Exhibit 99.1

Corporate Overview January 2018 A Novel Class of Living Medicines Synthetic BioticTM medicines to perform and deliver critical therapeutic functions to treat diseases throughout the body

Forward Looking Statements This presentation contains “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this presentation regarding strategy, future operations, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward-looking statements. In addition, when or if used in this presentation, the words “may,” “could,” “should,” “anticipate,” “believe,” “estimate,” “expect,” “intend,” “plan,” “predict” and similar expressions and their variants may identify forward-looking statements. Examples of forward-looking statements include, but are not limited to, the approach we are taking to discover and develop novel therapeutics using synthetic biology; statements regarding the potential of our platform to develop therapeutics to address a wide range of diseases including: inborn errors of metabolism, liver disease, inflammatory and immune disorders, and cancer; the future clinical development of Synthetic Biotic medicines; the potential of our technology to treat hyperammonemia and phenylketonuria; the expected timing of our anticipated clinical trial initiations; the benefit of orphan drug and fast track status; the adequacy of our capital to support our future operations and our ability to successfully initiate and complete clinical trials; the results of our collaborations; and the difficulty in predicting the time and cost of development of our product candidates. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: the uncertainties inherent in the preclinical development process; our ability to protect our intellectual property rights; and legislative, regulatory, political and economic developments, as well as those risks identified under the heading “Risk Factors” in our filings with the SEC. The foregoing review of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors included in our Quarterly Report on Form 10-Q filed with the SEC on November 13, 2017. The forward-looking statements contained in this presentation reflect our current views with respect to future events. We anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements in the future, we specifically disclaim any obligation to do so. These forward-looking statements should not be relied upon as representing our view as of any date subsequent to the date hereof.
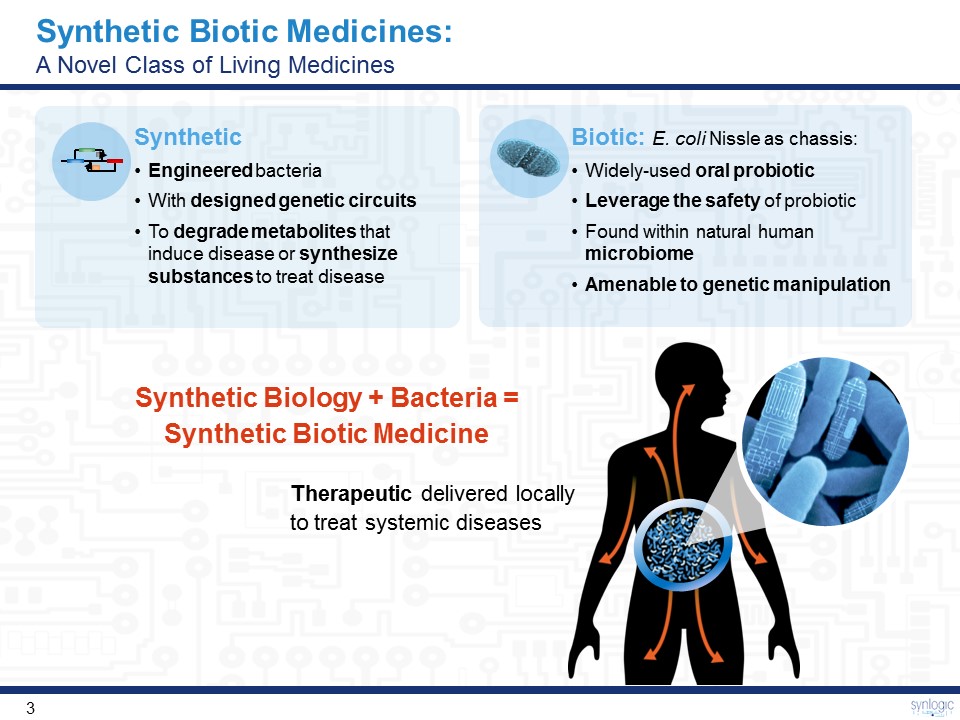
Synthetic Biotic Medicines: A Novel Class of Living Medicines Synthetic Biology + Bacteria = Synthetic Biotic Medicine Therapeutic delivered locally to treat systemic diseases SyntheticEngineered bacteriaWith designed genetic circuitsTo degrade metabolites that induce disease or synthesize substances to treat disease Biotic: E. coli Nissle as chassis: Widely-used oral probioticLeverage the safety of probioticFound within natural human microbiomeAmenable to genetic manipulation
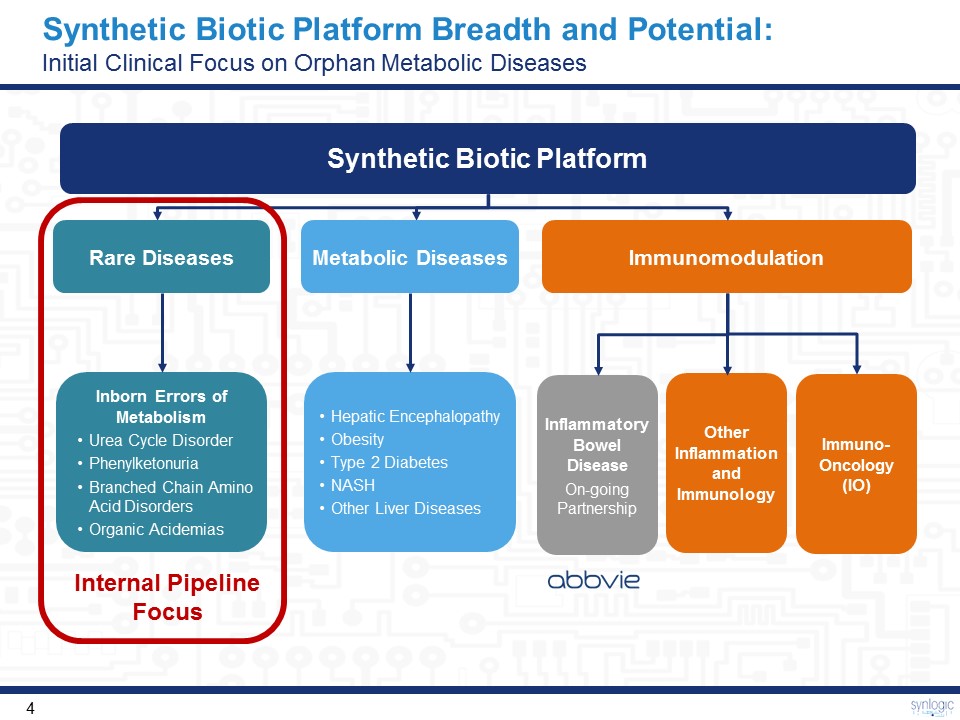
Synthetic Biotic Platform Breadth and Potential: Initial Clinical Focus on Orphan Metabolic Diseases Immuno-Oncology (IO) Synthetic Biotic Platform Internal Pipeline Focus Immunomodulation Inflammatory Bowel DiseaseOn-going Partnership Other Inflammation and Immunology Metabolic Diseases Rare Diseases Inborn Errors of MetabolismUrea Cycle DisorderPhenylketonuriaBranched Chain Amino Acid DisordersOrganic Acidemias Hepatic EncephalopathyObesityType 2 DiabetesNASHOther Liver Diseases
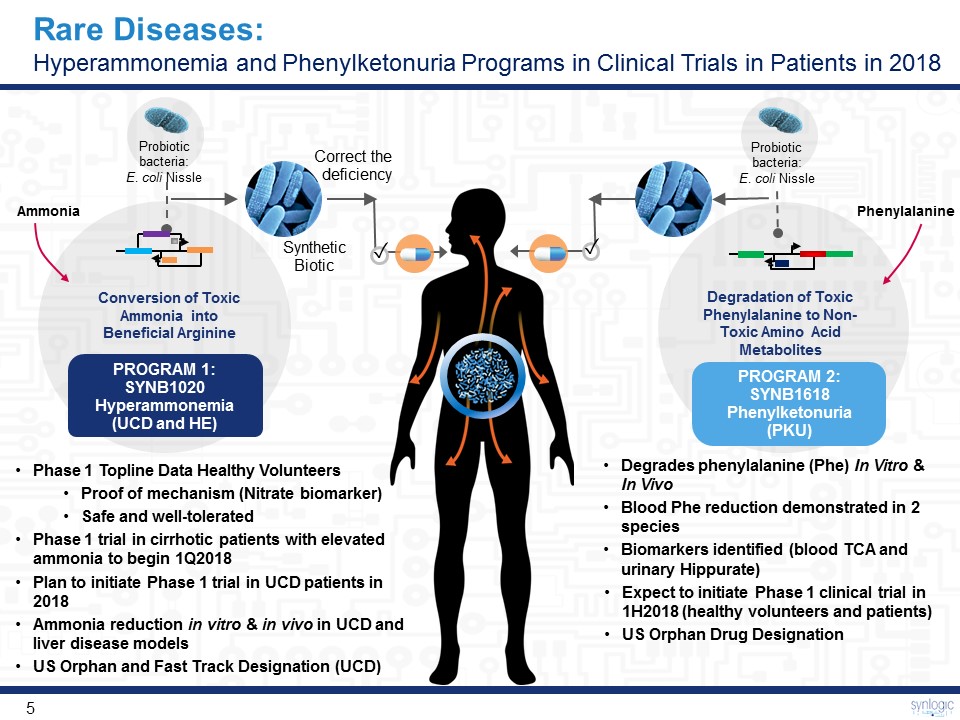
