
Enabling Oral Drug Delivery to Improve Patient Compliance March 1 - 3, 2021 Raymond James 42nd Annual Institutional Investors Conference Exhibit 99.2

Forward - Looking Statements This presentation contains forward - looking statements about Lipocine Inc . (the “Company”) . These forward - looking statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 . These forward - looking statements relate to the Company’s products and product candidates, FDA’s approval of TLANDO Œ , the expected timing of Phase 3 trials for TLANDO XR and LPCN 1107 and Phase 2 studies for LPCN 1144 and LPCN 1148 , clinical and regulatory processes and objectives, potential benefits of the Company’s product candidates, intellectual property and related matters, all of which involve known and unknown risks and uncertainties . Actual results may differ materially from the forward - looking statements discussed in this presentation . Accordingly, the Company cautions investors not to place undue reliance on the forward - looking statements contained in, or made in connection with, this presentation . Several factors may affect the initiation and completion of clinical trials and studies, the potential advantages of the Company’s product candidates and the Company’s capital needs . The forward - looking statements contained in this presentation are qualified by the detailed discussion of risks and uncertainties set forth in the Company’s annual report on Form 10 - K and other periodic reports filed by the Company with the Securities and Exchange Commission, all of which can be obtained on the Company’s website at www . lipocine . com or on the SEC website at www . sec . gov . The forward - looking statements contained in this document represent the Company’s estimates and assumptions only as of the date of this presentation and the Company undertakes no duty or obligation to update or revise publicly any forward - looking statements contained in this presentation as a result of new information, future events or changes in the Company’s expectations .

Raymond James 42nd Annual Institutional Investors Conference I Clinical Stage Biopharmaceutical Company Innovative Product Candidates for Metabolic and Endocrine Disorders PRODUCT (Indication) PRE - CLINICAL PHASE 1 PHASE 2 PHASE 3 NDA Propriety Drug Delivery Platform TLANDO Œ (Oral Testosterone for Testosterone Replacement Therapy “TRT”) Tentative Approval LPCN 1144 (Oral Testosterone for Non - Cirrhotic NASH ) LiFT Phase 2 Clinical Study Ongoing TLANDO XR (Long Acting Oral Testosterone for Testosterone Replacement Therapy “ TRT” ) Next Step: Food Effect Study LPCN 1148 (Oral Testosterone for Cirrhosis) Next Step: POC Phase 2 Clinical Study LPCN 1107 (Oral HPC for Prevention of PTB) Next Step: Food Effect Study 3

TLANDO ΠThe Convenient TRT without Titration Requirement
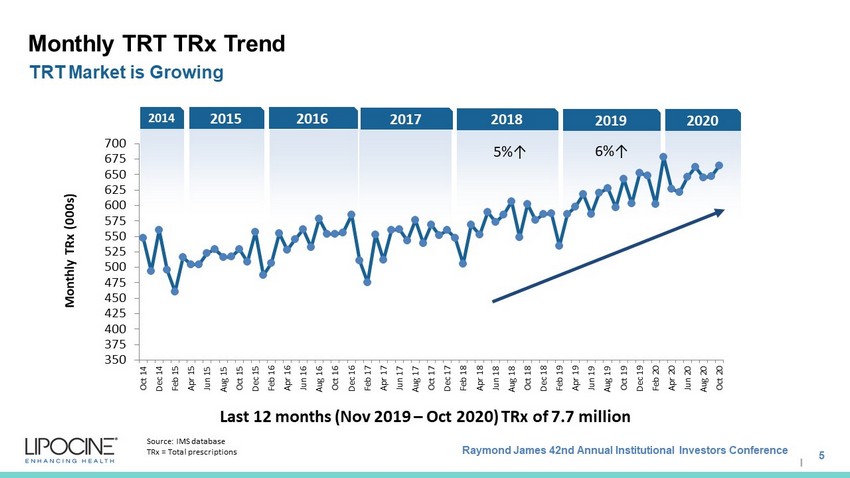
Raymond James 42nd Annual Institutional Investors Conference I 2020 2019 2018 2017 2016 2014 2015 350 375 400 425 450 475 500 525 550 575 600 625 650 675 700 Oct 14 Dec 14 Feb 15 Apr 15 Jun 15 Aug 15 Oct 15 Dec 15 Feb 16 Apr 16 Jun 16 Aug 16 Oct 16 Dec 16 Feb 17 Apr 17 Jun 17 Aug 17 Oct 17 Dec 17 Feb 18 Apr 18 Jun 18 Aug 18 Oct 18 Dec 18 Feb 19 Apr 19 Jun 19 Aug 19 Oct 19 Dec 19 Feb 20 Apr 20 Jun 20 Aug 20 Oct 20 Monthly TRx (000s) Monthly TRT TRx Trend 5 TRT Market is Growing Last 12 months (Nov 2019 – Oct 2020) TRx of 7.7 million Source: IMS database TRx = Total prescriptions 6%↑ 5%↑

