FORM 10-K
TABLE OF CONTENTS
|
|
| | | |
| 2 |
| | | |
| | 3 |
| | | |
| | 48 |
| | | |
| | 76 |
| | | |
| | 76 |
| | | |
| | 77 |
| | | |
| | 77 |
| | | |
| 77 |
| | | |
| | 77 |
| | | |
| | 77 |
| | | |
| | 77 |
| | | |
| | 85 |
| | | |
| | 86 |
| | | |
| | 86 |
| | | |
| | 86 |
| | | |
| | 86 |
| | | |
| | 86 |
| | | |
| 87 |
| | | |
| | 87 |
| | | |
| | 93 |
| | | |
| | 101 |
| | | |
| | 103 |
| | | |
| | 105
|
| | | |
| 106
|
| | | |
| | 106 |
| | | |
| | 107 |
PART I
In this Annual Report on Form 10-K, unless the context otherwise requires:
| • | references to “Chemomab Therapeutics Ltd.”, “Chemomab,” the “Company,” “us,” “we” and “our” refer to Chemomab Therapeutics Ltd., an Israeli company and its consolidated subsidiaries; however, with respect to the presentation of financial results for historical periods that preceded the Merger (as defined below), these terms refer to the financial results of the Company’s wholly owned subsidiary, Chemomab Ltd., which was the accounting acquirer in the Merger; |
| • | references to “ordinary shares,” “our shares” and similar expressions refer to the Company’s ordinary shares, no nominal (par) value; |
| • | references to “ADS” refer to the American Depositary Shares listed on the Nasdaq Capital Market (“Nasdaq”) under the symbol “CMMB,” each representing twenty (20) ordinary shares; |
| • | references to “dollars,” “U.S. dollars” and “$” are to United States Dollars; |
| • | references to “NIS” are to New Israeli Shekels; |
| • | references to the “Companies Law” are to Israel’s Companies Law, 5759-1999, as amended; |
| • | references to the “SEC” are to the U.S. Securities and Exchange Commission; and |
| • | references to the “Merger” refer to the merger involving Anchiano Therapeutics Ltd., or Anchiano, and Chemomab Ltd., whereby a wholly owned subsidiary of Anchiano merged with and into Chemomab Ltd., with Chemomab Ltd. surviving as a wholly owned subsidiary of Anchiano. Upon consummation of the Merger on March 16, 2021, Anchiano changed its name to “Chemomab Therapeutics Ltd.” and the business conducted by Chemomab Ltd. became primarily the business conducted by the Company. |
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
The statements set forth under the captions “Business,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Risk Factors,” and other statements included elsewhere in this Annual Report on Form 10-K, which are not historical, constitute “forward-looking statements” within the meanings of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), including statements regarding expectations, beliefs, intentions or strategies for the future. These statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases, you can identify forward-looking statements by terms including “anticipates,” “believes,” “could,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “projects,” “should,” “will,” “would,” and similar expressions intended to identify forward-looking statements, but these are not the only ways these statements are identified. Forward-looking statements reflect our current views with respect to future events and are based on assumptions and subject to risks and uncertainties. Such forward-looking statements include, but are not limited to: the initiation, timing, progress and results of our preclinical studies and other therapeutic candidate development efforts; our ability to develop and advance a future therapeutic candidate into clinical trial or to successfully complete our preclinical studies; our receipt of regulatory approvals for a future therapeutic candidate, and the timing of other regulatory filings and approvals; the clinical development, commercialization and market acceptance of a future therapeutic candidate; our ability to establish and maintain corporate collaborations and integrate new therapeutic candidates and new personnel; the interpretation of the properties and characteristics of a future therapeutic candidate; the implementation of our business model and strategic plans for our business and future therapeutic candidates; the scope of protection we are able to establish and maintain for intellectual property rights covering future therapeutic candidates and our ability to operate our business without infringing the intellectual property rights of others; estimates of our expenses, future revenues, capital requirements and our needs for additional financing; risks relating to our ability to finance our activities and research programs; our dependence on performance by third-party providers of services and supplies, including without limitation, clinical research organizations; the inherent risks and uncertainties in developing the types of preclinical products we are attempting to develop; competitive companies, technologies and our industry; risks related to our ability to maintain compliance with the continued listing standards of Nasdaq; our future revenue, expenses, inflationary pressures, capital requirements and our needs for additional financing, including our ability to continue as a going concern; our ability to obtain additional financing in future offerings; the volatility of the trading price of the ADSs; and other risks and uncertainties, including those listed in the section titled “Risk Factors.”
Item 1. Business
Overview
Chemomab is a clinical-stage biotechnology company focused on the discovery and development of innovative therapeutics for fibrotic and inflammatory diseases with high unmet needs. Based on the unique and pivotal role of the soluble protein CCL24 in promoting fibrosis and inflammation, Chemomab developed CM-101, a monoclonal antibody designed to bind and block CCL24 activity. CM-101 has demonstrated the potential to treat multiple severe and life-threatening fibrotic and inflammatory diseases.
Chemomab has pioneered the therapeutic targeting of CCL24, a chemokine that promotes various types of cellular processes that regulate inflammatory and fibrotic activities through the CCR3 receptor. The chemokine is expressed in various types of cells, including immune cells, endothelial cells and epithelial cells. We have developed a novel CCL24 inhibiting product candidate with dual anti-fibrotic and anti-inflammatory activity that modulates the complex interplay of both of these inflammatory and fibrotic mechanisms, which drive abnormal states of fibrosis and clinical fibrotic diseases. This innovative approach is being developed for difficult to treat rare diseases, also known as orphan indications or diseases, such as primary sclerosing cholangitis, or PSC, and systemic sclerosis, or SSc, for which patients have no established disease modifying standard of care treatment options. We estimate that there are approximately 77 thousand patients suffering from PSC in the U.S., EU and Japan, representing over a $1 billion market opportunity, and approximately 170 thousand patients suffering from SSc in those same markets, representing over a $1.5 billion market opportunity.
CM-101, our lead clinical product candidate, is a first-in-class humanized monoclonal antibody that attenuates the basic function of the soluble chemokine CCL24, also known as eotaxin-2, as a regulator of major inflammatory and fibrotic pathways. We have demonstrated that CM-101 interferes with the underlying biology of inflammation and fibrosis through a novel and differentiated mechanism of action. Based on these findings, we are actively advancing CM-101 in Phase 2 clinical studies directed toward two distinct clinical indications that include patients with liver or skin, and/or lung fibrosis. We are currently conducting a Phase 2 clinical study in PSC, a rare obstructive and cholestatic liver disease. The study is actively recruiting patients in the U.S., Europe and Israel and is being expanded by adding clinical sites, an additional high dose arm (20mg/kg) as well as an open label extension. We had earlier proposed to add both low and high dose arms to the study but the recent encouraging results reported from our Phase 2 liver fibrosis trial in NASH patients, dosed at 5mg/kg, along the positive Phase 1b data we previously reported in non-alcoholic fibrotic liver disease (NAFLD) patients dosed at 5mg/kg and 2,5mg/kg, are seen as providing us sufficient data on the performance of the lower dose to drop it from the current trial, which is focusing on the 10mg/kg and 20g/kg doses. We believe this change will facilitate timely conduct and completion of the trial. If regulators in the future do not agree that the existing low dose data are sufficient, we always have the option to add a lower dose group as part of the Phase 3 clinical program.
We are also planning to open a Phase 2 clinical trial in SSc about midyear 2023. The trial in SSc, a rare autoimmune rheumatic disease characterized by fibrosis in the skin and lung and other organs, will focus on establishing biological and clinical proof of concept in this patient population. Although our primary focus is on these two rare indications, as we noted, an additional Phase 2 clinical study in patients with liver fibrosis due to non-alcoholic steatohepatitis, or NASH has recently been completed. This trial provided safety and pharmacokinetic (PK) data and is informative in determining whether the company advances the development of its current subcutaneous formulation of CM-101. Additionally, the trial measured a number of biomarkers that may be relevant to the potential activity of CM-101 in other fibro-inflammatory conditions. We recently reported results from this trial, which showed that the trial met its primary endpoint of safety and tolerability, and that CM-101 demonstrated encouraging activity in secondary endpoints that include a range of liver fibrosis biomarkers and physiologic assessments.
Fibrosis is the abnormal and excessive accumulation of collagen and extracellular matrix, the non-cellular component in all tissues and organs, which provides structural and biochemical support to surrounding cells. When present in excessive amounts, collagen and extracellular matrix lead to scarring and thickening of connective tissues, affecting tissue properties and potentially leading to organ dysfunction and failure. Fibrosis can occur in many different tissues, including lung, liver, kidney, muscle, skin, and the gastrointestinal tract, resulting in a wide array of progressive fibrotic conditions. Fibrosis and inflammation are intrinsically linked. While a healthy inflammatory response is necessary for efficient tissue repair; after disease or injury, an excessive, uncontrolled inflammatory response can lead to tissue fibrosis that in turn can further stimulate inflammatory processes in a fibro-inflammatory vicious cycle.
FDA Clearance of our IND Application for CM-101 in Phase 2 Trial in SSc Patients
On February 21, 2023, we reported U.S. Food and Drug Administration (FDA) clearance of our Investigational New Drug (IND) Application to evaluate CM-101 in a Phase 2 trial in adults with systemic sclerosis (SSc). The Phase 2 ABATE trial is a multicenter, randomized, double-blind, proof-of-biology study to evaluate the sAfety, toleraBility, and Activity of CM-101 in patients with sysTEmic sclerosis. It expects to enroll 45 patients with clinically active dermatologic, vascular or pulmonary SSc. The study population is expected to be roughly split between patients with diffuse SSc and patients with limited SSc. The primary outcome measure is safety. Secondary endpoints include multiple serum-based biological markers and a variety of exploratory biological and clinical outcomes, including the American College of Rheumatology Composite Response Index in Systemic Sclerosis (ACR-CRISS) score and its revisions (rCRISS). The trial includes a 24-week double blind period during which active treatment patients will receive 10 mg/kg of CM-101 by intravenous infusion every three weeks, followed by a 24-week open label extension, where all patients will receive a 10 mg/kg dose. The trial includes multiple clinical assessments of the skin, vasculature and pulmonary function. It is expected to generate additional information about disease mechanisms, provide data relevant to future patient stratification strategies and inform the selection of appropriate endpoints for future studies. The trial is expected to begin enrolling patients midyear of 2023.A topline data read-out is targeted for the second half of 2024.
Report Topline Results from CM-101 Phase 2a Liver Fibrosis Biomarker Trial in NASH Patients
On January 3, 2023, we reported positive topline results from our Phase 2a liver fibrosis biomarker trial of CM-101 in NASH patients. This trial was primarily designed to assess a subcutaneous formulation of CM-101 and to evaluate the drug’s impact on liver fibrosis biomarkers relevant to both NASH and the fibro-inflammatory conditions that represent the focus for the company, such as PSC and SSc. The trial met its primary endpoint of safety and tolerability, and CM-101 demonstrated encouraging activity in secondary endpoints that include a range of liver fibrosis biomarkers and physiologic assessments measured at baseline and at week 20.
The randomized, placebo-controlled trial enrolled 23 NASH patients with stage F1c, F2 and F3 disease who were randomized to receive either CM-101 or placebo. Patients received a dose of 5 mg/kg of study drug administered by subcutaneous (SC) injection once every two weeks, for a treatment period of 16 weeks. Key findings of the CM-101 Phase 2a trial included the following.
| • | CM-101 appeared to be safe and well tolerated when administered subcutaneously. Most reported adverse events observed were mild, with one unrelated serious adverse event reported. No significant injection site reactions were reported and no anti-drug antibodies were detected. |
| • | CM-101 administered subcutaneously demonstrated favorable pharmacokinetics and target engagement profiles as expected, and were similar to what the company has previously reported. |
| • | CM-101-treated patients showed greater improvements than the placebo group in a number of liver fibrosis-related biomarkers, including ProC-3, ProC-4, ProC-18, TIMP-1 and ELF. |
| • | A majority of CM-101-treated patients showed improvements in multiple liver fibrosis-related biomarkers—almost 60% of CM-101 patients were “multiple responders”, responding in at least three biomarkers at week 20, compared to no patients in the placebo group. |
| • | CM-101-treated patients with higher CCL24 levels at baseline showed greater reductions in fibrosis-related biomarkers than patients with lower levels of CCL24 at baseline. More CM-101-treated patients with higher CCL24 levels also were “multiple responders”, responding in three or more of the fibrosis-related biomarkers, compared to patients with lower CCL24 levels at baseline. These findings further add to the growing body of evidence validating the role of CCL24 in the pathophysiology of fibrotic liver disease. |
| • | A higher proportion of patients in the CM-101-treated group showed improvement in a physiologic measure of liver stiffness as compared to placebo (reduction of at least one grade of fibrosis score as assessed by the non-invasive elastography method known as FibroScan®). |
| • | After completion of the study, the unblinded data showed that patients in the CM-101-treated group had higher baseline levels of fibrosis compared to placebo patients. The impact of this difference on the results, if any, is unknown. |
We believe that the data from this trial provide important insights in support of the CM-101 development program, including the favorable safety and tolerability of CM-101 in patients with serious liver disease, confirmation of early signs of biomarker activity that are relevant for a number of fibro-inflammatory disorders, and support for the tolerability and pharmacokinetic data needed to assess next steps in the development of our SC formulation.
Results Reported for Clinical Study of CM-101 in Patients with COVID-19-Derived Lung Damage
On November 9, 2022, positive clinical data from an investigator-initiated clinical study assessing CM-101 activity and safety in hospitalized patients with severe lung injury derived from COVID-19 was presented at the 2022 Union Conference, an international conference on lung health. Some of the mechanisms underlying lung inflammation resulting from COVID-19 infection are similar to those seen in systemic sclerosis and other chronic diseases involving lung inflammation and fibrosis. The objective of the study was to evaluate the drug’s safety and activity in hospitalized COVID-19 patients with severe pneumonia, including its impact on biomarkers related to lung inflammation that are also relevant in SSc. The open label, single arm trial enrolled 16 hospitalized adult COVID-19 patients with severe respiratory involvement. All patients were receiving standard of care therapy. All were treated with a single 10mg/kg intravenous dose of CM-101 on the first day of the study and followed for 30 days.
Administration of CM-101 to this acutely ill patient population appeared safe and was well tolerated. CM-101 exposures and target engagement profiles were similar to what our researchers have seen in previous clinical studies of CM-101. Importantly, rapid reductions in serum biomarkers of lung inflammation, fibrogenesis and neutrophil activity were observed post-treatment with CM-101. Overall, this study confirmed and extended the safety and tolerability profile of CM-101 and demonstrated clinically relevant changes in biomarkers associated with lung inflammation and fibrogenesis, further supporting CM-101’s anti-inflammatory and anti-fibrotic effects. Moreover, we believe that these results add to the data suggesting that CM-101 has the potential to attenuate lung inflammation and fibrosis, further strengthening the rationale for treating SSc patients with this drug. These new clinical data also contribute to a growing body of evidence demonstrating CM-101’s anti-fibrotic and anti-inflammatory effects in varied organs including the lung, liver and skin.
Pipeline

Chemomab’s lead product candidate, CM-101, is a first-in-class humanized monoclonal antibody targeting CCL24 that is being advanced in two orphan indications: PSC and SSc. CCL24 has been extensively studied in airway inflammation and, more recently, Chemomab has demonstrated in preclinical studies and early clinical studies that it plays an important role in additional indication areas, including inflammation and fibrosis of the liver, skin and lung. Although found in low levels in blood or tissue samples taken from healthy volunteers, elevated levels of both CCL24 and its receptor CCR3, have been found in patients with PSC, SSc and NASH. CCL24 levels have even been correlated to different phases of disease. Chemomab expects that neutralizing CCL24 with an antibody will exert anti-fibrotic and anti-inflammatory effects in patients. CM-101 has been granted orphan drug designation by both the FDA and the EMA in its primary indications of PSC and SSc based on extensive preclinical and non-clinical data. This designation provides multiple benefits, including the potential for exclusive marketing and development rights for a period of time for these indications.
PSC is a rare, chronic cholestatic liver disease characterized by progressive inflammation, fibrosis, and destruction of the intrahepatic and extrahepatic bile ducts with no identifiable cause. Cholestasis is a symptom of liver injury and is characterized as the interruption of bile flow from hepatocytes to the intestine, which leads to bile acid accumulation in the liver, resulting in oxidative stress, inflammation, apoptosis, and fibrosis. PSC affects approximately 30,000-45,000 patients in the United States and is commonly associated with inflammatory bowel disease. Median survival is between 10-12 years.
Fibrosis and inflammatory responses induce a progressive spread of the fibrotic condition. No treatment aside from a liver transplant has been associated with change of the disease course or significant long-term improvement in the clinical outcome. PSC is a clear serious unmet medical need with no FDA approved therapeutics for which the current standard of care is inadequate.
SSc is a rare connective tissue disease characterized by excessive fibrosis and extracellular matrix accumulation in the skin, lung, and other visceral organs. The disease initiates with an early inflammatory phase involving the immune cell network, as well as endothelial cells. As the disease progresses, the inflammation increases and fibroblasts and myofibroblasts generate tissue fibrosis, while endothelial cells promote vascular injury, which can lead to skin fibrosis, interstitial lung disease, myocardial insufficiency, vascular obliteration, distal ulcerations, and gangrene. SSC affects approximately 75,000-100,000 patients in the United States. SSc has the highest mortality rate among the systemic rheumatic diseases and has high unmet need, as current treatments manage only disease manifestations and there is no disease modifying drug available.
Chemomab is primarily focused on the orphan indications PSC and SSc, but believes that it may have additional opportunities in other fibrotic-inflammatory disease areas such as idiopathic pulmonary fibrosis, or IPF and nonalcoholic steatohepatitis, or NASH. CM-101 has shown promising anti-fibrotic and anti-inflammatory effects in preclinical studies of liver fibrosis and PSC, with significant reductions in fibrotic genes, liver enzymes, bile acid and cholangiocyte proliferation, all reflecting a potential improvement in disease status. In preclinical studies of SSc, CM-101 reduced inflammatory and fibrotic injury resulting in reductions in dermal thickness, collagen concentration in the skin and the lung,
and immune cell infiltration in the lung.
Chemomab has completed two Phase 1a single ascending dose studies with intravenous, or IV, and subcutaneous, or SC, administrations of CM-101 in 40 healthy volunteers. The drug was shown to be safe and well-tolerated, with a PK profile supporting dosing once every 2-4 weeks. The Company also completed a Phase 1b multiple administration ascending dose study in 16 non-alcoholic fatty liver disease (NAFLD) patients, expanding its safety, tolerability, and pharmacodynamics database with patients with early liver disease. Early evidence of an anti-fibrotic effect was also seen in this study.
Chemomab recently reported topline results from its Phase 2a randomized, double-blind, placebo-controlled study in patients with liver fibrosis derived due to NASH. The trial met its primary endpoint of safety and tolerability, and CM-101 demonstrated promising activity in secondary endpoints that included pharmacokinetic and target engagement profiles of the SC formulation as well as a range of liver fibrosis biomarkers and physiologic assessments measured at baseline and at week 20.
The randomized, placebo-controlled trial enrolled 23 NASH patients with stage F1c, F2 and F3 disease who were randomized to receive either CM-101 or placebo. Patients received eight doses of 5 mg/kg of study drug administered by SC injection once every two weeks, for a treatment period of 16 weeks. Key findings of the CM-101 Phase 2a trial included the following.
| • | CM-101 appeared to be safe and was well tolerated when administered subcutaneously. Most reported adverse events observed were mild, with one unrelated serious adverse event reported. No significant injection site reactions were reported and no anti-drug antibodies, or ADAs, were detected. |
| • | CM-101 administered subcutaneously demonstrated favorable pharmacokinetics and target engagement profiles as expected, which were similar to what the company has previously reported. |
| • | CM-101-treated patients showed greater improvements than the placebo group in a number of liver fibrosis-related biomarkers, including ProC-3, ProC-4, ProC-18, TIMP-1 and ELF. |
| • | A majority of CM-101-treated patients showed improvements in more than one liver fibrosis-related biomarker—almost 60% of CM-101 patients responded in at least three biomarkers at week 20, compared to no patients in the placebo group. |
| • | A higher proportion of patients in the CM-101-treated group showed improvement in a physiologic measure of liver stiffness as compared to placebo (reduction of at least one grade of fibrosis score as assessed by the non-invasive elastography method known as FibroScan®). |
| • | CM-101-treated patients with higher CCL24 levels at baseline showed greater reductions in fibrosis-related biomarkers than patients with lower levels. Multiple fibrosis-related biomarkers showed more pronounced reductions in CM-101-treated patients who had higher CCL24 levels at baseline than in patients with lower CCL24 baseline levels, adding to the growing body of evidence validating the role of CCL24 in the pathophysiology of fibrotic liver disease. |
| • | After completion of the study, the unblinded data showed that patients in the CM-101-treated group had higher baseline levels of fibrosis compared to placebo patients. The impact of this difference on the results, if any, is unknown. |
Data from this trial provide important insights in support of the CM-101 development program, including the favorable safety and tolerability of CM-101 in patients with serious liver disease, confirmation of early signs of biomarker activity that are relevant for a number of fibro-inflammatory disorders, and additional tolerability and pharmacokinetic data needed to assess next steps in the development of our current subcutaneous formulation.
Recently, Chemomab also reported positive clinical data from an investigator-initiated clinical study assessing CM-101 activity and safety in hospitalized patients with severe lung injury derived from COVID-19. The objective of the study was to evaluate the drug’s safety and activity in hospitalized COVID-19 patients with severe pneumonia, including its impact on biomarkers related to lung inflammation that are also relevant in systemic sclerosis. The open label, single arm trial enrolled 16 hospitalized adult COVID-19 patients with severe respiratory involvement. All patients were receiving standard of care therapy. All were treated with a single 10mg/kg intravenous dose of CM-101 on the first day of the study and followed for 30 days. Administration of CM-101 to this acutely ill patient population appeared to be safe and was well tolerated. CM-101 exposures and target engagement profiles were similar to what our researchers have seen in previous clinical studies of CM-101. Importantly, rapid reductions in serum biomarkers of lung inflammation, fibrogenesis and neutrophil activity were observed post-treatment with CM-101. Overall, this study confirmed and extended the safety and tolerability profile of CM-101 and demonstrated clinically relevant changes in biomarkers associated with lung inflammation and fibrogenesis, further supporting CM-101’s anti-inflammatory and anti-fibrotic effects.
Chemomab is currently conducting a randomized, double-blind, placebo-controlled study of CM-101 in PSC patients, and is planning to initiate a new study in SSc patients around midyear of 2023.
The Phase 2 randomized, double-blind, placebo-controlled SPRING study is currently enrolling patients with PSC who are treated with CM-101 or placebo for 15 weeks. The company has expanded the trial by implementing a dose finding component to the CM-101 development program and evaluating a higher dose level of CM-101 (20mg/kg) to support future potential registrational trials. In addition, the company is adding an open-label extension to the trial to evaluate the safety, tolerability and durability of effect over longer treatment durations.
Chemomab is currently conducting a randomized, double-blind, placebo-controlled study of CM-101 in PSC patients, and is planning to initiate a new study in SSc patients around midyear of 2023.
Chemomab may also explore CM-101 in other indications, where the dual activity of CM-101 acting on both inflammation and fibrosis could provide new avenues for treating inflammatory and fibrotic conditions.
Chemomab was founded in 2011, based on a novel discovery originating from the Sourasky Medical Center in Tel-Aviv, Israel, where Professor Jacob George first identified CCL24 as a key regulator of unstable plaque formation in atherosclerotic patients. In its early years, Chemomab focused on research directed at clarifying the role and effectiveness of CCL24 blockade. In 2015, Chemomab selected its proprietary lead product candidate, CM-101, and started product development directed towards human testing.
Chemomab has assembled an executive team with highly relevant experience in inflammation and fibrosis, and biologics drug discovery and clinical development. Adi Mor, Ph.D., Chemomab’s Chief Scientific Officer and Co-founder, has 15 years of experience in Immunology and has led the CM-101 program from discovery stage into Phase 2 clinical studies. Matthew Frankel, M.D., Chemomab’s Chief Medical Officer, brings deep experience in clinical development. Dr. Frankel, who is based in the U.S., has more than 25 years of experience in the discovery and clinical development of novel therapeutics and has held senior executive roles at both global pharmaceutical and biotechnology companies. Jack Lawler, Chemomab’s Vice President of Global Development Operations, is highly experienced in managing clinical trials across a wide range of indications and geographies.
Company strategy
Chemomab aims to become a world-leading company for the treatment of diseases involving inflammation and fibrosis, developing novel therapies across a wide range of indications. To achieve this, the company is focused on the following key strategies:
| • | Advance Chemomab’s lead product, CM-101, for the treatment of PSC and SSc, through clinical development to approval |
The clinical development plan of lead product candidate CM-101 was optimized to maximize the clinical information obtained, generating additional important data to support future advancement to registration trials, and decreasing the overall risk in the CM-101 clinical development program in the lead indications of PSC and SSc, as well as potentially in additional indications where the scientific rationale is strong.
The Company expects that the current trial designs will provide important data on the clinical dose response relationship to inform the broader development program and to identify the optimal dose to advance in later PSC and SSc trials. The study design is also expected to generate proof of concept data on clinically relevant aspects of SSc, a complex rheumatological disorder, to best inform the development path for a novel, first-in-class therapeutic like CM-101, along with relevant safety and tolerability data to support the evaluation of higher doses and inform decisions on next steps in the development of the subcutaneous formulation.
| • | Expand Chemomab’s next generation pipeline |
Based on the know-how, knowledge and experience it has gathered in diseases involving both inflammation and fibrosis, Chemomab is assessing opportunities to expand its pipeline with novel products developed against new targets. Chemomab may also explore targeting CCL24 with additional, complementary fibrotic and/or inflammatory mechanisms, including acquiring or in-licensing innovative product candidates.
| • | Selectively evaluate partnership opportunities |
Chemomab continuously explores partnership opportunities to advance CM-101 development in PSC and SSc, identifying companies with drugs (either approved or in development) that could possibly be combined with CM-101, extending the development of CM-101 to new indications beyond PSC and SSc, and seeking additional significant commercial or drug development capabilities that may accelerate CM-101’s time to market. The Company also continues to evaluate opportunities to in-license or acquire other novel investigational therapeutics addressing relevant fibro-inflammatory disorders.
| • | Explore opportunities for CM-101 in additional inflammatory/fibrotic indications |
Chemomab continuously evaluates the potential benefit of CM-101 outside of its two lead indications, PSC and SSc, in order to maximize the product’s potential. CM-101 has shown anti-fibrotic activity in animal models and human tissue studies of IPF and NASH. Chemomab will continue to assess ways to leverage the dual anti-inflammatory an anti-fibrotic activity of CM-101 into new disease areas and to form additional collaborations with global medical researchers and drug developers.
| • | Strengthen Chemomab’s intellectual property portfolio |
Chemomab believes that it has developed a strong intellectual property portfolio and will continue to seek, maintain, and defend its patent rights, whether developed internally or licensed to protect and enhance the proprietary technology, inventions, and improvements that are commercially important to the development of its business proprietary position in the field of inflammation and fibrosis.
Fibrosis and inflammation
Tissue damage activates a repair process that includes acute inflammation followed by either successful complete repair or tissue replacement by fibrosis. However, persistent and repeated damage can result in continuous activation of the repair process leading to chronic inflammation, progressive tissue fibrosis and sclerosis.
Fibrosis is an accumulation of non-functional tissue and can occur in many different tissues, including lung, liver, kidney, muscle, skin and the gastrointestinal tract, resulting in a number of chronic fibrotic conditions. Liver fibrosis is the process of excessive accumulation of extracellular matrix proteins, predominantly collagen, which occurs as the result of liver injury. In cases of acute temporary damage, these changes are transient and liver fibrosis may resolve. In chronic cases, however, the liver damage persists and chronic inflammation and accumulation of the extracellular matrix eventually lead to cirrhosis. The various fibrotic manifestations in conditions like SSc are still not well understood. Disease progression is characterized by an early inflammatory onset followed by tissue fibrosis, vascular injury and organ damage. Fibrosis, and specifically lung fibrosis, is the main cause of disease progression and mortality in SSc, although manifestation of the disease in other organs can cause patients considerable distress and adversely impact their health and their quality of life.
Fibrosis and inflammation are intrinsically linked; a healthy inflammatory response is necessary for efficient wound healing, however, a prolonged response can contribute to the pathogenesis of fibrosis. The inflammatory response during chronic liver injury is a dynamic process with intrahepatic accumulation of diverse immune cells. Recruitment and infiltration of these cells to the liver and their localization is mainly determined by chemokines and cytokines that are produced by hepatocytes, immune cells, biliary epithelial cells, and endothelial cells. Notably, activated liver fibroblasts, the hepatic stellate cells, or HSCs, secrete various chemokines, thereby contributing to the ongoing immune response during fibrotic liver diseases. Similarly, for SSc, the early inflammatory phase leading to fibrosis in multiple organs of the body includes activation of the immune cell network of lymphocytes, eosinophils, and monocytes, as well as endothelial and endothelial progenitor cells. In the advanced SSc phase, fibroblasts and myofibroblasts take the lead to generate tissue fibrosis.
Chemokine involvement in inflammation and fibrosis
Chemokines are a group of small signaling proteins thought to be involved in the etiology, or causation, of multiple inflammatory diseases. They are not only implicated in immune cell recruitment during inflammation, but also contribute to immune surveillance, direct cells to target organs in homeostasis, and exert pleiotropic, or diverse, effects on non-immune cells, for instance, directly influencing the functionality of fibrogenic cells. Chemokines and their corresponding chemokine receptors are key players in orchestrating the sequential influx of immune cells into damaged or diseased organs, driving inflammatory responses to specific triggers.
In the liver, chemokines have a key role in the development of inflammation and wound healing responses, which can lead to either resolution of liver injury or promote, if ongoing, maladaptive responses with chronic inflammation, fibrosis, and development of clinically manifest liver disease. Although the pathophysiology underlying PSC has not yet been fully clarified, animal models of PSC have contributed to dissecting the molecular basis of this disease and highlighting the role of cytokines and chemokines as important pathogenetic mediators of liver inflammation and fibrosis. Recently published studies demonstrated that in most of the processes suggested for the onset and development of PSC, chemokines and chemokine receptors play a key role. HSCs may be the main producers of cytokines and play an initial role in the progression of liver fibrosis by attracting different types of immune cells, resulting in further production of cytokines and liver injury in a vicious disease cycle. Extensive proliferation, trans-differentiation and activation of HSCs result in ongoing chronic tissue remodeling and severe fibrosis. In addition, chemokines are also involved in promoting polarization of the recruited immune cells. Therefore, chemokines may participate in PSC by promoting migration of inflammatory and fibrotic cells, by activating inflammatory and fibrotic cells locally, or by inducing cytokines that promote collagen and matrix deposition.
Likewise, in SSc pathogenesis, chemokines foster migration and activation of inflammatory and fibrotic cells, inducing the secretion of cytokines that promote collagen and matrix deposition in affected organs. Indeed, patients with SSc exhibit increased systemic levels of proinflammatory chemokines and some of these have also been shown to correlate with limited or diffuse cutaneous disease phenotype and/or to organ-specific pathology such as lung disease or skin vascular inflammation.
The role of CCL24
CCL24 is a chemokine that promotes various types of cellular processes that regulate inflammatory and fibrotic activities through the CCR3 receptor. This chemokine is known to be expressed by activated T-cells, monocytes, epithelial cells and endothelial cells, as well as by activated fibroblasts. CCL24 induces chemotaxis and activation of CCR3-expressing cells, including immune cells and fibroblasts.
Chemomab has been the driving force in establishing the role of CCL24 in the pathogenesis of PSC and SSc, however, others have highlighted its contribution to other indications. For example, published work has shown that both CCL24 and CCR3 are involved in lung and skin inflammation and fibrosis. CCR3 is robustly expressed on eosinophils and recent data has suggested that eosinophilic inflammation may be involved in the pathogenesis and progression of SSc. For example, in SSc patients, eosinophil counts, but not total leukocytes, were significantly higher than in patients with other connective autoimmune diseases. Eosinophil counts correlated positively with both interstitial lung disease severity and the modified Rodnan skin thickness score, or mRSS.
Notably, CCR3 was shown to be expressed on oral and dermal fibroblasts where it modulates wound healing and tissue remodeling processes. A recent academic study also demonstrated overexpression of CCR3 on monocyte populations isolated from SSc patients. CCL24 was shown to be involved in proinflammatory reactions, specifically contributing to the type 2 immune reaction involving Th2 lymphocytes and M2 macrophages that were shown to be present in skin lesions of SSc patients. Accordingly, CCL24 was found to play a dominant role in inducing profibrotic effects and to be overexpressed in fibrotic lungs and bronchoalveolar lavage fluid from patients with idiopathic pulmonary fibrosis, a disease sharing similar lung dysfunction features with SSc. Furthermore, CCL24 was shown to promote collagen production in human lung fibroblasts and to be constitutively expressed by dermal fibroblasts.
Prior studies support the role of CCL24/CCR3 signaling in the pathogenesis of SSc and these findings have been further explored by Chemomab in SSc and for the first time, in PSC.
CCL24 is a critical mediator promoting inflammation and fibrosis
Challenges to drug development in fibrosis and inflammation
Successful treatment of fibrotic disorders has in large part remained elusive, primarily due to incomplete understanding of the complexity and multi-mechanism contributions to disease progression. This has complicated preclinical investigations for new products and new targets, with animal models having limited resemblance to human disease. Additionally, preclinical animal data is often of short treatment duration and does not capture the effects of treating chronic fibrotic indications. This is particularly applicable to complex, orphan indications like SSc, where there is still no approved standard of care or proven target mechanism. Most drug approvals in this space have been focused on fibrosis of the lungs i.e., idiopathic pulmonary fibrosis, or IPF, interstitial lung disease, or ILD, and pulmonary arterial hypertension, or PAH.
Most approved anti-fibrotic products target extracellular components, given their biological accessibility, and inhibition of receptors and ligands preventing downstream signaling is considered to be a potentially effective option for alleviating fibrosis. PDGF and TGF-β are commonly studied targets in fibrosis and there are two approved products that target these pathways, pirfenidone and nintedanib. Both pirfenidone and nintedanib are approved for the treatment of IPF, with nintedanib also recently approved for treatment of systemic sclerosis associated interstitial lung disease and chronic fibrosing interstitial lung diseases. Due to the strong associations between inflammation and fibrosis, companies have devoted efforts to anti-inflammatory drugs with the hope that reduction in inflammation will attenuate fibrosis. For example, companies have targeted TNF-α, a commonly explored anti-inflammatory mechanism in fibrotic indications. Despite the success of targeting cytokines, inflammatory factors and immune cells in pure inflammatory autoimmune diseases, such as blocking TNF-α, these results have generally not been reproduced in studies targeting inflammatory fibrotic indications. Nonetheless, tocilizumab, a monoclonal antibody targeting IL-6R, was recently approved for the treatment of ILD associated with systemic sclerosis. Treatments that inhibit certain pure anti-fibrotic pathways, such as nintedanib and pirfenidone, have resulted in limited clinical benefit. Chemomab believes that these results highlight the importance of a dual mechanism that, with adequate selectivity, is designed to target inflammatory processes and also directly prevent fibrosis resulting in blockage of multiple disease-contributing mechanisms.
Notwithstanding challenges in the field of fibrosis and inflammation, there is significant and growing industry interest given the associated unmet medical need and the continuing opportunity to identify better therapeutic targets. For example, in 2019 Novartis completed two transactions related to the treatment of NASH, a liver metabolic fibrotic disease. It acquired IFM Tre for NLRP3 antagonists for a $310 million upfront payment and total potential consideration of $1.5 billion and licensed an integrin inhibitor from Pliant Therapeutics for an $80 million upfront payment. Additionally, Gilead Sciences licensed two preclinical programs, one in NASH for a $15 million upfront payment (total potential consideration of $785 million) and the other for TGF-β inhibitors in fibrosis for an $80 million upfront payment and total potential consideration of $1.4 billion. In 2020, Roche acquired Promedior for a $390 million upfront payment and total potential consideration of $1 billion in milestones for its Phase 2 product in pulmonary fibrosis, and Bayer partnered with Recursion Pharmaceuticals to develop and commercialize preclinical-stage small molecule treatments for fibrotic conditions for a $30 million upfront payment and total potential consideration of $1 billion. Boehringer Ingelheim also acquired Enleofen Bio in a deal potentially worth $1 billion for its NASH and ILD anti-IL11 platform. More recently, Mediar, a high-profile start-up targeting novel mechanisms for fibrotic diseases, reported a $105 million Series A financing.
Targeting chemokines as a treatment for fibrotic indications
Chemomab believes that its approach, selectively targeting fibrotic conditions by attenuating both inflammation and fibrosis, may be an optimal approach for both effectiveness and reduction of toxicity. As central regulators of initiation and progression of fibrotic disorders, chemokines are an ideal target to impact both inflammation and fibrosis. Some chemokines are also disease-specific, allowing for potential selectivity.
Chemokine receptors, or CCRs, have been more extensively studied as drug targets in fibrotic conditions compared to chemokine ligands, however, the therapeutic effects of CCR inhibitors have generally fallen short in the clinic. Pharmaceutical companies have previously explored the CCL24 ligand receptor, CCR3, and its other ligands CCL7 and CCL11, with small or large molecule inhibitors. These programs were directed at inhibiting eosinophilic trafficking in respiratory and allergic inflammation, however, despite promising preclinical data, most programs were discontinued largely due to poor safety profiles and limited efficacy of the antagonist used. To Chemomab’s knowledge, only Alkahest has an active program that explores CCR3 inhibition, which is under license from Boehringer Ingelheim and is being developed as a treatment for wet AMD. In contrast, Chemomab believes CCL24 presents a more promising opportunity. Unlike other CCR3 ligands, CCL24 binds only to the CCR3 receptor and is also organ/disease-specific, which together could provide enhanced selectivity and tolerability. For example, in PSC, CCL24 is elevated in the liver and cholangiocytes (bile duct epithelia) and immune cells that play a key role in the progression of the disease. Likewise, elevation of CCL24 has been shown in fibrotic lungs and bronchoalveolar lavage fluid from patients with idiopathic pulmonary fibrosis, a disease sharing similar lung dysfunction features with SSc and which recently was correlated, by Chemomab, with disease severity and lung involvement in a cohort of SSc patients from the United Kingdom. Furthermore, CCL24 is constitutively expressed by skin and dermal fibroblasts. The use of an antibody in targeting this chemokine is a novel approach to targeting fibrosis.
