
Exhibit 99.2 Transforming Medicine Through Synthetic Biology Proof of Concept for SYNB8802 for Enteric Hyperoxaluria Top-Line Results from Phase 1b Study December 15, 2022 © © 2 20 02 22 2 SY SYN NLO LOG GIIC C.. P PR RO OO OF F O OF F C CO ON NC CE EP PT T F FO OR R SY SYN NB B8 88 80 02 2 IIN N E EH H.. A ALL R LL RIIG GH HT TS R S RE ESE SER RV VE ED D..

Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this presentation regarding strategy, future operations, clinical development plans, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward-looking statements. In addition, when or if used in this presentation, the words may, could, should, anticipate, believe, “look forward,” estimate, expect, intend, on track,” plan, predict and similar expressions and their variants, as they relate to Synlogic, may identify forward-looking statements. Examples of forward-looking statements, include, but are not limited to, statements regarding the potential of Synlogic's approach to Synthetic Biotics to develop therapeutics to address a wide range of diseases including: inborn errors of metabolism and inflammatory and immune disorders; our expectations about sufficiency of our existing cash balance; the future clinical development of Synthetic Biotics; the approach Synlogic is taking to discover and develop novel therapeutics using synthetic biology; and the expected timing of Synlogic's clinical trials of SYNB1618, SYNB1934, SYNB1353, SYNB8802 and SYNB2081 and availability of clinical trial data. Actual results could differ materially from those contained in any forward-looking statements as a result of various factors, including: the uncertainties inherent in the clinical and preclinical development process; the ability of Synlogic to protect its intellectual property rights; and legislative, regulatory, political and economic developments, as well as those risks identified under the heading Risk Factors in Synlogic's filings with the U.S. Securities and Exchange Commission. The forward-looking statements contained in this presentation reflect Synlogic's current views with respect to future events. Synlogic anticipates that subsequent events and developments will cause its views to change. However, while Synlogic may elect to update these forward-looking statements in the future, Synlogic specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing Synlogic's view as of any date subsequent to the date hereof. © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Speakers Aoife Brennan, MB ChB Kyle Wood, MD Caroline Kurtz, PhD Dave Hava, PhD President & CEO Associate Professor, Urology, Chief Development Officer Chief Scientific Officer University of Alabama at Birmingham © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Opening Remarks Dr. Aoife Brennan President & CEO © © 2 20 02 22 2 SY SYN NLO LOG GIIC C.. P PR RO OO OF F O OF F C CO ON NC CE EP PT T F FO OR R SY SYN NB B8 88 80 02 2 IIN N E EH H.. A ALL R LL RIIG GH HT TS R S RE ESE SER RV VE ED D..

SYNB8802: Enteric POC Achieved with Potential for Hyperoxaluria & Urinary Oxalate First Approved the Burden of Lowering from Treatment for Recurrent Phase 1b Study Enteric Kidney Stones Hyperoxaluria © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Enteric Hyperoxaluria & Recurrent Kidney Stones: An Overview Dr. Kyle Wood Associate Professor, Urology University of Alabama at Birmingham © 2020 SYNLOGIC. QU ARTE R LY R ES U LT S.A LLRIG H TS RESERVED .| 3 © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. © 201 9 SY NLO G IC. ALL RI G HTS RESERVE D. | 3

