Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
CMMB similar filings
- 11 Oct 24 Current report (foreign)
- 21 Aug 24 Current report (foreign)
- 25 Jul 24 Current report (foreign)
- 12 Jun 24 Current report (foreign)
- 9 May 24 Chemomab Therapeutics Announces First Quarter 2024 Financial Results and Provides
- 8 May 24 Chemomab Therapeutics Granted 180-Day Extension by Nasdaq to Regain Compliance
Filing view
External links
Exhibit 99.2

CM-101 Phase 2 SPRING TRIALPreliminary Topline Results JULY 25, 2024 NASDAQ: CMMB

This presentation contains forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995 that involve substantial risks and uncertainties. All statements other than statements of historical facts contained in this presentation, including statements regarding our future financial condition, results of operations, business strategy and plans, and objectives of management for future operations, as well as statements regarding industry trends, are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “estimate,” “intend,” “may,” “plan,” “potentially” “will” or the negative of these terms or other similar expressions. We have based these forward-looking statements largely on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including, among other things: the risk that the full data set from the CM-101 study or data generated in further clinical trials of CM-101 will not be consistent with the topline results of the CM-101 Phase 2 PSC trial; failure to obtain, or delays in obtaining, regulatory approvals for CM-101 in the U.S., Europe or other territories; failure to successfully commercialize CM-101, if approved by applicable regulatory authorities, in the U.S., Europe or other territories, or to maintain U.S., European or other territory regulatory approval for CM-101 if approved; uncertainties in the degree of market acceptance of CM-101 by physicians, patients, third-party payors and others