase, resell and distribute Abattis' line of natural products in Canada. Under the terms of the Agreement Hedley has acquired the exclusive right to sell and distribute Abattis' products to all retail distribution channels, which include health food stores, grocery stores, fitness facilities, and similar retail establishments. | |
iii) | On November 1, 2012, the Company renewed a three year office lease with Toro Holdings Ltd. The Company's minimum annual lease payments are as follows: |
| Year | ||
| 2015 | $ | 27,218 |
| 2016 | 3,063 | |
| $ | 30,281 |
iv) | On December 27, 2012, the Company entered into a license agreement with Vertical Designs Ltd. ("Vertical Designs"), a company controlled by the former director of the Company. Under the agreement, the Company has been granted the exclusive, worldwide rights to a patent license, with the right to grant sublicenses, to use the Bio Pharma technology for growing products at licensed facilities, which products may only be used as ingredients in the pharmaceutical, nutraceutical, cosmetic and wellness markets. The royalty provisions of the license agreement reflect that: (i) the royalty payable on net sales of all products sold by Abattis was 4%; (ii) in consideration for the grant of the Company's right to grant sublicenses, the Company will pay to Vertical Designs Ltd. a sublicense royalty of 15% of any monies or other consideration that the Company receives from any sublicense; and (iii) after two years, the Company will be required to pay to Vertical Designs Ltd. a minimum royalty payment of $25,000 per year and if the combined royalty payments paid from (i) and (ii) above do not equal $25,000 in any given year then the Company will be permitted to top up such amount with a cash payment. Under the terms of the agreement, the patent license will revert to Vertical Designs Ltd. in certain circumstances, including: (i) if the Company terminates the agreement; (ii) if the Company materially breaches or defaults in the performance of the agreement and has not cured such default within 60 days, or in the case of failure to pay any amounts due, then within 30 days, after receiving written notice from Vertical Designs Ltd. specifying the breach; (iii) if the Company discontinues its business of producing ingredients for pharmaceutical, nutraceutical, cosmetic or wellness markets; (iv) if the Company fails to pay the annual $25,000 minimum royalty payment for any year ending after the second anniversary of the agreement; or (v) if the Company becomes insolvent, makes an assignment for the benefit of creditors or has a petition of bankruptcy filed by or against it, which petition is not vacated or otherwise removed within 90 days after the filing thereof. The Company also agreed to pay Vertical Designs $250,000 for the purchase and sale of six complete Vertical Designs operational units. The purchase price will be paid in instalments, dates and amounts are to be determined between the parties, with the first payment due on or before the earlier of five business days following the Company completing an equity and/or debt financing of any |
119
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 18. | COMMITMENTS (CONTINUED) |
| amount or the first business day in the seventh month following the date of the Bill of Sale. See Note 23 for events after the reporting period related to this agreement. | |
v) | On January 6, 2013, the Company entered into a two year consulting agreement with Georges Laraque Management Inc. Under the agreement, the Company will pay consulting fees of $5,000 per month. | |
vi) | On October 1, 2013, the Company entered into a consulting agreement with Crimson Opportunities Ltd., a company controlled by the CFO of the Company for his services as CFO and COO. Under the agreement, the Company will pay annual consulting fees of $125,000 (excluding GST). This agreement is in effect until terminated. | |
vii) | On January 1, 2014, the Company entered into a consulting agreement with the Chiron Capital Corp., a company controlled by the CEO of the Company for his services as CEO. Under the agreement, the Company will pay annual consulting fees of $175,000 (excluding GST). This agreement is in effect until terminated. | |
viii) | On January 1, 2014, the Company entered into a five year consulting agreement with Voelpel Gold Medal Investment Ltd. Under the agreement, the Company will pay monthly consulting fees of $3,000 (excluding GST). | |
ix) | On March 16, 2014, the Company entered into a one year consulting agreement with Think Sharp Inc. Under the agreement, the Company will pay monthly consulting fees of $10,000 and monthly administration fees of $100 (excluding GST) in cash and 6,000 common shares per month. On May 1, 2014, this agreement was amended such that the Company will pay monthly consulting fees of $12,000 and monthly administration fees of $120 (excluding GST) in cash and 10,000 common shares per month. | |
x) | On June 25, 2014, the Company entered into an 18 month consulting agreement with Brazos Minshew for his services as the President of one of the Company's subsidiaries. Pursuant to the agreement, the Company will pay, for consulting services, an aggregate of 200,000 shares of the Company payable in monthly instalments for the period of July 1, 2014 to December 31, 2014. Following this period, the Company will pay the consultant $5,000 per month. As of December 31, 2014, 100,001 shares remain to be issued under this arrangement. | |
xi) | During the year ended September 30, 2014, the Company entered into 34 month office lease. The Company's minimum annual lease payments are as follows: |
| Year | ||
| 2015 | $ | 51,645 |
| 2016 | 71,727 | |
| 2017 | 73,962 | |
| $ | 197,334 |
120
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 18. | COMMITMENTS (CONTINUED) |
xii) | During the year ended September 30, 2014, the Company entered into 13 month facility lease. The Company's minimum annual lease payments are as follows: |
| Year | ||
| 2015 | $ | 11,714 |
| 19. | CONTINGENCIES |
On September 20, 2012, a claim, which is based on a contract dated June 29, 2009 between the Company and the plaintiff, was filed against the Company. The plaintiff and the Company entered into an agreement dated May 16, 2011 to settle a dispute between the two parties over the contract dated June 29, 2009. The Company made an initial payment of $5,000 to the plaintiff, as per the agreement dated May 16, 2011. However, the plaintiff did not transfer the payment to an individual named in the agreement nor did the plaintiff instruct this individual appropriately. As such, the Company refused to make any further payments under this agreement until those events have taken place. The plaintiff claims that the agreement of May 16, 2011 is not binding and is seeking payment of $145,000. The outcome of this claim is not determinable and therefore no amount has been recorded for any potential payments which may have to be made. |
The Company is defending a claim from one of its former consultants for breaching the consulting contract which the plaintiff should entitle for 75,000 options of the Company. Legal advice received supports the Company's belief that the claim is without merit. The outcome of this claim is not determinable and therefore no amounts have been recorded for any potential payments which may have to be made. |
The Company is defending a claim from one of its former consultants for breaching a contract to pay for marketing services for approximately $23,000. The Company has filed a counter claim that the plaintiff failed to provide the requested services. The outcome of the claim is not determinable and therefore no amounts have been recorded for any potential payments which have to be made. |
The Company is defending a claim from one of its former directors for amounts payable to him which he claims were to be settled in common shares. The plaintiff has claimed damages of approximately $300,000. The outcome of this claim is not determinable. |
The Company is defending a claim from one of its former consultants for breaching a settlement agreement dated Feb 17, 2011. The plaintiff is seeking payment of $37,356 plus interest. The outcome of this claim is not determinable. |
121
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 19. | CONTINGENCIES (CONTINUED) |
On December 29, 2014, the Company obtained a obtained a preliminary injunction from the Washington state court in King County against Herbal Analytics, LLC, James Baxter, Kaleb Lund and Lauren Hilty, and Erin Leary, Affinor Growers, LLC, and Nicholas Brusatore. Affinor Growers, LLC is a wholly owned subsidiary of defendant Affinor Growers, Inc. (collectively "Defendants"). According to the conclusions of law, the Defendants shall cease and desist from any and all use of PhytaLabs trade secrets and confidential information and documents; (b) are restrained from copying, transferring, using or disclosing to any other person or entity any documentation taken from PhytaLab; (c) shall retain and preserve all existing documents and files that mention, refer to, or are derived from PhytaLab, PhytaLab and Abattis' customers, or PhytaLab and Abattis' prospective customers; and (d) shall keep a detailed, complete, and accurate accounting of its business operations. |
It is the opinion of management, based in part on advice of legal counsel, that the ultimate resolution of these contingencies, to the extent not previously provided for, will not have a material adverse effect on the financial condition of the Corporation.
| 20. | MANAGEMENT OF CAPITAL |
The Company's objectives when managing capital are to safeguard its ability to continue as a going concern in order to pursue the development of its technologies and to maintain a flexible capital structure, which optimizes the costs of capital at an acceptable risk. The Company considers its capital for this purpose to be its shareholders' equity.
The Company's primary source of capital is through the issuance of equity. The Company manages and adjusts its capital structure when changes in economic conditions occur. To maintain or adjust the capital structure, the Company may seek additional funding. The Company may require additional capital resources to meet its administrative overhead expenses in the long term. The Company believes it will be able to raise capital as required in the long term, but recognizes there will be risks involved that may be beyond its control. There are no external restrictions on the management of capital.
| 21. | FINANCIAL INSTRUMENTS |
a) | Fair value In accordance with IFRS, financial instruments are classified into one of the five following categories: held-for-trading, held-to-maturity investments, loans and receivables, available-for-sale financial assets and other financial liabilities. Investments with quoted prices in active markets are designated as held-for-trading. Investments without quoted prices in active markets designated as available for sale and are carried at cost. Cash and trade and other receivable are classified as loans and receivables. Term deposits are classified as held-to-maturity. Their carrying value approximates fair value due to their limited time to maturity and ability to convert them to cash in the normal course of business. Trade payable and accrued liabilities, interest payable, short-term loan and advanced payable are classified as other financial liabilities. Their carrying values also approximate fair value due to their short term maturities. |
122
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 21. | FINANCIAL INSTRUMENTS (CONTINUED) |
a) | Fair value (continued) IFRS 13 establishes a fair value hierarchy that reflects the significance of inputs used in making fair value measurements as follows: |
Level 1 | quoted prices in active markets for identical assets or liabilities; |
Level 2 | inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly (i.e. as prices) or indirectly (i.e. from derived prices); and |
Level 3 | inputs for the asset or liability that are not based upon observable market data. |
The Company has determined the estimated fair values of its financial instruments based upon appropriate valuation methodologies. At December 31, 2014, cash of $139,032 (September 30, 2014 - $135,171), cash held in trust of $248,990 (September 30, 2014 - $248,990) and marketable securities of $160,518 (September 30, 2014 - $25,475) have been measured and recognized in the balance sheet using Level 1 inputs. Investments of $200,000 (September 30, 2014 - $200,000) have been designated as available for sale and carried at cost on the balance sheet as at December 31, 2014, there was no observable market data available. At December 31, 2014 and September 30, 2014, there were no financial assets or liabilities measured and recognized in the balance sheet at fair value that would be categorized as Level 2 and 3 in the fair value hierarchy above.
The Company's financial assets and financial liabilities are categorized as follows:
December 31, 2014 | September 30, 2014 | |||
Financial Assets | ||||
| Held-for-trading | ||||
Cash | $ | 139,032 | $ | 135,171 |
Marketable securities | 160,518 | 25,475 | ||
| Loans and receivable | ||||
Cash held in trust | 248,990 | 248,990 | ||
Term deposits | 164,692 | 949,692 | ||
Trade and other receivables | 174,823 | 152,693 | ||
Loan receivable | 116,290 | - | ||
| Available-for-sale | ||||
Investments | $ | 200,000 | $ | 200,000 |
Financial Liabilities | ||||
| Other financial liabilities | ||||
Trade and other payables | 1,143,885 | 858,181 | ||
Advances payable | 18,871 | 18,871 |
123
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 21. | FINANCIAL INSTRUMENTS (CONTINUED) |
b) | Financial risk management Credit risk Credit risk is the risk that one party to a financial instrument will cause a financial loss for the other party by failing to discharge an obligation. The Company's cash, cash held in trust, term deposits, trade and other receivables and loan receivable are exposed to credit risk. The Company reduces its credit risk on cash and cash equivalents by placing these instruments with institutions of high credit worthiness. As at December 31, 2014 and September 30, 2014, the Company's exposure is the carrying value of the financial instruments. The Company's maximum exposure to credit risk is the carrying value of its financial assets. Liquidity risk Liquidity risk is the risk that an entity will encounter difficulty in raising funds to meet commitments associated with financial instruments. The Company manages liquidity by maintaining adequate cash balances to meet liabilities as they become due. The Company maintained cash at December 31, 2014 in the amount of $139,032 (September 30, 2014 - $135,171), in order to meet short-term business requirements. At December 31, 2014, the Company had accounts payable and accrued liabilities and advances payable of $1,143,885 and $18,871, respectively (September 30, 2014 - $824,862 and $18,871, respectively). All accounts payable and accrued liabilities and advances payables are current. Market risk The significant market risks to which the Company is exposed are interest rate risk and currency risk. Interest rate risk Interest rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. Included in the loss for the year in the financial statements is interest income on Canadian dollar cash and cash equivalents and term deposits. The Company is not exposed to significant other price risk. Currency risk The Company is exposed to currency risk to the extent that monetary assets and liabilities held by the Company are not denominated in Canadian dollars. The Company has not entered into any foreign currency contracts to mitigate this risk. The Company's cash and cash equivalents and accounts payable and accrued liabilities are partly held in US dollars ("USD"); therefore, USD accounts are subject to fluctuation against the Canadian dollar. |
124
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 21. | FINANCIAL INSTRUMENTS (CONTINUED) |
b) | Financial risk management (continued) Market risk (continued) Currency risk (continued) The Company had the following balances in Canadian and foreign currencies as at December 31, 2014: |
| in CAD | in USD | |||
| Cash | $ | 21,979 | $ | 100,674 |
| Cash held in trust | 248,990 | - | ||
| Term deposit | 164,692 | - | ||
| Marketable securities | 160,518 | - | ||
| Amounts receivable | 174,986 | (140) | ||
| Loan receivable | 116,290 | - | ||
| Accounts payable and accrued liabilities | (720,817) | (363,867) | ||
| Advances payable | (18,871) | - | ||
147,767 | (263,333) | |||
| Rate to convert to $1.00 CAD | 1.000 | 1.1627 | ||
| Equivalent to Canadian dollars | 147,767 | (306,177) |
Based on the above net exposures as at December 31, 2014, and assuming that all other variables remain constant, a 10% appreciation or depreciation of the CAD against the USD by 10% would increase/ decrease profit or loss by $30,618.
| 22. | SEGMENTED INFORMATION |
The Company has one reportable operating segment of producing, licensing and marketing proprietary ingredients and formulas for use in the BioPharma, Nutraceutical, Cosmetic and Animal Nutrition markets. Non-current assets (other than financial instruments) by geographic location are as:
|
| Canada |
| US |
| Total |
As at December 31, 2014 |
|
|
|
|
|
|
Property and equipment | $ | 434,851 | $ | 458,634 | $ | 893,485 |
Intangible assets |
| 1,378,502 |
| 1,245,812 |
| 2,624,314 |
Other assets |
| 2,000 |
| - |
| 2,000 |
| $ | 1,815,353 | $ | 1,704,446 | $ | 3,519,799 |
As at September 30, 2014 |
|
|
|
|
|
|
Property and equipment | $ | 334,390 | $ | 458,634 | $ | 793,024 |
Intangible assets |
| 1,395,271 |
| 1,245,812 |
| 2,641,083 |
Other assets |
| 2,000 |
| - |
| 2,000 |
| $ | 1,731,661 | $ | 1,704,446 | $ | 3,436,107 |
125
Abattis Bioceuticals Corp.
Notes to the Condensed Interim Consolidated Financial Statements
For the three months ended December 31, 2014
(Expressed in Canadian Dollars)
| 23. | EVENTS AFTER THE REPORTING DATE |
Subsequent to December 31, 2014:
VDL sent a letter advising they were terminating the license agreement discussed in Note 18(iv) by citing that the Company failed to comply with certain terms and conditions included in the license agreement. The Company believes that the terms in the license agreement have been followed; as a result, the license agreement should be valid. The Company intends to continue to honor the agreement and make any payments or provide any information required under the license. The Company provides for costs related to contingencies when a loss is probable and the amount is reasonably determinable. In the opinion of management, no grounds exist that justify the termination of the license agreement. It is the opinion of management, based in part on advice of legal counsel, that the ultimate resolution of the termination of the license agreement is undeterminable; therefore there has been no provision made with respect to the license in the consolidated financial statements for the for the three months ended December 31, 2014. |
The Company is defending a defamation claim from one of its former directors of the Company. Legal advice received supports the Company's belief that the claim is without merit. The outcome of this claim is not determinable and therefore no amounts have been recorded for any potential payments which may have to be made. |
The Company is defending a claim from one of its former consultants for breaching the terms of an agreement. The plaintiff is claiming an entitlement to 5% of the common shares of Abattis Bioceuticals Corp., damages, punitive damages, and costs. The Company has not yet filed a response to the civil claim. The outcome of this claim is not determinable and therefore no amounts have been recorded for any potential payments which may have to be made. |
The Company was granted a patent which was originally acquired on April 16, 2009 from PRB and Pacific Bio. |
One of the directors of the Company forgave $235,418 in amounts owing to him for consulting services in exchange for cash settlement of $32,000. |
368,000 warrants were exercised for proceeds of $36,800. |
357,642 common shares issued to the Company's directors and officers for the services provided to the Company. |
The Company granted 175,000 stock options to certain of its directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. |
126

Consolidated Financial Statements
For the years ended September 30, 2014 and 2013
(Expressed in Canadian Dollars)
127
Independent Auditors' Report
To the Board of Directors and Shareholders of Abattis Bioceuticals Corp.:
We have audited the accompanying consolidated financial statements of Abattis Bioceuticals Corp. (the "Company"), which comprise the statement of financial position as at September 30, 2014 and the consolidated statements of comprehensive loss, changes in equity and cash flows for the ended September 30, 2014, and the related notes, which comprise significant accounting policies and other explanatory information.
Management's Responsibility for the Consolidated Financial Statements
Management is responsible for the preparation and fair presentation of these consolidated financial statements in accordance with International Financial Reporting Standards as issued by the International Accounting Standards Board, and for such internal control as management determines is necessary to enable the preparation of consolidated financial statements that are free from material misstatement, whether due to fraud or error.
Auditors' Responsibility
Our responsibility is to express an opinion on these consolidated financial statements based on our audit. We conducted our audit in accordance with Canadian generally accepted auditing standards and the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we comply with ethical requirements and plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free from material misstatement.
An audit involves performing procedures to obtain audit evidence, on a test basis, about the amounts and disclosures in the consolidated financial statements. The procedures selected depend on the auditors' judgment, including the assessment of the risks of material misstatement of the consolidated financial statements, whether due to fraud or error. We were not engaged to perform an audit of the Company's internal control over financial reporting. Our audit included consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion. An audit also includes evaluating the appropriateness of accounting principles and policies used and the reasonableness of accounting estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that the audit evidence we have obtained in our audits is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the consolidated financial statements present fairly, in all material respects, the consolidated financial position of Abattis Bioceuticals Corp. as at September 30, 2014 and its consolidated financial performance and its cash flows for the year ended September 30, 2014 in accordance with International Financial Reporting Standards as issued by the International Accounting Standards Board.
Emphasis of Matter
Without qualifying our opinion, we draw attention to Note 1 to these consolidated financial statements, which states that Abattis Bioceuticals Corp. incurred significant losses from operations, negative cash flows from operating activities and has an accumulated deficit. These matters, along with other matters as described in Note 1, indicate the existence of a material uncertainty that raises substantial doubt about the ability of Abattis Bioceuticals Corp. to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Other Matter
The consolidated financial statements of Abattis Bioceuticals Corp. for the year ended September 30, 2013, were audited by another auditor who expressed an unmodified opinion on those statements on January 28, 2014.
Vancouver, BC |
| /s/ MNP LLP |
April 22, 2015 |
| Chartered Accountants |
128
Abattis Bioceuticals Corp.
Consolidated Statements of Financial Position
(Expressed in Canadian Dollars)
| As at | September 30, 2014 | September 30, 2013 | ||
| ASSETS | ||||
| CURRENT ASSETS | ||||
| Cash | $ | 135,171 | $ | 5,327 |
| Cash held in trust (note 4) | 248,990 | - | ||
| Term deposits (note 5) | 949,692 | - | ||
| Marketable securities (note 6) | 25,475 | - | ||
| Other receivables (note 7) | 152,693 | 26,763 | ||
| Prepaid expenses and other deposits | 60,920 | 39,799 | ||
| 1,572,941 | 71,889 | |||
| NON CURRENT ASSETS | ||||
| Property and equipment (note 8) | 793,024 | 481,296 | ||
| Intangible assets (notes 3 and 9) | 2,641,083 | 1,074,161 | ||
| Other assets | 2,000 | 2,000 | ||
| Investments (note 10) | 200,000 | - | ||
| Investment in associates (note 11) | 271,179 | - | ||
| Goodwill (note 3) | 423,576 | - | ||
| 4,330,862 | 1,557,457 | |||
| TOTAL ASSETS | $ | 5,903,803 | $ | 1,629,346 |
| LIABILITIES | ||||
| CURRENT LIABILITIES | ||||
| Trade and other payables (notes 12 and 18) | $ | 858,181 | $ | 727,610 |
| Interest payable (note 13) | - | 34,689 | ||
| Short-term loans (note 13) | - | 290,159 | ||
| Advances payable (note 14) | 18,871 | 18,871 | ||
| 877,052 | 1,071,329 | |||
| LONG TERM LIABILITIES | ||||
| Deferred tax liability (notes 3 and 23) | 383,715 | - | ||
| TOTAL LIABILITIES | 1,260,767 | 1,071,329 | ||
| SHAREHOLDERS' EQUITY | ||||
| Share capital (note 15) | $ | 10,073,190 | $ | 4,291,204 |
| Equity settled share-based payments | 1,637,196 | 316,900 | ||
| Warrants | 1,441,805 | 606,459 | ||
| Accumulated deficit | (9,108,966) | (4,656,546) | ||
| TOTAL SHAREHOLDERS' EQUITY | 4,043,225 | 558,017 | ||
| NON-CONTROLLING INTEREST (note 16) | 599,811 | - | ||
| TOTAL LIABILITIES AND SHAREHOLDERS' EQUITY | $ | 5,903,803 | $ | 1,629,346 |
Nature of operations and going concern (note 1)
The accompanying notes are an integral part of these consolidated financial statements.
These consolidated financial statements were authorized for issue by the Board of Directors on April 22, 2015. They are signed on behalf of the Board of Directors by:
| "Douglas Sorocco" | "William Fleming" | |
| Director | Director | |
129
Abattis Bioceuticals Corp.
Consolidated Statements of Loss and Comprehensive Loss
(Expressed in Canadian Dollars)
| For the years ended | September 30, 2014 | September 30, 2013 | ||
| REVENUE | ||||
Sales | $ | 7,720 | $ | 17,448 |
| EXPENSES |
| |||
Accounting and audit fees | $ | 116,813 | $ | 64,543 |
Advertising | 309,631 | 70,145 | ||
Amortization | 67,350 | 67,314 | ||
Bank service charge | 5,185 | 1,047 | ||
Depreciation | 37,360 | 19,680 | ||
Domain names | 9,522 | 4,919 | ||
Interest | 9,046 | 12,635 | ||
Legal fees | 722,707 | 167,663 | ||
Management and consulting fees | 804,755 | 325,239 | ||
Office and general administration | 369,627 | 83,906 | ||
Regulatory and transfer agents fees | 77,555 | 25,274 | ||
Research | 129,057 | 123,559 | ||
Share-based compensation (note 15) | 4,701,541 | 85,033 | ||
Sponsorship | 18,410 | 10,000 | ||
| (7,378,559) | (1,060,957) | |||
| OTHER INCOME (EXPENSES) |
| |||
Foreign exchange gain (loss) | (10,261) | (6,489) | ||
Gain (loss) on cancellation and settlement of trade payables (note 11) | (127,039) | 132,013 | ||
Interest income | 9,692 | - | ||
Investment loss (note 6) | (13,033) | - | ||
Loss from investment in associates (note 11) | (18,381) | - | ||
Loss on write-off of abandoned patent application | - | (189,489) | ||
Other income | 6,904 | 4,983 | ||
Financing costs | (128,444) | - | ||
| $ | (280,562) | $ | (58,982) | |
| LOSS BEFORE TAXES | $ | (7,651,401) | $ | (1,102,491) |
| Deferred income tax recovery | 39,861 | - | ||
| TOTAL COMPREHENSIVE LOSS FOR THE YEAR | $ | (7,611,540) | $ | (1,102,491) |
| Comprehensive loss for the period attributable to: | ||||
Common shareholders | (7,569,411) | (1,102,491) | ||
Non-controlling interest | (42,129) | - | ||
| $ | (7,611,540) | $ | (1,102,491) | |
| ||||
Basic and diluted loss per share basic, for loss for the period attributable to common shareholders (options and warrants not included as the impact would be anti-dilutive) | $ | (0.16) | $ | (0.04) |
| Weighted average number of common shares outstanding | 48,803,786 | 26,066,009 | ||
The accompanying notes are an integral part of these consolidated financial statements.
130
Abattis Bioceuticals Corp.
