Synlogic DESIGNED FOR LIFE June 2019 Corporate Presentation Exhibit 99.1

Forward Looking Statements This presentation contains “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this presentation regarding strategy, future operations, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward-looking statements. In addition, when or if used in this presentation, the words “may,” “could,” “should,” “anticipate,” “believe,” “estimate,” “expect,” “intend,” “plan,” “predict” and similar expressions and their variants may identify forward-looking statements. Examples of forward-looking statements include, but are not limited to, the approach we are taking to discover and develop novel therapeutics using synthetic biology; statements regarding the potential of our platform to develop therapeutics to address a wide range of diseases, including: inborn errors of metabolism, liver disease, inflammatory and immune disorders, and cancer; the future clinical development of Synthetic Biotic medicines; the potential of our technology to treat hyperammonemia and phenylketonuria; the expected timing of our anticipated clinical trial initiations; the benefit of orphan drug and fast track status; the adequacy of our capital to support our future operations and our ability to successfully initiate and complete clinical trials; the results of our collaborations; and the difficulty in predicting the time and cost of development of our product candidates. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: the uncertainties inherent in the preclinical development process; our ability to protect our intellectual property rights; and legislative, regulatory, political and economic developments, as well as those risks identified under the heading “Risk Factors” in our filings with the SEC. The foregoing review of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors included in our Annual Report on Form 10-K filed with the SEC on May 9, 2019. The forward-looking statements contained in this presentation reflect our current views with respect to future events. We anticipate that subsequent events and developments will cause our views to change. However, while we may elect to update these forward-looking statements in the future, we specifically disclaim any obligation to do so. These forward-looking statements should not be relied upon as representing our view as of any date subsequent to the date hereof.