Probiotic bacteria:E. coli Nissle Correct the deficiency Ammonia ✓ Phenylalanine ✓ Synthetic Biotic Probiotic bacteria:E. coli Nissle Conversion of Toxic Ammonia into Beneficial Arginine PROGRAM 1: SYNB1020Hyperammonemia (UCD and HE) Degradation of Toxic Phenylalanine to Non-Toxic Amino Acid Metabolites PROGRAM 2: SYNB1618Phenylketonuria (PKU) Rare Diseases: Hyperammonemia and Phenylketonuria Programs in Clinical Trials in Patients in 2018 Phase 1 Topline Data Healthy VolunteersProof of mechanism (Nitrate biomarker)Safe and well-toleratedPhase 1 trial in cirrhotic patients with elevated ammonia to begin 1Q2018Plan to initiate Phase 1 trial in UCD patients in 2018Ammonia reduction in vitro & in vivo in UCD and liver disease modelsUS Orphan and Fast Track Designation (UCD) Degrades phenylalanine (Phe) In Vitro & In VivoBlood Phe reduction demonstrated in 2 speciesBiomarkers identified (blood TCA and urinary Hippurate)Expect to initiate Phase 1 clinical trial in 1H2018 (healthy volunteers and patients)US Orphan Drug Designation

Synthetic Biotic Platform Breadth and Potential:Current Pipeline Inborn Errors of MetabolismMetabolic DiseaseImmunomodulation Immuno Oncology 1: STING A/Kyn Consumer Lead Discovery Lead Optimization IND-Enabling Studies Phase I Phase II Hyperammonemia -Urea Cycle Disorder SYNB1020 Phenylketonuria SYNB1618 Organic Acidemias Maple Syrup Urine Disease Inflammatory Bowel Disease Immuno Oncology 2 Hyperammonemia -Hepatic Encephalopathy Immuno Oncology 3 SYNB1020
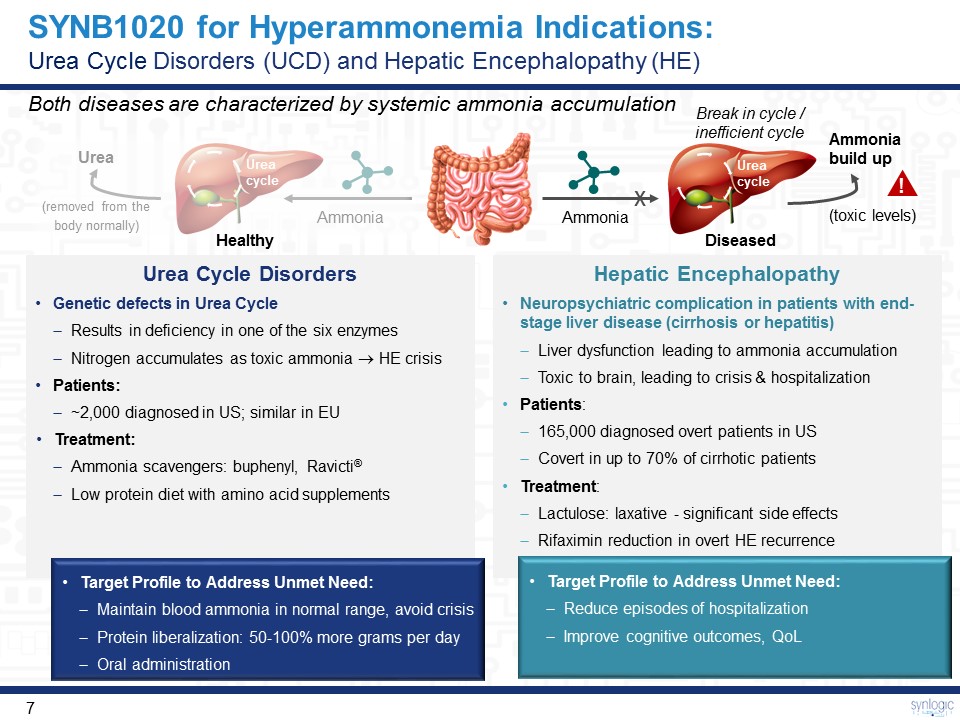
SYNB1020 for Hyperammonemia Indications: Urea Cycle Disorders (UCD) and Hepatic Encephalopathy (HE) Both diseases are characterized by systemic ammonia accumulation Break in cycle / inefficient cycle Ammonia x Ammoniabuild up (toxic levels) Urea cycle ! Urea(removed from the body normally) Urea cycle Ammonia Healthy Diseased Urea Cycle DisordersGenetic defects in Urea CycleResults in deficiency in one of the six enzymes Nitrogen accumulates as toxic ammonia HE crisisPatients: ~2,000 diagnosed in US; similar in EUTreatment: Ammonia scavengers: buphenyl, Ravicti®Low protein diet with amino acid supplements Hepatic EncephalopathyNeuropsychiatric complication in patients with end-stage liver disease (cirrhosis or hepatitis)Liver dysfunction leading to ammonia accumulationToxic to brain, leading to crisis & hospitalizationPatients: 165,000 diagnosed overt patients in USCovert in up to 70% of cirrhotic patientsTreatment: Lactulose: laxative - significant side effectsRifaximin reduction in overt HE recurrence Target Profile to Address Unmet Need:Maintain blood ammonia in normal range, avoid crisisProtein liberalization: 50-100% more grams per dayOral administration Target Profile to Address Unmet Need:Reduce episodes of hospitalizationImprove cognitive outcomes, QoL
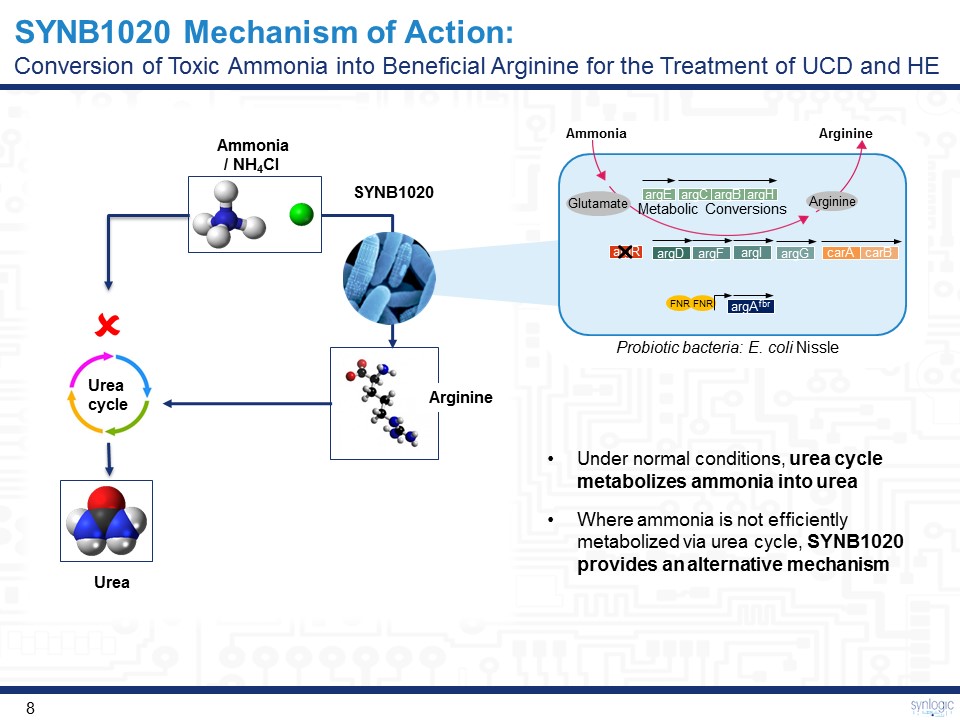
SYNB1020 Mechanism of Action:Conversion of Toxic Ammonia into Beneficial Arginine for the Treatment of UCD and HE Urea cycle Ammonia/ NH4Cl Urea Under normal conditions, urea cycle metabolizes ammonia into ureaWhere ammonia is not efficiently metabolized via urea cycle, SYNB1020 provides an alternative mechanism Arginine Probiotic bacteria: E. coli Nissle Arginine argE argC argB argH argR FNR FNR carA carB argG argl argD argAfbr Ammonia Metabolic Conversions Arginine Glutamate argF SYNB1020
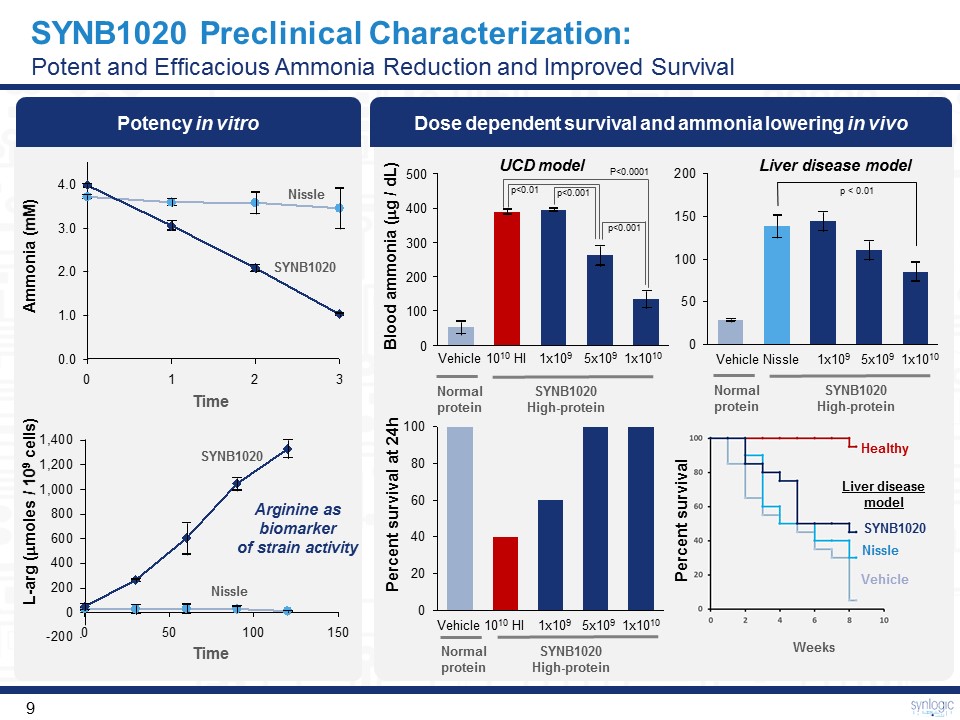
SYNB1020 Preclinical Characterization:Potent and Efficacious Ammonia Reduction and Improved Survival Potency in vitro Arginine as biomarker of strain activity Dose dependent survival and ammonia lowering in vivo 300 400 200 500 0 100 Vehicle 1010 HI 5x109 1x1010 1x109 Vehicle 1010 HI 5x109 1x1010 1x109 Vehicle Nissle 5x109 1x1010 1x109 Blood ammonia (mg / dL) Liver disease model Normalprotein SYNB1020High-protein Normalprotein SYNB1020High-protein Normalprotein SYNB1020High-protein Percent survival at 24h UCD model 4.0 0.0 3.0 2.0 1.0 Nissle SYNB1020 Time Nissle SYNB1020 Ammonia (mM) L-arg (mmoles / 109 cells) Time P<0.0001 p<0.01 p<0.001 p<0.001 Vehicle Nissle SYNB1020 Healthy Weeks Percent survival Liver disease model p < 0.01
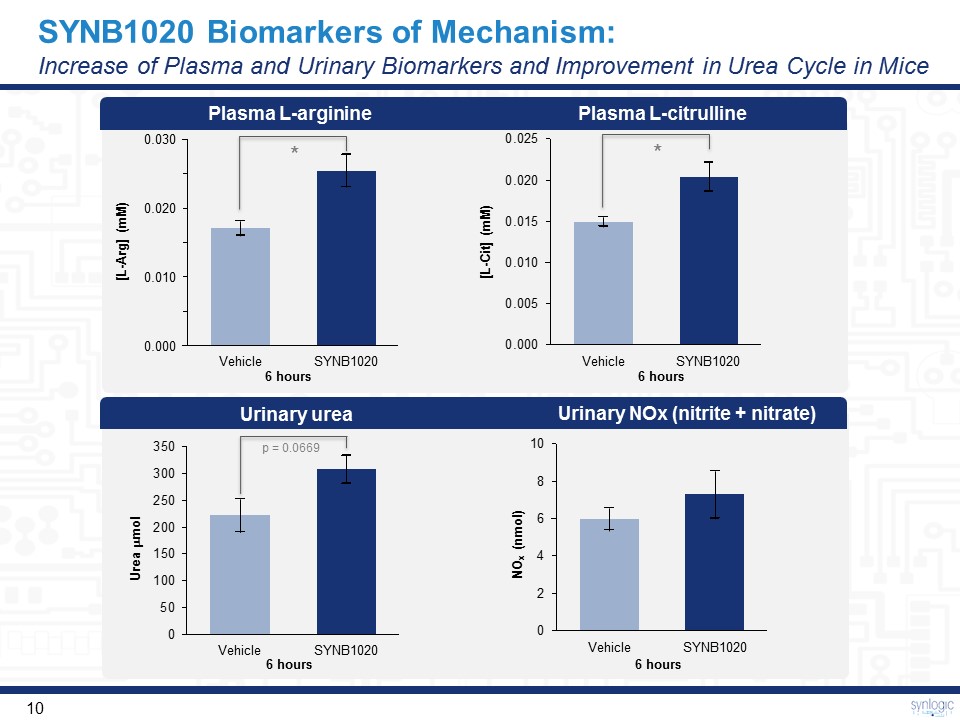