Consolidated Statements of Changes in Shareholders' Equity
(Expressed in Canadian Dollars)
Share capital | Reserves | |||||||||||||||
Number of common shares | Amount | Equity settled share-based payments | Warrants reserve | Total | Accumulated deficit | Non-controlling interest | Total | |||||||||
| Balance at September 30, 2012 | 18,489,928 | $ | 3,350,373 | $ | 233,518 | $ | 593,631 | $ | 827,149 | $ | (3,554,055) | $ | - | $ | 623,467 | |
| Private placement | 2,100,000 | 92,172 | - | 12,828 | 12,828 | - | - | 105,000 | ||||||||
| Share issuance costs | - | (4,400) | - | - | - | - | - | (4,400) | ||||||||
| Shares issued for acquisition of assets | 6,400,000 | 578,000 | - | - | - | - | - | 578,000 | ||||||||
| Shares issued as settlement of trade payables | 1,494,166 | 263,408 | - | - | - | - | - | 263,408 | ||||||||
| Options exercised for cash | 100,000 | 11,651 | (1,651) | - | (1,651) | - | - | 10,000 | ||||||||
| Share-based payments | - | - | 85,033 | - | 85,033 | - | - | 85,033 | ||||||||
| Net loss for the period | - | - | - | - | - | (1,102,491) | - | (1,102,491) | ||||||||
| Balance at September 30, 2013 | 28,584,094 | $ | 4,291,204 | $ | 316,900 | $ | 606,459 | $ | 923,359 | $ | (4,656,546) | - | $ | 558,017 | ||
| Private placement | 17,333,331 | 1,557,646 | - | 1,442,353 | 1,442,353 | - | - | 2,999,999 | ||||||||
| Share issue costs | - | (42,100) | - | - | - | - | - | (42,100) | ||||||||
| Shares issued for acquisition of assets | 425,000 | 270,700 | - | - | - | - | - | 270,700 | ||||||||
| Shares issued for acquisition of Instant Payment Systems LLC | 200,000 | 180,000 | - | - | - | - | - | 180,000 | ||||||||
| Shares issued for acquisition of Phytalytics LLC | 827,657 | 579,360 | - | - | - | - | - | 579,360 | ||||||||
| Shares issued as prepayment | 243,460 | 111,992 | - | - | - | - | - | 111,992 | ||||||||
| Shares issued as settlement of trade payables | 5,668,440 | 407,376 | - | - | - | - | - | 407,376 | ||||||||
| Shares issued for consulting fees | 246,538 | 153,653 | - | - | - | - | - | 153,653 | ||||||||
| Shares issued for directors' fees | 216,666 | 83,333 | - | - | - | - | - | 83,333 | ||||||||
| Warrants exercised for cash | 8,049,500 | 1,061,625 | - | - | - | - | - | 1,061,625 | ||||||||
| Options exercised for cash | 3,131,000 | 547,140 | - | - | - | - | 547,140 | |||||||||
| Reclassification of grant-date fair value on exercise of warrants | - | 609,210 | - | (609,210) | (609,210) | - | - | - | ||||||||
| Reclassification of grant-date fair value on exercise of stock options | - | 264,254 | (264,254) | - | (264,254) | - | - | - | ||||||||
| Reclassification of grant-date fair value on cancelled options | - | - | (3,116,991) | - | (3,116,991) | 3,116,991 | - | - | ||||||||
| Fair value of modification of warrants | - | (2,203) | - | 2,203 | 2,203 | - | - | - | ||||||||
| Non-controlling interest on acquisition | - | - | - | - | - | - | 641,940 | 641,940 | ||||||||
| Share-based payments | - | - | 4,701,541 | - | 4,701,541 | - | - | 4,701,541 | ||||||||
| Net loss for the year | - | - | - | - | - | (7,569,411) | (42,129) | (7,611,540) | ||||||||
| Balance at September 30, 2014 | 64,925,686 | $ | 10,073,190 | $ | 1,637,196 | $ | 1,441,805 | $ | 3,079,001 | $ | (9,108,966) | $ | 599,811 | $ | 4,643,036 | |
The accompanying notes are an integral part of these consolidated financial statements.
131
Abattis Bioceuticals Corp.
Consolidated Statements of Cash Flows
(Unaudited, Expressed in Canadian Dollars)
| For the years ended | September 30, 2014 | September 30, 2013 | ||
| Cash flows provided from (used by): | ||||
| OPERATING ACTIVITIES | ||||
| Net loss for the year | $ | (7,611,540) | $ | (1,102,491) |
Adjustments for items not affecting cash: |
| |||
| Amortization | 67,350 | 67,314 | ||
| Depreciation | 37,360 | 19,680 | ||
| Loss on settlement on trade payables | 133,075 | 72,987 | ||
| Loss on write-off patent application | - | 189,489 | ||
| Share-based payments | 4,701,541 | 85,033 | ||
| Investment loss | 13,033 | - | ||
| Loss from investment in associates | 18,381 | - | ||
| Shares issued for consulting fees | 153,653 | 67,322 | ||
| Shares issued for directors' fees | 83,333 | - | ||
| Shares issued for financing costs | 111,992 | - | ||
| Deferred income tax recovery | (39,861) | - | ||
| (2,331,683) | (600,666) | |||
Net changes in non-cash working capital items: |
| |||
| Trade and other receivables | (125,930) | (13,298) | ||
| Prepaid expenses | (21,121) | - | ||
| Trade and other payables | 404,872 | 371,630 | ||
| Interest payable | (34,689) | 12,183 | ||
| Net cash flows used in operating activities | (2,108,551) | (230,151) | ||
| FINANCING ACTIVITIES |
| |||
| Common shares issued for cash, net of cash share issue costs | 4,566,664 | 110,600 | ||
| Funding received from Agriculture Investment Foundation | - | 18,870 | ||
| Short-term loan received from related party | - | 102,509 | ||
| Repayment of short-term loan | (290,159) | - | ||
| Net cash flows from financing activities | 4,276,505 | 231,979 | ||
| INVESTING ACTIVITIES |
| |||
| Cash held in trust | (248,990) | - | ||
| Purchase of term deposits | (949,692) | - | ||
| Purchase of investments and marketable securities | (238,508) | - | ||
| Investment in subsidiaries, net of cash assumed on acquisition | (26,778) | - | ||
| Investment in associates | (109,560) | - | ||
| Purchase of intangible assets | (117,760) | - | ||
| Purchase of equipment | (346,822) | - | ||
| Purchase of other assets | - | (2,000) | ||
| Net cash flows used in investing activities | (2,038,110) | (2,000) | ||
| Net increase (decrease) in cash | 129,844 | (172) | ||
| Cash, beginning of year | 5,327 | 5,499 | ||
| Cash, end of year | $ | 135,171 | $ | 5,327 |
| ||||
| Cash paid during the year for interest | - | - | ||
| Cash paid during the year for income taxes | - | - | ||
Supplementary cash flow information (Note 17)
The accompanying notes are an integral part of these consolidated financial statements.
132
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 1. | NATURE OF OPERATIONS AND GOING CONCERN |
Abattis Bioceuticals Corp. (the "Company" or "Abattis") was incorporated as Sinocan Capital Group Inc. under the Company Act (Canada British Columbia) on June 30, 1997 and listed and began trading on the Canadian National Stock Exchange ("the Exchange") under the symbol "FLU" on December 23, 2010. From February 21, 2014, the Company commenced trading under the new symbol "ATT". The Company's head office is located at Suite 1000-355 Burrard Street, Vancouver, British Columbia, V6C 2G8, Canada.
Abattis is a biotechnology company with capabilities, through its wholly owned subsidiaries, of producing, licensing and marketing proprietary ingredients and formulas for use in the BioPharma, Nutraceutical, Cosmetic and Animal Nutrition markets.
These consolidated financial statements have been prepared on a going concern basis, which assumes the realization of assets and liquidation of liabilities in the normal course of business in the foreseeable future. These consolidated financial statements do not include any adjustments to the carrying value and classification of recorded asset amounts and classification of liabilities that might be necessary should the Company be unable to continue as a going concern.
The Company's operations to date have been financed by issuing common shares, debt instruments and government assistance. The Company's ability to continue as a going concern is dependent upon profitable commercialization of its technologies and the continuing ability to obtain debt or equity financing to fund ongoing operations and research and development activities. The current cash position on hand and expected cash flows for the next 12 months are not sufficient to fund the Company's ongoing operational needs. Therefore, the Company will need funding through equity or debt financing, joint venture arrangements or a combination thereof. There is no assurance that additional funding or suitable joint venture arrangements will be available on a timely basis or on terms acceptable to the Company. If the Company is unable to obtain sufficient funding in this fashion, the ability of the Company to meet its obligations as they come due and, accordingly, the appropriateness of the use of the going concern assumption will be in significant doubt.
During the year ended September 30, 2014, the Company has incurred a net loss of $7,611,540 (September 30, 2013 - $1,102,491). As at September 30, 2014, the Company had a working capital of $695,889 (September 30, 2013 - working capital deficiency of $999,440) and an accumulated deficit of $9,108,966 (September 30, 2013 $4,656,546). These factors indicate the presence of a material uncertainty that may cast significant doubt on the ability of the Company to continue as a going concern.
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION |
Statement of compliance
These consolidated financial statements have been prepared in accordance with International Financial Reporting Standards ("IFRS") as issued by the International Accounting Standards Board ("IASB").
Basis of presentation
These consolidated financial statements have been prepared on a historical cost basis except for certain financial assets classified as at fair value through profit or loss or available for sale, which are measured at fair value. In addition, the consolidated financial statements have been prepared using the accrual basis of accounting, except for cash flow information.
Presentation and functional currency
The presentation and functional currency of the Company and its subsidiaries is the Canadian dollar. All amounts in these consolidated financial statements are expressed in Canadian dollars, unless otherwise indicated.
133
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting estimates and judgments
The preparation of these consolidated financial statements requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements, and expenses during the reporting period. The estimates and associated assumptions are based on historical experience and various other factors that are believed to be reasonable under the circumstances. Actual outcomes could differ from these estimates. The consolidated financial statements include estimates which, by their nature, are uncertain. The estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimate is revised if the revision affects only that period or in both the period of revision and future periods if the revision affects both current and future periods.
Significant estimates are estimates and assumptions about the future and other sources of estimation uncertainty that management has made, that could result in a material adjustment to the carrying amounts of assets and liabilities. Significant estimates used in the preparation of these consolidated financial statements include, but are not limited to, the following:
Investment in associates Including in the carrying value of the Company's investment in associates is the Company's share of loss of the associates for the year ended September 30, 2014. The associates have not released full financial statements for the year ended September 30, 2014 and the Company's share of the loss of the associate has been estimated based on available information, including the associates' internal financial records. These estimates may change when full financial statements become available and this may impact the carrying value of the investment in associates. |
Business combinations The company makes estimates related to the values assigned to assets in the purchase price allocation in a business combination. Changes in these assumptions could result in a change in the value of intangible assets, property and equipment, and non-controlling interests. |
Provisions and contingencies The amount recognized as a provision, including legal, contractual, constructive and other exposures or obligations, is the best estimate of the consideration required to settle the related liability, including any related interest charges, taking into account the risks and uncertainties surrounding the obligation. In addition, contingencies will only be resolved when one or more future events occur or fail to occur. Therefore, assessment of contingencies inherently involves the exercise of significant judgment and estimates of the outcome of future events. The Company assesses its liabilities and contingencies based upon the best information available. |
Impairment Assets, including intangible assets, property and equipment, goodwill and investment in associates, are reviewed for impairment whenever events or changes in circumstances indicate that their carrying amounts may exceed their recoverable amounts. As at September 30, 2014, there are no indications that existing intangible assets of the Company are impaired. |
134
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting estimates and judgments (continued)
Inputs used in determining the estimated fair values of options and warrants issued during the year The Company has an equity-settled share-based compensation plan for directors, officers and consultants. Services received, and the corresponding increase in equity, are measured by reference to the fair value of the equity instruments at the date of grant, excluding the impact of any non-market vesting conditions. The fair value of share options are estimated using the Black-Scholes model on the date of grant based on certain assumptions. Those assumptions are described in Note 15 and include, among others, expected volatility, expected life of the options and number of options expected to vest. |
Estimated useful lives of property and equipment and intangible assets The Company makes estimates and utilizes assumptions in determining the useful lives of property and equipment and intangible assets, and the related depreciation and amortization. Uncertainties in these estimates relate to technical obsolescence that may change the utilization of certain assets. |
While management believes the estimates contained within these consolidated financial statements are reasonable, actual results could differ from those estimates and could impact future results of operations and cash flows.
Significant accounting judgments are accounting policies that have been identified as being complex or involving subjective judgments or assessments. Critical accounting judgments used by the Company include, but are not limited to, the following:
Income taxes The Company is subject to income taxes in various jurisdictions and subject to various rates and rules of taxation. Significant judgment is required in determining the provision for income taxes. There are many transactions and calculations undertaken during the ordinary course of business for which the ultimate tax determination is uncertain. The Company recognizes liabilities for anticipated tax audit issues based on the Company's current understanding of the tax law. Where the final tax outcome of these matters is different from the amounts that were initially recorded, such differences will impact the current and deferred tax provisions in the period in which such determination is made. In addition, the Company has not recognized deferred tax assets relating to tax losses carried forward. Future realization of the tax losses depends on the ability of the entity to satisfy certain tests at the time the losses are recouped, including current and future economic conditions and tax law. |
Going concern The Company's ability to execute its strategy by funding future working capital requirements requires judgment. Estimates and assumptions are continually evaluated and are based on historical experience and other factors, such as expectations of future events that are believed to be reasonable under the circumstances. |
135
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting estimates and judgments (continued)
Impairment of non-financial assets Judgment is involved in assessing whether there is any indication that an asset or cash generating unit may be impaired. This assessment is made based on the analysis of, amongst other factors, changes in the market or business environment, events that have transpired that have impacted the asset or cash generating unit, and information from internal reporting. |
Basis of consolidation
These consolidated financial statements include the accounts of the Company and its subsidiaries.
Subsidiaries are controlled by the Company. Control exists when the parent entity has the power to govern the financial and operating policies of an entity so as to obtain benefits from its activities. Subsidiaries are included in the consolidated financial statements from the date control is obtained until the date control is lost.
At September 30, 2014 and September 30, 2013, the Company's subsidiaries are as follows:
Percentage owned* | ||||
| Country of Incorporation | Principal Activity | September 30, 2014 | September 30, 2013 |
| Ijuana Cannabis Inc | Canada | Holds certain licenses | 100% | 100% |
| Abattis Bioceuticals International Inc. | United States | Biotechnology | 100% | Nil |
| BioCell Labs Inc. ("BLI") | Canada | Biotechnology | 100% | 100% |
| North American BioExtracts Inc. ("NAB") | Canada | Biotechnology | 100% | 100% |
| Biocube Green Grow Systems Corp. | Canada | Biotechnology | 100% | Nil |
| True Plant Technologies | Canada | Biotechnology | 100% | Nil |
| Northern Vine Canada Inc. ("Northern Vine") | Canada | Biotechnology | 75% | 100% |
| Phytalytics LLC | United States | Biotechnology | 51% | Nil |
Intercompany transactions and balances between the Company and its subsidiary are eliminated in full on consolidation.
On April 7, 2014, the Company acquired a 51% membership interest in Phytalytics LLC. On April 8, 2014, the Company incorporated another subsidiary National Access Pharmacy Corp. in Canada. On April 10, 2014, the Company through its wholly owned subsidiary, Northern Vine Canada Inc., entered into a share exchange agreement with Experion Biotechnologies Inc. ("Experion"). Pursuant to the terms of the agreement, Experion and Northern Vine have exchanged 25% of each parties' issued and outstanding common shares. Abattis maintains a 75% ownership in Northern Vine. On January 28, 2014, the Company changed the name of its subsidiary Animo Wellness Corporation to Ijuana Cannabis Inc. On September 23, 2014, the Company changed the name of its subsidiary National Access Pharmacy Corp. to True Plant Technologies.
136
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Non-controlling interests
Non-controlling interests in the Company's less than wholly-owned subsidiaries are classified as a separate component of equity. On initial recognition, non-controlling interests are measured at their proportionate share of the acquisition date fair value of identifiable net assets of the related subsidiary acquired by the Company. Subsequent to the acquisition date, adjustments are made to the carrying amount of non-controlling interests for the non-controlling interests' share of changes to the subsidiary's equity. Adjustments to recognize the non-controlling interests' share of changes to the subsidiary's equity are made even if this results in the non-controlling interests having a deficit balance.
Changes in the Company's ownership interest in a subsidiary that do not result in a loss of control are recorded as equity transactions. The carrying amount of non-controlling interests is adjusted to reflect the change in the non-controlling interests' relative interests in the subsidiary and the difference between the adjustment to the carrying amount of non-controlling interests and the Company's share of proceeds received and/or consideration paid is recognized directly in equity and attributed to the shareholders of the Company.
Significant accounting policies
The significant accounting policies used in the preparation of these consolidated financial statements are as follows:
Business combinations
The Company accounts for a transaction as a business combination when the acquisition of an asset or group of assets constitutes a business and when the Company obtains control of the entity being acquired.
Business combinations are accounted for using the acquisition method. In applying the acquisition method, the Company separately records the identifiable assets acquired, the liabilities assumed, any goodwill acquired and any non-controlling interests in the acquired entity.
The Company measures the identifiable assets acquired and the liabilities assumed at their acquisition-date fair values, less any non-controlling interest at fair value. Goodwill is measured as the excess of the fair value of the consideration transferred, less any non-controlling interest in the entity being acquired over the fair value of the net identifiable assets acquired. The consideration transferred in a business combination is measured as the aggregate of the acquisition date fair values of the assets transferred by the acquirer, the liabilities incurred by the acquirer to former owners of the acquired entity and the equity interests issued by the Company.
Acquisition costs in connection with a business combination are expensed as incurred. Those costs include finder's fees, professional fees, consulting fees and general administrative costs.
Investment in associates
An associate is an entity over which the Company has significant influence and which is neither a subsidiary nor a joint venture.
Significant influence is the power to participate in the financial and operating policy decisions of the investee but is not control or joint control over those policies. Significant influence is presumed to exist when the Company holds between 20% and 50% of the voting power of another entity, but can also arise where the Company holds less than 20% if it has the power to be actively involved and influential in policy decision affecting the entity.
137
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Investment in associates (continued)
An investment in associate is accounted for using the equity method. Under the equity method, investments in associates are carried in the statement of financial position at cost adjusted for post-acquisition changes in the Company's share of net assets of the associate, less any impairment losses. Losses in an associate in excess of the Company's interest in that associate are recognized only to the extent that the Company has incurred a legal or constructive obligation to make payments on behalf of the associate.
Unrealized profits or losses on transactions between the Company and an associate are eliminated to the extent of the Company's interest therein.
At the end of each reporting period, the Company assesses whether there is any evidence that an investment in associate is impaired. This assessment is generally made with reference to the status of licence applications, operating results achieved, and an assessment of the likely results to be achieved from future business operations of the associate. When there is evidence that an investment in associate is impaired, the carrying amount of such investment is compared to its recoverable amount. If the recoverable amount of an investment in associate is less than its carrying amount, the carrying amount is reduced to its recoverable amount and an impairment loss, being the excess of carrying amount over the recoverable amount, is recognized in the period of impairment. When an impairment loss reverses in a subsequent period, the carrying amount of the investment in associate is increased to the revised estimate of recoverable amount to the extent that the increased carrying amount does not exceed the carrying amount that would have been determined had an impairment loss not been previously recognized. A reversal of an impairment loss is recognized in net earnings in the period the reversal occurs.
Related party transactions
Parties are considered to be related if one party has the ability, directly or indirectly, to control the other party or exercise significant influence over the other party in making financial and operating decisions. Parties are also considered to be related if they are subject to common control. Related parties may be individuals or corporate entities. A transaction is considered to be a related party transaction when there is a transfer of resources or obligations between related parties.
Foreign currency
Transactions in currencies other than the functional currency are recorded at the exchange rates prevailing on the dates of the transactions. At each statement of financial position reporting date, monetary assets and liabilities that are denominated in foreign currencies are translated at the exchange rates prevailing at the date of the statement of financial position. Non-monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rates at the acquisition dates.
Gains and losses arising from this translation are included in profit or loss for the period.
138
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Financial instruments
Financial assets and financial liabilities are recognized on the statements of financial position when the Company becomes a party to the contractual provisions of the financial instrument. All financial instruments are initially recorded at fair value.
Financial assets
The Company classifies its financial assets at initial recognition as either financial assets at fair value through profit or loss, loans and receivables, held-to-maturity investments or available-for-sale, depending on the purpose for which the asset was acquired. The Company's accounting policy for each of these categories is as follows:
Fair value through profit or loss - This category comprises derivatives or financial assets acquired or incurred principally for the purpose of selling or repurchasing in the near term. Subsequent to initial recognition, they continue to be recorded in the statement of financial position at fair value with changes in fair value recognized in profit or loss.
Loans and receivables - These assets are non-derivative financial assets with fixed or determinable payments that are not quoted in an active market. Subsequent to initial recognition, they are recorded at amortized cost less any provision for impairment. Individually significant receivables are considered for impairment when they are past due or when other objective evidence is received that a specific counterparty will default.
Held to maturity investments - These assets are non-derivative financial assets with fixed or determinable payments and fixed maturities that the Company's management has the positive intention and ability to hold to maturity. Subsequent to initial recognition, these assets are recorded at amortized cost using the effective interest rate method. If there is objective evidence that the investment is impaired, determined by reference to external credit ratings and other relevant indicators, the financial asset is measured at the present value of estimated future cash flows. Any changes to the carrying amount of the investment, including impairment losses, are recognized in profit or loss.
Available for sale - Non-derivative financial assets not included in the above categories are classified as available for sale. Subsequent to initial recognition, they continue to be recorded at fair value with changes in fair value recognized directly in equity. If there is no quoted price in an active market and fair value cannot be readily determined, available for sale investments are carried at cost. Where a decline in the fair value of an available for sale financial asset constitutes objective evidence of impairment, the amount of the loss is removed from equity and recognized in profit or loss.
The Company's financial assets are cash, term deposits, marketable securities, trade and other receivables and investments. The Company classifies its financial assets as follows:
Cash and investments with quoted prices in active markets are classified as held for trading financial assets at fair value through profit or loss. |
Marketable securities are classified as held for trading financial assets at fair value through profit or loss. |
Investments without quoted prices in active markets are classified as available for sale. |
Term deposits and trade and other receivables are classified as loans and receivables. |
139
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Financial instruments (continued)
Financial assets (continued)
Transaction costs associated with financial assets at fair value through profit or loss are expensed as incurred, while transaction costs associated with all other financial assets are included in the initial carrying amount of the asset.
All financial assets except for those at fair value through profit or loss are subject to review for impairment at least at each reporting date. Financial assets are impaired when there is any objective evidence that a financial asset or a group of financial assets is impaired.
Financial liabilities
The Company classifies its financial liabilities as either financial liabilities at fair value through profit or loss or other financial liabilities, depending on the purpose for which the liability was incurred. The Company's accounting policy for each of these categories is as follows:
Fair value through profit or loss: This category comprises derivatives or liabilities acquired or incurred principally for the purpose of selling or repurchasing in the near term. Subsequent to initial recognition, they continue to be recorded in the statement of financial position at fair value with changes in fair value recognized in profit or loss.
Other financial liabilities: Financial liabilities other than those classified as fair value through profit or loss are classified as other financial liabilities. Subsequent to initial recognition, other financial liabilities are measured at amortized cost using the effective interest rate method.
The Company's financial liabilities are trade and other payables, advances payable, interest payable and the short-term loans. The Company classifies these financial liabilities as other financial liabilities.
The Company classifies and discloses fair value measurements based on a three-level hierarchy:
Level 1 - inputs are unadjusted quoted prices in active markets for identical assets or liabilities; |
Level 2 - inputs other than quoted prices in Level 1 that are observable for the asset or liability, either directly or indirectly; and |
Level 3 - inputs for the asset or liability that are not based on observable market |
Cash
Cash in the statements of financial position comprise cash, bank deposits and short-term investments that are readily converted to known amounts of cash with original maturities of three months or less.
140
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Property and equipment
Property and equipment is carried at cost, less accumulated depreciation and accumulated impairment losses.
The cost of an item of property and equipment consists of the purchase price, any costs directly attributable to bringing the asset to the location and condition necessary for its intended use and an initial estimate of the costs of dismantling and removing the item and restoring the site on which it is located.
Property and equipment is depreciated annually on the following basis:
Computer equipment - 30% declining-balance |
Office equipment - 30% declining-balance |
Plant equipment - 20% declining-balance |
Leasehold improvement - 20 years straight-line |
Depreciation commences when an item of equipment becomes available for use.
An item of property and equipment is derecognized upon disposal or when no future economic benefits are expected to arise from the continued use of the asset. Any gain or loss arising on disposal of the asset, determined as the difference between the net disposal proceeds and the carrying amount of the asset, is recognized in profit or loss.
Where an item of property and equipment comprises major components with different useful lives, the components are accounted for separately. Expenditures incurred to replace a component of an item of property and equipment that is accounted for separately, including major inspection and overhaul expenditures, are capitalized.
Intangible assets
Research and development
Expenditure on research activities, undertaken with the prospect of gaining new scientific or technical knowledge and understanding, is recognized in profit or loss as incurred.
Development activities involve a plan or design for the production of new or substantially improved products and processes. Development expenditure is capitalized only if development costs can be measured reliably, the product or process is technically and commercially feasible, future economic benefits are probable, and the Company intends to and has sufficient resources to complete development and has the ability to use or sell the asset. The expenditures capitalized includes the cost of materials, direct labor, overhead costs that are directly attributable to preparing the asset for its intended use, and borrowing costs on qualifying assets for which the commencement date for capitalization is on or after October 1, 2010. Other development expenditure is recognized in profit or loss as incurred.
Capitalized development expenditure is measured at cost less accumulated amortization and accumulated impairment losses.
141
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Intangible assets (continued)
Other intangible assets
Other intangible assets that are acquired by the Company and have finite useful lives are measured at cost less accumulated amortization and accumulated impairment losses.
Intangible assets are amortized annually on a straight-line basis at the following rates:
Patents - 20 years |
Formulae - 20 years |
Licenses - 5 to 20 years |
Impairment of non-current assets
At each reporting date, the carrying amounts of the Company's assets are reviewed to determine whether there is any indication that those assets are impaired. If any such indication exists, the recoverable amount of the asset is estimated in order to determine the extent of the impairment, if any. Where the asset does not generate cash flows that are independent from other assets, the Company estimates the recoverable amount of the cash-generating unit to which the asset belongs.
The recoverable amount of an asset is the higher of fair value less costs to sell and value in use. Fair value is determined as the amount that would be obtained from the sale of the asset in an arm's length transaction between knowledgeable and willing parties. In assessing value in use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset. If the recoverable amount of an asset or cash generating unit is estimated to be less than its carrying amount, the carrying amount of the asset is reduced to its recoverable amount and the impairment loss is recognized in profit or loss.
Where an impairment loss subsequently reverses, the carrying amount of the asset (or cash generating unit) is increased to the revised estimate of its recoverable amount, to the extent the revised carrying amount does not exceed the carrying amount that would have been determined had no impairment loss been recognized for the asset (or cash generating unit) in prior years. A reversal of an impairment loss is recognized in profit or loss.
Share capital
Common shares
Common shares are classified as equity. Transaction costs directly attributable to the issue of common shares and share options are recognized as a deduction from equity, net of any tax effects.
Equity units
Proceeds received on the issuance of units are allocated between the common shares and warrants using the relative fair value method. The fair value of the warrants is determined using the Black Scholes valuation model on the date the units are issued.
142
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Loss per share
The Company presents basic and diluted loss per share data for its common shares, calculated by dividing the loss attributable to common shareholders of the Company by the weighted average number of common shares outstanding during the period. Diluted loss per share is determined by adjusting the loss attributable to common shareholders and the weighted average number of common shares outstanding for the effects of all dilutive potential common shares.
Share-based compensation
The Company's share purchase option plan allows directors, executive officers, employees and consultants to acquire shares of the Company. The fair value of options granted is recognized as a share-based compensation expense with a corresponding increase in equity. An individual is classified as an employee when the individual is an employee for legal or tax purposes (direct employee) or provides services similar to those performed by a direct employee.
