
Transforming Medicine through Synthetic Biology Q4 and Full Year 2021 Financial Results & Business Update March 17, 2022 Exhibit 99.2

Forward Looking Statements This presentation contains "forward-looking statements" that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this press release regarding strategy, future operations, clinical development plans, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward-looking statements. In addition, when or if used in this press release, the words "may," "could," "should," "anticipate," "believe," "estimate," "expect," "intend," "plan," "predict" and similar expressions and their variants, as they relate to Synlogic may identify forward-looking statements. Examples of forward-looking statements, include, but are not limited to, statements regarding the potential of Synlogic's approach to Synthetic Biotics to develop therapeutics to address a wide range of diseases including: inborn errors of metabolism, and inflammatory and immune disorders; our expectations about sufficiency of our existing cash balance; the future clinical development of Synthetic Biotics; the approach Synlogic is taking to discover and develop novel therapeutics using synthetic biology; and the expected timing of Synlogic's clinical trials of SYNB1618, SYNB1934, SYNB1353 and SYNB8802 and availability of clinical trial data. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including: the uncertainties inherent in the clinical and preclinical development process; the ability of Synlogic to protect its intellectual property rights; and legislative, regulatory, political and economic developments, as well as those risks identified under the heading "Risk Factors" in Synlogic's filings with the SEC. The forward-looking statements contained in this press release reflect Synlogic's current views with respect to future events. Synlogic anticipates that subsequent events and developments will cause its views to change. However, while Synlogic may elect to update these forward-looking statements in the future, Synlogic specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing Synlogic's view as of any date subsequent to the date hereof..

© 2020 SYNLOGIC. QU©AR2T0E1R9LYSYRNELSOUGLTICS.. AALLLL RRIIGGHHTTSS RREESSEERRVVEEDD.. || 33 Opening Remarks Dr. Aoife Brennan MB CHB President & CEO

