Exhibit 99.1
|
RXi Pharmaceuticals Corporation March 2013 OTC: RXII Next Generation in RNAi ®
|
Forward Looking Statements This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believes,” “anticipates,” “plans,” “expects,” “indicates,” “will,” “intends,” “potential,” “suggests” and similar expressions are intended to identify forward-looking statements. These statements are based on RXi Pharmaceuticals Corporation’s (the “Company”) current beliefs and expectations. Such statements include, but are not limited to, statements about the future development of the Company’s products (including timing of clinical trials and related matters associated therewith), the expected timing of certain developmental milestones, the reporting of unblinded data, potential partnership opportunities, the Company’s competition and market opportunity and pro forma estimates. The inclusion of forward-looking statements should not be regarded as a representation by the Company that any of its plans will be achieved. Actual results may differ from those set forth in this presentation due to risks and uncertainties in the Company’s business, including those identified under “Risk Factors” in the Company’s most recently filed Quarterly Report on Form 10-Q and in other filings the Company periodically makes with the U.S Securities and Exchange Commission. The Company does not undertake to update any of these forward-looking statements to reflect a change in its views or events or circumstances that occur after the date of this presentation.
|
Corporate Highlights (1) RXi is a leader in development of advanced RNAi-based therapeutics with strong IP position Self-delivering technology possesses certain advantages in safety, potency & selectivity Experienced scientific and development team (2) RXI-109, RXi’s lead product, is designed to silence a clinically validated target and circumvents many challenges facing other RNAi-based drugs Reduction of CTGF with an antisense oligonucleotide correlated with improved scars in well-controlled clinical trials Delivered locally; avoiding many delivery challenges of other RNA-based drugs Large market opportunity in scar prevention/revision (3) RXi has broad development opportunities based on its RNAi platform Ophthalmology, organ fibrosis, spinal cord injury Platform potential enhanced by recent OPKO Health, Inc. asset purchase
|
Corporate History Spin-off from Galena Biopharma, Inc. as a public, independent company in April 2012 (OTC:RXII) Approximately $50 million invested to date to develop a broad- based RNAi technology platform and our lead compound, RXI-109 RXi received FDA clearance to begin clinical trials with RXI-109 in mid-2012 Conducting 2 Phase 1 clinical trials with RXI-109 with a plan to initiate Phase 2 in H2 2013 Progress in ophthalmology, neurology and liver fibrosis supported by research grants and collaborative projects Located in the greater Boston area—Westborough, MA
|
RXi’s Unique RNAi Platform
|
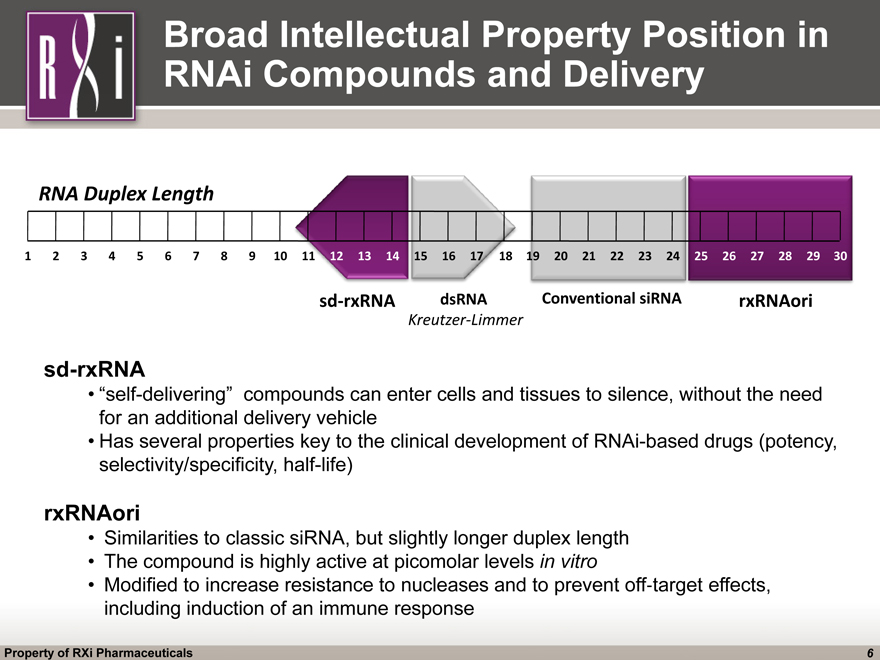