The fair value of employee options is measured at the option's grant date, and the fair value of non-employee options is measured at the date when goods or services are received. The fair value of each tranche of options granted which do not vest immediately on grant, is recognized over the period during which each tranche of options vest. The fair value of the options granted is measured using the Black-Scholes option pricing model taking into account the terms and conditions upon which the options were granted. At each financial position reporting date, the amount recognized as an expense is adjusted to reflect the actual number of share options that are expected to vest.
Share-based compensation expense is credited to the equity settled share-based payment reserve. If the options are later exercised, their fair value is transferred from the reserve to share capital.
Provisions
Provisions are recognized where a legal or constructive obligation has been incurred as a result of past events, it is probable that an outflow of resources embodying economic benefit will be required to settle the obligation, and a reliable estimate of the amount of the obligation can be made. If material, provisions are measured at the present value of the expenditures expected to be required to settle the obligation.
Income tax
Income tax expense comprises current and deferred tax. Income tax is recognized in the statement of operations and comprehensive income (loss) except to the extent it relates to items recognized in other comprehensive income or directly in equity.
Current income tax expense is based on the results for the period as adjusted for items that are not taxable or not deductible. Current income tax is calculated using tax rates that were enacted or substantively enacted at the end of the reporting period. Management periodically evaluates positions taken in tax returns with respect to situations in which applicable tax regulation is subject to interpretation. Provisions are established where appropriate on the basis of amounts expected to be paid to the tax authorities.
Deferred taxes are expected to be payable or recoverable between the carrying amounts of assets in the consolidated statement of financial position and their corresponding tax bases used in the computation of taxable profit, and are accounted for using the balance sheet liability method. Deferred tax liabilities are generally recognized for all taxable temporary differences between the carrying amounts of assets and their corresponding tax bases. Deferred tax assets are recognized to the extent that it is probable that taxable profits will be available against which deductible temporary differences can be utilized.
143
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
Significant accounting policies (continued)
Income tax (continued)
Deferred tax liabilities:
| - | are generally recognized for all taxable temporary differences; |
| - | are recognized for all temporary differences arising on investments in subsidiaries except where the reversal of the temporary difference can be controlled and it is probable that the difference will not reverse in the foreseeable future; and |
| - | are not recognized on temporary differences that arise from goodwill which is not deductible for tax purposes. |
Deferred tax assets:
| - | are recognized to the extent it is probable that taxable profits will be available against which the deductible temporary differences can be utilized; and |
| - | are reviewed at the end of the reporting period and reduced to the extent that it is no longer probable that sufficient taxable profits will be available to allow all or part of an asset to be recovered. |
Revenue recognition
Revenue from the sales of goods is recognised when the Company has transferred the significant risks and rewards of ownership to the buyer and it is probable that the Company will receive the previously agreed upon payment. These criteria are considered to be met when the goods are delivered to the buyer. Where the buyer has a right of return, the Company defers recognition of revenue until the right to return has lapsed. However, where high volumes of sales are made to established wholesale customers, revenue is recognised in the period where the goods are delivered less an appropriate provision for returns based on past experience. Provided the amount of revenue can be measured reliably and it is probable that the Group will receive any consideration, revenue for services is recognised in the period in which they are rendered.
Interest from cash and cash equivalents, if applicable, are recorded on an accrual basis when collection is reasonably assured.
New standards and interpretations not yet adopted
During the year ended September 30, 2014, the Company adopted the following standards:
IFRS 7, Financial Instruments: Disclosures ("IFRS 7") IFRS 7 provides disclosure requirements of the information about significance of financial instruments to an entity, and the nature and extent of risks arising from those financial instruments, both in qualitative and quantitative terms. The amendment is effective for annual periods beginning on or after January 1,2013. The Company has evaluated the amendments to IFRS 7 and determined that it did not have material impact on the consolidated financial statements. |
144
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
New standards and interpretations not yet adopted (continued)
IFRS 10, Consolidated Financial Statements ("IFRS 10") IFRS 10 replaces the guidance on control and consolidation in IAS 27 Consolidated and Separate Financial Statements, and SIC 12 Consolidation - Special Purpose Entities. IFRS 10 requires consolidation of an investee only if the investor possesses power over the investee, has exposure to variable returns from its involvement with the investee and has the ability to use its power over the investee to affects its returns. Detailed guidance is provided on applying the definition of control. The accounting requirements for consolidation have remained largely consistent with IAS 27. The Company has evaluated IFRS 10 and determined that it did not result in any change in the consolidation status of any of its subsidiaries and investees. |
IFRS 11, Joint Arrangements ("IFRS 11") IFRS 11 supersedes IAS 31, Interests in Joint Ventures, and requires joint arrangements to be classified either as joint operations or joint ventures depending on the contractual rights and obligations of each investor that jointly controls the arrangement. For joint operations, a corporation recognizes its share of assets, liabilities, revenues and expenses of the joint operation. An investment in a joint venture is accounted for using the equity method as set out in IAS 28, Investments in Associates and Joint Ventures (amended in 2011). The other amendments to IAS 28 did not affect the Company. The Company has no joint arrangements and concluded that the adoption of IFRS 11 did not result in any changes in the accounting for joint arrangements. |
IFRS 12, Disclosure of Interest in Other Entities ("IFRS 12") IFRS 12 provides disclosure requirements for entities that have an interest in a subsidiary, a joint arrangement, an associate or an unconsolidated structured entity. IFRS 12 requires the Company to disclose the significant judgments and assumptions made in determining the nature of the interest in other entities and information about its interest in all other entities. The Company adopted IFRS 12 on October 1, 2013 on a prospective basis. The Company has assessed the disclosure of interest in other entities and concluded that the adoption of IFRS 12 did not result in any changes in disclosures. |
IFRS 13, Fair value measurement ("IFRS 13") IFRS 13, Fair value measurement , provides a single frame work for measuring fair value. The measurement of the fair value of an asset or liability is based on assumptions that market participants would use when pricing the asset or liability under current market conditions, including assumptions about risk. The Company adopted IFRS 13 on a prospective basis. The adoption of IFRS 13 did not require any adjustments to the valuation techniques used by the Company to measure fair value and did not result in any measurement adjustments as at October 1, 2013. The Company has assessed the disclosure of fair value measurement and concluded that the adoption of IFRS 13 did not result in any changes in disclosures. |
145
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 2. | SIGNIFICANT ACCOUNTING POLICIES AND BASIS OF PREPARATION (CONTINUED) |
New standards and interpretations not yet adopted (continued)
The following standards are not effective until fiscal years beginning on or after January 1, 2015, and, unless otherwise indicated, have no effect on the Company's financial performance:
IFRS 9, Financial Instruments ("IFRS 9") IFRS 9 replaces the guidance in IAS 39 Financial Instruments: Recognition and Measurement, on the classification and measurement of financial assets. The Standard eliminates the existing IAS 39 categories of held to maturity, available-for-sale and loans and receivable. Financial assets will be classified into one of two categories on initial recognition, financial assets measured at amortized cost or financial assets measured at fair value. Gains and losses on re-measurement of financial assets measured at fair value will be recognized in profit or loss, except that for an investment in an equity instrument which is not held-for-trading, IFRS 9 provides, on initial recognition, an irrevocable election to present all fair value changes from the investment in other comprehensive income (OCI). |
IFRS 15, Revenue from Contracts with Customers ("IFRS 15") In May 2014, the IASB issued IFRS 15, Revenue from Contracts with Customers. IFRS 15 is effective for periods beginning on or after January 1, 2017 and is to be applied retrospectively. IFRS 15 clarifies the principles for recognizing revenue from contracts with customers. IFRS 15 will also result in enhanced disclosures about revenue, provide guidance for transactions that were not previously addressed comprehensively (i.e. service revenue and contract modifications) and improve guidance for multiple-element arrangements. The Company intends to adopt IFRS 15 in its financial statements for the annual period beginning October 1, 2018, and may consider earlier adoption. The extent of the impact of adoption of IFRS 15 has not yet been determined. |
IAS 32, Financial Instruments: Presentation IAS 32 is effective for annual periods beginning on or after January 1, 2014, is amended to provide guidance on the offsetting of financial assets and financial liabilities. |
| 3. | BUSINESS COMBINATION |
On April 7, 2014, the Company acquired a 51% membership interest in Phytalytics LLC. by making a cash payment of US$20,000 ($22,196), issuing 827,657 common shares with a fair value of $579,360 to the members of Phytalytics LLC and advancing a loan of US$60,000 ($66,588).
The acquisition of Phytalytics LLC will provide a mutually beneficial relationship to Phytalytics LLC and the Company. The new analytics lab testing team in Washington will have the support of the Company's resources to secure a WSLCB license in addition to sourcing the requisite equipment to scale its operations as demand for its services escalate.
The transaction has been accounted for as a business combination by the acquisition method, with the Company identified as the acquirer. The Company's consolidated statements of loss and comprehensive loss include the operating results of Phytalytic LLC from April 7, 2014, the date of acquisition.
146
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 3. | BUSINESS COMBINATION (CONTINUED) |
At the date of acquisition, the Company determined the fair value of the net identified assets of Phytalytics and recognized an intangible assets of $1,245,813 which related to the license application for the cannabis analysis laboratory services. Subsequent to the acquisition, Phytalytics received conditional certification and fully permitted to test and receive payment for testing cannibas under the I-502 legislation. In addition, The Company have passed the state audit of the Company's standards of practice.
The following table summarizes the preliminary allocation of the purchase price to the fair value of the assets acquired and liabilities assumed at the date of acquisition.
| Cash | $ | 62,005 |
| Property and equipment | 2,265 | |
| Intangible assets | 1,245,813 | |
| Defered tax liability | (423,576) | |
| Non-controlling interest | (641,940) | |
| Total net assets acquired | $ | 244,567 |
| Consideration paid: | ||
- Cash (USD 20,000) | $ | 22,196 |
- Shares | 579,360 | |
- Promissory note | 66,587 | |
| $ | 668,143 | |
| Goodwill | $ | 423,576 |
The goodwill recognized at the date of acquisition is attributable to temporary differences between the carrying amounts of assets for financial reporting purposes and their tax values.
Since the date of acquisition, Phytalytics LLC contributed revenue of $7,720 and loss of $72,531 to the Company's consolidated results.
The following table presents unaudited pro forma results of operations for the year ended September 30, 2014 as if the acquisitions had occurred on October 1, 2013. The unaudited pro forma information is not necessarily indicative of the combined results that would have occurred had the acquisitions taken place at the beginning of the periods presented, nor is it necessarily indicative of results that may occur in the future.
| For the years ended | ||||
September 30, 2014 | September 30, 2013 | |||
| Revenue | $ | 7,720 | $ | 17,448 |
| Loss for the year | (7,614,623) | (1,102,491) | ||
| Basic and diluted loss per common share | (0.16) | (0.04) | ||
147
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 3. | BUSINESS COMBINATION (CONTINUED) |
Following is the summary of the financial position of Phytalytics LLC as at September 30, 2014 and the operating result of Phytalyics LLC from the date of acquisition to September 30, 2014:
| As at September 30, 2014 | ||
| Current assets | $ | 37,337 |
| Property and equipment | 83,634 | |
| Intangible assets | 1,245,813 | |
| Goodwill | 423,576 | |
| Current liabilities | (17,813) | |
| Deferred income tax liability | (383,715) | |
| Other non-current liabilities | (151,281) | |
| From the date of acquisition to September 30, 2014 | ||
| Revenue | $ | 7,720 |
| Loss for the period | (72,531) |
| 4. | CASH HELD IN TRUST |
As at September 30, 2014, the Company has $248,990 held in trust (September 30, 2013 - $nil). This amount was held in a lawyer's trust account at year end. Subsequent to year end this cash has been returned less $78,895 in legal fees.
| 5. | TERM DEPOSITS |
The Company's term deposit matures on March 17, 2015, and bears interest at an annual rate of prime - 1.95%.
| 6. | MARKETABLE SECURITIES |
During the year ended September 30, 2014, the Company purchased 254,750 shares of BI Optic Ventures Inc. at a cost of $38,508. These securities are classified as held for trading. As at September 30, 2014, the market value of BI Optic Ventures Inc. is $25,475; as a result, a loss of $13,033 was recognized in statement of loss and comprehensive loss.
| 7. | ACCOUNTS RECEIVABLE |
September 30, 2014 | September 30, 2013 | |||
| GST receivable | $ | 107,040 | $ | 26,763 |
| Other receivable | 21,110 | - | ||
| Due from an associate | 24,543 | - | ||
| $ | 152,693 | $ | 26,763 |
148
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 8. | PROPERTY AND EQUIPMENT |
| Computer equipment | Office equipment | Plant equipment | Leasehold Improvement | Total | ||||||
| COST | ||||||||||
| Balance at September 30, 2012 | $ | 2,047 | $ | 14,792 | $ | 440,000 | $ | 47,463 | $ | 504,302 |
Additions | - | - | - | - | - | |||||
| Balance at September 30, 2013 | 2,047 | 14,792 | 440,000 | 47,463 | 504,302 | |||||
Additions | 28,624 | 95,441 | 215,604 | 9,419 | 349,088 | |||||
| Balance at September 30, 2014 | $ | 30,671 | $ | 110,233 | $ | 655,604 | $ | 56,882 | $ | 853,390 |
| ACCUMULATED DEPRECIATION | ||||||||||
| Balance at September 30, 2012 | $ | (829) | $ | (559) | $ | (1,638) | $ | (300) | $ | (3,326) |
Depreciation | (365) | (4,270) | (12,672) | (2,373) | (19,680) | |||||
| Balance at September 30, 2013 | (1,194) | (4,829) | (14,310) | (2,673) | (23,006) | |||||
Depreciation | (1,066) | (5,328) | (28,358) | (2,608) | (37,360) | |||||
| Balance at September 30, 2014 | $ | (2,260) | $ | (10,157) | $ | (42,668) | $ | (5,281) | $ | (60,366) |
| CARRYING AMOUNT | ||||||||||
| At September 30, 2013 | $ | 853 | $ | 9,963 | $ | 425,690 | $ | 44,790 | $ | 481,296 |
| At September 30, 2014 | $ | 28,411 | $ | 100,076 | $ | 612,936 | $ | 51,601 | $ | 793,024 |
Equipment which is not ready for use as at September 30, 2014 with a cost of $375,000 (September 30, 2013 - $375,000) is not being depreciated.
149
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 9. | INTANGIBLE ASSETS |
| Patents | Formulae | Licenses | Proprietary processes | Total | ||||||
| COST | ||||||||||
| Balance at September 30, 2012 | $ | 500,000 | $ | 250,000 | $ | 116,331 | $ | - | $ | 866,331 |
Additions | - | 78,000 | 500,000 | - | 578,000 | |||||
Write-off application | (250,000) | - | - | - | (250,000) | |||||
| Balance at September 30, 2013 | 250,000 | 328,000 | 616,331 | - | 1,194,331 | |||||
Additions | - | 257,840 | 130,620 | 1,245,812 | 1,634,272 | |||||
| Balance at September 30, 2014 | $ | 250,000 | $ | 585,840 | $ | 746,951 | $ | 1,245,812 | $ | 2,828,603 |
| ACCUMULATED AMORTIZATION | ||||||||||
| Balance at September 30, 2012 | $ | (93,889) | $ | (17,688) | $ | (1,790) | $ | - | $ | (113,367) |
Amortization | (27,134) | (14,487) | (25,693) | - | (67,314) | |||||
Write-off application | 60,511 | - | - | - | 60,511 | |||||
| Balance at September 30, 2013 | (60,512) | (32,175) | (27,483) | - | (120,170) | |||||
Amortization | (12,500) | (22,623) | (32,227) | - | (67,350) | |||||
| Balance at September 30, 2014 | $ | (73,012) | $ | (54,798) | $ | (59,710) | $ | - | $ | (187,520) |
| CARRYING AMOUNT | ||||||||||
| At September 30, 2013 | $ | 189,488 | $ | 295,825 | $ | 588,848 | $ | - | $ | 1,074,161 |
| At September 30, 2014 | $ | 176,988 | $ | 531,042 | $ | 687,241 | $ | 1,245,812 | $ | 2,641,083 |
Amortization of intangible assets is included in 'Amortization' on the statement of comprehensive loss.
The Company's intangible assets consist of assets for both finite and indefinite life. The Company amortized the intangible assets based on their expected useful life.
150
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 9. | INTANGIBLE ASSETS (CONTINUED) |
The intangible assets include the following key agreements:
On April 16, 2009 the Company entered into an agreement with PRB and Pacific Bio for the purchase of their interest in patents and intellectual property related to anti-viral products designed to prevent avian influenza in humans and poultry. Accordingly, Pacific Bio relinquished its license for the use of the patents to PRB in return for 4,800,000 common shares in the Company, assigned a value of $480,000 and PRB sold its interest in the patents to the Company for 200,000 common shares at an assigned value of $20,000. During the year ended September 30, 2013, the Company abandoned the patent related to humans and wrote off the amortized value of $189,489. The Company retained the patent related to animals. (Note 25) |
On December 27, 2012 the Company entered into a worldwide exclusive agreement with Vertical Designs Ltd. ("VDL") and acquired the license to apparatus engineered using vertical farming technology. The license allows the Company to grow plants using the technology for use as ingredients in pharmaceuticals, nutraceutical, wellness and cosmetics, among other uses. Total consideration for the intangible asset was $500,000 paid by way of issuance of 6,000,000 common shares of the Company. (Note 25) |
On August 6, 2014 the Company entered into an agreement with TerraSphere Systems LLC which granted the Company non-exclusive rights to proprietary and patented vertical farming technology for cash consideration of $109,500. The patent related to the technology has a remaining life of 15 years. |
On February 27, 2014 the Company purchased organic and hydroponic fertilizer and nutritional proprietary formulas from Green-Grow Garden Products Ltd. in consideration for 300,000 common shares of the Company. |
On April 7, 2014, the Company acquired a 51% membership interest in Phytalytics LLC. by making a cash payment of US$20,000 ($22,196), issuing 827,657 common shares with a fair value of $579,360 to the members of Phytalytics LLC and advancing a loan of US$60,000 ($66,588). At the date of acquisition, the Company determined the fair value of the net identified assets of Phytalytics and recognized an intangible asset of $1,245,813 which related to the accumulated research, trade secrets and established standard operating procedures for cannabis analysis laboratory services. Key management personnel remain shareholders in Phytalytics and have active management contracts with the Company. |
151
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 10. | INVESTMENTS |
During the year ended September 30, 2014, the Company purchased 800,000 shares of True Leaf Medicine Corp. at a cost of $200,000. These securities are classified as available for sale. The shares of True Leaf Medicine Corp. do not have a quoted price in an active market and their fair value cannot be reliably measured; accordingly, these shares are measured at their cost in the consolidated financial statements.
| 11. | INVESTMENT IN ASSOCIATES |
The following is a summary of the investment in associates for the year ended September 30, 2014:
September 30, 2014 | ||||
Experion Biotechnologies Inc. | Instant Payment Systems LLC | |||
Initial investment | $ | - | $ | 289,560 |
Share of loss | - | (18,381) | ||
Carrying value as at September 30, 2014 | $ | - | $ | 271,179 |
On April 10, 2014, the Company through its wholly owned subsidiary, Northern Vine, entered into a share exchange agreement with Experion Biotechnologies Inc. ("Experion"). Experion is incorporated under the laws of British Columbia, Canada and is located in Vancouver, BC. Pursuant to the terms of the agreement, Experion and Northern Vine have exchanged 25% of each parties' issued and outstanding common shares. The Company maintains a 75% ownership in Northern Vine.
April 30, 2014, the Company through its wholly owned subsidiary, Abattis Bioceuticals International Inc., is acquired 34% interest in Instant Payment Systems LLC (IPS), a US entity based in Washington State, in consideration for $100,000 cash payments and 200,000 common shares of the Company with a fair value of $180,000. IPS was incorporated under the laws of Washington, US.
The following table summarizes the financial information of Experion and IPS for the year ended September 30, 2014:
September 30, 2014 | ||||
Experion Biotechnologies Inc. | Instant Payment Systems LLC | |||
| Current assets | $ | - | $ | 167 |
| Current liabilities | 14,298 | 26,774 | ||
| Revenue | - | - | ||
| Loss | (14,298) | (54,061) | ||
| $ | - | $ | (27,120) | |
152
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 12. | TRADE AND OTHER PAYABLES |
September 30, 2014 | September 30, 2013 | |||
| Trade payables | $ | 317,475 | $ | 238,956 |
| Accrued liabilities | 225,675 | 179,472 | ||
| Due to related parties | 309,725 | 306,363 | ||
| Payroll liabilities | 5,306 | 2,819 | ||
| $ | 858,181 | $ | 727,610 |
During the year ended September 30, 2014, the Company issued 5,668,440 common shares (September 30, 2014 - 1,494,166 common shares) with a fair value of $407,376 (September 30, 2014 - $578,000) to settle the accounts payable of $280,337 (September 30, 2013 - $710,013). As a result, a loss on cancellation and settlement of trade payables of $127,039 (September 30, 2013 - gain of $132,013) was recognized in statement of loss and comprehensive loss.
| 13. | SHORT-TERM LOANS |
Loan from Chief Executive Officer
| Principle | Interest | Total | ||||
| Balance at September 30, 2012 | $ | 157,650 | $ | 22,507 | $ | 180,157 |
| Additions | 132,509 | 12,182 | 144,691 | |||
| Balance at September 30, 2013 | $ | 290,159 | $ | 34,689 | $ | 324,848 |
| Additions | 1,550 | 8,726 | 10,276 | |||
| Repayment | (291,709) | (43,415) | (335,124) | |||
| Balance at September 30, 2014 | $ | - | $ | - | $ | - |
During the year ended September 30, 2012, the Company obtained a $140,000 loan from the Chief Executive Officer ("CEO") of the Company, which bears interest at a rate of 8%. The interest on the loan is payable on a monthly basis, and the principal was repayable six months from the date of advance. The loan is secured by all of the Company's property and assets.
During the year ended September 30, 2014, an additional $1,550 in operating cash was advanced by the CEO of the Company without interest. During the year ended June 30, 2014, $8,726 (September 30, 2014 - $12,182) in interest was accrued as interest payable.
During the year ended September 30, 2014, the Company repaid the loan from Chief Executive Officer in the amount of $335,124 which included accrued interest of $43,415.
| 14. | ADVANCES PAYABLE |
On January 30, 2013, the Investment Agriculture Foundation provided $18,871 to a subsidiary acquired by the Company on March 1, 2013 to develop high value, high quality fractionation processes for surplus berries. Focus has moved away from this project during the year ended September 30, 2013 and therefore funds advanced by the Investment Agriculture Foundation will be repaid. During the year ended September 30, 2014, no funds were repaid by the Company.
153
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL |
Authorized share capital
Unlimited number of common shares without par value.
Issued share capital
At September 30, 2014, there were 64,925,686 issued and fully paid common shares (September 30, 2013 - 28,584,094).
During the year ended September 30, 2014:
On December 6, 2013, the Company issued 3,500,000 common shares with a fair value of $87,500 to settle a trade payable of $105,000. |
On January 16, 2014, the Company issued 625,000 common shares with a fair value of $31,250 to settle a trade payable of $31,250. |
On January 28, 2014, the Company issued 1,500,000 common shares with a fair value of $210,000 to settle a trade payable of $75,000. |
On February 11, 2014, the Company completed a non brokered private placement of 12,000,000 units at a price of $0.05 per unit for gross proceeds of $600,000. Each unit consists of one common share and one half share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.10 per share for a period of one year. $166,813 of the proceeds was allocated to share capital and $433,187 of the proceeds was allocated to warrant reserve. Share issuance costs of $36,300 were paid for this private placement. |
On February 21, 2014, the Company issued 100,000 common shares with a fair value of $17,000 as consideration to acquire the exclusive worldwide distribution rights to Jiangsu Jiahui New Material Co Ltd's. innovative MgO (Magnesium Oxide) products for use in any building or facility designed to cultivate botanicals. 10,000 common shares with a fair value of $1,700 was paid to the finder. |
On March 7, 2014, the Company issued 827,657 common shares with a fair value of $579,360 to consultants of Phytalytics LLC. (Note 3). |
On March 10, 2014, the Company issued 300,000 common shares with a fair value of $240,000 as consideration to acquire the organic and hydroponic fertilizer and nutritional formulas from Green Gro Garden Products Ltd. 15,000 common shares with a fair value of $12,000 was paid to the finder. |
On March 17, 2014, the Company completed a non brokered private placement of 5,333,331 units at a price of $0.45 per unit for gross proceeds of $2,399,999. Each unit consists of one common share and one share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.50 per share for a period of 18 months, subject to forced acceleration in the event the Company's shares close at a price of $0.75 or higher per share on the Canadian National Stock Exchange (or such other stock exchange as the majority of the trading volume of the Company's shares occur) for 10 consecutive trading days. $1,390,834 of the proceeds was allocated to share capital and $1,009,165 of the proceeds was allocated to warrant reserve. Share issuance costs of $5,800 were paid for this private placement. |
154
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Issued share capital (continued)
On April 1, 2014, the Company issued 43,440 common shares to a law firm, of which a director is a one third partner,to settle a trade payable of US$62,554 ($78,626). |
On April 30, 2014, the Company issued 200,000 common shares of the Company with a fair value of $180,000 to acquire 34% interest in Instant Payment Systems LLC (IPS) (Note 11). |
On June 19, 2014, the Company issued 100,000 common shares with fair value of $38,000 to two directors for the consulting services provided. |
During the year ended September 30, 2014, the Company issued 216,538 common shares with a deemed value of $135,773 to TDM Financial for investor relations services provided to the Company. |
During the year ended September 30, 2014, the Company issued 30,000 common shares with a deemed value of $17,880 to Think Sharp for consulting services provided to the Company. |
During the year ended September 30, 2014, the Company issued 116,666 common shares to two directors for the directors' fees of $51,333. |
During the year ended September 30, 2014, 3,131,000 options were exercised for proceeds of $547,140. A fair value of $264,254 was transferred to share capital from reserves in connection with these exercises. | ||
During the year ended September 30, 2014, 8,049,500 warrants were exercised for proceeds of $1,061,625. A fair value of $609,210 was transferred to share capital from reserves in connection with these exercises. | ||
On August 5, 2014, the Company entered into a US$5 million standby equity financing agreement with Kodiak Capital Group, LLC ("Kodiak"), a Newport Beach-based institutional investor. The Company has agreed to file a registration statement with the U.S. Securities and Exchange Commission ("SEC") covering the Company's shares that may be issued to Kodiak under this financing. After the SEC has declared the registration statement related to the transaction effective, the Company has the right at its sole discretion over a period of one year to sell up to US$5 million of common shares to Kodiak. The Company has agreed to pay Kodiak a 5% commitment fee in shares. During the year ended September 30, 2014, 243,460 shares with a fair value of $111,991 were issued as part of a commitment fee and charged to statement of loss (Note 25). |
155
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Issued share capital (continued)
During the year ended September 30, 2013:
On December 12, 2012, the Company completed a non-brokered private placement of 2,100,000 units at a price of $0.05 per unit for gross proceeds of $105,000. Each unit consists of one common share and one share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.13 per share for a period of one year, subject to forced acceleration in the event the Company's shares close at a price of $0.20 or higher per share on the Canadian National Stock Exchange (or such other stock exchange as the majority of the trading volume of the Company's shares occur) for 10 consecutive trading days. $92,172 of the proceeds was allocated to share capital and $12,828 of the proceeds was allocated to warrant reserve. Share issuance costs of $4,400 were paid for this private placement. |
On November 26, 2012, the Company issued 2,500,000 common shares with a fair value of $150,000 and on December 27, 2012, the Company issued 3,500,000 common shares with a fair value of $350,000 as consideration to acquire the exclusive, worldwide rights to a patent license from Vertical Designs Ltd. |
On January 14, 2013, the Company issued 200,000 common shares with a fair value of $36,000 to settle a trade payable of $36,000. |
On January 16, 2013, the Company issued 200,000 common shares to consultants of the Company with aggregate fair value of $25,000 for consulting services. |
On January 16, 2013, the Company issued 213,333 common shares with a fair value of $57,600 to settle a trade payable of $40,000. |
On February 6, 2013, the Company issued 122,500 common shares with a fair value of $25,725 to the CEO of the Company to settle a trade payable of $18,988. |
On March 13, 2013, 100,000 share purchase options were exercised at an exercise price of $0.10 per share purchase option for total proceeds of $10,000. |
On March 18, 2013, the Company issued 133,333 common shares with a fair value of $25,333 to settle a trade payable of $20,434. |
On March 28, 2013, the Company issued 400,000 common shares, with a fair value of $78,000 to acquire a portfolio of natural health product formulae focusing on pain management, immunity and inflammation and cognitive function. |
On April 15, 2013, the Company issued 625,000 common shares with a fair value of $93,750 to settle a trade payable of $50,000. |
156
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Share purchase warrants
The changes in warrants during the years ended September 30, 2014 and 2013 are as follows:
| September 30, 2014 | September 30, 2013 | |||||
| Number outstanding | Weighted average exercise price | Number outstanding | Weighted average exercise price | |||
| Outstanding, beginning of year | 4,570,000 | $ | 0.19 | 3,670,000 | $ | 0.28 |
Issued | 11,333,331 | 0.29 | 2,100,000 | 0.13 | ||
Exercised | (8,049,500) | 0.13 | - | - | ||
Expired | - | - | (1,200,000) | 0.35 | ||
| Outstanding, end of year | 7,853,831 | $ | 0.40 | 4,570,000 | $ | 0.19 |
The following summarizes information about stock options outstanding and exercisable at September 30, 2014:
| Expiry date | Warrants outstanding | Exercise price | Estimated grant date fair value | Weighted average remaining contractual life (in years) | ||
| February 14, 2015 | 1,300,000 | $ | 0.100 | $ | 95,906 | 0.38 |
| September 18, 2015 | 5,308,331 | 0.500 | 999,705 | 0.97 | ||
| October 7, 2015 | 1,245,500 | 0.250 | 251,681 | 1.02 | ||
| 7,853,831 | $ | $ | 1,347,292 | 0.88 |
During the year ended September 30, 2014, the Company extended the expiry date of the warrants which originally expired on December 12, 2013 to December 31, 2014. As a result of the modification, $2,203 was recognized and adjusted to share capital.