PKU Proof of concept from Phase 2 interim analysis with lead candidate PKU Next generation candidate showed greater potency HCU 2nd rare metabolic disease drug candidate entered pre-IND studies HOX Achieved proof of mechanism in healthy volunteers IBD Established Roche collaboration, achieved first research milestone PKU Phase 2 data Æ H1 2022 PKU Phase 3 initiation Æ H2 2022 HCU Phase 1 data from healthy volunteers Æ H2 2022 Enteric hyperoxaluria proof of concept Æ 2022 Multiple Anticipated Milestones as PKU Program Approaches Phase 3 2021 2022
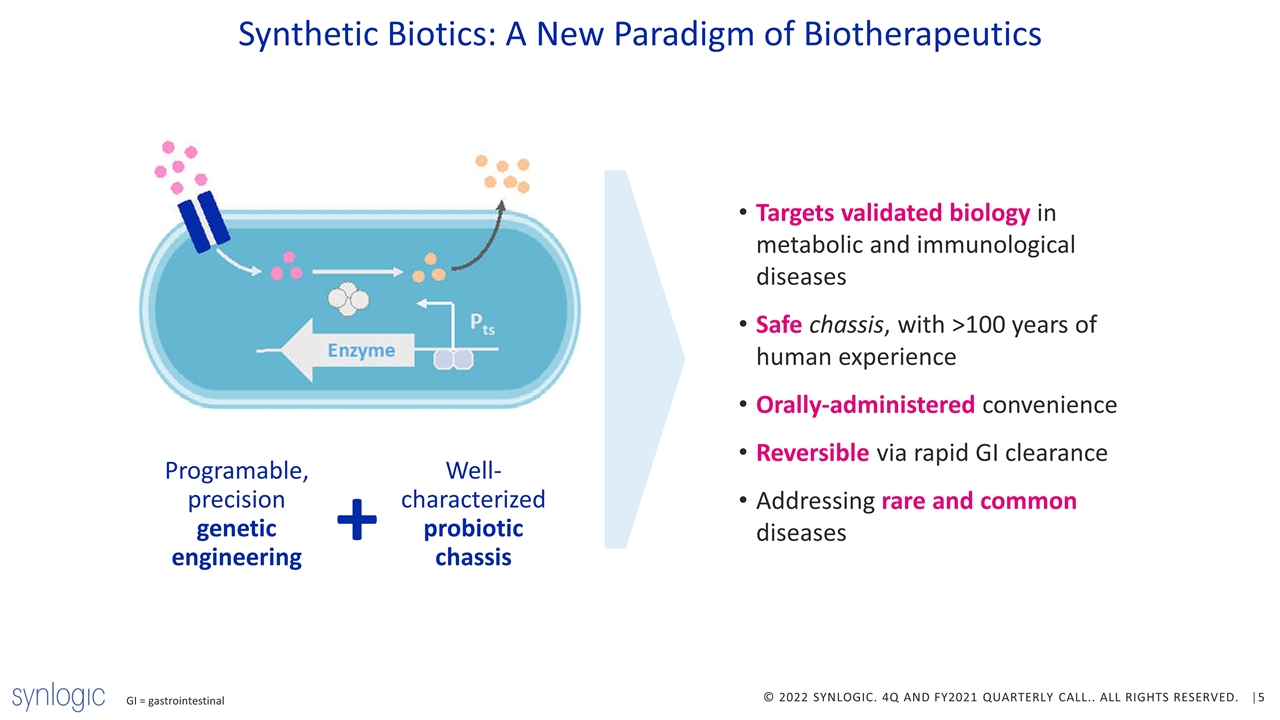
Synthetic Biotics: A New Paradigm of Biotherapeutics GI = gastrointestinal Well-characterized probiotic chassis Programable, precision genetic engineering + Targets validated biology in metabolic and immunological diseases Safe chassis, with >100 years of human experience Orally-administered convenience Reversible via rapid GI clearance Addressing rare and common diseases
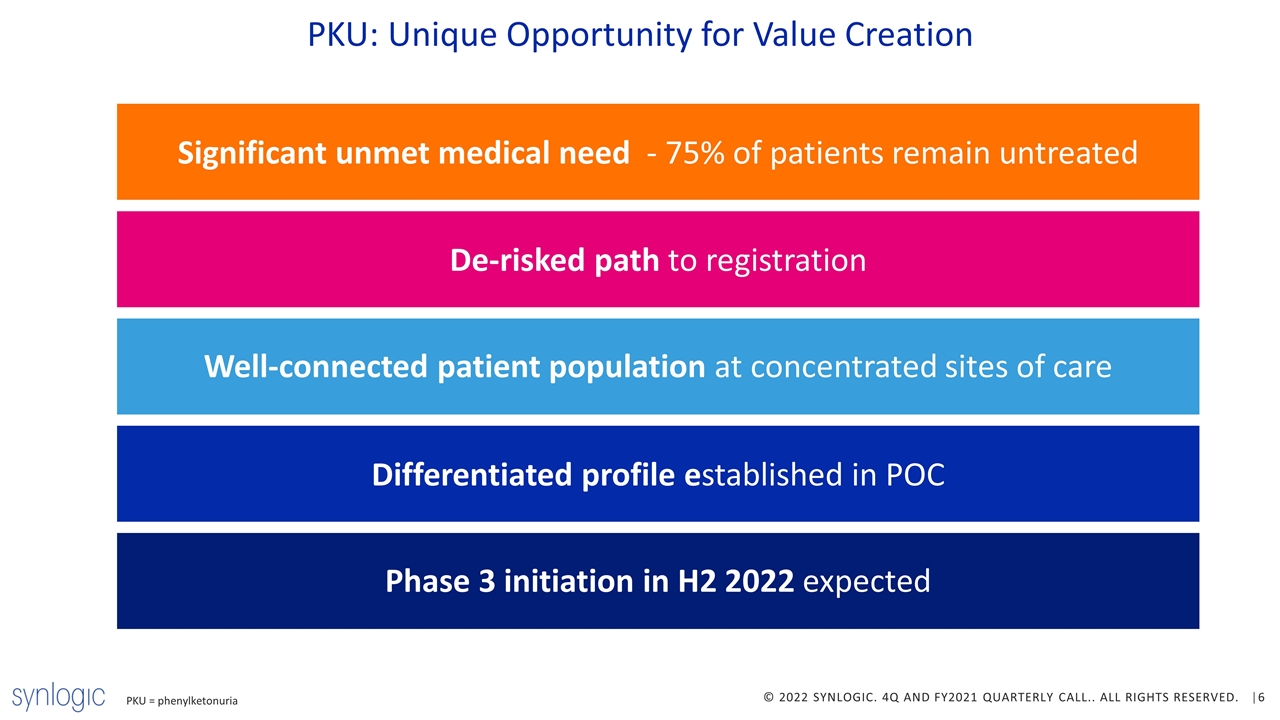
PKU: Unique Opportunity for Value Creation PKU = phenylketonuria Significant unmet medical need - 75% of patients remain untreated De-risked path to registration Well-connected patient population at concentrated sites of care Differentiated profile established in POC Phase 3 initiation in H2 2022 expected