Chemomab’s expertise and approach to drug discovery
Chemomab is a clinical stage biotechnology company focused on the discovery and development of novel drugs to address fibrotic indications with unmet medical needs. CCL24 is a key target promoting fibrosis as it regulates the two main processes that drive fibrosis: fibroblast activation and immune cell migration and activation. Using Chemomab’s expertise in monoclonal antibody, or mAb, development and deep knowledge of chemokine biology, Chemomab is developing CM-101, a proprietary, first-in-class, fully humanized mAb that through research and studies to date, has been shown to neutralize CCL24 and by so doing inhibits its disease-related functions in both inflammation and fibrosis. This represents an innovative approach to anti-fibrotic drug discovery and is a key differentiator for Chemomab. The ability of CM-101 to directly attenuate fibroblast activation and concurrently attenuate recruitment of immune cells is novel and could address a wide range of hard-to-treat fibrotic diseases.
Chemomab’s ongoing collaborations are complementary to both preclinical and clinical aspects of research and development. Chemomab has created an extensive panel of in vitro, ex vivo and in vivo assays which it has used to further the understanding of fibrotic processes together with the role of CCL24 in various diseases and the effects of its neutralization by CM-101. These assays have allowed Chemomab to sequentially explore target validation and proof of mechanism in disease relevant human and animal samples that continues to de-risk the translation of CM-101 into the clinic.
Target expression and engagement
Chemomab regularly collaborates with leading academic centers around the world to investigate the role of CCL24 and CM-101 in various indications. For example, Chemomab works with The Royal Free Hospital, or RFH, in London, and Birmingham University in Birmingham, United Kingdom to access liver biopsy and serum samples from patients with PSC. Using immunohistochemistry and florescence microscopy to stain CCL24 and CCR3, it explores the expression patterns of these targets in disease relevant human samples and compares them to healthy volunteers. Similarly, Chemomab has tested biopsies of SSc patients through a collaboration with the University of Florence in Italy and serum samples of SSc patients through a collaboration with Leeds University in the UK.
Proof of mechanism
Chemomab explores fibroblast activation and immune cell recruitment in response to CM-101 treatment through inhouse ex vivo and in vitro assays. Chemomab has executed multiple validated genetic and treatment-based disease models in fibrotic and inflammatory indications in which it has investigated CM-101’s effects. Additionally, as part of a collaboration with Nordic Biosciences, Copenhagen, Denmark, Chemomab has gained access to proprietary tools and expertise to explore the effects of CM-101 on key fibrogenesis and fibrolysis biomarkers. Nordic Biosciences is a world-leading extracellular matrix specialist and continues to contribute additional analyses to Chemomab’s clinical samples.
Chemomab has created a broad array of biological assays to explore CCL24 and CM-101
Chemomab may explore next-generation biologic products, and, based on its wide database of patient samples and extensive knowledge and experience in fibrosis, may identify targets that could complement CCL24 inhibition. Next-generation assets may therefore be dual targeting and would be screened through the panel of assays available at Chemomab that evaluate target expression in fibrotic tissues as well as the anti-fibrotic activity of potential candidates. Similar to CM-101, this process would establish proof-of-biological-mechanism in both animal models and human tissue prior to commencing product development and initiating clinical studies.
The Chemomab pipeline
CM-101 in PSC and SSc
Chemomab’s lead product, CM-101, is a first-in-class humanized monoclonal antibody targeting CCL24 that is being developed initially for the treatment of PSC and SSc, with potential future opportunities in other fibrotic-inflammatory indications. Chemomab has completed two Phase 1a studies of CM-101 in healthy volunteers as well as a Phase 1b safety, tolerability and proof-of-mechanism study in NAFLD patients and a Phase 2a liver fibrosis biomarker study in NASH patients with liver fibrosis, which was recently reported. Topline results showed favorable safety and tolerability profiles for CM-101 in patients with serious liver disease, confirmed early signs of biomarker activity that are also relevant for a number of fibro-inflammatory disorders, and reinforced tolerability and pharmacokinetic data relevant to the development of our current subcutaneous formulation.
A Phase 2 study in PSC is now ongoing in Europe, the United States and Israel and is currently expanding to include an additional dose level cohort as well as an open label extension. A global Phase 2 study in SSc that will assess the clinical and biological effects of CM-101 in this patient population is expected to begin enrolling patients around midyear of 2023.
Primary Sclerosing Cholangitis
PSC is a progressive, rare, and chronic cholestatic liver disorder that is characterized by thickening, inflammation, and fibrosis of the intra- and extra-hepatic bile ducts. This generally leads to cholestasis, liver damage, cirrhosis, and eventually liver failure. The exact cause of PSC remains mostly unknown; however, immune system dysregulation, genes, viruses, and bacteria may be involved. PSC is commonly associated with inflammatory bowel disease, or IBD. Approximately three in every four individuals with PSC also have ulcerative colitis. Most individuals affected with PSC are adults with an average age of 40 years at diagnosis; however, it may also occur in children. Disease progression, symptoms, and severity may vary greatly between individuals. Patients in the initial stages of PSC are generally asymptomatic or have only mild symptoms.
Abdominal discomfort, fatigue, and pruritus, or itching, are common initial symptoms of PSC that can be severe and debilitating. The initial step in diagnosing PSC is to evaluate liver enzyme levels through blood tests. Physicians will then confirm a diagnosis with cholangiography ultrasound and, in rare cases, a liver biopsy. As the disease progresses, bile flow from the liver is obstructed and is subsequently absorbed into the bloodstream leading to the yellowing of the mucous membranes, whites of the eyes, and skin. Furthermore, individuals may also experience abdominal pain, malaise, light-colored stools, nausea, dark urine, weight loss, and/or hepatomegaly or splenomegaly. PSC patients have a 40-fold increased risk of liver cancer and a 400-fold increased risk of cholangiocarcinoma, and the disease may lead to other conditions including osteoporosis, bacterial cholangitis, portal hypertension, bleeding, as well as vitamin deficiencies.
There are currently no specific medical therapies that can alter or cure the course of the disease; instead, available treatments are directed towards slowing the progression of PSC and treating symptoms. In certain individuals, endoscopic surgery may be performed to enlarge the narrowed bile ducts and to remove blockages. Complications due to vitamin deficiencies can be prevented with the help of vitamin supplements, while infections and inflammation can be controlled by using antibiotics. Cholestyramine and UCDA can be effective in managing itching and can be used with or without antihistamines. Patients with advanced symptoms such as end-stage liver disease, recurrent bacterial cholangitis and intractable pruritus, will often undergo liver transplantation, however, in 30% of cases, PSC will recur even after liver transplantation. The median survival is 10-12 years without intervention.
Systemic Sclerosis
SSc is an autoimmune inflammatory condition that results in widespread fibrosis and vascular abnormalities affecting the skin, lungs, gastrointestinal tract, heart and kidneys. Other key features of SSc include thickening and hardening of the skin, autoantibody production and abnormal nail fold capillaries. The underlying mechanisms that cause SSc are complex and for the most part unknown but most likely involve a combination of factors including the immune system, genetics, and environmental triggers. Various pathways are involved in the pathogenesis of SSc including cytokines that injure blood vessels, growth factors that stimulate collagen, integrin signaling, morphogen pathways, and co-stimulatory pathways. SSc is generally diagnosed between the age of 30 and 50 years and is more prevalent in women.
Given that SSc can affect many different parts of the body there are a multitude of different symptoms of the disease. The most widely observed symptoms include fatigue, arthralgia, and myalgia. However, the earliest sign is often the Raynaud phenomenon in which the body’s normal response to cold or emotional stress is exaggerated, resulting in abnormal spasms in arterioles. Cutaneous features include sclerosis of the skin, particularly the face and hands. Gastrointestinal symptoms of the upper tract include acid reflux and of the lower tract include bloating, nausea and incontinence. Cardiopulmonary presentations include interstitial lung disease, pulmonary arterial hypertension and cardiac scleroderma. Renal and ocular symptoms can also present and 20% of SSc patients have an overlapping diagnosis with other connective tissue diseases and can develop arthritis, lupus or myositis. SSc is subdivided into two main types related to the distribution of skin involvement: diffuse cutaneous (two-thirds of cases) and limited cutaneous. Diffuse SSc, or dcSSc, is rapidly progressive with more significant organ involvement.
There is no cure for SSc. Established treatments can help with symptoms and may modify the disease outcome only if given early in the disease course. Prescribed medications, used off-label, primarily focus on suppressing inflammation with NSAIDs and dilating abnormal or constricted blood vessels with losartan, sildenafil, iloprost and SSRIs, or selective serotonin reuptake inhibitors, as well as treatments to manage individual organ involvement. The only three drugs that are approved for the treatment of SSc symptoms are bosentan by Actelion Pharmaceuticals, approved in Europe for the prevention of digital ulcer development, nintedanib by Boehringer Ingelheim, and tocilizumab by Roche, approved in the United States, Europe and Japan for the treatment of SSc associated interstitial lung disease. The clinical course of SSc is determined by the extent of vascular and fibrosis complications and has the highest mortality rate among the systemic rheumatic diseases. Forty percent of patients die within 10 years of disease onset, with pulmonary involvement being the leading cause of death.
Chemomab’s CM-101may have disease-modifying potential
The dual anti-fibrotic and anti-inflammatory activity of CM-101 enables the targeting of a wide range of pathogenic mechanisms and may afford patients a new treatment that may have a more impactful effect on disease progression.
Targeting CCL24 offers a dual activity approach
In order to understand CCL24’s role in disease pathophysiology, Chemomab has collected data on CCL24 levels from patients with multiple fibrotic-inflammatory indications, including those with PSC, SSc and NASH. PSC patients’ liver biopsies and SSc skin samples were stained for CCL24 and its receptor, CCR3. Blood samples taken from PSC and SSc patients were used to further evaluate the role of the CCL24-CCR3 axis exploring levels of circulating CCL24 and CCR3. To explore the influence of CCL24 on disease status, CCL24 serum levels were correlated with fibrotic biomarkers and disease severity markers.
CCL24 levels in liver biopsies from PSC patients
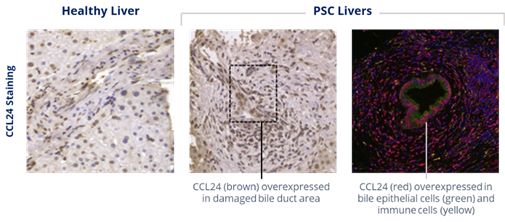
PSC pathology generally initiates with bile duct damage leading to cholestasis, bile duct inflammation and fibrosis and finally to substantial liver damage. Chemomab assessed the accumulation and cellular localization of CCL24 in livers of PSC patients focusing on CCL24 levels in the periductal damaged zone that is most relevant to disease pathology. CCL24 was mainly found in inflammatory cells in the liver of PSC patients. Due to the robust liver inflammatory insult in PSC, reflected by massive accumulation of resident and recruited immune cells in the periductal space, CCL24 positive staining was extensive. Specific and robust CCL24 staining was also shown in cholangiocytes, the epithelial cells of the bile ducts. Activated myofibroblasts that surround the bile ducts, whether they originate from hepatic stellate cells or portal fibroblasts, are the main drivers of the excess extracellular matrix accumulation in this area, comprising the unique “onion ring” shape seen in PSC liver sections. The collective expression pattern shows high CCL24 levels in areas that are most affected in PSC and highlights its central role in PSC related liver pathology.
Elevated CCL24 staining in liver biopsies from PSC patients
CCR3 levels in liver biopsies from PSC patients
To evaluate the levels of CCR3, the receptor of CCL24, and identify the cells that can potentially respond to CCL24 secretion, biopsies were stained for CCR3. As was seen for similar studies with CCL24, specific CCR3 staining was evident in cholangiocytes, surrounding immune cells and fibroblasts.
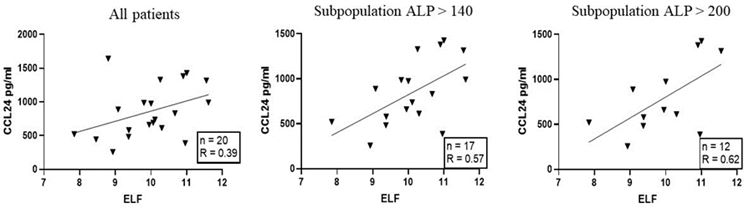
CCL24 levels in serum and correlation to a fibrotic biomarker in PSC patients
Together with the Royal Free Hospital (RFH), Chemomab analyzed serum levels of CCL24 in PSC patients at various stages of disease. CCL24 levels showed a positive correlation to the liver fibrosis biomarker ELF score, which is a commercially available test that reflects liver fibrosis stage based on serum concentrations of several fibrosis-related proteins. When dividing this cohort of PSC serum samples by ALP levels, a circulating parameter used for monitoring PSC activity, there was a stronger relation of the fibrotic biomarker and CCL24 with increased ALP.
CCL24 levels correlate with ELF score
CCR3 levels in circulating PBMCs in PSC patients
Chronic liver inflammation is driven in most hepatic injuries by several different immune cell populations originating from either resident hepatic immune cells or recruited cells from the circulation to the damaged site. In collaboration with the Kaplan Medical Center, Israel, Chemomab explored systemic changes of CCR3, given that this could impact cell recruitment to the PSC damaged liver. PBMCs from ten PSC patients and healthy controls were stained for expression of CCR3 and demonstrated that levels were significantly higher in PSC patient samples compared to healthy donors.
PSC patients showed significantly higher expression of CCR3 on PBMCs
CCL24 and CCR3 levels in skin biopsies from SSc patients
Chemomab analyzed skin samples from diffuse SSc patients and healthy volunteers and the SSc samples showed elevations in CCL24 and CCR3. Specifically, higher accumulation of CCL24 on immune cells skin infiltration was shown in the SSc samples and CCR3 was evident in skin fibroblasts, immune cells and endothelial cells. These elevations led to a CCL24-mediated robust activation of CCR3 expressing cells, which enhances the recruitment of immune cells and fibroblasts to the diseased organ.
SSc patients showed elevated levels of CCL24 in skin tissue
SSc patients showed elevated levels of CCR3 in skin tissue
CCL24 levels in serum samples from SSc patients and correlation with fibrotic biomarkers
Chemomab researchers analyzed SSc serum samples that showed that CCL24 levels were significantly increased in SSc patients compared with healthy individuals. Notably, in diffuse SSc patients, CCL24 levels were fourfold higher than in healthy control patients. Additionally, the levels of CCL24 were correlated with a biomarker of SSc severity, anti-topoisomerase, an autoantibody seen in diffuse SSc patients.
Preclinical Efficacy of CM-101 in models of PSC
Preclinical experiments in models of PSC
| • | Human hepatic stellate cells demonstrated reduced transition to myofibroblasts following incubation of CM-101 with CCL24. |
| • | Human hepatic stellate cells showed reduced motility towards CCL24 following treatment with CM-101. |
| • | CM-101 demonstrated in vivo activity on liver fibrosis and cholangiocyte proliferation induced by bile duct ligation in the Sprague Dawley rat model. |
| • | CM-101 (D8-a murine surrogate of CM-101) inhibits the progression of liver fibrosis and bile duct damage in a chronic cholangitis cholestasis model using the hepatobiliary toxin ANIT. |
| • | CM-101 (D8) reduces bile duct epithelial cell (cholangiocyte) proliferation, collagen deposition, macrophage infiltration, liver enzymes, bile acid and circulating inflammatory monocytes in an experimental cholangitis model in MDR2 knockout mice. |
| • | CM-101 reduces liver enzymes, fibrosis, collagen, and fibrotic gene expression in a TAA-induced liver fibrosis model in rats. |
| • | CM-101 (D8) prevented fibrosis and inflammation in a TAA-induced liver fibrosis model in mice. |
Results from the multi-drug resistant 2, or MDR2, knock out mouse model that reflects sclerosing cholangitis and the thioacetamide (TAA) rat model reflecting liver fibrosis are described below.
CM-101 demonstrates anti-cholestatic, anti-inflammatory, and anti-fibrotic activity in MDR2 knock out mouse model in vivo
Mice with targeted disruption of the MDR2, transporter gene develop chronic and progressive hepatic sclerosing cholangitis that closely resembles PSC and therefore this model has been extensively used to study the pathogenesis and progression of PSC. Using MDR2 knockout mice (six weeks of age), the Company tested the ability of CM-101 (D8) to attenuate PSC related symptoms. Mice (n=15/group) received either vehicle control, or CM-101 10 mg/kg SC twice weekly during weeks 6-12 following established disease and were sacrificed at the end of week 12. In this study mice were tested for changes in alkaline phosphatase, or ALP, bile acid levels, collagen deposition (histology, Sirius red), macrophage presence in the liver and cholangiocyte proliferation. The Company observed a significant decrease in all three core pathologies that play a role in PSC: inflammation, fibrosis and cholangiocyte proliferation after CM-101 (D8) treatment compared to non-active treatment. Reduction in the serum markers that represent the cholestatic state, ALP and bile acid, was also observed.
CM-101 reduces liver fibrosis, inflammation and bile duct epithelial proliferation in MDR2 knockout model
CM-101 demonstrates in vivo activity in a thioacetamide induced liver fibrosis model in rats using a therapeutic model
To assess potential efficacy of CM-101 on liver fibrosis, the Company used the TAA-induced liver fibrosis model. Liver fibrosis was induced by intraperitoneal administration of TAA at a dose of 250 mg/kg twice weekly for eight weeks. Rats (n=10/group) received either vehicle control or CM-101 2.5 mg/kg IV twice weekly during weeks four to eight following established fibrosis and were sacrificed at week eight. After eight weeks of TAA treatment, all vehicle-treated animals had developed liver fibrosis, as confirmed by Sirius-red-stained liver histology.
CM-101 reduces fibrosis in rat livers
Plasma ALP, ALT, and AST levels decreased in the CM-101 study arm. Liver collagen content and fibrotic areas were significantly reduced in the CM-101 treated group compared to non-active treatment. CM-101 was also shown to reduce fibrotic markers in the TAA treated rats.
Efficacy of CM-101 in models of SSc
Preclinical experiments in models of SSc
| • | CM-101 reduces SSc serum-induced dermal fibroblast activation and transition to myofibroblasts and interferes with endothelial cell activation. |
| • | CM-101 treatment attenuated skin fibrotic remodeling in the bleomycin (BLM)-induced dermal fibrosis mouse model. |
| • | CM-101 attenuated lung fibrosis and inflammation in the bleomycin (BLM)-induced pulmonary fibrosis mouse model. |
Results from the bleomycin (BLM)-induced dermal and lung fibrosis mouse models are discussed below in more detail.
CM-101 treatment attenuates skin fibrotic remodeling in the bleomycin (BLM)-induced dermal fibrosis mouse model
The activity of CM-101 (D8) in SSc was tested in the dermal bleomycin model. Treatment started after the onset of fibrotic signs, eight days following the first BLM injection. Histological assessment of skin lesions stained with H&E and Masson’s trichrome revealed significant elevation of dermal thickness and collagen deposition following 21 days of BLM administration. This elevation was significantly reduced when mice were treated with 2.5 mg/kg CM-101 with significant reductions in both skin thickness and collagen deposition compared with the mouse group treated with BLM alone.
CM-101 treatment attenuates skin fibrotic remodeling in the bleomycin-induced dermal fibrosis mouse model
Another feature that characterizes the BLM model and is representative of human SSc is the development of bronchoalveolar inflammation. To evaluate the effect of CM-101 on lung inflammation, the Company collected bronchoalveolar lavage, or, BAL, fluid, and assessed the number of white blood cells, or WBC, and mononuclear cells. Treatment with BLM for 21 days significantly increased WBC and mononuclear cells in BAL fluid and the number of WBC and mononuclear cells was decreased significantly following CM-101 treatment compared with the group that was administered only BLM. This data supports the anti-inflammatory effect of CM-101 in SSc.
CM-101 inhibits lung fibrosis in the BLM-induced pulmonary fibrosis mouse model
The Company also tested CM-101 in the experimental lung SSc model where mice were given a single intratracheal administration of BLM followed by either CM-101, non-active treatments (PBS or control immunoglobulin G (IgG)) or the approved anti-fibrosis drugs, pirfenidone and nintedanib. CM-101 had a significant anti-fibrotic and anti-inflammatory effect in the experimental BLM-induced lung fibrosis model as compared with non-active treatment-treated animals. BLM animals treated with non-active treatments showed massive immune cell infiltration, extensive fibrosis and severe tissue injury. CM-101-treated mice exhibited significantly reduced levels of lung fibrosis similar to levels in healthy animals and showed superior effects compared to the approved fibrosis drugs pirfenidone and nintedanib.
CM-101 attenuates lung fibrosis and collagen deposition in the bleomycin (BLM)-induced pulmonary fibrosis mouse model
Preclinical safety and toxicology of CM-101
Preclinical safety evaluation of CM-101 included tissue cross reactivity, assessment of the effect of CM-101 on pro-inflammatory cytokine secretion ex-vivo, and in vivo GLP toxicology studies in mice and non-human primates. No safety concerns were observed in these preclinical assessments.
Immunogenicity may be triggered following administration of humanized monoclonal antibodies, an effect that is frequently seen with approved mAbs. To date, no meaningful ADA were identified in three completed clinical studies, which supports a preliminary conclusion that CM-101 may have low immunogenic potential.
As summarized below, there were no safety concerns related to CM-101 in any of the other preclinical safety experiments.
Summary of key preclinical safety experiments
| Preclinical findings | | Observation |
| Ex vivo | |
|
| |
|
| Antibody dependent cell-cytotoxic (ADCC) and complement dependent cell-cytotoxic (CDC) activity was tested in PBMCs from healthy volunteers | | CM-101 did not have Fc-related effector functions such as ADCC and CDC |
| |
|
| Cytokine release was assessed in human whole blood from healthy volunteers. | | CM-101 did not induce pro-inflammatory cytokine secretion |
| | | |
| Tissue cross reactivity was evaluated from healthy human tissues. | | CM-101 does not bind non-specifically to healthy tissues, and therefore is expected to only bind to its target, circulating CCL24 |
| | |
| In vivo | | |
| | | |
GLP repeated dose 4-week toxicity study of CM-101 (IV) in mice | | 1. No obvious treatment related adverse reactions
2. No gross or microscopic pathological findings
3. No cases of treatment related mortality were observed
4. No significant elevation was seen in IL1β, IL2, IL4, IL5, IL10, GM-CSF, IFN and TNFα |
GLP repeated dose (up to 50 mg/kg) 6-month toxicity study of CM-101 (SC) in Cynomolgus Monkey | | 1. No obvious treatment related adverse reactions
2. No clinical signs or injection site reactions
3. No cases of treatment related mortality were observed
4. Blood and urine tests were found to be within normal ranges for monkeys
5. No treatment-related organ weight changes and no treatment-related necropsy findings
6. No treatment-related histopathology findings
7. Three samples from treated animals were confirmed ADA positive but there was no obvious correlation between positive ADA results and CM-101 serum concentrations or systemic exposure |
Preclinical proof of mechanism studies for CM-101
The Company conducted a series of in vitro and in vivo studies to demonstrate the proposed mechanism of action and provide proof-of-concept for administering CM-101 in the clinic for target indications.
Affinity, selectivity, and binding kinetics
The Company evaluated the kinetic binding parameters of CM-101 to human CCL24, as well as the specificity of CM-101 binding to other chemokines using commercial binding assays. CM-101 demonstrated a strong and stable, high affinity, binding to CCL24.
CM-101 reduced CCL24 dependent CCR3 activation
In an in vitro assay, CM-101 was shown to robustly attenuate the ability of CCL24 to induce activation of the CCR3 receptor following pre-incubation of CCL24 with CM-101.
Clinical Development of CM-101
Completed studies
The CM-101 Phase 1 program included two Phase 1a single administration, or SAD, studies, using IV and SC administration with doses ranging from 0.75-10 mg/kg, in healthy volunteers and a Phase 1b multiple administration (MAD) study (5 administrations) in NAFLD patients with normal liver function, testing 2.5 mg/kg IV and 5 mg/kg SC. In the Phase 1 studies 42 subjects have received at least one CM-101 dose, the majority by IV infusion (12/42 subjects received SC).
Safety
The first Phase 1a study, which was a single-center, randomized double-blind, placebo-controlled, single-dose, dose-escalation study, included four escalating dose groups of eight subjects each. In each dose group subjects were randomized in a 3:1 ratio to receive a single IV infusion of either CM-101 (n=6) or placebo (n=2). A total of 24 subjects were enrolled into the study and randomized to the treatment groups (0.75 mg/ kg, 2.5 mg/kg, 5.0 mg/kg, 10 mg/kg) and eight subjects received a placebo. All 32 subjects completed the study as planned. Single, IV doses of CM-101 were safe and well tolerated up to the highest dose level (10 mg/ kg) in healthy subjects. No severe or serious adverse events, or AEs, occurred during the study and all CM-101 related AEs were mild, with one moderate AE reported in the placebo group (myalgia).
The second Phase 1a study was also a single-center, randomized double-blind, placebo-controlled, single-dose study, but evaluated only one dose group. Subjects were randomized in a 3:1 ratio to receive a single SC injection of either CM-101 5 mg/kg (n=6) or matching placebo (n=2). A total of eight subjects were enrolled into the study and randomized; all eight subjects completed the study as planned. Single, SC administration of 5 mg/kg of CM-101 was safe and well tolerated with no severe or serious AEs occurring during the study. A total of 6 AEs were reported in two subjects treated with CM-101; only one AE was classified as related to CM-101 (change in diastolic blood pressure) and that AE was classified as mild in intensity.
In both Phase 1a studies, all AEs reported were resolved; no subjects discontinued the study prematurely due to AEs, and no concomitant medications were required for treatment of any drug-related AEs. No clinically significant changes in laboratory tests (hematology, chemistry or urinalysis), vital signs, ECG, physical examination or infusion site examination were observed. In the first Phase 1a study with CM-101 delivered by IV administration, the effect on cytokine secretion was tested pre-treatment and one hour, eight hours and 24 hours post drug administration. Serum levels of a panel of cytokines including IL-6, IFNγ, GM-CSF, TNF-α, IL-2, IL-4, IL-8 and IL-10 showed no significant change at all tested CM-101 doses and timepoints. These findings suggest that single CM-101 administration does not cause immune activation nor cytokine secretion. Additionally, none of the subjects in either of the Phase 1a studies tested positive for anti-drug antibodies (ADA).
The multiple administration randomized, placebo-controlled, Phase 1b study in NAFLD patients with normal liver function tests evaluated two dose levels. The first dose level of 2.5 mg/kg CM-101 was administered as an IV infusion and the second dose level of 5 mg/kg was administered as an SC injection. Both dose levels involved five drug administrations over 12 weeks (Q3W), providing 15 weeks of treatment coverage. At both dose levels, subjects were randomized in a 3:1 ratio to receive either CM-101 (n=6 per cohort) (2.5 mg/kg IV or 5 mg/kg SC) or matching placebo (n=2 per cohort). Five repeated IV and SC CM-101 administrations were safe and well tolerated and there were no deaths, or severe or serious drug related AEs reported throughout the study. Only mild to moderate AEs were reported in the CM-101 treatment groups of which only two AEs were classified as possibly related to CM-101. No injection site reactions or clinically significant trends in laboratory tests (hematology, chemistry, or urinalysis), vital signs, ECG or physical examination were observed. One patient experienced a non-drug-related SAE. This patient was a 61-year-old female that was subsequently diagnosed with a non-treatment related meningioma. The tumor was treated surgically, and the patient was discontinued from the study.
Pharmacokinetics with single-dose administration
PK analysis was conducted for the Phase 1 studies and the quantification of CM-101 in plasma samples was performed using a validated ELISA-based assay by Eurofins (UK). Following IV infusion in healthy volunteers, CM-101 exhibited a biphasic serum concentration vs. time curve (rapid distribution phase and slow elimination phase) which is typical for monoclonal antibodies. Target-mediated drug disposition (TMDD), or presence of ADAs, was not evident in the analyzed concentration vs. time curves of CM-101, which exhibited linear terminal slope without apparent TMDD kinetics or other concentration-dependent changes of the elimination kinetics. Comparison of the PK data of 5 mg/kg CM-101 using IV administration against SC administration indicates consistent distribution and elimination behavior of CM-101.
At either IV or SC administration, the values of the PK parameters obtained in the non-compartmental and compartmental analysis of CM-101 concentration vs. time data appear to be typical for monoclonal antibodies that undergo FcRn-mediated recycling. The terminal half-life of CM-101 was long for both SC and IV formulations, which supports administration of CM-101 at a frequency of once every 2-4 weeks.
Pharmacokinetics with multiple-dose administration
PK analysis of the data from the Phase 1b study was conducted to evaluate CM-101 following multiple IV infusion of 2.5 mg/kg or 5 mg/kg SC injections of CM-101 in NAFLD patients. Following repeated IV infusions (2.5 mg/kg Q3W) and SC injection (5 mg/kg Q3W), CM-101 exhibited a long terminal half-life, similar to the terminal half-life seen in the single dose studies. CM-101 accumulated over time, resulting in significant systemic exposure over time and potentially reaching a steady state.
Overall, CM-101 reached steady state conditions more slowly following SC injection, as compared to IV infusion. The inter-patient variability in CM-101 serum concentrations was higher for SC dosing injection, as compared to IV. The trough CM-101 serum concentrations after repeated 5 mg/kg SC injections were proportionally higher than those after 2.5 mg/kg IV infusions, considering the difference in administration modes. Comparison of the PK data of CM-101 in the Phase 1b to the Phase 1a studies indicates a consistency in PK behavior of CM-101.
Pharmacodynamics and target engagement of CM-101
Serum was taken from patients in all three Phase 1 studies at different times and the levels of both CCL24 and CM-101 were measured. Total CCL24 levels represent CM-101’s engagement to its target. Total CCL24 levels were increased following administration of the drug, which indicates that CM-101 is effective in target engagement, as the higher levels of CCL24 correlated significantly with greater doses of CM-101, and such levels decreased gradually from the peak of CM-101 administration. These findings demonstrate that CM-101 effectively binds to CCL24 in the circulation, which reflects a strong drug-target interaction.
In the Phase 1b study, CM-101 treatment of 2.5mg/kg IV attained the highest levels of total CCL24 by the third administration, maintaining these levels until the end of treatment. CM-101 5mg/kg administered by SC injection reached the highest levels of CCL24 by the fourth treatment and maintained these levels until the end of treatment. The matching placebo did not have any effect on CCL24 levels.
As exemplified in the in-vitro studies, binding of CCL24 by CM-101 attenuates the binding of CCL24 to its cognate CCR3 receptor, thereby reducing its downstream activation. Altogether, CCL24 levels following treatment with CM-101 provide strong evidence for target engagement and pharmacodynamic response of CM-101 in healthy volunteers and patients.
Phase 1b exploratory endpoints
Fibrotic biomarkers were analyzed as part of the Phase 1b study in NAFLD patients with normal liver function. Circulating fibrotic biomarkers were tested in serum pre- and post-treatment. The analysis included data from patients that presented with more active disease, reflected by baseline elastography (FibroScan) score >4 kPa. Tissue inhibitor of metalloproteinases-1 (TIMP-1) and tissue inhibitor of metalloproteinases-2 (TIMP-2), considered well established fibrotic biomarkers, were evaluated, and showed that CM-101 treatment led to reductions of both markers by week 15. The growth factor PDGF-AA, known as a pro-fibrotic secreted factor, was also reduced in CM-101 treated patients. Conversely, in the placebo group TIMP-1, TIMP-2 and PDGF-AA all increased.
Evaluation of the fibrogenesis and fibrolysis/inflammatory biomarkers, Pro-C3, Pro-C4 and C3M measured in serum, conducted by Nordic Bioscience, Copenhagen, Denmark, were also used as sensitive indicators of the liver’s fibrotic state. In accordance with reduced liver stiffness, Pro-C3, Pro-C4 and C3M were all reduced in the CM-101 treated groups. No reductions were identified in the placebo control group.
Changes in liver stiffness, a measurement of liver fibrosis, were also evaluated using FibroScan measurements taken at screening and end of treatment (EoT) following 15 weeks of treatment coverage. 80% of CM-101 treated patients had significant decreases in FibroScan measurements, unlike placebo patients where there was no significant change from baseline
Results of investigator-initiated clinical study of CM-101 in patients with COVID-19-derived lung damage
On November 9, 2022, encouraging clinical data from an investigator-initiated clinical study assessing CM-101 activity and safety in hospitalized patients with severe lung injury derived from COVID-19 infection was presented at the 2022 Union Conference, an international conference on lung health. A key rationale for the study is that some of the mechanisms underlying lung inflammation resulting from COVID-19 infection are similar to those seen in systemic sclerosis and other chronic diseases involving lung inflammation and fibrosis.
The objective was to evaluate the safety and activity of CM-101 in hospitalized COVID-19 patients with severe pneumonia, including its impact on biomarkers related to lung inflammation that are also relevant in SSc. The open label, single arm trial enrolled 16 hospitalized adult COVID-19 patients with severe respiratory involvement. All patients were receiving standard of care therapy. All were treated with a single 10mg/kg intravenous dose of CM-101 on the first day of the study and followed for 30 days.
Administration of CM-101 to this acutely ill patient population appeared safe and was well tolerated. CM-101 exposures and target engagement profiles were similar to what our researchers have seen in previous clinical studies of CM-101.
Importantly, rapid reductions in serum biomarkers of lung inflammation, fibrogenesis and neutrophil activity were observed post-treatment with CM-101. Overall, this study confirmed and extended the safety and tolerability profile of CM-101 and demonstrated clinically relevant changes in biomarkers associated with lung inflammation and fibrogenesis, further supporting CM-101’s anti-inflammatory and anti-fibrotic effects.
Moreover, we believe that these results add to the data suggesting that CM-101 has the potential to attenuate lung inflammation and fibrosis, further strengthening the rationale for treating SSc patients with this drug. These new clinical data also contribute to a growing body of evidence demonstrating CM-101’s anti-fibrotic and anti-inflammatory effects in varied organs including the lung, liver and skin.
Topline results of Phase 2a study in patients with liver fibrosis derived from NASH
Primary endpoints for the study were safety and tolerability. Secondary endpoints included the evaluation of the pharmacokinetic and target engagement profile of the SC formulation as well as changes in relevant biomarkers that may provide further mechanistic understanding of CM-101 effects on liver fibrosis. This trial was primarily designed to assess a subcutaneous formulation of CM-101 and to evaluate the drug’s impact on liver fibrosis biomarkers relevant to both NASH and fibro-inflammatory conditions that represent the focus for the company, such as PSC and SSc.
The randomized, placebo-controlled trial enrolled 23 NASH patients with stage F1c, F2 and F3 disease who were randomized to receive either CM-101 or placebo. Patients received eight doses of 5 mg/kg of study drug administered by subcutaneous injection once every two weeks, for a treatment period of 16 weeks. Key findings of the CM-101 Phase 2a trial included the following.
• CM-101 appeared to be safe and was well tolerated when administered subcutaneously. Most reported adverse events observed were mild, with one unrelated serious adverse event reported. No significant injection site reactions were reported and no anti-drug antibodies were detected.
• CM-101 administered subcutaneously demonstrated favorable pharmacokinetics and target engagement profiles as expected and were similar to what the company has previously reported.
• CM-101-treated patients showed greater improvements than the placebo group in a number of liver fibrosis-related biomarkers, including ProC-3, ProC-4, ProC-18, TIMP-1 and ELF.
• A majority of CM-101-treated patients showed improvements in more than one liver fibrosis-related biomarker—almost 60% of CM-101 patients responded in at least three biomarkers at week 20, compared to no patients in the placebo group.
• A higher proportion of patients in the CM-101-treated group showed improvement in a physiologic measure of liver stiffness as compared to placebo (reduction of at least one grade of fibrosis score as assessed by the non-invasive elastography method known as FibroScan®).
• CM-101-treated patients with higher CCL24 levels at baseline showed greater reductions in fibrosis-related biomarkers than patients with lower levels. Multiple fibrosis-related biomarkers showed more pronounced reductions in CM-101-treated patients who had higher CCL24 levels at baseline than in patients with lower CCL24 levels at baseline, adding to the growing body of evidence validating the role of CCL24 in the pathophysiology of fibrotic liver disease.
• After completion of the study, the unblinded data showed that patients in the CM-101-treated group had higher baseline levels of fibrosis compared to placebo patients. The impact of this difference on the results, if any, is unknown.
We believe that the data from this trial provide important insights in support of the CM-101 development program, including the favorable safety and tolerability of CM-101 in patients with serious liver disease, confirmation of early signs of biomarker activity that are relevant for a number of fibro-inflammatory disorders, and support of the tolerability and pharmacokinetic data needed to assess next steps in the development of our current subcutaneous formulation.
Other clinical development activities for CM-101
Recently, Chemomab also reported positive clinical data from an investigator-initiated clinical study assessing CM-101 activity and safety in hospitalized patients with severe lung injury derived from COVID-19. The objective of the study was to evaluate the drug’s safety and activity in hospitalized COVID-19 patients with severe pneumonia, including its impact on biomarkers related to lung inflammation that are also relevant in systemic sclerosis. The open label, single arm trial enrolled 16 hospitalized adult COVID-19 patients with severe respiratory involvement. All patients were receiving standard of care therapy. All were treated with a single 10mg/kg intravenous dose of CM-101 on the first day of the study and followed for 30 days. Administration of CM-101 to this acutely ill patient population appeared to be safe and was well tolerated. CM-101 exposures and target engagement profiles were similar to what our researchers have seen in previous clinical studies of CM-101. Importantly, rapid reductions in serum biomarkers of lung inflammation, fibrogenesis and neutrophil activity were observed post-treatment with CM-101. Overall, this study confirmed and extended the safety and tolerability profile of CM-101 and demonstrated clinically relevant changes in biomarkers associated with lung inflammation and fibrogenesis, further supporting CM-101’s anti-inflammatory and anti-fibrotic effects.
Together, these promising results provide initial support for CM-101’s anti-fibrotic and anti-inflammatory mechanisms in humans and support further testing of CM-101 in PSC and SSc patients.
Current and planned clinical studies for PSC and SSc
The Company is currently recruiting and treating PSC patients in a Phase 2 study at multiple sites in Israel, the United States and Europe and expanding the trial to include an additional dose level and an open label extension. The Company is also planning to initiate a Phase 2 study in SSc around midyear of 2023 that will enroll patients across multiple sites in Israel, the United States and Europe.
The ongoing Phase 2 trial in PSC is a randomized, double-blind, placebo-controlled, study designed to evaluate the safety and efficacy of CM-101 in adult subjects with PSC. Participants must have a serum alkaline phosphatase, or ALP, level of at least 1.5 times the upper limit of normal (x 1.5 ULN). Subjects with concomitant IBD are eligible for recruitment if their disease is stable and there is an absence of high-grade dysplasia in colonic biopsies within 18 months of randomization. To date, subjects are randomized to receive 10 mg/kg CM-101 IV, or placebo, in a 2:1 ratio. Chemomab has amended the study to include a higher dose level (20mg/kg) of CM-101. Patients will receive a dose of investigational product once every three weeks for a total of five administrations resulting in a total coverage of 15 weeks during the double-blind portion of the study. Chemomab is adding a 33-week long open-label extension to the study.