in the healthcare community; inaccuracies in the Company's estimates of the size of the potential markets for CM-101 or in data the Company has used to identify physicians; expected rates of patient uptake, duration of expected treatment, or expected patient adherence or discontinuation rates; development of unexpected safety or efficacy concerns related to CM-101; failure to successfully conduct future clinical trials for CM-101, including due to the Company's potential inability to enroll or retain sufficient patients to conduct and complete the trials or generate data necessary for regulatory approval, among other things; risks that the Company's clinical studies will be delayed or that serious side effects will be identified during drug development; failure of third parties on which the Company is dependent to manufacture sufficient quantities of CM-101 for commercial or clinical needs, to conduct the Company's clinical trials, or to comply with the Company's agreements or laws and regulations that impact the Company's business or agreements with the Company; the strength and enforceability of the Company’s intellectual property rights or the rights of third parties; the cost and potential reputational damage resulting from litigation to which the Company may become a party, including product liability claims; changes in laws and regulations applicable to the Company's business and failure to comply with such laws and regulations; business or economic disruptions due to catastrophes or other events, including natural disasters or public health crises; and inability to repay the Company's existing indebtedness and uncertainties with respect to the Company's need and ability to access future capital; and the intensity and duration of the current war in Israel, and its impact on our operations in Israel. These risks are not exhaustive. You should carefully consider the risks and uncertainties described