© 2020 SYNLOGIC. QU©AR2T0E1R9LYSYRNELSOUGLTICS.. AALLLL RRIIGGHHTTSS RREESSEERRVVEEDD.. || 33 PKU Opportunity Molly Harper Chief Business Officer
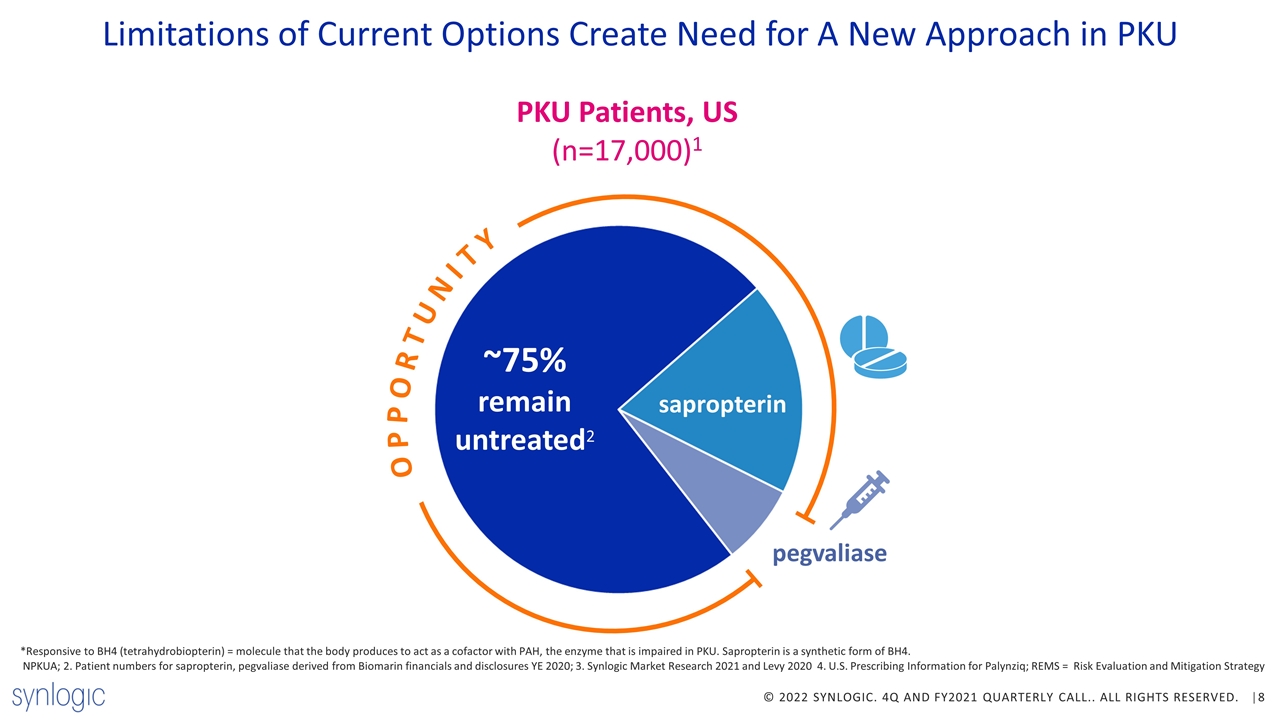
*Responsive to BH4 (tetrahydrobiopterin) = molecule that the body produces to act as a cofactor with PAH, the enzyme that is impaired in PKU. Sapropterin is a synthetic form of BH4. NPKUA; 2. Patient numbers for sapropterin, pegvaliase derived from Biomarin financials and disclosures YE 2020; 3. Synlogic Market Research 2021 and Levy 2020 4. U.S. Prescribing Information for Palynziq; REMS = Risk Evaluation and Mitigation Strategy ~75% remain untreated2 Limitations of Current Options Create Need for A New Approach in PKU OPPORTUNITY PKU Patients, US (n=17,000)1 pegvaliase sapropterin