Raymond James 42nd Annual Institutional Investors Conference I Issues with Current TRT Options • Black box warning – Secondary exposure to testosterone – Pulmonary oil micro embolism (POME) and anaphylactic shock • Inconvenient application or painful injection • High incidence of erythrocytosis was reported in patients on non - oral – 67% on injectable T, 35% on pellets* • Most require dose titrations - Majority of patients are not on efficacious dose at start of therapy and require up titration – Avg titration time to reach efficacious dose takes 3 - 6 months – Finding appropriate dose through titration is burdensome for physicians and patients – Requires additional clinic/pharmacy visits and invasive samplings • Poor persistence with products requiring titration – 50 - 80% drop off in 3 - 6 months – Insufficient T levels at the beginning of therapy is one of the top reasons patients stop using TRT Potential Barriers To Newly Diagnosed and Existing Patients 6 * Pastuszak et al. Sex Med. 2015

Raymond James 42nd Annual Institutional Investors Conference I f TLANDO Œ Attributes* Convenient Oral Route No inadvertent transference or pulmonary oil micro embolism risks Single strength and dose TRT without titration requirement Enables selection of an effective dose at the start of therapy without delay No “efficacy gap” upon switching from other TRTs No additional pharmacy and clinic copays to reach efficacious dose No dose adjustment clinic and pharmacy visits No dose adjustment invasive samplings No titration decision errors Bioequivalent exposure in low/med/high fat food Not known to produce hepatic adverse events associated with 17 - methylated testosterone 7 Physician Research: Physicians View No Titration Product as Positive ▪ Cited “easy/less titration” as an important advantage of TLANDO Œ ▪ Finding the adequate TRT dose through titration is burdensome for physicians and patients *pending final approval

Raymond James 42nd Annual Institutional Investors Conference I TLANDO: De - Risked Regulatory Profile Opportunities • Post COVID pandemic launch • Adequate time for launch preparation/partnering • Potential exclusivity • Improves TLANDO XR development prospect Eligible to Launch After March 27, 2022 Current Status • Tentative approvals are clearances for drugs to be marketed, but for the existence of patents or market exclusivities • Initial launch supply manufactured • Commercialization partnering process ongoing 8