Broad Intellectual Property Position in RNAi Compounds and Delivery 1 3 9 15 17 19 21 23 25 27 6 13 16 20 22 24 26 28 29 2 4 5 7 8 10 11 12 14 30 18 RNA Duplex Length sd-rxRNA Conventional siRNA rxRNAori dsRNA Kreutzer-Limmer sd-rxRNA “self-delivering” compounds can enter cells and tissues to silence, without the need for an additional delivery vehicle Has several properties key to the clinical development of RNAi-based drugs (potency, selectivity/specificity, half-life) rxRNAori Similarities to classic siRNA, but slightly longer duplex length The compound is highly active at picomolar levels in vitro Modified to increase resistance to nucleases and to prevent off^target effects, including induction of an immune response
|
sd-rxRNA Combines Features of RNAi and Antisense Technologies sd-rxRNA therapeutic compounds with drug-like properties Conventional Antisense Clinically relevant, validated PK/PD Conventional RNAi Potent, long-lasting activity Medicinal Chemistry Improved cell uptake and PK/PD O sd-rxRNA Single compound designed to not require delivery vehicle Robust uptake & silencing in multiple preclinical models Structural diversity = novel intellectual property Combining many positives of RNAi & antisense, while avoiding many negatives Provides for broad pipeline of RNAi drugs for unmet medical needs
|
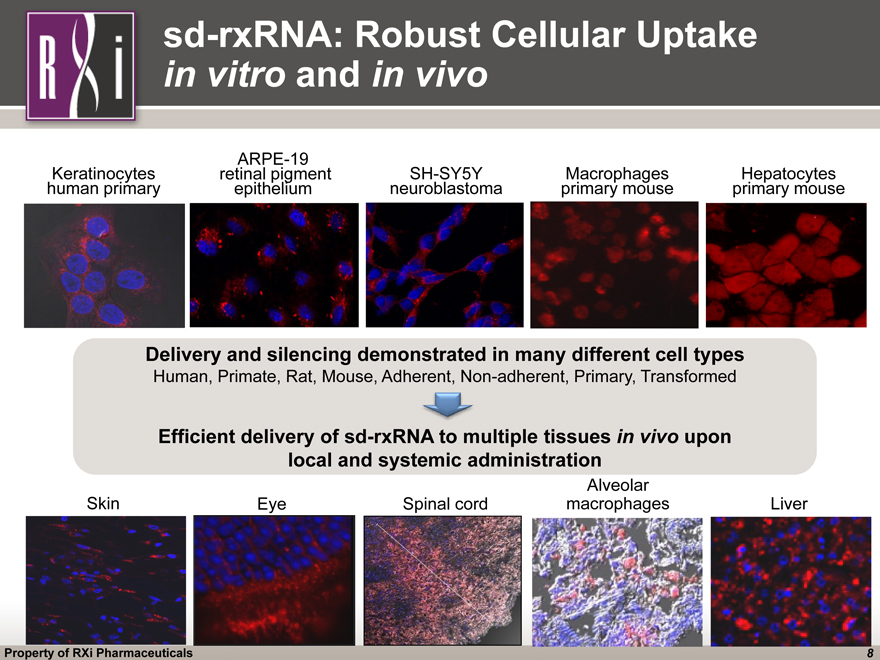
sd-rxRNA: Robust Cellular Uptake in vitro and in vivo Hepatocytes primary mouse ARPE-19 retinal pigment epithelium Macrophages primary mouse Keratinocytes human primary SH-SY5Y neuroblastoma Skin Eye Liver Spinal cord Delivery and silencing demonstrated in many different cell types Human, Primate, Rat, Mouse, Adherent, Non-adherent, Primary, Transformed Efficient delivery of sd-rxRNA to multiple tissues in vivo upon local and systemic administration Alveolar macrophages
|
Selection of Area of Focus: Dermal Scarring Attractive Therapeutic Opportunity Unmet need with limited competition for truly effective therapies No prescription drugs approved Clear development precedent (Excaliard antisense) Large market in scar prevention/revision 42.3 million surgical procedures per year in the U.S.* *US anti-scarring market – The Nemetz Group – December 2009
|
RXi’s Lead Clinical Product Candidate: RXI-109
|
RXI-109 Selection Rationale & Overview Numerous studies implicate Connective Tissue Growth Factor (CTGF) overexpression in scarring and fibrotic diseases CTGF-targeting antisense oligo demonstrated improved scarring (vs. placebo) in 3, well-controlled Phase 2 trials by another company RXI-109, an anti-CTGF sd-rxRNA compound, is delivered locally, avoiding delivery challenges of systemic RNA-based drugs Preclinical data demonstrate potent, selective, dose-dependent and long-lasting silencing of CTGF with RXI-109 RXI-109 addresses a large market opportunity in scar prevention/revision, that today is underserved RXI-109 has a clear development path with early efficacy end points and entered clinical trials in mid-2012
|
The Potential for an Effective Anti- Scarring Agent Is Large Scarring Agent Is Large Scarring Agent Is Large US anti-scarring market – The Nemetz Group – December 2009
|
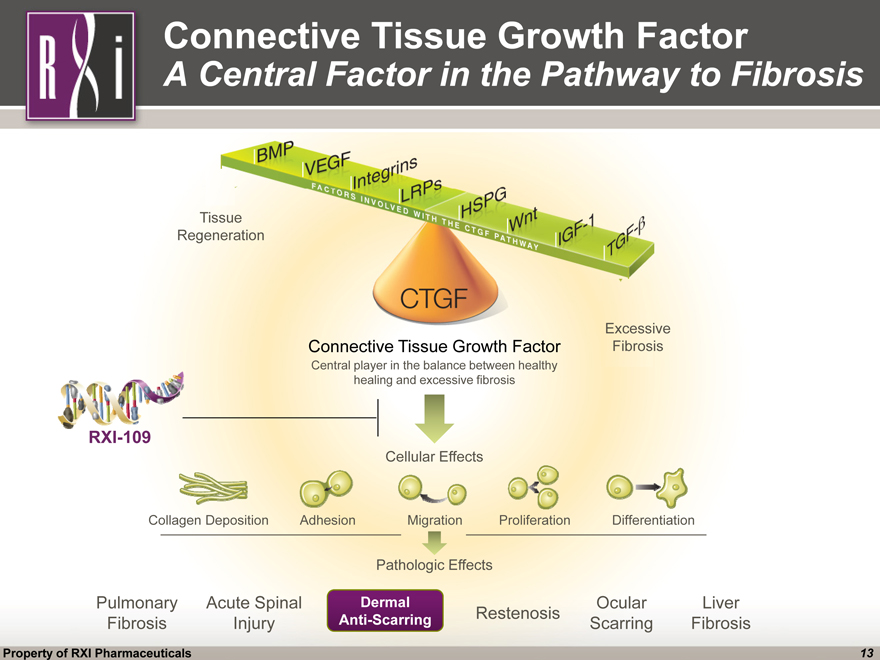
Connective Tissue Growth Factor A Central Factor in the Pathway to Fibrosis RXI-109 Connective Tissue Growth Factor Central player in the balance between healthy healing and excessive fibrosis Cellular Effects Pathologic Effects Pulmonary Fibrosis Acute Spinal Injury Restenosis Ocular Scarring Liver Fibrosis Collagen Deposition Adhesion Migration Proliferation Differentiation Excessive Fibrosis Tissue Regeneration Dermal Anti-Scarring Property of RXI Pharmaceuticals 13