157
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Stock options
The Company has a share purchase option plan (dated June 18, 2012) which specifies that a maximum of 10% of the issued and outstanding common shares of the Company may be reserved for issuance pursuant to the exercise of share options. The term of the share options granted are fixed by the board of directors and are not to exceed ten years. The exercise prices of the share options shall not be less than the closing price of the Company's common shares on the day preceding the day on which the directors grant the share purchase options, less any discount permitted by the Exchange. Vesting of options will be at the discretion of the Board.
The changes in options during the years ended September 30, 2014 and 2013 are as follows:
| September 30, 2014 | September 30, 2013 | |||||
| Number outstanding | Weighted average exercise price | Number outstanding | Weighted average exercise price | |||
| Outstanding, beginning of year | 2,575,000 | $ | 0.17 | 890,000 | $ | 0.33 |
Granted | 9,217,200 | 0.87 | 2,290,000 | 0.11 | ||
Exercised | (3,131,000) | 0.17 | (100,000) | 0.10 | ||
Expired | (605,000) | 0.13 | (355,000) | 0.21 | ||
Cancelled | (2,613,100) | 1.99 | (150,000) | 0.10 | ||
| Outstanding, end of year | 5,443,100 | $ | 0.49 | 2,575,000 | $ | 0.17 |
The weighted average share price at the date of exercise was $0.79.
Option issued during the year ended September 30, 2014:
On January 16, 2014, The Company granted 250,000 options with an exercise price of $0.10 to its officers. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On January 29, 2014, The Company granted 700,000 options with an exercise price of $0.115 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On February 28, 2014, The Company granted 1,075,000 options with an exercise price of $0.17 to its officers, directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On February 26, 2014, The Company granted 110,000 options with an exercise price of $0.26 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On February 27, 2014, The Company granted 310,000 options with an exercise price of $0.36 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On March 4, 2014, The Company granted 440,000 options with an exercise price of $0.45 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
158
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Stock options (continued)
On March 5, 2014, The Company granted 121,000 options with an exercise price of $0.64 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On March 10, 2014, The Company granted 330,000 options with an exercise price of $0.80 to its officers. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On March 19, 2014, The Company granted 1,533,100 options with an exercise price of $2.20 to its officers, directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On March 19, 2014, The Company granted 250,000 options with an exercise price of $2.26 to its officers. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On March 31, 2014, The Company granted 500,000 options with an exercise price of $2.00 to its officers, directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On July 22, 2014, The Company granted 2,438,100 options with an exercise price of $0.64 to its officers, directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On August 8, 2014, The Company granted 605,000 options with an exercise price of $0.48 to its officers. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On August 13, 2014, The Company granted 455,000 options with an exercise price of $0.43 to its officers, directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On September 16, 2014, The Company granted 100,000 options with an exercise price of $0.33 to its directors. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
Options issued during the year ended September 30, 2013:
On December 24, 2012, The Company granted 1,615,000 options with an exercise price of $0.10 to its officers, directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On December 28, 2012, The Company granted 350,000 options with an exercise price of $0.10 to its directors and consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
159
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Stock options (continued)
On January 31, 2013, The Company granted 125,000 options with an exercise price of $0.205 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On March 28, 2013, The Company granted 75,000 options with an exercise price of $0.165 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On May 23, 2013, The Company granted 50,000 options with an exercise price of $0.10 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
On May 24, 2013, The Company granted 75,000 options with an exercise price of $0.10 to its consultants. The options are exercisable for a period of five years. All options vested immediately at the grant date. |
The following summarizes information about stock options outstanding and exercisable at September 30, 2014:
| Expiry date | Options outstanding | Options exercisable | Exercise price | Estimated grant date fair value | Weighted average remaining contractual life (in years) | ||
| May 12, 2016 | 660,000 | 660,000 | $ | 0.350 | $ | 189,050 | 1.62 |
| December 24, 2017 | 350,000 | 350,000 | 0.100 | 13,057 | 3.24 | ||
| December 28, 2017 | 20,000 | 20,000 | 0.100 | 1,635 | 3.25 | ||
| January 29, 2019 | 200,000 | 200,000 | 0.115 | 9,051 | 4.33 | ||
| February 18, 2019 | 175,000 | 175,000 | 0.170 | 13,151 | 4.39 | ||
| March 4, 2019 | 440,000 | 440,000 | 0.450 | 91,782 | 4.43 | ||
| July 22, 2019 | 2,438,100 | 2,438,100 | 0.640 | 978,427 | 4.81 | ||
| August 8, 2019 | 605,000 | 605,000 | 0.480 | 182,984 | 4.86 | ||
| August 13, 2019 | 455,000 | 455,000 | 0.430 | 127,227 | 4.87 | ||
| September 16, 2019 | 100,000 | 100,000 | 0.330 | 20,422 | 4.96 | ||
| 5,443,100 | 5,443,100 | $ | 1,626,786 | 4.27 |
160
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 15. | SHARE CAPITAL (continued) |
Share-based payments
The estimated grant date fair value of the options granted during the years ended September 30, 2014 and 2013 was calculated using the Black-Scholes option pricing model with the following assumptions:
| For the year ended | ||||
September 30, 2014 | September 30, 2013 | |||
| Share price | $ | 0.79 | $ | 0.07 |
| Exercise price | 0.87 | 0.11 | ||
| Risk-free interest rate: | 1.29% | 1.14% | ||
| Estimated Volatility: | 93% | 102% | ||
| Expected lives: | 4.07 | 3.10 | ||
| Expected dividend yield | 0% | 0.00% | ||
Option pricing models require the input of highly subjective assumptions, including the expected price volatility. Changes in the subjective input assumptions can materially affect the fair value estimate, and therefore the existing assumptions do not necessarily provide a reliable measure of the future fair value of the Company's share purchase options.
As the Company is a newly listed company which does not have sufficient information on historical volatility, the expected volatility was determined based on the historical volatility of similar entities following a comparable period in their lives.
161
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 16. | NON-CONTROLLING INTEREST |
The following table summarizes the information relating to the non-controlling interest ("NCI") of the Company's subsidiaries before any inter-company eliminations:
Phytalytics LLC |
| Northern Vine | ||
NCI percentage |
| 49% |
| 25% |
As at September 30, 2014 | ||||
Current assets | $ | 37,337 | $ | 34,285 |
Property and equipment | 83,634 | 97,323 | ||
Intangible assets | 1,245,813 | |||
Goodwill | 423,576 | |||
Current liabilities | (17,813) | (10,737) | ||
Deferred income tax liability | (383,715) | |||
Other non-current liabilities | (151,281) | (189,703) | ||
Net assets | 1,237,551 | (68,832) | ||
Carrying amount for NCI | $ | 606,400 | $ | (17,208) |
For the year ended September 30, 2014 | ||||
| Revenue | $ | 7,720 | $ | - |
| Loss | (72,531) | (52,710) | ||
| Loss allocated to NCI | $ | (35,540) | $ | (6,589) |
As the September 30, 2014, the carrying value of NCI was $599,811 which was determined as follows:
Phytalytics LLC |
| Northern Vine | ||
| Initial recognition at the date of acquisition | $ | 641,940 | $ | - |
| During the year ended September 30, 2014 | ||||
Loss allocated to NCI | (35,540) | (6,589) | ||
| As at September 30, 2014 | $ | 606,400 | $ | (6,589) |
162
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 17. | SUPPLEMENTAL CASH FLOW INFORMATION |
For the years ended | September 30, 2014 | September 30, 2013 | ||
| Shares issued for acquisition of assets | $ | 270,700 | $ | 578,000 |
| Shares issued for acquisition of Instant Payment Systems LLC |
| 180,000 | - | |
| Shares issued for acquisition of Phytalytics LLC |
| 579,360 | - | |
| Shares issued for financing costs |
| 111,992 | - | |
| Shares issued for settlement of trade payable |
| 407,376 | 238,408 | |
| Shares issued for consulting fees |
| 153,653 | 55,000 | |
| Shares issued for directors' fees |
| 83,333 | - | |
| Transfer from warrants reserve to share capital on exercise of warrants |
| 609,210 | - | |
| Transfer to warrants reserve fair value of warrants issued on private placement |
| 1,442,353 | 12,828 | |
| Transfer from equity settled share based payments reserve to share capital on exercise of options |
| 264,254 | (1,651) | |
| Transfer from equity settled share based payments reserve to deficit of the cancelled options |
| 3,116,991 | - |
163
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 18. | RELATED PARTY TRANSACTIONS |
Transactions with associates
During the year ended September 30, 2014, the Company provided a short-term loan of $24,543 to IPS (September 30, 2013 - $nil). This amount remains outstanding as at September 30, 2014 (September 30, 2013 - $nil).
Key management personnel compensation
| For the year ended September 30, 2014 | |||||
| Name | Position | Management and consulting fees | Director's fees | Share-based compensation | Total |
($) | ($) | ($) | ($) | ||
| Mike Withrow (i) | CEO | 172,134 | - | 2,226,591 | 2,398,725 |
| Rene David (ii) | CFO | 127,425 | - | 983,105 | 1,110,530 |
| Terence Fealey (iii) | Director | 104,182 | - | - | 104,182 |
| Brazos Minshew (iv) | Director | - | 30,333 | - | 30,333 |
| Guy Dancosse (v) | Director | - | 15,000 | 20,422 | 35,422 |
| Douglas Sorocco (vi) | Director | - | - | 357,306 | 357,306 |
| Robert Hedley (vii) | Director | - | - | 89,326 | 89,326 |
| Dunlap Codding, P.C. (viii) | Director | 75,618 | - | 325,683 | 401,301 |
| Emanuel "Manny" Montenegrino (ix) | Director | 110,860 | 19,000 | - | 129,800 |
| William Fleming (x) | Director | - | 19,000 | - | 19,000 |
| 590,219 | 83,333 | 4,002,433 | 4,675,985 | ||
| For the year ended September 30, 2013 | |||||
| Name | Position | Management and consulting fees | Director's fees | Share-based compensation | Total |
($) | ($) | ($) | ($) | ||
| Mike Withrow (i) | CEO | 142,500 | - | 14,549 | 157,049 |
| Terence Fealey (iii) | Director | 97,816 | - | 2,798 | 100,614 |
| Guy Dancosse (v) | Director | - | - | - | - |
| Douglas Sorocco (vi) | Director | - | - | 2,798 | 2,798 |
| Robert Hedley (vii) | Director | - | - | 2,798 | 2,798 |
| Nick Brusatore | Former Director | - | - | 26,981 | 26,981 |
| Dunlap Codding, P.C. (viii) | Director | 33,150 | - | - | 33,150 |
| 273,466 | - | 49,924 | 323,390 | ||
164
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 18. | RELATED PARTY TRANSACTIONS (CONTINUED) |
Key management personnel compensation (continued)
i)
| The Company paid management fees of $172,134 to Chiron Capital Inc., a company controlled by Mr. Withrow during the year ended September 30, 2014 (September 30, 2013 - $142,500). During the year ended September 30, 2014, 3,230,000 stock options (September 30, 2013 - 390,000), with an estimated fair value of $2,226,591 (September 30, 2013 - $14,549) were granted by the Company to Mr. Withrow and Chiron Capital Inc., a company controlled by Mr. Withrow. During the year ended September 30, 2014, 890,000 options were exercised for cash proceeds of $124,000 (September 30, 2013 - nil) and 1,150,000 options were cancelled (September 30, 2013 - nil). During the year ended September 30, 2014, 1,360,000 warrants were exercised for cash proceeds of $220,000 (September 30, 2013 - nil). During the year ended September 30, 2014, 5,000,000 common shares (September 30, 2013 - 747,500), with an estimated fair value of $297,500 (September 30, 2013 - $119,475), were issued by the Company to Mr. Withrow to settle a trade payable of $180,000 (September 30, 2013 - $68,988). At September 30, 2014, $18,270 due to Mr. Withrow was included in trade and other payables (September 30, 2013 - $142,344). In addition, as at September 30, 2013, operating cash of $260,159 provided by Mr. Withrow and $30,000 in common shares due from the Company were included in short-term loans and accrued interest of $14,468 was included in interest payable. This amount was fully paid during the year ended September 30, 2014. | |
ii)
| The Company paid management fees of $127,425 to Crimson Opportunities Ltd., a company controlled by Mr. David during the year ended September 30, 2014 (September 30, 2013 - $nil). During the year ended September 30, 2014, 2,315,000 stock options (September 30, 2013 - nil), with an estimated fair value of $983,105 (September 30, 2013 - $nil) were granted by the Company to Crimson Opportunities Ltd., a company controlled by Mr. David. During the year ended September 30, 2014, 550,000 options were exercised for cash proceeds of $76,000 (September 30, 2013 - nil) and 580,000 options were cancelled (September 30, 2013 - nil). During the year ended September 30, 2014, 625,000 common shares (September 30, 2013 - nil), with an estimated fair value of $31,250 (September 30, 2013 - $nil) were issued by the Company to Crimson Opportunities Ltd. to settle a trade payable of $31,250 (September 30, 2013 - $nil).
|
165
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 18. | RELATED PARTY TRANSACTIONS (CONTINUED) |
Key management personnel compensation (continued)
During the year ended September 30, 2014, the Company leased a facility from Crimson Opportunities Ltd. Until the Company secures a license, the Company will use the facility to manufacture and warehouse its proprietary Biocube systems. Minimum lease payments are as follows: |
| Year | ||
| 2015 | 36,450 | |
| 2016 | 36,450 | |
| 2017 | 36,450 | |
| 2018 | 36,450 | |
| 2019 | 24,300 | |
| $ | 170,100 |
During the year ended September 30, 2014, the Company recognized $11,340 in related lease expense (September 30, 2013 - $nil). The amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014. At September 30, 2014, $10,809 due to Mr. David was included in trade and other payables (September 30, 2013 - $nil). | ||
| iii) | The Company paid consulting fees of $104,182 to Mr. Fealey during the year ended September 30, 2014 (September 30, 2013 - $97,816). During the year ended September 30, 2014, nil stock options (September 30, 2013 - 75,000), with an estimated fair value of $nil (September 30, 2013 - $2,798) were granted by the Company to Mr. Fealey. At September 30, 2014, $235,418 due to Mr. Fealey was included in trade and other payables (September 30, 2013 - $118,302). |
| iv) | During the year ended September 30, 2014, the Company issued 66,666 common shares to Mr. Minshew for the directors' fees of $30,333 (September 30, 2013 - $nil). During the year ended September 30, 2014, 244,500 warrants were exercised for cash proceeds of $61,125 (September 30, 2013 - nil). |
| v) | During the year ended September 30, 2014, the Company issued 50,000 common shares to Mr. Dancosse for the directors' fees of $15,000 (September 30, 2013 - $nil). During the year ended September 30, 2014, 100,000 stock options (September 30, 2013 - nil), with an estimated fair value of $20,422 (September 30, 2013 - $nil) were granted by the Company to Mr. Dancosse. |
166
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 18. | RELATED PARTY TRANSACTIONS (CONTINUED) |
Key management personnel compensation (continued)
vi) | During the year ended September 30, 2014, 400,000 stock options (September 30, 2013 - 75,000), with an estimated fair value of $357,306 (September 30, 2013 - $2,798) were granted by the Company to Mr. Sorocco. During the year ended September 30, 2014, 200,000 options were cancelled (September 30, 2013 - nil). | |
vii) | During the year ended September 30, 2014, 100,000 stock options (September 30, 2013 - 75,000), with an estimated fair value of $89,326 (September 30, 2013 - $2,798) were granted by the Company to Mr. Hedley. During the year ended September 30, 2014, 75,000 options were exercised for cash proceeds of $7,500 (September 30, 2013 - nil) and 50,000 options were cancelled (September 30, 2013 - nil). | |
viii) | The Company paid legal fees of $75,618 to Dunlap Codding, P.C., of which of Mr. Sorocco is a one-third partner, during the year ended September 30, 2014 (September 30, 2013 - $33,150). During the year ended September 30, 2014, 200,000 stock options (September 30, 2013 - nil), with an estimated fair value of $325,683 (September 30, 2013 - $nil) were granted by the Company to Dunlap Codding, P.C. During the year ended September 30, 2014, 200,000 options were cancelled (September 30, 2013 - nil). During the year ended September 30, 2014, 43,400 common shares (September 30, 2013 - nil), with a deemed value of $78,626 (September 30, 2013 - $nil) were issued by the Company to Dunlap Codding, P.C. to settle a trade payable of $68,591 (September 30, 2013 - $nil). At September 30, 2013, $31,040 (September 30, 2014 - $45,717) due to Dunlap Codding, P.C. was included in trade and other payables. | |
ix) | The Company paid consulting fees of $110,860 and directors' fees of $19,000 to Think Sharp, a company controlled by Emanuel Montenegrino, the director of the Company, during the year ended September 30, 2014 (September 30, 2013 - $nil). During the year ended September 30, 2014, the Company issued 80,000 common shares with a fair value of $36,880 in lieu of cash payments (September 30, 2013 - nil). At September 30, 2013, $14,187 (September 30, 2013 - $nil) due to Think Sharp was included in trade and other payables. | |
| x) | During the year ended September 30, 2014, the Company issued 50,000 common shares to Mr. Fleming for the directors' fees of $19,000 (September 30, 2013 - $nil). |
Transactions with related parties are measured at the exchange amount of consideration established and agreed to by the related parties.
167
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 19. | COMMITMENTS |
i)
| On March 1, 2012, the Company entered into a three year consulting agreement with one of the directors of the Company. Under the agreement, the Company will pay US $8,000 per month to this director for consulting and research and development services. The contract expires on March 1, 2015 and if the contract is terminated at the Company's discretion, the director is entitled to receive three months' fees over and above the thirty day notice period. | |
ii) | On April 20, 2012, the Company entered into a five year exclusive distribution agreement with Hedley Enterprises Ltd. ("Hedley") to purchase, resell and distribute Abattis' line of natural products in Canada. Under the terms of the Agreement Hedley has acquired the exclusive right to sell and distribute Abattis' products to all retail distribution channels, which include health food stores, grocery stores, fitness facilities, and similar retail establishments. | |
iii) | On November 1, 2012, the Company renewed a three year office lease with Toro Holdings Ltd. The Company's minimum annual lease payments are as follows: |
| Year | ||
| 2015 | $ | 36,291 |
| 2016 | 3,063 | |
| $ | 39,354 |
During the year ended September 30, 2014, the Company paid $30,884 lease payments (September 30, 2013 - $30,021). These amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014. | ||
iv) | On December 27, 2012, the Company entered into a license agreement with Vertical Designs Ltd. ("Vertical Designs"), a company controlled by the former director of the Company. Under the agreement, the Company has been granted the exclusive, worldwide rights to a patent license, with the right to grant sublicenses, to use the Bio Pharma technology for growing products at licensed facilities, which products may only be used as ingredients in the pharmaceutical, nutraceutical, cosmetic and wellness markets. The royalty provisions of the license agreement reflect that: (i) the royalty payable on net sales of all products sold by Abattis was 4%; (ii) in consideration for the grant of the Company's right to grant sublicenses, the Company will pay to Vertical Designs Ltd. a sublicense royalty of 15% of any monies or other consideration that the Company receives from any sublicense; and (iii) after two years, the Company will be required to pay to Vertical Designs Ltd. a minimum royalty payment of $25,000 per year and if the combined royalty payments paid from (i) and (ii) above do not equal $25,000 in any given year then the Company will be permitted to top up such amount with a cash payment. Under the terms of the agreement, the patent license will revert to Vertical Designs Ltd. in certain circumstances, including: (i) if the Company terminates the agreement; (ii) if the Company materially breaches or defaults in the performance of the agreement and has not cured such default within 60 days, or in the case of failure to pay any amounts due, then within 30 days, after receiving written notice from Vertical Designs Ltd. specifying the breach; (iii) if the Company discontinues its business of producing ingredients for pharmaceutical, nutraceutical, cosmetic or wellness markets; (iv) if the Company fails to pay the annual $25,000 minimum royalty payment for any year ending after the second anniversary of the agreement; or |
168
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 19. | COMMITMENTS (CONTINUED) |
| (v) if the Company becomes insolvent, makes an assignment for the benefit of creditors or has a petition of bankruptcy filed by or against it, which petition is not vacated or otherwise removed within 90 days after the filing thereof. The Company also agreed to pay Vertical Designs $250,000 for the purchase and sale of six complete Vertical Designs operational units. The purchase price will be paid in instalments, dates and amounts are to be determined between the parties, with the first payment due on or before the earlier of five business days following the Company completing an equity and/or debt financing of any amount or the first business day in the seventh month following the date of the Bill of Sale. See Note 25 for events after the reporting period related to this agreement. | |
v) | On January 6, 2013, the Company entered into a two year consulting agreement with Georges Laraque Management Inc. Under the agreement, the Company will pay consulting fees of $5,000 per month. | |
vi) | On October 1, 2013, the Company entered into a consulting agreement with Crimson Opportunities Ltd., a company controlled by the CFO of the Company for his services as CFO and COO. Under the agreement, the Company will pay annual consulting fees of $125,000 (excluding GST). This agreement is in effect until terminated. | |
vii) | On January 1, 2014, the Company entered into a consulting agreement with the Chiron Capital Corp., a company controlled by the CEO of the Company for his services as CEO. Under the agreement, the Company will pay annual consulting fees of $175,000 (excluding GST). This agreement is in effect until terminated. | |
viii) | On January 1, 2014, the Company entered into a five year consulting agreement with Voelpel Gold Medal Investment Ltd. Under the agreement, the Company will pay monthly consulting fees of $3,000 (excluding GST). | |
ix) | On March 16, 2014, the Company entered into a one year consulting agreement with Think Sharp Inc. Under the agreement, the Company will pay monthly consulting fees of $10,000 and monthly administration fees of $100 (excluding GST) in cash and 6,000 common shares per month. On May 1, 2014, this agreement was amended such that the Company will pay monthly consulting fees of $12,000 and monthly administration fees of $120 (excluding GST) in cash and 10,000 common shares per month. | |
x) | On June 25, 2014, the Company entered into an 18 month consulting agreement with Brazos Minshew for his services as the President of one of the Company's subsidiaries. Pursuant to the agreement, the Company will pay, for consulting services, an aggregate of 200,000 shares of the Company payable in monthly instalments for the period of July 1, 2014 to December 31, 2014. Following this period, the Company will pay the consultant $5,000 per month. As of September 30, 2014, 100,001 shares remain to be issued under this arrangement. |
169
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 19. | COMMITMENTS (CONTINUED) |
xi) | During the year ended September 30, 2014, the Company entered into 34 month office lease. The Company's minimum annual lease payments are as follows: |
| Year | ||
| 2015 | $ | 68,860 |
| 2016 | 71,727 | |
| 2017 | 73,962 | |
| $ | 214,549 |
During the year ended September 30, 2014, the Company paid $5,591 lease payments (September 30, 2013 - $nil). These amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014. |
xii) | During the year ended September 30, 2014, the Company entered into 13 month facility lease. The Company's minimum annual lease payments are as follows: |
| Year | ||
| 2015 | $ | 15,618 |
During the year ended September 30, 2014, the Company paid $10,879 lease payments (September 30, 2013 - $nil). These amount had been charged to statement of loss and comprehensive loss during the year ended September 30, 2014. |
| 20. | CONTINGENCIES |
On September 20, 2012, a claim, which is based on a contract dated June 29, 2009 between the Company and the plaintiff, was filed against the Company. The plaintiff and the Company entered into an agreement dated May 16, 2011 to settle a dispute between the two parties over the contract dated June 29, 2009. The Company made an initial payment of $5,000 to the plaintiff, as per the agreement dated May 16, 2011. However, the plaintiff did not transfer the payment to an individual named in the agreement nor did the plaintiff instruct this individual appropriately. As such, the Company refused to make any further payments under this agreement until those events have taken place. The plaintiff claims that the agreement of May 16, 2011 is not binding and is seeking payment of $145,000. The outcome of this claim is not determinable and therefore no amount has been recorded for any potential payments which may have to be made. |
The Company is defending a claim from one of its former consultants for breaching the consulting contract which the plaintiff should entitle for 75,000 options of the Company. Legal advice received supports the Company's belief that the claim is without merit. The outcome of this claim are not determinable and therefore no amounts have been recorded for any potential payments which may have to be made. | ||
The Company is defending a claim from one of its former consultants for breaching a contract to pay for marketing services for approximately $23,000. The Company has filed a counter claim that the plaintiff failed to provide the requested services. The outcome of the claim is not determinable and therefore no amounts have been recorded for any potential payments which have to be made. |
170
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 20. | CONTINGENCIES (CONTINUED) |
The Company is defending a claim from one of its former directors for amounts payable to him which he claims were to be settled in common shares. The plaintiff has claimed damages of approximately $300,000. The outcome of this claim is not determinable. |
The Company is defending a claim from one of its former consultants for breaching a settlement agreement dated Feb 17, 2011. The plaintiff is seeking payment of $37,356 plus interest. Subsequent to year end judgment was found in favour of the plaintiff. The settlement amount has not yet been finalized. |
It is the opinion of management, based in part on advice of legal counsel, that the ultimate resolution of these contingencies, to the extent not previously provided for, will not have a material adverse effect on the financial condition of the Corporation.
| 21. | MANAGEMENT OF CAPITAL |
The Company's objectives when managing capital are to safeguard its ability to continue as a going concern in order to pursue the development of its technologies and to maintain a flexible capital structure, which optimizes the costs of capital at an acceptable risk. The Company considers its capital for this purpose to be its shareholders' equity.