SYNB1020 Biomarkers of Mechanism:Increase of Plasma and Urinary Biomarkers and Improvement in Urea Cycle in Mice Plasma L-arginine Plasma L-citrulline 0.000 0.010 0.020 0.030 SYNB1020 Vehicle [L-Arg] (mM) 6 hours SYNB1020 Vehicle 6 hours [L-Cit] (mM) Urinary urea Urinary NOx (nitrite + nitrate) SYNB1020 Vehicle 6 hours NOx (nmol) SYNB1020 Vehicle Urea mmol 6 hours * * p = 0.0669
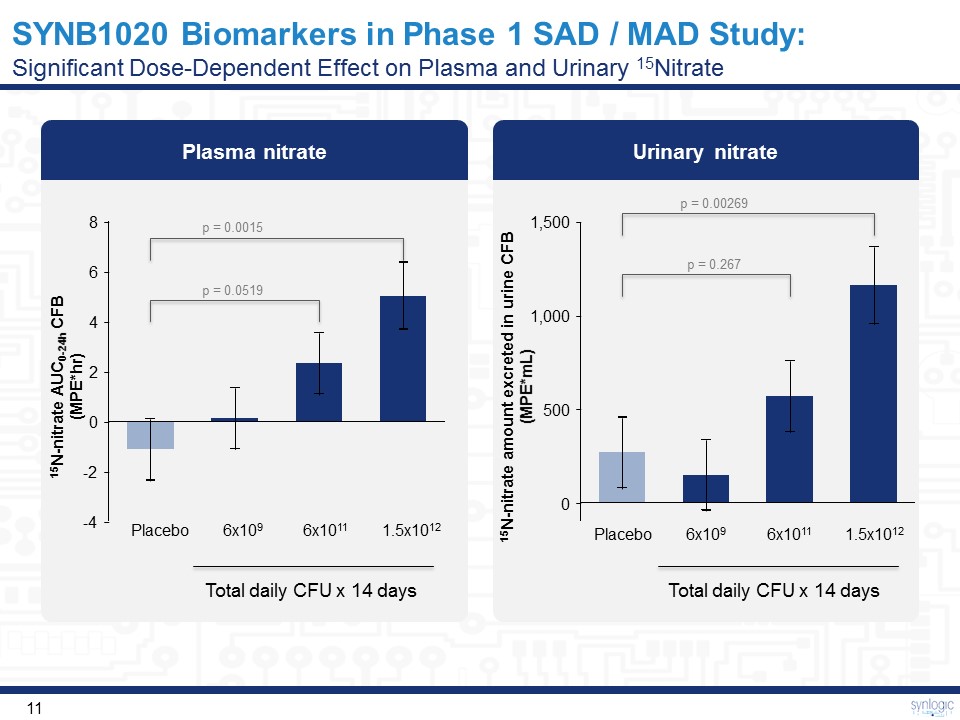
Urinary nitrate SYNB1020 Biomarkers in Phase 1 SAD / MAD Study:Significant Dose-Dependent Effect on Plasma and Urinary 15Nitrate 500 1,500 0 1,000 Placebo 6x109 6x1011 1.5x1012 15N-nitrate amount excreted in urine CFB(MPE*mL) Total daily CFU x 14 days Plasma nitrate 6 8 4 -4 -2 2 0 Placebo 6x109 6x1011 1.5x1012 Total daily CFU x 14 days 15N-nitrate AUC0-24h CFB(MPE*hr) p = 0.0519 p = 0.0015 p = 0.267 p = 0.00269
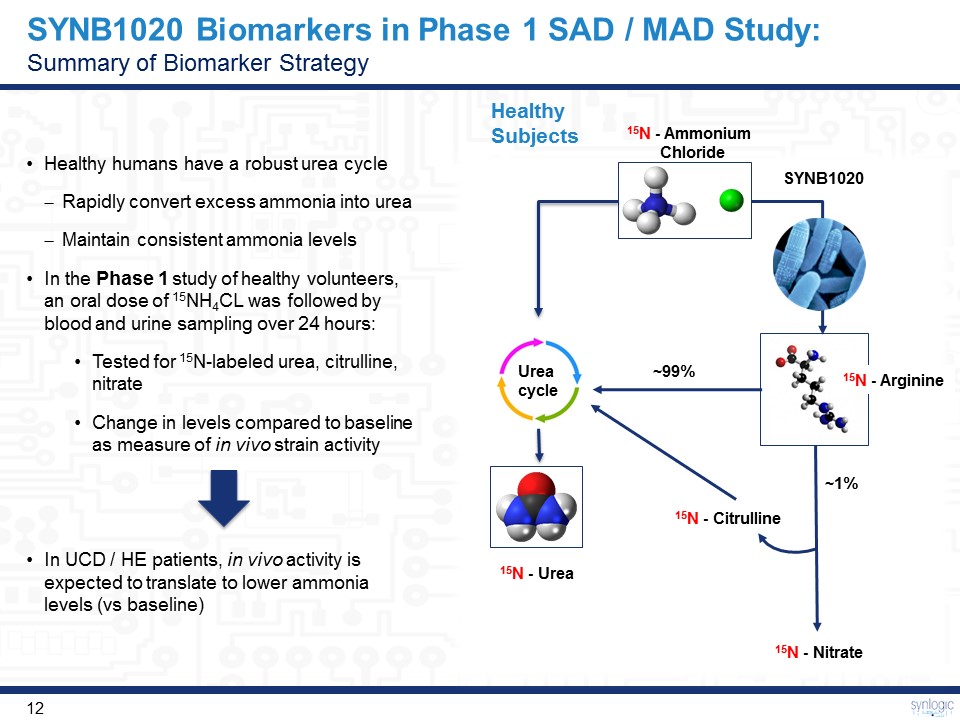
SYNB1020 Biomarkers in Phase 1 SAD / MAD Study:Summary of Biomarker Strategy Healthy humans have a robust urea cycleRapidly convert excess ammonia into ureaMaintain consistent ammonia levelsIn the Phase 1 study of healthy volunteers, an oral dose of 15NH4CL was followed by blood and urine sampling over 24 hours: Tested for 15N-labeled urea, citrulline, nitrateChange in levels compared to baseline as measure of in vivo strain activityIn UCD / HE patients, in vivo activity is expected to translate to lower ammonia levels (vs baseline) Healthy Subjects Urea cycle ~99% ~1% SYNB1020 15N - Ammonium Chloride 15N - Arginine 15N - Citrulline 15N - Nitrate 15N - Urea
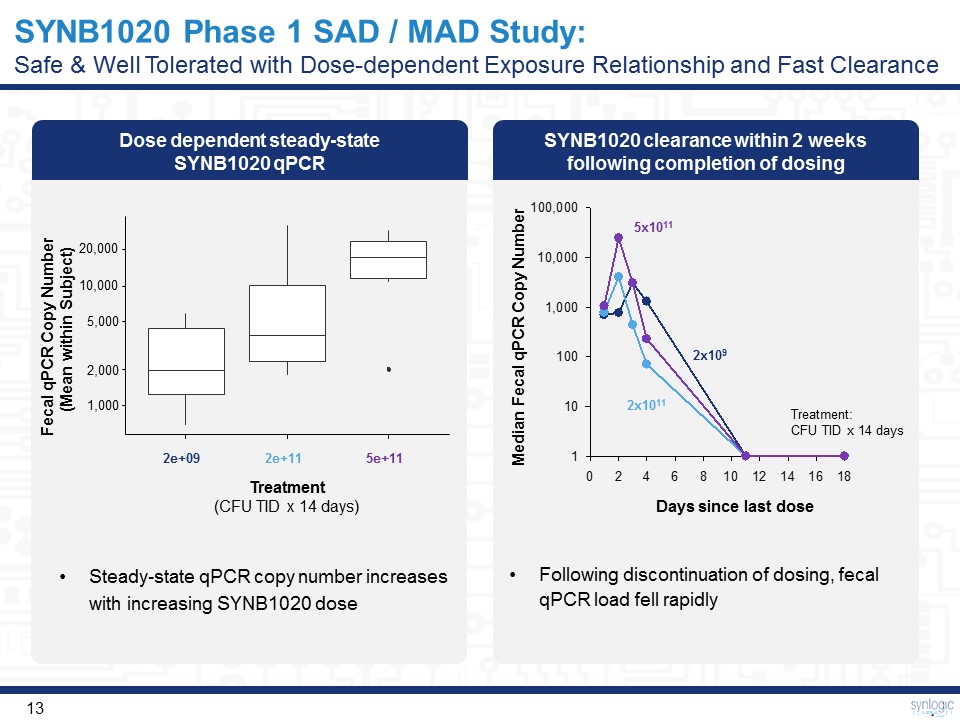
SYNB1020 clearance within 2 weeks following completion of dosing Dose dependent steady-state SYNB1020 qPCR SYNB1020 Phase 1 SAD / MAD Study:Safe & Well Tolerated with Dose-dependent Exposure Relationship and Fast Clearance Steady-state qPCR copy number increases with increasing SYNB1020 dose Following discontinuation of dosing, fecal qPCR load fell rapidly 2x109 2x1011 5x1011 Median Fecal qPCR Copy Number Treatment: CFU TID x 14 days 1,000 2,000 5,000 10,000 20,000 2e+09 2e+11 5e+11 Treatment(CFU TID x 14 days) Fecal qPCR Copy Number (Mean within Subject) Days since last dose
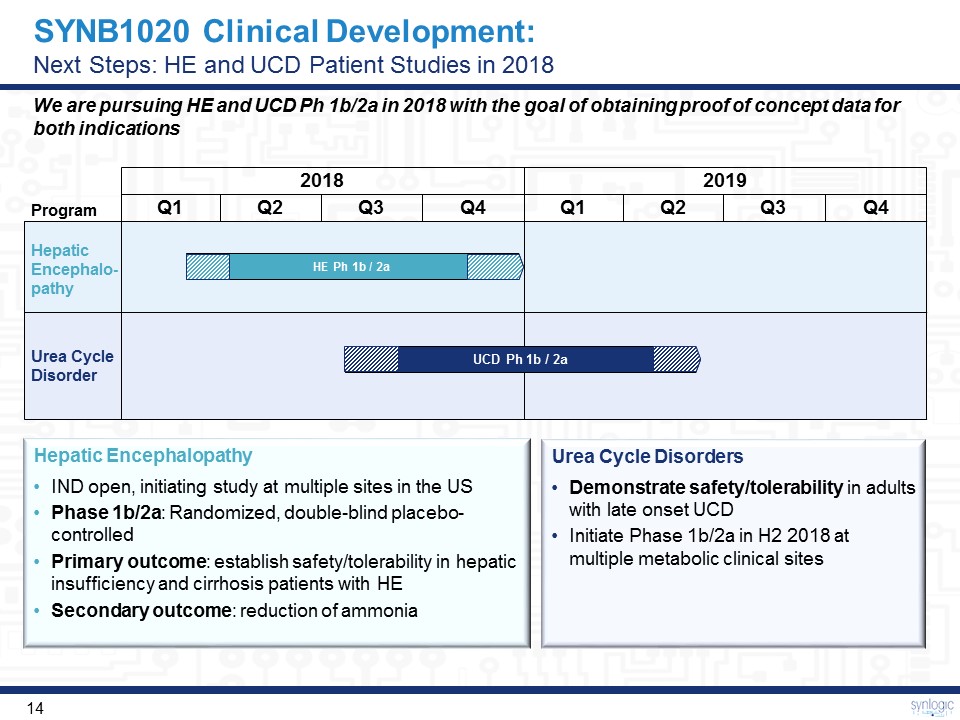
2018 2019 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 HE Ph 1b / 2a UCD Ph 1b / 2a Program Urea CycleDisorder Hepatic Encephalo-pathy SYNB1020 Clinical Development: Next Steps: HE and UCD Patient Studies in 2018 Urea Cycle DisordersDemonstrate safety/tolerability in adults with late onset UCDInitiate Phase 1b/2a in H2 2018 at multiple metabolic clinical sites Hepatic EncephalopathyIND open, initiating study at multiple sites in the USPhase 1b/2a: Randomized, double-blind placebo-controlledPrimary outcome: establish safety/tolerability in hepatic insufficiency and cirrhosis patients with HESecondary outcome: reduction of ammonia We are pursuing HE and UCD Ph 1b/2a in 2018 with the goal of obtaining proof of concept data for both indications

SYNB1618 for Phenylketonuria (PKU):Facilitating Normalization of Plasma Phe Levels Rare Inherited amino acid metabolism disorder Causes build up of amino acid phenylalanine (Phe) in the bodyPhenylalanine is found in all proteinsDiagnosed: 16,500 in US, similar in EU5If left untreated, symptoms include cognitive impairment, convulsions, behavior problems, skin rash, musty body odorTreatment: Low protein diet (no meat, dairy, nuts, eggs)Kuvan: PAH cofactor. 20-40% of patients Target Profile to Address Unmet Need:Normalize Phe: less than half manage to target (120 - 360 mmol / L, source: NPKUA)Increase protein intake to >25g (vs less than 10g typically)Oral dosing without systemic toxicity
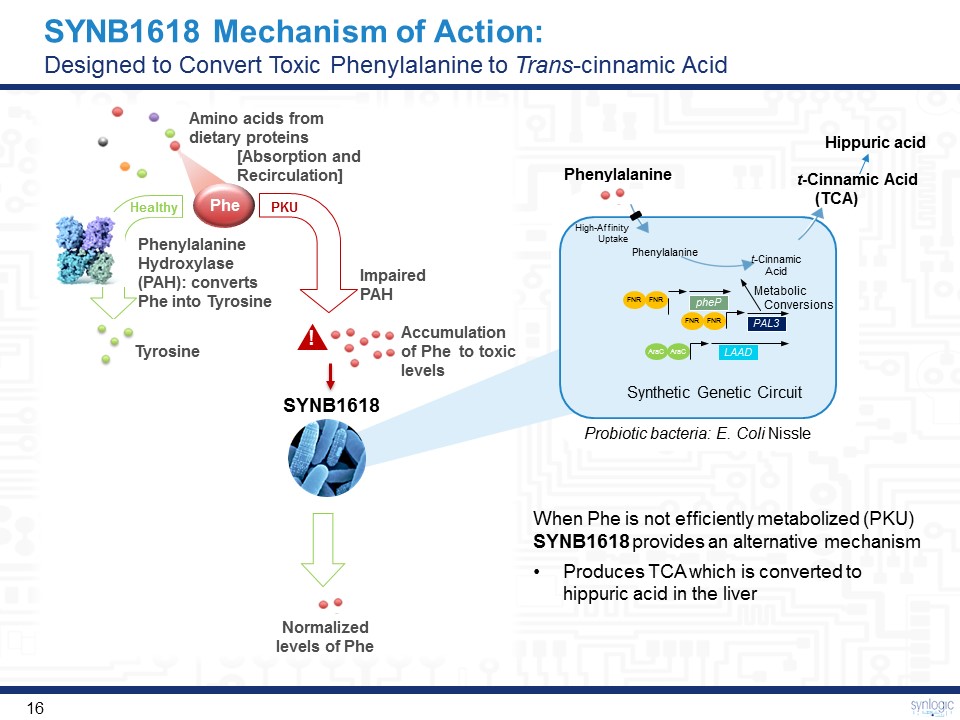
SYNB1618 Mechanism of Action:Designed to Convert Toxic Phenylalanine to Trans-cinnamic Acid Phe PKU Healthy Amino acids fromdietary proteins [Absorption andRecirculation] Phenylalanine Hydroxylase (PAH): converts Phe into Tyrosine Impaired PAH Tyrosine ! Phenylalanine Hippuric acid Normalized levels of Phe Synthetic Genetic Circuit Phenylalanine t-CinnamicAcid Metabolic Conversions High-Affinity Uptake LAAD FNR FNR PAL3 FNR FNR pheP AraC AraC t-Cinnamic Acid (TCA) SYNB1618 When Phe is not efficiently metabolized (PKU) SYNB1618 provides an alternative mechanismProduces TCA which is converted to hippuric acid in the liver Probiotic bacteria: E. Coli Nissle Accumulation of Phe to toxic levels