|
Excaliard’s (Now Pfizer’s) CTGF-Targeting Antisense Oligonucleotide Study Results In all three Phase 2 clinical trials, Excaliard’s EXC 001 demonstrated decreased scar severity compared to placebo* Study 201 in surgical incisions in the abdominal pannus demonstrated a statistically significant decrease in scarring with EXC 001 in the physician assessment scale Study 202 in abdominoplasty demonstrated a statistically significant decrease in scarring with EXC 001 in all 3 scar assessment scales at 12 weeks Study 203 in breast scar revision demonstrated a statistically significant decrease in scarring with EXC 001 in all 3 scar assessment scales at both 12 and 24 weeks Physician Assessment at week 24 (p<0.001) Subject Assessment at week 24 (p=0.003) Expert Panel VAS at week 24 (p<0.001) *Results with Excaliard compound not necessarily indicative of results with RXI-109
|
Positive Excaliard Data from 3 Phase 2 Studies of EXC 001 * Significant Improvement in the Appearance of Scars from Revision Surgery Significant Improvement in the Appearance of Scars from Revision Surgery Significant Improvement in the Appearance of Scars from Revision Surgery Significant Improvement in the Appearance of Scars from Revision Surgery *Results with Excaliard compound not necessarily indicative of results with RXI-109
|
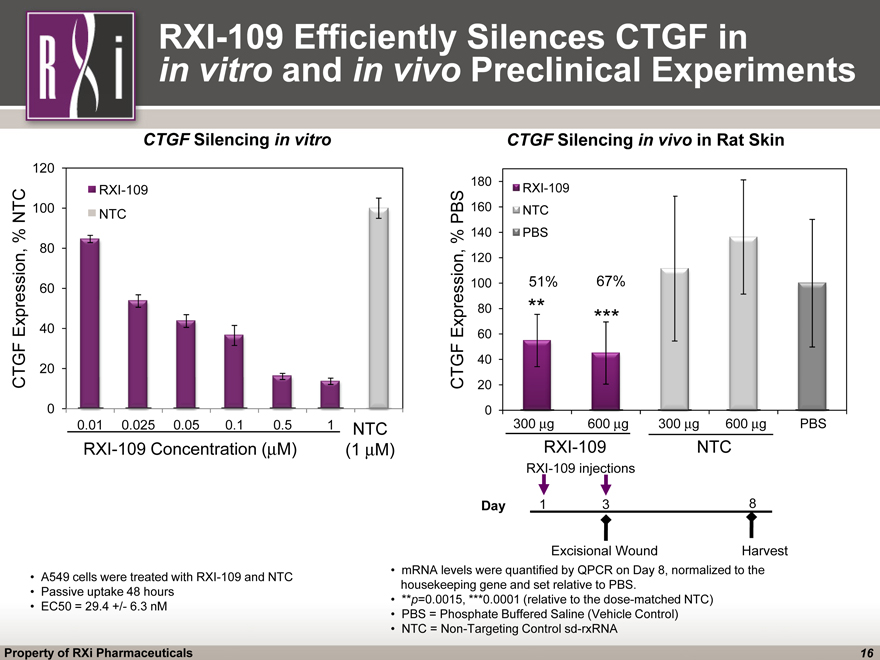
CTGF Silencing in vitro A549 cells were treated with RXI-109 and NTC Passive uptake 48 hours EC50 = 29.4 +/- 6.3 nM (CHART) RXI-109 Efficiently Silences CTGF in in vitro and in vivo Preclinical Experiments RXI-109 Concentration (mM) mRNA levels were quantified by QPCR on Day 8, normalized to the housekeeping gene and set relative to PBS. **p=0.0015, ***0.0001 (relative to the dose-matched NTC) PBS = Phosphate Buffered Saline (Vehicle Control) NTC = Non-Targeting Control sd-rxRNA RXI-109 NTC NTC (1 mM) (CHART) 300 mg 600 mg 300 mg 600 mg PBS RXI-109 NTC CTGF Silencing in vivo in Rat Skin ** ***
|
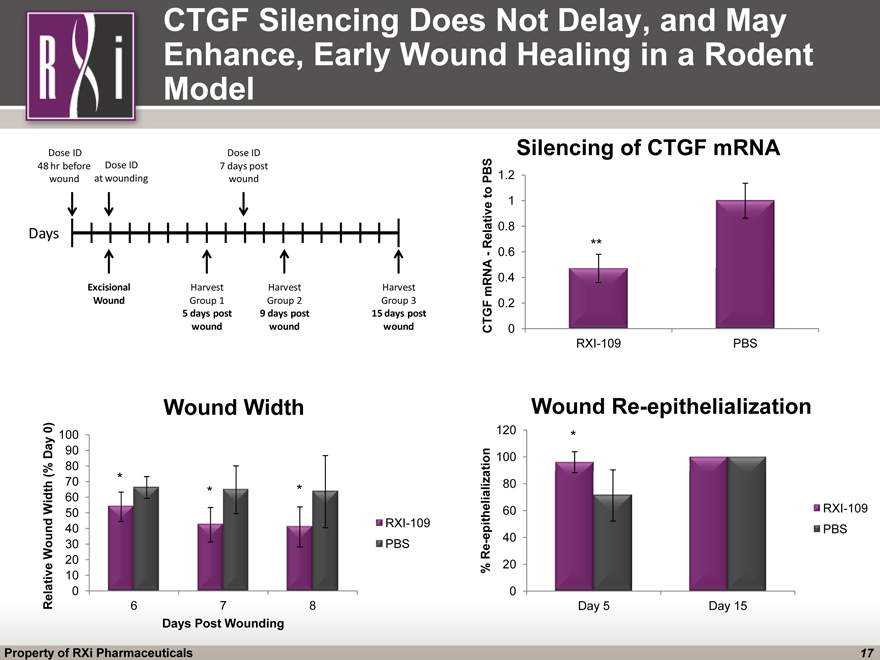
CTGF Silencing Does Not Delay, and May Enhance, Early Wound Healing in a Rodent Model (CHART) (CHART) (CHART) ** * * *
|
RXI-109 Phase 1 Clinical Trials Study 1201: Phase 1 single center, randomized, single-dose, double-blind, ascending dose, within-subject controlled study of RXI-109 for the treatment of incision scars Study 1202: Phase 1 single center, randomized, multi-dose double-blind, ascending dose, within-subject controlled study of RXI-109 for the treatment of incision scars Parameters evaluated: Safety & side effect assessment versus vehicle Photographic comparison versus vehicle Histological comparison of the scar sites versus vehicle Pharmacokinetic parameters after local intradermal injection
|
RXI-109-1201: Abdominal Incision Layout Wound Addresses Column A(Right) Column B(Left) Row 1 1A 1B Row 2 2A 2B Four injections and incisions (2 cm in length) were made on the abdomen. The A and B ‘columns’ were at least 4 cm lateral to the midline of the abdomen. Rows were spaced at least 4 cm apart. Treatment at each incision site was made by intradermal injection according to a predetermined randomization pattern for each subject. Half of the sites were treated with RXI^109, half with placebo.