The Company's primary source of capital is through the issuance of equity. The Company manages and adjusts its capital structure when changes in economic conditions occur. To maintain or adjust the capital structure, the Company may seek additional funding. The Company may require additional capital resources to meet its administrative overhead expenses in the long term. The Company believes it will be able to raise capital as required in the long term, but recognizes there will be risks involved that may be beyond its control. There are no external restrictions on the management of capital.
| 22. | FINANCIAL INSTRUMENTS |
a) | Fair value In accordance with IFRS, financial instruments are classified into one of the five following categories: held-for-trading, held-to-maturity investments, loans and receivables, available-for-sale financial assets and other financial liabilities. Investments with quoted prices in active markets are designated as held-for-trading. Investments without quoted prices in active markets designated as available for sale and are carried at cost. Cash and trade and other receivable are classified as loans and receivables. Term deposits are classified as held-to-maturity. Their carrying value approximates fair value due to their limited time to maturity and ability to convert them to cash in the normal course of business. Trade payable and accrued liabilities, interest payable, short-term loan and advanced payable are classified as other financial liabilities. Their carrying values also approximate fair value due to their short term maturities. IFRS 13 establishes a fair value hierarchy that reflects the significance of inputs used in making fair value measurements as follows: |
Level 1 | quoted prices in active markets for identical assets or liabilities; |
Level 2 | inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly (i.e. as prices) or indirectly (i.e. from derived prices); and |
Level 3 | inputs for the asset or liability that are not based upon observable market data. |
171
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 22. | FINANCIAL INSTRUMENTS (CONTINUED) |
a) | Fair value (continued) The Company has determined the estimated fair values of its financial instruments based upon appropriate valuation methodologies. At September 30, 2014, cash of $135,171 (September 30, 2013 - $5,327), cash held in trust of $248,990 (September 30, 2013 - $nil) and marketable securities of $25,475 (September 30, 2013 - $nil) have been measured and recognized in the balance sheet using Level 1 inputs. Investments of $200,000 (September 30, 2013 - $nil) have been designated as available for sale and carried at cost on the balance sheet as at September 30, 2014, there was no observable market data available. At September 30, 2014 and 2013, there were no financial assets or liabilities measured and recognized in the balance sheet at fair value that would be categorized as Level 2 and 3 in the fair value hierarchy above. The Company's financial assets and financial liabilities are categorized as follows:
|
September 30, 2014 | September 30, 2013 | |||
Financial Assets | ||||
| Held-for-trading | ||||
Cash | $ | 135,171 | $ | 5,327 |
Marketable securities | 25,475 | - | ||
| Loans and receivable | ||||
Cash held in trust | 248,990 | - | ||
Term deposits | 949,692 | - | ||
Trade and other receivables | 152,693 | 26,763 | ||
| Available-for-sale | ||||
Investments | $ | 200,000 | $ | - |
Financial Liabilities | ||||
| Other financial liabilities | ||||
Trade and other payables | 858,181 | 727,610 | ||
Interest payable | - | 34,689 | ||
Short-term loans | - | 290,159 | ||
Advances payable | 18,871 | 18,871 |
b) | Financial risk management Credit Risk Credit risk is the risk that one party to a financial instrument will cause a financial loss for the other party by failing to discharge an obligation. The Company's cash, cash held in trust, term deposits and trade and other receivables are exposed to credit risk. The Company reduces its credit risk on cash and cash equivalents by placing these instruments with institutions of high credit worthiness. As at September 30, 2014 and 2013, the Company's exposure is the carrying value of the financial instruments. The Company's maximum exposure to credit risk is the carrying value of its financial assets. |
172
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 22. | FINANCIAL INSTRUMENTS (CONTINUED) |
b) | Financial risk management (continued) Liquidity risk Liquidity risk is the risk that an entity will encounter difficulty in raising funds to meet commitments associated with financial instruments. The Company manages liquidity by maintaining adequate cash balances to meet liabilities as they become due. The Company maintained cash at September 30, 2014 in the amount of $135,171 (September 30, 2013 - $5,327), in order to meet short-term business requirements. At September 30, 2014, the Company had accounts payable and accrued liabilities and advances payable of $824,862 and $18,871, respectively (September 30, 2013 - $$727,610 and $18,871, respectively). All accounts payable and accrued liabilities and advances payables are current. Market risk The significant market risks to which the Company is exposed are interest rate risk and currency risk. Interest rate risk Interest rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market interest rates. Included in the loss for the year in the financial statements is interest income on Canadian dollar cash and cash equivalents and term deposits. The Company is not exposed to significant other price risk. Currency risk The Company is exposed to currency risk to the extent that monetary assets and liabilities held by the Company are not denominated in Canadian dollars. The Company has not entered into any foreign currency contracts to mitigate this risk. The Company's cash and cash equivalents and accounts payable and accrued liabilities are partly held in US dollars ("USD"); therefore, USD accounts are subject to fluctuation against the Canadian dollar. The Company had the following balances in Canadian and foreign currencies as at September 31, 2014 and 2013: |
173
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 22. | FINANCIAL INSTRUMENTS (CONTINUED) |
b) | Financial risk management (continued) Market risk (continued) Currency risk (continued) |
As at September 30, 2014
| in CAD | in USD | |||
| Cash | $ | 128,811 | $ | 5,701 |
| Cash held in trust | 248,990 | - | ||
| Term deposit | 949,692 | - | ||
| Marketable securities | 25,475 | - | ||
| Amounts receivable | 128,150 | 22,000 | ||
| Investments | 200,000 | - | ||
| Accounts payable and accrued liabilities | (547,331) | (278,639) | ||
| Advances payable | (18,871) | - | ||
| 1,114,916 | (250,938) | |||
| Rate to convert to $1.00 CAD | 1.000 | 1.1156 | ||
| Equivalent to Canadian dollars | 1,114,916 | (279,946) |
Based on the above net exposures as at September 30, 2014, and assuming that all other variables remain constant, a 10% appreciation or depreciation of the CAD against the USD by 10% would increase/ decrease profit or loss by $27,995.
As at September 30, 2013
| in CAD | in USD | |||
| Cash | $ | 5,327 | $ | - |
| Amounts receivable | 26,763 | - | ||
| Accounts payable and accrued liabilities | (544,372) | (178,160) | ||
| Interest payable | (34,689) | - | ||
| Short-term loans | (290,159) | - | ||
| Advances payable | (18,871) | - | ||
| (856,001) | (178,160) | |||
| Rate to convert to $1.00 CAD | 1.000 | 1.0285 | ||
| Equivalent to Canadian dollars | (856,001) | (183,238) |
Based on the above net exposures as at September 30, 2013, and assuming that all other variables remain constant, a 10% appreciation or depreciation of the CAD against the USD by 10% would increase/ decrease profit or loss by $18,324.
174
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 23. | INCOME TAXES |
The following table reconciles the expected income taxes expense (recovery) at the Canadian statutory income tax rates to the amounts recognized in the consolidated statements of operations for the years ended September 30, 2014 and 2013:
September 30, 2014 | September 30, 2013 | |||
| Loss before taxes | $ | (7,651,401) | $ | (1,102,491) |
| Statutory income tax rate | 26.00% | 25.80% | ||
| Expected income tax recovery | (1,989,364) | (284,443) | ||
| Differences resulting from: | ||||
| Non-deductible items | 1,237,362 | 99,705 | ||
| Changes in estimates | (60,927) | - | ||
| Share issuance costs | (10,946) | - | ||
| Changes in tax rate | - | (57,522) | ||
| Functional currency adjustments | (1,161) | - | ||
| Foreign tax rate differences | (9,144) | - | ||
| Change in deferred tax asset not recognized | 794,319 | 242,260 | ||
| Total income taxes recovery | $ | (39,861) | $ | - |
| Current tax expense (recovery) | $ | - | $ | - |
| Deferred tax expense (recovery) | (39,681) | - | ||
| Total income taxes recovery | $ | (39,681) | $ | - |
The British Columbia corporate tax rate has increased during the year, resulting in an increase in the Company's combined statutory tax rate.
The Company recognized a deferred tax liability of $423,576 as a result of the business combination with Phytalytics LLC (Note 3).
175
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 23. | INCOME TAXES (CONTINUED) |
Deferred taxes reflect the tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and their tax values. Deferred tax assets (liabilities) at September 30, 2014 and 2013 are comprised of the following:
| Canada | September 30, 2014 | September 30, 2013 | ||
| Non-capital loss carry forwards | $ | 1,484,967 | $ | 773,025 |
| Other | 9,448 | - | ||
| Intangible assets | 17,258 | (10,266) | ||
| Property and equipment | 35,688 | 26,282 | ||
| Financial instruments | 2,902 | - | ||
| Financing costs | 40,005 | 6,908 | ||
| 1,590,268 | 795,949 | |||
| Deferred tax asset not recognized | (1,590,268) | (795,949) | ||
| Net deferred tax asset (liability) | $ | - | $ | - |
| USA | September 30, 2014 | September 30, 2013 | ||
| Net operating loss carryforwards | $ | 36,789 | $ | - |
| Financial instruments | (423,576) | - | ||
| Property and equipment | 3,072 | - | ||
| (383,715) | - | |||
| Deferred tax asset not recognized | - | - | ||
| Net deferred tax asset (liability) | $ | (383,715) | $ | - |
176
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 23. | INCOME TAXES (CONTINUED) |
The Company has non capital loss carryforwards of approximately $5,711,413 (2013 - $2,974,000) which may be carried forward to apply against future year income tax for Canadian tax purposes, subject to the final determination by taxation authorities, expiring in the following years:
| 2028 | $ | 65,764 |
| 2029 | 64,815 | |
| 2030 | 609,226 | |
| 2031 | 682,635 | |
| 2032 | 834,223 | |
| 2033 | 904,700 | |
| 2034 | 2,550,050 | |
| Total | $ | 5,711,413 |
The Company has net operating loss carryforwards of approximately $108,204 which may be carried forward to apply against future year income tax for US tax purposes.
| 2034 | $ | 108,204 |
| Total | $ | 108,204 |
| 24. | SEGMENTED INFORMATION |
The Company has one reportable operating segment of producing, licensing and marketing proprietary ingredients and formulas for use in the BioPharma, Nutraceutical, Cosmetic and Animal Nutrition markets. Non-current assets (other than financial instruments) by geographic location are as:
|
| Canada |
| US |
| Total |
As at September 30, 2014 |
|
|
|
|
|
|
Property and equipment | $ | 334,390 | $ | 458,634 | $ | 793,024 |
Intangible assets |
| 1,395,271 |
| 1,245,812 |
| 2,641,083 |
Other assets |
| 2,000 |
| - |
| 2,000 |
| $ | 1,731,661 | $ | 1,704,446 | $ | 3,436,107 |
As at September 30, 2013 |
|
|
|
|
|
|
Property and equipment | $ | 106,296 | $ | 375,000 | $ | 481,296 |
Intangible assets |
| 1,074,161 |
| - |
| 1,074,161 |
Other assets |
| 2,000 |
| - |
| 2,000 |
| $ | 1,182,457 | $ | 375,000 | $ | 1,557,457 |
177
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
| 25. | EVENTS AFTER THE REPORTING DATE |
Subsequent to September 30, 2014:
1,200,000 warrants were exercised for proceeds of $120,000. |
1,104,779 common shares issued to the Company's directors and officers for the services provided to the Company. | ||
72,414 common shares were issued to settle accounts payable. |
VDL sent a letter advising they were terminating the license agreement discussed in Note 19(iv) by citing that the Company failed to comply with certain terms and conditions included in the license agreement. The Company believes that the terms in the license agreement have been followed; as a result, the license agreement should be valid. The Company intends to continue to honor the agreement and make any payments or provide any information required under the license. The Company provides for costs related to contingencies when a loss is probable and the amount is reasonably determinable. In the opinion of management, no grounds exist that justify the termination of the license agreement. It is the opinion of management, based in part on advice of legal counsel, that the ultimate resolution of the termination of the license agreement is undeterminable; therefore there has been no provision made with respect to the license in the consolidated financial statements for the year ended September 30, 2014. |
On December 29, 2014, the Company obtained a obtained a preliminary injunction from the Washington state court in King County against Herbal Analytics, LLC, James Baxter, Kaleb Lund and Lauren Hilty (former employees of PhytaLab - see Note 3), and Erin Leary, Affinor Growers, LLC, and Nicholas Brusatore (collectively, "Injunction Defendants") . Affinor Growers, LLC is a wholly owned subsidiary of defendant Affinor Growers, Inc. According to the conclusions of law, the Injunction Defendants shall cease and desist from any and all use of PhytaLabs trade secrets and confidential information and documents; (b) are restrained from copying, transferring, using or disclosing to any other person or entity any documentation taken from PhytaLab; (c) shall retain and preserve all existing documents and files that mention, refer to, or are derived from PhytaLab, PhytaLab and Abattis' customers, or PhytaLab and Abattis' prospective customers; and (d) Herbal Analytics, LLC shall keep a detailed, complete, and accurate accounting of its business operations. |
On January 27, 2015, the Company granted 175,000 stock options to certain of its directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. |
On March 6, 2015 the Company granted 125,000 stock options to certain of its directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. | ||
The Company is defending a defamation claim from one of its former directors of the Company. Legal advice received supports the Company's belief that the claim is without merit. The outcome of this claim are not determinable and therefore no amounts have been recorded for any potential payments which may have to be made. |
178
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
For the year ended September 30, 2014
(Expressed in Canadian Dollars)
The Company is defending a claim from one of its former consultants for breaching the terms of an agreement. The plaintiff is claiming an entitlement to 5% of the common shares of Abattis Bioceuticals Corp., damages, punitive damages, and costs. The Company has offered to settle with the plaintiff for $55,000 and the issuance of 75,000 common shares. |
The Company was granted a patent which was originally acquired on April 16, 2009 from PRB and Pacific Bio. |
One of the directors of the Company forgave $235,418 in amounts owing to him for consulting services in exchange for cash settlement of $32,000. | ||
| The Company completed a private placement in the amount of 2,365,072 Units for gross proceeds of $307,460. Each Unit was issued at $0.13 per Unit and consists of one common share and one warrant to purchase a common share. Each warrant is exercisable into one common share at a price of $0.18 per common share for a period of 18 months after the closing date of the private placement. The warrants contain an acceleration component whereby the Company will have the right to call for the exercise of the warrants if the common shares of the Company close above $0.35 per share for a period of 10 consecutive business days after the four month and one day hold period on the common shares expires. | |
The Company terminated its agreement with Kodiak as described in note 15. | ||
| The Company entered into a US $25,000,000 equity line facility agreement (the "Agreement") with Dutchess opportunity Fund, II, LP ("Dutchess"). The Company has agreed to file a registration statement with the US Securities & Exchange Commission ("SEC") to register the shares that Abattis may issue to Dutchess under the Agreement. After the SEC has declared the registration statement to be effective, the Company has the right at its sole discretion to sell up to US $25,000,000 of common shares to Dutchess over a period of three years. The common shares will be issued at the current market price less permitted discounts in effect during such issuances. |
179

Consolidated Financial Statements
For the years ended September 30, 2013 and 2012
(Expressed in Canadian Dollars)
180
Abattis Bioceuticals Corp.
Consolidated Financial Statements
For the years ended September 30, 2013 and 2012
TABLE OF CONTENTS
Page | |
| Independent Auditor's Report | 1 |
| Consolidated Statements of Financial Position | 2 |
| Consolidated Statements of Loss and Comprehensive Loss | 3 |
| Consolidated Statements of Changes in Shareholders' Equity | 4 |
| Consolidated Statements of Cash Flows | 5 |
| Notes to the Consolidated Financial Statements | 6-29 |
181
INDEPENDENT AUDITOR'S REPORT
To the Shareholders of Abattis Bioceuticals Corp.
We have audited the consolidated financial statements of Abattis Bioceuticals Corp., which comprise the statements of financial position as at September 30, 2013 and 2012 and statements of loss and comprehensive loss, changes in shareholders' equity and cash flows for the years ended September 30, 2013 and 2012, and a summary of significant accounting policies and other explanatory information.
Management's Responsibility for the Financial Statements
Management is responsible for the preparation and fair presentation of these financial statements in accordance with International Financial Reporting Standards, and for such internal control as management determines is necessary to enable the preparation of financial statements that are free from material misstatement, whether due to fraud or error.
Auditor's Responsibility
Our responsibility is to express an opinion on these financial statements based on our audit. We conducted our audit in accordance with Canadian generally accepted auditing standards. Those standards require that we comply with ethical requirements and plan and perform the audit to obtain reasonable assurance about whether the financial statements are free from material misstatement.
An audit involves performing procedures to obtain audit evidence about the amounts and disclosures in the financial statements. The procedures selected depend on the auditor's judgment, including the assessment of the risks of material misstatement of the financial statements, whether due to fraud or error. In making those risk assessments, the auditor considers internal control relevant to the entity's preparation and fair presentation of the financial statements in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the entity's internal control. An audit also includes evaluating the appropriateness of accounting policies used and the reasonableness of accounting estimates made by management, as well as evaluating the overall presentation of the financial statements.
We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as at September 30, 2013 and 2012, and its financial performance and its cash flows for the years ended September 30, 2013 and 2012 in accordance with International Financial Reporting Standards.
Emphasis of Matter
Without qualifying our opinion, we draw attention to Note 1 in the financial statements which indicates that the Company incurred a net loss of $1,102,491 during the year ended September 30, 2013, has an accumulated deficit of $4,656,546 and has a working capital deficiency at September 30, 2013 of $999,440. These conditions, along with other matters as set forth in Note 1, indicate the existence of material uncertainties that may cast significant doubt about the Company's ability to continue as a going concern.
/s/ Hay and Watson
Chartered Accountants
Vancouver, British Columbia
January 28, 2014
182
Abattis Bioceuticals Corp.
Consolidated Statements of Financial Position
(Expressed in Canadian Dollars)
September 30, 2013 | September 30, 2012 | |||
| ASSETS | ||||
| CURRENT ASSETS | $ | $ | ||
| Cash and cash equivalents | 5,327 | 5,499 | ||
| Trade and other receivables (Note 10(a)) | 26,763 | 13,465 | ||
| Prepaid expenses and other deposits | 39,799 | 52,121 | ||
| 71,889 | 71,085 | |||
| Property and equipment (Note 4) | 481,296 | 500,976 | ||
| Intangible assets (Note 5) | 1,074,161 | 752,964 | ||
| Other assets | 2,000 | - | ||
| TOTAL ASSETS | $ | 1,629,346 | $ | 1,325,025 |
| LIABILITIES | ||||
| CURRENT LIABILITIES | ||||
| Trade and other payables (Note 10(a)) | $ | 727,610 | $ | 521,402 |
| Interest payable (Note 6&12) | 34,689 | 22,506 | ||
| Short-term loans (Note 6&12) | 290,159 | 157,650 | ||
| Advances payable (Note 7) | 18,871 | - | ||
| 1,071,329 | 701,558 | |||
| SHAREHOLDERS' EQUITY | ||||
| Share capital (Note 8) | 4,291,204 | 3,350,373 | ||
| Reserves | 923,359 | 827,149 | ||
| Accumulated deficit | (4,656,546) | (3,554,055) | ||
| TOTAL LIABILITIES AND SHAREHOLDERS' EQUITY | 558,017 | 623,467 | ||
| $ | 1,629,346 | $ | 1,325,025 |
Nature of operations and going concern (Note 1)
Commitments (Note 13)
Events after the reporting date (Note 16)
These consolidated financial statements were authorized for issue by the Board of Directors on January 28, 2014. They are signed on the company's behalf by:
| "Timothy Fealey" | "Mike Withrow" | |
| Director | Director | |
The accompanying notes are an integral part of these consolidated financial statements
183
Abattis Bioceuticals Corp.
Consolidated Statements of Loss and Comprehensive Loss
(Expressed in Canadian Dollars)
| For the years ended | ||||
September 30, 2013 | September 30, 2012 | |||
| REVENUE | ||||
| Sales | $ | 17,448 | $ | - |
| Cost of goods sold | (11,806) | - | ||
| GROSS PROFIT | 5,642 | - | ||
| EXPENSES | ||||
| Accounting and audit fees | 64,543 | 76,319 | ||
| Advertising | 70,145 | 68,549 | ||
| Amortization (Note 5) | 67,314 | 41,532 | ||
| Appraisal | - | 12,793 | ||
| Bank service charge | 1,047 | 3,625 | ||
| Depreciation (Note 4) | 19,680 | 3,019 | ||
| Domain names | 4,919 | 136,364 | ||
| Interest (Note 6&12) | 12,635 | 2,468 | ||
| Legal fees | 167,663 | 164,409 | ||
| Management and consulting fees | 313,433 | 274,350 | ||
| Office and general administration | 83,906 | 76,268 | ||
| Regulatory and transfer agents fees | 25,274 | 24,025 | ||
| Research | 123,559 | 124,308 | ||
| Share-based compensation (Note 8(c)) | 85,033 | 8,542 | ||
| Sponsorship | 10,000 | - | ||
| (1,049,151) | (1,016,571) | |||
| OTHER (EXPENSE) INCOME | ||||
| Loss on write-off of abandoned patent application (Note 5) | (189,489) | - | ||
| Gain on cancellation and settlement of trade payables | 132,013 | 19,884 | ||
| Foreign exchange (loss) gain | (6,489) | 1,434 | ||
| Other income | 4,983 | 2,864 | ||
| (58,982) | 24,182 | |||
| NET LOSS AND COMPREHENSIVE LOSS FOR THE YEAR | $ | (1,102,491) | $ | (992,389) |
| Basic and diluted loss per share | $ | 0.04 | $ | 0.09 |
| Weighted average number of common shares outstanding | 26,066,009 | 11,051,220 | ||
The accompanying notes are an integral part of these consolidated financial statements
184
Abattis Bioceuticals Corp.
Consolidated Statements of Changes in Shareholders' Equity
For the years ended September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| Reserves | |||||||||||||
| Number of common shares | Share capital | Equity settled share-based payments | Warrants | Total | Accumulated deficit | Total shareholders' equity | |||||||
| Balance, September 30, 2011 | 10,947,581 | $ | 2,422,110 | $ | 224,976 | $ | 505,180 | $ | 730,156 | $ | (2,561,666) | $ | 590,600 |
| Net loss and comprehensive loss for the year | - | - | - | - | - | (992,389) | (992,389) | ||||||
| Private placement | 1,200,000 | 205,486 | - | 94,514 | 94,514 | - | 300,000 | ||||||
| Share-based compensation | - | - | 8,542 | - | 8,542 | - | 8,542 | ||||||
| Shares cancelled | (2,050,000) | (205,000) | - | - | - | - | (205,000) | ||||||
| Shares issued for acquisition of assets | 3,509,090 | 400,000 | - | - | - | - | 400,000 | ||||||
| Shares issued as bonus | 800,000 | 100,000 | - | - | - | - | 100,000 | ||||||
| Shares issued in business combination | 200,000 | 30,000 | - | - | - | - | 30,000 | ||||||
| Shares issued for domain names | 2,727,272 | 136,364 | - | - | - | - | 136,364 | ||||||
| Shares issued as settlement of trade payables | 525,985 | 131,326 | - | - | - | - | 131,326 | ||||||
| Shares issued for consulting fees | 600,000 | 150,000 | - | - | - | - | 150,000 | ||||||
| Share issuance costs | - | (33,476) | - | - | - | - | (33,476) | ||||||
| Warrants exercised for cash | 30,000 | 13,563 | - | (6,063) | (6,063) | - | 7,500 | ||||||
| Balance, September 30, 2012 | 18,489,928 | 3,350,373 | 233,518 | 593,631 | 827,149 | (3,554,055) | 623,467 | ||||||
| Net loss and comprehensive loss for the year | - | - | - | - | - | (1,102,491) | (1,102,491) | ||||||
| Private placement | 2,100,000 | 92,172 | - | 12,828 | 12,828 | - | 105,000 | ||||||
| Share issuance costs | - | (4,400) | - | - | - | - | (4,400) | ||||||
| Share-based compensation | - | - | 85,033 | - | 85,033 | - | 85,033 | ||||||
| Shares issued for acquisition of assets (Note 8) | 6,400,000 | 578,000 | - | - | - | - | 578,000 | ||||||
| Shares issued as settlement of trade payables | 1,494,166 | 263,408 | - | - | - | - | 263,408 | ||||||
| Options exercised for cash | 100,000 | 11,651 | (1,651) | - | (1,651) | - | 10,000 | ||||||
| Balance, September 30, 2013 | 28,584,094 | $ | 4,291,204 | $ | 316,900 | $ | 606,459 | $ | 923,359 | $ | (4,656,546) | $ | 558,017 |
The accompanying notes are an integral part of these consolidated financial statements
185
Abattis Bioceuticals Corp.