`` `` Enteric Hyperoxaluria (EH) & Recurrent Kidney Stones • Oxalate crystals damage kidneys, impair renal Excruciating function Pain • Can lead to CKD, ESRD, Dietary Oxalate*: nephrocalcinosis, plant-derived; in systemic oxalosis excess, absorbed Circulating from GI tract into oxalate results in circulation crystal formation and chronic, recurrent kidney stones © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. GI = gastrointestinal, CKD = chronic kidney disease, ESRD = end stage renal disease. 1. D’Costa, et al. Nephrol Dial Transpla nt (2020)
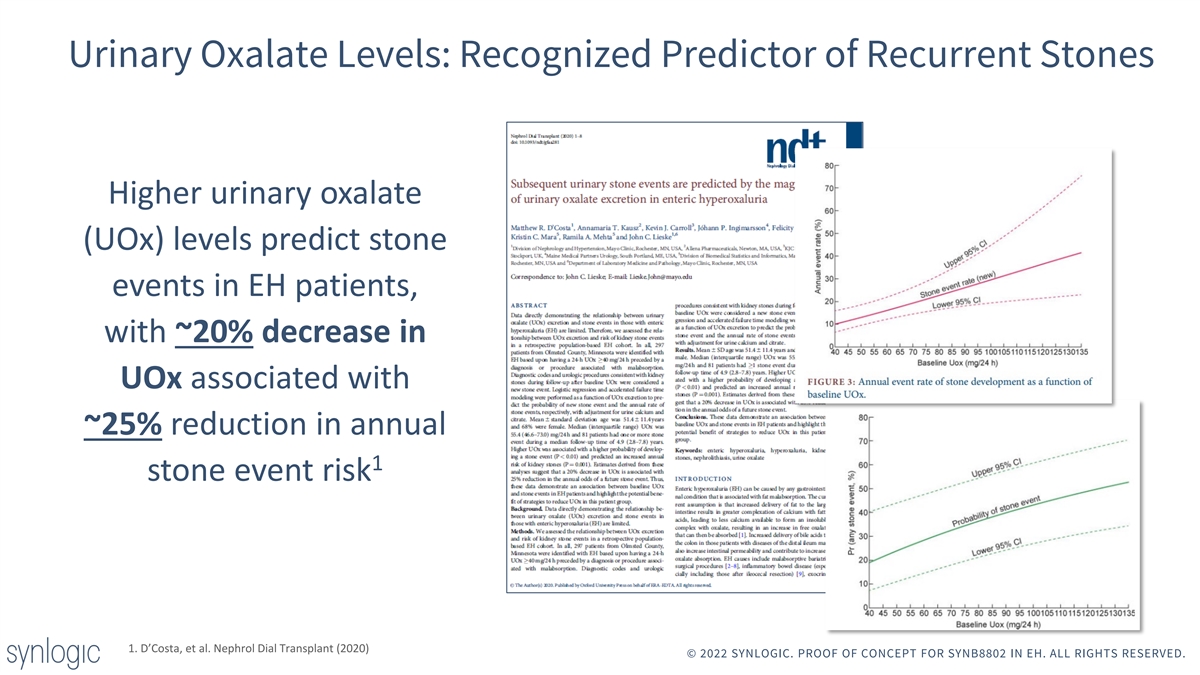
Urinary Oxalate Levels: Recognized Predictor of Recurrent Stones Higher urinary oxalate (UOx) levels predict stone events in EH patients, with ~20% decrease in UOx associated with ~25% reduction in annual 1 stone event risk 1. D’Costa, et al. Nephrol Dial Transplant (2020) © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Urinary Oxalate Levels – and GI Malabsorption - Also Increase Risk for CKD Incident CKD Risk vs. Baseline Methodology • Largest population-based study on the Urinary Oxalate & GI Malabsorption relationship of urinary oxalate levels and incident All Subjects CKD to date 3.0 Malabsorption Condition • Dataset includes 426,896 patients without CKD at 2.5 baseline and includes 12,522 with GI malabsorption 2.0 1.5 Findings • Among patients without a history of CKD, higher 1.0 urine oxalate is associated with higher risk of 0.5 developing incident CKD 0.0 • Prevalence of CKD was twice as high in patients with UOx > 80 mg/d compared with < 20 mg/d • Risk is substantially higher among those with an Baseline UOx underlying malabsorptive condition © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. Puurunen et al, Data presented at ASN in November 2021 <20 20-29 30-39 40-44 45-49 50-59 60-69 70-79 80+ MV Odds Ratio

Hyperoxaluria May Have Genetic or Enteric Etiology Primary Hyperoxaluria (PH) Enteric Hyperoxaluria (EH) Pathology Rare genetic condition Dietary oxalate hyperabsorption Onset Pediatric Adult Underlying insult to bowel: including IBD, Etiology Genetic liver enzyme deficiency bariatric surgery, other chronic GI conditions 90 – 500 mg / 24 hrs 45 – 130 mg / 24 hrs UOx. Levels (~10x normal) (~3x normal) Number of Patients Affected © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Clinical Path: Differential Diagnosis to Enteric Hyperoxaluria Etiology of Oxalate Clinical Presentation Stone Composition Malabsorption Source of GI Injury Other Genetic High Risk of Recurrent Kidney Calcium Oxalate Stones Surgery Idiopathic 1 Stones (“annual stone 1 formers”) 2 Enteric IBD/Crohn’s 2 Other Medical (e.g. CF) 1,2 Estimated U.S. Population: (>3mm) x (~75%) x (~10%) = ~200,000-300,000 1. Ziemba 2017 2. Synlogic 2022 Qualitative Market Research & Real World Evidence analyses by Trinity Partners © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Today, Treatment for EH is Limited to Dietary Restrictions Stone Etiology Current Management Strategy Low Volume Increase fluid intake Ø Modest efficacy Hypocitraturia Citrate supplements Ø Avoids healthy foods (e.g. green vegetables) Low oxalate diet, calcium supplements, Hyperoxaluria Ø Calcium supplements can change IBD Tx (if applicable) exacerbate hypercalciuria May include low sodium diet or thiazide Ø Treatment is even less effective Hypercalciuria diuretics for enteric hyperoxaluria High Uric Acid Increase fluid intake Source: Synlogic 2022 Qualitative Market Research, In-depth interviews with U.S. © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. stone clinic physicians treating patients with recurrent stones