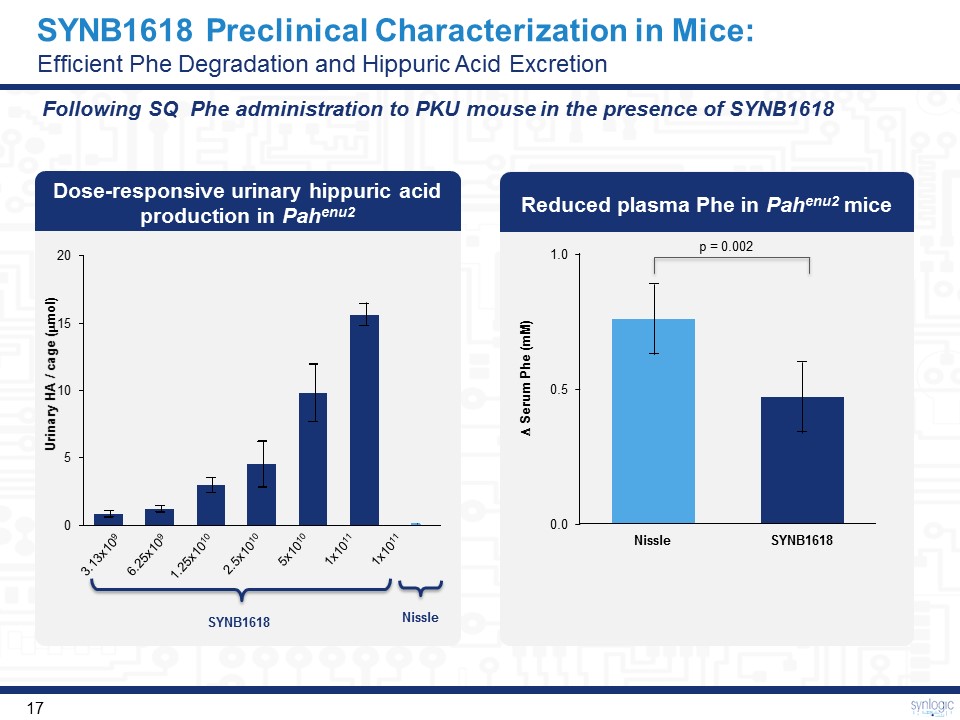
SYNB1618 Preclinical Characterization in Mice: Efficient Phe Degradation and Hippuric Acid Excretion Dose-responsive urinary hippuric acid production in Pahenu2 Reduced plasma Phe in Pahenu2 mice 3.13x109 6.25x109 1.25x1010 2.5x1010 5x1010 1x1011 1x1011 Urinary HA / cage (mmol) SYNB1618 Nissle 1.0 0.0 0.5 SYNB1618 Nissle Serum Phe (mM) p = 0.002 Following SQ Phe administration to PKU mouse in the presence of SYNB1618
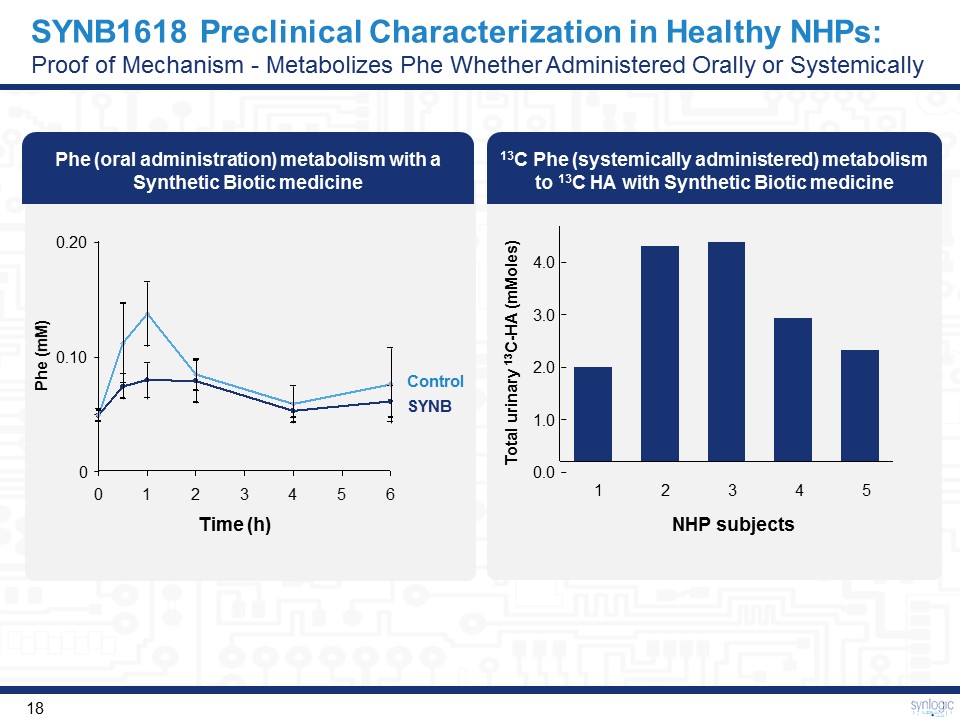
SYNB1618 Preclinical Characterization in Healthy NHPs: Proof of Mechanism - Metabolizes Phe Whether Administered Orally or Systemically Phe (oral administration) metabolism with a Synthetic Biotic medicine 13C Phe (systemically administered) metabolism to 13C HA with Synthetic Biotic medicine 0.10 0.20 0 SYNB Control Phe (mM) Time (h) 1.0 0.0 3.0 2.0 4.0 2 5 3 4 1 Total urinary 13C-HA (mMoles) NHP subjects
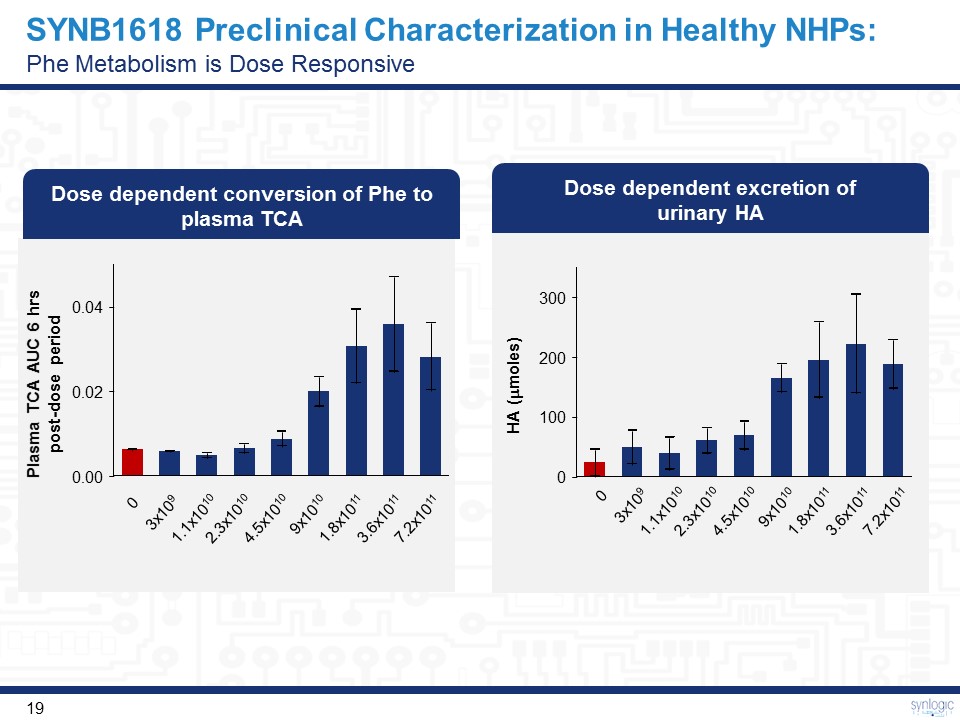
300 100 200 0 SYNB1618 Preclinical Characterization in Healthy NHPs:Phe Metabolism is Dose Responsive Dose dependent excretion of urinary HA 0 3x109 1.1x1010 2.3x1010 4.5x1010 9x1010 1.8x1011 3.6x1011 7.2x1011 HA (mmoles) Dose dependent conversion of Phe to plasma TCA 0 3x109 1.1x1010 2.3x1010 4.5x1010 9x1010 1.8x1011 3.6x1011 7.2x1011 0.04 0.00 0.02 Plasma TCA AUC 6 hrs post-dose period
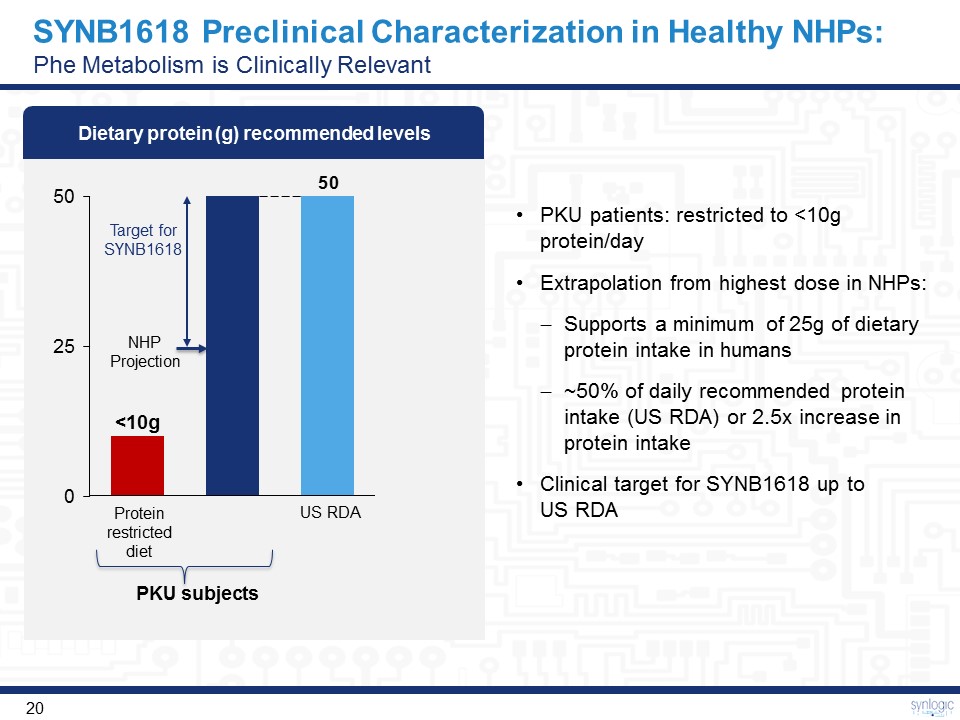
SYNB1618 Preclinical Characterization in Healthy NHPs:Phe Metabolism is Clinically Relevant PKU patients: restricted to <10g protein/dayExtrapolation from highest dose in NHPs: Supports a minimum of 25g of dietary protein intake in humans~50% of daily recommended protein intake (US RDA) or 2.5x increase in protein intakeClinical target for SYNB1618 up to US RDA PKU subjects Dietary protein (g) recommended levels 0 25 50 <10g Protein restricted diet NHP Projection US RDA Target for SYNB1618
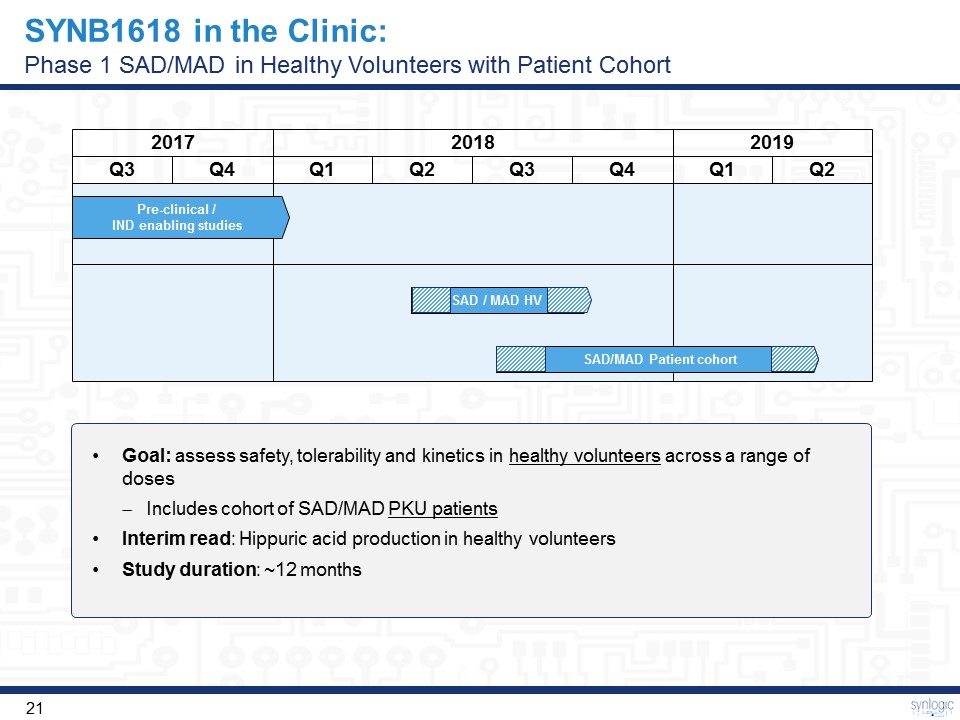
SYNB1618 in the Clinic:Phase 1 SAD/MAD in Healthy Volunteers with Patient Cohort 2017 2018 2019 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Pre-clinical / IND enabling studies SAD/MAD Patient cohort SAD / MAD HV Goal: assess safety, tolerability and kinetics in healthy volunteers across a range of dosesIncludes cohort of SAD/MAD PKU patientsInterim read: Hippuric acid production in healthy volunteersStudy duration: ~12 months
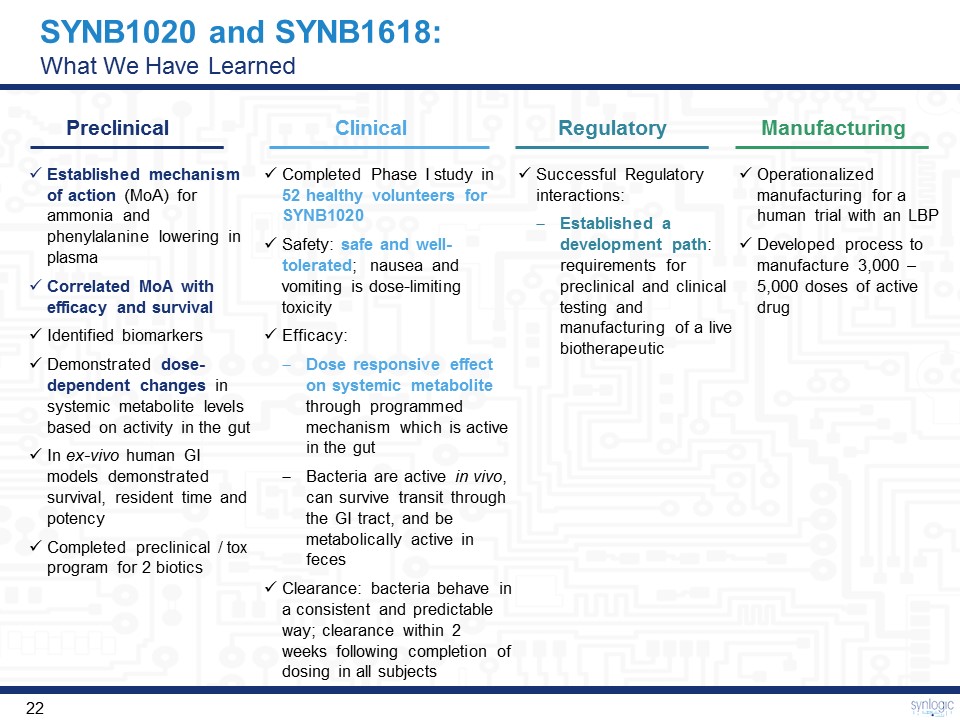
SYNB1020 and SYNB1618:What We Have Learned Preclinical Clinical Regulatory Manufacturing Successful Regulatory interactions: Established a development path: requirements for preclinical and clinical testing and manufacturing of a live biotherapeutic Completed Phase I study in 52 healthy volunteers for SYNB1020Safety: safe and well-tolerated; nausea and vomiting is dose-limiting toxicityEfficacy: Dose responsive effect on systemic metabolite through programmed mechanism which is active in the gut Bacteria are active in vivo, can survive transit through the GI tract, and be metabolically active in fecesClearance: bacteria behave in a consistent and predictable way; clearance within 2 weeks following completion of dosing in all subjects Established mechanism of action (MoA) for ammonia and phenylalanine lowering in plasmaCorrelated MoA with efficacy and survivalIdentified biomarkersDemonstrated dose-dependent changes in systemic metabolite levels based on activity in the gutIn ex-vivo human GI models demonstrated survival, resident time and potencyCompleted preclinical / tox program for 2 biotics Operationalized manufacturing for a human trial with an LBPDeveloped process to manufacture 3,000 – 5,000 doses of active drug