Consolidated Statements of Cash Flows
(Expressed in Canadian Dollars)
| For the years ended | ||||
September 30, 2013 | September 30, 2012 | |||
| Cash provided by (used in): | ||||
| OPERATING ACTIVITIES | ||||
Net loss and comprehensive loss for the year | $ | (1,102,491) | $ | (922,389) |
Adjustments for non-cash / non-operating items: | ||||
Amortization | 67,314 | 41,532 | ||
Depreciation | 19,680 | 3,019 | ||
Loss on write-off of patent application | 189,489 | - | ||
Loss (gain) on settlement on trade payables | 72,987 | (19,884) | ||
Shares issued as bonus | - | 100,000 | ||
Shares issued for consulting fees | 67,322 | 100,000 | ||
Shares issued for domain names | - | 136,364 | ||
Share-based compensation | 85,033 | 8,542 | ||
| (600,666) | (622,816) | |||
Net changes in working capital items: | ||||
Trade and other receivables | (13,298) | 25,151 | ||
Trade and other payables | 371,630 | 343,161 | ||
Interest payable | 12,183 | 2,285 | ||
| (230,151) | (252,219) | |||
| FINANCING ACTIVITIES | ||||
Common shares issued for cash, net of cash share issuance costs | 100,600 | 274,024 | ||
Funding received from Investment Agriculture Foundation | 18,871 | - | ||
Line of credit assumed on acquisition of Northern Vine Canada Inc. | - | (69,994) | ||
Short-term loan received from related party | 102,509 | 157,650 | ||
Options exercised for cash | 10,000 | - | ||
| 231,979 | 361,680 | |||
| INVESTING ACTIVITIES | ||||
Acquisition of licenses | - | (81,000) | ||
Acquisitions of other assets | (2,000) | - | ||
Expenditure on license applications | - | (3,188) | ||
Pre-acquisition advances to Northern Vine Canada Inc. | - | (25,640) | ||
| (2,000) | (109,828) | |||
| Decrease in cash | (172) | (367) | ||
| Cash and cash equivalents, beginning of the year | 5,499 | 5,866 | ||
| Cash and cash equivalents, end of the year | $ | 5,327 | $ | 5,499 |
| Cash and cash equivalents are comprised of: | ||||
Cash in bank | $ | 5,327 | $ | 5,499 |
Supplemental cash flow information (Note 10(b))
The accompanying notes are an integral part of these consolidated financial statements
186
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 1. | NATURE OF OPERATIONS AND GOING CONCERN |
Abattis Bioceuticals Corp. (the "Company" or "Abattis") was incorporated as Sinocan Capital Group Inc. under the Company Act (Canada British Columbia) on June 30, 1997 and listed and began trading on the Canadian National Stock Exchange ("the Exchange") under the symbol "FLU" on December 23, 2010. The Company's head office is located at Suite 1000 - 355 Burrard Street, Vancouver, British Columbia, V6C 2G8, Canada.
Abattis Bioceuticals Corporation is a biotechnology company with capabilities, through its wholly owned subsidiaries, of producing, licensing and marketing proprietary ingredients and formulas for use in the BioPharma, Nutraceutical, Cosmetic and Animal Nutrition markets. The Company has a pipeline of proprietary products ready for sale in the Functional Foods and Supplements business.
These financial statements have been prepared on a going concern basis, which assumes the realization of assets and liquidation of liabilities in the normal course of business in the foreseeable future. These consolidated financial statements do not include any adjustments to the carrying value and classification of recorded asset amounts and classification of liabilities that might be necessary should the Company be unable to continue as a going concern.
During the year ended September 30, 2013, the Company incurred a net loss of $1,102,491 (September 30, 2013 - $992,389). As at September 30, 2013, the Company had a working capital deficiency of $999,440 (September 30, 2012 - $630,473) and an accumulated deficit of $4,656,546 (September 30, 2012 - $3,554,055).
The Company's operations to date have been financed by issuing common shares, debt instruments and government assistance. The Company's ability to continue as a going-concern is dependent upon profitable commercialization of its technologies and the continuing ability to obtain debt or equity financing to fund ongoing operations and research and development activities. The current cash position on hand and expected cash flows for the next 12 months are not sufficient to fund the Company's ongoing operational needs. Therefore, the Company will need funding through equity or debt financing, joint venture arrangements or a combination thereof. There is no assurance that additional funding or suitable joint venture arrangements will be available on a timely basis or on terms acceptable to the Company. If the Company is unable to obtain sufficient funding in this fashion, the ability of the Company to meet its obligations as they come due and, accordingly, the appropriateness of the use of the going concern assumption will be in significant doubt.
| 2. | BASIS OF PREPARATION |
(a) Statement of compliance
These consolidated financial statements have been prepared in accordance with International Financial Reporting Standards ("IFRS") as issued by the International Accounting Standards Board ("IASB").
(b) Basis of presentation
These consolidated financial statements have been prepared on a historical cost basis except for financial instruments that have been measured at fair value. In addition, the consolidated financial statements have been prepared using the accrual basis of accounting, except for cash flow information.
187
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 2. | BASIS OF PREPARATION (continued) |
(c) Presentation and functional currency
The presentation and functional currency of the Company and its subsidiaries is the Canadian dollar. All amounts in these consolidated financial statements are expressed in Canadian dollars, unless otherwise indicated.
(d) Significant accounting estimates and judgments
The preparation of these consolidated financial statements requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements, and expenses during the reporting period. The estimates and associated assumptions are based on historical experience and various other factors that are believed to be reasonable under the circumstances. Actual outcomes could differ from these estimates. The consolidated financial statements include estimates which, by their nature, are uncertain. The estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimate is revised if the revision affects only that period or in both the period of revision and future periods if the revision affects both current and future periods.
Significant estimates are estimates and assumptions about the future and other sources of estimation uncertainty that management has made, that could result in a material adjustment to the carrying amounts of assets and liabilities. Significant estimates used in the preparation of these consolidated financial statements include, but are not limited to, the following:
| the recoverability of trade and other receivables and prepaid expenses and other deposits; |
| the carrying value and recoverability of intangible assets and the related amortization; |
| the inputs used in measuring share based payments; |
| the valuation of deferred tax assets; and |
| the inputs used in determining the estimated fair values of options and warrants issued during the year. |
While management believes the estimates contained within these consolidated financial statements are reasonable, actual results could differ from those estimates and could impact future results of operations and cash flows.
Significant accounting judgments are accounting policies that have been identified as being complex or involving subjective judgments or assessments. Critical accounting judgments used by the Company include, but are not limited to, the following:
| the estimated useful lives of property and equipment and the related depreciation; |
| the estimated future operating results; and |
| the net cash flows from property and equipment and intangible assets. |
188
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES |
The significant accounting policies used in the preparation of these consolidated financial statements are as follows:
(a) Basis of consolidation
These consolidated financial statements include the accounts of the Company and its subsidiaries. Subsidiaries are controlled by the Company. Control exists when the parent entity has the power to govern the financial and operating policies of an entity so as to obtain benefits from its activities. Subsidiaries are included in the consolidated financial statements from the date control is obtained until the date control is lost.
At September 30, 2013, the Company's subsidiaries are as follows:
Name | Country of Incorporation | Principal Activity |
Company Interest |
| Animo Wellness Corporation ("Animo") | Canada | Holds certain licenses | 100% |
| BioCell Labs Inc. ("BLI") | Canada | Biotechnology | 100% |
| North American BioExtracts Inc. ("NAB") | Canada | Biotechnology | 100% |
| Northern Vine Canada Inc. ("Northern Vine") | Canada | Biotechnology | 100% |
Intercompany transactions and balances between the Company and its subsidiaries are eliminated in full on consolidation.
(b) Business combinations
The Company accounts for a transaction as a business combination when the acquisition of an asset or group of assets constitutes a business and when the Company obtains control of the entity being acquired.
Business combinations are accounted for using the acquisition method. In applying the acquisition method, the Company separately records the identifiable assets acquired, the liabilities assumed, any goodwill acquired and any non-controlling interests in the acquired entity.
The Company measures the identifiable assets acquired and the liabilities assumed at their acquisition-date fair values, less any non-controlling interest at fair value. Goodwill is measured as the excess of the fair value of the consideration transferred, less any non-controlling interest in the entity being acquired over the fair value of the net identifiable assets acquired. The consideration transferred in a business combination is measured as the aggregate of the acquisition date fair values of the assets transferred by the acquirer, the liabilities incurred by the acquirer to former owners of the acquired entity and the equity interests issued by the Company.
Acquisition costs in connection with a business combination are expensed as incurred. Those costs include finder's fees, professional fees, consulting fees and general administrative costs.
(c) Foreign currency
Transactions in currencies other than the functional currency are recorded at the exchange rates prevailing on the dates of the transactions. At each statement of financial position reporting date, monetary assets and liabilities that are denominated in foreign currencies are translated at the exchange rates prevailing at the date of the statement of financial position. Non-monetary items that are measured in terms of historical cost in a foreign currency are translated using the exchange rates at the acquisition dates.
189
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(c) Foreign currency (continued)
Gains and losses arising from this translation are included in profit or loss for the period.
(d) Financial instruments
Financial assets and financial liabilities are recognized on the statements of financial position when the Company becomes a party to the contractual provisions of the financial instrument. All financial instruments are initially recorded at fair value.
Financial assets
The Company classifies its financial assets at initial recognition as either financial assets at fair value through profit or loss, loans and receivables, held-to-maturity investments or available-for-sale, depending on the purpose for which the asset was acquired. The Company's accounting policy for each of these categories is as follows:
Fair value through profit or loss - This category comprises derivatives or financial assets acquired or incurred principally for the purpose of selling or repurchasing in the near term. Subsequent to initial recognition, they continue to be recorded in the statement of financial position at fair value with changes in fair value recognized in profit or loss.
Loans and receivables - These assets are non-derivative financial assets with fixed or determinable payments that are not quoted in an active market. Subsequent to initial recognition, they are recorded at amortized cost less any provision for impairment. Individually significant receivables are considered for impairment when they are past due or when other objective evidence is received that a specific counterparty will default.
Held to maturity investments - These assets are non-derivative financial assets with fixed or determinable payments and fixed maturities that the Company's management has the positive intention and ability to hold to maturity. Subsequent to initial recognition, these assets are recorded at amortized cost using the effective interest rate method. If there is objective evidence that the investment is impaired, determined by reference to external credit ratings and other relevant indicators, the financial asset is measured at the present value of estimated future cash flows. Any changes to the carrying amount of the investment, including impairment losses, are recognized in profit or loss.
Available for sale - Non-derivative financial assets not included in the above categories are classified as available for sale. Subsequent to initial recognition, they continue to be recorded at fair value with changes in fair value recognized directly in equity. Where a decline in the fair value of an available for sale financial asset constitutes objective evidence of impairment, the amount of the loss is removed from equity and recognized in profit or loss.
The Company's financial assets are cash and cash equivalents and trade and other receivables. The Company classifies its cash and cash equivalents as financial assets at fair value through profit or loss and its trade and other receivables as loans and receivables.
Transactions costs associated with financial assets at fair value through profit or loss are expensed as incurred, while transaction costs associated with all other financial assets are included in the initial carrying amount of the asset.
190
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(d) Financial instruments (continued)
All financial assets except for those at fair value through profit or loss are subject to review for impairment at least at each reporting date. Financial assets are impaired when there is any objective evidence that a financial asset or a group of financial assets is impaired.
Financial liabilities
The Company classifies its financial liabilities as either financial liabilities at fair value through profit or loss or other financial liabilities, depending on the purpose for which the liability was incurred. The Company's accounting policy for each of these categories is as follows:
Fair value through profit or loss: This category comprises derivatives or liabilities acquired or incurred principally for the purpose of selling or repurchasing in the near term. Subsequent to initial recognition, they continue to be recorded in the statement of financial position at fair value with changes in fair value recognized in profit or loss.
Other financial liabilities: Financial liabilities other than those classified as fair value through profit or loss are classified as other financial liabilities. Subsequent to initial recognition, other financial liabilities are measured at amortized cost using the effective interest rate method.
The Company's financial liabilities are trade and other payables, interest payable and the short-term loans. The Company classifies these financial liabilities as other financial liabilities.
The Company does not have any derivative financial instruments.
The Company classifies and discloses fair value measurements based on a three-level hierarchy:
| Level 1 - inputs are unadjusted quoted pries in active markets for identical assets or liabilities; |
| Level 2 - inputs other than quoted pries in Level 1 that are observable for the asset or liability, either directly or indirectly; and |
| Level 3 - inputs for the asset or liability that are not based on observable market |
(e) Cash and cash equivalents
Cash and cash equivalents in the statements of financial position comprise cash, bank deposits and short-term investments that are readily converted to known amounts of cash with original maturities of three months or less.
(f) Property and equipment
Property and equipment is carried at cost, less accumulated depreciation and accumulated impairment losses.
The cost of an item of property and equipment consists of the purchase price, any costs directly attributable to bringing the asset to the location and condition necessary for its intended use and an initial estimate of the costs of dismantling and removing the item and restoring the site on which it is located.
191
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(f) Property and equipment (continued)
Property and equipment is depreciated annually on the following basis:
| Computer equipment | - 30% | declining-balance |
| Office equipment | - 30% | declining-balance |
| Plant equipment | - 20% | declining-balance |
| Leasehold improvement | - 20 years | straight-line |
Depreciation commences when an item of equipment becomes available for use.
An item of property and equipment is derecognized upon disposal or when no future economic benefits are expected to arise from the continued use of the asset. Any gain or loss arising on disposal of the asset, determined as the difference between the net disposal proceeds and the carrying amount of the asset, is recognized in profit or loss.
Where an item of property and equipment comprises major components with different useful lives, the components are accounted for separately. Expenditures incurred to replace a component of an item of property and equipment that is accounted for separately, including major inspection and overhaul expenditures, are capitalized.
(g) Intangible assets
Research and development
Expenditure on research activities, undertaken with the prospect of gaining new scientific or technical knowledge and understanding, is recognized in profit or loss as incurred.
Development activities involve a plan or design for the production of new or substantially improved products and processes. Development expenditure is capitalized only if development costs can be measured reliably, the product or process is technically and commercially feasible, future economic benefits are probable, and the Company intends to and has sufficient resources to complete development and has the ability to use or sell the asset. The expenditures capitalized includes the cost of materials, direct labor, overhead costs that are directly attributable to preparing the asset for its intended use, and borrowing costs on qualifying assets for which the commencement date for capitalization is on or after October 1, 2010. Other development expenditure is recognized in profit or loss as incurred.
Capitalized development expenditure is measured at cost less accumulated amortization and accumulated impairment losses.
Other intangible assets
Other intangible assets that are acquired by the Company and have finite useful lives are measured at cost less accumulated amortization and accumulated impairment losses.
Intangible assets are amortized annually on a straight-line basis at the following rates:
| Patents | - 20 years |
| Formulae | - 20 years |
| Licenses | - 20 years |
192
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(h) Impairment of non-current assets
At each reporting date, the carrying amounts of the Company's assets are reviewed to determine whether there is any indication that those assets are impaired. If any such indication exists, the recoverable amount of the asset is estimated in order to determine the extent of the impairment, if any. Where the asset does not generate cash flows that are independent from other assets, the Company estimates the recoverable amount of the cash-generating unit to which the asset belongs.
The recoverable amount of an asset is the higher of fair value less costs to sell and value in use. Fair value is determined as the amount that would be obtained from the sale of the asset in an arm's length transaction between knowledgeable and willing parties. In assessing value in use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset. If the recoverable amount of an asset or cash generating unit is estimated to be less than its carrying amount, the carrying amount of the asset is reduced to its recoverable amount and the impairment loss is recognized in profit or loss.
Where an impairment loss subsequently reverses, the carrying amount of the asset (or cash generating unit) is increased to the revised estimate of its recoverable amount, to the extent the revised carrying amount does not exceed the carrying amount that would have been determined had no impairment loss been recognized for the asset (or cash generating unit) in prior years. A reversal of an impairment loss is recognized in profit or loss.
(i) Share capital
Common shares
Common shares are classified as equity. Transaction costs directly attributable to the issue of common shares and share options are recognized as a deduction from equity, net of any tax effects.
Equity units
Proceeds received on the issuance of units, are allocated between the common shares and warrants using the relative fair value method. The fair value of the warrants is determined using the Black Scholes valuation model on the date the units are issued.
(j) Loss per share
The Company presents basic and diluted loss per share data for its common shares, calculated by dividing the loss attributable to common shareholders of the Company by the weighted average number of common shares outstanding during the period. Diluted loss per share is determined by adjusting the loss attributable to common shareholders and the weighted average number of common shares outstanding for the effects of all dilutive potential common shares.
193
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(k) Share-based compensation
The Company's share purchase option plan allows directors, executive officers, employees and consultants to acquire shares of the Company. The fair value of options granted is recognized as a share-based compensation expense with a corresponding increase in equity. An individual is classified as an employee when the individual is an employee for legal or tax purposes (direct employee) or provides services similar to those performed by a direct employee.
The fair value of employee options is measured at the option's grant date, and the fair value of non-employee options is measured at the date when goods or services are received. The fair value of each tranche of options granted which do not vest immediately on grant, is recognized over the period during which each tranche of options vest. The fair value of the options granted is measured using the Black-Scholes option pricing model taking into account the terms and conditions upon which the options were granted. At each financial position reporting date, the amount recognized as an expense is adjusted to reflect the actual number of share options that are expected to vest.
Share-based compensation expense is credited to the equity settled share-based payment reserve. If the options are later exercised, their fair value is transferred from the reserve to share capital.
(l) Provisions
Provisions are recognized where a legal or constructive obligation has been incurred as a result of past events, it is probable that an outflow of resources embodying economic benefit will be required to settle the obligation, and a reliable estimate of the amount of the obligation can be made. If material, provisions are measured at the present value of the expenditures expected to be required to settle the obligation.
(m) Income taxes
Income tax on the profit or loss for the periods presented comprises of current and deferred taxes. Income tax is recognized in profit or loss except to the extent that it relates to items recognized directly in equity, in which case it is recognized as equity.
Current tax expense is the expected tax payable on the taxable income for the year, using tax rates enacted or substantively enacted at the end of a period, adjusted for amendments to tax payable with regards to previous years.
Deferred tax assets and liabilities are computed using the asset and liability method on temporary differences between the carrying amounts of assets and liabilities on the statement of financial position and their corresponding tax values, using the enacted or substantially enacted income tax rates at each statement of financial position date. Deferred tax assets also result from unused tax losses and other deductions carried forward. Deferred tax assets are recognized only to the extent that they are probable that future taxable profits will be available against which the assets can be utilized. A valuation allowance is used to reflect the estimated realizable amount of deferred tax assets.
Deferred tax assets and liabilities are offset when there is a legally enforceable right to set off current tax assets against current tax liabilities and when they relate to income taxes levied by the same taxation authority and the Company intends to settle its current tax assets and liabilities on a net basis.
194
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(n) New standards and interpretations not yet adopted
The following is a summary of new standards, amendments and interpretations that have been issued but not yet adopted in these annual financial statements:
IFRS 7, Financial Instruments: Disclosures ("IFRS 7") - amendments
In December 2011, the IASB issued new disclosure requirements for financial assets and liabilities that (1) are offset in the statement of financial position; or (2) subject to master netting agreements or similar arrangements. These new disclosure requirements are effective for annual periods beginning on or after January 1, 2013 and are to be applied retrospectively. The Company is currently evaluating the impact of these new disclosure requirements on its financial statement; however, the impact, if any, is not expected to be significant.
IFRS 9, Financial Instruments ("IFRS 9")
IFRS 9 uses a single approach to determine whether a financial asset is measured at amortized cost or fair value, replacing the multiple classification options in IAS 39. The approach in IFRS 9 is based on how an entity manages its financial impairment methods in IAS 39. IFRS 9 is effective for annual periods beginning on or after January 1, 2015, with earlier adoption permitted. The Company is currently evaluating the impact of IFRS 9 on its financial instruments; however, the impact, if any, is not expected to be significant.
IFRS 10, Consolidated Financial Statements ("IFRS 10")
IFRS 10 replaces the consolidation requirements in IAS 27, Consolidated and Separate Financial Statements, and Standing Interpretations Committee ("SIC") Interpretation 12, Consolidation - Special Purpose Entities. IFRS 10 introduces a single consolidation model for all entities based on control, irrespective of the nature of the investee, and is effective for annual periods beginning on or after January 1, 2013, with earlier application permitted. The Company currently does not anticipate the adoption of IFRS 10 to have a significant impact on its financial statements.
IFRS 11, Joint Arrangements ("IFRS 11")
In May 2011, the IASB issued guidance establishing principles for financial reporting by parties to a joint arrangement. IFRS 11 replaces IAS 31, Interests in Joint Ventures. Under IFRS 11, joint arrangements are classified as joint operations or joint ventures based on the rights and obligations of the parties to the joint arrangements. A joint operation is a joint arrangement whereby the parties that have joint control of the arrangement ("joint operators") have direct rights to the assets, and obligations for the liabilities, relating to and in accordance with the arrangement.
A joint venture is a joint arrangement whereby the parties that have joint control of the arrangement ("joint venturers") have rights to the net assets of the arrangement. IFRS 11 requires that a joint operator recognize its portion of assets, liabilities, revenues and expenses of a joint arrangement, while a joint venturer recognizes its investment in a joint arrangement using the equity method. IFRS 11 is effective for annual periods beginning on or after January 1, 2013, with early adoption permitted. The Company is reviewing the standard to determine the potential impact, if any, on its financial statements.
195
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 3. | SIGNIFICANT ACCOUNTING POLICIES (continued) |
(n) New standards and interpretations not yet adopted (continued)
IFRS 12, Disclosure of Interests in Other Entities ("IFRS 12")
IFRS 12 requires the disclosure of information that enables users of financial statements to evaluate the nature of, and risks associated with, its interests in other entities and the effects of those interests on its financial position, financial performance and cash flows. The disclosure requirements are applicable to all forms of interests in other entities, including subsidiaries, joint arrangements, associates and unconsolidated structured entities. IFRS 12 is effective for annual periods beginning on or after January 1, 2013 with earlier adoption permitted. The Company is reviewing the standard to determine the potential impact, if any, on its financial statements.
IFRS 13, Fair Value Measurement ("IFRS 13")
In May 2011, the IASB issued guidance establishing a single source for fair value measurement. IFRS 13 defines fair value, provides guidance on how to determine fair value and requires disclosures about fair value measurements. However, IFRS 13 does not change the requirements regarding which items should be measured or disclosed at fair value. Rather, the measurement and disclosure requirements of IFRS 13 apply when another standard requires or permits the item to be measured at fair value, with limited exceptions. IFRS 13 is effective for annual periods beginning on or after January 1, 2013 with early application permitted. The Company is currently evaluating the impact of this standard on its financial statements.
IAS 28, Investments in Associates and Joint Ventures ("IAS 28")
IAS 28 was amended as a consequence of the issuance of IFRS 10, 11 and 12. IAS 28 provides additional guidance for investments in associates and sets out the requirements for the application of the equity method when accounting for investments in associates and joint ventures. This standard will be applied by the Company when there is joint control or significant influence over an investee. Amendments to IAS 28 are effective for annual periods beginning on or after January 1, 2013 with early application permitted. The Company is in the process of evaluating the impact of the new standard on its financial statements.
IAS 32, Financial Instruments: presentation ("IAS 32")
In December 2011, the IASB issued amendments to IAS 32. The amendments clarify that an entity currently has a legally enforceable right to set-off financial assets and liabilities if that right is (1) not contingent on a future event; and (2) enforceable both in the normal course of business and in the event of default, insolvency or bankruptcy of the entity and all counterparties. These amendments are effective for annual periods beginning on or after January 1, 2014 with early application permitted and are to be applied retrospectively. The Company is currently evaluating the impact of the adoption of the amendments on its financial statement; however, the impact, if any, is not expected to be significant.
196
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 4. | PROPERTY AND EQUIPMENT |
| Computer Equipment | Office Equipment | Plant Equipment | Leasehold Improvements | Total | ||||||
| COST | ||||||||||
Balance at September 30, 2011 | $ | 2,047 | $ | - | $ | - | $ | - | $ | 2,047 |
Additions / disposals | - | 14,792 | 440,000 | 47,463 | 502,255 | |||||
Balance at September 30, 2012 | 2,047 | 14,792 | 440,000 | 47,463 | 504,302 | |||||
Additions / disposals | - | - | - | - | - | |||||
Balance at September 30, 2013 | $ | 2,047 | $ | 14,792 | $ | 440,000 | $ | 47,463 | $ | 504,302 |
| ACCUMULATED DEPRECIATION | ||||||||||
Balance at September 30, 2011 | $ | 307 | $ | - | $ | - | $ | - | $ | 307 |
Depreciation for the year | 522 | 559 | 1,638 | 300 | 3,019 | |||||
Balance at September 30, 2012 | 829 | 559 | 1,638 | 300 | 3,326 | |||||
Depreciation for the year | 365 | 4,270 | 12,672 | 2,373 | 19,680 | |||||
Balance at September 30, 2013 | $ | 1,194 | 4,829 | $ | 14,310 | 2,673 | $ | 23,006 | ||
| CARRYING AMOUNTS | ||||||||||
At September 30, 2011 | $ | 1,740 | $ | - | $ | - | $ | - | $ | 1,740 |
At September 30, 2012 | 1,218 | 14,233 | 438,362 | 47,163 | 500,976 | |||||
At September 30, 2013 | $ | 853 | $ | 9,963 | $ | 425,690 | $ | 44,790 | $ | 481,296 |
| 5. | INTANGIBLE ASSETS |
| Patents | Formulae | Licenses | Total | |||||
| COST | ||||||||
Balance at September 30, 2011 | $ | 500,000 | $ | 250,000 | $ | - | $ | 750,000 |
Additions / disposals | - | - | 116,331 | 116,331 | ||||
Balance at September 30, 2012 | 500,000 | 250,000 | 116,331 | 866,331 | ||||
Additions / disposals | - | 78,000 | 500,000 | 578,000 | ||||
Write-off of patent application | (250,000) | - | - | (250,000) | ||||
Balance at September 30, 2013 | $ | 250,000 | $ | 328,000 | $ | 616,331 | $ | 1,194,331 |
| ACCUMULATED AMORTIZATION | ||||||||
Balance at September 30, 2011 | $ | 66,681 | $ | 5,154 | $ | - | $ | 71,835 |
Amortization | 27,208 | 12,534 | 1,790 | 41,532 | ||||
Balance at September 30, 2012 | 93,889 | 17,688 | 1,790 | 113,367 | ||||
Amortization | 27,134 | 14,487 | 25,693 | 67,314 | ||||
Write-off of patent application | (60,511) | - | - | (60,511) | ||||
Balance at September 30, 2013 | $ | 60,512 | $ | 32,175 | $ | 27,483 | $ | 120,170 |
| CARRYING AMOUNTS | ||||||||
At September 30, 2011 | $ | 433,319 | $ | 244,846 | $ | - | $ | 678,165 |
At September 30, 2012 | 406,111 | 232,312 | 114,541 | 752,964 | ||||
At September 30, 2013 | $ | 189,488 | $ | 295,825 | $ | 588,848 | $ | 1,074,161 |
197
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 6. | SHORT-TERM LOANS |
A continuity schedule of the Company's short-term loans for the year ended September 30, 2013 and the year ended September 30, 2012 is as follows:
| Loans from CEO | ||
| Balance at September 30, 2012 | $ | 157,650 |
| Additions | 132,509 | |
| Balance at September 30, 2013 | $ | 290,159 |
During 2012, the Company obtained a $140,000 loan from the Chief Executive Officer ("CEO") of the Company, which bears interest at a rate of 8%. The interest on the loan is payable on a monthly basis, and the principal was repayable six months from the date of advance. During the year ended September 30, 2013, $12,182 (2012 - $2,286) in interest was accrued. The loan is secured by all of the Company's property and assets.