Harnessing nature and technology to create LIVING medicines designed to significantly improve patients’ LIVES Synthetic BioticTM Medicines Designed For Life
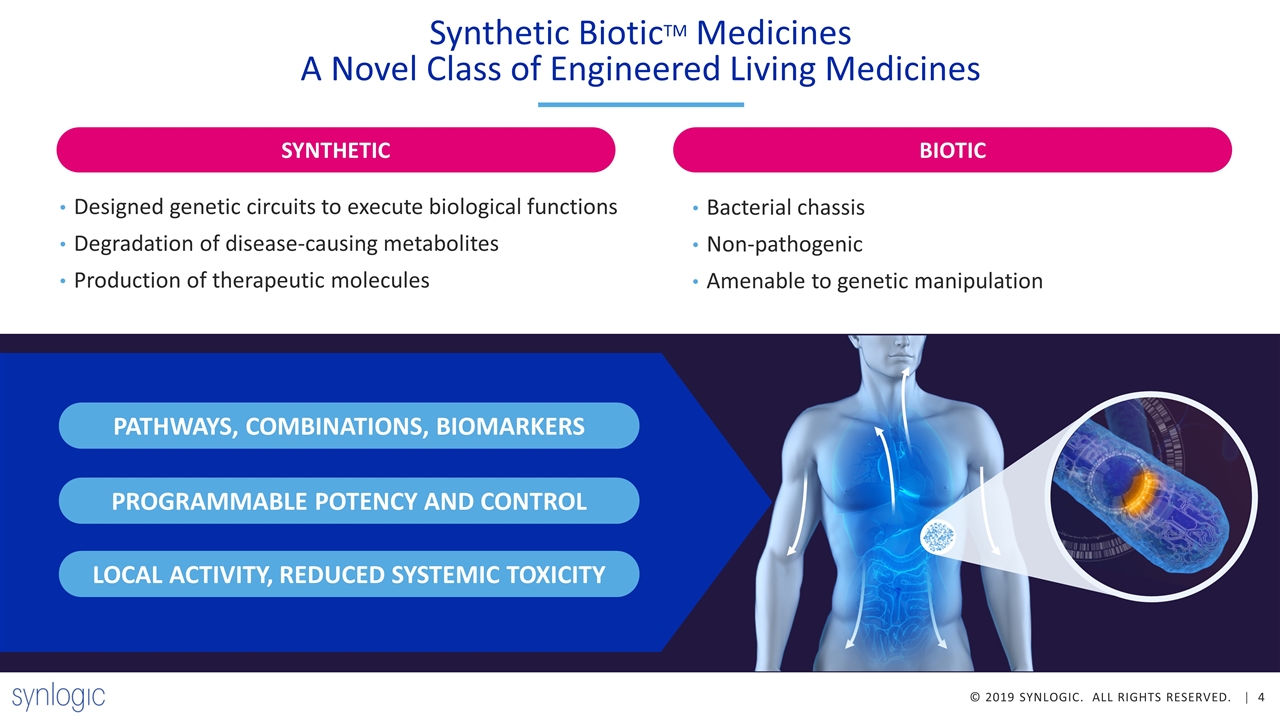
v Synthetic BioticTM Medicines A Novel Class of Engineered Living Medicines Designed genetic circuits to execute biological functions Degradation of disease-causing metabolites Production of therapeutic molecules Bacterial chassis Non-pathogenic Amenable to genetic manipulation PATHWAYS, COMBINATIONS, BIOMARKERS PROGRAMMABLE POTENCY AND CONTROL LOCAL ACTIVITY, REDUCED SYSTEMIC TOXICITY BIOTIC SYNTHETIC
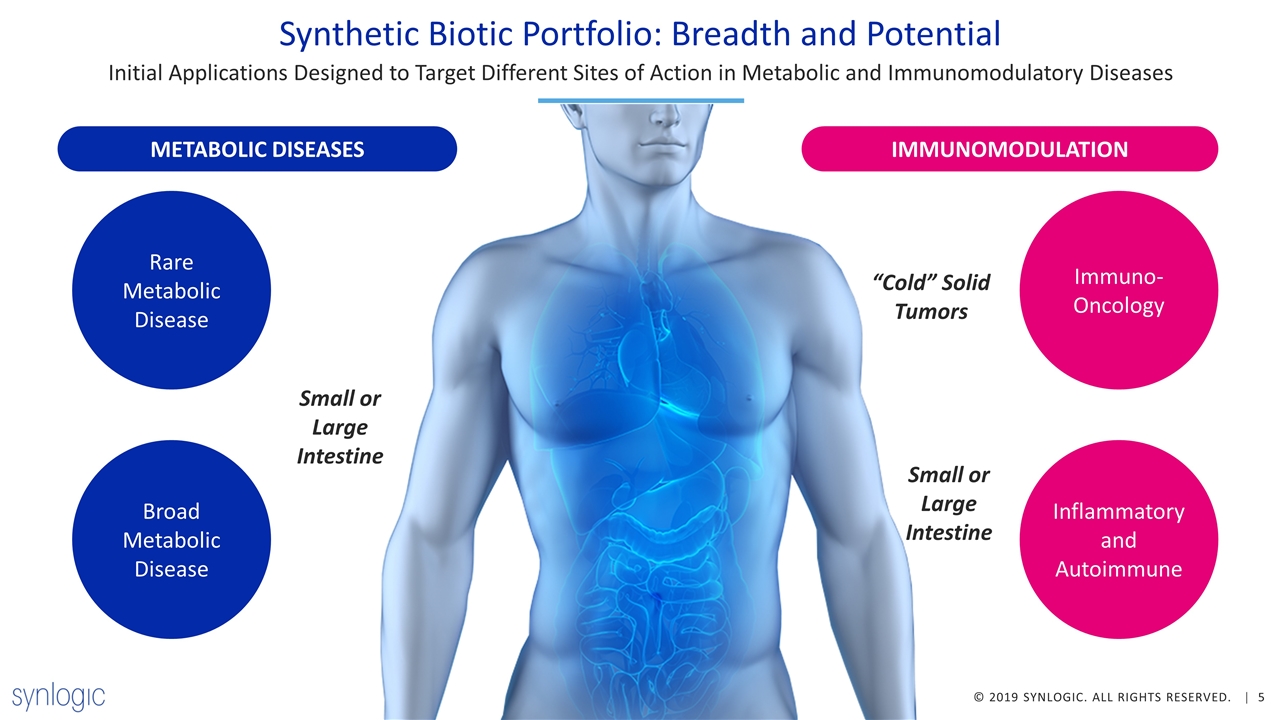
Synthetic Biotic Portfolio: Breadth and Potential Initial Applications Designed to Target Different Sites of Action in Metabolic and Immunomodulatory Diseases Oral Administration IMMUNOMODULATION METABOLIC DISEASES Immuno-Oncology Inflammatory and Autoimmune Small or Large Intestine “Cold” Solid Tumors Small or Large Intestine Rare Metabolic Disease Broad Metabolic Disease

Builds off validated pilot program initiated in 2017 Provides access to Ginkgo’s industrial scale, high-throughput strain optimization and screening Enables screening and identification of higher quality optimized candidates, increasing potential for success Delivers novel tools for increased candidate potency Includes equity investment at a premium, extending runway through multiple milestones Platform Collaboration to Accelerate Development of Synlogic’s Synthetic Biotic Medicines

Platform Collaboration to Accelerate Development of Synlogic’s Synthetic Biotic Medicines Industry leader in the construction and editing of microbial strains and organisms Leaders in non-therapeutic commercial applications of synthetic biology Comprehensive database of microbial genome sequences and unparalleled automated foundry Top-tier platform companies and collaborations High-quality investor base Rapid prototyping and screening enables efficient iteration through 1000’s of microbial strains
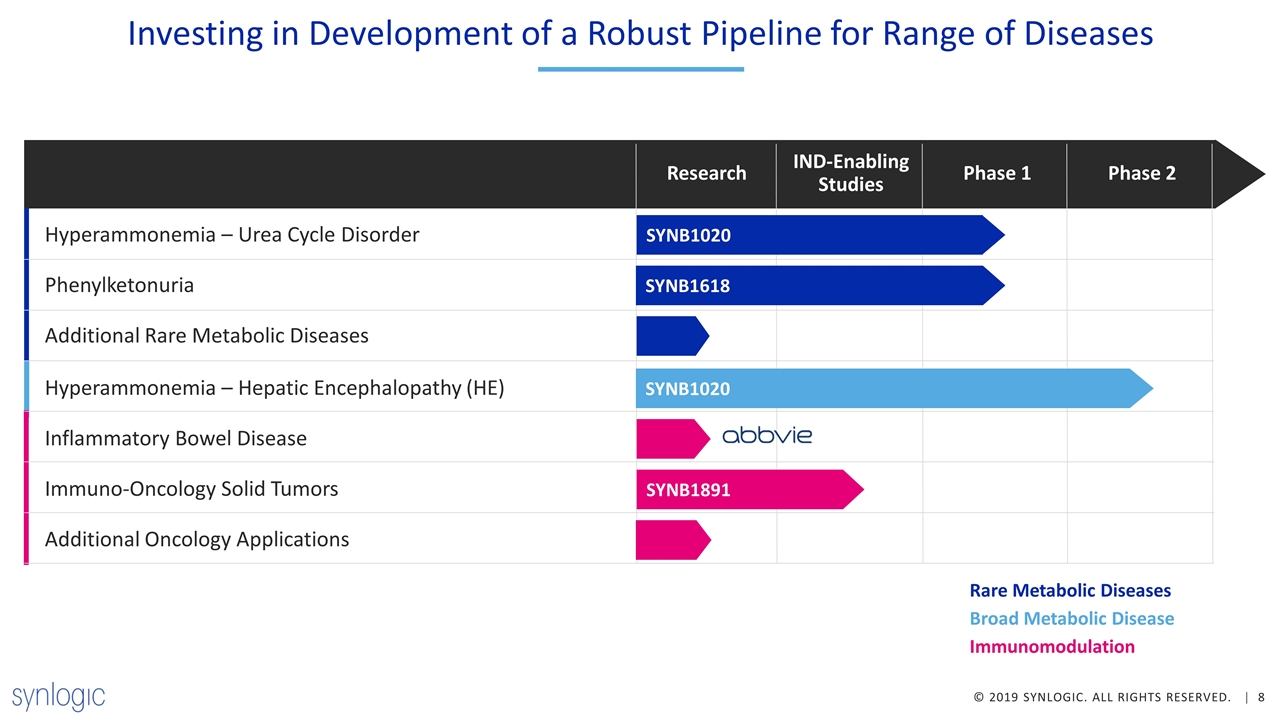
Investing in Development of a Robust Pipeline for Range of Diseases Hyperammonemia – Urea Cycle Disorder Research IND-Enabling Studies Phase 1 Phase 2 Phenylketonuria Additional Rare Metabolic Diseases Hyperammonemia – Hepatic Encephalopathy (HE) Inflammatory Bowel Disease Immuno-Oncology Solid Tumors Additional Oncology Applications SYNB1020 SYNB1618 SYNB1020 SYNB1891 Rare Metabolic Diseases Broad Metabolic Disease Immunomodulation