Potentially Transformative Product Profile Efficacy Safety Oral administration Monotherapy or Adjunctive Use .Sources: Synlogic PKU Program Update, September 20, 20211, Synlogic Market Research 4Q 2021 Designed and Differentiated for PKU

© 2020 SYNLOGIC. QU©AR2T0E1R9LYSYRNELSOUGLTICS.. AALLLL RRIIGGHHTTSS RREESSEERRVVEEDD.. || 33 PKU Clinical Development Dr. Aoife Brennan MB CHB President & CEO
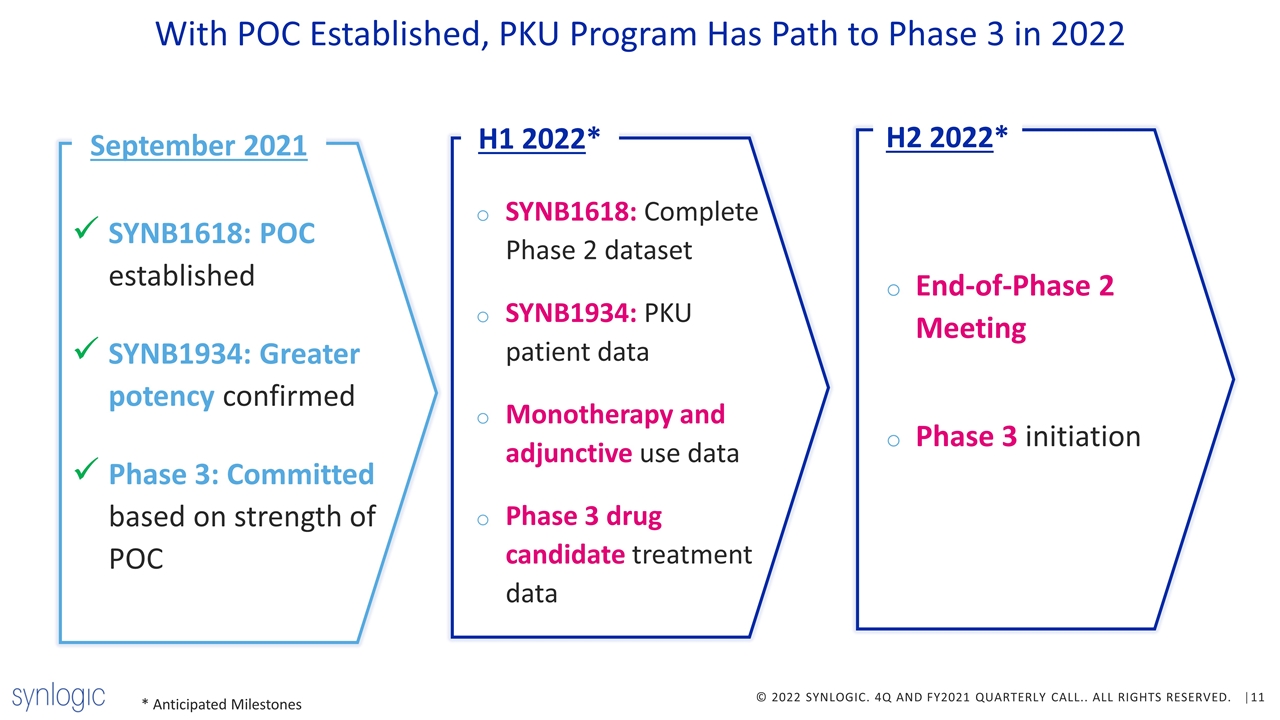
With POC Established, PKU Program Has Path to Phase 3 in 2022 September 2021 SYNB1618: POC established SYNB1934: Greater potency confirmed Phase 3: Committed based on strength of POC H1 2022* SYNB1618: Complete Phase 2 dataset SYNB1934: PKU patient data Monotherapy and adjunctive use data Phase 3 drug candidate treatment data H2 2022* End-of-Phase 2 Meeting Phase 3 initiation * Anticipated Milestones