Proof of Concept Data for SYNB8802 Caroline Kurtz, PhD. Chief Development Officer © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

SYNB8802: Consuming Oxalate in the GI Tract to Prevent Absorption Our Approach Precision Engineering of Probiotic Oral therapy Consume oxalate in the GI tract Gut lumen Intestinal epithelium Reduce oxalate in the urine Consumes Oxalate Throughout GI Tract © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. GI = gastrointestinal

SYNB8802 Differentiation: Targeting Oxalate Throughout the GI Tract Oxalate Absorption ABSORPTION SITES OF ACTION Dietary Oxalate Healthy Disease State State Stomach • SYNB8802 consumes oxalate throughout Small Intestine the GI tract • Extends duration of action, increasing Colon oxalate-lowering efficacy potential © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. GI=gastrointestinal

SYNB8802-CP-002 Phase 1b Study Design Phase 1b Study Design Randomized, placebo-controlled Day -3 -2 -1 1 2 3 4 5 6 7 8 9 10 11 12 parallel arm, in-patient study Treatment Treatment Primary endpoint: safety and tolerability 11 11 3 x 10 1 x 10 Placebo Treatment Controlled diet: Standardized dietary Run-In 1 oxalate intake Placebo Patient population: prior Roux-en-Y gastric bypass, with abnormal GI ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ physiology typical of EH Frequency ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ ◉ Disposition: 11 patients, 7 received 24h urine SYNB8802 and 4 received placebo, 1 patient discontinued during Daily collections Plasma placebo treatment Fecal 1 Meals contained an average level of oxalate and low calcium, divided over 3 meals a day during in patient stay © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.
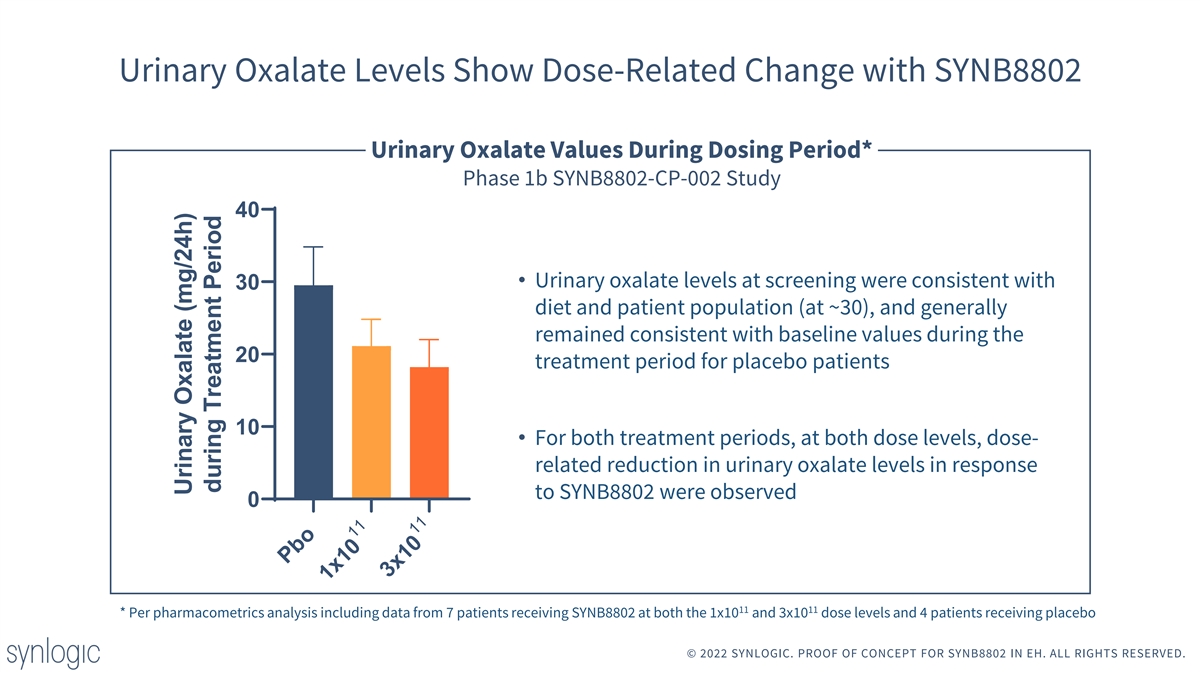
Urinary Oxalate Levels Show Dose-Related Change with SYNB8802 Urinary Oxalate Values During Dosing Period* Phase 1b SYNB8802-CP-002 Study 40 • Urinary oxalate levels at screening were consistent with 30 diet and patient population (at ~30), and generally remained consistent with baseline values during the 20 treatment period for placebo patients 10 • For both treatment periods, at both dose levels, dose- related reduction in urinary oxalate levels in response to SYNB8802 were observed 0 11 11 * Per pharmacometrics analysis including data from 7 patients receiving SYNB8802 at both the 1x10 and 3x10 dose levels and 4 patients receiving placebo © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. Pbo 11 1x10 11 3x10 Urinary Oxalate (mg/24h) during Treatment Period