UREA CYCLE DISORDERS (UCD) SYNB1020 for Hyperammonemia Indications Characterized by Systemic Ammonia Accumulation Neuropsychiatric complication in patients with end-stage liver disease (cirrhosis) Liver dysfunction leads to ammonia accumulation Toxic to brain, leading to HE crisis & hospitalization Patients: 165,000 diagnosed overt patients in US Up to 70% of patients with cirrhosis characterized as covert (subclinical) Treatment: Lactulose: laxative with significant side effects Rifaximin: reduction in overt HE recurrence Genetic defects in Urea Cycle Deficiency in one of the six enzymes Nitrogen accumulates as toxic ammonia leading to metabolic crisis Patients: ~2,000 diagnosed in US; similar in EU Treatment: Ammonia scavengers: Buphenyl® (sodium phenylbutyrate), Ravicti® (glycerol phenylbuterate) Low protein diet with amino acid supplements Target Profile to Address Unmet Need: Reduce episodes of hospitalization Improve cognitive outcomes, Quality of Life Target Profile to Address Unmet Need: Maintain blood ammonia in normal range, avoid crisis Protein liberalization: 50-100% more per day Oral administration HEPATIC ENCEPHALOPATHY (HE)
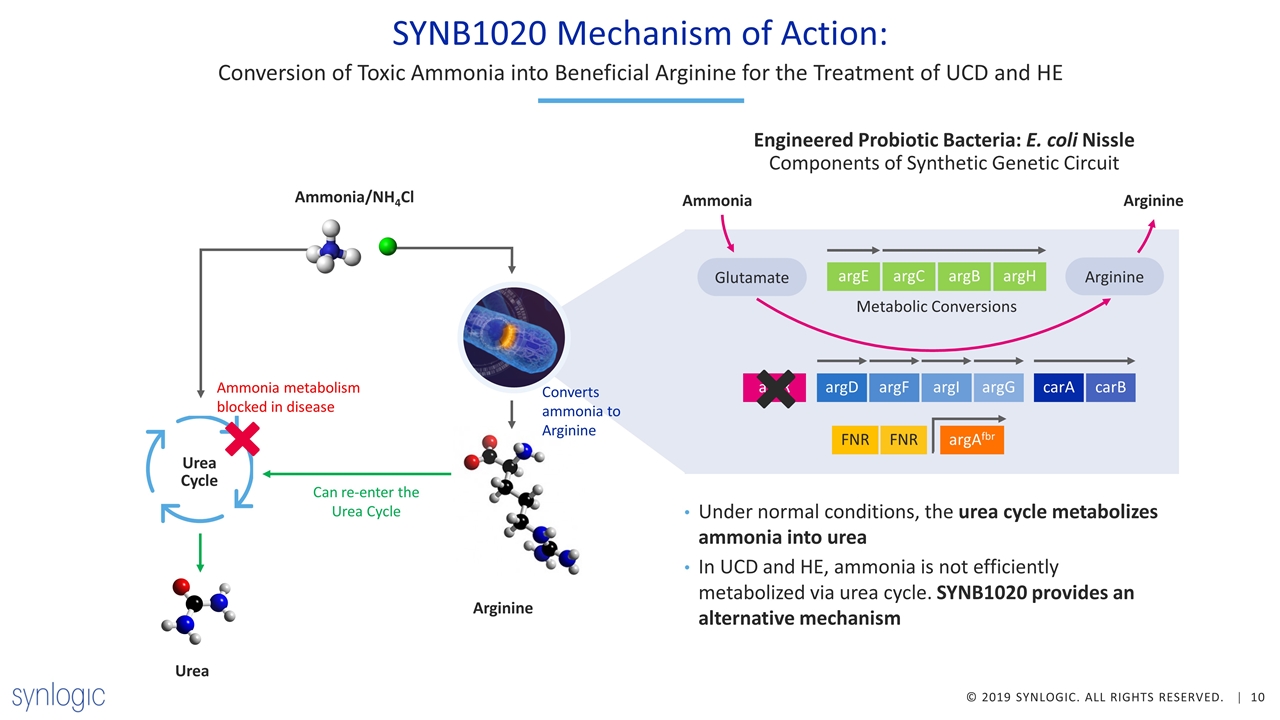
Arginine Urea SYNB1020 Mechanism of Action: Under normal conditions, the urea cycle metabolizes ammonia into urea In UCD and HE, ammonia is not efficiently metabolized via urea cycle. SYNB1020 provides an alternative mechanism Conversion of Toxic Ammonia into Beneficial Arginine for the Treatment of UCD and HE Ammonia/NH4Cl Urea Cycle Ammonia Arginine Arginine argD argF argI argG carA carB argR argE argC argB argH Metabolic Conversions FNR FNR argAfbr Glutamate Converts ammonia to Arginine Engineered Probiotic Bacteria: E. coli Nissle Components of Synthetic Genetic Circuit Can re-enter the Urea Cycle Ammonia metabolism blocked in disease
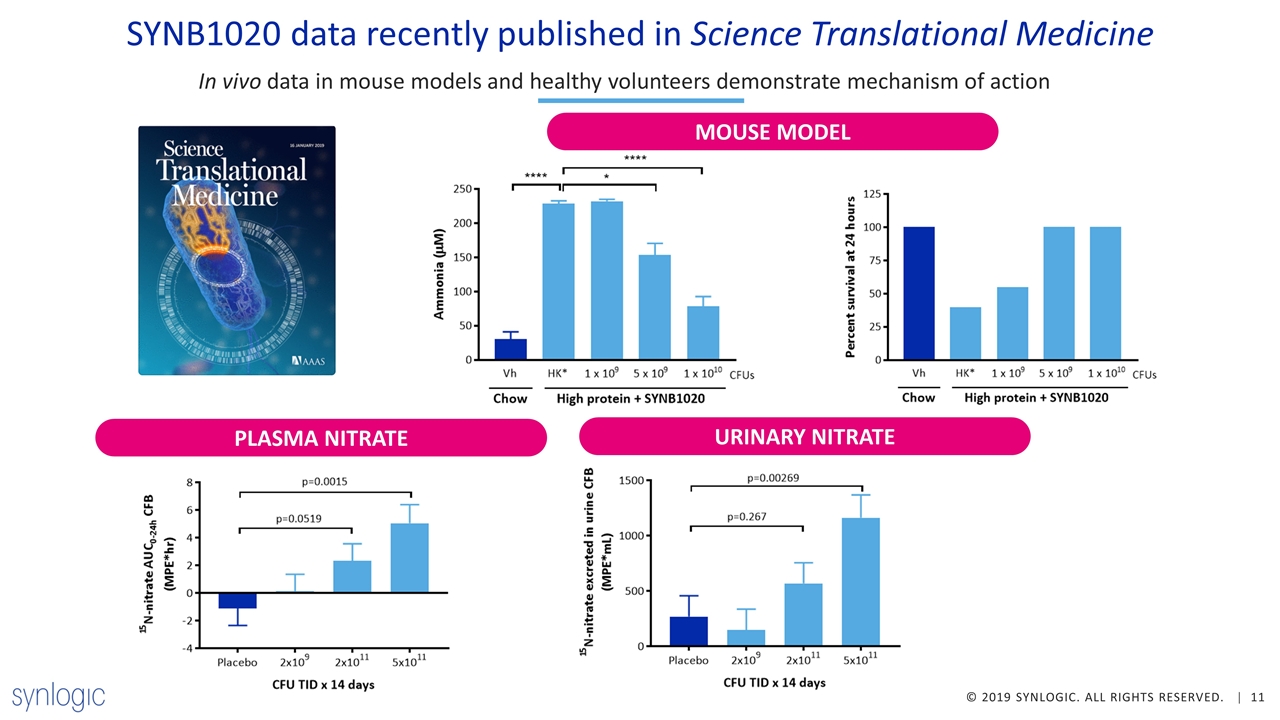
In vivo data in mouse models and healthy volunteers demonstrate mechanism of action SYNB1020 data recently published in Science Translational Medicine PLASMA NITRATE URINARY NITRATE MOUSE MODEL
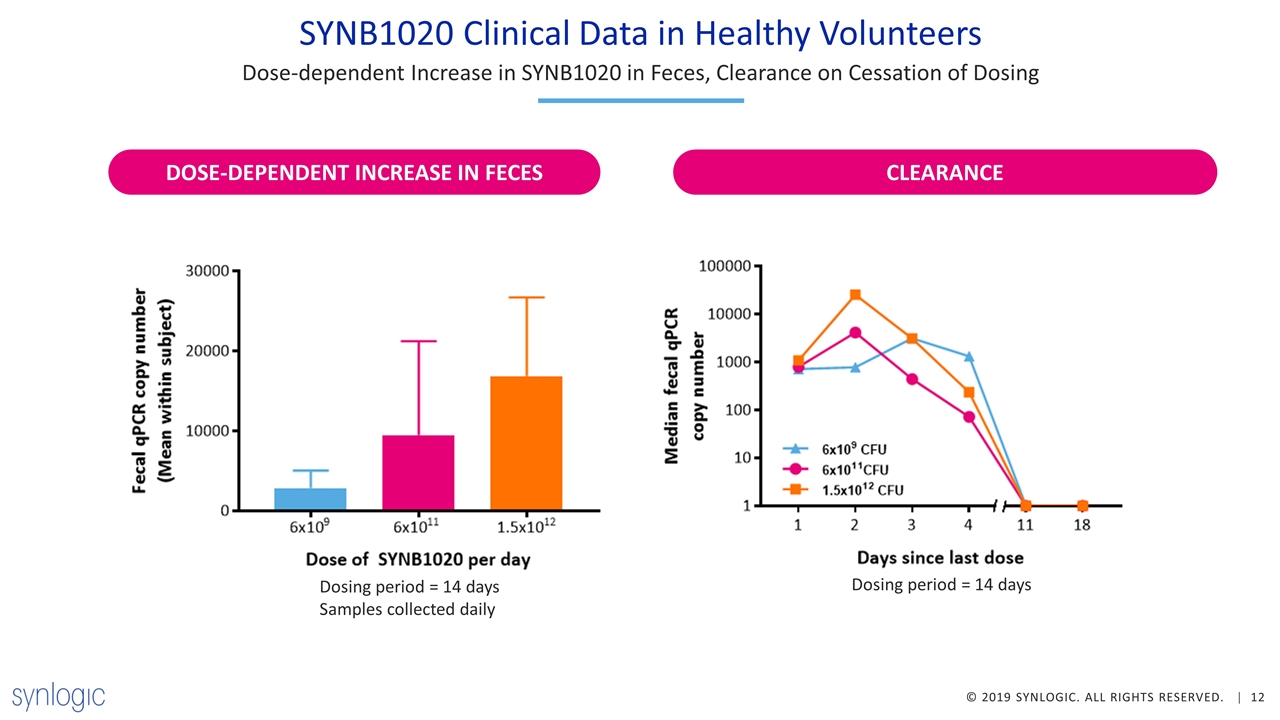
SYNB1020 Clinical Data in Healthy Volunteers Dose-dependent Increase in SYNB1020 in Feces, Clearance on Cessation of Dosing DOSE-DEPENDENT INCREASE IN FECES CLEARANCE Dosing period = 14 days Dosing period = 14 days Samples collected daily
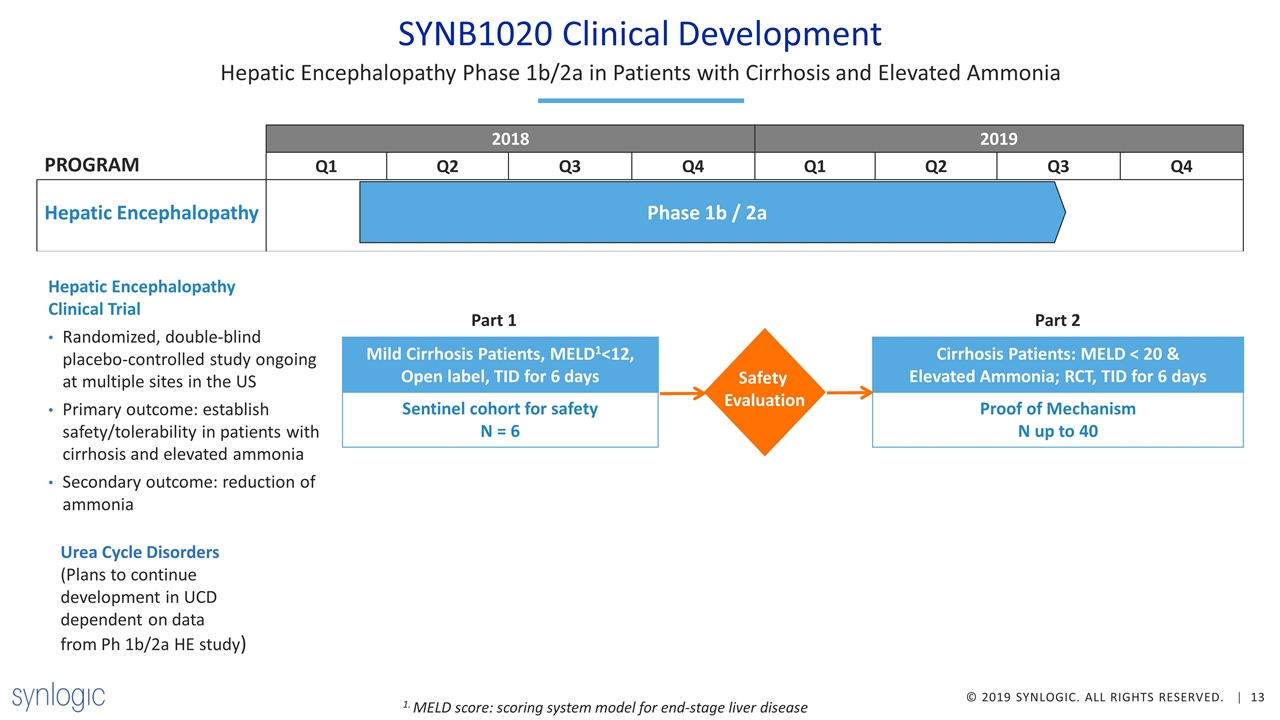