|
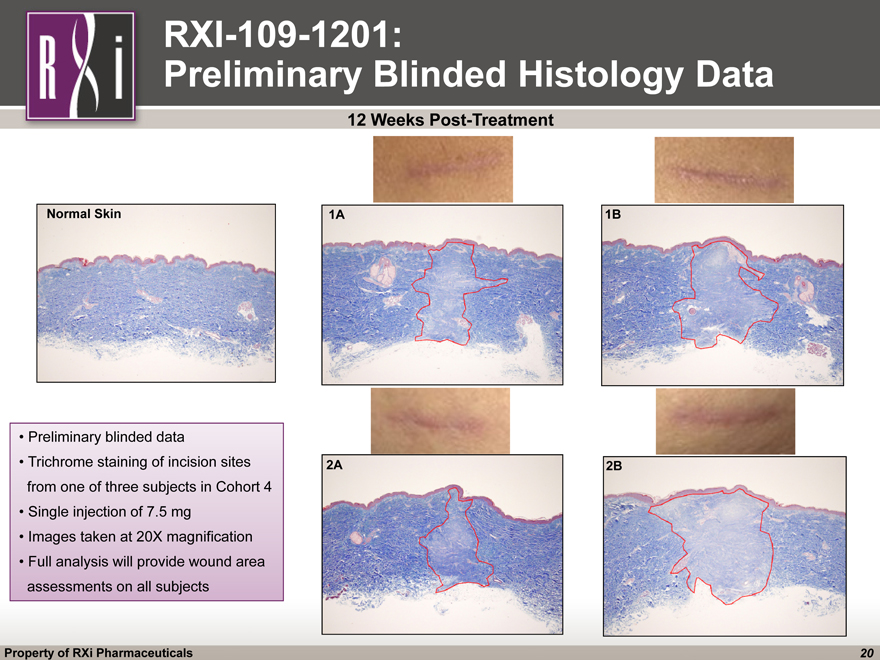
RXI-109-1201: Preliminary Blinded Histology Data 12 Weeks Post-Treatment Normal Skin 1A 2A 1B 2B Preliminary blinded data Trichrome staining of incision sites from one of three subjects in Cohort 4 Single injection of 7.5 mg Images taken at 20X magnification Full analysis will provide wound area assessments on all subjects
|
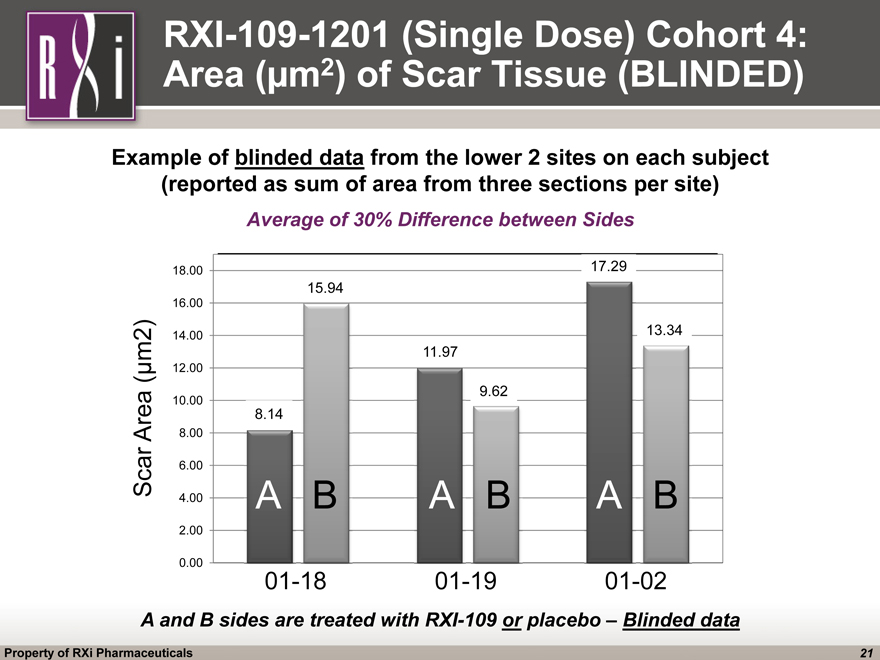
RXI-109-1201 (Single Dose) Cohort 4: Area (µm2) of Scar Tissue (BLINDED) A and B sides are treated with RXI-109 or placebo – Blinded data Average of 30% Difference between Sides Example of blinded data from the lower 2 sites on each subject (reported as sum of area from three sections per site) Scar Area (µm2) 01-18 01-19 01-02 A B A B A B
|
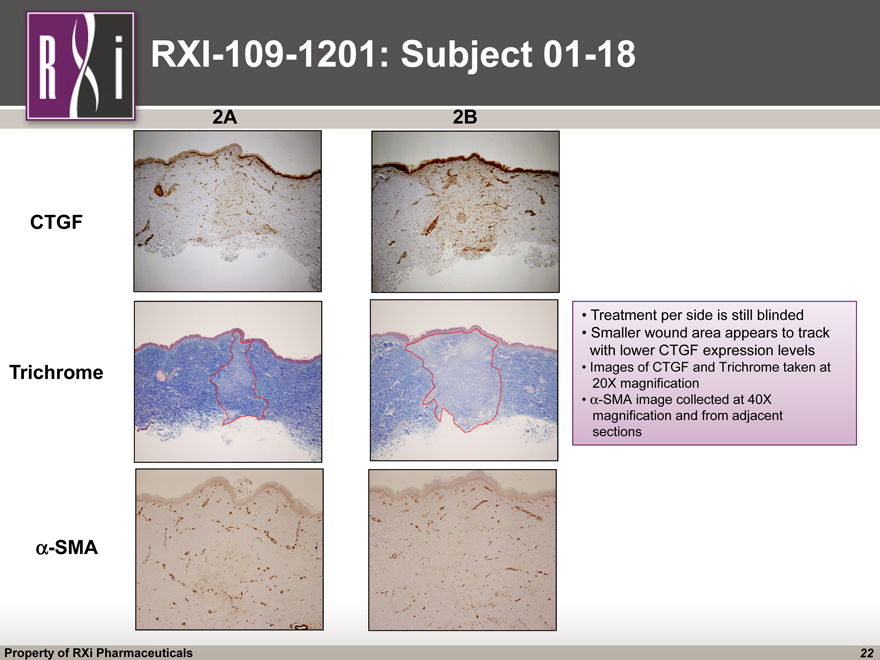
RXI-109-1201: Subject 01-18 2A Trichrome CTGF a-SMA 2B Treatment per side is still blinded Smaller wound area appears to track with lower CTGF expression levels Images of CTGF and Trichrome taken at 20X magnification a-SMA image collected at 40X magnification and from adjacent sections
|
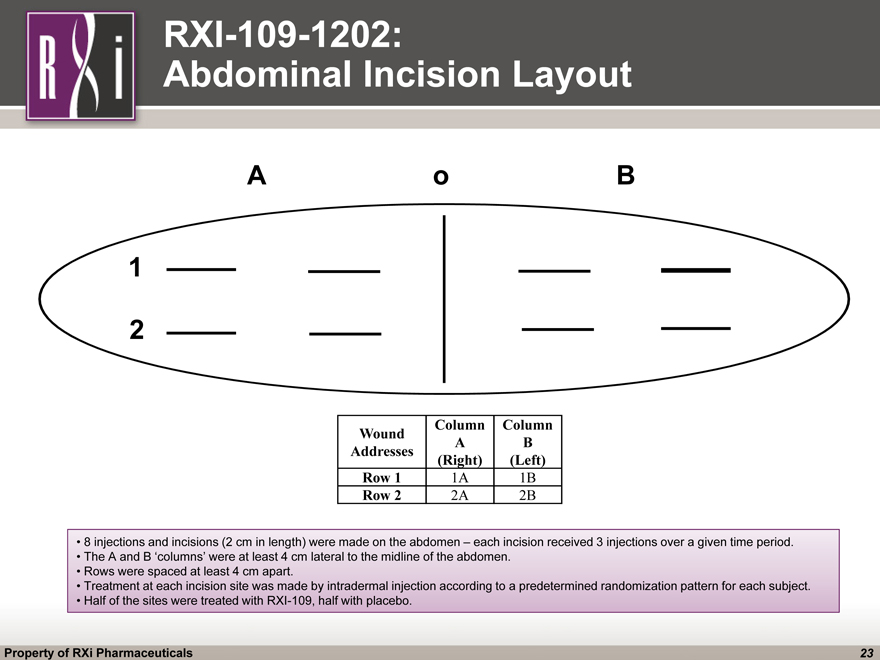
RXI-109-1202: Abdominal Incision Layout Wound Addresses Column A(Right) Column B(Left) Row 1 1A 1B Row 2 2A 2B 8 injections and incisions (2 cm in length) were made on the abdomen – each incision received 3 injections over a given time period. The A and B ‘columns’ were at least 4 cm lateral to the midline of the abdomen. Rows were spaced at least 4 cm apart. Treatment at each incision site was made by intradermal injection according to a predetermined randomization pattern for each subject. Half of the sites were treated with RXI^109, half with placebo. A o B 1 2
|
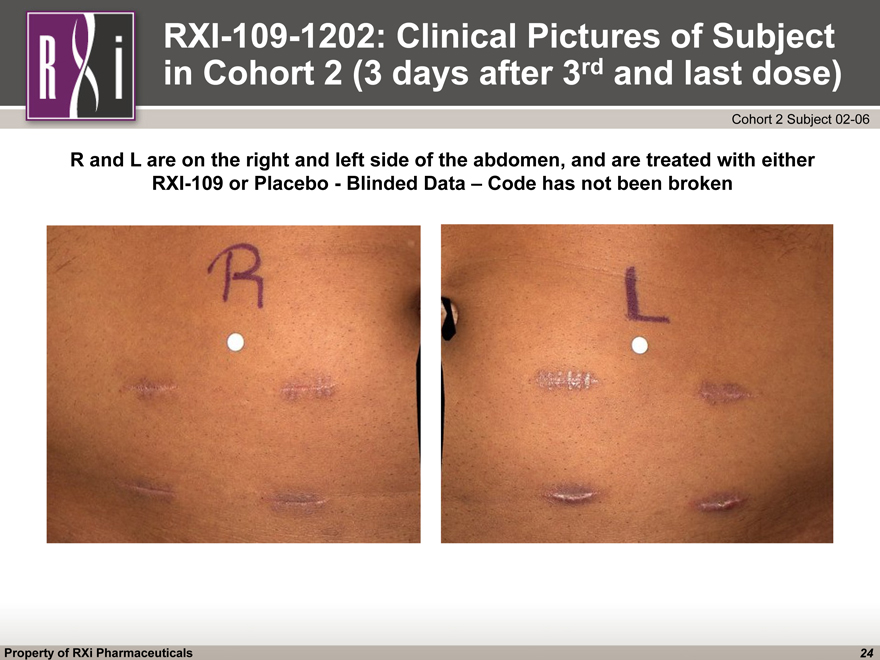
RXI-109-1202: Clinical Pictures of Subject in Cohort 2 (3 days after 3rd and last dose) R and L are on the right and left side of the abdomen, and are treated with either RXI-109 or Placebo—Blinded Data – Code has not been broken Cohort 2 Subject 02-06
|
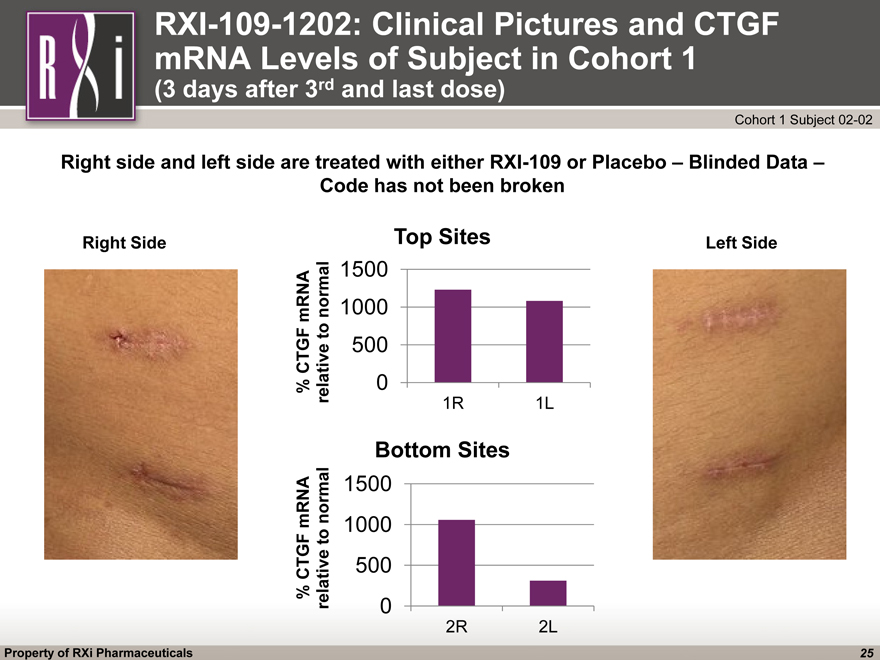
RXI-109-1202: Clinical Pictures and CTGF mRNA Levels of Subject in Cohort 1 (3 days after 3rd and last dose) (CHART) (CHART) Right Side Left Side Right side and left side are treated with either RXI-109 or Placebo – Blinded Data – Code has not been broken Cohort 1 Subject 02-02