in the “Risk Factors” sections of our 20-F for the year ended December 31, 2023. New risk factors emerge from time to time, and it is not possible for our management to predict all risk factors, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in, or implied by, any forward-looking statements. You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this presentation. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities law of any such state or jurisdiction. 2 Forward Looking Statements

Clinical proof-of concept established Achieved primary and secondary exploratory efficacy endpoints across anti-inflammatory, anti-fibrotic and anti-cholestatic elements of PSC Achieved statistically significant improvement in Liver Stiffness measurement (FibroScan®) Reduced pruritis (itch) across all time points Broad and consistent activity of 20 mg/kg dose observed Greater activity in moderate/advanced patients observed Next steps: Targeting FDA End-of-Phase 2 meeting regarding potential registration trial in 4Q24 Completion of Open Label Extension in 1Q25 3 SPRING Trial: Positive Results Support Advancing CM-101 in PSC Provides strong foundation for advancing CM-101 in PSC and other indications

Primarily affects men in their 40’s Symptoms include fatigue, pruritis, abdominal pain and jaundice Diagnosed via serum liver enzyme abnormalities, cholangiography Unknown cause; associated with IBD in ~70% of patients 50% of patients require liver transplantation; PSC re-occurs in ~20% of recipients Leads to end-stage liver disease and cancer, which causes half of all deaths Median transplant-free survival is 10-20 years Debilitating orphan liver disorder with no FDA-approved therapies (1) Rupp C (2018) United European Gastroenterology Journal. 6(2): 255–262. (2) Palmela C (2018) Gut Liver. 12(1): 17-29. (3) Emek E (2019) Transplantation Proceedings 51(7): 2439-2441. (4) Bakhshi Z (2020). Journal of gastroenterology, 55(5), 523–532. Figure adapted from John Hopkins. 