SYNB1020 Clinical Development Hepatic Encephalopathy Phase 1b/2a in Patients with Cirrhosis and Elevated Ammonia Hepatic Encephalopathy Clinical Trial Randomized, double-blind placebo-controlled study ongoing at multiple sites in the US Primary outcome: establish safety/tolerability in patients with cirrhosis and elevated ammonia Secondary outcome: reduction of ammonia Phase 1b / 2a PROGRAM Hepatic Encephalopathy Mild Cirrhosis Patients, MELD1<12, Open label, TID for 6 days Sentinel cohort for safety N = 6 Cirrhosis Patients: MELD < 20 & Elevated Ammonia; RCT, TID for 6 days Proof of Mechanism N up to 40 Safety Evaluation Part 1 Part 2 1. MELD score: scoring system model for end-stage liver disease Urea Cycle Disorders (Plans to continue development in UCD dependent on data from Ph 1b/2a HE study)

PKU is a rare inherited amino acid metabolism disorder Causes build up of amino acid phenylalanine (Phe) in the body Today, less than half of adults are at or below target Phe levels of 120-360 mmol / L If left untreated, symptoms include cognitive impairment, convulsions, behavioral problems, skin rash Patients: 16,500 diagnosed in US, similar in EU5 Treatment: Phenylalanine is found in all proteins therefore low protein diet is followed (no meat, dairy, nuts, eggs) KUVAN® (sapropterin dihydrochloride): PAH cofactor. 20-40% of patients are responders PalynziqTM (pegvaliase-pqpz): injectable, pegylated, bacterial enzyme (phenylalanine ammonia-lyase or PAL) for treatment of adult patients SYNB1618 for Phenylketonuria (PKU) Target Profile to Address Unmet Need: Manage Phe below target levels to prevent irreversible cognitive damage Increase natural protein intake: classic PKU patients’ natural protein intake is typically less than 10g Oral dosing without systemic toxicity Goal: Managing Plasma Phe Levels
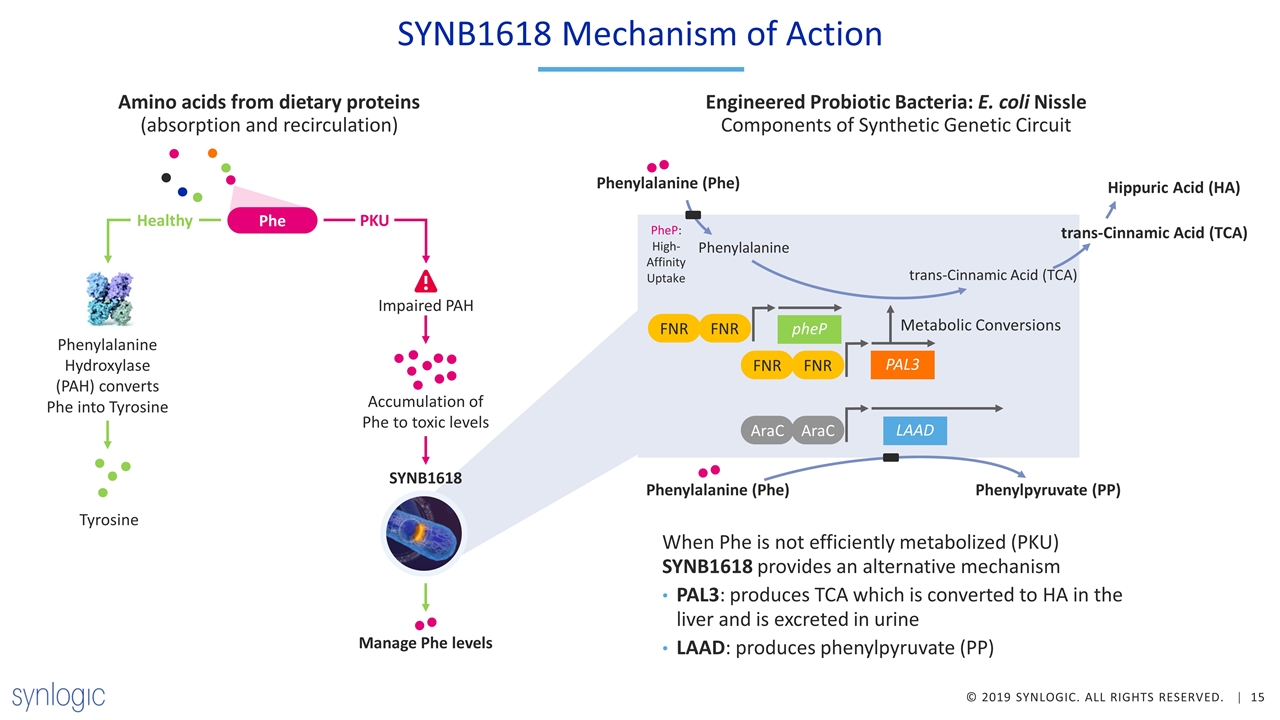
SYNB1618 Mechanism of Action Amino acids from dietary proteins (absorption and recirculation) Phe PKU Healthy Phenylalanine Hydroxylase (PAH) converts Phe into Tyrosine Tyrosine Accumulation of Phe to toxic levels Impaired PAH SYNB1618 Manage Phe levels When Phe is not efficiently metabolized (PKU) SYNB1618 provides an alternative mechanism PAL3: produces TCA which is converted to HA in the liver and is excreted in urine LAAD: produces phenylpyruvate (PP) Phenylalanine (Phe) Hippuric Acid (HA) Engineered Probiotic Bacteria: E. coli Nissle Components of Synthetic Genetic Circuit trans-Cinnamic Acid (TCA) PheP: High-Affinity Uptake trans-Cinnamic Acid (TCA) pheP PAL3 Metabolic Conversions FNR FNR FNR FNR Phenylalanine LAAD AraC AraC Phenylalanine (Phe) Phenylpyruvate (PP)
