Exhibit 99.1

Discussion of Interim Analysis of Data from Phase 1/2a Clinical Trial of SYNB1618 September 4, 2018 A Novel Class of Living Medicines Synthetic BioticTM medicines to perform and deliver critical therapeutic functions to treat diseases throughout the body

Forward-Looking Statements This presentation and various remarks which may be made during this presentation contain “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995, including statements regarding Synlogic’s plans and expectations for the development of SYNB1618. All statements, other than statements of historical facts, regarding strategy, future operations, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward-looking statements. In addition, when or if used in this presentation and various remarks which may be made during this presentation, the words “may,” “could,” “should,” “anticipate,” “believe,” “estimate,” “expect,” “intend,” “plan,” “predict” and similar expressions and their variants, as they relate to Synlogic may identify forward-looking statements. Examples of forward-looking statements, include, but are not limited to, statements regarding the potential of Synlogic’s platform to develop therapeutics to address a wide range of diseases including: cancer, inborn errors of metabolism, liver disease, and inflammatory and immune disorders; the future clinical development of Synthetic Biotic medicines; the approach Synlogic is taking to discover and develop novel therapeutics using synthetic biology; and the expected timing of Synlogic’s clinical trials and availability of clinical trial data. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including: the uncertainties inherent in the preclinical development process; the ability of Synlogic to protect its intellectual property rights; and legislative, regulatory, political and economic developments, as well as those risks identified under the heading “Risk Factors” in Synlogic’s filings with the SEC. The forward-looking statements reflect Synlogic’s current views with respect to future events. Synlogic anticipates that subsequent events and developments will cause its views to change. However, while Synlogic may elect to update these forward-looking statements in the future, Synlogic specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing Synlogic’s view as of any date subsequent to the date hereof.

Synthetic Biotic Platform Breadth and Potential:Current Pipeline Inborn Errors of MetabolismMetabolic DiseaseImmunomodulation Immuno Oncology 1 Lead Discovery Lead Optimization IND-Enabling Studies Phase I Phase II Hyperammonemia -Urea Cycle Disorder SYNB1020 Phenylketonuria SYNB1618 Organic Acidemias Maple Syrup Urine Disease Inflammatory Bowel Disease Immuno Oncology 2 Hyperammonemia -Hepatic Encephalopathy Immuno Oncology 3 SYNB1020

SYNB1618 for Phenylketonuria (PKU):Goal: Managing Plasma Phe Levels to Enable Increased Intake of Natural Protein PKU is a rare inherited amino acid metabolism disorder Causes build up of amino acid phenylalanine (Phe) in the bodyPhenylalanine is found in all proteinsDiagnosed: 16,500 in US, similar in EU5If left untreated, symptoms include cognitive impairment, convulsions, behavior problems, skin rashTreatment: Low protein diet (no meat, dairy, nuts, eggs)Kuvan: PAH cofactor. 