The primary endpoint for the study is safety and tolerability. Secondary endpoints include evaluations of changes from baseline in serum alkaline phosphatase, or ALP, levels and the fibrotic marker enhanced liver function, or ELF, score at week 15. ALP is a liver enzyme that is elevated in cholestasis and the ELF score is a biochemical test panel made up of serum markers that are indicators of the extracellular matrix. Additional secondary endpoints include evaluations of changes from baseline in other liver enzymes and additional fibrotic markers, to include AST, ALT, Pro-C3 and Pro-C5. PK, PD and ADA parameters.
The planned Phase 2 study in SSc will enable an expedited path to proof-of-concept data and further elucidation of different CM-101 mechanisms of actions in treating SSc skin, lung and vascular damage. To that end, we will slightly enrich the study with SSc patients who have higher levels of CCL24 and may therefore be more likely to respond to neutralization of this critical chemokine Chemomab is currently finalizing the design. The U.S. FDA recently cleared the Company’s IND application to commence the Phase 2 SSc trial and we plan to initiate the study around midyear of 2023. Key design elements include the following.
| | • | The trial is a randomized, double-blind, placebo-controlled study that will enroll 45 SSc patients. |
| | • | To be eligible for the study, patients must manifest two key characteristics: the presence of clinically active disease, either dermatologic or pulmonary, and higher serum levels of circulating CCL24. |
| | • | Thirty patients will be randomized to treatment with CM-101 and 15 will be randomized to placebo. |
| | • | Of the patients on active treatment, approximately half will have limited SSc, and half will have diffuse cutaneous disease. |
| | • | The study includes a 24-week double blind period during which patients assigned to active treatment will receive CM-101 at a dose of 10 mg/kg, via intravenous infusion, every three weeks. |
| | • | Following the double-blind period, patients will enter a 24-week open label treatment period, where all patients will receive CM-101 at a dose of 10 mg/kg via intravenous infusion every three weeks. |
| | • | All patients enrolled will undergo a skin biopsy at baseline and again after the double-blind treatment period, along with multiple clinical assessments of skin, vascular and pulmonary function. |
| | • | The primary outcome measure for the trial will be demonstration of the safety and tolerability of treatment with CM-101. |
| | • | All other outcome measures will be principally assessed as changes from baseline to the end of the double-blind treatment period. |
The secondary outcome measures of the trial are focused on highly relevant and informative biological read-outs. Key secondary outcomes include: evaluation of multiple serum-based biological markers that are known to be associated with different manifestations of SSc including:
| • | Inflammatory cytokines (such a CCL2, IL6 and CXCL10), vascular and growth factor-related biomarkers (such as VEGF and PDGF), pulmonary-related biomarkers (such as KL-6, SPD and CCL18), and fibrogenesis and extracellular matrix biomarkers (collagens, MMPs and ELF score). |
| • | Inflammatory, fibrotic and target expression markers in skin biopsies, including but not limited to CCL24 and CCR3 expression levels. |
| • | Pharmacokinetics and target engagement of CM-101. |
| • | Monitoring for the presence of any potential anti-drug antibodies during the study. |
Exploratory biological outcomes assessments will include immune cell phenotyping, assessments of neutrophil function, and ex-vivo biological assays. Exploratory clinical outcomes will include evaluation of: vascular involvement, using nail fold capillaroscopy; vascular imaging and digital ulcer burden; skin involvement using modified Rodnan scoring; pulmonary disease involvement using pulmonary function tests; and multiple patient-reported outcome measures. The data collected should also enable evaluation of global effects of intervention with CM-101 using the American College of Rheumatology CRISS scale (ACR-CRISS) and the revised CRISS scale (rCRISS). We intend to conduct this study at multiple sites in Israel, the United States and Europe.
Competition
The development and commercialization of new drug products is highly competitive across major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. The Company faces competition with respect to its current product and expects to face competition with respect to any product candidates that it may develop or commercialize in the future. Specifically, there are a number of companies developing treatments for fibrotic/inflammatory diseases, including multiple major pharmaceutical and biotechnology companies with substantially greater resources than the Company. The Company is a small biotech company with limited resources compared to the major pharmaceutical companies, however, the Company believes that the unique CM-101 platform together with its knowledge and experience in inflammatory-fibrotic research provides it with competitive advantages.
Therapeutic options for PSC and SSc are limited and despite significant biopharmaceutical industry investment, the FDA has not approved any disease modifying therapies for the treatment of PSC or SSc. Liver transplant is currently the only treatment shown to improve clinical outcomes for PSC patients while SSc patients are being treated with drugs that were approved for different manifestations of the disease like interstitial lung disease (nintedanib, Boehringer Ingelheim and tocilizumab, Hoffmann-La Roche).
The Company is advancing CM-101, a first-in-class monoclonal antibody that interferes directly with both inflammation and fibrosis, into clinical development for the treatment of PSC and SSc. There are a number of large biopharmaceutical and biotechnology companies that are currently pursuing the development of products for the treatment of fibrotic indications like PSC and SSc, such as, Mitsubishi Tanabe Pharma, Horizon Therapeutics, Pliant Therapeutics, Prometheus Biosciences and others. However, the Company knows of no other companies currently in clinical development with a monoclonal antibody that targets CCL24.
Although the approach is novel with respect to targeting both inflammation and fibrosis, the Company will need to compete with products further advanced in the pipeline towards market approval. Investigational products, include:
There are currently no FDA-approved therapies for the treatment of PSC. Companies currently developing product candidates in Phase 3 clinical studies include Gilead and Dr. Falk Pharma, targeting cholestasis and liver metabolism (Gilead; Cilofexor, Dr. Falk; norUrso). Additional companies with clinical candidates in earlier stages of development include HighTide Biopharmaceutical, Mirum Pharmaceuticals and Pliant Inc.
There are currently two FDA approved products for the treatment of clinical manifestations of SSc--nintedanib, marketed by Boehringer Ingelheim GmbH and tocilizumab, marketed by Hoffmann-La Roche for the treatment of interstitial lung disease. Companies currently developing product candidates in SSc in early clinical stage include Horizon, Mitsubishi Tanabe, GS Johnson & Johnson, Vicore, Sanofi, Prometheos Biosciences and others.
The availability of reimbursement from government and other third-party payors will affect the pricing and competitiveness of CM-101 and any future products. More advanced competitors also may obtain regulatory approval for their products more rapidly than the Company, which could result in competitors establishing a strong market position.
Intellectual Property
Overview
The Company strives to protect and enhance the proprietary technology, inventions, and improvements that are commercially important to the development of its business, including seeking, maintaining, and defending patent rights, whether developed internally or licensed from third parties. The Company also relies on trade secrets relating to its proprietary technology platform and know-how, continuing technological innovation and in-licensing opportunities to develop, strengthen, and maintain its proprietary position in the field of inflammation and fibrosis that may present areas of opportunity for the development of its business. The Company may also rely on regulatory protection afforded through data exclusivity, market exclusivity, and patent term extensions, where available.
The Company’s commercial success may depend in part on its ability to: obtain and maintain patent and other proprietary protection for commercially important technology, inventions and know-how related to its business; defend and enforce its patents; preserve the confidentiality of its trade secrets; and operate without infringing the valid enforceable patents and proprietary rights of third parties. The Company’s ability to prevent third parties from making, using, selling, offering to sell, or importing the Company’s products may depend on the extent to which it has rights under valid and enforceable licenses, patents, or trade secrets that cover these activities. In certain cases, enforcement of these rights may depend on third party licensors. With respect to both licensed and company-owned intellectual property rights, the Company cannot be sure that patents will be granted with respect to any of its pending patent applications or with respect to any patent applications that may be filed by the Company in the future, nor can the Company be sure that any of its existing patents or any patents that may be granted to it in the future will be commercially useful in protecting its commercial products and methods of manufacturing the same.
As of the date of this Annual Report on Form 10-K, the Company owned or licensed seven pending or issued US patents and patent applications as well as patents and patent applications in other jurisdictions. The first patent family has been issued in each of the United States, Europe (validated in France, Germany and the United Kingdom) and Israel to the Tel Aviv Souraski Medical Center, whose rights have been licensed to the Company on an exclusive basis. A composition of matter patent was issued in United States and certain corresponding foreign jurisdictions. To date, three additional patent families were filed by the Company concerning the use of anti CCL24 antibodies in specific indications, dosing regimens, and routes of administration. The Company will seek United States and foreign patent protection for a variety of additional technologies, including: research compounds and methods, candidate compounds and antibodies for modulating the activity of CCL24, methods for treating diseases of interest, and methods for treating its products. The Company will seek additional protection, in part, through confidentiality and proprietary information agreements.
Company Owned Intellectual Property
The Company owns multiple families of patent applications that pertain to anti-CCL24 monoclonal antibody compositions capable of blocking CCL24 activity and methods for treating or preventing diseases associated with inflammation and fibrosis. Certain applications in these families relate to the Company’s CM-101 antibody, backup variants, various unit dosages, dosing regimens, and other routes of administration. Patents that are or will be issued from these submissions will expire between the years 2035 to 2041, subject to possible patent term adjustments and/or extensions.
In addition to the above, the Company has established expertise and development capabilities focused in the areas of preclinical research and development, manufacturing and manufacturing process development, quality control, quality assurance, regulatory affairs, and clinical study design and implementation. The Company believes that its focus and expertise will help the Company develop products based on its proprietary intellectual property.
Licensed IP
As mentioned above, the Company has obtained an exclusive license from the Tel Aviv Souraski Medical Center for one patent, which is expected to expire in 2029. This patent was issued in each of the United States, Europe and Israel, and pertains to anti CCL24 inhibitors (specifically, anti CCL24 antibodies) and methods of using such inhibitors for treating inflammatory, autoimmune and cardiovascular diseases.
Trade Secret Protection
The Company may rely, in some circumstances, on trade secrets to protect its technology. The Company seeks to protect its proprietary technology and processes, in part, by entering into confidentiality agreements with its employees, consultants, scientific advisors, and contractors. The Company also seeks to preserve the integrity and confidentiality of its data and trade secrets by maintaining physical security of its premises and physical and electronic security of its information technology systems.
Material Agreements
Tel-Aviv Souraski Medical Center (TASMC) License Agreement
In December 2011, the Company entered into a license agreement, or the TASMC Agreement, with the Medical Research, Infrastructure, Health Services Fund of the Tel Aviv Souraski Medical Center., or TASMC, for the research, development and commercialization of the CCL24 platform and CCR3 blockade platform (CM-101), which license includes patent rights covering the foregoing platforms and related know how and products. Under the terms of the TASMC Agreement, the Company is responsible for the research, development, manufacturing and commercialization of CM-101. This license was granted on an exclusive basis and the Company was also granted rights to sublicense the instant license to third parties pursuant to certain terms described therein.
In accordance with the TASMC Agreement, the Company paid TASMC a non-refundable and non-creditable payment in four milestone installments, related to TASMC’s past patent maintenance and prosecution costs.
Certain additional terms of the TASMC Agreement include:
| • | The Company will be required to pay TASMC non-refundable and non-creditable milestone payments of up to (i) $300,000 upon the submission of an NDA, BLA or equivalent for each of the licensed products to the FDA and to equivalent European and Asian foreign regulatory agencies, and (ii) $600,000 upon the grant by the FDA or equivalent European and/or Asian regulatory agencies of their marketing approval for each licensed product; |
| • | In the event of an “exit,” as such term is defined therein, the Company must pay TASMC an exit fee of 1% of the transaction consideration (which shall be capped at $3 million); |
| • | In the event the Company sublicenses a licensed product, the Company must pay TASMC a sublicense fee of 10% of all attributed income, in addition to a low-single digit percentage tiered royalty payment of our earned royalties. |
Unless terminated earlier, the TASMC Agreement will expire upon the later of the expiration of the last-to-expire valid patent claim and any extension granted prior thereto. The termination of the TASMC Agreement will not preclude TASMC from receiving sublicense payments or royalties. In addition to the foregoing, the TASMC Agreement includes customary termination provisions.
CMC Collaboration Agreement
In June 2015, the Company entered into a collaboration agreement, or the CMC Agreement, with CMC ICOS Biologics, Inc. (acquired by AGC Biologics in 2018), or CMC, which, under the terms thereof, granted the Company certain licenses to use proprietary rights, materials and know-how of CMC for purposes of research and development of CM-101 as well as commercialization thereof. Pursuant to the terms of the CMC Agreement, the Company received (i) a worldwide, non-exclusive, non-transferable, non-sublicensable license for research purposes, or the Research License, and (ii) an option, or the Option License, to a worldwide, non-exclusive, non-transferable, sublicensable license for commercialization purposes, subject to a fee schedule in addition to that described below.
In accordance with the terms of the CMC Agreement, the Company agreed to pay in exchange for the foregoing license payments to CMC upon the achievement of certain pre-determined clinical and regulatory events, an amount stipulated in the CMC Agreement, aggregating a six-digit number. Additionally, for any product that is commercialized pursuant to the CMC Agreement, the Company is required to pay CMC a royalty payment based on annual aggregate worldwide net sales thresholds for such products. In the event CMC exclusively manufactures the Company’s products, CMC agrees to waive the foregoing royalty.
Unless terminated earlier pursuant to the customary termination provisions set forth in the CMC Agreement, the Research License will expire upon the conclusion of the term as defined therein, and the Option License will expire upon the later of (a) the tenth anniversary following the Company’s obtainment of regulatory approval, or (b) the last to expire of the patent rights and country-by-country basis.
Manufacturing
The Company’s product candidate, CM-101, is a monoclonal antibody amenable to standard formulation technologies. The Company has developed the biological process and manufactured kilogram quantities through processes similar to the manufacturing processes that will be required to provide drug product for the Phase 2 clinical studies. The manufacturing process of the drug substance used for such product candidates is robust, well established and requires the use of readily available starting materials. The biological route is amenable to large-scale production and does not require unconventional equipment or handling during the manufacturing process. The Company has obtained an adequate supply chain of the drug substance for CM-101 from the Company’s contract manufacturing organization, or CMO, to satisfy both the Company’s clinical and preclinical requirements for this year. The Company relies on a sole supplier for the manufacture of CM-101. The Company’s manufacturer has the capabilities to support late stage clinical studies as well as product launch and marketing.
The Company does not own or operate facilities for clinical drug manufacturing, storage, distribution or quality testing. Currently, all of the Company’s clinical manufacturing is outsourced to third-party manufacturers. As the Company’s development programs expand and it builds new process efficiencies, the Company expects to continually evaluate this strategy with the objective of satisfying demand for its clinical studies and, if approved, the manufacture, sale and distribution of commercial products.
Commercialization
The Company intends to develop and, if approved by the FDA, to commercialize its product candidates alone or in collaboration with others. The Company may work in combination with one or more large pharmaceutical partners for certain indications, where specialist capabilities are needed. The Company intends to enter into distribution or licensing arrangements for global or regional commercialization rights. The Company will, however, continuously review its partnering strategy in the light of new clinical data and market understanding.
Regulatory Matters
The Food and Drug Administration, or FDA, and comparable regulatory authorities in state and local jurisdictions and in other countries impose substantial and burdensome requirements upon companies involved in the clinical development, manufacture, marketing and distribution of drugs, such as those the Company is developing. These agencies and other federal, state and local entities regulate, among other things, the research and development, testing, manufacture, quality control, safety, effectiveness, labelling, storage, record keeping, approval, advertising and promotion, distribution, post-approval monitoring and reporting, sampling and export and import of the Company’s product candidates.
United States government regulation of drug products
Drugs in the United States are subject to rigorous regulation under the Food, Drug, and Cosmetic Act, or FDCA, and its implementing regulations. The FDA also regulates biological products under the FDCA and the Public Health Service Act, or PHSA. If the Company advances clinical development of a biologic candidate in the future, these development activities will be subject to additional regulatory requirements specific to biologics. The process of obtaining regulatory approvals and the subsequent compliance with applicable federal, state, local and foreign statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable United States requirements at any time during the product development process, approval process or after approval, may subject an applicant to a variety of administrative or judicial sanctions, such as the FDA’s refusal to approve a pending New Drug Application, or NDA, withdrawal of an approval, imposition of a clinical hold, issuance of warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement or civil or criminal penalties.
The process required by the FDA before a drug or biologic may be marketed in the United States generally involves the following:
| | • | Completion of preclinical laboratory tests, animal studies and formulation studies in compliance with the FDA’s Good Laboratory Practice, or GLP, regulations; |
| | • | Submission to the FDA of an Investigational New Drug application, or IND, which must become effective before human clinical studies may begin; |
| | • | Approval by an Institutional Review Board, or IRB, at each clinical site before each study may be initiated; |
| | • | Performance of adequate and well-controlled human clinical studies in accordance with Good Clinical Practice, or GCP requirements to establish the safety and efficacy of the proposed drug product for each indication; |
| | • | Completion of all manufacturing requirements to ensure robust manufacturing process, and product quality and safety as per Good Manufacturing Practice, or cGMP guidelines; |
| | • | Completion of non-clinical reproductive studies, as applicable, prior to late stage clinical studies and NDA or Biologics License Application, or BLA, submission; |
| | • | Development of an appropriate pediatric plan for clinical testing or exclusion, pre- or post-approval, as applicable; |
| | • | Submission to the FDA of an NDA or BLA; |
| | • | Satisfactory completion of an FDA advisory committee review, if applicable; |
| | • | Satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the product is produced to assess compliance with cGMP requirements and to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality and purity; |
| | • | Satisfactory completion of FDA audits of clinical study sites to assure compliance with GCPs and the integrity of the clinical data; |
| | • | Payment of user fees and securing FDA approval of the NDA; |
| | • | FDA review and approval of an NDA or BLA; and |
| | • | Compliance with any post-approval requirements, including the potential requirement to implement a Risk Evaluation and Mitigation Strategies, or REMS, and the potential requirement to conduct post-approval studies. |
Preclinical studies
Preclinical studies include laboratory evaluation of product chemistry, toxicity and formulation, as well as animal studies to assess potential safety and efficacy. An IND sponsor must submit the results of the preclinical tests, together with manufacturing information, analytical data and any available clinical data or literature, among other things, to the FDA as part of an IND. Some preclinical testing may continue even after the IND is submitted. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions related to one or more proposed clinical studies and places the clinical study on a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical study can begin. As a result, submission of an IND may not result in the FDA allowing clinical studies to initiate.
Clinical studies
Clinical studies involve the administration of the investigational new drug to human subjects under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research subjects provide their informed consent in writing for their participation in any clinical study. Clinical studies are conducted under protocols detailing, among other things, the objectives of the study, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. A protocol for each clinical study and any subsequent protocol amendments must be submitted to the FDA as part of the IND. In addition, an IRB at each institution participating in the clinical study must review and approve the plan for any clinical study before it initiates at that institution. Information about certain clinical studies must be submitted within specific timeframes to the National Institutes of Health, or NIH, for public dissemination on their www.clinicaltrials.gov website.
Human clinical studies are typically conducted in three sequential phases, which may overlap or be combined. A fourth, or post-approval, phase may include additional clinical studies. These phases generally include the following:
| | • | Phase 1: The drug or biologic is initially introduced into healthy human subjects or patients with the target disease or condition and tested for safety, dosage tolerance, absorption, metabolism, distribution, excretion and, if possible, to gain an early indication of its effectiveness. For some products for severe or life-threatening diseases, especially if the product may be too toxic to administer to healthy humans, the initial clinical trials may be conducted in individuals having a specific disease for which use the tested product is indicated. |
| | • | Phase 2: The drug or biologic is administered is administered to a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance and optimal dosage. |
| | • | Phase 3: The drug or biologic is administered to an expanded patient population, generally at geographically dispersed clinical study sites, in well-controlled clinical studies to generate enough data to statistically evaluate the efficacy and safety of the product for approval, to establish the overall risk-benefit profile of the product, and to provide adequate information for the labeling of the product. |
| | • | Phase 4: Phase 4 clinical trials are studies required of, or agreed to by, a sponsor that are conducted after the FDA has approved a product for marketing. These studies are used to gain additional experience from the treatment of patients in the intended therapeutic indication and to document a clinical benefit in the case of drugs approved under accelerated approval regulations. If the FDA approves a product while a company has ongoing clinical trials that were not necessary for approval, a company may be able to use the data from these clinical trials to meet all or part of any Phase 4 clinical trial requirement. Failure to promptly conduct Phase 4 clinical trials where necessary could result in withdrawal of approval for products approved under accelerated approval regulations. |
Progress reports detailing the results of the clinical studies must be submitted at least annually to the FDA and more frequently if serious adverse events occur. Phase 1, Phase 2 and Phase 3 studies may not be completed successfully within any specified period, or at all. Furthermore, the FDA or the sponsor may suspend or terminate a clinical study at any time on various grounds, including a finding that the research subjects are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical study at its institution if the clinical study is not being conducted in accordance with the IRB’s requirements or if the drug has been associated with unexpected serious harm to patients.
Marketing approval
Assuming successful completion of the required clinical testing, the results of the preclinical and clinical studies, together with detailed information relating to the product’s chemistry, manufacture, controls and proposed labeling, among other things, are submitted to the FDA as part of an NDA or BLA requesting approval to market the product for one or more indications. In most cases, the submission of an NDA or BLA is subject to a substantial application user fee. Under the Prescription Drug User Fee Act, or PDUFA, guidelines that are currently in effect, the FDA has a goal of ten months from the date of “filing” of a standard NDA, for a new molecular entity to review and act on the submission. This review typically takes twelve months from the date the NDA or BLA is submitted to FDA because the FDA has approximately two months to make a “filing” decision.
In addition, under the Pediatric Research Equity Act of 2003, or PREA, as amended and reauthorized, certain NDAs/BLAs or supplements thereof must contain data that are adequate to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements. An Agreed Initial Pediatric Study Plan requesting a waiver from the requirement to conduct clinical studies may be submitted to the FDA.
The FDA also may require submission of a REMS plan to ensure that the benefits of the drug outweigh its risks. The REMS plan could include medication guides, physician communication plans, assessment plans, and/or elements to assure safe use, such as restricted distribution methods, patient registries, or other risk minimization tools.
The FDA conducts a preliminary review of all NDAs/BLAs within the first 60 days after submission, before accepting them for filing, to determine whether they are sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an NDA/BLA for filing. In this event, the application must be resubmitted with the additional information. The resubmitted application is also subject to review before the FDA accepts it for filing. Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews an NDA/BLA to determine, among other things, whether the drug is safe and effective and whether the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product’s continued safety, quality and purity.
The FDA may refer an application for a novel drug to an advisory committee. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, which reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
Before approving an NDA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving an NDA, the FDA may inspect one or more clinical study sites to assure compliance with GCP requirements.
After evaluating the NDA/BLA and all related information, including the advisory committee recommendation, if any, and inspection reports regarding the manufacturing facilities and clinical study sites, the FDA may issue an approval letter, or, in some cases, a complete response letter. A complete response letter generally contains a statement of specific conditions that must be met in order to secure final approval of the NDA/BLA and may require additional clinical or preclinical testing in order for the FDA to reconsider the application. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. If and when those conditions have been met to the FDA’s satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Even if the FDA approves a product, it may limit the approved indications for use of the product, require that contraindications, warnings or precautions be included in the product labeling, require that post-approval studies, including Phase 4 clinical studies, be conducted to further assess a drug’s safety after approval, require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution and use restrictions or other risk management mechanisms under a REMS, which can materially affect the potential market and profitability of the product. The FDA may prevent or limit further marketing of a product based on the results of post-marketing studies or surveillance programs. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval.
FDA Expedited Development and Review Programs
The FDA has various programs, including fast track designation, priority review, accelerated approval, and breakthrough therapy designation, which are intended to expedite or simplify the process for the development and FDA review of drugs that are intended for the treatment of serious or life-threatening diseases or conditions and demonstrate the potential to address unmet medical needs. The purpose of these programs is to provide important new drugs to patients earlier than under standard FDA review procedures.
The FDA has a fast track designation program that is intended to expedite or facilitate the process for reviewing new drug products that meet certain criteria. Specifically, new drugs are eligible for fast track designation if they are intended to treat a serious or life-threatening disease or condition and demonstrate the potential to address unmet medical needs for the disease or condition. With regard to a fast track product, the FDA may consider for review sections of the NDA/BLA on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the NDA/BLA, the FDA agrees to accept sections of the NDA/BLA and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the NDA/BLA.
Any product submitted to the FDA for approval, including a product with a fast track designation, may also be eligible for other types of FDA programs intended to expedite development and review, such as priority review and accelerated approval. A product is eligible for priority review if it has the potential to provide safe and effective therapy where no satisfactory alternative therapy exists or a significant improvement in the treatment, diagnosis, or prevention of a disease compared to marketed products. The FDA will attempt to direct additional resources to the evaluation of an application for a new drug designated for priority review in an effort to facilitate the review.
In addition, a product may be eligible for accelerated approval. Drug products intended to treat serious or life-threatening diseases or conditions may be eligible for accelerated approval upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA may require a sponsor of a drug receiving accelerated approval to perform post-marketing studies to verify and describe the predicted effect on irreversible morbidity or mortality, or other clinical endpoint and to submit promotional materials for preapproval and pre-use review, which could adversely impact the timing of the commercial launch of the product. In addition, the drug may be subject to accelerated withdrawal procedures. On December 29, 2022, the Consolidated Appropriations Act, 2023, including the Food and Drug Omnibus Reform Act (FDORA), was signed into law. FDORA made several changes to the FDA’s authorities and its regulatory framework, including, among other changes, reforms to the accelerated approval pathway, such as requiring the FDA to specify conditions for post-approval study requirements and setting forth procedures for the FDA to withdraw a product on an expedited basis for non-compliance with post-approval requirements.
The Food and Drug Administration Safety and Innovation Act established a category of drugs referred to as “breakthrough therapies” that may be eligible to receive breakthrough therapy designation. A sponsor may seek FDA designation of a product candidate as a “breakthrough therapy” if the product is intended, alone or in combination with one or more other products, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The designation includes all of the fast track program features, as well as more intensive FDA interaction and guidance. The breakthrough therapy designation is a distinct status from both accelerated approval and priority review, which can also be granted to the same drug if relevant criteria are met. If a product is designated as breakthrough therapy, the FDA will work to expedite the development and review of such drug.
In addition, the FDA may review new drug applications under the Oncology Center of Excellence Real-Time Oncology Review (“RTOR”), which, according to the FDA, aims to explore a more efficient review process to ensure that safe and effective treatments are available to patients as early as possible, while maintaining and improving review quality. Drugs considered for review under RTOR must be likely to demonstrate substantial improvements over available therapy, which may include drugs previously granted breakthrough therapy designation for the same or other indications, and must have straight-forward study designs and endpoints that can be easily interpreted. RTOR allows the FDA to review much of the data in an NDA/BLA earlier, before the applicant formally submits the complete application. This analysis of the pre-submission package gives the FDA and applicants an early opportunity to address data quality and potential review issues and allows the FDA to provide early feedback regarding the most effective way to analyze data to properly address key regulatory questions.
Fast track designation, priority review, accelerated approval, and breakthrough therapy designation do not change the standards for approval, but may expedite the development or approval process. Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.
Orphan drug designation and exclusivity
Under the Orphan Drug Act, the FDA may designate a drug product as an “orphan drug” if it is intended to treat a rare disease or condition (generally meaning that it affects fewer than 200,000 individuals in the United States, or more in cases in which there is no reasonable expectation that the cost of developing and making a drug product available in the United States for treatment of the disease or condition will be recovered from sales of the product). A company must request orphan product designation before submitting an NDA. If the request is granted, the FDA will disclose the identity of the therapeutic agent and its potential use. Orphan product designation does not convey any advantage in or shorten the duration of the regulatory review and approval process. As of the current date, the Company has obtained orphan drug designation for three indications, PSC, SSc and IPF.
If a product with orphan status receives the first FDA approval for the disease or condition for which it has such designation or for a select indication or use within the rare disease or condition for which it was designated, the product generally will be receiving orphan product exclusivity. Orphan product exclusivity means that the FDA may not approve any other applications for the same product for the same indication for seven years, except in certain limited circumstances. If a drug or drug product designated as an orphan product ultimately receives marketing approval for an indication broader than what was designated in its orphan product application, it may not be entitled to exclusivity. Orphan exclusivity will not bar approval of another product under certain circumstances, including if a subsequent product with the same active ingredient for the same indication is shown to be clinically superior to the approved product on the basis of greater efficacy or safety, or providing a major contribution to patient care, or if the company with orphan drug exclusivity is not able to meet market demand. Further, the FDA may approve more than one product for the same orphan indication or disease as long as the products contain different active ingredients. Moreover, competitors may receive approval of different products for the indication for which the orphan product has exclusivity or obtain approval for the same product but for a different indication for which the orphan product has exclusivity.
In Catalyst Pharms., Inc. v. Becerra, 14 F.4th 1299 (11th Cir. 2021), the court disagreed with the FDA’s longstanding position that the orphan drug exclusivity only applies to the approved use or indication within an eligible disease. This decision created uncertainty in the application of the orphan drug exclusivity. On January 24, 2023, the FDA published a notice in the Federal Register to clarify that while the agency complies with the court’s order in Catalyst, FDA intends to continue to apply its longstanding interpretation of the regulations to matters outside of the scope of the Catalyst order – that is, the agency will continue tying the scope of orphan-drug exclusivity to the uses or indications for which a drug is approved, which permits other sponsors to obtain approval of a drug for new uses or indications within the same orphan designated disease or condition that have not yet been approved. It is unclear how future litigation, legislation, agency decisions, and administrative actions will impact the scope of the orphan drug exclusivity.
United States marketing exclusivity
Market exclusivity provisions under the FDCA also can delay the submission or the approval of certain applications. The FDCA provides a five-year period of non-patent marketing exclusivity within the United States to the first applicant to gain approval of an NDA for a new chemical entity. A drug is a new chemical entity if the FDA has not previously approved any other new drug containing the same active moiety, which is the molecule or ion responsible for the action of the drug substance. During the exclusivity period, the FDA may not accept for review an Abbreviated New Drug Application, or ANDA, or a 505(b)(2) NDA submitted by another company for another version of such drug where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted after four years if it contains a certification of patent invalidity or non-infringement. The FDCA also provides three years of marketing exclusivity for an NDA, 505(b)(2) NDA or supplement to an existing NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example, new indications, dosages or strengths of an existing drug. This three-year exclusivity covers only the conditions of use associated with the new clinical investigations and does not prohibit the FDA from approving ANDAs for the original non-modified version of the drug. Five-year and three-year exclusivity will not delay the submission or approval of a full NDA. However, an applicant submitting a full NDA would be required to conduct or obtain a right of reference to all of the preclinical studies and adequate and well-controlled clinical studies necessary to demonstrate safety and effectiveness.
Abbreviated Licensure Pathway of Biological Products as Biosimilars or Interchangeable Biosimilars
The Patient Protection and Affordable Care Act (Affordable Care Act or ACA), signed into law in 2010, includes the Biologics Price Competition and Innovation Act of 2009 (BPCIA), which created an abbreviated approval pathway for biological products shown to be highly similar to an FDA-licensed reference biological product. The BPCIA attempts to minimize duplicative testing, and thereby lower development costs and increase patient access to affordable treatments. An application for licensure of a biosimilar product must include information demonstrating biosimilarity based upon the following, unless the FDA determines otherwise:
| • | Analytical studies demonstrating that the proposed biosimilar product is highly similar to the approved product notwithstanding minor differences in clinically inactive components; |
| • | Animal studies (including the assessment of toxicity); and |
| • | A clinical trial or trials (including the assessment of immunogenicity and pharmacokinetic or pharmacodynamic) sufficient to demonstrate safety, purity and potency in one or more conditions for which the reference product is licensed and intended to be used. |
In addition, an application must include information demonstrating that:
| • | The proposed biosimilar product and reference product utilize the same mechanism of action for the condition(s) of use prescribed, recommended or suggested in the proposed labeling, but only to the extent the mechanism(s) of action are known for the reference product; |
| • | The condition or conditions of use prescribed, recommended or suggested in the labeling for the proposed biosimilar product have been previously approved for the reference product; |
| • | The route of administration, the dosage form and the strength of the proposed biosimilar product are the same as those for the reference product; and |
| • | The facility in which the biological product is manufactured, processed, packed or held meets standards designed to assure that the biological product continues to be safe, pure and potent. |
Biosimilarity means that the biological product is highly similar to the reference product notwithstanding minor differences in clinically inactive components, and that there are no clinically meaningful differences between the biological product and the reference product in terms of the safety, purity and potency of the product. In addition, the law provides for a designation of “interchangeability” between the reference and biosimilar products, whereby the biosimilar may be substituted for the reference product without the intervention of the healthcare provider who prescribed the reference product. The higher standard of interchangeability must be demonstrated by information sufficient to show that:
| • | The proposed product is biosimilar to the reference product; |
| • | The proposed product is expected to produce the same clinical result as the reference product in any given patient; and |
| • | For a product that is administered more than once to an individual, the risk to the patient in terms of safety or diminished efficacy of alternating or switching between the biosimilar and the reference product is no greater than the risk of using the reference product without such alternation or switch. |
FDA approval is required before a biosimilar may be marketed in the United States. However, complexities associated with the large and intricate structures of biological products and the process by which such products are manufactured pose significant hurdles to the FDA’s implementation of the law that are still being worked out by the FDA. For example, the FDA has discretion over the kind and amount of scientific evidence—laboratory, preclinical and/or clinical—required to demonstrate biosimilarity to a licensed biological product.
The FDA intends to consider the totality of the evidence provided by a sponsor to support a demonstration of biosimilarity and recommends that sponsors use a stepwise approach in the development of their biosimilar products. Biosimilar product applications thus may not be required to duplicate the entirety of preclinical and clinical testing used to establish the underlying safety and effectiveness of the reference product. However, the FDA may refuse to approve a biosimilar application if there is insufficient information to show that the active ingredients are the same or to demonstrate that any impurities or differences in active ingredients do not affect the safety, purity or potency of the biosimilar product. In addition, as with BLAs, biosimilar product applications will not be approved unless the product is manufactured in facilities designed to assure and preserve the biological product’s safety, purity and potency.
The submission of a biosimilar application does not guarantee that the FDA will accept the application for filing and review, as the FDA may refuse to accept applications that it finds are insufficiently complete. The FDA will treat a biosimilar application or supplement as incomplete if, among other reasons, any applicable user fees assessed under the Biosimilar User Fee Act of 2012 have not been paid. In addition, the FDA may accept an application for filing but deny approval on the basis that the sponsor has not demonstrated biosimilarity, in which case the sponsor may choose to conduct further analytical, preclinical or clinical studies and submit a BLA for licensure as a new biological product.
The timing of final FDA approval of a biosimilar for commercial distribution depends on a variety of factors, including whether the manufacturer of the branded product is entitled to one or more statutory exclusivity periods, during which time the FDA is prohibited from approving any products that are biosimilar to the branded product. The FDA cannot approve a biosimilar application for twelve years from the date of first licensure of the reference product.
Additionally, a biosimilar product sponsor may not submit an application for four years from the date of first licensure of the reference product. A reference product may also be entitled to exclusivity under other statutory provisions. For example, a reference product designated for a rare disease or condition (an orphan drug) may be entitled to seven years of exclusivity, in which case no product that is biosimilar to the reference product may be approved until either the end of the twelve-year period provided under the biosimilarity statute or the end of the seven-year orphan drug exclusivity period, whichever occurs later. In certain circumstances, a regulatory exclusivity period can extend beyond the life of a patent, and thus block biosimilarity applications from being approved on or after the patent expiration date. In addition, the FDA may under certain circumstances extend the exclusivity period for the reference product by an additional six months if the FDA requests, and the manufacturer undertakes, studies on the effect of its product in children, a so-called pediatric extension.
Pediatric exclusivity is another type of regulatory market exclusivity in the United States. Pediatric exclusivity, if granted, adds six months to existing regulatory exclusivity periods. This six-month exclusivity may be granted based on the voluntary completion of a pediatric study in accordance with an FDA-issued “Written Request” for such a study.
Post-approval requirements
Drugs and biologics manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior FDA review and approval. There are continuing, annual user fee requirements for any marketed products and the establishments where such products are manufactured, as well as new application fees for supplemental applications with clinical data.
The FDA may impose a number of post-approval requirements as a condition of approval of an NDA. For example, the FDA may require post-marketing testing, including Phase 4 clinical studies, and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization.
In addition, drug and biologic manufacturers and other entities involved in the manufacture and distribution of approved drugs and biologics are required to register their establishments with the FDA and state agencies, and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP requirements and impose reporting and documentation requirements upon the sponsor and any third-party manufacturers that the sponsor may decide to use. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance.
Once an approval of a drug or biologic is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical studies to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, among other things:
| • | Restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| • | Fines, warning letters or holds on post-approval clinical studies; |
| • | Refusal of the FDA to approve pending NDAs or BLAs or supplements to approved NDAs or BLAs, or suspension or revocation of product approvals; |
| • | Product seizure or detention, or refusal to permit the import or export of products; and |
| • | Injunctions or the imposition of civil or criminal penalties. |
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Drugs and biologics may be promoted by a manufacturer and any third parties acting on behalf of a manufacturer only for the approved indications and in a manner consistent with the approved label for the product. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability.