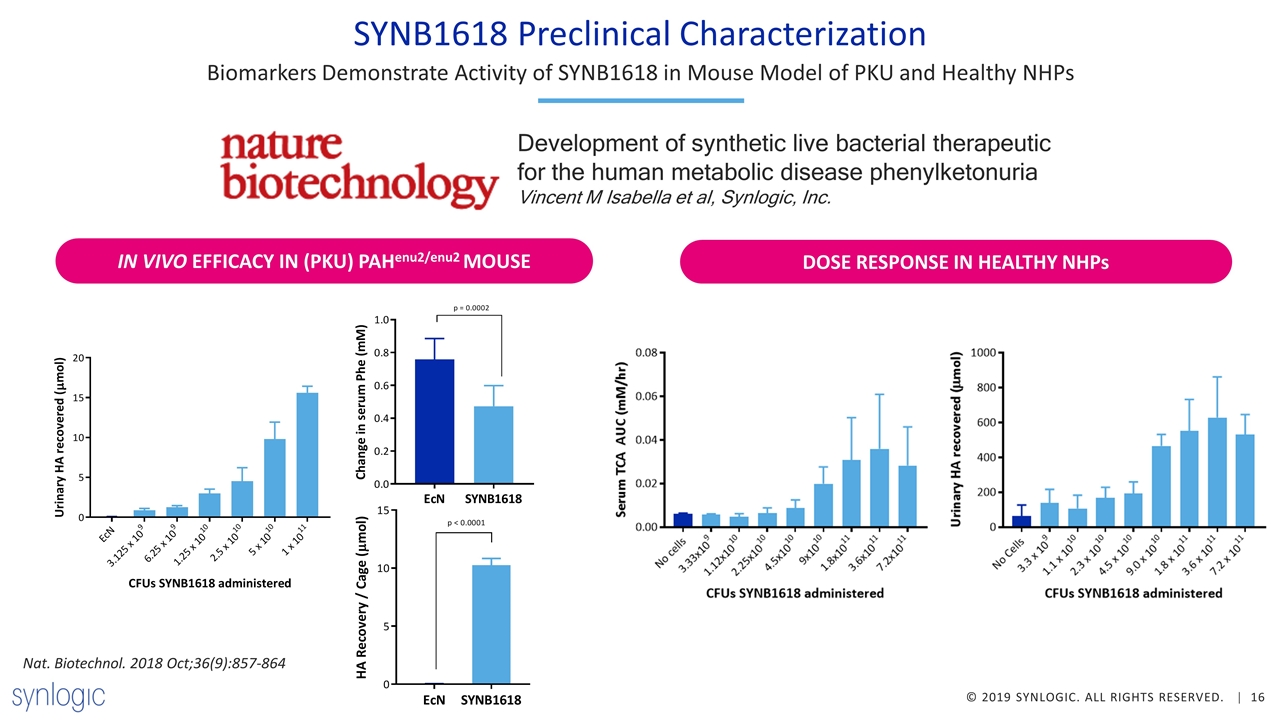
SYNB1618 Preclinical Characterization Biomarkers Demonstrate Activity of SYNB1618 in Mouse Model of PKU and Healthy NHPs IN VIVO EFFICACY IN (PKU) PAHenu2/enu2 MOUSE DOSE RESPONSE IN HEALTHY NHPs Nat. Biotechnol. 2018 Oct;36(9):857-864 Development of synthetic live bacterial therapeutic for the human metabolic disease phenylketonuria Vincent M Isabella et al, Synlogic, Inc.

SYNB1618 in the Clinic: Safety There were no treatment-related serious adverse events, no systemic toxicity or infections Treatment-emergent adverse events were either mild or moderate in severity, and reversible. Most adverse events were GI-related Single dose MTD was defined as 2x1011 CFU. Doses above this level were associated with dose-limiting GI adverse events All subjects cleared the bacteria. There was no evidence of colonization, and no subject required antibiotics Interim Analysis of Phase 1/2a SAD/MAD Study Demonstrates Safety and Clearance in Healthy Volunteers Based on pharmacodynamic data and tolerability profile, a dose of 7x1010 CFU was identified for the second part of the study in PKU patients 56 healthy volunteers Received at least one dose of SYNB1618 or placebo Adults Age range: 18-62 yrs old
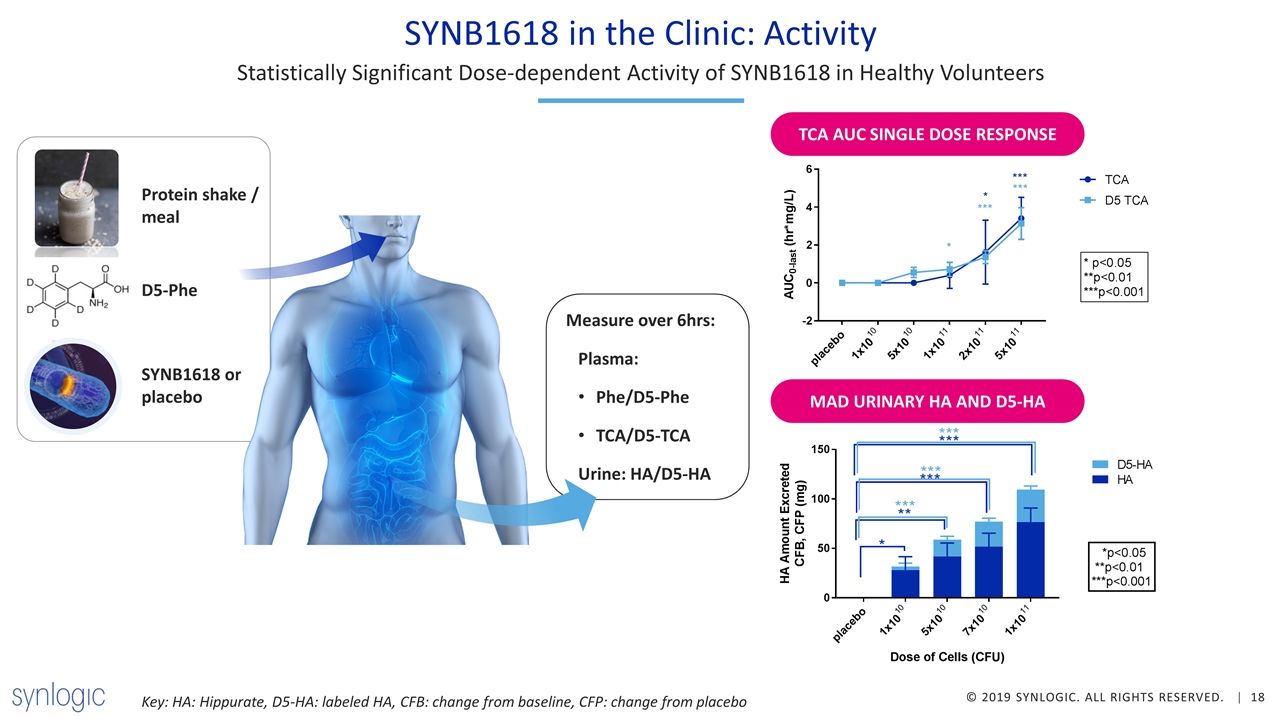
SYNB1618 in the Clinic: Activity Statistically Significant Dose-dependent Activity of SYNB1618 in Healthy Volunteers SYNB1618 or placebo Protein shake / meal D5-Phe Measure over 6hrs: Plasma: Phe/D5-Phe TCA/D5-TCA Urine: HA/D5-HA TCA AUC SINGLE DOSE RESPONSE MAD URINARY HA AND D5-HA Key: HA: Hippurate, D5-HA: labeled HA, CFB: change from baseline, CFP: change from placebo
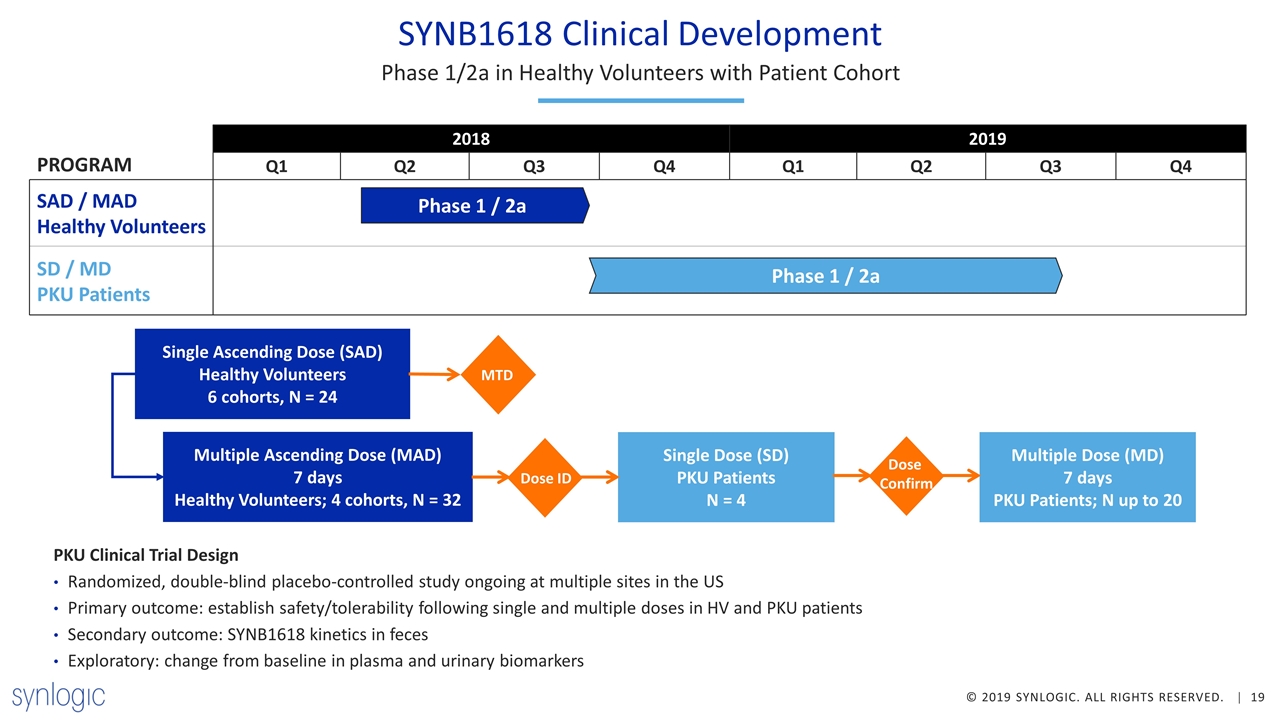
SYNB1618 Clinical Development Phase 1/2a in Healthy Volunteers with Patient Cohort Phase 1 / 2a Phase 1 / 2a SAD / MAD Healthy Volunteers SD / MD PKU Patients PROGRAM PKU Clinical Trial Design Randomized, double-blind placebo-controlled study ongoing at multiple sites in the US Primary outcome: establish safety/tolerability following single and multiple doses in HV and PKU patients Secondary outcome: SYNB1618 kinetics in feces Exploratory: change from baseline in plasma and urinary biomarkers Single Ascending Dose (SAD) Healthy Volunteers 6 cohorts, N = 24 Multiple Ascending Dose (MAD) 7 days Healthy Volunteers; 4 cohorts, N = 32 MTD Dose ID Single Dose (SD) PKU Patients N = 4 Multiple Dose (MD) 7 days PKU Patients; N up to 20 Dose Confirm