4 PSC Has High Unmet Need & Large Commercial Potential Progressive disease characterized by inflammation and fibrosis Fibrotic Stricture of Bile Duct Inflammationof Duct Wall Liver Gall bladder Stomach + Blocks Bile Duct backup damages liver Sizeable Market Opportunity, Orphan & Fast Track Incentives ~80,000 PSC patients in 7 major markets: U.S., Europe and Japan Commercial opportunity worldwide estimated at ~$1 billion

CM-101 Primary Sclerosing Cholangitis SPRING Trial Key Enrollment Criteria PSC patients with large duct disease of >24 weeks duration ALP > 1.5 ULN Stable IBD allowed Stable UDCA treatment allowed Outcome Measures Primary – Safety and tolerability Secondary - Change from baseline to Week 15 in: Liver stiffness by VCTE ( e.g. FibroScan®) ELF score Fibrotic biomarkers (Pro-C3) Pruritis Liver enzymes (ALP, ALT, AST, GGT) & Total Bilirubin Pharmacokinetics Pharmacodynamic parameters Prespecified Analysis Populations & Subgroup By all patients vs. patients with moderate/advanced disease* Territories: US, UK, Germany, Spain, Israel Phase 2 PSC SPRING Trial Design and Endpoints Randomized, Double-Blind, Placebo-Controlled--Includes dose-finding & open label extension Safety Follow-Up 6-Week Screening 33-Week Open Label Phase 15-Week Treatment Period 12-Week F/U 5 5 CM-101 10 mg/kg IV Q3W (n~25) CM-101 20 mg/kg IV Q3W (n~25) Placebo IV Q3W (n~18) Planned N~68 Patients CM-101 10 mg/kg IV Q3W CM-101 20 mg/kg IV Q3W CM-10 10/20 mg/kg IV Q3W PSC-primary sclerosing cholangitis; Q3W-once every 3 weeks; IV-intravenous; ALP-alkaline phosphatase; IBD-inflammatory bowel disease; UDCA-ursodeoxycholic acid; VCTE-Vibration controlled transient elastography-measure of liver stiffness; ELF–enhanced liver fibrosis score; Pro-C3-type lll collagen biomarker; AST-aspartate aminotransferase; ALT-alanine aminotransferase, GGT-gamma-glutamyl transferase; ULN –upper limit of normal *Includes patients with VCTE at baseline > 8.7 kPa