Other healthcare laws
Healthcare providers, physicians, and third party payors play a primary role in the recommendation and prescription of drug products for which the Company obtains marketing approval. Arrangements with third party payors, healthcare providers and physicians, in connection with the clinical research, sales, marketing and promotion of products, once approved, and related activities, may expose a pharmaceutical manufacturer to broadly applicable fraud and abuse and other healthcare laws and regulations. In the United States, these laws include, without limitation, state and federal anti-kickback, physician self-referral prohibitions, false claims, physician transparency, and patient data privacy and security laws and regulations, including but not limited to those described below:
| • | The federal Anti-Kickback Statute, or AKS, which makes it illegal for any person, including a prescription drug manufacturer (or a party acting on its behalf) to knowingly and willfully solicit, receive, offer or pay any remuneration (including any kickback, bribe, or rebate), directly or indirectly, overtly or covertly, in cash or in kind, that is intended to induce or reward, referrals including the purchase recommendation, order or prescription of a particular drug for which payment may be made under a federal healthcare program, such as the Medicare and Medicaid programs. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the federal False Claims Act, or FCA; |
| • | The federal civil and criminal false claims laws, including the FCA, which can be enforced through “qui tam” or “whistleblower” actions, and civil monetary penalty laws, which impose criminal and civil penalties against individuals or entities for, among other things, knowingly presenting, or causing to be presented, claims for payment or approval from Medicare, Medicaid, or other federal health care programs that are false or fraudulent; knowingly making or causing a false statement material to a false or fraudulent claim or an obligation to pay or transmit money or property to the federal government; or knowingly concealing or knowingly and improperly avoiding or decreasing such an obligation. Similar to the AKS and Stark Law, a person or entity does not need to have actual knowledge of these statutes or specific intent to violate them in order to have committed a violation; |
| • | The federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created additional federal criminal statutes that prohibit knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program or obtain, by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the payor (e.g., public or private) and knowingly and willfully falsifying, concealing or covering up by any trick or device a material fact or making any materially false statements in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters; |
| • | HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, or HITECH, and their respective implementing regulations, which impose requirements on certain covered healthcare providers, health plans, and healthcare clearinghouses as well as their respective business associates that perform services for them that involve the creation, use, receipt, maintenance or disclosure of individually identifiable health information, relating to the privacy, security and transmission of individually identifiable health information; |
| • | The federal Physician Payments Sunshine Act, created under Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010, or collectively, the ACA, and its implementing regulations, which require manufacturers of drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health Insurance Program to report annually to the Centers for Medicare and Medicaid Services, or CMS, under the Open Payments Program, information related to payments or other transfers of value made to physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain non-physician healthcare professionals (such as physician assistants and nurse practitioners, among others), and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members; and |
| • | Analogous state and foreign laws and regulations, such as state and foreign anti-kickback, physician self-referral prohibitions, false claims, consumer protection and unfair competition laws which may apply to pharmaceutical business practices, including but not limited to, research, distribution, sales and marketing arrangements as well as submitting claims involving healthcare items or services reimbursed by any third-party payor, including commercial insurers; state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government that otherwise restricts payments that may be made to healthcare providers and other potential referral sources; state laws that require drug manufacturers to file reports with states regarding pricing and marketing information, such as the tracking and reporting of gifts, compensations and other remuneration and items of value provided to healthcare professionals and entities; state and local laws requiring the registration of pharmaceutical sales representatives; and state and foreign laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. |
Because of the breadth of these laws and the narrowness of the statutory exceptions and regulatory safe harbors available, it is possible that some of a pharmaceutical manufacturer’s business activities could be subject to challenge under one or more of such laws. Efforts to ensure that business arrangements comply with applicable healthcare laws involve substantial costs. It is possible that governmental and enforcement authorities will conclude that a pharmaceutical manufacturer’s business practices do not comply with current or future statutes, regulations or case law interpreting applicable fraud and abuse or other healthcare laws and regulations. If any such actions are instituted against a pharmaceutical manufacturer, and it is not successful in defending itself or asserting its rights, those actions could have a significant impact on its business, including the imposition of significant civil, criminal and administrative penalties, damages, disgorgement, imprisonment, monetary fines, possible exclusion from participation in Medicare, Medicaid and other federal healthcare programs, reporting obligations and oversight if the Company becomes subject to integrity and oversight agreements to resolve allegations of non-compliance, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of operations, any of which could adversely affect a pharmaceutical manufacturer’s ability to operate its business and the results of operations. In addition, commercialization of any drug product outside the United States will also likely be subject to foreign equivalents of the healthcare laws mentioned above, among other foreign laws.
Prescription drug advertising is subject to federal, state and foreign regulations. In the United States, the FDA regulates prescription drug promotion, including direct-to-consumer advertising. Prescription drug promotional materials must be submitted to the FDA in conjunction with their first use. Any distribution of prescription drug products and pharmaceutical samples must comply with the United States Prescription Drug Marketing Act, or PDMA, a part of the FDCA. In addition, Title II of the Federal Drug Quality and Security Act of 2013, known as the Drug Supply Chain Security Act, or DSCSA, has imposed new “track and trace” requirements on the distribution of prescription drug products by manufacturers, distributors, and other entities in the drug supply chain. The DSCSA requires product identifiers (i.e., serialization) on prescription drug products in order to eventually establish an electronic interoperable prescription product system to identify and trace certain prescription drugs distributed in the United States and preempts existing state drug pedigree laws and regulations on this topic. The DSCSA also establishes new requirements for the licensing of wholesale distributors and third-party logistic providers. The FDA is in the process of finalizing regulations addressing national standards for the licensure of wholesale distributors and third-party logistics providers.
In the United States, numerous federal and state laws and regulations, including state data breach notification laws, state health information privacy laws, and federal and state consumer protection laws, govern the collection, use, disclosure, and protection of health-related and other personal information. For example, in June 2018, the State of California enacted the California Consumer Privacy Act of 2018, or the CCPA, which came into effect on January 1, 2020 and provides new data privacy rights for consumers and new operational requirements for companies, which may increase the Company’s compliance costs and potential liability. The CCPA gives California residents expanded rights to access and delete their personal information, opt out of certain personal information sharing, and receive detailed information about how their personal information is used. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches that is expected to increase data breach litigation. While there is currently an exception for protected health information that is subject to HIPAA and clinical study regulations, as currently written, the CCPA may impact certain of the Company’s business activities. The CCPA could mark the beginning of a trend toward more stringent state privacy legislation in the United States, which could increase the Company’s potential liability and adversely affect its business.
In the event the Company decides to conduct clinical studies or continue to enroll subjects in its ongoing or future clinical studies, the Company may be subject to additional privacy restrictions. The collection, use, storage, disclosure, transfer, or other processing of personal data regarding individuals in the European Economic Area, or EEA, including personal health data, is subject to the EU General Data Protection Regulation, or GDPR, which became effective on May 25, 2018. The GDPR is wide-ranging in scope and imposes numerous requirements on companies that process personal data, including requirements relating to processing health and other sensitive data, obtaining consent of the individuals to whom the personal data relates, providing information to individuals regarding data processing activities, implementing safeguards to protect the security and confidentiality of personal data, providing notification of data breaches, and taking certain measures when engaging third-party processors. The GDPR also imposes strict rules on the transfer of personal data to countries outside the EEA, including the United States, and permits data protection authorities to impose large penalties for violations of the GDPR, including potential fines of up to €20 million or 4% of annual global revenues, whichever is greater. The GDPR also confers a private right of action on data subjects and consumer associations to lodge complaints with supervisory authorities, seek judicial remedies, and obtain compensation for damages resulting from violations of the GDPR. In addition, the GDPR includes restrictions on cross-border data transfers. The GDPR may increase the Company’s responsibility and liability with respect to personal data that the Company processes where such processing is subject to the GDPR, and it may be required to put in place additional mechanisms to ensure compliance with the GDPR, including as implemented by individual countries. Compliance with the GDPR will be a rigorous and time-intensive process that may increase the Company’s cost of doing business or require it to change its business practices, and despite those efforts, there is a risk that the Company may be subject to fines and penalties, litigation, and reputational harm in connection with its European activities. Further, the United Kingdom’s decision to leave the EU, often referred to as Brexit, has created uncertainty with regard to data protection regulation in the United Kingdom and transfers of personal data to the UK and from the UK to both the EEA and countries outside the UK/EEA. For the time being, transfers of personal data from the EU to the UK are covered by an adequacy decision of the EU Commission, and the UK has recently implemented its own regime for safeguarding transfers from the UK to countries outside the UK/EEA which sit alongside the new EU safeguards which were brought in during 2021. However, both the adequacy decision and the UK regime remain vulnerable to withdrawal or legal challenge. Further both the new UK and EU personal data transfer regimes remain relatively untested and therefore impose risk that a transfer of personal data and/or its subsequent processing would be held unlawful and give rise to liabilities from administrative fines and/or damages claims from data subjects.
Current and future healthcare reform legislation
In both the United States and certain foreign jurisdictions, there have been a number of legislative and regulatory changes to the health care system. In particular, in 2010 the ACA was enacted, which, among other things, increased the minimum Medicaid rebates owed by most manufacturers under the Medicaid Drug Rebate Program, extended the Medicaid Drug Rebate Program to utilization of prescriptions of individuals enrolled in Medicaid managed care organizations, subjected manufacturers to new annual fees and taxes for certain branded prescription drugs, and provided incentives to programs that increase the federal government’s comparative effectiveness research.
In addition, other legislative changes have been proposed and adopted in the United States since the ACA was enacted. In August 2011, the Budget Control Act of 2011, among other things, created measures for spending reductions by Congress. A Joint Select Committee on Deficit Reduction, tasked with recommending a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, was unable to reach required goals, thereby triggering the legislation’s automatic reduction to several government programs. This includes aggregate reductions of Medicare payments to providers of 2% per fiscal year, which went into effect in 2013, and, due to subsequent legislative amendments, will remain in effect through 2031, with the exception of a temporary suspension implemented under various COVID-19 relief legislation from May 1, 2020, through March 31, 2022. Under current legislation, the actual reduction in Medicare payments can vary from 1% in 2022 to up to 4% in the final fiscal year of this sequester. The American Taxpayer Relief Act of 2012 further reduced Medicare payments to several providers, including hospitals and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. The Bipartisan Budget Act of 2018, also amended the ACA, effective January 1, 2019, by increasing the point-of-sale discount that is owed by pharmaceutical manufacturers who participate in Medicare Part D and closing the coverage gap in most Medicare drug plans, commonly referred to as the “donut hole”.
Additionally, there has been heightened governmental scrutiny in the United States of pharmaceutical pricing practices in light of the rising cost of prescription drugs and biologics. In August 2022, Congress passed the Inflation Reduction Act of 2022, which includes prescription drug provisions that have significant implications for the pharmaceutical industry and Medicare beneficiaries, including allowing the federal government to negotiate a maximum fair price for certain high-priced single source Medicare drugs, imposing penalties and excise tax for manufacturers that fail to comply with the drug price negotiation requirements, requiring inflation rebates for all Medicare Part B and Part D drugs, with limited exceptions, if their drug prices increase faster than inflation, and redesigning Medicare Part D to reduce out-of-pocket prescription drug costs for beneficiaries, among other changes. The impact of these legislative, executive, and administrative actions and any future healthcare measures and agency rules implemented by the Biden administration on us and the pharmaceutical industry as a whole is unclear. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize any of the product candidates for which we receive approval. At the state level, legislatures have increasingly passed legislation and implemented regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures and, in some cases, designed to encourage importation from other countries and bulk purchasing.
Legislative and regulatory proposals, and enactment of laws, at the foreign, federal and state levels, directed at containing or lowering the cost of healthcare, will continue into the future.
For other countries outside of the European Union and the United States, such as countries in Eastern Europe, Latin America or Asia, the requirements governing product development, the conduct of clinical studies, manufacturing, distribution, marketing approval, product licensing, pricing and reimbursement vary from country to country. Additionally, clinical studies must be conducted in accordance with GCP requirements and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
If the Company fails to comply with applicable foreign regulatory requirements, the Company may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Additionally, to the extent that any of the Company’s product candidates, once approved, are sold in a foreign country, it may be subject to applicable post-marketing requirements, including safety surveillance, anti-fraud and abuse laws and implementation of corporate compliance programs and reporting of payments or other transfers of value to healthcare professionals.
Coverage and reimbursement
Successful commercialization of new drug products depends in part on the extent to which reimbursement for those drug products will be available from government health administration authorities, private health insurers, and other organizations. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which drug products they will pay for and establish reimbursement levels. The availability and extent of reimbursement by governmental and private payors is essential for most patients to be able to afford a drug product. Sales of drug products depend substantially, both domestically and abroad, on the extent to which the costs of drugs products are paid for by health maintenance, managed care, pharmacy benefit and similar healthcare management organizations, or reimbursed by government health administration authorities, private health coverage insurers and other third-party payors.
A primary trend in the United States healthcare industry and elsewhere is cost containment. Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular drug products. In many countries, the prices of drug products are subject to varying price control mechanisms as part of national health systems. In general, the prices of drug products under such systems are substantially lower than in the United States. Other countries allow companies to fix their own prices for drug products, but monitor and control company profits. Accordingly, in markets outside the United States, the reimbursement for drug products may be reduced compared with the United States.
In the United States, the principal decisions about reimbursement for new drug products are typically made by CMS, an agency within the U.S. Department of Health and Human Services. CMS decides whether and to what extent a new drug product will be covered and reimbursed under Medicare, and private payors tend to follow CMS to a substantial degree. However, no uniform policy of coverage and reimbursement for drug products exists among third-party payors and coverage and reimbursement levels for drug products can differ significantly from payor to payor.
The Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or the MMA, established the Medicare Part D program to provide a voluntary prescription drug benefit to Medicare beneficiaries. Under Part D, Medicare beneficiaries may enroll in prescription drug plans offered by private entities that provide coverage of outpatient prescription drugs. While all Medicare drug plans must give at least a standard level of coverage set by Medicare, Part D prescription drug plan sponsors are not required to pay for all covered Part D drugs, and each Part D prescription drug plan can develop its own drug formulary that identifies which drugs it will cover and at what tier or level. However, Part D prescription drug formularies must include drugs within each therapeutic category and class of covered Part D drugs, though not necessarily all the drugs in each category or class. Any formulary used by a Part D prescription drug plan must be developed and reviewed by a pharmacy and therapeutic committee. Government payment for some of the costs of prescription drugs may increase demand for drugs for which the Company obtains marketing approval. Any negotiated prices for any of the Company’s products covered by a Part D prescription drug plan will likely be lower than the prices it might otherwise obtain. Moreover, while the MMA applies only to drug benefits for Medicare beneficiaries, private payors often follow Medicare coverage policy and payment limitations in setting their own payment rates. Any reduction in payment that results from the MMA may result in a similar reduction in payments from non-governmental payors.
For a drug product to receive federal reimbursement under the Medicaid or Medicare Part B programs or to be sold directly to United States government agencies, the manufacturer must extend discounts to entities eligible to participate in the 340B drug pricing program. The required 340B discount on a given product is calculated based on the average manufacturer price, or AMP, and Medicaid rebate amounts reported by the manufacturer. As of 2010, the ACA expanded the types of entities eligible to receive discounted 340B pricing, although under the current state of the law these newly eligible entities (with the exception of children’s hospitals) will not be eligible to receive discounted 340B pricing on orphan drugs. As 340B drug pricing is determined based on AMP and Medicaid rebate data, the revisions to the Medicaid rebate formula and AMP definition described above could cause the required 340B discount to increase. If third-party payors do not consider the Company’s drugs to be cost-effective compared to other available therapies, they may not cover the Company’s drugs after approval as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow the Company to sell its drugs on a profitable basis.
These laws, and state and federal healthcare reform measures that may be adopted in the future, may result in additional reductions in Medicare and other healthcare funding and otherwise affect the prices the Company may obtain for any product candidates for which it may obtain regulatory approval or the frequency with which any such product candidate is prescribed or used.
Outside of the United States, the pricing of pharmaceutical products and medical devices is subject to governmental control in many countries. For example, in the European Union, pricing and reimbursement schemes vary widely from country to country. Some countries provide that products may be marketed only after a reimbursement price has been agreed. Some countries may require the completion of additional studies that compare the cost effectiveness of a particular therapy to currently available therapies or so-called health technology assessments, in order to obtain reimbursement or pricing approval. Other countries may allow companies to fix their own prices for products, but monitor and control product volumes and issue guidance to physicians to limit prescriptions. Efforts to control prices and utilization of pharmaceutical products and medical devices will likely continue as countries attempt to manage healthcare expenditures.
Employees and Human Capital Resources
As of December 31, 2022, the Company had 37 full-time employees/consultants, including 10 with Ph.D. or M.D. degrees and 13 who are engaged in research and development activities. The Company is dependent on its management and scientific personnel, and it is crucial that it continues to attract and retain valuable employees. To facilitate attraction and retention, the Company strives to make itself an inclusive and safe workplace, with opportunities for its employees to grow and develop in their careers, supported by strong compensation and benefits programs. None of the Company’s employees are represented by labor unions or covered by collective bargaining agreements.
Corporate Information and History
We were incorporated on September 22, 2011, under the laws of the State of Israel. In March 2021, in connection with the Merger, we changed our name from Anchiano Therapeutics Ltd. to Chemomab Therapeutics Ltd. Our principal executive offices are located at Kiryat Atidim, Building 7, Tel Aviv, Israel 6158002, and our phone number is +972-77-331-0156. Our website is: www.chemomab.com. The information contained on, or that can be accessed through, our website is not incorporated by reference into this Annual Report.
Available Information
Our investor relations website is https://investors.chemomab.com/. We promptly make available on our investor relations website, free of charge, the reports that we file or furnish with the SEC, corporate governance information (including our Code of Business Conduct and Ethics) and all press releases. We file annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy and information statements and amendments to reports filed or furnished pursuant to the Exchange Act. The SEC maintains a website at www.sec.gov that contains reports, proxy and information statements and other information regarding Chemomab and other issuers that file electronically with the SEC.
Item 1A. Risk Factors
You should consider carefully the following information about the risks described below, together with the other information contained in this Annual Report on Form 10-K and in our other public filings, in evaluating our business. If any of the following risks actually occurs, our business, financial condition, results of operations and future growth prospects would likely be materially and adversely affected. In these circumstances, the market price of our common stock would likely decline.
Risks Related to Chemomab’s Business, Research and Development and the Biopharmaceutical Industry
Chemomab has a limited operating history and funding, which may make it difficult to evaluate its prospects and likelihood of success.
Chemomab is a clinical-stage biopharmaceutical company with a limited operating history. Chemomab was incorporated in 2015, has no products approved for commercial sale and has not generated any revenue. Its operations to date have been limited to organizing and staffing the company, business planning, raising capital, establishing its intellectual property portfolio and conducting research and development of its product candidates, technology related to CCL24 and novel therapies for the treatment of inflammation and fibrosis. Chemomab’s approach to the discovery and development of product candidates is unproven, and Chemomab does not know whether it will be able to develop any products of commercial value. In addition, Chemomab’s lead product candidate, CM-101, is in early clinical development for the treatment of PSC and SSc. The clinical programs will require substantial additional development and clinical research, both in time and resources, before Chemomab is in a position to apply for or receive regulatory approvals and begin generating revenue in connection with the sale of such product candidates. Chemomab has not yet demonstrated the ability to successfully complete a large-scale, pivotal clinical trial, obtain marketing approval, manufacture a commercial scale product, or arrange for a third party to do so on its behalf, or conduct sales and marketing activities necessary for successful product commercialization. Consequently, predictions about Chemomab’s future success or viability may not be as accurate as they could be if Chemomab had a longer operating history or a history of successfully developing and commercializing pharmaceutical products.
In addition, as a business with a limited operating history, Chemomab may encounter unforeseen expenses, difficulties, complications, delays and other known or unknown factors and risks frequently experienced by early-stage biopharmaceutical companies in rapidly evolving fields. Consequently, Chemomab has no meaningful history of operations upon which to evaluate its business, and predictions about its future success or viability may not be as accurate as they could be if Chemomab had a longer operating history or a history of successfully developing and commercializing drug products. Chemomab will eventually need to transition from a company with a research and development focus to a company capable of supporting commercial activities. Chemomab may not be successful in such a transition and, as a result, its business may be adversely affected.
As Chemomab continues to build its business, it expects its financial condition and operating results to fluctuate significantly from quarter to quarter and year to year due to a variety of factors, many of which are beyond Chemomab’s control.
Chemomab’s business is highly dependent on the success of its lead product candidate, CM-101, and any other product candidates that it advances into clinical studies. All of Chemomab’s programs will require significant additional clinical development.
Chemomab currently has no products that are approved for commercial sale and may never be able to develop marketable products. Chemomab is very early in its development efforts and has only one product candidate, CM-101, in early clinical development. Because CM-101 is Chemomab’s lead product candidate, if CM-101 encounters safety or efficacy problems, development delays, regulatory issues or other problems, Chemomab’s development plans and business would be significantly harmed. Chemomab has completed a Phase 1a SAD study with healthy volunteers, a Phase 1b MAD study of CM-101 in non-alcoholic fatty liver disease, or NAFLD, a Phase 2a safety, Pk and liver fibrosis biomarker study in NASH patients, an open-label exploratory study in severe lung injury in hospitalized COVID-19 patients and is recruiting patients in a Phase 2 PSC trial. Chemomab plans to initiate a Phase 2 SSc study around midyear of 2023.
Chemomab expects that a substantial portion of its efforts and expenditures over the next few years will be devoted to CM-101, which will require additional clinical development, management of clinical and manufacturing activities, regulatory approval in multiple jurisdictions, obtaining manufacturing supply, building of a commercial organization, substantial investment and significant marketing efforts before it can generate any revenues from any commercial sales. Chemomab cannot be certain that it will be able to successfully complete any of these activities. In addition, if one or more of Chemomab’s product candidates are approved, Chemomab may need to ensure access to sufficient commercial manufacturing capacity and conduct significant marketing efforts in connection with any commercial launch. These efforts will require substantial investment, and Chemomab may not have the financial resources to continue the development of its product candidates.
Chemomab will need to raise substantial additional funds through public or private equity or debt transactions and/or complete one or more strategic transactions or partnerships, to complete development of CM-101 or any other product candidates. If Chemomab is unable to raise such financing or complete such a transaction, it may not be able to fund the clinical trials of its product candidates and potentially commercialize those product candidates.
As a result of the expected development timeline to potentially obtain FDA approval for CM-101, the substantial additional costs associated with the development of our product candidates, including the costs associated with clinical trials related thereto, and the substantial cost of commercializing CM-101, Chemomab will need to raise substantial additional funding through public or private equity or debt transactions or a strategic combination or partnership. If Chemomab is delayed in obtaining funding or is unable to complete a strategic transaction, Chemomab may have to delay or discontinue development activities on CM-101 and our other product candidates. Even if Chemomab is able to fund continued development of CM-101 or any of our other product candidates is approved, Chemomab expects that it will need to raise substantial additional funding through public or private equity or debt securities or complete a strategic transaction or partnership to successfully commercialize CM-101 or any other product candidate.
Chemomab believes its cash and cash equivalents and bank deposits as of December 31, 2022 will be sufficient to fund its operations at least through March 31, 2024. Sales of Chemomab’s ADSs dilute the ownership interest of its shareholders and may cause the price per ADS to decrease. Changing circumstances may cause us to consume capital significantly faster or slower than Chemomab currently anticipates. Chemomab has based these estimates on assumptions that may prove to be wrong, and Chemomab could exhaust its available financial resources sooner than currently anticipated.
Chemomab’s liquidity, and ability to raise additional capital or complete any strategic transaction, depends on a number of factors, including, but not limited to, the following:
| • | the costs and timing for potential additional clinical trials in order to gain possible regulatory approval for CM-101and our other product candidates; |
| • | the market price of Chemomab’s ADSs and the availability and cost of additional equity capital from existing and potential new investors; |
| • | Chemomab’s ability to retain the listing of its ADSs on the Nasdaq Capital Market; |
| • | general economic and industry conditions affecting the availability and cost of capital, including as a result of deteriorating market conditions due to investor concerns regarding inflation and continued hostilities between Russia and Ukraine; |
| • | Chemomab’s ability to control costs associated with its operations; |
| • | the costs of filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; and |
| • | the terms and conditions of our existing collaborative and licensing agreements. |
The sale of additional equity or convertible debt securities would likely result in substantial dilution to our stockholders. If Chemomab raise additional funds through the incurrence of indebtedness, the obligations related to such indebtedness would be senior to rights of holders of our capital stock and could contain covenants that would restrict our operations. Chemomab also cannot predict what consideration might be available, if any, to us or our stockholders, in connection with any strategic transaction. Should strategic alternatives or additional capital not be available to us, or not be available on acceptable terms, Chemomab may be unable to realize value from its assets and discharge its liabilities in the normal course of business which may, among other alternatives, cause Chemomab to further delay, substantially reduce or discontinue operational activities to conserve our cash resources.
Chemomab’s approach in the area of fibrotic diseases is novel and unproven and may not result in marketable products.
Chemomab’s central objective is to design and develop targeted treatments for inflammation and fibrosis with an initial focus on the neutralization of CCL24 signaling, which is known to regulate fibrotic and inflammatory processes. While several studies are currently underway, this mechanism has not yet been definitively proven to successfully treat inflammation and fibrosis. Targeting CCL24 to treat inflammation and fibrosis is a novel approach in a rapidly developing field, and there can be no assurance that Chemomab can avoid unforeseen problems or delays in the development of its product candidates, that such problems or delays will not result in unanticipated costs, or that any such development problems can or will be solved. Chemomab has only tested its lead product candidate, CM-101, in early trials in healthy volunteers, NAFLD, NASH and COVID-19 lung injury patients. Therefore, Chemomab may ultimately discover that its approach does not possess properties required for therapeutic effectiveness. As a result, Chemomab may elect to abandon the program or never succeed in developing a marketable product, which would have a significant effect on the success and profitability of its business.
Clinical development involves a lengthy, complex and expensive process, with an uncertain outcome.
Before obtaining the requisite regulatory approvals from the FDA or other comparable foreign regulatory authorities for the sale of any of its product candidates, Chemomab must support its application with clinical studies that prove that such product candidate is safe and effective in humans. Clinical testing is expensive and can take many years to complete, and its outcome is inherently uncertain. In particular, the general approach for FDA approval of a new drug requires positive data from two well-controlled Phase 3 clinical studies of the relevant drug in the relevant patient population. Failure can occur at any time during the clinical study process. Chemomab may experience delays in initiating and completing any clinical studies that it is conducting or intends to conduct, including as a result of the COVID-19 pandemic or other public health emergencies, and Chemomab does not know whether its ongoing or planned clinical studies will begin or progress on schedule, need to be redesigned, enroll patients on time or be completed on schedule, or at all.
Phase 3 clinical studies typically involve hundreds of patients, have significant costs and take years to complete. A product candidate can fail at any stage of testing, even after observing promising signals of activity in earlier preclinical studies or clinical trials. The results of preclinical studies and early clinical trials of Chemomab’s product candidates may not be predictive of the results of later-stage clinical studies. In addition, initial or interim success in clinical studies may not be indicative of results obtained when such studies are completed. There is typically an extremely high rate of attrition from the failure of product candidates proceeding through clinical studies. Product candidates in later stages of clinical studies may fail to show the desired safety and efficacy profile despite having progressed through preclinical studies and initial clinical trials. A number of companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical studies due to lack of efficacy or unacceptable safety issues, notwithstanding promising results in earlier studies. Most product candidates that commence clinical studies are never approved as products and there can be no assurance that any of Chemomab’s future clinical studies will ultimately be successful or support further clinical development of CM-101. Product candidates that appear promising in the early phases of development may fail to reach the market for several reasons, including:
| | • | the FDA or comparable foreign regulatory authorities disagreeing as to the design or implementation of Chemomab’s clinical studies; |
| | • | obtaining regulatory authorizations to commence a trial or consensus with regulatory authorities on trial’s design; |
| | • | reaching an agreement on acceptable terms with prospective clinical research organizations, or CROs, and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; |
| | • | obtaining IRB approval at each site, or Independent Ethics Committee, or IEC, approval at sites outside the United States; |
| | • | imposition of a clinical hold by regulatory authorities, including as a result of unforeseen safety issues or side effects or failure of trial sites to adhere to regulatory requirements or follow trial protocols; |
| | • | clinical studies may show the product candidates to be less effective than expected (e.g., a clinical study could fail to meet its primary endpoint(s)) or to have unacceptable side effects or toxicities; |
| | • | failure to establish clinical endpoints that applicable regulatory authorities would consider clinically meaningful; |
| | • | the occurrence of serious adverse events in trials of the same class of agents conducted by other companies; |
| | • | adding a sufficient number of clinical study sites; |
| | • | manufacturing sufficient quantities of product candidate with sufficient quality for use in clinical studies; |
| | • | having patients complete a trial or return for post-treatment follow-up; |
| | • | recruiting suitable patients to participate in a trial in a timely manner and in sufficient numbers; |
| | • | a facility manufacturing Chemomab’s product candidates or any of their components being ordered by the FDA or comparable foreign regulatory authorities to temporarily or permanently shut down due to violations of current good manufacturing practice, or cGMP, regulations or other applicable requirements, or infections or cross-contaminations of product candidates in the manufacturing process; |
| | • | third-party clinical investigators losing the licenses or permits necessary to perform Chemomab’s clinical studies, not performing its clinical studies on its anticipated schedule or consistent with the clinical study protocol, GCP, or other regulatory requirements; |
| | • | third-party contractors not performing data collection or analysis in a timely or accurate manner; |
| | • | manufacturing costs, formulation issues, pricing or reimbursement issues, or other factors that make a product candidate uneconomical; or |
| | • | the proprietary rights of others and their competing products and technologies that may prevent Chemomab’s product candidates from being commercialized. |
In addition, differences in trial design between early-stage clinical studies and later-stage clinical studies make it difficult to extrapolate the results of earlier clinical studies to later clinical studies. Moreover, clinical data are often susceptible to varying interpretations and analyses, and many companies that have believed their product candidates performed satisfactorily in clinical studies have nonetheless failed to obtain marketing approval of their products.
In addition, the standards used by the FDA and comparable foreign regulatory authorities when regulating Chemomab require judgment and can change, which makes it difficult to predict with certainty how they will be applied. For more information, see “Risk Factors - Risks Related to Chemomab’s Regulatory Approvals.”
Successful completion of clinical studies is a prerequisite to submitting a marketing application to the FDA and similar marketing applications to comparable foreign regulatory authorities, for each product candidate and, consequently, the ultimate approval and commercial marketing of any product candidates. Chemomab may experience negative or inconclusive results, which may result in it deciding, or it being required by regulators, to conduct additional clinical studies or trials or abandon some or all of its product development programs, which could have a material adverse effect on Chemomab’s business.
Chemomab may incur additional costs or experience delays in completing the development and commercialization of CM-101 or any other product candidates.
Chemomab may experience delays in initiating or completing clinical studies. It also may experience numerous unforeseen events during, or as a result of, any future clinical studies that could delay or prevent its ability to receive marketing approval or commercialize CM-101 or any other product candidates, including:
| | • | regulators, IRBs, or IECs may not authorize Chemomab or its investigators to commence a clinical study or conduct a clinical study at a prospective trial site;
|
| | • | the FDA or other comparable regulatory authorities may disagree with Chemomab’s clinical study design, including with respect to dosing levels administered in its planned clinical studies, which may delay or prevent Chemomab from initiating its clinical studies with its originally intended trial design; |
| | • | Chemomab may experience delays in reaching, or fail to reach, agreement on acceptable terms with prospective trial sites and prospective CROs, which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; |
| | • | the number of subjects required for clinical studies of any product candidates may be larger than Chemomab anticipates or subjects may drop out of these clinical studies or fail to return for post-treatment follow-up at a higher rate than it anticipates;
|
| | • | Chemomab’s third-party contractors may fail to comply with regulatory requirements or meet its contractual obligations to Chemomab in a timely manner, or at all, or may deviate from the clinical study protocol or drop out of the trial, which may require that Chemomab add new clinical study sites or investigators;
|
| | • | due to the impact of the COVID-19 pandemic, or other emerging public health threats, Chemomab has experienced, and may continue to experience, delays and interruptions to clinical studies, it may experience delays or interruptions to its manufacturing supply chain, or it could suffer delays in reaching, or it may fail to reach, agreement on acceptable terms with third-party service providers on whom it relies;
|
| | • | additional delays and interruptions to Chemomab’s clinical studies could extend the duration of the trials and increase the overall costs to finish the trials as its fixed costs are not substantially reduced during delays;
|
| | • | Chemomab may elect to, or regulators, IRBs, Data Safety Monitoring Boards or ethics committees may require that it or its investigators, suspend or terminate clinical research or trials for various reasons, including noncompliance with regulatory requirements or a finding that the participants are being exposed to unacceptable health risks;
|
| | • | Chemomab may not have the financial resources available to begin and complete the planned trials, or the cost of clinical studies of any product candidates may be greater than it anticipates; and
|
| | •
| the supply or quality of Chemomab’s product candidates or other materials necessary to conduct clinical studies of its product candidates may be insufficient or inadequate to initiate or complete a given clinical study. |
Chemomab’s product development costs will increase if it experiences additional delays in clinical testing or in obtaining marketing approvals. Chemomab does not know whether any of its clinical studies will begin as planned, will need to be restructured or will be completed on schedule, or at all. If Chemomab does not achieve its product development goals in the time frames it announces and expects, the approval and commercialization of its product candidates may be delayed or prevented entirely. Significant clinical study delays also could shorten any periods during which it may have the exclusive right to commercialize its product candidates and may allow its competitors to bring products to market before Chemomab does, potentially impairing its ability to successfully commercialize its product candidates and harming its business and results of operations. Any delays in Chemomab’s clinical development programs may harm its business, financial condition and results of operations significantly.
Chemomab’s ongoing and future clinical studies may reveal significant adverse events or immunogenicity related responses and may result in a safety profile that could delay or prevent regulatory approval or market acceptance of its product candidate.
Chemomab completed its Phase 1a and Phase 1b and Phase 2a clinical studies of its lead product candidate, CM-101, in healthy volunteers, NAFLD, NASH and COVID-19 lung injury patients, and, with the exception of a number of reported minor adverse events (including mild headaches, changes in blood pressure and mild-moderate increases in liver enzymes,) and one serious adverse event (a transient ischemic attack or seizure judged to be unrelated to administration of CM-101), CM-101 was observed to be generally well-tolerated across all doses in about 70 trial participants. Some potential therapeutics developed in the biopharmaceutical industry that initially showed therapeutic promise in early-stage trials have later been found to cause side effects that prevented their further development and ultimately commercialization. Even if side effects do not preclude the product candidate from obtaining or maintaining marketing approval, undesirable side effects may inhibit market acceptance of the approved product due to its tolerability versus other therapies.
Protein biopharmaceuticals, including, monoclonal antibodies, or mAbs, may be immunogenic and promote immune responses against themselves. In particular, anti-drug antibodies, or ADAs, may be produced by patients following infusion mAbs and may disturb the pharmacokinetics of mAbs, neutralize their therapeutic activities or induce allergic or autoimmune symptoms. Clinical immunogenicity can range from mild, transient antibody responses with no apparent clinical manifestations to loss of therapeutic efficacy and even life-threatening reactions. Several approved therapeutic antibodies have been found to induce neutralizing antibodies, as illustrated by the approved anti-TNFa antibodies infliximab and adalimumab as well as the approved anti-IL-17 mAb ixekizumab. Chemomab’s product candidate, CM-101, is a humanized antibody that, similar to other humanized approved mAbs, was shown to include several non-germline sequences that may serve as a source for immunogenicity in therapeutic antibodies. Clinical studies to date have not identified any anti-drug antibodies, or ADAs. Additional larger clinical studies will be needed to address the risk of immunogenicity and, if discovered, Chemomab’s business will be materially and adversely affected.
Additionally, if unacceptable side effects, including materialized risks of immunogenicity, do arise in the development of Chemomab’s product candidates, Chemomab, the FDA or the IRBs at the institutions in which its studies are conducted, or the Data Safety Monitoring Board, if constituted for its clinical studies, could recommend a suspension or termination of Chemomab’s clinical studies, or the FDA or comparable foreign regulatory authorities could order Chemomab to cease further development of or deny approval of a product candidate for any or all targeted indications. In addition, drug-related side effects could affect patient recruitment or the ability of enrolled patients to complete a trial or result in potential product liability claims. In addition, these side effects may not be appropriately recognized or managed by the treating medical staff. Chemomab expects to have to train medical personnel using its product candidates to understand the side effect profiles for its clinical studies and upon any commercialization of any of its product candidates. Inadequate training in recognizing or managing the potential side effects of its product candidates could result in patient injury or death. Any of these occurrences may harm Chemomab’s business, financial condition and prospects significantly.
Additionally, if one or more of Chemomab’s product candidates receives marketing approval, and Chemomab or others later identify undesirable side effects caused by such products, a number of potentially significant negative consequences could result, including:
| | • | regulatory authorities may withdraw approvals of such product; |
| | • | regulatory authorities may require additional warnings on the label, such as a “black box” warning or contraindication; |
| | • | additional restrictions may be imposed on the marketing of the particular product or the manufacturing processes for the product or any component thereof; |
| | • | Chemomab may be required to implement a Risk Evaluation and Mitigation Strategy, or REMS, or create a medication guide outlining the risks of such side effects for distribution to patients;
|
| | • | Chemomab could be sued and held liable for harm caused to patients; |
| | • | the product may become less competitive; and |
| | • | Chemomab’s reputation may suffer. |
Any of these events could prevent Chemomab from achieving or maintaining market acceptance of a product candidate, if approved, and could significantly harm Chemomab’s business, results of operations and prospects.
Interim, topline and preliminary data from our clinical trials that we announce or publish from time to time may change as more patient data become available or as additional analyses are conducted and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose preliminary, interim or topline data from our clinical trials. The preliminary data is based on a preliminary analysis of then available data, and the results and related findings and conclusions are subject to change following a more comprehensive review of the data related to the particular study or trial. For example, we may report tumor responses in certain patients that are unconfirmed at the time and which do not ultimately result in confirmed responses to treatment after follow-up evaluations. We also make assumptions, estimations, calculations and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the topline results that we report may differ from future results of the same studies, or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Topline data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, topline data should be viewed with caution until the final data are available. In addition, we may report interim analyses of only certain endpoints rather than all endpoints. Interim data from clinical trials that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Adverse changes between interim data and final data could significantly harm our business and prospects. Further, additional disclosure of interim data by us or by our competitors in the future could result in volatility in the price of our common stock.
In addition, the information we choose to publicly disclose regarding a particular clinical trial is typically selected from a more extensive amount of available information. You or others may not agree with what we determine is the material or otherwise appropriate information to include in our disclosure, and any information we determine not to disclose may ultimately be deemed significant with respect to future decisions, conclusions, views, activities or otherwise regarding a particular product candidate or our business. If the preliminary or topline data that we report differ from late, final or actual results, or if others, including regulatory authorities, disagree with the conclusions reached, our ability to obtain approval for any product candidates that we may develop in the future may be harmed, which could harm our business, financial condition, results of operations and prospects.
Changes in methods of product candidate manufacturing or formulation may result in additional costs or delay.
As product candidates progress through preclinical studies and clinical trials to regulatory approval and commercialization, it is common that various aspects of the development program, such as manufacturing methods and formulation, are altered along the way in an effort to optimize yield and manufacturing batch size, minimize costs and achieve consistent quality and results. Any material manufacturing changes made to any product candidate that we may develop could perform differently and affect the results of planned clinical trials or other clinical trials conducted with the altered materials. This could delay completion of clinical trials, require the conduct of bridging clinical trials or the repetition of one or more clinical trials, increase clinical trial costs, delay approval of our product candidates and jeopardize our ability to commercialize any product candidates that we may develop in the future, if approved, and generate revenue.
If Chemomab encounters difficulties enrolling patients in its clinical studies, including due to COVID-19 or other public health emergencies, its clinical development activities could be delayed or otherwise adversely affected.