© 2019 SYNLOGIC. ALL RIGHTS RESERVED. | Immuno-Oncology
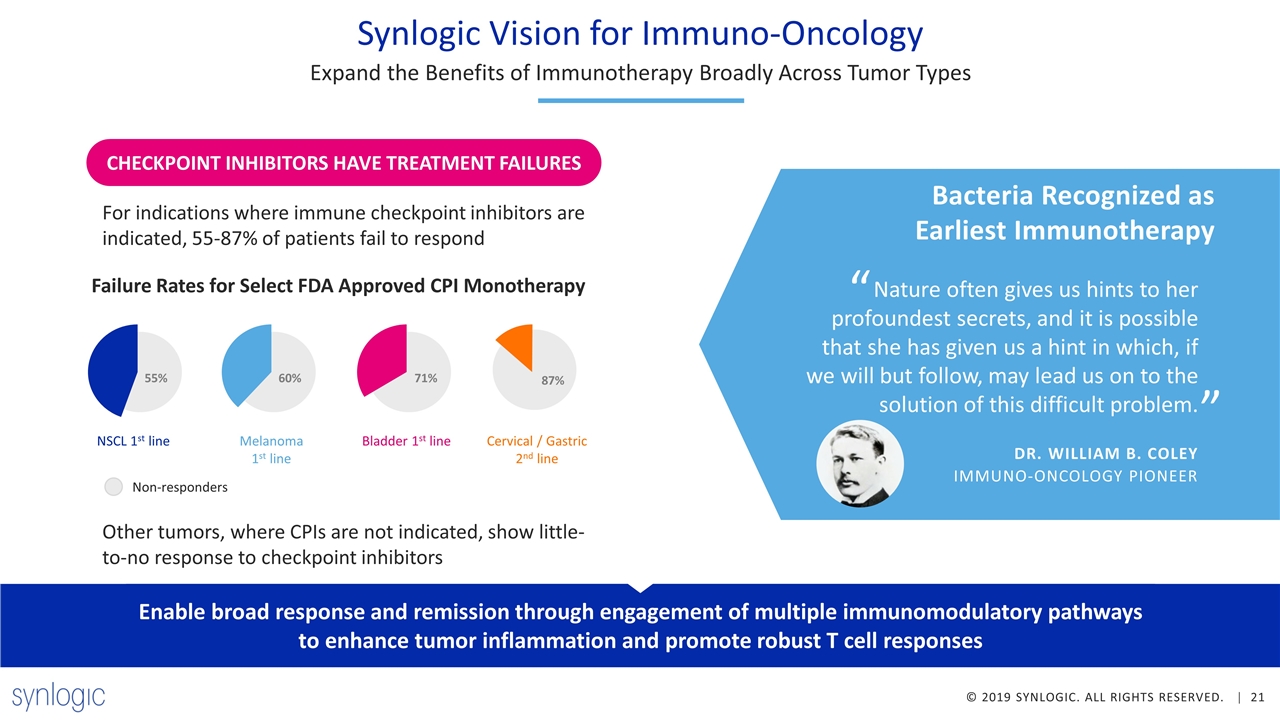
CHECKPOINT INHIBITORS HAVE TREATMENT FAILURES Synlogic Vision for Immuno-Oncology Expand the Benefits of Immunotherapy Broadly Across Tumor Types Other tumors, where CPIs are not indicated, show little-to-no response to checkpoint inhibitors Failure Rates for Select FDA Approved CPI Monotherapy 55% NSCL 1st line 60% Melanoma 1st line 71% Bladder 1st line 87% Cervical / Gastric 2nd line Non-responders For indications where immune checkpoint inhibitors are indicated, 55-87% of patients fail to respond Nature often gives us hints to her profoundest secrets, and it is possible that she has given us a hint in which, if we will but follow, may lead us on to the solution of this difficult problem. “ ” Bacteria Recognized as Earliest Immunotherapy DR. WILLIAM B. COLEY IMMUNO-ONCOLOGY PIONEER Enable broad response and remission through engagement of multiple immunomodulatory pathways to enhance tumor inflammation and promote robust T cell responses
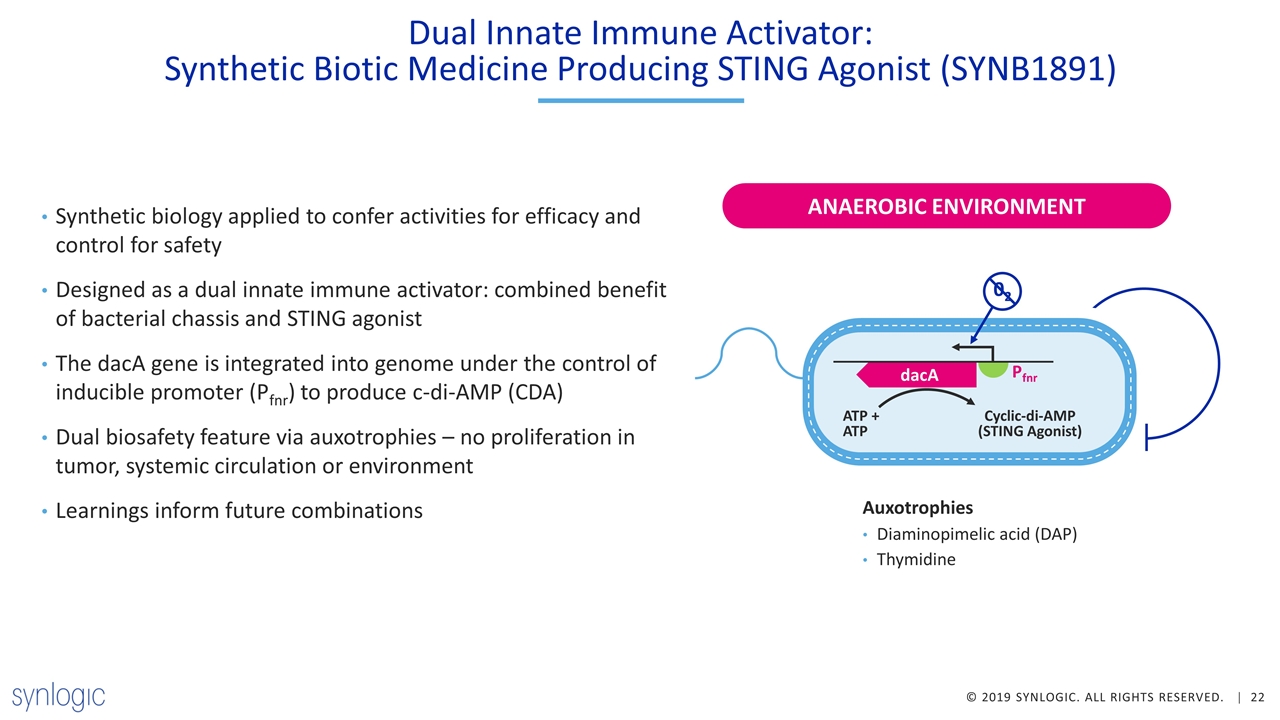
Dual Innate Immune Activator: Synthetic Biotic Medicine Producing STING Agonist (SYNB1891) Synthetic biology applied to confer activities for efficacy and control for safety Designed as a dual innate immune activator: combined benefit of bacterial chassis and STING agonist The dacA gene is integrated into genome under the control of inducible promoter (Pfnr) to produce c-di-AMP (CDA) Dual biosafety feature via auxotrophies – no proliferation in tumor, systemic circulation or environment Learnings inform future combinations ANAEROBIC ENVIRONMENT dacA Pfnr ATP + ATP Cyclic-di-AMP (STING Agonist) 02 Auxotrophies Diaminopimelic acid (DAP) Thymidine
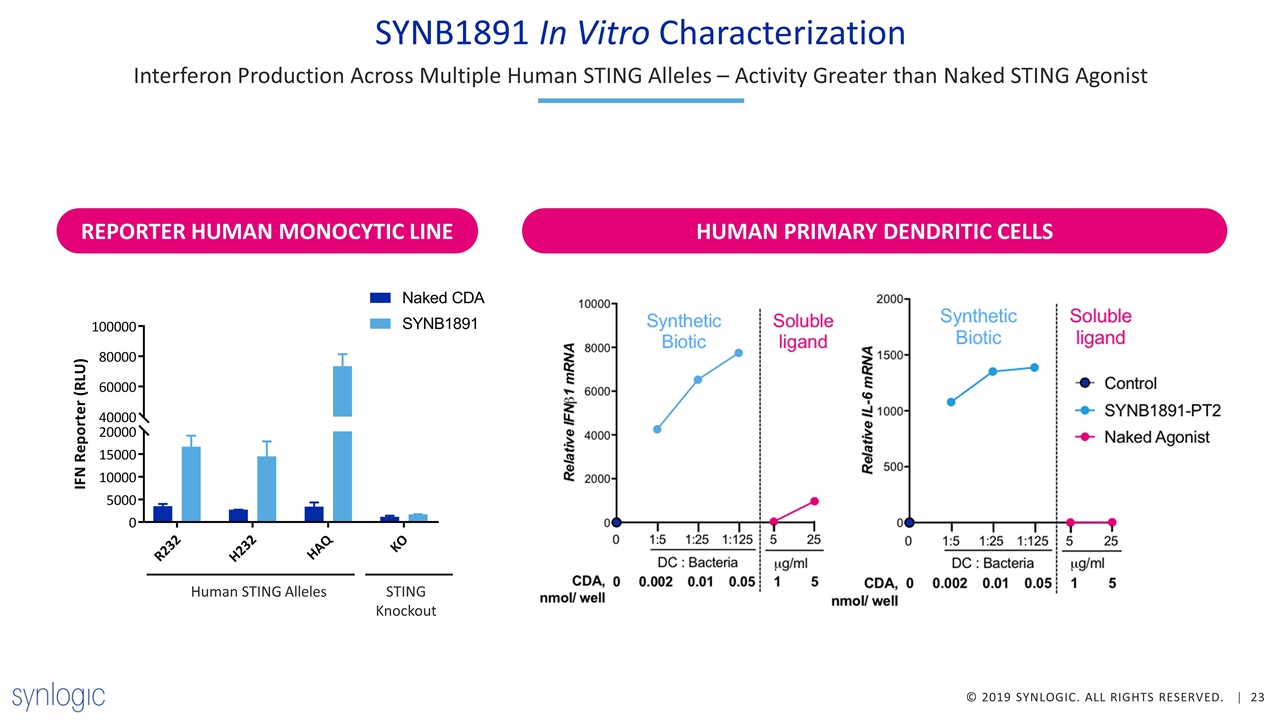
SYNB1891 In Vitro Characterization Interferon Production Across Multiple Human STING Alleles – Activity Greater than Naked STING Agonist HUMAN PRIMARY DENDRITIC CELLS REPORTER HUMAN MONOCYTIC LINE Human STING Alleles STING Knockout
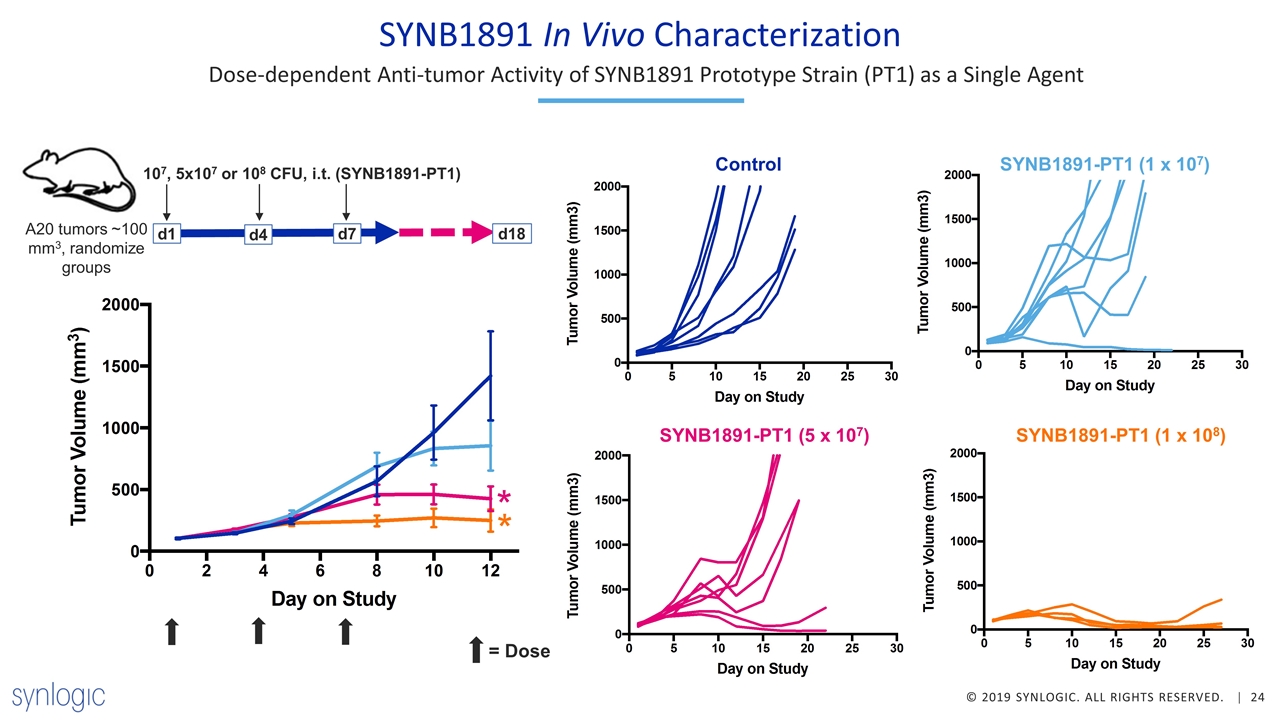
SYNB1891 In Vivo Characterization d1 107, 5x107 or 108 CFU, i.t. (SYNB1891-PT1) A20 tumors ~100 mm3, randomize groups d7 d4 d18 Dose-dependent Anti-tumor Activity of SYNB1891 Prototype Strain (PT1) as a Single Agent Control Control SYNB1891-PT1 (1 x 107) SYNB1891-PT1 (5 x 107) SYNB1891-PT1 (1 x 108) = Dose