During the year ended September 30, 2013, an additional $102,509 (2012 - $17,650) was advanced by the CEO of the Company without interest and $30,000 in common shares of the Company owned by the CEO were transferred to investor relation consultants on behalf of the Company.
On January 20, 2013, the Company and the CEO agreed to extend the repayment date of the short-term loans provided by the CEO unless either parties wishes to intervene and at that time, the loan must be paid in full by the Company. Although the loan must be paid in full by the Company, the parties continue discussions regarding partial repayment and extensions.
| 7. | ADVANCES PAYABLE |
On January 30, 2013, the Investment Agriculture Foundation provided $18,871 to a subsidiary acquired by the Company on March 1, 2013 to develop high value, high quality fractionation processes for surplus berries. Focus has moved away from this project during the year and therefore funds advanced by the Investment Agriculture Foundation will be repaid.
198
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 8. | SHARE CAPITAL |
(a) Common shares
Authorized
There are an unlimited number of common shares without par value authorized for issuance.
Issued
Year ended September 30, 2012
On October 12, 2011, the Company issued 100,000 (pre consolidation 500,000) common shares to the Company's CEO, with a fair value of $22,500 to settle trade payable of $25,000.
On January 24, 2012, the Company terminated its prior agreement with the Hensley Group Inc. dated October 8, 2010 pursuant to which it had been granted the exclusive license to make, have made, use and sell the nutraceutical anti-viral nasal spray product code named ZITEK. Further to the termination of the Company's agreement with the Hensley Group Inc., on February 16, 2012, 2,050,000 (pre consolidation 10,250,000) common shares previously issued in relation to the agreement, with a fair value of $205,000 were cancelled and returned to treasury.
On February 7, 2012, the Company issued 140,000 (pre consolidation 700,000) common shares to the Company's CEO, with a fair value of $38,500 to settle a trade payable of $49,000.
On February 14, 2012, the Company completed a non-brokered private placement of 1,200,000 (pre consolidation 6,000,000) units for gross proceeds of $300,000. Each unit consists of one common share and one share purchase warrant of the issuer. Each warrant is exercisable into a share for a period of 12 months at an exercise price of $0.35 (pre consolidation $0.07) per share. $205,486 of the proceeds was allocated to share capital and $94,514 of the proceeds was allocated to the warrant reserve. Cash share issuance costs of $33,476 were incurred.
On February 21, 2012, the Company issued 57,143 (pre consolidation 285,714) common shares to a consultant of the Company, with a fair value of $20,000 for consulting services.
On March 16, 2012, the Company issued 100,000 (pre consolidation 500,000) common shares, with a fair value of $25,000, as partial consideration for the acquisition of 100% of the outstanding shares of Animo Wellness Corporation.
On March 26, 2012, 30,000 (pre consolidation 150,000) warrants were exercised at an exercise price of $0.25 (pre consolidation $0.05) per warrant for total proceeds of $7,500.
On June 13, 2012 and August 15, 2012, the Company issued 600,000 (pre consolidation 3,000,000) common shares in total, with a fair value of $150,000 to Wakabayashi Fund LLC for consulting services.
On August 15, 2012, the Company issued 200,000 (pre consolidation 1,000,000) common shares, with a fair value of $30,000 to acquire 100% of the outstanding shares of Northern Vine Canada Inc. On the same day the Company issued 68,842 (pre consolidation 344,209) common shares to settle $17,210 of Northern Vine Canada Inc.'s loan payable to the former shareholders of that company.
On August 21, 2012, the Company issued 800,000 (pre consolidation 4,000,000) common shares to certain directors, employees and consultants of the Company as a bonus for their services provided to the Company.
199
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 8. | SHARE CAPITAL (continued) |
(a) Common shares (continued)
On August 21, 2012, the Company issued 160,000 (pre consolidation 800,000) common shares to a director of the Company, with a fair value of $40,000, to settle trade payable of $40,000.
On September 11, 2012, the Company consolidated its common shares on the basis of five pre consolidation common shares for one new post consolidation common share, such that the 61,767,823 issued and outstanding common shares on that date were consolidated into 12,353,565 common shares.
On September 27, 2012, the Company issued 3,409,090 common shares to the Company's CEO to acquire plant equipment with an estimated fair value of $375,000 and 2,727,272 common shares to acquire domain names with an estimated fair value of $136,364. Acquisition costs of domain names were expensed in the year.
Year ended September 30, 2013
On December 12, 2012, the Company completed a non-brokered private placement of 2,100,000 units at a price of $0.05 per unit for gross proceeds of $105,000. Each unit consists of one common share and one share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.13 per share for a period of one year, subject to forced acceleration in the event the Company's shares close at a price of $0.20 or higher per share on the Canadian National Stock Exchange (or such other stock exchange as the majority of the trading volume of the Company's shares occur) for 10 consecutive trading days. $92,172 of the proceeds was allocated to share capital and $12,828 of the proceeds was allocated to warrant reserve. Share issuance costs of $4,400 were paid for this private placement.
On November 26, 2012, the Company issued 2,500,000 common shares with a fair value of $150,000 and on December 27, 2012, the Company issued 3,500,000 common shares with a fair value of $350,000 as consideration to acquire the exclusive, worldwide rights to a patent license from Vertical Designs Ltd. (Note 5)
On January 14, 2013, the Company issued 200,000 common shares with a fair value of $36,000 to settle a trade payable of $36,000.
On January 16, 2013, the Company issued 200,000 common shares to consultants of the Company with aggregate fair value of $25,000 for consulting services.
On January 16, 2013, the Company issued 213,333 common shares with a fair value of $57,600 to settle a trade payable of $40,000.
On February 6, 2013, the Company issued 122,500 common shares with a fair value of $25,725 to the CEO of the Company to settle a trade payable of $18,988.
On March 13, 2013, 100,000 share purchase options were exercised at an exercise price of $0.10 per share purchase option for total proceeds of $10,000.
On March 18, 2013, the Company issued 133,333 common shares with a fair value of $25,333 to settle a trade payable of $20,434.
On March 28, 2013, the Company issued 400,000 common shares, with a fair value of $78,000 to acquire a portfolio of natural health product formulae focusing on pain management, immunity and inflammation and cognitive function. (Note 5)
200
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 8. | SHARE CAPITAL (continued) |
(a) Common shares (continued)
On April 15, 2013, the Company issued 625,000 common shares with a fair value of $93,750 to settle a trade payable of $50,000.
(b) Share purchase options
The Company has a share purchase option plan (dated June 18, 2012) which specifies that a maximum of 10% of the issued and outstanding common shares of the Company may be reserved for issuance pursuant to the exercise of share options. The term of the share options granted are fixed by the board of directors and are not to exceed ten years. The exercise prices of the share options shall not be less than the closing price of the Company's common shares on the day preceding the day on which the directors grant the share purchase options, less any discount permitted by the Exchange. Vesting of options will be at the discretion of the Board.
A continuity schedule of the Company's outstanding options for the year ended September 30, 2013 and the year ended September 30, 2012 is as follows:
| Number outstanding | Weighted average exercise price | ||
| Balance at September 30, 2011 | 860,000 | $ | 0.33 |
Granted | 30,000 | 0.38 | |
| Balance at September 30, 2012 | 890,000 | $ | 0.33 |
Granted | 2,290,000 | 0.11 | |
Exercised | (100,000) | 0.10 | |
Cancelled | (150,000) | 0.10 | |
Expired | (355,000) | 0.21 | |
| Balance at September 30, 2013 | 2,575,000 | $ | 0.17 |
As of September 30, 2013, the Company had options outstanding and exercisable to acquire common shares of the Company as follows:
| Expiry date | Number of options outstanding | Number of options exercisable | Exercise price $ | Weighted average remaining contractual life (in years) |
| May 12, 2016 | 660,000 | 660,000 | 0.35 | 2.62 |
| December 24, 2017 | 1,240,000 | 1,240,000 | 0.10 | 4.24 |
| December 28, 2017 | 350,000 | 350,000 | 0.10 | 4.25 |
| January 31, 2018 | 125,000 | 125,000 | 0.21 | 4.34 |
| March 28, 2018 | 75,000 | 75,000 | 0.17 | 4.49 |
| May 23, 2018 | 50,000 | 50,000 | 0.10 | 4.65 |
| May 24, 2018 | 75,000 | 75,000 | 0.10 | 4.65 |
| 2,575,000 | 2,575,000 | 3.85 |
(c) Share-based compensation
The estimated fair value of options granted to executive officers, directors, and consultants is recognized over the vesting period of the options. During the year ended September 30, 2013, $85,033 (2012 - $8,542) share-based compensation expense was recorded. The weighted average fair value of share options granted during the year ended September 30, 2013 was $0.04 per option (2012 - $0.28).
201
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 8. | SHARE CAPITAL (continued) |
(c) Share-based compensation (continued)
The estimated fair value of share options granted was determined using the Black-Scholes option pricing model using the following weighted average assumptions:
| 2013 | 2012 | |
| Annualized volatility | 102.38% | 118.18% |
| Risk-free interest rate | 1.14% | 1.35% |
| Expected life | 3.1 years | 5 years |
| Dividend yield | nil% | nil% |
| Share price | $0.07 | $0.35 |
| Exercise price | $0.11 | $0.38 |
Option pricing models require the input of highly subjective assumptions, including the expected price volatility. Changes in the subjective input assumptions can materially affect the fair value estimate, and therefore the existing assumptions do not necessarily provide a reliable measure of the future fair value of the Company's share purchase options.
(d) Warrants
A continuity schedule of the Company's outstanding warrants for the year ended September 30, 2013 and year ended September 30, 2012 is as follows:
Number outstanding | Weighted average exercise price | ||
| Balance at September 30, 2011 | 2,500,000 | $ | 0.25 |
| Granted | 1,200,000 | 0.35 | |
| Exercised | (30,000) | 0.25 | |
| Balance at September 30, 2012 | 3,670,000 | $ | 0.28 |
| Granted | 2,100,000 | 0.13 | |
| Expired | (1,200,000) | 0.35 | |
| Balance at September 30, 2013 | 4,570,000 | $ | 0.20 |
As of September 30, 2013, the Company had warrants outstanding as follows:
Expiry date | Number of common shares issuable upon exercise |
Exercise price $ | Weighted average remaining contractual life (in years) |
| October 7, 2015 | 2,470,000 | 0.25 | 2.02 |
| December 12, 2013 (Note 16) | 2,100,000 | 0.13 | 0.20 |
| 4,570,000 | 1.18 |
202
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 9. | INCOME TAXES |
A reconciliation of income taxes at statutory rates with taxes reported in these financial statements:
| 2013 | 2012 | |||
| Net loss for the year | $ | (1,102,491) | $ | (992,389) |
| Canadian statutory income tax rates | 25.80 % | 25.00 % | ||
| Expected recovery of income taxes based on Canadian statutory tax rate | (284,443) | (248,097) | ||
Increase (decrease) in income tax recovery resulting from: | ||||
Share based compensation | 10,600 | 27,135 | ||
Net tax effect of other items not taxable or deductible for income tax purposes |
| 89,105 |
| 42,906 |
Tax losses acquired on the acquisition of Northern Vine Canada Inc. | - | (47,454) | ||
| Effect of income tax rate change | (57,522) | (6,794) | ||
| Increase in valuation allowance | 242,260 | 232,304 | ||
| Total income tax recovery | - | - | ||
| Deferred tax assets and liabilities are: | ||||
| Deferred tax assets (liabilities) | 2013 | 2012 | ||
Property and equipment | 26,282 | 20,351 | ||
Intangible assets | (10,266) | (39,321) | ||
Share issuance costs | 6,908 | 7,826 | ||
Non-capital tax losses carried forward | 773,025 | 564,833 | ||
| Valuation allowance | (795,949) | (553,689) | ||
| Net deferred tax assets | - | - |
At September 30, 2013, the Company has accumulated non-capital losses for income tax purposes of approximately $2,974,000. If unused, the non-capital losses will expire as follows:
| Year of expiration | Non-capital loss | |
| 2029 | $ | 66,000 |
| 2030 | 634,000 | |
| 2031 | 704,000 | |
| 2032 | 855,000 | |
| 2033 | 715,000 | |
| $ | 2,974,000 |
203
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 10. | ADDITIONAL FINANCIAL INFORMATION |
(a) Statement of financial position
September 30, 2013 | September 30, 2012 | |
| Trade and other receivables | ||
HST/GST receivable | 26,763 | 13,465 |
| 26,763 | 13,465 | |
| Trade and other payables | ||
Trade payables | 238,956 | 329,146 |
Accrued liabilities | 179,472 | 110,125 |
Due to related parties (Note 12) | 306,363 | 80,058 |
Payroll liabilities | 2,819 | 2,073 |
| 727,610 | 521,402 |
The Company reversed an account payable of $205,000 for research fees and recorded this reversal as a gain on cancellation of trade payables in the statement of loss and comprehensive loss for the year ended September 30, 2013.
(b) Supplemental cash flow information
| 2013 | 2012 | |
| Shares cancelled | - | (205,000) |
| Shares issued for acquisition of assets | 578,000 | 400,000 |
| Shares issued in business combination | - | 30,000 |
| Shares issued for prepaid consulting fees | 55,000 | 50,000 |
| Shares issued for settlement of trade payable | 238,408 | 131,326 |
| Transfer from warrants reserve to share capital on exercise of warrants | - | (6,062) |
| Transfer to warrants reserve fair value of warrants issued on private placement | 12,828 | 94,514 |
| Transfer from equity settled share based payments reserve to share capital on exercise of options | (1,651) | - |
| 11. | FINANCIAL INSTRUMENTS |
The Company's financial assets and financial liabilities are categorized as follows:
September 30, 2013 | September 30, 2012 | |||
Financial Assets | ||||
Held for trading: | ||||
| Cash and cash equivalents | $ | 5,327 | $ | 5,499 |
Loans and receivable: | ||||
| Trade and other receivables | $ | 26,763 | $ | 13,465 |
Financial Liabilities | ||||
Other financial liabilities: | ||||
| Trade and other payables | $ | 727,610 | $ | 521,402 |
| Interest payable | $ | 34,689 | $ | 22,506 |
| Short-term loans | $ | 290,159 | $ | 157,650 |
204
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 11. | FINANCIAL INSTRUMENTS (continued) |
The fair values of the Company's financial assets and financial liabilities approximate their carry values due to the short-term nature of these instruments.
The Company's financial instruments are exposed to certain risks.
(a) Credit risk
Credit risk is the risk that one party to a financial instrument will fail to discharge an obligation and cause the other party to incur a financial loss. The Company is exposed to credit risk on its cash and cash equivalents and trades and other receivables.
The Company's maximum credit risk exposure is as follows:
| September 30, 2013 | September 30, 2012 | |||
| Cash and cash equivalents | $ | 5,327 | $ | 5,499 |
| Trade and other receivables | 26,763 | 13,465 | ||
| $ | 32,090 | $ | 18,964 |
The Company deposits the majority of its cash with high credit quality financial institutions in Canada and the Company's trade and other receivables consists of refundable taxes due from the Federal Government of Canada. Therefore, management considers the risk of non-performance related to cash and cash equivalents and trade and other receivables to be minimal.
(b) Liquidity risk
Liquidity risk is the risk that the Company will not be able to meet its financial obligations as they come due. To the extent that the Company does not believe it has sufficient liquidity to meet its current obligations, the board of directors considers securing additional funds through equity, debt or partnering transactions. The board of directors approves the Company's annual operation and capital budgets as well as any material transactions outside the ordinary course of business.
At September 30, 2013, the Company has current liabilities of $1,071,329, which are due within the operating period, (September 30, 2012 - $701,558), and cash of $5,327 (September 30, 2012 - $5,499).
The Company will have to obtain additional funding from loans or equity financings to have sufficient capital in order to meet short term business requirements, after taking into account its limited cash flows from operations and the Company's low cash balances.
(c) Market risk
Market risk consists of currency risk, interest rate risk, and other price risk. These are discussed further below.
Currency risk
The Company is exposed to financial risk related to fluctuations in foreign exchange rates. Foreign currency risk is limited to the portion of the Company's business transactions denominated in currencies other than the Canadian Dollar.
205
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 11. | FINANCIAL INSTRUMENTS (continued) |
(c) Market risk (continued)
A portion of the Company's cash and trade and other payables are denominated in US Dollars as follows.
| September 30, 2013 | September 30, 2012 | |||
| USD | USD | |||
| Cash | $ | - | $ | 120 |
| Trade and other payables | 178,160 | 66,830 | ||
| Net exposure | $ | 178,160 | $ | 66,710 |
| Canadian dollar equivalent | $ | 183,238 | $ | 65,556 |
A 4.66% (September 30, 2012 - 5.41%) weakening or strengthening in the Canadian / US exchange rate (based on prior year fluctuations in the relative exchange rate) would result in an estimated increase or decrease of approximately $8,302 in the Company's operations and comprehensive loss for the year ended September 30, 2013 (September 30, 2012 - $3,609).
The Company has not entered into any foreign currency contracts to mitigate this risk, but manages the risk by minimizing the value of financial instruments denominated in foreign currency.
Interest rate risk
Interest rate risk consists of two components:
i) To the extent that payments made or received on the Company's monetary assets and liabilities are affected by changes in the prevailing market interest rates, the Company is exposed to interest rate cash flow risk.
ii) To the extent that changes in prevailing market rates differ from the interest rate in the Company's monetary assets and liabilities, the Company is exposed to interest rate price risk.
Current financial assets and financial liabilities are generally not exposed to interest rate risk because of their short term to maturity.
| 12. | RELATED PARTY TRANSACTIONS |
(a) Management transactions
Management transactions with related parties during the years ended September 30, 2013 and September 30, 2012 were as follows:
| September 30, 2013 | September 30, 2012 | |||||
| Short-term employee benefits | Share-based compensation | Total | Short-term employee benefits | Share-based compensation | Total | |
| Mike Withrow (i) | $ 142,500 | $ 14,549 | $ 157,049 | $ 120,000 | $ - | $ 120,000 |
206
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 12. | RELATED PARTY TRANSACTIONS (continued) |
(a) Management transactions (continued)
(i) Mike Withrow is the President and CEO and director of the Company. During the year ended September 30, 2013, 390,000 (September 30, 2012 - nil) share purchase options were granted by the Company to Mr. Withrow, with an estimated fair value of $14,549 (September 30, 2012 - $nil). During the year ended September 30, 2013, 747,500 (September 30, 2012 - 240,000) common shares, with an estimated fair value of $119,475 (September 30, 2012 - $61,000), were issued by the Company to Mr. Withrow to settle a trade payable of $68,988 (September 30, 2012 - $74,000). At September 30, 2013, $142,344 due to Mr. Withrow was included in trade and other payables (September 30, 2012 - $63,711), operating cash of $260,159 (September 30, 2012 - $157,650) provided by Mr. Withrow and $30,000 in common shares due from the Company were included in short-term loans and accrued interest of $14,468 (September 30, 2012 - $2,286) was included in interest payable.
Transactions with related parties have been in the normal course of operations and, in management's opinion, undertaken on the same terms and conditions as transactions with unrelated parties. These costs are measured at exchange amounts agreed upon by the parties.
(b) Directors' transactions
| September 30, 2013 | September 30, 2012 | |||||
| Consulting fees | Share-based compensation | Total | Consulting fees | Share-based compensation | Total | |
| Terence Fealey (i) | $ 97,816 | $ 2,798 | $ 100,614 | $ 56,000 | $ 50,000 | $ 106,000 |
(i) Mr. Fealey is a director of the Company. During the year ended September 30, 2013, 75,000 share purchase options (September 30, 2012 - nil) were granted by the Company to Mr. Fealey, with an estimated fair value of $2,798 (September 30, 2012 - $nil). During the year ended September 30, 2013, no bonus shares were issued to Mr. Fealey (September 30, 2012 - 400,000 bonus shares with a fair value of $50,000). At September 30, 2013, $118,302 due to Mr. Fealey was included in trade and other payables (September 30, 2012 - $16,073).
(ii) During the year ended September 30, 2013, 480,000 share purchase options (September 30, 2012 - nil) were granted to other directors of the Company with a fair value of $22,169 (September 30, 2012 - $nil). During the year ended September 30, 2013, no bonus shares (September 30, 2012 - 180,000 bonus shares with a fair value of $22,500) were issued to other directors of the Company.
(iii) During the year ended September 30, 2013, a law firm, of which a director is a one-third partner, provided legal services totaling $33,150 (2012 - $38,962) to the Company. At September 30, 2013, $45,717 (2012 - $31,262) due to this law firm was included in trade and other payables.
207
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 13. | COMMITMENTS |
On January 10, 2011, the Company entered into a consulting agreement with the CEO of the Company for his services as CEO. Under the agreement, the Company will pay consulting fees of $10,000 (excluding HST) per month. The contract expires on January 10, 2014. If the contract is terminated at the Company's discretion, the CEO is entitled to receive three months' fees over and above the thirty day notice period. This agreement was amended in May 2013 and pursuant to the amended agreement, the CEO's consulting fees were increased to $12,500 (excluding HST) per month, retroactive to January 1, 2013, and the term of his contract extended to January 10, 2016.
On March 1, 2012, the Company entered into a 3 year consulting agreement with one of the directors of the Company. Under the agreement, the Company will pay US $8,000 per month to this director for consulting and research and development services. The contract expires on March 1, 2015 and if the contract is terminated at the Company's discretion, the director is entitled to receive three months' fees over and above the thirty day notice period.
On April 20, 2012, the Company entered into an exclusive distribution agreement with Hedley Enterprises Ltd. ("Hedley") to purchase, resell and distribute Abattis' line of natural products in Canada. Under the terms of the Agreement Hedley has acquired the exclusive right to sell and distribute Abattis' products to all retail distribution channels, which include health food stores, grocery stores, fitness facilities, and similar retail establishments.
On May 21, 2012, the Company entered into a two year finder's fee agreement with VentureCorp 2 Capital Inc. Under the agreement, the Company will pay to the finder a cash fee equal to 10% of the gross proceeds raised by the Company through the sale of shares or units to investors introduced by the finder. The Company will also issue to the finder non-transferable compensation warrants in an amount equal to 5% of the aggregate number of shares or units subscribed for by the investors introduced by the finder. Each finder's warrant will entitle the finder to acquire one common share of the Company, exercisable for a period of 12 months from closing date at an exercise price equal to the price per share or unit.
On November 1, 2012, the Company renewed a three year office lease with Toro Holdings Ltd. The Company's minimum annual lease commitments over the next two years are as follows:
2014 - $22,541
2015 - $27,772
On December 27, 2012, the Company entered into a license agreement with Vertical Designs Ltd. ("Vertical Designs") Under the agreement, the Company has been granted the exclusive, worldwide rights to a patent license, with the right to grant sublicenses, to use the Bio Pharma technology for growing products at licensed facilities, which products may only be used as ingredients in the pharmaceutical, nutraceutical, cosmetic and wellness markets. The royalty provisions of the license agreement reflect that: (i) the royalty payable on net sales of all products sold by Abattis was 4%; (ii) in consideration for the grant of the Company's right to grant sublicenses, the Company will pay to Vertical Designs Ltd. a sublicense royalty of 15% of any monies or other consideration that the Company receives from any sublicense; and (iii) after two years, the Company will be required to pay to Vertical Designs Ltd. a minimum royalty payment of $25,000 per year and if the combined royalty payments paid from (i) and (ii) above do not equal $25,000 in any given year then the Company will be permitted to top up such amount with a cash payment. Under the terms of the agreement, the patent license will revert to Vertical Designs Ltd. in certain circumstances, including: (i) if the Company terminates the agreement; (ii) if the Company materially breaches or defaults in the performance of the agreement and has not cured such default within 60 days, or in the case of failure to pay any amounts due, then within 30 days, after receiving written notice from Vertical Designs Ltd. specifying the breach; (iii) if the Company discontinues its business of producing ingredients for pharmaceutical, nutraceutical, cosmetic or wellness markets; (iv) if the Company fails to pay the annual $25,000 minimum royalty payment for any year ending after the second anniversary of the agreement; or (v) if the Company becomes insolvent, makes an assignment for the benefit of creditors or has a petition of bankruptcy filed by or against it, which petition is not vacated or otherwise removed within 90 days after the filing thereof. The Company also agreed to pay Vertical Designs $250,000
208
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 13. | COMMITMENTS (continued) |
for the purchase and sale of six complete Vertical Designs operational units. The purchase price will be paid in instalments, dates and amounts are to be determined between the parties, with the first payment due on or before the earlier of five business days following the Company completing an equity and/or debt financing of any amount or the first business day in the seventh month following the date of the Bill of Sale.
On January 6, 2013, the Company entered into a two year consulting agreement with Georges Laraque Management Inc. Under the agreement, the Company will pay consulting fees of $5,000 per month.
On February 1, 2013, the Company entered into a one year consulting agreement with Triangle Consultants Inc. ("Triangle"). Under the agreement, the Company will pay consulting fees of $8,000 per month to Triangle for consulting and research services.