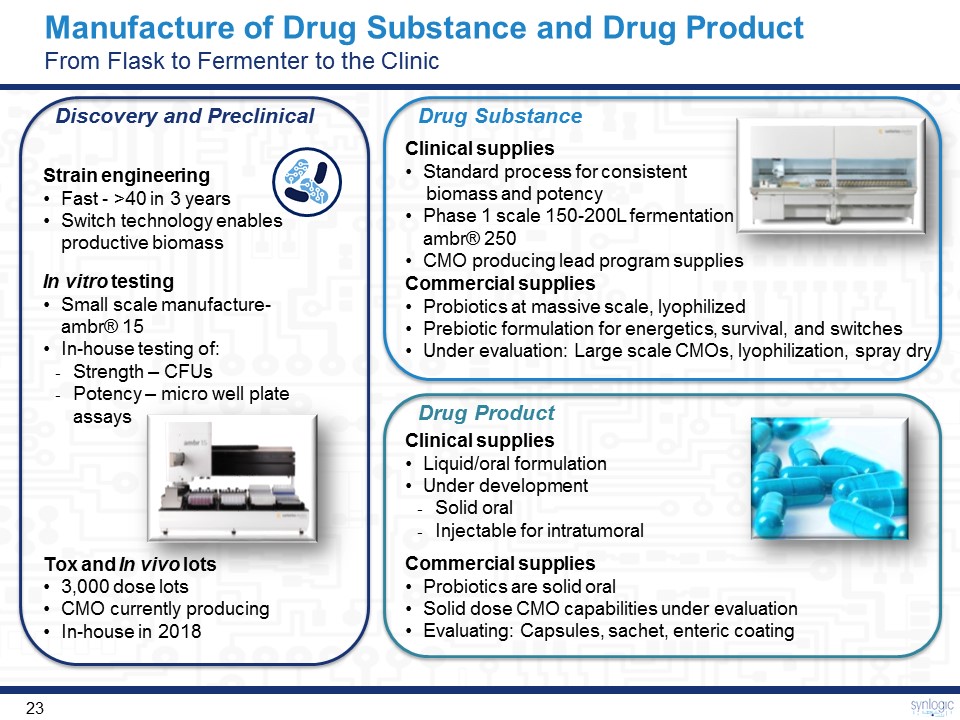
Strain engineeringFast - >40 in 3 yearsSwitch technology enables productive biomass In vitro testingSmall scale manufacture- ambr® 15In-house testing of:Strength – CFUsPotency – micro well plate assays Discovery and Preclinical Drug Substance Clinical suppliesStandard process for consistent biomass and potencyPhase 1 scale 150-200L fermentation - ambr® 250CMO producing lead program supplies Commercial suppliesProbiotics at massive scale, lyophilizedPrebiotic formulation for energetics, survival, and switchesUnder evaluation: Large scale CMOs, lyophilization, spray dry Drug Product Clinical suppliesLiquid/oral formulationUnder developmentSolid oralInjectable for intratumoral Commercial suppliesProbiotics are solid oralSolid dose CMO capabilities under evaluationEvaluating: Capsules, sachet, enteric coating Tox and In vivo lots3,000 dose lotsCMO currently producingIn-house in 2018 Manufacture of Drug Substance and Drug ProductFrom Flask to Fermenter to the Clinic
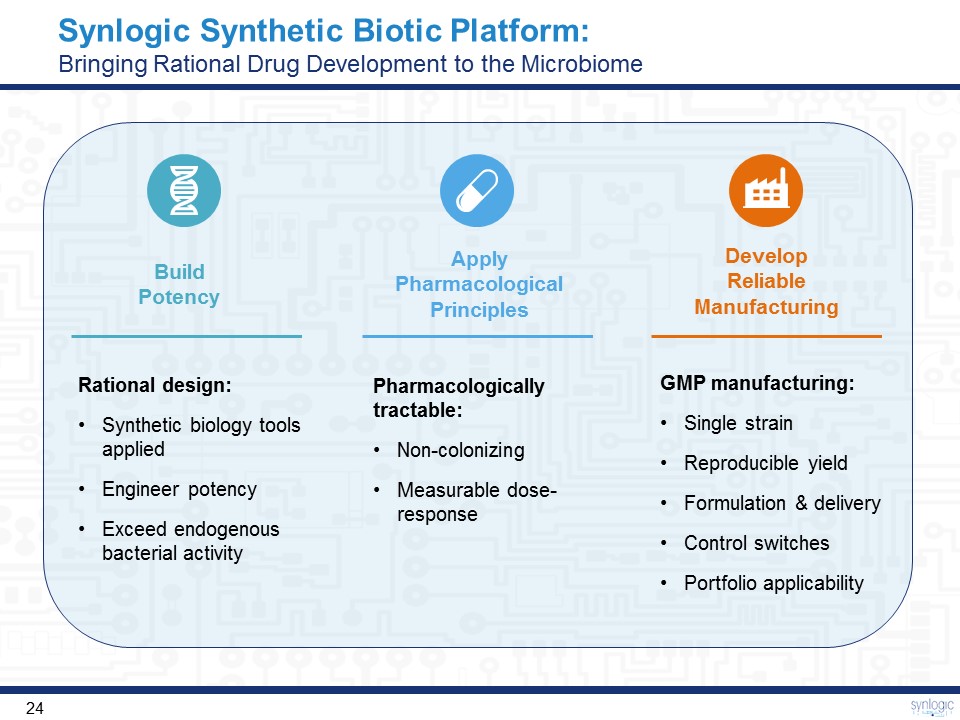
GMP manufacturing:Single strainReproducible yieldFormulation & deliveryControl switchesPortfolio applicability Synlogic Synthetic Biotic Platform: Bringing Rational Drug Development to the Microbiome Rational design:Synthetic biology tools appliedEngineer potencyExceed endogenous bacterial activity Pharmacologically tractable:Non-colonizing Measurable dose-response Apply Pharmacological Principles Develop Reliable Manufacturing Build Potency
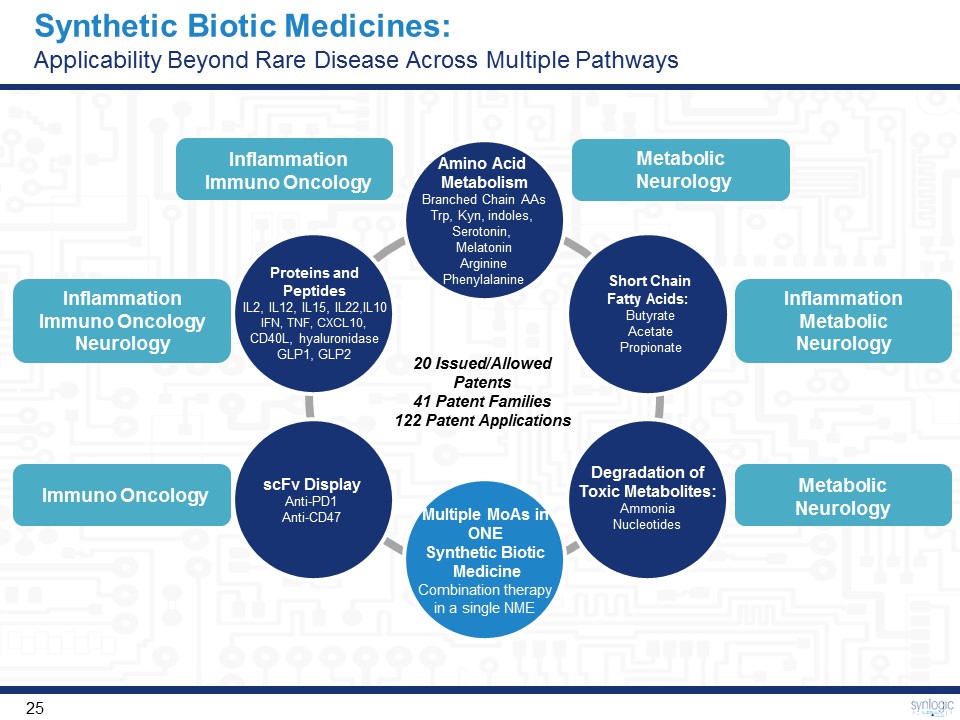
Synthetic Biotic Medicines: Applicability Beyond Rare Disease Across Multiple Pathways Short Chain Fatty Acids: ButyrateAcetatePropionate Proteins and PeptidesIL2, IL12, IL15, IL22,IL10IFN, TNF, CXCL10, CD40L, hyaluronidaseGLP1, GLP2 Amino Acid MetabolismBranched Chain AAsTrp, Kyn, indoles, Serotonin, MelatoninArgininePhenylalanine Multiple MoAs in ONE Synthetic Biotic MedicineCombination therapy in a single NME scFv DisplayAnti-PD1Anti-CD47 Degradation ofToxic Metabolites:AmmoniaNucleotides 20 Issued/Allowed Patents 41 Patent Families122 Patent Applications Inflammation MetabolicNeurology MetabolicNeurology MetabolicNeurology Inflammation Immuno Oncology Inflammation Immuno OncologyNeurology Immuno Oncology
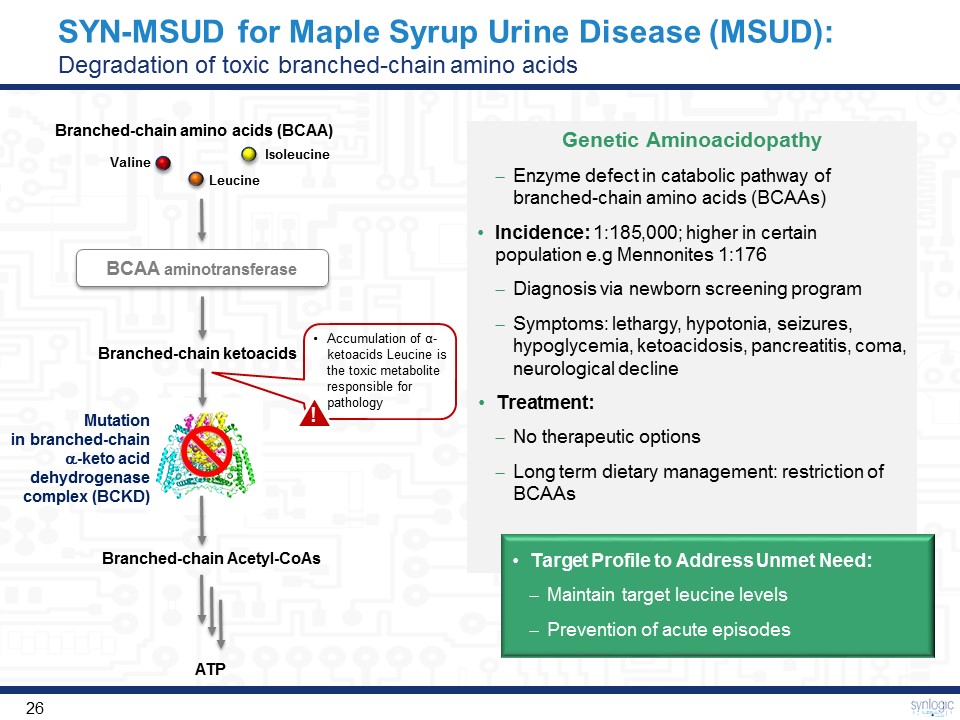
SYN-MSUD for Maple Syrup Urine Disease (MSUD):Degradation of toxic branched-chain amino acids Branched-chain amino acids (BCAA) Valine Leucine Isoleucine BCAA aminotransferase Branched-chain ketoacids Branched-chain Acetyl-CoAs Mutation in branched-chain a-keto acid dehydrogenase complex (BCKD) ATP Accumulation of α-ketoacids Leucine is the toxic metabolite responsible for pathology ! Genetic AminoacidopathyEnzyme defect in catabolic pathway of branched-chain amino acids (BCAAs)Incidence: 1:185,000; higher in certain population e.g Mennonites 1:176Diagnosis via newborn screening programSymptoms: lethargy, hypotonia, seizures, hypoglycemia, ketoacidosis, pancreatitis, coma, neurological declineTreatment: No therapeutic optionsLong term dietary management: restriction of BCAAs Target Profile to Address Unmet Need:Maintain target leucine levelsPrevention of acute episodes
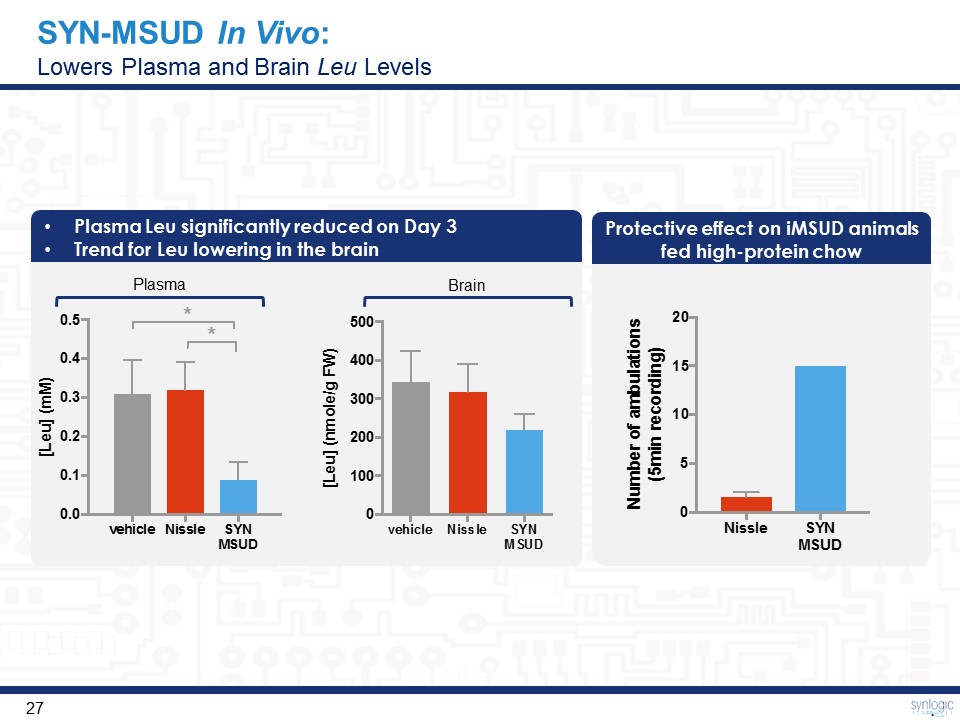
SYN-MSUD In Vivo: Lowers Plasma and Brain Leu Levels Plasma Leu significantly reduced on Day 3Trend for Leu lowering in the brain Plasma Brain Protective effect on iMSUD animals fed high-protein chow

Metabolite production, consumption, transformation Neurotransmitter or hormone production/consumption,Toxin consumption,Neuroprotectant production Bianconi et al., Atlas of Human Biology 2013Sender et al, PLoS Biology 2016 Engineered Probiotics to Modulate Cross-talk between the Gut and the Liver or Brain Chassis Pathway Preclinical Translation Delivery Engineerability Effectiveness Safety Pharmacology, modelingDoseToxicityBioavailabilityPotency Residence TimeConversion Efficiency ManufacturabilityDrug Substance and Drug Product Manufacturing: High Throughput Strain EngineeringEnzyme optimization Pathway balancingIndustrial scale gene synthesisAutomated and Parallel TestingMutli-dimensional analyticsIntegrated manufacturabilityPredictive translational Understand disease pathology Identify mechanisms to restore healthDesignBioinformaticsPathway enumerationIn silico designSystems-level characterization Unprecedented Engine for Rational Drug Development of Living Medicines