|
RXI-109 Summary of Observations & Clinical Development Plan Single-dose study started in June 2012; multi-dose study started in December 2012 First studies aimed at showing safety and initial indications of clinical efficacy Tolerance and safety to date considered excellent by investigators & volunteers Side effects minimal with injection site redness as possibly drug related; the redness was NOT dose related Based on human and animal exposure, intradermal injection provides only 5% systemic exposure as compared to intravenous injection Differences between R & L sites can be seen, but studies are still blinded Unblinded results from Phase 1 studies to be reported in mid-2013 Phase 2 studies to be started in H2 2013
|
Preliminary Plan for Phase 2 Program for RXI-109 Two or three randomized, double-blind, within-subject controlled studies Potential indications include: Breast augmentation scar revision Hysterectomy scar revision Cesarean section scar revision Bilateral keloid scar revision Study objectives include: Determination of optimal dose and schedule Demonstration of safety and efficacy Planned initiation in H2 2013
|
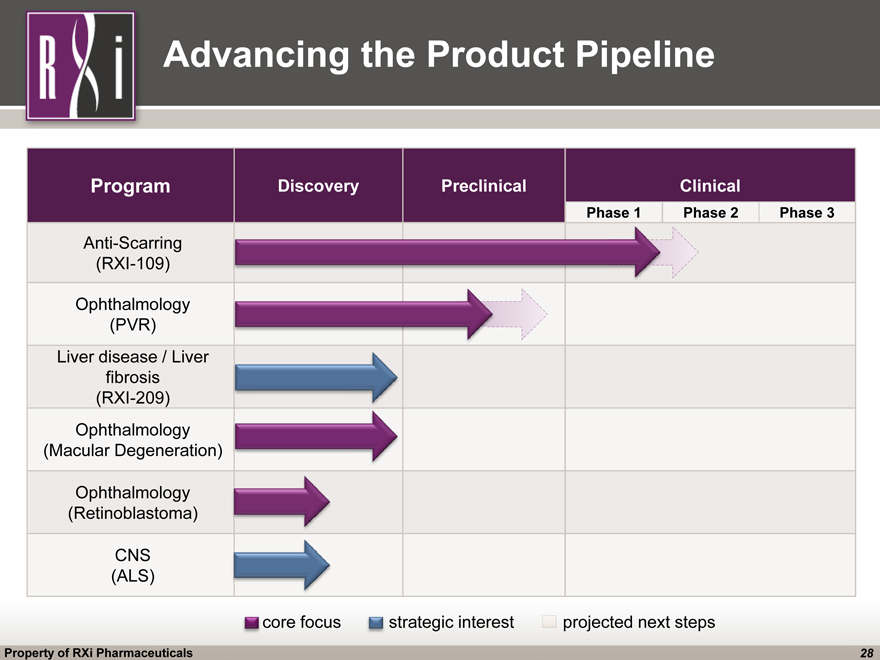
Advancing the Product Pipeline Program Discovery Preclinical Clinical Anti-Scarring(RXI-109) Ophthalmology(PVR) Liver disease / Liver fibrosis(RXI-209) Ophthalmology(Macular Degeneration) Ophthalmology(Retinoblastoma) CNS(ALS) strategic interest projected next steps Phase 1 Phase 2 Phase 3
|
RXI-109 in Ophthalmology PVR (Proliferative Vitreoretinopathy) Proliferative vitreoretinopathy (PVR) is caused by scar tissue formation within the eye Most common complication of a retinal detachment (RD) Occurs in approximately 8-10% of patients who develop an RD Preclinical data strongly support CTGF involvement Preclinical studies show that sd-rxRNA separates effective doses in the eye from toxic doses A potential Orphan indication with unmet need Collaboration with Geoff Lewis, Ph.D. (UCSB) to demonstrate efficacy in the rat retina scar model Potential to file an IND based on the RXI-109 anti-scarring IND with minimal ocular toxicity studies
|
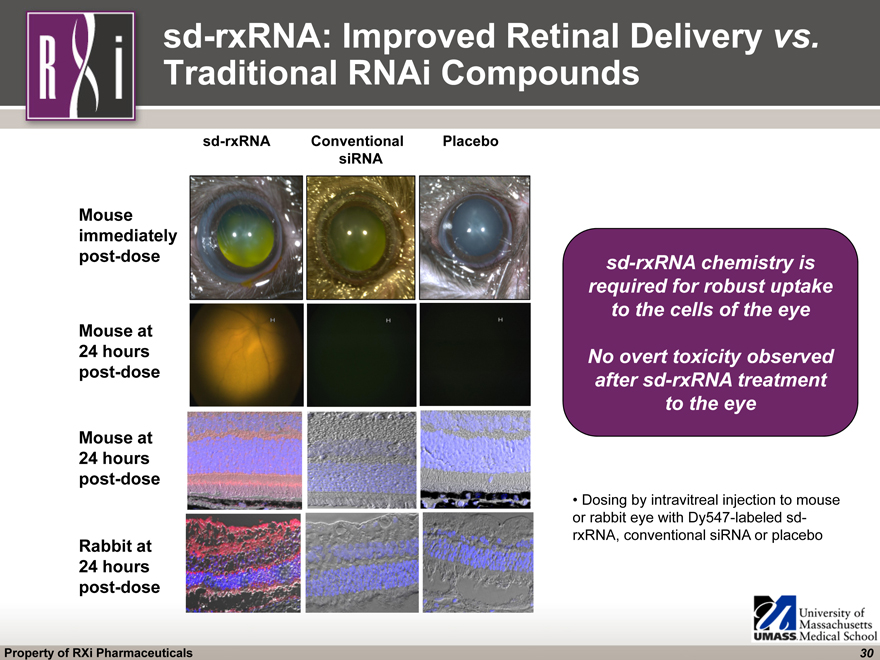