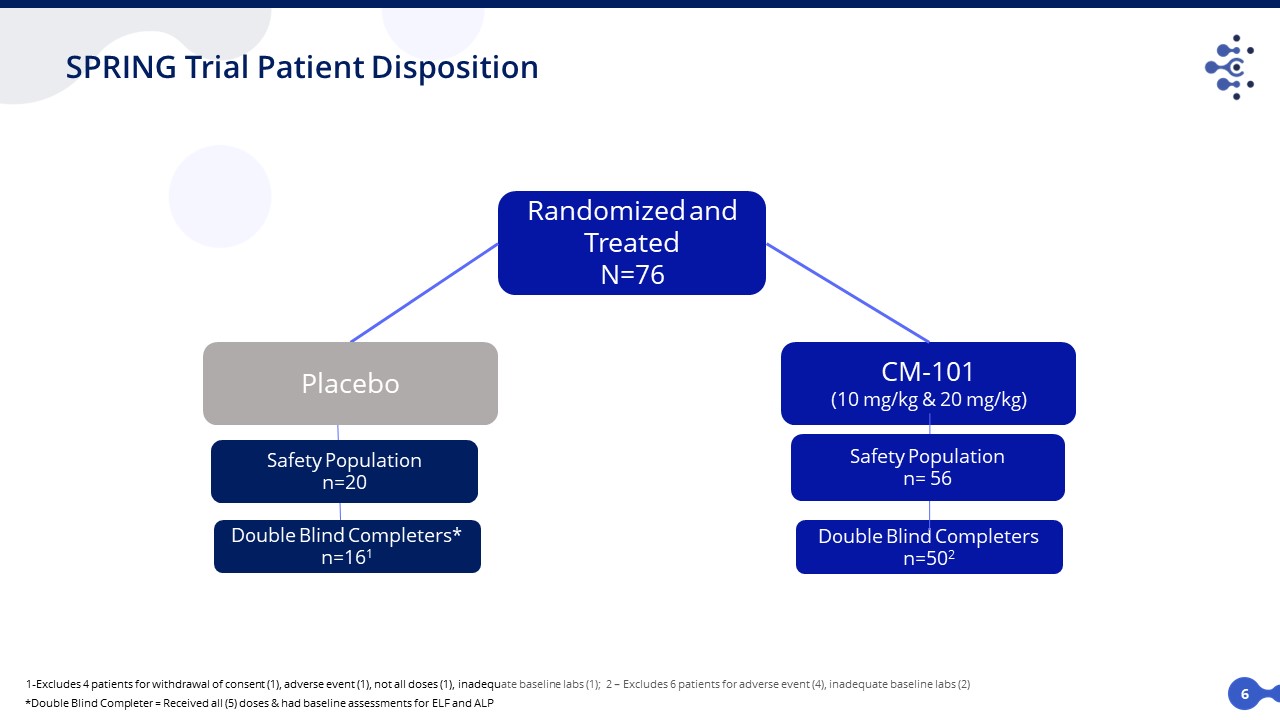
SPRING Trial Patient Disposition *Double Blind Completer = Received all (5) doses & had baseline assessments for ELF and ALP Randomized and Treated N=76 CM-101 (10 mg/kg & 20 mg/kg) Double Blind Completers n=502 Double Blind Completers* n=161 Safety Population n=20 Safety Population n= 56 Placebo 1-Excludes 4 patients for withdrawal of consent (1), adverse event (1), not all doses (1), inadequate baseline labs (1); 2 – Excludes 6 patients for adverse event (4), inadequate baseline labs (2) 6

Characteristic Placebo n=20 CM-101 10 mg/kg n=25 CM-101 20 mg/kg n=31 CM-101 (All) n=56 Age (year), mean, (range) 40.5 (23–75) 48.6 (23–74) 45.6 (23–67) 46.9 (23–74) Male Gender, n, (%) 13 (65%) 16 (64%) 17 (55%) 33 (59%) Race, n, (%) White Black Asian Multiple/ not reported/ unknown 19 (95%) 1 (5%) 0 (0) 0 (0) 19 (76%) 1 (4%) 0 (0) 5 (20%) 28 (90%) 2 (7%) 1 (3%) 0 (0) 47 (84%) 3 (5%) 1 (2%) 5 (9%) Concomitant UDCA use, n, (%) 15 (75%) 15 (60%) 18 (58%) 33 (59%) IBD, n (%) Crohn’s disease Ulcerative colitis 9 (45%) 3 (33%) 6 (67%) 18 (72%) 3 (17%) 15 (83%) 20 (65%) 5 (25%) 15 (75%) 38 (68%) 8 (21%) 30 (79%) Pruritis 5-D Itch scale total score, mean, (SD) 11.3 (4.5) 10.1 (5.0) 8.5 (3.3) 9.2 (4.2) Fatigue Severity Scale, mean, (SD) 32.2 (15.7) 30.0 (13.8) 30.2 (17.1) 30.1 (15.7) 7 SPRING Trial Patient Demographics (Safety Population) In general, placebo and treatment arms had similar demographics UDCA-ursodeoxycholic acid

Serum Liver Function Tests mean, (SD) Placebo n=20 CM-101 10 mg/kg n=25 CM-101 20 mg/kg n=31 CM-101 (All) n=56 Alkaline Phosphatase (U/L) 355 (203) 333 (189) 325 (167) 329 (176) Alanine Aminotransferase (U/L) 121 (83) 91 (63) 83 (51) 86 (56) Aspartate Aminotransferase (U/L) 87 (54) 59 (27) 64 (31) 62 (29) Total Bilirubin (umol/L) 15.2 (9.3) 14.6 (9.8) 16.8 (7.6) 15.9 (8.6) 8 SPRING Trial Baseline