
Exhibit 99.2 Enabling Oral Drug Delivery to Improve Patient Compliance
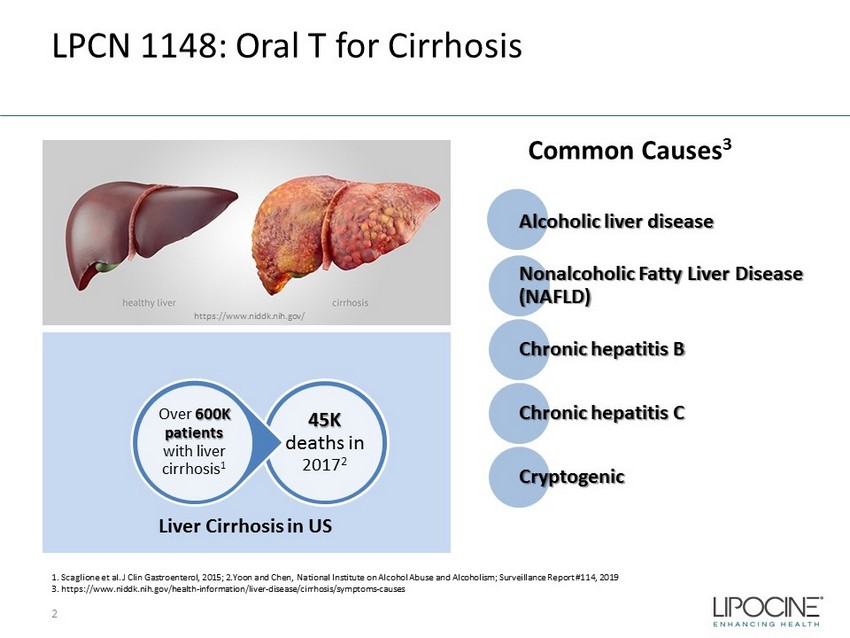
LPCN 1148: Oral T for Cirrhosis 2 Alcoholic liver disease Nonalcoholic Fatty Liver Disease (NAFLD) Chronic hepatitis B Chronic hepatitis C Cryptogenic 45K deaths in 2017 2 Over 600K patients with liver cirrhosis 1 Liver Cirrhosis in US 1. Scaglione et al. J Clin Gastroenterol, 2015; 2.Yoon and Chen, National Institute on Alcohol Abuse and Alcoholism; Surveil lan ce Report #114, 2019 3. https://www.niddk.nih.gov/health - information/liver - disease/cirrhosis/symptoms - causes Common Causes 3 https://www.niddk.nih.gov/

Complications of Cirrhotic Patients 3 Portal hypertension • Varices in esophagus, stomach, or intestines leading to internal bleeding • Edema • Ascites • Hepatic encephalopathy Infections Cachexia, malnutrition, and weight loss Sarcopenia, loss muscle mass and function Frailty with compromised energetics Bone diseases (e.g., osteoporosis) Liver cancer Insulin resistance and type 2 diabetes https://www.niddk.nih.gov/health - information/liver - disease/cirrhosis/definition - facts
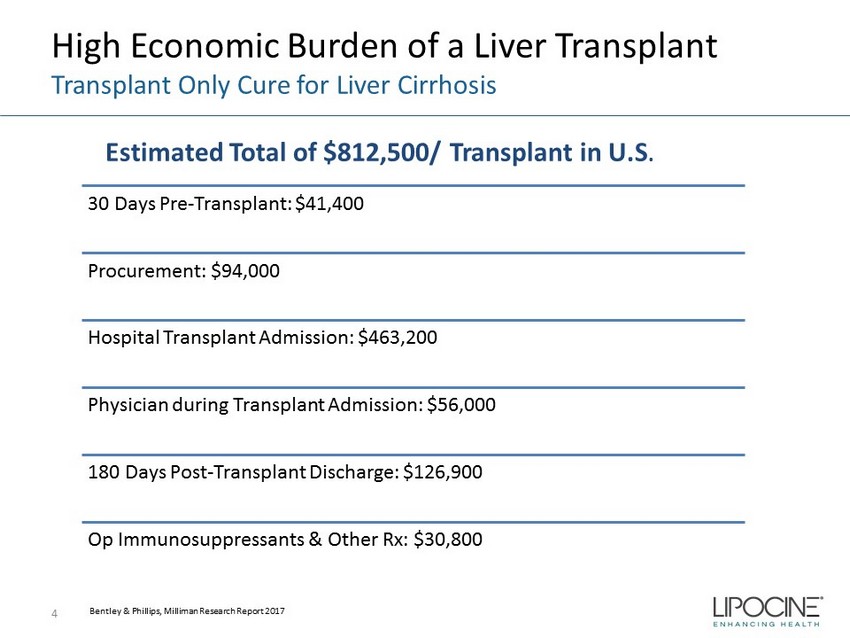
High Economic Burden of a Liver Transplant Transplant Only Cure for Liver Cirrhosis 4 30 Days Pre - Transplant: $41,400 Procurement: $94,000 Hospital Transplant Admission: $463,200 Physician during Transplant Admission: $56,000 180 Days Post - Transplant Discharge: $126,900 Op Immunosuppressants & Other Rx: $30,800 Bentley & Phillips, Milliman Research Report 2017 Estimated Total of $812,500/ Transplant in U.S .