sd-rxRNA Conventional Placebo siRNA Mouse immediately post-dose Mouse at 24 hours post-dose Mouse at 24 hours post-dose Rabbit at 24 hours post-dose Dosing by intravitreal injection to mouse or rabbit eye with Dy547-labeled sd- rxRNA, conventional siRNA or placebo sd-rxRNA: Improved Retinal Delivery vs. Traditional RNAi Compounds sd-rxRNA chemistry is required for robust uptake to the cells of the eye No overt toxicity observed after sd-rxRNA treatment to the eye
|
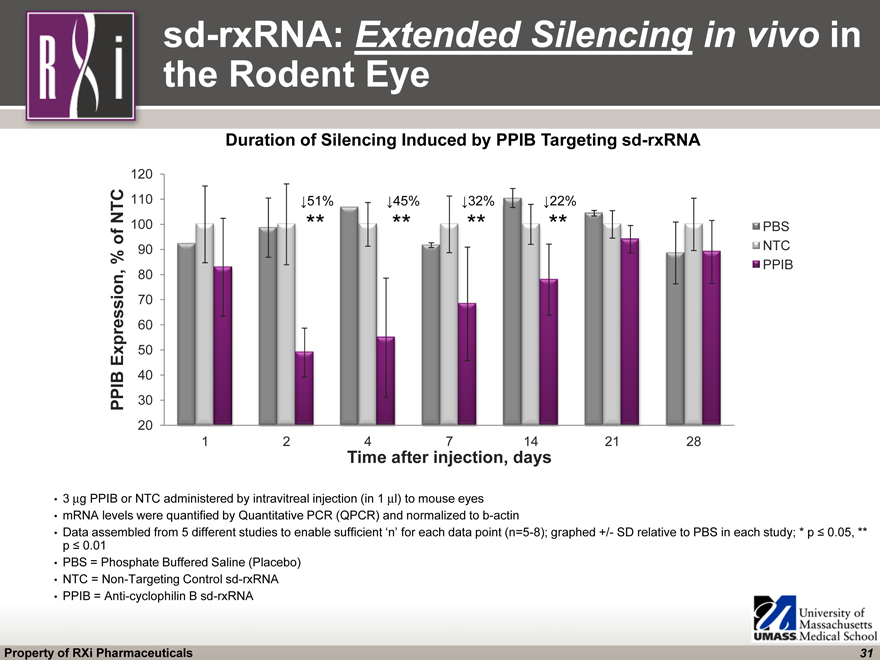
sd-rxRNA: Extended Silencing in vivo in the Rodent Eye 3 mg PPIB or NTC administered by intravitreal injection (in 1 ml) to mouse eyes mRNA levels were quantified by Quantitative PCR (QPCR) and normalized to b-actin Data assembled from 5 different studies to enable sufficient ‘n’ for each data point (n=5-8); graphed +/- SD relative to PBS in each study; * p ^ 0.05, ** p ^ 0.01 PBS = Phosphate Buffered Saline (Placebo) NTC = Non-Targeting Control sd-rxRNA PPIB = Anti-cyclophilin B sd-rxRNA (CHART) ^51% ** ^45% ** ^32% ** ^22% ** Duration of Silencing Induced by PPIB Targeting sd-rxRNA
|
Anti-SOD1 sd-rxRNA in Amyotrophic Lateral Sclerosis (ALS) ALS (Lou Gehrig’s Disease) is a paralytic disease caused by degeneration of motor neurons in the spinal cord, with a 5-year mortality rate of 80% Current reimbursed patient care costs up to $200,000 per year Misfolded SOD1 accumulation is believed to be the primary reason for motor neuron degeneration, and SOD1 gene mutation is responsible for 20% of familial cases of ALS Not targetable with small molecules; no effective treatment Strong support from non profit organization and government funding (~$1.5M currently) Collaboration with Robert Brown, M.D., PhD. (top neurologist in the field)
|
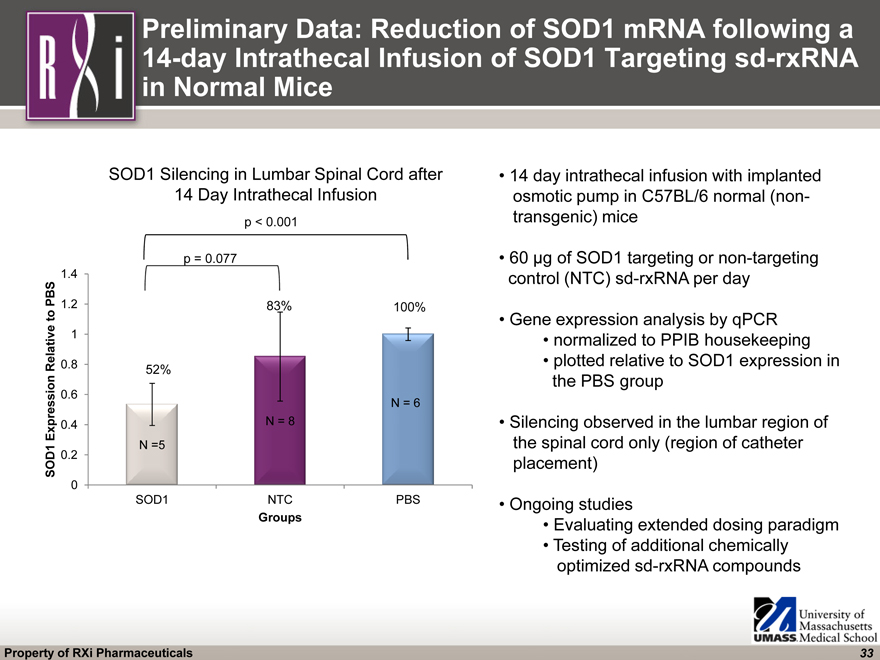
(CHART) 52% 83% 100% Preliminary Data: Reduction of SOD1 mRNA following a 14-day Intrathecal Infusion of SOD1 Targeting sd-rxRNA in Normal Mice 14 day intrathecal infusion with implanted osmotic pump in C57BL/6 normal (non- transgenic) mice 60 µg of SOD1 targeting or non-targeting control (NTC) sd-rxRNA per day Gene expression analysis by qPCR normalized to PPIB housekeeping plotted relative to SOD1 expression in the PBS group Silencing observed in the lumbar region of the spinal cord only (region of catheter placement) Ongoing studies Evaluating extended dosing paradigm Testing of additional chemically optimized sd-rxRNA compounds p = 0.077 p < 0.001 SOD1 Silencing in Lumbar Spinal Cord after 14 Day Intrathecal Infusion