Characteristics (Safety Population) Disease Markers of Fibrosis mean, (SD) Placebo n=20 CM-101 10 mg/kg n=25 CM-101 20 mg/kg n=31 CM-101 (All) n=56 Serum CCL24 (ng/L) 947 (594) 1139 (710) 978 (757) 1048 (734) PRO-C3 (ng/mL) 49.4 (16.5) 57.6 (28.2) 49.6 (22.4) 53.2 (25.2) Liver stiffness by VCTE (kPa) 9.3 (4.0) 12.5 (13.1) 11.7 (12.0) 12.0 (12.4) Enhanced Liver Fibrosis Score (ELF) 9.75 (1.06) 9.66 (1.03) 9.84 (1.11) 9.76 (1.07) In general, placebo & treatment cohorts had similar baseline values VCTE-vibration-controlled transient elastography

Treatment Emergent Adverse Events N, (%) Placebo n=20 CM-101 10mg/kg n=25 CM-101 20mg/kg n=31 CM-101 (All) n=56 Treatment Emergent Adverse Events (TEAE) 15 (75%) 18 (72%) 28 (90%) 46 (82%) Related to study drug 9 (45%) 7 (28%) 16 (52%) 23 (41%) Serious TEAE 1 (5%) 2 (8%) 0 (0) 2 (4%) Related to study drug 0 0 0 0 TEAE leading to death 0 (0) 0 (0) 0 (0) 0 (0) TEAE leading to treatment discontinuation 1 (5%) 3 (12%) 2 (7%) 5 (9%) 9 SPRING Trial: CM-101 Demonstrated Favorable Safety & Tolerability Profile No Serious TEAEs were related to study drug & overall TEAEs were mostly mild & distributed relatively similarly across study arms
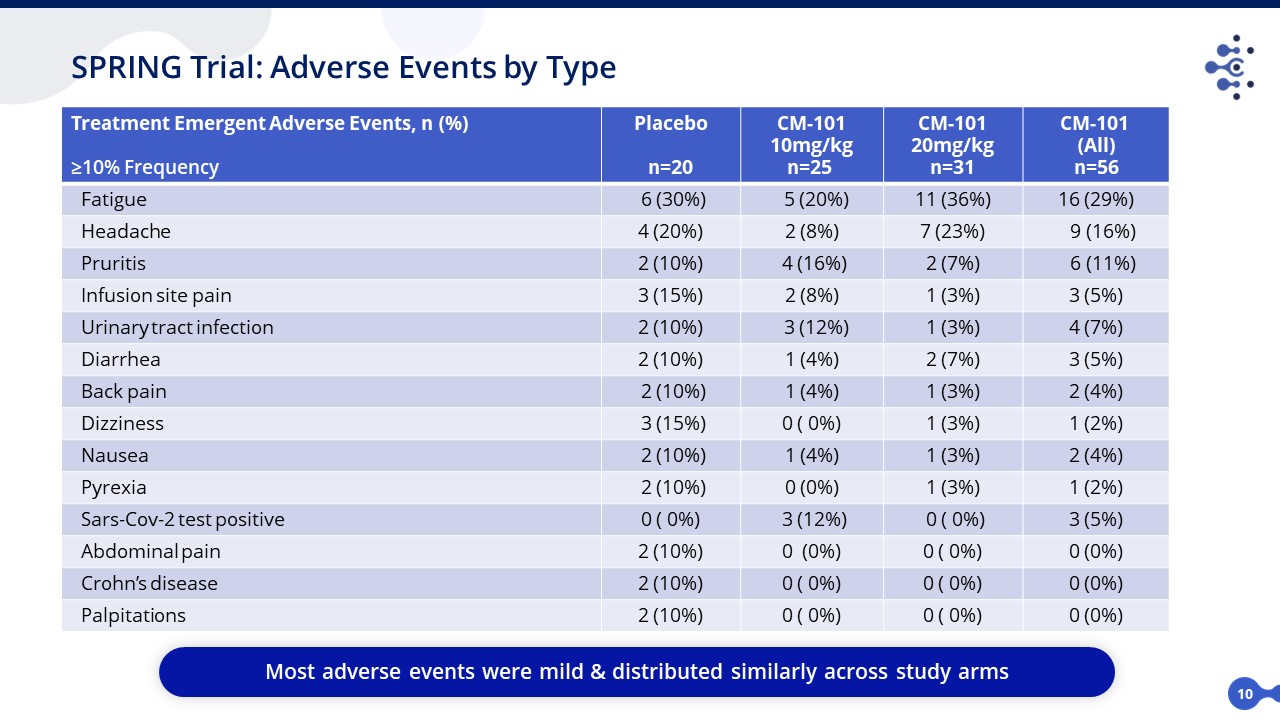
Treatment Emergent Adverse Events, n (%) ≥10% Frequency Placebo n=20 CM-101 10mg/kg n=25 CM-101 20mg/kg n=31 CM-101 (All) n=56 Fatigue 6 (30%) 5 (20%) 11 (36%) 16 (29%) Headache 4 (20%) 2 (8%) 7 (23%) 9 (16%) Pruritis 2 (10%) 4 (16%) 2 (7%) 6 (11%) Infusion site pain 3 (15%) 2 (8%) 1 (3%) 3 (5%) Urinary tract infection 2 (10%) 3 (12%) 1 (3%) 4 (7%) Diarrhea 2 (10%) 1 (4%) 2 (7%) 3 (5%) Back pain 2 (10%) 1 (4%) 1 (3%) 2 (4%) Dizziness 3 (15%) 0 ( 0%) 1 (3%) 1 (2%) Nausea 2 (10%) 1 (4%) 1 (3%) 2 (4%) Pyrexia 2 (10%) 0 (0%) 1 (3%) 1 (2%) Sars-Cov-2 test positive 0 ( 0%) 3 (12%) 0 ( 0%) 3 (5%) Abdominal pain 2 (10%) 0 (0%) 0 ( 0%) 0 (0%) Crohn’s disease 2 (10%) 0 ( 0%) 0 ( 0%) 0 (0%) Palpitations 2 (10%) 0 ( 0%) 0 ( 0%) 0 (0%) 10 SPRING Trial: Adverse Events by Type Most adverse events were mild & distributed similarly across study arms