Regulatory Path De-Risked at Multiple Levels Disease Precedents in two FDA-approved drugs Platform Synthetic Biotics Drug Candidate SYNB1618/SYNB1934

© 2020 SYNLOGIC. QU©AR2T0E1R9LYSYRNELSOUGLTICS.. AALLLL RRIIGGHHTTSS RREESSEERRVVEEDD.. || 33 Enteric Hyperoxaluria, Homocystinuria and Preclinical Programs Dave Hava, PhD Chief Scientific Officer
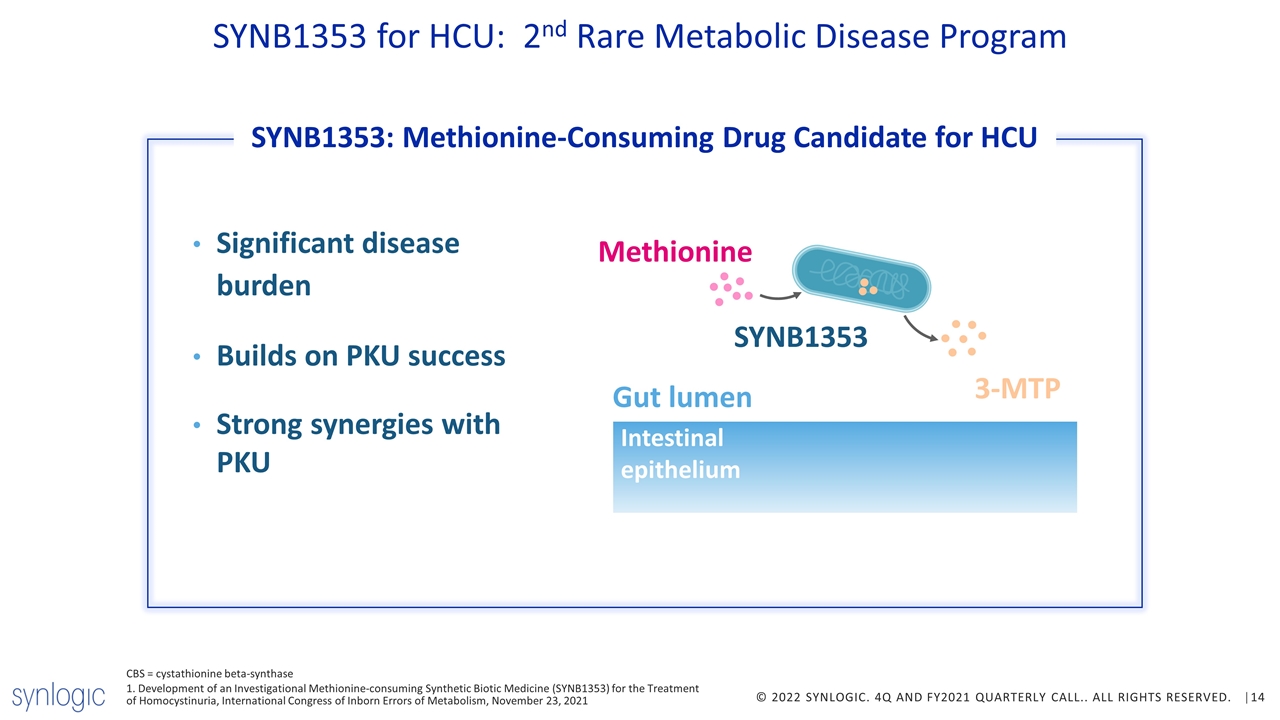
SYNB1353 for HCU: 2nd Rare Metabolic Disease Program Significant disease burden Builds on PKU success Strong synergies with PKU Gut lumen Methionine SYNB1353 Intestinal epithelium CBS = cystathionine beta-synthase 1. Development of an Investigational Methionine-consuming Synthetic Biotic Medicine (SYNB1353) for the Treatment of Homocystinuria, International Congress of Inborn Errors of Metabolism, November 23, 2021 3-MTP SYNB1353: Methionine-Consuming Drug Candidate for HCU

