Exhibit 99.2
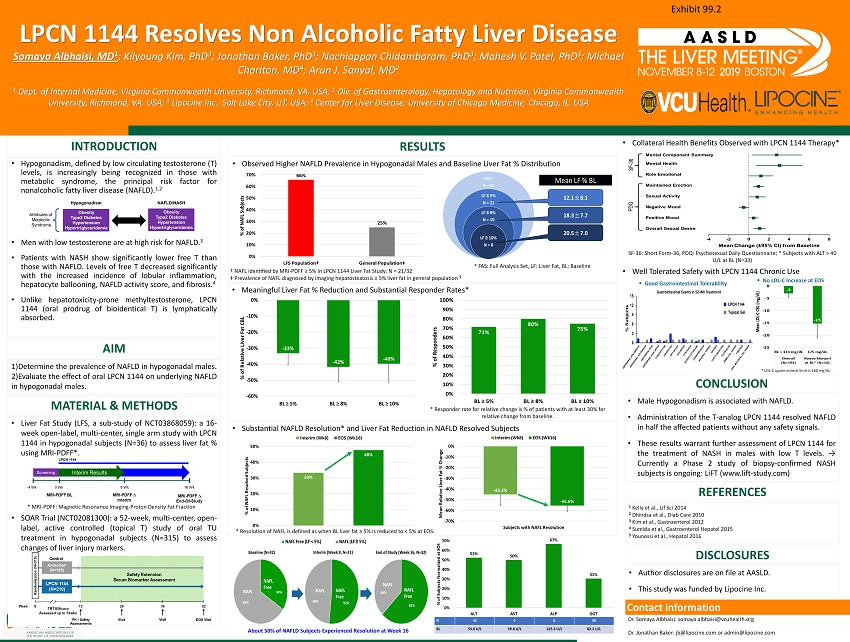
• Liver Fat Study (LFS, a sub - study of NCT 03868059 ) : a 16 - week open - label, multi - center, single arm study with LPCN 1144 in hypogonadal subjects (N= 36 ) to assess liver fat % using MRI - PDFF* . • SOAR Trial (NCT 02081300 ) : a 52 - week, multi - center, open - label, active controlled (topical T) study of oral TU treatment in hypogonadal subjects (N= 315 ) to assess changes of liver injury markers . LPCN 1144 Resolves Non Alcoholic Fatty Liver Disease Somaya Albhaisi, MD 1 ; Kilyoung Kim, PhD 3 ; Jonathan Baker, PhD 3 ; Nachiappan Chidambaram, PhD 3 ; Mahesh V. Patel, PhD 3 ; Michael Charlton, MD 4 ; Arun J. Sanyal, MD 2 1 Dept. of Internal Medicine, Virginia Commonwealth University, Richmond, VA, USA; 2 Div. of Gastroenterology, Hepatology and Nutrition, Virginia Commonwealth University, Richmond, VA , USA; 3 Lipocine Inc., Salt Lake City, UT, USA; 4 Center for Liver Disease, University of Chicago Medicine, Chicago, IL, USA INTRODUCTION RESULTS • Hypogonadism, defined by low circulating testosterone (T) levels, is increasingly being recognized in those with metabolic syndrome, the principal risk factor for nonalcoholic fatty liver disease (NAFLD) . 1 , 2 • Men with low testosterone are at high risk for NAFLD . 3 • Patients with NASH show significantly lower free T than those with NAFLD . Levels of free T decreased significantly with the increased incidence of lobular inflammation, hepatocyte ballooning, NAFLD activity score, and fibrosis . 4 • Unlike hepatotoxicity - prone methyltestosterone , LPCN 1144 (oral prodrug of bioidentical T) is lymphatically absorbed . • Male Hypogonadism is associated with NAFLD . • Administration of the T - analog LPCN 1144 resolved NAFLD in half the affected patients without any safety signals . • These results warrant further assessment of LPCN 1144 for the treatment of NASH in males with low T levels . → Currently a Phase 2 study of biopsy - confirmed NASH subjects is ongoing : LiFT (www . lift - study . com) 1 )Determine the prevalence of NAFLD in hypogonadal males . 2 )Evaluate the effect of oral LPCN 1144 on underlying NAFLD in hypogonadal males . • Observed Higher NAFLD Prevalence in Hypogonadal Males and Baseline Liver Fat % Distribution 1 Kelly et al., Lif Sci 2014 2 Dhindsa et al., Diab Care 2010 3 Kim et al., Gastroenterol 2012 4 Sumida et al., Gastroenterol Hepatol 2015 5 Younossi et al., Hepatol 2016 • Substantial NAFLD Resolution* and Liver Fat Reduction in NAFLD Resolved Subjects CONCLUSION AIM MATERIAL & METHODS DISCLOSURES Dr . Somaya Albhaisi : somaya . albhaisi@vcuhealth . org Dr . Jonathan Baker : jb@lipocine . com or admin@lipocine . com Cont act information • Author disclosures are on file at AASLD . • This study was funded by Lipocine Inc . REFERENCES • Meaningful Liver Fat % Reduction and Substantial Responder Rates* * MRI - PDFF: Magnetic Resonance Imaging - Proton Density Fat Fraction † NAFL identified by MRI - PDFF ≥ 5% in LPCN 1144 Liver Fat Study: N = 21/32 ‡ Prevalence of NAFL diagnosed by imaging hepatosteatosis ≥ 5% liver fat in general population 5 * FAS: Full Analysis Set, LF: Liver Fat, BL: Baseline * Responder rate for relative change is % of patients with at least 30% for relative change from baseline. * Resolution of NAFL is defined as when BL liver fat ≥ 5% is reduced to < 5% at EOS. About 50% of NAFLD Subjects Experienced Resolution at Week 16 SF - 36: Short Form - 36, PDQ: Psychosexual Daily Questionnaire; * Subjects with ALT > 40 U/L at BL (N=33) ▪ Good Gastrointestinal Tolerability ▪ No LDL - C Increase at EOS * LDL - C upper normal limit is 160 mg/dL. • Collateral Health Benefits Observed with LPCN 1144 Therapy* • Well Tolerated Safety with LPCN 1144 Chronic Use Exhibit 99.2