SPRING Trial Data: Biological Activity Observed Across Multiple Parameters 11
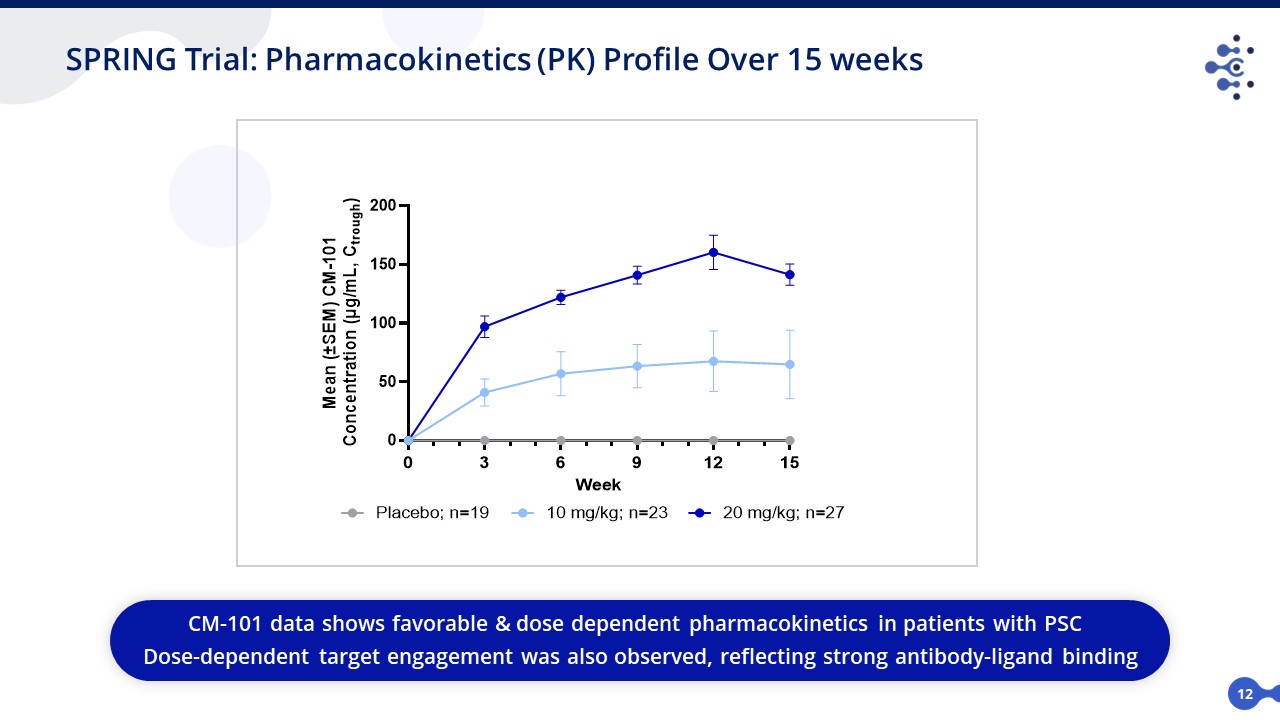
12 SPRING Trial: Pharmacokinetics (PK) Profile Over 15 weeks CM-101 data shows favorable & dose dependent pharmacokinetics in patients with PSC a Dose-dependent target engagement was also observed, reflecting strong antibody-ligand binding

13 SPRING Trial: Liver Stiffness Measures Improved in CM-101-Treated Patients *VCTE>8.7 kPa; 1Chazouillèreset al, EASL 2024 Significantly improved liver stiffness observed with CM-101 compared to placebo in patients with moderate/advanced disease All patients Patients with baseline moderate/advanced disease* Large prospective FICUS study presented at EASL 2024 validated the prognostic value of liver stiffness in PSC1 *Measured by VCTE (vibration-controlled transient elastography) e.g. FibroScan®
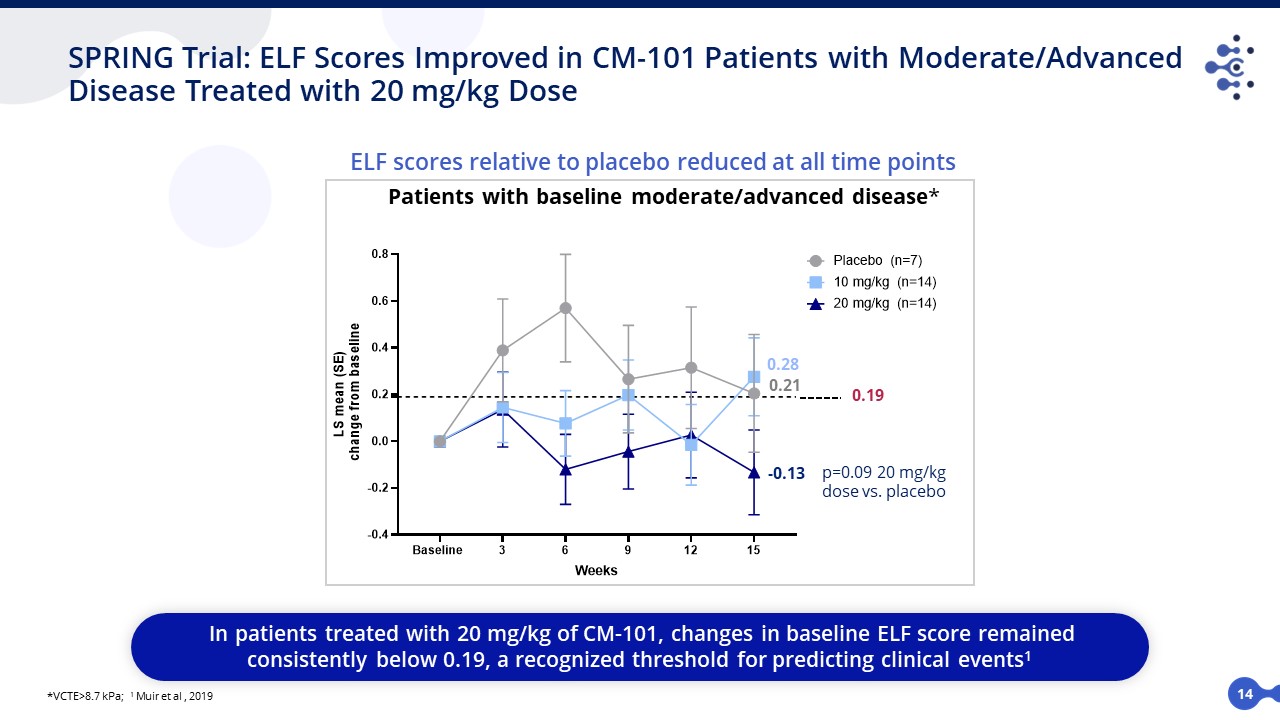
14 SPRING Trial: ELF Scores Improved in CM-101 Patients with Moderate/Advanced Disease Treated with 20 mg/kg Dose ELF scores relative to placebo reduced at all time points *VCTE>8.7 kPa; 1 Muir et al , 2019 In patients treated with 20 mg/kg of CM-101, changes in baseline ELF score remained consistently below 0.19, a recognized threshold for predicting clinical events1 Patients with baseline moderate/advanced disease* 0.19 -0.13 0.28 0.21 p=0.09 20 mg/kg dose vs. placebo

15 SPRING Trial: PRO-C3 Improved in CM-101-Treated Patients with Moderate/Advanced Disease Reduced PRO-C3 at week 15 relative to placebo observed for both 10 mg/kg & 20 mg/kg doses PROC-C3: neo-epitope pro-peptide of type III collagen formation. *VCTE>8.7 kPa; 1-Nielsen et al, 2018 Patients with baseline moderate/advanced disease* PRO-C3, a serum biomarker of type III collagen synthesis, was shown to be elevated in patients with PSC & identified as an independent predictor of transplant-free survival in PSC1