Chemomab may experience difficulties in patient enrollment in its clinical studies for a variety of reasons. The timely completion of clinical studies in accordance with its protocols depends, among other things, on Chemomab’s ability to enroll a sufficient number of patients who remain in the trial until its conclusion. The enrollment of patients depends on many factors, including:
| | • | the patient eligibility and exclusion criteria defined in the protocol;
|
| | • | the need to receive study drug via an IV infusion;
|
| | • | the size of the patient population required for analysis of the trial’s primary endpoints and the process for identifying patients; |
| | • | the willingness or availability of patients to participate in Chemomab’s trials (including due to fears of contracting COVID-19); |
| | • | the proximity of patients to trial sites; |
| | • | the design of the trial; |
| | • | Chemomab’s ability to recruit clinical study investigators with the appropriate competencies and experience; |
| | • | clinicians’ and patients’ perceptions as to the potential advantages and risks of the product candidate being studied with respect to other available therapies, including any new products that may be approved for the indications Chemomab is investigating; |
| | • | the availability of competing commercially available therapies and other competing product candidates’ clinical studies; |
| | • | Chemomab’s ability to obtain and maintain patient informed consents; and |
| | • | the risk that patients enrolled in clinical studies will drop out of the trials before completion. |
Further, timely enrollment in clinical studies is reliant on clinical study sites which may be adversely affected by global health matters, including, among other things, pandemics. For example, Chemomab’s clinical study sites have been affected by the COVID-19 pandemic. Commencement of the enrollment of Chemomab’s clinical studies of CM-101 in PSC had been delayed due to the pandemic.
Some factors from a resurgent COVID-19 pandemic or other emerging public health emergencies that Chemomab believes could potentially adversely affect enrollment in its trials include:
| | • | the diversion of healthcare resources away from the conduct of clinical study matters to focus on pandemic concerns, including the attention of infectious disease physicians serving as Chemomab’s clinical study investigators, hospitals serving as Chemomab’s clinical study sites and hospital staff supporting the conduct of its clinical studies; |
| | • | the inability of patients to come to hospitals to participate in Chemomab’s trials, which may force Chemomab to conduct its trials in patients’ homes, rendering the trials more difficult and costly to conduct; |
| | • | limitations on travel that interrupt key trial activities, such as clinical study site initiations and monitoring; and |
| | • | employee furlough days that delay necessary interactions with local regulators, ethics committees and other important agencies and contractors. |
These and other factors arising from a resurgent COVID-19 pandemic or other emerging public health emergencies could adversely impact Chemomab’s clinical studies.
The market opportunities for CM-101, if approved, may be smaller than Chemomab anticipates.
Chemomab expects to initially seek approval of CM-101 for the treatment of PSC and SSc. Its projections of the number of PSC and SSc patients is based on its beliefs and estimates. These estimates have been derived from a variety of sources, including scientific literature, patient foundations and publicly available databases, and may prove to be incorrect. Further, new sources may reveal a change in the estimated number of patients, and the number of patients may turn out to be lower than Chemomab expected. The potential addressable patient population for Chemomab’s current programs or future product candidates may be limited. The ultimate market opportunity for Chemomab’s product candidates will depend on, among other things, the final labeling for such product candidates as agreed with the FDA or comparable foreign regulatory authorities, acceptance by the medical community and patient access, potential competition and drug pricing and reimbursement. Even if Chemomab obtains significant market share for any product candidate, if approved, if the potential target populations are small, Chemomab may never achieve profitability without obtaining marketing approval for additional indications.
Chemomab may not be successful in its efforts to identify or discover additional product candidates in the future.
Chemomab’s research programs may initially show promise in identifying potential product candidates, yet may fail to yield product candidates for clinical development for a number of reasons, including:
| | • | Chemomab’s inability to design such product candidates with the pharmacological properties that it desires or attractive pharmacokinetics; or |
| | • | potential product candidates may, on further study, be shown to have harmful side effects or other characteristics that indicate that they are unlikely to be medicines that will receive marketing approval and achieve market acceptance. |
Research programs to identify new product candidates require substantial technical, financial and human resources. If Chemomab is unable to identify suitable compounds for preclinical and clinical development, it will not be able to obtain product revenue in future periods, which likely would result in significant harm to Chemomab’s financial position and adversely impact the price of its ADSs.
Certain of Chemomab’s key strategic initiatives, including investing in the internal discovery of new product candidates and in-licensing or acquiring new assets to expand Chemomab’s current pipeline, involve various risks that may impair Chemomab’s ability to actualize the foregoing strategies.
| • | The competitive landscape for in-licensing or acquiring assets in the biopharmaceutical sector is intense with several companies employing this growth and diversification strategy. |
| • | Even if appropriate assets are identified, there can be no assurance that a potential transaction can be consummated between the parties. |
| • | If a transaction is concluded on acceptable terms, there can be assurance that the assets in-licensed or acquired will be successful in preclinical and subsequent clinical development. |
| • | The Company will likely need to raise additional capital to close any transaction of significance. As such, there can be no assurance that a fundraising effort will be successful and if successful, it could result in dilution to current shareholders. |
Due to Chemomab’s limited resources and access to capital, it must make decisions on the allocation of resources to certain programs and product candidates; these decisions may prove to be wrong and may adversely affect Chemomab’s business.
Chemomab has limited financial and human resources and intends to initially focus on research programs and product candidates for a limited set of indications. As a result, Chemomab may forgo or delay pursuit of opportunities with other product candidates or for other indications that later prove to have greater commercial potential or a greater likelihood of success.
There can be no assurance that Chemomab will ever be able to identify additional therapeutic opportunities for its product candidates or to develop suitable potential product candidates through internal research programs, which could materially adversely affect its future growth and prospects. Chemomab may focus its efforts and resources on potential product candidates or other potential programs that ultimately prove to be unsuccessful.
If product liability lawsuits are brought against Chemomab, it may incur substantial financial or other liabilities and may be required to limit commercialization of its product candidates.
Chemomab faces an inherent risk of product liability as a result of testing CM-101, and will face an even greater risk if Chemomab commercializes any products. For example, Chemomab may be sued if any of its product candidates cause or are perceived to cause injury or are found to be otherwise unsuitable during clinical studies, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability or a breach of warranties. Claims could also be asserted under state consumer protection acts. If Chemomab cannot successfully defend itself against product liability claims, it may incur substantial liabilities or be required to limit commercialization of its product candidates. Even successful defense would require significant financial and management resources. Chemomab’s inability to obtain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the commercialization of products Chemomab develops. Chemomab will need to obtain additional insurance for clinical studies as it continues clinical development of CM-101 and as additional product candidates enter clinical studies. However, Chemomab may be unable to obtain, or may obtain on unfavorable terms, clinical study insurance in amounts adequate to cover any liabilities from any of its clinical studies. Chemomab’s insurance policies may also have various exclusions, and Chemomab may be subject to a product liability claim for which it has no coverage. Chemomab may have to pay any amount awarded by a court or negotiated in a settlement that exceed its coverage limitations or that are not covered by insurance, and Chemomab may not have, or be able to obtain, sufficient capital to pay such amounts. Even if Chemomab’s agreements with any future corporate collaborators entitles Chemomab to indemnification against losses, such indemnification may not be available or adequate should any claim arise.
Chemomab has been granted Orphan Drug Designation for CM-101 in connection with three indications and may seek Orphan Drug Designation for other indications or product candidates, and Chemomab may be unable to maintain the benefits associated with Orphan Drug Designation, including the potential for market exclusivity, and may not receive Orphan Drug Designation for other indications or for its other product candidates.
Regulatory authorities in some jurisdictions, including the United States and Europe, may designate drugs intended for relatively small patient populations as orphan drugs. Under the Orphan Drug Act, the FDA may designate a drug as an orphan drug if it is a drug intended to treat a rare disease or condition, which is generally defined as a patient population of fewer than 200,000 individuals in the United States, or a patient population greater than 200,000 in the United States where there is no reasonable expectation that the cost of developing the drug will be recovered from sales in the United States. In the United States, Orphan Drug Designation has entitled a party to financial incentives such as opportunities for grant funding toward clinical study costs, tax advantages and user-fee waivers. In addition, if a product that has Orphan Drug Designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other applications, including a full NDA to market the same product for the same indication for seven years, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity or where the manufacturer is unable to assure sufficient product quantity. However, Orphan Drug Designation neither shortens the development time or regulatory review time of a drug nor gives the drug any advantage in the regulatory review or approval process. In 2022, the Eleventh Circuit’s decision in Catalyst Pharmaceuticals, Inc. v. FDA challenges FDA’s long-standing interpretation and provides that the orphan drug exclusivity should be applied to block FDA approval of the same drug for the “same disease or condition” instead of the approved indication during the exclusivity period. If the Catalyst decision is applied beyond the facts of that case, FDA may revoke approvals or the grant of subsequent orphan exclusivity periods for the same drugs approved for different indications within the same orphan-designated disease or condition. Catalyst has created some uncertainty with respect to the scope of the orphan drug exclusivity and may increase legal challenges in the field. FDA may work with the Congress to amend the orphan drug provisions in the law to provide more clarity to stakeholders. The extent of the impact of the Catalyst decision on the industry and on FDA’s regulation and policies with respect to orphan exclusivity as well as the impact of any future legislation on orphan drug approval and exclusivity is unclear.
In Catalyst Pharms., Inc. v. Becerra, 14 F.4th 1299 (11th Cir. 2021), the court disagreed with the FDA’s longstanding position that the orphan drug exclusivity only applies to the approved use or indication within an eligible disease. This decision created uncertainty in the application of the orphan drug exclusivity. On January 24, 2023, the FDA published a notice in the Federal Register to clarify that while the agency complies with the court’s order in Catalyst, FDA intends to continue to apply its longstanding interpretation of the regulations to matters outside of the scope of the Catalyst order – that is, the agency will continue tying the scope of orphan-drug exclusivity to the uses or indications for which a drug is approved, which permits other sponsors to obtain approval of a drug for new uses or indications within the same orphan designated disease or condition that have not yet been approved. It is unclear how future litigation, legislation, agency decisions, and administrative actions will impact the scope of the orphan drug exclusivity.
The FDA and EMA granted Orphan Drug Designation to CM-101 in its primary indications of PSC, SSc and idiopathic pulmonary fibrosis, or IPF. Chemomab may seek Orphan Drug Designations for CM-101 in other indications or for other product candidates. There can be no assurance that Chemomab will be able to obtain such designations.
Even if Chemomab obtains Orphan Drug Designation for any product candidate in specific indications, it may not be the first to obtain marketing approval of such product candidate for the orphan-designated indication due to the uncertainties associated with developing pharmaceutical products. In addition, exclusive marketing rights in the United States may be limited if Chemomab seeks approval for an indication broader than the orphan-designated indication or may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition.
Further, even if Chemomab obtains orphan drug exclusivity in the United States for a product, that exclusivity may not effectively protect the product from competition because different drugs with different active moieties can be approved for the same condition. Even after an orphan product is approved, the FDA can subsequently approve the same drug with the same active moiety for the same condition if the FDA concludes that the later drug is safer, more effective or makes a major contribution to patient care.
Chemomab will need to expand its organization, and it may experience difficulties in managing this growth, which could disrupt its operations.
As of December 31, 2022, the Company had 37 employees / full time consultants. The Company expect to experience significant growth in the number of its employees and the scope of its operations, particularly in the areas of product candidate development, regulatory affairs and sales and marketing. Chemomab may have difficulty identifying, hiring and integrating new personnel. Future growth would impose significant additional responsibilities on its management, including the need to identify, recruit, maintain, motivate and integrate additional employees, consultants and contractors. Also, Chemomab’s management may need to divert a disproportionate amount of its attention away from its day-to-day activities and devote a substantial amount of time to managing these growth activities. Chemomab may not be able to effectively manage the expansion of its operations, which may result in weaknesses in its infrastructure, give rise to operational mistakes, loss of business opportunities, loss of employees and reduced productivity among remaining employees. Chemomab’s expected growth could require significant capital expenditures and may divert financial resources from other projects, such as the development of product candidates. If Chemomab’s management is unable to effectively manage its growth, its expenses may increase more than expected, its ability to generate and/or grow revenues could be reduced, and it may not be able to implement its business strategy. Chemomab’s future financial performance and its ability to commercialize its product candidates and compete effectively will depend, in part, on its ability to effectively manage any future growth.
Many of the biopharmaceutical companies that Chemomab competes against for qualified personnel and consultants have greater financial and other resources, different risk profiles and a longer history in the industry than Chemomab does. If Chemomab is unable to continue to attract and retain high-quality personnel and consultants, the rate and success at which it can discover and develop product candidates and operate its business will be limited.
Chemomab has incurred significant operating losses since its inception and anticipates it will incur continued losses for the foreseeable future.
The Company has funded its operations to date through proceeds from sales of its equity and grants from the Israel Innovation Authority, or the IIA, which as of December 31, 2022, resulted in gross proceeds of approximately $96 million. As of December 31, 2022, Chemomab’s cash, cash equivalents and deposits were approximately $40million. Chemomab has incurred net losses in each year since its inception, and it has an accumulated deficit of $63.8 million as of December 31, 2022. Chemomab expects its existing cash, cash equivalents and bank deposits will allow it to fund its operating expenses and capital expenditure requirements at least through March 31, 2024.
Substantially all of Chemomab’s operating losses have resulted from general and administrative costs associated with its operations, and costs associated with its research and development programs, including for its preclinical and clinical product candidates. Chemomab expects to incur increasing levels of operating losses over the next several years and for the foreseeable future. Chemomab’s prior losses, combined with expected future losses, have had and will continue to have an adverse effect on its shareholders’ deficit and working capital. In any particular quarter or quarters, Chemomab’s operating results could be below the expectations of securities analysts or investors, which could cause the price of Chemomab’s ADSs to decline.
Chemomab expects its research and development expenses to significantly increase in connection with its clinical studies of its product candidates. In addition, if Chemomab obtains marketing approval for its product candidates, it will incur significant sales and marketing, legal, and outsourced-manufacturing expenses. As a public company, Chemomab expects to continue to incur significant and increasing operating losses for the foreseeable future. Because of the numerous risks and uncertainties associated with developing pharmaceutical products, Chemomab is also unable to predict the extent of any future losses or when it will become profitable, if at all. Even if Chemomab does become profitable, it may not be able to sustain or increase its profitability on a quarterly or annual basis.
A possible resurgent COVID-19 pandemic or the future outbreak of other highly infectious or contagious diseases could seriously harm Chemomab’s research, development and potential future commercialization efforts, increase its costs and expenses and have a material adverse effect on its business, financial condition and results of operations.
Broad-based business or economic disruptions have, and could continue to, adversely affect Chemomab’s ongoing or planned research and development activities. For example, over the past few years the COVID-19 pandemic has caused significant disruptions to the Israeli, United States, European and global economy and has contributed to significant volatility and negative pressure in financial markets and created many barriers to the successful conduct of clinical trials.
Chemomab cannot presently predict the scope and severity of any potential business shutdowns or disruptions from crises related to either disease outbreaks or possible political or social turmoil,. If Chemomab or any of the third parties with whom it engages, however, were to experience shutdowns or other business disruptions, Chemomab’s ability to conduct its business in the manner and on the timelines presently planned could be materially and negatively affected, which could have a material adverse impact on its business and its results of operation and financial condition.
Risks Related to Chemomab’s Intellectual Property Rights
If Chemomab is unable to protect its patents or other proprietary rights, or if Chemomab infringes the patents or other proprietary rights of others, its competitiveness and business prospects may be materially damaged.
Patent and other proprietary rights are essential to Chemomab’s business. Chemomab’s success depends to a significant degree on its ability to obtain and enforce patents and licenses to patent rights, both in the United States and in other countries. Chemomab cannot guarantee that pending patent applications will result in issued patents, that patents issued or licensed will not be challenged or circumvented by competitors, that the patents and other intellectual property rights of Chemomab and its business partners will not be found to be invalid or that the intellectual property rights of others will not prevent Chemomab from selling its products or from executing on its strategies.
The patent position of a biopharmaceutical company is often uncertain and involves complex legal and factual questions. Significant litigation concerning patents and products is pervasive in Chemomab’s industry. Patent claims include challenges to the coverage and validity of Chemomab’s patents on products or processes as well as allegations that its products infringe patents held by competitors or other third parties. A loss in any of these types of cases could result in a loss of patent protection or the ability to market products, which could lead to a significant loss of sales, or otherwise materially affect future results of operations. Chemomab also relies on trademarks, copyrights, trade secrets and know-how to develop, maintain and strengthen its competitive positions. Third parties may know, discover or independently develop equivalent proprietary information or techniques, or they may gain access to Chemomab’s trade secrets or disclose such trade secrets to the public.
Although Chemomab’s employees, consultants, parties to collaboration agreements and other business partners are generally subject to confidentiality or similar agreements to protect its confidential and proprietary information, these agreements may be breached, and Chemomab may not have adequate remedies for any breach. In addition, Chemomab’s trade secrets may otherwise become known or be independently discovered by competitors. To the extent that Chemomab’s employees, consultants, parties to collaboration agreements and other business partners use intellectual property owned by others in their work for the company, disputes may arise as to the rights in related or resulting know-how and inventions.
Furthermore, Chemomab’s intellectual property, other proprietary technology and other sensitive company data is potentially vulnerable to loss, damage or misappropriation from system malfunction, computer viruses, unauthorized access to data or misappropriation or misuse thereof by those with permitted access and other events. While Chemomab has invested to protect its intellectual property and other data, and continue to work diligently in this area, there can be no assurance that its precautionary measures will prevent breakdowns, breaches, cyber incidents or other events. Such events could have a material adverse effect on Chemomab’s reputation, business, financial condition or results of operations. Misappropriation or other loss of Chemomab’s intellectual property from any of the foregoing could have a material adverse effect on its competitive position and may cause it to incur substantial litigation costs.
Chemomab may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent, which might adversely affect its ability to develop, manufacture and market its product candidates.
From time to time Chemomab may identify patents or applications in the same general area as its products and product candidates. Chemomab may determine these third-party patents are irrelevant to its business based on various factors, including its interpretation of the scope of the patent claims and its interpretation of when the patent expires. If the patents are asserted against Chemomab, however, a court may disagree with its determinations. Further, while Chemomab may determine that the scope of claims that will issue from a patent application does not present a risk, it is difficult to accurately predict the scope of claims that will issue from a patent application, its determination may be incorrect, and the issuing patent may be asserted against Chemomab. Chemomab cannot guarantee that it will be able to successfully settle or otherwise resolve such infringement claims. If Chemomab fails in any such dispute, in addition to being forced to pay monetary damages, it may be temporarily or permanently prohibited from commercializing its product candidates or be required to obtain a license under such patent, which may not be available on reasonable terms or at all. Chemomab might, if possible, also be forced to redesign its product candidates so that it no longer infringes, misappropriates or otherwise violates the third-party intellectual property rights. Any of these events, even if Chemomab were ultimately to prevail, could require it to divert substantial financial and management resources that it would otherwise be able to devote to its business. Any of the foregoing could have a material adverse effect on Chemomab’s business, financial condition, results of operations, and prospects.
Changes in patent laws or patent jurisprudence could diminish the value of patents in general, thereby impairing Chemomab’s ability to protect its product candidates.
As is the case with other biopharmaceutical and pharmaceutical companies, Chemomab’s success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biopharmaceutical and pharmaceutical industries involve both technological and legal complexity. Therefore, obtaining and enforcing biopharmaceutical and pharmaceutical patents is costly, time-consuming and inherently uncertain. In addition, the Leahy-Smith America Invents Act, or the AIA, which was passed in September 2011, resulted in significant changes to the United States patent system.
An important change introduced by the AIA is that, as of March 16, 2013, the United States transitioned from a “first-to-invent” to a “first-to-file” system for deciding which party should be granted a patent when two or more patent applications are filed by different parties claiming the same invention. Under a “first-to-file” system, assuming the other requirements for patentability are met, the first inventor to file a patent application generally will be entitled to a patent on the invention regardless of whether another inventor had made the invention earlier. A third party that files a patent application in the USPTO after that date but before Chemomab could therefore be awarded a patent covering an invention of ours even if it made the invention before it was made by the third party. This will require Chemomab to be cognizant of the time from invention to filing of a patent application and be diligent in filing patent applications, but circumstances could prevent it from promptly filing patent applications on its inventions.
Among some of the other changes introduced by the AIA are changes that limit where a patentee may file a patent infringement suit and providing opportunities for third parties to challenge any issued patent with the USPTO. This applies to all of Chemomab’s United States patents, even those issued before March 16, 2013. Because of a lower evidentiary standard in USPTO proceedings compared to the evidentiary standard in United States federal courts necessary to invalidate a patent claim, a third party could potentially provide evidence in a USPTO proceeding sufficient for the USPTO to hold a claim invalid even though the same evidence would be insufficient to invalidate the claim if first presented in a district court action.
Accordingly, a third party may attempt to use the USPTO procedures to invalidate Chemomab’s patent claims that would not have been invalidated if first challenged by the third party as a defendant in a district court action. It is not clear what, if any, impact the AIA will have on the operation of its business. However, the AIA and its implementation could increase the uncertainties and costs surrounding the prosecution of Chemomab or its licensors’ patent applications and the enforcement or defense of Chemomab or its licensors’ issued patents.
Chemomab may become involved in opposition, interference, derivation, inter partes review, post-grant review, reexamination or other proceedings challenging Chemomab or its licensors’ patent rights, and the outcome of any proceedings are highly uncertain. An adverse determination in any such proceeding could reduce the scope of, or invalidate, Chemomab’s owned or in-licensed patent rights, in whole or in part, allow third parties to commercialize its technology or products and compete directly with Chemomab, without payment to it, or result in Chemomab’s inability to manufacture or commercialize products without infringing third-party patent rights.
Additionally, the United States Supreme Court has ruled on several patent cases in recent years either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing uncertainty with regard to Chemomab’s ability to obtain patents in the future, this combination of events has created uncertainty with respect to the validity, enforceability and value of patents, once obtained. Depending on decisions by Congress, the federal courts and the USPTO, as well as similar bodies in other jurisdictions, the laws and regulations governing patents could change in unpredictable ways that could weaken Chemomab’s ability to obtain new patents or to enforce its existing patents and patents that it might obtain in the future. Similarly, the complexity and uncertainty of European patent laws have also increased in recent years. In addition, the European patent system is relatively stringent in the type of amendments that are allowed during prosecution. Complying with these laws and regulations could limit Chemomab’s ability to obtain new patents in the future that may be important for its business, and these laws and regulations patents could continue to change in unpredictable ways that could have a material adverse effect on Chemomab’s existing patent rights and its ability to protect and enforce its intellectual property in the future.
Obtaining and maintaining Chemomab’s patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and Chemomab’s patent protection could be reduced or eliminated for non-compliance with these requirements.
The USPTO and European and other patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. In addition, periodic maintenance, renewal and annuity fees on any issued patent are due to be paid to the USPTO and European and other patent agencies over the lifetime of a patent. While an inadvertent failure to make payment of such fees or to comply with such provisions can in many cases be cured by additional payment of a late fee or by other means in accordance with the applicable rules, there are situations in which such noncompliance will result in the abandonment or lapse of the patent or patent application, and the partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents within prescribed time limits. If Chemomab or its licensors fails to maintain the patents and patent applications covering its product candidates or if Chemomab or its licensors otherwise allow its patents or patent applications to be abandoned or lapse, its competitors might be able to enter the market, which would hurt Chemomab’s competitive position and could impair its ability to successfully commercialize its product candidates in any indication for which they are approved, which could have a material adverse effect on Chemomab’s business, financial condition, results of operations, and prospects.
Risks Related to Chemomab’s Regulatory Approvals
The regulatory approval processes of the FDA and comparable foreign authorities are lengthy, time consuming and inherently unpredictable, and if Chemomab is ultimately unable to obtain regulatory approval for CM-101 or any other product candidates, its business will be substantially harmed.
The time required to obtain approval by the FDA and comparable foreign authorities is unpredictable but typically takes many years following the commencement of clinical studies and depends upon numerous factors, including the substantial discretion of the regulatory authorities. In addition, approval policies, regulations or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s clinical development and may vary among jurisdictions, which may cause delays in the approval or the decision not to approve an application. Regulatory authorities have substantial discretion in the approval process and may refuse to accept any application or may decide that Chemomab’s data is insufficient for approval and require additional preclinical, clinical or other data. Even if Chemomab eventually completes clinical testing and receives approval of any regulatory filing for its product candidates, the FDA and other comparable foreign regulatory authorities may approve Chemomab’s product candidates for a more limited indication or a narrower patient population than it originally requested. Chemomab has not obtained regulatory approval for any product candidate and it is possible that it will never obtain regulatory approval for CM-101 or any other product candidate. Chemomab is not permitted to market any of its product candidates in the United States until it receives regulatory approval of an NDA from the FDA.
Prior to obtaining approval to commercialize a product candidate in the United States or abroad, Chemomab must demonstrate with substantial evidence from well-controlled clinical studies, and to the satisfaction of the FDA or foreign regulatory agencies, that such product candidate is safe and effective for its intended use. Results from preclinical studies and clinical trials can be interpreted in different ways. Even if Chemomab believes the preclinical or clinical data for its product candidates are promising, such data may not be sufficient to support approval by the FDA and other regulatory authorities.
The FDA or any foreign regulatory bodies can delay, limit or deny approval of Chemomab’s product candidates or require it to conduct additional preclinical or clinical testing or abandon a program for many reasons, including:
| | • | the FDA or comparable foreign regulatory authorities may disagree with the design or implementation of Chemomab’s clinical studies; |
| | • | Chemomab may be unable to demonstrate to the satisfaction of the FDA or comparable foreign regulatory authorities that a product candidate is safe and effective for its proposed indication; |
| | • | serious and unexpected drug-related side effects experienced by participants in Chemomab’s clinical studies or by individuals using drugs similar to its product candidates, or other products containing the active ingredient in Chemomab’s product candidates; |
| | • | negative or ambiguous results from Chemomab’s clinical studies or results that may not meet the level of statistical significance required by the FDA or comparable foreign regulatory authorities for approval;
|
| | • | the population studied in the clinical study may not be sufficiently broad or representative to assure efficacy and safety in the full population for which Chemomab seeks approval; |
| | • | Chemomab may be unable to demonstrate that a product candidate’s clinical and other benefits outweigh its safety risks; |
| | • | the FDA or comparable foreign regulatory authorities may disagree with Chemomab’s interpretation of data from preclinical studies or clinical trials; |
| | • | the data collected from clinical studies of Chemomab’s product candidates may not be acceptable or sufficient to support the submission of an NDA or other submission or to obtain regulatory approval in the United States or elsewhere, and Chemomab may be required to conduct additional clinical studies; |
| | • | the FDA’s or the applicable foreign regulatory agency’s disagreement regarding the formulation, labeling and/or the specifications of Chemomab’s product candidates; |
| | • | the FDA or comparable foreign regulatory authorities may fail to approve or find deficiencies with the manufacturing processes or facilities of third-party manufacturers with which Chemomab contracts for clinical and commercial supplies; and |
| | • | the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering Chemomab’s clinical data insufficient for approval. |
Moreover, preclinical and clinical data are often susceptible to varying interpretations and analyses and many companies that believed their product candidates performed satisfactorily in preclinical studies and clinical trials nonetheless failed to obtain FDA or comparable foreign regulatory authority approval. Chemomab cannot guarantee that the FDA or foreign regulatory authorities will interpret trial results as it does, and more trials could be required before Chemomab is able to submit applications seeking approval of its product candidates. To the extent that the results of the trials are not satisfactory to the FDA or foreign regulatory authorities for support of a marketing application, Chemomab may be required to expend significant resources, which may not be available to it, to conduct additional trials in support of potential approval of Chemomab’s product candidates. Furthermore, the approval policies or regulations of the FDA or comparable foreign regulatory authorities may significantly change in a manner rendering Chemomab’s clinical data insufficient for approval, which may lead to the FDA or comparable foreign regulatory authorities delaying, limiting or denying approval of its product candidates.
Of the large number of drugs in development, only a small percentage successfully complete the regulatory approval processes and are commercialized. This lengthy approval process, as well as the unpredictability of future clinical trial results, may result in Chemomab failing to obtain regulatory approval to market CM-101 or any other product candidate, which would significantly harm Chemomab’s business, results of operations and prospects.
In addition, the FDA or the applicable foreign regulatory agency also may approve a product candidate for a more limited indication or patient population than Chemomab originally requested, and the FDA or applicable foreign regulatory agency may approve a product candidate with a REMS or a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product candidate. Regulatory authorities may also grant approval contingent on the performance of costly post-marketing clinical trials. Any of the foregoing scenarios could materially harm the commercial prospects for Chemomab’s product candidates.
Obtaining and maintaining regulatory approval of Chemomab’s product candidates in one jurisdiction does not mean that it will be successful in obtaining regulatory approval of its product candidates in other jurisdictions.
In order to market any product outside of the United States, Chemomab must establish and comply with the numerous and varying safety, efficacy, and other regulatory requirements of other countries. Obtaining and maintaining regulatory approval of its product candidates in one jurisdiction does not guarantee that Chemomab will be able to obtain or maintain regulatory approval in any other jurisdiction, but a failure or delay in obtaining regulatory approval in one jurisdiction may have a negative effect on the regulatory approval process in others. Chemomab’s product candidates may not receive marketing approval even if they achieve their primary endpoints in future Phase 3 clinical studies or registrational trials. The FDA or comparable foreign regulatory authorities may disagree with Chemomab’s trial designs and its interpretation of data from preclinical studies or clinical trials. In addition, any of these regulatory authorities may change requirements for the approval of a product candidate even after reviewing and providing comments or advice on a protocol for a pivotal Phase 3 or registrational clinical study. In addition, any of these regulatory authorities may also approve a product candidate for fewer or more limited indications than Chemomab’s request or may grant approval contingent on the performance of costly post-marketing clinical trials. The FDA or comparable foreign regulatory authorities may not approve the labeling claims that Chemomab believes would be necessary or desirable for the successful commercialization of its product candidates, if approved.
Furthermore, even if the FDA or other comparable foreign regulatory authority grants marketing approval of a product candidate, comparable regulatory authorities in foreign jurisdictions must also approve the manufacturing, marketing, and promotion of the product candidate in those countries. Approval procedures vary among jurisdictions and can involve requirements and administrative review periods different from those in the United States, including additional preclinical studies or clinical trials as clinical studies conducted in one jurisdiction may not be accepted by regulatory authorities in other jurisdictions. The marketing approval processes in other countries may implicate all of the risks detailed above regarding FDA approval in the United States, as well as other risks. In many jurisdictions outside the United States, a product candidate must be approved for reimbursement before it can be approved for sale in that jurisdiction. In some cases, the price that Chemomab intends to charge for its products is also subject to approval.
Obtaining foreign regulatory approvals and compliance with foreign regulatory requirements could result in significant delays, difficulties, and costs for Chemomab and could delay or prevent the introduction of its products in certain countries. Failure to obtain marketing approval in other countries or any delay or other setback in obtaining such approval would impair its ability to market its product candidates in such foreign markets. Any such impairment would reduce the size of its potential market, which could have a material adverse impact on its business, results of operations, and prospects.
Even if Chemomab obtains regulatory approval for CM-101 or any product candidate, it will still face extensive and ongoing regulatory requirements and obligations and any product candidates, if approved, may face future development and regulatory difficulties.
Any product candidate for which Chemomab obtains marketing approval, along with the manufacturing processes, post-approval clinical data, labeling, packaging, distribution, adverse event reporting, storage, recordkeeping, export, import, advertising and promotional activities for such product, among other things, will be subject to extensive and ongoing requirements of and review by the FDA and other regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, establishment registration and drug listing requirements, continued compliance with cGMP requirements relating to manufacturing, quality control, quality assurance and corresponding maintenance of records and documents, requirements regarding the distribution of samples to physicians and recordkeeping and GCP requirements for any clinical studies that Chemomab conducts post-approval.
Even if marketing approval of a product candidate is granted, the approval may be subject to limitations on the indicated uses for which the product candidate may be marketed or to the conditions of approval, including a requirement to implement a REMS. If any of Chemomab’s product candidates receives marketing approval, the accompanying label may limit the approved indicated use of the product candidate, which could limit sales of the product candidate. The FDA may also impose requirements for costly post-marketing studies or clinical trials and surveillance to monitor the safety or efficacy of a product. Violations of the Federal Food, Drug, and Cosmetic Act, or FDCA, relating to the promotion of prescription drugs may lead to FDA enforcement actions and investigations alleging violations of federal and state healthcare fraud and abuse laws, as well as state consumer protection laws.
In addition, later discovery of previously unknown adverse events or other problems with Chemomab’s products, manufacturers or manufacturing processes or failure to comply with regulatory requirements, may yield various results, including:
| | • | restrictions on manufacturing such products; |
| | • | restrictions on the labeling or marketing of products; |
| | • | restrictions on product manufacturing, distribution or use; |
| | • | requirements to conduct post-marketing studies or clinical trials; |
| | • | warning letters or untitled letters; |
| | • | withdrawal of the products from the market; |
| | • | refusal to approve pending applications or supplements to approved applications that Chemomab submits; |
| | • | fines, restitution or disgorgement of profits or revenues; |
| | • | suspension or withdrawal of marketing approvals; |
| | • | refusal to permit the import or export of Chemomab’s products; |
| | • | injunctions or the imposition of civil or criminal penalties. |
Further, the FDA’s policies may change, and additional government regulations may be enacted that could impose extensive and ongoing regulatory requirements and obligations on any product candidate for which Chemomab obtains marketing approval. If Chemomab is slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if Chemomab is not able to maintain regulatory compliance, it may lose any marketing approval that it may have obtained, which would adversely affect its business, prospects and ability to achieve or sustain profitability.
Disruptions at the FDA and other government agencies caused by funding shortages or global health concerns could hinder Chemomab’s ability to hire, retain or deploy key leadership and other personnel, or otherwise prevent new or modified products from being developed, approved or commercialized in a timely manner or at all, which could negatively impact its business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory, and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would harm Chemomab’s business. For example, over the last several years, including for 35 days beginning on December 22, 2018, the United States government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough critical FDA employees and stop critical activities. If a prolonged government shutdown occurs, it could significantly impact the ability of the FDA to timely review and process Chemomab’s regulatory submissions, which could harm its business.
The COVID-19 pandemic has also resulted in the FDA imposing preventive measures, including postponements of non-United States manufacturing and product inspections. If global health concerns continue to prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews, or other regulatory activities, it could significantly impact the ability of the FDA or other regulatory authorities to timely review and process Chemomab’s regulatory submissions, which could have a material adverse effect on its business.
Risks Related to Commercialization of Chemomab’s Product Candidates
If Chemomab does not achieve its projected development and commercialization goals in the timeframes it announces and expects, the commercialization of its product candidates may be delayed and Chemomab’s business will be harmed.
For planning purposes, Chemomab sometimes estimates the timing of the accomplishment of various scientific, clinical, regulatory and other product development objectives. These milestones may include Chemomab’s expectations regarding the commencement or completion of scientific studies and clinical trials, the regulatory submissions or commercialization objectives. From time to time, Chemomab may publicly announce the expected timing of some of these milestones, such as the completion of an ongoing clinical study, the initiation of other clinical studies, receipt of regulatory approval or the commercial launch of a product. The achievement of many of these milestones may be outside of Chemomab’s control. All of these milestones are based on a variety of assumptions which may cause the timing of achievement of the milestones to vary considerably from Chemomab’s estimates, including:
| | • | Chemomab’s available capital resources or capital constraints it experiences; |
| | • | the rate of progress, costs and results of Chemomab’s clinical studies and research and development activities, including the extent of scheduling conflicts with participating clinicians and collaborators; |
| | • | Chemomab’s ability to identify and enroll patients who meet clinical study eligibility criteria; |
| | • | Chemomab’s receipt of authorizations by the FDA and comparable foreign regulatory authorities, and the timing thereof; |
| | • | other actions, decisions or rules issued by regulators; |
| | • | Chemomab’s ability to access sufficient, reliable and affordable supplies of materials used in the manufacture of its product candidates; |
| | • | Chemomab’s ability to manufacture and supply clinical study materials to its clinical sites on a timely basis; |
| | • | the severity, duration and impact of the COVID-19 pandemic; |
| | • | the efforts of Chemomab’s collaborators with respect to the commercialization of its products, if any; and |
| | • | the securing of, costs related to, and timing issues associated with, commercial product manufacturing as well as sales and marketing activities. |
If Chemomab fails to achieve announced milestones in the timeframes it expects, the commercialization of any of its product candidates may be delayed, and its business, results of operations, financial condition and prospects may be adversely affected.
Chemomab faces substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than it.
The development and commercialization of new drug products is highly competitive. Chemomab may face competition with respect to any product candidates that it seeks to develop or commercialize in the future from major biopharmaceutical companies, specialty biopharmaceutical companies, and biotechnology companies worldwide. Potential competitors also include academic institutions, government agencies, and other public and private research organizations that conduct research, seek patent protection, and establish collaborative arrangements for research, development, manufacturing, and commercialization.
There are a number of large biopharmaceutical and biotechnology companies that are currently pursuing the development of products for the treatment of inflammation and fibrosis. Companies that Chemomab is aware of that are targeting the treatment of inflammation and fibrosis include large companies with significant financial resources such as Biogen, Inc., AbbVie Inc., Gilead Sciences, Inc., FibroGen, Inc., Galapagos NV, Bristol Myers Squibb Co., and Novartis AG. However, Chemomab does not know of any other companies currently in clinical development with an anti CCL24 mAb. For additional information regarding Chemomab’s competition, see “Chemomab Business - Competition.”
Many of Chemomab’s current or potential competitors, either alone or with their strategic partners, have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical studies, obtaining regulatory approvals, and marketing approved products than Chemomab does.
Even if CM-101 or any other product candidate Chemomab develops receives marketing approval, it may fail to achieve market acceptance by physicians, patients, third-party payors or others in the medical community necessary for commercial success.
If CM-101 or any other product candidate Chemomab develops receives marketing approval, it may nonetheless fail to gain sufficient market acceptance by physicians, patients, third-party payors and others in the medical community. If it does not achieve an adequate level of acceptance, Chemomab may not generate significant product revenues or become profitable. The degree of market acceptance of Chemomab’s product candidates, if approved, will depend on a number of factors, including but not limited to:
| | • | the efficacy and potential advantages compared to alternative treatments; |
| | • | effectiveness of sales and marketing efforts; |
| | • | the cost of treatment with respect to alternative treatments, including any similar generic treatments; |
| | • | Chemomab’s ability to offer its products for sale at competitive prices; |
| | • | the convenience and ease of administration compared to alternative treatments; |
| | • | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| | • | the strength of marketing and distribution support; |
| | • | the timing of market introduction of competitive products; |
| | • | the availability of third-party coverage and adequate reimbursement; |
| | • | product labeling or product insert requirements of the FDA, EMA or other regulatory authorities, including any limitations on warnings contained in a product’s approved labeling; |
| | • | the prevalence and severity of any side effects; and |
| | • | any restrictions on the use of Chemomab’ product together with other medications. |
Because Chemomab expects sales of its product candidates, if approved, to generate substantially all of its revenues for the foreseeable future, the failure of its product candidates to find market acceptance would harm its business and could require it to seek additional financing.