HORIZON 3 HORIZON 2 Test simple synthetic biology concepts in the local tumor microenvironment Harness microbiome metabolomics to widen response to cancer immunotherapies Rationally build full minimal genomes solely designed for wide therapeutic use in cancer HORIZON 1 Synlogic Mission: To Re-define Medicine through Synthetic BiologyBringing Minimal Therapeutic Genomes to Oncology
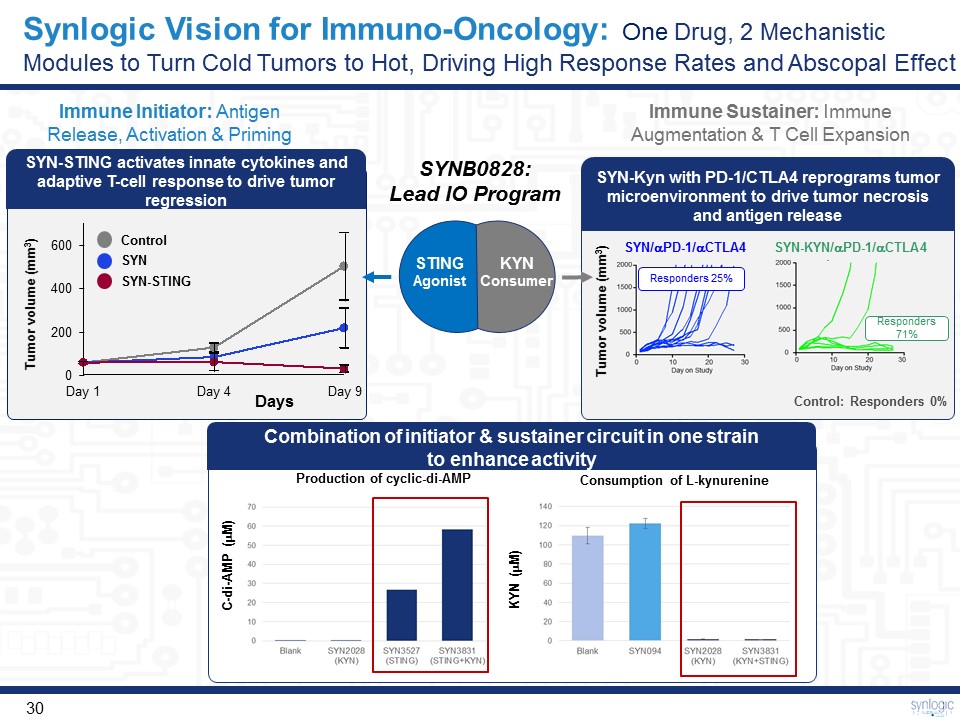
Synlogic Vision for Immuno-Oncology: One Drug, 2 Mechanistic Modules to Turn Cold Tumors to Hot, Driving High Response Rates and Abscopal Effect STING Agonist KYNConsumer SYNB0828: Lead IO Program Immune Initiator: Antigen Release, Activation & Priming Immune Sustainer: Immune Augmentation & T Cell Expansion SYN-Kyn with PD-1/CTLA4 reprograms tumor microenvironment to drive tumor necrosis and antigen release SYN-STING activates innate cytokines and adaptive T-cell response to drive tumor regression Combination of initiator & sustainer circuit in one strain to enhance activity Production of cyclic-di-AMP Consumption of L-kynurenine KYN (mM) C-di-AMP (mM) 200 400 600 0 Day 1 Day 9 Day 4 Control SYN SYN-STING Days Tumor volume (mm3) Control: Responders 0% SYN/aPD-1/aCTLA4 SYN-KYN/aPD-1/aCTLA4 Responders71% Responders 25% Tumor volume (mm3)
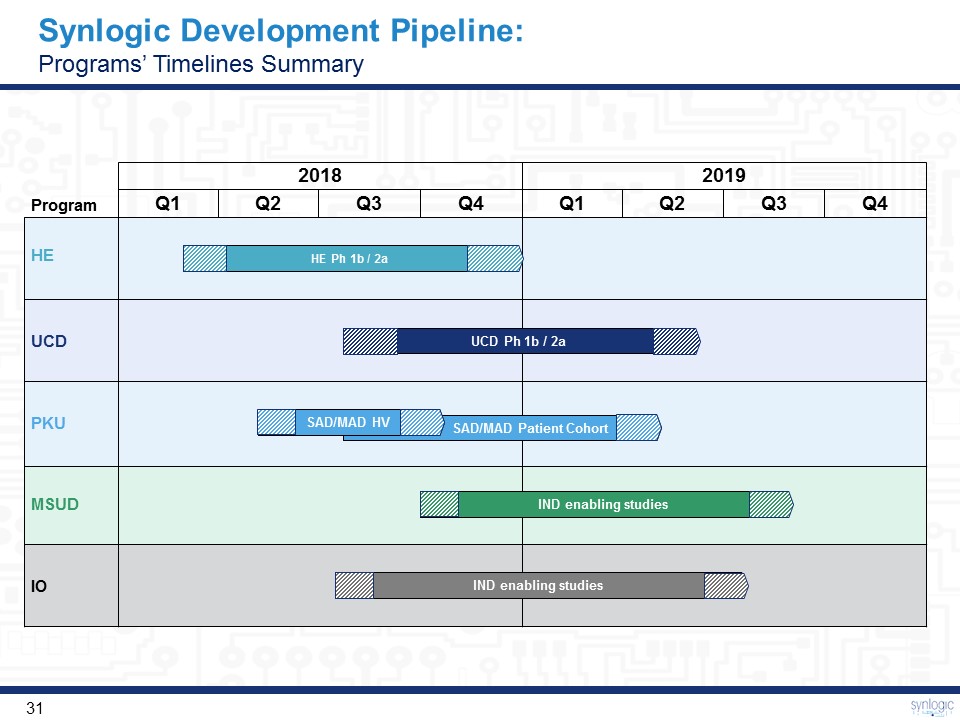
Synlogic Development Pipeline:Programs’ Timelines Summary 2018 2019 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 SAD/MAD Patient Cohort HE Ph 1b / 2a IND enabling studies IND enabling studies SAD/MAD HV UCD Ph 1b / 2a PKU Program IO UCD MSUD HE

Synlogic:Significant Delivery in 2017 …. Primed to Deliver in 2018 2017 Accomplishments 2018 Goals Significant pipeline progress: SYNB1020: UCD / HEPhase 1 completionFast track designationSYNB1618: PKUOrphan statusIND on track for Q1 ’18Corporate:Public listing on NASDAQ1st milestone in AbbVie collaboration in IBDStrategic collaboration with Gingko BioworksOrganization growth: hiring into key roles to support clinical and manufacturing functions Programs:SYNB1020: UCD / HEPhase 1 results presentation at medical conferences: Q1 ’18Initiate Phase 1b / 2a in HE in Q1 ’18Initiate Phase 1b / 2a in UCD mid ’18SYNB 1618: PKUPhase 1 SAD / MAD study in HV and PKU patients in H1 ’18Early pipeline: new indications (including IO) data presentation at major meetingsCorporate:Advance existing collaborationsExpand platform reach through new partnerships

Synlogic Overview NovelTherapeuticClass Dominant Synthetic Biotic IP PortfolioAs of Jan 2018 Strong Balance Sheet Leader of therapeutic synthetic biology, genetically reprogram probiotics for transformative impact on disease treatmentSimple, robust and rapid process for the discovery, development and manufacturing of drug candidatesKey differentiation: potency, pharmacology/dose responses, reproducible manufacturing Robust Pipelinewith Orphan Drug Programs SYNB1020 for Hyperammonemia including Urea Cycle Disorder (UCD) & Hepatic Encephalopathy (HE). Healthy volunteer study completed November 2017: Safe and well tolerated, positive Proof of Mechanism SYNB1618 for Phenylketonuria (PKU); Positive PoM in NHPs. 2018 IND and clinical study $96.6M in cash as of 3Q 2017Investors include: Aju IB Investment, Ally Bridge Group, Atlas Venture, Deerfield Management, New Enterprise Associates (NEA), OrbiMed, Perceptive Advisors, Rock Springs Capital Highly Experienced Management Team JC Gutierrez-Ramos, CEOAoife Brennan, CMOTodd Shegog, CFOAndrew Gengos, COO Paul Miller, CSODean Falb, CTORichard Schwartz, SVP ManufacturingCaroline Kurtz, VP Translational Science Broad Platform - Multiple Product Opportunities Immuno Oncology: Non pathogenic bacterial chasis “armed” synthetically with effector functions that activate/expand immune response for tumor regression and memory response Inflammatory Bowel Disease (IBD) partnered with Abbvie Liver Diseases and CNS diseases: direct exposure to Liver/plasma of designed metabolites 20 Issued/Allowed Patents41 Patent Families122 Pending Patent Applications