16 SPRING Trial: Pruritis Scores* Improved in CM-101-Treated Patients *Pruritis assessments based on 5D Itch Score-Total Domain; DBC population CM-101-treated patients experienced decreased pruritis scores across all timepoints compared to placebo
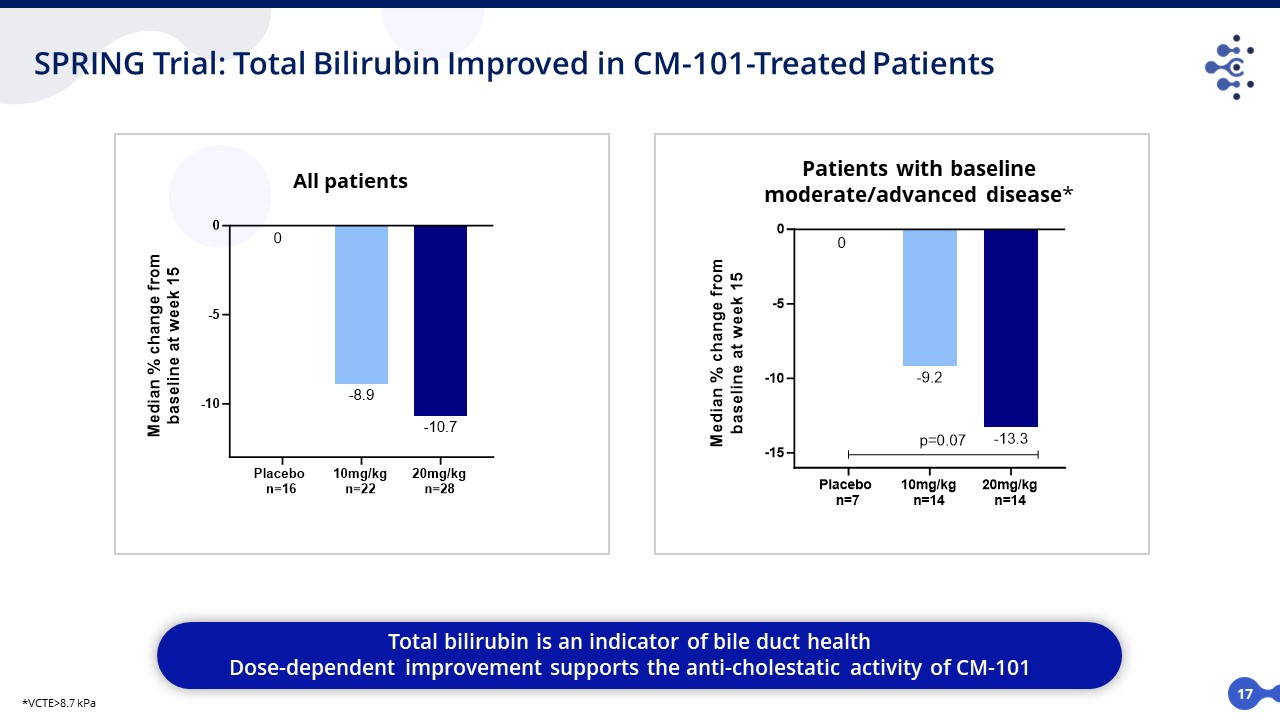
17 SPRING Trial: Total Bilirubin Improved in CM-101-Treated Patients Total bilirubin is an indicator of bile duct health Dose-dependent improvement supports the anti-cholestatic activity of CM-101 Patients with baseline moderate/advanced disease* *VCTE>8.7 kPa All patients

18 SPRING Trial: Liver Function Tests Improved in CM-101-Treated Patients Key liver function tests all improved in CM-101-treated patients Patients with baseline moderate/advanced disease* All patients *VCTE>8.7 kPa
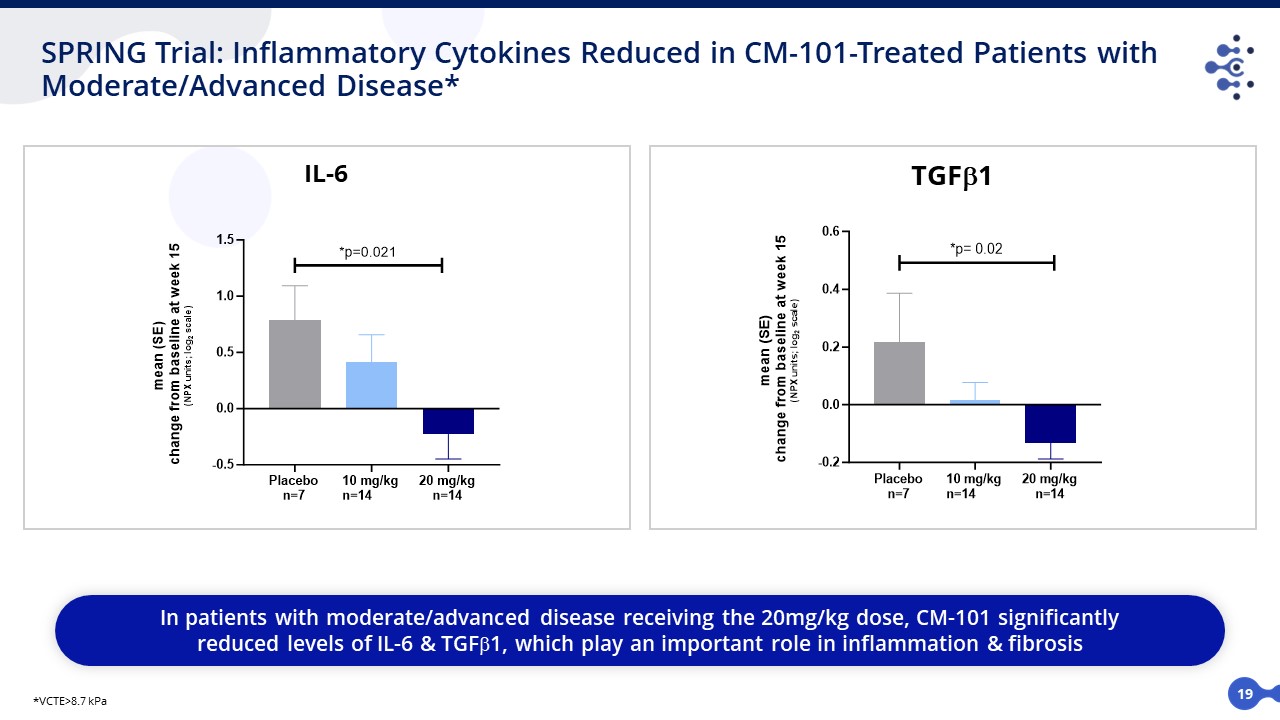
19 SPRING Trial: Inflammatory Cytokines Reduced in CM-101-Treated Patients with Moderate/Advanced Disease* IL-6 TGFb1 In patients with moderate/advanced disease receiving the 20mg/kg dose, CM-101 significantly reduced levels of IL-6 & TGF1, which play an important role in inflammation & fibrosis *VCTE>8.7 kPa

Clinical proof-of concept established Achieved primary and secondary exploratory efficacy endpoints across anti-inflammatory, anti-fibrotic and anti-cholestatic elements of PSC Achieved statistically significant improvement in Liver Stiffness measurement (FibroScan®) Reduced pruritis (itch) across all time points Broad and consistent activity of 20 mg/kg dose observed Greater activity in moderate/advanced patients observed Next steps: Targeting FDA End-of-Phase 2 meeting regarding potential registration trial in 4Q24 Completion of Open Label Extension in 1Q25 20 SPRING Trial: Positive Results Support Advancing CM-101 in PSC Provides strong foundation for advancing CM-101 in PSC and other indications