Chemomab relies completely on third-party suppliers to manufacture its clinical drug supplies for its product candidates, and Chemomab intends to rely on third parties to produce preclinical, clinical, and commercial supplies of any future product candidates.
Chemomab does not currently have, nor does Chemomab plan to acquire, the infrastructure or capability to internally manufacture its clinical drug supply of its product candidates, or any future product candidates, for use in the conduct of its preclinical studies and clinical trials.
Chemomab lacks the internal resources and the capabilities to manufacture any product candidates on a clinical or commercial scale. The facilities used by its contract manufacturers to manufacture the active pharmaceutical ingredient and final drug product must complete a pre-approval inspection by the FDA and other comparable foreign regulatory agencies to assess compliance with applicable requirements, including cGMPs, after Chemomab submits its NDA or relevant foreign regulatory market application to the applicable regulatory agency.
Chemomab is responsible for setting the product specifications and approving master batch records, but does not oversee the manufacturing process itself, and is completely dependent on its contract manufacturers to comply with cGMPs for manufacture of both active drug substances and finished drug products. If its contract manufacturers cannot successfully manufacture material that conforms to its specifications and the strict regulatory requirements of the FDA or applicable foreign regulatory agencies, they will not be able to pass a pre-approval inspection or secure and/or maintain regulatory approval for their manufacturing facilities. In addition, Chemomab has no direct control over its contract manufacturers’ ability to maintain adequate quality control, quality assurance, and qualified personnel. Furthermore, all of its contract manufacturers are engaged with other companies to supply and/or manufacture materials or products for such companies, which exposes its manufacturers to regulatory risks for the production of such materials and products. As a result, failure to satisfy the regulatory requirements for the production of those materials and products may affect the regulatory clearance of its contract manufacturers’ facilities generally. If the FDA or an applicable foreign regulatory agency determines now or in the future that these facilities for the manufacture of its product candidates are noncompliant, Chemomab may need to find alternative manufacturing facilities, which would adversely impact its ability to develop, obtain regulatory approval for or market its product candidates. Its reliance on contract manufacturers also exposes Chemomab to the possibility that they, or third parties with access to their facilities, will have access to and may compromise its trade secrets or other proprietary information.
If Chemomab is unable to establish sales, marketing and distribution capabilities either on its own or in collaboration with third parties, it may not be successful in commercializing CM-101, if approved.
Chemomab does not have any infrastructure for the sales, marketing or distribution of CM-101, and the cost of establishing and maintaining such an organization may exceed the cost-effectiveness of doing so. In order to market and successfully commercialize CM-101 or any other product candidate Chemomab develops, if approved, it must build its sales, distribution, marketing, managerial and other non-technical capabilities or make arrangements with third parties to perform these services. Chemomab expects to build a focused sales, distribution and marketing infrastructure to market CM-101, if approved. There are significant expenses and risks involved with establishing Chemomab’s own sales, marketing and distribution capabilities, including its ability to hire, retain and appropriately incentivize qualified individuals, generate sufficient sales leads, provide adequate training to sales and marketing personnel and effectively manage a geographically dispersed sales and marketing team. Any failure or delay in the development of Chemomab’s internal sales, marketing and distribution capabilities could delay any product launch, which would adversely impact the commercialization of that product. Additionally, if the commercial launch of CM-101 for which Chemomab recruits a sales force and establishes marketing capabilities is delayed or does not occur for any reason, it would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and Chemomab’s investment would be lost if it cannot retain or reposition its sales and marketing personnel.
Factors that may inhibit Chemomab’s efforts to commercialize its product candidates on its own include:
| | • | Chemomab’s inability to recruit, train and retain adequate numbers of effective sales and marketing personnel; |
| | • | the inability of sales personnel to obtain access to physicians or attain adequate numbers of physicians to prescribe Chemomab’s products; and |
| | • | unforeseen costs and expenses associated with creating an independent sales and marketing organization. |
Chemomab does not anticipate having the resources in the foreseeable future to allocate to the sales and marketing of its product candidates, if approved, in certain markets overseas. Therefore, Chemomab’s future success will depend, in part, on its ability to enter into and maintain collaborative relationships for such capabilities, the collaborator’s strategic interest in a product and such collaborator’s ability to successfully market and sell the product. Chemomab intends to pursue collaborative arrangements regarding the sale and marketing of CM-101, if approved, for certain markets overseas; however, Chemomab cannot guarantee that it will be able to establish or maintain such collaborative arrangements, or if able to do so, that it will have effective sales forces. To the extent that Chemomab depends on third parties for marketing and distribution, any revenues it receives will depend upon the efforts of such third parties, and there can be no assurance that such efforts will be successful.
If Chemomab is unable to build its own sales force or negotiate a collaborative relationship for the commercialization of CM-101, Chemomab may be forced to delay the potential commercialization of CM-101 or reduce the scope of its sales or marketing activities for CM-101. If Chemomab needs to increase its expenditures to fund commercialization activities for CM-101, it will need to obtain additional capital, which may not be available to it on acceptable terms, or at all. Chemomab may also have to enter into collaborative arrangements for CM-101 at an earlier stage than otherwise would be ideal and it may be required to relinquish rights to CM-101 or otherwise agree to terms unfavorable to it. Any of these occurrences may have an adverse effect on Chemomab’s business, operating results and prospects.
If Chemomab is unable to establish adequate sales, marketing and distribution capabilities, either on its own or in collaboration with third parties, it will not be successful in commercializing its product candidates and may never become profitable. Chemomab will be competing with many companies that currently have extensive and well-funded marketing and sales operations. Without an internal team or the support of a third party to perform marketing and sales functions, it may be unable to compete successfully against these more established companies.
A variety of risks associated with operating internationally could materially adversely affect Chemomab’s business.
Chemomab’s research and development facilities and certain of its executives are located in Israel and certain of its product candidates may be manufactured at third-party facilities located in Europe. In addition, Chemomab’s business strategy includes potentially expanding internationally if any of its product candidates receives regulatory approval. Doing business internationally involves a number of risks, including but not limited to:
| | • | multiple, conflicting and changing laws and regulations, such as privacy regulations, tax laws, export and import restrictions, employment laws, regulatory requirements and other governmental approvals, permits and licenses; |
| | • | failure by Chemomab to obtain and maintain regulatory approvals for the use of its products in various countries; |
| | • | additional potentially relevant third-party patent rights; |
| | • | complexities and difficulties in obtaining protection and enforcing Chemomab’s intellectual property; |
| | • | difficulties in staffing and managing foreign operations; |
| | • | complexities associated with managing multiple payor reimbursement regimes, government payors or patient self-pay systems; |
| | • | limits in Chemomab’s ability to penetrate international markets; |
| | • | financial risks, such as longer payment cycles, difficulty collecting accounts receivable, the impact of local and regional financial crises on demand and payment for Chemomab’s products and exposure to foreign currency exchange rate fluctuations; |
| | • | natural disasters, political and economic instability, including wars, terrorism and political unrest, outbreak of disease, boycotts, curtailment of trade and other business restrictions; |
| | • | certain expenses including, among others, expenses for travel, translation and insurance; and |
| | • | regulatory and compliance risks that relate to maintaining accurate information and control over sales and activities that may fall within the purview of the United States Foreign Corrupt Practices Act, its books and records provisions, or its anti-bribery provisions. |
Any of these factors could significantly harm Chemomab’s international expansion and operations and, consequently, its results of operations.
Risks Related to Chemomab’s Incorporation and Location in Israel
Conditions in Israel could materially and adversely affect Chemomab’s business.
Many of Chemomab’s employees, including certain management members operate from its offices that are located in Tel Aviv, Israel. In addition, a number of Chemomab’s officers and directors are residents of Israel. Accordingly, political, economic, and military conditions in Israel and the surrounding region may directly affect its business and operations. In recent years, Israel has been engaged in sporadic armed conflicts with Hamas, an Islamist terrorist group that controls the Gaza Strip, with Hezbollah, an Islamist terrorist group that controls large portions of southern Lebanon, and with Iranian-backed military forces in Syria. In addition, Iran has threatened to attack Israel and may be developing nuclear weapons. Some of these hostilities were accompanied by missiles being fired from the Gaza Strip against civilian targets in various parts of Israel, including areas in which Chemomab’s employees and some of its consultants are located, and negatively affected business conditions in Israel. Any hostilities involving Israel or the interruption or curtailment of trade between Israel and its trading partners could adversely affect Chemomab’s operations and results of operations.
Chemomab’s commercial insurance does not cover losses that may occur as a result of events associated with war and terrorism. Although the Israeli government currently covers the reinstatement value of direct damages that are caused by terrorist attacks or acts of war, Chemomab cannot guarantee that this government coverage will be maintained or that it will sufficiently cover its potential damages. Any losses or damages incurred by Chemomab could have a material adverse effect on its business. Any armed conflicts or political instability in the region would likely negatively affect business conditions and could harm Chemomab’s results of operations.
Further, in the past, the State of Israel and Israeli companies have been subjected to economic boycotts. Several countries still restrict business with the State of Israel and with Israeli companies. These restrictive laws and policies may have an adverse impact on Chemomab’s operating results, financial condition or the expansion of its business. A campaign of boycotts, divestment and sanctions has been undertaken against Israel, which could also adversely impact Chemomab’s business. The recent installation of a new government in Israel that includes far right wing parties and personnel may lead to greater international sanctions or other actions that are detrimental to international business for Israeli companies.
In addition, many Israeli citizens are obligated to perform several days, and in some cases more, of annual military reserve duty each year until they reach the age of 40 (or older, for reservists who are military officers or who have certain occupations) and, in the event of a military conflict, may be called to active duty. In response to increases in terrorist activity, there have been periods of significant call-ups of military reservists. It is possible that there will be military reserve duty call-ups in the future. Chemomab’s operations could be disrupted by such call-ups, which may include the call-up of members of Chemomab’s management. Such disruption could materially adversely affect Chemomab’s business, prospects, financial condition and results of operations.
Furthermore, the Israeli government is currently pursuing extensive changes to Israel’s judicial system. In response to the foregoing developments, individuals, organizations and institutions, both within and outside of Israel, have voiced concerns that the proposed changes may negatively impact the business environment in Israel including due to reluctance of foreign investors to invest or conduct business in Israel, as well as to increased currency fluctuations, downgrades in credit rating, increased interest rates, increased volatility in securities markets, and other changes in macroeconomic conditions. Such proposed changes may also adversely affect the labor market in Israel or lead to political instability or civil unrest. To the extent that any of these negative developments do occur, they may have an adverse effect on our business, our results of operations and our ability to raise additional funds, if deemed necessary by our management and board of directors.
Because a certain portion of Chemomab’s expenses are incurred in currencies other than the U.S. Dollar, its results of operations may be harmed by currency fluctuations and inflation.
Chemomab’s reporting and functional currency is the United States Dollar, but some portion of its clinical studies and operations expenses are in NIS. As a result, Chemomab is exposed to some currency fluctuation risks. Fluctuation in the exchange rates of foreign currency has an influence on the cost of goods sold and Chemomab’s financing revenues and expenses. Chemomab may, in the future, decide to enter into currency hedging transactions to decrease the risk of financial exposure from fluctuations in the exchange rate of the currencies mentioned above with respect to the U.S. Dollar. These measures, however, may not adequately protect Chemomab from adverse effects.
Chemomab received Israeli government grants for certain of their research and development activities as detailed below. The terms of those grants require us to satisfy specified conditions in order to transfer outside of Israel the manufacture of products based on know-how funded by the Israel Innovation Authority or to transfer outside of Israel the know-how itself. If we fail to comply with the requirements of Israeli law in this regard, we may be required to pay penalties, and it may impair our ability to sell our technology outside of Israel.
Some of Chemomab’s research and development efforts were financed through grants that were received from the Israel Innovation Authority of the Israeli Ministry of Economy and Industry, or the IIA (formerly known as the Office of the Chief Scientist). When know-how is developed using IIA grants, the Encouragement of Research, Development and Technological Innovation in Industry Law 5744-1984, or the Innovation Law, and the regulations thereunder, restrict our ability to transfer outside of Israel either the manufacture of products based on IIA-funded know-how or the know-how itself. Such restrictions continue to apply even after financial obligations to the IIA are paid in full. The consideration available to our shareholders in a future transaction involving the transfer outside of Israel of know-how developed with IIA funding (such as a merger or similar transaction) may be reduced by any amounts that we are required to pay to the IIA.
Risks Related to our ADSs
We will need to raise additional capital to fund our operations, which may be unavailable to us on acceptable terms or at all, or may cause dilution or place significant restrictions on our ability to operate our business.
If our available cash resources are insufficient to satisfy our liquidity requirements, we will be required to raise additional capital through issuances of equity or convertible debt securities, or seek debt financing or other form of third-party funding.
If we are unable to obtain adequate financing or financing on terms satisfactory to us when needed, our ability to continue to pursue our business objectives and to respond to business opportunities, challenges, or unforeseen circumstances could be significantly limited, and could have a material adverse effect on our business, financial condition, results of operations and prospects.
The various ways we could raise additional capital carry potential risks. If we raise funds by issuing equity securities, dilution to our shareholders would result. If we raise funds by issuing debt securities, those debt securities would have rights, preferences and privileges senior to those of holders of our ADSs. The terms of debt securities issued or borrowings pursuant to a credit agreement could impose significant restrictions on our operations. If we raise funds through collaborations or licensing arrangements, we might be required to relinquish significant rights to our product candidates or grant licenses on terms that are not favorable to us.
The trading price of the ADSs has been highly volatile, and is expected to continue to be volatile.
The trading price of the ADSs has been highly volatile, particularly over the last year. For example, on January 11, 2022, the closing price of the ADSs was $6.98 per ADS and on March 13, 2023, it was $1.48 per ADS. This volatility may affect the price at which you are able to sell ADSs. Our ADS price is likely to continue to be volatile and subject to significant price and volume fluctuations in response to market and economic factors that are beyond our control. In addition, while the stock market in general has experienced high volatility, biotechnology companies in particular have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to operating performance. Broad market and industry factors may negatively affect the market price of the ADSs, regardless of our actual operating performance.
We have not paid dividends in the past and do not expect to pay dividends in the future, and, as a result, any return on investment may be limited to the value of the ADSs.
We have never paid dividends and do not anticipate paying dividends in the foreseeable future. The payment of dividends will depend on our earnings, capital requirements, financial condition, prospects and other factors our board of directors may deem relevant. If we do not pay dividends, the ADSs may be less valuable because a return on your investment will only occur if our ADS price appreciates and you sell your ADS thereafter. In addition, the Companies Law imposes restrictions on our ability to declare and pay dividends.
If we fail to continue to meet all applicable Nasdaq requirements, Nasdaq may delist the ADSs, which could have an adverse impact on the liquidity and market price of the ADSs.
The ADSs are currently listed on Nasdaq, which has qualitative and quantitative listing criteria. If we are unable to meet any of the Nasdaq listing requirements in the future, including, for example, if the closing bid price for the ADSs falls below $1.00 per share for 30 consecutive trading days, Nasdaq could determine to delist the ADSs, which could adversely affect the market liquidity of the ADSs and the market price of the ADSs could decrease. Such delisting could also adversely affect our ability to obtain financing for the continuation of our operations and could result in the loss of confidence by investors, customers and employees.
Holders of ADSs are not treated as holders of our ordinary shares.
Holders of ADSs are not treated as holders of our ordinary shares, unless they withdraw the ordinary shares underlying their ADSs in accordance with the deposit agreement and applicable laws and regulations. The depositary is the holder of the ordinary shares underlying the ADSs. Holders of ADSs therefore do not have any rights as holders of our ordinary shares, other than the rights that they have pursuant to the deposit agreement.
You may not have the same voting rights as the holders of our ordinary shares and may not receive voting materials in time to be able to exercise your right to vote.
Except as described in the deposit agreement, holders of the ADSs will not be able to exercise voting rights attaching to the ordinary shares represented by the ADSs. If we request the depositary to solicit your voting instructions (and we are not required to do so), the depositary will notify you of a shareholders’ meeting and send or make voting materials available to you. Those materials will describe the matters to be voted on and explain how ADS holders may instruct the depositary how to vote. We cannot guarantee that ADS holders will receive the voting materials in time to ensure that they can instruct the depositary to vote the ordinary shares underlying their ADSs. For instructions to be valid, they must reach the depositary by a date set by the depositary. The depositary will try, as far as practical, subject to the laws of Israel and the provisions of our articles of association or similar documents, to vote or to have its agents vote the ordinary shares or other deposited securities as instructed by ADS holders. If we do not request the depositary to solicit your voting instructions, you can still send voting instructions, and, in that case, the depositary may try to vote as you instruct, but it is not required to do so.
Otherwise, ADS holders will not be able to exercise their right to vote, unless they withdraw the ordinary shares underlying the ADSs they hold. However, ADS holders may not know about the meeting far enough in advance to withdraw those ordinary shares. In any event, the depositary will not exercise any discretion in voting deposited securities and it will only vote or attempt to vote as instructed. In addition, the depositary and its agents are not responsible for failing to carry out voting instructions or for the manner of carrying out voting instructions. This means that you may not be able to exercise your right to vote and there may be nothing you can do if your ordinary shares are not voted as you requested.
Holders of ADSs may be subject to limitations on the transfer of their ADSs and the withdrawal of the underlying ordinary shares.
ADSs are transferable on the books of the depositary. However, the depositary may refuse to deliver ADSs or register transfers of ADSs when the transfer books of the depositary or our transfer books are closed or at any time if the depositary or we think it advisable to do so. These limitations on transfer may have a material adverse effect on the value of the ADSs.
We are entitled to amend the deposit agreement and to change the rights of ADS holders under the terms of such agreement, or to terminate the deposit agreement, without the prior consent of the ADS holders.
We are entitled to amend the deposit agreement and to change the rights of the ADS holders under the terms of such agreement without the prior consent of the ADS holders. We and the depositary may agree to amend the deposit agreement in any way we decide is necessary or advantageous to us or to the depositary. Amendments may reflect, among other things, operational changes in the ADS program, legal developments affecting ADSs or changes in the terms of our business relationship with the depositary. If an amendment adds or increases fees or charges, except for taxes and other governmental charges or expenses of the depositary for registration fees, facsimile costs, delivery charges or similar items, or prejudices a substantial right of ADS holders, it will not become effective for outstanding ADSs until 30 days after the depositary notifies ADS holders of the amendment. At the time an amendment becomes effective, you are considered, by continuing to hold the ADSs, to agree to the amendment and to be bound by the ADRs and the deposit agreement as amended. If we make an amendment to the deposit agreement that is disadvantageous to ADS holders or terminate the deposit agreement, the ADS holders may choose to sell their ADSs or surrender their ADSs and become direct holders of the underlying ordinary shares, but they will have no right to any compensation whatsoever.
ADSs holders may not be entitled to a jury trial with respect to claims arising under the deposit agreement, which could result in less favorable outcomes to the plaintiff(s) in any such action.
The deposit agreement governing the ADSs representing our ordinary shares provides that, to the fullest extent permitted by law, holders and beneficial owners of ADSs irrevocably waive the right to a jury trial of any claim they may have against us or the depositary arising out of or relating to the ADSs or the deposit agreement.
If this jury trial waiver provision is not permitted by applicable law, an action could proceed under the terms of the deposit agreement with a jury trial. If we or the depositary opposed a jury trial demand based on the waiver, the court would determine whether the waiver was enforceable based on the facts and circumstances of that case in accordance with the applicable state and federal law. We believe that a contractual pre-dispute jury trial waiver provision is generally enforceable, including under the laws of the State of New York, which governs the deposit agreement. In determining whether to enforce a contractual pre-dispute jury trial waiver provision, courts will generally consider whether a party knowingly, intelligently and voluntarily waived the right to a jury trial. We believe that this is the case with respect to the deposit agreement and the ADSs.
If any holders or beneficial owners of ADSs bring a claim against us or the depositary in connection with matters arising under the deposit agreement or the ADSs, including claims under federal securities laws, such holder or beneficial owner may not be entitled to a jury trial with respect to such claims, which may have the effect of limiting and discouraging lawsuits against us or the depositary. If a lawsuit is brought against us or the depositary under the deposit agreement, it may be heard only by a judge or justice of the applicable trial court, which would be conducted according to different civil procedures and may result in different outcomes than a trial by jury would have had, including results that could be less favorable to the plaintiff(s) in any such action, depending on, among other things, the nature of the claims, the judge or justice hearing such claims, and the venue of the hearing.
No condition, stipulation or provision of the deposit agreement or ADSs serves as a waiver by any holder or beneficial owner of ADSs or by us or the depositary of compliance with any substantive provision of the U.S. federal securities laws and the rules and regulations promulgated thereunder.
We presently anticipate that we will be classified as a passive foreign investment company, which could result in adverse U.S. federal income tax consequences to U.S. Holders of our ordinary shares.
We would be classified as a passive foreign investment company, or PFIC, for any taxable year if, after the application of certain look-through rules, either: (i) 75% or more of our gross income for such year is “passive income” (as defined in the relevant provisions of the Internal Revenue Code of 1986, as amended, or the Code), or (ii) 50% or more of the value of our assets (generally determined on the basis of a quarterly average) during such year is attributable to assets that produce or are held for the production of passive income. Passive income generally includes, among other things, rents, dividends, interest, royalties, gains from the disposition of passive assets, and gains from commodities and securities transactions. For purposes of this test, we will be treated as owning a proportionate share of the assets and earning a proportionate share of the income of any other corporation of which we own, directly or indirectly, at least 25% (by value) of the stock. Based on the nature, composition and value of our income, operations and assets currently and in the future, we presently anticipate that we will be a PFIC for United States federal income tax purposes for the current taxable year and in the foreseeable future.
Item 1B. Unresolved Staff Comments
None.
Item 2. Properties
We lease a facility containing 3,961 square feet of laboratory and office space, which is located at Kiryat Atidim, Building 7, Tel Aviv, Israel 6158002. The lease expires in October 30, 2024 with an option to extend for an additional 36 months. We believe that our current facilities are sufficient to meet our current and near-term needs and that, should it be needed, suitable additional space will be available.
Item 3. Legal Proceedings
From time to time, we may be party to litigation or other legal proceedings that we consider to be a part of the ordinary course of our business. We are not currently involved in any legal proceedings that could reasonably be expected to have a material adverse effect on our business, prospects, financial condition or results of operations.
Item 4. Mine Safety Disclosures
Not applicable.
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market for our ADSs
The ADSs, representing our ordinary shares, have traded on Nasdaq under the symbol “CMMB” since the closing of the Merger on March 16, 2021. From February 12, 2019 until the Merger, the ADSs, representing our ordinary shares, traded on Nasdaq under the symbol “ANCN”. Prior to February 12, 2019, there was no public trading market for our ADSs.
As of March 15, 2023, there were seven holders of record of our ordinary shares.
Dividend Policy
We have never declared nor paid any cash dividends on our shares. We currently intend to retain any future earnings and do not expect to pay any dividends in the foreseeable future. Any future determination to declare cash dividends will be made at the discretion of our Board of Directors, subject to applicable laws, and will depend on a number of factors, including our financial condition, results of operations, capital requirements, contractual restrictions, general business conditions, and other factors that our Board of Directors may deem relevant.
Purchases of Equity Securities by the Issuer and Affiliated Purchasers
The following table summarizes the stock repurchase activity for the three months ended December 31, 2022:
| Period | | Total Number of ADSs Purchased | | | Average Price Paid per Share | | | Total Number of ADSs Purchased as Part of Publicly Announced Programs | | | Approximate Dollar Value of Shares that May Yet Be Purchased Under the Programs(1) | |
November 16, 2022 (1) | | | 582,023 | | | $ | 2.0848 | | | | 582,023 | | | $ | 1,218,000 | |
| Total | | | 582,023 | | | | | | | | 582,023 | | | | | |
(1) For additional information about the Repurchase Arrangement, see Item 13 – Certain Relationships and Related Transactions.
Item 6. [Reserved]
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes included elsewhere in this Annual Report on Form 10-K. Some of the information contained in this discussion and analysis, particularly with respect to our plans and strategy for our business and related financing, includes forward-looking statements that involve risks and uncertainties. You should read “Cautionary Statement Regarding Forward-Looking Statements” and Item 1A “Risk Factors” of this Annual Report on Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
We are a clinical-stage biotechnology company focused on the discovery and development of innovative therapeutics for fibrotic and inflammatory diseases with high unmet needs. Based on the unique and pivotal role of the soluble protein CCL24 in promoting fibrosis and inflammation, we developed CM-101, a monoclonal antibody designed to bind and block CCL24 activity. We believe CM-101 has demonstrated the potential to treat multiple severe and life-threatening fibrotic and inflammatory diseases.
We have pioneered the therapeutic targeting of CCL24, a chemokine that promotes various types of cellular processes that regulate inflammatory and fibrotic activities through the CCR3 receptor. The chemokine is expressed in various types of cells, including immune cells, endothelial cells and epithelial cells. We have developed a novel CCL24 inhibiting product candidate with dual anti-fibrotic and anti-inflammatory activity that modulates the complex interplay of both of these inflammatory and fibrotic mechanisms, which drive abnormal states of fibrosis and clinical fibrotic diseases. This innovative approach is being developed for difficult to treat rare diseases, also known as orphan indications or diseases, such as PSC and SSc, for which patients have no established disease-modifying treatment options. We estimate that there are approximately 77 thousand patients suffering from PSC in the U.S., EU and Japan, representing over a $1 billion market opportunity, and approximately 170 thousand patients suffering from SSc in those same markets, representing over a $1.5 billion market opportunity.
CM-101, our lead clinical product candidate, is a first-in-class humanized monoclonal antibody that attenuates the basic function of the soluble chemokine CCL24, also known as eotaxin-2, as a regulator of major inflammatory and fibrotic pathways. We have demonstrated that CM-101 interferes with the underlying biology of inflammation and fibrosis through a novel and differentiated mechanism of action. Based on these findings, we are actively advancing CM-101 in Phase 2 clinical studies directed toward two distinct clinical indications that include patients with liver or skin, and/or lung fibrosis. We are currently conducting a Phase 2 clinical study in PSC, a rare obstructive and cholestatic liver disease. The study is actively recruiting patients in the U.S., Europe and Israel and is being expanded by adding clinical sites, an additional high dose arm (20mg/kg) as well as an open label extension. We had earlier proposed to add both low and high dose arms to the study but the recent encouraging results reported from our Phase 2 liver fibrosis trial in NASH patients, dosed at 5mg/kg, along with the positive Phase 1b data we previously reported in NAFLD patients dosed at 5mg/kg and 2.5mg/kg, are seen as providing us sufficient data on the performance of the lower dose to drop it from the current trial, which is focusing on the 10mg/kg and 20g/kg doses. We believe this change will facilitate timely conduct and completion of the trial. If regulators in the future do not agree that the existing low dose data is sufficient, we always have the option to add a lower dose group as part of the Phase 3 clinical program.
We are also planning to open a Phase 2 clinical trial in SSc around midyear of 2023. The trial in SSc, a rare autoimmune rheumatic disease characterized by fibrosis in the skin and lung and other organs, will focus on establishing biological and clinical proof of concept in this patient population. Although our primary focus is on these two rare indications, as we noted, an additional Phase 2 clinical study in patients with liver fibrosis due to non-alcoholic steatohepatitis (NASH) has recently been completed. This trial provided safety and pharmacokinetic (PK) data and was informative in providing data that will help us determine whether we will advance the development of our current subcutaneous formulation of CM-101. Additionally, the trial measured a number of biomarkers that may be relevant to the potential activity of CM-101 in other fibro-inflammatory conditions. We recently reported results from this trial, which showed that the trial met its primary endpoint of safety and tolerability, and that CM-101 demonstrated encouraging activity in secondary endpoints that include a range of liver fibrosis biomarkers and physiologic assessments.
Components of Operating Results
Revenues
To date, we have not generated any revenue from product sales and do not expect to generate any revenue from product sales in the near future. If development efforts for our product candidates are successful and result in our receipt of necessary regulatory approvals, or if our development efforts otherwise lead to any commercialized products or additional license agreements with third parties, then we may generate revenue in the future from product sales.
Research and Development Expenses, net
Research and development expenses consist primarily of costs incurred in connection with the development of our product candidates. These expenses include:
| | • | expenses incurred under agreements with clinical research organizations and contract manufacturing organizations, as well as investigative sites and consultants that conduct our clinical trials, preclinical studies and other scientific development services; |
| | • | manufacturing scale-up expenses and the cost of acquiring and manufacturing preclinical and clinical trial materials; |
| | • | employee-related expenses, including salaries, related benefits, travel and share-based compensation expenses for employees engaged in research and development functions, as well as external costs, such as fees paid to outside consultants engaged in such activities; |
| | • | license maintenance fees and milestone fees incurred in connection with various license agreements; |
| | • | costs related to compliance with regulatory requirements; and |
| | • | depreciation and other expenses. |
We recognize external development costs based on an evaluation of the progress to completion of specific tasks using information provided to us by our service providers.
We do not allocate employee costs or facility expenses, including depreciation or other indirect costs, to specific programs because these resources are deployed across multiple programs and, as such, the related costs are not separately classified. We use internal resources primarily to oversee our research, as well as for managing our preclinical development, process development, manufacturing and clinical development activities. Our employees work across multiple programs; therefore, we do not track the related expenses by program.
Research and development activities are fundamental to our business. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. As a result, we expect that our research and development expenses will increase substantially over the next several years as we continue to advance the development of our product candidates. We also expect to incur additional expenses related to milestone and royalty payments payable to third parties with whom we have entered into license agreements.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries and related benefits, share-based compensation expenses for personnel in executive and administrative functions, insurance and professional fees for legal, consulting, accounting and audit services.
We anticipate that our general and administrative expenses will increase in the future due to increased headcount and professional fees to support our continued research activities and development of our product candidates. We also anticipate that we will continue to incur accounting, audit, legal, regulatory, compliance, director and officer insurance costs, as well as investor and public relations expenses associated with being a public company. Additionally, once we believe that regulatory approval of a product candidate appears likely, we will begin to incur a material increase in payroll and related expenses as a result of preparation for commercial operations, particularly in respect of sales and marketing.
Financing expenses, net consist primarily of income or expenses related to revaluation of foreign currencies and interest income on our bank deposits.
The following table summarizes our results of operations for the years ended December 31, 2022 and 2021:
| | | Year ended December 31, | |
| | | 2022 | | | 2021 | |
| | | (in thousands) | |
| Operating Expenses: | |
| |
| Research and development | | $ | 16,977 | | | $ | 6,334 | |
| General and administrative | | | 11,556 | | | | 6,033 | |
| Total operating expenses | | | 28,533 | | | | 12,367 | |
| | | | | | | | | |
| Financing (income) expense, net | | | (353 | ) | | | 111 | |
| Loss before taxes | | | 28,180 | | | | 12,478 | |
| Taxes on income (benefit) | | | (534 | ) | | | — | |
| | | | | | | | | |
| Net loss | | $ | 27,646 | | | $ | 12,478 | |
Our results of operations have varied in the past and can be expected to vary in the future due to numerous factors. We believe that period-to-period comparisons of our operating results are not necessarily meaningful and should not be relied upon as indications of future performance.
Year ended December 31, 2022 Compared to the Year Ended December 31, 2021
Research and development expenses
Research and development expenses increased by approximately $10.7 million, or 168%, to approximately $17.0 million for the year ended December 31, 2022 compared to approximately $6.3 million for the year ended December 31, 2021. The increase resulted primarily from an increase in headcount and payments to consultants and subcontractors for clinical and pre-clinical activities.
General and administrative expenses
General and administrative expenses increased by approximately $5.6 million, or 92%, to approximately $11.6 million for the year ended December 31, 2022 compared to approximately $6.0 million for the year ended December 31, 2021. This increase was primarily due to increase in headcount and professional fees and insurance expense and share-based compensation.
Financing (income) expense, net
Financing expenses, net, increased by approximately $464 thousand, or 418%, to net income of $353 thousand for the year ended December 31, 2022 compared to a net loss of $111 thousand for the year ended December 31, 2021. Financing expense, net for the year ended December 31, 2022 was primarily related to foreign currency exchange rate differences, offset by interest income on deposits. Financing income, net for 2021 was primarily related to interest income on deposits, offset by foreign currency exchange rate differences.
Taxes on income, net, for the year ended December 31, 2022 were $534 thousand. The tax benefit is related to a tax return of Chemomab Therapeutics Inc., a wholly owned subsidiary of the Company, derived by carryback of net operating losses. Chemomab Therapeutics Inc. received $351 thousand in December 2022 on account of previous years and expects to receive the remainder $183 thousand in 2023.
The following table summarizes our cash flows for the years ended December 31, 2022 and 2021:
| | | Year ended
December 31, | | | Increase/(decrease) | |
| | | 2022 | | | 2021 | | | $ | | | %
| |
| | | (in thousands) | | | | | | | | |
| Net cash used in operating activities | | $ | (20,370 | ) | | $ | (12,374 | ) | | $ | (7,996 | ) | | | 65 | % |
| Net cash provided by (used in) investing activities | | | 19,533 | | | | (45,186 | ) | | | 64,719 | | | | (143 | )% |
| Net cash provided by (used in) financing activities | | | (808 | ) | | | 61,074 | | | | (61,882 | ) | | | (101 | )% |
| Net increase (decrease) in cash, cash equivalents and restricted cash | | $ | (1,645 | ) | | $ | 3,514 | | | $ | (5,159 | ) | | | (147 | )% |
Net cash used in operating activities for the year ended December 31, 2022 was approximately $20.4 million and included net loss of $27.6 million, partially offset by net cash used by changes in operating assets and liabilities of approximately $4.0 million and non-cash charges of $3.3 million, which mainly included share-based compensation expenses.
Net cash used in operating activities for the years ended December 31, 2021 was approximately $12.4 million and included net loss of $12.5 million, partially offset by net cash provided by changes in operating assets and liabilities of $1.9 million and non-cash charges of $2.0 million, which mainly included share-based compensation expenses.
Net cash provided by investing activities for the year ended December 31, 2022 was approximately $19.5 million, which was primarily related to investment in short-term deposits offset by purchasing of fixed assets.
Net cash used in investing activities for the year ended December 31, 2021 was $45.2 million, which was primarily related to purchase of fixed assets and investment in bank deposits.
Net cash used in financing activities for the year ended December 31, 2022 was approximately $0.8 million, consisting of $0.3 million of proceeds from the sale of ADSs, $0.1 million of proceeds from the exercise of stock options offset by the repurchase of shares in the amount of $1.2 million.
Net cash provided by financing activities for the year ended December 31, 2021 was $61.1 million, consisting of $58.7 million of proceeds from the sale of ADSs, primarily from the Private Placement (as defined and described below) and issuances under the Sales Agreement with Cantor, and $2.4 million of cash acquired in the Merger.
We expect our expenses to increase substantially as we advance the clinical trials of our product candidate. In addition, we expect to continue to incur additional costs associated with operating as a public company.
We believe that our existing cash, cash equivalents and bank deposits will enable us to fund our operating expenses and capital expenditure requirements at least through March 31, 2024. We have based these estimates on assumptions that may prove to be wrong, and we could expend our capital resources sooner than we expect. If we receive regulatory approval for any of our product candidates, we expect to incur significant commercialization expenses related to product manufacturing, sales, marketing and distribution.
Until such time, if ever, that we generate product revenue sufficient to achieve profitability, we expect to finance our cash needs through the sales of our securities and through other outside funding sources. Debt financing and preferred equity financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise additional funds through government and other third-party funding, collaboration agreements, strategic alliances, licensing arrangements or marketing and distribution arrangements, then we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings when needed, then we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market products or product candidates that we would otherwise prefer to develop and market.
Liquidity and Capital Resources
In connection with the Merger, on March 15, 2021, we entered into Securities Purchase Agreements with certain investors, pursuant to which we agreed to sell approximately $45.5 million of the ADSs in a private placement transaction (the Private Placement). The Private Placement closed on March 22, 2021, at which time we sold 2,619,270 ADSs together with warrants to purchase up to 261,929 ADSs at an exercise price of $17.35 per ADS. The warrants expire five years from the date of issuance, and, if exercised in full, will provide proceeds of approximately $4.5 million.
On April 30, 2021, we entered into the Sales Agreement with Cantor Fitzgerald & Co. (Cantor). Pursuant to the Sales Agreement, we may offer and sell, from time to time, ADSs having an aggregate offering price of up to $75 million through Cantor (the ATM Facility). Sales of ADSs, if any, under the Sales Agreement will be issued and sold pursuant to our Registration Statement on Form S-3 which was declared effective on May 17, 2021 and will be made in sales deemed to be an “at the market offering” as defined in Rule 415(a)(4) promulgated under the Securities Act. Pursuant to the Sales Agreement, Cantor has agreed to act as sales agent on a best efforts basis and use commercially reasonable efforts to sell on our behalf all of the ADSs we requested to be sold in accordance with the Sales Agreement, consistent with Cantor’s normal trading and sales practices, on mutually agreed terms.
On April 25, 2022, we filed a prospectus supplement with the SEC for the issuance and sale of up to $18,125,000 of ADSs in connection with the reactivation of the ATM Facility and pursuant to General Instruction I.B.6 of Form S-3, which, subject to certain exceptions, limits the amount of securities we are able to offer and sell under such registration statement during any twelve month period to one-third of our unaffiliated public float.
During the year ended December 31, 2022, we sold 130,505 ADSs at an average price of USD 2.11 per ADS, through the ATM facility, resulting in gross proceeds of $275,000.
As shown in the accompanying consolidated financial statements, we have incurred losses and cash flow deficits from operations since inception, resulting in an accumulated deficit at December 31, 2022 of approximately $64 million. We have financed operations to date primarily through public and private placements of equity securities. We anticipate that we will continue to incur net losses for the foreseeable future. We believe that our existing cash, cash equivalents and bank deposits will be sufficient to fund our projected cash needs at least through March 31, 2024. To meet future capital needs we would need to raise additional capital through equity or debt financing or other strategic transactions. However, any such financing may not be available to us on favorable terms or at all. Our failure to obtain sufficient funds on commercially acceptable terms when needed would have a material adverse effect on our business, results of operations and financial condition.
We estimate that our current liquidity resources will allow us to execute our business plans at least through March 31, 2024.