Synlogic Management Team: From Funding of Platform to Clinic in Less than Three Years JC Gutierrez-Ramos, CEO Group SVP Biotherapeutics, PfizerSVP, Head Immunoinflammation Center for Drug Discovery, GSKCSO & Site Head, Amgen Mountain View Dean Falb, CTOEntrepreneur in Residence, Atlas VentureVP, R&D, Stryker Regenerative Medicine Aoife Brennan, CMOVP, Rare Disease Innovation Unit, BiogenMedical Director, Tolerx Caroline Kurtz, SVP, Translational Science Vice President, GCC Platform Lead, Ironwood PharmaceuticalsDirector, Infectious Diseases, Genzyme Todd Shegog, CFOSVP & CFO, Forum PharmaceuticalsSVP & CFO, Millennium Pharmaceuticals Dick Schwartz, SVP, ManufacturingChief, Vaccine Production Program Lab, NIHSenior Director, Process & Manufacturing Sciences, MedImmune Paul Miller, CSO VP, Infection iScience, AstraZenecaVP, Antibacterials Research Unit, Pfizer Andrew Gengos, COO & Head of Corp. Dev.President and CEO ImmunoCellular TherapeuticsPresident & CEO Neuraltus PharmaceuticalsVP, Strategy & Corp. Development Amgen,VP, CFO & CBO Dynavax Technologies Adam Thomas, CHROVP, Head of Human Resources for R&D, ShireHead of HR for Research, Development & Engineering, S.C. Johnson Co Maiken Keson-Brooks, General CounselSVP, General Counsel, uniQureSVP, General Counsel, Forum Pharmaceuticals
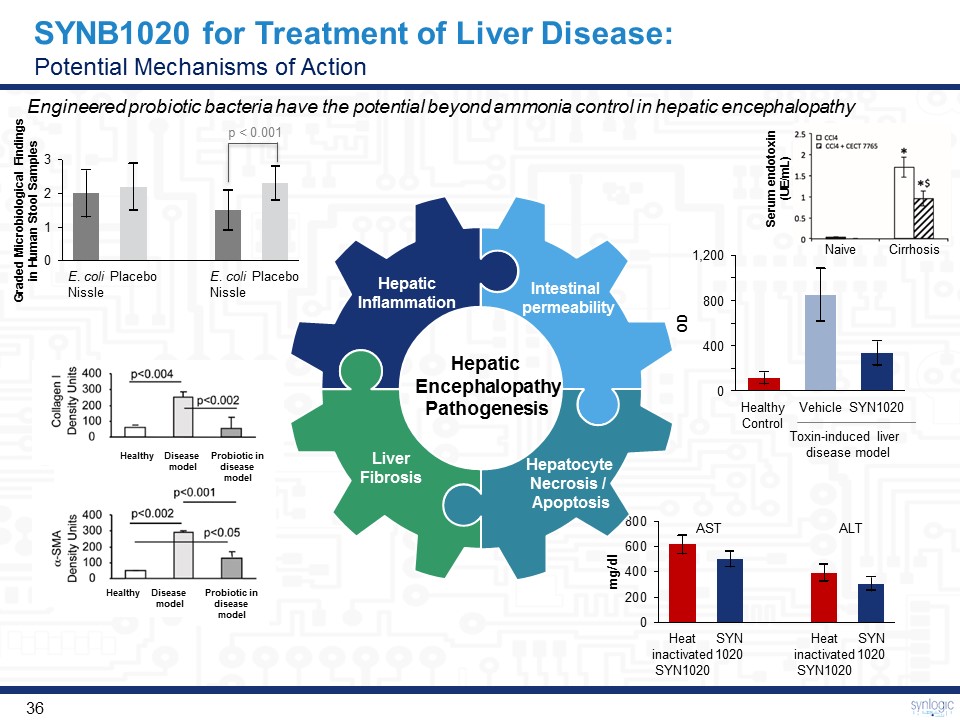
SYNB1020 for Treatment of Liver Disease:Potential Mechanisms of Action Engineered probiotic bacteria have the potential beyond ammonia control in hepatic encephalopathy Hepatic EncephalopathyPathogenesis 800 400 0 1,200 HealthyControl SYN1020 Vehicle SYN1020 Heat inactivated SYN1020 Heat inactivated SYN1020 SYN1020 AST ALT OD mg/dl Toxin-induced liver disease model Probiotic in disease model Probiotic in disease model Healthy Healthy Disease model Disease model Intestinal permeability Hepatocyte Necrosis / Apoptosis LiverFibrosis HepaticInflammation E. coli Nissle Placebo E. coli Nissle Placebo Graded Microbiological Findings in Human Stool Samples p < 0.001 Serum endotoxin (UE/mL) Naive Cirrhosis
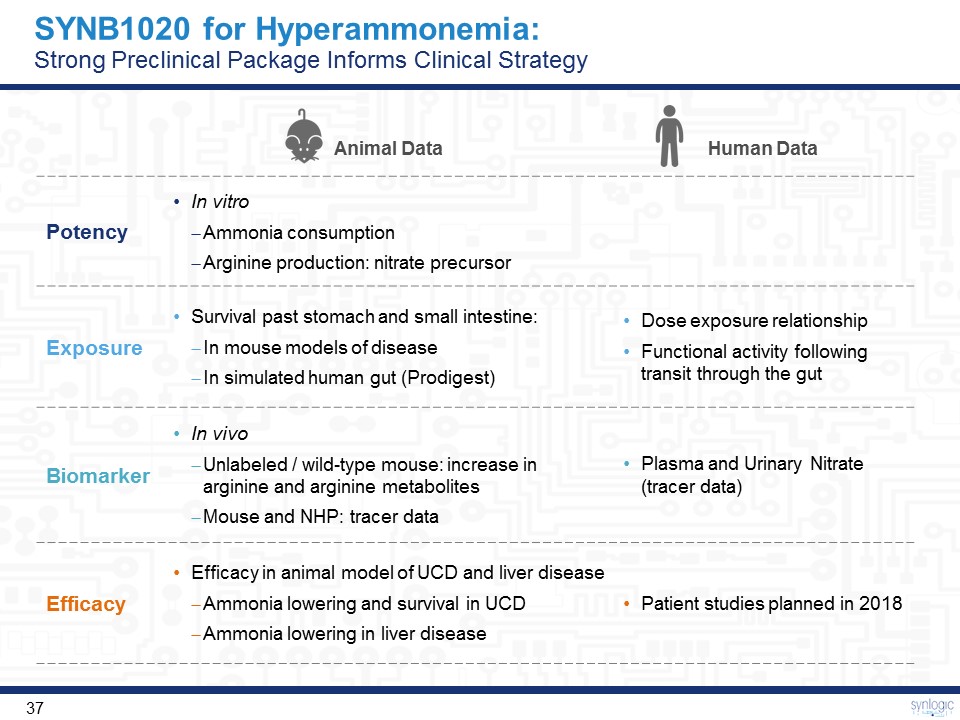
Animal Data Human Data Potency In vitroAmmonia consumptionArginine production: nitrate precursor Exposure Survival past stomach and small intestine:In mouse models of disease In simulated human gut (Prodigest) Dose exposure relationshipFunctional activity following transit through the gut Biomarker In vivoUnlabeled / wild-type mouse: increase in arginine and arginine metabolitesMouse and NHP: tracer data Plasma and Urinary Nitrate (tracer data) Efficacy Efficacy in animal model of UCD and liver diseaseAmmonia lowering and survival in UCDAmmonia lowering in liver disease Patient studies planned in 2018 SYNB1020 for Hyperammonemia:Strong Preclinical Package Informs Clinical Strategy
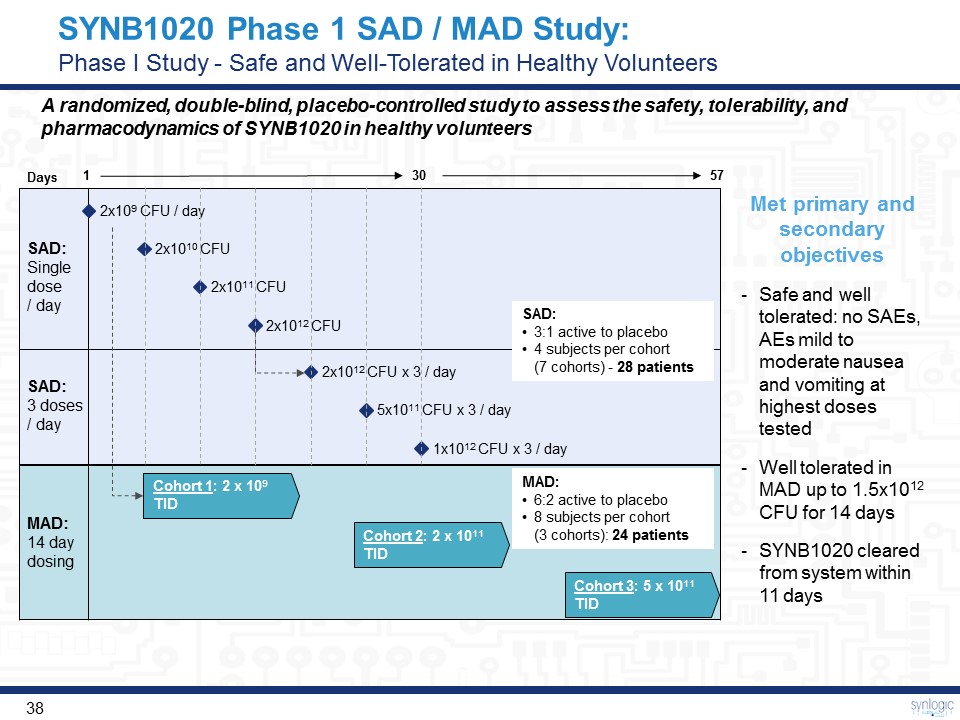
Cohort 2: 2 x 1011 TID Cohort 1: 2 x 109 TID Cohort 3: 5 x 1011 TID 1x1012 CFU x 3 / day Days 2x1010 CFU 5x1011 CFU x 3 / day 2x109 CFU / day 2x1011 CFU 2x1012 CFU x 3 / day 2x1012 CFU A randomized, double-blind, placebo-controlled study to assess the safety, tolerability, and pharmacodynamics of SYNB1020 in healthy volunteers SAD:Single dose / day SAD:3 doses/ day MAD:14 day dosing 1 30 57 MAD:6:2 active to placebo8 subjects per cohort (3 cohorts): 24 patients SAD:3:1 active to placebo4 subjects per cohort (7 cohorts) - 28 patients Met primary and secondary objectives Safe and well tolerated: no SAEs, AEs mild to moderate nausea and vomiting at highest doses testedWell tolerated in MAD up to 1.5x1012 CFU for 14 daysSYNB1020 cleared from system within 11 days SYNB1020 Phase 1 SAD / MAD Study:Phase I Study - Safe and Well-Tolerated in Healthy Volunteers