|
RNAi-Related Assets to Be Acquired from OPKO OPKO and collaborators/licensors among early pioneers in RNAi field Extensive intellectual property portfolio including 12 patent families Granted or undergoing prosecution in several key territories including US and EU Coverage of important biological targets, including: Potential indications include: VEGF Ang2 HIF-1-alpha C3 ICAM-1 Diseases of the eye Immune disorders Cancer Inflammatory diseases
|
Management Team Geert Cauwenbergh, Dr. Med. Sc. – Chief Executive Officer Vice President, Research & Development, Johnson & Johnson’s Skin Research Center Founder, Chairman & CEO of Barrier Therapeutics, Inc. (acquired by Stiefel Laboratories, Inc.) Chairman & CEO of Rhei Pharmaceuticals HK Ltd. Doctoral degree in Medicine from K.U., Leuven, Belgium Pamela Pavco, Ph.D. – Chief Development Officer VP of Pharmaceutical Development of Galena Biopharma, Inc. Senior Director, R&D Project Management, Sirna Therapeutics, Inc., a subsidiary of Merck & Co., Inc. Responsible for Sirna-027,1st chemically modified siRNA to enter clinical trials Ph.D. in Biochemistry from Virginia Commonwealth University and post-doctoral work at Duke University Lyn Libertine, M.D. – Vice President Medical Affairs & Safety Assessment Director Pharmacology of Galena Biopharma, Inc. Scientist, Critical Therapeutics, DMPK and clinical development for pulmonary & cardiovascular programs Contributed to development of Zyflo CR®, the only FDA-approved leukotriene synthesis inhibitor for asthma M.D. from University of Massachusetts Karen Bulock, Ph.D. – Vice President Research Associate Director Discovery of Galena Biopharma, Inc. Project Lead for program leading to discovery of RXI-109 Group Leader, Discovery/HTS of Cytrx Corp. Ph.D. in Pharmacology from Yale University
|