LPCN 1144 for Non - Cirrhotic NASH Currently No Approved Treatment
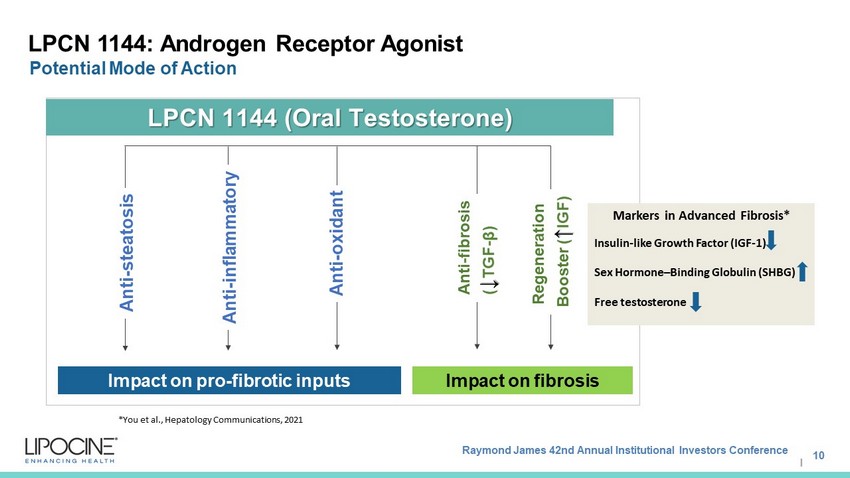
Raymond James 42nd Annual Institutional Investors Conference I LPCN 1144: Androgen Receptor Agonist Potential Mode of Action 10 Impact on pro - fibrotic inputs Anti - steatosis Anti - inflammatory Anti - oxidant LPCN 1144 (Oral Testosterone) Regeneration Booster ( ↑ IGF) Impact on fibrosis Anti - fibrosis ( ↓ TGF - β) *You et al., Hepatology Communications, 2021 Markers in Advanced Fibrosis* Insulin - like G rowth F actor (IGF - 1) Sex Hormone – Binding Globulin (SHBG) Free testosterone
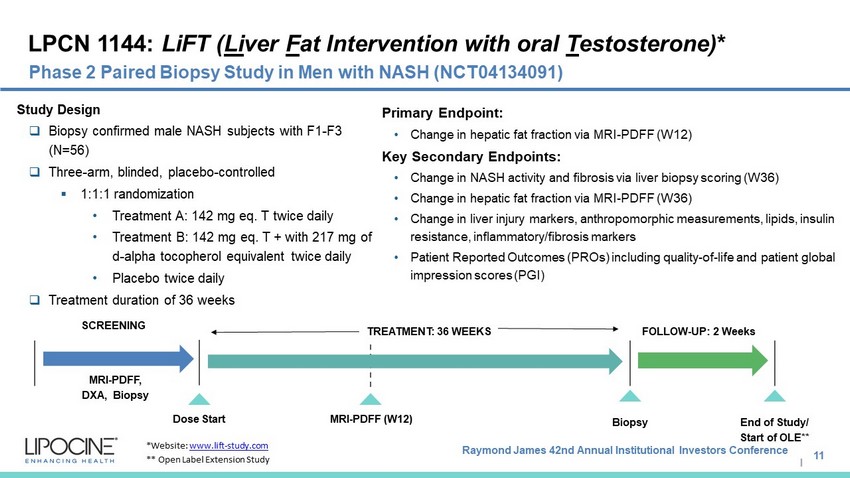
Raymond James 42nd Annual Institutional Investors Conference I LPCN 1144: LiFT ( Li ver F at Intervention with oral T estosterone) * 11 Phase 2 Paired Biopsy Study in Men with NASH (NCT04134091) *Website: www.lift - study.com ** Open Label Extension Study SCREENING MRI - PDFF (W12) Dose Start MRI - PDFF, DXA, Biopsy Study Design □ Biopsy confirmed male NASH subjects with F1 - F3 (N=56) □ Three - arm, blinded, placebo - controlled ▪ 1:1:1 randomization • Treatment A: 142 mg eq. T twice daily • Treatment B: 142 mg eq. T + with 217 mg of d - alpha tocopherol equivalent twice daily • Placebo twice daily □ Treatment duration