POC Achieved by Lowering of Urinary Oxalate SYNB8802 Urinary Oxalate Lowering vs. Baseline Compared to Placebo* Phase 1b SYNB8802-CP-002 Study 0% • Findings demonstrate that by consuming oxalate in the GI tract to prevent its absorption, SYNB8802 resulted in changes -20% in urinary oxalate in gastric bypass -28.3 patients -37.8 -40% * • Data analyzed using a pharmacometric model that enabled use of data from all * patients on all study days -60% 11 11 * Per pharmacometrics analysis including data from 7 patients receiving SYNB8802 at both the 1x10 and 3x10 dose levels and 4 patients receiving placebo CFB=change from baseline, CFP=change from placebo © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. 11 1x10 11 3x10 %Change Urinary Oxalate CFB, CFP

Safety and Tolerability Findings SYNB8802 – Proof of Concept from 002 Study Top-Line Results • SYNB8802 was well tolerated, with no serious adverse events • The most common adverse events were GI-related, mild, and transient • The GI-related AEs occurred at a similar frequency in active and placebo groups • One patient in the placebo group discontinued during dosing due to the need for antibiotics © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

SYNB8802: Proof of Concept Achieved, Focused Path Forward • EH: Well-recognized burden of recurrent stones, with no FDA approved specific medical treatment options • SYNB8802: POC data shows potential for a powerfully differentiated treatment • Dose related lowering of UOx established in patients with gastric bypass • Validated mechanism of metabolizing oxalate in GI tract to prevent its absorption 11 11 • Both 1x10 and 3x10 doses exceeded -20% threshold for clinically meaningful reduction in recurrent 11 stone risk, with urinary oxalate lowering of -38% vs. placebo at the 3x10 dose TID • Enriched, concentrated target patient population: EH with highly recurrent stones • Profoundly affected by pain, interventions of recurrent stones • Connected to specialists (e.g. stone clinics) • Path forward: plan to advance towards registrational trial, likely clinical endpoint of stone disease progression © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Cross-Platform Implications Dave Hava, PhD Chief Scientific Officer © 2020 SYNLOGIC. QU ARTE R LY R ES U LT S.A LLRIG H TS RESERVED .| 3 © 201 9 SY NLO G IC. ALL RI G HTS RESERVE D. | 3 © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

SYNB8802 POC: A Milestone for Synthetic Biotic Platform rd ü 3 positive data readout in 4Q 2022, following proof of concept in PKU, and proof of mechanism in HCU ü 2nd disease state with POC achieved ü Platform experience accelerates learnings, for dosing, tolerability © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Concluding Remarks Dr. Aoife Brennan President & CEO © © 2 20 02 22 2 SY SYN NLO LOG GIIC C.. P PR RO OO OF F O OF F C CO ON NC CE EP PT T F FO OR R SY SYN NB B8 88 80 02 2 IIN N E EH H.. A ALL R LL RIIG GH HT TS R S RE ESE SER RV VE ED D..

Advancing a New Class of Biotherapeutics IND- Exploratory Preclinical Enabling Phase 1 Phase 2 Phase 3 Studies Phenylketonuria (PKU) FT SYNB1934* ODD (US) ODD SYNB1353 FT ODD Homocystinuria (HCU) SYNB8802 (US) Metabolic SYNB8802 Enteric Hyperoxaluria ODD Gout SYNB2081 Inflammatory Bowel Disease (IBD) Combo study late ‘21 Immunology IBD Program - Single Target FT = Fast Track granted by FDA ODD = Orphan Drug Designation granted by FDA *First generation SYNB1618 for PKU received both ODD and FT designations by the FDA and orphan medicinal product designation by the EMA.

Available For Questions Aoife Brennan, MB ChB Caroline Kurtz, PhD Dave Hava, PhD Kyle Wood, MD President & CEO Chief Development Officer Chief Scientific Officer Associate Professor, Urology, University of Alabama at Birmingham Michael Jensen Molly Harper Antoine Awad Chief Financial Officer Chief Business Officer Chief Operating Officer © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.

Thank You © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED. © 2022 SYNLOGIC. PROOF OF CONCEPT FOR SYNB8802 IN EH. ALL RIGHTS RESERVED.