Governance & Scientific Leadership Robert Bitterman—Board of Directors: Interim Chairman, Chairman of the Compensation Committee and Chairman of the Nominating Committee President & CEO of Cutanea Life Sciences, Inc., a wholly owned subsidiary of Maruho Company, LTD., a specialty pharma development company focused on diseased and aging skin More than 18 years of executive leadership experience in the pharmaceutical and biologic life science industry Keith Brownlie—Board of Directors: Chairman of the Audit Committee and Chairman of Corporate Governance Distinguished career with Ernst & Young that spanned 36 years. Held the position of Metro New York Area Life Sciences Industry Leader at Ernst & Young Craig Mello, Ph.D.—Chairman, Scientific Advisory Board Co-recipient of the 2006 Nobel Prize in Medicine for RNAi, co-discovered RNAi and co-invented RNAi therapeutics Blais University Chair in Molecular Medicine at the University of Massachusetts Medical School, a Howard Hughes Investigator and a member of the National Academy of Sciences Leroy Young, M.D.—Scientific Advisory Board Director of the BodyAesthetic Research Center in St. Louis, Missouri. Immediate past President of the Aesthetic Surgery Education and Research Foundation (ASERF) Jeannette Graf, M.D.—Scientific Advisory Board Assistant Clinical Professor of Dermatology at the Mount Sinai School of Medicine Independent consultant and advisory board member for a number of cosmetic and pharmaceutical companies, including Neutrogena, Johnson & Johnson, RoC, Allergan, Aveeno, Merz/Bioform and Medicis
|
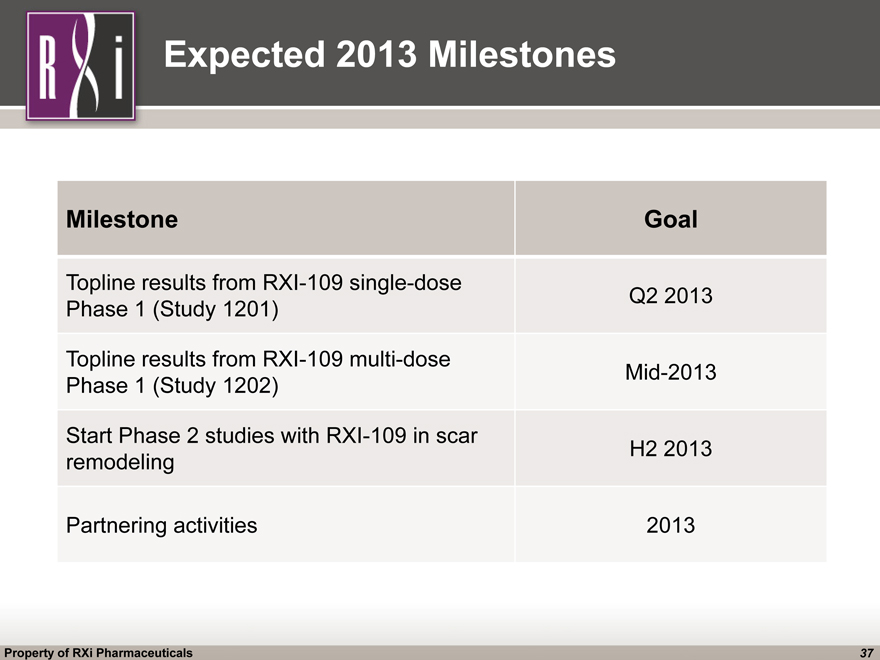
Expected 2013 Milestones Expected 2013 Milestones
|
Corporate Highlights (1) RXi is a leader in development of advanced RNAi-based therapeutics with strong IP position Self-delivering technology possesses certain advantages in safety, potency & selectivity Experienced scientific and development team (2) RXI-109, RXi’s lead product, is designed to silence a clinically validated target and circumvents many challenges facing other RNAi-based drugs Reduction of CTGF with an antisense oligonucleotide correlated with improved scars in well-controlled clinical trials Delivered locally; avoiding many delivery challenges of other RNA-based drugs Large market opportunity in scar prevention/revision (3) RXi has broad development opportunities based on its RNAi platform Ophthalmology, organ fibrosis, spinal cord injury Platform potential enhanced by recent OPKO Health, Inc. asset purchase