of 36 weeks 12 Weeks TREATMENT: 36 WEEKS Primary Endpoint: • Change in hepatic fat fraction via MRI - PDFF (W12) Key Secondary Endpoints: • Change in NASH activity and fibrosis via liver biopsy scoring (W36) • Change in hepatic fat fraction via MRI - PDFF (W36) • Change in liver injury markers, anthropomorphic measurements, lipids, insulin resistance, inflammatory/fibrosis markers • Patient Reported Outcomes (PROs) including quality - of - life and patient global impression scores (PGI) Biopsy FOLLOW - UP: 2 Weeks End of Study/ Start of OLE**
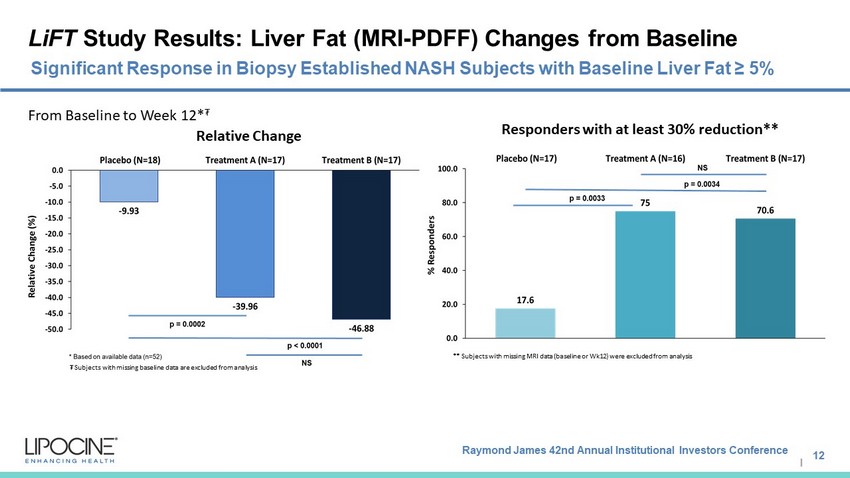
Raymond James 42nd Annual Institutional Investors Conference I LiFT Study Results : Liver Fat (MRI - PDFF) Changes from Baseline 12 Significant Response in Biopsy Established NASH Subjects with Baseline Liver Fat ≥ 5% Relative Change Responders with at least 30% reduction** From Baseline to Week 12* ₮ ₮ Subjects with missing baseline data are excluded from analysis ** Subjects with missing MRI data (baseline or Wk12) were excluded from analysis MB452
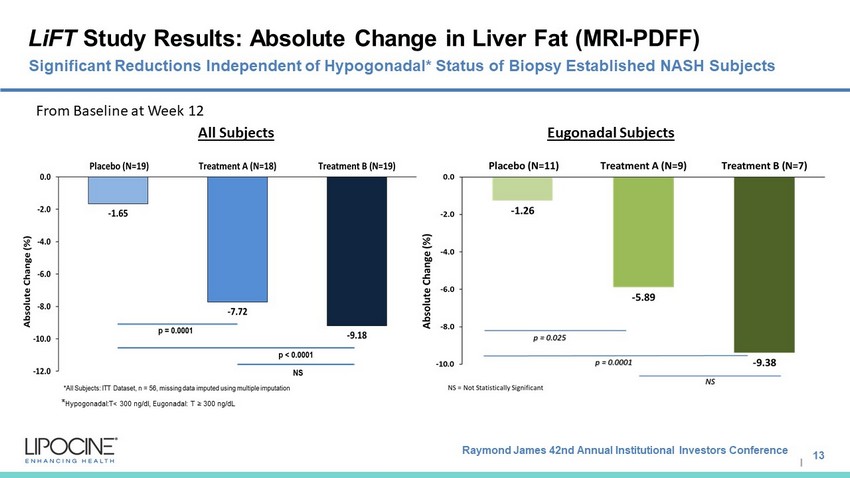
Raymond James 42nd Annual Institutional Investors Conference I LiFT Study Results: Absolute Change in Liver Fat (MRI - PDFF) 13 Significant Reductions Independent of Hypogonadal* Status of Biopsy Established NASH Subjects All Subjects Eugonadal Subjects From Baseline at Week 12 * Hypogonadal:T < 300 ng/dl, Eugonadal : T ≥ 300 ng/dL