20-40% of patientsPalynziq: injectable, pegylated, bacterial enzyme (PAL) (Adults) Target Profile to Address Unmet Need:Manage Phe: Currently < half adults at target (120 - 360 mmol / L, source: NPKUA)Increase natural protein intake (less than 10g typically)Oral dosing without systemic toxicity
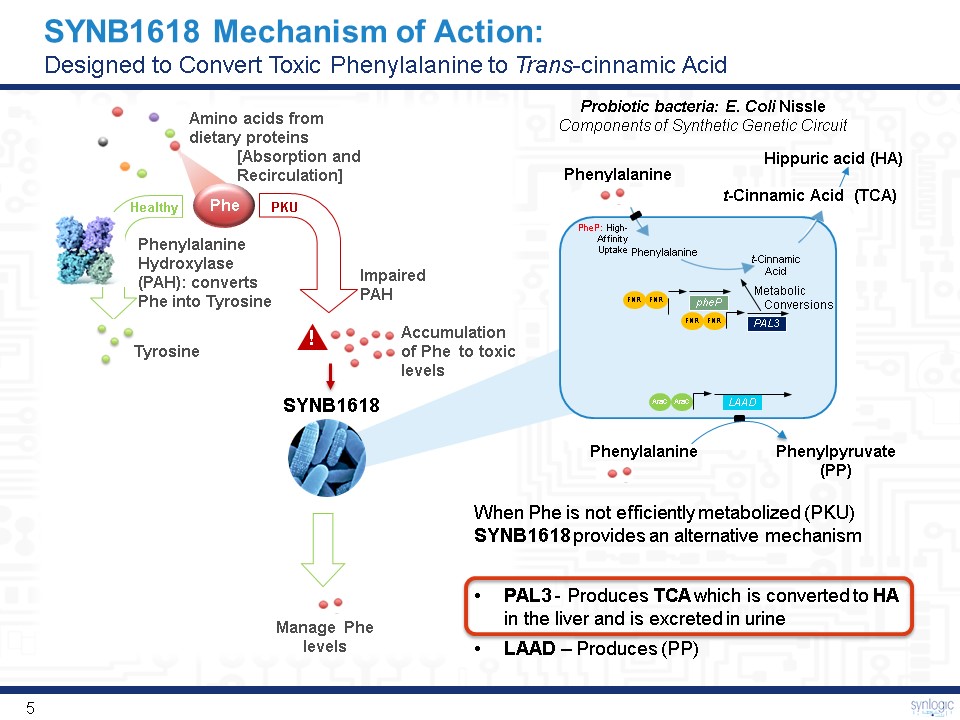
SYNB1618 Mechanism of Action:Designed to Convert Toxic Phenylalanine to Trans-cinnamic Acid Phe PKU Healthy Amino acids fromdietary proteins [Absorption andRecirculation] Phenylalanine Hydroxylase (PAH): converts Phe into Tyrosine Impaired PAH Tyrosine ! Phenylalanine Hippuric acid (HA) Manage Phe levels Phenylalanine t-CinnamicAcid Metabolic Conversions PheP: High-Affinity Uptake FNR FNR PAL3 FNR FNR pheP LAAD AraC AraC t-Cinnamic Acid (TCA) SYNB1618 When Phe is not efficiently metabolized (PKU) SYNB1618 provides an alternative mechanismPAL3 - Produces TCA which is converted to HA in the liver and is excreted in urineLAAD – Produces (PP) Accumulation of Phe to toxic levels Phenylalanine Probiotic bacteria: E. Coli NissleComponents of Synthetic Genetic Circuit Phenylpyruvate (PP)
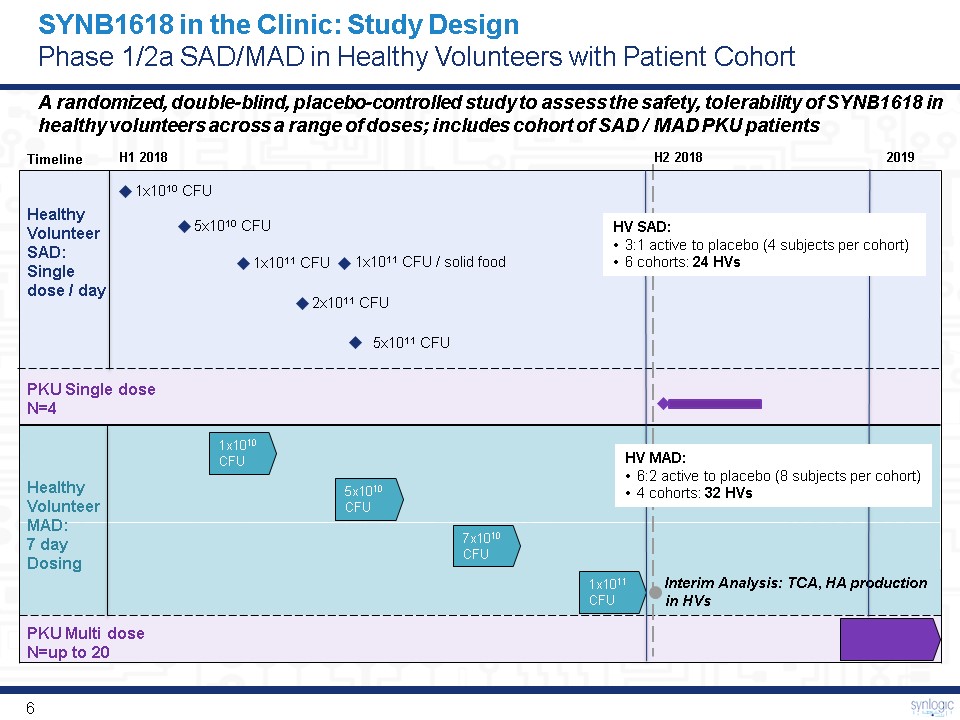
5x1010 CFU 7x1010 CFU 1x1010 CFU 1x1011 CFU 5x1011 CFU 5x1010 CFU 1x1010 CFU 1x1011 CFU 2x1011 CFU Timeline Interim Analysis: TCA, HA production in HVs 1x1011 CFU / solid food A randomized, double-blind, placebo-controlled study to assess the safety, tolerability of SYNB1618 in healthy volunteers across a range of doses; includes cohort of SAD / MAD PKU patients Healthy Volunteer SAD:Single dose / day PKU Single dose N=4 Healthy Volunteer MAD:7 day Dosing SYNB1618 in the Clinic: Study DesignPhase 1/2a SAD/MAD in Healthy Volunteers with Patient Cohort PKU Multi dose N=up to 20 H1 2018 H2 2018 2019 HV MAD:6:2 active to placebo (8 subjects per cohort)4 cohorts: 32 HVs HV SAD:3:1 active to placebo (4 subjects per cohort) 6 cohorts: 24 HVs

