Exhibit 99.3

IMPACT OF A NEW ORAL TESTOSTERONE UNDECANOATE (TLANDO™) ON BLOOD PRESSURE AND CARDIOVASCULAR RISK MP45 - 14 Presenter: Mohit Khera, MD Professor of Urology Baylor College of Medicine, Houston, TX May 15, 2020

Introduction ▪ Cardiovascular (CV) risk of chronic use of testosterone replacement therapy (TRT) is unclear. Objective ▪ To assess the effects of TLANDO™ ( fixed dose oral TRT) on BP (specifically, SBP changes in CV risk categories based on Framingham Risk Score) in hypogonadal men Methods ▪ A 16 - week, multicenter, single arm, 24 - hr ambulatory blood pressure monitoring (ABPM) study in hypogonadal men (n=138) ▪ 126 completed study and 118 are available for 24 - hr ABPM data BP: Blood Pressure, SBP: Systolic BP
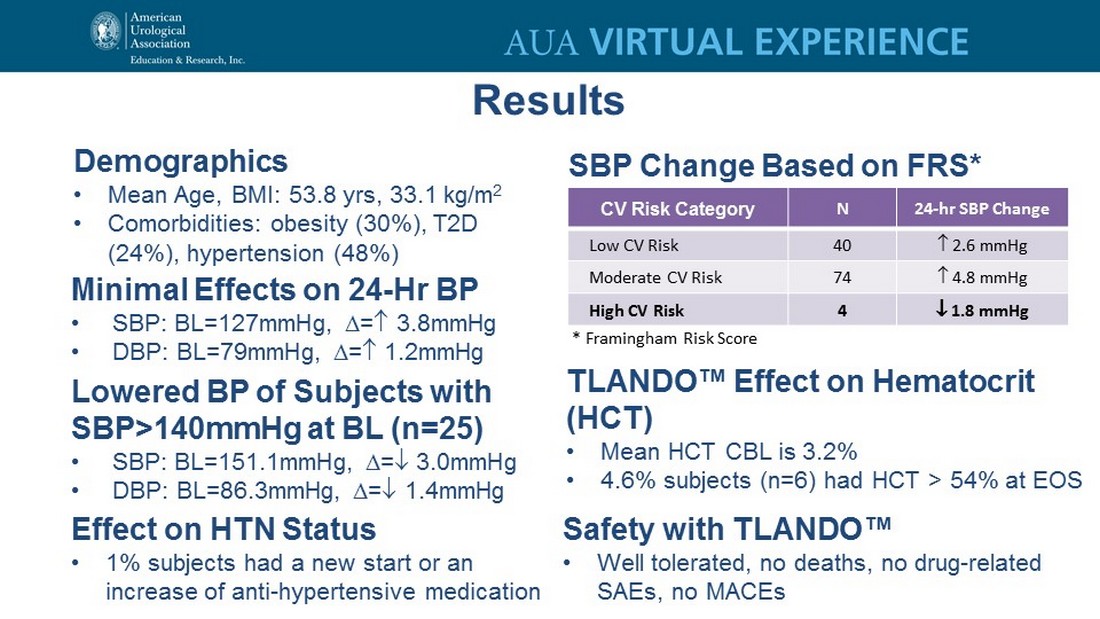
Demographics • Mean Age, BMI: 53.8 yrs , 33.1 kg/m 2 • Comorbidities: obesity (30%), T2D (24%), hypertension (48%) Minimal Effects on 24 - Hr BP • SBP: BL=127mmHg, D = 3.8mmHg • DBP: BL=79mmHg, D = 1.2mmHg Lowered BP of Subjects with SBP>140mmHg at BL (n=25) • SBP: BL=151.1mmHg, D = 3.0mmHg • DBP: BL=86.3mmHg, D = 1.4mmHg Effect on HTN Status • 1% subjects had a new start or an increase of anti - hypertensive medication Safety with TLANDO™ • Well tolerated, no deaths, no drug - related SAEs, no MACEs CV Risk Category N 24 - hr SBP Change Low CV Risk 40 2.6 mmHg Moderate CV Risk 74 4.8 mmHg High CV Risk 4 1.8 mmHg SBP Change Based on FRS* TLANDO™ Effect on Hematocrit (HCT) • Mean HCT CBL is 3.2% • 4.6% subjects (n=6) had HCT > 54% at EOS * Framingham Risk Score Results