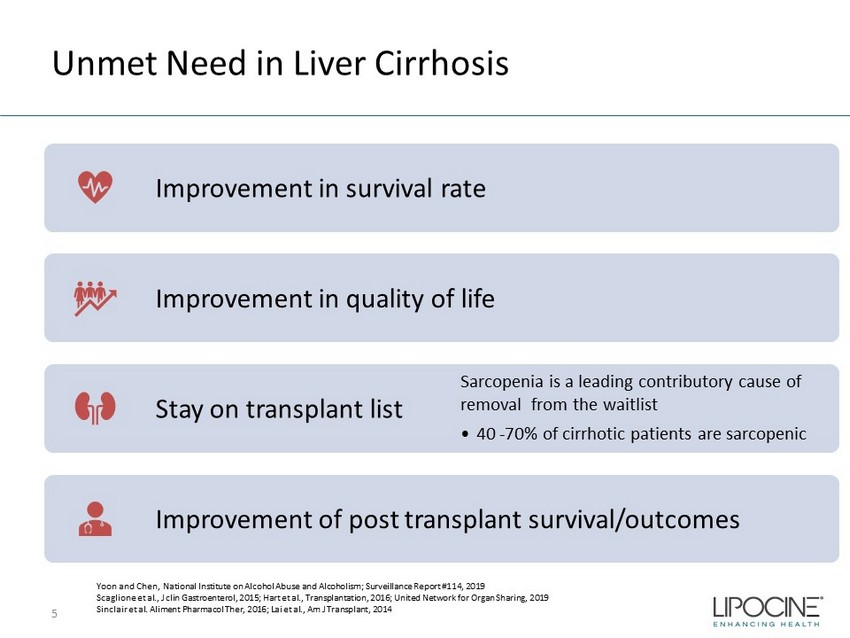
Unmet Need in Liver Cirrhosis 5 Improvement in survival rate Improvement in quality of life Stay on transplant list Sarcopenia is a leading contributory cause of removal from the waitlist • 40 - 70% of cirrhotic patients are sarcopenic Improvement of post transplant survival/outcomes Yoon and Chen, National Institute on Alcohol Abuse and Alcoholism; Surveillance Report #114, 2019 Scaglione et al., J clin Gastroenterol, 2015; Hart et al., Transplantation, 2016; United Network for Organ Sharing, 2019 Sinclair et al. Aliment Pharmacol Ther , 2016; Lai et al., Am J Transplant, 2014
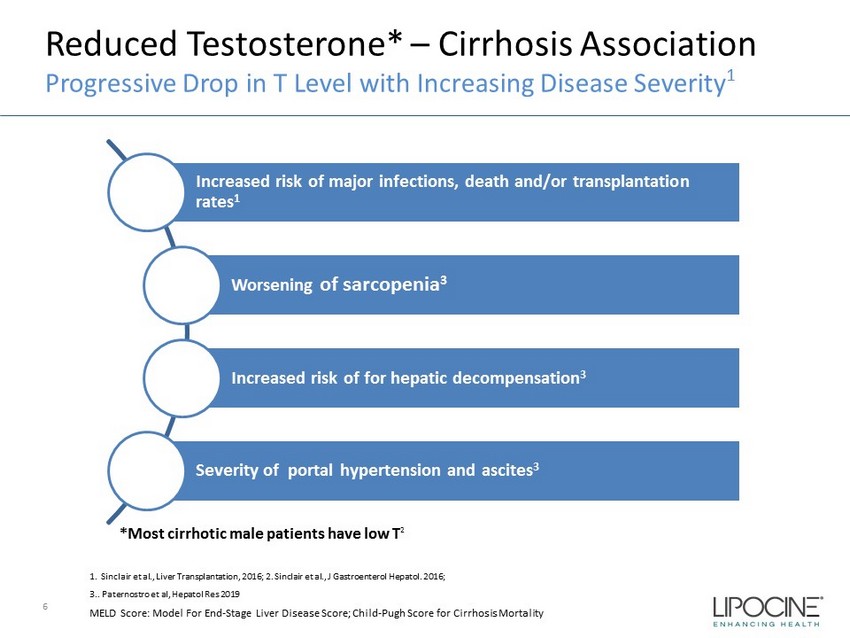
Reduced Testosterone* – Cirrhosis Association Progressive Drop in T Level with Increasing Disease Severity 1 6 Increased risk of major infections, death and/or transplantation rates 1 Worsening of sarcopenia 3 Increased risk of for hepatic decompensation 3 Severity of portal hypertension and ascites 3 1. Sinclair et al., Liver Transplantation, 2016; 2. Sinclair et al., J Gastroenterol Hepatol . 2016; 3.. Paternostro et al, Hepatol Res 2019 MELD Score: Model For End - Stage Liver Disease Score; Child - Pugh Score for Cirrhosis Mortality *Most cirrhotic male patients have low T 2
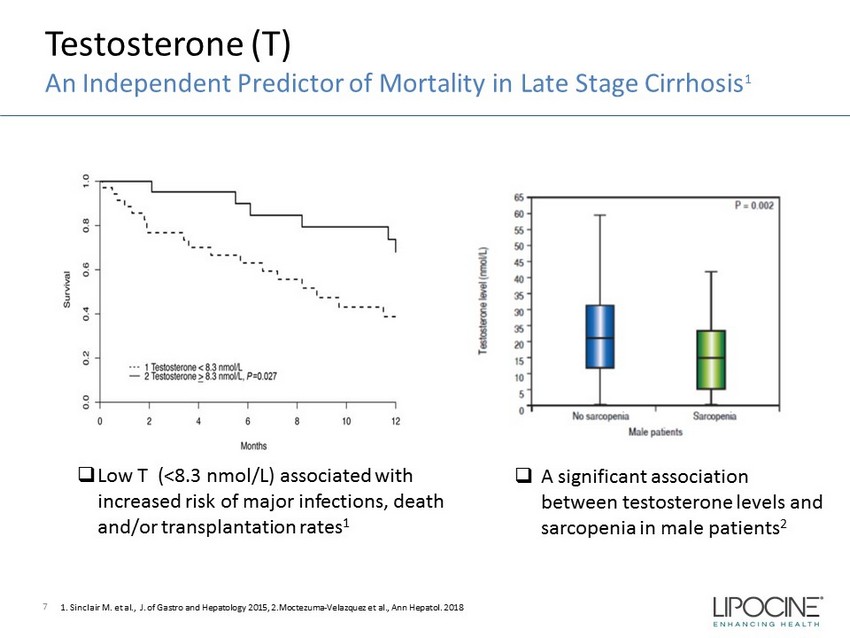