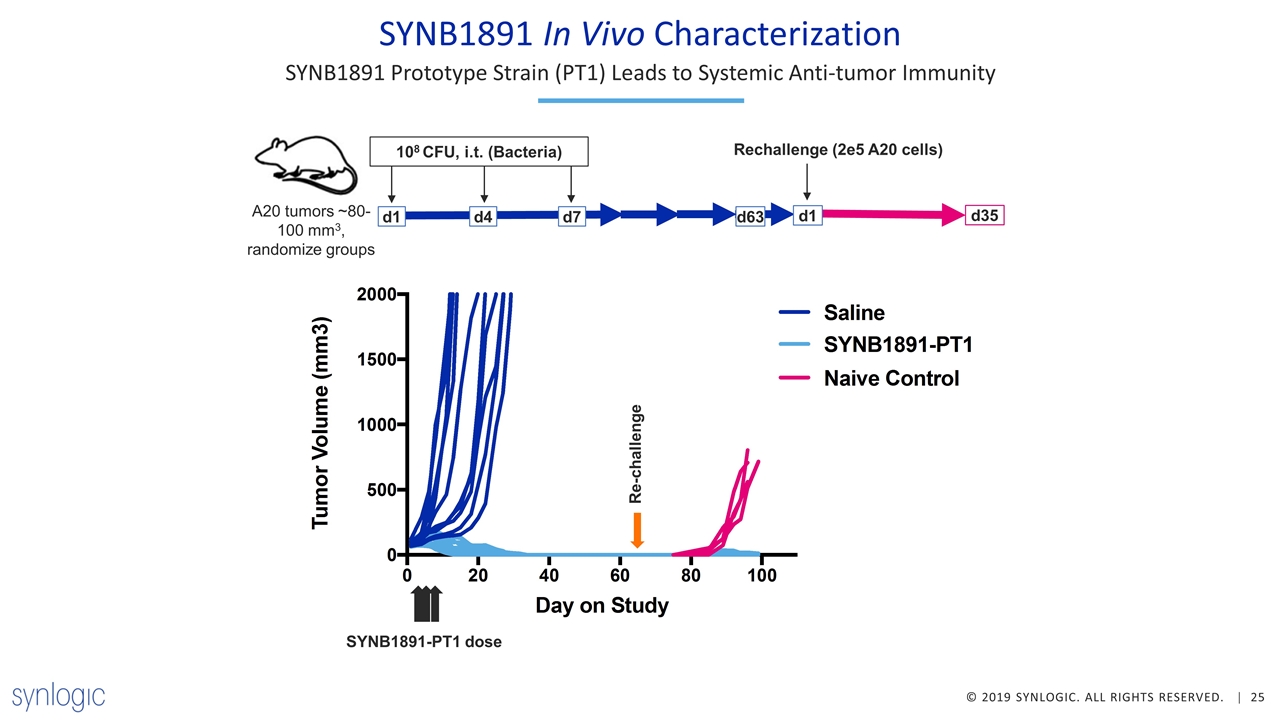
SYNB1891 In Vivo Characterization SYNB1891 Prototype Strain (PT1) Leads to Systemic Anti-tumor Immunity SYNB1891-PT1 dose Re-challenge d1 108 CFU, i.t. (Bacteria) A20 tumors ~80-100 mm3, randomize groups d7 d4 d35 d63 Rechallenge (2e5 A20 cells) d1
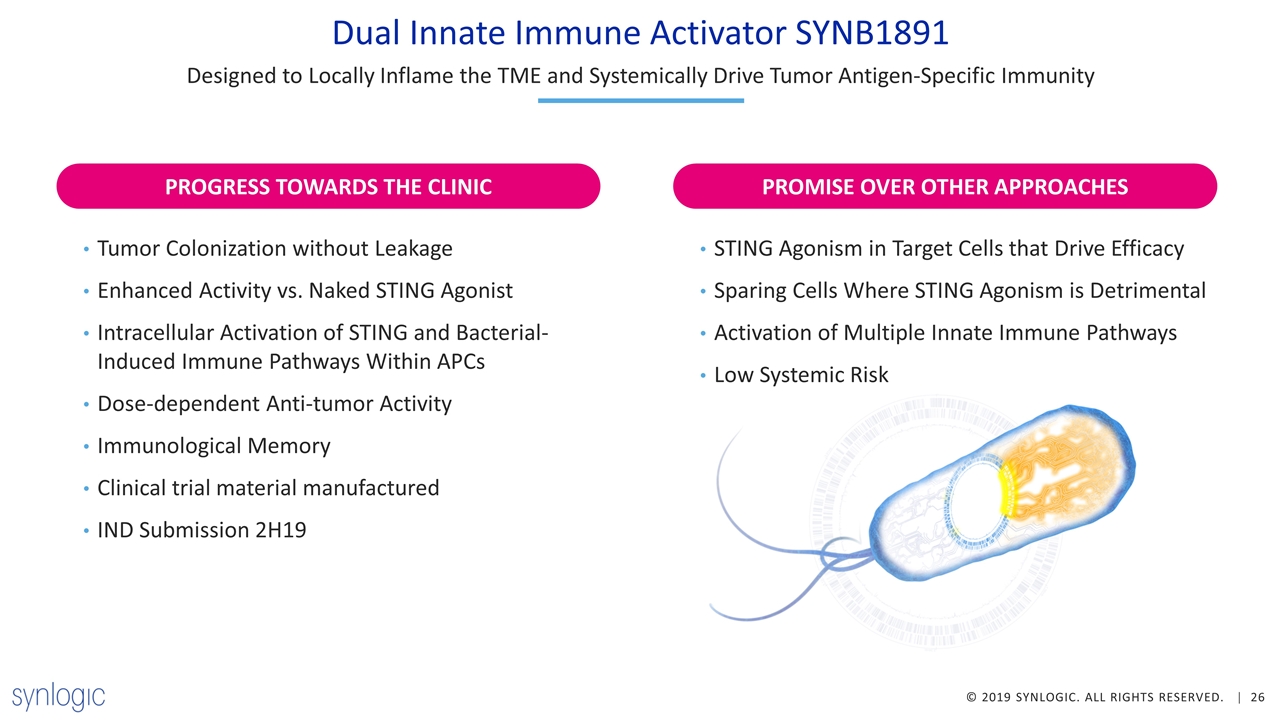
Dual Innate Immune Activator SYNB1891 Designed to Locally Inflame the TME and Systemically Drive Tumor Antigen-Specific Immunity Tumor Colonization without Leakage Enhanced Activity vs. Naked STING Agonist Intracellular Activation of STING and Bacterial-Induced Immune Pathways Within APCs Dose-dependent Anti-tumor Activity Immunological Memory Clinical trial material manufactured IND Submission 2H19 STING Agonism in Target Cells that Drive Efficacy Sparing Cells Where STING Agonism is Detrimental Activation of Multiple Innate Immune Pathways Low Systemic Risk PROMISE OVER OTHER APPROACHES PROGRESS TOWARDS THE CLINIC
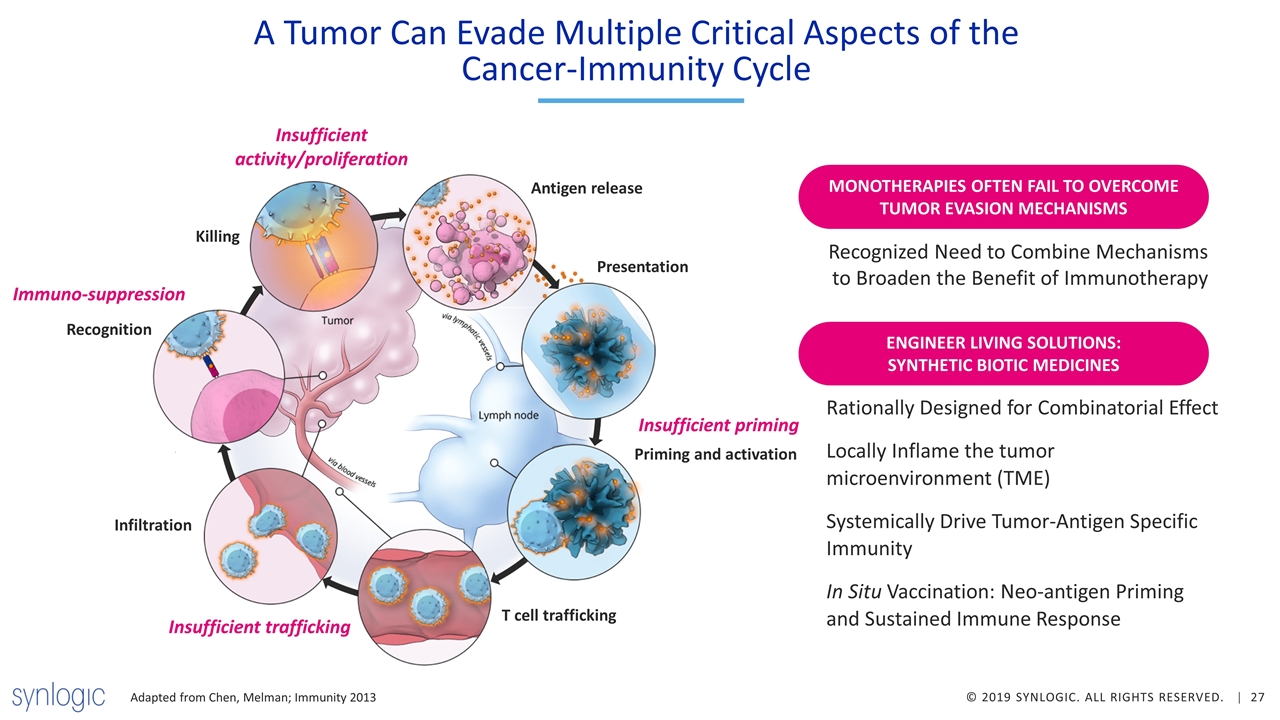
A Tumor Can Evade Multiple Critical Aspects of the Cancer-Immunity Cycle Killing Recognition Infiltration Antigen release Presentation Priming and activation T cell trafficking Insufficient activity/proliferation Immuno-suppression Insufficient trafficking Insufficient priming Recognized Need to Combine Mechanisms to Broaden the Benefit of Immunotherapy Adapted from Chen, Melman; Immunity 2013 MONOTHERAPIES OFTEN FAIL TO OVERCOME TUMOR EVASION MECHANISMS Rationally Designed for Combinatorial Effect Locally Inflame the tumor microenvironment (TME) Systemically Drive Tumor-Antigen Specific Immunity In Situ Vaccination: Neo-antigen Priming and Sustained Immune Response ENGINEER LIVING SOLUTIONS: SYNTHETIC BIOTIC MEDICINES
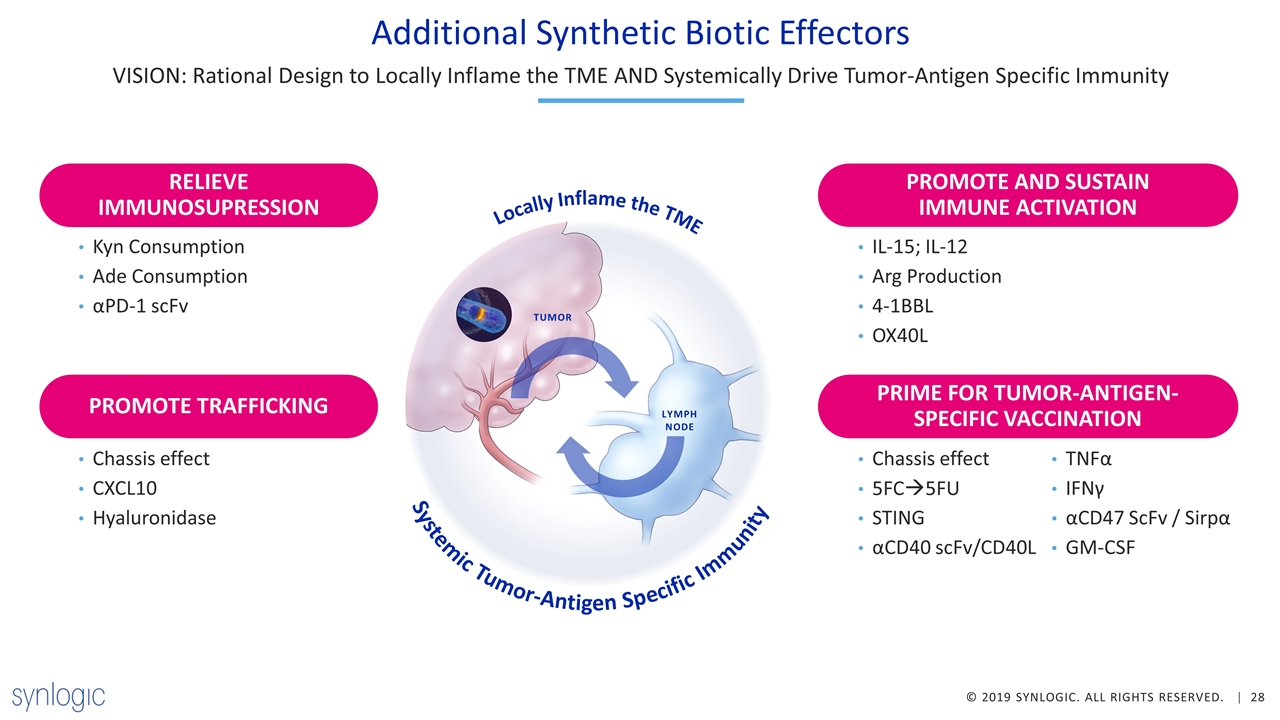
Additional Synthetic Biotic Effectors VISION: Rational Design to Locally Inflame the TME AND Systemically Drive Tumor-Antigen Specific Immunity Chassis effect CXCL10 Hyaluronidase Kyn Consumption Ade Consumption αPD-1 scFv Chassis effect 5FCà5FU STING αCD40 scFv/CD40L TNFα IFNγ αCD47 ScFv / Sirpα GM-CSF TUMOR LYMPH NODE Systemic Tumor-Antigen Specific Immunity Locally Inflame the TME IL-15; IL-12 Arg Production 4-1BBL OX40L RELIEVE IMMUNOSUPRESSION PROMOTE AND SUSTAIN IMMUNE ACTIVATION PRIME FOR TUMOR-ANTIGEN-SPECIFIC VACCINATION PROMOTE TRAFFICKING