SYNB1618 in the Clinic: SafetyInterim Analysis of Phase 1/2a SAD/MAD Study Healthy Volunteer Cohorts The study enrolled 56 healthy volunteers, all of whom received at least one dose of SYNB1618 or placebo. The subjects were predominantly male Caucasians and the age range of enrolled subjects was 18-62 yearsThere were no treatment-related serious adverse events, no systemic toxicity or infections Treatment-emergent adverse events were either mild or moderate in severity, and reversible. Most AEs were GI-relatedAll subjects cleared the bacteria. There was no evidence of colonization, and no subject required antibioticsSingle dose MTD was defined as 2x1011 CFU. Doses above this level were associated with dose-limiting GI adverse eventsBased on pharmacodynamic data and tolerability profile a dose was identified for the second part of the study in PKU patients
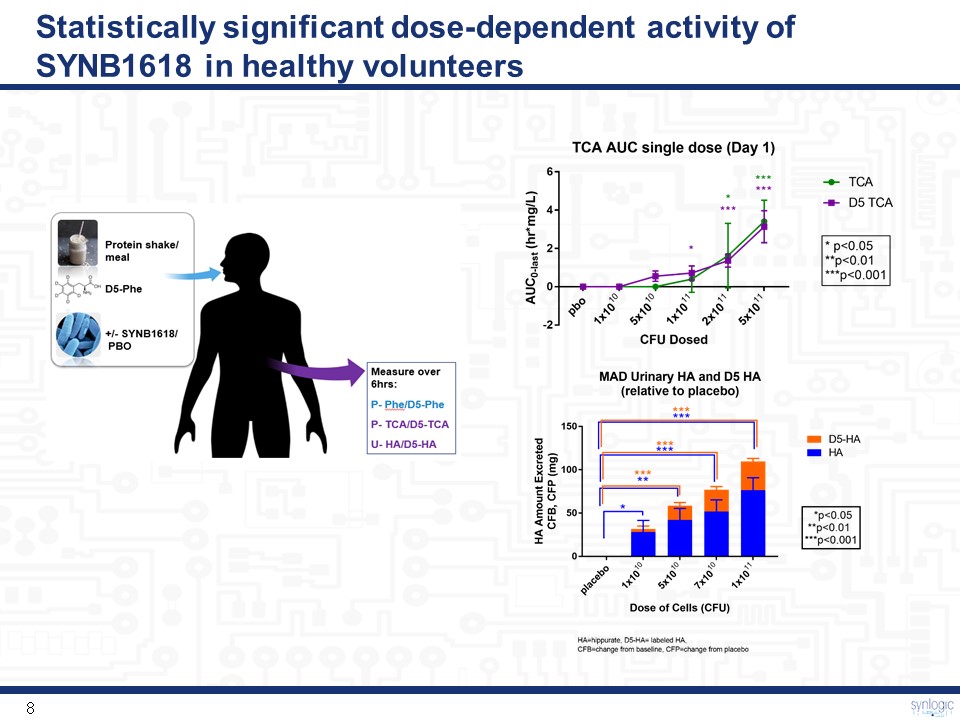
Statistically significant dose-dependent activity of SYNB1618 in healthy volunteers
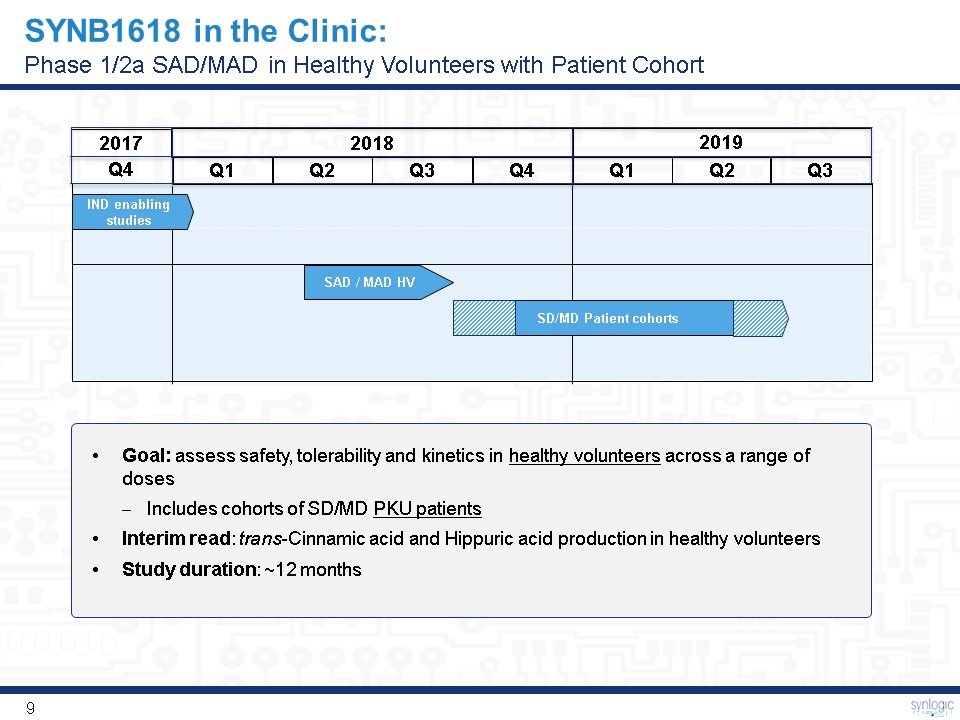
SYNB1618 in the Clinic:Phase 1/2a SAD/MAD in Healthy Volunteers with Patient Cohort 2017 2018 2019 Q1 Q2 Q3 Q4 Q1 Q2 IND enabling studies SD/MD Patient cohorts SAD / MAD HV Goal: assess safety, tolerability and kinetics in healthy volunteers across a range of dosesIncludes cohorts of SD/MD PKU patientsInterim read: trans-Cinnamic acid and Hippuric acid production in healthy volunteersStudy duration: ~12 months Q4 Q3