SYNB8802 for Enteric Hyperoxaluria: On Track for POC in 2022 GI = gastrointestinal, CKD = chronic kidney disease, ESRD = end stage renal disease Acute Crises, Long-Term Damage Stomach Small Intestine Colon Consumes Oxalate Across GI Tract Dietary Oxalate SYNB8802
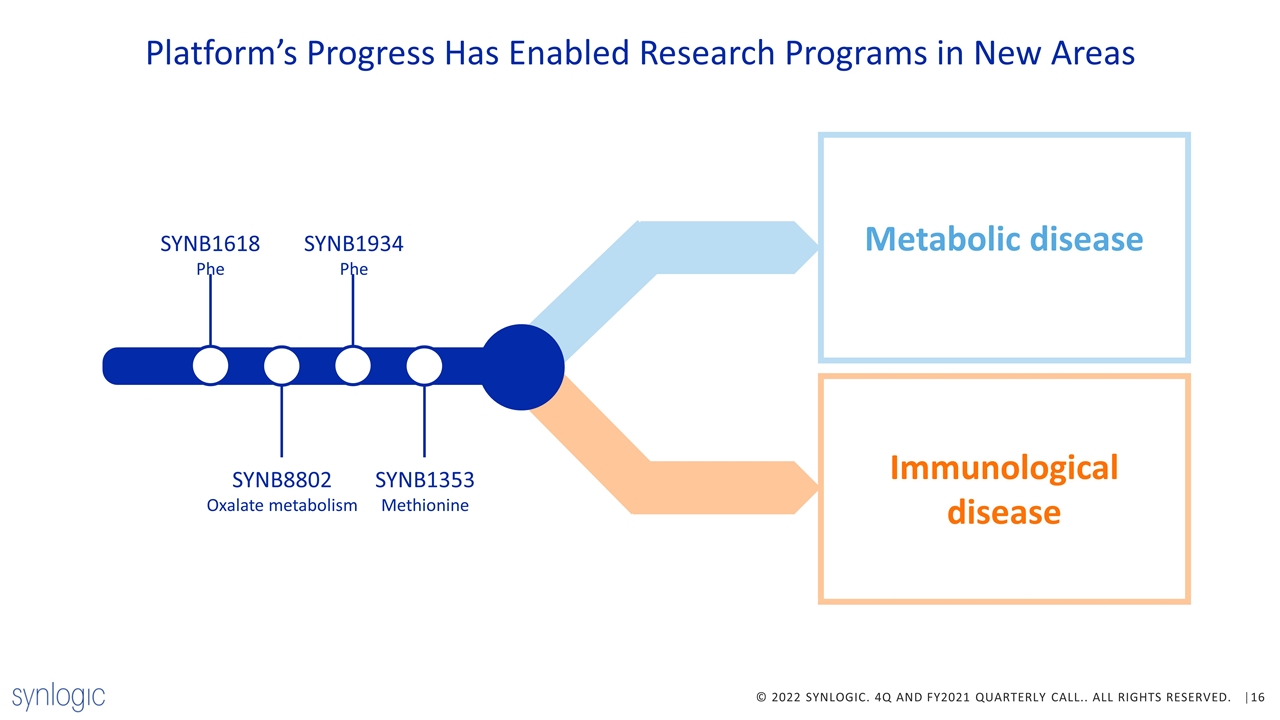
Platform’s Progress Has Enabled Research Programs in New Areas Immunological disease SYNB1618 Phe SYNB8802 Oxalate metabolism SYNB1934 Phe SYNB1353 Methionine Metabolic disease
Strength of Partners Reflects Platform Progress and Potential IND = Investigational New Drug