Developing drugs, conducting preclinical and clinical trials, obtaining commercial manufacturing capabilities and commercializing products is expensive, and we will need to raise substantial additional funds to achieve our strategic objectives. We will require significant additional financing in the future to fund our operations, including if and when we progress into clinical trials of our product candidates, obtain regulatory approval for one or more of our product candidates, obtain commercial manufacturing capabilities and commercialize one or more of our product candidates. Our future capital requirements will depend on many factors, including, but not limited to:
| | ● | the progress and costs of our preclinical and clinical trials and other research and development activities; |
| | ● | the scope, prioritization and number of our preclinical and clinical trials and other research and development programs; |
| | ● | the amount of revenues and contributions we receive under future licensing, collaboration, development and commercialization arrangements with respect to our product candidates; |
| | ● | the costs of development and expansion of our operational infrastructure; |
| | ● | the costs and timing of obtaining regulatory approval for one or more of our product candidates; |
| | ● | our ability, or that of our collaborators, to achieve development milestones, marketing approval and other events or developments under potential future licensing agreements; |
| | ● | the costs of filing, prosecuting, enforcing and defending patent claims and other intellectual property rights; |
| | ● | the costs and timing of securing manufacturing arrangements for clinical or commercial production; |
| | ● | the costs of contracting with third parties to provide sales and marketing capabilities for us or establishing such capabilities ourselves; |
| | ● | the costs of acquiring or undertaking development and commercialization efforts for any future products, product candidates or technology; |
| | ● | the magnitude of our general and administrative expenses; and |
| | ● | any additional costs that we may incur under future in- and out-licensing arrangements relating to one or more of our product candidates. |
Until we can generate significant recurring revenues, we expect to satisfy our future cash needs through capital raising or by out-licensing and/or co-developing applications of one or more of our product candidates. We cannot be certain that additional funding will be available to us on acceptable terms, if at all. If funds are not available, we may be required to delay, reduce the scope of or eliminate research or development plans for, or commercialization efforts with respect to, one or more of our product candidates and make the necessary change to our operations to reduce the level of our expenditures in line with available resources.
We are a development-stage company and it is not possible for us to predict with any degree of accuracy the outcome of our research and development efforts. As such, it is not possible for us to predict with any degree of accuracy any significant trends, uncertainties, demands, commitments or events that are reasonably likely to have a material effect on our net loss, liquidity or capital resources, or that would cause financial information to not necessarily be indicative of future operating results or financial condition. However, to the extent possible, certain trends, uncertainties, demands, commitments and events are described in this item.
Critical Accounting Estimates
Our financial statements are prepared in accordance with generally accepted accounting principles in the United States (GAAP). The preparation of our financial statements and related disclosures in accordance with GAAP requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, revenue, costs and expenses, and the disclosure of contingent assets and liabilities in our financial statements. We base our estimates on historical experience, known trends and events and various other factors that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. We evaluate our estimates and assumptions on an ongoing basis. Our actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in Note 2 to our consolidated financial statements, we believe that the following accounting estimates are those that include a higher degree of judgment or complexity and are reasonably likely to have a material impact on our financial condition or results of operations and are therefore considered critical accounting estimates.
We apply Accounting Standard Codification (ASC) 718-10, “Share-Based Payment,” which requires the measurement and recognition of compensation expenses for all share-based payment awards made to employees and directors, including employee options under our option plans based on estimated fair values.
ASC 718-10 requires that we estimate the fair value of equity-based payment awards on the date of grant using an option-pricing model. The fair value of the award is recognized as an expense over the requisite service periods in our statements of comprehensive loss. We recognize share-based award forfeitures as they occur, rather than estimate by applying a forfeiture rate.
In June 2018, the Financial Accounting Standards Board (FASB) issued Accounting Standards Update (ASU) 2018-07, “Compensation-Stock Compensation (Topic 718): Improvements to Nonemployee Share-Based Payment Accounting”, which simplifies the accounting for nonemployee share-based payment transactions by aligning the measurement and classification guidance, with certain exceptions, to that for share-based payment awards to employees. The amendments expand the scope of the accounting standard for share-based payment awards to include share-based payment awards granted to non-employees in exchange for goods or services used or consumed in an entity’s own operations and supersedes the guidance related to equity-based payments to non-employees. We adopted these amendments on January 1, 2019.
We recognize compensation expenses for the fair value of non-employee awards over the requisite service period of each award.
We estimate the fair value of options granted as equity awards using a Black-Scholes options pricing model. The option-pricing model requires a number of assumptions, of which the most significant are share price, expected volatility and the expected option term (the time from the grant date until the options are exercised or expire). We determine the fair value per share of the underlying stock by taking into consideration our most recent sales of stock, as well as additional factors that we deem relevant. Our board determined the fair value of ordinary shares based on valuations performed using the Option Pricing Method subject to relevant facts and circumstances. We have historically been a private company and lack company-specific historical and implied volatility information of our stock. Expected volatility is estimated based on volatility of similar companies in the biotechnology sector. Historically, we have not paid dividends and have no foreseeable plans to issue dividends. The risk-free interest rate is based on the yield from governmental zero-coupon bonds with an equivalent term. The expected option term is calculated for options granted to employees and directors using the “simplified” method. Grants to non-employees are based on the contractual term. Changes in the determination of each of the inputs can affect the fair value of the options granted and our results of operations.
Quantitative and Qualitative Disclosures about Market Risks
Foreign Currency Exchange Risk
Our functional currency is the U.S. Dollar. We are exposed to foreign exchange rate risk. We are located in Israel, where part of our general and administrative expenses costs is incurred in New Israeli Shekels. During each of the years ended December 31, 2022 and 2021, we recognized foreign currency transaction loss of $609 thousand and $176 thousand, respectively. These foreign currency transaction gains and losses were recorded in financial expenses. We believe that a 10% change in the exchange rate between the U.S. Dollar and New Israeli Shekel would not have a material impact on our financial position or results of operations.
As we continue to grow our business, our results of operations and cash flows will be subject to fluctuations due to changes in foreign currency exchange rates, which could adversely impact our results of operations. To date, we have not entered into any foreign currency hedging contracts to mitigate our exposure to foreign currency exchange risk.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
As a smaller reporting company, we are not required to provide the information required by this item.
Item 8. Financial Statements and Supplementary Data
See the Index to Consolidated Financial Statements on Page F-1 attached hereto.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
A. Disclosure Controls and Procedures
We have evaluated, with the participation of our chief executive officer and chief financial officer, the effectiveness of our disclosure controls and procedures (as that term is defined in Rule 13a-15(e) and 15d-15(e) under the Exchange Act) as of December 31, 2022. There are inherent limitations to the effectiveness of any system of disclosure controls and procedures, including the possibility of human error and the circumvention or overriding of the controls and procedures. Accordingly, even effective disclosure controls and procedures can only provide reasonable assurance of achieving their control objectives. Based upon our evaluation, our chief executive officer and chief financial officer concluded that our disclosure controls and procedures were effective as of December 31, 2022 to provide reasonable assurance that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the applicable rules and forms, and that it is accumulated and communicated to our management, including our chief executive officer and chief financial officer, as appropriate to allow timely decisions regarding required disclosure.
B. Management’s Annual Report on Internal Controls Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over our financial reporting. Internal control over financial reporting is defined in Rules 13a-15(f) and 15d-15(f) promulgated under the Exchange Act as a process designed by, or under the supervision of, our principal executive and principal financial officers and effected by our board of directors, management and other personnel to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP and includes those policies and procedures that:
| • | pertain to the maintenance of records that in reasonable detail accurately and fairly reflect the transactions and dispositions of our assets; |
| • | provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with GAAP, and that our receipts and expenditures are being made only in accordance with authorizations of our management; and |
| • | provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Projections of any evaluation of effectiveness to future periods are subject to the risks that controls may become inadequate because of changes in conditions or that the degree of compliance with the policies or procedures may deteriorate.
Our management, including our Chief Executive Officer and Chief Financial Officer, assessed the effectiveness of our internal control over financial reporting at December 31, 2022. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control—Integrated Framework (2013). Based on that assessment under those criteria, management determined that, as of December 31, 2022, our internal control over financial reporting was effective.
C. Attestation Report of the Registered Public Accounting Firm
This Annual Report on Form 10-K does not include an attestation report of our registered public accounting firm due to the Company’s status as an emerging growth company, as defined in Rule 12b-2 of the Exchange Act.
D. Changes in Internal Control over Financial Reporting
There were no changes in our internal controls over financing reporting during the last fiscal year.
Item 9B. Other Information
None.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
None.
PART III
Item 10. Directors, Executive Officers and Corporate Governance
Our directors and executive officers, their ages and positions as of the date of this Annual Report on Form 10-K are as follows:
| Name | | Age | | Position |
| Executive Officers: | | | | |
| Dale Pfost | | 65 | | Chairman of the Board, Chief Executive Officer |
| Donald Marvin | | 70 | | Chief Financial Officer, Executive Vice President and Chief Operating Officer |
| Adi Mor | | 41 | | Director, Chief Scientific Officer |
| Matthew Frankel | | 54 | | Chief Medical Officer |
| Non-Employee Directors: | | | | |
| Nissim Darvish†(2)(3) | | 57 | | Director |
| Alan Moses†(1) | | 74 | | Director |
| Claude Nicaise†(1) | | 69 | | Director |
| Neil Cohen†(2)(3) | | 58 | | Director |
| Jill Quigley†(1) | | 47 | | Director |
† Independent Director
| (1) | Member of Audit Committee |
| (2) | Member of Compensation Committee |
| (3) | Member of Corporate Governance and Nominating Committee |
Executive Officers
Dr. Dale Pfost has served as our Chief Executive Officer and a member of our Board since October 2021, and also as chairman of our Board since February 2022. Dr. Pfost has 35 years of experience as an entrepreneur, investor and business executive. From 2019 to 2021, Dr. Pfost served as Chief Executive Officer and Chairman of the Board of Lodo Therapeutics Corporation. From 2009 until 2019, Dr. Pfost served in a variety of roles at Microbiome Therapeutics, LLC, a company which he co-founded, including as a board member and Chief Executive Officer from 2009 until 2010 and again from 2016 until 2019. From 2010 until 2019, he served as a General Partner at Advent Life Sciences. From 2013 until 2017, Dr. Pfost served in a variety of roles at Vestagen Protective Technologies, including Chairman, Executive Chairman from 2013 until 2017, and Chief Executive Officer from 2015 until 2016. Prior to that, Dr. Pfost held various executive positions at the following companies: Chief Executive Officer at Receptor Biologix Inc. (from 2008 until 2009), President, Chief Executive Officer and Chairman at Acuity Pharmaceuticals, Inc. (from 2003 until 2007), President, Chief Executive Officer and Chairman at Orchid BioSciences, Inc. (from 1996 until 2002), President, Chief Executive Officer and Managing Director at Oxford Glycosciences Ltd. (from 1988 until 1996) and Director of Robotics and Automated Chemistry Systems at SmithKline Beckman (1984 until 1988). In addition to the foregoing, Dr. Pfost has served on the board of directors of several companies, including Bioelectronica Corp., AxoSim, Inc., JMB Companies LLC, Aura Bioscience, Louisiana BIO, CN Creative and Ancilia Biosciences. Dr. Pfost earned his BS degree from the University of California, Santa Barbara, and a PhD in physics from Brown University.
Donald Marvin has served as our Chief Financial Officer, Executive Vice President and Chief Operating Officer since November 8, 2021. Mr. Marvin has more than 35 years of experience at growth enterprises in corporate finance and fundraising, strategy, corporate development, mergers and acquisitions, and operations. Mr. Marvin served as Executive Vice President and Chief Financial Officer of Lodo Therapeutics from 2020 until 2021. He was previously Chairman, President and CEO of Concentric from 2014 to 2021. Prior to his positions at Concentric, Mr. Marvin was Managing Partner of Cairn Associates from 2006 until 2014. He was President and CEO of IdentiGEN from 2006 until 2009. Mr. Marvin was a co-founder of Orchid BioSciences, where he served as Chief Operating Officer, CFO and Senior Vice President of Corporate Development from 1997 to 2003, and President and Chief Executive Officer of Diatron Corporation from 1986 until 1994. Earlier in his career, Mr. Marvin held positions of increasing responsibility at Abbott, Boehringer Ingelheim, Bayer, and PepsiCo. Mr. Marvin earned a Bachelor of Science degree from The Ohio State University and an MBA from Iona College.
Dr. Adi Mor is the co-founder of Chemomab Ltd. and served as Chemomab Ltd.’s Chief Executive Officer, Chief Scientific Officer and a member of Chemomab Ltd.’s board of directors from its formation in 2011 until the Chemomab Ltd.-Anchiano Therapeutics Ltd. merger that was consummated on March 16, 2021 (the “Merger”), and has continued to serve in those capacities for our company thereafter. Dr. Mor’s tenure as the Company’s Chief Executive Officer concluded simultaneously with the approval of Dr. Dale Pfost’s employment by the Company’s shareholders on October 25, 2021. Dr. Mor has in-depth knowledge in immunology focusing on rare diseases and broad experience in designing, developing and patenting a novel class of monoclonal antibodies to treat inflammatory and fibrotic diseases. Dr. Mor received her Ph.D. in immunology from Tel Aviv University in the Department of Neurobiochemistry in Israel and is the lead author of numerous scientific journal publications in immunology and inflammatory disorders.
Dr. Matthew B. Frankel has 20 years of experience in the pharmaceutical industry, encompassing clinical development and medical affairs. From 2018 until November 2022, Dr. Frankel served as Vice President, Clinical Development and Medical Affairs, Specialty Pharma at Boehringer-Ingelheim Pharmaceuticals, Inc. Prior to that, he served as the Vice President & Head, Immunology and Dermatology Medical Unit at Novartis Pharmaceuticals Corporation from 2016 to 2018. From 2012 to 2016, Dr. Frankel served as the Executive Medical Director of Sandoz and from 2010 through 2012 held the role of Global Medical Director, Clinical Development at Reata Pharmaceuticals (RETA). From 2003 through 2010, he held various senior roles across different companies in the research field. Dr. Frankel received his undergraduate degree from Vassar College, his MD from the University of California, Los Angeles School of Medicine, and his MBA from the J. L. Kellogg Graduate School of Management.
Directors
Nissim Darvish, M.D., Ph.D. has served on our Board since March 16, 2021. Dr. Darvish is a General Partner at MeOhr Ventures, a venture capital fund. Dr. Darvish currently serves as a director of several private companies. Prior to his current position, Dr. Darvish served as a Venture Partner at OrbiMed Israel and as a member of the boards of directors of 9 Meters Biopharma Inc. and Medigus Ltd. Previously, Dr. Darvish was employed at Pitango Venture Capital, where he was a General Partner managing life sciences investments. He was also the founder and CEO of Impulse Dynamics, where he oversaw a $250 million realization event. Dr. Darvish obtained his M.D. and Ph.D. in Biophysics and Physiology from the Technion in Israel, and subsequently conducted his post-doctoral research at NIH. He has published over 100 patents and authored over 20 publications.
Alan Moses, MD, FACP has served on our Board since March 16, 2021. Dr. Moses is board certified by the ABIM with subspecialty certification in Endocrinology and Metabolism and is a Fellow of the American College of Physicians. Dr. Moses currently serves on the board of directors of BioFabUSA, a position he has held since 2018. Prior to that time, from 2008 to 2018, Dr. Moses served as the Global Chief Medical Officer of Novo Nordisk A/S (CPH: NOVO-B), a company he joined in 2004. Dr. Moses served as a Professor of Medicine at Harvard Medical School from 2002 to 2006, and in collaboration with MIT, he co-founded and co-directed the Clinical Investigator Training Program, which focused on training physician-scientists in translational research. Dr. Moses previously served as the Senior Vice President and Chief Medical Officer of the Joslin Diabetes Center from 1998 to 2004. Dr. Moses holds a BS from Duke University, North Carolina and an MD from Washington University School of Medicine, Missouri.
Claude Nicaise, MD has served on our Board since March 16, 2021. Dr. Nicaise is a physician with extensive U.S. and international experience in clinical drug development, strategic management, worldwide regulatory strategy, pharmaceuticals, biotechnology, including clinical cancer research, infectious diseases and neuroscience. Dr. Nicaise is the owner and founder of Clinical Regulatory Services, which provides consulting services to the life science and biotechnology industry in support of all aspects of clinical and regulatory development. Since 2015, Dr. Nicaise has served on the board of directors and as the Chairman of the Compensation Committee of Sarepta Therapeutics, Inc. (NASDAQ: SRPT). Dr Nicaise has also served on the board of directors of Mynoryx Therapeutics since 2017. Prior to that time, from 2008 to 2014, Dr. Nicaise served as the Senior Vice President of Alexion Pharmaceuticals Inc. (NASDAQ: ALXN), and between 1984 and 2008, he held numerous senior management roles at Bristol Myers Squibb (NYSE: BMY). Dr. Nicaise holds an MD and a degree in Internal Medicine, Clinical Oncology, from Brussels University, Belgium.
Neil Cohen has served as a member of our Board since April 2020 and served as our interim Chief Executive Officer from October 2020 until the consummation of the Merger. Mr. Cohen has served as the Chairman and Chief Executive Officer of Castel Partners Ltd. since January 2012. In 1994, he co-founded Israel Seed Partners, a leading venture capital firm, and managed the firm until 2019. Mr. Cohen has invested in and served on the boards of directors of many private technology companies, including a large number which were acquired or completed successful initial public offerings, including Compugen (Nasdaq: CGEN), Shopping.com (Nasdaq: SHOP, acquired by EBAY), Broadlight (acquired by Broadcom, Nasdaq: AVGO) and Cyota (acquired by RSA). He is a venture partner at SKY, an Israeli middle-market private equity firm, Hetz Ventures Management Ltd., an early-stage Israeli venture capital fund, and Shavit Capital. Mr. Cohen was previously the Business Editor of The Jerusalem Post and began his career in the private equity group at N M Rothschild & Sons Limited in London. Mr. Cohen received a B.A. and M.A. in Oriental Studies, with first class honors, from Oxford University.
Jill M. Quigley has served as a member of our Board since June 2022. Since December 2020, Ms. Quigley has served as a member of the board of directors of Terns Pharmaceuticals, Inc. (Nasdaq: TERN), including her role as chairperson of its audit committee. From November 2018 until December 2021, Ms. Quigley served as Chief Operating Officer of Passage BIO, Inc. (Nasdaq: PASG). Previously, she served as the Interim Chief Executive Officer and General Counsel of Nutrinia, Inc., from January 2016 to November 2018. From July 2012 to January 2016, Ms. Quigley served in various roles at Shire plc, most recently as Senior Legal Counsel. Ms. Quigley received her undergraduate degree in Communications, Legal Institutions, Economics & Governance (CLEG) from American University and J.D. from Rutgers School of Law.
Overall Role of the Board and Board Leadership Structure
Under the Israeli Companies Law, 5759-1999 (the “Companies Law”), our Board is responsible for setting our general policies and supervising the performance of management. Our Board may exercise all powers and may take all actions that are not specifically granted by the Companies Law or our articles of association to our shareholders or to management. Our executive officers are responsible for our day-to-day management and have individual responsibilities established by our Board.
Under the Companies Law, the chief executive officer of a public company, or a relative of the chief executive officer, may not serve as the chairperson of the board of directors of such public company, and the chairperson of the board of directors of a public company, or a relative of the chairperson, may not be vested with authorities of the chief executive officer of such public company without obtaining shareholder approval pursuant to special majority requirements set forth in the Companies Law.
During a special general meeting of our shareholders held on February 10, 2022, our shareholders approved the appointment of Dr. Dale Pfost as Chairperson of our board of directors in addition to his role as our Chief Executive Officer. According to the Companies Law and the regulations promulgated thereunder, such appointment is valid for a three-year term and such re-appointment shall be subject to shareholder approval.
Our Board is currently composed of seven (7) members. Our Board consists of three classes of directors (as illustrated below), each serving staggered three-year terms. Upon expiration of the term of a class of directors, directors in that class will be elected for a three-year term at the Annual General Meeting of shareholders in the year in which that term expires. Each director’s term continues until the election and qualification of his or her successor, or his or her earlier death, resignation or removal. Any increase or decrease in the number of directors will be distributed among the three classes so that, as nearly as possible, each class will consist of one-third of the directors. The division of our board of directors into three classes with staggered three-year terms may delay or prevent a change of management or a change of control of our company.
| | • | Class I consists of Nissim Darvish and Jill Quigley, each with a term expiring at the 2025 annual meeting of shareholders. |
| | • | Class II consists of Neil Cohen and Claude Nicaise, each with a term expiring at the 2023 annual meeting of shareholders. |
| | • | Class III consists of Adi Mor, Alan Moses and Dale Pfost, each with a term expiring at the 2024 annual meeting of shareholders. |
Board Leadership Structure
The Company is led by Dr. Dale Pfost, who has served as our Chief Executive Officer and Chairman of the Board since October 2021 and February 2022, respectively. Although the Board does not have a formal policy on whether the roles of Chief Executive Officer and Chairman of the Board should be separated, we believe that our current board leadership structure is suitable for us. The Chief Executive Officer is the individual selected by the Board to manage our company on a day to day basis, and his direct involvement in our business operations makes him best positioned to lead productive Board strategic planning sessions and determine the time allocated to each agenda item in discussions of our company’s short- and long-term objectives.
Role of the Board in Risk Oversight
Our Board is responsible for overseeing our risk management process. Our Board focuses on our general risk management strategy, the most significant risks facing us, and oversees the implementation of risk mitigation strategies by management. Our Board is also apprised by management of particular risk management matters in connection with its general oversight and approval of corporate matters and significant transactions. The Board’s independent oversight function is further enhanced by the fact that all of the Board’s Committees are composed entirely of independent directors, the directors have complete access to management and the Board and its committees may retain their own respective advisors.
Corporate Governance Guidelines
Our Board strongly supports effective corporate governance and has developed and followed a program of strong corporate governance. Our Corporate Governance and Nominating Committee is responsible for overseeing our guidelines and reporting and making recommendations to the Board concerning corporate governance matters. Our guidelines are published on our website at https://investors.chemomab.com/documents and are available in print to any shareholder who requests them from our Corporate Secretary (or, in the absence thereof, by the Chief Executive Officer of the Company).
Based upon information requested from and provided by each director concerning their background, employment and affiliations, including family relationships, our Board has determined that each of the directors is independent as defined under Nasdaq listing standards, with the exceptions of Dr. Mor and Dr. Pfost. Our Board also determined that Nissim Darvish and Neil Cohen, who comprise the Compensation Committee and Neil Cohen and Nissim Darvish, who comprise the Corporate Governance and Nominating Committee, all satisfy the independence standards for such committees established by the SEC and Nasdaq listing standards, as applicable. With respect to the Audit Committee, our Board has determined that Jill Quigley, Alan Moses and Claude Nicaise satisfy the independence standards for such committee established by Rule 10A-3 under the Exchange Act, the SEC and Nasdaq listing standards, as applicable, and that Jill Quigley is a financial expert under the rules of the SEC. The Board considered the relationships between such directors and certain of the investors of the Company and determined that such relationships did not affect such directors’ independence under the standards of Nasdaq, or, where applicable, under SEC rules.
Code of Business Conduct and Ethics
We have adopted a Code of Business Conduct and Ethics that includes provisions ranging from restrictions on gifts to conflicts of interest. All of our employees and directors are bound by this Code of Business Conduct and Ethics. Violations of our Code of Business Conduct and Ethics may be reported to the Audit Committee. The Code of Business Conduct and Ethics includes provisions applicable to all of our employees, including senior financial officers and members of our Board and is posted on our website (https://investors.chemomab.com/). We intend to post amendments to or waivers from any such Code of Business Conduct and Ethics.
Our insider trading and blackout policy prohibits directors, officers and employees from engaging in transactions in publicly traded options, such as puts and calls, and other derivative securities with respect to our securities. This prohibition extends to any hedging or similar transactions designed to decrease the risks associated with holding our securities.
The Board meets regularly during the year and holds special meetings and acts by unanimous written consent whenever circumstances require. Independent directors meet at regular executive sessions without management present. During fiscal year 2022, the Board held 12 meetings and conducted certain business by unanimous consent. All of our directors are encouraged to attend our Annual Meeting of shareholders. Each of our directors attended at least 75% of the aggregate of the total number of meetings of the Board and the total number of meetings held by all committees of the Board on which such director served. Directors are expected to attend the Annual General Meeting.
Our Board has established the following committees. Each committee operates in accordance with a written charter that sets forth the committee’s structure, operations, membership requirements, responsibilities and authority to engage advisors.
Under the Companies Law, the Exchange Act and Nasdaq rules, we are required to establish an Audit Committee, and we have a separately-designated standing audit committee established in accordance with section 3(a)(58)(A) of the Exchange Act.
The responsibilities of an Audit Committee under the Companies Law include identifying and addressing flaws in the business management of the company, reviewing and approving related party transactions, establishing whistleblower procedures, overseeing the company’s internal audit system and the performance of its internal auditor, and assessing the scope of the work and recommending the fees of the company’s independent accounting firm. In addition, the Audit Committee is required to determine whether certain related party actions and transactions are “material” or “extraordinary” for the purpose of the requisite approval procedures under the Companies Law and to establish procedures for considering proposed transactions with a controlling shareholder.
In accordance with U.S. law and Nasdaq requirements, our Audit Committee is also responsible for the appointment, compensation and oversight of the work of our independent auditors and for assisting our Board in monitoring our financial statements, the effectiveness of our internal controls and our compliance with legal and regulatory requirements.
Under the Companies Law and related regulations, the Audit Committee must consist of at least three directors who meet certain independence criteria. Under the Nasdaq rules, we are required to maintain an Audit Committee consisting of at least three independent directors, all of whom are financially literate and one of whom has accounting or related financial management expertise. Each of the members of the Audit Committee is required to be “independent” as such term is defined in Rule 10A-3(b)(1) under the Exchange Act.
The members of the Audit Committee are Jill Quigley, Alan Moses and Claude Nicaise. Jill Quigley is the chairperson of the Audit Committee and is a financial expert under the rules of the SEC. Our Board has concluded that the composition of the Audit Committee meets the requirements for independence under the rules and regulations of Nasdaq and the SEC. During fiscal year 2022, the Audit Committee held 5 meetings.
Under both the Companies Law and Nasdaq rules, we are required to establish a Compensation Committee.
The responsibilities of a Compensation Committee under the Companies Law include recommending to the Board, for ultimate shareholder approval by a special majority, a policy governing the compensation of directors and officers based on specified criteria, reviewing modifications to and implementing such compensation policy from time to time, and approving the actual compensation terms of directors and officers prior to approval by the Board.
In accordance with U.S. law and Nasdaq requirements, our Compensation Committee is also responsible for the appointment, compensation and oversight of the work of any compensation consultant, independent legal counsel and other advisors retained by the Compensation Committee.
The Companies Law and related regulations require the appointment of a Compensation Committee that complies with the requirements of Nasdaq. Under Nasdaq rules, we are required to maintain a Compensation Committee consisting of at least two independent directors; each of the members of the Compensation Committee is required to be independent under Nasdaq rules relating to Compensation Committee members, which are different from the general test for independence of board and committee members. The members of the Compensation Committee are Nissim Darvish and Neil Cohen. Nissim Darvish is the chairperson of the Compensation Committee. Our Board has determined that each member of the Compensation Committee is independent within the meaning of the independent director guidelines of Nasdaq and under Rule 10C-1 under the Exchange Act. During fiscal year 2022, the Compensation Committee held six meetings and conducted certain business by unanimous consent.
Corporate Governance and Nominating Committee
We have established a Corporate Governance and Nominating Committee, responsible for making recommendations to the Board regarding candidates for directorships and the size and composition of the board. In addition, the committee is responsible for overseeing our corporate governance guidelines and reporting and making recommendations to the board concerning corporate governance matters. Under the Companies Law, nominations for director may also, under certain circumstances, be made by shareholders in accordance with the conditions prescribed by applicable law and our articles of association. The members of the Corporate Governance and Nominating Committee are Neil Cohen and Nissim Darvish. Neil Cohen is the chairperson of the Corporate Governance and Nominating Committee. Our Board has determined that each member of the Corporate Governance and Nominating Committee is independent within the meaning of the independent director guidelines of Nasdaq. During fiscal year 2022, the Compensation Committee held one meeting and conducted certain business by unanimous consent.
Board Diversity
While we do not currently have a formal diversity policy with respect to Board composition, the Board believes it is important for the Board to have gender, race and ethnicity diversity along with diversity of knowledge base, professional experience and skills. The Corporate Governance and Nominating Committee takes these qualities into account when considering director nominees for recommendation to the Board. We believe diversity enhances our effectiveness. Our Board currently has two female directors and two members with racial diversity. However, the Board remains committed to achieving a Board composition that represents a diversity of background and experience, inclusive of race, ethnicity, gender and sexual orientation. We will disclose a matrix with additional information about our board diversity in our proxy statement on Schedule 14A in connection with our annual general meeting of the shareholders to be convened in 2023.
Internal Auditor
Under the Companies Law, the board of directors is required to appoint an internal auditor recommended by the Audit Committee. The role of the internal auditor is to examine, among other things, whether the company’s actions comply with applicable law and proper business procedures. The internal auditor may not be an interested party, a director or an officer of the company, or a relative of any of the foregoing, nor may the internal auditor be our independent accountant or a representative thereof. Grant Thornton Israel currently serves as our internal auditor.
Section 16(a) Beneficial Ownership Reporting Compliance
Section 16(a) of the Exchange Act requires our directors, executive officers and holders of more than 10% of our ADSs to file with the SEC reports regarding their ownership and change in ownership of our equity securities. We believe that all Section 16 filings requirements were met by such persons during the fiscal year ended December 31, 2022.
Code of Business Conduct and Ethics
We have adopted a Code of Business Conduct and Ethics that includes provisions ranging from restrictions on gifts to conflicts of interest. All of our employees and directors are bound by this Code of Business Conduct and Ethics. Violations of our Code of Business Conduct and Ethics may be reported to the Audit Committee. The Code of Business Conduct and Ethics includes provisions applicable to all of our employees, including senior financial officers and members of our Board of Directors and is posted on our website. We intend to post amendments to or waivers from any such Code of Business Conduct and Ethics.
Item 11. Executive Compensation
Summary Compensation Table
The table and summary below outline the compensation granted to individuals who served in the role of chief executive officer during the previous fiscal year and our three most highly compensated executive officers with respect to the year ended December 31, 2022. For purposes of the table and the summary below, “compensation” includes base salary, bonuses, equity-based compensation, retirement or termination payments, benefits and perquisites such as car, phone and social benefits and any undertaking to provide such compensation.
| Name and Principal Position | | Year | | Salary (1) ($) | | | Bonus (2) ($) | | | Option
Awards (3) ($) | | | All Other
Compensation (4) ($) | | | Total ($) | |
| Dale Pfost | | 2021 | | | 182,557 | | | | - | | | | 300,000 | | | | 22,868 | | | | 505,425 | |
Chief Executive Officer and Chairman (5) | | 2022 | | | 600,000 | | | | 300,000 | | | | 1,500,000 | | | | 75,160 | | | | 2,475,160 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| Adi Mor | | 2021 | | | 248,547 | | | | 167,000 | | | | 8,000 | | | | 64,453 | | | | 488,000 | |
Chief Scientific Officer, Director and Previous Chief Executive Officer (6) | | 2022 | | | 298,470 | | | | 120,000 | | | | - | | | | 16,926 | | | | 435,396 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| Donald Marvin | | 2021 | | | 88,276 | | | | - | | | | 102,390 | | | | 11,590 | | | | 202,256 | |
Chief Financial Officer, Executive Vice President and Chief Operating Officer (7) | | 2022 | | | 460,000 | | | | 207,000 | | | | 660,252 | | | | 60,397 | | | | 1,387,649 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| Sigal Fattal | | 2021 | | | 127,050 | | | | 122,000 | | | | 616,000 | | | | 8,952 | | | | 874,002 | |
Previous Interim Chief Financial Officer (8) | | | | | | | | | | | | | | | | | | | | | | |
(1) Salary includes gross base salary.
(2) Bonuses disclosed under row “2021” relate to bonuses accrued in 2021 and paid in 2021 and 2022. Bonuses disclosed under “2022” represent target bonuses for 2022 (the information is not final and based on certain estimates due to the fact that the Company has not yet determined actual bonuses for 2022).
(3) Represents the equity-based compensation expenses recorded in our consolidated financial statements for the years ended December 31, 2021 and 2022, based on the options’ fair value on the grant date, calculated in accordance with applicable accounting guidance for equity-based compensation. For a discussion of the assumptions used in reaching this valuation, see Note 8C to our consolidated financial statements filed with our Annual Report on Form 10-K for the year ended December 31, 2021 incorporated by reference in this Annual Report.
(4) Represents payments by us of social benefits on behalf of the officer. Such benefits may include, to the extent applicable, payments, contributions and/or allocations for risk insurance (e.g., life, or work disability insurance), payments for social security, vacation, medical insurance and benefits, and other benefits and perquisites consistent with our policies.
(5) Dr. Dale Pfost’s terms of compensation were approved by the Company’s shareholders on October 25, 2021, which includes an annual base salary of $600,000. Dr. Pfost’s compensation data set forth in the above table is pro-rated to reflect time served as the Company’s chief executive officer in 2021.
(6) In addition to her current positions of Chief Scientific Officer and a member of our Board, Dr. Adi Mor previously served as our Chief Executive Officer, and resigned from such role concurrent with the commencement of Dr. Dale Pfost’s service as our Chief Executive Officer on October 25, 2021.
(7) Mr. Donald Marvin’s employment with the Company commenced on November 4, 2021, which include an annual base salary of $460,000. The compensation data set forth in the above table is pro-rated to reflect time served as the Company’s Chief Financial Officer, Executive Vice President and Chief Operating Officer in 2021.
(8) Ms. Sigal Fattal previously served as our interim Chief Financial Officer, and resigned from such role concurrent with the commencement of Mr. Marvin’s service as our Chief Financial Officer on November 8, 2021.
Outstanding Equity Awards at Fiscal Year-End
The table below outlines the options to purchase ADSs held by our named executive officers outstanding as of December 31, 2022.
| | | Option awards |
| Name | | Number of securities
underlying
unexercised options
(#) exercisable | | | Number of
securities
underlying
unexercised options
(#)
unexercsiable | | Option
exercise
price ($) | | Option expiration
date |
Dale Pfost, Chief Executive Officer and Chairman of the Board | | | 133,977 | | | | 325,376(1) |
| | 10.05 | | October 25, 2031 |
| | | | | | | | | | | | | |
Adi Mor, Chief Scientific Officer, Director and Previous Chief Executive Officer | | | 131,698 | | | | - | | | 1.49 | | March 15, 2028 |
| | | | | | | | |
| | | | |
Donald Marvin, Chief Financial Officer, Executive Vice President and Chief Operating Officer | | | 53,320 | | | | 143,555(2) |
|
| 9.77 | | November 8, 2031 |
(1) The options granted to Dr. Pfost vest and become exercisable over a period of four (4) years with one quarter (1/4) of the options vesting on October 25, 2022, the first anniversary of the grant date, and the remainder in equal amounts over the ensuing 36 monthly periods, subject to Dr. Pfost’s continued service.
(2) The options granted to Mr. Marvin vest and become exercisable over a period of four (4) years, with one quarter (1/4) of the options vesting on November 8, 2022, the first anniversary of the grant date, and the remainder in equal amounts over the ensuing 36 monthly periods, subject to Mr. Marvin’s continued service.
Employment Agreements
Employment Agreement with Dr. Dale Pfost, our Chief Executive Officer
On September 1, 2021, the Company’s board of directors approved the appointment of Dr. Dale Pfost as Chief Executive Officer of the Company, and on October 25, 2021, the shareholders of the Company approved the terms of employment of Dr. Pfost, pursuant to an Executive Employment Agreement entered into between Dr. Pfost and Chemomab Therapeutics Inc., the Company’s wholly owned subsidiary. In accordance with terms of Dr. Pfost’s employment agreement, Dr. Pfost receives: (i) an annual base salary of $600,000 (the “Base Salary”); (ii) an initial target annual cash incentive bonus of 50% of the Base Salary and an additional potential bonus of 10% of the Base Salary based on Dr. Pfost’s achievement of certain predetermined goals, which shall be determined at the discretion of the Company’s board of directors; (iii) options to purchase 459,353 ADSs of the Company (the ”Options”), constituting 3.5% of the outstanding and issued ADSs of the Company (on a fully diluted basis) which will vest over a period of four (4) years with one quarter (1/4) of the Options vesting on the first anniversary of the grant date and the remainder in equal amounts over the ensuing 36 monthly periods, unless such options have been cancelled in accordance with the terms and conditions of the 2015 Plan (as defined below); (iv) a one-time signing bonus in an amount of $80,000; (v) a one-time bonus in an amount of $80,000 upon the establishment of a new office in the United State and Dr. Pfost’s domiciling in the location of the new office, which will be payable within 15 days of such domiciling; (vi) 25 days of paid time off (“PTO”) per year, capped at 50 days of accrued PTO; and (vii) certain severance benefits payable in the event that the Company terminates Dr. Pfost’s employment without Cause (as defined in the employment agreement), provided that the total amount of the cash portion of severance benefits will not exceed two hundred percent (200%) of Dr. Pfost’s annual base salary at the rate in effect on the date of termination.
Additionally, in the event of termination of Dr. Pfost’s employment without Cause (as defined therein) as the result of a merger or sale of all or substantially all of the Company’s capital stock or assets, (i.e. a change in control of the Company), vesting of all unvested Options will accelerate and all unvested Options will immediately vest and become exercisable. Furthermore, in the event of termination of Dr. Pfost’s employment without Cause, other than as a result of a merger or sale of all or substantially all of the Company’s capital stock or assets (i.e. a change in control of the Company) or if Dr. Pfost terminates his employment for Good Reason (as defined in the employment agreement): (a) any time-based Options then outstanding and due to vest on the twelve (12) month anniversary of Dr. Pfost’s employment commencement date will accelerate and become exercisable if Dr. Pfost has been employed by the Company at such time for six (6) months or more but less than twelve (12) months; and (b) all time-based Options then outstanding will accelerate and become exercisable if Dr. Pfost has been employed by the Company at such time for twelve (12) months or longer.
The foregoing description of Dr. Pfost’s employment agreement is qualified in its entirety by reference to the full text of the employment agreement, a copy of which is filed as Exhibit 10.9 hereto.
Employment Agreement with Dr. Adi Mor, our Chief Scientific Officer (and former Chief Executive Officer)
Dr. Adi Mor provides services to the Company pursuant to the terms of a consulting agreement among Dr. Mor, an Israeli company co-owned by Dr. Mor, and the Subsidiary, dated April 18, 2022 (the “Consulting Agreement”). Pursuant to the terms of the Consulting Agreement, Dr. Mor is entitled to a gross monthly payment, an annual performance bonus, subject to her meeting certain performance milestones, as to be determined by our board of directors on an annual basis, and certain other benefits. Dr. Mor’s current monthly payment and annual performance bonus target were set and approved by the Company’s shareholders as described below. In addition, Dr. Mor is entitled to other benefits that are provided for by Israeli law or that are customary for senior executives in Israel, including reimbursement for reasonable expenses incurred in connection with her services and payment for variable and fixed costs of a car. The Consulting Agreement is terminable by either party upon 60 days prior written notice, and contains customary provisions regarding noncompetition, confidentiality of information, and assignment of inventions.