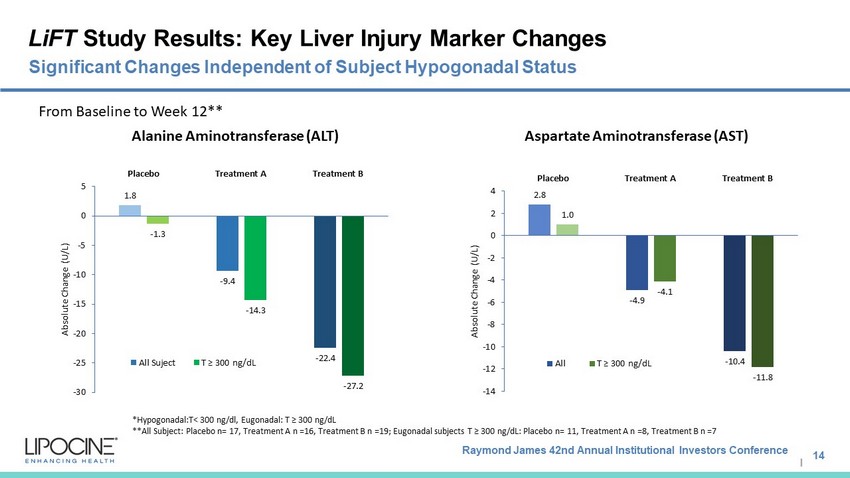
Raymond James 42nd Annual Institutional Investors Conference I LiFT Study Results: Key Liver Injury Marker Changes 14 Significant Changes Independent of Subject Hypogonadal Status * Hypogonadal:T < 300 ng/dl, Eugonadal : T ≥ 300 ng/dL **All Subject: Placebo n= 17, Treatment A n =16, Treatment B n =19; Eugonadal subjects T ≥ 300 ng/dL: Placebo n= 11, Treatment A n =8, Treatment B n =7 2.8 - 4.9 - 10.4 1.0 - 4.1 - 11.8 -14 -12 -10 -8 -6 -4 -2 0 2 4 Placebo Treatment A Treatment B Absolute Change (U/L) All T ≥ 300 ng/dL 1.8 - 9.4 - 22.4 - 1.3 - 14.3 - 27.2 -30 -25 -20 -15 -10 -5 0 5 Placebo Treatment A Treatment B Absolute Change (U/L) All Suject T ≥ 300 ng/dL Alanine Aminotransferase (ALT) Aspartate Aminotransferase (AST) From Baseline to Week 12**
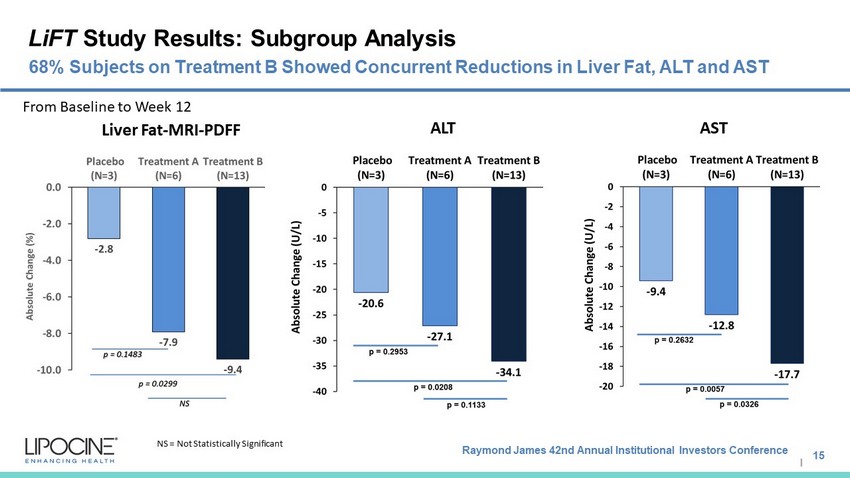
Raymond James 42nd Annual Institutional Investors Conference I LiFT Study Results: Subgroup Analysis 15 68% Subjects on Treatment B Showed Concurrent Reductions in Liver Fat, ALT and AST NS = Not Statistically Significant Liver Fat - MRI - PDFF ALT AST From Baseline to Week 12