On August 6, 2013, the Company entered into a six month investor relation services agreement with Carmel Advisors LLC. Under the agreement, the Company will grant an aggregate of 650,000 share purchase options, a bonus of 8% (in USD) and 500,000 bonus share purchase options upon the Company's successful completion of a private placement.
| 14. | CONTINGENT LIABILITIES |
On September 20, 2012, a claim, which is based on a contract dated June 29, 2009 between the Company and the plaintiff, was filed against the Company. The plaintiff and the Company entered into an agreement dated May 16, 2011 to settle a dispute between the two parties over the contract dated June 29, 2009. The Company made an initial payment of $5,000 to the plaintiff, as per the agreement dated May 16, 2011. However, the plaintiff did not transfer the payment to an individual named in the agreement nor did the plaintiff instruct this individual appropriately. As such, the Company refused to make any further payments under this agreement until those events have taken place. The plaintiff claims that the agreement of May 16, 2011 is not binding and is seeking payment of $145,000. The outcome of this claim is not determinable and therefore no amount has been recorded for any potential payments which may have to be made.
During the year, four claims were filed against the Company, one of which was settled subsequent to year end and an accrual of $7,000 has been recorded for the settlement amount. The other three claims are outstanding as at September 30, 2013 and the plaintiffs are claiming an aggregate of approximately $421,000. The outcomes of these claims are not determinable and therefore no amounts have been recorded for any potential payments which may have to be made.
| 15. | MANAGEMENT OF CAPITAL |
The Company's objectives when managing capital are to safeguard its ability to continue as a going concern in order to pursue the development of its technologies and to maintain a flexible capital structure, which optimizes the costs of capital at an acceptable risk. The Company considers its capital for this purpose to be its shareholders' equity.
The Company's primary source of capital is through the issuance of equity. The Company manages and adjusts its capital structure when changes in economic conditions occur. To maintain or adjust the capital structure, the Company may seek additional funding. The Company may require additional capital resources to meet its administrative overhead expenses in the long term. The Company believes it will be able to raise capital as required in the long term, but recognizes there will be risks involved that may be beyond its control. There are no external restrictions on the management of capital.
209
Abattis Bioceuticals Corp.
Notes to the Consolidated Financial Statements
September 30, 2013 and 2012
(Expressed in Canadian Dollars)
| 16. | EVENTS AFTER THE REPORTING DATE |
The following events occurred subsequent to September 30, 2013:
| 150,000 options were cancelled following the resignation of a director. |
The expiry date of 2,000,000 share purchase warrants previously issued on December 12, 2012 has been extended from December 12, 2013 to December 12, 2014. The warrants are exercisable at an exercise price of $0.13 per common share, subject to an acceleration provision in the event that the Company's shares close on the Exchange for 10 consecutive trading days at $0.20 per share or higher. |
| On December 6, 2013, the Company issued 3,500,000 common shares to an officer to settle $105,000 in outstanding debt owed to the officer. |
On December 18, 2013 the Company through its wholly owned subsidiary Animo Wellness Corporation, doing business as Medical Marijuana Labs ("MMLC") signed a five year lease with PurGenesis Technologies, Inc. for the lease of approximately 5,000 square feet of lab and production space at a cost of $24 per square foot or $120,000 in annual gross rent. |
On January 16, 2014, the Company announced a non-brokered private placement of up to 12,000,000 units of the Company at a price of $0.05 per unit for gross proceeds of up to $600,000. Each share unit will be comprised of one common share and one share purchase warrant, exercisable at $0.10 per share. |
| The Company negotiated debt settlement arrangements with two officers of the Company for settlement of debt totaling $106,250 with 2,125,000 common shares of the Company. |
| 250,000 share purchase options, exercisable at $0.10 per share for five years, were granted to an officer of the Company. |
210
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
OTHER EXPENSES OF ISSUANCE AND DISTRIBUTION
The following table sets forth costs and expenses payable by the Company in connection with the issuance and distribution of securities being registered.
| Fees | Amount | ||
| SEC Filing Fees | $ | 3,000 | |
| Blue Sky Fees and Expenses | $ | 10,000 | |
| Legal Fees and Expenses | $ | 70,000 | |
| Accounting Fees and Expenses | $ | 30,000 | |
| Document Preparation Fee | $ | 15,000 | |
| Miscellaneous | $ | 15,000 | |
| Total | $ | 143,000 | |
INDEMNIFICATION OF DIRECTORS AND OFFICERS
The corporate laws of British Columbia allow us, and our corporate articles require us (subject to the provisions of the British Columbia Business Corporations Act ("BCBCA") ( noted below), to indemnify our Directors, former Directors, alternate Directors and their heirs and legal personal representatives against all eligible penalties to which such person is or may be liable, and the Company must, after the final disposition of an eligible proceeding, pay the expenses actually and reasonably incurred by such person in respect of that proceeding. Each Director and alternate Director is deemed to have contracted with the Company on the terms of the indemnity contained in our articles.
For the purposes of such an indemnification:
"eligible party", in relation to the Company, means an individual who
| (1) | is or was a Director or officer of the Company, |
| (2) | is or was a director or officer of another corporation |
(i) at a time when the corporation is or was an affiliate of the Company, or
(ii) at the request of the Company, or
| (3) | at the request of the Company, is or was, or holds or held a position equivalent to that of, a director or officer of a partnership, trust, joint venture or other unincorporated entity, |
and includes, except in the definition of "eligible proceeding" and certain other cases, the heirs and personal or other legal representatives of that individual;
211
"eligible penalty" means a judgment, penalty or fine awarded or imposed in, or an amount paid in settlement of, an eligible proceeding;
"eligible proceeding" means a proceeding in which an eligible party or any of the heirs and personal or other legal representatives of the eligible party, by reason of the eligible party being or having been a director or officer of, or holding or having held a position equivalent to that of a director or officer of, the Company or an associated corporation:
| (1) | is or may be joined as a party, or |
| (2) | is or may be liable for or in respect of a judgment, penalty or fine in, or expenses related to, the proceeding; |
"expenses" includes costs, charges and expenses, including legal and other fees, but does not include judgments, penalties, fines or amounts paid in settlement of a proceeding; and
"proceeding" includes any legal proceeding or investigative action, whether current, threatened, pending or completed.
In addition, under the BCBCA, the Company may pay, as they are incurred in advance of the final disposition of an eligible proceeding, the expenses actually and reasonably incurred by an eligible party in respect of that proceeding, provided that the Company first receives from the eligible party a written undertaking that, if it is ultimately determined that the payment of expenses is prohibited by the restrictions noted below, the eligible party will repay the amounts advanced.
Notwithstanding the provisions of the Company's articles noted above, the Company must not indemnify an eligible party or pay the expenses of an eligible party, if any of the following circumstances apply:
| (1) | if the indemnity or payment is made under an earlier agreement to indemnify or pay expenses and, at the time that the agreement to indemnify or pay expenses was made, the company was prohibited from giving the indemnity or paying the expenses by its memorandum or articles; |
| (2) | if the indemnity or payment is made otherwise than under an earlier agreement to indemnify or pay expenses and, at the time that the indemnity or payment is made, the company is prohibited from giving the indemnity or paying the expenses by its memorandum or articles; |
| (3) | if, in relation to the subject matter of the eligible proceeding, the eligible party did not act honestly and in good faith with a view to the best interests of the company or the associated corporation, as the case may be; |
| (4) | in the case of an eligible proceeding other than a civil proceeding, if the eligible party did not have reasonable grounds for believing that the eligible party's conduct in respect of which the proceeding was brought was lawful. |
In addition, if an eligible proceeding is brought against an eligible party by or on behalf of the Company or by or on behalf of an associated corporation, the Company must not do either of the following:
| (1) | indemnify the eligible party under section 160 (a) in respect of the proceeding; or |
| (2) | pay the expenses of the eligible party in respect of the proceeding. |
212
Notwithstanding any of the foregoing, and whether or not payment of expenses or indemnification has been sought, authorized or declined under the BCBCA or the articles of the Company, on the application of the Company or an eligible party, the Supreme Court of British Columbia may do one or more of the following:
| (1) | order a company to indemnify an eligible party against any liability incurred by the eligible party in respect of an eligible proceeding; |
| (2) | order a company to pay some or all of the expenses incurred by an eligible party in respect of an eligible proceeding; |
| (3) | order the enforcement of, or any payment under, an agreement of indemnification entered into by a company; |
| (4) | order a company to pay some or all of the expenses actually and reasonably incurred by any person in obtaining an order under this section; |
| (5) | make any other order the court considers appropriate. |
RECENT SALES OF UNREGISTERED SECURITIES
The following information is furnished with regard to all securities issued by the Company within the last three years that were not registered under the Securities Act. The issuance of such shares was deemed exempt from registration requirements of the Securities Act as such securities were offered and sold outside of the United States to persons who were neither citizens nor residents of the United States or such sales were exempt from registration under Section 4(a)(2) of Securities Act.
On April 14, 2015, 86,436 common shares were issued to certain consultants of the Company. | ||
On April 14, 2015, 72,414 common shares were issued to Allan Echino to settle a debt. | ||
On March 19, 2015, 2,365,072 units were issued at a price of $0.13 per unit for gross proceeds of $307,459. Each unit consists of one common share and one share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.18 per share for a period of 18 months, subject to acceleration terms. | ||
On March 6, 2015, 125,000 stock options were granted to certain directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. | ||
On March 4, 2015, 539,498 common shares were issued to certain directors, officers and consultants of the Company. | ||
On February 11, 2015, 50,000 warrants were exercised at a price of $0.10 per share for gross proceeds of $5,000. | ||
On February 11, 2015, 250,000 warrants were exercised at a price of $0.10 per share for gross proceeds of $25,000. | ||
On February 3, 2015, 55,926 common shares were issued to certain consultants of the Company. | ||
On January 27, 2015, 175,000 stock options were granted to certain directors, officers and consultants, with each option being exercisable into a common share of the Company at $0.16 per share for a period of five years. |
213
| On January 2, 2015, New Horizons Inc. exercised 68,000 warrants at $0.10 per share for gross proceeds of $6,800. |
| On January 2, 2015, 283,334 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On January 2, 2015, 18,382 common shares were issued to Jaouad Fichatli for consulting services to the Company. |
| On December 3, 2014, New Horizons Inc. exercised 40,000 warrants at $0.10 per share for gross proceeds of $4,000. |
| On December 1, 2014, 33,334 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On December 1, 2014, 14,535 common shares were issued to Jaouad Fichatli for consulting services to the Company. |
| On December 1, 2014, 40,000 common shares were issued to ThinkSharp Inc., a consulting firm. |
| On November 27, 2014, New Horizons Inc. exercised 42,000 warrants at $0.10 per share for gross proceeds of $4,200. |
| On November 20, 2014, Michael Sweeney exercised 75,000 warrants at $0.10 per share for gross proceeds of $7,500. |
| On November 18, 2014, Michael Sweeney exercised 25,000 warrants at $0.10 per share for gross proceeds of $2,500. |
On November 14, 2014, New Horizons Inc. exercised 150,000 warrants at $0.10 per share for gross proceeds of $15,000. | ||
| On November 4, 2014, Stone Throw exercised 500,000 warrants at $0.10 per share for gross proceeds of $50,000. |
| On November 3, 2014, 33,334 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On September 17, 2014, 50,000 common shares with a deemed value of $16,500 was issued to Guy P. Dancosse, a director of the Company. |
| On September 16, 2014, 100,000 incentive stock options were granted to certain directors, with each option being exercisable into a common share of the Company at $0.33 per share for a period of five years. |
| On September 12, 2014, 100,000 common shares with a deemed value of $10,000 was issued to Henry Der Professional Corporation for consulting services to the Company. |
| On September 3, 2014, 33,333 common shares were issued to Brazos Minshew for consulting services to the Company. |
| On September 3, 2014, 55,964 common shares were issued to Emerging Growth Ltd. (TDM Financial) for investor relations services provided to the Company. |
| On August 13, 2014, 455,000 stock options were granted to certain of its directors, officers, and consultants, with each option being exercisable into a common share of the Company at $0.43 per share for a period of five years. |
| On August 6, 2014, 243,460 common shares with a deemed value of $111,991 were issued to Kodiak pursuant to the Equity Purchase Agreement. |
| On August 1, 2014, 50,416 common shares were issued to Emerging Growth Ltd. (TDM Financial) for investor relations services provided to the Company. |
| On August 1, 2014, 33,333 common shares with a deemed value of $14,000 was issued to Brazos Minshew for consulting services to the Company. |
| On August 1, 2014, 24,000 common shares with a deemed value of $12,240 was issued to ThinkSharp Inc. |
214
| On August 1, 2014, the CFO for gross proceeds of $25,000 exercised 250,000 stock options at $0.10 per share. |
| On August 1, 2014, the CFO for gross proceeds of $51,000 exercised 300,000 stock options at $0.17 per share. |
| On July 28, 2014, 100,000 options were exercised at $0.10 per share for gross proceeds of $10,000. |
| On July 22, 2014, 2,438,100 stock options were granted to certain of its directors, officers, and consultants, with each option being exercisable into a common share of the Company at $0.64 per share for a period of five years. |
| On July 21, 2014, 25,000 warrants were exercised for proceeds of $12,500. |
| On July 16, 2014, 50,000 warrants were exercised for proceeds of $5,000. |
| On July 16, 2014, 100,000 warrants were exercised for proceeds of $10,000. |
| On July 16, 2014, 200,000 options were exercised at $0.36 per share for gross proceeds of $72,000. |
| On July 15, 2014, 143,550 warrants were exercised for proceeds of $35,888. |
| On July 15, 2014, 100,000 options were exercised at $0.115 per share for gross proceeds of $11,500. |
| On July 15, 2014, 110,000 options were exercised at $0.26 per share for gross proceeds of $28,600. |
| On July 10, 2014, 100,000 warrants were exercised for proceeds of $10,000. |
| On July 3, 2014, 50,000 warrants were exercised for proceeds of $5,000. |
| On July 2, 2014, 46,824 common shares were issued to Emerging Growth Ltd. (TDM Financial) for investor relations services provided to the Company. |
| On June 19, 2014, 100,000 common shares with a deemed value of $38,000 were issued to two directors for the consulting services provided. |
| On June 2, 2014, the Company issued 35,665 common shares with a deemed value of US$24,609 to Emerging Growth Ltd. (TDM Financial). |
| On April 30, 2014, the Company through its wholly owned Washington subsidiary Abattis Bioceuticals International Inc. acquired 34% interest in Instant Payment Systems LLC (IPS), in consideration for 200,000 common shares at a deemed value of $180,000. |
| On April 30, 2014, 33,669 common shares with a deemed value of $33,333 were issued to two service providers for services provided to the Company. |
| On April 28, 2014, 40,000 warrants were exercised for proceeds of $10,000. |
| On April 7, 2014, 827,657 common shares with a deemed value of $579,360 were issued to Phytalytics LLC to acquire a 51% membership interest. |
| On April 1, 2014, 43,440 common shares were issued to a law firm to settle a trade payable of US $62,554. |
On March 17, 2014, 5,333,331 units at a price of $0.45 per unit for gross proceeds of $2,399,999 was issued to arm's length parties. Each unit consists of one common share and one share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.50 per share for a period of 18 months. |
| On March 12, 2014, the CEO of the Company for proceeds of $124,000 exercised 890,000 stock options. | ||
| On March 7, 2014, 315,000 common shares with a deemed value of $252,000 was issued to Green Gro Garden Products Ltd. |
215
| On February 26, 2014, 110,000 incentive stock options was granted to a consultant of the Company, with each option being exercisable into a common share of the Company at $0.26 per share for a period of five years. | ||
| On February 24, 2014, the CEO of the Company exercised 1,000,000 warrants for proceeds of $130,000. | ||
| On February 21, 2014, 110,000 common shares with a deemed value of $18,700 was issued to Jiangsu Jiahui New Material Co Ltd. | ||
| On February 18, 2014, 1,075,000 incentive stock options were granted to certain directors, officers and consultants of the Company, with each option exercisable into a common share of the Company at $0.17 per share for a period of five years. | ||
On February 11, 2014, 12,000,000 units at a price of $0.05 per unit for gross proceeds of $600,000 was issued to arm's length parties. Each unit consists of one common share and one-half share purchase warrant of the Company. Each share purchase warrant will entitle the holder to purchase one additional common share at a price of $0.10 per share for a period of one year. | ||
On January 29, 2014, 700,000 incentive stock options were granted to certain of its directors and consultants, with each option exercisable into a common share of the Company at $0.10 per share for a period of five years. On January 30, 2014, the Company amended the exercise price of the 700,000 five-year incentive stock options to $0.115 per share. | ||
| On January 28, 2014, 1,500,000 common shares were issued with a deemed value of $210,000 to settle a trade payable of $75,000 with the CEO of the Company. |
| On January 16, 2014, 625,000 common shares were issued with a deemed value of $31,250 to settle a trade payable of $31,250 with the CFO of the Company. |
| On December 6, 2013, 3,500,000 common shares were issued with a deemed value of $87,500 to settle a trade payable of $105,000 with the CEO of the Company. |
On August 6, 2013, the Company entered into a six month investor relation services agreement with Carmel Advisors LLC. Under the agreement, the Company will grant an aggregate of 650,000 share purchase options, a bonus of 8% (in USD) and 500,000 bonus share purchase options upon the Company's successful completion of a private placement. |
| On May 24, 2013, 50,000 incentive stock options was granted to one of its consultants, with each option being exercisable into a common share at $0.10 per share for a period of five years. |
| On May 23, 2013, 75,000 incentive stock options was granted to certain consultants, with each option being exercisable into a common share at $0.10 per share for a period of five years. |
| On April 15, 2013, 625,000 common shares were issued at a deemed value of $93,750 to settle a trade payable of $50,000. |
| On March 28, 2013, 400,000 common shares were issued with a deemed value of $78,000 to Dr. Paula Brown. |
| On March 18, 2013, 133,333 common shares were issued with a deemed value of $25,333 to settle a debt payable of $20,434 to a consulting company. |
| On March 13, 2013, 100,000 share purchase options were exercised at $0.10 per share purchase option for proceeds of $10,000. |
| On February 6, 2013, 122,500 common shares were issued with a deemed value of $25,725 to settle a debt payable of $18,988 to an officer of the Company. |
| On January 31, 2013, 125,000 share purchase options were granted to certain directors, officers and consultants, exercisable at $0.10 per share for a period of five years. |
| On January 16, 2013, 213,333 common shares were issued with a deemed value of $57,600 to settle a debt payable of $40,000 to a consultant of the Company. |
216
| On January 16, 2013, 200,000 common shares were issued with a deemed value of $25,000 to consultants of the Company. |
| On January 14, 2013, 200,000 common shares were issued with a deemed value of $36,000 to Bacchus Law Corporation for services provided in January 2013. |
| On January 1, 2013, the Company entered into an agreement with Georges Laraque Management Inc. and as partial consideration 150,000 common shares, with a deemed price of $0.10, of the Company were issued on January 16, 2013. |
| On January 1, 2013, 100,000 incentive share purchase options were granted to Cors Group Corporate Solutions, with each option exercisable at a price of $0.10 per share for a period of five years. |
| On December 28, 2012, 350,000 incentive stock options were granted to certain directors and consultants, with each option exercisable at $0.10 per share for a period of five years. |
| On December 27, 2012, 3,500,000 common shares were issued with a deemed value of $350,000 to Vertical Designs Ltd. |
| On December 27, 2012, 1,615,000 share purchase options were granted to certain directors, officers and consultants, with each option exercisable at $0.10 per share for a period of five years. |
On December 12, 2012, 2,100,000 units were issued at a price of $0.05 per unit for gross proceeds of $105,000 to arm's length parties. Each unit consists of one common share and one share purchase warrant of the Company. Each share purchase warrant entitles the holder to purchase one additional common share at a price of $0.13 per share for a period of one year. |
| On November 26, 2012, 2,500,000 common shares were issued with a fair value of $150,000. |
| On September 27, 2012, 3,409,090 common shares were issued with a deemed value of $375,000 and 2,727,272 common shares with a deemed value of $136,364 were issued to the CEO of the Company. |
| On August 21, 2012, 160,000 (pre consolidation 800,000) shares were issued with a deemed value of $40,000 to settle a debt payable of $40,000 to a director. |
| On August 21, 2012, 800,000 (pre consolidation 4,000,000) common shares were issued to certain directors, officers, consultants and consultant companies at a deemed value of $200,000. |
| On August 15, 2012, 200,000 (pre consolidation 1,000,000) common shares were issued with a deemed value of $30,000 to Northern Vine Canada Inc. |
| On August 15, 2012, 68,842 (pre consolidation 344,209) common shares were issued with a deemed value of $17,210 to settle a debt payable of $17,210 to certain creditors of Northern Vine Canada Inc. |
| On June 13, 2012 and August 15, 2012, 600,000 (pre consolidation 3,000,000) common shares were issued with a deemed value of $150,000 to Wakabayashi Fund LLC for consulting services. |
| On March 26, 2012, 30,000 warrants (pre consolidation 150,000) were exercised at $0.25 (pre consolidation $0.05) per warrant for proceeds of $7,500. |
| On March 16, 2012, the Company acquired Animo Wellness Corporation for $25,000 cash and 100,000 (pre-consolidation 500,000) common shares, which had an estimated fair value of $25,000 on closing date. Amino Wellness Corporation changed its name to iJuana Cannabis Inc. January 28, 2014. |
| On February 21, 2012, 57,143 (pre consolidation 285,714) common shares were issued with a fair value of $20,000 to a consultant for consulting services. |
On February 14, 2012, 1,200,000 (pre consolidation 6,000,000) units were issued at a price of $0.35 (pre consolidation 6,000,000) units for gross proceeds of $300,000. Each unit consists of one common share and one share purchase warrant of the issuer. Each warrant is exercisable into a share for a period of 12 months at an exercise price of $0.35 (pre consolidation $0.07) per share. |
217
| On February 7, 2012, 140,000 (pre consolidation 700,000) common shares were issued with a deemed value of $38,500 to settle a trade payable of $49,000. |
| On October 12, 2011, 100,000 (pre consolidation 500,000) common shares were issued with a deemed value of $22,500 to settle a debt to one of the directors. |
| On September 30, 2011, 500,000 warrants were exercised at $0.05 per common share for proceeds of $25,000. |
218
The undersigned Registrant hereby undertakes that,
| 1. | For purposes of determining any liability under the Securities Act of 1933, the information omitted from the form of prospectus filed as part of this registration statement in reliance upon Rule 430A and contained in the form of prospectus filed by the Registrant pursuant to Rule 424(b)(1) or (4) or 497(h) under the Securities Act shall be deemed to be part of this registration statement as of the time it was declared effective. |
| 2. | For the purpose of determining any liability under the Securities Act of 1933, each post-effective amendment that contains a form of prospectus shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| 3. | To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
| i. | To include any prospectus required by Section 10(a)(3) of the Securities Act of 1933; |
| ii. | To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20% change in the maximum aggregate offering price set forth in the "Calculation of Registration Fee" table in the effective registration statement. |
| iii. | To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement; |
| 4. | That, for the purpose of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| 5. | To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering. |
| 6. | That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use. |
| 7. | That, for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of securities: |
219
| i. | The undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to the registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer and sell such securities to such purchaser: |
| Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; |
| Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; |
| The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and |
| Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
| Exhibit Number | Description |
| 3(i) | Articles of Incorporation |
| 3(ii) | Articles |
| 5 | Legal Opinion |
| 10.1 | Audit Committee Charter |
| 10.2 | Investment Agreement dated January 28, 2015 with Dutchess |
| 10.3 | Registration Rights Agreement dated January 28, 2015 with Dutchess |
| 10.4 | Amending Registration Rights Agreement dated April 1, 2015 |
| 10.5 | Stock Option Plan |
| 10.6 | Consulting Agreement dated March 1, 2012 with Terence Fealey |
| 10.7 | Consulting Agreement dated January 6, 2013 with Georges Laraque Management Inc. |
| 10.8 | Consulting Agreement dated October 1, 2013 with Crimson Opportunities Ltd. |
| 10.9 | Consulting Agreement dated January 1, 2014 with Chiron Capital Corp. |
| 10.10 | Consulting Agreement dated January 1, 2014 with Growing Strategies Inc. |
| 10.11 | Consulting Agreement dated January 1, 2014 with Voelpel Gold Medal Investments Ltd. |
| 10.12 | Consulting Agreement dated February 1, 2014 with Golden Straw Consulting Group Inc. |
| 10.13 | Consulting Agreement dated March 16, 2014 with Think Sharp Inc. |
| 10.14 | Consulting Agreement dated April 1, 2014 with TDM Financial. |
| 10.15 | Distribution Agreement dated April 20, 2012 with Hedley Enterprises Ltd. |
| 10.16 | Licensing Agreement dated December 27, 2012 with Vertical Designs Ltd. |
| 10.17 | Sublicensing Agreement dated July 31, 2014 with Company subsidiary Biocube Green Grow Systems Corp. |
| 10.18 | Licensing Agreement dated August 6, 2014 between Company subsidiary Biocube Green Grow Systems Corp. and TerraSphere Systems LLC |
| 10.19 | Office Lease Agreement dated November 1, 2012 with Toro Holdings Ltd. |
| 10.20 | Office Lease Agreement dated December 18, 2013 between Company's subsidiary Animo Wellness Corporation (nka iJuana Cannabis Inc.) and PurGenesis Technologies Inc. |
| 10.21 | Facility Lease Agreement dated May 26, 2014 with Crimson Opportunities Ltd. |
220
| Exhibit Number | Description |
| 10.22 | Share Exchange Agreement dated April 10, 2014 between Company's subsidiary Northern Vine Canada Inc. and Experion Biotechnologies Inc. |
| 21 | List of Subsidiaries of Company |
| 23.1 | Consent of MNP LLP, Chartered Accountants |
| 23.2 | Consent of Hay & Watson, Chartered Accountants |
| 23.3 | Consent of Venture Law Corporation (included in exhibit 5)* |
221
Pursuant to the requirements of the Securities Act of 1933, the registrant certifies that it has reasonable grounds to believe that it meets all of the requirements for filing on Form F-1 and has duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Vancouver, Province of British Columbia on April 27, 2015.
| ABATTIS BIOCEUTICALS CORP. | ||
| By: | /s/ William Fleming | |
| William Fleming | ||
| Chief Executive Officer and Director | ||
222
Pursuant to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
/s/ William Fleming | /s/ Terrence Fealey | |
William Fleming Chief Executive Officer and Director | Terrence Fealey Director and Global Product Development Advisor | |
/s/ Douglas Sorocco | /s/ Guy Dancosse | |
Douglas Sorocco Director | Guy Dancosse Director | |
| /s/ Mike Withrow |
/s/ Emanuel Montenegrino | |
Mike Withrow International Business Advisor | Emanuel Montenegrino Director | |
/s/ Rene David | ||
Rene David Chief Financial Officer and Chief Operating Officer | ||
223