© 2020 SYNLOGIC. QU©AR2T0E1R9LYSYRNELSOUGLTICS.. AALLLL RRIIGGHHTTSS RREESSEERRVVEEDD.. || 33 Financial Summary Michael Jensen Chief Financial Officer
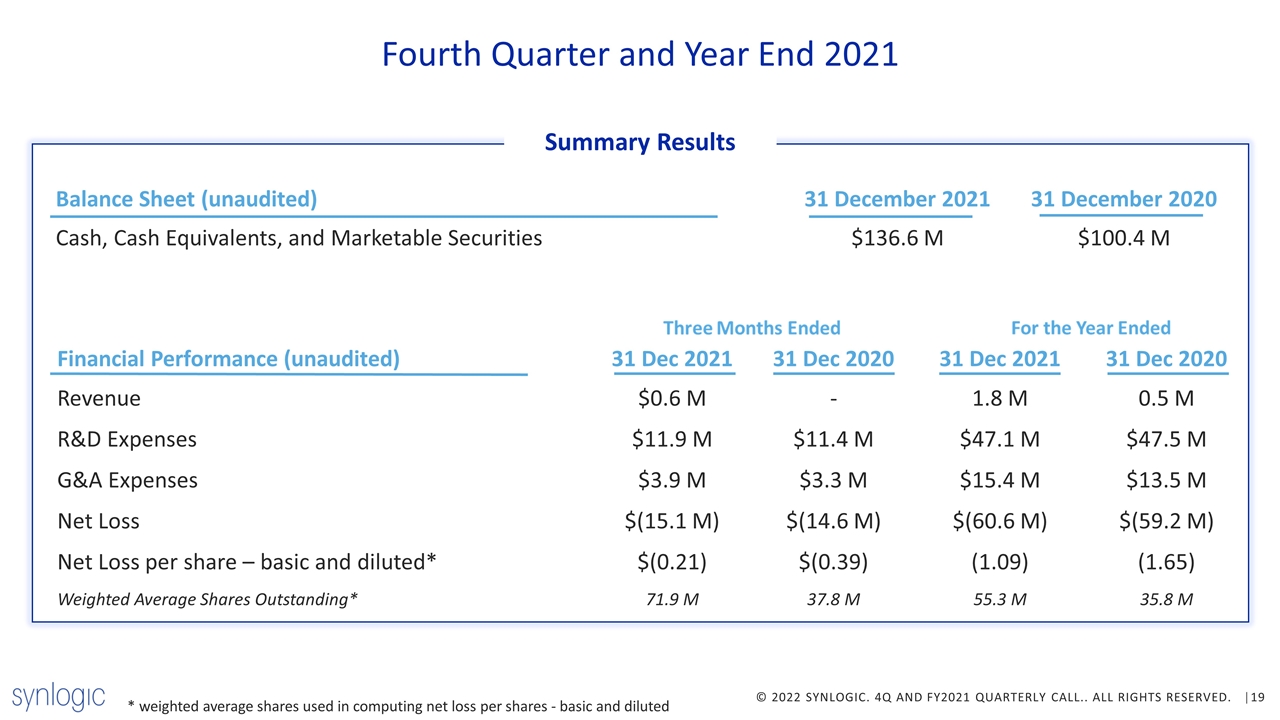
Balance Sheet (unaudited) 31 December 2021 31 December 2020 Cash, Cash Equivalents, and Marketable Securities $136.6 M $100.4 M * weighted average shares used in computing net loss per shares - basic and diluted Fourth Quarter and Year End 2021 Summary Results Financial Performance (unaudited) 31 Dec 2021 31 Dec 2020 31 Dec 2021 31 Dec 2020 Revenue $0.6 M - 1.8 M 0.5 M R&D Expenses $11.9 M $11.4 M $47.1 M $47.5 M G&A Expenses $3.9 M $3.3 M $15.4 M $13.5 M Net Loss $(15.1 M) $(14.6 M) $(60.6 M) $(59.2 M) Net Loss per share – basic and diluted* $(0.21) $(0.39) (1.09) (1.65) Weighted Average Shares Outstanding* 71.9 M 37.8 M 55.3 M 35.8 M

© 2020 SYNLOGIC. QU©AR2TE1R9LYSYRNELSOUGLTICS.. AALLLL RRIIGGHHTTSS RREESSEERRVVEEDD.. || 1414 Concluding Remarks Dr. Aoife Brennan MB CHB President & CEO

PKU Phase 2 data readout Æ H1 2022 PKU Phase 3 initiation Æ H2 2022 HCU Phase 1 data from healthy volunteers Æ H2 2022 Enteric hyperoxaluria proof of concept Æ 2022 Multiple Expected Near-Term Milestones Strong balance sheet: $136.6m* with projected runway into 2024 * As of December 31, 2021

Available For Questions Dave Hava, PhD Chief Scientific Officer Michael Jensen Chief Financial Officer Aoife Brennan, MB ChB President & CEO Antoine Awad Chief Operating Officer Molly Harper Chief Business Officer