Testosterone (T) An Independent Predictor of Mortality in Late Stage Cirrhosis 1 7 □ Low T (<8.3 nmol/L) associated with increased risk of major infections, death and/or transplantation rates 1 1. Sinclair M. et al., J. of Gastro and Hepatology 2015, 2.Moctezuma - Velazquez et al., Ann Hepatol . 2018 □ A significant association between testosterone levels and sarcopenia in male patients 2
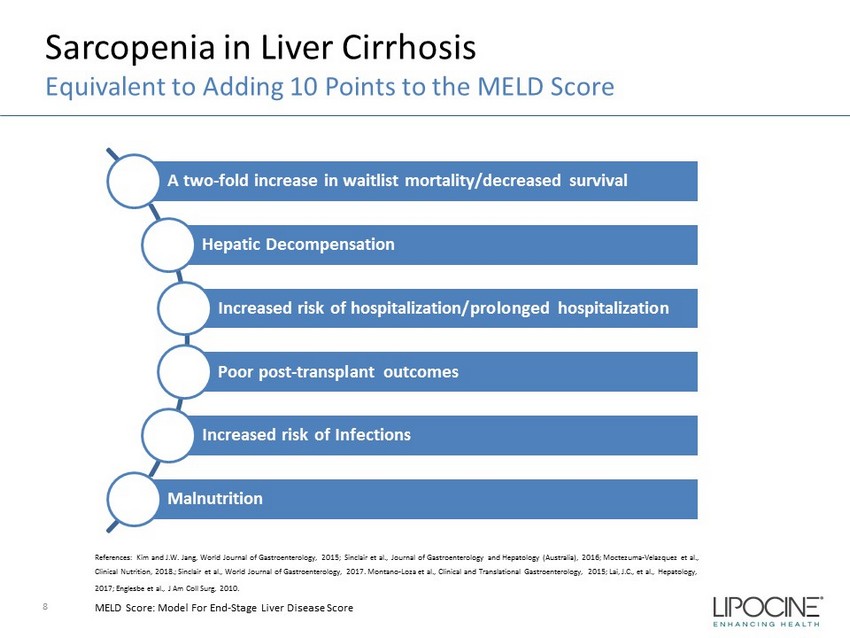
Sarcopenia in Liver Cirrhosis Equivalent to Adding 10 Points to the MELD Score 8 References: Kim and J.W. Jang, World Journal of Gastroenterology, 2015; Sinclair et al., Journal of Gastroenterology and Hep ato logy (Australia), 2016; Moctezuma - Velazquez et al., Clinical Nutrition, 2018.; Sinclair et al., World Journal of Gastroenterology, 2017. Montano - Loza et al., Clinical and Translational Gastroenterology, 2015; Lai, J.C., et al., Hepatology, 2017; Englesbe et al., J Am Coll Surg, 2010. MELD Score: Model For End - Stage Liver Disease Score A two - fold increase in waitlist mortality/decreased survival Hepatic Decompensation Increased risk of hospitalization/prolonged hospitalization Poor post - transplant outcomes Increased risk of Infections Malnutrition

Potential Key Effects of LPCN 1148 in Cirrhotic Patients 9 References: Trivedi and Tapper, Gastroenterol Rep ( Oxf ), 2018; Berzigotti et al., Hepatology, 2017; Chen and Dunn, Clin Liver Dis (Hoboken), 2018; Sinclair et.al, Liver international, 