Raymond James 42nd Annual Institutional Investors Conference I Summary: Positive Topline Results Statistically significant reduction in liver fat was observed compared to placebo independent of hypogonadal status Statistically significant reduction in markers of liver injury were observed compared to placebo independent of hypogonadal status 68% on Treatment B had concurrent reductions of liver fat, ALT, and AST Adverse events in both the treatment arms were comparable to the placebo arm 16 Both LPCN 1144 Treatment A rms M et the Primary E ndpoint with Statistical S ignificance

Raymond James 42nd Annual Institutional Investors Conference I LiFT Study: Upcoming Data 17 • Histological change in NASH activity and fibrosis at week 36 • MRI - PDFF liver fat data at week 36 • Body composition data from DXA scan (at 20 weeks and 36 weeks) • Liver and other markers including (fibrosis and inflammation markers) at week 36 • PROs for periodic visits up to week 36 – Quality - of - life and patient global impression scores (PGI)

TLANDO XR Once Daily Oral TRT

Raymond James 42nd Annual Institutional Investors Conference I TLANDO XR: An Innovative Opportunity 19 Next Generation TRT Option Strong IP (new molecule & proprietary drug delivery technology) Positive Phase 2b study results Patients and physicians prefer once a day oral testosterone TLANDO XR is positioned to be the first Oral QD Product TLANDO XR is clinically differentiated from other orals

LPCN 1148 for Liver Cirrhosis
Raymond James 42nd Annual Institutional Investors Conference I Common Causes 5 Alcoholic liver disease Nonalcoholic Fatty Liver Disease (NAFLD) Chronic hepatitis B Chronic hepatitis C Cryptogenic LPCN 1148: Oral T for Cirrhosis 21 1. Bentley & Phillips, Milliman Research Report 2017 4. Sarkar et al. J Hepatol. 2015 2. Sinclair et al., Liver Transplantation, 2016 5. https://www.niddk.nih.gov/health - information/liver - disease/cirrhosis/ symptoms - causes 3. Sinclair et al., J Gastroenterol Hepatol . 2016; Transplant Only Cure for Liver Cirrhosis High Economic Burden of a Liver Transplant 1 Liver Cirrhosis in US Healthy Liver Over 600K patients with liver cirrhosis 2 45K deaths in 2017 3 62% male vs. 38% female on the liver transplant (LT) waitlist 4 Background: Prevalence and Common Causes of Cirrhosis Liver with Cirrhosis
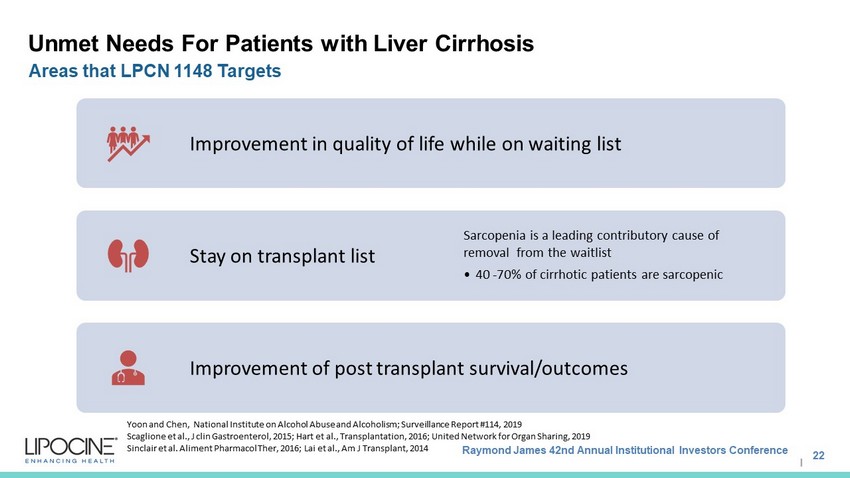
Raymond James 42nd Annual Institutional Investors Conference I Unmet Needs For Patients with Liver Cirrhosis Improvement in quality of life while on waiting list Stay on transplant list Sarcopenia is a leading contributory cause of removal from the waitlist • 40 - 70% of cirrhotic patients are sarcopenic Improvement of post transplant survival/outcomes Yoon and Chen, National Institute on Alcohol Abuse and Alcoholism; Surveillance Report #114, 2019 Scaglione et al., J clin Gastroenterol, 2015; Hart et al., Transplantation, 2016; United Network for Organ Sharing, 2019 Sinclair et al. Aliment Pharmacol Ther , 2016; Lai et al., Am J Transplant, 2014 22 Areas that LPCN 1148 Targets