SYNB1020: Toxicology Package and Regulatory Path Orphan Drug Designation (UCD, PKU)Fast Track Designation (UCD)Feedback from FDA Office of Vaccines Research and Review (CBER)No Recombinant DNA Advisory Committee (RAC) requiredLowering of blood ammonia level is an approvable end-point (UCD) Preclinical:No toxicity at highest feasible dose in two speciesNo evidence of distribution outside the GI tractClinical:Well tolerated in MAD up to 1.5 x 1012 CFU for 14 daysMild nausea and vomiting at higher dose52 healthy volunteers dosed orally with either SYNB1020 or placeboSAD-28 subjects in 7 cohortsMAD-24 subjects in 3 cohortsSYNB1020 cleared from system within expected timeframe Regulatory Preclinical and Clinical Safety & Toxicology
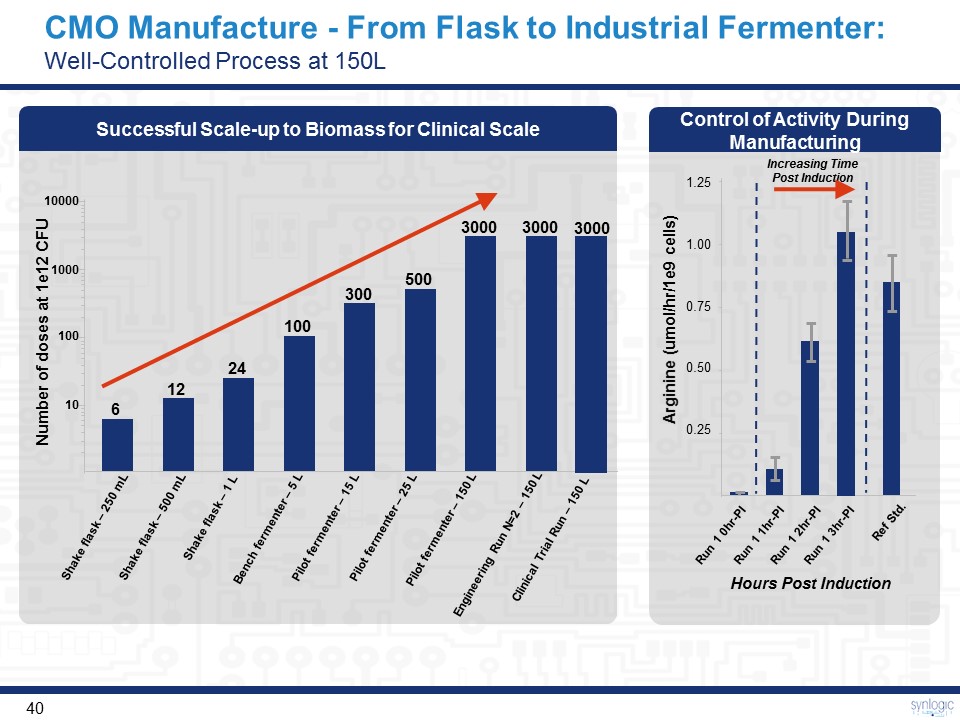
CMO Manufacture - From Flask to Industrial Fermenter: Well-Controlled Process at 150L Successful Scale-up to Biomass for Clinical Scale Number of doses at 1e12 CFU 10000 1000 100 10 Shake flask – 250 mL Shake flask – 500 mL Shake flask – 1 L Bench fermenter – 5 L Pilot fermenter – 25 L Pilot fermenter – 150 L Engineering Run N=2 – 150 L Pilot fermenter – 15 L 6 12 24 100 300 500 3000 3000 Increasing Time Post Induction Run 1 0hr-PI Run 1 1hr-PI Run 1 2hr-PI Run 1 3hr-PI Ref Std. 1.25 1.00 0.75 0.50 0.25 Arginine (umol/hr/1e9 cells) Control of Activity During Manufacturing Hours Post Induction Clinical Trial Run – 150 L 3000
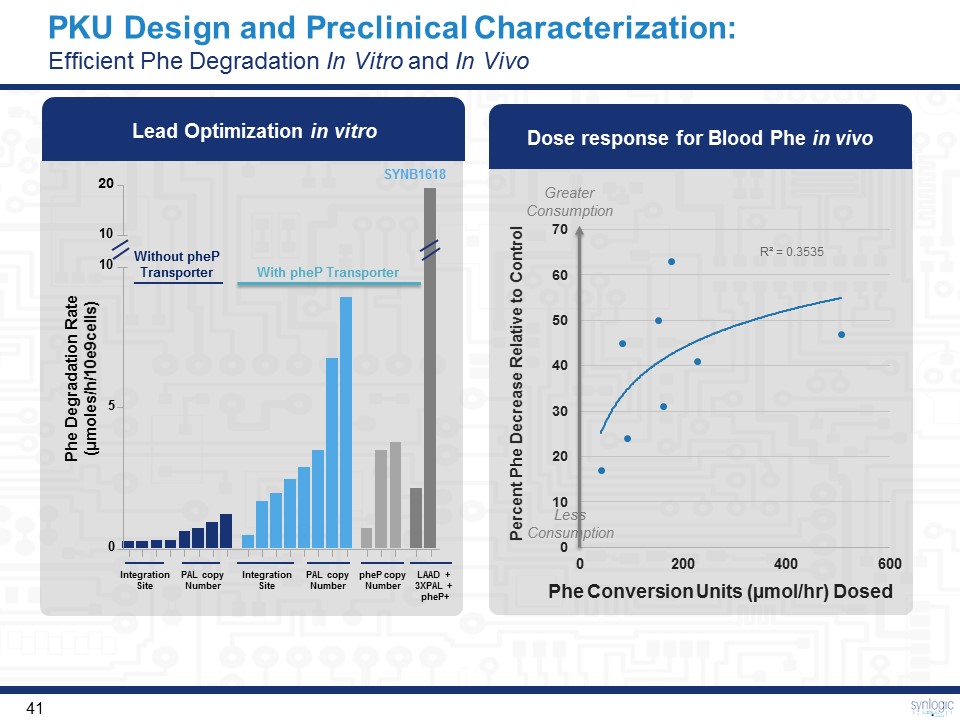
SYNB1618 PKU Design and Preclinical Characterization:Efficient Phe Degradation In Vitro and In Vivo With pheP Transporter Without pheP Transporter 20 10 10 0 5 Phe Degradation Rate(μmoles/h/10e9cells) IntegrationSite PAL copyNumber IntegrationSite PAL copyNumber pheP copyNumber LAAD +3XPAL + pheP+ Lead Optimization in vitro Greater Consumption Less Consumption Dose response for Blood Phe in vivo
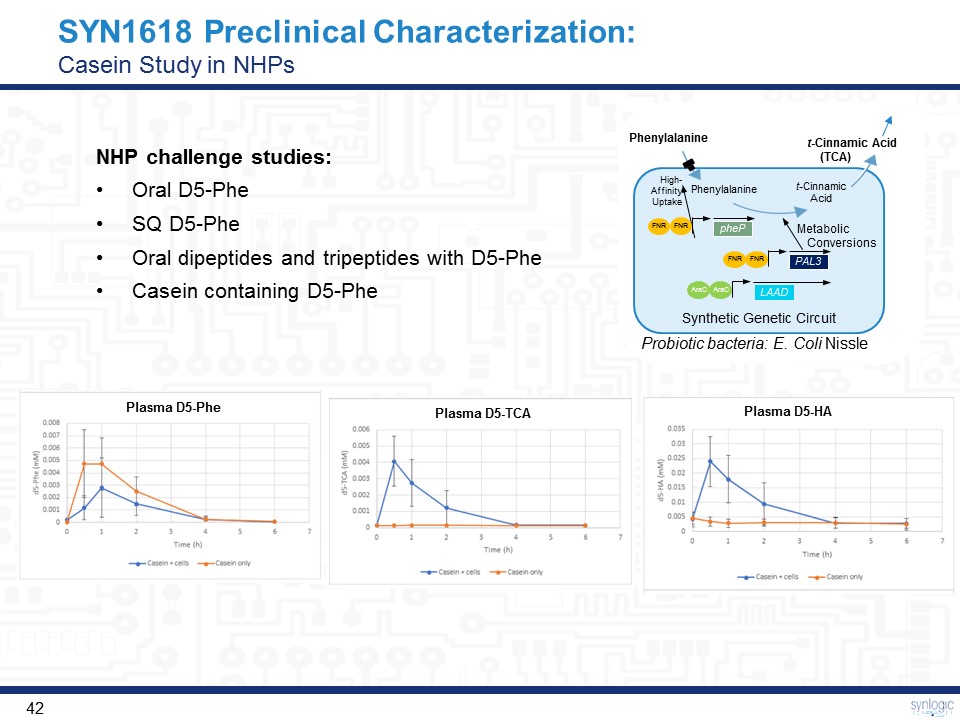
SYN1618 Preclinical Characterization:Casein Study in NHPs NHP challenge studies:Oral D5-PheSQ D5-PheOral dipeptides and tripeptides with D5-PheCasein containing D5-Phe Plasma D5-Phe Plasma D5-TCA Plasma D5-HA Phenylalanine Probiotic bacteria: E. Coli Nissle Synthetic Genetic Circuit Phenylalanine t-CinnamicAcid Metabolic Conversions High-Affinity Uptake LAAD FNR FNR PAL3 FNR FNR pheP AraC AraC t-Cinnamic Acid (TCA)
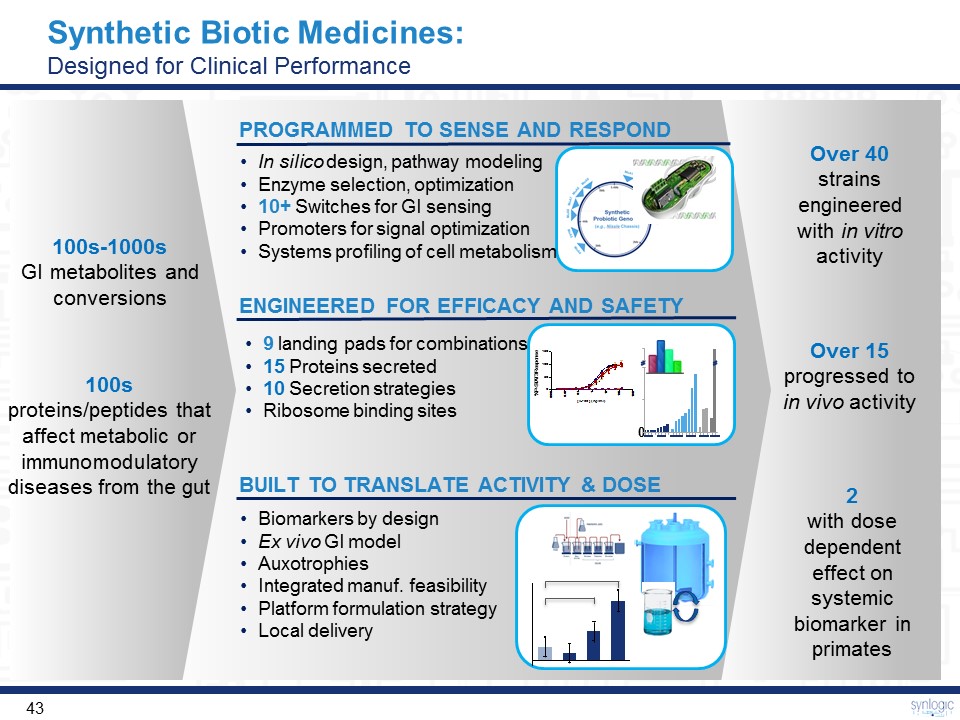
Synthetic Biotic Medicines: Designed for Clinical Performance BUILT TO TRANSLATE ACTIVITY & DOSE In silico design, pathway modelingEnzyme selection, optimization10+ Switches for GI sensingPromoters for signal optimizationSystems profiling of cell metabolism PROGRAMMED TO SENSE AND RESPOND ENGINEERED FOR EFFICACY AND SAFETY Biomarkers by designEx vivo GI modelAuxotrophiesIntegrated manuf. feasibilityPlatform formulation strategyLocal delivery 100sproteins/peptides that affect metabolic or immunomodulatory diseases from the gut 100s-1000sGI metabolites and conversions Over 15progressed to in vivo activity 2 with dose dependent effect on systemic biomarker in primates Over 40strains engineered with in vitro activity 9 landing pads for combinations15 Proteins secreted10 Secretion strategiesRibosome binding sites 0
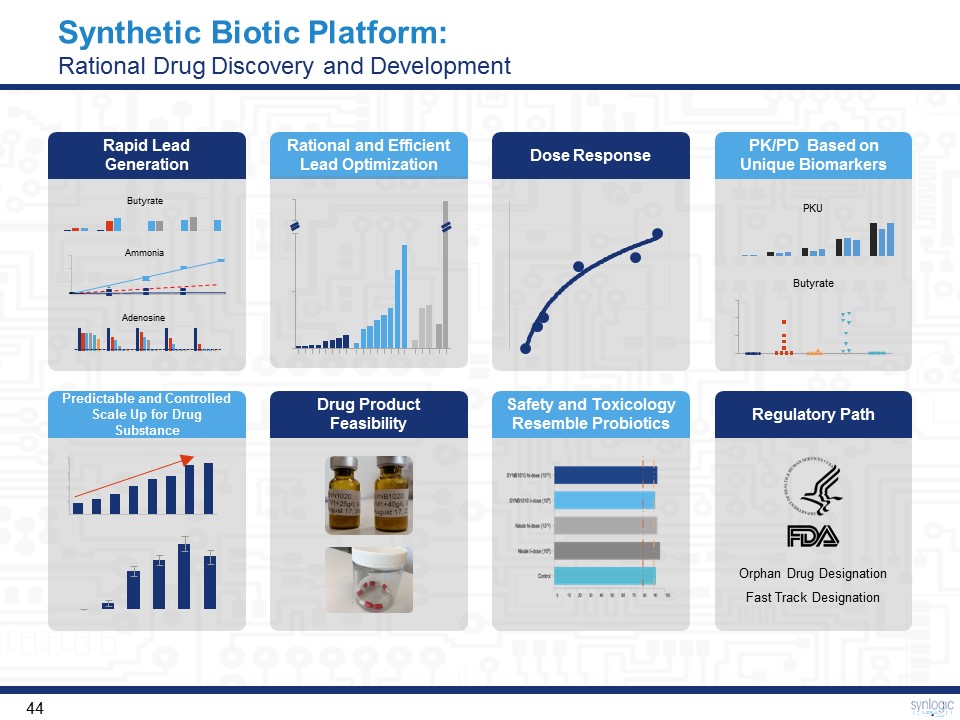
Synthetic Biotic Platform: Rational Drug Discovery and Development Rapid Lead Generation Predictable and Controlled Scale Up for Drug Substance Rational and Efficient Lead Optimization Drug Product Feasibility PK/PD Based on Unique Biomarkers Regulatory Path Dose Response Safety and ToxicologyResemble Probiotics Butyrate Ammonia Adenosine PKU Orphan Drug DesignationFast Track Designation Butyrate