Broad Ambitions in Immuno-Oncology SYNB1891 DISCOVERY PORTFOLIO INTRATUMORAL Vision: Expand and Exceed the Effect of Cancer Immunotherapies COMBINATIONS HARNESS THE MICROBIOME ORAL
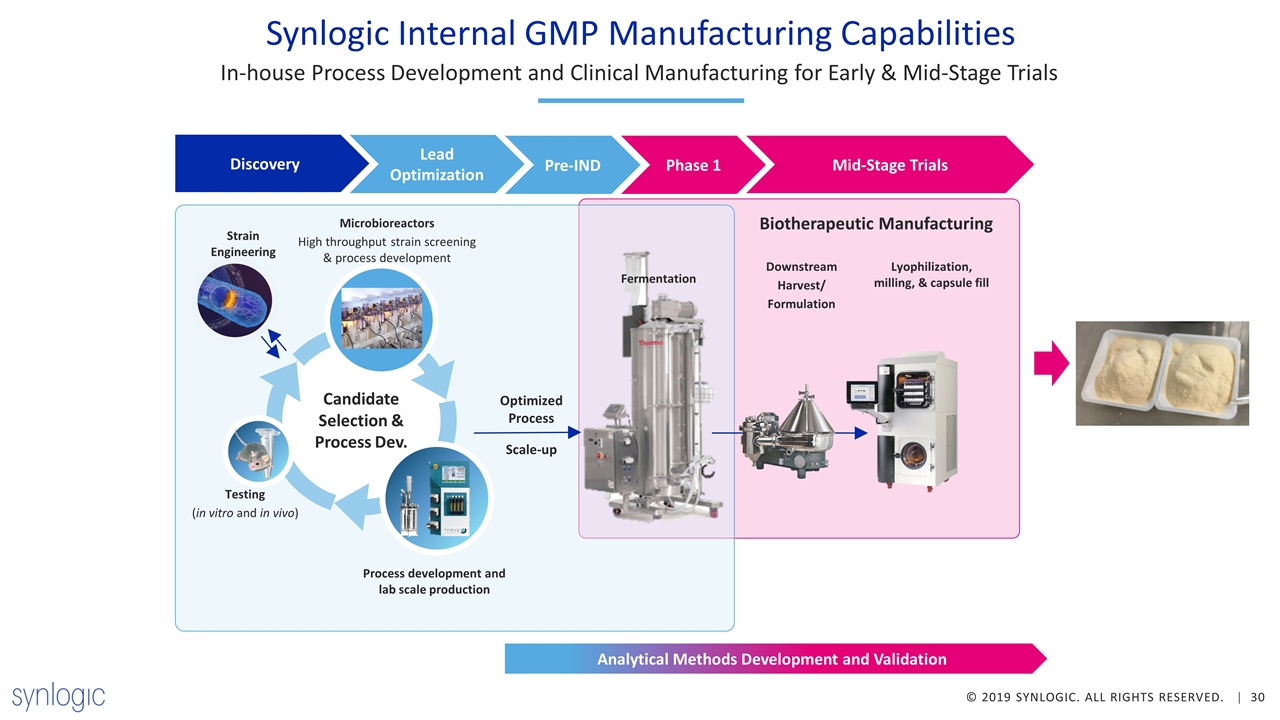
Synlogic Internal GMP Manufacturing Capabilities In-house Process Development and Clinical Manufacturing for Early & Mid-Stage Trials Biotherapeutic Manufacturing Candidate Selection & Process Dev. Testing (in vitro and in vivo) Strain Engineering Optimized Process Scale-up Process development and lab scale production Microbioreactors High throughput strain screening & process development Fermentation Downstream Harvest/ Formulation Lyophilization, milling, & capsule fill Discovery Lead Optimization Pre-IND Phase 1 Mid-Stage Trials Analytical Methods Development and Validation

Demonstrated Progress in Development of Lyophilized SYNB1618 Improved fermentation process enables production of a solid formulation of SYNB1618 with minimal impact on cell viability or activity Lyophilized SYNB1618 is similarly active to frozen liquid in terms of consumption of Phe or production of TCA/HA in vitro and in vivo New solid process material is expected to have improved quality attributes including less free protein and reduced viscosity Process is robust and reproducible at 30 L production scale Lyophilized SYNB1618 is stable for >90 days at 2-8 ◦C and >30 days at room temperature Suite build-out complete and ready to manufacture cGMP lyophilized SYNB1618
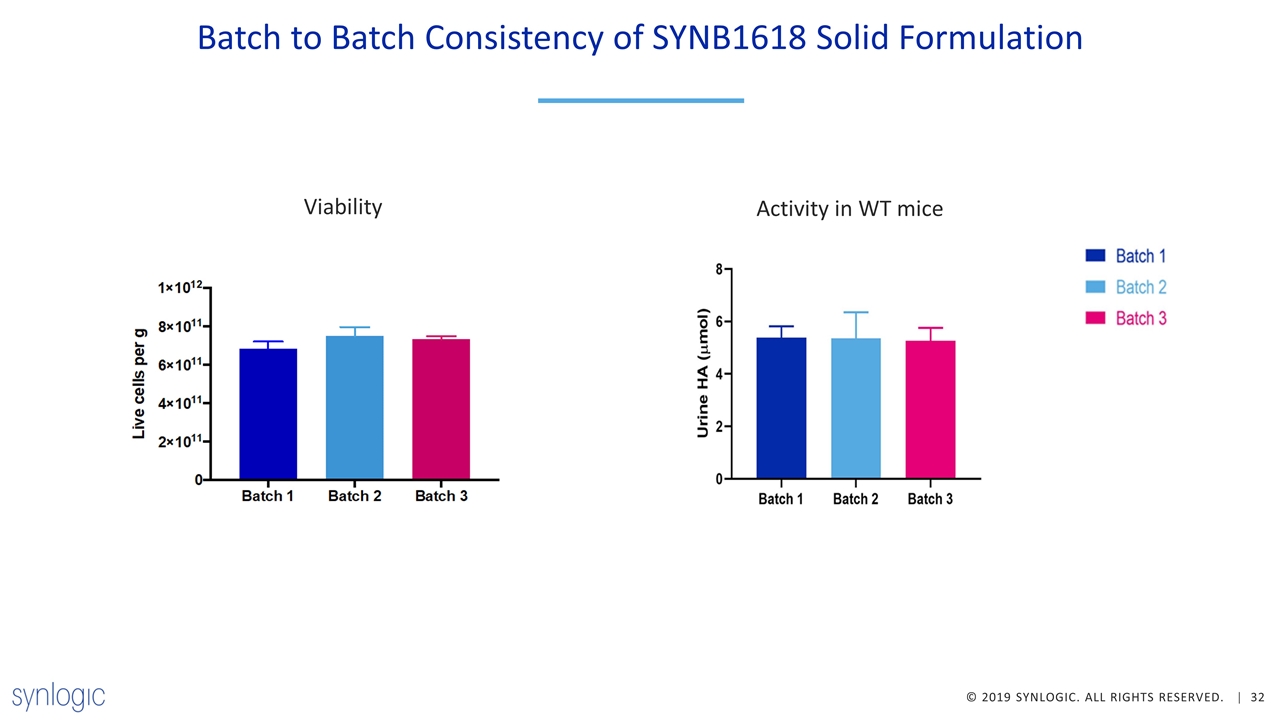
Batch to Batch Consistency of SYNB1618 Solid Formulation Viability Activity in WT mice
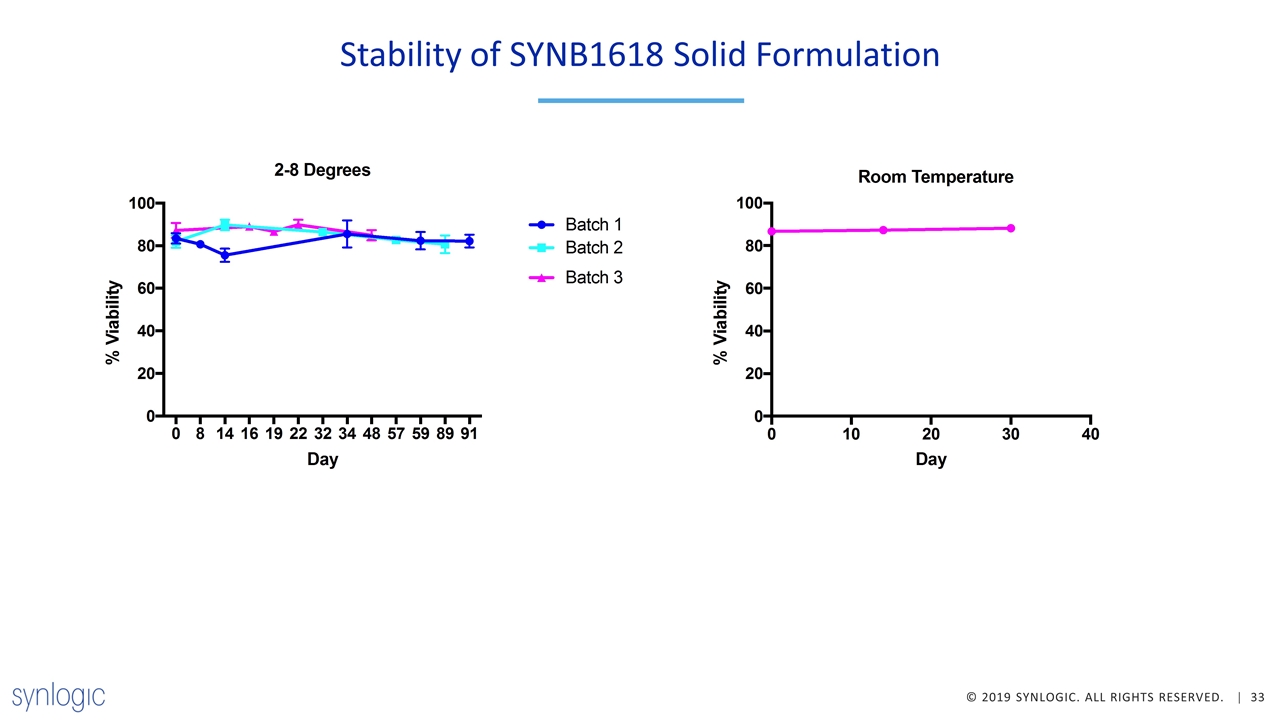
Stability of SYNB1618 Solid Formulation

2019 Progress and Milestones SYNB1618 in PKU Complete ongoing study in patients Data expected 3Q2019 (safety, tolerability and biomarkers) SYNB1020 in Hyperammonemia Preclin. and HV clin. data published in Sci. Transl. Med. Complete ongoing study in patients with cirrhosis Data expected 3Q2019 (safety, tolerability and ammonia-lowering) With ammonia-lowering data define development plan SYNB1891 in Immuno-Oncology IND submission 2H2019 Clinical trial material manufactured Advance AbbVie collaboration Advance preclinical pipeline

Harnessing nature and technology to create LIVING medicines designed to significantly improve patients’ LIVES Synthetic BioticTM Medicines Designed For Life

301 BINNEY ST., #402, CAMBRIDGE, MA 02142 TEL: 617-401-9975 WEB: WWW.SYNLOGICTX.COM | EMAIL: INFO@SYNLOGICTX.COM © SYNLOGIC. CONFIDENTIAL. ALL RIGHTS RESERVED.