LPCN 1107 Prevention of Preterm Birth

Raymond James 42nd Annual Institutional Investors Conference I Unmet Medical Need • One Preterm birth (PTB) every minute 1 • ≥ $26 billion economic impact 2 • 10% of all US pregnancies 3 • A leading cause of neonatal mortality and morbidity • Medical costs for PTB infants are ~10x higher than for full - term infants 4 24 Preterm Birth is a Significant Public Health Issue in the U.S 1. Pediatric Research (2006) 60, 775 – 776 2. Institute of Medicine of the National Academies. Jul.200 3. National Vital Statistics Reports, Vol 67, No. 8, Nov 7, 2018: Centers for Disease Control and Prevention (2017) 4. J. Maternal - Fetal and Neonatal Medicine, Dec. 2006, 19(12), 773 – 782

Raymond James 42nd Annual Institutional Investors Conference I LPCN 1107: Potential To Be The Standard of Care for Prevention of PTB 25 Oral Hydroxyprogesterone Caproate (HPC) Convenient dosing No injection site reactions First and only oral enabling technology Solid IP coverage Orphan drug designated Next Step: Food Effect Study

Raymond James 42nd Annual Institutional Investors Conference I Upcoming Milestones Near Term Value Drivers Event Expected Timing TLANDO ΠFinal FDA Approval Decision March 2022 LPCN 1144 LiFT Biopsy Results July/August 2021 26
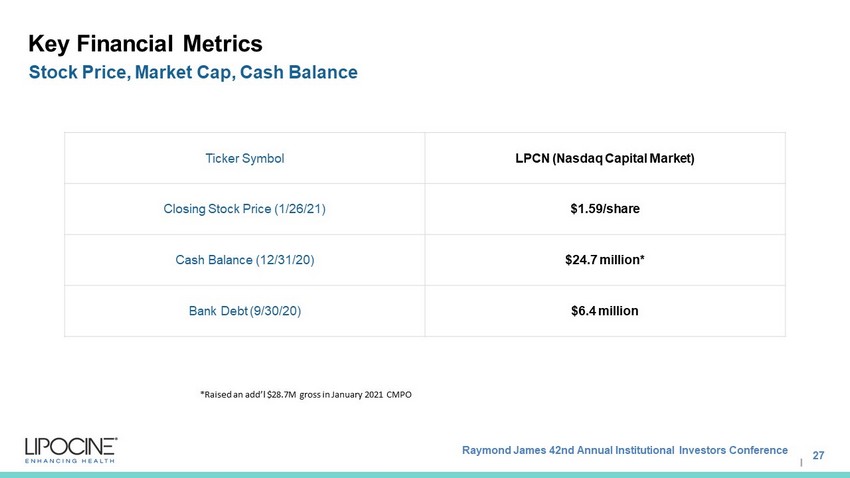
Raymond James 42nd Annual Institutional Investors Conference I Key Financial Metrics 27 Stock Price, Market Cap, Cash Balance Ticker Symbol LPCN (Nasdaq Capital Market) Closing Stock Price (1/26/21) $1.59/share Cash Balance (12/31/20) $24.7 million* Bank Debt (9/30/20) $6.4 million *Raised an add’l $28.7M gross in January 2021 CMPO