2016; Neff et al., Digestive Diseases and Sciences, 2004; Puliyel et al., Australian and New Zealand Journal of Medicine, 1977; Brown et al., Cleve Clin Q, 1960; Girolami M, Am Geriatr Soc, 1958; Neff et al., Transplant Proc, 2004; Wells R., The Lancet, 1960; Yurci et al., Clinics and Research in Hepatology and Gastroenterology, 2011; Muting D., Verh Dtsch Ges Inn Med, 1969; Gluud C., Liver, 1984. *individual's health condition as it is influenced by the intake and utilization of nutrients Anabolic • Increase muscle mass and strength • Reduce fat mass • Increase bone density • Inhibit myostatin • Improve nutritional status* Androgenic • Induce hematopoiesis • Improve sexual dysfunction Anti - Inflammatory • Reduce IL - 1, IL - 6, and TNF - α Improve immuno - dysregulation • Lower infection rate SHBG lowering • Increase free T

LPCN 1148: Oral Testosterone Therapy Comprising Testosterone Tridecanoate 10 Prodrug of endogenous testosterone (non - methylated) Known anabolic and androgenic agent Preferred route for self - administration

Phase 2: Proof of Concept Study • A Phase 2, randomized double - blind, placebo - controlled, multi - center study to assess the efficacy, safety, and tolerability of a 52 - week treatment period of oral LPCN 1148 in male subjects with cirrhosis of the liver and sarcopenia (with interim results at 24 weeks) 11 11