As required under Israeli law, the terms of Dr. Mor’s Consulting Agreement with the Subsidiary were approved by the board of directors and presented to the shareholders for approval. At the annual meeting of our shareholders that took place on June 7, 2022, our shareholders approved for following terms of employment for Dr. Mor: (a) an increase to her base gross monthly salary from 67,500 NIS (approximately $21,090) (plus social benefits) to 74,250 NIS (approximately $23,200), (plus social benefits) which represents an increase of 6,750 NIS (approximately $2,110), effective as of March 7, 2022; (b) an increase in her 2021 gross annual bonus from $100,000 to $110,250, an increase of approximately 10%; and (c) an increase in her annual gross target bonus opportunity from $100,000 to 45% of Dr. Mor’s annual gross payment, effective as of January 1, 2022. In lieu of the monthly salary payment of 74,250 NIS (plus social benefits), as approved by the shareholders, Dr. Mor receives the equivalent amount in the form of a monthly payment equal to 98,730 NIS under the Consulting Agreement.
The foregoing description of the Consulting Agreement is qualified in its entirety by reference to the full text of the consulting agreement, a copy of which is filed as Exhibit 10.11 hereto.
Employment Agreement with Mr. Donald Marvin, our Chief Financial Officer, Executive Vice President and Chief Operating Officer
On November 8, 2021, Chemomab Therapeutics Inc. entered into an Executive Employment Agreement with Mr. Donald Marvin. In accordance with his employment agreement, Mr. Marvin receives an annual base salary of $460,000, and is a part of the Company’s bonus program with a yearly bonus potential of 45% of his base annual base salary, which bonus will be based on the achievement of mutually agreeable objectives to be determined by Mr. Marvin and the Chief Executive Officer of the Company. Additionally, in accordance with his employment agreement, Mr. Marvin received (i) 1.5% of the outstanding equity of the Company, which exercise price is based on the average of the ADS market value over the 30 calendar days preceding November 8, 2021, and vesting over four years, (ii) an additional 0.5% of the outstanding equity of the Company for the achievement of strategic goals as agreed upon with the Compensation Committee and approved by the Board of Directors, (iii) a $25,000 signing bonus, and (iv) a 12-month initial severance package, which will increase by one month every two years that Mr. Marvin is employed by the Company, provided however that such amount does not exceed 18 months.
Additionally, in the event of termination of Mr. Marvin’s employment without Cause as the result of a merger or sale of all or substantially all of the Company’s capital stock or assets, (i.e. a change in control of the Company), vesting of all unvested Options will accelerate and all unvested Options will immediately vest and become exercisable. Furthermore, in the event of termination of Mr. Marvin’s employment without Cause, other than as a result of a merger or sale of all or substantially all of the Company’s capital stock or assets (i.e. a change in control of the Company) or if Mr. Marvin terminates his employment for Good Reason (as defined in the employment agreement): (a) any time-based Options then outstanding and due to vest on the twelve (12) month anniversary of Mr. Marvin’s employment commencement date will accelerate and become exercisable if Mr. Marvin has been employed by the Company at such time for six (6) months or more but less than twelve (12) months; and (b) all time-based Options due to vest on or before December 31 of the year in which termination occurs then outstanding will accelerate and become exercisable if Mr. Marvin has been employed by the Company at such time for twelve (12) months or longer.
The foregoing description of Mr. Marvin’s employment agreement is qualified in its entirety by reference to the full text of the employment agreement, a copy of which is filed as Exhibit 10.10 hereto.
Director Compensation Table
The table below outlines compensation earned by our non-executive directors for the fiscal year ended December 31, 2022, including fees earned in cash and options awarded for services provided as a director:
| Name | | Fees earned
or paid
in cash ($) | | | Option
awards ($) | | | Total ($) | |
| Nissim Darvish | | | 47,000 | | | | 76,000 | (1) | | | 123,000 | |
| Jill Quigley | | | 23,000 | | | | 15,000 | (2) | | | 38,000 | |
| Alan Moses | | | 43,000 | | | | 76,000 | (3) | | | 119,000 | |
| Claude Nicaise | | | 47,000 | | | | 76,000 | (4) | | | 123,000 | |
| Neil Cohen | | | 47,000 | | | | 76,000 | (5) | | | 123,000 | |
(1) (i) 11,884 of the options granted to Dr. Darvish vest and become exercisable in equal monthly installments over a 36 month period commencing on March 16, 2021, and (ii) 6,820 options vest and become exercisable on March 16, 2023, subject to Dr. Darvish’s continued service. Additional 10,123 options have fully vested.
(2) The options granted to Ms. Quigley vest and become exercisable in equal monthly installments over a 36 month period commencing on June 16, 2022, subject to Ms. Quigley’s continued service.
(3) (i) 11,884 of the options granted to Dr. Moses vest and become in equal monthly installments over a 36 month period commencing on March 16, 2021, and (ii) 6,820 of the options granted to Dr. Moses vest and become exercisable on March 16, 2023, subject to Dr. Moses’ continued service.
(4) (i) 11,884 of the options granted to Dr. Nicaise vest and become in equal monthly installments over a 36 month period commencing on March 16, 2021, and (ii) 6,820 of the options granted to Dr. Nicaise vest and become exercisable on March 16, 2023, subject to Dr. Nicaise's continued service.
(5) (i) 11,884 of the options granted to Mr. Cohen vest and become exercisable in equal monthly installments over a 36 month period commencing on March 16, 2021, (ii) 6,820 options vest and become exercisable on March 16, 2023, and (iii) 688 options vest and become exercisable over a period of three (3) years with one quarter (1/4) of the options vesting on July 16, 2021, the first anniversary of the grant date, and the remainder in equal amounts over the ensuing 24 monthly periods commencing on October 16, 2021, subject to Mr. Cohen’s continued service.
Director Compensation
We have adopted a Compensation Policy regarding the terms of office and employment of its “office holders” (as defined in the Israeli Companies Law, 5759-1999), including cash compensation, equity-based awards, releases from liability, indemnification and insurance, severance and other benefits. Chemomab compensates its directors and senior management team in accordance with the recommendation of its compensation committee and, generally, subject to the approval of our Board and shareholders. That compensation will generally need to be consistent with the terms of our Compensation Policy, which will require periodic approval, in accordance with the requirements of the Companies Law.
In addition to the Compensation Policy, on March 15, 2021, our shareholders approved certain actual compensation terms applicable to our current and future directors (the “Director Compensation Package”), pursuant to which (and among other terms) (i) our non-employee directors are entitled to receive an annual option grant of up to 0.05% of the Company’s share capital on a fully diluted basis, (ii) the chairman of our Board is entitled to receive an annual option grant of up to 0.2% of the Company’s share capital on a fully diluted basis and (iii) our non-employee directors are entitled to receive an annual cash fee of $35,000. The Director Compensation Policy implements terms for actual equity compensation that will be granted to directors on a regular basis and determines the annual cash fee that may be paid to non-employee directors. In accordance with the Companies Law, equity grants to our directors that are within the limitations of the Director Compensation Package require the approval of our compensation committee and Board, but not our shareholders, while any contemplated compensation made in excess of the Director Compensation Package will require the approval of our shareholders. Furthermore, any such compensation, including the annual cash fee payable to our non-employee directors, must be limited to the framework of the Director Compensation Package.
Outstanding Equity Awards at Fiscal Year End
The table below outlines options to purchase ADSs held by our non-employee directors outstanding as of December 31, 2022.
| | | Option awards |
| Name | | Number of ADSs
underlying
unexercised options
(#) exercisable | | | Number of
ADSs
underlying
unexercised options
(#)
unexercsiable | | | Option
exercise
price ($) | | Option
expiration
date |
| Nissim Darvish | | | 10,123 | | | | - | | | | 0.80 | | October 27, 2026 |
| Nissim Darvish | | | 6,932 | | | | 4,952 | | | | 27.26 | | April 19, 2031 |
| Nissim Darvish | | | - | | | | 6,820 | | | | 3.53 | | March 7, 2032 |
| | | | | | | | | | | | | | |
| Alan Moses | | | 6,932 | | | | 4,952 | | | | 27.26 | | April 19, 2031 |
| Alan Moses | | | - | | | | 6,820 | | | | 3.53 | | March 7, 2032 |
| | | | | | | | | | | | | | |
| Claude Nicaise | | | 6,932 | | | | 4,952 | | | | 27.26 | | April 19, 2031 |
| Claude Nicaise | | | - | | | | 6,820 | | | | 3.53 | | March 7, 2032 |
| | | | | | | | | | | | | | |
| Neil Cohen | | | 515 | | | | 173 | | | | 13.20 | | July 16, 2030 |
| Neil Cohen | | | 6,932 | | | | 4,952 | | | | 27.26 | | April 19, 2031 |
| Neil Cohen | | | - | | | | 6,820 | | | | 3.53 | | March 7, 2032 |
| | | | | | | | | | | | | | |
| Jill Quigley | | | 2,273 | | | | 11,367 | | | | 3.25 | | June 16, 2032 |
Equity Incentive Plans
We maintain (i) the 2011 Share Option Plan (the “2011 Plan”), (ii) the 2017 Plan and (iii) the 2015 Plan, which was assumed by our company from the Subsidiary upon the effectiveness of the Merger. At that time, outstanding options under the 2015 Plan became exercisable for such number of ADSs of our company (formerly known as Anchiano Therapeutics Ltd.) as was determined based on the exchange ratio in the Merger Agreement, with a reciprocal adjustment to exercise price. As of December 31, 2022, a total of 1,422,153 of ADSs were reserved for issuance under the 2015 Plan, of which 172,276 ADSs had been issued pursuant to previous exercises options, and 1,173,037 ADSs were issuable under outstanding options. Of such outstanding options, options to purchase 620,036 ADSs had vested and were exercisable as of that date, with a weighted average exercise price of $5.96 per ADS.
As of December 31, 2022, a total of 625,581 of ADSs were reserved for issuance under the 2017 Plan, of which 586,540 ADSs were issuable under outstanding options. Of such outstanding options, options to purchase 21,377 ADSs had vested and were exercisable as of that date, with a weighted average exercise price of $6.98 per ADS. No ADSs had been issued pursuant to previous exercises options.
2011 Plan
On December 19, 2011, our board of directors adopted the 2011 Plan to allocate options to purchase our ordinary shares to our directors, officers, employees and consultants, and those of our affiliated companies (as such term is defined under the 2011 Plan), or the Grantees. The 2011 Plan is administered by our board of directors or a committee that was designated by our board of directors for such purpose (the “Administrator”).
Under the 2011 Plan, we may grant options to purchase ordinary shares (“Options”), under four tracks: (i) Approved 102 capital gains Options through a trustee, which was approved by the Israeli Tax Authority in accordance with Section 102(a) of the Israeli Income Tax Ordinance (“ITO”), and granted under the tax track set forth in Section 102(b)(2) of the ITO, or the Approved 102 Capital Gains Options. The holding period under this tax track is 24 months from the date of allocation of Options to the trustee or such period as may be determined in any amendment of Section 102 of the ITO, or any applicable tax ruling or guidelines; (ii) Approved 102 Earned Income Options through a trustee, granted under the tax track set forth is Section 102(b)(1) of the ITO, or the Approved 102 Earned Income Options. The holding period under this tax track is 12 months from the date of allocation of Options to the trustee or such period as may be determined in any amendment of Section 102 of the ITO; (iii) Unapproved 102 Options (the Options will not be allocated through a trustee and will not be subject to a holding period), or the Unapproved 102 Options; and (iv) 3(i) Options (the Options will not be subject to a holding period). These Options shall be subject to taxation pursuant to Section 3(i) of the ITO, or Section 3(i).
Options pursuant to the first three tax tracks (under Section 102 of the ITO) can be granted to our employees and directors and the grant of Options under Section 3(i) can be granted to our consultants and controlling shareholders (a controlling shareholder is defined under the Section 102 of the ITO is a person who holds, directly or indirectly, alone or together with a “relative,” (i) the right to at least 10% of the company’s issued capital or 10% of the voting power; (ii) the right to hold at least 10% of the company’s issued capital or 10% of the voting power, or the right to purchase such rights; (iii) the right to receive at least 10% of the company’s profits; or (iv) the right to appoint a company’s director). Grantees who are not Israeli residents may be granted options that are subject to the applicable tax laws in their respective jurisdictions.
We determine, in our sole discretion, under which of the first three tax tracks above the Options are granted and we notify the Grantee in a grant letter, as to the elected tax track. As mentioned above, consultants and controlling shareholders can only be granted Section 3(i) Options.
The number of ordinary shares authorized to be issued under the 2011 Plan will be proportionately adjusted for any increase or decrease in the number of ordinary shares issued as a result of a distribution of bonus shares, change in our capitalization (split, combination, reclassification of the shares or other capital change), or issuance of rights to purchase ordinary shares or payment of a dividend. We will not allocate fractions of ordinary shares and the number of ordinary shares shall be rounded up to the closest number of ordinary shares.
Unless otherwise determined by the Administrator, the exercise price of an Option granted under the 2011 Plan will be the average of the market price of the Company’s ordinary shares during the 22 business days prior to the date on which our board of directors authorized the grant of Options; provided, however, that such exercise price cannot be lower than the market price at the close of the trading day at which it was granted by our board of directors. The exercise price will be specified in the grant letter every Grantee received from us in which the Grantee notifies of the decision to grant him/her Options under the 2011 Plan.
Unless otherwise determined by the Administrator, the Options granted under the 2011 Plan will become vested and may be exercised in 16 equal portions of 6.25% of the total number of Options, at the end of each quarter following the day the Options were granted. Unless otherwise determined by our board of directors, the Options may be exercised for ten years following the date of grant, unless terminated earlier, and as long as the Grantee is employed by the Company (or by an affiliated company), or provides service to the Company (or an affiliated company).
The Administrator may, in its absolute discretion, accelerate the time at which Options granted under the 2011 Plan or any portion of which will vest.
Unless otherwise determined by the Administrator, in the event that the Grantee’s employment was terminated, not for Cause (as defined in the 2011 Plan), the Grantee may exercise that portion of the Options that had vested as of the date of such termination until the end of the specified term in the grant letter or the 2011 Plan. The portion of the Options that had not vested at such date, will be forfeited and can be re-granted according to the terms of the 2011 Plan.
2015 Plan
In November 2015, the Subsidiary’s board of directors adopted, and its shareholders subsequently approved, the 2015 Plan. The 2015 Plan provides for the grant of options, restricted shares, restricted share units and other share-based awards to the Subsidiary’s (following the Merger, the Company’s) and its subsidiaries’ and affiliates’ directors, employees, officers, consultants, advisors, and any other person whose services are considered valuable to Chemomab or its affiliates. Any such grants are intended to incentivize the foregoing persons to continue as service providers, to increase their efforts on Chemomab’s behalf or on behalf of its subsidiaries or affiliates, and to promote the success of its business.
The 2015 Plan is administered by Chemomab’s board of directors or by a committee designated by the board of directors, which determines, subject to Israeli law, the grantees of awards and the terms of the grant, including, exercise prices, vesting schedules, acceleration of vesting and the other matters necessary in the administration of the 2015 Plan. The 2015 Plan enables Chemomab to issue awards under various tax regimes, including, without limitation, pursuant to Section 102 of the Israeli Income Tax Ordinance, or the Ordinance, and under Section 3(i) of the Ordinance and Section 422 of the United States Internal Revenue Code of 1986, as amended, or the Code.
The 2015 Plan provides that options granted to Chemomab’s employees, directors and officers who are not controlling shareholders and who are considered Israeli residents are intended to qualify for special tax treatment under the “capital gain track” provisions of Section 102(b) of the Ordinance. Chemomab’s Israeli non-employee service providers and controlling shareholders may only be granted options under Section 3(i) of the Ordinance, which does not provide for similar tax benefits.
Options granted under the 2015 Plan to U.S. residents may qualify as “incentive stock options” within the meaning of Section 422 of the Code, or may be non-qualified. The exercise price for “incentive stock options” must not be less than the fair market value on the date on which an option is granted, or 110% of the fair market value if the option holder holds more than 10% of Chemomab’s share capital.
Options and other awards granted under the 2015 Plan generally vest over four years commencing on the date of grant, such that 25% vests on the first anniversary of the date of grant and an additional 6.25% vests at the end of each subsequent calendar quarter over the course of the next three years, provided that the participant remains continuously employed or engaged by Chemomab.
Options, other than certain incentive share options, that are not exercised within ten years from the grant date expire, unless otherwise determined by Chemomab’s board of directors or its designated committee, as applicable. Share options that qualify as “incentive stock options” and are granted to a person holding more than 10% of Chemomab’s voting power will expire within five years from the date of the grant. In the event of the death of a grantee while employed by or performing service for Chemomab or its subsidiary or within three months after the date of the employee’s termination, or the termination of a grantee’s employment or services for reasons of disability, the grantee, or in the case of death, his or her legal successor, may exercise options or other awards that have vested prior to termination within a period of one year from the date of disability or death. If Chemomab terminates a grantee’s employment or service for cause, all of the grantee’s vested and unvested options or other awards will expire on the date of termination. If a grantee’s employment or service is terminated for any other reason, the grantee may generally exercise his or her vested options or other award within three months of the date of termination. Any expired or unvested options return to the pool and become available for reissuance. From time to time, Chemomab may consider issuing options with slightly different terms or accelerating, extending or otherwise modifying options in accordance with applicable law and regulation and the terms of the 2015 Plan.
In the event of a merger or consolidation of Chemomab, or a sale of all, or substantially all, of Chemomab’s shares or assets or other transaction having a similar effect on Chemomab, then without the consent of the option holder, Chemomab’s board of directors or its designated committee, as applicable, may, but is not required, to (i) cause any outstanding award to be assumed or an equivalent award to be substituted by such successor corporation, or (ii) in case the successor corporation does not assume or substitute the award (a) provide the grantee with the option to exercise the award as to all or part of the shares or (b) cancel the options and pay in cash an amount determined by the board of directors or the committee as fair in the circumstances. Notwithstanding the foregoing, Chemomab’s board of directors or its designated committee may upon such event amend, modify or terminate the terms of any award, including conferring the right to purchase any other security or asset that the board of directors or the committee shall deem, in good faith, appropriate.
The 2015 plan was assumed by our company from the Subsidiary upon the effectiveness of the Merger.
2017 Plan
On February 22, 2017, our board of directors adopted the 2017 Plan to allocate a variety of share-based awards to our directors, officers, employees, consultants, advisors and service providers, and those of our affiliates (companies that control us, are controlled by us or are under common control with us) (the “Participants”). The 2017 Plan is currently administered by our board of directors, and may be administered by a committee designated by our board of directors for such purpose.
Under the 2017 Plan, we may grant options to purchase ordinary shares or ADSs, restricted shares or ADSs, restricted share units and other awards based on our ordinary shares, all of which are referred to as Awards. We may grant Awards under the same four tracks as described above with respect to the 2011 Plan, subject to the same conditions as apply for the 2011 Plan. In addition, we may grant incentive stock options and nonqualified stock options to Participants who are residents of the United States, and we may grant awards to Participants who are residents of other countries that comply with the laws of those jurisdictions.
The number of ordinary shares authorized to be issued under the 2017 Plan will be proportionately adjusted for any increase or decrease in the number of ordinary shares issued as a result of a distribution of bonus shares, change in our capitalization (split, combination, reclassification of the shares or other capital change), issuance of rights to purchase ordinary shares or payment of a dividend. We will not allocate fractions of ordinary shares and the number of ordinary shares shall be rounded down to the closest number of ordinary shares.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The following table sets forth information, as of March 16, 2023, regarding beneficial ownership of our ordinary shares represented by ADSs:
| • | each person who is known by us to own beneficially more than 5% of our ordinary shares; |
| • | each executive officer; and |
| • | all of our directors and executive officers collectively. |
The percentages of ordinary shares beneficially owned are reported on the basis of regulations of the SEC governing the determination of beneficial ownership of securities. Under the rules of the SEC, a person is deemed to be a beneficial owner of a security if that person has or shares voting power, which includes the power to vote or to direct the voting of the security, or investment power, which includes the power to dispose of or to direct the disposition of the security.
Unless otherwise noted, the address of each director and current and former executive officer of Chemomab is Kiryat Atidim, Building 7, Tel Aviv, Israel 6158002.
| NAME OF BENEFICIAL OWNER | | Total Beneficial Ownership (ADSs) | | | Percentage of ADSs Beneficially Owned† | |
| 5% and Greater Shareholders | | | | | | |
| OrbiMed Israel (1) | | | 2,270,091 | | | | 20.5 | % |
| The Centillion Fund(2) | | | 661,370 | | | | 6.0 | % |
| Rivendell Investments 2017-9(3) | | | 1,131,563 | | | | 10.2 | % |
| Kobi George(4) | | | 747,445 | | | | 6.7 | % |
| Apeiron Group(5) | | | 770,388 | | | | 6.9 | % |
| Directors and Executive Officers | | | | | | | | |
| Dale Pfost (6) | | | 174,757 | | | | 1.6 | % |
| Donald Marvin (7) | | | 75,828 | | | | * | % |
| Adi Mor (8) | | | 747,445 | | | | 6.7 | % |
| Neil Cohen (9) | | | 25,702 | | | | * | |
| Nissim Darvish (10) | | | 26,395 | | | | * | |
| Alan Moses (11) | | | 15,072 | | | | * | |
| Claude Nicaise (12) | | | 15,072 | | | | * | |
| Jill Quigley (13) | | | 3,788 | | | | * | |
| Matthew Frankel | | | - | | | | - | |
| All current executive officers and directors as a group (9 persons) | | | 1,084,059 | | | | 9.40 | % |
† Percentage ownership based on 11,049,812 ADSs outstanding as of March 16, 2023
* Less than one percent (1%)
| (1) | Pursuant to a Schedule 13D/A filed with the SEC by OrbiMed Israel BioFund GP Limited Partnership (“OrbiMed BioFund”) and OrbiMed Israel GP Ltd. (“OrbiMed GP”, and together with OrbiMed BioFund, “OrbiMed Israel”) on January 5, 2023, such amount consists of (i) 2,241,274 ADSs and (ii) 28,817 ADSs issuable upon the exercise of warrants to purchase ADSs. OrbiMed GP, a company that acts as general partner of certain limited partnerships, is the general partner of OrbiMed BioFund, which is the general partner of OrbiMed Israel Partners Limited Partnership, which is the entity that holds the foregoing securities. The address of OrbiMed Israel is 89 Medinat HaYehudim St., Build E, 11th Floor, Herzliya 46766 Israel. |
| (2) | The address of Centillion Fund, Inc. is 10 Manoel Street, Castries, Saint Lucia. |
| (3) | Represents 1,108,509 ADSs, representing 22,170,180 ordinary shares, held by Rivendell Investments 2017-9 LLC, or Rivendell, as reported by Rivendell on Schedule 13G filed with the SEC on March 26, 2021, and 23,054 ADSs, representing 461,080 Ordinary Shares, issuable upon the exercise of warrants. Rivendell is the shareholder of record. Peter Thiel is the beneficial owner of Rivendell and has sole voting and investment power over the securities held by Rivendell. The address of Rivendell is 1209 Orange Street, Wilmington, Delaware 19801. |
| (4) | Consists of (i) 257,247 ADSs owned directly by Dr. George, (ii) 324,775 ADSs owned by Dr. Adi Mor (Dr. George’s spouse), (iii) 33,725 options to purchase 33,725 ADSs issued directly to Dr. George, issuable upon the exercise of options, and (iv) 131,698 options to purchase 131,698 ADSs, issued to Dr. Mor, (Dr. George’s spouse), as reported by Dr. Adi Mor on Schedule 13D/A filed with the SEC on November 17, 2022. |
| (5) | The Apeiron Group consists of (i) Apeiron SICAV Ltd. - Presight Capital Fund One, of which owns 438,993 ADSs, (ii) Apeiron Presight Capital Fund II, LP, of which owns 288,170 ADSs and 28,817 ADSs issuable upon the exercise of warrants and (iii) Apeiron Investment Group Ltd., of which owns 14,408 ADSs issuable upon the exercise of warrants. Each of Fabian Hansen and Christian Angermayer may be deemed to share voting and investment power with respect to the ADSs held by the Apeiron Group. |
| (6) | Includes 2,500 ADSs and 172,257ADSs issuable upon the exercise of options within 60 days of the date hereof as reported by Dr. Dale Pfost on Form 4 filed with the SEC on March 15, 2022. |
| (7) | Includes 2,000 ADSs and 73,828ADSs issuable upon the exercise of options within 60 days of the date hereof as reported by Mr. Donald Marvin on Form 4 filed with the SEC on June 21, 2022. |
| (8) | Consists of (i) 324,775 ADSs owned directly by Dr. Mor, (ii) 257,247 ADSs owned by Dr. George, (Dr. Mor’s spouse), (iii) 131,698 ADSs issued to Dr. Mor, issuable upon the exercise of options within 60 days of the date hereof, and (iv) 33,725 options to purchase 33,725 ADSs issued to Dr. George, (Dr. Mor’s spouse) issuable upon the exercise of options within 60 days of the date hereof, as reported by Dr. Adi Mor on Schedule 13D/A filed with the SEC on November 17, 2022. |
| (9) | Includes 10,000 ADSs, and 15,702 ADSs issuable upon the exercise of options within 60 days of the date hereof, as reported by Mr. Neil Cohen on Form 4 filed with the SEC on November 11, 2022. |
| (10) | Includes 1,200 ADSs, and 25,195 ADSs issuable upon the exercise of options within 60 days of the date hereof, as reported by Dr. Nissim Darvish on Form 4 filed with the SEC on March 14, 2022. |
| (11) | Represents 15,072 ADSs issuable upon the exercise of options within 60 days of the date hereof, as reported by Dr. Alan Moses on Form 4 filed with the SEC on March 9, 2022. |
| (12) | Represents 15,072 ADSs issuable upon the exercise of options within 60 days of the date hereof, as reported by Dr. Claude Nicaise on Form 4 filed with the SEC on March 9, 2022. |
| (13) | Represents 3,788 ADSs issuable upon the exercise of options within 60 days of the date hereof, as reported by Ms. Jill Quigley on Form 4 filed with the SEC on June 16, 2022. |
Securities Authorized for Issuance under Equity Compensation Plans
The following table provides certain information as of December 31, 2022, with respect to our equity compensation plans under which our equity securities are authorized for issuance:
| Plan Category | | Number of securities to be issued upon exercise of outstanding options, warrants and rights | | | Weighted-average exercise price of outstanding options, warrants and rights | | | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a)) | |
| | | (a) | | | (b) | | | (c) | |
| Equity compensation plans approved by security holders | | | 1,350,163 | | | $ | 7.65 | | | | 107,755 | |
| Equity compensation plans not approved by security holders | | | - | | | | - | | | | - | |
| Total | | | 1,350,163 | | | $ | 7.65 | | | | 107,755 | |
Item 13. Certain Relationships and Related Transactions, and Director Independence
Certain Relationships and Related Transactions
Described below are any transactions occurring since the beginning of our last fiscal year, and any currently proposed transactions to which either the Company was a party and in which:
| • | The amounts involved exceeded or will exceed$120,000; and |
| • | A director, executive officer, holder of more than 5% of the outstanding share capital of the Company, or any member of such person’s immediate family had or will have a direct or indirect material interest. |
The following transactions relate to the Registrant, formerly known as Anchiano Therapeutics Ltd., prior to the consummation of the Merger.
Related Party Transactions
Under the Israel’s Companies Law, 5759-1999, or the Companies Law, a related party transaction in which an “office holder” has a personal interest may be approved only if it is for the benefit of the company. An office holder is defined in the Companies Law as a director, a general manager, chief business manager, deputy general manager, vice general manager, any other person assuming the responsibilities of any of these positions regardless of such person’s title, and any other manager directly subordinate to the general manager. A transaction that is not an extraordinary transaction in which an office holder has a personal interest requires the approval of the board of directors, unless the articles of association of the company provide otherwise. If the transaction is an extraordinary transaction, it must be approved by the audit committee and the board of directors, and, under certain circumstances, by the shareholders of the company. An “extraordinary transaction” is a transaction other than in the ordinary course of business, other than on market terms or that is likely to have a material impact on the company’s profitability, assets or liabilities.
Pursuant to the Companies Law, extraordinary transactions in which a controlling shareholder has a personal interest require the approval of the audit committee, or the compensation committee if the transaction is in connection with employment or service with the company, the board of directors and the shareholders of the company. The shareholder approval must be by a simple majority of all votes cast, provided that (i) such majority includes a simple majority of the votes cast by non-controlling shareholders having no personal interest in the matter or (ii) the total number of votes of shareholders mentioned in clause (i) above who voted against such transaction does not exceed 2% of the total voting rights in the company.
In most cases, the Companies Law prohibits any director who has a personal interest in a transaction from being present for the discussion or voting pertaining to such transaction in the audit committee or board of directors. Nevertheless, a director who has a personal interest may be present at the meeting and vote on the matter if a majority of the directors or members of the audit committee have a personal interest in the approval of such transaction; in this case, however, the transaction also requires shareholder approval.
Director and Officer Compensation
Under the Companies Law, Chemomab is required to approve, at least once every three years, a compensation policy with respect to office holders. Following the recommendation of Chemomab’s compensation committee, the compensation policy must be approved by the board of directors and shareholders. The shareholder approval must be by a simple majority of all votes cast, provided that (i) such majority includes a simple majority of the votes cast by non-controlling shareholders having no personal interest in the matter or (ii) the total number of votes of shareholders mentioned in clause (i) above who voted against such transaction does not exceed 2% of the total voting rights in the company. In general, the terms of compensation of directors, the chief executive officer and any employee or service provider who is considered a controlling shareholder must be approved separately by the compensation committee, the board of directors and the shareholders. The compensation terms of other officers who report directly to the chief executive officer requires the approval of the compensation committee and the board of directors.
The Repurchase Arrangement
As previously reported in our Registration Statement on Form S-4 (File No. 333-252070), filed with SEC on January 13, 2021 and declared effective by the SEC on February 10, 2021, Chemomab Ltd. filed an application with the Israel Tax Authority for a tax ruling (the “Tax Ruling”) in connection with the Merger, pursuant to which certain of Chemomab Ltd.’s shareholders were entitled to defer an immediate Israeli tax liability resulting from the exchange of shares that otherwise would have been deemed a sale. The deferral of the aforementioned tax liability is set to lapse on March 16, 2023, which is the two-year anniversary of the closing date of the Merger. Dr. Adi Mor, co-founder of Chemomab Ltd. and both our Chief Scientific Officer and a Class III director, and Professor Kobi George, co-founder of Chemomab Ltd. (together with Dr. Adi Mor, the “Co-Founders”), will be required to pay a substantial tax liability to the Israeli Tax Authority upon the expiration date of the deferral period. In order to pay this tax liability, the Co-Founders had to sell part of their holdings in the Company. In light of the foregoing, we elected to enter into a share purchase agreement (the “Repurchase Arrangement”) with the Co-Founders whereby we agreed, subject to the requisite court approval required under Section 303(a) of the Companies Law, which we received on November 14, 2022, to repurchase up to 582,023 of our ADSs owned by the Co-Founders, for consideration not to exceed an aggregate amount of $2,500,000, depending on the market price of the ADSs at the time of any repurchase. Accordingly, on November 16, 2022, we repurchased the entire amount of 582,023 ADSs from the Co-Founders at a weighted average price of $2.0848 and for total consideration of approximately $1,213,400.
We do not expect any change in our cash runway as a result of the Repurchase Arrangement. Our current cash, cash equivalents and bank deposits are expected to last through at least March 31, 2024. We are likely to need to secure additional financing to complete our clinical development programs for PSC and SSc that are projected to have topline readouts in the second half of 2024. We continue to manage our cash runway and believe additional capital will be available when required.
Corporate Governance and Independent Directors
In compliance with the listing requirements of Nasdaq, we have a comprehensive plan of corporate governance for the purpose of defining responsibilities, setting high standards of professional and personal conduct and assuring compliance with such responsibilities and standards. We currently regularly monitor developments in the area of corporate governance to ensure we are in compliance with the standards and regulations required by Nasdaq.
Based upon information requested from and provided by each director concerning their background, employment and affiliations, including family relationships, our board of directors has determined that each of the directors is independent as defined under Nasdaq listing standards, with the exceptions of Dr. Mor and Dr. Pfost. Our board of directors also determined that Nissim Darvish and Neil Cohen, who comprise each of the compensation committee and the corporate governance and nominating committee, all satisfy the independence standards for such committees established by the SEC and Nasdaq listing standards, as applicable. With respect to the audit committee, our board of directors has determined that Jill Quigley, Alan Moses and Claude Nicaise satisfy the independence standards for such committee established by Rule 10A-3 under the Exchange Act, the SEC and Nasdaq listing standards, as applicable, and that Jill Quigley is a financial expert under the rules of the SEC. The board of directors considered the relationships between such directors and certain of the investors of the Company and determined that such relationships did not affect such directors’ independence under the standards of Nasdaq, or, where applicable, under SEC rules.
In addition, our articles of association allow our board of directors to appoint new directors to fill vacancies which occurred for any reason or as additional directors, provided that the number of board members shall not exceed the maximum numbers of directors mentioned above. The appointment of a director by the board shall be in effect until the following annual general meeting of the shareholders or until the end of his tenure in accordance with our articles of association. Our board of directors may continue to operate for as long as the number of directors is not less than the minimum number of directors mentioned above.
In addition, under the Companies Law, our board of directors must determine the minimum number of directors who are required to have financial and accounting expertise. Under applicable regulations, a director with financial and accounting expertise is a director who, by reason of his or her education, professional experience and skill, has a high level of proficiency in and understanding of business accounting matters and financial statements. He or she must be able to thoroughly comprehend the financial statements of the company and initiate discussion regarding the manner in which financial information is presented. In determining the number of directors required to have such expertise, the board of directors must consider, among other things, the type and size of the company and the scope and complexity of its operations. Our board of directors has determined that we require at least one director with the requisite financial and accounting expertise and that Jill Quigley has such expertise.
Item 14. Principal Accountant Fees and Services
Somekh Chaikin, a member firm of KPMG International, located in Tel Aviv, Israel, PCAOB ID 1057, has served as our independent registered public accounting firm for 2022 and 2021. The following table sets forth fees billed to us by our independent registered public accounting firm during the fiscal years ended December 31, 2022 and 2021 for (i) services rendered for the audit of our annual financial statements and the review of our quarterly financial statements; (ii) services by our independent registered public accounting firm that are reasonably related to the performance of the audit or review of our financial statements and that are not reported as audit fees; (iii) services rendered during the period in connection with tax compliance, tax advice and tax planning; and (iv) all other fees for services rendered.
| | | Year Ended December 31, | |
| | | 2022 | | | 2021 | |
Audit Fees
| | | 223 | | | | 173 | |
Tax Fees
| | | 30 | | | | 29 | |
All Other Fees
| | | -
| | | | - | |
Total
| | | 253 | | | | 202 | |
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Auditors
Our audit committee has the sole authority to approve the scope of the audit and any audit-related services, as well as all audit fees and terms. The audit committee must pre-approve any audit and non-audit services provided by our independent registered public accounting firm. The audit committee will not approve the engagement of the independent registered public accounting firm to perform any services that the independent registered public accounting firm would be prohibited from providing under applicable laws, rules and regulations, including those of self-regulating organizations. The audit committee will approve permitted non-audit services by our independent registered public accounting firm only if it determines that using a different firm to perform such services will be less efficient or cost-effective. The audit committee reviews and pre-approves the statutory audit fees that can be provided by the independent registered public accounting firm on an annual basis.
PART IV
Item 15. Exhibits and Financial Statement Schedules
| |
| | |
| |
| | |
| |
| | |
| |
| | |
| |
| | |
| |
| | |
| |
| | |
| |
| | |
| |
| | |
| 101.INS | Inline XBRL Instance Document |
| | |
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document |
| | |
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase Document |
| | |
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase Document |
| | |
| 101.LAB | Inline XBRL Taxonomy Extension Label Linkbase Document |
| | |
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase Document |
| | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
+ Indicates management contract or compensatory plan.
† The certifications furnished in Exhibits 32.1 and 32.2 hereto are deemed to accompany this Annual Report on Form 10-K and will not be deemed “filed” for purposes of Section 18 or the Securities Exchange Act of 1934, as amended, except to the extent that the registrant specifically incorporates it by reference.
++ Portions of this Exhibit (indicated with [***]) have been omitted as the Registrant has determined that (i) the omitted information is not material and (ii) the omitted information would likely cause competitive harm to the Registrant if publicly disclosed.
Item 16. Form 10-K Summary
Not applicable.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| | CHEMOMAB THERAPEUTICS LTD. |
| | | |
| Date: March 20, 2023 | By: | /s/ Dale Pfost |
| | | Dale Pfost
Chief Executive Officer |
POWER OF ATTORNEY
KNOW ALL PERSONS BY THESE PRESENTS, that each person whose signature appears below constitutes and appoints Dale Pfost and Donald Marvin, and each of them, as his or her true and lawful attorneys-in-fact and agents, with full power of substitution and re-substitution, for him or her and in his or her name, place and stead, in any and all capacities, to sign any and all amendments to this Annual Report on Form 10-K, and to file the same, with all exhibits thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he or she might or could do in person, hereby ratifying and confirming that said attorneys-in-fact and agents, or any of them, or their or his or her substitute or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
| Signature | | Title | | Date |
| | | | | |
| /s/ Dale Pfost | | Chief Executive Officer and Chairman of the Board | | March 20, 2023 |
| Dale Pfost | | (Principal Executive Officer) | | |
| | | | | |
| /s/ Donald Marvin | | Chief Financial Officer, Executive Vice President and Chief Operating Officer | | March 20, 2023 |
| Donald Marvin | | (Principal Financial and Accounting Officer) | | |
| | | | | |
| /s/ Adi Mor | | Director, Chief Scientific Officer | | March 20, 2023 |
| Adi Mor | | | | |
| | | | | |
| /s/ Nissim Darvish | | Director | | March 20, 2023 |
| Nissim Darvish | | | | |
| | | | | |
| /s/ Jill Quigley | | Director | | March 20, 2023 |
| Jill Quigley | | | | |
| | | | | |
| /s/ Alan Moses | | Director | | March 20, 2023 |
| Alan Moses | | | | |
| | | | | |
| /s/ Claude Nicaise | | Director | | March 20, 2023 |
| Claude Nicaise | | | | |
| | | | | |
| /s/ Neil Cohen | | Director | | March 20, 2023 |
| Neil Cohen | | | | |